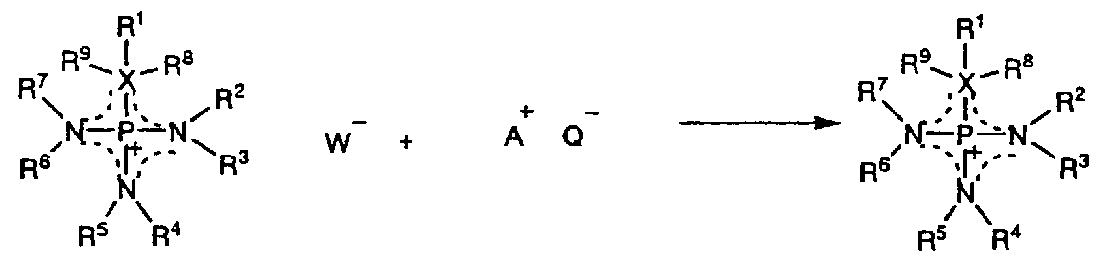WO2006118232A1 - ホスホニウムカチオンを有するイオン液体およびその製造方法 - Google Patents
ホスホニウムカチオンを有するイオン液体およびその製造方法 Download PDFInfo
- Publication number
- WO2006118232A1 WO2006118232A1 PCT/JP2006/308948 JP2006308948W WO2006118232A1 WO 2006118232 A1 WO2006118232 A1 WO 2006118232A1 JP 2006308948 W JP2006308948 W JP 2006308948W WO 2006118232 A1 WO2006118232 A1 WO 2006118232A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- atom
- substituents
- ionic liquid
- partially
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic Table
- C07F9/02—Phosphorus compounds
- C07F9/06—Phosphorus compounds without P—C bonds
- C07F9/22—Amides of acids of phosphorus
- C07F9/24—Esteramides
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/056—Accumulators with non-aqueous electrolyte characterised by the materials used as electrolytes, e.g. mixed inorganic/organic electrolytes
- H01M10/0564—Accumulators with non-aqueous electrolyte characterised by the materials used as electrolytes, e.g. mixed inorganic/organic electrolytes the electrolyte being constituted of organic materials only
- H01M10/0566—Liquid materials
- H01M10/0569—Liquid materials characterised by the solvents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C305/00—Esters of sulfuric acids
- C07C305/02—Esters of sulfuric acids having oxygen atoms of sulfate groups bound to acyclic carbon atoms of a carbon skeleton
- C07C305/04—Esters of sulfuric acids having oxygen atoms of sulfate groups bound to acyclic carbon atoms of a carbon skeleton being acyclic and saturated
- C07C305/06—Hydrogenosulfates
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C311/00—Amides of sulfonic acids, i.e. compounds having singly-bound oxygen atoms of sulfo groups replaced by nitrogen atoms, not being part of nitro or nitroso groups
- C07C311/48—Amides of sulfonic acids, i.e. compounds having singly-bound oxygen atoms of sulfo groups replaced by nitrogen atoms, not being part of nitro or nitroso groups having nitrogen atoms of sulfonamide groups further bound to another hetero atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic Table
- C07F9/02—Phosphorus compounds
- C07F9/06—Phosphorus compounds without P—C bonds
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic Table
- C07F9/02—Phosphorus compounds
- C07F9/28—Phosphorus compounds with one or more P—C bonds
- C07F9/54—Quaternary phosphonium compounds
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic Table
- C07F9/02—Phosphorus compounds
- C07F9/28—Phosphorus compounds with one or more P—C bonds
- C07F9/54—Quaternary phosphonium compounds
- C07F9/5463—Compounds of the type "quasi-phosphonium", e.g. (C)a-P-(Y)b wherein a+b=4, b>=1 and Y=heteroatom, generally N or O
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01G—CAPACITORS; CAPACITORS, RECTIFIERS, DETECTORS, SWITCHING DEVICES, LIGHT-SENSITIVE OR TEMPERATURE-SENSITIVE DEVICES OF THE ELECTROLYTIC TYPE
- H01G9/00—Electrolytic capacitors, rectifiers, detectors, switching devices, light-sensitive or temperature-sensitive devices; Processes of their manufacture
- H01G9/20—Light-sensitive devices
- H01G9/2004—Light-sensitive devices characterised by the electrolyte, e.g. comprising an organic electrolyte
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M14/00—Electrochemical current or voltage generators not provided for in groups H01M6/00 - H01M12/00; Manufacture thereof
- H01M14/005—Photoelectrochemical storage cells
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M8/00—Fuel cells; Manufacture thereof
- H01M8/02—Details
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M8/00—Fuel cells; Manufacture thereof
- H01M8/10—Fuel cells with solid electrolytes
- H01M8/1016—Fuel cells with solid electrolytes characterised by the electrolyte material
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10F—INORGANIC SEMICONDUCTOR DEVICES SENSITIVE TO INFRARED RADIATION, LIGHT, ELECTROMAGNETIC RADIATION OF SHORTER WAVELENGTH OR CORPUSCULAR RADIATION
- H10F10/00—Individual photovoltaic cells, e.g. solar cells
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/052—Li-accumulators
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M2300/00—Electrolytes
- H01M2300/0017—Non-aqueous electrolytes
- H01M2300/0025—Organic electrolyte
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E10/00—Energy generation through renewable energy sources
- Y02E10/50—Photovoltaic [PV] energy
- Y02E10/542—Dye sensitized solar cells
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/10—Energy storage using batteries
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/13—Energy storage using capacitors
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/30—Hydrogen technology
- Y02E60/50—Fuel cells
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02P—CLIMATE CHANGE MITIGATION TECHNOLOGIES IN THE PRODUCTION OR PROCESSING OF GOODS
- Y02P70/00—Climate change mitigation technologies in the production process for final industrial or consumer products
- Y02P70/50—Manufacturing or production processes characterised by the final manufactured product
Definitions
- Ionic liquid having phosphonium cation and method for producing the same
- the present invention relates to an ionic liquid which exhibits a liquid state in a wide temperature range from a low temperature and has a low viscosity and an excellent electrochemical stability, a method for producing the ionic liquid, a storage device using the ionic liquid, and a lithium secondary battery.
- the present invention relates to a secondary battery, an electric double layer capacitor, a dye-sensitized solar cell, a fuel cell, or a reaction solvent.
- Non-Patent Documents 4 and 5 ionic liquids with relatively low viscosity and melting point, such as imidazolium-based cations and containing cations, are poor in stability due to low reduction stability and narrow potential window, and are not applicable to electrolytes for power storage devices. There are many things that have drawbacks such as difficulties. (See Patent Document 3, Non-Patent Documents 4 and 5)
- the ability to have a small body is a major obstacle to the application as an electrolyte, electrolyte, or additive for lithium secondary batteries, electric double layer capacitors, fuel cells or dye-sensitized solar cells, or power storage devices. .
- Patent Document 1 Pamphlet of International Publication No. 02/076924
- Patent Document 2 JP 2003-331918 A
- Patent Document 3 ::: Special Table 2001-517205
- Non-Patent Literature 1 Matsumoto Hajime, Amagasaki Yoshinori, Molten Salt and High Temperature Chemistry, 44, 7 (2001)
- Non-Patent Document 2 H. Matsumoto, M. Yanagida, K. Tanimoto, M. Nomura, Y. Kitagawa and Y. Miyazaki, Chem. Lett, 8, 922 (2000)
- Non-Patent Document 3 D.R.MacFarlane, J.Sun, J.Golding, P.Meakin and M.Forsyth, Electro chemica Acta, 45, 1271 (2000)
- Non-Patent Document 4 Kaji principle, Electrochemistry, 70, No.2, 130 (2002)
- Non-Special Reference 5 Y. Katayama, b. Dan, T. Miura and T. Kishi, Journal of The Electrochemical Society, 148 (2), C102— C105 (2001)
- An object of the present invention is to provide an ionic liquid having low viscosity, good electrical conductivity, and excellent electrochemical stability, and a method for producing the ionic liquid.
- Ionic liquids that can be used as materials for lithium secondary batteries, electric double layer capacitors, dye-sensitized solar cells, fuel cells or reaction solvents, especially ionic liquids that exhibit a stable liquid at room temperature Specifically, it is an object to provide an ionic liquid containing a novel phosphonium cation. Means for solving the problem
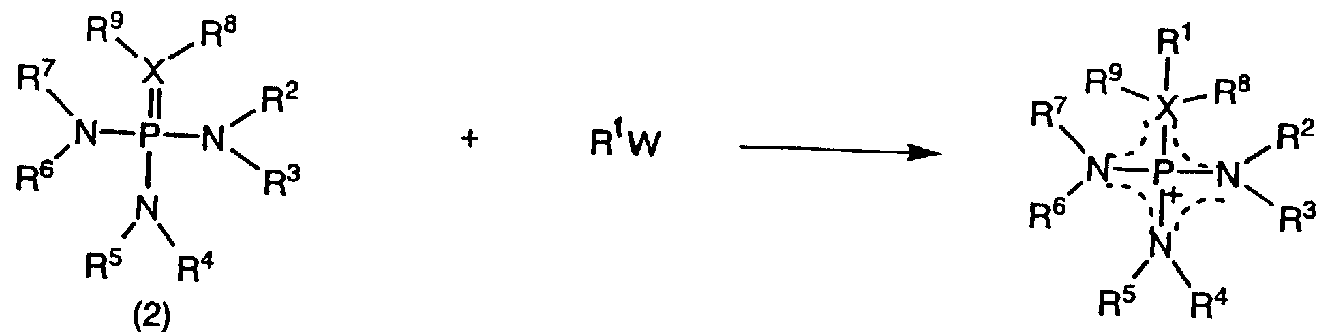
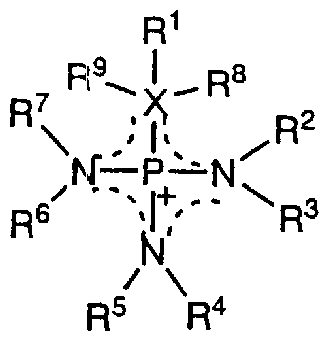
- the inventors of the present invention synthesized a large number of salts having a cationic component and a cation component, and intensively studied an ionic liquid for achieving the above object. As a result, the following general formula (1) was obtained.
- the ionic liquid containing one or more selected components as the cation component constitutes an ionic liquid with low viscosity, good conductivity, and excellent electrochemical stability. I found that I could do it.
- Substituents R 1 to R 9 in the formula are independent of each other and may be the same or different.
- Substituents R 1 to R 9 are each an H atom, an alkyl having a C to C linear or side chain.
- Lucenyl group having a linear or side chain with single or multiple triple bonds of c to C
- the H atom contained in one or a plurality of substituents among these substituents R 1 to R 9 can be partially or completely substituted with a halogen atom, or can be partially substituted with a CN group or a NO group. Can be replaced with
- substituents among the substituents R 1 to R 9 may jointly form a cyclic structure.
- Atoms and / or atoms selected from the group R'-0-,-0-P (0) R'-0-, and -P (R ') N-
- R ′ is an alkyl having a C to C linear or side chain
- X represents an S atom, an O atom, or a C atom.
- R 8 and R 9 are substituents that are present only when X is a C atom, and when X is a C atom, X, R 1 , R 8 and R 9 are jointly saturated or partially Form a completely or completely unsaturated ring structure! Furthermore, a dotted line shows a conjugate structure. ]
- the present invention relates to "an ionic liquid containing an organic substance represented by the general formula (1) as a cation component" and "an ionic liquid having both a cation component and a cation component, Brief description of the drawings is achieved by providing an ionic liquid whose component is one or more selected from the group forces of the cation component represented by the general formula (1)
- FIG. 1 is a graph showing a CV curve of tri (dimethylamino) butoxyphospho-umbistrifluoromethanesulfonylimide of Example 3.
- FIG. 2 Tri (dimethylamino) butylphospho-umbis trifluoromethane sulfone of Example 4 It is a graph which shows the cv curve of a diluimide.
- Examples of the cation component represented by the general formula (1) include an alkyl group having a linear or side chain of the substituents R 1 to R 9 1S C to C in the general formula (1), a saturated state Or partial
- a fully unsaturated cycloalkyl group, aryl group, or heterocyclic group and the H atom contained in one or more of these substituents R 1 to R 9 is a halogen atom.
- R 1 to R 9 is a halogen atom.
- R 1 to R 9 are -0-, -C (0)-, -C (0) 0-, -S-, -S (0)-, -NR '-, And -N (R') [where R 'is a straight chain of C to C
- R 1 to R 9 in the general formula (1) are each an alkyl group or an alkoxy group (R 1
- X is an S atom, an O atom or a C atom.
- the H atom contained in these substituents R may be partially or completely by a halogen atom, or by a CN group or NO group. Can be partially replaced.
- R f is a fluorine-containing substituent.
- One or more kinds of forces are also selected, and more preferably, [RS0] ", [R f SO
- the group 2 2 3 3 3 2 4 4 4 3 group is also one or more selected.
- the combination of the cationic component and these ion components it is more preferable than the combination of the cationic component and these ion components, and it has characteristics, that is, it exhibits a stable liquid in a wide temperature range of low-temperature force, and has low viscosity and good conductivity.
- an ionic liquid having particularly excellent electrochemical stability can be formed.
- ionic liquids having an alkyl group or an alkoxy group having R 1 to R 9 (R 1 to R 9 may be the same or different from each other!).
- X in the cation component represented by the general formula (1) is an S atom or an O atom. Those substituted with these atoms, which are more preferred when used, can constitute low melting ionic liquids. More preferred is an ionic liquid in which X is an o atom.
- R 7 is an alkyl group having a C to C straight chain
- R 8 and R 9 are H atoms
- an ionic liquid that exhibits a stable liquid state in a wide temperature range from a low temperature, has a low viscosity, good conductivity, and excellent electrochemical stability can be formed.
- the ionic liquid of the present invention described above is an ionic liquid having excellent conductivity, low viscosity, and excellent electrochemical stability. Therefore, the ionic liquid of the present invention can be used as a material used in lithium secondary batteries, electric double layer capacitors, fuel cells, or dye-sensitized solar cells, such as electrolytes, electrolytes, or additives for storage devices. It is useful as a reaction solvent for various reactions. In particular, an ionic liquid having both low viscosity and electrochemical stability has not been obtained so far, and the ionic liquid proposed in the present invention is compatible with both.
- the cation represented by the general formula (1) may have a positive charge in the molecule depending on the type of heteroatom represented by force X expressed as a phosphorous cation with a positive charge on the P atom for convenience. It is thought to be delocalized.
- Alkylating agent (R 1 W) is added dropwise to the organic material represented by the general formula (2), which is a raw material, and reacted at a predetermined temperature and time. After washing with jetyl ether or the like, vacuum drying is performed.
- Alkylating agents (R 1 W) include dialkyl sulfate, dialkyl ester sulfonate, dialkyl ester carbonate, trialkyl ester phosphate, mono- or poly-fluoroalkyl sulfonate alkyl ester, perfluoroalkyl.
- Examples include sulfonic acid alkyl esters, mono- or polyfluorocarboxylic acid alkyls, perfluorocarbonic acid alkyls, alkyl iodides, alkyl bromides, alkyl chlorides, sulfuric acid, nitric acid, and hydrochloric acid.
- ionic liquids having different anions can be obtained by exchanging anions by the following method.
- the ion binding compound AQ for example, LiN (CF 2 SO 4), NaN (CF 3 S
- substituents R 1 to R 9 in the general formula (3) are independent of each other and may be the same or different.
- Substituents R 1 to R 9 are each H atom, halogen atom, C
- an H atom contained in one or more substituents among these substituents R 1 to R 9 Can be partially or fully substituted by a halogen atom, or CN group, NO
- any substituents among R 1 to R 9 may jointly form a cyclic structure.
- X represents an S atom, an O atom or a C atom.
- R 8 and R 9 are substituents that exist only when is a C atom, and in the case of an X force atom, X, R 1 , R 8 and R 9 are jointly saturated or partially Alternatively, a completely unsaturated ring structure may be formed.
- halogen atom examples include F, Cl, Br and I.
- cycloalkyl group examples include cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, cyclooctyl, cyclonol, cyclodecyl, and the like.
- the cycloalkyl group includes those having an unsaturated bond such as a cycloalkenyl group and a cycloalkyl group, and can be partially or completely substituted by a halogen atom, or can be a CN group, NO Can be partially substituted by a group.
- heterocyclic group examples include pyrrolidyl, pyrrolinyl, imidazolidinyl, imidazolinyl, pyrarozolidinyl, pyrazonyl, piperidyl, piperazinyl, morpholinyl, and benzyl groups.
- These heterocyclic groups contain one or more alkyl groups, alkoxy groups, hydroxyl groups, carboxyl groups, amino groups, alkylamino groups, dialkylamino groups, thiol groups, alkylthio groups, and halogen atoms. It's good.
- aryl group examples include phenol, tamenyl, mesityl, tolyl, xylyl group and the like (these aryl groups include an alkyl group, an alkoxy group, a hydroxyl group, a carboxyl group, an acyl group, a formyl group, Amino group, alkylamino group, dialkylamino group, thiol group,
- the alkylthio group and the halogen atom may contain one or a plurality of halogen atoms.
- alkoxyalkyl groups such as methoxymethyl, methoxyethyl, ethoxymethyl, ethoxyethyl and the like can be mentioned.
- hetero atom represented by X in the formula examples include S, O, and C atoms. Particularly preferred are S or O atoms, and substitution with these atoms makes it possible to obtain an ionic liquid having a lower melting point.
- the above-mentioned key-on component can be exemplified as the key-on component Q to be combined with the compound represented by the general formula (3).
- the compound was identified by a nuclear magnetic resonance analyzer (BRUKER Ultra Shield 300 NMR Spectrometer manufactured by BRUKER). The spectrum data is shown below.
- the compound was identified by a nuclear magnetic resonance analyzer (BRUKER Ultra Shield 300 N MR Spectrometer manufactured by BRUKER) and confirmed to be the target product, tri (dimethylamino) methoxyphospho-umbis trifluoromethanesulfurimide.
- the spectrum data is shown below.
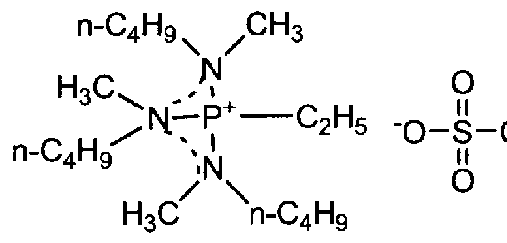
- hexane methyl phosphate triamide (2.0 g, 11.2 mmol) was dissolved in disulfate at room temperature under an N atmosphere.
- the compound was identified by a nuclear magnetic resonance analyzer (BRUKER Ultra Shield 300 NMR Spectrometer manufactured by BRUKER). The spectrum data is shown below.
- the compound was identified with a nuclear magnetic resonance analyzer (BRUKER Ultra Shield 300 NMR Spectrometer manufactured by BRUKER) and confirmed to be the target product, tri (dimethylamino) ethoxyphospho-umbitrifluorotrifluorosulfimide. .
- the spectrum data is shown below.
- the compound was identified by a nuclear magnetic resonance analyzer (BRUKER Ultra Shield 300 NMR Spectrometer manufactured by BRUKER). The spectrum data is shown below.
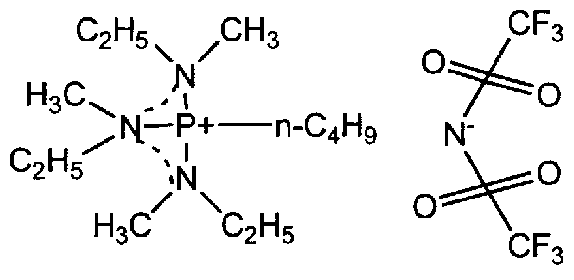
- the compound was identified with a nuclear magnetic resonance analyzer (BRUKER Ultra Shield 300 NMR Spectrometer manufactured by BRUKER) and confirmed to be the target product, tri (dimethylamino) butoxyphospho- umbitrifluoromethanesulfurimide. . Spectral data below Shown in
- the viscosity at 25 ° C. by a vibration viscometer was 45 mPa ′s.
- the conductivity at 25 ° C by the AC impedance method was 0.3 Sm- 1 .
- the compound was identified by a nuclear magnetic resonance analyzer (BRUKER Ultra Shield 300 NMR Spectrometer manufactured by BRUKER). The spectrum data is shown below.
- the compound was identified with a nuclear magnetic resonance analyzer (BRUKER Ultra Shield 300 NMR Spectrometer manufactured by BRUKER) and confirmed to be the target product, tri (dimethylamino) butylphospho-mumbitrifluoromethanesulfurimide.
- BRUKER Ultra Shield 300 NMR Spectrometer manufactured by BRUKER
- the spectrum data is shown below.
- the viscosity at 40 ° C. by a vibration viscometer was 53 mPa's.
- the conductivity at 40 ° C by the AC impedance method was 0.3 Sm- 1 .
- the working electrode and the counter electrode are Pt
- the reference electrode is Li, manufactured by Hokuto Denko Co., Ltd. From the cyclic voltammogram measured using the electrochemical measurement system HZ-3000, the potential window was 0V to 4.9V with respect to Li / Li +.
- Figure 2 shows the CV curve of tri (dimethylamino) butylphospho- trifluorobistrifluoromethanesulfonylimide.
- the compound was identified by a nuclear magnetic resonance analyzer (BRUKER Ultra Shield 300 NMR Spectrometer manufactured by BRUKER). The spectrum data is shown below.
- the compound was identified by a nuclear magnetic resonance analyzer (BRUKER Ultra Shield 300 NMR Spectrometer manufactured by BRUKER). The spectrum data is shown below.
- the compound was identified by a nuclear magnetic resonance analyzer (BRUKER Ultra Shield 300 NMR Spectrometer manufactured by BRUKER). The spectrum data is shown below.
- the compound was identified by a nuclear magnetic resonance analyzer (BRUKER Ultra Shield 300 NMR Spectrometer manufactured by BRUKER). The spectrum data is shown below.
- the compound was identified by a nuclear magnetic resonance analyzer (BRUKER Ultra Shield 300 NMR Spectrometer manufactured by BRUKER). The spectrum data is shown below.
- the compound was identified by a nuclear magnetic resonance analyzer (BRUKER Ultra Shield 300 NMR Spectrometer manufactured by BRUKER). The spectrum data is shown below.
- the melting point was measured with a scanning differential calorimeter (DSC8230, manufactured by Shimadzu Corporation), but the peak considered to be the melting point was strong.
- the thermal decomposition temperature was measured with a thermogravimetric analyzer (TG8120, manufactured by Rigaku Corporation).
- the 5% weight loss temperature measured at a heating rate of 10 ° C Zmin was 250.5 ° C.
- the compound was identified by a nuclear magnetic resonance analyzer (BRUKER Ultra Shield 300 NMR Spectrometer manufactured by BRUKER). The spectrum data is shown below.
- the conductivity at 25 ° C. by the AC impedance method was 0.085 Sm 1 .
- the compound was identified by a nuclear magnetic resonance analyzer (BRUKER Ultra Shield 300 NMR Spectrometer manufactured by BRUKER). The spectrum data is shown below.
- the crystallization temperature was 0 ° C, -32.7 ° C, and the glass transition temperature was -75.5 ° C.
- the thermal decomposition temperature was measured with a thermogravimetric analyzer (TG8120, manufactured by Rigaku Corporation). Measured at a heating rate of 10 ° CZmin The specified 5% weight loss temperature was 389.1 ° C.
- the compound was identified by a nuclear magnetic resonance analyzer (BRUKER Ultra Shield 300 NMR Spectrometer manufactured by BRUKER). The spectrum data is shown below.
- the compound was identified by a nuclear magnetic resonance analyzer (BRUKER Ultra Shield 300 NMR Spectrometer manufactured by BRUKER). The spectrum data is shown below.
- the compound was identified by a nuclear magnetic resonance analyzer (BRUKER Ultra Shield 300 NMR Spectrometer manufactured by BRUKER). The spectrum data is shown below.
- an ionic liquid that exhibits a stable liquid state in a wide temperature range from a low temperature, has a low viscosity, good electrical conductivity, and excellent electrochemical stability. Can be provided.
- the ionic liquid of the present invention can be applied to lithium secondary batteries, electric double layer capacitors, fuel cells, dye-sensitized solar cells, storage device electrolytes, electrolytic solutions or additives, reaction solvents, and the like. .
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Electrochemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Biochemistry (AREA)
- General Health & Medical Sciences (AREA)
- Molecular Biology (AREA)
- Health & Medical Sciences (AREA)
- Manufacturing & Machinery (AREA)
- Power Engineering (AREA)
- Sustainable Energy (AREA)
- Sustainable Development (AREA)
- Microelectronics & Electronic Packaging (AREA)
- Physics & Mathematics (AREA)
- Condensed Matter Physics & Semiconductors (AREA)
- General Physics & Mathematics (AREA)
- Inorganic Chemistry (AREA)
- Secondary Cells (AREA)
- Hybrid Cells (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Electric Double-Layer Capacitors Or The Like (AREA)
Abstract
Description
Claims
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US11/918,859 US20090023954A1 (en) | 2005-04-28 | 2006-04-28 | Ionic liquid containing phosphonium ion and method for producing the same |
| JP2007514827A JP5070044B2 (ja) | 2005-04-28 | 2006-04-28 | ホスホニウムカチオンを有するイオン液体およびその製造方法 |
| CN2006800143225A CN101166747B (zh) | 2005-04-28 | 2006-04-28 | 含有鏻阳离子的离子液体及其制造方法 |
| CA002606482A CA2606482A1 (en) | 2005-04-28 | 2006-04-28 | Ionic liquid containing phosphonium ion and method for producing same |
| EP06745829.9A EP1876181B1 (en) | 2005-04-28 | 2006-04-28 | Ionic liquid containing phosphonium ion and method for producing same |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PCT/JP2005/008229 WO2006117872A1 (ja) | 2005-04-28 | 2005-04-28 | ホスホニウムカチオンを有するイオン液体およびその製造方法 |
| JPPCT/JP2005/008229 | 2005-04-28 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2006118232A1 true WO2006118232A1 (ja) | 2006-11-09 |
Family
ID=37307679
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2005/008229 Ceased WO2006117872A1 (ja) | 2005-04-28 | 2005-04-28 | ホスホニウムカチオンを有するイオン液体およびその製造方法 |
| PCT/JP2006/308948 Ceased WO2006118232A1 (ja) | 2005-04-28 | 2006-04-28 | ホスホニウムカチオンを有するイオン液体およびその製造方法 |
Family Applications Before (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2005/008229 Ceased WO2006117872A1 (ja) | 2005-04-28 | 2005-04-28 | ホスホニウムカチオンを有するイオン液体およびその製造方法 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US20090023954A1 (ja) |
| EP (1) | EP1876181B1 (ja) |
| KR (1) | KR100961041B1 (ja) |
| CN (1) | CN101166747B (ja) |
| CA (1) | CA2606482A1 (ja) |
| RU (1) | RU2374257C2 (ja) |
| WO (2) | WO2006117872A1 (ja) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2007063959A1 (ja) * | 2005-12-02 | 2007-06-07 | Kanto Denka Kogyo Co., Ltd. | P-n結合を含むホスホニウムカチオンを有するイオン液体およびその製造方法 |
| JP2014011098A (ja) * | 2012-07-02 | 2014-01-20 | Toyota Motor Corp | 空気電池用イオン性液体、当該イオン性液体を含有するリチウム空気電池用電解液及び空気電池 |
Families Citing this family (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP5070044B2 (ja) * | 2005-04-28 | 2012-11-07 | 関東電化工業株式会社 | ホスホニウムカチオンを有するイオン液体およびその製造方法 |
| JP5134783B2 (ja) * | 2005-12-26 | 2013-01-30 | 株式会社ブリヂストン | 電池用非水電解液及びそれを備えた非水電解液電池 |
| WO2007074609A1 (ja) * | 2005-12-26 | 2007-07-05 | Bridgestone Corporation | 電池用非水電解液及びそれを備えた非水電解液電池、並びに電気二重層キャパシタ用電解液及びそれを備えた電気二重層キャパシタ |
| SG191886A1 (en) | 2011-01-10 | 2013-08-30 | Reliance Ind Ltd | Process for preparation of acetals |
| CN103402959B (zh) | 2011-01-10 | 2016-05-18 | 瑞来斯实业有限公司 | 在含水介质中制成二缩醛化合物的方法 |
| JP5827694B2 (ja) | 2011-01-10 | 2015-12-02 | リライアンス、インダストリーズ、リミテッドReliance Industries Limited | アルジトールアセタールの製造方法 |
| US9318271B2 (en) * | 2012-06-21 | 2016-04-19 | Schlumberger Technology Corporation | High temperature supercapacitor |
| JP2015011823A (ja) * | 2013-06-27 | 2015-01-19 | 住友電気工業株式会社 | リチウム電池 |
| KR20160077270A (ko) * | 2014-12-22 | 2016-07-04 | 삼성에스디아이 주식회사 | 리튬 이차전지용 전해액 및 이를 구비한 리튬 이차전지 |
| KR102166742B1 (ko) | 2015-12-31 | 2020-10-16 | 에스케이텔레콤 주식회사 | 내비게이션 방법, 이를 위한 장치 및 시스템 |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2002076924A1 (en) * | 2001-03-26 | 2002-10-03 | Nisshinbo Industries, Inc., | Ionic liquid, electrolyte salt for storage device, electrolytic solution for storage device, electric double layer capacitor, and secondary battery |
Family Cites Families (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2774658A (en) * | 1955-08-30 | 1956-12-18 | Mousanto Chemical Company | Herbicidal alkyl-amino-phosphonium halides |
| US5541287A (en) * | 1992-06-09 | 1996-07-30 | Neorx Corporation | Pretargeting methods and compounds |
| GB9616264D0 (en) * | 1996-08-02 | 1996-09-11 | British Nuclear Fuels Plc | Reprocessing irradiated fuel |
| EP2243539A3 (en) * | 2002-04-05 | 2011-06-01 | University Of South Alabama | Functionalized ionic liquids, and methods of use thereof |
-
2005
- 2005-04-28 WO PCT/JP2005/008229 patent/WO2006117872A1/ja not_active Ceased
-
2006
- 2006-04-28 RU RU2007140882/04A patent/RU2374257C2/ru not_active IP Right Cessation
- 2006-04-28 KR KR1020077025417A patent/KR100961041B1/ko not_active Expired - Lifetime
- 2006-04-28 CN CN2006800143225A patent/CN101166747B/zh not_active Expired - Lifetime
- 2006-04-28 WO PCT/JP2006/308948 patent/WO2006118232A1/ja not_active Ceased
- 2006-04-28 EP EP06745829.9A patent/EP1876181B1/en not_active Expired - Lifetime
- 2006-04-28 US US11/918,859 patent/US20090023954A1/en not_active Abandoned
- 2006-04-28 CA CA002606482A patent/CA2606482A1/en not_active Abandoned
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2002076924A1 (en) * | 2001-03-26 | 2002-10-03 | Nisshinbo Industries, Inc., | Ionic liquid, electrolyte salt for storage device, electrolytic solution for storage device, electric double layer capacitor, and secondary battery |
Non-Patent Citations (4)
| Title |
|---|
| HAYNES R.K. ET AL.: "The preparation of (methylthio)- and (methylseleno)tri(alkyl or aryl)phosphonium salts and their reactions with carboxylic acid and alcohols", AUSTRALIAN JOURNAL OF CHEMISTRY, vol. 37, no. 6, 1984, pages 1183 - 1194, XP003003063 * |
| KIM T.C. ET AL.: "Synthesis of new bis(dialkylamino)-1,4,2(lambda 4)-diazaphospholanium salts from phosphenium cations and imines", TETRAHEDRON LETTERS, vol. 31, no. 31, 1990, pages 4459 - 4462, XP003003062 * |
| PETZ W. ET AL.: "The reactions of CH2:P(NMe2)3 with Fe(CO)5, Cr(CO)6, and CS2. Molecular structures of [MeP(NMe2)3][(CO)5CrC(0)CH:P(NMe2)3] and (CO)4Fe:C(OMe)CH:P(NMe2)3", ZEITSCHRIFT FUER ANORGANISCHE UND ALLGEMEINE CHEMIE, vol. 624, no. 7, 1998, pages 1123 - 1129, XP003003061 * |
| See also references of EP1876181A4 * |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2007063959A1 (ja) * | 2005-12-02 | 2007-06-07 | Kanto Denka Kogyo Co., Ltd. | P-n結合を含むホスホニウムカチオンを有するイオン液体およびその製造方法 |
| US8871974B2 (en) | 2005-12-02 | 2014-10-28 | Kanto Denka Kogyo Co., Ltd. | Ionic liquid containing phosphonium cation having P—N bond and method for producing same |
| JP2014011098A (ja) * | 2012-07-02 | 2014-01-20 | Toyota Motor Corp | 空気電池用イオン性液体、当該イオン性液体を含有するリチウム空気電池用電解液及び空気電池 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP1876181A4 (en) | 2010-11-17 |
| RU2007140882A (ru) | 2009-05-10 |
| CA2606482A1 (en) | 2006-11-09 |
| CN101166747A (zh) | 2008-04-23 |
| US20090023954A1 (en) | 2009-01-22 |
| CN101166747B (zh) | 2011-08-31 |
| EP1876181A1 (en) | 2008-01-09 |
| RU2374257C2 (ru) | 2009-11-27 |
| WO2006117872A1 (ja) | 2006-11-09 |
| KR100961041B1 (ko) | 2010-06-01 |
| KR20080003855A (ko) | 2008-01-08 |
| EP1876181B1 (en) | 2013-07-24 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5265197B2 (ja) | P−n結合を含むホスホニウムカチオンを有するイオン液体およびその製造方法 | |
| KR101650207B1 (ko) | 반응성 이온성 액체 | |
| EP3601233B1 (en) | Salts for use in electrolyte compositions or as electrode additives | |
| WO2006118232A1 (ja) | ホスホニウムカチオンを有するイオン液体およびその製造方法 | |
| JP6170646B1 (ja) | 電解質組成物、二次電池、及び二次電池の使用方法 | |
| US20020160261A1 (en) | Borate salts for use in electrochemical cells | |
| US8026394B2 (en) | Lithium salt and method for producing thereof | |
| JP5227524B2 (ja) | S−n結合を含むスルホニウムカチオンを有するイオン液体およびその製造方法 | |
| CN101723976A (zh) | 含有氟代烷基三氟硼酸根阴离子的离子液体 | |
| JP2005179254A (ja) | 常温溶融塩およびその製造方法 | |
| KR101156825B1 (ko) | 직류 전지용 전도성 염, 이의 제조방법 및 용도 | |
| JP5070044B2 (ja) | ホスホニウムカチオンを有するイオン液体およびその製造方法 | |
| JP4815127B2 (ja) | 常温溶融塩の製造方法 | |
| JP4766914B2 (ja) | Sを含む環状イオン液体およびその製造方法 | |
| JP2004262896A (ja) | 4級アンモニウム系常温溶融塩及び該化合物の製造法 | |
| HK40119322A (en) | Salts for use in electrolyte compositions or as electrode additives |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200680014322.5 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2007514827 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 11918859 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2006745829 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2606482 Country of ref document: CA |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1020077025417 Country of ref document: KR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2007140882 Country of ref document: RU |
|
| WWP | Wipo information: published in national office |
Ref document number: 2006745829 Country of ref document: EP |