KR20200067009A - 이차전지용 양극 활물질, 이의 제조 방법, 이를 포함하는 이차전지용 양극 및 리튬 이차전지 - Google Patents
이차전지용 양극 활물질, 이의 제조 방법, 이를 포함하는 이차전지용 양극 및 리튬 이차전지 Download PDFInfo
- Publication number
- KR20200067009A KR20200067009A KR1020180153838A KR20180153838A KR20200067009A KR 20200067009 A KR20200067009 A KR 20200067009A KR 1020180153838 A KR1020180153838 A KR 1020180153838A KR 20180153838 A KR20180153838 A KR 20180153838A KR 20200067009 A KR20200067009 A KR 20200067009A
- Authority
- KR
- South Korea
- Prior art keywords
- positive electrode
- secondary battery
- active material
- electrode active
- transition metal
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 239000007774 positive electrode material Substances 0.000 title claims abstract description 154
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 title claims abstract description 138
- 229910052744 lithium Inorganic materials 0.000 title claims abstract description 138
- 238000004519 manufacturing process Methods 0.000 title claims description 24
- 238000000034 method Methods 0.000 title claims description 24
- 239000002245 particle Substances 0.000 claims abstract description 177
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 claims abstract description 115
- 239000002131 composite material Substances 0.000 claims abstract description 75
- 229910000314 transition metal oxide Inorganic materials 0.000 claims abstract description 74
- 239000011572 manganese Substances 0.000 claims abstract description 42
- 229910052759 nickel Inorganic materials 0.000 claims abstract description 36
- 229910052782 aluminium Inorganic materials 0.000 claims abstract description 27
- 229910052751 metal Inorganic materials 0.000 claims abstract description 27
- 239000002184 metal Substances 0.000 claims abstract description 27
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 claims abstract description 16
- 229910052748 manganese Inorganic materials 0.000 claims abstract description 16
- PWHULOQIROXLJO-UHFFFAOYSA-N Manganese Chemical compound [Mn] PWHULOQIROXLJO-UHFFFAOYSA-N 0.000 claims abstract description 12
- 229910017052 cobalt Inorganic materials 0.000 claims abstract description 12
- 239000010941 cobalt Substances 0.000 claims abstract description 12
- GUTLYIVDDKVIGB-UHFFFAOYSA-N cobalt atom Chemical compound [Co] GUTLYIVDDKVIGB-UHFFFAOYSA-N 0.000 claims abstract description 12
- 229910052723 transition metal Inorganic materials 0.000 claims description 26
- 150000003624 transition metals Chemical class 0.000 claims description 26
- 238000010304 firing Methods 0.000 claims description 25
- 150000002739 metals Chemical class 0.000 claims description 21
- 239000003792 electrolyte Substances 0.000 claims description 15
- 229910052719 titanium Inorganic materials 0.000 claims description 13
- 150000001875 compounds Chemical class 0.000 claims description 11
- 229910052726 zirconium Inorganic materials 0.000 claims description 11
- 150000002642 lithium compounds Chemical class 0.000 claims description 10
- 229910052721 tungsten Inorganic materials 0.000 claims description 10
- 229910052796 boron Inorganic materials 0.000 claims description 9
- 229910052710 silicon Inorganic materials 0.000 claims description 8
- 238000002156 mixing Methods 0.000 claims description 7
- 229910052698 phosphorus Inorganic materials 0.000 claims description 7
- 239000011149 active material Substances 0.000 claims description 4
- 230000007423 decrease Effects 0.000 claims description 3
- 230000000052 comparative effect Effects 0.000 description 43
- WMFOQBRAJBCJND-UHFFFAOYSA-M Lithium hydroxide Chemical compound [Li+].[OH-] WMFOQBRAJBCJND-UHFFFAOYSA-M 0.000 description 29
- 239000000203 mixture Substances 0.000 description 17
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 16
- -1 etc.) Chemical compound 0.000 description 16
- 239000010410 layer Substances 0.000 description 12
- 239000011230 binding agent Substances 0.000 description 11
- 239000004020 conductor Substances 0.000 description 11
- 238000002360 preparation method Methods 0.000 description 11
- 238000005336 cracking Methods 0.000 description 10
- 239000010936 titanium Substances 0.000 description 10
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 9
- 229910052799 carbon Inorganic materials 0.000 description 9
- 238000009826 distribution Methods 0.000 description 9
- 230000000694 effects Effects 0.000 description 9
- 239000007773 negative electrode material Substances 0.000 description 9
- 238000005096 rolling process Methods 0.000 description 9
- JBTWLSYIZRCDFO-UHFFFAOYSA-N ethyl methyl carbonate Chemical compound CCOC(=O)OC JBTWLSYIZRCDFO-UHFFFAOYSA-N 0.000 description 8
- 239000000126 substance Substances 0.000 description 8
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 7
- 230000014759 maintenance of location Effects 0.000 description 7
- 229910052760 oxygen Inorganic materials 0.000 description 7
- 239000001301 oxygen Substances 0.000 description 7
- 239000006182 cathode active material Substances 0.000 description 6
- KMTRUDSVKNLOMY-UHFFFAOYSA-N Ethylene carbonate Chemical compound O=C1OCCO1 KMTRUDSVKNLOMY-UHFFFAOYSA-N 0.000 description 5
- HBBGRARXTFLTSG-UHFFFAOYSA-N Lithium ion Chemical compound [Li+] HBBGRARXTFLTSG-UHFFFAOYSA-N 0.000 description 5
- 229920001577 copolymer Polymers 0.000 description 5
- 229910001416 lithium ion Inorganic materials 0.000 description 5
- 229910003002 lithium salt Inorganic materials 0.000 description 5
- 159000000002 lithium salts Chemical class 0.000 description 5
- 239000002904 solvent Substances 0.000 description 5
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 4
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 4
- VGGSQFUCUMXWEO-UHFFFAOYSA-N Ethene Chemical compound C=C VGGSQFUCUMXWEO-UHFFFAOYSA-N 0.000 description 4
- 239000005977 Ethylene Substances 0.000 description 4
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 4
- 229910021383 artificial graphite Inorganic materials 0.000 description 4
- 239000003575 carbonaceous material Substances 0.000 description 4
- 229910052802 copper Inorganic materials 0.000 description 4
- 239000010949 copper Substances 0.000 description 4
- 238000002149 energy-dispersive X-ray emission spectroscopy Methods 0.000 description 4
- 239000010408 film Substances 0.000 description 4
- 230000006872 improvement Effects 0.000 description 4
- 229910021382 natural graphite Inorganic materials 0.000 description 4
- 239000004745 nonwoven fabric Substances 0.000 description 4
- 239000003960 organic solvent Substances 0.000 description 4
- 229910001220 stainless steel Inorganic materials 0.000 description 4
- 239000010935 stainless steel Substances 0.000 description 4
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 3
- BVKZGUZCCUSVTD-UHFFFAOYSA-L Carbonate Chemical compound [O-]C([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-L 0.000 description 3
- OIFBSDVPJOWBCH-UHFFFAOYSA-N Diethyl carbonate Chemical compound CCOC(=O)OCC OIFBSDVPJOWBCH-UHFFFAOYSA-N 0.000 description 3
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 3
- SECXISVLQFMRJM-UHFFFAOYSA-N N-Methylpyrrolidone Chemical compound CN1CCCC1=O SECXISVLQFMRJM-UHFFFAOYSA-N 0.000 description 3
- 239000011248 coating agent Substances 0.000 description 3
- 238000000576 coating method Methods 0.000 description 3
- IEJIGPNLZYLLBP-UHFFFAOYSA-N dimethyl carbonate Chemical compound COC(=O)OC IEJIGPNLZYLLBP-UHFFFAOYSA-N 0.000 description 3
- 229910002804 graphite Inorganic materials 0.000 description 3
- 239000010439 graphite Substances 0.000 description 3
- 238000003780 insertion Methods 0.000 description 3
- 230000037431 insertion Effects 0.000 description 3
- KWGKDLIKAYFUFQ-UHFFFAOYSA-M lithium chloride Chemical compound [Li+].[Cl-] KWGKDLIKAYFUFQ-UHFFFAOYSA-M 0.000 description 3
- 239000000463 material Substances 0.000 description 3
- 229910044991 metal oxide Inorganic materials 0.000 description 3
- 150000004706 metal oxides Chemical class 0.000 description 3
- 239000005518 polymer electrolyte Substances 0.000 description 3
- 229920002981 polyvinylidene fluoride Polymers 0.000 description 3
- RUOJZAUFBMNUDX-UHFFFAOYSA-N propylene carbonate Chemical compound CC1COC(=O)O1 RUOJZAUFBMNUDX-UHFFFAOYSA-N 0.000 description 3
- 229910052709 silver Inorganic materials 0.000 description 3
- 239000004332 silver Substances 0.000 description 3
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 2
- 229920000049 Carbon (fiber) Polymers 0.000 description 2
- 229920002134 Carboxymethyl cellulose Polymers 0.000 description 2
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 2
- 229910013870 LiPF 6 Inorganic materials 0.000 description 2
- 239000002033 PVDF binder Substances 0.000 description 2
- 239000004698 Polyethylene Substances 0.000 description 2
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 2
- KAESVJOAVNADME-UHFFFAOYSA-N Pyrrole Chemical compound C=1C=CNC=1 KAESVJOAVNADME-UHFFFAOYSA-N 0.000 description 2
- BQCADISMDOOEFD-UHFFFAOYSA-N Silver Chemical compound [Ag] BQCADISMDOOEFD-UHFFFAOYSA-N 0.000 description 2
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 2
- XLOMVQKBTHCTTD-UHFFFAOYSA-N Zinc monoxide Chemical compound [Zn]=O XLOMVQKBTHCTTD-UHFFFAOYSA-N 0.000 description 2
- 239000000654 additive Substances 0.000 description 2
- 239000004917 carbon fiber Substances 0.000 description 2
- 239000003660 carbonate based solvent Substances 0.000 description 2
- 230000008859 change Effects 0.000 description 2
- 238000007600 charging Methods 0.000 description 2
- 239000013078 crystal Substances 0.000 description 2
- 150000005676 cyclic carbonates Chemical class 0.000 description 2
- JHIVVAPYMSGYDF-UHFFFAOYSA-N cyclohexanone Chemical compound O=C1CCCCC1 JHIVVAPYMSGYDF-UHFFFAOYSA-N 0.000 description 2
- 238000009831 deintercalation Methods 0.000 description 2
- 238000003795 desorption Methods 0.000 description 2
- 238000007599 discharging Methods 0.000 description 2
- 238000001035 drying Methods 0.000 description 2
- 239000011267 electrode slurry Substances 0.000 description 2
- LYCAIKOWRPUZTN-UHFFFAOYSA-N ethylene glycol Natural products OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 2
- 239000000835 fiber Substances 0.000 description 2
- 239000006260 foam Substances 0.000 description 2
- 239000011888 foil Substances 0.000 description 2
- 230000014509 gene expression Effects 0.000 description 2
- 238000009830 intercalation Methods 0.000 description 2
- 230000002687 intercalation Effects 0.000 description 2
- 238000010030 laminating Methods 0.000 description 2
- 238000007561 laser diffraction method Methods 0.000 description 2
- 239000011244 liquid electrolyte Substances 0.000 description 2
- IIPYXGDZVMZOAP-UHFFFAOYSA-N lithium nitrate Chemical compound [Li+].[O-][N+]([O-])=O IIPYXGDZVMZOAP-UHFFFAOYSA-N 0.000 description 2
- 238000005259 measurement Methods 0.000 description 2
- 150000002736 metal compounds Chemical class 0.000 description 2
- 229920000573 polyethylene Polymers 0.000 description 2
- 229920000642 polymer Polymers 0.000 description 2
- 239000002243 precursor Substances 0.000 description 2
- 238000006722 reduction reaction Methods 0.000 description 2
- 238000007789 sealing Methods 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- 229920003048 styrene butadiene rubber Polymers 0.000 description 2
- 230000007704 transition Effects 0.000 description 2
- WNXJIVFYUVYPPR-UHFFFAOYSA-N 1,3-dioxolane Chemical compound C1COCO1 WNXJIVFYUVYPPR-UHFFFAOYSA-N 0.000 description 1
- DURPTKYDGMDSBL-UHFFFAOYSA-N 1-butoxybutane Chemical compound CCCCOCCCC DURPTKYDGMDSBL-UHFFFAOYSA-N 0.000 description 1
- XNWFRZJHXBZDAG-UHFFFAOYSA-N 2-METHOXYETHANOL Chemical compound COCCO XNWFRZJHXBZDAG-UHFFFAOYSA-N 0.000 description 1
- DSMUTQTWFHVVGQ-UHFFFAOYSA-N 4,5-difluoro-1,3-dioxolan-2-one Chemical compound FC1OC(=O)OC1F DSMUTQTWFHVVGQ-UHFFFAOYSA-N 0.000 description 1
- 229910000838 Al alloy Inorganic materials 0.000 description 1
- 229910000925 Cd alloy Inorganic materials 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 1
- PIICEJLVQHRZGT-UHFFFAOYSA-N Ethylenediamine Chemical compound NCCN PIICEJLVQHRZGT-UHFFFAOYSA-N 0.000 description 1
- YCKRFDGAMUMZLT-UHFFFAOYSA-N Fluorine atom Chemical compound [F] YCKRFDGAMUMZLT-UHFFFAOYSA-N 0.000 description 1
- 229920002153 Hydroxypropyl cellulose Polymers 0.000 description 1
- 229910010238 LiAlCl 4 Inorganic materials 0.000 description 1
- 229910015015 LiAsF 6 Inorganic materials 0.000 description 1
- 229910013063 LiBF 4 Inorganic materials 0.000 description 1
- 229910013372 LiC 4 Inorganic materials 0.000 description 1
- 229910013684 LiClO 4 Inorganic materials 0.000 description 1
- 229910012851 LiCoO 2 Inorganic materials 0.000 description 1
- 229910015643 LiMn 2 O 4 Inorganic materials 0.000 description 1
- 229910014689 LiMnO Inorganic materials 0.000 description 1
- 229910013553 LiNO Inorganic materials 0.000 description 1
- 229910012513 LiSbF 6 Inorganic materials 0.000 description 1
- MKYBYDHXWVHEJW-UHFFFAOYSA-N N-[1-oxo-1-(2,4,6,7-tetrahydrotriazolo[4,5-c]pyridin-5-yl)propan-2-yl]-2-[[3-(trifluoromethoxy)phenyl]methylamino]pyrimidine-5-carboxamide Chemical compound O=C(C(C)NC(=O)C=1C=NC(=NC=1)NCC1=CC(=CC=C1)OC(F)(F)F)N1CC2=C(CC1)NN=N2 MKYBYDHXWVHEJW-UHFFFAOYSA-N 0.000 description 1
- 229910002651 NO3 Inorganic materials 0.000 description 1
- NHNBFGGVMKEFGY-UHFFFAOYSA-N Nitrate Chemical compound [O-][N+]([O-])=O NHNBFGGVMKEFGY-UHFFFAOYSA-N 0.000 description 1
- YVNYRZGQMJTPLA-UHFFFAOYSA-N O=C1CCCCCO1.O=C1CCCCCO1 Chemical compound O=C1CCCCCO1.O=C1CCCCCO1 YVNYRZGQMJTPLA-UHFFFAOYSA-N 0.000 description 1
- PKVGKJDLLALEMP-UHFFFAOYSA-N O=C1CCCO1.O=C1CCCO1 Chemical compound O=C1CCCO1.O=C1CCCO1 PKVGKJDLLALEMP-UHFFFAOYSA-N 0.000 description 1
- 229920000265 Polyparaphenylene Polymers 0.000 description 1
- 239000004743 Polypropylene Substances 0.000 description 1
- 239000004372 Polyvinyl alcohol Substances 0.000 description 1
- 229910000676 Si alloy Inorganic materials 0.000 description 1
- 229910001128 Sn alloy Inorganic materials 0.000 description 1
- 229910006404 SnO 2 Inorganic materials 0.000 description 1
- 229920002472 Starch Polymers 0.000 description 1
- 239000002174 Styrene-butadiene Substances 0.000 description 1
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 1
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 1
- GSEJCLTVZPLZKY-UHFFFAOYSA-N Triethanolamine Chemical compound OCCN(CCO)CCO GSEJCLTVZPLZKY-UHFFFAOYSA-N 0.000 description 1
- RLTFLELMPUMVEH-UHFFFAOYSA-N [Li+].[O--].[O--].[O--].[V+5] Chemical compound [Li+].[O--].[O--].[O--].[V+5] RLTFLELMPUMVEH-UHFFFAOYSA-N 0.000 description 1
- XHCLAFWTIXFWPH-UHFFFAOYSA-N [O-2].[O-2].[O-2].[O-2].[O-2].[V+5].[V+5] Chemical compound [O-2].[O-2].[O-2].[O-2].[O-2].[V+5].[V+5] XHCLAFWTIXFWPH-UHFFFAOYSA-N 0.000 description 1
- 238000009825 accumulation Methods 0.000 description 1
- 239000006230 acetylene black Substances 0.000 description 1
- 230000000996 additive effect Effects 0.000 description 1
- 239000005456 alcohol based solvent Substances 0.000 description 1
- 238000005275 alloying Methods 0.000 description 1
- VSCWAEJMTAWNJL-UHFFFAOYSA-K aluminium trichloride Chemical compound Cl[Al](Cl)Cl VSCWAEJMTAWNJL-UHFFFAOYSA-K 0.000 description 1
- 150000001408 amides Chemical class 0.000 description 1
- 150000003863 ammonium salts Chemical class 0.000 description 1
- 229910003481 amorphous carbon Inorganic materials 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 150000004945 aromatic hydrocarbons Chemical class 0.000 description 1
- 125000003118 aryl group Chemical group 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- 229910052797 bismuth Inorganic materials 0.000 description 1
- HTRXGEPDTFSKLI-UHFFFAOYSA-N butanoic acid;ethyl acetate Chemical compound CCCC(O)=O.CCOC(C)=O HTRXGEPDTFSKLI-UHFFFAOYSA-N 0.000 description 1
- IAQRGUVFOMOMEM-UHFFFAOYSA-N butene Natural products CC=CC IAQRGUVFOMOMEM-UHFFFAOYSA-N 0.000 description 1
- 229910052793 cadmium Inorganic materials 0.000 description 1
- 238000001354 calcination Methods 0.000 description 1
- 239000003990 capacitor Substances 0.000 description 1
- 239000006229 carbon black Substances 0.000 description 1
- 239000001768 carboxy methyl cellulose Substances 0.000 description 1
- 235000010948 carboxy methyl cellulose Nutrition 0.000 description 1
- 239000008112 carboxymethyl-cellulose Substances 0.000 description 1
- 238000005266 casting Methods 0.000 description 1
- 230000015556 catabolic process Effects 0.000 description 1
- 239000000919 ceramic Substances 0.000 description 1
- 150000005678 chain carbonates Chemical class 0.000 description 1
- 239000006231 channel black Substances 0.000 description 1
- 239000011294 coal tar pitch Substances 0.000 description 1
- 239000000571 coke Substances 0.000 description 1
- 230000006835 compression Effects 0.000 description 1
- 238000007906 compression Methods 0.000 description 1
- 229920001940 conductive polymer Polymers 0.000 description 1
- 150000004292 cyclic ethers Chemical class 0.000 description 1
- 125000000753 cycloalkyl group Chemical group 0.000 description 1
- 238000006731 degradation reaction Methods 0.000 description 1
- 230000006866 deterioration Effects 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 150000004862 dioxolanes Chemical class 0.000 description 1
- NJLLQSBAHIKGKF-UHFFFAOYSA-N dipotassium dioxido(oxo)titanium Chemical compound [K+].[K+].[O-][Ti]([O-])=O NJLLQSBAHIKGKF-UHFFFAOYSA-N 0.000 description 1
- 229920001971 elastomer Polymers 0.000 description 1
- 238000003487 electrochemical reaction Methods 0.000 description 1
- 239000003759 ester based solvent Substances 0.000 description 1
- 235000019441 ethanol Nutrition 0.000 description 1
- RTZKZFJDLAIYFH-UHFFFAOYSA-N ether Chemical group CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 1
- 239000004210 ether based solvent Substances 0.000 description 1
- ZUNGGJHBMLMRFJ-UHFFFAOYSA-O ethoxy-hydroxy-oxophosphanium Chemical compound CCO[P+](O)=O ZUNGGJHBMLMRFJ-UHFFFAOYSA-O 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 230000001747 exhibiting effect Effects 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 238000000445 field-emission scanning electron microscopy Methods 0.000 description 1
- 239000011737 fluorine Substances 0.000 description 1
- 229910052731 fluorine Inorganic materials 0.000 description 1
- 239000006232 furnace black Substances 0.000 description 1
- 239000003365 glass fiber Substances 0.000 description 1
- 239000011357 graphitized carbon fiber Substances 0.000 description 1
- 229910021385 hard carbon Inorganic materials 0.000 description 1
- 229920001519 homopolymer Polymers 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-M hydroxide Chemical compound [OH-] XLYOFNOQVPJJNP-UHFFFAOYSA-M 0.000 description 1
- 239000001863 hydroxypropyl cellulose Substances 0.000 description 1
- 235000010977 hydroxypropyl cellulose Nutrition 0.000 description 1
- 239000012535 impurity Substances 0.000 description 1
- 229910052738 indium Inorganic materials 0.000 description 1
- 150000002500 ions Chemical class 0.000 description 1
- 239000003273 ketjen black Substances 0.000 description 1
- 239000005453 ketone based solvent Substances 0.000 description 1
- 239000006233 lamp black Substances 0.000 description 1
- 229910052745 lead Inorganic materials 0.000 description 1
- 229910000625 lithium cobalt oxide Inorganic materials 0.000 description 1
- 229910002102 lithium manganese oxide Inorganic materials 0.000 description 1
- QEXMICRJPVUPSN-UHFFFAOYSA-N lithium manganese(2+) oxygen(2-) Chemical class [O-2].[Mn+2].[Li+] QEXMICRJPVUPSN-UHFFFAOYSA-N 0.000 description 1
- FUJCRWPEOMXPAD-UHFFFAOYSA-N lithium oxide Chemical compound [Li+].[Li+].[O-2] FUJCRWPEOMXPAD-UHFFFAOYSA-N 0.000 description 1
- 229910001947 lithium oxide Inorganic materials 0.000 description 1
- 229910000686 lithium vanadium oxide Inorganic materials 0.000 description 1
- ACFSQHQYDZIPRL-UHFFFAOYSA-N lithium;bis(1,1,2,2,2-pentafluoroethylsulfonyl)azanide Chemical compound [Li+].FC(F)(F)C(F)(F)S(=O)(=O)[N-]S(=O)(=O)C(F)(F)C(F)(F)F ACFSQHQYDZIPRL-UHFFFAOYSA-N 0.000 description 1
- BFZPBUKRYWOWDV-UHFFFAOYSA-N lithium;oxido(oxo)cobalt Chemical compound [Li+].[O-][Co]=O BFZPBUKRYWOWDV-UHFFFAOYSA-N 0.000 description 1
- VROAXDSNYPAOBJ-UHFFFAOYSA-N lithium;oxido(oxo)nickel Chemical compound [Li+].[O-][Ni]=O VROAXDSNYPAOBJ-UHFFFAOYSA-N 0.000 description 1
- 238000002844 melting Methods 0.000 description 1
- 239000002931 mesocarbon microbead Substances 0.000 description 1
- 239000011302 mesophase pitch Substances 0.000 description 1
- 229910000000 metal hydroxide Inorganic materials 0.000 description 1
- 150000004692 metal hydroxides Chemical class 0.000 description 1
- VNWKTOKETHGBQD-UHFFFAOYSA-N methane Chemical compound C VNWKTOKETHGBQD-UHFFFAOYSA-N 0.000 description 1
- RCYSGAYIEFAJTG-UHFFFAOYSA-N methyl acetate;propanoic acid Chemical compound CCC(O)=O.COC(C)=O RCYSGAYIEFAJTG-UHFFFAOYSA-N 0.000 description 1
- PYLWMHQQBFSUBP-UHFFFAOYSA-N monofluorobenzene Chemical compound FC1=CC=CC=C1 PYLWMHQQBFSUBP-UHFFFAOYSA-N 0.000 description 1
- 150000002825 nitriles Chemical class 0.000 description 1
- 150000005181 nitrobenzenes Chemical class 0.000 description 1
- 239000005486 organic electrolyte Substances 0.000 description 1
- 239000011301 petroleum pitch Substances 0.000 description 1
- 239000011295 pitch Substances 0.000 description 1
- 229920002239 polyacrylonitrile Polymers 0.000 description 1
- 229920000139 polyethylene terephthalate Polymers 0.000 description 1
- 239000005020 polyethylene terephthalate Substances 0.000 description 1
- 229920006254 polymer film Polymers 0.000 description 1
- 239000002861 polymer material Substances 0.000 description 1
- 229920000098 polyolefin Polymers 0.000 description 1
- 229920001155 polypropylene Polymers 0.000 description 1
- 229920002451 polyvinyl alcohol Polymers 0.000 description 1
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 1
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 1
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 238000003825 pressing Methods 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- 229920001384 propylene homopolymer Polymers 0.000 description 1
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 1
- 239000002296 pyrolytic carbon Substances 0.000 description 1
- 239000001008 quinone-imine dye Substances 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 239000004627 regenerated cellulose Substances 0.000 description 1
- 238000012827 research and development Methods 0.000 description 1
- 230000002441 reversible effect Effects 0.000 description 1
- 239000005060 rubber Substances 0.000 description 1
- 239000002153 silicon-carbon composite material Substances 0.000 description 1
- 239000002356 single layer Substances 0.000 description 1
- 239000002002 slurry Substances 0.000 description 1
- 229910021384 soft carbon Inorganic materials 0.000 description 1
- 229910052596 spinel Inorganic materials 0.000 description 1
- 239000011029 spinel Substances 0.000 description 1
- 239000008107 starch Substances 0.000 description 1
- 235000019698 starch Nutrition 0.000 description 1
- 238000003860 storage Methods 0.000 description 1
- HXJUTPCZVOIRIF-UHFFFAOYSA-N sulfolane Chemical class O=S1(=O)CCCC1 HXJUTPCZVOIRIF-UHFFFAOYSA-N 0.000 description 1
- 229920005608 sulfonated EPDM Polymers 0.000 description 1
- 229910052717 sulfur Inorganic materials 0.000 description 1
- 239000011593 sulfur Substances 0.000 description 1
- 238000004381 surface treatment Methods 0.000 description 1
- 238000012360 testing method Methods 0.000 description 1
- BFKJFAAPBSQJPD-UHFFFAOYSA-N tetrafluoroethene Chemical group FC(F)=C(F)F BFKJFAAPBSQJPD-UHFFFAOYSA-N 0.000 description 1
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 1
- 239000010409 thin film Substances 0.000 description 1
- 229910052718 tin Inorganic materials 0.000 description 1
- 239000002733 tin-carbon composite material Substances 0.000 description 1
- OGIDPMRJRNCKJF-UHFFFAOYSA-N titanium oxide Inorganic materials [Ti]=O OGIDPMRJRNCKJF-UHFFFAOYSA-N 0.000 description 1
- 229910001935 vanadium oxide Inorganic materials 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 1
- 235000015041 whisky Nutrition 0.000 description 1
- 229910052725 zinc Inorganic materials 0.000 description 1
- 239000011701 zinc Substances 0.000 description 1
- 239000011787 zinc oxide Substances 0.000 description 1
Images
Classifications
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/48—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides
- H01M4/52—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of nickel, cobalt or iron
- H01M4/525—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of nickel, cobalt or iron of mixed oxides or hydroxides containing iron, cobalt or nickel for inserting or intercalating light metals, e.g. LiNiO2, LiCoO2 or LiCoOxFy
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01G—COMPOUNDS CONTAINING METALS NOT COVERED BY SUBCLASSES C01D OR C01F
- C01G53/00—Compounds of nickel
- C01G53/40—Complex oxides containing nickel and at least one other metal element
- C01G53/42—Complex oxides containing nickel and at least one other metal element containing alkali metals, e.g. LiNiO2
- C01G53/44—Complex oxides containing nickel and at least one other metal element containing alkali metals, e.g. LiNiO2 containing manganese
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01G—COMPOUNDS CONTAINING METALS NOT COVERED BY SUBCLASSES C01D OR C01F
- C01G53/00—Compounds of nickel
- C01G53/40—Complex oxides containing nickel and at least one other metal element
- C01G53/42—Complex oxides containing nickel and at least one other metal element containing alkali metals, e.g. LiNiO2
- C01G53/44—Complex oxides containing nickel and at least one other metal element containing alkali metals, e.g. LiNiO2 containing manganese
- C01G53/50—Complex oxides containing nickel and at least one other metal element containing alkali metals, e.g. LiNiO2 containing manganese of the type (MnO2)n-, e.g. Li(NixMn1-x)O2 or Li(MyNixMn1-x-y)O2
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01G—COMPOUNDS CONTAINING METALS NOT COVERED BY SUBCLASSES C01D OR C01F
- C01G53/00—Compounds of nickel
- C01G53/80—Compounds containing nickel, with or without oxygen or hydrogen, and containing one or more other elements
- C01G53/82—Compounds containing nickel, with or without oxygen or hydrogen, and containing two or more other elements
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/052—Li-accumulators
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/052—Li-accumulators
- H01M10/0525—Rocking-chair batteries, i.e. batteries with lithium insertion or intercalation in both electrodes; Lithium-ion batteries
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/04—Processes of manufacture in general
- H01M4/0471—Processes of manufacture in general involving thermal treatment, e.g. firing, sintering, backing particulate active material, thermal decomposition, pyrolysis
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/13—Electrodes for accumulators with non-aqueous electrolyte, e.g. for lithium-accumulators; Processes of manufacture thereof
- H01M4/131—Electrodes based on mixed oxides or hydroxides, or on mixtures of oxides or hydroxides, e.g. LiCoOx
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/48—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides
- H01M4/485—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of mixed oxides or hydroxides for inserting or intercalating light metals, e.g. LiTi2O4 or LiTi2OxFy
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/48—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides
- H01M4/50—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of manganese
- H01M4/505—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of manganese of mixed oxides or hydroxides containing manganese for inserting or intercalating light metals, e.g. LiMn2O4 or LiMn2OxFy
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2002/00—Crystal-structural characteristics
- C01P2002/50—Solid solutions
- C01P2002/52—Solid solutions containing elements as dopants
- C01P2002/54—Solid solutions containing elements as dopants one element only
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2002/00—Crystal-structural characteristics
- C01P2002/80—Crystal-structural characteristics defined by measured data other than those specified in group C01P2002/70
- C01P2002/85—Crystal-structural characteristics defined by measured data other than those specified in group C01P2002/70 by XPS, EDX or EDAX data
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2004/00—Particle morphology
- C01P2004/01—Particle morphology depicted by an image
- C01P2004/03—Particle morphology depicted by an image obtained by SEM
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2004/00—Particle morphology
- C01P2004/51—Particles with a specific particle size distribution
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2004/00—Particle morphology
- C01P2004/60—Particles characterised by their size
- C01P2004/61—Micrometer sized, i.e. from 1-100 micrometer
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2006/00—Physical properties of inorganic compounds
- C01P2006/40—Electric properties
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M2004/021—Physical characteristics, e.g. porosity, surface area
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M2004/026—Electrodes composed of, or comprising, active material characterised by the polarity
- H01M2004/028—Positive electrodes
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/10—Energy storage using batteries
Landscapes
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Electrochemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Inorganic Chemistry (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Manufacturing & Machinery (AREA)
- Materials Engineering (AREA)
- Battery Electrode And Active Subsutance (AREA)
- Inorganic Compounds Of Heavy Metals (AREA)
Abstract
Description
도 2는 실시예 1의 이차전지용 양극 활물질을 포함하는 리튬 이차전지에 있어서, 고온(45℃)에서 충방전 400회 실시한 이후의 양극 활물질 입자 깨짐 여부를 확인하기 위한 주사전파현미경(SEM) 사진이다.
도 3은 실시예 2의 이차전지용 양극 활물질을 포함하는 리튬 이차전지에 있어서, 고온(45℃)에서 충방전 400회 실시한 이후의 양극 활물질 입자 깨짐 여부를 확인하기 위한 주사전파현미경(SEM) 사진이다.
도 4는 실시예 3의 이차전지용 양극 활물질을 포함하는 리튬 이차전지에 있어서, 고온(45℃)에서 충방전 400회 실시한 이후의 양극 활물질 입자 깨짐 여부를 확인하기 위한 주사전파현미경(SEM) 사진이다.
도 5는 실시예 4의 이차전지용 양극 활물질을 포함하는 리튬 이차전지에 있어서, 고온(45℃)에서 충방전 400회 실시한 이후의 양극 활물질 입자 깨짐 여부를 확인하기 위한 주사전파현미경(SEM) 사진이다.
도 6은 비교예 1의 이차전지용 양극 활물질을 포함하는 리튬 이차전지에 있어서, 고온(45℃)에서 충방전 400회 실시한 이후의 양극 활물질 입자 깨짐 여부를 확인하기 위한 주사전파현미경(SEM) 사진이다.
도 7은 비교예 2의 이차전지용 양극 활물질을 포함하는 리튬 이차전지에 있어서, 고온(45℃)에서 충방전 400회 실시한 이후의 양극 활물질 입자 깨짐 여부를 확인하기 위한 주사전파현미경(SEM) 사진이다.
도 8은 비교예 3의 이차전지용 양극 활물질을 포함하는 리튬 이차전지에 있어서, 고온(45℃)에서 충방전 400회 실시한 이후의 양극 활물질 입자 깨짐 여부를 확인하기 위한 주사전파현미경(SEM) 사진이다.
도 9는 비교예 4의 이차전지용 양극 활물질을 포함하는 리튬 이차전지에 있어서, 고온(45℃)에서 충방전 400회 실시한 이후의 양극 활물질 입자 깨짐 여부를 확인하기 위한 주사전파현미경(SEM) 사진이다.
도 10은 비교예 5의 이차전지용 양극 활물질을 포함하는 리튬 이차전지에 있어서, 고온(45℃)에서 충방전 400회 실시한 이후의 양극 활물질 입자 깨짐 여부를 확인하기 위한 주사전파현미경(SEM) 사진이다.
도 11은 비교예 6의 이차전지용 양극 활물질을 포함하는 리튬 이차전지에 있어서, 고온(45℃)에서 충방전 400회 실시한 이후의 양극 활물질 입자 깨짐 여부를 확인하기 위한 주사전파현미경(SEM) 사진이다.
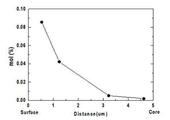
도 12는 실시예 1의 이차전지용 양극 활물질에 있어서, 위치에 따른 도핑 원소 분포 및 함량을 EDS(Energy Dispersive Spectrometer)를 통해 분석한 그래프이다.
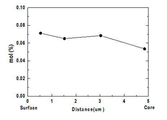
도 13은 비교예 4의 이차전지용 양극 활물질에 있어서, 위치에 따른 도핑 원소 분포 및 함량을 EDS를 통해 분석한 그래프이다.
| 실시예 | 비교예 | |||||||||
| 1 | 2 | 3 | 4 | 1 | 2 | 3 | 4 | 5 | 6 | |
| 입자 강도(MPa) | 226.2 | 237.3 | 218.5 | 253.4 | 114.7 | 171.7 | 182.2 | 190.1 | 183.2 | 298.2 |
| 45℃에서의 400회 사이클 용량유지율(%) |
|
| 실시예 1 | 87.7 |
| 실시예 2 | 90.4 |
| 실시예 3 | 86.2 |
| 실시예 4 | 85.3 |
| 비교예 1 | 64.2 |
| 비교예 2 | 75.9 |
| 비교예 3 | 80.2 |
| 비교예 4 | 83.9 |
| 비교예 5 | 82.4 |
| 비교예 6 | 73.8 |
Claims (16)
- 니켈(Ni) 및 코발트(Co)를 포함하고, 망간(Mn) 및 알루미늄(Al) 중 적어도 1종 이상을 포함하는 리튬 복합 전이금속 산화물 입자이며,
상기 리튬 복합 전이금속 산화물 입자는 상기 니켈(Ni)이 리튬을 제외한 전체 금속 중 60몰% 이상이고,
상기 리튬 복합 전이금속 산화물 입자에 도핑 원소가 도핑되고,
입자 강도가 210MPa 내지 290MPa인, 이차전지용 양극 활물질.
- 청구항 1에 있어서,
상기 도핑 원소는 리튬을 제외한 전체 금속에 대하여 6몰% 내지 10몰%로 도핑되는, 이차전지용 양극 활물질.
- 청구항 1에 있어서,
상기 도핑 원소는 P, B, Al, Si, W, Zr 및 Ti로 이루어진 군에서 선택된 적어도 1종을 포함하는, 이차전지용 양극 활물질.
- 청구항 1에 있어서,
상기 리튬 복합 전이금속 산화물 입자는 하기 화학식 1로 표시되는 화합물을 포함하는, 이차전지용 양극 활물질.
[화학식 1]
LipNi1-x-y-zCoxMa yMb zO2
화학식 1 중, 1.0≤p≤1.5, 0<x≤0.2, 0<y≤0.2, 0.06≤z≤0.1, 0<x+y+z≤0.4이고,
Ma은 Mn 및 Al으로 이루어지는 군으로부터 선택되는 적어도 1종이고,
Mb는 P, B, Al, Si, W, Zr 및 Ti으로 이루어지는 군으로부터 선택되는 적어도 1종이다.
- 청구항 1에 있어서,
평균 입경(D50)이 8㎛ 내지 30㎛인, 이차전지용 양극 활물질.
- 청구항 1에 있어서,
상기 도핑 원소는 상기 리튬 복합 전이금속 산화물 입자의 표면으로부터 중심까지 도핑 원소의 농도가 감소하도록 도핑되는, 이차전지용 양극 활물질.
- 청구항 1에 있어서,
상기 리튬 복합 전이금속 산화물 입자의 반직경에 대하여, 입자 중심으로부터 60% 내지 100%의 거리에 해당하는 영역에 상기 도핑 원소가 전체 도핑 원소 몰수의 70몰% 이상으로 도핑되는, 이차전지용 양극 활물질.
- 청구항 1에 있어서,
상기 리튬 복합 전이금속 산화물 입자는 EDS(Energy Dispersive Spectrometer)로 분석한 입자 표면과 입자 중심의 도핑 원소 농도를 하기 수학식 1에 대입하여 얻은 값이 0.7 내지 1인, 이차전지용 양극 활물질:
[수학식 1]
(Hs-Hc)/Hs
수학식 1 중, Hs는 상기 리튬 복합 전이금속 산화물 입자를 EDS로 분석하였을 때 입자 표면의 도핑 원소 농도이고, Hc는 상기 리튬 복합 전이금속 산화물 입자를 EDS로 분석하였을 때 입자 중심의 도핑 원소 농도이다.
- 니켈(Ni) 및 코발트(Co)를 포함하고, 망간(Mn) 및 알루미늄(Al) 중 적어도 1종 이상을 포함하며, 전체 금속 중 상기 니켈(Ni)이 60몰% 이상인 전이금속 수산화물 및 리튬 화합물을 혼합하고 1차 소성하여 리튬 복합 전이금속 산화물 입자를 제조하는 단계; 및
상기 리튬 복합 전이금속 산화물 입자와, 상기 도핑 원소를 포함하는 도핑 소스를 혼합하고 2차 소성하여 상기 도핑 원소를 상기 리튬 복합 전이금속 산화물 입자에 도핑시키는 단계를 포함하는, 청구항 1에 따른 이차전지용 양극 활물질의 제조방법.
- 청구항 9에 있어서,
상기 도핑 소스는 P, B, Al, Si, W, Zr 및 Ti로 이루어진 군에서 선택된 적어도 1종의 도핑 원소를 포함하는, 이차전지용 양극 활물질의 제조방법.
- 청구항 9에 있어서,
상기 1차 소성은 10 내지 25시간 동안 수행되는, 이차전지용 양극 활물질의 제조방법.
- 청구항 9에 있어서,
상기 1차 소성은 750 내지 1,000℃에서 수행되는, 이차전지용 양극 활물질의 제조방법.
- 청구항 9에 있어서,
상기 2차 소성은 3 내지 12시간 동안 수행되는, 이차전지용 양극 활물질의 제조방법.
- 청구항 9에 있어서,
상기 2차 소성은 700 내지 900℃에서 수행되는, 이차전지용 양극 활물질의 제조방법.
- 양극 집전체; 및
상기 양극 집전체 상에 형성된 양극 활물질층을 포함하고,
상기 양극 활물질층은 청구항 1에 따른 이차전지용 양극 활물질을 포함하는, 이차전지용 양극.
- 청구항 15에 따른 이차전지용 양극;
상기 이차전지용 양극과 대향하는 음극;
상기 이차전지용 양극과 음극 사이에 개재되는 세퍼레이터; 및
전해질을 포함하는, 리튬 이차전지.
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020180153838A KR102533811B1 (ko) | 2018-12-03 | 2018-12-03 | 이차전지용 양극 활물질, 이의 제조 방법, 이를 포함하는 이차전지용 양극 및 리튬 이차전지 |
| PCT/KR2019/016579 WO2020116858A1 (ko) | 2018-12-03 | 2019-11-28 | 이차전지용 양극 활물질, 이의 제조 방법, 이를 포함하는 이차전지용 양극 |
| JP2021530229A JP7214299B2 (ja) | 2018-12-03 | 2019-11-28 | 二次電池用正極活物質、この製造方法、これを含む二次電池用正極 |
| CN201980076406.9A CN113646930B (zh) | 2018-12-03 | 2019-11-28 | 二次电池用正极活性材料、其制备方法及包含其的二次电池正极 |
| US17/294,923 US12046750B2 (en) | 2018-12-03 | 2019-11-28 | Positive electrode active material for secondary battery, preparation method thereof, and positive electrode for secondary battery including same |
| EP19894008.2A EP3869595A4 (en) | 2019-11-28 | Positive electrode active material for rechargeable battery, production method therefor and rechargeable battery positive electrode comprising same |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020180153838A KR102533811B1 (ko) | 2018-12-03 | 2018-12-03 | 이차전지용 양극 활물질, 이의 제조 방법, 이를 포함하는 이차전지용 양극 및 리튬 이차전지 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| KR20200067009A true KR20200067009A (ko) | 2020-06-11 |
| KR102533811B1 KR102533811B1 (ko) | 2023-05-19 |
Family
ID=70973936
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020180153838A Active KR102533811B1 (ko) | 2018-12-03 | 2018-12-03 | 이차전지용 양극 활물질, 이의 제조 방법, 이를 포함하는 이차전지용 양극 및 리튬 이차전지 |
Country Status (5)
| Country | Link |
|---|---|
| US (1) | US12046750B2 (ko) |
| JP (1) | JP7214299B2 (ko) |
| KR (1) | KR102533811B1 (ko) |
| CN (1) | CN113646930B (ko) |
| WO (1) | WO2020116858A1 (ko) |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2023080286A1 (ko) * | 2021-11-05 | 2023-05-11 | 주식회사 엘 앤 에프 | 이차전지용 양극 활물질 |
| WO2023132641A1 (ko) * | 2022-01-07 | 2023-07-13 | 주식회사 릴엠 | 이차전지용 집전체 및 그의 제조 방법 |
| KR20240156037A (ko) * | 2023-04-21 | 2024-10-29 | (주)포스코퓨처엠 | 리튬 이차 전지용 양극 활물질 표면부의 코팅 원소 농도 분석 방법 |
| WO2026043181A1 (ko) * | 2024-08-23 | 2026-02-26 | 주식회사 엘지화학 | 양극 활물질, 이의 제조방법, 및 이를 포함하는 양극 |
Families Citing this family (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR102816582B1 (ko) * | 2020-05-27 | 2025-06-04 | 주식회사 엘지에너지솔루션 | 리튬 이차전지의 퇴화 원인 진단 방법 |
| US12126019B2 (en) * | 2020-06-15 | 2024-10-22 | Lg Chem, Ltd. | Method of preparing positive electrode active material |
| KR20220076407A (ko) * | 2020-11-30 | 2022-06-08 | 주식회사 엘지에너지솔루션 | 리튬 이차 전지용 양극 활물질, 그 제조방법 및 이를 포함하는 리튬 이차 전지 |
| CN114436347B (zh) * | 2022-03-21 | 2024-02-20 | 宁波容百新能源科技股份有限公司 | 一种高镍三元正极材料及其制备方法和应用 |
| CN115101713B (zh) * | 2022-08-26 | 2022-11-11 | 蜂巢能源科技股份有限公司 | 一种锂离子电池极片及电池 |
| KR20240158926A (ko) * | 2022-09-23 | 2024-11-05 | 베이징 이스프링 머티리얼 테크놀로지 컴퍼니 리미티드 | 리튬 함유 산화물 양극재 전구체, 리튬 함유 산화물 양극재 및 그 제조방법과 응용, 양극시트 및 그 응용 |
| CN121002672A (zh) * | 2023-08-11 | 2025-11-21 | 株式会社Lg新能源 | 正极活性材料、包含其的正极和锂二次电池 |
| US20250210651A1 (en) * | 2023-12-22 | 2025-06-26 | Lg Energy Solution, Ltd. | Lithium Secondary Battery |
| WO2025165076A1 (ko) * | 2024-02-01 | 2025-08-07 | 주식회사 엘지에너지솔루션 | 리튬 이차 전지 |
| CN119812299B (zh) * | 2025-01-07 | 2025-12-09 | 中南大学 | 一种钛掺杂高镍三元正极材料及其制备方法 |
Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20150050509A (ko) * | 2013-10-31 | 2015-05-08 | 주식회사 엘지화학 | 양극 활물질, 이의 제조방법, 및 이를 포함하는 리튬 이차전지 |
| KR20160038789A (ko) * | 2014-09-30 | 2016-04-07 | 주식회사 엘지화학 | 양극 활물질 및 이의 제조방법 |
| KR20160053849A (ko) | 2016-04-05 | 2016-05-13 | 한양대학교 산학협력단 | 양극활물질, 및 이를 포함하는 이차 전지 |
| KR20160081545A (ko) * | 2014-12-31 | 2016-07-08 | 주식회사 에코프로 | 양극활물질 및 이의 제조 방법 |
| JP2016139583A (ja) * | 2015-01-29 | 2016-08-04 | 輔仁大學學校財團法人輔仁大學 | リチウムイオン電池の金属勾配ドープ正極材料 |
| KR20170063373A (ko) * | 2015-11-30 | 2017-06-08 | 주식회사 엘지화학 | 이차전지용 양극활물질, 이를 포함하는 이차전지용 양극 및 이차전지 |
| JP2018095523A (ja) * | 2016-12-14 | 2018-06-21 | 住友化学株式会社 | リチウム金属複合酸化物粉末、リチウム二次電池用正極活物質、リチウム二次電池用正極及びリチウム二次電池 |
| KR20180077090A (ko) * | 2016-12-28 | 2018-07-06 | 주식회사 엘지화학 | 리튬 이차전지용 양극활물질, 이의 제조방법, 및 이를 포함하는 리튬 이차전지 |
| KR20180110249A (ko) * | 2017-03-27 | 2018-10-10 | 전자부품연구원 | 표면 도핑 처리된 리튬이차전지용 양극 활물질 및 그의 제조 방법 |
Family Cites Families (18)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7241532B2 (en) | 2002-03-28 | 2007-07-10 | Mitsubishi Chemical Corporation | Positive-electrode material for lithium secondary battery, secondary battery employing the same, and process for producing positive-electrode material for lithium secondary battery |
| WO2004082046A1 (ja) | 2003-03-14 | 2004-09-23 | Seimi Chemical Co., Ltd. | リチウム二次電池用正極活物質粉末 |
| JP5389620B2 (ja) | 2009-11-27 | 2014-01-15 | 株式会社日立製作所 | リチウムイオン二次電池用正極材料およびそれを用いたリチウムイオン二次電池 |
| US8871113B2 (en) * | 2010-03-31 | 2014-10-28 | Samsung Sdi Co., Ltd. | Positive active material, and positive electrode and lithium battery including positive active material |
| JP5971109B2 (ja) | 2011-12-20 | 2016-08-17 | 住友金属鉱山株式会社 | ニッケル複合水酸化物とその製造方法、非水系電解質二次電池用正極活物質とその製造方法、および非水系電解質二次電池 |
| KR101689213B1 (ko) | 2012-06-21 | 2016-12-23 | 삼성에스디아이 주식회사 | 리튬 이차 전지용 양극 활물질, 그 제조방법, 이를 포함한 리튬 이차 전지용 양극 및 이를 구비한 리튬 이차 전지 |
| CN103715424B (zh) | 2014-01-06 | 2016-06-08 | 中国科学院宁波材料技术与工程研究所 | 一种核壳结构正极材料及其制备方法 |
| KR102312369B1 (ko) | 2014-12-16 | 2021-10-12 | 에스케이이노베이션 주식회사 | 리튬 이차 전지 |
| KR20170009557A (ko) | 2015-07-17 | 2017-01-25 | 주식회사 엘지화학 | 양극 활물질 입자 강도 변경을 통해 안전성이 향상된 원통형 이차전지 |
| WO2017095081A1 (ko) | 2015-11-30 | 2017-06-08 | 주식회사 엘지화학 | 이차전지용 양극활물질, 이를 포함하는 이차전지용 양극 및 이차전지 |
| CN108028369B (zh) * | 2016-03-04 | 2020-11-17 | 株式会社Lg化学 | 二次电池用正极活性材料、其制备方法以及包含其的二次电池 |
| JP2018085221A (ja) | 2016-11-24 | 2018-05-31 | 株式会社豊田自動織機 | リチウムイオン二次電池 |
| KR102176633B1 (ko) * | 2017-02-28 | 2020-11-09 | 주식회사 엘지화학 | 리튬 이차전지용 양극 활물질, 그 제조방법 및 이를 포함하는 리튬 이차전지 |
| EP3611785A4 (en) * | 2017-04-13 | 2021-01-20 | IUCF-HYU (Industry-University Cooperation Foundation Hanyang University) | CATHODE ACTIVE MATERIAL, ITS MANUFACTURING PROCESS AND LITHIUM ACCUMULATOR CONTAINING IT |
| KR102402388B1 (ko) | 2017-04-28 | 2022-05-26 | 삼성에스디아이 주식회사 | 양극 활물질, 이를 채용한 양극과 리튬 이차 전지, 및 상기 양극 활물질의 제조방법 |
| KR101882878B1 (ko) * | 2017-09-25 | 2018-07-30 | 주식회사 엘 앤 에프 | 리튬 이차 전지용 양극 활물질, 및 이를 포함하는 리튬 이차 전지 |
| US11784310B2 (en) * | 2017-10-31 | 2023-10-10 | Sumitomo Metal Mining Co., Ltd. | Non-aqueous electrolyte secondary battery positive electrode active material, method for producing same, and non-aqueous electrolyte secondary battery which uses positive electrode active material |
| EP3712989B1 (en) | 2018-01-24 | 2024-05-22 | LG Energy Solution, Ltd. | Positive electrode active material for secondary battery, method of preparing the same, and lithium secondary battery including the positive electrode active material |
-
2018
- 2018-12-03 KR KR1020180153838A patent/KR102533811B1/ko active Active
-
2019
- 2019-11-28 WO PCT/KR2019/016579 patent/WO2020116858A1/ko not_active Ceased
- 2019-11-28 JP JP2021530229A patent/JP7214299B2/ja active Active
- 2019-11-28 CN CN201980076406.9A patent/CN113646930B/zh active Active
- 2019-11-28 US US17/294,923 patent/US12046750B2/en active Active
Patent Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20150050509A (ko) * | 2013-10-31 | 2015-05-08 | 주식회사 엘지화학 | 양극 활물질, 이의 제조방법, 및 이를 포함하는 리튬 이차전지 |
| KR20160038789A (ko) * | 2014-09-30 | 2016-04-07 | 주식회사 엘지화학 | 양극 활물질 및 이의 제조방법 |
| KR20160081545A (ko) * | 2014-12-31 | 2016-07-08 | 주식회사 에코프로 | 양극활물질 및 이의 제조 방법 |
| JP2016139583A (ja) * | 2015-01-29 | 2016-08-04 | 輔仁大學學校財團法人輔仁大學 | リチウムイオン電池の金属勾配ドープ正極材料 |
| KR20170063373A (ko) * | 2015-11-30 | 2017-06-08 | 주식회사 엘지화학 | 이차전지용 양극활물질, 이를 포함하는 이차전지용 양극 및 이차전지 |
| KR20160053849A (ko) | 2016-04-05 | 2016-05-13 | 한양대학교 산학협력단 | 양극활물질, 및 이를 포함하는 이차 전지 |
| JP2018095523A (ja) * | 2016-12-14 | 2018-06-21 | 住友化学株式会社 | リチウム金属複合酸化物粉末、リチウム二次電池用正極活物質、リチウム二次電池用正極及びリチウム二次電池 |
| KR20180077090A (ko) * | 2016-12-28 | 2018-07-06 | 주식회사 엘지화학 | 리튬 이차전지용 양극활물질, 이의 제조방법, 및 이를 포함하는 리튬 이차전지 |
| KR20180110249A (ko) * | 2017-03-27 | 2018-10-10 | 전자부품연구원 | 표면 도핑 처리된 리튬이차전지용 양극 활물질 및 그의 제조 방법 |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2023080286A1 (ko) * | 2021-11-05 | 2023-05-11 | 주식회사 엘 앤 에프 | 이차전지용 양극 활물질 |
| WO2023132641A1 (ko) * | 2022-01-07 | 2023-07-13 | 주식회사 릴엠 | 이차전지용 집전체 및 그의 제조 방법 |
| KR20240156037A (ko) * | 2023-04-21 | 2024-10-29 | (주)포스코퓨처엠 | 리튬 이차 전지용 양극 활물질 표면부의 코팅 원소 농도 분석 방법 |
| WO2026043181A1 (ko) * | 2024-08-23 | 2026-02-26 | 주식회사 엘지화학 | 양극 활물질, 이의 제조방법, 및 이를 포함하는 양극 |
Also Published As
| Publication number | Publication date |
|---|---|
| KR102533811B1 (ko) | 2023-05-19 |
| CN113646930B (zh) | 2024-09-13 |
| CN113646930A (zh) | 2021-11-12 |
| US12046750B2 (en) | 2024-07-23 |
| JP2022510653A (ja) | 2022-01-27 |
| JP7214299B2 (ja) | 2023-01-30 |
| US20220020983A1 (en) | 2022-01-20 |
| EP3869595A1 (en) | 2021-08-25 |
| WO2020116858A1 (ko) | 2020-06-11 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR102417199B1 (ko) | 이차전지용 양극 활물질, 그 제조방법 및 이를 포함하는 리튬 이차전지 | |
| KR102459883B1 (ko) | 리튬 이차전지용 양극 및 이를 포함하는 리튬 이차전지 | |
| KR102533811B1 (ko) | 이차전지용 양극 활물질, 이의 제조 방법, 이를 포함하는 이차전지용 양극 및 리튬 이차전지 | |
| KR102359103B1 (ko) | 이차전지용 양극 활물질, 그 제조방법 및 이를 포함하는 리튬 이차전지 | |
| KR102227313B1 (ko) | 이차전지용 양극재 및 이를 포함하는 리튬 이차전지 | |
| KR102453273B1 (ko) | 리튬 이차전지용 양극재, 이를 포함하는 리튬 이차전지용 양극 및 리튬 이차전지 | |
| KR102270113B1 (ko) | 이차전지용 양극의 제조방법, 이와 같이 제조된 이차전지용 양극 및 이를 포함하는 리튬 이차전지 | |
| KR102158680B1 (ko) | 이차전지용 양극의 제조방법 | |
| KR102807740B1 (ko) | 리튬 이차전지용 양극재, 상기 양극재의 제조 방법 | |
| KR102292889B1 (ko) | 리튬 복합 산화물 및 이를 포함하는 리튬 이차전지 | |
| KR102177798B1 (ko) | 이차전지용 양극 활물질, 그 제조방법 및 이를 포함하는 리튬 이차전지 | |
| EP3955344B1 (en) | Positive electrode active material for secondary battery and lithium secondary battery comprising same | |
| KR20210117212A (ko) | 리튬 이차전지용 양극재, 이를 포함하는 양극 및 리튬 이차전지 | |
| KR20210031325A (ko) | 이차전지용 양극재 및 이를 포함하는 리튬 이차전지 | |
| KR20210097025A (ko) | 이차전지용 양극 활물질 및 이를 포함하는 리튬 이차전지 | |
| KR102462952B1 (ko) | 이차전지용 양극 활물질, 그 제조방법 및 이를 포함하는 리튬 이차전지 | |
| KR102179968B1 (ko) | 리튬 이차전지용 양극 활물질의 제조방법, 이에 의해 제조된 양극 활물질, 이를 포함하는 리튬 이차전지용 양극 및 리튬 이차전지 | |
| US12021228B2 (en) | Method of producing positive electrode material for secondary battery | |
| KR102459882B1 (ko) | 리튬 이차전지용 양극 및 이를 포함하는 리튬 이차전지 | |
| KR20200095884A (ko) | 이차전지용 양극 활물질 및 이를 포함하는 리튬 이차전지 | |
| KR20210136842A (ko) | 수분과의 반응성이 완화된 고-니켈 전극 시트 및 이의 제조방법 | |
| KR20220101316A (ko) | 양극 활물질의 제조방법 및 양극 활물질 | |
| KR20230162576A (ko) | 양극 활물질 및 이의 제조방법 | |
| KR20230029562A (ko) | 양극 활물질 및 양극 활물질의 제조 방법 | |
| KR20190092825A (ko) | 리튬 이차전지용 양극 활물질, 이를 포함하는 리튬 이차전지용 양극 및 리튬 이차전지 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0109 | Patent application |
Patent event code: PA01091R01D Comment text: Patent Application Patent event date: 20181203 |
|
| PG1501 | Laying open of application | ||
| A201 | Request for examination | ||
| PA0201 | Request for examination |
Patent event code: PA02012R01D Patent event date: 20201014 Comment text: Request for Examination of Application Patent event code: PA02011R01I Patent event date: 20181203 Comment text: Patent Application |
|
| N231 | Notification of change of applicant | ||
| PN2301 | Change of applicant |
Patent event date: 20210524 Comment text: Notification of Change of Applicant Patent event code: PN23011R01D |
|
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
Comment text: Notification of reason for refusal Patent event date: 20220903 Patent event code: PE09021S01D |
|
| E701 | Decision to grant or registration of patent right | ||
| PE0701 | Decision of registration |
Patent event code: PE07011S01D Comment text: Decision to Grant Registration Patent event date: 20230315 |
|
| GRNT | Written decision to grant | ||
| PR0701 | Registration of establishment |
Comment text: Registration of Establishment Patent event date: 20230515 Patent event code: PR07011E01D |
|
| PR1002 | Payment of registration fee |
Payment date: 20230516 End annual number: 3 Start annual number: 1 |
|
| PG1601 | Publication of registration |