WO2013183617A1 - 糖液の製造方法 - Google Patents
糖液の製造方法 Download PDFInfo
- Publication number
- WO2013183617A1 WO2013183617A1 PCT/JP2013/065431 JP2013065431W WO2013183617A1 WO 2013183617 A1 WO2013183617 A1 WO 2013183617A1 JP 2013065431 W JP2013065431 W JP 2013065431W WO 2013183617 A1 WO2013183617 A1 WO 2013183617A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- membrane
- sugar
- sugar solution
- producing
- solution
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C13—SUGAR INDUSTRY
- C13K—SACCHARIDES OBTAINED FROM NATURAL SOURCES OR BY HYDROLYSIS OF NATURALLY OCCURRING DISACCHARIDES, OLIGOSACCHARIDES OR POLYSACCHARIDES
- C13K1/00—Glucose; Glucose-containing syrups
- C13K1/02—Glucose; Glucose-containing syrups obtained by saccharification of cellulosic materials
- C13K1/04—Purifying
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12P—FERMENTATION OR ENZYME-USING PROCESSES TO SYNTHESISE A DESIRED CHEMICAL COMPOUND OR COMPOSITION OR TO SEPARATE OPTICAL ISOMERS FROM A RACEMIC MIXTURE
- C12P19/00—Preparation of compounds containing saccharide radicals
- C12P19/02—Monosaccharides
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12P—FERMENTATION OR ENZYME-USING PROCESSES TO SYNTHESISE A DESIRED CHEMICAL COMPOUND OR COMPOSITION OR TO SEPARATE OPTICAL ISOMERS FROM A RACEMIC MIXTURE
- C12P19/00—Preparation of compounds containing saccharide radicals
- C12P19/14—Preparation of compounds containing saccharide radicals produced by the action of a carbohydrase (EC 3.2.x), e.g. by alpha-amylase, e.g. by cellulase, hemicellulase
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12P—FERMENTATION OR ENZYME-USING PROCESSES TO SYNTHESISE A DESIRED CHEMICAL COMPOUND OR COMPOSITION OR TO SEPARATE OPTICAL ISOMERS FROM A RACEMIC MIXTURE
- C12P7/00—Preparation of oxygen-containing organic compounds
- C12P7/40—Preparation of oxygen-containing organic compounds containing a carboxyl group including Peroxycarboxylic acids
- C12P7/56—Lactic acid
-
- C—CHEMISTRY; METALLURGY
- C13—SUGAR INDUSTRY
- C13K—SACCHARIDES OBTAINED FROM NATURAL SOURCES OR BY HYDROLYSIS OF NATURALLY OCCURRING DISACCHARIDES, OLIGOSACCHARIDES OR POLYSACCHARIDES
- C13K13/00—Sugars not otherwise provided for in this class
- C13K13/002—Xylose
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12P—FERMENTATION OR ENZYME-USING PROCESSES TO SYNTHESISE A DESIRED CHEMICAL COMPOUND OR COMPOSITION OR TO SEPARATE OPTICAL ISOMERS FROM A RACEMIC MIXTURE
- C12P2203/00—Fermentation products obtained from optionally pretreated or hydrolyzed cellulosic or lignocellulosic material as the carbon source
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E50/00—Technologies for the production of fuel of non-fossil origin
- Y02E50/10—Biofuels, e.g. bio-diesel
Definitions
- the present invention relates to a method for producing a sugar liquid from biomass.
- the object of the present invention is to prevent the above problems, namely, scale adhesion to fermentation apparatus, piping clogging, separation membrane clogging, pH / DO sensor malfunction occurrence and scale adhesion to separation membrane in continuous culture, and culture solution and fermentation.
- An object of the present invention is to provide a method for producing a sugar solution that enables membrane separation of products.
- the present invention intends to solve the above-mentioned problem, and the method for producing a sugar liquid according to the present invention adds an alkali to a cellulosic biomass concentrated sugar liquid, adjusts the pH to 7 or more, and contains at least magnesium.
- a method for producing a sugar solution comprising the steps of precipitating an insoluble substance, and filtering through a microfiltration membrane to remove the insoluble substance and obtaining a sugar liquid as a permeate.
- the adjustment of the cellulose-based biomass concentrated sugar liquid with an alkali is adjusted to pH 8 or more.
- the average pore diameter of the microfiltration membrane is in the range of 0.01 ⁇ m to 1 ⁇ m.
- the sugar liquid further comprises an additive selected from the group consisting of a nitrogen source, a metal salt, a vitamin, an amino acid, a sugar, an antifoaming agent and a surfactant. It is a manufacturing method.
- microorganisms are cultured using the sugar solution obtained by the above production method as a fermentation raw material to produce chemical products in the culture solution, and the microorganisms and chemical products are continuously or intermittently passed through the separation membrane. And filtered to recover the chemical.
- a simple method that is, an alkali is added to a cellulosic biomass concentrated sugar solution, the pH is adjusted to 7 or more, and an insoluble substance containing at least magnesium is added. After depositing, the insoluble material is filtered and removed through a microfiltration membrane to obtain a sugar solution as a permeate, thereby suppressing scale adhesion to the fermenter or separation membrane, which was the above-mentioned problem. can do. Furthermore, by carrying out this operation, a new effect of improving the fermentation yield can be obtained when the cellulosic biomass concentrated sugar liquid is used as a fermentation raw material.
- the method for producing a sugar liquid in the present invention can be used for producing a sugar liquid as a fermentation raw material from biomass containing cellulose. Moreover, the sugar liquid manufactured by this invention can be used as a fermentation raw material of various chemical products.
- FIG. 1 is a drawing showing a block flow of a method for producing a sugar solution of the present invention.
- FIG. 2 is a drawing showing a block flow of another method for producing a sugar liquid of the present invention.
- FIG. 3 is a chromatograph obtained by redissolving an insoluble substance in an aqueous hydrochloric acid solution and then separating it using ion chromatography.
- FIG. 4 is a schematic cross-sectional view for explaining the configuration of a simple module using a hollow fiber membrane for the method for producing a sugar liquid of the present invention.
- FIG. 5 is a side view showing an example of an apparatus used in the method for producing a sugar liquid of the present invention.
- FIG. 6 is a side view showing an example of another apparatus used in the method for producing a sugar liquid of the present invention.
- FIG. 7 is a side view showing an example of an apparatus for producing a chemical product using the sugar solution of the present invention as a fermentation raw material.
- the method for producing a sugar liquid according to the present invention includes a step of adding an alkali to a cellulosic biomass concentrated sugar liquid, adjusting the pH to 7 or more, and precipitating an insoluble substance containing at least magnesium, and filtering through a microfiltration membrane. And a method for producing a sugar solution comprising a step of removing the insoluble substance and obtaining a sugar solution as a permeate.
- FIG. 1 is a drawing showing a block flow of a method for producing a sugar liquid of the present invention.
- step (1) the step of adding an alkali to the cellulosic biomass concentrated sugar solution of the present invention, adjusting the pH to 7 or more, and precipitating an insoluble substance containing at least magnesium (step (1)) will be described.
- the cellulosic biomass concentrated sugar solution used in the present invention is an aqueous solution containing sugar obtained by hydrolyzing cellulosic biomass as a raw material, and is a step of concentrating by at least one concentration operation. It refers to the sugar solution that has passed.
- Cellulosic biomass here refers to biomass containing cellulose.
- cellulosic biomass examples include herbaceous biomass such as bagasse, switchgrass, napiergrass, Eliansus, corn stover, rice straw, straw, rice husk and coconut husk, or woody biomass such as trees, poplar and waste building materials Furthermore, it refers to biomass derived from the aquatic environment such as algae and seaweed.
- Such biomass contains lignin, which is an aromatic polymer, in addition to cellulose and hemicellulose (hereinafter sometimes referred to as “cellulose” as a generic term for cellulose and hemicellulose).
- sucrose solution refers to hydrolysis of the cellulose component or hemicellulose component contained in the cellulosic biomass by subjecting the cellulosic biomass to one or more of acid treatment, enzyme treatment, alkali treatment and pulverization treatment. It refers to what has been done. Further, not only those immediately after hydrolysis, but also those after adding microorganisms to the hydrolyzate and fermenting it, an aqueous solution containing sugar can be regarded as a sugar solution and used in the present invention. be able to.
- This hydrolyzate mainly contains 6 monosaccharides such as glucose and 5 monosaccharides such as sylose as sugars.
- the concentrated sugar liquid means a sugar liquid obtained by concentrating the cellulose biomass sugar liquid by a known method such as evaporation method or membrane concentration method. Moreover, the concentration method may combine various methods.
- the concentrated sugar solution may be a solution concentrated by the above-described concentration method, or a solution obtained by diluting a sugar obtained by removing water by concentration to a solid state by adding water or the like again.
- an operation of adding an alkali to the cellulosic biomass concentrated sugar solution and adjusting the pH to 7 or more to precipitate an insoluble substance containing at least magnesium is performed.
- ammonia As the alkali to be added, ammonia, aqueous ammonia, sodium hydroxide, potassium hydroxide and the like are preferably used.
- Alkaline such as calcium hydroxide can also be used as the alkali, but calcium, like magnesium, is a factor in the generation of scale, so there is no advantage of using it as an alkali.
- ammonia is particularly preferably used as the alkali.
- ammonium sulfate In the hydrolysis of cellulosic biomass, when sulfuric acid is used, the resulting hydrolyzate and the concentrated sugar solution often contain sulfate ions, and by adding ammonia, ammonium sulfate is produced as a salt. be able to. As is well known, ammonium sulfate can be effectively used as a nitrogen source in the growth of microorganisms, fermentation production, and the like. That is, ammonia is most preferable as the alkali used for the pH adjustment. By adjusting the pH to 7 or more, magnesium dissolved in the cellulosic biomass concentrated sugar liquid becomes magnesium hydroxide and can be precipitated as insoluble crystals.
- operations such as stirring and mixing may be performed in order to make the added alkali uniform.
- the precipitation of magnesium hydroxide may be appropriately performed by operations such as heat retention and cooling.
- the time for precipitation may be set as appropriate, but it is preferably 1 minute or longer, more preferably 5 minutes or longer, and most preferably 3 hours or longer.
- FIG. 2 is a drawing showing a block flow of another method for producing a sugar liquid of the present invention.
- FIG. 2 is a flow in which a period of transportation, storage / storage or transportation / storage / storage of the concentrated sugar liquid of cellulosic biomass is utilized in the step of depositing magnesium hydroxide. Since a certain period of time is required for the precipitation of the insoluble substance, these times can be effectively used for the purpose.
- the pH is adjusted with an alkali, the storage stability against microbial contamination and the like is improved.
- nutrients and auxiliary materials required when sugar solution is used as a fermentation raw material nitrogen sources, metal salts, vitamins, amino acids, sugars, antibiotics, surfactants and antifoaming agents Etc. may be added.
- Examples of the metal salt include molybdenum, cobalt, iron, copper, zinc, manganese, nickel, chromium, selenium, iodine, fluorine, silicon, and panadium.
- Examples of vitamins include vitamin B12, thiamine, biotin and vitamin B1.
- saccharides include glucose, arabinose, xylose, furucose, psicose, galactose, mannose, xylulose, threose, erythrose, ribose and the like.
- amino acids include alanine, arginine, asparagine, aspartic acid, cysteine, glutamine, glutamic acid, glycine, histidine, isoleucine, leucine, lysine, methionine, phenylalanine, proline, serine, threonine, tryptophan, tyrosine and valine. it can.
- antibiotics examples include tetracycline, ⁇ -lactam, aminoglycoside, macrolide, and chloramphenicol.
- surfactant examples include nonionic surfactants, anionic surfactants, and cationic surfactants.
- Such components are preferably added in advance at this stage, when the sugar solution is used as a fermentation raw material. This is intended to prevent the insoluble material from being produced again by adding these components to the sugar solution at the stage of fermentation.
- the cellulose-based biomass concentrated sugar solution used in the present invention is preferably a sugar solution concentrated using a nanofiltration membrane and / or a reverse osmosis membrane.
- the nanofiltration membrane is also called a nanofilter (nanofiltration membrane, NF membrane), and is a membrane generally defined as “a membrane that transmits monovalent ions and blocks divalent ions”. . It is a membrane that is considered to have a minute gap of about several nanometers, and is mainly used to block minute particles, molecules, ions, salts, and the like in water.
- the reverse osmosis membrane is also called an RO membrane, and is a membrane generally defined as “a membrane having a desalting function including monovalent ions”. It is a membrane that is thought to have ultrafine pores of several angstroms to several nanometers, and is mainly used for removing ionic components such as seawater desalination and ultrapure water production.
- the nanofiltration membrane used in the present invention is preferably a spiral membrane element.
- preferred nanofiltration membrane elements include, for example, GE Osmonics GEsepa, which is a cellulose acetate nanofiltration membrane element, Alfa Laval nanofiltration membrane element NF99 or NF99HF with polyamide as a functional layer, and crosslinked piperazine Nanofiltration membrane element manufactured by FILM TEC with a functional layer of polyamide NF-45, NF-90, NF-200, NF-270 or NF-400, or nanofiltration manufactured by Toray Industries, Inc., which is mainly composed of crosslinked piperazine polyamide
- the nanofiltration membrane element SU-210, SU-220, SU-600 or SU-610 manufactured by the same company including the UTC 60 of the membrane may be mentioned, more preferably NF99, NF99HF, NF-45, NF-90, NF-200, NF -400, SU-2 0, SU-220, a SU-600 or SU-610,
- the reverse osmosis membrane used in the present invention includes a composite membrane having a cellulose acetate-based polymer as a functional layer (hereinafter sometimes referred to as a cellulose acetate-based reverse osmosis membrane), and a composite having a polyamide as a functional layer. And a membrane (hereinafter also referred to as a polyamide-based reverse osmosis membrane).
- examples of the cellulose acetate-based polymer include organic acid esters of cellulose such as cellulose acetate, cellulose diacetate, cellulose triacetate, cellulose propionate, and cellulose butyrate alone or a mixture thereof and those using mixed esters. It is done.
- the polyamide includes a linear polymer or a crosslinked polymer having an aliphatic and / or aromatic diamine as a monomer.
- reverse osmosis membrane used in the present invention include, for example, ultra-low pressure type SUL-G10, SUL-G20, low pressure type SU-710, SU-, which are polyamide-based reverse osmosis membrane modules manufactured by Toray Industries, Inc.
- SU-720F SU-710L, SU-720L, SU-720LF, SU-720R, SU-710P, or SU-720P
- a sugar concentration in the sugar solution can be increased and a fermentation inhibitor can be removed as a permeate.
- the term “fermentation-inhibiting substance” as used herein refers to a component other than a sugar that inhibits fermentation in the subsequent fermentation step, and specifically includes aromatic compounds, furan compounds, organic acids, monovalent inorganic salts, and the like. be able to.
- aromatic compounds and furan compounds include furfural, hydroxymethylfurfural, vanillin, vanillic acid, syringic acid, coniferyl aldehyde, coumaric acid and ferulic acid. It can be shown.
- organic acids and inorganic salts examples include acetic acid, formic acid, and salts such as potassium and sodium.
- the sugar concentration of the concentrated sugar solution can be arbitrarily set within a range of preferably 50 g / L to 400 g / L, and may be arbitrarily set according to the use of the concentrated sugar solution.
- it may be added to a sugar solution or a concentrated sugar solution and concentrated to a target sugar concentration with a nanofiltration membrane and / or a reverse osmosis membrane.
- a nanofiltration membrane As a fermentation inhibitor, it can be removed.
- the use of a nanofiltration membrane has a higher effect of removing a fermentation inhibitor and is a preferred embodiment. Whether to use a nanofiltration membrane or a reverse osmosis membrane may be selected in view of the influence of the concentration of fermentation-inhibiting substances contained in the mixed sugar solution or the subsequent fermentation.
- Step (2) the process [Step (2)] of filtering through the microfiltration membrane of the present invention to remove the insoluble material to obtain a sugar solution as a permeate will be described.
- a microfiltration membrane also called membrane filtration, is a separation membrane that can separate and remove particles of about 0.01 to 10 ⁇ m from a fine particle suspension by using a pressure difference as a driving force.
- the surface of the microfiltration membrane has pores in the range of 0.01 to 10 ⁇ m, and fine particle components exceeding the pores can be separated and removed to the membrane side.
- microfiltration membrane examples include cellulose acetate, aromatic polyamide, polyvinyl alcohol, polysulfone, polyvinylidene fluoride, polyethylene, polyacrylonitrile, ceramic, polypropylene, polycarbonate, and polytetrafluoroethylene (“Teflon” (registered trademark)).
- Teflon polytetrafluoroethylene
- a microfiltration membrane made of polyvinylidene fluoride is preferable.
- the average pore diameter of the microfiltration membrane used in the present invention is preferably 0.01 ⁇ m to 1 ⁇ m. This is because, in the cellulose-based biomass concentrated sugar solution, the size of the insoluble material that is alkali-deposited is about 2 ⁇ m, and if the average pore size of the microfiltration membrane is 1 ⁇ m, the precipitated insoluble material is almost completely removed by the microfiltration membrane. Can be filtered off. On the other hand, if the average pore size of the microfiltration membrane is less than 0.01 ⁇ m, the average pore size is small, so it is theoretically possible to completely remove the precipitated insoluble material, but on the other hand, the filtration flow rate (Flux) is reduced, and a high pressure is required for filtration.
- Flux filtration flow rate
- a known solid-liquid separation such as a centrifugal separation method such as a screw decanter, a filtration method such as pressure / suction filtration, or a membrane filtration method such as microfiltration may be performed as a pretreatment.
- a centrifugal separation method such as a screw decanter
- a filtration method such as pressure / suction filtration
- a membrane filtration method such as microfiltration
- insoluble substances including magnesium hydroxide cannot be removed without filtration through a microfiltration membrane.
- the microfiltration membrane may be a crossflow method or a dead end filtration method, but a crossflow filtration method is preferred in terms of fouling or flux.
- the properties of the microfiltration membrane can be classified into a flat membrane and a hollow fiber membrane, but a hollow fiber membrane is preferable.
- the backwashing can be performed by applying pressure to the dirt or scale component adhering to the membrane surface with a solution containing a chemical solution from the membrane secondary side.
- Hollow fiber membranes can be classified into two types: internal pressure type hollow fiber membranes (filtered from the inside to the outside) and external pressure type hollow fiber membranes (filtered from the outside to the inside).
- an external pressure type hollow fiber membrane can be preferably used.
- backwashing is preferably performed with an acidic chemical.
- the acidic drug those having a pH of 0.4 to 4 and containing sulfuric acid or hydrochloric acid can be preferably used.
- Various chemical products can be produced by culturing microorganisms having the ability to produce chemical products using the sugar solution obtained according to the present invention as a fermentation raw material.
- culturing a microorganism as a fermentation raw material means that a sugar component or an amino source contained in a sugar solution is used as a nutrient for the microorganism to perform the growth of the microorganism and the metabolic conversion of the sugar.
- chemical products include substances that are mass-produced in the fermentation industry, such as alcohols, organic acids, amino acids, nucleic acids, and enzymes. Such chemical products are accumulated and produced as chemical products in and out of the living body in the process of metabolism using the sugar component in the sugar solution as a carbon source.
- chemicals that can be produced by microorganisms include alcohols such as ethanol, 1,3-propanediol, 1,4-butanediol or glycerol, acetic acid, lactic acid, pyruvic acid, succinic acid, malic acid, itaconic acid or citric acid.
- the sugar solution obtained in the present invention can be applied to the production of enzymes, antibiotics, recombinant proteins and the like.
- the microorganism used for the production of such a chemical product may be any microorganism that can efficiently produce the target chemical product, and microorganisms such as Escherichia coli, yeast, filamentous fungus, and basidiomycete can be used.
- the sugar solution obtained by the method for producing a sugar solution of the present invention is preferable for a method for producing a chemical product that is intermittently or continuously filtered using a separation membrane because the magnesium component is removed.
- a separation membrane used here may be either an organic polymer membrane such as PVDF or an inorganic separation membrane such as zeolite.
- the sugar solution subjected to the treatment of the present invention has an advantage that the long-term filterability is remarkably excellent since the magnesium component is removed.
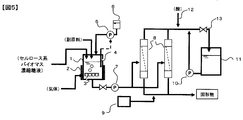
- FIG. 5 is a side view showing an example of an apparatus used in the method for producing a sugar liquid of the present invention.
- the cellulosic biomass concentrated sugar solution is held in the precipitation tank 1.
- pH is adjusted in the precipitation tank 1.
- Examples of pH adjustment include a method of adding alkali from the alkali storage tank 6 and a method of supplying alkali in a gaseous state such as ammonia gas from the air diffuser 3.
- the pH sensor 4 can monitor the pH of the precipitation tank 1 and send the signal to the alkali supply adjusting pump 5 to adjust the amount of alkali added.
- the pH of ammonia gas can be adjusted while controlling the amount of gas with a valve.
- air can be supplied from the air diffuser 3 and the cellulose-based biomass concentrated sugar solution held in the precipitation tank 1 can be mixed to make the pH uniform and promote the precipitation of magnesium hydroxide.
- the precipitation tank 1 may be provided with a temperature control facility 2. In the temperature control facility 2, either heat retention or cooling may be performed, but it is better to cool the magnesium hydroxide so that it can be further precipitated.
- the cooling temperature is not particularly limited as long as the cellulosic biomass concentrated sugar solution is not frozen.
- the precipitation tank 1 is connected to a microfiltration membrane module 8 via a microfiltration membrane pump 7. In the microfiltration membrane module 8, the above-described microfiltration membrane is disposed in the module.
- the microfiltration membrane module 8 may be provided with a compressed air supply device 9 that performs aeration for cleaning the membrane surface. Periodically, the compressed air supply device 9 can be used to remove dirt components adhering to or depositing on the surface of the microfiltration membrane.
- the filtrate component of the microfiltration membrane module 8 is collected in the MF filtrate tank 11.
- the solid matter separated on the primary side of the microfiltration membrane module 8 is discharged timely.
- the backwash pump 10 is used to apply pressure from the filtrate side, and the filtrate stored in the MF filtrate tank 11 is used to backwash the hollow fiber membrane. It can be carried out. At that time, acid is supplied from the acid supply line 12 into the pipe, the cleaning valve 13 is closed, and pressure is applied by the backwash pump 10 to supply an acid aqueous solution from the acid supply line 12.
- the hollow fiber membrane can be washed.
- a secondary raw material may be supplied to the precipitation tank 1.
- insoluble precipitates may be generated when adding the auxiliary raw material to the cellulose-based biomass concentrated sugar liquid.
- the deposition tank 1 may be supplied with a gas.
- ammonia gas is introduced in the form of gas as an alkali, it is preferable to supply it from the air diffuser 3.
- FIG. 6 is a side view showing an example of another apparatus used in the method for producing a sugar liquid of the present invention.
- This apparatus of FIG. 6 has a cross flow return line 14 with respect to the apparatus of FIG. It is an added device.
- the membrane surface of the microfiltration membrane module 8 can be filtered by crossflow by generating a liquid flow with the microfiltration membrane pump 7.
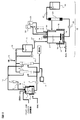
- FIG. 7 is a side view showing an example of an apparatus for producing a chemical product using the sugar solution of the present invention as a fermentation raw material.
- a fermenter 21 and a stirring device 19 are installed in the fermenter 15.
- the heat retention apparatus 18 is arrange
- the DO sensor 17 is installed in the fermenter 21, the dissolved oxygen concentration during fermentation is measured, and this signal is valve-controlled, whereby the vent pipe 16 The amount of gas vented to the fermenter 21 can be controlled more.
- the gas is selected from nitrogen, oxygen, air and the like.
- the pH sensor 20 is installed, and the acid supply from the acid supply tank 22 and the alkali charging from the alkali supply tank 23 can be controlled by this signal.
- the microfiltration membrane module 24 for isolate
- the microfiltration membrane module 24 preferably performs crossflow filtration with a crossflow pump 25.
- the filtrate of the microfiltration membrane module 24 is collected in the culture filtrate storage tank 26.
- the amount of the sugar liquid charged into the fermenter 21 and the amount of the filtrate of the microfiltration membrane are preferably controlled to be the same, and controlled by the sugar liquid flow rate control device 27.
- the obtained hydrothermally treated liquid decomposed product and cellulose fraction decomposed product were subjected to solid-liquid separation by centrifugation, and then the supernatant was filtered through a microfiltration membrane.
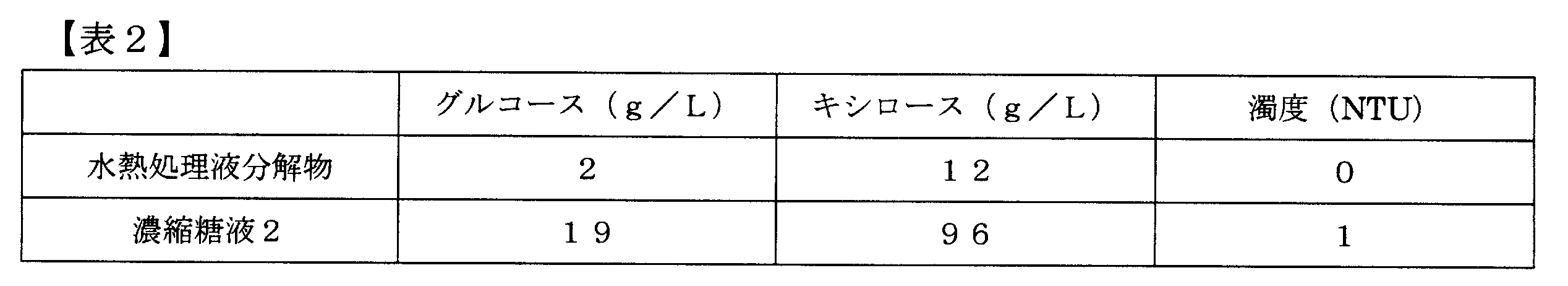
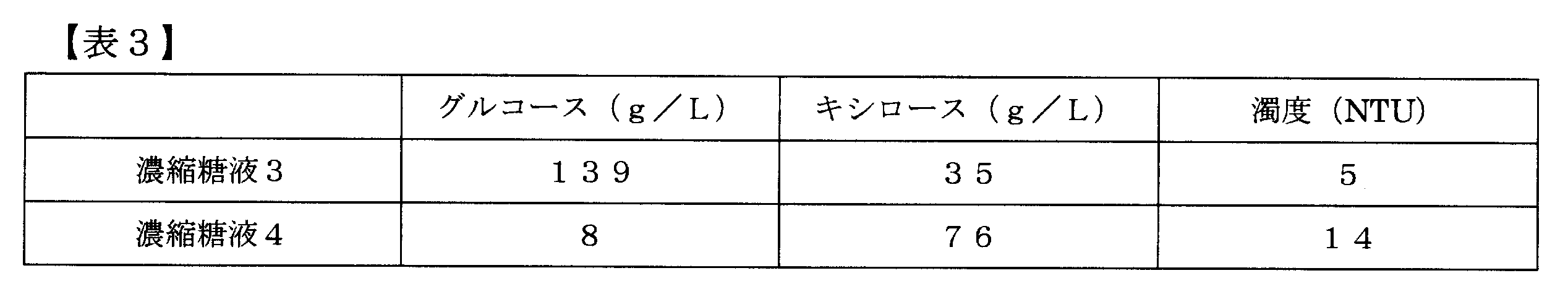
- the sugar concentration of hydrolyzed hydrolyzed product and cellulose fraction decomposed product was measured according to Reference Example 1 and summarized in Tables 1 and 2.
- the cellulose fraction decomposition product and the hydrothermal treatment liquid decomposition product were subjected to sugar concentration using a nanofiltration membrane to obtain a concentrated sugar solution 1 and a concentrated sugar solution 2.
- a nanofiltration membrane a flat membrane of “UTC-60” used for the nanofiltration membrane “SU-610” manufactured by Toray Industries, Inc. was cut out and used.
- the sugar concentrations of the hydrolyzate and the cellulose-based biomass concentrated sugar solution were measured according to Reference Example 1. The results are shown in Tables 1 and 2. Further, the turbidity (Nephelometric Turbidity Units; NTU) of each sugar solution was quantified using a HACH indoor high turbidimeter (2100N). Further, the pH of the concentrated sugar solution 1 was 4.8, and the pH of the concentrated sugar solution 2 was 3.8.
- Example 1 Adjustment to pH 7 or higher by adding alkali to cellulosic biomass concentrated sugar liquid
- Cellulose biomass concentrated liquids 1 and 2 prepared in Reference Example 1 described above were used using sodium hydroxide (1N), The pH was adjusted to 6, 7, 8, 9, 10, 11, 12, and 13. After adjusting to a predetermined pH, it was left at a temperature of 25 ° C. for 1 hour. Thereafter, the turbidity (Nephelometric Turbidity Units; NTU) was measured. The turbidity of the sugar solution was quantified using a HACH indoor high turbidimeter (2100N). The results are shown in Table 4. The turbidity of each sugar solution before pH adjustment was 0 (zero) NTU.
- Example 2 Ion chromatographic analysis of insoluble substances
- the pH of the concentrated sugar solution 1 obtained in Example 1 above was adjusted to 10, and 1 mL of the sample left for 1 hour was centrifuged (15,000 rpm, 5 minutes). The insoluble material was separated and recovered as a precipitate. 1 mL of 1N sulfuric acid aqueous solution was added to the obtained precipitate to dissolve the insoluble material again. The solution was subjected to ion chromatography analysis (cation analysis) under the following conditions.
- the chromatogram obtained by the above analysis is shown in FIG.
- a peak can be confirmed at a position corresponding to sodium ion (Na ion), potassium ion (K ion), magnesium ion (Mg ion), and calcium ion (Ca ion), and the amount of Mg ion is extremely large. It was found that it was included.
- the alkali or salt formed by Na ions and K ions is known to have high solubility even under alkaline conditions, so it must be a component dissolved in a sugar solution that coexists with insoluble substances. Was guessed.
- Mg ions are known to form magnesium hydroxide (Mg (OH) 2 ) under alkaline conditions, and their solubility product (K sp ) is 1.2 ⁇ 10 12 , especially under alkaline conditions. Insolubilized. That is, it was confirmed that the insoluble substance produced by adjusting the pH was a substance containing at least magnesium (magnesium hydroxide) as a component.
- Example 3 Particle Size Analysis of Insoluble Components Containing Magnesium Using the above Example 3, concentrated sugar solution 1, 1 mL of a sample adjusted to pH 10 and left for 1 hour, the dynamic light scattering method (Otsuka Electronics) ) To measure the particle size of the insoluble material. The number of integrations was set to 100 times. The results are shown in Table 5.
- Example 4 Microfiltration Membrane Treatment of Cellulose Biomass Concentrated Sugar Liquid Adjusted for pH Concentrated sugar liquid 1, concentrated sugar liquid 2, concentrated sugar liquid 3 and concentrated sugar liquid 4 prepared in Reference Example 2 and Reference Example 3 Each aqueous sugar solution (concentrated sugar solution 1A, concentrated sugar solution 2A, concentrated sugar solution 3A and concentrated sugar solution 4A) which was adjusted to pH 10 using 28% aqueous ammonia (Wako Pure Chemical Industries) and left for 1 hour was tested. It used as a sample (1L) and filtered using the microfiltration membrane from which an average pore diameter differs. Table 6 summarizes the types of membranes used and the average pore size.
- Concentrated sugar solutions 1A to 4A were supplied at a pressure of 30 kPa at a temperature of 25 ° C. and subjected to cross flow filtration, and an attempt was made to recover a 0.5 L sugar solution from the membrane permeation side.
- the membrane surface linear velocity at the time of cross-flow filtration was set to 30 cm / second, and filtration was performed by setting each microfiltration membrane under the condition of membrane permeation flux of 0.1 m / day.
- MF-250 was reduced in filtration flow rate immediately after filtration, and filtration could not be performed at about 100 mL.
- the average particle size of the insoluble material produced in the cellulose-based biomass concentrated sugar liquid is close to about 2 ⁇ m compared to the average pore size of 2.5 ⁇ m of MF-250, and the insoluble material particles are in the pores of the microfiltration membrane. Is estimated to have caused intrusion fouling.
- a 0.4 ⁇ m to 0.9 ⁇ m microfiltration membrane 0.5 L filtration could be completed without clogging the membrane.
- the turbidity of the filtrate was measured, the turbidity was 0 (zero) NTU no matter which membrane except MF-250 was used.
- Example 5 Ethanol Fermentation Production Using Sugar Liquid as Fermentation Raw Material Using the filtrates (sugar liquid 1 and sugar liquid 3) obtained using the microfiltration membrane (HVLP) in Example 4, yeast (Saccharomyces) cerevisiae OC-2: wine yeast).
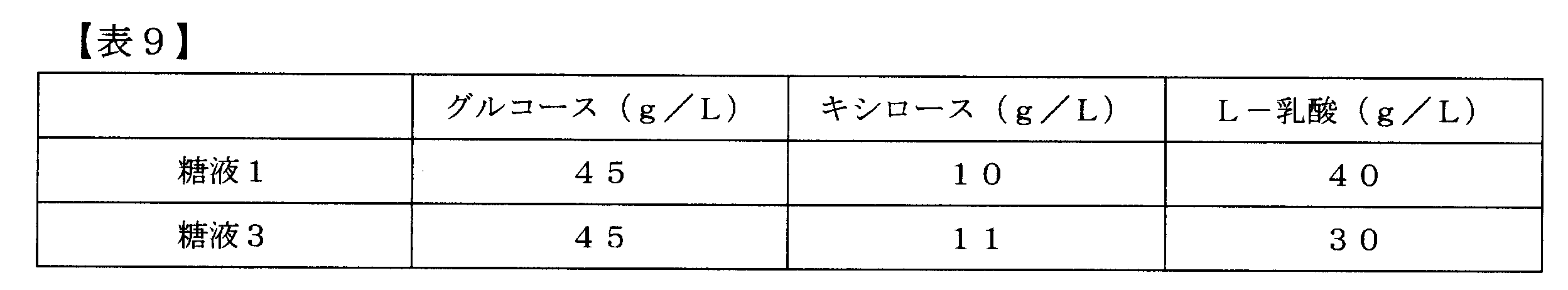
- the lactic acid bacteria were used after adjusting the concentrated sugar solution 1 and the concentrated sugar solution 3 to pH 6 using 1N sulfuric acid and then diluting them with sterilized water to the sugar concentrations shown in Table 9.
- a pre-culture solution containing lactic acid bacteria was added to these sugar solutions so as to be 5%.
- yeast was added, it was incubated at a temperature of 25 ° C. for 35 hours.
- Static culture was performed at a temperature of 37 ° C. for 24 hours.
- the concentration of L-lactic acid contained in the culture solution was analyzed under the following conditions.
- Table 9 shows the fermentation results of L-lactic acid using Sugar Solution 1 and Sugar Solution 3.
- L-lactic acid can be produced using either sugar solution 1 or sugar solution 3. Lactic acid production was lower in the sugar liquid 3 than in the sugar liquid 1.
- Example 7 Filtration with a hollow fiber microfiltration membrane and washing of the hollow fiber membrane Using the sugar solution (concentrated sugar solution 1AA) before filtration with a microfiltration membrane in Example 4, the average pore size was 0.08 ⁇ m.
- a hollow fiber ultrafiltration membrane (“Torayfil” (registered trademark) HFS, manufactured by Toray Industries, Inc.).
- Trefil HFS is an external pressure hollow fiber membrane made of PVDF, and is an external pressure hollow fiber membrane that filters the solution from the outside to the inside of the hollow fiber.
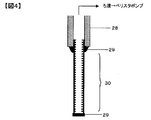
- Trefill HFS was cut to 10 cm, and one end was sealed with a silicon-based adhesive. On the other side, a silicon tube (laboran, 2 ⁇ 4) was bonded with the adhesive to form a simple membrane module (FIG. 4).
- the silicon tube 28 to which the hollow fiber microfiltration membrane 30 is connected is connected by a silicon-based adhesive 29, and the inside of the hollow fiber microfiltration membrane 30 is set to a negative pressure so that the solution outside the hollow fiber microfiltration membrane 30 is removed. It was made possible to filter.
- One end of the hollow fiber microfiltration membrane 30 was sealed with a silicon adhesive 29.
- Example 8 Filtration of culture solution using hollow fiber microfiltration membrane Culture solution 1 and comparative example after culturing sugar solution 1 of Example 6 using the simple hollow fiber module prepared in Example 7 above After culturing the concentrated sugar solution 1A of No. 2, the culture solution 1A was filtered, and the separability between the lactic acid aqueous solution as a product and the microbial cell (lactic acid bacterium) was evaluated.
- the simple hollow fiber module of Example 7 was placed in a beaker containing 100 mL of each culture solution, a magnetic stirrer was placed in the beaker, and filtration was performed while stirring at 100 rpm using a stirrer. Filtration flux was initially set to 0.5 m / day and filtration was started. In the culture solution 1A, filtration became difficult in 20 minutes from the start, and in the culture solution 1, filtration was performed for 2 hours or more, and an 8 mL filtrate (lactic acid aqueous solution) was obtained. That is, when the chemical product (lactic acid) is produced using the concentrated sugar solution (concentrated sugar solution 1) obtained in the present invention, the fermentation product (lactic acid aqueous solution) is separated from the culture solution after fermentation (membrane separation). ).
- Example 9 L-lactic acid continuous fermentation
- the filtrate obtained by using a microfiltration membrane (HVLP) in Example 4 using the continuous culture apparatus described in JP-A-2008-237213 (FIG. 2) Continuous fermentation was carried out using the lactic acid bacteria described in Example 7 using the sugar solution 1) and the concentrated sugar solution (concentrated sugar solution 1A) before being filtered through the microfiltration membrane in Example 5.
- the membrane was blocked after 200 hours of culture, making it difficult to culture.
- the sugar solution 1 subjected to the microfiltration membrane treatment continuous culture for 500 hours or more was possible. That is, it was confirmed that the sugar solution produced in the present invention can be preferably used as a sugar solution used for continuous culture.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Biochemistry (AREA)
- Zoology (AREA)
- Engineering & Computer Science (AREA)
- Wood Science & Technology (AREA)
- General Health & Medical Sciences (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Microbiology (AREA)
- General Chemical & Material Sciences (AREA)
- Bioinformatics & Cheminformatics (AREA)
- General Engineering & Computer Science (AREA)
- Biotechnology (AREA)
- Genetics & Genomics (AREA)
- Emergency Medicine (AREA)
- Separation Using Semi-Permeable Membranes (AREA)
- Preparation Of Compounds By Using Micro-Organisms (AREA)
Description
本発明により得られた糖液を、発酵原料として化学品を生産する能力を有する微生物を培養することにより、各種化学品を製造することができる。ここでいう発酵原料として微生物を培養するとは、糖液に含まれる糖成分あるいはアミノ源を微生物の栄養素として利用し、微生物の増殖と糖の代謝変換を行うことを意味している。
次に、本発明の糖液を製造する装置に関して説明する。
次に、本発明の糖液を発酵原料として、化学品を製造する装置に関して説明する。
糖液に含まれるグルコースおよびキシロース濃度は、下記に示すHPLC条件で、標品との比較により定量した。
・カラム:Luna NH2(Phenomenex社製)
・移動相:ミリQ:アセトニトリル=25:75(流速0.6mL/分)
・反応液:なし
・検出方法:RI(示差屈折率)
・温度:30℃。
セルロースとして、稲藁を使用した。前記のセルロースを水に浸し、撹拌しながら180℃の温度で20分間オートクレーブ処理(日東高圧株式会社製)した。処理後、遠心分離(3000G)を行い、溶液成分(水熱処理液)と固形物(セルロース画分)に分離を行った。これら水熱処理液およびセルロース画分に対し、ジェネンコア製「アクセルレースデュエット」(酵素濃度:40g/L)を添加(最終濃度1mg/L)して、50℃の温度で、24時間保温し、加水分解を実施した。得られた水熱処理液分解物およびセルロース画分分解物は、遠心分離によって固液分離した後、上澄みを精密濾過膜にて濾過した。水熱処理液分解物およびセルロース画分分解物の糖濃度を参考例1に準じて測定を行い、表1および表2にまとめた。
参考例2記載の手順で調製した、セルロース画分分解物および水熱処理液分解物を減圧濃縮により濃縮し、濃縮糖液3および濃縮糖液4を得た。減圧濃縮はロータリーエバポレーター(アズワン製)を使用して行い、80℃、200hPaまで減圧して、糖濃縮を行った。得られた濃縮糖液の糖濃度および濁度を、参考例2に準じて測定を行った。結果を表3に示す。
前記の参考例1で調製したセルロース系バイオマス濃縮液1および2を、水酸化ナトリウム(1N)を使用して、pHを6、7、8、9,10、11、12および13に調製した。所定pHに調製した後、1時間25℃の温度で放置した。その後、濁度(Nephelometric Turbidity Units;NTU)を測定した。糖液の濁度は、HACH社製室内用高度濁度計(2100N)を用いて定量した。その結果を表4に示す。pH調整前の各糖液の濁度は、0(ゼロ)NTUであった。
前記の実施例1で得られた濃縮糖液1のpHを10に調整し、1時間放置したサンプル1mLを遠心分離(15,000rpm、5分)し、不溶性物質を沈殿として分離回収した。得られた沈殿に対し、1N硫酸水溶液1mLを添加し、不溶性物質を再度溶解させた。溶解液を、次の条件でイオンクロマト分析(カチオン分析)を実施した。
分析条件:
・カラム:Ion Pac AS22(DIONEX社製)
・移動相:4.5mM Na2CO3/1.4mM NaHCO3(流速1.0mL/分)
・反応液:なし
・検出方法:電気伝導度(サプレッサ使用)
・温度:30℃。
前記の実施例3、濃縮糖液1、pHを10に調整し1時間放置したサンプル1mLを使用して、動的光散乱法(大塚電子)による不溶性物質の粒径測定を行った。積算回数は100回に設定した。その結果を表5に示す。
参考例2および参考例3で調製した濃縮糖液1、濃縮糖液2、濃縮糖液3および濃縮糖液4を、28%アンモニア水(和光純薬工業)を用いてpH10に調整し、1時間放置した各糖水溶液(濃縮糖液1A、濃縮糖液2A、濃縮糖液3Aおよび濃縮糖液4A)を、試験サンプル(1L)として使用し、平均細孔径の異なる精密濾過膜を用いて濾過を行った。使用した膜種および平均細孔径を、表6にまとめて示す。
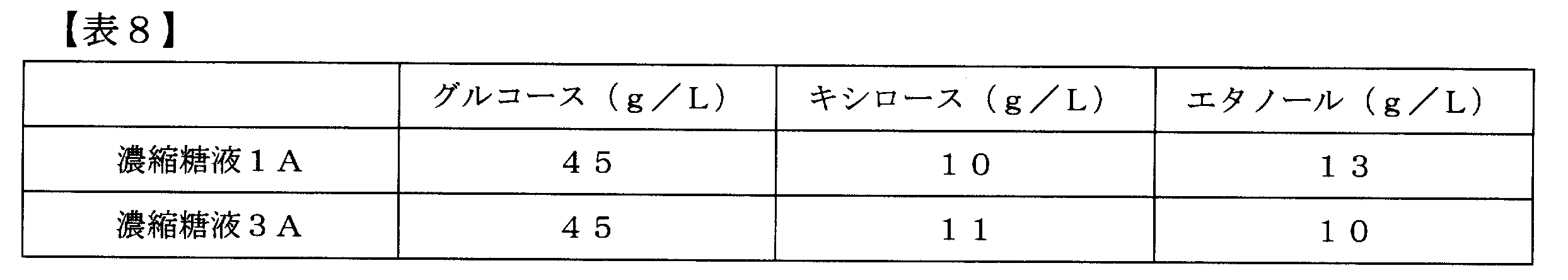
実施例4で精密濾過膜(HVLP)を使用して得られた濾液(糖液1および糖液3)を使用して、酵母(Saccharomycecs cerevisiae OC-2:ワイン酵母)によるエタノール発酵試験を行った。
比較のために、実施例4で精密濾過膜による濾過を行う前の糖液(実施例4の濃縮糖液1Aおよび濃縮糖液3A:pH調整のみ実施)を使用して、実施例5に準じてエタノール発酵試験を行った。結果を、表8に示す。実施例5の精密濾過膜によって処理した本発明の糖液に比べ、培養液中のエタノール蓄積濃度が低いことが判明した。
実施例4で精密濾過膜(HVLP)を使用して得られた濾液(濃縮糖液1および濃縮糖液3)およびラクトコッカス・ラクティスJCM7638株を使用して、乳酸発酵生産を検討した。
・カラム:Shim-Pack SPR-H(株式会社島津製作所製)
・移動相:5mM p-トルエンスルホン酸(流速0.8mL/min)
・反応液:5mM p-トルエンスルホン酸、20mM ビストリス、0.1mM EDTA2Na(流速0.8mL/min)
・検出方法:電気伝導度
・温度:45℃。
比較のために、実施例4で精密濾過膜による濾過を行う前の糖液(濃縮糖液1Aおよび濃縮糖液3A:pH調整のみ実施)を使用してラクトコッカス・ラクティスJCM7638株を24時間、37℃の温度で静置培養した。手順は、濃縮糖液が精密濾過を行っていないものを使用したこと以外は、実施例6と同じ手順で行った。濃縮糖液1Aおよび濃縮糖液3Aを使用して発酵した結果を表10に示す。実施例6に比べ、L-乳酸濃度が低いことが判明した。
実施例4で精密濾過膜による濾過を行う前の糖液(濃縮糖液1AA)を使用して、平均細孔径0.08μmの中空糸限外濾過膜(東レ製、“トレフィル”(登録商標)HFS)に通じて濾過を行った。トレフィルHFSは、PVDF製外圧式中空糸膜であり、中空糸外側から内側に向かって溶液の濾過を行う外圧式中空糸膜である。トレフィルHFSは、10cmにカットし片末端は、シリコン系接着剤を用いて封止した。もう一方には、シリコンチューブ(ラボラン、2x4)を前記接着剤で接着し、簡易膜モジュールとした(図4)。図4において、中空糸精密濾過膜30を接続したシリコンチューブ28は、シリコン系接着剤29で接続し、中空糸精密濾過膜30内部を陰圧にすることにより中空糸精密濾過膜外側の溶液を濾過できるようにした。なお中空糸精密濾過膜30の片端は、シリコン系接着剤29で封止した。
前記の実施例7で作製した簡易中空糸モジュールを使用して、実施例6の糖液1を培養した後の培養液1および比較例2のの濃縮糖液1Aを培養した後の培養液1Aの濾過を行い、生産物である乳酸水溶液と微生物菌体(乳酸菌)の分離性を評価した。
特開2008-237213号公報(図2)記載の連続培養装置を使用して、実施例4で精密濾過膜(HVLP)を使用して得られた濾液(糖液1)および、実施例5で精密濾過膜による濾過を行う前の濃縮糖液(濃縮糖液1A)を使用して、前記実施例7記載の乳酸菌を使用して連続発酵を実施した。その結果、濃縮糖液1Aでは、培養200時間で膜の閉塞が確認され培養困難となった。一方、精密濾過膜処理を行った糖液1では、500時間以上の連続培養が可能であった。すなわち、本発明で製造された糖液は、連続培養に使用する糖液として好ましく使用できることが確認できた。
2.温度調節装置
3.散気管
4.pHセンサー
5.アルカリ供給ポンプ
6.アルカリ貯槽
7.精密濾過膜ポンプ
8.精密濾過膜モジュール
9.圧空供給装置
10.逆洗ポンプ
11.MF濾液槽
12.酸供給ライン
13.洗浄バルブ
14.クロスフロー戻りライン
15.発酵装置
16.通気管
17.DOセンサー
18.保温装置
19.攪拌装置
20.pHセンサー(発酵)
21.発酵槽
22.酸供給槽
23.アルカリ供給槽
24.精密濾過膜モジュール
25.クロスフローポンプ
26.培養濾液貯槽
27.糖液流量制御装置
28.シリコンチューブ
29.シリコン系接着剤
30.中空糸精密濾過膜
Claims (8)
- セルロース系バイオマス濃縮糖液にアルカリを添加し、pHを7以上に調整し、少なくともマグネシウムを含む不溶性物質を析出させる工程、および、精密濾過膜に通じて濾過し、該不溶性物質を除去し、透過液として糖液を得る工程を含む糖液の製造方法。
- セルロース系バイオマス濃縮糖液が、セルロース系バイオマスの水熱処理、酸処理、アルカリ処理および酵素処理のいずれか1以上の処理により得られた加水分解物を、膜濃縮、減圧濃縮および熱濃縮のいずれか1以上の処理で濃縮した糖液である請求項1記載の糖液の製造方法。
- pHを8以上に調整する請求項1または2記載の糖液の製造方法。
- 精密濾過膜の平均細孔径が0.01μm~1μmの範囲である請求項1~3のいずれかに記載の糖液の製造方法。
- 精密濾過膜が中空糸精密濾過膜である請求項1~4のいずれかに記載の糖液の製造方法。
- 窒素源、金属塩、ビタミン、アミノ酸、糖類、消泡剤および界面活性剤からなる群から選ばれた添加剤をさらに添加する請求項1から7のいずれかに記載の糖液の製造方法。
- 請求項1から6いずれかに記載の糖液の製造方法で得られた糖液を発酵原料として使用して、微生物を培養する化学品の製造方法。
- 請求項1から7いずれかに記載の糖液の製造方法で得られた糖液を発酵原料として微生物を培養し、培養液中に化学品を産生させるとともに、微生物と化学品を連続的あるいは間欠的に分離膜に通じて濾過し、化学品を回収する化学品の製造方法。
Priority Applications (7)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US14/405,239 US9765412B2 (en) | 2012-06-05 | 2013-06-04 | Process of producing sugar solution |
| CA2875083A CA2875083C (en) | 2012-06-05 | 2013-06-04 | Process for producing sugar solution |
| ES13801065.7T ES2682273T3 (es) | 2012-06-05 | 2013-06-04 | Procedimiento para la producción de una solución que contiene azúcar |
| JP2013535194A JP6330328B2 (ja) | 2012-06-05 | 2013-06-04 | 糖液の製造方法 |
| EP13801065.7A EP2857528B1 (en) | 2012-06-05 | 2013-06-04 | Process for producing a sugar solution |
| BR112014029529-8A BR112014029529B1 (pt) | 2012-06-05 | 2013-06-04 | método para a produção de um líquido de açúcar |
| AU2013272652A AU2013272652B2 (en) | 2012-06-05 | 2013-06-04 | Process for producing sugar solution |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2012-127704 | 2012-06-05 | ||
| JP2012127704 | 2012-06-05 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2013183617A1 true WO2013183617A1 (ja) | 2013-12-12 |
Family
ID=49712008
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2013/065431 Ceased WO2013183617A1 (ja) | 2012-06-05 | 2013-06-04 | 糖液の製造方法 |
Country Status (8)
| Country | Link |
|---|---|
| US (1) | US9765412B2 (ja) |
| EP (1) | EP2857528B1 (ja) |
| JP (1) | JP6330328B2 (ja) |
| AU (1) | AU2013272652B2 (ja) |
| BR (1) | BR112014029529B1 (ja) |
| CA (1) | CA2875083C (ja) |
| ES (1) | ES2682273T3 (ja) |
| WO (1) | WO2013183617A1 (ja) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2015119038A1 (ja) * | 2014-02-05 | 2015-08-13 | 東レ株式会社 | 糖液の製造方法 |
| JP2017109187A (ja) * | 2015-12-18 | 2017-06-22 | 昭和電工株式会社 | 植物性バイオマスの加水分解方法及び装置 |
| WO2018147289A1 (ja) * | 2017-02-07 | 2018-08-16 | 東レ株式会社 | 連続発酵によるアルコールの製造方法およびそれに用いる連続発酵装置 |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN103936109B (zh) * | 2014-04-12 | 2016-08-17 | 大连双迪创新科技研究院有限公司 | 饮水电解制取装置 |
| KR101763367B1 (ko) * | 2015-04-09 | 2017-07-31 | 한국화학연구원 | 오염 미생물의 대사산물 생성을 최소화하는 바이오매스의 효소당화 방법 및 그 장치 |
| GB201807987D0 (en) * | 2018-05-17 | 2018-07-04 | Univ Leeds Innovations Ltd | Reduction in microbial growth |
Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH0767399B2 (ja) * | 1987-04-08 | 1995-07-26 | 三菱化成エンジニアリング株式会社 | 甘蔗糖蜜中の蔗糖分の回収方法 |
| JPH07242788A (ja) * | 1994-02-14 | 1995-09-19 | Wr Grace & Co Connecticut | 濃縮用の蒸発器内での堆積の防止方法 |
| JP2001157599A (ja) * | 1999-12-02 | 2001-06-12 | Tsukishima Kikai Co Ltd | 炭酸ソーダの添加による軟化処理を含む限外ろ過処理による甘蔗からの精製糖製造プロセス |
| JP2005027807A (ja) | 2003-07-10 | 2005-02-03 | Matsushita Electric Ind Co Ltd | 電気掃除機 |
| JP2006304634A (ja) * | 2005-04-27 | 2006-11-09 | Hokkaido Sugar Co Ltd | ビート含蜜糖乃至液糖組成物 |
| JP2008237213A (ja) | 2007-02-26 | 2008-10-09 | Toray Ind Inc | 連続発酵装置 |
| WO2009041009A1 (ja) | 2007-09-25 | 2009-04-02 | Kubota Corporation | 有機性廃棄物の処理方法および装置 |
| WO2011162009A1 (ja) * | 2010-06-24 | 2011-12-29 | 東レ株式会社 | 精製糖水溶液の製造方法 |
Family Cites Families (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2073616A (en) * | 1927-07-15 | 1937-03-16 | Arthur B Foster | Galactan product and process of making same |
| US2892683A (en) * | 1953-07-08 | 1959-06-30 | Montedison Spa | Process of precipitating and filtering magnesium hydroxide |
| US3972775A (en) * | 1974-06-28 | 1976-08-03 | The United States Of America As Represented By The United States Energy Research And Development Administration | Conversion of cellulosic materials to sugar |
| US4743682A (en) * | 1984-11-27 | 1988-05-10 | Nestec S.A. | Methods for controlling the viscosity of protein hydrolysates |
| JPS6467173A (en) * | 1987-09-04 | 1989-03-13 | Sanei Kagaku Kogyo Kk | Preparation of vegetable or fruit juices |
| US5782982A (en) * | 1993-03-26 | 1998-07-21 | Arkenol, Inc. | Method of removing silica or silicates from solids resulting from the strong acid hydrolysis of cellulosic and hemicellulosic materials |
| JP4182524B2 (ja) | 2004-03-26 | 2008-11-19 | 北海道糖業株式会社 | ビート含蜜糖の製造方法 |
| BRPI0816477A2 (pt) * | 2007-08-27 | 2014-10-14 | Iogen Energy Corp | Método para a produção de um produto de fermentação a partir de uma matéria-prima lignocelulósica pré-tratada |
| CN101348429B (zh) | 2008-09-05 | 2011-03-16 | 江南大学 | 一种阳离子树脂交换提取发酵液中丁二酸的方法 |
| WO2010081217A1 (en) * | 2009-01-14 | 2010-07-22 | Iogen Energy Corporation | Improved method for the production of glucose from lignocellulosic feedstocks |
-
2013
- 2013-06-04 JP JP2013535194A patent/JP6330328B2/ja active Active
- 2013-06-04 CA CA2875083A patent/CA2875083C/en not_active Expired - Fee Related
- 2013-06-04 WO PCT/JP2013/065431 patent/WO2013183617A1/ja not_active Ceased
- 2013-06-04 ES ES13801065.7T patent/ES2682273T3/es active Active
- 2013-06-04 AU AU2013272652A patent/AU2013272652B2/en active Active
- 2013-06-04 US US14/405,239 patent/US9765412B2/en active Active
- 2013-06-04 EP EP13801065.7A patent/EP2857528B1/en not_active Not-in-force
- 2013-06-04 BR BR112014029529-8A patent/BR112014029529B1/pt active IP Right Grant
Patent Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH0767399B2 (ja) * | 1987-04-08 | 1995-07-26 | 三菱化成エンジニアリング株式会社 | 甘蔗糖蜜中の蔗糖分の回収方法 |
| JPH07242788A (ja) * | 1994-02-14 | 1995-09-19 | Wr Grace & Co Connecticut | 濃縮用の蒸発器内での堆積の防止方法 |
| JP2001157599A (ja) * | 1999-12-02 | 2001-06-12 | Tsukishima Kikai Co Ltd | 炭酸ソーダの添加による軟化処理を含む限外ろ過処理による甘蔗からの精製糖製造プロセス |
| JP2005027807A (ja) | 2003-07-10 | 2005-02-03 | Matsushita Electric Ind Co Ltd | 電気掃除機 |
| JP2006304634A (ja) * | 2005-04-27 | 2006-11-09 | Hokkaido Sugar Co Ltd | ビート含蜜糖乃至液糖組成物 |
| JP2008237213A (ja) | 2007-02-26 | 2008-10-09 | Toray Ind Inc | 連続発酵装置 |
| WO2009041009A1 (ja) | 2007-09-25 | 2009-04-02 | Kubota Corporation | 有機性廃棄物の処理方法および装置 |
| WO2011162009A1 (ja) * | 2010-06-24 | 2011-12-29 | 東レ株式会社 | 精製糖水溶液の製造方法 |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP2857528A4 |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2015119038A1 (ja) * | 2014-02-05 | 2015-08-13 | 東レ株式会社 | 糖液の製造方法 |
| JPWO2015119038A1 (ja) * | 2014-02-05 | 2017-03-23 | 東レ株式会社 | 糖液の製造方法 |
| US10767236B2 (en) | 2014-02-05 | 2020-09-08 | Toray Industries, Inc. | Method of producing sugar solution |
| JP2017109187A (ja) * | 2015-12-18 | 2017-06-22 | 昭和電工株式会社 | 植物性バイオマスの加水分解方法及び装置 |
| WO2018147289A1 (ja) * | 2017-02-07 | 2018-08-16 | 東レ株式会社 | 連続発酵によるアルコールの製造方法およびそれに用いる連続発酵装置 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP2857528A4 (en) | 2015-12-09 |
| AU2013272652A1 (en) | 2014-12-04 |
| EP2857528A1 (en) | 2015-04-08 |
| ES2682273T3 (es) | 2018-09-19 |
| EP2857528B1 (en) | 2018-07-18 |
| CA2875083C (en) | 2020-03-31 |
| AU2013272652B2 (en) | 2017-09-07 |
| US20150147787A1 (en) | 2015-05-28 |
| US9765412B2 (en) | 2017-09-19 |
| CA2875083A1 (en) | 2013-12-12 |
| BR112014029529A2 (pt) | 2017-06-27 |
| JPWO2013183617A1 (ja) | 2016-02-01 |
| JP6330328B2 (ja) | 2018-05-30 |
| BR112014029529B1 (pt) | 2021-03-09 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| RU2582649C2 (ru) | Способ получения сахарного раствора | |
| CA2831543C (en) | Method for producing sugar solution | |
| EP2860269B1 (en) | Method for producing a sugar solution | |
| JP6330328B2 (ja) | 糖液の製造方法 | |
| EP2546353A1 (en) | Method for producing pure sugar solution, and method for producing chemical product | |
| JPWO2011162009A1 (ja) | 精製糖水溶液の製造方法 | |
| JP5728817B2 (ja) | キシロース糖液の製造方法 | |
| JP6167902B2 (ja) | 糖液の製造方法 | |
| JP2012191878A (ja) | 糖液の製造方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| ENP | Entry into the national phase |
Ref document number: 2013535194 Country of ref document: JP Kind code of ref document: A |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 13801065 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2875083 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14405239 Country of ref document: US |
|
| ENP | Entry into the national phase |
Ref document number: 2013272652 Country of ref document: AU Date of ref document: 20130604 Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: IDP00201408332 Country of ref document: ID |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112014029529 Country of ref document: BR |
|
| ENP | Entry into the national phase |
Ref document number: 112014029529 Country of ref document: BR Kind code of ref document: A2 Effective date: 20141126 |