JP2017178902A - Chlorine removing agent and external preparation for skin - Google Patents
Chlorine removing agent and external preparation for skin Download PDFInfo
- Publication number
- JP2017178902A JP2017178902A JP2016072574A JP2016072574A JP2017178902A JP 2017178902 A JP2017178902 A JP 2017178902A JP 2016072574 A JP2016072574 A JP 2016072574A JP 2016072574 A JP2016072574 A JP 2016072574A JP 2017178902 A JP2017178902 A JP 2017178902A
- Authority
- JP
- Japan
- Prior art keywords
- chlorine
- extract
- skin
- gemmifera
- sumawiki
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 title claims abstract description 61
- 239000000460 chlorine Substances 0.000 title claims abstract description 61
- 229910052801 chlorine Inorganic materials 0.000 title claims abstract description 61
- 239000003795 chemical substances by application Substances 0.000 title claims abstract description 14
- 238000002360 preparation method Methods 0.000 title claims abstract description 12
- 239000000284 extract Substances 0.000 claims abstract description 40
- 240000007124 Brassica oleracea Species 0.000 claims description 19
- 235000011303 Brassica alboglabra Nutrition 0.000 claims description 15
- 235000011302 Brassica oleracea Nutrition 0.000 claims description 15
- 239000002537 cosmetic Substances 0.000 claims description 9
- 239000013040 bath agent Substances 0.000 claims description 2
- 239000003599 detergent Substances 0.000 claims description 2
- 230000009885 systemic effect Effects 0.000 claims 1
- 230000000694 effects Effects 0.000 abstract description 15
- 239000008399 tap water Substances 0.000 abstract description 7
- 235000020679 tap water Nutrition 0.000 abstract description 7
- 235000004221 Brassica oleracea var gemmifera Nutrition 0.000 abstract description 3
- 244000308368 Brassica oleracea var. gemmifera Species 0.000 abstract description 3
- 230000003699 hair surface Effects 0.000 abstract description 2
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 17
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 16
- 238000011156 evaluation Methods 0.000 description 11
- 239000000419 plant extract Substances 0.000 description 10
- 239000000243 solution Substances 0.000 description 9
- PUPZLCDOIYMWBV-UHFFFAOYSA-N (+/-)-1,3-Butanediol Chemical compound CC(O)CCO PUPZLCDOIYMWBV-UHFFFAOYSA-N 0.000 description 8
- 229960005070 ascorbic acid Drugs 0.000 description 8
- 238000000605 extraction Methods 0.000 description 8
- 239000000203 mixture Substances 0.000 description 8
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 7
- 238000000034 method Methods 0.000 description 7
- 239000002904 solvent Substances 0.000 description 7
- 238000012360 testing method Methods 0.000 description 7
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 6
- 239000002211 L-ascorbic acid Substances 0.000 description 6
- 235000000069 L-ascorbic acid Nutrition 0.000 description 6
- 239000005708 Sodium hypochlorite Substances 0.000 description 6
- POULHZVOKOAJMA-UHFFFAOYSA-N dodecanoic acid Chemical compound CCCCCCCCCCCC(O)=O POULHZVOKOAJMA-UHFFFAOYSA-N 0.000 description 6
- SUKJFIGYRHOWBL-UHFFFAOYSA-N sodium hypochlorite Chemical compound [Na+].Cl[O-] SUKJFIGYRHOWBL-UHFFFAOYSA-N 0.000 description 6
- 239000007787 solid Substances 0.000 description 6
- 235000003899 Brassica oleracea var acephala Nutrition 0.000 description 5
- 241000196324 Embryophyta Species 0.000 description 5
- 235000019441 ethanol Nutrition 0.000 description 5
- 238000004659 sterilization and disinfection Methods 0.000 description 5
- 229940058015 1,3-butylene glycol Drugs 0.000 description 4
- 235000019437 butane-1,3-diol Nutrition 0.000 description 4
- 230000000052 comparative effect Effects 0.000 description 4
- 238000009472 formulation Methods 0.000 description 4
- -1 hypochlorite ions Chemical class 0.000 description 4
- 238000002156 mixing Methods 0.000 description 4
- 239000000843 powder Substances 0.000 description 4
- ZWEHNKRNPOVVGH-UHFFFAOYSA-N 2-Butanone Chemical compound CCC(C)=O ZWEHNKRNPOVVGH-UHFFFAOYSA-N 0.000 description 3
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 3
- 235000011331 Brassica Nutrition 0.000 description 3
- 241000219198 Brassica Species 0.000 description 3
- 235000012905 Brassica oleracea var viridis Nutrition 0.000 description 3
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 3
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 3
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 3
- 239000005639 Lauric acid Substances 0.000 description 3
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 3
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 3
- 239000007864 aqueous solution Substances 0.000 description 3
- 239000003814 drug Substances 0.000 description 3
- 235000011187 glycerol Nutrition 0.000 description 3
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 3
- MWUXSHHQAYIFBG-UHFFFAOYSA-N nitrogen oxide Inorganic materials O=[N] MWUXSHHQAYIFBG-UHFFFAOYSA-N 0.000 description 3
- 239000003755 preservative agent Substances 0.000 description 3
- 230000002335 preservative effect Effects 0.000 description 3
- 239000008213 purified water Substances 0.000 description 3
- 230000001954 sterilising effect Effects 0.000 description 3
- 230000001629 suppression Effects 0.000 description 3
- 208000024891 symptom Diseases 0.000 description 3
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 2
- 235000011301 Brassica oleracea var capitata Nutrition 0.000 description 2
- 235000001169 Brassica oleracea var oleracea Nutrition 0.000 description 2
- WWZKQHOCKIZLMA-UHFFFAOYSA-N Caprylic acid Natural products CCCCCCCC(O)=O WWZKQHOCKIZLMA-UHFFFAOYSA-N 0.000 description 2
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 2
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 2
- 206010012438 Dermatitis atopic Diseases 0.000 description 2
- 239000003463 adsorbent Substances 0.000 description 2
- 150000001298 alcohols Chemical class 0.000 description 2
- 239000003963 antioxidant agent Substances 0.000 description 2
- 235000006708 antioxidants Nutrition 0.000 description 2
- 235000010323 ascorbic acid Nutrition 0.000 description 2
- 239000011668 ascorbic acid Substances 0.000 description 2
- 201000008937 atopic dermatitis Diseases 0.000 description 2
- 239000003788 bath preparation Substances 0.000 description 2
- 238000004140 cleaning Methods 0.000 description 2
- 238000007796 conventional method Methods 0.000 description 2
- 239000006071 cream Substances 0.000 description 2
- GHVNFZFCNZKVNT-UHFFFAOYSA-N decanoic acid Chemical compound CCCCCCCCCC(O)=O GHVNFZFCNZKVNT-UHFFFAOYSA-N 0.000 description 2
- 229930195733 hydrocarbon Natural products 0.000 description 2
- 150000002430 hydrocarbons Chemical class 0.000 description 2
- WQYVRQLZKVEZGA-UHFFFAOYSA-N hypochlorite Inorganic materials Cl[O-] WQYVRQLZKVEZGA-UHFFFAOYSA-N 0.000 description 2
- QWPPOHNGKGFGJK-UHFFFAOYSA-N hypochlorous acid Chemical compound ClO QWPPOHNGKGFGJK-UHFFFAOYSA-N 0.000 description 2
- 238000007654 immersion Methods 0.000 description 2
- 230000002401 inhibitory effect Effects 0.000 description 2
- 239000007788 liquid Substances 0.000 description 2
- 238000005259 measurement Methods 0.000 description 2
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 description 2
- 239000003921 oil Substances 0.000 description 2
- 230000001590 oxidative effect Effects 0.000 description 2
- 239000003002 pH adjusting agent Substances 0.000 description 2
- 102000004169 proteins and genes Human genes 0.000 description 2
- 108090000623 proteins and genes Proteins 0.000 description 2
- 239000011734 sodium Substances 0.000 description 2
- VBICKXHEKHSIBG-UHFFFAOYSA-N 1-monostearoylglycerol Chemical compound CCCCCCCCCCCCCCCCCC(=O)OCC(O)CO VBICKXHEKHSIBG-UHFFFAOYSA-N 0.000 description 1
- RZRNAYUHWVFMIP-KTKRTIGZSA-N 1-oleoylglycerol Chemical compound CCCCCCCC\C=C/CCCCCCCC(=O)OCC(O)CO RZRNAYUHWVFMIP-KTKRTIGZSA-N 0.000 description 1
- TWJNQYPJQDRXPH-UHFFFAOYSA-N 2-cyanobenzohydrazide Chemical compound NNC(=O)C1=CC=CC=C1C#N TWJNQYPJQDRXPH-UHFFFAOYSA-N 0.000 description 1
- KWIUHFFTVRNATP-UHFFFAOYSA-N Betaine Natural products C[N+](C)(C)CC([O-])=O KWIUHFFTVRNATP-UHFFFAOYSA-N 0.000 description 1
- 240000004073 Brassica oleracea var. viridis Species 0.000 description 1
- 239000005632 Capric acid (CAS 334-48-5) Substances 0.000 description 1
- 239000005635 Caprylic acid (CAS 124-07-2) Substances 0.000 description 1
- 239000004215 Carbon black (E152) Substances 0.000 description 1
- QDHHCQZDFGDHMP-UHFFFAOYSA-N Chloramine Chemical compound ClN QDHHCQZDFGDHMP-UHFFFAOYSA-N 0.000 description 1
- 208000035473 Communicable disease Diseases 0.000 description 1
- FCKYPQBAHLOOJQ-UHFFFAOYSA-N Cyclohexane-1,2-diaminetetraacetic acid Chemical compound OC(=O)CN(CC(O)=O)C1CCCCC1N(CC(O)=O)CC(O)=O FCKYPQBAHLOOJQ-UHFFFAOYSA-N 0.000 description 1
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 1
- QZKRHPLGUJDVAR-UHFFFAOYSA-K EDTA trisodium salt Chemical compound [Na+].[Na+].[Na+].OC(=O)CN(CC([O-])=O)CCN(CC([O-])=O)CC([O-])=O QZKRHPLGUJDVAR-UHFFFAOYSA-K 0.000 description 1
- FPVVYTCTZKCSOJ-UHFFFAOYSA-N Ethylene glycol distearate Chemical compound CCCCCCCCCCCCCCCCCC(=O)OCCOC(=O)CCCCCCCCCCCCCCCCC FPVVYTCTZKCSOJ-UHFFFAOYSA-N 0.000 description 1
- 239000004354 Hydroxyethyl cellulose Substances 0.000 description 1
- 229920000663 Hydroxyethyl cellulose Polymers 0.000 description 1
- 150000000996 L-ascorbic acids Chemical class 0.000 description 1
- 239000004909 Moisturizer Substances 0.000 description 1
- 235000021360 Myristic acid Nutrition 0.000 description 1
- TUNFSRHWOTWDNC-UHFFFAOYSA-N Myristic acid Natural products CCCCCCCCCCCCCC(O)=O TUNFSRHWOTWDNC-UHFFFAOYSA-N 0.000 description 1
- KWIUHFFTVRNATP-UHFFFAOYSA-O N,N,N-trimethylglycinium Chemical compound C[N+](C)(C)CC(O)=O KWIUHFFTVRNATP-UHFFFAOYSA-O 0.000 description 1
- SUZRRICLUFMAQD-UHFFFAOYSA-N N-Methyltaurine Chemical compound CNCCS(O)(=O)=O SUZRRICLUFMAQD-UHFFFAOYSA-N 0.000 description 1
- 229920001213 Polysorbate 20 Polymers 0.000 description 1
- 208000003251 Pruritus Diseases 0.000 description 1
- 206010040880 Skin irritation Diseases 0.000 description 1
- 229920002385 Sodium hyaluronate Polymers 0.000 description 1
- 235000021355 Stearic acid Nutrition 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 description 1
- PHYFQTYBJUILEZ-UHFFFAOYSA-N Trioleoylglycerol Natural products CCCCCCCCC=CCCCCCCCC(=O)OCC(OC(=O)CCCCCCCC=CCCCCCCCC)COC(=O)CCCCCCCC=CCCCCCCCC PHYFQTYBJUILEZ-UHFFFAOYSA-N 0.000 description 1
- 241000700605 Viruses Species 0.000 description 1
- 238000002835 absorbance Methods 0.000 description 1
- 239000006096 absorbing agent Substances 0.000 description 1
- 239000008351 acetate buffer Substances 0.000 description 1
- 150000001335 aliphatic alkanes Chemical class 0.000 description 1
- 125000005907 alkyl ester group Chemical group 0.000 description 1
- 150000001412 amines Chemical class 0.000 description 1
- 150000001413 amino acids Chemical class 0.000 description 1
- 230000003712 anti-aging effect Effects 0.000 description 1
- 239000004599 antimicrobial Substances 0.000 description 1
- 230000003078 antioxidant effect Effects 0.000 description 1
- 238000003287 bathing Methods 0.000 description 1
- 229960003237 betaine Drugs 0.000 description 1
- 210000000170 cell membrane Anatomy 0.000 description 1
- 210000002421 cell wall Anatomy 0.000 description 1
- 239000012459 cleaning agent Substances 0.000 description 1
- MRUAUOIMASANKQ-UHFFFAOYSA-N cocamidopropyl betaine Chemical compound CCCCCCCCCCCC(=O)NCCC[N+](C)(C)CC([O-])=O MRUAUOIMASANKQ-UHFFFAOYSA-N 0.000 description 1
- 229940073507 cocamidopropyl betaine Drugs 0.000 description 1
- 238000013329 compounding Methods 0.000 description 1
- 230000000249 desinfective effect Effects 0.000 description 1
- JSYGRUBHOCKMGQ-UHFFFAOYSA-N dichloramine Chemical compound ClNCl JSYGRUBHOCKMGQ-UHFFFAOYSA-N 0.000 description 1
- 230000037213 diet Effects 0.000 description 1
- 235000005911 diet Nutrition 0.000 description 1
- 238000007598 dipping method Methods 0.000 description 1
- 239000002552 dosage form Substances 0.000 description 1
- 229940079593 drug Drugs 0.000 description 1
- 239000000975 dye Substances 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 238000003912 environmental pollution Methods 0.000 description 1
- 239000010696 ester oil Substances 0.000 description 1
- 150000002170 ethers Chemical class 0.000 description 1
- SFNALCNOMXIBKG-UHFFFAOYSA-N ethylene glycol monododecyl ether Chemical compound CCCCCCCCCCCCOCCO SFNALCNOMXIBKG-UHFFFAOYSA-N 0.000 description 1
- MJEMIOXXNCZZFK-UHFFFAOYSA-N ethylone Chemical compound CCNC(C)C(=O)C1=CC=C2OCOC2=C1 MJEMIOXXNCZZFK-UHFFFAOYSA-N 0.000 description 1
- 230000005713 exacerbation Effects 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 230000001815 facial effect Effects 0.000 description 1
- 238000001914 filtration Methods 0.000 description 1
- 239000003063 flame retardant Substances 0.000 description 1
- 235000013305 food Nutrition 0.000 description 1
- 239000003205 fragrance Substances 0.000 description 1
- 125000003976 glyceryl group Chemical group [H]C([*])([H])C(O[H])([H])C(O[H])([H])[H] 0.000 description 1
- 229940075529 glyceryl stearate Drugs 0.000 description 1
- 229940100608 glycol distearate Drugs 0.000 description 1
- 239000003676 hair preparation Substances 0.000 description 1
- IIRDTKBZINWQAW-UHFFFAOYSA-N hexaethylene glycol Chemical compound OCCOCCOCCOCCOCCOCCO IIRDTKBZINWQAW-UHFFFAOYSA-N 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- 229910052739 hydrogen Inorganic materials 0.000 description 1
- 235000019447 hydroxyethyl cellulose Nutrition 0.000 description 1
- 230000001771 impaired effect Effects 0.000 description 1
- 230000007794 irritation Effects 0.000 description 1
- 230000005722 itchiness Effects 0.000 description 1
- 230000007803 itching Effects 0.000 description 1
- 229940119170 jojoba wax Drugs 0.000 description 1
- 150000002576 ketones Chemical class 0.000 description 1
- 239000006210 lotion Substances 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 229910044991 metal oxide Inorganic materials 0.000 description 1
- 150000004706 metal oxides Chemical class 0.000 description 1
- 244000005700 microbiome Species 0.000 description 1
- 239000012046 mixed solvent Substances 0.000 description 1
- 230000001333 moisturizer Effects 0.000 description 1
- 230000003472 neutralizing effect Effects 0.000 description 1
- 102000039446 nucleic acids Human genes 0.000 description 1
- 108020004707 nucleic acids Proteins 0.000 description 1
- 150000007523 nucleic acids Chemical class 0.000 description 1
- OQCDKBAXFALNLD-UHFFFAOYSA-N octadecanoic acid Natural products CCCCCCCC(C)CCCCCCCCC(O)=O OQCDKBAXFALNLD-UHFFFAOYSA-N 0.000 description 1
- 239000003960 organic solvent Substances 0.000 description 1
- 239000002245 particle Substances 0.000 description 1
- 244000052769 pathogen Species 0.000 description 1
- 230000008506 pathogenesis Effects 0.000 description 1
- 239000000049 pigment Substances 0.000 description 1
- 239000000256 polyoxyethylene sorbitan monolaurate Substances 0.000 description 1
- 235000010486 polyoxyethylene sorbitan monolaurate Nutrition 0.000 description 1
- 150000008442 polyphenolic compounds Chemical class 0.000 description 1
- 235000013824 polyphenols Nutrition 0.000 description 1
- 235000013772 propylene glycol Nutrition 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 230000037307 sensitive skin Effects 0.000 description 1
- 239000003352 sequestering agent Substances 0.000 description 1
- 239000002453 shampoo Substances 0.000 description 1
- 150000003377 silicon compounds Chemical class 0.000 description 1
- 229920002545 silicone oil Polymers 0.000 description 1
- 230000036556 skin irritation Effects 0.000 description 1
- 231100000475 skin irritation Toxicity 0.000 description 1
- 239000000344 soap Substances 0.000 description 1
- 229940010747 sodium hyaluronate Drugs 0.000 description 1
- YWIVKILSMZOHHF-QJZPQSOGSA-N sodium;(2s,3s,4s,5r,6r)-6-[(2s,3r,4r,5s,6r)-3-acetamido-2-[(2s,3s,4r,5r,6r)-6-[(2r,3r,4r,5s,6r)-3-acetamido-2,5-dihydroxy-6-(hydroxymethyl)oxan-4-yl]oxy-2-carboxy-4,5-dihydroxyoxan-3-yl]oxy-5-hydroxy-6-(hydroxymethyl)oxan-4-yl]oxy-3,4,5-trihydroxyoxane-2- Chemical compound [Na+].CC(=O)N[C@H]1[C@H](O)O[C@H](CO)[C@@H](O)[C@@H]1O[C@H]1[C@H](O)[C@@H](O)[C@H](O[C@H]2[C@@H]([C@@H](O[C@H]3[C@@H]([C@@H](O)[C@H](O)[C@H](O3)C(O)=O)O)[C@H](O)[C@@H](CO)O2)NC(C)=O)[C@@H](C(O)=O)O1 YWIVKILSMZOHHF-QJZPQSOGSA-N 0.000 description 1
- 239000000600 sorbitol Substances 0.000 description 1
- 241000894007 species Species 0.000 description 1
- 238000005507 spraying Methods 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 239000008117 stearic acid Substances 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 238000003860 storage Methods 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 150000005846 sugar alcohols Polymers 0.000 description 1
- 239000004094 surface-active agent Substances 0.000 description 1
- 239000002562 thickening agent Substances 0.000 description 1
- 229940088594 vitamin Drugs 0.000 description 1
- 239000011782 vitamin Substances 0.000 description 1
- 229930003231 vitamin Natural products 0.000 description 1
- 235000013343 vitamin Nutrition 0.000 description 1
- 229920003169 water-soluble polymer Polymers 0.000 description 1
- 239000001993 wax Substances 0.000 description 1
- 230000002087 whitening effect Effects 0.000 description 1
- 230000037303 wrinkles Effects 0.000 description 1
Images
Landscapes
- Cosmetics (AREA)
- Removal Of Specific Substances (AREA)
Abstract
Description
本発明は水道水中に含まれる塩素を除去することができる塩素除去剤および皮膚外用剤等に関する。具体的には、水道水中に含まれ、残留塩素の除去に関する。 The present invention relates to a chlorine remover and a skin external preparation that can remove chlorine contained in tap water. Specifically, it relates to the removal of residual chlorine contained in tap water.
近年、外部環境や食生活などの外的要因、また寝不足や過労、ストレスといった内的要因に起因したアトピー性皮膚炎や肌のカサつきやかゆみ、赤味などの自覚症状を伴ういわゆる敏感肌に悩む人が増加している。こうした症状を引き起こす原因は種々存在し、外的要因としては、窒素酸化物などにより引き起こされる環境汚染や紫外線などが考えられているが、その1つとして塩素などの化学物質による皮膚刺激も挙げられる。 In recent years, for sensitive skin with subjective symptoms such as atopic dermatitis, skin itchiness, itching and redness caused by external factors such as the external environment and diet, and internal factors such as lack of sleep, overwork, and stress. The number of people who are worried is increasing. There are various causes that cause such symptoms, and external factors such as environmental pollution caused by nitrogen oxides and ultraviolet rays are considered, and one of them is skin irritation caused by chemical substances such as chlorine. .
塩素は、我々が日常的に使用している生活用水の水質を確保するため、水道水に含まれることが国の定める基準となっており、我々の生活とは切っても切り離せない存在である。一般的に水道水などの消毒には水系伝染病の病原駆除にも非常に有効であることから、これまで次亜塩素酸ナトリウムが多用されてきた。次亜塩素酸の一部は水中で次亜塩素酸イオンと水素イオンに解離し、この次亜塩素酸と次亜塩素酸イオンは総称して遊離残留塩素または遊離有効塩素と呼ばれる。これら遊離残留塩素はその強い酸化力で微生物やウイルスなどの病原生物の細胞膜や細胞壁を破壊し、内部のタンパク質や核酸を変性させることで殺菌または消毒の効果を発揮している。これに対して、水中のアミン類やアミノ酸類などと遊離残留塩素が結合するとモノクロラミン、ジクロラミンなどの結合型有効塩素となり、結合残留塩素と呼ばれる。この結合残留塩素は、遊離残留塩素に比べるとおよそ数分の一の効果ではあるが、酸化力に由来する比較的強い殺菌または消毒力を持ち、これら遊離残留塩素と結合残留塩素は総じて残留塩素と呼ばれている。(非特許文献1)。 Chlorine is a standard established by the national government to ensure the quality of daily water we use, and it is inseparable from our daily lives. . In general, sodium hypochlorite has been frequently used for disinfection of tap water and the like because it is very effective in controlling the pathogenesis of waterborne infectious diseases. A part of hypochlorous acid dissociates in water into hypochlorite ions and hydrogen ions, and these hypochlorous acid and hypochlorite ions are collectively called free residual chlorine or free effective chlorine. These free residual chlorine exerts the effect of sterilization or disinfection by destroying cell membranes and cell walls of pathogenic organisms such as microorganisms and viruses with its strong oxidizing power, and denaturing internal proteins and nucleic acids. On the other hand, when free residual chlorine is combined with amines or amino acids in water, it becomes combined effective chlorine such as monochloramine and dichloroamine, and is called combined residual chlorine. Although this combined residual chlorine is about a fraction of the effect of free residual chlorine, it has a relatively strong sterilizing or disinfecting power derived from oxidizing power. These free residual chlorine and combined residual chlorine are generally residual chlorine. is called. (Non-Patent Document 1).
残留塩素は高い消毒、殺菌効果を有する反面、近年、人体への影響も懸念されるようになってきた。例えば、プールに入った後に髪がパサつく、きしむといった悩みや、一番風呂に入ると皮膚がピリピリするといった症状は、水中に含まれる残留塩素が皮膚や毛髪を構成しているタンパク質に作用することで起きると考えられる。先行研究でもプールや入浴時に接触する水に含まれている残留塩素がアトピー性皮膚炎の増悪因子であること (非特許文献2) などが報告されており、遊離残留塩素は水を衛生的に保つという大きなメリットがある反面、近年、デメリットも徐々に顕在化してきている。こうした流れを受けて、残留塩素の除去が望まれるようになっているが、日常生活における残留塩素との接触は使用者が積極的な塩素除去手段を講じない限り避けがたいのが現状である。 Residual chlorine has a high disinfection and sterilization effect, but in recent years there have been concerns about the effects on the human body. For example, troubles such as hair becoming crusty or squeaky after entering the pool, or symptoms such as skin tingling when taking the first bath, the residual chlorine contained in the water acts on the proteins that make up the skin and hair It is thought that this happens. In previous studies, it has been reported that residual chlorine contained in water that comes into contact with pools and bathing is an exacerbation factor for atopic dermatitis (Non-patent Document 2). On the other hand, there is a great merit of keeping it, but in recent years, the demerit has gradually become apparent. In response to these trends, removal of residual chlorine is desired, but it is difficult to avoid contact with residual chlorine in daily life unless the user takes active measures to remove chlorine. .
水道水中に含まれる塩素を除去する手段としては、既に様々な方法が提案されており、代表的な方法としては、アスコルビン酸やアスコルビン酸誘導体などを塩素中和剤として用い、残留塩素を中和除去する方法(特許文献1)やケイ素化合物で表面修飾された親水性金属酸化物粒子を多孔質体に添着させたことを特徴とする親水化吸着剤(特許文献2)などがある。 Various methods have already been proposed as means for removing chlorine contained in tap water. As representative methods, ascorbic acid or ascorbic acid derivatives are used as a chlorine neutralizing agent to neutralize residual chlorine. There are a removal method (Patent Document 1) and a hydrophilic adsorbent (Patent Document 2) characterized in that hydrophilic metal oxide particles surface-modified with a silicon compound are attached to a porous body.
しかしながら、アスコルビン酸やその誘導体は高濃度で使用すると刺激や乾燥を感じることがあり、それ自身が不安定で劣化しやすいため保管にも注意を払う必要がある上、例えば、これらを含有する入浴剤の場合は、特に湯船に浸からない頭部や顔面などの洗浄に際しては残留塩素との接触を避けることができない。
また、特許文献2の親水化吸着剤は浄水器やシャワーヘッドのフィルターとして適用するものであるため、定期的なフィルターの交換が必要となり、継続的な使用が容易とは言い難く、限られた場所での使用となるため、所望の場所で簡易に塩素を除去できるものではない。
However, ascorbic acid and its derivatives may cause irritation and dryness when used at high concentrations, and as such, it is unstable and easily deteriorates, so it is necessary to pay attention to storage. For example, baths containing these In the case of an agent, contact with residual chlorine cannot be avoided particularly when cleaning the head or face that is not immersed in the bathtub.
Moreover, since the hydrophilization adsorbent of patent document 2 is applied as a filter for a water purifier or a shower head, it is necessary to periodically replace the filter, and it is difficult to say that continuous use is easy and limited. Since it is used at a place, chlorine cannot be easily removed at a desired place.
本発明は、上記背景に鑑みてなされたものであり、従来ある塩素除去手段の欠点を克服した有用性の高い塩素除去剤、特に水道水中の残留塩素を除去するための塩素除去剤および皮膚外用剤等を提供することを目的とする。 The present invention has been made in view of the above background, and is a highly useful chlorine remover that has overcome the drawbacks of conventional chlorine removing means, in particular, a chlorine remover for removing residual chlorine in tap water, and a skin external application. The purpose is to provide agents.
本発明者らは、上記の背景に鑑み、水道水中に含まれる残留塩素が皮膚および毛髪に及ぼす影響を抑制するために鋭意研究を重ねた結果、Brassica Oleracea var. gemmiferaおよびスクマウィキの抽出物が高い塩素除去作用を有することを見出し、本発明を完成するに至った。
すなわち、本発明は、Brassica Oleracea var. gemmiferaおよびスクマウィキの抽出物から選ばれる1種または2種以上を含むことを特徴とする塩素除去剤を提供する。また、本発明はBrassica Oleracea var. gemmiferaおよびスクマウィキの抽出物から選ばれる1種または2種以上を含むことを特徴とする皮膚外用剤および全身用洗浄剤、頭髪用化粧料、入浴剤を提供するものである。
In view of the above background, the present inventors have conducted extensive research to suppress the influence of residual chlorine contained in tap water on the skin and hair, and as a result, Brassica Oleracea var. It was found that the extract of gemmifera and sumawiki has a high chlorine removing action, and the present invention has been completed.
That is, the present invention relates to Brassica Oleracea var. A chlorine removing agent comprising one or more selected from gemmifera and sumawiki extract is provided. The present invention also relates to Brassica Oleracea var. The present invention provides an external preparation for skin, a cleansing agent for whole body, a cosmetic for hair, and a bath agent characterized by containing one or more selected from gemmifera and sumawiki extract.
本発明は、優れた塩素除去能を有するものである。 The present invention has excellent chlorine removing ability.
以下、本発明について更に詳しく説明する。 Hereinafter, the present invention will be described in more detail.
本発明で用いるBrassica Oleracea var. gemmiferaは、ケールとメキャベツの交配種であり、例えばプチヴェール(登録商標)という名称で市販されている。使用される部位は特に限定されない。葉、花、根、全草等が用いられるが、特に葉が好適で、最も顕著な効果を示した。評価試験では葉を用いているが、その他の部位でも同様の効果が得られる。 Brassica Oleracea var. Used in the present invention. gemmifera is a hybrid of kale and mecha cabbage, and is commercially available, for example, under the name Petitvale (registered trademark). The site used is not particularly limited. Leaves, flowers, roots, whole plants, etc. are used, but leaves are particularly suitable and show the most remarkable effects. In the evaluation test, leaves are used, but the same effect can be obtained at other sites.
本発明で用いるスクマウィキ、は、アブラナ科(Brassicaceae)アブラナ属(Brassica)学名:Brassica Oleracea var.acephalaであり、アフリカ原産の在来種である。使用される部位は特に限定されない。葉、花、根、全草等が用いられるが、特に葉が好適で、最も顕著な効果を示した。評価試験では葉を用いているが、その他の部位でも同様の効果が得られる。 The suma wiki used in the present invention is a Brassicaaceae genus Brassica. Scientific name: Brassica Oleracea var. acephala, a native species native to Africa. The site used is not particularly limited. Leaves, flowers, roots, whole plants, etc. are used, but leaves are particularly suitable and show the most remarkable effects. In the evaluation test, leaves are used, but the same effect can be obtained at other sites.
これらの植物抽出物の調製は特に限定されないが、例えば種々の適当な有機溶媒を用いて、低温下から加温下で抽出される。抽出溶媒としては、例えば、水;メチルアルコール、エチルアルコール等の低級1価アルコール;グリセリン、プロピレングリコール、1,3−ブチレングリコール等の液状多価アルコール;アセトン、メチルエチルケトン等のケトン;酢酸エチルなどのアルキルエステル;ベンゼン、ヘキサン等の炭化水素;ジエチルエーテル等のエーテル類;ジクロルメタン、クロロホルム等のハロゲン化アルカン等の1種または2種以上を用いることが出来る。中でも、水、エチルアルコール、1,3−ブチレングリコールの1種または2種以上の混合溶媒が特に好適である。 The preparation of these plant extracts is not particularly limited. For example, the plant extracts are extracted from a low temperature to a warm temperature using various appropriate organic solvents. Examples of the extraction solvent include water; lower monohydric alcohols such as methyl alcohol and ethyl alcohol; liquid polyhydric alcohols such as glycerin, propylene glycol, and 1,3-butylene glycol; ketones such as acetone and methyl ethyl ketone; and ethyl acetate. One or more of alkyl esters; hydrocarbons such as benzene and hexane; ethers such as diethyl ether; and halogenated alkanes such as dichloromethane and chloroform can be used. Among these, water, ethyl alcohol, and one or more mixed solvents of 1,3-butylene glycol are particularly suitable.
本発明に用いることのできる植物抽出物の抽出方法は特に限定されないが、例えば乾燥したものを用いる場合、質量比で1〜1000倍量、特に10〜100倍量の溶媒を用い、0℃以上、特に20℃〜40℃で1時間以上、特に3〜7日間行うのが好ましい。また、60〜100℃で1〜4時間、加熱抽出しても良い。乾燥粉末は市販のものを用いても良い。また、乾燥したものでなくとも抽出可能である。 Although the extraction method of the plant extract which can be used for this invention is not specifically limited, For example, when using what was dried, 1 to 1000 times amount by mass ratio, especially 10 to 100 times amount of solvent are used, and 0 degreeC or more In particular, it is preferably performed at 20 ° C. to 40 ° C. for 1 hour or longer, particularly 3 to 7 days. Moreover, you may heat-extract for 1 to 4 hours at 60-100 degreeC. A commercially available dry powder may be used. Moreover, extraction is possible even if it is not dried.
以上のような条件で得られる上記各抽出物は、抽出された溶液のまま用いても良いが、さらに必要により、濾過や活性炭等の処理をして、濃縮、脱色、粉末化したものを適宜使い分けて用いることが出来る。 Each of the above-mentioned extracts obtained under the above conditions may be used as an extracted solution, but if necessary, the extract, which has been concentrated, decolored, or powdered by filtration, activated carbon, etc. You can use it properly.
本発明に用いることのできる植物抽出物の各剤組成物における配合量は特に限定されないが、乾燥固形分に換算して0.0001〜20.0質量%が好ましく、特に0.001〜10.0質量%の範囲が最適である。 Although the compounding quantity in each agent composition of the plant extract which can be used for this invention is not specifically limited, 0.0001-20.0 mass% is preferable converted into dry solid content, and especially 0.001-10. A range of 0% by weight is optimal.
本発明は、清浄用化粧品、基礎化粧品、頭髪化粧品、入浴剤、メークアップ化粧品、石鹸類、洗顔料類、医薬部外品、医薬品などに利用できるが、その利用分野は特に限定されるものではない。 The present invention can be used for cleaning cosmetics, basic cosmetics, hair cosmetics, bath preparations, makeup cosmetics, soaps, facial cleansers, quasi drugs, pharmaceuticals, etc., but the field of use is not particularly limited. Absent.
本発明には、本発明の効果を損なわない範囲で、通常化粧品や医薬品等の皮膚外用剤や洗浄剤等に用いられる成分、たとえば、油脂、ロウ類、炭化水素油、エステル油、高級アルコール、シリコーン油、紫外線吸収剤、紫外線散乱剤、保湿剤、界面活性剤、水溶性高分子、増粘剤、粉体、皮膚保護剤、美白剤、シワ改善剤、老化防止剤、植物抽出物、防腐剤、消炎剤、pH調整剤、金属イオン封鎖剤、酸化防止剤、安定化剤、香料、色素、顔料等などを必要に応じて適宜配合することができる。 In the present invention, within the range not impairing the effects of the present invention, components usually used in external preparations for skin and cosmetics such as cosmetics and pharmaceuticals, cleaning agents, such as oils, waxes, hydrocarbon oils, ester oils, higher alcohols, Silicone oil, UV absorber, UV scattering agent, moisturizer, surfactant, water-soluble polymer, thickener, powder, skin protectant, whitening agent, wrinkle improver, anti-aging agent, plant extract, antiseptic Agents, flame retardants, pH adjusters, sequestering agents, antioxidants, stabilizers, fragrances, dyes, pigments, and the like can be appropriately blended as necessary.
本発明は、噴霧用剤、液状、ジェル状、クリーム状、固形状、パック、浴用剤等、または二剤式などこれらを混合して用いる剤型に利用できる。 INDUSTRIAL APPLICATION This invention can be utilized for the dosage form which mixes these, such as a spraying agent, liquid form, gel form, cream form, solid form, a pack, a bath preparation, etc., or these two types.
以下、本発明を実施例によりさらに具体的に説明するが、本発明はこれらの実施例により限定されるものではない。また、特記しない限り配合量は質量%で示す。 EXAMPLES Hereinafter, the present invention will be described more specifically with reference to examples. However, the present invention is not limited to these examples. Unless otherwise specified, the blending amount is expressed in mass%.
実施例1および2、比較例1〜10について塩素除去率を比較した。
<評価方法>
塩素除去率の評価は、一定濃度の次亜塩素酸ナトリウム溶液に各抽出乾燥物の水溶液を添加した試料の残留塩素濃度を測定することで行った。
The chlorine removal rates of Examples 1 and 2 and Comparative Examples 1 to 10 were compared.
<Evaluation method>
The evaluation of the chlorine removal rate was performed by measuring the residual chlorine concentration of a sample obtained by adding an aqueous solution of each extraction dry product to a sodium hypochlorite solution having a constant concentration.
<調製例1>
Brassica Oleracea var. gemmiferaの葉乾燥粉末25gに10倍質量の水を加え分散後、60℃で4時間抽出し、Brassica Oleracea var. gemmifera抽出物を得た。乾燥固形分は23%の収率であった。スクマウィキの葉乾燥粉末25gに10倍質量の水を加え分散後、60℃で4時間抽出し、スクマウィキ抽出物を得た。乾燥固形分は22.6%の収率であった。水を溶媒とし、各抽出物の乾燥固形分の濃度が10mg/Lとなるよう調製し、残留塩素濃度測定試験に供した。また、比較対象として用いたケール葉抽出物、メキャベツ抽出物についても、同様の方法で調製した。
<Preparation Example 1>
Brassica Oleracea var. 10 times the mass of water was added to 25 g of Gemifera leaf dry powder and dispersed, followed by extraction at 60 ° C. for 4 hours. Gemifera extract was obtained. The dry solid content was 23%. 10 g of water was added to and dispersed in 25 g of dried Sumawiki leaf powder, followed by extraction at 60 ° C. for 4 hours to obtain a Sumawiki extract. The dry solid content was 22.6%. Water was used as a solvent, and the concentration of dry solids of each extract was adjusted to 10 mg / L and subjected to a residual chlorine concentration measurement test. In addition, the kale leaf extract and mecha cabbage extract used as comparison targets were prepared in the same manner.
また、陽性対象には、塩素除去効果が知られているL−アスコルビン酸を用いた。まず、各抽出物の乾燥分と比較するためにL−アスコルビン酸は水を溶媒として、10mg/Lの濃度(比較例9)で比較した。また、Brassica Oleracea var. gemmifera抽出物およびスクマウィキ抽出物中に含まれるL−アスコルビン酸量を定量すると、約0.2%であった(表1)。このことからBrassica Oleracea var. gemmifera抽出物、またはスクマウィキ抽出物中のL−アスコルビン酸含有量と同じになるように、L−アスコルビン酸を0.02mg/L(比較例10)になるよう調製した。 Moreover, L-ascorbic acid with a known chlorine removal effect was used as a positive target. First, in order to compare with the dry content of each extract, L-ascorbic acid was compared at a concentration of 10 mg / L (Comparative Example 9) using water as a solvent. Also, Brassica Oleracea var. The amount of L-ascorbic acid contained in the gemmifera extract and the sumawiki extract was determined to be about 0.2% (Table 1). From this, Brassica Oleracea var. L-ascorbic acid was prepared to be 0.02 mg / L (Comparative Example 10) so as to be the same as the L-ascorbic acid content in the gemmifera extract or the Sumawiki extract.
<残留塩素濃度測定試験>
(試験例1)
上記調製例1の方法で10mg/Lになるよう調製したBrassica Oleracea var. gemmifera抽出物、又はスクマウィキ抽出物、比較対象として下記表2に記載した植物抽出物およびL−アスコルビン酸溶液を0.62mLと、2mg/Lになるように調製した次亜塩素酸ナトリウム溶液1.88mLとを混合したものを試料とした。その試料中に含まれる残留塩素濃度を測定し、塩素除去率を算出した(表2)。
より具体的には、培養面積3.38cm2のマルチウェルプレートに0.25% CyDTAを含む3mol/L 酢酸緩衝液 (pH5.2) 30μLを分注し、これに上記試料2.5mLを混合した後、20μLのSBTをさらに混合し、速やかにマイクロプレートリーダー(TECAN,Infinite(登録商標)200PRO)にて655nmにおける吸光度を測定した。なお、塩素除去率は、以下の式1に従い、前述の測定法により測定した吸光度から求めた。
<Residual chlorine concentration measurement test>
(Test Example 1)
Brassica Olacea var. Prepared to 10 mg / L by the method of Preparation Example 1 above. 1. A sodium hypochlorite solution prepared as follows: gemmifera extract or sumawiki extract, plant extract and L-ascorbic acid solution described in Table 2 below as comparison targets, and 0.62 mL, 2 mg / L A sample mixed with 88 mL was used as a sample. The residual chlorine concentration contained in the sample was measured, and the chlorine removal rate was calculated (Table 2).
More specifically, 30 μL of 3 mol / L acetate buffer solution (pH 5.2) containing 0.25% CyDTA was dispensed into a multi-well plate with a culture area of 3.38 cm 2 , and 2.5 mL of the sample was mixed therewith. After that, 20 μL of SBT was further mixed, and the absorbance at 655 nm was quickly measured with a microplate reader (TECAN, Infinite (registered trademark) 200PRO). In addition, the chlorine removal rate was calculated | required from the light absorbency measured by the above-mentioned measuring method according to the following formula | equation 1.
<式1>
<Formula 1>
塩素除去能評価結果を以下に示す。 The results of chlorine removal ability evaluation are shown below.
<植物抽出物の塩素除去率>
試験の結果、Brassica Oleracea var. gemmifera抽出物、スクマウィキ抽出物のどちらにおいても残留塩素が低減していることが確認された。一方、植物ポリフェノールやビタミン類を含有し、抗酸化能を有することが知られている比較例1〜10の植物抽出物では塩素除去効果が低く、実使用において十分な効果を発揮するためには化粧料組成物への配合量が増加することが考えられ、さらには、配合量の増加によって化粧料組成物へ安定して配合することが困難になる、経時安定性が損なわれるといったことが懸念される。 As a result of the test, Brassica Oleracea var. It was confirmed that the residual chlorine was reduced in both the gemmifera extract and the sumawiki extract. On the other hand, in the plant extracts of Comparative Examples 1 to 10, which contain plant polyphenols and vitamins and are known to have antioxidant ability, the chlorine removal effect is low, and in order to exert a sufficient effect in actual use It is conceivable that the blending amount in the cosmetic composition will increase, and further, it is difficult to stably blend into the cosmetic composition due to the increase in blending amount, and there is concern that stability over time will be impaired. Is done.
(試験例1)
残留塩素に曝露した毛髪における実施例1および2のダメージ抑制効果の評価
<評価方法>
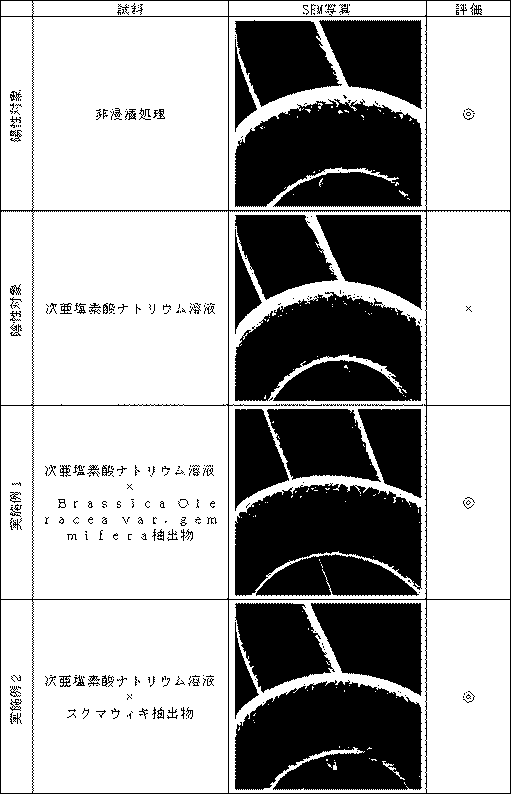
毛髪におけるダメージ抑制効果の評価は、一定濃度の次亜塩素酸ナトリウム溶液に各抽出物の水溶液を添加した試料に毛髪を浸漬処理した際の毛髪表面構造を走査型電子顕微鏡で観察することによって行った。
(Test Example 1)
Evaluation of damage suppression effect of Examples 1 and 2 on hair exposed to residual chlorine <Evaluation Method>
Evaluation of damage-inhibiting effect on hair is performed by observing the hair surface structure with a scanning electron microscope when the hair is immersed in a sample in which an aqueous solution of each extract is added to a sodium hypochlorite solution at a constant concentration. It was.
調整例1に記述した方法で抽出したBrassica Oleracea var. gemmifera抽出物を、水を溶媒として乾燥固形分の濃度が60mg/Lになるように調製し、同じく調製例1に記述した方法で抽出したスクマウィキ抽出物を、水を溶媒として乾燥固形分の濃度が40mg/Lになるように調製した。調製した各抽出物6.7mLと、10mg/Lになるように調製した次亜塩素酸ナトリウム溶液33.5mLとを混合し、試料とした。この試料中に無作為に選択した毛髪(人毛黒毛)を浸漬させ、攪拌しながら、室温、暗所にて30分間処理した。浸漬処理後、濡れた状態の毛髪をドライヤーで5分間風乾させた。この一連の流れを総浸漬時間が16時間になるまで繰り返した後、毛髪の一部を切り取り、走査型電子顕微鏡(SEM)により写真撮影を行った。このSEM写真を図2に示す。SEM写真における毛髪の状況は図1の評価基準にて評価した。陽性対象には浸漬処理していない部位の毛髪を用い、陰性対象には10mg/Lになるよう調製した次亜塩素酸ナトリウム溶液で浸漬処理した毛髪を用いた。 Brassica Oleracea var. Extracted by the method described in Adjustment Example 1. Gemifera extract was prepared using water as a solvent so that the concentration of dry solids was 60 mg / L. Similarly, the Sumawiki extract extracted by the method described in Preparation Example 1 was used as a solvent. Was adjusted to 40 mg / L. 6.7 mL of each prepared extract and 33.5 mL of sodium hypochlorite solution prepared to 10 mg / L were mixed to prepare a sample. Randomly selected hair (human hair black hair) was immersed in this sample and treated for 30 minutes at room temperature in the dark with stirring. After the dipping treatment, the wet hair was air-dried with a dryer for 5 minutes. After repeating this series of flow until the total immersion time reached 16 hours, a part of the hair was cut out and photographed with a scanning electron microscope (SEM). This SEM photograph is shown in FIG. The condition of the hair in the SEM photograph was evaluated according to the evaluation criteria shown in FIG. For the positive target, the hair of the part not subjected to the immersion treatment was used, and for the negative target, the hair immersed in the sodium hypochlorite solution prepared to be 10 mg / L was used.
毛髪におけるダメージ抑制効果の評価結果を図2に示す。 The evaluation result of the damage suppression effect in hair is shown in FIG.
実験の結果、図2より明らかなようにBrassica Oleracea var. gemmifera抽出物、スクマウィキ抽出物を配合することにより残留塩素によるキューティクル層の剥離、脱落等を防ぐことが確認された。 As a result of the experiment, as apparent from FIG. 2, Brassica Oleracea var. It was confirmed that the cuticle layer was prevented from peeling off or falling off due to residual chlorine by blending the gemmifera extract and the sumawiki extract.
以下、本発明に係る処方例を示す。なお、含有量は質量%である。製法は、常法による。なお、処方は代表例であり、これに限定されない。処方例中に配合してある植物の抽出物については、Brassica Oleracea var. gemmifera抽出乾燥物、スクマウィキ抽出乾燥物いずれも抽出部位は葉を用いており、抽出溶媒は水である。一方の植物しか配合していないものも、もう一方の植物の抽出物および他の部位の抽出物を使用した場合にも同種の効果が確認されている。 Hereinafter, formulation examples according to the present invention will be shown. In addition, content is mass%. A manufacturing method is a conventional method. In addition, prescription is a representative example and is not limited to this. For the plant extract incorporated in the formulation examples, see Brassica Olacea var. Both the gemmifera extract dry product and the sumawiki extract dry product use leaves as the extraction site, and the extraction solvent is water. Even when only one plant is blended, the same type of effect has been confirmed when the extract of the other plant and the extract of another part are used.
<処方例1:化粧水>
Brassica Oleracea var. gemmifera抽出乾燥物 0.01
ポリオキシエチレンソルビタンモノラウレート(20E.O.) 1.5
1,3−ブチレングリコール 4.5
グリセリン 3.0
エタノール 2.0
ヒアルロン酸ナトリウム(1%水溶液) 5.0
エデト酸三ナトリウム 0.1
防腐剤 適量
pH調整剤 適量
精製水 残部
合計 100
<Formulation Example 1: Lotion>
Brassica Oleracea var. gemmifera extract dried product 0.01
Polyoxyethylene sorbitan monolaurate (20E.O.) 1.5
1,3-butylene glycol 4.5
Glycerin 3.0
Ethanol 2.0
Sodium hyaluronate (1% aqueous solution) 5.0
Edetate trisodium 0.1
Preservative appropriate amount pH adjuster appropriate amount purified water remaining balance 100
<処方例2:洗顔クリーム>
Brassica Oleracea var. gemmifera抽出乾燥物 3.0
ラウリン酸アミドプロピルベタイン液 2.0
ヒドロキシエチルセルロース 0.7
ソルビトール 1.0
PEG−6(カプリル/カプリン酸)グリセリル 1.0
グリセリン 16.0
PEG−30 4.0
ステアリン酸 8.0
ミリスチン酸 16.0
ラウリン酸 4.0
ステアリン酸グリセリル(SE) 2.0
ホホバ油 4.0
水酸化Ca 5.2
防腐剤 適量
精製水 残部
合計 100
<Prescription Example 2: Face-wash cream>
Brassica Oleracea var. gemmifera extract dried product 3.0
Lauric acid amidopropyl betaine solution 2.0
Hydroxyethyl cellulose 0.7
Sorbitol 1.0
PEG-6 (capryl / capric acid) glyceryl 1.0
Glycerin 16.0
PEG-30 4.0
Stearic acid 8.0
Myristic acid 16.0
Lauric acid 4.0
Glyceryl stearate (SE) 2.0
Jojoba oil 4.0
Hydroxylated Ca 5.2
Preservative Appropriate amount of purified water, remaining balance 100
<処方例3:シャンプー>
スクマウィキ抽出乾燥物 3.0
ポリクオタニウム−10 0.4
1,3‐ブチレングリコール 5.0
ココイルメチルタウリンNa 10.5
ラウリン酸BG 1.2
ラウレス硫酸Na 15.0
コカミドプロピルベタイン 4.0
オレイン酸PEG−18グリセリル 1.2
ジステアリン酸グリコール 1.0
カチオン化ヒドロキシセルロース 1.0
防腐剤 適量
pH調整剤 適量
精製水 残部
合計 100
<Prescription Example 3: Shampoo>
Sumawiki Extract Dried 3.0
Polyquaternium-10 0.4
1,3-butylene glycol 5.0
Cocoyl Methyl Taurine Na 10.5
Lauric acid BG 1.2
Laureth sulfate Na 15.0
Cocamidopropyl betaine 4.0
PEG-18 glyceryl oleate 1.2
Glycol distearate 1.0
Cationized hydroxycellulose 1.0
Preservative appropriate amount pH adjuster appropriate amount purified water remaining balance 100
各処方例の製剤を常法にて調製した。いずれも有意に塩素除去効果を確認できた。
The preparation of each formulation example was prepared by a conventional method. In both cases, the chlorine removal effect was significantly confirmed.
Claims (5)
Brassica Oleracea var. The bath agent containing 1 type, or 2 or more types chosen from the extract of gemmifera and Sumawiki.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2016072574A JP6534957B2 (en) | 2016-03-31 | 2016-03-31 | Chlorine remover and skin external preparation |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2016072574A JP6534957B2 (en) | 2016-03-31 | 2016-03-31 | Chlorine remover and skin external preparation |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2017178902A true JP2017178902A (en) | 2017-10-05 |
| JP6534957B2 JP6534957B2 (en) | 2019-06-26 |
Family
ID=60003739
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2016072574A Active JP6534957B2 (en) | 2016-03-31 | 2016-03-31 | Chlorine remover and skin external preparation |
Country Status (1)
| Country | Link |
|---|---|
| JP (1) | JP6534957B2 (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN116327624A (en) * | 2023-04-21 | 2023-06-27 | 安蒂加(上海)生物科技有限公司 | Moisturizing dechlorination shower gel and preparation method thereof |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2000070956A (en) * | 1998-08-28 | 2000-03-07 | Goshu Yakuhin Kk | Removing agent containing extract of galenical with hot water for free residual chlorine in water and bathing agent containing the same |
| JP2006083078A (en) * | 2004-09-14 | 2006-03-30 | Umebayashi Wataru | Bathing agent composition |
| JP2009213451A (en) * | 2008-03-13 | 2009-09-24 | Niigata Bio Research Park Kk | Food and drink preventing life-style disease |
| KR20140090896A (en) * | 2013-01-10 | 2014-07-18 | 홍소영 | Cosmetic composition comprising Brassica oleracea L. var. gemmifera Zenker extracts |
-
2016
- 2016-03-31 JP JP2016072574A patent/JP6534957B2/en active Active
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2000070956A (en) * | 1998-08-28 | 2000-03-07 | Goshu Yakuhin Kk | Removing agent containing extract of galenical with hot water for free residual chlorine in water and bathing agent containing the same |
| JP2006083078A (en) * | 2004-09-14 | 2006-03-30 | Umebayashi Wataru | Bathing agent composition |
| JP2009213451A (en) * | 2008-03-13 | 2009-09-24 | Niigata Bio Research Park Kk | Food and drink preventing life-style disease |
| KR20140090896A (en) * | 2013-01-10 | 2014-07-18 | 홍소영 | Cosmetic composition comprising Brassica oleracea L. var. gemmifera Zenker extracts |
Non-Patent Citations (2)
| Title |
|---|
| "エチュードエブリマンスクレンジングクリーム ソフト&モイスチャー", COSMETIC-INFO.JP[ONLINE], JPN6019007209, May 2014 (2014-05-01), ISSN: 0003989296 * |
| 竹内若子: ""新葉菜類および新品種ナスの抗酸化性機能成分"", 名古屋女子大学紀要, vol. 第52号, JPN6019007208, 2006, pages 59 - 66, ISSN: 0003989295 * |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN116327624A (en) * | 2023-04-21 | 2023-06-27 | 安蒂加(上海)生物科技有限公司 | Moisturizing dechlorination shower gel and preparation method thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| JP6534957B2 (en) | 2019-06-26 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| RU2494748C2 (en) | High-stable electrolytic water with reduced width of nuclear magnetic resonance peak at half height | |
| CN101569596B (en) | Antioxidation cosmetic combination containing nanometer platinum colloid ion water | |
| CN114423400A (en) | Cosmetic composition containing graphene quantum dots as active ingredient | |
| KR101839491B1 (en) | Extraction Method of Houttuynia cordata Thunberg Extract and Cosmetic materials Using the same | |
| EP3102181B1 (en) | Active complex for a skin anti-ageing cosmetic | |
| JP4644589B2 (en) | Negative ion water production method and negative ion water | |
| KR101056129B1 (en) | Anti-inflammatory composition | |
| JP5701480B2 (en) | Antioxidant | |
| JP2023153398A (en) | Skin external preparation | |
| JP4881537B2 (en) | Moisturizing moisturizing water | |
| JP6534957B2 (en) | Chlorine remover and skin external preparation | |
| JP5196416B2 (en) | Cosmetic composition containing Limnocitrus litoritalis extract | |
| WO2012042970A1 (en) | Prophylactic or therapeutic agent for atopic dermatitis, and preparation for external application | |
| EP2747739B1 (en) | Dermocosmetic compositions based on a synergistic combination of colloidal silver and deoxyribonucleic acid | |
| JP2008120717A (en) | Cosmetic composition | |
| JP2006052198A (en) | Deodorant agent, deodorant preparation, and fiber deodorant treatment method | |
| KR20180126912A (en) | A composition for water tissue containing lava seawater | |
| KR20110057843A (en) | Skin cleanser composition | |
| JP2006241148A (en) | Collagenase inhibitor and anti-aging skin external preparation | |
| KR101965420B1 (en) | Cosmetic composition for improving atopic dermatitis containing extract of pseudostellaria palibiniana ohwi | |
| CN113577000B (en) | Facial cleanser containing dendrobium officinale extract and preparation method thereof | |
| KR20150116504A (en) | A cosmetics composition containing tea tree and solt | |
| JP2011006376A (en) | Shampoo using pure water and photocatalyst, and method for producing the same | |
| JP2007204418A (en) | Topical skin preparation | |
| JP2014062058A (en) | Antimicrobial agent and skin external preparation |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20180319 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20190125 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20190312 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20190510 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20190528 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20190530 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 6534957 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |