WO2024253070A1 - Temperature-responsive nonwoven fabric - Google Patents
Temperature-responsive nonwoven fabric Download PDFInfo
- Publication number
- WO2024253070A1 WO2024253070A1 PCT/JP2024/020258 JP2024020258W WO2024253070A1 WO 2024253070 A1 WO2024253070 A1 WO 2024253070A1 JP 2024020258 W JP2024020258 W JP 2024020258W WO 2024253070 A1 WO2024253070 A1 WO 2024253070A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- nonwoven fabric
- cells
- temperature
- less
- responsive
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F293/00—Macromolecular compounds obtained by polymerisation on to a macromolecule having groups capable of inducing the formation of new polymer chains bound exclusively at one or both ends of the starting macromolecule
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12M—APPARATUS FOR ENZYMOLOGY OR MICROBIOLOGY; APPARATUS FOR CULTURING MICROORGANISMS FOR PRODUCING BIOMASS, FOR GROWING CELLS OR FOR OBTAINING FERMENTATION OR METABOLIC PRODUCTS, i.e. BIOREACTORS OR FERMENTERS
- C12M3/00—Tissue, human, animal or plant cell, or virus culture apparatus
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M15/00—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment
- D06M15/19—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment with synthetic macromolecular compounds
- D06M15/21—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds
- D06M15/263—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds of unsaturated carboxylic acids; Salts or esters thereof
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M15/00—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment
- D06M15/19—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment with synthetic macromolecular compounds
- D06M15/21—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds
- D06M15/285—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds of unsaturated carboxylic acid amides or imides
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M2101/00—Chemical constitution of the fibres, threads, yarns, fabrics or fibrous goods made from such materials, to be treated
- D06M2101/16—Synthetic fibres, other than mineral fibres
- D06M2101/30—Synthetic polymers consisting of macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds
- D06M2101/32—Polyesters
Definitions
- the purpose of this patent is to provide a culture substrate that has high cell productivity per unit medium and enables the recovery of a cell suspension that does not contain fibrous impurities.
- the present inventors have conducted extensive research and found that a nonwoven fabric made of plastic with a true density of 1.0 to 1.5 g/ cm3 and a water absorption rate of less than 0.3%, the surface of which is coated with a block copolymer containing the following blocks (A), (B), and (C), has high cell productivity per unit medium and enables the recovery of a cell suspension free of fibrous impurities, thereby completing the present invention. That is, the present invention encompasses the following aspects.
- C A temperature-responsive polymer block having a lower critical solution temperature (LCST) in water in the range of 0°C to 50°C.
- ⁇ 2> The nonwoven fabric according to ⁇ 1>, characterized in that the water absorption rate is 0.2% or less.
- ⁇ 3> The nonwoven fabric according to ⁇ 1> or ⁇ 2>, characterized in that the plastic is polyethylene terephthalate.
- ⁇ 4> The nonwoven fabric according to any one of ⁇ 1> to ⁇ 3>, wherein the coating amount of the block copolymer is 1.0 to 100.0 ⁇ g/ cm2 .
- ⁇ 5> The nonwoven fabric according to any one of ⁇ 1> to ⁇ 4>, wherein the fibers constituting the nonwoven fabric have a thickness of 1 to 1000 ⁇ m.
- ⁇ 6> The nonwoven fabric according to any one of ⁇ 1> to ⁇ 5>, wherein the proportion of the block (C) in the block copolymer is 30 to 90 mol %.
- ⁇ 7> The nonwoven fabric according to any one of ⁇ 1> to ⁇ 6>, wherein the block copolymer has a number average molecular weight of 1,000 to 1,000,000.
- ⁇ 8> A method for producing the nonwoven fabric according to any one of ⁇ 1> to ⁇ 7>.
- a method for producing a nonwoven fabric according to any one of ⁇ 1> to ⁇ 7> comprising a step of coating a surface of a nonwoven fabric made of a plastic having a true density of 1.0 to 1.5 g/ cm3 and a water absorption rate of less than 0.3% with a block copolymer containing the following blocks (A), (B), and (C): (A) A polymer block having an HLB value (Griffin method) in the range of 7 or more and 20 or less. (B) A polymer block having an HLB value (Griffin method) in the range of 0 or more and less than 7.
- (C) A temperature-responsive polymer block having a lower critical solution temperature (LCST) in water in the range of 0°C to 50°C.
- LCST lower critical solution temperature
- ⁇ 10> The method according to ⁇ 9>, wherein the water absorption rate is 0.2% or less.
- ⁇ 11> A cell culture method using the nonwoven fabric according to any one of ⁇ 1> to ⁇ 7>.
- ⁇ 12> A cell culture method comprising a step of culturing cells in a medium containing the nonwoven fabric according to any one of ⁇ 1> to ⁇ 7>.
- ⁇ 13> The cell culture method according to ⁇ 11> or ⁇ 12>, further comprising a cooling cell detachment step of detaching cells from microcarriers using a cooling liquid containing a chelating agent.
- a nonwoven fabric made of plastic having a true density of 1.0 to 1.5 g/ cm3 and a water absorption rate of less than 0.3%, the surface of which is coated with a block copolymer containing the following blocks (A), (B), and (C), has high cell productivity per unit medium and can recover a cell suspension that does not contain fibrous impurities.
- B A polymer block having an HLB value (Griffin method) in the range of 0 or more and less than 7.
- C A temperature-responsive polymer block having a lower critical solution temperature (LCST) in water in the range of 0°C to 50°C.
- the present invention relates to a nonwoven fabric made of plastic having a true density of 1.0 to 1.5 g/ cm3 and a water absorption rate of less than 0.3%, the surface of which is coated with a block copolymer containing blocks (A), (B), and (C).
- nonwoven fabric is fabric made by bonding or intertwining fibers through heat, mechanical or chemical action.
- nonwoven fabric is fabric made by forming fibers into a cloth and bonding the fibers together.
- sheeting method used to form the nonwoven fabric typically examples include the dry method in which the fabric is formed in air, the wet method in which the fabric is formed in water, and the spunbond method.
- bonding method used to form bonds between the fibers typically examples include thermal bonding, chemical bonding, and mechanical bonding. As long as it is within the scope of the present invention, it can be selected appropriately depending on the ease of processing.
- the true density of the fibers constituting the nonwoven fabric is a density calculated from the volume excluding the gaps (pores) between the fibers constituting the nonwoven fabric, and is 1.0 to 1.5 g/cm 3 in order to settle in water, and is preferably 1.0 to 1.3 g/cm 3 , and more preferably 1.0 to 1.2 g/cm 3 in order to easily disperse under stirring.
- the water absorption rate of the fibers constituting the nonwoven fabric is less than 0.3% in order to maintain strength without decomposing in water, preferably 0.2% or less, more preferably 0.15% or less, and particularly preferably 0.1% or less.
- the method for evaluating the water absorption rate refers to JIS K 7209:2000, "Plastics - Determination of water absorption," which is a translation of ISO 62:1999, Plastics - Determination of water absorption.
- the material of the fibers constituting the nonwoven fabric is not particularly limited, but examples of materials having true densities and water absorption rates within the ranges of the present invention include polyamideimide, polyoxymethylene, polycarbonate, polyethylene terephthalate, polybutylene terephthalate, polyvinyl chloride, polyether ether ketone, polyphenylene sulfide, polystyrene, polypropylene, etc., and polyethylene terephthalate is preferred because of its ease of processing.
- the material may be made of a copolymer or may be a multilayer structure yarn as long as it is within the true density and water absorption rate ranges of the present invention.
- organic and/or inorganic substances and/or metals may be added to the nonwoven fabric as long as it is within the true density and water absorption rate ranges of the present invention.
- the thickness of the fibers that make up the nonwoven fabric is preferably 1 to 1000 ⁇ m, and more preferably 10 to 300 ⁇ m, but it is preferable to select an appropriate thickness taking into consideration the type and characteristics of the cells used.
- the structure of the fibers that make up the nonwoven fabric is not particularly limited, and an example of a cross-sectional structure is a circle, but it may also be flower-shaped or flattened and multi-lobed to increase the surface area, or it may have a hollow fiber structure to allow the cell culture medium to reach the entire nonwoven fabric. It is preferable to select an appropriate structure taking into account the type and characteristics of the cells used.
- the thickness of the nonwoven fabric is not particularly limited, but as an example, it is 0.01 to 5 mm, and preferably 0.1 to 3 mm, but it is preferable to select an appropriate thickness taking into consideration the type and characteristics of the cells used.
- the mesh size of the nonwoven fabric is 1 ⁇ m to 1000 ⁇ m.
- the block copolymer with which the nonwoven fabric of the present invention is coated contains the following blocks (A), (B), and (C).
- B A polymer block having an HLB value (Griffin method) in the range of 0 or more and less than 7.
- C A temperature-responsive polymer block having a lower critical solution temperature (LCST) in water in the range of 0°C to 50°C.
- LCST lower critical solution temperature
- the HLB value (Hydrophile-Lipophile Balance: HLB) is a value expressing the degree of affinity for water and oil, as described in W. C. Griffin, Journal of the Society of Cosmetic Chemists, 1, 311 (1949). It takes a value from 0 to 20, and the closer to 0, the higher the hydrophobicity, and the closer to 20, the higher the hydrophilicity.
- Methods for calculating the HLB value by a formula include the Atlas method, the Griffin method, the Davis method, and the Kawakami method.
- the value calculated by the Griffin method is used, and the value is calculated by the following formula based on the formula weight of the hydrophilic portion in the repeating unit of each block constituting the block copolymer of the present invention and the total formula weight of the repeating units.
- HLB value 20 x (formula weight of hydrophilic portion in repeating unit) ⁇ (total formula weight of repeating units)
- hydrophilic moieties in the repeating units of each block include a sulfone moiety (-SO 3 -), a phosphono group (-PO 3 -), a carboxyl group (-COOH), an ester group (-COO-), an amide group (-CONH-), an imide group (-CON-), an aldehyde group (-CHO), a carbonyl group (-CO-), a hydroxyl group (-OH), an amino group (-NH 2 ), an acetyl group (-COCH 3 ), an ethyleneamine group (-CH 2 CH 2 N-), an ethyleneoxy group (-CH 2 CH 2 O-), an alkali metal ion, an alkaline earth metal ion, an ammonium ion, a halide ion, and an acetate ion.
- the atoms constituting the hydrophilic portion must not overlap with the atoms constituting other hydrophilic portions.
- An example of calculating the HLB value in a repeating unit is shown below.
- the hydrophilic portion is one part ester portion, one part phosphono group portion, and one part ethyleneamine portion, and the molecular weight of the hydrophilic portion is 181.04, so the HLB value is 12.3.
- the proportion of block (C) is preferably 30 to 90 mol%, and more preferably 50 to 80 mol%.
- the method for producing the nonwoven fabric of the present invention is not particularly limited, and examples include the impregnation method and the immersion method, in which the nonwoven fabric is immersed or sprayed with a polymer solution, and then dried to remove the solvent.
- the nonwoven fabric of the present invention may be coated with a polymer other than the block copolymer containing blocks (A), (B), and (C), and as an example, coating with a polymer having a positive or negative charge also facilitates cell adhesion.
- the method for producing a nonwoven fabric of the present invention may be, for example, a method characterized by including a step of coating a surface of a nonwoven fabric made of a plastic having a true density of 1.0 to 1.5 g/ cm3 and a water absorption rate of less than 0.3%, with a block copolymer containing the following blocks (A), (B), and (C):
- a polymer block having an HLB value (Griffin method) in the range of 7 or more and 20 or less.
- B A polymer block having an HLB value (Griffin method) in the range of 0 or more and less than 7.
- C A temperature-responsive polymer block having a lower critical solution temperature (LCST) in water in the range of 0°C to 50°C.
- the nonwoven fabric of the present invention can be used for cell culture, particularly for culturing adherent cells.
- Adherent cells are cells that grow while adhering to the surface of a cell culture substrate such as a microcarrier.
- a cell culture substrate such as a microcarrier.
- adherent cells There are no particular limitations on the origin of the adherent cells to be cultured, but examples include mammals such as humans, monkeys, dogs, cats, rabbits, rats, nude mice, mice, guinea pigs, pigs, sheep, Chinese hamsters, and cows, and birds such as chickens and ducks.
- the type of cells (adherent cells) to be cultured is not particularly limited, and examples thereof include mesenchymal stem cells, CHO cells derived from Chinese hamster ovaries, VERO cells derived from African green monkey kidney epithelium, MDCK cells derived from dog kidneys, L929 cells derived from mouse connective tissue, HEK293 cells derived from human fetal kidneys, MDBK cells derived from bovine kidneys, CRFK cells derived from cat kidneys, CPK cells derived from guinea pig kidneys, HeLa cells derived from human cervical cancer, epithelial cells and endothelial cells constituting each tissue and organ in the body, skeletal muscle cells exhibiting contractility, smooth muscle cells, cardiac muscle cells, neuron cells constituting the nervous system, glial cells, fibroblasts, hepatic parenchymal cells involved in the metabolism of the body, non-parenchymal liver cells and adipocytes, and cells having differentiation potential, such as stem cells present in various tissues
- examples include cells contained in blood, lymph, cerebrospinal fluid, sputum, urine or stool, and microorganisms, viruses, protozoa, etc. present in the body or in the environment.
- the type of medium used it may be a serum medium or a serum-free medium, and can be selected appropriately depending on the type of cells.
- the type of basal medium used in the culture of the present invention is not particularly limited, and for example, MEM, ⁇ MEM, DMEM, EMEM, GMEM, DMEM/Ham's F-12, Ham's F-12, Ham's F-10, Medium 199, RPMI 1640, etc. can be used.
- it may be a serum medium or a serum-free medium, but the serum used is not particularly limited, and for example, fetal bovine serum (FBS), calf serum, adult bovine serum, horse serum, sheep serum, goat serum, pig serum, chicken serum, rabbit serum, and human serum can be used.
- the complete medium may or may not contain antibiotics, and can be selected appropriately depending on the type of cells.
- the cell culture method using the nonwoven fabric of the present invention is not particularly limited, and may be cultured under static conditions, shaking conditions, stirring conditions, or a combination of the above methods under intermittent shaking and/or intermittent stirring conditions.
- culturing under shaking conditions there is no particular limit to the shaking speed, and there is no particular limit to the shaking direction.
- stirring speed there is no particular limit to the stirring speed, and one example is 1 to 1000 rpm.
- the culture temperature during culture but it is preferable to carry out the culture at a temperature above the LCST, which can provide an environment in which the temperature-responsive polymer becomes hydrophobic and cells can easily adhere. After culture, the cells on the nonwoven fabric can be recovered by cooling to a temperature below the LCST.
- the cooling method there is no particular limit to the cooling method, and the cells may be directly cooled using a cooler, or the culture medium may be replaced with a cooling liquid.
- the cooling liquid can be a culture medium, and the culture medium may be a complete medium containing growth factors, serum, etc., or a basal medium not containing growth factors, serum, etc.
- the cooling liquid may be a phosphate buffer or a cell detachment liquid containing a protease, but by selecting a cell detachment liquid containing a protease, the number of cells recovered is likely to be high.
- the protease is trypsin, and it may be recombinant trypsin that does not contain animal-derived components.
- the cells are more likely to be detached from the nonwoven fabric as single cells.
- single cells mean that the cells have escaped from a state in which they were connected to each other and are separated into individual cells.
- the concentration of the chelating agent in the phosphate buffer is not particularly limited and is 1 ⁇ M to 1 mM.
- the cooling liquid has a flow, since this can promote the cooling of the nonwoven fabric. For example, the flow can be obtained by stirring or pipetting.
- the cell culture method using the nonwoven fabric of the present invention may be, for example, a method characterized by including a step of culturing cells in a medium containing the nonwoven fabric of the present invention.
- the cell culture method using the nonwoven fabric of the present invention may further include a cooling cell detachment step of detaching cells from microcarriers using a cooling liquid containing a chelating agent.
- the culture performance of the nonwoven fabric of the present invention is evaluated based on the number of cells recovered using a cell detachment solution containing a chilled protease and/or a chilled detachment solution not containing a protease and/or a cell detachment solution containing a chelating agent, and the presence or absence of nonwoven fabric fragments in the recovered cell suspension.
- a cell detachment solution containing a chilled protease and/or a chilled detachment solution not containing a protease and/or a cell detachment solution containing a chelating agent and the presence or absence of nonwoven fabric fragments in the recovered cell suspension.
- the protease but an example is TrypLE Express Enzyme (manufactured by Thermo Fisher Scientific).
- the chilled detachment solution not containing a protease but an example is phosphate buffered saline (PBS(-)) not containing calcium and magnesium.
- the cell detachment solution containing a chelating agent there is no particular limitation on the cell detachment solution containing a chelating agent, but an example is PBS(-) containing 1 mM EDTA.
- the method for measuring the number of cells there is no particular limitation on the method for measuring the number of cells, but the evaluation can be performed using a hemocytometer or the like.
- the GPC device used was a Tosoh Corporation HLC-8320GPC, and two Tosoh Corporation TSKgel SuperAWM-H columns were used. The column temperature was set to 40°C, and the eluent was 2,2,2-trifluoroethanol containing 10 mM sodium trifluoroacetate.
- the measurement sample was prepared at 1.0 mg/mL and measured.
- polymethylmethacrylate Sigma-Aldrich
- the reaction solution was purified by reprecipitation with water and dried under reduced pressure to obtain a yellow solid.
- the obtained yellow solid was dissolved in chloroform, and the chloroform phase was collected using a separatory funnel.
- the collected chloroform phase was concentrated in an evaporator and purified by reprecipitation with hexane.
- the precipitate was collected by filtration and dried under reduced pressure to obtain 5.805 g of polymer MBI.
- ⁇ Polymer CStS having negative charge> 1.156 g (8 mmol) of p-carboxystyrene (CSt) and 1.271 g (12 mmol) of styrene (St) were added to a 100 mL two-neck flask, and 3.3 mg (20 ⁇ mol) of azobisisobutyronitrile and 20 mL of tert-butyl alcohol were added. After argon gas replacement, the mixture was heated and stirred at 64° C. for 24 hours. The reaction solution was purified by reprecipitation with n-heptane and dried under reduced pressure to obtain 0.92 g of polymer CStS. The composition ratio of polymer CStS was CSt/St 33/67 (mol%), the number average molecular weight Mn was 106,000, and the molecular weight distribution Mw/Mn was 1.84.
- Example 1 10 g of a 1.00 wt% 1-methoxy-2-propanol solution of polymer MBI and 10 g of a 0.03 wt% 1-methoxy-2-propanol solution of polymer CStS were added to a 50 mL beaker. 5 g of BioNOCII (manufactured by CESCO BIOENGINEERING, specific surface area 2400 cm 2 /g, water absorption rate 0.1%), which is a polyethylene terephthalate nonwoven fabric, was immersed in the solution in the beaker, and after leaving it to stand for 5 minutes, the nonwoven fabric was removed from the solution and dried at room temperature for 2 hours.
- BioNOCII manufactured by CESCO BIOENGINEERING, specific surface area 2400 cm 2 /g, water absorption rate 0.15%
- the nonwoven fabric was dried under reduced pressure at 50°C to obtain a temperature-responsive nonwoven fabric 1.
- 1 g of the block copolymer coating film of the temperature-responsive nonwoven fabric 1 was extracted with 2,2,2-trifluoroethanol, and the coating amount (fixed amount) of the block copolymer was evaluated from the peak intensity of GPC, which was 5.0 ⁇ g/cm 2 .
- 0.1 g of the temperature-responsive nonwoven fabric 1 and 10 mL of mesenchymal stem cell proliferation medium 2 (PromoCell) were added to a 100 mm untreated dish (Corning).
- 5 x 10 cells of human bone marrow-derived mesenchymal stem cells (passage number 2) (Lonza) were further added and cultured for 6 days in an incubator at 37°C with a 5 vol% CO2 atmosphere.
- the dish was transferred to a safety cabinet at room temperature, the medium was removed, 10 mL of calcium- and magnesium-free phosphate buffered saline (PBS(-)) at 37 ° C was added and left to stand for 1 minute, and the PBS(-) was removed. Furthermore, 5 mL of TrypLE Express Enzyme (manufactured by Thermo Fisher Scientific) at 4 ° C was added and left to stand for 30 minutes.
- PBS(-) calcium- and magnesium-free phosphate buffered saline
- mesenchymal stem cell growth medium 2 5 mL of mesenchymal stem cell growth medium 2 was added, and after pipetting 30 times with a 1 mL pipette, the cell suspension was passed through a 15 mL tube with a cell strainer with a mesh size of 70 ⁇ m set, and 2 mL of PBS(-) was added on the cell strainer, the cell suspension was collected in a tube, concentrated by centrifugation, and the cell number was counted. The cell number was 38.0 ⁇ 10 5 cells, and no fibrous impurities were observed.
- Example 2 A temperature-responsive nonwoven fabric 2 was obtained in the same manner as in Example 1, except that 10 g of a 4.00 wt % 1-methoxy-2-propanol solution of polymer MBI and 10 g of a 0.12 wt % 1-methoxy-2-propanol solution of polymer CStS were added to a 50 mL beaker. The coating amount (fixed amount) of the block copolymer of the temperature-responsive nonwoven fabric 2 was evaluated and found to be 35.0 ⁇ g/ cm2 .
- Human bone marrow-derived mesenchymal stem cells were cultured in the same manner as in Example 1, except that temperature-responsive nonwoven fabric 2 was used.
- the cell suspension was collected in the same manner as in Example 1 except that temperature-responsive nonwoven fabric 2 was used, concentrated by centrifugation, and the cell number was counted.
- the cell number was 38.8 x 10 5 cells, and no fibrous impurities were found.
- Example 3 Temperature-responsive nonwoven fabric 3 was obtained in the same manner as in Example 1, except that 10 g of a 0.50 wt % 1-methoxy-2-propanol solution of polymer MBI and 10 g of a 0.015 wt % 1-methoxy-2-propanol solution of polymer CStS were added to a 50 mL beaker. The coating amount (fixed amount) of the block copolymer of temperature-responsive nonwoven fabric 3 was evaluated and found to be 1.7 ⁇ g/ cm2 .
- Human bone marrow-derived mesenchymal stem cells were cultured in the same manner as in Example 1, except that temperature-responsive nonwoven fabric 3 was used.
- the cell suspension was collected in the same manner as in Example 1 except that temperature-responsive nonwoven fabric 3 was used, concentrated by centrifugation, and the cell number was counted.
- the cell number was 33.3 x 10 5 cells, and no fibrous impurities were found.
- Comparative Example 1 Human bone marrow-derived mesenchymal stem cells were cultured in the same manner as in Example 1, except that 0.1 g of untreated BioNOCII (defined as non-temperature responsive nonwoven fabric A) was used in a 100 mm untreated dish (manufactured by Corning).
- BioNOCII defined as non-temperature responsive nonwoven fabric A
- Example 1 A cell suspension was collected in the same manner as in Example 1 except that non-temperature responsive nonwoven fabric A was used, concentrated by centrifugation, and the cell count was measured. No fibrous impurities were found, but the cell count was 10.2 ⁇ 10 5 cells, which was only about one-third of the number of cells collected in Examples 1 to 3.
- Example 2 A temperature-responsive nonwoven fabric A was obtained in the same manner as in Example 1, except that a nylon nonwoven fabric (specific surface area: 3,500 cm2 /g, water absorption rate: 1.1%) was used. The coating amount (fixed amount) of the block copolymer on the temperature-responsive nonwoven fabric A was evaluated to be 6.0 ⁇ g/ cm2 .
- Human bone marrow-derived mesenchymal stem cells were cultured in the same manner as in Example 1, except that temperature-responsive nonwoven fabric A was used.
- the cell suspension was collected in the same manner as in Example 1 except that temperature-responsive nonwoven fabric A was used, concentrated by centrifugation, and the cell number was counted.
- the cell number was 32.8 ⁇ 10 5 cells, but fibrous impurities were found.
- Comparative Example 3 Human bone marrow-derived mesenchymal stem cells were cultured in the same manner as in Comparative Example 1, except that a nylon nonwoven fabric (specific surface area: 3500 cm 2 /g, water absorption rate: 1.1%) (defined as non-temperature responsive nonwoven fabric B) was used.
- a cell suspension was collected in the same manner as in Example 1 except that non-temperature responsive nonwoven fabric B was used, concentrated by centrifugation, and the cell count was measured.
- the cell count was 10.0 ⁇ 10 5 cells, which was only about one-third of the number of cells collected in Examples 1 to 3, and fibrous impurities were also found.
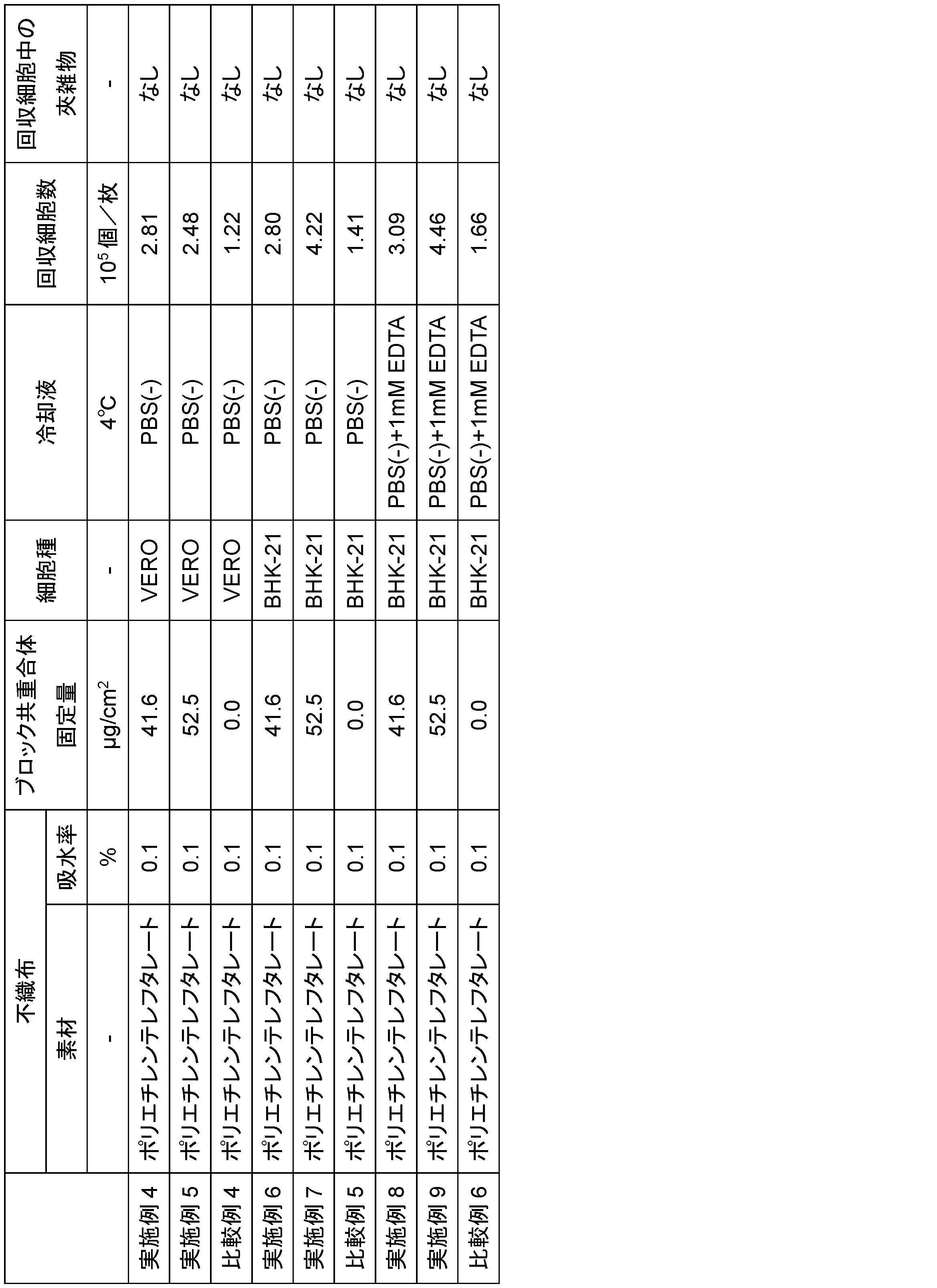
- Example 4 Temperature-responsive nonwoven fabric 4 was obtained in the same manner as in Example 1, except that 10 g of a 6.00 wt % 1-methoxy-2-propanol solution of polymer MBI and 10 g of a 0.09 wt % 1-methoxy-2-propanol solution of polymer CStS were added to a 50 mL beaker. The coating amount (fixed amount) of the block copolymer of temperature-responsive nonwoven fabric 4 was evaluated to be 41.6 ⁇ g/ cm2 .
- the 24-well plate was transferred to a safety cabinet at room temperature, the medium was removed, 2 mL of 37°C PBS(-) was added, and the plate was left to stand for 1 minute for washing, and the PBS(-) was removed. Furthermore, 2 mL of 4°C PBS(-) was added and left to stand for 10 minutes. Then, the plate was pipetted 20 times with a 1 mL pipette for cooling treatment.
- the cell suspension obtained after the cooling treatment was passed through a 50 mL tube equipped with a cell strainer (manufactured by ASONEO, product number: VCS-70) with a mesh size of 70 ⁇ m, and 2 mL of fresh 4°C PBS(-) was added on the cell strainer for washing, and the cell suspension was collected in a tube and concentrated by centrifugation to count the number of cells.
- the cell number was 2.81 ⁇ 10 5 cells/sheet, and no fibrous impurities were observed.
- Example 5 Temperature responsive nonwoven fabric 5 was obtained in the same manner as in Example 1, except that 10 g of a 1-methoxy-2-propanol solution of 8.00 wt % polymer MBI and 10 g of a 1-methoxy-2-propanol solution of 0.16 wt % polymer CStS were added to a 50 mL beaker. The coating amount (fixed amount) of the block copolymer of temperature responsive nonwoven fabric 5 was evaluated to be 52.5 ⁇ g/ cm2 .
- VERO cells were cultured in the same manner as in Example 4, except that temperature-responsive nonwoven fabric 5 was used.
- the cell suspension was recovered in the same manner as in Example 4 except that temperature responsive nonwoven fabric 5 was used, concentrated by centrifugation, and the cell number was counted.
- the cell number was 2.48 ⁇ 10 5 cells/sheet, and no fibrous impurities were found.
- Comparative Example 4 VERO cells were cultured in the same manner as in Example 4, except that non-temperature responsive nonwoven fabric A was used.
- Example 4 A cell suspension was collected in the same manner as in Example 4 except that non-temperature responsive nonwoven fabric A was used, concentrated by centrifugation, and the cell count was counted. No fibrous impurities were found, but the cell count was 1.22 ⁇ 10 5 cells/sheet, which was about half the number of cells collected compared to Examples 4 and 5.
- Example 6 Two sheets of temperature-responsive nonwoven fabric 4 and 2 mL of DMEM (Dulbecco's Modified Eagle Medium, Wako Pure Chemical Industries, Ltd.) containing 10% fetal bovine serum (FBS, Biowest Co., Ltd.) and 1% antibiotic-antimycotic solution (Wako Pure Chemical Industries, Ltd.) were added to an ultra-low attachment surface 24-well plate (Corning, product number: 3473). Furthermore, BHK-21 cells (ATCC, product number: CCL-10) were added at a seeding concentration of 0.25 x 10 5 cells/sheet and cultured for 6 days in an incubator with a 5 vol% CO 2 atmosphere at 37°C.
- DMEM Dynamic Eagle Medium
- FBS fetal bovine serum
- antibiotic-antimycotic solution Wako Pure Chemical Industries, Ltd.
- the 24-well plate was transferred to a safety cabinet at room temperature, the medium was removed, 2 mL of 37°C PBS(-) was added, and the plate was left to stand for 1 minute for washing, and the PBS(-) was removed. Furthermore, 2 mL of 4°C PBS(-) was added and left to stand for 10 minutes. Then, the plate was pipetted 20 times with a 1 mL pipette for cooling treatment.
- the cell suspension obtained after the cooling treatment was passed through a 50 mL tube equipped with a cell strainer (manufactured by ASONEO, product number: VCS-70) with a mesh size of 70 ⁇ m, and 2 mL of fresh 4°C PBS(-) was added on the cell strainer for washing, and the cell suspension was collected in a tube and concentrated by centrifugation to count the number of cells.
- the cell number was 2.80 ⁇ 10 5 cells/sheet, and no fibrous impurities were observed.
- Example 7 BHK-21 cells were cultured in the same manner as in Example 6, except that temperature-responsive nonwoven fabric 5 was used.
- the cell suspension was collected in the same manner as in Example 6 except that temperature-responsive nonwoven fabric 5 was used, concentrated by centrifugation, and the cell count was measured. The cell count was 4.22 ⁇ 10 5 cells/sheet, and no fibrous impurities were observed.
- Example 6 A cell suspension was collected in the same manner as in Example 6 except that non-temperature responsive nonwoven fabric A was used, concentrated by centrifugation, and the cell count was measured. No fibrous impurities were found, but the cell count was 1.41 ⁇ 10 5 cells/sheet, which was only about one-third of the number of cells collected in Examples 6 and 7.
- Example 8 BHK-21 cells were cultured in the same manner as in Example 6, except that temperature-responsive nonwoven fabric 4 was used.
- the 24-well plate was transferred to a safety cabinet at room temperature, the medium was removed, 2 mL of 37°C PBS(-) was added, washed for 1 minute, and the PBS(-) was removed. Furthermore, 2 mL of PBS(-) containing 1 mM EDTA at 4°C was added and left to stand for 10 minutes. Then, the plate was pipetted 20 times with a 1 mL pipette and cooled.
- the cell suspension obtained after the cooling treatment was passed through a 50 mL tube with a cell strainer (manufactured by ASONEO, product number: VCS-70) with a mesh size of 70 ⁇ m, washed by adding 2 mL of fresh 4°C PBS(-), the cell suspension was collected in a tube, concentrated by centrifugation, and the number of cells was counted.
- the number of cells was 3.09 ⁇ 10 5 cells/sheet, which was an increase in the number of cells collected compared to Example 6. In addition, no fibrous impurities were observed.
- Example 9 BHK-21 cells were cultured in the same manner as in Example 6, except that temperature-responsive nonwoven fabric 5 was used.
- Example 8 A cell suspension was collected in the same manner as in Example 8 except that temperature-responsive nonwoven fabric 5 was used, concentrated by centrifugation, and the number of cells was counted, resulting in a cell count of 4.46 ⁇ 10 5 cells/sheet, which was an increase in the number of cells collected compared to Example 7. In addition, no fibrous impurities were observed.
- Comparative Example 6 BHK-21 cells were cultured in the same manner as in Example 6, except that non-temperature responsive nonwoven fabric A was used.
- Example 8 A cell suspension was collected in the same manner as in Example 8, except that non-temperature responsive nonwoven fabric A was used, concentrated by centrifugation, and the cell count was counted. No fibrous impurities were found, but the cell count was 1.66 ⁇ 10 5 cells/sheet, which was about one-third of the number of cells collected in Examples 8 and 9.
Landscapes
- Engineering & Computer Science (AREA)
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Organic Chemistry (AREA)
- Biomedical Technology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Zoology (AREA)
- Wood Science & Technology (AREA)
- Genetics & Genomics (AREA)
- Biotechnology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Health & Medical Sciences (AREA)
- Textile Engineering (AREA)
- Biochemistry (AREA)
- General Engineering & Computer Science (AREA)
- Cell Biology (AREA)
- Microbiology (AREA)
- Virology (AREA)
- Sustainable Development (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
Abstract
Description
本発明は、温度応答性不織布に関する。 The present invention relates to a temperature-responsive nonwoven fabric.
バイオテクノロジーは医療や食品をはじめ幅広く活用されている。バイオテクノロジーの中でも細胞を原料とした開発が加速しており、医療分野では細胞医薬品が難治疾患の治療を目指しており、食品分野では培養肉が省資源省スペースで生産効率の高い食用肉の生産を目指している。これら技術の実用化には原料細胞の生産コスト低減が不可欠である。 Biotechnology is used in a wide range of applications, including in medicine and food. Within biotechnology, development using cells as raw materials is accelerating. In the medical field, cell-based medicines aim to treat intractable diseases, while in the food field, cultured meat aims to produce edible meat that requires fewer resources and space and is highly efficient to produce. Reducing the production costs of the raw cells is essential to putting these technologies to practical use.
一般的に多くの動物細胞は足場依存性があり、細胞を増殖する際は足場となる培養基材が必要である。培養基材としてプラズマ処理を施したポリスチレン製のディッシュがあり、細胞を回収する際はトリプシンなどのタンパク質分解酵素を用いて処理するが、処理が煩雑な課題があった。これに対し特許文献1では、温度応答性ポリマーを基材表面に被覆した培養基材が記載されている。温度応答性ポリマーの相転移を利用し、タンパク質分解酵素を用いずに冷却することで細胞の回収を可能にしており煩雑操作を解消した。 Generally, many animal cells are anchorage-dependent, and require a culture substrate to act as a scaffold when growing cells. Plasma-treated polystyrene dishes are available as culture substrates, and cells are harvested using protease such as trypsin, but this process is cumbersome. In response to this, Patent Document 1 describes a culture substrate whose surface is coated with a temperature-responsive polymer. By utilizing the phase transition of the temperature-responsive polymer, cells can be harvested by cooling without using protease, eliminating the need for cumbersome operations.
また一般的な細胞培養用ディッシュは、ディッシュ底面でしか培養できず単位培地当たりの細胞生産性が低い課題があった。これに対し特許文献2では、温度応答性ポリマーを含んだ不織布が記載されている。温度応答性ポリマーを含んだ不織布は表面積が広いため単位培地量当たりの細胞生産性を高めることができる。さらに細胞回収時は冷却することで繊維の集合構造である不織布が繊維構造まで崩壊し、不織布表面の細胞だけでなく不織布内部の細胞も回収できる。 Also, with typical cell culture dishes, cells can only be cultured on the bottom surface of the dish, which poses the problem of low cell productivity per unit of medium. In response to this, Patent Document 2 describes a nonwoven fabric containing a temperature-responsive polymer. The nonwoven fabric containing a temperature-responsive polymer has a large surface area, which makes it possible to increase cell productivity per unit of medium volume. Furthermore, when recovering cells, the nonwoven fabric, which is an aggregate structure of fibers, is cooled so that it collapses down to the fiber structure, allowing cells to be recovered not only on the surface of the nonwoven fabric, but also inside the nonwoven fabric.
しかしながら、特許文献2記載の温度応答性ポリマーを含んだ不織布を用いた細胞回収方法では、細胞懸濁液中に不織布成分である繊維が含まれてしまうため、繊維を除去する工程が必要となり操作が煩雑な課題が残存した。 However, in the cell recovery method using a nonwoven fabric containing a temperature-responsive polymer as described in Patent Document 2, the fibers that are components of the nonwoven fabric are contained in the cell suspension, necessitating a process for removing the fibers, which makes the process complicated.
本特許の目的は、単位培地当たりの細胞生産性が高く、且つ、繊維状の夾雑物を含まない細胞懸濁液を回収可能な培養基材を提供することにある。 The purpose of this patent is to provide a culture substrate that has high cell productivity per unit medium and enables the recovery of a cell suspension that does not contain fibrous impurities.
本発明者らは、以上の点を鑑み、鋭意研究を重ねた結果、真密度が1.0~1.5g/cm3で吸水率が0.3%未満のプラスチックからなる不織布表面に下記(A)、(B)、(C)のブロックを含むブロック共重合体が被覆されている不織布は、単位培地当たりの細胞生産性が高く、且つ、繊維状の夾雑物を含まない細胞懸濁液を回収できることを見出し、本発明を完成した。すなわち本発明は以下の態様を含包する。
<1>真密度が1.0~1.5g/cm3で吸水率が0.3%未満のプラスチックからなる不織布表面に下記(A)、(B)、(C)のブロックを含むブロック共重合体が被覆されている不織布。
(A)HLB値(グリフィン法)が7以上20以下の範囲にある重合体ブロック。
(B)HLB値(グリフィン法)が0以上7未満の範囲にある重合体ブロック。
(C)水に対する下限臨界溶解温度(LCST)が0℃~50℃の範囲にある温度応答性重合体ブロック。
<2>前記吸水率が0.2%以下であることを特徴とする<1>記載の不織布。<3>プラスチックがポリエチレンテレフタレートであることを特徴とする<1>又は<2>記載の不織布。
<4>ブロック共重合体の被覆量が1.0~100.0μg/cm2であることを特徴とする<1>~<3>記載の不織布。
<5>不織布を構成する繊維の太さが1~1000μmであることを特徴とする<1>~<4>記載の不織布。
<6>ブロック共重合体のブロック(C)の割合が30~90mol%であることを特徴とする<1>~<5>記載の不織布。
<7>ブロック共重合体の数平均分子量が1000~1000000であることを特徴とする<1>~<6>記載の不織布。
<8><1>~<7>記載の不織布の製造方法。
<9>真密度が1.0~1.5g/cm3で吸水率が0.3%未満のプラスチックからなる不織布表面に下記(A)、(B)、(C)のブロックを含むブロック共重合体を被覆する工程を含むことを特徴とする、<1>~<7>記載の不織布の製造方法。
(A)HLB値(グリフィン法)が7以上20以下の範囲にある重合体ブロック。
(B)HLB値(グリフィン法)が0以上7未満の範囲にある重合体ブロック。
(C)水に対する下限臨界溶解温度(LCST)が0℃~50℃の範囲にある温度応答性重合体ブロック。
<10>前記吸水率が0.2%以下であることを特徴とする<9>記載の製造方法。
<11><1>~<7>記載の不織布を用いた細胞培養方法。
<12><1>~<7>記載の不織布を含む培地で、細胞を培養する工程を含むことを特徴とする細胞培養方法。
<13>キレート剤を含む冷却液を用いて、細胞をマイクロキャリアから剥離する冷却細胞剥離工程を含むことを特徴とする<11>又は<12>記載の細胞培養方法。
In view of the above, the present inventors have conducted extensive research and found that a nonwoven fabric made of plastic with a true density of 1.0 to 1.5 g/ cm3 and a water absorption rate of less than 0.3%, the surface of which is coated with a block copolymer containing the following blocks (A), (B), and (C), has high cell productivity per unit medium and enables the recovery of a cell suspension free of fibrous impurities, thereby completing the present invention. That is, the present invention encompasses the following aspects.
<1> A nonwoven fabric made of plastic having a true density of 1.0 to 1.5 g/ cm3 and a water absorption rate of less than 0.3%, the surface of which is coated with a block copolymer containing the following blocks (A), (B), and (C):
(A) A polymer block having an HLB value (Griffin method) in the range of 7 or more and 20 or less.
(B) A polymer block having an HLB value (Griffin method) in the range of 0 or more and less than 7.
(C) A temperature-responsive polymer block having a lower critical solution temperature (LCST) in water in the range of 0°C to 50°C.
<2> The nonwoven fabric according to <1>, characterized in that the water absorption rate is 0.2% or less. <3> The nonwoven fabric according to <1> or <2>, characterized in that the plastic is polyethylene terephthalate.
<4> The nonwoven fabric according to any one of <1> to <3>, wherein the coating amount of the block copolymer is 1.0 to 100.0 μg/ cm2 .
<5> The nonwoven fabric according to any one of <1> to <4>, wherein the fibers constituting the nonwoven fabric have a thickness of 1 to 1000 μm.
<6> The nonwoven fabric according to any one of <1> to <5>, wherein the proportion of the block (C) in the block copolymer is 30 to 90 mol %.
<7> The nonwoven fabric according to any one of <1> to <6>, wherein the block copolymer has a number average molecular weight of 1,000 to 1,000,000.
<8> A method for producing the nonwoven fabric according to any one of <1> to <7>.
<9> A method for producing a nonwoven fabric according to any one of <1> to <7>, comprising a step of coating a surface of a nonwoven fabric made of a plastic having a true density of 1.0 to 1.5 g/ cm3 and a water absorption rate of less than 0.3% with a block copolymer containing the following blocks (A), (B), and (C):
(A) A polymer block having an HLB value (Griffin method) in the range of 7 or more and 20 or less.
(B) A polymer block having an HLB value (Griffin method) in the range of 0 or more and less than 7.
(C) A temperature-responsive polymer block having a lower critical solution temperature (LCST) in water in the range of 0°C to 50°C.
<10> The method according to <9>, wherein the water absorption rate is 0.2% or less.
<11> A cell culture method using the nonwoven fabric according to any one of <1> to <7>.
<12> A cell culture method comprising a step of culturing cells in a medium containing the nonwoven fabric according to any one of <1> to <7>.
<13> The cell culture method according to <11> or <12>, further comprising a cooling cell detachment step of detaching cells from microcarriers using a cooling liquid containing a chelating agent.
真密度が1.0~1.5g/cm3で吸水率が0.3%未満のプラスチックからなる不織布表面に下記(A)、(B)、(C)のブロックを含むブロック共重合体が被覆されている不織布は、単位培地当たりの細胞生産性が高く、且つ、繊維状の夾雑物を含まない細胞懸濁液を回収できる。
(A)HLB値(グリフィン法)が7以上20以下の範囲にある重合体ブロック。
(B)HLB値(グリフィン法)が0以上7未満の範囲にある重合体ブロック。
(C)水に対する下限臨界溶解温度(LCST)が0℃~50℃の範囲にある温度応答性重合体ブロック。
A nonwoven fabric made of plastic having a true density of 1.0 to 1.5 g/ cm3 and a water absorption rate of less than 0.3%, the surface of which is coated with a block copolymer containing the following blocks (A), (B), and (C), has high cell productivity per unit medium and can recover a cell suspension that does not contain fibrous impurities.
(A) A polymer block having an HLB value (Griffin method) in the range of 7 or more and 20 or less.
(B) A polymer block having an HLB value (Griffin method) in the range of 0 or more and less than 7.
(C) A temperature-responsive polymer block having a lower critical solution temperature (LCST) in water in the range of 0°C to 50°C.
以下、本発明を実施するための形態について詳細に説明するが、本発明を以下の内容に限定する趣旨ではない。本発明は、その趣旨の範囲内で適宜変更して実施できる。 Below, we will explain in detail the form for carrying out the present invention, but it is not intended to limit the present invention to the contents below. The present invention can be carried out with appropriate modifications within the scope of its intent.
本発明は真密度が1.0~1.5g/cm3で吸水率が0.3%未満のプラスチックからなる不織布表面に(A)、(B)、(C)のブロックを含むブロック共重合体が被覆されている不織布である。 The present invention relates to a nonwoven fabric made of plastic having a true density of 1.0 to 1.5 g/ cm3 and a water absorption rate of less than 0.3%, the surface of which is coated with a block copolymer containing blocks (A), (B), and (C).
本発明において不織布とは繊維を熱または機械的または化学的な作用によって接着または絡み合わせる事で布にしたものである。 In the present invention, nonwoven fabric is fabric made by bonding or intertwining fibers through heat, mechanical or chemical action.
本発明において不織布とは、繊維を布状にし、繊維を結合させたものであり、不織布を形成させるシーティング方法に特に限定はないが、一般的に空気中で形成させる乾式法、水中で形成させる湿式法、スパンボンド法等が挙げられ、また、繊維間の結合を形成させるボンディング方法に特に限定はないが、一般的に熱による結合、化学的結合、機械的結合等が挙げられ、本発明の範囲にあれば加工のしやすさに応じ適宜選択できる。 In the present invention, nonwoven fabric is fabric made by forming fibers into a cloth and bonding the fibers together. There are no particular limitations on the sheeting method used to form the nonwoven fabric, but typical examples include the dry method in which the fabric is formed in air, the wet method in which the fabric is formed in water, and the spunbond method. There are no particular limitations on the bonding method used to form bonds between the fibers, but typical examples include thermal bonding, chemical bonding, and mechanical bonding. As long as it is within the scope of the present invention, it can be selected appropriately depending on the ease of processing.
本発明において不織布を構成する繊維の真密度は、不織布を構成する繊維間にある隙間部分(孔隙部分)を除いた体積から算出される密度であり、水中で沈降するために1.0~1.5g/cm3であり、撹拌下で分散しやすいように、好ましくは1.0~1.3g/cm3であり、さらに好ましくは1.0~1.2g/cm3である。不織布を構成する繊維の吸水率は、水中で分解せずに強度を保つために0.3%未満であり、好ましくは0.2%以下であり、さらに好ましくは0.15%以下であり、特に好ましくは0.1%以下である。吸水率の評価方法はISO 62:1999,Plastics-Determination of water absorptionを翻訳した、JIS K 7209:2000,「プラスチック-吸水率の求め方」を参照する。不織布を構成する繊維の材質は特に限定はないが、本発明の真密度と吸水率の範囲にある材質の一例として、ポリアミドイミド、ポリオキシメチレン、ポリカーボネート、ポリエチレンテレフタレート、ポリブチレンテレフタレート、ポリ塩化ビニル、ポリエーテルエーテルケトン、ポリフェニレンスルファイド、ポリスチレン、ポリプロピレン等が挙げられるが、加工のしやすさから好ましくはポリエチレンテレフタレートである。また本発明の真密度と吸水率の範囲にあれば材質は共重合体からなっても良く、多層構造糸であっても良い。また、本発明の真密度と吸水率の範囲にあれば不織布に有機物及び/又は無機物及び/又は金属を添加しても良い。 In the present invention, the true density of the fibers constituting the nonwoven fabric is a density calculated from the volume excluding the gaps (pores) between the fibers constituting the nonwoven fabric, and is 1.0 to 1.5 g/cm 3 in order to settle in water, and is preferably 1.0 to 1.3 g/cm 3 , and more preferably 1.0 to 1.2 g/cm 3 in order to easily disperse under stirring. The water absorption rate of the fibers constituting the nonwoven fabric is less than 0.3% in order to maintain strength without decomposing in water, preferably 0.2% or less, more preferably 0.15% or less, and particularly preferably 0.1% or less. The method for evaluating the water absorption rate refers to JIS K 7209:2000, "Plastics - Determination of water absorption," which is a translation of ISO 62:1999, Plastics - Determination of water absorption. The material of the fibers constituting the nonwoven fabric is not particularly limited, but examples of materials having true densities and water absorption rates within the ranges of the present invention include polyamideimide, polyoxymethylene, polycarbonate, polyethylene terephthalate, polybutylene terephthalate, polyvinyl chloride, polyether ether ketone, polyphenylene sulfide, polystyrene, polypropylene, etc., and polyethylene terephthalate is preferred because of its ease of processing. In addition, the material may be made of a copolymer or may be a multilayer structure yarn as long as it is within the true density and water absorption rate ranges of the present invention. In addition, organic and/or inorganic substances and/or metals may be added to the nonwoven fabric as long as it is within the true density and water absorption rate ranges of the present invention.
不織布を構成する繊維の太さは特に限定はないが、細胞が吸着するために、好ましくは1~1000μmであり、さらに好ましくは10~300μmであるが、使用細胞種や特性を考慮し適宜選択することが好ましい。 There are no particular limitations on the thickness of the fibers that make up the nonwoven fabric, but in order to allow cells to adhere to it, it is preferably 1 to 1000 μm, and more preferably 10 to 300 μm, but it is preferable to select an appropriate thickness taking into consideration the type and characteristics of the cells used.
不織布を構成する繊維の構造は特に限定はなく、断面構造の一例として、円形であり、表面積を高めるために花形や扁平多葉型にしても良いし、細胞培養用培地を不織布全体に行き届かせるために中空糸構造であっても良く、使用細胞種や特性を考慮し適宜選択することが好ましい。 The structure of the fibers that make up the nonwoven fabric is not particularly limited, and an example of a cross-sectional structure is a circle, but it may also be flower-shaped or flattened and multi-lobed to increase the surface area, or it may have a hollow fiber structure to allow the cell culture medium to reach the entire nonwoven fabric. It is preferable to select an appropriate structure taking into account the type and characteristics of the cells used.
不織布の厚みは特に限定はないが、一例として、0.01~5mmであり、好ましくは0.1~3mmであるが、使用細胞種や特性を考慮し適宜選択することが好ましい。 The thickness of the nonwoven fabric is not particularly limited, but as an example, it is 0.01 to 5 mm, and preferably 0.1 to 3 mm, but it is preferable to select an appropriate thickness taking into consideration the type and characteristics of the cells used.
不織布の目開きは特に限定はなく、一例として1μm~1000μmである。 There are no particular limitations on the mesh size of the nonwoven fabric, but as an example it is 1 μm to 1000 μm.
本発明の不織布に被覆するブロック共重合体は下記(A)、(B)、(C)のブロックを含む。
(A)HLB値(グリフィン法)が7以上20以下の範囲にある重合体ブロック。
(B)HLB値(グリフィン法)が0以上7未満の範囲にある重合体ブロック。
(C)水に対する下限臨界溶解温度(LCST)が0℃~50℃の範囲にある温度応答性重合体ブロック。
The block copolymer with which the nonwoven fabric of the present invention is coated contains the following blocks (A), (B), and (C).
(A) A polymer block having an HLB value (Griffin method) in the range of 7 or more and 20 or less.
(B) A polymer block having an HLB value (Griffin method) in the range of 0 or more and less than 7.
(C) A temperature-responsive polymer block having a lower critical solution temperature (LCST) in water in the range of 0°C to 50°C.
HLB値(Hydrophile-Lipophile Balance:HLB)とは、W.C.Griffin, Journal of the Society of Cosmetic Chemists, 1, 311(1949).に記載の、水と油への親和性の程度を表す値であり、0から20までの値を取り、0に近いほど疎水性が高く、20に近いほど親水性が高くなる。計算式によりHLB値を算出方法として、アトラス法、グリフィン法、デイビス法、川上法があるが、本明細書においては、グリフィン法により算出した値を使用し、本発明のブロック共重合体を構成する各ブロックの繰り返し単位中の親水部の式量と繰り返し単位の総式量を元に、下記の計算式により算出した。
HLB値=20×(繰り返し単位中の親水部の式量)÷(繰り返し単位の総式量)
The HLB value (Hydrophile-Lipophile Balance: HLB) is a value expressing the degree of affinity for water and oil, as described in W. C. Griffin, Journal of the Society of Cosmetic Chemists, 1, 311 (1949). It takes a value from 0 to 20, and the closer to 0, the higher the hydrophobicity, and the closer to 20, the higher the hydrophilicity. Methods for calculating the HLB value by a formula include the Atlas method, the Griffin method, the Davis method, and the Kawakami method. In this specification, the value calculated by the Griffin method is used, and the value is calculated by the following formula based on the formula weight of the hydrophilic portion in the repeating unit of each block constituting the block copolymer of the present invention and the total formula weight of the repeating units.
HLB value = 20 x (formula weight of hydrophilic portion in repeating unit) ÷ (total formula weight of repeating units)
前述の、各ブロックの繰り返し単位中の親水部の定義として、スルホン部(-SO3-)、ホスホノ基部(-PO3-)、カルボキシル基部(-COOH)、エステル部(-COO-)、アミド部(-CONH-)、イミド部(-CON-)、アルデヒド基部(-CHO)、カルボニル基部(-CO-)、ヒドロキシル基部(-OH)、アミノ基部(-NH2)、アセチル基部(-COCH3)、エチレンアミン部(-CH2CH2N-)、エチレンオキシ部(-CH2CH2O-)、アルカリ金属イオン、アルカリ土類金属イオン、アンモニウムイオン、ハロゲン化物イオン、酢酸イオンを例示することができる。 As mentioned above, examples of hydrophilic moieties in the repeating units of each block include a sulfone moiety (-SO 3 -), a phosphono group (-PO 3 -), a carboxyl group (-COOH), an ester group (-COO-), an amide group (-CONH-), an imide group (-CON-), an aldehyde group (-CHO), a carbonyl group (-CO-), a hydroxyl group (-OH), an amino group (-NH 2 ), an acetyl group (-COCH 3 ), an ethyleneamine group (-CH 2 CH 2 N-), an ethyleneoxy group (-CH 2 CH 2 O-), an alkali metal ion, an alkaline earth metal ion, an ammonium ion, a halide ion, and an acetate ion.
繰り返し単位中の親水部の算出では、親水部を構成する原子が、他の親水部を構成する原子として重複してはならない。繰り返し単位中のHLB値の算出例を以下に記載した。例えば、2-メタクリロイルオキシエチルホスホリルコリン(分子量:295.27)の場合、親水部は、エステル部が1部、ホスホノ基部が1部およびエチレンアミン部が1部であり、親水部の分子量は181.04であるから、HLB値は12.3である。2-ジメチルアミノエチルメタクリレート(分子量:157.11)の場合、親水部は、エステル部が1部およびエチレンアミン部が1部であり、親水部の分子量は86.07であるから、HLB値は11.0である。メチルメタクリレート(分子量:100.12)の場合、親水部は、エステル部が1部であり、親水部の分子量は44.01であるから、HLB値は8.8である。n-ブチルメタクリレート(分子量:142.20)の場合、親水部は、エステル部が1部であり、親水部の分子量は44.01であるから、HLB値は6.2である。 When calculating the hydrophilic portion in a repeating unit, the atoms constituting the hydrophilic portion must not overlap with the atoms constituting other hydrophilic portions. An example of calculating the HLB value in a repeating unit is shown below. For example, in the case of 2-methacryloyloxyethyl phosphorylcholine (molecular weight: 295.27), the hydrophilic portion is one part ester portion, one part phosphono group portion, and one part ethyleneamine portion, and the molecular weight of the hydrophilic portion is 181.04, so the HLB value is 12.3. In the case of 2-dimethylaminoethyl methacrylate (molecular weight: 157.11), the hydrophilic portion is one part ester portion and one part ethyleneamine portion, and the molecular weight of the hydrophilic portion is 86.07, so the HLB value is 11.0. In the case of methyl methacrylate (molecular weight: 100.12), the hydrophilic portion is one part ester portion, and the molecular weight of the hydrophilic portion is 44.01, so the HLB value is 8.8. In the case of n-butyl methacrylate (molecular weight: 142.20), the hydrophilic portion is one part ester, and the molecular weight of the hydrophilic portion is 44.01, so the HLB value is 6.2.
下限臨界溶解温度(LCST;Lower Critical Solution Temperature)とは、この温度よりも低い温度では高分子が水に溶解して透明の溶液になるが、この温度よりも高い温度では不溶化して白濁するか沈殿が生じ、相分離する温度である。ブロック(C)に含まれる繰返し単位とその水に対するLCSTは、例えば、N-イソプロピルアクリルアミド(LCST=32℃)、N-n-プロピルメタクリルアミド(LCST=22℃)、N-テトラヒドロフルフリルアクリルアミド(LCST=28℃)、N-エトキシエチルアクリルアミド(LCST=35℃)、N,N-ジエチルアクリルアミド(LCST=32℃)、N-イソプロピルメタクリルアミド(LCST=44℃)、N-n-プロピルメタクリルアミド(LCST=28℃)、N-テトラヒドロフルフリルメタクリルアミド(LCST=35℃)、N-メチル-N-イソプロピルアクリルアミド(LCST=23℃)、またはN-メチル-N-n-プロピルアクリルアミド(LCST=20℃)などが例示できる。本発明におけるブロック(C)は、前記繰り返し単位を1種類のみ用いてもよく、2種類以上を組み合わせて用いてもよい。また温度応答性を有するのであれば、前記温度応答性繰返し単位の他に、異なる繰返し単位を含んでも良い。 The lower critical solution temperature (LCST) is the temperature below which a polymer dissolves in water to form a transparent solution, but above which it becomes insoluble and either becomes cloudy or precipitates, resulting in phase separation. Examples of the repeating units contained in block (C) and their LCSTs with respect to water include N-isopropylacrylamide (LCST = 32°C), N-n-propylmethacrylamide (LCST = 22°C), N-tetrahydrofurfuryl acrylamide (LCST = 28°C), N-ethoxyethylacrylamide (LCST = 35°C), N,N-diethylacrylamide (LCST = 32°C), N-isopropylmethacrylamide (LCST = 44°C), N-n-propylmethacrylamide (LCST = 28°C), N-tetrahydrofurfurylmethacrylamide (LCST = 35°C), N-methyl-N-isopropylacrylamide (LCST = 23°C), and N-methyl-N-n-propylacrylamide (LCST = 20°C). The block (C) in the present invention may use only one type of the repeating unit, or may use two or more types in combination. In addition, as long as it has temperature responsiveness, it may contain a different repeating unit in addition to the temperature responsive repeating unit.
(A)、(B)、(C)のブロックを含むブロック共重合体の一例として、(A)2-メトキシエチルアクリレート(HLB値=13.5)の繰り返し単位、(B)n-ブチルアクリレート(HLB値=6.9)の繰り返し単位、(C)N-イソプロピルアクリルアミド(LCST=32℃)の繰り返し単位からなるブロック共重合体が挙げられる。 An example of a block copolymer containing blocks (A), (B), and (C) is a block copolymer consisting of repeating units of (A) 2-methoxyethyl acrylate (HLB value = 13.5), (B) n-butyl acrylate (HLB value = 6.9), and (C) N-isopropylacrylamide (LCST = 32°C).
ブロック共重合体の数平均分子量は特に限定はないが、一例として1000~1000000であり、好ましくは5000~500000、さらに好ましくは10000~200000である。 The number average molecular weight of the block copolymer is not particularly limited, but an example is 1,000 to 1,000,000, preferably 5,000 to 500,000, and more preferably 10,000 to 200,000.
ブロック共重合体の組成比は特に限定はないが、不織布上の培養細胞を冷却で回収するために、ブロック(C)の割合は好ましくは30~90mol%であり、さらに好ましくは50~80mol%である。 There are no particular limitations on the composition ratio of the block copolymer, but in order to recover the cultured cells on the nonwoven fabric by cooling, the proportion of block (C) is preferably 30 to 90 mol%, and more preferably 50 to 80 mol%.
本発明の不織布に被覆するブロック共重合体の被覆量(本発明の不織布へのブロック共重合体の固定量ともいう。)は特に限定はないが、不織布上の培養細胞を冷却で回収するために、好ましくは1.0~100.0μg/cm2であり、さらに好ましくは2.0~10.0μg/cm2である。 The amount of the block copolymer coated on the nonwoven fabric of the present invention (also referred to as the amount of the block copolymer fixed on the nonwoven fabric of the present invention) is not particularly limited, but is preferably 1.0 to 100.0 μg/ cm2 , and more preferably 2.0 to 10.0 μg/ cm2 , in order to recover the cultured cells on the nonwoven fabric by cooling.
本発明の不織布の製造方法は特に限定はなく、一例として含浸法や浸漬法が挙げられ、不織布にポリマー溶液を浸漬ないし吹付を行い、乾燥して溶媒を除去することで得られる。本発明の不織布には(A)、(B)、(C)のブロックを含むブロック共重合体以外のポリマーが被覆されても良く、一例として正電荷や負電荷を有するポリマーも被覆すると細胞が接着しやすくなる。 The method for producing the nonwoven fabric of the present invention is not particularly limited, and examples include the impregnation method and the immersion method, in which the nonwoven fabric is immersed or sprayed with a polymer solution, and then dried to remove the solvent. The nonwoven fabric of the present invention may be coated with a polymer other than the block copolymer containing blocks (A), (B), and (C), and as an example, coating with a polymer having a positive or negative charge also facilitates cell adhesion.
本発明の不織布の製造方法は、例えば、真密度が1.0~1.5g/cm3で吸水率が0.3%未満のプラスチックからなる不織布表面に下記(A)、(B)、(C)のブロックを含むブロック共重合体を被覆する工程を含むことを特徴とする方法であってもよい。
(A)HLB値(グリフィン法)が7以上20以下の範囲にある重合体ブロック。
(B)HLB値(グリフィン法)が0以上7未満の範囲にある重合体ブロック。
(C)水に対する下限臨界溶解温度(LCST)が0℃~50℃の範囲にある温度応答性重合体ブロック。
The method for producing a nonwoven fabric of the present invention may be, for example, a method characterized by including a step of coating a surface of a nonwoven fabric made of a plastic having a true density of 1.0 to 1.5 g/ cm3 and a water absorption rate of less than 0.3%, with a block copolymer containing the following blocks (A), (B), and (C):
(A) A polymer block having an HLB value (Griffin method) in the range of 7 or more and 20 or less.
(B) A polymer block having an HLB value (Griffin method) in the range of 0 or more and less than 7.
(C) A temperature-responsive polymer block having a lower critical solution temperature (LCST) in water in the range of 0°C to 50°C.
本発明の不織布は細胞培養、特に接着細胞の培養に使用できる。接着性細胞とはマイクロキャリア等の細胞培養基材の表面に付着しながら増殖する細胞である。培養する接着細胞の由来は特に限定はないが、一例としてヒト、サル、イヌ、ネコ、ウサギ、ラット、ヌードマウス、マウス、モルモット、ブタ、ヒツジ、チャイニーズハムスター、ウシ等の哺乳類やニワトリ、アヒル等の鳥類等が挙げられる。また、培養する細胞(接着細胞)の種類に特に限定はなく、一例として間葉系幹細胞、チャイニーズハムスター卵巣由来のCHO細胞、アフリカミドリザルの腎臓上皮由来のVERO細胞、イヌ腎臓由来のMDCK細胞、マウス結合組織由来のL929細胞、ヒト胎児腎臓由来細胞のHEK293細胞、ウシ腎由来のMDBK細胞、ネコ腎臓由来のCRFK細胞、モルモット腎臓由来のCPK細胞、ヒト子宮頸癌由来のHeLa細胞、更に生体内の各組織、臓器を構成する上皮細胞や内皮細胞、収縮性を示す骨格筋細胞、平滑筋細胞、心筋細胞、神経系を構成するニューロン細胞、グリア細胞、繊維芽細胞、生体の代謝に関与する肝実質細胞、肝非実質細胞や脂肪細胞、分化能を有する細胞として、種々の組織に存在する幹細胞、さらにはそれらから分化誘導した細胞が挙げられる。これら以外でも、血液、リンパ液、髄液、喀痰、尿又は便に含まれる細胞や、体内あるいは環境中に存在する微生物、ウイルス、原虫等を例示できる。使用する培地の種類は特に限定はなく、血清培地であっても無血清培地であっても良く、細胞の種類に応じて適宜選択できる。 The nonwoven fabric of the present invention can be used for cell culture, particularly for culturing adherent cells. Adherent cells are cells that grow while adhering to the surface of a cell culture substrate such as a microcarrier. There are no particular limitations on the origin of the adherent cells to be cultured, but examples include mammals such as humans, monkeys, dogs, cats, rabbits, rats, nude mice, mice, guinea pigs, pigs, sheep, Chinese hamsters, and cows, and birds such as chickens and ducks. In addition, the type of cells (adherent cells) to be cultured is not particularly limited, and examples thereof include mesenchymal stem cells, CHO cells derived from Chinese hamster ovaries, VERO cells derived from African green monkey kidney epithelium, MDCK cells derived from dog kidneys, L929 cells derived from mouse connective tissue, HEK293 cells derived from human fetal kidneys, MDBK cells derived from bovine kidneys, CRFK cells derived from cat kidneys, CPK cells derived from guinea pig kidneys, HeLa cells derived from human cervical cancer, epithelial cells and endothelial cells constituting each tissue and organ in the body, skeletal muscle cells exhibiting contractility, smooth muscle cells, cardiac muscle cells, neuron cells constituting the nervous system, glial cells, fibroblasts, hepatic parenchymal cells involved in the metabolism of the body, non-parenchymal liver cells and adipocytes, and cells having differentiation potential, such as stem cells present in various tissues, and cells induced to differentiate from them. In addition to these, examples include cells contained in blood, lymph, cerebrospinal fluid, sputum, urine or stool, and microorganisms, viruses, protozoa, etc. present in the body or in the environment. There are no particular limitations on the type of medium used; it may be a serum medium or a serum-free medium, and can be selected appropriately depending on the type of cells.
本発明の培養で使用される基礎培地の種類は特に限定はなく、例えば、MEM、αMEM、DMEM、EMEM、GMEM、DMEM/Ham’sF-12、Ham’sF-12、Ham’sF-10、Medium199、RPMI1640などを用いることができる。また、血清培地であっても無血清培地であっても良いが、使用血清は特に限定はなく、例えば、牛胎児血清(Fetal Bovine Serum:FBS)、児牛血清、成牛血清、ウマ血清、ヒツジ血清、ヤギ血清、ブタ血清、ニワトリ血清、ウサギ血清、ヒト血清が使用できる。また、完成培地は抗生物質を含有してもしなくても良く、細胞の種類に応じて適宜選択できる。 The type of basal medium used in the culture of the present invention is not particularly limited, and for example, MEM, αMEM, DMEM, EMEM, GMEM, DMEM/Ham's F-12, Ham's F-12, Ham's F-10, Medium 199, RPMI 1640, etc. can be used. In addition, it may be a serum medium or a serum-free medium, but the serum used is not particularly limited, and for example, fetal bovine serum (FBS), calf serum, adult bovine serum, horse serum, sheep serum, goat serum, pig serum, chicken serum, rabbit serum, and human serum can be used. In addition, the complete medium may or may not contain antibiotics, and can be selected appropriately depending on the type of cells.
本発明の不織布を用いた細胞培養方法は特に限定はなく、静置条件で培養しても良いし、振とう条件で培養しても良いし、撹拌条件で培養しても良く、前記方法を組みあせた方法による間欠的な振とう条件及び/又は間欠的な攪拌条件でも良い。振とう条件で培養する場合、振とう速度に特に限定はなく、振とう方向にも特に限定はない。撹拌条件で培養する場合は撹拌速度に特に限定はなく、一例として1~1000rpmである。培養時の培養温度は特に限定はないが、温度応答性ポリマーが疎水化し細胞が接着しやすい環境の提供ができるLCST以上で行うことが好ましい。培養後はLCST未満の温度に冷却することで不織布上の細胞を回収できる。冷却方法は特に限定はなく、冷却器を使用して直接冷却しても良いし、培地を冷却液で交換しても良い。冷却液は培地を使うことができ、培地は成長因子や血清等を含む完成培地であっても、成長因子や血清等を含まない基礎培地であっても良い。冷却液がリン酸緩衝液であってもタンパク質分解酵素を含む細胞剥離液であっても良いが、タンパク質分解酵素を含む細胞剥離液を選択することで細胞の回収数が高くなりやすい。タンパク質分解酵素は一例としてトリプシンであり、動物由来成分を含まないリコンビナントトリプシンであっても良い。また、リン酸緩衝液または細胞剥離液にEDTA、EGTAなどのキレート剤を導入すると細胞がさらに不織布からシングルセルとして剥離しやすくなる。ここでシングルセルとは、細胞同士が繋がっている状態から脱し、1細胞ごとに分離していることを意味している。冷却液がリン酸緩衝液である場合、リン酸緩衝液中のキレート剤の濃度は特に限定はなく、1μM~1mMである。細胞を回収する際は、冷却液に流動があると不織布の冷却を促進できるため好ましく、一例として撹拌やピペッティングで流動を得ることができる。 The cell culture method using the nonwoven fabric of the present invention is not particularly limited, and may be cultured under static conditions, shaking conditions, stirring conditions, or a combination of the above methods under intermittent shaking and/or intermittent stirring conditions. When culturing under shaking conditions, there is no particular limit to the shaking speed, and there is no particular limit to the shaking direction. When culturing under stirring conditions, there is no particular limit to the stirring speed, and one example is 1 to 1000 rpm. There is no particular limit to the culture temperature during culture, but it is preferable to carry out the culture at a temperature above the LCST, which can provide an environment in which the temperature-responsive polymer becomes hydrophobic and cells can easily adhere. After culture, the cells on the nonwoven fabric can be recovered by cooling to a temperature below the LCST. There is no particular limit to the cooling method, and the cells may be directly cooled using a cooler, or the culture medium may be replaced with a cooling liquid. The cooling liquid can be a culture medium, and the culture medium may be a complete medium containing growth factors, serum, etc., or a basal medium not containing growth factors, serum, etc. The cooling liquid may be a phosphate buffer or a cell detachment liquid containing a protease, but by selecting a cell detachment liquid containing a protease, the number of cells recovered is likely to be high. One example of the protease is trypsin, and it may be recombinant trypsin that does not contain animal-derived components. Furthermore, by introducing a chelating agent such as EDTA or EGTA into the phosphate buffer or cell detachment liquid, the cells are more likely to be detached from the nonwoven fabric as single cells. Here, single cells mean that the cells have escaped from a state in which they were connected to each other and are separated into individual cells. When the cooling liquid is a phosphate buffer, the concentration of the chelating agent in the phosphate buffer is not particularly limited and is 1 μM to 1 mM. When recovering cells, it is preferable that the cooling liquid has a flow, since this can promote the cooling of the nonwoven fabric. For example, the flow can be obtained by stirring or pipetting.
本発明の不織布を用いた細胞培養方法は、例えば、本発明の不織布を含む培地で、細胞を培養する工程を含むことを特徴とする方法であってもよい。本発明の不織布を用いた細胞培養方法は、キレート剤を導入した冷却液を用いて、細胞をマイクロキャリアから剥離する冷却細胞剥離工程を更に含んでいてもよい。 The cell culture method using the nonwoven fabric of the present invention may be, for example, a method characterized by including a step of culturing cells in a medium containing the nonwoven fabric of the present invention. The cell culture method using the nonwoven fabric of the present invention may further include a cooling cell detachment step of detaching cells from microcarriers using a cooling liquid containing a chelating agent.
本発明の不織布の培養性能は、冷却したタンパク質分解酵素を含む細胞剥離液及び/又はタンパク質分解酵素を含まない冷却剥離液及び/又はキレート剤を含む細胞剥離液を用いて回収できた細胞数と回収した細胞懸濁液中の不織布断片混入の有無で評価する。タンパク質分解酵素は特に限定はないが、一例としてTrypLE Express Enzyme(Thermo Fisher Scientific製)が挙げられる。タンパク質分解酵素を含まない冷却剥離液は特に限定はないが、一例としてカルシウムとマグネシウムが不含のリン酸緩衝生理食塩水(PBS(-))が挙げられる。キレート剤を含む細胞剥離液は特に限定はないが、一例として1mMのEDTAを含むPBS(-)が挙げられる。細胞数の計測方法は特に限定はないが、血球計算板などを用いて評価できる。 The culture performance of the nonwoven fabric of the present invention is evaluated based on the number of cells recovered using a cell detachment solution containing a chilled protease and/or a chilled detachment solution not containing a protease and/or a cell detachment solution containing a chelating agent, and the presence or absence of nonwoven fabric fragments in the recovered cell suspension. There is no particular limitation on the protease, but an example is TrypLE Express Enzyme (manufactured by Thermo Fisher Scientific). There is no particular limitation on the chilled detachment solution not containing a protease, but an example is phosphate buffered saline (PBS(-)) not containing calcium and magnesium. There is no particular limitation on the cell detachment solution containing a chelating agent, but an example is PBS(-) containing 1 mM EDTA. There is no particular limitation on the method for measuring the number of cells, but the evaluation can be performed using a hemocytometer or the like.
以下に本発明の実施例を説明するが、本発明はこれら実施例により何ら制限されるものではない。なお、断りのない限り、試薬は市販品を用いた。 The following describes examples of the present invention, but the present invention is not limited to these examples. Unless otherwise specified, commercially available reagents were used.
<ブロック共重合体の組成解析>
核磁気共鳴測定装置(日本電子製、商品名JNM-ECZ400S/L1)を用いたプロトン核磁気共鳴分光(1H-NMR)スペクトル分析より求めた。
<Composition analysis of block copolymers>
The amount was determined by proton nuclear magnetic resonance spectroscopy (1H-NMR) spectrum analysis using a nuclear magnetic resonance measurement device (manufactured by JEOL Ltd., trade name JNM-ECZ400S/L1).
<ブロック共重合体の分子量、分子量分布の解析>
重量平均分子量(Mw)、数平均分子量(Mn)及び分子量分布(Mw/Mn)は、ゲル・パーミエーション・クロマトグラフィー(GPC)によって測定した。GPC装置は東ソー(株)製 HLC-8320GPCを用い、カラムは東ソー(株)製 TSKgel SuperAWM-Hを2本用い、カラム温度を40℃に設定し、溶離液は10mMトリフルオロ酢酸ナトリウムを含む2,2,2-トリフルオロエタノールを用いて測定した。測定試料は1.0mg/mLで調製して測定した。分子量の検量線は、分子量既知のポリメタクリル酸メチル(Sigma-Aldrich社製)を用いた。
<Analysis of molecular weight and molecular weight distribution of block copolymer>
Weight average molecular weight (Mw), number average molecular weight (Mn) and molecular weight distribution (Mw/Mn) were measured by gel permeation chromatography (GPC). The GPC device used was a Tosoh Corporation HLC-8320GPC, and two Tosoh Corporation TSKgel SuperAWM-H columns were used. The column temperature was set to 40°C, and the eluent was 2,2,2-trifluoroethanol containing 10 mM sodium trifluoroacetate. The measurement sample was prepared at 1.0 mg/mL and measured. For the molecular weight calibration curve, polymethylmethacrylate (Sigma-Aldrich) with a known molecular weight was used.
<温度応答性ポリマーMBIの合成>
100mL2口フラスコに2-メトキシエチルアクリレート(MEA)0.650g(5mmol)を加え、さらにシアノメチルドデシルカルボナトを31.8mg(100μmol)とアゾビスイソブチロニトリル1.6mg(10μmol)と1,4-ジオキサン10mLを加え、アルゴンガス置換後、62℃で24時間加熱撹拌した。
<Synthesis of temperature-responsive polymer MBI>
0.650 g (5 mmol) of 2-methoxyethyl acrylate (MEA) was added to a 100 mL two-neck flask, and 31.8 mg (100 μmol) of cyanomethyl dodecyl carbonate, 1.6 mg (10 μmol) of azobisisobutyronitrile, and 10 mL of 1,4-dioxane were further added. After replacing with argon gas, the mixture was heated and stirred at 62° C. for 24 hours.
1回目の加熱撹拌後、上記にn-ブチルアクリレート(BA)3.845g(30mmol)を加え、さらにアゾビスイソブチロニトリル1.6mg(10μmol)と1,4-ジオキサン5mLを加え、アルゴンガス置換後、62℃で48時間加熱撹拌した。 After the first heating and stirring, 3.845 g (30 mmol) of n-butyl acrylate (BA) was added to the above, followed by 1.6 mg (10 μmol) of azobisisobutyronitrile and 5 mL of 1,4-dioxane. After replacing with argon gas, the mixture was heated and stirred at 62°C for 48 hours.
2回目の加熱撹拌後、上記にN-イソプロピルアクリルアミド(IPAAm,LCST=32℃)7.355g(65mmol)を加え、さらにアゾビスイソブチロニトリル1.6mg(10μmol)と1,4-ジオキサン35mLを加え、アルゴンガス置換後、62℃で48時間加熱撹拌した。 After the second heating and stirring, 7.355 g (65 mmol) of N-isopropylacrylamide (IPAAm, LCST = 32°C) was added to the above, followed by 1.6 mg (10 μmol) of azobisisobutyronitrile and 35 mL of 1,4-dioxane. After replacing with argon gas, the mixture was heated and stirred at 62°C for 48 hours.
3回目の加熱撹拌後、反応液を水で再沈精製し、減圧乾燥することで黄色固体を得た。得られた黄色固体をクロロホルムに溶解し、分液ロートを用いクロロホルム相を回収した。回収したクロロホルム相をエバポレーターで濃縮し、ヘキサンで再沈精製した。沈殿物をろ過で回収し、減圧乾燥することで、ポリマーMBIを5.805g得た。得られたブロック共重合体MBIの組成比はMEA/BA/IPAAm=5/26/69(mol%)であり、数平均分子量Mnは8.5万、分子量分布Mw/Mnは1.78であった。 After the third heating and stirring, the reaction solution was purified by reprecipitation with water and dried under reduced pressure to obtain a yellow solid. The obtained yellow solid was dissolved in chloroform, and the chloroform phase was collected using a separatory funnel. The collected chloroform phase was concentrated in an evaporator and purified by reprecipitation with hexane. The precipitate was collected by filtration and dried under reduced pressure to obtain 5.805 g of polymer MBI. The composition ratio of the obtained block copolymer MBI was MEA/BA/IPAAm = 5/26/69 (mol%), the number average molecular weight Mn was 85,000, and the molecular weight distribution Mw/Mn was 1.78.
<負電荷を有するポリマーCStS>
100mL2口フラスコにp-カルボキシスチレン(CSt)1.156g(8mmol)とスチレン(St)1.271g(12mmol)を加え、さらにアゾビスイソブチロニトリル3.3mg(20μmol)とtert-ブチルアルコール20mLを加え、アルゴンガス置換後、64℃で24時間加熱撹拌した。反応液をn-ヘプタンで再沈精製し、減圧乾燥することで、ポリマーCStSを0.92g得た。ポリマーCStSの組成比はCSt/St=33/67(mol%)、数平均分子量Mnは10.6万、分子量分布Mw/Mnは1.84であった。
<Polymer CStS having negative charge>
1.156 g (8 mmol) of p-carboxystyrene (CSt) and 1.271 g (12 mmol) of styrene (St) were added to a 100 mL two-neck flask, and 3.3 mg (20 μmol) of azobisisobutyronitrile and 20 mL of tert-butyl alcohol were added. After argon gas replacement, the mixture was heated and stirred at 64° C. for 24 hours. The reaction solution was purified by reprecipitation with n-heptane and dried under reduced pressure to obtain 0.92 g of polymer CStS. The composition ratio of polymer CStS was CSt/St=33/67 (mol%), the number average molecular weight Mn was 106,000, and the molecular weight distribution Mw/Mn was 1.84.
<細胞数と細胞生存率の計測>
細胞懸濁液中から10μLを細胞数測定用スライド(Thermo Fisher Scientific製、商品名Countess Cell Counting Chamber Slid)に添加し自動セルカウンター(Thermo Fisher Scientific製、商品名CountessII)を用いて、細胞数を測定した。
<Measurement of cell number and cell viability>
10 μL of the cell suspension was added to a slide for measuring cell count (manufactured by Thermo Fisher Scientific, product name: Countess Cell Counting Chamber Slide), and the cell number was measured using an automatic cell counter (manufactured by Thermo Fisher Scientific, product name: Countess II).
実施例1
50mLのビーカーに1.00wt%のポリマーMBIの1-メトキシ-2-プロパノール溶液を10gと0.03wt%のポリマーCStSの1-メトキシ-2-プロパノール溶液を10g加えた。ビーカー内の溶液にポリエチレンテレフタレートの不織布であるBioNOCII(CESCO BIOENGINEERING製、比表面積2400cm2/g、吸水率0.1%)を5g浸漬させ、5分間静置した後に不織布を溶液から取り出し、室温で2時間乾燥した。さらに50℃で減圧乾燥することで温度応答性不織布1を得た。1gの温度応答性不織布1のブロック共重合体コート膜を2,2,2-トリフルオロエタノールで抽出し、GPCのピーク強度からブロック共重合体の被覆量(固定量)を評価したところ、5.0μg/cm2であった。
Example 1
10 g of a 1.00 wt% 1-methoxy-2-propanol solution of polymer MBI and 10 g of a 0.03 wt% 1-methoxy-2-propanol solution of polymer CStS were added to a 50 mL beaker. 5 g of BioNOCII (manufactured by CESCO BIOENGINEERING, specific surface area 2400 cm 2 /g, water absorption rate 0.1%), which is a polyethylene terephthalate nonwoven fabric, was immersed in the solution in the beaker, and after leaving it to stand for 5 minutes, the nonwoven fabric was removed from the solution and dried at room temperature for 2 hours. Further, the nonwoven fabric was dried under reduced pressure at 50°C to obtain a temperature-responsive nonwoven fabric 1. 1 g of the block copolymer coating film of the temperature-responsive nonwoven fabric 1 was extracted with 2,2,2-trifluoroethanol, and the coating amount (fixed amount) of the block copolymer was evaluated from the peak intensity of GPC, which was 5.0 μg/cm 2 .
100mmの無処理ディッシュ(Corning製)に0.1gの温度応答性不織布1と10mLの間葉系幹細胞増殖培地2(PromoCell製)を加えた。さらにロンザ製のヒト骨髄由来間葉系幹細胞(継代数2)を5×105cells加えて37℃の5vol%CO2雰囲気のインキュベーター内で6日間培養した。 0.1 g of the temperature-responsive nonwoven fabric 1 and 10 mL of mesenchymal stem cell proliferation medium 2 (PromoCell) were added to a 100 mm untreated dish (Corning). 5 x 10 cells of human bone marrow-derived mesenchymal stem cells (passage number 2) (Lonza) were further added and cultured for 6 days in an incubator at 37°C with a 5 vol% CO2 atmosphere.
培養後、ディッシュを室温の安全キャビネットに移し、培地を抜取り、37℃のカルシウムとマグネシウムが不含のリン酸緩衝生理食塩水(PBS(-))を10mL加えて1分間静置し、PBS(-)を抜取った。さらに新たに4℃のTrypLE Express Enzyme(Thermo Fisher Scientific製)を5mL加え、30分間静置した。5mLの間葉系幹細胞増殖培地2を加え、1mLのピペットで30回ピペッティングを行った後、細胞懸濁液を目開き70μmのセルストレーナーをセットした15mLのチューブに通し、さらにセルストレーナー上に2mLのPBS(-)を加え、細胞懸濁液をチューブに回収し、遠心分離で濃縮して細胞数を計測した。細胞数は38.0×105cellsで、繊維状の夾雑物は認められなかった。 After culturing, the dish was transferred to a safety cabinet at room temperature, the medium was removed, 10 mL of calcium- and magnesium-free phosphate buffered saline (PBS(-)) at 37 ° C was added and left to stand for 1 minute, and the PBS(-) was removed. Furthermore, 5 mL of TrypLE Express Enzyme (manufactured by Thermo Fisher Scientific) at 4 ° C was added and left to stand for 30 minutes. 5 mL of mesenchymal stem cell growth medium 2 was added, and after pipetting 30 times with a 1 mL pipette, the cell suspension was passed through a 15 mL tube with a cell strainer with a mesh size of 70 μm set, and 2 mL of PBS(-) was added on the cell strainer, the cell suspension was collected in a tube, concentrated by centrifugation, and the cell number was counted. The cell number was 38.0 × 10 5 cells, and no fibrous impurities were observed.
実施例2
50mLのビーカーに4.00wt%のポリマーMBIの1-メトキシ-2-プロパノール溶液を10gと0.12wt%のポリマーCStSの1-メトキシ-2-プロパノール溶液を10g加えたこと以外は実施例1と同じ方法で温度応答性不織布2を得た。温度応答性不織布2のブロック共重合体の被覆量(固定量)を評価したところ35.0μg/cm2であった。
Example 2
A temperature-responsive nonwoven fabric 2 was obtained in the same manner as in Example 1, except that 10 g of a 4.00 wt % 1-methoxy-2-propanol solution of polymer MBI and 10 g of a 0.12 wt % 1-methoxy-2-propanol solution of polymer CStS were added to a 50 mL beaker. The coating amount (fixed amount) of the block copolymer of the temperature-responsive nonwoven fabric 2 was evaluated and found to be 35.0 μg/ cm2 .
温度応答性不織布2を用いたこと以外は実施例1と同じ方法でヒト骨髄由来間葉系幹細胞を培養した。 Human bone marrow-derived mesenchymal stem cells were cultured in the same manner as in Example 1, except that temperature-responsive nonwoven fabric 2 was used.
温度応答性不織布2を用いたこと以外は実施例1と同じ方法で細胞懸濁液を回収し、遠心分離で濃縮して細胞数を計測したところ、細胞数は38.8×105cellsで、繊維状の夾雑物は認められなかった。 The cell suspension was collected in the same manner as in Example 1 except that temperature-responsive nonwoven fabric 2 was used, concentrated by centrifugation, and the cell number was counted. The cell number was 38.8 x 10 5 cells, and no fibrous impurities were found.
実施例3
50mLのビーカーに0.50wt%のポリマーMBIの1-メトキシ-2-プロパノール溶液を10gと0.015wt%のポリマーCStSの1-メトキシ-2-プロパノール溶液を10g加えたこと以外は実施例1と同じ方法で温度応答性不織布3を得た。温度応答性不織布3のブロック共重合体の被覆量(固定量)を評価したところ1.7μg/cm2であった。
Example 3
Temperature-responsive nonwoven fabric 3 was obtained in the same manner as in Example 1, except that 10 g of a 0.50 wt % 1-methoxy-2-propanol solution of polymer MBI and 10 g of a 0.015 wt % 1-methoxy-2-propanol solution of polymer CStS were added to a 50 mL beaker. The coating amount (fixed amount) of the block copolymer of temperature-responsive nonwoven fabric 3 was evaluated and found to be 1.7 μg/ cm2 .
温度応答性不織布3を用いたこと以外は実施例1と同じ方法でヒト骨髄由来間葉系幹細胞を培養した。 Human bone marrow-derived mesenchymal stem cells were cultured in the same manner as in Example 1, except that temperature-responsive nonwoven fabric 3 was used.
温度応答性不織布3を用いたこと以外は実施例1と同じ方法で細胞懸濁液を回収し、遠心分離で濃縮して細胞数を計測したところ、細胞数は33.3×105cellsで、繊維状の夾雑物は認められなかった。 The cell suspension was collected in the same manner as in Example 1 except that temperature-responsive nonwoven fabric 3 was used, concentrated by centrifugation, and the cell number was counted. The cell number was 33.3 x 10 5 cells, and no fibrous impurities were found.
比較例1
100mmの無処理ディッシュ(Corning製)に0.1gの未処理のBioNOCII(非温度応答性不織布Aと規定)を用いたこと以外は実施例1と同じ方法でヒト骨髄由来間葉系幹細胞を培養した。
Comparative Example 1
Human bone marrow-derived mesenchymal stem cells were cultured in the same manner as in Example 1, except that 0.1 g of untreated BioNOCII (defined as non-temperature responsive nonwoven fabric A) was used in a 100 mm untreated dish (manufactured by Corning).
非温度応答性不織布Aを用いたこと以外は実施例1と同じ方法で細胞懸濁液を回収し、遠心分離で濃縮して細胞数を計測したところ、繊維状の夾雑物は認められなかったが、細胞数は10.2×105cellsと、実施例1~3と比較して3分の1程度の細胞回収数にとどまった。 A cell suspension was collected in the same manner as in Example 1 except that non-temperature responsive nonwoven fabric A was used, concentrated by centrifugation, and the cell count was measured. No fibrous impurities were found, but the cell count was 10.2 × 10 5 cells, which was only about one-third of the number of cells collected in Examples 1 to 3.
比較例2
ナイロン製の不織布(比表面積3500cm2/g、吸水率1.1%)を用いたこと以外は実施例1と同じ方法で温度応答性不織布Aを得た。温度応答性不織布Aのブロック共重合体の被覆量(固定量)を評価したところ6.0μg/cm2であった。
Comparative Example 2
A temperature-responsive nonwoven fabric A was obtained in the same manner as in Example 1, except that a nylon nonwoven fabric (specific surface area: 3,500 cm2 /g, water absorption rate: 1.1%) was used. The coating amount (fixed amount) of the block copolymer on the temperature-responsive nonwoven fabric A was evaluated to be 6.0 μg/ cm2 .
温度応答性不織布Aを用いたこと以外は実施例1と同じ方法でヒト骨髄由来間葉系幹細胞を培養した。 Human bone marrow-derived mesenchymal stem cells were cultured in the same manner as in Example 1, except that temperature-responsive nonwoven fabric A was used.
温度応答性不織布Aを用いたこと以外は実施例1と同じ方法で細胞懸濁液を回収し、遠心分離で濃縮して細胞数を計測したところ、細胞数は32.8×105cellsだったが、繊維状の夾雑物が認められた。 The cell suspension was collected in the same manner as in Example 1 except that temperature-responsive nonwoven fabric A was used, concentrated by centrifugation, and the cell number was counted. The cell number was 32.8 × 10 5 cells, but fibrous impurities were found.
比較例3
ナイロン製の不織布(比表面積3500cm2/g、吸水率1.1%)(非温度応答性不織布Bと規定)を用いたこと以外は比較例1と同じ方法でヒト骨髄由来間葉系幹細胞を培養した。
Comparative Example 3
Human bone marrow-derived mesenchymal stem cells were cultured in the same manner as in Comparative Example 1, except that a nylon nonwoven fabric (specific surface area: 3500 cm 2 /g, water absorption rate: 1.1%) (defined as non-temperature responsive nonwoven fabric B) was used.
非温度応答性不織布Bを用いたこと以外は実施例1と同じ方法で細胞懸濁液を回収し、遠心分離で濃縮して細胞数を計測したところ、細胞数は10.0×105cellsと、実施例1~3と比較して3分の1程度の細胞回収数にとどまり、さらに繊維状の夾雑物が認められた。 A cell suspension was collected in the same manner as in Example 1 except that non-temperature responsive nonwoven fabric B was used, concentrated by centrifugation, and the cell count was measured. The cell count was 10.0 × 10 5 cells, which was only about one-third of the number of cells collected in Examples 1 to 3, and fibrous impurities were also found.
実施例4
50mLのビーカーに6.00wt%のポリマーMBIの1-メトキシ-2-プロパノール溶液を10gと0.09wt%のポリマーCStSの1-メトキシ-2-プロパノール溶液を10g加えたこと以外は実施例1と同じ方法で温度応答性不織布4を得た。温度応答性不織布4のブロック共重合体の被覆量(固定量)を評価したところ41.6μg/cm2であった。
Example 4
Temperature-responsive nonwoven fabric 4 was obtained in the same manner as in Example 1, except that 10 g of a 6.00 wt % 1-methoxy-2-propanol solution of polymer MBI and 10 g of a 0.09 wt % 1-methoxy-2-propanol solution of polymer CStS were added to a 50 mL beaker. The coating amount (fixed amount) of the block copolymer of temperature-responsive nonwoven fabric 4 was evaluated to be 41.6 μg/ cm2 .
超低接着表面24ウェルプレート(Corning社製、製品番号:3473)に、2枚の温度応答性不織布4と2mLの10%のウシ胎児血清(FBS、バイオウエスト社製)と1%の抗生物質-抗真菌剤溶液(和光純薬社製)を含有するDMEM(Dulbecco’s Modified Eagle Medium、和光純薬社製)を加えた。さらに、VERO細胞(ATCC製、製品番号:CCL-81)を0.25×105cells/枚の播種濃度で加えて37℃の5vol%CO2雰囲気のインキュベーター内で6日間培養した。 Two sheets of temperature-responsive nonwoven fabric 4 and 2 mL of DMEM (Dulbecco's Modified Eagle Medium, Wako Pure Chemical Industries) containing 10% fetal bovine serum (FBS, Biowest) and 1% antibiotic-antimycotic solution (Wako Pure Chemical Industries) were added to an ultra-low attachment surface 24-well plate (Corning, product number: 3473). Furthermore, VERO cells (ATCC, product number: CCL-81) were added at a seeding concentration of 0.25 x 10 5 cells/sheet and cultured for 6 days in an incubator with a 5 vol% CO 2 atmosphere at 37°C.
培養後、24ウェルプレートを室温の安全キャビネットに移し、培地を抜取り、37℃のPBS(-)を2mL加えて1分間静置洗浄し、PBS(-)を抜取った。さらに、4℃のPBS(-)を2mL加え、10分間静置した。その後、1mLのピペットで20回ピペッティングし冷却処理を行った。冷却処理後得られた細胞懸濁液を目開き70μmのセルストレーナー(ASONE社製、製品番号:VCS―70)をセットした50mLのチューブに通し、セルストレーナー上に2mLの新たな4℃のPBS(-)を加え洗浄し、細胞懸濁液をチューブに回収し、遠心分離で濃縮して細胞数を計測したところ、細胞数は2.81×105cells/枚で、繊維状の夾雑物は認められなかった。 After the culture, the 24-well plate was transferred to a safety cabinet at room temperature, the medium was removed, 2 mL of 37°C PBS(-) was added, and the plate was left to stand for 1 minute for washing, and the PBS(-) was removed. Furthermore, 2 mL of 4°C PBS(-) was added and left to stand for 10 minutes. Then, the plate was pipetted 20 times with a 1 mL pipette for cooling treatment. The cell suspension obtained after the cooling treatment was passed through a 50 mL tube equipped with a cell strainer (manufactured by ASONEO, product number: VCS-70) with a mesh size of 70 μm, and 2 mL of fresh 4°C PBS(-) was added on the cell strainer for washing, and the cell suspension was collected in a tube and concentrated by centrifugation to count the number of cells. The cell number was 2.81 × 10 5 cells/sheet, and no fibrous impurities were observed.
実施例5
50mLのビーカーに8.00wt%のポリマーMBIの1-メトキシ-2-プロパノール溶液を10gと0.16wt%のポリマーCStSの1-メトキシ-2-プロパノール溶液を10g加えたこと以外は実施例1と同じ方法で温度応答性不織布5を得た。温度応答性不織布5のブロック共重合体の被覆量(固定量)を評価したところ52.5μg/cm2であった。
Example 5
Temperature responsive nonwoven fabric 5 was obtained in the same manner as in Example 1, except that 10 g of a 1-methoxy-2-propanol solution of 8.00 wt % polymer MBI and 10 g of a 1-methoxy-2-propanol solution of 0.16 wt % polymer CStS were added to a 50 mL beaker. The coating amount (fixed amount) of the block copolymer of temperature responsive nonwoven fabric 5 was evaluated to be 52.5 μg/ cm2 .
温度応答性不織布5を用いたこと以外は実施例4と同じ方法でVERO細胞を培養した。 VERO cells were cultured in the same manner as in Example 4, except that temperature-responsive nonwoven fabric 5 was used.
温度応答性不織布5を用いたこと以外は実施例4と同じ方法で細胞懸濁液を回収し、遠心分離で濃縮して細胞数を計測したところ、細胞数は2.48×105cells/枚、繊維状の夾雑物は認められなかった。 The cell suspension was recovered in the same manner as in Example 4 except that temperature responsive nonwoven fabric 5 was used, concentrated by centrifugation, and the cell number was counted. The cell number was 2.48 × 10 5 cells/sheet, and no fibrous impurities were found.
比較例4
非温度応答性不織布Aを用いたこと以外は実施例4と同じ方法でVERO細胞を培養した。
Comparative Example 4
VERO cells were cultured in the same manner as in Example 4, except that non-temperature responsive nonwoven fabric A was used.
非温度応答性不織布Aを用いたこと以外は実施例4と同じ方法で細胞懸濁液を回収し、遠心分離で濃縮して細胞数を計測したところ、繊維状の夾雑物は認められなかったが、細胞数は1.22×105cells/枚と、実施例4~5と比較して2分の1程度の細胞回収数にとどまった。 A cell suspension was collected in the same manner as in Example 4 except that non-temperature responsive nonwoven fabric A was used, concentrated by centrifugation, and the cell count was counted. No fibrous impurities were found, but the cell count was 1.22 × 10 5 cells/sheet, which was about half the number of cells collected compared to Examples 4 and 5.
実施例6
超低接着表面24ウェルプレート(Corning社製、製品番号:3473)に、2枚の温度応答性不織布4と2mLの10%のウシ胎児血清(FBS、バイオウエスト社製)と1%の抗生物質-抗真菌剤溶液(和光純薬社製)を含有するDMEM(Dulbecco’s Modified Eagle Medium、和光純薬社製)を加えた。さらに、BHK-21細胞(ATCC製、製品番号:CCL-10)を0.25×105cells/枚の播種濃度で加えて37℃の5vol%CO2雰囲気のインキュベーター内で6日間培養した。
Example 6
Two sheets of temperature-responsive nonwoven fabric 4 and 2 mL of DMEM (Dulbecco's Modified Eagle Medium, Wako Pure Chemical Industries, Ltd.) containing 10% fetal bovine serum (FBS, Biowest Co., Ltd.) and 1% antibiotic-antimycotic solution (Wako Pure Chemical Industries, Ltd.) were added to an ultra-low attachment surface 24-well plate (Corning, product number: 3473). Furthermore, BHK-21 cells (ATCC, product number: CCL-10) were added at a seeding concentration of 0.25 x 10 5 cells/sheet and cultured for 6 days in an incubator with a 5 vol% CO 2 atmosphere at 37°C.
培養後、24ウェルプレートを室温の安全キャビネットに移し、培地を抜取り、37℃のPBS(-)を2mL加えて1分間静置洗浄し、PBS(-)を抜取った。さらに、4℃のPBS(-)を2mL加え、10分間静置した。その後、1mLのピペットで20回ピペッティングし冷却処理を行った。冷却処理後得られた細胞懸濁液を目開き70μmのセルストレーナー(ASONE社製、製品番号:VCS-70)をセットした50mLのチューブに通し、セルストレーナー上に2mLの新たな4℃のPBS(-)を加え洗浄し、細胞懸濁液をチューブに回収し、遠心分離で濃縮して細胞数を計測したところ、細胞数は2.80×105cells/枚で、繊維状の夾雑物は認められなかった。 After the culture, the 24-well plate was transferred to a safety cabinet at room temperature, the medium was removed, 2 mL of 37°C PBS(-) was added, and the plate was left to stand for 1 minute for washing, and the PBS(-) was removed. Furthermore, 2 mL of 4°C PBS(-) was added and left to stand for 10 minutes. Then, the plate was pipetted 20 times with a 1 mL pipette for cooling treatment. The cell suspension obtained after the cooling treatment was passed through a 50 mL tube equipped with a cell strainer (manufactured by ASONEO, product number: VCS-70) with a mesh size of 70 μm, and 2 mL of fresh 4°C PBS(-) was added on the cell strainer for washing, and the cell suspension was collected in a tube and concentrated by centrifugation to count the number of cells. The cell number was 2.80 × 10 5 cells/sheet, and no fibrous impurities were observed.
実施例7
温度応答性不織布5を用いたこと以外は実施例6と同じ方法でBHK-21細胞を培養した。
Example 7
BHK-21 cells were cultured in the same manner as in Example 6, except that temperature-responsive nonwoven fabric 5 was used.
温度応答性不織布5を用いたこと以外は実施例6と同じ方法で細胞懸濁液を回収し、遠心分離で濃縮して細胞数を計測したところ、細胞数は4.22×105cells/枚、繊維状の夾雑物は認められなかった。 The cell suspension was collected in the same manner as in Example 6 except that temperature-responsive nonwoven fabric 5 was used, concentrated by centrifugation, and the cell count was measured. The cell count was 4.22 × 10 5 cells/sheet, and no fibrous impurities were observed.
比較例5
非温度応答性不織布Aを用いたこと以外は実施例6と同じ方法でBHK-21細胞を培養した。
Comparative Example 5
BHK-21 cells were cultured in the same manner as in Example 6, except that non-temperature responsive nonwoven fabric A was used.
非温度応答性不織布Aを用いたこと以外は実施例6と同じ方法で細胞懸濁液を回収し、遠心分離で濃縮して細胞数を計測したところ、繊維状の夾雑物は認められなかったが、細胞数は1.41×105cells/枚と、実施例6~7と比較して3分の1程度の細胞回収数にとどまった。 A cell suspension was collected in the same manner as in Example 6 except that non-temperature responsive nonwoven fabric A was used, concentrated by centrifugation, and the cell count was measured. No fibrous impurities were found, but the cell count was 1.41 × 10 5 cells/sheet, which was only about one-third of the number of cells collected in Examples 6 and 7.
実施例8
温度応答性不織布4を用いたこと以外は実施例6と同じ方法でBHK-21細胞を培養した。
Example 8
BHK-21 cells were cultured in the same manner as in Example 6, except that temperature-responsive nonwoven fabric 4 was used.
培養後、24ウェルプレートを室温の安全キャビネットに移し、培地を抜取り、37℃のPBS(-)を2mL加えて1分間洗浄し、PBS(-)を抜取った。さらに、4℃の1mMのEDTAを含むPBS(-)を2mL加え、10分間静置した。その後、1mLのピペットで20回ピペッティングし冷却処理を行った。冷却処理後得られた細胞懸濁液を目開き70μmのセルストレーナー(ASONE社製、製品番号:VCS-70)をセットした50mLのチューブに通し、セルストレーナー上に2mLの新たな4℃のPBS(-)を加え洗浄し、細胞懸濁液をチューブに回収し、遠心分離で濃縮して細胞数を計測したところ、細胞数は3.09×105cells/枚と、実施例6と比較し細胞回収数が増加された。また、繊維状の夾雑物は認められなかった。 After the culture, the 24-well plate was transferred to a safety cabinet at room temperature, the medium was removed, 2 mL of 37°C PBS(-) was added, washed for 1 minute, and the PBS(-) was removed. Furthermore, 2 mL of PBS(-) containing 1 mM EDTA at 4°C was added and left to stand for 10 minutes. Then, the plate was pipetted 20 times with a 1 mL pipette and cooled. The cell suspension obtained after the cooling treatment was passed through a 50 mL tube with a cell strainer (manufactured by ASONEO, product number: VCS-70) with a mesh size of 70 μm, washed by adding 2 mL of fresh 4°C PBS(-), the cell suspension was collected in a tube, concentrated by centrifugation, and the number of cells was counted. The number of cells was 3.09 × 10 5 cells/sheet, which was an increase in the number of cells collected compared to Example 6. In addition, no fibrous impurities were observed.
実施例9
温度応答性不織布5を用いたこと以外は実施例6と同じ方法でBHK-21細胞を培養した。
Example 9
BHK-21 cells were cultured in the same manner as in Example 6, except that temperature-responsive nonwoven fabric 5 was used.
温度応答性不織布5を用いたこと以外は実施例8と同じ方法で細胞懸濁液を回収し、遠心分離で濃縮して細胞数を計測したところ、細胞数は4.46×105cells/枚と、実施例7と比較し細胞回収数が増加された。また、繊維状の夾雑物は認められなかった。 A cell suspension was collected in the same manner as in Example 8 except that temperature-responsive nonwoven fabric 5 was used, concentrated by centrifugation, and the number of cells was counted, resulting in a cell count of 4.46 × 10 5 cells/sheet, which was an increase in the number of cells collected compared to Example 7. In addition, no fibrous impurities were observed.
比較例6
非温度応答性不織布Aを用いたこと以外は実施例6と同じ方法でBHK-21細胞を培養した。
Comparative Example 6
BHK-21 cells were cultured in the same manner as in Example 6, except that non-temperature responsive nonwoven fabric A was used.
非温度応答性不織布Aを用いたこと以外は実施例8と同じ方法で細胞懸濁液を回収し、遠心分離で濃縮して細胞数を計測したところ、繊維状の夾雑物は認められなかったが、細胞数は1.66×105cells/枚と、実施例8~9と比較して3分の1程度の細胞回収数にとどまった。 A cell suspension was collected in the same manner as in Example 8, except that non-temperature responsive nonwoven fabric A was used, concentrated by centrifugation, and the cell count was counted. No fibrous impurities were found, but the cell count was 1.66 × 10 5 cells/sheet, which was about one-third of the number of cells collected in Examples 8 and 9.
Claims (13)
(A)HLB値(グリフィン法)が7以上20以下の範囲にある重合体ブロック。
(B)HLB値(グリフィン法)が0以上7未満の範囲にある重合体ブロック。
(C)水に対する下限臨界溶解温度(LCST)が0℃~50℃の範囲にある温度応答性重合体ブロック。 A nonwoven fabric made of plastic having a true density of 1.0 to 1.5 g/ cm3 and a water absorption rate of less than 0.3%, the surface of which is coated with a block copolymer containing the following blocks (A), (B), and (C):
(A) A polymer block having an HLB value (Griffin method) in the range of 7 or more and 20 or less.
(B) A polymer block having an HLB value (Griffin method) in the range of 0 or more and less than 7.
(C) A temperature-responsive polymer block having a lower critical solution temperature (LCST) in water in the range of 0°C to 50°C.
(A)HLB値(グリフィン法)が7以上20以下の範囲にある重合体ブロック。
(B)HLB値(グリフィン法)が0以上7未満の範囲にある重合体ブロック。
(C)水に対する下限臨界溶解温度(LCST)が0℃~50℃の範囲にある温度応答性重合体ブロック。 8. A method for producing a nonwoven fabric according to any one of claims 1 to 7 , comprising a step of coating a surface of a nonwoven fabric made of a plastic having a true density of 1.0 to 1.5 g/cm3 and a water absorption rate of less than 0.3% with a block copolymer containing the following blocks (A), (B), and (C):
(A) A polymer block having an HLB value (Griffin method) in the range of 7 or more and 20 or less.
(B) A polymer block having an HLB value (Griffin method) in the range of 0 or more and less than 7.
(C) A temperature-responsive polymer block having a lower critical solution temperature (LCST) in water in the range of 0°C to 50°C.
12. The cell culture method according to claim 11, further comprising a cooling cell detachment step of detaching the cells from the microcarriers using a cooling liquid containing a chelating agent.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2025526106A JPWO2024253070A1 (en) | 2023-06-06 | 2024-06-03 |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2023-093313 | 2023-06-06 | ||
| JP2023093313 | 2023-06-06 | ||
| JP2023143854 | 2023-09-05 | ||
| JP2023-143854 | 2023-09-05 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2024253070A1 true WO2024253070A1 (en) | 2024-12-12 |
Family
ID=93796032
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2024/020258 Ceased WO2024253070A1 (en) | 2023-06-06 | 2024-06-03 | Temperature-responsive nonwoven fabric |
Country Status (2)
| Country | Link |
|---|---|
| JP (1) | JPWO2024253070A1 (en) |
| WO (1) | WO2024253070A1 (en) |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH1156353A (en) * | 1997-11-20 | 1999-03-02 | Nippon Bio Serapii Kk | Culture of adherent cell such as animal tissue by using pet (polyethylene terephthalate) nonwoven fabric |
| WO2004058936A1 (en) * | 2002-12-25 | 2004-07-15 | National Institute Of Advanced Industrial Science And Technology | Cell separation and collection apparatus and separation and collection method |
| JP2018174919A (en) * | 2017-04-12 | 2018-11-15 | 東ソー株式会社 | Block copolymer and surface treating agent using the same |
| JP2020110140A (en) * | 2019-01-11 | 2020-07-27 | 東ソー株式会社 | Cell culture method |
| JP2022073687A (en) * | 2020-11-02 | 2022-05-17 | 東ソー株式会社 | Cooling recovery method suitable for cytometry |
-
2024
- 2024-06-03 WO PCT/JP2024/020258 patent/WO2024253070A1/en not_active Ceased
- 2024-06-03 JP JP2025526106A patent/JPWO2024253070A1/ja active Pending
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH1156353A (en) * | 1997-11-20 | 1999-03-02 | Nippon Bio Serapii Kk | Culture of adherent cell such as animal tissue by using pet (polyethylene terephthalate) nonwoven fabric |
| WO2004058936A1 (en) * | 2002-12-25 | 2004-07-15 | National Institute Of Advanced Industrial Science And Technology | Cell separation and collection apparatus and separation and collection method |
| JP2018174919A (en) * | 2017-04-12 | 2018-11-15 | 東ソー株式会社 | Block copolymer and surface treating agent using the same |
| JP2020110140A (en) * | 2019-01-11 | 2020-07-27 | 東ソー株式会社 | Cell culture method |
| JP2022073687A (en) * | 2020-11-02 | 2022-05-17 | 東ソー株式会社 | Cooling recovery method suitable for cytometry |
Also Published As
| Publication number | Publication date |
|---|---|
| JPWO2024253070A1 (en) | 2024-12-12 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Chen et al. | Control of cell attachment on pH-responsive chitosan surface by precise adjustment of medium pH | |
| JP6638706B2 (en) | Block copolymer and surface treatment agent using the same | |
| JP2018174919A (en) | Block copolymer and surface treating agent using the same | |
| Verma et al. | Three-dimensional multiscale fiber matrices: development and characterization for increased HepG2 functional maintenance for bio-artificial liver application | |
| JP6150071B2 (en) | Organosilicon compound and silane coupling agent containing the same | |
| JP2020110140A (en) | Cell culture method | |
| Xia et al. | Cell attachment/detachment behavior on poly (N-isopropylacrylamide)-based microgel films: the effect of microgel structure and swelling ratio | |
| WO2005001019A1 (en) | Container for germ layer formation and method of forming germ layer | |
| WO2019035436A1 (en) | Culture substrate for pluripotent stem cell and method for producing pluripotent stem cell | |
| JPWO2018198495A1 (en) | Temperature-responsive cell culture substrate and method for producing the same | |
| JP2013506416A (en) | Methods and kits for cell release | |
| WO2024253070A1 (en) | Temperature-responsive nonwoven fabric | |
| Gutsche et al. | N‐acetylglucosamine and adenosine derivatized surfaces for cell culture: 3T3 fibroblast and chicken hepatocyte response | |
| Krasteva et al. | Influence of polymer membrane porosity on C3A hepatoblastoma cell adhesive interaction and function | |
| Pettersson et al. | Cell expansion of human articular chondrocytes on macroporous gelatine scaffolds—impact of microcarrier selection on cell proliferation | |
| JP7647047B2 (en) | Low tissue factor activity cells and method for producing same | |
| JP7250248B2 (en) | BLOCK COPOLYMER, SURFACE TREATMENT AGENT AND MEMBRANE CONTAINING THE SAME, AND CELL CULTURE EQUIPMENT AND CELL CULTURE METHOD USING THE SAME | |
| WO2022202911A1 (en) | Temperature-responsive polymer surface treatment agent | |
| WO2024253069A1 (en) | Temperature-responsive microcarrier | |
| Park et al. | Phenotype of hepatocyte spheroids in Arg-GLY-Asp (RGD) containing a thermo-reversible extracellular matrix | |
| JP2018164412A (en) | Cell aggregate formation substrate, cell aggregate formation method, substance screening method, cell function search method, cell culture substrate coating agent, and cell aggregate formation accelerator | |
| JPH06153905A (en) | Cell culture base and method for cell culture | |
| JP2025127417A (en) | Expansion culture method using temperature-responsive microcarriers | |
| JPWO2024075722A5 (en) | ||
| JP2025134229A (en) | Method for producing temperature-responsive beads |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24819300 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2025526106 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2025526106 Country of ref document: JP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |