WO2020163964A1 - The use of protoporphyrin ix derivatives to improve the health of plants - Google Patents
The use of protoporphyrin ix derivatives to improve the health of plants Download PDFInfo
- Publication number
- WO2020163964A1 WO2020163964A1 PCT/CA2020/050197 CA2020050197W WO2020163964A1 WO 2020163964 A1 WO2020163964 A1 WO 2020163964A1 CA 2020050197 W CA2020050197 W CA 2020050197W WO 2020163964 A1 WO2020163964 A1 WO 2020163964A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- compound
- acceptable salt
- agriculturally acceptable
- substituted
- alkyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 *C(C1=*2*3(*45)*(C(C(*)=C6C(F)(F)F)=C7)C6=C1)=C(*)C2=CC4=C(*)C(CCC(N)=O)=C5C=C1*3=C7C(*)=C1CCC(N)=O Chemical compound *C(C1=*2*3(*45)*(C(C(*)=C6C(F)(F)F)=C7)C6=C1)=C(*)C2=CC4=C(*)C(CCC(N)=O)=C5C=C1*3=C7C(*)=C1CCC(N)=O 0.000 description 5
- DUMCQJIQTJATBB-REDNZTQVSA-N Cc1c(/C=C2\N=C(/C=C(/C(C=C)=C3C)\N/C3=C\C(C(C)=C3CCC(O)=O)=N/C3=C3)C(C)=C2C=C)[nH]c3c1CCC(NCC[N](C)(C)C)=O Chemical compound Cc1c(/C=C2\N=C(/C=C(/C(C=C)=C3C)\N/C3=C\C(C(C)=C3CCC(O)=O)=N/C3=C3)C(C)=C2C=C)[nH]c3c1CCC(NCC[N](C)(C)C)=O DUMCQJIQTJATBB-REDNZTQVSA-N 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N55/00—Biocides, pest repellants or attractants, or plant growth regulators, containing organic compounds containing elements other than carbon, hydrogen, halogen, oxygen, nitrogen and sulfur
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N55/00—Biocides, pest repellants or attractants, or plant growth regulators, containing organic compounds containing elements other than carbon, hydrogen, halogen, oxygen, nitrogen and sulfur
- A01N55/02—Biocides, pest repellants or attractants, or plant growth regulators, containing organic compounds containing elements other than carbon, hydrogen, halogen, oxygen, nitrogen and sulfur containing metal atoms
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/90—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having two or more relevant hetero rings, condensed among themselves or with a common carbocyclic ring system
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01P—BIOCIDAL, PEST REPELLANT, PEST ATTRACTANT OR PLANT GROWTH REGULATORY ACTIVITY OF CHEMICAL COMPOUNDS OR PREPARATIONS
- A01P1/00—Disinfectants; Antimicrobial compounds or mixtures thereof
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01P—BIOCIDAL, PEST REPELLANT, PEST ATTRACTANT OR PLANT GROWTH REGULATORY ACTIVITY OF CHEMICAL COMPOUNDS OR PREPARATIONS
- A01P3/00—Fungicides
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01P—BIOCIDAL, PEST REPELLANT, PEST ATTRACTANT OR PLANT GROWTH REGULATORY ACTIVITY OF CHEMICAL COMPOUNDS OR PREPARATIONS
- A01P7/00—Arthropodicides
- A01P7/04—Insecticides
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D487/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00
- C07D487/22—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00 in which the condensed system contains four or more hetero rings
Definitions
- the technical field generally relates to photosensitizer tetrapyrrole compounds and their use for promoting the health of plants. More particularly, the technical field relates to modified protoporphyrin IX (PP IX) compounds and use thereof for photodynamic inhibition of microbial pathogens, such as fungal or bacterial pathogens, in plants.
- the modified PP IX compounds can also be used for increasing abiotic stress resistance or tolerance in plants and/or as insecticides to protect plants from plant pests.
- Photodynamic inhibition of microbial pathogens involves exposing a photosensitive agent to light in order to generate reactive oxygen species (ROS), such as singlet oxygen, which can have detrimental effects on the microbial pathogens.
- ROS reactive oxygen species
- Z 1 and Z 2 are each independently OR 1 or NR 2 R 3 ;
- each R 1 , R 2 and R 3 is independently H, alkyl, substituted alkyl, aryl, substituted aryl, alkenyl, substituted alkenyl, alkynyl, or substituted alkynyl, wherein:
- each R a , R B , R c , R d , R e and R f is, independently, H, alkyl, substituted alkyl, aryl, substituted aryl, alkenyl, substituted alkenyl, alkynyl or substituted alkynyl;
- : : ::: :: is a single bond or a double bond
- M is 2H or a metal species
- each X is independently a halogen: F, Cl, Br or I;
- each R B is independently H, alkyl, aryl, arylalkyl, a heterocycle, an alkyloxy group such as poly(ethyleneoxy), PEG or poly(methyleneoxy), a capped poly(ethyleneoxy), capped PEG or capped polymethyleneoxy, or a protecting group;
- the capped poly(ethyleneoxy), capped PEG and capped poly(methyleneoxy) groups being each independently capped with alkyl, aryl, arylalkyl, alkenyl, alkynyl, CO(alkyl), CO(aryl), CO(arylalkyl), CO(alkenyl) or CO(alkynyl);
- each R c is independently alkyl, aryl, arylalkyl, O(alkyl), O(aryl), O(arylalkyl), or O(tri- substituted silyl); each tri-substituted silyl is independently substituted with three functional groups selected from alkyl, alkenyl, alkynyl, aryl and arylalkyl; and
- each Aik is independently alkylene, alkenylene, or alkynylene.
- one of Z 1 and Z 2 is OR 1 ;
- Z 1 and Z 2 is NR 2 R 3 , NR 2 -(CH 2 ) n -NR 4 R 5 , NR 2 -(CH 2 ) n -N + R 4 R 5 R 6 Y-,
- each R 1 and R 2 is, independently, H, alkyl, substituted alkyl, aryl, substituted aryl, alkenyl, substituted alkenyl, alkynyl or substituted alkynyl;
- R 3 is alkyl, substituted alkyl, aryl, substituted aryl, alkenyl, substituted alkenyl, alkynyl or substituted alkynyl;
- each R 4 , R 6 , R 8 , R 9 , R 10 and R 11 is, independently, H, alkyl, substituted alkyl, aryl, substituted aryl, alkenyl, substituted alkenyl, alkynyl, substituted alkynyl or -(CH 2 ) q - (CH 2 CH 2 O) m -R 13 ;
- R 5 is alkyl, substituted alkyl, aryl, substituted aryl, alkenyl, substituted alkenyl, alkynyl, substituted alkynyl or -(CH 2 ) q -(CH 2 CH 2 O)m-R 13 ;
- R 7 is alkyl, O(alkyl) or O(tri-substituted silyl);
- R 13 is H, alkyl, substituted alkyl, aryl, substituted aryl, alkenyl, substituted alkenyl, alkynyl, substituted alkynyl, CO(alkyl), CO(substituted alkyl), CO(alkenyl), CO(substituted alkenyl), CO(alkynyl) or CO(substituted alkynyl);
- W + is an agriculturally acceptable cation
- Y- is an agriculturally acceptable anion
- n is an integer selected from 1 to 16;
- p is an integer selected from 1 to 16;
- n is an integer selected from 1 to 100;
- q is an integer selected from 0 to 16;
- each R a , R B , R c , R d , R e and R f is, independently, H, alkyl, substituted alkyl, aryl, substituted aryl, alkenyl, substituted alkenyl, alkynyl or substituted alkynyl;
- M is 2H or a metal species
- each substituted alkyl, substituted aryl, substituted alkenyl and substituted alkynyl groups are, independently, substituted with one or more OH, F, Cl, Br, I, CN and N 3 .
- one of Z 1 and Z 2 is NR 2 R 3 ;
- each R 1 and R 2 is, independently, H, alkyl or substituted alkyl
- R 3 is alkyl or substituted alkyl
- each R a , R B , R c , R d , R e and R f is, independently, H, alkyl, substituted alkyl, alkenyl, substituted alkenyl, alkynyl or substituted alkynyl;
- M is 2H or a metal species
- substituted alkyl, substituted alkenyl and substituted alkynyl groups are, independently, substituted with one or more OH, F, Cl, Br, I, CN and N3.
- Z 1 and Z 2 is NR 2 -(CH 2 ) n -NR 4 R 5 or O-(CH 2 ) n -NR 4 R 5 ;
- Z 1 NR 2 -(CH 2 ) n -NR 4 R 5 or O-(CH 2 ) n -NR 4 R 5 ;
- R 5 is alkyl, substituted alkyl or -(CH 2 ) p -NR 9 R 10 ;
- each R 1 , R 2 , R 4 , R 9 and R 10 is, independently, H, alkyl or substituted alkyl;
- n is an integer selected from 1 to 16;
- p is an integer selected from 1 to 16;
- each R a , R B , R c , R d , R e and R f is, independently, H, alkyl, substituted alkyl, alkenyl, substituted alkenyl, alkynyl or substituted alkynyl;
- M is 2H or a metal species
- substituted alkyl, substituted alkenyl and substituted alkynyl groups are, independently, substituted with one or more OH, F, Cl, Br, I, CN and N3.
- Z 1 and Z 2 is NR 2 -(CH 2 ) n -Si(R 7 ) 3 , O-(CH 2 ) n -Si(R 7 ) 3 , NR 2 -(CH 2 ) n -SR 8 or O- (CH 2 ) n -SR 8 ; and
- each R 1 and R 2 is, independently, H, alkyl or substituted alkyl
- R 7 is alkyl, O(alkyl) or O(trisubstituted silyl);
- R 8 is H, alkyl, substituted alkyl or -(CH 2 ) q -(CH 2 CH 2 O) m -R 13 ;
- R 13 is H, alkyl, substituted alkyl, alkenyl, substituted alkenyl, alkynyl, substituted alkynyl, CO(alkyl), CO(substituted alkyl), CO(alkenyl), CO(substituted alkenyl), CO(alkynyl) or CO(substituted alkynyl);
- n is an integer selected from 1 to 16;
- n is an integer selected from 1 to 100;
- q is an integer selected from 0 to 16;
- each R a , R B , R c , R d , R e and R f is, independently, H, alkyl, substituted alkyl, alkenyl, substituted alkenyl, alkynyl or substituted alkynyl;
- M is 2H or a metal species
- substituted alkyl, substituted alkenyl and substituted alkynyl groups are, independently, substituted with one or more OH, F, Cl, Br, I, CN and N 3 .
- each R 1 and R 2 is, independently, H, alkyl or substituted alkyl
- n is an integer selected from 1 to 16;
- W + is an agriculturally acceptable cation
- each R a , R B , R c , R d , R e and R f is, independently, H, alkyl, substituted alkyl, alkenyl, substituted alkenyl, alkynyl or substituted alkynyl;
- M is 2H or a metal species
- substituted alkyl, substituted alkenyl and substituted alkynyl groups are, independently, substituted with one or more OH, F, Cl, Br, I, CN and N3.
- Z 1 and Z 2 is NR 2 -(CH 2 ) n -NR 4 R 5 R 6+ Y- or O-(CH 2 ) n -NR 4 R 5 R 6+ Y-; and the other one of Z 1 and Z 2 is OR 1 ;
- Z 1 N R 2 -(CH 2 ) n - N R 4 R 5 R 6+ Y- or O-(CH 2 ) n -NR 4 R 5 R 6+ Y-;
- each R 1 and R 2 is, independently, H, alkyl or substituted alkyl
- each R 4 , R 5 and R 6 is, independently, alkyl or substituted alkyl
- n is an integer selected from 1 to 16;
- Y- is an agriculturally acceptable anion
- each R a , R B , R c , R d , R e and R f is, independently, H, alkyl, substituted alkyl, alkenyl, substituted alkenyl, alkynyl or substituted alkynyl;
- M is 2H or a metal species
- substituted alkyl, substituted alkenyl and substituted alkynyl groups are, independently, substituted with one or more OH, F, Cl, Br, I, CN and N3.
- Z 1 and Z 2 is NR 2 -(CH 2 CH 2 O) m -R 13 or O-(CH 2 CH 2 O) m -R 13 ;
- Z 1 NR 2 -(CH 2 CH 2 O) m -R 13 or O-(CH 2 CH 2 O) m -R 13 ;
- each R 1 and R 2 is, independently, H, alkyl or substituted alkyl
- R 13 is H, alkyl, substituted alkyl, alkenyl, substituted alkenyl, alkynyl, substituted alkynyl, CO(alkyl), CO(substituted alkyl), CO(alkenyl), CO(substituted alkenyl), CO(alkynyl) or CO(substituted alkynyl);
- n is an integer selected from 1 to 100;
- each R a , R B , R c , R d , R e and R f is, independently, H, alkyl, substituted alkyl, alkenyl, substituted alkenyl, alkynyl or substituted alkynyl;
- M is 2H or a metal species
- substituted alkyl, substituted alkenyl and substituted alkynyl groups are, independently, substituted with one or more OH, F, Cl, Br, I, CN and N3.
- Z 1 and Z 2 is a natural amino acid attached to the compound by its amino group bonded to the alpha carbon;
- Z 1 is a natural amino acid attached to the compound by its amino group bonded to the alpha carbon
- each R 1 and R 2 is, independently, H, alkyl or substituted alkyl
- each R a , R B , R c , R d , R e and R f is, independently, H, alkyl, substituted alkyl, alkenyl, substituted alkenyl, alkynyl or substituted alkynyl;
- M is 2H or a metal species
- substituted alkyl, substituted alkenyl and substituted alkynyl groups are, independently, substituted with one or more OH, F, Cl, Br, I, CN and N3.
- compositions for use in promoting the health of a plant comprising at least one compound as defined herein, or an agriculturally acceptable salt thereof, and a carrier fluid.
- the present description also provides a method for promoting the health of a plant, comprising: applying to the plant a compound as defined herein or an agriculturally acceptable salt thereof, or a composition as defined herein; and exposing the plant to light. Promoting the health of the plant can include at least one of preventing or inhibiting growth of a microbial pathogen of the plant (e.g., a fungus or a bacterial pathogen), increasing resistance of the plant to one or more abiotic stress, and controlling a pest of the plant (e.g., a noxious insect or corresponding larva).
- a microbial pathogen of the plant e.g., a fungus or a bacterial pathogen
- a pest of the plant e.g., a noxious insect or corresponding larva.
- Some microbial pathogens such as Gram-negative bacteria and certain types of fungi have a cellular membrane that is difficult to penetrate. More specifically, these microbial pathogens sometimes have an impermeable outer cell membrane that contains endotoxins and can block small molecules such as antibiotics, dyes and detergents, thereby protecting the sensitive inner membrane and cell wall. It can therefore be challenging to use photodynamic therapy to inhibit growth of certain microbial pathogens in plants because the photosensitizer compounds tend to not achieve good penetration inside the cell wall. It can also be challenging to increase resistance of plants to damage caused by abiotic stresses.
- photodynamic inhibition of microbial pathogens that are present on plants can be achieved by applying a photosensitizer compound.
- the photosensitizer compound reacts to light by generating reactive oxygen species (ROS).
- ROS reactive oxygen species
- a photosensitizer compound can be used to increase resistance of plants to damage caused by one or more abiotic stress.
- Some of the compounds of the present description are photosensitizer compounds that can be derived from the protoporphyrin IX (hereinafter“PP IX”) scaffold.
- the compounds derived from PP IX can also be referred to as“modified PP IX”.
- Some of the compounds of the present description are photosensitizer compounds that have a scaffold similar to that of PP IX, but that are not necessarily derived from PP IX. In some scenarios, these compounds can be used to improve the health of plants.
- the compounds of the present description can be applied to plants to photodynamically inhibit microbial pathogens on plants, to increase resistance of plants to damage caused by one or more abiotic stress and/or to be used as insecticides.
- the phrase“a compound of Formula I” means a compound of Formula I or an agriculturally acceptable salt thereof.
- the phrase“a compound of Formula (number)” means a compound of that formula and salts thereof, and optionally agriculturally acceptable salts thereof.
- Alkyl means a hydrocarbon containing primary, secondary, tertiary or cyclic carbon atoms.
- an alkyl group can have 1 to 20 carbon atoms (/.e, C1-C20 alkyl), 1 to 8 carbon atoms (/.e., C 1 -C 8 alkyl),
- alkyl groups include, but are not limited to, methyl (Me, -CH 3 ), ethyl (Et, -CH 2 CH 3 ), 1 -propyl (n-Pr, n-propyl, -CH 2 CH 2 CH 3 ), 2-propyl (/- Pr, /-propyl, -CH(CH 3 ) 2 ), 1 -butyl (n-Bu, n- butyl, -CH 2 CH 2 CH 2 CH 3 ), 2-methyl-1-propyl (/- Bu, /-butyl, -CH 2 CH(CH 3 ) 2 ), 2-butyl (s-Bu, s- butyl, -CH(CH 3 )CH 2 CH 3 ), 2-methyl-2-propyl (t-Bu, t-But
- alkenyl means a hydrocarbon containing primary, secondary, tertiary or cyclic carbon atoms with at least one site of unsaturation, /.e. a carbon- carbon sp 2 double bond.
- an alkenyl group can have
- Alkynyl means a hydrocarbon containing primary, secondary, tertiary or cyclic carbon atoms with at least one site of unsaturation, /.e. a carbon- carbon, sp triple bond.
- an alkynyl group can have 2 to 20 carbon atoms (/.e., C 2 -C 20 alkynyl), 2 to 8 carbon atoms (/.e., C 2 -C 8 alkynyl), 2 to 6 carbon atoms (/.e., C 2 -C 6 alkynyl) or 2 to 4 carbon atoms (i.e. , C 2 -C 4 alkynyl).
- suitable alkynyl groups include, but are not limited to, acetylenic (-CoCH) and propargyl (-CH 2 CoCH).
- alkyl portion of an O(Alkyl) group can have 1 to 20 carbon atoms (i.e, C 1 -C 20 alkyl), 1 to 8 carbon atoms (i.e., C 1 C 8 alkyl), 1 to 6 carbon atoms (/.e., C 1 -C 6 alkyl) or 1 to 4 carbon atoms (i.e., C 1 -C 4 alkyl).
- Alkoxy or O(Alkyl) groups include, but are not limited to, methoxy (-OCH 3 or -OMe), ethoxy (-OCH 2 CH 3 or -OEt) and t-Bu toxy (-O-C(CH 3 ) 3 or -OtBu).
- ethoxy ethoxy
- t-Bu toxy -O-C(CH 3 ) 3 or -OtBu
- O(alkenyl)”, O(alkynyl)” and the corresponding substituted groups will be understood by a person skilled in the art.
- suitable Acyl groups include, but are not limited to, formyl (i.e., a carboxyaldehyde group), acetyl, trifluoroacetyl, propionyl, and butanoyl.
- Alkylene means a saturated, branched or straight chain or cyclic hydrocarbon radical having two monovalent radical centers derived by the removal of two hydrogen atoms from the same or two different carbon atoms of a parent alkane.
- an alkylene group can have 1 to 20 carbon atoms, 1 to 10 carbon atoms, 1 to 6 carbon atoms or 1 to 4 carbon atoms.
- Typical alkylene radicals include, but are not limited to, methylene (-CH 2 -), 1 , 1-ethyl (-CH(CH3)-), 1 ,2-ethyl (-CH 2 CH 2 -), 1 , 1-propyl (-CH(CH 2 CH 3 )-), 1 ,2-propyl (-CH 2 CH(CH 3 )-), 1 ,3-propyl (-CH 2 CH 2 CH 2 -) and 1 ,4- butyl (-CH 2 CH 2 CH 2 CH 2 -).
- alkenylene means an unsaturated, branched or straight chain or cyclic hydrocarbon radical having two monovalent radical centers derived by the removal of two hydrogen atoms from the same or two different carbon atoms of a parent alkene.
- alkenylene group can have 1 to 20 carbon atoms, 1 to 10 carbon atoms, 1 to 6 carbon atoms or 1 to 4 carbon atoms.
- Alkynylene means an unsaturated, branched or straight chain or cyclic hydrocarbon radical having two monovalent radical centers derived by the removal of two hydrogen atoms from the same or two different carbon atoms of a parent alkyne.
- an alkynylene group can have 2 to 20 carbon atoms, 2 to 10 carbon atoms, 2 to 6 carbon atoms or 2 to 4 carbon atoms.
- Typical alkynylene radicals include, but are not limited to, acetylene (-CoC-), propargyl (-CH 2 CoC-), and 4- pentynyl (-CH 2 CH 2 CH 2 CoC-).
- Aryl means an aromatic hydrocarbon radical derived by the removal of one hydrogen atom from a single carbon atom of a parent aromatic ring system.
- an aryl group can have 6 to 20 carbon atoms, 6 to 14 carbon atoms, or 6 to 10 carbon atoms.
- Typical aryl groups include, but are not limited to, radicals derived from benzene (e.g., phenyl), substituted benzene, naphthalene, anthracene and biphenyl.
- Arylalkyl means an acyclic alkyl radical in which one of the hydrogen atoms bonded to a carbon atom, typically a terminal or sp 3 carbon atom, is replaced with an aryl radical.
- Typical arylalkyl groups include, but are not limited to, benzyl, 2-phenylethan-1-yl, naphthylmethyl, 2-naphthylethan-1-yl, naphthobenzyl, 2-naphthophenylethan-1-yl and the like.
- the arylalkyl group can include 7 to 20 carbon atoms, e.g., the alkyl moiety is 1 to 6 carbon atoms and the aryl moiety is 6 to 14 carbon atoms.
- Arylalkenyl means an acyclic alkenyl radical in which one of the hydrogen atoms bonded to a carbon atom, typically a terminal or sp 3 carbon atom, but also an sp 2 carbon atom, is replaced with an aryl radical.
- the aryl portion of the arylalkenyl can include, for example, any of the aryl groups described herein, and the alkenyl portion of the arylalkenyl can include, for example, any of the alkenyl groups described herein.
- the arylalkenyl group can include 8 to 20 carbon atoms, e.g., the alkenyl moiety is 2 to 6 carbon atoms and the aryl moiety is 6 to 14 carbon atoms.
- the term“Arylalkynyl”, as used herein, means an acyclic alkynyl radical in which one of the hydrogen atoms bonded to a carbon atom, typically a terminal or sp 3 carbon atom, but also an sp carbon atom, is replaced with an aryl radical.
- the aryl portion of the arylalkynyl can include, for example, any of the aryl groups disclosed herein, and the alkynyl portion of the arylalkynyl can include, for example, any of the alkynyl groups disclosed herein.
- the arylalkynyl group can include 8 to 20 carbon atoms, e.g., the alkynyl moiety is 2 to 6 carbon atoms and the aryl moiety is 6 to 14 carbon atoms.
- heterocycle means a group including a covalently closed ring wherein at least one atom forming the ring is a heteroatom.
- heterocyclic rings can be formed by three, four, five, six, seven, eight, nine, or more than nine atoms. Any number of those atoms can be heteroatoms (/.e., a heterocyclic ring can include one, two, three, four, five, six, seven, eight, nine, or more than nine heteroatoms). In heterocyclic rings including two or more heteroatoms, those two or more heteroatoms can be the same or different from one another. Heterocycles can be substituted. Binding to a heterocycle can be at a heteroatom or via a carbon atom. It should also be understood that in the present description, the term “heterocycle” also encompasses “heteroaryl” groups.
- protecting group means a moiety of a compound that masks or alters the properties of a functional group or the properties of the compound as a whole.
- the chemical substructure of a protecting group can greatly vary.
- One function of a protecting group is to serve as an intermediate in the synthesis of the parental active substance.
- Chemical protecting groups and strategies for protection/deprotection are well known in the art. See: “Protective Groups in Organic Chemistry", Theodora W. Greene (John Wiley & Sons, Inc., New York, 1991).
- substituted as used herein in reference to alkyl, alkylene, alkoxy, alkenyl, alkynyl, alkenylene, aryl, alkynylene, etc., for example “substituted alkyl”, “substituted alkylene”,“substituted alkoxy” -“or substituted O(Alkyl)”,“substituted alkenyl”,“substituted alkynyl”, “substituted alkenylene”, “substituted aryl” and “substituted alkynylene”, unless otherwise indicated, means alkyl, alkylene, alkoxy, alkenyl, alkynyl, alkenylene, aryl and alkynylene, respectively, in which one or more hydrogen atoms are each independently replaced with a non-hydrogen substituent.
- substituted when used in conjunction with groups such as arylalkyl, which have two or more moieties capable of substitution, the substituents can be attached to the aryl moiety, the alkyl moiety, or both.
- tri-substituted silyl refers to a silyl group that is independently substituted with three functional groups selected from alkyl, alkenyl, alkynyl, aryl and arylalkyl.
- tri-substituted silyl groups include trimethylsilyl and dimethylphenylsilyl.
- PEG poly(ethylene glycol)
- PEG chains of the present description can include one of the following structures: -(CH 2 CH 2 O) m - or -(CH 2 CH 2 O) m -iCH 2 CH 2 -, depending on if the terminal oxygen has been displaced, where m is an integer, optionally selected from 1 to 100, 1 to 50, 1 to 30, 5 to 30, 5 to 20 or 5 to 15.
- the PEG can be capped with an“end capping group” that is generally a non-reactive carbon-containing group attached to a terminal oxygen or other terminal atom of the PEG.
- end capping groups can include alkyl, substituted alkyl, aryl, substituted aryl, alkenyl, substituted alkenyl, alkynyl, substituted alkynyl, CO(alkyl), CO(substituted alkyl), CO(alkenyl), CO(substituted alkenyl), CO(alkynyl) or CO(substituted alkynyl).
- natural amino acid refers to the twenty natural amino acids. More specifically, the natural amino acid can be selected from the group consisting of: Alanine (Ala), Glycine (Gly), Isoleucine (lie), Leucine (Leu), Proline (Pro), Valine (Val), Phenylalanine (Phe), Tryptophan (Trp), Tyrosine (Tyr), Aspartic acid (Asp), Glutamic acid (Glu), Arginine (Arg), Histidine (His), Lysine (Lys), Serine (Ser), Threonine (Thr), Cysteine (Cys), Methionine (Met), Asparagine (Asn) and Glutamine (Gin).
- the natural amino acids bear an asymmetric carbon (the alpha-carbon) that is of (S) configuration (also referred to as L-amino acids).
- the expression“natural amino acid attached to the compound by its amino group bonded to the alpha-carbon”, as used herein, means that a hydrogen of the amino group bonded to the alpha-carbon is removed and replaced by a bond between the amino acid and the rest of the compound.
- substituents and other moieties of the compounds of the present description should be selected in order to provide an agriculturally useful compound which can be formulated into an acceptably stable agricultural composition that can be applied to plants.
- the definitions and substituents for various genus and subgenus of the compounds of the present description are described and illustrated herein. It should be understood by a person skilled in the art that any combination of the definitions and substituents described herein should not result in an inoperable species or compound. It should also be understood that the phrase “inoperable species or compound” means compound structures that violate relevant scientific principles (such as, for example, a carbon atom connecting to more than four covalent bonds) or compounds too unstable to permit isolation and formulation into agriculturally acceptable compositions.
- R x includes a R y substituent.
- R y can be R.
- R can be W 3 .
- W 3 can be W 4 and W 4 can be R or include substituents including R y .
- a person skilled in the art of organic chemistry understands that the total number of such substituents is to be reasonably limited by the desired properties of the compound intended.
- each recursive substituent can independently occur 20, 19, 18, 17, 16, 15, 14, 13, 12, 1 1 , 10, 9, 8, 7, 6, 5, 4, 3, 2, 1 , or 0, times in a given implementation.
- each recursive substituent can independently occur 3 or fewer times in a given embodiment.
- Recursive substituents are an intended aspect of the compounds of the present description. A person skilled in the art of organic chemistry understands the versatility of such substituents.
- the term“agriculturally acceptable salt”, as used herein, refers to salts that exhibit pesticidal activity (/.e., that are active against one or more biotic stress) or that can improve resistance of a plant to one or more abiotic stresss.
- the term also refers to salts that are or can be converted in plants, water or soil to a compound or salt that exhibits pesticidal activity or that can improve resistance of a plant to one or more abiotic stress.
- The“agriculturally acceptable salt” can be an agriculturally acceptable cation or agriculturally acceptable anion.
- Non-limiting examples of agriculturally acceptable cations can include cations derived from alkali or alkaline earth metals and cations derived from ammonia and amines.
- agriculturally acceptable cations can include sodium, potassium, magnesium, alkylammonium and ammonium cations.
- Non-limiting examples of agriculturally acceptable anions can include halide, phosphate, alkylsulfate and carboxylate anions.
- agriculturally acceptable anions can include chloride, bromide, methylsulfate, ethylsulfate, acetate, lactate, dimethyl phosphate or polyalkoxylated phosphate anions.
- a compound of the present description and its agriculturally acceptable salts may exist as different polymorphs or pseudopolymorphs.
- crystalline polymorphism means the ability of a crystalline compound to exist in different crystal structures. The crystalline polymorphism may result from differences in crystal packing (packing polymorphism) or differences in packing between different conformers of the same molecule (conformational polymorphism).
- crystalline pseudopolymorphism means the ability of a hydrate or solvate of a compound to exist in different crystal structures.
- Pseudopolymorphs of the compounds of the present description may exist due to differences in crystal packing (packing pseudopolymorphism) or due to differences in packing between different conformers of the same molecule (conformational pseudopolymorphism).
- the description and depiction of the compounds of the present description is intended to include all polymorphs and pseudopolymorphs of the compounds and their agriculturally acceptable salts.
- a compound of the present description and its agriculturally acceptable salts may also exist as an amorphous solid.
- an amorphous solid is a solid in which there is no long-range order of the positions of the atoms in the solid.
- the description and depiction of the compounds of the present description is intended to include all amorphous forms of the compounds and their agriculturally acceptable salts.
- the modifier“about” used in connection with a quantity is inclusive of the stated value and has the meaning dictated by the context.
- the modifier“about” can include the degree of error associated with the measurement of the quantity.
- salts of the compounds of the present description are agriculturally acceptable salts.
- salts which are not agriculturally acceptable can also find use, for example, in the preparation or purification of an agriculturally acceptable compound. All salts, whether or not they are agriculturally acceptable salts, are therefore to be understood as within the scope of the present description.
- each group is independently selected.
- Si(OR 7 )3 with each R 7 being independently alkyl or aryl
- each R 7 can independently be selected from alkyl groups and aryl groups.
- Si(OR 7 )3 therefore includes both symmetrical groups where all three R 7 are the same and asymmetrical groups where at least one R 7 group is different from the other two R 7 groups, or where each R 7 group is different.
- R q or Z q groups defined herein (e.g., q being selected from 1 to 17, from a to f or from A to C).
- the compounds described herein can also exist as tautomeric forms in certain cases. Although only one delocalized resonance structure will typically be depicted, all such forms are contemplated within the scope of the present description. For example, various tautomers can exist for the tetrapyrole ring systems described herein, and all their possible tautomeric forms are within the scope of the present description.
- the term“growing medium”, as used herein, refers to any soil (of any composition) or soil-free (e.g., hydroponic) medium that is suitable for growing and cultivating a plant.
- the growing medium can further include any naturally occurring and/or synthetic substance(s) that are suitable for growing and cultivating the plant.
- the phrase“any surface of the growing medium” or“a surface of the growing medium”, as used herein, refers to a surface that is directly exposed to natural and/or simulated light and/or weather.
- applying refers to contacting a surface of the growing medium with at least one compound of the present description (e.g., combinations, compositions, solutions, emulsions including at least one compound of the present description), by any means known in the art (e.g., pouring, root bathing, soil drenching, drip irrigation, etc.), or contacting an area that is beneath the surface of the growing medium with at least one compound of the present description (e.g., by soil injection), or any combination thereof, or directly contacting the plant with at least one compound of the present description (e.g., spraying).
- a compound of the present description e.g., combinations, compositions, solutions, emulsions including at least one compound of the present description
- any means known in the art e.g., pouring, root bathing, soil drenching, drip irrigation, etc.
- contacting an area that is beneath the surface of the growing medium with at least one compound of the present description e.g., by soil injection
- the term“crop plant”, as used herein, refers to a non-woody plant, which is grown, tended to, and harvested in a cycle of one year or less as source of foodstuffs and/or energy.
- crop plants include sugar cane, wheat, rice, corn (maize), potatoes, sugar beets, barley, sweet potatoes, cassava, soybeans, tomatoes, and legumes (beans and peas).
- the crop plant can be a monocot or a dicot.
- the term“woody plant”, as used herein, refers to a woody perennial plant having a single stem or trunk, and bearing lateral branches at some distance from the ground (e.g., a tree).
- the woody plant can be a deciduous tree, an evergreen tree (e.g., a coniferous) or a shrub.
- woody plants include maple trees, citrus trees, apple trees, pear trees, oak trees, ash trees, pine trees, and spruce trees.
- Turf grass refers to a cultivated grass that provides groundcover, for example a turf or lawn that is periodically cut or mowed to maintain a consistent height.
- Grasses belong to the Poaceae family, which is subdivided into six subfamilies, three of which include common turf grasses: the Festucoideae subfamily of cool- season turf grasses; and the Panicoideae and Eragrostoideae subfamiles of warm-season turf grasses.
- a limited number of species are in widespread use as turf grasses, generally meeting the criteria of forming uniform soil coverage and tolerating mowing and traffic.
- turf grasses have a compressed crown that facilitates mowing without cutting off the growing point.
- the term "turf grass” includes areas in which one or more grass species are cultivated to form relatively uniform soil coverage, including blends that are a combination of different cultivars of the same species, or mixtures that are a combination of different species and/or cultivars.
- Non-limiting examples of turf grasses include: bluegrasses (e.g., Kentucky bluegrass), bentgrasses (e.g., creeping bentgrass), Redtop, fescues (e.g., red fescue), ryegrasses (e.g., annual ryegrass), wheatgrasses (e.g., crested wheatgrass), beachgrass, Brome grasses (e.g., Arizona Brome), cattails (e.g., sand cattail), Alkaligrass ( Puccinellia distans), crested dog's-tail ( Cynosurus cristatus), bermudagrass ( Cynodon spp.
- bluegrasses e.g., Kentucky bluegrass
- bentgrasses e.g., creeping bentgrass
- ryegrasses e.g., annual ryegrass
- wheatgrasses e.g.,
- Cynodon dactylon such as Cynodon dactylon
- hybrid bermudagrass e.g.,tifdwarf bermudagrass
- Zoysiagrasses e.g., Zoysia japonica
- St. Augustinegrass e.g., Bitter Blue St.
- the phrase“promoting the health of a plant”, as used herein, includes at least one of controlling a disease, condition, or injury caused by a pest of a plant and increasing abiotic stress resistance or tolerance in a plant.
- the phrase“promoting the health of a plant” includes at least one of“controlling infection of a plant by one or more biotic agent”, “controlling infestation of a plant by one or more insect” and“increasing resistance of a plant to one or more abiotic stress”.
- controlling infection of a plant by a biotic agent means to diminish, ameliorate, or stabilize the infection and/or any other existing unwanted condition or side effect that is caused by the association of a microbial pathogen or infestation of an insect on the plant.
- the microbial pathogen can include fungi, bacteria (gram positive or gram negative), viruses, viroids, virus-like organisms, phytoplasma, etc.
- abiotic stress refers to environmental conditions that negatively impact growth, development, yield and yield quality of crop and other plants below optimum levels.
- Non-limiting examples of abiotic stresses include, for example: photooxidative conditions, drought (water deficit), excessive watering (flooding, and submergence), extreme temperatures (chilling, freezing and heat), extreme levels of light (high and low), radiation (UV-B and UV-A), salinity due to excessive Na + (sodicity), chemical factors (e.g., pH), mineral (metal and metalloid) toxicity, deficiency or excess of essential nutrients, gaseous pollutants (ozone, sulfur dioxide), wind, mechanical factors, and other stressors.
- the term“increasing stress resistance” refers to an increase in the ability of a plant to survive or thrive in stress conditions. Enhanced resistance or tolerance can be specific for a particular stressor, e.g., drought, excess water, nutrient deficiency, salt, cold, shade or heat, or multiple stressors. In some scenarios, increased resistance to one or more abiotic stresses can be exemplified by the reduction in degradation of quality of the plant, as compared to an untreated plant subjected to the same stress. In other scenarios, increased resistance to one or more abiotic stress can be exemplified by maintained or improved plant quality, as compared to an untreated plant subjected to the same stress.
- photosensitizer compounds can be used to enable photodynamic inhibition of biotic agents (i.e., microbial pathogens and/or insects) that are present on plants.
- the photosensitizer compounds react to light by generating reactive oxygen species (ROS).
- ROS reactive oxygen species
- photosensitizers can be classified into two classes, namely Type I photosensitizers and Type II photosensitizers.
- Type I photosensitizers form short lived free radicals through electron abstraction or transfer from a substrate when excited at an appropriate wavelength in the presence of oxygen.
- Type II photosensitizers form a highly reactive oxygen state known as“singlet oxygen”, also referred to herein as“reactive singlet oxygen species”. Singlet oxygen species are generally relatively long lived and can have a large radius of action.
- the photosensitizer compound can be metallated or non- metallated.
- the metal can be selected to generate either a Type I or a Type II photosensitizer in response to light exposure.
- the ROS that are generated are typically Type I photosensitizers.
- the ROS that are generated are typically Type II photosensitizers.
- Both Type I and Type II photosensitizers can be used to enable photodynamic inhibition of biotic agents that are present on plants or to protect a plant from abiotic stress.
- the term“singlet oxygen photosensitizer”, as used herein, refers to a compound that produces reactive singlet oxygen species when excited by light.
- the term“singlet oxygen photosensitizer” refers to a photosensitizer in which the Type II process defined above is dominant compared to the Type I process.
- Protoporphyrin IX is an organic compound, which is one of the most common porphyrins in nature. PP IX is a deeply colored pigment that is not very soluble even in basic water. PP IX is encountered in nature in the form of its iron complexes. When complexed with ferrous iron, the molecule is called heme. Other iron complexes have also been synthesized, for example with Fe(lll) or Fe(IV). PP IX is a largely planar tetrapyrrole having a 20-carbon atom macrocyclic ring, each pyrrole being linked to two other pyrroles of the macrocyclic ring by a one-carbon bridge.
- tetrapyrrole it is meant four pyrrole like rings.
- a“pyrrole-like” ring is a five-atom ring with four carbon atoms and one nitrogen atom.
- the carbons of the macrocyclic ring are numbered from 1 to 20.
- two carboxylic acid-bearing moieties are provided at the C13 (CH 2 CH 2 COOH) and C17 (CH 2 CH 2 COOH) positions.
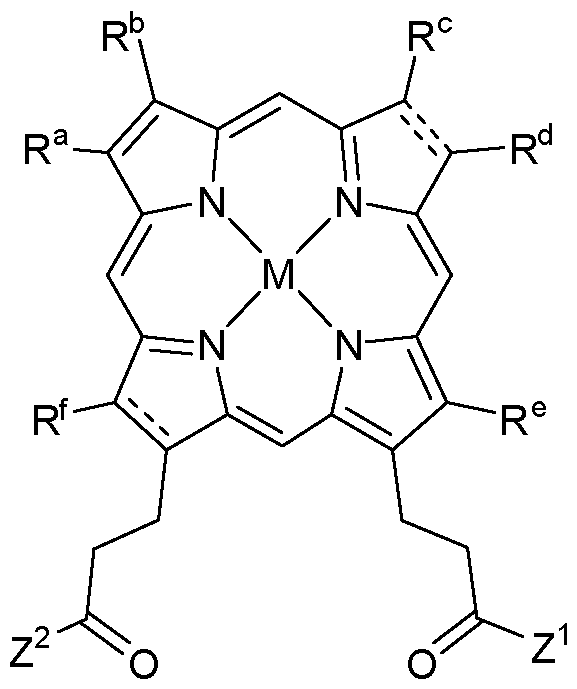
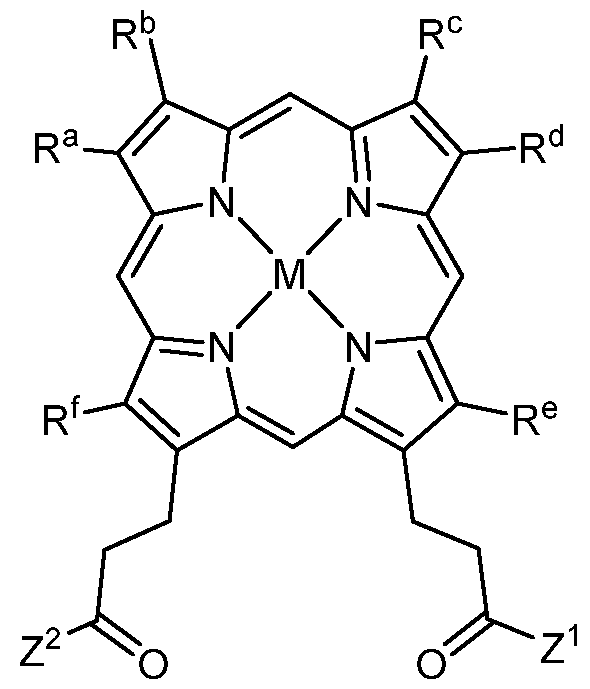
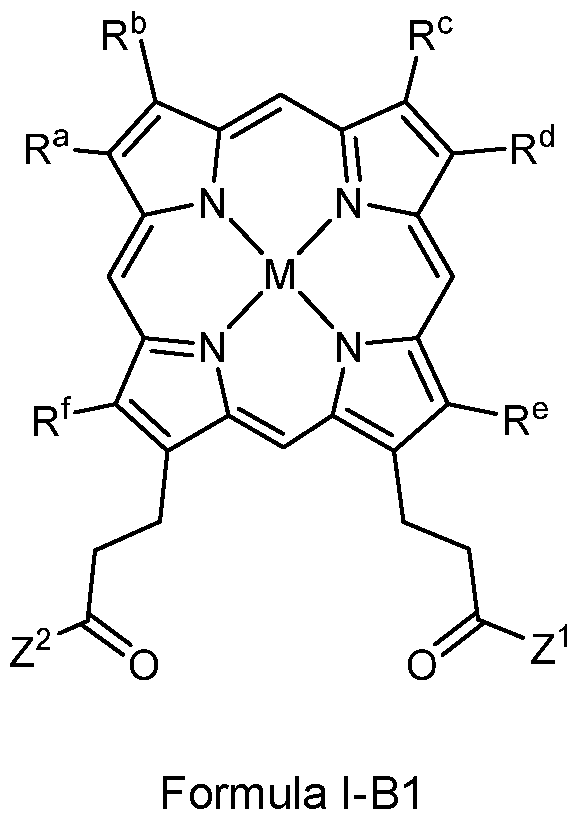
- the compounds of the present description include photosensitizer compounds based on or similar to the PP IX scaffold above, that are of general Formula I represented below, or an agriculturally acceptable salt thereof.
- the term“photosensitizer compound” refers to one or more compounds of Formula I.
- the term“photosensitizer compound” can refer to one compound of Formula I or to a combination or mixture of two or more compounds of Formula I.
- Z 1 and Z 2 are each independently OR 1 or NR 2 R 3 ;
- each R 1 , R 2 and R 3 is independently H, alkyl, substituted alkyl, aryl, substituted aryl, alkenyl, substituted alkenyl, alkynyl, or substituted alkynyl, wherein:
- each R a , R B , R c , R d , R e and R f is, independently, H, alkyl, substituted alkyl, aryl, substituted aryl, alkenyl, substituted alkenyl, alkynyl or substituted alkynyl;
- M is 2H or a metal species
- each X is independently a halogen: F, Cl, Br or I;
- each R B is independently H, alkyl, aryl, arylalkyl, a heterocycle, an alkyloxy group such as poly(ethyleneoxy), PEG or poly(methyleneoxy), a capped poly(ethyleneoxy), capped PEG or capped polymethyleneoxy, or a protecting group;
- the capped poly(ethyleneoxy), capped PEG and capped poly(methyleneoxy) groups being each independently capped with alkyl, aryl, arylalkyl, alkenyl, alkynyl, CO(alkyl), CO(aryl), CO(arylalkyl), CO(alkenyl) or CO(alkynyl);
- each R c is independently alkyl, aryl, arylalkyl, O(alkyl), O(aryl), O(arylalkyl), or O(tri- substituted silyl);
- each tri-substituted silyl is independently substituted with three functional groups selected from alkyl, alkenyl, alkynyl, aryl and arylalkyl;
- each Aik is independently alkylene, alkenylene, or alkynylene.
- the compound of Formula I is such that:
- one of Z 1 and Z 2 is OR 1 ;
- Z 1 and Z 2 is NR 2 R 3 , NR 2 -(CH 2 ) n -NR 4 R 5 , NR 2 -(CH 2 ) n -N + R 4 R 5 R 6 Y-,
- Z 1 is NR 2 R 3 , N R 2 -(CH 2 ) n - N R 4 R 5 , NR 2 -(CH 2 ) n -N + R 4 R 5 R 6 Y-, NR 2 -(CH 2 ) n -O(PO 3 H)- W + , NR 2 -(CH 2 ) n -Si(R 7 ) 3 , NR 2 -(CH 2 ) n -SR 8 NR 2 -(CH 2 ) n -NR 4 -(CH 2 ) p -NR 9 R 10 ,
- each R 1 and R 2 is, independently, H, alkyl, substituted alkyl, aryl, substituted aryl, alkenyl, substituted alkenyl, alkynyl or substituted alkynyl;
- R 3 is alkyl, substituted alkyl, aryl, substituted aryl, alkenyl, substituted alkenyl, alkynyl or substituted alkynyl;
- each R 4 , R 6 , R 8 , R 9 , R 10 and R 11 is, independently, H, alkyl, substituted alkyl, aryl, substituted aryl, alkenyl, substituted alkenyl, alkynyl, substituted alkynyl or -(CH 2 ) q - (CH 2 CH 2 O) m -R 13 ;
- R 5 is alkyl, substituted alkyl, aryl, substituted aryl, alkenyl, substituted alkenyl, alkynyl, substituted alkynyl or -(CH 2 ) q -(CH 2 CH 2 O)m-R 13 ;
- R 7 is alkyl, O(alkyl) or O(tri-substituted silyl);
- R 13 is H, alkyl, substituted alkyl, aryl, substituted aryl, alkenyl, substituted alkenyl, alkynyl, substituted alkynyl, CO(alkyl), CO(substituted alkyl), CO(alkenyl), CO(substituted alkenyl), CO(alkynyl) or CO(substituted alkynyl);
- W + is an agriculturally acceptable cation
- Y- is an agriculturally acceptable anion
- n is an integer selected from 1 to 16;
- p is an integer selected from 1 to 16;
- n is an integer selected from 1 to 100;
- q is an integer selected from 0 to 16;
- each R a , R B , R c , R d , R e and R f is, independently, H, alkyl, substituted alkyl, aryl, substituted aryl, alkenyl, substituted alkenyl, alkynyl or substituted alkynyl;
- M is 2H or a metal species
- each substituted alkyl, substituted aryl, substituted alkenyl and substituted alkynyl groups are, independently, substituted with one or more OH, F, Cl, Br, I, CN and N 3 .
- Z 1 is NR 2 R 3 and Z 2 is OH, or Z 1 is OH and Z 2 is NR 2 R 3 .
- R 3 can for example be alkyl or substituted alkyl.
- each R a , R B , R c , R d , R e and R f is, independently, alkyl or alkenyl.
- R a , R c , R e and R f are methyl while R B and R d are vinyl.
- M is 2H.
- M is a metal species selected from the group consisting of Mg, Zn, Pd, Sn, Al, Pt, Si, Ge, Ga, In, Cu, Co, Fe and Mn.
- M is a metal species selected from the group consisting of Mg(ll), Zn(ll), Pd(ll), Sn(IV), Al(lll), Pt(ll), Si(IV), Ge(IV), Ga(lll) and In(lll).
- M is a metal species selected from the group consisting of Cu(ll), Co(ll), Fe(ll) and Mn(ll).
- M is a metal species selected from the group consisting of Cu(ll), Co(lll), Fe(ll l) and Mn(lll).
- each R 1 , R 2 , R 4 , R 6 , R 8 , R 9 , R 10 and R 11 is, independently, H, alkyl or substituted alkyl.
- each R 3 and R 5 is, independently, alkyl or substituted alkyl.
- R 13 is H, alkyl, substituted alkyl, CO(alkyl) or CO(substituted alkyl).
- the compound of Formula I is selected such that at least one of the following is true: R 1 is H, R 2 is H, R 3 is alkyl, R 4 is H or alkyl, R 5 is alkyl, R 6 is alkyl, R 7 is O(tri-substituted silyl), R 8 is H or alkyl, R 9 is alkyl, R 10 is alkyl, R 11 is alkyl and R 13 is H, alkyl, alkenyl, CO(alkyl) or CO(alkenyl).
- W + is selected from the group consisting of sodium, potassium, magnesium and ammonium cations.
- Y- is selected from the group consisting of chloride, bromide, phosphate, dimethylphosphate, methylsulfate, ethylsulfate, acetate and lactate.
- n is an integer selected from 1 to 16, or from 1 to 12, or from 1 to 8, or from 1 to 6, or from 1 to 4, or from 2 to 4.
- p is an integer selected from 1 to 16, or from 1 to 12, or from 1 to 8, or from 1 to 6, or from 1 to 4, or from 2 to 4.
- m is an integer that can be selected from 1 to 100, or from 1 to 80, or from 1 to 60, or from 1 to 50, or from 1 to 30, or from 1 to 20, or from 1 to 10, or from 5 to 30, or from 5 to 20, or from 5 to 10.
- Z 1 is NR 2 R 3 , NR 2 -(CH 2 ) n -NR 4 R 5 , NR 2 -(CH 2 ) n -N + R 4 R 5 R 6 Y-, NR 2 -(CH 2 ) n -O(PO 3 H) W ⁇ NR 2 -(CH 2 ) n -Si(R 7 ) 3 , NR 2 -(CH 2 ) n -SR 8 NR 2 -(CH 2 ) n -NR 4 -(CH 2 ) p -NR 9 R 10 , NR 2 -(CH 2 ) n -NR 4 -(CH 2 ) p -N + R 9 R 10 R 11 Y-,
- one of Z 1 and Z 2 is NR 2 R 3 , NR 2 -(CH 2 ) n -NR 4 R 5 , NR 2 - (CH 2 ) n -N + R 4 R 5 R 6 Y , NR 2 -(CH 2 ) n -O(PO 3 H)- W ⁇ NR 2 -(CH 2 ) n -Si(R 7 ) 3 , NR 2 -(CH 2 ) n -SR 8 or NR 2 -(CH 2 ) n -NR 4 -(CH 2 ) p -NR 9 R 10 ; and the other one of Z 1 and Z 2 is OR 1 ; or Z 1 is NR 2 R 3 , NR 2 -(CH 2 ) n -NR 4 R 5 , NR 2 -(CH 2 ) n -N + R 4 R 5 R 6 Y , NR 2 -(CH 2 ) n -O(
- one of Z 1 and Z 2 is NR 2 R 3 , NR 2 -(CH 2 ) n -NR 4 R 5 , NR 2 - (CH 2 ) n -N + R 4 R 5 R 6 Y , NR 2 -(CH 2 ) n -O(PO 3 H)- W ⁇ NR 2 -(CH 2 ) n -Si(R 7 ) 3 , NR 2 -(CH 2 ) n -SR 8 or NR 2 -(CH 2 ) n -NR 4 -(CH 2 ) p -NR 9 R 10 ; and the other one of Z 1 and Z 2 is OR 1 .
- Z 1 is NR 2 R 3 , NR 2 -(CH 2 ) n -NR 4 R 5 , NR 2 -(CH 2 ) n -N + R 4 R 5 R 6 Y-,
- one of Z 1 and Z 2 is NR 2 R 3 ; and the other one of Z 1 and Z 2 is OR 1 ;
- each R 1 and R 2 is, independently, H, alkyl or substituted alkyl
- R 3 is alkyl or substituted alkyl
- each R a , R B , R c , R d , R e and R f is, independently, H, alkyl, substituted alkyl, alkenyl, substituted alkenyl, alkynyl or substituted alkynyl;
- M is 2H or a metal species
- substituted alkyl, substituted alkenyl and substituted alkynyl groups are, independently, substituted with one or more OH, F, Cl, Br, I, CN and N3.
- R 1 is H
- R 2 is H and/or R 3 is alkyl.
- R 3 can for example be a (Ci-Ci2)alkyl, a (C 1 -C 8 )alkyl or a (C 1 -C 4 )alkyl.
- one of Z 1 and Z 2 is NR 2 R 3 ; and the other one of Z 1 and Z 2 is OR 1 .
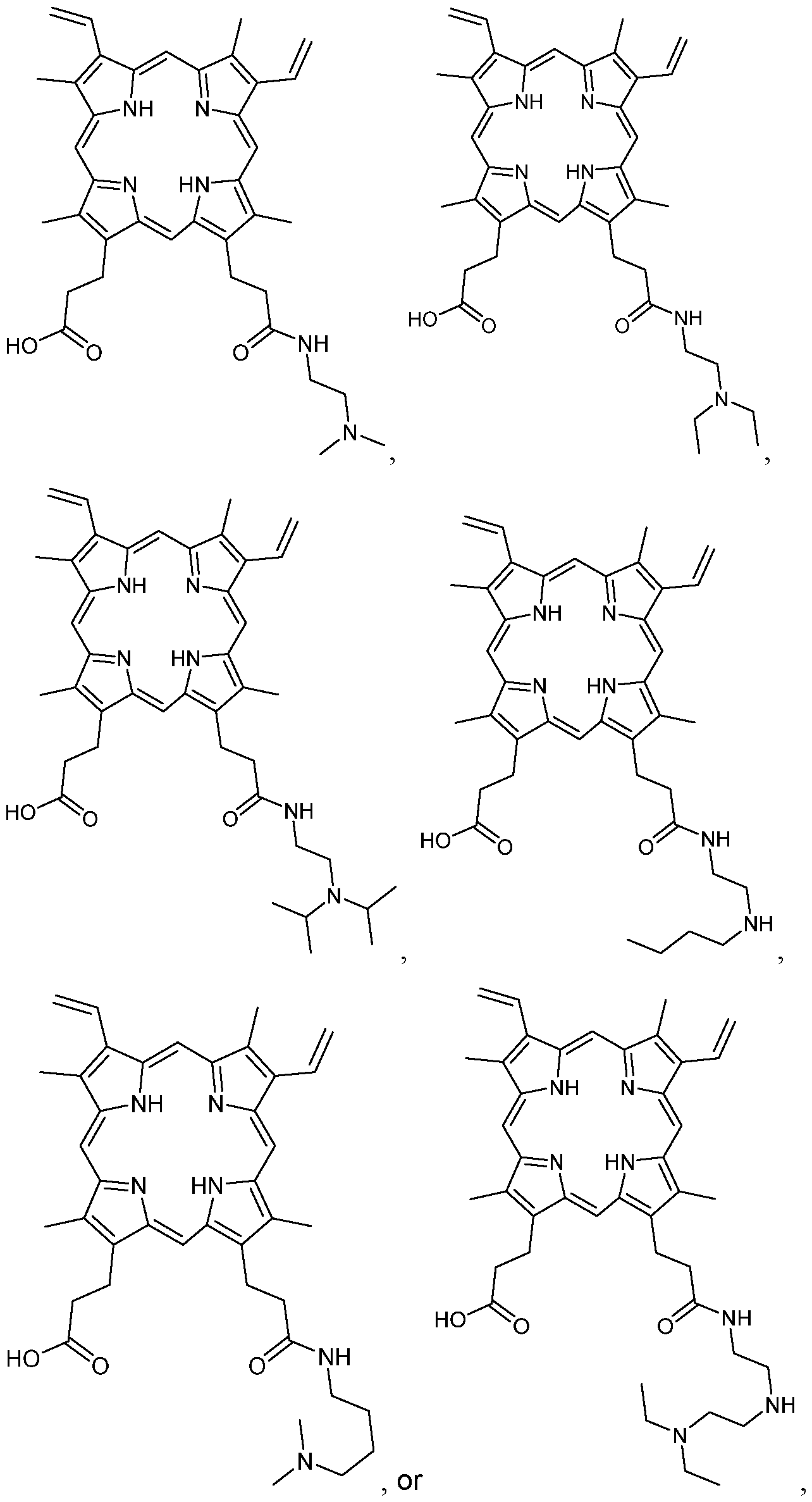
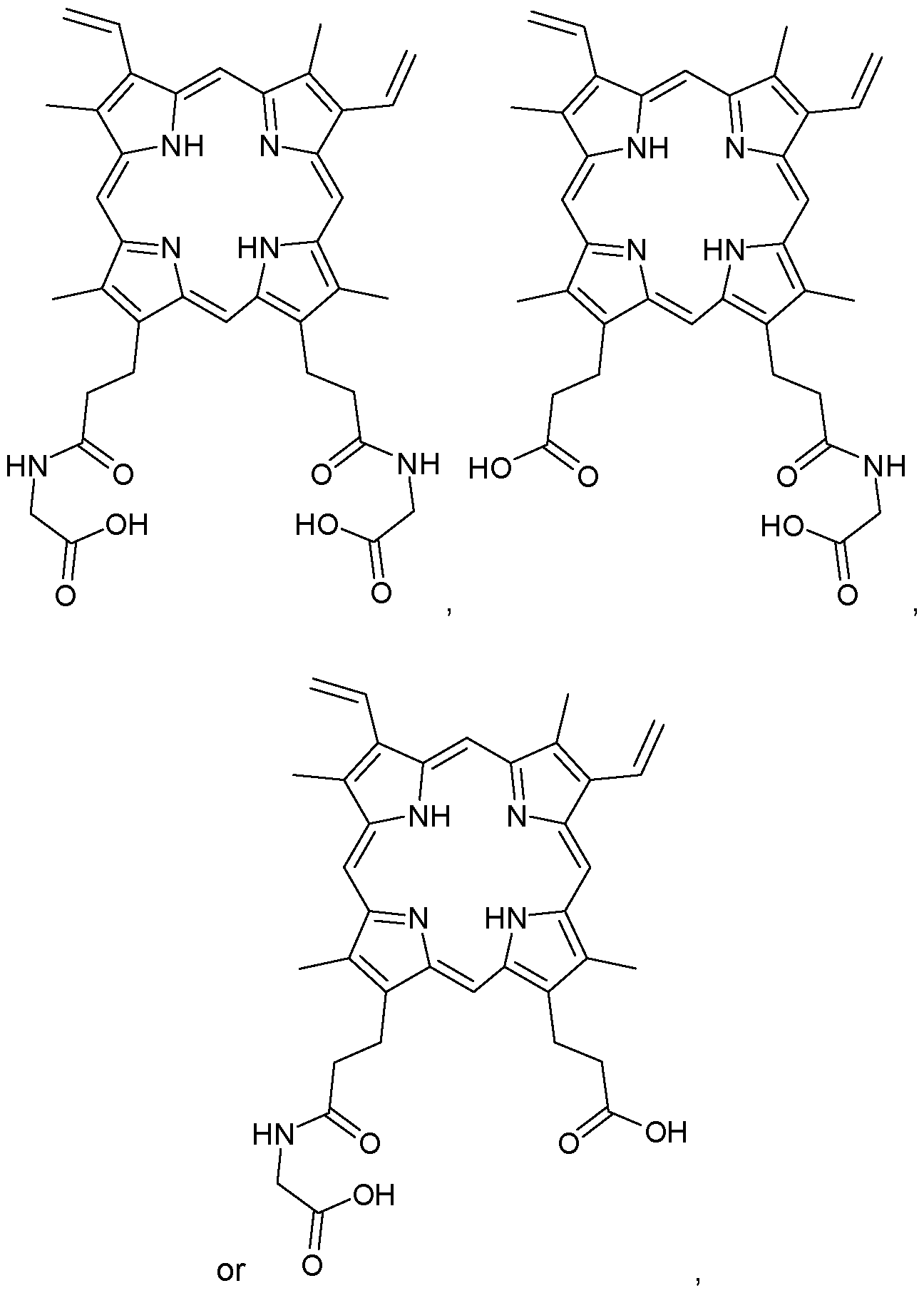
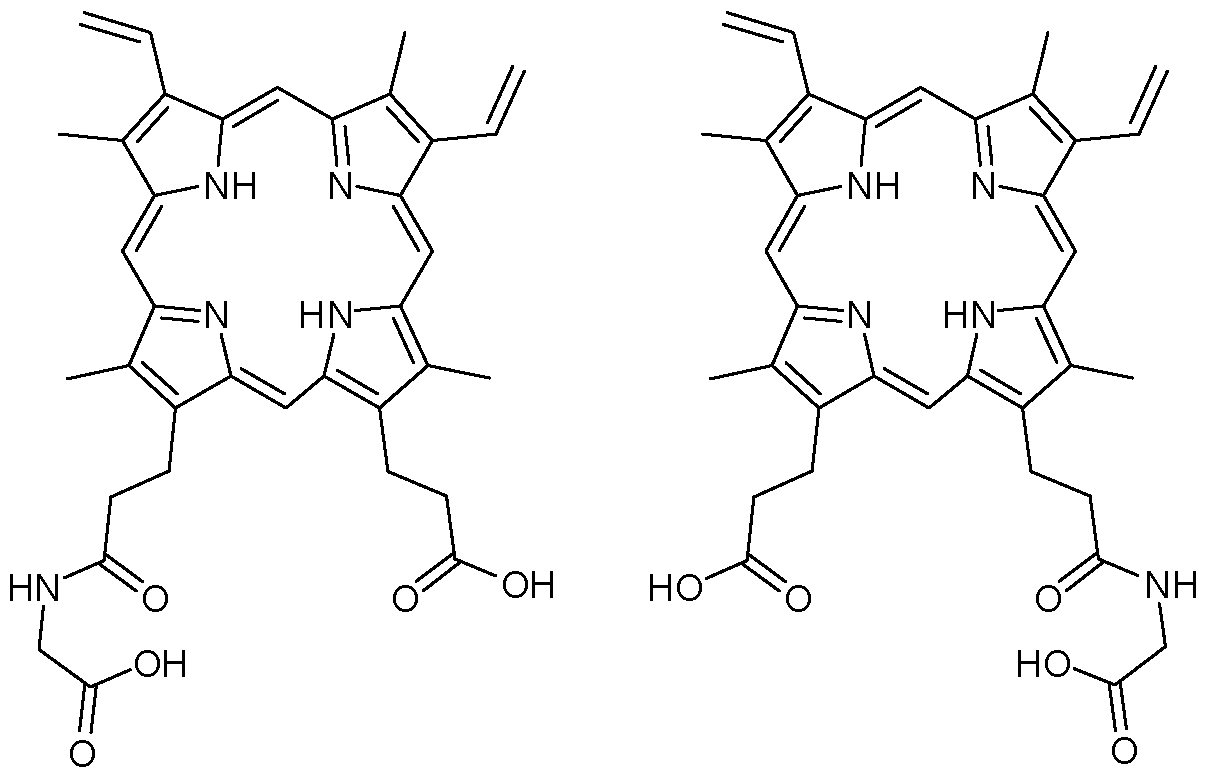
- the compound of Formula I-B1 is:
- the compound of Formula I-B1 is:
- the compound of Formula I-B1 is:
- Formula I-B1 or an agriculturally acceptable salt thereof, for use in promoting the health of a plant wherein:
- Z 1 and Z 2 is NR 2 -(CH 2 ) n -NR 4 R 5 or O-(CH 2 ) n -NR 4 R 5 ;
- Z 1 NR 2 -(CH 2 ) n -NR 4 R 5 or O-(CH 2 ) n -NR 4 R 5 ;
- R 5 is alkyl, substituted alkyl or -(CH 2 ) p -NR 9 R 10 ;
- each R 1 , R 2 , R 4 , R 9 and R 10 is, independently, H, alkyl or substituted alkyl;
- n is an integer selected from 1 to 16;
- p is an integer selected from 1 to 16;
- each R a , R B , R c , R d , R e and R f is, independently, H, alkyl, substituted alkyl, alkenyl, substituted alkenyl, alkynyl or substituted alkynyl;
- M is 2H or a metal species
- substituted alkyl, substituted alkenyl and substituted alkynyl groups are, independently, substituted with one or more OH, F, Cl, Br, I, CN and N3.
- R 1 is H
- R 2 is H
- R 4 is H or alkyl.
- R 4 is H and R 5 is alkyl. In some implementations, R 4 and R 5 are alkyl. R 4 and/or R 5 can for example each independently be a (C 1 -C 12 )alkyl, a (C 1 -C 8 )alkyl or a (C 1 - C 4 )alkyl. In some implementations, R 5 is -(CH 2 ) P -NR 9 R 10 . In some implementations, R 9 and R 10 are alkyl, or R 9 is H and R 10 is alkyl. R 9 and/or R 10 can for example each independently be a (C 1 -C 12 )alkyl, a (C 1 -C 8 )alkyl or a ( C 1 -C 4 )alkyl.
- n is an integer selected from 1 to 16, or from 1 to 12, or from 1 to 8, or from 1 to 6, or from 1 to 4, or from 2 to 4.
- p is an integer selected from 1 to 16, or from 1 to 12, or from 1 to 8, or from 1 to 6, or from 1 to 4, or from 2 to 4.
- one of Z 1 and Z 2 is NR 2 -(CH 2 ) n -NR 4 R 5 ; and the other one of Z 1 and Z 2 is OR 1 .
- the compound of Formula I-B1 is:
- the compound of Formula I-B1 is:
- the compound of Formula I-B1 is:
- Z 1 and Z 2 is NR 2 -(CH 2 ) n -Si(R 7 ) 3 , O-(CH 2 ) n -Si(R 7 ) 3 , NR 2 -(CH 2 ) n -SR 8 or O- (CH 2 ) n -SR 8 ; and
- each R 1 and R 2 is, independently, H, alkyl or substituted alkyl
- R 7 is alkyl, O(alkyl) or O(trisubstituted silyl);
- R 8 is H, alkyl, substituted alkyl or -(CH 2 ) q -(CH 2 CH 2 O) m -R 13 ;
- R 13 is H, alkyl, substituted alkyl, alkenyl, substituted alkenyl, alkynyl, substituted alkynyl, CO(alkyl), CO(substituted alkyl), CO(alkenyl), CO(substituted alkenyl), CO(alkynyl) or CO(substituted alkynyl);
- n is an integer selected from 1 to 16;
- n is an integer selected from 1 to 100;
- q is an integer selected from 0 to 16;
- each R a , R B , R c , R d , R e and R f is, independently, H, alkyl, substituted alkyl, alkenyl, substituted alkenyl, alkynyl or substituted alkynyl;
- M is 2H or a metal species
- R 1 is H and/or R 2 is H.
- R 7 is alkyl, O(alkyl) or O(tri-substituted silyl).
- the alkyl groups for R 1 , R 2 and R 7 can each independently be a (Ci-Ci2)alkyl, a (C 1 -C 8 )alkyl or a (C 1 -C 4 )alkyl.
- R 8 is -(CH 2 )q-(CH 2 CH 2 O) m -R 13 .
- R 13 can be H and m can be an integer selected from 1 to 20.
- n is an integer selected from 1 to 16, or from 1 to 12, or from 1 to 8, or from 1 to 6, or from 1 to 4, or from 2 to 4.
- q is an integer selected from 0 to 16, or from 1 to 8, or from 0 to 4, or from 0 to 2.
- one of Z 1 and Z 2 is NR 2 -(CH 2 ) n -Si(R 7 )3, O-(CH 2 ) n -Si(R 7 )3, N R 2 -(CH 2 )n-SR 8 or O-(CH 2 ) n -SR 8 ; and the other one of Z 1 and Z 2 is OR 1 .
- the compound of Formula I-B1 is:
- n is an integer selected from 4 to 15.
- the compound of Formula I-B1 is:
- the compound of Formula I-B1 is:
- each R 1 and R 2 is, independently, H, alkyl or substituted alkyl; n is an integer selected from 1 to 16;
- W + is an agriculturally acceptable cation
- each R a , R B , R c , R d , R e and R f is, independently, H, alkyl, substituted alkyl, alkenyl, substituted alkenyl, alkynyl or substituted alkynyl;
- M is 2H or a metal species
- substituted alkyl, substituted alkenyl and substituted alkynyl groups are, independently, substituted with one or more OH, F, Cl, Br, I, CN and N 3 .
- R 1 is H and/or R 2 is H.
- n is an integer selected from 1 to 16, or from 1 to 12, or from 1 to 8, or from 1 to 6, or from 1 to 4, or from 2 to 4.
- W + can be selected from the group consisting of sodium, potassium, magnesium and ammonium cations.
- Z 2 Z 1 .
- the compound of Formula I-B1 is:
- the compound of Formula I-B1 is:
- W+ is a cation that can be selected from the group consisting of: of sodium, potassium, magnesium, ammonium and alkylammonium cations.
- W+ can be an n-octylammonium cation.
- Z 1 and Z 2 is NR 2 -(CH 2 ) n -NR 4 R 5 R 6+ Y- or O-(CH 2 ) n -NR 4 R 5 R 6+ Y-; and the other one of Z 1 and Z 2 is OR 1 ;
- Z 1 N R 2 -(CH 2 ) n - N R 4 R 5 R 6+ Y- or O-(CH 2 ) n -NR 4 R 5 R 6+ Y-;
- each R 1 and R 2 is, independently, H, alkyl or substituted alkyl
- each R 4 , R 5 and R 6 is, independently, alkyl or substituted alkyl
- n is an integer selected from 1 to 16;
- Y- is an agriculturally acceptable anion
- each R a , R B , R c , R d , R e and R f is, independently, H, alkyl, substituted alkyl, alkenyl, substituted alkenyl, alkynyl or substituted alkynyl;
- M is 2H or a metal species
- substituted alkyl, substituted alkenyl and substituted alkynyl groups are, independently, substituted with one or more OH, F, Cl, Br, I, CN and N3.
- R 1 is H and/or R 2 is H.
- n is an integer selected from 1 to 16, or from 1 to 12, or from 1 to 8, or from 1 to 6, or from 1 to 4, or from 2 to 4.
- Y- is selected from the group consisting of chloride, bromide, phosphate, dimethylphosphate, methylsulfate, ethylsulfate, acetate and lactate.
- one of Z 1 and Z 2 is NR 2 -(CH 2 ) n -NR 4 R 5 R 6+ Y- or O-(CH 2 ) n - NR 4 R 5 R 6+ Y-; and the other one of Z 1 and Z 2 is OR 1 .
- the compound of Formula I-B1 is:
- Y- is an agriculturally acceptable anion that can be selected from the group consisting of: chloride, bromide, phosphate, dimethylphosphate, methylsulfate, ethylsulfate, acetate, citrate, tartrate and lactate.
- Z 1 and Z 2 is NR 2 -(CH 2 CH 2 O) m -R 13 or O-(CH 2 CH 2 O) m -R 13 ;
- Z 1 NR 2 -(CH 2 CH 2 O) m -R 13 or O-(CH 2 CH 2 O) m -R 13 ;
- each R 1 and R 2 is, independently, H, alkyl or substituted alkyl
- R 13 is H, alkyl, substituted alkyl, alkenyl, substituted alkenyl, alkynyl, substituted alkynyl, CO(alkyl), CO(substituted alkyl), CO(alkenyl), CO(substituted alkenyl), CO(alkynyl) or CO(substituted alkynyl);
- n is an integer selected from 1 to 100;
- each R a , R B , R c , R d , R e and R f is, independently, H, alkyl, substituted alkyl, alkenyl, substituted alkenyl, alkynyl or substituted alkynyl;
- M is 2H or a metal species
- substituted alkyl, substituted alkenyl and substituted alkynyl groups are, independently, substituted with one or more OH, F, Cl, Br, I, CN and N3.
- R 1 is H and/or R 12 is H.
- m is an integer selected from 5 to 100, or from 5 to 80, or from 5 to 50, or from 5 to 20, or from 5 to 10.
- R 13 is H, alkyl, alkenyl, CO(alkyl) or CO(alkenyl).
- one of Z 1 and Z 2 is NR 2 -(CH 2 CH 2 O) m -R 13 or O-(CH 2 CH 2 O) m - R 13 ; and the other one of Z 1 and Z 2 is OR 1 .
- the compound of Formula I-B1 is:
- n is an integer selected from 1 to 100;
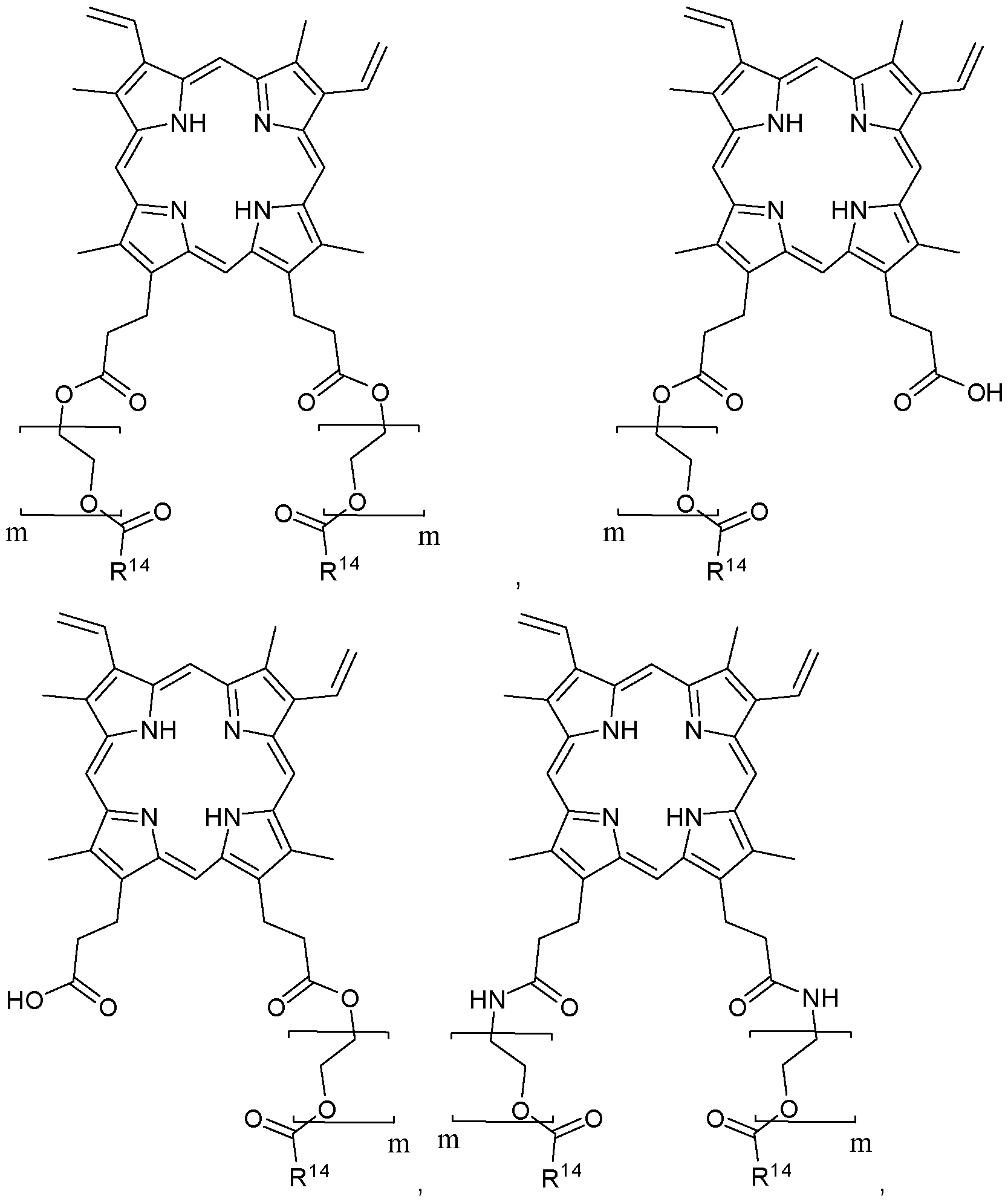
- R 14 is alkyl, alkenyl, alkynyl, substituted alkyl, substituted alkenyl or substituted alkynyl.
- m is an integer selected from 5 to 20 and (CH 2 CH 2 O) m COR 14 is PEGeoo-oleate, PEG4oo-oleate, PEGeoo-allyl or PEG4oo-allyl.
- R 13 is H. In some implementations, (CH 2 CH 2 O)R 13 is PEG6OO or PEG400 ⁇
- the compound of Formula I-B1 is:
- Z 1 and Z 2 is a natural amino acid attached to the compound by its amino group bonded to the alpha carbon;
- Z 1 is a natural amino acid attached to the compound by its amino group bonded to the alpha carbon
- each R 1 and R 2 is, independently, H, alkyl or substituted alkyl
- each R a , R B , R c , R d , R e and R f is, independently, H, alkyl, substituted alkyl, alkenyl, substituted alkenyl, alkynyl or substituted alkynyl;
- M is 2H or a metal species
- substituted alkyl, substituted alkenyl and substituted alkynyl groups are, independently, substituted with one or more OH, F, Cl, Br, I, CN and N3.
- one of Z 1 and Z 2 is a natural amino acid attached to the compound by its amino group bonded to the alpha carbon; and the other one of Z 1 and Z 2 is OR 1 .
- Z 1 is one of the natural amino acids and Z 2 is OH;
- Z 2 is one of the natural amino acids and Z 1 is OH; or
- Z 1 is Glycine, L-Alanine or L-Valine and Z 2 is OH;
- Z 2 is Glycine, L-Alanine or L-Valine and Z 1 is OH; or
- the compound of Formula I-B1 is:
- the compound of Formula I-B1 is:
- the compound of Formula I-B1 is: or an agriculturally acceptable salt thereof.
- the compound of Formula I-B1 is:
- R 17 is the side chain of one of the twenty natural amino acids.
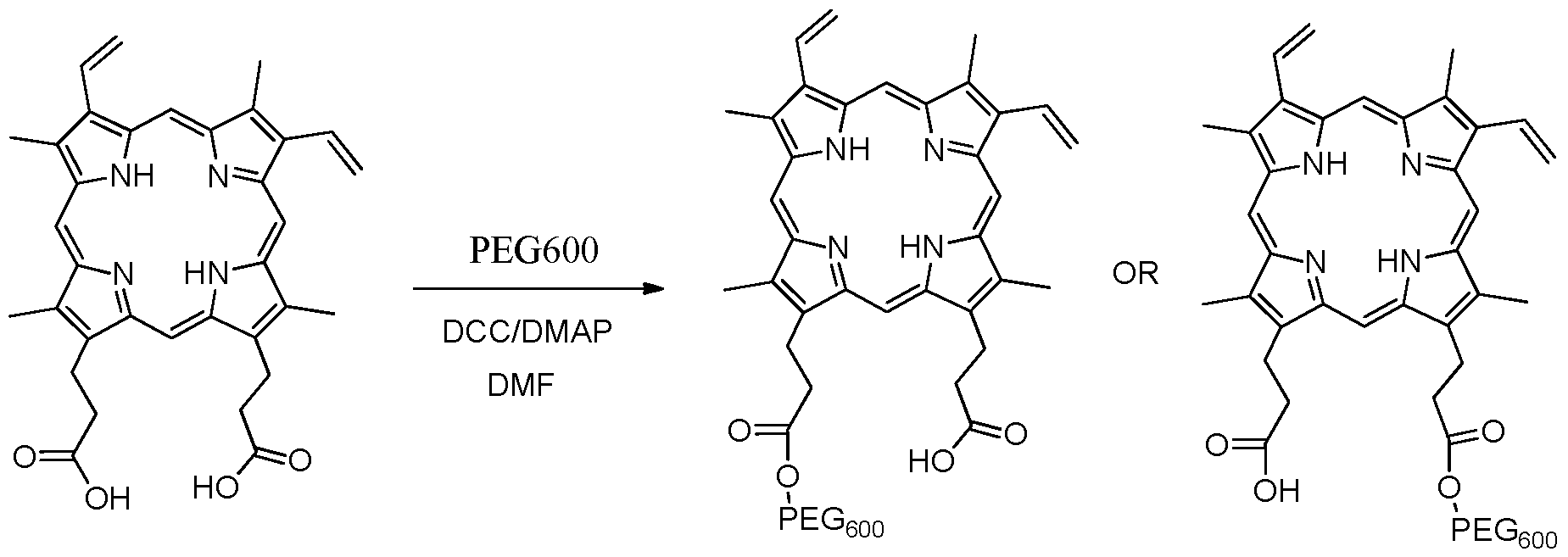
- a compound of Formula II can first be reacted with a coupling agent (e.g., DCC, EDC etc.) before being reacted with an amine, as shown in the scheme below, to give a mixture of mono and bis substituted amide compounds.
- a coupling agent e.g., DCC, EDC etc.
- the compound of Formula II can be esterified using esterification methods known in the art.
- the photosensitizer compound can be applied to a plant in combination with a chelating agent (also referred to herein as a permeabilizing agent).
- a chelating agent also referred to herein as a permeabilizing agent.
- the photosensitizer compound reacts to light by generating ROS, while the chelating agent can increase the overall impact of suppression of the growth of the microbial pathogen, for example by increasing the permeability of the outer membrane of the microbial pathogen to the photosensitizer.
- the term“chelating agent”, as used herein refers generally to a compound that can form several chelating bonds to one or several metals or ions.
- the chelating agent can include at least one carboxylic group, at least one hydroxyl group, at least one phenol group and/or at least one amino group or an agriculturally acceptable salt thereof.
- the chelating agent can include an aminocarboxylic acid compound or an agriculturally acceptable salt thereof.
- the aminocarboxylic acid or agriculturally acceptable salt thereof can include an amino polycarboxyl ic acid or an agriculturally acceptable salt thereof.
- the amino polycarboxyl ic acid can include two amino groups and two alkylcarboxyl groups bound to each amino group.
- the alkylcarboxyl groups can be methylcarboxyl groups.
- the chelating agent is selected from the group consisting of: an aminopolycarboxylic acid, an aromatic or aliphatic carboxylic acid, an amino acid, a phosphonic acid, and a hydroxycarboxylic acid or an agriculturally acceptable salt thereof.
- the methods and compositions described herein include one or more aminopolycarboxylic acid chelating agents.
- aminopolycarboxylic acid chelating agents include, without limitation, ethylenediaminetetraacetic acid (EDTA), diethylenetriaminepentaacetic acid (DTPA), hydroxyethylenediaminetriacetic acid (HEDTA), and ethylenediaminedisuccinate (EDDS), cyclohexanediaminetetraacetic acid (CDTA), N-(2- hydroxyethyl)ethylenediaminetriacetic acid (EDTA-OH) glycol ether diaminetetraacetic acid (GEDTA), alanine diacetic acid (ADA), alkoyl ethylene diamine triacetic acids (e.g., lauroyl ethylene diamine triacetic acids (LED3A)), aspartic acid diacetic acid (ASDA), aspartic acid monoacetic acid, diamino cyclohexane tetra
- EDTA ethylened
- chelating agent is ethylenediaminetetraacetic acid (EDTA) or an agriculturally acceptable salt thereof.
- EDTA ethylenediaminetetraacetic acid
- the aminocarboxylate salt can for example be a sodium or calcium salt.
- chelating agent is polyaspartic acid or an agriculturally acceptable salt thereof (i.e., a polyaspartate), such as sodium polyaspartate.
- a polyaspartate such as sodium polyaspartate.
- the molecular weight of the polyaspartate salt can for example be between 2,000 and 3,000.
- the chelating agent can thus be a polymeric compound, which can include aspartate units, carboxylic groups, and other features found in polyaspartates.
- the polyaspartate can be a co-polymer that has alpha and beta linkages, which may be in various proportions (e.g., 30% alpha, 70% beta, randomly distributed along the polymer chain).
- alpha and beta linkages e.g., 30% alpha, 70% beta, randomly distributed along the polymer chain.
- One non-limiting example of a sodium polyaspartate is Baypure® DS 100.
- chelating agents include EDDS (ethylenediamine- N,N’-disuccinic acid), IDS (iminodisuccinic acid (N-1 ,2-dicarboxyethyl)-D,L-aspartic acid), isopropylamine, triethanolamine, triethylamine, ammonium hydroxide, tetrabutylammonium hydroxide, hexamine, GLDA (L-glutamic acid N,N-diacetic acid), or agriculturally acceptable salts thereof.
- the chelating agent can be metallated or non-metallated.
- IDS can be used as a tetrasodium salt of IDS (e.g., tetrasodium iminodisuccinate), which can be Baypure® CX100.
- EDDS can be used as a trisodium salt of EDDS.
- GLDA can be used as a tetrasodium salt of GLDA.
- the chelating agent can include one or more amino acid chelating agents.
- amino acid chelating agents include, without limitation, alanine, arginine, asparagine, aspartic acid, glutamic acid, glutamine, glycine, histidine, isoleucine, leucine, lysine, methionine, proline, serine, threonine, tyrosine, valine, or salts (for example, the sodium salts, calcium salts and/or potassium salts) and combinations thereof.
- the chelating agent can include one or more aromatic or aliphatic carboxylic acid chelating agents.
- aromatic or aliphatic carboxylic acid chelating agents include, without limitation, oxalic acid, succinic acid, pyruvic acid malic, acid, malonic acid, salicylic acid, and anthranilic acid, and salts (for example, the sodium salts, calcium salts and/or potassium salts) thereof.
- the methods and compositions described herein include one or more polyphenol chelating agents.
- a polyphenol chelating agent is tannins such as tannic acid.
- the chelating agent can include one or more hydroxycarboxylic acid chelating agents.
- the hydroxycarboxylic acid type chelating agents include, without limitation, malic acid, citric acid, glycolic acid, heptonic acid, tartaric acid and salts (for example, the sodium salts, calcium salts and/or potassium salts) thereof.
- the one or more chelating agents can be provided as the free acid, as an agriculturally acceptable salt, or as combinations thereof.
- each of one or more the chelating agent(s) is applied as the free acid.
- the chelating agent(s) can be applied as a salt.
- Exemplary salts include sodium salts, potassium salts, calcium salts, ammonium salts, amine salts, amide salts, and combinations thereof.
- at least one of the chelating agents is applied as a free acid, and at least one of the chelating agents is applied as a salt.
- the photosensitizer compound can be applied to a plant in combination with one or more agriculturally suitable adjuvants.
- Each of the one or more agriculturally suitable adjuvants can be independently selected from the group consisting of one or more activator adjuvants (e.g., one or more surfactants; e.g., one or more oil adjuvants, e.g., one or more penetrants) and one or more utility adjuvants (e.g., one or more wetting or spreading agents; one or more humectants; one or more emulsifiers; one or more drift control agents; one or more thickening agents; one or more deposition agents; one or more water conditioners; one or more buffers; one or more anti-foaming agents; one or more UV blockers; one or more antioxidants; one or more fertilizers, nutrients, and/or micronutrients; and/or one or more herbicide safeners).
- activator adjuvants e.g., one or more surfact
- the photosensitizer compound can be applied to a plant in combination with oil.
- the oil can be selected from the group consisting of a mineral oil (e.g., paraffinic oil), a vegetable oil, an essential oil, and a mixture thereof.
- a mineral oil e.g., paraffinic oil
- a vegetable oil e.g., a vegetable oil
- an essential oil e.g., a mixture thereof.
- combining the photosensitizer compound with an oil can improve solubility of the photosensitizer compound when in contact with the plant.
- the oil can be added with the photosensitizer compound, or separately, in the presence or absence of a carrier fluid such as water.
- Non-limiting examples of vegetable oils include oils that contain medium chain triglycerides (MCT), or oil extracted from nuts.
- Other non-limiting examples of vegetable oils include coconut oil, canola oil, soybean oil, rapeseed oil, sunflower oil, safflower oil, peanut oil, cottonseed oil, palm oil, rice bran oil or mixtures thereof.
- Non-limiting examples of mineral oils include paraffinic oils, branched paraffinic oils, naphthenic oils, aromatic oils or mixtures thereof.
- paraffinic oils include various grades of poly-alpha-olefin (PAO).
- PAO poly-alpha-olefin
- the paraffinic oil can include HT60TM, HT100TM, High Flash Jet, LSRDTM, and N65DWTM.
- the paraffinic oil can include a paraffin having a number of carbon atoms ranging from about 12 to about 50, or from about 16 to 35. In some scenarios, the paraffin can have an average number of carbon atoms of 23. In some implementations, the oil can have a paraffin content of at least 80 wt%, or at least 90 wt%, or at least 99 wt%.
- the photosensitizer compound and the oil can be added sequentially or simultaneously. When added simultaneously, the nitrogen-bearing macrocyclic compound and the oil can be added as part of the same composition or as part of two separate compositions. In some implementations, the nitrogen-bearing macrocyclic compound and the oil can be combined in an oil-in-water emulsion. That is, the combination can include the nitrogen-bearing macrocyclic compound combined with the oil and water so that the combination is formulated as an oil-in-water emulsion.
- the oil-in-water emulsion can also include other additives such as a chelating agent, a surfactant, or combinations thereof.
- oil-in-water emulsion refers to a mixture in which one of the oil (e.g., the paraffinic oil) and water is dispersed as droplets in the other (e.g., the water).
- an oil-in-water emulsion is prepared by a process that includes combining the paraffinic oil, water, and any other components and the paraffinic oil and applying shear until the emulsion is obtained.
- an oil-in-water emulsion is prepared by a process that includes combining the paraffinic oil, water, and any other components in the mixing tank and spraying through the nozzle of a spray gun.
- the photosensitizer compound is part of a composition that includes a carrier fluid.
- a suitable carrier fluid can allow obtaining a stable solution, suspension and/or emulsion of the components of the composition in the carrier fluid.
- the carrier fluid is water.
- the carrier fluid is a mixture of water and other solvents or oils that are non-miscible or only partially soluble in water.
- a combination of photosensitizer compound and oil can be used to inhibit growth of a microbial pathogen in a plant.
- the combination can be an oil-in- water emulsion, where the surfactant is selected such that the photosensitizer compound is maintained in dispersion in the oil-in-water emulsion for delivery to the plant.
- the combination can include a surfactant (also referred to as an emulsifier).
- the surfactant can be selected from the group consisting of an ethoxylated alcohol, a polymeric surfactant, a fatty acid ester, a poly(ethylene glycol), an ethoxylated alkyl alcohol, a monoglyceride, an alkyl monoglyceride, an amphipathic glycoside, and a mixture thereof.
- the fatty acid ester can be a sorbitan fatty acid ester.
- the surfactant can include a plant derived glycoside such as a saponin.
- the surfactant can be present as an adjuvant to aid coverage of plant foliage.
- the surfactant can be an acceptable polysorbate type surfactant (e.g., Tween 80), a nonionic surfactant blend (e.g., AltoxTM 3273), or another suitable surfactant.
- the poly(ethylene glycol) can include a poly(ethylene glycol) of Formula R 15 -O-(CH 2 CH 2 O) f -R 16 , wherein: each R 15 and R 16 is each, independently, H, alkyl, substituted alkyl, aryl, substituted aryl, CO(alkyl) or CO(substituted alkyl); and f is an integer selected from 1 to 100; wherein the substituted alkyl groups are, independently, substituted with one or more F, Cl, Br, I, hydroxy, alkenyl, CN and N 3 .
- compositions including a photosensitizer compound
- the photosensitizer compounds and the other agents can be provided to a plant separately or together as part of the same composition.
- the components of the compositions can be packaged in a concentrated form, without carrier fluid, and the carrier fluid (e.g ., water) can be added to form the composition directly by the operator that can then apply the composition to plants.
- the composition can be provided to have certain concentrations and relative proportions of components.
- the composition can have between about 100 nM and about 50 mM, between about 5 micromolar and about 10 mM, between about 1 micromolar and about 1000 micromolar, between about 5 micromolar and about 200 micromolar of the photosensitizer compound, between about 10 micromolar and about 150 micromolar of the nitrogen-bearing macrocyclic compound, between about 25 micromolar and about 100 micromolar of the nitrogen-bearing macrocyclic compound, or between about 50 micromolar and about 75 micromolar of the photosensitizer compound.
- the composition can also include between about 2 micromolar and about 10,000 micromolar of the chelating agent, between about 5 micromolar and about 5,000 micromolar of the chelating agent, between about 10 micromolar and about 1 ,000 micromolar of the chelating agent, between about 25 micromolar and about 500 micromolar of the chelating agent, or between about 50 micromolar and about 100 micromolar of the chelating agent.
- the relative proportion, by weight, of the nitrogen-bearing macrocyclic compound and the chelating agent in the composition can be between about 50: 1 and about 1 : 1000, between about 20: 1 and about 1 :500, between about 10:1 and about 1 : 100, or between about 1 : 1 and about 1 : 10.
- the photosensitizer compound and the oil can be applied in a relative proportion, by weight, between about 50: 1 and about 1 : 1000, between about 20:1 and about 1 :500, between about 10: 1 and about 1 : 100, or between about 1 : 1 and about 1 : 10.
- the composition including the photosensitizer compound can be applied to plants in various ways.
- the composition can be prepared to include the photosensitizer compound, a chelating agent as well as a delivery fluid, such as water or a water-oil emulsion.
- the composition can be applied to the plant by spraying, misting, sprinkling, pouring, or any other suitable method.
- the anti-microbial composition can be applied to the foliage, roots and/or stem of the plant. Other additives can also be included in the anti-microbial composition, and other application methods can also be performed.
- the plants on which the composition is applied can be outdoors or indoors ( e.g ., greenhouse) where they are exposed to natural sunlight, or in an indoor location where they are exposed to artificial light.
- the exposure to the incident light is provided such that the photosensitizer compound can generate ROS that, in turn, facilitate disruption of microbial growth.
- the photosensitizer compound can be used to treat seeds or seedlings.
- the treatment of seeds or seedlings can stimulate germination and growth, and/or can increase resistance of the plant to abiotic stresses.
- the seeds or seedlings can be treated with the photosensitizer compound prior to being planted into a growing medium.
- the seeds or seedlings can be treated with the photosensitizer compound after being planted into a growing medium.
- the photosensitizer compound can be directly surface-coated onto the seeds, applied to seedlings roots or seedlings leafs (foliar application on seedlings).
- a solution or emulsion containing the photosensitizer compound can be directly sprayed onto the seeds or seedlings.
- the seeds or seedlings can be dipped into a solution or emulsion containing the photosensitizer compound.
- the root of the seedling can be dipped into a solution or emulsion containing the photosensitizer compound.
- the seeds can be placed into a container, and a solution containing the photosensitizer compound can be introduced into the container. The container can then be shaken for an appropriate period (e.g., between about 1 minute to several minutes) such that the solution contacts the seeds. The shaken seeds can then be dried (e.g., air dried) prior to being planted.
- the photosensitizer compound can be applied once, twice, or more than twice to seeds or seedlings, using various modes of application.
- the seeds can be treated after having been planted into a growing medium.
- the seeds and/or seedlings can be treated prior to having been planted and after having been planted (e.g., in furrow treatment and/or foliar application).
- the seed can be treated prior to having been planted and/or after having been planted, and the ensuing seedling can be further treated (e.g., root treatment and/or foliar treatment).
- the microbial pathogens to which the composition including the photosensitizer compound can be applied include fungal and bacterial pathogens.
- the composition can be referred to as an“anti-microbial composition”.
- the fungal pathogens to which the anti-microbial composition can be applied include Alternaria solani, which can infect plants such as tomatoes and potatoes; Botrytis cinerea, which can infect grapes, as well as soft fruits and bulb crops; or Sclerotinia homoeocarpa, which can commonly infect turfgrasses. Other fungal pathogens in the Alternaria, Botrytis or Sclerotinia genera can also receive application of the anti-microbial composition.
- the anti microbial composition can be applied to plants that are affected or susceptible to pathogens that cause various plant diseases, e.g., Colletotrichum, Fusarium, Puccinia, Erysiphaceae, Cercospora, Rhizoctonia, Bipolaris, Microdochium, Venturia inaequalis, Monilinia fructicola, Gymnosporangium juniperi-virginianae, Plasmodiophora brassicae, Ustilago zeae, Phytophthora, Pythium, Fusarium oxysporum, Phytophthora infestans, Taphrina deformans, Powdery Mildew, Phragmidium spp., or other fungal pathogens.
- plant diseases e.g., Colletotrichum, Fusarium, Puccinia, Erysiphaceae, Cercospora, Rhizoctonia, Bipolaris, Microdochium, Venturia inaequalis,
- the bacterial pathogens to which the anti-microbial composition can be applied include gram-negative bacteria, such as Erwinia amylovara, or other bacterial pathogens in the genus Erwinia that can infect woody plants.
- E. amylovara causes fire blight on various plants, including pears, apples, and other Rosaceae crops.
- the anti-microbial composition can be applied to plants that are affected or susceptible to pathogens that cause various plant diseases, e.g., Pseudomonas, Xanthomonas, Agrobacterium, Curtobacterium, Streptomyces, E. Coli, Xylella fastidiosa (which causes Olive Quick Decline Syndrome (OQDS) disease), or other bacterial pathogens.
- Pseudomonas e.g., Pseudomonas, Xanthomonas, Agrobacterium, Curtobacterium, Streptomyces, E. Coli, Xylella fastidio
- the anti-microbial compositions described herein can have various inhibitory effects on the microbial pathogens depending on the type of plant and pathogen as well as the state of microbial infection. While herein it is described that the anti-microbial composition can inhibit microbial pathogen growth on a plant, such expressions should not be limiting but should be understood to include suppression of microbial pathogens, prevention against microbial pathogens, killing of microbial pathogens or generally increase toxicity toward microbial pathogens. Abiotic stresses
- the photosensitizer compounds and compositions of the present description can be used to increase tolerance of plants to one or more abiotic stresses such as photooxidative conditions, drought (water deficit), excessive watering (flooding, and submergence), extreme temperatures (chilling, freezing and heat), extreme levels of light (high and low), radiation (UV-B and UV-A), salinity due to excessive Na + (sodicity), chemical factors (e.g., pH), mineral (metal and metalloid) toxicity, deficiency or excess of essential nutrients, gaseous pollutants (ozone, sulfur dioxide), wind, mechanical factors, and other stressors.
- abiotic stresses such as photooxidative conditions, drought (water deficit), excessive watering (flooding, and submergence), extreme temperatures (chilling, freezing and heat), extreme levels of light (high and low), radiation (UV-B and UV-A), salinity due to excessive Na + (sodicity), chemical factors (e.g., pH), mineral (metal and metalloid) toxicity,
- the photosensitizer compound when the abiotic stress is cold stress, application of the photosensitizer compound, alone or in combination with additives such as an oil, a surfactant and/or a chelating agent, can improve cold hardiness of the plant. That is, application of the photosensitizer compound can allow the plant to withstand temperature conditions that are colder than would typically be experienced in the plant’s optimal or native growing conditions.
- Various types of cold stress are possible, such as unexpected frost (for example an early fall frost when healthy crop, fruit, grain, seeds or leaves are still present on the plant, or a late spring frost that occurs after spring plant growth has begun), a cooler than average growing season, colder than native winter conditions, minimal winter snow cover, ice accumulation, etc.
- a cold stress condition for one plant may not be a cold stress condition for another plant.
- a cold stress condition for a zone 9 plant may in fact be a native growing condition for a zone 8 plant.
- the depth of snow cover required for survival of one type of plant may not be required for a second type of plant. It is therefore understood that various types of cold stress are possible, depending on the type of plant in question.
- the photosensitizer compound, compositions or combinations described herein may be used to protect plants, including woody plants, non-woody plants and turfgrasses, from frost injury.
- the frost can be an early frost, for example before harvest, after harvest and before dormancy.
- the frost can be a late frost, for example after budding.
- the cold damage can also be winter kill induced by winter temperatures, which may result in a loss of viable branches or shoots and lead to plant mortality.
- Plants treated by the photosensitizer compound, compositions or combinations described herein can be frost or cold sensitive plants, in that they are naturally susceptible to frost, freezing or cold damage or injury in economically or aesthetically significant amounts.
- Increasing resistance to cold stress can be exemplified by a delayed onset of dormancy.
- Plant dormancy can be triggered by a drop in temperature, e.g., the onset of cold stress.
- dormancy of the plant can be delayed until triggered by a further drop in temperature.
- the photosensitizer compound, compositions or combinations described herein can be used periodically (e.g., at 2 or 3-week intervals starting with spring at breaking the dormancy) and/or by applying one or more treatments (e.g., 2 in the fall), to provide a response in reducing or delaying the dormancy period of certain plants.
- reducing dormancy period refers to a plant that has a reduced dormancy period or extended growing period relative to a control, e.g., a non-treated plant.
- the harvesting step may be carried out one week, one month, two months or more after the last application of the photosensitizer compound, compositions or combinations described herein, with the active agent still being effective to reduce the effects of cold stress on the plant during the intervening period.
- resistance to cold stress includes resistance to early or late frost, or winter damage.
- the photosensitizer compound, compositions or combinations described herein can be used to protect early growth from cold during fluctuations in temperature (e.g., in early spring).
- the photosensitizer compound, compositions or combinations described herein can be used to protect plants from cold during the cold months (e.g., in winter).
- the photosensitizer compound, compositions or combinations described herein can be applied by soil drenching and/or foliar application (e.g., sprayed until run-off) at the onset or prior to exposure to the low temperature (e.g., fall when the trees have full healthy and vigorous foliage).
- the photosensitizer compound, compositions or combinations described herein can be applied by soil drenching and/or foliar application (e.g., sprayed until run-off) during late fall and winter (e.g., for warm climates).
- the photosensitizer compound, compositions or combinations described herein can be applied by soil drenching in the late fall following by a foliar application ⁇ e.g., sprayed until run-off) in the winter in order to reach maximum hardiness.
- the photosensitizer compound, compositions or combinations described herein can be applied 1-4 times at a 1 to 6-month interval (e.g., every 2 to 3 months). Further treatments may be applied in the spring and/or during the growing season to improve resistance to subsequent cold stress conditions.
- the photosensitizer compound, compositions or combinations described herein can improve tolerance to high temperatures during the growing season. That is, application of the photosensitizer compound, compositions or combinations described herein can allow the plant to withstand temperature conditions that are higher than would typically be experienced in the plant’s optimal or native growing conditions. Heat stress can have various causes, such as lack of shade for plants that typically require shaded growing conditions, or higher than normal soil and air temperatures.
- the photosensitizer compound, compositions or combinations described herein can improve tolerance to stressful light condition during periods of increased generation of reactive oxygen species. That is, application of the photosensitizer compound, compositions or combinations described herein can allow the plant to withstand light exposure conditions (e.g., ultraviolet irradiation conditions) that are higher than would typically be experienced in the plant’s optimal or native growing conditions.
- Photooxidative stress can have various causes, such as high light conditions or certain types of lighting that induce formation of free radicals.
- Shade stress or“low light (LL) stress” can be a problem that influences plant growth and quality.
- abiotic stress is shade stress
- application of the photosensitizer compound, compositions or combinations described herein can improve shade hardiness of the plant. That is, application of the photosensitizer compound, compositions or combinations described herein can allow the plant to withstand shady conditions for plants whose optimal or native growing conditions typically require partial or full sun exposure.
- shade stress are possible, such as a prolonged period of cloudy weather, excessive growth of adjacent plants or trees that cast shade onto the plant, or lack of availability of a sunny planting location.
- Shade can be a periodic problem. For example, during certain months of the year, a structure situated near a plant may cast a shadow on the plant, causing a shade stress. As the earth moves over the course of a year, the structure may no longer cast the shadow on the plant for another series of months and then the situation can be repeated during the next annual cycle. In such instances, the photosensitizer compound, compositions or combinations described herein can be applied to the plant prior to onset of the period of shade stress and can also be applied during the period of shade stress. The damage to the plant that would typically result on account of the period of shade stress can be prevented or reduced.
- Shade conditions are not considered to be an abiotic stress condition for many types of plants, as some plants have a requirement for shade as part of their optimal growing conditions. It should also be noted that what constitutes a shade stress condition for one plant may not be a shade stress condition for another plant.
- Drought can be defined as the absence of rainfall or irrigation for a period of time sufficient to deplete soil moisture and injure plants. Drought stress results when water loss from the plant exceeds the ability of the plant's roots to absorb water and/or when the plant's water content is reduced enough to interfere with normal plant processes. The severity of the effect of a drought condition may vary between plants, as the plant’s need for water may vary by plant type, plant phenological stage, plant age, root depth, soil quality, etc. [0184] The photosensitizer compound, compositions or combinations described herein can be applied to a plant prior to onset of a drought and/or during a drought.
- Increasing resistance can include maintaining or increasing a quality of the plant as compared to an untreated plant subjected to the same drought stress.
- Increasing resistance can include reducing the degradation in quality of the plant, as compared to an untreated plant subjected to the same drought stress. If plants do not receive adequate rainfall or irrigation, the resulting drought stress can reduce growth more than all other environmental stresses combined.
- Salts can be naturally present in the growing environment of a plant.
- Salinity stress refers to osmotic forces exerted on a plant when the plant is growing in a saline soil or under other excessively saline conditions.
- plants growing near a body of salt water can be exposed to salt present in the air or in water used to water the plants.
- salt applied to road, sidewalk and driveway surfaces during the winter for improved driving conditions can be transferred and/or leach into the soil of plants growing in the proximity.
- Such increased salt content in a growing environment of the plant can result in salinity stress, which can damage the plant.
- a plant that is subjected to transplanting from one growing environment to another, e.g., from a pot to flower bed or garden, can be subjected to transplant shock stress as a result of exposure to new environmental conditions such as wind, direct sun, or new soil conditions.
- Application of the photosensitizer compound, compositions or combinations described herein to the roots of the plant can reduce the impact to the plant caused by the transplanting. In some scenarios, stunting of plant growth and/or development of a transplanted plant can be reduced or prevented by application of the photosensitizer compound, compositions or combinations described herein.
- transplant shock stress condition for one plant may not be a transplant shock stress condition for another plant.
- the compounds and combinations of the present description can be used to protect the plant from a plant pest.
- plant pest or“pest”, as used herein, refers to insects and/or their larvae, which are known to or have the potential to cause damage to the plant.
- the compounds and combinations of the present description can induce photoinduced mortality in plant pests.
- the insect pests are selected from the order of Hemiptera (groups of aphids, whiteflies, scales, mealybugs, stink bugs), Coleoptera (groups of beetles), Lepidoptera (groups of butterflies, moths), Diptera (groups of flies), Thysanoptera (group of thrips), Orthoptera (group of grasshoppers, locusts), Hymenoptera (groups of wasps, ants), Blattodea (groups of cockroaches and termites) and mite pests (spider mites).
- Hemiptera groups of aphids, whiteflies, scales, mealybugs, stink bugs
- Coleoptera groups of beetles
- Lepidoptera groups of butterflies, moths
- Diptera groups of flies
- Thysanoptera group of thrips
- Orthoptera group of grasshoppers, locusts
- Non-limiting examples of insect pests include: larvae of the order Lepidoptera, such as armyworms, (e.g., beet armyworm ( Spodoptera exigua )), cutworms, loopers, (e.g., cabbage looper ( Trichoplusia ni)) and heliothines, in the family Noctuidae (e.g., fall armyworm ( Spodoptera fugiperda J. E.
- armyworms e.g., beet armyworm ( Spodoptera exigua )
- cutworms e.g., cabbage looper ( Trichoplusia ni)
- loopers e.g., cabbage looper ( Trichoplusia ni)
- heliothines in the family Noctuidae (e.g., fall armyworm ( Spodoptera fugiperda J. E.
- borers, casebearers, webworms, coneworms, cabbageworms and skeletonizers from the family Pyralidae e.g., European corn borer ( Ostrinia nubilalis Hubner)), navel orangeworm ( Amyelois transitella Walker), corn root webworm ( Crambus caliginosellus Clemens), and sod webworms (Pyralidae: Crambinae) such as sod webworm ( Herpetogramma licarsisalis Walker), leafrollers, budworms, seed worms, and fruit worms in the family Tortricidae (e.g., codling moth ( Cydia pomonella Linnaeus)), grape berry moth ( Endopiza viteana Clemens), oriental
- psyllids from the family Psyllidae( e.g. Citrus psyllid Diaphorina citri)), whiteflies from the family Aleyrodidae (silverleaf whitefly ( Bemisia argentifolii )); aphids from the family Aphididae, such as cotton melon aphid ( Aphis gossypii), pea aphid ( Acyrthisiphon pisum Harris), cowpea aphid ( Aphis craccivora Koch), black bean aphid ( Aphis fabae Scopoli), melon or cotton aphid ( Aphis gossypii Glover), apple aphid (Aphis pomi De Geer), spirea aphid ( Aphis spiraecola Patch), foxglove aphid ( Aula
- Agronomic pests also include invertebrate arthropods sush as mites from the family Tetranychidae: twospotted spider mite (e.g. Tetranychus urticae Koch), flat mite from family Rutacea (e.g., citrus flat mite ( Brevipalpus lewisi McGregor); rust and bud mites from the family Eriophyidae and other foliar feeding mites.
- Tetranychidae twospotted spider mite (e.g. Tetranychus urticae Koch), flat mite from family Rutacea (e.g., citrus flat mite ( Brevipalpus lewisi McGregor); rust and bud mites from the family Eriophyidae and other foliar feeding mites.
- Economically important agricultural pests nematodes e.g., root knot nematodes in the genus Meloidogyne, lesion nematodes in the genus Pratylenchus, and stubby root nematodes in the genus Trichodorus
- members of the classes Nematoda, Cestoda, Trematoda, and Acanthocephala from orders of Strongylida, Ascaridida, Oxyurida, Rhabditida, Spirurida, and Enoplida e.g., root knot nematodes in the genus Meloidogyne, lesion nematodes in the genus Pratylenchus, and stubby root nematodes in the genus Trichodorus
- the photosensitizer compounds of the present description can be applied to the plant before, at or after infestation of the plant by the insect pests.
- the photosensitizer compounds of the present description can be used as insecticides by applying them to insects (i.e. , without applying the photosensitizers to a plant).
- the present description therefore also provides a method for controlling insect population. The method includes applying to the insects a photosensitizer compound of the present description; and exposing the insects to light to activate the photosensitizer compound and generate reactive oxygen species.
- applying the photosensitizer to the insects can include indirectly applying the photosensitizer to the insect pests (e.g., by applying the photosensitizer to a food source that is then fed to the insects) and/or directly applying the photosensitizer to the insect pests (e.g., by directly contacting the insects with the photosensitizer, such as by spraying a composition including the photosensitizer onto the insects).
- the photosensitizer compounds and compositions of the present description can be used for various types of plants.
- the plant can be a non-woody crop plant, a woody plant or a turfgrass.
- the plant can be selected from the group consisting of a crop plant, a fruit plant, a vegetable plant, a legume plant, a cereal plant, a fodder plant, an oil seed plant, a field plant, a garden plant, a green-house plant, a house plant, a flower plant, a lawn plant, a turfgrass, a tree such as a fruit-bearing tree, and other plants that may be affected by microbial pathogens and/or one or more abiotic stress.
- the plant is a crop plant selected from the group consisting of sugar cane, wheat, rice, corn (maize), potatoes, sugar beets, barley, sweet potatoes, cassava, soybeans, tomatoes, and legumes (beans and peas).
- the plant is a tree selected from the group consisting of deciduous trees and evergreen trees.
- trees include, without limitation, maple trees, fruit trees such as citrus trees, apple trees, and pear trees, an oak tree, an ash tree, a pine tree, and a spruce tree.
- the plant is a shrub.
- the plant is a fruit or nut plant.
- Non-limiting examples of such plants include: acerola (barbados cherry), atemoya, carambola (star fruit), rambutan, almonds, apricots, cherries, nectarines, peaches, pistachio, apples, avocados, bananas, plantains, figs, grapes, mango, olives, papaya, pears, pineapple, plums, strawberries, grapefruit, lemons, limes, oranges (e.g., navel and Valencia), tangelos, tangerines, mandarins and plants from the berry and small fruits plant group.
- the plant is a vegetable plant.
- Non-limiting examples of such plants include: asparagus, bean, beets, broccoli, Chinese broccoli, broccoli raab, brussels sprouts, cabbage, cauliflower, Chinese cabbage (e.g., bok choy and mapa), Chinese mustard cabbage (gai choy), cavalo broccoli, collards, kale, kohlrabi, mizuna, mustard greens, mustard spinach, rape greens, celery, chayote, Chinese waxgourd, citron melon, cucumber, gherkin, hyotan, cucuzza, hechima, Chinese okra, balsam apple, balsam pear, bitter melon, Chinese cucumber, true cantaloupe, cantaloupe, casaba, crenshaw melon, golden pershaw melon, honeydew melon, honey galls, mango melon, Persian melon, pumpkin, summer squash, winter squash, watermelon, dashe
- the plant is a flowering plant, such as roses, flowering shrubs or ornamentals.
- a flowering plant such as roses, flowering shrubs or ornamentals.
- Non-limiting examples of such plants include: flowering and foliage plants including roses and other flowering shrubs, foliage ornamentals & bedding plants, fruit bearing trees such as apple, cherry, peach, and pear trees, non-fruit-bearing trees, shade trees, ornamental trees, and shrubs (e.g ., conifers, deciduous and broadleaf evergreens & woody ornamentals).
- the plant is a houseplant.
- Non-limiting examples of such plants include: chrysanthemum, dieffenbachia, dracaena, ferns, gardenias, geranium, jade plant, palms, philodendron, and schefflera.
- the plant is a plant grown in a greenhouse.
- Non- limiting examples of such plants include: ageratum, crown of thorns, dieffenbachia, dogwood, dracaena, ferns, ficus, holly, lisianthus, magnolia, orchid, palms, petunia, poinsettia, schefflera, sunflower, aglaonema, aster, azaleas, begonias, browallia, camellias, carnation, celosia, chrysanthemum, coleus, cosmos, crepe myrtle, dusty miller, easier lilies, fuchsia, gardenias, gerbera, hellichrysum, hibiscus foliage, hydrangea, impatiens, jade plant, marigold, new guinea, impatiens, nicotonia, philodendron, portulaca, re
- the combinations can exhibit a synergistic response for inhibiting growth of microbial pathogens in plants.
- the terms“synergy” or “synergistic”, as used herein refer to the interaction of two or more components of a combination (or composition) so that their combined effect is greater than the sum of their individual effects. This may include, in the context of the present description, the action of two or more of the nitrogen-bearing macrocyclic compounds, the oil, and the chelating agent. In some scenarios, the nitrogen-bearing macrocyclic compound and the oil can be present in synergistically effective amounts.
- the nitrogen-bearing macrocyclic compound and the chelating agent can be present in synergistically effective amounts. In some scenarios, the oil and the chelating agent can be present in synergistically effective amounts. In some scenarios, the nitrogen-bearing macrocyclic compound, the oil and the chelating agent can be present in synergistically effective amounts.
- the two components are said to be present in synergistically effective amounts when the observed efficacy is higher than the expected efficacy.
- the product contained about 50% di-substituted compound and 50% mono- substituted compound.
- the ratio was brought up to 80% di-substituted compound and 20% mono-substituted compound.
- PP IX-mono-DMAE amide MS-ESI (HRMS) m/z calculated for C38 H44 N6 O3 (M+H + ): 633.35. Found: 633.087.
- PP IX-di-DMAE amide MS-ESI (HRMS) m/z calculated for C42 H54 N8 O2 (M): 703.44 Found: 703.611.
- the filtrate was slowly dripped into methyl tert-butyl ether (200 ml) at 0°C and filtered to obtain PP IX-mono- L-Analine as a mixture containing mainly the two L-Alanine mono-substituted PP IX compounds.
- control of the gram-negative bacterial plant pathogen Pseudomonas syringae pv. tabaci with PP IX and modified PP IX was assessed, with and without chelating agents.
- Treatments were prepared in Phosphate Buffered Saline (PBS) in 96 well plates at desired concentrations. A bacterial suspension was inoculated into the PBS and incubated at 28°C in the dark for 30 minutes. After 30 minutes, the 96 well plate was placed under illumination for 1 hour (at 21 °C).
- PBS Phosphate Buffered Saline
- bacterial suspensions were serially diluted and 10 mL of each dilution is spread uniformly on Tryptic Soy Agar (TSA) plates and placed in the dark in an incubator at 28°C for 48 hours. After 48 hours, bacterial colonies were counted, and the results were log transformed (log colony forming units (CFU)/ml_). The relative inactivation was determined by taking the difference between logCFU(PBS control) and logCFU(treatments). Sample Illumination was provided by LED lights (Heliospectra RX30) emitting about 1000 mmol/m 2 /s photosynthetically active radiation (PAR). The results are summarized in Table 1 below.
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Pest Control & Pesticides (AREA)
- Plant Pathology (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Environmental Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Dentistry (AREA)
- Agronomy & Crop Science (AREA)
- Health & Medical Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Organic Chemistry (AREA)
- Mycology (AREA)
- Insects & Arthropods (AREA)
- Microbiology (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
- Cultivation Of Plants (AREA)
- Nitrogen Condensed Heterocyclic Rings (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
Abstract
Description
Claims
Priority Applications (11)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2021547492A JP7588289B2 (en) | 2019-02-15 | 2020-02-14 | Protoporphyrin IX derivatives for promoting plant health and uses thereof |
| BR112021016109A BR112021016109A2 (en) | 2019-02-15 | 2020-02-14 | Protoporphyrin ix derivatives and their use to improve plant health |
| CN202080028335.8A CN113677204B (en) | 2019-02-15 | 2020-02-14 | Use of protoporphyrin IX derivatives to improve plant health |
| CA3128733A CA3128733C (en) | 2019-02-15 | 2020-02-14 | The use of protoporphyrin ix derivatives to improve the health of plants |
| US17/431,294 US12207655B2 (en) | 2019-02-15 | 2020-02-14 | Protoporphyrin IX derivatives and use thereof to improve the health of plants |
| ES20755957T ES3051057T3 (en) | 2019-02-15 | 2020-02-14 | The use of protoporphyrin ix derivatives to improve the health of plants |
| AU2020222515A AU2020222515B2 (en) | 2019-02-15 | 2020-02-14 | The use of protoporphyrin IX derivatives to improve the health of plants |
| HRP20251337TT HRP20251337T1 (en) | 2019-02-15 | 2020-02-14 | The use of protoporphyrin ix derivatives to improve the health of plants |
| MX2021009781A MX2021009781A (en) | 2019-02-15 | 2020-02-14 | DERIVATIVES OF PROTOPORPHIRIN IX AND THEIR USE TO IMPROVE THE HEALTH OF PLANTS. |
| EP20755957.6A EP3923729B1 (en) | 2019-02-15 | 2020-02-14 | The use of protoporphyrin ix derivatives to improve the health of plants |
| ZA2021/05283A ZA202105283B (en) | 2019-02-15 | 2021-07-26 | The use of protoporphyrin ix derivatives to improve the health of plants |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201962806084P | 2019-02-15 | 2019-02-15 | |
| US62/806,084 | 2019-02-15 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2020163964A1 true WO2020163964A1 (en) | 2020-08-20 |
Family
ID=72043778
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/CA2020/050197 Ceased WO2020163964A1 (en) | 2019-02-15 | 2020-02-14 | The use of protoporphyrin ix derivatives to improve the health of plants |
Country Status (12)
| Country | Link |
|---|---|
| US (1) | US12207655B2 (en) |
| EP (1) | EP3923729B1 (en) |
| JP (1) | JP7588289B2 (en) |
| CN (1) | CN113677204B (en) |
| AU (1) | AU2020222515B2 (en) |
| BR (1) | BR112021016109A2 (en) |
| CL (1) | CL2021002023A1 (en) |
| ES (1) | ES3051057T3 (en) |
| HR (1) | HRP20251337T1 (en) |
| MX (1) | MX2021009781A (en) |
| WO (1) | WO2020163964A1 (en) |
| ZA (1) | ZA202105283B (en) |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11910795B2 (en) | 2013-03-15 | 2024-02-27 | Suncor Energy Inc. | Natural indole auxin and aminopolycarboxylic acid herbicidal compositions |
| US12207655B2 (en) | 2019-02-15 | 2025-01-28 | Nutrien Ag Solutions (Canada) Inc. | Protoporphyrin IX derivatives and use thereof to improve the health of plants |
| WO2026012814A1 (en) | 2024-07-10 | 2026-01-15 | Basf Se | Compositions and methods to enhance crop yield and plant health |
| US12583872B2 (en) | 2019-01-25 | 2026-03-24 | Nutrien Ag Solutions (Canada) Inc. | Photosensitizer compounds, methods of manufacture and application to plants |
| EP4578859A4 (en) * | 2022-09-06 | 2026-03-25 | Nanjing Bostec Biological Eng Co Ltd | NATURAL PORPHINE SALT AND USE OF IT AS A PLANT GROWTH REGULATOR AND IMMUNE RESISTANCE INDUCTOR |
Families Citing this family (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2013181738A1 (en) | 2012-06-04 | 2013-12-12 | Suncor Energy Inc. | Formulations containing paraffinic oil and anti-settling agent |
| AR118050A1 (en) | 2019-02-15 | 2021-09-15 | Bristol Myers Squibb Co | BICYCLIC COMPOUNDS REPLACED AS MODULATORS OF THE FARNESOID X RECEIVER |
| CN119912459B (en) * | 2025-03-31 | 2025-09-12 | 上海光声制药有限公司 | Mono-heterocyclic substituted porphyrin derivatives, preparation method thereof and application thereof as photosensitizer |
| CN119912463B (en) * | 2025-03-31 | 2025-09-05 | 上海光声制药有限公司 | Disubstituted amide porphyrin derivative, preparation method thereof and use thereof as photosensitizer |
| CN119912461B (en) * | 2025-03-31 | 2025-09-05 | 上海光声制药有限公司 | Disubstituted ester porphyrin derivative, preparation method thereof and use thereof as photosensitizer |
| CN119912464B (en) * | 2025-03-31 | 2025-07-29 | 上海光声制药有限公司 | Monosubstituted ester porphyrin derivative, preparation method thereof and application of monosubstituted ester porphyrin derivative as photosensitizer |
| CN119912462B (en) * | 2025-03-31 | 2025-09-12 | 上海光声制药有限公司 | Biheterocycle-substituted porphyrin derivatives, process for their preparation and their use as photosensitizers |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2009052638A1 (en) | 2007-10-25 | 2009-04-30 | Innovotech Inc. | Natural photodynamic agents and their use |
| WO2010080819A1 (en) * | 2009-01-06 | 2010-07-15 | C3 Jian, Inc. | Targeted antimicrobial moieties |
| CN103601727A (en) | 2013-10-23 | 2014-02-26 | 中国医学科学院生物医学工程研究所 | Use of novel amine compound modified protoporphyrin |
Family Cites Families (318)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2870037A (en) | 1959-01-20 | Process for treating grass | ||
| GB191208748A (en) | 1912-04-13 | 1913-04-14 | Thomas Hurry | Composition for Destroying Red Spider. |
| GB191419295A (en) | 1914-09-01 | 1914-11-19 | Ambrose Shardlow & Company Ltd | An Improved Liquid Fuel Injector for Internal Combustion Engines. |
| GB745360A (en) | 1952-10-20 | 1956-02-22 | Boots Pure Drug Co Ltd | Acaricidal preparations |
| GB747909A (en) | 1953-08-07 | 1956-04-18 | Boots Pure Drug Co Ltd | New compositions comprising sulphur containing compounds |
| GB758926A (en) | 1953-08-07 | 1956-10-10 | Boots Pure Drug Co Ltd | New sulphur containing compounds |
| GB762866A (en) | 1953-08-07 | 1956-12-05 | Boots Pure Drug Co Ltd | New compositions comprising sulphur containing compounds |
| GB748422A (en) | 1953-08-07 | 1956-05-02 | Boots Pure Drug Co Ltd | Thiadiazole derivatives and compositions containing them |
| GB763246A (en) | 1953-08-20 | 1956-12-12 | Boots Pure Drug Co Ltd | A new diaryl disulphide and acaricidal compositions containing it |
| GB765459A (en) | 1953-08-20 | 1957-01-09 | Boots Pure Drug Co Ltd | New compositions comprising sulphur containing compounds |
| GB753976A (en) | 1953-11-24 | 1956-08-01 | Cilag Ltd | Novel amides, their production and use |
| GB792045A (en) | 1954-02-18 | 1958-03-19 | Boots Pure Drug Co Ltd | New sulphur containing compounds |
| US2714062A (en) | 1955-01-07 | 1955-07-26 | Lockrey | Composition for and process of restoring the green color of grass |
| US2786821A (en) | 1955-02-02 | 1957-03-26 | Henry A Gardner | Composition for coloring dormant grass |
| BE602261A (en) | 1960-04-06 | |||
| US3113066A (en) | 1961-01-03 | 1963-12-03 | Exxon Research Engineering Co | Bacillus thuringiensis spore pesticide |
| US3426126A (en) | 1963-10-31 | 1969-02-04 | Union Oil Co | Process for the control of nematodes using petroleum oil and water emulsions |
| GB1044895A (en) | 1964-05-04 | 1966-10-05 | George Garnham Turner | Stable copper emulsion for controlling plant diseases |
| NL136866C (en) | 1966-04-08 | |||
| US3615799A (en) | 1968-01-26 | 1971-10-26 | Ashland Oil Inc | Method of preparing sprayable bituminous composition |
| US4015970A (en) | 1968-09-06 | 1977-04-05 | Airwick Industries, Inc. | Granular products for soil treatment |
| GB1249674A (en) | 1968-11-28 | 1971-10-13 | Exxon Research Engineering Co | Novel agricultural compositions and leaf-desiccating method |
| US3948635A (en) | 1969-06-04 | 1976-04-06 | Societe Seppic | Process for selective post-emergence weeding of crops, and new products by which the process can be implemented |
| US3689574A (en) | 1970-04-28 | 1972-09-05 | Exxon Research Engineering Co | 3,4,5-trimethylcyclohexanol |
| US3997322A (en) | 1970-09-09 | 1976-12-14 | Sun Oil Company Of Pennsylvania | Herbicide carrier oil composition |
| US4124720A (en) | 1970-11-09 | 1978-11-07 | Wenmaekers Georges E J | Therapeutic hydrodispersible emulsion |
| US3799758A (en) | 1971-08-09 | 1974-03-26 | Monsanto Co | N-phosphonomethyl-glycine phytotoxicant compositions |
| US3877921A (en) | 1971-06-28 | 1975-04-15 | Scott & Sons Co O M | Herbicidal Phenoxyalkyloxazolines |
| DE2315641A1 (en) | 1973-03-29 | 1974-10-17 | Hoechst Ag | ACARICIDAL DISPERSIONS |
| US4002628A (en) | 1973-06-18 | 1977-01-11 | Eli Lilly And Company | Novel fluoroalkoxyphenyl-substituted nitrogen heterocycles |
| NL7412787A (en) | 1973-10-04 | 1975-04-08 | Hoechst Ag | FUNGICIDE DISPERSIONS OF CARBONIC ACID AMIDES. |
| DE2511077A1 (en) | 1975-03-13 | 1976-09-23 | Nippon Chemical Works Co | Turf grass coloration - using hexadentate cobalt phthalocyanine amine complex salts |
| GB1499397A (en) | 1975-04-24 | 1978-02-01 | British Petroleum Co | Pesticidal compositions |
| US4094845A (en) | 1975-09-19 | 1978-06-13 | United States Trading International, Inc. | Method of reducing loss of water by transpiration and composition therefor |
| IE45765B1 (en) | 1976-08-19 | 1982-11-17 | Ici Ltd | Triazoles and imidazoles useful as plant fungicides and growth regulating agents |
| FR2377155A1 (en) | 1977-01-14 | 1978-08-11 | Philagro Sa | FUNGICIDE COMPOSITIONS BASED ON ALCOYLPHOSPHITES |
| GB2002357A (en) | 1977-08-16 | 1979-02-21 | Bp Chem Int Ltd | Vinyl chloride monomer production |
| JPS55129213A (en) | 1979-03-22 | 1980-10-06 | Nippon Nohyaku Co Ltd | Agricultural and horticultural microbicide |
| JPS5728184A (en) | 1980-07-29 | 1982-02-15 | Toa Doro Kogyo Kk | Coloring material for sodding and post-planting care and its preparation |
| CH646193A5 (en) | 1980-09-12 | 1984-11-15 | Alusuisse | OIL-IN-WATER EMULSION FOR THE COLD ROLLING OF LIGHT METALS. |
| NO151687C (en) | 1980-11-10 | 1985-05-22 | Stauffer Chemical Co | HERBICIDE AND PLANT GROWTH REGULATORY PREPARATIONS IN THE FORM OF A Aqueous, RUNNING CONCENTRATE OF 2- (ALFA-NAPHETOXY) -N, N-DIETHYL-PROPIONAMIDE |
| SU1021416A1 (en) | 1981-12-03 | 1983-06-07 | Предприятие П/Я В-2913 | Composition for protecting planting material |
| IL68822A (en) | 1982-06-18 | 1990-07-12 | Dow Chemical Co | Pyridyl(oxy/thio)phenoxy compounds,herbicidal compositions and methods of using them |
| HU191184B (en) | 1982-07-09 | 1987-01-28 | Chinoin Gyogyszer Es Vegyeszeti Termekek Gyara Rt,Hu | Stabilized herbicide suspension |
| US5185151A (en) | 1982-12-27 | 1993-02-09 | Union Oil Company Of California | Pesticidal compositions and methods for controlling pests |
| DE3476195D1 (en) | 1983-05-18 | 1989-02-23 | Ciba Geigy Ag | Cyclohexanedione-carboxylic-acid derivatives having a herbicidal and plant growth regulating activity |
| JPS61501027A (en) | 1984-01-06 | 1986-05-22 | キノイン ギオギスゼル−エス ベギエスゼチ テルメケク ギヤラ ア−ル.テイ−. | Seed protection coating additive |
| JPS59210007A (en) | 1984-04-16 | 1984-11-28 | Hokko Chem Ind Co Ltd | Agricultural and horticultural fungicidal composition |
| US5242892A (en) | 1984-07-27 | 1993-09-07 | The Board Of Trustees Of The University Of Illinois | Chlorophyll biosynthesis modulators |
| US5300526A (en) | 1984-07-27 | 1994-04-05 | The Board Of Trustees Of The University Of Illinois | Porphyric insecticides |
| GB8507806D0 (en) | 1985-03-26 | 1985-05-01 | Robinson E | Restoring colour of grass |
| US4977177A (en) * | 1985-04-30 | 1990-12-11 | Nippon Petrochemicals Company, Ltd. | Tetrapyrrole polyaminomonocarboxylic acid therapeutic agents |
| DE3541630A1 (en) | 1985-11-26 | 1987-05-27 | Bayer Ag | FUNGICIDAL USE OF TETRAHYDROBENZOTHIENYL UREA DERIVATIVES |
| US4844756A (en) | 1985-12-06 | 1989-07-04 | The Lubrizol Corporation | Water-in-oil emulsions |
| CA1315780C (en) | 1986-01-17 | 1993-04-06 | Yozo Fukuda | Porphyrin derivatives |
| JP2559704B2 (en) | 1986-04-12 | 1996-12-04 | 有恒薬品工業 株式会社 | Oil-in-water composition of pesticide active ingredient |
| DE3614788A1 (en) | 1986-05-02 | 1987-11-05 | Hoechst Ag | HERBICIDE EMULSIONS |
| JPH0625140B2 (en) | 1986-11-10 | 1994-04-06 | 呉羽化学工業株式会社 | Novel azole derivative, method for producing the same and agricultural / horticultural drug of the derivative |
| US5102442A (en) | 1987-10-05 | 1992-04-07 | Basf Corporation | Antagonism defeating crop oil concentrates |
| US5238604A (en) | 1987-10-05 | 1993-08-24 | Basf Corporation | Crop oil concentrates |
| US4834908A (en) | 1987-10-05 | 1989-05-30 | Basf Corporation | Antagonism defeating crop oil concentrates |
| US4902333A (en) | 1987-10-30 | 1990-02-20 | The United States Of America As Represented By The Secretary Of Agriculture | Control of undesirable vegetation |
| NZ227505A (en) | 1988-01-13 | 1992-02-25 | Univ Illinois | Insecticidal composition comprising delta-ala or inducers and enhancers thereof |
| JPH01246286A (en) * | 1988-03-28 | 1989-10-02 | Hamari Yakuhin Kogyo Kk | Porphyrin derivative |
| US4896029A (en) | 1988-04-08 | 1990-01-23 | United Parcel Service Of America, Inc. | Polygonal information encoding article, process and system |
| US5206021A (en) | 1988-05-09 | 1993-04-27 | Rhone-Poulenc Ag Company | Stabilized oil-in-water emulsions or suspoemulsions containing pesticidal substances in both oil and water phases |
| US5002962A (en) | 1988-07-20 | 1991-03-26 | Health Research, Inc. | Photosensitizing agents |
| JPH02138376A (en) | 1988-11-18 | 1990-05-28 | Mitsubishi Yuka Badische Co Ltd | Hay coloring composition |
| EP0379851A1 (en) | 1988-12-27 | 1990-08-01 | Monsanto Company | Dispensable pesticide-containing compositions |
| NL8900381A (en) | 1989-02-16 | 1990-09-17 | Klook Beheer Asten B V | Root crop preservation - by covering with layer(s) of a wax material |
| EP0390743A1 (en) | 1989-03-31 | 1990-10-03 | Ciba-Geigy Ag | Pesticides with improved effect |
| US5084087A (en) | 1989-04-26 | 1992-01-28 | Basf Corporation | Ready to dilute adjuvant-containing postemergent herbicide formulations |
| AU621218B2 (en) | 1989-04-26 | 1992-03-05 | Sumitomo Chemical Company, Limited | Emulsifiable pesticidal solid compositions |
| ATA237789A (en) | 1989-07-03 | 1992-06-15 | Robert Boho | METHOD FOR TREATING WET NEEDLE OR DECUMBER LUMBER |
| US5599768A (en) | 1989-09-21 | 1997-02-04 | E. I. Du Pont De Nemours And Company | Stabilization of non-aqueous suspensions |
| US5035741A (en) | 1989-10-13 | 1991-07-30 | Safer, Inc. | Fatty acid based emulsifiable concentrate having herbicidal activity |
| JPH03183505A (en) | 1989-12-13 | 1991-08-09 | Sumika Color Kk | Treated composition of rush and treatment |
| JP2799469B2 (en) | 1990-01-27 | 1998-09-17 | 三菱化学株式会社 | Lawn colorants |
| US5741502A (en) | 1990-07-19 | 1998-04-21 | Helena Chemical Co. | Homogeneous, essentially nonaqueous adjuvant compositions with buffering capability |
| US5580567A (en) | 1990-07-19 | 1996-12-03 | Helena Chemical Company | Homogeneous, essentially nonaqueous adjuvant compositions with buffering capability |
| US5178795A (en) | 1990-07-19 | 1993-01-12 | Helena Chemical Company | Homogeneous, essentially nonaqueous adjuvant compositions with buffering capability |
| US5393791A (en) | 1990-07-19 | 1995-02-28 | Helena Chemical Company | Homogeneous, essentially nonaqueous adjuvant compositions with buffering capability |
| DE4029304A1 (en) | 1990-09-15 | 1992-03-19 | Hoechst Ag | Synergistic herbicide mixts. - contain alkyl-polyglycol-ether! sulphate surfactant and a leaf-effective herbicide |
| JPH04128003A (en) | 1990-09-19 | 1992-04-28 | Sumika Color Kk | Rush treated composition and rush treatment method |
| US5308827A (en) | 1990-11-28 | 1994-05-03 | Fumakilla Limited | Herbicidal foam composition |
| GB9102757D0 (en) | 1991-02-08 | 1991-03-27 | Albright & Wilson | Biocidal and agrochemical suspensions |
| FR2678807A1 (en) | 1991-07-09 | 1993-01-15 | Rhone Poulenc Chimie | HOMOGENEOUS, STABLE AND VERSABLE AQUEOUS MIXTURES AND USE OF SAID MIXTURES FOR PREPARING DISPERSIONS OF WATER-INSOLUBLE SUBSTANCES. |
| GB9116557D0 (en) | 1991-07-31 | 1991-09-11 | Shell Int Research | Fungicidal compositions |
| US5229356A (en) | 1991-08-23 | 1993-07-20 | E. I. Du Pont De Nemours And Company | Slow release compositions comprising heterocyclic sulfonylurea herbicides, paraffin wax, hydrocarbon polymers, and particulate fillers |
| DE4137476A1 (en) | 1991-11-14 | 1993-05-19 | Basf Ag | USE OF PIGMENT PREPARATIONS FOR THE PRODUCTION OF PASTE, PRINTING INKS AND VARNISHES |
| IL103614A (en) | 1991-11-22 | 1998-09-24 | Basf Ag | Carboxamides for controlling botrytis and certain novel such compounds |
| MY109293A (en) | 1991-12-18 | 1996-12-31 | Gillette Co | Fiber marker including an erasable ink |
| US5976535A (en) | 1992-06-09 | 1999-11-02 | Neorx Corporation | Pretargeting protocols for the enhanced localization of cytotoxins to target sites and cytotoxic combinations useful therefore |
| US6217869B1 (en) | 1992-06-09 | 2001-04-17 | Neorx Corporation | Pretargeting methods and compounds |
| US5558806A (en) | 1992-07-23 | 1996-09-24 | Osi Specialties, Inc. | Surfactant blend of a polyalkleneoxide polysiloxane and an organic compound having a short chain hydrophobic moiety |
| DK0598515T3 (en) | 1992-11-18 | 1999-06-21 | Ishihara Sangyo Kaisha | Process for Reinforcing a Herbicidal Activity, Herbicidal Composition with Enhanced Activity and Activity Forest |
| US5336661A (en) | 1993-01-13 | 1994-08-09 | North Carolina State University | Method for enhancing turf quality of bent grass |
| US5643852A (en) | 1993-01-13 | 1997-07-01 | North Carolina State University | Fungicidal compositions for the enhancement of turf quality |
| US5665672A (en) | 1993-01-13 | 1997-09-09 | North Carolina State University | Fungicidal compositions for the treatment of crown and root rot in turfgrass |
| WO1994022311A1 (en) | 1993-03-30 | 1994-10-13 | Osi Specialties, Inc. | Super-spreading, low-foam surfactant for agricultural spray mixtures |
| US6713518B1 (en) | 1993-05-21 | 2004-03-30 | Ecosmart Technologies, Inc. | Non-hazardous pest control |
| US5658851A (en) | 1993-10-13 | 1997-08-19 | Osi Specialties, Inc. | Lipophilic siloxanes as adjuvants for agriculture |
| US6015897A (en) | 1993-12-07 | 2000-01-18 | Neorx Corporation | Biotinamido-n-methylglycyl-seryl-o-succinamido-benzyl dota |
| EP1346730A1 (en) | 1993-12-07 | 2003-09-24 | Neorx Corporation | Pretargeting methods and novel pretargeting conjugates |
| BR9406470A (en) | 1993-12-28 | 1996-01-23 | Kao Corp | Composition and process for improving chemicals applicable in agriculture and chemical composition applicable in agriculture |
| US5668086A (en) | 1994-05-20 | 1997-09-16 | Kao Corporation | Herbicide composition |
| AU691730B2 (en) | 1995-01-13 | 1998-05-21 | Environmentally Safe Systems, Inc. | Environmentally safe pesticide and plant growth accelerator |
| CA2209920C (en) | 1995-01-13 | 2007-01-02 | Environmentally Safe Systems, Inc. | Environmentally safe pesticide and plant growth accelerator |
| US5614203A (en) | 1995-01-30 | 1997-03-25 | Environmentally Safe Systems, Inc. | Environmentally safe pesticide and plant growth accelerator |
| JPH08218225A (en) | 1995-02-14 | 1996-08-27 | Teijin Ltd | Polyamide fiber for human turf and method for producing the same |
| US5599804A (en) | 1995-04-03 | 1997-02-04 | Rhone-Poulenc, Inc. | Fungicidal compositions for the enhancement of turf quality |
| IT1275571B (en) | 1995-07-19 | 1997-08-07 | Consiglio Nazionale Ricerche | FLUOROGENIC SUBSTRATES SUSCEPTIBLE FOR PHOTOACTIVATION AFTER ENZYMATIC TRANSFORMATION SUITABLE FOR DIAGNOSIS AND PHOTODYNAMIC CANCER THERAPY |
| US5989331A (en) | 1996-05-28 | 1999-11-23 | Southern Clay Products, Inc. | TiO2 slurry stabilization |
| JPH1029901A (en) | 1996-07-17 | 1998-02-03 | Mitsui Petrochem Ind Ltd | Soil fumigant and soil fumigating method |
| US5913884A (en) | 1996-09-19 | 1999-06-22 | The General Hospital Corporation | Inhibition of fibrosis by photodynamic therapy |
| US5703016A (en) | 1996-09-30 | 1997-12-30 | Albemarle Corporation | Surfactant composition for use with glyphosate comprising dimethyl amine oxide, polyethoxylated alcohol, and pyridinium halide |
| FR2754451B1 (en) | 1996-10-14 | 1998-11-06 | Oreal | SELF-FOAMING CREAM |
| US7353829B1 (en) | 1996-10-30 | 2008-04-08 | Provectus Devicetech, Inc. | Methods and apparatus for multi-photon photo-activation of therapeutic agents |
| CA2277821A1 (en) | 1997-01-24 | 1998-07-30 | The Procter & Gamble Company | Photochemical singlet oxygen generators having enhanced singlet oxygen yields |
| ES2171009T3 (en) | 1997-02-14 | 2002-08-16 | Monsanto Technology Llc | WATERPROOF COMPOSITIONS OF HERBICIDES AND TENSIOACTIVES OF APPLICATION TO THE BASE OR SLEEPING PARTS OF THE MALEZA SIZE. |
| US6462070B1 (en) | 1997-03-06 | 2002-10-08 | The General Hospital Corporation | Photosensitizer conjugates for pathogen targeting |
| US6221811B1 (en) | 1997-03-06 | 2001-04-24 | Crompton Corporation | Siloxane nonionic blends useful in agriculture |
| AU749607B2 (en) | 1997-04-14 | 2002-06-27 | Dow Agrosciences Llc | Pesticide compositions |
| GB9710049D0 (en) | 1997-05-19 | 1997-07-09 | Nycomed Imaging As | Method |
| US5739371A (en) | 1997-07-07 | 1998-04-14 | Lambent Technologies Inc. | Carboxy silicone amphoteric surfactant complexes |
| GB9718139D0 (en) | 1997-08-27 | 1997-10-29 | Zeneca Ltd | Agrochemical Formulation |
| US20010019728A1 (en) | 1997-08-28 | 2001-09-06 | Basinger William H. | Methods and compositions for protecting plants and crops |
| US6486214B1 (en) | 1997-09-10 | 2002-11-26 | Rutgers, The State University Of New Jersey | Polyanhydride linkers for production of drug polymers and drug polymer compositions produced thereby |
| US7122615B1 (en) | 1998-09-10 | 2006-10-17 | Rutgers, The State University Of New Jersey | Polyanhydrides with therapeutically useful degradation products |
| US5958104A (en) | 1997-09-11 | 1999-09-28 | Nonomura; Arthur M. | Methods and compositions for enhancing plant growth |
| JP3277146B2 (en) | 1997-11-12 | 2002-04-22 | 大日精化工業株式会社 | Vegetable grass coloring agent and vegetation grass coloring method |
| US6123923A (en) | 1997-12-18 | 2000-09-26 | Imarx Pharmaceutical Corp. | Optoacoustic contrast agents and methods for their use |
| JP3227424B2 (en) | 1998-02-13 | 2001-11-12 | 大日精化工業株式会社 | Lawn colorant composition with growing effect |
| JPH11349588A (en) | 1998-04-10 | 1999-12-21 | Mitsubishi Chemical Corp | Method for producing phthalocyanine derivative |
| JP2000016909A (en) | 1998-04-28 | 2000-01-18 | Japan Tobacco Inc | Herbicidal composition for poa annua |
| US6379687B2 (en) | 1998-05-28 | 2002-04-30 | Bayer Corporation | Inhibiting phase separation in low viscosity water-based pesticide suspensions |
| US20030194454A1 (en) | 1998-07-28 | 2003-10-16 | Bessette Steven M. | Pesticidal compositions containing rosemary oil and wintergreen oil |
| US6346535B1 (en) | 1999-01-29 | 2002-02-12 | American Cyanamid Company | Fungicidal mixtures |
| UA72760C2 (en) | 1999-04-23 | 2005-04-15 | Monsanto Technology Llc | Method to treat plant foliage |
| FR2799965B1 (en) | 1999-10-22 | 2002-10-31 | Oreal | EMULSION-FREE EMULSIONS CONTAINING AT LEAST ONE INSOLUBLE ORGANIC UV FILTER |
| FR2799963B1 (en) | 1999-10-22 | 2002-07-19 | Oreal | EMULSIONS CONTAINING AT LEAST ONE INSOLUBLE ORGANIC UV FILTER AND A NON-FILTERING ORGANOMODIFIED SILICONE |
| US6432877B2 (en) | 2000-01-21 | 2002-08-13 | Dainichiseika Color & Chemicals Mfg. Co., Ltd. | Lawn colorant composition having rearing effects |
| PL358278A1 (en) | 2000-03-13 | 2004-08-09 | Basf Aktiengesellschaft | Agrotechnical formulation |
| US6444614B2 (en) | 2000-04-12 | 2002-09-03 | Lidochem Inc. | Aspartic acid derivative-containing compositions and use thereof in stimulating and/or regulating plant and plant precursor growth |
| US7135437B2 (en) | 2000-05-19 | 2006-11-14 | Monsanto Technology Llc | Stable liquid pesticide compositions |
| US6858634B2 (en) | 2000-09-15 | 2005-02-22 | Monsanto Technology Llc | Controlled release formulations and methods for their production and use |
| US6573258B2 (en) | 2000-09-27 | 2003-06-03 | Frontier Scientific, Inc. | Photodynamic porphyrin antimicrobial agents |
| AU1312502A (en) | 2000-10-11 | 2002-04-22 | Pe Corp Ny | Fluorescent nucleobase conjugates having anionic linkers |
| US20030198696A1 (en) | 2000-10-19 | 2003-10-23 | David Keen | Pesticide composition of plant oils and fish oils |
| CA2324677A1 (en) | 2000-10-26 | 2002-04-26 | James F. Stewart | Agrochemical formulation aid |
| US20030185754A1 (en) | 2001-01-16 | 2003-10-02 | Genset, S.A. | Treatment of CNS disorders using D-amino acid oxidase and D-aspartate oxidase antagonists |
| US20020137901A1 (en) | 2001-01-22 | 2002-09-26 | Cavanaugh Philip Gerard | Synthesis, and photodynamic therapy-mediated anti-cancer, and other uses of chlorin e6-transferrin |
| US6515031B2 (en) | 2001-02-13 | 2003-02-04 | Platte Chemical Company | Technique for emulsifying highly saturated hydroisomerized fluids |
| WO2002089573A1 (en) | 2001-05-04 | 2002-11-14 | Bayer Cropscience Gmbh | Pesticide oil-in-water-in-oil emulsion |
| AUPR510001A0 (en) | 2001-05-18 | 2001-06-14 | Jupitar Pty Ltd | Formulation and method |
| WO2002098225A2 (en) | 2001-06-07 | 2002-12-12 | Basf Corporation | Plant growth regulators and methods for control of annual bluegrass |
| EP1455568A2 (en) | 2001-06-15 | 2004-09-15 | The University Of Toledo | Method for transformation of mono- and di- cotyledonous plants using meristematic tissue and nodal callus from dycotiledonous plants |
| CA2462122C (en) | 2001-09-26 | 2010-02-09 | Platte Chemical Company | Herbicide microemulsion-forming-concentrates, microemulsions, and methods |
| EP1450774A4 (en) | 2001-10-03 | 2005-10-26 | Miravant Pharm Inc | CHLORINE PHOTOSENSITIZING AGENTS FOR USE IN PHOTODYNAMIC THERAPY |
| EP1450790A4 (en) | 2001-10-03 | 2005-10-26 | Miravant Pharm Inc | PHOTOSENSITIZER CARBAMATE DERIVATIVES |
| JP2003128501A (en) | 2001-10-18 | 2003-05-08 | Nissan Chem Ind Ltd | Agrochemical emulsion composition |
| US20030198659A1 (en) | 2001-10-25 | 2003-10-23 | Hoffmann Michael P. | Fibrous pest control |
| AU2002339696A1 (en) | 2001-12-03 | 2003-06-17 | Genset S.A. | Treatment of cns disorders using d-amino acid oxidase and d-aspartate oxidase inhibitors |
| US6972273B2 (en) | 2002-03-01 | 2005-12-06 | W. Neudorff Gmbh Kg | Composition and method for selective herbicide |
| US8076267B2 (en) | 2002-03-01 | 2011-12-13 | W. Neudorff Gmbh Kg | Enhancers for water soluble selective auxin type herbicides |
| IL149978A0 (en) | 2002-06-02 | 2002-12-01 | Biotan Biocides For Paints And | A biocide and method for making the same |
| UA78326C2 (en) | 2002-06-12 | 2007-03-15 | Сінгента Партісіпейшнс Аг | Herbicidal synergetic composition and method to control growth of undesirable vegetation |
| GB0215534D0 (en) | 2002-07-04 | 2002-08-14 | Ecole Polytech | Selective photochemotherapy using oligonucleotide targeting agents |
| DE10245727A1 (en) | 2002-10-01 | 2004-04-15 | Beiersdorf Ag | Sprayable W / O emulsions as aerosol |
| US20060276339A1 (en) | 2002-10-16 | 2006-12-07 | Windsor J B | Methods and compositions for increasing the efficacy of biologically-active ingredients |
| US7700076B2 (en) | 2002-10-25 | 2010-04-20 | Foamix, Ltd. | Penetrating pharmaceutical foam |
| HRP20050585A2 (en) | 2002-11-25 | 2006-02-28 | Attenuon | Peptides which inhibit angiogenesis, cell migration, cell invasion and cell proliferation, compositions and uses thereof |
| US20040192556A1 (en) | 2002-12-17 | 2004-09-30 | Schregenberger Sandra Denise | Crop oil adjuvants |
| DE10258867A1 (en) | 2002-12-17 | 2004-07-08 | Bayer Cropscience Gmbh | Microemulsion concentrates |
| US7135435B2 (en) | 2002-12-27 | 2006-11-14 | Sepro Corporation | Plant growth regulation compositions and methods using gibberellin biosynthesis inhibitor combinations |
| EP1592299A1 (en) | 2003-01-10 | 2005-11-09 | Battelle Memorial Institute | Sprayable non-aqueous, oil-continuous microemulsions and methods of making same |
| US8138120B2 (en) | 2003-03-11 | 2012-03-20 | Cognis Ip Management Gmbh | Microemulsions as adjuvants for agricultural chemicals |
| EP1457113A1 (en) | 2003-03-12 | 2004-09-15 | Salbert Co Ltd | Alkylated porphyrins as pesticides acting both in the presence and absence of light |
| AU2003902064A0 (en) | 2003-04-30 | 2003-05-15 | Australian Agricultural Chemicals | Colouring agent for turf |
| US20080112909A1 (en) | 2003-06-24 | 2008-05-15 | Ppg Industries Ohio, Inc. | Compositions for providing color to animate objects and related methods |
| US8575067B2 (en) | 2003-07-25 | 2013-11-05 | Ishihara Sangyo Kaisha, Ltd. | Herbicidal composition having the herbicidal effect enhanced, and method for enhancing the herbicidal effect |
| DE10334300A1 (en) | 2003-07-28 | 2005-03-03 | Bayer Cropscience Gmbh | Oil suspension concentrate |
| US20070148200A1 (en) | 2003-08-08 | 2007-06-28 | Quest Products Corporation | Agricultural compositions for through bark application to woody plants |
| EP1659864B1 (en) | 2003-08-26 | 2013-09-18 | Basf Se | Method of plant growth promotion using amide compounds |
| WO2005021723A2 (en) | 2003-08-27 | 2005-03-10 | Syngenta Participations Ag | Nucleic acid molecules from rice controlling abiotic stress tolerance |
| US8133482B2 (en) | 2003-11-14 | 2012-03-13 | The Trustees Of The University Of Pennsylvania | Activatable photodynamic therapy agents |
| WO2005048944A2 (en) | 2003-11-14 | 2005-06-02 | University Of Pennsylvania | Activatable photodynamic therapy agents |
| ES2451623T3 (en) | 2003-12-04 | 2014-03-28 | Syngenta Participations Ag | Herbicidal composition |
| CN102010382A (en) | 2003-12-29 | 2011-04-13 | 塞普拉科公司 | Benzo[d]isoxazol-3-ol DAAO Inhibitors |
| US20050181949A1 (en) | 2004-02-13 | 2005-08-18 | Norton Lawrence H. | Method of improving turfgrass quality |
| US8153558B2 (en) | 2004-02-13 | 2012-04-10 | Bayer Cropscience Lp | Method of improving grass quality |
| EP1570733A1 (en) | 2004-02-26 | 2005-09-07 | Wolfgang Willi Deppert | Mammal for studying DCIS and the effect of inactivated tumor suppressors |
| US20050244357A1 (en) | 2004-02-26 | 2005-11-03 | Eward Sieverding | Oil-containing emulsifiable formulations containing active agents |
| DE102004014704A1 (en) | 2004-03-25 | 2005-10-13 | Wacker-Chemie Gmbh | Particle-stabilized emulsions |
| JP2006124337A (en) | 2004-10-29 | 2006-05-18 | Idemitsu Kosan Co Ltd | Plant disease control agent |
| CA2472806A1 (en) | 2004-05-18 | 2005-11-18 | Petro-Canada | Compositions and methods for treating turf insect pests and diseases such as fungal infestations |
| CA2507482C (en) | 2004-05-18 | 2014-02-25 | Petro-Canada | Spray oil and method of use thereof for controlling turfgrass pests |
| WO2005123105A1 (en) | 2004-06-10 | 2005-12-29 | The Trustees Of Boston College | Phototoxic compounds |
| US20050274164A1 (en) | 2004-06-14 | 2005-12-15 | Brian Coates | Combination grass colorant and fertilizer |
| TW200603738A (en) | 2004-06-17 | 2006-02-01 | Basf Ag | Use of (e)-5-(4-chlorobenzylidene)-2,2-dimethyl-1-(1h-1,2,4-triazol-1-ylmethyl)cyclopentanol for controlling rust disease on soybean plants |
| WO2006023899A1 (en) | 2004-08-24 | 2006-03-02 | Bayer Cropscience Lp | Method of fungal pathogen control in grass or turf |
| US20060063676A1 (en) | 2004-09-17 | 2006-03-23 | Brigance Mickey R | Method for enhancing deposition of an agrochemical |
| WO2006086662A2 (en) | 2005-02-11 | 2006-08-17 | Dow Agrosciences Llc | Safer control of broadleaf weeds in turf with granular formulations of als inhibiting herbicides |
| US9326509B2 (en) | 2005-02-22 | 2016-05-03 | Basf Se | Composition and method for improving plant health |
| NL1028485C2 (en) | 2005-03-08 | 2006-09-11 | Photobiochem N V | Method for preparing a porphyrin derivative, a porphyrin derivative, use of said porphyrin derivative, and a pharmaceutical preparation containing said porphyrin derivative. |
| CA2600079A1 (en) | 2005-03-21 | 2006-09-28 | Basf Aktiengesellschaft | Insecticidal mixtures |
| WO2006132712A2 (en) | 2005-04-21 | 2006-12-14 | Goldstein Glenn A | N-acetylcysteine amide (nac amide) for enhancing plant resistance and tolerance to environmental stress |
| AU2005332196A1 (en) | 2005-05-27 | 2006-11-30 | Indian Oil Corporation Limited | Composition of an agricultural spray oil |
| US7576113B2 (en) | 2005-06-16 | 2009-08-18 | Basf Aktiengesellschaft | Use of (E)-5-(4-chlorbenzyliden)-2,2-dimethyl-1-(1H-1,2,4-triazol-1-ylmethyl) cyclopentanol for combating rust attacks on soya plants |
| UA90035C2 (en) | 2005-11-10 | 2010-03-25 | Басф Се | Fungicidal mixture comprising boscalid and pyrimethanil and method for control of phytopathogenic fungi |
| CN101374856A (en) | 2005-11-29 | 2009-02-25 | 斯克里普斯研究学院 | Inhibit tumor cell invasion, metastasis and angiogenesis |
| CA2633987A1 (en) | 2005-12-22 | 2008-02-21 | Syngenta Participations Ag | Methods and composition for growth engineering and disease control |
| GB0526474D0 (en) | 2005-12-24 | 2006-02-08 | Destiny Pharma Ltd | Novel process |
| US7935842B2 (en) | 2006-02-09 | 2011-05-03 | Momentive Performance Materials Inc. | Hydrolysis resistant organomodified trisiloxane surfactants |
| US8652493B2 (en) | 2006-04-05 | 2014-02-18 | Elc Management, Llc | Hydrophobic agents stably dispersed in oil-in-water emulsions |
| CN101415327B (en) | 2006-04-07 | 2012-10-10 | 先正达参股股份有限公司 | Method of controlling phytopathogenic diseases on turfgrass |
| WO2007133683A2 (en) | 2006-05-10 | 2007-11-22 | E. I. Du Pont De Nemours And Company | Formulated tick and insect repellent compositions |
| US8734821B2 (en) | 2006-05-15 | 2014-05-27 | Oms Investments, Inc. | Silicone surfactant-based agricultural formulations and methods for the use thereof |
| PT2046123E (en) | 2006-07-24 | 2014-11-18 | Syngenta Participations Ag | Controlled release granules |
| JP2010502725A (en) | 2006-09-06 | 2010-01-28 | シンジェンタ パーティシペーションズ アクチェンゲゼルシャフト | Pickering emulsion formulation |
| CA2605092C (en) | 2006-10-05 | 2014-03-25 | Petro-Canada | Herbicidal composition with increased herbicidal efficacy |
| US9357768B2 (en) | 2006-10-05 | 2016-06-07 | Suncor Energy Inc. | Herbicidal composition with increased herbicidal efficacy |
| CA2562718A1 (en) | 2006-10-05 | 2008-04-05 | Petro-Canada | Herbicidal composition with increased herbicidal efficacy |
| CN100428884C (en) * | 2006-10-24 | 2008-10-29 | 南京农业大学 | Chlorohemin contaning plant growth regulator |
| TWI484910B (en) | 2006-12-01 | 2015-05-21 | Du Pont | Liquid formulations of carboxamide arthropodicides |
| US7879916B2 (en) | 2006-12-11 | 2011-02-01 | Momentive Performance Materials Inc. | Hydrolysis resistant organomodified silylated ionic surfactants |
| CN100556289C (en) | 2007-02-05 | 2009-11-04 | 广东省昆虫研究所 | Mineral oil is as the application of fruit tree bud inhibitor |
| CA2625415C (en) | 2007-03-14 | 2015-05-05 | W. Neudorff Gmbh Kg | Enhancers for water soluble selective auxin type herbicides |
| US8748345B2 (en) | 2007-05-10 | 2014-06-10 | Tessenderlo Kerley Inc. | Method and composition for improving turfgrass |
| EP2064330A2 (en) | 2007-05-22 | 2009-06-03 | BASF Plant Science GmbH | Plants with increased tolerance and/or resistance to environmental stress and increased biomass production |
| US20090093470A1 (en) | 2007-10-08 | 2009-04-09 | Ondine International, Ltd. | Photodynamic therapy process and photosensitizer compositions therefor |
| DE102007061455A1 (en) | 2007-12-20 | 2009-06-25 | Evonik Degussa Gmbh | defoamer |
| EP2234489B1 (en) | 2008-01-15 | 2017-04-12 | Bayer Intellectual Property GmbH | Pesticide composition comprising a tetrazolyloxime derivative and a fungicide or an insecticide active substance |
| ES2658852T3 (en) | 2008-01-18 | 2018-03-12 | Visen Medical, Inc. | Fluorescent Imaging Agents |
| US7822554B2 (en) | 2008-01-24 | 2010-10-26 | Schlumberger Technology Corporation | Methods and apparatus for analysis of downhole compositional gradients and applications thereof |
| CA2711660C (en) | 2008-01-25 | 2016-10-11 | Syngenta Limited | Herbicidal compositions |
| EP4410100A3 (en) | 2008-02-05 | 2024-10-09 | Basf Se | Plant health composition |
| US20100009970A1 (en) | 2008-03-19 | 2010-01-14 | Combinatorx (Singapore) Pte. Ltd. | Compositions and methods for treatment of viral diseases |
| WO2009126370A1 (en) | 2008-04-08 | 2009-10-15 | Bayer Cropscience Lp | Composition and system for turf maintenance |
| WO2009137062A2 (en) | 2008-05-05 | 2009-11-12 | The General Hospital Corporation | Photoactivatable antimicrobial agents and therapeutic and diagnostic methods of using same |
| AU2009247547B2 (en) | 2008-05-13 | 2013-10-10 | Cosmo Trade & Service Co., Ltd. | Agent for improving lawn grass qualities |
| JP5596678B2 (en) | 2008-06-26 | 2014-09-24 | サンコー エナジー インコーポレーテッド | Improved turfgrass disinfectant formulation containing pigments |
| ES2620442T3 (en) * | 2008-07-29 | 2017-06-28 | Frontier Scientific, Inc. | Use of tetrakis (N-alkylpyridinium) -porphyrin derivatives for the destruction of microbes or the prevention of their growth |
| EP2349981A4 (en) | 2008-10-11 | 2016-11-30 | Univ Rutgers | BIOCOMPATIBLE POLYMERS FOR MEDICAL DEVICES |
| BRPI0920125B1 (en) | 2008-10-17 | 2017-03-28 | Evonik Degussa Gmbh | agrochemical oily composition comprising high character silicone alkyl polysiloxane auxiliaries, and method for controlling unwanted weeds or treating plants against pests |
| JP5584133B2 (en) | 2008-10-20 | 2014-09-03 | 株式会社理研グリーン | Growth regulator for grass weeds |
| HRP20140721T1 (en) * | 2008-10-24 | 2014-09-26 | Destiny Pharma Limited | Porphyrin compounds for killing, inhibiting or preventing the growth of microbial biofilms |
| CN101390517B (en) | 2008-11-07 | 2011-06-15 | 四川大学 | Kiwi growth cold resistant agent |
| BRPI0922509B1 (en) | 2008-12-23 | 2018-03-06 | Nufarm Australia Limited | WATER LIQUID HERBICIDE COMPOSITION AND WATER SOLUBLE SOLID COMPOSITION, THEIR PREPARATION METHODS AS WELL AS A PLANT DEVELOPMENT CONTROL METHOD |
| AU2010204310B2 (en) | 2009-01-07 | 2014-07-24 | Basf Se | Pesticidal mixtures |
| CN101473849B (en) | 2009-01-17 | 2012-01-04 | 邹一平 | Application of paraffin oil composition in preventing and treating diseases and pests of plant |
| GB0904825D0 (en) | 2009-03-20 | 2009-05-06 | Photobiotics Ltd | Biological materials and uses thereof |
| WO2010116259A2 (en) | 2009-04-07 | 2010-10-14 | Taminco, Naamloze Vennootschap | Plant growth regulator additive |
| AR076485A1 (en) | 2009-04-28 | 2011-06-15 | Basf Corp | FOAM PESTICIDES COMPOSITIONS |
| US8377850B2 (en) | 2009-05-15 | 2013-02-19 | Bayer Cropscience Lp | Fungicidal compositions for turf treatment and improvement |
| EP2429287A4 (en) | 2009-05-15 | 2014-12-24 | Bayer Cropscience Lp | Fungicidal active compound compositions for turf treatment |
| CN101904802B (en) | 2009-06-03 | 2015-03-11 | 赢创德固赛有限公司 | Water-in-oil emulsion system and preparation method thereof |
| PL2445341T3 (en) | 2009-06-25 | 2018-01-31 | Basf Se | Use of agrochemical mixtures for increasing the health of a plant |
| WO2011028987A2 (en) | 2009-09-03 | 2011-03-10 | University Of Tennessee Research Foundation | Methods of improving nutritional value of plants |
| FR2953369B1 (en) | 2009-12-07 | 2016-02-05 | Total Raffinage Marketing | EMULSIFIABLE CONCENTRATE FOR PHYTOSANITARY COMPOSITION, EMULSIFIABLE CONCENTRATE AND PHYTOSANITARY FILM |
| PL2509417T3 (en) | 2009-12-08 | 2017-09-29 | Basf Se | Pesticidal mixtures |
| WO2011075805A1 (en) | 2009-12-23 | 2011-06-30 | Fundação Educacional Da Região De Joinville-Furj | Non-toxic and biodegradable insecticide formulation |
| CN101773113B (en) | 2010-02-09 | 2013-02-13 | 安徽农业大学 | Hot fogging concentrate containing propiconazole and chlorpyrifos and preparation method thereof |
| CN103118536B (en) | 2010-04-26 | 2015-07-01 | 陶氏益农公司 | Stabilized agricultural oil dispersions |
| US8987171B2 (en) | 2010-06-11 | 2015-03-24 | W. Neudorff Gmbh Kg | Selective herbicides with activator adjuvants |
| PE20131200A1 (en) | 2010-07-15 | 2013-10-13 | Dow Agrosciences Llc | SOLID HERBICIDE COMPOSITIONS WITH AN INTEGRATED ADJUVANT |
| CN103338642A (en) | 2010-09-09 | 2013-10-02 | 桑科能源股份有限公司 | Synergistic paraffinic oil and boscalid fungicides |
| BRPI1003373A2 (en) | 2010-09-29 | 2013-01-29 | Fmc Quimica Do Brasil Ltda | synergistic combinations of triazoles, strobirulins and benzimidazoles, uses, formulations, production processes and applications using the same |
| IT1402153B1 (en) | 2010-10-08 | 2013-08-28 | Uni Degli Studi Camerino | COMPOSITION INCLUDING A LARVICID PHOTOACTIVABLE |
| US20130247630A1 (en) | 2010-10-28 | 2013-09-26 | Akzo Nobel Chemicals International B.V. | Complex of divalent metal cations and chelating agent |
| CN103459551B (en) | 2011-01-27 | 2016-06-15 | 日东电工株式会社 | The phototherapy device of the terphenyl that comprises any replacement and quaterphenyl compound and method |
| GB201103578D0 (en) | 2011-03-02 | 2011-04-13 | Sabrepharm Ltd | Dipyridinium derivatives |
| US20140106967A1 (en) | 2011-03-21 | 2014-04-17 | The Governors Of The University Of Alberta | Auxin plant growth regulators |
| EP2704566A1 (en) | 2011-05-02 | 2014-03-12 | Basf Se | A method for enhancing the performance of a pesticide with guanidines |
| US9451773B2 (en) | 2011-06-03 | 2016-09-27 | Suncor Energy Inc. | Paraffinic oil-in-water emulsions for controlling infection of crop plants by fungal pathogens |
| EP2713748A4 (en) | 2011-06-03 | 2014-11-19 | Suncor Energy Inc | Paraffinic oil and class b gibberellin biosynthesis inhibitor compositions |
| US20140256556A1 (en) | 2011-06-13 | 2014-09-11 | Suncor Energy Inc. | Delivery of paraffinic oil-containing compositions to root tissue of plants |
| CN102273467B (en) | 2011-06-28 | 2013-10-09 | 南京师范大学 | Application of Metal Derivatives Containing Chlorophyll and Its Hydrolyzed Products as Plant Growth Regulators |
| CN102285992B (en) | 2011-06-28 | 2013-12-18 | 南京师范大学 | Dihydroporphin (III) chelates having plant growth regulating activity and use thereof as plant growth regulator |
| RU2475498C1 (en) | 2011-11-17 | 2013-02-20 | Общество С Ограниченной Ответственностью "Фарминтерпрайсез" | Novel hemin derivatives having antibacterial and antiviral activity |
| CA2839775A1 (en) | 2011-12-02 | 2013-06-06 | Bright Ray Solar Corp. | Plant treatment and method |
| KR101518298B1 (en) | 2011-12-12 | 2015-05-08 | 가톨릭대학교 산학협력단 | Gene Nanocomplex and Method for internalizing Gene using the same |
| US9204638B2 (en) | 2011-12-30 | 2015-12-08 | Plant Health Care, Inc. | Method for increasing plant yield, and yield improving compositions |
| EP2820397A4 (en) | 2012-02-27 | 2015-09-09 | Sergei Vinogradov | Improved phosphorescent molecules for measuring oxygen and imaging methods |
| US8748446B2 (en) | 2012-03-03 | 2014-06-10 | Nanoquantum Sciences, Inc. | Halogenated compounds for photodynamic therapy |
| CN102701836B (en) | 2012-05-11 | 2014-04-16 | 浙江农林大学 | Slowly-releasing health-care liquid special for directly-spraying landscape plants and preparation method and use thereof |
| WO2013181738A1 (en) | 2012-06-04 | 2013-12-12 | Suncor Energy Inc. | Formulations containing paraffinic oil and anti-settling agent |
| US20160073634A1 (en) | 2012-06-22 | 2016-03-17 | The General Hospital Corporation | Beta-lactamase targeted photosensitizer for pesticide and pest detection |
| EP2922403A1 (en) | 2012-11-22 | 2015-09-30 | BASF Corporation | Pesticidal mixtures |
| KR102381965B1 (en) | 2013-01-11 | 2022-04-04 | 임파서블 푸즈 인크. | Methods and compositions for consumables |
| CA2899925A1 (en) | 2013-02-04 | 2014-08-07 | Seres Therapeutics, Inc. | Compositions and methods for inhibition of pathogenic bacterial growth |
| EP2967056B1 (en) | 2013-03-15 | 2018-02-21 | Suncor Energy Inc. | Herbicidal compositions |
| EP3011972B1 (en) | 2013-06-19 | 2019-10-23 | SBI Pharmaceuticals Co., Ltd. | Medicinal composition for promoting synthesis of protoporphyrin ix |
| CA2836757C (en) | 2013-12-06 | 2019-09-10 | Suncor Energy Inc. | Methods for increasing resistance of plants to abiotic stresses |
| WO2015106770A1 (en) | 2014-01-16 | 2015-07-23 | Tarek Abd Allah El-Tayeb | Suncide agri-pest: a green pesticide formula against agricultural pests |
| EP3142684A4 (en) | 2014-05-12 | 2018-05-23 | Purdue Research Foundation | Selectin and icam/vcam peptide ligand conjugates |
| CN106794264B (en) | 2014-06-10 | 2021-03-23 | 3B制药有限公司 | Conjugates comprising neurotensin receptor ligands and uses thereof |
| EP2954933A1 (en) | 2014-06-10 | 2015-12-16 | 3B Pharmaceuticals GmbH | Conjugate comprising a neurotensin receptor ligand |
| EP2954934A1 (en) | 2014-06-11 | 2015-12-16 | 3B Pharmaceuticals GmbH | Conjugate comprising a neurotensin receptor ligand and use thereof |
| CN107108863B (en) | 2014-12-23 | 2021-03-19 | 新泽西鲁特格斯州立大学 | Biocompatible iodinated diphenol monomers and polymers |
| CN104513250B (en) | 2014-12-23 | 2016-11-23 | 南开大学 | The synthetic method of a kind of water-soluble porphyrin and photocatalytic applications thereof |
| US20160205925A1 (en) | 2015-01-15 | 2016-07-21 | Ariel-University Research And Development Company Ltd. | Antimicrobial compositions made of a thermoplastic polymer and a photosensitizer |
| CN105111219B (en) | 2015-07-29 | 2018-10-16 | 大连理工大学 | Hydrophily long wavelength's dihydro porphin light, sound sensitiser and the preparation method and application thereof |
| DK3331997T5 (en) | 2015-08-03 | 2024-10-07 | Monsanto Technology Llc | METHODS AND COMPOSITIONS FOR HERBICIDE TOLERANCE IN PLANTS |
| JP2018528207A (en) | 2015-08-31 | 2018-09-27 | ザ ユニバーシティー オブ アデレード | Methods and products for preventing and / or treating microbial infections comprising iron chelators and non-ferrous porphyrins |
| CN105601638B (en) | 2016-01-28 | 2018-10-26 | 东北林业大学 | A kind of chlorophyllin derivative and preparation method thereof, as the application of photosensitive bacteriostatic agent and light sensitive pesticides |
| CN105748439A (en) | 2016-01-29 | 2016-07-13 | 四川大学 | Ph-responsive nanometer drug delivery system based on dendrimers modified by short-chain alkane and preparation method and application of drug delivery system |
| EP3225112B2 (en) | 2016-04-01 | 2025-03-26 | TriOptoTec GmbH | Photosensitiser dispersion and use of the same |
| US10070648B2 (en) | 2016-04-15 | 2018-09-11 | Rosalind Franklin University Of Medicine And Science | Photodynamic insecticides |
| US11331304B2 (en) * | 2016-05-11 | 2022-05-17 | The Jackson Laboratory | YAP1 inhibitors and methods |
| WO2018135882A1 (en) | 2017-01-19 | 2018-07-26 | 가톨릭대학교 산학협력단 | Substance having a recognition function for virus diagnosis and therapy and method for producing same. |
| CN107417706B (en) | 2017-08-04 | 2019-07-16 | 大连理工大学 | Chlorin artesunate conjugate with photo- and sound-sensitizing activity and its preparation method and application |
| AR112694A1 (en) | 2017-08-16 | 2019-11-27 | Suncor Energy Inc | PHOTODYNAMIC INHIBITION OF MICROBIAL PATHOGENS IN PLANTS |
| CA3097038A1 (en) | 2018-04-30 | 2019-11-07 | Suncor Energy Inc. | Macrocyclic tetrapyrrole compounds, compositions and methods for increasing abiotic stress resistance in plants |
| BR112021014592A2 (en) | 2019-01-25 | 2021-10-05 | Suncor Energy Inc. | PHOTOSENSITIZING COMPOUNDS, MANUFACTURING METHODS AND APPLICATION IN PLANTS |
| CA3128734C (en) | 2019-02-15 | 2024-10-15 | Nutrien Ag Solutions (Canada) Inc. | Photosensitizer and chelating agent combinations for use as insecticides |
| AU2020222515B2 (en) | 2019-02-15 | 2025-10-09 | Nutrien Ag Solutions (Canada) Inc. | The use of protoporphyrin IX derivatives to improve the health of plants |
| US20230128730A1 (en) | 2020-02-20 | 2023-04-27 | Suncor Energy Inc. | Oxygen impermeable porphyrin photosensitizer film composition for application to plants |
-
2020
- 2020-02-14 AU AU2020222515A patent/AU2020222515B2/en active Active
- 2020-02-14 EP EP20755957.6A patent/EP3923729B1/en active Active
- 2020-02-14 HR HRP20251337TT patent/HRP20251337T1/en unknown
- 2020-02-14 JP JP2021547492A patent/JP7588289B2/en active Active
- 2020-02-14 CN CN202080028335.8A patent/CN113677204B/en active Active
- 2020-02-14 BR BR112021016109A patent/BR112021016109A2/en not_active Application Discontinuation
- 2020-02-14 WO PCT/CA2020/050197 patent/WO2020163964A1/en not_active Ceased
- 2020-02-14 US US17/431,294 patent/US12207655B2/en active Active
- 2020-02-14 ES ES20755957T patent/ES3051057T3/en active Active
- 2020-02-14 MX MX2021009781A patent/MX2021009781A/en unknown
-
2021
- 2021-07-26 ZA ZA2021/05283A patent/ZA202105283B/en unknown
- 2021-07-30 CL CL2021002023A patent/CL2021002023A1/en unknown
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2009052638A1 (en) | 2007-10-25 | 2009-04-30 | Innovotech Inc. | Natural photodynamic agents and their use |
| WO2010080819A1 (en) * | 2009-01-06 | 2010-07-15 | C3 Jian, Inc. | Targeted antimicrobial moieties |
| CN103601727A (en) | 2013-10-23 | 2014-02-26 | 中国医学科学院生物医学工程研究所 | Use of novel amine compound modified protoporphyrin |
Non-Patent Citations (6)
| Title |
|---|
| FRACAROLLI ET AL., JOURNAL OF PHOTOCHEMISTRY AND PHOTOBIOLOGY B: BIOLOGY, vol. 162, pages 402 - 411 |
| HAZEN, J.L., WEED TECHNOLOGY, vol. 14, 2000, pages 773 - 784 |
| JUNG ET AL.: "Toxic Tetrapyrrole Accumulation in Protoporphyrinogen IX Oxidase- Overexpressing Transgenic Rice Plants", PLANT MOL. BIOL., vol. 67, 2008, pages 535 - 546, XP019613459 * |
| S. R. COLBY: "Calculating synergistic and antagonistic responses of herbicide combinations", WEEDS, vol. 15, 1967, pages 20 - 22, XP001112961 |
| See also references of EP3923729A4 |
| THEODORA W. GREENE: "Protective Groups in Organic Chemistry", 1991, JOHN WILEY & SONS, INC. |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11910795B2 (en) | 2013-03-15 | 2024-02-27 | Suncor Energy Inc. | Natural indole auxin and aminopolycarboxylic acid herbicidal compositions |
| US12583872B2 (en) | 2019-01-25 | 2026-03-24 | Nutrien Ag Solutions (Canada) Inc. | Photosensitizer compounds, methods of manufacture and application to plants |
| US12207655B2 (en) | 2019-02-15 | 2025-01-28 | Nutrien Ag Solutions (Canada) Inc. | Protoporphyrin IX derivatives and use thereof to improve the health of plants |
| EP4578859A4 (en) * | 2022-09-06 | 2026-03-25 | Nanjing Bostec Biological Eng Co Ltd | NATURAL PORPHINE SALT AND USE OF IT AS A PLANT GROWTH REGULATOR AND IMMUNE RESISTANCE INDUCTOR |
| WO2026012814A1 (en) | 2024-07-10 | 2026-01-15 | Basf Se | Compositions and methods to enhance crop yield and plant health |
Also Published As
| Publication number | Publication date |
|---|---|
| CN113677204A (en) | 2021-11-19 |
| AU2020222515B2 (en) | 2025-10-09 |
| EP3923729C0 (en) | 2025-08-13 |
| MX2021009781A (en) | 2021-09-08 |
| EP3923729B1 (en) | 2025-08-13 |
| AU2020222515A1 (en) | 2021-08-19 |
| ES3051057T3 (en) | 2025-12-26 |
| CA3128733A1 (en) | 2020-08-20 |
| US20220132855A1 (en) | 2022-05-05 |
| EP3923729A4 (en) | 2022-11-23 |
| CL2021002023A1 (en) | 2022-01-07 |
| US12207655B2 (en) | 2025-01-28 |
| ZA202105283B (en) | 2025-11-26 |
| HRP20251337T1 (en) | 2025-12-19 |
| JP7588289B2 (en) | 2024-11-22 |
| EP3923729A1 (en) | 2021-12-22 |
| BR112021016109A2 (en) | 2021-11-09 |
| CN113677204B (en) | 2024-03-05 |
| JP2022520450A (en) | 2022-03-30 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP3923729B1 (en) | The use of protoporphyrin ix derivatives to improve the health of plants | |
| AU2020220245B2 (en) | Photosensitizer and chelating agent combinations for use as insecticides | |
| JP7713455B2 (en) | Oxygen-impermeable porphyrin photosensitizer film compositions for plant applications - Patents.com | |
| AU2020211650B2 (en) | Photosensitizer compounds, methods of manufacture and application to plants | |
| KR101897840B1 (en) | Mesoionic pyrido [1,2-a] pyrimidine pesticides | |
| CA3128733C (en) | The use of protoporphyrin ix derivatives to improve the health of plants |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 20755957 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 3128733 Country of ref document: CA |
|
| ENP | Entry into the national phase |
Ref document number: 2021547492 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2020222515 Country of ref document: AU Date of ref document: 20200214 Kind code of ref document: A |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112021016109 Country of ref document: BR |
|
| ENP | Entry into the national phase |
Ref document number: 2020755957 Country of ref document: EP Effective date: 20210915 |
|
| ENP | Entry into the national phase |
Ref document number: 112021016109 Country of ref document: BR Kind code of ref document: A2 Effective date: 20210814 |
|
| WWG | Wipo information: grant in national office |
Ref document number: MX/A/2021/009781 Country of ref document: MX |
|
| WWG | Wipo information: grant in national office |
Ref document number: 2020755957 Country of ref document: EP |