WO2017005698A1 - Organische moleküle zur verwendung in organischen optoelektronischen vorrichtungen - Google Patents
Organische moleküle zur verwendung in organischen optoelektronischen vorrichtungen Download PDFInfo
- Publication number
- WO2017005698A1 WO2017005698A1 PCT/EP2016/065723 EP2016065723W WO2017005698A1 WO 2017005698 A1 WO2017005698 A1 WO 2017005698A1 EP 2016065723 W EP2016065723 W EP 2016065723W WO 2017005698 A1 WO2017005698 A1 WO 2017005698A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- substituted
- organic
- group
- atoms
- formula
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- UUBOEJWLQGCYPO-UHFFFAOYSA-N CN(C)c(cc1)ccc1-c(cc1c2cc(-c(cc3)ccc3N(C)C)ccc22)ccc1[n]2-c(c(-c1ccccc1)c1)ccc1C#N Chemical compound CN(C)c(cc1)ccc1-c(cc1c2cc(-c(cc3)ccc3N(C)C)ccc22)ccc1[n]2-c(c(-c1ccccc1)c1)ccc1C#N UUBOEJWLQGCYPO-UHFFFAOYSA-N 0.000 description 1
- 0 CNc1c(*)c(*)c(*)c(*)c1*B* Chemical compound CNc1c(*)c(*)c(*)c(*)c1*B* 0.000 description 1
- PMUCKJDYIZQSDX-UHFFFAOYSA-N N#Cc(cc1-c2ccccc2)ccc1N1c(cccc2)c2Oc2c1cccc2 Chemical compound N#Cc(cc1-c2ccccc2)ccc1N1c(cccc2)c2Oc2c1cccc2 PMUCKJDYIZQSDX-UHFFFAOYSA-N 0.000 description 1
- TZMSYXZUNZXBOL-UHFFFAOYSA-N c(cc1)cc2c1Oc1ccccc1N2 Chemical compound c(cc1)cc2c1Oc1ccccc1N2 TZMSYXZUNZXBOL-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D215/00—Heterocyclic compounds containing quinoline or hydrogenated quinoline ring systems
- C07D215/02—Heterocyclic compounds containing quinoline or hydrogenated quinoline ring systems having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen atoms or carbon atoms directly attached to the ring nitrogen atom
- C07D215/04—Heterocyclic compounds containing quinoline or hydrogenated quinoline ring systems having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen atoms or carbon atoms directly attached to the ring nitrogen atom with only hydrogen atoms or radicals containing only hydrogen and carbon atoms, directly attached to the ring carbon atoms
- C07D215/06—Heterocyclic compounds containing quinoline or hydrogenated quinoline ring systems having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen atoms or carbon atoms directly attached to the ring nitrogen atom with only hydrogen atoms or radicals containing only hydrogen and carbon atoms, directly attached to the ring carbon atoms having only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, attached to the ring nitrogen atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D209/00—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom
- C07D209/56—Ring systems containing three or more rings
- C07D209/80—[b, c]- or [b, d]-condensed
- C07D209/82—Carbazoles; Hydrogenated carbazoles
- C07D209/86—Carbazoles; Hydrogenated carbazoles with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to carbon atoms of the ring system
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D209/00—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom
- C07D209/56—Ring systems containing three or more rings
- C07D209/80—[b, c]- or [b, d]-condensed
- C07D209/82—Carbazoles; Hydrogenated carbazoles
- C07D209/88—Carbazoles; Hydrogenated carbazoles with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to carbon atoms of the ring system
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D219/00—Heterocyclic compounds containing acridine or hydrogenated acridine ring systems
- C07D219/04—Heterocyclic compounds containing acridine or hydrogenated acridine ring systems with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to carbon atoms of the ring system
- C07D219/08—Nitrogen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D219/00—Heterocyclic compounds containing acridine or hydrogenated acridine ring systems
- C07D219/14—Heterocyclic compounds containing acridine or hydrogenated acridine ring systems with hydrocarbon radicals, substituted by nitrogen atoms, attached to the ring nitrogen atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D241/00—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings
- C07D241/36—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings condensed with carbocyclic rings or ring systems
- C07D241/38—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings condensed with carbocyclic rings or ring systems with only hydrogen or carbon atoms directly attached to the ring nitrogen atoms
- C07D241/46—Phenazines
- C07D241/48—Phenazines with hydrocarbon radicals, substituted by nitrogen atoms, directly attached to the ring nitrogen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D279/00—Heterocyclic compounds containing six-membered rings having one nitrogen atom and one sulfur atom as the only ring hetero atoms
- C07D279/02—1,2-Thiazines; Hydrogenated 1,2-thiazines
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D279/00—Heterocyclic compounds containing six-membered rings having one nitrogen atom and one sulfur atom as the only ring hetero atoms
- C07D279/10—1,4-Thiazines; Hydrogenated 1,4-thiazines
- C07D279/14—1,4-Thiazines; Hydrogenated 1,4-thiazines condensed with carbocyclic rings or ring systems
- C07D279/18—[b, e]-condensed with two six-membered rings
- C07D279/22—[b, e]-condensed with two six-membered rings with carbon atoms directly attached to the ring nitrogen atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D279/00—Heterocyclic compounds containing six-membered rings having one nitrogen atom and one sulfur atom as the only ring hetero atoms
- C07D279/10—1,4-Thiazines; Hydrogenated 1,4-thiazines
- C07D279/14—1,4-Thiazines; Hydrogenated 1,4-thiazines condensed with carbocyclic rings or ring systems
- C07D279/18—[b, e]-condensed with two six-membered rings
- C07D279/22—[b, e]-condensed with two six-membered rings with carbon atoms directly attached to the ring nitrogen atom
- C07D279/24—[b, e]-condensed with two six-membered rings with carbon atoms directly attached to the ring nitrogen atom with hydrocarbon radicals, substituted by amino radicals, attached to the ring nitrogen atom
- C07D279/26—[b, e]-condensed with two six-membered rings with carbon atoms directly attached to the ring nitrogen atom with hydrocarbon radicals, substituted by amino radicals, attached to the ring nitrogen atom without other substituents attached to the ring system
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D279/00—Heterocyclic compounds containing six-membered rings having one nitrogen atom and one sulfur atom as the only ring hetero atoms
- C07D279/10—1,4-Thiazines; Hydrogenated 1,4-thiazines
- C07D279/14—1,4-Thiazines; Hydrogenated 1,4-thiazines condensed with carbocyclic rings or ring systems
- C07D279/18—[b, e]-condensed with two six-membered rings
- C07D279/34—[b, e]-condensed with two six-membered rings with hetero atoms directly attached to the ring sulfur atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D409/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms
- C07D409/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings
- C07D409/10—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings linked by a carbon chain containing aromatic rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D409/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms
- C07D409/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D417/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00
- C07D417/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F7/00—Compounds containing elements of Groups 4 or 14 of the Periodic Table
- C07F7/02—Silicon compounds
- C07F7/08—Compounds having one or more C—Si linkages
- C07F7/0803—Compounds with Si-C or Si-Si linkages
- C07F7/081—Compounds with Si-C or Si-Si linkages comprising at least one atom selected from the elements N, O, halogen, S, Se or Te
- C07F7/0812—Compounds with Si-C or Si-Si linkages comprising at least one atom selected from the elements N, O, halogen, S, Se or Te comprising a heterocyclic ring
- C07F7/0816—Compounds with Si-C or Si-Si linkages comprising at least one atom selected from the elements N, O, halogen, S, Se or Te comprising a heterocyclic ring said ring comprising Si as a ring atom
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B15/00—Acridine dyes
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B19/00—Oxazine dyes
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B57/00—Other synthetic dyes of known constitution
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B57/00—Other synthetic dyes of known constitution
- C09B57/008—Triarylamine dyes containing no other chromophores
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K11/00—Luminescent materials, e.g. electroluminescent or chemiluminescent
- C09K11/06—Luminescent materials, e.g. electroluminescent or chemiluminescent containing organic luminescent materials
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/657—Polycyclic condensed heteroaromatic hydrocarbons
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/657—Polycyclic condensed heteroaromatic hydrocarbons
- H10K85/6572—Polycyclic condensed heteroaromatic hydrocarbons comprising only nitrogen in the heteroaromatic polycondensed ring system, e.g. phenanthroline or carbazole
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E10/00—Energy generation through renewable energy sources
- Y02E10/50—Photovoltaic [PV] energy
- Y02E10/549—Organic PV cells
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02P—CLIMATE CHANGE MITIGATION TECHNOLOGIES IN THE PRODUCTION OR PROCESSING OF GOODS
- Y02P70/00—Climate change mitigation technologies in the production process for final industrial or consumer products
- Y02P70/50—Manufacturing or production processes characterised by the final manufactured product
Definitions
- the invention relates to purely organic molecules and their use in organic light emitting diodes (OLEDs) and in other organic optoelectronic devices.
- Organic optoelectronic devices are characterized in that either electrical energy is converted into photons (organic light-emitting diodes, OLED or light-emitting electrochemical cells, LEEC) or the reverse process takes place (organic photovoltaics, OPV). It is important that these processes run as efficiently as possible. For the range of OLEDs, therefore, ideally materials with the highest possible photoluminescent quantum yield must be used.
- Limited efficiencies of OLED materials can be improved by using efficient materials that exhibit thermally activated delayed fluorescence (TADF), as up to 100% of the excitons can be used instead of 25% of the excitons formed in an OLED, in contrast to purely fluorescent materials ,
- TADF thermally activated delayed fluorescence
- the resulting triplet excitons can be converted into singlet excitons, from which state photons can then be emitted.
- the prerequisite for such a thermal reoccupation is a small energetic distance between the lowest excited singlet (Si) and triplet level (Ti). This can be achieved, for example, by using copper (I) complexes (see, for example, H. Yersin, U. Monkowius, T. Fischer, T.
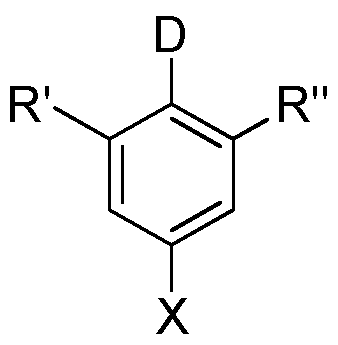
- the invention provides a new class of molecules having a structure of formula I or having a structure of formula I:
- X an electron-withdrawing unit, in particular CN or CF3;
- a and B independently selected from the group consisting of CRR 1 , CR, NR, N, wherein between A and B is a single or double bond and between B and Z is a single or double bond;
- R 3 for each occurrence is identical or different H, deuterium, F, CF3 or an aliphatic, aromatic and / or heteroaromatic hydrocarbon radical having 1 to 20 carbon atoms, in which one or more H atoms are replaced by F or CF3 can; can do it two or more substituents R 3 also together form a mono- or polycyclic aliphatic ring system;
- R ' selected from the group consisting of H, N (R 4 ) 2, OR 4 , a linear alkyl or alkoxy group having 1 to 40 carbon atoms or a branched or cyclic alkyl or alkoxy group having 3 to 40 carbon atoms each of which may be substituted with one or more R 4 radicals and an aromatic or heteroaromatic ring system of 5 to 60 aromatic ring atoms, each of which may be substituted by one or more R 4 radicals;
- R “ selected from the group consisting of N (R 4 ) 2, OR 4 , a linear alkyl or alkoxy group having 1 to 40 C atoms or a branched or cyclic alkyl or alkoxy group having 3 to 40 C atoms, the each may be substituted with one or more radicals R 4 , and an aromatic or heteroaromatic ring system having 5 to 60 aromatic ring atoms, each of which may be substituted by one or more radicals R 4 ;
- R 4 is identical or different at each instance and is H, deuterium, N (R 5 ) 2, Si (R 5 ) 3, a linear alkyl, alkoxy or thioalkoxy group having 1 to 40 C atoms or a branched or cyclic alkyl group.
- R 5 is identical or different at each instance and is H, deuterium, or an aliphatic, aromatic and / or heteroaromatic hydrocarbon radical having 1 to 20 C atoms; here may also form a mono- or polycyclic, aliphatic ring system with two or more substituents R. 5
- R ' is selected from the group consisting of H, N (R 4 ) 2, OR 4 , thiophene, a linear alkyl or alkoxy group having 1 to 40 carbon atoms, or one branched or cyclic alkyl or alkoxy group having 3 to 40 carbon atoms, each of which may be substituted by one or more R 4 , and an aromatic ring system having 5 to 60 aromatic ring atoms, which may be substituted by one or more R 4 radicals and R "is selected from the group consisting of N (R 4 ) 2, OR 4 , thiophene, a linear alkyl or alkoxy group having 1 to 40 carbon atoms or a branched or cyclic alkyl or alkoxy group having 3 to 40 ° C -Atomen, which may be substituted in each case with one or more radicals R 4 , and an aromatic ring system having 5 to 60 aromatic ring atoms, which may be substituted by one or more radicals R 4 .
- An aryl group in the sense of this invention contains 6 to 60 aromatic ring atoms;
- a heteroaryl group contains 5 to 60 aromatic ring atoms, at least one of which represents a heteroatom.
- the heteroatoms are in particular N, O, and / or S. This is the basic definition. If other preferences are given in the description of the present invention, for example with regard to the number of aromatic ring atoms or the heteroatoms contained, these apply.
- an aryl group or heteroaryl group is understood as meaning a simple aromatic cycle, ie benzene, or a simple heteroaromatic cycle, for example pyridine, pyrimidine or thiophene, or a heteroaromatic polycycle, for example phenanthrene, quinoline or carbazole.
- a condensed (fused) aromatic or heteroaromatic polycycle consists in the context of the present application of two or more fused simple aromatic or heteroaromatic cycles.
- An aryl or heteroaryl group which may be substituted in each case by the abovementioned radicals and which may be linked via any position on the aromatic or heteroaromatic compounds is understood in particular to mean groups which are derived from benzene, naphthalene, anthracene, phenanthrene, pyrene, Dihydropyrenes, chrysene, perylene, fluoranthene, benzanthracene, benzphenanthrene, tetracene, pentacene, benzpyrene, furan, benzofuran, isobenzofuran, dibenzofuran, thiophene, benzothiophene, isobenzothiophene, dibenzothiophene; Pyrrole, indole, isoindole, carbazole, pyridine, quinoline, isoquinoline, acridine, phenanthridine, benzo-5,6-quinoline, isoquinoline
- a cyclic alkyl, alkoxy or thioalkoxy group is understood as meaning a monocyclic, a bicyclic or a polycyclic group.
- a C 1 - to C 4 -alkyl group in which also individual H atoms or CH groups can be substituted by the abovementioned groups for example the radicals methyl, ethyl, n-propyl, i-propyl, Cyclopropyl, n-butyl, i-butyl, s-butyl, t-butyl, cyclobutyl, 2-methylbutyl, n-pentyl, s-pentyl, t-pentyl, 2-pentyl, neo-pentyl, cyclopentyl, n-hexyl, s-hexyl, t -hexyl, 2-hexyl, 3-hexyl, neo -hexyl, cyclohexyl, 1-methylcyclopentyl, 2-methylpentyl, n-heptyl, 2-heptyl,
- alkenyl group is understood as meaning, for example, ethenyl, propenyl, butenyl, pentenyl, cyclopentenyl, hexenyl, cyclohexenyl, heptenyl, cycloheptenyl, octenyl, cyclooctenyl or cyclooctadienyl.
- alkynyl group is meant, for example, ethynyl, propynyl, butynyl, pentynyl, hexynyl, heptynyl or octynyl.
- a C 1 to C 4 o-alkoxy group is meant for example methoxy, trifluoromethoxy, ethoxy, n-propoxy, i-propoxy, n-butoxy, i-butoxy, s-butoxy, t-butoxy or 2-methylbutoxy.
- the chemical entity D of the molecules according to the invention has donor properties. What is understood by donor or acceptor properties is known in principle to a person skilled in the art.
- the chemical entity D is electron-donating in one embodiment. It has a + M effect (positive mesomeric effect).
- suitable donor substituents include an atom having a lone pair of electrons, such as an N, O, or S atom.
- 5-membered heteroaryl groups having exactly one hetero ring atom are preferred. At this additional aryl groups can be fused.
- carbazole groups or carbazole derivatives are preferred.
- Further suitable donor substituents are phenoxazine groups or phenoxazine derivatives.
- the electron withdrawing moiety X exerts an M effect (negative mesomeric effect) or an I effect (negative inductive effect).
- the radical X is correspondingly an acceptor substituent. Suitable acceptor substituents are especially cyano groups or CF3.
- the molecules according to the invention have a substituent in the ortho position to the donor on the aromatic. This allows for effective separation of HOMO and LUMO of the organic molecule.
- the molecules according to the invention exhibit thermally activated delayed fluorescence and emit in particular in the deep blue region of the visible spectrum.
- OLED organic light-emitting diode
- an optoelectronic device for example an OLED
- OLEDs can be realized in the deep blue color spectrum.
- Corresponding OLEDs have a higher stability than OLEDs with known emitter materials and comparable color.
- the crosslinkable units QE in one embodiment are a compound selected from the group consisting of oxetanes, alkynes and azides, in particular for a click reaction, and the following alkene derivatives:
- D is independently a donor group with electron donating properties selected from the group consisting of substituted and unsubstituted carbazole, substituted and unsubstituted indole, substituted and unsubstituted indoline, substituted and unsubstituted dihydroacridine, substituted and unsubstituted benzimidazole, substituted and unsubstituted 2,3,4,9-tetrahydrocarbazole, substituted and unsubstituted 1,2,3,4-tetrahydroquinoline , substituted and unsubstituted phenothiazine, substituted and unsubstituted phenoxazine, substituted and unsubstituted dihydrophenazine, substituted and unsubstituted spiro compounds.
- the donor group electron-donating properties of the formula 1-1 has a structure of the formula II:
- the donor group with electron-donating properties of the formula 1-1 can be any organic compound with electron-donating properties of the formula 1-1.
- the accepting unit X of formula I in one embodiment is CN, in another embodiment CF3.
- the radical R 'of the formula I is a hydrogen atom, ie H.
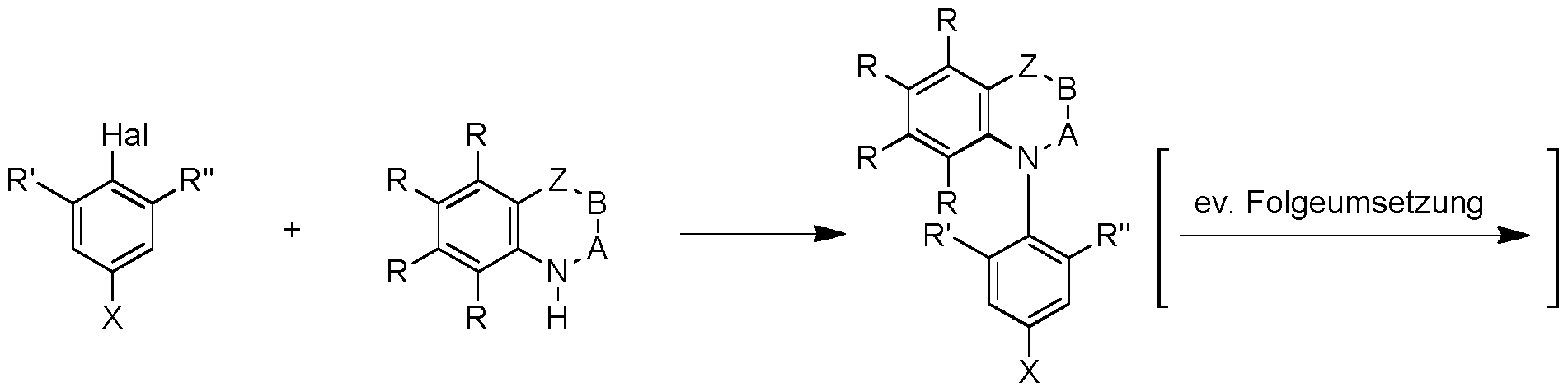
- the invention relates to a process for the preparation of an organic molecule according to the invention of the type described here (with a possible subsequent reaction).
- a nitrogen heterocycle is reacted in the sense of a nucleophilic aromatic substitution with an aryl halide, in particular an aryl fluoride.
- Typical conditions include the use of a base such as tribasic potassium phosphate or sodium hydride in an aprotic polar solvent such as dimetylsulfoxide (DMSO) or ⁇ , ⁇ -dimethylformamide (DMF).
- An alternative synthetic route involves the introduction of a nitrogen heterocycle via a copper- or palladium-catalyzed coupling with an aryl halide or aryl pseudohalide, in particular an aryl bromide, an aryl iodide, aryl triflate or an aryl tosylate.
- the described methods of preparation can represent both the last synthetic reaction and provide a precursor molecule, which can be implemented by subsequent reactions, for example by changing the radicals R, R 'or R "to the molecule of the invention.
- a further aspect of the invention relates to the use of an organic molecule of the type described here as a luminescent emitter or as an absorber, and / or as a host material and / or as an electron transport material, and / or as a hole injection material, and / or as a hole blocking material in an organic optoelectronic device.
- the organic optoelectronic device is in particular selected from the group consisting of:
- OLEDs Organic light-emitting diodes
- OLED sensors especially in non-hermetically shielded gas and vapor sensors
- the proportion of the organic molecule on the emission layer in an organic optoelectronic device in particular in OLEDs, is 1% to 99%, in particular 5% to 80% (% by weight). In an alternative embodiment, the proportion of the organic molecule at the emission layer is 100%.
- the light-emitting layer has a host material whose triplet (T1) and singlet (S1) energy levels are higher in energy than the triplet (T1) and singlet (S1) energy levels of the organic molecule.
- the invention relates to an organic optoelectronic device comprising an organic molecule of the type described here, in particular formed as a device selected from the group consisting of organic light emitting diode (OLED), light emitting electrochemical cell, OLED sensor, in particular non-hermetic externally shielded gas and vapor sensors, organic diode, organic solar cell, organic transistor, organic field effect transistor, organic laser and down-conversion element.
- OLED organic light emitting diode
- OLED sensor in particular non-hermetic externally shielded gas and vapor sensors
- organic diode organic solar cell
- organic transistor organic field effect transistor
- organic laser and down-conversion element organic laser and down-conversion element
- Such an organic optoelectronic device has, in one embodiment:
- a cathode wherein in particular the anode or the cathode are applied directly to the substrate, and
- At least one light-emitting layer which is arranged between the anode and cathode and having the organic molecule according to the invention.
- the optoelectronic device is an OLED.
- a typical OLED has, for example, the following layer structure:
- HIL hole injection layer
- HTL hole transport layer
- Electron blocking layer (EBL)
- EML emitter layer
- HBL hole blocking layer
- ETL electron transport layer
- EIL Electron Injection Layer
- individual layers are only available in an optional manner. Furthermore, several of these layers can coincide. And there may be multiple layers in the component multiple times.
- At least one electrode of the organic component is made translucent.
- translucent refers to a layer that is transparent to visible light.
- the translucent layer can be clear translucent, that is transparent, or at least partially light-absorbing and / or partially light-scattering, so that the translucent layer can also be translucent, for example, diffuse or milky.
- a layer designated here as translucent is formed as transparent as possible, so that in particular the absorption of light is as low as possible.
- the organic component in particular an OLED, has an inverted structure.
- the inverted structure is characterized in that the cathode is located on the substrate and the other layers are applied correspondingly inverted:
- EIL electron injection layer
- ETL electron transport layer
- HBL hole blocking layer
- Emission layer or emitter layer Emission layer or emitter layer (EML)
- Electron blocking layer (EBL)
- HTL hole transport layer
- HIL Hole injection layer
- the anode layer of the typical structure e.g. an ITO layer (indium tin oxide), connected as a cathode.
- the organic component in particular an OLED, has a stacked construction.
- the individual OLEDs are arranged one above the other and not next to each other as usual.
- the generation of mixed light can be made possible.
- this design can be used in the generation of white light, the entire visible spectrum of which is typically imaged by the combination of the emitted light from blue, green and red emitters.
- significantly longer lifetimes compared to conventional OLEDs can be achieved with virtually the same efficiency and identical luminance.
- a so-called charge generation layer (CGL) is optionally inserted between two OLEDs. This consists of an n-doped and a p-doped layer, wherein the n-doped layer is typically applied closer to the anode.
- a so-called tandem OLED - occur two or more emission layers between the anode and cathode.
- three emission layers are arranged one above the other, wherein an emission layer emits red light, an emission layer emits green light, and an emission layer emits blue light, and optionally further charge generation, blocking or transport layers are applied between the individual emission layers.
- the respective emission layers are applied directly adjacent.
- a charge generation layer is in each case located between the emission layers.
- emission layers directly adjacent and separated by charge generation layers can be combined in an OLED.
- An encapsulation arrangement can furthermore be arranged above the electrodes and the organic layers.
- the encapsulation arrangement can be embodied, for example, in the form of a glass cover or in the form of a thin-layer encapsulation.
- carrier material of the optoelectronic device for example, glass, quartz, plastic, metal, silicon wafers or any other suitable solid or flexible, optional serve transparent material.
- the carrier material may comprise, for example, one or more materials in the form of a layer, a film, a plate or a laminate.
- Transparent conductive metal oxides such as, for example, ITO (indium-tin-oxide), zinc oxide, tin oxide, cadmium oxide, titanium oxide, indium oxide or aluminum zinc oxide (AZO), Zn 2 Sn 4 , CdSnC, ZnSnC, Mgln 2 ⁇ D 4 , GaNO 3, can be used as the anode of the optoelectronic device.
- ITO indium-tin-oxide
- ZO aluminum zinc oxide
- Zn 2 Sn 4 CdSnC, ZnSnC, Mgln 2 ⁇ D 4 , GaNO 3
- Zn2ln20s or ln 4 Sn30i2 or mixtures of different transparent conductive oxides.
- PEDOT PSS (poly-3,4-ethylenedioxythiophene: polystyrene sulfonic acid), PEDOT (poly-3,4-ethylenedioxythiophene), m-MTDATA (4,4 ', 4 "-tris [phenyl (m) tolyl) amino] triphenylamine), spiro-TAD (2,2 ', 7,7'-tetrakis (N, N-diphenylamino) -9,9-spirobifluorene), DNTPD (4,4'-bis [N- [4 - ⁇ N, N-bis (3-methylphenyl) amino ⁇ phenyl] -N-phenylamino] biphenyl), NPNPB (N, N'-diphenyl-N, N'-di- [4- (N, N-) diphenyl-amino) phenyl] benzene), MeO-TPD (N,
- tertiary amines As materials of an HTL, tertiary amines, carbazole derivatives, polystyrenesulfonic acid-doped polyethylenedioxythiophene, camphorsulfonic acid-doped polyaniline poly-TPD (poly (4-butylphenyl-diphenyl-amine)), [alpha] -NPD (poly (4-butylphenyl-diphenyl-amine) )), TAPC (4,4'-cyclohexylidene bis [/ V, / V-bis (4-methylphenyl) benzenamine]), TCTA (tris (4-carbazoyl-9-ylphenyl) amine), 2-TNATA (4 , 4 ', 4 "-tris [2-naphthyl (phenyl) amino] triphenylamine), spiro-TAD, DNTPD, NPNPB, MeO-TPD, HAT-CN or TrisPcz
- the HTL may comprise a p-doped layer comprising an inorganic or organic dopant in an organic hole-conducting matrix.
- inorganic dopant for example, transition metal oxides such as vanadium oxide, molybdenum oxide or tungsten oxide can be used.
- organic dopants for example, tetrafluorotetracyanoquinodimethane (F4-TCNQ), copper pentafluorobenzoate (Cu (I) pFBz) or transition metal complexes can be used.
- an electron-blocking layer As materials of an electron-blocking layer it is possible to use, for example, mCP (1,3-bis (carbazol-9-yl) benzene), TCTA, 2-TNATA, mCBP (3,3-di (9H-carbazol-9-yl) biphenyl), tris Pcz (9,9'-diphenyl-6- (9-phenyl-9H-carbazol-3-yl) -9H, 9'H-3,3'-bicarbazole), CzSi (9- (4-tert-butylphenyl) 3,6-bis (triphenylsilyl) -9H-carbazole) or DCB (N, N'-dicarbazolyl-1, 4-dimethylbenzene).
- mCP 1,3-bis (carbazol-9-yl) benzene
- TCTA 2,3-di (9H-carbazol-9-yl) biphenyl
- Pcz 9,9'-diphenyl
- the emitter layer EML contains or consists of emitter material or a mixture comprising at least two emitter materials and optionally one or more host materials.
- Suitable host materials include, for example, mCP, TCTA, 2-TNATA, mCBP, Sif87 (dibenzo [b, d] thiophen-2-yltriphenylsilane), Sif88 (dibenzo [b, d] thiophen-2-yl) diphenylsilane), CBP (4, 4'-bis (N-carbazolyl) biphenyl) or DPEPO (bis [2- ((oxo) diphenylphosphino) phenyl] ether).
- the common matrix materials such as CBP are suitable.
- UHG matrix materials ultra-high energy gap materials
- CBP wide-gap Matrix materials
- the electron transport layer ETL can be, for example, materials based on AlQ3, TSP01, BPyTP2 (2,7-di (2,2'-bipyridin-5-yl) triphenyl) or BTB (4,4'-bis- [2- (4, 6-diphenyl-1, 3,5-triazinyl)] - 1, 1-biphenyl).
- EIL materials of a thin electron injection layer EIL, for example, CsF, LiF, 8-hydroxyquinolinolatolithium (Liq), L12O, BaF2, MgO or NaF can be used.
- the materials of the cathode layer may be metals or alloys, for example Al, Al> AIF, Ag, Pt, Au, Mg, Ag: Mg.
- Typical layer thicknesses are from 100 nm to 200 nm.
- one or more metals are used which are stable in air and / or which are self-passivating, for example by forming a thin protective oxide layer.
- alumina, vanadium oxide, zinc oxide, zirconium oxide, titanium oxide, hafnium oxide, lanthanum oxide, tantalum oxide are suitable.
- the person skilled in the art is aware of which combinations of materials are to be used for an optoelectronic device comprising an organic molecule according to the invention.
- the organic molecule according to the invention is used as the emission material in a light-emitting layer, wherein it is used either as a pure layer or in combination with a matrix material.
- the proportion of the organic molecule according to the invention at the emission layer in a light-emitting layer in optical light-emitting devices, in particular in OLEDs is between 5% and 80%.
- the light-emitting layer is applied to a substrate, wherein in particular an anode and a cathode are applied to the substrate and the light-emitting layer is applied between anode and cathode.
- the light-emitting layer may comprise only an organic molecule according to the invention in 100% concentration, wherein the anode and the cathode are applied to the substrate, and the light-emitting layer between the anode and cathode is applied.
- a hole and electron injecting layer between anode and cathode, and a hole and electron transporting layer between hole and electron injecting layer, and the light emitting layer between holes and electron transporting layer are applied.
- the organic optoelectronic device comprises in a further embodiment of the invention: a substrate, an anode, a cathode and at least one hole- and electron-injecting layer, and at least one hole- and electron-transporting layer, and at least one light-emitting layer, the organic according to the invention Molecule and a host material whose triplet (T1) and singlet (S1) energy levels are higher in energy than the triplet (T1) and singlet (S1) energy levels of the organic molecule, with the anode and cathode applied to the substrate and the hole and electron injecting layer is deposited between the anode and the cathode, and the hole and electron transporting layer is sandwiched between the hole and electron injecting layers Electron-injecting layer is applied, and the light-emitting layer between holes and electron-transporting layer is applied.
- the present invention also provides a light-emitting material comprising an organic molecule of the invention and a host material, wherein the triplet (T1) and singlet (S1) energy levels of the host material are higher than the triplet (T1) and singlet (S1) energy levels of the host material organic molecule, and wherein the light emitting material emits fluorescence or thermally activated delayed fluorescence, and a deltaE (S1 - T1) value between the lowest excited singlet (S1) and the underlying triplet (Tl) state of less than 3000 cm -1 .
- the invention relates to a method for producing an optoelectronic component.
- an organic molecule according to the invention is used.
- the manufacturing method comprises processing the organic molecule of the invention by a vacuum evaporation method or from a solution.
- the invention also provides a method for producing an optoelectronic device according to the invention, in which at least one layer of the optoelectronic device
- OVPD Organic Vapor Phase Deposition
- the optical glasses (cuvettes and substrates of quartz glass, diameter: 1 cm) were cleaned. Wash three times with dichloromethane, acetone, ethanol, demineralized water. Place in 5% Hellmanex solution for 24 h, rinse thoroughly with demineralised water, dry to rinse with nitrogen.
- the sample concentration corresponded to 10 mg / ml, stated in toluene or chlorobenzene.
- UV-VIS spectra were recorded on a Thermo Scientific Model Evolution 201 instrument. (See Sample Preparation: Solutions)
- Steady-state emission spectroscopy was performed with a Horiba Scientific FluoroMax-4 fluorescence spectrometer equipped with a 150 W xenon-arc lamp, excitation and emission monochromators and a Hamamatsu R928 photomultiplier tube, and a TCSPC option. Emission and excitation spectra were corrected by standard correction curves.
- the photoluminescence quantum yield was measured by means of an Absolute PL Quantum Yield Measurement C9920-03G system from Hamamatsu Photonics. It consists of a 150 W xenon gas discharge lamp, automatically adjustable Czerny-Turner monochromators (250-950 nm) and an Ulbricht sphere with highly reflective spectral coating (a Teflon derivative), which has a fiber optic cable with a PMA-12 multichannel detector BT (back thinned) CCD chip with 1024 x 122 pixels (size 24 x 24 ⁇ ) is connected.
- the evaluation of the quantum efficiency and the CIE coordinates took place with the help of the software U6039-05 version 3.6.0.
- the emission maximum is given in nm, the quantum yield ⁇ in% and the CIE color coordinates as x, y values.
- PLQY was determined for polymer films, solutions and powder samples according to the following protocol:
- the reference material is anthracenes in ethanol of known concentration. First, the absorbance maximum of the sample was determined and excited with it. Subsequently, degassed solutions and films under a nitrogen atmosphere were used to determine the absolute quantum yield.
- BP86 functional (Becke, AD Phys Rev. A1988, 38, 3098-3100, Perdew, JP Phys Rev. B1986, 33, 8822-8827) was used, with the resolution-of-identity Approach (RI) (Sierka, M., Hogekamp, A., Ahlrichs, RJ Chem. Phys., 2003, 18, 9136-9148; Becke, AD, J. Chem. Phys., 98 (1993) 5648-5652; Lee, C; Yang, W; Parr, RG Phys. Rev. B 37 (1988) 785-789).
- RI resolution-of-identity Approach
- Excitation energies were determined in the BP86 optimized structure using the time-dependent DFT method (TD-DFT) using the B3LYP functional (Becke, AD, J. Chem. Phys. 98 (1993) 5648-5652, Lee, C; Yang, W; Parr, RG Phys Rev. B 37 (1988) 785-789; Vosko, SH; Wilk, L; Nusair, M. Can. J. Phys. 58 (1980) 1200-121 1; Stephens, PJ Devlin, FJ; Chabalowski, CF; Frisch, MJJ Phys. Chem 98 (1994) 1 1623-1 1627).
- def2-SV (P) base sets Weigend, F., Ahlrichs, R. Phys. Chem. Chem. Phys., 2005, 7, 3297-3305, Rappoport, D .; Furche, FJ Chem. Phys. 2010, 133, 134105 / 1-134105 / 1
- All DFT calculations were performed with the Turbomole program package (version 6.5) (TURBOMOLE V6.4 2012, a development of University of Düsseldorf and Anlagens scholar Düsseldorf GmbH, 1989-2007, TURBOMOLE GmbH, since 2007; available from http: // www. turbomole.com). example 1
- the film emission of 1 (10% in PMMA) can be seen in FIG.
- the emission maximum is 443 nm.
- the photoluminescence quantum yield (PLQY) is 56%.
- the density functional theory calculations show a singlet (S1) energy of 2.86 eV and a triplet (T1) energy of 2.59 eV.
- the absorption spectrum of 2 as a solution in 2-methyltetrahydrofuran is shown in FIG.
- the film emission of 2 (10% in PMMA) can be seen in FIG.
- the emission maximum is 441 nm.
- the photoluminescence quantum yield (PLQY) is 55%.
- the film emission of 3 (10% in PMMA) is shown in FIG.
- the emission maximum is 494 nm.
- the photoluminescence quantum yield (PLQY) is 65%.
- the film emission of 4 (10% in PMMA) can be seen in FIG.
- the emission maximum is 466 nm.
- the photoluminescence quantum yield (PLQY) is 31%.
- the emission lifetime is 51 s.
- the film emission of 5 (10% in PMMA) can be seen in FIG.
- the emission maximum is 449 nm.
- the photoluminescence quantum yield (PLQY) is 65%.
- the film emission of 7 (10% in PMMA) is shown in FIG.
- the emission maximum is 477 nm.
- the photoluminescence quantum yield (PLQY) is 71%.
- the film emission of 8 (10% in PMMA) is shown in FIG. 11.
- the emission maximum is 462 nm.
- the photoluminescence quantum yield (PLQY) is 40%.
- the film emission of 9 (10% in PMMA) can be seen in FIG.
- the emission maximum is 404 nm.
- the photoluminescence quantum yield (PLQY) is 28%.
- Example 10 OLED component A
- Molecule 1 was tested in an OLED device having the following structure: ITO / m-MTDATA / HAT-CN / tris-Pcz / 1: DPEPO (20%) / TSPO1 / BPyTP2 / Liq / Al
- Table 1 Component data for component A.
- the current density and luminance over the voltage are shown in FIG.
- the power efficiency and current efficiency versus voltage are shown in FIG.
- the external quantum efficiency versus current density is shown in FIG.
- the electroluminescence spectrum operated at 10 V, the OLED is shown in Figure 16.
- Example 11 OLED component B
- Molecule 1 was tested in an OLED device having the following structure: ITO / m-MTDATA / a-NPD / TCTA / CzSi / 1 (10%): DPEPO / DPEPO / TPBi / Liq / Al Table 2: Component data for component B.
- the film emission of 19 (10% in PMMA) was measured.
- the emission maximum is 407 nm.
- the photoluminescence quantum yield (PLQY) is 30%.
- the film emission of 20 (10% in PMMA) was measured.
- the emission maximum is 440 nm.
- the photoluminescence quantum yield (PLQY) is 50%.
- the film emission of 20 (10% in PMMA) was measured.
- the emission maximum is 442 nm.
- the photoluminescence quantum yield (PLQY) is 52%.
- the film emission of 22 (10% in PMMA) was measured.
- the emission maximum is 408 nm.
- the photoluminescence quantum yield (PLQY) is 41%.
- the film emission of 23 (10% in PMMA) was measured.
- the emission maximum is 399 nm.
- the photoluminescence quantum yield (PLQY) is 31%.
- the film emission of 24 (10% in PMMA) was measured.
- the emission maximum is 441 nm.
- the photoluminescence quantum yield (PLQY) is 32%.
- the film emission of 25 (10% in PMMA) is shown in Figure F9.
- the emission maximum is 474 nm.
- the photoluminescence quantum yield (PLQY) is 52%.
- the film emission of 26 (10% in PMMA) was measured.
- the emission maximum is 448 nm.
- the photoluminescence quantum yield (PLQY) is 53%.
- the film emission of 27 (10% in PMMA) was measured.
- the emission maximum is 392 nm.
- the photoluminescence quantum yield (PLQY) is 30%.
- the film emission of 29 (10% in PMMA) was measured.
- the emission maximum is 416 nm.
- the photoluminescence quantum yield (PLQY) is 13%.
- the film emission of 30 (10% in PMMA) was measured.
- the emission maximum is 433 nm.
- the photoluminescence quantum yield (PLQY) is 34%.
- the film emission of 31 (10% in P MM A) was measured.
- the emission maximum is 397 nm.
- the photoluminescence quantum yield (PLQY) is 33%.
- the film emission of 32 (10% in PMMA) was measured.
- the emission maximum is 405 nm.
- the photoluminescence quantum yield (PLQY) is 37%.
- the film emission of 33 (10% in PMMA) was measured.
- the emission maximum is 394 nm.
- the photoluminescence quantum yield (PLQY) is 37%.
- the film emission of 34 (10% in PMMA) was measured.
- the emission maximum is 477 nm.
- the photoluminescence quantum yield (PLQY) is 33% and the half-width is 99 nm.
- Molecule 5 was tested in an OLED component ("component X2) with the following structure (proportion of the molecule according to the invention at the emission layer is given in mass percent):
- Figure 1 Absorption spectrum of 1 as a solution in 2-methyltetrahydrofuran.
- Figure 4 Absorption spectrum of 2 as a solution in 2-methyltetrahydrofuran.
- Figure 7 Film emission of 4 (10% in PMMA).
- Figure 8 Film emission of 5 (10% in PMMA).
- Figure 1 1 Film emission of 8 (10% in PMMA).
- Figure 12 Film emission of 9 (10% in PMMA).
- FIG. 13 Current density and luminance of the OLED component ITO / m-MTDATA / HATCN / tris-Pcz / 1: DPEPO (20%) / TSP01 / BPyTP2 / Liq / Al.
- Figure 15 External quantum efficiency over the current density of the OLED device ITO / m- MTDATA / HAT-CN / tris-Pcz / 1: DPEPO (20%) / TSP01 / BPyTP2 / Liq / AI.
- FIG. 16 Electroluminescence spectrum operated at 10 V of the OLED component ITO / m-MTDATA / HAT-CN / tris-Pcz / 1: DPEPO (20%) / TSP01 / BPyTP2 / Liq / Al.
- FIG. 17 Current density and luminance of the OLED component X2.
- FIG. 18 Power efficiency of the OLED component X2.
- FIG. 19 Current efficiency of the OLED component X2.
- FIG. 20 External quantum efficiency of the OLED component X2.
- FIG. 21 Electroluminescence spectrum of the OLED component X2, operated at 14 V.
- FIG. 21 Electroluminescence spectrum of the OLED component X2, operated at 14 V.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Physics & Mathematics (AREA)
- Spectroscopy & Molecular Physics (AREA)
- Electroluminescent Light Sources (AREA)
Abstract
Description
Claims
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US15/740,044 US12262635B2 (en) | 2015-07-03 | 2016-07-04 | Organic molecules for use in optoelectronic devices |
| CN201680039549.9A CN107925004B (zh) | 2015-07-03 | 2016-07-04 | 用于有机光电器件的有机分子 |
| KR1020187002739A KR102541444B1 (ko) | 2015-07-03 | 2016-07-04 | 유기 광전자 디바이스에 사용되는 유기 분자 |
| EP16747735.5A EP3317904B1 (de) | 2015-07-03 | 2016-07-04 | Organische moleküle zur verwendung in organischen optoelektronischen vorrichtungen |
| JP2017568275A JP6738838B2 (ja) | 2015-07-03 | 2016-07-04 | 有機光電子デバイスに使用するための有機分子 |
Applications Claiming Priority (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP15175358.9 | 2015-07-03 | ||
| EP15175358 | 2015-07-03 | ||
| EP15200813.2A EP3113239A1 (de) | 2015-07-03 | 2015-12-17 | Organische moleküle zur verwendung in organischen optoelektronischen vorrichtungen |
| EP15200813.2 | 2015-12-17 | ||
| EP16168821.3 | 2016-05-09 | ||
| EP16168821 | 2016-05-09 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2017005698A1 true WO2017005698A1 (de) | 2017-01-12 |
Family
ID=56372889
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2016/065723 Ceased WO2017005698A1 (de) | 2015-07-03 | 2016-07-04 | Organische moleküle zur verwendung in organischen optoelektronischen vorrichtungen |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2017005698A1 (de) |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2018202840A1 (en) * | 2017-05-04 | 2018-11-08 | Cynora Gmbh | Organic molecules, in particular for use in optoelectronic devices |
| WO2019053049A1 (en) * | 2017-09-18 | 2019-03-21 | Cynora Gmbh | ORGANIC MOLECULES, ESPECIALLY FOR USE IN OPTOELECTRONIC DEVICES |
| JP2020525438A (ja) * | 2017-06-23 | 2020-08-27 | 株式会社Kyulux | 有機発光ダイオードに用いられる組成物 |
| US11424417B2 (en) | 2018-11-16 | 2022-08-23 | Samsung Display Co., Ltd. | Organic electroluminescence device and compound for organic electroluminescence device |
| US11985893B2 (en) | 2019-11-08 | 2024-05-14 | Samsung Display Co., Ltd. | Organic electroluminescence device and aromatic compound for organic electroluminescence device |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2010149748A1 (de) | 2009-06-24 | 2010-12-29 | Hartmut Yersin | Kupfer-komplexe für optoelektronische anwendungen |
| WO2013161437A1 (ja) | 2012-04-25 | 2013-10-31 | 国立大学法人九州大学 | 発光材料および有機発光素子 |
| WO2014146752A1 (de) * | 2013-03-22 | 2014-09-25 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| WO2014146750A1 (de) * | 2013-03-22 | 2014-09-25 | Merck Patent Gmbh | Synthesebausteine für die herstellung von materialien für organische elektrolumineszenzvorrichtungen |
-
2016
- 2016-07-04 WO PCT/EP2016/065723 patent/WO2017005698A1/de not_active Ceased
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2010149748A1 (de) | 2009-06-24 | 2010-12-29 | Hartmut Yersin | Kupfer-komplexe für optoelektronische anwendungen |
| WO2013161437A1 (ja) | 2012-04-25 | 2013-10-31 | 国立大学法人九州大学 | 発光材料および有機発光素子 |
| WO2014146752A1 (de) * | 2013-03-22 | 2014-09-25 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| WO2014146750A1 (de) * | 2013-03-22 | 2014-09-25 | Merck Patent Gmbh | Synthesebausteine für die herstellung von materialien für organische elektrolumineszenzvorrichtungen |
Non-Patent Citations (13)
| Title |
|---|
| BECKE, A. D., PHYS. REV. A, vol. 38, 1988, pages 3098 - 3100 |
| BECKE, A.D., J.CHEM.PHYS, vol. 98, 1993, pages 5648 - 5652 |
| BECKE, A.D., J.CHEM.PHYS., vol. 98, 1993, pages 5648 - 5652 |
| LEE, C; YANG, W; PARR, R.G., PHYS. REV. B, vol. 37, 1988, pages 785 - 789 |
| M.E. THOMPSON ET AL., CHEM. MATER., vol. 16, 2004, pages 4743 |
| PERDEW, J. P., PHYS. REV. B, vol. 33, 1986, pages 8822 - 8827 |
| Q. ZHANG ET AL., J. AM. CHEM. SOC., vol. 134, 2012, pages 14706 |
| RAPPOPORT, D.; FURCHE, F., J. CHEM. PHYS., vol. 133, 2010, pages 134105,1 - 134105,11 |
| SHAOLONG GONG ET AL: "Simple CBP isomers with high triplet energies for highly efficient blue electrophosphorescence", JOURNAL OF MATERIALS CHEMISTRY, ROYAL SOCIETY OF CHEMISTRY, GB, vol. 22, no. 7, 1 January 2012 (2012-01-01), pages 2894 - 2899, XP002734092, ISSN: 0959-9428, [retrieved on 20111221], DOI: 10.1039/C1JM14903B * |
| SIERKA, M.; HOGEKAMP, A.; AHLRICHS, R. J., CHEM. PHYS., vol. 118, 2003, pages 9136 - 9148 |
| STEPHENS, P. J.; DEVLIN, F. J.; CHABALOWSKI, C. F.; FRISCH, M. J., J.PHYS.CHEM, vol. 98, 1994, pages 11623 - 11627 |
| VOSKO, S. H.; WILK, L.; NUSAIR, M., CAN. J. PHYS., vol. 58, 1980, pages 1200 - 1211 |
| WEIGEND, F.; AHLRICHS, R., PHYS. CHEM. CHEM. PHYS., vol. 7, 2005, pages 3297 - 3305 |
Cited By (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2018202840A1 (en) * | 2017-05-04 | 2018-11-08 | Cynora Gmbh | Organic molecules, in particular for use in optoelectronic devices |
| CN109923191A (zh) * | 2017-05-04 | 2019-06-21 | 西诺拉股份有限公司 | 特别用于光电器件的有机分子 |
| US10981930B2 (en) | 2017-05-04 | 2021-04-20 | Cynora Gmbh | Organic molecules for use in optoelectronic devices |
| JP2020525438A (ja) * | 2017-06-23 | 2020-08-27 | 株式会社Kyulux | 有機発光ダイオードに用いられる組成物 |
| JP7226806B2 (ja) | 2017-06-23 | 2023-02-21 | 株式会社Kyulux | 有機発光ダイオードに用いられる組成物 |
| US11638390B2 (en) | 2017-06-23 | 2023-04-25 | Kyulux, Inc. | Composition of matter for use in organic light-emitting diodes |
| WO2019053049A1 (en) * | 2017-09-18 | 2019-03-21 | Cynora Gmbh | ORGANIC MOLECULES, ESPECIALLY FOR USE IN OPTOELECTRONIC DEVICES |
| US11424417B2 (en) | 2018-11-16 | 2022-08-23 | Samsung Display Co., Ltd. | Organic electroluminescence device and compound for organic electroluminescence device |
| US11985893B2 (en) | 2019-11-08 | 2024-05-14 | Samsung Display Co., Ltd. | Organic electroluminescence device and aromatic compound for organic electroluminescence device |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP3317904B1 (de) | Organische moleküle zur verwendung in organischen optoelektronischen vorrichtungen | |
| EP3452471B1 (de) | Organische moleküle, insbesondere zur verwendung in optoelektronischen vorrichtungen | |
| DE102016112377B4 (de) | Organische Moleküle, insbesondere zur Verwendung in organischen optoelektronischen Vorrichtungen | |
| EP3113239A1 (de) | Organische moleküle zur verwendung in organischen optoelektronischen vorrichtungen | |
| DE102016110004B3 (de) | Organische Moleküle, insbesondere zur Verwendung in organischen optoelektronischen Vorrichtungen | |
| EP3507285A1 (de) | Organische moleküle, insbesondere zur verwendung in organischen optoelektronischen vorrichtungen | |
| EP3478657B1 (de) | Dicarbazolbiphenylderivate zur verwendung in optoelektronischen vorrichtungen | |
| DE102016108332B3 (de) | Organische Moleküle, insbesondere zur Verwendung in organischen optoelektronischen Vorrichtungen | |
| EP3478656B1 (de) | Dicarbazolbiphenylderivate zur verwendung in optoelektronischen vorrichtungen | |
| DE102016112082B4 (de) | Organische Moleküle, insbesondere zur Verwendung in organischen optoelektronischen Vorrichtungen | |
| DE102016108334B3 (de) | Organische Moleküle, insbesondere zur Verwendung in organischen optoelektronischen Vorrichtungen | |
| DE102016115853B4 (de) | Organische Moleküle, insbesondere zur Verwendung in organischen optoelektronischen Vorrichtungen | |
| WO2018153510A1 (de) | Organische moleküle, insbesondere zur verwendung in organischen optoelektronischen vorrichtungen | |
| WO2017005698A1 (de) | Organische moleküle zur verwendung in organischen optoelektronischen vorrichtungen | |
| EP3494112B1 (de) | Organische moleküle zur verwendung in organischen optoelektronischen vorrichtungen | |
| DE102017102662B4 (de) | Organische Moleküle, insbesondere zur Verwendung in organischen optoelektronischen Vorrichtungen | |
| DE102016108335B3 (de) | Organische Moleküle, insbesondere zur Verwendung in organischen optoelektronischen Vorrichtungen | |
| DE102016108327B3 (de) | Organische Moleküle, insbesondere zur Verwendung in organischen optoelektronischen Vorrichtungen | |
| WO2018077492A1 (de) | Organische moleküle, insbesondere zur verwendung in organischen optoelektronischen vorrichtungen | |
| WO2019002175A1 (de) | Organische moleküle, insbesondere zur verwendung in optoelektronischen vorrichtungen | |
| DE102017102363A1 (de) | Organische Moleküle, insbesondere zur Verwendung in organischen optoelektronischen Vorrichtungen | |
| WO2018145995A2 (de) | Organische moleküle, insbesondere zur verwendung in organischen optoelektronischen vorrichtungen | |
| EP3494113B1 (de) | Organische moleküle zur verwendung in optoelektronischen vorrichtungen | |
| WO2019197407A1 (de) | Pyridin-substituierte 2,2'-di-9h-carbazol-9-yl-[1,1'-biphenyl]-dicarbonitril-derivate und verwandte verbindungen zur verwendung in optoelektronischen vorrichtungen | |
| DE102016115728A1 (de) | Organische Moleküle, insbesondere zur Verwendung in organischen optoelektronischen Vorrichtungen |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 16747735 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2017568275 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 20187002739 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2016747735 Country of ref document: EP |
|
| WWG | Wipo information: grant in national office |
Ref document number: 15740044 Country of ref document: US |