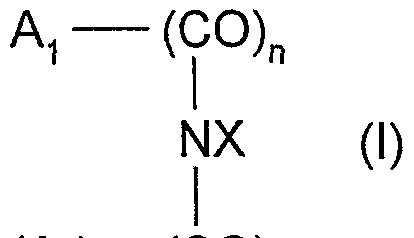WO2002007687A1 - Composition a base d'enzymes pour la decoloration des fibres keratiniques humaines et procede de decoloration - Google Patents
Composition a base d'enzymes pour la decoloration des fibres keratiniques humaines et procede de decoloration Download PDFInfo
- Publication number
- WO2002007687A1 WO2002007687A1 PCT/FR2001/002091 FR0102091W WO0207687A1 WO 2002007687 A1 WO2002007687 A1 WO 2002007687A1 FR 0102091 W FR0102091 W FR 0102091W WO 0207687 A1 WO0207687 A1 WO 0207687A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- composition according
- composition
- phenothiazine
- chosen
- radical
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 C*CCC1CCCC1 Chemical compound C*CCC1CCCC1 0.000 description 3
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q5/00—Preparations for care of the hair
- A61Q5/08—Preparations for bleaching the hair
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/49—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds
- A61K8/494—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds with more than one nitrogen as the only hetero atom
- A61K8/4966—Triazines or their condensed derivatives
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/64—Proteins; Peptides; Derivatives or degradation products thereof
- A61K8/66—Enzymes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/96—Cosmetics or similar toiletry preparations characterised by the composition containing materials, or derivatives thereof of undetermined constitution
- A61K8/97—Cosmetics or similar toiletry preparations characterised by the composition containing materials, or derivatives thereof of undetermined constitution from algae, fungi, lichens or plants; from derivatives thereof
- A61K8/9728—Fungi, e.g. yeasts
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/96—Cosmetics or similar toiletry preparations characterised by the composition containing materials, or derivatives thereof of undetermined constitution
- A61K8/97—Cosmetics or similar toiletry preparations characterised by the composition containing materials, or derivatives thereof of undetermined constitution from algae, fungi, lichens or plants; from derivatives thereof
- A61K8/9755—Gymnosperms [Coniferophyta]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/96—Cosmetics or similar toiletry preparations characterised by the composition containing materials, or derivatives thereof of undetermined constitution
- A61K8/97—Cosmetics or similar toiletry preparations characterised by the composition containing materials, or derivatives thereof of undetermined constitution from algae, fungi, lichens or plants; from derivatives thereof
- A61K8/9771—Ginkgophyta, e.g. Ginkgoaceae [Ginkgo family]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/96—Cosmetics or similar toiletry preparations characterised by the composition containing materials, or derivatives thereof of undetermined constitution
- A61K8/97—Cosmetics or similar toiletry preparations characterised by the composition containing materials, or derivatives thereof of undetermined constitution from algae, fungi, lichens or plants; from derivatives thereof
- A61K8/9783—Angiosperms [Magnoliophyta]
- A61K8/9789—Magnoliopsida [dicotyledons]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/96—Cosmetics or similar toiletry preparations characterised by the composition containing materials, or derivatives thereof of undetermined constitution
- A61K8/97—Cosmetics or similar toiletry preparations characterised by the composition containing materials, or derivatives thereof of undetermined constitution from algae, fungi, lichens or plants; from derivatives thereof
- A61K8/9783—Angiosperms [Magnoliophyta]
- A61K8/9794—Liliopsida [monocotyledons]
Definitions
- the subject of the invention is a ready-to-use composition for bleaching human keratin fibers previously dyed with direct dyes, in particular hair, comprising at least one enzyme of the 4-electron oxidoreductase type, and at least one enzymatic mediator, as well as the bleaching process using this composition.
- direct dyes such as benzene, neutral, acidic or cationic direct dyes, direct azo, neutral, acid or cationic dyes, direct dyes quinonics and in particular anthraquinonics, neutral, acidic or cationic, indigoids, neutral, acidic or cationic dyes, azine direct dyes, triarylmethane direct dyes, indoamine direct dyes and natural direct dyes.
- the Applicant has now just discovered in a completely unexpected and surprising manner, that it is possible to partially or totally bleach human keratin fibers previously dyed with direct dyes, and in particular hair, using a composition comprising at least at least one 4-electron oxidoreductase type enzyme, and at least one enzyme mediator.
- the discoloration obtained is regular and homogeneous without causing significant degradation of the keratin fibers.
- the first object of the invention is therefore a ready-to-use composition for bleaching human keratin fibers previously dyed with at least one direct dye, in particular hair, characterized in that it comprises at least one enzyme. oxidoreductase type with 4 electrons, and at least one enzymatic mediator, said composition being free of oxidation base. Said discoloration may be partial or total.
- the subject of the invention is also a bleaching process for bleaching human keratin fibers previously dyed with at least one direct dye, in particular hair, using a ready-to-use composition for bleaching as described above .
- enzymatic mediator any compound capable of increasing the enzymatic activity of said 4-electron oxidoreductase.
- ready-to-use composition within the meaning of the invention, a composition intended to be applied as it is to keratin fibers, that is to say that it can be stored as it is before use or result from an extemporaneous mixture of two or more compositions, for example, a composition containing at least one 4-electron oxidoreductase and another comprising at least one enzymatic mediator .
- the enzymatic mediator can be chosen from the compounds of formula (I) below and their possible tautomeric forms:
- the nitrogen atom of the NX group which can form, with the groups A ⁇ - (CO) n and A 2 - (CO) p, a heterocycle comprising from 5 to 18 members, said heterocycle may or may not be substituted by one or more alkyl radicals in C ⁇ -C 4 , hydroxyl, phenyl, halogen, sulfo, carboxy or nitro;
- n and p identical or different, are whole numbers equal to 0 or 1.
- hydroxylamine N, N-dipropyl-hydroxy! Amine, N, N-diisopropyl-hydroxylamine, phenyl-hydroxylamine, N-acetyl hydroxylamine, 1-phenyl-1 H-1, 2,3-triazole-1-oxide, 2,4,5-triphenyl-2H- 1,2,3-triazoM-oxide, 1-hydroxy-benzotriazole, 1-hydroxy benzotriazole sulfonic acid, 1-hydroxy-benzimidazole, N-hydroxy-phthalimide, N-hydroxy-succinimide, quinoline-N-oxide, isoquinoline-N-oxide, 1-hydroxy- piperidine, violuric acid, 4-hydroxy-3-nitroso-coumarin, 1, 3-dimethyl-5-nitroso-barbituric acid, 1-nitroso-2-naphthol, 2-nitroso-1 acid -napht
- the enzymatic mediator can also be chosen from the compounds of formula (II) or of formula (III) below:
- R-l represents a group COR CH ⁇ CHR ⁇ CH ⁇ CH-CH ⁇ CHR
- CH CHCOR4, SO2R4, POR4R5 ;
- acetosyringone syringaldehyde, methylsyringate, syringic acid, ethylsyringate, butylsyringate, hexylsyringate, octylsyringate or the ethyl ester of 3- (4-hydroxy-3,5-dimethoxyphenyl) -acrylic acid.
- the enzymatic mediator can also be chosen from the compounds of formula (IV) below:
- X represents a sulfur or oxygen atom
- RQ to R-16 independently of one another, denote a hydrogen atom, a halogen atom, a hydroxy, formyl, carboxy, carboxyalkyl, carbamoyl, sulfo, sulfoalkyl, sulfamoyl, nitro, amino, phenyl radical , alkyl, alkoxy, carbonylalkyl, arylalkyl, these radicals being able to be substituted by one or more substituents R-17;
- R-17 denotes a halogen atom or a hydroxy, formyl, carboxy, carboxyalkyl, carbamoyl, sulfo, sulfoalkyl, sulfamoyl, nitro, amino, phenyl, alkyl, aminoalkyl, piperidino, piperazinyl, pyrrolidino, alkoxy radical, these substituents being able to, where appropriate, be themselves substituted by one or more R-17 substituents; two of the substituents Rs to RJ Q which can, together with the carbon atoms carrying them, form a saturated or unsaturated ring, containing or not one or more heteroatoms, substituted or not by one or more substituents RQ.
- the salts of 2,2'-azinobis (3-alkylbenzothiazoline-6-sulfonic) acids such as the diammonium salt of 2,2'-azinobis (3-ethylbenzothiazoline-6-) can also be used as an enzyme mediator sulfonic acid).
- the enzymatic mediator (s) used in the composition in accordance with the invention preferably represent from 0.0001 to 5% by weight approximately relative to the total weight of the composition and preferably from 0.005 to 0.5% by weight approximately. weight.
- the 4-electron oxidoreductase (s) used in the composition according to the invention can in particular be chosen from laccases, tyrosinases, catechol oxidases and polyphenol oxidases.
- the 4-electron oxidoreductase (s) are chosen from laccases.
- laccases can in particular be chosen from laccases of plant origin, of animal origin, of fungal origin (yeasts, molds, fungi) or of bacterial origin, the organisms of origin being able to be mono- or multicellular. Laccases can also be obtained by biotechnology.
- laccases of plant origin which can be used according to the invention, mention may be made of laccases produced by plants carrying out chlorophyll synthesis such as those indicated in patent application FR-A-2 694 018. Mention may in particular be made of the laccases present in extracts of Anacardiaceae such as for example extracts of Magnifera indica, Schinus molle or Pleiogynium timoriense; in extracts from Podocarpaceae; of Rosmarinus off. ; Solanum tuberosum; Iris sp. ; from Coffea sp. ; Daucus carrota; of Vinca minor; Persea americana; Catharenthus roseus; from Musa sp.
- Anacardiaceae such as for example extracts of Magnifera indica, Schinus molle or Pleiogynium timoriense
- extracts from Podocarpaceae of Rosmarinus off.
- Solanum tuberosum Iris sp
- laccases of fungal origin possibly obtained by biotechnology, which can be used according to the invention, mention may be made of the laccase (s) obtained from Polyporus versicolor, Rhizoctonia pratola and Rhus vernicifera as described for example in patent applications FR- A-2 112 549 and EP-A-504005; the laccases described in the patent applications
- Podospora anserina Agaricus bisporus, Ganoderma lucidum, Glomerella cingulata, from Lactarius piperatus, from Russula delica, from Heterobasidion annosum, from Thelephora terrestris, from Cladosporium cladosporioides, from Cerrena unicolor, from Coriolus hirsutus, from Ceriporiopsis subvermispora, from Coprinus cinereus, from Panaeolus papinus ses, of Dichomitius squalens, and their variants.
- laccases of fungal origin possibly obtained by biotechnology
- the enzymatic activity of the laccases used in accordance with the invention and having syringaldazine among their substrates can be defined from the oxidation of syringaldazine in aerobic condition.
- the Lacu unit corresponds to the quantity of enzyme catalyzing the conversion of 1 mmol of syringaldazine per minute at a pH of 5.5 and at a temperature of 30 ° C.
- Unit U corresponds to the quantity of enzyme producing an absorbance delta of 0.001 per minute, at a wavelength of 530 nm, using syringaldazine-as a substrate, at 30 ° C. and at a pH of 6 5.
- the enzymatic activity of the laccases used according to the invention can also be defined from the oxidation of paraphenylenediamine.
- the ulac unit corresponds to the quantity of enzyme producing an absorbance delta of 0.001 per minute, at a wavelength of 496.5 nm, using paraphenylenediamine as substrate (64 mM), at 30 ° C and at a pH of 5.
- the 4-electron oxidoreductase (s) in accordance with the invention preferably represent from 0.01 to 20% by weight approximately of the total weight of the composition, and even more preferably from 0.1 to 5% by weight. weight about that weight.
- the amount of laccase (s) present in the composition according to the invention will vary depending on the nature of the laccase (s) used.
- the amount of laccase (s) is between 0.5 and 200 Lacu approximately (either between 10,000 and 4.10 6 units U approximately or between 20 and 2.10 6 units ulac) per 100 g of composition.
- Human keratin fibers which can be discolored according to the process of the invention are those dyed with at least one direct dye.
- the direct dyes that can be used are preferably chosen from benzene, neutral, acidic or cationic direct dyes, azo, neutral, acidic or cationic direct dyes, quinone direct dyes and in particular anthraquinone, neutral, acidic or cationic dyes, indigo dyes , neutral, acidic or cationic, azine direct dyes, triarylmethane direct dyes, indoamine direct dyes and natural direct dyes.
- azo direct dyes mention may be made of the cationic azo dyes described in patent applications WO 95/15144, WO-95/01772 and EP-714954, the content of which forms an integral part of the invention.
- the following dyes may be mentioned -Disperse Red 15; Solvent Violet 13; Acid Violet 43; Disperse Violet 1; Disperse Violet 4; Disperse Blue 1; Disperse Violet 8; Disperse Blue 3; Disperse Red 11; Acid Blue 62; Disperse Blue 7; Basic Blue 22; Disperse Violet 15; Basic Blue 99, as well as the following compounds:
- azine dyes mention may be made of the following compounds such as Basic Blue 17 and Basic Red 2.
- triarylmethane dyes mention may be made of the following compounds " : -Basic Green 1; Acid blue 9; Basic Violet 3; Basic Violet 14; Basic Blue 7; Acid Violet 49; Basic Blue 26; Acid Blue 7.
- indoamine dyes mention may be made of the following compounds: -2- ⁇ -hydroxyethylamino-5- [bis- (-4'-hydroxyethyl) -amino] -anilino-1,4-benzoquinone; -2- ⁇ -hydroxyethylamino-5- (2'-methoxy-4'-amino) -anilino-1, 4-benzoquinone;
- the bleaching method according to the invention will be used to bleach hair totally or partially dyed with benzene, neutral, acidic or cationic direct dyes, direct azo, neutral or cationic dyes, direct quinone dyes and especially cationic anthraquinonics and indoamine direct dyes.
- the medium suitable for bleaching (or support) of the ready-to-use composition for bleaching generally consists of water or of a mixture of water and at least one solvent organic to dissolve the compounds which would not be sufficiently soluble in water.
- organic solvent of C 1 -C 4 alkanols, such as ethanol and isopropanol; glycerol; glycols and glycol ethers such as 2-butoxyethanol, propylene glycol, propylene glycol monomethyl ether, monoethyl ether and diethylene glycol monomethyl ether, as well as aromatic alcohols such as benzyl alcohol or phenoxyethanol, analogous products and mixtures thereof.
- the solvents may be present in proportions preferably of between 1 and 40% by weight approximately relative to the total weight of the composition ready for use for bleaching, and even more preferably between 5 and 30% by weight approximately.
- the pH of the composition ready for use for bleaching is chosen such that the enzymatic activity of the 4-electron oxidoreductase is sufficient. It is generally between 3 and
- acidifying agents there may be mentioned, by way of example, mineral or organic acids such as hydrochloric acid, orthophosphoric acid, sulfuric acid, carboxylic acids such as acetic acid, tartaric acid, citric acid, lactic acid, sulfonic acids.
- mineral or organic acids such as hydrochloric acid, orthophosphoric acid, sulfuric acid, carboxylic acids such as acetic acid, tartaric acid, citric acid, lactic acid, sulfonic acids.
- basifying agents there may be mentioned, by way of example, ammonia, alkali carbonates, alkanolamines such as mono-, di- and triethanolamines, 2-methyl-2-amino-1-propanol as well as their derivatives, sodium or potassium hydroxides and the compounds of formula (V) below:
- W is a propylene residue optionally substituted by a hydroxyl group or a C 1 -C 4 alkyl radical
- 6, R 17 and R 18, identical or different, represent a hydrogen atom, an alkyl radical in C 1 -C 4 alkyl or hydroxyalkyl, C ⁇ -C 4.
- the ready-to-use composition for bleaching in accordance with the invention, can also contain various adjuvants conventionally used in compositions for bleaching the hair, such as anionic, cationic, nonionic, amphoteric surfactants, zwitterionics or their mixtures, anionic, cationic, nonionic, amphoteric, zwitterionic polymers or their mixtures, mineral or organic thickeners, antioxidants, enzymes other than the 4-electron oxidoreductases used in accordance with the invention such as for example peroxidases and / or oxidoreductases with two electrons with their possible cofactors, penetration agents, agents sequestering agents, perfumes, buffers, dispersing agents, conditioning agents such as, for example, volatile or non-volatile silicones, modified or unmodified, film-forming agents, ceramides, preserving agents, opacifying agents.
- adjuvants conventionally used in compositions for bleaching the hair
- the ready-to-use composition for bleaching in accordance with the invention, can be in various forms, such as in the form of liquids, creams, gels, optionally pressurized, or in any other form suitable for producing a discoloration of human keratin fibers, and in particular of the hair.
- the composition In the case where the composition is stored as it is before use, it must be free of gaseous oxygen, so as to avoid any premature degradation of the mediator (s).
- At least one composition ready to use for bleaching as defined above is applied to the fibers, at an application temperature between room temperature and 80 ° C., for a time sufficient to partially or completely degrade the direct dyeing of human keratin fibers.
- the fibers are then rinsed, or optionally washed with shampoo, then dried.
- the application temperature is preferably between room temperature and 60 ° C and even more preferably between 35 ° C and 50 ° C.
- the time sufficient for the development of discoloration of human keratin fibers is generally between 1 and 60 minutes and even more precisely between 5 and 30 minutes.
- the method comprises a preliminary step consisting in storing in separate form, on the one hand, a composition (A) comprising, in a medium suitable for discoloration, at least one mediator as defined previously, and on the other hand, a composition (B) containing, in a medium suitable for discoloration, at least one enzyme of the 4-electron oxidoreductase type, then mixing them at the time of use before d apply this mixture to the keratin fibers.
- a composition (A) comprising, in a medium suitable for discoloration, at least one mediator as defined previously
- a composition (B) containing, in a medium suitable for discoloration, at least one enzyme of the 4-electron oxidoreductase type then mixing them at the time of use before d apply this mixture to the keratin fibers.
- Another object of the invention is a device with several compartments or "kit” for bleaching according to the invention or any other packaging system with several compartments of which at least one first compartment contains the composition (A) as defined above. above and at least one second compartment contains the composition (B) as defined above.
- These devices can be equipped with a means enabling the desired mixture to be delivered to the hair, such as the devices described in patent FR-2,586,913 in the name of the applicant.
- the ready-to-use composition was prepared for the following bleaching (contents in grams):
- composition ready for use for bleaching described above was applied for 30 minutes at a temperature of 30 ° C to locks of natural gray hair containing 90% white hairs previously dyed with a 0.5% solution of 2-nitro-paraphenylenediamine in a mixture of Water (90) and ethanol (10). The hair was then rinsed, washed with a standard shampoo, and then dried. The initial red shade then weakened considerably.
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Epidemiology (AREA)
- Birds (AREA)
- Microbiology (AREA)
- Mycology (AREA)
- Engineering & Computer Science (AREA)
- Biotechnology (AREA)
- Botany (AREA)
- Natural Medicines & Medicinal Plants (AREA)
- Cosmetics (AREA)
Abstract
Description
Claims
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AU2001270714A AU2001270714A1 (en) | 2000-07-21 | 2001-06-29 | Enzyme composition for bleaching human keratinous fibres and bleaching method |
| EP01949588A EP1304996A1 (fr) | 2000-07-21 | 2001-06-29 | Composition a base d'enzymes pour la decoloration des fibres keratiniques humaines et procede de decoloration |
| US10/333,327 US20040049859A1 (en) | 2000-07-21 | 2001-06-29 | Enzyme composition for bleaching human keratinous fibres and bleaching method |
| JP2002513425A JP2004521858A (ja) | 2000-07-21 | 2001-06-29 | ヒトのケラチン繊維を脱色するための酵素組成物及び脱色方法 |
| BR0113003-0A BR0113003A (pt) | 2000-07-21 | 2001-06-29 | Composição pronta para o uso, processo de descoloração das fibras queratìnicas humanas previamente tingida por pelo menos um colorante direto, e dispositivo com vários compartimentos |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| FR00/09620 | 2000-07-21 | ||
| FR0009620A FR2811888A1 (fr) | 2000-07-21 | 2000-07-21 | Composition a base d'enzymes pour la decoloration des fibres keratiniques et procede de decoloration |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2002007687A1 true WO2002007687A1 (fr) | 2002-01-31 |
Family
ID=8852804
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/FR2001/002091 Ceased WO2002007687A1 (fr) | 2000-07-21 | 2001-06-29 | Composition a base d'enzymes pour la decoloration des fibres keratiniques humaines et procede de decoloration |
Country Status (8)
| Country | Link |
|---|---|
| US (1) | US20040049859A1 (fr) |
| EP (1) | EP1304996A1 (fr) |
| JP (1) | JP2004521858A (fr) |
| CN (1) | CN1443060A (fr) |
| AU (1) | AU2001270714A1 (fr) |
| BR (1) | BR0113003A (fr) |
| FR (1) | FR2811888A1 (fr) |
| WO (1) | WO2002007687A1 (fr) |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8009556B2 (en) | 2003-10-17 | 2011-08-30 | Ip Infusion, Inc. | System and method for providing redundant routing capabilities for a network node |
| DE102004062431A1 (de) * | 2004-12-20 | 2006-06-29 | Henkel Kgaa | Haarbehandlungs-Kit mit Färbe- und Entfärbemittel |
| CN108078849A (zh) * | 2017-12-30 | 2018-05-29 | 广州润虹医药科技股份有限公司 | 一种含有壳聚糖的漱口液及其制备方法 |
| US12458576B2 (en) | 2022-09-29 | 2025-11-04 | L'oreal | Method for removing color from artificially colored hair |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB2304107A (en) * | 1995-08-08 | 1997-03-12 | Ciba Geigy Ag | Enzyme activity enhancement by addition of an (hetero)aromatic compound |
| WO1997041215A1 (fr) * | 1996-04-29 | 1997-11-06 | Novo Nordisk A/S | Compositions liquides, non aqueuses et a base d'enzyme |
| WO1998040471A1 (fr) * | 1997-03-12 | 1998-09-17 | Novo Nordisk A/S | Formulation liquide stable en stockage comprenant une laccase |
| US5899212A (en) * | 1998-05-07 | 1999-05-04 | Novo Nordisk A/S | Re-formation of keratinous fibre cross links |
| EP1062938A1 (fr) * | 1999-06-01 | 2000-12-27 | L'oreal | Composition de teinture d'oxydation des fibres kératiniques et procédé de teinture |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| ES2215944T3 (es) * | 1994-11-03 | 2004-10-16 | Ciba Specialty Chemicals Holding Inc. | Colorantes imidazolazoicos cationicos. |
| US6540791B1 (en) * | 2000-03-27 | 2003-04-01 | The Procter & Gamble Company | Stable alkaline hair bleaching compositions and method for use thereof |
-
2000
- 2000-07-21 FR FR0009620A patent/FR2811888A1/fr not_active Withdrawn
-
2001
- 2001-06-29 JP JP2002513425A patent/JP2004521858A/ja not_active Withdrawn
- 2001-06-29 US US10/333,327 patent/US20040049859A1/en not_active Abandoned
- 2001-06-29 EP EP01949588A patent/EP1304996A1/fr not_active Withdrawn
- 2001-06-29 AU AU2001270714A patent/AU2001270714A1/en not_active Abandoned
- 2001-06-29 WO PCT/FR2001/002091 patent/WO2002007687A1/fr not_active Ceased
- 2001-06-29 CN CN01813070A patent/CN1443060A/zh active Pending
- 2001-06-29 BR BR0113003-0A patent/BR0113003A/pt not_active IP Right Cessation
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB2304107A (en) * | 1995-08-08 | 1997-03-12 | Ciba Geigy Ag | Enzyme activity enhancement by addition of an (hetero)aromatic compound |
| WO1997041215A1 (fr) * | 1996-04-29 | 1997-11-06 | Novo Nordisk A/S | Compositions liquides, non aqueuses et a base d'enzyme |
| WO1998040471A1 (fr) * | 1997-03-12 | 1998-09-17 | Novo Nordisk A/S | Formulation liquide stable en stockage comprenant une laccase |
| US5899212A (en) * | 1998-05-07 | 1999-05-04 | Novo Nordisk A/S | Re-formation of keratinous fibre cross links |
| EP1062938A1 (fr) * | 1999-06-01 | 2000-12-27 | L'oreal | Composition de teinture d'oxydation des fibres kératiniques et procédé de teinture |
Also Published As
| Publication number | Publication date |
|---|---|
| EP1304996A1 (fr) | 2003-05-02 |
| FR2811888A1 (fr) | 2002-01-25 |
| JP2004521858A (ja) | 2004-07-22 |
| US20040049859A1 (en) | 2004-03-18 |
| CN1443060A (zh) | 2003-09-17 |
| BR0113003A (pt) | 2003-06-24 |
| AU2001270714A1 (en) | 2002-02-05 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO1999036037A1 (fr) | Composition de teinture d'oxydation des fibres keratiniques contenant une laccase et procede de teinture mettant en oeuvre cette composition | |
| EP1043012A2 (fr) | Procédé de teinture d'oxydation utilisant un cétose à titre d'agent réducteur et une laccase à titre d'agent oxydant | |
| EP1138318A2 (fr) | Composition de teinture d'oxydation des fibres kératiniques et procédé de teinture mettant en oeuvre cette composition | |
| EP1062937A1 (fr) | Composition de teinture d'oxydation des fibres kératiniques et procédé de teinture mettant en oeuvre cette composition | |
| CA2347560A1 (fr) | Composition oxydante et utilisations pour la teinture, pour la deformation permanente ou pour la decoloration des fibres keratiniques | |
| CA2319852A1 (fr) | Composition de teinture d'oxydation des fibres keratiniques procede de teinture mettant en oeuvre cette composition | |
| EP1047376A1 (fr) | Composition de teinture d'oxydation des fibres keratiniques contenant une laccase et procede de teinture mettant en oeuvre cette composition | |
| EP1304996A1 (fr) | Composition a base d'enzymes pour la decoloration des fibres keratiniques humaines et procede de decoloration | |
| EP1047383A1 (fr) | Composition de teinture d'oxydation des fibres keratiniques contenant une laccase et procede de teinture mettant en oeuvre cette composition | |
| WO2000057848A1 (fr) | Procede de teinture d'oxydation utilisant la n-acetylcysteine a titre d'agent reducteur et une laccase a titre d'agent oxydant | |
| EP1304997A1 (fr) | Composition a base d'enzymes pour la decoloration des fibres keratiniques humaines et procede de decoloration | |
| WO1999017732A1 (fr) | Composition de teinture d'oxydation des fibres keratiniques et procede de teinture mettant en oeuvre cette composition | |
| EP1062938A1 (fr) | Composition de teinture d'oxydation des fibres kératiniques et procédé de teinture | |
| WO2002007689A1 (fr) | Composition a base d'enzymes pour la decoloration des fibres keratiniques humaines et procede de decoloration | |
| EP1047382A1 (fr) | Composition de teinture d'oxydation des fibres keratiniques contenant une laccase et procede de teinture mettant en oeuvre cette composition | |
| WO1999036038A1 (fr) | Composition de teinture d'oxydation des fibres keratiniques contenant une laccase et procede de teinture mettant en oeuvre cette composition | |
| WO1999036042A1 (fr) | Composition de teinture d'oxydation des fibres keratiniques contenant une laccase et procede de teinture mettant en oeuvre cette composition | |
| EP1432392A2 (fr) | Composition tinctoriale comprenant une base d'oxydation du type diaminopyrazole et un agent oxydant de nature enzymatique |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NO NZ PL PT RO RU SD SE SG SI SK SL TJ TM TR TT TZ UA UG US UZ VN YU ZA ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): GH GM KE LS MW MZ SD SL SZ TZ UG ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE TR BF BJ CF CG CI CM GA GN GW ML MR NE SN TD TG |
|
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2001949588 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 018130704 Country of ref document: CN |
|
| WWP | Wipo information: published in national office |
Ref document number: 2001949588 Country of ref document: EP |
|
| REG | Reference to national code |
Ref country code: DE Ref legal event code: 8642 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 10333327 Country of ref document: US |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: 2001949588 Country of ref document: EP |