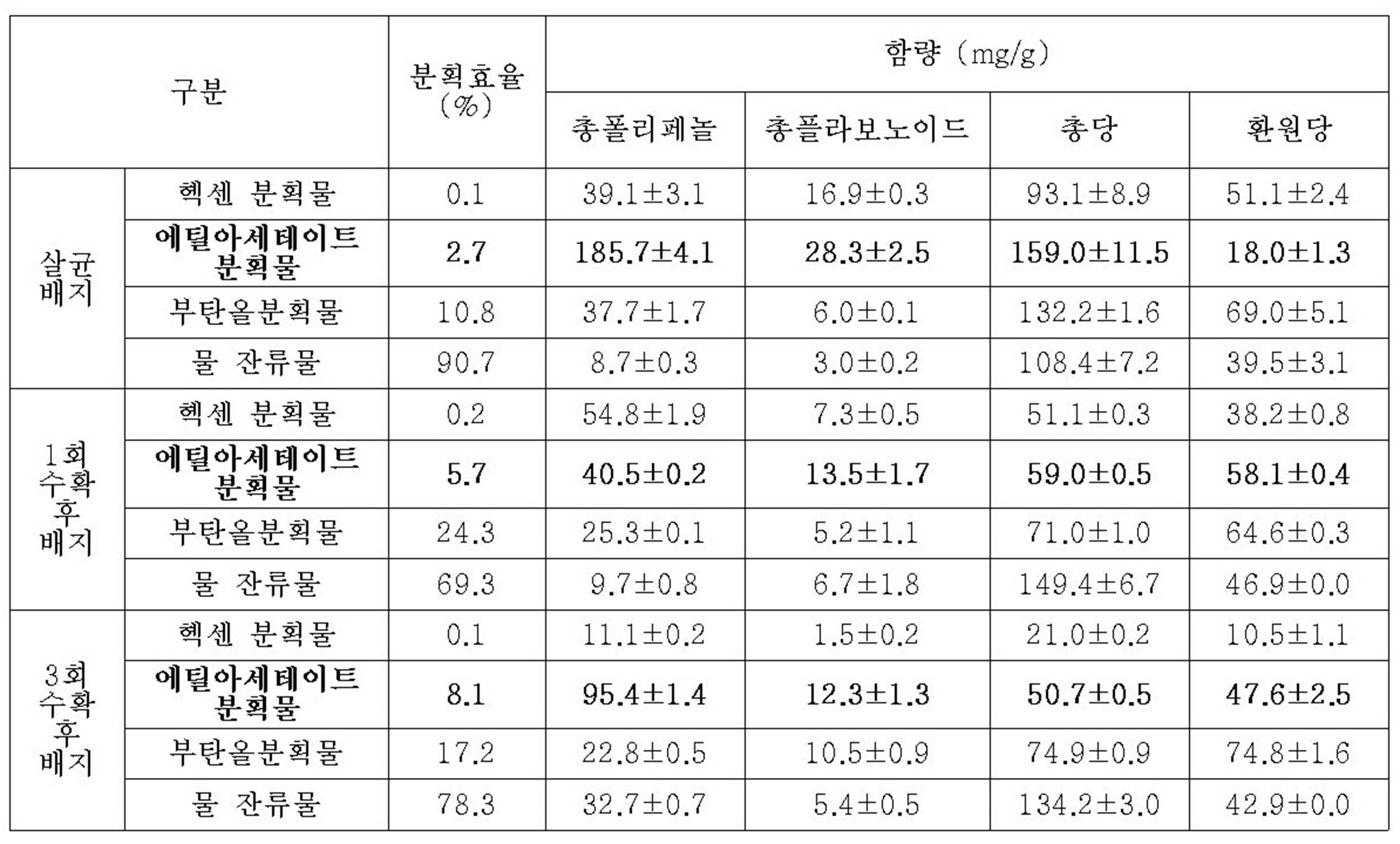
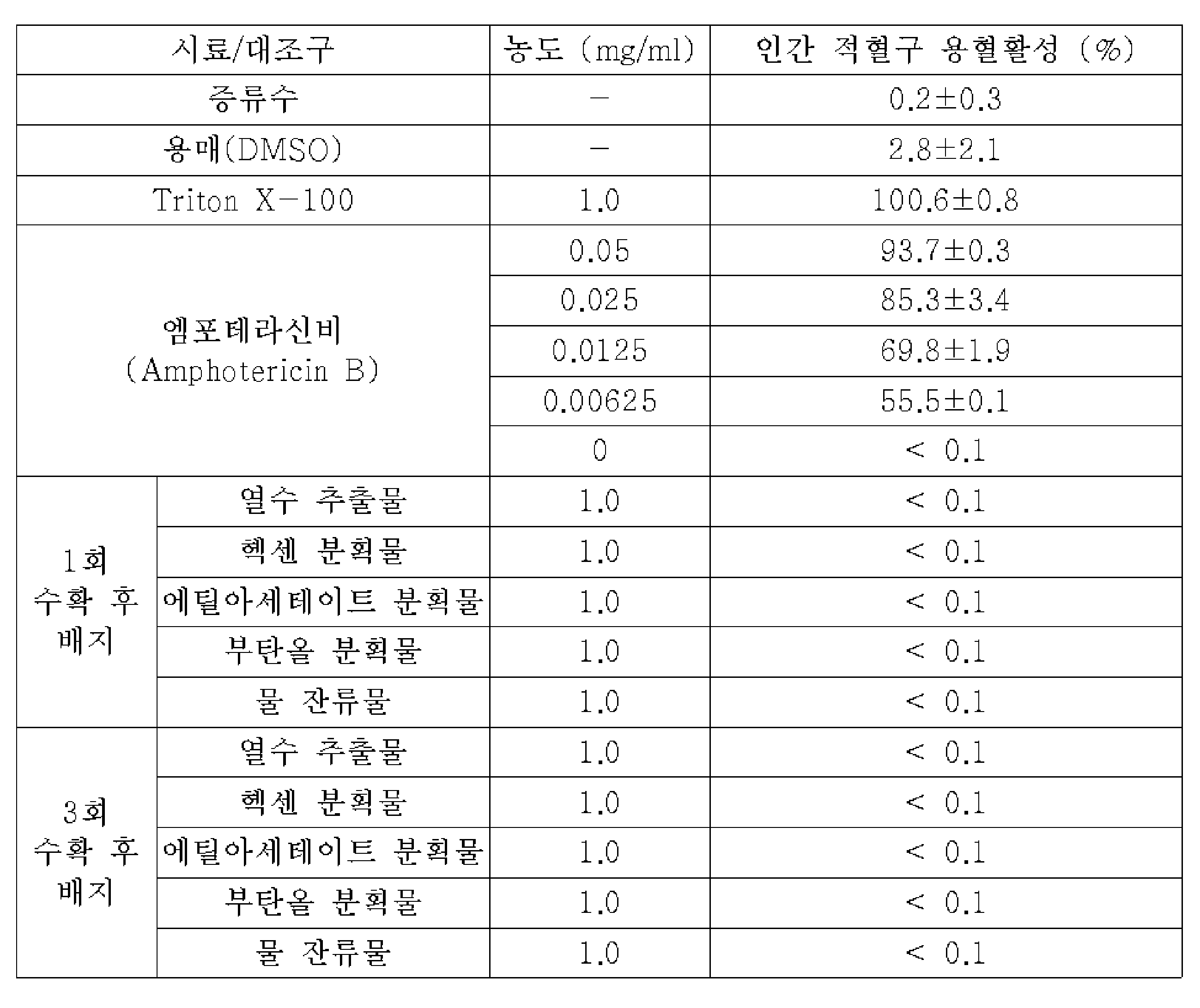
KR20230077110A - Antimicrobial composition comprising the extract of spent mushroom substrate for lentinus edodes - Google Patents
Antimicrobial composition comprising the extract of spent mushroom substrate for lentinus edodes Download PDFInfo
- Publication number
- KR20230077110A KR20230077110A KR1020210164021A KR20210164021A KR20230077110A KR 20230077110 A KR20230077110 A KR 20230077110A KR 1020210164021 A KR1020210164021 A KR 1020210164021A KR 20210164021 A KR20210164021 A KR 20210164021A KR 20230077110 A KR20230077110 A KR 20230077110A
- Authority
- KR
- South Korea
- Prior art keywords
- mushroom
- medium
- shiitake
- antibacterial
- waste
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 235000001674 Agaricus brunnescens Nutrition 0.000 title claims abstract description 170
- 240000000599 Lentinula edodes Species 0.000 title claims abstract description 131
- 239000000284 extract Substances 0.000 title claims abstract description 86
- 239000000203 mixture Substances 0.000 title claims abstract description 81
- 235000001715 Lentinula edodes Nutrition 0.000 title claims abstract description 13
- 239000000758 substrate Substances 0.000 title claims abstract description 6
- 230000000845 anti-microbial effect Effects 0.000 title claims description 13
- 238000003306 harvesting Methods 0.000 claims abstract description 119
- 230000000844 anti-bacterial effect Effects 0.000 claims abstract description 77
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims abstract description 66
- 239000002537 cosmetic Substances 0.000 claims abstract description 18
- 241001465754 Metazoa Species 0.000 claims abstract description 17
- 230000036541 health Effects 0.000 claims abstract description 17
- 239000008194 pharmaceutical composition Substances 0.000 claims abstract description 16
- 239000003599 detergent Substances 0.000 claims abstract description 14
- 235000013376 functional food Nutrition 0.000 claims abstract description 13
- 239000002699 waste material Substances 0.000 claims description 138
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 claims description 27
- 238000000034 method Methods 0.000 claims description 8
- 235000013373 food additive Nutrition 0.000 claims description 4
- 239000002778 food additive Substances 0.000 claims description 4
- 235000019730 animal feed additive Nutrition 0.000 claims description 3
- 239000000490 cosmetic additive Substances 0.000 claims description 3
- 239000002038 ethyl acetate fraction Substances 0.000 abstract description 24
- 235000013305 food Nutrition 0.000 abstract description 24
- 239000000843 powder Substances 0.000 abstract description 21
- 239000000654 additive Substances 0.000 abstract description 17
- 239000004480 active ingredient Substances 0.000 abstract description 16
- 230000000843 anti-fungal effect Effects 0.000 abstract description 13
- 230000000694 effects Effects 0.000 abstract description 13
- 239000006227 byproduct Substances 0.000 abstract description 7
- 239000012459 cleaning agent Substances 0.000 abstract description 6
- 238000012258 culturing Methods 0.000 abstract description 5
- 239000003826 tablet Substances 0.000 abstract description 4
- 239000006071 cream Substances 0.000 abstract description 3
- 239000007788 liquid Substances 0.000 abstract description 3
- 230000002378 acidificating effect Effects 0.000 abstract description 2
- 244000144972 livestock Species 0.000 abstract description 2
- 239000006187 pill Substances 0.000 abstract description 2
- 239000002609 medium Substances 0.000 description 197
- 230000001954 sterilising effect Effects 0.000 description 37
- 238000004659 sterilization and disinfection Methods 0.000 description 37
- 238000004519 manufacturing process Methods 0.000 description 24
- 241000282414 Homo sapiens Species 0.000 description 17
- 238000004458 analytical method Methods 0.000 description 17
- 230000002949 hemolytic effect Effects 0.000 description 13
- 235000000346 sugar Nutrition 0.000 description 12
- 210000003743 erythrocyte Anatomy 0.000 description 10
- 239000003960 organic solvent Substances 0.000 description 10
- LIKMAJRDDDTEIG-UHFFFAOYSA-N 1-hexene Chemical compound CCCCC=C LIKMAJRDDDTEIG-UHFFFAOYSA-N 0.000 description 9
- 239000003242 anti bacterial agent Substances 0.000 description 9
- 239000000047 product Substances 0.000 description 9
- 241000894006 Bacteria Species 0.000 description 8
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 8
- LRHPLDYGYMQRHN-UHFFFAOYSA-N N-Butanol Chemical compound CCCCO LRHPLDYGYMQRHN-UHFFFAOYSA-N 0.000 description 8
- 239000003814 drug Substances 0.000 description 8
- 208000015181 infectious disease Diseases 0.000 description 8
- 235000015097 nutrients Nutrition 0.000 description 8
- 239000000796 flavoring agent Substances 0.000 description 7
- 150000008442 polyphenolic compounds Chemical class 0.000 description 7
- 235000013824 polyphenols Nutrition 0.000 description 7
- 239000003755 preservative agent Substances 0.000 description 7
- 238000011160 research Methods 0.000 description 7
- 239000000523 sample Substances 0.000 description 7
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 6
- 241000233866 Fungi Species 0.000 description 6
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 6
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 description 6
- 239000002034 butanolic fraction Substances 0.000 description 6
- 238000000605 extraction Methods 0.000 description 6
- 229930003935 flavonoid Natural products 0.000 description 6
- 150000002215 flavonoids Chemical class 0.000 description 6
- 235000017173 flavonoids Nutrition 0.000 description 6
- 238000000227 grinding Methods 0.000 description 6
- 238000002360 preparation method Methods 0.000 description 6
- 230000002829 reductive effect Effects 0.000 description 6
- 108090000790 Enzymes Proteins 0.000 description 5
- 102000004190 Enzymes Human genes 0.000 description 5
- 241000192125 Firmicutes Species 0.000 description 5
- 235000013361 beverage Nutrition 0.000 description 5
- 201000010099 disease Diseases 0.000 description 5
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 5
- 229940088598 enzyme Drugs 0.000 description 5
- 238000011156 evaluation Methods 0.000 description 5
- 230000012010 growth Effects 0.000 description 5
- 230000001766 physiological effect Effects 0.000 description 5
- 229920001282 polysaccharide Polymers 0.000 description 5
- 239000005017 polysaccharide Substances 0.000 description 5
- 150000004804 polysaccharides Chemical class 0.000 description 5
- 241000196324 Embryophyta Species 0.000 description 4
- 239000004599 antimicrobial Substances 0.000 description 4
- 150000001720 carbohydrates Chemical class 0.000 description 4
- 235000014633 carbohydrates Nutrition 0.000 description 4
- 238000011161 development Methods 0.000 description 4
- 229940079593 drug Drugs 0.000 description 4
- 235000019634 flavors Nutrition 0.000 description 4
- 239000004615 ingredient Substances 0.000 description 4
- 230000002401 inhibitory effect Effects 0.000 description 4
- 230000002265 prevention Effects 0.000 description 4
- 239000000126 substance Substances 0.000 description 4
- 241000251468 Actinopterygii Species 0.000 description 3
- 241000222122 Candida albicans Species 0.000 description 3
- 241000238557 Decapoda Species 0.000 description 3
- 241000588724 Escherichia coli Species 0.000 description 3
- 206010017533 Fungal infection Diseases 0.000 description 3
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 3
- 241000124008 Mammalia Species 0.000 description 3
- 208000031888 Mycoses Diseases 0.000 description 3
- 240000007594 Oryza sativa Species 0.000 description 3
- 235000007164 Oryza sativa Nutrition 0.000 description 3
- 240000001462 Pleurotus ostreatus Species 0.000 description 3
- 235000001603 Pleurotus ostreatus Nutrition 0.000 description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- 244000269722 Thea sinensis Species 0.000 description 3
- 239000002253 acid Substances 0.000 description 3
- 230000000996 additive effect Effects 0.000 description 3
- 229940088710 antibiotic agent Drugs 0.000 description 3
- 239000003795 chemical substances by application Substances 0.000 description 3
- 238000007796 conventional method Methods 0.000 description 3
- MTHSVFCYNBDYFN-UHFFFAOYSA-N diethylene glycol Chemical compound OCCOCCO MTHSVFCYNBDYFN-UHFFFAOYSA-N 0.000 description 3
- 235000013355 food flavoring agent Nutrition 0.000 description 3
- 238000005194 fractionation Methods 0.000 description 3
- 239000008187 granular material Substances 0.000 description 3
- 239000007924 injection Substances 0.000 description 3
- 238000002347 injection Methods 0.000 description 3
- 238000011081 inoculation Methods 0.000 description 3
- 150000002632 lipids Chemical class 0.000 description 3
- 244000000010 microbial pathogen Species 0.000 description 3
- 235000016709 nutrition Nutrition 0.000 description 3
- -1 pH adjusters Substances 0.000 description 3
- 238000004806 packaging method and process Methods 0.000 description 3
- 239000000049 pigment Substances 0.000 description 3
- 230000003405 preventing effect Effects 0.000 description 3
- 235000009566 rice Nutrition 0.000 description 3
- 239000012488 sample solution Substances 0.000 description 3
- 239000002904 solvent Substances 0.000 description 3
- 238000003860 storage Methods 0.000 description 3
- 239000004094 surface-active agent Substances 0.000 description 3
- 229940124597 therapeutic agent Drugs 0.000 description 3
- MYPYJXKWCTUITO-LYRMYLQWSA-N vancomycin Chemical compound O([C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@H]1OC1=C2C=C3C=C1OC1=CC=C(C=C1Cl)[C@@H](O)[C@H](C(N[C@@H](CC(N)=O)C(=O)N[C@H]3C(=O)N[C@H]1C(=O)N[C@H](C(N[C@@H](C3=CC(O)=CC(O)=C3C=3C(O)=CC=C1C=3)C(O)=O)=O)[C@H](O)C1=CC=C(C(=C1)Cl)O2)=O)NC(=O)[C@@H](CC(C)C)NC)[C@H]1C[C@](C)(N)[C@H](O)[C@H](C)O1 MYPYJXKWCTUITO-LYRMYLQWSA-N 0.000 description 3
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 2
- 241000271566 Aves Species 0.000 description 2
- 208000035143 Bacterial infection Diseases 0.000 description 2
- 241000283690 Bos taurus Species 0.000 description 2
- 235000002566 Capsicum Nutrition 0.000 description 2
- 235000009091 Cordyline terminalis Nutrition 0.000 description 2
- 244000289527 Cordyline terminalis Species 0.000 description 2
- 241000238424 Crustacea Species 0.000 description 2
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 2
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 2
- 108010082495 Dietary Plant Proteins Proteins 0.000 description 2
- 241000258955 Echinodermata Species 0.000 description 2
- 241000289667 Erinaceus Species 0.000 description 2
- 239000004386 Erythritol Substances 0.000 description 2
- UNXHWFMMPAWVPI-UHFFFAOYSA-N Erythritol Natural products OCC(O)C(O)CO UNXHWFMMPAWVPI-UHFFFAOYSA-N 0.000 description 2
- 241000287828 Gallus gallus Species 0.000 description 2
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 2
- 244000068988 Glycine max Species 0.000 description 2
- 235000010469 Glycine max Nutrition 0.000 description 2
- 206010018910 Haemolysis Diseases 0.000 description 2
- 241000237852 Mollusca Species 0.000 description 2
- 239000006002 Pepper Substances 0.000 description 2
- 241000286209 Phasianidae Species 0.000 description 2
- 235000016761 Piper aduncum Nutrition 0.000 description 2
- 240000003889 Piper guineense Species 0.000 description 2
- 235000017804 Piper guineense Nutrition 0.000 description 2
- 235000008184 Piper nigrum Nutrition 0.000 description 2
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 2
- 229930006000 Sucrose Natural products 0.000 description 2
- 239000013504 Triton X-100 Substances 0.000 description 2
- 229920004890 Triton X-100 Polymers 0.000 description 2
- 108010059993 Vancomycin Proteins 0.000 description 2
- TVXBFESIOXBWNM-UHFFFAOYSA-N Xylitol Natural products OCCC(O)C(O)C(O)CCO TVXBFESIOXBWNM-UHFFFAOYSA-N 0.000 description 2
- 238000002835 absorbance Methods 0.000 description 2
- 235000010443 alginic acid Nutrition 0.000 description 2
- 229920000615 alginic acid Polymers 0.000 description 2
- 229960000723 ampicillin Drugs 0.000 description 2
- AVKUERGKIZMTKX-NJBDSQKTSA-N ampicillin Chemical compound C1([C@@H](N)C(=O)N[C@H]2[C@H]3SC([C@@H](N3C2=O)C(O)=O)(C)C)=CC=CC=C1 AVKUERGKIZMTKX-NJBDSQKTSA-N 0.000 description 2
- 235000021120 animal protein Nutrition 0.000 description 2
- 229940121375 antifungal agent Drugs 0.000 description 2
- 239000003429 antifungal agent Substances 0.000 description 2
- 230000003078 antioxidant effect Effects 0.000 description 2
- 208000022362 bacterial infectious disease Diseases 0.000 description 2
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 2
- 210000004369 blood Anatomy 0.000 description 2
- 239000008280 blood Substances 0.000 description 2
- 239000002775 capsule Substances 0.000 description 2
- 239000000969 carrier Substances 0.000 description 2
- 239000001913 cellulose Substances 0.000 description 2
- 229920002678 cellulose Polymers 0.000 description 2
- 235000010980 cellulose Nutrition 0.000 description 2
- 235000013339 cereals Nutrition 0.000 description 2
- 238000006243 chemical reaction Methods 0.000 description 2
- 239000003153 chemical reaction reagent Substances 0.000 description 2
- 235000013330 chicken meat Nutrition 0.000 description 2
- 235000013365 dairy product Nutrition 0.000 description 2
- 238000000354 decomposition reaction Methods 0.000 description 2
- 230000003247 decreasing effect Effects 0.000 description 2
- 239000008121 dextrose Substances 0.000 description 2
- 235000015872 dietary supplement Nutrition 0.000 description 2
- 239000003085 diluting agent Substances 0.000 description 2
- 238000001035 drying Methods 0.000 description 2
- 239000003623 enhancer Substances 0.000 description 2
- UNXHWFMMPAWVPI-ZXZARUISSA-N erythritol Chemical compound OC[C@H](O)[C@H](O)CO UNXHWFMMPAWVPI-ZXZARUISSA-N 0.000 description 2
- 235000019414 erythritol Nutrition 0.000 description 2
- 229940009714 erythritol Drugs 0.000 description 2
- 238000002474 experimental method Methods 0.000 description 2
- 238000001914 filtration Methods 0.000 description 2
- 235000015203 fruit juice Nutrition 0.000 description 2
- 239000000446 fuel Substances 0.000 description 2
- 230000009036 growth inhibition Effects 0.000 description 2
- 235000013402 health food Nutrition 0.000 description 2
- 230000008588 hemolysis Effects 0.000 description 2
- 238000001990 intravenous administration Methods 0.000 description 2
- 230000007774 longterm Effects 0.000 description 2
- 239000006210 lotion Substances 0.000 description 2
- 210000004072 lung Anatomy 0.000 description 2
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 2
- HEBKCHPVOIAQTA-UHFFFAOYSA-N meso ribitol Natural products OCC(O)C(O)C(O)CO HEBKCHPVOIAQTA-UHFFFAOYSA-N 0.000 description 2
- 244000005700 microbiome Species 0.000 description 2
- 235000006408 oxalic acid Nutrition 0.000 description 2
- 230000001717 pathogenic effect Effects 0.000 description 2
- 230000007170 pathology Effects 0.000 description 2
- 239000008188 pellet Substances 0.000 description 2
- 239000000546 pharmaceutical excipient Substances 0.000 description 2
- 150000003839 salts Chemical class 0.000 description 2
- 239000000243 solution Substances 0.000 description 2
- 239000000600 sorbitol Substances 0.000 description 2
- 235000010356 sorbitol Nutrition 0.000 description 2
- 238000007920 subcutaneous administration Methods 0.000 description 2
- 239000005720 sucrose Substances 0.000 description 2
- 210000001519 tissue Anatomy 0.000 description 2
- 230000000699 topical effect Effects 0.000 description 2
- 229960003165 vancomycin Drugs 0.000 description 2
- MYPYJXKWCTUITO-UHFFFAOYSA-N vancomycin Natural products O1C(C(=C2)Cl)=CC=C2C(O)C(C(NC(C2=CC(O)=CC(O)=C2C=2C(O)=CC=C3C=2)C(O)=O)=O)NC(=O)C3NC(=O)C2NC(=O)C(CC(N)=O)NC(=O)C(NC(=O)C(CC(C)C)NC)C(O)C(C=C3Cl)=CC=C3OC3=CC2=CC1=C3OC1OC(CO)C(O)C(O)C1OC1CC(C)(N)C(O)C(C)O1 MYPYJXKWCTUITO-UHFFFAOYSA-N 0.000 description 2
- 239000011782 vitamin Substances 0.000 description 2
- 229930003231 vitamin Natural products 0.000 description 2
- 235000013343 vitamin Nutrition 0.000 description 2
- 229940088594 vitamin Drugs 0.000 description 2
- 235000010447 xylitol Nutrition 0.000 description 2
- 239000000811 xylitol Substances 0.000 description 2
- HEBKCHPVOIAQTA-SCDXWVJYSA-N xylitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)CO HEBKCHPVOIAQTA-SCDXWVJYSA-N 0.000 description 2
- 229960002675 xylitol Drugs 0.000 description 2
- OILXMJHPFNGGTO-UHFFFAOYSA-N (22E)-(24xi)-24-methylcholesta-5,22-dien-3beta-ol Natural products C1C=C2CC(O)CCC2(C)C2C1C1CCC(C(C)C=CC(C)C(C)C)C1(C)CC2 OILXMJHPFNGGTO-UHFFFAOYSA-N 0.000 description 1
- RQOCXCFLRBRBCS-UHFFFAOYSA-N (22E)-cholesta-5,7,22-trien-3beta-ol Natural products C1C(O)CCC2(C)C(CCC3(C(C(C)C=CCC(C)C)CCC33)C)C3=CC=C21 RQOCXCFLRBRBCS-UHFFFAOYSA-N 0.000 description 1
- FYGDTMLNYKFZSV-URKRLVJHSA-N (2s,3r,4s,5s,6r)-2-[(2r,4r,5r,6s)-4,5-dihydroxy-2-(hydroxymethyl)-6-[(2r,4r,5r,6s)-4,5,6-trihydroxy-2-(hydroxymethyl)oxan-3-yl]oxyoxan-3-yl]oxy-6-(hydroxymethyl)oxane-3,4,5-triol Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@H]1OC1[C@@H](CO)O[C@@H](OC2[C@H](O[C@H](O)[C@H](O)[C@H]2O)CO)[C@H](O)[C@H]1O FYGDTMLNYKFZSV-URKRLVJHSA-N 0.000 description 1
- TUSDEZXZIZRFGC-UHFFFAOYSA-N 1-O-galloyl-3,6-(R)-HHDP-beta-D-glucose Natural products OC1C(O2)COC(=O)C3=CC(O)=C(O)C(O)=C3C3=C(O)C(O)=C(O)C=C3C(=O)OC1C(O)C2OC(=O)C1=CC(O)=C(O)C(O)=C1 TUSDEZXZIZRFGC-UHFFFAOYSA-N 0.000 description 1
- OWEGMIWEEQEYGQ-UHFFFAOYSA-N 100676-05-9 Natural products OC1C(O)C(O)C(CO)OC1OCC1C(O)C(O)C(O)C(OC2C(OC(O)C(O)C2O)CO)O1 OWEGMIWEEQEYGQ-UHFFFAOYSA-N 0.000 description 1
- OQMZNAMGEHIHNN-UHFFFAOYSA-N 7-Dehydrostigmasterol Natural products C1C(O)CCC2(C)C(CCC3(C(C(C)C=CC(CC)C(C)C)CCC33)C)C3=CC=C21 OQMZNAMGEHIHNN-UHFFFAOYSA-N 0.000 description 1
- 244000215068 Acacia senegal Species 0.000 description 1
- 235000006491 Acacia senegal Nutrition 0.000 description 1
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 1
- 241000222518 Agaricus Species 0.000 description 1
- 244000251953 Agaricus brunnescens Species 0.000 description 1
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 1
- 241000473391 Archosargus rhomboidalis Species 0.000 description 1
- 206010003210 Arteriosclerosis Diseases 0.000 description 1
- 241000238421 Arthropoda Species 0.000 description 1
- 108010011485 Aspartame Proteins 0.000 description 1
- 206010003645 Atopy Diseases 0.000 description 1
- 235000007319 Avena orientalis Nutrition 0.000 description 1
- 244000075850 Avena orientalis Species 0.000 description 1
- 244000063299 Bacillus subtilis Species 0.000 description 1
- 235000014469 Bacillus subtilis Nutrition 0.000 description 1
- 229920002498 Beta-glucan Polymers 0.000 description 1
- JMGZEFIQIZZSBH-UHFFFAOYSA-N Bioquercetin Natural products CC1OC(OCC(O)C2OC(OC3=C(Oc4cc(O)cc(O)c4C3=O)c5ccc(O)c(O)c5)C(O)C2O)C(O)C(O)C1O JMGZEFIQIZZSBH-UHFFFAOYSA-N 0.000 description 1
- 239000005996 Blood meal Substances 0.000 description 1
- 208000014644 Brain disease Diseases 0.000 description 1
- 240000002791 Brassica napus Species 0.000 description 1
- 235000004977 Brassica sinapistrum Nutrition 0.000 description 1
- 206010007134 Candida infections Diseases 0.000 description 1
- 241000283707 Capra Species 0.000 description 1
- 241000700199 Cavia porcellus Species 0.000 description 1
- 108010059892 Cellulase Proteins 0.000 description 1
- 241000238366 Cephalopoda Species 0.000 description 1
- 241000282693 Cercopithecidae Species 0.000 description 1
- 229920001661 Chitosan Polymers 0.000 description 1
- 241000938605 Crocodylia Species 0.000 description 1
- 206010011409 Cross infection Diseases 0.000 description 1
- 235000019750 Crude protein Nutrition 0.000 description 1
- 229920000858 Cyclodextrin Polymers 0.000 description 1
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 1
- ZZZCUOFIHGPKAK-UHFFFAOYSA-N D-erythro-ascorbic acid Natural products OCC1OC(=O)C(O)=C1O ZZZCUOFIHGPKAK-UHFFFAOYSA-N 0.000 description 1
- 206010012442 Dermatitis contact Diseases 0.000 description 1
- 229920001353 Dextrin Polymers 0.000 description 1
- 239000004375 Dextrin Substances 0.000 description 1
- 208000035240 Disease Resistance Diseases 0.000 description 1
- 241000257465 Echinoidea Species 0.000 description 1
- 241000194033 Enterococcus Species 0.000 description 1
- 241000283073 Equus caballus Species 0.000 description 1
- DNVPQKQSNYMLRS-NXVQYWJNSA-N Ergosterol Natural products CC(C)[C@@H](C)C=C[C@H](C)[C@H]1CC[C@H]2C3=CC=C4C[C@@H](O)CC[C@]4(C)[C@@H]3CC[C@]12C DNVPQKQSNYMLRS-NXVQYWJNSA-N 0.000 description 1
- 239000001263 FEMA 3042 Substances 0.000 description 1
- 239000001512 FEMA 4601 Substances 0.000 description 1
- 241000282326 Felis catus Species 0.000 description 1
- 235000019733 Fish meal Nutrition 0.000 description 1
- 229930091371 Fructose Natural products 0.000 description 1
- 239000005715 Fructose Substances 0.000 description 1
- RFSUNEUAIZKAJO-ARQDHWQXSA-N Fructose Chemical compound OC[C@H]1O[C@](O)(CO)[C@@H](O)[C@@H]1O RFSUNEUAIZKAJO-ARQDHWQXSA-N 0.000 description 1
- 241000276438 Gadus morhua Species 0.000 description 1
- 208000007882 Gastritis Diseases 0.000 description 1
- 208000018522 Gastrointestinal disease Diseases 0.000 description 1
- 241000237858 Gastropoda Species 0.000 description 1
- 108010010803 Gelatin Proteins 0.000 description 1
- 239000004378 Glycyrrhizin Substances 0.000 description 1
- 229920000084 Gum arabic Polymers 0.000 description 1
- 244000020551 Helianthus annuus Species 0.000 description 1
- 235000003222 Helianthus annuus Nutrition 0.000 description 1
- 102000001554 Hemoglobins Human genes 0.000 description 1
- 108010054147 Hemoglobins Proteins 0.000 description 1
- 241000251511 Holothuroidea Species 0.000 description 1
- 241000282412 Homo Species 0.000 description 1
- 240000005979 Hordeum vulgare Species 0.000 description 1
- 235000007340 Hordeum vulgare Nutrition 0.000 description 1
- 206010020751 Hypersensitivity Diseases 0.000 description 1
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 1
- 241000186779 Listeria monocytogenes Species 0.000 description 1
- 208000019693 Lung disease Diseases 0.000 description 1
- 235000007688 Lycopersicon esculentum Nutrition 0.000 description 1
- GUBGYTABKSRVRQ-PICCSMPSSA-N Maltose Natural products O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@@H](CO)OC(O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-PICCSMPSSA-N 0.000 description 1
- 229930195725 Mannitol Natural products 0.000 description 1
- 206010027304 Menopausal symptoms Diseases 0.000 description 1
- BYBLEWFAAKGYCD-UHFFFAOYSA-N Miconazole Chemical compound ClC1=CC(Cl)=CC=C1COC(C=1C(=CC(Cl)=CC=1)Cl)CN1C=NC=C1 BYBLEWFAAKGYCD-UHFFFAOYSA-N 0.000 description 1
- 244000294411 Mirabilis expansa Species 0.000 description 1
- 235000015429 Mirabilis expansa Nutrition 0.000 description 1
- 241000699666 Mus <mouse, genus> Species 0.000 description 1
- 206010028980 Neoplasm Diseases 0.000 description 1
- 208000008589 Obesity Diseases 0.000 description 1
- 206010030113 Oedema Diseases 0.000 description 1
- 241000283973 Oryctolagus cuniculus Species 0.000 description 1
- 241001282110 Pagrus major Species 0.000 description 1
- 241001494479 Pecora Species 0.000 description 1
- 229920002230 Pectic acid Polymers 0.000 description 1
- LRBQNJMCXXYXIU-PPKXGCFTSA-N Penta-digallate-beta-D-glucose Natural products OC1=C(O)C(O)=CC(C(=O)OC=2C(=C(O)C=C(C=2)C(=O)OC[C@@H]2[C@H]([C@H](OC(=O)C=3C=C(OC(=O)C=4C=C(O)C(O)=C(O)C=4)C(O)=C(O)C=3)[C@@H](OC(=O)C=3C=C(OC(=O)C=4C=C(O)C(O)=C(O)C=4)C(O)=C(O)C=3)[C@H](OC(=O)C=3C=C(OC(=O)C=4C=C(O)C(O)=C(O)C=4)C(O)=C(O)C=3)O2)OC(=O)C=2C=C(OC(=O)C=3C=C(O)C(O)=C(O)C=3)C(O)=C(O)C=2)O)=C1 LRBQNJMCXXYXIU-PPKXGCFTSA-N 0.000 description 1
- 241000009328 Perro Species 0.000 description 1
- 235000008331 Pinus X rigitaeda Nutrition 0.000 description 1
- 241000018646 Pinus brutia Species 0.000 description 1
- 235000011613 Pinus brutia Nutrition 0.000 description 1
- 241000269908 Platichthys flesus Species 0.000 description 1
- 241000269980 Pleuronectidae Species 0.000 description 1
- 241000222350 Pleurotus Species 0.000 description 1
- 244000252132 Pleurotus eryngii Species 0.000 description 1
- 241001529596 Pontinus kuhlii Species 0.000 description 1
- 241000206607 Porphyra umbilicalis Species 0.000 description 1
- 241000588767 Proteus vulgaris Species 0.000 description 1
- 241000589517 Pseudomonas aeruginosa Species 0.000 description 1
- 241000700159 Rattus Species 0.000 description 1
- HELXLJCILKEWJH-SEAGSNCFSA-N Rebaudioside A Natural products O=C(O[C@H]1[C@@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1)[C@@]1(C)[C@@H]2[C@](C)([C@H]3[C@@]4(CC(=C)[C@@](O[C@H]5[C@H](O[C@H]6[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O6)[C@@H](O[C@H]6[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O6)[C@H](O)[C@@H](CO)O5)(C4)CC3)CC2)CCC1 HELXLJCILKEWJH-SEAGSNCFSA-N 0.000 description 1
- 240000004808 Saccharomyces cerevisiae Species 0.000 description 1
- 235000014680 Saccharomyces cerevisiae Nutrition 0.000 description 1
- 241000293869 Salmonella enterica subsp. enterica serovar Typhimurium Species 0.000 description 1
- 206010040047 Sepsis Diseases 0.000 description 1
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 1
- 240000003768 Solanum lycopersicum Species 0.000 description 1
- 206010041925 Staphylococcal infections Diseases 0.000 description 1
- 241000191940 Staphylococcus Species 0.000 description 1
- 241000191967 Staphylococcus aureus Species 0.000 description 1
- 241000751182 Staphylococcus epidermidis ATCC 12228 Species 0.000 description 1
- 229920002472 Starch Polymers 0.000 description 1
- 241000282887 Suidae Species 0.000 description 1
- 241000282898 Sus scrofa Species 0.000 description 1
- 208000007536 Thrombosis Diseases 0.000 description 1
- 235000021307 Triticum Nutrition 0.000 description 1
- 244000098338 Triticum aestivum Species 0.000 description 1
- 241000700605 Viruses Species 0.000 description 1
- 229930003268 Vitamin C Natural products 0.000 description 1
- 239000005862 Whey Substances 0.000 description 1
- 102000007544 Whey Proteins Human genes 0.000 description 1
- 108010046377 Whey Proteins Proteins 0.000 description 1
- 241000607479 Yersinia pestis Species 0.000 description 1
- 240000008042 Zea mays Species 0.000 description 1
- 235000005824 Zea mays ssp. parviglumis Nutrition 0.000 description 1
- 235000002017 Zea mays subsp mays Nutrition 0.000 description 1
- 230000002159 abnormal effect Effects 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- 235000010489 acacia gum Nutrition 0.000 description 1
- 229920001284 acidic polysaccharide Polymers 0.000 description 1
- 150000004805 acidic polysaccharides Chemical class 0.000 description 1
- 239000011149 active material Substances 0.000 description 1
- 239000013543 active substance Substances 0.000 description 1
- 230000007059 acute toxicity Effects 0.000 description 1
- 231100000403 acute toxicity Toxicity 0.000 description 1
- 239000000443 aerosol Substances 0.000 description 1
- 239000000783 alginic acid Substances 0.000 description 1
- 229960001126 alginic acid Drugs 0.000 description 1
- 150000004781 alginic acids Chemical class 0.000 description 1
- 208000026935 allergic disease Diseases 0.000 description 1
- 230000007815 allergy Effects 0.000 description 1
- 150000001413 amino acids Chemical class 0.000 description 1
- 208000007502 anemia Diseases 0.000 description 1
- 230000001093 anti-cancer Effects 0.000 description 1
- 230000003178 anti-diabetic effect Effects 0.000 description 1
- 230000003110 anti-inflammatory effect Effects 0.000 description 1
- 230000002785 anti-thrombosis Effects 0.000 description 1
- 239000003146 anticoagulant agent Substances 0.000 description 1
- 239000002246 antineoplastic agent Substances 0.000 description 1
- 229940041181 antineoplastic drug Drugs 0.000 description 1
- 208000011775 arteriosclerosis disease Diseases 0.000 description 1
- 239000000605 aspartame Substances 0.000 description 1
- 235000010357 aspartame Nutrition 0.000 description 1
- IAOZJIPTCAWIRG-QWRGUYRKSA-N aspartame Chemical compound OC(=O)C[C@H](N)C(=O)N[C@H](C(=O)OC)CC1=CC=CC=C1 IAOZJIPTCAWIRG-QWRGUYRKSA-N 0.000 description 1
- 229960003438 aspartame Drugs 0.000 description 1
- 230000001580 bacterial effect Effects 0.000 description 1
- 244000052616 bacterial pathogen Species 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- 108010047754 beta-Glucosidase Proteins 0.000 description 1
- 102000006995 beta-Glucosidase Human genes 0.000 description 1
- GUBGYTABKSRVRQ-QUYVBRFLSA-N beta-maltose Chemical compound OC[C@H]1O[C@H](O[C@H]2[C@H](O)[C@@H](O)[C@H](O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@@H]1O GUBGYTABKSRVRQ-QUYVBRFLSA-N 0.000 description 1
- 230000017531 blood circulation Effects 0.000 description 1
- 230000036772 blood pressure Effects 0.000 description 1
- 208000037815 bloodstream infection Diseases 0.000 description 1
- 230000037396 body weight Effects 0.000 description 1
- 230000008468 bone growth Effects 0.000 description 1
- 210000001185 bone marrow Anatomy 0.000 description 1
- 229940036811 bone meal Drugs 0.000 description 1
- 239000002374 bone meal Substances 0.000 description 1
- 239000001506 calcium phosphate Substances 0.000 description 1
- 229910000389 calcium phosphate Inorganic materials 0.000 description 1
- 235000011010 calcium phosphates Nutrition 0.000 description 1
- 239000000378 calcium silicate Substances 0.000 description 1
- 229910052918 calcium silicate Inorganic materials 0.000 description 1
- 235000012241 calcium silicate Nutrition 0.000 description 1
- OYACROKNLOSFPA-UHFFFAOYSA-N calcium;dioxido(oxo)silane Chemical compound [Ca+2].[O-][Si]([O-])=O OYACROKNLOSFPA-UHFFFAOYSA-N 0.000 description 1
- 201000011510 cancer Diseases 0.000 description 1
- 229940095731 candida albicans Drugs 0.000 description 1
- 201000003984 candidiasis Diseases 0.000 description 1
- 125000004432 carbon atom Chemical group C* 0.000 description 1
- 235000014171 carbonated beverage Nutrition 0.000 description 1
- 210000004027 cell Anatomy 0.000 description 1
- 230000010261 cell growth Effects 0.000 description 1
- 229940106157 cellulase Drugs 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 229960005091 chloramphenicol Drugs 0.000 description 1
- WIIZWVCIJKGZOK-RKDXNWHRSA-N chloramphenicol Chemical compound ClC(Cl)C(=O)N[C@H](CO)[C@H](O)C1=CC=C([N+]([O-])=O)C=C1 WIIZWVCIJKGZOK-RKDXNWHRSA-N 0.000 description 1
- 235000020230 cinnamon extract Nutrition 0.000 description 1
- 238000004140 cleaning Methods 0.000 description 1
- 239000000084 colloidal system Substances 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 239000002361 compost Substances 0.000 description 1
- 150000001875 compounds Chemical class 0.000 description 1
- 208000010247 contact dermatitis Diseases 0.000 description 1
- 235000005822 corn Nutrition 0.000 description 1
- 230000003412 degenerative effect Effects 0.000 description 1
- 230000000593 degrading effect Effects 0.000 description 1
- 235000019425 dextrin Nutrition 0.000 description 1
- 206010012601 diabetes mellitus Diseases 0.000 description 1
- 230000029087 digestion Effects 0.000 description 1
- 230000001079 digestive effect Effects 0.000 description 1
- 208000010643 digestive system disease Diseases 0.000 description 1
- 150000002016 disaccharides Chemical class 0.000 description 1
- 238000002828 disc diffusion antibiotic sensitivity testing Methods 0.000 description 1
- 231100000676 disease causative agent Toxicity 0.000 description 1
- 230000006806 disease prevention Effects 0.000 description 1
- 230000002554 disease preventive effect Effects 0.000 description 1
- 239000012153 distilled water Substances 0.000 description 1
- 238000009826 distribution Methods 0.000 description 1
- 235000013399 edible fruits Nutrition 0.000 description 1
- 239000003995 emulsifying agent Substances 0.000 description 1
- 239000000839 emulsion Substances 0.000 description 1
- HELXLJCILKEWJH-UHFFFAOYSA-N entered according to Sigma 01432 Natural products C1CC2C3(C)CCCC(C)(C(=O)OC4C(C(O)C(O)C(CO)O4)O)C3CCC2(C2)CC(=C)C21OC(C1OC2C(C(O)C(O)C(CO)O2)O)OC(CO)C(O)C1OC1OC(CO)C(O)C(O)C1O HELXLJCILKEWJH-UHFFFAOYSA-N 0.000 description 1
- 230000007613 environmental effect Effects 0.000 description 1
- DNVPQKQSNYMLRS-SOWFXMKYSA-N ergosterol Chemical compound C1[C@@H](O)CC[C@]2(C)[C@H](CC[C@]3([C@H]([C@H](C)/C=C/[C@@H](C)C(C)C)CC[C@H]33)C)C3=CC=C21 DNVPQKQSNYMLRS-SOWFXMKYSA-N 0.000 description 1
- IVTMALDHFAHOGL-UHFFFAOYSA-N eriodictyol 7-O-rutinoside Natural products OC1C(O)C(O)C(C)OC1OCC1C(O)C(O)C(O)C(OC=2C=C3C(C(C(O)=C(O3)C=3C=C(O)C(O)=CC=3)=O)=C(O)C=2)O1 IVTMALDHFAHOGL-UHFFFAOYSA-N 0.000 description 1
- 239000000469 ethanolic extract Substances 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- 230000029142 excretion Effects 0.000 description 1
- 230000001747 exhibiting effect Effects 0.000 description 1
- 238000000855 fermentation Methods 0.000 description 1
- 230000004151 fermentation Effects 0.000 description 1
- 239000000945 filler Substances 0.000 description 1
- 239000004467 fishmeal Substances 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- 238000004108 freeze drying Methods 0.000 description 1
- 208000018685 gastrointestinal system disease Diseases 0.000 description 1
- 239000008273 gelatin Substances 0.000 description 1
- 229920000159 gelatin Polymers 0.000 description 1
- 235000019322 gelatine Nutrition 0.000 description 1
- 235000011852 gelatine desserts Nutrition 0.000 description 1
- 239000008103 glucose Substances 0.000 description 1
- 235000011187 glycerol Nutrition 0.000 description 1
- LPLVUJXQOOQHMX-UHFFFAOYSA-N glycyrrhetinic acid glycoside Natural products C1CC(C2C(C3(CCC4(C)CCC(C)(CC4C3=CC2=O)C(O)=O)C)(C)CC2)(C)C2C(C)(C)C1OC1OC(C(O)=O)C(O)C(O)C1OC1OC(C(O)=O)C(O)C(O)C1O LPLVUJXQOOQHMX-UHFFFAOYSA-N 0.000 description 1
- UYRUBYNTXSDKQT-UHFFFAOYSA-N glycyrrhizic acid Natural products CC1(C)C(CCC2(C)C1CCC3(C)C2C(=O)C=C4C5CC(C)(CCC5(C)CCC34C)C(=O)O)OC6OC(C(O)C(O)C6OC7OC(O)C(O)C(O)C7C(=O)O)C(=O)O UYRUBYNTXSDKQT-UHFFFAOYSA-N 0.000 description 1
- 229960004949 glycyrrhizic acid Drugs 0.000 description 1
- 235000019410 glycyrrhizin Nutrition 0.000 description 1
- LPLVUJXQOOQHMX-QWBHMCJMSA-N glycyrrhizinic acid Chemical compound O([C@@H]1[C@@H](O)[C@H](O)[C@H](O[C@@H]1O[C@@H]1C([C@H]2[C@]([C@@H]3[C@@]([C@@]4(CC[C@@]5(C)CC[C@@](C)(C[C@H]5C4=CC3=O)C(O)=O)C)(C)CC2)(C)CC1)(C)C)C(O)=O)[C@@H]1O[C@H](C(O)=O)[C@@H](O)[C@H](O)[C@H]1O LPLVUJXQOOQHMX-QWBHMCJMSA-N 0.000 description 1
- 239000001963 growth medium Substances 0.000 description 1
- 239000007952 growth promoter Substances 0.000 description 1
- 239000010440 gypsum Substances 0.000 description 1
- 229910052602 gypsum Inorganic materials 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- 230000036039 immunity Effects 0.000 description 1
- 230000016784 immunoglobulin production Effects 0.000 description 1
- 230000008676 import Effects 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 238000011534 incubation Methods 0.000 description 1
- 230000005764 inhibitory process Effects 0.000 description 1
- 208000014674 injury Diseases 0.000 description 1
- 229910052500 inorganic mineral Inorganic materials 0.000 description 1
- 238000000185 intracerebroventricular administration Methods 0.000 description 1
- 238000007918 intramuscular administration Methods 0.000 description 1
- 238000007912 intraperitoneal administration Methods 0.000 description 1
- 238000007913 intrathecal administration Methods 0.000 description 1
- 230000009545 invasion Effects 0.000 description 1
- 238000002955 isolation Methods 0.000 description 1
- 239000008101 lactose Substances 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 235000019359 magnesium stearate Nutrition 0.000 description 1
- 239000000845 maltitol Substances 0.000 description 1
- 235000010449 maltitol Nutrition 0.000 description 1
- VQHSOMBJVWLPSR-WUJBLJFYSA-N maltitol Chemical compound OC[C@H](O)[C@@H](O)[C@@H]([C@H](O)CO)O[C@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O VQHSOMBJVWLPSR-WUJBLJFYSA-N 0.000 description 1
- 229940035436 maltitol Drugs 0.000 description 1
- 239000000594 mannitol Substances 0.000 description 1
- 235000010355 mannitol Nutrition 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 239000008204 material by function Substances 0.000 description 1
- 235000012054 meals Nutrition 0.000 description 1
- 235000013372 meat Nutrition 0.000 description 1
- 208000015688 methicillin-resistant staphylococcus aureus infectious disease Diseases 0.000 description 1
- 229920000609 methyl cellulose Polymers 0.000 description 1
- 239000001923 methylcellulose Substances 0.000 description 1
- 235000010981 methylcellulose Nutrition 0.000 description 1
- LXCFILQKKLGQFO-UHFFFAOYSA-N methylparaben Chemical compound COC(=O)C1=CC=C(O)C=C1 LXCFILQKKLGQFO-UHFFFAOYSA-N 0.000 description 1
- 229960002509 miconazole Drugs 0.000 description 1
- 239000011707 mineral Substances 0.000 description 1
- 235000010755 mineral Nutrition 0.000 description 1
- 239000002480 mineral oil Substances 0.000 description 1
- 235000010446 mineral oil Nutrition 0.000 description 1
- 235000013536 miso Nutrition 0.000 description 1
- 239000012046 mixed solvent Substances 0.000 description 1
- 230000003020 moisturizing effect Effects 0.000 description 1
- 150000002772 monosaccharides Chemical class 0.000 description 1
- 229930014626 natural product Natural products 0.000 description 1
- 239000006916 nutrient agar Substances 0.000 description 1
- 230000035764 nutrition Effects 0.000 description 1
- 235000020824 obesity Nutrition 0.000 description 1
- 210000000056 organ Anatomy 0.000 description 1
- 150000007524 organic acids Chemical class 0.000 description 1
- 235000005985 organic acids Nutrition 0.000 description 1
- 239000005416 organic matter Substances 0.000 description 1
- 244000052769 pathogen Species 0.000 description 1
- LCLHHZYHLXDRQG-ZNKJPWOQSA-N pectic acid Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)O[C@H](C(O)=O)[C@@H]1OC1[C@H](O)[C@@H](O)[C@@H](OC2[C@@H]([C@@H](O)[C@@H](O)[C@H](O2)C(O)=O)O)[C@@H](C(O)=O)O1 LCLHHZYHLXDRQG-ZNKJPWOQSA-N 0.000 description 1
- OQUKIQWCVTZJAF-UHFFFAOYSA-N phenol;sulfuric acid Chemical compound OS(O)(=O)=O.OC1=CC=CC=C1 OQUKIQWCVTZJAF-UHFFFAOYSA-N 0.000 description 1
- 238000013379 physicochemical characterization Methods 0.000 description 1
- 230000008635 plant growth Effects 0.000 description 1
- 239000010318 polygalacturonic acid Substances 0.000 description 1
- 239000003910 polypeptide antibiotic agent Substances 0.000 description 1
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 1
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 1
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 1
- 230000003389 potentiating effect Effects 0.000 description 1
- 244000144977 poultry Species 0.000 description 1
- 235000013594 poultry meat Nutrition 0.000 description 1
- 235000008476 powdered milk Nutrition 0.000 description 1
- 238000004321 preservation Methods 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 230000001737 promoting effect Effects 0.000 description 1
- BDERNNFJNOPAEC-UHFFFAOYSA-N propan-1-ol Chemical compound CCCO BDERNNFJNOPAEC-UHFFFAOYSA-N 0.000 description 1
- QELSKZZBTMNZEB-UHFFFAOYSA-N propylparaben Chemical compound CCCOC(=O)C1=CC=C(O)C=C1 QELSKZZBTMNZEB-UHFFFAOYSA-N 0.000 description 1
- 229960003415 propylparaben Drugs 0.000 description 1
- 230000001681 protective effect Effects 0.000 description 1
- 229940007042 proteus vulgaris Drugs 0.000 description 1
- FDRQPMVGJOQVTL-UHFFFAOYSA-N quercetin rutinoside Natural products OC1C(O)C(O)C(CO)OC1OCC1C(O)C(O)C(O)C(OC=2C(C3=C(O)C=C(O)C=C3OC=2C=2C=C(O)C(O)=CC=2)=O)O1 FDRQPMVGJOQVTL-UHFFFAOYSA-N 0.000 description 1
- 235000019203 rebaudioside A Nutrition 0.000 description 1
- HELXLJCILKEWJH-NCGAPWICSA-N rebaudioside A Chemical compound O([C@H]1[C@H](O)[C@@H](CO)O[C@H]([C@@H]1O[C@H]1[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O1)O)O[C@]12C(=C)C[C@@]3(C1)CC[C@@H]1[C@@](C)(CCC[C@]1([C@@H]3CC2)C)C(=O)O[C@H]1[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O1)O)[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O HELXLJCILKEWJH-NCGAPWICSA-N 0.000 description 1
- 230000000241 respiratory effect Effects 0.000 description 1
- IKGXIBQEEMLURG-BKUODXTLSA-N rutin Chemical compound O[C@H]1[C@H](O)[C@@H](O)[C@H](C)O[C@@H]1OC[C@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](OC=2C(C3=C(O)C=C(O)C=C3OC=2C=2C=C(O)C(O)=CC=2)=O)O1 IKGXIBQEEMLURG-BKUODXTLSA-N 0.000 description 1
- ALABRVAAKCSLSC-UHFFFAOYSA-N rutin Natural products CC1OC(OCC2OC(O)C(O)C(O)C2O)C(O)C(O)C1OC3=C(Oc4cc(O)cc(O)c4C3=O)c5ccc(O)c(O)c5 ALABRVAAKCSLSC-UHFFFAOYSA-N 0.000 description 1
- 235000005493 rutin Nutrition 0.000 description 1
- 229960004555 rutoside Drugs 0.000 description 1
- 235000019204 saccharin Nutrition 0.000 description 1
- CVHZOJJKTDOEJC-UHFFFAOYSA-N saccharin Chemical compound C1=CC=C2C(=O)NS(=O)(=O)C2=C1 CVHZOJJKTDOEJC-UHFFFAOYSA-N 0.000 description 1
- 229940081974 saccharin Drugs 0.000 description 1
- 239000000901 saccharin and its Na,K and Ca salt Substances 0.000 description 1
- 239000012047 saturated solution Substances 0.000 description 1
- HFHDHCJBZVLPGP-UHFFFAOYSA-N schardinger α-dextrin Chemical compound O1C(C(C2O)O)C(CO)OC2OC(C(C2O)O)C(CO)OC2OC(C(C2O)O)C(CO)OC2OC(C(O)C2O)C(CO)OC2OC(C(C2O)O)C(CO)OC2OC2C(O)C(O)C1OC2CO HFHDHCJBZVLPGP-UHFFFAOYSA-N 0.000 description 1
- 230000035945 sensitivity Effects 0.000 description 1
- 239000002453 shampoo Substances 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- 210000004872 soft tissue Anatomy 0.000 description 1
- 229960002920 sorbitol Drugs 0.000 description 1
- 238000001228 spectrum Methods 0.000 description 1
- 238000001694 spray drying Methods 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 239000008107 starch Substances 0.000 description 1
- 235000019698 starch Nutrition 0.000 description 1
- 210000002784 stomach Anatomy 0.000 description 1
- 150000005846 sugar alcohols Chemical class 0.000 description 1
- 239000006228 supernatant Substances 0.000 description 1
- 230000001629 suppression Effects 0.000 description 1
- 238000001356 surgical procedure Methods 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 239000006188 syrup Substances 0.000 description 1
- 235000020357 syrup Nutrition 0.000 description 1
- 239000000454 talc Substances 0.000 description 1
- 229910052623 talc Inorganic materials 0.000 description 1
- 235000012222 talc Nutrition 0.000 description 1
- 235000015523 tannic acid Nutrition 0.000 description 1
- 229920002258 tannic acid Polymers 0.000 description 1
- LRBQNJMCXXYXIU-NRMVVENXSA-N tannic acid Chemical compound OC1=C(O)C(O)=CC(C(=O)OC=2C(=C(O)C=C(C=2)C(=O)OC[C@@H]2[C@H]([C@H](OC(=O)C=3C=C(OC(=O)C=4C=C(O)C(O)=C(O)C=4)C(O)=C(O)C=3)[C@@H](OC(=O)C=3C=C(OC(=O)C=4C=C(O)C(O)=C(O)C=4)C(O)=C(O)C=3)[C@@H](OC(=O)C=3C=C(OC(=O)C=4C=C(O)C(O)=C(O)C=4)C(O)=C(O)C=3)O2)OC(=O)C=2C=C(OC(=O)C=3C=C(O)C(O)=C(O)C=3)C(O)=C(O)C=2)O)=C1 LRBQNJMCXXYXIU-NRMVVENXSA-N 0.000 description 1
- 229940033123 tannic acid Drugs 0.000 description 1
- 238000012360 testing method Methods 0.000 description 1
- 239000000892 thaumatin Substances 0.000 description 1
- 235000010436 thaumatin Nutrition 0.000 description 1
- 239000002562 thickening agent Substances 0.000 description 1
- 230000008733 trauma Effects 0.000 description 1
- QORWJWZARLRLPR-UHFFFAOYSA-H tricalcium bis(phosphate) Chemical compound [Ca+2].[Ca+2].[Ca+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O QORWJWZARLRLPR-UHFFFAOYSA-H 0.000 description 1
- 230000002792 vascular Effects 0.000 description 1
- 208000019553 vascular disease Diseases 0.000 description 1
- 235000013311 vegetables Nutrition 0.000 description 1
- 235000019154 vitamin C Nutrition 0.000 description 1
- 239000011718 vitamin C Substances 0.000 description 1
- 150000003722 vitamin derivatives Chemical class 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
- 238000003809 water extraction Methods 0.000 description 1
- 230000003442 weekly effect Effects 0.000 description 1
- 239000000080 wetting agent Substances 0.000 description 1
- 235000015099 wheat brans Nutrition 0.000 description 1
- 230000002087 whitening effect Effects 0.000 description 1
- 239000002023 wood Substances 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N61/00—Biocides, pest repellants or attractants, or plant growth regulators containing substances of unknown or undetermined composition, e.g. substances characterised only by the mode of action
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01G—HORTICULTURE; CULTIVATION OF VEGETABLES, FLOWERS, RICE, FRUIT, VINES, HOPS OR SEAWEED; FORESTRY; WATERING
- A01G18/00—Cultivation of mushrooms
- A01G18/20—Culture media, e.g. compost
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N63/00—Biocides, pest repellants or attractants, or plant growth regulators containing microorganisms, viruses, microbial fungi, animals or substances produced by, or obtained from, microorganisms, viruses, microbial fungi or animals, e.g. enzymes or fermentates
- A01N63/30—Microbial fungi; Substances produced thereby or obtained therefrom
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Health & Medical Sciences (AREA)
- Zoology (AREA)
- Environmental Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Dentistry (AREA)
- Plant Pathology (AREA)
- Pest Control & Pesticides (AREA)
- Agronomy & Crop Science (AREA)
- Wood Science & Technology (AREA)
- Mycology (AREA)
- Microbiology (AREA)
- Biotechnology (AREA)
- Virology (AREA)
- Medicines Containing Plant Substances (AREA)
Abstract
Description
본 발명은 표고버섯(Lentinus edodes) 재배 시 부산물로 발생하는, 표고버섯 수확 후 버섯폐배지(spent mushroom substrate, SMS)의 추출물을 포함하는 항균 조성물에 관한 것으로서, 보다 구체적으로는, 표고버섯 인공 재배용 배지에 표고버섯 종균을 접종 및 배양하여 표고버섯을 수확한 이후의 남은 버섯폐배지 열수 추출물의 에틸아세테이트 분획물을 포함하는 항세균 및 항진균 활성이 우수한 항균 조성물에 관한 것이다.The present invention relates to an antibacterial composition comprising an extract of spent mushroom substrate (SMS) after harvesting shiitake mushrooms, which is generated as a by-product during cultivation of shiitake mushrooms ( Lentinus edodes ), and more specifically, for artificial cultivation of shiitake mushrooms The present invention relates to an antibacterial composition containing an ethyl acetate fraction of a hot-water extract of a mushroom waste medium remaining after harvesting shiitake mushrooms by inoculating and culturing shiitake mushroom spawn in a medium, and having excellent antibacterial and antifungal activity.
인류는 병원성 미생물의 침입을 방어하고, 식품 부패 미생물을 제어하기 위하여 다양한 항균제와 방부제를 사용하여 왔다. 그러나, 화학 항균제 및 방부제의 경우, 다양한 세균과 진균에 우수한 항균력을 보이는 반면, 장기간 사용시에는 알러지(allergy)에 의한 발적, 혈관성 부종, 골수 억제, 빈혈, 위염, 퇴행성 뇌질환 등의 많은 부작용이 나타나게 된다. 실제 미국에서는 클로람페니콜(chloramphenicol) 등의 부작용이 심한 항생제 사용을 금지한 바 있다. 따라서, 안전성이 확보되어 있으면서 장기간 사용 경험이 있는 천연물을 이용한 천연 항균제 개발에 관한 다양한 연구가 진행 중에 있다.Humans have been using various antibacterial agents and preservatives to defend against the invasion of pathogenic microorganisms and control food spoilage microorganisms. However, in the case of chemical antibacterial agents and preservatives, while showing excellent antibacterial activity against various bacteria and fungi, long-term use causes many side effects such as allergy-induced flare-ups, vascular edema, bone marrow suppression, anemia, gastritis, and degenerative brain disease. do. In fact, the United States has banned the use of antibiotics with severe side effects, such as chloramphenicol. Therefore, various studies on the development of natural antibacterial agents using natural products with long-term experience of use while ensuring safety are in progress.
미국 질병관리예방센터(U.S. Center for Disease Control and Prevention)에 따르면, 병원 내 혈류 감염이 미국에서 주된 사망 원인 중 하나이다. 또한, 피부 및 연조직을 통한 병원성 세균 및 진균의 감염은 보편적인 의학적 질병으로 인식되어 있으며, 주로 외상 또는 수술에 의하여 유발되는 것으로 이해되고 있다. 많은 병원성 세균 및 진균의 감염은 감염의 중증도에 따라 그 처치 방법이 다르지만, 경구용 항미생물제 및 국소 청결로 성공적인 치료가 가능하다. 그러나, 근원적인 위험 인자(예를 들면, 혈관 혈류 이상, 당뇨) 및/또는 치료하기 어렵거나 다중-내성인 세균 및 진균에 의해 유발된 감염을 갖는 환자에서 흔히 발생하는 보다 심하거나 복잡한 감염은 정맥 내 항미생물제 치료 및 침입성 외과적 조직 제거 등의 조치가 필요할 수도 있다. 실례로, 글라이코펩타이드계 항생물질인 반코마이신은 다중-약물-내성 그람-양성균, 특히 MRSA, 응고효소-음성 스태필로코커스 및 엔테로코커스에 의해 야기된 심한 병원 내 감염에 대해 성공적으로 사용되어왔다. 그러나, 최근 반코마이신에 대해 내성을 갖는 균주의 분리가 증가하고 있음이 보고되고 있으며, 현재의 항생제로는 병원균 중 일부균에 의해 유발된 심한 감염에 대한 치료가 매우 제한되어 있는 것이 사실이다. 따라서, 점차 증가되고 있는 내성균에 대한 보다 효과적인 새로운 항생물질 개발의 필요성이 증가되고 있다.According to the U.S. Center for Disease Control and Prevention, hospitalized bloodstream infections are one of the leading causes of death in the United States. In addition, infection of pathogenic bacteria and fungi through the skin and soft tissue is recognized as a universal medical disease, and is understood to be mainly caused by trauma or surgery. Many pathogenic bacterial and fungal infections can be successfully treated with oral antimicrobial agents and topical cleansing, although treatment methods vary depending on the severity of the infection. However, more severe or complex infections, which often occur in patients with underlying risk factors (e.g., abnormal blood flow, diabetes) and/or infections caused by difficult-to-treat or multi-resistant bacteria and fungi, are Treatment with antimicrobial agents and invasive surgical tissue removal may be necessary. For example, vancomycin, a glycopeptide antibiotic, has been successfully used against severe nosocomial infections caused by multi-drug-resistant Gram-positive bacteria, particularly MRSA, coagulase-negative Staphylococcus and Enterococcus. However, it has recently been reported that the isolation of strains resistant to vancomycin is increasing, and it is true that current antibiotics are very limited in treating severe infections caused by some of the pathogens. Therefore, there is an increasing need for the development of new antibiotics that are more effective against the gradually increasing resistant bacteria.
한편, 표고버섯은 담자균강 주름버섯목 느타리과 잣버섯속에 속하는 식용 버섯으로 버섯 중 비타민 C의 함량이 가장 많으며, 각종 아미노산과 에르고스테롤 (ergosterol)을 다량 함유하고 있다. 건강 기능성 측면에서 항암, 폐질환 및 위장 질환 예방, 바이러스에 대한 면역 증강, 항체 생성 촉진, 혈압 강하, 동맥경화 예방, 혈중 지질 농도 조절 기능, 당뇨 억제 효과 등이 알려져 있다. On the other hand, shiitake mushrooms are edible mushrooms belonging to the genus Pleurotus and Pine nut, and have the highest content of vitamin C among mushrooms, and contain a large amount of various amino acids and ergosterol. In terms of health functionalities, anticancer, lung and gastrointestinal disease prevention, immunity enhancement against viruses, promotion of antibody production, lowering blood pressure, prevention of arteriosclerosis, blood lipid concentration control function, antidiabetic effect, etc. are known.
표고버섯과 관련된 특허로는 대한민국 등록특허 제10-1839507호 [표고버섯과 표고버섯 발효물이 함유된 양념 창난 젓갈 제조방법], 제10-1830763호 [표고버섯 추출물을 포함하는 쌀된장의 제조방법 및 이로부터 제조된 쌀된장], 제10-1823460호 [표고버섯분말을 이용한 반건조 우럭의 제조방법], 제10-1759312호 [표고버섯가루의 제조방법, 표고버섯가루를 이용한 영양바의 제조방법 및 이에 의해 제조된 영양바], 제10-1673573호 [표고버섯차의 제조방법 및 이로 제조된 표고버섯차]가 개시되어 있으며, 대한민국 공개특허 제10-2004-0038957호 [표고버섯분말을 함유한 김(laver) 제조를 주제로 한 식품]이 공개되어 있다. 현재까지 알려진 대부분의 특허는 표고버섯 재배법 및 표고버섯을 이용한 다양한 식품 제조에 대한 것으로, 표고버섯의 유용 생리 활성에 대한 특허는 매우 제한되어 있는 실정이다.Patents related to shiitake mushrooms include Korean Registered Patent No. 10-1839507 [Method of manufacturing seasoned salted fish marinated with shiitake mushrooms and fermented shiitake mushrooms], No. 10-1830763 [Method of manufacturing rice doenjang containing shiitake mushroom extract] and rice miso paste prepared therefrom], No. 10-1823460 [Method for producing semi-dried rockfish using shiitake mushroom powder], No. 10-1759312 [Method for producing shiitake mushroom powder, production of nutritional bar using shiitake mushroom powder] Method and nutritional bar produced thereby], and No. 10-1673573 [Method for manufacturing shiitake mushroom tea and shiitake mushroom tea produced thereby] are disclosed, and Korean Patent Publication No. 10-2004-0038957 [shiitake mushroom powder Foods with the subject of laver production] are disclosed. Most of the patents known to date relate to shiitake mushroom cultivation and various food preparations using shiitake mushrooms, and patents on useful physiological activities of shiitake mushrooms are very limited.
또한, 표고버섯의 유용 생리 활성 관련 특허로는 대한민국 등록특허 제10-1487742호 [표고버섯과 천마 추출물을 포함하는 항산화 효능을 갖는 식품 조성물], 제10-1806808호 [표고버섯 균사체를 이용하여 발효시킨 적하수오 발효물을 유효성분으로 함유하는 여성 갱년기 증상의 예방, 개선 또는 치료용 조성물], 제10-1611947호 [혈관 질환 예방 및 개선용 건강 식품 조성물 및 그 제조방법], 제10-1266528호 [표고버섯 균사체 배양물에서 추출된 다당체를 유효성분으로 함유하는 보습 및/또는 미백용 화장료 조성물]이 개시되어 있으며, 공개특허 제10-2017-0036912호 [표고버섯 추출물을 유효성분으로 함유하는 아토피 및 접촉성 피부염 예방 및 개선용 조성물], 공개특허 제10-2003-0056753호 [표고버섯 균사체, 아가리쿠스 균사체, 수용성 키토산을 함유하는 비만 조절용 건강 식품 조성물], 제10-2008-0110212호 [표고버섯 열수 추출물을 이용한 골길이 성장에 도움을 주는 조성물]이 공개되어 있다.In addition, patents related to useful physiological activity of shiitake mushrooms include Korean Patent Registration No. 10-1487742 [Food composition having antioxidant effect containing shiitake mushroom and cinnamon extract], No. 10-1806808 [fermentation using shiitake mushroom mycelium] [Composition for prevention, improvement, or treatment of female menopausal symptoms containing fermented saline fermented soybeans as an active ingredient], No. 10-1611947 [Health food composition for preventing and improving vascular disease and manufacturing method thereof], No. 10-1266528 [A cosmetic composition for moisturizing and/or whitening containing a polysaccharide extracted from shiitake mushroom mycelium culture as an active ingredient] is disclosed, and Patent Publication No. 10-2017-0036912 [Atopy containing shiitake mushroom extract as an active ingredient] and composition for preventing and improving contact dermatitis], Patent Publication No. 10-2003-0056753 [Health food composition for controlling obesity containing shiitake mushroom mycelium, Agaricus mycelium, and water-soluble chitosan], No. 10-2008-0110212 [shiitake mushroom A composition that helps bone growth using a hot water extract] has been disclosed.
국내 식용버섯의 년간 총 생산량은 2013년 173,354톤에서 2017년 162,292톤으로 보고되어 있으며, 2017년의 경우 느타리버섯이 58,784톤, 새송이버섯이 48,588톤, 팽이버섯이 38,092톤, 양송이버섯이 10,173톤이 생산되었으며, 표고버섯 생산량은 25,000톤으로 추정되고 있다(농림축산식품부, 2016. 특용작물 생산실적). 이러한 버섯 재배는 천연 배지에서 점차 특정의 인공 배지를 사용하는 방식으로 변화되고 있다. 버섯의 인공 재배 및 생산량이 증가하면서, 버섯폐배지의 배출도 급속도로 증가되고 있다. 일반적으로 버섯 1kg 생산에 버섯 배지가 5kg이 소비되는 것을 감안할 때, 국내 버섯폐배지는 년간 200만톤 이상이 배출되는 것으로 추정된다. 특히 표고버섯의 경우, 다양한 나무의 톱밥 배지를 사용하는데, 2015년 중국에서 수입된 톱밥 배지는 36,285톤으로 2012년 11,825톤이었던 수입량과 비교하면 3년만에 2배 이상 증가하였다(관세청 수출입통계; 주간조선, 2016. 4. 11).The total annual production of edible mushrooms in Korea is reported to be 162,292 tons in 2017 from 173,354 tons in 2013. In 2017, oyster mushrooms were 58,784 tons, king oyster mushrooms 48,588 tons, enoki mushrooms 38,092 tons, and button mushrooms 10,173 tons. production, and shiitake mushroom production is estimated to be 25,000 tons (Ministry of Agriculture, Food and Rural Affairs, 2016. Production of special crops). These mushroom cultivations are gradually changing from natural media to using specific artificial media. As the artificial cultivation and production of mushrooms increase, the emission of mushroom waste media is also rapidly increasing. Considering that 5 kg of mushroom medium is generally consumed for the production of 1 kg of mushrooms, it is estimated that more than 2 million tons of waste mushroom media are emitted annually in Korea. In particular, in the case of shiitake mushrooms, sawdust medium from various trees is used. In 2015, sawdust medium imported from China was 36,285 tons, more than doubled in three years compared to 11,825 tons in 2012 (Export and import statistics of the Customs Service; Weekly Chosun, 2016. 4. 11).
인공 배지를 사용하는 경우, 버섯 재배에는 20~25%의 영양 성분이 소요된다고 알려져 있으며(Williams et al, 2001. Biores Technol. 79:227-230.; Bae et al, 2006. J Anim Sci & Technol Kor. 48:237-246). 따라서, 버섯폐배지에는 상당량의 영양성분이 잔존하게 된다. 또한, 버섯폐배지에는 이미 버섯 균사체가 대량 배양된 상태이므로 버섯 자실체 및 균사체로부터 유래하는 β-glucosidase, cellulase 등의 다양한 효소와 식물 생육 촉진 유효성분, 항균 물질, 병 저항성 유도체 등의 다양한 생리 활성을 가진 이차대사산물이 포함되어 있는 것으로 알려져 있다(Hautzel and Anke., 1990, Z Naturforsch. 45: 1093-1098; Lim et al., 2012. Mycobiology. 41: 160-166; Parada et al., 2012 Crop Protection 30: 443-450; Suess, 2006, Agriculture and Lands. pp. 1-101). 따라서, 버섯폐배지는 영양적으로나 생리 활성적으로 우수한 소재로 사용될 수 있다. 그러나, 현재까지 버섯폐배지를 이용한 고부가가치 기능성 소재로의 개발 연구는 매우 제한된 실정이며, 표고버섯 재 이후 발생하는 수확 후 버섯폐배지 추출물의 강력한 항균 조성물은 알려진 바 없는 실정이다.It is known that 20-25% of nutrients are required for mushroom cultivation when artificial media are used (Williams et al, 2001. Biores Technol. 79:227-230.; Bae et al, 2006. J Anim Sci & Technol. 48:237-246). Therefore, a significant amount of nutrients remain in the mushroom waste medium. In addition, since the mushroom mycelium is already mass-cultured in the mushroom waste medium, various enzymes such as β-glucosidase and cellulase derived from mushroom fruiting bodies and mycelia, as well as various physiological activities such as plant growth promoting active ingredients, antibacterial substances, and disease resistance derivatives, etc. (Hautzel and Anke., 1990, Z Naturforsch. 45: 1093-1098; Lim et al., 2012. Mycobiology. 41: 160-166; Parada et al., 2012 Crop Protection 30: 443-450; Suess, 2006, Agriculture and Lands. pp. 1-101). Therefore, the mushroom waste medium can be used as a nutritionally or physiologically active material. However, to date, research on the development of high value-added functional materials using waste mushroom medium has been very limited, and a strong antibacterial composition of post-harvest mushroom waste medium extracts generated after shiitake mushroom ash has not been known.
국내의 경우, 버섯폐배지의 일부는 사료용으로 사용되기도 하였으며(정종천 외, 2012. J. Mushroom Sci. Prod. 10: 174-178), 버섯 재배에 10~30% 첨가하여 인공배지로 재이용(이남길 외, 2015. J. Mushrooms, 13: 310-313)되기도 하였으나, 인공 배지에 톱밥 함량이 높은 경우, 사료나 버섯 재배에 재이용되지 못하고 거의 퇴비로 사용되고 있는 실정이다. 최근, 외국에서는 버섯폐배지의 다양한 미생물 및 효소를 이용하여 축사 폐기물을 효율적으로 정화하고자 하는 연구가 활발히 진행되고 있어(Luo X 등, 2018, Bioresour. Technol. 250: 611-620; Meng L 등, 2018. Bioresour. Technol. 253: 197-203; Meng X 등, 2018. Bioresour. Technol. 251: 22-30), 향후 버섯폐배지는 새로운 환경정화제로 사용 가능성이 제시되고 있다. In Korea, some of the waste mushroom medium was used for feed (Jeong Jong-cheon et al., 2012. J. Mushroom Sci. Prod. 10: 174-178), and 10 to 30% was added to mushroom cultivation and reused as an artificial medium (Lee Nam-gil et al., 2015. J. Mushrooms, 13: 310-313), but when the sawdust content is high in the artificial medium, it is not reused for feed or mushroom cultivation and is almost used as compost. Recently, studies have been actively conducted in foreign countries to efficiently purify barn waste using various microorganisms and enzymes of mushroom waste medium (Luo X et al., 2018, Bioresour. Technol. 250: 611-620; Meng L et al., 2018. Bioresour. Technol. 253: 197-203; Meng X et al., 2018. Bioresour. Technol. 251: 22-30), suggesting the possibility of using mushroom waste medium as a new environmental cleaning agent in the future.
버섯폐배지의 유용 물질 함량은, 버섯 접종 전의 배지 조성 및 배양 버섯의 종류 및 상태에 의해 달라지게 된다. 현재까지 대부분의 연구는 팽이버섯 버섯폐배지에 집중되어 있었으나(정종천 외, 2012. J. Mushroom Sci. Prod. 10: 174-178), 최근에는 노루궁뎅이버섯의 버섯폐배지(이상엽, 2015, The Korean Journal of Mycology 43: 185-190) 및 표고 톱밥 배지의 베타-글루칸 함량에 대해서도 연구되고 있다(박영애, 2016. 한국균학회지, 44: 296-299). 현재까지 버섯폐배지의 알려진 유용 생리 활성으로는 큰느타리버섯, 표고버섯, 노루궁뎅이버섯의 버섯폐배지의 실온에서의 물 추출물의 고추 생육 촉진 및 역병 억제 효과(곽아민 외, 2015. J. Mushrooms, 13: 16-20; Kang DS 외, 2017. Plant Pathology J. 33: 264-275) 및 느타리버섯 버섯폐배지의 물 추출물의 항염증 효과가 알려져 있다(Rivero-Perez N. 등, 2016. Indian J Pharmacol. 48: 141-144). 또한, 버섯폐배지의 주요 활성 성분으로는 수용성 고분자 다당류(polysaccharide)가 주로 보고되어 있으며, 최근 표고버섯 버섯폐배지에서 항산화 활성의 고분자 다당류가 보고된 바 있으며(Zhu H 등, 2018, Int. J. Biol. Macromol. 112: 976-978), 암세포 생육 억제 활성의 산성 다당류(Zhang Y 등, 2017. Int. J. Med. Mushrooms. 19: 395-403) 및 항균 활성의 다당류도 확인되어 있다(Zhu H, 2012, Int. J. Biol. Macromol. 50: 840-843). 그러나, 현재까지 표고버섯 버섯폐배지에서 강력한 항균 활성이 보고된 바는 없다. The content of useful substances in the waste mushroom medium varies depending on the composition of the medium before mushroom inoculation and the type and condition of the cultured mushrooms. Until now, most studies have been focused on the waste medium of mushrooms for enoki mushrooms (Jeong Jong-cheon et al., 2012. J. Mushroom Sci. Prod. 10: 174-178), but recently, the waste medium for ericium erinaceus (Lee Sang-yeop, 2015, The Korean Journal of Mycology 43: 185-190) and beta-glucan content of shiitake sawdust medium are also being studied (Park Young-ae, 2016. Korean Journal of Mycology, 44: 296-299). To date, the known useful physiological activities of mushroom waste media include the growth promotion and pest control effects of pepper growth promotion and pestilence inhibition effects of water extracts of mushroom waste media of oyster mushrooms, shiitake mushrooms, and ericium erinaceus at room temperature (Kwak Amin et al., 2015. J. Mushrooms , 13: 16-20; Kang DS et al., 2017. Plant Pathology J. 33: 264-275) and the anti-inflammatory effect of the water extract of oyster mushroom mushroom waste medium is known (Rivero-Perez N. et al., 2016. Indian J Pharmacol. 48: 141-144). In addition, water-soluble polysaccharides have been reported as the main active ingredient of mushroom waste medium, and recently, antioxidant activity of polysaccharides has been reported in shiitake mushroom mushroom waste medium (Zhu H et al., 2018, Int. J Biol. Macromol. 112: 976-978), acidic polysaccharides with cancer cell growth inhibitory activity (Zhang Y et al., 2017. Int. J. Med. Mushrooms. 19: 395-403), and polysaccharides with antibacterial activity have also been identified ( Zhu H, 2012, Int. J. Biol. Macromol. 50: 840-843). However, no strong antibacterial activity has been reported in shiitake mushroom waste medium.
또한, 버섯폐배지 관련 특허는 모두 버섯폐배지를 이용한 연료용 팰릿 제조(대한민국 등록특허 제10-1728233호, 버섯 재배 후의 폐배지 또는 폐목재를 활용한 연료용 펠릿 제조방법), 사료 제조(대한민국 공개특허 제10-2014-0066036호, 버섯 폐배지를 이용한 사료 및 이의 제조방법), 및 당화용 효소 제조(대한민국 등록특허 제10-1458628호, 버섯 폐배지로부터 당화효소를 제조하는 방법), 항혈전 활성 조성물(대한민국 등록특허 제10-2128625호, 표고버섯 수확후의 배지 추출물을 유효성분으로 함유하는 혈전증의 예방 또는 치료용 약학적 조성물 및 건강기능식품) 및 옥살산을 활성성분으로 함유하는 고추역병/토마토 풋마름병 방제 조성물(대한민국 등록특허 제10-1873978호, 표고버섯 수확 후의 배지 추출물을 포함하는 식물병 방제용 조성물 및 그의 제조방법)에 대한 것이 알려져 있으나, 표고버섯 버섯폐배지의 열수 추출물로부터 수득되는 에틸아세테이트 분획물의 강력한 광범위 항세균 및 항진균 활성이 보고된 바는 없다. In addition, all patents related to mushroom waste medium are manufacturing fuel pellets using mushroom waste medium (Korean Registered Patent No. 10-1728233, method of manufacturing fuel pellets using waste medium or waste wood after mushroom cultivation), feed manufacturing (Korea Patent Publication No. 10-2014-0066036, feed using mushroom waste medium and its manufacturing method), and saccharification enzyme preparation (Korean Patent No. 10-1458628, method for producing saccharification enzyme from mushroom waste medium), section Antithrombotic active composition (Republic of Korea Patent No. 10-2128625, pharmaceutical composition and health functional food for preventing or treating thrombosis containing medium extract after harvesting shiitake mushroom as an active ingredient) and pepper blight containing oxalic acid as an active ingredient / Tomato green blight control composition (Republic of Korea Patent No. 10-1873978, composition for controlling plant diseases including medium extract after shiitake mushroom harvest and method for producing the same) is known, but obtained from hot water extract of shiitake mushroom waste medium There have been no reports of potent broad-spectrum antibacterial and antifungal activity of ethyl acetate fractions.
본 발명은 상기와 같은 종래 기술의 문제점을 해결하기 위하여 안출된 것으로서, 본 발명에서 해결하고자 하는 과제는 표고버섯 인공 재배시 부산물로 발생하는 버섯폐배지의 추출물을 포함하는 항균 조성물을 제공하고자 하는 것이다.The present invention has been made to solve the problems of the prior art as described above, and the problem to be solved in the present invention is to provide an antimicrobial composition containing an extract of mushroom waste medium generated as a by-product during artificial cultivation of shiitake mushrooms. .
상기와 같은 과제를 해결하기 위하여, 본 발명은 표고버섯(Lentinus edodes) 수확 후 버섯폐배지(Spent Mushroom Substrate, SMS) 추출물을 포함하는 항균 조성물을 제공한다.In order to solve the above problems, the present invention provides an antibacterial composition comprising a mushroom waste medium (Spent Mushroom Substrate, SMS) extract after harvesting shiitake mushrooms ( Lentinus edodes ).
상기 버섯폐배지는 표고버섯을 1~3회 수확 후의 버섯폐배지인 것이 바람직하다.The waste mushroom medium is preferably a waste mushroom medium after harvesting shiitake mushrooms 1 to 3 times.
상기 추출물은 열수로 추출한 후 에틸아세테이트로 분획한 것이 바람직하다.The extract is preferably extracted with hot water and then fractionated with ethyl acetate.
상기 항균 조성물은 약학 조성물, 건강 기능 식품, 화장료 조성물, 동물 사료 조성물, 식품 첨가물, 화장품 첨가물, 동물 사료용 첨가물, 세정제 및 청결제로 이루어지는 군으로부터 선택되는 것이 바람직하다.The antibacterial composition is preferably selected from the group consisting of pharmaceutical compositions, health functional foods, cosmetic compositions, animal feed compositions, food additives, cosmetic additives, animal feed additives, detergents and cleaners.
본 발명의 광범위 항균 조성물의 유효성분으로서의 표고버섯 인공 재배시 부산물로 발생하는 버섯폐배지의 열수 추출물의 에틸아세테이트 분획물은 우수한 항균 활성을 나타내어, 항균용 약학 조성물, 건강 기능 식품, 화장료 조성물, 동물 사료 조성물, 식품, 화장품 또는 동물 사료용 첨가물, 각종 세정제 및 각종 청결제 등의 용도로 사용할 수 있는 뛰어난 효과가 있다. 뿐만 아니라, 본 발명의 표고버섯 인공 재배시 부산물로 발생하는 버섯폐배지의 열수 추출물의 에틸아세테이트 분획물은 열 안정성이 우수하고, pH 2의 산성조건 및 혈장 내에서도 활성의 손실이 나타나지 않아, 액상, 크림, 분말, 환, 정 등의 다양한 형태로 손쉽게 가공될 수 있어 제약 산업, 식품 산업, 축산업, 보건 산업 및 화장품 산업상 매우 유용하게 이용될 수 있다.The ethyl acetate fraction of the hot-water extract of the mushroom waste medium generated as a by-product during the artificial cultivation of shiitake mushrooms as an active ingredient of the broad-spectrum antibacterial composition of the present invention exhibits excellent antibacterial activity, making it an antibacterial pharmaceutical composition, health functional food, cosmetic composition, and animal feed It has excellent effects that can be used for applications such as compositions, food, cosmetics, additives for animal feed, various detergents, and various cleaning agents. In addition, the ethyl acetate fraction of the hot water extract of the mushroom waste medium generated as a by-product during the artificial cultivation of shiitake mushrooms of the present invention has excellent heat stability and does not show loss of activity even in acidic conditions of pH 2 and plasma, so it can be used in liquid, cream , It can be easily processed into various forms such as powder, pill, tablet, etc., so it can be used very usefully in the pharmaceutical industry, food industry, livestock industry, health industry and cosmetics industry.
도 1은 좌측으로부터 우측 방향으로 실험에 사용한 표고버섯 재배용 살균 배지, 살균 배지에 표고버섯 종균을 접종 및 배양하여 표고버섯을 1회 수확한 이후의 버섯폐배지, 3회 수확한 이후의 버섯폐배지, 및 수확한 표고버섯의 사진도이다.
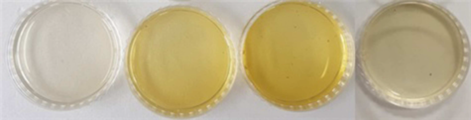
도 2는 좌측으로부터 우측 방향으로 실험에 사용한 표고버섯 재배용 살균 배지, 살균 배지에 표고버섯 종균을 접종 및 배양하여 표고버섯을 1회 수확한 이후의 버섯폐배지, 3회 수확한 이후의 버섯폐배지, 및 수확한 표고버섯 각각의 열수 추출물을 나타낸 사진도이다. Figure 1 shows, from left to right, a sterilization medium for growing shiitake mushrooms used in the experiment, a waste mushroom medium after harvesting shiitake mushrooms once by inoculating and culturing shiitake spawn in the sterilization medium, and a waste mushroom medium after harvesting three times. , and photographs of harvested shiitake mushrooms.
Figure 2 is a sterilization medium for shiitake mushroom cultivation used in the experiment from left to right, a waste mushroom medium after harvesting shiitake mushrooms once by inoculating and culturing shiitake spawn in a sterilization medium, and a waste mushroom medium after harvesting three times. , and a photograph showing the hot water extract of each of the harvested shiitake mushrooms.
이하, 본 발명을 상세하게 설명한다.Hereinafter, the present invention will be described in detail.
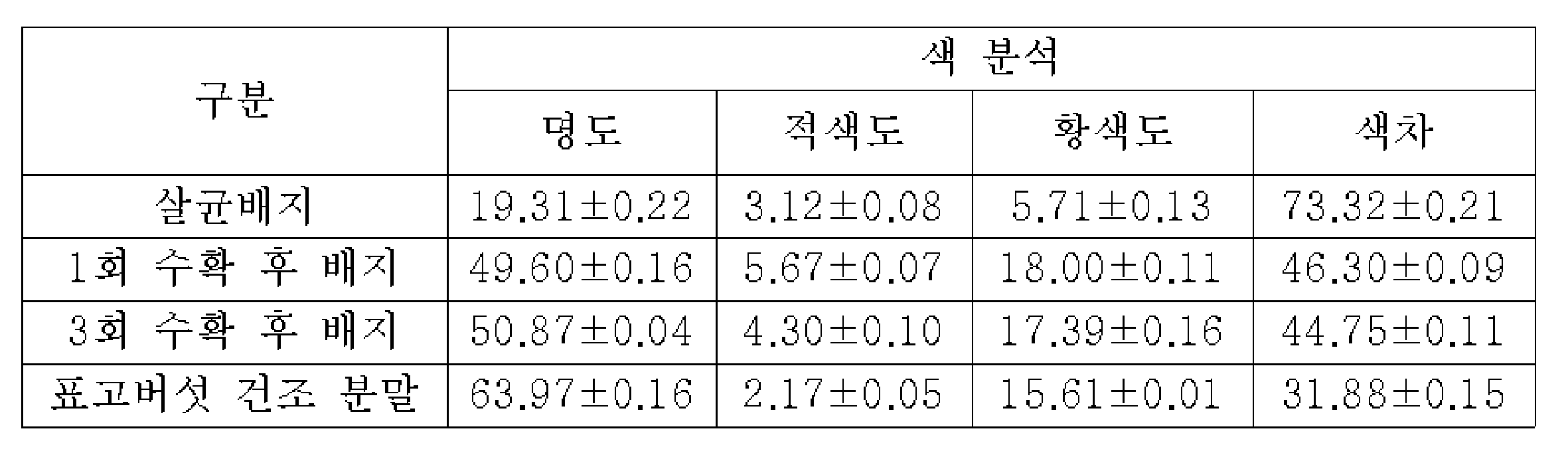
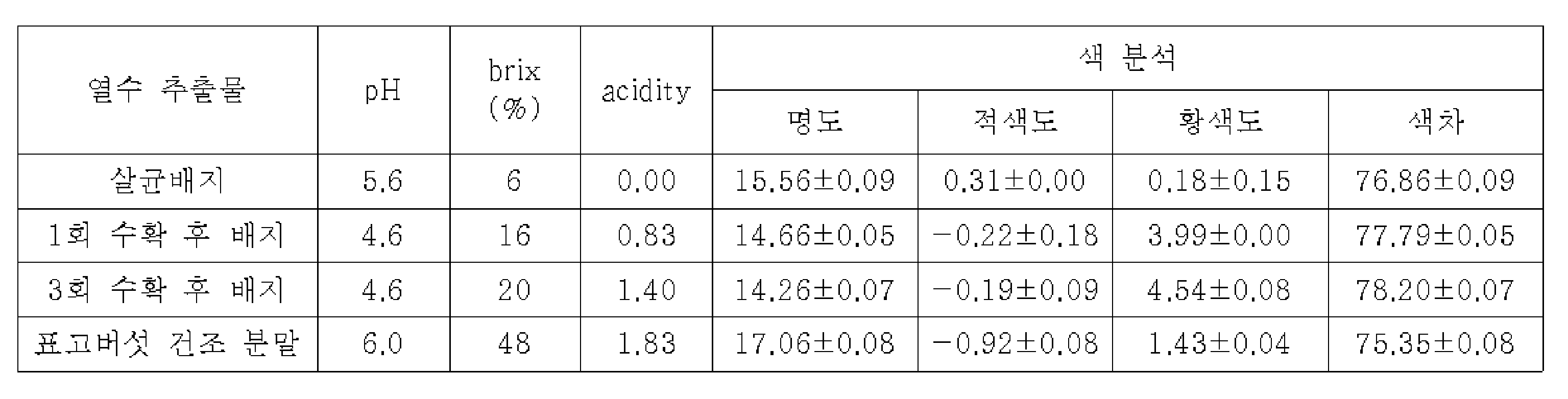
본 발명의 발명자들은 표고버섯 버섯폐배지의 항균 효능을 검증하기 위하여, 표고버섯 재배를 위한 살균 배지(접종 이전의 새 배지), 살균 배지에 표고버섯 종균을 접종 및 배양하여 표고버섯을 1회 수확한 이후의 버섯폐배지, 3회 수확한 이후의 버섯폐배지 및 수확한 표고버섯 각각의 건조분말을 준비하였다. 이후, 일정 방법으로 각각의 열수 추출물을 조제하고, 이들의 항세균 및 항진균 활성을 평가한 결과, 모든 추출물에서 의미있는 항균 활성이 나타나지 않았다. 이는 기존 보고(대한민국 등록특허 제10-1873978호)에서와 같이 버섯폐배지의 옥살산을 농축하지 않는 경우, 표고버섯 분말, 살균 배지, 수확 후 배지에서는 항균활성이 매우 미약함을 의미하였다. In order to verify the antibacterial efficacy of the shiitake mushroom waste medium, the inventors of the present invention inoculated and cultured the shiitake mushroom spawn in a sterilization medium for shiitake mushroom cultivation (new medium before inoculation) and a sterilization medium to harvest the shiitake mushrooms once. Waste mushroom medium after harvesting, waste mushroom medium after harvesting three times, and dry powder of each harvested shiitake mushroom were prepared. Thereafter, each of the hot water extracts was prepared in a certain way, and as a result of evaluating their antibacterial and antifungal activities, no significant antibacterial activity was found in all of the extracts. This meant that, as in the previous report (Korean Patent No. 10-1873978), when the oxalic acid of the mushroom waste medium was not concentrated, the antimicrobial activity was very weak in the shiitake mushroom powder, sterilized medium, and post-harvest medium.
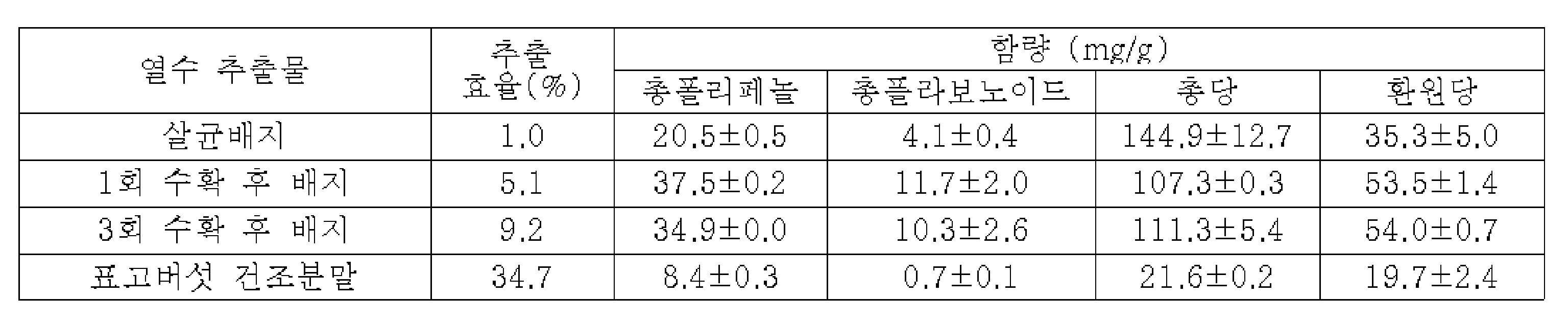
이후, 각각의 추출물로부터 헥센 분획물, 에틸아세테이트 분획물, 부탄올 분획물 및 유기용매 분획 후의 물 잔류물을 각각 조제하였으며, 이들에 대한 항균 활성 평가 결과, 표고버섯 재배 후의 버섯폐배지의 열수 추출물의 에틸아세테이트 분획물을 광범위 항균 활성 성분으로 회수하였으며, 상기 분획물은 인간 적혈구에 대해 용혈 활성은 전혀 나타내지 않으면서도, 열 안정성과 산 안정성이 우수한 특징을 가짐을 확인함으로써 상기 에틸아세테이트 분획물을 그람 양성 세균, 그람 음성 세균 및 진균 감염병에 대한 광범위 항균제 등으로 개발될 수 있음을 확인하여 본 발명을 완성하였다.Then, hexene fractions, ethyl acetate fractions, butanol fractions, and water residues after organic solvent fractionation were prepared from each of the extracts, and as a result of evaluating the antibacterial activity thereof, the ethyl acetate fraction of the hot water extract of the mushroom waste medium after cultivation of shiitake mushrooms was recovered as a broad-spectrum antibacterial active component, and it was confirmed that the fraction had excellent heat stability and acid stability while not exhibiting any hemolytic activity against human red blood cells, and thus the ethyl acetate fraction was classified into gram-positive bacteria, gram-negative bacteria and The present invention was completed by confirming that it could be developed as a broad-spectrum antibacterial agent for fungal infections.
구체적으로, 본 발명자들은 표고버섯의 균사체 및 미성숙 자실체가 대량 배양된 후의 버섯폐배지에서 항균 조성물을 개발하기 위하여, 살균 배지, 표고버섯 1회 수확 후 버섯폐배지, 표고버섯 3회 수확 후 버섯폐배지, 표고버섯의 영양 성분 분석, 이화학적 특성 분석에 이어, 상기 시료들을 대상으로 열수 추출물을 각각 조제하고 항균 활성을 평가하였으며, 이후 각각의 추출물에 대상으로 순차적 유기용매 분획물을 조제하여 1회 및 3회 수확 후 버섯폐배지 열수 추출물의 에틸아세테이트 분획물에서 강력한 항세균 및 항진균 활성을 확인하였다. 또한, 상기 표고버섯 수확 후 버섯폐배지 열수 추출물의 에틸아세테이트 분획물은 인간 적혈구에 대한 용혈 활성은 나타내지 않음을 확인하였다. Specifically, the inventors of the present invention, in order to develop an antibacterial composition in mushroom waste medium after mass cultivation of shiitake mycelium and immature fruiting bodies, sterilized medium, mushroom waste medium after one harvest of shiitake mushrooms, mushroom waste after three shiitake mushrooms Following analysis of the nutrient components and physicochemical properties of the culture medium and shiitake mushrooms, hot water extracts were prepared for each of the above samples and antibacterial activity was evaluated, and then sequential organic solvent fractions were prepared for each extract, and After harvesting three times, strong antibacterial and antifungal activities were confirmed in the ethyl acetate fraction of the mushroom waste medium hot water extract. In addition, it was confirmed that the ethyl acetate fraction of the mushroom waste medium hot water extract after harvesting the shiitake mushroom did not exhibit hemolytic activity on human red blood cells.
따라서, 본 발명은 본 발명은 표고버섯(Lentinus edodes) 수확 후 버섯폐배지(Spent Mushroom Substrate, SMS) 추출물을 포함하는 항균 조성물을 제공한다.Therefore, the present invention provides an antibacterial composition comprising an extract of shiitake mushroom ( Lentinus edodes ) after harvesting mushroom waste medium (Spent Mushroom Substrate, SMS).
상기 버섯폐배지는 표고버섯을 1~3회 수확 후의 버섯폐배지인 것이 바람직하다.The waste mushroom medium is preferably a waste mushroom medium after harvesting shiitake mushrooms 1 to 3 times.
상기 추출물은 열수로 추출한 후 에틸아세테이트로 분획한 것이 바람직하다.The extract is preferably extracted with hot water and then fractionated with ethyl acetate.
상기 항균 조성물은 약학 조성물, 건강 기능 식품, 화장료 조성물, 동물 사료 조성물, 식품 첨가물, 화장품 첨가물, 동물 사료용 첨가물, 세정제 및 청결제로 이루어지는 군으로부터 선택되는 것이 바람직하다.The antibacterial composition is preferably selected from the group consisting of pharmaceutical compositions, health functional foods, cosmetic compositions, animal feed compositions, food additives, cosmetic additives, animal feed additives, detergents and cleaners.
이하에서는, 본 발명의 표고버섯 인공재배 시의 수확 후 버섯폐배지 에탄올 추출물의 에틸아세테이트 활성 분획물의 제조 방법 및 효능 실험 등을 보다 구체적으로 설명한다.Hereinafter, the production method and efficacy test of the ethyl acetate active fraction of the mushroom waste medium ethanol extract after harvest during artificial cultivation of shiitake mushrooms of the present invention will be described in more detail.
본 발명은 표고버섯 인공 재배를 위한 살균 배지, 살균 배지에 표고버섯 종균을 접종 및 배양하여 표고버섯을 1회 수확한 이후의 버섯폐배지, 3회 수확한 이후의 버섯폐배지, 및 수확한 표고버섯의 각각의 분말을 준비하는 단계; 상기 4종 시료로부터 추출물을 조제하는 단계; 상기 추출물의 항세균 및 항진균 활성 평가 단계; 상기 열수 추출물들을 대상으로 헥센, 에틸아세테이트 및 부탄올의 순차적 유기용매 분획물 조제 및 이후 얻어지는 물 잔류물의 조제 단계; 및 상기 분획물의 항세균 및 항진균 활성 평가 단계 및 추출물과 활성 분획물의 활성 물질의 안정성 및 활성 분획물의 인간 적혈구 용혈활성 조사 단계를 포함한다.The present invention is a sterilization medium for artificial cultivation of shiitake mushrooms, a waste mushroom medium after harvesting shiitake mushrooms once by inoculating and culturing shiitake spawn in a sterilization medium, a waste mushroom medium after harvesting three times, and harvested shiitake Preparing each powder of mushrooms; preparing an extract from the four samples; Evaluating the antibacterial and antifungal activity of the extract; preparing sequential organic solvent fractions of hexene, ethyl acetate and butanol from the hot water extracts and preparing a water residue obtained thereafter; and evaluating the antibacterial and antifungal activities of the fractions and examining the stability of the active substances of the extracts and active fractions and the hemolytic activity of human red blood cells of the active fractions.
본 발명의 조성물에 포함되는 "표고버섯 인공재배 시의 수확 후 버섯폐배지 추출물"은 표고버섯을 접종 및 배양한 후 표고버섯을 1~3회 수확한 이후의 폐기되는 버섯폐배지를 회수하여 분쇄하는 단계; 상기 분쇄된 버섯폐배지를 용매로 추출하는 단계 및 상기 추출액을 0.06mm 이하의 여과망을 사용하여 여과하고, 이를 감압농축하는 단계에 의해 수득될 수 있다."Post-harvest mushroom waste medium extract during artificial cultivation of shiitake mushrooms" included in the composition of the present invention collects and pulverizes discarded mushroom waste media after harvesting shiitake mushrooms 1 to 3 times after inoculation and cultivation of shiitake mushrooms. doing; It can be obtained by extracting the pulverized mushroom waste medium with a solvent, filtering the extract using a filter net of 0.06 mm or less, and concentrating it under reduced pressure.
본 발명에서 사용되는 용매는 물(냉수, 열수), 주정, 탄소수 1~4의 무수 또는 함수 저급 알코올(메탄올, 에탄올, 주정, 프로판올, 부탄올 등), 상기 저급알코올과 물과의 혼합용매 등을 이용할 수 있으며, 열수 추출이 가장 바람직하다.The solvent used in the present invention is water (cold water, hot water), alcohol, anhydrous or hydrous lower alcohol having 1 to 4 carbon atoms (methanol, ethanol, alcohol, propanol, butanol, etc.), a mixed solvent of the lower alcohol and water, etc. It can be used, and hot water extraction is most preferred.
상기 추출물, 바람직하게는 열수 추출물은 헥센, 에틸아세테이트 및 부탄올의 유기용매로 순차 또는 각각 분획하여 헥센 분획물, 에틸아세테이트 분획물, 부탄올 분획물 및 유기용매 분획 후의 물 잔류물을 추가적으로 수득할 수 있다.The extract, preferably a hot water extract, may be sequentially or individually fractionated with organic solvents of hexene, ethyl acetate and butanol to additionally obtain a hexene fraction, an ethyl acetate fraction, a butanol fraction, and a water residue after organic solvent fractionation.
본 발명의 표고버섯 인공 재배 시의 버섯 수확 후 버섯폐배지 추출물 및 이의 활성 분획물은 감압건조 및 동결건조, 또는 분무건조 등과 같은 통상적인 분말화 과정을 거쳐 분말로 제조될 수 있다. 이들은 혈장 내의 다양한 분해효소에 분해되지 않으며, 100 ℃의 열처리와 pH 2의 인체 위 내의 pH에서도 활성을 유지한다.After mushroom harvesting in artificial cultivation of shiitake mushrooms of the present invention, the mushroom waste medium extract and its active fraction may be prepared into powder through a conventional powdering process such as reduced pressure drying, freeze drying, or spray drying. They are not degraded by various degrading enzymes in plasma, and maintain their activity even in heat treatment at 100 ° C and pH in the human stomach of pH 2.
본 발명의 표고버섯 재배 후의 버섯폐배지의 열수 추출물의 에틸아세테이트 분획물은 강력한 항균 활성을 나타내어, 병원성 세균의 감염의 예방 또는 치료와 관련되는 약학 조성물, 건강 기능 식품, 화장료 조성물, 동물 사료 조성물, 식품, 화장품, 동물 사료용 첨가물 및 각종 청결제의 소재로 사용될 수 있다.The ethyl acetate fraction of the hot water extract of the mushroom waste medium after cultivation of the shiitake mushroom of the present invention exhibits strong antibacterial activity, and is suitable for pharmaceutical compositions, health functional foods, cosmetic compositions, animal feed compositions, and foods related to the prevention or treatment of pathogenic bacterial infections. , it can be used as a material for cosmetics, additives for animal feed, and various cleaners.
바람직한 구체예로서, 본 발명의 항균 조성물은 약학적 조성물의 용도로서 적용될 수 있다. As a preferred embodiment, the antibacterial composition of the present invention can be applied as a pharmaceutical composition.
상기 약학적 조성물은 각각의 사용 목적에 맞게 통상의 방법에 따라 산제, 과립제, 정제, 캡슐제, 현탁제, 에멀젼, 시럽, 에어로졸 등의 경구 제형, 멸균 주사용액의 주사제 등 다양한 형태로 제형화하여 사용할 수 있으며, 경구 투여하거나 정맥 내, 복강 내, 피하, 직장, 국소 투여 등을 포함한 다양한 경로를 통해 투여될 수 있다. 이러한 약학적 조성물에는 추가적으로 담체, 부형제 또는 희석제 등이 더 포함될 수 있으며, 포함될 수 있는 적합한 담체, 부형제 또는 희석제의 예로는 락토오스, 덱스트로오스, 수크로오스, 솔비톨, 만니톨, 자일리톨, 에리쓰리톨, 말티톨, 전분, 아카시아 고무, 알지네이트, 젤라틴, 칼슘 포스페이트, 칼슘 실리케이트, 셀룰로스, 메틸 셀룰로스, 비정질 셀룰로스, 폴리비닐 피롤리돈, 물, 메틸하이드록시벤조에이트, 프로필하이드록시벤조에이트, 탈크, 마그네슘 스테아레이트 및 광물유 등을 들 수 있다. 또한, 본 발명의 약학적 조성물은 충전제, 항응집제, 윤활제, 습윤제, 향료, 유화제, 방부제 등을 추가로 더 포함할 수도 있다. The pharmaceutical composition is formulated in various forms such as oral formulations such as powders, granules, tablets, capsules, suspensions, emulsions, syrups, aerosols, and injections of sterile injection solutions according to conventional methods according to each purpose of use. It can be used and can be administered orally or through various routes including intravenous, intraperitoneal, subcutaneous, rectal, topical, and the like. These pharmaceutical compositions may further include carriers, excipients, or diluents, and examples of suitable carriers, excipients, or diluents that may be included include lactose, dextrose, sucrose, sorbitol, mannitol, xylitol, erythritol, maltitol, Starch, gum acacia, alginates, gelatin, calcium phosphate, calcium silicate, cellulose, methyl cellulose, amorphous cellulose, polyvinyl pyrrolidone, water, methylhydroxybenzoate, propylhydroxybenzoate, talc, magnesium stearate and mineral oil. etc. can be mentioned. In addition, the pharmaceutical composition of the present invention may further include fillers, anti-agglomerating agents, lubricants, wetting agents, flavoring agents, emulsifiers, preservatives, and the like.
본 발명의 약학적 조성물은 약제학적으로 유효한 양으로 투여한다. 본 발명에서, "약제학적으로 유효한 양"은 의학적 치료에 적용 가능한 합리적인 수혜/위험 비율로 질환을 치료하기에 충분한 양을 의미하며, 유효 용량 수준은 환자의 질환의 종류, 중증도, 약물의 활성, 약물에 대한 민감도, 투여 시간, 투여 경로 및 배출 비율, 치료 기간, 동시 사용되는 약물을 포함한 요소 및 기타 의학 분야에 잘 알려진 요소에 따라 결정될 수 있다. 본 발명의 약학적 조성물은 개별 치료제로 투여하거나 다른 치료제와 병용하여 투여될 수 있고, 종래의 치료제와 순차적으로 또는 동시에 투여될 수 있으며, 단일 또는 다중 투여될 수 있다. 상기한 요소들을 모두 고려하여 부작용 없이 최소한의 양으로 최대 효과를 얻을 수 있는 양을 투여하는 것이 중요하며, 이는 당업자에 의해 용이하게 결정될 수 있다. The pharmaceutical composition of the present invention is administered in a pharmaceutically effective amount. In the present invention, "pharmaceutically effective amount" means an amount sufficient to treat a disease with a reasonable benefit / risk ratio applicable to medical treatment, and the effective dose level is the type, severity, drug activity, It may be determined according to factors including sensitivity to the drug, time of administration, route of administration and excretion rate, duration of treatment, drugs used concurrently, and other factors well known in the medical field. The pharmaceutical composition of the present invention may be administered as an individual therapeutic agent or in combination with other therapeutic agents, may be administered sequentially or simultaneously with conventional therapeutic agents, and may be administered single or multiple times. Considering all of the above factors, it is important to administer an amount that can obtain the maximum effect with the minimum amount without side effects, which can be easily determined by those skilled in the art.
본 발명의 약학적 조성물에서 항균 활성을 갖는 성분의 유효량은 환자의 나이, 성별, 체중에 따라 달라질 수 있으며, 일반적으로는 체중 당 1 내지 5,000mg, 바람직하게는 100 내지 3,000mg을 매일 또는 격일 투여하거나 1일 1 내지 3회로 나누어 투여할 수 있다. 그러나, 투여 경로, 질병의 중증도, 성별, 체중, 연령 등에 따라서 증감될 수 있으므로 상기 투여량이 어떠한 방법으로도 본 발명의 범위를 한정하는 것은 아니다. 본 발명의 약학적 조성물은 다양한 경로를 통하여 대상에 투여될 수 있다. 투여의 모든 방식은 예상될 수 있는데, 예를 들면, 경구, 직장 또는 정맥, 근육, 피하, 자궁내 경막 또는 뇌혈관 내(intra-cerebroventricular) 주사에 의해 투여될 수 있다. 본 발명에서 "투여"는 임의의 적절한 방법으로 환자에게 소정의 물질을 제공하는 것을 의미하며, 본 발명의 약학적 조성물의 투여 경로는 목적 조직에 도달할 수 있는 한 일반적인 모든 경로를 통하여 경구 또는 비경구 투여될 수 있다. 또한, 본 발명의 조성물은 유효성분을 표적 세포로 전달할 수 있는 임의의 장치를 이용해 투여될 수도 있다. 본 발명에서 "대상"은, 특별히 한정되는 것은 아니지만, 예를 들어, 인간, 원숭이, 소, 말, 양, 돼지, 닭, 칠면조, 메추라기, 고양이, 개, 마우스, 쥐, 토끼 또는 기니아 피그를 포함하고, 바람직하게는 포유류, 보다 바람직하게는 인간을 의미한다. The effective amount of the component having antibacterial activity in the pharmaceutical composition of the present invention may vary depending on the patient's age, sex, and weight, and is generally administered in an amount of 1 to 5,000 mg per body weight, preferably 100 to 3,000 mg per day or every other day. Alternatively, it may be divided into 1 to 3 doses per day. However, since it may increase or decrease according to the route of administration, severity of disease, sex, weight, age, etc., the dosage is not limited to the scope of the present invention in any way. The pharmaceutical composition of the present invention may be administered to a subject through various routes. All modes of administration can be envisaged, for example by oral, rectal or intravenous, intramuscular, subcutaneous, intrauterine intrathecal or intra-cerebroventricular injection. In the present invention, "administration" means providing a predetermined substance to a patient by any suitable method, and the route of administration of the pharmaceutical composition of the present invention is oral or parenteral through all common routes as long as it can reach the target tissue. can be administered orally. In addition, the composition of the present invention may be administered using any device capable of delivering active ingredients to target cells. In the present invention, "subject" includes, but is not particularly limited to, for example, human, monkey, cow, horse, sheep, pig, chicken, turkey, quail, cat, dog, mouse, rat, rabbit or guinea pig. and, preferably, a mammal, more preferably a human.
바람직한 구체예로서, 본 발명의 항균 조성물은 건강 기능 식품의 용도로서 적용될 수 있다. As a preferred embodiment, the antibacterial composition of the present invention can be applied as a health functional food.
본 발명의 건강 기능 식품은 병원성 미생물의 감염 우려가 높은 대상으로부터 감염의 가능성을 낮추어 줄 수 있는 효과적인 식품 및 음료 등에 다양하게 이용될 수 있다. 본 발명의 항균 활성이 우수한 유효성분을 포함하는 식품으로는, 예를 들어, 각종 식품류, 음료, 껌, 차, 비타민 복합제, 건강보조 식품류 등이 있고, 분말, 과립, 정제, 캡슐 또는 음료인 형태로 사용할 수 있다. 본 발명의 활성 성분은 일반적으로 전체 식품 중량의 0.01 내지 15중량%로 가할 수 있으며, 건강음료 조성물은 100 ml를 기준으로 0.02 내지 10g, 바람직하게는 0.3 내지 1g의 비율로 가할 수 있다. 본 발명의 건강 기능 식품은 지시된 비율로 필수 성분으로서 상기 화합물을 함유하는 것 외에 식품학적으로 허용 가능한 식품보조 첨가제, 예컨대, 천연 탄수화물 및 다양한 향미제 등을 추가 성분으로서 함유할 수 있다. 상기 천연 탄수화물의 예로는 포도당, 과당 등의 단당류, 말토오스, 수크로오스 등의 이당류 및 덱스트린, 시클로덱스트린 등의 다당류와 같은 통상적인 당 및 자일리톨, 소르비톨, 에리쓰리톨 등의 당알코올이 있다. 상기 향미제로는 타우마틴, 레바우디오시드 A, 글리시르히진, 사카린, 아스파르탐 등을 사용할 수 있다. 상기 향미제의 비율은 본 발명의 건강 기능 식품 100ml당 일반적으로 약 1 내지 20g, 바람직하게는 약 5 내지 12g을 사용한다. 상기 외에 본 발명의 건강 기능 식품은 여러 가지 영양제, 비타민, 광물, 합성 풍미제 및 천연 풍미제 등의 풍미제, 착색제 및 중진제, 펙트산 및 그의 염, 알긴산 및 그의 염, 유기산, 보호성 콜로이드 증점제, pH 조절제, 안정화제, 방부제, 글리세린, 알코올, 탄산음료에 사용되는 탄산화제 등을 함유할 수 있다. 그 밖에 본 발명의 건강 기능 식품은 천연 과일 주스 및 과일 주스 음료 및 야채 음료 등의 제조를 위한 과육을 함유할 수도 있다. 이러한 성분은 독립적으로 또는 조합하여 사용할 수 있다. 이러한 첨가제의 비율은 본 발명의 상기 활성 분획물 100 중량부 당 0.01 내지 약 20중량부의 범위에서 선택되는 것이 일반적이다. The health functional food of the present invention can be used in various ways such as effective foods and beverages that can lower the possibility of infection from a subject at high risk of infection by pathogenic microorganisms. Foods containing the active ingredient with excellent antibacterial activity of the present invention include, for example, various foods, beverages, gum, tea, vitamin complexes, health supplements, etc., in the form of powders, granules, tablets, capsules or beverages can be used as The active ingredient of the present invention may generally be added in an amount of 0.01 to 15% by weight of the total food weight, and the health drink composition may be added in an amount of 0.02 to 10g, preferably 0.3 to 1g, based on 100 ml. In addition to containing the above compound as an essential component in the indicated ratio, the health functional food of the present invention may contain food additives such as natural carbohydrates and various flavors as additional components. Examples of the natural carbohydrate include monosaccharides such as glucose and fructose, disaccharides such as maltose and sucrose, polysaccharides such as dextrin and cyclodextrin, and sugar alcohols such as xylitol, sorbitol, and erythritol. Thaumatin, rebaudioside A, glycyrrhizin, saccharin, aspartame, etc. may be used as the flavoring agent. The proportion of the flavoring agent is generally about 1 to 20g, preferably about 5 to 12g, per 100ml of the health functional food of the present invention. In addition to the above, the health functional food of the present invention is various nutrients, vitamins, minerals, flavors such as synthetic flavors and natural flavors, colorants and enhancers, pectic acid and its salts, alginic acid and its salts, organic acids, protective colloids It may contain thickeners, pH adjusters, stabilizers, preservatives, glycerin, alcohol, carbonation agents used in carbonated beverages, and the like. In addition, the health functional food of the present invention may contain fruit flesh for production of natural fruit juice, fruit juice beverages, and vegetable beverages. These components may be used independently or in combination. The proportion of these additives is generally selected from the range of 0.01 to about 20 parts by weight per 100 parts by weight of the active fraction of the present invention.
바람직한 구체예로서, 본 발명의 항균 조성물은 화장료 조성물의 용도로서 적용될 수 있다. As a preferred embodiment, the antibacterial composition of the present invention can be applied as a cosmetic composition.
본 발명의 화장료 조성물은 본 발명의 항균 활성이 우수한 성분을, 예를 들어, 에센스, 영양크림, 바디로션, 린스, 샴푸, 바디클렌저, 로션, 스킨, 마스크팩 등의 형태로 제조될 수 있으며, 여기에 한정되는 것은 아니다. 상기 형태로의 제조는 당업자에게 알려진 다양한 제조 공정에 따라 용이하게 수행될 수 있을 것이다.The cosmetic composition of the present invention can be prepared in the form of an ingredient having excellent antibacterial activity of the present invention, for example, essence, nutrient cream, body lotion, rinse, shampoo, body cleanser, lotion, toner, mask pack, etc. It is not limited here. Preparation in the form can be easily carried out according to various manufacturing processes known to those skilled in the art.
바람직한 구체예로서, 본 발명의 항균 조성물은 동물 사료 조성물의 용도로서 적용될 수 있다. As a preferred embodiment, the antibacterial composition of the present invention can be applied as an animal feed composition.
상기 동물은 식물에 대응하는 생물군으로 주로 유기물을 영양분으로 섭취하며, 소화나 배성 및 호흡기관이 분화되어 있는 것을 말하고, 구체적으로는 극피동물, 갑각류, 연체동물, 어류, 양서류, 파충류, 조류, 포유류일 수 있으며, 바람직하게는 성게류 또는 해삼류와 같은 극피동물, 게, 새우, 대하 등의 갑각류를 포함하는 절지동물, 두족류, 복족류 또는 이매패류 등의 연체동물, 참돔, 도미, 대구, 가자미, 넙치 등의 어류, 꿩 또는 닭 등의 가금류를 포함하는 조류 또는 돼지, 소, 염소 등의 포유류일 수 있다.The animals are a group of organisms corresponding to plants and mainly consume organic matter as nutrients, and have differentiated digestive, embryonic, and respiratory organs. Specifically, echinoderms, crustaceans, mollusks, fish, amphibians, reptiles, birds It may be mammals, preferably echinoderms such as sea urchins or sea cucumbers, arthropods including crustaceans such as crabs, shrimps and lobsters, molluscs such as cephalopods, gastropods or bivalves, red sea bream, sea bream, cod, flounder , fish such as halibut, birds including poultry such as pheasants or chickens, or mammals such as pigs, cows, and goats.
상기 동물 사료용 조성물은 본 발명의 항균 성분을 포함하는 항균 조성물에 곡물, 식물성 단백질 사료, 동물성 단백질 사료, 당분 또는 유제품을 추가로 포함할 수 있다. 상기 곡물은 구체적으로는 분쇄 또는 파쇄된 밀, 귀리, 보리, 옥수수 및 쌀일 수 있고, 상기 식물성 단백질 사료는 구체적으로는 평지, 콩 및 해바라기를 주성분으로 하는 것일 수 있으며, 상기 동물성 단백질 사료는 구체적으로는 혈분, 육분, 골분 및 생선분일 수 있고, 상기 당분 또는 유제품은 구체적으로는 각종 분유 및 유장 분말로 이루어지는 건조성분일 수 있다.The composition for animal feed may further include grain, vegetable protein feed, animal protein feed, sugar or dairy products to the antibacterial composition containing the antibacterial component of the present invention. The grain may be specifically ground or crushed wheat, oats, barley, corn and rice, the vegetable protein feed may be specifically rapeseed, soybean and sunflower as main components, and the animal protein feed is specifically may be blood meal, meat meal, bone meal, and fish meal, and the sugar or dairy product may be a dry ingredient composed of various powdered milk and whey powder.
상기 동물 사료용 조성물은 추가로 영양 보충제, 소화 및 흡수 향상제, 성창 촉진제 또는 질병 예방제와 같은 성분을 함께 사용할 수 있다.The composition for animal feed may additionally use ingredients such as nutritional supplements, digestion and absorption enhancers, growth promoters, or disease preventive agents.
본 발명의 동물 사료용 조성물에 포함되는 항균 조성물은 사료의 사용목적 및 사용조건에 따라 달라질 수 있으며, 일 예로 최종 생산된 사료 1kg을 기준으로 상기 항균성 조성물이 0.1g 내지 100g 포함될 수 있다.The antimicrobial composition included in the composition for animal feed of the present invention may vary depending on the purpose and conditions of use of the feed, and for example, 0.1 g to 100 g of the antimicrobial composition may be included based on 1 kg of the final produced feed.
또한, 상기 사료 조성물은 성분들의 분쇄 정도에 따라 경점성의 조립 또는 과립 물질로 제조될 수 있으며, 상기 조성물은 메쉬로 공급되거나 추가 가공 및 포장을 위해 원하는 분리된 형상으로 형성시킬 수 있으며, 저장을 위해 펠렛화, 팽창화 또는 압출 공정을 거칠 수 있으며, 저장의 용이성을 위해 바람직하게는 과잉의 물을 건조 제거될 수 있다.In addition, the feed composition may be made of a hard granular or granular material depending on the degree of grinding of the ingredients, and the composition may be supplied as a mesh or formed into a desired separated shape for further processing and packaging, and may be stored It may be pelletized, expanded, or extruded for storage, and excess water may preferably be removed by drying for ease of storage.
바람직한 구체예로서, 본 발명의 항균 조성물은 식품, 화장품 또는 동물 사료용 첨가제의 용도로서 적용될 수 있다. As a preferred embodiment, the antibacterial composition of the present invention can be applied as an additive for food, cosmetics or animal feed.
상기 첨가제는 사용되는 식품, 화장품 또는 동물 사료에 보존성을 부여하기 위하여 첨가되는 것으로서, 상기 항균 조성물은 천연성분으로 인체에 유해하지 아니하므로 식품, 화장품 또는 동물용 사료에 안전하게 적용할 수 있다.The additive is added to impart preservation to food, cosmetics, or animal feed, and the antibacterial composition is a natural component and is not harmful to the human body, so it can be safely applied to food, cosmetics, or animal feed.
상기 첨가제는 그 사용 목적 및 용도에 따라 적의 선택된 추가적인 여타의 첨가물을 추가로 더 포함할 수 있으며, 통상적으로 사용되는 첨가물 등 다른 유효성분이나 색소, 계면활성제, 방부제 등의 첨가물과 혼합하여 사용할 수 있다. 상기 첨가제는 그 사용 목적 및 용도에 따라 분말화, 과립화, 정제화 또는 액상화하여 제조될 수 있으며, 제품화를 위하여 포장을 위해 통상의 방법을 사용할 수 있다.The additive may further include other additives appropriately selected depending on the purpose and purpose of use, and may be mixed with other active ingredients such as commonly used additives or additives such as pigments, surfactants, and preservatives. . The additives may be prepared by powdering, granulating, tableting or liquefying according to the purpose and purpose of use, and a conventional method may be used for packaging for commercialization.
또한, 상기 첨가제 중 항균 조성물의 사용량은 사용목적과 적용형태, 적용부위, 적용대상물품 등에 따라 적절히 사용될 수 있으며, 예를 들면 전체 조성물 기준으로 0.01 중량부 내지 50 중량부를 포함할 수 있으나, 상기 항균 조성물의 함량은 이에 한정되는 것은 아니다. 본원 발명의 항균 조성물은 천연성분으로 인체에 유해하지 아니하므로, 그 제품의 특성상 적용목적을 달성할 수 있는 경우라면 다양한 양으로 사용가능하다.In addition, the amount of the antimicrobial composition among the additives may be appropriately used depending on the purpose of use, application form, application area, application target product, etc., for example, may include 0.01 part by weight to 50 parts by weight based on the total composition, but the antibacterial The content of the composition is not limited thereto. Since the antibacterial composition of the present invention is a natural component and is not harmful to the human body, it can be used in various amounts if the purpose of application can be achieved due to the nature of the product.
바람직한 구체예로서, 본 발명은 항균 조성물은 세정제의 용도로서 적용될 수 있다.As a preferred embodiment, the antibacterial composition of the present invention can be applied for use as a detergent.
상기 세정제는 바람직하게는 의류용 세제, 식기 세척제, 식품 세정제, 가전제품 세척제일 수 있다. 상기 항균 조성물은 천연성분으로 인체에 유해하지 아니하므로, 더욱 바람직하게는 주방용품 또는 식품세척 등을 포함하는 각종 생활용품의 세척이나 항균성 부여를 목적으로 사용할 수 있다.The detergent may preferably be a detergent for clothes, a dish detergent, a food detergent, or a home appliance detergent. Since the antimicrobial composition is a natural component and is not harmful to the human body, more preferably, it can be used for cleaning or imparting antibacterial properties to various household products including kitchen utensils or food washing.
상기 세정제는 그 사용 목적 및 용도에 따라 적의 선택된 첨가물을 추가로 포함할 수 있으며, 통상적으로 사용되는 세정제 등 다른 유효성분이나 색소, 계면활성제, 방부제 등의 첨가제와 혼합하여 사용할 수 있다. 상기 세정제는 그 사용 목적 및 용도에 따라 분말화, 과립화, 정제화 또는 액상화하여 제조될 수 있으며, 제품화를 위하여 포장을 위해 통상의 방법을 사용할 수 있다.The detergent may further include appropriately selected additives depending on the purpose and purpose of use, and may be mixed with other active ingredients such as commonly used detergents or additives such as pigments, surfactants, and preservatives. The detergent may be prepared by powdering, granulating, tableting or liquefying depending on the purpose and purpose of use, and a conventional method may be used for packaging for commercialization.
또한, 상기 세정제 중 항균성 조성물의 사용량은 사용목적과 적용형태, 적용부위, 적용대상물품 등에 따라 적절히 사용될 수 있으며, 예를 들면 전체 조성물 기준으로 0.01 중량부 내지 50 중량부를 포함할 수 있으나, 상기 항균성 조성물의 함량은 이에 한정되는 것은 아니다. 본원 발명의 항균성 조성물은 천연성분으로 인체에 유해하지 아니하므로, 그 제품의 특성상 적용목적을 달성할 수 있는 경우라면 다양한 양으로 사용가능하다.In addition, the amount of the antimicrobial composition used in the cleaning agent may be appropriately used depending on the purpose of use, application form, application area, application target product, etc., for example, may include 0.01 part by weight to 50 parts by weight based on the total composition, but the antibacterial The content of the composition is not limited thereto. Since the antimicrobial composition of the present invention is a natural component and is not harmful to the human body, it can be used in various amounts if the purpose of application can be achieved due to the nature of the product.
바람직한 구체예로서, 본 발명은 항균 조성물은 청결제의 용도로서 적용될 수 있다.As a preferred embodiment, the antibacterial composition of the present invention can be applied for use as a cleaning agent.
상기 청결제는 일상 생활에서 흔히 감염되기 쉬운 병원성 미생물의 체내 감염을 예방 차원에서 수행하기 위하여 간단한 방법으로 손, 구강 또는 여성용 청결제 등의 용도로 사용될 수 있다. 상기 항균 조성물은 천연성분으로 인체에 유해하지 아니하므로 손, 구강, 여성용 청결제로 사용하는 경우에도 안전하게 적용할 수 있다.The cleaning agent can be used as a hand, oral, or feminine cleaner in a simple way to prevent infection in the body of pathogenic microorganisms that are often easily infected in daily life. Since the antibacterial composition is a natural component and is not harmful to the human body, it can be safely applied even when used as a hand, oral, and feminine cleaner.
상기 청결제는 그 사용 목적 및 용도에 따라 적의 선택된 추가적인 여타의 첨가물을 추가로 더 포함할 수 있으며, 통상적으로 사용되는 첨가물 등 다른 유효성분이나 색소, 계면활성제, 방부제 등의 첨가물과 혼합하여 사용할 수 있다. 상기 청결제는 그 사용 목적 및 용도에 따라 분말화, 과립화, 정제화 또는 액상화하여 제조될 수 있으나, 액상형의 경우 편리한 사용이 가능할 것이다.The cleaner may further include other additives appropriately selected according to the purpose and purpose of use, and may be mixed with other active ingredients such as commonly used additives or additives such as pigments, surfactants, and preservatives. . The cleaner may be prepared by powdering, granulating, tableting or liquefying depending on the purpose and purpose of use, but in the case of a liquid type, convenient use will be possible.
또한, 상기 청결제 중 항균 조성물의 사용량은 사용목적과 적용형태, 적용부위 등에 따라 적절히 사용될 수 있으며, 예를 들면 전체 조성물 기준으로 0.01중량부 내지 50중량부를 포함할 수 있으나, 상기 항균 조성물의 함량은 이에 한정되는 것은 아니다. 본원 발명의 항균 조성물은 천연성분으로 인체에 유해하지 아니하므로, 그 제품의 특성상 적용목적을 달성할 수 있는 경우라면 다양한 양으로 사용가능하다.In addition, the amount of the antimicrobial composition used in the cleaner may be appropriately used depending on the purpose of use, application form, application area, etc., for example, may include 0.01 part by weight to 50 parts by weight based on the total composition, but the content of the antibacterial composition It is not limited to this. Since the antibacterial composition of the present invention is a natural component and is not harmful to the human body, it can be used in various amounts if the purpose of application can be achieved due to the nature of the product.
이하에서는 실시예를 통하여 본 발명을 더욱 상세하게 설명한다. 하기 실시예는 본 발명의 바람직한 일 구체예일 뿐이며, 본 발명의 권리범위가 하기 실시예의 범위로 한정되는 것은 아니다.Hereinafter, the present invention will be described in more detail through examples. The following examples are only one preferred embodiment of the present invention, and the scope of the present invention is not limited to the scope of the following examples.
[실시예][Example]
실시예 1: 표고버섯 재배용 살균 배지, 1 회 수확 후 버섯폐배지, 3 회 수확 후 버섯폐배지 및 표고버섯의 이화학적 특성 분석 및 색 분석 Example 1: Physicochemical Characteristics Analysis and Color Analysis of Sterilized Medium for Shiitake Mushroom Cultivation, Wasted Mushroom Medium after 1st Harvest, Wasted Mushroom Medium after 3rd Harvest and Shiitake Mushrooms
2017년 중국 금전시에 제조된 표고버섯 재배용 배지(조성: 참나무 80%, 밀기울 19%, 식용석고 1%)를 압착 밀봉하고, 이를 100℃에서 8시간 살균하여 무균 배지를 조제한 다음, 표고버섯 종균 808을 접종하여 약 120일간 배양실 배양하고, 다시 재배사에서 40일간 23℃, 습도 70% 조건에서 배양한 후 발생 작업을 하였다. 이후, 생성된 표고버섯을 1회 수확(1주기 수확)한 이후 1회 수확 후 버섯폐배지를 준비하였으며, 이후 표고버섯을 재발생하여 2회(2주기 수확) 및 3회 수확(3주기 수확)하였으며, 이후 남은 3회 수확 후 버섯폐배지를 회수하였다(도 1).A medium for growing shiitake mushrooms (composition: oak 80%, bran 19%, edible gypsum 1%) manufactured in Jinzhen, China in 2017 was pressed and sealed, sterilized at 100 ° C for 8 hours to prepare an aseptic medium, and then shiitake mushroom spawn 808 was inoculated and cultured in a culture room for about 120 days, and then cultured again in a cultivation house at 23 ° C and 70% humidity for 40 days, and then generation work was performed. Afterwards, the generated shiitake mushrooms were harvested once (1st cycle harvest), and after the first harvest, a mushroom waste medium was prepared, and then the shiitake mushrooms were regenerated and harvested 2 times (2nd cycle harvest) and 3rd harvest (3rd cycle harvest) After harvesting the remaining three times, the mushroom waste medium was recovered (FIG. 1).
상기의 살균 배지, 1회 수확 후 버섯폐배지, 3회 수확 후 버섯폐배지 및 수확한 표고버섯의 사진도는 도 1에 나타내었으며, 이의 조단백, 조탄수화물, 조지질, 회분 및 수분함량을 식품공전에 따라 측정하여 열량을 표 1에 나타내었다.Photographs of the sterilization medium, the waste mushroom medium after the first harvest, the waste mushroom medium after the third harvest, and the harvested shiitake mushrooms are shown in Figure 1, and their crude protein, crude carbohydrates, crude lipid, ash and moisture content are shown in food Table 1 shows the amount of heat measured according to revolution.
[표 1] 살균 배지, 1 회 수확 후 버섯폐배지, 3 회 수확 후 버섯폐배지 및 표고버섯의 영양 성분 분석[Table 1] Nutrient composition analysis of sterilization medium, waste mushroom medium after 1st harvest, waste mushroom medium after 3rd harvest and shiitake mushrooms
수분함량은 초기 46.8%에서 1차 수확 후 52.6%, 3차 수확 후 54.0%로 변화하였으며, 조단백은 살균 배지에서 3.1%이었으나, 수확 후 버섯폐배지에서는 6.75~7.5%로 2배 이상 증가하였다. 회분 함량 역시 살균 배지에서는 6.55%이었으나, 수확 후 버섯폐배지에서는 14.77~14.93%로 급격히 증가하였다. 표고버섯의 회분 함량은 0.8%를 나타내었다. 반면, 조지질 함량은 변화가 미미하였으며, 조탄수화물은 1차 수확 후에는 43.3%에서 25.7%로 급격히 감소하였다가 3차 수확 후에는 23.2%를 나타내었다. 이는 1차, 3차 수확 후의 배지가 다량의 표고버섯 균사체 및 자실체를 함유하여 나타난 결과로 판단된다. Moisture content changed from 46.8% at the beginning to 52.6% after the first harvest and 54.0% after the third harvest. The ash content was also 6.55% in the sterilized medium, but increased rapidly to 14.77~14.93% in the mushroom waste medium after harvest. The ash content of shiitake mushrooms was 0.8%. On the other hand, the change in the crude lipid content was insignificant, and the crude carbohydrate decreased rapidly from 43.3% to 25.7% after the first harvest, and then showed 23.2% after the third harvest. This is judged to be the result of the medium after the 1st and 3rd harvests containing a large amount of shiitake mushroom mycelium and fruiting bodies.
한편, 표고버섯 배지 및 표고버섯의 분쇄 후 각각의 시료의 색 분석 결과, 살균 배지는 상대적으로 낮은 명도, 적색도, 황색도를 나타내어 색차는 73.3을 나타내었으며, 1회 수확 후 버섯폐배지 및 3회 수확 후 버섯폐배지는 각각 46.3 및 44.75를 나타내어 유사한 명도, 적색도, 황색도를 나타내었다(표 2). 이때, 색차 분석은 Hunter Color Difference meter(Super color SP-80 Colormeter, Tokyo Denshoku Co., Japan)를 이용하여 측정하였으며, 명도(백색 100 ~ 0 검정색), 적색도(적색 100 ~ -80 녹색), 황색도(황색 70 ~ -80 검정색)를 측정하였다. 이때, 표준백판의 색도는 L 값이 92.44, a 값이 -0.06, b 값이 1.35로 기준을 정하였으며, 시료 당 3회 측정하여 평균값을 구하여 나타내었고, 색차(△E)는 다음의 식을 이용하여 계산하였다.On the other hand, as a result of color analysis of each sample after grinding the shiitake mushroom medium and shiitake mushrooms, the sterilized medium showed relatively low brightness, redness, and yellowness, and the color difference was 73.3. After one harvest, the mushroom waste medium and 3 After harvesting, the mushroom waste medium showed 46.3 and 44.75, respectively, showing similar brightness, redness, and yellowness (Table 2). At this time, color difference analysis was measured using a Hunter Color Difference meter (Super color SP-80 Colormeter, Tokyo Denshoku Co., Japan), brightness (white 100 ~ 0 black), redness (red 100 ~ -80 green), Yellowness (yellow 70 to -80 black) was measured. At this time, the chromaticity of the standard white board was set as L value of 92.44, a value of -0.06, and b value of 1.35, and was measured three times per sample to obtain an average value. It was calculated using
[표 2] 살균 배지, 1회 수확 후 버섯폐배지, 3회 수확 후 버섯폐배지 및 표고버섯 분말의 색차 분석[Table 2] Color difference analysis of sterilization medium, waste mushroom medium after 1st harvest, waste mushroom medium after 3rd harvest and shiitake mushroom powder
실시예 2: 살균 배지, 1회 수확 후 버섯폐배지, 3회 수확 후 버섯폐배지 및 표고버섯 열수 추출물 조제 및 이의 이화학적 특성 분석 Example 2: Sterilization medium, mushroom waste medium after 1st harvest, mushroom waste medium after 3rd harvest and shiitake mushroom hot water extract preparation and physicochemical characterization analysis thereof
실시예 1에서 준비된 살균 배지, 1회 수확 후 버섯폐배지, 3회 수확 후 버섯폐배지, 표고버섯 건조 분말을 대상으로 열수 추출물을 조제하였다. 각각의 시료에 대해 10배의 증류수를 가하고, 100℃에서 1시간 추출한 후, 얻어진 추출물의 pH, brix 및 색차를 측정하였으며, 그 결과는 표 3에 나타내었다.Hot water extracts were prepared from the sterilization medium prepared in Example 1, the waste mushroom medium after the first harvest, the waste mushroom medium after the third harvest, and dried shiitake mushroom powder. After adding 10 times distilled water to each sample and extracting at 100 ° C. for 1 hour, the pH, brix and color difference of the obtained extract were measured, and the results are shown in Table 3.
[표 3] 살균 배지, 1회 수확 후 버섯폐배지, 3회 수확 후 버섯폐배지, 표고버섯 분말 열수 추출물의 이화학적 특성 [Table 3] Physicochemical properties of sterilization medium, mushroom waste medium after 1st harvest, mushroom waste medium after 3rd harvest, and shiitake mushroom powder hot water extract
살균 배지의 초기 pH는 5.6이었으나, 1회 및 3회 수확 후 버섯폐배지의 경우, 4.6으로 감소되었다. 또한, 살균 배지의 초기 brix는 6.0에서 1회 및 3회 수확 후 버섯폐배지는 16.0과 20.0으로 증가되었으며, 산도 역시 초기 0에서 1회 및 3회 수확 후 버섯폐배지는 0.83, 1.40으로 증가하였다. 반면, 열수 추출물의 색 분석에서는 수확 횟수 증가에 따라 명도와 적색도의 감소 및 황색도의 증가가 나타났으며, 색차는 초기 살균 배지 76.8에서 3차 수확 후 버섯폐배지에서는 78.2로 증가하였다. 즉, 표고버섯의 배양이 길어질수록 버섯폐배지는 산도 및 brix의 증가, 황색도의 증가가 두드러졌다(도 2). The initial pH of the sterilization medium was 5.6, but it was reduced to 4.6 in the waste mushroom medium after the first and third harvests. In addition, the initial brix of the sterilization medium increased from 6.0 to 16.0 and 20.0 for the mushroom waste medium after the first and third harvests, and the acidity also increased from 0 initially to 0.83 and 1.40 after the first and third harvests. . On the other hand, in the color analysis of the hot water extract, the brightness and redness decreased and the yellowness increased as the number of harvests increased, and the color difference increased from 76.8 in the initial sterilization medium to 78.2 in the mushroom waste medium after the third harvest. That is, the longer the culture of shiitake mushrooms, the higher the acidity, brix, and yellowness of the mushroom waste medium (FIG. 2).
실시예 3: 살균 배지, 1회 수확 후 버섯폐배지, 3회 수확 후 버섯폐배지 및 표고버섯 열수 추출물의 성분 분석Example 3: Component analysis of sterilization medium, mushroom waste medium after 1 harvest, mushroom waste medium after 3 harvest, and hot water extract of shiitake mushroom
실시예 2에서 제조된 열수 추출물을 필터링한 후, 감압 농축하여 분말로 제조하였으며, 각각의 추출 효율 및 추출물의 성분 분석 결과는 표 4에 나타내었다. 성분 분석으로 총 폴리페놀, 총 플라보노이드, 총당 및 환원당 함량을 측정하였다. 총 폴리페놀 함량은 추출 검액 400μl에 50μl의 Folin-ciocalteau, 100μl의 Na2CO3 포화 용액을 넣고, 실온에서 1시간 방치한 후 725nm에서 흡광도를 측정하였다. 표준시약으로는 tannic acid를 사용하였다. 총 플라보노이드 함량은 각각의 시료를 18시간 메탄올 교반 추출하고, 여과한 추출 검액 400μl에 90% diethylene glycol 4ml를 첨가하고, 다시 1N NaOH 40μl를 넣고, 37℃에서 1시간 반응 후 420nm에서 흡광도를 측정하였다. 표준시약으로는 rutin을 사용하였다. 환원당은 DNS법으로, 총당은 phenol-sulfuric acid법을 이용하여 정량하였다. After filtering the hot water extract prepared in Example 2, it was concentrated under reduced pressure to prepare a powder, and the extraction efficiency and component analysis results of each extract are shown in Table 4. Component analysis determined total polyphenol, total flavonoid, total sugar and reducing sugar content. For the total polyphenol content, 50 μl of Folin-ciocalteau and 100 μl of a saturated solution of Na 2 CO 3 were added to 400 μl of the extraction sample solution, and the absorbance was measured at 725 nm after being allowed to stand at room temperature for 1 hour. As a standard reagent, tannic acid was used. For the total flavonoid content, each sample was stirred and extracted with methanol for 18 hours, 4ml of 90% diethylene glycol was added to 400μl of the filtered extraction sample solution, 40μl of 1N NaOH was added, and the absorbance was measured at 420nm after reaction at 37℃ for 1 hour. . Rutin was used as a standard reagent. Reducing sugar was quantified using the DNS method, and total sugar was quantified using the phenol-sulfuric acid method.
[표 4] 살균 배지, 1회 수확 후 버섯폐배지, 3회 수확 후 버섯폐배지 및 표고버섯 열수 추출물의 성분 분석[Table 4] Component analysis of sterilization medium, waste mushroom medium after 1 harvest, waste mushroom medium after 3 harvests, and hot water extract of shiitake mushroom
표 4에 나타낸 바와 같이, 추출 효율은 살균 배지에서는 1%로 매우 낮았으나, 버섯폐배지는 5.1~9.2%의 높은 수율을 보였으며, 1차 수확 후 버섯폐배지(5.1%)보다 3차 수확 후 버섯폐배지(9.2%)가 약 1.8배 높은 추출 수율을 보였다. 표고버섯 건조 분말의 열수 추출물의 추출 효율은 34.7%로 매우 높았다. 이는 표고버섯 재배가 진행될수록 살균 배지의 분해로 인한 가용성 성분의 증가 때문으로 판단된다. As shown in Table 4, the extraction efficiency was very low at 1% in the sterile medium, but the mushroom waste medium showed a high yield of 5.1 to 9.2%, and the third harvest was higher than the mushroom waste medium (5.1%) after the first harvest. After the mushroom waste medium (9.2%), the extraction yield was about 1.8 times higher. The extraction efficiency of the hot water extract of dried shiitake mushroom powder was very high at 34.7%. This is considered to be due to the increase in soluble components due to the decomposition of the sterilization medium as the cultivation of shiitake mushrooms progresses.
총 폴리페놀 분석 결과, 살균 배지는 20.5mg/g을 나타내어 표고버섯보다 2.5배 높은 함량을 보였으며, 1차 및 3차 수확 후 버섯폐배지의 경우 34.9~37.5mg/g을 함유하여 살균 배지 및 수확된 표고버섯보다 월등히 높은 함량을 보였다. 이러한 패턴은 총 플라보노이드 함량 분석에서도 동일하게 나타났다(표 4). 따라서, 1차 및 3차 수확 후 버섯폐배지는 버섯 재배용 살균 배지 및 수확한 표고버섯보다 우수한 생리 활성을 나타내리라 예측되었다. As a result of total polyphenol analysis, the sterilization medium showed 20.5mg/g, which was 2.5 times higher than that of shiitake mushrooms. It showed a significantly higher content than harvested shiitake mushrooms. This pattern was also found in the total flavonoid content analysis (Table 4). Therefore, it was predicted that the mushroom waste medium after the first and third harvests would show better physiological activity than the sterilized medium for mushroom cultivation and the harvested shiitake mushrooms.
한편, 총당의 경우 살균 배지에서 가장 높은 144.9mg/g함량을 나타내었으며, 환원당의 경우에는 1차 및 3차 수확 후 버섯폐배지에서 가장 높은 53.4~54.0mg/g 함량을 보였다. 반면, 표고버섯 추출물에서는 다른 시료와 달리 매우 낮은 총당(21.6mg/g) 및 환원당(19.7mg/g) 함량을 보였다.On the other hand, in the case of total sugar, the highest content of 144.9mg / g was shown in the sterilized medium, and in the case of reduced sugar, the highest content of 53.4 ~ 54.0mg / g was shown in the mushroom waste medium after the 1st and 3rd harvest. On the other hand, shiitake mushroom extract showed very low total sugar (21.6 mg/g) and reducing sugar (19.7 mg/g) content, unlike other samples.
실시예 4: 살균 배지, 1회 수확 후 버섯폐배지, 3회 수확 후 버섯폐배지 및 표고버섯 열수 추출물의 항균 활성 Example 4: Antibacterial activity of sterilization medium, mushroom waste medium after 1 harvest, mushroom waste medium after 3 harvest and hot water extract of shiitake mushroom
실시예 2에서 조제된 다양한 추출물을 대상으로 항균 활성을 평가하여 그 결과를 표 5에 나타내었다. 이때, 항균활성은 disc diffusion법을 사용하였으며, disc당 시료 농도은 0.5mg으로 조정하였다. Antibacterial activity was evaluated for various extracts prepared in Example 2, and the results are shown in Table 5. At this time, the disc diffusion method was used for antibacterial activity, and the sample concentration per disc was adjusted to 0.5 mg.
항균 활성 평가에 사용된 균주로는, 그람 음성균으로서 Escherichia coli KCTC 1682, Proteus vulgaris KCTC 2433, Pseudomonas aeruginosa KACC 10186, Salmonella typhimurium KCTC 1926을 사용하였고, 그람 양성균으로는 Staphylococcus aureus KCTC 1916, Staphylococcus epidermidis ATCC 12228, Listeria monocytogenes KACC 10550, Bacillus subtilis KCTC 1924를 사용하였다. 한편, 항진균 활성 평가를 위해서는 Saccharomyces cerevisiae IF0 0233 및 캔디다증 진균감염증 원인균 Candida albicans KCTC 1940을 사용하였다. Escherichia coli KCTC 1682, Proteus vulgaris KCTC 2433, Pseudomonas aeruginosa KACC 10186, and Salmonella typhimurium KCTC 1926 were used as strains for antibacterial activity evaluation, and Staphylococcus aureus KCTC 1916 and Staphylococcus epidermidis ATCC 12228 were used as gram-positive bacteria. , Listeria monocytogenes KACC 10550 and Bacillus subtilis KCTC 1924 were used. On the other hand, for evaluation of antifungal activity, Saccharomyces cerevisiae IF0 0233 and Candida albicans KCTC 1940, the causative agent of candidiasis fungal infection, were used.
먼저, 항세균 활성 평가의 경우, Nutrient broth(Difco Co., USA)에 각각의 세균을 접종하여 37℃에서 24시간 동안 배양한 후, 각 균주를 O.D.600 0.1로 조정하여 Nutrient agar(Difco Co., USA) 배지를 포함하는 멸균 petri dish(90mm, 녹십자, 한국)에 100μl씩 도말하고, 각각의 시료 5μl를 멸균 disc-paper(지름 6.5mm, Whatsman No. 2)에 가하여, 37℃에서 24시간 동안 배양하였으며, 진균의 경우에는, Sabouraud dextrose(Difco Co. USA)를 이용하여 동일한 방법으로 30℃에서 24시간 동안 배양 후, 생육저지환의 크기를 측정하여 항균활성을 평가하였다. 대조구로는 항세균제인 엠피실린(ampicillin)과 항진균제인 마이코나졸(miconazole, Sigma Co., USA)을 각각 1μg/disc 농도로 사용하였으며, 생육저지환의 크기는 육안으로 생육이 나타나지 않는 부분의 지름을 mm 단위로 측정하였고, 3회 이상 평가 후 대표 결과로 나타내었다.First, in the case of antibacterial activity evaluation, each bacterium was inoculated into Nutrient broth (Difco Co., USA) and cultured at 37° C. for 24 hours, and each strain was adjusted to an OD 600 of 0.1 to Nutrient agar (Difco Co. , USA) 100 μl each was spread on a sterile petri dish (90 mm, Green Cross, Korea) containing the medium, and 5 μl of each sample was applied to a sterile disc-paper (diameter 6.5 mm, Whatsman No. 2) for 24 hours at 37 ° C. In the case of fungi, after incubation at 30 ° C. for 24 hours in the same manner using Sabouraud dextrose (Difco Co. USA), the antibacterial activity was evaluated by measuring the size of the growth inhibition ring. As a control, antibacterial agent ampicillin and antifungal agent miconazole (Sigma Co., USA) were used at a concentration of 1 μg/disc, respectively. It was measured in mm, and it was shown as a representative result after 3 or more evaluations.
[표 5] 살균 배지, 1회 수확 후 버섯폐배지, 3회 수확 후 버섯폐배지, 수확 표고버섯 분말 열수 추출물의 항균 활성[Table 5] Sterilization medium, mushroom waste medium after 1 harvest, mushroom waste medium after 3 harvest, antibacterial activity of harvested shiitake mushroom powder hot water extract
표 5에 나타낸 바와 같이, 항세균 활성 대조구로 사용된 엠피실린(1μg/disc)의 경우 대장균(E. coli)과 녹농균(P. aeruginosa)을 제외한 모든 세균에 20.0~30.0mm의 생육억제환을 나타내어 강력한 항세균 활성을 나타냄을 확인하였으며, 항진균제인 마이코나졸(1μg/disc)의 경우 C. albicans 및 S. cerevisiae에 대해 각각 10.5mm 및 9.5mm의 생육억제환을 나타내었다. 한편, 표고버섯 재배를 위한 살균배지, 1회 수확 후 버섯폐배지, 3회 수확 후 버섯폐배지 및 표고버섯 건조분말의 열수 추출물은 500μg/disc 농도에서 실험 세균 및 진균 모두에 대해 전혀 생육억제환이 나타나지 않았다. 표고버섯 추출물의 경우 43.1mg/disc의 매우 고농도에서 항균활성이 보고(김용두 외, 2003. 한국식품저장유통학회지 10: 89-93)된 바 있으나, 이러한 고농도는 실제적 적용 가능한 부분을 넘어서므로 의미가 없는 보고이다(인간의 혈액을 4리터로 가정하는 경우, 43mg/disc 농도는 50g에 육박하는 항균제를 섭취해야 하는 농도임), 통상 1mg/disc 이하의 농도에서 활성이 인정되어야 실제 항균제로 이용 가능하다고 알려져 있으므로, 이러한 결과는 표고버섯 재배를 위한 살균배지, 1회 수확 후 버섯폐배지, 3회 수확 후 버섯폐배지 및 표고버섯 건조분말 추출물은 항세균 및 항진균 활성이 없음을 의미하고 있다.As shown in Table 5, in the case of ampicillin (1μg/disc) used as an antibacterial activity control, growth inhibitory rings of 20.0 to 30.0 mm were applied to all bacteria except Escherichia coli ( E. coli ) and P. aeruginosa ( P. aeruginosa ). In the case of myconazole (1 μg/disc), which is an antifungal agent, growth inhibitory rings of 10.5 mm and 9.5 mm were shown for C. albicans and S. cerevisiae , respectively. On the other hand, the sterilization medium for shiitake mushroom cultivation, the waste mushroom medium after the 1st harvest, the waste mushroom medium after the 3rd harvest, and the hot water extract of dried shiitake mushroom powder showed no growth inhibition for all of the experimental bacteria and fungi at a concentration of 500 μg/disc. Didn't show up. In the case of shiitake mushroom extract, antibacterial activity has been reported at a very high concentration of 43.1mg/disc (Kim Yong-doo et al., 2003. Journal of the Korean Food Storage and Distribution Society 10: 89-93), but this high concentration is beyond the practically applicable part, so it is meaningless. There is no report (assuming that human blood is 4 liters, 43mg/disc concentration is the concentration at which an antimicrobial agent should be ingested close to 50g). Normally, activity must be recognized at a concentration of 1mg/disc or less to be used as an actual antibacterial agent. Therefore, these results mean that the sterilization medium for shiitake mushroom cultivation, the waste mushroom medium after the first harvest, the waste mushroom medium after the third harvest, and the dried shiitake mushroom dry powder extract have no antibacterial and antifungal activity.
실시예 5: 표고버섯 재배를 위한 살균배지, 1회 수확 후 버섯폐배지 및 3회 수확 후 버섯폐배지 열수 추출물의 순차적 유기용매 분획물의 조제 및 성분 분석Example 5: Sterilization medium for shiitake mushroom cultivation, preparation of sequential organic solvent fractions of waste mushroom medium after 1 harvest and hot water extract of mushroom waste medium after 3 harvest and analysis of components
실시예 2에서 조제된 표고버섯 재배를 위한 살균 배지, 1회 수확 후 버섯폐배지와 3회 수확 후 버섯폐배지 열수 추출물의 활성 성분을 확인하기 위해, 각각의 추출물을 대상으로 헥센, 에틸아세테이트, 부탄올을 이용하여 순차적 유기용매 분획을 수행하였으며, 최종적으로 물 잔류물을 회수하였다. 각각의 회수된 분획물의 성분 분석은 실시예 3과 동일한 방법으로 성분을 분석하였으며, 그 결과는 표 6에 나타내었다. In order to confirm the active ingredients of the sterilization medium for shiitake mushroom cultivation prepared in Example 2, the mushroom waste medium after the first harvest and the hot water extract of the mushroom waste medium after the third harvest, hexene, ethyl acetate, Sequential organic solvent fractionation was performed using butanol, and finally a water residue was recovered. Components of each recovered fraction were analyzed in the same manner as in Example 3, and the results are shown in Table 6.
[표 6] 살균 배지, 1회 수확 후 버섯폐배지와 3회 수확 후 버섯폐배지 열수 추출물의 순차적 유기용매 분획물의 조제 및 성분 분석[Table 6] Sterilization medium, preparation and component analysis of sequential organic solvent fractions of mushroom waste medium after 1 harvest and hot water extract of mushroom waste medium after 3 harvest
표 6에 나타낸 바와 같이, 표고버섯 재배를 위한 살균배지의 열수 추출물의 경우 90.7%는 물 잔류물로 이행되어 열수 추출물은 수용성 성분을 상당량 함유하고 있음을 알 수 있었다. 반면, 1회 수확 후 버섯폐배지 및 3회 수확 후 버섯폐배지의 추출물의 물 잔류물은 각각 69.3% 및 78.3%를 나타내어 상대적으로 수용성 성분이 감소됨을 알 수 있었다. 표고버섯 재배를 위한 살균배지, 1회 수확 후 버섯폐배지 및 3회 수확 후 버섯폐배지의 열수 추출물의 지용성 헥센 분획물은 각각 열수 추출물의 0.1, 0.2 및 0.1%를 차지하여 무시할 만 하였다. 반면, 폴리페놀, 플라보노이드 성분을 고농도로 함유하고 있는 에틸아세테이트 분획물의 수율은 각각의 열수 추출물에 대해 2.7, 5.7 및 8.1%를 차지하여 재배기간이 증가할수록 점차 증가하였다. 부탄올 분획물의 경우 살균배지, 1회 수확 후 버섯폐배지 및 3회 수확 후 버섯폐배지의 열수 추출물에서 각각 10.8, 24.3 및 17.2%를 차지하여 1차 수확 후 버섯폐배지에서 가장 높은 분획 수율을 나타내었다. As shown in Table 6, in the case of the hot-water extract of the sterilization medium for shiitake mushroom cultivation, 90.7% was converted to water residue, indicating that the hot-water extract contained a significant amount of water-soluble components. On the other hand, the water residues of the mushroom waste medium after the first harvest and the extract of the mushroom waste medium after the third harvest were 69.3% and 78.3%, respectively, indicating a relatively reduced water-soluble component. The fat-soluble hexene fraction of the hot-water extract of the sterilization medium for shiitake cultivation, the waste mushroom medium after the first harvest, and the waste mushroom medium after the third harvest accounted for 0.1, 0.2, and 0.1% of the hot-water extract, respectively, and were negligible. On the other hand, the yields of ethyl acetate fractions containing high concentrations of polyphenols and flavonoids accounted for 2.7, 5.7, and 8.1% of each hot water extract, and gradually increased as the cultivation period increased. In the case of the butanol fraction, it accounted for 10.8, 24.3, and 17.2% in the hot water extract of the sterilization medium, the waste mushroom medium after the first harvest, and the waste mushroom medium after the third harvest, respectively, showing the highest fraction yield in the waste mushroom medium after the first harvest. was
한편, 폴리페놀, 플라보노이드 성분을 고농도로 함유하고 있는 에틸아세테이트 분획물의 경우, 총 폴리페놀 및 플라보노이드 함량은 살균배지에서 가장 높게 나타났으며, 총당 함량의 경우에도 살균배지의 에틸아세테이트 분획물에서 가장 높게 나타났다. 그러나, 환원당 함량은 1회 및 3회 수확 후 버섯폐배지의 에틸아세테이트 분획물에서 상대적으로 높게 나타났다. 이러한 변화는 살균배지에 접종한 표고버섯이 성장하면서 배지성분인 참나무톱밥, 밀기울의 분해와 이용에 따라 나타난 변화로 이해된다. On the other hand, in the case of the ethyl acetate fraction containing polyphenols and flavonoids at high concentrations, the total polyphenol and flavonoid content was highest in the sterilized medium, and the total sugar content was also the highest in the ethyl acetate fraction of the sterilized medium. . However, the reducing sugar content was relatively high in the ethyl acetate fraction of the mushroom waste medium after the first and third harvests. These changes are understood to be the changes that occurred due to the decomposition and use of oak sawdust and wheat bran, which are the components of the medium, as the shiitake mushrooms inoculated in the sterilization medium grew.
특히, 각각의 분획물의 총 폴리페놀 함량 분석 결과, 살균 배지의 경우 에틸아세테이트 분획물이 가장 높은 185.7mg/g을 나타내었으며, 헥센 분획물 > 부탄올 분획물 > 물 잔류물 순으로 나타났다. 그러나, 1회 수확 후 버섯폐배지의 경우 헥센 분획물이 가장 높은 54.8mg/g을 나타내었으며, 에틸아세테이트 분획물 > 부탄올 분획물 > 물 잔류물 순으로 나타내었으며, 3회 수확 후 버섯폐배지의 경우, 에틸아세테이트 분획물이 가장 높은 95.4mg/g을 나타내었으며, 물 잔류물 > 부탄올 분획물 > 헥센 분획물 순으로 나타났다. 이러한 결과는 살균배지와 수확 후 버섯폐배지 추출물의 성분이 전혀 다르며, 또한 1회 수확 후 버섯폐배지 추출물과 3회 수확 후 버섯폐배지 추출물의 성분이 상이함을 의미하고 있다. In particular, as a result of analyzing the total polyphenol content of each fraction, in the case of the sterilization medium, the ethyl acetate fraction showed the highest value of 185.7 mg/g, followed by the hexene fraction > butanol fraction > water residue. However, in the case of the mushroom waste medium after the first harvest, the hexene fraction showed the highest 54.8mg / g, followed by the ethyl acetate fraction > butanol fraction > water residue, and in the case of the mushroom waste medium after the third harvest, ethyl The acetate fraction showed the highest value of 95.4 mg/g, followed by the water residue > butanol fraction > hexene fraction. These results mean that the components of the sterilization medium and the mushroom waste medium extract after harvest are completely different, and the components of the mushroom waste medium extract after one harvest and the mushroom waste medium extract after three harvests are different.
실시예 6: 표고버섯 재배용 살균 배지, 1회 수확 후 버섯폐배지 및 3회 수확 후 버섯폐배지의 열수 추출물의 순차적 유기용매 분획물의 항균 활성 Example 6: Antibacterial Activity of Sequential Organic Solvent Fractions of Sterilization Medium for Shiitake Mushroom Cultivation, Wasted Mushroom Medium after 1st Harvest and Hot Water Extract of Wasted Mushroom Medium after 3rd Harvest
실시예 5에서 얻어진 다양한 분획물들을 대상으로 항세균 및 항진균 활성을 실시예 4의 방법과 동일하게 평가하였다. 표 7에 나타낸 바와 같이, 표고버섯 재배용 살균배지, 1회 수확 후 버섯폐배지 및 3회 수확 후 버섯폐배지의 열수 추출물의 경우 항균 활성이 나타나지 않았으나, 각각의 추출물의 에틸아세테이트 분획물에서는 매우 강력한 항균 활성이 나타났으며, 특히 1회 수확 후 버섯폐배지 및 3회 수확 후 버섯폐배지의 열수 추출물의 에틸아세테이트 분획물에서는 그람 양성 세균, 그람 음성 세균 및 진균에 대해 광범위한 항균 스펙트럼과 함께 우수한 항세균 및 항진균 활성을 나타내었다. 1회 수확 후 버섯폐배지에서 제조된 에틸아세테이트 분획물의 경우 3회 수확 후 버섯폐배지에서 제조된 에틸아세테이트 분획물보다 전체적으로 항균력이 우수하였으며, 그람 양성세균에 대한 생육억제능이 더욱 강력하였다. The antibacterial and antifungal activities of the various fractions obtained in Example 5 were evaluated in the same manner as in Example 4. As shown in Table 7, antibacterial activity was not shown in the case of the sterilization medium for shiitake mushroom cultivation, the mushroom waste medium after the first harvest, and the hot water extract of the mushroom waste medium after the third harvest, but the ethyl acetate fraction of each extract had very strong antibacterial activity. In particular, the ethyl acetate fraction of the mushroom waste medium after the first harvest and the hot water extract of the mushroom waste medium after the third harvest showed excellent antibacterial and antibacterial activity with a broad antibacterial spectrum against gram-positive bacteria, gram-negative bacteria and fungi. showed antifungal activity. In the case of the ethyl acetate fraction prepared from the mushroom waste medium after the first harvest, the overall antibacterial activity was superior to the ethyl acetate fraction prepared from the mushroom waste medium after the third harvest, and the growth inhibitory activity against Gram-positive bacteria was more powerful.
[표 7] 표고버섯 재배용 살균 배지, 1회 수확 후 버섯폐배지와 3회 수확 후 버섯폐배지의 열수 추출물의 순차적 유기용매 분획물의 항균 활성[Table 7] Sterilization medium for shiitake mushroom cultivation, antibacterial activity of the sequential organic solvent fractions of the waste mushroom medium after the first harvest and the hot water extract of the mushroom waste medium after the third harvest
실시예 7: 1회 수확 후 버섯폐배지와 3회 수확 후 버섯폐배지 열수 추출물 및 이의 활성 분획물의 인간 적혈구 용혈 활성Example 7: Human erythrocyte hemolytic activity of mushroom waste medium after the first harvest and hot-water extract of the mushroom waste medium after the third harvest and active fractions thereof
표고버섯은 오랫동안 식용과 약용으로 사용되어 왔으며, 표고버섯 수확 후 버섯폐배지는 사료로 사용되어 왔다. 본 발명에서는 버섯폐배지 추출물 및 이들의 활성 분획물의 급성 독성을 평가하기 위해 인간 적혈구 용혈 활성 시험을 하였으며, 그 결과는 표 8에 나타내었다. 이때, 용혈 활성은 기존의 보고(정인창, 손호용, 2014년, Korean J. Microbiol. Biotechnol. 42: 285-292)에 준해 평가하였으며, 간단하게는 PBS로 3회 수세한 인간 적혈구 100μl를 96-well microplate에 가하고, 다양한 농도의 시료용액 100μl를 가한 다음 37℃에서 30분간 반응시켰으며, 이후, 반응액을 10분간 원심분리(1,500rpm)하여 상등액 100μl를 새로운 microtiter plate로 옮긴 후 용혈에 따른 헤모글로빈 유출 정도를 414nm에서 측정하였다. 시료의 용매 대조구로는 DMSO(2%)를 사용하였으며, 적혈구 용혈을 위한 실험 대조구로는 Triton X-100(1mg/ml)를 사용하였다. 용혈 활성은 다음의 수식을 이용하여 계산하였다.Shiitake mushrooms have been used for food and medicine for a long time, and mushroom waste medium after harvesting shiitake mushrooms has been used as feed. In the present invention, human erythrocyte hemolytic activity was tested to evaluate the acute toxicity of mushroom lung medium extracts and their active fractions, and the results are shown in Table 8. At this time, the hemolytic activity was evaluated according to the previous report (Jung In-chang, Son Ho-yong, 2014, Korean J. Microbiol. Biotechnol. 42: 285-292), and simply 100 μl of human red blood cells washed three times with PBS was added to a 96-well It was added to a microplate, 100μl of sample solution of various concentrations was added, reacted at 37 ° C for 30 minutes, and then the reaction solution was centrifuged for 10 minutes (1,500 rpm), and 100μl of the supernatant was transferred to a new microtiter plate, and hemoglobin flow due to hemolysis The degree was measured at 414 nm. DMSO (2%) was used as a solvent control for the sample, and Triton X-100 (1 mg/ml) was used as a control for red blood cell hemolysis. Hemolytic activity was calculated using the following formula.
[표 8] 1회 수확 후 버섯폐배지와 3회 수확 후 버섯폐배지 열수 추출물 및 이의 활성 분획물의 인간 적혈구 용혈 활성[Table 8] Human erythrocyte hemolytic activity of the mushroom waste medium after the first harvest and the hot water extract of the mushroom waste medium after the third harvest and active fractions thereof
먼저, 대조구로 사용된 DMSO와 물은 적혈구 용혈 활성이 없었으며, triton X-100은 1mg/ml 농도에서 적혈구를 100% 용혈시킴을 확인하였다. 한편, 1회와 3회 수확 후 버섯폐배지 추출물 및 이의 활성 분획물들은 1mg/ml에서도 용혈 활성은 전혀 나타나지 않았다. 용혈 활성이 있다고 알려진 항암제인 엠포테라신 B는 0.00625mg/ml 농도에서 55.5%, 0.05mg/ml 농도에서 93.7%의 용혈활성을 나타냄을 고려할 때, 표고버섯 수확 후 버섯폐배지 추출물 및 이의 활성 분획물들은 용혈 활성이 없는 것으로 판단된다. First, it was confirmed that DMSO and water used as controls had no erythrocyte hemolytic activity, and triton X-100 100% hemolyzed erythrocytes at a concentration of 1 mg/ml. On the other hand, after the first and third harvests, the mushroom waste medium extract and its active fractions showed no hemolytic activity even at 1 mg/ml. Considering that amphoteracin B, an anticancer drug known to have hemolytic activity, exhibits hemolytic activity of 55.5% at a concentration of 0.00625mg/ml and 93.7% at a concentration of 0.05mg/ml, mushroom waste medium extract and its active fraction after harvesting shiitake mushrooms are considered to have no hemolytic activity.
실시예 8: 1회 수확 후 버섯폐배지와 3회 수확 후 버섯폐배지 열수 추출물 및 이의 활성 분획물의 혈장, 산 및 열 안정성 평가 Example 8: Evaluation of Plasma, Acid and Thermal Stability of Mushroom Waste Medium After 1st Harvest and Hot-Water Extract and Active Fractions of Mushroom Waste Medium After 3rd Harvest
상기 실시예 2 및 실시예 5에서 얻어진 표고버섯 1회 수확 후 버섯폐배지 및 3회 수확 후 버섯폐배지 추출물의 에틸아세테이트 활성 분획물을 대상으로 항균 활성에 대한 열 안정성 및 산 안정성을 확인하였다. 상기 에틸아세테이트 활성 분획물들은 100℃에서 1시간 열 처리, pH 2(0.01M HCl)에서의 1시간 처리시에도 항세균 및 항진균 활성의 감소가 나타나지 않았다. 따라서, 상기의 활성 분획물은 다양한 식품, 화장품, 생활용품 제조 과정 중에도 항세균 및 항진균 활성을 유지할 것으로 예상된다.Thermal stability and acid stability for antibacterial activity were confirmed for the ethyl acetate active fraction of the mushroom waste medium after the first harvest and the mushroom waste medium extract after the third harvest obtained in Examples 2 and 5. The ethyl acetate active fractions showed no decrease in antibacterial and antifungal activity even when heat treated at 100° C. for 1 hour and treated at pH 2 (0.01M HCl) for 1 hour. Therefore, the active fraction is expected to maintain antibacterial and antifungal activity during the manufacturing process of various foods, cosmetics, and household products.
[연구개발과제][R&D task]
[과제고유번호] 2021-0357[Assignment identification number] 2021-0357
[주관부처명] 중소벤처기업부[Name of department in charge] Ministry of SMEs and Startups
[연구관리전문기관] 안동대학교 산학협력단[Research management institution] Andong National University Industry-University Cooperation Foundation
[연구사업명] 경북지역센터 가치만드소(所) 스타기업 육성 R&D 연구[Research project name] R&D research on fostering star companies at the Gyeongbuk Regional Center for Value Creation
[연구과제명] 지역농산물을 이용한 고부가가치 제품 개발[Name of research project] Development of high value-added products using local agricultural products
[기여율] 100%[Contribution rate] 100%
[연구기간] 2021. 09.27.-2021.12.25.[Research period] 2021. 09.27.-2021.12.25.
[주관기관] (재)장애인기업종합지원센터 경북지역센터[Organizer] Corporate Support Center for the Disabled Gyeongbuk Regional Center
[연구수행기관] 안동대학교 식품영양학과[Research Institution] Department of Food and Nutrition, Andong National University
Claims (4)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020210164021A KR20230077110A (en) | 2021-11-25 | 2021-11-25 | Antimicrobial composition comprising the extract of spent mushroom substrate for lentinus edodes |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020210164021A KR20230077110A (en) | 2021-11-25 | 2021-11-25 | Antimicrobial composition comprising the extract of spent mushroom substrate for lentinus edodes |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| KR20230077110A true KR20230077110A (en) | 2023-06-01 |
Family
ID=86770903
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020210164021A Ceased KR20230077110A (en) | 2021-11-25 | 2021-11-25 | Antimicrobial composition comprising the extract of spent mushroom substrate for lentinus edodes |
Country Status (1)
| Country | Link |
|---|---|
| KR (1) | KR20230077110A (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2024258238A1 (en) | 2023-06-15 | 2024-12-19 | 에이비온 주식회사 | Use of claudin-3 as target for diagnosis and treatment of small cell lung cancer |
| KR102754680B1 (en) | 2024-08-30 | 2025-01-21 | 서스테이너블랩 주식회사 | Plastic-free thermoplastic wood-based natural material utilizing agricultural and forestry by-products |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR101611947B1 (en) | 2015-06-11 | 2016-04-12 | 정재경 | Functional food composition for preventing and treating blood vessel disease and producing method thefeof |
| KR101873978B1 (en) | 2016-11-25 | 2018-07-04 | 한경대학교 산학협력단 | COMPOSITION FOR PREVENTING PLANT PATHOGENS COMPRISING EXTRACT FROM SPENT SUBSTRATE OF Lentinula edodes AND METHOD FOR PREPARING THE SAME |
-
2021
- 2021-11-25 KR KR1020210164021A patent/KR20230077110A/en not_active Ceased
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR101611947B1 (en) | 2015-06-11 | 2016-04-12 | 정재경 | Functional food composition for preventing and treating blood vessel disease and producing method thefeof |
| KR101873978B1 (en) | 2016-11-25 | 2018-07-04 | 한경대학교 산학협력단 | COMPOSITION FOR PREVENTING PLANT PATHOGENS COMPRISING EXTRACT FROM SPENT SUBSTRATE OF Lentinula edodes AND METHOD FOR PREPARING THE SAME |
Non-Patent Citations (4)
| Title |
|---|
| Kang DS 외, 2017. Plant Pathology J. 33: 264-275 |
| Zhu H, 2012, Int. J. Biol. Macromol. 50: 840-843 |
| 곽아민 외, 2015. J. Mushrooms, 13: 16-20 |
| 유혜린 외, 2018. 한국버섯학회지 16: 170-174 |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2024258238A1 (en) | 2023-06-15 | 2024-12-19 | 에이비온 주식회사 | Use of claudin-3 as target for diagnosis and treatment of small cell lung cancer |
| KR102754680B1 (en) | 2024-08-30 | 2025-01-21 | 서스테이너블랩 주식회사 | Plastic-free thermoplastic wood-based natural material utilizing agricultural and forestry by-products |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN105087423B (en) | Novel Bei Laisi bacillus CJBV and the bactericidal composition containing it | |
| KR102086166B1 (en) | Antimicrobial composition comprising the extracts of rambutan peel | |
| KR102243591B1 (en) | Antimicrobial composition comprising the extract of rodgersia podophylla | |
| KR20210027722A (en) | Antimicrobial composition comprising the fermentation extract of medicinal or edible natural products | |
| KR20200003993A (en) | Antimicrobial composition comprising the leaf extract of polygonium ticnctorium and use thereof | |
| KR101791549B1 (en) | An antibacterial composition comprising the pachyrhizus erosus root bark extract as an effective component | |
| KR20230077110A (en) | Antimicrobial composition comprising the extract of spent mushroom substrate for lentinus edodes | |
| KR101602552B1 (en) | Antimicrobial composition comprising the extract of cornus officinalis | |
| KR20200058353A (en) | Antimicrobial composition comprising the hexane and butanol fractions of teleogryllus emma extract | |
| KR102534868B1 (en) | Antimicrobial composition comprising the extract from coccon of protaetia brevitarsis | |
| KR102254424B1 (en) | Antimicrobial composition comprising the extract of sageretia thea | |
| KR101976201B1 (en) | Antimicrobial composition comprising the sprout extract of zingiber officinale | |
| KR101911847B1 (en) | Antimicrobial composition comprising organic solvent fractions of arctium lappa root extracts as an effective component | |
| KR102669103B1 (en) | Antimicrobial composition comprising the micro scaled powder of bast fiber of cannabis sativa l. | |
| KR101776511B1 (en) | Antibacterial-listeria composition of culture medium containing chinese juniper berries fermented with lactobacillus plantarum | |
| KR102787850B1 (en) | Antimicrobial composition comprising the extract of songhwa mushroom fruiting body | |
| KR102572181B1 (en) | Antimicrobial composition comprising the extract from wasp | |
| KR102428964B1 (en) | Antimicrobial composition comprising the extracts of hermetia illucens | |
| KR20220053247A (en) | Antimicrobial composition comprising the ethyl acetate fraction of aerial part of glehnia littoralis extract as an effective component | |
| KR102677601B1 (en) | Antimicrobial composition comprising the kenaf seed extract of jang-dae cultivar | |
| KR102287391B1 (en) | Antimicrobial composition comprising the extract of moringa root | |
| KR102254425B1 (en) | Antimicrobial composition comprising the extract of immature fruit extract of magnolia denudata | |
| KR102287389B1 (en) | Antimicrobial composition comprising the extract of flower bud of panax ginseng c. a. meyer | |
| KR102131454B1 (en) | Antimicrobial composition comprising the extract of yunnongchamssal | |
| KR20240021683A (en) | Antimicrobial composition comprising the extracts of pollen of cannabis sativa l. |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0109 | Patent application |
Patent event code: PA01091R01D Comment text: Patent Application Patent event date: 20211125 |
|
| PA0201 | Request for examination | ||
| PG1501 | Laying open of application | ||
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
Comment text: Notification of reason for refusal Patent event date: 20231023 Patent event code: PE09021S01D |
|
| E601 | Decision to refuse application | ||
| PE0601 | Decision on rejection of patent |
Patent event date: 20240501 Comment text: Decision to Refuse Application Patent event code: PE06012S01D Patent event date: 20231023 Comment text: Notification of reason for refusal Patent event code: PE06011S01I |