WO2014181852A1 - 浴用剤組成物 - Google Patents
浴用剤組成物 Download PDFInfo
- Publication number
- WO2014181852A1 WO2014181852A1 PCT/JP2014/062430 JP2014062430W WO2014181852A1 WO 2014181852 A1 WO2014181852 A1 WO 2014181852A1 JP 2014062430 W JP2014062430 W JP 2014062430W WO 2014181852 A1 WO2014181852 A1 WO 2014181852A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- mass
- component
- ginsenoside
- agent composition
- bath
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/19—Cosmetics or similar toiletry preparations characterised by the composition containing inorganic ingredients
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/36—Carboxylic acids; Salts or anhydrides thereof
- A61K8/362—Polycarboxylic acids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/63—Steroids; Derivatives thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/96—Cosmetics or similar toiletry preparations characterised by the composition containing materials, or derivatives thereof of undetermined constitution
- A61K8/97—Cosmetics or similar toiletry preparations characterised by the composition containing materials, or derivatives thereof of undetermined constitution from algae, fungi, lichens or plants; from derivatives thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/96—Cosmetics or similar toiletry preparations characterised by the composition containing materials, or derivatives thereof of undetermined constitution
- A61K8/97—Cosmetics or similar toiletry preparations characterised by the composition containing materials, or derivatives thereof of undetermined constitution from algae, fungi, lichens or plants; from derivatives thereof
- A61K8/9783—Angiosperms [Magnoliophyta]
- A61K8/9789—Magnoliopsida [dicotyledons]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q19/00—Preparations for care of the skin
- A61Q19/10—Washing or bathing preparations
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2800/00—Properties of cosmetic compositions or active ingredients thereof or formulation aids used therein and process related aspects
- A61K2800/20—Chemical, physico-chemical or functional or structural properties of the composition as a whole
- A61K2800/22—Gas releasing
- A61K2800/222—Effervescent
Definitions
- the present invention relates to a bath agent composition.
- a bath preparation containing a carbonate and an organic acid is widely known as an excellent bath preparation because it generates bubbles of carbon dioxide gas in the bath water and the blood circulation promoting effect is obtained by the carbon dioxide gas.
- studies have been made to bring additional effects and synergistic effects by further blending various components with such bath agents.
- herbal medicine such as ginseng is known as a component that can be blended in a so-called foaming bath agent blended with an organic acid such as carbonate and succinic acid.
- An effervescent bath agent that can synergistically enhance the moist feeling effect of hot water has been developed (see Patent Document 1).
- Ginseng is also known as Panax ginseng, a medicinal plant that has been known for a long time. Ginsenoside is known as a medicinal component, and 50 or more types of ginsenoside have been isolated from ginseng so far.
- antitumor action anti-arteriosclerosis / antihypertensive action, anti-stress action, immunoregulatory action, anti-inflammatory / anti-allergic action, anti-diabetes mellitus
- Various effects such as action, action on the central nervous system, improvement of memory and learning, neuroprotection, neurotransmitter release or uptake action have been found (Non-Patent Documents 1 and 2).
- Patent Document 1 Japanese Patent Application Laid-Open No. 60-215618 (Non-Patent Document 1) Advances in Food and Nutrition Research, 2009, 55: 1-99 (Non-Patent Document 2) Phytochemical Analysis, 2008, 19: 2-16
- the present invention includes the following components (A), (B) and (C): (A) Ginsenoside Rg3 and / or ginsenoside Rh2 in total 0.00001% by mass to 0.5% by mass, (B) Carbonate whose content of dialkali metal carbonate in carbonate is 30% by mass or more, and (C) Mass of organic acid, ginsenoside Rb2, Rb1, Rc and Rd, and ginsenoside Rg3 and Rh2 A bath agent composition having a ratio ((Rb2 + Rb1 + Rc + Rd) / (Rg3 + Rh2)) of 0 or more and 30 or less is provided.
- the present invention provides a bath preparation capable of enhancing the skin feel at the time of bathing while suppressing the coloring and discoloration as well as the stability in appearance while sufficiently exerting the blood circulation promoting effect by the combination of carbonate and organic acid. Relates to the composition.
- the present inventors have made various studies, and among the ginsenosides known to be mainly contained in ginseng, they contain two specific types in a specific quantitative ratio and use a specific carbonate.
- a bath preparation composition that can improve the stability on appearance while synergistically enhancing the blood circulation promoting effect by the generation of carbon dioxide gas, and can also improve the skin feel during bathing.
- the present invention was completed.
- the blood circulation promoting effect is synergistically increased, and the appearance stability is also improved, so that the bath agent composition can be colored and discolored over time. It can be effectively suppressed.
- the skin feel at the time of bathing can be made smooth with a moist and oily feeling.
- the bath agent composition of the present invention contains 0.00001% by mass to 0.5% by mass of ginsenoside Rg3 and / or ginsenoside Rh2 (A).
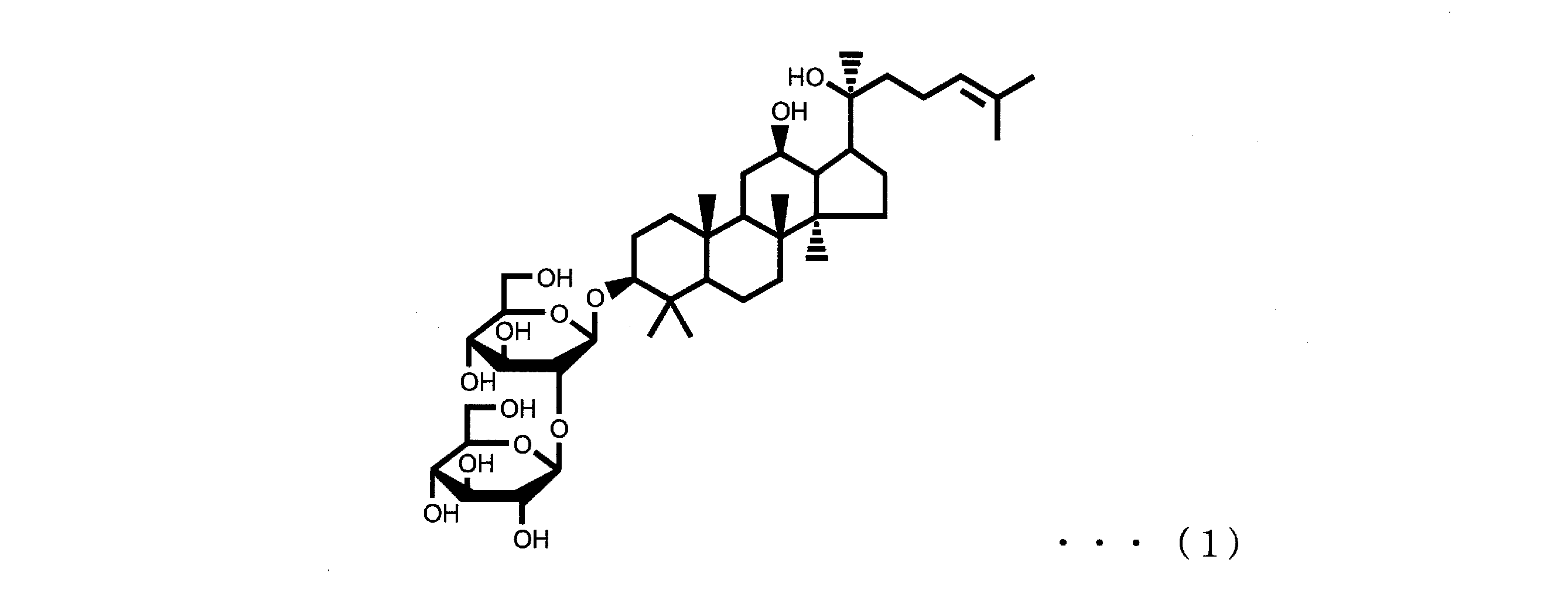
- Ginsenoside Rg3 and ginsenoside Rh2 are also known as active ingredients of ginseng, ginsenoside Rg3 is represented by the following formula (1), and ginsenoside Rh2 is represented by the following formula (2).
- Both the ginsenoside Rg3 and ginsenoside Rh2 of the component (A) can act effectively and exhibit a blood circulation promoting effect, and are combined with the carbonate of the component (B) and the organic acid of the component (C) described later.
- By generating carbon dioxide gas it is possible to enhance the blood circulation promoting effect together.
- the stability in appearance be improved and coloring of the bath agent composition can be prevented, but also the color change with time can be effectively suppressed.
- the skin feel when bathing is smooth, moist and oily that is, when touching the skin in bath water, you can feel a moderate slime feeling as if it was slightly covered with an oil film. It is possible to suppress a crisp feeling of friction.
- ginsenoside Rg3 and ginsenoside Rh2 may be used alone, or both may be used together.
- ginsenoside Rg3 is preferable from the viewpoint of synergistically enhancing the blood circulation promoting effect and enhancing the effect of suppressing coloring and discoloration.
- the content of component (A) is in the bath preparation composition of the present invention from the viewpoint of enhancing the blood circulation promoting effect and the effect of improving the skin feel during bathing, and the effect of suppressing coloring and discoloration.
- the total amount is 0.00001% by mass or more, preferably 0.00002% by mass or more, and more preferably 0.00003% by mass or more.
- Content of a component (A) is 0.5 mass% in total in the bath agent composition of this invention from a viewpoint of ensuring generation
- the content of the component (A) is 0.00001 to 0.5% by mass in total in the bath agent composition of the present invention, preferably 0.00002 to 0.3% by mass, More preferably, it is 0.00003 to 0.1% by mass.
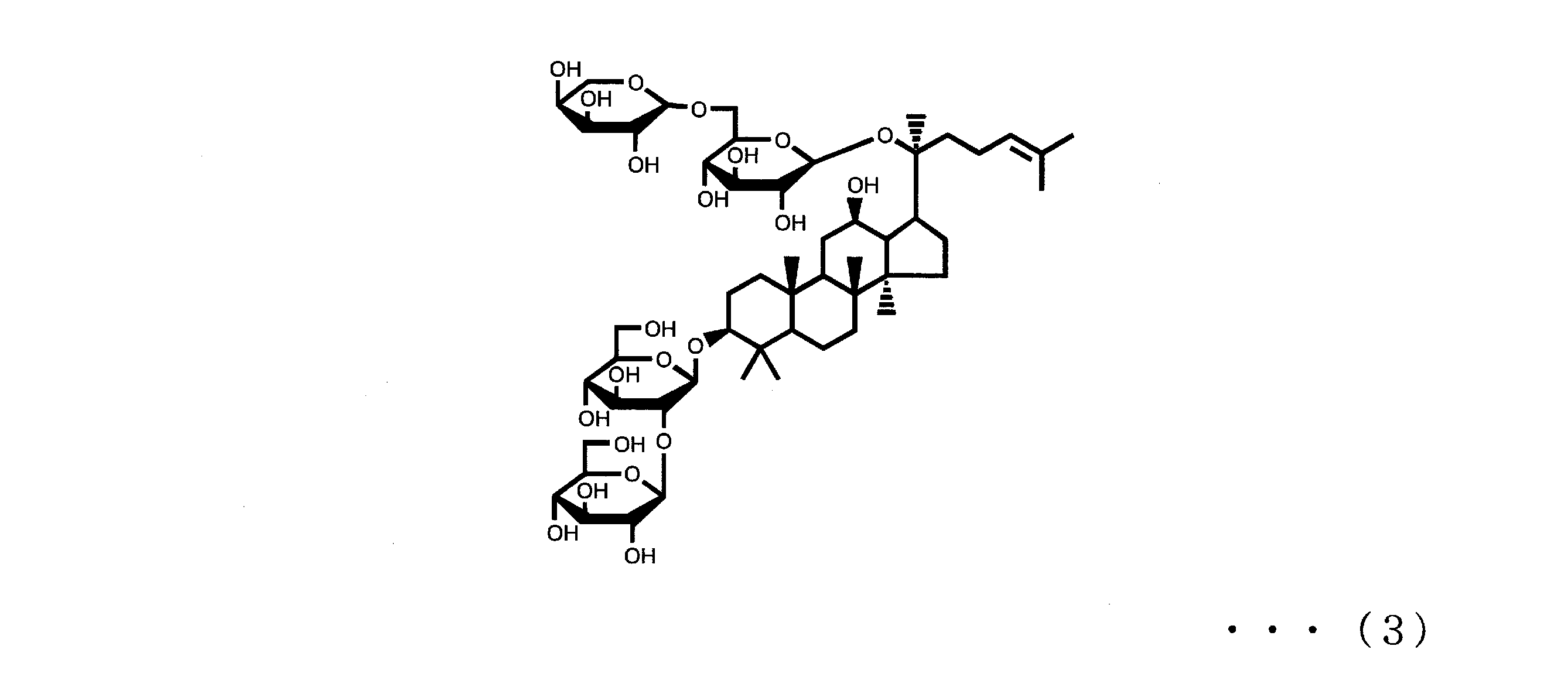
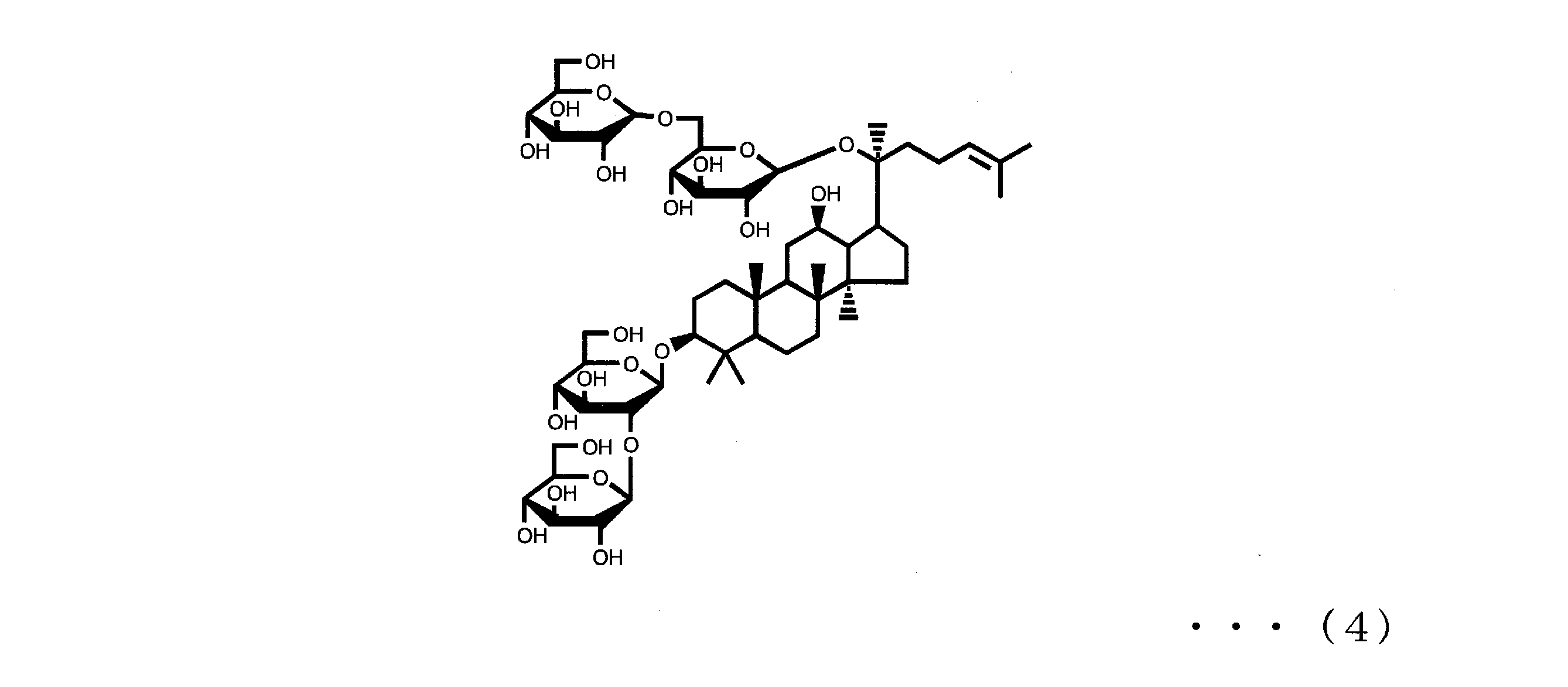
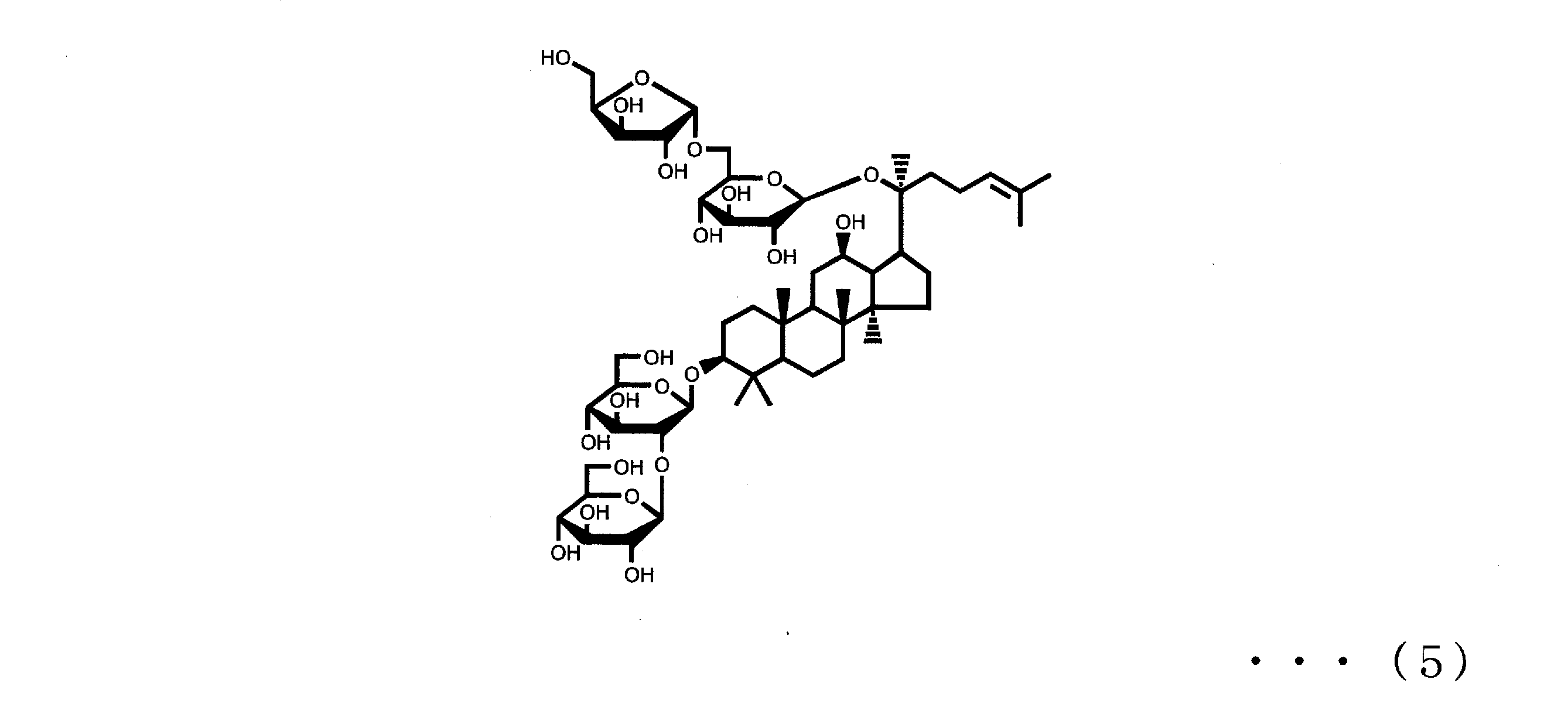
- Ginsenoside Rb2, Rb1, Rc and Rd are also known as components that can be contained in ginseng together with ginsenoside Rg3 and Rh2 of component (A).
- the mass ratio of ginsenoside Rb2, Rb1, Rc and Rd to ginsenoside Rg3 and Rh2 ((Rb2 + Rb1 + Rc + Rd) / (Rg3 + Rh2)) of component (A) enhances the effect of suppressing coloring and discoloration. From the viewpoint, it is 0 or more, and from the viewpoint of satisfactorily maintaining the effect of suppressing coloring and discoloration, it is 30 or less, preferably 20 or less, more preferably 10 or less, and even more preferably 7 or less.
- the mass ratio of ginsenoside Rb2, Rb1, Rc and Rd to ginsenoside Rg3 and Rh2 is 0-30, preferably 0-20, more preferably 0-10. More preferably 0 to 7, still more preferably 0 to 3, and still more preferably 0 to 1.
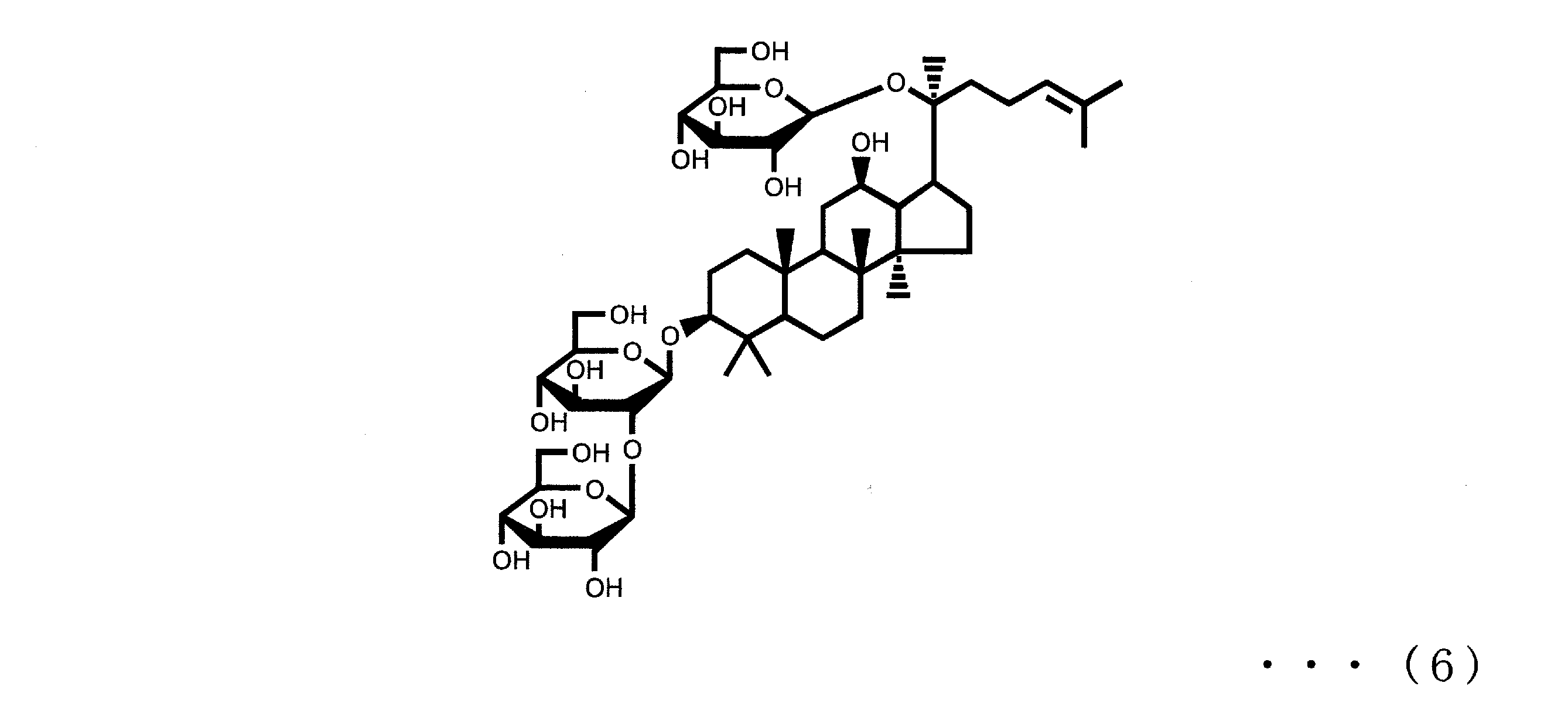
- Ginsenoside Rb2, Rb1, Rc and Rd are respectively represented by the following formulas (3), (4), (5) and (6).
- ginsenoside when ginsenoside is extracted from ginseng, usually not only ginsenoside Rg3 and Rh2, but also a plurality of types of ginsenoside are extracted.
- ginsenosides Rg3 and Rh2 paying attention to ginsenosides Rg3 and Rh2, and setting them to a specific content while (Rb2 + Rb1 + Rc + Rd) / (Rg3 + Rh2) is in the above range, blood circulation promoting effects, coloring and discoloration by ginsenoside Rg3 and Rh2 are achieved. It is possible to fully enjoy the suppression effect and the effect of improving the skin feel during bathing.
- component (A) when component (A) is obtained by extracting ginsenoside from ginseng, in order to set (Rb2 + Rb1 + Rc + Rd) / (Rg3 + Rh2) to the above range, extraction is performed using a method and an enzyme that are extracted under high temperature and high pressure.
- a method, a method of extracting using an acid such as vinegar or mineral acid, a method of extracting using an ion exchange resin, or the like may be adopted to achieve the above range.
- Panaxanaginseng root is used as a raw material, and water, ethanol, 2-propanol, butanol, or a mixed solvent thereof is used as an extraction solvent at a temperature of 60 to 100 ° C.
- the extraction solvent is preferably water, the extraction temperature is preferably 90 to 100 ° C., and the extraction time is preferably 20 to 40 hours.
- the extracted extract is isolated by separation by means of column chromatography using an aromatic column such as Diaion HP20 or an HPLC-ODS column, and (Rb2 + Rb1 + Rc + Rd) / (Rg3 + Rh2) is adjusted to the above range. Can do.
- Panax ginseng In addition to Panax ginseng, Panax notoginseng, Panax bipinnatifidus, Panax japonicus, Panax quinquefolius, Panax vietnamensis, Panax wangianus, Panax zingiberensis, Panax pseudogseng, Panax pseudogseng, Panax pseudogseng, Panax pseudogseng Good.
- the bath agent composition of the present invention contains a carbonate (B) in which the content of a dialkali metal carbonate in the carbonate is 30% by mass or more.
- component (B) When component (B) is added to the bath water of the bath preparation composition of the present invention, carbon dioxide is generated together with the organic acid of component (C) described later to bring about blood circulation promoting effect, and the skin is warmed well. A feeling can be imparted.
- the dialkali metal carbonate include one or two selected from sodium carbonate and potassium carbonate, and sodium carbonate is preferable from the viewpoint of ensuring high storage stability.
- the content of the dialkali metal carbonate in the carbonate (B) is 30% by mass or more, preferably 40% by mass or more, from the viewpoint of securing the appearance stability and enhancing the effect of suppressing coloring and discoloration. More preferably, it is 50 mass% or more.
- the content of the dialkali metal carbonate in the carbonate (B) is from the viewpoint of ensuring stability in appearance, from the viewpoint of maintaining good coloration and discoloration suppressing effect, and from the viewpoint of enhancing solubility in bath water. , Preferably it is 100 mass% or less, More preferably, it is 99 mass% or less, More preferably, it is 95 mass% or less.
- the content of the dialkali metal carbonate in the carbonate (B) is 30% by mass or more, preferably 30 to 100% by mass, more preferably 40 to 100% by mass, and still more preferably. It is 40 to 99% by mass, and more preferably 50 to 95% by mass.
- the bath agent composition of the present invention comprises, in component (B), carbonates other than dialkali metal carbonates, that is, monoalkali metal carbonates such as sodium hydrogen carbonate and potassium hydrogen carbonate, and divalent or higher metals such as calcium carbonate. 1 type (s) or 2 or more types selected from carbonates of Among these, as the carbonate other than the dialkali metal carbonate, a monoalkali metal carbonate is preferable from the viewpoint of securing the amount of carbon dioxide gas generated, and it is one or two selected from sodium hydrogen carbonate and potassium hydrogen carbonate. Is more preferable.
- the content of the monoalkali metal carbonate is preferably 60% by mass or less in the component (B) from the viewpoint of securing the amount of carbon dioxide gas generated and ensuring stability in appearance. It is 50 mass% or less.
- the content of the carbonate of the component (B) is sufficient in the bath preparation composition of the present invention from the viewpoint of securing a sufficient amount of carbon dioxide gas to produce a sufficient blood circulation promoting effect and enhancing the effect of suppressing coloring and discoloration.
- it is 20 mass% or more, More preferably, it is 25 mass% or more, More preferably, it is 30 mass% or more.
- the content of the carbonate of the component (B) is the bath preparation of the present invention from the viewpoint of securing the amount of carbon dioxide generated, the viewpoint of providing a sufficient blood circulation promoting effect, and the viewpoint of satisfactorily maintaining the effect of suppressing coloring and discoloration.
- a composition Preferably it is 70 mass% or less, More preferably, it is 65 mass% or less, More preferably, it is 60 mass% or less.
- the content of the carbonate of component (B) is preferably 20 to 70% by mass, more preferably 25 to 65% by mass, and further preferably 30 to 30% by mass in the bath agent composition of the present invention. 60% by mass.
- the mass ratio ((Rg3 + Rh2) / (B)) of the ginsenosides Rg3 and Rh2 of the component (A) and the component (B) is preferably from the viewpoint of enhancing both the blood circulation promoting effect and the coloring and discoloration suppressing effect. It is 0005 ⁇ 10 ⁇ 3 or more, more preferably 0.0025 ⁇ 10 ⁇ 3 or more, and further preferably 0.02 ⁇ 10 ⁇ 3 or more.
- the mass ratio of the component (A) ginsenosides Rg3 and Rh2 to the component (B) ((Rg3 + Rh2) / (B)) is preferably from the viewpoint of maintaining a good blood circulation promoting effect and a coloring and discoloration suppressing effect in a balanced manner.
- the mass ratio ((Rg3 + Rh2) / (B)) of ginsenosides Rg3 and Rh2 of component (A) to component (B) is preferably 0.0005 ⁇ 10 ⁇ 3 to 3 ⁇ 10 ⁇ 3 , more preferably 0 0025 ⁇ 10 ⁇ 3 to 2.5 ⁇ 10 ⁇ 3 , more preferably 0.02 ⁇ 10 ⁇ 3 to 2 ⁇ 10 ⁇ 3 .
- the bath agent composition of the present invention contains an organic acid (C).
- Such components (C) are solid at room temperature (25 ° C.) such as malic acid, tartaric acid, citric acid, maleic acid, succinic acid, phthalic acid, fumaric acid, glutaric acid, adipic acid, benzoic acid, salicylic acid and oxalic acid.
- the organic acid is preferred. These may be used alone or in combination of two or more. Among these, from the viewpoint of securing stability in appearance, one or more selected from citric acid, malic acid, succinic acid, and fumaric acid are preferable.
- the content of the component (C) is preferably in the bath preparation composition of the present invention from the viewpoint of securing a carbon dioxide gas generation amount and providing a sufficient blood circulation promoting effect and enhancing the effect of suppressing coloring and discoloration. Is 20% by mass or more, more preferably 25% by mass or more, and further preferably 30% by mass or more.
- the content of the component (C) is in the bath preparation composition of the present invention, from the viewpoint of securing a carbon dioxide gas generation amount and providing a sufficient blood circulation promoting effect, and maintaining a satisfactory effect of suppressing coloring and discoloration.
- it is 70 mass% or less, More preferably, it is 65 mass% or less, More preferably, it is 60 mass% or less.
- the content of the component (C) is preferably 20 to 70% by mass, more preferably 25 to 65% by mass, and further preferably 30 to 60% by mass in the bath agent composition of the present invention. It is.
- the bath agent composition of the present invention may further contain a dye (D), for example, 0.0001% by mass to 5.0% by mass, and 0.5% by mass or less. It is more preferable to contain 0.05 mass% or less, or (D) it is preferable not to contain a pigment
- the bath agent composition of the present invention contains the specific amount of the component (A), the component (B) and the component (C), and the (Rb2 + Rb1 + Rc + Rd) / (Rg3 + Rh2) is within a specific range. Since the appearance stability is high and coloring of the bath agent composition and discoloration over time can be effectively suppressed, the content of the pigment of the component (D) can be reduced or contained. There is no need, and good color can be maintained over a long period of time.
- the pigment of component (D) is meant to include pigments. Specifically, as component (D), specifically, Blue No. 1, Red No. 106, Red Tar pigments such as No. 2, Yellow No. 4, Yellow No. 202 (1) and Green No. 3; Natural pigments recognized as food additives such as chlorophyll, riboflavin, and natto; titanium oxide, zinc oxide, talc, kaolin, Examples include pigments such as bentonite and mica titanium. These dyes may be used alone or in combination of two or more.
- the bath agent composition of the present invention preferably contains a water-soluble polymer (E) from the viewpoint of enhancing the dispersibility of each of the above components when charged into bath water.
- the component (E) include glue, gelatin, collagen protein, casein, sodium alginate, carrageenan, fur celerane, tamarind gum, pectin, gum arabic, guar gum, xanthan gum, tragacanth gum, locust bean gum, dextrin, dextran, agar, Natural water-soluble polymers such as starch; semi-synthetic water-soluble such as carboxymethylcellulose, methylcellulose, hydroxyethylcellulose, hydroxypropylcellulose, cellulose acetate phthalate, propylene glycol alginate, oxidized starch, esterified starch, etherified starch, cationic starch Polymer: Synthetic water such as sodium polyacrylate, polyethyleneimine, polyvinyl alcohol, polyethylene glycol, polyvinylpyrrolidone Poly
- components other than the component (B) are coated with the component (E).
- the component other than the component (B) is an organic acid of the component (C) from the viewpoint of maintaining good storage stability while ensuring separability from the component (B) and increasing the amount of carbon dioxide generation. It is preferable.
- component (E) in the case of coating components other than component (B) polyethylene glycol is preferred.
- the mass ratio (the coated component / the coated component (E)) between the component other than the coated component (B) and the component (E) coated with such a component is preferably 4 to 90 More preferably, it is 6 to 40.
- the bath agent composition of this invention can contain a component (E) further as another component.
- the component (E) that covers components other than the component (B) and the component (E) as other components in the bath agent composition of the present invention may be the same. May be different.
- the content of the component (E) is preferably 0.5% by mass or more, more preferably 1% by mass or more in total in the bath agent composition of the present invention from the viewpoint of enhancing the dispersibility of each component. More preferably, it is 3% by mass or more, and further preferably 5% by mass or more.
- the content of the component (E) is, in total, preferably 30% by mass or less, more preferably 25% by mass or less in the bath agent composition of the present invention, from the viewpoint of securing the amount of carbon dioxide gas generated. .
- components other than the above components components usually used in bath agents, for example, nonionic surfactants, inorganic salts other than carbonates, vitamins, proteolytic enzymes, bactericidal preservatives Agents, excipients, fragrances, herbal medicines and the like may be contained as appropriate.
- nonionic surfactants for example, nonionic surfactants, inorganic salts other than carbonates, vitamins, proteolytic enzymes, bactericidal preservatives Agents, excipients, fragrances, herbal medicines and the like may be contained as appropriate.
- nonionic surfactants for example, nonionic surfactants, inorganic salts other than carbonates, vitamins, proteolytic enzymes, bactericidal preservatives Agents, excipients, fragrances, herbal medicines and the like may be contained as appropriate.
- anhydrous silicic acid having liquid absorbency, sodium sulfate capable of holding water as crystallization water, and the like.
- the bath agent composition of the present invention can be produced according to a conventional method such as a compression granulation method such as extrusion granulation or a compression molding method using a press tableting machine or a briquette machine after sufficiently mixing.
- the form may be any of powder, granule, granule, briquette tablet, tablet and the like.
- a part of the components may be granulated or molded in advance and then mixed with the other components and then molded.
- a component other than the component (B) is coated with the component (E) In this case, these components are mixed in advance and then granulated, and then mixed with the remaining components.
- the bath agent composition of the present invention is used by dissolving in bath water to generate carbon dioxide bubbles and immersing at least a part of the body in the bath water.
- the body part to be immersed may be a whole body bath in which the entire body is immersed, or may be only a part of the body such as the neck and shoulders, hands and arms, legs and legs.
- the bath preparation composition of the present invention is effective in accelerating blood circulation even in a body part with a thick stratum corneum, so that the feet can be soaked from the viewpoint of realizing good skin feel. Ideal as a footbath for use.
- the bath agent composition of the present invention has a pH of 4 to 7 in the bath water. It is preferable that the pH is 4.5 to 6.5.
- the bath agent composition of the present invention preferably has a carbon dioxide gas concentration of bath water of 30 ppm or more, and more preferably 60 ppm or more. It is preferable.
- the bathing temperature is preferably 30 to 45 ° C. in order to fully enjoy the blood circulation promoting effect and the effect of enhancing the skin feel during bathing by the bath preparation composition of the present invention.
- the bathing time for one time is preferably 3 minutes or more, more preferably 5 minutes or more, and even more preferably 10 minutes or more.
- the following bath agent composition is further disclosed regarding the embodiment of the present invention described above.
- the mass ratio ((Rb2 + Rb1 + Rc + Rd) / (Rg3 + Rh2)) is preferably 20 or less, more preferably 10 or less, still more preferably 7 or less, and even more preferably 3 or less.
- the content of the dialkali metal carbonate in component (B) is preferably 40% by mass or more, more preferably 50% by mass or more, and preferably 100% by mass or less, more preferably. Is 99% by mass or less, more preferably 95% by mass or less, and any one of [1] to [3] above.
- the dialkali metal carbonate is preferably one or two selected from sodium carbonate and potassium carbonate, more preferably sodium carbonate, the bath agent composition according to any one of the above [1] to [4] object.
- the carbonate other than the dialkali metal carbonate in component (B) is preferably one or more selected from a monoalkali metal carbonate and a carbonate of a divalent or higher metal, and more preferably.
- the content of component (B) is preferably 20% by mass or more, more preferably 25% by mass or more, still more preferably 30% by mass or more, and preferably 70% by mass or less.
- the mass ratio ((Rg3 + Rh2) / (B)) between ginsenosides Rg3 and Rh2 of component (A) and component (B) is preferably 0.0005 ⁇ 10 ⁇ 3 or more, more preferably 0.0025. ⁇ 10 ⁇ 3 or more, more preferably 0.02 ⁇ 10 ⁇ 3 or more, preferably 3 ⁇ 10 ⁇ 3 or less, more preferably 2.5 ⁇ 10 ⁇ 3 or less,
- the bath agent composition according to any one of the above [1] to [7], which is preferably 2 ⁇ 10 ⁇ 3 or less.
- Component (C) is preferably one selected from malic acid, tartaric acid, citric acid, maleic acid, succinic acid, phthalic acid, fumaric acid, glutaric acid, adipic acid, benzoic acid, salicylic acid and oxalic acid
- the content of component (C) is preferably 20% by mass or more, more preferably 25% by mass or more, still more preferably 30% by mass or more, and preferably 70% by mass or less.
- Component (D) is preferably one or more selected from tar dyes, natural dyes, and pigments, more preferably Blue No. 1, Red No. 106, Red No. 2, Yellow No.
- the content of the component (D) is preferably 0.0001% by mass or more and 5.0% by mass or less, more preferably 0.5% by mass or less, and more preferably 0.05% by mass or less. More preferably, or (D) the bath agent composition according to the above [11] or [12], which preferably contains no dye.
- the content of component (E) is preferably 0.5% by mass or more, more preferably 1% by mass or more, further preferably 3% by mass or more, and further preferably 5% by mass.
- Component (E) is preferably one or more selected from natural water-soluble polymers, semi-synthetic water-soluble polymers, and synthetic water-soluble polymers, more preferably polyethylene glycol and dextrin
- the bath agent composition according to [14] or [15] above which is one or two selected from the group consisting of polyethylene glycol and more preferably polyethylene glycol.
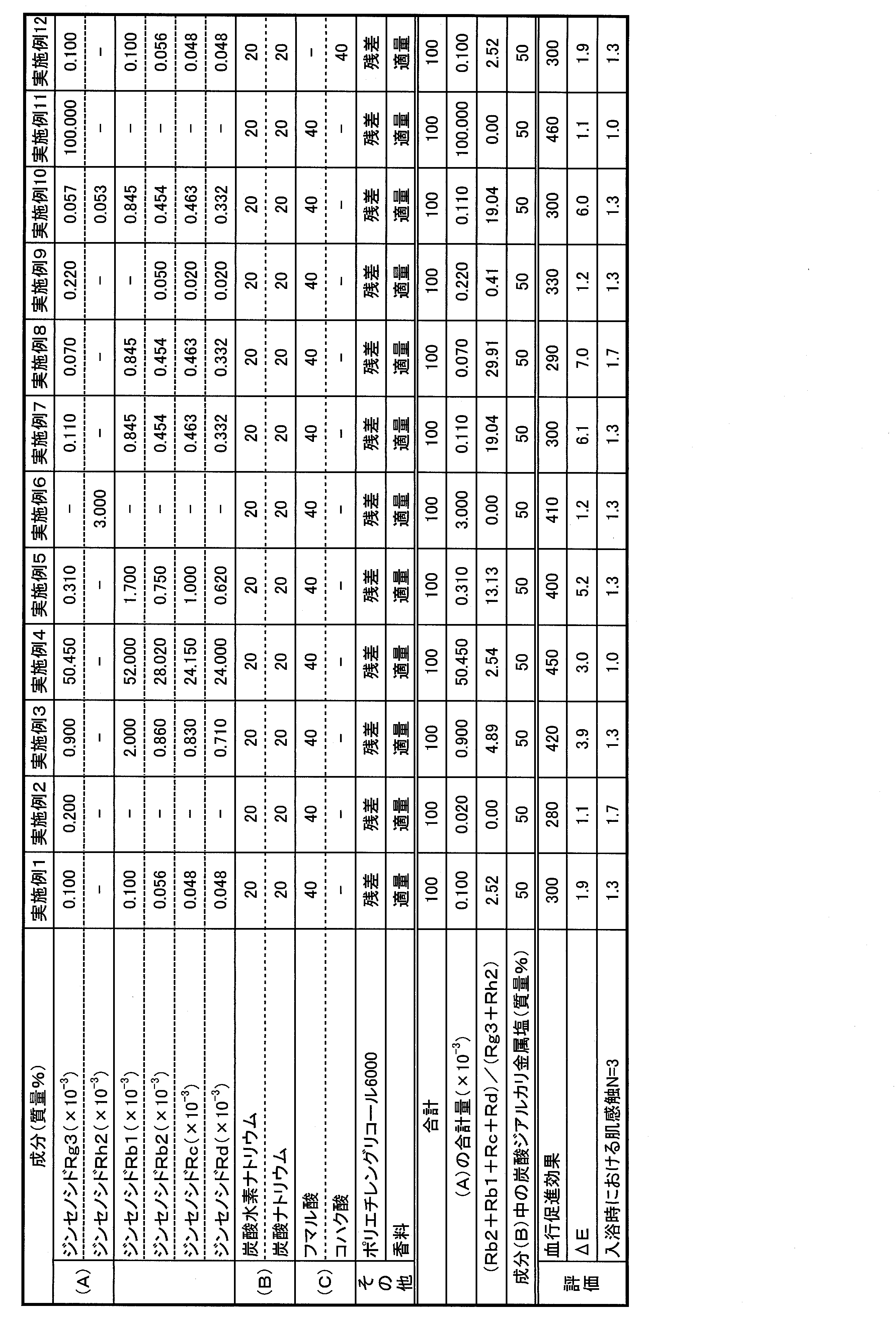
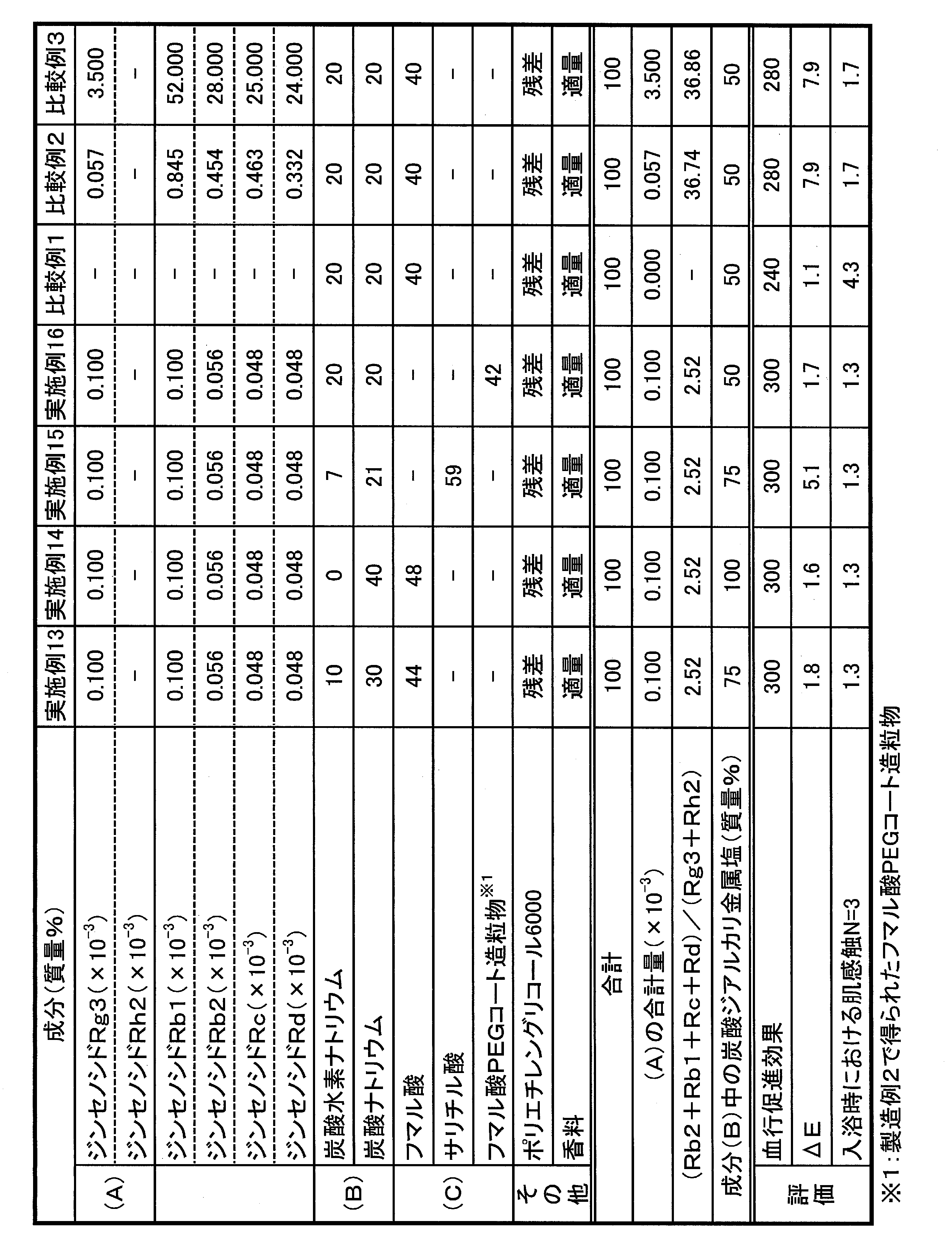
- Examples 1 to 16 Comparative Examples 1 to 3
- each component was mixed using the extract obtained in Production Example 1, and compression-molded into 45 g foamed tablets, thereby obtaining a bath preparation.
- the amounts of ginsenosides Rg3, Rh2, Rb2, Rb1, Rc and Rd in the bath preparation were determined by analysis using high performance liquid chromatography (HPLC) under the conditions shown in Table 1 below. HPLC conditions
- ⁇ Evaluation of blood circulation promotion effect 45 g of each bathing agent is put into 6 L of 40 ° C. hot water, and five subjects are immersed in the lower part from the ankle for 15 minutes in bath water with carbon dioxide concentration of 1000 ppm and pH 5.1. Measure skin blood flow (mL / min) before foot bathing and immediately after foot bathing, each blood flow before starting foot bath as 100, and individual blood flow 15 minutes after foot bathing is displayed as an index And the average value of 5 persons was calculated. The skin blood flow rate was measured using a laser Doppler blood flow meter (ALF21, manufactured by Advance Co., Ltd.) with an electrode attached to the back of the foot.
- ALF21 laser Doppler blood flow meter
- the bath preparations of Examples 1 to 16 were comparative example 1 containing no ginsenoside, and the mass ratio of ginsenoside Rg3 and Rh2 ((Rb2 + Rb1 + Rc + Rd) / (Rg3 + Rh2)) exceeded 30.
- the blood circulation promoting effect is exhibited well, the effect of suppressing coloring and discoloration, and the effect of improving skin feel during bathing are well balanced. I understand.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- General Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Epidemiology (AREA)
- Birds (AREA)
- Mycology (AREA)
- Microbiology (AREA)
- Botany (AREA)
- Biotechnology (AREA)
- Engineering & Computer Science (AREA)
- Emergency Medicine (AREA)
- Dermatology (AREA)
- Chemical & Material Sciences (AREA)
- Inorganic Chemistry (AREA)
- Cosmetics (AREA)
- Detergent Compositions (AREA)
Abstract
Description
(非特許文献1)Advances in Food and Nutrition Research, 2009, 55:1-99
(非特許文献2)Phytochemical Analysis, 2008, 19:2-16
(A)ジンセノシドRg3及び/又はジンセノシドRh2 合計で0.00001質量%以上0.5質量%以下、
(B)炭酸塩中の炭酸ジアルカリ金属塩の含有量が30質量%以上である炭酸塩、及び
(C)有機酸
を含有し、ジンセノシドRb2、Rb1、Rc及びRdと、ジンセノシドRg3及びRh2の質量比((Rb2+Rb1+Rc+Rd)/(Rg3+Rh2))が0以上30以下である浴用剤組成物を提供するものである。
本発明の浴用剤組成物は、ジンセノシドRg3及び/又はジンセノシドRh2(A)を0.00001質量%以上0.5質量%以下含有する。ジンセノシドRg3及びジンセノシドRh2は、オタネニンジンの有効成分としても知られるものであり、ジンセノシドRg3は下記式(1)で表され、ジンセノシドRh2は下記式(2)で表されるものである。
[1]次の成分(A)、(B)及び(C):
(A)ジンセノシドRg3及び/又はジンセノシドRh2 合計で0.00001質量%以上0.5質量%以下、
(B)炭酸塩中の炭酸ジアルカリ金属塩の含有量が30質量%以上である炭酸塩、及び
(C)有機酸
を含有し、ジンセノシドRb2、Rb1、Rc及びRdと、ジンセノシドRg3及びRh2の質量比((Rb2+Rb1+Rc+Rd)/(Rg3+Rh2))が0以上30以下である浴用剤組成物。
[2]成分(A)の含有量は、合計で、好ましくは0.00002質量%以上であり、好ましくは0.3質量%以下である上記[1]の浴用剤組成物。
[3]質量比((Rb2+Rb1+Rc+Rd)/(Rg3+Rh2))は、好ましくは20以下であり、より好ましくは10以下であり、さらに好ましくは7以下であり、またさらに好ましくは3以下であり、ことさらに好ましくは1以下である上記[1]又は[2]の浴用剤組成物。
[5]炭酸ジアルカリ金属塩は、好ましくは炭酸ナトリウム、及び炭酸カリウムから選ばれる1種又は2種であり、より好ましくは炭酸ナトリウムである上記[1]~[4]いずれか1の浴用剤組成物。
[6]成分(B)中における炭酸ジアルカリ金属塩以外の炭酸塩は、好ましくは炭酸モノアルカリ金属塩、及び二価以上の金属の炭酸塩から選ばれる1種又は2種以上であり、より好ましくは炭酸モノアルカリ金属塩であり、さらに好ましくは炭酸水素ナトリウム及び炭酸水素カリウムから選ばれる1種又は2種以上である上記[1]~[5]いずれか1の浴用剤組成物。
[7]成分(B)の含有量は、好ましくは20質量%以上であり、より好ましくは25質量%以上であり、さらに好ましくは30質量%以上であり、また、好ましくは70質量%以下であり、より好ましくは65質量%以下であり、さらに好ましくは60質量%以下である上記[1]~[6]いずれか1の浴用剤組成物。
[8]成分(A)のジンセノシドRg3及びRh2と成分(B)の質量比((Rg3+Rh2)/(B))は、好ましくは0.0005×10-3以上であり、より好ましくは0.0025×10-3以上であり、さらに好ましくは0.02×10-3以上であり、また、好ましくは3×10-3以下であり、より好ましくは2.5×10-3以下であり、さらに好ましくは2×10-3以下である上記[1]~[7]いずれか1の浴用剤組成物。
[9]成分(C)は、好ましくはリンゴ酸、酒石酸、クエン酸、マレイン酸、コハク酸、フタル酸、フマル酸、グルタル酸、アジピン酸、安息香酸、サリチル酸及びシュウ酸から選ばれる1種又は2種以上であり、より好ましくはクエン酸、リンゴ酸、コハク酸、及びフマル酸から選ばれる1種又は2種である上記[1]~[8]いずれか1の浴用剤組成物。
[10]成分(C)の含有量は、好ましくは20質量%以上であり、より好ましくは25質量%以上であり、さらに好ましくは30質量%以上であり、また、好ましくは70質量%以下であり、より好ましくは65質量%以下であり、さらに好ましくは60質量%以下である上記[1]~[9]いずれか1の浴用剤組成物。
[11]さらに、色素(D)を含有する、或いは色素(D)を含有しない、上記[1]~[10]いずれか1の浴用剤組成物。
[12]成分(D)は、好ましくはタール色素、天然色素、及び顔料から選ばれる1種又は2種以上であり、より好ましくは青色1号、赤色106号、赤色2号、黄色4号、黄色202(1)号、緑色3号等のタール色素、クロロフィル、リボフラビン、アンナット等の天然色素、酸化チタン、酸化亜鉛、タルク、カオリン、ベントナイト、雲母チタン等の顔料から選ばれる1種又は2種である上記[11]の浴用剤組成物。
[13]成分(D)の含有量は、0.0001質量%以上5.0質量%以下含有することが好ましく、0.5質量%以下含有することがより好ましく、0.05質量%以下含有することがさらに好ましく、或いは(D)色素を含有しないことが好ましい上記[11]又は[12]の浴用剤組成物。
[15]成分(E)の含有量は、好ましくは0.5質量%以上であり、より好ましくは1質量%以上であり、さらに好ましくは3質量%以上であり、ことさらに好ましくは5質量%以上であり、また、好ましくは30質量%以下であり、より好ましくは25質量%以下である上記[14]の浴用剤組成物。
[16]成分(E)は、好ましくは天然水溶性高分子、半合成水溶性高分子、及び合成水溶性高分子から選ばれる1種又は2種以上であり、より好ましくはポリエチレングリコール、及びデキストリンから選ばれる1種又は2種であり、さらに好ましくはポリエチレングリコールである上記[14]又は[15]の浴用剤組成物。
[17]成分(B)以外の成分が、成分(E)により被覆されてなる上記[14]~[16]いずれか1の浴用剤組成物。
[18]成分(B)以外の成分が、成分(C)である上記[17]の浴用剤組成物。
[19]さらに、油性成分を含有する上記[1]~[18]いずれか1の浴用剤組成物。
[21]組成物のpHが、好ましくは4~7であり、より好ましくは4.5~6.5である上記[1]~[20]いずれか1の浴用剤組成物。
[22]足浴剤である上記[1]~[21]いずれか1の浴用剤組成物。
[23]足浴のための上記[1]~[21]いずれか1の浴用剤組成物の使用。
[24]着色及び変色が抑制された浴用剤組成物を製造するための、上記[1]~[21]いずれか1の浴用剤組成物の使用。
チョウセンニンジン(Panax ginseng)の根(中国産)100gに水850mLを加え、100℃で30時間抽出し、ろ過後濃縮してニンジンエキスを得た。
7.6kgのフマル酸と0.4kgのPEG6000をヘンシェルミキサー(FM20B型)に投入し、加熱して70℃になるまで混合し、その後冷却して造粒物を得た。フマル酸とPEGの質量比(フマル酸/PEG)は、95/5であった。
表2~3に示す処方にしたがい、製造例1で得られたエキスを用いて各成分を混合し、1錠45gの発泡錠剤に圧縮成形して浴用剤を得た。なお、浴用剤中におけるジンセノシドRg3、Rh2、Rb2、Rb1、Rc及びRdの量は、高速液体クロマトグラフィー(HPLC)を下記表1に示す条件下で用いて分析することにより求めた。
HPLC条件
40℃の湯6Lに各浴用剤45gを投入して、炭酸ガス濃度1000ppm、pH5.1とした浴湯へ、5名の被験者に、15分間足首から下の部分を浸漬して足浴してもらい、足浴を開始する前と、足浴直後の皮膚血流量(mL/min)を測定し、足浴を開始する前の個々の血流量を100として、足浴開始15分後の個々の血流量を指数表示し、5人の平均値を算出した。なお、皮膚血流量は、足背部に電極を装着し、レーザードップラー血流計(ALF21、(株)アドバンス社製)を用いて測定した。
分光測色計CM-700D(コニカミノルタ製)を用い、製造した直後のものと、アルミ(7μm)積層フィルムにて包装し50℃で1日間保存したものとの色差ΔEを測定した。
各浴用剤45gを、40℃、6Lの浴水に溶解した浴水に、健常肌の者3名により、15分間入浴してもらい、入浴中の肌の感触(潤いのある油性感の高い滑らかなものであることを実感できる感触)を以下の基準で官能評価し、平均点を求めた。かかる値が低いほど、良好な結果であることを示す。
1:非常になめらか
2:なめらか
3:どちらとも言えない
4:あまりなめらかでない
5:なめらかでない
Claims (8)
- 次の成分(A)、(B)及び(C):
(A)ジンセノシドRg3及び/又はジンセノシドRh2 合計で0.00001質量%以上0.5質量%以下、
(B)炭酸塩中の炭酸ジアルカリ金属塩の含有量が30質量%以上である炭酸塩、及び
(C)有機酸
を含有し、ジンセノシドRb2、Rb1、Rc及びRdと、ジンセノシドRg3及びRh2の質量比((Rb2+Rb1+Rc+Rd)/(Rg3+Rh2))が0以上30以下である浴用剤組成物。 - さらに、(D)色素を0.001質量%以上0.1質量%以下含有するか、或いは(D)色素を含有しない請求項1に記載の浴用剤組成物。
- さらに(E)水溶性高分子を含有する請求項1又は2に記載の浴用剤組成物。
- 成分(B)以外の成分が、成分(E)により被覆されてなる請求項3に記載の浴用剤組成物。
- 成分(E)が、ポリエチレングリコールである請求項3又は4に記載の浴用剤組成物。
- ジンセノシドRb2、Rb1、Rc及びRdと、ジンセノシドRg3及びRh2の質量比((Rb2+Rb1+Rc+Rd)/(Rg3+Rh2))が、0以上10以下である請求項1~5のいずれか1項に記載の浴用剤組成物。
- ジンセノシドRg3及び/又はジンセノシドRh2が、オタネニンジンから得られるものである請求項1~6のいずれか1項に記載の浴用剤組成物。
- 成分(C)が、クエン酸、リンゴ酸、コハク酸、及びフマル酸から選ばれる1種又は2種以上である請求項1~7のいずれか1項に記載の浴用剤組成物。
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201480025778.6A CN105188658B (zh) | 2013-05-10 | 2014-05-09 | 浴用剂组合物 |
| EP14794702.2A EP2995294B1 (en) | 2013-05-10 | 2014-05-09 | Bathing agent composition |
| HK16104070.7A HK1216081B (zh) | 2013-05-10 | 2014-05-09 | 浴用剂组合物 |
| RU2015152777A RU2667641C2 (ru) | 2013-05-10 | 2014-05-09 | Композиция моющего средства |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2013-099817 | 2013-05-10 | ||
| JP2013099817 | 2013-05-10 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2014181852A1 true WO2014181852A1 (ja) | 2014-11-13 |
Family
ID=51867324
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2014/062430 Ceased WO2014181852A1 (ja) | 2013-05-10 | 2014-05-09 | 浴用剤組成物 |
Country Status (6)
| Country | Link |
|---|---|
| EP (1) | EP2995294B1 (ja) |
| JP (1) | JP6246656B2 (ja) |
| CN (1) | CN105188658B (ja) |
| RU (1) | RU2667641C2 (ja) |
| TW (1) | TWI627972B (ja) |
| WO (1) | WO2014181852A1 (ja) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2020534248A (ja) * | 2017-07-17 | 2020-11-26 | コスメティック ウォリアーズ エルティーディーCosmetic Warriors Ltd | 組成物 |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP6640481B2 (ja) * | 2015-07-30 | 2020-02-05 | 花王株式会社 | 固形状発泡性浴用剤 |
| CN107773696A (zh) * | 2016-08-31 | 2018-03-09 | 段工华 | 一种减压型沐浴养生宝 |
| KR102095387B1 (ko) * | 2018-12-10 | 2020-04-01 | 주식회사 상일기업 | 발 전용 티슈용 조성물 |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS60215618A (ja) | 1984-04-12 | 1985-10-29 | Kao Corp | 弱酸性入浴剤 |
| JP2003160497A (ja) * | 2001-11-22 | 2003-06-03 | Toshin Kagaku Kk | 皮膚外用剤 |
| JP2003286155A (ja) * | 2002-03-28 | 2003-10-07 | Rejino Color Kogyo Kk | 入浴剤用組成物 |
Family Cites Families (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS60215616A (ja) * | 1984-04-12 | 1985-10-29 | Kao Corp | 弱酸性入浴剤 |
| JPS60215617A (ja) * | 1984-04-12 | 1985-10-29 | Kao Corp | 弱酸性入浴剤 |
| JPH07116017B2 (ja) * | 1986-05-17 | 1995-12-13 | アース製薬株式会社 | 入浴剤 |
| JP2558188B2 (ja) * | 1991-06-10 | 1996-11-27 | 花王株式会社 | 入浴剤組成物 |
| RU2234913C1 (ru) * | 2003-12-04 | 2004-08-27 | Кочеров Максим Борисович | Средство для ванн "женьшень-2" |
| KR101096372B1 (ko) * | 2004-04-14 | 2011-12-20 | (주)아모레퍼시픽 | 진세노사이드를 함유하는 고분자 캡슐 및 그 제조 방법,및 이를 함유하는 화장료 조성물 |
| KR20060114774A (ko) * | 2005-05-02 | 2006-11-08 | 에스케이케미칼주식회사 | (20S)-진세노사이드 Rg3을 함유하는 주름 예방 및개선용 화장료 |
| KR100706111B1 (ko) * | 2005-07-12 | 2007-04-12 | 고성권 | 비타민 씨를 이용한 인삼 제제 및 이의 제조방법 |
| CN1718191A (zh) * | 2005-07-14 | 2006-01-11 | 中国医药研究开发中心有限公司 | 人参总次苷口腔崩解片及其制备方法和应用 |
| CN100423732C (zh) * | 2005-12-28 | 2008-10-08 | 金凤燮 | 高活性红参的制作方法 |
| US9193756B2 (en) * | 2008-02-19 | 2015-11-24 | National University Corporation Asahikawa Medical College | Adrenomedullin production enhancer |
| KR101074102B1 (ko) * | 2008-12-31 | 2011-10-17 | 주식회사 엔에이치한삼인 | 홍삼박을 주재로 한 입욕제조성물 및 그 제조방법 |
| JP6076719B2 (ja) * | 2012-12-06 | 2017-02-08 | 花王株式会社 | Cgrp応答性促進剤 |
-
2014
- 2014-05-09 EP EP14794702.2A patent/EP2995294B1/en active Active
- 2014-05-09 WO PCT/JP2014/062430 patent/WO2014181852A1/ja not_active Ceased
- 2014-05-09 CN CN201480025778.6A patent/CN105188658B/zh active Active
- 2014-05-09 TW TW103116614A patent/TWI627972B/zh active
- 2014-05-09 RU RU2015152777A patent/RU2667641C2/ru active
- 2014-05-09 JP JP2014097273A patent/JP6246656B2/ja active Active
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS60215618A (ja) | 1984-04-12 | 1985-10-29 | Kao Corp | 弱酸性入浴剤 |
| JP2003160497A (ja) * | 2001-11-22 | 2003-06-03 | Toshin Kagaku Kk | 皮膚外用剤 |
| JP2003286155A (ja) * | 2002-03-28 | 2003-10-07 | Rejino Color Kogyo Kk | 入浴剤用組成物 |
Non-Patent Citations (3)
| Title |
|---|
| ADVANCES IN FOOD AND NUTRITION RESEARCH, vol. 55, 2009, pages 1 - 99 |
| PHYTOCHEMICAL ANALYSIS, vol. 19, 2008, pages 2 - 16 |
| See also references of EP2995294A4 |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2020534248A (ja) * | 2017-07-17 | 2020-11-26 | コスメティック ウォリアーズ エルティーディーCosmetic Warriors Ltd | 組成物 |
Also Published As
| Publication number | Publication date |
|---|---|
| RU2667641C2 (ru) | 2018-09-21 |
| JP2014237633A (ja) | 2014-12-18 |
| TWI627972B (zh) | 2018-07-01 |
| EP2995294A4 (en) | 2017-01-18 |
| CN105188658B (zh) | 2020-03-10 |
| EP2995294B1 (en) | 2020-01-08 |
| JP6246656B2 (ja) | 2017-12-13 |
| CN105188658A (zh) | 2015-12-23 |
| EP2995294A1 (en) | 2016-03-16 |
| RU2015152777A3 (ja) | 2018-03-06 |
| RU2015152777A (ru) | 2017-06-20 |
| TW201519910A (zh) | 2015-06-01 |
| HK1216081A1 (zh) | 2016-10-14 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN103156243A (zh) | 电解质功能性运动饮料及其制备方法 | |
| CN102670445A (zh) | 一种中药修颜乳及其制备方法 | |
| JP6246656B2 (ja) | 浴用剤組成物 | |
| CN103027884B (zh) | 洗面盐砂及其制备方法 | |
| KR102093620B1 (ko) | 유용 성분이 고온용융 혼합된 기능성 소금 및 이의 제조 방법 | |
| JP2004010526A (ja) | 皮膚外用剤 | |
| KR20100064120A (ko) | 알로에베라와 알로에아보레센스를 주성분으로 함유하는 변비개선 및 예방기능을 갖는 겔상 식품조성물 | |
| CN107375007B (zh) | 一种护肤品组合物、氨基酸皂及其制备方法 | |
| CN105250199A (zh) | 含珍珠粉的可食用面膜 | |
| CN106720822A (zh) | 一种具有补肺益气作用的巧克力 | |
| JPH07277949A (ja) | 浴用剤組成物 | |
| CN108066275A (zh) | 一种可食玫瑰面膜及其制备方法 | |
| HK1216081B (zh) | 浴用剂组合物 | |
| CN114903129A (zh) | 一种浆果花色苷-胶原蛋白-透明质酸钠稳态化复合果汁的制备方法及应用 | |
| JP2005312301A (ja) | 食塩組成物 | |
| CN105394514B (zh) | 马齿苋固体饮料及其制备方法、以及用其制成的面膜粉 | |
| JP3403438B2 (ja) | 米発酵エキスの着色防止方法 | |
| CN105326659A (zh) | 美白抗皱精华液 | |
| CN105055212A (zh) | 芦荟花露水 | |
| KR100665966B1 (ko) | 규산염과 은행잎추출물로 제조되는 입욕제의 조성물 및제조제법 | |
| CN101637206A (zh) | 一种嚼茶 | |
| CN108522739A (zh) | 一种保健茶饮料及其制备方法 | |
| CN106074345A (zh) | 凹凸棒女士防晒霜 | |
| JPH0769862A (ja) | 入浴剤組成物 | |
| CN105919845A (zh) | 黄连杀菌爽口液 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 201480025778.6 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 14794702 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2014794702 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2015152777 Country of ref document: RU Kind code of ref document: A |