WO2024254540A2 - Treatment of amyotrophic lateral sclerosis with extracellular vesicle composition - Google Patents
Treatment of amyotrophic lateral sclerosis with extracellular vesicle composition Download PDFInfo
- Publication number
- WO2024254540A2 WO2024254540A2 PCT/US2024/033123 US2024033123W WO2024254540A2 WO 2024254540 A2 WO2024254540 A2 WO 2024254540A2 US 2024033123 W US2024033123 W US 2024033123W WO 2024254540 A2 WO2024254540 A2 WO 2024254540A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- hsa
- mir
- subject
- alpha
- alsfrs
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K35/00—Medicinal preparations containing materials or reaction products thereof with undetermined constitution
- A61K35/12—Materials from mammals; Compositions comprising non-specified tissues or cells; Compositions comprising non-embryonic stem cells; Genetically modified cells
- A61K35/28—Bone marrow; Haematopoietic stem cells; Mesenchymal stem cells of any origin, e.g. adipose-derived stem cells
Definitions
- ALS Amyotrophic lateral sclerosis
- ALS is a devastating terminal nenrodegenerative disease affecting approximately 4.5 out of 100,000 people.
- ALS is the most common type of motor neuron disease and third most common neurode generative disease behind Alzheimer’s disease and Parkinson’s disease.
- ALS is a nervous system disease that affects the brain and spinal cord, causing the loss of muscle control.
- Approximately 4.5 per 100,000 people are living with the disease, but, unfortunately, there has been little advancement in theunderstanding and treatment of ALS.
- the current diagnosis is one of exclusion based on symptoms.
- a method of treating amyotrophic lateral sclerosis (ALS) in a subject in need thereof comprising administering to the subject a composition comprising a therapeutic mesenchymal stem cell (MSC) secretome composition comprising extracellular vesicles, wherein at least 80% of the extracellular vesicles in the therapeutic MSC secretome composition are CD63 + CD9 CDS! .
- MSC mesenchymal stem cell
- the subject has an increase of at least abou t 0.1 point per month in ALS Functional Rating Scale-Revised (ALSFRS-R) scores or has a decline of less than about 3.0 points per month in ALSFRS-R scores after administration compared to ALSFRS-R scores measured prior to administration.
- the subject has an increase of at least about 0.2, 0.3, 0.4, 0.5, 0.6, 0.7, 0.8, 0.9, 1.0, 1.1, 1.2, 1.3, 1.4, 1.5, 1.6, 1.7, 1.8, 1.9, or 2.0 points per month in ALSFRS-R scores after administration compared to ALSFRS-R scores measured prior to administration.
- the subject has a decline of less than about 2.9, 2.8, 2.7, 2.6, 2.5, 2.4, 2.3, 2.2, 2.1 , 2.0, 1.9, 1.8, 1.7, 1.6, 1.5, 1.4, 1.3, 1.2, 1.1, 1.0. 0.9. 0.8. 0.7, 0.6. 0.5, 0.4, 0.3, 0.2, or 0.1 points per month in ALSFRS- R scores after administration compared to ALSFRS-R scores measured prior to administration.
- the subject has a history of a decline in ALSFRS-R scores of about 3.0 points per month prior io administration of the therapeutic MSC secretome composition.
- a method of treating amyotrophic lateral sclerosis (ALS) in a subject in need thereof comprising administering to the subject a composition comprising a therapeutic mesenchymal stem cell (MSC) secretome composition comprising extracellular vesicles, wherein the subject has an increase of at least about 0.1 point per month in ALS Functional Rating Scale-Revised (ALSFRS-R) scores or has a deciinc of less than about 3.0 points per month in ALSFRS-R scores after administration compared to ALSFRS-R scores measured prior to administration.
- MSC mesenchymal stem cell
- the subject has an increase of at least about 0.2, 0.3. 0.4. 0.5. 0.6, 0.7, 0.8, 0.9, 1.0, 1.1 , 1.2, 1.3, 1.4, 1.5, 1.6, 1.7, 1.8, 1.9, or 2.0 points per month in ALSFRS-R scores after administration compared to ALSFRS-R scores measured prior to administration.
- the subject has a decline of less than about 2.9, 2.8, 2.7, 2.6, 2.5, 2.4, 2.3, 2.2, 2.1, 2.0, 1.9, 1.8, 1.7, 1.6, 1.5, 1.4, 1.3, 1.2, 1.1 , 1.0, 0.9, 0.8, 0.7, 0.6, 0.5, 0.4, 0.3, 0.2, or 0.1 points per month in ALSFRS-R scores after administration compared to ALSFRS-R scores measured prior to administration.
- the subject has a history' of a decline in ALSFRS-R scores of about 3.0 points per month prior to administration of the therapeutic MSC secretome composition.
- the extracellular vesicles in the therapeutic MSC secretome composition are CD63 f CD9 C DS ;
- the therapeutic MSC secretome composition comprises one or more of the following proteins: Ferritin, NUP85, LAMP2, GPR1 15, Setpin Fl, OPN, PAI-1, DAPPL Cathepsin B, Semaphorin 6C, PDGF R alpha, Sortilin, Scrpin B6, Dkk-3, Thrombomodulin, PF4, MIF, Periosiin, Fur in, TIMP- 1, Decorin, PCK.1 , CD99, CD63, CD9, CD81 , Transferrin, DcR3, Lumican, TIMP-2, SLITRK5, FAP, Artemin, DPPIL cIAP-1 , Pentraxin 3, Visfatin, Neprilysin, Albumin, Galectin-1, UNC5H3, IL-20 R beta, SREC-11, JAM-C,
- the therapeutic MSC secretome composition comprises one or more of the following nucleic acids: hsa-let-7a-5p, hsa-let-7b-5p, hsa-let-7c-5p, hsa-let-7d-3p, hsa-let-7e-5p, hsa-let-7g-5p, hsa-let-7i, hsa-let-7i-5p, hsa-miR-100-5p, hsa-miR-103a-3p, hsa- miR-106a-5p, hsa-miR-106b-5p, hsa-mir-lOb, hsa-miR-10b-5p, hsa-mir-1246, hsa-miR-1246, hsa-miR-125a-5p, hsa-miR-125b-5p, hsa-miR-130a-3p,
- bsa-miR-486-5p hsa-miR-570-3p, hsa- miR-574-3p, hsa-miR-663a, hsa-miR-874-3p, hsa-mir-92a-1, hsa-mir-92a-2, hsa-miR-92a- 3p.
- hsa*miR-92b-3p hsa-mir-93, hsa-miR-93-5p, hsa-niiR-940, hsa-miR*99a-5p, or hsa-.mi.R- 99b-5p.
- the composition is produced by: (a) culturing bone marrow- derived MSCs under the following conditions to produce an MSC conditioned media: (i) oxygen tension below 5%; and (ii) culture media having a pH below 7: (b) harvesting the MSC conditioned media; and (c) formulating the MSC conditioned media to produce the therapeutic MSC secretome composition, wherein the therapeutic MSC secretome composition comprises proteins and extracellular vesicles produced by the bone m arrow - derived MSCs in step (a).
- the culture media is serum- tree.
- the culture media has a glucose concentration below 4.5 g L.
- the subject has spinal onset type ACS. In some embodiments, the subject has bulbar onset type ALS. In some embodiments, the subject has advanced ALS. In some embodiments, the subject presents with limb-related symptoms. In some embodiments, the subject presents with dysphagia or speech difficulties. In some embodiments, the treating delays the progression of ALS.
- the subject carries one or more amino acid variations in SOD I protein. In some embodiments, the one or more amino acid variations comprise G93A. In some embodiments, the subject carries one or more dipeptide repeats in C9ORF72 protein. In some embodiments, the one or more dipeptide repeats comprise poly-GA, poly-GP poly-GR, poly- PA, or poly -PR.
- the subject is a human.
- the bone marrow -derived MSCs are derived from human bone marrow.
- administering comprises intravenous administration.
- the dosage of the therapeutic MSC secretome composition administered to the subject is a cell-equivalent dosage of 0.7 to 7 million cells/kg.
- the therapeutic MSC secretome composition comprises 4x10 10 to 10x 10 10 cells/ml.
- the therapeutic MSC secretome composition comprises 5x10 11 to 1.5xl0 12 extracellular vesicles.
- the composition is administered monthly for two or more months, or once every 1, 2, or 3 or more months.
- a composition comprising a therapeutic mesenchymal stem cell (MSC) secretome composition for treating amyotrophic lateral sclerosis (ALS) in a subject in need thereof, the method comprising: (a) culturing bone marrow-derived MSCs under the following conditions to produce an MSC conditioned media: (i) oxygen tension below 5%: and (ii) culture media having a pH below' 7; (b) harvesting the MSC conditioned media; and (c) formulating the MSC conditioned media to produce the therapeutic MSC secretome composition, wherein the therapeutic MSC secretome composition comprises proteins and extracellular vesicles produced by the bone marrow-derived MSCs in step (a).
- MSC mesenchymal stem cell
- the culture media is serum- free. In some embodiments, the culture media has a glucose concentration below 4.5 gZL. In some embodiments, at least 80% of the extracellular vesicles in the therapeutic MSC secretome composition are CD63" CD9 CD81 In some embodiments, the bone marrow -derived MSCs are derived from human bone marrow.
- the therapeutic MSC secretome composition further comprises one or more of the following proteins: Ferritin, NUP85, I..AMP2, GPR1 15, Serpin F1, OPN, PAI-1 , DAPP1, Cathepsin B, Semaphorin 6C, PDGF R alpha, Sortiiin, Serpin B6, Dkk-3, Thrombomodulin, PF4, MIF, Periostin, Furin, TIMP-l, Decorin, PCK 1, CD99, CD63, CD9, CD81, Transferrin, DcR3, Lum lean, TIMP-2, SLITRK5, FAP, Artemin, DPPII, cIAP-1, Pentraxin 3, Visfatin, Neprilysin, Albumin, Galectin-1, UNC5H3, TL-20 R beta, SREC-1I, JAM-C, TNF RI, htPAPP-A, cNOS, MSP R, TPP1, LAMP1 , B2M, N
- Thrombospondin- 2 SMPD1, B2M, MFRP, LRP-6, ST3GAL1, NCAM-1 (CD56), Granzyme B, Adipo nedin, IL-22BP, TPST2, PD-ECGF, LH, LEDGE, Cyr61, ULBP-3, IFNb, THSD 1 , FGF-23, LAMA4, Adipsin, AIF, SorCS2, SULT2A1 , CD39L2, Insulin R, HIF-1 alpha, OX40 Ligand, Pax3, IJCH-L3, cMASP3, Langerin, Desmin, SOX9, ST6GAL1 , MEP1 B, CD99-L2, Plexin A4, Semaphorin 4D, ROBO2, PDX-1 , APRIL, Neurturin, Kremen-2, EMMPRIN, Activin RIB, Neuroligin 2, Epiregulin, CASA, MMP-12, GALNT2, CEACAM-5, VEGF R
- MEPE Activin RIIA, ANG-2, Cochlin, Presenilin 1, NPTXR, SLAM, COMT, SPHK1, RBP4, Nectin-1, GUSB,Nidogen-2, IL-17F, SR-AI, TAFA2, N-Cadherin, IL-17B, IL-17 RC, MIP-3b, Cystatin C, Cystatin D, AMSH, FcERI, CLEC10A, HGF R, ANG-1, Prolactin R, FGF-20, CD28, Nogo-A, HSD17B1, IL-19, Enteropeptidase, Cathepsin E, TSLP, TCN2, GDF-15, Epimorphin, GRKS, PD-1, Serpin A4, ADAM23, NOV, Galectin-2, Neurexin 3 beta, TLR3, Sirtuin 2, Numb, IL- 28 R alpha, IL-
- VE- Cadherin Procalcitonin, SOX15, Kallikrein 11, BCMA, Dectin-2, EpCAM, HCC-4, TGFa, IP-10, BLAME, CILP-1, PIGF, LOX-1, MCP-2, Resistin, HVEM, ENPP-7, Syndecan-4, IL-2 Rg, MICA, Dopa Decarboxylase, NPDC-1, MCP-4, EG-VEGF, Glycoprotein V, Semaphorin 4G, IL-12p40, PSA-total, IL-15, MAP1D, CLq, TNF4, Dtk, Endoglin, ENA-78, Reg3A, MIP- lb, FGF-17, IL-6R, IL-8, Galectin-8, CA4, Cystatin E M, FUT8, B7-H3, GCP-2, CD40L, MDC, 4-1BB, HO-1, SOST, S100A13, Kallikrein 7, or IL-13.
- the extracellular vesicles comprise one or more of the following nucleic acids: hsa-let-7a-5p, hsa-let-7b-5p, hsa-let-7c-5p, hsa-let-7d-3p, hsa-let-7e- 5p, hsa-let-7g-5p, hsa-let-7i, hsa-let-7i-5p, hsa-miR-100-5p, hsa-miR-103a-3p, hsa-miR- 106a-5p, hsa-miR-106b-5p, hsa-mir-lOb, hsa-miR-10b-5p, hsa-mir-1246, hsa-miR-1246, hsa- miR-125a-5p, hsa-miR-125b-5p, hsa-miR-130a-3
- ALS amyotrophic lateral sclerosis
- the subject has spinal onset type ALS.
- the subject has bulbar onset type ALS.
- the subject has advanced ALS.
- the subject presents with Limb-related symptoms.
- the subject presents with dysphagia or speech difficulties.
- the treating delays the progression of ALS.
- the subject carries one or more amino acid variations in SOD1 protein.
- the one or more amino acid variations comprise G93A.
- tire subject carries one or more dipeptide repeats in C9ORF72 protein.
- the one or more dipeptide repeats comprise poly-GA, poly-GP poly-GR, poly-PA, or poly-PR.
- the subject is a human.
- the composition is intravenously administered to the subject.
- the subject has an increase of at least about 0.1 point per month in ALS Functional Rating Scale-Revised (ALSFRS-R) scores or has a decline of less than about 3.0 points per month in ALSFRS-R scores after administration compared to ALSFRS-R scores measured prior to administration.
- ALSFRS-R ALS Functional Rating Scale-Revised
- the subject has an increase of at least about 0.2, 0.3, 0.4, 0.5, 0.6, 0.7, 0.8, 0.9, 1.0, 1.1 , 1.2, 1.3, 1.4, 1.5, 1.6, 1 .7, 1 .8, 1 .9, or 2.0 points per month in ALSFRS-R scores after administration compared to ALSFRS-R scores measured prior to administration.
- the subject has a decline of less than about 2.9, 2.8, 2.7, 2.6, 2.5, 2.4, 2.3, 2.2, 2.1, 2.0, 1.9, 1.8, 1.7, 1.6, 1.5, 1.4, 1.3, 1.2, 1.1 , 1.0, 0.9, 0.8, 0.7, 0.6, 0.5, 0.4, 0.3, 0.2, or 0.1 points per month in ALSFRS- R scores after administration compared to ALSFRS-R scores measured prior to administration.
- the subject has a history' of a decline in ALSFRS-R scores of about 3.0 pointe per month prior to administration of the therapeutic MSC secretome compositi on.
- the dosage of the therapeutic MSC secretome composition administered to the subject is a cell-equivalent dosage of 0.7 to 7 million cells/kg.
- the therapeutic MSC secretome composition comprises 4x10 10 to 10x10 10 cells/ml.
- the therapeutic MSC secretome composition comprises 5x10 11 to 1.5x10 12 extracellular vesicles.
- the composition is administered monthly tor two or more months, or once every 1, 2, or 3 or more months.
- compositions comprising a therapeutic mesenchymal stem cell (MSC) secretome composition comprising extracellular vesicles in treating amyotrophic lateral sclerosis (ALS) in a subject in need thereof, wherein at least 80% of the extracellular vesicles in the therapeutic MSC secretome composition are CD63- CD9 CD81
- the composition is intravenously administered to the subject.
- the subject has an increase of at least abou t 0,1 point per month in ALS Functional Rating Scale-Re vised (ALSFRS-R) scores or has a decline of less than about 3.0 points per month in ALSFRS-R scores after administration compared to ALSFRS-R scores measured prior to administration.
- the subject has an increase of at least about 0.2, 0.3, 0.4, 0.5, 0.6, 0.7, 0.8, 0.9, 1.0, 1,1, 1.2, 1.3, 1.4, 1.5, 1.6,
- the subject has a decline of less than about 2.9, 2.8, 2.7, 2.6, 2.5, 2.4, 2.3, 2.2, 2, 1, 2.0, 1.9, 1.8, 1.7, 1.6, 1 .5, 1.4, 1.3, 1.2, 1.1, 1.0, 0.9, 0.8, 0.7, 0.6, 0.5, 0.4, 0.3, 0.2, or 0.1 points per month in ALSFRS- R scores after administration compared to ALSFRS-R scores measured prior to administration.
- the subject has a history of a decline in ALSFRS-R scores of about 3.0 points per month prior to administration of the therapeutic MSC secretome composition.
- a composition comprising a therapeutic mesenchymal stem cell (MSC) secretome composition comprising extracellular vesicles in treating amyotrophic lateral sclerosis (ALS) in a subject in need thereof, wherein the subject has an increase of at least about 0.1 point per month in ALS Functional Rating Scale-Revised (ALSFRS-R) scores or has a decline of less than about 3.0 points per month in ALSFRS-R scores after administration compared to ALSFRS-R scores measured prior to administration, [0027] In some embodiments, the subject has an increase of at least about 0.2, 0.3, 0.4, 0.5, 0.6, 0.7, 0.8, 0.9, 1 .0, 1.1, 1 .2, 1 .3, 1 .4, 1 .5, 1 .6, 1 .7, 1 .8, 1 .9, or 2.0 points per month in ALSFRS-R scores after administration compared to ALSFRS-R scores measured prior
- the subject has a history of a decline in ALSFRS-R scores of about 3.0 points per month prior to administration of the therapeutic MSC secretome composition.
- the therapeutic MSC secretome composition further comprises one or more of the following proteins: Ferritin, NUP85, LAMP2, GPR115, Serpin Fl, OPN, PAI-1, DAPP1, Cathepsin B, Semaphorin 6C, PDGF R alpha, Sortilin, Serpin B6, Dkk-3, Thrombomodulin, PF4, MIF, Periostin, Furin, TIMP-1, Decorin, PCK1, CD99, CD63, CD9, CD81, Transferrin, DcR3, Lumican, TIMP-2, SLITRK5, FAP, Artemin, DPPII, cIAP-1, Pentraxin 3, Visfatin, Neprilysin, Albumin, Galectin-1, UNC5H3, IL-20 R beta, SREC-II, JAM-C
- the extracellular vesicles comprise one or more of the following nucleic acids: hsa-let-7a-5p, hsa-let-7b-5p, hsa-let-7c-5p, hsa-let-7d-3p, hsa-let-7e- 5p, hsa-let-7g-5p, hsa-let-7i, hsa-let-7i-5p, hsa-miR-100-5p, hsa-miR-103a-3p, hsa-miR- 106a-5p, hsa-miR-106b-5p, hsa-mir-lOb, hsa-miR-10b-5p, hsa-mir-1246, hsa-miR-1246, hsa- miR-125a-5p, hsa-miR-125b-5p, hsa-miR-130a-3
- the composition is produced by: (a) culturing bone marrow- derived MSCs under the following conditions to produce an MSG conditioned media: (i) oxygen tension below 5%; and (ii) culture media having a pH below 7; (b) harvesting the M.SC conditioned media; and (c) formulating the MSG conditioned media to produce the therapeutic MSC sec-retome composition, wherein the therapeutic MSC secretome composition comprises proteins and extracellular vesicles produced by the bone marrow- derived MSCs in step (a), [0031]
- the culture media is serum- free.
- the cul ture media has a glucose concentration below 4.5 g 1...
- the subject has spinal onset type ALS. In some embodiments, the subject has bulbar onset type ALS. In some embodiments, the subject has advanced ALS. In some embodiments, the subject presents with limb-related symptoms. In some embodiments, the subject presents with dysphagia or speech difficulties. In some embodiments, the treating delays the progression of ALS.
- the subject carries one or more amino acid variations in SOD1 protein. In some embodiments, the one or more amino acid variations comprise G93A. In some embodiments, the subject carries one or more dipeptide repeats in C9ORF72 protein. In some embodiments, the one or more dipeptide repeats comprise poly-GA, poly-GP poly-GR, poly- PA, or poly -PR.
- the subject is a human.
- the bone marrow -derived MSCs are derived from human bone marrow,
- the composition is intravenously administered to the subject.
- the dosage of the therapeutic MSC see.ret.ome composition administered to the subject is a cell-equivalent dosage of 0.7 to 7 million cells/kg.
- the therapeutic MSC secretome composition comprises 4x10 10 to 10x10 10 cells/ml.
- the therapeutic MSC secretome composition comprises 5x10 11 to 1.5x10 12 extracellular vesicles.
- the composition is administered monthly tor two or more months, or once every 1, 2, or 3 or more months.
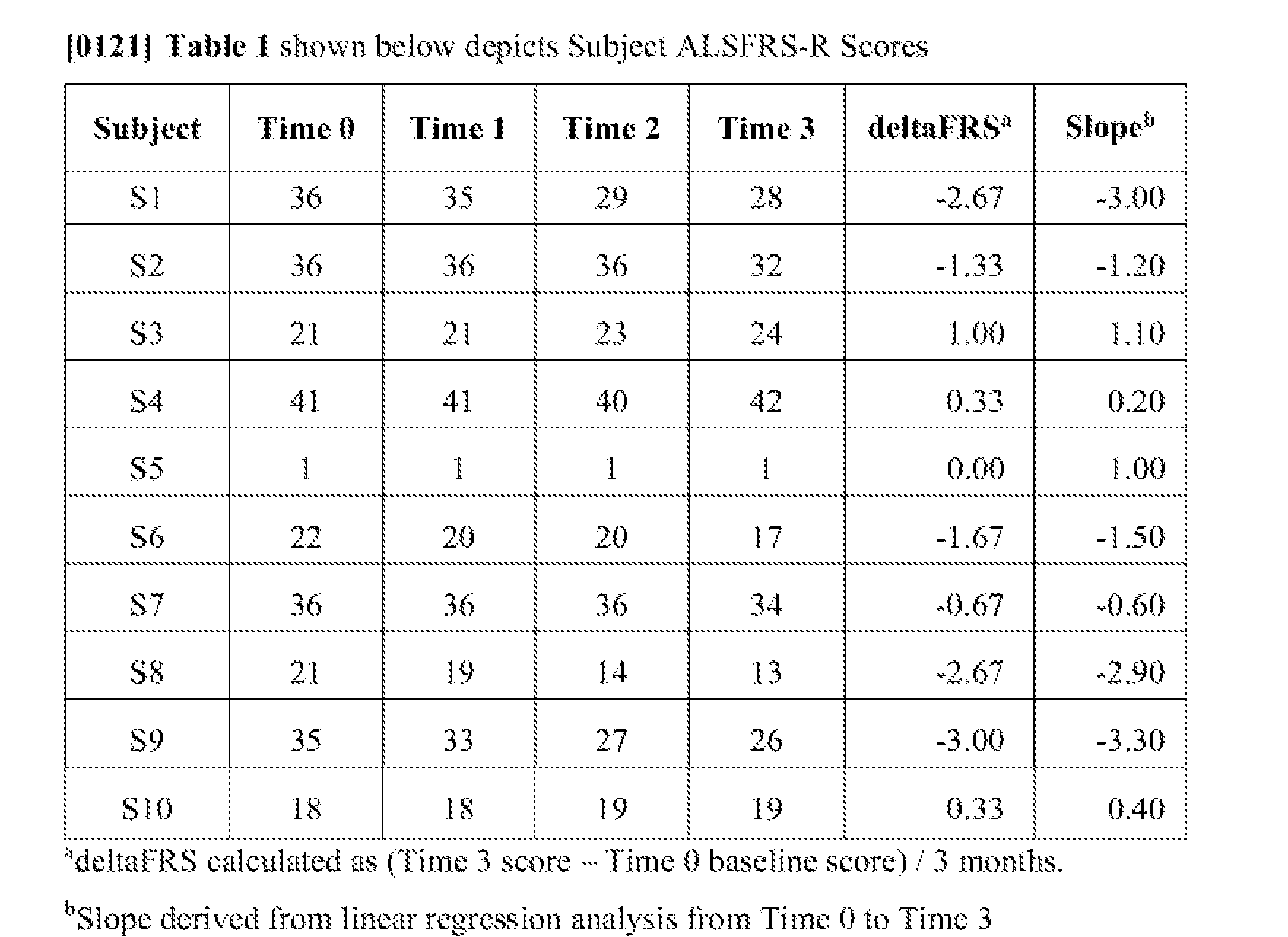
- FIG. I A illustrates the differences in raw ALSFR.S-R. scores amongst all subjects at each measurement time period.
- FIG. 1B shows the lilted linear regression analysis for each subject over time to illustrate disease progression during the study.
- the term “about” or “approximately” can mean within an acceptable error range for the particular value, which may depend in part on how the value is measured or determined, e.g., the limitations of the measurement system. For example, “about” can mean within 1 or more than I standard deviation. Alternatively, “about” can mean a range of up to 20%, up to 10%, up to 5%, or up io 1% of a given value. Alternatively, particularly with respect to biological systems or processes, the term can mean within an order of magnitude, within 5- fokl, or within 2 -fold, of a value. Where particular values are described in the application and claims, unless otherwise stated the term “about” meaning within an acceptable error range for the particular value should be assumed.
- MSC secretome including, but not limited to a MSC exosome (with or without growth factors) .referred to herein as an extracellular vesicle isolate product (E VIP)
- E VIP extracellular vesicle isolate product
- a primary trophic property of MSC s is the secretion of growth factors and exosomes to induce cell proliferation and angiogenesis.
- Exosomes express mitogenic proteins such as transforming growth factor-alpha (TGF-a), TGFp, hepatocyte growth factor (HGF), epithelial growth factor (EGF), basic fibroblast growth factor (FGF-2) and insulin-1ike growth factor- 1 (IGF-1), These increase fibroblast, epithelial and endothelial cell division.
- VEGF vascular endothelial growth factor
- IGF-1 IGF-1
- EGF hepatocyte growth factor
- FGF-2 basic fibroblast growth factor
- IGF-1 insulin-1ike growth factor- 1
- MSCs assist via paracrine mechanisms and modulate the regenerative environment via anti-inflammatory and immunomodulatory' mechanisms.
- inflammatory molecules such as interleukin- 1 (IL-1), IL-6, IL-2, IL-12, tumor necrosis factor-a (TNF-a) and interferon-gamma (INF-y)
- IL-1 interleukin-1
- IL-6 interleukin-6
- IL-2 tumor necrosis factor-a
- INF-y interferon-gamma
- the key immunomodulatory cytokines include prostaglandin 2, TGF-131, HGF, SDF-1, nitrous oxide, indoleaniine 2, 3 -dioxygenase, IL-4, IL- 10, IL-1 receptor antagonist and soluble tumor necrosis factor-a receptor, MSCs prevent proliferation and function of many inflammatory immune cells, including T-cells, natural killer cells, B- cells, monocytes, macrophages, and dendritic cells. Although MSCs across species are able to regulate T-cell activity, the mechanisms are not identical across mammalian species.
- a characteristic of chronically inflamed environments is a persistent imbalance in the types of helper T-cells and macrophages. MSC exosomes indirectly promote the transition of TH1 to TH2 cells by reducing INF-y and increasing IL-4 and IL- 10. The .restored TH1/TH2 balance has been shown to improve tissue regeneration in cartilage, muscle, and other soft tissue injuries, alleviate symptoms of autoimmune diseases, and have an anti-diabetic effect.
- Ml proinflammatory, anti- angiogenic and tissue growth inhibition
- M2 anti-inflammatory, pro-remodeling and tissue healing
- compositions comprising a therapeutically effective amount of a MSC secretome (such as, for example, including, but not limited to MSC growth factor, MSC cxosome, MSC extracts and/or extracellular vesicle comprising compositions) and one or more biomolecules (such as, for example, a peptide, polypeptide, protein, siRNA, shRNA, and/or microRNA (miRNA)).
- MSC secretome such as, for example, including, but not limited to MSC growth factor, MSC cxosome, MSC extracts and/or extracellular vesicle comprising compositions
- biomolecules such as, for example, a peptide, polypeptide, protein, siRNA, shRNA, and/or microRNA (miRNA).
- the therapeutic composition comprises 1 , 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13. 14, 15, 16, 17, 18, 19, 20. 21 , 22, 23, 24, 25, 26, 27, 28, 29. 30, 31 , 32, 33, 34, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, or 100 or more of the following proteins, in any combination, or all of the following proteins; Ferritin, NIJP85, LAMP2, GPR115, Serpin Fl, OPN, PAI-1, DAPP1, Cathepsin B, Semaphorin 6C, PDGF R alpha, Sortilin, Serpin B6, Dkk-3, Thrombomodulin, PF4, M1F, Perioslin, Furin, T1MP-1 , Decorin, PCK1 , CD99, CD63, CD9, CD81, Transferrin, DcR3, Lumican, TIMP-2, SL1TRK5, FAP, Artemin, DPPII, cIAP
- IL-22BP IL-22BP
- TPST2 PD-ECGF
- LH LEDGF
- Cyr61 ULBP-3
- IFNb THSD1
- FGF-23 LAMA4
- Adipsin Adipsin
- AIF SorCS2
- SULT2A1 CD39L2 Insulin R, HIF-1 alpha, OX40 Ligand, Pax3, UCH-L3, cMASP3, Langerin, Desmin, SOX9, ST6GAL1, MEP1B, CD99-L2, Plexin A4, Semaphorin 4D, ROBO2, PDX-1, APRIL, Neurturin, Kremen-2, EMMPRIN, Activin RIB, Neuroligin 2, Epiregulin.
- VEGF vascular endothelial growth factor
- IL-7 alpha
- GPR56 CE AC AM-3
- MCEMP 1 MCEMP 1
- FABP2 Plexin B3, MEPE
- Activin RIA ANG-2
- Cochlin Presenilin 1
- NPTXR NPTXR
- SLAM COMT
- SPHK1 RBP4
- Neciin-1 GUSB
- Nidogen-2 IL-17F.
- SR-AI TAFA2
- N-Cadherin N-Cadherin
- Cystatin C Cystatin C.
- Cystatin D AMSH, FcERI, CLEC10A, HGF R, ANG-1, Prolactin R, FGF-20, CD28, Nogo-A, HSD17B 1, IL- 19, Enteropeptidase, Cathepsin E, TSLP.
- TCN2 TCN2, GDF-15, Epimorphin, GRKS, PD-1, Serpin A4, ADAM23, NOV, Galeciin-2, Neurexin 3 beta, TLR3, Sirtuin 2, Numb, IL- 28 R alpha, IL-33, Lin28, FCRL1, KLF4, NKp30, Lymphotactin, Cystatin SN, JAM-A, Calreuculin-2, ErbB4, BMP-8, IL-27 Ra, Fas, IL-4 Ra, Kallikrein 14, Matriiin-3, Olig2, Kallikrein 12, CA1 3, IL-9, Neclin-3, MP1F-1 , Cystatin S, ADA, IL-2 Rb, GFR alpha-1 , Smad4, ICAM-1, MEF2C, TREM-1, L-Selectin, Hepsin, CD42b, MCSF.
- EGF R TAFAS, Galectin-9, vWF-A2, TACE, Aciivin RIM, Cathepsin S, LDL R, BMPR-IA, OX40, IL- 13 R2, B7- H4, MMP-13, ANGPTL7, TRAIL R4, IGSF4B, Sirtuin 5, PEAR1 , SH2D1A, Cerberus 1, GDF-11, Nrf2, TROP-2, NUDTS, ROR2, EphB4, Glypican 1, LAP(TGFb1), Gash, Contactin- 1, IL-27, UNC5H4, ICAM-2, MBL, HS3ST3B1, RCOR1, IL- 10 Rb, XEDAR, IL-22, PILR-alpha, NRG 1- 131 , FABP4, RGM-A, RELT, TrkC, CSa, SREC-1, Nestin, TPO, ErbB3, Kirrel3, FLRT1, Galectin-3, CXCL16, J
- CTRC CEACAM-1, TARC, MIP-3a, SDF-1b, NKp46, MCP-3, IL-32 alpha, TGFb3 FOLR2, CD58, IL-23, CD36, TNFb, Shh-N, Ficol in- 1, Reg4, ILT2, Mer, TREM-2, Fli-3L, CDS, IL-6, CD229, Insulin, Syntaxin 6, GRO, Bcl-w, Lipocalin-2, PDGF-AA, IL-2 Ra, Angiogenin, LYVE-1 , CD4, R AGE, CDNF, Brevican, NAP-2, PU.l, EDAR,
- ADAMTS13 Kynureninase, PTH1R, IFN-gamma R1 , CrkL, B7-1, PARC, Draxin, VE- Cadherin, Procalcitonin, SOX 15, Kall
- Extracellular vesicles are small membrane bound spheres containing proteins and RNA (of which exosomes are a subset). Exosomes are small (e.g., 20 - 150 nm) diameter lipid bilayer vesicles secreted by cells to enable paracrine communication. Other EV populations are derived directly from the plasma membrane or are formed during apoptosis (apoptotic bodies).
- compositions comprising a therapeutically effective amount of an MSC secretome (such as, for example, including, but not limited to MSC growth factor, MSC exosome, MSC extracts and/or extracellular vesicle comprising compositions).
- the therapeutic composition comprises 1 , 2, 3, 4, 5, 6, 7, 8, 9, 10, 1 1, 12, 13, 14, 15, .16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31 , 32, 33, 34, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, or 100 or more of the following nucleic acids, in any combination, or all of the following nucleic acids: hsa-1et-7a-5p, hsa-1et-7b-5p, hsa-1et-7c-5p, hsa-1et-7d-3p, hsa-1et-7e-5p, hsa-1et-7g-5p, hsa-1et-7i, hsa-1et-7i-5p, hsa-miR-100-5p, hsa-miR- 103a-3p, hsa-miR-106a-5p, hsa-miR-106b-5p
- Exemplary microRNA content may include human miRNA sequences hsa-1et-7a-5p, hsa-1et-7b-5p, hsa-1et-7c-5p, hsa-1et-7g-5p, hsa-1et-7i-5p, bsa-mi.R-214-3p, and hsa-miR-27a- 3p, which all have binding sites in mRNA for TMPRSS2.
- the therapeutic composition comprises extracellular vesicles with a phenotype of CD63" CD9" CD81".
- at least 70, 75, 80, 85, 90, 91, 92, 93, 94, or 95% of the extracellular vesicles in the therapeutic composition are CD63 + CD9- CD81-.
- At least 50, 60, 70, 80, 85, 90, 91, 92, 93, 94, or 95% of the extracellular vesicles in the therapeutic composition are CD9 , In some embodiments, at least 50, 60, 70, 80, 85, 90, 91 , 92, 93, 94, or 95% of the extracellular vesicles in the therapeutic composition are CD81 -
- the MSCs cultured to produce the therapeutic composition have the capacity to undergo trilineage differentiation in vitro toward adipocyte, osteoblast, and chondrocyte phenotypes.
- the MSCs are positive for CD73, CD105, CD 166, and CD90 and are negative for CD 14, CD31 , CD34, and CD45.
- the MSC secretome comprises exosomes and growth factors.
- the growth factors and exosomes can be allogenic or autogenic.
- the growth factors and exosomes can be derived from any cell in the human body, such as from ectodermal cells, endodermal cells, or mesodermal cells.
- the MSC secretomes may comprise mesenchymal stem cell (MSC) derived growth factors, MSC derived exosomes, or both MSC derived growth factors and exosomes.
- the method further comprises adding at least one additive with the exosomes and growth factors.
- MSCs under appropriate wound healing conditions may produce suitable therapeutic agents, such as exosomes and growth factors, that can provide therapy for inflammatory lung diseases.
- the MSC secretome composition further comprises prostaglandin E2 (PGE2), transforming growth factor 131 (TGF-I31), hepatocyte growth factor (HGF), stromal cell derived factor- 1 (SDF-1 ), nitric oxide, indoleamine 2,3-di oxygenase, interieukin-4 (IL-4), IL-6, interleukin-10 (IL- 10), IL-1 receptor antagonist and soluble TM F-a receptor, insulin -1ike growth factors, fibroblast growth factors (FGF) 1 -23 (especially, FGF1 and FGF2), bone morphogenetic proteins (BMPs) 1 -15, epidermal growth factor (EGF), transforming growth factor-a (TGF-a) macrophagestimulating protein (MSP), platelet derived growth factor (PLGF), vascular endothelial growth factor (ethelial growth
- Embodiments of a therapeutic composition described herein may comprise proteins and microRNAs, some of which may be embedded in or surrounded by a lipid membrane to create vesicles in the size range of about >20nm io about 200 nm in size.
- the number of vesicles within the composition may be between about 1 million to about 100 billion vesicles per ml. when suspended or about 10 million to about 1 trillion when formulated as a lyophilized powder.
- compositions described herein may be administered in vivo in a pharmaceutically acceptable carrier.
- pharmaceutically acceptable is meant a material that is not biologically or otherwise undesirable, i.e., the material may be administered to a subject, along with the nucleic acid or vector, without causing any undesirable biological effects or interacting in a deleterious manner with any of the other components of the pharmaceutical composition in which it is contained.
- the carrier would naturally be selected to minimize any degradation of the active ingredient and to minimize any adverse side effects in the subject, as would be well known io one of skill in the art.
- compositions may be administered orally, parenterally (e.g., intravenously), by intramuscular injection, by intraperitoneal injection, transdermally, extracorporeally, topically or the like, including topical intranasal administration or administration by inhalant.
- parenterally e.g., intravenously
- intramuscular injection by intraperitoneal injection
- transdermally extracorporeally, topically or the like
- topical intranasal administration means delivery of the compositions into the nose and nasal passages through one or both of the nares and can comprise delivery by a spraying mechanism or droplet mechanism, or through aerosolization of the nucleic acid or vector.
- Administration of the compositions by inhalant can be through the nose or mouth via delivery by a spraying or droplet mechanism such as, for example, a metered-dose inhaler, a dry powder inhaler, a nebulizer, a vaporization device, or the like. Delivery can also be directly to any area of the respiratory system (e.g,, lungs) via intubation.
- a spraying or droplet mechanism such as, for example, a metered-dose inhaler, a dry powder inhaler, a nebulizer, a vaporization device, or the like. Delivery can also be directly to any area of the respiratory system (e.g, lungs) via intubation.
- the exact amount of the compositions required will vary from subject to subject, depending on the species, age, weight and general condition of the subject, the severity of the disorder being treated, mode of administration and the like,
- Parenteral administration of the composition is generally characterized by injection.
- Injectables can be prepared in conventional forms, either as liquid solutions or suspensions, solid forms suitable for solution of suspension in liquid prior to injection, or as emulsions.
- Parenteral administration may involve use of a slow release or sustained release system such that a constant dosage is maintained.
- Suitable carriers and their formulations are described in Remington: The Science and Practice of Pharmacy (19th ed.) ed. A.R. Gennaro, Mack Publishing Company, Easton, PA 1995, Typically, an appropriate amount of a phannaceutically-acceptable salt is used in the formulation to render the formulation isotonic.
- the pharmaceutically -acceptable carrier examples include, but are not limited to, saline, Ringer's solution, and dextrose solution, 'Idle pH of the solution is preferably from about 5 to about 8, and more preferably from about 7 to about 7.5, Further carriers include sustained release preparations such as semipermeablc matrices of solid hydrophobic polymers containing the antibody, which matrices are in the form of shaped articles, e.g., films, liposomes or microparticles. It will be apparent to those persons skilled in the art that certain carriers may be more preferable depending upon, for instance, the route of administration and concentration of composition being administered. [0061 ] Pharmaceutical carriers are known to those skilled in the art.
- compositions can be administered intramuscularly or subcutaneously.
- Other compounds will be administered according to standard procedures used by those skilled in the art,
- compositions may include carriers, thickeners, diluents, buffers, preservatives, surface active agents and the like in addition io the molecule of choice.
- Pharmaceutical compositions may also inente one or more active ingredients such as antimicrobial agents, anti -inflammatory agents, anesthetics, and the like,
- the pharmaceutical composition may be administered in a number of ways depending on whether local or systemic treatment is desired, and on the area to be treated.
- Administration may be topically (including ophthalmieahy, vaginally, .rectally, intranasally), orally, by inhalation, or parenterally, for example by intravenous drip, subcutaneous, intraperitoneal or intramuscular injection.
- the disclosed antibodies can be administered intravenously, intraperitoneally, intramuscularly, subcutaneously, intracavity, or transdermally.
- Preparations for parenteral administration include sterile aqueous or non-aqueous solutions, suspensions, and emulsions.
- non-aqueous solvents are propylene glycol, polyethylene glycol, vegetable oils such as olive oil, and injectable organic esters such as ethyl oleate.
- Aqueous carriers include water, alcohoh’c/aqueous solutions, emulsions or suspensions, including saline and buffered media.
- Parenteral vehicles include sodium chloride solution, Ringer's dextrose, dextrose and sodium chloride, lactated Ringer’s, or fixed oils.
- Intravenous vehicles include fluid and nutrient replenishes, electrolyte replenishers (such as those based on Ringer's dextrose), and the like. Preservatives and other additives may also be present such as, for example, antimicrobials, anti-oxidants, chelating agents, and inert gases and the like.
- Formulations for topical administration may include ointments, lotions, creams, gels, drops, suppositories, sprays, liquids and powders. Conventional pharmaceutical carriers, aqueous, powder or oily bases, thickeners and the like may be access ary or desirable.
- compositions for oral administration may include powders or granules, suspensions or solutions in water or non-aqueous media, capsules, sachets, or tablets. Thickeners, flavorings, diluents, emulsifiers, dispersing aids or binders may be desirable.
- compositions may potentially be administered as a pharmaceutically acceptable acid- or base- addition salt, formed by reaction with inorganic acids such as hydrochloric acid, hydrobromic acid, perchloric acid, nitric acid, thiocyanic acid, sulfuric acid, and phosphoric acid, and organic acids such as formic acid, acetic acid, propionic acid, glycolic acid, lactic acid, pyruvic acid, oxalic acid, malonic acid, succinic acid, maleic acid, and fumaric acid, or by reaction with an inorganic base such as sodium hydroxide, ammonium hydroxide, potassium hydroxide, and organic bases such as mono-, di-, trialkyl and aryl amines and substituted ethanolam ines.
- inorganic acids such as hydrochloric acid, hydrobromic acid, perchloric acid, nitric acid, thiocyanic acid, sulfuric acid, and phosphoric acid
- organic acids such as formic acid, acetic acid, propionic acid,
- Effective dosages and schedules for administering the compositions may be determined empirically, and making such determinations is within the skill in the art.
- the dosage ranges for the administration of the compositions are those large enough to produce the desired effect in which the symptoms of the disorder are affected.
- the dosage should not be so large as to cause adverse side effects, such as unwanted cross-reactions, anaphylactic reactions, and the like.
- the dosage will vary with the age, condition, sex and extent of the disease in the patient, route of administration, or whether other drugs are included in the regimen, and can be determined by one of skill in the art.
- the dosage can be adjusted by the individual physician in the event of any counterindications.
- Dosage can vary, and can be administered in one or more dose administrations daily, tor one or several days, and/or one or more dose administrations monthly for two or more months, or once every 1, 2, or 3 or more months.
- Guidance can be found in the literature for appropriate dosages for given classes of ph arm a c eu tic al produc ts .
- the treatment compositions disclosed herein utilize MSC secretomes and/or growth factors derived from mesenchymal stem cells (MSCs).
- MSC seeretome compositions inchiding, but not limited to MSC growth factor, MSC exosome, MSC extracts and/or extracellular vesicle comprising compositions).
- the treatment compositions may be used in the treatment, inhibition, decrease, reduction, amelioration, and/or prevention of conditions such as, for example, amyotrophic lateral sclerosis (A'LS).
- A'LS amyotrophic lateral sclerosis
- MSCs are multipotent cells that have the ability to differentiate into a multitude of cell types including myocytes, chondrocytes, adipocytes, and osteoblasts. Typically, these cells can be found in the placenta, umbilical cord blood, adipose tissue, bone marrow, or amniotic fluid, including perivascular tissue.
- MSC refers to non-terminally differentiated cells including but not limited to .multipotential stern cell, multipotential stromal cell, stromal vascular cells, pericytes, perivascular cells, stromal cells, pluripotent cells, multipotent cells, adipose-derived fibroblast-1ike cells, adipose-derived stromal vascular fraction, adipose-derived MSC, bone marrow-derived fibroblast-1ike cells, bone marrow- derived stromal vascular fraction, bone marrow-derived MSC, tissue-derived fibroblast-1ike cells, adult stem cells, adult stromal cells, keratinocytes, and/or melanocytes.
- MSCs in addition to their differentiation potential, have immunomodulatory 1 abilities resulting in the expression of many different cytokines and growth factors.
- a ”MSC preparation” or “MSC seeretome composition” refers to a composition comprising MSC growth factors, MSC exosomes, extracellular vesicles, extracellular vesicle isolate product (EVIP), or acellular extracts of MSCs and/or MSC lysates obtained from human MSCs, fibroblast-1ike cells, and non-human animal MSCs including, but not limited to MSCs from horses, cows, pigs, sheep, non-human primates, dogs, cats, rabbits, rats, and mice, in embodiments, the MSCs may be derived from the patient to which the composition will be applied (autologous) or derived from another individual (allogeneic).
- the MSCs may be culture expanded to collect the conditioned media or to increase the quantity of cells for the lysate or used freshly prior to incorporation into the composition of the present disclosure.
- the MSC secretome compositions may comprise about 0,00001 to about 20 wt.%, such as from about 0.01 to abou t 10 wt%, of a mesenchymal stem cell (MSC) extract, MSC exosome, or MSC growth factor preparation.
- the MSC preparation may comprise either MSC conditioned media or MSC lysate from cell culture expanded MSCs.
- the composition may further comprise from about 0.01 to about 10 wt.% of a cell-free medium conditioned by growth of MSCs or MSC lineage ceils, wherein the cells are cultured under normal hyperoxic culturing conditions or under artificial wound healing conditions.
- the MSCs used to produce the disclosed MSC additives can be selectively stimulated to produce MSC growth factors, secretomes, cytokines, chemokin es, mesenchymal stem cell proteins, peptides, glycosaminoglycans, extracellular matrix (ECM), proteoglycans, secretomes, and exosomes.
- the growth factors and exosomes may be derived from any cell in the human body, such as from ectodermal cells, endodermal cells, or mesodermal cells.
- MSC growth factors include but are not limited io prostaglandin E2 (PGE2), transforming growth factor 131 (TGF-(31), hepatocyte growth factor (HGF), stromal cell derived factor- 1 (SDF-1 ), nitric oxide, indoleamine 2,3- dioxygenasc, interleukin-4 (IL-4), IL-6, interleukin- 10 (IL-10), IL-1 receptor antagonist and soluble TNF-a receptor, insulin-1ike growth factors, fibroblast growth factors (FGF) 1-23 (especially, FGF1 and FGF2), bone morphogenetic proteins (BMPs) 1 -15, epidermal growth factor (EGF), transforming growth factor-a (TGF-a) macrophage-stimulating protein (MSP), platelet derived growth factor (PLGF), vascular endothelial growth factor (VEGF), macrophage colony stimulating factor (M-CSF), insulin, granulocyte colony stimulating factor (G-CSF), granulo prostaglan
- C lulturing the MSCs may occur under wound healing and- or hypoxic conditions.
- Hypoxic conditions may comprise about 1% to about 5% oxygen, reduced or no serum, reduced glucose, or these elements in various combinations.
- the combined reduced nutrient and metabolite environment may trigger the cultured cells to produce wound healing and anti-inflammatory ECM proteins and growth factors to direct tissue healing.
- Direct tissue healing likely is in the form of .new ECM proteins, such as collagen and glycosaminoglycans (GAGs), as well as growth factors and cytokines.
- the MSC preparation (such as, for example, a MSC sec-retome composition) comprises MSC growth factors, MSC exosomes, and/or cellular extracts of MSCs or MSC lysates obtained from MSCs cultured under standard hyperoxic- culturing conditions (for example, 21% oxygen) or MSCs cultured under artificial wound healing conditions (such as, for example, 0.1% to about 5% oxygen).
- artificial wound healing conditions simulate growth conditions in real wounds where there is a reduction in nutrient supply and reduction of waste removal that is usually caused by a disruption in local blood circulation. This creates a harsh environment for cells until new blood vessels are created and blood circulation is restored.
- artificial wound healing conditions used to culture MSCs may include one or more of the following growth conditions reduction in glucose availability, reduction in oxygen tension, reduction in pH, and increased temperature.
- the glucose availability can be reduced relative to normal control (e.g,, 4.5 g/L).
- Modified culture media to reduce glucose, but not damage the cells can be between 0 and 50% reduction in glucose, more preferably between about 5% and 40% reduction in glucose.
- MSC artificial wound healing culture conditions can comprise glucose reduction of about 5% to about 15%, from about 10% to about 20%, from about 15% to about 25%, from about 20% to about 30%, or from about 25% to about 35%.
- glucose is present at a concentration of about 0.
- glucose is present at a concentration of less than or no more than 0.1, 0.2, 0.3, 0.4, 0.5, 0.6, 0.7, 0.8, 0.9, 1 .0, 1.1 , 1.2, 1 .3, 1 .4, 1 .5, 1 .6, 1 .7, 1 .8, 1 .9, 2.0, 2.
- oxygen tension can be reduced to oxygen levels to hypoxic conditions.
- Normal atmospheric oxygen is approximately 21% and any reduction is considered hypoxic.
- MSCs can be cultured at between 0.0% and 20.9% oxygen, from about ().! %> io about 0.5% oxygen, from about 0.1 % to about 2.0%, from about 0.1% to about 5.0% oxygen, from about 0.5% to 5.0%, from about 1 .O’* ⁇ •> to about 1 ()",> oxygen, about 5.0% to about 10.0% oxygen; and from about 10.0% to about 15.0%.
- the hypoxic oxygen conditions may be an aspect of artificial wound healing conditions.
- Oxygen tension may be between about 0.5% and 20.5% oxygen when culturing MSCs to produce a therapeutic see.ret.ome composition comprising extracellular vesicles and/or MSC-secreted growth factors, such as, for example, 0.1, 0,2, 0.3, 0.4, 0.5, 0.6, 0.7, 0.8, 0.9, 1 , 1.1, 1.2, 1.3,
- the pH can also be reduced during MSC culturing.
- the pH can be from about 6.0 to about 7.4, for example, from 6.0 to about 6.4, from about 6.2 io about 6.4, from about 6.2 io about 6.6, from about 6.4 to about 6.6, from about 6.4 to about 6,8, or from about 6.6 to about 7.0, such as 6.0, 6. 1, 6.2, 6.3, 6.4, 6.5, 6.6, 6.7, 6.8, 6.9, 7.0, 7.1 , 7.2, 7.3 or 7.4.
- the temperature of the culture environment may be raised relative to physiologic homeostasis temperature (e.g., 37°C).
- the culture conditions for the MSCs can comprise from about 35 ,5 C to about 39°C, from about 35°C to about 36°C, from about 36°C to about 37°C, from about 37°C to about 38 °C, from about 38°C to about 39°C, from about 39 C C to about 40°C.
- the temperature of the culture can be 35.0, 35.1, 35.2,
- the culture media is serum free.
- the scram free culture media comprises platelet lysate.
- the platelet lysate is human platelet lysate ( HPL).
- the serum free culture media comprises at least 1%, 2%, 3%, 4%, 5%, 6%, 7%, 8%, 9%, 10%, 11 %, 12%, 13%, 14%, 15%, 16%, 17%, 18%, 19% , or 20% of HPL by volume, or a range between any two of these values. 1.n some embodiments, the culture media comprises from 8% to 12%, 5% to 15%, or 9% to 1 1 % of HPL by volume.
- the MSC secreiome compositions can further comprise a protective coating (such as, for example, a cryoprotectant oligosaccharide and a protein solution) to reduce degradation of the growth factors.
- a protective coating such as, for example, a cryoprotectant oligosaccharide and a protein solution
- the protective coating can be engineered as a polymer.
- Polymer refers to a relatively high molecular weight organic compound, natural or synthetic, whose structure can be represented by a repeated small unit, the monomer. Non-1imiting examples of polymers include polyethylene, rubber, cellulose.
- Synthetic polymers are typically formed by addition or condensation polymerization of monomers.
- copolymer refers to a polymer formed from two or more different repeating units (monomer residues).
- a copolymer can be an alternating copolymer, a random copolymer, a block copolymer, or a graft copolymer. It is also contemplated that, in certain aspects, various block segments of a block copolymer can themselves comprise copolymers.
- polymer encompasses all forms of polymers including, but not limited to, natural polymers, synthetic polymers, homopolymers, heteropolymcrs or copolymers, addition polymers, etc.
- the gel matrix can comprise copolymers, block copolymers, diblock copolymers, and/or triblock copolymers.
- the protective coating can comprise a biocompatible polymer.
- biocompatible polymer can be crosslinked. Such polymers can also serve to slowly release the adipose browning agent and/or fat modulating agent into tissue.
- biocompatible polymers include, but are not limited to polysaccharides; hydrophilic polypeptides; poly(amino acids) such as poly-L- glutamic acid (PGS), gamma -poly glutamic acid, poly-L-aspartic acid, poly-L- serine, or poly-L- lysine; poly alkylene glycols and polyalkylene oxides such as polyethylene glycol (PEG), polypropylene glycol (PPG), and po1y(ethy1ene oxide) (PEO); poly( oxy ethylated polyol); poly (olefinic alcohol); polyvinylpyrrolidone); poly(hydroxyalkylmethacrylainide); polyfhydroxy alkyl methacrylate); polyf saccharides); poly(bydroxy acids); polyfvinyl alcohol), polyhydroxyacids such as poly(laciic acid), poly (gly colic acid), and poly (lactic acid-co-gly colic acids); poly
- Biocompatible polymers can also include polyamides, polycarbonates, polyalkylenes, polyalkylene glycols, polyalkylene oxides, polyalkylene tereptbalates, polyvinyl alcohols (PVA), methacrylate PVA(m-PVA), polyvinyl ethers, polyvinyl esters, polyvinyl halides, polyvinylpyrrolidone, polyglycolides, polysiloxanes, polyurethanes and copolymers thereof, alkyl cellulose, hydroxyalkyl celluloses, cellulose ethers, cellulose esters, nitro celluloses, polymers of acrylic and methacrylic esters, methyl cellulose, ethyl cellulose, hydroxypropyl cellulose, hydroxy-propyl methyl cellulose, hydroxy butyl methyl cellulose, cellulose acetate, cellulose propionate, cellulose acetate butyrate, cellulose acetate phthalate, carboxylethyl cellulose, cellulose tri
- biodegradable polymers include polyesters, poly(ortho esters), polyethylene amines), poly (caprolactones), p oly (hydroxyb uty rat e s) , poly (hydroxy v alerates), poly anhy dri des, p oly ( aery lie acids) , polyglycolides, poly (urethanes), polycarbonates, polyphosphate esters, polyphospli azenes, derivatives thereof, linear and branched copolymers and block copolymers thereof, and blends thereof,
- the protective coating comprises carbohydrate construction of monosaccharides as well as carbohydrate polymers such as disaccharides or polysaccharides including but not limited to non-reducing poly or disaccharides as well as any combination thereof.
- carbohydrates that can be used in the protective coaling comprise Glucose, Aldoses (D-Allose, D-Altrose, D-Mannose, etc.), Glucopyranose, Pentahydroxyhexanal, a-D-Glucopyranosyl-D-glucose, a-D-Glucopyranosyl-dihydrate, Polymer of P-D-Glycopyranosyl units, P-D-Fractofuranosyl a-D-glucopyntnoside (anhydrous / dihydrate), f3-DGalactopyranosyl-D -glucose, a-D-Glucopyranosyl-a-D-glucopyrauoside (an
- the protective coating contains biocompatible and/or biodegradable polyesters or polyanhydrides such as poly(lactic acid), poly(glycolic acid), and poly (lactic-co -glycolic acid).
- the particles can contain one more of the following polyesters: homopolymers including glycolic acid units, referred to herein as "PGA", and lactic acid units, such as poly-L-1actic acid, poly-D-1actic acid, poly-D,L-1actie acid, poly-L-1actide, poly- D-lactide, and poly-D,L-1actide5 collectively referred to herein as "Pl.
- caprolactone units such as poly(e-caprolactone), collectively referred to herein as "PCL”; and copolymers including lactic acid and glycolic acid units, such as various forms of polyflactic acid-co- gly colic acid) and poly(lactide-co-glycolide) characterized by the ratio of lactic ac.id:glycolic acid, collectively referred to herein as "PLGA”; and polyacry tales, and derivatives thereof.
- PCL poly(e-caprolactone)
- copolymers including lactic acid and glycolic acid units such as various forms of polyflactic acid-co- gly colic acid) and poly(lactide-co-glycolide) characterized by the ratio of lactic ac.id:glycolic acid, collectively referred to herein as "PLGA”; and polyacry tales, and derivatives thereof.
- Exemplary' polymers also include copolymers of polyethylene glycol (PEG) and the aforementioned polyesters, such as various forms of PLGA-PEG or PEA-PEG copolymers, collectively referred io herein as "PEGylated polymers".
- PEG polyethylene glycol
- the PEG region can be covalently associated with polymer to yield "PEGylated polymers" by a cleavable linker.
- the polymer comprises at least 60, 65, 70, 75, 80, 85, 89, 90, 91, 92, 93, 94, 95, 96, 97, 98, or 99 percent acetal pendant groups.
- the triblock copolymers disclosed herein comprise a core polymer such as, example, polyethylene glycol (PEG), polyvinyl acetate, polyvinyl alcohol, polyvinyl pyrrolidone (PVP), polyethyleneoxide (PEO), polyfvinyl pyrrolidone-co-vinyl acetate), polymeihacryiates, polyoxyethylene alkyl ethers, polyoxyethylene castor oils, polycaprolactam, polylactic acid, polygly colic acid, poly (lactic-glycolic) acid, poly(lactic co- gly colic) acid (PLGA), cellulose derivatives, such as hydroxymethylcellulose, hydroxypropylcellulose and the like.
- a core polymer such as, example, polyethylene glycol (PEG), polyvinyl acetate, polyvinyl alcohol, polyvinyl pyrrolidone (PVP), polyethyleneoxide (PEO), polyfvinyl pyrrolidone-co-vinyl acetate
- diblock copolymers that can be used in the protective coatings disclosed herein comprise a polymer such as, example, polyethylene glycol (PEG), poly vinyl acetate, poly vinyl alcohol (PVA), polyvinyl pyrrolidone (PVP), polyethyleneoxide (PEO), poly( vinyl pyrrolidone-co-vinyl acetate), poly methacrylates, polyoxyethylene alkyl ethers, polyoxyethylene castor oils, polycaprolactam, polylaciic acid, polyglycolic acid, poly(lactic-glycolic) acid, poly(lactic co-glycolic) acid (PLGA).
- PEG polyethylene glycol
- PVA poly vinyl alcohol
- PVP polyvinyl pyrrolidone
- PEO polyethyleneoxide
- methacrylates polyoxyethylene alkyl ethers
- polyoxyethylene castor oils polycaprolactam
- polylaciic acid polyglycoli
- the protective coaling contains (i.e., the encapsulated, the encapsulated compositions can further comprise lecithin or hydrolyzed lecithin as a carrier or as encapsulation material.
- lecithin and/or hydrolyzed lecithin coatings include coatings comprising phosphatidyl choline, phosphatidyl inositol, phosphatidyl ethanolamine, phosphatidylserine, and phosphatidic acid.
- Sources of the lecithin can be plant or animal sources.
- any of the polymers, monosaccharides, disaccharides, or polysaccharides used to form the protective coating formed by placing the MSC additive in an encapsulating solution can be at an appropriate concentration for form the protective coating.
- polymers, monosaccharides, disaccharides, or polysaccharides can be at any concentration between 0.01 mM and 10.0 M concentration, for example, from about 0,01 M to about 0, 1 M, from about 0,1 mM to about 1 .0 M, from about 1 .0 M to about 10.0 M.
- the MSC secreiome compositions may comprise any known ingredients typically found pharmaceutical fields such as agents for combating free radicals; bactericides; sequestering agents; preservatives; basifying or acidifying agents; fragrances; surfactants; .fillers; natural products or extracts of natural product, such as aloe or green tea extract; vitamins; or coloring materials.
- Other ingredients that may be combined with the powder may include an antioxidant, which can be selected from a variety of antioxidants.
- Suitable antioxidants include vitamins, such as Vitamin C (L -Ascorbate, Ascorbate-2 Phosphate magnesium salt, Ascorbyl Palmitate, Tetrahexyl decyl Ascorbate), Vitamin E (Tocotrienol), Vitamin A (retinol, retinal, retinoic acid, provitamin A carotenoids, such as beta-carotene), N-acetyl glucosamine, or other deri vatives of glucosamine.
- Vitamin C L -Ascorbate, Ascorbate-2 Phosphate magnesium salt, Ascorbyl Palmitate, Tetrahexyl decyl Ascorbate
- Vitamin E Tocotrienol
- Vitamin A retinol, retinal, retinoic acid, provitamin A carotenoids, such as beta-carotene
- N-acetyl glucosamine or other deri vatives of glucosamine.
- ingredients may include at least one essential fatty acid, such as S2-3, S2-6, and S2-9 polyunsaturated fatty acids, such as linoleic acid (LA), gamma-1inoleic acid (GLA), alpha-1inoleic acid (ALA), dihomo-y-1inolenic acid (DGLA), arachidonic acid (ARA), and others.
- the fatty acids may be derived from various sources including evening primrose oil, black currant oil, borage oil, or GLA modified safflower seeds.
- Other ingredients may include a platelet rich fibrin matrix, at least one ingredient to support ECM production and production of hyaluronic acid, such as N-acetyl glucosamine or other derivatives of glucosamine, ultra- low molecular weight (ULMW) hyaluronic acid, chondroitin sulfate, or keratin sulfate.
- hyaluronic acid such as N-acetyl glucosamine or other derivatives of glucosamine, ultra- low molecular weight (ULMW) hyaluronic acid, chondroitin sulfate, or keratin sulfate.
- Producing the MSC secretome compositions can comprise culturing MSCs collected from a donor to create a cultured media under culturing conditions including, in some embodiments, reduced oxygen and nutrition; stimulating the cultured cells to selectively secrete desired anti-inflammatory proteins, peptides, glycosaminoglycans, proteoglycans exosomes, and secretomes by adjusting the cell growth conditions; collecting, combining the conglomerate mixture with an encapsulation solution, and freezing the conglomerate mixture, wherein the conglomerate mixture comprises exosomes, peptides, proteins, cytokines, growth factors, extracellular matrix (ECM), proteoglycans, glycosaminoglycans; and chemokines selected from the group consisting of human MSCs, animal MSCs, multipotential stromal cells, fibroblasts, and fibroblast cells; combining the conglomerate mixture with an encapsulation solution, such as oligosaccharides, like a
- the MSCs may be lysed to collect all of the M SCs from the culture process, creating an extracted lysate; concentrating the extracted lysate and combining the extracted lysate with an encapsulation solution, such as oligosaccharides like a trehalose solution or protein solution and freezing the mixture; and lyophilizing or freeze-drying the frozen mixture, creating a dry powder.
- the powder contains a highly concentrated collection of analgesic MSC secretomes and exosomes and extracellular matrix components that are specific to anti-in (lamination.
- the method may also include filter-sterilizing, concentrating, freezing, or freeze drying the MSC conditioned culture medium. Additionally, the MSC culture medium may be combined with a cryoprotectant prior to freezing.
- T here are various methods for lysing the MSCs. Lysing may be achieved by the addition of a hypotonic solution or repeated freeze-thaw processes to disrupt the cell membranes. Moreover, the cells may be lysed while attached to the culture surface or in suspension. The cells may also be enzymatically released and/or lysed by mechanical homogenization.
- Stimulating the MSC to selectively secrete the desired anti-inflammatory proteins, peptides, glycosaminoglycans, proteoglycans, exosomes and secretomes may be achieved by adjusting the cell growth conditions, such as cell confluency, culture media supplements, nutritional supplements, oxygen levels, length of culture in those conditions, cell passage number or combinations of those, and the like.
- the therapeutic compositions disclosed herein are used in methods of treating amyotrophic lateral sclerosis (ALS) in a subject. Any of the therapeutic compositions described herein may be used in such a method.
- ALS is a complex, progressive, and fatal neurodegenerative disorder that affects motor neurons; motor neurons die (atrophy) over time, leading to muscle weakness, a loss of muscle mass, and an inability to control movement, and resulting in death from respiratory failure.
- the subject has spinal onset type ALS. In some embodiments, the subject has bulbar onset type ALS. The subject may have advanced ALS. The subject may present with limb-related symptoms. The subject may present with dysphagia or speech difficulties.
- treatment with, the therapeutic compositions disclosed herein delays the p rogre ssi on o f A L S .
- genetic mutations may be associated with ALS.
- mutations in superoxide dismutase type 1 have been found in ALS patients.
- hexanucleotide repeat expansions of six nucleotides, GGGGCC (G4C2), in the C9ORF72 gene are considered to be associated with familial ALS.
- subjects with ALS may carry a mutation in SOD1 gene or C9ORF72 gene, that can lead to amino acid variations in SOD1 protein or C9ORF72 protein, respectively.
- the subject with ALS may carry one or more amino acid variations in SOD 1 protein.
- the subject with ALS may carry' one or more amino acid variations comprising K3E, A4V, W32*, G38R, G41S, G72S, N86S, D90A, G93A, S105L, D109Y, Cl 11 Y, Il 12M, L 126*. N139D, L144S, or a combination thereof, wherein * denotes truncation due to introduction of a premature stop codon in SOD1 protein.
- the subject with ALS may carry G4C2 expansion in C9ORF72 gene, which leads io production of a longer form of C9ORF72 protein with dipeptide repeats.
- Nonlimiting dipeptide repeats can include poly-GA, poly-GP poly-GR, poly-PA, and poly-PR.
- the mutation in SOD1 gene or C9ORF72 gene may be located in a noncoding region intron).
- ALS Functional Rating Scale-Revised a questionnaire-based scale can be used to measure and track changes in an ALS patient’s physical function over time.
- ALSFRS-R measures 12 aspects of physical function comprising speech, salivation, swallowing, handwriting, cutting food, climbing stairs, turning in bed, walking, dressing and hygiene, dyspnea (difficulty breathing), orthopnea (shortness of breath while lying down), and breathing insufficiency.
- Each function is scored from 4 (normal) to 0 (no ability), with a maximum total score of 48 and a minimum total score of 0.
- a patient with higher ALSFRS-R scores across the 12 aspects is considered to have more physical function.
- /XLSFRS-R can be administered by a healthcare provider.
- a subject may have an increase of ALSFRS-R scores after treatment with the therapeutic composition described herein.
- a subject may have an increase of at least about 0.1 point per month in ALSFRS-R scores after treatment with the therapeutic composition described herein compared to ALSFRS-R scores measured prior to treatment.
- a subject may have an increase of at least about 0.1 , 0.2, 0.3, 0.4, 0.5, 0.6, 0.7, 0.8, 0.9, 1 .0, 1.1, 1 .2, 1 .3, 1 .4, 1.5, 1.6, 1.7, 1.8, 1.9, or 2.0 points per month in ALSFRS-R scores after administration of the therapeutic composition described herein compared to ALSFRS-R scores measured prior to administration.
- a subject may have an increase of at least about 0. 1 , 0.2, 0.3, 0.4, 0.5, 0.6, 0.7, 0.8, 0.9, i .0, i .
- ALSFRS-R scores alter administration of the therapeutic composition described herein monthly for at. least 1 , 2, 3, 4, 5, 6, 7, 8, 9, 10, 1 1 , 12, or more than 12 months compared to ALSFRS-R scores measured prior to administration.
- a subject may have a decline of ALSFRS-R scores after treatment with the therapeutic composition described herein.
- a subject may have a decline of less than about 3.0 points per month in ALSFRS-R scores after treatment with the therapeutic composition described herein compared to ALSFRS-R scores measured prior to treatment.
- a subject may have a decline of less than about 3.0, 2.9, 2.8, 2.7, 2.6, 2.5, 2.4, 2.3, 2.2, 2.1, 2.0, 1.9, 1.8, 1.7, 1.6, 1.5, 1.4, 1.3, 1.2, 1.1 , 1.0, 0.9, 0.8, 0.7, 0.6, 0.5, 0.4, 0.3, 0.2, or 0.1 points per month in ALSFRS-R scores after administration of the therapeutic composition described herein compared to ALSFRS-R scores measured prior to administration.
- a subject may have a decline of less than about 3.0, 2.9, 2.8, 2.7, 2.6, 2.5, 2.4, 2.3, 2.2, 2.1 , 2.0, 1.9, 1.8, 1.7, 1.6, 1.5, 1.4, 1.3, 1.2, 1.1, 1.0, 0.9, 0.8, 0.7, 0.6, 0.5, 0.4, 0.3, 0.2, or 0.1 points per month in ALSFRS-R scores after administration of the therapeutic composition described herein monthly for at Least 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 1 1 , 12, or more than 12 months compared to /XLSFRS-R scores measured prior to administration.
- a subject may have a history of a decline in ALSFRS-R scores prior to administration of the therapeutic MSC secretome composition.
- a subject may have ahistory of a decline in ALSFRS-R scores of about 0.1, 0.2, 0.3, 0.4, 0.5, 0.6, 0.7, 0.8, 0.9, 1.0, 1.1, 1.2, 1.3, 1.4, 1.5, 1.6, 1.7, 1.8, 1.9, 2.0, 2.1, 2.2, 2.3, 2.4, 2.5, 2.6, 2.7, 2.8, 2.9, or 3.0 points per month prior to administration of the therapeutic MSC secretome composition.
- arc methods of treating ALS in a subject comprising administering to the subject a composition comprising secreted extracellular vesicles that contain a composition that includes any combination of proteins and/or miRNAs, selected from the following: Ferritin, NUP85, LAMP2, GPR115, Serpin Fl, OPN, PALI, DAPP1, Cathepsin B, Semaphorin 6C, PDGF R alpha, Sortilin, Serpin B6, Dkk-3, Thrombomodulin, PF4, MTF, Periostin, Furin, TIMP-1, Decorin, PCK1, CD99, CD63, CD9, CD81, Transferrin, DcR3, Lumican, TIMP-2, SLITRK5, FAP, Artemin, DPPII, cIAP-1, Pentraxin 3, Visfatin, Neprilysin, Albumin, Galectin-1, UNC5H3, IL-20 R beta, SREC-H, JAM-C, TNF
- ESAM IL-1 R4, CXCL14, IL-31 , SIRP alpha, Uromodulin, CTRL, CEACAM-1 , TARC, MIP-3a, SDF-1b, NKp46, MCP-3, IL-32 alpha, TGFb3 FOLR2, CD5S, IL-23, CD36, TNFb, Shh-N, Ficolin-1 , Reg4, ILT2, Mer, TREM-2, Flt-3L, CDS, IL-6, CD229, Insulin, Syntaxin 6, GRO, Bcl-w, LipocaIin-2, PDGF-AA, IL-2 Ra, Angiogenin, LYVE-1, CD4, RAGE, CDNF, Brevican, NAP-2, PUT , EDAR, ADAMTS13, Kymireninase, PTH1R, IFN-garnma R 1 CrkL.
- hsa-1et-7i-5p hsa-miR-100- 5p, hsa-miR-103 a-3p, hsa-miR-106a-5p, hsa-miR- 106b-5p, hsa-mir-lOb, bsa-miR-10b-5p, hsa-mir- 1246, hsa-miR- 1246, hsa-miR- 125a- 5p, hsa-nuR-125b-5p, hsa-miR-13Oa-3p, hsa-mir- 130b, hsa-miR-130b-3p, hsa-miR-132-3p, hsa-miR-136-5p, hsa-miR-138-5p, hsa-miR-139-5p, hsa-mir- 140, hsa-miR- 140
- bsa-m.iR-152-3p hsa-miR-15a- 5p, hsa-miR-15b-5p, hsa-mir- 16-1, hsa-mir-16-2, hsa-miR-16-5p, hsa-miR-1'7-5p, hsa-miR- 181a-5p, hsa-miR-191 -5p, hsa-miR- 193a-5p, hsa-miR-193b-3p, hsa-miR-19’7-3p, hsa-miR- 199a-3p, hsa-miR-199a- 5p, hsa-miR-199b-5p.
- the therapeutic product is cells per ml and is administered at a dose of 10 io 20 ml. In some embodiments, the therapeutic product comprises 6xl() k ’ to 8x10’° extracellular vesicles or cells per ml and is administered at a dose of 10, 1 1 , 12, 13, 14, 15, 16, 17, 18, 19, or 20 ml, or a range between any two of these values.
- a subject that can be treated with the therapeutic product and methods described herein can comprise any subject suffering from or diagnosed with AI..S.
- a subject can comprise a mammal.
- a subject can comprise a human.
- a subject can comprise a non-human mammal.
- Non-1imiting examples of a non-human mammal can include a non-human primate such as chimpanzee, and other apes and monkey species; farm animals such as cattle, horses, sheep, goats, swine; domestic animals such as rabbits, dogs, and cats; laboratory animals including rodents, such as rats, mice and guinea pigs, and the like.
- An MSC secretome therapeutic composition was made by the following method: human bone marrow-derived MSCs were cultured in culture vessels with growth media to expand the MSC population. Growth media was then removed, and the cells were washed with PBS. The MSCs were then cultured in reduced glucose media with a pH below 7.0 under hypoxic conditions. The conditioned media was then collected and subjected to diafiltration followed by filler sterilization. The production process for the therapeutic product was done under current Good Manufacturing Practices and Current Good Tissue Practices.
- nucleic acid content of the therapeutic product was determined, and the following nucleic acids were found to be present: hsa-let-7a-5p, hsa-let-7b-5p, hsa-let-7c-5p, hsa-let-7d- 3p, hsa-let-7e-5p, hsa-let-7g-5p, hsa-let-7i, hsa-let-7i-5p, hsa-miR-100-5p, hsa-miR-103a-3p, hsa-miR-106a-5p, hsa-miR-106b-5p, hsa-mir-lOb, hsa-miR- 10b-5p, hsa-mir-1246, hsa-miR- 1246, hsa-miR-125a-5p, hsa-miR-125b -5p, hsa-miR
- hBM-MSC EV human bone matrow stem cell derived extracellular vesicle
- IP amyotrophic lateral sclerosis
- IV intravenous
- HBM-MSC EVs appeared safe in ALS patients. This early investigation suggests a controlled study of EVs for the treatment of ALS is warranted.
- ALS Amyotrophic lateral sclerosis
- MSC EV mesenchymal stem cell extracellular vesicles
- hBM- MSC EV may have potential for delaying the progression of ALS. This indicates that more, larger studies need to be carried out to find out treatment specifics, such as dose (how much of the treatment to give) and frequency (how often to give the treatment), and how they could be related to patient outcomes.
- ALS is the third most common neurodegcnerative disease and, behind only Alzheimer’s disease and Parkinson’s disease, it. is the most common type of motor neuron disease. There has been little advancement in the understanding and treatment of ALS despite an incidence rale of approximately 0.005%, The disease presents with both spinal and bulbar forms. Genetically linked causes have been defined but these events account for a minority of cases. Less well defined environmental factors are also therefore assumed to underlie 90%- 95% of ALS cases, and until further research clarifies additional causes, the latter eases are deemed sporadic in origin. The remaining 5- 10% of cases with a clear genetic link to family history are classified as familial, yet effective therapies directed at such defined targets have not been developed.
- Riluzole which decreases glutamate levels within neurons, is defined as an oral glutamatergie neurotransmission inhibitor. It demonstrates limited results in the clinic, and patients only survive an additional five to six months. As a potent antioxidant that is administered intravenously (IV), Edaravone slows the progression of early stage ALS, but recent publications are questioning the findings of earlier clinical trials. Currently, these are the only two FDA approved drugs for the treatment of ALS and access to these treatments can be restricted and limited. Given these limitations, it is urgent that additional medical treatments be discovered and developed to stop or reverse the progress of both sporadic and familial ALS.
- EXOF1O IM the investigational product (IP) used herein, is a human BM-MSC derived EV (hBM-MSC EV) preparation.
- the IP is a consistent EV product with extensive characterization including advanced particle analysis, proteomic evaluation and USP ⁇ 71> sterility assurance. Additionally, through manufacturing in a cGMP environment, the IP is a quality bio-pharmacological product that is consistent regarding dose and biological activity.
- Two peer reviewed studies have demonstrated the safety of administering IV Infusions of the IP to severe CO VID- 19 patients. The IP has also demonstrated efficacy in a subset of these severely ill patients. We hypothesized that the IP would be safe to administer intravenously (IV) to ALS patients and could potentially demonstrate efficacy in the patient population.
- IV intravenously
- Subjects meeting the following original inclusion criteria were included: 30-65 years old, a diagnosis of ALS, and written informed consent by the subject or legal representative was obtained. An IRB-approved protocol deviation was written to extend the age limit io 72 io improve recruitment io 10 subjects. Subjects were categorized as spinal onset type (initially presenting with limb involvement) or bulbar onset type (initially presenting with dysphagia or speech difficulties) based on initial symptoms. Informed consent was obtained on all subjects. ALS type was based on primary symptom presentation of their disease, and the rate of decline determined as rapid or normal. Spinal onset ALS declines at one point per month on average whereas bulbar onset ALS can decline much faster. Time with symptoms was defined as the time lapsed from initial reporting of ALS symptoms, typically much later than onset of any related symptoms prior to a diagnosis.
- Safety was defined as the lack of adverse or serious adverse events related to the investigational product (TP).
- TP investigational product
- Subject #5 who began the study with an amyotrophic lateral sclerosis functional rating scale-revised (ALSFRS-R) score of 1 , subjective assessment by the patient’s caregiver was provided. This included eye contact, focus, and blinking communication as the patient could not move or speak.
- ALSFRS-R amyotrophic lateral sclerosis functional rating scale-revised
- the IP dosing was calculated based on (1 ) the phase I START trial using IV administration of BM-MSC for Acute Respiratory Distress Syndrome, which demonstrated safety at up to 5 million cell /Kg and a ceil ing dose of 10 mi llion cell/Kg; (2) observation of approximately 2.000 EVs secreted per cell: (3) lab analysis indicating 60-80 billion EV/mL; and (4) safe administration of 10 mL doses io a single ALS patient previously. This indicated an IV IP ceiling dose of 17.5 mL/70 Kg adult, and 10 ml. of IV IP was determined as a reasonable dose providing 0.6 ••• 0.8 trillion EV particles per dose.
- ALSFRS-R. Score amyotrophic lateral sclerosis functional rating scale-revised
- Time 0 baseline ALSFRS-R scores ranged from 1 -41 with a mean of 26.7 across all
- Time 0 and Time 1 scores were those obtained just prior io the initiation of the first and second treatments, respectively. Each progressi ve score was recorded at one-month intervals for a total of three consecutive months of scoring (Time 0 through Time 3). One subject was an outlier with a prior ALSFRS-R score of 1 and was included in the study to contribute to the safety evaluation in all stages of the disease’s progression.
- FIG. 1A illustrates the differences in raw ALSFRS-R scores amongst all subjects at each measurement time period.
- FIG. IB shows the fitted linear regression analysis for each subject over time to illustrate disease progression during the study.
- One subject (S3) demonstrated steady improvement in ALSFRS-R score and two others (S4 and S10) showed no apparent decrease in ALSFRS-R score. The remainder of the subjects exhibited score declines.
- the mean (+/- SD) dehaFRS calculated from Table 1 across all 10 subjects from baseline (Time 0) to study end (36 months. Time 3) was -1.03 A- 1 .44.
- the mean slope derived from the linear regression analysis was -0.98 A- 1.67.
- EVs derived from MSCs may offer a novel therapeutic due to the preclinical and clinical evidence of safety and efficacy.
- Safety is a critical aspect of all early phase studies of a new investigational product in a new disease indication. In this study, ten ALS subjects were treated with the hBM-MSC EV IP to evaluate safety risk and potential for efficacy. There were no adverse or serious adverse events related to the investigational product. This safety profile was consistent with the excellent safety profile observed upon IV administration of up to two doses (15 ml. each) of the IP in patients with severe COVID- 19. This small open-1abel pilot safety study suggests that the intravenous delivery of bone marrow derived MSC EVs is safe in ALS patients.
- BM-MSC EVs can modulate the inflammatory, anti-inflammatory and neurotoxic activities of astrocytes and/or microglia isolated from SOD1G93A mice (a mouse model of ALS) and of motor neurons differentiated from inducible neural progenitor cells of ALS patients carrying SOD1G93A or C9orf72 mutations.
- SOD1G93A mice a mouse model of ALS
- motor neurons differentiated from inducible neural progenitor cells of ALS patients carrying SOD1G93A or C9orf72 mutations The net effect of BM-MSC EVs in the in vitro and in vivo models is a reduction of both neurotoxicity and neuroinllammation.
- BM-MSC EVs to pass through the blood brain barrier provides the opportunity to treat this terminal disease using a safer IV administration approach. It also would allow for more convenient and frequent dosing to help continue and boost any possible gains from this potential treatment option.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Immunology (AREA)
- Cell Biology (AREA)
- Developmental Biology & Embryology (AREA)
- Zoology (AREA)
- Medicinal Chemistry (AREA)
- Biomedical Technology (AREA)
- Biotechnology (AREA)
- Virology (AREA)
- Hematology (AREA)
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Pharmacology & Pharmacy (AREA)
- Epidemiology (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Medicines Containing Material From Animals Or Micro-Organisms (AREA)
Abstract
Description
Claims
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202480051907.2A CN121729236A (en) | 2023-06-09 | 2024-06-07 | Amyotrophic Lateral Sclerosis Treatment Using Extracellular Vesicle Composition |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202363507204P | 2023-06-09 | 2023-06-09 | |
| US63/507,204 | 2023-06-09 | ||
| US202463570632P | 2024-03-27 | 2024-03-27 | |
| US63/570,632 | 2024-03-27 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2024254540A2 true WO2024254540A2 (en) | 2024-12-12 |
| WO2024254540A3 WO2024254540A3 (en) | 2025-02-20 |
Family
ID=93796466
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2024/033123 Pending WO2024254540A2 (en) | 2023-06-09 | 2024-06-07 | Treatment of amyotrophic lateral sclerosis with extracellular vesicle composition |
Country Status (2)
| Country | Link |
|---|---|
| CN (1) | CN121729236A (en) |
| WO (1) | WO2024254540A2 (en) |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US12453743B2 (en) | 2018-05-30 | 2025-10-28 | Direct Biologics, Llc | Mesenchymal stem cell (MSC) growth factor and extracellular vesicle preparation in frozen or powdered form and methods of use |
| US12502407B2 (en) | 2024-04-25 | 2025-12-23 | Direct Biologics, Llc | Treatment of fistula with bone marrow mesenchymal stem cell derived extracellular vesicles |
| US12569517B2 (en) | 2019-02-07 | 2026-03-10 | Direct Biologics, Llc | Method for treating osteoarthritis with mesenchymal stem cell exosomes |
| US12570980B2 (en) | 2020-04-22 | 2026-03-10 | Direct Biologics, Llc | Methods and compositions for treating inflammatory conditions associated with infectious disease |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2019527218A (en) * | 2016-07-18 | 2019-09-26 | ブレインストーム セル セラペウティクス リミテッド | Method for treating amyotrophic lateral sclerosis (ALS) |
| US12053493B2 (en) * | 2016-11-03 | 2024-08-06 | Exostem Biotec Ltd. | Mesenchymal stem cells populations, their products, and use thereof |
| WO2019126068A1 (en) * | 2017-12-20 | 2019-06-27 | Cedars-Sinai Medical Center | Engineered extracellular vesicles for enhanced tissue delivery |
-
2024
- 2024-06-07 WO PCT/US2024/033123 patent/WO2024254540A2/en active Pending
- 2024-06-07 CN CN202480051907.2A patent/CN121729236A/en active Pending
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US12453743B2 (en) | 2018-05-30 | 2025-10-28 | Direct Biologics, Llc | Mesenchymal stem cell (MSC) growth factor and extracellular vesicle preparation in frozen or powdered form and methods of use |
| US12569517B2 (en) | 2019-02-07 | 2026-03-10 | Direct Biologics, Llc | Method for treating osteoarthritis with mesenchymal stem cell exosomes |
| US12570980B2 (en) | 2020-04-22 | 2026-03-10 | Direct Biologics, Llc | Methods and compositions for treating inflammatory conditions associated with infectious disease |
| US12502407B2 (en) | 2024-04-25 | 2025-12-23 | Direct Biologics, Llc | Treatment of fistula with bone marrow mesenchymal stem cell derived extracellular vesicles |
Also Published As
| Publication number | Publication date |
|---|---|
| CN121729236A (en) | 2026-03-24 |
| WO2024254540A3 (en) | 2025-02-20 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US12570980B2 (en) | Methods and compositions for treating inflammatory conditions associated with infectious disease | |
| WO2024254540A2 (en) | Treatment of amyotrophic lateral sclerosis with extracellular vesicle composition | |
| Bazzoni et al. | Extracellular vesicle-dependent communication between mesenchymal stromal cells and immune effector cells | |
| Liu et al. | Therapeutic prospects of MicroRNAs carried by mesenchymal stem cells-derived extracellular vesicles in autoimmune diseases | |
| WO2024254459A2 (en) | Treatment of complex regional pain syndrome with extracellular vesicle composition | |
| JP2025020190A (en) | Human oral mucosa stem cell secretome | |
| EP3653217A1 (en) | Mesenchymal stem cells for use in improving pulmonary function | |
| WO2025101663A1 (en) | Extracellular vesicle composition for use in the treatment of graft-versus-host disease | |
| WO2025101653A1 (en) | Extracellular vesicle composition for use in the treatment of long covid | |
| WO2025101659A1 (en) | Extracellular vesicle composition for use in the treatment of irradiated skin | |
| WO2024192119A1 (en) | Wound healing compositions and methods thereof | |
| Saad-Naguib et al. | Impaired receptivity of thin endometrium: therapeutic potential of mesenchymal stem cells | |
| WO2025101658A1 (en) | Extracellular vesicle composition for use in the treatment of lumbar facet joint syndrome | |
| EP3813858B1 (en) | Therapeutic lung repair by inhalation of lung spheroid exosomes | |
| EP4114419A1 (en) | Fibroblast and tlr activated fibroblast treatment of viral induced acute respiratory distress syndrome | |
| US20230293598A1 (en) | Prevention of radiation exposure associated pathology by treatment with umbilical cord derived regenerative cells | |
| WO2021232064A2 (en) | Treatment of ovarian failure using regenerative cells | |
| WO2025226276A1 (en) | Treatment of fistula with bone marrow mesenchymal stem cell derived extracellular vesicles | |
| WO2025226866A1 (en) | Bone marrow mesenchymal stem cell derived extracellular vesicles to improve perioral area and face fat graft retention | |
| US12502407B2 (en) | Treatment of fistula with bone marrow mesenchymal stem cell derived extracellular vesicles | |
| Fareez et al. | Molecular and Epigenetic Basis of Extracellular Vesicles Cell Repair Phenotypes in Targeted Organ-specific Regeneration | |
| US12590310B2 (en) | Methods and compositions for treating inflammatory conditions associated with infectious disease | |
| Lin et al. | Mechanisms of stem cells and their secreted exosomes in the treatment of autoimmune diseases | |
| Surya et al. | Stem cells as therapeutics and their implications in cytokine storm in COVID-19 | |
| CA3178941A1 (en) | Compositions and methods for treatment of inflammatory disorders |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24820188 Country of ref document: EP Kind code of ref document: A2 |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24820188 Country of ref document: EP Kind code of ref document: A2 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2024820188 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2024820188 Country of ref document: EP Effective date: 20260109 |
|
| ENP | Entry into the national phase |
Ref document number: 2024820188 Country of ref document: EP Effective date: 20260109 |
|
| ENP | Entry into the national phase |
Ref document number: 2024820188 Country of ref document: EP Effective date: 20260109 |
|
| ENP | Entry into the national phase |
Ref document number: 2024820188 Country of ref document: EP Effective date: 20260109 |
|
| ENP | Entry into the national phase |
Ref document number: 2024820188 Country of ref document: EP Effective date: 20260109 |