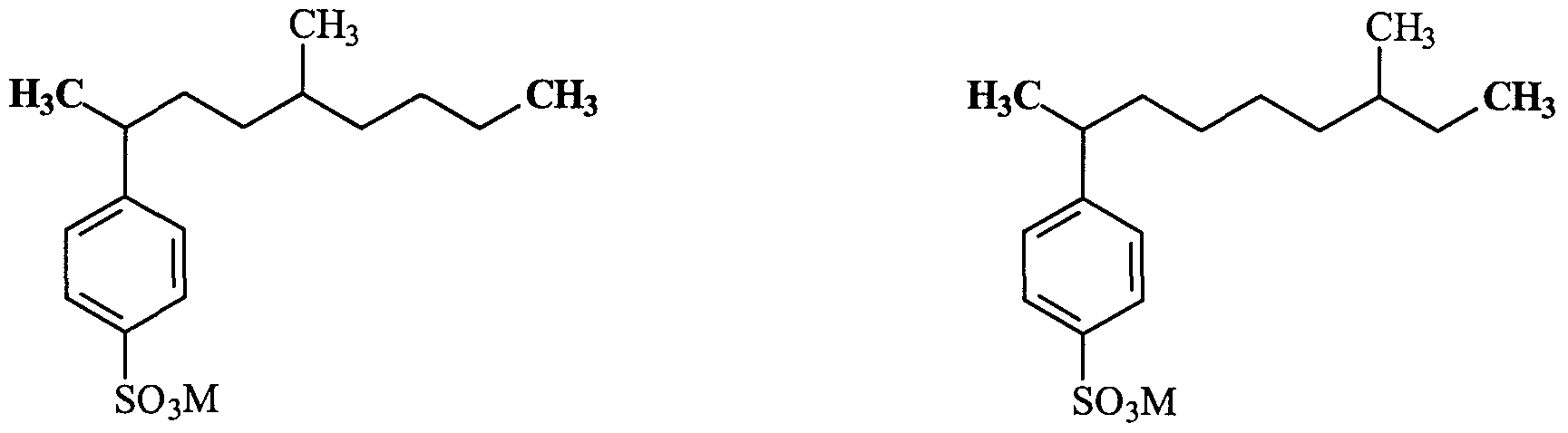WO2000043473A2 - Aqueous heavy duty liquid detergent compositions comprising modified alkylbenzene sulfonates - Google Patents
Aqueous heavy duty liquid detergent compositions comprising modified alkylbenzene sulfonates Download PDFInfo
- Publication number
- WO2000043473A2 WO2000043473A2 PCT/US1999/029717 US9929717W WO0043473A2 WO 2000043473 A2 WO2000043473 A2 WO 2000043473A2 US 9929717 W US9929717 W US 9929717W WO 0043473 A2 WO0043473 A2 WO 0043473A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- mixture
- branched
- alkylbenzene sulfonate
- alkyl
- surfactant
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 CC1***(C*)C1 Chemical compound CC1***(C*)C1 0.000 description 9
Classifications
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D11/00—Special methods for preparing compositions containing mixtures of detergents
- C11D11/04—Special methods for preparing compositions containing mixtures of detergents by chemical means, e.g. by sulfonating in the presence of other compounding ingredients followed by neutralising
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D1/00—Detergent compositions based essentially on surface-active compounds; Use of these compounds as a detergent
- C11D1/38—Cationic compounds
- C11D1/65—Mixtures of anionic with cationic compounds
- C11D1/652—Mixtures of anionic compounds with carboxylic amides or alkylol amides
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D1/00—Detergent compositions based essentially on surface-active compounds; Use of these compounds as a detergent
- C11D1/02—Anionic compounds
- C11D1/12—Sulfonic acids or sulfuric acid esters; Salts thereof
- C11D1/22—Sulfonic acids or sulfuric acid esters; Salts thereof derived from aromatic compounds
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D1/00—Detergent compositions based essentially on surface-active compounds; Use of these compounds as a detergent
- C11D1/38—Cationic compounds
- C11D1/52—Carboxylic amides, alkylolamides or imides or their condensation products with alkylene oxides
- C11D1/525—Carboxylic amides (R1-CO-NR2R3), where R1, R2 or R3 contain two or more hydroxy groups per alkyl group, e.g. R3 being a reducing sugar rest
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D1/00—Detergent compositions based essentially on surface-active compounds; Use of these compounds as a detergent
- C11D1/38—Cationic compounds
- C11D1/52—Carboxylic amides, alkylolamides or imides or their condensation products with alkylene oxides
- C11D1/528—Carboxylic amides (R1-CO-NR2R3), where at least one of the chains R1, R2 or R3 is interrupted by a functional group, e.g. a -NH-, -NR-, -CO-, or -CON- group
Definitions
- This invention relates to heavy duty liquid laundry detergent products which are aqueous in nature and which include particular types of improved alkylbenzene sulfonate surfactant mixtures adapted for use by controlling compositional parameters, especially a
- compositions containing both branched and linear alkylbenzene sulfonate surfactants are complex.
- such compositions can be highly varied, containing one or more different kinds of branching in any of a number of positions on the aliphatic chain.
- a very large number, e.g., hundreds, of distinct chemical species are possible in such mixtures. Accordingly there is an onerous burden of experimentation if it is desired to improve such compositions so that they can clean better in detergent compositions while at the same time remaining biodegradable.
- the formulator's knowledge is key to guiding this effort.
- the present invention provides aqueous heavy-duty liquid detergent compositions comprising modified alkylbenzene sulfonate surfactant mixtures.
- the present invention comprises an aqueous heavy-duty liquid detergent composition.
- aqueous based heavy duty laundry detergent compositions herein preferably contain a surfactant system comprising surfactants selected from nonionic detersive surfactant, anionic detersive surfactant, zwitterionic detersive surfactant, amine oxide detersive surfactant, and mixtures thereof.
- the first embodiment of the present invention comprises an aqueous based heavy duty laundry detergent composition comprising:
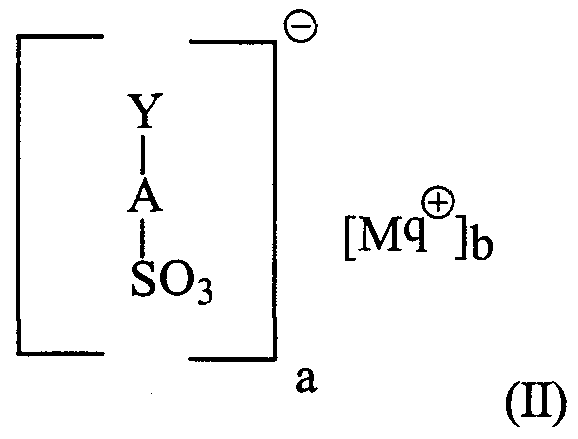
- composition of a modified alkylbenzene sulfonate surfactant mixture comprising:
- L is an acyclic aliphatic moiety consisting of carbon and hydrogen, said L having two methyl termini and said L having no substituents other than A, R 1 and R 2 ; and wherein said mixture of branched alkylbenzene sulfonates contains two or more, preferably at least three, optionally more, of said branched alkylbenzene sulfonates differing in molecular weight of the anion of said formula (I) and wherein said mixture of branched alkylbenzene sulfonates has
- R 1 , L and R 2 a sum of carbon atoms in R 1 , L and R 2 of from 9 to 15, preferably from 10 to 14;
- M is a cation or cation mixture, preferably M is selected from H, Na, K, Ca, Mg and mixtures thereof, more preferably M is selected from H, Na, K and mixtures thereof, more preferably still, M is selected from H, Na, and mixtures thereof, M having a valence q, typically from 1 to 2, preferably 1; a and b are integers selected such that said branched alkylbenzene sulfonates are electroneutral (a is typically from 1 to 2, preferably 1, b is 1); R 1 is d-C alkyl, preferably d-C 2 alkyl, more preferably methyl; R 2 is selected from H and d-C 3 alkyl (preferably H and d-
- Y is an unsubstituted linear aliphatic moiety consisting of carbon and hydrogen having two methyl termini, and wherein said Y has a sum of carbon atoms of from 9 to 15, preferably from 10 to 14, and said Y has an average aliphatic carbon content of from about 10.0 to about 14.0, preferably from about 11.0 to about 13.0, more preferably 11.5 to 12.5 carbon atoms; and wherein said modified alkylbenzene sulfonate surfactant mixture is further characterized by a 2/3-phenyl index of from about 275 to about 10,000, preferably from about 350 to about 1200, more preferably from about 500 to about 700; and also preferably wherein said modified alkylbenzene sulfonate surfactant mixture has a 2-methyl-2 -phenyl index of less than about 0.3, preferably less than about 0.2, more preferably less than about 0J, more preferably still, from
- Such alternate embodiments of the invention nonlimitingly include those termed herein as "medium 2/3-phenyl surfactant system".
- Such surfactant system essentially contain useful amounts of the modified alkylbenzene sulfonate surfactant, along with other known alkylbenzene sulfonates subject to specific provisions of the 2/3-phenyl index of the overall composition.
- Such an aqueous based heavy duty laundry detergent compositions include:
- composition of a medium 2/3-phenyl surfactant system consisting essentially of:
- surfactant system of a first alkylbenzene sulfonate surfactant, wherein said first alkylbenzene sulfonate surfactant is a modified alkylbenzene sulfonate surfactant mixture, said surfactant mixture comprising:
- L is an acyclic aliphatic moiety consisting of carbon and hydrogen, said L having two methyl termini and said L having no substituents other than A, R 1 and R 2 ; and wherein said mixture of branched alkylbenzene sulfonates contains two or more of said branched alkylbenzene sulfonates differing in molecular weight of the anion of said formula (I) and wherein said mixture of branched alkylbenzene sulfonates has
- a, b, M, A and q are as defined hereinbefore and Y is an unsubstituted linear aliphatic moiety consisting of carbon and hydrogen having two methyl termini, and wherein said Y has a sum of carbon atoms of from 9 to 15, preferably from 10 to 14, and said Y has an average aliphatic carbon content of from about 10.0 to about 14.0; and wherein said modified alkylbenzene sulfonate surfactant mixture is further characterized by a 2/3-phenyl index of from about 275 to about 10,000; and
- surfactant system of a second alkylbenzene sulfonate surfactant, wherein said second alkylbenzene sulfonate surfactant is an alkylbenzene sulfonate surfactant mixture other than said modified alkylbenzene sulfonate surfactant mixture (1)
- said second alkylbenzene sulfonate surfactant is a commercial C ⁇ 0 -C ⁇ 4 linear alkylbenzene sulfonate surfactant, e.g., DETAL ® process LAS or HF process LAS though in general any commercial linear (LAS) or branched (ABS, TPBS) type can be used
- said second alkylbenzene sulfonate surfactant has a 2/3-phenyl index of from about 75 to about 160; provided that said medium 2/3-phenyl surfactant system has a 2/3-phenyl index of from about 160 to about 275, (preferably
- the third embodiment of the present invention comprises an aqueous based heavy duty laundry detergent composition comprising:
- the fourth embodiment of the present invention comprises an aqueous based heavy duty laundry detergent composition comprising:
- a modified alkylbenzene sulfonate surfactant mixture consisting essentially of the product of a process comprising the steps, in sequence, of: (I) alkylating benzene with an alkylating mixture; (LI) sulfonating the product of (I); and (LH) neutralizing the product of (LI); wherein said alkylating mixture comprises:
- the fifth embodiment of the present invention comprises an aqueous based heavy duty laundry detergent composition comprising:
- composition of a modified alkylbenzene sulfonate surfactant mixture comprising:
- L is an acyclic aliphatic moiety consisting of carbon and hydrogen, said L having two methyl termini and said L having no
- M is a cation or cation mixture having a valence q; a and b are integers selected such that said branched alkylbenzene sulfonates are electroneutral; R ! is Cj-C 3 alkyl; R 2 is selected from H and C ⁇ -C 3 alkyl; A is a benzene moiety; and
- Y is an unsubstituted linear aliphatic moiety consisting of carbon and hydrogen having two methyl termini, and wherein said Y has a sum of carbon atoms of from 9 to 15, preferably from 10 to 14, and said Y has an average aliphatic carbon content of from about 10.0 to about 14.0; and wherein said modified alkylbenzene sulfonate surfactant mixture is further characterized by a 2/3-phenyl index of from about 275 to about 10,000 and wherein said modified alkylbenzene sulfonate surfactant mixture has a 2-methyl-
- composition from about 0.00001% to about 99.9% of composition of a surfactant selected from the group consisting of anionic surfactants other than those of (i), nonionic surfactants, zwitterionic surfactants, cationic surfactants, amphoteric surfactant and mixtures thereof; wherein said composition is further characterized by a 2/3-phenyl index of from about
- composition comprises any alkylbenzene sulfonate surfactant other than said modified alkylbenzene sulfonate surfactant mixture, said composition is further characterized by an overall 2/3-phenyl index of at least about
- said overall 2/3-phenyl index is determined by measuring 2/3-phenyl index, as defined herein, on a blend of said modified alkylbenzene sulfonate surfactant mixture and said any other alkylbenzene sulfonate to be added to said composition, said blend, for purposes of measurement, being prepared from aliquots of said modified alkylbenzene sulfonate surfactant mixture and said other alkylbenzene sulfonate not yet exposed to any other of the components of said composition; and further provided that when said composition comprises any alkylbenzene sulfonate surfactant other than said modified alkylbenzene sulfonate surfactant mixture, said composition is further characterized by an overall 2-methyl-2 -phenyl index of less than about 0.3, wherein said overall 2-methyl-2-phenyl index is to be determined by measuring 2-methyl-2-phenyl index, as defined herein, on a blend of said modified alkylbenzene s
- the detergent compositions defined herein also comprise from about 1% to 80% by weight of the composition of additional detergent ingredients such as builders, enzymes, colorants, bleaching agents, bleach activators, colored speckles, organic detergent builders, inorganic alkalinity sources and mixtures thereof.
- additional detergent ingredients such as builders, enzymes, colorants, bleaching agents, bleach activators, colored speckles, organic detergent builders, inorganic alkalinity sources and mixtures thereof.
- the aqueous liquid detergent compositions of this invention comprise a modified alkylbenzene sulfonate surfactant mixture.
- the essential and optional components of the modified alkylbenzene sulfonate surfactant mixture and other optional materials of the aqueous liquid detergent compositions herein, as well as composition form, preparation and use, are described in greater detail as follows: (All concentrations and ratios are on a weight basis unless otherwise specified.)
- the invention is not intended to encompass any wholly conventional liquid detergent compositions, such as those based exclusively on linear alkylbenzene sulfonates made by any process, or exclusively on known unacceptably branched alkylbenzene sulfonates such as ABS or
- the detergent compositions of the present invention comprise any alkylbenzene sulfonate surfactant other than said modified alkylbenzene sulfonate surfactant mixture (for example as a result of blending into the detergent composition one or more commercial, especially linear, typically linear C 10 -C 14 , alkylbenzene sulfonate surfactants), said composition is further characterized by an overall 2/3-phenyl index of at least about 200, preferably at least about 250, more preferably at least about 350, more preferably still, at least about 500, wherein said overall 2/3-phenyl index is determined by measuring 2/3-phenyl index, as defined herein, on a blend of said modified alkylbenzene sulfonate surfactant mixture and said any other alkylbenzene sulfonate to be added to said composition, said blend, for pu ⁇ oses of measurement, being prepared from aliquots of said modified alkylbenzene sulfonate surfactant
- the invention encompasses the addition of useful hydrotrope precursors and/or hydrotropes, such as C ⁇ -C 8 alkylbenzenes, more typically toluenes, cumenes, xylenes, naphthalenes, or the sulfonated derivatives of any such materials, minor amounts of any other materials, such as tribranched alkylbenzene sulfonate surfactants, dialkylbenzenes and their derivatives, dialkyl tetralins, wetting agents, processing aids, and the like. It will be understood that, with the exception of hydrotropes, it will not be usual practice in the present invention to include any such materials.
- useful hydrotrope precursors and/or hydrotropes such as C ⁇ -C 8 alkylbenzenes, more typically toluenes, cumenes, xylenes, naphthalenes, or the sulfonated derivatives of any such materials, minor amounts of any other materials, such
- Such a modified alkylbenzene sulfonate surfactant mixture can be made as the product of a process using as catalyst a zeolite selected from mordenite, offretite and H-ZSM-12 in at least partially acidic form, preferably an acidic mordenite (in general certain forms of zeolite beta can be used as an alternative but are not preferred).
- zeolite selected from mordenite, offretite and H-ZSM-12 in at least partially acidic form, preferably an acidic mordenite (in general certain forms of zeolite beta can be used as an alternative but are not preferred).
- said modified alkylbenzene sulfonate surfactant mixture consists essentially of said mixture of (a) and (b), wherein said 2-methyl-2-phenyl index of said modified alkylbenzene sulfonate surfactant mixture is less than about 0J, and said average aliphatic carbon content is from about 11.5 to about 12.5 carbon atoms; said R 1 is methyl; said R is selected from H and methyl provided that in at least about 0.7 mole fraction of said branched alkylbenzene sulfonates R is H; and wherein said sum of carbon atoms in R 1 , L and R 2 is from 10 to 14; and further wherein in said mixture of nonbranched alkylbenzene sulfonates, said Y has a sum of carbon atoms of from 10 to 14 carbon atoms, said average aliphatic carbon content of said nonbranched alkylbenzene sulfonates is from about
- methyl termini and/or “terminal methyl” mean the carbon atoms which are the terminal carbon atoms in alkyl moieties, that is L, and/or Y of formula (I) and formula (H) respectively are always bonded to three hydrogen atoms. That is, they will form a CH 3 - group.
- the structure below shows the two terminal methyl groups in an alkylbenzene sulfonate.
- ABS alkylbenzene
- LAB linear alkylbenzene
- MLAS modified alkylbenzene sulfonate mixtures of the invention.
- the surfactant mixtures herein are preferably substantially free from impurities selected from tribranched impurities, dialkyl tetralin impurities and mixtures thereof.
- substantially free it is meant that the amounts of such impurities are insufficient to contribute positively or negatively to the cleaning effectiveness of the composition.
- Structures (w) and (x) nonlimitingly illustrate less preferred compounds of Formula (I) which can be present, at lower levels than the above-illustrated preferred types of stuctures, in the modified alkylbenzene sulfonate surfactant mixtures of the invention and the resulting detergent compositions.
- Formula (I) that are not preferred but which can be present in the modified alkylbenzene sulfonate surfactant mixtures of the invention and the resulting detergent compositions.
- Structure (bb) is illustrative of a tri-branched structure not within Formula (I), but that can be present as an impurity.
- the modified alkylbenzene sulfonate surfactant mixtures herein are the product of sulfonating a modified alkylbenzene, (other than well known tetrapropylene or AB types) wherein the modified alkylbenzene is produced by alkylating benzene with a branched olefin, other than tetrapropylene, and more particularly the lightly branched types described in more detail hereinafter, over an acidic mordenite-type catalyst or other suitable catalyst as defined elsewhere herein.
- a modified alkylbenzene other than well known tetrapropylene or AB types
- the modified alkylbenzene is produced by alkylating benzene with a branched olefin, other than tetrapropylene, and more particularly the lightly branched types described in more detail hereinafter, over an acidic mordenite-type catalyst or other suitable catalyst as defined elsewhere herein.
- said modified alkylbenzene sulfonate surfactant mixtures herein can also be prepared by blending.
- the invention includes a detergent composition using a modified alkylbenzene sulfonate surfactant mixture according to the first embodiment wherein said modified alkylbenzene sulfonate surfactant mixture is prepared by a process comprising a step selected from: (i) blending a mixture of branched and linear alkylbenzene sulfonate surfactants having a 2/3-phenyl index of 500 to 700 with an alkylbenzene sulfonate surfactant mixture having a 2/3-phenyl index of 75 to 160 and (ii) blending a mixture of branched and linear alkylbenzenes having a 2/3-phenyl index of 500 to 700 with an alkylbenzene mixture having a 2/3-phenyl index of 75 to 160 and sulfonating said blend.
- modified alkylbenzene sulfonate surfactant mixtures herein can be made by the steps of:
- step (I) is a modified alkylbenzene mixture in accordance with the invention.
- product of step (H) is a modified alkylbenzene sulfonic acid mixture in accordance with the invention.
- neutralization step (__) is conducted as generally taught herein, the product of step (HI) is a modified alkylbenzene sulfonate surfactant mixture in accordance with the invention.
- Preferred modified alkylbenzene sulfonate surfactant mixtures herein comprise the product of a process comprising the steps of: (I) alkylating benzene with an alkylating mixture; (II) sulfonating the product of (I); and (optionally but very preferably) (III) neutralizing the product of (H); wherein said alkylating mixture comprises: (a) from about 1% to about 99.9%, by weight of branched C -C 2 o (preferably C -C 15 , more preferably C ⁇ 0 -C 1 ) monoolefins, said branched monoolefins having structures identical with those of the branched monoolefins formed by dehydrogenating branched paraffins of formula R ] LR 2 wherein L is an acyclic aliphatic moiety consisting of carbon and hydrogen and containing two terminal methyls; R 1 is d to C 3 alkyl; and R 2 is selected from H and d to
- the invention encompasses a modified alkylbenzene sulfonate surfactant mixture prepared in accordance with the above- outlined steps wherein said alkylating mixture consists essentially of: (a) from about 0.5%) to about 47.5%, by weight of said branched alkylating agent selected from: (G) C - C 14 internal monoolefins R*LR 2 wherein L is an acyclic olefinic moiety consisting of carbon and hydrogen and containing two terminal methyls; (H) C -C ⁇ 4 alpha monoolefins
- R AR wherein A is an acyclic alpha-olefinic moiety consisting of carbon and hydrogen and containing one terminal methyl and one terminal olefinic methylene; and (J) mixtures thereof; wherein in any of (G)-(H), said R is methyl, and said R is H or methyl provided that in at least about 0.7 mole fraction of the total of said monoolefins, R 2 is H; and (b) from about 0.1% to about 25%, by weight of C 9 -C ⁇ 4 linear aliphatic olefins; and
- step (I) modified alkylbenzene sulfonate surfactant mixtures herein are made by the above-outlined processes wherein in step (I), said alkylation is performed in the presence of an alkylation catalyst, said alkylation catalyst is an intermediate acidity solid porous alkylation catalyst, and step (LI) comprises removal of components other than monoalkylbenzene prior to contacting the product of step (I) with sulfonating agent.
- modified alkylbenzene sulfonate surfactant mixture according to the above-defined processes wherein said alkylation catalyst is other than a member selected from the group consisting of HF, A1C1 3 , sulfuric acid and mixtures thereof.
- the alkylation catalyst is selected from the group consisting of non-fluoridated acidic mordenite-type catalyst, fluoridated acidic mordenite-type catalyst and mixtures thereof. Catalysts are described in more detail hereinafter.
- the processes are tolerant of variation, for example conventional steps can be added before, in parallel with, or after the outlined steps (I), (II) and (EH). This is especially the case for accomodating the use of hydrotropes or their precursors.
- the invention encompasses a modified alkylbenzene sulfonate surfactant mixture according to the above-outlined processes wherein a hydrotrope, hydrotrope precursor, or mixtures thereof is added after step (I); or the hydrotrope, hydrotrope precursor or mixtures thereof is added during or after step (H) and prior to step (HI); or a hydrotrope can be added during or after step (HI).
- sulfonation of the modified alkylbenzenes in the instant process can be accomplished using any of the well-known sulfonation systems, including those described in “Detergent Manufacture Including Zeolite Builders and other New Materials", Ed. Sittig., Noyes Data Co ⁇ ., 1979, as well as in Vol. 56 in “Surfactant Science” series, Marcel Dekker, New York, 1996, including in particular Chapter 2 entitled “Alkylarylsulfonates: History, Manufacture, Analysis and Environmental Properties", pages 39-108 which includes 297 literature references.
- any convenient workup steps may be used in the present process.
- Common practice is to neutralize after sulfonation with any suitable alkali.
- the neutralization step can be conducted using alkali selected from sodium, potassium, ammonium, magnesium and substituted ammonium alkalis and mixtures thereof.
- Potassium can assist solubility
- magnesium can promote soft water performance and substituted ammonium can be helpful for formulating specialty variations of the instant surfactants.
- the invention encompasses any of these derivative forms of the modified alkylbenzenesulfonate surfactants as produced by the present process and their use in consumer product compositions.
- acid form of the present surfactants can be added directly to acidic cleaning products, or can be mixed with cleaning ingredients and then neutralized.
- hydrotropes or hydrotrope precursors useful herein can in general be selected from any suitable hydrotrope or hydrotrope precursor, including lower alkyl (d-C 8 ) aromatics and their sulfonic acids and sulfonate salts, but are more typically based on a sulfonic acid or sodium sulfonate salt of toluene, cumene, xylene, napthalene or mixtures thereof.
- the hydrotrope precursors are selected from any suitable hydrotrope precursor, typically toluene, cumene, xylene, napthalene or mixtures thereof.
- a hydrotrope precursor is a compound that during step (HI), namely the sulfonation step, is converted into a hydrotrope.
- the invention encompasses a modified alkylbenzene sulfonate.
- surfactant mixture wherein in step (I) said alkylation is performed at a temperature of from about 125°C to about 230°C (preferably from about 175°C to about 215°C) and at a pressure of from about 50 psig to about 1000 psig (preferably from about 100 psig to about 250 psig).
- said alkylation is performed at a temperature of from about 175°C to about 215°C, at a pressure of from about 100 psig to about 250 psig.
- step (I) LAB manufacturers will conduct step (I), with detergent manufacturers conducting step (LH).
- step (H) is typically conducted by either, or can even be conducted by third party manufacturers.
- step (I) it is found preferable in step (I) to couple together the use of relatively low temperatures (e.g., 175°C to about 215°C) with reaction times of medium duration (1 hour to about 8 hours) in the above-indicated ranges.
- relatively low temperatures e.g., 175°C to about 215°C
- reaction times of medium duration e.g., 1 hour to about 8 hours
- the alkylation "step" (I) herein can be "staged” so that two or more reactors operating under different conditions in the defined ranges may be useful. By operating a plurality of such reactors, it is possible to allow for material with less preferred 2-methyl-2-phenyl index to be initially formed and, su ⁇ risingly, to convert such material into material with a more preferred 2-methyl-2- phenyl index.
- the invention encompasses a modified alkylbenzene sulfonate surfactant mixture wherein step (H) is performed using a sulfonating agent selected from the group consisting of sulfur trioxide, sulfur trioxide/air mixtures, and sulfuric acid (including oleum).
- a sulfonating agent selected from the group consisting of sulfur trioxide, sulfur trioxide/air mixtures, and sulfuric acid (including oleum).
- Chlorosulfonic acid or other known sulfonating agents while less commercially relevant, are also useful and are included for use in the invention.
- the invention includes a modified alkylbenzene sulfonate surfactant mixture wherein said step (HI) is performed using a basic salt, said basic salt having a cation selected from the group consisting of alkali metal, alkaline earth metal, ammonium, substituted ammonium, and mixtures thereof and an anion selected from hydroxide, oxide, carbonate, silicate, phosphate, and mixtures thereof.
- a basic salt having a cation selected from the group consisting of alkali metal, alkaline earth metal, ammonium, substituted ammonium, and mixtures thereof and an anion selected from hydroxide, oxide, carbonate, silicate, phosphate, and mixtures thereof.
- Preferred basic salt is selected from the group consisting of sodium hydroxide, sodium silicate, potassium hydroxide, potassium silicate, magnesium hydroxide, ammonium hydroxide, and mixtures thereof.
- the present invention uses a particularly defined alkylation catalyst.
- Said alkylation catalyst is an intermediate acidity solid porous alkylation catalyst defined in detail hereinafter.
- Particularly preferred alkylation catalysts comprise at least partially dealuminized acidic fluoridated mordenites, at least partially dealuminized acidic nonfluoridated mordenites, and mixtures thereof.
- alkylation catalysts are unsuitable for making the present modified alkylbenzene mixtures and modified alkylbenzene sulfonate surfactant mixtures.
- Unsuitable alkylation catalysts include any of: sulfuric acid, aluminum chloride, and HF. Also unsuitable are non-acidic calcium mordenite, and many others.
- Other catalysts, such as the DETAL® process catalysts of UOP are also unsuitable, at least in their current commercial executions. Indeed no alkylation catalyst currently used for alkylation in the commercial production of detergent C10-C14 linear alkylbenzene sulfonates for use in laundry products are suitable.
- suitable alkylation catalysts herein are selected from shape-selective moderately acidic alkylation catalysts, preferably zeolitic.
- the zeolite catalyst used for the alkylation step (I) is preferably selected from the group consisting of mordenite, HZSM-12, and offretite, any of these being in at least partially acidic form. Mixtures can be used and the catalysts can be combined with binders etc. as described hereinafter. More preferably, the zeolite is substantially in acid form and is contained in a catalyst pellet comprising a conventional binder and further wherein said catalyst pellet comprises at least about 1 %, more preferably at least 5%, more typically from 50% to about 90%, of said zeolite.
- a suitable alkylation catalyst is typically at least partially crystalline, more preferably substantially crystalline not including binders or other materials used to form catalyst pellets, aggregates or composites. Moreover the catalyst is typically at least partially acidic. Fully exchanged Ca-form mordenite, for example, is unsuitable whereas H-form mordenite is suitable.
- the pores characterizing the zeolites useful in the present alkylation process may be substantially circular, uniform pores of about 6.2 Angstrom, or preferably may be somewhat elliptical, such as in mordenite. It should be understood that, in any case, the zeolites used as catalysts in the alkylation step of the present process have a major pore dimension intermediate between that of the large pore zeolites, such as the X and Y zeolites, and the relatively small pore size zeolites ZSM-5 and ZSM-11, and preferably between about 6 Angstrom and about 7 Angstrom. Indeed ZSM-5 has been tried and found inoperable in the present invention.
- zeolites The pore size dimensions and crystal structures of certain zeolites are specified in ATLAS OF ZEOLITE STRUCTURE TYPES by W. M. Meier and D. H. Olson, published by the Structure Commission of the International Zeolite Association (1978 and more recent editions) and distributed by Polycrystal Book Service, Pittsburgh, Pa.
- the zeolites useful in the alkylation step of the instant process generally have at least 10 percent of the cationic sites thereof occupied by ions other than alkali or alkaline-earth metals.
- Typical but non-limiting replacing ions include ammonium, hydrogen, rare earth, zinc, copper and aluminum. Of this group, particular preference is accorded ammonium, hydrogen, rare earth or combinations thereof.
- the zeolites are converted to the predominantly hydrogen form, generally by replacement of the alkali metal or other ion originally present with hydrogen ion precursors, e.g., ammonium ions, which upon calcination yield the hydrogen form.
- This exchange is conveniently carried out by contact of the zeolite with an ammonium salt solution, e.g., ammonium chloride, utilizing well known ion exchange techniques.
- an ammonium salt solution e.g., ammonium chloride
- the extent of replacement is such as to produce a zeolite material in which at least 50 percent of the cationic sites are occupied by hydrogen ions.
- the zeolites may be subjected to various chemical treatments, including alumina extraction (dealumination) and combination with one or more metal components, particularly the metals of Groups HB, HI, IN, NI, NH and NHL It is also contemplated that the zeolites may, in some instances, desirably be subjected to thermal treatment, including steaming or calcination in air, hydrogen or an inert gas, e.g. nitrogen or helium.
- a suitable modifying treatment entails steaming of the zeolite by contact with an atmosphere containing from about 5 to about 100% steam at a temperature of from about 250°C to 1000°C. Steaming may last for a period of between about 0.25 and about 100 hours and may be conducted at pressures ranging from sub-atmospheric to several hundred atmospheres.
- matrix materials include synthetic or naturally occurring substances as well as inorganic materials such as clay, silica, and/or metal oxides.
- Matrix materials can be in the form of gels including mixtures of silica and metal oxides. The latter may be either naturally occurring or in the form of gels or gelatinous precipitates.
- Naturally occurring clays which can be composited with the zeolite include those of the montmorillonite and kaolin families, which families include the sub-bentonites and the kaolins commonly known as Dixie, McNamee-Georgia and Florida clays or others in which the main mineral constituent is halloysite, kaolinite, dickite, nacrite or anauxite.
- Such clays can be used in the raw state as originally mined or initially subjected to calcination, acid treatment or chemical modification.
- the intermediate pore size zeolites employed herein may be compounded with a porous matrix material, such as alumina, silica-alumina, silica-magnesia, silica-zirconia, silica-thoria, silica-beryllia, and silica- titania, as well as ternary combinations, such as silica-alumina-thoria, silica-alumina- zirconia, silica-alumina-magnesia and silica-magnesia-zirconia.
- the matrix may be in the form of a cogel.
- the relative proportions of finely divided zeolite and inorganic oxide gel matrix may vary widely, with the zeolite content ranging from between about 1 to about 99% by weight and more usually in the range of about 5 to about 80% by weight of the composite.
- a group of zeolites which includes some useful for the alkylation step herein have a silica: alumina ratio of at least 2:1, preferably at least 10:1 more preferably at least 20:1.
- the silica: alumina ratios referred to in this specification are the structural or framework ratios, that is, the ratio for the SiO 4 to the AlO 4 tetrahedra.
- silica: alumina ratios as determined by various physical and chemical methods are acceptable for use herein. It should be understood that such methods may acceptably give some variation. For example, a gross chemical analysis may include aluminum which is present in the form of cations associated with the acidic sites on the zeolite, thereby giving a somewhat low experimentally determined silica: alumina ratio.
- thermogravimetric analysis TGA
- a somewhat low ammonia titration may be obtained if cationic aluminum prevents exchange of the ammonium ions onto the acidic sites.
- the zeolites When the zeolites have been prepared in the presence of organic cations they are typically catalytically inactive, commonly because the intracrystalline free space is occupied by organic cations from the forming solution. They may be activated by heating in an inert atmosphere at 540°C for one hour, for example, followed by base exchange with ammonium salts followed by calcination at 540°C in air. The presence of organic cations in the forming solution may not be absolutely essential to the formation of the zeolite; but it does appear to favor the formation of this special type of zeolite. Some natural zeolites may sometimes be converted to zeolites of the desired type by various activation procedures and other treatments such as base exchange, steaming, alumina extraction and calcination.
- the zeolites preferably have a crystal framework density, in the dry hydrogen form, not substantially below about 1.6 g/cm 3 .
- the dry density for known structures may be calculated from the number of silicon plus aluminum atoms per 1000 cubic Angstroms, as given, e.g., on page 19 of the article on Zeolite Structure by W. M. Meier included in "Proceedings of the Conference on Molecular Sieves, London, April 1967", published by the Society of Chemical Industry, London, 1968. Reference is made to this paper for a discussion of the crystal framework density. A further discussion of crystal framework density, together with values for some typical zeolites, is given in U.S. Pat. No. 4,016,218, to which reference is made.
- the zeolite When synthesized in the alkali metal form, the zeolite is conveniently converted to the hydrogen (acidic) form, generally via intermediate formation of the ammonium form by ammonium ion exchange and calcination of the ammonium form to yield the hydrogen form. It has been found that although the hydrogen form of the zeolite catalyzes the reaction successfully, the zeolite may also be partly in the alkali metal form and/or the form of other metal salts.
- EP 466,558 describes an acidic mordenite type alkylation catalyst also of possible use herein having overall Si/Al atomic ratio of 15-85 (15-60), Na weight content is less than 1000 ppm (preferably less than 250 ppm), and there is a low or zero content of extra-network Al species; the elementary mesh volume as defined in EP 466,558 is below 2,760 nm 3 .
- US 5,057,472 is likewise useful for preparing alkylation catalysts herein and relates to concurrent dealumination and ion-exchange of an acid-stable Na ion-containing zeolite, preferably mordenite, effected by contact of the zeolite with a 0.5-3 (preferably 1- 2.5) M HNO 3 solution containing sufficient NH NO 3 to fully exchange the Na + ions for NH + and H + ions.
- the resulting zeolites can have a Si ⁇ 2:Al 2 O 3 ratio of 15:1 to 26:1, preferably 17:1 to 23:1, and are preferably calcined to at least partially convert the NH 4 + /H + form to the H + form.
- the catalyst can contain a Group VIH metal (and optionally also an inorganic oxide) together with the calcined zeolite of '472.
- alkylation catalyst useful herein is described in US 5,175,135 which is an acid mordenite zeolite having a silica/alumina molar ratio of at least 50:1, a Symmetry Index of at least 1.0 as determined by X-ray diffraction analysis, and a porosity such that the total pore volume is in the range from about 0J8 cc/g to about 0.45 cc/g and the ratio of the combined meso- and macropore- volume to the total pore volume is from about 0.25 to about 0.75.
- Particularly preferred alkylation catalysts herein include the acidic mordenite catalysts Zeocat FM-8/25H available from Zeochem; CBN 90 A available from Zeolyst International, and LZM-8 available from UOP Chemical Catalysts as well as fluoridated versions of the above commercial catalysts.
- Fluoridated mordenites can be prepared by a number of ways. A method of providing a particularly useful fluoridated mordenite is described in US 5,777,187. The invention encompasses preferred embodiments in which the mordenites are fluoridated, but also has other preferred embodiments in which the mordenites are non-fluoridated.
- any alkylation catalyst may be used herein provided that the alkylation catalyst can (a) accommodate branched olefins as described elsewhere herein into the smallest pore diameter of said catalyst and (b) selectively alkylate benzene with said branched olefins and optionally mixtures thereof with nonbranched olefins.
- Acceptable selectivity is in accordance with a 2/3-Phenyl index of about 275 to about 10,000 as defined herein.
- the catalyst selections herein are made in part with the intention of minimizing internal alkylbenzene formation (e.g., 4-phenyl, 5-phenyl ”).
- the formulators contributing to the present invention have unexpectedly discovered that control of internal alkylbenzene sulfonate isomers in the present inventive surfactant mixtures in conjunction with introduction of limited methyl branching is very helpful for improving their performance.
- the present invention connects this discovery to discoveries of the synthesis chemists in the present invention, who have determined how to control internal isomer content while providing limited methyl branching in the modified alkylbenzene sulfonate surfactant mixtures in accordance with the formulators' prescriptions.
- internal isomer content needs to be controlled can vary depending on the consumer product application and on whether outright best performance or a balance of performance and cost is required.
- the amount of internal isomer such as internal alkylbenzene isomer is preferably always kept below 25% by weight, but for best results, from 0 to 10%, preferably less than about 5% by weight.
- "Internal alkylbenzene” isomers as defined herein include alkylbenzenes having phenyl attachment to an aliphatic chain in the 4,5,6 or 7 position.
- the prefered alkylation catalysts are the above-described shape selective zeolitic type catalysts, especially mordenites.
- the first reason is to provide the selectivity of formation of preferred compounds such as branched and nonbranched 2-phenyl and 3- phenylalkylbenzenes. This selectivity is measured by the 2/3-phenyl index.
- the second reason is to control the amount of quaternary alkylbenzenes and thus quaternary alkylbenzenesulfonates.
- the detergent composition which comprises, at least about 0.1%, preferably no more than about 10%, more preferably no more than about 5%, more preferably still, no more than about 1% by weight of composition, of a commercial C ⁇ 0 -C ⁇ 4 linear alkylbenzene sulfonate surfactant;
- the detergent composition which comprises, at least about 0.1 %, preferably no more than about 10%, more preferably no more than about 5%, more preferably still, no more than about 1% by weight of composition, of a commercial highly branched alkylbenzene sulfonate surfactant, (e.g., TPBS or tetrapropylbenzene sulfonate);
- a commercial highly branched alkylbenzene sulfonate surfactant e.g., TPBS or tetrapropylbenzene sulfonate
- the detergent composition which comprises, a nonionic surfactant at a level of from about 0.5% to about 25% by weight of composition, and wherein said nonionic surfactant is a polyalkoxylated alcohol in capped or non-capped form having: - a hydrophobic group selected from linear C 10 -C ⁇ 6 alkyl, mid- chain C ⁇ -C 3 branched C 10 -C 16 alkyl, guerbet branched do-C ⁇ 6 alkyl, and mixtures thereof and - a hydrophihc group selected from 1-15 ethoxylates, 1- 15 propoxylates 1-15 butoxylates and mixtures thereof, in capped or uncapped form, (when uncapped, there is also present a terminal primary -OH moiety and when capped, there is also present a terminal moiety of the form - OR wherein R is a d-C 6 hydrocarbyl moiety, optionally comprising a primary or, preferably when present, a secondary alcohol.);
- the detergent composition which comprises, an alkyl sulfate surfactant at a level of from about 0.5% to about 25% by weight of composition, wherein said alkyl sulfate surfactant has a hydrophobic group selected from linear C 10 - C 18 alkyl, mid-chain C ⁇ -C 3 branched C 10 -C ⁇ 8 alkyl, guerbet branched C ⁇ o-C 18 alkyl, and mixtures thereof and a cation selected from Na, K and mixtures thereof;
- the detergent composition which comprises, an alkyl(polyalkoxy)sulfate surfactant at a level of from about 0.5% to about 25% by weight composition, wherein said alkyl(polyalkoxy)sulfate surfactant has - a hydrophobic group selected from linear do-Ci ⁇ alkyl, mid-chain d-C 3 branched C 10 -C 16 alkyl, guerbet branched C 10 -C 16 alkyl, and mixtures thereof and - a (polyalkoxy)sulfate hydrophihc group selected from 1-15 polyethoxysulfate, 1-15 polypropoxysulfate, 1-15 polybutoxysulfate, 1-15 mixed poly(ethoxy/propoxy/butoxy)sulfates, and mixtures thereof, in capped or uncapped form; and - a cation selected from Na, K and mixtures thereof;
- the present invention includes a detergent composition
- a detergent composition comprising (preferably consisting essentially of): (i) from about 0.01% to about 95%), by weight of composition, (preferably from about 0.5% to about 50%, more preferably from about 1%, preferably at least 2%, more preferably at least 4%, more preferably at least 6%, more preferably still at least 8% to about 35%) of modified alkylbenzene sulfonate surfactant mixture according to the invention; (ii) from about 0.00001% to about 99.9% by weight of composition (preferably from about 5% to about 98%, more preferably from about 50% to about 95%) of a conventional hand dishwasing adjunct; and (iii) from about 0.00001% to about 99.9% by weight of composition (preferably from about 0.1% to about 50%, more preferably from about 0.2% to about 40%, even more preferably form abour 0.5% to about 30%), of a surfactant selected from the group consisting of anionic surfactants other than said modified alkylbenzene s
- the invention includes a 2/3-phenyl surfactant mixture consisting essentially of: from 1% (preferably at least about 5%, more preferably at least about 10 %) to about 60% (in one mode preferably less than about 50%, more preferably less than about 40 %), by weight of surfactant system of a first alkylbenzene sulfonate surfactant, wherein said first alkylbenzene sulfonate surfactant is a modified alkylbenzene sulfonate surfactant mixture according to the first embodiment; and from 40% (in one mode preferably at least about 50%, more preferably at least about 60 %) to about 99% (preferably less than about 95%, more preferably less than about 90%), by weight of surfactant system of a second alkylbenzene sulfonate surfactant, wherein said second alkylbenzene sulfonate surfactant is an alkylbenzene sulfonate surfactant mixture other than said modified alkylbenzene
- Processes for preparing a medium 2/3-phenyl surfactant mixture include those comprising a step selected from: (i) blending said first alkylbenzene sulfonate surfactant and said second alkylbenzene sulfonate surfactant; and (ii) blending the nonsulfonated precursor of said first alkylbenzene sulfonate surfactant and the nonsulfonated precursor of said second alkylbenzene sulfonate surfactant and sulfonating said blend.
- Example 1 Mixture of 4-methyl-4-nonanol, 5-methyl-5-decanol, 6-methyl-6-undecanol and 6-methyl-6-dodecanol (A starting-material for branched olefins) A mixture of 4.65 g of 2-pentanone, 20.7 g of 2-hexanone, 51.0 g of 2-heptanone, 36.7 g of 2-octanone and 72.6 g of diethyl ether is added to an addition funnel.
- the ketone mixture is then added dropwise over a period of 2.25 hours to a nitrogen blanketed stirred three neck 2 L round bottom flask, fitted with a reflux condenser and containing 600 mL of 2.0 M n-pentylmagnesium bromide in diethyl ether and an additional 400 mL of diethyl ether. After the addition is complete the reaction mixture is stirred an additional 2.5 hours at 20°C. The reaction mixture is then added to 1kg of cracked ice with stirring. To this mixture is added 393.3 g of 30% sulphuric acid solution. The aqueous acid layer is drained and the remaining ether layer is washed twice with 750 mL of water. The ether layer is then evaporated under vacuum to yield 176J g of a mixture of 4-methyl-4-nonanol, 5-methyl-5-decanol, 6-methyl-6-undecanol and 6-methyl-6- dodecanol.
- Example 2a The olefin mixture of Example 2a is combined with 36g of a shape selective zeolite catalyst (acidic mordenite catalyst ZeocatTM FM-8/25H) and reacted according to example 2a with the following changes.
- the reaction temperature is raised to 190-200°C for a period of about 1-2 hours to randomize the specific branch positions in the olefin mixture.
- the substantially mono methyl branched olefin mixture with randomized branching remaining in the flask along with the substantially mono methyl branched olefin mixture with randomized branching collected in the dean stark trap are recombined and filtered to remove catalyst.
- the solid filter cake is washed twice with 100 mL portions of hexane.
- the hexane filtrate is evaporated under vacuum and the resulting product is combined with the first filtrate to give 147.5 g of a substantially mono methyl branched olefin mixture with randomized branching.
- a modified alkylbenzene mixture in accordance with the invention 147 g of the substantially mono methyl branched olefin mixture of example 2 and 36 g of a shape selective zeolite catalyst (acidic mordenite catalyst ZeocatTM FM-8/25H) are added to a 2 gallon stainless steel, stirred autoclave. Residual olefin and catalyst in the container are washed into the autoclave with 300 mL of n-hexane and the autoclave is sealed. From outside the autoclave cell, 2000 g of benzene (contained in a isolated vessel and added by way of an isolated pumping system inside the isolated autoclave cell) is added to the autoclave.
- a shape selective zeolite catalyst acidic mordenite catalyst ZeocatTM FM-8/25H
- the autoclave is purged twice with 250 psig N2, and then charged to 60 psig N2.
- the mixture is stirred and heated to about 200°C for about 4-5 hours.
- the autoclave is cooled to about 20°C overnight.
- the valve is opened leading from the autoclave to the benzene condenser and collection tank.
- the autoclave is heated to about 120°C with continuous collection of benzene. No more benzene is collected by the time the reactor reaches 120°C.
- the reactor is then cooled to 40°C and 750 g of n- hexane is pumped into the autoclave with mixing.
- the autoclave is then drained to remove the reaction mixture.
- the reaction mixture is filtered to remove catalyst and the n-hexane is removed under vacuum.
- the product is distilled under vacuum (1-5 mm of Hg).
- the substantially mono methyl branched alkylbenzene mixture with a 2/3 -Phenyl index of about 550 and a 2-methyl-2-phenyl index of about 0.02 is collected from 76°C - 130°C (167 g).
- EXAMPLE 4 Substantially Mono Methyl Branched Alkylbenzenesulfonic Acid Mixture with a 2/3-Phenyl Lndex of about 550 and a 2-Methyl-2-Phenyl Index of about 0.02 (A modified alkylbenzene sulfonic acid mixture in accordance with the invention)
- the product of example 3 is sulfonated with a molar equivalent of chlorosulfonic acid using methylene chloride as solvent.
- EXAMPLE 5 Substantially Mono Methyl Branched Alkylbenzene sulfonate, Sodium Salt Mixture with a 2/3-Phenyl index of about 550 (A modified alkylbenzene sulfonate surfactant mixture in accordance with the invention)
- the product of example 4 is neutralized with a molar equivalent of sodium methoxide in methanol and the methanol is evaporated to give 225 g of a substantially mono methyl branched alkylbenzene sulfonate, sodium salt mixture with a 2/3-Phenyl index of about 550 and a 2-methyl-2 -phenyl index of about 0.02.
- Neodene A mixture of 15J g of Neodene (R)10, 136.6 g of Neodene(R)1112, 89.5 g of Neodene(R)12 and 109J g of 1- tridecene is added to a 2 gallon stainless steel, stirred autoclave along with 70 g of a shape selective catalyst (acidic mordenite catalyst ZeocatTM FM-8/25H).
- Neodene is a trade name for olefins from Shell Chemical Company. Residual olefin and catalyst in the container are washed into the autoclave with 200 mL of n-hexane and the autoclave is sealed.
- benzene (contained in a isolated vessel and added by way of an isolated pumping system inside the isolated autoclave cell) is added to the autoclave.
- the autoclave is purged twice with 250 psig N2, and then charged to 60 psig N2- The mixture is stirred and heated to about 200-205°C for about 4- 5 hours then cooled to 70-80°C.
- the valve is opened leading from the autoclave to the benzene condenser and collection tank.
- the autoclave is heated to about 120°C with continuous collection of benzene in collection tank. No more benzene is collected by the time the reactor reaches 120°C.
- the reactor is then cooled to 40°C and 1 kg of n-hexane is pumped into the autoclave with mixing.
- the autoclave is then drained to remove the reaction mixture.
- the reaction mixture is filtered to remove catalyst and the n-hexane is evaporated under low vacuum.
- the product is then distilled under high vacuum (1-5 mm of Hg).
- the substantially linear alkylbenzene mixture with a 2/3-Phenyl index of about 550 and a 2-methyl-2-phenyl index of about 0.02 is collected from 85°C - 150°C (426.2 g)-
- the substantially linear alkylbenzenesulfonic acid mixture of example 7 is neutralized with a molar equivalent of sodium methoxide in methanol and the methanol is evaporated to give 613 g of the substantially linear alkylbenzene sulfonate, sodium salt mixture with a 2/3-Phenyl index of about 550 and a 2-methyl-2-phenyl index of about
- EXAMPLE 9 10-Dimethyl-2-undecanol (A starting-material for branched olefins)
- a glass autoclave liner is added 299 g of geranylacetone, 3.8 g of 5% rutheniumon carbon and 150 ml of methanol.
- the glass liner is sealed inside a 3 L, stainless steel, rocking autoclave and the autoclave purged once with 250 psig N 2 , once with 250 psig H 2 and then charged with 1000 psig H 2 .
- the reaction mixture is heated. At about 75°C, the reaction initiates and begins consuming H 2 and exotherms to 170-180°C.
- the temperature has dropped to 100-110°C and the pressure dropped to 500 psig.
- the autoclave is boosted to 1000 psig with H2 and mixed at 100-110°C for an additional 1 hour and 40 minutes with the reaction consuming an additional 160 psig H 2 but at which time no more H 2 consumption is observed.
- the reaction mixture removed, filtered to remove catalyst and concentrated by evaporation of methanol under vacuum to yield 297.75 g of 6J0-dimethyl-2-undecanol.
- a starting-material for branched olefins A mixture of 671.2 g of citral and 185.6 g of diethyl ether is added to an addition funnel. The citral mixture is then added dropwise over a five hour period to a nitrogen blanketed, stirred, 5 L, 3-neck, round bottom flask equipped with a reflux condenser containing 1.6 L of 3.0 M methylmagnesium bromide solution and an additional 740 ml of diethyl ether. The reaction flask is situated in an ice water bath to control exotherm and subsequent ether reflux.
- the dimethyl branched olefin mixture remaining in the flask along with the dimethyl branched olefin mixture that distilled over are recombined and filtered to remove the catalyst.
- the catalyst filter cake is slurried with 500 ml of hexane and vacuum filtered.
- the catalyst filter cake is washed twice with 100 ml of hexane and the filtrate concentrated by evaporation of the hexane under vacuum.
- the resulting product is combined with the first filtrate to give 820 g of dimethyl branched olefin mixture with randomized branching.
- a modified alkylbenzene mixture in accordance with the invention 820 g of the dimethyl branched olefin mixture of example 12 and 160 g of a shape selective zeolite catalyst (acidic mordenite catalyst ZeocatTM FM-8/25H) is added to a 2 gallon stainless steel, stirred autoclave and the autoclave is sealed.
- the autoclave is purged twice with 80 psig N2 and then charged to 60 psig N2-
- 3000 g of benzene (contained in a isolated vessel and added by way of an isolated pumping system inside the isolated autoclave cell) is added to the autoclave.
- the mixture is stirred and heated to 205°C to about 210°C.
- the reaction is continued for about 10 minutes at which time the product mixture is sampled.
- the 10 minute sample is filtered to remove catalyst and vacuum pulled on the mixture to remove any residual traces of benzene.
- the sample is distilled under vacuum (1-5 mm of Hg).
- the dimethyl branched alkylbenzene mixture with randomized branching and 2/3-Phenyl index of about 600 and a 2-methyl-2-phenyl index of about 0.26 is collected from 90°C - 140°C.
- the reaction is continued at 205°C to about 210°C for about 8 hours.
- the autoclave is cooled to about 30°C overnight.
- the valve is opened leading from the autoclave to the benzene condenser and collection tank.
- the autoclave is heated to about 120°C with continuous collection of benzene. No more benzene is collected by the time the reactor reaches 120°C and the reactor is then cooled to 40°C. The autoclave is then drained to remove the reaction mixture. The reaction mixture is filtered to remove catalyst and vacuum pulled on the mixture to remove any residual traces of benzene. The product is distilled under vacuum (1-5 mm of Hg). The dimethyl branched alkylbenzene mixture with randomized branching and 2/3-Phenyl index of about 600 and a 2-methyl-2-phenyl index of about 0.04 is collected from 90°C - 140°C.
- the dimethyl branched alkylbenzene product of example 13 is sulfonated with a molar equivalent of chlorosulfonic acid using methylene chloride as solvent with HC1 evolved as a side product.
- the resulting sulfonic acid product is concentrated by evaporation of methylene chloride under vacuum.
- the substantially dimethyl branched alkylbenzenesulfonic acid mixture has a 2/3 Phenyl Index of about 2/3-Phenyl index of about 600 and a 2-methyl-2-phenyl index of about 0.04.
- Each of the above blends has a 2/3-phenyl index in the range from about 160 to about 275.
- Each of the above blends has a 2/3-phenyl index in the range from about 160 to about 275.
- Each of the above blends has a 2/3-phenyl index in the range from about 160 to about 275.
- Each of the above blends has a 2/3-phenyl index in the range from about 160 to about 275.
- Modified alkylbenzene mixtures according to the invention (Medium 2/3-phenyl type) Blends are prepared of:
- Each of the above blends has a 2/3-phenyl index in the range from about 160 to about 275.
- Modified alkylbenzene mixtures according to the invention (Medium 2/3-phenyl type) Blends are prepared of:
- Each of the above blends has a 2/3-phenyl index in the range from about 160 to about 275.
- EXAMPLE 22 Modified Alkylbenzene Mixture according to the invention With a 2/3-Phenyl Index of about 550 and a 2-Methyl-2-Phenyl Index of about 0.02 110.25 g of the substantially mono methyl branched olefin mixture of example 2, 36.75 g a nonbranched olefin mixture (decene : undecene : dodecene : tridecene ratio of 2 : 9 : 20 : 18) and 36 g of a shape selective zeolite catalyst (acidic mordenite catalyst ZeocatTM FM-8/25H) are added to a 2 gallon stainless steel, stirred autoclave.
- a shape selective zeolite catalyst acidic mordenite catalyst ZeocatTM FM-8/25H
- Residual olefin and catalyst in the container are washed into the autoclave with 300 mL of n-hexane and the autoclave is sealed.
- 2000 g of benzene (contained in a isolated vessel and added by way of an isolated pumping system inside the isolated autoclave cell) is added to the autoclave.
- the autoclave is purged twice with 250 psig N2, and then charged to 60 psig N2- The mixture is stirred and heated to about 200°C for about 4-5 hours.
- the autoclave is cooled to about 20°C overnight.
- the valve is opened leading from the autoclave to the benzene condenser and collection tank.
- the autoclave is heated to about 120°C with continuous collection of benzene. No more benzene is collected by the time the reactor reaches 120°C.
- the reactor is then cooled to 40°C and 750 g of n-hexane is pumped into the autoclave with mixing.
- the autoclave is then drained to remove the reaction mixture.
- the reaction mixture is filtered to remove catalyst and the n-hexane is removed under vacuum.
- the product is distilled under vacuum (1-5 mm of Hg).
- a modified alkylbenzene mixture with a 2/3-Phenyl index of about 550 and a 2-methyl-2-phenyl index of about 0.02 is collected from 76°C - 130°C (167 g).
- EXAMPLE 23 Modified Alkylbenzenesulfonic Acid Mixture according to the invention (Branched and Nonbranched Alkylbenzenesulfonic Acid Mixture) with a 2/3-Phenyl Index of about 550 and a 2-Methyl-2-Phenyl Index of about 0.02
- the modified alkylbenzene mixture of example 22 is sulfonated with a molar equivalent of chlorosulfonic acid using methylene chloride as solvent. The methylene chloride is removed to give 210 g of a modified alkylbenzenesulfonic acid mixture with a 2/3- Phenyl index of about 550 and a 2-methyl-2-phenyl index of about 0.02.
- EXAMPLE 24 Modified Alkylbenzenesulfonate, Sodium Salt Mixture According to the invention (Branched and Nonbranched Alkylbenzenesulfonate, Sodium Salt Mixture) with a 2/3-Phenyl Index of about 550 and a 2-Methyl-2-Phenyl Index of about 0.02
- the modified alkylbenzenesulfonic acid of example 23 is neutralized with a molar equivalent of sodium methoxide in methanol and the methanol is evaporated to give 225 g of a modified alkylbenzenesulfonate, sodium salt mixture with a 2/3-Phenyl index of about 550 and a 2-methyl-2-phenyl index of about 0.02.
- compositional parameters of conventional linear alkylbenzenes and/or highly branched alkylbenzenesulfonates See, for example Surfactant Science Series, Nolume 40, Chapter 7 and Surfactant Science Series, Nolume 73, Chapter 7.
- this is done by GC and/or GC-mass spectroscopy for the alkylbenzenes and HPLC for the alkylbenzenesulfonates or sulfonic acids; 13 C nmr is also commonly used.
- Another common practice is desulfonation. This permits GC and/or GC-mass spectroscopy to be used, since desulfonation converts the sulfonates or sulfonic acids to the alkylbenzenes which are tractable by such methods.
- the present invention provides unique and relatively complex mixtures of alkylbenzenes, and similarly complex surfactant mixtures of alkylbenzenesulfonates and/or alkylbenzenesulfonic acids.
- Compositional parameters of such compositions can be determined using variations and combinations of the art-known methods.
- the material contains more than about 10% impurities such as dialkylbenzenes, olefins, paraffins, hydrotropes, dialkylbenzenesulfonates, etc.
- Carrier Gas Hydrogen Column Head Pressure: 9 psi Flows: Column Flow @ 1 ml/min. Split Vent @ ⁇ 3ml/min. Septum Purge @ 1 ml/min.
- Injection HP 7673 Autosampler, 10 ul syringe, lul injection Injector Temperature: 350 °C Detector Temperature: 400 °C Oven Temperature Program: initial 70 °C hold 1 min. rate 1 °C/min. final 180 °C hold 10 min.
- Standards required for this method are 2-phenyloctane and 2-phenylpentadecane, each freshly distilled to a purity of greater than 98%. Run both standards using the conditions specified above to define the retention time for each standard.
- a rentention time range which is the retention time range to be used for characterizing any alkylbenzenes or alkylbenzene mixtures in the context of this invention (e.g., test samples).
- Test samples pass the GC test provided that greater than 90% of the total GC area percent is within the retention time range defined by the two standards.
- Test samples that pass the GC test can be used directly in the NMRl and NMR2 test methods.
- Test samples that do not pass the GC test must be further purified by distillation until the test sample passes the GC test.
- the desulfonation method is a standard method described in "The Analysis of Detergents and Detergent Products" by G. F. Longman on pages 197-199. Two other useful descriptions of this standard method are given on page 230-231 of volume 40 of the Surfactant Sience Series edited by T. M. Schmitt: "Analysis of Surfactants” and on page 272 of volume 73 of the Surfactant Science Series: “Anionic Surfactants” edited by John Cross.
- This is an alternative method to the HPLC method, described herein, for evaluation of the branched and nonbranched alkylbenzenesulfonic acid and/or salt mixtures (Modified Alkylbenzensulfonic acid and or salt Mixtures).
- the method provides a means of converting the sulfonic acid and/or salt mixture into branched and nonbranched alkylbenzene mixtures which can then be analyzed by means of the GC and NMR methods NMRl and NMR2 described herein. HPLC
- Mobile phase B Prepare 2000 mL of 60% acetonitrile in HPLC grade water. Filter through an LC eluent membrane filter and degas prior to use.
- Wash Solutions Transfer 250 ⁇ L of the standard solution to a 1 mL autosampler vial and add 750 ⁇ L of the wash solution. Cap and place in the autosampler tray.
- Mobile phase B 40% H 2 O/60% ACN time 0 min. 100% Mobile phase A 0% Mobile Phase B time 75 min. 5% Mobile phase A 95% Mobile Phase B time 98 min. 5% Mobile phase A 95% Mobile Phase B time 110 min. 100% Mobile phase A 0% Mobile Phase B time 120 min. 100% Mobile phase A 0% Mobile Phase B Note: A gradient delay time of 5-10 minutes may be needed depending on dead volume of HPLC system.
- the column should be washed with 100% water followed by 100% acetonitrile and stored in 80/20 ACN/water.
- the HPLC elution time of the 2-phenyloctylbenzenesulfonate defines the lower limit and the elution time of the 2-phenylpentadecanesulfonate standard defines the upper limit of the HPLC analysis relating to the alkylbenzenesulfonic acid/salt mixture of the invention. If 90% of the alkylbenzenesulfonic acid/salt mixture components have retention times within the range of the above standards then the sample can be further defined by methods NMR 3 and NMR 4.
- the alkylbenzenesulfonic acid/salt mixture contains 10% or more of components outside the retention limits defined by the standards then the mixture should be further purified by method HPLC-P or by DE, DIS methods.
- HPLC-P HPLC Preparative
- Alkylbenzenesulfonic acids and/or the salts which contain substantial impurities (10% or greater) are purified by preparative HPLC. See L.R. Snyder and JJ. Kirkland, "Introduction to Modern Liquid Chromatography", 2nd. Ed., Wiley, NY, 1979. This is routine to one skilled in the art. A sufficient quantity should be purified to meet the requirements of the NMR 3 and NMR 4.
- Alkylbenzenesulfonic acids and/or the salts which contain substantial impurities (10% or greater) can also be purified by an LC method (also defined herein as HPLC-P).
- the purified MLAS salt can be isolated and ready for freeze drying within 2 hours.
- a 100 mg sample of Modified alkylbenzenesulfonate salt can be loaded onto a 5g(20ml) Bond Elut Sep Pak and ready within the same amount of time.
- HPLC Waters Model 600E gradient pump, Model 717 Autosampler, Water's
- HPLC Autosampler Vials 4mL glass vials with Teflon caps and glass low volume inserts and pipette capable of accurately delivering 1, 2, and 5mL volumes
- DI-H2O Distilled, deionized water from a Millipore, Milli-Q system or equivalent
- Reservoir A 60/40, H 2 O/C AN with salt and Reservoir B: 40/60,
- Adjustments in organic modifier concentration may be necessary for optimum separation and isolation.
- a 5 liter, 3 -necked round bottom flask with 24/40 joints is equipped with a magnetic stir bar.
- a few boiling chips (Hengar Granules, catalog #136-C) are added to the flask.
- a 91/2 inch long vigreux condenser with a 24/40 joint is placed in the center neck of the flask.
- a water cooled condenser is attached to the top of the vigreux condenser which is fitted with a calibrated thermometer.
- a vacuum receiving flask is attached to the end of the condenser.
- a glass stopper is placed in one side arm of the 5 liter flask and a calibrated thermometer in the other. The flask and the vigreux condenser are wrapped with aluminum foil.
- Fraction B (1881 g) contains the alkylbenzene mixture of interest.
- the method can be scaled according to the practitioner's needs provided that sufficient quantity of the alkylbenzene mixture remains after distillation for evaluation by NMR methods NMRl and NMR2.
- Salts of alkylbenzenesulfonic acids are acidified by common means such as reaction in a solvent with HC1 or sulfuric acid or by use of an acidic resin such as Amberlyst 15.
- the 2/3-Phenyl index of an alkylbenzene mixture is defined by the following equation:
- a 400 mg sample of an anhydrous alkylbenzene mixture is dissolved in 1 ml of anhydrous deuterated chloroform containing 1% v/v TMS as reference and placed in a standard NMR tube.
- the 13 C NMR is run on the sample on a 300 MHz NMR spectrometer using a 20 second recycle time, a 40° 13 C pulse width and gated heteronuclear decoupling. At least 2000 scans are recorded.
- the 13 C NMR spectrum region between about 145.00 ppm to about 150.00 ppm is integrated.
- the 2-methyl-2- phenyl index of an alkylbenzene mixture is defined by the following equation:
- 2-methyl-2-phenyl index (Integral from about 149.35 ppm to about 149.80 ppm)/(Integral from about 145.00 ppm to about 150.00 ppm).
- a 400 mg sample of an anhydrous alkylbenzenesulfonic acid mixture is dissolved in 1 ml of anhydrous deuterated chloroform containing 1% v/v TMS as reference and placed in a standard NMR tube.
- the 13 C NMR is run on the sample on a 300 MHz NMR spectrometer using a 20 second recycle time, a 40° 13 C pulse width and gated heteronuclear decoupling. At least 2000 scans are recorded.
- the 13 C NMR spectrum region between about 152.50 ppm to about 156.90 ppm is integrated.
- a 400 mg sample of an anhydrous alkylbenzenesulfonic acid mixture is dissolved in 1 ml of anhydrous deuterated chloroform containing 1% v/v TMS as reference and placed in a standard NMR tube.
- the 13 C NMR is run on the sample on a 300 MHz NMR spectrometer using a 20 second recycle time, a 40° 13 C pulse width and gated heteronuclear decoupling. At least 2000 scans are recorded.
- the 13 C NMR spectrum region between about 152.50 ppm to about 156.90 ppm is integrated.
- the 2-methyl-2- phenyl Index for an alkylbenzenesulfonic acid mixture is defined by the following equation:
- 2-methyl-2-phenyl index (Integral from about 156.40 ppm to about 156.65 ppm)/(Integral from about 152.50 ppm to about 156.90 ppm).
- the present invention also comprises aqueous based liquid detergent compositions.
- the aqueous liquid detergent compositions preferably comprise from about 10% to about 98%, preferably from about 30% to about 95%), by weight of an aqueous liquid carrier which is preferably water.
- the aqueous liquid detergent compositions of the present invention comprise a surfactant system which preferably contains one or more detersive co-surfactants in addition to the branched surfactants disclosed above.
- the additional co-surfactants can be selected from nonionic detersive surfactant, anionic detersive surfactant, zwitterionic detersive surfactant, amine oxide detersive surfactant, and mixtures thereof.
- the surfactant system typically comprises from about 5% to about 70%, preferably from about 15% to about 30%, by weight of the detergent composition.
- Anionic surfactants include Cn-Ci 8 alkyl benzene sulfonates (LAS) and primary, branched-chain and random C10-C20 alkyl sulfates (AS), the C ⁇ Q-CIS secondary (2,3) alkyl sulfates of the formula CH3(CH 2 ) x (CHOSO3 " M + ) CH 3 and CH 3 (CH2)y(CHOSO3 " M + ) CH2CH3 where x and (y + 1) are integers of at least about 7, preferably at least about 9, and M is a water-solubilizing cation, especially sodium, unsaturated sulfates such as oleyl sulfate, the C ⁇ Q-CI S alkyl alkoxy sulfates ("AE X S"; especially EO 1-7 ethoxy sulfates), CJO-CIS alkyl alkoxy carboxylates (especially the EO 1-5 ethoxycarboxylates), the CJO-18 gly
- anionic surfactants useful herein are disclosed in U.S. Patent No. 4,285,841, Barrat et al, issued August 25, 1981, and in U.S. Patent No. 3,919,678, Laughlin et al, issued December 30, 1975.
- Useful anionic surfactants include the water-soluble salts, particularly the alkali metal, ammonium and alkylolammonium (e.g., monoethanolammonium or triethanolammonium) salts, of organic sulfuric reaction products having in their molecular structure an alkyl group containing from about 10 to about 20 carbon atoms and a sulfonic acid or sulfuric acid ester group.
- water-soluble salts particularly the alkali metal, ammonium and alkylolammonium (e.g., monoethanolammonium or triethanolammonium) salts, of organic sulfuric reaction products having in their molecular structure an alkyl group containing from about 10 to about 20 carbon atoms and a sulfonic acid or sulfuric acid ester group.
- alkyl is the alkyl portion of aryl groups.
- alkyl sulfates especially those obtained by sulfating the higher alcohols (Cg-C ⁇ g carbon atoms) such as those produced by reducing the glycerides of tallow or coconut oil.
- anionic surfactants herein are the water-soluble salts of alkyl phenol ethylene oxide ether sulfates containing from about 1 to about 4 units of ethylene oxide per molecule and from about 8 to about 12 carbon atoms in the alkyl group.
- Other useful anionic surfactants herein include the water-soluble salts of esters of a- sulfonated fatty acids containing from about 6 to 20 carbon atoms in the fatty acid group and from about 1 to 10 carbon atoms in the ester group; water-soluble salts of 2-acyloxy- alkane-1 -sulfonic acids containing from about 2 to 9 carbon atoms in the acyl group and from about 9 to about 23 carbon atoms in the alkane moiety; water-soluble salts of olefin sulfonates containing from about 12 to 24 carbon atoms; and b-alkyloxy alkane sulfonates containing from about 1 to 3 carbon atoms in the alkyl group and from about 8 to 20 carbon atoms in the alkane moiety.
- Particularly preferred anionic surfactants herein are the alkyl polyethoxylate sulfates of the formula RO(C 2 H 4 O) x SO3-M + wherein R is an alkyl chain having from about 10 to about 22 carbon atoms, saturated or unsaturated, M is a cation which makes the compound water-soluble, especially an alkali metal, ammonium or substituted arnmonium cation, and x averages from about 1 to about 15.
- Preferred alkyl sulfate surfactants are the non-ethoxylated C12J5 primary and secondary alkyl sulfates. Under cold water washing conditions, i.e., less than abut 65°F (18.3°C), it is preferred that there be a mixture of such ethoxylated and non-ethoxylated alkyl sulfates.
- fatty acids include capric, lauric, myristic, palmitic, stearic, arachidic, and behenic acid. Other fatty acids include palmitoleic, oleic, linoleic, linolenic, and ricinoleic acid.
- Nonionic and amphoteric surfactants include C12-C18 alkyl ethoxylates (AE) including the so-called narrow peaked alkyl ethoxylates and Cg-C ⁇ alkyl phenol alkoxylates (especially ethoxylates and mixed ethoxy/propoxy).
- AE alkyl ethoxylates
- Cg-C ⁇ alkyl phenol alkoxylates especially ethoxylates and mixed ethoxy/propoxy
- the CI Q- C j g N-alkyl polyhydroxy fatty acid amides can also be used. Typical examples include the Ci2"Ci N-methylglucamides. See WO 9,206,154.
- Other sugar-derived surfactants include the N-alkoxy polyhydroxy fatty acid amides, such as Cjo-Cig N-(3- methoxypropyl) glucamide.
- N-propyl through N-hexyl Ci2-C ⁇ g glucamides can be used for low sudsing.
- C10-C20 conventional soaps may also be used.
- the branched-chain CjQ-Cig soaps may be used. Examples of nonionic surfactants are described in U.S. Patent No. 4,285,841, Barrat et al, issued August 25, 1981.
- these surfactants include ethoxylated alcohols and ethoxylated alkyl phenols of the formula R(OC2H4) n OH, wherein R is selected from the group consisting of aliphatic hydrocarbon radicals containing from about 8 to about 15 carbon atoms and alkyl phenyl radicals in which the alkyl groups contain from about 8 to about 12 carbon atoms, and the average value of n is from about 5 to about 15.
- R is selected from the group consisting of aliphatic hydrocarbon radicals containing from about 8 to about 15 carbon atoms and alkyl phenyl radicals in which the alkyl groups contain from about 8 to about 12 carbon atoms, and the average value of n is from about 5 to about 15.
- ethoxylated alcohols having an average of from about 10 to abut 15 carbon atoms in the alcohol and an average degree of ethoxylation of from about 6 to about 12 moles of ethylene oxide per mole of alcohol. Mixtures of anionic and nonionic surfactants are especially useful.
- compositions herein also contain amine oxide surfactants of the formula: Rl (EO) x (PO) y (BO) z N(O)(CH2R , )2 qH2 ⁇ (I)
- the structure (I) provides one long-chain moiety Rl(EO) x (PO)y(BO) z and two short chain moieties, C ⁇ R'.
- R' is preferably selected from hydrogen, methyl and -CH2OH.
- R* is a primary or branched hydrocarbyl moiety which can be saturated or unsaturated, preferably, R* is a primary alkyl moiety.
- R* is a hydrocarbyl moiety having chainlength of from about 8 to about 18.
- Rl may be somewhat longer, having a chainlength in the range C12-C24.
- amine oxides are illustrated by Cj2-14 alkyldimethyl amine oxide, hexadecyl dimethylamine oxide, octadecylamine oxide and their hydrates, especially the dihydrates as disclosed in U.S. Patents 5,075,501 and 5,071,594, inco ⁇ orated herein by reference.
- the invention also encompasses amine oxides wherein x+y+z is different from zero, specifically x+y+z is from about 1 to about 10, Rl is a primary alkyl group containing 8 to about 24 carbons, preferably from about 12 to about 16 carbon atoms; in these embodiments y + z is preferably 0 and x is preferably from about 1 to about 6, more preferably from about 2 to about 4; EO represents ethyleneoxy; PO represents propyleneoxy; and BO represents butyleneoxy.
- amine oxides can be prepared by conventional synthetic methods, e.g., by the reaction of alkylethoxysulfates with dimethylamine followed by oxidation of the ethoxylated amine with hydrogen peroxide.
- Highly preferred amine oxides herein are solids at ambient temperature, more preferably they have melting-points in the range 30°C to 90°C.
- Amine oxides suitable for use herein are made commercially by a number of suppliers, including Akzo Chemie, Ethyl Co ⁇ ., and Procter & Gamble. See McCutcheon's compilation and Kirk-Othmer review article for alternate amine oxide manufacturers.
- Preferred commercially available amine oxides are the solid, dihydrate ADMOX 16 and ADMOX 18, ADMOX 12 and especially ADMOX 14 from Ethyl Co ⁇ .
- Preferred embodiments include dodecyldimethylamine oxide dihydrate, hexadecyldimethylamine oxide dihydrate, octadecyldimethylamine oxide dihydrate, hexadecyltris(ethyleneoxy)dimethyl-amine oxide, tetradecyldimethylamine oxide dihydrate, and mixtures thereof.
- R' is H
- R' is CH2OH, such as hexadecylbis(2- hydroxyethyl)amine oxide, tallowbis(2-hydroxyethyl)amine oxide, stearylbis(2- hydroxyethyl)amine oxide and oleylbis(2- hydroxyethyl)amine oxide.
- compositions herein also optionally, but preferably, contain up to about 50%, more preferably from about 1% to about 40%, even more preferably from about 5% to about 30%, by weight of a detergent builder material. Lower or higher levels of builder, however, are not meant to be excluded.
- Detergent builders can optionally be included in the compositions herein to assist in controlling mineral hardness. Inorganic as well as organic builders can be used. Builders are typically used in fabric laundering compositions to assist in the removal of particulate soils. Detergent builders are described in U.S. Patent No. 4,321,165, Smith et al, issued March 23, 1982. Preferred builders for use in liquid detergents herein are described in U.S. Patent No. 4,284,532, Leikhim et al, issued August 18, 1981.
- silicate builders are the alkali metal silicates, particularly those having a Si ⁇ 2:Na2 ⁇ ratio in the range 1.6:1 to 3.2.J and layered silicates, such as the layered sodium silicates described in U.S. Patent 4,664,839, issued May 12, 1987 to H. P. Rieck.
- NaSKS-6 is the trademark for a crystalline layered silicate marketed by Hoechst (commonly abbreviated herein as "SKS-6").
- Hoechst commonly abbreviated herein as "SKS-6”
- the Na SKS-6 silicate builder does not contain aluminum.
- NaSKS-6 has the delta-Na2Si ⁇ 5 mo ⁇ hology form of layered silicate.
- SKS-6 is a highly preferred layered silicate for use herein, but other such layered silicates, such as those having the general formula NaMSi x O2 x +ryH2O wherein M is sodium or hydrogen, x is a number from 1.9 to 4, preferably 2, and y is a number from 0 to 20, preferably 0 can be used herein.
- Various other layered silicates from Hoechst include NaSKS-5, NaSKS-7 and NaSKS-11, as the alpha, beta and gamma forms.
- delta-Na2Si ⁇ 5 (NaSKS-6 form) is most preferred for use herein.
- Other silicates may also be useful such as for example magnesium silicate, which can serve as a stabilizing agent for oxygen bleaches and as a component of suds control systems.
- carbonate builders are the alkaline earth and alkali metal carbonates as disclosed in German Patent Application No. 2,321,001 published on November 15, 1973.
- Aluminosilicate builders are useful in the present invention.
- Aluminosilicate builders can be a significant builder ingredient in liquid detergent formulations.
- Aluminosilicate builders include those having the empirical formula: M z (zAlO 2 ) y ]-xH 2 O wherein z and y are integers of at least 6, the molar ratio of z to y is in the range from 1.0 to about 0.5, and x is an integer from about 15 to about 264.
- aluminosilicate ion exchange materials are commercially available. These aluminosilicates can be crystalline or amo ⁇ hous in structure and can be naturally- occurring aluminosilicates or synthetically derived. A method for producing aluminosilicate ion exchange materials is disclosed in U.S. Patent 3,985,669, Krummel, et al, issued October 12, 1976. Preferred synthetic crystalline aluminosilicate ion exchange materials useful herein are available under the designations Zeolite A, Zeolite P (B), Zeolite MAP and Zeolite X.
- the crystalline aluminosilicate ion exchange material has the formula: Na 12 [(Al ⁇ 2)l2(Si ⁇ 2)l2]-xH 2 O wherein x is from about 20 to about 30, especially about 27.
- This material is known as Zeolite A.
- the aluminosilicate has a particle size of about OJ-10 microns in diameter.
- Organic detergent builders suitable for the memeposes of the present invention include, but are not restricted to, a wide variety of polycarboxylate compounds.
- polycarboxylate refers to compounds having a plurality of carboxylate groups, preferably at least 3 carboxylates.
- Polycarboxylate builder can generally be added to the composition in acid form, but can also be added in the form of a neutralized salt. When utilized in salt form, alkali metals, such as sodium, potassium, and lithium, or alkanolammomum salts are preferred.
- polycarboxylate builders include a variety of categories of useful materials.
- One important category of polycarboxylate builders encompasses the ether polycarboxylates, including oxydisuccinate, as disclosed in Berg, U.S. Patent 3,128,287, issued April 7, 1964, and Lamberti et al, U.S. Patent 3,635,830, issued January 18, 1972. See also "TMS/TDS" builders of U.S. Patent 4,663,071, issued to Bush et al, on May 5, 1987.
- Suitable ether polycarboxylates also include cyclic compounds, particularly alicyclic compounds, such as those described in U.S. Patents 3,923,679; 3,835,163; 4,158,635; 4,120,874 and 4,102,903.
- ether hydroxypolycarboxylates copolymers of maleic anhydride with ethylene or vinyl methyl ether, 1, 3, 5-trihydroxy benzene-2, 4, 6-trisulphonic acid, and carboxymethyloxysuccinic acid
- various alkali metal, ammonium and substituted ammonium salts of polyacetic acids such as ethylenediamine tetraacetic acid and nitrilotriacetic acid
- polycarboxylates such as mellitic acid, succinic acid, oxydisuccinic acid, polymaleic acid, benzene 1,3,5- tricarboxylic acid, carboxymethyloxysuccinic acid, and soluble salts thereof.
- Citrate builders e.g., citric acid and soluble salts thereof (particularly sodium salt), are polycarboxylate builders of particular importance for heavy duty liquid detergent formulations due to their availability from renewable resources and their biodegradability. Oxydisuccinates are also especially useful in such compositions and combinations.
- succinic acid builders include the C5- C20 alkyl and alkenyl succinic acids and salts thereof.
- a particularly preferred compound of this type is dodecenylsuccinic acid.
- succinate builders include: laurylsuccinate, myristylsuccinate, palmitylsuccinate, 2-dodecenylsuccinate (preferred), 2-pentadecenylsuccinate, and the like. Laurylsuccinates are the preferred builders of this group, and are described in European Patent Application 86200690.5/0,200,263, published November 5, 1986.
- Fatty acids e.g., C12-C18 monocarboxylic acids
- the aforesaid builders especially citrate and/or the succinate builders, to provide additional builder activity.
- Such use of fatty acids will generally result in a diminution of sudsing, which should be taken into account by the formulator.
- phosphorus-based builders can be used the various alkali metal phosphates such as the well-known sodium tripolyphosphates, sodium pyrophosphate and sodium orthophosphate can be used.
- Phosphonate builders such as ethane-1-hydroxy-lJ- diphosphonate and other known phosphonates (see, for example, U.S. Patents 3,159,581; 3,213,030; 3,422,021; 3,400,148 and 3,422,137) can also be used.
- the aqueous based detergent compositions can, and preferably will, contain various other optional components.
- Such optional components may be in either liquid or solid form.
- the optional components may either dissolve in the liquid phase or may be dispersed within the liquid phase in the form of fine particles or droplets.
- the detergent compositions herein may also optionally contain one or more types of inorganic detergent builders beyond those listed hereinbefore that also function as alkalinity sources.
- optional inorganic builders can include, for example, aluminosilicates such as zeolites. Aluminosilicate zeolites, and their use as detergent builders are more fully discussed in Corkill et al., U.S. Patent No. 4,605,509; Issued August 12, 1986, the disclosure of which is inco ⁇ orated herein by reference.
- crystalline layered silicates such as those discussed in this '509 U.S. patent, are also suitable for use in the detergent compositions herein.
- optional inorganic detergent builders can comprise from about 2% to 15% by weight of the compositions herein.
- the detergent compositions herein may also optionally contain one or more types of detergent enzymes.
- Such enzymes can include proteases, amylases, cellulases and Upases.
- Such materials are known in the art and are commercially available. They may be inco ⁇ orated into the aqueous liquid detergent compositions herein in the form of suspensions, "marumes" or "prills".
- Another suitable type of enzyme comprises those in the form of slurries of enzymes in nonionic surfactants, e.g., the enzymes marketed by Novo Nordisk under the tradename "SL” or the microencapsulated enzymes marketed by Novo Nordisk under the tradename "LDP.”
- Enzymes added to the compositions herein in the form of conventional enzyme prills are especially preferred for use herein. Such prills will generally range in size from about 100 to 1,000 microns, more preferably from about 200 to 800 microns and will be suspended throughout the non-aqueous liquid phase of the composition. Prills in the compositions of the present invention have been found, in comparison with other enzyme forms, to exhibit especially desirable enzyme stability in terms of retention of enzymatic activity over time. Thus, compositions which utilize enzyme prills need not contain conventional enzyme stabilizing such as must frequently be used when enzymes are inco ⁇ orated into aqueous liquid detergents.
- non-aqueous liquid detergent compositions herein will typically comprise from about 0.001% to 5%, preferably from about 0.01% to 1% by weight, of a commercial enzyme preparation.
- Protease enzymes for example, are usually present in such commercial preparations at levels sufficient to provide from 0.005 to 0J Anson units (AU) of activity per gram of composition.
- AU Anson units
- the detergent compositions herein may also optionally contain a chelating agent which serves to chelate metal ions, e.g., iron and/or manganese, within the non-aqueous detergent compositions herein.
- a chelating agent which serves to chelate metal ions, e.g., iron and/or manganese, within the non-aqueous detergent compositions herein.
- Such chelating agents thus serve to form complexes with metal impurities in the composition which would otherwise tend to deactivate composition components such as the peroxygen bleaching agent.
- Useful chelating agents can include amino carboxylates, phosphonates, amino phosphonates, polyfunctionally- substituted aromatic chelating agents and mixtures thereof.
- Amino carboxylates useful as optional chelating agents include ethylenediaminetetraacetates, N-hydroxyethyl-ethylenediaminetriacetates, nitrilotriacetates, ethylene-diamine tetrapropionates, triethylenetetraaminehexacetates, diethylenetriaminepentaacetates, ethylenediaminedisuccinates and ethanol diglycines.
- the alkali metal salts of these materials are preferred.
- Amino phosphonates are also suitable for use as chelating agents in the compositions of this invention when at least low levels of total phosphorus are permitted in detergent compositions, and include ethylenediaminetetrakis (methylene- phosphonates) as DEQUEST. Preferably, these amino phosphonates do not contain alkyl or alkenyl groups with more than about 6 carbon atoms.
- Preferred chelating agents include hydroxy-ethyldiphosphonic acid (HEDP), diethylene triamine penta acetic acid (DTP A), ethylenediamine disuccinic acid (EDDS) and dipicolinic acid (DP A) and salts thereof.
- the chelating agent may, of course, also act as a detergent builder during use of the compositions herein for fabric laundering/bleaching.
- the chelating agent if employed, can comprise from about 0.1% to 4% by weight of the compositions herein. More preferably, the chelating agent will comprise from about 0.2% to 2% by weight of the detergent compositions herein.
- the detergent compositions herein may also optionally contain a polymeric material which serves to enhance the ability of the composition to maintain its solid particulate components in suspension.
- a polymeric material which serves to enhance the ability of the composition to maintain its solid particulate components in suspension.
- Such materials may thus act as thickeners, viscosity control agents and/or dispersing agents.
- Such materials are frequently polymeric polycarboxylates but can include other polymeric materials such as polyvinylpyrrolidone (PVP) or polyamide resins.
- Polymeric polycarboxylate materials can be prepared by polymerizing or copolymerizing suitable unsaturated monomers, preferably in their acid form.
- Unsaturated monomeric acids that can be polymerized to form suitable polymeric polycarboxylates include acrylic acid, maleic acid (or maleic anhydride), fumaric acid, itaconic acid, aconitic acid, mesaconic acid, citraconic acid and methylenemalonic acid.
- the presence in the polymeric polycarboxylates herein of monomeric segments, containing no carboxylate radicals such as vinylmethyl ether, styrene, ethylene, etc. is suitable provided that such segments do not constitute more than about 40% by weight of the polymer.
- Particularly suitable polymeric polycarboxylates can be derived from acrylic acid.
- acrylic acid-based polymers which are useful herein are the water-soluble salts of polymerized acrylic acid.
- the average molecular weight of such polymers in the acid form preferably ranges from about 2,000 to 100,000, more preferably from about 2,000 to 10,000, even more preferably from about 4,000 to 7,000, and most preferably from about 4,000 to 5,000.
- Water-soluble salts of such acrylic acid polymers can include, for example, the alkali metal, salts.
- Soluble polymers of this type are known materials. Use of polyacrylates of this type in detergent compositions has been disclosed, for example, Diehl, U.S. Patent 3,308,067, issued March 7, 1967. Such materials may also perform a builder function.
- the optional thickening, viscosity control and/or dispersing agents should be present in the compositions herein to the extent of from about 0.1% to 4% by weight. More preferably, such materials can comprise from about 0.5% to 2% by weight of the detergents compositions herein.
- compositions of the present invention can also optionally contain water-soluble ethoxylated amines having clay soil removal and anti-redeposition properties. If used, soil materials can contain from about 0.01% to about 5% by weight of the compositions herein.
- the most preferred soil release and anti-redeposition agent is ethoxylated tetraethylenepentamine. Exemplary ethoxylated amines are further described in U.S. Patent 4,597,898, VanderMeer, issued July 1, 1986.
- Another group of preferred clay soil removal-anti-redeposition agents are the cationic compounds disclosed in European Patent Application 111,965, Oh and Gosselink, published June 27, 1984.
- Other clay soil removal/anti-redeposition agents which can be used include the ethoxylated amine polymers disclosed in European Patent Application 111,984, Gosselink, published June 27, 1984; the zwitterionic polymers disclosed in European Patent Application 112,592, Gosselink, published July 4, 1984; and the amine oxides disclosed in U.S.
- CMC carboxy methyl cellulose
- Peroxygen bleaching agents may be organic or inorganic in nature. Inorganic peroxygen bleaching agents are frequently utilized in combination with a bleach activator.
- Useful organic peroxygen bleaching agents include percarboxylic acid bleaching agents and salts thereof. Suitable examples of this class of agents include magnesium monoperoxyphthalate hexahydrate, the magnesium salt of metachloro perbenzoic acid, 4- nonylamino-4-oxoperoxybutyric acid and diperoxydodecanedioic acid.
- Such bleaching agents are disclosed in U.S. Patent 4,483,781, Hartman, Issued November 20, 1984; European Patent Application EP-A-133,354, Banks et al., Published February 20, 1985; and U.S. Patent 4,412,934, Chung et al., Issued November 1, 1983.
- Highly preferred bleaching agents also include 6-nonylamino-6-oxoperoxycaproic acid (NAPAA) as described in U.S. Patent 4,634,551, Issued January 6, 1987 to Burns et al.
- NAPAA 6-nonylamino-6-oxoperoxycaproic acid
- Inorganic peroxygen bleaching agents may also be used in the detergent compositions herein.
- Inorganic bleaching agents are in fact preferred.
- Such inorganic peroxygen compounds include alkali metal perborate and percarbonate materials, most preferably the percarbonates.
- sodium perborate e.g. mono- or tetra- hydrate
- Suitable inorganic bleaching agents can also include sodium or potassium carbonate peroxyhydrate and equivalent "percarbonate" bleaches, sodium pyrophosphate peroxyhydrate, urea peroxyhydrate, and sodium peroxide.
- Persulfate bleach e.g., OXONE, manufactured commercially by DuPont
- OXONE manufactured commercially by DuPont
- inorganic peroxygen bleaches will be coated with silicate, borate, sulfate or water-soluble surfactants.
- coated percarbonate particles are available from various commercial sources such as FMC, Solvay Interox, Tokai Denka and Degussa.
- Inorganic peroxygen bleaching agents e.g., the perborates, the percarbonates, etc.
- bleach activators which lead to the in situ production in aqueous solution (i.e., during use of the compositions herein for fabric laundering/bleaching) of the peroxy acid corresponding to the bleach activator.
- Various non-limiting examples of activators are disclosed in U.S. Patent 4,915,854, Issued April 10, 1990 to Mao et al.; and U.S. Patent 4,412,934 Issued November 1, 1983 to Chung et al.
- NOBS nonanoyloxybenzene sulfonate
- TAED tetraacetyl ethylene diamine
- R is an alkyl group containing from about 6 to about 12 carbon atoms
- R 2 is an alkylene containing from 1 to about 6 carbon atoms
- R ⁇ is H or alkyl, aryl, or alkaryl containing from about 1 to about 10 carbon atoms
- L is any suitable leaving group, for example, oxybenzene sulfonate, -OOH, -OOM.
- a leaving group is any group that is displaced from the bleach activator as a consequence of the nucleophilic attack on the bleach activator by the perhydrolysis anion.
- a preferred leaving group is phenol sulfonate.
- bleach activators of the above formulae include (6- octanamido-caproyl)oxybenzenesulfonate, (6-nonanamidocaproyl) oxybenzenesulfonate, (6-decanamido-caproyl)oxybenzenesulfonate and mixtures thereof as described in the hereinbefore referenced U.S. Patent 4,634,551. Such mixtures are characterized herein as (6-Cg-C 1 o alkamido-caproyl)oxybenzenesulfonate.
- Another class of useful bleach activators comprises the benzoxazin-type activators disclosed by Hodge et al. in U.S. Patent 4,966, 723, Issued October 30, 1990, inco ⁇ orated herein by reference.
- a highly preferred activator of the benzoxazin-type is:
- Still another class of useful bleach activators includes the acyl lactam activators, especially acyl caprolactams and acyl valerolactams of the formulae:
- R ⁇ is H or an alkyl, aryl, alkoxyaryl, or alkaryl group containing from 1 to about 12 carbon atoms.
- Highly preferred lactam activators include benzoyl capro lactam, octanoyl caprolactam, 3,5,5-trimethylhexanoyl caprolactam, nonanoyl caprolactam, decanoyl caprolactam, undecenoyl caprolactam, benzoyl valerolactam, octanoyl valerolactam, decanoyl valerolactam, undecenoyl valerolactam, 3,5,5-trimethylhexanoyl valerolactam and mixtures thereof.
- peroxygen bleaching agents they will generally comprise from about 0.1% to 30% by weight of the composition. More preferably, peroxygen bleaching agent will comprise from about 1% to 20% by weight of the composition. Most preferably, peroxygen bleaching agent will be present to the extent of from about 5% to 20% by weight of the composition. If utilized, bleach activators can comprise from about 0.5% to 20%, more preferably from about 3% to 10%, by weight of the composition. Frequently, activators are employed such that the molar ratio of bleaching agent to activator ranges from about 1:1 to 10:1, more preferably from about 1.5:1 to 5:1.
- the bleaching compounds can be catalyzed by means of a manganese compound.
- a manganese compound Such compounds are well known in the art and include, for example, the manganese-based catalysts disclosed in U.S. Pat. 5,246,621, U.S. Pat. 5,244,594; U.S. Pat. 5,194,416; U.S. Pat. 5,114,606; and European Pat. App. Pub. Nos.
- Preferred examples of these catalysts include - n ⁇ 2( u -°)3( 1 ' 4 ' 7 - t ⁇ me 1 - 1 ' 4 ' 7 - tnazac clononane )2( PF 6)2' Mn ⁇ Cu-O i - OAc)2( 1 ,4,7-trimethyl- 1 ,4,7-triazacyclononane)2(Cl ⁇ 4)2, Mn rV 4(u-O)6( 1 ,4,7- triazacyclononane)4(Cl ⁇ 4)4, Mn ⁇ ⁇ Mn ⁇ -O) i (u-O Ac)2_( 1 ,4,7-trimethyl- 1 ,4,7- triazacyclononane)2(Cl ⁇ 4)3, Mn ⁇ l ,4,7-trimethyl- 1 ,4,7-triazacyclononane)-
- compositions and processes herein can be adjusted to provide on the order of at least one part per ten million of the active bleach catalyst species in the aqueous washing liquor, and will preferably provide from about 0J ppm to about 700 ppm, more preferably from about 1 ppm to about 500 ppm, of the catalyst species in the laundry liquor.
- Cobalt bleach catalysts useful herein are known, and are described, for example, in M. L. Tobe, "Base Hydrolysis of Transition-Metal Complexes", Adv. Inorg. Bioinorg. Mech., (1983), 2, pages 1-94.
- the most preferred cobalt catalyst useful herein are cobalt pentaamine acetate salts having the formula [Co(NH3)5OAc] T v , wherein "OAc” represents an acetate moiety and “Ty” is an anion, and especially cobalt pentaamine acetate chloride, [Co(NH3)5OAc]Cl2; as well as [Co(NH3)5OAc](OAc)2; [Co(NH 3 ) 5 OAc](PF 6 )2; [Co(NH 3 ) 5 OAc](SO4); [Co(NH 3 ) 5 OAc](BF 4 )2; and [Co(NH3) 5 OAc](N ⁇ 3) 2 (herein "PAC").
- compositions and cleaning processes herein can be adjusted to provide on the order of at least one part per hundred million of the active bleach catalyst species in the aqueous washing medium, and will preferably provide from about 0.01 ppm to about 25 ppm, more preferably from about 0.05 ppm to about 10 ppm, and most preferably from about 0.1 ppm to about 5 ppm, of the bleach catalyst species in the wash liquor.
- typical compositions herein will comprise from about 0.0005% to about 0.2%, more preferably from about 0.004% to about 0.08%, of bleach catalyst, especially manganese or cobalt catalysts, by weight of the cleaning compositions.
- the detergent compositions herein may also optionally contain conventional brighteners, suds suppressors, dyes and/or perfume materials.
- Such brighteners, suds suppressors, silicone oils, dyes and perfumes must, of course, be compatible and non- reactive with the other composition components in a non-aqueous environment. If present, brighteners suds suppressors, dyes and/or perfumes will typically comprise from about 0.0001% to 2% by weight of the compositions herein.
- Polymeric soil release agents are characterized by having both hydrophihc segments, to hydrophilize the surface of hydrophobic fibers, such as polyester and nylon, and hydrophobic segments, to deposit upon hydrophobic fibers and remain adhered thereto through completion of washing and rinsing cycles and, thus, serve as an anchor for the hydrophihc segments. This can enable stains occurring subsequent to treatment with the soil release agent to be more easily cleaned in later washing procedures.
- polymeric soil release agents useful herein include U.S. Patent 4,721,580, issued January 26, 1988 to Gosselink; U.S. Patent 4,000,093, issued December 28, 1976 to Nicol, et al.; European Patent Application 0 219 048, published April 22, 1987 by Kud, et al.; U.S. Patent 4,702,857, issued October 27, 1987 to Gosselink; U.S. Patent 4,968,451, issued November 6, 1990 to JJ. Scheibel.
- Commercially available soil release agents include the SOKALAN type of material, e.g., SOKALAN HP-22, available from BASF (West Germany). Also see U.S. Patent 3,959,230 to Hays, issued May 25, 1976 and U.S.
- this polymer examples include the commercially available material ZELCON 5126 (from Dupont) and MILEASE T (from ICI).
- Other suitable polymeric soil release agents include the terephthalate polyesters of U.S. Patent 4,711,730, issued December 8, 1987 to Gosselink et al, the anionic end-capped oligomeric esters of U.S. Patent 4,721,580, issued January 26, 1988 to Gosselink, and the block polyester oligomeric compounds of U.S. Patent 4,702,857, issued October 27, 1987 to Gosselink.
- Preferred polymeric soil release agents also include the soil release agents of U.S. Patent 4,877,896, issued October 31, 1989 to Maldonado et al. If utilized, soil release agents will generally comprise from about 0.01% to about 10.0%, by weight, of the detergent compositions herein, typically from about 0.1% to about 5%, preferably from about 0.2% to about 3.0%.
- the detergent compositions herein may also optionally contain one or more iron and/or manganese chelating agents.
- chelating agents can be selected from the group consisting of amino carboxylates, amino phosphonates, polyfunctionally-substituted aromatic chelating agents and mixtures therein, all as hereinafter defined. Without intending to be bound by theory, it is believed that the benefit of these materials is due in part to their exceptional ability to remove iron and manganese ions from washing solutions by formation of soluble chelates.
- Amino carboxylates useful as optional chelating agents include ethylenediamine- tetracetates, N-hydroxyethylethylenediaminetriacetates, nitrilo-triacetates, ethylenediamine tetraproprionates, triethylenetetraaminehexacetates, diethylenetriaminepentaacetates, and ethanoldiglycines, alkali metal, ammonium, and substituted ammonium salts therein and mixtures therein.
- Amino phosphonates are also suitable for use as chelating agents in the compositions of the invention when at lease low levels of total phosphorus are permitted in detergent compositions, and include ethylenediaminetetrakis (methylenephosphonates) as DEQUEST. Preferred, these amino phosphonates to not contain alkyl or alkenyl groups with more than about 6 carbon atoms.
- Polyfunctionally-substituted aromatic chelating agents are also useful in the compositions herein. See U.S. Patent 3,812,044, issued May 21, 1974, to Connor et al.
- Preferred compounds of this type in acid form are dihydroxydisulfobenzenes such as 1,2- dihydroxy-3,5-disulfobenzene.
- EDDS ethylenediamine disuccinate
- [S,S] isomer as described in U.S. Patent 4,704,233, November 3, 1987, to Hartman and Perkins.
- these chelating agents will generally comprise from about 0.1% to about 10% by weight of the detergent compositions herein. More preferably, if utilized, the chelating agents will comprise from about 0.1% to about 3.0% by weight of such compositions.
- Clay Soil Removal/ Anti-redeposition Agents will generally comprise from about 0.1% to about 10% by weight of the detergent compositions herein. More preferably, if utilized, the chelating agents will comprise from about 0.1% to about 3.0% by weight of such compositions.
- compositions of the present invention can also optionally contain water-soluble ethoxylated amines having clay soil removal and antiredeposition properties.
- Liquid detergent compositions typically contain about 0.01% to about 5% of these compositions.
- the most preferred soil release and anti-redeposition agent is ethoxylated tetraethylenepentamine.
- Exemplary ethoxylated amines are further described in U.S. Patent 4,597,898, VanderMeer, issued July 1, 1986.
- Another group of preferred clay soil removal-antiredeposition agents are the cationic compounds disclosed in European Patent Application 111,965, Oh and Gosselink, published June 27, 1984.
- Clay soil removal/antiredeposition agents which can be used include the ethoxylated amine polymers disclosed in European Patent Application 111,984, Gosselink, published June 27, 1984; the zwitterionic polymers disclosed in European Patent Application 112,592, Gosselink, published July 4, 1984; and the amine oxides disclosed in U.S. Patent 4,548,744, Connor, issued October 22, 1985.
- Other clay soil removal and/or anti redeposition agents known in the art can also be utilized in the compositions herein.
- Another type of preferred antiredeposition agent includes the carboxy methyl cellulose (CMC) materials. These materials are well known in the art.
- CMC carboxy methyl cellulose
- Polymeric dispersing agents can advantageously be utilized at levels from about 0.1% to about 7%, by weight, in the compositions herein, especially in the presence of zeolite and/or layered silicate builders.
- Suitable polymeric dispersing agents include polymeric polycarboxylates and polyethylene glycols, although others known in the art can also be used. It is believed, though it is not intended to be limited by theory, that polymeric dispersing agents enhance overall detergent builder performance, when used in combination with other builders (including lower molecular weight polycarboxylates) by crystal growth inhibition, particulate soil release peptization, and anti-redeposition.
- Polymeric polycarboxylate materials can be prepared by polymerizing or copolymerizing suitable unsaturated monomers, preferably in their acid form.
- Unsaturated monomeric acids that can be polymerized to form suitable polymeric polycarboxylates include acrylic acid, maleic acid (or maleic anhydride), f ⁇ imaric acid, itaconic acid, aconitic acid, mesaconic acid, citraconic acid and methylenemalonic acid.
- the presence in the polymeric polycarboxylates herein or monomeric segments, containing no carboxylate radicals such as vinylmethyl ether, styrene, ethylene, etc. is suitable provided that such segments do not constitute more than about 40% by weight.
- Particularly suitable polymeric polycarboxylates can be derived from acrylic acid.
- acrylic acid-based polymers which are useful herein are the water-soluble salts of polymerized acrylic acid.
- the average molecular weight of such polymers in the acid form preferably ranges from about 2,000 to 10,000, more preferably from about 4,000 to 7,000 and most preferably from about 4,000 to 5,000.
- Water-soluble salts of such acrylic acid polymers can include, for example, the alkali metal, ammonium and substituted ammonium salts. Soluble polymers of this type are known materials. Use of polyacrylates of this type in detergent compositions has been disclosed, for example, in Diehl, U.S. Patent 3,308,067, issued march 7, 1967.
- Acrylic/maleic-based copolymers may also be used as a preferred component of the dispersing/anti-redeposition agent.
- Such materials include the water-soluble salts of copolymers of acrylic acid and maleic acid.
- the average molecular weight of such copolymers in the acid form preferably ranges from about 2,000 to 100,000, more preferably from about 5,000 to 75,000, most preferably from about 7,000 to 65,000.
- the ratio of acrylate to maleate segments in such copolymers will generally range from about 30:1 to about 1:1, more preferably from about 10:1 to 2:1.
- Water-soluble salts of such acrylic acid/maleic acid copolymers can include, for example, the alkali metal, ammonium and substituted ammonium salts.
- Soluble acrylate/maleate copolymers of this type are known materials which are described in European Patent Application No. 66915, published December 15, 1982, as well as in EP 193,360, published September 3, 1986, which also describes such polymers comprising hydroxypropylacrylate.
- Still other useful dispersing agents include the maleic/acrylic/vinyl alcohol te ⁇ olymers.
- Such materials are also disclosed in EP 193,360, including, for example, the 45/45/10 te ⁇ olymer of acrylic/maleic/vinyl alcohol.
- PEG polyethylene glycol
- PEG can exhibit dispersing agent performance as well as act as a clay soil removal- antiredeposition agent.
- Typical molecular weight ranges for these pu ⁇ oses range from about 500 to about 100,000, preferably from about 1,000 to about 50,000, more preferably from about 1,500 to about 10,000.
- Polyaspartate and polyglutamate dispersing agents may also be used, especially in conjunction with zeolite builders.
- Dispersing agents such as polyaspartate preferably have a molecular weight (avg.) of about 10,000.
- compositions of the present invention may also include one or more materials effective for inhibiting the transfer of dyes from one fabric to another during the cleaning process.
- dye transfer inhibiting agents include polyvinyl pyrrolidone polymers, polyamine N-oxide polymers, copolymers of N-vinylpyrrolidone and N- vinylimidazole, manganese phthalocyanine, peroxidases, and mixtures thereof. If used, these agents typically comprise from about 0.01% to about 10% by weight of the composition, preferably from about 0.01% to about 5%, and more preferably from about 0.05% to about 2%.
- Preferred polyamine N-oxides are those wherein R is a heterocyclic group such as pyridine, pyrrole, imidazole, pyrrolidine, piperidine and derivatives thereof.
- the N-O group can be represented by the following general structures:
- Ri , R2, R3 are aliphatic, aromatic, heterocyclic or alicyclic groups or combinations thereof; x, y and z are 0 or 1 ; and the nitrogen of the N-O group can be attached or form part of any of the aforementioned groups.
- the amine oxide unit of the polyamine N-oxides has a pKa ⁇ 10, preferably pKa ⁇ 7, more preferred pKa ⁇ 6.
- Any polymer backbone can be used as long as the amine oxide polymer formed is water-soluble and has dye transfer inhibiting properties.
- suitable polymeric backbones are polyvinyls, polyalkylenes, polyesters, polyethers, polyamide, polyimides, polyacrylates and mixtures thereof. These polymers include random or block copolymers where one monomer type is an amine N-oxide and the other monomer type is an N-oxide.
- the amine N-oxide polymers typically have a ratio of amine to the amine N-oxide of 10:1 to 1:1,000,000. However, the number of amine oxide groups present in the polyamine oxide polymer can be varied by appropriate copolymerization or by an appropriate degree of N-oxidation.
- the polyamine oxides can be obtained in almost any degree of polymerization. Typically, the average molecular weight is within the range of 500 to 1,000,000; more preferred 1,000 to 500,000; most preferred 5,000 to 100,000. This preferred class of materials can be referred to as "PVNO".
- poly(4-vinylpyridine-N-oxide) which as an average molecular weight of about 50,000 and an amine to amine N-oxide ratio of about 1:4.
- Copolymers of N-vinylpyrrolidone and N-vinylimidazole polymers are also preferred for use herein.
- the PVPVI has an average molecular weight range from 5,000 to 1,000,000, more preferably from 5,000 to 200,000, and most preferably from 10,000 to 20,000. (The average molecular weight range is determined by light scattering as described in Barth, et al., Chemical Analysis. Vol 113.
- the PVPVI copolymers typically have a molar ratio of N-vinylimidazole to N-vinylpyrrolidone from 1:1 to 0.2:1, more preferably from 0.8:1 to 0.3:1, most preferably from 0.6:1 to 0.4:1. These copolymers can be either linear or branched.
- compositions also may employ a polyvinylpyrrolidone (“PVP”) having an average molecular weight of from about 5,000 to about 400,000, preferably from about 5,000 to about 200,000, and more preferably from about 5,000 to about 50,000.
- PVP's are known to persons skilled in the detergent field; see, for example, EP-A-262,897 and EP-A-256,696, inco ⁇ orated herein by reference.
- Compositions containing PVP can also contain polyethylene glycol (“PEG”) having an average molecular weight from about 500 to about 100,000, preferably from about 1,000 to about 10,000.
- PEG polyethylene glycol
- the ratio of PEG to PVP on a ppm basis delivered in wash solutions is from about 2:1 to about 50:1, and more preferably from about 3:1 to about 10:1.
- the detergent compositions herein may also optionally contain from about 0.005% to 5% by weight of certain types of hydrophihc optical brighteners which also provide a dye transfer inhibition action. If used, the compositions herein will preferably comprise from about 0.01% to 1% by weight of such optical brighteners.
- hydrophihc optical brighteners useful in the present invention are those having the structural formula:
- Ri is selected from anilino, N-2-bis-hydroxyethyl and NH-2-hydroxyethyl
- R2 is selected from N-2-bis-hydroxyethyl, N-2-hydroxyethyl-N-methylamino, mo ⁇ hilino, chloro and amino
- M is a salt-forming cation such as sodium or potassium.
- Rj is anilino
- R2 is N-2-bis-hydroxyethyl and M is a cation such as sodium
- the brightener is 4,4',-bis[(4-anilino-6-(N-2-bis-hydroxyethyl)-s- triazine-2-yl)amino]-2,2'-stilbenedisulfonic acid and disodium salt.
- This particular brightener species is commercially marketed under the tradename Tinopal-UNPA-GX by Ciba-Geigy Co ⁇ oration. Tinopal-UNPA-GX is the preferred hydrophihc optical brightener useful in the detergent compositions herein.
- R ⁇ is anilino
- R2 is N-2-hydroxyethyl-N-2- methylamino
- M is a cation such as sodium
- the brightener is 4,4'-bis[(4-anilino-6-(N- 2-hydroxyethyl-N-methylamino)-s-triazine-2-yl)amino]2,2 , -stilbenedisulfonic acid disodium salt.
- This particular brightener species is commercially marketed under the tradename Tinopal 5BM-GX by Ciba-Geigy Co ⁇ oration.
- R ⁇ is anilino
- R2 is mo ⁇ hilino
- M is a cation such as sodium
- the brightener is 4,4'-bis[(4-anilino-6-mo ⁇ hilino-s-triazine-2-yl)amino]2,2'- stilbenedisulfonic acid, sodium salt.
- This particular brightener species is commercially marketed under the tradename Tinopal AMS-GX by Ciba Geigy Co ⁇ oration.
- the specific optical brightener species selected for use in the present invention provide especially effective dye transfer inhibition performance benefits when used in combination with the selected polymeric dye transfer inhibiting agents hereinbefore described.
- the combination of such selected polymeric materials (e.g., PVNO and/or PVPVI) with such selected optical brighteners (e.g., Tinopal UNPA-GX, Tinopal 5BM- GX and/or Tinopal AMS-GX) provides significantly better dye transfer inhibition in aqueous wash solutions than does either of these two detergent composition components when used alone. Without being bound by theory, it is believed that such brighteners work this way because they have high affinity for fabrics in the wash solution and therefore deposit relatively quick on these fabrics.
- the extent to which brighteners deposit on fabrics in the wash solution can be defined by a parameter called the "exhaustion coefficient".
- the exhaustion coefficient is in general as the ratio of a) the brightener material deposited on fabric to b) the initial brightener concentration in the wash liquor. Brighteners with relatively high exhaustion coefficients are the most suitable for inhibiting dye transfer in the context of the present invention.
- the aqueous based heavy-duty liquid detergent compositions disclosed herein can contain water and other solvents as carriers.
- Low molecular weight primary or secondary alcohols exemplified by methanol, ethanol, propanol, and isopropanol are suitable.
- Monohydric alcohols are preferred for solubilizing surfactant, but polyols such as those containing from 2 to about 6 carbon atoms and from 2 to about 6 hydroxy groups (e.g., 1,3-propanediol, ethylene glycol, glycerine, and 1,2-propanediol) can also be used.
- the compositions may contain from 5% to 90%, typically 10% to 50% of such carriers.
- the detergent compositions herein will preferably be formulated such that, during use in aqueous cleaning operations, the wash water will have a pH of between about 6.5 and about 11, preferably between about 7.5 and 11.
- Techniques for controlling pH at recommended usage levels include the use of buffers, alkalis, acids, etc., and are well known to those skilled in the art. COMPOSITION PREPARATION AND USE
- aqueous based heavy-duty liquid detergent compositions of the present invention can be made by mixing and blending the desired ingredients with the desired sovent. In a typical process for preparing such compositions, essential and certain preferred optional components will be combined in a particular order and under certain conditions.
- compositions of this invention can be used to form aqueous washing solutions for use in the laundering and bleaching of fabrics.
- an effective amount of such compositions is added to water, preferably in a conventional fabric laundering automatic washing machine, to form such aqueous laundering/bleaching solutions.
- the aqueous washing/bleaching solution so formed is then contacted, preferably under agitation, with the fabrics to be laundered and bleached therewith.
- An effective amount of the liquid detergent compositions herein added to water to form aqueous laundering/bleaching solutions can comprise amounts sufficient to form from about 500 to 7,000 ppm of composition in aqueous solution. More preferably, from about 800 to 3,000 ppm of the detergent compositions herein will be provided in aqueous washing/bleaching solution.
- MBAE X Mid-chain branched primary alkyl (average total carbons
- Citric acid Anhydrous citric acid
- Carbonate Anhydrous sodium carbonate with a particle size between 200 ⁇ m and 900 ⁇ m
- TPK Fatty Acid
- Nai2(A102SiO2)i2- 27H2O having a primary particle size in the range from 0J to 10 micrometers
- Bicarbonate Anhydrous sodium bicarbonate with a particle size distribution between 400 ⁇ m and 1200 ⁇ m
- MA/AA Copolymer of 1 :4 maleic/acrylic acid average molecular weight about 70,000.
- Brightener 1 Disodium 4,4'-bis(2-sulphostyryl)biphenyl
- Brightener 2 Disodium 4,4'-bis(4-anilino-6-mo ⁇ holino-l .3.5- triazin-2-yl)amino) stilbene-2 :2'-disulfonate.
- liquid detergent compositions (A-D) are found to be very efficient in the removal of a wide range of stains and soils from fabrics under various usage conditions.
- EXAMPLE 26 The following compositions (E to J) are heavy duty liquid laundry detergent com ositions accordin to the resent invention.
- aqueous liquid laundry detergent compositions K to O are prepared in accord with the invention:
Landscapes
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Wood Science & Technology (AREA)
- Organic Chemistry (AREA)
- Detergent Compositions (AREA)
- Low-Molecular Organic Synthesis Reactions Using Catalysts (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
Abstract
Description
Claims
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP99968121A EP1144571A3 (en) | 1999-01-20 | 1999-12-15 | Aqueous heavy duty liquid detergent compositions comprising modified alkylbenzene sulfonates |
| AU24801/00A AU2480100A (en) | 1999-01-20 | 1999-12-15 | Aqueous heavy duty liquid detergent compositions comprising modified alkylbenzene sulfonates |
| BR9916940-1A BR9916940A (en) | 1999-01-20 | 1999-12-15 | Heavy-duty aqueous liquid detergent compositions comprising modified alkyl benzenesulfonates |
| CA002358856A CA2358856A1 (en) | 1999-01-20 | 1999-12-15 | Aqueous heavy duty liquid detergent compositions comprising modified alkylbenzene sulfonates |
| JP2000594882A JP2003518518A (en) | 1999-01-20 | 1999-12-15 | Aqueous heavy-duty liquid detergent composition comprising a modified alkylbenzene sulfonate |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US11649799P | 1999-01-20 | 1999-01-20 | |
| US60/116,497 | 1999-01-20 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2000043473A2 true WO2000043473A2 (en) | 2000-07-27 |
| WO2000043473A3 WO2000043473A3 (en) | 2002-10-03 |
Family
ID=22367514
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US1999/029717 Ceased WO2000043473A2 (en) | 1999-01-20 | 1999-12-15 | Aqueous heavy duty liquid detergent compositions comprising modified alkylbenzene sulfonates |
Country Status (6)
| Country | Link |
|---|---|
| EP (1) | EP1144571A3 (en) |
| JP (1) | JP2003518518A (en) |
| AU (1) | AU2480100A (en) |
| BR (1) | BR9916940A (en) |
| CA (1) | CA2358856A1 (en) |
| WO (1) | WO2000043473A2 (en) |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2002092737A1 (en) * | 2001-05-16 | 2002-11-21 | The Procter & Gamble Company | Laundry detergents comprising modified and enhanced alkylbenzene sulfonates |
| US6696402B2 (en) | 1999-11-09 | 2004-02-24 | The Procter & Gamble Company | Laundry detergent compositions comprising zwitterionic polyamines |
| US7163985B2 (en) | 2002-09-12 | 2007-01-16 | The Procter & Gamble Co. | Polymer systems and cleaning compositions comprising the same |
| WO2010031689A1 (en) * | 2008-09-17 | 2010-03-25 | Henkel Ag & Co. Kgaa | All-purpose cleaner with improved cleaning performance in diluted application |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| BR112015028674A2 (en) * | 2013-05-24 | 2017-07-25 | Procter & Gamble | fluid compact laundry detergent composition |
| EP3039109B1 (en) | 2013-08-26 | 2017-09-13 | The Procter and Gamble Company | Compositions comprising alkoxylated polyamines having low melting points |
Family Cites Families (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE69318393T2 (en) * | 1991-12-23 | 1998-09-03 | Uop Inc | Alkylation process for benzene using a fluorinated silica / alumina and a C6-C20 monoolefin |
| PH11998001775B1 (en) * | 1997-07-21 | 2004-02-11 | Procter & Gamble | Improved alkyl aryl sulfonate surfactants |
| AU737587B2 (en) * | 1997-08-08 | 2001-08-23 | Procter & Gamble Company, The | Improved processes for making surfactants via adsorptive separation and products thereof |
| BR9913663A (en) * | 1998-09-02 | 2001-06-05 | Procter & Gamble | Improved processes for producing surfactants via adsorbable separation and products from these processes |
-
1999
- 1999-12-15 WO PCT/US1999/029717 patent/WO2000043473A2/en not_active Ceased
- 1999-12-15 JP JP2000594882A patent/JP2003518518A/en not_active Withdrawn
- 1999-12-15 EP EP99968121A patent/EP1144571A3/en not_active Withdrawn
- 1999-12-15 AU AU24801/00A patent/AU2480100A/en not_active Abandoned
- 1999-12-15 CA CA002358856A patent/CA2358856A1/en not_active Abandoned
- 1999-12-15 BR BR9916940-1A patent/BR9916940A/en not_active IP Right Cessation
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6596680B2 (en) | 1997-07-21 | 2003-07-22 | The Procter & Gamble Company | Enhanced alkylbenzene surfactant mixture |
| US6696402B2 (en) | 1999-11-09 | 2004-02-24 | The Procter & Gamble Company | Laundry detergent compositions comprising zwitterionic polyamines |
| WO2002092737A1 (en) * | 2001-05-16 | 2002-11-21 | The Procter & Gamble Company | Laundry detergents comprising modified and enhanced alkylbenzene sulfonates |
| US7163985B2 (en) | 2002-09-12 | 2007-01-16 | The Procter & Gamble Co. | Polymer systems and cleaning compositions comprising the same |
| US7442213B2 (en) | 2002-09-12 | 2008-10-28 | The Procter & Gamble Company | Methods of cleaning a situs with a cleaning composition comprising a polymer system |
| WO2010031689A1 (en) * | 2008-09-17 | 2010-03-25 | Henkel Ag & Co. Kgaa | All-purpose cleaner with improved cleaning performance in diluted application |
Also Published As
| Publication number | Publication date |
|---|---|
| JP2003518518A (en) | 2003-06-10 |
| WO2000043473A3 (en) | 2002-10-03 |
| CA2358856A1 (en) | 2000-07-27 |
| EP1144571A3 (en) | 2002-11-20 |
| EP1144571A2 (en) | 2001-10-17 |
| AU2480100A (en) | 2000-08-07 |
| BR9916940A (en) | 2001-11-20 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| AU763324B2 (en) | Laundry detergents comprising modified alkylbenzene sulfonates | |
| US6583096B1 (en) | Laundry detergents comprising modified alkylbenzene sulfonates | |
| US6274540B1 (en) | Detergent compositions containing mixtures of crystallinity-disrupted surfactants | |
| US6596680B2 (en) | Enhanced alkylbenzene surfactant mixture | |
| US6593285B1 (en) | Alkylbenzenesulfonate surfactants | |
| EP1194402B1 (en) | Detergent compositions containing modified alkylaryl sulfonate surfactants | |
| JP2020125478A (en) | Detergent for cold-water cleaning | |
| KR20000005494A (en) | Mid-chain branched primary alkyl sulphate as surfactants | |
| KR20000005562A (en) | Heavy chain side-chained detergent | |
| WO2000043478A1 (en) | Aqueous heavy duty liquid detergent compositions comprising modified alkylbenzene sulfonates | |
| WO2000043473A2 (en) | Aqueous heavy duty liquid detergent compositions comprising modified alkylbenzene sulfonates | |
| WO1999019450A1 (en) | Aqueous and non-aqueous heavy duty liquid detergent compositions comprising mid-chain branched surfactants | |
| MXPA01007351A (en) | Aqueous heavy duty liquid detergent compositions comprising modified alkylbenzene sulfonates | |
| MXPA01007344A (en) | Aqueous heavy duty liquid detergent compositions comprising modified alkylbenzene sulfonates | |
| CN1457358A (en) | Aqueous heavy duty liquid detergent composition containing modified alkylbenzene sulfonate | |
| MXPA01004008A (en) | Laundry detergents comprising modified alkylbenzene sulfonates |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 99815763.5 Country of ref document: CN |
|
| AK | Designated states |
Kind code of ref document: A2 Designated state(s): AE AL AM AT AT AU AZ BA BB BG BR BY CA CH CN CR CU CZ CZ DE DE DK DK DM EE EE ES FI FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX NO NZ PL PT RO RU SD SE SG SI SK SK SL TJ TM TR TT UA UG US UZ VN YU ZA ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A2 Designated state(s): GH GM KE LS MW SD SL SZ TZ UG ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE BF BJ CF CG CI CM GA GN GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| WWE | Wipo information: entry into national phase |
Ref document number: IN/PCT/2001/00587/DE Country of ref document: IN |
|
| ENP | Entry into the national phase |
Ref document number: 2358856 Country of ref document: CA Ref document number: 2358856 Country of ref document: CA Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: PA/a/2001/007344 Country of ref document: MX Ref document number: 09889637 Country of ref document: US |
|
| ENP | Entry into the national phase |
Ref document number: 2000 594882 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1999968121 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 1999968121 Country of ref document: EP |
|
| REG | Reference to national code |
Ref country code: DE Ref legal event code: 8642 |
|
| AK | Designated states |
Kind code of ref document: A3 Designated state(s): AE AL AM AT AT AU AZ BA BB BG BR BY CA CH CN CR CU CZ CZ DE DE DK DK DM EE EE ES FI FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX NO NZ PL PT RO RU SD SE SG SI SK SK SL TJ TM TR TT UA UG US UZ VN YU ZA ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A3 Designated state(s): GH GM KE LS MW SD SL SZ TZ UG ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE BF BJ CF CG CI CM GA GN GW ML MR NE SN TD TG |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: 1999968121 Country of ref document: EP |