KR20170052702A - N-(4-{〔6,7-비스(메틸옥시)퀴놀린-4-일〕옥시}페닐)-n'-(4-플루오로페닐)사이클로프로판-1,1-디카르복사미드의 말산염 및 그 결정형 - Google Patents
N-(4-{〔6,7-비스(메틸옥시)퀴놀린-4-일〕옥시}페닐)-n'-(4-플루오로페닐)사이클로프로판-1,1-디카르복사미드의 말산염 및 그 결정형 Download PDFInfo
- Publication number
- KR20170052702A KR20170052702A KR1020177011913A KR20177011913A KR20170052702A KR 20170052702 A KR20170052702 A KR 20170052702A KR 1020177011913 A KR1020177011913 A KR 1020177011913A KR 20177011913 A KR20177011913 A KR 20177011913A KR 20170052702 A KR20170052702 A KR 20170052702A
- Authority
- KR
- South Korea
- Prior art keywords
- compound
- quinolin
- methyloxy
- bis
- cyclopropane
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- OGFLDKVKRARCNN-UHFFFAOYSA-N CC(C1(CC1)C(Nc(cc1)ccc1F)=O)Nc(cc1)ccc1Oc1c(cc(c(OC)c2)OC)c2ncc1 Chemical compound CC(C1(CC1)C(Nc(cc1)ccc1F)=O)Nc(cc1)ccc1Oc1c(cc(c(OC)c2)OC)c2ncc1 OGFLDKVKRARCNN-UHFFFAOYSA-N 0.000 description 1
- WRVHQEYBCDPZEU-UHFFFAOYSA-N COc(c(OC)cc1ncc2)cc1c2Cl Chemical compound COc(c(OC)cc1ncc2)cc1c2Cl WRVHQEYBCDPZEU-UHFFFAOYSA-N 0.000 description 1
- QOGPNCUTXVZQSL-UHFFFAOYSA-N COc(c(OC)cc1ncc2)cc1c2O Chemical compound COc(c(OC)cc1ncc2)cc1c2O QOGPNCUTXVZQSL-UHFFFAOYSA-N 0.000 description 1
- VXEQRXJATQUJSN-UHFFFAOYSA-N COc(c(OC)cc1ncc2)cc1c2Oc(cc1)ccc1N Chemical compound COc(c(OC)cc1ncc2)cc1c2Oc(cc1)ccc1N VXEQRXJATQUJSN-UHFFFAOYSA-N 0.000 description 1
- YFINVJAUWMDAJX-UHFFFAOYSA-N COc(c(OC)cc1ncc2)cc1c2Oc(cc1)ccc1NC(CCCCC(Nc(cc1)ccc1F)=O)=O Chemical compound COc(c(OC)cc1ncc2)cc1c2Oc(cc1)ccc1NC(CCCCC(Nc(cc1)ccc1F)=O)=O YFINVJAUWMDAJX-UHFFFAOYSA-N 0.000 description 1
- YRYKZXCOGVCXBN-UHFFFAOYSA-N COc(c(OC)cc1ncc2)cc1c2Oc(cc1)ccc1[N+]([O-])=O Chemical compound COc(c(OC)cc1ncc2)cc1c2Oc(cc1)ccc1[N+]([O-])=O YRYKZXCOGVCXBN-UHFFFAOYSA-N 0.000 description 1
- KRZCOLNOCZKSDF-UHFFFAOYSA-N Nc(cc1)ccc1F Chemical compound Nc(cc1)ccc1F KRZCOLNOCZKSDF-UHFFFAOYSA-N 0.000 description 1
- JQWTVIFNRAALHM-UHFFFAOYSA-N O=C(C1(CC1)C(Cl)=O)Nc(cc1)ccc1F Chemical compound O=C(C1(CC1)C(Cl)=O)Nc(cc1)ccc1F JQWTVIFNRAALHM-UHFFFAOYSA-N 0.000 description 1
- CTFRYFUQPJDALN-UHFFFAOYSA-N OC(C1(CC1)C(NC1C=CC(F)=CC1)=O)=O Chemical compound OC(C1(CC1)C(NC1C=CC(F)=CC1)=O)=O CTFRYFUQPJDALN-UHFFFAOYSA-N 0.000 description 1
- BTJIUGUIPKRLHP-UHFFFAOYSA-N [O-][N+](c(cc1)ccc1O)=O Chemical compound [O-][N+](c(cc1)ccc1O)=O BTJIUGUIPKRLHP-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D215/00—Heterocyclic compounds containing quinoline or hydrogenated quinoline ring systems
- C07D215/02—Heterocyclic compounds containing quinoline or hydrogenated quinoline ring systems having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen atoms or carbon atoms directly attached to the ring nitrogen atom
- C07D215/16—Heterocyclic compounds containing quinoline or hydrogenated quinoline ring systems having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen atoms or carbon atoms directly attached to the ring nitrogen atom with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D215/20—Oxygen atoms
- C07D215/22—Oxygen atoms attached in position 2 or 4
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/47—Quinolines; Isoquinolines
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P5/00—Drugs for disorders of the endocrine system
- A61P5/14—Drugs for disorders of the endocrine system of the thyroid hormones, e.g. T3, T4
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D215/00—Heterocyclic compounds containing quinoline or hydrogenated quinoline ring systems
- C07D215/02—Heterocyclic compounds containing quinoline or hydrogenated quinoline ring systems having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen atoms or carbon atoms directly attached to the ring nitrogen atom
- C07D215/16—Heterocyclic compounds containing quinoline or hydrogenated quinoline ring systems having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen atoms or carbon atoms directly attached to the ring nitrogen atom with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D215/20—Oxygen atoms
- C07D215/22—Oxygen atoms attached in position 2 or 4
- C07D215/233—Oxygen atoms attached in position 2 or 4 only one oxygen atom which is attached in position 4
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D417/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00
- C07D417/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07B—GENERAL METHODS OF ORGANIC CHEMISTRY; APPARATUS THEREFOR
- C07B2200/00—Indexing scheme relating to specific properties of organic compounds
- C07B2200/13—Crystalline forms, e.g. polymorphs
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Epidemiology (AREA)
- Diabetes (AREA)
- Endocrinology (AREA)
- Biomedical Technology (AREA)
- Neurology (AREA)
- Neurosurgery (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Quinoline Compounds (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Low-Molecular Organic Synthesis Reactions Using Catalysts (AREA)
Abstract
Description
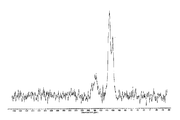
도 2는 결정형 화합물 (I), 제형 N-1의 고체 13C NMR 스펙트럼을 도시한 도이다.
도 3은 결정형 화합물 (I), 제형 N-1의 고체 15N NMR 스펙트럼을 도시한 도이다.
도 4는 결정형 화합물 (I), 제형 N-1의 고체 19F NMR 스펙트럼을 도시한 도이다.
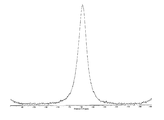
도 5는 결정형 화합물 (I), 제형 N-1의 열 무게측정 분석(thermal gravimetric analysis, TGA)을 도시한 도이다.
도 6은 결정형 화합물 (I), 제형 N-1의 시차 주사 열량계(differential scanning calorimetry, DSC)를 도시한 도이다.
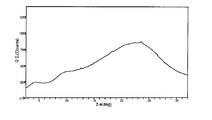
도 7은 결정형 화합물 (I), 제형 N-1의 흡습/탈습성(moisture sorption)을 도시한 도이다.
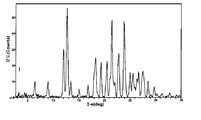
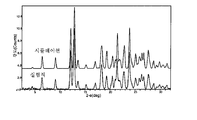
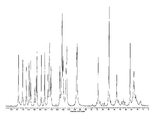
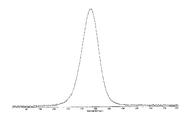
도 8은 25℃에서 결정형 화합물 (I), 제형 N-2의 실험적 XRPD 패턴을 도시한 도이다.
도 9는 결정형 화합물 (I), 제형 N-2의 고체 13C NMR 스펙트럼을 도시한 도이다.
도 10은 결정형 화합물 (I), 제형 N-2의 고체 15N NMR 스펙트럼을 도시한 도이다.
도 11은 결정형 화합물 (I), 제형 N-2의 고체 19F NMR 스펙트럼을 도시한 도이다.
도 12는 결정형 화합물 (I), 제형 N-2의 열 무게측정 분석(thermal gravimetric analysis, TGA)을 도시한 도이다.
도 13은 결정형 화합물 (I), 제형 N-2의 시차 주사 열량계(differential scanning calorimetry, DSC)를 도시한 도이다.
도 14는 결정형 화합물 (I), 제형 N-2의 흡습/탈습성(moisture sorption)을 도시한 도이다.
도 15는 실온에서 결정형 화합물 (III), 제형 N-1의 실험적 XRPD 패턴을 도시한 도이다.
도 16은 결정형 화합물 (III), 제형 N-1의 고체 13C NMR 스펙트럼을 도시한 도이다.
도 17은 결정형 화합물 (III), 제형 N-1의 고체 15N NMR 스펙트럼을 도시한 도이다.
도 18은 결정형 화합물 (III), 제형 N-1의 고체 19F NMR 스펙트럼을 도시한 도이다.
도 19는 결정형 화합물 (III), 제형 N-1의 열 무게측정 분석(thermal gravimetric analysis, TGA)을 도시한 도이다.
도 20은 결정형 화합물 (III), 제형 N-1의 시차 주사 열량계(differential scanning calorimetry, DSC)를 도시한 도이다.
도 21은 결정형 화합물 (III), 제형 N-1의 흡습/탈습성(moisture sorption)을 도시한 도이다.
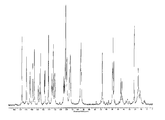
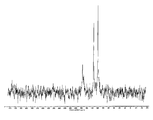
도 22는 실온에서 비결정형 화합물 (I)의 XRPD 패턴을 도시한 도이다.
도 23은 비결정형 화합물 (I)의 고체 13C NMR 스펙트럼을 도시한 도이다.
도 24는 비결정형 화합물 (I)의 고체 15N NMR 스펙트럼을 도시한 도이다.
도 25는 비결정형 화합물 (I)의 고체 19F NMR 스펙트럼을 도시한 도이다.
도 26은 비결정형 화합물 (I)의 시차 주사 열량계(differential scanning calorimetry, DSC)를 도시한 도이다.
도 27은 비결정형 화합물 (I)의 흡습/탈습성(moisture sorption)을 도시한 도이다.
Claims (15)
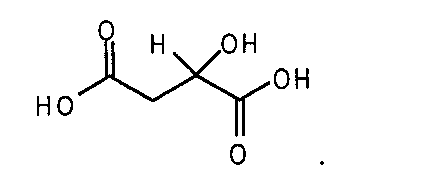
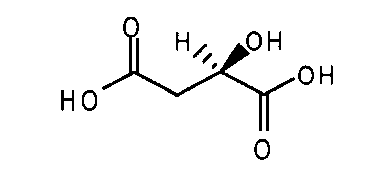
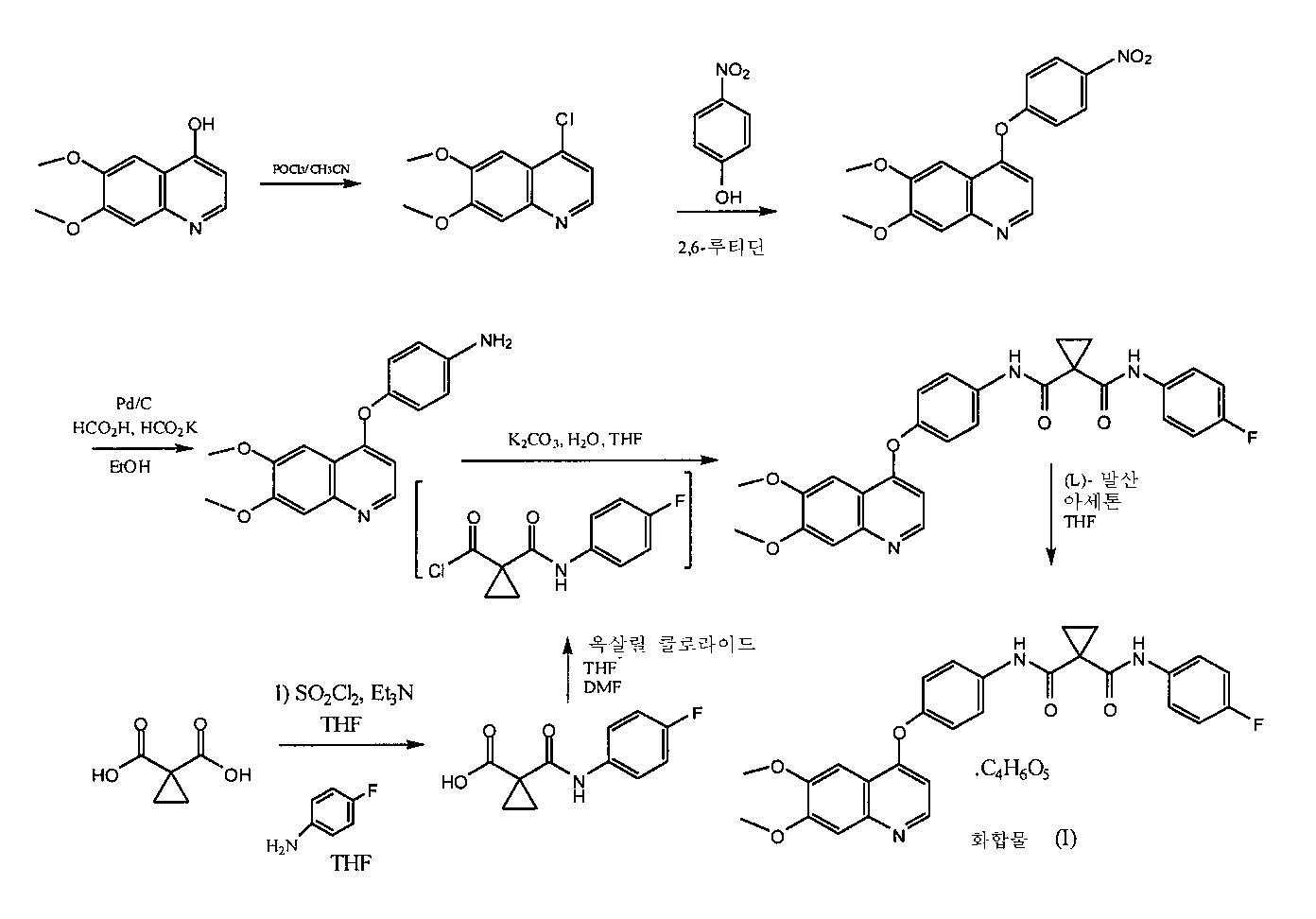
- N-(4-{[6,7-비스(메틸옥시)퀴놀린-4-일]옥시}페닐)-N'-(4-플루오로페닐)사이클로프로판-1,1-디카르복사미드의 말산염.
- 제 1항에 있어서, 상기 염이 (DL)-말산염인, N-(4-{[6,7-비스(메틸옥시)퀴놀린-4-일]옥시}페닐)-N'-(4-플루오로페닐)사이클로프로판-1,1-디카르복사미드의 말산염.
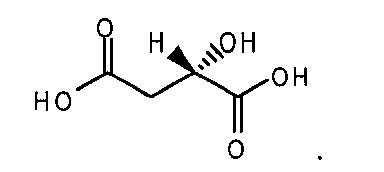
- 제 1항에 있어서, 상기 염이 (L)-말산염 또는 (D)-말산염인, N-(4-{[6,7-비스(메틸옥시)퀴놀린-4-일]옥시}페닐)-N'-(4-플루오로페닐)사이클로프로판-1,1-디카르복사미드의 말산염.
- 제 3항에 있어서, 상기 염이 (L)-말산염인, N-(4-{[6,7-비스(메틸옥시)퀴놀린-4-일]옥시}페닐)-N'-(4-플루오로페닐)사이클로프로판-1,1-디카르복사미드의 말산염.
- 제 3항에 있어서, 상기 염이 (D)-말산염인, N-(4-{[6,7-비스(메틸옥시)퀴놀린-4-일]옥시}페닐)-N'-(4-플루오로페닐)사이클로프로판-1,1-디카르복사미드의 말산염.
- 제 3항 내지 제 5항 중 어느 한 항에 있어서, 상기 염이 결정형인, N-(4-{[6,7-비스(메틸옥시)퀴놀린-4-일]옥시}페닐)-N'-(4-플루오로페닐)사이클로프로판-1,1-디카르복사미드의 말산염.
- 제 6항에 있어서, 상기 염이 N-1 결정형이고, 상기 N-1 결정형이
(i) 18.1, 42.9, 44.5, 70.4, 123.2, 156.2, 170.8, 175.7 및 182.1 ppm, ± 0.2 ppm에서 선택된 4개 이상의 피크가 나타나는 고체상 13C NMR 스펙트럼;
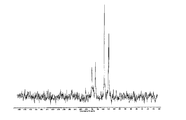
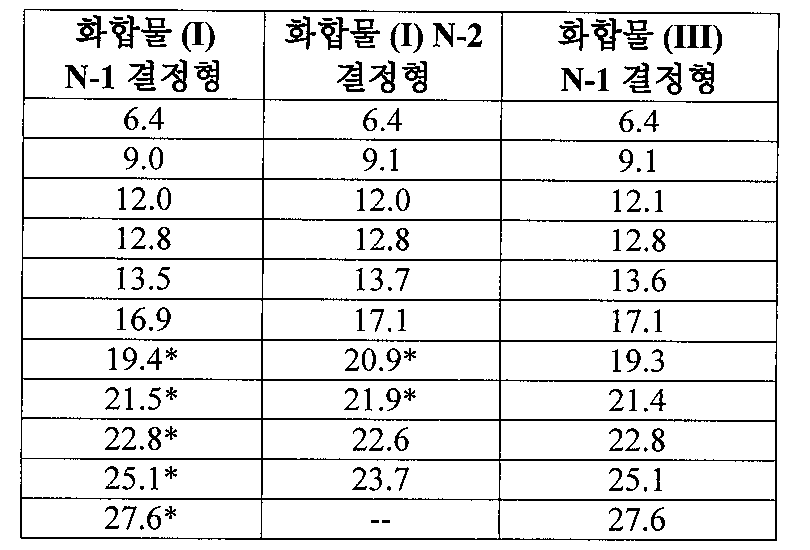
(ii) 실온에서 결정형 측정 시 12.8±0.2 °2θ, 13.5±0.2 °2θ, 16.9±0.2 °2θ, 19.4±0.2 °2θ, 21.5±0.2 °2θ, 22.8±0.2 °2θ, 25.1±0.2 °2θ, 27.6±0.2 °2θ에서 선택된 4개 이상의 2θ치로 이루어진 x-선 분말 회절 패턴(CuKα λ=1.5418A); 및/또는
(iii) 도 1에 나타난 패턴과 상당 부분 일치하는 x-선 분말 회절(XRPD) 스펙트럼 중 하나 이상에 의해 특징되는, N-(4-{[6,7-비스(메틸옥시)퀴놀린-4-일]옥시}페닐)-N'-(4-플루오로페닐)사이클로프로판-1,1-디카르복사미드의 말산염. - 제 7항에 있어서, 상기 염이 상기 염의 중량을 기준으로 하여 90 중량% 이상이 N-1 결정형인, N-(4-{[6,7-비스(메틸옥시)퀴놀린-4-일]옥시}페닐)-N'-(4-플루오로페닐)사이클로프로판-1,1-디카르복사미드의 말산염.
- 제 6항에 있어서, 상기 염이 N-2 결정형이고, 상기 N-2 결정형이
(i) 23.0, 25.9, 38.0, 41.7, 69.7, 102.0, 122.5, 177.3, 179.3, 180.0 및 180.3 ppm, ± 0.2 ppm에서 선택된 4개 이상의 피크가 나타나는 고체상 13C NMR 스펙트럼;
(ii) 실온에서 결정형 측정 시 20.9±0.2 °2θ 및 21.9±0.2 °2θ 등의 2θ치와 6.4±0.2 °2θ, 9.1±0.2 °2θ, 12.0±0.2 °2θ, 12.8±0.2, 13.7±0.2, 17.1±0.2, 22.6±0.2, 23.7±0.2에서 선택된 2개 이상의 2θ치로 이루어진 x-선 분말 회절 패턴(CuKα λ=1.5418A); 및/또는
(iii) 도 8에 나타난 패턴과 상당 부분 일치하는 x-선 분말 회절(XRPD) 스펙트럼 중 하나 이상에 의해 특징되는, N-(4-{[6,7-비스(메틸옥시)퀴놀린-4-일]옥시}페닐)-N'-(4-플루오로페닐)사이클로프로판-1,1-디카르복사미드의 말산염. - 제 9항에 있어서, 상기 염이 상기 염의 중량을 기준으로 하여 90 중량% 이상이 N-2 결정형인, N-(4-{[6,7-비스(메틸옥시)퀴놀린-4-일]옥시}페닐)-N'-(4-플루오로페닐)사이클로프로판-1,1-디카르복사미드의 말산염.
- 제 4항 내지 제 10항 중 어느 한 항에 따른 N-(4-{[6,7-비스(메틸옥시)퀴놀린-4-일]옥시}페닐)-N'-(4-플루오로페닐)사이클로프로판-1,1-디카르복사미드의 말산염, 및 약제학적으로 허용가능한 부형제를 포함하는 약제 조성물.
- 암 치료용 의약을 제조하기 위한, 제 3항 내지 제 10항 중 어느 한 항에 따른 N-(4-{[6,7-비스(메틸옥시)퀴놀린-4-일]옥시}페닐)-N'-(4-플루오로페닐)사이클로프로판-1,1-디카르복사미드의 말산염의 용도.
- 제 3항 내지 제 10항 중 어느 한 항에 있어서, 암 치료 요법에 사용하기 위한, N-(4-{[6,7-비스(메틸옥시)퀴놀린-4-일]옥시}페닐)-N'-(4-플루오로페닐)사이클로프로판-1,1-디카르복사미드의 말산염.
- 환자의 갑상선암 치료용 의약으로서 사용하기 위한, 제 6항 내지 제 10항 중 어느 한 항에 따른 N-(4-{[6,7-비스(메틸옥시)퀴놀린-4-일]옥시}페닐)-N'-(4-플루오로페닐)사이클로프로판-1,1-디카르복사미드의 (L)-말산염의 결정형.
- 환자의 교모세포종 치료용 의약으로서 사용하기 위한, 제 6항 내지 제 10항 중 어느 한 항에 따른 N-(4-{[6,7-비스(메틸옥시)퀴놀린-4-일]옥시}페닐)-N'-(4-플루오로페닐)사이클로프로판-1,1-디카르복사미드의 (L)-말산염의 결정형.
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US14542109P | 2009-01-16 | 2009-01-16 | |
| US61/145,421 | 2009-01-16 | ||
| PCT/US2010/021194 WO2010083414A1 (en) | 2009-01-16 | 2010-01-15 | Malate salt of n- (4- { [ 6, 7-bis (methyloxy) quin0lin-4-yl] oxy}phenyl-n' - (4 -fluorophenyl) cyclopropane-1-dicarboxamide, and crystalline forms therof for the treatment of cancer |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020117018924A Division KR101733773B1 (ko) | 2009-01-16 | 2010-01-15 | N-(4-{〔6,7-비스(메틸옥시)퀴놀린-4-일〕옥시}페닐)-n'-(4-플루오로페닐)사이클로프로판-1,1-디카르복사미드의 말산염 및 그 결정형 |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020187026467A Division KR102088588B1 (ko) | 2009-01-16 | 2010-01-15 | N-(4-{〔6,7-비스(메틸옥시)퀴놀린-4-일〕옥시}페닐)-n'-(4-플루오로페닐)사이클로프로판-1,1-디카르복사미드의 말산염 및 그 결정형 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| KR20170052702A true KR20170052702A (ko) | 2017-05-12 |
Family
ID=41820137
Family Applications (9)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020237000106A Ceased KR20230008268A (ko) | 2009-01-16 | 2010-01-15 | 암의 치료를 위한 n-(4-{〔6,7-비스(메틸옥시)퀴놀린-4-일〕옥시}페닐)-n'-(4-플루오로페닐)사이클로프로판-1,1-디카르복사미드의 말산염 및 그 결정형 |
| KR1020207006546A Active KR102187034B1 (ko) | 2009-01-16 | 2010-01-15 | 암 치료용 n-(4-{〔6,7-비스(메틸옥시)퀴놀린-4-일〕옥시}페닐)-n'-(4-플루오로페닐)사이클로프로판-1,1-디카르복사미드의 말산염 및 그 결정형 |
| KR1020247001034A Ceased KR20240011244A (ko) | 2009-01-16 | 2010-01-15 | 암의 치료를 위한 n-(4-{〔6,7-비스(메틸옥시)퀴놀린-4-일〕옥시}페닐)-n'-(4-플루오로페닐)사이클로프로판-1,1-디카르복사미드의 말산염 및 그 결정형 |
| KR1020177011913A Ceased KR20170052702A (ko) | 2009-01-16 | 2010-01-15 | N-(4-{〔6,7-비스(메틸옥시)퀴놀린-4-일〕옥시}페닐)-n'-(4-플루오로페닐)사이클로프로판-1,1-디카르복사미드의 말산염 및 그 결정형 |
| KR1020207034429A Ceased KR20200137052A (ko) | 2009-01-16 | 2010-01-15 | 암의 치료를 위한 n-(4-{〔6,7-비스(메틸옥시)퀴놀린-4-일〕옥시}페닐)-n'-(4-플루오로페닐)사이클로프로판-1,1-디카르복사미드의 말산염 및 그 결정형 |
| KR1020187026467A Active KR102088588B1 (ko) | 2009-01-16 | 2010-01-15 | N-(4-{〔6,7-비스(메틸옥시)퀴놀린-4-일〕옥시}페닐)-n'-(4-플루오로페닐)사이클로프로판-1,1-디카르복사미드의 말산염 및 그 결정형 |
| KR1020117018924A Active KR101733773B1 (ko) | 2009-01-16 | 2010-01-15 | N-(4-{〔6,7-비스(메틸옥시)퀴놀린-4-일〕옥시}페닐)-n'-(4-플루오로페닐)사이클로프로판-1,1-디카르복사미드의 말산염 및 그 결정형 |
| KR1020257026007A Pending KR20250123237A (ko) | 2009-01-16 | 2010-01-15 | 암의 치료를 위한 n-(4-{〔6,7-비스(메틸옥시)퀴놀린-4-일〕옥시}페닐)-n'-(4-플루오로페닐)사이클로프로판-1,1-디카르복사미드의 말산염 및 그 결정형 |
| KR1020217039169A Ceased KR20210151988A (ko) | 2009-01-16 | 2010-01-15 | 암의 치료를 위한 n-(4-{〔6,7-비스(메틸옥시)퀴놀린-4-일〕옥시}페닐)-n'-(4-플루오로페닐)사이클로프로판-1,1-디카르복사미드의 말산염 및 그 결정형 |
Family Applications Before (3)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020237000106A Ceased KR20230008268A (ko) | 2009-01-16 | 2010-01-15 | 암의 치료를 위한 n-(4-{〔6,7-비스(메틸옥시)퀴놀린-4-일〕옥시}페닐)-n'-(4-플루오로페닐)사이클로프로판-1,1-디카르복사미드의 말산염 및 그 결정형 |
| KR1020207006546A Active KR102187034B1 (ko) | 2009-01-16 | 2010-01-15 | 암 치료용 n-(4-{〔6,7-비스(메틸옥시)퀴놀린-4-일〕옥시}페닐)-n'-(4-플루오로페닐)사이클로프로판-1,1-디카르복사미드의 말산염 및 그 결정형 |
| KR1020247001034A Ceased KR20240011244A (ko) | 2009-01-16 | 2010-01-15 | 암의 치료를 위한 n-(4-{〔6,7-비스(메틸옥시)퀴놀린-4-일〕옥시}페닐)-n'-(4-플루오로페닐)사이클로프로판-1,1-디카르복사미드의 말산염 및 그 결정형 |
Family Applications After (5)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020207034429A Ceased KR20200137052A (ko) | 2009-01-16 | 2010-01-15 | 암의 치료를 위한 n-(4-{〔6,7-비스(메틸옥시)퀴놀린-4-일〕옥시}페닐)-n'-(4-플루오로페닐)사이클로프로판-1,1-디카르복사미드의 말산염 및 그 결정형 |
| KR1020187026467A Active KR102088588B1 (ko) | 2009-01-16 | 2010-01-15 | N-(4-{〔6,7-비스(메틸옥시)퀴놀린-4-일〕옥시}페닐)-n'-(4-플루오로페닐)사이클로프로판-1,1-디카르복사미드의 말산염 및 그 결정형 |
| KR1020117018924A Active KR101733773B1 (ko) | 2009-01-16 | 2010-01-15 | N-(4-{〔6,7-비스(메틸옥시)퀴놀린-4-일〕옥시}페닐)-n'-(4-플루오로페닐)사이클로프로판-1,1-디카르복사미드의 말산염 및 그 결정형 |
| KR1020257026007A Pending KR20250123237A (ko) | 2009-01-16 | 2010-01-15 | 암의 치료를 위한 n-(4-{〔6,7-비스(메틸옥시)퀴놀린-4-일〕옥시}페닐)-n'-(4-플루오로페닐)사이클로프로판-1,1-디카르복사미드의 말산염 및 그 결정형 |
| KR1020217039169A Ceased KR20210151988A (ko) | 2009-01-16 | 2010-01-15 | 암의 치료를 위한 n-(4-{〔6,7-비스(메틸옥시)퀴놀린-4-일〕옥시}페닐)-n'-(4-플루오로페닐)사이클로프로판-1,1-디카르복사미드의 말산염 및 그 결정형 |
Country Status (24)
| Country | Link |
|---|---|
| US (15) | US8877776B2 (ko) |
| EP (1) | EP2387563B2 (ko) |
| JP (6) | JP5677318B2 (ko) |
| KR (9) | KR20230008268A (ko) |
| CN (3) | CN106083714B (ko) |
| AR (1) | AR075025A1 (ko) |
| AU (3) | AU2010204619B2 (ko) |
| BR (1) | BRPI1006812B8 (ko) |
| CA (2) | CA2995880C (ko) |
| DK (1) | DK2387563T4 (ko) |
| EA (3) | EA019959B1 (ko) |
| ES (1) | ES2402524T5 (ko) |
| HR (1) | HRP20130458T4 (ko) |
| IL (2) | IL214086A (ko) |
| MX (3) | MX350898B (ko) |
| NZ (3) | NZ779754A (ko) |
| PL (1) | PL2387563T5 (ko) |
| PT (1) | PT2387563E (ko) |
| RS (1) | RS52754B2 (ko) |
| SG (1) | SG173014A1 (ko) |
| SM (1) | SMT201300050B (ko) |
| TW (7) | TW202112751A (ko) |
| WO (1) | WO2010083414A1 (ko) |
| ZA (1) | ZA201105167B (ko) |
Families Citing this family (66)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2888102B2 (ja) | 1993-08-26 | 1999-05-10 | 三菱電機株式会社 | 時間ダイバーシティ通信装置用送信機および受信機、並びに時間ダイバーシティ通信装置 |
| PL2210607T3 (pl) * | 2003-09-26 | 2012-01-31 | Exelixis Inc | N-[3-fluoro-4-({6-(metyloksy)-7-[(3-morfolin-4-ylopropylo)oksy]chinolin-4-ylo} oxy)fenylo]-N'-(4-fluorofenylo)cyklopropano-1,1-dikarboksamid do leczenia raka |
| US8877776B2 (en) | 2009-01-16 | 2014-11-04 | Exelixis, Inc. | (L)-malate salt of N-(4-{[6,7-bis(methyloxy) quinolin-4-yl]oxy}phenyl)-N'-(4-fluorophenyl)cyclopropane-1,1-dicarboxamide |
| UA108618C2 (uk) | 2009-08-07 | 2015-05-25 | Застосування c-met-модуляторів в комбінації з темозоломідом та/або променевою терапією для лікування раку | |
| US20120070368A1 (en) * | 2010-04-16 | 2012-03-22 | Exelixis, Inc. | Methods of Using C-Met Modulators |
| CN114209699A (zh) | 2010-07-16 | 2022-03-22 | 埃克塞里艾克西斯公司 | C-met调节剂药物组合物 |
| PL2621481T5 (pl) * | 2010-09-27 | 2023-03-13 | Exelixis, Inc. | Podwójne inhibitory MET i VEGF do leczenia opornego na kastrację raka prostaty i osteoblastycznych przerzutów do kości |
| KR20200031711A (ko) * | 2011-02-10 | 2020-03-24 | 엑셀리시스, 인코포레이티드 | 퀴놀린 화합물들의 제조 방법들 및 상기 화합물들을 함유하는 약학 조성물들 |
| US20120252840A1 (en) * | 2011-04-04 | 2012-10-04 | Exelixis, Inc. | Method of Treating Cancer |
| TW201806598A (zh) | 2011-05-02 | 2018-03-01 | 艾克塞里克斯公司 | 治療癌症及骨癌疼痛之方法 |
| AU2012312364B2 (en) * | 2011-09-22 | 2017-11-09 | Exelixis, Inc. | Method for treating osteoporosis |
| TWI642650B (zh) | 2011-10-20 | 2018-12-01 | 艾克塞里克斯公司 | 用於製備喹啉衍生物之方法 |
| JP6238311B2 (ja) | 2012-01-13 | 2017-11-29 | エックススプレイ ファーマ パブリーク・アクチエボラグXSpray Pharma AB(publ) | 少なくとも1種のプロテインキナーゼ阻害剤及び少なくとも1種のポリマー性安定化マトリックス形成性成分の安定な非晶質のハイブリッドナノ粒子を含む医薬組成物 |
| EP2844254A1 (en) | 2012-05-02 | 2015-03-11 | Exelixis, Inc. | A dual met - vegf modulator for treating osteolytic bone metastases |
| EP2892532B1 (en) * | 2012-09-07 | 2019-02-13 | Exelixis, Inc. | Inhibitors of met, vegfr and ret for use in the treatment of lung adenocarcinoma |
| CN103664776B (zh) * | 2012-09-26 | 2016-05-04 | 正大天晴药业集团股份有限公司 | 一种酪氨酸激酶抑制剂及其中间体的制备方法 |
| UA119321C2 (uk) | 2013-03-15 | 2019-06-10 | Екселіксіс, Інк. | Метаболіти n-(4-{[6,7-біс(метилокси)хінолін-4-іл]окси}феніл)-n'-(4-фторфеніл)циклопропан-1,1-дикарбоксаміду |
| ES2927651T3 (es) * | 2013-04-04 | 2022-11-10 | Exelixis Inc | Forma de dosificación de cabozantinib y uso en el tratamiento del cáncer |
| HK1221424A1 (zh) * | 2013-04-04 | 2017-06-02 | Exelixis, Inc. | 治疗癌症的药物组合 |
| CN104109124B (zh) * | 2013-04-19 | 2016-08-31 | 正大天晴药业集团股份有限公司 | 卡博替尼·0.5苹果酸盐的晶体 |
| CN104370811B (zh) * | 2013-08-15 | 2019-02-12 | 广东东阳光药业有限公司 | 一种喹啉化合物的新晶型及其制备方法 |
| CN104649969B (zh) * | 2013-11-22 | 2019-02-12 | 广东东阳光药业有限公司 | 一种替尼类药物的盐及其制备方法 |
| CN106068255A (zh) | 2014-02-14 | 2016-11-02 | 埃克塞里艾克西斯公司 | 结晶固体形式的n‑{4‑[(6,7‑二甲氧基喹啉‑4‑基)氧基]苯基}‑n′‑(4‑氟苯基)环丙烷‑1,1‑二甲酰胺、制备方法以及使用方法 |
| EA201691850A1 (ru) * | 2014-03-17 | 2016-12-30 | Экселиксис, Инк. | Дозирование составов, содержащих кабозантиниб |
| BR112016024672A2 (pt) * | 2014-04-25 | 2021-02-02 | Exelixis, Inc | método para o tratamento de adenocarcinoma de pulmão |
| WO2015177758A1 (en) | 2014-05-23 | 2015-11-26 | Mylan Laboratories Ltd | Novel polymorphs of cabozantinib (s)-malate and cabozantinib free base |
| CN104788372B (zh) * | 2014-07-25 | 2018-01-30 | 上海圣考医药科技有限公司 | 一种氘代卡博替尼衍生物、其制备方法、应用及其中间体 |
| CN106715397B (zh) | 2014-07-31 | 2021-07-23 | 埃克塞里艾克西斯公司 | 制备氟-18标记的卡博替尼及其类似物的方法 |
| UA121482C2 (uk) | 2014-08-05 | 2020-06-10 | Екселіксіс, Інк. | Комбінація інгібітору протеасом, яким є бортезоміб, та інгібітору c-met для лікування множинної мієломи |
| CN105503717A (zh) * | 2014-09-24 | 2016-04-20 | 江苏奥赛康药业股份有限公司 | 一种苹果酸卡博替尼化合物及其药物组合物 |
| CN104961680B (zh) * | 2014-11-07 | 2017-09-12 | 苏州晶云药物科技有限公司 | N‑(4‑{[6,7‑双(甲基氧基)喹啉‑4‑基]氧基}苯基)‑n’‑(4‑氟苯基)环丙烷‑1,1‑二甲酰胺的盐酸盐及其多晶型 |
| CN104961681B (zh) * | 2014-11-13 | 2017-06-13 | 苏州晶云药物科技有限公司 | 卡博替尼的粘酸盐及其晶型 |
| US10206916B2 (en) | 2015-03-25 | 2019-02-19 | Sandoz Ag | Cabozantinib salts and their use as anti-cancer agents |
| ES2716633T3 (es) | 2015-03-25 | 2019-06-13 | Sandoz Ag | Formas cristalinas de fosfato de cabozantinib e hidrocloruro de cabozantinib |
| EP3337786B1 (en) | 2015-08-19 | 2019-12-11 | Sandoz AG | Asymmetric bisamidation of malonic ester derivatives |
| CN109069499A (zh) * | 2016-04-15 | 2018-12-21 | 埃克塞里艾克西斯公司 | 使用 n-(4-(6,7-二甲氧基喹啉-4-基氧基)苯基)-n’-(4-氟苯基)环丙烷-1,1-二甲酰胺,(2s)-羟基丁二酸盐治疗肾细胞癌的方法 |
| US10980792B2 (en) | 2016-09-12 | 2021-04-20 | Zhuhai Beihai Biotech Co., Ltd. | Formulations of Cabozantinib |
| KR101829706B1 (ko) * | 2016-09-21 | 2018-02-19 | 씨제이헬스케어 주식회사 | 벤즈이미다졸 유도체의 산부가염 |
| MX389966B (es) * | 2017-01-20 | 2025-03-20 | Exelixis Inc | Combinaciones de cabozantinib y atezolizumab para tratar cancer. |
| CN108341773A (zh) * | 2017-01-21 | 2018-07-31 | 南京华威医药科技开发有限公司 | 卡博替尼苹果酸盐的晶型ii |
| ES2909390T3 (es) | 2017-05-26 | 2022-05-06 | Exelixis Inc | Formas sólidas cristalinas de sales de N-{4-[(6,7-dimetoxiquinolin-4-il)oxi]fenil}-N'-(4-fluorofenil)ciclopropano-1,1-dicarboxamida, procedimientos de fabricación y procedimientos de uso |
| JP7249962B2 (ja) | 2017-06-09 | 2023-03-31 | エグゼリクシス, インコーポレイテッド | がんを治療するための液体剤形 |
| CN109836382B (zh) * | 2017-11-29 | 2021-11-05 | 江苏豪森药业集团有限公司 | 苹果酸卡博替尼及其中间体的制备方法 |
| DK3743070T3 (da) | 2018-01-26 | 2025-09-29 | Exelixis Inc | Forbindelser til behandling af kinaseafhængige lidelser |
| WO2019234761A1 (en) * | 2018-06-05 | 2019-12-12 | Natco Pharma Limited | An improved process for the preparation of cabozantinib and its pharmaceutically acceptable salts thereof |
| TWI770624B (zh) | 2018-06-15 | 2022-07-11 | 漢達生技醫藥股份有限公司 | 尼洛替尼十二烷基硫酸鹽在製備用於治療慢性骨髓性白血病之劑型的用途 |
| WO2020075196A1 (en) | 2018-10-11 | 2020-04-16 | Cipla Limited | Polymorphs of n-(4-(6,7-dimethoxyquinolin-4-yloxy) phenyl)-n'-(4-fluorophenyl)cyclopropane-1, 1-dicarboxamide, (s)- malate, methods of production and pharmaceutical uses thereof |
| AU2019395419B2 (en) | 2018-12-13 | 2025-06-26 | Exelixis, Inc. | Crystalline forms and salt forms of a kinase inhibitor |
| IL289941B2 (en) | 2019-07-19 | 2025-07-01 | Biosynth Ag | Method of making nicotinamide ribofuranoside salts, nicotinamide ribofuranoside salts as such, and uses thereof |
| CN112979544B (zh) * | 2019-12-17 | 2024-06-28 | 江苏先声药业有限公司 | 一种卡博替尼或其盐的制备方法 |
| WO2021209940A1 (en) * | 2020-04-14 | 2021-10-21 | Msn Laboratories Private Limited, R&D Center | Pharmaceutical composition containing n-(4-(6,7-dimethoxyquinolin-4-yloxy)phenyl)-n'-(4-fluorophenyl)cyclopropane-1,1-dicarboxamide, (2s)-hydroxybutanedioate |
| CA3176042A1 (en) * | 2020-04-30 | 2021-11-04 | Exelixis, Inc. | Processes for the preparation of a kinase inhibitor |
| CN114456109B (zh) * | 2020-11-09 | 2025-11-28 | 北京轩升制药有限公司 | 一种高纯度卡博替尼苹果酸盐的制备方法及其应用 |
| IT202000027678A1 (it) | 2020-11-18 | 2022-05-18 | Indena Spa | Dispersioni solide amorfe di cabozantinib-(s)-malato e processi per la loro preparazione |
| WO2022115464A1 (en) | 2020-11-25 | 2022-06-02 | Nanocopoeia, Llc | Amorphous cabozantinib particles and uses thereof |
| WO2022177983A1 (en) | 2021-02-19 | 2022-08-25 | Slayback Pharma Llc | Pharmaceutical compositions of cabozantinib |
| US20240190825A1 (en) * | 2021-03-24 | 2024-06-13 | Biocon Limited | Process for preparation of cabozantinib |
| CN115215797B (zh) * | 2021-04-15 | 2024-04-12 | 成都苑东生物制药股份有限公司 | 一种苹果酸卡博替尼新晶型及其制备方法 |
| CN118339144A (zh) * | 2021-12-03 | 2024-07-12 | 湖南湘源美东医药科技有限公司 | 卡博替尼共晶及制备方法以及作为药物或在药物制剂中的应用 |
| WO2023165948A1 (en) | 2022-03-01 | 2023-09-07 | Synthon B.V. | Cabozantinib salt with l-(+)-tartaric acid and solid forms thereof |
| WO2023222946A1 (en) | 2022-05-18 | 2023-11-23 | Fermion Oy | Process for the preparation of cabozantinib |
| AU2024215796A1 (en) | 2023-01-31 | 2025-05-15 | Handa Oncology, Llc | Improved cabozantinib compositions and methods of use |
| US11814356B1 (en) | 2023-03-29 | 2023-11-14 | Apotex Inc. | Salt of cabozantinib |
| WO2025079089A1 (en) * | 2023-10-09 | 2025-04-17 | Msn Laboratories Private Limited, R&D Center | Pharmaceutical composition of cabozantinib |
| WO2025090032A1 (en) * | 2023-10-24 | 2025-05-01 | Deva Holding | Novel polymorph of cabozantinib l-tartrate (form a) and method of preparation |
| CN120058603A (zh) * | 2025-03-04 | 2025-05-30 | 鲁南制药集团股份有限公司 | 一种卡博替尼新晶型 |
Family Cites Families (208)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB2160201B (en) | 1984-06-14 | 1988-05-11 | Wyeth John & Brother Ltd | Quinazoline and cinnoline derivatives |
| US5123951A (en) | 1986-03-31 | 1992-06-23 | Rhone-Poulenc Nederland B.V. | Synergistic plant growth regulator compositions |
| JPS646261A (en) | 1987-03-31 | 1989-01-10 | Nisshin Flour Milling Co | 4-thioquinazoline derivative, its production and antiulcer agent containing said derivative as active component |
| US5143854A (en) | 1989-06-07 | 1992-09-01 | Affymax Technologies N.V. | Large scale photolithographic solid phase synthesis of polypeptides and receptor binding screening thereof |
| US5034393A (en) | 1989-07-27 | 1991-07-23 | Dowelanco | Fungicidal use of pyridopyrimidine, pteridine, pyrimidopyrimidine, pyrimidopyridazine, and pyrimido-1,2,4-triazine derivatives |
| US5238951A (en) | 1991-02-01 | 1993-08-24 | E. R. Squibb & Sons, Inc. | Heterocyclic amido prostaglandin analogs |
| DE4114733A1 (de) | 1991-05-06 | 1992-11-12 | Huels Chemische Werke Ag | Verfahren zur herstellung von substituierten malonesteraniliden und malonsaeure-monoaniliden |
| US5710158A (en) | 1991-05-10 | 1998-01-20 | Rhone-Poulenc Rorer Pharmaceuticals Inc. | Aryl and heteroaryl quinazoline compounds which inhibit EGF and/or PDGF receptor tyrosine kinase |
| US5480883A (en) | 1991-05-10 | 1996-01-02 | Rhone-Poulenc Rorer Pharmaceuticals Inc. | Bis mono- and bicyclic aryl and heteroaryl compounds which inhibit EGF and/or PDGF receptor tyrosine kinase |
| US6057344A (en) | 1991-11-26 | 2000-05-02 | Sepracor, Inc. | Methods for treating hypertension, and angina using optically pure (-) amlodipine |
| US6498144B1 (en) | 1993-10-18 | 2002-12-24 | North Shore - Long Island Jewish Research Institute | Use of scatter factor to enhance angiogenesis |
| IL112249A (en) | 1994-01-25 | 2001-11-25 | Warner Lambert Co | Pharmaceutical compositions containing di and tricyclic pyrimidine derivatives for inhibiting tyrosine kinases of the epidermal growth factor receptor family and some new such compounds |
| GB9510757D0 (en) | 1994-09-19 | 1995-07-19 | Wellcome Found | Therapeuticaly active compounds |
| TW321649B (ko) | 1994-11-12 | 1997-12-01 | Zeneca Ltd | |
| GB9508538D0 (en) | 1995-04-27 | 1995-06-14 | Zeneca Ltd | Quinazoline derivatives |
| US5747498A (en) | 1996-05-28 | 1998-05-05 | Pfizer Inc. | Alkynyl and azido-substituted 4-anilinoquinazolines |
| WO1996040648A1 (en) | 1995-06-07 | 1996-12-19 | Sugen, Inc. | Quinazolines and pharmaceutical compositions |
| US5650415A (en) | 1995-06-07 | 1997-07-22 | Sugen, Inc. | Quinoline compounds |
| DE69531558T2 (de) | 1995-06-07 | 2004-03-18 | Pfizer Inc. | Heterocyclische kondensierte pyrimidin-derivate |
| GB9514265D0 (en) | 1995-07-13 | 1995-09-13 | Wellcome Found | Hetrocyclic compounds |
| EP0860433B1 (en) | 1995-11-07 | 2002-07-03 | Kirin Beer Kabushiki Kaisha | Quinoline derivatives and quinazoline derivatives inhibiting autophosphorylation of growth factor receptor originating in platelet and pharmaceutical compositions containing the same |
| GB9523675D0 (en) | 1995-11-20 | 1996-01-24 | Celltech Therapeutics Ltd | Chemical compounds |
| GB9624482D0 (en) | 1995-12-18 | 1997-01-15 | Zeneca Phaema S A | Chemical compounds |
| GB9526546D0 (en) | 1995-12-23 | 1996-02-28 | Pfizer Ltd | Compounds useful in therapy |
| DK0880508T3 (da) | 1996-02-13 | 2003-06-30 | Astrazeneca Ab | Quinazolinderivater som VEGF-inhibitorer |
| GB9603095D0 (en) | 1996-02-14 | 1996-04-10 | Zeneca Ltd | Quinazoline derivatives |
| CN1116286C (zh) | 1996-03-05 | 2003-07-30 | 曾尼卡有限公司 | 4-苯胺基喹唑啉衍生物 |
| US6514971B1 (en) | 1996-03-15 | 2003-02-04 | Zeneca Limited | Cinnoline derivatives and use as medicine |
| US6107300A (en) | 1996-03-27 | 2000-08-22 | Dupont Pharmaceuticals | Arylamino fused pyrimidines |
| SK284073B6 (sk) | 1996-04-12 | 2004-09-08 | Warner-Lambert Company | Polycyklické zlúčeniny, ich použitie a farmaceutické kompozície na ich báze |
| CZ370298A3 (cs) | 1996-05-20 | 1999-05-12 | G. D. Searle Co. | Draselné, sodné a trisoxaprozinové soli farmaceutických formulací |
| HRP970371A2 (en) | 1996-07-13 | 1998-08-31 | Kathryn Jane Smith | Heterocyclic compounds |
| ATE227283T1 (de) | 1996-07-13 | 2002-11-15 | Glaxo Group Ltd | Kondensierte heterozyklische verbindungen als protein kinase inhibitoren |
| US5962407A (en) | 1996-07-26 | 1999-10-05 | Kelly; Michael T. | Loloatin derivatives and analogs |
| ATE300521T1 (de) | 1996-09-25 | 2005-08-15 | Astrazeneca Ab | Chinolin-derivate die den effekt von wachstumsfaktoren wie vegf vezögern |
| GB9718972D0 (en) | 1996-09-25 | 1997-11-12 | Zeneca Ltd | Chemical compounds |
| JP4073961B2 (ja) | 1996-10-01 | 2008-04-09 | 協和醗酵工業株式会社 | 含窒素複素環化合物 |
| GB9700504D0 (en) | 1997-01-11 | 1997-02-26 | Pfizer Ltd | Pharmaceutical compounds |
| GB9705361D0 (en) | 1997-03-14 | 1997-04-30 | Celltech Therapeutics Ltd | Chemical compounds |
| UA73073C2 (uk) | 1997-04-03 | 2005-06-15 | Уайт Холдінгз Корпорейшн | Заміщені 3-ціанохіноліни, спосіб їх одержання та фармацевтична композиція |
| EP0990647B1 (en) | 1997-04-18 | 2003-07-02 | Kirin Beer Kabushiki Kaisha | Process for producing quinolone derivatives |
| NZ335822A (en) | 1997-04-22 | 2001-05-25 | Neurocrine Biosciences Inc | Preparation and use of 2-methyl-4-dipropylamino-8(2',4'-dichlorophenyl)-quinoline and 2-methyl-4-(N-propyl-N-cyclopropanemethyl)amino-8(2',4'-dichlorophenyl)-quinoline for treating corticotropin-releasing factor related diseases |
| GB9708917D0 (en) | 1997-05-01 | 1997-06-25 | Pfizer Ltd | Compounds useful in therapy |
| AR012634A1 (es) | 1997-05-02 | 2000-11-08 | Sugen Inc | Compuesto basado en quinazolina, composicion famaceutica que lo comprende, metodo para sintetizarlo, su uso, metodos de modulacion de la funcion deserina/treonina proteinaquinasa con dicho compuesto y metodo in vitro para identificar compuestos que modulan dicha funcion |
| ZA986732B (en) | 1997-07-29 | 1999-02-02 | Warner Lambert Co | Irreversible inhibitiors of tyrosine kinases |
| ATE368665T1 (de) | 1997-08-22 | 2007-08-15 | Astrazeneca Ab | Oxindolylchinazolinderivate als angiogenesehemmer |
| RS49779B (sr) | 1998-01-12 | 2008-06-05 | Glaxo Group Limited, | Biciklična heteroaromatična jedinjenja kao inhibitori protein tirozin kinaze |
| WO1999061428A1 (en) | 1998-05-28 | 1999-12-02 | Parker Hughes Institute | Quinazolines for treating brain tumor |
| PT1107964E (pt) | 1998-08-11 | 2010-06-11 | Novartis Ag | Derivados de isoquinolina com actividade inibidora da angiogénese |
| JP2002523403A (ja) | 1998-08-21 | 2002-07-30 | パーカー ヒューズ インスティテュート | キナゾリン誘導体 |
| US6184226B1 (en) | 1998-08-28 | 2001-02-06 | Scios Inc. | Quinazoline derivatives as inhibitors of P-38 α |
| US6740326B1 (en) | 1998-09-10 | 2004-05-25 | Bioequal Ag | Topical nail care compositions |
| US6288082B1 (en) | 1998-09-29 | 2001-09-11 | American Cyanamid Company | Substituted 3-cyanoquinolines |
| CA2344169C (en) | 1998-09-29 | 2011-07-19 | American Cyanamid Company | Substituted 3-cyanoquinolines as protein tyrosine kinases inhibitors |
| JP2002526538A (ja) | 1998-10-01 | 2002-08-20 | アストラゼネカ アクチボラグ | 化学化合物 |
| FR2784030B1 (fr) | 1998-10-02 | 2002-12-20 | Inst Nat Sante Rech Med | Utilisation de bloqueurs des canaux calciques et/ou cgmp-dependants pour le traitement de pathologies de la retine |
| DK1119567T3 (da) | 1998-10-08 | 2005-07-25 | Astrazeneca Ab | Quinazolinderivater |
| JP3687900B2 (ja) | 1998-11-19 | 2005-08-24 | ワーナー−ランバート・カンパニー、リミテッド、ライアビリティ、カンパニー | チロシンキナーゼの不可逆的阻害剤であるn−[4−(3−クロロ−4−フルオロフェニルアミノ)−7−(3−モルホリン−4−イルプロポキシ)キナゾリン−6−イル]アクリルアミド |
| WO2000043366A1 (en) | 1999-01-22 | 2000-07-27 | Kirin Beer Kabushiki Kaisha | Quinoline derivatives and quinazoline derivatives |
| KR20080015482A (ko) | 1999-02-10 | 2008-02-19 | 아스트라제네카 아베 | 혈관형성 억제제로서의 퀴나졸린 유도체 |
| GB9904103D0 (en) | 1999-02-24 | 1999-04-14 | Zeneca Ltd | Quinoline derivatives |
| US6080747A (en) | 1999-03-05 | 2000-06-27 | Hughes Institute | JAK-3 inhibitors for treating allergic disorders |
| DE19911509A1 (de) | 1999-03-15 | 2000-09-21 | Boehringer Ingelheim Pharma | Bicyclische Heterocyclen, diese Verbindungen enthaltende Arzneimittel, deren Verwendung und Verfahren zu ihrer Herstellung |
| WO2000056338A1 (en) | 1999-03-19 | 2000-09-28 | Parker Hughes Institute | Quinazoline formulations and therapeutic use thereof |
| US6258820B1 (en) | 1999-03-19 | 2001-07-10 | Parker Hughes Institute | Synthesis and anti-tumor activity of 6,7-dialkoxy-4-phenylamino-quinazolines |
| YU13200A (sh) | 1999-03-31 | 2002-10-18 | Pfizer Products Inc. | Postupci i intermedijeri za dobijanje anti-kancernih jedinjenja |
| EP1169041A2 (en) | 1999-03-31 | 2002-01-09 | The Procter & Gamble Company | Use of carbamic acid derivatives for the treatment of viral infections |
| GB9910580D0 (en) | 1999-05-08 | 1999-07-07 | Zeneca Ltd | Chemical compounds |
| GB9910577D0 (en) | 1999-05-08 | 1999-07-07 | Zeneca Ltd | Chemical compounds |
| US6126917A (en) | 1999-06-01 | 2000-10-03 | Hadasit Medical Research Services And Development Ltd. | Epidermal growth factor receptor binding compounds for positron emission tomography |
| CA2375259C (en) | 1999-06-21 | 2009-04-28 | Boehringer Ingelheim Pharma Kg | Bicyclic heterocycles, pharmaceutical compositions containing these compounds, their use and processes for preparing them |
| GB9922171D0 (en) | 1999-09-21 | 1999-11-17 | Zeneca Ltd | Chemical compounds |
| JP4870304B2 (ja) | 1999-09-21 | 2012-02-08 | アストラゼネカ アクチボラグ | キナゾリン誘導体およびそれらの医薬品としての使用 |
| US6759410B1 (en) | 1999-11-23 | 2004-07-06 | Smithline Beecham Corporation | 3,4-dihydro-(1H)-quinazolin-2-ones and their use as CSBP/p38 kinase inhibitors |
| CA2392554A1 (en) | 1999-11-30 | 2001-06-28 | Parker Hughes Institute | Inhibitors of thrombin induced platelet aggregation |
| US20020002169A1 (en) | 1999-12-08 | 2002-01-03 | Griffin John H. | Protein kinase inhibitors |
| US7135466B2 (en) | 1999-12-24 | 2006-11-14 | Kirin Beer Kabushiki Kaisha | Quinoline and quinazoline derivatives and drugs containing the same |
| US6525046B1 (en) | 2000-01-18 | 2003-02-25 | Boehringer Ingelheim Pharmaceuticals, Inc. | Aromatic heterocyclic compounds as antiinflammatory agents |
| US6977259B2 (en) | 2000-01-28 | 2005-12-20 | Astrazeneca Ab | Quinoline derivatives and their use as aurora 2 kinase inhibitors |
| US6664390B2 (en) | 2000-02-02 | 2003-12-16 | Warner-Lambert Company Llc | Method for the simplified production of (3-chloro-4-fluorophenyl)-[7-(3-morpholin-4-yl-propoxy)-6-nitro-quinazoline-4-yl]-amine or (3-chloro-4-fluorophenyl)-[7-(3-morpholin-4-yl-propoxy)-6-amino-quinazoline-4-yl]-amine |
| KR20020084116A (ko) | 2000-02-07 | 2002-11-04 | 애보트 게엠베하 운트 콤파니 카게 | 2-벤조티아졸릴 우레아 유도체 및 이의 단백질 키나제억제제로서의 용도 |
| CZ303705B6 (cs) | 2000-02-15 | 2013-03-27 | Sugen, Inc. | Pyrrolem substituovaná 2-indolinonová sloucenina pro pouzití jako inhibitor proteinkináz a farmaceutická kompozice s jejím obsahem |
| HUP0300547A2 (hu) | 2000-03-13 | 2003-07-28 | American Cyanamid Co. | Ciano-kinolin vegyületek alkalmazása vastagbél polipok kezelésére és gátlására alkalmas gyógyszerkészítmények előállítására |
| US6521618B2 (en) | 2000-03-28 | 2003-02-18 | Wyeth | 3-cyanoquinolines, 3-cyano-1,6-naphthyridines, and 3-cyano-1,7-naphthyridines as protein kinase inhibitors |
| US6608048B2 (en) | 2000-03-28 | 2003-08-19 | Wyeth Holdings | Tricyclic protein kinase inhibitors |
| US6627634B2 (en) | 2000-04-08 | 2003-09-30 | Boehringer Ingelheim Pharma Kg | Bicyclic heterocycles, pharmaceutical compositions containing them, their use, and processes for preparing them |
| UA73993C2 (uk) | 2000-06-06 | 2005-10-17 | Астразенека Аб | Хіназолінові похідні для лікування пухлин та фармацевтична композиція |
| PT1292591E (pt) | 2000-06-22 | 2005-06-30 | Pfizer Prod Inc | Derivados biciclicos substituidos para o tratamento de crescimento de celula anormal |
| EP1174118A1 (de) | 2000-06-28 | 2002-01-23 | Cognis France S.A. | Verwendung von Inulinen und Inulinderivaten |
| PL360439A1 (en) | 2000-06-28 | 2004-09-06 | Astrazeneca Ab | Substituted quinazoline derivatives and their use as inhibitors |
| FR2811658B1 (fr) | 2000-07-17 | 2004-07-02 | Cfpi Nufarm | Reacteur biologique a lit fixe immerge et procede de traitement d'effluents liquides |
| JP2002030083A (ja) * | 2000-07-18 | 2002-01-29 | Kirin Brewery Co Ltd | N−(2−クロロ−4−{[6−メトキシ−7−(3−ピリジルメトキシ)−4−キノリル]オキシ}フェニル)−n’−プロピルウレアの二塩酸塩 |
| US7427689B2 (en) | 2000-07-28 | 2008-09-23 | Georgetown University | ErbB-2 selective small molecule kinase inhibitors |
| KR100821446B1 (ko) | 2000-08-21 | 2008-04-10 | 아스트라제네카 아베 | 퀴나졸린 유도체 |
| FR2813307B1 (fr) | 2000-08-23 | 2002-11-08 | Sanofi Synthelabo | Aminoalkenylbenzoyl-benzofurannes ou benzothiophenes, leur procede de preparation et les compositions les contenant |
| DE10042058A1 (de) | 2000-08-26 | 2002-03-07 | Boehringer Ingelheim Pharma | Bicyclische Heterocyclen, diese Verbindungen enthaltende Arzneimittel, deren Verwendung und Verfahren zu ihrer Herstellung |
| US6617329B2 (en) | 2000-08-26 | 2003-09-09 | Boehringer Ingelheim Pharma Kg | Aminoquinazolines and their use as medicaments |
| US6403580B1 (en) | 2000-08-26 | 2002-06-11 | Boehringer Ingelheim Pharma Kg | Quinazolines, pharmaceutical compositions containing these compounds, their use and processes for preparing them |
| US6656946B2 (en) | 2000-08-26 | 2003-12-02 | Boehringer Ingelheim Pharma Kg | Aminoquinazolines which inhibit signal transduction mediated by tyrosine kinases |
| US6740651B2 (en) | 2000-08-26 | 2004-05-25 | Boehringer Ingelheim Pharma Kg | Aminoquinazolines which inhibit signal transduction mediated by tyrosine kinases |
| US6653305B2 (en) | 2000-08-26 | 2003-11-25 | Boehringer Ingelheim Pharma Kg | Bicyclic heterocycles, pharmaceutical compositions containing them, their use, and processes for preparing them |
| JP2004511479A (ja) | 2000-10-13 | 2004-04-15 | アストラゼネカ アクチボラグ | キナゾリン誘導体 |
| EP1326860A1 (en) | 2000-10-13 | 2003-07-16 | AstraZeneca AB | Quinazoline derivatives |
| DE60137273D1 (de) | 2000-10-20 | 2009-02-12 | Eisai R&D Man Co Ltd | Verfahren zur Herstellung von 4-Phenoxy chinolin Derivaten |
| US20040063733A1 (en) | 2000-10-25 | 2004-04-01 | Lambert Christine Marie Paul | Quinazoline derivatives |
| US7067532B2 (en) | 2000-11-02 | 2006-06-27 | Astrazeneca | Substituted quinolines as antitumor agents |
| EP1340748B1 (en) | 2000-11-02 | 2008-01-16 | Nippon Shinyaku Co., Ltd. | Quinazoline derivatives and drugs |
| EP1337513A1 (en) | 2000-11-02 | 2003-08-27 | AstraZeneca AB | 4-substituted quinolines as antitumor agents |
| US7019012B2 (en) | 2000-12-20 | 2006-03-28 | Boehringer Ingelheim International Pharma Gmbh & Co. Kg | Quinazoline derivatives and pharmaceutical compositions containing them |
| US6900220B2 (en) | 2001-01-02 | 2005-05-31 | Syntex (U.S.A.) Llc | Quinazolone derivatives as alpha 1A/B adrenergic receptor antagonists |
| GB0103046D0 (en) | 2001-02-07 | 2001-03-21 | Novartis Ag | Organic Compounds |
| JP2002265365A (ja) | 2001-03-08 | 2002-09-18 | Koyo Chemical Kk | 好中球機能抑制剤 |
| DE60229046D1 (de) | 2001-04-19 | 2008-11-06 | Astrazeneca Ab | Chinazolin derivate |
| WO2002088110A1 (en) | 2001-04-27 | 2002-11-07 | Kirin Beer Kabushiki Kaisha | Quinoline derivative having azolyl group and quinazoline derivative |
| SE0101675D0 (sv) | 2001-05-11 | 2001-05-11 | Astrazeneca Ab | Novel composition |
| WO2002092577A1 (en) | 2001-05-14 | 2002-11-21 | Astrazeneca Ab | Quinazoline derivatives |
| WO2002092578A1 (en) | 2001-05-14 | 2002-11-21 | Astrazeneca Ab | Quinazoline derivatives |
| WO2002092579A1 (en) | 2001-05-14 | 2002-11-21 | Astrazeneca Ab | 4-anilinoquinazoline derivatives |
| US6734303B2 (en) | 2001-05-18 | 2004-05-11 | Pfizer Inc. | Process for the production of quinazolines |
| DE10125432A1 (de) | 2001-05-25 | 2002-11-28 | Bayer Ag | Substituierte Benzoylketone |
| US7132427B2 (en) | 2001-06-21 | 2006-11-07 | Ariad Pharmaceuticals, Inc. | Quinazolines and uses thereof |
| US7425564B2 (en) | 2001-06-22 | 2008-09-16 | Kirin Beer Kabushiki Kaisha | Quinoline derivative and quinazoline derivative inhibiting self-phosphorylation of hepatocytus prolifertor receptor and medicinal composition containing the same |
| KR100397792B1 (ko) | 2001-06-28 | 2003-09-13 | 한국과학기술연구원 | 4-(페닐아미노)-[1,4]디옥사노[2,3-g]퀴나졸린 유도체 및그의 제조방법 |
| GB0118752D0 (en) | 2001-08-01 | 2001-09-26 | Pfizer Ltd | Process for the production of quinazolines |
| US7229774B2 (en) | 2001-08-02 | 2007-06-12 | Regents Of The University Of Michigan | Expression profile of prostate cancer |
| EP2332934B1 (en) | 2001-08-15 | 2017-03-01 | Pharmacia & Upjohn Company LLC | Processes for the preparation of crystals including a malic acid salt of n-[2-(diethylamino)ethyl]-5-[(5-fluoro-1,2-dihydro-2-oxo-3h-indole-3-ylidene)methyl]-2,4-dimethyl-1h-pyrrole-3-carboxamide. |
| US20030066060A1 (en) | 2001-09-28 | 2003-04-03 | Ford Richard L. | Cross profile guided optimization of program execution |
| EP1447405A4 (en) | 2001-10-17 | 2005-01-12 | Kirin Brewery | CHINOLIN OR CHINAZOLINE DERIVATIVES THAT PREVENT THE AUTOPHOSPHORYLATION OF RECEPTORS FOR THE FIBROBLAST GROWTH FACTOR |
| US7169788B2 (en) | 2001-10-30 | 2007-01-30 | Merck & Co., Inc. | Tyrosine kinase inhibitors |
| GB0126433D0 (en) | 2001-11-03 | 2002-01-02 | Astrazeneca Ab | Compounds |
| US7319107B2 (en) | 2001-11-08 | 2008-01-15 | Johnson & Johnson Consumer Companies, Inc. | 1,2,4-thiadiazolium derivatives as melanocortin receptor modulators |
| GB0128108D0 (en) | 2001-11-23 | 2002-01-16 | Astrazeneca Ab | Therapeutic use |
| MXPA04004969A (es) | 2001-11-27 | 2004-08-11 | Wyeth Corp | 3-cianoquinolinas como inhibidores del receptor del factor de crecimiento epidermico y her2 cinasas. |
| GB0129099D0 (en) | 2001-12-05 | 2002-01-23 | Astrazeneca Ab | Chemical compounds |
| AU2002347336A1 (en) | 2001-12-05 | 2003-06-17 | Astrazeneca Ab | Quinoline derivatives |
| WO2003047584A1 (en) | 2001-12-05 | 2003-06-12 | Astrazeneca Ab | Quinoline derivatives |
| AU2002360582B2 (en) | 2001-12-10 | 2009-06-11 | Aryx Therapeutics | Novel compounds for the treatment of cardiac arrhythmia, synthesis, and methods of use |
| AU2002339687A1 (en) | 2001-12-12 | 2003-06-23 | Pfizer Products Inc. | Quinazoline derivatives for the treatement of abnormal cell growth |
| GEP20063872B (en) | 2001-12-12 | 2006-07-10 | Pfizer Prod Inc | Salt forms of e-2-methoxy-n-(3-{4-[3-methyl-4-(6-methyl-pyridin-3-yloxy)-phenylamino]-quina-zolin-6-yl}-allyl)-acetamide and method of production |
| TW200301123A (en) | 2001-12-21 | 2003-07-01 | Astrazeneca Uk Ltd | New use |
| WO2003055866A1 (en) | 2001-12-21 | 2003-07-10 | Bayer Pharmaceuticals Corporation | Quinazoline and quinoline derivative compounds as inhibitors of prolylpeptidase, inducers of apoptosis and cancer treatment agents |
| JP4498741B2 (ja) | 2001-12-24 | 2010-07-07 | アストラゼネカ アクチボラグ | オーロラキナーゼ阻害剤としての置換キナゾリン誘導体 |
| TW200406410A (en) | 2002-01-29 | 2004-05-01 | Glaxo Group Ltd | Compounds |
| JP4508650B2 (ja) | 2002-01-29 | 2010-07-21 | グラクソ グループ リミテッド | アミノピペリジン化合物、当該化合物の製法および当該化合物を含有する医薬組成物 |
| RU2365588C2 (ru) | 2002-02-01 | 2009-08-27 | Астразенека Аб | Хиназолиновые соединения |
| DE10204462A1 (de) | 2002-02-05 | 2003-08-07 | Boehringer Ingelheim Pharma | Verwendung von Tyrosinkinase-Inhibitoren zur Behandlung inflammatorischer Prozesse |
| TW200813014A (en) | 2002-03-28 | 2008-03-16 | Astrazeneca Ab | Quinazoline derivatives |
| DE10217689A1 (de) | 2002-04-19 | 2003-11-13 | Boehringer Ingelheim Pharma | Bicyclische Heterocyclen, diese Verbindungen enthaltende Arzneimittel, ihre Verwendung und Verfahren zu ihrer Herstellung |
| US6693198B2 (en) | 2002-04-22 | 2004-02-17 | Xanthus Life Sciences, Inc. | Amonafide salts |
| AU2003235838A1 (en) | 2002-05-01 | 2003-11-17 | Kirin Beer Kabushiki Kaisha | Quinoline derivatives and quinazoline derivatives inhibiting autophosphorylation of macrophage colony stimulating factor receptor |
| US7323479B2 (en) | 2002-05-17 | 2008-01-29 | Celgene Corporation | Methods for treatment and management of brain cancer using 1-oxo-2-(2,6-dioxopiperidin-3-yl)-4-methylisoindoline |
| US7576074B2 (en) | 2002-07-15 | 2009-08-18 | Rice Kenneth D | Receptor-type kinase modulators and methods of use |
| GB0219746D0 (en) | 2002-08-23 | 2002-10-02 | Inst Of Ex Botany Ascr | Azapurine derivatives |
| EP1548008A4 (en) | 2002-08-23 | 2008-08-06 | Kirin Pharma Kk | COMPOUND WITH TGF-BETA-HEMMENDER EFFECT AND THIS MEDICAL COMPOSITION CONTAINING |
| US7419984B2 (en) | 2002-10-17 | 2008-09-02 | Cell Therapeutics, Inc. | Pyrimidines and uses thereof |
| DE60316590T2 (de) | 2002-10-21 | 2008-06-19 | Kirin Beer K.K. | N-(2-CHLOR-4-((6,7-DIMETHOXY-4-CHINOLYL)OXYÜPHENYLü-N'-(5-METHYL-3-ISOXAZOLYL)HARNSTOFFSALZIN KRISTALLINER FORM |
| AU2003280599A1 (en) | 2002-10-29 | 2004-05-25 | Kirin Beer Kabushiki Kaisha | QUINOLINE DERIVATIVES AND QUINAZOLINE DERIVATIVES INHIBITING AUTOPHOSPHORYLATION OF Flt3 AND MEDICINAL COMPOSITIONS CONTAINING THE SAME |
| CA2503371C (en) | 2002-11-04 | 2011-01-11 | Astrazeneca Ab | Quinazoline derivatives as src tyrosine kinase inhibitors |
| EP1569925A1 (en) | 2002-12-13 | 2005-09-07 | Neurogen Corporation | 2-substituted quinazolin-4-ylamine analogues as capsaicin receptor modulators |
| PL377686A1 (pl) | 2002-12-18 | 2006-02-06 | Pfizer Products Inc. | Pochodne 4-anilinochinazoliny do leczenia anormalnego wzrostu komórek |
| EP1575591A4 (en) | 2002-12-23 | 2007-09-12 | Ariad Pharma Inc | HETEROCYCLES AND ITS USE |
| WO2004060373A1 (ja) | 2002-12-27 | 2004-07-22 | Santen Pharmaceutical Co., Ltd. | 滲出型加齢黄斑変性治療剤 |
| US8176532B1 (en) | 2003-03-17 | 2012-05-08 | Sprint Communications Company L.P. | Secure access point for scada devices |
| EP1608373A4 (en) | 2003-03-19 | 2010-09-29 | Exelixis Inc | TIE-2 MODULATORS AND USE METHOD |
| KR100559180B1 (ko) | 2003-05-20 | 2006-03-14 | 김민서 | 조건부 거래에 따른 전자결제 방법 및 전자결제 서버 |
| WO2005003140A1 (en) | 2003-07-02 | 2005-01-13 | Pharmacia & Upjohn Company Llc | 4-oxo-4,7-dihydrothieno[2,3-b]pyridine-5-carboxamides as antiviral agents |
| WO2005005389A2 (en) | 2003-07-07 | 2005-01-20 | Merck Patent Gmbh | Malonamide derivatives |
| JP4299090B2 (ja) * | 2003-09-24 | 2009-07-22 | 株式会社東海理化電機製作所 | 車両用空調装置の操作装置 |
| PL2210607T3 (pl) * | 2003-09-26 | 2012-01-31 | Exelixis Inc | N-[3-fluoro-4-({6-(metyloksy)-7-[(3-morfolin-4-ylopropylo)oksy]chinolin-4-ylo} oxy)fenylo]-N'-(4-fluorofenylo)cyklopropano-1,1-dikarboksamid do leczenia raka |
| AU2004288709B2 (en) | 2003-11-07 | 2011-01-06 | Novartis Vaccines And Diagnostics, Inc. | Methods for synthesizing quinolinone compounds |
| EP2567955A3 (en) | 2003-12-25 | 2013-03-27 | Eisai R&D Management Co., Ltd. | Crystal of salt of 4-(3-chloro-4-(cyclopropylaminocarbonyl)aminophenoxy)-7-methoxy-6-quinolinecarboxamide or of solvate thereof and processes for producing these |
| EP1711495A2 (en) | 2004-01-23 | 2006-10-18 | Amgen Inc. | Quinoline, quinazoline, pyridine and pyrimidine counds and their use in the treatment of inflammation, angiogenesis and cancer |
| AR047530A1 (es) | 2004-02-04 | 2006-01-25 | Novartis Ag | Formas de sal de 4-(4-metilpiperazin-1-ilmetil)-n-(4-metil-3-(4-piridin-3-il)pirimidin-2-ilamino)fenil)-benzamida |
| US20060035940A1 (en) | 2004-03-16 | 2006-02-16 | Sepracor Inc. | (S)-Amlodipine malate |
| US7173031B2 (en) | 2004-06-28 | 2007-02-06 | Bristol-Myers Squibb Company | Pyrrolotriazine kinase inhibitors |
| JP5368701B2 (ja) | 2004-07-02 | 2013-12-18 | エクセリクシス、インコーポレイテッド | c−Metモジュレーター及び使用方法 |
| EP1853968A2 (en) * | 2005-03-04 | 2007-11-14 | Colorlink, Inc. | Four panel projection system |
| AU2006231646A1 (en) | 2005-04-06 | 2006-10-12 | Exelixis, Inc. | C-Met modulators and methods of use |
| WO2007062135A2 (en) | 2005-11-23 | 2007-05-31 | Junji Shiraishi | Computer-aided method for detection of interval changes in successive whole-body bone scans and related computer program product and system |
| WO2007102074A2 (en) | 2006-03-07 | 2007-09-13 | Cadila Healthcare Limited | Salts of quetiapine |
| CA2645137A1 (en) | 2006-03-07 | 2007-09-13 | James F. Blake | Heterobicyclic pyrazole compounds and methods of use |
| WO2007109799A2 (en) | 2006-03-23 | 2007-09-27 | Teva Pharmaceutical Industries Ltd. | Polymorphs of eszopiclone malate |
| US20080004273A1 (en) | 2006-05-30 | 2008-01-03 | Stephane Raeppel | Inhibitors of protein tyrosine kinase activity |
| ES2375284T3 (es) | 2006-08-23 | 2012-02-28 | Eisai R&D Management Co., Ltd. | Sal de un derivado de fenoxipiridina, o cristal de la misma, y procedimiento de producción de la misma. |
| US20100074897A1 (en) | 2006-12-01 | 2010-03-25 | University Of Utah Research Foundation | Methods and Compositions related to HIF-1 alpha |
| EP2101759B1 (en) | 2006-12-14 | 2018-10-10 | Exelixis, Inc. | Methods of using mek inhibitors |
| US20080200515A1 (en) | 2006-12-29 | 2008-08-21 | Tap Pharmaceutical Products Inc. | Solid state forms of enantiopure ilaprazole |
| CA2812705C (en) | 2007-12-19 | 2015-08-18 | Sunovion Pharmaceuticals Inc. | Maleate, besylate and l-malate salts of 6-(5-chloro-2-pyridyl)-5-[(4-methyl-1-piperazinyl)carbonyloxy]-7-oxo-6,7-dihydro-5h-pyrrolo[3,4-b]pyrazine |
| UY31800A (es) | 2008-05-05 | 2009-11-10 | Smithkline Beckman Corp | Metodo de tratamiento de cancer usando un inhibidor de cmet y axl y un inhibidor de erbb |
| TW201035017A (en) | 2008-09-26 | 2010-10-01 | Smithkline Beecham Corp | Preparation of a quinolinyloxydiphenylcyclopropanedicarboxamide |
| AU2009300328A1 (en) | 2008-10-01 | 2010-04-08 | Ludwig Institute For Cancer Research | Methods for the treatment of cancer |
| CN102282134B (zh) | 2008-11-13 | 2015-04-01 | 埃克塞里艾克西斯公司 | 喹啉衍生物制备方法 |
| TW201028383A (en) | 2008-12-04 | 2010-08-01 | Exelixis Inc | Methods of preparing quinoline derivatives |
| US8877776B2 (en) | 2009-01-16 | 2014-11-04 | Exelixis, Inc. | (L)-malate salt of N-(4-{[6,7-bis(methyloxy) quinolin-4-yl]oxy}phenyl)-N'-(4-fluorophenyl)cyclopropane-1,1-dicarboxamide |
| WO2011009095A1 (en) | 2009-07-17 | 2011-01-20 | Exelixis, Inc. | Crystalline forms of n-[3-fluoro-4-({6-(methyloxy)-7-[(3-morpholin-4-ylpropyl)oxy]-quin0lin-4-yl}oxy)phenyl]-n'-(4-fluorophenyl)cyclopropane-1, 1-dicarboxamide |
| UA108618C2 (uk) | 2009-08-07 | 2015-05-25 | Застосування c-met-модуляторів в комбінації з темозоломідом та/або променевою терапією для лікування раку | |
| WO2011031840A1 (en) | 2009-09-09 | 2011-03-17 | Quintiles Transnational Corp. | Methods and compositions for the treatment of receptor tyrosine kinase mediated diseases or disorders |
| TW201202228A (en) | 2010-03-12 | 2012-01-16 | Exelixis Inc | Hydrated crystalline forms of N-[3-fluoro-4-({6-(methyloxy)-7-[(3-morpholin-4-ylpropyl)oxy]-quinolin-4-yl}oxy)phenyl]-N'-(4-fluorophenyl)cyclopropane-1, 1-dicarboxamide |
| US20120070368A1 (en) | 2010-04-16 | 2012-03-22 | Exelixis, Inc. | Methods of Using C-Met Modulators |
| CN114209699A (zh) | 2010-07-16 | 2022-03-22 | 埃克塞里艾克西斯公司 | C-met调节剂药物组合物 |
| PL2621481T5 (pl) | 2010-09-27 | 2023-03-13 | Exelixis, Inc. | Podwójne inhibitory MET i VEGF do leczenia opornego na kastrację raka prostaty i osteoblastycznych przerzutów do kości |
| KR20200031711A (ko) | 2011-02-10 | 2020-03-24 | 엑셀리시스, 인코포레이티드 | 퀴놀린 화합물들의 제조 방법들 및 상기 화합물들을 함유하는 약학 조성물들 |
| US20120252840A1 (en) | 2011-04-04 | 2012-10-04 | Exelixis, Inc. | Method of Treating Cancer |
| AU2012312364B2 (en) | 2011-09-22 | 2017-11-09 | Exelixis, Inc. | Method for treating osteoporosis |
| EP2844254A1 (en) | 2012-05-02 | 2015-03-11 | Exelixis, Inc. | A dual met - vegf modulator for treating osteolytic bone metastases |
| EP2892532B1 (en) | 2012-09-07 | 2019-02-13 | Exelixis, Inc. | Inhibitors of met, vegfr and ret for use in the treatment of lung adenocarcinoma |
| HK1221424A1 (zh) | 2013-04-04 | 2017-06-02 | Exelixis, Inc. | 治疗癌症的药物组合 |
| EA201691850A1 (ru) | 2014-03-17 | 2016-12-30 | Экселиксис, Инк. | Дозирование составов, содержащих кабозантиниб |
-
2010
- 2010-01-15 US US13/145,054 patent/US8877776B2/en active Active
- 2010-01-15 TW TW109120834A patent/TW202112751A/zh unknown
- 2010-01-15 HR HRP20130458TT patent/HRP20130458T4/hr unknown
- 2010-01-15 RS RS20130135A patent/RS52754B2/sr unknown
- 2010-01-15 PL PL10701175.1T patent/PL2387563T5/pl unknown
- 2010-01-15 NZ NZ779754A patent/NZ779754A/en unknown
- 2010-01-15 KR KR1020237000106A patent/KR20230008268A/ko not_active Ceased
- 2010-01-15 EA EA201170941A patent/EA019959B1/ru unknown
- 2010-01-15 CA CA2995880A patent/CA2995880C/en active Active
- 2010-01-15 TW TW104129563A patent/TWI577664B/zh active
- 2010-01-15 CN CN201610140984.2A patent/CN106083714B/zh not_active Ceased
- 2010-01-15 KR KR1020207006546A patent/KR102187034B1/ko active Active
- 2010-01-15 KR KR1020247001034A patent/KR20240011244A/ko not_active Ceased
- 2010-01-15 PT PT107011751T patent/PT2387563E/pt unknown
- 2010-01-15 KR KR1020177011913A patent/KR20170052702A/ko not_active Ceased
- 2010-01-15 TW TW107128219A patent/TW201920110A/zh unknown
- 2010-01-15 BR BRPI1006812A patent/BRPI1006812B8/pt active IP Right Grant
- 2010-01-15 AR ARP100100098A patent/AR075025A1/es not_active Application Discontinuation
- 2010-01-15 MX MX2012010502A patent/MX350898B/es unknown
- 2010-01-15 EA EA201692224A patent/EA038195B1/ru active Protection Beyond IP Right Term
- 2010-01-15 SG SG2011051422A patent/SG173014A1/en unknown
- 2010-01-15 CN CN2010800126565A patent/CN102388024A/zh not_active Withdrawn
- 2010-01-15 MX MX2011007620A patent/MX2011007620A/es active IP Right Grant
- 2010-01-15 CN CN201910791380.8A patent/CN110818633A/zh active Pending
- 2010-01-15 CA CA2758030A patent/CA2758030C/en active Active
- 2010-01-15 DK DK10701175.1T patent/DK2387563T4/da active
- 2010-01-15 TW TW105143226A patent/TWI641593B/zh active
- 2010-01-15 EA EA201400110A patent/EA026425B1/ru active Protection Beyond IP Right Term
- 2010-01-15 KR KR1020207034429A patent/KR20200137052A/ko not_active Ceased
- 2010-01-15 TW TW103115685A patent/TWI511956B/zh active
- 2010-01-15 KR KR1020187026467A patent/KR102088588B1/ko active Active
- 2010-01-15 AU AU2010204619A patent/AU2010204619B2/en active Active
- 2010-01-15 NZ NZ594594A patent/NZ594594A/xx unknown
- 2010-01-15 TW TW111100787A patent/TW202241853A/zh unknown
- 2010-01-15 KR KR1020117018924A patent/KR101733773B1/ko active Active
- 2010-01-15 NZ NZ618004A patent/NZ618004A/en unknown
- 2010-01-15 EP EP10701175.1A patent/EP2387563B2/en active Active
- 2010-01-15 WO PCT/US2010/021194 patent/WO2010083414A1/en not_active Ceased
- 2010-01-15 KR KR1020257026007A patent/KR20250123237A/ko active Pending
- 2010-01-15 KR KR1020217039169A patent/KR20210151988A/ko not_active Ceased
- 2010-01-15 TW TW099101140A patent/TWI447108B/zh active
- 2010-01-15 ES ES10701175T patent/ES2402524T5/es active Active
- 2010-01-15 JP JP2011546390A patent/JP5677318B2/ja active Active
-
2011
- 2011-07-13 ZA ZA2011/05167A patent/ZA201105167B/en unknown
- 2011-07-14 IL IL214086A patent/IL214086A/en active IP Right Grant
- 2011-07-15 MX MX2022014444A patent/MX2022014444A/es unknown
-
2013
- 2013-04-11 AU AU2013203780A patent/AU2013203780B2/en active Active
- 2013-05-15 SM SM201300050T patent/SMT201300050B/xx unknown
-
2014
- 2014-07-25 US US14/340,871 patent/US9809549B2/en active Active
- 2014-08-13 IL IL234097A patent/IL234097B/en active IP Right Grant
- 2014-12-26 JP JP2014264071A patent/JP5980894B2/ja active Active
-
2016
- 2016-07-27 JP JP2016146934A patent/JP6342456B2/ja active Active
- 2016-11-24 AU AU2016262732A patent/AU2016262732B2/en active Active
-
2017
- 2017-06-08 US US15/617,725 patent/US20180002289A1/en not_active Abandoned
- 2017-06-09 US US15/618,593 patent/US20180037552A1/en not_active Abandoned
- 2017-06-13 US US15/621,527 patent/US20170275251A1/en not_active Abandoned
-
2018
- 2018-05-16 JP JP2018094391A patent/JP6542429B2/ja active Active
-
2019
- 2019-06-12 JP JP2019109267A patent/JP6931372B2/ja active Active
-
2020
- 2020-02-20 US US16/796,250 patent/US20200190032A1/en not_active Abandoned
- 2020-10-14 US US17/070,514 patent/US11091439B2/en active Active
-
2021
- 2021-01-14 US US17/149,365 patent/US11091440B2/en active Active
- 2021-02-09 US US17/171,752 patent/US11098015B2/en active Active
- 2021-07-09 US US17/371,372 patent/US20220259151A9/en not_active Abandoned
- 2021-08-13 JP JP2021131901A patent/JP2021191756A/ja active Pending
- 2021-10-19 US US17/505,275 patent/US20220033357A1/en not_active Abandoned
-
2023
- 2023-01-20 US US18/157,568 patent/US20230167063A1/en not_active Abandoned
-
2024
- 2024-03-21 US US18/612,538 patent/US20240279181A1/en not_active Abandoned
- 2024-11-25 US US18/958,628 patent/US12275706B2/en active Active
-
2025
- 2025-03-04 US US19/069,917 patent/US20250243165A1/en not_active Abandoned
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US12275706B2 (en) | Malate salt of N-(4-{[6,7-bis(methyloxy) quinolin-4-yl]oxy}phenyl)-N′-(4-fluorophenyl)cyclopropane-1,1-dicarboxamide, and crystalline forms thereof for the treatment of cancer | |
| HK1231054B (zh) | N-(4-{[6,7-雙(甲基氧基)喹啉-4-基]氧基}苯基)-n'-(4-氟苯基)環丙烷-1,1-二甲酰胺蘋果酸鹽及其結晶型 | |
| HK1164305B (en) | Malate salt of n- (4- { [ 6, 7-bis (methyloxy) quinolin-4-yl]oxy}phenyl-n' - (4 -fluorophenyl) cyclopropane-1,1-dicarboxamide, and crystalline forms thereof for the treatment of cancer |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A107 | Divisional application of patent | ||
| PA0104 | Divisional application for international application |
Comment text: Divisional Application for International Patent Patent event code: PA01041R01D Patent event date: 20170428 Application number text: 1020117018924 Filing date: 20110812 |
|
| PG1501 | Laying open of application | ||
| A201 | Request for examination | ||
| PA0201 | Request for examination |
Patent event code: PA02012R01D Patent event date: 20170526 Comment text: Request for Examination of Application |
|
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
Comment text: Notification of reason for refusal Patent event date: 20170720 Patent event code: PE09021S01D |
|
| E601 | Decision to refuse application | ||
| PE0601 | Decision on rejection of patent |
Patent event date: 20180614 Comment text: Decision to Refuse Application Patent event code: PE06012S01D Patent event date: 20170720 Comment text: Notification of reason for refusal Patent event code: PE06011S01I |