KR20150100902A - 옥시부티닌의 투여를 위한 방법 및 조성물 - Google Patents
옥시부티닌의 투여를 위한 방법 및 조성물 Download PDFInfo
- Publication number
- KR20150100902A KR20150100902A KR1020157020335A KR20157020335A KR20150100902A KR 20150100902 A KR20150100902 A KR 20150100902A KR 1020157020335 A KR1020157020335 A KR 1020157020335A KR 20157020335 A KR20157020335 A KR 20157020335A KR 20150100902 A KR20150100902 A KR 20150100902A
- Authority
- KR
- South Korea
- Prior art keywords
- oxybutynin
- salt
- vitamin
- agent
- agents
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
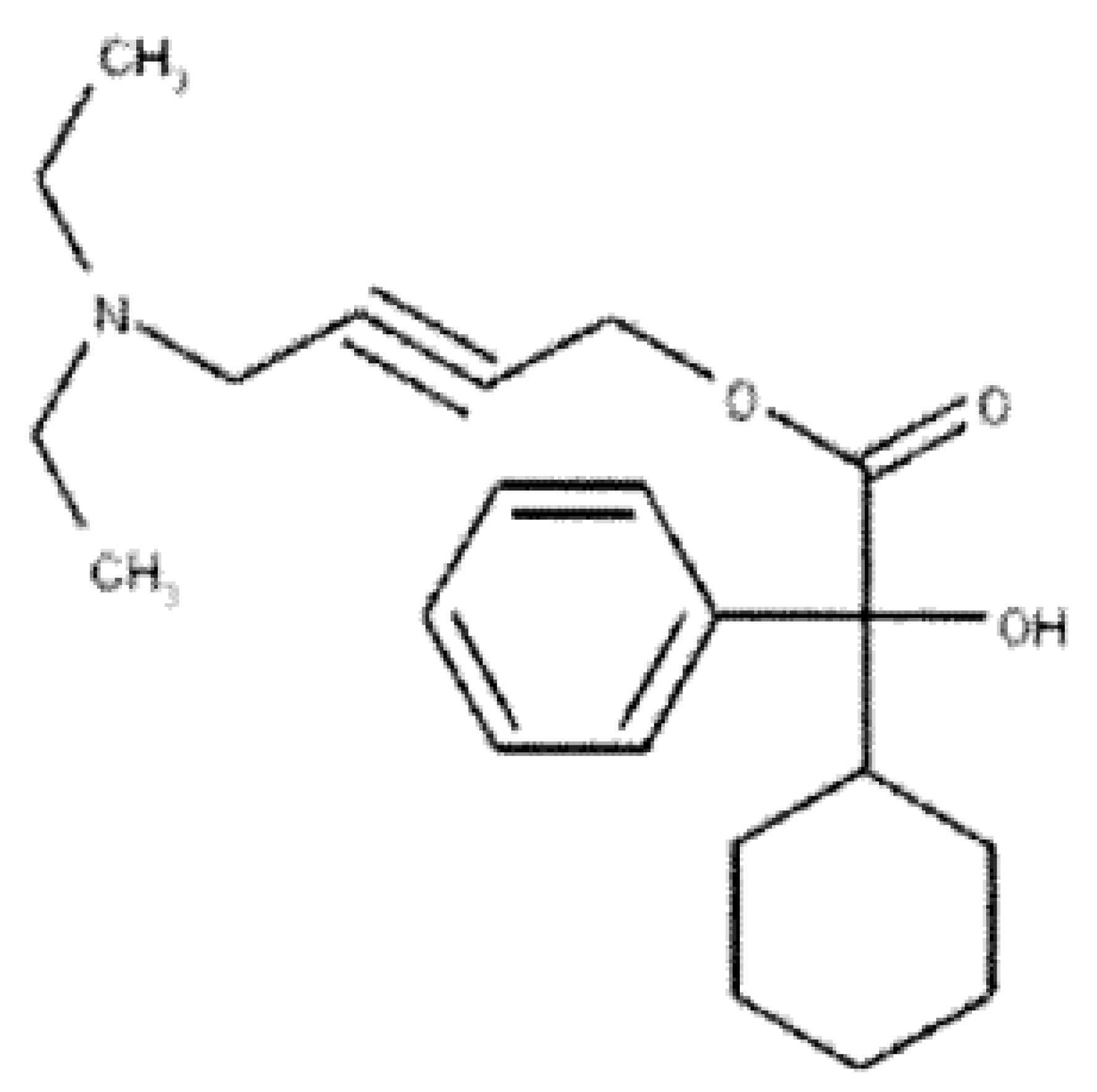
- XIQVNETUBQGFHX-UHFFFAOYSA-N Ditropan Chemical compound C=1C=CC=CC=1C(O)(C(=O)OCC#CCN(CC)CC)C1CCCCC1 XIQVNETUBQGFHX-UHFFFAOYSA-N 0.000 title claims abstract description 161
- 229960005434 oxybutynin Drugs 0.000 title claims abstract description 155
- 239000000203 mixture Substances 0.000 title claims abstract description 34
- 238000000034 method Methods 0.000 title claims abstract description 33
- 239000003795 chemical substances by application Substances 0.000 claims abstract description 52
- 150000003839 salts Chemical class 0.000 claims abstract description 42
- 210000004072 lung Anatomy 0.000 claims abstract description 27
- 239000003246 corticosteroid Substances 0.000 claims abstract description 21
- 208000019693 Lung disease Diseases 0.000 claims abstract description 18
- -1 cinnamoyl Chemical group 0.000 claims abstract description 15
- 229940124630 bronchodilator Drugs 0.000 claims abstract description 12
- 229960001334 corticosteroids Drugs 0.000 claims abstract description 6
- 229940121363 anti-inflammatory agent Drugs 0.000 claims abstract description 5
- 239000002260 anti-inflammatory agent Substances 0.000 claims abstract description 5
- 239000013543 active substance Substances 0.000 claims abstract description 4
- 239000000137 peptide hydrolase inhibitor Substances 0.000 claims abstract description 3
- 208000006545 Chronic Obstructive Pulmonary Disease Diseases 0.000 claims description 23
- 239000000843 powder Substances 0.000 claims description 20
- 239000002245 particle Substances 0.000 claims description 19
- 206010057190 Respiratory tract infections Diseases 0.000 claims description 18
- 102000003702 retinoic acid receptors Human genes 0.000 claims description 18
- 108090000064 retinoic acid receptors Proteins 0.000 claims description 18
- QYSXJUFSXHHAJI-XFEUOLMDSA-N Vitamin D3 Natural products C1(/[C@@H]2CC[C@@H]([C@]2(CCC1)C)[C@H](C)CCCC(C)C)=C/C=C1\C[C@@H](O)CCC1=C QYSXJUFSXHHAJI-XFEUOLMDSA-N 0.000 claims description 16
- 229930003316 Vitamin D Natural products 0.000 claims description 15
- 238000009472 formulation Methods 0.000 claims description 15
- 235000019166 vitamin D Nutrition 0.000 claims description 15
- 239000011710 vitamin D Substances 0.000 claims description 15
- 150000003710 vitamin D derivatives Chemical class 0.000 claims description 15
- 229940046008 vitamin d Drugs 0.000 claims description 15
- 229940112141 dry powder inhaler Drugs 0.000 claims description 11
- SHGAZHPCJJPHSC-YCNIQYBTSA-N all-trans-retinoic acid Chemical compound OC(=O)\C=C(/C)\C=C\C=C(/C)\C=C\C1=C(C)CCCC1(C)C SHGAZHPCJJPHSC-YCNIQYBTSA-N 0.000 claims description 10
- 239000003112 inhibitor Substances 0.000 claims description 10
- 239000008194 pharmaceutical composition Substances 0.000 claims description 10
- 208000006673 asthma Diseases 0.000 claims description 9
- 102000009310 vitamin D receptors Human genes 0.000 claims description 9
- 108050000156 vitamin D receptors Proteins 0.000 claims description 9
- 239000000556 agonist Substances 0.000 claims description 8
- WBYWAXJHAXSJNI-UHFFFAOYSA-N cinnamic acid Chemical compound OC(=O)C=CC1=CC=CC=C1 WBYWAXJHAXSJNI-UHFFFAOYSA-N 0.000 claims description 8
- ZFXYFBGIUFBOJW-UHFFFAOYSA-N theophylline Chemical compound O=C1N(C)C(=O)N(C)C2=C1NC=N2 ZFXYFBGIUFBOJW-UHFFFAOYSA-N 0.000 claims description 8
- MECHNRXZTMCUDQ-RKHKHRCZSA-N vitamin D2 Chemical compound C1(/[C@@H]2CC[C@@H]([C@]2(CCC1)C)[C@H](C)/C=C/[C@H](C)C(C)C)=C\C=C1\C[C@@H](O)CCC1=C MECHNRXZTMCUDQ-RKHKHRCZSA-N 0.000 claims description 8
- 229960002848 formoterol Drugs 0.000 claims description 7
- BPZSYCZIITTYBL-UHFFFAOYSA-N formoterol Chemical compound C1=CC(OC)=CC=C1CC(C)NCC(O)C1=CC=C(O)C(NC=O)=C1 BPZSYCZIITTYBL-UHFFFAOYSA-N 0.000 claims description 7
- VOVIALXJUBGFJZ-KWVAZRHASA-N Budesonide Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@@H]2[C@@H]1[C@@H]1C[C@H]3OC(CCC)O[C@@]3(C(=O)CO)[C@@]1(C)C[C@@H]2O VOVIALXJUBGFJZ-KWVAZRHASA-N 0.000 claims description 6
- 229960004436 budesonide Drugs 0.000 claims description 6
- 229960002714 fluticasone Drugs 0.000 claims description 6
- MGNNYOODZCAHBA-GQKYHHCASA-N fluticasone Chemical compound C1([C@@H](F)C2)=CC(=O)C=C[C@]1(C)[C@]1(F)[C@@H]2[C@@H]2C[C@@H](C)[C@@](C(=O)SCF)(O)[C@@]2(C)C[C@@H]1O MGNNYOODZCAHBA-GQKYHHCASA-N 0.000 claims description 6
- 229940071648 metered dose inhaler Drugs 0.000 claims description 6
- 229940044601 receptor agonist Drugs 0.000 claims description 6
- 239000000018 receptor agonist Substances 0.000 claims description 6
- WBYWAXJHAXSJNI-VOTSOKGWSA-M trans-cinnamate Chemical class [O-]C(=O)\C=C\C1=CC=CC=C1 WBYWAXJHAXSJNI-VOTSOKGWSA-M 0.000 claims description 6
- GIIZNNXWQWCKIB-UHFFFAOYSA-N Serevent Chemical compound C1=C(O)C(CO)=CC(C(O)CNCCCCCCOCCCCC=2C=CC=CC=2)=C1 GIIZNNXWQWCKIB-UHFFFAOYSA-N 0.000 claims description 5
- 229960005084 calcitriol Drugs 0.000 claims description 5
- 235000020964 calcitriol Nutrition 0.000 claims description 5
- 239000011612 calcitriol Substances 0.000 claims description 5
- GMRQFYUYWCNGIN-NKMMMXOESA-N calcitriol Chemical compound C1(/[C@@H]2CC[C@@H]([C@]2(CCC1)C)[C@@H](CCCC(C)(C)O)C)=C\C=C1\C[C@@H](O)C[C@H](O)C1=C GMRQFYUYWCNGIN-NKMMMXOESA-N 0.000 claims description 5
- 239000007788 liquid Substances 0.000 claims description 5
- 229960001664 mometasone Drugs 0.000 claims description 5
- QLIIKPVHVRXHRI-CXSFZGCWSA-N mometasone Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@]2(Cl)[C@@H]1[C@@H]1C[C@@H](C)[C@@](C(=O)CCl)(O)[C@@]1(C)C[C@@H]2O QLIIKPVHVRXHRI-CXSFZGCWSA-N 0.000 claims description 5
- 230000002085 persistent effect Effects 0.000 claims description 5
- 229940044551 receptor antagonist Drugs 0.000 claims description 5
- 239000002464 receptor antagonist Substances 0.000 claims description 5
- 229960004017 salmeterol Drugs 0.000 claims description 5
- 230000002459 sustained effect Effects 0.000 claims description 5
- 206010014561 Emphysema Diseases 0.000 claims description 4
- 229940121948 Muscarinic receptor antagonist Drugs 0.000 claims description 4
- 206010006451 bronchitis Diseases 0.000 claims description 4
- POULHZVOKOAJMA-UHFFFAOYSA-N dodecanoic acid Chemical class CCCCCCCCCCCC(O)=O POULHZVOKOAJMA-UHFFFAOYSA-N 0.000 claims description 4
- 239000003120 macrolide antibiotic agent Substances 0.000 claims description 4
- 229940041033 macrolides Drugs 0.000 claims description 4
- 239000003149 muscarinic antagonist Substances 0.000 claims description 4
- 150000002942 palmitic acid derivatives Chemical class 0.000 claims description 4
- 150000003431 steroids Chemical class 0.000 claims description 4
- 229960000278 theophylline Drugs 0.000 claims description 4
- SHGAZHPCJJPHSC-NUEINMDLSA-N Isotretinoin Chemical compound OC(=O)C=C(C)/C=C/C=C(C)C=CC1=C(C)CCCC1(C)C SHGAZHPCJJPHSC-NUEINMDLSA-N 0.000 claims description 3
- 102100029438 Nitric oxide synthase, inducible Human genes 0.000 claims description 3
- 101710089543 Nitric oxide synthase, inducible Proteins 0.000 claims description 3
- 229940049937 Pgp inhibitor Drugs 0.000 claims description 3
- 239000003963 antioxidant agent Substances 0.000 claims description 3
- 229940092705 beclomethasone Drugs 0.000 claims description 3
- NBMKJKDGKREAPL-DVTGEIKXSA-N beclomethasone Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@]2(Cl)[C@@H]1[C@@H]1C[C@H](C)[C@@](C(=O)CO)(O)[C@@]1(C)C[C@@H]2O NBMKJKDGKREAPL-DVTGEIKXSA-N 0.000 claims description 3
- FAOZLTXFLGPHNG-KNAQIMQKSA-N fluorometholone Chemical compound C([C@@]12C)=CC(=O)C=C1[C@@H](C)C[C@@H]1[C@]2(F)[C@@H](O)C[C@]2(C)[C@@](O)(C(C)=O)CC[C@H]21 FAOZLTXFLGPHNG-KNAQIMQKSA-N 0.000 claims description 3
- 229960001048 fluorometholone Drugs 0.000 claims description 3
- 239000002748 glycoprotein P inhibitor Substances 0.000 claims description 3
- 239000013386 metal-inorganic framework Substances 0.000 claims description 3
- 239000006199 nebulizer Substances 0.000 claims description 3
- 229960005294 triamcinolone Drugs 0.000 claims description 3
- GFNANZIMVAIWHM-OBYCQNJPSA-N triamcinolone Chemical compound O=C1C=C[C@]2(C)[C@@]3(F)[C@@H](O)C[C@](C)([C@@]([C@H](O)C4)(O)C(=O)CO)[C@@H]4[C@@H]3CCC2=C1 GFNANZIMVAIWHM-OBYCQNJPSA-N 0.000 claims description 3
- 206010003598 Atelectasis Diseases 0.000 claims description 2
- 229940121710 HMGCoA reductase inhibitor Drugs 0.000 claims description 2
- 239000012825 JNK inhibitor Substances 0.000 claims description 2
- YTFHCXIPDIHOIA-DHZHZOJOSA-N Palovarotene Chemical compound C1=CC=NN1CC=1C=C2C(C)(C)CCC(C)(C)C2=CC=1\C=C\C1=CC=C(C(O)=O)C=C1 YTFHCXIPDIHOIA-DHZHZOJOSA-N 0.000 claims description 2
- 206010035664 Pneumonia Diseases 0.000 claims description 2
- 208000007123 Pulmonary Atelectasis Diseases 0.000 claims description 2
- 230000003078 antioxidant effect Effects 0.000 claims description 2
- 239000002471 hydroxymethylglutaryl coenzyme A reductase inhibitor Substances 0.000 claims description 2
- 229940065725 leukotriene receptor antagonists for obstructive airway diseases Drugs 0.000 claims description 2
- 239000003199 leukotriene receptor blocking agent Substances 0.000 claims description 2
- 102000002574 p38 Mitogen-Activated Protein Kinases Human genes 0.000 claims description 2
- 108010068338 p38 Mitogen-Activated Protein Kinases Proteins 0.000 claims description 2
- 229950000473 palovarotene Drugs 0.000 claims description 2
- 239000004031 partial agonist Substances 0.000 claims description 2
- 239000002571 phosphodiesterase inhibitor Substances 0.000 claims description 2
- 239000007787 solid Substances 0.000 claims description 2
- 229940125388 beta agonist Drugs 0.000 claims 4
- 206010058467 Lung neoplasm malignant Diseases 0.000 claims 1
- 229940122696 MAP kinase inhibitor Drugs 0.000 claims 1
- 229940099471 Phosphodiesterase inhibitor Drugs 0.000 claims 1
- 229940124158 Protease/peptidase inhibitor Drugs 0.000 claims 1
- 206010037423 Pulmonary oedema Diseases 0.000 claims 1
- 239000003966 growth inhibitor Substances 0.000 claims 1
- 239000003630 growth substance Substances 0.000 claims 1
- 201000005202 lung cancer Diseases 0.000 claims 1
- 208000020816 lung neoplasm Diseases 0.000 claims 1
- 239000002829 mitogen activated protein kinase inhibitor Substances 0.000 claims 1
- 208000005333 pulmonary edema Diseases 0.000 claims 1
- 239000000168 bronchodilator agent Substances 0.000 abstract description 4
- IPCSVZSSVZVIGE-UHFFFAOYSA-M hexadecanoate Chemical compound CCCCCCCCCCCCCCCC([O-])=O IPCSVZSSVZVIGE-UHFFFAOYSA-M 0.000 abstract description 3
- 102000035195 Peptidases Human genes 0.000 abstract description 2
- 108091005804 Peptidases Proteins 0.000 abstract description 2
- 239000004365 Protease Substances 0.000 abstract description 2
- 229940042399 direct acting antivirals protease inhibitors Drugs 0.000 abstract description 2
- POULHZVOKOAJMA-UHFFFAOYSA-M dodecanoate Chemical compound CCCCCCCCCCCC([O-])=O POULHZVOKOAJMA-UHFFFAOYSA-M 0.000 abstract description 2
- 229940070765 laurate Drugs 0.000 abstract description 2
- BZLVMXJERCGZMT-UHFFFAOYSA-N Methyl tert-butyl ether Chemical compound COC(C)(C)C BZLVMXJERCGZMT-UHFFFAOYSA-N 0.000 description 34
- 230000000694 effects Effects 0.000 description 26
- 238000011282 treatment Methods 0.000 description 22
- 239000003814 drug Substances 0.000 description 21
- SNIBJKHIKIIGPR-UHFFFAOYSA-N N-desethyloxybutynin Chemical compound C=1C=CC=CC=1C(O)(C(=O)OCC#CCNCC)C1CCCCC1 SNIBJKHIKIIGPR-UHFFFAOYSA-N 0.000 description 14
- 229940079593 drug Drugs 0.000 description 13
- 230000037317 transdermal delivery Effects 0.000 description 13
- PZSMUPGANZGPBF-UHFFFAOYSA-N 4-[5-(dithiolan-3-yl)pentanoylamino]butanoic acid Chemical compound OC(=O)CCCNC(=O)CCCCC1CCSS1 PZSMUPGANZGPBF-UHFFFAOYSA-N 0.000 description 12
- 229940125389 long-acting beta agonist Drugs 0.000 description 12
- 208000005069 pulmonary fibrosis Diseases 0.000 description 11
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 10
- 238000005481 NMR spectroscopy Methods 0.000 description 10
- 230000015572 biosynthetic process Effects 0.000 description 10
- 230000002685 pulmonary effect Effects 0.000 description 10
- 150000001875 compounds Chemical class 0.000 description 8
- 238000001704 evaporation Methods 0.000 description 8
- 230000008020 evaporation Effects 0.000 description 8
- 206010066218 Stress Urinary Incontinence Diseases 0.000 description 7
- 206010046543 Urinary incontinence Diseases 0.000 description 7
- 229930002330 retinoic acid Natural products 0.000 description 7
- 208000024891 symptom Diseases 0.000 description 7
- 230000001225 therapeutic effect Effects 0.000 description 7
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 6
- 241000700198 Cavia Species 0.000 description 6
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 6
- 241001465754 Metazoa Species 0.000 description 6
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 6
- 239000004480 active ingredient Substances 0.000 description 6
- 238000002425 crystallisation Methods 0.000 description 6
- 208000002551 irritable bowel syndrome Diseases 0.000 description 6
- 239000008101 lactose Substances 0.000 description 6
- 239000002207 metabolite Substances 0.000 description 6
- NZWOPGCLSHLLPA-UHFFFAOYSA-N methacholine Chemical compound C[N+](C)(C)CC(C)OC(C)=O NZWOPGCLSHLLPA-UHFFFAOYSA-N 0.000 description 6
- 208000023504 respiratory system disease Diseases 0.000 description 6
- 230000005068 transpiration Effects 0.000 description 6
- 206010006482 Bronchospasm Diseases 0.000 description 5
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical class Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 5
- 230000007885 bronchoconstriction Effects 0.000 description 5
- 230000008025 crystallization Effects 0.000 description 5
- JMMWKPVZQRWMSS-UHFFFAOYSA-N isopropyl acetate Chemical compound CC(C)OC(C)=O JMMWKPVZQRWMSS-UHFFFAOYSA-N 0.000 description 5
- 229960002329 methacholine Drugs 0.000 description 5
- 231100000252 nontoxic Toxicity 0.000 description 5
- 230000003000 nontoxic effect Effects 0.000 description 5
- 239000002904 solvent Substances 0.000 description 5
- 238000003786 synthesis reaction Methods 0.000 description 5
- 229940124597 therapeutic agent Drugs 0.000 description 5
- IHOXNOQMRZISPV-YJYMSZOUSA-N 5-[(1r)-1-hydroxy-2-[[(2r)-1-(4-methoxyphenyl)propan-2-yl]azaniumyl]ethyl]-2-oxo-1h-quinolin-8-olate Chemical compound C1=CC(OC)=CC=C1C[C@@H](C)NC[C@H](O)C1=CC=C(O)C2=C1C=CC(=O)N2 IHOXNOQMRZISPV-YJYMSZOUSA-N 0.000 description 4
- 206010021639 Incontinence Diseases 0.000 description 4
- 238000010521 absorption reaction Methods 0.000 description 4
- 229950010713 carmoterol Drugs 0.000 description 4
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 4
- 230000014759 maintenance of location Effects 0.000 description 4
- 238000002844 melting Methods 0.000 description 4
- 230000008018 melting Effects 0.000 description 4
- 238000012986 modification Methods 0.000 description 4
- 230000004048 modification Effects 0.000 description 4
- 238000000634 powder X-ray diffraction Methods 0.000 description 4
- 230000009885 systemic effect Effects 0.000 description 4
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 4
- 238000001061 Dunnett's test Methods 0.000 description 3
- 208000000059 Dyspnea Diseases 0.000 description 3
- 206010013975 Dyspnoeas Diseases 0.000 description 3
- VPNYRYCIDCJBOM-UHFFFAOYSA-M Glycopyrronium bromide Chemical compound [Br-].C1[N+](C)(C)CCC1OC(=O)C(O)(C=1C=CC=CC=1)C1CCCC1 VPNYRYCIDCJBOM-UHFFFAOYSA-M 0.000 description 3
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 3
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 3
- 230000009471 action Effects 0.000 description 3
- 238000000540 analysis of variance Methods 0.000 description 3
- 230000001684 chronic effect Effects 0.000 description 3
- 230000034994 death Effects 0.000 description 3
- 231100000517 death Toxicity 0.000 description 3
- 201000010099 disease Diseases 0.000 description 3
- 238000004090 dissolution Methods 0.000 description 3
- 229940015042 glycopyrrolate Drugs 0.000 description 3
- 229910052757 nitrogen Inorganic materials 0.000 description 3
- 230000002688 persistence Effects 0.000 description 3
- 238000002360 preparation method Methods 0.000 description 3
- 229930010796 primary metabolite Natural products 0.000 description 3
- 208000022170 stress incontinence Diseases 0.000 description 3
- FPIPGXGPPPQFEQ-UHFFFAOYSA-N 13-cis retinol Natural products OCC=C(C)C=CC=C(C)C=CC1=C(C)CCCC1(C)C FPIPGXGPPPQFEQ-UHFFFAOYSA-N 0.000 description 2
- ZNOVTXRBGFNYRX-UHFFFAOYSA-N 2-[[4-[(2-amino-5-methyl-4-oxo-1,6,7,8-tetrahydropteridin-6-yl)methylamino]benzoyl]amino]pentanedioic acid Chemical compound C1NC=2NC(N)=NC(=O)C=2N(C)C1CNC1=CC=C(C(=O)NC(CCC(O)=O)C(O)=O)C=C1 ZNOVTXRBGFNYRX-UHFFFAOYSA-N 0.000 description 2
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 2
- 206010001052 Acute respiratory distress syndrome Diseases 0.000 description 2
- 241000700199 Cavia porcellus Species 0.000 description 2
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 2
- 238000005033 Fourier transform infrared spectroscopy Methods 0.000 description 2
- 206010052402 Gastrointestinal hypermotility Diseases 0.000 description 2
- 241000124008 Mammalia Species 0.000 description 2
- 102000014415 Muscarinic acetylcholine receptor Human genes 0.000 description 2
- 108050003473 Muscarinic acetylcholine receptor Proteins 0.000 description 2
- 208000000921 Urge Urinary Incontinence Diseases 0.000 description 2
- FPIPGXGPPPQFEQ-BOOMUCAASA-N Vitamin A Natural products OC/C=C(/C)\C=C\C=C(\C)/C=C/C1=C(C)CCCC1(C)C FPIPGXGPPPQFEQ-BOOMUCAASA-N 0.000 description 2
- 208000005946 Xerostomia Diseases 0.000 description 2
- 230000002411 adverse Effects 0.000 description 2
- FPIPGXGPPPQFEQ-OVSJKPMPSA-N all-trans-retinol Chemical compound OC\C=C(/C)\C=C\C=C(/C)\C=C\C1=C(C)CCCC1(C)C FPIPGXGPPPQFEQ-OVSJKPMPSA-N 0.000 description 2
- 230000001078 anti-cholinergic effect Effects 0.000 description 2
- 235000006708 antioxidants Nutrition 0.000 description 2
- 230000004872 arterial blood pressure Effects 0.000 description 2
- 239000012298 atmosphere Substances 0.000 description 2
- 239000008280 blood Substances 0.000 description 2
- 210000004369 blood Anatomy 0.000 description 2
- 230000036765 blood level Effects 0.000 description 2
- 229940046731 calcineurin inhibitors Drugs 0.000 description 2
- 238000006243 chemical reaction Methods 0.000 description 2
- 238000001816 cooling Methods 0.000 description 2
- 230000002939 deleterious effect Effects 0.000 description 2
- 239000003937 drug carrier Substances 0.000 description 2
- 206010013781 dry mouth Diseases 0.000 description 2
- 238000002474 experimental method Methods 0.000 description 2
- 239000006260 foam Substances 0.000 description 2
- 230000002496 gastric effect Effects 0.000 description 2
- 239000000499 gel Substances 0.000 description 2
- 238000004128 high performance liquid chromatography Methods 0.000 description 2
- 238000001727 in vivo Methods 0.000 description 2
- 230000005764 inhibitory process Effects 0.000 description 2
- 238000002347 injection Methods 0.000 description 2
- 239000007924 injection Substances 0.000 description 2
- 208000018936 intestinal hypermotility Diseases 0.000 description 2
- 230000037036 intestinal hypermotility Effects 0.000 description 2
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 2
- 230000004060 metabolic process Effects 0.000 description 2
- 230000004118 muscle contraction Effects 0.000 description 2
- 229940021182 non-steroidal anti-inflammatory drug Drugs 0.000 description 2
- 239000003960 organic solvent Substances 0.000 description 2
- 239000008177 pharmaceutical agent Substances 0.000 description 2
- 230000036470 plasma concentration Effects 0.000 description 2
- 230000000069 prophylactic effect Effects 0.000 description 2
- 208000002815 pulmonary hypertension Diseases 0.000 description 2
- 102000005962 receptors Human genes 0.000 description 2
- 108020003175 receptors Proteins 0.000 description 2
- 210000003079 salivary gland Anatomy 0.000 description 2
- 230000000391 smoking effect Effects 0.000 description 2
- 230000035882 stress Effects 0.000 description 2
- 208000011580 syndromic disease Diseases 0.000 description 2
- 230000001839 systemic circulation Effects 0.000 description 2
- 238000002560 therapeutic procedure Methods 0.000 description 2
- 210000001519 tissue Anatomy 0.000 description 2
- 231100000027 toxicology Toxicity 0.000 description 2
- 206010046494 urge incontinence Diseases 0.000 description 2
- 235000019155 vitamin A Nutrition 0.000 description 2
- 239000011719 vitamin A Substances 0.000 description 2
- 229940045997 vitamin a Drugs 0.000 description 2
- ZKNJEOBYOLUGKJ-ALCCZGGFSA-N (z)-2-propylpent-2-enoic acid Chemical compound CCC\C(C(O)=O)=C\CC ZKNJEOBYOLUGKJ-ALCCZGGFSA-N 0.000 description 1
- 238000005160 1H NMR spectroscopy Methods 0.000 description 1
- 208000033116 Asbestos intoxication Diseases 0.000 description 1
- 201000002909 Aspergillosis Diseases 0.000 description 1
- 208000036641 Aspergillus infections Diseases 0.000 description 1
- 239000002028 Biomass Substances 0.000 description 1
- 201000004569 Blindness Diseases 0.000 description 1
- 229940127291 Calcium channel antagonist Drugs 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 1
- WBYWAXJHAXSJNI-SREVYHEPSA-N Cinnamic acid Chemical compound OC(=O)\C=C/C1=CC=CC=C1 WBYWAXJHAXSJNI-SREVYHEPSA-N 0.000 description 1
- 208000035473 Communicable disease Diseases 0.000 description 1
- 206010010774 Constipation Diseases 0.000 description 1
- 229920002261 Corn starch Polymers 0.000 description 1
- 201000003883 Cystic fibrosis Diseases 0.000 description 1
- 241000228402 Histoplasma Species 0.000 description 1
- 241000342334 Human metapneumovirus Species 0.000 description 1
- 206010020853 Hypertonic bladder Diseases 0.000 description 1
- 229940110339 Long-acting muscarinic antagonist Drugs 0.000 description 1
- 206010025219 Lymphangioma Diseases 0.000 description 1
- 206010027406 Mesothelioma Diseases 0.000 description 1
- NTIZESTWPVYFNL-UHFFFAOYSA-N Methyl isobutyl ketone Chemical compound CC(C)CC(C)=O NTIZESTWPVYFNL-UHFFFAOYSA-N 0.000 description 1
- 208000007101 Muscle Cramp Diseases 0.000 description 1
- 241000186359 Mycobacterium Species 0.000 description 1
- 206010028813 Nausea Diseases 0.000 description 1
- 208000005119 Necrotizing Pneumonia Diseases 0.000 description 1
- 241000150452 Orthohantavirus Species 0.000 description 1
- 208000009722 Overactive Urinary Bladder Diseases 0.000 description 1
- 206010033557 Palpitations Diseases 0.000 description 1
- 208000031481 Pathologic Constriction Diseases 0.000 description 1
- 208000011191 Pulmonary vascular disease Diseases 0.000 description 1
- 208000013616 Respiratory Distress Syndrome Diseases 0.000 description 1
- 206010038687 Respiratory distress Diseases 0.000 description 1
- 201000003176 Severe Acute Respiratory Syndrome Diseases 0.000 description 1
- 201000010001 Silicosis Diseases 0.000 description 1
- 206010040880 Skin irritation Diseases 0.000 description 1
- 208000005392 Spasm Diseases 0.000 description 1
- 208000034972 Sudden Infant Death Diseases 0.000 description 1
- 206010042440 Sudden infant death syndrome Diseases 0.000 description 1
- 208000001871 Tachycardia Diseases 0.000 description 1
- 206010046555 Urinary retention Diseases 0.000 description 1
- 241000700605 Viruses Species 0.000 description 1
- 206010047513 Vision blurred Diseases 0.000 description 1
- MECHNRXZTMCUDQ-UHFFFAOYSA-N Vitamin D2 Chemical class C1CCC2(C)C(C(C)C=CC(C)C(C)C)CCC2C1=CC=C1CC(O)CCC1=C MECHNRXZTMCUDQ-UHFFFAOYSA-N 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 201000000028 adult respiratory distress syndrome Diseases 0.000 description 1
- 239000000443 aerosol Substances 0.000 description 1
- 238000012387 aerosolization Methods 0.000 description 1
- 230000000172 allergic effect Effects 0.000 description 1
- 229940065524 anticholinergics inhalants for obstructive airway diseases Drugs 0.000 description 1
- 206010003441 asbestosis Diseases 0.000 description 1
- 208000010668 atopic eczema Diseases 0.000 description 1
- 229940124748 beta 2 agonist Drugs 0.000 description 1
- 210000000621 bronchi Anatomy 0.000 description 1
- 201000009267 bronchiectasis Diseases 0.000 description 1
- 210000003123 bronchiole Anatomy 0.000 description 1
- 230000007883 bronchodilation Effects 0.000 description 1
- 230000000178 bronchoprotective effect Effects 0.000 description 1
- 206010006475 bronchopulmonary dysplasia Diseases 0.000 description 1
- 239000000480 calcium channel blocker Substances 0.000 description 1
- 229940082638 cardiac stimulant phosphodiesterase inhibitors Drugs 0.000 description 1
- 150000003841 chloride salts Chemical class 0.000 description 1
- 239000000812 cholinergic antagonist Substances 0.000 description 1
- 230000001713 cholinergic effect Effects 0.000 description 1
- 229930016911 cinnamic acid Natural products 0.000 description 1
- 235000013985 cinnamic acid Nutrition 0.000 description 1
- 230000008602 contraction Effects 0.000 description 1
- 239000008120 corn starch Substances 0.000 description 1
- 239000013078 crystal Substances 0.000 description 1
- 239000002178 crystalline material Substances 0.000 description 1
- 238000011461 current therapy Methods 0.000 description 1
- 238000000354 decomposition reaction Methods 0.000 description 1
- 238000002716 delivery method Methods 0.000 description 1
- 238000001514 detection method Methods 0.000 description 1
- FSBVERYRVPGNGG-UHFFFAOYSA-N dimagnesium dioxido-bis[[oxido(oxo)silyl]oxy]silane hydrate Chemical compound O.[Mg+2].[Mg+2].[O-][Si](=O)O[Si]([O-])([O-])O[Si]([O-])=O FSBVERYRVPGNGG-UHFFFAOYSA-N 0.000 description 1
- 208000035475 disorder Diseases 0.000 description 1
- 229940099170 ditropan Drugs 0.000 description 1
- 208000002173 dizziness Diseases 0.000 description 1
- 230000004530 effect on cardiovascular disease Effects 0.000 description 1
- 201000009580 eosinophilic pneumonia Diseases 0.000 description 1
- 229960002061 ergocalciferol Drugs 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- 235000019439 ethyl acetate Nutrition 0.000 description 1
- 230000029142 excretion Effects 0.000 description 1
- 239000012065 filter cake Substances 0.000 description 1
- 238000010579 first pass effect Methods 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 229960000289 fluticasone propionate Drugs 0.000 description 1
- WMWTYOKRWGGJOA-CENSZEJFSA-N fluticasone propionate Chemical compound C1([C@@H](F)C2)=CC(=O)C=C[C@]1(C)[C@]1(F)[C@@H]2[C@@H]2C[C@@H](C)[C@@](C(=O)SCF)(OC(=O)CC)[C@@]2(C)C[C@@H]1O WMWTYOKRWGGJOA-CENSZEJFSA-N 0.000 description 1
- 239000012458 free base Substances 0.000 description 1
- 239000000446 fuel Substances 0.000 description 1
- 230000004927 fusion Effects 0.000 description 1
- 229940040165 gelnique Drugs 0.000 description 1
- 238000011554 guinea pig model Methods 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 150000004677 hydrates Chemical class 0.000 description 1
- 230000036571 hydration Effects 0.000 description 1
- 238000006703 hydration reaction Methods 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 239000005414 inactive ingredient Substances 0.000 description 1
- 230000028709 inflammatory response Effects 0.000 description 1
- 229940125369 inhaled corticosteroids Drugs 0.000 description 1
- 230000000968 intestinal effect Effects 0.000 description 1
- 230000001788 irregular Effects 0.000 description 1
- 238000002955 isolation Methods 0.000 description 1
- 229960005280 isotretinoin Drugs 0.000 description 1
- 208000017169 kidney disease Diseases 0.000 description 1
- 229940043355 kinase inhibitor Drugs 0.000 description 1
- 238000011068 loading method Methods 0.000 description 1
- 239000003589 local anesthetic agent Substances 0.000 description 1
- 229940127212 long-acting beta 2 agonist Drugs 0.000 description 1
- 230000007774 longterm Effects 0.000 description 1
- 239000000391 magnesium silicate Substances 0.000 description 1
- 229910052919 magnesium silicate Inorganic materials 0.000 description 1
- 235000019792 magnesium silicate Nutrition 0.000 description 1
- 235000019359 magnesium stearate Nutrition 0.000 description 1
- 238000012423 maintenance Methods 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 239000012528 membrane Substances 0.000 description 1
- 230000000116 mitigating effect Effects 0.000 description 1
- 230000008693 nausea Effects 0.000 description 1
- 230000031990 negative regulation of inflammatory response Effects 0.000 description 1
- 230000000414 obstructive effect Effects 0.000 description 1
- QIQXTHQIDYTFRH-UHFFFAOYSA-M octadecanoate Chemical class CCCCCCCCCCCCCCCCCC([O-])=O QIQXTHQIDYTFRH-UHFFFAOYSA-M 0.000 description 1
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical class CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 description 1
- 230000008520 organization Effects 0.000 description 1
- 208000020629 overactive bladder Diseases 0.000 description 1
- SWIJYDAEGSIQPZ-UHFFFAOYSA-N oxybutynin chloride Chemical compound [H+].[Cl-].C=1C=CC=CC=1C(O)(C(=O)OCC#CCN(CC)CC)C1CCCCC1 SWIJYDAEGSIQPZ-UHFFFAOYSA-N 0.000 description 1
- 229960002016 oxybutynin chloride Drugs 0.000 description 1
- 229940023569 palmate Drugs 0.000 description 1
- WLJNZVDCPSBLRP-UHFFFAOYSA-N pamoic acid Chemical compound C1=CC=C2C(CC=3C4=CC=CC=C4C=C(C=3O)C(=O)O)=C(O)C(C(O)=O)=CC2=C1 WLJNZVDCPSBLRP-UHFFFAOYSA-N 0.000 description 1
- 239000000825 pharmaceutical preparation Substances 0.000 description 1
- 239000003757 phosphotransferase inhibitor Substances 0.000 description 1
- 230000007084 physiological dysfunction Effects 0.000 description 1
- 239000012254 powdered material Substances 0.000 description 1
- 238000001556 precipitation Methods 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- 239000000651 prodrug Substances 0.000 description 1
- 229940002612 prodrug Drugs 0.000 description 1
- 239000000047 product Substances 0.000 description 1
- 230000002035 prolonged effect Effects 0.000 description 1
- 238000011321 prophylaxis Methods 0.000 description 1
- 201000009732 pulmonary eosinophilia Diseases 0.000 description 1
- 239000011802 pulverized particle Substances 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 230000000241 respiratory effect Effects 0.000 description 1
- 230000029058 respiratory gaseous exchange Effects 0.000 description 1
- 238000012552 review Methods 0.000 description 1
- 201000000306 sarcoidosis Diseases 0.000 description 1
- 238000004062 sedimentation Methods 0.000 description 1
- 229940125390 short-acting beta agonist Drugs 0.000 description 1
- 208000013220 shortness of breath Diseases 0.000 description 1
- 230000036556 skin irritation Effects 0.000 description 1
- 231100000475 skin irritation Toxicity 0.000 description 1
- 201000002859 sleep apnea Diseases 0.000 description 1
- 210000002460 smooth muscle Anatomy 0.000 description 1
- 210000000329 smooth muscle myocyte Anatomy 0.000 description 1
- 230000002048 spasmolytic effect Effects 0.000 description 1
- 238000005507 spraying Methods 0.000 description 1
- 230000036262 stenosis Effects 0.000 description 1
- 208000037804 stenosis Diseases 0.000 description 1
- 238000012916 structural analysis Methods 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 230000009747 swallowing Effects 0.000 description 1
- 238000001308 synthesis method Methods 0.000 description 1
- 235000020357 syrup Nutrition 0.000 description 1
- 239000006188 syrup Substances 0.000 description 1
- 230000006794 tachycardia Effects 0.000 description 1
- 239000000454 talc Substances 0.000 description 1
- 229910052623 talc Inorganic materials 0.000 description 1
- 235000012222 talc Nutrition 0.000 description 1
- 238000012360 testing method Methods 0.000 description 1
- 230000004797 therapeutic response Effects 0.000 description 1
- 238000004448 titration Methods 0.000 description 1
- 230000000699 topical effect Effects 0.000 description 1
- 238000012546 transfer Methods 0.000 description 1
- 229960002117 triamcinolone acetonide Drugs 0.000 description 1
- YNDXUCZADRHECN-JNQJZLCISA-N triamcinolone acetonide Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@]2(F)[C@@H]1[C@@H]1C[C@H]3OC(C)(C)O[C@@]3(C(=O)CO)[C@@]1(C)C[C@@H]2O YNDXUCZADRHECN-JNQJZLCISA-N 0.000 description 1
- 201000008827 tuberculosis Diseases 0.000 description 1
- 239000003981 vehicle Substances 0.000 description 1
- 239000011653 vitamin D2 Chemical class 0.000 description 1
- 235000001892 vitamin D2 Nutrition 0.000 description 1
- QYSXJUFSXHHAJI-YRZJJWOYSA-N vitamin D3 Chemical class C1(/[C@@H]2CC[C@@H]([C@]2(CCC1)C)[C@H](C)CCCC(C)C)=C\C=C1\C[C@@H](O)CCC1=C QYSXJUFSXHHAJI-YRZJJWOYSA-N 0.000 description 1
- 239000011647 vitamin D3 Chemical class 0.000 description 1
- 235000005282 vitamin D3 Nutrition 0.000 description 1
- 229940021056 vitamin d3 Drugs 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/007—Pulmonary tract; Aromatherapy
- A61K9/0073—Sprays or powders for inhalation; Aerolised or nebulised preparations generated by other means than thermal energy
- A61K9/0075—Sprays or powders for inhalation; Aerolised or nebulised preparations generated by other means than thermal energy for inhalation via a dry powder inhaler [DPI], e.g. comprising micronized drug mixed with lactose carrier particles
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/21—Esters, e.g. nitroglycerine, selenocyanates
- A61K31/215—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids
- A61K31/216—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids of acids having aromatic rings, e.g. benactizyne, clofibrate
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
- A61P11/06—Antiasthmatics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
- A61P11/08—Bronchodilators
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/12—Antihypertensives
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Veterinary Medicine (AREA)
- Chemical & Material Sciences (AREA)
- Public Health (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Pharmacology & Pharmacy (AREA)
- Pulmonology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Organic Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Epidemiology (AREA)
- Otolaryngology (AREA)
- Emergency Medicine (AREA)
- Heart & Thoracic Surgery (AREA)
- Cardiology (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
- Medicinal Preparation (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
도 1에서는 18시간째에서 옥시부티닌 및 옥시부티닌 염의 메타콜린 유도된 기관지 수축의 억제를 플롯팅한다. 보다 구체적으로, 옥시부티닌 치료된 동물에 대한 기관지 수축에서의 변화를 ANOVA 및 후속 던네트(Dunnetts) 시험을 사용하여 시간 정합된(time matched) 락토오스 대조 동물과 비교하였다. ** P<0.01.
도 2에서는 18시간째 및 24시간째에서 옥시부티닌, 옥시부티닌 염, 티오트로피움 및 글리코피롤레이트에 의한 메타콜린 유도된 기관지 수축의 억제를 비교한다. 보다 구체적으로, 도 2에서는 락토오스(2 mg, i.t., n=6), 옥시부티닌 염기(2 mg, i.t., n=6), 옥시부티닌 HCl(2.5 mg, i.t., n=6), 옥시부티닌 시나포에이트(3 mg, i.t., n=6), 티오트로피움(1 mg/kg, i.t., n=6) 또는 글리코피롤레이트(1 mg/kg, i.t., n=6) 투여 후 18 h, 및 24 h째의 메타콜린(MCh, 10 μg/kg, i.v.)에 의해 유발된 기관지 수축의 비교를 플롯팅한다. 각 바(bar)는 평균값을 나타내고 수직선은 표준 오차 평균(s.e. mean)을 나타낸다. 옥시부티닌 또는 티오트로피움 처리된 동물(마취된 기니 피그)에 대한 기관지 수축에서의 변화를 ANOVA 및 후속 던네트 시험을 사용하여 시간 정합된 락토오스 대조 동물과 비교하였다. ** P<0.01.
도 3에서는, 폐 팽창압, 평균 동맥압 및 심박수에서, 옥시부티닌 시나포에이트 및 티오트로피움에 의한, 시간에 대한 메타콜린에 의해 유발된 대조 반응으로부터 변화를 비교하는 일련의 그래프이다. 보다 구체적으로, 도 3에서는 폐 팽창압(PIP), 평균 동맥압(MAP) 및 심박수(HR)에서, 락토오스(i.t., n=6), 옥시부티닌 시나포에이트(7.5% w/w, i.t., n=6), 또는 티오트로피움(1 mg, i.t., n=6)의 존재 하에 시간(h)에 대한 메타콜린(10 μg kg-1, i.v.)에 의해 유발된 대조 반응으로부터 변화의 비교를 플롯팅한다. 각 점은 평균값을 나타내고 수직선은 표준 오차 평균을 나타낸다. 옥시부티닌 또는 티오트로피움 처리된 동물에서의 백분율 변화를 ANOVA 및 후속 던네트 시험을 사용하여 각각의 락토오스 대조 동물(마취된 기니 피그)과 비교하였다. * P<0.05, ** P<0.01.
도 4에서는 시간에 대한 옥시부티닌의 폐 투여의 약동학을 나타낸다. 보다 구체적으로, 도 4에서는 건조 분말 흡입에 이은 옥시부티닌, 옥시부티닌 에난티오머, 및 주요 대사산물(데스에틸옥시부티닌)의 마취된 기니 피그(n=5) 약동학 프로필을 플롯팅한다.
도 5 및 도 6은 1H NMR 및 FT-IR 구조 분석을 제공한다.
도 7에서는 HPLC 분석을 제공한다.
도 8 및 도 9에서는 XRPD 및 DSC에 의해 측정된 결정도, 화합물 순도, 및 융점을 제공한다.
| 용매 | 용매 유형 | 옥시부티닌 중량 (mg) | 용매 부피 | 온도(℃) | 결과 |
| THF | II | 20.4 | 5 | 23 | 가용성 |
| 메틸 THF | II | 18.4 | 5 | 23 | 가용성 |
| i-PrOAc | III | 20.7 | 5 | 23 | 가용성 |
| EtOAc | III | 19.5 | 5 | 23 | 가용성 |
| MTBE | III | 21.7 | 5 | 30 | 가용성; 재결정 없음 |
| 톨루엔 | II | 22.0 | 5 | 23 | 가용성 |
| 에탄올 | III | 21.9 | 5 | 31 | 가용성; 재결정 없음 |
| 2-프로판올 | III | 20.8 | 5 | 39 | 가용성; 재결정 없음 |
| 아세톤 | III | 20.8 | 5 | 23 | 가용성 |
| 메탄올 | II | 18.1 | 5 | 23 | 가용성 |
| 항목 (규모) |
산 (당량) |
용매(1) (부피) |
용매(2) (부피) |
온도 (℃) |
단리 | % 회수 |
비고 |
| 1 (0.5g) |
시나포산 (1) |
MTBE (3부피) |
EtOH (4) |
45 | 증발 | 94 | 폼; NMR에 의한 1:1 염; 1.2% 잔류 EtOH |
| 2 (0.5g) |
시나포산 (1) |
MTBE (3부피) |
MTBE (5.4) |
50 | 5℃로 냉각; 이후 증발 |
92 | 폼; NMR에 의한 1:1 염; 3% 잔류 MTBE |
| 3 (0.5g) |
시나포산 (1) |
i-PrOAc (6부피) |
없음 | 50 | 5℃로 냉각; 이후 증발 |
101 | 오일; NMR에 의한 1:1 염; 5.5% 잔류 IPAc |
| 4 (0.5g) |
시나포산 (1) |
2-PrOH (10부피) |
없음 | 80 | 증발 | 86 | 오일; NMR에 의한 1:1 염 |
| 5 (0.5g) |
시나포산 (1) |
메틸 THF (10부피) |
없음 | 80 | 증발 | 106 | 오일; NMR에 의한 1:1 염 |
| 6 (0.5g) |
시나포산 (1) |
톨루엔 (10부피) |
없음 | 80 | 증발 | 112 | 오일; NMR에 의한 1:1 염 |
| 7 (0.5g) |
시나포산 (1) |
MIBK (10부피) |
없음 | 80 | 증발 | 118 | 오일; NMR에 의한 1:1 염 |
| 8 (0.5g) |
시나포산 (1) |
물 (10부피) |
2-PrOH (10) |
80 | 증발 | 99 | 오일; NMR에 의한 1:1 염 |
| 9 (3g) |
시나포산 (1) |
MTBE (3부피) |
EtOH (4) |
50 | 결정화 | 77 | 1:1 염; 0.16% 잔류 MTBE; XRPD에 의한 결정질; DSC: 104-106℃ |
| 10 (3g) |
시나포산 (1) |
MTBE (5부피) |
없음 | 50 | 결정화 | 89 | 1:1 염; 0.23% 잔류 MTBE; XRPD에 의한 결정질; DSC: 104-106℃ |
| 11 (20g) |
시나포산 (1) |
MTBE (5부피) |
없음 | 50 | 결정화 | 89 | NMR에 의한 1:1 염; 0.27% 잔류 MTBE; 0.057% 잔류 H2O(KF) |
Claims (15)
- 치료적 유효량의 옥시부티닌을 하나 이상의 약학적으로 유효한 제제와의 조합으로 환자의 폐에 직접 전달하는 것을 포함하는 폐질환을 치료하는 방법.
- 제1항에 있어서, 옥시부티닌 및/또는 약학적으로 유효한 제제는 건조 분말 형태로 전달되는 것인 방법.
- 제1항 또는 제2항에 있어서, 건조 분말 옥시부티닌은 시나포에이트 염, 팔미테이트 염, 파모산 염, 레조네이트(resonate) 염 및 라우레이트 염으로 이루어진 군으로부터 선택되는 것인 방법.
- 제1항 또는 제2항에 있어서, 옥시부티닌은 옥시부티닌 시나포에이트를 포함하는 것인 방법.
- 제1항 내지 제4항 중 어느 하나의 항에 있어서, 약학적으로 유효한 제제는 기관지 확장제, 소염제, 코르티코스테로이드, 흡입형 코르티코스테로이드, 코르티코스테로이드 역전제, 폐포 생장제, 프로테이나제 억제제, 또는 프로테아제 억제제를 포함하는 것인 방법.
- 제5항에 있어서, 약학적으로 유효한 제제는 지속성 베타 작용제 및 속효성 베타 작용제 및 이들의 유도체 또는 약학적으로 허용 가능한 염을 포함하는 기관지 확장제, 또는 흡입형 코르티코스테로이드, 포스포디에스테라제 억제제 또는 류코트리엔 수용체 길항제, 또는 부데소니드, 플루티카손, 베클로메타손, 플루니솔리드, 트리암시놀론, 시클레소니드, 로테프레드놀, 플루오로메톨론, 및 이들의 유도체 또는 약학적으로 허용 가능한 염을 포함하는 코르티코스테로이드, 또는 비타민 D, 합성 비타민 D, 비타민 D 유사체, 비타민 D 수용체 작용제, 비타민 D 수용체 부분 작용제, 칼시트리올, 항산화제, iNOS 억제제, 포스포이노시티드-3-키나제-델타 억제제, p38 MAP 키나제 억제제, JNK 억제제, MIF 억제제, 저용량 테오필린, p-당단백질 억제제, 매크로라이드, 칼시뉴린 억제제, 스타틴 및 이의 동등물을 포함하는 코르티코스테로이드 역전제, 또는 비타민 A, 올 트랜스 레티노산(All Trans Retinoic Acid, ATRA), 레티노산 수용체(RAR) 작용제 및 RAR 선택적 폐포 생장제, RAR 선택적 작용제, 팔로바로텐(palovarotene) 및 이의 동등물을 포함하는 폐포 생장제로부터 선택되는 것인 방법.
- 제6항에 있어서, 옥시부티닌은 옥시부티닌 시노포에이트(xinofoate)를 포함하고, 지속성 베타 작용제는 포르모테롤, 살메테롤, 오달라테롤(odalaterol), 카르모테롤 또는 빌란테롤을 포함하는 것인 방법.
- 제7항에 있어서, 추가로 흡입형 코르티코스테로이드를 포함하고, 흡입형 코르티코스테로이드는 부데소니드, 플루티카손, 또는 모메타손을 포함하며, 임의로 소프트 스테로이드류로부터 선택된 선택적 제제(selective agent) 및/또는 CR 역전제를 추가로 포함하며, 소프트 스테로이드류는 시클레소니드 또는 로테프레드놀을 포함하고, CR 역전제는 비타민 D, 비타민 D 유사체, 합성 비타민 D, 비타민 D 수용체 작용제 및 길항제, 칼시톨(calcitol) 및 이의 동등물로 이루어진 군으로부터 선택되는 것인 방법.
- 제1항에 있어서, 옥시부티닌은 옥시부티닌 시노포에이트를 포함하고, 약학적으로 유효한 제제는
포르모테롤을 포함하는 지속성 베타 작용제, 및
ATRA, 시스-레티오노산(cis-retionoic acid) 및 팔로바로텐으로 이루어진 군으로부터 선택된 폐포 생장제
를 포함하는 것인 방법. - 제1항 내지 제9항 중 어느 하나의 항에 있어서, 폐질환은 천식, 무기폐, 기관지염, 만성 폐쇄성 폐질환, 폐기종, 폐암, 폐렴 또는 폐부종을 포함하는 것인 방법.
- 제1항에 있어서, 폐질환은 만성 폐쇄성 폐질환을 포함하고,
옥시부티닌은 옥시부티닌 시노포에이트를 포함하며,
약학적으로 유효한 제제는 지속성 무스카린 길항제를 포함하는 것인 방법. - 제1항 내지 제11항 중 어느 하나의 항에 있어서, 옥시부티닌 및 약학적으로 유효한 제제는 건조 분말 흡입기(dry powder inhaler, DPI) 또는 정량 흡입기(metered dose inhaler, MDI) 또는 액체 네뷸라이저(liquid nebulizer)를 사용하여 전달되는 것인 방법.
- 제12항에 있어서, 건조 분말 흡입기는 압전 진동자(piezo vibrator)를 포함하는 것인 방법.
- 제1항 내지 제13항 중 어느 하나의 항에 있어서, 옥시부티닌은 0.5 - 20 마이크론, 0.5 - 15 마이크론, 0.5 - 10 마이크론, 또는 0.5 - 5 마이크론으로 이루어진 군으로부터 선택된 공기역학적 질량 중앙 입자 크기를 갖는 건조 분말 형태로 전달되고/되거나, 하나 이상의 약학적으로 유효한 제제와 조합되는 치료적 유효량의 옥시부티닌의 복용량은 1일당 0.001 내지 20 mg, 1일당 0.02 내지 15 mg, 또는 1일당 0.05 내지 10 mg 범위 내로 필요한 만큼 투여되는 것인 방법.
- 치료적 유효량의 옥시부티닌을 하나 이상의 약학적으로 유효한 제제와의 조합으로 환자의 폐에 직접 전달하는 것을 포함하는 만성 폐쇄성 폐질환을 치료하는 방법으로서, 옥시부티닌은 바람직하게는 시나포에이트 염, 팔미테이트 염, 파모산 염, 레조네이트 염 및 라우레이트 염으로 이루어진 군으로부터 선택되는 것인 방법.
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US13/728,706 | 2012-12-27 | ||
| US13/728,706 US9119777B2 (en) | 2008-05-30 | 2012-12-27 | Methods and compositions for administration of oxybutynin |
| PCT/US2013/074759 WO2014105446A1 (en) | 2012-12-27 | 2013-12-12 | Methods and compositions for administration of oxybutynin |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| KR20150100902A true KR20150100902A (ko) | 2015-09-02 |
Family
ID=51021931
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020157020335A Ceased KR20150100902A (ko) | 2012-12-27 | 2013-12-12 | 옥시부티닌의 투여를 위한 방법 및 조성물 |
Country Status (15)
| Country | Link |
|---|---|
| EP (1) | EP2938329A4 (ko) |
| JP (1) | JP2016504358A (ko) |
| KR (1) | KR20150100902A (ko) |
| CN (1) | CN104955444A (ko) |
| AR (1) | AR094287A1 (ko) |
| AU (1) | AU2013368298B2 (ko) |
| BR (1) | BR112015015421A2 (ko) |
| CA (1) | CA2895955A1 (ko) |
| EA (1) | EA201591218A1 (ko) |
| HK (2) | HK1215396A1 (ko) |
| IL (1) | IL239623A0 (ko) |
| MX (1) | MX2015008333A (ko) |
| NZ (1) | NZ628479A (ko) |
| UY (1) | UY35230A (ko) |
| WO (1) | WO2014105446A1 (ko) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20180091041A (ko) * | 2016-02-25 | 2018-08-14 | 히사미쓰 세이야꾸 가부시키가이샤 | 외용 액제 |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US12448347B2 (en) * | 2020-05-05 | 2025-10-21 | Apnimed, Inc. (Delaware) | Polymorphic forms of (R)-oxybutynin hydrochloride |
Family Cites Families (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| AU767877B2 (en) * | 1999-05-20 | 2003-11-27 | Sepracor, Inc. | Methods for treatment of asthma using S-oxybutynin |
| US6339107B1 (en) * | 2000-08-02 | 2002-01-15 | Syntex (U.S.A.) Llc | Methods for treatment of Emphysema using 13-cis retinoic acid |
| AU2003267796A1 (en) * | 2002-10-29 | 2004-05-25 | Pharmacia & Upjohn Company Llc | Quaternary ammonium compounds |
| GB0410398D0 (en) * | 2004-05-10 | 2004-06-16 | Arakis Ltd | The treatment of respiratory disease |
| US20060110449A1 (en) * | 2004-10-25 | 2006-05-25 | Lorber Richard R | Pharmaceutical composition |
| US8415390B2 (en) * | 2008-05-30 | 2013-04-09 | Microdose Therapeutx, Inc. | Methods and compositions for administration of oxybutynin |
| GB2468073B (en) * | 2008-02-26 | 2012-09-05 | Elevation Pharmaceuticals Inc | Method and system for the treatment of chronic obstructive pulmonary disease with nebulized anticholinergic administrations |
| AU2010260513A1 (en) * | 2009-06-16 | 2012-02-02 | Wen Tan | Use of R-Bambuterol as inhaled medicament and combination therapies for treatment of respiratory disorders |
| GB0918450D0 (en) * | 2009-10-21 | 2009-12-09 | Innovata Ltd | Composition |
| BR112012024059B1 (pt) * | 2010-04-01 | 2021-06-01 | Chiesi Farmaceutici S.P.A. | Processo de preparo de um excipiente para composições farmacêuticas em pó para inalação, partículas carreadoras para uma formulação farmacêutica de pó seco, composição farmacêutica em forma de pó seco para inalação e inalador de pó seco |
| JP2013538830A (ja) * | 2010-09-27 | 2013-10-17 | マイクロドース セラピューテクス,インコーポレイテッド | 吸入を用いる疾患治療のための方法及び組成物 |
-
2013
- 2013-12-12 HK HK16103431.3A patent/HK1215396A1/zh unknown
- 2013-12-12 JP JP2015550444A patent/JP2016504358A/ja active Pending
- 2013-12-12 EP EP13867956.8A patent/EP2938329A4/en not_active Withdrawn
- 2013-12-12 KR KR1020157020335A patent/KR20150100902A/ko not_active Ceased
- 2013-12-12 AU AU2013368298A patent/AU2013368298B2/en not_active Ceased
- 2013-12-12 CN CN201380068230.5A patent/CN104955444A/zh active Pending
- 2013-12-12 EA EA201591218A patent/EA201591218A1/ru unknown
- 2013-12-12 NZ NZ628479A patent/NZ628479A/en not_active IP Right Cessation
- 2013-12-12 WO PCT/US2013/074759 patent/WO2014105446A1/en not_active Ceased
- 2013-12-12 CA CA2895955A patent/CA2895955A1/en not_active Abandoned
- 2013-12-12 MX MX2015008333A patent/MX2015008333A/es unknown
- 2013-12-12 HK HK16104715.8A patent/HK1216843A1/zh unknown
- 2013-12-12 BR BR112015015421A patent/BR112015015421A2/pt not_active IP Right Cessation
- 2013-12-20 UY UY0001035230A patent/UY35230A/es not_active Application Discontinuation
- 2013-12-27 AR ARP130105037A patent/AR094287A1/es unknown
-
2015
- 2015-06-24 IL IL239623A patent/IL239623A0/en unknown
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20180091041A (ko) * | 2016-02-25 | 2018-08-14 | 히사미쓰 세이야꾸 가부시키가이샤 | 외용 액제 |
Also Published As
| Publication number | Publication date |
|---|---|
| HK1216843A1 (zh) | 2016-12-09 |
| JP2016504358A (ja) | 2016-02-12 |
| NZ628479A (en) | 2016-06-24 |
| BR112015015421A2 (pt) | 2017-07-11 |
| IL239623A0 (en) | 2015-08-31 |
| WO2014105446A1 (en) | 2014-07-03 |
| UY35230A (es) | 2015-01-30 |
| AU2013368298B2 (en) | 2016-08-11 |
| CA2895955A1 (en) | 2014-07-03 |
| EP2938329A4 (en) | 2016-08-10 |
| HK1215396A1 (zh) | 2016-08-26 |
| EP2938329A1 (en) | 2015-11-04 |
| EA201591218A1 (ru) | 2015-11-30 |
| CN104955444A (zh) | 2015-09-30 |
| AU2013368298A1 (en) | 2015-07-02 |
| AR094287A1 (es) | 2015-07-22 |
| MX2015008333A (es) | 2015-11-09 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| TWI272106B (en) | New medicament compositions based on anticholinergically-effective compounds and beta-mimetics | |
| CN102946868B (zh) | 包含抗毒蕈碱药的干粉制剂 | |
| US20150238456A1 (en) | Methods and compositions for administration of oxybutynin | |
| EP3377064B1 (en) | Orvepitant for the treatment of chronic cough | |
| AU2008259864C1 (en) | Methods and compositions for administration of Oxybutynin | |
| CN106470700A (zh) | 用于治疗copd的噻托溴铵、福莫特罗和布地奈德的组合 | |
| AU2005315337B2 (en) | Pharmaceutical compounds and compositions | |
| US8748488B2 (en) | Methods and compositions for administration of oxybutynin | |
| MX2010014223A (es) | Composiciones pediatricas para el tratamiento de esclerosis multiple. | |
| US20060189642A1 (en) | Synergistic combination comprising roflumilast and an anticholinergic agent selected from ipratropium, oxitropium and tiotropium salts for the treatment of respiratory diseases | |
| AU2013368298B2 (en) | Methods and compositions for administration of oxybutynin | |
| WO2010097114A1 (en) | Novel combination of therapeutic agents | |
| JP7048629B2 (ja) | 医薬組成物およびその使用 | |
| RU2440972C2 (ru) | Кристаллический сульфат левосальбутамола, способ его получения и фармацевтическая композиция, содержащая его | |
| KR20050094810A (ko) | 로플루미래스트와 r,r-포르모테롤을 포함하는 상승작용성조합물 | |
| WO2004084894A1 (en) | Synergistic combination comprising roflumilast and revatropate for the treatment of respiratory diseases | |
| JP2000302761A (ja) | モルファン誘導体またはその塩 | |
| HK1232788A1 (en) | Combinations of tiotropium bromide, formoterol and budesonide for the treatment of copd |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A201 | Request for examination | ||
| PA0105 | International application |
Patent event date: 20150727 Patent event code: PA01051R01D Comment text: International Patent Application |
|
| PA0201 | Request for examination | ||
| PG1501 | Laying open of application | ||
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
Comment text: Notification of reason for refusal Patent event date: 20160927 Patent event code: PE09021S01D |
|
| E601 | Decision to refuse application | ||
| PE0601 | Decision on rejection of patent |
Patent event date: 20170217 Comment text: Decision to Refuse Application Patent event code: PE06012S01D Patent event date: 20160927 Comment text: Notification of reason for refusal Patent event code: PE06011S01I |