WO2017170896A1 - Procédé de production de glycoprotéine contenant un groupe mannose-6-phosphate et procédé de détection de la distribution intracellulaire de glycoprotéine contenant un groupe mannose-6-phosphate capable de se lier à un groupe fluorescent - Google Patents
Procédé de production de glycoprotéine contenant un groupe mannose-6-phosphate et procédé de détection de la distribution intracellulaire de glycoprotéine contenant un groupe mannose-6-phosphate capable de se lier à un groupe fluorescent Download PDFInfo
- Publication number
- WO2017170896A1 WO2017170896A1 PCT/JP2017/013322 JP2017013322W WO2017170896A1 WO 2017170896 A1 WO2017170896 A1 WO 2017170896A1 JP 2017013322 W JP2017013322 W JP 2017013322W WO 2017170896 A1 WO2017170896 A1 WO 2017170896A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- sugar chain
- man
- man6p
- glcnac
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N9/00—Enzymes; Proenzymes; Compositions thereof; Processes for preparing, activating, inhibiting, separating or purifying enzymes
- C12N9/14—Hydrolases (3)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12P—FERMENTATION OR ENZYME-USING PROCESSES TO SYNTHESISE A DESIRED CHEMICAL COMPOUND OR COMPOSITION OR TO SEPARATE OPTICAL ISOMERS FROM A RACEMIC MIXTURE
- C12P19/00—Preparation of compounds containing saccharide radicals
- C12P19/26—Preparation of nitrogen-containing carbohydrates
- C12P19/28—N-glycosides
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12P—FERMENTATION OR ENZYME-USING PROCESSES TO SYNTHESISE A DESIRED CHEMICAL COMPOUND OR COMPOSITION OR TO SEPARATE OPTICAL ISOMERS FROM A RACEMIC MIXTURE
- C12P21/00—Preparation of peptides or proteins
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
Definitions
- the present disclosure relates to a method for producing a mannose-6-phosphate group-containing glycoprotein and a method for detecting the intracellular distribution of a fluorescent group-bound mannose-6-phosphate group-containing glycoprotein.
- lysosomal diseases are intractable diseases that develop due to genetic defects such as hydrolases (lysosomal enzymes) present in lysosomes, which are intracellular organelles.
- Current treatment methods include prepared enzymes (recombinant enzymes).
- Enzyme replacement therapy is known which is administered by intravenous administration. Enzyme replacement therapy is associated with binding of a non-reducing mannose-6-phosphate group (hereinafter referred to as Man6P) of an N-linked sugar chain of a glycoprotein to Man6P receptors present on the cell surface and lysosomes.
- Man6P non-reducing mannose-6-phosphate group
- a target glycoprotein is to be introduced into a target cell or organelle with high efficiency, the glycoprotein in which Man6P is added to the non-reducing end of the N-linked sugar chain is used. It is important that enzyme replacement therapy with a high therapeutic effect is possible.
- the phosphate group is not necessarily added to the glycoprotein, and there is a problem that the content of Man6P cannot be increased depending on the glycoprotein. Another problem is that many glycoproteins that are not bound to Man6P are mixed into the resulting glycoprotein due to the influence of phosphatases from cells serving as hosts.
- end M reports a method in which a large oligosaccharide chain site on the non-reducing end side of an N-linked sugar chain, which is a sugar chain donor, is transferred to a sugar chain acceptor at one time.
- a mutant (glycosynthase) in which the amino acid of endo M is modified can efficiently transfer a sugar chain of a sugar chain donor containing a structure derived from a target N-linked sugar chain to a sugar chain acceptor.
- the glycosynthase can transfer a sugar chain to a sugar chain acceptor using an N-linked sugar chain having Man6P as a sugar chain donor, the desired sugar having a high content of Man6P is consequently obtained. It can be expected that protein will be obtained.
- the sugar chain donor used in the above document uses an oxazolined compound that requires strong acidic conditions for its preparation
- an oxazolineated sugar chain donor having Man6P is used.
- it has a drawback that it is necessary to oxazoline so as not to eliminate a phosphate group that is weak in acidic conditions.
- the oxazoline ring is unstable, it is difficult to introduce a phosphate group after oxazolinization.
- sugar chain transfer reaction of the oxazolineized sugar chain donor there is a concern that the sugar chain transfer also occurs on residues other than GlcNAc of the sugar chain acceptor, for example, amino acid residues.
- the present disclosure has been made in view of the above, and a production method capable of easily producing a Man6P-containing glycoprotein using a glycosyl transfer reaction, and a fluorescent group-bound mannose-6-phosphate group-containing glycoprotein cell It is an object to provide a method for detecting an internal distribution.
- a mannose-6-phosphate group-containing glycoprotein is produced by producing a mannose-6-phosphate group-containing glycoprotein by a sugar chain transfer reaction between a sugar chain receptor represented by the following general formula (1):
- (b) Other than the 175th amino acid residue of the amino acid sequence of (a)
- An endo M mutant having an activity of catalyzing a chain transfer reaction.
- Y 1 represents an acylamino group containing a structure derived from a glycoprotein.
- GlcNAc represents an N-acetylglucosaminyl group.
- Y 1 in the general formula (1) is an acylamino group containing a structure derived from a lysosomal enzyme.
- ⁇ 3> The method for producing a mannose-6-phosphate group-containing glycoprotein according to ⁇ 1> or ⁇ 2>, wherein the sugar chain donor is represented by the following general formula (2).
- X 1 , X 2 , X 3 , X 4 , X 5 and X 6 each independently represent a hydrogen atom or a saccharide-derived group, and X 1 , X 2 , X 3 , X 4, at least one of X 5 and X 6, the non-reducing end is a group derived from a saccharide having a mannose-6-phosphate group .
- Z 1 represents a hydrogen atom or a GlcNAc
- Z 1 is GlcNAc
- the GlcNAc is bound to Man linked to GlcNAc at ⁇ 1-4 by ⁇ 1-4
- Y 2 represents a monovalent substituent
- GlcNAc represents an N-acetylglucosaminyl group.
- ⁇ 1-4 represents a ⁇ glycoside bond between position 1 of GlcNAc and position 4 of GlcNAc, or ⁇ glycoside bond between position 1 of Man and position 4 of GlcNAc
- Man represents a mannosyl group
- ⁇ 1-6 represents Man 1st and Man 6th It represents ⁇ -glycosidic bond
- [alpha] 1-3 represents a ⁇ -glycosidic bond between the 1- and 3-position of Man of Man.
- ⁇ 4> The method for producing a mannose-6-phosphate group-containing glycoprotein according to any one of ⁇ 1> to ⁇ 3>, wherein the sugar chain donor is represented by the following general formula (3).
- X 7 , X 8 and X 9 each independently represent a hydrogen atom, Man, Man ⁇ 1-2Man, Man6P, Man6P ⁇ 1-2Man or Man6P ⁇ 1-6Man.
- Man represents a mannosyl group.
- Man6P represents a mannosyl group phosphoric acid group is bonded to the 6-position.
- X 7 -6 indicates the X 7 bound to 6-position of Man
- X 8 -3 is attached to the 3-position of Man and showed X 8, X 9 -2 at least one .
- X 7, X 8 and X 9 showing the X 9 attached to the 2-position of Man is Man6P
- either Man6P ⁇ 1-2Man and Man6P ⁇ 1-6Man GlcNAc represents an N-acetylglucosaminyl group
- ⁇ 1-6 is an ⁇ -glycoside bond between position 1 of Man and position 6 of Man or position 1 of Man6P and M
- n represents the ⁇ -glycoside bond at the 6-position
- ⁇ 1-3 represents the ⁇ -glycoside bond between the 1-position of Man and the 3-position of Man
- ⁇ 1-2 represents the ⁇ -glycoside between Man's 1-position and Man's 2-position
- ⁇ 1-4 represents a ⁇ -glyco
- Fluorescent group-bound mannose obtained by further introducing a fluorescent group into the mannose-6-phosphate group-containing glycoprotein obtained by the production method according to any one of ⁇ 1> to ⁇ 4> A method for detecting intracellular distribution of a fluorescent group-bound mannose-6-phosphate group-containing glycoprotein by applying a -6-phosphate group-containing glycoprotein to cells.
- a production method capable of easily producing a Man6P-containing glycoprotein using a glycosyl transfer reaction, and a method for detecting intracellular distribution of a fluorescent group-bound mannose-6-phosphate group-containing glycoprotein. can be provided.
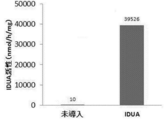
- FIG. 1 is a diagram showing the IDUA activity of an extract obtained from a middle silk gland produced by a transgenic silkworm introduced with a human ⁇ -iduronidase (IDUA) gene.
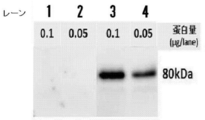
- FIG. 2 is a diagram showing the results of Western blotting using an anti-IDUA antibody on an extract obtained from a middle silk gland produced by a transgenic silkworm into which an IDUA gene has been introduced.
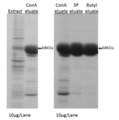
- FIG. 3 is a diagram showing the results of SDS-PAGE of a solution obtained by purifying the extract obtained from the middle silk gland of a transgenic silkworm introduced with the IDUA gene by each chromatography operation.
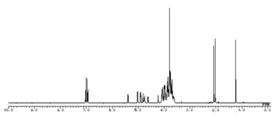
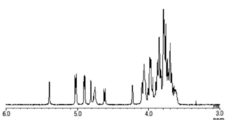
- FIG. 4 is a diagram showing a 1 H-NMR spectrum of Compound 12, which is a sugar chain donor in the present disclosure.
- FIG. 5 is an enlarged view of the 1 H-NMR spectrum of FIG.
- FIG. 6 is a diagram showing a mass spectrum of Compound 12 obtained by MALDI-TOF MS measurement.
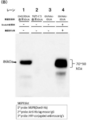
- FIG. 7A is a graph showing the results of SDS-PAGE of a sample after sugar chain transfer to a sugar chain acceptor (IDUA after endoenzyme treatment (GlcNAc-IDUA)) of a sugar chain donor (compound 12). It is.
- Lane 1 is a sample containing IDUA obtained from Chinese hamster (CHO) cells into which IDUA gene has been introduced
- Lane 2 is a sample containing IDUA before endo-enzyme treatment
- Lane 3 is a sugar chain receptor (endo).
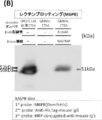
- FIG. 7B is a view showing the results of lectin blotting with the same sample as FIG. 7A using a lectin (Dom9-His) that specifically binds to Man6P.
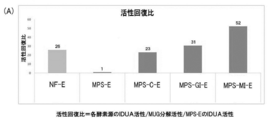
- FIG. 8A is a graph showing an activity recovery ratio in intracellular IDUA activity after adding M6P-containing IDUA to mucopolysaccharidosis type 1 (MPS-1) patient skin fibroblasts and the like and incubating for 24 hours.
- MPS-1 mucopolysaccharidosis type 1
- FIG. 8B shows 4-methylumbelliferyl (intracellular solution (enzyme source)) of 4-methylumbelliferyl obtained after adding M6P-containing IDUA to mucopolysaccharidosis type 1 (MPS-1) patient skin fibroblasts and the like.
- FIG. 4 is a graph showing the measurement of ⁇ -hexosaminidase (Hex) (control enzyme) activity against 4MU) -N-acetyl- ⁇ -D-glucopyranoside (MUG).
- FIG. 9 is a schematic diagram showing a method for modifying a fluorescent group to a Man6P-containing compound (Man6P-Man5-AFO).
- FIG. 10 is a diagram showing a MALDI-TOF MS spectrum of a fluorescent group-modified Man6P-containing compound (Man6P-Man5-AFO).
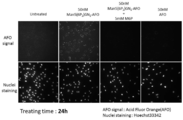
- FIG. 11 shows mucopolysaccharidosis type 1 (MPS-1) patient skin fibroblasts observed with a fluorescence microscope after addition of a fluorescent group-modified Man6P-containing compound (Man6P-Man5-AFO) and incubation for 24 hours. It is a figure which shows a result.
- FIG. 12 shows that mucopolysaccharidosis type 1 (MPS-1) patient skin fibroblasts after addition of fluorescent group-modified Man6P-containing IDUA (Man6P-IDUA-AFO) and incubation for 24 hours were observed with a fluorescence microscope.
- FIG. 13A is a graph showing the results of SDS-PAGE of a sample after sugar chain transfer to a sugar chain acceptor (CTSA after endoenzyme treatment (GlcNAc-CTSA)) of a sugar chain donor (compound 12). It is.
- Lane 1 is a sample containing CTSA obtained from Chinese hamster (CHO) cells into which a CTSA gene has been introduced
- lane 2 is a sample containing a sugar chain receptor (CTSA after endoenzyme treatment (GlcNAc-CTSA)).
- lane 3 is a sample after the sugar chain transfer of the sugar chain donor (compound 12) to the sugar chain acceptor (GlcNAc-CTSA).
- FIG. 13B is a diagram showing the result of lectin blotting with the same sample as FIG. 13A using a lectin (Dom9-His) that specifically binds to Man6P.
- Endo M is a kind of “Endoenzyme”, and “Endoenzyme” has the same meaning as “Endo- ⁇ -N-acetylglucosaminidase” and extends the end of the arrow in the following general formula (4).
- An “endoenzyme variant” is one in which one or more amino acid residues of an endoenzyme amino acid have been deleted, added or substituted.
- the substance to be subjected to sugar chain transfer refers to a sugar chain donor and a sugar chain acceptor in a sugar chain transfer reaction.
- X represents an oligosaccharide that binds to the 4-position of GlcNAc on the non-reducing end side of the reducing end chitobiosyl residue (GlcNAc ⁇ 1-4GlcNAc) in the N-linked sugar chain
- R represents a polypeptide Indicates.
- the arrow represents the position where the endoenzyme or endoenzyme variant is hydrolyzed.
- Transglycosylation means a part of a sugar chain structure of a sugar chain donor, for example, in the case of the general formula (4), a part on the left side from the position where the tip of the arrow is extended ( ⁇ 1-4 glycoside bond part). , Refers to binding (transfer) to a sugar chain receptor.
- the sugar chain receptor in the present disclosure refers to a sugar-containing substance having GlcNAc represented by the general formula (1) at the non-reducing end.
- Transglycosylation activity refers to the ability of Endo M or Endo M mutant to transfer a sugar chain donor to a sugar chain acceptor to generate a new product (transfer to a sugar chain).
- Transglycosylation yield is a product generated after the transglycosylation reaction, and is transferred to the glycan acceptor from the position where the arrow in the above general formula (4) is extended to the glycan acceptor.
- the product may be referred to as a product after sugar chain transfer.
- endo M mutant refers to an endo enzyme mutant in which the amino acid of the amino acid sequence of endo M (see SEQ ID NO: 1) has been deleted, added, or substituted.
- End M means an endoenzyme derived from the hair mold Mucor Himaris (GenBank Accession No. BAB43869)
- End D means an endoenzyme derived from Streptococcus pneumoniae (GenBank Accession No. BAB62042.1). .
- “Homology” refers to residues in a protein amino acid sequence variant that are identical after aligning the sequence by introducing gaps, if necessary, to achieve maximum homology (percent). Defined as a percentage. Methods and computer programs for alignment are well known in the art and use ClustralW2 (http://www.ebi.ac.uk/Tools/msa/clustalw2/) herein. . In addition, as a method of describing amino acid residues, it is expressed by either one of three letters or one letter.
- the 175th asparagine residue is indicated as N175.
- an amino acid residue substituted that is, an amino acid residue substituted with another amino acid
- a residue obtained by substituting the 175th asparagine residue of endo M with a glutamine residue (or alanine) is N175Q ( Alternatively, End M, which is designated N175A) and contains it, may be referred to as the N175Q variant of Endo M, or simply the N175Q variant.
- endo M mutant of the present disclosure a mutant having an amino acid sequence in which the 175th amino acid residue of the amino acid sequence represented by SEQ ID NO: 1 is glutamine or alanine is referred to as “endo M mutant of the present disclosure”. There is.
- the amino acid sequence of 175 of the amino acid sequence represented by SEQ ID NO: 1 has an amino acid sequence of glutamine or alanine, and 175 of the amino acid sequence represented by SEQ ID NO: 1 Amino acids modified within a range of homology of 80% or more to the amino acid sequence of the endo-M mutant by deletion, addition or substitution of one or more amino acid residues other than the amino acid residue
- the endo-M mutant homolog of the present disclosure it may be particularly referred to as “the endo-M mutant homolog of the present disclosure”. is there.
- the transglycosylation activity by the endo M mutant can be confirmed by measuring the Man6P-containing glycoprotein produced in the solution after the reaction, for example, by SDS-PAGE, lectin blotting and MALDI-TOF MS.
- Examples of the MALDI-TOF MS include the following method. That is, a certain amount of acetone was added to the solution after the reaction, the dissolved portion was dried, and then a certain amount of DHBA solution (20 mg / mL 2,5-dihydroxybenzoic acid dissolved in 50% aqueous methanol solution) was added. Dissolve. Thereafter, a part of the dissolved solution is spotted on a plate for MALDI-TOF MS analysis, dried, and measured with an autoflex speed-tko1 reflector system (manufactured by Bruker Daltonics) under the following conditions: The mass of the product after the reaction can be confirmed.
- Measurement mode positive ion mode or negative ion mode and reflector mode or linear mode
- Measurement voltage 1.5Kv ⁇ 2.5Kv
- Measurement molecular weight range 0 to 10,000 (m / z) ⁇ Total number of times: 500-10000
- a lectin blot for example, a lectin against Man6P can be used, and the lectin described in the literature by Akeboshi et al. [APPLIED AND ENVIRONMENTAL MICROBIOLOGY, Vol. 73, 4805-4812, (2007)] Man6PR (sometimes referred to as Dom9-His).
- the product after sugar chain transfer can be identified by, for example, measurement by SDS-PAGE, lectin blot and MALDI-TOF MS as described above, and the sugar obtained by SDS-PAGE and lectin blot From the comparison of the density of the band of the product after chain transfer, it is possible to qualitatively confirm the magnitude of the yield of the product after sugar chain transfer (yield of sugar chain transfer). Moreover, from this result, it is possible to qualitatively confirm the size of the endo-M mutant for the transglycosylation activity.
- the carbohydrate portion is described with reference to the nomenclature usually used for describing oligosaccharides. These nomenclatures are found, for example, in Hubbard et al. [Ann. Rev. Biochem., Vol. 50, 555, (1981)].
- Mannose is represented by Man
- 2-N-acetylglucosamine is represented by GlcNAc
- galactose is Gal
- fucose is Fuc
- glucose Glc
- Sialic acid is represented by the abbreviated notation NeuAc for 5-N-acetylneuraminic acid and NeuGc for 5-glycolylneuraminic acid.
- the N-acetylglucosaminyl group may be referred to as a GlcNAc residue
- the mannosyl group may be referred to as a Man residue.
- the monosaccharide means only the above-mentioned sugars such as Man and GlcNAc itself.
- the position of the carbon that forms the sugar is represented by the 1st position of the reducing end and the 2nd position of the adjacent carbon atom.
- a combination of these two or more monosaccharides is called an oligosaccharide, and its derivative is called an oligosaccharide derivative. That is, the N-linked sugar chain is an oligosaccharide.
- “Glycoside bond” refers to a bond in which the hydroxyl group at the 1-position of a sugar unit in a sugar chain and the hydroxyl group of another sugar are dehydrated and bonded together via an oxygen atom.
- an ⁇ 1-6 glycoside bond The term “sugar” refers to a glycosidic bond in which the 1-position (carbon) of a sugar and the 6-position (oxygen atom at the 6-position) of another sugar are bonded in an ⁇ -type.
- the hydroxyl group at the 1-position of the sugar has ⁇ type and ⁇ type.
- the glucosaminyl group (GlcNAc) on the non-reducing end side of the chitobiosyl site (-GlcNAc ⁇ 1-4GlcNAc-) does not contain the oxygen atom at the 1-position of the GlcNAc, and It indicates that it contains a 4-position oxygen atom.
- GlcNAc that binds to Y on the reducing end side does not include the oxygen atom at the 1-position and includes an oxygen atom at the 4-position.
- the 2nd, 3rd, 4th and 6th carbons that are not glycosidic bonded indicate that a hydroxyl group is bonded.
- a sugar chain including three monosaccharides may be referred to as three sugars, and a five sugar chain including five sugars.
- the “core sugar chain” refers to the sugar chain part represented by C-1 below in the N-linked sugar chain, and “trimannosyl” refers to the three mannose parts in the core sugar chain. . Further, as shown in the broken line portion on the left side of C-1, among the trimannosyl portion, Man binding at ⁇ 1-6 on the non-reducing end side is Man2, and Man binding at ⁇ 1-3 on the non-reducing end side is Man3. And Man on the reducing end side is referred to as Man1.
- oligosaccharide as shown on the left side of the position where the tip of the arrow in the general formula (4) is extended may be referred to as a trimannosyl GlcNAc-containing sugar chain.
- protein means containing a peptide. That is, the glycoprotein includes a glycopeptide.
- glycopeptide refers to a glycoprotein having 50 or less amino acid residues.
- protein includes both glycoproteins and non-glycoproteins unless otherwise specified.
- glycoprotein indicates that at least one or more N-linked sugar chains are present in the peptide or polypeptide portion. Unless otherwise specified, the type of the plurality of N-linked sugar chains is 1 Species and two or more types are included.
- lysosomal enzyme refers to a glycoprotein that functions as an enzyme in lysosomes in eukaryotic cells.
- a stable natural-type substrate can be used in the preparation of a chemo-enzymatic Man6P-containing glycoprotein, and a Man6P-containing glycoprotein that cannot be achieved by the prior art can be produced.
- an N-linked sugar chain derivative having a chitobiosyl skeleton is used as a sugar chain donor in the sugar chain transfer reaction of endo M
- the reactivity in the sugar chain transfer is due to the sugar chain structure on the non-reducing terminal side of endo M. It was known to change significantly.
- an N-linked sugar chain having a large polar residue such as a phosphate group is expected to be unsuitable as a sugar chain donor for a sugar chain transfer reaction by an endo M mutant.
- the endo M mutant recognizes an N-linked sugar chain having Man6P at the non-reducing end, but recognizes it as a sugar chain donor, and the sugar chain acceptor represented by the general formula (1) is water. It is considered that the transglycosylation reaction proceeds by preferentially binding to the glycan donor rather than the glycan donor, and once the product after the transglycosylation is formed, the endo M mutant is re-linked to the saccharide. Since it is difficult to recognize the product after chain transfer, it is presumed that the yield of the product after sugar chain transfer increases as a result.
- the sugar chain donor having no oxazoline skeleton used in the above-mentioned sugar chain transfer reaction has an advantage that it can be synthesized by a method in which the phosphate group is not exposed to acidic conditions, and is stable even in a buffer solution. Can exist.
- the sugar chain donor having a chitobiosyl skeleton in the present disclosure is extremely unlikely to bind to a group other than GlcNAc of the sugar chain acceptor, like an oxazolineized sugar chain donor.
- the sugar chain transfer reaction in the present disclosure is a reaction that facilitates the production of a Man6P-containing glycoprotein that could not be easily produced by conventional oxazolineated sugar chain donors. Application to large-scale preparation can also be considered.
- the method for producing a Man6P-containing glycoprotein of the present disclosure includes a sugar chain having a structure derived from an N-linked sugar chain in which Man6P is bound to a non-reducing end in the presence of the endo M mutant of (a) or (b) below: This is a method for producing a Man6P-containing glycoprotein by producing a Man6P-containing glycoprotein by a sugar chain transfer reaction between a donor and the sugar chain acceptor represented by the general formula (1).
- Man6P-containing glycoproteins produced by the production method of the present disclosure include those found in nature and those synthesized.
- the second amino acid residue from the asparagine is necessarily a threonine residue or a serine residue on the C-terminal side of the asparagine to which the N-linked sugar chain binds in a naturally occurring glycoprotein.
- the glycoprotein of the Man6P-containing glycoprotein produced by the production method of the present disclosure is not particularly limited.
- glycoproteins secreted inside or outside the cell or on the cell surface and having an N-linked sugar chain are exemplified.
- examples include various enzymes, various hormones, cell adhesion factors, various receptors, and the like.
- Examples include various cytokines.
- various enzymes are preferable.
- examples of various enzymes include enzymes that function in cells, and among them, lysosomal enzymes are preferable.
- ⁇ -hexosaminidase As lysosomal enzymes, ⁇ -hexosaminidase ( ⁇ N-acetylglucosaminidase, ⁇ N-acetylgalactosaminidase), ⁇ N-acetylglucosaminidase, ⁇ galactosidase, ⁇ galactosidase, ⁇ glucosidase, ⁇ iduronidase, ⁇ iduronic acid 2 sulfatase, ⁇ glucuronidase, ⁇ -glucocerebrosidase, ⁇ -galactocerebrosidase, ⁇ N-acetylgalactosaminidase, ⁇ fucosidase, cathepsin A, cathepsin B, cathepsin D, cathepsin H, cathepsin L, aspartyl glucosaminidase, endo ⁇ -galactosidase
- the origin of the glycoprotein is not particularly limited as long as it does not impair the effect of the method of the present disclosure. However, from the viewpoint of introducing and functioning in an animal cell, it is preferably derived from an animal cell, and further to humans. From the viewpoint of enhancing the effect of enzyme replacement therapy, it is more preferably derived from a human.
- the endo M variant in the present disclosure is (a) an endo M variant having an amino acid sequence in which the amino acid residue at position 175 of the amino acid sequence represented by SEQ ID NO: 1 is glutamine or alanine, or (b) Modification within 80% or more homology with the amino acid sequence of (a) above by deletion, addition or substitution of one or more amino acid residues other than the 175th amino acid residue of the amino acid sequence And an endo-M mutant (endo-M mutant homologue) having an amino acid sequence and an activity of catalyzing a sugar chain transfer reaction.
- the sugar chain transfer reaction refers to a sugar chain donor having a structure derived from an N-linked sugar chain in which Man6P is bonded to the non-reducing end, and a sugar chain acceptor represented by the above general formula (1): This refers to the transglycosylation reaction.
- the endo M mutant of the present disclosure is a mutant in which a mutation is introduced into the amino acid of endo M.
- the endoenzyme represented by SEQ ID NO: 1 is an endo ⁇ -N acetylglucosaminidase (GenBank Accession No. BAB43869) derived from Mucor Himalis.
- the endo M mutant is N175Q or N175A, so that hydrolysis of the product after the transglycosylation reaction can be sufficiently suppressed. Further, from the viewpoint of sugar chain transfer yield, N175Q mutant is preferable.
- the endo M mutant of the present disclosure can be prepared by a normal genetic engineering technique, and can be prepared using various types of hosts and corresponding appropriate protein expression vectors.
- the host include Escherichia coli, Brevibacillus, cyanobacteria, lactic acid bacteria, yeast, insect cells and animal cells.
- E. coli Escherichia coli
- Brevibacillus cyanobacteria
- lactic acid bacteria yeast
- insect cells insect cells
- animal cells from the viewpoint of ease of preparation and expression level, it is preferable to prepare by a method using E. coli as a host or a method using yeast as a host.
- the specific production method is described in detail in the literature of Umekawa et al. [J. Biol. Chem., Vol.285 (1), 511-521, ⁇ (2010)] for Escherichia coli. This is described in detail in JP-A-11-332568.
- the endo-M mutant and endo-M mutant homologue of the present disclosure are fused endo-M mutants (or fused forms) fused with other peptides or proteins on the C-terminal side or the N-terminal side in a transglycosylation reaction. Endo M mutant homologue).
- the peptide or protein that can be fused is not particularly limited as long as it does not inhibit the transglycosylation reaction.
- hexahistidine peptide (amino acid sequence is from N-terminal to HHHHHH), flag peptide (amino acid sequence is N-terminal) To DYKDDDDK), influenza HA polypeptide (amino acid sequence is YPYDVPDYA from the N-terminus), glutathione-S-transferase, luciferase, avidin, chitin-binding protein, c-myc, thioredoxin, disulfide isomerase (DsbA), maltose-binding protein ( MBP) and green fluorescent protein (GFP).
- hexahistidine peptides and flag peptides are particularly preferable from the viewpoint of ease of preparation of endo M mutant or endo M mutant homolog.
- fusion-type endo M mutant or fusion-type endo M mutant homolog there is a gap between the polypeptide part of the peptide or protein to be fused and the polypeptide part of the endo-M mutant or endo-M mutant homolog.
- the linker region may include an amino acid sequence portion (protease site) that is hydrolyzed by a protease.
- protease site For example, a factor Xa site, a thrombin site, an enterokinase site, a precision protease site is mentioned.
- saccharide hydrolase found in nature has a family of hundreds of glycosylhydrases from the homology of amino acid sequences. (GH family).
- Endo M is a glycosyl hydrolase belonging to the GH family 85, and other proteins belonging to the GH family 85 are known to be widely distributed from humans to bacteria.
- the endo M mutant homologue of the present disclosure may be any amino acid other than the 175th amino acid residue as long as it has 80% homology with the N175Q mutant or N175A mutant and has transglycosylation activity. Those prepared by substituting, deleting or adding residues to other amino acids may be used.
- an endo M mutation caused by deletion, addition, or substitution of one or more amino acid residues other than the 175th amino acid residue of the amino acid sequence of the endo M mutant of the present disclosure
- the homology of the amino acid sequence of the body homologue to the amino acid sequence of the endo M mutant is 80% or more, the transglycosylation activity can be sufficiently maintained.
- it is 90% or more it is preferable from the sugar-transfer yield easily improving, More preferably, it is 95% or more, Especially preferably, it is 98% or more, Most preferably, it is 99% or more.
- the concentration of the endo M mutant or endo M mutant homolog in the reaction solution is preferably 0.1 U / ml to 1 U / ml.
- the concentration is more preferably 0.1 U / ml to 1 U / ml, and particularly preferably 0.2 U / ml to 0.5 U / ml.
- the purity of the endo M mutant or endo M mutant homolog used in the reaction is 50% from the viewpoint of shortening the reaction time and the yield of sugar chain transfer from the results of SDS-polyacrylamide electrophoresis. % Or more, more preferably 70% or more, particularly preferably 80% or more, and most preferably 90% or more. When the degree of purification is 50% or more, the reaction time can be shortened and the sugar chain transfer yield can be easily increased.
- Man6P has a structure derived from an N-linked sugar chain bonded to the non-reducing end, and the sugar chain can be transferred to the sugar chain acceptor represented by the general formula (1). If it is a thing, it will not specifically limit.
- the structure derived from the N-linked sugar chain means that the N-linked sugar chain itself and a derivative of the N-linked sugar chain are included.
- a sugar chain donor represented by the following general formula (2) is preferable.
- X 1 to X 6 each independently represents a hydrogen atom or a saccharide-derived group, and at least one of X 1 to X 6 is derived from a saccharide having Man6P at the non-reducing end. It is a group.
- Z 1 represents a hydrogen atom or GlcNAc, and when Z 1 is GlcNAc, the GlcNAc is bonded to Man linked to GlcNAc at ⁇ 1-4 at ⁇ 1-4.
- Y 2 represents a monovalent substituent.
- X 1 to X 6 may be a hydrogen atom or a saccharide-derived group, and at least one of X 1 to X 6 may be a saccharide-derived group having Man6P at the non-reducing end.
- the saccharide-derived group is not particularly limited as long as it does not impair the effect of the method of the present disclosure.
- the saccharide-derived group having Man6P includes Man6P itself.
- groups other than the saccharide-derived group having Man6P at the non-reducing end include, for example, GlcNAc ⁇ 1-2, Gal ⁇ 1-4GlcNAc ⁇ 1-2, NeuAc ⁇ 2-6Gal ⁇ 1-4GlcNAc ⁇ 1-2, NeuAc ⁇ 2-3Gal ⁇ 1-4GlcNAc ⁇ 1-2, NeuGc ⁇ 2-6Gal ⁇ 1-4GlcNAc ⁇ 1-2 and NeuGc ⁇ 2-3Gal ⁇ 1-4GlcNAc ⁇ 1-2, heterologous antigens: Galc1-4GlcNAc ⁇ 1-2 4GlcNAc ⁇ 1-3] nGal ⁇ 1-4GlcNAc ⁇ 1-2 (n is an arbitrary number), keratan sulfate [Gal ⁇ 1-4GlcNAc (6SO 3) ⁇ 1-3] nGal ⁇ 1-4GlcNAc ⁇ 1-2 (n is an arbitrary number) [Gal (6SO 3) ⁇ 1-4GlcNAc ( 6SO 3) ⁇ 1-3] nGal
- the saccharide-derived groups having Man6P at the non-reducing end include GlcNAc ⁇ 1-2, Gal ⁇ 1-4GlcNAc ⁇ 1-2, [Gal ⁇ 1-4GlcNAc ⁇ 1-3] nGal ⁇ 1-4GlcNAc ⁇ 1-2 (n is Any number), [Gal ⁇ 1-4GlcNAc (6SO 3 ) ⁇ 1-3] nGal ⁇ 1-4GlcNAc ⁇ 1-2 (where n is an arbitrary number), etc. Things.
- a group other than a saccharide-derived group having Man6P at the non-reducing end may have any of a structure derived from a high mannose-type sugar chain in addition to a hydrogen atom.
- Examples of X 1 to X 6 include Man ⁇ 1-6, Man ⁇ 1-3, Man ⁇ 1-2Man ⁇ 1-3, Man ⁇ 1-2Man ⁇ 1-6, Man ⁇ 1-2, Man ⁇ 1-2Man ⁇ 1-2, and the like.
- saccharide-derived groups having Man6P at the non-reducing end include, for example, Man6P ⁇ 1-6, Man6P ⁇ 1-3, Man6P ⁇ 1-2Man ⁇ 1-3, Man6P ⁇ 1-2Man ⁇ 1-6, Man6P ⁇ 1-2 , Man6P ⁇ 1-2Man ⁇ 1-2 and the like.
- Z 1 may be a hydrogen atom or GlcNAc ⁇ 1-4.
- examples of Y 2 include a substituent containing a structure in which an oxygen atom, a nitrogen atom, a carbon atom, or a sulfur atom is directly bonded to the 1-position carbon of GlcNAc.
- Y 2 is not particularly limited as long as it does not reduce the transglycosylation activity.
- alkoxy group, acylamino group, aryloxy group, alkenyl examples thereof include an oxy group, an acyloxy group, an alkylsulfinyl group, an arylsulfinyl group, an alkylsulfonyl group, an arylsulfonyl group, an alkylsulfonyloxy group, and an arylsulfonyloxy group.
- substituents may further have a substituent.
- Y 2 is a hydroxyl group, an alkoxy group having 1 to 30 carbon atoms which may have a substituent, or an alkyl group having 6 to 30 carbon atoms which may have a substituent.
- An aryloxy group, an optionally substituted alkenyloxy group having 1 to 30 carbon atoms and an optionally substituted acylamino group are preferred.
- Y 2 is an alkoxy group optionally having a substituent having 1 to 10 carbon atoms and a substitution having 2 to 10 carbon atoms from the viewpoint of ease of preparation as a sugar chain donor and a yield of sugar chain transfer.
- An alkenyloxy group which may have a group, an aryloxy group which may have a substituent having 6 to 24 carbon atoms, and an acylamino group which may have a substituent are preferable, and more preferably 1 to An alkoxy group optionally having 8 substituents, an alkenyloxy group optionally having 2 to 6 carbon atoms, and an aryloxy optionally having 6 to 12 carbon atoms
- An acylamino group which may have a group and a substituent is particularly preferred. Specific examples include a methoxy group, an ethoxy group, a phenoxy group, a paramethoxyphenoxy group, and a paranitrophenoxy group.
- the acylamino group which may have a substituent is preferably an acylamino group to which the amino group of the side chain of the asparagine of the peptide or protein is bonded, and the solubility in the reaction solution and the transglycosylation yield. From the viewpoint, an acylamino group to which the amino group of the side chain of the asparagine of the peptide is bonded is more preferable.
- substituents which may be substituted include an alkyl group having 1 to 6 carbon atoms, an alkenyl group having 2 to 12 carbon atoms, an aryl group having 6 to 12 carbon atoms, a halogen atom, a cyano group, a nitro group, When it is at least one selected from an amino group, a carboxyl group and a pyridyl group and there are two or more substituents, each substituent may be the same or different.
- the method for preparing the sugar chain donor represented by the general formula (2) is not particularly limited, but for the ease of the preparation method, for example, an N-linked sugar chain is prepared and Man6P is chemically introduced. A method is mentioned.
- N-linked sugar chains include high mannose sugar chains and complex sugar chains.
- the high mannose sugar chains can be prepared from natural products, even by chemical synthesis. There may be.
- As a method by chemical synthesis for example, as described in the literature of Matsuo et al. [Tetrahedron, Vol. 62, 8262-8277, (2006)], As described in Japanese Patent Application Publication No. 2007-297429, a method combining a chemical synthesis method and mannosidase can be mentioned.
- Examples of the method of preparing from natural products include a method of extracting from egg white albumin as described in Japanese Patent No. 3776952 and JP-A-10-251304.
- a synthetic preparation method is preferred from the viewpoint of introducing Man6P into the non-reducing end.
- the method for preparing the complex type sugar chain may be a method by chemical synthesis or a method of preparing from a natural product.
- a method by chemical synthesis for example, it can be prepared by the method described in Wang et al. [J. Am. Chem. Soc., Vol. 134 (29), 12308, (2012)].
- Examples of the method of preparing from a natural product include a method of extracting from egg yolk (for example, International Publication No. 962255 and Japanese Patent Application Laid-Open No. 2011-231293).
- those obtained by synthesis are preferable from the viewpoint of introducing Man6P into the non-reducing end.
- a chemical introduction method can be mentioned.
- any synthetic route may be used as long as Man6P can be introduced into the non-reducing end of the N-linked sugar chain, and it is appropriately selected depending on the structure of the target sugar chain donor.
- the method for introducing a phosphate group into mannose is not particularly limited.
- the phosphate group can be introduced by reacting a phosphorylating reagent with the 6-position hydroxyl group of the mannose residue at the non-reducing end of an N-linked sugar chain. .
- Examples of the phosphorylating reagent include tetrabenzyl pyrophosphate, cyclic phosphite amide, phosphite, phosphate triester, and amidite.
- a phosphate group from the viewpoint of introducing a phosphate group only to the 6-position hydroxyl group of the mannose residue at the non-reducing end of the N-linked sugar chain, other than the hydroxyl group is protected by a protecting group. It is preferable.
- protection means that the hydroxyl group is converted into a group that is not reactive with the phosphorylating reagent.
- a sugar chain donor represented by the following general formula (3) is preferable.
- X 7 , X 8 and X 9 each independently represent a hydrogen atom, Man, Man ⁇ 1-2Man, Man6P, Man6P ⁇ 1-2Man or Man6P ⁇ 1-6Man.
- Man represents a mannosyl group.
- Man6P represents a mannosyl group having a phosphate group bonded to the 6-position.
- X 7 -6 indicates the X 7 bound to 6-position of Man
- X 8 -3 shows the X 8 attached to the 3-position of Man
- X 9 -2 is attached to the 2-position of Man X 9 is shown.
- At least one of X 7 , X 8 and X 9 represents any of Man6P, Man6P ⁇ 1-2Man and Man6P ⁇ 1-6Man.
- GlcNAc represents an N-acetylglucosaminyl group.
- ⁇ 1-6 represents an ⁇ -glycosidic bond between the 1-position of Man and the 6-position of Man or an ⁇ -glycosidic bond between the 1-position of Man6P and the 6-position of Man
- ⁇ 1-3 represents ⁇ 1-2 represents an ⁇ -glycoside bond between the 1-position of Man and the 2-position of Man, or an ⁇ -glycoside bond between the 1-position of Man6P and the 2-position of Man.
- ⁇ 1-4 represents a ⁇ glycosidic bond between position 1 of GlcNAc and position 4 of GlcNAc or ⁇ glycoside bond between position 1 of Man and position 4 of GlcNAc.
- Y 3 represents a monovalent substituent.
- X 7 , X 8 and X 9 when at least one of X 7 , X 8 and X 9 is any one of Man6P, Man6P ⁇ 1-2Man or Man6P ⁇ 1-6Man, the other is a hydrogen atom, Man, Man ⁇ 1-2Man, Man6P, Man6P ⁇ 1 -2Man or Man6P ⁇ 1-6Man.
- the one having Man6P at the non-reducing end may be any of X 7 to X 9 and is appropriately adjusted depending on the type of the target peptide or glycoprotein to be transferred to the sugar chain. It is preferable.
- X 7 is more preferably any of Man6P, Man6P ⁇ 1-2Man, or Man6P ⁇ 1-6Man.
- the number of Man6Ps among X 7 to X 9 is more preferably 2 or more, and particularly preferably 3.
- sugar chain donors represented by the general formula (3) from the viewpoint of being a high mannose type sugar chain already found in natural glycoproteins, the following general formulas (3-1) to (3) Those represented by the general formula (3-10) are preferable, and among them, the general formula 3-3 is more preferable.
- Y 3 represents a monovalent substituent.
- Man6P represents a mannosyl group having a phosphate group bonded to the 6-position.
- Y 3 has the same meaning as Y 2 in the general formula (2), and the preferred range is also the same.
- the compound represented by the general formula (3) can be prepared by appropriately combining, for example, a known chemical glycosylation method and the above-described method for introducing a phosphate group or the like.
- the sugar chain receptor is a GlcNAc-containing glycoprotein represented by the following general formula (1).
- the sugar chain receptor is not particularly limited as long as it is represented by the general formula (1) as long as it does not inhibit the sugar chain transfer activity of the endo M mutant.
- Y 1 represents an acylamino group containing a structure derived from a glycoprotein.
- GlcNAc represents an N-acetylglucosaminyl group.
- Y 1 as the glycoprotein, for example, various enzymes, various hormones, cell adhesion factors, and various receptors and various cytokines and the like. Among them, various enzymes are preferable from the viewpoint of functioning in cells, and lysosomal enzymes are preferable from the viewpoint of functioning in lysosomes using the Man6P receptor on the lysosomal membrane.
- ⁇ -hexosaminidase As lysosomal enzymes, ⁇ -hexosaminidase ( ⁇ N-acetylglucosaminidase, ⁇ N-acetylgalactosaminidase), ⁇ N-acetylglucosaminidase, ⁇ galactosidase, ⁇ galactosidase, ⁇ glucosidase, ⁇ iduronidase, ⁇ iduronic acid 2 sulfatase, ⁇ glucuronidase, ⁇ -glucocerebrosidase, ⁇ -galactocerebrosidase, ⁇ N-acetylgalactosaminidase, ⁇ fucosidase, cathepsin A, cathepsin B, cathepsin D, cathepsin H, cathepsin L, aspartyl glucosaminidase, endo ⁇ -galactosidase
- the origin of the glycoprotein is not particularly limited as long as it does not impair the effect of the method of the present disclosure. However, from the viewpoint of introducing and functioning in an animal cell, it is preferably derived from an animal cell, and further to humans. From the viewpoint of enhancing the effect of enzyme replacement therapy, it is more preferably derived from a human.
- Y 1 is an acylamino group containing a structure derived from a glycoprotein having 30 or less amino acid residues
- Y 1 is an acylamino group containing a structure derived from a glycoprotein having 30 or less amino acid residues
- a chemical method or a chemistry including a peptide extension step as shown in a patent document (Japanese Patent Laid-Open No. 10-45788).
- a glycopeptide or N-linked sugar chain derivative obtained by a reagent manufacturer can be easily prepared by hydrolysis with the endo M mutant of the present disclosure or a commercially available endo enzyme.
- Y 1 is an acylamino group containing a structure derived from a glycoprotein is a glycoprotein obtained by purchasing a commercially available reagent or food, a glycoprotein prepared by a genetic engineering technique, or a food or natural product. Can be easily prepared by hydrolyzing the glycoprotein obtained by a general extraction / separation method with the endo-M mutant of the present disclosure or a commercially available endo-enzyme.
- End H manufactured by New England Biolabs
- End S manufactured by Sigma-Aldrich
- End D manufactured by Cosmo Bio
- End M manufactured by Tokyo Chemical Industry Co., Ltd.
- End F1 ⁇ And F3 manufactured by Sigma-Aldrich
- Non-commercial enzymes include glycosyl hydrolases belonging to GH family 85, or enzymes that belong to GH family 18 and have been shown to hydrolyze N-linked sugar chains. Can do.
- a gene encoding a target glycoprotein is introduced (transgenic) into a host cell or individual to prepare a desired glycoprotein.
- it does not specifically limit as a host.
- yeast, an insect cell the cell derived from various animals, animals, such as an insect which is an individual, an amphibian, and a mammal, are mentioned.
- insect cells or insects that are individuals are more preferred, silkworm cells or silkworms are particularly preferred, and silkworms that are individuals are most preferred.
- silkworms for example, as shown in the literature of Ito et al.
- transgenic silkworms introduced with foreign genes (transgenic silkworms) It is known that foreign proteins are expressed in large quantities in silk glands.
- the expressed sugar chain of the protein contains a human-type-like partial structure, and has an advantage that an insect-specific sugar chain structure is not added.
- glycoproteins prepared from transgenic silkworms for example, as shown in JP-A-2015-208260, N-linked glycoprotein sugar chains are mainly high mannose sugar chains, many endoenzymes are used. This is advantageous for the preparation of sugar chain receptors. For this reason, it can be said that the silkworm which can be raised in large quantities and cheaply is economically advantageous for preparation of said glycoprotein.
- the reaction means a sugar chain transfer reaction, and the sugar chain transfer reaction is performed in a solution in which an endo M mutant, a sugar chain donor, and a sugar chain acceptor are dissolved.
- the solution used for the reaction is not particularly limited as long as it does not inhibit the transglycosylation activity of the endo M mutant.
- phosphate buffer, citrate buffer, carbonate buffer, Tris-HCl buffer, MES Examples include a buffer solution, a MOPS buffer solution, a HEPES buffer solution, a borate buffer solution, and a tartrate buffer solution. These buffers may be used alone or in combination.
- MES buffer solution MOPS buffer solution
- HEPES buffer solution phosphate buffer solution
- phosphate buffer solution is preferable from the viewpoint of glycosyltransferase activity of endo M mutant or endo M mutant homolog.
- concentration of the phosphate buffer is preferably 10 mM to 250 mM, more preferably 20 mM to 150 mM, and particularly preferably 50 mM to 100 mM, from the viewpoint of glycosyl transfer activity of the endo M mutant or endo M mutant homolog. .
- the buffering capacity is increased, and by setting the concentration to 250 mM or less, the sugar chain transfer activity of the endo M mutant or the endo M mutant homolog can be increased and the sugar chain transfer yield can be increased.
- the pH of the solution in the reaction is preferably 5.5 to 8.5, more preferably 6.0 to 8.0, and particularly preferably 6.5 to 7.5 from the viewpoint of increasing the sugar chain transfer yield.
- the temperature in the reaction is 4 from the viewpoint of transglycosylation activity of the endo M mutant or endo M mutant homolog, from the viewpoint of increasing the transglycosylation yield, and from the viewpoint of the stability of the sugar chain receptor, endo M mutant, etc. It is preferably from 40 ° C to 40 ° C, more preferably from 20 ° C to 40 ° C, and particularly preferably from 25 ° C to 35 ° C. By setting the temperature to 4 ° C. or higher, the transglycosylation activity of the endo M mutant can be increased, and by setting the temperature to 40 ° C. or lower, the stability of the endo M mutant or the endo M mutant homolog can be increased. .
- the concentration of the endo M mutant (or endo M mutant homolog) in the reaction solution is preferably 0.005 ⁇ g / ⁇ L to 0.5 ⁇ g / ⁇ L.
- the transglycosylation activity is improved, and when it is 0.5 ⁇ g / ⁇ L or less, the product of the endo-M mutant (or endo-M mutant homolog) after the transglycosylation Rehydrolysis can be suppressed, and the sugar chain transfer yield can be improved.
- the concentration of the sugar chain receptor in the reaction solution is not particularly limited as long as it does not inhibit the sugar chain transfer reaction of the End M mutant or End M mutant homolog. It is adjusted as appropriate in consideration of the sugar chain transfer yield and the solubility of the sugar chain receptor in the reaction solution.
- the sugar chain receptor represented by the general formula (1) contains a protein
- 1.0 mg / mL to 20 mg / mL is preferable. By being in this range, a higher sugar chain transfer yield can be obtained. Of these, 2.0 mg / mL to 10 mg / mL is more preferable.
- the molar ratio of the sugar chain donor to the sugar chain acceptor is the type of sugar chain donor or sugar chain acceptor used. In addition, it can be appropriately set in consideration of recovery of the reaction after the reaction. However, from the viewpoint of transglycosylation yield, the molar ratio is preferably 10 to 3000, more preferably 100 to 2000, particularly preferably 200 to 1500, and further preferably 300 to 1000. Is most preferred. When the molar ratio is 10 or more, a more sufficient transglycosylation yield can be obtained, and when the molar ratio is 3000 or less, a higher transglycosylation yield can be obtained.
- the reaction time in the transglycosylation reaction is appropriately set depending on the reaction temperature and the concentration of the endo M mutant or endo M mutant homolog in the solution, but is preferably 3 hours to 100 hours. By being 3 hours or longer, the sugar chain transfer yield can be further increased. By being 100 hours or shorter, the influence of hydrolysis of the product after the sugar chain transfer is further suppressed, and the sugar chain transfer yield is increased. be able to. Among the above reaction times, it is more preferably 10 hours to 50 hours.
- the sugar chain donor and the endo M mutant or the endo M mutant homolog are added to the reaction solution again. It is preferable to react for a certain time.
- Man6P containing glycoprotein manufactured with said manufacturing method can be provided as a pharmaceutical composition and the chemical
- the pharmaceutical composition includes a pharmaceutically acceptable stabilizer, buffer, excipient, binder, disintegrant, flavoring agent, A coloring agent, a fragrance
- a protein having a high proportion of many types of Man6P-containing sugar chains can be obtained using many types of sugar chain donors. They are also useful as glycoprotein preparations.
- the method for detecting the intracellular distribution of the fluorescent group-bound Man6P-containing glycoprotein of the present disclosure includes a fluorescent group-bound Man6P-containing product obtained by further introducing a fluorescent group into the Man6P-containing glycoprotein obtained by the above production method. This is a method for detecting the intracellular distribution of a glycoprotein by applying the glycoprotein to a cell.
- the fluorescent group binding type means that the fluorescent group is bound to the Man6P-containing glycoprotein by a covalent bond or an ionic bond.
- the distribution of the Man6P-containing glycoprotein in the cells is distributed. Can be detected.
- fluorescent group-bound Man6P-containing glycoproteins and the like easily enter the cell by the Man6P receptor, and thus the behavior of the fluorescent Man6P glycoprotein and the like after the entry in the cell is traced by the fluorescence emitted by the fluorescent group. can do.
- the detection method for example, by adding the fluorescent Man6P glycoprotein to a target cell or tissue, incubating at a constant atmosphere, temperature, and time, and observing with a fluorescence microscope or the like, The localization and distribution of the fluorescent Man6P glycoprotein in the cell can be known.
- the amount, temperature, and time of addition of the fluorescent Man6P glycoprotein to the cells are appropriately adjusted depending on the type of cells, the function and properties of the protein in the fluorescent Man6P glycoprotein, and the like.
- IDUA human ⁇ -iduronidase
- IDUA refers to an enzyme having activity as an enzyme.
- GlcNAc-IDUA is a protein obtained by hydrolyzing an IDUA N-linked sugar chain with an endoenzyme, and at least one GlcNAc is bound to a polypeptide constituting IDUA. It shows that.
- Man6P-IDUA refers to a Man6P-containing glycoprotein, and unless otherwise specified, is obtained from a transgenic silkworm (also referred to as TG silkworm) described later and then purified by three-stage chromatography. Further, human IDUA usually has 6 N-linked sugar chains, and IDUA prepared by TG silkworm hereinafter is a mixture of 1 to 6 N-linked sugar chains.
- Detection of protein in the gel after SDS-PAGE was performed by shaking in CBB staining solution [10% CBB-R350 (manufactured by GE), 27% methanol, 9% acetic acid, 0.1% CuSO 4 ] for 1 hour or longer. And then decoloring for 6 hours or more using a decolorizing solution (10% acetic acid).
- 1 st IDUA antibodies as probes (Anti-IDUA, sheep IgG polyclonal antibody, (R & D Co., Ltd.)) was used to shake the PVDF membrane in 50% Blocking one / TBS (4 °C, O / N). Thereafter, the PVDF membrane was washed 5 times at room temperature in 0.1% Tween20 / TBS (TBST) 5 times, and further washed in TBS at room temperature for 5 minutes.
- Use as 2 nd probe anti-sheep antibodies (Biotin-conjugated anti-sheep antibody (Vector Co.)), in 50% Blocking one / TBS, was shaken for 1 hour PVDF membranes at room temperature.
- the PVDF membrane in TBST was repeatedly washed 5 times at room temperature for 5 minutes, and further washed in TBS at room temperature for 5 minutes. 3 rd using HRP-conjugated anti-biotin antibody as a probe (Cell Signaling Inc.), the PVDF membrane was shaken for 1 hour at room temperature in 50% Blocking one / TBS. Thereafter, the PVDF membrane in TBST was washed 5 times at room temperature for 5 minutes, and further washed in TBS at room temperature for 5 minutes.
- the PVDF film was subjected to chemiluminescence using Western lighting Chemiluminescence Reagent Ultra / Plus (manufactured by Perkin Elmer), and signal (band) detection was performed using an image analyzer [LAS-4000miniEPUV (manufactured by Fuji Film)].
- IDUA activity is sodium citrate buffer using 4-methylumbelliferyl (4MU) - ⁇ -L-idpyranoside (manufactured by Toronto Research Chemicals) (hereinafter sometimes referred to as 4MU-Ido) as a substrate.
- 4MU-Ido 4-methylumbelliferyl
- the enzyme activity using 4MU-Ido as a substrate may be referred to as IDUA activity.
- the middle silk gland from silkworms was collected by a known general method. Break the outer skin (by hand with surgical gloves) of the TG silkworm from the 5th to the mature stage, and pull out the silk gland that is pushed out from the cracked part. It was. In physiological saline, the front and rear silk glands and the attached fat body tissue were removed from the whole collected silk gland tissue, and only the middle silk gland was fractionated and collected. After washing twice with physiological saline, 50 heads were packed in a conical tube and stored frozen at ⁇ 20 to ⁇ 30 ° C.
- IDUA purification from middle silk gland> IDUA was obtained by purifying by performing the three-stage chromatography operation (affinity chromatography, cation exchange chromatography, hydrophobic interaction chromatography) in combination with the above IDUA activity measurement method.
- the middle silk gland collected from the above TG silkworm was thawed and then shredded and sonicated in 20 mM sodium acetate buffer (pH 4.5) containing 0.5 M NaCl to prepare an extract A. did.
- FIG. 1 shows the results of measuring the enzyme activity (IDUA activity) against 4MU-Ido for the obtained extract A.
- the term “not introduced” means that the IDUA activity was measured for the extract B of the middle silk gland obtained from the silkworm not introduced with the IDUA gene in the same manner as described above. From the results, the extract A obtained from the TG silkworm into which the IDUA gene was introduced showed significantly higher IDUA activity than the extract B. Further, when Western blotting using human IDUA antibody was performed on the obtained extract, as shown in lanes 1 and 2 in FIG. 2, a protein-containing solution extracted from silkworms into which no IDUA gene had been introduced, While no band was obtained, as shown in lanes 3 and 4, a strong band was found around 80 kDa in the protein-containing solution extracted from the TG silkworm into which the IDUA gene was introduced. From the above results, an extract A containing IDUA having IDUA activity was obtained from a TG silkworm into which a human IDUA gene was introduced.
- the extract A obtained above was centrifuged by centrifugation [20,000 ⁇ g, 4 ° C., 20 minutes (Beckman Coulter, Avanti J-E, rotor: JRA 16.250)], and the supernatant was collected.
- CaCl 2 and MnCl 2 were added to the resulting supernatant to a final concentration of 1 mM, and a binding buffer [20 mM sodium acetate buffer, 500 mM NaCl, 1 mM CaCl 2 and 1 mM MnCl 2 (pH 4) was previously added.
- elution was performed with an elution buffer [20 mM sodium acetate buffer, 500 mM NaCl, 1 mM CaCl 2 , 1 mM MnCl 2 , and 0.5 M methyl- ⁇ -D-mannopyranoside (pH 4.5)].
- the obtained elution fraction (hereinafter referred to as ConA elution fraction) was concentrated using an Amicon ultra filter unit (fractional molecular weight 30 kDa) (Millipore), and then a cation exchange column binding buffer [10 mM acetic acid. Buffer exchange was performed using sodium buffer, 150 mM NaCl (pH 6.0)].
- SP elution fraction The elution fraction showing IDUA activity (hereinafter referred to as SP elution fraction) was concentrated using an Amicon ultra filter unit (fractional molecular weight 30 kDa) (Millipore), and then a butyl-Sepharose column binding buffer [50 mM acetic acid.
- Buffer exchange with sodium, 1M ammonium sulfate, 150 mM NaCl (pH 4.5)] was performed. Thereafter, the mixture was filtered through a 0.22 ⁇ m filter, and applied to a hydrophobic interaction chromatography column (HiTrap Butyl FF, manufactured by GE) equipped with AKTA purifier UPC-10, and the ammonium sulfate concentration was changed from 1M to 0M.
- a purification operation was performed under the condition of continuously decreasing, the IDUA activity of each fractionated fraction was measured, and fractions having activity (hereinafter referred to as Butyl-eluting fractions) were collected. Table 1 shows the results of the specific activities of the fractions obtained by the respective purification methods. Further, FIG.
- Extract indicates the extract A
- ConA elute indicates the Con A elution fraction
- SP elute indicates the SP elution fraction
- Butyl elate indicates the Butyl. Represents the eluted fraction.
- SDS-PAGE was performed by applying the same amount (10 ⁇ g) of protein to each lane.
- the specific activity for 4MU-Ido of the Butyl elution fraction obtained by the above purification method is improved to 30 times or more the specific activity of Extract A (Extract) for 4MU-Ido.
- Highly purified IDUA could be obtained from TG silkworm.
- SDS-PAGE in FIG. 3 the staining concentration of bands other than the band around 80 kDa of the SP elution fraction and the Butyl elution fraction is significantly thinner than that of the extract, It was shown that IDUA present in the obtained SP elution fraction and Butyl elution fraction was highly purified.
- IDUA present in the Butyl elution fraction is referred to as TG-IDUA.
- the fraction containing IDUA obtained by the above purification operation was used for the preparation of the sugar chain receptor shown below.
- the sugar chain receptor is obtained by converting the TG-IDUA obtained above into an N-linked sugar chain having a mannose residue number of 5 or less using an endoenzyme (Endo-D, New England Biolabs).
- Endo-D an endoenzyme
- the sugar chain receptor is obtained by converting the TG-IDUA obtained above into an N-linked sugar chain having a mannose residue number of 5 or less using an endoenzyme (Endo-D, New England Biolabs).
- Endo-D Endo-D, New England Biolabs
- MALDI-TOF MS A solution obtained by dissolving the reaction solution as it is or after purification in a solvent (for example, methylene chloride, ethyl acetate, methanol, toluene, acetone, water) and a DHBA solution (20 mg / ml 2,5-dihydroxybenzoic acid in 50% methanol) 0.5 to 2 ⁇ L each of the solution dissolved in an aqueous solution was spotted on a plate for MALDI-TOF MS analysis and dried, and MALDI-TOF MS analysis was performed using an autoflex speed-tko1 reflector system (manufactured by Bruker Daltonics). ) And the following measurement conditions were used.
- a solvent for example, methylene chloride, ethyl acetate, methanol, toluene, acetone, water
- a DHBA solution 20 mg / ml 2,5-dihydroxybenzoic acid in 50% methanol
- Measurement mode positive ion mode or negative ion mode and reflector mode or linear mode
- Measurement voltage 1.5Kv ⁇ 2.5Kv
- Measurement molecular weight range 0 to 10,000 (m / z) ⁇ Total number of times: 500-10000
- a specific method for preparing a sugar chain donor will be described.
- a hexasaccharide derivative (Compound 6) was synthesized by a known glycosylation method. Thereafter, the paramethoxyphenyl group at the reducing end is converted to a fluoro group which is a leaving group, and glycosylation is performed under the glycosylation conditions described below to synthesize compound 9 which is a heptasaccharide derivative, Deprotection of two protecting groups at the position where an acid group is introduced, followed by introduction of a phosphate group, followed by deprotection of the protecting group, a parasaccharide phenyl derivative of 7 sugars having Man6P at the terminal (sugar chain Donor) was obtained.
- reaction solution was cooled to ⁇ 30 ° C., 1.6 g (7.0 mmol) of N-iodosuccinimide and 61 ⁇ L (0.7 mmol) of trifluoromethanesulfonic acid were added, and the mixture was stirred for 3 hours. After confirming the reaction by TLC, 210 ⁇ L (1.5 mmol) of triethylamine was added to the reaction solution to stop the reaction, followed by filtration through Celite. The reaction mixture was diluted with ethyl acetate, washed successively with saturated aqueous sodium thiosulfate solution, saturated aqueous sodium hydrogen carbonate solution and saturated brine, dried over magnesium sulfate, filtered and concentrated under reduced pressure.
- the solution was filtered through celite, diluted with ethyl acetate, washed successively with saturated aqueous sodium hydrogen carbonate solution and saturated brine, dried over magnesium sulfate, filtered, and concentrated under reduced pressure.
- the product was subjected to NMR measurement, and the following results were obtained.
- 0.81 g (0.31 mmol) of the obtained 1-OH compound was dissolved in 8.1 mL of methylene chloride and then cooled to ⁇ 20 ° C., and 0.16 mL (1.2 mmol) of (diethylamino) sulfur trifluoride was added. Stir for 5 hours.
- 0.34 g (0.12 mmol) of the obtained compound was dissolved in a mixed solvent of 1.7 mL of tetrahydrofuran and 1.7 mL of methanol, sodium methoxide (2.5 ⁇ L, 0.012 mmol) was added, and the mixture was stirred for 2.5 hours. . Thereafter, sodium methoxide (5.0 ⁇ L, 0.023 mmol) was further added and stirred for 17 hours.
- a lectin (CI-Man6PR) for detecting Man6P was prepared as follows. That is, a plasmid vector for expression of CI-Man6PR (Dom9-His) prepared by referring to Akeboshi et al. [APPLIED AND ENVIRONMENTAL MICROBIOLOGY, Vol. 73, 4805-4812, (2007)] was used as a methanol-assimilating yeast strain. (Pichia pastoris GS11) was introduced into 5 strains by electroporation.
- the whole culture solution containing yeast after culturing is centrifuged (6000 rpm, 4 ° C., 1 h, KUBOTA 6800 (rotor: RA-1500)), and the culture supernatant is recovered. did.
- the culture supernatant was filtered using a 0.22 ⁇ m filter (Millipore) and concentrated by ultrafiltration [Labscale TFF System, Pellicon XL5K (Millipore)].
- the concentrated culture supernatant was bound to a His tag purification resin (Ni-Sepharose 6 Fast Flow, manufactured by GE), and then washed with a washing buffer [20 mM sodium phosphate according to the protocol attached to the resin. , 0.5 M NaCl (pH 8.0)] and elution with elution buffer [20 mM sodium phosphate, 0.5 M NaCl, 50 mM imidazole (pH 8.0)] A solution containing CI-Man6PR (Dom9-His) was obtained. The obtained solution was appropriately diluted and used for detection of Man6P in the product in the solution after the transglycosylation reaction described later.
- the PVDF membrane was shaken for 1 hour at room temperature in 50% Blocking one / TBS. Thereafter, the PVDF membrane in TBST was washed 5 times at room temperature for 5 minutes, and further washed in TBS at room temperature for 5 minutes.
- the PVDF film was chemiluminescent using Western lightning Chemiluminescence Reagent Ultra / Plus (manufactured by Perkin Elmer), and using an image analyzer [LAS-4000miniEPUV (manufactured by Fujifilm)] with high exposure conditions with an exposure time of 1 second. The signal (band) was detected at.
- FIG. 7A shows the result of SDS-PAGE for the solution after the above reaction
- FIG. 7B shows the result of lectin blotting.
- lane 1 is a sample containing IDUA (hereinafter referred to as CHO-IDUA) obtained by culturing after culturing CHO cells (K1 strain, RIKEN Cell Bank) into which IDUA gene has been introduced.
- Lane 2 is the result of the sample containing TG-IDUA
- Lane 3 is the result of the sample after treating TG-IDUA with End D
- Lane 4 is the result after the transglycosylation reaction. It is a result. From FIG.
- Man6P-IDUA ⁇ Preparation of Man6P-containing glycoprotein (Man6P-IDUA)>
- the solution after the transglycosylation reaction of Example 1 was subjected to ultrafiltration using an Amicon ultra filter unit (fractionated molecular weight of 30 kDa) (manufactured by Millipore) to remove unreacted sugar chain donors. Further, the buffer solution was replaced with a phosphate physiological buffer solution (hereinafter referred to as PBS) to obtain Man6P-IDUA (10.5 ⁇ g).
- PBS phosphate physiological buffer solution
- the obtained Man6P-IDUA was used for examining the supplementary effect on the following mucopolysaccharidosis type 1 patient fibroblasts (MPS1 patient-derived fibroblasts) and the like.
- TG-IDUA, GlcNAc-IDUA, and Man6P-IDUA may be collectively referred to as supplementary enzymes.
- MPS1 patient-derived fibroblasts are present in Ham's F-10 medium (+ 10% Fetal bovine serum) from oral mucosal tissues after approval by Tokushima University Hospital Small Ethics Committee and informed consent to patients It was obtained by inducing under and establishing by adhesion culture and subculture.
- Each enzyme source was prepared as follows. To each 1 mL of culture solution having 5 ⁇ 10 4 cells / well of MPS1 patient-derived cells or healthy human fibroblasts (HS68) cultured in a collagen-coated 24-well plate (Collagen-coated microplate 24well, manufactured by IWAKI) TG-IDUA (endoenzyme-free IDUA) was added at 2000 nmol / h (2.5 ⁇ g), GlcNAc-IDUA was added at 2000 nmol / h (2.85 ⁇ g), and Man6P-IDUA 2000 nmol / h (4.6 ⁇ g) was added. The cells were cultured for 24 hours at 37 ° C. in a 5% CO 2 atmosphere.
- the cells were washed twice with 1 mL of PBS, and further 0.2 mL of 0.5% trypsin EDTA solution was added, and the cells were detached from the wells by incubation for 5 minutes. After adding 1 mL of the culture solution to stop the trypsin reaction, the cells were collected by centrifugation at 4 ° C. and 200 ⁇ g for 5 minutes.
- the enzyme source obtained by the above preparation method is referred to as MPS-E, and the healthy human fibroblasts (HS68)
- the enzyme source obtained by the above preparation method after incubation without adding any of the supplemental enzymes is referred to as NF-E.
- the enzyme source obtained by the above preparation method was referred to as MPS-CE, and GlcNAc-IDUA was added to MPS1 patient-derived fibroblasts.
- the enzyme source obtained by the above preparation method is called MPS-GI-E, and Man6P-IDUA is added to MPS1 patient-derived fibroblasts and incubated, and then the above preparation method
- MPS-MI-E The enzyme source obtained by this is referred to as MPS-MI-E.
- IDUA activity 4-methylumbelliferyl (4MU) - ⁇ -L-idpyranoside (manufactured by Toronto Research Chemicals) (hereinafter sometimes referred to as 4MU-Ido) as a substrate in a sodium citrate buffer (pH 4. After incubating at 37 ° C. for 30 minutes in 5), the fluorescence intensity (Ex: 355 nm, Em: 460 nm) of the released 4MU was measured as an index.
- the enzyme activity using 4MU-Ido as a substrate may be referred to as IDUA activity.
- MUG degradation activity which is ⁇ -hexosaminidase activity
- MUG degradation activity is sodium citrate buffer (pH 4) using 4-methylumbelliferyl (4MU) -N-acetyl- ⁇ -D-glucosaminide (manufactured by SIGMA) as a substrate. .5) was incubated at 37 ° C. for 30 minutes, and the fluorescence intensity (Ex: 355 nm, Em: 460 nm) of the released 4MU was measured as an index.
- FIG. 8A shows the activity recovery ratio in intracellular IDUA activity after adding each of the above-mentioned supplementary enzymes to MPS1 patient-derived fibroblasts and incubating for 24 hours
- FIG. 8B shows the MUG of each of the above enzyme sources. Degradation activity is shown. Further, the numbers on each column in FIG. 8A indicate the value of the activity recovery ratio, and the numbers on each column in FIG. 8B indicate the value of the MUG decomposition activity. From the results, the activity recovery ratio of other enzyme sources other than MPS-E showed a very high value of 23 to 52 times that of MPS-E. In addition, the activity recovery ratio of MPS-MI-E was shown to be the highest in the activity recovery ratio compared to the activity recovery ratios of other enzyme sources.
- a fluorescent group-modified Man6P-containing compound Man6P-Man5-AFO
- a fluorescent group-modified Man6P-IDUA were prepared and used to produce the Man6P-containing compound and Man6P-IDUA in cells. The localization and distribution of was investigated.
- a compound having an n-propoxy group having an amino group at the 3-position as shown in FIG. 9 by using a method similar to the method of synthesizing compound 12 using -2-deoxy-2-phthalimido- ⁇ -D-glucopyranoside AF was synthesized. Further, compound AF1 was prepared by binding with a fluorescent molecule (AcidiFluor ORANGE-NHS, manufactured by Goryuka Kagaku Co., Ltd.) that is activated at acidic pH.
- FIG. 10 shows MALDI-TOF MS spectra of the obtained compound AF1 and compound AF.
- Man6P addition compound Man6P-Man5-AFO modified with a fluorescent group was obtained from the compound AF which is a raw material compound.
- AFO means a fluorescent compound (AcidiFluor ORANGE).
- Man6P-Man5-AFO which is a derivative of 6-saccharide oligosaccharide, was taken into cells through binding to the Man6P receptor on the surface of MPS1 patient-derived fibroblasts.
- Man6P-Man5-AFO was added to a culture solution containing MPS1 patient-derived fibroblasts cultured in a 35 mm collagen-coated dish (35 mm / Collagen-coated dish, manufactured by IWAKI) at a final concentration of 50 nM, and 24 hours later, The fluorescence of Man6P-Man5-AFO was observed with a fluorescence microscope (BZ-9000, manufactured by KEYENCE). The results are shown in FIG.
- Man6P-Man5-AFO was taken up into the cell name, and fluorescence was observed.
- M6P mannose 6-phosphate
- Man6P-Man5-AFO is mediated by M6P receptor on the cell surface. It has been shown to be taken up and transported to late endosomes / lysosomes, intracellular organelles with acidic pH. Thus, Man6P-Man5-AFO has been shown to function as a tag for delivery of molecules bound to Man6P to intracellular organelles (lysosomes).
- DMSO DMSO amount: 6 ⁇ L
- Man6P-IDUA Fluorescent group-modified (fluorescent group binding type) Man6P-IDUA (ultrafluorescent group binding type) by ultrafiltration with an Amicon ultra filter unit (fractional molecular weight 30 kDa) (manufactured by Millipore) and removing unreacted AcidiFluor ORANGE-NHS Man6P-IDUA-AFO) was obtained.
- the obtained Man6P-IDUA-AFO was stored at ⁇ 80 ° C. until just before use.
- Man6P-IDUA-AFO ⁇ Detection of intracellular distribution of Man6P-IDUA-AFO> The Man6P-IDUA-AFO obtained above was added to a culture solution of MPS1 patient-derived fibroblasts cultured on an 8-well chamber slide (manufactured by Nunc) at a final concentration of 70 nM. After a period of time, the cells were washed with PBS, and then the fluorescence of AFO was observed with a fluorescence microscope (BZ-9000, manufactured by KEYENCE). The results are shown in FIG. From the results, it was taken up into cells and fluorescence was observed in acidic pH organelles (late endosome / lysosome).
- Man6P-IDUA-AFO was taken up through the M6P receptor on the surface of fibroblasts and was shown to be transported to the late endosome / lysosome, which is an acidic organelle.
- ConA Sepharose 4B (manufactured by GE Healthcare) was performed on the culture supernatant of the CHO / CTSA strain to purify the CTSA protein. Specifically, the culture supernatant was collected, and CaCl 2 and MnCl 2 were added to the culture supernatant so that the final concentration was 1 mM.
- ConA Sepharose was equilibrated with a binding buffer [20 mM MES buffer, 150 mM NaCl, 1 mM CaCl 2 , 1 mM MnCl 2 (pH 5.5)], and the culture supernatant was applied thereto for adsorption to Con A. It was.
- ConA Sepharose was washed with ConA washing buffer [20 mM MES buffer, 500 mM NaCl, 1 mM CaCl 2 , 1 mM MnCl 2 (pH 5.5)]. Thereafter, elution was performed with an elution buffer [20 mM MES buffer, 150 mM NaCl, 1 mM CaCl 2 , 1 mM MnCl 2, 0.5 M methyl- ⁇ -D-mannopyranoside (pH 5.5)].
- the ConA column elution fraction obtained was concentrated with an Amicon ultrafilter unit 30 kDa cut, and the buffer solution was exchanged with a buffer solution for butyl binding [50 mM MES, 1 M ammonium sulfate (pH 5.5)]. Thereafter, filtration was performed with a 0.22 ⁇ m filter, and purification was performed by hydrophobic interaction chromatography using a HiTrap Butyl HP column (GE Healthcare). For purification, AKTA purifier UPC-10 was used, and elution was performed while the ammonium sulfate concentration was continuously changed from 1 M to 0 M after adsorption of CTSA protein to the column.
- Butyl column elution fraction was buffered in Phos-tag (registered trademark) binding buffer [100 mM Tris-acetate buffer solution (pH 7.5)] using an Amicon ultra filter unit 30 kDa cut (manufactured by Millipore). The liquid was changed.
- Phos-tag registered trademark
- Phos-tag registered trademark
- agarose manufactured by Wako
- Phos-tag® agarose was equilibrated with a binding buffer [100 mM [Tris-acetate buffer (pH 7.5)], and a sample was applied thereto for adsorption to Phos-tag TM agarose. It was. After removing the flow-through fraction, the Phos-tag (registered trademark) agarose was washed with Phos-tag (registered trademark) washing buffer [100 mM Tris-acetate buffer solution (pH 7.5)]. Thereafter, CTSA was eluted with an elution buffer [20 mM sodium phosphate buffer (pH 6.5)].
- reaction solution was replaced with 20 mM Tris-HCl buffer (pH 7.0) and 200 mM NaCl using an Amicon ultra filter unit 10 kDa cut (manufactured by Millipore). Thereafter, filtration with a 0.22 ⁇ m filter (Millipore) was performed, and the reaction solution was added to 1 mL of an MBP trap column equilibrated with 20 mM Tris-HCl buffer (pH 7.0) and 200 mM NaCl. Hf was adsorbed on the MBP trap column.
- the flow-through fraction was collected, and 20 mM Tris-HCl buffer (pH 7.0) and 2OmL of 200 mM NaCl were added to the MBP trap column to wash out the flow-through fraction.
- the flow-through fraction was concentrated using an Amicon ultra filter unit 10 kDa cut (manufactured by Millipore), and the buffer solution was exchanged with 50 mM MES buffer solution (pH 6.0).
- the obtained fraction was designated as GlcNAc-CTSA (sugar chain receptor).
- Glycosyltransferase Glycosynthase (Endo-M-N175Q) 2 mU -Molar ratio (D / A) of sugar chain donor (D) to sugar chain acceptor (A): 1000 -Reaction temperature: 30 ° C ⁇ Reaction time: 24h
- FIG. 13A For the solution after the above reaction, the result of CBB staining of the gel after SDS-PAGE is shown in FIG. 13A, and the lectin (Dom9-His (Akeboshi H. et al., Glycobiology (2009) 19 (9)): FIG. 13B shows the result of lectin blotting using 1002-1009))) as the primary probe.
- lane 1 is the result of the sample containing CTSA
- lane 2 is the result of the sample containing GlcNAc-CTSA
- lane 3 is the result after the transglycosylation reaction.
- FIG. 13A shows that GlcNAc-CTSA is obtained from CTSA by the above Endo-Hf treatment because the band of lane 2 appears on the lower molecular weight side than the band of lane 1. It was.
- the lane 3 band appeared on the higher molecular weight side than the lane 2 band. Therefore, it was shown that a glycosyltransferase reaction occurred in GlcNAc-CTSA, which is a sugar chain receptor, and a post-glycosylation product in which one or two terminal Man6P-containing N-type sugar chains were linked was obtained.
- FIG. 13B since the luminescence signal disappeared in lane 2 of FIG. 13B is newly obtained in lane 3, the sugar chain containing Man6P in the sugar chain donor is added to the sugar chain acceptor. Showed metastasis.
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Genetics & Genomics (AREA)
- Molecular Biology (AREA)
- General Health & Medical Sciences (AREA)
- Biochemistry (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Biotechnology (AREA)
- Microbiology (AREA)
- General Engineering & Computer Science (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Biomedical Technology (AREA)
- Hematology (AREA)
- Urology & Nephrology (AREA)
- Food Science & Technology (AREA)
- Physics & Mathematics (AREA)
- Analytical Chemistry (AREA)
- General Physics & Mathematics (AREA)
- Immunology (AREA)
- Pathology (AREA)
- Preparation Of Compounds By Using Micro-Organisms (AREA)
- Enzymes And Modification Thereof (AREA)
- Measuring Or Testing Involving Enzymes Or Micro-Organisms (AREA)
Abstract
L'invention concerne un procédé de production de glycoprotéine contenant un groupe mannose-6-phosphate (Man6P) pour produire une glycoprotéine contenant du Man6P par une réaction de transglycosylation entre un donneur de chaîne de sucre qui présente une structure dérivée d'une chaîne de sucre se liant à N présentant un Man6P lié à une extrémité non réductrice correspondante et un accepteur de chaîne de sucre représenté par la formule générale (1) en présence d'un mutant Endo-M (a) : un mutant Endo-M présentant une séquence d'acides aminés représentée par la séquence SEQ ID NO: 1 dans laquelle le 175ème résidu d'acide aminé est remplacé par un résidu de glutamine ou d'alanine. Dans la formule générale (1), Y1 représente un groupe acylamino contenant une structure dérivée de glycoprotéine.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2016069437 | 2016-03-30 | ||
| JP2016-069437 | 2016-03-30 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2017170896A1 true WO2017170896A1 (fr) | 2017-10-05 |
Family
ID=59964713
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2017/013322 Ceased WO2017170896A1 (fr) | 2016-03-30 | 2017-03-30 | Procédé de production de glycoprotéine contenant un groupe mannose-6-phosphate et procédé de détection de la distribution intracellulaire de glycoprotéine contenant un groupe mannose-6-phosphate capable de se lier à un groupe fluorescent |
Country Status (2)
| Country | Link |
|---|---|
| JP (1) | JP6996676B2 (fr) |
| WO (1) | WO2017170896A1 (fr) |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2005538941A (ja) * | 2002-04-25 | 2005-12-22 | トランスカリヨティック セラピーズ インコーポレーテッド | α−ガラクトシダーゼA欠損症の治療 |
| JP2012512313A (ja) * | 2008-12-16 | 2012-05-31 | ジェンザイム・コーポレーション | オリゴ糖−タンパク複合体 |
| WO2012147933A1 (fr) * | 2011-04-28 | 2012-11-01 | 国立大学法人大阪大学 | Composition pharmaceutique destinée au traitement de la maladie du stockage lysosomial |
| WO2012157136A1 (fr) * | 2011-05-19 | 2012-11-22 | 財団法人東京都医学総合研究所 | Protéine recombinée de saposine b humaine contenant un cycle glucose phosphorylé et son utilisation |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP4742191B2 (ja) * | 2001-06-14 | 2011-08-10 | 独立行政法人産業技術総合研究所 | 糖蛋白質およびその製造方法 |
-
2017
- 2017-03-30 JP JP2017068970A patent/JP6996676B2/ja active Active
- 2017-03-30 WO PCT/JP2017/013322 patent/WO2017170896A1/fr not_active Ceased
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2005538941A (ja) * | 2002-04-25 | 2005-12-22 | トランスカリヨティック セラピーズ インコーポレーテッド | α−ガラクトシダーゼA欠損症の治療 |
| JP2012512313A (ja) * | 2008-12-16 | 2012-05-31 | ジェンザイム・コーポレーション | オリゴ糖−タンパク複合体 |
| WO2012147933A1 (fr) * | 2011-04-28 | 2012-11-01 | 国立大学法人大阪大学 | Composition pharmaceutique destinée au traitement de la maladie du stockage lysosomial |
| WO2012157136A1 (fr) * | 2011-05-19 | 2012-11-22 | 財団法人東京都医学総合研究所 | Protéine recombinée de saposine b humaine contenant un cycle glucose phosphorylé et son utilisation |
Non-Patent Citations (2)
| Title |
|---|
| FUJITA K. ET AL.: "Molecular cloning of Mucor hiemalis endo-beta-N-acetylglucosaminidase and some properties of the recombinant enzyme", ARCH. BIOCHEM. BIOPHYS., vol. 432, no. 1, 2004, pages 41 - 49, XP004616696, ISSN: 0003-9861 * |
| UMEKAWA M. ET AL.: "Efficient Glycosynthase Mutant Derived from Mucor hiemalis Endo-beta-N- acetylglucosaminidase Capable of Transferring Oligosaccharide from Both Sugar Oxazoline and Natural N-Glycan", J. BIOL. CHEM., vol. 285, no. 1, 2010, pages 511 - 521, XP055429703, ISSN: 0021-9258 * |
Also Published As
| Publication number | Publication date |
|---|---|
| JP6996676B2 (ja) | 2022-02-04 |
| JP2017184736A (ja) | 2017-10-12 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP2949665B1 (fr) | Peptide natriurétique auriculaire glycosylé | |
| Mandal et al. | Functions and biosynthesis of O-acetylated sialic acids | |
| JP2011184445A (ja) | 植物培養における高マンノースタンパク質の製造 | |
| Sugiura et al. | Chondroitinase from baculovirus Bombyx mori nucleopolyhedrovirus and chondroitin sulfate from silkworm Bombyx mori | |
| Geissner et al. | 7-Fluorosialyl glycosides are hydrolysis resistant but readily assembled by sialyltransferases providing easy access to more metabolically stable glycoproteins | |
| Fujita et al. | Glycoside hydrolase family 89 α-N-acetylglucosaminidase from Clostridium perfringens specifically acts on GlcNAcα1, 4Galβ1R at the non-reducing terminus of O-glycans in gastric mucin | |
| Speak et al. | CD1d presentation of glycolipids | |
| Heine et al. | Identifying efficient Clostridium difficile toxin A binders with a multivalent neo-glycoprotein glycan library | |
| JP6996676B2 (ja) | マンノース-6-リン酸基含有糖蛋白質の製造方法、及び蛍光基結合型マンノース-6-リン酸基含有糖蛋白質の細胞内分布を検出する方法 | |
| JP6888207B2 (ja) | 糖ペプチド又は糖蛋白質の製造方法 | |
| Mamedov et al. | Engineering, and production of functionally active human Furin in N. benthamiana plant | |
| Ledeen et al. | Neurobiology meets glycosciences | |
| KR101457514B1 (ko) | 우렁쉥이(멍게) 유래의 시알산 전이효소 및 이를 이용한 시알화 복합당질 합성 방법 | |
| Low | Structure and function of GPI-specific phospholipases | |
| JP6830576B2 (ja) | エンドm変異体、及びn結合型糖鎖含有化合物又はn結合型糖鎖含有蛋白質の製造方法 | |
| Bakar et al. | Biosynthesis and Function of Glycoconjugates | |
| Pillai | Heterologous expression of human ST6GAL1 in trichoderma reesei | |
| Bigi | Characterization of human sialidase NEU4: role of the proline-rich region in signal transduction | |
| Morrison et al. | Modification of a recombinant GPI‐anchored metalloproteinase for secretion alters the protein glycosylation | |
| Schaus et al. | Degradation of mucin O-glycans by a human gut symbiont requires a complex enzyme repertoire and promotes colonization | |
| Brito-Arias | Glycoconjugates | |
| Cheng | Mucin biosynthesis: purification and characterization of a mucin beta 6N-acetylglucosaminyltransferase | |
| Ferguson | International Glycoconjugate Organization Award | |
| Degani | Molecular characterization of membrane-bound glycoproteins involved in human diseases and potential targets for new therapies | |
| Levy-Ontman | N-Glycosylation of the 66-kDa cell-wall glycoprotein of a red microalga |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 17775430 Country of ref document: EP Kind code of ref document: A1 |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 17775430 Country of ref document: EP Kind code of ref document: A1 |