JP2017018012A - 歯肉炎・歯周炎の簡易判定方法及びその簡易判定キット - Google Patents
歯肉炎・歯周炎の簡易判定方法及びその簡易判定キット Download PDFInfo
- Publication number
- JP2017018012A JP2017018012A JP2015136596A JP2015136596A JP2017018012A JP 2017018012 A JP2017018012 A JP 2017018012A JP 2015136596 A JP2015136596 A JP 2015136596A JP 2015136596 A JP2015136596 A JP 2015136596A JP 2017018012 A JP2017018012 A JP 2017018012A
- Authority
- JP
- Japan
- Prior art keywords
- gingivitis
- periodontitis
- color tone
- test paper
- patients
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 208000007565 gingivitis Diseases 0.000 title claims abstract description 70
- 201000001245 periodontitis Diseases 0.000 title claims abstract description 68
- 238000000034 method Methods 0.000 title claims abstract description 15
- 238000012360 testing method Methods 0.000 claims abstract description 64
- 239000000758 substrate Substances 0.000 claims abstract description 38
- 102000003855 L-lactate dehydrogenase Human genes 0.000 claims abstract description 31
- 108700023483 L-lactate dehydrogenases Proteins 0.000 claims abstract description 31
- 210000003296 saliva Anatomy 0.000 claims abstract description 27
- 239000005515 coenzyme Substances 0.000 claims abstract description 22
- 238000009826 distribution Methods 0.000 claims abstract description 15
- 235000019646 color tone Nutrition 0.000 claims description 52
- 102000004190 Enzymes Human genes 0.000 claims description 24
- 108090000790 Enzymes Proteins 0.000 claims description 24
- 239000007864 aqueous solution Substances 0.000 claims description 23
- 125000003831 tetrazolyl group Chemical group 0.000 claims description 22
- 239000003381 stabilizer Substances 0.000 claims description 21
- 239000006174 pH buffer Substances 0.000 claims description 19
- 239000003086 colorant Substances 0.000 claims description 12
- JVTAAEKCZFNVCJ-UHFFFAOYSA-N lactic acid Chemical compound CC(O)C(O)=O JVTAAEKCZFNVCJ-UHFFFAOYSA-N 0.000 claims description 12
- HCAONSFIGUBAPW-UHFFFAOYSA-N methyl hydrogen sulfate;phenazine Chemical class COS(O)(=O)=O.C1=CC=CC2=NC3=CC=CC=C3N=C21 HCAONSFIGUBAPW-UHFFFAOYSA-N 0.000 claims description 9
- 239000004310 lactic acid Substances 0.000 claims description 6
- 235000014655 lactic acid Nutrition 0.000 claims description 6
- 238000001035 drying Methods 0.000 claims description 5
- 239000000463 material Substances 0.000 claims description 5
- 238000002791 soaking Methods 0.000 claims description 2
- 101710088194 Dehydrogenase Proteins 0.000 claims 3
- 239000002250 absorbent Substances 0.000 abstract 1
- 239000000243 solution Substances 0.000 description 17
- 208000028169 periodontal disease Diseases 0.000 description 16
- 230000000694 effects Effects 0.000 description 13
- 208000005888 Periodontal Pocket Diseases 0.000 description 8
- 239000000523 sample Substances 0.000 description 8
- 210000000214 mouth Anatomy 0.000 description 7
- BAWFJGJZGIEFAR-NNYOXOHSSA-N NAD zwitterion Chemical compound NC(=O)C1=CC=C[N+]([C@H]2[C@@H]([C@H](O)[C@@H](COP([O-])(=O)OP(O)(=O)OC[C@@H]3[C@H]([C@@H](O)[C@@H](O3)N3C4=NC=NC(N)=C4N=C3)O)O2)O)=C1 BAWFJGJZGIEFAR-NNYOXOHSSA-N 0.000 description 6
- 229950006238 nadide Drugs 0.000 description 6
- 238000004040 coloring Methods 0.000 description 4
- 238000003745 diagnosis Methods 0.000 description 4
- JUJBNYBVVQSIOU-UHFFFAOYSA-M sodium;4-[2-(4-iodophenyl)-3-(4-nitrophenyl)tetrazol-2-ium-5-yl]benzene-1,3-disulfonate Chemical compound [Na+].C1=CC([N+](=O)[O-])=CC=C1N1[N+](C=2C=CC(I)=CC=2)=NC(C=2C(=CC(=CC=2)S([O-])(=O)=O)S([O-])(=O)=O)=N1 JUJBNYBVVQSIOU-UHFFFAOYSA-M 0.000 description 4
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 4
- 201000010099 disease Diseases 0.000 description 3
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 3
- 229910003002 lithium salt Inorganic materials 0.000 description 3
- 159000000002 lithium salts Chemical class 0.000 description 3
- QKNYBSVHEMOAJP-UHFFFAOYSA-N 2-amino-2-(hydroxymethyl)propane-1,3-diol;hydron;chloride Chemical compound Cl.OCC(N)(CO)CO QKNYBSVHEMOAJP-UHFFFAOYSA-N 0.000 description 2
- 108091003079 Bovine Serum Albumin Proteins 0.000 description 2
- DHMQDGOQFOQNFH-UHFFFAOYSA-N Glycine Chemical compound NCC(O)=O DHMQDGOQFOQNFH-UHFFFAOYSA-N 0.000 description 2
- 206010061218 Inflammation Diseases 0.000 description 2
- SEQKRHFRPICQDD-UHFFFAOYSA-N N-tris(hydroxymethyl)methylglycine Chemical compound OCC(CO)(CO)[NH2+]CC([O-])=O SEQKRHFRPICQDD-UHFFFAOYSA-N 0.000 description 2
- DFPAKSUCGFBDDF-UHFFFAOYSA-N Nicotinamide Chemical compound NC(=O)C1=CC=CN=C1 DFPAKSUCGFBDDF-UHFFFAOYSA-N 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 2
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 2
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 2
- XSQUKJJJFZCRTK-UHFFFAOYSA-N Urea Chemical compound NC(N)=O XSQUKJJJFZCRTK-UHFFFAOYSA-N 0.000 description 2
- 229940098773 bovine serum albumin Drugs 0.000 description 2
- 239000000872 buffer Substances 0.000 description 2
- 239000007853 buffer solution Substances 0.000 description 2
- DDRJAANPRJIHGJ-UHFFFAOYSA-N creatinine Chemical compound CN1CC(=O)NC1=N DDRJAANPRJIHGJ-UHFFFAOYSA-N 0.000 description 2
- 238000002405 diagnostic procedure Methods 0.000 description 2
- ZUOUZKKEUPVFJK-UHFFFAOYSA-N diphenyl Chemical group C1=CC=CC=C1C1=CC=CC=C1 ZUOUZKKEUPVFJK-UHFFFAOYSA-N 0.000 description 2
- 239000012153 distilled water Substances 0.000 description 2
- 210000004195 gingiva Anatomy 0.000 description 2
- 230000004054 inflammatory process Effects 0.000 description 2
- GKQWYZBANWAFMQ-UHFFFAOYSA-M lithium;2-hydroxypropanoate Chemical compound [Li+].CC(O)C([O-])=O GKQWYZBANWAFMQ-UHFFFAOYSA-M 0.000 description 2
- 239000006179 pH buffering agent Substances 0.000 description 2
- 230000003239 periodontal effect Effects 0.000 description 2
- 238000010340 saliva test Methods 0.000 description 2
- 239000006228 supernatant Substances 0.000 description 2
- VVJYUAYZJAKGRQ-UHFFFAOYSA-N 1-[4,5-dihydroxy-6-(hydroxymethyl)oxan-2-yl]-5-methylpyrimidine-2,4-dione Chemical compound O=C1NC(=O)C(C)=CN1C1OC(CO)C(O)C(O)C1 VVJYUAYZJAKGRQ-UHFFFAOYSA-N 0.000 description 1
- MASUWVVNWALEEM-UHFFFAOYSA-M 1-methoxy-5-methylphenazin-5-ium;methyl sulfate Chemical compound COS([O-])(=O)=O.C1=CC=C2N=C3C(OC)=CC=CC3=[N+](C)C2=C1 MASUWVVNWALEEM-UHFFFAOYSA-M 0.000 description 1
- GVEDOIATHPCYGS-UHFFFAOYSA-N 1-methyl-3-(3-methylphenyl)benzene Chemical group CC1=CC=CC(C=2C=C(C)C=CC=2)=C1 GVEDOIATHPCYGS-UHFFFAOYSA-N 0.000 description 1
- GZCWLCBFPRFLKL-UHFFFAOYSA-N 1-prop-2-ynoxypropan-2-ol Chemical compound CC(O)COCC#C GZCWLCBFPRFLKL-UHFFFAOYSA-N 0.000 description 1
- NBWRJAOOMGASJP-UHFFFAOYSA-N 2-(3,5-diphenyl-1h-tetrazol-1-ium-2-yl)-4,5-dimethyl-1,3-thiazole;bromide Chemical compound [Br-].S1C(C)=C(C)N=C1N1N(C=2C=CC=CC=2)N=C(C=2C=CC=CC=2)[NH2+]1 NBWRJAOOMGASJP-UHFFFAOYSA-N 0.000 description 1
- JKMHFZQWWAIEOD-UHFFFAOYSA-N 2-[4-(2-hydroxyethyl)piperazin-1-yl]ethanesulfonic acid Chemical compound OCC[NH+]1CCN(CCS([O-])(=O)=O)CC1 JKMHFZQWWAIEOD-UHFFFAOYSA-N 0.000 description 1
- JJMQRJKPLUACSO-UHFFFAOYSA-N 3-(4-iodophenyl)-2-(4-nitrophenyl)-5-phenyl-1,3-dihydrotetrazol-3-ium;chloride Chemical compound [Cl-].C1=CC([N+](=O)[O-])=CC=C1N1N(C=2C=CC(I)=CC=2)[NH2+]C(C=2C=CC=CC=2)=N1 JJMQRJKPLUACSO-UHFFFAOYSA-N 0.000 description 1
- CYDQOEWLBCCFJZ-UHFFFAOYSA-N 4-(4-fluorophenyl)oxane-4-carboxylic acid Chemical compound C=1C=C(F)C=CC=1C1(C(=O)O)CCOCC1 CYDQOEWLBCCFJZ-UHFFFAOYSA-N 0.000 description 1
- 102000013563 Acid Phosphatase Human genes 0.000 description 1
- 108010051457 Acid Phosphatase Proteins 0.000 description 1
- 102100036475 Alanine aminotransferase 1 Human genes 0.000 description 1
- 108010082126 Alanine transaminase Proteins 0.000 description 1
- 102000002260 Alkaline Phosphatase Human genes 0.000 description 1
- 108020004774 Alkaline Phosphatase Proteins 0.000 description 1
- 239000004251 Ammonium lactate Substances 0.000 description 1
- 108010003415 Aspartate Aminotransferases Proteins 0.000 description 1
- 102000004625 Aspartate Aminotransferases Human genes 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 1
- 241000766026 Coregonus nasus Species 0.000 description 1
- 208000006558 Dental Calculus Diseases 0.000 description 1
- 102000003779 Dipeptidyl-peptidases and tripeptidyl-peptidases Human genes 0.000 description 1
- 108090000194 Dipeptidyl-peptidases and tripeptidyl-peptidases Proteins 0.000 description 1
- 108091006149 Electron carriers Proteins 0.000 description 1
- 108020004206 Gamma-glutamyltransferase Proteins 0.000 description 1
- 239000004471 Glycine Substances 0.000 description 1
- 239000006173 Good's buffer Substances 0.000 description 1
- 239000007995 HEPES buffer Substances 0.000 description 1
- OWXMKDGYPWMGEB-UHFFFAOYSA-N HEPPS Chemical compound OCCN1CCN(CCCS(O)(=O)=O)CC1 OWXMKDGYPWMGEB-UHFFFAOYSA-N 0.000 description 1
- 239000007996 HEPPS buffer Substances 0.000 description 1
- 102000001554 Hemoglobins Human genes 0.000 description 1
- 108010054147 Hemoglobins Proteins 0.000 description 1
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 1
- JVTAAEKCZFNVCJ-UHFFFAOYSA-M Lactate Chemical compound CC(O)C([O-])=O JVTAAEKCZFNVCJ-UHFFFAOYSA-M 0.000 description 1
- VAYOSLLFUXYJDT-RDTXWAMCSA-N Lysergic acid diethylamide Chemical compound C1=CC(C=2[C@H](N(C)C[C@@H](C=2)C(=O)N(CC)CC)C2)=C3C2=CNC3=C1 VAYOSLLFUXYJDT-RDTXWAMCSA-N 0.000 description 1
- 208000001145 Metabolic Syndrome Diseases 0.000 description 1
- 102000005741 Metalloproteases Human genes 0.000 description 1
- 108010006035 Metalloproteases Proteins 0.000 description 1
- FSVCELGFZIQNCK-UHFFFAOYSA-N N,N-bis(2-hydroxyethyl)glycine Chemical compound OCCN(CCO)CC(O)=O FSVCELGFZIQNCK-UHFFFAOYSA-N 0.000 description 1
- 206010028851 Necrosis Diseases 0.000 description 1
- 208000001132 Osteoporosis Diseases 0.000 description 1
- 206010035664 Pneumonia Diseases 0.000 description 1
- 208000005107 Premature Birth Diseases 0.000 description 1
- 206010036590 Premature baby Diseases 0.000 description 1
- KEAYESYHFKHZAL-UHFFFAOYSA-N Sodium Chemical class [Na] KEAYESYHFKHZAL-UHFFFAOYSA-N 0.000 description 1
- UZMAPBJVXOGOFT-UHFFFAOYSA-N Syringetin Natural products COC1=C(O)C(OC)=CC(C2=C(C(=O)C3=C(O)C=C(O)C=C3O2)O)=C1 UZMAPBJVXOGOFT-UHFFFAOYSA-N 0.000 description 1
- 208000008312 Tooth Loss Diseases 0.000 description 1
- 239000007997 Tricine buffer Substances 0.000 description 1
- 208000034953 Twin anemia-polycythemia sequence Diseases 0.000 description 1
- 201000000690 abdominal obesity-metabolic syndrome Diseases 0.000 description 1
- 239000006096 absorbing agent Substances 0.000 description 1
- 238000009825 accumulation Methods 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 1
- 229940059265 ammonium lactate Drugs 0.000 description 1
- 235000019286 ammonium lactate Nutrition 0.000 description 1
- RZOBLYBZQXQGFY-HSHFZTNMSA-N azanium;(2r)-2-hydroxypropanoate Chemical compound [NH4+].C[C@@H](O)C([O-])=O RZOBLYBZQXQGFY-HSHFZTNMSA-N 0.000 description 1
- 239000007998 bicine buffer Substances 0.000 description 1
- 235000010290 biphenyl Nutrition 0.000 description 1
- 230000000740 bleeding effect Effects 0.000 description 1
- 210000000988 bone and bone Anatomy 0.000 description 1
- KGBXLFKZBHKPEV-UHFFFAOYSA-N boric acid Chemical compound OB(O)O KGBXLFKZBHKPEV-UHFFFAOYSA-N 0.000 description 1
- 239000004327 boric acid Substances 0.000 description 1
- 230000001680 brushing effect Effects 0.000 description 1
- 230000003139 buffering effect Effects 0.000 description 1
- 239000004202 carbamide Substances 0.000 description 1
- 210000004027 cell Anatomy 0.000 description 1
- 230000005779 cell damage Effects 0.000 description 1
- 208000037887 cell injury Diseases 0.000 description 1
- 238000005119 centrifugation Methods 0.000 description 1
- 238000004140 cleaning Methods 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 229940109239 creatinine Drugs 0.000 description 1
- 210000000805 cytoplasm Anatomy 0.000 description 1
- 238000001514 detection method Methods 0.000 description 1
- 206010012601 diabetes mellitus Diseases 0.000 description 1
- 238000010586 diagram Methods 0.000 description 1
- ZBCBWPMODOFKDW-UHFFFAOYSA-N diethanolamine Chemical compound OCCNCCO ZBCBWPMODOFKDW-UHFFFAOYSA-N 0.000 description 1
- KCFYHBSOLOXZIF-UHFFFAOYSA-N dihydrochrysin Natural products COC1=C(O)C(OC)=CC(C2OC3=CC(O)=CC(O)=C3C(=O)C2)=C1 KCFYHBSOLOXZIF-UHFFFAOYSA-N 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 239000011888 foil Substances 0.000 description 1
- 102000006640 gamma-Glutamyltransferase Human genes 0.000 description 1
- 210000005260 human cell Anatomy 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- 229910052739 hydrogen Inorganic materials 0.000 description 1
- 229940001447 lactate Drugs 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- OVGXLJDWSLQDRT-UHFFFAOYSA-L magnesium lactate Chemical compound [Mg+2].CC(O)C([O-])=O.CC(O)C([O-])=O OVGXLJDWSLQDRT-UHFFFAOYSA-L 0.000 description 1
- 239000000626 magnesium lactate Substances 0.000 description 1
- 235000015229 magnesium lactate Nutrition 0.000 description 1
- 229960004658 magnesium lactate Drugs 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 239000003550 marker Substances 0.000 description 1
- 239000011159 matrix material Substances 0.000 description 1
- 230000003340 mental effect Effects 0.000 description 1
- 208000010125 myocardial infarction Diseases 0.000 description 1
- 230000017074 necrotic cell death Effects 0.000 description 1
- 229960003966 nicotinamide Drugs 0.000 description 1
- 235000005152 nicotinamide Nutrition 0.000 description 1
- 239000011570 nicotinamide Substances 0.000 description 1
- 229930027945 nicotinamide-adenine dinucleotide Natural products 0.000 description 1
- 239000004745 nonwoven fabric Substances 0.000 description 1
- 125000000636 p-nitrophenyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1*)[N+]([O-])=O 0.000 description 1
- 206010034674 peritonitis Diseases 0.000 description 1
- 239000012466 permeate Substances 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- PHZLMBHDXVLRIX-UHFFFAOYSA-M potassium lactate Chemical compound [K+].CC(O)C([O-])=O PHZLMBHDXVLRIX-UHFFFAOYSA-M 0.000 description 1
- 239000001521 potassium lactate Substances 0.000 description 1
- 235000011085 potassium lactate Nutrition 0.000 description 1
- 229960001304 potassium lactate Drugs 0.000 description 1
- 230000002265 prevention Effects 0.000 description 1
- 230000002250 progressing effect Effects 0.000 description 1
- 230000000284 resting effect Effects 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- 235000017557 sodium bicarbonate Nutrition 0.000 description 1
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 1
- 229910000029 sodium carbonate Inorganic materials 0.000 description 1
- 239000001540 sodium lactate Substances 0.000 description 1
- 235000011088 sodium lactate Nutrition 0.000 description 1
- 229940005581 sodium lactate Drugs 0.000 description 1
- 159000000000 sodium salts Chemical class 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 238000001356 surgical procedure Methods 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- 238000009210 therapy by ultrasound Methods 0.000 description 1
- 210000001519 tissue Anatomy 0.000 description 1
Images
Landscapes
- Measuring Or Testing Involving Enzymes Or Micro-Organisms (AREA)
Abstract
Description
−nitrophenyl)−5−phenyl−2H−tetrazoliumchloride(以下、INTという)、3−(4,5−Dimethyl−2−thiazolyl)−2,5−diphenyl−2H−tetrazoliumbromide、2−(4−Iodophenyl)−3−(4−nitrophenyl)−5−(2,4−disulfophenyl)−2H−tetrazolium,monosodiumsalt(以下、WST−1という)、2−(4−Iodophenyl)−3−(2,4−dinitrophenyl)−5−(2,4−disulfophenyl)−2H−tetrazolium,monosodiumsalt等が好適に用いられる。テトラゾリウム塩はLDH活性試験紙用発色液中の最終濃度が0.05〜20mg/mLとなるように添加することが望ましい。
また、吸水性基材には通常の濾紙、書道用吸い取り紙、高分子吸収体、不織布などが用いられる。
〔唾液検査の方法〕
46人の成人被験者に水でうがいをさせた後、1分間後に安静時唾液を約100μL、アルミホイル上に吐出させた。試験紙を唾液に浸した後に遮光し、1分間後の色の濃さを10種類の濃度の色見本の中で最も近いものから選んだ。なお、色の濃さは10が最も濃く、1が最も薄いものとした。
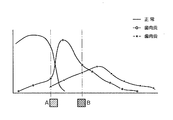
各被験者の全ての歯を対象に、歯周ポケットプローブを用いて、1歯に対して6ヶ所を検査し、プロービング後の出血の有無及び歯周ポケットの深さから、正常、歯肉炎、軽度歯周炎、中度歯周炎及び重度歯周炎を評価し、試験紙呈色の色調と正常、歯肉炎、軽度歯周炎、中度歯周炎及び重度歯周炎の関係を求め、図1に示される関係図を得た。
本例の簡易判定キットを用いると、正常、歯肉炎、歯周炎の簡易判定が可能であった。また、簡易判定を行った患者、あるいは歯石の除去を行った患者に、ブラッシングを指導してプラークコントロールを行い、2週間の経過後、再度試験紙を用いて簡易判定を行ったところ、病状の改善が見られること、あるいは病状が進行していることを簡易判定することができた。
Claims (4)
- 乳酸脱水素酵素基質、乳酸脱水素酵素補酵素、ジアホラーゼ、テトラゾリウム塩、酵素安定化剤およびpH緩衝剤を含むpH7〜11の水溶液が吸水性基材に浸透され乾燥されてなる試験紙、又は乳酸脱水素酵素基質、乳酸脱水素酵素補酵素、Phenaziniummethylsulfate誘導体、テトラゾリウム塩、酵素安定化剤およびpH緩衝剤を含むpH7〜11の水溶液が吸水性基材に浸透され乾燥されてなる試験紙を用い、
正常者、歯肉炎患者及び歯周炎患者の唾液を試験紙に浸して一定時間の経過したときの試験紙呈色の色調に対する正常者数の分布、歯肉炎患者及び歯周炎患者の各患者数の分布を求め、
正常者の最大数の色調と歯肉炎患者の最大数の色調との間の中間の色調を第1境界値とし、歯肉炎患者の最大数の色調と歯周炎患者の最大数の色調との間の中間の色調を第2境界値とし、
患者の唾液を試験紙に浸して一定時間経過後の呈色が第1境界値を超える色調の患者を歯肉炎と、第2境界値を超える色調の患者を歯周炎と簡易判定するようにしたことを特徴とする歯肉炎・歯周炎の簡易判定方法。 - 乳酸脱水素酵素基質、乳酸脱水素酵素補酵素、ジアホラーゼ、テトラゾリウム塩、酵素安定化剤およびpH緩衝剤を含むpH7〜11の水溶液が吸水性基材に浸透され乾燥されてなる試験紙、又は乳酸脱水素酵素基質、乳酸脱水素酵素補酵素、Phenaziniummethylsulfate誘導体、テトラゾリウム塩、酵素安定化剤およびpH緩衝剤を含むpH7〜11の水溶液が吸水性基材に浸透され乾燥されてなる試験紙を用い、
正常者、歯肉炎患者及び歯周炎患者の唾液を試験紙に浸して一定時間の経過したときの試験紙呈色の色調に対する正常者数の分布、歯肉炎患者及び歯周炎患者の各患者数の分布における、正常者の最大数の色調と歯肉炎患者の最大数の色調との間の中間の色調を第1境界値とし、歯肉炎患者の最大数の色調と歯周炎患者の最大数の色調との間の中間の色調を第2境界値としたとき、患者の唾液を試験紙に浸して一定時間経過後の呈色が第1境界値を超える色調の患者を歯肉炎と、第2境界値を超える色調の患者を歯周炎と簡易判定するようにしたことを特徴とする歯肉炎・歯周炎の簡易判定方法。 - 上記簡易判定を行った患者の唾液を試験紙に浸して一定時間経過後の呈色が第1境界値又は第2境界値を超える色調から正常者又は第1境界値の色調に変化したときに歯肉炎又は歯周炎が改善され、第1境界値を超える色調から第2境界値を超える色調に変化したときには歯肉炎が悪化したと簡易判定するようにした請求項1又は2記載の歯肉炎・歯周炎の簡易判定方法。
- 乳酸脱水素酵素基質、乳酸脱水素酵素補酵素、ジアホラーゼ、テトラゾリウム塩、酵素安定化剤およびpH緩衝剤を含むpH7〜11の水溶液が吸水性基材に浸透され乾燥されてなる試験紙、又は乳酸脱水素酵素基質、乳酸脱水素酵素補酵素、Phenaziniummethylsulfate誘導体、テトラゾリウム塩、酵素安定化剤およびpH緩衝剤を含むpH7〜11の水溶液が吸水性基材に浸透され乾燥されてなる試験紙と、
正常者、歯肉炎患者及び歯周炎患者の唾液を、乳酸脱水素酵素基質、乳酸脱水素酵素補酵素、ジアホラーゼ、テトラゾリウム塩、酵素安定化剤およびpH緩衝剤を含むpH7〜11の水溶液が吸水性基材に浸透され乾燥されてなる試験紙、又は乳酸脱水素酵素基質、乳酸脱水素酵素補酵素、Phenaziniummethylsulfate誘導体、テトラゾリウム塩、酵素安定化剤およびpH緩衝剤を含むpH7〜11の水溶液が吸水性基材に浸透され乾燥されてなる試験紙に浸して一定時間の経過したときの試験紙呈色の色調に対する正常者数の分布、歯肉炎患者及び歯周炎患者の各患者数の分布における、正常者の最大数の色調と歯肉炎患者の最大数の色調との間の中間の色調を第1境界値とし、歯肉炎患者の最大数の色調と歯周炎患者の最大数の色調との間の中間の色調を第2境界値としたとき、患者の唾液を試験紙に浸して一定時間経過後の呈色が第1境界値を超える色調の患者を歯肉炎と、第2境界値を超える色調の患者を歯周炎と簡易判定する第1境界値の色調及び第2境界値の色調を示す色見本と、を備えたことを特徴とする歯肉炎・歯周炎の簡易判定キット。
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2015136596A JP6480278B2 (ja) | 2015-07-08 | 2015-07-08 | 歯肉炎・歯周炎の簡易判定補助方法 |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2015136596A JP6480278B2 (ja) | 2015-07-08 | 2015-07-08 | 歯肉炎・歯周炎の簡易判定補助方法 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| JP2017018012A true JP2017018012A (ja) | 2017-01-26 |
| JP2017018012A5 JP2017018012A5 (ja) | 2018-02-22 |
| JP6480278B2 JP6480278B2 (ja) | 2019-03-06 |
Family
ID=57887007
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2015136596A Active JP6480278B2 (ja) | 2015-07-08 | 2015-07-08 | 歯肉炎・歯周炎の簡易判定補助方法 |
Country Status (1)
| Country | Link |
|---|---|
| JP (1) | JP6480278B2 (ja) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN111226118A (zh) * | 2017-10-27 | 2020-06-02 | 加利福尼亚大学董事会 | 用于牙龈炎的唾液细胞外rna生物标记物 |
| CN111830023A (zh) * | 2019-08-19 | 2020-10-27 | 杭州爱光医疗器械有限公司 | 巯基化合物检测试剂、检测试纸、试剂盒、试纸盒及其制备方法 |
Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1990004787A1 (en) * | 1988-10-26 | 1990-05-03 | The Board Of Trustees Of The University Of Illinois | Method for determination of the type and severity of periodontal disease states |
| JP2004229537A (ja) * | 2003-01-29 | 2004-08-19 | Bml Inc | 歯周疾患の検出方法 |
| WO2005001484A1 (ja) * | 2003-06-30 | 2005-01-06 | Applied Cell Biotechnologies, Inc. | 歯周炎の発症を判定する方法 |
| JP2005201768A (ja) * | 2004-01-15 | 2005-07-28 | Bml Inc | 歯周疾患の検出方法 |
| JP2010130924A (ja) * | 2008-12-03 | 2010-06-17 | Nagata Sangyo Kk | 歯周病診断試験紙 |
| JP2010256190A (ja) * | 2009-04-24 | 2010-11-11 | Kao Corp | 歯周病マーカー |
| JP2011130924A (ja) * | 2009-12-24 | 2011-07-07 | Unicharm Corp | 排泄検出装置及び吸収性物品 |
| WO2012090995A1 (ja) * | 2010-12-28 | 2012-07-05 | ライオン株式会社 | 口腔状態の判定方法、並びにそのために用いられる分析用具、装置、及びプログラム |
-
2015
- 2015-07-08 JP JP2015136596A patent/JP6480278B2/ja active Active
Patent Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1990004787A1 (en) * | 1988-10-26 | 1990-05-03 | The Board Of Trustees Of The University Of Illinois | Method for determination of the type and severity of periodontal disease states |
| JPH03502890A (ja) * | 1988-10-26 | 1991-07-04 | ザ ボード オブ トラスティーズ オブ ザ ユニバーシティ オブ イリノイ | 歯周疾患状態の型および程度の決定法 |
| JP2004229537A (ja) * | 2003-01-29 | 2004-08-19 | Bml Inc | 歯周疾患の検出方法 |
| WO2005001484A1 (ja) * | 2003-06-30 | 2005-01-06 | Applied Cell Biotechnologies, Inc. | 歯周炎の発症を判定する方法 |
| JP2005201768A (ja) * | 2004-01-15 | 2005-07-28 | Bml Inc | 歯周疾患の検出方法 |
| JP2010130924A (ja) * | 2008-12-03 | 2010-06-17 | Nagata Sangyo Kk | 歯周病診断試験紙 |
| JP2010256190A (ja) * | 2009-04-24 | 2010-11-11 | Kao Corp | 歯周病マーカー |
| JP2011130924A (ja) * | 2009-12-24 | 2011-07-07 | Unicharm Corp | 排泄検出装置及び吸収性物品 |
| WO2012090995A1 (ja) * | 2010-12-28 | 2012-07-05 | ライオン株式会社 | 口腔状態の判定方法、並びにそのために用いられる分析用具、装置、及びプログラム |
Non-Patent Citations (1)
| Title |
|---|
| 日歯周誌, vol. 33, no. 1, JPN6018041476, 1991, pages 154 - 163, ISSN: 0003903746 * |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN111226118A (zh) * | 2017-10-27 | 2020-06-02 | 加利福尼亚大学董事会 | 用于牙龈炎的唾液细胞外rna生物标记物 |
| US12077824B2 (en) | 2017-10-27 | 2024-09-03 | Colgate-Palmolive Company | Salivary extracellular RNA biomarkers for gingivitis |
| CN111830023A (zh) * | 2019-08-19 | 2020-10-27 | 杭州爱光医疗器械有限公司 | 巯基化合物检测试剂、检测试纸、试剂盒、试纸盒及其制备方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| JP6480278B2 (ja) | 2019-03-06 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Agrawal et al. | Noninvasive method for glucose level estimation by saliva | |
| Cho‐Yan Lee et al. | Residual periodontal pockets are a risk indicator for peri‐implantitis in patients treated for periodontitis | |
| Santos et al. | Comparison between conventional and chemomechanical approaches for the removal of carious dentin: an in vitro study | |
| Borghi et al. | Relationship among α amylase and carbonic anhydrase VI in saliva, visible biofilm, and early childhood caries: a longitudinal study | |
| El Masry | Salivary carbonic anhydrase, pH and phosphate buffer concentrations as potential biomarkers of caries risk in children | |
| US20030100842A1 (en) | Method and kit for indicating the level of bad breath | |
| JP2010130924A (ja) | 歯周病診断試験紙 | |
| Pallos et al. | Salivary microbial dysbiosis is associated with peri-implantitis: a case-control study in a Brazilian population | |
| Persson et al. | A multicenter clinical trial of PerioGard™ in distinguishing between diseased and healthy periodontal sites:(I). Study design, methodology and therapeutic outcome. | |
| JP6480278B2 (ja) | 歯肉炎・歯周炎の簡易判定補助方法 | |
| Hannig et al. | Transaminases in the acquired pellicle | |
| Parihar et al. | Comparative evaluation of salivary, serum, and GCF alkaline phosphatase levels in chronic periodontitis patients before and after nonsurgical periodontal therapy: A clinico-biochemical study | |
| Szkaradkiewicz-Karpinska et al. | Salivary glucose levels and pH in healthy adults with dental caries | |
| Bachtiar et al. | Relationships between Solobacterium moorei and Prevotella intermedia in subgingival microbiota of periodontitis patients with halitosis: A preliminary study using qPCR | |
| Sheth et al. | Analysis of aspartate aminotransferase in gingival crevicular fluid: A study with initial therapy | |
| Fathima et al. | Evaluation of alkaline phosphatase in gingival crevicular fluid among chronic periodontitis patients with smoking habit | |
| Cheaib et al. | Comparison of three strip-type tests and two laboratory methods for salivary buffering analysis | |
| JP4792585B2 (ja) | 口腔細菌の迅速検出方法 | |
| Lee et al. | Distinguishing discolored caries lesions using biofluorescence and dental bleaching: An in vitro simulation model study | |
| Menassa et al. | Periodontal diagnosis: current status and future directions | |
| WO2000065033A1 (en) | Method and kit for indicating the level of bad breath | |
| Sterer et al. | Effect of chemomechanical excavation (CarisolvTM) on residual cariogenic bacteria | |
| Sharma | Evaluation of serum and salivary alkaline phosphatase levels in chronic periodontitis patients | |
| QIZI et al. | ASSOCIATION BETWEEN SALIVARY BIOMARKERS AND CARIES RISK ASSESSMENT IN ADULTS: A CLINICAL OBSERVATIONAL STUDY | |
| RU2764672C2 (ru) | Способ диагностики жизнеспособности пульпы при лечении начального пульпита |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20180115 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20180115 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20181017 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20181023 |
|
| RD02 | Notification of acceptance of power of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: A7422 Effective date: 20181126 |
|
| RD04 | Notification of resignation of power of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: A7424 Effective date: 20181204 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A821 Effective date: 20181126 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20181219 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20190115 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20190207 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 6480278 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |