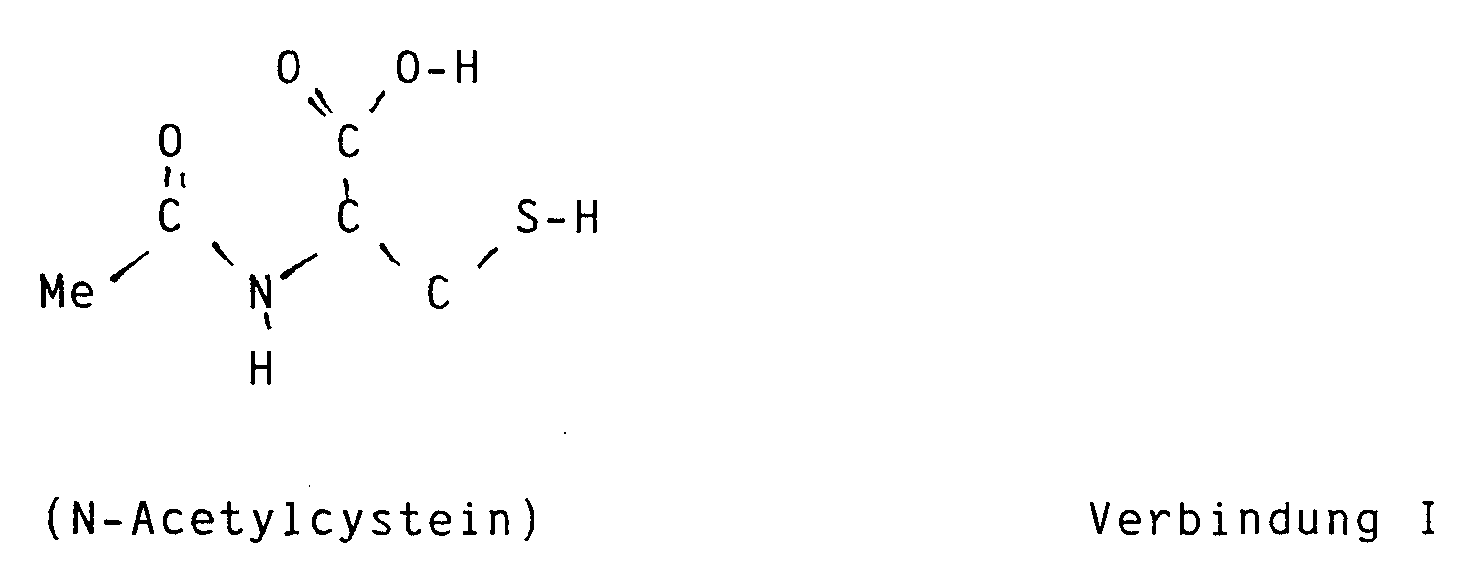WO1994004128A2 - Verwendung von radikalfängern als immunmodulierende agentien in kosmetischen und dermatologischen zubereitungen - Google Patents
Verwendung von radikalfängern als immunmodulierende agentien in kosmetischen und dermatologischen zubereitungen Download PDFInfo
- Publication number
- WO1994004128A2 WO1994004128A2 PCT/DE1993/000773 DE9300773W WO9404128A2 WO 1994004128 A2 WO1994004128 A2 WO 1994004128A2 DE 9300773 W DE9300773 W DE 9300773W WO 9404128 A2 WO9404128 A2 WO 9404128A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- derivatives
- cosmetic
- radical
- thiols
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/49—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds
- A61K8/4973—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds with oxygen as the only hetero atom
- A61K8/498—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds with oxygen as the only hetero atom having 6-membered rings or their condensed derivatives, e.g. coumarin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/13—Amines
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/185—Acids; Anhydrides, halides or salts thereof, e.g. sulfur acids, imidic, hydrazonic or hydroximic acids
- A61K31/19—Carboxylic acids, e.g. valproic acid
- A61K31/195—Carboxylic acids, e.g. valproic acid having an amino group
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/21—Esters, e.g. nitroglycerine, selenocyanates
- A61K31/215—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids
- A61K31/22—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids of acyclic acids, e.g. pravastatin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/335—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin
- A61K31/34—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin having five-membered rings with one oxygen as the only ring hetero atom, e.g. isosorbide
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/335—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin
- A61K31/35—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin having six-membered rings with one oxygen as the only ring hetero atom
- A61K31/352—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin having six-membered rings with one oxygen as the only ring hetero atom condensed with carbocyclic rings, e.g. methantheline
- A61K31/353—3,4-Dihydrobenzopyrans, e.g. chroman, catechin
- A61K31/355—Tocopherols, e.g. vitamin E
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/40—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/41—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with two or more ring hetero atoms, at least one of which being nitrogen, e.g. tetrazole
- A61K31/415—1,2-Diazoles
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/35—Ketones, e.g. benzophenone
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/40—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing nitrogen
- A61K8/44—Aminocarboxylic acids or derivatives thereof, e.g. aminocarboxylic acids containing sulfur; Salts; Esters or N-acylated derivatives thereof
- A61K8/447—Aminocarboxylic acids or derivatives thereof, e.g. aminocarboxylic acids containing sulfur; Salts; Esters or N-acylated derivatives thereof containing sulfur
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/46—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing sulfur
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/67—Vitamins
- A61K8/671—Vitamin A; Derivatives thereof, e.g. ester of vitamin A acid, ester of retinol, retinol, retinal
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/67—Vitamins
- A61K8/676—Ascorbic acid, i.e. vitamin C
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
- A61P37/04—Immunostimulants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q17/00—Barrier preparations; Preparations brought into direct contact with the skin for affording protection against external influences, e.g. sunlight, X-rays or other harmful rays, corrosive materials, bacteria or insect stings
- A61Q17/04—Topical preparations for affording protection against sunlight or other radiation; Topical sun tanning preparations
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2800/00—Properties of cosmetic compositions or active ingredients thereof or formulation aids used therein and process related aspects
- A61K2800/40—Chemical, physico-chemical or functional or structural properties of particular ingredients
- A61K2800/52—Stabilizers
- A61K2800/522—Antioxidants; Radical scavengers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2800/00—Properties of cosmetic compositions or active ingredients thereof or formulation aids used therein and process related aspects
- A61K2800/40—Chemical, physico-chemical or functional or structural properties of particular ingredients
- A61K2800/59—Mixtures
Definitions
- radical scavengers as immunomodulating agents in cosmetic and dermatological preparations
- the present invention relates to active substances and preparations containing such active substances for cosmetic or dermatological treatment and / or prophylaxis of inflammatory, allergic or autoimmune-reactive symptoms, in particular dermatoses.
- the skin especially the epidermis, as a barrier organ of the human organism, is particularly exposed to external influences.
- the skin represents an immunological organ which, as an immunocompetent peripheral compartment, plays its own role in inductive, effective and regulative immune processes of the whole organism.
- UVC range rays with wavelengths less than 290 nm
- UVB range rays in the range between 290 and 320 nm
- the narrower range around 308 nm is given as a maximum of the erythema effectiveness of sunlight.
- UVA range UV range
- UVA radiation also causes damage to the elastic and collagen fibers of the connective tissue, which causes the skin to age prematurely (so-called photoaging), and that it can be seen as the cause of numerous phototoxic and photoallergic reactions.
- the damaging influence of UVB radiation can be intensified by UVA radiation.
- UVB radiation is of particular interest in the development of topical sunscreens, since this is where the spectrum of actions for acute inflammatory processes (sunburn) and chronic damage (photoaging) is located.
- UVB-induced immunosuppression a serious change in the intraepidermal immunological situation, which is referred to as UVB-induced immunosuppression.
- UVB-induced immunosuppression a serious change in the intraepidermal immunological situation.
- Immunosuppression in general is the suppression or weakening of the reactivity of the immune system.
- UVB-induced immunosuppression can be broken down into local and systemic effects. Ultimately, it encompasses a variety of different aspects, all of which involve a reduction in the normal immunological defense mechanisms of the skin. So was the model of UVB-irradiated mice the connection between the increased tumor growth and the immunosuppressive effect of UVB light was related very early on. This UVB-induced immunosuppression is discussed today as a mechanism by means of which highly immunogenic, UVB-induced neoplastic cells escape the immunological defense, and thus their own destruction.
- This cell-surface glycoprotein also called ICAM-1 is one of the most important cellular communication structures through which direct cell-cell contacts between epidermal keratinocytes and leukocytes such as e.g. T lymphocytes and monocytes are regulated.
- UVB-induced immunosuppression therefore affects a broad spectrum of immunological dysfunctions, which result in a reduction in the normal immune response.
- Exposure to ultraviolet rays Agents namely to use the usual sunscreen substances:
- Derivatives of dibenzoylmethane, for example, are predominantly used to protect against UVA rays.
- UVB radiation which are predominantly derivatives of 3-benzylidene camphor, 4-aminobenzoic acid, cinnamic acid, salicylic acid, benzophenone and also 2-phenylbenzimidazole.
- free radical scavengers as agents acting against UV-induced photoxidative phenomena in the skin.
- photochemical reaction products are predominantly free radical compounds, for example hydroxy radicals.
- Undefined radical photo products which are created in the skin itself, can also display uncontrolled subsequent reactions due to their high reactivity.
- singlet oxygen a non-radical excited state of the oxygen molecule can occur with UV radiation, just as short-lived epoxies and many others.
- Singlet oxygen for example, is characterized by increased reactivity compared to the triplet oxygen that is normally present (radical ground state). However, there are also excited, reactive (radical isch) triplet states of the oxygen molecule.
- vitamin E a substance with a known antioxidant effect, in light protection formulations.
- the background was however, always UV protection through light absorption or protection against photooxidative processes.
- the object of the present invention was therefore to provide active substances and preparations containing such active substances, with the aid of which an effective prophylaxis against UVB immunosuppression can be brought about
- damaged immune system can be strengthened again.
- Cosmetic or dermatological preparations for the treatment and / or prophylaxis of immunosuppression induced by UVB radiation characterized by a therapeutically or cosmetically effective content of free radical scavengers and the
- the radical scavengers according to the invention are particularly advantageously selected from the group of thiols and / or thiol derivatives, from the group of tocopherols and their derivatives and from the group consisting of 2,4-0-furfurylidene sorbitol and / or its alkyl ethers.
- Preparations which act as tanning agents and which contain N-acetylcysteine were indeed known from EP-A 219 455.
- this document refers to the fact that N-acetylcysteine promotes pigment formation and is mainly used for this purpose.
- a certain additional antierythematous effect which is claimed at the specified location, makes it suitable for cosmetic preparations.
- this document does not honor the advantageous properties of the present invention, namely the advantages described above.
- EP-A 138 262 describes a combination of panthenol
- Methionine, cysteine, N-acetyl-cysteine, S-acetyl-cysteine, and other ingredients is used to treat eczema and dishydrosis caused by sunlight. This document also does not convey any teaching that goes in the direction of the present
- thiols are understood to mean organic compounds which are distinguished by the group -SH.
- thiol derivatives are organic compounds which are either derivatives while maintaining the group -SH
- the rest X can be selected from the group -O-R ', -NRR'.
- the radicals R, R 'and R "represent independently
- the rest X can be selected from the group -O-R ', -NRR'.
- the radicals R, R 'and R "represent independently
- organic radicals R, R 'and R "independently of one another represent: H, ethyl, acetyl.
- X can be any organic radicals such that the organic radicals R, R 'and R "independently of one another represent: H, ethyl, acetyl.
- salts or acid or base adducts of these particularly preferred thiols or thiol derivatives are also preferred.
- Radical scavengers which are advantageous according to the invention are also to be understood to mean 2,4-0-furfurylidene sorbitol and / or its alkyl ether.
- 2,4-0-furfurylidene sorbitol is synonymous with the names 2,4-0-furfurylidene glucitol, 2,4-0- (2-furanylmethylene) glucitol, 2,4-monofurfuryl idensorbitol. It is characterized by the following structure:
- R 1-4 independently of one another denote H
- R 1-4 are particularly preferably identical and represent H or Methyl groups
- the configuration of the four asymmetric carbon atoms in the part of the molecule derived from sorbitol preferably corresponds to that of the carbon atoms in natural sorbitol, although the other stereoisomers are also fundamentally advantageous.
- the configuration of the asymmetric carbon atom of the furfurylidene group can equally advantageously be R or S.
- a racemate of R and S configuration, based on this carbon atom, is of particular advantage according to the invention.
- radical scavengers according to the invention can advantageously be selected from the group of customary cosmetic and dermatological radical scavengers, in particular from the group consisting of tocopherols and their derivatives, especially ⁇ -tocopherol or ⁇ -tocopheryl esters, in particular ⁇ -tocopheryl acetate, furthermore sesamol, bile acid derivatives such as methyl -, ethyl, propyl, amyl, butyl and lauryl gallate, the Konyferylbenzoat benzoate of benzoin, butylhydroxyanisole, butylhydroxytoluene, citric acid, phosphoric acid, lecithin, trihydroxybutyrophenone, carotenes, vitamin A and its derivatives, in particular retinyl palmitate, ascorbic acid, ascorbyl palmitate, dilauryl thiodipropionate, distearyl , Monoisopropyl citrate, phytic acid and its
- the tocopherols also called vitamin E, are derived from the base body tocol ((2-methyl-2- (4,8,12-trimethyltridecyl) -chroman-6-ol) and are characterized by the following structures:
- K represents either H or an acyl radical
- R, R 'and R "independently of one another denote H or a methyl group, e.g .:
- K H ⁇ -tocopherol
- R "" can represent an alkyl or alkenyl radical having 1 to 21 atoms.
- the tocopherol derivatives preferred according to the invention are the ⁇ -tocopherol and its esters, in particular the ⁇ -tocopheryl acetate. It is known from EP patent application 345 362 to use 2,4-furfurylidene sorbitol in cosmetic and dermatological formulations. In such formulations it primarily serves as an intercepting agent for free radicals, which can be caused, inter alia, by UV radiation.
- the cosmetic and / or dermatological formulations according to the invention can be composed as usual and can be used for prophylaxis and / or for the treatment of the skin in the sense of a dermatological treatment or a prophylaxis and / or treatment in the sense of cosmetics. But they can also be used in make-up products in decorative cosmetics. They preferably contain 0.01% by weight to 10% by weight, but in particular 0.1% by weight to 6% by weight, based on the total weight of one or more radical scavengers.
- radical scavengers it is particularly advantageous to use combinations of several radical scavengers, in particular if at least one of the components is selected from the group of the thiols or thiol derivatives and 2,4-0-furfurylidene sorbitol and its alkyl ethers.
- radical scavengers or preparations according to the invention containing such radical scavengers are preferably used, preferably one or more thiols or thiol derivatives or one or more tocopherols or their derivatives or
- 2,4-0-furfurylidene sorbitol or its alkyl ether is applied to the skin in a sufficient amount in the manner customary for cosmetics or dermatics.
- Cosmetic and dermatological preparations in the form of a sunscreen are particularly preferred. These advantageously additionally contain at least one UVA filter and / or at least one UVB filter and / or at least one inorganic pigment.
- Cosmetic preparations according to the invention for protecting the skin from UV rays can be in various forms, e.g. are usually used for this type of preparation. So you can e.g. a solution, an emulsion of the type water-in-oil (W / O) or of the type oil-in-water (0 / W), or a multiple emulsions, for example of the type water-in-oil-in-water (W / O / W), a gel, a hydrodispersion, a solid stick or an aerosol.
- the cosmetic preparations according to the invention can contain cosmetic auxiliaries, as is customary used in such preparations, for example preservatives, bactericides, perfumes, anti-foaming agents, dyes, pigments which have a coloring effect, thickeners, surface-active substances, emulsifiers, softening substances, moisturizing and / or moisturizing substances, fats, oils, Waxes or other common components of a cosmetic formulation such as alcohols, polyols, polymers, foam stabilizers, electrolytes, organic solvents or silicone derivatives.
- cosmetic auxiliaries for example preservatives, bactericides, perfumes, anti-foaming agents, dyes, pigments which have a coloring effect, thickeners, surface-active substances, emulsifiers, softening substances, moisturizing and / or moisturizing substances, fats, oils, Waxes or other common components of a cosmetic formulation such as alcohols, polyols, polymers, foam stabilizers, electrolytes, organic
- the cosmetic or dermatological preparation is a solution or lotion
- the following can be used as solvents: - water or aqueous solutions; - oils, such as triglycerides of the capric or
- Caprylic acid but preferably castor oil
- Fats, waxes and other natural and synthetic fat bodies preferably esters of fatty acids with alcohols of low C number, e.g. with isopropanol, propylene glycol or glycerin, or esters of
- Alcohols with low C number alkanoic acids or with fatty acids Alcohols, diols or polyols of low C number, and also their ethers, preferably ethanol, isopropanol, propylene glycol, glycerol, ethylene glycol,
- Emulsions according to the invention e.g. in form of a
- Sun protection cream, a sun protection lotion or a sun protection milk are advantageous and contain e.g. the fats, oils, waxes and other fat bodies mentioned, and also water and an emulsifier, as is usually used for such a type of formulation.
- Gels according to the invention usually contain low C number alcohols, e.g. Ethanol, isopropanol, 1,2-propanediol, glycerol and water or an oil mentioned above in the presence of a thickener which is preferably silicon dioxide or an aluminum silicate in the case of oily-alcoholic gels, and is preferably a polyacrylate in the case of aqueous-alcoholic or alcoholic gels.
- low C number alcohols e.g. Ethanol, isopropanol, 1,2-propanediol, glycerol and water or an oil mentioned above in the presence of a thickener which is preferably silicon dioxide or an aluminum silicate in the case of oily-alcoholic gels, and is preferably a polyacrylate in the case of aqueous-alcoholic or alcoholic gels.
- Fixed pins according to the invention contain e.g. natural or synthetic waxes, fatty alcohols or fatty acid esters. Lip care pens are preferred.
- Suitable blowing agents for cosmetic or dermatological preparations according to the invention which can be sprayed from aerosol containers are the customary known volatile, liquefied blowing agents, for example hydrocarbons (propane, butane, isobutane), which can be used alone or in a mixture with one another. Compressed air can also be used advantageously.
- hydrocarbons propane, butane, isobutane
- the preparations according to the invention can preferably also contain substances which absorb UV radiation in the UVB range, the total amount of the filter substances e.g. 0.1% by weight to 30% by weight, preferably 0.5 to 10% by weight, in particular 1 to 6% by weight, based on the total weight of the preparation, in order to provide cosmetic preparations, that protect the skin from the entire range of ultraviolet radiation. They can also serve as sunscreens.
- the UVB filters can be oil-soluble or water-soluble.
- oil-soluble substances e.g. to call:
- UVB filters which can be used in combination with the radical scavengers according to the invention, is of course not intended to be limiting.
- the invention also relates to the combination of one or more radical scavengers according to the invention with one or more UVB filters or cosmetic or dermatological preparations according to the invention which also contain one or more UVB filters. It may also be advantageous to combine one or more radical scavengers according to the invention with UVA filters which have hitherto usually been contained in cosmetic and / or dermatological ones. These substances are preferably derivatives of dibenzoylmethane, in particular 1- (4'-tert-butylphenyl) -3- (4'-methoxyphenyl) propane-1,3-dione and 1-phenyl-3- ( 4'-isopropylphenyl) propane-1,3-dione. These combinations or preparations containing these combinations are also the subject of the invention. The quantities used for the UVB combination can be used.
- radical scavengers according to the invention are combined with UVA and UVB filters.
- Cosmetic preparations containing radical scavengers according to the invention can also contain inorganic pigments, which are usually used in cosmetics to protect the skin from UV rays. These are oxides of titanium, zinc, iron, zirconium, silicon, manganese, aluminum, cerium and mixtures thereof, as well as modifications in which the oxides are the active agents. It is particularly preferred to use pigments based on titanium dioxide.
- UVA filter and / or UVB filter and pigment or preparations which contain this combination are also the subject of the invention.
- the amounts given for the above combinations can be used.
- Cosmetic preparations for protecting the scalp from UV rays according to the invention are, for example, shampooing agents, preparations which are used in the Rinsing of hair before or after shampooing, before or after permanent wave treatment, before or after dyeing or decolorizing the hair can be used to formulate hair drying or inlaying preparations, to dye or decolorize a hairdressing and treatment lotion, a Hair lacquer or permanent waving.
- the cosmetic preparations contain active ingredients and auxiliaries, as are usually used for this type of preparations for hair care and hair treatment.
- Auxiliaries are preservatives, surface-active substances, substances to prevent foaming, emulsifiers, thickeners, fats, oils, waxes, organic solvents, bactericides, perfumes, dyes or pigments, the task of which is to dye the hair or the preparation itself, electrolytes , Preparations against greasy hair.
- Cosmetic preparations which are a shampooing agent preferably contain at least one anionic, nonionic or amphoteric surface-active substance or mixtures thereof, at least one radical scavenger according to the invention in an aqueous medium and auxiliaries as are usually used therefor.
- the surface-active substance can be present in the shampoo in a concentration between 1% by weight and 50% by weight.
- the cosmetic or dermatological preparation is in the form of a lotion which is rinsed out and used, for example, before or after decolorization, before or after shampooing, between two shampooing steps, before or after permanent waving, it is, for example, aqueous or aqueous alcoholic solutions, which may contain surface-active substances, preferably non-ionic or cationic surface surface-active substances, the concentration of which can be between 0.1 and 10% by weight, preferably between 0.2 and 5% by weight.
- aqueous or aqueous alcoholic solutions which may contain surface-active substances, preferably non-ionic or cationic surface surface-active substances, the concentration of which can be between 0.1 and 10% by weight, preferably between 0.2 and 5% by weight.
- Preparation can also be an aerosol with the auxiliaries usually used for this.
- a cosmetic preparation in the form of a lotion that is not rinsed out, in particular a lotion for inlaying the hair, a lotion used for blow-drying the hair, a styling and treatment lotion generally provides an aqueous, alcoholic or aqueous-alcoholic solution represents and contains at least one cationic, anionic, non-ionic or amphoteric polymer or mixtures thereof, and at least one radical scavenger according to the invention.
- the amount of the polymers used is e.g. between 0.1 and 10% by weight, preferably between 0.1 and 3% by weight.
- Active ingredient combinations I - V refer to the total weight of the active ingredient combinations.
- Vitamin E acetate 75.00
- UVB-MLR The UVB mixed lymphocyte reaction method
- the UVB-MLR is a method to analyze effects of test substances on the UVB-induced suppression of a cellular immune response. It is a modification of the MLR, an immunological in-vitro standard procedure, which serves as a measure for the activation and functionalization of the T-lymphocyte system.
- test substances are added to the cell cultures in various concentrations.
- a Phillips TL 20W / 12 lamp was used as the radiation source.
- 7.5 mJ UVB / cm 2 , 15 mJ UVB / cm 2 , and 30 mJ UVBZcm 2 were used as the radiation dose.
- Mononuclear cells of the peripheral blood of two healthy human donors are made using a density gradient
- the individual culture consists of 3.0 * 10 5 mitocytin-treated stimulator cells (donor A) and 2.5 * 10 responder cells (donor B) (incubation at 37 ° C, 7.5% CO 2 , 10% FCS ( Fetal calf serum) in RPMI 1640 medium).
- the stimulator cells are physiologically locked by treatment with mitocytin, so that only they serve as a cellular antigen for the responder cells whose proliferation is due to the incorporation of
- 3H-thymidine is determined.
- the responder cells are no longer recognized as antigen by the stimulator cells.
- H-thymidine is analyzed after separation of all cells, i.e. stimulator and responder cells.
- the amount of 3 H-thymidine incorporated correlates with the ability to respond to the immune system: the less 3 H-thymidine is incorporated, the stronger the UVB immunosuppression.
- the stimulator cells are irradiated with the corresponding UVB dose before they are incubated with the responder cells.
- the corresponding test substance is present in the culture medium during the irradiation.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- General Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Epidemiology (AREA)
- Pharmacology & Pharmacy (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Birds (AREA)
- Immunology (AREA)
- Dermatology (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Organic Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Emergency Medicine (AREA)
- Cosmetics (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
Abstract
Description
Claims
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP6505780A JPH08506085A (ja) | 1992-08-26 | 1993-08-24 | 化粧用及び皮膚用製剤中における免疫調節剤としての遊離基補捉剤の使用 |
| AU49422/93A AU690075B2 (en) | 1992-08-26 | 1993-08-24 | Use of radical catchers as immunomodulating agents in cosmetic and dermatological compositions |
| DE59306051T DE59306051D1 (de) | 1992-08-26 | 1993-08-24 | Verwendung von radikalfängern als immunmodulierende agentien in kosmetischen und dermatologischen zubereitungen |
| EP93918914A EP0656773B1 (de) | 1992-08-26 | 1993-08-24 | Verwendung von radikalfängern als immunmodulierende agentien in kosmetischen und dermatologischen zubereitungen |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DEP4228455.4 | 1992-08-26 | ||
| DE4228455A DE4228455A1 (de) | 1992-08-26 | 1992-08-26 | Kosmetische und dermatologische Lichtschutzformulierungen mit einem Gehalt an Thiolen und/oder Thiolderivaten |
| DE4305788A DE4305788C2 (de) | 1993-02-25 | 1993-02-25 | Kosmetische und dermatologische Formulierungen zum Schutze der Haut gegen Oxidationsprozesse |
| DEP4305788.8 | 1993-02-25 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO1994004128A2 true WO1994004128A2 (de) | 1994-03-03 |
| WO1994004128A3 WO1994004128A3 (de) | 1994-07-07 |
Family
ID=25917922
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/DE1993/000773 Ceased WO1994004128A2 (de) | 1992-08-26 | 1993-08-24 | Verwendung von radikalfängern als immunmodulierende agentien in kosmetischen und dermatologischen zubereitungen |
Country Status (8)
| Country | Link |
|---|---|
| EP (1) | EP0656773B1 (de) |
| JP (1) | JPH08506085A (de) |
| AT (1) | ATE150960T1 (de) |
| AU (1) | AU690075B2 (de) |
| DE (1) | DE59306051D1 (de) |
| DK (1) | DK0656773T3 (de) |
| ES (1) | ES2102670T3 (de) |
| WO (1) | WO1994004128A2 (de) |
Cited By (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2736826A1 (fr) * | 1995-07-20 | 1997-01-24 | Oreal | Composition filtrante a usage cosmetique ou dermatologique |
| FR2736825A1 (fr) * | 1995-07-20 | 1997-01-24 | Oreal | Composition filtrante a usage cosmetique ou dermatologique contenant un chelateur de fer |
| US5645825A (en) * | 1995-06-07 | 1997-07-08 | The Procter & Gamble Company | Depilatory compositions comprising sulfhydryl compounds |
| WO1999029294A1 (en) * | 1997-12-08 | 1999-06-17 | Arizona Board Of Regents | Long-acting, chemical-resistant skin emollients, moisturizers, and strengtheners |
| FR2810548A1 (fr) * | 2000-06-26 | 2001-12-28 | Oreal | Utilisation d'ergothioneine et/ou de ses derives comme agent anti-glycation |
| WO2001072287A3 (de) * | 2000-03-24 | 2002-03-28 | Merck Patent Gmbh | Verwendung von ectoin oder ectoin-derivaten zur prophylaxe und/oder behandlung von uv-induzierter immunsuppression |
| WO2004073700A1 (en) * | 2003-02-19 | 2004-09-02 | Omega Bio-Pharma (I.P.2) Limited | Composition comprising cysteamine for improving immunity of animals |
| US7048910B2 (en) | 2000-09-07 | 2006-05-23 | Merck Patent Gmbh | Use of ectoine or ectoine derivatives for oral care |
| EP1961453A2 (de) | 2004-05-03 | 2008-08-27 | Symrise GmbH & Co. KG | Benzyliden-ß-dicarbonylverbindungen als UV-Absorber |
| EP2108369A1 (de) * | 2008-04-08 | 2009-10-14 | Giuliani S.P.A. | Verbindungen zur Behandlung der durch aggressive Wirkstoffe verursachten Immunsuppression der Haut |
| US7981899B2 (en) | 2002-03-28 | 2011-07-19 | Merck Patent Gmbh | Use of compatible solutes for inhibiting the release of ceramides |
Family Cites Families (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE2331821A1 (de) * | 1973-06-22 | 1975-01-23 | Henkel & Cie Gmbh | Entuendungshemmer fuer kosmetische praeparationen |
| AU509899B2 (en) * | 1976-02-13 | 1980-05-29 | E.R. Squibb & Sons, Inc. | Proline derivatives and related compounds |
| JPS52136925A (en) * | 1976-05-12 | 1977-11-16 | Sansho Seiyaku Kk | Skin cosmetic |
| US4248861A (en) * | 1979-02-21 | 1981-02-03 | Schutt Steven R | Skin treatment methods |
| CA1277663C (en) * | 1985-02-04 | 1990-12-11 | Richard A. Mueller | Heterocyclic amides |
| GB2180153A (en) * | 1985-09-10 | 1987-03-25 | Rory Ltd | Compositions containing acetylcysteine |
| DE3542309A1 (de) * | 1985-11-29 | 1987-06-04 | Cardona Federico Dr | Medizinisches antioxidativum |
| US4847267A (en) * | 1986-03-17 | 1989-07-11 | Charles Of The Ritz Group Ltd. | Skin treatment composition and method |
| FR2608425B1 (fr) * | 1986-12-17 | 1989-04-28 | Rofsa Cerd | Composition cosmetique pour la depigmentation selective partielle de la peau et procede pour sa fabrication |
| IT1205111B (it) * | 1987-05-28 | 1989-03-15 | Fitocosmesi & Farma Lab | Impiego del 2,4-monofurfuriliden-sorbitolo ad uso cosmetico |
| EP0496433B1 (de) * | 1987-10-22 | 1999-03-24 | The Procter & Gamble Company | Chelatbildner enthaltende Lichtschutzmittel |
| US4847072A (en) * | 1987-10-22 | 1989-07-11 | The Procter & Gamble Company | Photoprotection compositions comprising tocopherol sorbate |
| ATE112171T1 (de) * | 1989-03-14 | 1994-10-15 | Univ Texas | Uv-induzierter faktor für immununterdrückung. |
| FR2666226B1 (fr) * | 1990-08-30 | 1994-10-28 | Jean Noel Thorel | Composition protectrice de la peau. |
| DE69225631T2 (de) * | 1991-02-19 | 1998-12-17 | Cci Corp., Gifu | Acylaminosäure-Verbindungen und Verfahren zu ihrer Herstellung |
| CA2122923C (en) * | 1991-11-25 | 1999-01-19 | Roy L. Blank | Compositions for regulating skin wrinkles and/or skin atrophy |
-
1993
- 1993-08-24 ES ES93918914T patent/ES2102670T3/es not_active Expired - Lifetime
- 1993-08-24 DK DK93918914.8T patent/DK0656773T3/da active
- 1993-08-24 WO PCT/DE1993/000773 patent/WO1994004128A2/de not_active Ceased
- 1993-08-24 DE DE59306051T patent/DE59306051D1/de not_active Expired - Lifetime
- 1993-08-24 AU AU49422/93A patent/AU690075B2/en not_active Expired - Fee Related
- 1993-08-24 AT AT93918914T patent/ATE150960T1/de not_active IP Right Cessation
- 1993-08-24 JP JP6505780A patent/JPH08506085A/ja active Pending
- 1993-08-24 EP EP93918914A patent/EP0656773B1/de not_active Expired - Lifetime
Cited By (18)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5645825A (en) * | 1995-06-07 | 1997-07-08 | The Procter & Gamble Company | Depilatory compositions comprising sulfhydryl compounds |
| US5897857A (en) * | 1995-06-07 | 1999-04-27 | The Procter & Gamble Company | Depilatory compositions comprising sulfhydryl compounds |
| FR2736826A1 (fr) * | 1995-07-20 | 1997-01-24 | Oreal | Composition filtrante a usage cosmetique ou dermatologique |
| FR2736825A1 (fr) * | 1995-07-20 | 1997-01-24 | Oreal | Composition filtrante a usage cosmetique ou dermatologique contenant un chelateur de fer |
| AU677556B2 (en) * | 1995-07-20 | 1997-04-24 | L'oreal | Screening composition for cosmetic or dermatological use containing an iron chelator |
| EP0770377A1 (de) * | 1995-07-20 | 1997-05-02 | L'oreal | Filtrierende kosmetische oder dermatologische Zusammensetzung enthaltend einen Eisen-Chelat-Bildner |
| US5776472A (en) * | 1995-07-20 | 1998-07-07 | Societe L'oreal S.A. | UVA photoprotective cosmetic/dermatological compositions comprising iron chelating agents |
| EP0754446A3 (de) * | 1995-07-20 | 2007-04-04 | L'oreal | Verwendung von einem Antioxidant und/oder einem Antiradikal in einer filtrierende kosmetische oder dermatologische Zusammensetzung |
| US6284258B1 (en) | 1997-12-08 | 2001-09-04 | Arizona Board Of Regents | Long-acting, chemical-resistant skin emollients, moisturizers, and strengtheners |
| WO1999029294A1 (en) * | 1997-12-08 | 1999-06-17 | Arizona Board Of Regents | Long-acting, chemical-resistant skin emollients, moisturizers, and strengtheners |
| WO2001072287A3 (de) * | 2000-03-24 | 2002-03-28 | Merck Patent Gmbh | Verwendung von ectoin oder ectoin-derivaten zur prophylaxe und/oder behandlung von uv-induzierter immunsuppression |
| FR2810548A1 (fr) * | 2000-06-26 | 2001-12-28 | Oreal | Utilisation d'ergothioneine et/ou de ses derives comme agent anti-glycation |
| EP1166768A1 (de) * | 2000-06-26 | 2002-01-02 | L'oreal | Verwendung von Ergothionein und seinen Derivaten als Glykierungsinhibitor |
| US7048910B2 (en) | 2000-09-07 | 2006-05-23 | Merck Patent Gmbh | Use of ectoine or ectoine derivatives for oral care |
| US7981899B2 (en) | 2002-03-28 | 2011-07-19 | Merck Patent Gmbh | Use of compatible solutes for inhibiting the release of ceramides |
| WO2004073700A1 (en) * | 2003-02-19 | 2004-09-02 | Omega Bio-Pharma (I.P.2) Limited | Composition comprising cysteamine for improving immunity of animals |
| EP1961453A2 (de) | 2004-05-03 | 2008-08-27 | Symrise GmbH & Co. KG | Benzyliden-ß-dicarbonylverbindungen als UV-Absorber |
| EP2108369A1 (de) * | 2008-04-08 | 2009-10-14 | Giuliani S.P.A. | Verbindungen zur Behandlung der durch aggressive Wirkstoffe verursachten Immunsuppression der Haut |
Also Published As
| Publication number | Publication date |
|---|---|
| ATE150960T1 (de) | 1997-04-15 |
| AU4942293A (en) | 1994-03-15 |
| JPH08506085A (ja) | 1996-07-02 |
| EP0656773A1 (de) | 1995-06-14 |
| AU690075B2 (en) | 1998-04-23 |
| DK0656773T3 (da) | 1997-10-13 |
| WO1994004128A3 (de) | 1994-07-07 |
| EP0656773B1 (de) | 1997-04-02 |
| DE59306051D1 (de) | 1997-05-07 |
| ES2102670T3 (es) | 1997-08-01 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0799023B1 (de) | Verwendung von flavonoiden als immunmodulierende oder immunschützende agenzien in kosmetischen und dermatologischen zubereitungen | |
| EP0633017B1 (de) | Kosmetische und dermatologische Zubereitungen mit einem Gehalt an Delta-Aminolävulinsäure | |
| DE10032964B4 (de) | Verwendung von Kreatin in kosmetischen oder dermatologischen Zubereitungen | |
| EP0656774B1 (de) | Kosmetische und dermatologische lichtschutzformulierungen mit einem gehalt an thiolen und/oder thiolderivaten | |
| EP0686026B1 (de) | Synergistische lichtschutzkombinationen und kosmetische und dermatologische formulierungen, solche kombinationen enthaltend | |
| EP1194115A1 (de) | Topisch anzuwendendes mittel mit schuetzender und regenerativer wirkung die idebenon enthalten | |
| EP0945126B1 (de) | Kosmetische oder dermatologische Zusammensetzung bestehend aus Carnitin und/oder einem Acylcarnitin, und mindestens einem Antioxidans | |
| DE19806947A1 (de) | Kosmetische oder dermatologische Wirkstoffkombinationen aus mindestens einer Substanz gewählt aus der Gruppe, bestehend aus Carnitin und den Acylcarnitinen, und mindestens einem Chinon und oder mindestens einem Hydrochinon sowie Zubereitungen mit einem Gehalt an solchen Wirkstoffkombinationen | |
| WO2002009652A2 (de) | Carnitin enthaltende kosmetische oder dermatologische wirkstoffkombinationen und deren verwendung | |
| EP1594455A1 (de) | Kosmetische oder dermatologische zubereitungen mit einem gehalt an kreatin, kreatinin und/oder seinen derivaten in kombination mit sojabohnenkeimextrakten | |
| DE10136077A1 (de) | Kosmetische oder dermatologische Zubereitungen mit einer Kombination von Kreatinin und/oder Kreatininderivaten mit Kreatin und/oder seinen Derivaten | |
| EP0656773B1 (de) | Verwendung von radikalfängern als immunmodulierende agentien in kosmetischen und dermatologischen zubereitungen | |
| DE10136076A1 (de) | Verwendung von Kreatinin und/oder Kreatininderivaten in kosmetischen oder dermatologischen Zubereitungen | |
| DE19806946A1 (de) | Kosmetische oder dermatologische Wirkstoffkombinationen aus mindestens einer Substanz gewählt aus der Gruppe, bestehend aus Carnitin und den Acylcarnitinen, und mindestens einem Retinoid sowie Zubereitungen mit einem Gehalt an solchen Wirkstoffkombinationen | |
| DE19806889A1 (de) | Verwendung von Acyl-Carnitin | |
| DE10316666B4 (de) | Kosmetische oder dermatologische Zubereitungen mit einer Kombination von Kreatinin mit Kreatin und Coenzym Q10 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A2 Designated state(s): AU JP US |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A2 Designated state(s): AT BE CH DE DK ES FR GB GR IE IT LU MC NL PT SE |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| AK | Designated states |
Kind code of ref document: A3 Designated state(s): AU JP US |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A3 Designated state(s): AT BE CH DE DK ES FR GB GR IE IT LU MC NL PT SE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1993918914 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 1993918914 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref country code: US Ref document number: 1995 387891 Date of ref document: 19950623 Kind code of ref document: A Format of ref document f/p: F |
|
| WWG | Wipo information: grant in national office |
Ref document number: 1993918914 Country of ref document: EP |