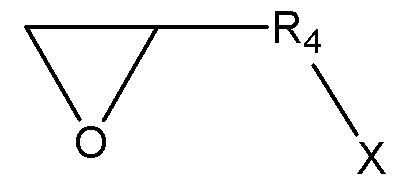KR20170100111A - 인계 에폭시 화합물 및 이의 제조방법, 이를 포함하는 에폭시 조성물 - Google Patents
인계 에폭시 화합물 및 이의 제조방법, 이를 포함하는 에폭시 조성물 Download PDFInfo
- Publication number
- KR20170100111A KR20170100111A KR1020160022168A KR20160022168A KR20170100111A KR 20170100111 A KR20170100111 A KR 20170100111A KR 1020160022168 A KR1020160022168 A KR 1020160022168A KR 20160022168 A KR20160022168 A KR 20160022168A KR 20170100111 A KR20170100111 A KR 20170100111A
- Authority
- KR
- South Korea
- Prior art keywords
- phosphorus
- hydrocarbon group
- epoxy compound
- based epoxy
- weight
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 239000004593 Epoxy Substances 0.000 title claims abstract description 175
- 229910052698 phosphorus Inorganic materials 0.000 title claims abstract description 160
- 239000011574 phosphorus Substances 0.000 title claims abstract description 160
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 title claims abstract description 154
- 150000001875 compounds Chemical class 0.000 title claims abstract description 123
- 239000000203 mixture Substances 0.000 title claims abstract description 57
- 238000000034 method Methods 0.000 title claims description 25
- 239000003795 chemical substances by application Substances 0.000 claims description 42
- 125000001931 aliphatic group Chemical group 0.000 claims description 32
- 125000002029 aromatic hydrocarbon group Chemical group 0.000 claims description 30
- 229910052760 oxygen Inorganic materials 0.000 claims description 28
- 229910052717 sulfur Inorganic materials 0.000 claims description 28
- 125000005842 heteroatom Chemical group 0.000 claims description 27
- 239000002904 solvent Substances 0.000 claims description 25
- 239000011541 reaction mixture Substances 0.000 claims description 18
- -1 pyridine compound Chemical class 0.000 claims description 17
- RAXXELZNTBOGNW-UHFFFAOYSA-N imidazole Natural products C1=CNC=N1 RAXXELZNTBOGNW-UHFFFAOYSA-N 0.000 claims description 15
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N Phenol Chemical compound OC1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 claims description 12
- 239000003054 catalyst Substances 0.000 claims description 11
- 150000002440 hydroxy compounds Chemical class 0.000 claims description 10
- 229920003986 novolac Polymers 0.000 claims description 10
- 239000000463 material Substances 0.000 claims description 9
- 239000000126 substance Substances 0.000 claims description 9
- SLAFUPJSGFVWPP-UHFFFAOYSA-M ethyl(triphenyl)phosphanium;iodide Chemical compound [I-].C=1C=CC=CC=1[P+](C=1C=CC=CC=1)(CC)C1=CC=CC=C1 SLAFUPJSGFVWPP-UHFFFAOYSA-M 0.000 claims description 7
- ICSNLGPSRYBMBD-UHFFFAOYSA-N 2-aminopyridine Chemical compound NC1=CC=CC=N1 ICSNLGPSRYBMBD-UHFFFAOYSA-N 0.000 claims description 6
- CUYKNJBYIJFRCU-UHFFFAOYSA-N 3-aminopyridine Chemical compound NC1=CC=CN=C1 CUYKNJBYIJFRCU-UHFFFAOYSA-N 0.000 claims description 6
- VHYFNPMBLIVWCW-UHFFFAOYSA-N 4-Dimethylaminopyridine Chemical compound CN(C)C1=CC=NC=C1 VHYFNPMBLIVWCW-UHFFFAOYSA-N 0.000 claims description 6
- 229920000642 polymer Polymers 0.000 claims description 6
- 229910052739 hydrogen Inorganic materials 0.000 claims description 5
- 239000001257 hydrogen Substances 0.000 claims description 5
- 239000012948 isocyanate Substances 0.000 claims description 5
- LXBGSDVWAMZHDD-UHFFFAOYSA-N 2-methyl-1h-imidazole Chemical compound CC1=NC=CN1 LXBGSDVWAMZHDD-UHFFFAOYSA-N 0.000 claims description 4
- 150000001408 amides Chemical class 0.000 claims description 4
- 230000009477 glass transition Effects 0.000 claims description 4
- 238000002360 preparation method Methods 0.000 claims description 4
- YTWBFUCJVWKCCK-UHFFFAOYSA-N 2-heptadecyl-1h-imidazole Chemical compound CCCCCCCCCCCCCCCCCC1=NC=CN1 YTWBFUCJVWKCCK-UHFFFAOYSA-N 0.000 claims description 3
- LLEASVZEQBICSN-UHFFFAOYSA-N 2-undecyl-1h-imidazole Chemical compound CCCCCCCCCCCC1=NC=CN1 LLEASVZEQBICSN-UHFFFAOYSA-N 0.000 claims description 3
- NUKYPUAOHBNCPY-UHFFFAOYSA-N 4-aminopyridine Chemical compound NC1=CC=NC=C1 NUKYPUAOHBNCPY-UHFFFAOYSA-N 0.000 claims description 3
- RGUKYNXWOWSRET-UHFFFAOYSA-N 4-pyrrolidin-1-ylpyridine Chemical compound C1CCCN1C1=CC=NC=C1 RGUKYNXWOWSRET-UHFFFAOYSA-N 0.000 claims description 3
- ULKLGIFJWFIQFF-UHFFFAOYSA-N 5K8XI641G3 Chemical compound CCC1=NC=C(C)N1 ULKLGIFJWFIQFF-UHFFFAOYSA-N 0.000 claims description 3
- QUXLCYFNVNNRBE-UHFFFAOYSA-N 6-methylpyridin-2-amine Chemical compound CC1=CC=CC(N)=N1 QUXLCYFNVNNRBE-UHFFFAOYSA-N 0.000 claims description 3
- 150000008065 acid anhydrides Chemical class 0.000 claims description 3
- 229960004979 fampridine Drugs 0.000 claims description 3
- 125000005843 halogen group Chemical group 0.000 claims description 3
- 125000004435 hydrogen atom Chemical class [H]* 0.000 claims description 3
- HMMPCBAWTWYFLR-UHFFFAOYSA-N n-pyridin-2-ylpyridin-2-amine Chemical compound C=1C=CC=NC=1NC1=CC=CC=N1 HMMPCBAWTWYFLR-UHFFFAOYSA-N 0.000 claims description 3
- 150000002894 organic compounds Chemical class 0.000 claims description 3
- 229920000768 polyamine Polymers 0.000 claims description 3
- JUJWROOIHBZHMG-UHFFFAOYSA-N pyridine Substances C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 claims description 3
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 claims description 3
- 150000003512 tertiary amines Chemical class 0.000 claims description 3
- XZZNDPSIHUTMOC-UHFFFAOYSA-N triphenyl phosphate Chemical compound C=1C=CC=CC=1OP(OC=1C=CC=CC=1)(=O)OC1=CC=CC=C1 XZZNDPSIHUTMOC-UHFFFAOYSA-N 0.000 claims description 3
- 150000004963 1-benzylimidazoles Chemical class 0.000 claims 1
- HAHXPMFFMRTIJV-UHFFFAOYSA-N 6-propan-2-ylpyridin-2-amine Chemical compound CC(C)C1=CC=CC(N)=N1 HAHXPMFFMRTIJV-UHFFFAOYSA-N 0.000 claims 1
- 238000004519 manufacturing process Methods 0.000 abstract description 7
- 229920000647 polyepoxide Polymers 0.000 abstract description 3
- 125000003700 epoxy group Chemical group 0.000 abstract description 2
- 239000003822 epoxy resin Substances 0.000 abstract description 2
- 150000003017 phosphorus Chemical class 0.000 abstract description 2
- BHEPBYXIRTUNPN-UHFFFAOYSA-N hydridophosphorus(.) (triplet) Chemical compound [PH] BHEPBYXIRTUNPN-UHFFFAOYSA-N 0.000 abstract 1
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 30
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 21
- 230000000052 comparative effect Effects 0.000 description 18
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 15
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 14
- 239000000047 product Substances 0.000 description 11
- RNFJDJUURJAICM-UHFFFAOYSA-N 2,2,4,4,6,6-hexaphenoxy-1,3,5-triaza-2$l^{5},4$l^{5},6$l^{5}-triphosphacyclohexa-1,3,5-triene Chemical compound N=1P(OC=2C=CC=CC=2)(OC=2C=CC=CC=2)=NP(OC=2C=CC=CC=2)(OC=2C=CC=CC=2)=NP=1(OC=1C=CC=CC=1)OC1=CC=CC=C1 RNFJDJUURJAICM-UHFFFAOYSA-N 0.000 description 10
- NTIZESTWPVYFNL-UHFFFAOYSA-N Methyl isobutyl ketone Chemical compound CC(C)CC(C)=O NTIZESTWPVYFNL-UHFFFAOYSA-N 0.000 description 10
- UIHCLUNTQKBZGK-UHFFFAOYSA-N Methyl isobutyl ketone Natural products CCC(C)C(C)=O UIHCLUNTQKBZGK-UHFFFAOYSA-N 0.000 description 10
- MTHSVFCYNBDYFN-UHFFFAOYSA-N diethylene glycol Chemical compound OCCOCCO MTHSVFCYNBDYFN-UHFFFAOYSA-N 0.000 description 10
- 239000003063 flame retardant Substances 0.000 description 10
- 229940043265 methyl isobutyl ketone Drugs 0.000 description 10
- 238000000746 purification Methods 0.000 description 9
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 8
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 8
- 238000006243 chemical reaction Methods 0.000 description 8
- UPMLOUAZCHDJJD-UHFFFAOYSA-N 4,4'-Diphenylmethane Diisocyanate Chemical compound C1=CC(N=C=O)=CC=C1CC1=CC=C(N=C=O)C=C1 UPMLOUAZCHDJJD-UHFFFAOYSA-N 0.000 description 7
- PGYPOBZJRVSMDS-UHFFFAOYSA-N loperamide hydrochloride Chemical compound Cl.C=1C=CC=CC=1C(C=1C=CC=CC=1)(C(=O)N(C)C)CCN(CC1)CCC1(O)C1=CC=C(Cl)C=C1 PGYPOBZJRVSMDS-UHFFFAOYSA-N 0.000 description 7
- 238000003786 synthesis reaction Methods 0.000 description 7
- ZWEHNKRNPOVVGH-UHFFFAOYSA-N 2-Butanone Chemical compound CCC(C)=O ZWEHNKRNPOVVGH-UHFFFAOYSA-N 0.000 description 6
- 229910052802 copper Inorganic materials 0.000 description 6
- 239000010949 copper Substances 0.000 description 6
- 239000000654 additive Substances 0.000 description 5
- 229910052783 alkali metal Inorganic materials 0.000 description 5
- 125000003118 aryl group Chemical group 0.000 description 5
- 125000001570 methylene group Chemical group [H]C([H])([*:1])[*:2] 0.000 description 5
- 239000005056 polyisocyanate Substances 0.000 description 5
- 229920001228 polyisocyanate Polymers 0.000 description 5
- 238000010992 reflux Methods 0.000 description 5
- 238000005406 washing Methods 0.000 description 5
- LRHPLDYGYMQRHN-UHFFFAOYSA-N N-Butanol Chemical compound CCCCO LRHPLDYGYMQRHN-UHFFFAOYSA-N 0.000 description 4
- 239000003513 alkali Substances 0.000 description 4
- 238000004090 dissolution Methods 0.000 description 4
- ZSIAUFGUXNUGDI-UHFFFAOYSA-N hexan-1-ol Chemical compound CCCCCCO ZSIAUFGUXNUGDI-UHFFFAOYSA-N 0.000 description 4
- 229910052757 nitrogen Inorganic materials 0.000 description 4
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 4
- 125000006832 (C1-C10) alkylene group Chemical group 0.000 description 3
- ARXJGSRGQADJSQ-UHFFFAOYSA-N 1-methoxypropan-2-ol Chemical compound COCC(C)O ARXJGSRGQADJSQ-UHFFFAOYSA-N 0.000 description 3
- LLOXZCFOAUCDAE-UHFFFAOYSA-N 2-diphenylphosphorylbenzene-1,4-diol Chemical compound OC1=CC=C(O)C(P(=O)(C=2C=CC=CC=2)C=2C=CC=CC=2)=C1 LLOXZCFOAUCDAE-UHFFFAOYSA-N 0.000 description 3
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 3
- BRLQWZUYTZBJKN-UHFFFAOYSA-N Epichlorohydrin Chemical compound ClCC1CO1 BRLQWZUYTZBJKN-UHFFFAOYSA-N 0.000 description 3
- WMFOQBRAJBCJND-UHFFFAOYSA-M Lithium hydroxide Chemical compound [Li+].[OH-] WMFOQBRAJBCJND-UHFFFAOYSA-M 0.000 description 3
- AMQJEAYHLZJPGS-UHFFFAOYSA-N N-Pentanol Chemical compound CCCCCO AMQJEAYHLZJPGS-UHFFFAOYSA-N 0.000 description 3
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 3
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 3
- 230000000996 additive effect Effects 0.000 description 3
- 150000001340 alkali metals Chemical class 0.000 description 3
- 230000015572 biosynthetic process Effects 0.000 description 3
- IISBACLAFKSPIT-UHFFFAOYSA-N bisphenol A Chemical compound C=1C=C(O)C=CC=1C(C)(C)C1=CC=C(O)C=C1 IISBACLAFKSPIT-UHFFFAOYSA-N 0.000 description 3
- 238000004132 cross linking Methods 0.000 description 3
- 230000000694 effects Effects 0.000 description 3
- 125000000816 ethylene group Chemical group [H]C([H])([*:1])C([H])([H])[*:2] 0.000 description 3
- 238000010438 heat treatment Methods 0.000 description 3
- 239000000843 powder Substances 0.000 description 3
- FBHPRUXJQNWTEW-UHFFFAOYSA-N 1-benzyl-2-methylimidazole Chemical compound CC1=NC=CN1CC1=CC=CC=C1 FBHPRUXJQNWTEW-UHFFFAOYSA-N 0.000 description 2
- PTTPXKJBFFKCEK-UHFFFAOYSA-N 2-Methyl-4-heptanone Chemical compound CC(C)CC(=O)CC(C)C PTTPXKJBFFKCEK-UHFFFAOYSA-N 0.000 description 2
- XYGFISRAXLLACA-UHFFFAOYSA-N 6-propan-2-ylpyridin-3-amine Chemical compound CC(C)C1=CC=C(N)C=N1 XYGFISRAXLLACA-UHFFFAOYSA-N 0.000 description 2
- DLFVBJFMPXGRIB-UHFFFAOYSA-N Acetamide Chemical compound CC(N)=O DLFVBJFMPXGRIB-UHFFFAOYSA-N 0.000 description 2
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 2
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 2
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 2
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 2
- SECXISVLQFMRJM-UHFFFAOYSA-N N-Methylpyrrolidone Chemical compound CN1CCCC1=O SECXISVLQFMRJM-UHFFFAOYSA-N 0.000 description 2
- ATHHXGZTWNVVOU-UHFFFAOYSA-N N-methylformamide Chemical compound CNC=O ATHHXGZTWNVVOU-UHFFFAOYSA-N 0.000 description 2
- WCUXLLCKKVVCTQ-UHFFFAOYSA-M Potassium chloride Chemical compound [Cl-].[K+] WCUXLLCKKVVCTQ-UHFFFAOYSA-M 0.000 description 2
- NBBJYMSMWIIQGU-UHFFFAOYSA-N Propionic aldehyde Chemical compound CCC=O NBBJYMSMWIIQGU-UHFFFAOYSA-N 0.000 description 2
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 2
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 2
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 2
- WQDUMFSSJAZKTM-UHFFFAOYSA-N Sodium methoxide Chemical compound [Na+].[O-]C WQDUMFSSJAZKTM-UHFFFAOYSA-N 0.000 description 2
- YRKCREAYFQTBPV-UHFFFAOYSA-N acetylacetone Chemical compound CC(=O)CC(C)=O YRKCREAYFQTBPV-UHFFFAOYSA-N 0.000 description 2
- 125000003172 aldehyde group Chemical group 0.000 description 2
- 125000005577 anthracene group Chemical group 0.000 description 2
- 125000005428 anthryl group Chemical group [H]C1=C([H])C([H])=C2C([H])=C3C(*)=C([H])C([H])=C([H])C3=C([H])C2=C1[H] 0.000 description 2
- TZCXTZWJZNENPQ-UHFFFAOYSA-L barium sulfate Chemical compound [Ba+2].[O-]S([O-])(=O)=O TZCXTZWJZNENPQ-UHFFFAOYSA-L 0.000 description 2
- 239000002585 base Substances 0.000 description 2
- HUMNYLRZRPPJDN-UHFFFAOYSA-N benzaldehyde Chemical compound O=CC1=CC=CC=C1 HUMNYLRZRPPJDN-UHFFFAOYSA-N 0.000 description 2
- 125000006267 biphenyl group Chemical group 0.000 description 2
- PXKLMJQFEQBVLD-UHFFFAOYSA-N bisphenol F Chemical compound C1=CC(O)=CC=C1CC1=CC=C(O)C=C1 PXKLMJQFEQBVLD-UHFFFAOYSA-N 0.000 description 2
- 125000001246 bromo group Chemical group Br* 0.000 description 2
- YCIMNLLNPGFGHC-UHFFFAOYSA-N catechol Chemical compound OC1=CC=CC=C1O YCIMNLLNPGFGHC-UHFFFAOYSA-N 0.000 description 2
- 125000001309 chloro group Chemical group Cl* 0.000 description 2
- 239000011889 copper foil Substances 0.000 description 2
- JHIVVAPYMSGYDF-UHFFFAOYSA-N cyclohexanone Chemical compound O=C1CCCCC1 JHIVVAPYMSGYDF-UHFFFAOYSA-N 0.000 description 2
- 230000007613 environmental effect Effects 0.000 description 2
- 239000010408 film Substances 0.000 description 2
- 238000009472 formulation Methods 0.000 description 2
- 239000011521 glass Substances 0.000 description 2
- 239000003365 glass fiber Substances 0.000 description 2
- 229910052736 halogen Inorganic materials 0.000 description 2
- 150000002367 halogens Chemical class 0.000 description 2
- 150000002431 hydrogen Chemical class 0.000 description 2
- 150000002460 imidazoles Chemical class 0.000 description 2
- 239000011256 inorganic filler Substances 0.000 description 2
- 229910003475 inorganic filler Inorganic materials 0.000 description 2
- 239000011810 insulating material Substances 0.000 description 2
- ZXEKIIBDNHEJCQ-UHFFFAOYSA-N isobutanol Chemical compound CC(C)CO ZXEKIIBDNHEJCQ-UHFFFAOYSA-N 0.000 description 2
- KWGKDLIKAYFUFQ-UHFFFAOYSA-M lithium chloride Chemical compound [Li+].[Cl-] KWGKDLIKAYFUFQ-UHFFFAOYSA-M 0.000 description 2
- 239000011777 magnesium Substances 0.000 description 2
- 229910052749 magnesium Inorganic materials 0.000 description 2
- 229910052751 metal Inorganic materials 0.000 description 2
- 239000002184 metal Substances 0.000 description 2
- 238000002156 mixing Methods 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 239000000178 monomer Substances 0.000 description 2
- 125000005561 phenanthryl group Chemical group 0.000 description 2
- 238000003825 pressing Methods 0.000 description 2
- BDERNNFJNOPAEC-UHFFFAOYSA-N propan-1-ol Chemical compound CCCO BDERNNFJNOPAEC-UHFFFAOYSA-N 0.000 description 2
- ZZYXNRREDYWPLN-UHFFFAOYSA-N pyridine-2,3-diamine Chemical compound NC1=CC=CN=C1N ZZYXNRREDYWPLN-UHFFFAOYSA-N 0.000 description 2
- MIROPXUFDXCYLG-UHFFFAOYSA-N pyridine-2,5-diamine Chemical compound NC1=CC=C(N)N=C1 MIROPXUFDXCYLG-UHFFFAOYSA-N 0.000 description 2
- VHNQIURBCCNWDN-UHFFFAOYSA-N pyridine-2,6-diamine Chemical compound NC1=CC=CC(N)=N1 VHNQIURBCCNWDN-UHFFFAOYSA-N 0.000 description 2
- 239000002994 raw material Substances 0.000 description 2
- SMQUZDBALVYZAC-UHFFFAOYSA-N salicylaldehyde Chemical compound OC1=CC=CC=C1C=O SMQUZDBALVYZAC-UHFFFAOYSA-N 0.000 description 2
- VWDWKYIASSYTQR-UHFFFAOYSA-N sodium nitrate Chemical compound [Na+].[O-][N+]([O-])=O VWDWKYIASSYTQR-UHFFFAOYSA-N 0.000 description 2
- 238000009849 vacuum degassing Methods 0.000 description 2
- MTZUIIAIAKMWLI-UHFFFAOYSA-N 1,2-diisocyanatobenzene Chemical compound O=C=NC1=CC=CC=C1N=C=O MTZUIIAIAKMWLI-UHFFFAOYSA-N 0.000 description 1
- ZXHZWRZAWJVPIC-UHFFFAOYSA-N 1,2-diisocyanatonaphthalene Chemical compound C1=CC=CC2=C(N=C=O)C(N=C=O)=CC=C21 ZXHZWRZAWJVPIC-UHFFFAOYSA-N 0.000 description 1
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 1
- FZTLLUYFWAOGGB-UHFFFAOYSA-N 1,4-dioxane dioxane Chemical compound C1COCCO1.C1COCCO1 FZTLLUYFWAOGGB-UHFFFAOYSA-N 0.000 description 1
- RWNUSVWFHDHRCJ-UHFFFAOYSA-N 1-butoxypropan-2-ol Chemical compound CCCCOCC(C)O RWNUSVWFHDHRCJ-UHFFFAOYSA-N 0.000 description 1
- PAUHLEIGHAUFAK-UHFFFAOYSA-N 1-isocyanato-1-[(1-isocyanatocyclohexyl)methyl]cyclohexane Chemical compound C1CCCCC1(N=C=O)CC1(N=C=O)CCCCC1 PAUHLEIGHAUFAK-UHFFFAOYSA-N 0.000 description 1
- KJCVRFUGPWSIIH-UHFFFAOYSA-N 1-naphthol Chemical compound C1=CC=C2C(O)=CC=CC2=C1 KJCVRFUGPWSIIH-UHFFFAOYSA-N 0.000 description 1
- HECLRDQVFMWTQS-RGOKHQFPSA-N 1755-01-7 Chemical compound C1[C@H]2[C@@H]3CC=C[C@@H]3[C@@H]1C=C2 HECLRDQVFMWTQS-RGOKHQFPSA-N 0.000 description 1
- KMRIWYPVRWEWRG-UHFFFAOYSA-N 2-(6-oxobenzo[c][2,1]benzoxaphosphinin-6-yl)benzene-1,4-diol Chemical compound OC1=CC=C(O)C(P2(=O)C3=CC=CC=C3C3=CC=CC=C3O2)=C1 KMRIWYPVRWEWRG-UHFFFAOYSA-N 0.000 description 1
- DJOYTAUERRJRAT-UHFFFAOYSA-N 2-(n-methyl-4-nitroanilino)acetonitrile Chemical compound N#CCN(C)C1=CC=C([N+]([O-])=O)C=C1 DJOYTAUERRJRAT-UHFFFAOYSA-N 0.000 description 1
- POAOYUHQDCAZBD-UHFFFAOYSA-N 2-butoxyethanol Chemical compound CCCCOCCO POAOYUHQDCAZBD-UHFFFAOYSA-N 0.000 description 1
- QTWJRLJHJPIABL-UHFFFAOYSA-N 2-methylphenol;3-methylphenol;4-methylphenol Chemical compound CC1=CC=C(O)C=C1.CC1=CC=CC(O)=C1.CC1=CC=CC=C1O QTWJRLJHJPIABL-UHFFFAOYSA-N 0.000 description 1
- VPWNQTHUCYMVMZ-UHFFFAOYSA-N 4,4'-sulfonyldiphenol Chemical compound C1=CC(O)=CC=C1S(=O)(=O)C1=CC=C(O)C=C1 VPWNQTHUCYMVMZ-UHFFFAOYSA-N 0.000 description 1
- 229910052582 BN Inorganic materials 0.000 description 1
- 229930185605 Bisphenol Natural products 0.000 description 1
- SDDLEVPIDBLVHC-UHFFFAOYSA-N Bisphenol Z Chemical compound C1=CC(O)=CC=C1C1(C=2C=CC(O)=CC=2)CCCCC1 SDDLEVPIDBLVHC-UHFFFAOYSA-N 0.000 description 1
- PZNSFCLAULLKQX-UHFFFAOYSA-N Boron nitride Chemical compound N#B PZNSFCLAULLKQX-UHFFFAOYSA-N 0.000 description 1
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- JOYRKODLDBILNP-UHFFFAOYSA-N Ethyl urethane Chemical compound CCOC(N)=O JOYRKODLDBILNP-UHFFFAOYSA-N 0.000 description 1
- 239000005057 Hexamethylene diisocyanate Substances 0.000 description 1
- 239000005058 Isophorone diisocyanate Substances 0.000 description 1
- FXHOOIRPVKKKFG-UHFFFAOYSA-N N,N-Dimethylacetamide Chemical compound CN(C)C(C)=O FXHOOIRPVKKKFG-UHFFFAOYSA-N 0.000 description 1
- OHLUUHNLEMFGTQ-UHFFFAOYSA-N N-methylacetamide Chemical compound CNC(C)=O OHLUUHNLEMFGTQ-UHFFFAOYSA-N 0.000 description 1
- OMRDSWJXRLDPBB-UHFFFAOYSA-N N=C=O.N=C=O.C1CCCCC1 Chemical compound N=C=O.N=C=O.C1CCCCC1 OMRDSWJXRLDPBB-UHFFFAOYSA-N 0.000 description 1
- IIGAAOXXRKTFAM-UHFFFAOYSA-N N=C=O.N=C=O.CC1=C(C)C(C)=C(C)C(C)=C1C Chemical compound N=C=O.N=C=O.CC1=C(C)C(C)=C(C)C(C)=C1C IIGAAOXXRKTFAM-UHFFFAOYSA-N 0.000 description 1
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 description 1
- KEAYESYHFKHZAL-UHFFFAOYSA-N Sodium Chemical compound [Na] KEAYESYHFKHZAL-UHFFFAOYSA-N 0.000 description 1
- UIIMBOGNXHQVGW-DEQYMQKBSA-M Sodium bicarbonate-14C Chemical compound [Na+].O[14C]([O-])=O UIIMBOGNXHQVGW-DEQYMQKBSA-M 0.000 description 1
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 1
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 1
- IKHGUXGNUITLKF-XPULMUKRSA-N acetaldehyde Chemical compound [14CH]([14CH3])=O IKHGUXGNUITLKF-XPULMUKRSA-N 0.000 description 1
- 239000000853 adhesive Substances 0.000 description 1
- 230000001070 adhesive effect Effects 0.000 description 1
- 150000004703 alkoxides Chemical class 0.000 description 1
- 125000002947 alkylene group Chemical group 0.000 description 1
- WNROFYMDJYEPJX-UHFFFAOYSA-K aluminium hydroxide Chemical compound [OH-].[OH-].[OH-].[Al+3] WNROFYMDJYEPJX-UHFFFAOYSA-K 0.000 description 1
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 1
- OJMOMXZKOWKUTA-UHFFFAOYSA-N aluminum;borate Chemical compound [Al+3].[O-]B([O-])[O-] OJMOMXZKOWKUTA-UHFFFAOYSA-N 0.000 description 1
- MWPLVEDNUUSJAV-UHFFFAOYSA-N anthracene Natural products C1=CC=CC2=CC3=CC=CC=C3C=C21 MWPLVEDNUUSJAV-UHFFFAOYSA-N 0.000 description 1
- 150000004945 aromatic hydrocarbons Chemical class 0.000 description 1
- 239000003849 aromatic solvent Substances 0.000 description 1
- JRPBQTZRNDNNOP-UHFFFAOYSA-N barium titanate Chemical compound [Ba+2].[Ba+2].[O-][Ti]([O-])([O-])[O-] JRPBQTZRNDNNOP-UHFFFAOYSA-N 0.000 description 1
- 229910002113 barium titanate Inorganic materials 0.000 description 1
- 229910021523 barium zirconate Inorganic materials 0.000 description 1
- DQBAOWPVHRWLJC-UHFFFAOYSA-N barium(2+);dioxido(oxo)zirconium Chemical compound [Ba+2].[O-][Zr]([O-])=O DQBAOWPVHRWLJC-UHFFFAOYSA-N 0.000 description 1
- 230000005540 biological transmission Effects 0.000 description 1
- 229910002115 bismuth titanate Inorganic materials 0.000 description 1
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 1
- 229910052794 bromium Inorganic materials 0.000 description 1
- ZTQSAGDEMFDKMZ-UHFFFAOYSA-N butyric aldehyde Natural products CCCC=O ZTQSAGDEMFDKMZ-UHFFFAOYSA-N 0.000 description 1
- 229910000019 calcium carbonate Inorganic materials 0.000 description 1
- AOWKSNWVBZGMTJ-UHFFFAOYSA-N calcium titanate Chemical compound [Ca+2].[O-][Ti]([O-])=O AOWKSNWVBZGMTJ-UHFFFAOYSA-N 0.000 description 1
- 150000001718 carbodiimides Chemical class 0.000 description 1
- 229910052799 carbon Inorganic materials 0.000 description 1
- 125000004432 carbon atom Chemical group C* 0.000 description 1
- 239000007795 chemical reaction product Substances 0.000 description 1
- 238000013329 compounding Methods 0.000 description 1
- 238000009833 condensation Methods 0.000 description 1
- 230000005494 condensation Effects 0.000 description 1
- DZCLVBQEPZQZNN-UHFFFAOYSA-N copper;phenol Chemical compound [Cu].OC1=CC=CC=C1 DZCLVBQEPZQZNN-UHFFFAOYSA-N 0.000 description 1
- 229930003836 cresol Natural products 0.000 description 1
- 238000000113 differential scanning calorimetry Methods 0.000 description 1
- 238000009792 diffusion process Methods 0.000 description 1
- GYZLOYUZLJXAJU-UHFFFAOYSA-N diglycidyl ether Chemical compound C1OC1COCC1CO1 GYZLOYUZLJXAJU-UHFFFAOYSA-N 0.000 description 1
- 125000005442 diisocyanate group Chemical group 0.000 description 1
- 239000003085 diluting agent Substances 0.000 description 1
- 238000010292 electrical insulation Methods 0.000 description 1
- 230000002708 enhancing effect Effects 0.000 description 1
- 239000000945 filler Substances 0.000 description 1
- 239000012467 final product Substances 0.000 description 1
- 125000000524 functional group Chemical group 0.000 description 1
- 150000002240 furans Chemical class 0.000 description 1
- 150000002366 halogen compounds Chemical class 0.000 description 1
- LNEPOXFFQSENCJ-UHFFFAOYSA-N haloperidol Chemical compound C1CC(O)(C=2C=CC(Cl)=CC=2)CCN1CCCC(=O)C1=CC=C(F)C=C1 LNEPOXFFQSENCJ-UHFFFAOYSA-N 0.000 description 1
- RRAMGCGOFNQTLD-UHFFFAOYSA-N hexamethylene diisocyanate Chemical compound O=C=NCCCCCCN=C=O RRAMGCGOFNQTLD-UHFFFAOYSA-N 0.000 description 1
- 150000004678 hydrides Chemical class 0.000 description 1
- 239000012433 hydrogen halide Substances 0.000 description 1
- 229910000039 hydrogen halide Inorganic materials 0.000 description 1
- 150000004679 hydroxides Chemical class 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- 239000012212 insulator Substances 0.000 description 1
- 239000013067 intermediate product Substances 0.000 description 1
- 229940035429 isobutyl alcohol Drugs 0.000 description 1
- IQPQWNKOIGAROB-UHFFFAOYSA-N isocyanate group Chemical group [N-]=C=O IQPQWNKOIGAROB-UHFFFAOYSA-N 0.000 description 1
- NIMLQBUJDJZYEJ-UHFFFAOYSA-N isophorone diisocyanate Chemical compound CC1(C)CC(N=C=O)CC(C)(CN=C=O)C1 NIMLQBUJDJZYEJ-UHFFFAOYSA-N 0.000 description 1
- 239000011499 joint compound Substances 0.000 description 1
- SIAPCJWMELPYOE-UHFFFAOYSA-N lithium hydride Chemical compound [LiH] SIAPCJWMELPYOE-UHFFFAOYSA-N 0.000 description 1
- 229910000103 lithium hydride Inorganic materials 0.000 description 1
- ZLNQQNXFFQJAID-UHFFFAOYSA-L magnesium carbonate Chemical compound [Mg+2].[O-]C([O-])=O ZLNQQNXFFQJAID-UHFFFAOYSA-L 0.000 description 1
- 239000001095 magnesium carbonate Substances 0.000 description 1
- 229910000021 magnesium carbonate Inorganic materials 0.000 description 1
- VTHJTEIRLNZDEV-UHFFFAOYSA-L magnesium dihydroxide Chemical compound [OH-].[OH-].[Mg+2] VTHJTEIRLNZDEV-UHFFFAOYSA-L 0.000 description 1
- 239000000347 magnesium hydroxide Substances 0.000 description 1
- 229910001862 magnesium hydroxide Inorganic materials 0.000 description 1
- WSFSSNUMVMOOMR-NJFSPNSNSA-N methanone Chemical compound O=[14CH2] WSFSSNUMVMOOMR-NJFSPNSNSA-N 0.000 description 1
- 239000010445 mica Substances 0.000 description 1
- 229910052618 mica group Inorganic materials 0.000 description 1
- 238000006011 modification reaction Methods 0.000 description 1
- NXPPAOGUKPJVDI-UHFFFAOYSA-N naphthalene-1,2-diol Chemical compound C1=CC=CC2=C(O)C(O)=CC=C21 NXPPAOGUKPJVDI-UHFFFAOYSA-N 0.000 description 1
- 150000007524 organic acids Chemical class 0.000 description 1
- 235000005985 organic acids Nutrition 0.000 description 1
- 239000003960 organic solvent Substances 0.000 description 1
- 239000003973 paint Substances 0.000 description 1
- QNGNSVIICDLXHT-UHFFFAOYSA-N para-ethylbenzaldehyde Natural products CCC1=CC=C(C=O)C=C1 QNGNSVIICDLXHT-UHFFFAOYSA-N 0.000 description 1
- ISWSIDIOOBJBQZ-UHFFFAOYSA-M phenolate Chemical compound [O-]C1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-M 0.000 description 1
- 150000002989 phenols Chemical class 0.000 description 1
- 230000000704 physical effect Effects 0.000 description 1
- 229920005862 polyol Polymers 0.000 description 1
- 150000003077 polyols Chemical class 0.000 description 1
- 239000001103 potassium chloride Substances 0.000 description 1
- 235000011164 potassium chloride Nutrition 0.000 description 1
- LLHKCFNBLRBOGN-UHFFFAOYSA-N propylene glycol methyl ether acetate Chemical compound COCC(C)OC(C)=O LLHKCFNBLRBOGN-UHFFFAOYSA-N 0.000 description 1
- HNJBEVLQSNELDL-UHFFFAOYSA-N pyrrolidin-2-one Chemical compound O=C1CCCN1 HNJBEVLQSNELDL-UHFFFAOYSA-N 0.000 description 1
- 230000009257 reactivity Effects 0.000 description 1
- 230000003014 reinforcing effect Effects 0.000 description 1
- 238000000518 rheometry Methods 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- 150000003333 secondary alcohols Chemical class 0.000 description 1
- 230000008054 signal transmission Effects 0.000 description 1
- 239000000377 silicon dioxide Substances 0.000 description 1
- 229910000029 sodium carbonate Inorganic materials 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- QDRKDTQENPPHOJ-UHFFFAOYSA-N sodium ethoxide Chemical compound [Na+].CC[O-] QDRKDTQENPPHOJ-UHFFFAOYSA-N 0.000 description 1
- 239000012312 sodium hydride Substances 0.000 description 1
- 229910000104 sodium hydride Inorganic materials 0.000 description 1
- 239000004317 sodium nitrate Substances 0.000 description 1
- 235000010344 sodium nitrate Nutrition 0.000 description 1
- RYYKJJJTJZKILX-UHFFFAOYSA-M sodium octadecanoate Chemical compound [Na+].CCCCCCCCCCCCCCCCCC([O-])=O RYYKJJJTJZKILX-UHFFFAOYSA-M 0.000 description 1
- 239000000454 talc Substances 0.000 description 1
- 229910052623 talc Inorganic materials 0.000 description 1
- 235000007586 terpenes Nutrition 0.000 description 1
- OBSZRRSYVTXPNB-UHFFFAOYSA-N tetraphosphorus Chemical compound P12P3P1P32 OBSZRRSYVTXPNB-UHFFFAOYSA-N 0.000 description 1
- 238000005979 thermal decomposition reaction Methods 0.000 description 1
- 239000010409 thin film Substances 0.000 description 1
- OGIDPMRJRNCKJF-UHFFFAOYSA-N titanium oxide Inorganic materials [Ti]=O OGIDPMRJRNCKJF-UHFFFAOYSA-N 0.000 description 1
- DVKJHBMWWAPEIU-UHFFFAOYSA-N toluene 2,4-diisocyanate Chemical compound CC1=CC=C(N=C=O)C=C1N=C=O DVKJHBMWWAPEIU-UHFFFAOYSA-N 0.000 description 1
- 239000008096 xylene Substances 0.000 description 1
- 150000003739 xylenols Chemical class 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G59/00—Polycondensates containing more than one epoxy group per molecule; Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups
- C08G59/18—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing
- C08G59/20—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing characterised by the epoxy compounds used
- C08G59/22—Di-epoxy compounds
- C08G59/30—Di-epoxy compounds containing atoms other than carbon, hydrogen, oxygen and nitrogen
- C08G59/304—Di-epoxy compounds containing atoms other than carbon, hydrogen, oxygen and nitrogen containing phosphorus
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D303/00—Compounds containing three-membered rings having one oxygen atom as the only ring hetero atom
- C07D303/02—Compounds containing oxirane rings
- C07D303/12—Compounds containing oxirane rings with hydrocarbon radicals, substituted by singly or doubly bound oxygen atoms
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G59/00—Polycondensates containing more than one epoxy group per molecule; Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups
- C08G59/18—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing
- C08G59/40—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing characterised by the curing agents used
- C08G59/4007—Curing agents not provided for by the groups C08G59/42 - C08G59/66
- C08G59/4014—Nitrogen containing compounds
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G59/00—Polycondensates containing more than one epoxy group per molecule; Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups
- C08G59/18—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing
- C08G59/40—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing characterised by the curing agents used
- C08G59/42—Polycarboxylic acids; Anhydrides, halides or low molecular weight esters thereof
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Epoxy Resins (AREA)
Abstract
Description
| 구분 | 인 함량(wt%) |
| 실시예 1 | 7.3 |
| 실시예 2 | 7.3 |
| 실시예 3 | 7.3 |
| 실시예 4 | 6.57 |
| 실시예 5 | 5.04 |
| 실시예 6 | 5.10 |
| 실시예 7 | 5.29 |
| 실시예 8 | 5.00 |
| 비교예 1 | 3.0 |
| 비교예 2 | 2.26 |
| 구분 | Tg() | Td(℃) (-5% Loss) |
Dk[1GHz] | Df[1GHz] |
| 실시예 9 | 175.8 | 401.7 | 3.277 | 0.0129 |
| 실시예 10 | 180.0 | 404.5 | 3.240 | 0.0126 |
| 실시예 11 | 172.8 | 397.8 | 3.200 | 0.0150 |
| 실시예 12 | 171.8 | 401.5 | 3.150 | 0.0120 |
| 비교예 3 | 129.0 | 378.9 | 3.330 | 0.0250 |
Claims (20)
- 인계 에폭시 화합물 및 경화제를 포함하는 인계 에폭시 조성물로서,
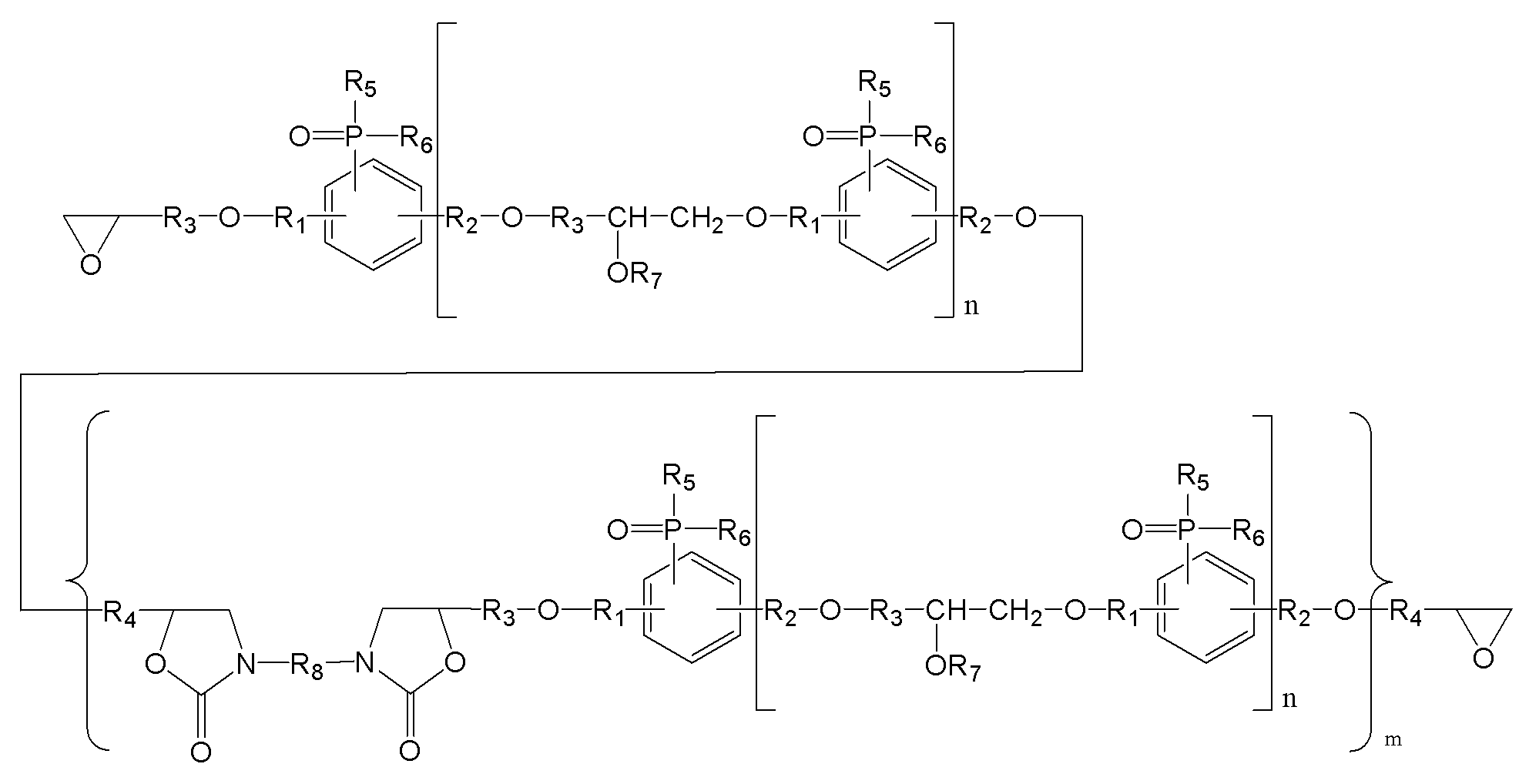
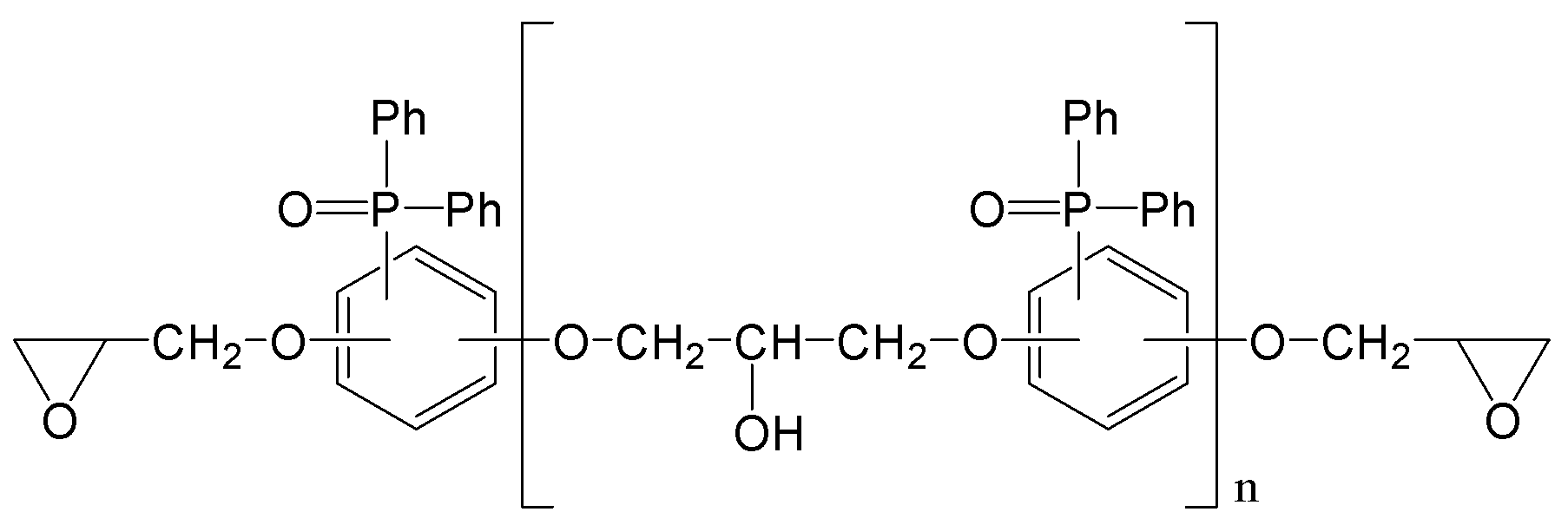
상기 인계 에폭시 화합물은 하기 화학식 1로 표현되는 인계 에폭시 화합물인 인계 에폭시 조성물.
[화학식 1]
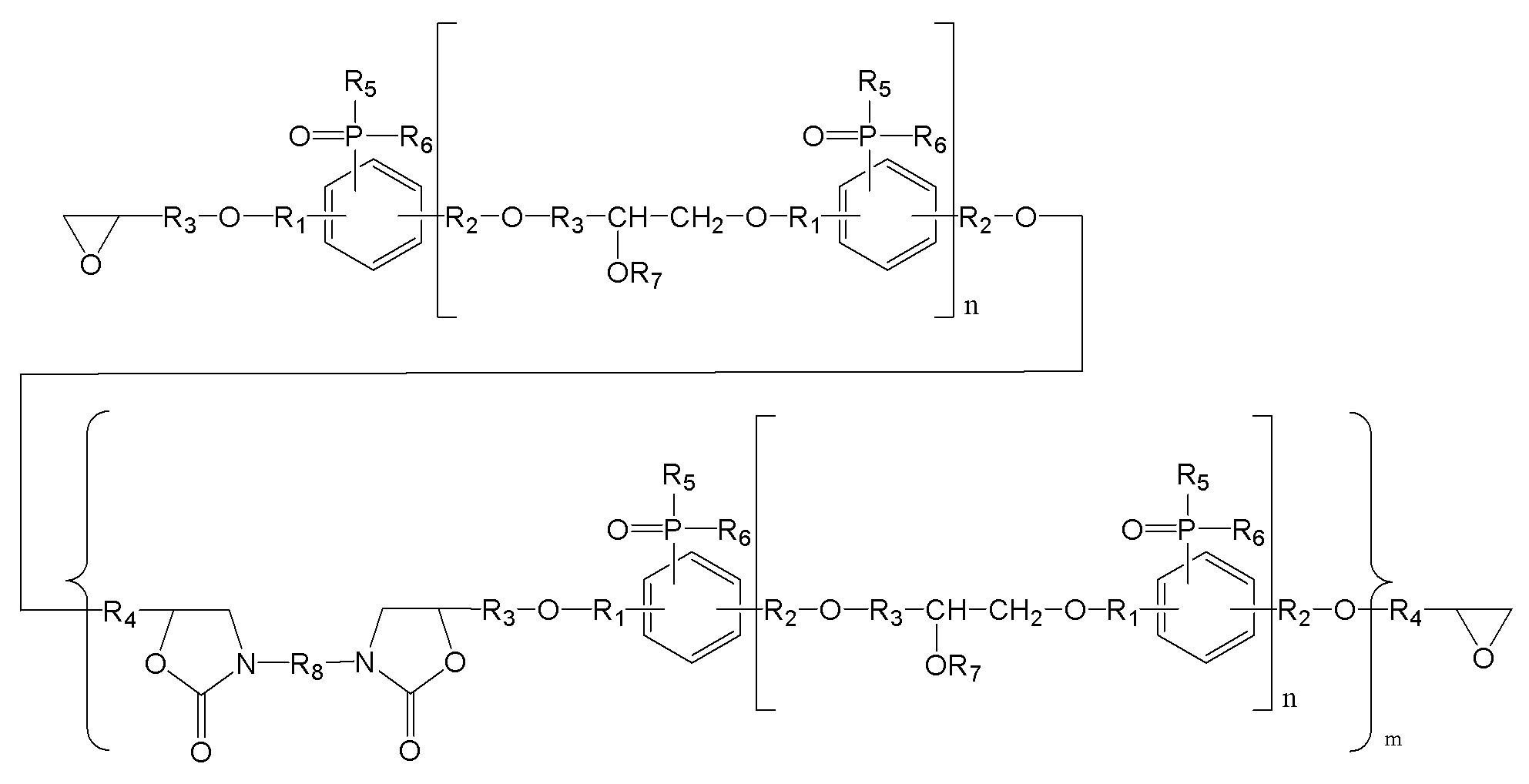
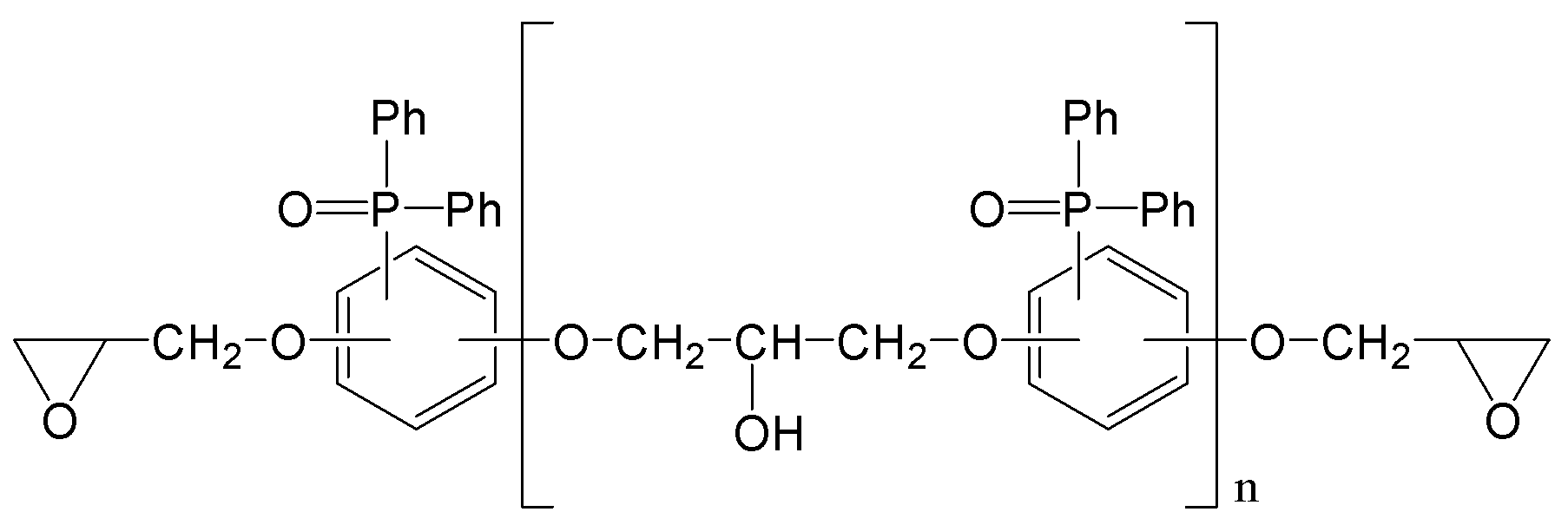
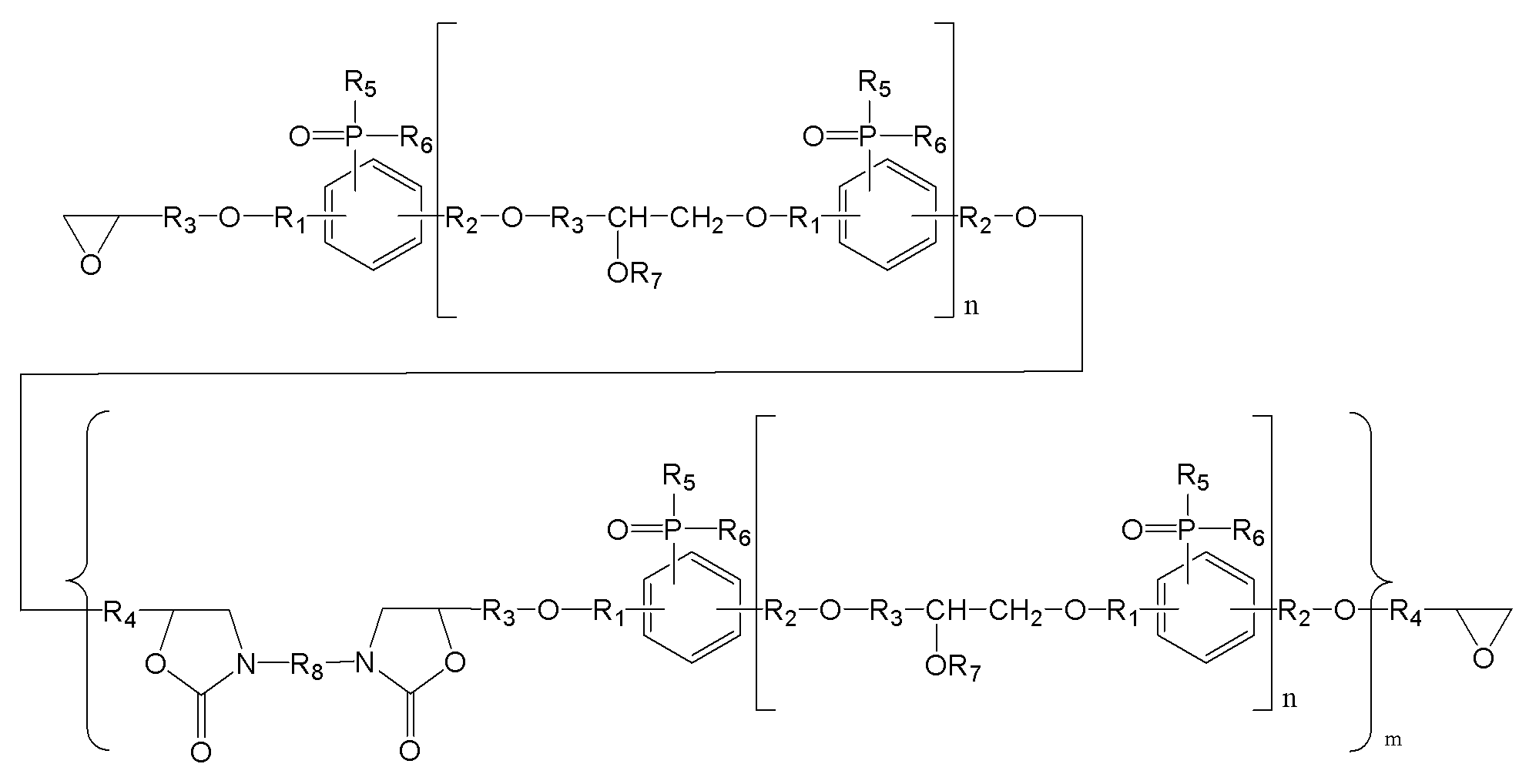
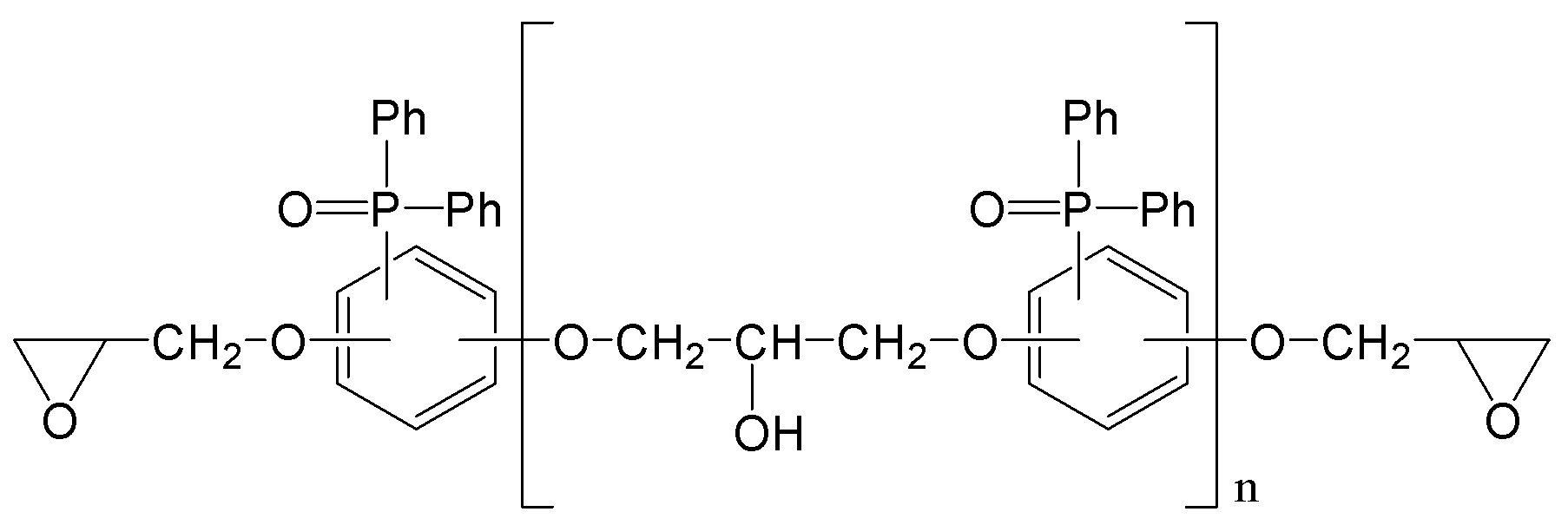
(m은 0 내지 3이며, n은 0 내지 5이며,
R1, R2는 각각 독립적으로, 존재하지 않거나 C1-C20의 지방족 탄화수소기 또는 C6-C30의 방향족 탄화수소기이거나 이들의 조합이며, 구조 내에 헤테로 원소(O, S, 또는 N)가 존재할 수 있으며,
R3, R4는 각각 독립적으로, C1-C20의 지방족 탄화수소기 또는 C6-C30의 방향족 탄화수소기이거나 이들의 조합이며, 구조 내에 헤테로 원소(O, S, 또는 N)가 존재할 수 있으며,
R5, R6는 각각 독립적으로, C1-C20의 지방족 탄화수소기 또는 C6-C30의 방향족 탄화수소기이거나 이들의 조합이며, 구조 내에 헤테로 원소(O, S, 또는 N)가 존재할 수 있으며,
R7는 각각 독립적으로 수소, C1-C20의 지방족 탄화수소기 또는 C6-C30의 방향족 탄화수소기이거나 이들의 조합이며, 구조 내에 헤테로 원소(O, S, 또는 N)가 존재할 수 있으며,
R8은 각각 독립적으로 C1-C20의 지방족 탄화수소기 또는 C6-C30의 방향족 탄화수소기 또는 이들의 조합이거나, 중량평균분자량 500 내지 5000의 고분자(또는 프리폴리머)이며, 구조 내에 헤테로 원소(O, S, 또는 N)가 존재할 수 있다.) - 제1항에 있어서,
상기 인계 에폭시 화합물 중 상기 n이 0인 인계 에폭시 화합물의 함량이 30 내지 99wt%인 인계 에폭시 조성물. - 제1항에 있어서,
상기 인계 에폭시 화합물과 상기 경화제의 당량비는 1 : 0.6 내지 1.5로 포함되는 인계 에폭시 조성물. - 제1항에 있어서,
상기 경화제는 아마이드계 경화제, 폴리아민계 경화제, 산무수물 경화제, 페놀노볼락형 경화제, 폴리메르캅탄 경화제, 제3아민 경화제 또는 이미다졸 경화제로 이루어진 군으로부터 적어도 하나 이상 선택되는 인계 에폭시 조성물. - 제1항에 있어서,
상기 인계 에폭시 조성물은 경화촉진제를 더 포함하며,
상기 경화촉진제는 2-메틸 이미다졸(2-methyl Imidazole), 2-에틸-4-메틸 이미다졸(2-Ethyl-4-Methyl Imidazole), 1-벤질-2-메틸 이미다졸(1-Benzyl-2-Methyl Imidazole), 2-헵타데실 이미다졸(2-Heptadecyl Imidazole) 및 2-운데실 이미다졸(2-Undecyl Imidazole) 중에서 선택된 1종 이상을 함유한 이미다졸 화합물;
트리페닐포스페이트(Triphenylphosphate)를 함유한 유기화합물; 및
ETPPI(Ethyl Triphenyl Phosphonium Iodide) 등의 트리 알킬 화합물, 4-디메틸아미노피리딘(4-Dimethylamino Pyridine), 2-아미노 피리딘, 3-아미노 피리딘, 4-아미노 피리딘, 2,3-디아미노 피리딘, 2,5-디아미노 피리딘, 2,6-디아미노 피리딘, 2-아미노-6-메틸 피리딘, 3-아미노-6-이소프로필 피리딘, 2,2- 디피리딜 아민 및 4-피롤리디노 피리딘 중에서 선택된 1종 이상을 함유한 피리딘 화합물; 중에서 선택된 1종 이상을 포함하는 인계 에폭시 조성물. - 제1항에 있어서,
상기 인계 에폭시 조성물의 전체 중량 대비 인(P) 함량은 2 내지 7 중량%인 인계 에폭시 조성물. - 제1항에 있어서,
상기 인계 에폭시 조성물로부터 얻어지는 경화물의 유리 전이 온도(Tg)가 150 내지 230℃ 인 것을 특징으로 하는 인계 에폭시 조성물. - 제1항에 있어서,
상기 인계 에폭시 조성물로부터 얻어지는 경화물의 유전상수(Dk)은 4.2 이하인 것을 특징으로 하는 인계 에폭시 조성물. - 제1항에 있어서,
상기 인계 에폭시 조성물로부터 얻어지는 경화물의 손실인자(Df)는 0.020 이하인 것을 특징으로 하는 인계 에폭시 조성물. - 하기 화학식 1로 표시되는 인계 에폭시 화합물.
[화학식 1]
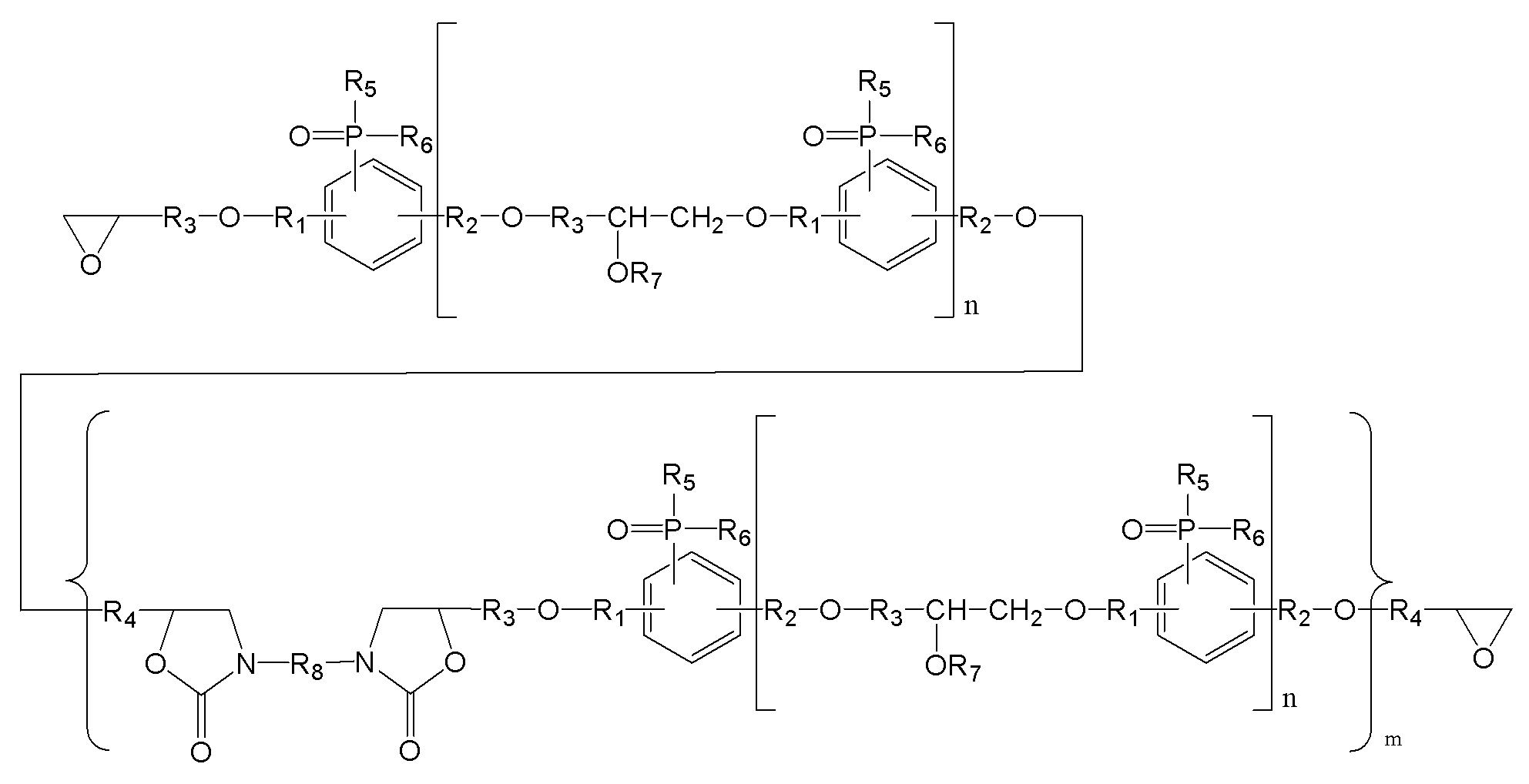
(m은 0 내지 3이며, n은 0 내지 5이며,
R1, R2는 각각 독립적으로, 존재하지 않거나 C1-C20의 지방족 탄화수소기 또는 C6-C30의 방향족 탄화수소기이거나 이들의 조합이며, 구조 내에 헤테로 원소(O, S, 또는 N)가 존재할 수 있으며,
R3, R4는 각각 독립적으로, C1-C20의 지방족 탄화수소기 또는 C6-C30의 방향족 탄화수소기이거나 이들의 조합이며, 구조 내에 헤테로 원소(O, S, 또는 N)가 존재할 수 있으며,
R5, R6는 각각 독립적으로, C1-C20의 지방족 탄화수소기 또는 C6-C30의 방향족 탄화수소기이거나 이들의 조합이며, 구조 내에 헤테로 원소(O, S, 또는 N)가 존재할 수 있으며,
R7는 각각 독립적으로 수소, C1-C20의 지방족 탄화수소기 또는 C6-C30의 방향족 탄화수소기이거나 이들의 조합이며, 구조 내에 헤테로 원소(O, S, 또는 N)가 존재할 수 있으며,
R8은 각각 독립적으로 C1-C20의 지방족 탄화수소기 또는 C6-C30의 방향족 탄화수소기 또는 이들의 조합이거나, 중량평균분자량 500 내지 5000의 고분자(또는 프리폴리머)이며, 구조 내에 헤테로 원소(O, S, 또는 N)가 존재할 수 있다.) - 제12항에 있어서,
상기 인계 에폭시 화합물의 전체 중량 대비 인(P) 함량은 6 내지 8 중량%인 인계 에폭시 화합물. - 제12항에 있어서,
상기 인계 에폭시 화합물은 인계 히드록시 화합물 및 에폭시 화합물을 알칼리 촉매 하에서 반응시켜 제조되는 것을 특징으로 하는 인계 에폭시 화합물. - 하기 화학식 3으로 표시되는 인계 히드록시 화합물, 하기 화학식 4-1과 하기 화학식 4-2로 표시되는 에폭시 화합물 중 선택되는 적어도 하나 이상의 에폭시 화합물, 및 제1 용매를 포함하는 반응 물질들을 반응기에 투입하여 반응 혼합물을 얻는 반응 혼합물 준비단계; 및
상기 반응 혼합물을 반응시켜 인이 함유된 에폭시 화합물을 얻는 단계;를 포함하는 인계 에폭시 화합물 제조방법.
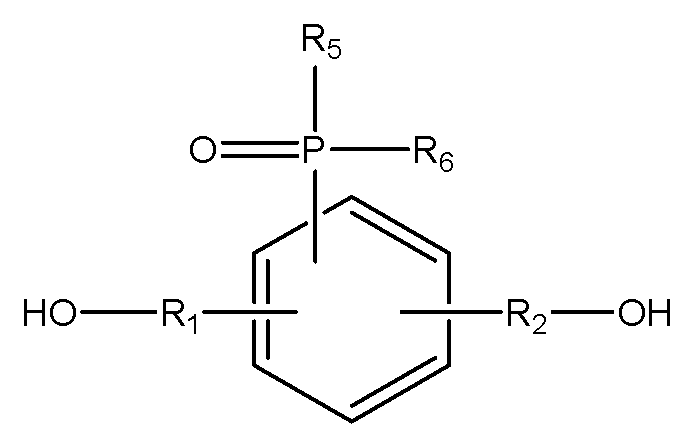
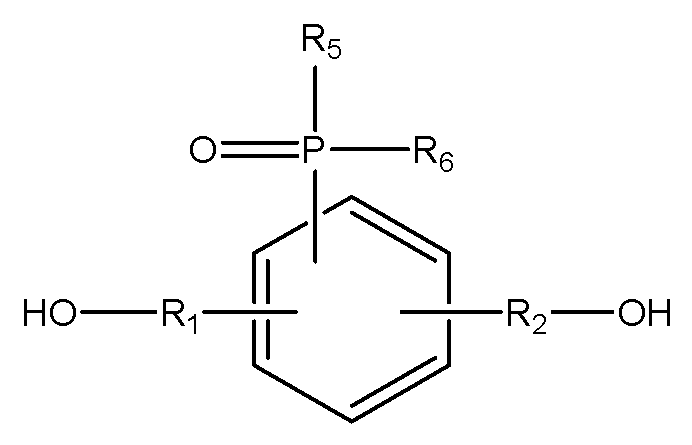
[화학식 3]
[화학식 4-1]
[화학식 4-2]
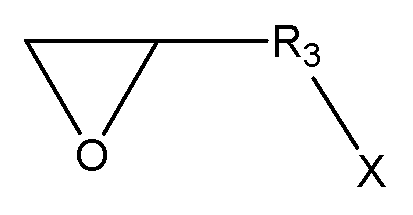
(R1, R2는 각각 독립적으로, 존재하지 않거나 C1-C20의 지방족 탄화수소기 또는 C6-C30의 방향족 탄화수소기이거나 이들의 조합이며, 구조 내에 헤테로 원소(O, S, 또는 N)가 존재할 수 있으며,
R3, R4는 각각 독립적으로, C1-C20의 지방족 탄화수소기 또는 C6-C30의 방향족 탄화수소기이거나 이들의 조합이며, 구조 내에 헤테로 원소(O, S, 또는 N)가 존재할 수 있으며,
R5, R6는 각각 독립적으로, C1-C20의 지방족 탄화수소기 또는 C6-C30의 방향족 탄화수소기이거나 이들의 조합이며, 구조 내에 헤테로 원소(O, S, 또는 N)가 존재할 수 있으며,
X는 할로겐기, -OTs(O-Tosyl), -OMs(O-Mesyl)이다.) - 제17항에 있어서,
상기 반응 혼합물 준비단계는,
상기 제1 용매 100 중량부에 대하여, 상기 화학식 3으로 표시되는 인계 히드록시 화합물이 100 내지 180 중량부, 상기 화학식 4-1과 상기 화학식 4-2로 표시되는 에폭시 화합물이 210 내지 290 중량부로 혼합되는 것을 특징으로 하는 인계 에폭시 화합물 제조방법. - 제17항에 있어서,
상기 반응 혼합물을 반응시켜 인이 함유된 에폭시 화합물을 얻는 단계 이후에, 상기 얻어진 에폭시 화합물의 전부 또는 일부를 다관능 이소시아네이트 화합물로 변성시키는 단계를 더 포함하는 것을 특징으로 하는 인계 에폭시 화합물 제조방법. - 제1항의 인계 에폭시 조성물을 포함하는 것을 특징으로 하는 적층판.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020160022168A KR102587599B1 (ko) | 2016-02-24 | 2016-02-24 | 인계 에폭시 화합물 및 이의 제조방법, 이를 포함하는 에폭시 조성물 |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020160022168A KR102587599B1 (ko) | 2016-02-24 | 2016-02-24 | 인계 에폭시 화합물 및 이의 제조방법, 이를 포함하는 에폭시 조성물 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| KR20170100111A true KR20170100111A (ko) | 2017-09-04 |
| KR102587599B1 KR102587599B1 (ko) | 2023-10-12 |
Family
ID=59924421
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020160022168A Active KR102587599B1 (ko) | 2016-02-24 | 2016-02-24 | 인계 에폭시 화합물 및 이의 제조방법, 이를 포함하는 에폭시 조성물 |
Country Status (1)
| Country | Link |
|---|---|
| KR (1) | KR102587599B1 (ko) |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2025116899A1 (en) * | 2023-11-29 | 2025-06-05 | Uniseal, Inc. | Storage stable, one-component, solvent-free, non-halogenated, heat curable compositions for use in encapsulation of electrical components |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5214070A (en) * | 1975-07-22 | 1977-02-02 | Asou Cement Kk | Method of treatment of alkali effluent by combustion waste gas |
| JPH0653785B2 (ja) * | 1991-08-02 | 1994-07-20 | 北興化学工業株式会社 | リン含有エポキシ樹脂およびその製造方法 |
| KR20050010847A (ko) * | 2002-05-30 | 2005-01-28 | 다우 글로벌 테크놀로지스 인크. | 할로겐 비함유 내점화성 열가소성 수지 조성물 |
| JP2009035586A (ja) * | 2007-07-31 | 2009-02-19 | Dic Corp | エポキシ樹脂組成物、その硬化物、プリント回路基板用ワニス、新規エポキシ樹脂及びその製造方法 |
| KR101341971B1 (ko) * | 2013-08-09 | 2013-12-16 | 주식회사 신아티앤씨 | 전기적 특성이 우수한 폴리 에폭시 수지, 이의 조성물 및 제조방법 |
-
2016
- 2016-02-24 KR KR1020160022168A patent/KR102587599B1/ko active Active
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5214070A (en) * | 1975-07-22 | 1977-02-02 | Asou Cement Kk | Method of treatment of alkali effluent by combustion waste gas |
| JPH0653785B2 (ja) * | 1991-08-02 | 1994-07-20 | 北興化学工業株式会社 | リン含有エポキシ樹脂およびその製造方法 |
| KR20050010847A (ko) * | 2002-05-30 | 2005-01-28 | 다우 글로벌 테크놀로지스 인크. | 할로겐 비함유 내점화성 열가소성 수지 조성물 |
| JP2009035586A (ja) * | 2007-07-31 | 2009-02-19 | Dic Corp | エポキシ樹脂組成物、その硬化物、プリント回路基板用ワニス、新規エポキシ樹脂及びその製造方法 |
| KR101341971B1 (ko) * | 2013-08-09 | 2013-12-16 | 주식회사 신아티앤씨 | 전기적 특성이 우수한 폴리 에폭시 수지, 이의 조성물 및 제조방법 |
Non-Patent Citations (1)
| Title |
|---|
| Journal of Polymer Science. Part A. Polymer Chemistry. 2007. Vol. 45. pp2142-2151 * |
Also Published As
| Publication number | Publication date |
|---|---|
| KR102587599B1 (ko) | 2023-10-12 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US9567481B2 (en) | Resin composition, resin varnish, prepreg, metal-clad laminate and printed wiring board | |
| KR101144566B1 (ko) | 에폭시 수지 조성물, 그 에폭시 수지 조성물을 이용한 프리프레그, 금속 클래드 적층판, 및 프린트 배선판 | |
| JP5264133B2 (ja) | エポキシ樹脂組成物、そのエポキシ樹脂組成物を用いたプリプレグ及び金属張積層板 | |
| KR101141305B1 (ko) | 인-변성 페놀 노볼락 수지, 이를 포함하는 경화제 및 에폭시 수지 조성물 | |
| KR101813527B1 (ko) | 인계 에폭시 화합물 및 이의 제조방법, 이를 포함하는 에폭시 조성물 | |
| JP6946578B2 (ja) | 樹脂組成物およびその製造方法 | |
| KR101596992B1 (ko) | 비할로겐 난연성 중합체 및 이를 함유하는 난연성 중합체 조성물 | |
| JP5752574B2 (ja) | フェノールノボラック樹脂及びそれを用いたエポキシ樹脂組成物 | |
| JP2017141389A (ja) | オキサジン樹脂組成物及びその硬化物 | |
| JP2011084697A (ja) | 重合性リン含有(ポリ)キシリレンアリールエーテル化合物、その製造方法、難燃性熱硬化性樹脂組成物、硬化物及び積層板 | |
| KR20020026408A (ko) | 에폭시 수지 조성물, 프리프레그 및 복합금속 적층판 | |
| CN104163903A (zh) | 含磷的酚树脂、其制造方法及其用途 | |
| JP7061944B2 (ja) | ワニスおよびその製造方法 | |
| KR102587599B1 (ko) | 인계 에폭시 화합물 및 이의 제조방법, 이를 포함하는 에폭시 조성물 | |
| JP6420616B2 (ja) | フェノール樹脂、その製造方法並びに該フェノール樹脂を含有するエポキシ樹脂組成物及びその硬化物 | |
| JP5793640B2 (ja) | プリント配線板用エポキシ樹脂組成物、そのプリント配線板用エポキシ樹脂組成物を用いたプリント配線板用プリプレグ及びプリント配線板用金属張積層板 | |
| KR20170080401A (ko) | 비할로겐 난연성 중합체, 이의 제조방법 및 이를 함유하는 난연성 중합체 조성물 | |
| EP2186816B1 (en) | Phosphorus compound and method for preparing the same | |
| KR101598244B1 (ko) | 비할로겐 난연성 중합체 및 이를 함유하는 난연성 중합체 조성물 | |
| JP5919576B2 (ja) | プリント配線板用エポキシ樹脂組成物、そのプリント配線板用エポキシ樹脂組成物を用いたプリント配線板用プリプレグ及びプリント配線板用金属張積層板 | |
| CN112409572A (zh) | 含磷阻燃低热膨胀系数环氧树脂及其制备方法和所涉及的中间产物以及应用 | |
| EP2374828B1 (en) | Preparation of a low dielectric brominated resin with a symmetric or saturated heterocyclic aliphatic molecular structure and composition comprising said resin | |
| KR101522697B1 (ko) | 활성 에스터 인 변성 경화제 조성물, 활성 에스터 인 변성 경화제 및 이의 제조방법 | |
| KR20140006140A (ko) | 비할로겐 난연성 중합체 및 이를 함유하는 난연성 중합체 조성물 | |
| KR20180009556A (ko) | 난연성 및 전기적 특성이 우수한 난연 화합물 및 이의 조성물 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0109 | Patent application |
Patent event code: PA01091R01D Comment text: Patent Application Patent event date: 20160224 |
|
| PG1501 | Laying open of application | ||
| PA0201 | Request for examination |
Patent event code: PA02012R01D Patent event date: 20210223 Comment text: Request for Examination of Application Patent event code: PA02011R01I Patent event date: 20160224 Comment text: Patent Application |
|
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
Comment text: Notification of reason for refusal Patent event date: 20220721 Patent event code: PE09021S01D |
|
| E90F | Notification of reason for final refusal | ||
| PE0902 | Notice of grounds for rejection |
Comment text: Final Notice of Reason for Refusal Patent event date: 20230201 Patent event code: PE09021S02D |
|
| E701 | Decision to grant or registration of patent right | ||
| PE0701 | Decision of registration |
Patent event code: PE07011S01D Comment text: Decision to Grant Registration Patent event date: 20230731 |
|
| GRNT | Written decision to grant | ||
| PR0701 | Registration of establishment |
Comment text: Registration of Establishment Patent event date: 20231005 Patent event code: PR07011E01D |
|
| PR1002 | Payment of registration fee |
Payment date: 20231005 End annual number: 3 Start annual number: 1 |
|
| PG1601 | Publication of registration |