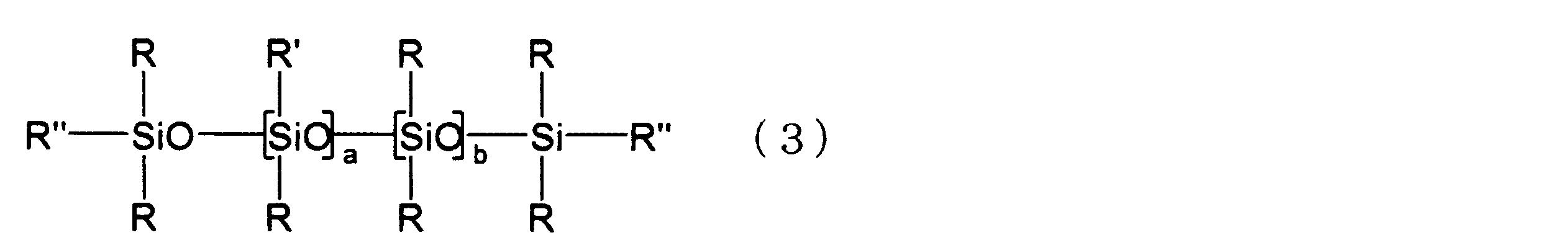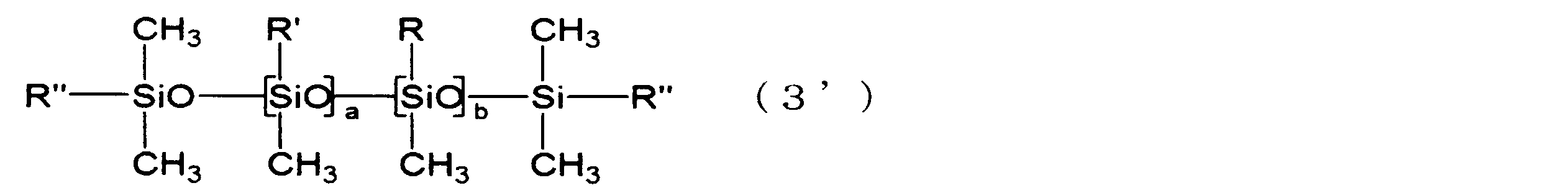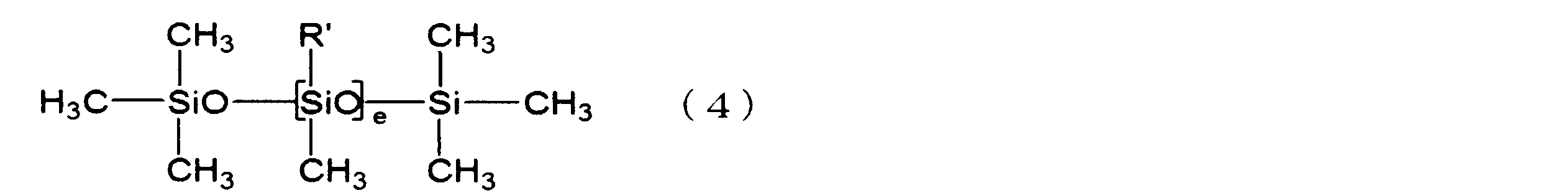WO2013137257A1 - 樹脂組成物、プリプレグ及び金属箔張積層板 - Google Patents
樹脂組成物、プリプレグ及び金属箔張積層板 Download PDFInfo
- Publication number
- WO2013137257A1 WO2013137257A1 PCT/JP2013/056817 JP2013056817W WO2013137257A1 WO 2013137257 A1 WO2013137257 A1 WO 2013137257A1 JP 2013056817 W JP2013056817 W JP 2013056817W WO 2013137257 A1 WO2013137257 A1 WO 2013137257A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- epoxy
- resin composition
- mass
- parts
- resin
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- AKSJTTWKULBVJI-UHFFFAOYSA-N CC(C)CNC(NCC1OC1)=O Chemical compound CC(C)CNC(NCC1OC1)=O AKSJTTWKULBVJI-UHFFFAOYSA-N 0.000 description 1
- ZSBDPRIWBYHIAF-UHFFFAOYSA-N CC(NC(C)=O)=O Chemical compound CC(NC(C)=O)=O ZSBDPRIWBYHIAF-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L83/00—Compositions of macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing silicon with or without sulfur, nitrogen, oxygen or carbon only; Compositions of derivatives of such polymers
- C08L83/04—Polysiloxanes
- C08L83/06—Polysiloxanes containing silicon bound to oxygen-containing groups
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D183/00—Coating compositions based on macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing silicon, with or without sulfur, nitrogen, oxygen, or carbon only; Coating compositions based on derivatives of such polymers
- C09D183/04—Polysiloxanes
- C09D183/06—Polysiloxanes containing silicon bound to oxygen-containing groups
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B15/00—Layered products comprising a layer of metal
- B32B15/04—Layered products comprising a layer of metal comprising metal as the main or only constituent of a layer, which is next to another layer of the same or of a different material
- B32B15/08—Layered products comprising a layer of metal comprising metal as the main or only constituent of a layer, which is next to another layer of the same or of a different material of synthetic resin
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B15/00—Layered products comprising a layer of metal
- B32B15/14—Layered products comprising a layer of metal next to a fibrous or filamentary layer
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B5/00—Layered products characterised by the non- homogeneity or physical structure, i.e. comprising a fibrous, filamentary, particulate or foam layer; Layered products characterised by having a layer differing constitutionally or physically in different parts
- B32B5/02—Layered products characterised by the non- homogeneity or physical structure, i.e. comprising a fibrous, filamentary, particulate or foam layer; Layered products characterised by having a layer differing constitutionally or physically in different parts characterised by structural features of a fibrous or filamentary layer
- B32B5/022—Non-woven fabric
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B5/00—Layered products characterised by the non- homogeneity or physical structure, i.e. comprising a fibrous, filamentary, particulate or foam layer; Layered products characterised by having a layer differing constitutionally or physically in different parts
- B32B5/02—Layered products characterised by the non- homogeneity or physical structure, i.e. comprising a fibrous, filamentary, particulate or foam layer; Layered products characterised by having a layer differing constitutionally or physically in different parts characterised by structural features of a fibrous or filamentary layer
- B32B5/024—Woven fabric
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G59/00—Polycondensates containing more than one epoxy group per molecule; Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups
- C08G59/18—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing
- C08G59/40—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing characterised by the curing agents used
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G73/00—Macromolecular compounds obtained by reactions forming a linkage containing nitrogen with or without oxygen or carbon in the main chain of the macromolecule, not provided for in groups C08G12/00 - C08G71/00
- C08G73/06—Polycondensates having nitrogen-containing heterocyclic rings in the main chain of the macromolecule
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G77/00—Macromolecular compounds obtained by reactions forming a linkage containing silicon with or without sulfur, nitrogen, oxygen or carbon in the main chain of the macromolecule
- C08G77/04—Polysiloxanes
- C08G77/38—Polysiloxanes modified by chemical after-treatment
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J5/00—Manufacture of articles or shaped materials containing macromolecular substances
- C08J5/24—Impregnating materials with prepolymers which can be polymerised in situ, e.g. manufacture of prepregs
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J5/00—Manufacture of articles or shaped materials containing macromolecular substances
- C08J5/24—Impregnating materials with prepolymers which can be polymerised in situ, e.g. manufacture of prepregs
- C08J5/241—Impregnating materials with prepolymers which can be polymerised in situ, e.g. manufacture of prepregs using inorganic fibres
- C08J5/244—Impregnating materials with prepolymers which can be polymerised in situ, e.g. manufacture of prepregs using inorganic fibres using glass fibres
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J5/00—Manufacture of articles or shaped materials containing macromolecular substances
- C08J5/24—Impregnating materials with prepolymers which can be polymerised in situ, e.g. manufacture of prepregs
- C08J5/249—Impregnating materials with prepolymers which can be polymerised in situ, e.g. manufacture of prepregs characterised by the additives used in the prepolymer mixture
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K3/00—Use of inorganic substances as compounding ingredients
- C08K3/18—Oxygen-containing compounds, e.g. metal carbonyls
- C08K3/20—Oxides; Hydroxides
- C08K3/22—Oxides; Hydroxides of metals
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/54—Silicon-containing compounds
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K9/00—Use of pretreated ingredients
- C08K9/02—Ingredients treated with inorganic substances
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L63/00—Compositions of epoxy resins; Compositions of derivatives of epoxy resins
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L79/00—Compositions of macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing nitrogen with or without oxygen or carbon only, not provided for in groups C08L61/00 - C08L77/00
- C08L79/04—Polycondensates having nitrogen-containing heterocyclic rings in the main chain; Polyhydrazides; Polyamide acids or similar polyimide precursors
-
- H—ELECTRICITY
- H05—ELECTRIC TECHNIQUES NOT OTHERWISE PROVIDED FOR
- H05K—PRINTED CIRCUITS; CASINGS OR CONSTRUCTIONAL DETAILS OF ELECTRIC APPARATUS; MANUFACTURE OF ASSEMBLAGES OF ELECTRICAL COMPONENTS
- H05K1/00—Printed circuits
- H05K1/02—Details
- H05K1/03—Use of materials for the substrate
- H05K1/0313—Organic insulating material
- H05K1/0353—Organic insulating material consisting of two or more materials, e.g. two or more polymers, polymer + filler, + reinforcement
- H05K1/0373—Organic insulating material consisting of two or more materials, e.g. two or more polymers, polymer + filler, + reinforcement containing additives, e.g. fillers
-
- H—ELECTRICITY
- H05—ELECTRIC TECHNIQUES NOT OTHERWISE PROVIDED FOR
- H05K—PRINTED CIRCUITS; CASINGS OR CONSTRUCTIONAL DETAILS OF ELECTRIC APPARATUS; MANUFACTURE OF ASSEMBLAGES OF ELECTRICAL COMPONENTS
- H05K1/00—Printed circuits
- H05K1/02—Details
- H05K1/03—Use of materials for the substrate
- H05K1/05—Insulated conductive substrates, e.g. insulated metal substrate
- H05K1/056—Insulated conductive substrates, e.g. insulated metal substrate the metal substrate being covered by an organic insulating layer
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2260/00—Layered product comprising an impregnated, embedded, or bonded layer wherein the layer comprises an impregnation, embedding, or binder material
- B32B2260/02—Composition of the impregnated, bonded or embedded layer
- B32B2260/021—Fibrous or filamentary layer
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2260/00—Layered product comprising an impregnated, embedded, or bonded layer wherein the layer comprises an impregnation, embedding, or binder material
- B32B2260/04—Impregnation, embedding, or binder material
- B32B2260/046—Synthetic resin
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2262/00—Composition or structural features of fibres which form a fibrous or filamentary layer or are present as additives
- B32B2262/02—Synthetic macromolecular fibres
- B32B2262/0261—Polyamide fibres
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2262/00—Composition or structural features of fibres which form a fibrous or filamentary layer or are present as additives
- B32B2262/02—Synthetic macromolecular fibres
- B32B2262/0276—Polyester fibres
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2262/00—Composition or structural features of fibres which form a fibrous or filamentary layer or are present as additives
- B32B2262/10—Inorganic fibres
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2262/00—Composition or structural features of fibres which form a fibrous or filamentary layer or are present as additives
- B32B2262/10—Inorganic fibres
- B32B2262/101—Glass fibres
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2307/00—Properties of the layers or laminate
- B32B2307/30—Properties of the layers or laminate having particular thermal properties
- B32B2307/306—Resistant to heat
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2307/00—Properties of the layers or laminate
- B32B2307/70—Other properties
- B32B2307/732—Dimensional properties
- B32B2307/734—Dimensional stability
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2457/00—Electrical equipment
- B32B2457/08—PCBs, i.e. printed circuit boards
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G77/00—Macromolecular compounds obtained by reactions forming a linkage containing silicon with or without sulfur, nitrogen, oxygen or carbon in the main chain of the macromolecule
- C08G77/04—Polysiloxanes
- C08G77/14—Polysiloxanes containing silicon bound to oxygen-containing groups
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2383/00—Characterised by the use of macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing silicon with or without sulfur, nitrogen, oxygen, or carbon only; Derivatives of such polymers
- C08J2383/04—Polysiloxanes
- C08J2383/06—Polysiloxanes containing silicon bound to oxygen-containing groups
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2479/00—Characterised by the use of macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing nitrogen with or without oxygen, or carbon only, not provided for in groups C08J2461/00 - C08J2477/00
- C08J2479/04—Polycondensates having nitrogen-containing heterocyclic rings in the main chain; Polyhydrazides; Polyamide acids or similar polyimide precursors
- C08J2479/08—Polyimides; Polyester-imides; Polyamide-imides; Polyamide acids or similar polyimide precursors
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K3/00—Use of inorganic substances as compounding ingredients
- C08K3/18—Oxygen-containing compounds, e.g. metal carbonyls
- C08K3/20—Oxides; Hydroxides
- C08K3/22—Oxides; Hydroxides of metals
- C08K2003/2237—Oxides; Hydroxides of metals of titanium
- C08K2003/2241—Titanium dioxide
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K2201/00—Specific properties of additives
- C08K2201/002—Physical properties
- C08K2201/003—Additives being defined by their diameter
-
- H—ELECTRICITY
- H05—ELECTRIC TECHNIQUES NOT OTHERWISE PROVIDED FOR
- H05K—PRINTED CIRCUITS; CASINGS OR CONSTRUCTIONAL DETAILS OF ELECTRIC APPARATUS; MANUFACTURE OF ASSEMBLAGES OF ELECTRICAL COMPONENTS
- H05K2201/00—Indexing scheme relating to printed circuits covered by H05K1/00
- H05K2201/02—Fillers; Particles; Fibers; Reinforcement materials
- H05K2201/0203—Fillers and particles
- H05K2201/0206—Materials
- H05K2201/0209—Inorganic, non-metallic particles
-
- H—ELECTRICITY
- H05—ELECTRIC TECHNIQUES NOT OTHERWISE PROVIDED FOR
- H05K—PRINTED CIRCUITS; CASINGS OR CONSTRUCTIONAL DETAILS OF ELECTRIC APPARATUS; MANUFACTURE OF ASSEMBLAGES OF ELECTRICAL COMPONENTS
- H05K2201/00—Indexing scheme relating to printed circuits covered by H05K1/00
- H05K2201/20—Details of printed circuits not provided for in H05K2201/01 - H05K2201/10
- H05K2201/2054—Light-reflecting surface, e.g. conductors, substrates, coatings, dielectrics
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10T—TECHNICAL SUBJECTS COVERED BY FORMER US CLASSIFICATION
- Y10T156/00—Adhesive bonding and miscellaneous chemical manufacture
- Y10T156/10—Methods of surface bonding and/or assembly therefor
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10T—TECHNICAL SUBJECTS COVERED BY FORMER US CLASSIFICATION
- Y10T428/00—Stock material or miscellaneous articles
- Y10T428/31504—Composite [nonstructural laminate]
- Y10T428/31511—Of epoxy ether
- Y10T428/31529—Next to metal
Definitions
- the present invention relates to a resin composition, and a prepreg, a metal foil-clad laminate, and a printed wiring board using the resin composition, and particularly suitable for printed wiring board materials, particularly printed wiring boards for mounting light emitting diodes (LEDs).
- the present invention relates to a resin composition, a prepreg, and a metal foil-clad laminate that can be used.
- a laminated board obtained by impregnating a glass woven fabric with an epoxy resin containing titanium dioxide and then heat-curing it (see, for example, Patent Document 1) is known. It has been.
- a laminated board using this type of epoxy resin usually has low heat resistance, the substrate surface is subjected to heat treatment in the manufacturing process of the printed wiring board or the LED mounting process, heating in the use after the LED mounting, or light irradiation. Can be discolored, which can cause the problem of significantly reduced reflectivity.
- laminates used for printed wiring boards for LED mounting are particularly required to have laminates with excellent heat resistance and light resistance. It is becoming. More specifically, it is required to have excellent heat resistance and not only high light reflectance in the ultraviolet light region and visible light region, but also a small decrease in light reflectance due to heat treatment or light irradiation treatment.
- Proposed prepreg consisting of a resin composition containing bisphenol A novolac type epoxy resin, alicyclic epoxy resin and titanium dioxide and a base material as a laminated board that has improved the decrease in reflectance at the time of such short wavelength light irradiation (For example, refer to Patent Document 2).
- a silicone laminate obtained by impregnating glass cloth with an addition-curable silicone resin composition containing an organopolysiloxane or organohydrogenpolysiloxane having a specific resin structure, a platinum group metal catalyst, and a filler. It is known that the substrate is excellent in mechanical properties, heat resistance and discoloration resistance, has little surface tack, and has little decrease in light reflectance upon heating and light irradiation (for example, Patent Document 3). reference.).
- a laminate for a printed wiring board requires high adhesion (peel strength) with a metal foil laminated when used as a metal foil-clad laminate, and is high when performing reflow with lead-free solder. Heat resistance is required.
- a resin composition that can realize a printed wiring board excellent in all of these characteristics has not been developed yet, and there is a problem in mountability in the mounting process and reliability in use after mounting.
- the present invention has been made in view of the above problems, and its purpose is to have high light reflectance in the ultraviolet light region and visible light region, and there is little reduction in light reflectance due to heat treatment and light irradiation treatment, Resin composition capable of realizing a prepreg or a metal foil-clad laminate having good peel strength with metal foil, excellent heat resistance during moisture absorption, and good appearance and excellent storage stability, as well as this It is to provide a prepreg, a metal foil-clad laminate and a printed wiring board.
- the inventors of the present invention contain at least an epoxy-modified silicone compound, a branched imide resin having an isocyanurate group and a carboxyl group, a phosphorus-based curing accelerator, titanium dioxide and a dispersant.
- the resin composition to be used the light reflectance is high in the ultraviolet light region and the visible light region, there is little decrease in light reflectance due to the heat treatment and light irradiation treatment, the peel strength with the metal foil is good, and when moisture is absorbed
- the present inventors have found that a prepreg or a metal foil-clad laminate having excellent heat resistance, excellent appearance and storage stability can be obtained, and the present invention has been achieved.
- ⁇ 1> Contains an epoxy-modified silicone compound (A), a branched imide resin (B) having an isocyanurate group and a carboxyl group, a phosphorus curing accelerator (C), titanium dioxide (D) and a dispersant (E). , Resin composition.
- branched imide resin (B) is at least one selected from the group consisting of an epoxy-modified branched imide resin, an alcohol-modified branched imide resin, and an amine-modified branched imide resin.
- the epoxy equivalent of the monomer epoxy compound (F) is 0.1 to 0.8 equivalent based on the carboxyl residue of the branched imide resin (B), described in ⁇ 5> above.
- Resin composition. ⁇ 7> The monomer epoxy compound (F) is represented by the following formulas (8) to (10):
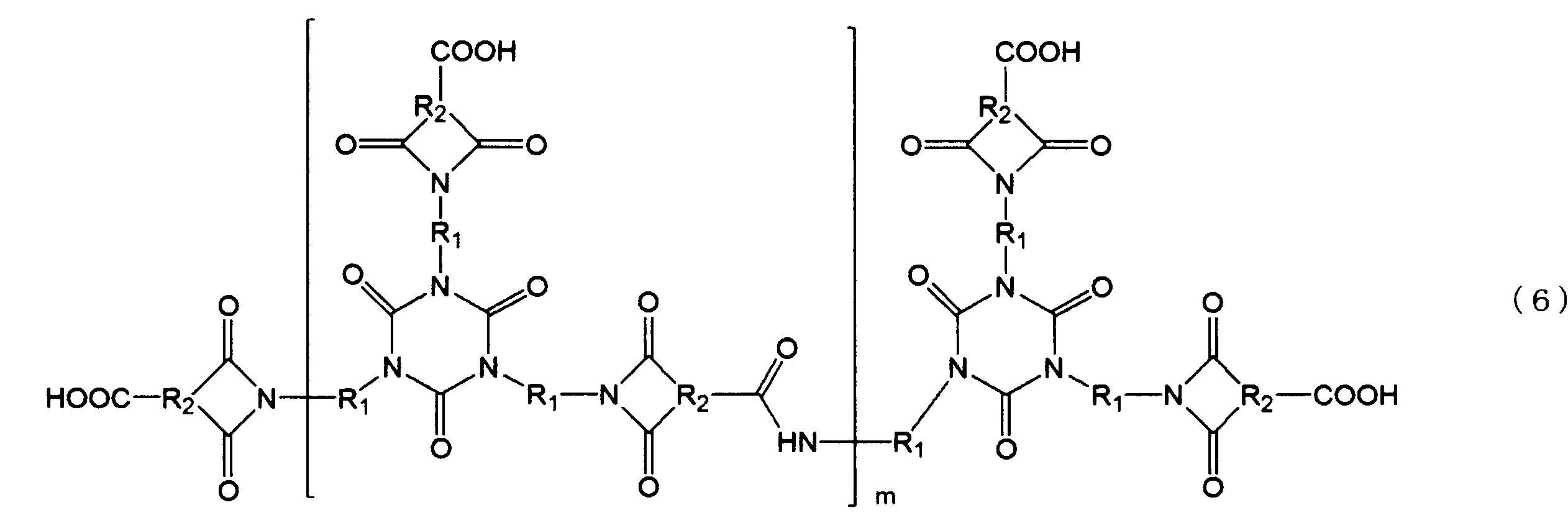
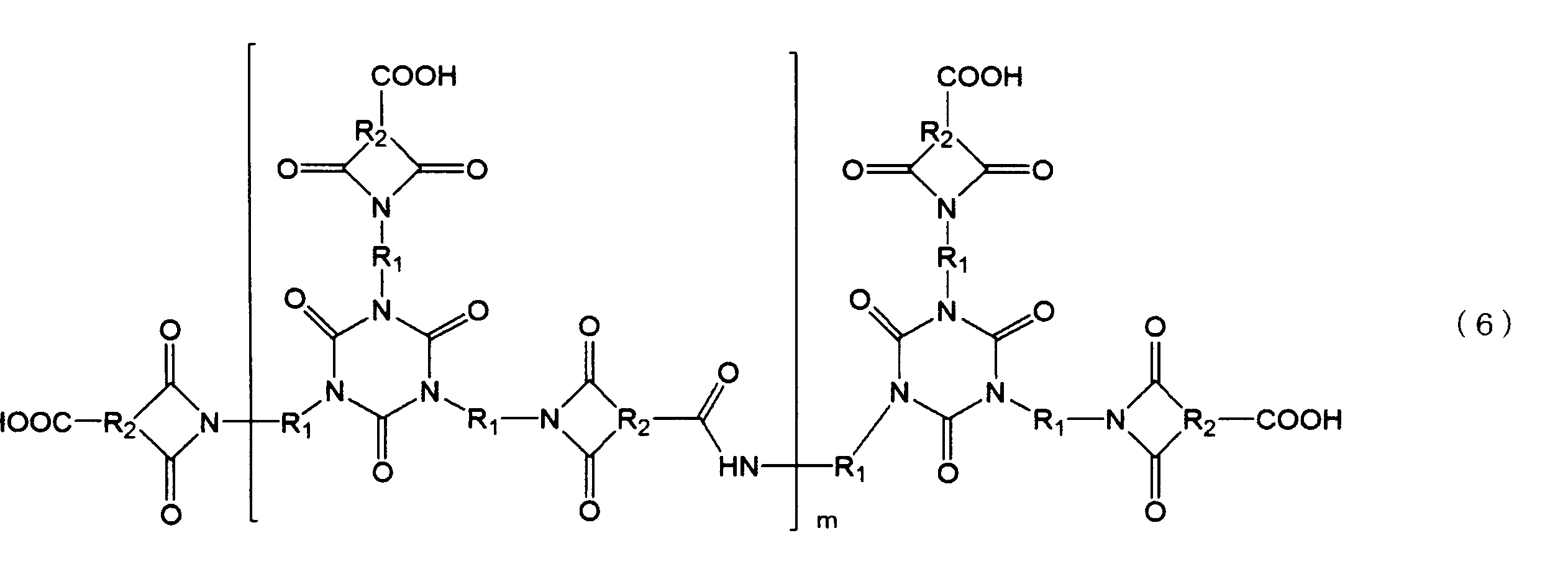
- the branched imide resin (B) is represented by the following formula (6).
- each R 1 is independently a divalent alicyclic group
- each R 2 is independently a trivalent alicyclic group
- m is 1 to (It is an integer of 10.)
- the resin composition according to any one of ⁇ 1> to ⁇ 10> above.
- ⁇ 12> The resin composition according to any one of ⁇ 1> to ⁇ 11>, further containing a silane coupling agent (H).
- ⁇ 13> The above ⁇ 1> to ⁇ 12>, wherein the phosphorus-based curing accelerator (C) is contained in an amount of 0.1 to 10 parts by mass with respect to 100 parts by mass in total of the components (A) and (B).
- the titanium dioxide (D) is surface-treated with SiO 2 , Al 2 O 3 , ZrO 2 , and / or ZnO, and the total amount of the titanium dioxide (D) is 100 parts by mass. 0.5 to 15 parts by mass of SiO 2 , 0.5 to 15 parts by mass of Al 2 O 3 , 0.5 to 15 parts by mass of ZrO 2 and / or 0.5 to 15 parts by mass of ZnO
- ⁇ 17> Any of the above ⁇ 1> to ⁇ 16>, wherein the dispersant (E) is contained in an amount of 0.05 to 10 parts by mass with respect to a total of 100 parts by mass of the components (A) and (B).
- ⁇ 18> The resin composition according to any one of ⁇ 1> to ⁇ 17>, wherein the dispersant (E) is a polymer wetting dispersant having an acid value of 20 to 200 mgKOH / g.
- the cycloaliphatic epoxy resin (G) is an alcohol adduct of vinylcyclohexene diepoxide, an alcohol adduct of 3,4-epoxycyclohexanecarboxylic acid-3 ′, 4′-epoxycyclohexylmethyl, bis 3 adipate , 4-epoxycyclohexylmethyl alcohol adduct, dicyclopentadiene diepoxide alcohol adduct, ⁇ -caprolactone modified bis (3,4-epoxycyclohexylmethyl) -4,5-epoxycyclohexane-1,2-dicarboxylic acid Alcohol adducts, ⁇ -caprolactone modified tetra (3,4-epoxycyclohexylmethyl) butane-tetracarboxylic acid alcohol adducts, dip
- the resin composition according to ⁇ 5> which is prepared by blending (A), titanium dioxide (D), and a dispersant (E).
- the titanium dioxide (D) is the epoxy-modified silicone compound (A), the branched imide resin (B), the monomer epoxy compound (F), and the alicyclic epoxy resin (G).
- the resin composition as described in ⁇ 22> above which is contained in an amount of 10 to 400 parts by mass with respect to 100 parts by mass in total.
- the dispersant (E) is the epoxy-modified silicone compound (A), the branched imide resin (B), the monomer epoxy compound (F), and the alicyclic epoxy resin (G).
- the resin composition according to ⁇ 22> or ⁇ 23> which is contained in an amount of 0.05 to 5 parts by mass with respect to 100 parts by mass in total.
- a resin composition for a printed wiring board for LED mounting comprising the resin composition according to any one of ⁇ 1> to ⁇ 25>.
- a prepreg obtained by impregnating or coating a substrate with the resin composition according to any one of ⁇ 1> to ⁇ 25> above.
- ⁇ 28> A metal foil-clad laminate obtained by laminating at least one prepreg according to the above ⁇ 27>, laminating a metal foil on one or both sides thereof.
- the combined use of the branched imide resin (B) having an isocyanurate group and a carboxyl group and the phosphorus-based curing accelerator (C) adjusts the reaction rate at the time of preparing the prepreg.
- High light reflectivity in the visible light region little decrease in light reflectivity due to heat treatment and light irradiation treatment, good peel strength with metal foil, excellent heat resistance during moisture absorption, and good appearance
- the use of the phosphorus-based curing accelerator (C) suppresses the activity of the branched imide resin (B) to prevent an increase in the varnish gel time and an increase in the prepreg viscosity, thereby improving the storage stability of the varnish and producing a prepreg. It is also possible to improve workability at the time.
- the heat resistance and handling properties are enhanced by the inclusion of the monomer epoxy compound and the prepreg meltability at the time of heating is enhanced, so that the moldability of the metal foil-clad laminate is further increased. improves. Therefore, the resin composition, prepreg, metal foil-clad laminate and the like of the present invention can be suitably used for printed wiring boards for LED mounting and the like, and their industrial practicality is extremely high.
- the resin composition of the present embodiment is a so-called thermosetting resin composition that is cured by heat, an epoxy-modified silicone compound (A), a branched imide resin (B) having an isocyanurate group and a carboxyl group, and a phosphorus-based curing. It contains at least an accelerator (C), titanium dioxide (D) and a dispersant (E).
- the epoxy-modified silicone compound (A) used in the resin composition of the present embodiment is not particularly limited as long as the organosilicon compound has an epoxy group, but a siloxane bond (Si—O—Si bond) is added to the main skeleton.
- An aliphatic epoxy-modified silicone compound in which a substituted or unsubstituted aliphatic hydrocarbon group having an epoxy group is introduced into the silicone compound is preferable.
- the light reflectance of the ultraviolet light region and the visible light region of the obtained metal foil-clad laminate is increased, the discoloration due to the heat treatment and the light irradiation treatment is suppressed, and the reduction of the light reflectance is suppressed, as well as the metal
- the peel strength and heat resistance with the foil tend to be particularly enhanced.
- a silicone compound having a repeating unit represented by the following formula (1) having at least three R ′ in one molecule, and not containing an alkoxy group is preferable. From the viewpoint of excellent workability, a liquid one is more preferable.
- having a repeating unit represented by the following formula (1) includes not only one having a plurality of units (preferably 3 or more) having the same R and / or R ′, but also R and / or It is an implication that includes both those in which R ′ has a plurality (preferably 3 or more) of different units.
- R is a hydrogen atom or a substituted or unsubstituted monovalent hydrocarbon group, and R ′ is an organic group having an epoxy group.
- R examples of the monovalent hydrocarbon group represented by R include a substituted or unsubstituted aliphatic hydrocarbon group, preferably having 1 to 20 carbon atoms, more preferably Is 1-8. More specifically, for example, alkyl groups such as a methyl group, an ethyl group, a propyl group, a butyl group, a hexyl group, and an octyl group, and a part or all of hydrogen atoms of these monovalent hydrocarbon groups are epoxy groups ( However, an epoxy cyclohexyl group is excluded), and a group substituted with a methacryl group, an acryl group, a mercapto group, an amino group, or the like is exemplified, but it is not particularly limited thereto. Among these, R is preferably a methyl group, an ethyl group, or a hydrogen atom, and more preferably a methyl group.
- R ′ examples of the organic group having an epoxy group represented by R ′ include a substituted or unsubstituted aliphatic hydrocarbon group having an epoxy group, the carbon number of which is 2 to 20 Is more preferable, and 2 to 12 is more preferable. More specifically, for example, a glycidoxypropyl group, a 3,4-epoxycyclohexylethyl group, and the like can be mentioned, but not limited thereto.
- R ′ is preferably an organic group having a 3,4-epoxycyclohexyl group.
- the silicone compound having a repeating unit represented by the above formula (1) preferably has 3 to 8 R's in one molecule.
- R ′ When the silicone compound having a unit represented by the above formula (1) has R ′ within this range, a cured product having high hardness and excellent toughness tends to be obtained.
- the silicone compound having a repeating unit represented by the above formula (1) preferably has a degree of polymerization of 3 to 100. Those having a polymerization degree in this range are easily available because they are easily synthesized industrially. In addition to these properties, the degree of polymerization is more preferably from 3 to 50, and even more preferably from 3 to 10 from the viewpoint of further suppressing cure shrinkage.
- the silicone compound having a repeating unit represented by the above formula (1) does not contain an alkoxy group. Therefore, there is no cure shrinkage due to dealcoholization reaction, and excellent dielectric breakdown characteristics can be obtained when used in combination with the branched imide resin (B).
- silicone compound having a repeating unit represented by the above formula (1) include, for example, those having a linear structure or a ring structure.
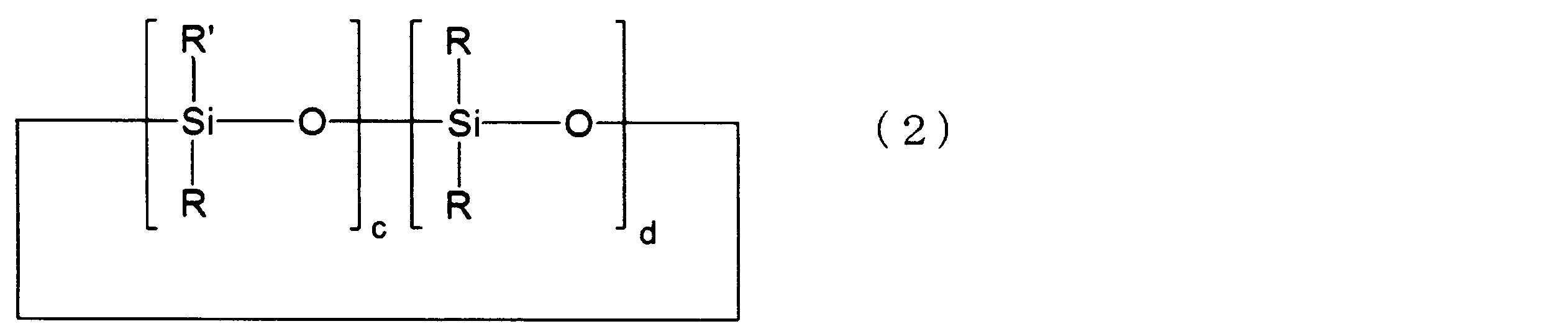
- Examples of the ring structure include a cyclic silicone compound represented by the following formula (2).
- the cyclic silicone compound represented by the following formula (2) is preferable from the viewpoint of small cure shrinkage.
- R and R ′ are as defined in the above formula (1), c represents an integer of 3 to 5, d represents an integer of 0 to 2, c and d Is an integer of 3 to 5, and each repeating unit can be bonded at random.
- c is an integer of 3 to 4
- d is an integer of 0 to 1
- the sum of c and d is 4.
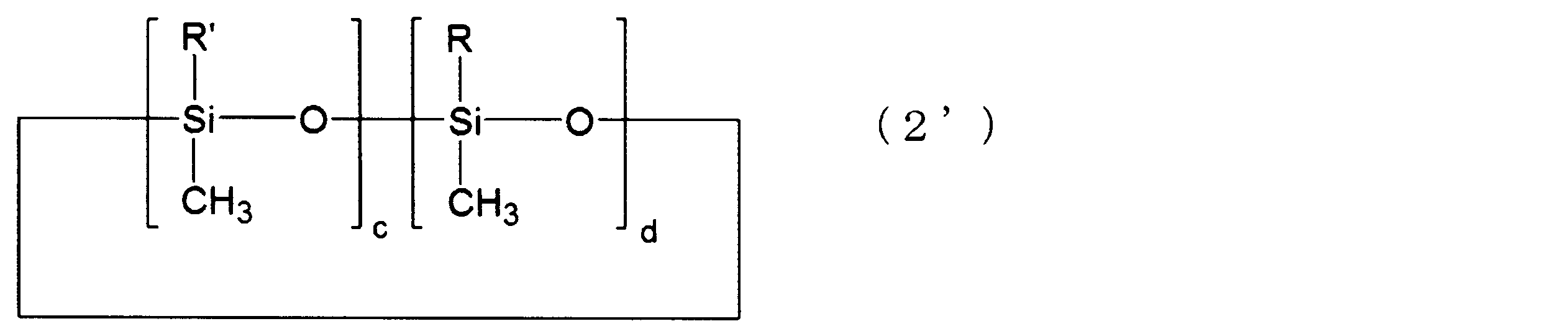
- cyclic silicone compounds represented by the above formula (2) a cyclic silicone compound represented by the following formula (2 ′) is more preferable.
- the linear silicone compound represented by following formula (3) is mentioned, for example.
- R and R ′ have the same meaning as described in the above formula (1)
- R ′′ represents R or R ′
- R, R ′ and R ′′ are the same as each other.
- A represents an integer of 1 to 10
- b represents an integer of 0 to 8
- a is an integer of 4 to 8
- b is an integer of 0 to 4
- the sum of a and b is 4 to 8.
- a linear silicone compound represented by the following formula (3 ′) is more preferable.
- R, R ′, R ′′, a and b have the same meanings as described in the above formulas (1) to (3).
- a linear silicone compound represented by the following formula (4) is more preferable.
- R ' has the same meaning as described in formula (1) above, and e represents an integer of 3 to 10.
- e is preferably an integer of 3 to 8.
- the silicone compound having a repeating unit represented by the above formula (1) is more preferably a cyclic silicone compound represented by the following formula (5).
- R ′ has the same meaning as described in the formula (1), and f represents an integer of 3 to 5)
- f is preferably 4.
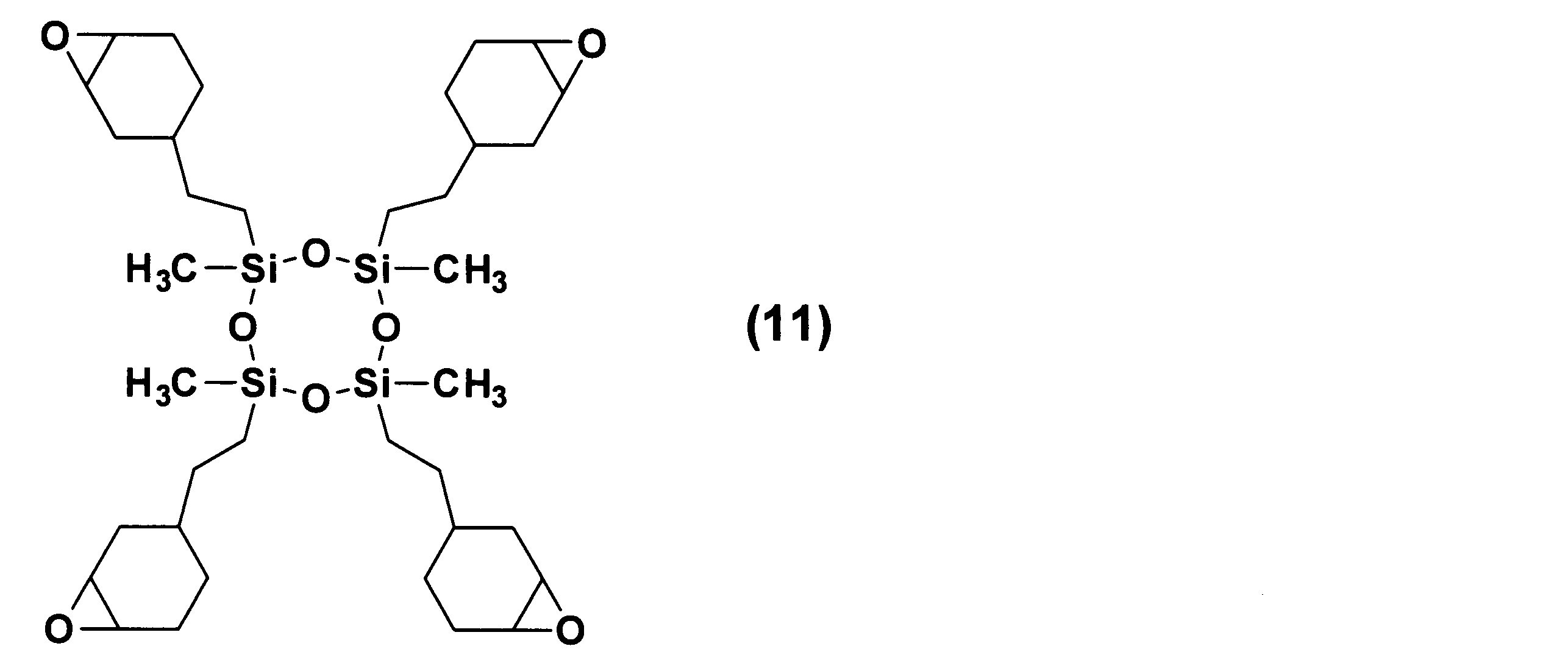
- aliphatic epoxy-modified silicone compound examples include, for example, (CH 3 ) 3 SiO (R′CH 3 SiO) 5 Si (CH 3 ) 3 , (CH 3 ) 3 SiO (R′CH 3 SiO).
- R ′ (CH 3 ) 2 SiO (R′CH 3 SiO) 6 ((CH 3 ) 2 SiO) 2 Si (CH 3 ) 2 R ′, R ′ (CH 3 ) 2 SiO (R′CH 3 SiO) 6 ((CH 3 ) 2 SiO) 3 Si (CH 3 ) 2 R ′, R ′ (CH 3 ) 2 SiO (R′CH 3 SiO) 7 ((CH 3 ) 2 SiO) Si (CH 3 ) 2 R ′ R ′ (CH 3 ) 2 SiO (R′CH 3 SiO) 7 ((CH 3 ) 2 SiO) 2 Si (CH 3 ) 2 R ′, R ′ (CH 3 ) 2 SiO (R′CH 3 SiO) 7 ((CH 3 ) 2 SiO) 3 Si (CH 3 ) 2 R ′, R ′ (CH 3 ) 2 SiO (R′CH 3 SiO) 7 ((CH 3 ) 2 SiO) 3 Si (CH 3 ) 2 R ′, R ′ (
- the above-mentioned epoxy-modified silicone compound (A) can be produced by a known method, and a commercially available product can be easily obtained. For example, it can be obtained by subjecting an organohydrogenpolysiloxane to an addition reaction (hydrosilylation) with an allyl epoxy compound (for example, 4-vinylcyclohexene oxide) using a catalyst such as a platinum compound.
- an organohydrogenpolysiloxane to an addition reaction (hydrosilylation) with an allyl epoxy compound (for example, 4-vinylcyclohexene oxide) using a catalyst such as a platinum compound.
- X-40-2670, X-22-163A, X-22-163B, X-22-169AS, X-22-169B, X-41-1053, KF-105, PRX413 (Shin-Etsu Chemical ( Etc.), BY16-752A, BY16-799, BY16-873 (manufactured by Toray Dow Corning Co., Ltd.), SE-02CM (manufactured by Nagase ChemteX Corporation)) and the like are commercially available.
- an epoxy-modified silicone compound can be used individually by 1 type or in combination of 2 or more types as appropriate.
- the content of the epoxy-modified silicone compound (A) in the resin composition of the present embodiment is not particularly limited, but from the viewpoint of suppressing discoloration due to heat treatment and light irradiation treatment of the obtained metal foil-clad laminate, epoxy modification
- the amount is preferably 20 to 80 parts by mass, more preferably 25 to 75 parts by mass with respect to 100 parts by mass in total of the silicone compound (A) and the branched imide resin (B).
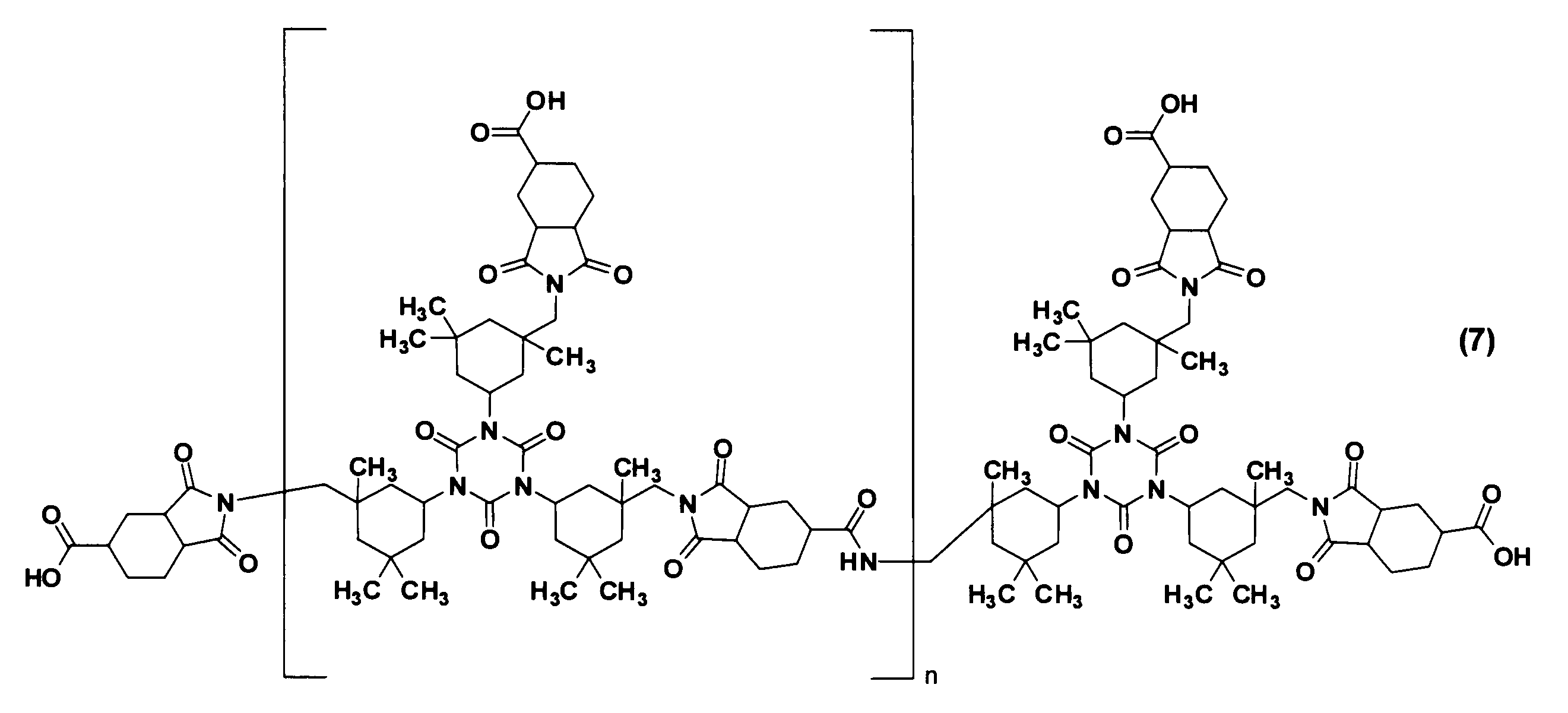
- the branched imide resin (B) used in the resin composition of the present embodiment is not particularly limited as long as it has an isocyanurate ring, a carboxyl group, and an imide group.
- a branched imide resin (hereinafter also referred to as “multi-branched imide resin”) having an isocyanurate ring and a plurality of cyclic imides having a carboxyl group, in which a large number of the cyclic imides are bonded to the isocyanurate ring.
- the branched imide resin (B) is more preferably an imide resin having an alicyclic structure, that is, an alicyclic imide resin (one having no aromatic ring).
- Such an alicyclic imide resin has an isocyanurate ring and a plurality of aliphatic cyclic imides having a carboxylic acid group bonded to each nitrogen atom of the isocyanurate ring via an aliphatic ring.
- Branched alicyclic imide resins are exemplified. Use of such a branched imide resin (B) having an isocyanurate ring and a carboxyl group together with an epoxy-modified silicone compound (A), a phosphorus curing accelerator (C), titanium dioxide (D) and a dispersant (E).
- the peel strength and heat resistance with the metal foil are particularly increased, but also the light reflectivity in the ultraviolet light region and the visible light region is particularly increased, and the light reflectivity is reduced by heat treatment and light irradiation treatment. It tends to be significantly suppressed.
- each R 1 is independently a divalent alicyclic group
- each R 2 is independently a trivalent alicyclic group
- m is 1 to (It is an integer of 10.)
- the alicyclic group represented by R 1 is a divalent group containing preferably 6 to 20 carbon atoms containing an aliphatic ring, and is a starting material alicyclic diamine or alicyclic group. It is a residue of diisocyanate.
- alicyclic diamine examples include 4,4′-diamino-dicyclohexylmethane, 3,3′-dimethyl-4,4′-diamino-dicyclohexylmethane, 3,3′-diethyl-4,4′-diamino- Dicyclohexylmethane, 3,3 ′, 5,5′-tetramethyl-4,4′-diamino-dicyclohexylmethane, 3,3 ′, 5,5′-tetraethyl-4,4′-diamino-dicyclohexylmethane, 3, 5-diethyl-3 ′, 5′-dimethyl-4,4′-diaminodicyclohexylmethane, 1,4-cyclohexanediamine, 1,3-cyclohexanediamine, isophoronediamine, 2,2-bis [4- (4-amino Cyclohexyloxy) cyclohexyl
- the alicyclic group represented by R 2 is a trivalent aliphatic group having an aliphatic ring, preferably having 6 to 20 carbon atoms, and the starting alicyclic tricarboxylic acid or It is an anhydride residue.
- Examples of the alicyclic tricarboxylic acid include cyclohexane-1,2,3-tricarboxylic acid, cyclohexane-1,2,4-tricarboxylic acid, cyclohexane-1,3,5-tricarboxylic acid, 5-methylcyclohexane-1, Examples include 2,4-tricarboxylic acid, 6-methylcyclohexane-1,2,4-tricarboxylic acid, and 3-methylcyclohexane-1,2,4-tricarboxylic acid, but are not particularly limited thereto.
- the branched imide resin (B) is represented by the above formula (6), its mass average molecular weight (Mw) is preferably 2,000 to 35,000, more preferably 2,000 to 10 , 000.
- the carboxyl group in the molecular structure has a curing accelerating ability in the curing reaction.
- the resin composition of the present embodiment since the phosphorus-based curing accelerator (C) having a curing accelerating ability is separately used, from the viewpoint of controlling the curing accelerating ability as a whole composition to an appropriate range, in the branched imide resin (B), at least a part of the carboxyl groups in the molecular structure is preferably modified.
- a modified branched imide resin obtained by reacting at least one selected from the group consisting of an epoxy compound, an alcohol compound and an amine compound with at least a part of the carboxyl group in the molecular structure.
- Branched imide resins, alcohol-modified branched imide resins, amine-modified branched imide resins are preferred.
- an epoxy-modified branched imide resin and an alcohol-modified branched imide resin are preferable, and an epoxy-modified branched imide resin is more preferable.
- a branched imide resin can be used individually by 1 type or in combination of 2 or more types.
- the degree of modification of the carboxyl group in the molecular structure of the branched imide resin (B) can be measured by the acid value.
- the acid value of the branched imide resin (B) is not particularly limited, but is 10 to 200 mgKOH / g in terms of solid content from the viewpoint of solubility in an organic solvent and curability of the resulting resin composition. More preferably, it is 10 to 100 mgKOH / g, and further preferably 20 to 80 mgKOH / g.
- the branched imide resin (B) can be produced by a known method, and a commercially available product can be easily obtained.
- Commercial products of the branched imide resin (B) include a branched imide compound having a structure represented by the following formula (7) (trade name: V-8002 (manufactured by DIC Corporation)), and this branched imide compound.
- Epoxy-modified (trade name: ELG-941 (manufactured by DIC)
- amine-modified from this branched imide compound (trade name: ELG-1301 (manufactured by DIC)
- this branched-type Examples thereof include those obtained by modifying an imide compound with alcohol (trade name: ELG-1302 (manufactured by DIC Corporation)).
- n an integer of 1 to 10.
- the content of the branched imide resin (B) in the resin composition of the present embodiment is not particularly limited, but increases the peel strength of the obtained metal foil-clad laminate and suppresses discoloration due to heat treatment and light irradiation treatment.
- the amount is preferably 20 to 80 parts by mass, more preferably 25 to 75 parts by mass with respect to 100 parts by mass in total of the epoxy-modified silicone compound (A) and the branched imide resin (B).
- the type of phosphorus-based curing accelerator (C) used in the resin composition of the present embodiment is not particularly limited as long as it is a compound containing phosphorus in the molecule and has curing acceleration ability.
- Specific examples thereof include methyltributylphosphonium dimethyl phosphate (trade name: PX-4MP (manufactured by Nippon Chemical Industry Co., Ltd.)), butylphosphonium diethyl phosphodithionate (trade name: PX-4ET (manufactured by Nippon Chemical Industry Co., Ltd.) )), Tetrabutylphosphonium tetrafluoroborate (trade name: PX-4FB (manufactured by Nippon Chemical Industry Co., Ltd.)), triphenylphosphine (manufactured by Tokyo Chemical Industry Co., Ltd.), phosphorus-containing cyanate ester (trade name: FR) -300 (manufactured by Lonza Corporation)).
- a phosphorus hardening accelerator can be used individually by 1 type or in combination of 2 or more types as appropriate. Among these, methyltributylphosphonium dimethyl phosphate is preferable from the viewpoint of light reflectance and glass transition temperature by heat treatment and light irradiation treatment.
- this phosphorus hardening accelerator (C) the activity of the branched imide resin (B) is suppressed, and as a result, the increase in the gel time of the varnish and the increase in the prepreg viscosity tend to be suppressed. Therefore, by using the phosphorus-based curing accelerator (C) together with the branched imide resin (B), it is possible to improve the storage stability of the varnish and the workability at the time of preparing the prepreg.
- content of the phosphorus hardening accelerator (C) in the resin composition of this embodiment is not specifically limited, From a viewpoint of the light reflectivity and glass transition temperature by heat processing and a light irradiation process, an epoxy-modified silicone compound ( The amount is preferably 0.1 to 10 parts by mass, more preferably 0.5 to 8 parts by mass with respect to 100 parts by mass in total of A) and the branched imide resin (B).
- the resin composition of the present embodiment contains titanium dioxide (D) as an essential inorganic filler. From the viewpoint of further increasing the light reflectance in the ultraviolet light region and the visible light region, titanium dioxide having a rutile type or anatase type crystal structure is preferable.
- the average particle diameter (D50) of titanium dioxide (D) is not particularly limited, but is preferably 0.1 to 5 ⁇ m, more preferably 0.2 to 0.5 ⁇ m. Titanium dioxide (D) can be used individually by 1 type or in combination of 2 or more types as appropriate. Further, for example, those having different particle size distributions and average particle sizes can be used in appropriate combination.
- the average particle diameter (D50) means a median diameter, and is a value in which the larger side and the smaller side are equivalent when the particle size distribution of the measured powder is divided into two. . More specifically, the particle size distribution of the powder is measured by a wet laser diffraction / scattering particle size distribution measuring device, and the volume is integrated from small particles to mean a value when 50% of the total volume is reached. .
- titanium dioxide (D) is surface-treated with SiO 2 , Al 2 O 3 , ZrO 2 and / or ZnO, in other words In this case, it is preferable to have a coating layer containing SiO 2 , Al 2 O 3 , ZrO 2 and / or ZnO.
- titanium dioxide (D) is surface-treated with SiO 2 , Al 2 O 3 , ZrO 2 and / or ZnO, and then treated with polyol, silane coupling agent and / or amine, in other words , SiO 2 , Al 2 O 3 , ZrO 2 and / or ZnO, and more preferably have a coating layer treated with polyol, silane coupling agent and / or amine.
- the surface-treated titanium dioxide (D) When the surface-treated titanium dioxide (D) is used, 0.5 to 15 parts by mass of SiO 2 and 0.5 to 15 of Al 2 O 3 with respect to 100 parts by mass of the total amount of titanium dioxide (D).
- 0.5 to 15 parts by mass of ZrO 2 and / or 0.5 to 15 parts by mass of ZnO are included, more preferably 1 to 11 parts by mass of SiO 2 and 1 of Al 2 O 3.
- the surface treatment amount By making the surface treatment amount within such a preferable range, discoloration due to heat treatment or light irradiation treatment is further suppressed without causing an excessive decrease in light reflectance in the ultraviolet light region and visible light region, and the light reflectance is reduced. The decrease tends to be further suppressed.
- the titanium dioxide (D) preferably contains 3 to 11 parts by mass of SiO 2 and 1 to 3 parts by mass of Al 2 O 3 with respect to 100 parts by mass of the total amount of titanium dioxide (D). .
- the content of titanium dioxide (D) in the resin composition of the present embodiment is not particularly limited, but is 10 to 10 parts per 100 parts by mass in total of the epoxy-modified silicone compound (A) and the branched imide resin (B).
- the amount is preferably 300 parts by mass, more preferably 50 to 250 parts by mass.
- the dispersion stabilizer generally used for coating materials can be used suitably.
- a copolymer-based wetting and dispersing agent is used.
- Specific examples thereof include polymer wetting and dispersing agents manufactured by Big Chemie Japan Co., Ltd., such as BYK-W903, BYK-W940, BYK-W996, BYK-W9010, Disper-BYK110, Disper-BYK111, Disper-BYK- 110, 111, 161, 180, etc. are mentioned, but not limited thereto.
- a dispersing agent (E) can be used individually by 1 type or in combination of 2 or more types as appropriate.
- content of the dispersing agent (E) in the resin composition of this embodiment is not specifically limited, It is 0.00 with respect to a total of 100 mass parts of an epoxy-modified silicone compound (A) and branched imide resin (B).
- the amount is preferably from 05 to 10 parts by mass, more preferably from 0.1 to 4.0 parts by mass, still more preferably from 0.5 to 3.0 parts by mass.
- the resin composition of the present embodiment may contain a monomer epoxy compound (F) having one or more epoxy rings as an epoxy compound other than the epoxy-modified silicone compound (A) described above.
- the type of the monomer epoxy compound (F) is not particularly limited as long as it is a monomer having one or more epoxy rings, but one monomer per molecule from the viewpoint of excellent heat resistance and light discoloration resistance. Or the monomer epoxy compound which has two epoxy rings is preferable.
- the monomer epoxy compound (F) can be used together with the phosphorus-based curing accelerator (C), but the phosphorus-based curing accelerator (C). Can be used in place of the phosphorus-based curing accelerator (C).
- monomer epoxy compound (F) compounds represented by the following formulas (8) to (10) are preferable.
- a monomer epoxy compound (F) can be used individually by 1 type or in combination of 2 or more types as appropriate.
- the molecular weight of the monomeric epoxy compound (F) is not particularly limited and can be appropriately set, but is preferably 80 to 800, more preferably 120 to 500.
- the monomeric epoxy compound (F) can be produced by a known method, and a commercially available product can be easily obtained.
- the compound represented by the above formula (8) includes, for example, DA-MGIC (manufactured by Shikoku Kasei Co., Ltd.), and the compound represented by the above formula (9) includes Celoxide 2021P (Daicel Chemical Industries).
- the compound represented by the above formula (10) includes Celoxide 2000 (manufactured by Daicel Chemical Industries, Ltd.) and the like.
- the content of the monomer epoxy compound (F) in the resin composition of the present embodiment is not particularly limited, but the storage stability of the prepreg, the moldability of the metal foil-clad laminate, the glass transition temperature, and the metal foil From the viewpoint of peel strength, it is preferably 0.05 to 25 parts by mass, more preferably 0.1 to 100 parts by mass with respect to 100 parts by mass in total of the epoxy-modified silicone compound (A) and the branched imide resin (B). 20 parts by mass, more preferably 0.5 to 15 parts by mass.
- the epoxy equivalent of the monomer epoxy compound (F) is not particularly limited, but from the viewpoint of improvement in manufacturability and suppression of viscosity increase of the resin, the epoxy equivalent to the carboxyl residue of the branched imide resin (B) is 0. It is preferably 1 to 0.8 equivalent.
- the resin composition of the present embodiment may contain an alicyclic epoxy resin (G) as an epoxy compound other than the epoxy-modified silicone compound (A) and the monomer epoxy compound (F) described above.
- an alicyclic epoxy resin (G) together with an epoxy-modified silicone compound (A), a branched imide resin (B), a phosphorus curing accelerator (C), titanium dioxide (D), and a dispersant (E).
- the light reflectance and the glass transition temperature tend to be increased by the heat treatment and the light irradiation treatment.
- this alicyclic epoxy resin (G) what carried out the ring-opening polymerization of the epoxy ring of the epoxy compound with alcohol is preferable.
- alcohol adducts of vinylcyclohexene diepoxide examples include alcohol adducts of 3,4-epoxycyclohexanecarboxylic acid-3 ′, 4′-epoxycyclohexylmethyl, alcohols of bis3,4-epoxycyclohexylmethyl adipate Adduct, alcohol adduct of dicyclopentadiene diepoxide, ⁇ -caprolactone modified bis (3,4-epoxycyclohexylmethyl) -4,5-epoxycyclohexane-1,2-dicarboxylic acid alcohol adduct, ⁇ -caprolactone modified Tetra (3,4-epoxycyclohexylmethyl) butane-tetracarboxylic acid alcohol adduct, dipentene dioxide alcohol adduct, 1,4-cyclooctadiene diepoxide alcohol adduct and bis (2,3-epoxy Examples include, but are not limited to, alcohol addition products of (
- the mass average molecular weight of the alicyclic epoxy resin (G) is not particularly limited and can be appropriately set, but is preferably 1500 to 10000, more preferably 2000 to 7000.
- the alicyclic epoxy resin (G) can be produced by a known method, and a commercially available product can be easily obtained.
- Examples of commercially available products include EHPE3150 (manufactured by Daicel Chemical Industries, Ltd.) and the like as a 2,2-bis (hydroxymethyl) -1-butanol adduct of vinylcyclohexene diepoxide.
- the content of the alicyclic epoxy resin (G) in the resin composition of the present embodiment is not particularly limited, but from the viewpoint of heat resistance and light discoloration resistance, the epoxy-modified silicone compound (A) and the branched imide resin (
- the total amount of B) is preferably 3 to 40 parts by mass, more preferably 5 to 30 parts by mass, and still more preferably 5 to 25 parts by mass.
- the resin composition of this embodiment is an aspect containing the monomer epoxy compound (F) and / or alicyclic epoxy resin (G) mentioned above, each in the resin composition of this embodiment
- the preferred content ratios of the components (A) to (G) are as follows.
- the content of the epoxy-modified silicone compound (A) in the resin composition is the epoxy-modified silicone compound (A), branched type, from the viewpoint of suppressing discoloration due to heat treatment and light irradiation treatment of the obtained metal foil-clad laminate.
- the amount is preferably 20 to 90 parts by mass, more preferably 30 to 80 parts by mass with respect to 100 parts by mass in total of the imide resin (B), the monomer epoxy compound (F) and the alicyclic epoxy resin (G). Part.
- the content of the branched imide resin (B) in the resin composition is epoxy-modified from the viewpoint of increasing the peel strength and heat resistance of the resulting metal foil-clad laminate and suppressing discoloration due to heat treatment and light irradiation treatment. 10 to 80 parts by mass with respect to 100 parts by mass in total of the silicone compound (A), the branched imide resin (B), the monomeric epoxy compound (F) and the alicyclic epoxy resin (G). The amount is preferably 15 to 70 parts by mass, and more preferably 20 to 60 parts by mass.
- the content of the phosphorus-based curing accelerator (C) in the resin composition is such that the epoxy-modified silicone compound (A), the branched imide resin ( B) is preferably 0.1 to 10 parts by weight, more preferably 0.5 to 8 parts by weight based on 100 parts by weight in total of the monomer epoxy compound (F) and the alicyclic epoxy resin (G). Part by mass.
- the content of titanium dioxide (D) in the resin composition is such that the epoxy-modified silicone compound (A), the branched imide resin (A), from the viewpoints of light reflectance, moldability and processability in the ultraviolet light region and visible light region ( B), preferably 10 to 400 parts by weight, more preferably 25 to 300 parts by weight with respect to a total of 100 parts by weight of the monomeric epoxy compound (F) and the alicyclic epoxy resin (G). More preferably, it is 100 to 250 parts by mass.
- the content of the dispersant (E) in the resin composition is such that the epoxy-modified silicone compound (A), the branched imide resin (B), the monomer epoxy compound (F), and the like from the viewpoints of heat resistance and dispersibility.
- the amount of the alicyclic epoxy resin (G) is preferably 0.05 to 5 parts by mass, more preferably 0.1 to 4.0 parts by mass, and still more preferably 0.5 to 5 parts by mass with respect to 100 parts by mass in total. 3.0 parts by mass.
- the content of the monomer epoxy compound (F) in the resin composition is determined from the viewpoint of the storage stability of the prepreg, the moldability of the metal foil-clad laminate, the glass transition temperature, and the peel strength with the metal foil.
- the amount is preferably 1 to 50 parts by mass with respect to 100 parts by mass in total of the compound (A), the branched imide resin (B), the monomeric epoxy compound (F) and the alicyclic epoxy resin (G). More preferably, it is 0.5 to 20 parts by mass, and still more preferably 1.0 to 10 parts by mass.
- the resin composition of the present embodiment may further contain a silane coupling agent (H) as necessary.
- the silanol group possessed by the silane coupling agent is excellent in affinity and reactivity with a material having a hydroxyl group on the surface, and therefore has an effect on the organic-inorganic bond, and the particle surface of the inorganic filler is combined with the silane coupling agent.
- the adhesiveness of a thermosetting resin and an inorganic filler is improved. Therefore, the combined use of the silane coupling agent (H) tends to improve the peel strength, elastic modulus, moisture absorption heat resistance, and cured product appearance of the obtained metal foil-clad laminate and printed wiring board. .
- silane coupling agent (H) used here what is generally used for the surface treatment of an inorganic substance can be used suitably, and the kind is not specifically limited.
- Specific examples thereof include aminosilanes such as ⁇ -aminopropyltriethoxysilane, N- ⁇ - (aminoethyl) - ⁇ -aminopropyltrimethoxysilane, epoxysilanes such as ⁇ -glycidoxypropyltrimethoxysilane, Examples include vinyl silanes such as ⁇ -methacryloxypropyltrimethoxysilane, cationic silanes such as N- ⁇ - (N-vinylbenzylaminoethyl) - ⁇ -aminopropyltrimethoxysilane hydrochloride, and phenylsilanes. . These can be used individually by 1 type or in combination of 2 or more types as appropriate.
- the content of the silane coupling agent (H) in the resin composition of the present embodiment can be appropriately set and is not particularly limited, but from the viewpoint of adhesion between the resin and the inorganic filler and the glass transition temperature,
- the amount is preferably 0.5 to 8 parts by mass, more preferably 1 to 5 parts by mass with respect to 100 parts by mass in total of the epoxy-modified silicone compound (A) and the branched imide resin (B).
- the resin composition of this embodiment is an epoxy resin (hereinafter simply referred to as an epoxy compound other than the epoxy-modified silicone compound (A), the monomer epoxy compound (F), and the alicyclic epoxy resin (G) described above. (It is also referred to as “other epoxy resin”).
- an epoxy resin if it is a compound which has a 2 or more epoxy group in 1 molecule, a well-known thing can be used, The kind is not specifically limited.
- bisphenol A type epoxy resin bisphenol E type epoxy resin, bisphenol F type epoxy resin, bisphenol S type epoxy resin, phenol novolac type epoxy resin, bisphenol A novolac type epoxy resin, cresol novolac type epoxy resin, biphenyl type epoxy resin, Naphthalene type epoxy resin, trifunctional phenol type epoxy resin, tetrafunctional phenol type epoxy resin, glycidyl ester type epoxy resin, phenol aralkyl type epoxy resin, biphenyl aralkyl type epoxy resin, aralkyl novolak type epoxy resin, naphthol aralkyl type epoxy resin, di Cyclopentadiene type epoxy resins, polyol type epoxy resins, isocyanurate ring-containing epoxies or their halides, etc. It is below.
- Other epoxy resins can be used singly or in appropriate combination of two or more.
- the resin composition of the present embodiment may contain other inorganic fillers in addition to the above-described titanium dioxide (D) as necessary.
- inorganic fillers those generally used in laminate applications can be suitably used.
- other inorganic fillers are preferably silicas and talc, from the viewpoint of electrical properties. More preferably, silicas are used.
- the other inorganic filler illustrated here can be used individually by 1 type or in combination of 2 or more types as appropriate.
- the average particle diameter (D50) of these other inorganic fillers is not particularly limited, but is 0.1 to 5 ⁇ m in consideration of dispersibility, flow characteristics during molding, and breakage when using a small diameter drill bit. It is preferably 0.2 to 3 ⁇ m.
- the content of these other inorganic fillers is not particularly limited, but is 1 to 300 parts by mass with respect to 100 parts by mass in total of the epoxy-modified silicone compound (A) and the branched imide resin (B).
- the amount is preferably 5 to 250 parts by mass or less.
- the resin composition of the present embodiment may contain a curing accelerator other than the phosphorus-based curing accelerator (C) described above, if necessary.
- a curing accelerator other than the phosphorus-based curing accelerator (C) described above, if necessary.
- the type of curing accelerator is not particularly limited as long as it is known in the art and is generally used.
- the curing accelerator include organic metal salts such as copper, zinc, cobalt, nickel, and manganese, imidazoles and derivatives thereof, and tertiary amines.
- the content of the curing accelerator can be appropriately adjusted and is not particularly limited.
- the epoxy-modified silicone compound (A) and the branched imide resin (B) The total amount of the monomer epoxy compound (F) and the alicyclic epoxy resin (G) is usually about 0.01 to 15 parts by mass, more preferably 0.02 to about 100 parts by mass. 3 parts by mass.
- the resin composition of the present embodiment may contain components other than those described above as long as the desired properties are not impaired.
- optional blends include various thermosetting compounds other than those described above, thermoplastic resins and oligomers thereof, various polymer compounds such as elastomers, flame retardant compounds, and various additives. These are not particularly limited as long as they are commonly used in the industry.
- flame retardant compounds include bromine compounds such as 4,4′-dibromobiphenyl, phosphorus compounds such as phosphate esters, melamine phosphate, and phosphorus-containing epoxy resins, nitrogen-containing compounds such as melamine and benzoguanamine, and oxazine rings Compound etc. are mentioned.
- additives include ultraviolet absorbers, antioxidants, photopolymerization initiators, fluorescent brighteners, photosensitizers, dyes, pigments, thickeners, lubricants, antifoaming agents, dispersants, and leveling agents. , Brighteners, polymerization inhibitors and the like. These arbitrary formulations can be used singly or in appropriate combination of two or more.
- the resin composition of this embodiment may contain a solvent as necessary.
- a solvent for example, when an organic solvent is used, the viscosity at the time of preparing the resin composition can be lowered, so that the handling property is improved and the impregnation property to the glass cloth is enhanced.
- the type of the solvent is not particularly limited as long as it can dissolve or be compatible with the mixture of the epoxy-modified silicone compound (A) and the branched imide resin (B) described above.
- ketones such as acetone, methyl ethyl ketone, methyl isobutyl ketone and cyclohexanone
- aromatic hydrocarbons such as benzene, toluene and xylene
- amides such as dimethylformamide and dimethylacetamide
- propylene glycol methyl ether and its acetate it is not particularly limited to these.
- a solvent can be used individually by 1 type or in combination of 2 or more types as appropriate.
- the resin composition of this embodiment can be prepared according to a conventional method.
- the preparation method is not particularly limited as long as it is a method by which a resin composition uniformly containing D), the dispersant (E), and other optional components described above is obtained.
- an epoxy-modified silicone compound (A), a branched imide resin (B), a phosphorus curing accelerator (C), titanium dioxide (D), and a dispersant (E) are sequentially blended in a solvent and sufficiently stirred.
- the resin composition of the present embodiment can be easily prepared.
- an organic solvent can be used as necessary.
- the kind of the organic solvent is not particularly limited as long as it can dissolve or be compatible with the mixture of the epoxy-modified silicone resin (A) and the branched imide resin (B). Specific examples thereof are as described above.
- a known process for uniformly dissolving or dispersing each component can be performed.
- a method for stirring and dispersing the resin composition of titanium dioxide (D) dispersibility in the resin composition is enhanced by performing stirring and dispersing treatment using a stirring tank provided with a stirrer having an appropriate stirring ability.
- the above stirring, mixing, and kneading treatment can be appropriately performed using, for example, a known device such as a ball mill, a bead mill or the like, or a revolution / spinning type mixing device.
- the branched imide resin (B), the phosphorus curing accelerator (C) and the monomer epoxy compound (F) are dissolved in a solvent, and the boiling point of the solvent
- the pretreatment for refluxing is performed, an increase in the molecular weight of the resulting resin composition can be controlled, and an excessive increase in viscosity can be suppressed.
- a branched imide resin (B) having an isocyanurate ring and a carboxyl group under a nitrogen atmosphere at normal pressure, a monomer epoxy compound (C), and a phosphorus curing accelerator (F) are converted into propylene glycol methyl ether.
- the solvent used for such pretreatment is not particularly limited, and examples thereof include aromatic hydrocarbons such as toluene and xylene, amides such as dimethylformamide, propylene glycol methyl ether and acetate thereof. These can be used individually by 1 type or in combination of 2 or more types as appropriate.
- the prepreg of the present embodiment can be obtained by combining the above resin composition with a base material, specifically, impregnating or applying the above resin composition to the base material.
- the method for producing the prepreg can be performed according to a conventional method, and is not particularly limited.
- the base material is impregnated with a resin composition containing an epoxy-modified silicone compound (A), a branched imide resin (B), a phosphorus curing accelerator (C), titanium dioxide (D), and a dispersant (E).
- the prepreg of this embodiment can be produced by semi-curing (B stage formation) by heating in a dryer at 100 to 200 ° C. for 1 to 30 minutes.
- the prepreg of this embodiment is not particularly limited, but the amount of the resin composition (including titanium dioxide (D) and other inorganic fillers) with respect to the total amount of prepreg is in the range of 30 to 90% by mass. It is preferable.
- the substrate used in the prepreg of the present embodiment is not particularly limited, and known materials used for various printed wiring board materials may be appropriately selected and used depending on the intended use and performance. Can do. Specific examples include glass fibers such as E glass, D glass, L glass, S glass, Q glass, spherical glass, NE glass, and T glass, inorganic fibers other than glass such as quartz and barium titanate, polyimide, and polyamide. Examples thereof include organic fibers such as polyester, and woven fabrics such as liquid crystal polyester, but are not particularly limited thereto.
- a base material can be used individually by 1 type or in combination of 2 or more types as appropriate.
- the weaving method of woven fabric is known as plain weave, Nanako weave, twill weave, etc. It doesn't matter.
- a base material can be used individually by 1 type or in combination of 2 or more types as appropriate.
- a glass woven fabric subjected to ultra-opening treatment or a glass woven fabric subjected to a plugging treatment is preferably used.
- a glass woven fabric surface-treated with a silane coupling agent such as epoxy silane treatment or amino silane treatment is preferred from the viewpoint of moisture absorption heat resistance.
- a liquid crystal polyester woven fabric is preferable from the viewpoint of electrical characteristics.
- the thickness and mass of the base material are not particularly limited, but usually about 0.01 to 0.3 mm is preferably used.
- the substrate is preferably a glass woven fabric having a thickness of 200 ⁇ m or less and a mass of 250 g / m 2 or less, and more preferably a glass woven fabric made of E-glass glass fibers. Used when producing prepreg.
- the metal foil-clad laminate of this embodiment is obtained by laminating and forming at least one prepreg as described above and arranging metal foil on one or both sides thereof. Specifically, one or a plurality of the prepregs described above are stacked, a metal foil such as copper or aluminum is placed on one or both sides thereof, and laminated, thereby producing the metal foil-clad laminate of this embodiment. be able to.
- the metal foil used here will not be specifically limited if it is used for printed wiring board material, Copper foil, such as a rolled copper foil and an electrolytic copper foil, is preferable. Considering the conductor loss in the high frequency region, an electrolytic copper foil having a small mat surface roughness is more preferable.
- the thickness of the metal foil is not particularly limited, but is preferably 2 to 70 ⁇ m, more preferably 2 to 35 ⁇ m.
- a multi-stage press machine, a multi-stage vacuum press machine, a continuous molding machine, an autoclave molding machine, etc. can be used for forming a metal foil-clad laminate, the temperature is 100 to 300 ° C., the pressure is a surface pressure of 2 to 100 kgf / cm 2 and the heating time is generally in the range of 0.05 to 5 hours.
- a multilayer board can be formed by combining and molding the prepreg of the present embodiment and a separately prepared wiring board for an inner layer.
- a method for producing a multilayer board for example, a 35 ⁇ m copper foil is disposed on both surfaces of one prepreg described above, laminated under the above conditions, an inner layer circuit is formed, and blackening treatment is performed on this circuit.
- the inner circuit board is then formed, and then the inner circuit board and the prepreg are alternately arranged one by one, and the copper foil is further disposed on the outermost layer, and preferably laminated under the above conditions, preferably under vacuum How to do is known.
- the metal foil-clad laminate of this embodiment not only has excellent heat resistance, but also has a high light reflectance in the ultraviolet light region and the visible light region, and also has a light reflectance of heat treatment and light irradiation treatment.
- the decrease is small, and the peel strength with the metal foil is excellent, and in a preferred embodiment, the heat resistance during moisture absorption is also excellent. Therefore, the metal foil-clad laminate of this embodiment can be used particularly effectively as a printed wiring board that requires such performance, particularly as a printed wiring board for LED mounting.
- the metal foil-clad laminate of the present embodiment described above can be suitably used as a printed wiring board by forming a predetermined wiring pattern.
- the printed wiring board can be manufactured according to a conventional method, and the manufacturing method is not particularly limited.
- an example of the manufacturing method of a printed wiring board is shown. First, a metal foil clad laminate such as the copper clad laminate described above is prepared. Next, an etching process is performed on the surface of the metal foil-clad laminate to form an inner layer circuit, thereby producing an inner layer substrate.
- the inner layer circuit surface of the inner layer substrate is subjected to a surface treatment to increase the adhesive strength as necessary, then the required number of the prepregs are stacked on the inner layer circuit surface, and a metal foil for the outer layer circuit is stacked on the outer surface. Then, it is integrally molded by heating and pressing. In this way, a multilayer laminate is produced in which an insulating layer made of a cured material of the base material and the thermosetting resin composition is formed between the inner layer circuit and the metal foil for the outer layer circuit. Next, after drilling for the through holes and via holes in the multilayer laminate, a plated metal film is formed on the wall surface of the hole to connect the inner layer circuit and the metal foil for the outer layer circuit. A printed wiring board is manufactured by performing an etching process on the metal foil for forming an outer layer circuit.
- the printed wiring board obtained in the above production example has an insulating layer and a conductor layer formed on the surface of the insulating layer, and the insulating layer includes the above-described resin composition of the present embodiment. That is, the prepreg of the present embodiment described above (the base material and the resin composition of the present embodiment impregnated or applied thereto), the layer of the resin composition of the metal foil-clad laminate of the present embodiment described above (the present embodiment).
- the layer made of the resin composition is composed of an insulating layer containing the resin composition of the present embodiment.
- part means “part by mass” unless otherwise specified.
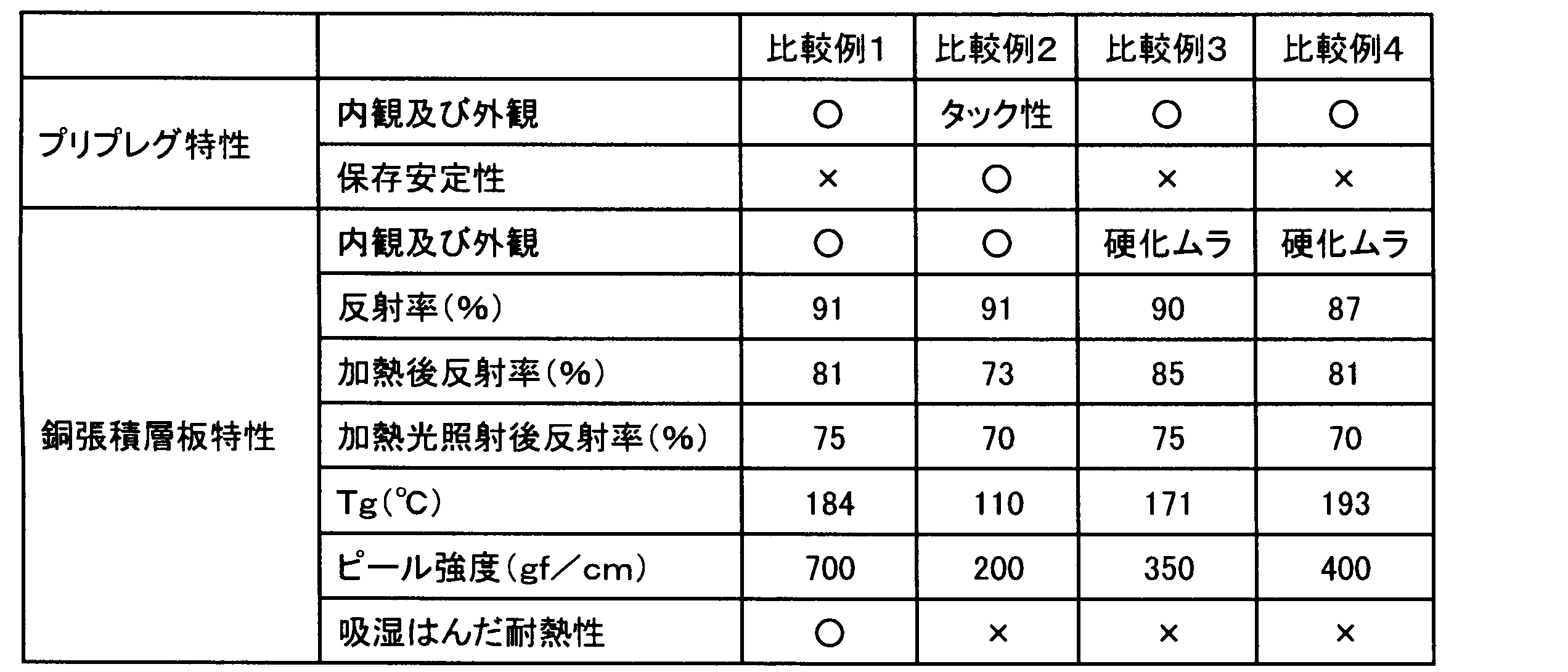
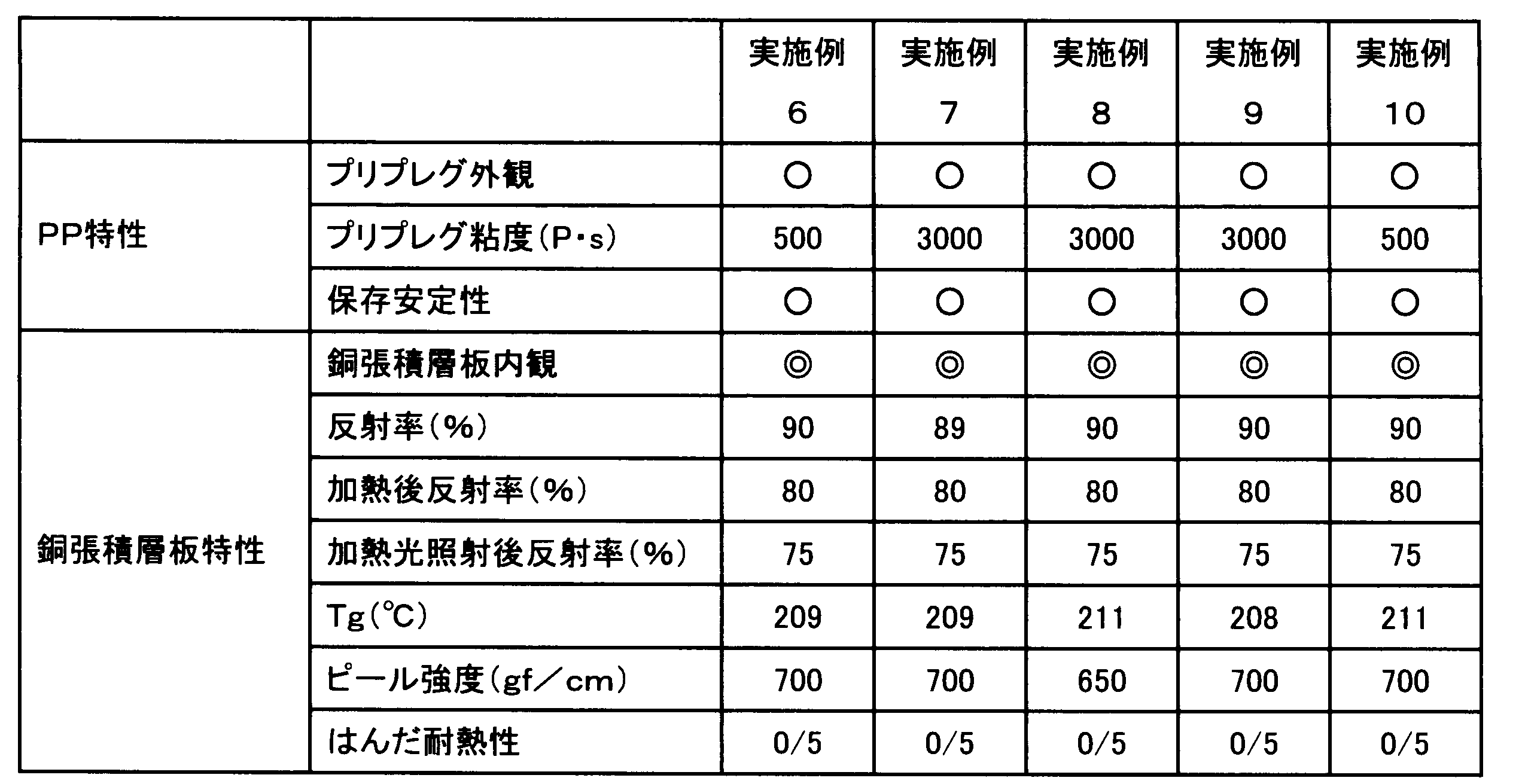
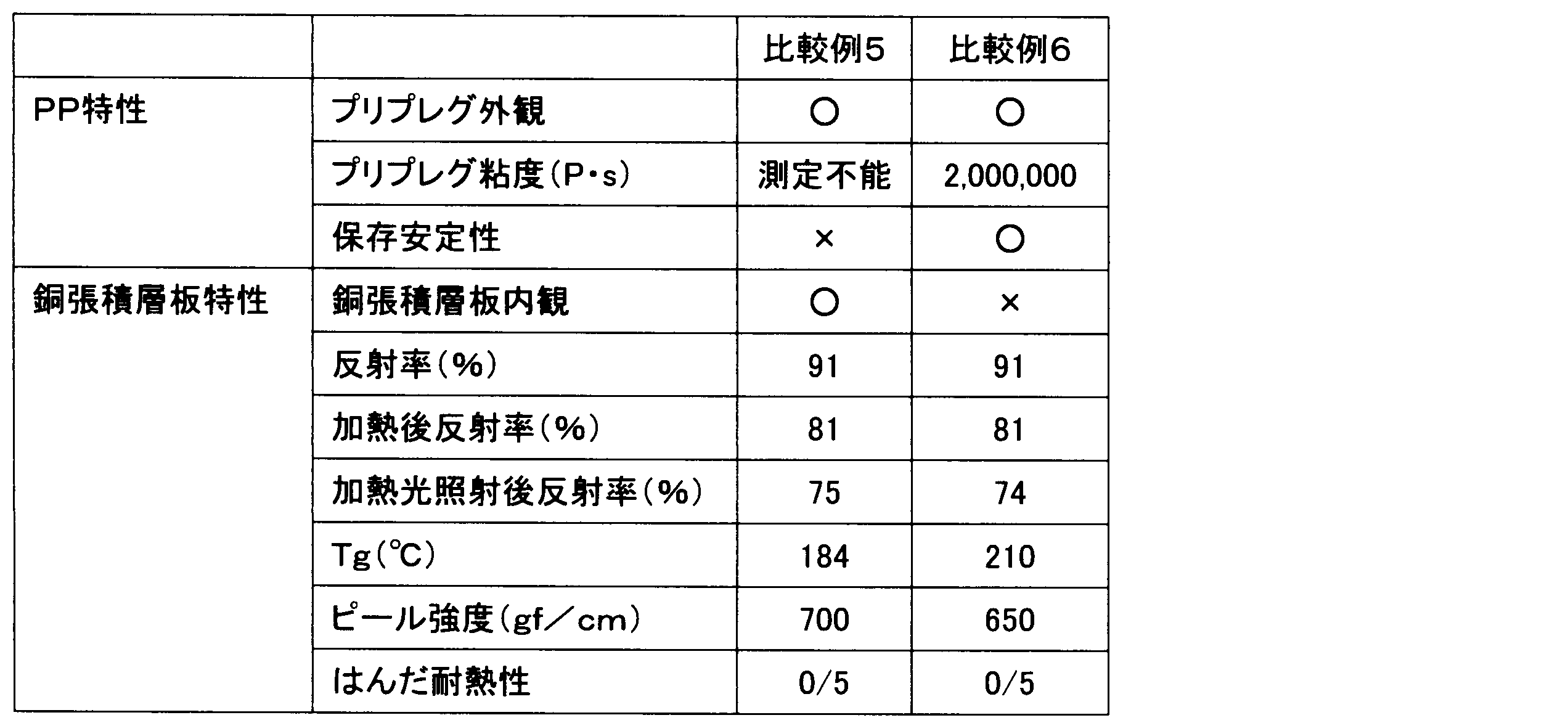
- Example 1 Epoxy-modified branched imide having 55 parts by mass of an aliphatic epoxy-modified silicone compound represented by the following formula (11) (X-40-2670 (manufactured by Shin-Etsu Chemical Co., Ltd.)) and a mass average molecular weight (Mw) of 33,000.
- Compound (ELG-941 (acid value 35 mgKOH / g), manufactured by DIC Corporation) 45 parts by mass, as a phosphorus curing accelerator, methyltributylphosphonium dimethyl phosphate (PX-4MP (manufactured by Nippon Chemical Industry Co., Ltd.)) 5 Parts by mass, titanium dioxide (CR90 (1 to 5 parts by mass of SiO 2 treatment and 1 to 3 parts by mass of Al 2 O 3 treatment with respect to 100 parts by mass of titanium dioxide), manufactured by Ishihara Sangyo Co., Ltd.) 200 parts by mass, 1.75 parts by mass of a wetting and dispersing agent (BYK-W903, manufactured by Big Chemie Japan Co., Ltd.), a silane coupling agent (Z6040, Toray Dowco) 3 parts by mass were stirred and mixed with a homomixer to obtain a varnish.
- PX-4MP manufactured by Nippon Chemical Industry Co., Ltd.
- titanium dioxide CR90 (1 to 5 parts by mass of SiO 2 treatment
- This varnish was diluted with methyl ethyl ketone at an equal mass ratio, impregnated into an E glass cloth having a thickness of 0.08 mm, and heated at 150 ° C. for 3 minutes to obtain a prepreg having a resin composition amount of 48 mass%. .
- an electrolytic copper foil JTC-LPZ foil, manufactured by Nikko Materials Co., Ltd.
- a double-sided copper foil-clad laminate having a thickness of about 0.2 mm was obtained by pressure molding using a vacuum press machine under a vacuum of 30 kgf / cm 2 and a surface pressure of 30 mmHg for 150 minutes.
- Example 2 Except for changing the compounding amount of the epoxy-modified silicone compound to 45 parts by mass and the compounding amount of the epoxy-modified branched imide compound to 55 parts by mass, respectively, the same procedure as in Example 1 was performed to prepare a varnish, prepreg and double-sided A copper foil-clad laminate was obtained.
- Example 3 The compounding amount of the epoxy-modified silicone compound was changed to 55 parts by mass, the compounding amount of the epoxy-modified branched imide compound was changed to 35 parts by mass, and 2,2-bis (hydroxymethyl) -1-butanol was used as the alicyclic epoxy resin.
- the varnish was prepared in the same manner as in Example 2 except that 10 parts by mass of 1,2-epoxy-4- (2-oxiranyl) cyclohexane adduct (EHPE-3150 (manufactured by Daicel Chemical Industries, Ltd.)) was further added.
- EHPE-3150 manufactured by Daicel Chemical Industries, Ltd.
- Example 4 Instead of the epoxy-modified branched imide compound, 25 parts by mass of an amine-modified branched imide compound having a mass average molecular weight (Mw) of 4800 (ELG-1301 (acid value 120 mgKOH / g), manufactured by DIC Corporation) is used. A varnish was prepared in the same manner as in Example 1 except that 20 parts by mass of the alicyclic epoxy resin used in Example 3 was further blended as a resin to obtain a prepreg and a double-sided copper foil-clad laminate.
- Mw mass average molecular weight
- Example 5 Instead of the amine-modified branched imide compound, 25 parts by mass of an alcohol-modified branched imide compound having a mass average molecular weight (Mw) of 3500 (ELG-1302 (acid value 70.8 mgKOH / g), manufactured by DIC Corporation) is used. Except for this, the same procedure as in Example 4 was performed to prepare a varnish to obtain a prepreg and a double-sided copper foil-clad laminate.
- Mw mass average molecular weight
- Comparative Example 2 A varnish was prepared in the same manner as in Comparative Example 1 except that 45 parts by mass of the epoxy-modified branched imide compound used in Example 1 was used instead of the unmodified branched imide compound. A laminate was obtained.
- Comparative Example 3 A varnish was prepared in the same manner as in Comparative Example 2 except that 1 part by mass of an aluminum-based curing accelerator (ALCH-TR (manufactured by Kawaken Fine Chemical Co., Ltd.)) was further blended, and a prepreg and a double-sided copper foil-clad laminate. I got a plate.
- ALCH-TR aluminum-based curing accelerator
- Comparative Example 4 Use 1 part by weight of a cationic curing accelerator (Sun-Aid-SI-150L (manufactured by Sanshin Chemical Industry Co., Ltd.)) instead of an aluminum curing accelerator (ALCH-TR (manufactured by Kawaken Fine Chemical Co., Ltd.)). Otherwise, the same procedure as in Comparative Example 3 was followed to prepare a varnish to obtain a prepreg and a double-sided copper foil-clad laminate.
- a cationic curing accelerator (Sun-Aid-SI-150L (manufactured by Sanshin Chemical Industry Co., Ltd.)) instead of an aluminum curing accelerator (ALCH-TR (manufactured by Kawaken Fine Chemical Co., Ltd.)
- the measuring method and evaluation method of each test method are as follows.
- JTC-LPZ foil 12 ⁇ m thick electrolytic copper foil
- Reflectance after heating light irradiation The sample obtained in 4) above was subjected to ultraviolet rays on a 180 ° C. hot plate installed in a weathering tester dryer (SUV-F11, manufactured by Iwasaki Electric Co., Ltd.).
- a measurement sample was obtained in which the surface copper foil was left.
- Hygroscopic solder heat resistance The obtained double-sided copper foil-clad laminate was cut into a size of 50 mm ⁇ 50 mm with a dicing saw, and then 3/4 of the copper foil on the surface was removed by etching to obtain a measurement sample. Using this measurement sample, moisture absorption was performed with a pressure cooker tester PCT (120 ° C./2 atm) for 3 hours, and the appearance change after immersion in a solder bath at 260 ° C. for 1 minute was visually observed. The number of tests was three, and no abnormalities were observed in all three “no abnormality: ( ⁇ )”, and even one that was swollen was evaluated as “blowing occurrence: ( ⁇ )”. The evaluation results are shown in Tables 1 and 2.
- Alcohol-modified multi-branched imide resin (ELG-1302, DIC Corporation) having 55 parts by mass of an aliphatic epoxy-modified silicone compound (X-40-2670, manufactured by Shin-Etsu Chemical Co., Ltd.) and a weight average molecular weight (Mw) of 3500 ), Acid value: 70.8 mg KOH / g) 20 parts by mass, monomer epoxy compound represented by the following formula (8) (DA-MGIC, manufactured by Shikoku Kasei Co., Ltd.), 5 parts by mass, alicyclic epoxy 20 parts by mass of 2,2-bis (hydroxymethyl) -1-butanol adduct of vinylcyclohexene diepoxide (EHPE3150, manufactured by Daicel Corporation) as a resin, methyltributylphosphonium dimethyl phosphate (PX- 4MP (manufactured by Nippon Chemical Industry Co., Ltd.) 3 parts by mass, titanium dioxide (CR90 (total 100 parts by mass of titanium dioxide
- This varnish was diluted with methyl ethyl ketone at the same mass ratio, impregnated into an E glass cloth having a thickness of 0.08 mm, and heated at 150 ° C. for 3 minutes to obtain a prepreg having a resin composition amount of 50% by mass. .
- an electrolytic copper foil JTC-LPZ foil, manufactured by Nikko Materials Co., Ltd.
- a double-sided copper foil-clad laminate having a thickness of about 0.2 mm was obtained by pressure molding using a vacuum press machine for 150 minutes under a vacuum of a surface pressure of 35 kgf / cm 2 and 30 mmHg or less.
- Example 7 The amount of monomer epoxy compound (DA-MGIC, manufactured by Shikoku Kasei Co., Ltd.) is 7.5 parts by mass, and the amount of alicyclic epoxy resin (EHPE3150, manufactured by Daicel Corp.) is 17.5 masses.
- the varnish was prepared in the same manner as in Example 6 except that the amount of titanium dioxide (CR90, manufactured by Ishihara Sangyo Co., Ltd.) was changed to 200 parts by mass, respectively, and a prepreg and double-sided copper foil-clad laminate were prepared. I got a plate.
- Example 8 Instead of the monomer epoxy compound (DA-MGIC, manufactured by Shikoku Kasei Co., Ltd.), 5 parts by mass of Celoxide 2021P (produced by Daicel Chemical Industries, Ltd.) represented by the following formula (9) was used, and titanium dioxide (CR90 A varnish was prepared in the same manner as in Example 6 except that the blending amount of Ishihara Sangyo Co., Ltd. was changed to 200 parts by mass to obtain a prepreg and a double-sided copper-clad laminate.
- DA-MGIC monomer epoxy compound manufactured by Shikoku Kasei Co., Ltd.
- Celoxide 2021P produced by Daicel Chemical Industries, Ltd.
- titanium dioxide (CR90 A varnish was prepared in the same manner as in Example 6 except that the blending amount of Ishihara Sangyo Co., Ltd. was changed to 200 parts by mass to obtain a prepreg and a double-sided copper-clad laminate.
- Example 9 Example 8 except that the amount of the alicyclic epoxy resin (EHPE3150, manufactured by Daicel Corporation) is changed to 19 parts by mass, and the amount of the monomer epoxy compound (Celoxide 2021P) is changed to 1 part by mass. In the same manner as above, a varnish was prepared to obtain a prepreg and a double-sided copper-clad laminate.
- EHPE3150 manufactured by Daicel Corporation
- Example 10 27 parts by mass of a multi-branched imide resin (ELG-1302, manufactured by DIC Corporation, acid value: 70.8 mgKOH / g) having a mass average molecular weight (Mw) of 3500, a single amount represented by the following formula (10) 3 parts by mass of an epoxy compound (Celoxide 2000, manufactured by Daicel Corporation), and 3 parts by mass of methyltributylphosphonium dimethyl phosphate (PX-4MP (manufactured by Nippon Chemical Industry Co., Ltd.)) as a phosphorus-based curing accelerator
- PX-4MP methyltributylphosphonium dimethyl phosphate
- Bis (hydroxymethyl) -1-butanol adduct (EHPE3150, manufactured by Daicel Corporation) 15 parts by mass, titanium dioxide (CR 0 (with respect to 100 parts by weight of titanium dioxide, SiO 2 process 1 to 5 parts by weight, and Al 2 O 3 process is 1-3 parts by weight), manufactured by Ishihara Sangyo Kaisha, Ltd.)) 180 parts by weight, wet 1.75 parts by mass of a dispersing agent (BYK-W903, manufactured by Big Chemie Japan Co., Ltd.) and 3 parts by mass of a silane coupling agent (Z6040, manufactured by Toray Dow Corning Co., Ltd.) were mixed with stirring in a homomixer and varnished.
- a prepreg and a double-sided copper-clad laminate were obtained in the same manner as in Example 6 except that the varnish thus obtained was used.
- the measuring method and evaluation method of each test method are as follows.
- the measurement conditions were a heater temperature of 120 ° C., a die hole diameter of 1.0 mm, a die hole length of 1.0 mm, a piston cross-sectional area of 1 cm 2 and a load of 0.5 kg. 3) Storage stability: The obtained prepreg was heat-treated in a hot air dryer at 40 ° C. for 120 hours. Two prepregs after the heat treatment were superposed, and 12 ⁇ m thick electrolytic copper foil (JTC-LPZ foil, manufactured by Nikko Materials Co., Ltd.) was placed on the upper and lower surfaces of the resulting laminate.
- JTC-LPZ foil manufactured by Nikko Materials Co., Ltd.
- Reflectance after irradiation with heating light The sample obtained in 5) above was subjected to ultraviolet rays on a 180 ° C. hot plate installed in a weathering tester dryer (SUV-F11, manufactured by Iwasaki Electric Co., Ltd.).
- the present invention can be used widely and effectively in various applications such as electric / electronic materials, machine tool materials, and aviation materials that require light resistance and heat resistance.
- the present invention can be effectively used in the fields of printed wiring boards and printed wiring boards for LED mounting, which require high peel strength with respect to heat resistance and heat resistance.
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Organic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Health & Medical Sciences (AREA)
- Materials Engineering (AREA)
- Manufacturing & Machinery (AREA)
- Microelectronics & Electronic Packaging (AREA)
- Textile Engineering (AREA)
- Wood Science & Technology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Inorganic Chemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Compositions Of Macromolecular Compounds (AREA)
- Epoxy Resins (AREA)
- Reinforced Plastic Materials (AREA)
- Laminated Bodies (AREA)
Description
<1> エポキシ変性シリコーン化合物(A)、イソシアヌレート基及びカルボキシル基を有する分岐型イミド樹脂(B)、リン系硬化促進剤(C)、二酸化チタン(D)及び分散剤(E)を含有する、樹脂組成物。
<2> 前記分岐型イミド樹脂(B)が、エポキシ変性分岐型イミド樹脂、アルコール変性分岐型イミド樹脂及びアミン変性分岐型イミド樹脂よりなる群から選択される少なくとも1種である、上記<1>に記載の樹脂組成物。
<3> 前記分岐型イミド樹脂(B)は、10~200mgKOH/gの酸価を有する、上記<1>又は<2>に記載の樹脂組成物。
<4> 前記分岐型イミド樹脂(B)は、10~100mgKOH/gの酸化を有する、上記<1>~<3>のいずれか一項に記載の樹脂組成物。
<6> 前記単量体エポキシ化合物(F)のエポキシ当量が、前記分岐型イミド樹脂(B)のカルボキシル残基に対して、0.1~0.8当量である、上記<5>に記載の樹脂組成物。
<7> 前記単量体エポキシ化合物(F)が、下記式(8)~(10):
<9> 前記エポキシ変性シリコーン化合物(A)が、下記式(1)で表される繰り返し単位を有し、1分子中に少なくとも3個のR′を有し、且つ、アルコキシ基を含有しないシリコーン化合物である、
上記<1>~<8>のいずれか一項に記載の樹脂組成物。
<10> 前記エポキシ変性シリコーン化合物(A)が、前記成分(A)及び(B)の合計100質量部に対して、20~80質量部含まれる、上記<1>~<9>のいずれか一項に記載の樹脂組成物。
上記<1>~<10>のいずれか一項に記載の樹脂組成物。
<13> 前記リン系硬化促進剤(C)が、前記成分(A)及び(B)の合計100質量部に対して、0.1~10質量部含まれる、上記<1>~<12>のいずれか一項に記載の樹脂組成物。
<14> 前記二酸化チタン(D)が、前記成分(A)及び(B)の合計100質量部に対して、10~300質量部含まれる、上記<1>~<13>のいずれか一項に記載の樹脂組成物。
<15> 前記二酸化チタン(D)は、5μm以下の平均粒径を有する、上記<1>~<14>のいずれか一項に記載の樹脂組成物。
<16> 前記二酸化チタン(D)は、SiO2、Al2O3、ZrO2、及び/又は、ZnOで表面処理されたものであり、前記二酸化チタン(D)の総量100質量部に対して、SiO2が0.5~15質量部、Al2O3が0.5~15質量部、ZrO2が0.5~15質量部、及び/又は、ZnOが0.5~15質量部含まれる、上記<1>~<15>のいずれか一項に記載の樹脂組成物。
<18> 前記分散剤(E)は、酸価が20~200mgKOH/gの高分子湿潤分散剤である、上記<1>~<17>のいずれか一項に記載の樹脂組成物。
<20> 前記脂環式エポキシ樹脂(G)が、ビニルシクロヘキセンジエポキシドのアルコール付加物、3,4-エポキシシクロヘキサンカルボン酸-3’,4’-エポキシシクロヘキシルメチルのアルコール付加物、アジピン酸ビス3,4-エポキシシクロヘキシルメチルのアルコール付加物、ジシクロペンタジエンジエポキシドのアルコール付加物、ε-カプロラクトン変性ビス(3,4-エポキシシクロヘキシルメチル)-4,5-エポキシシクロヘキサン-1,2-ジカルボン酸のアルコール付加物、ε-カプロラクトン変性テトラ(3,4-エポキシシクロヘキシルメチル)ブタン-テトラカルボン酸のアルコール付加物、ジペンテンジオキシドのアルコール付加物、1,4-シクロオクタジエンジエポキシドのアルコール付加物及びビス(2,3-エポキシシクロペンチル)エーテルのアルコール付加物よりなる群から選択される少なくとも1種である、上記<19>に記載の樹脂組成物。
<23> 前記二酸化チタン(D)が、前記エポキシ変性シリコーン化合物(A)、前記分枝型イミド樹脂(B)、前記単量体エポキシ化合物(F)、及び前記脂環式エポキシ樹脂(G)の合計100質量部に対して、10~400質量部含まれる、上記<22>に記載の樹脂組成物。
<24> 前記分散剤(E)が、前記エポキシ変性シリコーン化合物(A)、前記分枝型イミド樹脂(B)、前記単量体エポキシ化合物(F)、及び前記脂環式エポキシ樹脂(G)の合計100質量部に対して、0.05~5質量部含まれる、上記<22>又は<23>に記載の樹脂組成物。
<25> エポキシ変性シリコーン化合物(A)、イソシアヌレート基及びカルボキシル基を有する分岐型イミド樹脂(B)、二酸化チタン(D)、分散剤(E)及びエポキシ環を一以上有する、前記(A)以外の単量体エポキシ化合物(F)を含有する、樹脂組成物。この樹脂組成物においても、上記<2>~<20>で述べた具体的態様であることが好ましい。
<27> 上記<1>~<25>のいずれか一項に記載の樹脂組成物を基材に含浸又は塗布してなる、プリプレグ。
<28> 上記<27>に記載のプリプレグを少なくとも1枚以上重ね、その片面もしくは両面に金属箔を配して積層成形して得られる、金属箔張積層板。
<29> 絶縁層と、前記絶縁層の表面に形成された導体層とを含むプリント配線板であって、前記絶縁層が上記<1>~<25>のいずれか一項に記載の樹脂組成物を含む、プリント配線板。
下記式(11)で表される脂肪族エポキシ変性シリコーン化合物(X-40-2670(信越化学工業(株)製))55質量部、質量平均分子量(Mw)が33000であるエポキシ変性分岐型イミド化合物(ELG-941(酸価35mgKOH/g)、DIC(株)製)45質量部、リン系硬化促進剤として、メチルトリブチルホスホニウムジメチルホスフェート(PX-4MP(日本化学工業(株)製))5質量部、二酸化チタン(CR90(二酸化チタンの合計100質量部に対して、SiO2処理が1~5質量部、及びAl2O3処理が1~3質量部)、石原産業(株)製)200質量部、湿潤分散剤(BYK-W903、ビックケミー・ジャパン(株)製)1.75質量部、シランカップリング剤(Z6040、東レ・ダウコーニング(株)製))3質量部をホモミキサーにて攪拌混合しワニスを得た。このワニスをメチルエチルケトンを用いて質量基準で等倍に希釈し、厚さ0.08mmのEガラスクロスに含浸させ、150℃で3分加熱させて、樹脂組成物量が48質量%のプリプレグを得た。次に、このプリプレグを2枚重ね合わせ、得られた積層体の上下面に厚さ12μmの電解銅箔(JTC-LPZ箔、(株)日鉱マテリアルズ製)を配置し、その後、温度220℃、面圧30kgf/cm2、30mmHg以下の真空下で150分間、真空プレス機を用いて加圧成型することにより、厚さ約0.2mmの両面銅箔張積層板を得た。
エポキシ変性シリコーン化合物の配合量を45質量部に、エポキシ変性分岐型イミド化合物の配合量を55質量部にそれぞれ変更すること以外は、実施例1と同様に行い、ワニスを調製し、プリプレグ及び両面銅箔張積層板を得た。
エポキシ変性シリコーン化合物の配合量を55質量部に、エポキシ変性分岐型イミド化合物の配合量を35質量にそれぞれ変更し、脂環式エポキシ樹脂として2,2-ビス(ヒドロキシメチル)-1-ブタノールの1,2-エポキシ-4-(2-オキシラニル)シクロヘキサン付加物(EHPE-3150(ダイセル化学工業(株)製)10質量部をさらに配合すること以外は、実施例2と同様に行い、ワニスを調製し、プリプレグ及び両面銅箔張積層板を得た。
エポキシ変性分岐型イミド化合物に代えて質量平均分子量(Mw)が4800であるアミン変性分岐型イミド化合物(ELG-1301(酸価120mgKOH/g)、DIC(株)製)25質量部を用い、エポキシ樹脂として実施例3で用いた脂環式エポキシ樹脂20質量部をさらに配合すること以外は、実施例1と同様に行い、ワニスを調製し、プリプレグ及び両面銅箔張積層板を得た。
アミン変性分岐型イミド化合物に代えて質量平均分子量(Mw)が3500であるアルコール変性分岐型イミド化合物(ELG-1302(酸価70.8mgKOH/g)、DIC(株)製)25質量部を用いること以外は、実施例4と同様に行い、ワニスを調製し、プリプレグ及び両面銅箔張積層板を得た。
エポキシ変性分岐型イミド化合物に代えて質量平均分子量が11000である無変性の多分岐型イミド化合物((V-8002(酸価127mgKOH/g)、DIC(株)製)45質量部を用い、リン系硬化促進剤の配合を省略すること以外は、実施例1と同様に行い、ワニスを調製し、プリプレグ及び両面銅箔張積層板を得た。
無変性の分岐型イミド化合物に代えて実施例1で用いたエポキシ変性分岐型イミド化合物45質量部を用いること以外は、比較例1と同様に行い、ワニスを調製し、プリプレグ及び両面銅箔張積層板を得た。
アルミニウム系硬化促進剤(ALCH-TR(川研ファインケミカル(株)製))1質量部をさらに配合すること以外は、比較例2と同様に行い、ワニスを調製し、プリプレグ及び両面銅箔張積層板を得た。
アルミニウム系硬化促進剤(ALCH-TR(川研ファインケミカル(株)製))に代えてカチオン系硬化促進剤(サンエイド-SI-150L(三新化学工業(株)製))1質量部を用いること以外は、比較例3と同様に行い、ワニスを調製し、プリプレグ及び両面銅箔張積層板を得た。
(測定方法及び評価方法)
1)プリプレグ外観:得られたプリプレグ表面のタック性を指触にて確認した。ここでは、タック性が良好で、製品として使用することができるものを「○」と、タック性が不良で、製品として使用することができないものを「タック性」と評価した。
2)保存安定性:得られたプリプレグを40℃の熱風乾燥機で120時間加熱処理した。この熱処理後のプリプレグを2枚重ね合わせ、得られた積層体の上下面に厚さ12μmの電解銅箔(JTC-LPZ箔、(株)日鉱マテリアルズ製)を配置し、温度220℃、面圧30kgf/cm2、30mmHg以下の真空下で150分間、真空プレス機を用いて加圧成型し、厚さ0.2mmの両面銅張積層板の作製を試みた。そして、樹脂組成物のフローが良好であったものを○と、樹脂組成物のフローが不良で硬化ムラが確認されたものを「硬化ムラ」と評価した。
3)銅張積層板内観及び外観:得られた両面銅張積層板をダイシングソーでサイズ50×50mmに切断し、表面の銅箔をエッチングにより除去した後、研磨機を用いて研磨することで、両面銅張積層板の断面を露出させた測定用サンプルを得た。測定用サンプルの外観を目視観察するとともに、光学顕微鏡を用いて測定用サンプル断面のSEM観察を行ない、樹脂の欠落がなければ○、欠落がある場合を×とした。
4)反射率:得られた両面銅箔張積層板をダイシングソーでサイズ50×50mmに切断後、表面の銅箔をエッチングにより除去し、測定用サンプルを得た。この測定用サンプルを、JIS Z-8722に基づき、分光測色計(コニカミノルタホールディングス(株)製:CM3610d)を用いて、457nmでの反射率を測定した(n=5の平均値)。
5)加熱後反射率:上記4)で得られたサンプルを、180℃の熱風乾燥機で24時間加熱処理した後、上記4)の反射率の測定と同様にして、反射率を測定した(n=5の平均値)。
6)加熱光照射後反射率:上記4)で得られたサンプルを、耐候試験機乾燥機(SUV-F11、岩崎電気(株)製)内に設置された180℃のホットプレート上で、紫外線(波長:295~450nm)照射度100mW/cm2、24時間の加熱光照射処理(紫外線照射度100mW/cm2)を行った後、上記4)の反射率の測定と同様にして、反射率を測定した(n=5の平均値)。
7)Tg:得られた両面銅箔張積層板をダイシングソーでサイズ12.7×2.5mmに切断後、表面の銅箔をエッチングにより除去し、測定用サンプルを得た。この測定用サンプルを用い、JIS C6481に準拠して動的粘弾性分析装置(TAインスツルメント製)でDMA法によりガラス転移温度を測定した(n=3の平均値)。
8)ピール強度:得られた両面銅箔張積層板をダイシングソーでサイズ10×100mmに切断後、表面の銅箔を残した測定用サンプルを得た。JIS C6481のプリント配線板用銅張積層板試験方法(5.7 引き剥がし強さ参照。)に準じ、オートグラフ((株)島津製作所製:AG-IS)を用いて、測定用サンプルの銅箔の引き剥がし強度を測定した(n=5の平均値)。
9)吸湿はんだ耐熱性:得られた両面銅箔張積層板をダイシングソーでサイズ50mm×50mmに切断後、表面の銅箔の3/4をエッチングにより除去し、測定用サンプルを得た。この測定用サンプルを用い、プレッシャークッカー試験器PCT(120℃/2気圧)で3時間吸湿させた後、260℃の半田槽の中に1分浸漬した後の外観変化を目視で観察した。試験回数は3回とし、3つとも異常が認められなかったもの「異常なし:(○)」、1つでも膨れが発生したものを「膨れ発生:(×)」と評価した。
評価結果を、表1及び2に示す。
脂肪族エポキシ変性シリコーン化合物(X-40-2670、信越化学工業(株)製)55質量部、質量平均分子量(Mw)が3500であるアルコール変性多分枝型イミド樹脂(ELG-1302、DIC(株)製、酸価:70.8mgKOH/g)20質量部、下記式(8)で表される単量体エポキシ化合物(DA-MGIC、四国化成(株)製)5質量部、脂環式エポキシ樹脂としてビニルシクロヘキセンジエポキシドの2,2-ビス(ヒドロキシメチル)-1-ブタノール付加物(EHPE3150、ダイセル(株)製)20質量部、リン系硬化促進剤として、メチルトリブチルホスホニウムジメチルホスフェート(PX-4MP(日本化学工業(株)製)3質量部、二酸化チタン(CR90(二酸化チタンの合計100質量部に対して、SiO2処理が1~5質量部、及びAl2O3処理が1~3質量部)、石原産業(株)製))180質量部、湿潤分散剤(BYK-W903、ビックケミー・ジャパン(株)製)1.75質量部、シランカップリング剤(Z6040、東レ・ダウコーニング(株)製))3質量部をホモミキサーにて攪拌混合してワニスを得た。このワニスをメチルエチルケトンを用いて質量基準で等倍に希釈し、厚さ0.08mmのEガラスクロスに含浸させ、150℃で3分加熱させて、樹脂組成物量が50質量%のプリプレグを得た。次に、このプリプレグを2枚重ね合わせ、得られた積層板の上下面に厚さ12μmの電解銅箔(JTC-LPZ箔、(株)日鉱マテリアルズ製)を配置し、その後、温度230℃、面圧35kgf/cm2、30mmHg以下の真空下で150分間、真空プレス機を用いて加圧成型することにより、厚さ約0.2mmの両面銅箔張積層板を得た。
単量体エポキシ化合物(DA-MGIC、四国化成(株)製)の配合量を7.5質量部に、脂環式エポキシ樹脂(EHPE3150、ダイセル(株)製)の配合量を17.5質量部に、二酸化チタン(CR90、石原産業(株)製)の配合量を200質量部にそれぞれ変更すること以外は、実施例6と同様にして、ワニスを調製し、プリプレグ及び両面銅箔張積層板を得た。
単量体エポキシ化合物(DA-MGIC、四国化成(株)製)に代えて下記式(9)で表されるセロキサイド2021P(ダイセル化学工業(株)製)5質量部を用い、二酸化チタン(CR90、石原産業(株)製)の配合量を200質量部に変更すること以外は、実施例6と同様にして、ワニスを調製し、プリプレグ及び両面銅張積層板を得た。
脂環式エポキシ樹脂(EHPE3150、ダイセル(株)製)の配合量を19質量部に、単量体エポキシ化合物(セロキサイド2021P)の配合量を1質量部にそれぞれ変更すること以外は、実施例8と同様にして、ワニスを調製し、プリプレグ及び両面銅張積層板を得た。
質量平均分子量(Mw)が3500である多分枝型イミド樹脂(ELG-1302、DIC(株)製、酸価:70.8mgKOH/g)27質量部、下記式(10)で表される単量体エポキシ化合物(セロキサイド2000、ダイセル(株)製)3質量部、リン系硬化促進剤として、メチルトリブチルホスホニウムジメチルホスフェート(PX-4MP(日本化学工業(株)製)3質量部を溶媒の存在下で還流する前処理を行なった後に、脂肪族エポキシ変性シリコーン化合物(X-40-2670、信越化学工業(株)製)55質量部、脂環式エポキシ化合物としてビニルシクロヘキセンジエポキシドの2,2-ビス(ヒドロキシメチル)-1-ブタノール付加物(EHPE3150、ダイセル(株)製)15質量部、二酸化チタン(CR90(二酸化チタンの合計100質量部に対して、SiO2処理が1~5質量部、及びAl2O3処理が1~3質量部)、石原産業(株)製))180質量部、湿潤分散剤(BYK-W903、ビックケミー・ジャパン(株)製)1.75質量部、シランカップリング剤(Z6040、東レ・ダウコーニング(株)製))3質量部をホモミキサーにて攪拌混合しワニスを得た。
このようにして得られたワニスを用いること以外は、実施例6と同様にして、プリプレグ及び両面銅張積層板を得た。
脂肪族エポキシ変性シリコーン化合物(X-40-2670(信越化学工業(株)製))55質量部、質量平均分子量が11000であるエポキシ変性分岐型イミド化合物(V-8002、DIC(株)製、酸価:127mgKOH/g)45質量部、二酸化チタン(CR90、石原産業(株)製)200質量部、湿潤分散剤(BYK-W903、ビックケミー・ジャパン(株)製)1.75質量部、シランカップリング剤(Z6040、東レ・ダウコーニング(株)製))3質量部をホモミキサーにて攪拌混合しワニスを得た。このワニスを使用すること以外は、実施例6と同様に行い、プリプレグ及び厚さ0.2mmの両面銅箔張積層板を得た。
脂肪族エポキシ変性シリコーン化合物(X-40-2670、信越化学工業(株)製)55質量部、質量平均分子量(Mw)が4800である無変性の多分枝型イミド樹脂(ELG-1224、DIC(株)製、酸価:90.0mgKOH/g)20質量部、脂環式エポキシ樹としてビニルシクロヘキセンジエポキシドの2,2-ビス(ヒドロキシメチル)-1-ブタノール付加物(EHPE3150、ダイセル(株)製)25質量部、二酸化チタン(CR90(二酸化チタンの合計100質量部に対して、SiO2処理が1~5質量部、及びAl2O3処理が1~3質量部)、石原産業(株)製))180質量部、湿潤分散剤(BYK-W903、ビックケミー・ジャパン(株)製)1.75質量部、シランカップリング剤(Z6040、東レ・ダウコーニング(株)製))3質量部をホモミキサーにて攪拌混合しワニスを得た。このワニスを使用すること以外は、実施例6と同様に行い、プリプレグ及び両面銅箔張積層板を得た。
(測定方法及び評価方法)
1)プリプレグ外観:得られたプリプレグ表面のタック性を指触にて確認した。ここでは、タック性が良好で、製品として使用することができるものを「○」と、タック性が不良で、製品として使用することができないものを「タック性」と評価した。
2)プリプレグ粘度:得られたプリプレグの粘度をフローテスター(SHIMADZU(株)製:CFT-500D)を用いて測定した。測定条件は、ヒーター温度120℃,ダイ穴直径1.0mm,ダイ穴長1.0mm,ピストン断面積1cm2,荷重0.5kgとした。
3)保存安定性:得られたプリプレグを40℃の熱風乾燥機で120時間加熱処理した。この熱処理後のプリプレグを2枚重ね合わせ、得られた積層体の上下面に厚さ12μmの電解銅箔(JTC-LPZ箔、(株)日鉱マテリアルズ製)を配置し、温度220℃、面圧30kgf/cm2、30mmHg以下の真空下で150分間、真空プレス機を用いて加圧成型し、厚さ0.2mmの両面銅張積層板の作製を試みた。そして、樹脂組成物のフローが良好であったものを○と、樹脂組成物のフローが不良で硬化ムラが確認されたものを「硬化ムラ」と評価した。
4)銅張積層板内観:得られた両面銅張積層板をダイシングソーでサイズ50×50mmに切断し、表面の銅箔をエッチングにより除去した後、研磨機を用いて研磨することで、両面銅張積層板の断面を露出させた測定用サンプルを得た。光学顕微鏡を用いて、測定用サンプル断面のSEM観察を行ない、樹脂の欠落がなければ○、欠落がある場合を×とした。
5)反射率:得られた両面銅箔張積層板をダイシングソーでサイズ50×50mmに切断後、表面の銅箔をエッチングにより除去し、測定用サンプルを得た。この測定用サンプルを、JIS Z-8722に基づき、分光測色計(コニカミノルタホールディングス(株)製:CM3610d)を用いて、457nmでの反射率を測定した(n=5の平均値)。
6)加熱後反射率:上記5)で得られたサンプルを、180℃の熱風乾燥機で24時間加熱処理した後、上記5)の反射率の測定と同様にして、反射率を測定した(n=5の平均値)。
7)加熱光照射後反射率:上記5)で得られたサンプルを、耐候試験機乾燥機(SUV-F11、岩崎電気(株)製)内に設置された180℃のホットプレート上で、紫外線(波長:295~450nm)照射度100mW/cm2、24時間の加熱光照射処理(紫外線照射度100mW/cm2)を行った後、上記5)の反射率の測定と同様にして、反射率を測定した(n=5の平均値)。
8)Tg:得られた両面銅箔張積層板をダイシングソーでサイズ12.7×2.5mmに切断後、表面の銅箔をエッチングにより除去し、測定用サンプルを得た。この測定用サンプルを用い、JIS C6481に準拠して動的粘弾性分析装置(TAインスツルメント製)でDMA法によりガラス転移温度を測定した(n=3の平均値)。
9)ピール強度:得られた両面銅箔張積層板をダイシングソーでサイズ10×100mmに切断後、表面の銅箔を残した測定用サンプルを得た。JIS C6481のプリント配線板用銅張積層板試験方法(5.7 引き剥がし強さ参照。)に準じ、オートグラフ((株)島津製作所製:AG-IS)を用いて、J測定サンプルの銅箔の引き剥がし強度を測定した(n=5の平均値)。
10)吸湿はんだ耐熱性:得られた両面銅箔張積層板をダイシングソーでサイズ50mm×50mmに切断後、表面の銅箔を残した測定用サンプルを得た。この測定用サンプルを用い、280℃の半田槽の中に30分浸漬した後の外観変化を、目視で観察した(フクレ発生数/試験数:n=5)。
評価結果を表3及び4に示す。
Claims (29)
- エポキシ変性シリコーン化合物(A)、イソシアヌレート基及びカルボキシル基を有する分岐型イミド樹脂(B)、リン系硬化促進剤(C)、二酸化チタン(D)及び分散剤(E)を含有する、
樹脂組成物。 - 前記分岐型イミド樹脂(B)が、エポキシ変性分岐型イミド樹脂、アルコール変性分岐型イミド樹脂及びアミン変性分岐型イミド樹脂よりなる群から選択される少なくとも1種である、
請求項1に記載の樹脂組成物。 - 前記分岐型イミド樹脂(B)は、10~200mgKOH/gの酸価を有する、
請求項1又は2に記載の樹脂組成物。 - 前記分岐型イミド樹脂(B)は、10~100mgKOH/gの酸化を有する、
請求項1~3のいずれか一項に記載の樹脂組成物。 - エポキシ環を一以上有する、上記(A)以外の単量体エポキシ化合物(F)をさらに含有する、
請求項1~4のいずれか一項に記載の樹脂組成物。 - 前記単量体エポキシ化合物(F)のエポキシ当量が、前記分岐型イミド樹脂(B)のカルボキシル残基に対して、0.1~0.8当量である、
請求項5に記載の樹脂組成物。 - 前記エポキシ変性シリコーン化合物(A)が、脂肪族エポキシ変性シリコーン化合物である、
請求項1~7のいずれか一項に記載の樹脂組成物。 - 前記エポキシ変性シリコーン化合物(A)が、前記成分(A)及び(B)の合計100質量部に対して、20~80質量部含まれる、
請求項1~9のいずれか一項に記載の樹脂組成物。 - シランカップリング剤(H)をさらに含有する、
請求項1~11のいずれか一項に記載の樹脂組成物。 - 前記リン系硬化促進剤(C)が、前記成分(A)及び(B)の合計100質量部に対して、0.1~10質量部含まれる、
請求項1~12のいずれか一項に記載の樹脂組成物。 - 前記二酸化チタン(D)が、前記成分(A)及び(B)の合計100質量部に対して、10~300質量部含まれる、
請求項1~13のいずれか一項に記載の樹脂組成物。 - 前記二酸化チタン(D)は、5μm以下の平均粒径を有する、
請求項1~14のいずれか一項に記載の樹脂組成物。 - 前記二酸化チタン(D)は、SiO2、Al2O3、ZrO2、及び/又は、ZnOで表面処理されたものであり、前記二酸化チタン(D)の総量100質量部に対して、SiO2が0.5~15質量部、Al2O3が0.5~15質量部、ZrO2が0.5~15質量部、及び/又は、ZnOが0.5~15質量部含まれる、
請求項1~15のいずれか一項に記載の樹脂組成物。 - 前記分散剤(E)が、前記成分(A)及び(B)の合計100質量部に対して、0.05~10質量部含まれる、
請求項1~16のいずれか一項に記載の樹脂組成物。 - 前記分散剤(E)は、酸価が20~200mgKOH/gの高分子湿潤分散剤である、
請求項1~17のいずれか一項に記載の樹脂組成物。 - 脂環式エポキシ樹脂(G)をさらに含有する、
請求項1~18のいずれか一項に記載の樹脂組成物。 - 前記脂環式エポキシ樹脂(G)が、ビニルシクロヘキセンジエポキシドのアルコール付加物、3,4-エポキシシクロヘキサンカルボン酸-3’,4’-エポキシシクロヘキシルメチルのアルコール付加物、アジピン酸ビス3,4-エポキシシクロヘキシルメチルのアルコール付加物、ジシクロペンタジエンジエポキシドのアルコール付加物、ε-カプロラクトン変性ビス(3,4-エポキシシクロヘキシルメチル)-4,5-エポキシシクロヘキサン-1,2-ジカルボン酸のアルコール付加物、ε-カプロラクトン変性テトラ(3,4-エポキシシクロヘキシルメチル)ブタン-テトラカルボン酸のアルコール付加物、ジペンテンジオキシドのアルコール付加物、1,4-シクロオクタジエンジエポキシドのアルコール付加物及びビス(2,3-エポキシシクロペンチル)エーテルのアルコール付加物よりなる群から選択される少なくとも1種である、
請求項19に記載の樹脂組成物。 - 前記分岐型イミド樹脂(B)、前記リン系硬化促進剤(C)及び前記単量体エポキシ化合物(F)を溶媒の存在下で還流する前処理を行なった後に、エポキシ変性シリコーン化合物(A)、二酸化チタン(D)及び分散剤(E)を配合することで作製される、
請求項5に記載の樹脂組成物。 - エポキシ変性シリコーン化合物(A)、イソシアヌレート基及びカルボキシル基を有する分岐型イミド樹脂(B)、リン系硬化促進剤(C)、二酸化チタン(D)、分散剤(E)、エポキシ環を一以上有する上記(A)以外の単量体エポキシ化合物(F)、及び脂環式エポキシ樹脂(G)を含有し、
前記分枝型イミド樹脂(B)が、前記エポキシ変性シリコーン化合物(A)、前記分枝型イミド樹脂(B)、前記単量体エポキシ化合物(F)、及び前記脂環式エポキシ樹脂(G)の合計100質量部に対して、10~80質量部含まれる、
樹脂組成物。 - 前記二酸化チタン(D)が、前記エポキシ変性シリコーン化合物(A)、前記分枝型イミド樹脂(B)、前記単量体エポキシ化合物(F)、及び前記脂環式エポキシ樹脂(G)の合計100質量部に対して、10~400質量部含まれる、
請求項22に記載の樹脂組成物。 - 前記分散剤(E)が、前記エポキシ変性シリコーン化合物(A)、前記分枝型イミド樹脂(B)、前記単量体エポキシ化合物(F)、及び前記脂環式エポキシ樹脂(G)の合計100質量部に対して、0.05~5質量部含まれる、
請求項22又は23に記載の樹脂組成物。 - エポキシ変性シリコーン化合物(A)、イソシアヌレート基及びカルボキシル基を有する分岐型イミド樹脂(B)、二酸化チタン(D)、分散剤(E)及びエポキシ環を一以上有する、前記(A)以外の単量体エポキシ化合物(F)を含有する、
樹脂組成物。 - 請求項1~25のいずれか一項に記載の樹脂組成物からなる、
LED実装用プリント配線板用の樹脂組成物。 - 請求項1~25のいずれか一項に記載の樹脂組成物を基材に含浸又は塗布してなる、
プリプレグ。 - 請求項27に記載のプリプレグを少なくとも1枚以上重ね、その片面もしくは両面に金属箔を配して積層成形して得られる、
金属箔張積層板。 - 絶縁層と、前記絶縁層の表面に形成された導体層とを含むプリント配線板であって、前記絶縁層が請求項1~25のいずれか一項に記載の樹脂組成物を含む、
プリント配線板。
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| SG11201405224QA SG11201405224QA (en) | 2012-03-13 | 2013-03-12 | Resin composition, prepreg, and metal foil-clad laminate |
| EP13761906.0A EP2826800A4 (en) | 2012-03-13 | 2013-03-12 | RESIN COMPOSITION, PREPREG AND LAMINATE CASED WITH METAL FOIL |
| JP2014504930A JP6120102B2 (ja) | 2012-03-13 | 2013-03-12 | 樹脂組成物、プリプレグ及び金属箔張積層板 |
| CN201380014137.6A CN104245778B (zh) | 2012-03-13 | 2013-03-12 | 树脂组合物、预浸料以及覆金属箔层叠板 |
| US14/384,025 US20150056454A1 (en) | 2012-03-13 | 2013-03-12 | Resin composition, prepreg, and metal foil-clad laminate |
| KR1020147025478A KR101944577B1 (ko) | 2012-03-13 | 2013-03-12 | 수지 조성물, 프리프레그 및 금속박 피복 적층판 |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2012-055598 | 2012-03-13 | ||
| JP2012055598 | 2012-03-13 | ||
| JP2012131983 | 2012-06-11 | ||
| JP2012-131983 | 2012-06-11 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2013137257A1 true WO2013137257A1 (ja) | 2013-09-19 |
Family
ID=49161163
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2013/056817 Ceased WO2013137257A1 (ja) | 2012-03-13 | 2013-03-12 | 樹脂組成物、プリプレグ及び金属箔張積層板 |
Country Status (8)
| Country | Link |
|---|---|
| US (1) | US20150056454A1 (ja) |
| EP (1) | EP2826800A4 (ja) |
| JP (1) | JP6120102B2 (ja) |
| KR (1) | KR101944577B1 (ja) |
| CN (1) | CN104245778B (ja) |
| SG (1) | SG11201405224QA (ja) |
| TW (1) | TWI577545B (ja) |
| WO (1) | WO2013137257A1 (ja) |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2015147909A (ja) * | 2014-02-07 | 2015-08-20 | 太陽インキ製造株式会社 | 硬化性樹脂組成物、その硬化皮膜、およびこれを備えた加飾ガラス板 |
| JPWO2014027654A1 (ja) * | 2012-08-16 | 2016-07-28 | 三菱瓦斯化学株式会社 | 樹脂シート、樹脂層付き支持体、積層板及び金属箔張積層板 |
| WO2016157587A1 (ja) * | 2015-03-31 | 2016-10-06 | 太陽インキ製造株式会社 | 硬化性樹脂組成物、ドライフィルム、硬化物およびプリント配線板 |
| JP6963202B1 (ja) * | 2019-11-29 | 2021-11-05 | 石原産業株式会社 | 二酸化チタン微粒子の有機溶剤分散体及びその製造方法、並びにその用途 |
| JPWO2022239845A1 (ja) * | 2021-05-13 | 2022-11-17 | ||
| JP2024076959A (ja) * | 2022-11-25 | 2024-06-06 | 味の素株式会社 | 金属箔付き樹脂シート |
Families Citing this family (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2676999B1 (en) * | 2011-02-18 | 2018-10-17 | Mitsubishi Gas Chemical Company, Inc. | Resin composition, prepreg, and metal foil-clad laminate |
| CN105778506B (zh) | 2014-12-25 | 2019-04-30 | 广东生益科技股份有限公司 | 一种有机硅树脂组合物及使用它的预浸料、层压板、覆铜板以及铝基板 |
| KR102572390B1 (ko) * | 2015-07-06 | 2023-08-29 | 미츠비시 가스 가가쿠 가부시키가이샤 | 수지 조성물, 프리프레그, 레진 시트, 금속박 피복 적층판 및 프린트 배선판 |
| JP6826601B2 (ja) * | 2015-12-22 | 2021-02-03 | サイテック インダストリーズ インコーポレイテッド | 複合材部材のための離型性表面材 |
| TWI656172B (zh) * | 2017-09-18 | 2019-04-11 | 台燿科技股份有限公司 | 無溶劑之樹脂組合物及其應用 |
| JP7024672B2 (ja) * | 2018-09-12 | 2022-02-24 | 信越化学工業株式会社 | 熱硬化性樹脂組成物、熱硬化性樹脂フィルム及び半導体装置 |
| GB201911134D0 (en) * | 2019-08-05 | 2019-09-18 | Qinetiq Ltd | Materials and method |
| CN120020179B (zh) * | 2023-11-17 | 2025-11-21 | 广州立邦涂料有限公司 | 家装用低气味led-uv木器白底漆及其制备方法 |
| CN120432230B (zh) * | 2025-05-23 | 2026-03-24 | 丽水飞洲电缆有限公司 | 一种耐老化电缆及其制备方法 |
Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH0841168A (ja) * | 1994-07-27 | 1996-02-13 | Matsushita Electric Works Ltd | 液状エポキシ樹脂組成物及び半導体装置 |
| JPH10202789A (ja) | 1997-01-22 | 1998-08-04 | Shin Kobe Electric Mach Co Ltd | 積層板 |
| JP2008001880A (ja) | 2005-10-21 | 2008-01-10 | Mitsubishi Gas Chem Co Inc | プリプレグおよび銅張積層板 |
| JP2008297536A (ja) * | 2006-10-18 | 2008-12-11 | Hitachi Chem Co Ltd | 熱硬化性樹脂組成物、これを用いたフレキシブル基板及び電子部品 |
| JP2010089493A (ja) | 2008-09-11 | 2010-04-22 | Shin-Etsu Chemical Co Ltd | シリコーン積層基板、その製造方法、シリコーン積層基板製造用シリコーン樹脂組成物及びled装置 |
| JP2011093966A (ja) * | 2009-10-27 | 2011-05-12 | Panasonic Electric Works Co Ltd | 透明フィルム |
| WO2012165147A1 (ja) * | 2011-05-31 | 2012-12-06 | 三菱瓦斯化学株式会社 | 樹脂組成物、並びにこれを用いたプリプレグ及び金属箔張積層板 |
Family Cites Families (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5824149A (ja) * | 1981-08-06 | 1983-02-14 | Fuji Photo Film Co Ltd | 平版印刷用感光材料 |
| TW200427809A (en) * | 2003-05-19 | 2004-12-16 | Hitachi Chemical Co Ltd | Insulating material, film, circuit board and method for manufacture thereof |
| JP4941804B2 (ja) * | 2005-03-02 | 2012-05-30 | Dic株式会社 | エポキシ樹脂組成物、その硬化物、半導体封止材料、新規フェノール樹脂、および新規エポキシ樹脂 |
| JP4831992B2 (ja) * | 2005-04-08 | 2011-12-07 | ヘンケルエイブルスティックジャパン株式会社 | 透光性樹脂組成物 |
| JP2008189852A (ja) * | 2007-02-06 | 2008-08-21 | Taiyo Ink Mfg Ltd | 熱硬化性樹脂組成物及びそれを用いたプリント配線基板の製造方法 |
| EP2223969B1 (en) * | 2007-12-17 | 2015-02-25 | Mitsui Chemicals, Inc. | Resin composition, transparent member obtained from the resin composition, and use of the same |
| JP5449802B2 (ja) * | 2009-03-05 | 2014-03-19 | 新日鉄住金化学株式会社 | 脂環構造を有するエポキシシリコーン樹脂、及び硬化性樹脂組成物 |
| JPWO2010107045A1 (ja) * | 2009-03-18 | 2012-09-20 | Dic株式会社 | ポリイミド樹脂、硬化性樹脂組成物及びその硬化物 |
| JP2010254919A (ja) * | 2009-04-28 | 2010-11-11 | Hitachi Chem Co Ltd | 光反射用熱硬化性樹脂組成物、これを用いた光半導体素子搭載用基板及びその製造方法、並びに光半導体装置 |
| EP2540759B1 (en) * | 2009-05-29 | 2014-08-06 | DIC Corporation | Thermosetting resin composition and cured product of the same |
| JP5934463B2 (ja) * | 2009-07-07 | 2016-06-15 | 富士フイルム株式会社 | 遮光膜用着色組成物、遮光膜及び遮光パターンの形成方法、並びに固体撮像素子及びその製造方法 |
| AU2010276770A1 (en) * | 2009-07-27 | 2012-02-02 | Asahi Glass Company, Limited | Composite particle, composition for forming coating membrane, printing ink, coating material composition, coated article, and resin film with attached coating membrane |
| JP2011074355A (ja) * | 2009-09-07 | 2011-04-14 | Nitto Denko Corp | 光半導体装置用樹脂組成物およびそれを用いて得られる光半導体装置用リードフレーム、ならびに光半導体装置 |
| CN104619498B (zh) * | 2012-08-16 | 2016-08-24 | 三菱瓦斯化学株式会社 | 树脂片、带树脂层的支承体、层叠板和覆金属箔层叠板 |
-
2013
- 2013-03-12 JP JP2014504930A patent/JP6120102B2/ja active Active
- 2013-03-12 EP EP13761906.0A patent/EP2826800A4/en active Pending
- 2013-03-12 WO PCT/JP2013/056817 patent/WO2013137257A1/ja not_active Ceased
- 2013-03-12 KR KR1020147025478A patent/KR101944577B1/ko active Active
- 2013-03-12 SG SG11201405224QA patent/SG11201405224QA/en unknown
- 2013-03-12 US US14/384,025 patent/US20150056454A1/en not_active Abandoned
- 2013-03-12 CN CN201380014137.6A patent/CN104245778B/zh active Active
- 2013-03-13 TW TW102108853A patent/TWI577545B/zh active
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH0841168A (ja) * | 1994-07-27 | 1996-02-13 | Matsushita Electric Works Ltd | 液状エポキシ樹脂組成物及び半導体装置 |
| JPH10202789A (ja) | 1997-01-22 | 1998-08-04 | Shin Kobe Electric Mach Co Ltd | 積層板 |
| JP2008001880A (ja) | 2005-10-21 | 2008-01-10 | Mitsubishi Gas Chem Co Inc | プリプレグおよび銅張積層板 |
| JP2008297536A (ja) * | 2006-10-18 | 2008-12-11 | Hitachi Chem Co Ltd | 熱硬化性樹脂組成物、これを用いたフレキシブル基板及び電子部品 |
| JP2010089493A (ja) | 2008-09-11 | 2010-04-22 | Shin-Etsu Chemical Co Ltd | シリコーン積層基板、その製造方法、シリコーン積層基板製造用シリコーン樹脂組成物及びled装置 |
| JP2011093966A (ja) * | 2009-10-27 | 2011-05-12 | Panasonic Electric Works Co Ltd | 透明フィルム |
| WO2012165147A1 (ja) * | 2011-05-31 | 2012-12-06 | 三菱瓦斯化学株式会社 | 樹脂組成物、並びにこれを用いたプリプレグ及び金属箔張積層板 |
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPWO2014027654A1 (ja) * | 2012-08-16 | 2016-07-28 | 三菱瓦斯化学株式会社 | 樹脂シート、樹脂層付き支持体、積層板及び金属箔張積層板 |
| JP2015147909A (ja) * | 2014-02-07 | 2015-08-20 | 太陽インキ製造株式会社 | 硬化性樹脂組成物、その硬化皮膜、およびこれを備えた加飾ガラス板 |
| WO2016157587A1 (ja) * | 2015-03-31 | 2016-10-06 | 太陽インキ製造株式会社 | 硬化性樹脂組成物、ドライフィルム、硬化物およびプリント配線板 |
| JPWO2016157587A1 (ja) * | 2015-03-31 | 2018-01-25 | 太陽インキ製造株式会社 | 硬化性樹脂組成物、ドライフィルム、硬化物およびプリント配線板 |
| JP6963202B1 (ja) * | 2019-11-29 | 2021-11-05 | 石原産業株式会社 | 二酸化チタン微粒子の有機溶剤分散体及びその製造方法、並びにその用途 |
| JPWO2022239845A1 (ja) * | 2021-05-13 | 2022-11-17 | ||
| WO2022239845A1 (ja) * | 2021-05-13 | 2022-11-17 | パナソニックIpマネジメント株式会社 | 樹脂組成物、並びに、それを用いたプリプレグ、樹脂付きフィルム、樹脂付き金属箔、金属張積層板及び配線基板 |
| JP2024076959A (ja) * | 2022-11-25 | 2024-06-06 | 味の素株式会社 | 金属箔付き樹脂シート |
Also Published As
| Publication number | Publication date |
|---|---|
| CN104245778B (zh) | 2017-03-01 |
| JP6120102B2 (ja) | 2017-04-26 |
| TWI577545B (zh) | 2017-04-11 |
| JPWO2013137257A1 (ja) | 2015-08-03 |
| US20150056454A1 (en) | 2015-02-26 |
| TW201408478A (zh) | 2014-03-01 |
| EP2826800A4 (en) | 2015-11-04 |
| CN104245778A (zh) | 2014-12-24 |
| EP2826800A1 (en) | 2015-01-21 |
| KR20140133853A (ko) | 2014-11-20 |
| KR101944577B1 (ko) | 2019-01-31 |
| SG11201405224QA (en) | 2014-10-30 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6120102B2 (ja) | 樹脂組成物、プリプレグ及び金属箔張積層板 | |
| JP5888513B2 (ja) | 樹脂組成物、プリプレグ及び金属箔張積層板 | |
| WO2012132965A1 (ja) | 樹脂組成物、プリプレグ及び金属箔張積層板 | |
| JP6164500B2 (ja) | 樹脂シート、樹脂層付き支持体、積層板及び金属箔張積層板 | |
| KR102039874B1 (ko) | 프리프레그 및 이것을 사용한 적층판 그리고 프린트 배선판 | |
| EP2716710B1 (en) | Resin composition and prepreg and metal foil clad laminate using same | |
| JPWO2013146700A1 (ja) | 樹脂組成物、プリプレグ及び積層板 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 13761906 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14384025 Country of ref document: US |
|
| ENP | Entry into the national phase |
Ref document number: 2014504930 Country of ref document: JP Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 20147025478 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2013761906 Country of ref document: EP |