KR20140000717A - 신규한 항-혈소판 화합물의 부가염 - Google Patents
신규한 항-혈소판 화합물의 부가염 Download PDFInfo
- Publication number
- KR20140000717A KR20140000717A KR1020137030030A KR20137030030A KR20140000717A KR 20140000717 A KR20140000717 A KR 20140000717A KR 1020137030030 A KR1020137030030 A KR 1020137030030A KR 20137030030 A KR20137030030 A KR 20137030030A KR 20140000717 A KR20140000717 A KR 20140000717A
- Authority
- KR
- South Korea
- Prior art keywords
- formula
- acid addition
- sulfonic acid
- compound
- acid
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D495/00—Heterocyclic compounds containing in the condensed system at least one hetero ring having sulfur atoms as the only ring hetero atoms
- C07D495/02—Heterocyclic compounds containing in the condensed system at least one hetero ring having sulfur atoms as the only ring hetero atoms in which the condensed system contains two hetero rings
- C07D495/04—Ortho-condensed systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P7/00—Drugs for disorders of the blood or the extracellular fluid
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P7/00—Drugs for disorders of the blood or the extracellular fluid
- A61P7/02—Antithrombotic agents; Anticoagulants; Platelet aggregation inhibitors
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C309/00—Sulfonic acids; Halides, esters, or anhydrides thereof
- C07C309/01—Sulfonic acids
- C07C309/02—Sulfonic acids having sulfo groups bound to acyclic carbon atoms
- C07C309/03—Sulfonic acids having sulfo groups bound to acyclic carbon atoms of an acyclic saturated carbon skeleton
- C07C309/04—Sulfonic acids having sulfo groups bound to acyclic carbon atoms of an acyclic saturated carbon skeleton containing only one sulfo group
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C309/00—Sulfonic acids; Halides, esters, or anhydrides thereof
- C07C309/01—Sulfonic acids
- C07C309/02—Sulfonic acids having sulfo groups bound to acyclic carbon atoms
- C07C309/19—Sulfonic acids having sulfo groups bound to acyclic carbon atoms of a saturated carbon skeleton containing rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C309/00—Sulfonic acids; Halides, esters, or anhydrides thereof
- C07C309/01—Sulfonic acids
- C07C309/28—Sulfonic acids having sulfo groups bound to carbon atoms of six-membered aromatic rings of a carbon skeleton
- C07C309/29—Sulfonic acids having sulfo groups bound to carbon atoms of six-membered aromatic rings of a carbon skeleton of non-condensed six-membered aromatic rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C309/00—Sulfonic acids; Halides, esters, or anhydrides thereof
- C07C309/01—Sulfonic acids
- C07C309/28—Sulfonic acids having sulfo groups bound to carbon atoms of six-membered aromatic rings of a carbon skeleton
- C07C309/29—Sulfonic acids having sulfo groups bound to carbon atoms of six-membered aromatic rings of a carbon skeleton of non-condensed six-membered aromatic rings
- C07C309/30—Sulfonic acids having sulfo groups bound to carbon atoms of six-membered aromatic rings of a carbon skeleton of non-condensed six-membered aromatic rings of six-membered aromatic rings substituted by alkyl groups
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Public Health (AREA)
- General Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Veterinary Medicine (AREA)
- Diabetes (AREA)
- Hematology (AREA)
- Urology & Nephrology (AREA)
- Vascular Medicine (AREA)
- Cardiology (AREA)
- Heart & Thoracic Surgery (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Heterocyclic Carbon Compounds Containing A Hetero Ring Having Oxygen Or Sulfur (AREA)
Abstract
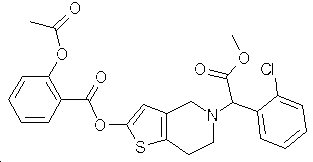
본 발명은 일반 화학식 I 화합물의 할로겐화수소산 및 설폰산 부가염, 이들의 제조방법, 및 혈전증 또는 색전증의 치료 또는 예방을 위한 약제의 제조에 있어서의 이들의 용도를 개시한다. 본 발명의 염은 유리 화합물 그 자체에 비해 더 안정적이며, 우수한 구강 흡수성 및 혈소판 응고에 대한 저해효과와 항-응고제로서의 높은 잠재성을 가지고 있다.
Description
| 번호 | 산 | 형성된 염의 물리적 상태 | 염의 특성 | 비고 |
| 1 | 폼산(formic acid) | 오일 | - | |
| 2 | 아세트산 | 오일 | - | |
| 3 | 프로피온산 | 오일 | - | |
| 4 | 뷰티르산 | 오일 | - | |
| 5 | 벤조산 | 오일 | - | |
| 6 | 파라벤 | 오일 | - | |
| 7 | L-만델산 | 오일 | - | |
| 8 | L-타르타르산 | 오일 | - | |
| 9 | L-캄포 설폰산 | 고체 | 백색의 고체 | |
| 10 | D-캄포 설폰산 | 끈적한 고체 | - | 염(base)이 순수하지 않은 상태에서 오일이 형성됨 |
| 11 | 메탄 설폰산 | 고체 | 백색의 고체 | |
| 12 | 벤젠설폰산 | 고체 | 백색의 고체 | |
| 13 | P-톨루엔 설폰산 | 고체 | 백색의 고체 | |
| 14 | 말레산 | 오일 | - | |
| 15 | 퓨말산 | 오일 | - | |
| 16 | 옥살산 | 오일 | - | |
| 17 | 아스파트산 | 오일 | - | |
| 18 | 염산 | 고체 | 백색의 고체 | |
| 19 | 브로민화수소산 | 고체 | 노르스름한 고체 | |
| 20 | 아이오딘화수소산 | 고체 | 황색의 고체 | |
| 21 | 황산 | 끈적한 고체 | - | 염형성 과정에서 끈적해지는 경향이 있음 |
| 22 | 인산 | 오일 | - |
| 번호 | 염 | 용해도 (mg/ml) |
비고 |
| 1 | L-캄포설폰산염 | 4.7 | |
| 2 | 메탄설폰산염 | 8.2 | |
| 3 | 벤젠설폰산염 | 2.5 | |
| 4 | P-톨루엔설폰산염 | 1.2 | |
| 5 | 염화수소염 | 0.02 | 용해과정 중 끈적해지는 경향이 있음 |
| 6 | 브로민화수소염 | 0.5 |
| 번호 | 염 | 안정성 |
| 1 | L-캄포설폰산염 | 안정적 |
| 2 | 벤젠설폰산염 | 안정적 |
| 3 | P-톨루엔설폰산염 | 안정적 |
| 4 | 메탄설폰산염 | 안정적 |
| 약물 | 투여량(mg/kg) | 혈전 형성 시간(s) | 혈전의 중량(mg) |
| 대조군 | 88.6±22.2 | 5.4±0.5 | |
| 클로피도그렐 그룹 | 10 | 249.9±58.0# | 3.2±0.5# |
| 유리 염기 그룹 | 3 | 165±22.4# | 4.0±0.6# |
| 9 | 261±54.3#,& | 3.0±0.5#,& | |
| 하이드로클로라이드 그룹 | 3 | 210.4±51.1# | 3.2±0.7#,Y |
| 9 | 319.6±35.2#,H,&,Y | 2.8±0.5# | |
| L-캄포설폰산염 그룹 | 3 | 250.4±35.2# ,Y,* | 3.0±0.7#,Y |
| 9 | 360.2±5.9#,H,&,Y,* | 2.5±0.4#, H,&,Y |
Claims (9)
- 1항에 있어서, 상기 화학식 I의 화합물은 라세미체인 할로겐화수소산 및 설폰산 부가염.
- 제1항에 있어서, 상기 화학식 I의 화합물은 S-이성질체인 할로겐화수소산 및 설폰산 부가염.
- 제1항에 있어서, 상기 할로겐화수소산 부가염은 염산염, 브로민화수소산염 및 아이오딘화수소산염에서 선택되는 것인 할로겐화수소산 및 설폰산 부가염.
- 제1항에 있어서, 상기 설폰산 부가염은 메탄설폰산염, 에탄설폰산염, 프로판 설폰산염, L-캄포설폰산염, D-캄포설폰산염, 벤젠설폰산염, 나프탈렌설폰산염 및 p-톨루엔설폰산염에서 선택되는 것인 할로겐화수소산 및 설폰산 부가염.
- 화학식 I 화합물 또는 이의 용매 중의 용액을, 상응하는 산 또는 이의 용매 중의 용액에 첨가하거나, 다른 방안으로 산 또는 이의 용매 중의 용액을 화학식 I 화합물 또는 이의 용액에 첨가하는 것을 포함하는 제1항에 따른 염의 제조방법.
- 제6항에 있어서, 상기 반응 용매는 헥산, 사이클로헥산, 헵탄, 리그로인 또는 페트롤륨 에테르; 톨루엔 또는 자일렌; 디클로로메탄, 클로로포름, 사염화탄소(carbon tetrachloride), 1,2-디클로로에탄, 클로로벤젠 또는 디클로로벤젠; 디에틸 에테르, 디이소프로필 에테르, 테트라하이드로퓨란, 디옥산, 디메톡시에탄 또는 디에틸렌 글리콜 디메틸 에테르; 아세트산 에틸, 아세트산 메틸, 아세트산 프로필 또는 아세트산 부틸, 또는 상기 언급된 시약과 메탄올, 에탄올, 프로판올, 이소프로판올, 부탄올, 아세톤, 메틸 에틸 케톤, 아밀 케톤 및 사이클로헥사논 중의 하나 또는 둘과의 조합에서 선택되는 것인 염의 제조방법.
- 제6항에 있어서, 상기 용매는 디에틸 에테르, 톨루엔, 아세톤, 이소프로판올 또는 이의 조합인 염의 제조방법.
- 혈전증 또는 색전증의 치료 또는 예방을 위한 약제의 제조에 있어서의 제1항 내지 제5항 중 어느 한 항에 따른 화학식 I 화합물의 염의 용도.
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201110098528 | 2011-04-14 | ||
| CN201110098528.3 | 2011-04-14 | ||
| PCT/CN2012/000468 WO2012139422A1 (zh) | 2011-04-14 | 2012-04-09 | 新型抗血小板化合物的加成盐 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| KR20140000717A true KR20140000717A (ko) | 2014-01-03 |
| KR101552691B1 KR101552691B1 (ko) | 2015-09-11 |
Family
ID=47008820
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020137030030A Expired - Fee Related KR101552691B1 (ko) | 2011-04-14 | 2012-04-09 | 신규한 항-혈소판 화합물의 부가염 |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US9556196B2 (ko) |
| EP (1) | EP2698371B1 (ko) |
| JP (1) | JP5695797B2 (ko) |
| KR (1) | KR101552691B1 (ko) |
| CN (1) | CN103517909B (ko) |
| WO (1) | WO2012139422A1 (ko) |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR102358548B1 (ko) * | 2014-10-15 | 2022-02-04 | 삼성전자주식회사 | 디바이스를 이용한 화면 처리 방법 및 장치 |
| CN107304215A (zh) * | 2016-04-20 | 2017-10-31 | 陕西合成药业股份有限公司 | 噻吩吡啶类衍生物及其制备方法和用途 |
| KR102934570B1 (ko) | 2022-10-21 | 2026-03-06 | 고려대학교 산학협력단 | 신규한 2-((1h-인돌-3-일)메틸렌)-n-페닐히드라진-1-카르복사마이드 유도체 및 이의 약학적 용도 |
Family Cites Families (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2652579B1 (fr) * | 1989-10-02 | 1992-01-24 | Sanofi Sa | Derives d'hydroxy-2 thiophene et furanne condenses avec un cycle azote, sur procede de preparation et leur application en therapeutique. |
| IL166593A0 (en) * | 2002-08-02 | 2006-01-15 | Racemization and enantiomer separation of clopidogrel | |
| HRP20140668T1 (hr) * | 2007-03-02 | 2014-10-10 | Daiichi Sankyo Company, Limited | Postupak proizvodnje prasugrel hidroklorida visoke äśistoä†e |
| CN101591344B (zh) | 2008-05-27 | 2012-12-05 | 连云港恒邦医药科技有限公司 | 一种抗血栓的化合物、其制备方法和用途 |
| CN101684124B (zh) * | 2008-09-22 | 2013-11-13 | 广州赫尔氏药物开发有限公司 | 新的具有抗凝血作用的化合物 |
| WO2011079407A1 (zh) * | 2009-12-28 | 2011-07-07 | 北京益君康医药技术有限公司 | 新的具有抗凝血作用的化合物 |
| CN102120744B (zh) * | 2010-02-02 | 2013-01-09 | 江苏威凯尔医药科技有限公司 | 光学活性2-羟基四氢噻吩并吡啶衍生物及其制备方法与在制药中的用途 |
-
2012
- 2012-04-09 EP EP12771534.0A patent/EP2698371B1/en active Active
- 2012-04-09 CN CN201280017876.6A patent/CN103517909B/zh active Active
- 2012-04-09 KR KR1020137030030A patent/KR101552691B1/ko not_active Expired - Fee Related
- 2012-04-09 US US14/110,898 patent/US9556196B2/en not_active Expired - Fee Related
- 2012-04-09 JP JP2014504145A patent/JP5695797B2/ja not_active Expired - Fee Related
- 2012-04-09 WO PCT/CN2012/000468 patent/WO2012139422A1/zh not_active Ceased
Also Published As
| Publication number | Publication date |
|---|---|
| JP5695797B2 (ja) | 2015-04-08 |
| EP2698371A4 (en) | 2014-09-03 |
| EP2698371B1 (en) | 2017-06-14 |
| EP2698371A1 (en) | 2014-02-19 |
| JP2014510765A (ja) | 2014-05-01 |
| CN103517909B (zh) | 2015-04-08 |
| CN103517909A (zh) | 2014-01-15 |
| US9556196B2 (en) | 2017-01-31 |
| WO2012139422A1 (zh) | 2012-10-18 |
| KR101552691B1 (ko) | 2015-09-11 |
| US20140031386A1 (en) | 2014-01-30 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP2112155B1 (en) | Hydrogensulfate salt of 2-acetoxy-5-(a-cyclopropylcarbonyl-2-fluorobenzyl)-4,5,6,7-tetrahydrothieno[3,2-c]pyridine and its preparation | |
| CN101260112B (zh) | 含噻吩并[3.2-c]吡啶的乙酰肼衍生物及其制备方法和用途 | |
| JPS63203684A (ja) | 右旋性α−5−(4,5,6,7−テトラヒドロ[3,2−c]チエノピリジル)−(2−クロロフェニル)酢酸メチル、その製法および医薬 | |
| JP7264999B2 (ja) | 2-(1-アシルオキシ-n-ペンチル)安息香酸と塩基性アミノ酸またはアミノグアニジンによって形成される塩と、その製造方法及び用途 | |
| JP5575979B2 (ja) | シアノ基を含有するチエノピリジンエステル誘導体、その調製方法、使用および組成物 | |
| KR100437307B1 (ko) | 토라세미드의 새로운 결정 변형태 ⅲ | |
| TW201643143A (zh) | 抑制氧化壓迫引發的神經細胞死亡之化合物 | |
| CN1318428C (zh) | 噻吩并吡啶取代的乙酰肼衍生物 | |
| JPH0428269B2 (ko) | ||
| KR101552691B1 (ko) | 신규한 항-혈소판 화합물의 부가염 | |
| BR112012028919B1 (pt) | Sal da rupatadina como um agente anti-histamínico | |
| CN109503548B (zh) | 一种丁苯酞衍生物及其制备方法和应用 | |
| KR20080003599A (ko) | 피롤로피리미디논 유도체의 겐티세이트 염 및 이의제조방법 | |
| JP3853389B2 (ja) | 新規の3−フェニルスルホニル−3,7−ジアザビシクロ[3,3,1]ノナン−化合物、その製法及び抗不整脈剤 | |
| EP3315505B1 (en) | Deuterated thienopiperidine derivatives, manufacturing method, and application thereof | |
| PT770082E (pt) | Derivados de acido dioxo-tiopirano-piridinocarboxilico e sua utilizacao como medicamentos | |
| CN103864756B (zh) | 丁二磺酸达比加群酯及其制备方法和用途 | |
| CN112778371B (zh) | 一种噻吩并吡啶衍生物及其制备方法和用途 | |
| US20080293772A1 (en) | Salts of picotamide | |
| KR100742134B1 (ko) | 결정성S-(+)-메틸-2-(2-클로로페닐)-2-(6,7-디히드로티에노[3,2-c]피리딘-5(4H)-일)아세테이트·캄실레이트를 포함하는약학적 조성물 | |
| JP7473554B2 (ja) | Par4阻害剤の結晶体 | |
| KR100563455B1 (ko) | 결정성 클로피도그렐 나프탈렌술폰산염 또는 이의 수화물,이의 제조방법 및 이를 함유하는 약학적 조성물 | |
| JP2020533401A (ja) | 不飽和脂肪族オレフィン性結合を含有するチエノピリジン誘導体、その調製方法および使用 | |
| CN106565698B (zh) | 取代噻唑类衍生物及其制备方法和用途 | |
| EA043816B1 (ru) | Кристаллические формы ингибитора par4 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A201 | Request for examination | ||
| PA0105 | International application |
St.27 status event code: A-0-1-A10-A15-nap-PA0105 |
|
| PA0201 | Request for examination |
St.27 status event code: A-1-2-D10-D11-exm-PA0201 |
|
| PG1501 | Laying open of application |
St.27 status event code: A-1-1-Q10-Q12-nap-PG1501 |
|
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
St.27 status event code: A-1-2-D10-D21-exm-PE0902 |
|
| AMND | Amendment | ||
| E13-X000 | Pre-grant limitation requested |
St.27 status event code: A-2-3-E10-E13-lim-X000 |
|
| P11-X000 | Amendment of application requested |
St.27 status event code: A-2-2-P10-P11-nap-X000 |
|
| P13-X000 | Application amended |
St.27 status event code: A-2-2-P10-P13-nap-X000 |
|
| E601 | Decision to refuse application | ||
| PE0601 | Decision on rejection of patent |
St.27 status event code: N-2-6-B10-B15-exm-PE0601 |
|
| AMND | Amendment | ||
| P11-X000 | Amendment of application requested |
St.27 status event code: A-2-2-P10-P11-nap-X000 |
|
| P13-X000 | Application amended |
St.27 status event code: A-2-2-P10-P13-nap-X000 |
|
| PX0901 | Re-examination |
St.27 status event code: A-2-3-E10-E12-rex-PX0901 |
|
| PX0701 | Decision of registration after re-examination |
St.27 status event code: A-3-4-F10-F13-rex-PX0701 |
|
| X701 | Decision to grant (after re-examination) | ||
| GRNT | Written decision to grant | ||
| PR0701 | Registration of establishment |
St.27 status event code: A-2-4-F10-F11-exm-PR0701 |
|
| PR1002 | Payment of registration fee |
St.27 status event code: A-2-2-U10-U12-oth-PR1002 Fee payment year number: 1 |
|
| PG1601 | Publication of registration |
St.27 status event code: A-4-4-Q10-Q13-nap-PG1601 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 4 |
|
| FPAY | Annual fee payment |
Payment date: 20190826 Year of fee payment: 5 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 5 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 6 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 7 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 8 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 9 |
|
| PC1903 | Unpaid annual fee |
St.27 status event code: A-4-4-U10-U13-oth-PC1903 Not in force date: 20240908 Payment event data comment text: Termination Category : DEFAULT_OF_REGISTRATION_FEE |
|
| PC1903 | Unpaid annual fee |
St.27 status event code: N-4-6-H10-H13-oth-PC1903 Ip right cessation event data comment text: Termination Category : DEFAULT_OF_REGISTRATION_FEE Not in force date: 20240908 |