KR20210077752A - Scm-18 알루미노포스페이트 분자체, 이의 제조 방법 및 용도 - Google Patents
Scm-18 알루미노포스페이트 분자체, 이의 제조 방법 및 용도 Download PDFInfo
- Publication number
- KR20210077752A KR20210077752A KR1020217015407A KR20217015407A KR20210077752A KR 20210077752 A KR20210077752 A KR 20210077752A KR 1020217015407 A KR1020217015407 A KR 1020217015407A KR 20217015407 A KR20217015407 A KR 20217015407A KR 20210077752 A KR20210077752 A KR 20210077752A
- Authority
- KR
- South Korea
- Prior art keywords
- molecular sieve
- aluminum
- aluminophosphate molecular
- precursor
- aluminophosphate
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 239000002808 molecular sieve Substances 0.000 title claims abstract description 225
- URGAHOPLAPQHLN-UHFFFAOYSA-N sodium aluminosilicate Chemical compound [Na+].[Al+3].[O-][Si]([O-])=O.[O-][Si]([O-])=O URGAHOPLAPQHLN-UHFFFAOYSA-N 0.000 title claims abstract description 225
- 238000002360 preparation method Methods 0.000 title abstract description 5
- 238000002441 X-ray diffraction Methods 0.000 claims abstract description 82
- 229910018072 Al 2 O 3 Inorganic materials 0.000 claims abstract description 57
- 239000000203 mixture Substances 0.000 claims abstract description 54
- 229910052782 aluminium Inorganic materials 0.000 claims abstract description 43
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 claims abstract description 43
- 238000000034 method Methods 0.000 claims abstract description 42
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 claims abstract description 41
- 229910052698 phosphorus Inorganic materials 0.000 claims abstract description 41
- 239000011574 phosphorus Substances 0.000 claims abstract description 41
- 239000000126 substance Substances 0.000 claims abstract description 26
- 239000003054 catalyst Substances 0.000 claims abstract description 19
- 239000003463 adsorbent Substances 0.000 claims abstract description 16
- 239000002243 precursor Substances 0.000 claims description 78
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 claims description 34
- 238000002425 crystallisation Methods 0.000 claims description 28
- 230000008025 crystallization Effects 0.000 claims description 28
- 230000001747 exhibiting effect Effects 0.000 claims description 24
- 239000011368 organic material Substances 0.000 claims description 22
- 239000012452 mother liquor Substances 0.000 claims description 19
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 18
- 229910000147 aluminium phosphate Inorganic materials 0.000 claims description 17
- AMNPXXIGUOKIPP-UHFFFAOYSA-N [4-(carbamothioylamino)phenyl]thiourea Chemical compound NC(=S)NC1=CC=C(NC(N)=S)C=C1 AMNPXXIGUOKIPP-UHFFFAOYSA-N 0.000 claims description 15
- 238000006243 chemical reaction Methods 0.000 claims description 15
- 125000000325 methylidene group Chemical group [H]C([H])=* 0.000 claims description 15
- 238000001354 calcination Methods 0.000 claims description 12
- SMZOGRDCAXLAAR-UHFFFAOYSA-N aluminium isopropoxide Chemical compound [Al+3].CC(C)[O-].CC(C)[O-].CC(C)[O-] SMZOGRDCAXLAAR-UHFFFAOYSA-N 0.000 claims description 8
- 125000006273 (C1-C3) alkyl group Chemical group 0.000 claims description 6
- 125000004169 (C1-C6) alkyl group Chemical group 0.000 claims description 6
- VXAUWWUXCIMFIM-UHFFFAOYSA-M aluminum;oxygen(2-);hydroxide Chemical compound [OH-].[O-2].[Al+3] VXAUWWUXCIMFIM-UHFFFAOYSA-M 0.000 claims description 6
- 238000001035 drying Methods 0.000 claims description 6
- 229910052739 hydrogen Inorganic materials 0.000 claims description 6
- 238000002156 mixing Methods 0.000 claims description 6
- DLYUQMMRRRQYAE-UHFFFAOYSA-N tetraphosphorus decaoxide Chemical compound O1P(O2)(=O)OP3(=O)OP1(=O)OP2(=O)O3 DLYUQMMRRRQYAE-UHFFFAOYSA-N 0.000 claims description 6
- 238000005406 washing Methods 0.000 claims description 6
- 239000011230 binding agent Substances 0.000 claims description 5
- 238000004519 manufacturing process Methods 0.000 claims description 5
- VSCWAEJMTAWNJL-UHFFFAOYSA-K aluminium trichloride Chemical compound Cl[Al](Cl)Cl VSCWAEJMTAWNJL-UHFFFAOYSA-K 0.000 claims description 4
- VHUUQVKOLVNVRT-UHFFFAOYSA-N Ammonium hydroxide Chemical group [NH4+].[OH-] VHUUQVKOLVNVRT-UHFFFAOYSA-N 0.000 claims description 3
- ABLZXFCXXLZCGV-UHFFFAOYSA-N Phosphorous acid Chemical compound OP(O)=O ABLZXFCXXLZCGV-UHFFFAOYSA-N 0.000 claims description 3
- 239000000908 ammonium hydroxide Substances 0.000 claims description 3
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 3
- TWNQGVIAIRXVLR-UHFFFAOYSA-N oxo(oxoalumanyloxy)alumane Chemical compound O=[Al]O[Al]=O TWNQGVIAIRXVLR-UHFFFAOYSA-N 0.000 claims description 3
- WNROFYMDJYEPJX-UHFFFAOYSA-K aluminium hydroxide Chemical compound [OH-].[OH-].[OH-].[Al+3] WNROFYMDJYEPJX-UHFFFAOYSA-K 0.000 claims description 2
- DIZPMCHEQGEION-UHFFFAOYSA-H aluminium sulfate (anhydrous) Chemical compound [Al+3].[Al+3].[O-]S([O-])(=O)=O.[O-]S([O-])(=O)=O.[O-]S([O-])(=O)=O DIZPMCHEQGEION-UHFFFAOYSA-H 0.000 claims description 2
- 238000002354 inductively-coupled plasma atomic emission spectroscopy Methods 0.000 description 26
- 239000000243 solution Substances 0.000 description 26
- 229910017119 AlPO Inorganic materials 0.000 description 17
- 239000011148 porous material Substances 0.000 description 16
- 239000007788 liquid Substances 0.000 description 13
- 238000001179 sorption measurement Methods 0.000 description 11
- 238000003756 stirring Methods 0.000 description 11
- MWUXSHHQAYIFBG-UHFFFAOYSA-N Nitric oxide Chemical compound O=[N] MWUXSHHQAYIFBG-UHFFFAOYSA-N 0.000 description 6
- 239000005416 organic matter Substances 0.000 description 5
- 239000000843 powder Substances 0.000 description 5
- 239000010457 zeolite Substances 0.000 description 5
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 4
- 230000015572 biosynthetic process Effects 0.000 description 4
- 238000003786 synthesis reaction Methods 0.000 description 4
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 3
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 3
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 3
- 239000012298 atmosphere Substances 0.000 description 3
- 239000007789 gas Substances 0.000 description 3
- 229910052751 metal Inorganic materials 0.000 description 3
- 239000002184 metal Substances 0.000 description 3
- -1 organic molecules Substances 0.000 description 3
- 229910052710 silicon Inorganic materials 0.000 description 3
- 239000010703 silicon Substances 0.000 description 3
- 239000007787 solid Substances 0.000 description 3
- 239000007858 starting material Substances 0.000 description 3
- RHQQHZQUAMFINJ-GKWSUJDHSA-N 1-[(3s,5s,8s,9s,10s,11s,13s,14s,17s)-3,11-dihydroxy-10,13-dimethyl-2,3,4,5,6,7,8,9,11,12,14,15,16,17-tetradecahydro-1h-cyclopenta[a]phenanthren-17-yl]-2-hydroxyethanone Chemical compound C1[C@@H](O)CC[C@]2(C)[C@H]3[C@@H](O)C[C@](C)([C@H](CC4)C(=O)CO)[C@@H]4[C@@H]3CC[C@H]21 RHQQHZQUAMFINJ-GKWSUJDHSA-N 0.000 description 2
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 2
- NQRYJNQNLNOLGT-UHFFFAOYSA-N Piperidine Chemical compound C1CCNCC1 NQRYJNQNLNOLGT-UHFFFAOYSA-N 0.000 description 2
- 229910021536 Zeolite Inorganic materials 0.000 description 2
- XLOMVQKBTHCTTD-UHFFFAOYSA-N Zinc monoxide Chemical compound [Zn]=O XLOMVQKBTHCTTD-UHFFFAOYSA-N 0.000 description 2
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 2
- 150000001412 amines Chemical class 0.000 description 2
- 239000007864 aqueous solution Substances 0.000 description 2
- HQABUPZFAYXKJW-UHFFFAOYSA-N butan-1-amine Chemical compound CCCCN HQABUPZFAYXKJW-UHFFFAOYSA-N 0.000 description 2
- 238000004364 calculation method Methods 0.000 description 2
- 239000003575 carbonaceous material Substances 0.000 description 2
- 239000013078 crystal Substances 0.000 description 2
- PAFZNILMFXTMIY-UHFFFAOYSA-N cyclohexylamine Chemical compound NC1CCCCC1 PAFZNILMFXTMIY-UHFFFAOYSA-N 0.000 description 2
- HNPSIPDUKPIQMN-UHFFFAOYSA-N dioxosilane;oxo(oxoalumanyloxy)alumane Chemical compound O=[Si]=O.O=[Al]O[Al]=O HNPSIPDUKPIQMN-UHFFFAOYSA-N 0.000 description 2
- 238000001027 hydrothermal synthesis Methods 0.000 description 2
- ZXEKIIBDNHEJCQ-UHFFFAOYSA-N isobutanol Chemical compound CC(C)CO ZXEKIIBDNHEJCQ-UHFFFAOYSA-N 0.000 description 2
- 239000000463 material Substances 0.000 description 2
- 238000005259 measurement Methods 0.000 description 2
- 229910044991 metal oxide Inorganic materials 0.000 description 2
- 150000004706 metal oxides Chemical class 0.000 description 2
- VNWKTOKETHGBQD-UHFFFAOYSA-N methane Chemical compound C VNWKTOKETHGBQD-UHFFFAOYSA-N 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- 230000007935 neutral effect Effects 0.000 description 2
- 238000011056 performance test Methods 0.000 description 2
- 230000005855 radiation Effects 0.000 description 2
- 229910002027 silica gel Inorganic materials 0.000 description 2
- 239000000741 silica gel Substances 0.000 description 2
- 230000002194 synthesizing effect Effects 0.000 description 2
- VDZOOKBUILJEDG-UHFFFAOYSA-M tetrabutylammonium hydroxide Chemical compound [OH-].CCCC[N+](CCCC)(CCCC)CCCC VDZOOKBUILJEDG-UHFFFAOYSA-M 0.000 description 2
- WGTYBPLFGIVFAS-UHFFFAOYSA-M tetramethylammonium hydroxide Chemical compound [OH-].C[N+](C)(C)C WGTYBPLFGIVFAS-UHFFFAOYSA-M 0.000 description 2
- PTTPXKJBFFKCEK-UHFFFAOYSA-N 2-Methyl-4-heptanone Chemical compound CC(C)CC(=O)CC(C)C PTTPXKJBFFKCEK-UHFFFAOYSA-N 0.000 description 1
- 241000269350 Anura Species 0.000 description 1
- 241000408939 Atalopedes campestris Species 0.000 description 1
- QPLDLSVMHZLSFG-UHFFFAOYSA-N Copper oxide Chemical compound [Cu]=O QPLDLSVMHZLSFG-UHFFFAOYSA-N 0.000 description 1
- 239000005751 Copper oxide Substances 0.000 description 1
- PIICEJLVQHRZGT-UHFFFAOYSA-N Ethylenediamine Chemical compound NCCN PIICEJLVQHRZGT-UHFFFAOYSA-N 0.000 description 1
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 1
- 239000005909 Kieselgur Substances 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- PWHULOQIROXLJO-UHFFFAOYSA-N Manganese Chemical compound [Mn] PWHULOQIROXLJO-UHFFFAOYSA-N 0.000 description 1
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 1
- 239000011398 Portland cement Substances 0.000 description 1
- 244000275012 Sesbania cannabina Species 0.000 description 1
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 1
- AVUYXHYHTTVPRX-UHFFFAOYSA-N Tris(2-methyl-1-aziridinyl)phosphine oxide Chemical compound CC1CN1P(=O)(N1C(C1)C)N1C(C)C1 AVUYXHYHTTVPRX-UHFFFAOYSA-N 0.000 description 1
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 1
- 229910052908 analcime Inorganic materials 0.000 description 1
- 239000010425 asbestos Substances 0.000 description 1
- UNYSKUBLZGJSLV-UHFFFAOYSA-L calcium;1,3,5,2,4,6$l^{2}-trioxadisilaluminane 2,4-dioxide;dihydroxide;hexahydrate Chemical compound O.O.O.O.O.O.[OH-].[OH-].[Ca+2].O=[Si]1O[Al]O[Si](=O)O1.O=[Si]1O[Al]O[Si](=O)O1 UNYSKUBLZGJSLV-UHFFFAOYSA-L 0.000 description 1
- 239000000969 carrier Substances 0.000 description 1
- 238000003421 catalytic decomposition reaction Methods 0.000 description 1
- 238000005119 centrifugation Methods 0.000 description 1
- 229910052676 chabazite Inorganic materials 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 239000004927 clay Substances 0.000 description 1
- 239000002734 clay mineral Substances 0.000 description 1
- 229910017052 cobalt Inorganic materials 0.000 description 1
- 239000010941 cobalt Substances 0.000 description 1
- GUTLYIVDDKVIGB-UHFFFAOYSA-N cobalt atom Chemical compound [Co] GUTLYIVDDKVIGB-UHFFFAOYSA-N 0.000 description 1
- 230000002301 combined effect Effects 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- 229910000431 copper oxide Inorganic materials 0.000 description 1
- 239000002178 crystalline material Substances 0.000 description 1
- 239000008367 deionised water Substances 0.000 description 1
- 229910021641 deionized water Inorganic materials 0.000 description 1
- 239000003599 detergent Substances 0.000 description 1
- 239000012973 diazabicyclooctane Substances 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 230000007613 environmental effect Effects 0.000 description 1
- 239000000835 fiber Substances 0.000 description 1
- 238000001914 filtration Methods 0.000 description 1
- 239000008187 granular material Substances 0.000 description 1
- 229910001690 harmotome Inorganic materials 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- 229910052742 iron Inorganic materials 0.000 description 1
- 229940035429 isobutyl alcohol Drugs 0.000 description 1
- JJWLVOIRVHMVIS-UHFFFAOYSA-N isopropylamine Chemical compound CC(C)N JJWLVOIRVHMVIS-UHFFFAOYSA-N 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 230000014759 maintenance of location Effects 0.000 description 1
- 229910052748 manganese Inorganic materials 0.000 description 1
- 239000011572 manganese Substances 0.000 description 1
- 150000002739 metals Chemical class 0.000 description 1
- 239000003345 natural gas Substances 0.000 description 1
- 229910017604 nitric acid Inorganic materials 0.000 description 1
- 239000012299 nitrogen atmosphere Substances 0.000 description 1
- 150000002894 organic compounds Chemical class 0.000 description 1
- 239000003960 organic solvent Substances 0.000 description 1
- 125000004430 oxygen atom Chemical group O* 0.000 description 1
- 239000002245 particle Substances 0.000 description 1
- 125000001453 quaternary ammonium group Chemical group 0.000 description 1
- SBYHFKPVCBCYGV-UHFFFAOYSA-N quinuclidine Chemical compound C1CC2CCN1CC2 SBYHFKPVCBCYGV-UHFFFAOYSA-N 0.000 description 1
- 230000002468 redox effect Effects 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 229910052895 riebeckite Inorganic materials 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 229910052814 silicon oxide Inorganic materials 0.000 description 1
- 238000002336 sorption--desorption measurement Methods 0.000 description 1
- 238000006467 substitution reaction Methods 0.000 description 1
- 238000000967 suction filtration Methods 0.000 description 1
- 238000001308 synthesis method Methods 0.000 description 1
- 238000012360 testing method Methods 0.000 description 1
- 229940073455 tetraethylammonium hydroxide Drugs 0.000 description 1
- LRGJRHZIDJQFCL-UHFFFAOYSA-M tetraethylazanium;hydroxide Chemical compound [OH-].CC[N+](CC)(CC)CC LRGJRHZIDJQFCL-UHFFFAOYSA-M 0.000 description 1
- LPSKDVINWQNWFE-UHFFFAOYSA-M tetrapropylazanium;hydroxide Chemical compound [OH-].CCC[N+](CCC)(CCC)CCC LPSKDVINWQNWFE-UHFFFAOYSA-M 0.000 description 1
- 238000002411 thermogravimetry Methods 0.000 description 1
- 239000010936 titanium Substances 0.000 description 1
- 229910052719 titanium Inorganic materials 0.000 description 1
- 229910052723 transition metal Inorganic materials 0.000 description 1
- IMNIMPAHZVJRPE-UHFFFAOYSA-N triethylenediamine Chemical compound C1CN2CCN1CC2 IMNIMPAHZVJRPE-UHFFFAOYSA-N 0.000 description 1
- YFTHZRPMJXBUME-UHFFFAOYSA-N tripropylamine Chemical compound CCCN(CCC)CCC YFTHZRPMJXBUME-UHFFFAOYSA-N 0.000 description 1
- 230000004580 weight loss Effects 0.000 description 1
- 239000011701 zinc Substances 0.000 description 1
- 229910052725 zinc Inorganic materials 0.000 description 1
- 239000011787 zinc oxide Substances 0.000 description 1
Images
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J29/00—Catalysts comprising molecular sieves
- B01J29/82—Phosphates
- B01J29/83—Aluminophosphates [APO compounds]
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/02—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof comprising inorganic material
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/02—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof comprising inorganic material
- B01J20/0203—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof comprising inorganic material comprising compounds of metals not provided for in B01J20/04
- B01J20/0274—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof comprising inorganic material comprising compounds of metals not provided for in B01J20/04 characterised by the type of anion
- B01J20/0292—Phosphates of compounds other than those provided for in B01J20/048
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/02—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof comprising inorganic material
- B01J20/10—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof comprising inorganic material comprising silica or silicate
- B01J20/16—Alumino-silicates
- B01J20/18—Synthetic zeolitic molecular sieves
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/28—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof characterised by their form or physical properties
- B01J20/28002—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof characterised by their form or physical properties characterised by their physical properties
- B01J20/28011—Other properties, e.g. density, crush strength
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/28—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof characterised by their form or physical properties
- B01J20/28054—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof characterised by their form or physical properties characterised by their surface properties or porosity
- B01J20/28057—Surface area, e.g. B.E.T specific surface area
- B01J20/28061—Surface area, e.g. B.E.T specific surface area being in the range 100-500 m2/g
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/28—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof characterised by their form or physical properties
- B01J20/28054—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof characterised by their form or physical properties characterised by their surface properties or porosity
- B01J20/28069—Pore volume, e.g. total pore volume, mesopore volume, micropore volume
- B01J20/28071—Pore volume, e.g. total pore volume, mesopore volume, micropore volume being less than 0.5 ml/g
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/30—Processes for preparing, regenerating, or reactivating
- B01J20/3078—Thermal treatment, e.g. calcining or pyrolizing
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/30—Processes for preparing, regenerating, or reactivating
- B01J20/3085—Chemical treatments not covered by groups B01J20/3007 - B01J20/3078
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J37/00—Processes, in general, for preparing catalysts; Processes, in general, for activation of catalysts
- B01J37/04—Mixing
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J37/00—Processes, in general, for preparing catalysts; Processes, in general, for activation of catalysts
- B01J37/06—Washing
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J37/00—Processes, in general, for preparing catalysts; Processes, in general, for activation of catalysts
- B01J37/08—Heat treatment
- B01J37/082—Decomposition and pyrolysis
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01B—NON-METALLIC ELEMENTS; COMPOUNDS THEREOF; METALLOIDS OR COMPOUNDS THEREOF NOT COVERED BY SUBCLASS C01C
- C01B37/00—Compounds having molecular sieve properties but not having base-exchange properties
- C01B37/04—Aluminophosphates [APO compounds]
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01B—NON-METALLIC ELEMENTS; COMPOUNDS THEREOF; METALLOIDS OR COMPOUNDS THEREOF NOT COVERED BY SUBCLASS C01C
- C01B39/00—Compounds having molecular sieve and base-exchange properties, e.g. crystalline zeolites; Their preparation; After-treatment, e.g. ion-exchange or dealumination
- C01B39/54—Phosphates, e.g. APO or SAPO compounds
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2002/00—Crystal-structural characteristics
- C01P2002/70—Crystal-structural characteristics defined by measured X-ray, neutron or electron diffraction data
- C01P2002/72—Crystal-structural characteristics defined by measured X-ray, neutron or electron diffraction data by d-values or two theta-values, e.g. as X-ray diagram
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2006/00—Physical properties of inorganic compounds
- C01P2006/12—Surface area
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2006/00—Physical properties of inorganic compounds
- C01P2006/14—Pore volume
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Inorganic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Analytical Chemistry (AREA)
- General Life Sciences & Earth Sciences (AREA)
- Geology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Physics & Mathematics (AREA)
- Thermal Sciences (AREA)
- Silicates, Zeolites, And Molecular Sieves (AREA)
- Catalysts (AREA)
- Solid-Sorbent Or Filter-Aiding Compositions (AREA)
Abstract
Description
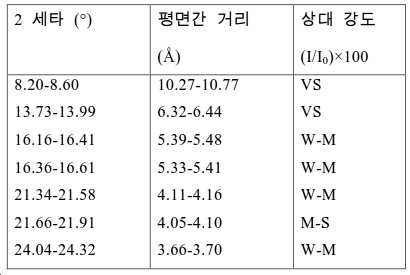
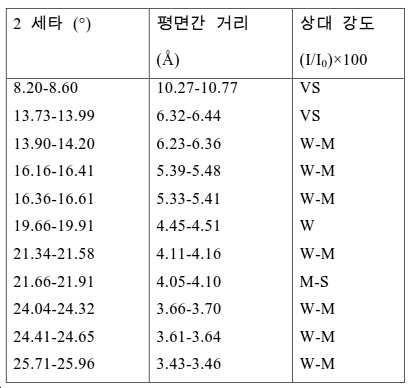
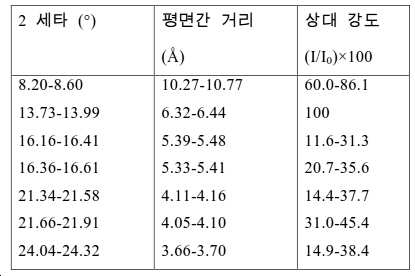
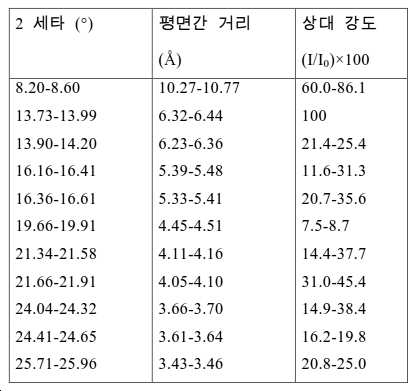
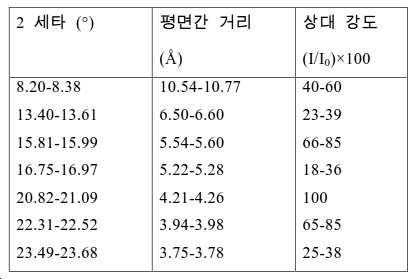
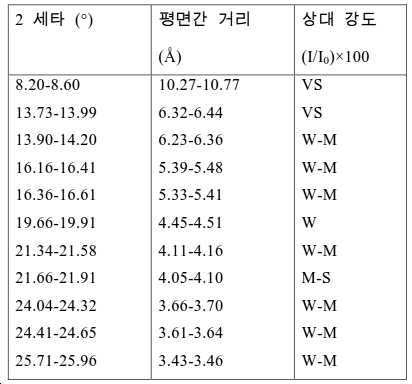
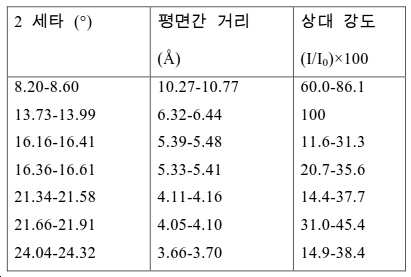
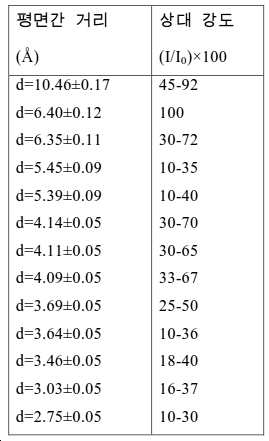
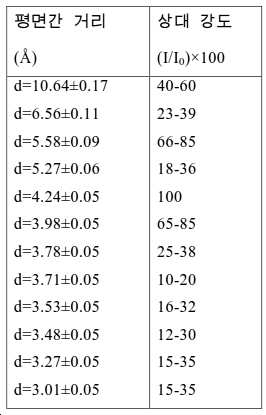
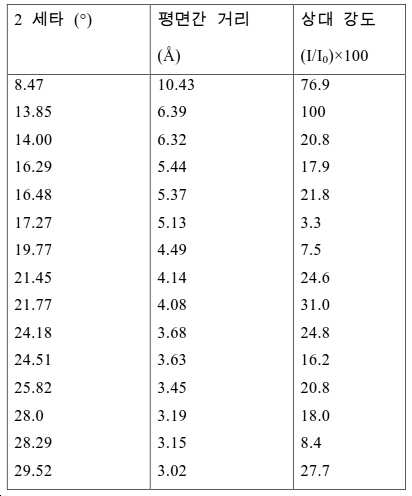
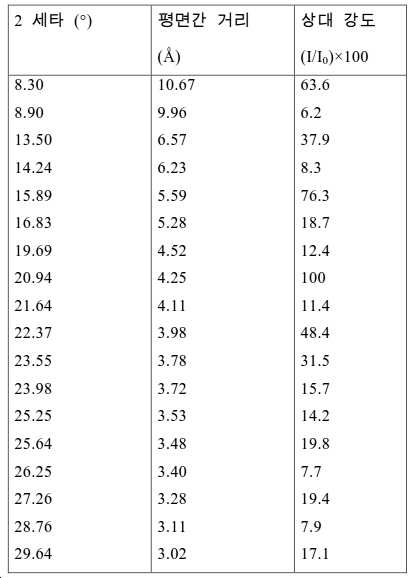
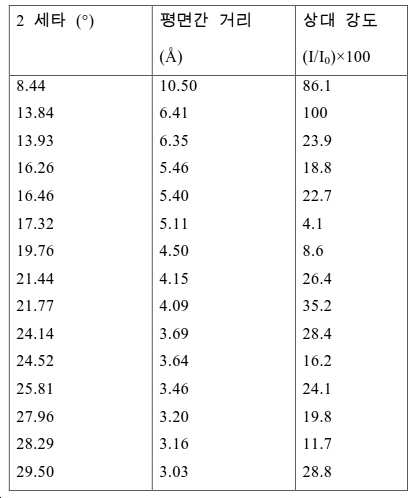
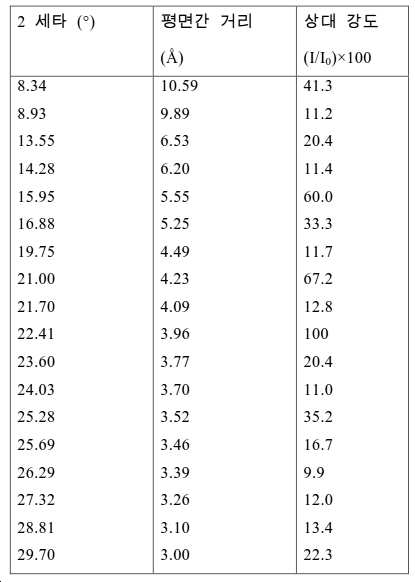
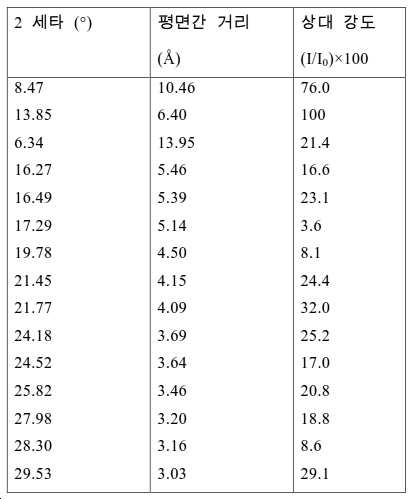
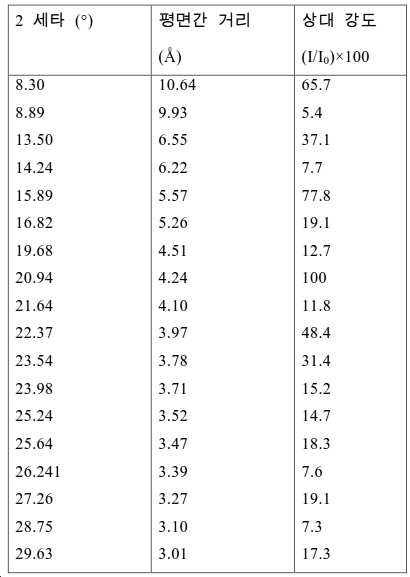
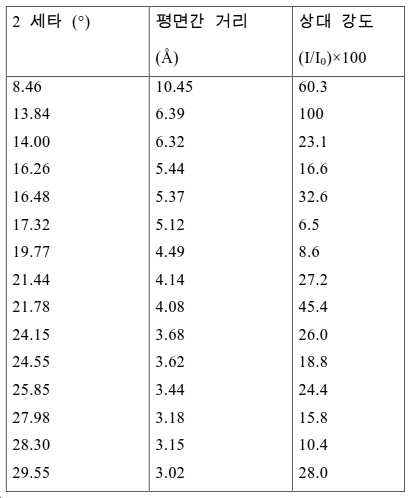
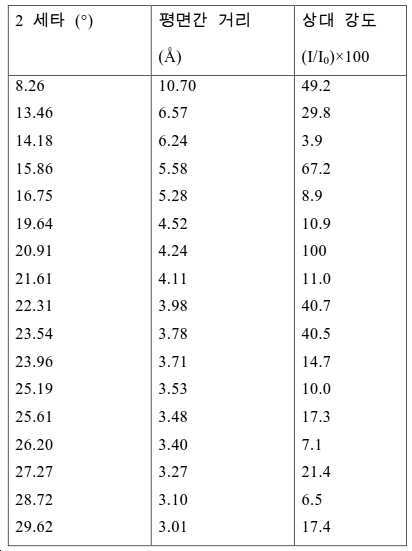
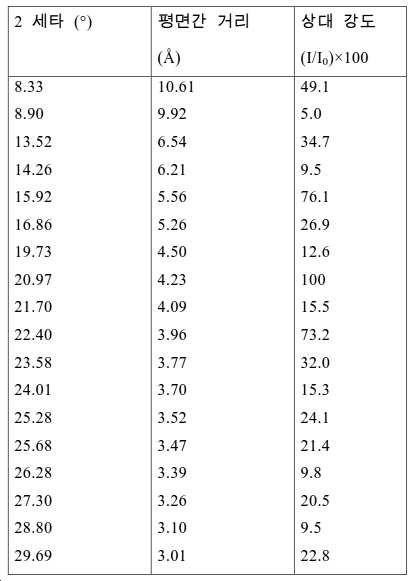
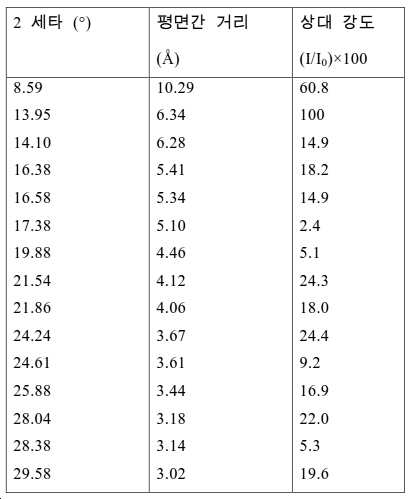
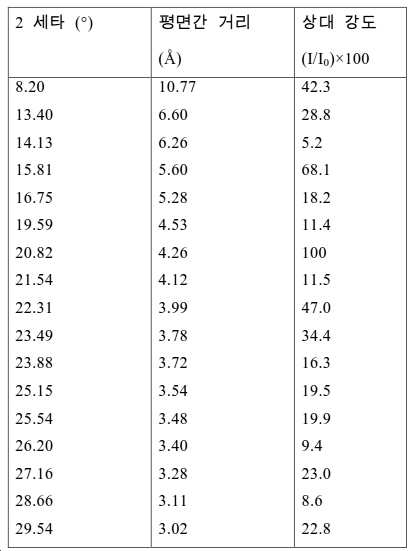
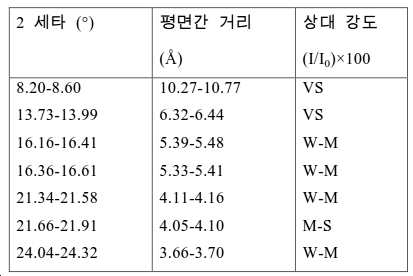
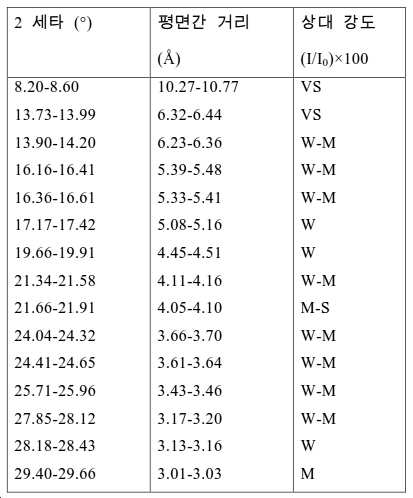
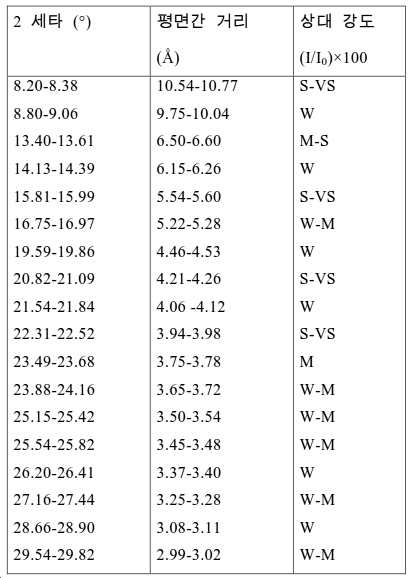
도 2는 실시예 1에서 수득된 알루미노포스페이트 분자체의 XRD 패턴을 나타낸다.
Claims (15)
- 제1항 내지 제3항 중 어느 한 항에 있어서,
상기 분자체는 약 150 내지 500 m2/g, 바람직하게는 약 200 내지 400 m2/g의 비표면적 및 약 0.9 내지 0.25 ml/g, 바람직하게는 약 0.10 내지 0.20 ml/g의 마이크로기공 부피를 가지는, 알루미노포스페이트 분자체. - 제5항 또는 제6항에 있어서,
상기 i) 단계가 하기 ia) 및 ib) 단계를 추가로 포함하고,
ia) 1 : (1.0-3.0) : (1.5-6.0) : (50-500)의 알루미늄 소스 (Al2O3로 계산) : 인 소스(P2O5로 계산) : R : H2O의 몰비로 알루미늄 소스, 인 소스, 유기 물질 R 및 물을 혼합하여, 합성 모액을 얻는 단계; 및
ib) 상기 합성 모액을 결정화시켜 알루미노포스페이트 분자체 전구체를 얻는 단계,
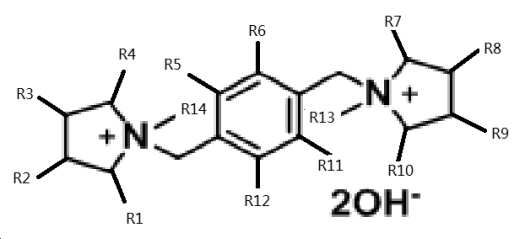
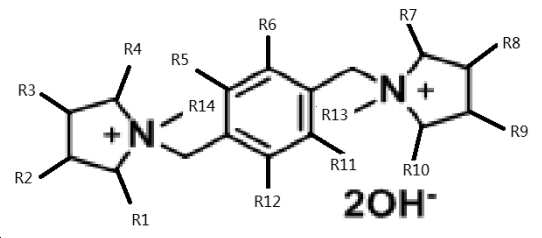
여기서 상기 유기 물질 R은 다음 식을 가지는 수산화 암모늄이며,
상기 식에서, R1-R12 기는 서로 동일하거나 상이할 수 있고, 독립적으로 H 및 C1-6 알킬기로부터, 바람직하게는 H 및 C1-3 알킬기로부터 선택되고, 더 바람직하게는 H이고;
R13 및 R14 기는 서로 동일하거나 상이할 수 있고, 독립적으로 C1-6 알킬기, 바람직하게는 C1-3 알킬기로부터 선택되고, 더 바람직하게는 메틸인, 방법. - 제7항에 있어서,
상기 ia) 단계에서, 알루미늄 소스, 인 소스, 유기 물질 R 및 물은, 약 1 : (1.0-2.0) : (2.5-4.8) : (100-300)의 알루미늄 소스 (Al2O3로 계산) : 인 소스(P2O5로 계산) : R : H2O의 몰비로 혼합되는, 방법. - 제7항 또는 제8항에 있어서,
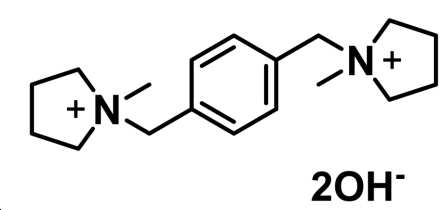
상기 유기 물질 R은 1,1-[1,4-페닐렌비스(메틸렌)]비스-1-메틸피롤리디늄 디하이드록사이드인, 방법. - 제7항 내지 제9항 중 어느 한 항에 있어서,
상기 ib) 단계는, 밀봉된 반응 용기, 약 130 내지 200 ℃, 바람직하게는 약 145 내지 185 ℃의 결정화 온도 및 약 24 내지 150 시간, 바람직하게는 약 48 내지 120 시간의 결정화 시간의 조건 하에서 수행되고,
바람직하게는, 상기 ib) 단계는 생성된 알루미노포스페이트 분자체 전구체를 세척 및 건조하는 단계를 추가로 포함하는, 방법. - 제7항 내지 제10항 중 어느 한 항에 있어서,
상기 알루미늄 소스는 슈도-보헤마이트(pseudo-boehmite), 알루미늄 이소프로폭사이드, 알루미늄 졸, 수산화 알루미늄, 황산 알루미늄, 염화 알루미늄 및 산화 알루미늄으로 이루어진 군에서 선택된 하나 이상, 바람직하게는 슈도-보헤마이트 및 알루미늄 이소프로폭사이드로 이루어진 군으로부터 선택된 하나 이상이고; 및/또는
상기 인 소스는 인산, 오르토아인산(orthophosphorous acid) 및 오산화인으로 이루어진 군에서 선택된 하나 이상, 바람직하게는 인산인, 방법. - 제5항 내지 제11항 중 어느 한 항에 있어서,
상기 알루미노포스페이트 분자체 전구체는, P2O5로 계산된 인 대 Al2O3로 계산된 알루미늄의 몰비(즉, P2O5/Al2O3)가 약 0.8 내지 약 1.2의 범위이고,
선택적으로 상기 알루미노포스페이트 분자체 전구체는, 알루미노포스페이트 분자체 전구체의 중량을 기준으로 약 8 중량% 내지 약 40 중량%의 유기 물질을 포함하는, 방법. - 제5항 내지 제12항 중 어느 한 항의 방법에 의해 얻어진 알루미노포스페이트 분자체.
- 제1항 내지 제4항 및 제13항 중 어느 한 항의 알루미노포스페이트 분자체 및 결합제를 포함하는, 분자체 조성물.
- 제1항 내지 제4항 및 제13항 중 어느 한 항의 알루미노포스페이트 분자체 또는 제14항의 분자체 조성물의 흡착제, 촉매 또는 촉매 담체로서의 용도.
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201811250883.6A CN111099603B (zh) | 2018-10-25 | 2018-10-25 | Scm-18分子筛及其制备方法 |
| CN201811250883.6 | 2018-10-25 | ||
| PCT/CN2019/106628 WO2020082944A1 (zh) | 2018-10-25 | 2019-09-19 | 磷铝分子筛scm-18、其制备方法及应用 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| KR20210077752A true KR20210077752A (ko) | 2021-06-25 |
| KR102709740B1 KR102709740B1 (ko) | 2024-09-24 |
Family
ID=70331296
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020217015407A Active KR102709740B1 (ko) | 2018-10-25 | 2019-09-19 | Scm-18 알루미노포스페이트 분자체, 이의 제조 방법 및 용도 |
Country Status (9)
| Country | Link |
|---|---|
| US (1) | US11819834B2 (ko) |
| EP (1) | EP3872032A4 (ko) |
| JP (1) | JP7373561B2 (ko) |
| KR (1) | KR102709740B1 (ko) |
| CN (1) | CN111099603B (ko) |
| CA (1) | CA3117388C (ko) |
| SG (1) | SG11202104152VA (ko) |
| TW (1) | TWI819099B (ko) |
| WO (1) | WO2020082944A1 (ko) |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN115504485B (zh) * | 2021-06-23 | 2023-08-29 | 中国石油化工股份有限公司 | 一种sapo-17分子筛的制备方法 |
| CN114180597B (zh) * | 2020-09-14 | 2023-06-06 | 中国石油化工股份有限公司 | Scm-34分子筛及其制备方法和应用 |
| US12194446B2 (en) | 2020-09-14 | 2025-01-14 | China Petroleum & Chemical Corporation | SCM-34 molecular sieve, preparation method therefor and use thereof |
| CN116618002B (zh) * | 2023-05-04 | 2024-08-23 | 鲁西催化剂有限公司 | 一种氧化铝基吸附剂及其制备方法 |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0043562A1 (en) * | 1980-07-07 | 1982-01-13 | Union Carbide Corporation | Crystalline metallophosphate compositions |
| EP0293939A2 (en) * | 1987-06-04 | 1988-12-07 | Union Carbide Corporation | Crystalline aluminophosphate composition |
| US20030129128A1 (en) * | 1999-11-18 | 2003-07-10 | Strohmaier Karl G | Method for the synthesis of molecular sieves |
| CN105776247A (zh) * | 2014-12-17 | 2016-07-20 | 中国科学院大连化学物理研究所 | 一种aei型金属磷酸铝分子筛的离子热合成方法 |
| CN106276943A (zh) * | 2016-08-08 | 2017-01-04 | 河南师范大学 | 一种固相合成有序多级孔磷酸铝及金属掺杂磷酸铝分子筛的方法 |
Family Cites Families (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4440871A (en) | 1982-07-26 | 1984-04-03 | Union Carbide Corporation | Crystalline silicoaluminophosphates |
| US4752651A (en) | 1986-06-16 | 1988-06-21 | Union Carbide Corporation | Production of light olefins |
| US4851204A (en) * | 1987-06-04 | 1989-07-25 | Uop | Crystalline aluminophosphate composition |
| RU2021975C1 (ru) | 1987-08-28 | 1994-10-30 | Дзе Дау Кемикал Компани | Кристаллическая алюмофосфатная композиция и способ ее получения |
| DD299288A5 (de) | 1989-07-07 | 1992-04-09 | ���������������@����@�������������@�K@����������K@��������@�K@���@�K@���@M�����]k�� | Verfahren zur herstellung eines kristallinen molekularsiebes |
| NO310106B1 (no) | 1996-03-13 | 2001-05-21 | Norsk Hydro As | Mikroporöse, krystallinske metallofosfatforbindelser, en fremgangsmåte for fremstilling og anvendelse derav |
| US6951830B2 (en) | 2003-08-05 | 2005-10-04 | Exxonmobil Chemical Patents Inc. | Molecular sieve catalyst compositions, their production and use in conversion processes |
| US6835363B1 (en) * | 2003-08-06 | 2004-12-28 | Exxonmobil Chemical Patents Inc. | Synthesis of molecular sieves of CHA framework type |
| US20060147364A1 (en) | 2004-12-30 | 2006-07-06 | Council Of Scientific And Industrial Research | Process for synthesising porous crystalline aluminophosphate molecular sieves |
| CN102092738B (zh) | 2011-03-30 | 2012-03-21 | 南开大学 | 水热法无氟合成AlPO4-34 |
| CN103204484B (zh) | 2013-04-09 | 2014-12-17 | 南开大学 | 一种有十元环和八元环交叉孔道的微孔亚磷酸铝及制备方法 |
| CN104445244B (zh) | 2014-11-28 | 2017-02-22 | 天津神能科技有限公司 | 一种磷酸铝分子筛AlPO4-34及其无氟制备方法 |
| CN104944434B (zh) * | 2015-05-19 | 2017-06-16 | 郑州大学 | 一种空心afi型分子筛及其制备方法 |
| SG11201804012WA (en) | 2015-12-04 | 2018-06-28 | Exxonmobil Res & Eng Co | Emm-28, a novel synthetic crystalline material, its preparation and use |
| CN107777701B (zh) * | 2016-08-30 | 2020-06-09 | 中国石油化工股份有限公司 | Scm-12分子筛及其制备方法 |
-
2018
- 2018-10-25 CN CN201811250883.6A patent/CN111099603B/zh active Active
-
2019
- 2019-09-19 JP JP2021523060A patent/JP7373561B2/ja active Active
- 2019-09-19 CA CA3117388A patent/CA3117388C/en active Active
- 2019-09-19 US US17/288,891 patent/US11819834B2/en active Active
- 2019-09-19 EP EP19876975.4A patent/EP3872032A4/en active Pending
- 2019-09-19 WO PCT/CN2019/106628 patent/WO2020082944A1/zh not_active Ceased
- 2019-09-19 KR KR1020217015407A patent/KR102709740B1/ko active Active
- 2019-09-19 SG SG11202104152VA patent/SG11202104152VA/en unknown
- 2019-09-20 TW TW108133991A patent/TWI819099B/zh active
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0043562A1 (en) * | 1980-07-07 | 1982-01-13 | Union Carbide Corporation | Crystalline metallophosphate compositions |
| EP0293939A2 (en) * | 1987-06-04 | 1988-12-07 | Union Carbide Corporation | Crystalline aluminophosphate composition |
| US20030129128A1 (en) * | 1999-11-18 | 2003-07-10 | Strohmaier Karl G | Method for the synthesis of molecular sieves |
| CN105776247A (zh) * | 2014-12-17 | 2016-07-20 | 中国科学院大连化学物理研究所 | 一种aei型金属磷酸铝分子筛的离子热合成方法 |
| CN106276943A (zh) * | 2016-08-08 | 2017-01-04 | 河南师范大学 | 一种固相合成有序多级孔磷酸铝及金属掺杂磷酸铝分子筛的方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| CA3117388C (en) | 2024-01-02 |
| WO2020082944A1 (zh) | 2020-04-30 |
| TWI819099B (zh) | 2023-10-21 |
| EP3872032A1 (en) | 2021-09-01 |
| EP3872032A4 (en) | 2022-08-03 |
| TW202028117A (zh) | 2020-08-01 |
| US20220001365A1 (en) | 2022-01-06 |
| SG11202104152VA (en) | 2021-05-28 |
| KR102709740B1 (ko) | 2024-09-24 |
| CN111099603B (zh) | 2022-12-09 |
| JP2022505968A (ja) | 2022-01-14 |
| US11819834B2 (en) | 2023-11-21 |
| JP7373561B2 (ja) | 2023-11-02 |
| CA3117388A1 (en) | 2020-04-30 |
| CN111099603A (zh) | 2020-05-05 |
| BR112021007662A2 (pt) | 2021-07-27 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Prodinger et al. | Sub-micron Cu/SSZ-13: Synthesis and application as selective catalytic reduction (SCR) catalysts | |
| KR102709740B1 (ko) | Scm-18 알루미노포스페이트 분자체, 이의 제조 방법 및 용도 | |
| JP5762816B2 (ja) | 8員環細孔開口構造を有するモレキュラーシーブまたはゼオライトを含んで成る新規マイクロポーラス結晶性物質およびその製法およびその使用 | |
| JP3371134B2 (ja) | 微孔質結晶シリコ−アルミノ−ホスフェートおよびその製造方法 | |
| JP4990145B2 (ja) | アルミノホスフェートモレキュラーシーブ、その合成及び使用 | |
| CN103482646A (zh) | 具有rho骨架结构的金属磷酸硅铝分子筛及其制备方法 | |
| WO2020252447A9 (en) | Chabazite zeolite synthesis with organic templates | |
| CN103864096B (zh) | 一种sapo-35分子筛及其合成方法 | |
| Wang et al. | Hollow nanocrystals of silicoaluminophosphate molecular sieves synthesized by an aminothermal co-templating strategy | |
| WO2018152829A1 (zh) | Cu-SAPO分子筛、合成方法及其催化应用 | |
| JP4729484B2 (ja) | Emm−3:新規結晶質微細孔物質 | |
| Zhou et al. | One-pot synthesis, characterization and ammonia-selective catalytic reduction performance of MnSAPO-18 molecular sieves | |
| Sanchez-Sanchez et al. | The extremely high specificity of N-methyldicyclohexylamine for the production of the large-pore microporous AFI material | |
| KR102715659B1 (ko) | 실리콘-포스포-알루미늄 분자체, 이의 제조 방법 및 용도 | |
| RU2811599C2 (ru) | Алюмофосфатное молекулярное сито scm-18, его получение и применение | |
| RU2811839C2 (ru) | Силикоалюмофосфатное молекулярное сито, его получение и применение | |
| BR112021007662B1 (pt) | Peneira molecular de aluminofosfato scm-18, método para preparação da mesma, composição de peneira molecular e seus usos | |
| CN108502900B (zh) | Cu-SAPO分子筛、合成方法及其催化应用 | |
| BR112021007627B1 (pt) | Peneira molecular de silicoaluminofosfato, seu método de preparação, composição de peneira molecular e uso das mesmas | |
| Pai et al. | Synthesis and characterization of cobalt substituted aluminophosphate molecular sieve: Co-SSZ-51 under microwave-hydrothermal conditions | |
| CN108502899B (zh) | Cu-SAPO分子筛、其合成方法及其在脱硝反应中的应用 | |
| Venkatathri | Synthesis of high silica content silicoaluminophosphate-5 (SAPO-5) from nonaqueous medium using hexamethyleneimine template | |
| Dumitriu et al. | Advances in the synthesis of pure aluminophosphate molecular sieves and derivatives |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0105 | International application |
Patent event date: 20210521 Patent event code: PA01051R01D Comment text: International Patent Application |
|
| PG1501 | Laying open of application | ||
| A201 | Request for examination | ||
| PA0201 | Request for examination |
Patent event code: PA02012R01D Patent event date: 20220317 Comment text: Request for Examination of Application |
|
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
Comment text: Notification of reason for refusal Patent event date: 20240124 Patent event code: PE09021S01D |
|
| PE0701 | Decision of registration |
Patent event code: PE07011S01D Comment text: Decision to Grant Registration Patent event date: 20240704 |
|
| GRNT | Written decision to grant | ||
| PR0701 | Registration of establishment |
Comment text: Registration of Establishment Patent event date: 20240920 Patent event code: PR07011E01D |
|
| PR1002 | Payment of registration fee |
Payment date: 20240920 End annual number: 3 Start annual number: 1 |
|
| PG1601 | Publication of registration |