WO2024251695A1 - Recombinant cd47 binding proteins and their use - Google Patents
Recombinant cd47 binding proteins and their use Download PDFInfo
- Publication number
- WO2024251695A1 WO2024251695A1 PCT/EP2024/065257 EP2024065257W WO2024251695A1 WO 2024251695 A1 WO2024251695 A1 WO 2024251695A1 EP 2024065257 W EP2024065257 W EP 2024065257W WO 2024251695 A1 WO2024251695 A1 WO 2024251695A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- seq
- ankyrin repeat
- amino acid
- acid sequence
- amino acids
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/705—Receptors; Cell surface antigens; Cell surface determinants
- C07K14/70596—Molecules with a "CD"-designation not provided for elsewhere
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/705—Receptors; Cell surface antigens; Cell surface determinants
- C07K14/70503—Immunoglobulin superfamily
Definitions
- the present invention relates to recombinant binding proteins comprising a designed ankyrin repeat domain with binding specificity for CD47.
- the invention relates to nucleic acids encoding such binding proteins, pharmaceutical compositions comprising such binding proteins or nucleic acids, and the use of such binding proteins, nucleic acids or pharmaceutical compositions in methods of treating diseases, such as cancer, in a mammal, including a human.
- innate immune checkpoint molecules expressed on certain antigen-presenting cells play a crucial role in immune evasion.
- APCs antigen-presenting cells
- Innate immune cells including macrophages, monocytes, dendritic cells (DCs), and natural killer (NK) cells, are the first line of the immune defense system. They establish proinflammatory responses to foreign invaders and repair damaged tissues (Liu et al; Signal Transduct Target Ther; 2023 Mar 7;8(1):104).
- Cancer cells can evade clearance by macrophages via overexpressing anti-phagocytic membrane proteins causing so-called “don’t eat me” signals, including cluster of differentiation 47 (CD47) (Zhang et al: Front Immunol; 2020 Jan 28; 11 :18).
- CD47 cluster of differentiation 47
- CD47 is a cell surface glycoprotein, which includes an amino terminal extracellular variable region, a transmembrane region formed of a highly hydrophobic transmembrane segment, and a hydrophilic carboxy-terminal cytoplasmic tail. It functions as a regulator of phagocytosis mediated by cells of the innate immune system by interacting with multiple ligands, such as integrins, signal regulatory protein alpha (SIRPa), signal regulatory protein gamma (SIRPy) and thrombospondins.
- SIRPa signal regulatory protein alpha
- SIRPy signal regulatory protein gamma
- CD47 inhibits phagocytosis by interacting with the transmembrane protein SIRPa on the surface of phagocytes, such as macrophages, neutrophils and dendritic cells, forming the CD47-SIRPa signaling complex and triggering a "don't eat me” signal.
- SIRPa transmembrane protein
- CD47 is overexpressed in different types of tumors, including in myeloma, leiomyosarcoma, acute lymphocytic leukaemia, non-Hodgkin’s lymphoma, breast cancer, osteosarcoma, and head and neck squamous cell carcinoma (Zhang et al: Front Immunol; 2020 Jan 28; 11 :18).
- CD47 Through upregulated expression of CD47, tumor cells manage to evade phagocytosis and escape from innate immune surveillance and therefore CD47 shows a remarkable potential as a therapeutic target.
- CD47 is also broadly expressed on normal cells, such as hematopoietic cells, red blood cells (RBCs) and platelets, thus posing safety and efficacy challenges, as targeting CD47, for example with a neutralizing antibody, could affect healthy cells, potentially leading to toxic effects (Ye at al; Acta Pharm Sin B. 2023 Apr;13(4):1467-1487). Additionally, broad expression of CD47 could also lead to rapid elimination of CD47 binding agents, leading to poor pharmacokinetics and decreased efficacy (Dizman et al; Cancers (Basel). 2021 Dec; 13(24): 6229).
- CD47 In addition to playing a critical role in cancer and the tumor microenvironment, CD47 is implicated in many other diseases. For example, in pulmonary arterial hypertension (PAH) the levels of CD47 are increased and promote hypoxia and ROS production in the environment (Novelli et al; Am. J. Physiol. Lung Cell Mol. Physiol. 316, L1150-L1164 (2019)). Activated CD47 was also found to promote acute kidney injury (AKI) by limiting autophagy, and has been demonstrated to be a target for preserving renal function following injury (El-Rashid et al; FASEB J. 33, 12735-12749 (2019)). Furthermore, CD47 mediates immune escape in infectious diseases caused by parasites, bacteria, and viruses, including SARS-CoV-2 in COVID pathogenesis (Cham et al; Antibodies; 9, 44 (2020)).
- PAH pulmonary arterial hypertension
- Activated CD47 was also found to promote acute kidney injury (AKI) by limiting autophag
- the present invention provides recombinant binding proteins comprising a designed ankyrin repeat domain with binding specificity for CD47. Further provided are such recombinant binding proteins linked to one or more binding agents, preferably ankyrin repeat domains, with binding specificity for a protein expressed on the surface of an immune cell, for human serum albumin and/or for a further Tumor Associated Antigen (TAA).
- TAA Tumor Associated Antigen
- the invention provides nucleic acids encoding such recombinant binding proteins and pharmaceutical compositions comprising such recombinant binding proteins or nucleic acids.
- the invention also provides the use of such recombinant binding proteins, nucleic acids or pharmaceutical compositions in methods of treating diseases, such as cancer, in a mammal, including a human.
- CD47-specific binding domains which are suitable to be used as building blocks in diagnostic or therapeutic agents, such as, e.g., multi-specific binding proteins, and have beneficial properties.
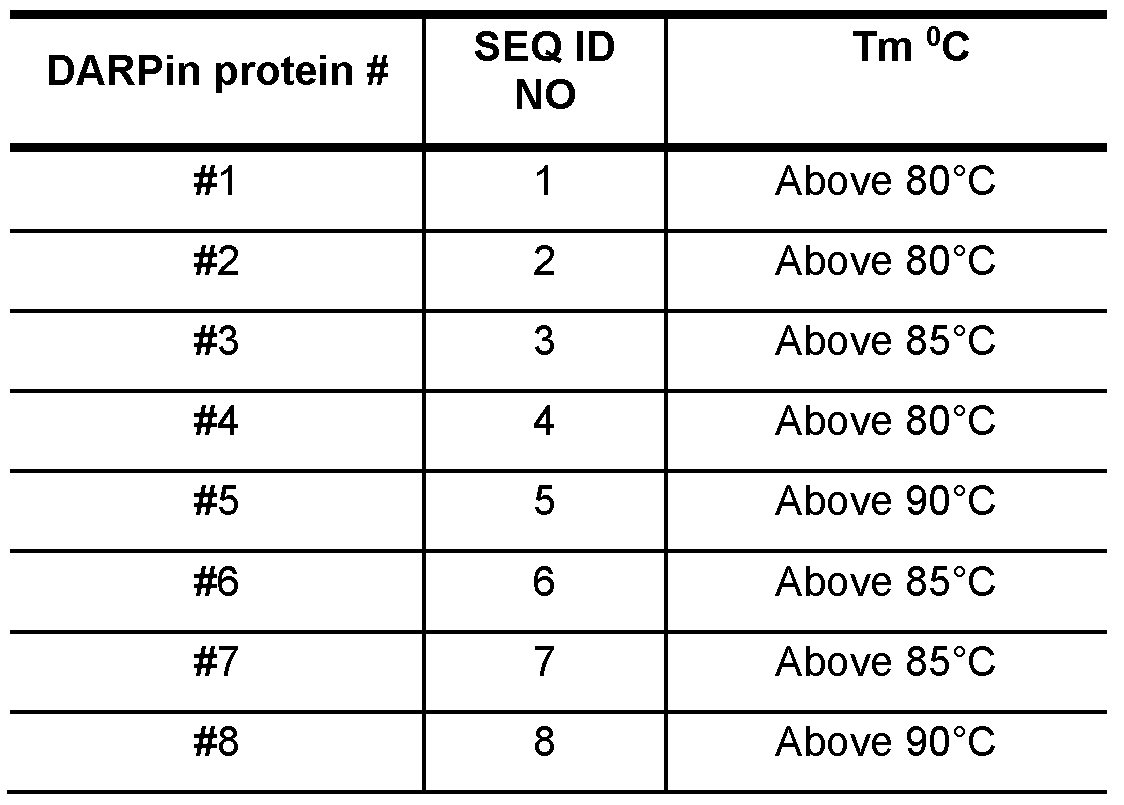
- multiple recombinant CD47-specific ankyrin repeat proteins were generated and produced, namely DARPin proteins comprising SEQ ID NO: 1 , SEQ ID NO: 2, SEQ ID NO: 3, SEQ ID NO: 4, SEQ ID NO: 5, SEQ ID NO: 6, SEQ ID NO: 7 and SEQ ID NO: 8.
- DARPin proteins comprising SEQ ID NO: 1 , SEQ ID NO: 2, SEQ ID NO: 3, SEQ ID NO: 4, SEQ ID NO: 5, SEQ ID NO: 6, SEQ ID NO: 7 and SEQ ID NO: 8.
- These recombinant binding proteins all comprise a designed ankyrin repeat domain with binding specificity for CD47.
- binding proteins are suitable to be linked to one or more Tumor Associated Antigen (TAA)-specific binding agent(s), which may result in avidity driven multi-specific molecules.
- TAA Tumor Associated Antigen
- binding proteins of the invention may further be linked to other binding agents, such as binding agents with binding specificity for surface molecules expressed by immune cells.
- a recombinant binding protein comprising an ankyrin repeat domain with binding specificity for CD47, wherein said ankyrin repeat domain comprises an ankyrin repeat module having an amino acid sequence selected from the group consisting of (1) SEQ ID NOs: 9 to 21 and (2) sequences in which up to 9 amino acids in any of SEQ ID NOs: 9 to 21 are substituted by other amino acids.
- E2 The recombinant binding protein of E1 , wherein said ankyrin repeat module is a first ankyrin repeat module and wherein said ankyrin repeat domain further comprises a second ankyrin repeat module having an amino acid sequence selected from the group consisting of (1) SEQ ID NOs: 9 to 21 and (2) sequences in which up to 9 amino acids in any of SEQ ID NOs: 9 to 21 are substituted by other amino acids.
- E3 The recombinant binding protein of E2, wherein said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 9 and (2) sequences in which up to 9 amino acids in SEQ ID NO: 9 are substituted by other amino acids, and wherein said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 10 and (2) sequences in which up to 9 amino acids of SEQ ID NO: 10 are substituted by other amino acids.
- E4 The recombinant binding protein of E2, wherein said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 11 and (2) sequences in which up to 9 amino acids in SEQ ID NO: 11 are substituted by other amino acids, and wherein said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 12 and (2) sequences in which up to 9 amino acids of SEQ ID NO: 12 are substituted by other amino acids.
- E5. The recombinant binding protein of E2, wherein said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 13 and (2) sequences in which up to 9 amino acids in SEQ ID NO: 13 are substituted by other amino acids, and wherein said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 14 and (2) sequences in which up to 9 amino acids of SEQ ID NO: 14 are substituted by other amino acids.
- E6 The recombinant binding protein of E2, wherein said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 15 and (2) sequences in which up to 9 amino acids in SEQ ID NO: 15 are substituted by other amino acids, and wherein said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 16 and (2) sequences in which up to 9 amino acids of SEQ ID NO: 16 are substituted by other amino acids.
- E7 The recombinant binding protein of E2, wherein said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 17 and (2) sequences in which up to 9 amino acids in SEQ ID NO: 17 are substituted by other amino acids, and wherein said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 18 and (2) sequences in which up to 9 amino acids of SEQ ID NO:
- E8 The recombinant binding protein of E2, wherein said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 13 and (2) sequences in which up to 9 amino acids in SEQ ID NO: 13 are substituted by other amino acids, and wherein said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 19 and (2) sequences in which up to 9 amino acids of SEQ ID NO:
- E9 The recombinant binding protein of E2, wherein said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 17 and (2) sequences in which up to 9 amino acids in SEQ ID NO: 17 are substituted by other amino acids, and wherein said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 20 and (2) sequences in which up to 9 amino acids of SEQ ID NO:
- E10 The recombinant binding protein of E2, wherein said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 17 and (2) sequences in which up to 9 amino acids in SEQ ID NO: 17 are substituted by other amino acids, and wherein said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 21 and (2) sequences in which up to 9 amino acids of SEQ ID NO:
- a recombinant binding protein comprising an ankyrin repeat domain with binding specificity for CD47, wherein said ankyrin repeat domain comprises an amino acid sequence with at least 80% amino acid sequence identity with any one of SEQ ID NOs: 1 to 8, and wherein A at the second last position of SEQ ID NOs: 1 to 8 is optionally substituted by L, and/or A at the last position of SEQ ID NOs: 1 to 8 is optionally substituted by N.
- E13 The recombinant binding protein of any of E1 to E12, wherein said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) of or below 10 -7 M, of or below 10 -8 M, of or below 10 -9 M.
- KD dissociation constant
- E14 The recombinant binding protein of any of E1 to E13, wherein said ankyrin repeat domain with binding specificity for CD47 comprises an amino acid sequence with at least 80% amino acid sequence identity with SEQ ID NO: 1 .
- E15 The recombinant binding protein of E14, wherein said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) of or below 10 -8 M or of or below 10 -9 M.
- KD dissociation constant
- E16 The recombinant binding protein of any of E1 to E13, wherein said ankyrin repeat domain with binding specificity for CD47 comprises an amino acid sequence with at least 80% amino acid sequence identity with SEQ ID NO: 2.
- E17 The recombinant binding protein of E16, wherein said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) of or below about 10 -8 M or of or below 10 -9 M.
- KD dissociation constant
- E18 The recombinant binding protein of any of E1 to E13, wherein said ankyrin repeat domain with binding specificity for CD47 comprises an amino acid sequence with at least 80% amino acid sequence identity with SEQ ID NO: 3.
- E19 The recombinant binding protein of E18, wherein said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) of or below about 10 -8 M or of or below 10 -9 M.
- KD dissociation constant
- E20 The recombinant binding protein of any of E1 to E13, wherein said ankyrin repeat domain with binding specificity for CD47 comprises an amino acid sequence with at least 80% amino acid sequence identity with SEQ ID NO: 4.
- E21 The recombinant binding protein of E20, wherein said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) of or below 10 -8 M or of or below 10 -9 M.
- KD dissociation constant
- E22 The recombinant binding protein of any of E1 to E13, wherein said ankyrin repeat domain with binding specificity for CD47 comprises an amino acid sequence with at least 80% amino acid sequence identity with SEQ ID NO: 5.
- E23 The recombinant binding protein of E22, wherein said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) of or below 10 -8 M or of or below 10 -9 M.
- KD dissociation constant
- E24 The recombinant binding protein of any of E1 to E13, wherein said ankyrin repeat domain with binding specificity for CD47 comprises an amino acid sequence with at least 80% amino acid sequence identity with SEQ ID NO: 6.
- E25 The recombinant binding protein of E24, wherein said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) of or below 10 -8 M or of or below 10 -9 M.
- KD dissociation constant
- E26 The recombinant binding protein of any of E1 to E13, wherein said ankyrin repeat domain with binding specificity for CD47 comprises an amino acid sequence with at least 80% amino acid sequence identity with SEQ ID NO: 7.
- E27 The recombinant binding protein of E26, wherein said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) of or below 10 -8 M or of or below 10 -9 M.
- KD dissociation constant
- E28 The recombinant binding protein of any of E1 to E13, wherein said ankyrin repeat domain with binding specificity for CD47 comprises an amino acid sequence with at least 80% amino acid sequence identity with SEQ ID NO: 8.
- E29 The recombinant binding protein of E28, wherein said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) of or below 10 -8 M or of or below 10 -9 M.
- KD dissociation constant
- E30 The recombinant binding protein of any of E1 to E29, wherein said recombinant binding protein further comprises a binding agent with binding specificity for a tumor-associated antigen.
- E31 The recombinant binding protein of E30, wherein said binding agent with binding specificity for a tumor-associated antigen is an ankyrin repeat domain.
- E32 The recombinant binding protein of any one of E1 to E31 , wherein said recombinant binding protein further comprises a binding agent with binding specificity for a protein expressed on the surface of an immune cell, preferably an NK cell or a macrophage.
- E33 The recombinant binding protein of E32, wherein said binding agent with binding specificity for a protein expressed on the surface of an immune cell, preferably an NK cell or a macrophage, has binding specificity for CD16a.
- E34 The recombinant binding protein of E32 or E33, wherein said binding agent with binding specificity for a protein expressed on the surface of an immune cell is a designed ankyrin repeat protein.
- E35 The recombinant binding protein of E34, wherein said binding agent with binding specificity said binding agent with binding specificity for a protein expressed on the surface of an immune cell comprises an amino acid sequence with at least 80%, 81 %, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% amino acid sequence identity with SEQ ID NO: 81 .
- E36 The recombinant binding protein of any one of E18 to E35, wherein said binding agent with binding specificity for a tumor-associated antigen and/or said binding agent with binding specificity for a protein expressed on the surface of an immune cell, preferably an NK cell or a macrophage, is covalently linked to said ankyrin repeat domain with binding specificity for CD47 with a peptide linker.
- E37 The recombinant binding protein of E36, wherein said peptide linker is a proline-threonine rich peptide linker or glycine-serine rich linker.
- E38 The recombinant binding protein of E34 to E37, wherein the amino acid sequence of said peptide linker has a length from 1 to 50 amino acids, preferably from 6 to 38 amino acids.
- E39 The recombinant binding protein of any of E1 to E38, wherein said recombinant binding protein further comprises a half-life extending moiety.
- E40 The recombinant binding protein of E39, wherein said half-life extending moiety comprises a binding agent with binding specificity for human serum albumin.
- E41 The recombinant binding protein of E40, wherein said binding agent with binding specificity for human serum albumin is an ankyrin repeat domain comprising an amino acid sequence of any one of SEQ ID NOs: 62 to 64, preferably SEQ ID NO: 63.
- E42 The recombinant binding protein of any one of E39 to E41 , wherein said half-life extending moiety is located N-terminally of said ankyrin repeat domain with binding specificity for CD47 within said binding protein.
- E43 A nucleic acid encoding the recombinant binding protein of any one of E1 to E42.
- E44 A cloning or expression vector comprising the nucleic acid of E43 wherein said vector is suitable for the recombinant production of a binding protein as defined in any one of E1 to E42.
- E45 The vector of E44, wherein said vector is a DNA vector, an RNA vector, a plasmid, a cosmid, or a viral vector.
- E46 A host cell comprising the nucleic acid of E43 or the vector of any one of E44 to E45.
- E47 A pharmaceutical composition comprising the recombinant binding protein of any one of E1 to E42 or the nucleic acid of E43, the vector of any one of E44 to E45 or the host cell of E46 and optionally a pharmaceutically acceptable carrier and/or diluent.
- E49 The recombinant binding protein of E48, wherein the medical condition is cancer.
- E50 A method of treating a medical condition, the method comprising the step of administering to a subject in need thereof a therapeutically effective amount of the recombinant binding protein of any one of E1 to E42, the nucleic acid of E43 or the pharmaceutical composition of E47.
- E51 The method of E50, wherein said medical condition is a cancer.
- E53 Use of the recombinant binding protein of any one of E1 to E42 or the nucleic acid of E43 or the pharmaceutical composition of E47 for the manufacture of a medicament for the treatment of a medical condition.
- E54 The use of E53, wherein said medical condition is a cancer.
- E56 A method for the production of protein according to any one of E1 to E42, comprising culturing a host cell according to E46, purifying and recovering said protein.
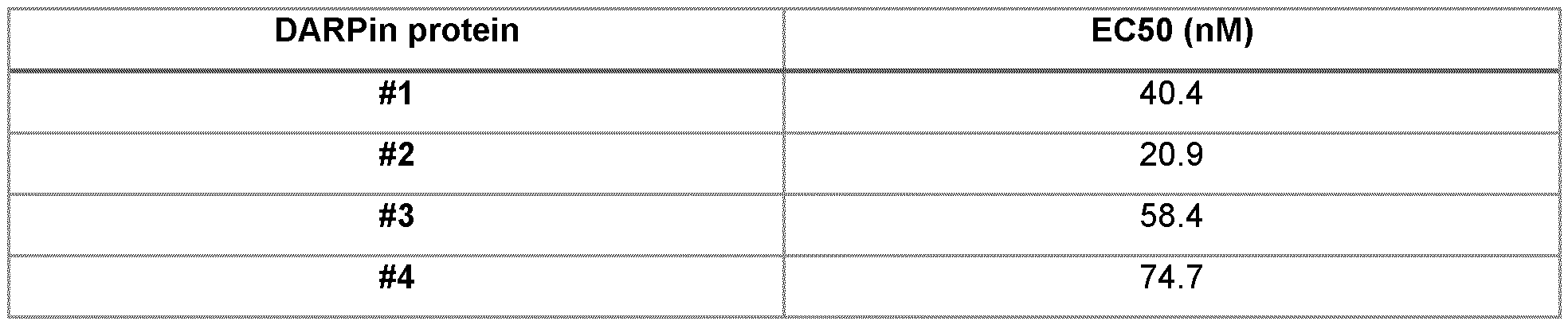
- FIG. 1 Binding of 40 nM Flag-tagged 96-well IMAC purified proteins to 10 nM biotinylated human CD47- Fc in Homogeneous Time Resolved Fluorescence (HTRF).
- the HTRF signal over PBS is shown for the eight selected CD47-specific designed ankyrin repeat proteins.
- Columns 1 to 8 show the HTRF signal of DARPin protein #1 , DARPin protein #2, DARPin protein #3, DARPin protein #4, DARPin protein #5, DARPin protein #6, DARPin protein #7 and DARPin protein #8, respectively.
- Figure 2 (A-D). SIRPIa competition on cells.
- DARPin protein #6 Titrated amounts of selected designed ankyrin repeat proteins (96 well purified) or anti-CD47 benchmark antibody control was added to the cells (CD47 presenting ligand cells and SIRPIa signaling cells). Reduction of signal indicates ligand competition.
- Fig. 2A DARPin protein #3; Fig.2B. DARPin protein #4 and DARPin protein #7; Fig. 2C. DARPin protein #1 , DARPin protein #2, DARPin protein #5 and DARPin protein #8; Fig. 2D. DARPin protein #6.
- Figure 3 (A-C). Cell binding of 96-well purified designed ankyrin repeat proteins. Titrated amount of selected designed ankyrin repeat proteins was added to CD47-expressing cells for 30 minutes at 4°C. Tested proteins bound to cells were detected with anti-DARPin 1.1.1 antibodies. Median fluorescence intensities obtained are shown.
- Fig. 3A Cell binding of DARPin protein #1 and #2;
- Fig. 3B Cell binding of DARPin protein #3, #4 and #5;
- Fig. 3C Cell binding of DARPin protein #6, #7 and #8.
- Figure 4 shows SPR traces obtained for DARPin molecule #1 (Fig. 4A), DARPin molecule #2 (Fig. 4B) and DARPin molecule #3 (Fig. 4C).
- Dissociation constants KD were calculated from the estimated on- and off-rates using standard procedures. KD values of the binding interactions of selected ankyrin repeat proteins with human CD47 were determined to be in the range of 0.1 to 0.8 nM (RU, Resonance Units; s, time in seconds).
- FIG. 5 Pharmacokinetic analysis of exemplary CD47-specific binding proteins in female BALB/c mice.
- FIG. 6 SIRPIa competition on cells. Titrated amounts of CD47-specific binding proteins (SEQ ID NOs: 1 to 7) in a trispecific format, or a benchmark anti-CD47 antibody as a control, were added to the cells (CD47 positive/TAA positive ligand cells and SIRPIa signaling cells). Reduction of signal indicates ligand competition.
- FIG. 7 A-B. SIRPIa competition on cells. Titrated amounts of the CD47-specific binding protein (SEQ ID NO: 8), in monospecific and trispecific format or the benchmark anti-CD47 antibody were added to the SIRPIa signaling cells in the presence of either CD47 positive/TAA negative ligand cells (Fig. 7A) or CD47 positive/TAA positive ligand cells (Fig. 7B). Reduction of signal indicates ligand competition.
- SEQ ID NO: 8 CD47-specific binding protein
- FIG. 8 SIRPIa competition on cells. Titrated amounts of selected DARPin proteins with binding specificity for CD47, CD117 and CD16a or the benchmark anti-CD47 antibody were added to the SIRPI a signaling cells in the presence of Kasumi-1 cells. Reduction of signal indicates ligand competition.
- the disclosure provides ankyrin repeat proteins that specifically bind target CD47.
- Designed ankyrin repeat protein libraries (W02002/020565; Binz et al., Nat. Biotechnol. 22, 575-582, 2004; Stumpp et al., Drug Discov. Today 13, 695-701 , 2008) can be used for the selection of target-specific designed ankyrin repeat domains that bind to their target with high affinity.
- target- specific designed ankyrin repeat domains in turn can be used as valuable components of recombinant binding proteins for the treatment of diseases.
- Designed ankyrin repeat proteins are a class of binding molecules which have the potential to overcome limitations of monoclonal antibodies, hence allowing novel therapeutic approaches.
- Such ankyrin repeat proteins may comprise a single designed ankyrin repeat domain, or may comprise a combination of two or more designed ankyrin repeat domains with the same or different target specificities (Stumpp et al., Drug Discov. Today 13, 695-701 , 2008; U.S. Patent No. 9,458,211).
- Ankyrin repeat proteins comprising only a single designed ankyrin repeat domain are small proteins (14 kDa) which can be selected to bind a given target protein with high affinity and specificity.
- ankyrin repeat proteins ideal agonistic, antagonistic and/or inhibitory drug candidates and allow for novel drug designs with increased safety profile, including, e.g., multispecific drugs comprising a CD47-specific binding agent and at least one further tumor localizer such as a tumor associated antigen binding agent, and wherein said CD47-specific agent is blocked or masked with a binding agent specific for the CD47-specific agent and wherein said CD47-specific agent is released only when said tumor associated antigen binding agent binds to the tumor associated agent, thus preventing binding of said CD47-specific binding agent to normal CD47-expressing cells.
- multispecific drugs comprising a CD47-specific binding agent and at least one further tumor localizer such as a tumor associated antigen binding agent, and wherein said CD47-specific agent is blocked or masked with a binding agent specific for the CD47-specific agent and wherein said CD47-specific agent is released only when said tumor associated antigen binding agent binds to the tumor associated agent, thus preventing binding of said CD47-specific binding agent to normal CD
- said recombinant binding protein comprises an ankyrin repeat domain with binding specificity for CD47 as described herein and further comprises an ankyrin repeat domain with dual binding specificity, wherein binding of said ankyrin repeat domain with dual binding specificity to its first and second binding targets is mutually exclusive.
- ankyrin repeat domains with dual binding specificity also referred to as “2-in-1 repeat domains”
- production methods are described in PCT/EP2022/085794.
- said recombinant binding protein comprises an ankyrin repeat domain with binding specificity for CD47 as described herein and further comprises a second ankyrin repeat domain, wherein the second ankyrin repeat domain is a 2-in-1 repeat domain having a first binding specificity for a TAA and a second binding specificity for said first ankyrin repeat domain having binding specificity for CD47.
- the 2-in-1 repeat domain binds the TAA and releases the CD47-specific binding domain from binding to the 2-in-1 repeat domain, thereby releasing the inhibition of the CD47-specific binding domain.
- ankyrin repeat proteins can be engineered to carry various effector functions, e.g. cytotoxic agents or half-life extending agents, enabling completely new drug formats.
- cytotoxic agents e.g. cytotoxic agents or half-life extending agents
- designed ankyrin repeat proteins are an example of the next generation of protein therapeutics with the potential to surpass existing antibody drugs.
- the invention relates to a recombinant binding protein comprising an ankyrin repeat domain, wherein said ankyrin repeat domain has binding specificity for CD47, and wherein said ankyrin repeat domain comprises an ankyrin repeat module having an amino acid sequence selected from the group consisting of (1) SEQ ID NOs: 9 to 21 and (2) sequences in which up to 9, or up to 8, or up to 7, or up to 6, or up to 5, or up to 4, or up to 3, or up to 2, or up to 1 amino acids in any of SEQ ID NOs: 9 to 21 are substituted by other amino acids.
- said ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NOs: 9 to 21 and (2) sequences in which up to 9 amino acids in any of SEQ ID NOs: 9 to 21 are substituted by other amino acids.
- said ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NOs: 9 to 21 and (2) sequences in which up to 8 amino acids in any of SEQ ID NOs: 9 to 21 are substituted by other amino acids.
- said ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NOs: 9 to 21 and (2) sequences in which up to 7 amino acids in any of SEQ ID NOs: 9 to 21 are substituted by other amino acids. In one embodiment, said ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NOs: 9 to 21 and (2) sequences in which up to 6 amino acids in any of SEQ ID NOs: 9 to 21 are substituted by other amino acids. In one embodiment, said ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NOs: 9 to 21 and (2) sequences in which up to 5 amino acids in any of SEQ ID NOs: 9 to 21 are substituted by other amino acids.
- said ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NOs: 9 to 21 and (2) sequences in which up to 4 amino acids in any of SEQ ID NOs: 9 to 21 are substituted by other amino acids. In one embodiment, said ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NOs: 9 to 21 and (2) sequences in which up to 3 amino acids in any of SEQ ID NOs: 9 to 21 are substituted by other amino acids. In one embodiment, said ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NOs: 9 to 21 and (2) sequences in which up to 2 amino acids in any of SEQ ID NOs: 9 to 21 are substituted by other amino acids.
- said ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NOs: 9 to 21 and (2) sequences in which up to 1 amino acid in any of SEQ ID NOs: 9 to 21 is substituted by another amino acid. In one embodiment, all of said 9, 8, 7, 6, 5, 4, 3, 2, or 1 amino acid substitutions occur in framework positions of said ankyrin repeat module(s). In one embodiment, said ankyrin repeat module comprises an amino acid sequence selected from the group consisting of SEQ ID NOs: 9 to 21 .
- said ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 9 or a sequence in which one or two amino acids in SEQ ID NO: 9 are substituted by other amino acids. In one embodiment, said ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 10 or a sequence in which one or two amino acids in SEQ ID NO: 10 are substituted by other amino acids. In one embodiment, said ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 11 or a sequence in which one or two amino acids in SEQ ID NO: 11 are substituted by other amino acids.
- said ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 12 or a sequence in which one or two amino acids in SEQ ID NO: 12 are substituted by other amino acids. In one embodiment, said ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 13 or a sequence in which one or two amino acids in SEQ ID NO: 13 are substituted by other amino acids. In one embodiment, said ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 14 or a sequence in which one or two amino acids in SEQ ID NO: 14 are substituted by other amino acids.
- said ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 15 or a sequence in which one or two amino acids in SEQ ID NO: 15 are substituted by other amino acids. In one embodiment, said ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 16 or a sequence in which one or two amino acids in SEQ ID NO: 16 are substituted by other amino acids. In one embodiment, said ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 17 or a sequence in which one or two amino acids in SEQ ID NO: 17 are substituted by other amino acids.
- said ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 18 or a sequence in which one or two amino acids in SEQ ID NO: 18 are substituted by other amino acids. In one embodiment, said ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 19 or a sequence in which one or two amino acids in SEQ ID NO: 19 are substituted by other amino acids. In one embodiment, said ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 20 or a sequence in which one or two amino acids in SEQ ID NO: 20 are substituted by other amino acids.
- said ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 21 or a sequence in which one or two amino acids in SEQ ID NO: 21 are substituted by other amino acids. In one embodiment, said ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 9. In one embodiment, said ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 10. In one embodiment, said ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 11. In one embodiment, said ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 12. In one embodiment, said ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 13. In one embodiment, said ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 14.
- said ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 15. In one embodiment, said ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 16. In one embodiment, said ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 17. In one embodiment, said ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 18. In one embodiment, said ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 19. In one embodiment, said ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 20. In one embodiment, said ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 21 .
- said ankyrin repeat domain comprises a first ankyrin repeat module and a second ankyrin repeat module.
- said first ankyrin repeat module is located N-terminally of said second ankyrin repeat module within said ankyrin repeat domain.
- said first and said second ankyrin repeat module each comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NOs: 9 to 21 and (2) sequences in which up to 9, or up to 8, or up to 7, or up to 6, or up to 5, or up to 4, or up to 3, or up to 2, or up to 1 amino acids in any of SEQ ID NOs: 9 to 21 are substituted by other amino acids.
- said ankyrin repeat domain comprises a first ankyrin repeat module having an amino acid sequence selected from the group consisting of (1) SEQ ID NOs: 9 to 21 and (2) sequences in which up to 9 amino acids in any of SEQ ID NOs: 9 to 21 are substituted by other amino acids and further comprises a second ankyrin repeat module having an amino acid sequence selected from the group consisting of (1) SEQ ID NOs: 9 to 21 and (2) sequences in which up to 9 amino acids in any of SEQ ID NOs: 9 to 21 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 9 and (2) sequences in which up to 9, or up to 8, or up to 7, or up to 6, or up to 5, or up to 4, or up to 3, or up to 2, or up to 1 amino acids in SEQ ID NO: 9 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 10 and (2) sequences in which up to 9, or up to 8, or up to 7, or up to 6, or up to 5, or up to 4, or up to 3, or up to 2, or up to 1 amino acids of SEQ ID NO: 10 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 9 and (2) sequences in which up to 9 amino acids in SEQ ID NO: 9 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 10 and (2) sequences in which up to 9 amino acids of SEQ ID NO: 10 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 9 and (2) sequences in which up to 6 amino acids in SEQ ID NO: 9 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 10 and (2) sequences in which up to 6 amino acids of SEQ ID NO: 10 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 9 and (2) sequences in which up to 5 amino acids in SEQ ID NO: 9 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 10 and (2) sequences in which up to 5 amino acids of SEQ ID NO: 10 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 9 and (2) sequences in which up to 4 amino acids in SEQ ID NO: 9 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 10 and (2) sequences in which up to 4 amino acids of SEQ ID NO: 10 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 9 and (2) sequences in which up to 3 amino acids in SEQ ID NO: 9 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 10 and (2) sequences in which up to 3 amino acids of SEQ ID NO: 10 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 9 and (2) sequences in which up to 2 amino acids in SEQ ID NO: 9 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 10 and (2) sequences in which up to 2 amino acids of SEQ ID NO: 10 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 9 and (2) sequences in which 1 amino acid in SEQ ID NO: 9 is substituted by another amino acid
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 10 and (2) sequences in which 1 amino acid of SEQ ID NO: 10 is substituted by another amino acid.
- said first ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 9
- said second ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 10.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 9 and (2) sequences in which up to 3, or up to 2, or up to 1 amino acids in SEQ ID NO: 9 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 10 and (2) sequences in which up to 3, or up to 2, or up to 1 amino acids of SEQ ID NO: 10 are substituted by other amino acids, wherein said first ankyrin repeat module is located N-terminally of said second ankyrin repeat module within said ankyrin repeat domain.
- said first ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 9 and said second ankyrin repeat module comprises the amino acid sequence of SEQ ID NO:10, wherein said first ankyrin repeat module is located N-terminally of said second ankyrin repeat module within said ankyrin repeat domain.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 11 and (2) sequences in which up to 9, or up to 8, or up to 7, or up to 6, or up to 5, or up to 4, or up to 3, or up to 2, or up to 1 amino acids in SEQ ID NO: 11 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 12 and (2) sequences in which up to 9, or up to 8, or up to 7, or up to 6, or up to 5, or up to 4, or up to 3, or up to 2, or up to 1 amino acids of SEQ ID NO: 12 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 11 and (2) sequences in which up to 9 amino acids in SEQ ID NO: 11 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 12 and (2) sequences in which up to 9 amino acids of SEQ ID NO: 12 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 11 and (2) sequences in which up to 6 amino acids in SEQ ID NO: 11 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 12 and (2) sequences in which up to 6 amino acids of SEQ ID NO: 12 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 11 and (2) sequences in which up to 5 amino acids in SEQ ID NO: 11 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 12 and (2) sequences in which up to 5 amino acids of SEQ ID NO: 12 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 11 and (2) sequences in which up to 4 amino acids in SEQ ID NO: 11 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 12 and (2) sequences in which up to 4 amino acids of SEQ ID NO: 12 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 11 and (2) sequences in which up to 3 amino acids in SEQ ID NO: 11 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 12 and (2) sequences in which up to 3 amino acids of SEQ ID NO: 12 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 11 and (2) sequences in which up to 2 amino acids in SEQ ID NO: 11 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 12 and (2) sequences in which up to 2 amino acids of SEQ ID NO: 12 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 11 and (2) sequences in which 1 amino acid in SEQ ID NO: 11 is substituted by another amino acid
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 12 and (2) sequences in which 1 amino acid of SEQ ID NO: 12 is substituted by another amino acid.
- said first ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 11 and said second ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 12.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 11 and (2) sequences in which up to 3, or up to 2, or up to 1 amino acids in SEQ ID NO: 11 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 12 and (2) sequences in which up to 3, or up to 2, or up to 1 amino acids of SEQ ID NO: 12 are substituted by other amino acids, wherein said first ankyrin repeat module is located N-terminally of said second ankyrin repeat module within said ankyrin repeat domain.
- said first ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 11 and said second ankyrin repeat module comprises the amino acid sequence of SEQ ID NO:12, wherein said first ankyrin repeat module is located N-terminally of said second ankyrin repeat module within said ankyrin repeat domain.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 13 and (2) sequences in which up to 9, or up to 8, or up to 7, or up to 6, or up to 5, or up to 4, or up to 3, or up to 2, or up to 1 amino acids in SEQ ID NO: 13 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 14 and (2) sequences in which up to 9, or up to 8, or up to 7, or up to 6, or up to 5, or up to 4, or up to 3, or up to 2, or up to 1 amino acids of SEQ ID NO: 14 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 13 and (2) sequences in which up to 9 amino acids in SEQ ID NO: 13 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 14 and (2) sequences in which up to 9 amino acids of SEQ ID NO: 14 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 13 and (2) sequences in which up to 6 amino acids in SEQ ID NO: 13 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 14 and (2) sequences in which up to 6 amino acids of SEQ ID NO: 14 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 13 and (2) sequences in which up to 5 amino acids in SEQ ID NO: 13 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 14 and (2) sequences in which up to 5 amino acids of SEQ ID NO: 14 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 13 and (2) sequences in which up to 4 amino acids in SEQ ID NO: 13 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 14 and (2) sequences in which up to 4 amino acids of SEQ ID NO: 14 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 13 and (2) sequences in which up to 3 amino acids in SEQ ID NO: 13 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 14 and (2) sequences in which up to 3 amino acids of SEQ ID NO: 14 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 13 and (2) sequences in which up to 2 amino acids in SEQ ID NO: 13 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 14 and (2) sequences in which up to 2 amino acids of SEQ ID NO: 14 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 13 and (2) sequences in which 1 amino acid in SEQ ID NO: 13 is substituted by another amino acid
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 14 and (2) sequences in which 1 amino acid of SEQ ID NO: 14 is substituted by another amino acid.
- said first ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 13
- said second ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 14.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 13 and (2) sequences in which up to 3, or up to 2, or up to 1 amino acids in SEQ ID NO: 13 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 14 and (2) sequences in which up to 3, or up to 2, or up to 1 amino acids of SEQ ID NO: 14 are substituted by other amino acids, wherein said first ankyrin repeat module is located N-terminally of said second ankyrin repeat module within said ankyrin repeat domain.
- said first ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 13 and said second ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 14, wherein said first ankyrin repeat module is located N-terminally of said second ankyrin repeat module within said ankyrin repeat domain.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 15 and (2) sequences in which up to 9, or up to 8, or up to 7, or up to 6, or up to 5, or up to 4, or up to 3, or up to 2, or up to 1 amino acids in SEQ ID NO: 15 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 16 and (2) sequences in which up to 9, or up to 8, or up to 7, or up to 6, or up to 5, or up to 4, or up to 3, or up to 2, or up to 1 amino acids of SEQ ID NO: 16 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 15 and (2) sequences in which up to 9 amino acids in SEQ ID NO: 15 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 16 and (2) sequences in which up to 9 amino acids of SEQ ID NO: 16 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 15 and (2) sequences in which up to 6 amino acids in SEQ ID NO: 15 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 16 and (2) sequences in which up to 6 amino acids of SEQ ID NO: 16 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 15 and (2) sequences in which up to 5 amino acids in SEQ ID NO: 15 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 16 and (2) sequences in which up to 5 amino acids of SEQ ID NO: 16 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 15 and (2) sequences in which up to 4 amino acids in SEQ ID NO: 15 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 16 and (2) sequences in which up to 4 amino acids of SEQ ID NO: 16 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 15 and (2) sequences in which up to 3 amino acids in SEQ ID NO: 15 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 16 and (2) sequences in which up to 3 amino acids of SEQ ID NO: 16 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 15 and (2) sequences in which up to 2 amino acids in SEQ ID NO: 15 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 16 and (2) sequences in which up to 2 amino acids of SEQ ID NO: 16 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 15 and (2) sequences in which 1 amino acid in SEQ ID NO: 15 is substituted by another amino acid
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 16 and (2) sequences in which 1 amino acid of SEQ ID NO: 16 is substituted by another amino acid.
- said first ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 15, and said second ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 16.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 15 and (2) sequences in which up to 3, or up to 2, or up to 1 amino acids in SEQ ID NO: 15 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 16 and (2) sequences in which up to 3, or up to 2, or up to 1 amino acids of SEQ ID NO: 16 are substituted by other amino acids, wherein said first ankyrin repeat module is located N-terminally of said second ankyrin repeat module within said ankyrin repeat domain.
- said first ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 15 and said second ankyrin repeat module comprises the amino acid sequence of SEQ ID NO:16, wherein said first ankyrin repeat module is located N-terminally of said second ankyrin repeat module within said ankyrin repeat domain.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 17 and (2) sequences in which up to 9, or up to 8, or up to 7, or up to 6, or up to 5, or up to 4, or up to 3, or up to 2, or up to 1 amino acids in SEQ ID NO: 17 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 18 and (2) sequences in which up to 9, or up to 8, or up to 7, or up to 6, or up to 5, or up to 4, or up to 3, or up to 2, or up to 1 amino acids of SEQ ID NO: 18 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 17 and (2) sequences in which up to 9 amino acids in SEQ ID NO: 17 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 18 and (2) sequences in which up to 9 amino acids of SEQ ID NO: 18 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 17 and (2) sequences in which up to 6 amino acids in SEQ ID NO: 17 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 18 and (2) sequences in which up to 6 amino acids of SEQ ID NO: 18 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 17 and (2) sequences in which up to 5 amino acids in SEQ ID NO: 17 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 18 and (2) sequences in which up to 5 amino acids of SEQ ID NO: 18 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 17 and (2) sequences in which up to 4 amino acids in SEQ ID NO: 17 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 18 and (2) sequences in which up to 4 amino acids of SEQ ID NO: 18 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 17 and (2) sequences in which up to 3 amino acids in SEQ ID NO: 17 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 18 and (2) sequences in which up to 3 amino acids of SEQ ID NO: 18 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 17 and (2) sequences in which up to 2 amino acids in SEQ ID NO: 17 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 18 and (2) sequences in which up to 2 amino acids of SEQ ID NO: 18 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 17 and (2) sequences in which 1 amino acid in SEQ ID NO: 17 is substituted by another amino acid
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 18 and (2) sequences in which 1 amino acid of SEQ ID NO: 18 is substituted by another amino acid.
- said first ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 17 and said second ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 18.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 17 and (2) sequences in which up to 3, or up to 2, or up to 1 amino acids in SEQ ID NO: 17 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 18 and (2) sequences in which up to 3, or up to 2, or up to 1 amino acids of SEQ ID NO: 18 are substituted by other amino acids, wherein said first ankyrin repeat module is located N-terminally of said second ankyrin repeat module within said ankyrin repeat domain.
- said first ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 17 and said second ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 18, wherein said first ankyrin repeat module is located N-terminally of said second ankyrin repeat module within said ankyrin repeat domain.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 13 and (2) sequences in which up to 9, or up to 8, or up to 7, or up to 6, or up to 5, or up to 4, or up to 3, or up to 2, or up to 1 amino acids in SEQ ID NO: 13 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 19 and (2) sequences in which up to 9, or up to 8, or up to 7, or up to 6, or up to 5, or up to 4, or up to 3, or up to 2, or up to 1 amino acids of SEQ ID NO: 19 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 13 and (2) sequences in which up to 9 amino acids in SEQ ID NO: 13 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 19 and (2) sequences in which up to 9 amino acids of SEQ ID NO: 19 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 13 and (2) sequences in which up to 6 amino acids in SEQ ID NO: 13 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 19 and (2) sequences in which up to 6 amino acids of SEQ ID NO: 19 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 13 and (2) sequences in which up to 5 amino acids in SEQ ID NO: 13 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 19 and (2) sequences in which up to 5 amino acids of SEQ ID NO: 19 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 13 and (2) sequences in which up to 4 amino acids in SEQ ID NO: 13 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 19 and (2) sequences in which up to 4 amino acids of SEQ ID NO: 19 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 13 and (2) sequences in which up to 3 amino acids in SEQ ID NO: 13 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 19 and (2) sequences in which up to 3 amino acids of SEQ ID NO: 19 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 13 and (2) sequences in which up to 2 amino acids in SEQ ID NO: 19 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 13 and (2) sequences in which up to 2 amino acids of SEQ ID NO: 13 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 13 and (2) sequences in which 1 amino acid in SEQ ID NO: 13 is substituted by another amino acid
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 19 and (2) sequences in which 1 amino acid of SEQ ID NO: 19 is substituted by another amino acid.
- said first ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 13
- said second ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 19.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 13 and (2) sequences in which up to 3, or up to 2, or up to 1 amino acids in SEQ ID NO: 13 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 19 and (2) sequences in which up to 3, or up to 2, or up to 1 amino acids of SEQ ID NO: 19 are substituted by other amino acids, wherein said first ankyrin repeat module is located N-terminally of said second ankyrin repeat module within said ankyrin repeat domain.
- said first ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 13 and said second ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 19, wherein said first ankyrin repeat module is located N-terminally of said second ankyrin repeat module within said ankyrin repeat domain.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 17 and (2) sequences in which up to 9, or up to 8, or up to 7, or up to 6, or up to 5, or up to 4, or up to 3, or up to 2, or up to 1 amino acids in SEQ ID NO: 17 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 20 and (2) sequences in which up to 9, or up to 8, or up to 7, or up to 6, or up to 5, or up to 4, or up to 3, or up to 2, or up to 1 amino acids of SEQ ID NO: 20 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 17 and (2) sequences in which up to 9 amino acids in SEQ ID NO: 17 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 20 and (2) sequences in which up to 9 amino acids of SEQ ID NO: 20 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 17 and (2) sequences in which up to 6 amino acids in SEQ ID NO: 17 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 20 and (2) sequences in which up to 6 amino acids of SEQ ID NO: 20 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 17 and (2) sequences in which up to 5 amino acids in SEQ ID NO: 17 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 20 and (2) sequences in which up to 5 amino acids of SEQ ID NO: 20 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO:17 and (2) sequences in which up to 4 amino acids in SEQ ID NO: 17 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 20 and (2) sequences in which up to 4 amino acids of SEQ ID NO: 20 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 17 and (2) sequences in which up to 3 amino acids in SEQ ID NO: 17 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 20 and (2) sequences in which up to 3 amino acids of SEQ ID NO: 20 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 17 and (2) sequences in which up to 2 amino acids in SEQ ID NO: 17 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 20 and (2) sequences in which up to 2 amino acids of SEQ ID NO: 20 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 17 and (2) sequences in which 1 amino acid in SEQ ID NO: 17 is substituted by another amino acid
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 20 and (2) sequences in which 1 amino acid of SEQ ID NO: 20 is substituted by another amino acid.
- said first ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 17 and said second ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 20.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 17 and (2) sequences in which up to 3, or up to 2, or up to 1 amino acids in SEQ ID NO: 17 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 20 and (2) sequences in which up to 3, or up to 2, or up to 1 amino acids of SEQ ID NO: 20 are substituted by other amino acids, wherein said first ankyrin repeat module is located N-terminally of said second ankyrin repeat module within said ankyrin repeat domain.
- said first ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 17 and said second ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 20, wherein said first ankyrin repeat module is located N-terminally of said second ankyrin repeat module within said ankyrin repeat domain.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 17 and (2) sequences in which up to 9, or up to 8, or up to 7, or up to 6, or up to 5, or up to 4, or up to 3, or up to 2, or up to 1 amino acids in SEQ ID NO: 17 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 21 and (2) sequences in which up to 9, or up to 8, or up to 7, or up to 6, or up to 5, or up to 4, or up to 3, or up to 2, or up to 1 amino acids of SEQ ID NO: 21 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 17 and (2) sequences in which up to 2 amino acids in SEQ ID NO: 17 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 21 and (2) sequences in which up to 2 amino acids of SEQ ID NO: 21 are substituted by other amino acids.
- said first ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 17 and (2) sequences in which up to 3, or up to 2, or up to 1 amino acids in SEQ ID NO: 17 are substituted by other amino acids
- said second ankyrin repeat module comprises an amino acid sequence selected from the group consisting of (1) SEQ ID NO: 21 and (2) sequences in which up to 3, or up to 2, or up to 1 amino acids of SEQ ID NO: 21 are substituted by other amino acids, wherein said first ankyrin repeat module is located N-terminally of said second ankyrin repeat module within said ankyrin repeat domain.
- said first ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 17 and said second ankyrin repeat module comprises the amino acid sequence of SEQ ID NO: 21 , wherein said first ankyrin repeat module is located N-terminally of said second ankyrin repeat module within said ankyrin repeat domain.
- all of said amino acid substitutions of said ankyrin repeat module(s) as described and referred to herein occur in framework positions of said ankyrin repeat module(s), wherein typically the overall structure of the module(s) is not affected by the substitutions.
- Such an embodiment of substitution in framework positions shall apply to all embodiments irrespective of whether such substitution is explicitly described.
- all of said amino acid substitutions of said ankyrin repeat module(s) as described and referred to herein occur in framework positions and in positions other than positions 3, 4, 6, 14 and 15, preferably other than positions 2, 3, 4, 5, 6, 11 , 14 and 15, of said ankyrin repeat module(s) of SEQ ID NOs: 9 to 21 , wherein typically the overall structure of the module(s) is not affected by the substitutions.
- the invention in another aspect, relates to a recombinant binding protein comprising an ankyrin repeat domain, wherein said ankyrin repeat domain has binding specificity for CD47, wherein said ankyrin repeat domain comprises an amino acid sequence with at least 80%, 81 %, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% amino acid sequence identity with any one of SEQ ID NOs: 1 to 8, and wherein A at the second last position of SEQ ID NOs: 1 to 8 is optionally substituted by L, and/or A at the last position of SEQ ID NOs: 1 to 8 is optionally substituted by N.
- said ankyrin repeat domain comprises an amino acid sequence with at least 80% amino acid sequence identity with any one of SEQ ID NOs: 1 to 8. In one embodiment, said ankyrin repeat domain comprises an amino acid sequence with at least 90% amino acid sequence identity with any one of SEQ ID NOs: 1 to 8. In another embodiment, said ankyrin repeat domain comprises an amino acid sequence with at least 93% amino acid sequence identity with any one of SEQ ID NOs: 1 to 8; and in a further embodiment, said ankyrin repeat domain comprises an amino acid sequence with at least 95% amino acid sequence identity with any one of SEQ ID NOs: 1 to 8.
- said ankyrin repeat domain comprises an amino acid sequence with at least 98% amino acid sequence identity with any one of SEQ ID NOs: 1 to 8; and in one embodiment, said ankyrin repeat domain comprises the amino acid sequence of any one of SEQ ID NOs: 1 to 8.
- said recombinant binding protein comprises an ankyrin repeat domain with binding specificity for CD47, wherein said ankyrin repeat domain comprises an amino acid sequence selected from SEQ ID NOs: 1 to 8, and wherein A at the second last position of SEQ ID NOs: 1 to 8 is optionally substituted by L, and/or A at the last position of SEQ ID NOs: 1 to 8 is optionally substituted by N.
- said ankyrin repeat domain comprises an amino acid sequence with at least 80%, 81 %, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% amino acid sequence identity with SEQ ID NO: 1 , and wherein A at the second last position of SEQ ID NO: 1 is optionally substituted by L, and/or A at the last position of SEQ ID NO: 1 is optionally substituted by N.
- said ankyrin repeat domain comprises an amino acid sequence with at least 80% amino acid sequence identity with SEQ ID NO: 1 .
- said ankyrin repeat domain comprises an amino acid sequence with at least 90% amino acid sequence identity with SEQ ID NO: 1. In another embodiment, said ankyrin repeat domain comprises an amino acid sequence with at least 93% amino acid sequence identity with SEQ ID NO: 1 ; and in a further embodiment, said ankyrin repeat domain comprises an amino acid sequence with at least 95% amino acid sequence identity with SEQ ID NO: 1. In one embodiment, said ankyrin repeat domain comprises an amino acid sequence with at least 98% amino acid sequence identity with SEQ ID NO: 1 ; and in one embodiment, said ankyrin repeat domain comprises the amino acid sequence of SEQ ID NO: 1 .
- said recombinant binding protein comprises an ankyrin repeat domain having binding specificity for CD47, wherein said ankyrin repeat domain comprises the amino acid sequence of SEQ ID NO: 1.
- said ankyrin repeat domain comprises an amino acid sequence with at least 80%, 81 %, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% amino acid sequence identity with SEQ ID NO: 2, and wherein A at the second last position of SEQ ID NO: 2 is optionally substituted by L, and/or A at the last position of SEQ ID NO: 2 is optionally substituted by N.
- said ankyrin repeat domain comprises an amino acid sequence with at least 80% amino acid sequence identity with SEQ ID NO: 2.
- said ankyrin repeat domain comprises an amino acid sequence with at least 90% amino acid sequence identity with SEQ ID NO: 2. In another embodiment, said ankyrin repeat domain comprises an amino acid sequence with at least 93% amino acid sequence identity with SEQ ID NO: 2; and in a further embodiment, said ankyrin repeat domain comprises an amino acid sequence with at least 95% amino acid sequence identity with SEQ ID NO: 2. In one embodiment, said ankyrin repeat domain comprises an amino acid sequence with at least 98% amino acid sequence identity with SEQ ID NO: 2; and in one embodiment, said ankyrin repeat domain comprises the amino acid sequence of SEQ ID NO: 2.
- said recombinant binding protein comprises an ankyrin repeat domain having binding specificity for CD47, wherein said ankyrin repeat domain comprises the amino acid sequence of SEQ ID NO: 2.
- said ankyrin repeat domain comprises an amino acid sequence with at least 80%, 81 %, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% amino acid sequence identity with SEQ ID NO: 3, and wherein A at the second last position of SEQ ID NO: 3 is optionally substituted by L, and/or A at the last position of SEQ ID NO: 3 is optionally substituted by N.
- said ankyrin repeat domain comprises an amino acid sequence with at least 80% amino acid sequence identity with SEQ ID NO: 3.
- said ankyrin repeat domain comprises an amino acid sequence with at least 90% amino acid sequence identity with SEQ ID NO: 3. In another embodiment, said ankyrin repeat domain comprises an amino acid sequence with at least 93% amino acid sequence identity with SEQ ID NO: 3; and in a further embodiment, said ankyrin repeat domain comprises an amino acid sequence with at least 95% amino acid sequence identity with SEQ ID NO: 3. In one embodiment, said ankyrin repeat domain comprises an amino acid sequence with at least 98% amino acid sequence identity with SEQ ID NO: 3; and in one embodiment, said ankyrin repeat domain comprises the amino acid sequence of SEQ ID NO: 3.
- said recombinant binding protein comprises an ankyrin repeat domain having binding specificity for CD47, wherein said ankyrin repeat domain comprises the amino acid sequence of SEQ ID NO: 3.
- said ankyrin repeat domain comprises an amino acid sequence with at least 80%, 81 %, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% amino acid sequence identity with SEQ ID NO: 4, and wherein A at the second last position of SEQ ID NO: 4 is optionally substituted by L, and/or A at the last position of SEQ ID NO: 4 is optionally substituted by N.
- said ankyrin repeat domain comprises an amino acid sequence with at least 80% amino acid sequence identity with SEQ ID NO: 4.
- said ankyrin repeat domain comprises an amino acid sequence with at least 90% amino acid sequence identity with SEQ ID NO: 4. In another embodiment, said ankyrin repeat domain comprises an amino acid sequence with at least 93% amino acid sequence identity with SEQ ID NO: 4; and in a further embodiment, said ankyrin repeat domain comprises an amino acid sequence with at least 95% amino acid sequence identity with SEQ ID NO: 4. In one embodiment, said ankyrin repeat domain comprises an amino acid sequence with at least 98% amino acid sequence identity with SEQ ID NO: 4; and in one embodiment, said ankyrin repeat domain comprises the amino acid sequence of SEQ ID NO: 4.
- said recombinant binding protein comprises an ankyrin repeat domain having binding specificity for CD47, wherein said ankyrin repeat domain comprises the amino acid sequence of SEQ ID NO: 4.
- said ankyrin repeat domain comprises an amino acid sequence with at least 80%, 81 %, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% amino acid sequence identity with SEQ ID NO: 5, and wherein A at the second last position of SEQ ID NO: 5 is optionally substituted by L, and/or A at the last position of SEQ ID NO: 5 is optionally substituted by N.
- said ankyrin repeat domain comprises an amino acid sequence with at least 80% amino acid sequence identity with SEQ ID NO: 5.
- said ankyrin repeat domain comprises an amino acid sequence with at least 90% amino acid sequence identity with SEQ ID NO: 5. In another embodiment, said ankyrin repeat domain comprises an amino acid sequence with at least 93% amino acid sequence identity with SEQ ID NO: 5; and in a further embodiment, said ankyrin repeat domain comprises an amino acid sequence with at least 95% amino acid sequence identity with SEQ ID NO: 5. In one embodiment, said ankyrin repeat domain comprises an amino acid sequence with at least 98% amino acid sequence identity with SEQ ID NO: 5; and in one embodiment, said ankyrin repeat domain comprises the amino acid sequence of SEQ ID NO: 5.
- said recombinant binding protein comprises an ankyrin repeat domain having binding specificity for CD47, wherein said ankyrin repeat domain comprises the amino acid sequence of SEQ ID NO: 5.
- said ankyrin repeat domain comprises an amino acid sequence with at least 80%, 81 %, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% amino acid sequence identity with SEQ ID NO: 6, and wherein A at the second last position of SEQ ID NO: 6 is optionally substituted by L, and/or A at the last position of SEQ ID NO: 6 is optionally substituted by N.
- said ankyrin repeat domain comprises an amino acid sequence with at least 80% amino acid sequence identity with SEQ ID NO: 6.
- said ankyrin repeat domain comprises an amino acid sequence with at least 90% amino acid sequence identity with SEQ ID NO: 6. In another embodiment, said ankyrin repeat domain comprises an amino acid sequence with at least 93% amino acid sequence identity with SEQ ID NO: 6; and in a further embodiment, said ankyrin repeat domain comprises an amino acid sequence with at least 95% amino acid sequence identity with SEQ ID NO: 6. In one embodiment, said ankyrin repeat domain comprises an amino acid sequence with at least 98% amino acid sequence identity with SEQ ID NO: 6; and in one embodiment, said ankyrin repeat domain comprises the amino acid sequence of SEQ ID NO: 6.
- said recombinant binding protein comprises an ankyrin repeat domain having binding specificity for CD47, wherein said ankyrin repeat domain comprises the amino acid sequence of SEQ ID NO: 6.
- said ankyrin repeat domain comprises an amino acid sequence with at least 80%, 81 %, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% amino acid sequence identity with SEQ ID NO: 7, and wherein A at the second last position of SEQ ID NO: 7 is optionally substituted by L, and/or A at the last position of SEQ ID NO: 7 is optionally substituted by N.
- said ankyrin repeat domain comprises an amino acid sequence with at least 80% amino acid sequence identity with SEQ ID NO: 7.
- said ankyrin repeat domain comprises an amino acid sequence with at least 90% amino acid sequence identity with SEQ ID NO: 7. In another embodiment, said ankyrin repeat domain comprises an amino acid sequence with at least 93% amino acid sequence identity with SEQ ID NO: 7; and in a further embodiment, said ankyrin repeat domain comprises an amino acid sequence with at least 95% amino acid sequence identity with SEQ ID NO: 7. In one embodiment, said ankyrin repeat domain comprises an amino acid sequence with at least 98% amino acid sequence identity with SEQ ID NO: 7; and in one embodiment, said ankyrin repeat domain comprises the amino acid sequence of SEQ ID NO: 7.
- said recombinant binding protein comprises an ankyrin repeat domain having binding specificity for CD47, wherein said ankyrin repeat domain comprises the amino acid sequence of SEQ ID NO: 7.
- said ankyrin repeat domain comprises an amino acid sequence with at least 80%, 81 %, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% amino acid sequence identity with SEQ ID NO: 8, and wherein A at the second last position of SEQ ID NO: 8 is optionally substituted by L, and/or A at the last position of SEQ ID NO: 8 is optionally substituted by N.
- said ankyrin repeat domain comprises an amino acid sequence with at least 80% amino acid sequence identity with SEQ ID NO: 8.
- said ankyrin repeat domain comprises an amino acid sequence with at least 90% amino acid sequence identity with SEQ ID NO: 8. In another embodiment, said ankyrin repeat domain comprises an amino acid sequence with at least 93% amino acid sequence identity with SEQ ID NO: 8; and in a further embodiment, said ankyrin repeat domain comprises an amino acid sequence with at least 95% amino acid sequence identity with SEQ ID NO: 8. In one embodiment, said ankyrin repeat domain comprises an amino acid sequence with at least 98% amino acid sequence identity with SEQ ID NO: 8; and in one embodiment, said ankyrin repeat domain comprises the amino acid sequence of SEQ ID NO: 8.
- said recombinant binding protein comprises an ankyrin repeat domain having binding specificity for CD47, wherein said ankyrin repeat domain comprises the amino acid sequence of SEQ ID NO: 8.
- said recombinant binding protein comprises an ankyrin repeat domain with binding specificity for CD47, wherein the potential interaction residues in said ankyrin repeat domain are identical to the corresponding positions in any one of the ankyrin repeat domains of SEQ ID NOs: 1 to 8.
- the invention relates to a recombinant binding protein comprising an ankyrin repeat domain with binding specificity for CD47, wherein said recombinant binding protein binds to human CD47 in PBS with a dissociation constant (KD) of or below about 10 -7 M, or of or below about 10 -8 M, or of or below about 10" 9 M.
- said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) of or below about 10 -7 M.
- said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) of or below about 10 -8 M.
- said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) of or below about 10 -9 M.
- said recombinant binding protein comprises an ankyrin repeat domain with binding specificity for CD47, wherein said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) of or below 10 -7 M, and wherein said ankyrin repeat domain comprises an amino acid sequence with at least 80%, 81 %, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, 100% amino acid sequence identity with any one of SEQ ID NOs: 1 to 8, wherein A at the second last position of SEQ ID NOs: 1 to 8 is optionally substituted by L, and/or A at the last position of SEQ ID NOs: 1 to 8 is optionally substituted by N.
- KD dissociation constant
- said recombinant binding protein comprises an ankyrin repeat domain with binding specificity for CD47, wherein said binding protein binds human CD47 in PBS with a dissociation constant (KD) of or below 10 -8 M, and wherein said ankyrin repeat domain comprises an amino acid sequence with at least 80%, 81 %, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, 100% amino acid sequence identity with any one of SEQ ID NOs: 1 to 8, wherein A at the second last position of SEQ ID NOs: 1 to 8 is optionally substituted by L, and/or A at the last position of SEQ ID NOs: 1 to 8 is optionally substituted by N.
- KD dissociation constant
- said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) below 10 -7 M
- said ankyrin repeat domain comprises an amino acid sequence with at least 80% amino acid sequence identity with any one of SEQ ID NOs: 1 to 8.
- said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) below 10 -7 M
- said ankyrin repeat domain comprises an amino acid sequence with at least 90% amino acid sequence identity with any one of SEQ ID NOs: 1 to 8.
- said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) below 10 -7 M, and said ankyrin repeat domain comprises an amino acid sequence with at least 93% amino acid sequence identity with any one of SEQ ID NOs: 1 to 8; and in a further embodiment, said binding protein binds human CD47 in PBS with a dissociation constant (KD) below 10 -7 M, and said ankyrin repeat domain comprises an amino acid sequence with at least 95% amino acid sequence identity with any one of SEQ ID NOs: 1 to 8.
- said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) below 10 -7 M, and said ankyrin repeat domain comprises an amino acid sequence with at least 98% amino acid sequence identity with any one of SEQ ID NOs: 1 to 8; and in one embodiment, said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) below 10 -7 M, and said ankyrin repeat domain comprises the amino acid sequence of any one of SEQ ID NOs: 1 to 8.
- said recombinant binding protein comprises an ankyrin repeat domain with binding specificity for CD47, wherein said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) of or below about 10 -7 M, and wherein said ankyrin repeat domain comprises the amino acid sequence of any one of SEQ ID NOs: 1 to 8, wherein A at the second last position of SEQ ID NOs: 1 to 8 is optionally substituted by L, and/or A at the last position of SEQ ID NOs: 1 to 8 is optionally substituted by N.
- KD dissociation constant
- said recombinant binding protein comprises an ankyrin repeat domain having binding specificity for CD47, wherein said binding protein binds human CD47 in PBS with a dissociation constant (KD) of or below about 10 -8 M, and wherein said ankyrin repeat domain comprises an amino acid sequence with at least 80%, 81 %, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, 100% amino acid sequence identity with SEQ ID NO: 1 , and wherein A at the second last position of SEQ ID NO: 1 is optionally substituted by L, and/or A at the last position of SEQ ID NO: 1 is optionally substituted by N.
- KD dissociation constant
- said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) of or below about 10 -8 M, and said ankyrin repeat domain comprises an amino acid sequence with at least 80% amino acid sequence identity with SEQ ID NO: 1.
- said binding protein binds human CD47 in PBS with a dissociation constant (KD) below 10 _ 8 M, and said ankyrin repeat domain comprises an amino acid sequence with at least 90% amino acid sequence identity with SEQ ID NO: 1.
- said recombinant binding protein comprises an ankyrin repeat domain with binding specificity for CD47, wherein said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) of or below about 10 -8 M, and wherein said ankyrin repeat domain comprises the amino acid sequence of SEQ ID NO: 1 , and wherein A at the second last position of SEQ ID NO: 1 is optionally substituted by L, and/or A at the last position of SEQ ID NO: 1 is optionally substituted by N.
- KD dissociation constant
- said recombinant binding protein comprises an ankyrin repeat domain having binding specificity for CD47, wherein said binding protein binds human CD47 in PBS with a dissociation constant (KD) of or below about 10 -8 M, and wherein said ankyrin repeat domain comprises an amino acid sequence with at least 80%, 81 %, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, 100% amino acid sequence identity with SEQ ID NO: 2, and wherein A at the second last position of SEQ ID NO: 2 is optionally substituted by L, and/or A at the last position of SEQ ID NO: 2 is optionally substituted by N.
- KD dissociation constant
- said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) below 10 -8 M, and said ankyrin repeat domain comprises an amino acid sequence with at least 93% amino acid sequence identity with SEQ ID NO: 2; and in a further embodiment, said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) below 10 -8 M, and said ankyrin repeat domain comprises an amino acid sequence with at least 95% amino acid sequence identity with SEQ ID NO: 2.
- said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) below 10 -8 M, and said ankyrin repeat domain comprises an amino acid sequence with at least 98% amino acid sequence identity with SEQ ID NO: 2; and in one embodiment, said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) below 10 -8 M, and said ankyrin repeat domain comprises the amino acid sequence of SEQ ID NO: 2.
- said recombinant binding protein comprises an ankyrin repeat domain with binding specificity for CD47, wherein said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) of or below about 10 -8 M, and wherein said ankyrin repeat domain comprises the amino acid sequence of SEQ ID NO: 2, and wherein A at the second last position of SEQ ID NO: 2 is optionally substituted by L, and/or A at the last position of SEQ ID NO: 2 is optionally substituted by N.
- KD dissociation constant
- said recombinant binding protein comprises an ankyrin repeat domain having binding specificity for CD47, wherein said binding protein binds human CD47 in PBS with a dissociation constant (KD) of or below about 10 -8 M, and wherein said ankyrin repeat domain comprises an amino acid sequence with at least 80%, 81 %, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, 100% amino acid sequence identity with SEQ ID NO: 3, and wherein A at the second last position of SEQ ID NO: 3 is optionally substituted by L, and/or A at the last position of SEQ ID NO: 3 is optionally substituted by N.
- KD dissociation constant
- said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) of or below about 10 -8 M, and said ankyrin repeat domain comprises an amino acid sequence with at least 80% amino acid sequence identity with SEQ ID NO: 3.
- said binding protein binds human CD47 in PBS with a dissociation constant (KD) below 10 _ 8 M, and said ankyrin repeat domain comprises an amino acid sequence with at least 90% amino acid sequence identity with SEQ ID NO: 3.
- said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) below 10 -8 M, and said ankyrin repeat domain comprises an amino acid sequence with at least 93% amino acid sequence identity with SEQ ID NO: 3; and in a further embodiment, said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) below 10 -8 M, and said ankyrin repeat domain comprises an amino acid sequence with at least 95% amino acid sequence identity with SEQ ID NO: 3.
- said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) below 10 -8 M, and said ankyrin repeat domain comprises an amino acid sequence with at least 98% amino acid sequence identity with SEQ ID NO: 3; and in one embodiment, said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) below 10 -8 M, and said ankyrin repeat domain comprises the amino acid sequence of SEQ ID NO: 3.
- said recombinant binding protein comprises an ankyrin repeat domain with binding specificity for CD47, wherein said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) of or below about 10 -8 M, and wherein said ankyrin repeat domain comprises the amino acid sequence of SEQ ID NO: 3, and wherein A at the second last position of SEQ ID NO: 3 is optionally substituted by L, and/or A at the last position of SEQ ID NO: 3 is optionally substituted by N.
- KD dissociation constant
- said recombinant binding protein comprises an ankyrin repeat domain having binding specificity for CD47, wherein said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) of or below about 10 -8 M, and wherein said ankyrin repeat domain comprises an amino acid sequence with at least 80%, 81 %, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, 100% amino acid sequence identity with SEQ ID NO: 4, and wherein A at the second last position of SEQ ID NO: 4 is optionally substituted by L, and/or A at the last position of SEQ ID NO: 4 is optionally substituted by N.
- KD dissociation constant
- said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) of or below about 10 -8 M, and said ankyrin repeat domain comprises an amino acid sequence with at least 80% amino acid sequence identity with SEQ ID NO: 4.
- said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) below 10 -8 M, and said ankyrin repeat domain comprises an amino acid sequence with at least 90% amino acid sequence identity with SEQ ID NO: 4.
- said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) below 10 -8 M, and said ankyrin repeat domain comprises an amino acid sequence with at least 93% amino acid sequence identity with SEQ ID NO: 4; and in a further embodiment, said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) below 10 -8 M, and said ankyrin repeat domain comprises an amino acid sequence with at least 95% amino acid sequence identity with SEQ ID NO: 4.
- said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) below 10 -8 M, and said ankyrin repeat domain comprises an amino acid sequence with at least 98% amino acid sequence identity with SEQ ID NO: 4; and in one embodiment, said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) below 10 -8 M, and said ankyrin repeat domain comprises the amino acid sequence of SEQ ID NO: 4.
- said recombinant binding protein comprises an ankyrin repeat domain with binding specificity for CD47, wherein said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) of or below about 10 -8 M, and wherein said ankyrin repeat domain comprises the amino acid sequence of SEQ ID NO: 4, and wherein A at the second last position of SEQ ID NO: 4 is optionally substituted by L, and/or A at the last position of SEQ ID NO: 4 is optionally substituted by N.
- KD dissociation constant
- said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) of or below about 10 -8 M, and said ankyrin repeat domain comprises an amino acid sequence with at least 80% amino acid sequence identity with SEQ ID NO: 5.
- said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) below 10 -8 M, and said ankyrin repeat domain comprises an amino acid sequence with at least 90% amino acid sequence identity with SEQ ID NO: 5.
- said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) below 10 -8 M, and said ankyrin repeat domain comprises an amino acid sequence with at least 93% amino acid sequence identity with SEQ ID NO: 5; and in a further embodiment, said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) below 10 -8 M, and said ankyrin repeat domain comprises an amino acid sequence with at least 95% amino acid sequence identity with SEQ ID NO: 5.
- said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) below 10 -8 M, and said ankyrin repeat domain comprises an amino acid sequence with at least 98% amino acid sequence identity with SEQ ID NO: 5; and in one embodiment, said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) below 10 -8 M, and said ankyrin repeat domain comprises the amino acid sequence of SEQ ID NO: 5.
- said recombinant binding protein comprises an ankyrin repeat domain with binding specificity for CD47, wherein said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) of or below about 10 -8 M, and wherein said ankyrin repeat domain comprises the amino acid sequence of SEQ ID NO: 5, and wherein A at the second last position of SEQ ID NO: 5 is optionally substituted by L, and/or A at the last position of SEQ ID NO: 5 is optionally substituted by N.
- KD dissociation constant
- said recombinant binding protein comprises an ankyrin repeat domain having binding specificity for CD47, wherein said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) of or below about 10 -8 M, and wherein said ankyrin repeat domain comprises an amino acid sequence with at least 80%, 81 %, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, 100% amino acid sequence identity with SEQ ID NO: 6, and wherein A at the second last position of SEQ ID NO: 6 is optionally substituted by L, and/or A at the last position of SEQ ID NO: 6 is optionally substituted by N.
- KD dissociation constant
- said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) of or below about 10 -8 M, and said ankyrin repeat domain comprises an amino acid sequence with at least 80% amino acid sequence identity with SEQ ID NO: 6.
- said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) below 10 -8 M, and said ankyrin repeat domain comprises an amino acid sequence with at least 90% amino acid sequence identity with SEQ ID NO: 6.
- said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) below 10 -8 M, and said ankyrin repeat domain comprises an amino acid sequence with at least 93% amino acid sequence identity with SEQ ID NO: 6; and in a further embodiment, said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) below 10 -8 M, and said ankyrin repeat domain comprises an amino acid sequence with at least 95% amino acid sequence identity with SEQ ID NO: 6.
- said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) below 10 -8 M, and said ankyrin repeat domain comprises an amino acid sequence with at least 98% amino acid sequence identity with SEQ ID NO: 6; and in one embodiment, said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) below 10 -8 M, and said ankyrin repeat domain comprises the amino acid sequence of SEQ ID NO: 6.
- said recombinant binding protein comprises an ankyrin repeat domain with binding specificity for CD47, wherein said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) of or below about 10 -8 M, and wherein said ankyrin repeat domain comprises the amino acid sequence of SEQ ID NO: 6, and wherein A at the second last position of SEQ ID NO: 6 is optionally substituted by L, and/or A at the last position of SEQ ID NO: 6 is optionally substituted by N.
- KD dissociation constant
- said recombinant binding protein comprises an ankyrin repeat domain having binding specificity for CD47, wherein said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) of or below about 10 -8 M, and wherein said ankyrin repeat domain comprises an amino acid sequence with at least 80%, 81 %, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, 100% amino acid sequence identity with SEQ ID NO: 7, and wherein A at the second last position of SEQ ID NO: 7 is optionally substituted by L, and/or A at the last position of SEQ ID NO: 7 is optionally substituted by N.
- KD dissociation constant
- said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) of or below about 10 -8 M, and said ankyrin repeat domain comprises an amino acid sequence with at least 80% amino acid sequence identity with SEQ ID NO: 7.
- said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) below 10 -8 M, and said ankyrin repeat domain comprises an amino acid sequence with at least 90% amino acid sequence identity with SEQ ID NO: 7.
- said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) below 10 -8 M, and said ankyrin repeat domain comprises an amino acid sequence with at least 93% amino acid sequence identity with SEQ ID NO: 7; and in a further embodiment, said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) below 10 -8 M, and said ankyrin repeat domain comprises an amino acid sequence with at least 95% amino acid sequence identity with SEQ ID NO: 7.
- said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) below 10 -8 M, and said ankyrin repeat domain comprises an amino acid sequence with at least 98% amino acid sequence identity with SEQ ID NO: 7; and in one embodiment, said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) below 10 -8 M, and said ankyrin repeat domain comprises the amino acid sequence of SEQ ID NO: 7.
- said recombinant binding protein comprises an ankyrin repeat domain with binding specificity for CD47, wherein said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) of or below about 10 -8 M, and wherein said ankyrin repeat domain comprises the amino acid sequence of SEQ ID NO: 7, and wherein A at the second last position of SEQ ID NO: 7 is optionally substituted by L, and/or A at the last position of SEQ ID NO: 7 is optionally substituted by N.
- KD dissociation constant
- said recombinant binding protein comprises an ankyrin repeat domain having binding specificity for CD47, wherein said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) of or below about 10 -8 M, and wherein said ankyrin repeat domain comprises an amino acid sequence with at least 80%, 81 %, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, 100% amino acid sequence identity with SEQ ID NO: 8, and wherein A at the second last position of SEQ ID NO: 8 is optionally substituted by L, and/or A at the last position of SEQ ID NO: 8 is optionally substituted by N.
- KD dissociation constant
- said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) of or below about 10 -8 M, and said ankyrin repeat domain comprises an amino acid sequence with at least 80% amino acid sequence identity with SEQ ID NO: 8.
- said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) below 10 -8 M, and said ankyrin repeat domain comprises an amino acid sequence with at least 90% amino acid sequence identity with SEQ ID NO: 8.
- said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) below 10 -8 M, and said ankyrin repeat domain comprises an amino acid sequence with at least 93% amino acid sequence identity with SEQ ID NO: 8; and in a further embodiment, said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) below 10 -8 M, and said ankyrin repeat domain comprises an amino acid sequence with at least 95% amino acid sequence identity with SEQ ID NO: 8.
- said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) below 10 -8 M, and said ankyrin repeat domain comprises an amino acid sequence with at least 98% amino acid sequence identity with SEQ ID NO: 8; and in one embodiment, said recombinant binding protein binds human CD47 in PBS with a dissociation constant (KD) below 10 -8 M, and said ankyrin repeat domain comprises the amino acid sequence of SEQ ID NO: 8.
- said ankyrin repeat domain has a melting temperature (T m ) of between 72°C and 88°C.
- said recombinant binding protein comprises an ankyrin repeat domain with binding specificity for CD47, wherein said ankyrin repeat domain comprises an amino acid sequence with at least 80%, 81 %, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% amino acid sequence identity with any one of SEQ ID NOs: 1 to 8, wherein A at the second last position of SEQ ID NOs: 1 to 8 is optionally substituted by L, and/or A at the last position of SEQ ID NOs: 1 to 8 is optionally substituted by N; and wherein said ankyrin repeat domain has a melting temperature (T m ) higherthan 65°C.
- said recombinant binding protein comprises an ankyrin repeat domain with binding specificity for CD47, wherein said ankyrin repeat domain comprises an amino acid sequence with at least 80%, 81 %, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% amino acid sequence identity with SEQ ID NO: 6, wherein A at the second last position of SEQ ID NO: 6 is optionally substituted by L, and/or A at the last position of SEQ ID NO: 6 is optionally substituted by N; and wherein said ankyrin repeat domain has a melting temperature (T m ) higherthan 75°C.
- T m melting temperature
- said melting temperature (T m ) of the recombinant binding proteins comprising an ankyrin repeat domains is determined in TBS pH 8.0 (50 mM Tris, 500 mM NaCI) by Circular Dichroism (CD) spectroscopy as described in Example 2.
- the N-and/or C-terminal capping modules may be derived from a capping unit or other structural unit found in a naturally occurring repeat protein adjacent to a repeat unit. Examples of capping sequences are described in International Patent Publication Nos. WO 2002/020565 and WO 2012/069655, in U.S. Patent Publication No. US20130296221 , and by Interlandi et al., J Mol Biol. 2008 Jan 18;375(3):837-54. Examples of N-terminal capping modules (i.e. N- terminal capping repeats) are provided with SEQ ID NOs: 22 to 36 and examples of C-terminal capping modules (i.e. C-terminal capping repeats) are provided with SEQ ID NOs: 37 to 60.
- the N-terminal capping module comprises the amino acid sequence of any one of SEQ ID NOs: 22 to 36, wherein up to 9, up to 8, up to 7, up to 6, up to 5, up to 4, up to 3, up to 2 or up to 1 amino acid(s) of any one of SEQ ID NOs: 22 to 36 are optionally exchanged by any amino acids.
- certain amino acid residues in the N-terminal capping module and/or the C-terminal capping module of the designed ankyrin repeat domain herein provided are altered, resulting in improved pharmacokinetic properties, including a prolonged terminal half-life, of the designed ankyrin repeat domain and of the recombinant binding proteins comprising the designed ankyrin repeat domain.
- the altered amino acid residues are mostly surface exposed residues.
- the ankyrin repeat domain with binding specificity for CD47 comprises the amino acid sequence of any one of SEQ ID NOs: 1 to 8, wherein the amino acid E at position 8 of any one of SEQ ID NOs: 1 to 8 is substituted with Q and/or the amino acid D at position 15 of any one of SEQ ID NOs: 1 to 8 is substituted with L.
- the ankyrin repeat domain with binding specificity for CD47 comprises the amino acid sequence of any one of SEQ ID NOs: 1 to 8, wherein the amino acid K at position 110 of any one of SEQ ID NOs: 1 to 8 is substituted with R and/or the amino acid E at position 114 of SEQ ID NOs: 1 and 8 is substituted with Q.
- the substitution is a conservative substitution according to Table 1.
- the substitution is made outside the structural core residues of the ankyrin repeat domain, e.g., in the beta loops that connect the alpha-helices.
- the CD47-binding domain of the invention may optionally further comprise a “G,” an “S,” or a “GS” sequence at its N-terminus.
- the CD47-binding domain provided herein comprises an amino acid sequence that is at least 80%, at least 81 %, at least 82%, at least 83%, at least 84%, at least 85%, at least 86%, at least 87%, at least 88%, at least 89%, at least 90%, at least 91 %, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical to any one of SEQ ID NOs: 1 to 8, and (ii) further comprises at its N-terminus a G, an S, or a GS.
- the CD47-binding domain comprises an amino acid sequence that is at least 90% identical to any one of SEQ ID NOs: 1 to 8, and further comprises at its N- terminus a G, an S, or a GS. In an exemplary embodiment, the CD47-binding domain comprises an amino acid sequence that is at least 95% identical to any one of SEQ ID NOs: 1 to 8, and further comprises at its N-terminus a G, an S, or a GS. In an exemplary embodiment, the CD47-binding domain comprises the amino acid sequence of any one of SEQ ID NOs: 1 to 8, and further comprises at its N-terminus a G, an S, or a GS.
- said designed ankyrin repeat domain comprises an amino acid sequence with at least 80%, 81 %, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% amino acid sequence identity with SEQ ID NO: 81 .
- a binding agent may be an antibody, an antibody mimetic, including a scaffold protein or a repeat protein, a designed repeat domain, preferably a designed ankyrin repeat domain, or any other suitable binding molecules known in the art.
- said binding agent is an antibody.
- said binding agent is a designed repeat domain, preferably a designed ankyrin repeat domain.
- said binding agent is linked, conjugated, fused or otherwise physically attached to said CD47-specific ankyrin repeat domain. In one embodiment, said binding agent is covalently linked to said CD47-specific ankyrin repeat domain. In one embodiment, said binding agent is covalently linked to said CD47-specific ankyrin repeat domain with a peptide linker. In one embodiment, said peptide linker is a proline-threonine rich peptide linker. In one embodiment, said peptide linker is the proline-threonine rich peptide linker of SEQ ID NO: 65.
- said binding agent is covalently linked to said CD47- specific ankyrin repeat domain with the proline-threonine rich peptide linker of SEQ ID NO: 65.
- said peptide linker is a glycine-serine rich peptide linker.
- said peptide linker is the glycine-serine rich peptide linker of SEQ ID NO: 66.
- said binding agent is covalently linked to said CD47-specific ankyrin repeat domain with the glycine-serine rich peptide linker of SEQ ID NO: 66.
- the recombinant binding protein of the invention further comprises a binding agent with binding specificity for a tumor-associated antigen.
- the recombinant binding protein of the invention comprises an ankyrin repeat domain with binding specificity for CD47 as described more specifically in any of the aspects or embodiments herein and further comprises an additional binding agent with binding specificity for a tumor- associated antigen.
- said binding agent with binding specificity for a tumor-associated antigen is linked, conjugated, fused or otherwise physically attached to said ankyrin repeat domain with binding specificity for CD47. In one embodiment, said binding agent with binding specificity for a tumor-associated antigen is covalently linked to said ankyrin repeat domain with binding specificity for CD47. In one embodiment, said binding agent with binding specificity for a tumor-associated antigen is covalently linked to said ankyrin repeat domain with binding specificity for CD47 with a peptide linker. In one embodiment, the amino acid sequence of said peptide linker has a length from 1 to 50 amino acids, preferably from 6 to 38 amino acids.
- said peptide linker is a proline-threonine rich peptide linker or glycine-serine rich linker. In one embodiment, said peptide linker is the proline-threonine rich peptide linker of SEQ ID NO:65 or the glycine-serine rich linker of SEQ ID NO: 66. In one embodiment, said binding agent with binding specificity for a tumor-associated antigen is covalently linked to said ankyrin repeat domain with binding specificity for CD47 with the proline-threonine rich peptide linker of SEQ ID NO: 65 or the glycine-serine rich linker of SEQ ID NO: 66. In one preferred embodiment, said binding agent with binding specificity for a tumor- associated antigen is a designed ankyrin repeat domain with binding specificity for a disease-associated antigen.
- the recombinant binding protein of the invention comprises an ankyrin repeat domain with binding specificity for CD47, an ankyrin repeat domain with binding specificity for one or more tumor associated antigen(s) and/or a binding agent with binding specificity for a protein expressed on the surface of an immune cell, preferably an NK cell or a macrophage.
- said recombinant binding protein comprises an ankyrin repeat domain with binding specificity for CD47 as described herein and further comprises an ankyrin repeat domain with dual binding specificity, wherein binding of said ankyrin repeat domain with dual binding specificity to its first and second binding targets is mutually exclusive.
- ankyrin repeat domains with dual binding specificity also referred to as “2-in-1 repeat domains”
- production methods are described in PCT/EP2022/085794.
- said recombinant binding protein comprises an ankyrin repeat domain with binding specificity for CD47 as described herein and further comprises a second ankyrin repeat domain, wherein the second ankyrin repeat domain is a 2-in-1 repeat domain having a first binding specificity for a TAA and a second binding specificity for said first ankyrin repeat domain having binding specificity for CD47.
- the 2-in-1 repeat domain binds the TAA and releases the CD47-specific binding domain from binding to the 2-in-1 repeat domain, thereby releasing the inhibition of the CD47-specific binding domain.
- the recombinant binding protein further comprises a half-life extending moiety.
- the recombinant binding protein of the invention comprises an ankyrin repeat domain with binding specificity for CD47 as described more specifically in any of the aspects or embodiments herein and further comprises a half-life extending moiety.
- the recombinant binding protein of the invention comprises an ankyrin repeat domain with binding specificity for CD47, an ankyrin repeat domain with binding specificity for one or more tumor associated antigen(s) and/or a binding agent with binding specificity for a protein expressed on the surface of an immune cell, preferably an NK cell or a macrophage, and further comprises a half-life extending moiety.
- said binding agent with binding specificity for said TAA and/or said protein expressed on the surface of an immune cell, preferably an NK cell or a macrophage, more preferably said protein being CD16a comprises or is an ankyrin repeat domain.
- said designed ankyrin repeat domain with binding specificity for CD16a comprises an amino acid sequence with at least 80%, 81 %, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% amino acid sequence identity with SEQ ID NO: 81 .
- said half-life extending moiety comprises a binding agent with binding specificity for human serum albumin.
- said half-life extending moiety is a designed ankyrin repeat domain with binding specificity for human serum albumin.
- said designed ankyrin repeat domain with binding specificity for human serum albumin comprises an amino acid sequence with at least 80%, 81 %, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% amino acid sequence identity with any one of SEQ ID NOs: 62 to 64.
- the recombinant binding protein of the invention comprises an ankyrin repeat domain with binding specificity for CD47 as described more specifically in any of the aspects or embodiments herein and further comprises an ankyrin repeat domain with binding specificity for human serum albumin having an amino acid sequence with at least 80% amino acid sequence identity with any one of SEQ ID NOs: 62 to 64.
- said ankyrin repeat domain with binding specificity for human serum albumin comprises an amino acid sequence with at least 90% amino acid sequence identity with any one of SEQ ID NOs: 62 to 64.
- said ankyrin repeat domain with binding specificity for human serum albumin comprises an amino acid sequence with at least 93% amino acid sequence identity with any one of SEQ ID NOs: 62 to 64; and in a further embodiment, said ankyrin repeat domain with binding specificity for human serum albumin comprises an amino acid sequence with at least 95% amino acid sequence identity with any one of SEQ ID NOs: 62 to 64.
- said ankyrin repeat domain with binding specificity for human serum albumin comprises an amino acid sequence with at least 98% amino acid sequence identity with any one of SEQ ID NOs: 62 to 64; and in a further embodiment, said ankyrin repeat domain with binding specificity for human serum albumin comprises the amino acid sequence of any one of SEQ ID NOs: 62 to 64.
- the recombinant binding protein of the invention comprises an ankyrin repeat domain with binding specificity for CD47 and further comprises an ankyrin repeat domain with binding specificity for human serum albumin, and wherein said ankyrin repeat domain with binding specificity for CD47 comprises the amino acid sequence of any one of SEQ ID NOs: 1 to 8 and wherein said ankyrin repeat domain with binding specificity for human serum albumin comprises the amino acid sequence of any one of SEQ ID NOs: 62 to 64.
- said ankyrin repeat domain with binding specificity for human serum albumin is located N-terminally of said ankyrin repeat domain with binding specificity for CD47 within said binding protein.
- said designed ankyrin repeat domain with binding specificity for human serum albumin comprises an amino acid sequence with at least 80%, 81 %, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% amino acid sequence identity with SEQ ID NO: 63.
- the recombinant binding protein of the invention comprises an ankyrin repeat domain with binding specificity for CD47 as described more specifically in any of the aspects or embodiments herein and further comprises an ankyrin repeat domain with binding specificity for human serum albumin comprising an amino acid sequence with at least 80% amino acid sequence identity with SEQ ID NO: 63.
- said ankyrin repeat domain with binding specificity for human serum albumin comprises an amino acid sequence with at least 90% amino acid sequence identity with SEQ ID NO: 63.
- said ankyrin repeat domain with binding specificity for human serum albumin comprises an amino acid sequence with at least 93% amino acid sequence identity with SEQ ID NO: 63; and in a further embodiment, said ankyrin repeat domain with binding specificity for human serum albumin comprises an amino acid sequence with at least 95% amino acid sequence identity with SEQ ID NO: 63. In one embodiment, said ankyrin repeat domain with binding specificity for human serum albumin comprises an amino acid sequence with at least 98% amino acid sequence identity with SEQ ID NO: 63; and in a further embodiment, said ankyrin repeat domain with binding specificity for human serum albumin comprises the amino acid sequence of SEQ ID NO: 63.
- the recombinant binding protein of the invention comprises an ankyrin repeat domain with binding specificity for CD47 and further comprises an ankyrin repeat domain with binding specificity for human serum albumin, and wherein ankyrin repeat domain with binding specificity for CD47 comprises the amino acid sequence of any one of SEQ ID NOs: 1 to 8, and wherein said ankyrin repeat domain with binding specificity for human serum albumin comprises the amino acid sequences of SEQ ID NO: 63.
- said ankyrin repeat domain with binding specificity for human serum albumin is located N-terminally of said ankyrin repeat domain with binding specificity for CD47.
- a recombinant binding protein of the present invention comprising an ankyrin repeat domain with binding specificity for CD47 and further comprising an ankyrin repeat domain with binding specificity for human serum albumin exhibits an increased terminal half-life, preferably an increased terminal half-life of at least 5%, preferably 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 60%, 70%, 80%, 90%, 100%, 150%, 200%, or 250%, compared to a corresponding recombinant binding protein comprising said ankyrin repeat domain with binding specificity for CD47 but not comprising said ankyrin repeat domain with binding specificity for human serum albumin.
- the recombinant binding protein of the invention comprises an ankyrin repeat domain having binding specificity for CD47 as described more specifically in any of the aspects or embodiments herein and further comprises two ankyrin repeat domains with binding specificity for human serum albumin as described more specifically in any of the aspects or embodiments herein.
- the half-life extending moiety comprises an immunoglobulin domain.
- the immunoglobulin domain comprises an Fc domain.
- the Fc domain is derived from any one of the known heavy chain isotypes: IgG (y), IgM (p), IgD (6), IgE (s), or IgA (a).
- the Fc domain is derived from any one of the known heavy chain isotypes or subtypes: IgGi (y1), lgG2 (y2), lgG3 (y3), lgG4 (y4), IgAi (a1), lgA2 (a2).
- the Fc domain is the Fc domain of human IgGi.
- the Fc domain comprises an uninterrupted native sequence (i.e., wild type sequence) of an Fc domain.
- the immunoglobulin Fc domain comprises a variant Fc domain resulting in altered biological activity. For example, at least one point mutation or deletion may be introduced into the Fc domain so as to reduce or eliminate the effector activity (e.g., International Patent Publication No. WO 2005/063815), and/or to increase the homogeneity during the production of the recombinant binding protein.
- the Fc domain is the Fc domain of human IgGi and comprises one or more of the following effector-null substitutions: L234A, L235A, and G237A (Eu numbering).
- the Fc domain does not comprise the lysine located at the C-terminal position of human lgG1 (i.e., K447 by Eu numbering). The absence of the lysine may increase homogeneity during the production of the recombinant binding protein.
- the Fc domain comprises the lysine located at the C-terminal position (K447, Eu numbering).
- the recombinant binding protein of the invention further comprises a polypeptide tag.
- a polypeptide tag is an amino acid sequence attached to a polypeptide/protein, wherein said amino acid sequence is useful for the purification, detection, and/or targeting of said polypeptide/protein, and/or wherein said amino acid sequence improves the physicochemical behavior of the polypeptide/protein, and/or wherein said amino acid sequence possesses an effector function.
- the individual polypeptide tags of a binding protein may be connected to other parts of the binding protein directly or via a peptide linker. Polypeptide tags are all known in the art.
- polypeptide tags are small polypeptide sequences, for example, His, HA, myc, FLAG, or Strep-tags, or polypeptides such as enzymes (for example alkaline phosphatase), which allow the detection of said polypeptide/protein, or polypeptides which can be used for targeting (such as immunoglobulins or fragments thereof) and/or as effector molecules.
- enzymes for example alkaline phosphatase
- the recombinant binding protein of the invention further comprises a peptide linker.
- a peptide linker is an amino acid sequence, which is able to link, for example, two protein domains, a polypeptide tag and a protein domain, a protein domain and a non-proteinaceous compound or polymer such as polyethylene glycol, a protein domain and a biologically active molecule, a protein domain and a localizer, or two sequence tags.
- Peptide linkers are known to the person skilled in the art. A list of examples is provided in the description of patent application W02002/020565. Particular examples of such linkers are glycine-serine-linkers and proline-threonine-linkers of variable lengths.
- Examples of a glycine-serine- linker are the amino acid sequence GS and the amino acid sequence of SEQ ID NO: 66, and an example of a proline-threonine-linker is the amino acid sequences of SEQ ID NO: 65.
- the invention relates to a nucleic acid encoding the amino acid sequence of an ankyrin repeat domain or a recombinant binding protein of the present invention. In one embodiment, the invention relates to a nucleic acid encoding the amino acid sequence of a recombinant binding protein of the present invention. In one embodiment, the invention relates to a nucleic acid encoding an amino acid sequence selected from the group consisting of SEQ ID NO: 1 to 8. In one embodiment, the invention relates to a nucleic acid encoding the amino acid sequence of SEQ ID NO: 1 . In one embodiment, the invention relates to a nucleic acid encoding the amino acid sequence of SEQ ID NO: 2.
- the invention relates to a nucleic acid encoding the amino acid sequence of SEQ ID NO: 3. In one embodiment, the invention relates to a nucleic acid encoding the amino acid sequence of SEQ ID NO: 4. In one embodiment, the invention relates to a nucleic acid encoding the amino acid sequence of SEQ ID NO: 5. In one embodiment, the invention relates to a nucleic acid encoding the amino acid sequence of SEQ ID NO: 6. In one embodiment, the invention relates to a nucleic acid encoding the amino acid sequence of SEQ ID NO: 7. In one embodiment, the invention relates to a nucleic acid encoding the amino acid sequence of SEQ ID NO: 8. In another embodiment, the invention relates to a nucleic acid sequence encoding the amino acid sequence of a recombinant protein comprising an ankyrin repeat domain comprising any one of the amino acid sequences with SEQ ID NOs: 9-21 .
- nucleic acids are well known to the skilled person in the art. In the examples, nucleic acids were used to produce designed ankyrin repeat domains or recombinant binding proteins of the invention in E. coli. Examples nucleic acids of the invention are provided by SEQ ID NOs: 73 to 80 which encode the amino acid sequences of SEQ ID NOs: 1 to 8, respectively.
- the invention relates to a pharmaceutical composition
- a pharmaceutical composition comprising a recombinant binding protein and/or a designed ankyrin repeat domain of the present invention, and/or a nucleic acid encoding a recombinant binding protein and/or a designed ankyrin repeat domain of the present invention, and optionally a pharmaceutically acceptable carrier and/or diluent.
- the invention relates to a pharmaceutical composition
- a pharmaceutical composition comprising a recombinant binding protein or a nucleic acid encoding a recombinant binding protein of the present invention, and optionally a pharmaceutically acceptable carrier and/or diluent.
- a pharmaceutical composition comprises a recombinant binding protein, and/or a designed ankyrin repeat domain, and/or a nucleic acid, preferably a recombinant binding protein and/or a nucleic acid, as described herein and a pharmaceutically acceptable carrier, excipient or stabilizer, for example as described in Remington's Pharmaceutical Sciences 16 th edition, Osol, A. Ed., 1980.
- Suitable carriers, excipients or stabilizers known to one of skill in the art include, for example, saline, Ringer's solution, dextrose solution, Hank's solution, fixed oils, ethyl oleate, 5% dextrose in saline, substances that enhance isotonicity and chemical stability, buffers and preservatives.
- Other suitable carriers include any carrier that does not itself induce the production of antibodies harmful to the individual receiving the composition such as proteins, polysaccharides, polylactic acids, polyglycolic acids, polymeric amino acids and amino acid copolymers.
- a pharmaceutical composition may also be a combination formulation, comprising an additional active agent, such as an anti-cancer agent or an anti-angiogenic agent, or an additional bioactive compound.
- the formulations to be used for/n vivo administration must be aseptic or sterile. This is readily accomplished by filtration through sterile filtration membranes.
- a recombinant binding protein of the present invention comprising an ankyrin repeat domain with binding specificity for CD47 and further comprising an ankyrin repeat domain with binding specificity for human serum albumin for manufacturing a pharmaceutical composition, wherein said recombinant binding protein exhibits an increased terminal half-life, preferably an increased terminal half-life of at least 5%, preferably 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 60%, 70%, 80%, 90%, 100%, 150%, 200%, or 250%, compared to a corresponding recombinant binding protein comprising said ankyrin repeat domain with binding specificity for CD47 but not comprising said ankyrin repeat domain with binding specificity for serum albumin.
- a recombinant binding protein comprises an ankyrin repeat domain having binding specificity for CD47 and further comprises two ankyrin repeat domains with binding specificity for serum albumin.
- a pharmaceutical composition comprises at least one recombinant binding protein as described herein and a detergent such as nonionic detergent, a buffer such as phosphate buffer, and a sugar such as sucrose.
- a composition comprises recombinant binding proteins as described above and PBS.
- the invention relates to a pharmaceutical composition, a recombinant binding protein, or a nucleic acid according to the present invention for use in the treatment of a disease.
- said disease is cancer.
- the pharmaceutical composition, the nucleic acid or the recombinant binding protein according to the present invention is administered to a subject in need thereof in a therapeutically effective amount. Administration may include topical administration, oral administration, and parenteral administration. The typical route of administration is parenteral administration.
- the pharmaceutical composition of this invention will be formulated in a unit dosage injectable form such as a solution, suspension or emulsion, in association with the pharmaceutically acceptable excipients as defined above. The dosage and mode of administration will depend on the individual to be treated and the particular disease.
- the invention provides a method of diagnosing a medical condition in a mammal, preferably a human, the method comprising the steps of: (i) contacting a cell or tissue sample obtained from said mammal with the CD47-specific recombinant binding protein of the invention; and
- binding protein refers to a protein comprising a binding domain.
- a binding protein may also comprise two, three, four, five or more binding domains.
- said binding protein is a recombinant binding protein.
- Binding proteins of the instant invention comprise an ankyrin repeat domain with binding specificity for CD47.
- repeat domains Such repeat domains, repeat modules, and capping modules, sequence motives, as well as structural homology and sequence homology are well known to the practitioner in the art from examples of ankyrin repeat domains (W02002/020565), leucine-rich repeat domains (W02002/020565), tetratricopeptide repeat domains (Main, E.R., Xiong, Y., Cocco, M.J., D'Andrea, L., Regan, L., Structure 11 (5), 497-508, 2003), and armadillo repeat domains (W02009/040338). It is further known to the practitioner in the art that such repeat domains are different from proteins comprising repeated amino acid sequences, where every repeated amino acid sequence is able to form an individual domain (for example FN3 domains of Fibronectin).
- designed refers to the property that such repeat proteins and repeat domains, respectively, are man-made and do not occur in nature.
- the recombinant binding proteins of the instant invention are designed repeat proteins and they comprise at least one designed ankyrin repeat domain.
- ankyrin repeat module refers to a repeat module, which is originally derived from the repeat units of naturally occurring ankyrin repeat proteins.
- Ankyrin repeat proteins are known to the person skilled in the art.
- amino acid residues 3, 4, 6, 11 , 14 and 15 of SEQ ID NOs: 9 to 21 amino acid residues 4, 8, 11 and 12 of SEQ ID NOs: 22 to 36, and amino acid residues 3, 4, 6, 14 and 15 of SEQ ID NOs: 37 to 60 are randomized positions of the ankyrin repeat modules of the instant invention. Randomized positions are generally not substituted in the variants of the module sequences disclosed herein.
- repeat sequence motif refers to an amino acid sequence, which is deduced from one or more repeat modules.
- said repeat modules are from repeat domains having binding specificity forthe same target.
- Such repeat sequence motifs comprise framework residue positions and target interaction residue positions. Said framework residue positions correspond to the positions of framework residues of the repeat modules. Likewise, said target interaction residue positions correspond to the positions of target interaction residues of the repeat modules.
- Repeat sequence motifs comprise non-randomized positions and randomized positions.
- repeat unit refers to amino acid sequences comprising sequence motifs of one or more naturally occurring proteins, wherein said "repeat units” are found in multiple copies, and exhibit a defined folding topology common to all said motifs determining the fold of the protein.
- repeat units include leucine-rich repeat units, ankyrin repeat units, armadillo repeat units, tetratricopeptide repeat units, HEAT repeat units, and leucine-rich variant repeat units.
- binding specificity “has binding specificity for a target”, “specifically binding to a target”, “binding to a target with high specificity”, “specific for a target” or “target specificity” and the like means that a binding protein or binding domain binds to a target with a lower dissociation constant (i.e. it binds with higher affinity) than it binds to an unrelated protein such as the E. coli maltose binding protein (MBP).
- the dissociation constant (“KD”) for the target is at least 10 2 ; more preferably, at least 10 3 ; more preferably, at least 10 4 ; or more preferably, at least 10 5 times lower than the corresponding dissociation constant for MBP.
- Binding of any molecule to another is governed by two forces, namely the association rate (k on ) and the dissociation rate (k O ff).
- the affinity of any binder [B] to a target [T] can then be expressed by the equilibrium dissociation constant KD, which is the quotient of koir/kon.
- kon is a second-order rate constant of the binding reaction, with the unit whereas the dissociation reaction kotr is a first-order rate constant with the unit s ⁇ 1 . From this it becomes clear that the association reaction depends on the concentration of the reactants, whereas the dissociation is independent of the concentration, following a simple exponential decay function.
- KD value refers to the dissociation constant of the binding moiety and the drug molecule target.
- KD is the ratio of the rate of dissociation, also called the “off-rate (kotr)”, to the association rate, or “on-rate (kon)”.
- LTD off-rate
- kon on-rate
- KD equals kotr/kon and is expressed as a molar concentration (M), and the smaller the KD, the stronger the affinity of binding.
- KD values can be determined using any suitable method.
- KD value may be measured by SPR using a biosensor system such as a BIACORE® system.
- BIAcore kinetic analysis comprises, e.g., analysing the binding and dissociation of an antigen from chips with immobilized molecules (e.g., molecules comprising epitope binding domains), on their surface.
- Another method for determining the KD of a protein is by using Bio-Layer Interferometry (see, e.g., Shah et al. J Vis Exp. 2014; (84): 51383).
- a KD value may be measured using OCTET® technology (Octet QKe system, ForteBio). Alternatively, or in addition, a KinExA® (Kinetic Exclusion Assay) assay, available from Sapidyne Instruments (Boise, Id.) can also be used. Any method suitable for assessing the binding affinity between two binding partners is encompassed herein. Surface plasmon resonance (SPR) is particularly preferred. Most preferably, the KD values are determined in PBS and by SPR.
- binding agent refers to any molecule capable of specifically binding a target molecule. Binding agents include, for example, antibodies, antibody fragments, aptamers, peptides (e.g., Williams et al., J Biol Chem 266:5182-5190 (1991)), antibody mimics, repeat proteins, e.g. designed ankyrin repeat proteins, receptor proteins and any other naturally occurring interaction partners of the target molecule, and can comprise natural proteins and proteins modified or genetically engineered, e.g., to include non-natural residues and/or to lack natural residues.
- binding agents include, for example, antibodies, antibody fragments, aptamers, peptides (e.g., Williams et al., J Biol Chem 266:5182-5190 (1991)), antibody mimics, repeat proteins, e.g. designed ankyrin repeat proteins, receptor proteins and any other naturally occurring interaction partners of the target molecule, and can comprise natural proteins and proteins modified or genetically engineered, e.g., to include non-
- PBS means a phosphate buffered water solution containing 137 mM NaCI, 10 mM phosphate and 2.7 mM KCI and having a pH of 7.4.
- the sequences are aligned for optimal comparison purposes (e.g., gaps can be introduced in a first sequence for optimal alignment with a second sequence).
- the nucleotides or amino acids at each position are then compared.
- a position in the first sequence is occupied by the same amino acid or nucleotide as the corresponding position in the second sequence, then the amino acids or nucleotides are identical at that position.
- the sequence comparison is carried out over the length of the reference sequence. For example, if the user wished to determine whether a given (“test”) sequence is 95% identical to SEQ ID NO: 1 , SEQ ID NO: 1 would be the reference sequence. To assess whether a sequence is at least 80% identical to SEQ ID NO: 1 (an example of a reference sequence), the skilled person would carry out an alignment over the length of SEQ ID NO: 1 , and identify how many positions in the test sequence were identical to those of SEQ ID NO: 1. If at least 80% of the positions are identical, the test sequence is at least 80% identical to SEQ ID NO: 1. If the sequence is shorter than SEQ ID NO: 1 , the gaps or missing positions should be considered to be non-identical positions.
- a comparison of sequences and determination of percent identity between two sequences can be accomplished using a mathematical algorithm.
- the percent identity between two amino acid or nucleic acid sequences can be determined using the Needleman and Wunsch (1970) algorithm which has been incorporated into the GAP program in the Accelrys GCG software package (available at http://www.accelrys.com/products/gcg/), using either a Blosum 62 matrix or a PAM250 matrix, and a gap weight of 16, 14, 12, 10, 8, 6, or 4 and a length weight of 1 , 2, 3, 4, 5, or 6.
- the term “mouse serum albumin” refers to UniProt accession number P07724, the term “cynomolgus monkey serum albumin” (i.e. macaca fascicularis) refers to UniProt accession number A2V9Z4, and the term “human serum albumin” refers to UniProt accession number P02768.
- clearance, and/or exposure, and/or terminal half-life are assessed in a mammal, more preferably mouse and/or cynomolgus monkey, more preferably cynomolgus monkey. Clearance, and/or exposure, and/or terminal half-life may be assessed as described in Example 3.
- the evaluation is done considering the data up to 48 h post-injection. More preferably, the evaluation of terminal half-life in mouse is calculated from 24 h to 48 h.
- the evaluation is done considering the data up to day 7 post-injection. More preferably, the evaluation of terminal half-life in cynomolgus monkey is calculated from day 1 to day 5. The person skilled in the art further is able to identify effects such as target-mediated clearance and consider them when calculating the terminal half-life.
- terminal half-life of a drug such as a recombinant binding protein of the invention refers to the time required to reach half the plasma concentration of the drug applied to a mammal after reaching pseudo-equilibrium (for example calculated from 24 hours to 48 hours in mouse or calculated from day 1 to day 5 in cynomolgus monkey). Terminal half-life is not defined as the time required to eliminate half the dose of the drug administered to the mammal.
- the term terminal half-life is known to the person skilled in the art.
- pharmacokinetic comparison is done at any dose, more preferably at equivalent dose (i.e. same mg/kg dose) or equimolar dose (i.e.
- a dose used for pharmacokinetic measurement is selected from 0.001 to 1000 mg/kg, more preferably 0.01 to 100 mg/kg, more preferably 0.1 to 50 mg/kg, more preferably 0.5 to 10 mg/kg.
- tumor-localized activation of immune cells means that immune cells are activated preferentially in tumor tissue as compared to a non-tumor tissue.
- treatment refers to both therapeutic treatment and prophylactic or preventive measures, wherein the object is to prevent or slow down an undesired physiological change or disorder.
- beneficial or desired clinical results include, but are not limited to, alleviation of symptoms, diminishment of extent of disease, stabilized (i.e., not worsening) state of disease, delay or slowing of disease progression, amelioration or palliation of the disease state, and remission (whether partial or total), whether detectable or undetectable.
- Treatment can also mean prolonging survival as compared to expected survival if not receiving treatment.
- the terms “treat”, “treatment” and “treating” referto the inhibition of the progression of a proliferative disorder, either physically by, e.g., stabilization of a discernible symptom, physiologically by, e.g., stabilization of a physical parameter, or both.
- the terms “treat”, “treatment” and “treating” refer to the reduction or stabilization of tumor size or cancerous cell count.
- peptide also encompasses peptides modified by, e.g, glycosylation, and proteins comprising two or more polypeptide chains, each of length of 4 to 600 amino acids long, cross-linked by, e.g., disulfide bonds, such as, e.g., insulin and immunoglobulins.
- chemical or biochemical agent is intended to include any naturally occurring or synthetic compound that may be administered to a recipient.
- the term “medical condition“(or disorder or disease) includes autoimmune disorders, inflammatory disorders, retinopathies (particularly proliferative retinopathies), neurodegenerative disorders, infections, metabolic diseases, and neoplastic diseases. Any of the recombinant binding proteins described herein may be used forthe preparation of a medicament for the treatment of such a disorder, particularly a disorder selected from the group comprising: an autoimmune disorder, an inflammatory disorder, an immune disorder, and a neoplastic disease.
- a “medical condition” may be one that is characterized by inappropriate cell proliferation.
- a medical condition may be a hyperproliferative condition.
- the invention particularly relates to a method of treating a medical condition, the method comprising the step of administering, to a subject in need of such treatment, a therapeutically effective amount of a recombinant binding protein or said pharmaceutical composition of the invention.
- said medical condition is a neoplastic disease.
- neoplastic disease refers to an abnormal state or condition of cells or tissue characterized by rapidly proliferating cell growth or neoplasm.
- said medical condition is a malignant neoplastic disease.
- said medical condition is cancer.
- therapeutically effective amount means an amount that is sufficient to produce a desired effect on a subject.
- antibody means not only intact antibody molecules, but also any fragments and variants of antibody molecules that retain immunogen-binding ability. Such fragments and variants are also known in the art and are regularly employed both in vitro and in vivo. Accordingly, the term “antibody” encompasses intact immunoglobulin molecules, antibody fragments such as, e.g., Fab, Fab', F(ab')2, and single chain V region fragments (scFv), bispecific antibodies, chimeric antibodies, antibody fusion polypeptides, and unconventional antibodies.
- Alternative scaffolds include any polypeptides or proteins comprising a binding domain that is capable of binding a target and that is not derived from an antibody or immunoglobulin molecule.
- alternative scaffolds may comprise or may be derived from a variety of different polypeptide or protein structures.
- Alternative scaffolds include, but are not limited to, adnectins (monobodies), affibodies, affilins, affimers and aptamers, affitins, alphabodies, anticalins, armadillo repeat protein-based scaffolds, atrimers, avimers, ankyrin repeat protein-based scaffolds (such as DARPin proteins), fynomers, knottins, and Kunitz domain peptides.
- Alternative scaffolds are described, e.g., in Yu et al., Annu Rev Anal Chem (Palo Alto Calif). 2017 June 12; 10(1): 293-320. doi:10.1146/annurevanchem-061516-045205.
- cancer and “cancerous” are used herein to refer to or describe the physiological condition in mammals that is typically characterized by unregulated cell growth.
- Cancer encompasses solid tumors and liquid tumors, as well as primary tumors and metastases.
- a "tumor” comprises one or more cancerous cells.
- Solid tumors typically also comprise tumor stroma. Examples of cancer include, but are not limited to, primary and metastatic carcinoma, lymphoma, blastoma, sarcoma, and leukemia, and any other epithelial and lymphoid malignancies.
- cancers include brain cancer, bladder cancer, breast cancer, ovarian cancer, clear cell kidney cancer, head/neck squamous cell carcinoma, lung adenocarcinoma, lung squamous cell carcinoma, malignant melanoma, non-small-cell lung cancer (NSCLC), ovarian cancer, pancreatic cancer, prostate cancer, renal cell carcinoma, small-cell lung cancer (SCLC), breast cancer, acute lymphoblastic leukemia (ALL), acute myeloid leukemia (AML), chronic lymphocytic leukemia (CLL), chronic myeloid leukemia (CML), diffuse large B-cell lymphoma (DLBCL), follicular lymphoma, Hodgkin's lymphoma (HL), mantle cell lymphoma (MCL), multiple myeloma (MM), myelodysplastic syndrome (MDS), non-Hodgkin's lymphoma (NHL), Squamous Cell Carcinoma of the Head and Neck (SCCHN
- Such randomized modules in such libraries may comprise additional polypeptide loop insertions with randomized amino acid positions.
- polypeptide loop insertions are complement determining region (CDR) loop libraries of antibodies or de novo generated peptide libraries.
- CDR complement determining region
- such a loop insertion could be designed using the structure of the N-terminal ankyrin repeat domain of human ribonuclease L (Tanaka, N., Nakanishi, M, Kusakabe, Y, Goto, Y., Kitade, Y, Nakamura, K.T., EMBO J. 23(30), 3929-3938, 2004) as guidance.
- ankyrin repeat proteins libraries may contain randomized loops (with fixed and randomized positions) of variable length (e.g. 1 to 20 amino acids) inserted in one or more beta-turns of an ankyrin repeat domain.
- any such N-terminal capping module of an ankyrin repeat protein library preferably possesses the RILLAA, RILLKA or RELLKA motif (e.g. present from position 21 to 26 in SEQ ID NO: 1) and any such C-terminal capping module of an ankyrin repeat protein library preferably possesses the KLN, KLA or KAA motif (e.g. present at the last three amino acids in SEQ ID NO: 1).
- SEQ ID NOs: 22 to 36 provide examples of N- terminal capping modules comprising the RILLAA, RILLKA or RELLKA motif
- SEQ ID NOs: 37 to 60 provide examples of C-terminal capping modules comprising the KLN, KLA or KAA motif.
- ankyrin repeat protein library may be guided by known structures of an ankyrin repeat domain interacting with a target.
- Examples of such structures identified by their Protein Data Bank (PDB) unique accession or identification codes (PDB-IDs), are 1WDY, 3V31 , 3V30, 3V2X, 3V2O, 3UXG, 3TWQ-3TWX, 1 N11 , 1 S70 and 2ZGD.
- PDB Protein Data Bank
- N2C and N3C designed ankyrin repeat protein libraries have been described (U.S. Patent No. 7,417,130; Binz et al. 2003, loc. cit.; Binz et al. 2004, loc. cit.).
- the digit in N2C and N3C describes the number of randomized repeat modules present between the N-terminal and C-terminal capping modules.
- ankyrin repeat domains of SEQ ID NOs: 1 to 8 constitute amino acid sequences of selected binding proteins comprising an ankyrin repeat domain with binding specificity for CD47.
- Individual ankyrin repeat modules from such ankyrin repeat domains with binding specificity for CD47 are provided, e.g., in SEQ ID NOs: 9 to 21.
- the target format chosen was based on single chain format, consisting of the extracellular domain of human CD47 protein (Uniprot ID: Q08722, residues 19 to 141 , SEQ ID NO: 67; purchased from BPS) linked to an Fc domain and a C-terminal Avi-tag for site-directed biotinylation. It was expressed in Human embryonic kidney 293 cells (HEK293) cells. A batch of biotinylated and a batch of non-biotinylated protein were purchased. The final material was formulated in 8 mM phosphate at pH 7.4, 110 mM NaCI, 2.2 mM KCI and 20% glycerol.
- CD47-specific ankyrin repeat proteins was performed by ribosome display (Hanes and Pluckthun, loc. cit.) using part of the extracellular domain of CD47 (SEQ ID NO: 67) as target protein, libraries of ankyrin repeat proteins as described above, and established protocols (see, e.g., Zahnd, C., Amstutz, P. and Pluckthun, A., Nat. Methods 4, 69-79, 2007). The number of reverse transcription (RT)- PCR cycles after each selection round was constantly thirty.
- RT reverse transcription
- the first four rounds of selection employed standard ribosome display selection, using decreasing target concentration (400 nM, 100 nM, 25 nM and 5 nM, respectively), followed by a 5 th off rate round using a target concentration of 1 nM and a competitor benchmark anti-CD47 antibody at 1000-fold higher concentration in order to select high affinity binders, and a 6 th recovery round at a target concentration of 5 nM.
- the pools from the ribosome display were expressed in E. coli cells in 96 well plates. 12 plates of DARPin proteins from ribosome display round 4 and 14 plates of DARPin proteins from round 6 were expressed in E. coli cells with a Flag-tag and a His-tag. Crude extracts thereof were prepared to test binding of the Flag-tagged DARPin proteins to human CD47 recombinant protein in an HTRF assay. The crude extracts were diluted 1 :1600 in PBS-TB (PBS supplemented with 0.2% (w/v) BSA and 0.1 % Tween20, pH 7.4) and used at 1 :6400 dilution (final) in the assay.
- PBS-TB PBS supplemented with 0.2% (w/v) BSA and 0.1 % Tween20, pH 7.4
- Binding was performed against 6 nM (final concentration) of human biotinylated CD47-Fc.
- FRET donor Streptavidin-Tb
- acceptor MAb Anti Flag M2-d2 conjugate
- RT room temperature
- the HTRF was read-out on a Tecan M1000 using a 340 nm excitation wavelength and a 665 ⁇ 10 nm emission filter.
- binders Four plates of binders were again expressed in E. coli cells and purified on 96 well plates using their His- tag according to standard protocols. The proteins were monomeric and soluble when concentrated to 10 mg/ml in TBS pH 8.0 (50 mM Tris, 500 mM NaCI). The following panel of eight designed ankyrin repeat proteins with binding specificity for human CD47 were selected:
- DARPin protein #1 (SEQ ID NO:1 with a His-tag (SEQ ID NO: 61) fused to its N terminus);
- DARPin protein #2 (SEQ ID NO:2 with a His-tag (SEQ ID NO: 61) fused to its N terminus);
- DARPin protein #3 (SEQ ID NO:3 with a His-tag (SEQ ID NO: 61) fused to its N terminus);
- DARPin protein #4 (SEQ ID NO:4 with a His-tag (SEQ ID NO: 61) fused to its N terminus);
- DARPin protein #5 (SEQ ID NO:5 with a His-tag (SEQ ID NO: 61) fused to its N terminus);
- DARPin protein #6 (SEQ ID NO:6 with a His-tag (SEQ ID NO: 61) fused to its N terminus);
- DARPin protein #7 (SEQ ID NO:7 with a His-tag (SEQ ID NO: 61) fused to its N terminus);
- DARPin protein #8 (SEQ ID NO:8 with a His-tag (SEQ ID NO: 61) fused to its N terminus).
- Binding of the purified designed ankyrin repeat proteins in HTRF was confirmed with 40 nM of each designed ankyrin repeat protein and 10 nM of human biotinylated CD47-Fc (see Figure 1).
- the eight selected designed ankyrin repeat proteins binding to human CD47 were tested for SIRPI a competition using the PathHunter Jurkat SIRPa Signaling Bioassay Kit purchased from Eurofins DiscoverX products LLC. Ligand engagement through co-culture of Jurkat CD47 presenting cells and Jurkat SIRPI a signaling cells results in phosphorylation and intracellular pathway activation leading to a chemiluminescence signal. This signal can be inhibited in the presence of an anti-CD47 antibody or anti- SIRPa antibody disrupting ligand-receptor interaction. As can be seen in Figures 2 (A- D), all eight designed ankyrin repeat proteins were able to inhibit ligand interaction. IC50 values of DARPin proteins #1 to 8 obtained in this assay are shown in Table 2.
- CD47 binding was confirmed by a cell binding assay using a Raji cell line (purchased from ATCC), a cancer cell line expressing CD47.
- the CD47 expressing cells were washed Ix with 500 pl PBS, spun at 350g for 5 minutes and the supernatant was discarded.
- a titration of selected CD47 designed ankyrin repeat proteins diluted in FACS buffer was added to the pellet of cells, resuspended and incubated for 30 minutes at 4°C. Cells were washed twice with 200 pl cold PBS, spun at 350g for 5 minutes and the supernatant was discarded.
- Example 2 Stability assessment of exemplary CD47-specific ankyrin repeat proteins
- CD47-specific designed ankyrin repeat proteins were analysed in-depth to assess their biophysical properties.
- purified CD47-specific ankyrin repeat proteins DARPin #1 (SEQ ID NO:1), DARPin #2 (SEQ ID NO: 2), DARPin #3 (SEQ ID NO: 3), DARPin #4 (SEQ ID NO: 4), DARPin #5 (SEQ ID NO: 5), DARPin #6 (SEQ ID NO: 6), DARPin #7 (SEQ ID NO: 7) and DARPin #8 (SEQ ID NO: 8) in PBS, pH7.4 were aliquoted into sterile glass vials (Schmidlin: LPP 11 09 0620) and stressed by incubation at 60°C for 1 week.
- the applied stress conditions allow a prediction of the biophysical properties after 2 years storage at 4°C.
- an aliquot was stored at -70 C as a reference.
- the reference and the heat-stressed samples were exposed to one freeze-thaw cycle before analysis.
- the samples were evaluated for multimerization, aggregation and fragmentation by SDS-PAGE and analytic SEC. Stressed samples were then compared to the respective reference samples. No colour change, turbidity or precipitation was detected by visual inspection. Furthermore, no significant degradation or aggregation was detected.
- the results of the experiments demonstrated that samples were stable in the applied stress conditions.
- the same CD47-specific ankyrin repeat proteins were also assessed for their thermal stability and unfolding/refolding propensity using a Jasco J-815 spectrophotometer (non-stressed proteins only).
- the Tm (melting temperature) of the selected proteins is determined by CD as a parameter for thermal stability. The ellipticity was recorded at 222 nM and a temperature range from 20°C to 90°C was applied followed by reverse scan to record the refolding behavior.
- the Tm of the selected CD47-specific ankyrin repeat proteins is the midpoint of the protein unfolding. Spectra from 190-250 nm were recorded before and after the temperature scan.
- Samples were prepared at a concentration of 2 pM in PBS, pH7.4. As it can be seen in Table 4, for all tested proteins a Tm higher than 80° C was obtained.
- Table 4 T m (melting temperature) of eight exemplary CD47-specific ankyrin repeat proteins
- Example 3 Determination of dissociation constants (KD) of ankyrin repeat proteins with binding specificity for human CD47 by Surface Plasmon Resonance (SPR) analysis
- binding affinities of eight purified ankyrin repeat proteins to biotinylated recombinant human CD47-Fc target were analyzed using a ProteOn XPR 36 instrument (NAHLC200M, Xantec ProteOn Sensor Chip) and the measurement was performed according to standard procedures.
- SEQ ID NO: 1 to SEQ ID NO: 8 were subcloned into derivatives of the pQE30 (Qiagen) expression vector, containing an N- terminal His-tag, and expressed and purified as described above (yielding DARPin protein #1 , DARPin protein #2, DARPin protein #3, DARPin protein #4, DARPin protein #5, DARPin protein #6, DARPin protein #7 and DARPin protein #8).
- biotinylated human CD47-Fc target was diluted in PBST (PBS, pH 7.4 containing 0.005% Tween 20®) and coated on an NAHLC200M chip (BioRad) to a level of around 350 and 800 resonance units (RU).
- PBST PBS, pH 7.4 containing 0.005% Tween 20®
- NAHLC200M chip BioRad
- ankyrin repeat protein and human CD47 were then measured by injecting 200 pl running buffer (PBS, pH 7.4 containing 0.005% Tween 20®) containing serial dilutions of ankyrin repeat proteins covering a concentration range between 16.7 nM, 5.6 nM, 1.85 nM and 0.62 nM for multi-trace SPR measurements, followed by a running buffer flow for at least 20 minutes at a constant flow rate of 100 pl/min (off-rate measurement).
- the regeneration was performed using 30 pl of 10 mM Glycine pH 2.5.
- the signals i.e. resonance unit (RU) values
- an empty lane (no target coated) and a reference injection i.e. injection of running buffer only
- Binding parameters (KD, on-rate, off-rate) against CD47 were determined for the constructs as shown in Table 4.
- Figure 4 shows SPR traces obtained for DARPin molecules #2, 4 and 7.
- Dissociation constants KD were calculated from the estimated on- and off-rates using standard procedures. KD values of the binding interactions of selected ankyrin repeat proteins with human CD47 were determined to be in the range of 0.1 to 0.8 nM (see Table 5).
- Table 5 Binding parameters for ankyrin repeat proteins with binding specificity for human CD47 by Surface Plasmon Resonance (SPR)
- DARPin proteins of SEQ ID NOs: 4 to 8 were analyzed in mice.
- DARPin sequences were subcloned and expressed as described above into derivatives of the pQE30 (Qiagen) expression vector.
- the constructs contained an N-terminal His-tag, followed by an HSA (human serum albumin) binding ankyrin repeat domain for half-life extension (SEQ ID NO: 63), and then followed by one of the CD47 specific binding domains.
- HSA human serum albumin binding ankyrin repeat domain for half-life extension
- DARPin protein #9 comprising SEQ ID NO: 68;
- DARPin protein #10 comprising SEQ ID NO: 69;
- DARPin protein #11 comprising SEQ ID NO: 70;
- DARPin protein #12 comprising SEQ ID NO: 71 ;
- DARPin protein #13 comprising SEQ ID NO: 72.
- DARPin protein #9, DARPin protein #10, DARPin protein #11 , DARPin protein #12 and DARPin protein #13 were administered as a single intravenous bolus injection into the tail vein of six mice for each ankyrin repeat fusion protein.
- the target dose level was 1 mg/kg with an application volume of 5 mL/kg.
- Ankyrin repeat fusion proteins were formulated in phosphate-buffered saline (PBS) solution. Mice were split into two groups with equal numbers of animals. Four serum samples were collected from each mouse.
- PBS phosphate-buffered saline
- Blood samples for pharmacokinetic investigations were collected from the saphenous vein at 5 min, 6 h, 24 h, 48 h, 76 h, 96 h and 168 h post dosing. Blood was kept at room temperature to allow clotting followed by centrifugation and collection of serum.
- Figure 5 shows the serum concentrations of DARPin protein #9, DARPin protein #10, DARPin protein #11 , DARPin protein #12 and DARPin protein #13 as a function of time after the single intravenous administration into mice.
- the traces indicate monoexponential elimination of the compounds.
- Pharmacokinetic data analysis was performed using Version 8.3 of the WinNonlin program as part of Phoenix 64, Pharsight, North Carolina. Calculation of the pharmacokinetic parameters based on the mean concentration-time data of the animals dosed via intravenous bolus injection was performed with noncompartmental analysis (NCA model 200-202, IV bolus, linear trapezoidal linear interpolation). The following pharmacokinetic parameters were calculated:
- Vss i.v. dose • AUMCinf I (AUCinf)2.
- AUMCinf denotes the total area under the first moment of drug concentration-time curve extrapolated to infinity using the same extrapolation procedure as described for calculation of AUCinf.
- Table 5 shows the summary of pharmacokinetic characteristics of the five tested ankyrin repeat proteins DARPin protein #9, DARPin protein #10, DARPin protein #11 , DARPin protein #12 and DARPin protein #13 following single intravenous administration of 1 mg/kg.
- Table 6 Pharmacokinetic parameters for five exemplary HSA/CD47 specific ankyrin repeat proteins As it can been seen in the Table 6 above and Figure 5, the half-life of all five tested designed ankyrin proteins in mouse is over 30h (32-38h) and in a similar range.
- Example 5 Potency assessment of selected CD47 specific binding proteins using a SIRPa signaling assay
- Selected CD47-specific designed ankyrin repeat proteins formatted as trispecific proteins to also include (1) a designed ankyrin repeat domain specific for a tumor associated antigen (TAA) and (2) a designed ankyrin repeat domain specific for CD16a (SEQ ID NO: 81) (CD16a (UniProt (www.uniprot.org) Ref. No. P08637) is expressed on the surface of certain immune cells), were cloned, produced and tested for SIRP1 a competition using the PathHunter Jurkat SIRPa Signaling Bioassay Kit as described above.
- TAA tumor associated antigen
- CD16a UniProt (www.uniprot.org) Ref. No. P08637
- ligand engagement through co-culture of TAA positive/CD47 positive ligand cells and Jurkat SIRPI a signaling cells results in phosphorylation and intracellular pathway activation leading to a chemiluminescence signal.
- This signal can be inhibited in the presence of an anti-CD47 antibody or anti- SIRPIa antibody disrupting ligand-receptor interaction.
- the eight selected CD47-specific designed ankyrin repeat proteins in the trispecific format were able to inhibit ligand interaction. More particularly, Fig.
- FIG. 6 and Table 7 show the inhibition caused by CD47-specific DARPin proteins (SEQ ID NOs: 1 to 7), linked to a CD16a-specific designed ankyrin domain (SEQ ID NO: 81) and to a TAA-specific designed ankyrin repeat domain, upon addition to CD47 positive/TAA positive ligand cells and Jurkat SIRPIa signaling cells.
- Figure 7A shows the inhibition caused by a CD47-specific DARPin protein (SEQ ID NO: 8) in the described trispecific format or in mono-domain (or mono-specific) format (DARPin protein #8) upon addition to the SIRPIa signaling cells in the presence of CD47 positive/TAA negative ligand cells.
- the CD47-specific DARPin protein inhibited SIRPI a signaling equally well in the mono- or tri-specific formats in the presence of CD47 positive/TAA negative ligand cells.
- Figure 7B shows the inhibition caused by a CD47-specific DARPin protein (SEQ ID NO: 8) in the described trispecific format or in mono-domain format (DARPin protein #8) upon addition to the SIRPI a signaling cells in the presence of CD47 positive/TAA positive ligand cells.
- the CD47-specific DARPin protein inhibited SIRPIa signaling much stronger in the tri-specific format than in the mono-specific format in the presence of CD47 positive/TAA positive ligand cells.
- DARPin protein #9 (SEQ ID NO: 94; including the ankyrin repeat domains of SEQ ID NO: 8, SEQ ID NO: 89 and SEQ ID NO: 81), DARPin protein #10 (SEQ ID NO: 95; including the ankyrin repeat domains of SEQ ID NO: 8, SEQ ID NO: 90 and SEQ ID NO: 81), DARPin protein #11 (SEQ ID NO: 96; including the ankyrin repeat domains of SEQ ID NO: 8, SEQ ID NO: 91 and SEQ ID NO: 81), DARPin protein #12 (SEQ ID NO: 97; including the ankyrin repeat domains of SEQ ID NO: 8, SEQ ID NO: 92 and SEQ ID NO: 81), and DARPin protein #13 (SEQ ID NO: 98; including the ankyrin repeat domains of SEQ ID NO: 8, SEQ ID NO: 93 and SEQ ID NO: 81) were able to inhibit ligand interaction, when added
- DARPin proteins #9 to #13 show increased SIRPI a signaling inhibition when compared to DARPin protein #8 in the co-culture of Jurkat and Kasumi 1 cell lines, indicating the avidity effect caused by the CD47-specific ankyrin repeat domain and the CD117-specific ankyrin repeat domain, which both bind to the surface of the Kasumi-1 cells.
- a benchmark anti-CD47 monoclonal antibody was used, having a bivalent structure.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Organic Chemistry (AREA)
- Biochemistry (AREA)
- Genetics & Genomics (AREA)
- Gastroenterology & Hepatology (AREA)
- Toxicology (AREA)
- Immunology (AREA)
- Biophysics (AREA)
- General Health & Medical Sciences (AREA)
- Zoology (AREA)
- Medicinal Chemistry (AREA)
- Molecular Biology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Cell Biology (AREA)
- Peptides Or Proteins (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
Abstract
Description
Claims
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP23177836.6 | 2023-06-06 | ||
| EP23177836 | 2023-06-06 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2024251695A1 true WO2024251695A1 (en) | 2024-12-12 |
Family
ID=86731972
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2024/065257 Ceased WO2024251695A1 (en) | 2023-06-06 | 2024-06-04 | Recombinant cd47 binding proteins and their use |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2024251695A1 (en) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2025146491A1 (en) | 2024-01-05 | 2025-07-10 | Molecular Partners Ag | Designed repeat domains with dual binding specificity for cd117 and cd47-binding agent |
| WO2025146490A1 (en) * | 2024-01-05 | 2025-07-10 | Molecular Partners Ag | Recombinant cd117 binding proteins and their use |
| WO2025146487A1 (en) | 2024-01-05 | 2025-07-10 | Molecular Partners Ag | Multispecific binding proteins |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2002020565A2 (en) | 2000-09-08 | 2002-03-14 | Universität Zürich | Collections of repeat proteins comprising repeat modules |
| WO2005063815A2 (en) | 2003-11-12 | 2005-07-14 | Biogen Idec Ma Inc. | Fcϝ receptor-binding polypeptide variants and methods related thereto |
| WO2009040338A1 (en) | 2007-09-24 | 2009-04-02 | University Of Zürich | Designed armadillo repeat proteins |
| WO2012069655A2 (en) | 2010-11-26 | 2012-05-31 | Molecular Partners Ag | Improved n-terminal capping modules for designed ankyrin repeat proteins |
| US9458211B1 (en) | 2015-04-02 | 2016-10-04 | Molecular Partners Ag | Recombinant proteins that simultaneously bind HGF, VEGF-A and serum albumin, comprising ankyrin repeat domains |
| WO2021229076A1 (en) * | 2020-05-14 | 2021-11-18 | Molecular Partners Ag | Recombinant cd40 binding proteins and their use |
-
2024
- 2024-06-04 WO PCT/EP2024/065257 patent/WO2024251695A1/en not_active Ceased
Patent Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2002020565A2 (en) | 2000-09-08 | 2002-03-14 | Universität Zürich | Collections of repeat proteins comprising repeat modules |
| US7417130B2 (en) | 2000-09-08 | 2008-08-26 | University Of Zurich | Collection of repeat proteins comprising repeat modules |
| WO2005063815A2 (en) | 2003-11-12 | 2005-07-14 | Biogen Idec Ma Inc. | Fcϝ receptor-binding polypeptide variants and methods related thereto |
| WO2009040338A1 (en) | 2007-09-24 | 2009-04-02 | University Of Zürich | Designed armadillo repeat proteins |
| WO2012069655A2 (en) | 2010-11-26 | 2012-05-31 | Molecular Partners Ag | Improved n-terminal capping modules for designed ankyrin repeat proteins |
| US20130296221A1 (en) | 2010-11-26 | 2013-11-07 | Molecular Partners Ag | Improved capping modules for designed ankyrin repeat proteins |
| US9458211B1 (en) | 2015-04-02 | 2016-10-04 | Molecular Partners Ag | Recombinant proteins that simultaneously bind HGF, VEGF-A and serum albumin, comprising ankyrin repeat domains |
| WO2021229076A1 (en) * | 2020-05-14 | 2021-11-18 | Molecular Partners Ag | Recombinant cd40 binding proteins and their use |
Non-Patent Citations (25)
| Title |
|---|
| "Remington's Pharmaceutical Sciences", 1980 |
| "UniProt", Database accession no. P02768 |
| BINZ ET AL., NAT. BIOTECHNOL., vol. 22, 2004, pages 575 - 582 |
| CHAM ET AL., ANTIBODIES, vol. 9, 2020, pages 44 |
| DIZMAN ET AL., CANCERS, vol. 13, no. 24, December 2021 (2021-12-01), pages 6229 |
| EI-RASHID ET AL., FASEB J, vol. 33, 2019, pages 12735 - 12749 |
| FORRERFORRERSTUMPP, M.TBINZ, H.KPLUCKTHUN, A ET AL., FEBS LETTERS, vol. 539, 2003, pages 2 - 6 |
| HANES, JPLUCKTHUN, A, PNAS, vol. 94, 1997, pages 4937 - 42 |
| INTERLANDI ET AL., J MOL BIOL., vol. 375, no. 3, 18 January 2008 (2008-01-18), pages 837 - 54 |
| LIU ET AL., SIGNAL TRANSDUCT TARGET THER, vol. 8, no. 1, 7 March 2023 (2023-03-07), pages 104 |
| MAIN, E.RXIONG, YCOCCO, M.JD'ANDREA, LREGAN, L, STRUCTURE, vol. 11, no. 5, 2003, pages 497 - 508 |
| NGUYEN ET AL., SENSORS, vol. 15, no. 5, 5 May 2015 (2015-05-05), pages 10481 - 510 |
| NICOLAS ET AL: "Functional interaction between Rh proteins and the spectrin-based skeleton in erythroid and epithelial cells", TRANSFUSION CLINIQUE ET BIOLOGIQUE, ARNETTE-BLACKWELL, PARIS, FR, vol. 13, no. 1-2, 1 March 2006 (2006-03-01), pages 23 - 28, XP005391593, ISSN: 1246-7820, DOI: 10.1016/J.TRACLI.2006.02.019 * |
| NOVELLI ET AL., AM. J. PHYSIOL. LUNG CELL MOL. PHYSIOL., vol. 316, 2019, pages L1150 - L1164 |
| SAMBROOK JFRITSCH E.FMANIATIS T: "Molecular Cloning: A Laboratory Manual", 1989, COLD SPRING HARBOR LABORATORY |
| SHAH ET AL., J VIS EXP, no. 84, 2014, pages 51383 |
| STUMPP ET AL., DRUG DISCOV. TODAY, vol. 13, 2008, pages 695 - 701 |
| SUN ET AL., LEUK RES REP, vol. 14, September 2021 (2021-09-01), pages 16 |
| TANAKA, N.NAKANISHI, MKUSAKABE, YGOTO, YKITADE, YNAKAMURA, K.T, EMBO J., vol. 23, no. 30, 2004, pages 3929 - 3938 |
| VELLIQUETTE ET AL., TRANSFUSION, vol. 59, no. 2, February 2019 (2019-02-01), pages 730 - 737 |
| WILLIAMS ET AL., J BIOL CHEM, vol. 266, 1991, pages 5182 - 5190 |
| YE, ACTA PHARM SIN B., vol. 13, no. 4, April 2023 (2023-04-01), pages 1467 - 1487 |
| YU ET AL., ANNU REV ANAL CHEM (PALO ALTO CALIF)., vol. 10, no. 1, 12 June 2017 (2017-06-12), pages 293 - 320 |
| ZAHND, CAMSTUTZ, PPLUCKTHUN, A, NAT. METHODS, vol. 4, 2007, pages 69 - 79 |
| ZHANG ET AL., FRONT IMMUNOL, vol. 11, 28 January 2020 (2020-01-28), pages 18 |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2025146491A1 (en) | 2024-01-05 | 2025-07-10 | Molecular Partners Ag | Designed repeat domains with dual binding specificity for cd117 and cd47-binding agent |
| WO2025146490A1 (en) * | 2024-01-05 | 2025-07-10 | Molecular Partners Ag | Recombinant cd117 binding proteins and their use |
| WO2025146487A1 (en) | 2024-01-05 | 2025-07-10 | Molecular Partners Ag | Multispecific binding proteins |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US11332501B2 (en) | Recombinant binding proteins and their use | |
| US20220242973A1 (en) | Recombinant fap binding proteins and their use | |
| WO2024251695A1 (en) | Recombinant cd47 binding proteins and their use | |
| US20240279309A1 (en) | Recombinant cd40 binding proteins and their use | |
| US20240317852A1 (en) | Novel darpin based cd33 engagers | |
| US20240150475A1 (en) | Novel darpin based cd123 engagers | |
| US20250074994A1 (en) | Novel darpin based cd70 engagers | |
| WO2024251628A1 (en) | Recombinant cd16a binding proteins and their use | |
| EP4263608A1 (en) | Recombinant cd3 binding proteins and their use | |
| WO2025146490A1 (en) | Recombinant cd117 binding proteins and their use | |
| WO2025163083A1 (en) | Recobminant dll3 binding proteins and their use | |
| CN116802213A (en) | Recombinant CD3 binding proteins and their uses | |
| CN117242100A (en) | Novel CD70 conjugate based on DARPin | |
| HK1244009B (en) | Designed ankyrin repeat domains with binding specificity for serum albumin |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24731842 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2024731842 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2024731842 Country of ref document: EP Effective date: 20260107 |
|
| ENP | Entry into the national phase |
Ref document number: 2024731842 Country of ref document: EP Effective date: 20260107 |
|
| ENP | Entry into the national phase |
Ref document number: 2024731842 Country of ref document: EP Effective date: 20260107 |
|
| ENP | Entry into the national phase |
Ref document number: 2024731842 Country of ref document: EP Effective date: 20260107 |
|
| ENP | Entry into the national phase |
Ref document number: 2024731842 Country of ref document: EP Effective date: 20260107 |