WO2012165488A1 - Procédé de lavage/tri et dispositif de lavage/tri pour des matières mixtes - Google Patents
Procédé de lavage/tri et dispositif de lavage/tri pour des matières mixtes Download PDFInfo
- Publication number
- WO2012165488A1 WO2012165488A1 PCT/JP2012/063951 JP2012063951W WO2012165488A1 WO 2012165488 A1 WO2012165488 A1 WO 2012165488A1 JP 2012063951 W JP2012063951 W JP 2012063951W WO 2012165488 A1 WO2012165488 A1 WO 2012165488A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- cleaning
- liquid
- separation
- specific gravity
- separating
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Images
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D61/00—Processes of separation using semi-permeable membranes, e.g. dialysis, osmosis or ultrafiltration; Apparatus, accessories or auxiliary operations specially adapted therefor
- B01D61/14—Ultrafiltration; Microfiltration
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D61/00—Processes of separation using semi-permeable membranes, e.g. dialysis, osmosis or ultrafiltration; Apparatus, accessories or auxiliary operations specially adapted therefor
- B01D61/02—Reverse osmosis; Hyperfiltration ; Nanofiltration
- B01D61/025—Reverse osmosis; Hyperfiltration
- B01D61/026—Reverse osmosis; Hyperfiltration comprising multiple reverse osmosis steps
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D61/00—Processes of separation using semi-permeable membranes, e.g. dialysis, osmosis or ultrafiltration; Apparatus, accessories or auxiliary operations specially adapted therefor
- B01D61/58—Multistep processes
-
- C—CHEMISTRY; METALLURGY
- C02—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F1/00—Treatment of water, waste water, or sewage
- C02F1/001—Processes for the treatment of water whereby the filtration technique is of importance
- C02F1/004—Processes for the treatment of water whereby the filtration technique is of importance using large scale industrial sized filters
-
- C—CHEMISTRY; METALLURGY
- C02—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F1/00—Treatment of water, waste water, or sewage
- C02F1/24—Treatment of water, waste water, or sewage by flotation
-
- C—CHEMISTRY; METALLURGY
- C02—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F1/00—Treatment of water, waste water, or sewage
- C02F1/28—Treatment of water, waste water, or sewage by sorption
-
- C—CHEMISTRY; METALLURGY
- C02—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F1/00—Treatment of water, waste water, or sewage
- C02F1/42—Treatment of water, waste water, or sewage by ion-exchange
-
- C—CHEMISTRY; METALLURGY
- C02—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F1/00—Treatment of water, waste water, or sewage
- C02F1/44—Treatment of water, waste water, or sewage by dialysis, osmosis or reverse osmosis
- C02F1/444—Treatment of water, waste water, or sewage by dialysis, osmosis or reverse osmosis by ultrafiltration or microfiltration
-
- C—CHEMISTRY; METALLURGY
- C02—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F1/00—Treatment of water, waste water, or sewage
- C02F1/52—Treatment of water, waste water, or sewage by flocculation or precipitation of suspended impurities
-
- G—PHYSICS
- G21—NUCLEAR PHYSICS; NUCLEAR ENGINEERING
- G21F—PROTECTION AGAINST X-RADIATION, GAMMA RADIATION, CORPUSCULAR RADIATION OR PARTICLE BOMBARDMENT; TREATING RADIOACTIVELY CONTAMINATED MATERIAL; DECONTAMINATION ARRANGEMENTS THEREFOR
- G21F9/00—Treating radioactively contaminated material; Decontamination arrangements therefor
- G21F9/04—Treating liquids
- G21F9/06—Processing
- G21F9/12—Processing by absorption; by adsorption; by ion-exchange
-
- G—PHYSICS
- G21—NUCLEAR PHYSICS; NUCLEAR ENGINEERING
- G21F—PROTECTION AGAINST X-RADIATION, GAMMA RADIATION, CORPUSCULAR RADIATION OR PARTICLE BOMBARDMENT; TREATING RADIOACTIVELY CONTAMINATED MATERIAL; DECONTAMINATION ARRANGEMENTS THEREFOR
- G21F9/00—Treating radioactively contaminated material; Decontamination arrangements therefor
- G21F9/28—Treating solids
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2317/00—Membrane module arrangements within a plant or an apparatus
- B01D2317/02—Elements in series
- B01D2317/022—Reject series
-
- C—CHEMISTRY; METALLURGY
- C02—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F1/00—Treatment of water, waste water, or sewage
- C02F1/28—Treatment of water, waste water, or sewage by sorption
- C02F1/281—Treatment of water, waste water, or sewage by sorption using inorganic sorbents
-
- C—CHEMISTRY; METALLURGY
- C02—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F1/00—Treatment of water, waste water, or sewage
- C02F1/28—Treatment of water, waste water, or sewage by sorption
- C02F1/283—Treatment of water, waste water, or sewage by sorption using coal, charred products, or inorganic mixtures containing them
-
- C—CHEMISTRY; METALLURGY
- C02—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F1/00—Treatment of water, waste water, or sewage
- C02F2001/007—Processes including a sedimentation step
-
- C—CHEMISTRY; METALLURGY
- C02—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F2101/00—Nature of the contaminant
- C02F2101/006—Radioactive compounds
-
- C—CHEMISTRY; METALLURGY
- C02—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F2101/00—Nature of the contaminant
- C02F2101/30—Organic compounds
- C02F2101/301—Detergents, surfactants
-
- C—CHEMISTRY; METALLURGY
- C02—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F2101/00—Nature of the contaminant
- C02F2101/30—Organic compounds
- C02F2101/32—Hydrocarbons, e.g. oil
-
- C—CHEMISTRY; METALLURGY
- C02—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F2103/00—Nature of the water, waste water, sewage or sludge to be treated
- C02F2103/34—Nature of the water, waste water, sewage or sludge to be treated from industrial activities not provided for in groups C02F2103/12 - C02F2103/32
-
- C—CHEMISTRY; METALLURGY
- C02—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F2209/00—Controlling or monitoring parameters in water treatment
- C02F2209/06—Controlling or monitoring parameters in water treatment pH
-
- C—CHEMISTRY; METALLURGY
- C02—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F2301/00—General aspects of water treatment
- C02F2301/04—Flow arrangements
- C02F2301/046—Recirculation with an external loop
-
- C—CHEMISTRY; METALLURGY
- C02—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F2301/00—General aspects of water treatment
- C02F2301/08—Multistage treatments, e.g. repetition of the same process step under different conditions
-
- C—CHEMISTRY; METALLURGY
- C02—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F2303/00—Specific treatment goals
- C02F2303/22—Eliminating or preventing deposits, scale removal, scale prevention
-
- C—CHEMISTRY; METALLURGY
- C02—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F5/00—Softening water; Preventing scale; Adding scale preventatives or scale removers to water, e.g. adding sequestering agents
- C02F5/08—Treatment of water with complexing chemicals or other solubilising agents for softening, scale prevention or scale removal, e.g. adding sequestering agents
Definitions
- the present invention relates to a cleaning separation method and a cleaning separation apparatus for mixed materials composed of a plurality of types of materials including waste materials such as rubble, and in particular, salts and harmful substances are removed from mixed materials containing salts and harmful substances.
- the present invention relates to a method and an apparatus for cleaning and removing the mixed material.
- waste disposal has become very important.
- industrial waste discharged from factories, construction and demolition sites contains plastic materials in addition to conventional wood and concrete, and recycling of these waste materials (waste) is important. .
- Non-Patent Document 1 A method of treating the leachate by guiding it to one place is used (for example, see Non-Patent Document 1).
- Patent Document 1 A method of forcibly purifying with washing water in order to accelerate leakage of harmful substances from waste materials (for example, see Patent Document 1), and a method of regenerating treated water from washing waste water or leachate and recycling it as washing water. It has been proposed (see, for example, Patent Document 2).
- the waste material contains only salt, there are many cases where leaching does not adversely affect the natural world.
- thermal recycling that is, direct incineration or RDF (Refuse Derived Fuel, waste solid fuel) conversion.
- RDF Refuse Derived Fuel, waste solid fuel
- the waste is dispersed in a separation liquid such as water, and the waste is separated by the difference in specific gravity.
- a separation liquid such as water
- the waste is separated by the difference in specific gravity.
- This method is widely used because it can be easily separated due to the specific gravity difference inherent to the material, but the solvent used for separation also has an effect of leaching out salts and soluble organic substances.
- This makes it possible to remove salts (especially chloride salts) and harmful substances that cause problems in furnace corrosion and generation of harmful gases during thermal recycling.
- the concentration of the salt or soluble organic substance in the separation liquid gradually increases, and the leaching cleaning efficiency gradually decreases.
- a method of injecting and diluting the separation liquid can be employed, but in that case, it is necessary to drain an amount corresponding to the injection. That is, increasing the injection amount of the separation liquid is not preferable because not only the necessary separation liquid amount increases but also the drainage amount increases. On the other hand, reducing the injection amount of the separation liquid increases the risk of reaching a concentration that is difficult to release into the environment due to an increase in the concentration of salts and soluble organic substances in the separation liquid. Become.
- Patent Document 4 since the salt and soluble organic matter in the separated liquid that has been leached remain in the separated waste, there is a considerable amount of waste, and in the recovery and reuse of the waste material, as illustrated in Patent Document 4, the cleaning liquid It is preferable to wash the waste material. However, the method of Patent Document 4 still requires a cleaning liquid, so that there remains a problem that the amount of waste liquid increases.
- Japanese Unexamined Patent Publication No. 11-179316 Japanese Unexamined Patent Publication No. 2001-79508 Japanese Unexamined Patent Publication No. 56-56246 Japanese Patent Laid-Open No. 11-76983 Japanese Unexamined Patent Publication No. 2006-21956
- the method of carbonizing once and then separating the reverse osmosis membrane can carbonize harmful substances and oils and surfactants that damage the reverse osmosis membrane.
- the amount of carbonization that can be carbonized is limited due to problems in equipment, and RDF conversion only confines harmful substances in the carbide, and the harmful substances contained in the carbide are harmless. Therefore, there is a risk of causing harm as a by-product when carbide is incinerated in a furnace as RDF.
- An object of the present invention is to separate salts and harmful substances from mixed waste materials and to efficiently wash the waste materials so that the waste materials can be safely disposed or recycled.
- it adheres to a large amount of wood and plastics, and in thermal recycling, it efficiently cleans and removes salt and harmful substances that cause damage to the furnace. It is to release safely and reuse.
- the present invention has the following configuration. (1) A separation step of putting a mixed material composed of a low specific gravity material and a high specific gravity material into a separation liquid tank and separating the mixture into a low specific gravity material that floats due to a specific gravity difference from the separation liquid and a high specific gravity material that settles; A cleaning step of cleaning the mixed material with a cleaning liquid before the separation step, or a cleaning step of cleaning at least one of the low specific gravity material and the high specific gravity material with a cleaning liquid after the separation step; and A method for cleaning and separating a mixed material, the method including a supply step of supplying cleaning waste liquid from the cleaning step to the separation liquid tank.
- the at least one additive selected from the group consisting of an aggregating agent, an adsorbent and an ion exchanger is added to the washing / separating effluent or the treatment liquid of the washing / separating effluent.
- At least one selected from the group consisting of flotation or sedimentation treatment, sand filtration, microfiltration, and ultrafiltration after adding the additive to the washing separation waste liquid or the treatment liquid of the washing separation waste liquid The method for cleaning and separating a mixed material according to (8), wherein one solid-liquid separation process and a separation process using a semipermeable membrane are sequentially performed. (10) The concentrated liquid is separated into a second permeated liquid and a second concentrated liquid by a second semipermeable membrane, and the second permeated liquid is reused as the cleaning liquid. 9. The method for washing and separating mixed materials according to any one of 9).
- a separation liquid tank that separates a mixed material composed of a low specific gravity material and a high specific gravity material into a low specific gravity material that floats due to a specific gravity difference from the separation liquid and a high specific gravity material that settles;
- a mixed material cleaning / separating device comprising: a supply line for supplying the cleaning waste liquid collected from the cleaning section to the separation liquid tank.
- a cleaning / separating drainage line that discharges at least a part of the cleaning drainage supplied to the separation liquid tank as cleaning / separating drainage.
- a semipermeable membrane that separates the washing separation drainage liquid into a permeate and a concentrate, and a discharge line that discharges the permeate to a river or the sea, or a reflux line that recirculates to the cleaning liquid.
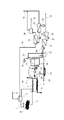
- FIG. 1 is a schematic flow diagram showing a first embodiment of a method for cleaning and separating mixed materials according to the present invention.
- FIG. 2 is a schematic flow diagram showing a second embodiment of the mixed material cleaning and separating method according to the present invention.
- FIG. 3 is a schematic flow diagram showing another aspect of the second embodiment of the method for cleaning and separating mixed materials according to the present invention.
- FIG. 1 shows a first embodiment of a cleaning / separating apparatus for separately cleaning a mixed material according to the present invention.
- the mixed material 8 is put into the separation liquid tank 7 and is separated into a low specific gravity material 3 that floats due to a specific gravity difference from the separation liquid and a high specific gravity material 9 that settles (separation step).
- the cleaning liquid 1 is supplied to the cleaning liquid tank 2, and the low specific gravity material 3 floated and separated in the cleaning section is cleaned with the cleaning liquid 1 sent through the cleaning liquid line 4, and the high specific gravity material 9 is a high specific gravity material.
- the cleaning drainage liquid after cleaning the low specific gravity material 3 or the high specific gravity material 9 is sent to the separation liquid tank 7 through the cleaning drainage line 5 (supply process), and as a separation liquid, It is subjected to specific gravity separation.
- the separation liquid in the cleaning liquid 1 and the separation liquid tank 7 is generally water or an aqueous solution.
- a specific gravity separation threshold value when a specific gravity separation threshold value is to be changed, a hydrocarbon solvent, alcohol, or a mixture thereof.
- water it is preferable to use natural water such as rain water, river water, ground water, etc., because costs can be reduced, but tap water can also be used.
- impurities such as turbidity are contained, turbidity, etc. It is also possible to apply the process.
- rainwater When rainwater is used for the cleaning liquid 1, rainwater can be collected in the cleaning liquid tank 2 using a rainwater collecting / collecting unit (not shown), or it can be directly mixed with rainwater without the cleaning liquid tank 2 or the cleaning liquid line 4. 8 can be washed, but in order to keep the amount of the washing liquid stable, it is preferable to provide the washing liquid line 2 from the washing liquid tank 2 or river or ground water.
- the separation liquid from which the suspended solids and the like are removed by specific gravity separation in the separation liquid tank 7 is appropriately discharged out of the separation liquid tank as a washing separation waste liquid in order to keep the amount constant. If there is no problem in discharging into the environment, it can be released as it is, but if the concentration of harmful substances exceeds the emission regulations, it must be treated.
- Such a treatment method is not particularly limited, but a separation treatment into a permeate and a concentrated solution by a semipermeable membrane is preferable both when the permeate is discharged and when the washing separation drainage is reused.
- reuse as the cleaning liquid in the present invention is preferable because the supply amount of the cleaning liquid can be reduced.
- the washing / separation drainage discharged from the separation liquid tank 7 through the washing / separation drainage line 37 is removed by the screen 10 after removing relatively large suspended matters that have overflowed without being settled, and then the agitation tank 14 by the supply pump 11. Sent to.
- At least one additive 13 selected from the group consisting of a flocculant, an adsorbent, and an ion exchanger is added, and in the washing separation drainage (that is, the liquid after having been separated by specific gravity using the washing drainage). Adsorbs salts, harmful substances, oils and surfactants dispersed or dissolved in Thereafter, the flocculant and the adsorbent are separated by a solid-liquid separation operation. Specifically, depending on the drainage properties, after flotation or sedimentation separation solid-liquid separation, at least one solid-liquid separation selected from the group consisting of sand filtration, microfiltration and ultrafiltration ensures reliable flotation and sedimentation separation. The fine particles that cannot be removed can be efficiently separated.
- the quality of the treated water is improved and contamination of the subsequent semipermeable membrane can be prevented.
- flotation separation is suitable, but in the case of flotation separation, pressurized flotation separation that introduces pressurized microbubbles is effective.
- sand filtration it is possible to apply gravity-type filtration that naturally flows down, and it is also possible to apply pressure-type filtration in which a pressure tank is filled with sand.
- sand to be filled single-component sand can be applied. For example, anthracite, silica sand, garnet, pumice, and the like can be combined to increase filtration efficiency.
- the microfiltration membrane and the ultrafiltration membrane are not particularly limited, and a flat membrane, a hollow fiber membrane, a tubular membrane, a pleat type, or any other shape can be used as appropriate.
- the material of the membrane is not particularly limited, and it is possible to use an inorganic material such as polyacrylonitrile, polyphenylene sulfone, polyphenylene sulfide sulfone, polyvinylidene fluoride, polypropylene, polyethylene, polysulfone, polyvinyl alcohol, cellulose acetate, or ceramic.
- an inorganic material such as polyacrylonitrile, polyphenylene sulfone, polyphenylene sulfide sulfone, polyvinylidene fluoride, polypropylene, polyethylene, polysulfone, polyvinyl alcohol, cellulose acetate, or ceramic.
- transmission side are applicable.
- the flocculant, adsorbent, and ion exchanger that can be charged into the agitation tank 14 are not particularly limited, and can be appropriately selected depending on the components of the washing waste water.
- inorganic flocculants such as polyaluminum chloride and ferric chloride are preferable because they are inexpensive and high removal efficiency can be obtained by using polymer flocculants, activated carbon, and ion exchangers. Can do.
- a plastic carrier is preferable because of its high removal performance, and in the case of a radioactive substance, zeolite and ferrocyanide are preferable because they selectively adsorb the radioactive substance.
- the water subjected to the pressure levitation treatment is stored in the pressure levitation treatment water tank 19, sent to the filtration unit 21 using the pressure pump 20, and processed by the filtration unit 21.
- the filtration unit 21 is preferably selected from the group consisting of sand filtration, filter cloth, microfiltration and ultrafiltration. However, when ultrafiltration with the best water quality is used, the latter semipermeable membrane unit is used. It is more preferable because contamination can be prevented. Although not shown, it is also preferable to increase the filtration efficiency in the filtration unit 21 by adding a flocculant or the like before the treatment in the filtration unit 21.
- the filtrate of the filtration unit 21 is stored in the filtrate tank 23, it is sent to the first semipermeable membrane unit 25 by the pressure pump 24 and separated into the permeate and the concentrate.
- the permeate of the first semipermeable membrane unit 25 is discharged out of the system (river, sea, etc.) as a final treated water 31 from which salt and harmful substances have been removed, or a reflux line is provided. It can be reused as the cleaning liquid 1.
- the concentrated liquid of the first semipermeable membrane unit 25 is pressurized by the booster pump 26 as needed, and then processed by the second semipermeable membrane unit 29, so that the second concentrated liquid and the second concentrated liquid Of permeate 30.
- the second permeate 30 is inferior in water quality to the permeate of the first semipermeable membrane unit 25, it is preferably reused as the cleaning liquid 1.
- the second concentrated liquid is preferably separated into a liquid that can be recovered and reused by the evaporation unit 28, etc., and a concentrated liquid or solid waste 35 and disposed of.
- the filtrate stored in the filtrate tank 23 may be discharged into the river or the sea as the discharge liquid 32 without being separated and reused by the semipermeable membrane unit 25, or by using the backwash pump 22. It may be processed again by the filtration unit 21.
- the mixed material 8 is not particularly limited, and examples thereof include those containing any of salts, harmful substances, oils, and surfactants, and the mixed material 8 containing these includes the method of the present invention. Can be effectively cleaned and removed.
- the salt is not particularly limited and may include alkali metals, alkaline earth metals, transition metal chloride salts, carbonates, sulfates, etc., and incinerators by reducing their concentrations. It is possible to prevent the deterioration of the battery and facilitate recycling.
- the chloride salt is converted to hydrogen chloride in the incinerator, and therefore it is highly necessary to remove it from the viewpoint of corrosion and toxic gas generation.
- alkali metal alkaline earth metal
- transition metal transition metal
- the cleaning liquid 1 used for this purpose is not particularly limited, and various waters such as rain water, river water, ground water, lake water, and tap water can be used. However, in view of cost performance and convenience, river water and lake water Can be used. Furthermore, since the amount of water taken in the cleaning liquid 1 can be reduced by reflux mixing the second permeate 30 as described above, it is preferable in terms of cost and environment.
- the cleaning liquid 1 it is preferable to use water having a salt concentration of 0.1% by weight or less as the cleaning liquid 1, but since the semipermeable membrane has a very high salt removal rate of 99% or more, the permeated solution obtained from the semipermeable membrane is a salt.
- the concentration is usually 0.05% by weight or less, and it is very preferable because the cleaning liquid 1 having high quality can be obtained by circulating and reusing the permeated liquid obtained by subjecting the mixed waste water to the semipermeable membrane treatment.
- the salt concentration referred to here is the total evaporation residue concentration of a generally defined dissolved component (component that has passed through 0.45 ⁇ m filter paper).
- the concentration of harmful substances, oil and surfactant contained in the mixed material 8 is appropriately reduced according to the regulations.
- harmful substances include radioactive substances, poisonous substances, and pathogens, but the substances are not limited to these, and any substances that are regulated according to the location and application may be used.
- Typical harmful radioactive substances include, for example, strontium, iodine, cesium, plutonium and the like.
- Toxic substances include poisonous substances specified by the Deleterious Substances Control Law and government ordinances, various insecticides and herbicides. Drugs are also included.
- pathogens include pathogenic E. coli, designated infectious pathogens, viruses, nematodes, roundworms, and the like.
- the semipermeable membrane unit applicable to the present invention is not particularly limited, but for easy handling, a hollow fiber membrane-like or flat membrane-like semipermeable membrane is housed in a casing to form a fluid separation element (element). It is preferable to use what was loaded in a pressure vessel.
- the fluid separation element is formed of a flat membrane, for example, generally a semipermeable membrane is wound in a cylindrical shape together with a flow path material (net) around a cylindrical central pipe having a large number of holes.
- TM700 series and TM800 series manufactured by Toray Industries, Inc. can be mentioned.
- These fluid separation elements may be one, and it is also preferable to configure a semipermeable membrane unit by connecting a plurality of fluid separation elements in series or in parallel.
- Polymer materials such as cellulose acetate polymer, polyamide, polyester, polyimide, vinyl polymer can be used for the semipermeable membrane material.
- the membrane structure has a dense layer on at least one side of the membrane, and on the asymmetric membrane having fine pores gradually increasing from the dense layer to the inside of the membrane or the other side, or on the dense layer of the asymmetric membrane. Any composite film having a very thin functional layer formed of another material may be used.
- a scale inhibitor, acid, or alkali is added to the supply liquid of each semipermeable membrane unit to prevent scale precipitation due to concentration or to adjust pH. It is possible to In addition, it is preferable to implement addition of a scale inhibitor upstream from pH adjustment so that the addition effect can be exhibited.
- an in-line mixer may be provided immediately after the addition of chemicals, or the addition port may be in direct contact with the flow of the supply liquid to prevent sudden changes in concentration and pH near the addition port. preferable.
- the scale inhibitor is a compound that forms a complex with a metal, metal ion, or the like in a solution and solubilizes the metal or metal salt, and an organic or inorganic ionic polymer or monomer can be used.
- Synthetic polymers such as polyacrylic acid, sulfonated polystyrene, polyacrylamide, and polyallylamine can be used as organic polymers, and natural polymers such as carboxymethylcellulose, chitosan, and alginic acid. Ethylenediaminetetraacetic acid and the like can be used as organic monomers. Can be used.
- polyphosphate etc. can be used as an inorganic type scale inhibitor.
- polyphosphate and ethylenediaminetetraacetic acid are particularly preferably used from the viewpoints of availability, ease of operation such as solubility, and cost.
- the polyphosphate refers to a polymerized inorganic phosphate material having two or more phosphorus atoms in a molecule typified by sodium hexametaphosphate and bonded with an alkali metal, an alkaline earth metal and a phosphate atom.
- Typical polyphosphates include tetrasodium pyrophosphate, disodium pyrophosphate, sodium tripolyphosphate, sodium tetrapolyphosphate, sodium heptapolyphosphate, sodium decapolyphosphate, sodium metaphosphate, sodium hexametaphosphate, and potassium salts thereof. Etc.
- sulfuric acid, sodium hydroxide, and calcium hydroxide are generally used as the acid and alkali, but hydrochloric acid, oxalic acid, potassium hydroxide, sodium bicarbonate, ammonium hydroxide, and the like can also be used.
- hydrochloric acid, oxalic acid, potassium hydroxide, sodium bicarbonate, ammonium hydroxide, and the like can also be used.
- the cleaning liquid line 4 is provided so as to clean both the low specific gravity material 3 and the high specific gravity material 9, but the cleaning liquid line 4 is cleaned so that only one of them is cleaned. There is no problem. Further, all of the washing waste liquid may be supplied to the separation liquid tank 7 or a part thereof may be supplied. Generally, since the low specific gravity material 3 that is floated and separated is a material that can be recycled or incinerated (thermal recycle), the method of cleaning only the low specific gravity material 3 using the cleaning liquid line 4 is economically preferable. Furthermore, when the water quality of the separation liquid in the separation liquid tank 7 deteriorates, a part of the cleaning liquid 1 can be directly supplied to the separation liquid tank 7.
- the cleaning liquid 1 is supplied to the cleaning liquid tank 2 and the mixed material 8 is cleaned through the cleaning liquid line 4 in the cleaning section (cleaning process).
- the cleaning drainage liquid after cleaning the mixed material 8 with the cleaning liquid 1 is sent to the separation liquid tank 7 through the cleaning drainage line 5 (supply process).
- suspended substances and solids mixed by washing are separated into a low specific gravity material 3 and a high specific gravity material 9, and separated from the low specific gravity material take-out line 3 and the high specific gravity material take-out line 6 as necessary. It is removed (fractionation process).
- the washing / separating waste liquid discharged from the separation liquid tank 7 is subjected to the same processing as in the first embodiment through the washing / separating drainage line 37, purified, and reused.
- FIG. 3 is a schematic flow diagram showing another aspect of the second embodiment of the method for cleaning and separating mixed materials according to the present invention.
- the washing liquid tank 7 when the washing waste liquid stored in the separation liquid tank 7 after washing the mixed material 8 is heavily contaminated, the amount of washing liquid can be increased, but as shown in FIG. 3, the washing liquid tank It is also preferable to supply the cleaning liquid directly from 2 to the separation liquid tank 7 through the bypass line 36 to reduce the drainage treatment and regeneration load after cleaning and separation.
- the mixed material 8 is put into the separation liquid tank 7 and the specific gravity separation is performed, and then the low specific gravity material 3 and the high specific gravity material 9 are washed with the washing liquid. There is no problem.
- the mixed material 8 is preliminarily washed in the separation liquid tank 7, it is washed with an uncontaminated washing liquid, which is very efficient.
- the untreated mixed material 8 having a large contamination comes into contact with the most contaminated separation liquid, and the mixed material 8 is separated by specific gravity. However, it is more efficient because it flows toward the relatively uncontaminated separation liquid.
- the present invention relates to a cleaning method and apparatus for efficiently cleaning and removing salt and harmful substances from waste materials containing salts and harmful substances, and disposing of the waste materials and making them recyclable. Efficient cleaning is possible by separating and reusing the cleaning and separation effluent used to remove salts and harmful substances with a semipermeable membrane.
Landscapes
- Engineering & Computer Science (AREA)
- Water Supply & Treatment (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Hydrology & Water Resources (AREA)
- Environmental & Geological Engineering (AREA)
- Organic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Engineering & Computer Science (AREA)
- High Energy & Nuclear Physics (AREA)
- Physics & Mathematics (AREA)
- Nanotechnology (AREA)
- Separation Using Semi-Permeable Membranes (AREA)
- Separation Of Suspended Particles By Flocculating Agents (AREA)
- Treatment Of Water By Ion Exchange (AREA)
- Physical Water Treatments (AREA)
- Separation Of Solids By Using Liquids Or Pneumatic Power (AREA)
- Water Treatment By Sorption (AREA)
- Processing Of Solid Wastes (AREA)
Abstract
La présente invention a pour but de retirer efficacement, à partir de matières de rebut contenant du sel, des substances toxiques et similaires, le sel, les substances toxiques et similaires par lavage pour permettre ainsi l'élimination et le recyclage des matières de rebut. La présente invention porte sur un procédé de lavage/tri pour des matières mixtes (8), les matières mixtes (8) étant jetées dans un réservoir de liquide séparé (7) et triées en des matières en surface de faible masse volumique (3) et des matières qui coulent de masse volumique élevée (9) selon la différence en masse volumique à partir de la solution séparée, et les matières de faible masse volumique (3) et/ou les matières de masse volumique élevée (9) sont lavées par une solution de lavage (1) et un drainage de lavage est appliqué au réservoir de liquide séparé (7).
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2012538893A JP5904123B2 (ja) | 2011-05-31 | 2012-05-30 | 混合材料の洗浄分別方法および洗浄分別装置 |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2011-121596 | 2011-05-31 | ||
| JP2011121595 | 2011-05-31 | ||
| JP2011121596 | 2011-05-31 | ||
| JP2011-121595 | 2011-05-31 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2012165488A1 true WO2012165488A1 (fr) | 2012-12-06 |
Family
ID=47259340
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2012/063951 Ceased WO2012165488A1 (fr) | 2011-05-31 | 2012-05-30 | Procédé de lavage/tri et dispositif de lavage/tri pour des matières mixtes |
Country Status (2)
| Country | Link |
|---|---|
| JP (1) | JP5904123B2 (fr) |
| WO (1) | WO2012165488A1 (fr) |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2016050780A (ja) * | 2014-08-28 | 2016-04-11 | 株式会社東芝 | 排水処理装置および排水処理方法 |
| JPWO2015137484A1 (ja) * | 2014-03-14 | 2017-04-06 | 株式会社ピーシーエス | 超微細気泡含有液体を用いる超微細気泡洗浄方法、その装置及び加圧浮上装置 |
| JP2019209324A (ja) * | 2018-05-30 | 2019-12-12 | 大矢建設工業株式会社 | 排水処理システム及び排水処理方法 |
| CN115805216A (zh) * | 2022-12-29 | 2023-03-17 | 苏州赢众环保有限公司 | 一种一体化填料清洗设备 |
| CN121060926A (zh) * | 2025-11-07 | 2025-12-05 | 中国资源循环集团塑料再生有限公司 | 一种利用垃圾渗滤液清洗废塑料及回收腐殖土的耦合方法 |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2002254063A (ja) * | 2001-03-01 | 2002-09-10 | Konoike Constr Ltd | 汚染土壌の浄化方法及びそれに用いる分離装置 |
| JP2005081247A (ja) * | 2003-09-09 | 2005-03-31 | Mitsubishi Heavy Ind Ltd | 汚染土壌の浄化装置 |
| JP2005279476A (ja) * | 2004-03-30 | 2005-10-13 | Kentaro Asakura | 有害物質の処理方法 |
| JP2009136846A (ja) * | 2007-12-11 | 2009-06-25 | Kubota Corp | 粉粒体処理システム及び粉粒体処理方法 |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5656246A (en) * | 1979-10-12 | 1981-05-18 | Toshiro Watanabe | Method and apparatus for sorting of synthetic resin waste |
| JP3120060B2 (ja) * | 1997-08-29 | 2000-12-25 | 川崎重工業株式会社 | 塩を含む燃料・廃棄物の処理方法 |
| JP3863274B2 (ja) * | 1997-12-22 | 2006-12-27 | 壯太郎 樋口 | 廃棄物の埋立処理方法 |
| JP2001079508A (ja) * | 1999-09-16 | 2001-03-27 | Shinko Pantec Co Ltd | 廃棄物の洗浄処理設備および洗浄方法 |
| JP2006021956A (ja) * | 2004-07-08 | 2006-01-26 | Takuma Co Ltd | 炭化物の脱塩設備および脱塩方法 |
-
2012
- 2012-05-30 JP JP2012538893A patent/JP5904123B2/ja not_active Expired - Fee Related
- 2012-05-30 WO PCT/JP2012/063951 patent/WO2012165488A1/fr not_active Ceased
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2002254063A (ja) * | 2001-03-01 | 2002-09-10 | Konoike Constr Ltd | 汚染土壌の浄化方法及びそれに用いる分離装置 |
| JP2005081247A (ja) * | 2003-09-09 | 2005-03-31 | Mitsubishi Heavy Ind Ltd | 汚染土壌の浄化装置 |
| JP2005279476A (ja) * | 2004-03-30 | 2005-10-13 | Kentaro Asakura | 有害物質の処理方法 |
| JP2009136846A (ja) * | 2007-12-11 | 2009-06-25 | Kubota Corp | 粉粒体処理システム及び粉粒体処理方法 |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPWO2015137484A1 (ja) * | 2014-03-14 | 2017-04-06 | 株式会社ピーシーエス | 超微細気泡含有液体を用いる超微細気泡洗浄方法、その装置及び加圧浮上装置 |
| US11311921B2 (en) | 2014-03-14 | 2022-04-26 | Pcs Co., Ltd. | Ultrafine bubble cleaning method using ultrafine bubble-containing liquid, apparatus therefor, and dissolved air floatation apparatus |
| JP2016050780A (ja) * | 2014-08-28 | 2016-04-11 | 株式会社東芝 | 排水処理装置および排水処理方法 |
| JP2019209324A (ja) * | 2018-05-30 | 2019-12-12 | 大矢建設工業株式会社 | 排水処理システム及び排水処理方法 |
| CN115805216A (zh) * | 2022-12-29 | 2023-03-17 | 苏州赢众环保有限公司 | 一种一体化填料清洗设备 |
| CN121060926A (zh) * | 2025-11-07 | 2025-12-05 | 中国资源循环集团塑料再生有限公司 | 一种利用垃圾渗滤液清洗废塑料及回收腐殖土的耦合方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| JP5904123B2 (ja) | 2016-04-13 |
| JPWO2012165488A1 (ja) | 2015-02-23 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5376683B2 (ja) | カルシウムスケールの防止方法 | |
| JP5904123B2 (ja) | 混合材料の洗浄分別方法および洗浄分別装置 | |
| JPWO2013031689A1 (ja) | 放射性物質および/または重金属含有水の浄化方法および浄化装置 | |
| KR102315734B1 (ko) | 순환모듈을 구비하는 오염토양 정화용 침전조 장치 | |
| CN116096629A (zh) | 浮游污染物质回收装置以及浮游污染物质回收系统 | |
| JP5852199B1 (ja) | 浸透探傷検査で発生する蛍光液と現像剤を含む浸透液水洗の排水処理装置と、その方法 | |
| WO2009107332A1 (fr) | Appareil de traitement des eaux usées et procédé de traitement des eaux usées | |
| JP2015031661A (ja) | 焼却飛灰の洗浄方法 | |
| KR101206399B1 (ko) | 배수의 처리방법 및 처리시스템 | |
| CN207210182U (zh) | 一种垃圾渗滤液膜法浓缩液减量处理设备 | |
| CN211004886U (zh) | 一种垃圾渗滤液处理系统 | |
| KR100550976B1 (ko) | 하수 방류수 처리 시스템 및 처리 방법 | |
| JP2011041907A (ja) | 水処理システム | |
| JP5845478B2 (ja) | 放射性物質含有排水の処理方法及び処理装置 | |
| Pabby et al. | Membrane applications in industrial waste management (including nuclear), environmental engineering and future trends in membrane science: Introduction | |
| SK5697Y1 (en) | Device for waste disposal | |
| KR102372410B1 (ko) | 오염토양 정화용 침전조 장치 | |
| JP2003340247A (ja) | 水処理装置および方法 | |
| JP5985415B2 (ja) | 両性イオン交換樹脂の再生方法 | |
| JP5594774B2 (ja) | ガス溶解機構 | |
| JP2015055542A (ja) | 焼却飛灰の洗浄方法 | |
| JP5875142B2 (ja) | カルシウムスケール防止装置及び防止方法 | |
| CN205999228U (zh) | 垃圾渗滤液处理系统 | |
| KR101522319B1 (ko) | 가압부상법과 막교체가 용이한 이동식 침지막 복합 방사성 폐액 처리시스템 및 방법 | |
| CN101654290A (zh) | 化学膜废水处理法及其装置 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| ENP | Entry into the national phase |
Ref document number: 2012538893 Country of ref document: JP Kind code of ref document: A |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 12793577 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 12793577 Country of ref document: EP Kind code of ref document: A1 |