WO2009142228A1 - 原子力用高強度Ni基合金管及びその製造方法 - Google Patents
原子力用高強度Ni基合金管及びその製造方法 Download PDFInfo
- Publication number
- WO2009142228A1 WO2009142228A1 PCT/JP2009/059249 JP2009059249W WO2009142228A1 WO 2009142228 A1 WO2009142228 A1 WO 2009142228A1 JP 2009059249 W JP2009059249 W JP 2009059249W WO 2009142228 A1 WO2009142228 A1 WO 2009142228A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- strength
- nuclear power
- base alloy
- less
- hot
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B21—MECHANICAL METAL-WORKING WITHOUT ESSENTIALLY REMOVING MATERIAL; PUNCHING METAL
- B21C—MANUFACTURE OF METAL SHEETS, WIRE, RODS, TUBES, PROFILES OR LIKE SEMI-MANUFACTURED PRODUCTS OTHERWISE THAN BY ROLLING; AUXILIARY OPERATIONS USED IN CONNECTION WITH METAL-WORKING WITHOUT ESSENTIALLY REMOVING MATERIAL
- B21C23/00—Extruding metal; Impact extrusion
- B21C23/02—Making uncoated products
- B21C23/04—Making uncoated products by direct extrusion
- B21C23/08—Making wire, rods or tubes
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B21—MECHANICAL METAL-WORKING WITHOUT ESSENTIALLY REMOVING MATERIAL; PUNCHING METAL
- B21J—FORGING; HAMMERING; PRESSING METAL; RIVETING; FORGE FURNACES
- B21J1/00—Preparing metal stock or similar ancillary operations prior, during or post forging, e.g. heating or cooling
- B21J1/02—Preliminary treatment of metal stock without particular shaping, e.g. salvaging segregated zones, forging or pressing in the rough
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D6/00—Heat treatment of ferrous alloys
- C21D6/004—Heat treatment of ferrous alloys containing Cr and Ni
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D7/00—Modifying the physical properties of iron or steel by deformation
- C21D7/13—Modifying the physical properties of iron or steel by deformation by hot working
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D8/00—Modifying the physical properties of ferrous metals or ferrous alloys by deformation combined with, or followed by, heat treatment
- C21D8/10—Modifying the physical properties of ferrous metals or ferrous alloys by deformation combined with, or followed by, heat treatment during manufacturing of tubular bodies
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D9/00—Heat treatment, e.g. annealing, hardening, quenching or tempering, adapted for particular articles; Furnaces therefor
- C21D9/08—Heat treatment, e.g. annealing, hardening, quenching or tempering, adapted for particular articles; Furnaces therefor for tubular bodies or pipes
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C19/00—Alloys based on nickel or cobalt
- C22C19/03—Alloys based on nickel or cobalt based on nickel
- C22C19/05—Alloys based on nickel or cobalt based on nickel with chromium
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C19/00—Alloys based on nickel or cobalt
- C22C19/03—Alloys based on nickel or cobalt based on nickel
- C22C19/05—Alloys based on nickel or cobalt based on nickel with chromium
- C22C19/058—Alloys based on nickel or cobalt based on nickel with chromium without Mo and W
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22F—CHANGING THE PHYSICAL STRUCTURE OF NON-FERROUS METALS AND NON-FERROUS ALLOYS
- C22F1/00—Changing the physical structure of non-ferrous metals or alloys by heat treatment or by hot or cold working
- C22F1/10—Changing the physical structure of non-ferrous metals or alloys by heat treatment or by hot or cold working of nickel or cobalt or alloys based thereon
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22B—PRODUCTION AND REFINING OF METALS; PRETREATMENT OF RAW MATERIALS
- C22B23/00—Obtaining nickel or cobalt
- C22B23/06—Refining
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22B—PRODUCTION AND REFINING OF METALS; PRETREATMENT OF RAW MATERIALS
- C22B9/00—General processes of refining or remelting of metals; Apparatus for electroslag or arc remelting of metals
- C22B9/16—Remelting metals
- C22B9/18—Electroslag remelting
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10T—TECHNICAL SUBJECTS COVERED BY FORMER US CLASSIFICATION
- Y10T428/00—Stock material or miscellaneous articles
- Y10T428/12—All metal or with adjacent metals
Definitions
- the present invention relates to a Ni-based alloy tube excellent in corrosion resistance in a high-temperature and high-pressure water environment of a nuclear power plant and a method for producing the same.
- the present invention relates to a Ni-based alloy tube suitable for a structural member such as a lid for a reactor vessel of a pressurized water reactor (PWR) and a method for manufacturing the same.
- a structural member such as a lid for a reactor vessel of a pressurized water reactor (PWR) and a method for manufacturing the same.
- PWR pressurized water reactor
- Inconel 600 (15% Cr-75% Ni) and Inconel are excellent as Ni-based alloys with excellent corrosion resistance. 690 (30% Cr-60% Ni) has been used.
- Patent Documents 1 and 2 disclose Ni-based alloys that have improved resistance to stress corrosion cracking by subjecting them to final annealing after defining the heating temperature and holding time after extrusion and cold working.
- Patent Document 3 discloses a Ni-based alloy in which a grain boundary is eliminated by forming an amorphous alloy layer on the surface layer, thereby improving the intergranular damage resistance.
- Patent Document 4 discloses a structure in which at least one of a ⁇ ′ phase and a ⁇ ′′ phase is contained in a ⁇ base, and M 23 C 6 is preferentially precipitated semi-continuously at a grain boundary. Also disclosed is a high-strength Ni-based alloy with improved stress corrosion cracking resistance.
- Patent Document 5 improved the intergranular corrosion resistance, intergranular stress corrosion cracking resistance and mechanical strength in the weld heat-affected zone by appropriately balancing the content of each component of C, N, and Nb.
- Ni-based alloys are disclosed.
- Patent Document 6 discloses a Ni-based alloy in which the grain boundary stress corrosion cracking resistance is improved by setting the low-angle grain boundary ratio in the grain boundaries to a structure of 4% or more.
- Ni-based alloy pipes have been many proposals aimed at improving the corrosion resistance of Ni-based alloy tubes.
- the crystal grain size and strength variation increase as a result of solution heat treatment and subsequent aging treatment for carbide precipitation.
- the strength at the end of the pipe may be low. .
- the defective portion has to be cut down, and there is a problem in that the yield decreases.
- An object of the present invention is to solve such problems, and in a high-strength Ni-base alloy tube for nuclear power, a Ni-base alloy tube having uniform high-temperature strength over the entire length of the tube and a method for manufacturing the same It is to provide.
- the present inventors have conducted various studies and experiments on factors that improve high-temperature strength in high-strength Ni-base alloy tubes for nuclear power, and as a result, obtained the following knowledge (a) to (j).
- Ti and Nb combine with C and N to precipitate carbonitride that has the effect of refining crystal grains.
- the heating temperature before hot extruding is a temperature at which the crystal does not coarsen and Cr carbonitride dissolves, but Ti and Nb charcoal have the effect of refining the crystal grain. It is preferable to set the temperature at which nitride does not dissolve.
- the average dissolution rate is preferably 200 to 600 kg / hr. This is because if the speed exceeds 600 kg / hr, the floating of impurities at the time of dissolution becomes insufficient, and the suppression of segregation may be insufficient, and if the speed is less than 200 kg / hr, the productivity is too low. It is.
- the conditions of the heating temperature before hot extrusion and the processing ratio during hot extrusion are obtained by the secondary melting method using the electroslag remelting (ESR) method or the vacuum arc melting (VAR) method. It is preferable that the Ni-based alloy material is hot-forged and then heated to 1000 to 1160 ° C. and then hot-extruded with an extruding ratio of 4 or more.
- the extrusion ratio is defined as [cross-sectional area before extrusion] / [cross-sectional area after extrusion].
- the upper limit of the heating temperature before hot extrusion is set to 1160 ° C., because Cr carbonitride is dissolved, but Ti and Nb carbonitride is not dissolved.
- the reason why the lower limit of the heating temperature before the inter-extrusion processing is set to 1000 ° C. is that if it is less than 1000 ° C., the deformation resistance during the hot extrusion processing is too large.
- the reason why the degree of hot extrusion processing is preferably set to 4 or more in terms of the extrusion ratio is that sufficient processing can be performed and uniform recrystallization can be performed, so that the crystal grains can be sufficiently refined. More preferably, the extrusion ratio is 5 or more.
- the upper limit of the extrusion ratio is not particularly defined, it is preferable to set the extrusion ratio to 30 or less because the larger the extrusion ratio, the more easily defects such as flaws appear in the product and the equipment needs to be enlarged.
- the purpose of the solution heat treatment is to sufficiently dissolve the carbide, and the heating temperature for this is preferably 980 to 1200 ° C. This is because when heated at 980 ° C. or higher, the carbide can be dissolved, and thus the corrosion resistance is improved. On the other hand, when it exceeds 1200 ° C., the strength may be reduced by coarsening. A more preferred upper limit temperature is 1090 ° C.
- the purpose of aging treatment is to precipitate carbides at the grain boundaries.
- the heating temperature is preferably 550 to 850 ° C. When heated in this temperature range, carbides can be sufficiently precipitated at the grain boundaries.
- the target value of the high temperature strength of the Ni-based alloy tube for nuclear power according to the present invention is, for example, the design yield point (proof stress) at 350 ° C specified in the nuclear power equipment standard JSME S NC-1. 199 MPa, design tensile strength (tensile strength) is 530 MPa.
- the crystal grain size of the high-strength Ni-base alloy tube for nuclear power after solution heat treatment and aging treatment is required to be fine particles having a grain size number of 6 or more according to JIS G0551. .
- the present invention has been made on the basis of the above-mentioned knowledge, and the gist thereof is the following high-strength Ni-base alloy tube for nuclear power and its manufacturing method.
- the present invention it is possible to provide a high-strength Ni-base alloy tube for nuclear power having a uniform high-temperature strength over the entire length of the tube and a method for manufacturing the same.
- C 0.04% or less C is an element necessary for ensuring strength. However, if the content exceeds 0.04%, Cr carbide increases and stress corrosion cracking resistance decreases. For this reason, the upper limit of the C content is set to 0.04%. A preferable upper limit is 0.03% or less. In addition, when ensuring intensity
- Si 0.10 to 0.50% Si is an element used as a deoxidizer, and in order to obtain this effect, it is necessary to contain 0.10% or more. On the other hand, if the content exceeds 0.50%, the weldability deteriorates and the cleanliness decreases. Therefore, the Si content is set to 0.10 to 0.50%. A more preferable Si content is 0.22 to 0.45%.
- Mn 0.05 to 0.50% Mn has an effect of improving hot extrusion processability by fixing S, which is an impurity, as MnS, and is also an effective element as a deoxidizer.
- S an impurity
- MnS an impurity
- MnS an impurity
- Mn content is set to 0.05 to 0.50%.
- Ni is an element effective for ensuring the corrosion resistance of the alloy. In particular, 55% or more needs to be contained in order to exhibit a remarkable effect in improving the resistance to intergranular stress corrosion cracking in high temperature water containing acid resistance and chlorine ions.
- the upper limit of the content is 70% in relation to the necessary content of other elements such as Cr, Mn, and Si. Therefore, the Ni content needs to be 55 to 70%.
- a more preferable range of Ni content is more than 58% and 65% or less.
- a more preferable range of Ni content is more than 60% and 65% or less.
- Cr more than 26% and 35% or less Cr is an element necessary for maintaining the corrosion resistance of the alloy, and in order to ensure the required corrosion resistance, the Cr content needs to exceed 26%. There is. On the other hand, when the content exceeds 35%, the hot extrusion processability is remarkably deteriorated. For this reason, Cr content needs to exceed 26% and to be 35% or less. Preferably it is more than 27% and not more than 32%, more preferably 28 to 31%.
- Al 0.005 to 0.5%
- Al is an element that acts as a deoxidizer and needs to be contained in an amount of 0.005% or more.
- the content exceeds 0.5%, the cleanliness of the alloy is lowered, so the Al content is set to 0.5% or less. More preferably, it is 0.02 to 0.3%.
- N 0.02 to 0.10% N not only increases the strength of the alloy by forming Ti or Nb carbonitride with C, but in the present invention, it is combined with the segregation suppressing effect of N, C, Ti, Nb by the secondary melting method.
- This carbonitride can be uniformly dispersed and precipitated, and the structure after hot extrusion can be refined. In order to acquire the effect, it is necessary to make it contain 0.02% or more.
- the N content is set to 0.02 to 0.10%. More preferably, it is 0.03 to 0.06%.
- Ti 0.01 to 0.5% and Nb: 0.02 to 1.0% Ti forms carbonitrides to increase the strength of the alloy and improve hot extrusion processability There is an action to make. In order to acquire these effects, it is necessary to contain 0.01% or more of Ti. On the other hand, if the Ti content exceeds 0.5%, not only the effect is saturated, but also ductility is impaired by the formation of intermetallic compounds. Therefore, the Ti content is set to 0.01 to 0.5%. More preferably, it is 0.05 to 0.3%.
- Nb like Ti, has the effect of forming carbonitrides to increase the strength of the alloy and improve hot extrusion processability. In order to obtain these effects, it is necessary to contain 0.02% or more of Nb. On the other hand, if the Nb content exceeds 1.0%, the effect is not only saturated, but ductility is impaired by the formation of intermetallic compounds. Therefore, the Nb content is set to 0.02 to 1.0%. More preferably, it is 0.1 to 0.6%.
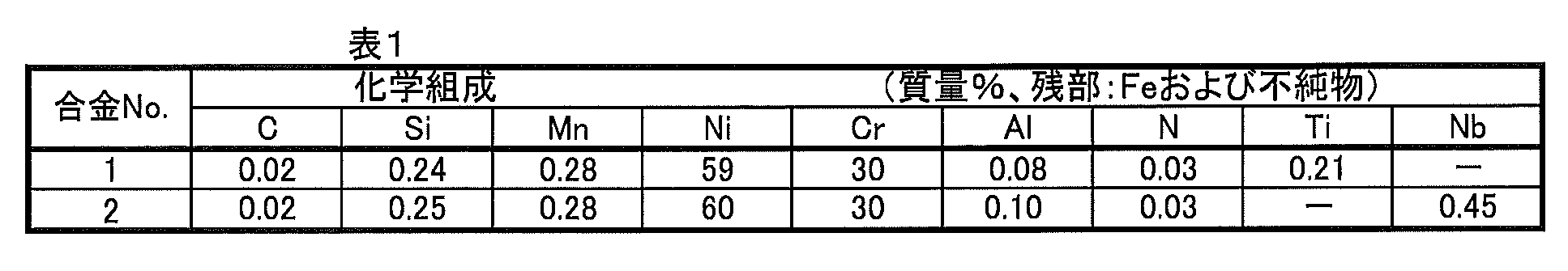
- a Ni-based alloy having the chemical composition shown in Table 1 is melted in an electric furnace, then refined by AOD and VOD, and then re-melted by ESR at a melting average speed of 500 kg / hr to obtain a Ni-based alloy material. It was. It was heated at 1270 ° C., hot forged at a forging ratio of 5, and then processed into a billet for hot extrusion. The billet was heated by changing the heating temperature of the billet and then subjected to hot extrusion at an extrusion ratio of 5 to obtain a Ni-based alloy tube having an outer diameter of 115 mm and a wall thickness of 27.5 mm. It was subjected to a solution heat treatment at 1075 ° C. ⁇ 30 minutes and an aging treatment at 700 ° C. ⁇ 900 minutes to obtain a final product. For comparison, a final product was obtained in the same manner for a Ni-based alloy material in which remelting by ESR was omitted.
- Table 2 shows the presence or absence of the secondary melting method by the ESR method, and the conditions under which the heating temperature before hot extrusion was changed.
- the Ni-based alloy has a fine structure and high strength at high temperatures (350 ° C) by appropriately selecting the application of the secondary melting method by the ESR method and the heating temperature before hot extrusion. was found to be obtained.
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Mechanical Engineering (AREA)
- Materials Engineering (AREA)
- Metallurgy (AREA)
- Organic Chemistry (AREA)
- Crystallography & Structural Chemistry (AREA)
- Physics & Mathematics (AREA)
- Thermal Sciences (AREA)
- Manufacturing & Machinery (AREA)
- Heat Treatment Of Steel (AREA)
- Extrusion Of Metal (AREA)
- Manufacture And Refinement Of Metals (AREA)
Abstract
Description
Nについても偏析が存在すると、同様にTiとNbの炭窒化物は均一には析出しないから、微細な結晶粒が均一に分散した組織を得ることができない。
(1) 質量%で、C:0.04%以下、Si:0.10~0.50%、Mn:0.05~0.50%、Ni:55~70%、Cr:26%超えて35%以下、Al:0.005~0.5%、N:0.02~0.10%、並びにTi:0.01~0.5%およびNb:0.02~1.0%のうちの一種以上を含有し、残部がFeおよび不純物からなり、結晶粒径がJIS G 0551での粒度番号6またはそれ以上の細粒であることを特徴とする原子力用高強度Ni基合金管。
(3) 質量%で、C:0.04%以下、Si:0.10~0.50%、Mn:0.05~0.50%、Ni:55~70%、Cr:26%超えて35%以下、Al:0.005~0.5%、N:0.02~0.10%、並びにTi:0.01~0.5%およびNb:0.02~1.0%のうちの一種以上を含有し、残部がFeおよび不純物からなり、二次溶解法によって得られたNi基合金素材を、熱間鍛造後、1000~1160℃に加熱した後に押出し比が4以上の加工度で熱間押出し加工し、さらに固溶化熱処理と時効処理を施すことを特徴とする原子力用高強度Ni基合金管の製造方法。
Cは、強度を確保するのに必要な元素であるが、含有量が0.04%を超えると、Cr炭化物が増え、耐応力腐食割れ性が低下する。このため、Cの含有量の上限は、0.04%とした。好ましい上限は0.03%以下である。なお、Cを含有させることによって強度確保を行う場合は、0.01%以上のCを含有させることが好ましい。
Siは、脱酸剤として使用される元素であり、この効果を得るためには0.10%以上含有させることが必要である。一方、0.50%を超えて含有させると、溶接性が悪化するとともに、清浄度が低下する。このため、Siの含有量は0.10~0.50%とした。より好ましいSi含有量は0.22~0.45%である。
Mnは、不純物であるSをMnSとして固定することで熱間押出し加工性の向上効果を有するとともに、脱酸剤としても有効な元素である。合金の熱間押出し加工性を確保するためには、Mnを0.05%以上含有させる必要がある。一方、0.50%を超えて過剰に含有させると、合金の清浄度が低下する。したがって、Mnの含有量は0.05~0.50%とした。
Niは、合金の耐食性を確保するのに有効な元素である。特に、耐酸性および塩素イオン含有高温水中における耐粒界応力腐食割れ性を向上させるのに顕著な作用を発揮するため、55%以上含有させる必要がある。一方、含有量の上限は、Cr、Mn、Si等の他元素の必要含有量との絡みで70%となる。このため、Ni含有量は55~70%とすることが必要である。より好ましいNi含有量の範囲は58%を超えて65%以下である。さらに好ましいNi含有量の範囲は60%を超えて65%以下である。
Crは、合金の耐食性を維持するために必要な元素であり、要求される耐食性を確保するためには、そのCrの含有量を26%超とする必要がある。一方、その含有量が35%を超えると、熱間押出し加工性が著しく悪化する。このため、Cr含有量は、26%を超えて35%以下とすることが必要である。好ましくは27%超えて32%以下、より好ましくは28~31%である。
Alは、前記Siと同様に、脱酸剤として作用する元素であり、0.005%以上の含有が必要である。一方、その含有量が0.5%を超えると、合金の清浄度を低下させるので、Alの含有量は0.5%以下とした。より好ましくは0.02~0.3%である。
NはCとともに、TiまたはNbの炭窒化物を形成して合金の強度を高めるだけでなく、本発明では二次溶解法によるN,C,Ti,Nbの偏析抑制効果と併せることで、それらの炭窒化物を均一に分散析出させ、熱間押出し加工後の組織を細粒化することができる。その効果を得るには、0.02%以上含有させる必要がある。一方、0.10%を超えると窒化物が増えすぎて逆に熱間押出し加工性や延性を劣化させる。このため、N含有量は0.02~0.10%とした。より好ましくは0.03~0.06%である。
Tiは、炭窒化物を形成して合金の強度を高め、熱間押出し加工性を向上させる作用がある。これらの効果を得るには、Tiを0.01%以上含有させる必要がある。一方、Tiの含有量が0.5%を超えると、その効果が飽和するだけでなく金属間化合物の生成により延性を損なう。このため、Tiの含有量は0.01~0.5%とした。より好ましくは0.05~0.3%である。
Claims (3)
- [規則91に基づく訂正 07.10.2009]
質量%で、C:0.04%以下、Si:0.10~0.50%、Mn:0.05~0.50%、Ni:55~70%、Cr:26%超えて35%以下、Al:0.005~0.5%、N:0.02~0.10%、並びにTi:0.01~0.5%およびNb:0.02~1.0%のうちの一種以上を含有し、残部がFeおよび不純物からなり、結晶粒径がJIS G0551での粒度番号6またはそれ以上の細粒であることを特徴とする原子力用高強度Ni基合金管。 - Ni基合金素材が二次溶解法によって得られたものであることを特徴とする、請求項1に記載の原子力用高強度Ni基合金管。
- [規則91に基づく訂正 07.10.2009]
質量%で、C:0.04%以下、Si:0.10~0.50%、Mn:0.05~0.50%、Ni:55~70%、Cr:26%超えて35%以下、Al:0.005~0.5%、N:0.02~0.10%、およびTi:0.01~0.5%およびNb:0.02~1.0%のうちの一種以上を含有し、残部がFeおよび不純物からなり、二次溶解法によって得られたNi基合金素材を、熱間鍛造後、1000~1160℃に加熱した後に押出し比が4以上の加工度で熱間押出し加工し、さらに固溶化熱処理と時効処理を施すことを特徴とする原子力用高強度Ni基合金管の製造方法。
Priority Applications (7)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| ES09750590T ES2758825T3 (es) | 2008-05-22 | 2009-05-20 | Tubo de gran resistencia, basado en una aleación de Ni, para ser usado en plantas de energía nuclear y su proceso de producción |
| JP2009521268A JP4433230B2 (ja) | 2008-05-22 | 2009-05-20 | 原子力用高強度Ni基合金管及びその製造方法 |
| KR1020107026142A KR101181166B1 (ko) | 2008-05-22 | 2009-05-20 | 원자력용 고강도 Ni기 합금관 및 그 제조 방법 |
| CN2009801158560A CN102016090B (zh) | 2008-05-22 | 2009-05-20 | 原子能用高强度Ni基合金管及其制造方法 |
| EP09750590.3A EP2281908B1 (en) | 2008-05-22 | 2009-05-20 | High-strength ni-base alloy pipe for use in nuclear power plants and process for production thereof |
| CA2723526A CA2723526C (en) | 2008-05-22 | 2009-05-20 | High-strength ni-based alloy tube for nuclear power use and method for manufacturing the same |
| US12/993,838 US8246766B2 (en) | 2008-05-22 | 2010-11-20 | High-strength Ni-based alloy tube for nuclear power use and method for manufacturing the same |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2008-134549 | 2008-05-22 | ||
| JP2008134549 | 2008-05-22 |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US12/993,838 Continuation US8246766B2 (en) | 2008-05-22 | 2010-11-20 | High-strength Ni-based alloy tube for nuclear power use and method for manufacturing the same |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2009142228A1 true WO2009142228A1 (ja) | 2009-11-26 |
Family
ID=41340156
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2009/059249 Ceased WO2009142228A1 (ja) | 2008-05-22 | 2009-05-20 | 原子力用高強度Ni基合金管及びその製造方法 |
Country Status (8)
| Country | Link |
|---|---|
| US (1) | US8246766B2 (ja) |
| EP (1) | EP2281908B1 (ja) |
| JP (1) | JP4433230B2 (ja) |
| KR (1) | KR101181166B1 (ja) |
| CN (1) | CN102016090B (ja) |
| CA (1) | CA2723526C (ja) |
| ES (1) | ES2758825T3 (ja) |
| WO (1) | WO2009142228A1 (ja) |
Cited By (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2011162819A (ja) * | 2010-02-05 | 2011-08-25 | Mitsubishi Materials Corp | Ni基合金およびNi基合金の製造方法 |
| CN102463273A (zh) * | 2010-11-08 | 2012-05-23 | 北京有色金属研究总院 | 一种大口径镍基合金薄壁管材的制备方法 |
| CN102463272A (zh) * | 2010-11-08 | 2012-05-23 | 北京有色金属研究总院 | 一种小口径镍基合金薄壁管材的短流程制备方法 |
| WO2018061317A1 (ja) * | 2016-09-30 | 2018-04-05 | 日立金属株式会社 | Ni基超耐熱合金押出材の製造方法およびNi基超耐熱合金押出材 |
| KR101850966B1 (ko) | 2016-10-28 | 2018-04-20 | 한국생산기술연구원 | 니켈계 석출경화형 초내열합금의 열처리 방법 및 이에 의하여 구현된 고온 밸브용 초내열 스프링 |
| WO2018225831A1 (ja) | 2017-06-08 | 2018-12-13 | 新日鐵住金株式会社 | 原子力用Ni基合金管 |
| US10287655B2 (en) | 2011-06-01 | 2019-05-14 | Ati Properties Llc | Nickel-base alloy and articles |
| CN109789458A (zh) * | 2016-09-29 | 2019-05-21 | 日立金属株式会社 | Ni基超耐热合金的热挤出成型方法及Ni基超耐热合金挤出材的制造方法 |
| US10619226B2 (en) | 2015-01-12 | 2020-04-14 | Ati Properties Llc | Titanium alloy |
| CN116079347A (zh) * | 2023-02-17 | 2023-05-09 | 河北亚都管道装备集团有限公司 | 镍基合金大口径厚壁无缝热压弯头的制造方法及弯头 |
Families Citing this family (28)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20040221929A1 (en) | 2003-05-09 | 2004-11-11 | Hebda John J. | Processing of titanium-aluminum-vanadium alloys and products made thereby |
| US7837812B2 (en) | 2004-05-21 | 2010-11-23 | Ati Properties, Inc. | Metastable beta-titanium alloys and methods of processing the same by direct aging |
| US10053758B2 (en) | 2010-01-22 | 2018-08-21 | Ati Properties Llc | Production of high strength titanium |
| US9255316B2 (en) | 2010-07-19 | 2016-02-09 | Ati Properties, Inc. | Processing of α+β titanium alloys |
| US8499605B2 (en) | 2010-07-28 | 2013-08-06 | Ati Properties, Inc. | Hot stretch straightening of high strength α/β processed titanium |
| US8613818B2 (en) | 2010-09-15 | 2013-12-24 | Ati Properties, Inc. | Processing routes for titanium and titanium alloys |
| US9206497B2 (en) | 2010-09-15 | 2015-12-08 | Ati Properties, Inc. | Methods for processing titanium alloys |
| US10513755B2 (en) | 2010-09-23 | 2019-12-24 | Ati Properties Llc | High strength alpha/beta titanium alloy fasteners and fastener stock |
| RU2492958C2 (ru) * | 2011-08-17 | 2013-09-20 | Федеральное Государственное Унитарное Предприятие "Центральный Научно-Исследовательский Институт Конструкционных Материалов "Прометей" | Способ изготовления заготовки обечайки активной зоны корпуса реактора типа ввэр |
| CN103128129A (zh) * | 2011-11-24 | 2013-06-05 | 北京有色金属研究总院 | 一种Ni-Cr-Mo耐蚀合金管材的短流程制备方法 |
| CN102758096B (zh) * | 2012-08-08 | 2013-09-25 | 贵州航天新力铸锻有限责任公司 | 核电站流量限制器用镍基高温合金材料的制备方法 |
| US9050647B2 (en) | 2013-03-15 | 2015-06-09 | Ati Properties, Inc. | Split-pass open-die forging for hard-to-forge, strain-path sensitive titanium-base and nickel-base alloys |
| US9869003B2 (en) | 2013-02-26 | 2018-01-16 | Ati Properties Llc | Methods for processing alloys |
| US9192981B2 (en) | 2013-03-11 | 2015-11-24 | Ati Properties, Inc. | Thermomechanical processing of high strength non-magnetic corrosion resistant material |
| US9777361B2 (en) | 2013-03-15 | 2017-10-03 | Ati Properties Llc | Thermomechanical processing of alpha-beta titanium alloys |
| CN103286154B (zh) * | 2013-06-30 | 2014-12-24 | 西安诺博尔稀贵金属材料有限公司 | 一种gh3600镍合金挤压管材的制备方法 |
| CN103556003A (zh) * | 2013-09-27 | 2014-02-05 | 贵州航天新力铸锻有限责任公司 | 核电站设备零部件用的镍基合金的制备方法 |
| US11111552B2 (en) | 2013-11-12 | 2021-09-07 | Ati Properties Llc | Methods for processing metal alloys |
| CN103882266B (zh) * | 2014-03-26 | 2016-01-20 | 中国科学院上海应用物理研究所 | 用于熔盐反应堆的镍基合金及其制备方法 |
| ES2761273T3 (es) * | 2015-06-26 | 2020-05-19 | Nippon Steel Corp | Tubo de aleación a base de Ni para energía atómica |
| US10502252B2 (en) | 2015-11-23 | 2019-12-10 | Ati Properties Llc | Processing of alpha-beta titanium alloys |
| CN105779698B (zh) * | 2016-03-17 | 2018-01-09 | 周海彬 | 含铌的铬镍铁合金的冶炼方法 |
| CN107322003B (zh) * | 2017-06-29 | 2019-07-09 | 厦门朋鹭金属工业有限公司 | 一种制备低杂质含量钨粉的工业方法 |
| CN109321781A (zh) * | 2018-10-09 | 2019-02-12 | 北京金达太钢石化配件有限公司 | 一种新的镍基合金及其制备方法 |
| CN114309131A (zh) * | 2021-12-28 | 2022-04-12 | 江阴市恒业锻造有限公司 | 一种均匀细晶镍基合金n08825大型厚壁管坯锻件的制造方法 |
| CN115228964B (zh) * | 2022-06-15 | 2024-03-26 | 江苏银环精密钢管有限公司 | 核反应堆压力容器密封圈用镍基合金小口径管的制造方法 |
| US12344918B2 (en) | 2023-07-12 | 2025-07-01 | Ati Properties Llc | Titanium alloys |
| CN119406965B (zh) * | 2024-12-11 | 2025-09-02 | 江苏银环精密钢管有限公司 | 一种uns n06690合金无缝换热管的制造方法 |
Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5867854A (ja) | 1981-10-16 | 1983-04-22 | Sumitomo Metal Ind Ltd | 耐応力腐食割れ性にすぐれたニツケル基高クロム合金の製造方法 |
| JPS60245773A (ja) | 1984-05-18 | 1985-12-05 | Sumitomo Metal Ind Ltd | 高耐食性Ni基合金の製造方法 |
| JPS6169938A (ja) | 1984-09-14 | 1986-04-10 | Sumitomo Metal Ind Ltd | 耐粒界損傷性Ni基合金及びその製造方法 |
| JPS62167836A (ja) | 1986-01-20 | 1987-07-24 | Mitsubishi Heavy Ind Ltd | Ni基合金及びその製造法 |
| JPH01132731A (ja) | 1988-05-20 | 1989-05-25 | Nippon Yakin Kogyo Co Ltd | 溶接部高温熱影響部における耐粒界腐食性,耐応力腐食割れ性および機械的強度に優れるNi基合金 |
| JPH04198444A (ja) * | 1990-11-29 | 1992-07-17 | Agency Of Ind Science & Technol | 耐応力腐食割れ性に優れたNi基合金 |
| JP2004218076A (ja) | 2002-12-25 | 2004-08-05 | Sumitomo Metal Ind Ltd | ニッケル基合金およびその製造方法 |
| JP2007224372A (ja) * | 2006-02-24 | 2007-09-06 | Sumitomo Metal Ind Ltd | 含Crニッケル基合金管の製造方法 |
Family Cites Families (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS58177444A (ja) * | 1982-04-12 | 1983-10-18 | Sumitomo Metal Ind Ltd | Ni−Cr合金の熱処理法 |
| JPS59211545A (ja) * | 1983-05-18 | 1984-11-30 | Sumitomo Metal Ind Ltd | 耐応力腐食割れ性の優れたニツケル基合金 |
| US5378427A (en) * | 1991-03-13 | 1995-01-03 | Sumitomo Metal Industries, Ltd. | Corrosion-resistant alloy heat transfer tubes for heat-recovery boilers |
| JPH0674475A (ja) | 1992-08-31 | 1994-03-15 | Matsushita Electric Ind Co Ltd | 面状暖房装置 |
| EP1325965B1 (en) * | 2001-12-21 | 2005-10-05 | Hitachi Metals, Ltd. | Ni-based alloy improved in oxidation-resistance, high temperature strength and hot workability |
| JP2004137549A (ja) * | 2002-10-17 | 2004-05-13 | Sumitomo Metal Ind Ltd | Ni−Cr合金 |
| WO2006003954A1 (ja) * | 2004-06-30 | 2006-01-12 | Sumitomo Metal Industries, Ltd. | Ni基合金素管及びその製造方法 |
-
2009
- 2009-05-20 ES ES09750590T patent/ES2758825T3/es active Active
- 2009-05-20 EP EP09750590.3A patent/EP2281908B1/en active Active
- 2009-05-20 KR KR1020107026142A patent/KR101181166B1/ko active Active
- 2009-05-20 CN CN2009801158560A patent/CN102016090B/zh active Active
- 2009-05-20 JP JP2009521268A patent/JP4433230B2/ja active Active
- 2009-05-20 CA CA2723526A patent/CA2723526C/en active Active
- 2009-05-20 WO PCT/JP2009/059249 patent/WO2009142228A1/ja not_active Ceased
-
2010
- 2010-11-20 US US12/993,838 patent/US8246766B2/en active Active
Patent Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5867854A (ja) | 1981-10-16 | 1983-04-22 | Sumitomo Metal Ind Ltd | 耐応力腐食割れ性にすぐれたニツケル基高クロム合金の製造方法 |
| JPS60245773A (ja) | 1984-05-18 | 1985-12-05 | Sumitomo Metal Ind Ltd | 高耐食性Ni基合金の製造方法 |
| JPS6169938A (ja) | 1984-09-14 | 1986-04-10 | Sumitomo Metal Ind Ltd | 耐粒界損傷性Ni基合金及びその製造方法 |
| JPS62167836A (ja) | 1986-01-20 | 1987-07-24 | Mitsubishi Heavy Ind Ltd | Ni基合金及びその製造法 |
| JPH01132731A (ja) | 1988-05-20 | 1989-05-25 | Nippon Yakin Kogyo Co Ltd | 溶接部高温熱影響部における耐粒界腐食性,耐応力腐食割れ性および機械的強度に優れるNi基合金 |
| JPH04198444A (ja) * | 1990-11-29 | 1992-07-17 | Agency Of Ind Science & Technol | 耐応力腐食割れ性に優れたNi基合金 |
| JP2004218076A (ja) | 2002-12-25 | 2004-08-05 | Sumitomo Metal Ind Ltd | ニッケル基合金およびその製造方法 |
| JP2007224372A (ja) * | 2006-02-24 | 2007-09-06 | Sumitomo Metal Ind Ltd | 含Crニッケル基合金管の製造方法 |
Cited By (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2011162819A (ja) * | 2010-02-05 | 2011-08-25 | Mitsubishi Materials Corp | Ni基合金およびNi基合金の製造方法 |
| CN102463273A (zh) * | 2010-11-08 | 2012-05-23 | 北京有色金属研究总院 | 一种大口径镍基合金薄壁管材的制备方法 |
| CN102463272A (zh) * | 2010-11-08 | 2012-05-23 | 北京有色金属研究总院 | 一种小口径镍基合金薄壁管材的短流程制备方法 |
| US10287655B2 (en) | 2011-06-01 | 2019-05-14 | Ati Properties Llc | Nickel-base alloy and articles |
| US10808298B2 (en) | 2015-01-12 | 2020-10-20 | Ati Properties Llc | Titanium alloy |
| US10619226B2 (en) | 2015-01-12 | 2020-04-14 | Ati Properties Llc | Titanium alloy |
| CN109789458A (zh) * | 2016-09-29 | 2019-05-21 | 日立金属株式会社 | Ni基超耐热合金的热挤出成型方法及Ni基超耐热合金挤出材的制造方法 |
| JPWO2018061317A1 (ja) * | 2016-09-30 | 2019-04-11 | 日立金属株式会社 | Ni基超耐熱合金押出材の製造方法およびNi基超耐熱合金押出材 |
| WO2018061317A1 (ja) * | 2016-09-30 | 2018-04-05 | 日立金属株式会社 | Ni基超耐熱合金押出材の製造方法およびNi基超耐熱合金押出材 |
| KR101850966B1 (ko) | 2016-10-28 | 2018-04-20 | 한국생산기술연구원 | 니켈계 석출경화형 초내열합금의 열처리 방법 및 이에 의하여 구현된 고온 밸브용 초내열 스프링 |
| WO2018225831A1 (ja) | 2017-06-08 | 2018-12-13 | 新日鐵住金株式会社 | 原子力用Ni基合金管 |
| KR20200016333A (ko) | 2017-06-08 | 2020-02-14 | 닛폰세이테츠 가부시키가이샤 | 원자력용 Ni기 합금관 |
| JPWO2018225831A1 (ja) * | 2017-06-08 | 2020-03-19 | 日本製鉄株式会社 | 原子力用Ni基合金管 |
| US11215356B2 (en) | 2017-06-08 | 2022-01-04 | Nippon Steel Corporation | Ni-based alloy pipe for nuclear power |
| CN116079347A (zh) * | 2023-02-17 | 2023-05-09 | 河北亚都管道装备集团有限公司 | 镍基合金大口径厚壁无缝热压弯头的制造方法及弯头 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP2281908A1 (en) | 2011-02-09 |
| JPWO2009142228A1 (ja) | 2011-09-29 |
| KR20100135304A (ko) | 2010-12-24 |
| EP2281908A4 (en) | 2017-07-19 |
| CA2723526C (en) | 2013-07-23 |
| JP4433230B2 (ja) | 2010-03-17 |
| CA2723526A1 (en) | 2009-11-26 |
| ES2758825T3 (es) | 2020-05-06 |
| CN102016090A (zh) | 2011-04-13 |
| US8246766B2 (en) | 2012-08-21 |
| EP2281908B1 (en) | 2019-10-23 |
| KR101181166B1 (ko) | 2012-09-18 |
| US20110183151A1 (en) | 2011-07-28 |
| CN102016090B (zh) | 2012-09-26 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP4433230B2 (ja) | 原子力用高強度Ni基合金管及びその製造方法 | |
| CN102066594B (zh) | 奥氏体系耐热合金以及由该合金构成的耐热耐压构件及其制造方法 | |
| CN104946932B (zh) | 奥氏体系耐热合金管的制造方法以及利用该制造方法制造的奥氏体系耐热合金管 | |
| JP6620475B2 (ja) | Ni基耐熱合金管の製造方法 | |
| JP6816779B2 (ja) | オーステナイト系耐熱合金部材およびその製造方法 | |
| JP6492747B2 (ja) | オーステナイト系耐熱合金管の製造方法およびその製造方法によって製造されたオーステナイト系耐熱合金管 | |
| JP6477252B2 (ja) | オーステナイト系耐熱合金および耐熱耐圧部材 | |
| WO2010093016A1 (ja) | チタン板 | |
| JP7167707B2 (ja) | オーステナイト系耐熱鋼 | |
| WO2019043882A1 (ja) | チタン板 | |
| JP2017036477A (ja) | オーステナイト系耐熱合金部材およびその製造方法 | |
| JP6822563B2 (ja) | 原子力用Ni基合金管 | |
| JP2021105204A (ja) | オーステナイト系耐熱鋼 | |
| EP3693487A1 (en) | Austenitic stainless steel | |
| CN109844148B (zh) | 镍材及镍材的制造方法 | |
| JP2017179478A (ja) | オーステナイト系耐熱合金部材およびその製造方法 | |
| JP5382518B2 (ja) | チタン材 | |
| TWI675921B (zh) | 鎳基沃斯田鐵系合金及其製造方法 | |
| WO2008047869A1 (fr) | Matériau de nickel pour installation chimique | |
| TWI612143B (zh) | 析出強化型鎳基合金及其製造方法 | |
| JP2020164919A (ja) | オーステナイト系耐熱鋼 | |
| JP2013100584A (ja) | 熱間押出性に優れたNi基合金管及びNi基合金管の製造方法 | |
| WO2025205764A1 (ja) | Ni-Fe-Cr基合金管およびその製造方法 | |
| JPH0995748A (ja) | 高温延性および耐焼鈍脆性に優れた耐食銅合金 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 2009521268 Country of ref document: JP Ref document number: 200980115856.0 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 09750590 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2723526 Country of ref document: CA |
|
| ENP | Entry into the national phase |
Ref document number: 20107026142 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2009750590 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |