WO2024251966A1 - Crystalline reduced nicotinamide mononucleotide (nmnh) and process thereof - Google Patents
Crystalline reduced nicotinamide mononucleotide (nmnh) and process thereof Download PDFInfo
- Publication number
- WO2024251966A1 WO2024251966A1 PCT/EP2024/065769 EP2024065769W WO2024251966A1 WO 2024251966 A1 WO2024251966 A1 WO 2024251966A1 EP 2024065769 W EP2024065769 W EP 2024065769W WO 2024251966 A1 WO2024251966 A1 WO 2024251966A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- compound
- nmnh
- crystalline
- formula
- iii
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07H—SUGARS; DERIVATIVES THEREOF; NUCLEOSIDES; NUCLEOTIDES; NUCLEIC ACIDS
- C07H19/00—Compounds containing a hetero ring sharing one ring hetero atom with a saccharide radical; Nucleosides; Mononucleotides; Anhydro-derivatives thereof
- C07H19/02—Compounds containing a hetero ring sharing one ring hetero atom with a saccharide radical; Nucleosides; Mononucleotides; Anhydro-derivatives thereof sharing nitrogen
- C07H19/04—Heterocyclic radicals containing only nitrogen atoms as ring hetero atom
- C07H19/048—Pyridine radicals
Definitions
- NMNH reduced nicotinamide mononucleotide
- NMNH reduced nicotinamide mononucleotide
- BACKGROUND OF THE INVENTION Nicotinamide adenine dinucleotide (NAD+) is essential to living organisms since it participates in various biological reactions and regulates key biological processes, such as metabolism and DNA repair. Nicotinamide adenine dinucleotide (NAD+) homeostasis is constantly compromised due to degradation by NAD+ -dependent enzymes which lead to various age related diseases.
- NMN nicotinamide mononucleotide
- NAD+ replenishment by supplementation with the NAD+ precursors nicotinamide mononucleotide (NMN) is thought to alleviate this imbalance.
- Recent studies have revealed that reduced nicotinamide mononucleotide (NMNH) is a potent NAD precursor than NMN both in vitro and in vivo.
- NMNH increases cellular NADH levels, suppresses glycolysis and TCA cycle, as well as cell growth.
- the researchers worldwide have focused on NMNH and its production on large scale such that NMNH would be available at lower prices than the precursors currently available.
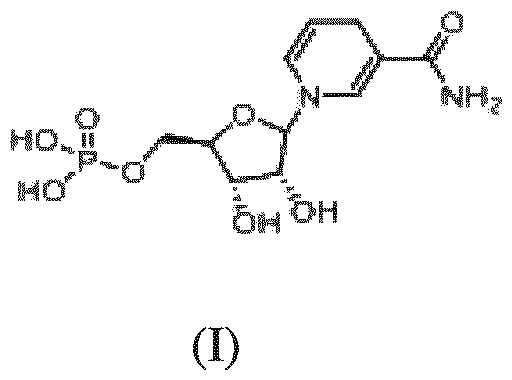
- NMNH chemically known as ((2R,3S,4R)-5-(3-carbamoylpyridin-1(4H)-yl)-3,4- dihydroxytetrahydrofuran-2-yl)methyl dihydrogen phosphate is the reduced form of NMN with the molecular formula of C 11 H 17 N 2 O 8 P, is stable under alkaline pH and low temperature conditions, but is unstable at neutral pH in solution.
- NMN-H Given the therapeutic benefits as nutraceuticals associated with NMN-H, the present inventors envisaged the need in the art to provide novel crystalline form of NMN-H and to the process for synthesis thereof which is efficient, industrially scalable and provides NMNH in high yield and purity.
- NMNH reduced nicotinamide mononucleotide
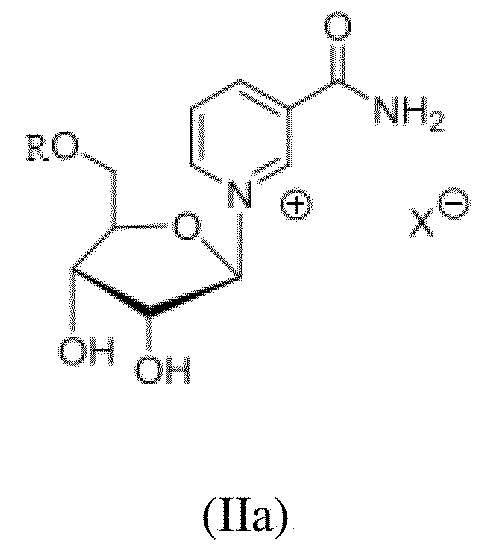
- the present invention provides a process for synthesis of crystalline form of reduced nicotinamide mononucleotide (NMNH) of Formula I (I) from the compound of general Formula (II) or (IIa); (II) or (IIa) wherein R is selected from hydrogen or –P(O)R1R2; R1 and R2 independently represent OH and O-respectively; and X- is halo, or triflate. comprising; i. Ketalizing the compound of Formula II, wherein R is hydrogen; X- is halo or triflate to a compound of Formula III
- R3 and R4 are alkyl, X- is halo or triflate ;
- Phosphorylating compound of Formula (III) with PO(OR 5 )Cl to obtain the compound of Formula (IV) wherein R3, R4, R5, R6 and R7 are independently alkyl, X- is halo or triflate;
- the present invention provides a composition comprising the crystalline compound of reduced nicotinamide mononucleotide of Formula (I) with suitable excipients.
- FIGURES Figure 1 depict the chromatogram of as prepared NMNH.
- Figure 2 depict the Masss pectra of as prepared NMNH
- Figure 3 depict the Powder X-RD of NMN
- Figure 4 depict the Powder X-RD of NMNH DETAILED DESCRIPTION OF THE INVENTION:
- the present invention will now be described in detail in its preferred and optional embodiments so that various aspects of the invention will be fully understood without limiting the scope of the invention.
- NMNH reduced nicotinamide mononucleot
- the present invention discloses a process for synthesis of crystalline form of reduced nicotinamide mononucleotide (NMNH)of Formula I from the compound of general Formula (II) or (IIa) ; (II) or (IIa) wherein R is selected from hydrogen or –P(O)R1R2; R1 and R2 independently represent OH and O-respectively; and X- is halo or triflate. comprising; i. Ketalizing the compound of Formula II, wherein R is hydrogen; X- is halo or triflate; to a compound of Formula III wherein R3 and R4 are alkyl, X- is halo or triflate; ii. Phosphorylating compound of Formula (III) with PO(OR 5 )Cl to obtain the compound of Formula (IV)
- R3, R4, R5, R6 and R7 are independently alkyl, X- is halo or triflate; iii. Reducing compound (IV) with dithionite under ice-cold conditions to obtain compound V; wherein R3, R4, R6 and R7 are independently alkyl; and iv. Deprotecting the compound of Formula (V) and purifying in methanol to obtain reduced nicotinamide mononucleotide of formula (I). OR i.
- the present invention relates to a process for synthesis of crystalline form of reduced nicotinamide mononucleotide (NMNH) of Formula I from the compound of general Formula (II); Wherein R is selected from hydrogen or –P(O)R1R2; R1 and R2 independently represent OH and O- respectively; and X- is halo or triflate.
- R is selected from hydrogen or –P(O)R1R2; R1 and R2 independently represent OH and O- respectively; and X- is halo or triflate.
- R3 and R4 are independently alkyl, X- is halo or triflate; ii. Phosphorylating compound of Formula (III) with PO(OR 5 )Cl to obtain the compound of Formula (IV) wherein R3, R4, R5, R6 and R7 are independently alkyl; iii. Reducing compound (IV) with dithionite under ice-cold conditions to obtain compound V; wherein R3, R4, R6 and R7 are independently alkyl; and iv. Deprotecting the compound of Formula (V) and purifying to obtain reduced nicotinamide mononucleotide of formula (I).
- the X- anion in the compound of Formula (II), (IIa), (III) and (IV) is preferably chloro. Accordingly, in the step (i) of the process of the present invention as shown above, the ketalization of compound (II) to compound (III) is carried out using the ketalizing agent selected from cyclic or acyclic ketones such as 2,2dimethoxy propane (2,2 DMP), 2,2 diethoxy propane, acetone, and the like.
- the ketalization process step (i) is catalyzed by acid, wherein the acid catalyst can be an inorganic acid catalyst such as sulfuric acid, hydrochloric acid, phosphoric acid, oleum and the like alone or mixtures thereof.
- the acid catalyst can be an organic acid catalyst, for example, p-toluenesulfonic acid, methylsulfonic acid, trifluoromethylsulfonic acid, and the like alone or mixtures thereof.
- the acid catalyst is a mixture of sulfuric acid and oleum.
- the solvent can be any suitable solvent such as acetonitrile, dichloromethane, acetone, dimethylformamide, dimethylsulfoxide, and the like alone or mixture thereof.
- the solvent is acetonitrile.
- the step of ketalization is performed at a suitable temperature ranging between 0 °C to 25°C
- the phosphorylation of compound (III) to compound (IV) is carried out using phosphorylating agent selected from suitable phosphorylating agent, preferably the phosphorylating agent is di-tert-butylchlorophosphate.
- the phosphorylation is carried out in presence of base selected from organic or inorganic base and in suitable solvent selected from polar or non-polar, protic or aprotic solvent alone or mixtures thereof.
- the reaction is carried out at a suitable temperature ranging between 0°C to 25°C
- the step (iii) of the process which comprises reduction of the salt of compound (IV) to the base (compound of formula V) is carried out in presence of reducing agent such as sodium dithionite at alkaline pH using a suitable base and under ice- cold conditions.
- the solvent is selected from water, acetates, ethers, alcohols, hydrocarbons and the like alone or mixtures thereof.
- the step (iv) of deprotection of compound (V) to NMNH is conducted preferably in methanol in the presence of HCl or formic acid or TFA catalyst.
- the reduced nicotinamide mononucleotide (NMNH) obtained by the process of the present invention is further isolated and purified from the solvent methanol.
- the present invention relate to a process for synthesis of crystalline NMNH (I) comprising; i. Ketalizing nicotinamide riboside chloride (NRCl) (II) with 2,2DMP in presence of H2SO4 and oleum in acetonitrile to obtain the intermediate compound (III); ii. Phosphorylating the compound (III) of step (i) with di-tert- butylchlorophosphate in presence of base and solvent to obtain the intermediate compound (IV); iii.
- the present invention discloses a process for synthesis of crystalline form of reduced nicotinamide mononucleotide (NMN-H) of formula (I); comprising; i. Adding portion wise sodium dithionite as reducing agnet in alkaline pH and under ice –cold conditions to the compound of Formula (IIa) wherein R is–P(O)R1R2 R1 and R2 independently represent OH and O-respectively; and X- is halo; ii. Concentrating the mixture of step (i) to half its volume by evaporation; iii. Cooling the temperature to 0 o C and precipitating the mixture from the solvent; iv.
- step (iii) Filtering the precipitate of step (iii), concentrating the filtrate followed by dissolving it in DI water and solvent at 0 o C until precipitation of salts from the filtrate; and v. Concentrating the filtrate and crystallizing to obtain desired product.
- base selected from suitable bases which include inorganic bases such as alkali and alkaline earth metal bases, e.g., those containing metallic cations such as sodium, potassium, magnesium, calcium and the like alone or mixtures thereof, organic base such as ethylamine, pyridine and the like followed by portion wise addition of the reducing agent such as sodium dithionite and allowing to stir at the same temperature. This is followed by concentrating the reaction mixture to half its volume and precipitating the mass from the mixture of solvents.
- suitable bases include inorganic bases such as alkali and alkaline earth metal bases, e.g., those containing metallic cations such as sodium, potassium, magnesium, calcium and the like alone or mixtures thereof, organic base such as ethylamine, pyridine and the like followed by portion wise addition of the reducing agent such as sodium dithionite and allowing to stir at the same temperature. This is followed by concentrating the reaction mixture to half its volume and precipitating the mass from the mixture of solvents.
- inorganic bases such as
- the precipitate is further washed, filtered and the filtrate is concentrated under vacuum to obtain crystalline NMNH of Formula (I).
- the solvent used in the process can be any suitable solvent selected from polar or non-polar, protic or aprotic solvent such as water, lower alcohols, ketone, ethers, acetates and such like alone or mixtures thereof.
- the present invention relates to a process for synthesis of crystalline reduced nicotinamide mononucleotide (NMNH) of formula (I) from nicotinamide mononucleotide (NMN) of formula (Ia) comprising; i.
- NMN nicotinamide mononucleotide
- IIa nicotinamide mononucleotide
- step (i) Concentrating the mixture of step (i) to half its volume by evaporation; iii. Cooling the temperature to 0 o C and precipitating the mixture from the solvent; iv. Filtering the precipitate of step (iii) and concentrating the filtrate followed by dissolving it in DI water and then addition of the solvent at 0 o C until precipitation of salts from the filtrate; and v. Concentrating the filtrate under reduced pressure followed by purification in methanol to obtain NMNH.
- NPN nicotinamide mononucleotide
- the process is depicted in Scheme 2 below:
- the crystalline reduced nicotinamide mononucleotide (NMNH) of formula I obtained by the process of the present invention is further characterized by chromatogram, Mass spectra, as shown in the figures 2 and 3.
- the process of the present invention avoids the use of costly chromatographic techniques used in the art, which makes the present process cost effective and industrially viable.
- the crystalline reduced nicotinamide mononucleotide (NMNH) of formula I is stable at 2-8oC for a period of 12-24months.
- the pharmaceutical composition and/or nutraceutical/dietary supplement compositions comprises the crystalline NMNH (I) prepared by the process of the present invention is provided herein.
- the pharmaceutical/nutraceutical/ dietary supplement composition may be formulated using suitable formulating excipients, adjuvants in appropriate amounts.
- the nutraceutical/dietary supplement composition may be in the form of powder, granules, pellets, syrups, suspensions and such suitable forms which may be taken along with food.
- the pharmaceutical composition may conveniently be provided in unit dosage form and may be prepared by any methods well known in the art. The amount of active ingredient which can be combined with a carrier material to produce a single dosage form will vary depending upon the host being treated, the particular mode of administration.
- Example 1 Synthesis of crystalline reduced Nicotinamide Mononucleotide (Formula I) from Nicotinamide Riboside Chloride (NRCl) (Formula II)
- Example 1a Preparation of compound (III) from Nicotinamide Riboside Chloride (NRCl) Formula (II)
- H2SO4 4.3 mL, 80 mmol
- Example 1b Preparation of compound (IV) through the phosphorylation of Formula III Compound-III (15g, 50.79 mmol) was dissolved in 100 mL DCM at 0 o C followed by addition of Et3N (14.15 mL, 101.59 mmol) and DMAP (1.24g, 10.15 mmol).
- Example 1c Preparation of compound (V) by the reduction of IV
- the compound-IV 14g, 28.71 mmol
- EtOAc 200 mL
- 65 mL sat.NaHCO3 solution were added, followed by addition of solid sodium dithionite (26.24g, 150.77mmol) and 35 mL of water at stirring and at room temperature.
- the biphasic reaction mixture was stirred at room temperature for 6h, and then EtOAc (200 mL) was added.
- the organic phase was separated, and evaporated under reduced pressure to give the reduced form of compound V.
- Example 1d Preparation of NMNH (I) from compound of Formula V.
- Example 2 Preparation of Crystalline Reduced ((2R,3S,4R)-5-(3- carbamoylpyridin-1(4H)-yl)-3,4-dihydroxytetrahydrofuran-2-yl)methyl di hydrogen phosphate (NMNH) from Nicotinamide Mononucleotide (NMN) (Formula IIa) To the stirred solution of NMN (8.15g, 1 equiv, 24.38 mmol) in 100 mL deionized water at 0 o C was added saturated NaHCO 3 solution (200 mL) in a single portion.
- NMN Nicotinamide Mononucleotide
- Example 3 Characterization of Crystalline NMNH and NMN Powder X-ray diffraction studies: Powder X-ray diffraction studies were carried out in Bragg–Brentano (reflection) geometry on a Bruker D8 Advance Davinci diffractometer and D2 Phaser diffractometers equipped with a Cu X-ray source (1.5418 ⁇ ) and an LYNXEYE- XET high-resolution position-sensitive detectors. The samples were placed into the plate sample holders and rotated at a rate of 15 rpm during the data acquisition.
- Powder X-RD of NMN 7.559 o , 11.434 o , 12.556 o , 15.947 o , 18.065 o , 18.870 o , 19.039 o , 20.027 o , 20.707 o, 20.914 o , 21.717 o , 22.962 o , 23.238 o , 25.387 o , 25.612 o , 26.491 o , 29.646 o , 30.244 o , 30.898 o ⁇ 2 ⁇ Powder X-RD of NMNH 19.112 o , 23.024 o , 25.340 o , 31.986 o , 33.569 o , 47.050 o ⁇ 2 ⁇ .
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Biochemistry (AREA)
- Biotechnology (AREA)
- General Health & Medical Sciences (AREA)
- Genetics & Genomics (AREA)
- Molecular Biology (AREA)
- Saccharide Compounds (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
The present invention discloses a novel crystalline form of reduced nicotinamide mononucleotide (NMNH) of formula (I) and to the process for preparation thereof.
Description
CRYSTALLINE REDUCED NICOTINAMIDE MONONUCLEOTIDE (NMNH) AND PROCESS THEREOF FIELD OF THE INVENTION: The present invention relates to a novel crystalline form of reduced nicotinamide mononucleotide (NMNH) of formula (I) and to the process for preparation thereof. BACKGROUND OF THE INVENTION: Nicotinamide adenine dinucleotide (NAD+) is essential to living organisms since it participates in various biological reactions and regulates key biological processes, such as metabolism and DNA repair. Nicotinamide adenine dinucleotide (NAD+) homeostasis is constantly compromised due to degradation by NAD+ -dependent enzymes which lead to various age related diseases. NAD+ replenishment by supplementation with the NAD+ precursors nicotinamide mononucleotide (NMN) is thought to alleviate this imbalance. However, NMN both have limitations due to their mild effect on the cellular NAD+ pool and hence requires considerably higher doses which is costly. Recent studies have revealed that reduced nicotinamide mononucleotide (NMNH) is a potent NAD precursor than NMN both in vitro and in vivo. Moreover, NMNH increases cellular NADH levels, suppresses glycolysis and TCA cycle, as well as cell growth. Hence, the researchers worldwide have focused on NMNH and its production on large scale such that NMNH would be available at lower prices than the precursors currently available.
Article titled “Reduced Nicotinamide Mononucleotide (NMNH) Potently Enhances NAD+, Suppresses Glycolysis, TCA Cycle and CellGrowth” by Yan Liu et al published in November 2020, disclose synthesis of NMNH by reduction of NMN using thiourea dioxide (TDO) as shown in the scheme below:
NMNH obtained by the process described above requires further purification by HPLC using an amide column which adds to the cost of the process. NMNH chemically known as ((2R,3S,4R)-5-(3-carbamoylpyridin-1(4H)-yl)-3,4- dihydroxytetrahydrofuran-2-yl)methyl dihydrogen phosphate is the reduced form of NMN with the molecular formula of C11H17N2O8P, is stable under alkaline pH and low temperature conditions, but is unstable at neutral pH in solution. In the article titled “Proton Magnetic Resonance Study of the intramolecular Association and Confirmation of the alpha- and beta-Nicotine Mononucleotides and Nucleosides” published in Biochemistry, 15(18), 3981-3983, disclose preparation of alpha-NMNH by dithionite reduction of alpha-NMN. It is known that aqueous solution of dithionites is unstable at ambient temperature conditions and hence could lead to undesirable formation of impurities. WO2017059249A1 relates to crystalline forms of a β-nicotinamide mononucleotide, methods of their preparation, and related pharmaceutical preparations thereof.
Given the therapeutic benefits as nutraceuticals associated with NMN-H, the present inventors envisaged the need in the art to provide novel crystalline form of NMN-H and to the process for synthesis thereof which is efficient, industrially scalable and provides NMNH in high yield and purity. SUMMARY OF THE INVENTION: In accordance with the above, the present invention provides crystalline form of reduced nicotinamide mononucleotide (NMNH) of the formula (I)
Characterized by PXRD 19.112 o, 23.024 o, 25.340 o, 31.986 o, 33.569 o, 47.050 o by 1H NMR (400 MHz, D2O) δ 7.06 (s, 1H), 6.14 (dd, J = 8.2, 1.4 Hz, 1H), 4.97 – 4.89 (m, 1H), 4.80 (d, J = 7.4 Hz, 1H), 4.24 (dd, J = 7.3, 5.6 Hz, 1H), 4.15 (dd, J = 5.4, 2.2 Hz, 1H), 4.00 (d, J = 1.5 Hz, 1H), 3.78 – 3.73 (m, 2H), 2.98 (d, J = 1.4 Hz, 2H). 13C NMR (101 MHz, D2O) δ 173.02, 138.32, 124.90, 105.46, 100.69, 94.98, 83.17, 83.09, 70.73, 70.65, 63.95, 63.91, 22.02.31P NMR (162 MHz, D2O) δ 3.88.Ms: m/z = 337.11 (M+H). In an aspect, the crystalline form of NMNH of formula (I) is in anhydrous powder form. In another aspect, the present invention provides a process for synthesis of crystalline form of reduced nicotinamide mononucleotide (NMNH) of Formula I
(I) from the compound of general Formula (II) or (IIa);
(II) or (IIa) wherein R is selected from hydrogen or –P(O)R1R2; R1 and R2 independently represent OH and O-respectively; and X- is halo, or triflate. comprising; i. Ketalizing the compound of Formula II,
wherein R is hydrogen; X- is halo or triflate to a compound of Formula III
wherein R3 and R4 are alkyl, X- is halo or triflate ; ii. Phosphorylating compound of Formula (III) with PO(OR5)Cl to obtain the compound of Formula (IV)
wherein R3, R4, R5, R6 and R7 are independently alkyl, X- is halo or triflate; iii. Reducing compound (IV) with dithionite under ice-cold conditions to obtain compound V;
wherein R3, R4, R6 and R7 are independently alkyl; and iv. Deprotecting the compound of Formula (V) and purifying in methanol to obtain the powder form of reduced nicotinamide mononucleotide of formula (I). OR
i. Reacting the compound of Formula (IIa) with sodium dithionite as reducing agent in alkaline pH and under ice –cold conditions
wherein R is–P(O)R1R2 R1 and R2 independently represent OH and O-respectively; and X- is halo or triflate; ii. Concentrating, crystallizing from suitable solvent and filtering; and iii. Concentrating the filtrate of step (ii) under reduced pressure to obtain crystalline reduced nicotinamide mononucleotide of formula (I). In another aspect, the present invention provides a composition comprising the crystalline compound of reduced nicotinamide mononucleotide of Formula (I) with suitable excipients. DESCRIPTION OF THE FIGURES Figure 1 depict the chromatogram of as prepared NMNH. Figure 2 depict the Masss pectra of as prepared NMNH Figure 3 depict the Powder X-RD of NMN Figure 4 depict the Powder X-RD of NMNH DETAILED DESCRIPTION OF THE INVENTION: The present invention will now be described in detail in its preferred and optional embodiments so that various aspects of the invention will be fully understood without limiting the scope of the invention.
In an embodiment, the present invention relates to a crystalline form of reduced nicotinamide mononucleotide (NMNH) of the formula (I)
characterized by PXRD 19.112 o, 23.024 o, 25.340 o, 31.986 o, 33.569 o, 47.050 o±2θ; by 1H NMR (400 MHz, D2O) δ 7.06 (s, 1H), 6.14 (dd, J = 8.2, 1.4 Hz, 1H), 4.97 – 4.89 (m, 1H), 4.80 (d, J = 7.4 Hz, 1H), 4.24 (dd, J = 7.3, 5.6 Hz, 1H), 4.15 (dd, J = 5.4, 2.2 Hz, 1H), 4.00 (d, J = 1.5 Hz, 1H), 3.78 – 3.73 (m, 2H), 2.98 (d, J = 1.4 Hz, 2H). 13C NMR (101 MHz, D2O) δ 173.02, 138.32, 124.90, 105.46, 100.69, 94.98, 83.17, 83.09, 70.73, 70.65, 63.95, 63.91, 22.02.31P NMR (162 MHz, D2O) δ 3.88.Ms: m/z = 337.11 (M+H). In another embodiment, the present invention discloses a process for synthesis of crystalline form of reduced nicotinamide mononucleotide (NMNH)of Formula I
from the compound of general Formula (II) or (IIa) ;
(II) or (IIa) wherein R is selected from hydrogen or –P(O)R1R2; R1 and R2 independently represent OH and O-respectively; and X- is halo or triflate. comprising; i. Ketalizing the compound of Formula II,
wherein R is hydrogen; X- is halo or triflate; to a compound of Formula III
wherein R3 and R4 are alkyl, X- is halo or triflate; ii. Phosphorylating compound of Formula (III) with PO(OR5)Cl to obtain the compound of Formula (IV)
wherein R3, R4, R5, R6 and R7 are independently alkyl, X- is halo or triflate; iii. Reducing compound (IV) with dithionite under ice-cold conditions to obtain compound V;
wherein R3, R4, R6 and R7 are independently alkyl; and iv. Deprotecting the compound of Formula (V) and purifying in methanol to obtain reduced nicotinamide mononucleotide of formula (I). OR i. Reacting the compound of Formula (IIa) with sodium dithionite as reducing agent in alkaline pH and under ice –cold conditions
wherein R is–P(O)R1R2 R1 and R2 independently represent OH and O-respectively; and X- is halo or triflate;
ii. Concentrating, crystallizing from suitable solvent and filtering; and iii. Concentrating the filtrate of step (ii) under reduced pressure to obtain crystalline reduced nicotinamide mononucleotide of formula (I). In an embodiment, the present invention relates to a process for synthesis of crystalline form of reduced nicotinamide mononucleotide (NMNH) of Formula I
from the compound of general Formula (II);
Wherein R is selected from hydrogen or –P(O)R1R2; R1 and R2 independently represent OH and O- respectively; and X- is halo or triflate. Comprising; i. Ketalizing the compound of Formula II,
wherein R is hydrogen; X- is halo or triflate to a compound of Formula III
wherein R3 and R4 are independently alkyl, X- is halo or triflate; ii. Phosphorylating compound of Formula (III) with PO(OR5)Cl to obtain the compound of Formula (IV)
wherein R3, R4, R5, R6 and R7 are independently alkyl; iii. Reducing compound (IV) with dithionite under ice-cold conditions to obtain compound V;
wherein R3, R4, R6 and R7 are independently alkyl; and iv. Deprotecting the compound of Formula (V) and purifying to obtain reduced nicotinamide mononucleotide of formula (I). The X- anion in the compound of Formula (II), (IIa), (III) and (IV) is preferably chloro.
Accordingly, in the step (i) of the process of the present invention as shown above, the ketalization of compound (II) to compound (III) is carried out using the ketalizing agent selected from cyclic or acyclic ketones such as 2,2dimethoxy propane (2,2 DMP), 2,2 diethoxy propane, acetone, and the like. The ketalization process step (i) is catalyzed by acid, wherein the acid catalyst can be an inorganic acid catalyst such as sulfuric acid, hydrochloric acid, phosphoric acid, oleum and the like alone or mixtures thereof. The acid catalyst can be an organic acid catalyst, for example, p-toluenesulfonic acid, methylsulfonic acid, trifluoromethylsulfonic acid, and the like alone or mixtures thereof. In a preferred embodiment, the acid catalyst is a mixture of sulfuric acid and oleum. The solvent can be any suitable solvent such as acetonitrile, dichloromethane, acetone, dimethylformamide, dimethylsulfoxide, and the like alone or mixture thereof. Preferably, the solvent is acetonitrile. The step of ketalization is performed at a suitable temperature ranging between 0 °C to 25°C In the process step (ii), the phosphorylation of compound (III) to compound (IV) is carried out using phosphorylating agent selected from suitable phosphorylating agent, preferably the phosphorylating agent is di-tert-butylchlorophosphate. The phosphorylation is carried out in presence of base selected from organic or inorganic base and in suitable solvent selected from polar or non-polar, protic or aprotic solvent alone or mixtures thereof. The reaction is carried out at a suitable temperature ranging between 0°C to 25°C The step (iii) of the process which comprises reduction of the salt of compound (IV) to the base (compound of formula V) is carried out in presence of reducing agent such as sodium dithionite at alkaline pH using a suitable base and under ice-
cold conditions. The solvent is selected from water, acetates, ethers, alcohols, hydrocarbons and the like alone or mixtures thereof. The step (iv) of deprotection of compound (V) to NMNH is conducted preferably in methanol in the presence of HCl or formic acid or TFA catalyst. The reduced nicotinamide mononucleotide (NMNH) obtained by the process of the present invention is further isolated and purified from the solvent methanol. In a preferred embodiment, the present invention relate to a process for synthesis of crystalline NMNH (I) comprising; i. Ketalizing nicotinamide riboside chloride (NRCl) (II) with 2,2DMP in presence of H2SO4 and oleum in acetonitrile to obtain the intermediate compound (III); ii. Phosphorylating the compound (III) of step (i) with di-tert- butylchlorophosphate in presence of base and solvent to obtain the intermediate compound (IV); iii. Reducing the compound (IV) of step (ii) with sodium dithionite at alkaline pH and under ice cold conditions to obtain the compound (V); and iv. Deprotecting the compound of formula (IV) obtained instep (iii) and crystallizing to obtain the crystalline NMNH compound (I). The process is depicted in Scheme 1 below:
Scheme 1 In yet another embodiment, the present invention discloses a process for synthesis of crystalline form of reduced nicotinamide mononucleotide (NMN-H) of formula (I);
comprising; i. Adding portion wise sodium dithionite as reducing agnet in alkaline pH and under ice –cold conditions to the compound of Formula (IIa)
wherein R is–P(O)R1R2
R1 and R2 independently represent OH and O-respectively; and X- is halo; ii. Concentrating the mixture of step (i) to half its volume by evaporation; iii. Cooling the temperature to 0oC and precipitating the mixture from the solvent; iv. Filtering the precipitate of step (iii), concentrating the filtrate followed by dissolving it in DI water and solvent at 0oC until precipitation of salts from the filtrate; and v. Concentrating the filtrate and crystallizing to obtain desired product. Accordingly, to the compound of formula (IIa), which is nicotinamide mononucleotide (NMN), dissolved in DI water and cooled to 0°C under an ice- bath, is added base selected from suitable bases which include inorganic bases such as alkali and alkaline earth metal bases, e.g., those containing metallic cations such as sodium, potassium, magnesium, calcium and the like alone or mixtures thereof, organic base such as ethylamine, pyridine and the like followed by portion wise addition of the reducing agent such as sodium dithionite and allowing to stir at the same temperature. This is followed by concentrating the reaction mixture to half its volume and precipitating the mass from the mixture of solvents. The precipitate is further washed, filtered and the filtrate is concentrated under vacuum to obtain crystalline NMNH of Formula (I). The solvent used in the process can be any suitable solvent selected from polar or non-polar, protic or aprotic solvent such as water, lower alcohols, ketone, ethers, acetates and such like alone or mixtures thereof. In another preferred embodiment the present invention relates to a process for synthesis of crystalline reduced nicotinamide mononucleotide (NMNH) of formula (I) from nicotinamide mononucleotide (NMN) of formula (Ia) comprising;
i. Adding portion wise sodium dithionite to the stirred mixture of nicotinamide mononucleotide (NMN)of formula (IIa) and the base at 0oC and maintaining the mixture at 0oCwithout stirring; ii. Concentrating the mixture of step (i) to half its volume by evaporation; iii. Cooling the temperature to 0oC and precipitating the mixture from the solvent; iv. Filtering the precipitate of step (iii) and concentrating the filtrate followed by dissolving it in DI water and then addition of the solvent at 0oC until precipitation of salts from the filtrate; and v. Concentrating the filtrate under reduced pressure followed by purification in methanol to obtain NMNH. The process is depicted in Scheme 2 below: In an embodiment, the crystalline reduced nicotinamide mononucleotide (NMNH) of formula I obtained by the process of the present invention is further characterized by chromatogram, Mass spectra, as shown in the figures 2 and 3. In an embodiment, the process of the present invention avoids the use of costly chromatographic techniques used in the art, which makes the present process cost effective and industrially viable. In an embodiment, the crystalline reduced nicotinamide mononucleotide (NMNH) of formula I is stable at 2-8ºC for a period of 12-24months.
In another embodiment, the pharmaceutical composition and/or nutraceutical/dietary supplement compositions comprises the crystalline NMNH (I) prepared by the process of the present invention is provided herein. The pharmaceutical/nutraceutical/ dietary supplement composition may be formulated using suitable formulating excipients, adjuvants in appropriate amounts. The nutraceutical/dietary supplement composition may be in the form of powder, granules, pellets, syrups, suspensions and such suitable forms which may be taken along with food. The pharmaceutical composition may conveniently be provided in unit dosage form and may be prepared by any methods well known in the art. The amount of active ingredient which can be combined with a carrier material to produce a single dosage form will vary depending upon the host being treated, the particular mode of administration. The amount of active ingredient that can be combined with a carrier material to produce a single dosage form will generally be that amount of the compound which produces a therapeutic effect. Experimental: Example 1: Synthesis of crystalline reduced Nicotinamide Mononucleotide (Formula I) from Nicotinamide Riboside Chloride (NRCl) (Formula II) Example 1a: Preparation of compound (III) from Nicotinamide Riboside Chloride (NRCl) Formula (II) In a flame dried flask under an argon atmosphere, H2SO4 (4.3 mL, 80 mmol) was slowly added to dry acetonitrile (250 mL) at 0°C. After 5 minutes, 2,2- dimethoxypropane (144 mL, 1175.3 mmol) was added to the stirred acetonitrile solution at the same temperature. NR chloride (30.0 g, 117.5 mmol) was added to the reaction mixture at 0°C, and the reaction was warmed up to room temperature. The progress of the reaction was monitored by thin-layer chromatography (TLC). The complete conversion was observed within 2h. The reaction mixture was cooled again to 0°C in an ice bath and quenched with solid Na2CO3 (6.4 g, 60.0 mmol) and stirred for another 1h followed by slow addition of 3 mL of water.
Upon complete neutralization of acid (pH = 6–7), the residual solids were filtered off, and the filtrate was concentrated under reduced pressure. The crude product was dissolved in a minimum volume of DCM and purified by silica gel chromatography (60 Å) using DCM/MeOH (9:1) as an eluent to obtain compound-III as a white solid. Example 1b: Preparation of compound (IV) through the phosphorylation of Formula III Compound-III (15g, 50.79 mmol) was dissolved in 100 mL DCM at 0oC followed by addition of Et3N (14.15 mL, 101.59 mmol) and DMAP (1.24g, 10.15 mmol). The resulting mixture was bought to room temperature and stirred for 1h and then a solution of di-tert-butyl chloro phosphate (17.42g, 76.19 mmol) in 25 mL DCM was added dropwise over 10 min. After complete addition, the reaction mixture was stirred for 24h at room temperature and then concentrated and purified over silica gel chromatography to yield NRCl (Compound-IV). Example 1c: Preparation of compound (V) by the reduction of IV In a round bottom flask flushed with nitrogen, the compound-IV (14g, 28.71 mmol) was dissolved in a nitrogen-purged EtOAc (200 mL) and 65 mL sat.NaHCO3 solution were added, followed by addition of solid sodium dithionite (26.24g, 150.77mmol) and 35 mL of water at stirring and at room temperature. The biphasic reaction mixture was stirred at room temperature for 6h, and then EtOAc (200 mL) was added. The organic phase was separated, and evaporated under reduced pressure to give the reduced form of compound V. Example 1d: Preparation of NMNH (I) from compound of Formula V. To a stirred solution of compound-V (5.0 g) in methanol (3 vol) in 250 mL round bottom flask at 0 °C was added anhydrous HCl gas (3 equiv). The resulting solution was stirred at 25°C over 24h. After complete conversion as indicated by TLC (3:7, MeOH:DCM), the solvent was concentrated to half of its volume by evaporation on rotavaporator at 40oC. The reaction mixture was then bought to
0oC and then 250 mL methanol was added slowly resulting in the formation of precipitate. The reaction mixture was stirred for another 3h for maximum precipitation. After 3h, the precipitate was filtered and dissolved in 100 mL distilled water. To this solution, 250 mL acetone was slowly added at 0oC and stirred for one hour. The precipitate formed was filtered and washed with 50mL chilled acetone. To the filtrate, 250 mL acetone was added and stirred for 6h for further precipitation of inorganic salts. The precipitate was filtered and the filtrate was concentrated under reduced pressure to provide the desired NMNH as a light yellow solid. Example 2: Preparation of Crystalline Reduced ((2R,3S,4R)-5-(3- carbamoylpyridin-1(4H)-yl)-3,4-dihydroxytetrahydrofuran-2-yl)methyl di hydrogen phosphate (NMNH) from Nicotinamide Mononucleotide (NMN) (Formula IIa) To the stirred solution of NMN (8.15g, 1 equiv, 24.38 mmol) in 100 mL deionized water at 0oC was added saturated NaHCO3 solution (200 mL) in a single portion. After stirring for five minutes, Na2S2O4 (14.85g, 3.5 equiv) was added in portion wise and the reaction mixture was stirred for one hour at the same temperature. Then a second portion of Na2S2O4 (6.36g, 1.5 equiv) was added and the contents of the reaction mixture was further stirred for two hours at 0oC. Purification: The reaction mixture was then placed at 0oC for 6h without stirring. After 6h, the reaction mixture was concentrated to half of its volume by evaporation on rotavaporator at 40oC. The reaction mixture was then bought to 0oC followed by slow addition of 250 mL methanol resulting in the formation of precipitate. The reaction mixture was stirred for 3h for maximum precipitation. After 3h, the precipitate was filtered and then dissolved in 100 mL distilled water. To this solution, 250 mL acetone was slowly added at 0oC and stirred for one hour. The precipitate formed was filtered and washed with 50 mL chilled acetone. To the
filtrate, 250 mL acetone was added and stirred for 6h for further precipitation of inorganic salts. The precipitate was filtered and the filtrate was concentrated under reduced pressure to yield NMNH (Weight- 6.2g, Yield- 83.6%, HPLC-94.5%). Example 3: Characterization of Crystalline NMNH and NMN Powder X-ray diffraction studies: Powder X-ray diffraction studies were carried out in Bragg–Brentano (reflection) geometry on a Bruker D8 Advance Davinci diffractometer and D2 Phaser diffractometers equipped with a Cu X-ray source (1.5418 Å) and an LYNXEYE- XET high-resolution position-sensitive detectors. The samples were placed into the plate sample holders and rotated at a rate of 15 rpm during the data acquisition. The X-rays were generated with 40 kV and 30 mA generator settings and the measurement was performed on the finely ground powder sample with a step size of 0.02° and step time of 1s across the 2θ range 2-40° and 4-30°. The diffractogram was processed using Bruker DIFFRAC.EVA version 4.0 software. Powder X-RD of NMN: 7.559o, 11.434 o, 12.556 o, 15.947 o, 18.065 o, 18.870 o, 19.039 o, 20.027 o, 20.707 o, 20.914 o, 21.717 o, 22.962 o, 23.238 o, 25.387 o, 25.612 o, 26.491 o, 29.646 o, 30.244 o, 30.898 o±2θ Powder X-RD of NMNH 19.112 o, 23.024 o, 25.340 o, 31.986 o, 33.569 o, 47.050 o±2θ. NMR of NMNH 1H NMR (400 MHz, D2O) δ 7.06 (s, 1H), 6.14 (dd, J = 8.2, 1.4 Hz, 1H), 4.97 – 4.89 (m, 1H), 4.80 (d, J = 7.4 Hz, 1H), 4.24 (dd, J = 7.3, 5.6 Hz, 1H), 4.15 (dd, J = 5.4, 2.2 Hz, 1H), 4.00 (d, J = 1.5 Hz, 1H), 3.78 – 3.73 (m, 2H), 2.98 (d, J = 1.4 Hz, 2H). 13C NMR (101 MHz, D2O) δ 173.02, 138.32, 124.90, 105.46, 100.69, 94.98, 83.17, 83.09, 70.73, 70.65, 63.95, 63.91, 22.02.31P NMR (162 MHz, D2O) δ 3.88.Ms: m/z = 337.11 (M+H). It will be understood that the above description is intended to be illustrative and not restrictive. The embodiments will be apparent to those in the art upon
reviewing the above description. The scope of the invention should therefore, be determined not with reference to the above description but should instead be determined by the appended claims along with full scope of equivalents to which such claims are entitled.
Claims
Claims 1. Crystalline reduced nicotinamide mononucleotide (NMNH) of the formula (I)
Formula I Characterized by PXRD 19.112 o, 23.024 o, 25.340 o, 31.986 o, 33.569 o, 47.050 oby 1H NMR (400 MHz, D2O) δ 7.06 (s, 1H), 6.14 (dd, J = 8.2, 1.4 Hz, 1H), 4.97 – 4.89 (m, 1H), 4.80 (d, J = 7.4 Hz, 1H), 4.24 (dd, J = 7.3, 5.6 Hz, 1H), 4.15 (dd, J = 5.4, 2.2 Hz, 1H), 4.00 (d, J = 1.5 Hz, 1H), 3.78 – 3.73 (m, 2H), 2.98 (d, J = 1.4 Hz, 2H). 13C NMR (101 MHz, D2O) δ 173.02, 138.32, 124.90, 105.46, 100.69, 94.98, 83.17, 83.09, 70.73, 70.65, 63.95, 63.91, 22.02.31P NMR (162 MHz, D2O) δ 3.88.Ms: m/z = 337.11 (M+H).
2. The crystalline NMNH as claimed in claim 1, wherein the crystalline NMNH is anhydrous.
3. The crystalline NMNH as claimed in claim 1, wherein said crystalline NMNH is prepared from the compound of Formula (Ia) or Formula (IIa)
Formula (II) or (IIa) wherein R is selected from hydrogen or –P(O)R1R2; R1 and R2 independently represent OH and O-respectively; and
X- is halo or triflate comprising; i. Ketalizing the compound of Formula II,
wherein R is hydrogen; X- is halo or triflate; to a compound of Formula III
wherein R3 and R4 are alkyl, X- is halo or triflate; ii. Phosphorylating compound of Formula (III) with PO(OR5)Cl to obtain the compound of Formula (IV)
wherein R3, R4, R5, R6 and R7 are independently alkyl, X- is halo or triflate; iii. Reducing compound (IV) with dithionite under ice-cold conditions to obtain compound V;
wherein R3, R4, R6 and R7 are independently alkyl; and iv. Deprotecting the compound of Formula (V) and purifying in methanol to obtain reduced nicotinamide mononucleotide of formula (I). OR iv. Reacting the compound of Formula (IIa) with the reducing agent selected from sodium dithionite at alkaline pH and under ice –cold conditions
wherein R is–P(O)R1R2 R1 and R2 independently represent OH and O-respectively; and X- is halo or triflate; v. Concentrating, crystallizing from suitable solvent and filtering; and vi. Concentrating the filtrate of step (ii) under reduced pressure to obtain crystalline reduced nicotinamide mononucleotide of formula (I).
4. The crystalline NMNH as claimed in claim 3, wherein the ketalization of compound (II) is carried out with the ketalizing agent selected from 2,2dimethoxy propane (2,2 DMP), 2,2 diethoxy propane, acetone, and the like.
5. The crystalline NMNH as claimed in claim 3, wherein the ketalization is catalyzed by acid selected from inorganic acids or organic acids.
6. The crystalline NMNH as claimed in claim 5, wherein the inorganic acid is selected from sulfuric acid, hydrochloric acid, phosphoric acid, oleum and the like alone or mixtures thereof.
7. The crystalline NMNH as claimed in claim 5, wherein the organic acid is selected from p-toluenesulfonic acid, methylsulfonic acid, trifluoromethylsulfonic acid and the like alone or mixtures thereof.
8. The crystalline NMNH as claimed in claim 3, wherein the ketalization is carried out at a temperature ranging between 0ºC-25ºC.
9. The crystalline NMNH as claimed in claim 3, wherein the phosphorylation of compound (III) is carried out in presence of base and a solvent at a temperature ranging between 0ºC-25ºC.
10. The crystalline NMNH as claimed in claim 3, wherein the reduction of compound (IV) is carried out at alkaline pH.
11. The crystalline NMNH as claimed in claim 3, wherein the step of deprotection of compound (V) is carried out in a solvent in presence of HCl or formic acid or TFA.
12. The crystalline NMNH as claimed in claim 3, wherein the solvent for the process is selected from water, lower alcohols, ketones, halogenated hydrocarbons, nitriles, ethers, acetates and the like alone or mixtures thereof.
13. The crystalline NMNH as claimed in any one of the claims 1 to 12, wherein crystalline NMNH is prepared from nicotinamide riboside chloride (NRCl) of Formula II by the process comprising;
i. Ketalizing nicotinamide riboside chloride (NRCl) (II) with 2,2DMP in presence of H2SO4 and oleum in acetonitrile to obtain the intermediate compound (III); ii. Phosphorylating the compound (III) of step (i) with di-tert- butylchlorophosphate in presence of base and solvent to obtain the intermediate compound (IV); iii. Reducing the compound (IV) of step (ii) with sodium dithionite at alkaline pH and under ice cold conditions to obtain the compound (V); and iv. Deprotecting the compound of formula (IV) obtained instep (iii) and crystallizing to obtain the crystalline NMNH compound (I).
14. The crystalline NMNH as claimed in any one of the claims 3 to 12, wherein crystalline NMNH is prepared from the compound of Formula II (a) by the process comprising; i. Adding portion wise the reducing agent selected from sodium dithionite at alkaline pH and under ice –cold conditions to the compound of Formula (IIa)
wherein R is–P(O)R1R2 R1 and R2 independently represent OH and O-respectively; and X- is halo or triflate; ii. Concentrating the mixture of step (i) to half its volume by evaporation; iii. Cooling the temperature to 0oC and precipitating the mixture from the solvent;
iv. Filtering the precipitate of step (iii), concentrating the filtrate followed by dissolving it in DI water and solvent at 0oC until precipitation of salts from the filtrate; and v. Concentrating the filtrate of step (iv) and crystallizing to obtain desired product.
15. A pharmaceutical composition comprising crystalline NMNH as claimed in any one of the claims 1-14 along with pharmaceutically acceptable excipients.
16. Nutraceutical or Dietary composition comprising crystalline NMNH as claimed in any one of the claims 1-14 along with acceptable excipients.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| IN202321039042 | 2023-06-07 | ||
| IN202321039042 | 2023-06-07 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2024251966A1 true WO2024251966A1 (en) | 2024-12-12 |
Family
ID=91482030
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2024/065769 Ceased WO2024251966A1 (en) | 2023-06-07 | 2024-06-07 | Crystalline reduced nicotinamide mononucleotide (nmnh) and process thereof |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2024251966A1 (en) |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2017059249A1 (en) | 2015-10-02 | 2017-04-06 | Metrobiotech, Llc | Crystal forms of beta-nicotinamide mononucleotide |
| WO2021098725A1 (en) * | 2019-11-19 | 2021-05-27 | Tsinghua University | Synthetic method for nmn derivative and medical applications of nmn and its derivative |
| WO2021214299A1 (en) * | 2020-04-24 | 2021-10-28 | Nuvamid Sa | Nicotinamide mononucleotide and nicotinamide riboside derivatives and use thereof in the treatment of viral infections and respiratory complications, in particular caused by influenzavirus or coronavirus |
-
2024
- 2024-06-07 WO PCT/EP2024/065769 patent/WO2024251966A1/en not_active Ceased
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2017059249A1 (en) | 2015-10-02 | 2017-04-06 | Metrobiotech, Llc | Crystal forms of beta-nicotinamide mononucleotide |
| WO2021098725A1 (en) * | 2019-11-19 | 2021-05-27 | Tsinghua University | Synthetic method for nmn derivative and medical applications of nmn and its derivative |
| WO2021214299A1 (en) * | 2020-04-24 | 2021-10-28 | Nuvamid Sa | Nicotinamide mononucleotide and nicotinamide riboside derivatives and use thereof in the treatment of viral infections and respiratory complications, in particular caused by influenzavirus or coronavirus |
Non-Patent Citations (1)
| Title |
|---|
| "Proton Magnetic Resonance Study of the intramolecular Association and Confirmation of the alpha- and beta-Nicotine Mononucleotides and Nucleosides", BIOCHEMISTRY, vol. 15, no. 18, pages 3981 - 3983 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP2015205887A (en) | Process for the preparation of methyl {4,6-diamino-2- [1- (2-fluorobenzyl) -1H-pyrazolo [3,4-b] pyridin-3-yl] pyrimidin-5-yl} methylcarbamate and pharmaceutical Purification method for use as an active compound | |
| EP0222192A2 (en) | Nucleosides of 5-monofluoromethyluracil and 5-difluoromethyluracil | |
| KR20210005071A (en) | (Thio)nicotinamide ribofuranoside salt and composition thereof, preparation method, and use | |
| US4057548A (en) | Process for preparing methotrexate or an N-substituted derivative thereof and/or a di (lower) alkyl ester thereof and precursor therefor | |
| Uenishi et al. | Syntheses and antitumor activities of D-and L-2′-deoxy-4′-thio pyrimidine nucleosides | |
| JP2023513761A (en) | 2-[2-methylazetidin-1-yl]-4-phenyl-6-(trifluoromethyl)-pyrimidine compounds | |
| US4067867A (en) | Process for preparing pyrazine precursor of methotrexate or an N-substituted derivative thereof and/or a di(lower)alkyl ester thereof | |
| EP0235590A2 (en) | Improved process for the resolution of 1-aminoindanes | |
| CN111454312A (en) | A kind of preparation method of P2Y2 receptor agonist diquafosol tetrasodium | |
| WO2024251966A1 (en) | Crystalline reduced nicotinamide mononucleotide (nmnh) and process thereof | |
| JPWO2004106352A1 (en) | Method for producing aldohexopyranose intermediate | |
| ES3028534T3 (en) | Novel spirobicyclic intermediates | |
| JPH0797391A (en) | Nucleoside derivative and its production | |
| SU1272988A3 (en) | Method of producing ergolines or physiologically acceptable salts thereof | |
| JP2014156493A (en) | Production intermediate of tetrahydropyran compound | |
| Kuchař et al. | Synthesis of substituted 6-cyclopropylpurine bases and nucleosides by cross-coupling reactions or cyclopropanations | |
| JP3388489B2 (en) | Method for producing indolopyrrolocarbazole derivative, intermediate for producing the same, and method for producing the same | |
| EP0111299B1 (en) | Fluorine-containing uridine derivative and preparation and use thereof | |
| CS9692A3 (en) | Process for preparing 3-fluoropyrimidine nucleosides | |
| CA3150028A1 (en) | A method for preparing ivosidenib and an intermediate thereof | |
| JP7566783B2 (en) | Carba cyclic phosphatidic acid compounds | |
| CN120794985A (en) | 1,4,5, 6-Tetrahydropyridazine spiro-imidazoline-2-ketone compound, and synthetic method and application thereof | |
| HK40119630A (en) | Crystalline forms of nicotinamide ribofuranoside salts and nutritional supplements and pharmaceutical compositions comprising the same | |
| HK40119316A (en) | Method of making nicotinamide ribofuranoside salts, nicotinamide ribofuranoside salts as such, and uses thereof | |
| FEHR et al. | 2-EPIMUTALOMYCIN AND 28-EPIMUTALOMYCIN, TWO NEW POLYETHER ANTIBIOTICS FROM STREPTOMYCES MUTABILIS DERIVATIZATION OF MUTALOMYCIN AND THE STRUCTURE ELUCIDATION OF TWO MINOR METABOLITES |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24732441 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |