WO2002072072A2 - Granules et granules enrobes au gout masque - Google Patents
Granules et granules enrobes au gout masque Download PDFInfo
- Publication number
- WO2002072072A2 WO2002072072A2 PCT/FR2002/000836 FR0200836W WO02072072A2 WO 2002072072 A2 WO2002072072 A2 WO 2002072072A2 FR 0200836 W FR0200836 W FR 0200836W WO 02072072 A2 WO02072072 A2 WO 02072072A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- granules
- coating
- coated
- waxy
- eudragit
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0087—Galenical forms not covered by A61K9/02 - A61K9/7023
- A61K9/0095—Drinks; Beverages; Syrups; Compositions for reconstitution thereof, e.g. powders or tablets to be dispersed in a glass of water; Veterinary drenches
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/14—Particulate form, e.g. powders, Processes for size reducing of pure drugs or the resulting products, Pure drug nanoparticles
- A61K9/16—Agglomerates; Granulates; Microbeadlets ; Microspheres; Pellets; Solid products obtained by spray drying, spray freeze drying, spray congealing,(multiple) emulsion solvent evaporation or extraction
- A61K9/1605—Excipients; Inactive ingredients
- A61K9/1617—Organic compounds, e.g. phospholipids, fats
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/48—Preparations in capsules, e.g. of gelatin, of chocolate
- A61K9/50—Microcapsules having a gas, liquid or semi-solid filling; Solid microparticles or pellets surrounded by a distinct coating layer, e.g. coated microspheres, coated drug crystals
- A61K9/5073—Microcapsules having a gas, liquid or semi-solid filling; Solid microparticles or pellets surrounded by a distinct coating layer, e.g. coated microspheres, coated drug crystals having two or more different coatings optionally including drug-containing subcoatings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/04—Antibacterial agents
Definitions
- the present invention relates to granules and coated granules. It also relates to dosage forms incorporating said granules or coated granules.
- solid oral forms such as tablets can be dangerous, especially for children and the elderly, who then prefer chewable tablets, tablets that melt in the mouth or in a spoon of water, granules, powders, solutions or suspensions.
- a number of active ingredients have an unpleasant taste, so it is essential to mask their taste.
- taste masking any process making it possible to delay or prevent the appearance of an unpleasant taste specific to a product during its administration by oral or buccal or even nasal route.
- compositions conditioned in the form of multidose vials and in particular in the case of dry suspensions intended for an extemporaneous reconstitution, also called dry reconstitutable suspensions, the absence of bitterness must be maintained for an equivalent time, either for the duration of the treatment, or for the duration of use of the bottle.
- the granule or coated active granule used in such formulations must therefore be stable in contact with an aqueous liquid phase for a time at least equal to 24 hours. In practice, this amounts to preventing the solubilization of the active principle in the liquid phase.
- the masking of the taste is carried out by encapsulation of the active principle inside a capsule or by microencapsulation techniques in which a polymeric coating is applied to the active principle (WO 92/11871).
- One of the solutions proposed consists in coating the particles of active principle with a cellulosic polymer.
- these polymers mention may in particular be made of ethylcellulose and hydroxypropylmethylcellulose.
- Another solution consists in coating the particle of active principle with an acrylic type polymer.
- acrylic type polymer a distinction is made between pH-dependent polymers, that is to say polymers whose solubility depends on pH and insoluble polymers whose intrinsic properties are not influenced by the pH of the medium.
- pH-dependent polymers that is to say polymers whose solubility depends on pH and insoluble polymers whose intrinsic properties are not influenced by the pH of the medium.
- these polymers interfere with the release of the active ingredient and require the use of agents promoting or delaying the solubilization of the active ingredient (GB 1 511 852; WO 91/16043).
- Matrix microspheres have also been stabilized, but they require additional coating to achieve the desired stability; correct stability can be obtained at acid pH with cellulose acetates, but there is a delay in release (EP 0 293 885).
- a granule or a granule comprising on the one hand a core containing an active principle possibly associated with at least one waxy compound and at least one polymer and on the other hand at least three layers coating, of which the second contains at least one waxy compound, makes it possible to isolate the active principle for a sufficient time to ensure the stability of the masking of the taste when the dry suspension incorporating said granule or coated granule is reconstituted by adding a volume of water defined at the time of the first intake.
- an immediate release, or a modified release, that is to say delayed or prolonged, of the active principle is an object of the present invention to solve the problems, or at least to improve the solutions implemented in the prior art to overcome the difficulties in developing this type of formulation.
- a subject of the present invention is therefore granules and coated granules characterized in that they comprise: a core containing at least one active principle possibly associated with at least one waxy compound and optionally with at least one polymer and / or at least a binding agent, and at least three successive coating layers starting from the core: • a functional polymeric coating (1) possibly containing a waxy compound, allowing immediate, delayed or prolonged release,
- the term immediate release means a release, the kinetics of which are not substantially modified by the formulation and / or by the parameters of the manufacturing process, which means that the dissolution profile of the active principle depends essentially of its intrinsic properties.
- modified release means a release whose kinetics is substantially modified by the formulation and / or by the parameters of the manufacturing process.
- the expression “complementary release” is understood to mean a release of the same nature as that obtained with the coating (1).
- conditioning the suspension medium means that the characteristics of the reconstituted suspension, obtained from the excipient grains, are chosen as a function of the release profile of the granule or active granule coated, in vi tro or after administration of said reconstituted suspension.
- additional layers can be applied, the composition of which is identical to that of layers (1) and (3).
- An overcoat intended to mask any bitterness linked to the components of the third coating layer (3), which does not substantially modify the release properties of the granules and of the granules, can be applied.
- the core is a neutral, preferentially spherical substrate of determined particle size, based on starch, sucrose, ethylcellulose, lactose or wax, to which the active principle is applied in layers. by spraying a suspension or a solution of said active principle, in an aqueous, organic solvent or in a mixture in the presence of at least one binding agent or at least one polymer or at least one waxy compound or of a mixture of at least two of these agents and optionally of lubricants.
- the core is the active principle itself, in the form of a spherical crystal or not, if its particle size enables an effective coating to be carried out directly.
- a layer application (mounting) of the active principle will be carried out by spraying a solution or a suspension of said active principle in the presence of at least one binding agent or at least one polymer or at least one compound. waxy or a mixture of at least two of these agents and optionally lubricants and organic solvents or water.
- the core is a granule based on active principle obtained by granulation.
- the granule can be obtained by wet granulation or in a fluidized air bed, or by spherical crystallization or by emulsion-diffusion of solvent preferably using (a) granulation solutions based on organic solutions of waxy compound (s) in the presence lubricants and plasticizers or (b) a polymer such as hydroxypropyl methylcellulose.
- an assembly of the active principle may be carried out using said granule as a support, by spraying a solution or suspension of active principle in organic solvents or in water, in the presence of at least one binder or 'at least one polymer or at least one waxy compound or a mixture of at least two of these agents and optionally lubricants.
- the heart can contain various agents; among these agents, there are insoluble agents, in particular talc, silicone dioxide, titanium dioxide, silica, alumina, starch and their mixtures; there are also soluble agents, in particular mannitol, sucrose, lactose, dextrose, sodium chloride, sorbitol and their mixtures, polyethylene glycol or amphiphilic compounds (magnesium stearate, polysorbates).
- insoluble agents in particular talc, silicone dioxide, titanium dioxide, silica, alumina, starch and their mixtures
- soluble agents in particular mannitol, sucrose, lactose, dextrose, sodium chloride, sorbitol and their mixtures, polyethylene glycol or amphiphilic compounds (magnesium stearate, polysorbates).
- the heart can contain up to 100% of active principle, preferably between 30 and 85% depending on the dosage of the final formulation and the proportion of dry mass in suspension to have a homogeneous suspension.
- the core containing the active ingredient can have any suitable size, but preferably the size distribution of the core containing the active ingredient has an average of between 100 to 500 ⁇ m, the average preferably being between 100 to 250 ⁇ m when the core is a granule or the active principle itself, and preferably between 400 and 500 ⁇ m when the core is a neutral support on which the active principle is applied in a layer.
- antacids As active principles, one can use in particular and without being limited to this list: antacids, anti-inflammatories, coronary or peripheral vasodilators, anti-infectives, antibiotics, antiparasitics, anxiolytics, psychotropics , neuroleptics, stimulants of the central nervous system, antihistamines, anti-diarrheal, nutritional supplements, antivirals, anti-spasmodics, vasoconstrictors, anti-thrombotics, antimigraine, analgesics, anti- pyretics, anti-asthmatics, cough suppressants, mucoregulators, decongestants, plant extracts and anti-nausea drugs.
- the active principle is an anti-infectious substance, chosen from macrolides.
- erythromycin and its derivatives mention may in particular be made of erythromycin and its derivatives, and clarithromycin.
- the coatings (1) and (3) are functional coatings, which aim to confer a property of release of the active principle, either immediate, or prolonged, or deferred; they consist of polymers conventionally known to those skilled in the art to confer said properties possibly associated with a waxy compound.
- polymers such as delayed release: polymethacrylates particular those marketed under the name Eudragit ® L, Eudragit ® S and Eudragit ® FS30D, 1 'cellulose acetate phthalate and cellulose acetate; as sustained release polymers: polymethacrylates particular those marketed under the name Eudragit ® NE, Eudragit ® RS and Eudragit ® RL, 1 ethyl cellulose, polyvinyl acetate, polyvinyl alcohol and copolymers thereof; and as immediate-release polymers: polymethacrylates, in particular those marketed under the name Eudragit E.
- delayed release polymethacrylates particular those marketed under the name Eudragit ® L, Eudragit ® S and Eudragit ® FS30D, 1 'cellulose acetate phthalate and cellulose acetate
- sustained release polymers polymethacrylates particular those marketed under the name Eudragit ® NE, Eudragit ® RS and Eudragit ®
- the waxy compounds used can be chosen in particular from the group consisting of: waxes, Novata waxes, gelucires and suppocires, glyceric macrogol, fatty acids (stearic acid), fatty acid esters, glycerol monostearate Precirol ® , Compritol ® .
- hydrophobic waxy compounds are advantageously used and even more advantageously hydrophobic waxy compounds having a low HLB (hydrophilic-lipophilic balance) and having a melting point of between 35 and 53 ° C., preferably between 37 and 43 °. vs.
- HLB hydrophilic-lipophilic balance
- waxy compounds sold under the names Gelucire ® 43/01 and Novata ® AB include, without implied limitation, waxy compounds sold under the names Gelucire ® 43/01 and Novata ® AB.
- GMS glycerol monostearate
- a coating consisting of a mixture of Eudragit ® E100 and optionally hydrophobic waxy compounds with a low HLB (hydrophilic-lipophilic balance) and having a melting point of between 35 and 53 ° C, preferably between 37 and 43 ° C in the presence of lubricants.
- HLB hydrophilic-lipophilic balance
- lubricants include, without limitation, Gelucire ® 43/01 and Novata ® AB, possibly associated with glycerol monostearate (GMS).
- functional coatings (1) and (3) may be based on an aqueous dispersion or an organic solution of ethylcellulose or Eudragit ® RL or RS or coating based on an organic solution of these polymers or Eudragit ® S in the presence or not of waxy compounds and / or lubricating agents, plasticizers and lubricants.
- the coating rate for coating (1) (calculated as a percentage (w / w) of dry matter applied to the starting substrate) is advantageously between 5 and 100% and preferably between 30 and 60%.
- the purpose of the hydrophobic coating (2) is to increase the stability of the grain in suspension. It is based on a solution of waxy compounds in a solvent and optionally includes a lubricating agent such as, for example, talc, hydrophobic colloidal silica or glycerol monostearate (GMS).
- a lubricating agent such as, for example, talc, hydrophobic colloidal silica or glycerol monostearate (GMS).
- the coating rate for this second coating (calculated as a percentage (w / w) of dry matter applied to the starting substrate) is advantageously between 5 and 100% and preferably between 20 and 80%.
- this hydrophobic coating (2) advantageously comprises a waxy compound or a combination of hydrophobic waxy compounds with low HLB and having a melting point of between 35 and 53 ° C, preferably 37 and 43 ° C in a solvent.
- these include including Gelucire 43/01 ®, Gelucire ® 53/01, Novata ® AB, glycerol monostearate and mixtures thereof.
- the functional polymeric coating (3) which has release functions complementary to those of the coating (1) is either identical to or analogous to said coating (1), but has the same properties with respect to the release of the active principle. and conditions the suspension medium.
- the coating rate at this coating (3) (calculated as a percentage (w / w) of material dry applied to the starting substrate) is advantageously between 5 and 200% and preferably between 80 and 160%.
- the coating (3) has a pronounced taste due to the excipients it comprises then an outer coating based Eudragits ® RL30D and RS30D or their mixtures, in the presence of plasticizers and lubricants is applied .
- the coating rate at this level will advantageously be between 0 and 15% and preferably between 0 and 5%.
- the lubrication agents are advantageously chosen from the group comprising talc, hydrophobic colloidal silica and glycerol monostearate.
- Plasticizers are advantageously selected from the group consisting of dibutylsébaccate, triethylcitrate, diethylphthalate, 1 'acétyltriéthyl- citrate, acetyl tributyl citrate, glyceryl monostearate (GMS) and Myvacet ®.
- the granules and the coated granules of the invention are prepared according to a process which comprises the production of the core or support and optionally includes an additional mounting step.
- the process can advantageously comprise the following stages: application of the active principle dissolved on the support, in the presence of preferably hydrophobic waxy compounds and / or polymers, and of at least one lubricating agent in a solvent or a mixture of solvents, application a first coating, functional polymeric coating (1) and optionally waxy compounds, said coating allowing immediate, delayed or prolonged release, application of a second coating, hydrophobic coating (2) containing at least one waxy compound or a combination of waxy compounds, application of a third coating, functional polymeric coating ( 3) and optionally of waxy compounds, said coating possibly having a structure different from that of the coating (1), but having a complementary release function, and optionally drying the granules or granules thus obtained.
- the coating solvents are those conventionally used by a person skilled in the art. Examples which may be mentioned are water, methylene chloride, ethanol, isopropanol and their mixtures.
- This process is carried out in a fluidized air bed or by any other similar industrial process known to those skilled in the art.
- the drying operation can be carried out in a fluidized air bed, in a rotary vacuum dryer or by any equivalent technique allowing the removal of residual solvents.
- the method further comprises the application of additional layers identical to the layers (1) and
- the granules and the granules coated according to the invention can be used in any suitable pharmaceutical formulation allowing a immediate reconstitution in liquid medium. They can in particular be used to prepare dry syrups, tablets, sachets and suspensions. Among the latter, advantageously the reconstitutable dry suspensions will be chosen, that is to say powders packaged in multidose vials which can be reconstituted before use as a suspension in a liquid such as water.
- the reconstitutable powders prepared from the granules and granules according to the invention are stable on storage and the suspensions, once reconstituted in the multidose vial, have a masked taste throughout the duration of the treatment or, in the case where the treatment requires several vials, during the entire time of use of the vial. In all cases, the reconstituted suspension is stable for at least 24 hours. These suspensions also have sufficient bioavailability and are particularly useful in pediatrics and geriatrics.
- the invention also relates to a reconstitutable dry suspension containing granules or granules according to the invention.
- This reconstitutable dry suspension also contains excipients giving the reconstituted formulation, particular organoleptic characteristics, and on the other hand, microbiological stability.
- excipients are chosen from those conventionally used by a person skilled in the art for producing these formulations. Mention may be made, among these excipients, of sweeteners, colorants, agents which confer viscosity or thickeners, pH modulating agents, preserving agents (antimicrobials or fungicides), surfactants, antioxidants.
- This suspension can be obtained in several ways: - by simple addition to the active grain of the excipients in the form of powder mixture, by addition to the active grain of a dry granule of excipients.
- the excipients are granules preferably obtained by wet granulation; by adding to the active grain excipients mounted on the active grain by a coating process advantageously carried out in a fluidized air bed.
- the invention also also relates to a dry mixture comprising granules or granules according to the present invention associated with any excipient suitable for having a dry suspension reconstitutable in liquid medium among which at least one is a thickening agent, one is a preservative and one is a pH modulating agent.
- thickener mention may be made of all the thickeners known to those skilled in the art, in particular those chosen from the group comprising gums such as xanthan, guar and tragacanth, magnesium silicate and their combinations, sodium alginate, propylene glycol alginate, cellulosic compounds such as hydroxyethyl cellulose, 1 hydroxypropyl cellulose, methyl cellulose, carboxymethyl cellulose, carbomers, gelatin, poloxamers, or combinations of these compounds and carrageenans.
- pH adjusting agent there may advantageously be mentioned those chosen from the group comprising citric acid, soda, sodium citrate, trisodium citrate or any pharmaceutically acceptable compound having the capacity to buffer an aqueous solution.
- a preservative mention may be made of those chosen from the group comprising potassium or sodium sorbate, sodium benzoate, azorubine, bronopol, ethylenediaminetetraacetic acid (EDTA), p -hydroxybenzoate (parabens) of methyl, ethyl, propyl and butyl as well as their salts, used alone or as a mixture, propionic acid, sulfites and cresol.
- potassium or sodium sorbate sodium benzoate
- azorubine azorubine
- bronopol ethylenediaminetetraacetic acid (EDTA)
- EDTA ethylenediaminetetraacetic acid
- parabens parabens of methyl, ethyl, propyl and butyl as well as their salts, used alone or as a mixture, propionic acid, sulfites and cresol.
- the suspension may also contain one or more sweetener (s) such as the saccharin salts and / or potassium acesulfame, or any other sweetener known to a person skilled in the art such as aspartame, sucrose and its derivatives, trehalose, sodium glycyrrhizinate or mixtures thereof, an opacifying agent such as Opadry ® OYB or titanium oxides and product capture agents such as cyclodextrins which amounts will vary depending on the size of the molecule and function to isolate.
- the suspension can also contain one or more aromatic composition (s) and a filler, in particular polyols, for example sorbitol (Neosorb ®), xylitol and lactitol.
- the grain of excipient can be obtained by a wet granulation process or any other similar industrial process known to those skilled in the art. It can in particular be obtained by making a hydro-alkanolic solution of sweeteners and / or preservatives which will serve as wetting liquid for a mixture of bulking agent such as sorbitol, thickening agent, opacifying agent, d pH adjusting agent, optionally aromatic compositions, the bulking agent having the function of creating a sufficient mass for granulation. Any other excipient fulfilling the same function may also be used. Another alternative consists in mounting the excipients on the active grain by any technique known to a person skilled in the art, in particular in a fluidized air bed.
- the suspension is prepared by adding a defined quantity of water (for example by volume, or by a dipstick on the bottle), directly in the bottle containing the final dry mixture.
- a defined quantity of water for example by volume, or by a dipstick on the bottle
- the excipient grains thus prepared allow rapid reconstitution of the suspension, which requires only manual stirring by inversion to homogenize the preparation; in addition, the suspension obtained has good bacteriological stability and masking stability greater than 7 days and independent of the pH of the suspension. It is particularly useful in pediatrics and geriatrics.
- the pH of the suspension is adjusted according to the properties of the granule or of the coated granule to be combined. In the case where immediate release is desired, the pH of the suspension will be between 5.5 and 10, preferably between 8.5 and 10. In the case of delayed release, the pH of the suspension will be between 3 and 7, preferably between 4 and 5.
- Waxy agents Thanks to the presence of waxy agents, the masking stability of the suspensions is improved. Waxy agents also make it possible to reduce the quantity of polymers used for coating, therefore the toxicity induced by said polymers.
- Step 0 a mixture of powders was produced and placed in the tank of the fluidized air bed:
- the dry concentration in methylene chloride - ethanol is equal to 11.9% by weight and the dry pulverized / substrate ratio is equal to 100% by weight.
- a solution based on Eudragit ® E 100 - Gélucire ® 43/01 - Talc M (10/1) (51.4% -5.7% -42.9%) in a methylene chloride / water mixture (10 / 1) is sprayed onto the granule obtained in step lb.
- the dry concentration in methylene chloride is equal to 12.9% by weight and the dry pulverized / substrate ratio is equal to 52.5% by weight.
- a solution based on Gélucire ® 43/01 - Talc M 10 (57.1% -42.9%) in methylene chloride is sprayed onto the granule obtained in step 2.
- the dry concentration in chloride of methylene is 18.2% and the dry spray / substrate ratio is 35% by weight.
- coating (3) functional polymeric coating
- a solution based on Eudragit ® E 100 - Gélucire ® 43/01 - Talc M (10/1) (45.7% -ll, 4% -42.9 %) in a mixture of methylene chloride / water (10/1) is sprayed onto the granule obtained in step 3.
- the dry concentration in the methylene chloride / water mixture is equal to 12.9% by weight and the dry pulverized / substrate ratio is equal to 105% by weight.
- the vial is filled according to the dose of CHL 13.05 taken for treatment. At the time of use, top up with mineral water. The reconstituted suspension is stable for at least 7 days.
- Step 1 the grain constitution steps are similar to those of the previous example.
- the dry concentration in total water is equal to 32.6% by weight and the dry sprayed / substrate ratio is equal to 39% by weight.
- the dry concentration in methylene chloride is equal to 19.4% by weight and the dry pulverized / substrate ratio is equal to 35% by weight.
- a solution based on Eudragit ® L30D (dry extract) Myvacet 9.45 - Talc M 10 (71.4% -10.7% -17.9%) diluted in purified water is sprayed onto the granule obtained at step 3.
- the concentration of dry matter in total water is equal to 34.5% by weight and the dry sprayed / substrate ratio is equal to 154% by weight.
- a solution based on Eudragit ® S100 - Myvacet 9.45 - Talc M 10 (83.3% -8.3% -8.3%) in ethanol is sprayed onto the granule obtained in step 4.
- the dry concentration in ethanol is equal to 9.8% by weight and the dry pulverized / substrate ratio is equal to 0.6% by weight.
- Distribution and reconstitution of the suspension The mixture consists of 75% of excipient grains and 25% of active grains and then treated as for the previous example.
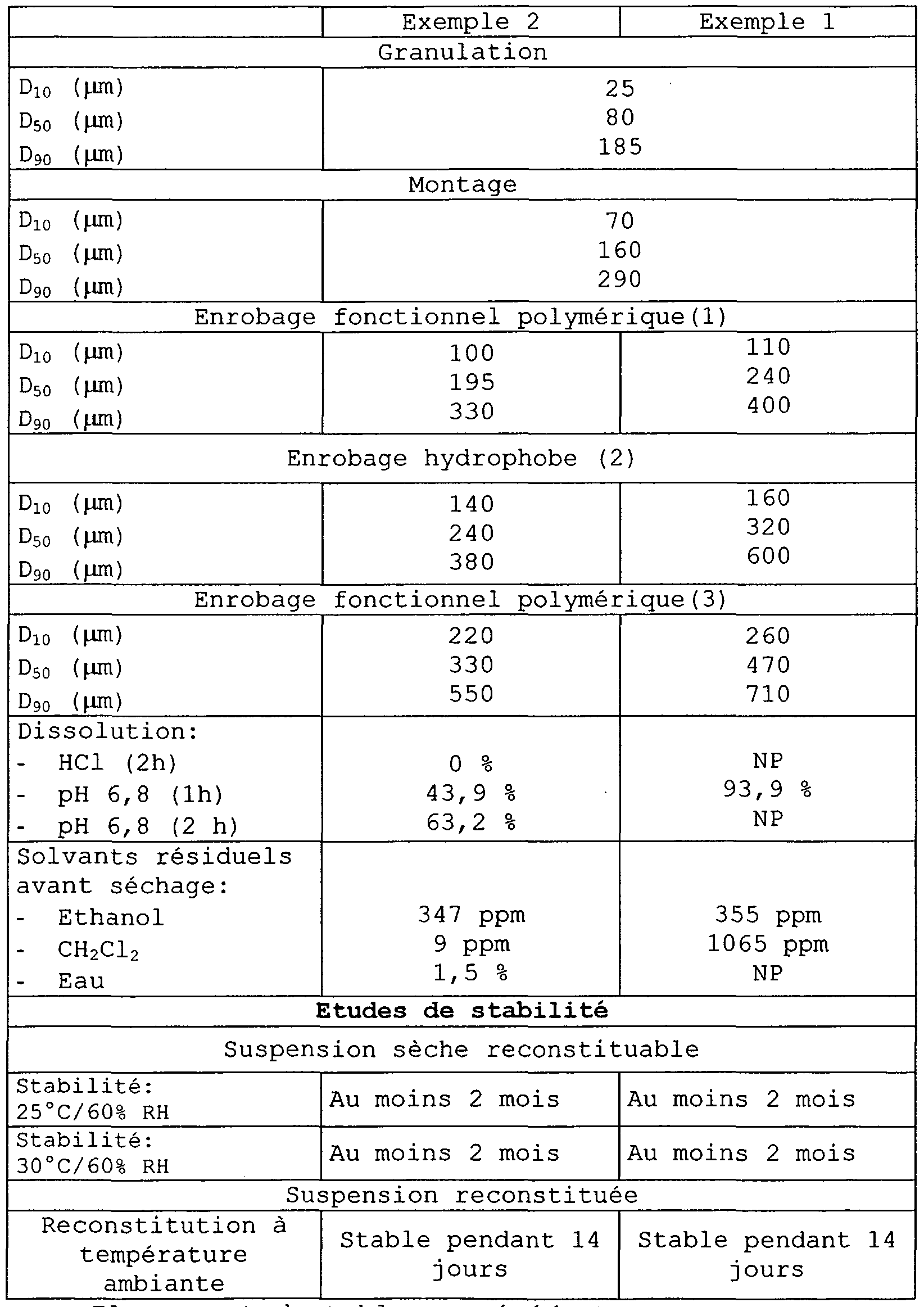
- the stability of the granules prepared according to the methods of Examples 1 and 2 is evaluated in terms of degradation products, kinetics of dissolution, taste and residual solvents.
- the stability of the suspensions obtained from the granules prepared according to the methods of Examples 1 and 2, is evaluated in terms of pH, taste masking and dosage of the released active principle.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Epidemiology (AREA)
- Molecular Biology (AREA)
- Biophysics (AREA)
- Communicable Diseases (AREA)
- Oncology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Medicinal Preparation (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
Claims
Priority Applications (8)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN02809252XA CN1525852B (zh) | 2001-03-09 | 2002-03-08 | 被掩味的包衣颗粒和粒状物 |
| AU2002247806A AU2002247806A1 (en) | 2001-03-09 | 2002-03-08 | Granules and granules coated with a masked taste |
| JP2002571031A JP4465152B2 (ja) | 2001-03-09 | 2002-03-08 | 味をマスキングした、顆粒及びコーティングされた顆粒剤 |
| ES02716883.0T ES2524197T3 (es) | 2001-03-09 | 2002-03-08 | Gránulos y granulados recubiertos con sabor enmascarado |
| US10/471,234 US20040241235A1 (en) | 2001-03-09 | 2002-03-08 | Granules and granules coated with a masked taste |
| EP02716883.0A EP1365751B1 (fr) | 2001-03-09 | 2002-03-08 | Granules et granules enrobes au gout masque |
| HK05101669.3A HK1068059B (zh) | 2001-03-09 | 2002-03-08 | 被掩味的包衣颗粒和粒状物 |
| US13/104,513 US20110212182A1 (en) | 2001-03-09 | 2011-05-10 | Masked taste pharmaceutical granules/granulates |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| FR01/03235 | 2001-03-09 | ||
| FR0103235A FR2821745B1 (fr) | 2001-03-09 | 2001-03-09 | Granules et granules enrobes au gout masque |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2002072072A2 true WO2002072072A2 (fr) | 2002-09-19 |
| WO2002072072A3 WO2002072072A3 (fr) | 2002-12-27 |
Family
ID=8860932
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/FR2002/000836 Ceased WO2002072072A2 (fr) | 2001-03-09 | 2002-03-08 | Granules et granules enrobes au gout masque |
Country Status (8)
| Country | Link |
|---|---|
| US (2) | US20040241235A1 (fr) |
| EP (1) | EP1365751B1 (fr) |
| JP (1) | JP4465152B2 (fr) |
| CN (1) | CN1525852B (fr) |
| AU (1) | AU2002247806A1 (fr) |
| ES (1) | ES2524197T3 (fr) |
| FR (1) | FR2821745B1 (fr) |
| WO (1) | WO2002072072A2 (fr) |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1485071A2 (fr) * | 2002-03-18 | 2004-12-15 | Flamel Technologies | Comprimes a base de microcapsules a liberation modifiee |
| WO2004096175A3 (fr) * | 2003-04-30 | 2005-01-13 | Ranbaxy Lab Ltd | Compositions au gout masque et leurs procedes de preparation |
| WO2006097456A1 (fr) * | 2005-03-16 | 2006-09-21 | Nycomed Gmbh | Forme posologique a gout masque |
| JP2006523620A (ja) * | 2003-02-19 | 2006-10-19 | バイオヴェイル ラボラトリーズ インコーポレイテッド | 急速吸収選択的5−ht作用剤製剤 |
| US9814684B2 (en) | 2002-04-09 | 2017-11-14 | Flamel Ireland Limited | Oral pharmaceutical formulation in the form of aqueous suspension for modified release of active principle(s) |
Families Citing this family (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8101209B2 (en) | 2001-10-09 | 2012-01-24 | Flamel Technologies | Microparticulate oral galenical form for the delayed and controlled release of pharmaceutical active principles |
| MY140561A (en) | 2002-02-20 | 2009-12-31 | Nycomed Gmbh | Dosage form containing pde 4 inhibitor as active ingredient |
| PT1606261E (pt) | 2003-03-10 | 2010-01-11 | Nycomed Gmbh | Novo processo para a preparação de roflumilast |
| US20050107438A1 (en) * | 2003-09-03 | 2005-05-19 | Boehringer Ingelheim International Gmbh | Pharmaceutical composition comprising 3-[(2-{[4-(Hexyloxycarbonylaminoiminomethyl) phenylamino]-methyl}-1-methyl-1H-benzimidazol-5-carbonyl)-pyridin-2-yl-amino]-propionic acid ethyl ester or a salt therefore |
| US20050181497A1 (en) * | 2003-12-04 | 2005-08-18 | Fuji Photo Film Co., Ltd. | Solid substrate used for sensors |
| PT2018153E (pt) | 2006-04-26 | 2012-07-04 | Rosemont Pharmaceuticals Ltd | Composições orais líquidas |
| KR101436516B1 (ko) * | 2007-07-23 | 2014-09-02 | 주식회사태평양제약 | 코팅된 펠렛을 함유하는 현탁정 및 이의 제조방법 |
| RU2011102777A (ru) * | 2008-06-26 | 2012-08-10 | МакНЕЙЛ-ППС, ИНК. (US) | Частицы с покрытием, содержащие фармацевтически активные агенты |
| TWI398274B (zh) * | 2009-12-21 | 2013-06-11 | Tci Co Ltd | Oral tablets |
| CN102805733B (zh) * | 2011-06-01 | 2016-03-09 | 日东电工株式会社 | 颗粒制剂及其制造方法 |
| GR1008992B (el) | 2015-12-17 | 2017-03-23 | Verisfield (Uk) Ltd, Υποκαταστημα Ελλαδας, Εμπορια Φαρμακων | Φαρμακευτικη συνθεση σε μορφη κοκκιων για χορηγηση απο το στομα που περιεχει μετρονιδαζολη ή παραγωγα αυτης και εναν παραγοντα καλυψης της γευσης |
| CN112438979B (zh) * | 2019-09-04 | 2022-05-27 | 普济生物科技(台州)有限公司 | 用于口腔掩味的含氢溴酸沃替西汀的包衣颗粒、固体分散体和制剂 |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2630912A1 (fr) | 1987-05-08 | 1989-11-10 | Orion Yhtymae Oy | Composition permettant d'administrer un medicament sous forme de gelules, d'une poudre ou d'un liquide |
| WO2002072111A1 (fr) | 2001-03-09 | 2002-09-19 | Ethypharm | Suspension de telithromycine a goût masque |
Family Cites Families (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5420572B2 (fr) * | 1973-04-27 | 1979-07-24 | ||
| EP0064485B1 (fr) * | 1981-04-27 | 1985-07-24 | Aktiebolaget Hässle | Préparation pharmaceutique |
| NZ210785A (en) * | 1984-01-13 | 1987-11-27 | Battelle Development Corp | Liquid dispersions of layered controlled release dosage forms |
| US5296236A (en) * | 1988-09-16 | 1994-03-22 | Recordati S.A., Chemical And Pharmaceutical Company | Controlled release therapeutic system for a liquid pharmaceutical formulations |
| JP3247511B2 (ja) * | 1993-09-07 | 2002-01-15 | 山之内製薬株式会社 | 医薬用組成物 |
| EP0758244B2 (fr) * | 1994-05-06 | 2008-02-13 | Pfizer Inc. | Formes galeniques a liberation controlee de l'azithromycine |
| IT1270594B (it) * | 1994-07-07 | 1997-05-07 | Recordati Chem Pharm | Composizione farmaceutica a rilascio controllato di moguisteina in sospensione liquida |
| SE9500422D0 (sv) * | 1995-02-06 | 1995-02-06 | Astra Ab | New oral pharmaceutical dosage forms |
| DE19724177A1 (de) * | 1997-06-09 | 1998-12-10 | Bosch Gmbh Robert | Elastische Lagerung eines hydraulischen Aggregates einer Fahrzeugbremsanlage |
| US6221402B1 (en) * | 1997-11-20 | 2001-04-24 | Pfizer Inc. | Rapidly releasing and taste-masking pharmaceutical dosage form |
| FR2793688B1 (fr) * | 1999-05-21 | 2003-06-13 | Ethypharm Lab Prod Ethiques | Microgranules gastroproteges, procede d'obtention et preparations pharmaceutiques |
| US6368620B2 (en) * | 1999-06-11 | 2002-04-09 | Abbott Laboratories | Formulations comprising lipid-regulating agents |
-
2001
- 2001-03-09 FR FR0103235A patent/FR2821745B1/fr not_active Expired - Lifetime
-
2002
- 2002-03-08 CN CN02809252XA patent/CN1525852B/zh not_active Expired - Lifetime
- 2002-03-08 US US10/471,234 patent/US20040241235A1/en not_active Abandoned
- 2002-03-08 AU AU2002247806A patent/AU2002247806A1/en not_active Abandoned
- 2002-03-08 EP EP02716883.0A patent/EP1365751B1/fr not_active Expired - Lifetime
- 2002-03-08 JP JP2002571031A patent/JP4465152B2/ja not_active Expired - Lifetime
- 2002-03-08 ES ES02716883.0T patent/ES2524197T3/es not_active Expired - Lifetime
- 2002-03-08 WO PCT/FR2002/000836 patent/WO2002072072A2/fr not_active Ceased
-
2011
- 2011-05-10 US US13/104,513 patent/US20110212182A1/en not_active Abandoned
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2630912A1 (fr) | 1987-05-08 | 1989-11-10 | Orion Yhtymae Oy | Composition permettant d'administrer un medicament sous forme de gelules, d'une poudre ou d'un liquide |
| WO2002072111A1 (fr) | 2001-03-09 | 2002-09-19 | Ethypharm | Suspension de telithromycine a goût masque |
Cited By (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1485071A2 (fr) * | 2002-03-18 | 2004-12-15 | Flamel Technologies | Comprimes a base de microcapsules a liberation modifiee |
| US8507003B2 (en) | 2002-03-18 | 2013-08-13 | Flamel Technologies | Compressed tablets comprising microcapsules with modified release |
| US9814684B2 (en) | 2002-04-09 | 2017-11-14 | Flamel Ireland Limited | Oral pharmaceutical formulation in the form of aqueous suspension for modified release of active principle(s) |
| US10004693B2 (en) | 2002-04-09 | 2018-06-26 | Flamel Ireland Limited | Oral pharmaceutical formulation in the form of aqueous suspension for modified release of active principle(s) |
| JP2006523620A (ja) * | 2003-02-19 | 2006-10-19 | バイオヴェイル ラボラトリーズ インコーポレイテッド | 急速吸収選択的5−ht作用剤製剤 |
| WO2004096175A3 (fr) * | 2003-04-30 | 2005-01-13 | Ranbaxy Lab Ltd | Compositions au gout masque et leurs procedes de preparation |
| WO2006097456A1 (fr) * | 2005-03-16 | 2006-09-21 | Nycomed Gmbh | Forme posologique a gout masque |
| EP2258350A3 (fr) * | 2005-03-16 | 2012-08-15 | Nycomed GmbH | Forme de dosage cachant le goût comprenant du roflumilas |
| US8663694B2 (en) | 2005-03-16 | 2014-03-04 | Takeda Gmbh | Taste masked dosage form containing roflumilast |
Also Published As
| Publication number | Publication date |
|---|---|
| JP4465152B2 (ja) | 2010-05-19 |
| EP1365751A2 (fr) | 2003-12-03 |
| CN1525852B (zh) | 2010-04-28 |
| ES2524197T3 (es) | 2014-12-04 |
| AU2002247806A1 (en) | 2002-09-24 |
| US20110212182A1 (en) | 2011-09-01 |
| FR2821745A1 (fr) | 2002-09-13 |
| CN1525852A (zh) | 2004-09-01 |
| JP2004522797A (ja) | 2004-07-29 |
| US20040241235A1 (en) | 2004-12-02 |
| HK1068059A1 (zh) | 2005-04-22 |
| EP1365751B1 (fr) | 2014-08-20 |
| FR2821745B1 (fr) | 2004-07-02 |
| WO2002072072A3 (fr) | 2002-12-27 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1365751B1 (fr) | Granules et granules enrobes au gout masque | |
| EP1194125B1 (fr) | Procede de fabrication de granules enrobes a gout masque et liberation immediate du principe actif | |
| EP1194140B1 (fr) | Composition pharmaceutique contenant du fenofibrate et procede de preparation | |
| EP2349217B1 (fr) | Granulé d'acide gamma-hydroxybutyrique | |
| FR2949061A1 (fr) | Microgranules flottants | |
| CH650674A5 (fr) | Poudre a base de sel d'addition d'acide de bacampicilline micro-encapsule et composition pharmaceutique la contenant. | |
| CZ20002709A3 (cs) | Mikrogranule omeprazolu s vnější vrstvou chránící před žaludečním prostředím, způsob přípravy mikrogranulí | |
| EP0830129B1 (fr) | Compositions solides contenant du polyethyleneoxyde et un principe actif non amorphe | |
| EP1126829B1 (fr) | Particules enrobees d'ibuprofene cristallin granule | |
| EP0216743B1 (fr) | Nouvelles formes galéniques du vérapamil, leur fabrication et médicaments contenant ces nouvelles formes galéniques | |
| EP1646379B1 (fr) | Nouvelle composition pharmaceutique solide comprenant de l amisulpride | |
| EP1401397B1 (fr) | Formulation pharmaceutique au gout masque et son procede de preparation | |
| EP1365775B1 (fr) | Suspension de telithromycine a gout masque | |
| HUT75962A (en) | A ranitidine tablet having a hydroxypropylmethycellulose containing coating and a method for producing said coating | |
| WO2004096175A2 (fr) | Compositions au gout masque et leurs procedes de preparation | |
| FR2791888A1 (fr) | Suspension pharmaceutique buvable | |
| CA2280980A1 (fr) | Composition pharmaceutique pour la liberation programmee de dexfenfluramine | |
| HK1068059B (zh) | 被掩味的包衣颗粒和粒状物 | |
| FR2747573A1 (fr) | Nouvelle composition contenant un benzimidazole acido-labile et son procede de preparation | |
| FR2791889A1 (fr) | Suspension pharmaceutique buvable d'ibuprofene | |
| FR2745181A1 (fr) | Nouvelle composition contenant un benzimidazole acido-labile et son procede de preparation |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A2 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NO NZ OM PH PL PT RO RU SD SE SG SI SK SL TJ TM TN TR TT TZ UA UG US UZ VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A2 Designated state(s): GH GM KE LS MW MZ SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| AK | Designated states |
Kind code of ref document: A3 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NO NZ OM PH PL PT RO RU SD SE SG SI SK SL TJ TM TN TR TT TZ UA UG US UZ VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A3 Designated state(s): GH GM KE LS MW MZ SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2002571031 Country of ref document: JP Ref document number: 01436/DELNP/2003 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2002716883 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 02809252X Country of ref document: CN |
|
| WWP | Wipo information: published in national office |
Ref document number: 2002716883 Country of ref document: EP |
|
| REG | Reference to national code |
Ref country code: DE Ref legal event code: 8642 |
|
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 10471234 Country of ref document: US |