WO2017191886A1 - Compositions nutritionnelles dessalées fonctionnellement renforcées dérivées d'halophytes et leur procédé de préparation - Google Patents
Compositions nutritionnelles dessalées fonctionnellement renforcées dérivées d'halophytes et leur procédé de préparation Download PDFInfo
- Publication number
- WO2017191886A1 WO2017191886A1 PCT/KR2017/000949 KR2017000949W WO2017191886A1 WO 2017191886 A1 WO2017191886 A1 WO 2017191886A1 KR 2017000949 W KR2017000949 W KR 2017000949W WO 2017191886 A1 WO2017191886 A1 WO 2017191886A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- desalted
- halophyte
- water
- nutritional composition
- extract
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES, NOT OTHERWISE PROVIDED FOR; PREPARATION OR TREATMENT THEREOF
- A23L33/00—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof
- A23L33/10—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof using additives
- A23L33/105—Plant extracts, their artificial duplicates or their derivatives
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES, NOT OTHERWISE PROVIDED FOR; PREPARATION OR TREATMENT THEREOF
- A23L5/00—Preparation or treatment of foods or foodstuffs, in general; Food or foodstuffs obtained thereby; Materials therefor
- A23L5/20—Removal of unwanted matter, e.g. deodorisation or detoxification
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES, NOT OTHERWISE PROVIDED FOR; PREPARATION OR TREATMENT THEREOF
- A23L33/00—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof
- A23L33/10—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof using additives
- A23L33/16—Inorganic salts, minerals or trace elements
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES, NOT OTHERWISE PROVIDED FOR; PREPARATION OR TREATMENT THEREOF
- A23L33/00—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof
- A23L33/20—Reducing nutritive value; Dietetic products with reduced nutritive value
Definitions
- the present invention relates to a functionally reinforced desalted nutritional composition, a desalted extract and a cold-water-extracted salt substitute, which are derived from halophytes that grow in coastal regions under highly saline conditions and thus retain high salt concentrations, and the present invention is also concerned with the use of the desalted nutritional composition to combat obesity.
- the present invention relates to a functionally reinforced desalted nutritional composition, a desalted extract and a salt substitute cold-water-extracted from halophytes that inhabit extreme environments of high salinity under high salt stress, the halophytes being desalted through a cold water extraction process at a low temperature based on the difference in water solubility of salts with change in temperature to allow only sodium chloride to be selectively removed, and the composition thus having decreased sodium content as well as having increased content of useful minerals such as potassium, as well as nutrients and physiologically active substances, which are naturally contained in halophytes.
- Halophytes are plants that naturally grow in saline habitats, such as in coastal regions and around salt fields, where most terrestrial plants can't survive due to the high salinity of soils. Halophytic plants, through metabolic responses that allow them to overcome salt stress, can retain high salt concentrations in their cells and tissues, and can take up seawater owing to their high osmotic potentials. When eaten, the plants taste very salty because they contain high salt concentrations. Halophytes are found growing in communities in high salt areas of salt marshes throughout the world. Representative examples of such halophytic species include Salicornia europaea , Suaeda asparagoides , and Suaeda japonica .
- Salicornia europaea which is an annual halophyte that belongs to the family Chenopodiaceae , is widely distributed in saline habitats, such as salt marshes or coastal areas, where agricultural crops generally cannot grow well, throughout the world including South Korea, Europe and North America.
- This plant has jointed stems, which are thick, fleshy and swollen and are deep green, and grows to 20 to 40 cm tall.
- This succulent herb is, in the classic manual of Chinese herbal medicine 'Shennong Bencaojing', referred to as 'Hamcho' and 'Yeomcho', which mean a salty herb, due to its salty taste, and is also called 'Shincho', which means a very rare numinous herb.
- glasswort In North America, the plant is known as glasswort. It is also known as 'Samphire' in Europe and 'Aatkaeso Sangoso' in Japan. Since S. europaea grows in salt marshes of high salinity, it can accumulate salts to high concentrations in its tissues so as to adjust to osmotic pressure. For this reason, glasswort powder has been used as a vegetable table salt substitute. Recent studies have revealed that, as well as sodium (Na), S. europaea retains calcium (Ca), potassium (K), magnesium (Mg) and iron (Fe) at higher levels relative to other plant species while it contains abundant amounts of essential amino acids, edible fibers, physiologically active nutrients, etc.
- Na sodium
- S. europaea retains calcium (Ca), potassium (K), magnesium (Mg) and iron (Fe) at higher levels relative to other plant species while it contains abundant amounts of essential amino acids, edible fibers, physiologically active nutrients, etc.
- the succulent herb has been reported to have various beneficial physiological effects, for example, anti-thrombotic, anti-diabetic, hypolipidemic, anti-hypertensive, and antioxidant effects, as well as melanin synthesis inhibition.
- S. europaea has been utilized in folk medicine and has been known to be used as a medicinal herb for lifestyle-related diseases.
- S. europaea has beneficial effects for circulatory and gastrointestinal systems.
- the Ohara Sanso Institute of incurable diseases in Japan revealed that S. europaea has excellent efficacies for several cancers, sinus infection, arthritis, hypertension, hypotension, backache, obesity, hemorrhoids, diabetes, etc.
- S. europaea In the Japanese ancient book about medicinal herbs 'Daehwaboncho', S. europaea is noted as 'Shincho', 'Bokcho', which means an herb bringing good fortune, or 'Yeomcho', and is described as eliminating toxins and coprostasis accumulated in the bodies and to have excellent therapeutic effects for various incurable diseases such as cancer, uterine myoma and sinus infection.
- S. europaea improves blood circulation, strengthens blood vessels, and has therapeutic effects for both hypertension and hypotension as well as for sinus infection, nephritis, arthritis, etc.
- S. europaea since S. europaea is effective in treating purulent inflammation and has diverse antibacterial activities, it has been applied to treat inflammations, arthritis-induced swelling, and the like. Also, S. europaea help relieve chronic fatigue and clear the brain to concentrate the mind.
- Seablite whose botanical name is ' Suaeda asparagoides ', is an annual halophytic plant that belongs to the family Chenopodiaceae , and is widely distributed in saline habitats such as coastal areas in South Korea, Japan, China, etc.
- This plant which is also known by the name ' Suaeda glauca ', has narrow and thin leaves like pine needles and, in South Korea, is commonly called 'Gaetsolnamul', which means a coastal herb having thin leaves like pine needles.
- S. asparagoides can be eaten, but its intake is limited owing to its high saline content and is thus merely used as a vegetable salt substitute.
- asparagoides has excellent effects of lowering fever and alleviating hypertension and poor hepatic function while degrading coprostasis and waste matter accumulated in the intestine and excreting them outside the body, thus being useful for constipation, obesity, etc.
- this plant contains physiologically active substances such as polyphenol compounds, and thus has antioxidant activity and inhibits the permeability of capillary vessels leading to strengthening of blood vessels, as well as having active-oxygen scavenging activity and inhibiting lipid peroxidation.
- S. asparagoides has the potential to be developed into a functional food.
- Suaeda japonica which is an annual halophyte that belongs to the family Chenopodiaceae , is a salt-tolerant plant that, like S. europaea , retains large quantities of salt in its tissues and can grow well in highly saline soils. This plant inhabits South Korea, Japan, etc., grows to 20 to 50 cm tall, and is green first and turns violet-red later. This plant also can be eaten, but its intake is limited owing to its high saline contents and is thus merely used as a vegetable salt substitute.
- Chinese herbal medicine the whole part of the plant other than the roots has been used as an herbal medicine and has been known to be effective in treating fever, hypertension, dyspepsia, constipation, obesity, and the like.
- the plant contains large quantities of natural minerals and is rich in secondary metabolites such as polyphenols, flavonoids and saponins, which are highly bioavailable.
- S. japonica has high potential to be utilized as a functional material.
- S . japonica has physiological activities including antioxidant activity and inhibitory activity against ⁇ -glucosidase, which is implicated in the postprandial rise in blood glucose.
- Some studies on components of S. japonica have revealed that this plant contains glycine betaine, which is involved in salt stress tolerance, 2'-hydroxy-6,7-methylenedioxy-isoflavone, loliolide, dehydrovomifoliol, uridine, and the like.
- seawater Over 97% of the water on the earth is seawater, and this huge amount of seawater can be used to alleviate drought and desertification as well as to create new food resources.
- halophytic plants which can be cultivated by seawater agriculture, may be potentially good alternatives for overcoming nutrient and food crises in situations of water and food shortage.
- halophytes have been known to be mainly used as food sources like salads and as vegetable table salt substitutes. Many studies have revealed that powders or extracts from halophytes have beneficial functions, but halophyte products have not been developed as functional foods or materials. This is because the high salinity of halophytes limits their utilization to use as salty sources or soybean sources.
- Korean Pat. No. 10-0724705 (entitled “Edible Liquid Type Composition Comprising Glasswort ( S. europaea ) Extract”) discloses a method of preparing a liquid-type composition containing glasswort as an effective ingredient, including extracting raw halophyte materials including glasswort and mixing the extract with food additives and others to yield a drink, in which the drink mixture can be further dried to yield a solid.
- This patent also describes a food manufacturing method characterized by kneading the drinkable composition at a predetermined ratio.
- these products are not desalted and thus have a high content of sodium chloride, their amounts when added or eaten are limited.
- excessive sodium intake may increase the risk of hypertension, cardiovascular disease, or the like, thereby causing health problems.
- Korean Pat. No. 10-1218355 discloses a method of preparing betacyanin from red glasswort ( S. europaea ).
- the method of preparing the natural edible pigment betacyanin is based on extracting red glasswort, desalting the extract by electrodialysis, and drying the desalted extract.
- this method only serves to obtain a red pigment from glasswort that has turned red, and glasswort necessarily turns red due to physiological changes as chlorophylls are destroyed right before it withers.
- some loss may occur, besides sodium salt, of minerals useful for human bodies, such as potassium, calcium, magnesium iron, and other useful low-molecular weight ingredients.
- Korean Laid-open Publication Pat. No. 10-2006-0110023 describes a method by which glasswort is extracted with hot water or ethanol, the extract powder then being mixed with starch paste and other ingredients to be made into pills. This method is problematic in that the hot water and ethanol extracts cannot contain all glasswort nutrient and in that the high salt concentrations of the extracts are not removed.
- Korean Pat. No. 10-1095619 discloses a method for lowering the salt content of glasswort and a storage method for the desalted glasswort.
- This method includes cutting glasswort into about 0.5 cm lengths, stirring the herb pieces in a 0.1% to 1.0% NaCl solution for 10 to 40 minutes, and storing the salt-reduced glasswort extract at 35°C and 50°C.
- this method has the following problems: since fresh herbs are cut, immersed in a salt solution and stirred at a high temperature above room temperature for a long period of time, most organic compounds contained in glasswort, except for water-insoluble dietary fiber, are lost, and the salt solution does not ensure a strong desalting effect.
- Korean Pat. No. 10-1287065 discloses a method for preparing glasswort powder having improved sanitation and digestibility. This method includes washing fresh glasswort herb, extracting juice from the herb, sterilizing the glasswort juice at 90 to 110°C for 5 to 60 minutes, heating the juice to 50 to 70°C, decompressing and concentrating the juice, degrading the spray-dried powder and remaining juice residues through enzymatic treatment, and pulverizing the resulting product.
- this method does not include a substantial desalting process, high salt concentrations still remain in the glasswort powder.
- Korean Pat. No. 10-1289769 describes a method for preparing desalted milk based on eliminating singly charged minerals contained in milk.
- this method includes passing raw milk through a chloride anion exchange resin and eliminating singly charged minerals through membrane separation.
- This method is applicable only to liquid-phase samples not containing insoluble solids, and has another problem in that milk acidity increases when milk is passed through the anion exchange resin.
- the anion exchange resin can absorb non-mineral organic substances, for example, essential amino acids and alkaloids, thereby causing the loss of a variety of such physiologically active ionic substances.
- Electrodialysis is a process of separating ionic components from a solution. This process is theoretically based on the mass transfer theory, in which ionic components in a solution are selectively passed through a cation exchange resin membrane and an anion exchange resin membrane by a voltage applied to an electric field. Also, electrodialysis is a membrane process that is most commonly used along with reverse osmosis and ultrafiltration. Such an electrodialysis process is mainly applied for desalination using an electrically charged membrane.
- Korean Pat. No. 10-0561103 discloses an electrodialytic method of lowering the salt concentration of Korean traditional soy sauce.
- ultrafiltration is unable to selectively eliminate only sodium salt, and is also disadvantageous in terms of removing useful minerals such as potassium, calcium and magnesium along with sodium.
- the ultrafiltration method has additional drawbacks in that low-molecular-weight organic compounds less than 200 daltons are lost in a sample and high costs are required for maintaining and managing the equipment.
- Osmosis is a natural process.
- a solvent moves across a membrane partition from the side of low solute concentration toward the side of high solute concentration.
- the driving force for the movement of the solvent is the chemical potential generated by the difference in solute concentration.
- pressure is generated and applied to the more highly concentrated solution, this pressure being called osmotic pressure.
- reverse when an external pressure higher than osmotic pressure is applied, the solvent is forced to move from the high to the low solution concentration, and this phenomenon is called reverse osmosis.
- Korean Pat. No. 10-1102259 discloses a desalting method of salted and fermented food using alcohol.
- desalination is achieved by adding alcohol in an amount 0.5 to 10 times more than a raw material to a salted and fermented food to lower salt solubility and thus precipitate the salt, followed by removing the salt through a physical process.
- this method a small amount of salt can be precipitated and removed.
- the alcohol addition rather than the salt removal effect, can cause proteins to coagulate and denature and can also reduce the solubility of polysaccharides, thus leading to precipitation.
- a large amount of acidic polysaccharides and protein-bound polysaccharides rapidly precipitate, thus resulting in a very large loss of nutrients contained in a raw material.
- the present inventors in order to solve the above mentioned problems of desalination, conducted thorough and intensive research into a method capable of effectively removing only sodium salt (NaCl) from halophytes without the loss of useful minerals such as potassium, calcium and iron, nutrients such as carbohydrates and proteins, and useful physiologically active substances such as chlorophylls, polyphenols and flavonoids.
- This research resulted in the development of a desalination method using the difference in water solubility of salts with change in temperature (see FIG. 1).
- the desalted halophyte powder compared with before desalination, was found to have remarkably improved activities, such as anti-oxidant, anti-thrombotic, anti-hypertensive and anti-diabetic activities.
- a cold-water-stirred extract obtained during the halophyte desalination process unlike conventional glasswort salts, was found to have a high content of sodium chloride and a clean salty taste having a savory (umami) flavor, and thus has the potential to be used as a 100% pure vegetable salt substitute.
- Obesity is a kind of metabolic disorder that is caused by various factors such as excess energy intake, genetic susceptibility and decreased physical activity.
- Obesity refers to a condition that is characterized not only by excess weight but also by an increased content of body fat.
- many people become obese owing to excess nutrient intake, and obesity is a growing serious socioeconomic health problem today.
- the prevalence of obesity has been rising mainly in advanced countries, but, in recent years, the population of overweight people has rapidly expanded in South Korea.
- Obesity has been known as a risk factor for many metabolic disorders such as cardiovascular disease, diabetes, non-alcoholic hepatitis, cancers, Alzheimer's disease and osteoarthritis, and it is thus becoming classified as a serious modern disorder.
- obesity increases intracellular oxidative stress, which facilitates the dysregulation of adipocytokine release from adipose tissues, which contributes to the development of several diseases, such as metabolic syndromes, including atherosclerosis and diabetes, and ischemic heart disease.
- metabolic syndromes including atherosclerosis and diabetes, and ischemic heart disease.
- Physical exercise, diet restriction, medication and surgical operations are the major preventive or therapeutic methods of treating obesity.
- anti-obesity drugs which are chemical synthetic substances, have been known to have strong anti-obesity effects, but also have many side effects. In this regard, recently, there is growing interest in natural plant materials that are safe and have mild side effects.
- anti-obesity natural plant materials include polyphenols, which suppress fat synthesis and adipocyte differentiation, chili pepper capsaicin, which reduces body fat by activating body energy metabolism, and vegetable dietary fiber, which inhibits fat absorption and gives a feeling of satiation.
- the halophyte-derived functionally reinforced desalted nutritional composition developed in the present invention sodium chloride could be effectively removed alone from glasswort.
- the present inventors thus felt that they were able to overcome the problems encountered in previous studies, and investigated to determine the anti-obesity effect of the desalted nutritional composition.
- the functionally reinforced desalted nutritional composition was found to have remarkably excellent anti-obesity and body-fat-reducing effects compared to before desalination, ensuring potential for application as a functional food and functional feedstuff effective in preventing and/or treating obesity, thus leading to the present invention.
- the present invention provides a method of preparing a functionally reinforced desalted nutritional composition from a halophyte, comprising the steps of (a) mixing dried powder of the halophyte with water at 9°C or lower and stirring the mixture; (b) centrifuging the stirred mixture and removing a supernatant having a high salt content to recover a desalted precipitate; and (c) drying the desalted precipitate.
- the present invention also provides a functionally reinforced nutritional composition from a halophyte, comprising sodium of 0.04 to 6.8 wt% and carbohydrates of 61 wt% or greater, based on the dry weight.
- the present invention provides a method of preparing a functionally reinforced desalted extract from a halophyte, comprising the steps of (a) mixing dried powder of the halophyte with water at 9°C or lower and stirring the mixture; (b) centrifuging the stirred mixture and removing a supernatant having a high salt content to recover a desalted precipitate; (c) extracting the desalted precipitate in a liquid phase to obtain an extract; and (d) drying the liquid-phase extract.
- the method of preparing a functionally reinforced desalted extract from a halophyte is characterized in that it further comprises drying the desalted precipitate before the liquid-phase extraction step of the desalted precipitate.
- the present invention provides a functionally reinforced desalted extract from a halophyte, which is characterized in that is extracted from a desalted product of the halophyte and has a total salt content of less than 11.0 wt% and insoluble dietary fiber of less than 3.2 wt%, based on the dry weight.
- the functionally reinforced desalted nutritional composition from a halophyte according to the present invention is characterized in that it comprises potassium (K) of 0.1 to 3.0 wt%, calcium (Ca) of 0.1 to 2.0 wt% and magnesium (Mg) of 0.1 to 1.5 wt%, based on the dry weight.
- the functionally reinforced desalted extract from a halophyte according to the present invention is characterized in that it comprises polyphenols of 0.1 to 10.0 wt% and flavonoids of 0.1 to 7.0 wt%, based on the dry weight.
- the halophyte-derived functionally reinforced desalted extract is characterized in that it comprises chlorophylls of 0.3 to 10.0 wt% based on the dry weight.
- the halophyte-derived functionally reinforced desalted nutritional composition is characterized in that it includes trans-ferulic acid.
- the present invention provides a method of preparing a cold-water-extracted salt substitute from a halophyte, comprising the steps of (a) mixing dried powder of the halophyte with water at 9°C or lower and stirring the mixture; (b) centrifuging the stirred mixture to obtain a supernatant; (c) concentrating the supernatant and purifying the concentrate with activated carbon; and (d) spray-drying the purified concentrate.
- the halophyte-derived cold-water-extracted salt substitute is characterized in that it has a total salt content of 50.0 wt% or more and a salt composition in which the weight ratio of potassium to sodium (K:Na) ranges from 1:10.1 to 1:19.0.
- the present invention provides a cold-water-extracted salt substitute from a halophyte characterized in that it has a total salt content of 50.0 wt% or more and a salt composition in which the weight ratio of potassium to sodium (K:Na) ranges from 1:10.1 to 1:19.0.
- the halophyte-derived cold-water-extracted salt substitute according to the present invention is characterized in that it comprises glutamic acid in an amount ranging from 0.1 to 50 mg/g.
- the present invention also provides a pharmaceutical composition for combatting obesity and for reducing body fat, comprising the halophyte-derived functionally reinforced desalted nutritional composition or trans-ferulic acid derived from a halophyte.
- the present invention further provides a functional food for combatting obesity and for reducing body fat, comprising the halophyte-derived functionally reinforced desalted nutritional composition or trans-ferulic acid derived from a halophyte.
- the present invention still further provides a feedstuff for combatting obesity and for reducing body fat, comprising the halophyte-derived functionally reinforced desalted nutritional composition or trans-ferulic acid derived from a halophyte.
- the method of preparing a functionally reinforced desalted nutritional composition or a desalted extract from a halophyte according to the present invention, through a cold water desalination process based on the difference in water solubility of salts with change in temperature, enables the effective removal of only sodium chloride with no loss in useful minerals such as potassium, calcium and magnesium, nutrients such as carbohydrates and proteins, and useful physiologically active substances such as chlorophylls, polyphenols and flavonoids.
- the removed sodium chloride solution can be utilized as a table salt substitute owing to its high contents of sodium chloride and glutamic acid.
- FIG. 1 is a flowchart showing processes of preparing a functionally reinforced desalted nutritional composition, a desalted extract and a salt substitute, which are derived from halophytes, according to the embodiments of the present invention
- FIG. 2 is a graph showing the water solubility of salts with varying temperature
- FIG. 3 is a photograph showing the appearance of Salicornia europaea powder before and after being desalted
- FIG. 4 is a photograph in which non-desalted Salicornia powder (SP), cold-water-desalted Salicornia powder (CW-DSP) and hot-water-desalted Salicornia powder (HW-DSP) were compared with each other for chlorophyll content;
- SP non-desalted Salicornia powder
- CW-DSP cold-water-desalted Salicornia powder
- HW-DSP hot-water-desalted Salicornia powder
- FIG. 5 is a photograph showing the results of colorimetric analysis for total polyphenols, total flavonoids and total proteins of hot-water extracts of non-desalted halophyte dried powder and hot-water extracts of cold-water-desalted halophyte dried powder' according to an embodiment of the present invention
- FIG. 6 is a photograph showing the results of colorimetric analysis for total sugars and total acidic sugars of hot-water extracts of non-desalted halophyte dried powder and hot-water extracts of cold-water-desalted halophyte dried powder according to an embodiment of the present invention
- FIG. 7 is a graph showing the antioxidant activity of hot-water extracts of non-desalted halophyte dried powder and hot-water extracts of cold-water-desalted halophyte dried powder according to an embodiment of the present invention depending on concentration;
- FIG. 8 is a graph showing inhibitory activity against Angiotensin-I-Converting Enzyme (ACE) of hot-water extracts of non-desalted halophyte dried powder and hot-water extracts of cold-water-desalted halophyte dried powder according to an embodiment of the present invention depending on concentration;
- ACE Angiotensin-I-Converting Enzyme
- FIG. 9 is a graph showing inhibitory activity against alpha-glucosidase of hot-water extracts of non-desalted halophyte dried powder and hot-water extracts of cold-water-desalted halophyte dried powder according to an embodiment of the present invention depending on concentration;
- FIG. 11 shows the body-fat-reducing effect of the Desalted Salicornia Powder (DSP) in obese rats induced by a high-fat diet at 6 and 12 weeks
- G1 normal control group
- G2 obesity control group induced by high-fat diet
- G3 administered with high-fat diet plus 200 mg/kg of Salicornia Powder (SP)
- G4 administered with high-fat diet plus 200 mg/kg of Desalted Salicornia Powder (DSP)
- FIG. 13 shows the results of HPLC chromatography of trans-ferulic acid, which is a marker contained in the Desalted Salicornia Powder (DSP)
- DSP Desalted Salicornia Powder
- A Analytical HPLC profile of DSP-EW
- B Analytical HPLC profile of authentic trans -ferulic acid
- C Multiple preparative HPLC profile of DSP-EW
- 1 caffeic acid
- 2 p -coumaric acid
- 3 trans -ferulic acid
- 4 isorhamnetin-3- ⁇ -D-glucoside
- FIG. 14 are photographs and graphs showing the inhibitory effects of trans-ferulic acid (TFA), isolated from the Desalted Salicornia Powder (DSP), on intracellular lipid accumulation and triglyceride formation in 3T3-L1 cells (One-way ANOVA test; *: p ⁇ 0.05, **: p ⁇ 0.01, ***: p ⁇ 0.001, #: p ⁇ 0.05, ##: p ⁇ 0.01, ###: p ⁇ 0.001); and
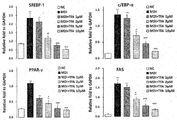
- FIG. 15 shows the results of Real-Time RT-PCR to determine the effect of trans-ferulic acid (TFA), isolated from the Desalted Salicornia Powder (DSP), on SREBP1, c/EBP ⁇ , PPAR ⁇ and FAS gene expression (One-way ANOVA test; *: p ⁇ 0.05, **: p ⁇ 0.01, ***: p ⁇ 0.001, #: p ⁇ 0.05, ##: p ⁇ 0.01, ###: p ⁇ 0.001).
- TFA trans-ferulic acid
- Halophytes which contain various useful substances such as dietary fiber, essential amino acids, vegetable minerals and physiologically active substances, have a limitation in their applicability owing to their high salt content.
- the present invention based on the difference in water solubility of salts with change in temperature, when dried powder of a halophyte was extracted with stirring in cold water at a low temperature for a short time, compared with the cases of being extracted with room-temperature water and hot water, the elution of useful minerals, except for sodium, and organic soluble components was remarkably reduced, whereas there was no great difference in the elution of sodium salt.
- halophyte to a halophyte were added cold water, room-temperature water and hot water, respectively, followed by stirring and centrifugation to remove a supernatant.
- the resulting desalted extract was recovered and dried so as to yield a halophyte-derived functionally reinforced desalted nutritional composition.
- the extraction with cold water was found to be able to effectively remove sodium salts while minimizing the elution of organic substances.
- the present invention relates to a method of preparing a functionally reinforced desalted nutritional composition from a halophyte, comprising the steps of (a) mixing dried powder of the halophyte with water at 9°C or lower and stirring the mixture, (b) centrifuging the stirred mixture and removing a supernatant having a high salt content to recover a desalted precipitate, and (c) drying the desalted precipitate, and a functionally reinforced desalted extract from a halophyte, including sodium of 0.04 to 6.8 wt% and carbohydrates of 61 wt% or greater, based on the dry weight.
- Halophytes are plants that naturally grow in saline habitats, such as in coastal regions and around salt fields. Examples of halophytes include, but are not limited to, glasswort ( Salicornia Spp . ), Suaeda asparagoides , and Suaeda japonica .
- the halophyte dried powder may be prepared by washing a halophyte to remove impurities followed by drying.
- a dried product itself can be used, but a powder form is preferred for more effective extraction.
- the halophyte-derived functionally reinforced desalted nutritional composition is prepared as follows. First, a dried halophyte product is mixed with water at 9°C or lower, preferably 0.1 to 4°C, and is then stirred. Preferred water is non-saline water, such as tap water or distilled water. If the stirring is carried out at a temperature of about 10°C or higher, as compared with the condition of 0.1 to 9°C, there is no great change in the elution degree of sodium salt, but other organic soluble components and minerals, such as potassium, are eluted, causing large loss of nutrients from the desalted dried product.
- the dried product of halophyte is preferably used in an amount of 40 to 70 g per 1 L for extraction.
- the solvent amount is relatively large, increasing the total amount to be centrifuged. This makes the extraction process ineffective.
- an amount greater than 70 g does not ensure effective stirring.
- the stirring is preferably carried out for 1 to 5 minutes. Stirring for less than one minute results in a decrease in desalination efficiency for halophytes. When the stirring time exceeds 5 minutes, the elution of soluble organic components as well as sodium salt is increased.
- the stirred mixture is centrifuged, and a supernatant having a high salt content is then removed to recover a desalted precipitate.
- the precipitate may be obtained according to a method commonly known in the art, and the method is not particularly limited as long as it can separate the stirred mixture into a supernatant and a precipitate.
- a filtration method may be used to obtain the precipitate.
- the desalted precipitate may be further stirred so as to further lower the remaining small amount of salt content.
- the finally desalted precipitate is recovered and dried.
- the desalted nutritional composition prepared according to the present invention may comprise sodium (Na) of 0.04 to 6.8 wt%, carbohydrates of 61 wt% or greater, potassium (K) of 0.1 to 3.0 wt%, calcium (Ca) of 0.1 to 2.0 wt%, magnesium (Mg) of 0.1 to 1.5 wt%, polyphenols of 0.1 to 10.0 wt%, flavonoids of 0.1 to 7.0 wt%, and chlorophylls of 0.3 to 10.0 wt%, based on the dry weight.
- the desalted precipitate obtained by desalting a halophyte with cold water, and its dried powder were extracted with water or ethanol, as compared with an extract from a non-desalted halophyte, they were found to have remarkably reduced salt content as well as having remarkably increased content of functional components and nutrients.
- the present invention provides a method of preparing a functionally reinforced desalted extract from a halophyte, comprising the steps of (a) mixing dried powder of the halophyte with water at 9°C or lower and stirring the mixture; (b) centrifuging the stirred mixture and removing a supernatant having a high salt content to recover a desalted precipitate; (c) extracting the desalted precipitate in a liquid phase to obtain an extract; and (d) drying the liquid-phase extract, and a functionally reinforced desalted extract from a halophyte, which is characterized in that it is extracted from a desalted product of the halophyte and has a total salt content of less than 11.0 wt% and insoluble dietary fiber of less than 3.2 wt%, based on the dry weight.
- the desalted precipitate from a halophyte is recovered using the same method described above.
- the desalted precipitate may be extracted with water, or may be dried and then extracted with an organic solvent, such as methanol, ethanol, butanol, ethyl acetate, acetone or diethyl ether, thus giving a desired extract.
- an organic solvent such as methanol, ethanol, butanol, ethyl acetate, acetone or diethyl ether, thus giving a desired extract.
- the desalted precipitate is preferably dried before being subjected to liquid-phase extraction.
- the liquid-phase extraction of the desalted halophyte precipitate with an organic solvent may be carried out by reflux extraction at room temperature or near a temperature at which the organic solvent becomes volatile.
- the desalted precipitate is preferably used in an amount of 40 to 75 g per liter of extraction solvent. An amount less than 40 g increases the cost of the extraction solvent. When the amount of solvent exceeds 75 g, extraction efficacy is reduced.
- the additional liquid-phase extraction process has an advantage of increasing the extract yield.
- the halophyte-derived functionally reinforced desalted extract prepared according to the present invention is characterized in that it has a total salt content of less than 11.0 wt% and insoluble dietary fiber of less than 3.2 wt%, and may comprise polyphenols of 0.1 to 10.0 wt%, flavonoids of 0.1 to 7.0 wt% and chlorophylls of 0.3 to 10.0 wt%.
- halophyte-derived functionally reinforced desalted extract has various in vivo physiological activities, such as antioxidant, anti-thrombotic, anti-hypertensive and anti-diabetic activities, it has the potential to be applied as a raw material in foods, cosmetics, medicines, and the like.
- the halophyte-derived functionally reinforced desalted nutritional composition comprises, as an effective component, trans-ferulic acid, which suppresses the adipocyte differentiation and genes involved in lipid synthesis, as well as containing dietary fiber in an amount greater than before being desalted.
- trans-ferulic acid which suppresses the adipocyte differentiation and genes involved in lipid synthesis, as well as containing dietary fiber in an amount greater than before being desalted.
- the composition has good anti-obesity and body-fat-reducing effects.
- the present invention relates to a pharmaceutical composition and functional food and feedstuff for anti-obesity and body-fat-reducing effects, comprising the above functionally reinforced desalted halophyte dried product.
- the remaining supernatant was found to have a high content of sodium chloride while having a low content of potassium and a high content of glutamic acid, and is thus applicable as a vegetable salt substitute having a clean salty taste with a savory (umami) flavor.
- the present invention relates to a method of preparing a cold-water-extracted salt substitute from a halophyte, comprising the steps of (a) mixing dried powder of the halophyte with water at 9°C or lower and stirring the mixture; (b) centrifuging the stirred mixture to obtain a supernatant; (c) concentrating the supernatant and purifying the concentrate with activated carbon; and (d) spray-drying the purified concentrate, and a halophyte-derived cold-water-extracted salt substitute prepared according to the method, which is characterized in that it has a total salt content of 50.0 wt% or more and a salt composition in which the weight ratio of potassium to sodium (K:Na) ranges from 1:10.1 to 1:19.0.
- the cold-water-stirred supernatant remaining after a desalted precipitated is recovered can be concentrated to a salinity of 15 to 19% and a total solid content of 20% or higher.
- the supernatant may be purified using activated carbon of 3-5% based on the total solid content of the concentrate, and then spray-dried to give a halophyte-derived cold-water-extracted salt.
- the amount of activated carbon used in the purification may be varied to control the content of organic substances and the color of salt.
- the concentration method of the cold-water-stirred supernatant is not particularly limited as long as it can concentrate the supernatant. Preferred is vacuum concentration.
- Example 1 Evaluation of cold water extraction of S. europaea for desalting effect
- the stirred mixture was centrifuged at a time interval of 5 min at 10,000 rpm for 20 min.
- Supernatants were vacuum-filtered through a membrane filter (0.45 ⁇ m pore size) and then analyzed for salinity (ATAGO ES-421, ATAGO Co. LTD. Japan) and Brix (ATAGO PAL-1, ATAGO Co. LTD. Japan).
- the supernatants were vacuum-concentrated, freeze-dried (EYELA FDU-2200, ETELA, Japan), and then analyzed for total solid content.
- the measured total solid contents are, along with salt percentage in total solid and content of solids other than salt, given in Table 1, below.
- FIG. 2 shows the water solubility of salts with varying temperatures.
- Table 1 correspond with the constant water solubility of NaCl according to temperature, as shown in FIG. 2.
- the soluble solid content (Brix, %)/salinity (%) index was measured. That is, the lower the Brix/salinity ratio, the lower the presumed loss of organic solids due to desalination. The index was found to increase gradually over time under all temperature conditions.
- Example 2 Preparation of desalted nutritional compositions from S. europaea , S. asparagoides and S. japonica
- Desalted nutritional compositions were prepared using three plants, Salicornia europaea , Suaeda asparagoides and Suaeda japonica , which are known to be extreme halophytes that naturally grow in South Korea. Fresh plants were washed with tap water and freeze-dried to be powdered. Based on the results from Example 1, in which, when extraction is conducted with cold water at 4°C or lower for 4 min or shorter, the extraction of organic substances is minimized while salts are effectively removed, 100 g of the dried powder was added to 2 L of cold water (4°C), stirred at 4°C for 4 min, and centrifuged at 10,000 rpm for 20 min.
- ICPS Inductively Coupled Plasma Spectrometry
- the total polyphenol content was determined in a 96-well microplate according to a modified Folin-Davis method.
- Non-desalted and desalted halophyte powders were extracted with 70% methanol, dried and dissolved in distilled water.
- 20 ⁇ l of each sample was mixed with 250 ⁇ l of 2% sodium carbonate and 15 ⁇ l of 50% Folin-Ciocalteu reagent (Sigma Co., USA), and the solution was allowed to react at room temperature for 30 min. Then, the absorbance was measured at 725 nm using a microreader (Bio-RAD, x-Mark, USA).
- tannic acid solutions of 0 to 500 ⁇ g/mL (Sigma Co., USA) were used instead of the samples, and, from the calibration curve thus obtained, the amount of total polyphenols contained in the extraction samples were calculated.
- the total flavonoid content was determined in a 96-well microplate according to a modified Abdel-Hameed method.
- Non-desalted and desalted halophyte powders were extracted with 70% methanol, dried and dissolved in distilled water.
- To 30 ⁇ l of each sample were added 200 ⁇ l of 90% diethylene glycol and 5 ⁇ l of 1 N NaOH.
- the solution was allowed to react at 37°C for 1 hr.
- the absorbance was measured at 420 nm using a microreader (Bio-RAD, x-Mark, USA).
- rutin solutions of 0 to 500 ⁇ g/mL (Sigma Co., USA) were used instead of the samples, and, from the calibration curve thus obtained, the amount of total flavonoids contained in the extraction samples were calculated.
- Chlorophyll b (mg/mL) 25.88OD645 - 5.50OD663
- the desalted halophyte samples were found to have increased contents of carbohydrates and crude proteins.
- the main component removed during desalination was found to be sodium (Na), while the concentrations of other minerals, such as potassium, calcium, magnesium and iron, were increased after desalting.
- FIG. 3 shows the appearances of Salicornia europaea powder (5 g) before and after being desalted.
- Salicornia powder was desalted in cold water for a short time, chlorophylls were eluted only in small amounts, with almost all remaining, while the desalted powder became lightened by desalination and its volume was thus increased.
- Non-desalted Salicornia powder SP
- cold-water-desalted Salicornia powder CW-DSP
- hot-water-desalted Salicornia powder HW-DSP
- Chlorophyll is a green pigment that is plentiful in the chloroplasts of plants in which photosynthesis occurs, and is weakly associated with proteins.
- Chlorophyll has a unique chemical structure that has a porphyrin (tetrapyrrole) ring with a magnesium atom at its center, and is a hydrophobic compound having a long hydrocarbon tail attached to the porphyrin ring (Rudiger, W.
- chlorophyll which is a functional raw material designated in the Health Functional Food Code, is a functional substance for improving antioxidant activity and immunity
- cold-water-extracted halophyte powder has the potential to be used as a functionally reinforced nutritional composition.
- chlorophyll is weakly resistant to heat and thus easily degraded by heat
- hot-water-desalted halophyte powder has a remarkably low chlorophyll content.
- EXAMPLE 3 Preparation of functionally reinforced desalted extract from halophytes (hot-water extracts and ethanol extracts)
- COMPARATIVE EXAMPLE 1 Preparation of hot-water extracts and ethanol extracts from non-desalted halophytes
- Hot-water extracts and ethanol extracts obtained from 2hrs extraction were prepared according to the same method as in Example 3 except that non-desalted halophytes ( S. europaea , S. asparagoides and S. japonica ) were used instead of the functionally reinforced desalted nutritional compositions (desalted powders) from S. europaea , S. asparagoides and S. japonica .
- the hot-water extracts of Comparative Example 1 from non-desalted halophytes S. europaea , S. asparagoides and S. japonica ) were found to have total salts of 55.8 to 62.0%, total carbohydrates of 25.8 to 33.3% and insoluble dietary fiber of 1.6 to 2.1%.
- the acidic sugar contents were found to range from 11.6 to 17.8%, which are relatively high compared to other general plants, indicating that acidic sugars were composed mainly of glucuronic acid and galacturonic acid.
- the hot-water extract of the cold-water-desalted dried powder, prepared in Example 3 showed a remarkable decrease of more than about 90% in total salt content while showing remarkable increases in total sugar content (51.0 to 63.0%), and, in particular, in total acidic sugar content (22.2 to 32.8%).
- acidic sugars in particular have good immune-enhancing, anti-coagulant, anti-thrombotic, and anticancer activities.
- the hot-water extracts of halophyte powder obtained through desalting with cold water can be used in functionally reinforced nutritional compositions because they contain high concentrations of acidic sugars.
- the hot-water extracts of Example 3 showed increases of 50 to 100% in concentrations of total polyphenols (up to 40.8 mg/g), total flavonoids (up to 31.0 mg/g) and total proteins (up to 15.9 wt%).
- the hot water extracts obtained from the three desalted halophytes showed higher total salt content (9.6 to 10.8%) than 2 hrs hot water-extracted samples.
- the ethanol extract of the cold-water-desalted dried powder, prepared in Example 3 showed a remarkable decrease of more than about 90% in total salt content while showing remarkable increases in concentrations of total neutral sugars and total acidic sugars.
- the ethanol extracts were found to contain plenty of polyphenols, flavonoids and chlorophylls compared with the hot-water extracts, and, compared with Comparative Example 1 (the ethanol extracts of dried powder before being desalted), showed remarkable increases in the content of total polyphenols (76.7 to 90.8 mg/g), total flavonoids (52.6 to 66.4 mg/g) and total chlorophylls (85.3 to 98.2 mg/g).
- the ethanol extracts obtained from the three desalted halophytes showed higher total salt content (5.5 to 6.9%) than 2 hrs ethanol-extracted samples.
- the methods of preparing a functionally reinforced desalted nutritional composition and a desalted extract from a halophyte according to the present invention through a cold water desalination process based on the difference in the water solubility of salts with change in temperature, enables the effective removal of only sodium chloride without causing useful functional plant compounds to be eluted, leading to a remarkable increase in their content compared to the case of not being desalted.
- the functionally reinforced desalted nutritional composition and the desalted extract from a halophyte according to the present invention have the potential to be applied as a functionally reinforced good nutritional material.
- Antioxidant activity was assayed using 1,1-diphenyl-2-picryl hydrazyl (DPPH, Sigma Co., USA) based on the method of Blois (Chen, et. al., 1999. J. Agric. Food Chem. 47. 2226-2228).
- DPPH DPPH
- concentrations 25, 50 and 100 ⁇ g/ml
- the free radical scavenging activity was expressed as the inhibition percentage of free radicals by the sample.
- the IC 50 value is defined as the sample concentration required to scavenge 50% of DPPH free radical.
- ROS reactive oxygen species
- free radicals generated in cells by our own metabolism When reactive oxygen species (ROS) and free radicals generated in cells by our own metabolism are excessively produced, they cause oxidative stress in respective parts of our bodies and thus make it difficult to maintain intracellular homeostasis, eventually leading to a wide variety of diseases, including cancer, brain diseases, such as stroke and Parkinson's disease, heart diseases, ischemia, arteriosclerosis, skin diseases, digestive diseases, inflammation, rheumatoid arthritis, autoimmune disease, and aging.
- antioxidant compounds which remove reactive oxygen species or inhibit free-radical production, can be used for preventing and/or treating various diseases and for suppressing skin aging, which are caused by intracellular oxidative stress.
- Anti-thrombotic activity was evaluated by assaying anti-coagulant activity using a previously known method (Sohn et al., 2004. Kor. J. Pharmacogn 35. 52-61; Kwon et al., 2004. J. Life Science, 14. 509-513; Ryu et al., 2010. J. Life Science, 20. 922-928), and Prothrombin Time (PT) and activated Partial Thromboplastin Time (aPTT) were measured. Used was commercially available control plasma (MD Pacific Technology Co., Ltd, Huayuan Industrial Area, China), and PT and aPTT levels were measured as follows.
- the blood which is a body constituent, has a wide variety of critical functions, such as transporting oxygen, nutrients and wastes, acting as a buffer, maintaining body temperature, regulating osmotic pressure, maintaining ion balance, keeping water content constant, humoral regulation, maintaining and regulating blood pressure, and protecting the body.
- the blood clotting system and the clot dissolution system are regulated in a mutually complementary manner to facilitate blood circulation.
- the normal blood clotting process occurs as follows. Platelets adhere to the wall of the damaged blood vessels and aggregate, promoting the formation of a platelet plug (primary clot). Then, the blood clotting system is activated, and a fibrin clot is formed around platelet clumps.
- Substances inhibiting the thrombin activity can be used to prevent and treat various clotting disorders caused by abnormal excessive blood clotting.
- the intrinsic coagulation pathway lead to the formation of a fibrin clot.
- the intrinsic pathway is activated by a sequential activation of coagulation factors, XII, XI, IX and X, in stepwise order to convert prothrombin to active thrombin.
- the specific inhibition of clotting factors is also a major target in developing therapeutic agents for clotting disorders.
- the S. europaea sample prepared in Comparative Example 1 displayed, at 5 mg/ml, slightly increased PT and aPTT values, 1.08 times and 1.21 times, respectively, whereas the S. europaea sample prepared in Example 3 (the cold-water-desalted hot-water extracts), at the same concentration, was found to have considerably prolonged PT and aPTT, 1.85 times and 2.22 times increased, respectively, indicating excellent anti-thrombotic activity.
- ACE Angiotensin-I-Converting Enzyme
- Angiotensin-I-Converting Enzyme cleaves the C-terminal dipeptide His-Leu from the decapeptide angiotensin I and thus converts angiotensin I to active angiotensin II, which stimulates blood vessel constriction.
- the elevated angiotensin II level due to ACE results in a sharp increase in blood pressure.
- Angiotensin II also stimulates the secretion of the anti-diuretic hormone aldosterone and promotes water and sodium retention by the kidneys, thereby increasing blood volume and blood pressure.
- ACE degrades and thereby inactivates bradykinin, which causes blood vessels to relax and thus causes blood pressure to fall, resulting in an increase in blood pressure.
- the inhibition of ACE activity may prevent vasoconstriction and thus lower blood pressure.
- compounds having inhibitory activity against ACE can be developed as a preventive and/or therapeutic agent for hypertension.
- the halophyte samples prepared in Comparative Example 1 all exhibited, at 1 mg/ml, low ACE inhibitory activity of less than 30%.
- the halophyte samples prepared in Example 3 were all found to have remarkably increased ACE inhibitory activity (65.3%, 59.7% and 56.9% inhibition with the samples from S. europaea , S. asparagoides and S. japonica , respectively).
- the carbohydrate digestive enzymes maltase, sucrase and glucoamylase are present in the brush border of the small intestine, which are also known as alpha-glucosidases.
- the inhibition of excessive activities of these enzymes blocks the breakdown of disaccharides and polysaccharides into monosaccharides and thereby delays excessive elevation of blood sugar levels.
- the inhibition of alpha-glucosidase activity has been used as a tool for measuring anti-diabetic efficacy.
- Alpha-glucosidase activity was determined using a slightly modified Ove method (Ove, N.; Cowell, G.M.; Tranum-Jenser, J. Hansen, O.; Welinder, K.G. J. Biol. Chem. 261:12306-12309, 1986).
- glucose oxidase-peroxidase reagent was added to 180 ⁇ l of the reaction mixture in the 96-well plate to generate hydrogen peroxide, which reacts with ⁇ -dianisidine to form a colored product.
- the color intensity was measured at 540 nm, and the absorbance of the reaction mixture was compared with that of a control not containing a sample.
- a positive control was used 0 to 10 ⁇ g/ml of acarbose (Sigma Co., USA).
- Mammalian alpha-glucosidases are digestive enzymes that are present along the brush border membrane of the differentiated enterocytes lining the villi of the small intestine.
- Alpha-glucosidases stimulate the hydrolysis of dietary carbohydrates in the form of oligosaccharides and polysaccharides into monosaccharides to allow them to be absorbed. Elevated activity of alpha-glucosidase increases such digestion and thus increase the rate of glucose absorption, causing hyperglycemia.
- Alpha-glucosidase inhibitors delay the digestion of carbohydrates in the small intestine and thereby lower postprandial blood sugar levels while delaying the insulin secretion induced by high blood sugar levels.
- alpha-glucosidase inhibitors include acarbose, miglitol and voglibose, which have been used for treating type 2 diabetes.
- Isorhamnetin- ⁇ -D-glucopyranoside which is an antioxidant flavonoid glucoside isolated from an extract of Salicornia europaea , has been reported to have anti-diabetic efficacy.
- the halophyte samples of Comparative Example 1 (the non-desalted hot-water extracts) exhibited about 15.2 to 40.2% alpha-glucosidase inhibition
- the halophyte samples of Example 3 (the cold-water-desalted hot-water extracts) were found to have remarkably increased inhibitory activity against alpha-glucosidase (70.8%, 76.3% and 65.2% inhibition for the samples from S. europaea , S. asparagoides and S. japonica , respectively).
- halophyte dried powder S. europaea , S. asparagoides and S. japonica .
- 100 g of halophyte dried powder S. europaea , S. asparagoides and S. japonica .
- the supernatant having a high salt content was separated while a desalted precipitate was recovered therefrom.
- the precipitate was further desalted in cold water once more according to the same method as described above, and the second desalted precipitate was recovered.
- the second supernatant was pooled together with the first supernatant and vacuum-concentrated at 90°C to achieve a salinity of 18 to 19% and a total solid content of about 26 to 28%.
- the concentrate was purified using activated carbon in an amount of 5% based on the total solid content, and spray-dried using a spray dryer (EYELA Spray Dryer SD1-1000, Japan), thereby yielding a halophyte-derived cold-water-extracted salt substitute.
- the cold-water-extracted salt substitute was then evaluated for total salt, cations and glutamic acid content, and the results are given in Table 6 below (the analysis was performed in the research institute of the Korean Food Industry Association).
- the cold-water-extracted salt substitute was found, compared to the hot water-extracted salt of Korean Pat. No. 10-0784229, to have low organic content while having high contents of sodium chloride and glutamic acid, which contribute to a clean salty taste having a savory (umami) flavor.
- the sodium to potassium ratio was greater than 10:1 (Na:K), and thus the present cold-water-extracted salt substitute was found to have a high sodium/potassium ratio.
- glutamic acid content was found to be remarkably increased compared to that of the conventional hot-water-extracted salt. This result comes from its nature whereby, since glutamic acid is an acidic amino acid that is highly water soluble, it is, unlike other organic substances including other amino acids, easily eluted even under conditions of stirring in cold water for a short time. In addition to this result, it is also a polar compound that is not adsorbed onto activated carbon during purification.
- the present invention enables only the sodium chloride component to be effectively removed from halophytes to obtain a functionally reinforced desalted nutritional composition from a halophyte, as well as enabling the residue of desalination to produce a pure vegetable salt having a clean salty taste with a savory (umami) flavor. Therefore, this invention is an innovative method capable of fully (100%) utilizing halophytes.
- the total carbohydrate content was found to be increased about 1.85- to 2.06-fold in the desalted nutritional compositions (desalted powder) through desalination of halophytes.
- These carbohydrates were analyzed and found to be composed of about 95% or greater dietary fiber in S. europaea , S. asparagoides and S. japonica powders.
- the dietary fiber content includes both soluble and insoluble dietary fiber (the analysis was performed in the research institute of the Korean Food Industry Association).
- Anti-obesity efficacy was investigated for the S. europaea - derived desalted nutritional composition (desalted powder), which has plenty of dietary fiber as well as polyphenols and flavonoids.
- TEST EXAMPLE 4-1 Evaluation for body-weight-reducing effect of the S. europaea- derived desalted nutritional composition in Sprague- Dawley rats with obesity induced by high-fat diet
- the desalted powder of Salicornia europaea (the desalted nutritional composition), from which 95% or more sodium was removed and which is made rich in dietary fiber, polyphenols and flavonoids through a cold water-desalting process, was evaluated for anti-obesity efficacy.
- the evaluation was performed in high-fat diet-induced obese Sprague-Dawley (SD) rats using the Desalted Salicornia Powder (DSP) prepared in Example 2, the non-desalted Salicornia Powder (SP) as a comparative control and, as a positive control, the commonly available natural anti-obesity material Garcinia Extract (GE), which is an extract from the roots of Garcinia cambogia .
- SD Desalted Salicornia Powder
- SP Desalted Salicornia Powder
- SP non-desalted Salicornia Powder
- GE the commonly available natural anti-obesity material
- SD rats were randomly divided into five groups each consisting of ten rats, as follows: G1: normal control group, G2: obesity control group induced by high-fat diet, G3: administered with 200 mg/kg of Salicornia Powder (SP), G4: administered with 200 mg/kg of Desalted Salicornia Powder (DSP), and G5: positive control group administered with 200 mg/kg of Garcinia Extract (GE).
- G1 normal control group
- G2 obesity control group induced by high-fat diet
- G3 administered with 200 mg/kg of Salicornia Powder
- DSP Desalted Salicornia Powder
- G5 positive control group administered with 200 mg/kg of Garcinia Extract (GE).
- FIG. 10 shows the changes in the average body weight of rats in the five groups over 12 weeks
- FIG. 11 shows the results of statistical analysis of the body weight of rats at 6 and 12 weeks.
- rats in the obesity control group fed with the high-fat diet showed increased body weight starting at three or four weeks compared with the normal control group, and, at 6 weeks, the body weight of the obesity control group was significantly increased compared to the normal control group (p ⁇ 0.05).
- the body weight of the group administered with 200 mg/kg of Desalted Salicornia Powder (DSP) was found to be significantly lower than that of the obesity control group (p ⁇ 0.05).
- the obesity control group showed a remarkable increase in body weight compared to the normal control group (p ⁇ 0.001), and the body weight of the group that received the Desalted Salicornia Powder (DSP) was kept at a significantly low level compared to the obesity control group (p ⁇ 0.001), while the body weight of the positive control group that received Garcinia Extract (GE) was also significantly low (p ⁇ 0.05).
- the body weight of rats in the group that received the non-desalted Salicornia Powder (SP) was found to be significantly high compared to the normal control group, in which obesity was not induced (p ⁇ 0.01), while being reduced compared to that of the obesity-induced control group.
- the body weight of rats in the obesity control group, the group administered with 200 mg/kg of the non-desalted Salicornia Powder (SP) and the positive control group was significantly high compared to the normal control group (p ⁇ 0.01 or p ⁇ 0.05), whereas the body weight of rats in the group administered with 200 mg/kg of Desalted Salicornia Powder (DSP) and the positive control group was significantly low compared to that of the obesity-induced control group (p ⁇ 0.001 and p ⁇ 0.01, respectively).
- the Desalted Salicornia Powder exhibited the most excellent body-weight-reducing effect, which was statistically significantly higher than that of the positive Garcinia Extract (GE) control (p ⁇ 0.001).
- the non-desalted Salicornia Powder showed a slight decrease in body weight in the obese rats, but this decrease was remarkably lower than that of DSP.
- the desalted Salicornia powder from which sodium chloride was removed and which was enriched in dietary fiber and functional compounds, may be a functional material highly effective for suppressing obesity.
- TEST EXAMPLE 4-2 Evaluation for body-fat-reducing effect of the S. europaea-derived desalted nutritional composition in SD rats having obesity induced by high-fat diet
- ALT alanine transaminase
- AST aspartate transaminase
- TC total cholesterol
- TG triglyceride
- HDL high density lipoprotein
- LDL low density lipoprotein
- AI atherosclerosis index
- AST levels of the obesity control group (HFD) induced with the high-fat diet (HFD) and the HFD+SP200 group, which was administered with HFD plus 200 mg/kg of the non-desalted Salicornia Powder (SP) were significantly higher than that of the normal control group (NC) (p ⁇ 0.01 and p ⁇ 0.001).
- the HFD+DSP200 group, which was administered with HFD plus 200 mg/kg of the Desalted Salicornia Powder (DSP) showed a significantly low AST level compared to the HFD group (p ⁇ 0.01).
- the HFD group, the HFD+SP200 group and the positive control group (HFD+GE200) showed a significant increase compared to the normal control group (NC) (p ⁇ 0.001, p ⁇ 0.01 and p ⁇ 0.05), while the HFD+DSP200 group and the positive control group (HFD+GE200) exhibited a significant decrease compared to the HFD group (p ⁇ 0.001 and p ⁇ 0.01).
- the HFD group, the HFD+SP200 group and the positive control group (HFD+GE200) showed a significant increase compared to the normal control group (NC) (p ⁇ 0.001 and p ⁇ 0.01). This tendency was also similarly observed in VLDL and ALT levels.
- DSP Desalted Salicornia Powder
- the total abdominal fat volume was significantly increased in the high-fat-diet-induced obesity control group (HFD) and the HFD+SP200 group administered with the non-desalted Salicornia Powder (SP), compared with the normal control group (NC) (p ⁇ 0.01 and p ⁇ 0.05), whereas it was significantly reduced in the HFD+DSP200 group administered with the Desalted Salicornia Powder (DSP) and the positive control group (HFD+GE200) compared with the HFD group (p ⁇ 0.01 and p ⁇ 0.05) (FIG. 12A and 12B).
- the visceral fat volume was found to be significantly increased in the HFD group, the HFD+SP200 group and the positive control group (HFD+GE200) compared with the normal control group (NC) (p ⁇ 0.01), whereas it was significantly reduced in the HFD+DSP200 group compared with the HFD group and the HFD+SP200 group (p ⁇ 0.01).
- HFD+GE200 positive control group
- NC normal control group
- the subcutaneous fat volume was found to be significantly increased in the HFD group and the HFD+SP200 group compared with the normal control group (NC) (p ⁇ 0.01 and p ⁇ 0.05), whereas it was significantly reduced in the HFD+DSP200 group and the positive control group (HFD+GE200) compared with the HFD group (p ⁇ 0.01 and p ⁇ 0.05).
- the desalted Salicornia powder was found to have a remarkably excellent effect of reducing body weight and suppressing body fat accumulation compared with the non-desalted Salicornia powder (SP), and this effect was higher than that of the positive control Garcinia Extract (GE).
- TEST EXAMPLE 5 Determination of a marker compound effective in suppressing adipocyte differentiation in the S. europaea-derived desalted nutritional composition
- Compound 1 a major peak compound (Compound 1) was observed near a retention time of 11.3 min.
- the UV spectrum of this peak component ( ⁇ max: 218-220, 240, 285-290sh, 325) showed the typical phenylpropanoid phenolic acid nature.
- the HPLC retention time and the UV spectrum were compared with standards of several kinds of phenylpropanoid phenolic acids (Sigma Co., USA).
- Compound 1 was identified to be trans-ferulic acid (FIG. 13B).
- trans-ferulic acid was purified by preparative high performance liquid chromatography (YMC-HPLC, Japan), and used in an experiment with 3T3-L1 cells.
- the analytical HPLC system used in this experiment was a model 1260 (Infinity, Agilent, USA) equipped with Zorbax Eclipse C18 column (5 ⁇ m, 4.5 ⁇ 250 mm, Agilent) and 1200 DAD detector.
- a model Multiple Preparative HPLC (LC-forte/R, YMC, Japan) equipped with a prep column (Triart C18, 20 mm ⁇ 150 mm, 5 ⁇ m, YMC, Japan).
- Preparative HPLC was performed under a gradient condition of methanol and triple-distilled water as a mobile phase at a flow rate of 15 ml/min using an YMC UV-3400 UV detector set at three wavelengths, namely 210, 254 and 320 nm, and four fractions were purified.
- the major peak (compound 3) was found to be trans-ferulic acid, and 230 mg was finally obtained.
- the other three peaks 1, 2 and 4 were found to be caffeic acid, p -coumaric acid and isorhamnetin-3- ⁇ -D-glucoside, respectively.
- the major marker component of the desalted Salicornia powder was evaluated for its inhibitory effect on adipocyte differentiation using 3T3-L1 cells as an in-vitro model of adipocyte differentiation.
- 3T3-L1 preadipocytes were checked for contamination every eight hours to improve confidence in the experiment.
- Primary preadipocytes were propagated, and were then induced to differentiate with a culture medium containing 3-isobutyl-1-methylxanthine (IBMX), dexamethasone and insulin. During the differentiation induction, the medium was changed twice every three days.
- IBMX 3-isobutyl-1-methylxanthine
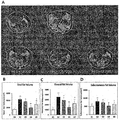
- 3T3-L1 cells were subjected to Oil Red O staining to detect the presence of intracellular lipid droplets.
- the medium was discarded from each well, and cells were fixed with 4% paraformaldehyde. Subsequently, cells were washed with 100% 1,2-propanediol dehydration solution for 5 min and then stained with Oil Red O stain solution. After Oil Red O staining, 85% 1,2-propanediol stain differential solution was added to each well for cell washing. Finally, distilled water was added to each well to prevent stained cells from drying, and cells were observed under a microscope to determine lipid accumulation.
- FIG. 14A shows the results of Oil Red O staining to determine the formation of lipid droplets during differentiation of 3T3-L1 preadipocytes to adipocytes so as to investigate the inhibitory effect of trans-ferulic acid (TFA) on adipocyte differentiation.
- TFA trans-ferulic acid
- TFA intracellular triglyceride formation was investigated.
- TFA reduced intracellular triglyceride formation in a dosage-dependent manner at various concentrations, namely 1, 2, 5 and 10 ⁇ M.
- concentrations of 5 ⁇ M and 10 ⁇ M of TFA triglyceride formation was remarkably significantly inhibited compared to a differentiation-induced control (MDI) ( ### p ⁇ 0.001).
- MDI differentiation-induced control
- PPAR ⁇ , FAS, SREBP-1 and C/EBP ⁇ are transcription factors involved in lipid metabolism, and are produced when 3T3-L1 preadipocytes differentiate into mature adipocytes. C/EBP ⁇ and PPAR ⁇ cooperate to accelerate adipogenesis. When preadipocytes proliferate to an early differentiation stage, C/EBP ⁇ is induced and stimulates PPAR ⁇ to induce a mature stage of differentiation. PPAR ⁇ is present mainly in adipose tissue and regulates overall lipid formation, and its capacity in adipocyte differentiation is much better than other transcription factors.
- FAS can be used as a marker gene when adipocyte differentiation reaches a late stage, and it is a lipid synthesis enzyme involved in lipid metabolism.
- FAS is most strongly expressed in adipose tissues, and is the last factor of adipose differentiation. FAS is thus a representative marker for anti-obesity effect, and is induced by SREBP-1, which is a transcription factor at an earlier stage.
- TFA trans-ferulic acid
- the Desalted Salicornia Powder (DSP) containing TFA as an effective component may effectively reduce body fat by inhibiting adipocyte differentiation and lipid droplet formation and may eventually lead to a decrease in body weight, and thus has the potential to be applied as a functional food and feedstuff for preventing and/or treating obesity.
- the halophyte-derived functionally reinforced desalted nutritional composition can be developed as a pharmaceutical composition and functional food and feedstuff for combatting obesity and for reducing body fat.
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Nutrition Science (AREA)
- Engineering & Computer Science (AREA)
- Food Science & Technology (AREA)
- Polymers & Plastics (AREA)
- Mycology (AREA)
- Inorganic Chemistry (AREA)
- Botany (AREA)
- Coloring Foods And Improving Nutritive Qualities (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
La présente invention concerne une composition nutritionnelle dessalée fonctionnellement renforcée, un extrait dessalé et un substitut de sel extrait à l'eau froide, qui sont dérivés d'halophytes qui poussent dans des régions côtières dans des conditions hautement salines et conservent ainsi des concentrations de sel élevées, ainsi que l'utilisation de la composition nutritionnelle dessalée pour lutter contre l'obésité. Plus précisément, la présente invention concerne une composition nutritionnelle dessalée fonctionnellement renforcée, un extrait dessalé et un substitut de sel extrait à l'eau froide dérivés d'halophytes qui poussent des environnements extrêmes à forte salinité et sont soumis à un stress salin important, les halophytes étant dessalés par un procédé d'extraction à l'eau froide à basse température sur la base de la différence de solubilité dans l'eau des sels avec un changement de température pour permettre uniquement le retrait sélectif du chlorure de sodium, et la composition ayant ainsi une teneur en sodium réduite ainsi qu'une teneur accrue en minéraux utiles tels que le potassium, ainsi qu'en nutriments et en substances physiologiquement actives, qui sont naturellement contenus dans des halophytes.
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201780027825.4A CN109152404B (zh) | 2016-05-04 | 2017-01-26 | 来自盐生植物的功能增强的脱盐营养组合物及其制备方法 |
| US16/099,070 US20190142046A1 (en) | 2016-05-04 | 2017-01-26 | Functionally reinforced desalted nutritional compositions from halophytes and preparation method thereof |
| EP17792789.4A EP3451859A4 (fr) | 2016-05-04 | 2017-01-26 | Compositions nutritionnelles dessalées fonctionnellement renforcées dérivées d'halophytes et leur procédé de préparation |
| JP2018558181A JP6876724B2 (ja) | 2016-05-04 | 2017-01-26 | 塩生植物由来の機能性が強化した脱塩栄養組成物とその製造方法 |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR20160055486 | 2016-05-04 | ||
| KR10-2016-0055486 | 2016-05-04 | ||
| KR10-2016-0183473 | 2016-12-30 | ||
| KR1020160183473A KR101922245B1 (ko) | 2016-05-04 | 2016-12-30 | 염생식물 유래의 기능성이 강화된 탈염 영양 조성물과 그 제조방법 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2017191886A1 true WO2017191886A1 (fr) | 2017-11-09 |
Family
ID=60203077
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/KR2017/000949 Ceased WO2017191886A1 (fr) | 2016-05-04 | 2017-01-26 | Compositions nutritionnelles dessalées fonctionnellement renforcées dérivées d'halophytes et leur procédé de préparation |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2017191886A1 (fr) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2019229379A1 (fr) * | 2018-06-01 | 2019-12-05 | Universite De Bretagne Occidentale | Complément alimentaire comportant un extrait de plantes halophyte |
Citations (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0906761A2 (fr) * | 1997-10-02 | 1999-04-07 | Archer-Daniels-Midland Company | Procédé de préparation et d'utilisation de produits phytochimiques |
| KR20050122447A (ko) | 2004-06-24 | 2005-12-29 | 학교법인 영남학원 | 역삼투법을 이용한 간장의 농축 및 저염화 방법 |
| US20060024422A1 (en) * | 2004-07-30 | 2006-02-02 | Cumberland Packing Corp. | Salt substitute compositions having N-neohexyl-a-aspartyl-l- phenylalanine methyl ester for modifying flavor and methods of manufacturing the same |
| KR100561103B1 (ko) | 2004-06-24 | 2006-03-15 | 학교법인 영남학원 | 전기투석법에 의한 간장의 저염화 방법 |
| KR20060110023A (ko) | 2005-04-19 | 2006-10-24 | 김용호 | 저염 가공 함초를 이용한 신장 기능 강화 스낵류, 과자류,빵류의 제조법 |
| KR100724705B1 (ko) | 2006-02-09 | 2007-06-04 | 최형식 | 함초추출물을 포함하는 음료용 액상 조성물 |
| KR100784229B1 (ko) | 2007-05-17 | 2007-12-11 | 주식회사 파이토코 | 퉁퉁마디 유래의 소금 대체물 및 그 제조방법 |
| KR101095619B1 (ko) | 2009-04-29 | 2011-12-19 | 전라남도 | 퉁퉁마디 저염화 방법 및 그 저장 방법 |
| KR101102259B1 (ko) | 2009-10-30 | 2012-01-05 | 대한민국 | 주정을 이용한 염장발효식품의 탈염방법 |
| KR101218355B1 (ko) | 2009-04-29 | 2013-01-03 | 전라남도 | 적색 퉁퉁마디로부터 천연 식용색소 베타시아닌의 제조 방법 |
| KR20130058230A (ko) * | 2011-11-25 | 2013-06-04 | 주식회사 태평소금 | 함초차의 제조방법 및 이에 의해 제조된 함초차 |
| KR101287065B1 (ko) | 2010-12-24 | 2013-07-17 | 천사섬함초영농조합법인 | 위생성 및 소화성이 향상된 함초분말의 제조방법 |
| KR101289769B1 (ko) | 2009-03-30 | 2013-07-26 | 모리나가 뉴교 가부시키가이샤 | 탈염유의 제조 방법, 탈염유 |
-
2017
- 2017-01-26 WO PCT/KR2017/000949 patent/WO2017191886A1/fr not_active Ceased
Patent Citations (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0906761A2 (fr) * | 1997-10-02 | 1999-04-07 | Archer-Daniels-Midland Company | Procédé de préparation et d'utilisation de produits phytochimiques |
| KR20050122447A (ko) | 2004-06-24 | 2005-12-29 | 학교법인 영남학원 | 역삼투법을 이용한 간장의 농축 및 저염화 방법 |
| KR100561103B1 (ko) | 2004-06-24 | 2006-03-15 | 학교법인 영남학원 | 전기투석법에 의한 간장의 저염화 방법 |
| US20060024422A1 (en) * | 2004-07-30 | 2006-02-02 | Cumberland Packing Corp. | Salt substitute compositions having N-neohexyl-a-aspartyl-l- phenylalanine methyl ester for modifying flavor and methods of manufacturing the same |
| KR20060110023A (ko) | 2005-04-19 | 2006-10-24 | 김용호 | 저염 가공 함초를 이용한 신장 기능 강화 스낵류, 과자류,빵류의 제조법 |
| KR100724705B1 (ko) | 2006-02-09 | 2007-06-04 | 최형식 | 함초추출물을 포함하는 음료용 액상 조성물 |
| KR100784229B1 (ko) | 2007-05-17 | 2007-12-11 | 주식회사 파이토코 | 퉁퉁마디 유래의 소금 대체물 및 그 제조방법 |
| KR101289769B1 (ko) | 2009-03-30 | 2013-07-26 | 모리나가 뉴교 가부시키가이샤 | 탈염유의 제조 방법, 탈염유 |
| KR101095619B1 (ko) | 2009-04-29 | 2011-12-19 | 전라남도 | 퉁퉁마디 저염화 방법 및 그 저장 방법 |
| KR101218355B1 (ko) | 2009-04-29 | 2013-01-03 | 전라남도 | 적색 퉁퉁마디로부터 천연 식용색소 베타시아닌의 제조 방법 |
| KR101102259B1 (ko) | 2009-10-30 | 2012-01-05 | 대한민국 | 주정을 이용한 염장발효식품의 탈염방법 |
| KR101287065B1 (ko) | 2010-12-24 | 2013-07-17 | 천사섬함초영농조합법인 | 위생성 및 소화성이 향상된 함초분말의 제조방법 |
| KR20130058230A (ko) * | 2011-11-25 | 2013-06-04 | 주식회사 태평소금 | 함초차의 제조방법 및 이에 의해 제조된 함초차 |
Non-Patent Citations (11)
| Title |
|---|
| BLUMENKRANTZ, ANALYTICAL BIOCHEM., vol. 54, 1973, pages 484 - 489 |
| CHEN, J. AGRIC. FOOD CHEM., vol. 47, 1999, pages 2226 - 2228 |
| JOURNAL OF THE SCIENCE OF FOOD AGRICULTURE, vol. 95, 2015, pages 3150 - 3159 |
| KIM, SOOK JA ET AL.: "Effects of insoluble dietary fiber extracted from Salicornia herbacea L. on large intestinal function in rats", KOREAN JOURNAL OF FOOD SCIENCE AND TECHNOLOGY, vol. 46, no. 5, 31 October 2014 (2014-10-31), pages 648 - 654, XP055581449, ISSN: 0367-6293, DOI: 10.9721/KJFST.2014.46.5.648 * |
| KWON, J. LIFE SCIENCE, vol. 14, 2004, pages 509 - 513 |
| OVE, N.COWELL, G.M.TRANUM-JENSER, J. HANSEN, O.WELINDER, K.G., J. BIOL. CHEM., vol. 261, 1986, pages 12306 - 12309 |
| PATEL ET AL.: "Salicornia: evaluating the halophytic extremophile as a food and a pharmaceutical candidate", 3 BIOTECH, vol. 6, no. 104, 18 April 2016 (2016-04-18), pages 1 - 10, XP055441982 * |
| RUDIGER, W.SCHOCH, S.: "Plant Pigments", 1988, ACADEMIC PRESS, article "Chlorophylls" |
| RYU ET AL., J. LIFE SCIENCE, vol. 20, 2010, pages 922 - 928 |
| See also references of EP3451859A4 |
| SOHN ET AL., KOR. J. PHARMACOGN, vol. 35, 2004, pages 52 - 61 |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2019229379A1 (fr) * | 2018-06-01 | 2019-12-05 | Universite De Bretagne Occidentale | Complément alimentaire comportant un extrait de plantes halophyte |
| FR3081683A1 (fr) * | 2018-06-01 | 2019-12-06 | Universite De Bretagne Occidentale | Complement alimentaire comportant un extrait de plantes halophyte |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP3451859A1 (fr) | Compositions nutritionnelles dessalées fonctionnellement renforcées dérivées d'halophytes et leur procédé de préparation | |
| Xie et al. | Protective effect of flavonoids from Cyclocarya paliurus leaves against carbon tetrachloride-induced acute liver injury in mice | |
| CN103467568B (zh) | 一种金乌贼蛋白抗氧化肽及其制备方法和用途 | |
| WO2015183027A1 (fr) | Composition pour améliorer la fonction hépatique, contenant un extrait de dendropanax morbifera | |
| WO2019135637A1 (fr) | Procédé de préparation d'un extrait de feuille de gynostemma pentaphyllum contenant une quantité accrue de saponine efficace de faible masse moléculaire et une quantité réduite de benzopyrène, et extrait de feuille de gynostemma pentaphyllum préparé selon ledit procédé | |
| AU2012338742B2 (en) | Composition comprising chicory extract | |
| WO2018062777A1 (fr) | Composition d'amélioration de la force musculaire et de la masse musculaire comprenant un peptide de jus de soja dérivé de soja germé traité avec de l'eau de tangerine non mûre | |
| WO2017043914A2 (fr) | Composition pharmaceutique comprenant salicornia spp. en tant que principe actif pour la prévention ou le traitement de la thrombose, et aliment fonctionnel de santé la comprenant | |
| KR100865022B1 (ko) | 피부 노화를 억제하는 화장료 조성물 | |
| WO2017191886A1 (fr) | Compositions nutritionnelles dessalées fonctionnellement renforcées dérivées d'halophytes et leur procédé de préparation | |
| KR102011349B1 (ko) | 항고혈압 활성이 개선된 혼합 씨앗즙 | |
| Wei et al. | Characteristics of pressurized hot water extract from abalone muscle and the antioxidant ability during simulated digestion in vitro | |
| CN110693025A (zh) | 一种多功能成分协同降血糖的组合物及其应用 | |
| RU2399379C2 (ru) | Экстракт salix, его использование и композиции, его содержащие | |
| WO2018044024A1 (fr) | Composition pour soulager les gueules de bois, contenant de l'eau de mer magmatique dessalée et un extrait de bois, et procédé de préparation de substances actives de celle-ci | |
| CN111450173A (zh) | 绞股蓝协同白茶预防2型糖尿病的配方制备方法及用途 | |
| KR101071044B1 (ko) | 황기, 복령 및 적소두의 혼합 생약 추출물을 유효성분으로 함유하는 비만증의 예방 및 치료용 조성물 | |
| WO2010090423A2 (fr) | Composition comprenant un extrait de substance médicamenteuse brute combinée pour la prévention et le traitement d'hyperlipidémie et d'hyperlipidémie diabétique | |
| WO2023003204A1 (fr) | Composition anti-obésité contenant un extrait de rosa davurica en tant que principe actif | |
| KR20110022472A (ko) | 해조류 추출물을 유효성분으로 함유하는 고혈압의 예방 및 치료용 조성물 | |
| WO2016204493A1 (fr) | Nouveau composé (ks 513) isolé de pseudolysimachion rotundum var. subintegrum, la composition le comprenant comme ingrédient actif pour la prévention ou le traitement de l'allergie, d'une maladie inflammatoire, de l'asthme ou d'une maladie pulmonaire obstructive chronique et son utilisation | |
| CN105646656A (zh) | 灰星鲨软骨蛋白抗氧化肽及其用途 | |
| KR20170142725A (ko) | 실크 단백질 및 하수오 추출물을 유효성분으로 포함하는 갱년기 증상 완화용 조성물 | |
| KR101886314B1 (ko) | 도화 및 마름 복합 추출물을 포함하는 비만 및 대사성 질환의 예방, 개선 또는 치료용 조성물 | |
| KR102453180B1 (ko) | 쥐눈이콩 청국장 및 장어 점액질을 포함하는 비만 및 피부 주름 개선용 효소 식품 조성물 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| ENP | Entry into the national phase |
Ref document number: 2018558181 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 17792789 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2017792789 Country of ref document: EP Effective date: 20181204 |