WO2024257764A1 - 免疫反応の評価方法 - Google Patents
免疫反応の評価方法 Download PDFInfo
- Publication number
- WO2024257764A1 WO2024257764A1 PCT/JP2024/021195 JP2024021195W WO2024257764A1 WO 2024257764 A1 WO2024257764 A1 WO 2024257764A1 JP 2024021195 W JP2024021195 W JP 2024021195W WO 2024257764 A1 WO2024257764 A1 WO 2024257764A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- sequences
- antigen
- vaccine
- cov
- immune
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/005—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from viruses
- C07K14/08—RNA viruses
- C07K14/11—Orthomyxoviridae, e.g. influenza virus
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/005—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from viruses
- C07K14/08—RNA viruses
- C07K14/165—Coronaviridae, e.g. avian infectious bronchitis virus
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/705—Receptors; Cell surface antigens; Cell surface determinants
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/53—Immunoassay; Biospecific binding assay; Materials therefor
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/53—Immunoassay; Biospecific binding assay; Materials therefor
- G01N33/569—Immunoassay; Biospecific binding assay; Materials therefor for microorganisms, e.g. protozoa, bacteria, viruses
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/68—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving proteins, peptides or amino acids
Definitions
- the present invention relates to a method for evaluating immune responses.
- Non-Patent Documents 1, 2 Since COVID-19, caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), is life-threatening for patients with hematological malignancies, hematologists generally recommend vaccination with mRNA SARS-CoV-2 vaccines (Non-Patent Documents 1, 2). However, it has been reported that some patients with hematological malignancies, such as those who have undergone hematopoietic stem cell transplantation (HSCT) or B cell depletion therapy, may not achieve sufficient humoral responses after vaccination (Non-Patent Documents 3-6).
- HSCT hematopoietic stem cell transplantation
- B cell depletion therapy may not achieve sufficient humoral responses after vaccination.
- Non-Patent Document 7 Even after the emergence of the omicron variant of SARS-CoV-2, which is considered to have low pathogenicity (Non-Patent Document 7), it has been reported that hospitalization and mortality rates in immunocompromised patients remain high, especially in patients who respond poorly to vaccination (Non-Patent Documents 8, 9).
- tixagevimab/silgavimab For such patients, the US Food and Drug Administration (FDA) issued an emergency use authorization for tixagevimab/silgavimab in December 2021.
- the drug contains neutralizing monoclonal antibodies against different epitopes in the receptor-binding domain of the SARS-CoV-2 spike protein and is used as a pre-exposure prophylaxis.
- tixagevimab/silgavimab is considered to be effective in preventing severe disease and shortening hospitalization in immunocompromised patients (Non-Patent Documents 10, 11).
- Non-Patent Documents 12 tixagevimab/silgavimab
- tixagevimab/silgavimab has low activity against certain omicron subspecies such as BQ.1 and XBB (Non-Patent Documents 13, 14).
- Tixagevimab/silgavimab which was developed before the emergence of the Omicron subspecies, has reduced activity against the currently dominant strains in certain countries (Non-Patent Documents 13, 14).
- Non-Patent Document 1 One study reported favorable outcomes of COVID-19 caused by the omicron variant as well as previous strains in a cohort of highly vaccinated hematopoietic stem cell transplant patients (Non-Patent Document 15).
- Non-Patent Document 16 Immunogenicity, especially humoral immunity, after SARS-CoV-2 vaccination is generally evaluated by enzyme-linked immunosorbent assay (ELISA) using anti-SARS-CoV-2 spike IgG antibodies.
- ELISA enzyme-linked immunosorbent assay
- tixagevimab/silgavimab is itself an anti-spike IgG antibody
- the antibody titer by ELISA is maintained at a fairly high level for a long period of time in all patients who received tixagevimab/silgavimab. Therefore, administration of tixagevimab/silgavimab masks the appearance of anti-spike antibodies after vaccination. In other words, there is a problem that the response to vaccination in these patients cannot be evaluated, even though vaccination is recommended.
- This problem of being unable to selectively evaluate only the targeted immune response is not unique to the situation after administration of tixagevimab/silgavimab, but is common to the situation after administration of all antibody drugs against SARS-CoV-2, such as casirivimab/imdevimab and sotrovimab, as well as other antibody drugs that may be clinically approved in the future.
- this problem occurs not only after administration of antibody drugs, but also in situations where antibody titers are maintained at a high level due to previous vaccination or infection, etc.
- this problem is not limited to vaccines against SARS-CoV-2, but is a universal problem that applies to the evaluation of any vaccine and, ultimately, any response to any antigen (e.g., infection, cancer immune response, autoimmune response, etc.).
- any antigen e.g., infection, cancer immune response, autoimmune response, etc.
- none of the currently used techniques can selectively evaluate only the targeted immune response.
- a new analytical method is needed that can evaluate immune responses at the level of mRNA expression, rather than at the level of protein expression, as in the case of conventional antibody titer measurement using ELISA.
- repertoire analysis cannot be simply applied to evaluate the post-vaccination response at the mRNA level is that repertoire analysis is a means essentially for evaluating the degree and changes in immune diversity, and is naturally not envisioned as a means for selectively identifying immune responses to specific antigens at the antibody sequence level.
- the object of the present invention is to provide a method for selectively identifying immune responses to specific antigens at the mRNA level using immune cell receptor repertoire data.
- the present invention was completed through further investigation based on this knowledge.
- Item 1 A step A of preparing repertoire data including a group of sequences of antigen recognition sites of immune cell receptors obtained from a sample of a subject; and B. comparing the repertoire data with a database including a group of sequences of antigen recognition sites of immune cell receptors for specific antigens, and detecting from the repertoire data a sequence that is identical to the sequence included in the database at the amino acid sequence level or that is identical in a portion other than two or less amino acid residues. This makes it possible to identify the sequence that immunologically reacted in the subject's body at the time the sample was collected.
- the method according to Item 1 further comprising a step C of deriving the number and/or frequency in the repertoire data of the sequences detected by the step B.
- Item 3. The method according to Item 2, further comprising a step D of confirming a change in the number and/or frequency over time.
- Item 4. The method according to Item 3, wherein in the step D, it is confirmed whether the time when the increase is observed as the change over time corresponds to 11 to 20 days or 3 to 10 days after stimulation with the specific antigen.
- Item 5. The method according to any one of Items 1 to 4, wherein the subject is a subject that has been exposed to the specific antigen.
- Item 6. The method according to Item 5, wherein the exposure to a specific antigen is vaccination. Clause 7.
- the method of clause 6, wherein the vaccine is a nucleic acid vaccine.
- Clause 8. The method of clause 6, wherein the vaccine is a coronavirus vaccine or an influenza vaccine.
- the method further comprises a step E of evaluating the scientific validity of the nucleic acid vaccine by checking additional information included in the database for the sequence whose number and/or frequency has been confirmed to increase over time, the additional information is selected from the group consisting of: [1] information on the presence or absence of neutralizing activity against an antigen, [2] information on the species or strain of the antigen, and [3] information on an epitope; Item 9.
- the specific antigen is a vaccine, 14.
- Item 15. The method according to any one of Items 5 to 14, wherein the sample is collected at the time of activation of mRNA of an immune cell receptor for the specific antigen.
- Item 16 The method according to Item 15, wherein the activation time is 11 to 20 days or 3 to 10 days after the exposure.
- Item 17 The method according to any one of Items 1 to 3, wherein the subject has undergone cancer immunotherapy.
- the method according to Item 16 wherein the sample is collected at the time of activation of the mRNA of an immune cell receptor for the specific antigen.
- Item 19 The method according to Item 17, wherein the activation period is 11 to 20 days or 3 to 10 days after the cancer immunotherapy.
- Item 20 The method according to any one of Items 1 to 16, wherein the subject is a subject receiving immunosuppressive treatment.
- Clause 21 The method of clause 20, wherein the immunosuppressive treatment is hematopoietic stem cell transplantation or administration of B cell depletion therapy.
- Item 23. The method according to any one of Items 1 to 16, wherein the subject is a patient with an autoimmune disease.
- the repertoire data is obtained from samples of subjects who have been vaccinated against a previously emerged virus strain ST1, the specific antigen is a virus strain ST2, the efficacy of which by the vaccine is unknown; Item 3.
- the method of item 1 or 2 further comprising step H of evaluating the vaccine as effective against the virus strain ST2 if the detected sequence is present.
- Item 25 The method according to any one of items 1 to 24, wherein the database comprises sequences of antigen recognition sites of the immune cell receptors for the specific antigens collected from a population of survey subjects in which an immune response to the specific antigen has occurred, and is obtained by the following steps: A process of acquiring repertoire data including a group of sequences of antigen recognition sites of the immune cell receptor from samples of each of the survey subjects, the repertoire data being in chronological order at the activation time T ex of the mRNA of the immune cell receptor for the specific antigen, T bf before the activation time, and T af after the activation time; and a process of selecting, as information to be collected in the database, sequences of antigen recognition sites of immune cell receptors that are observed to proliferate at the activation time T ex .
- Item 26 The method according to Item 25, wherein the activation time T ex is 11 to 20 days or 3 to 10 days after stimulation with the specific antigen.
- Item 27 The method according to any one of items 1 to 26, wherein the immune cells are T cells or B cells.
- the present invention provides a method for selectively identifying immune responses to specific antigens at the mRNA level using immune cell receptor repertoire data.
- FIG. 1 shows an outline of an example of a method for evaluating an immune response according to the present invention.
- FIG. 1 shows a conceptual diagram of an example of creating a database to be applied to the immune response evaluation method of the present invention.
- FIG. 1 shows a conceptual diagram of an example in which a large-scale data set group is applied as a comprehensive biosensor to the immune response evaluation method of the present invention.
- FIG. 1 shows a conceptual diagram of an example of the scientific validity of a vaccine that can be evaluated by the immune response evaluation method of the present invention (the ability of a healthy subject to produce, through vaccination, antibodies that completely match the antibodies acquired by a patient through infection with an antigen). Anti-spike antibody titers over time.
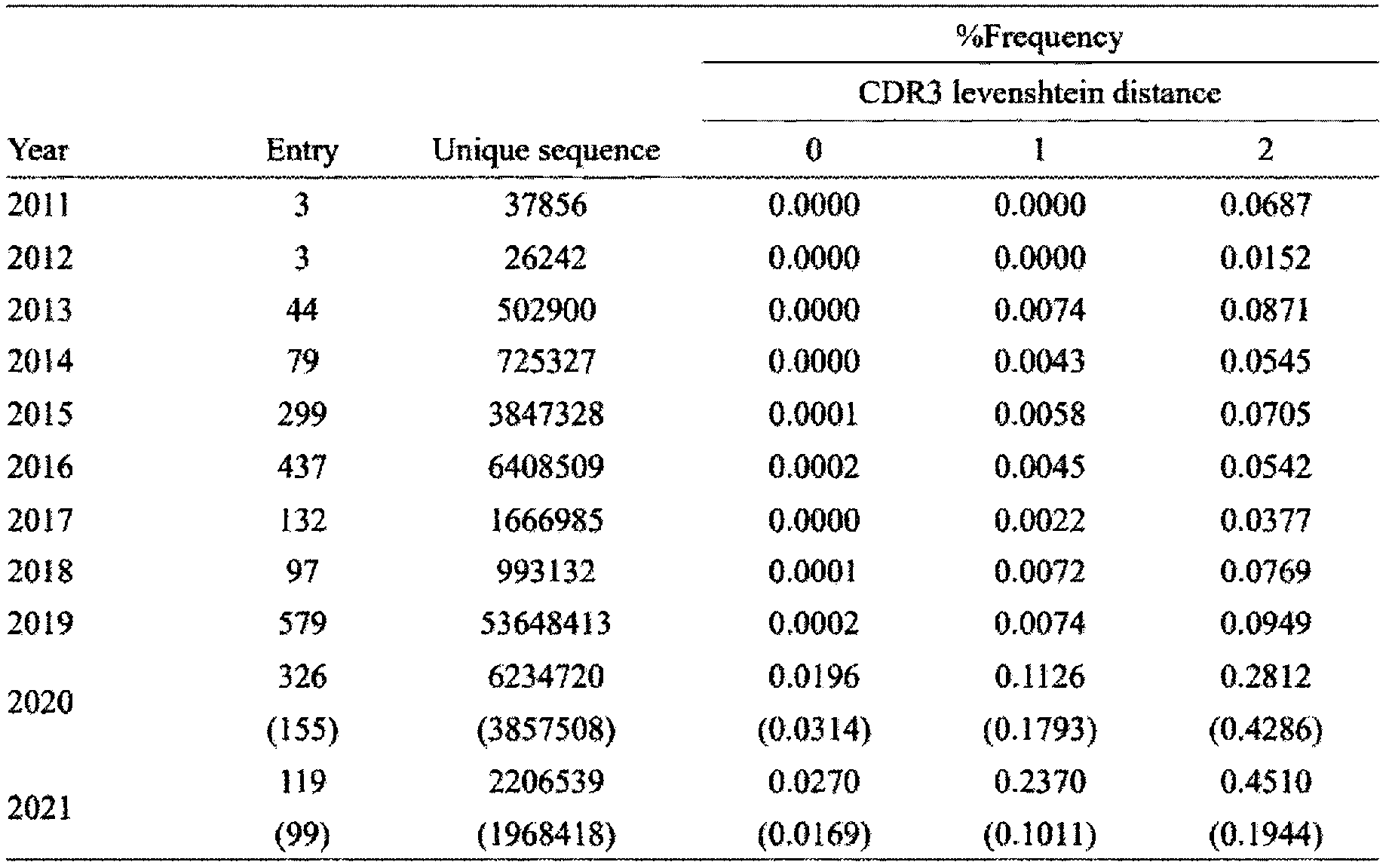
- Numbers indicate number of sequences used. Number and frequency of SARS-CoV-2-specific sequences over time in three patients infected with COVID-19. NGS analysis was performed on PBMCs collected over time after SARS-CoV-2 infection. Clone numbers (top) and frequencies (bottom) of SARS-CoV-2-specific sequences with identical V and J genes to the CoV-AbDab sequence and Levenshtein distances of 0 (left), 1 (middle) or 2 (right) for the CDR3 amino acid sequence are shown. The bottom circle plot shows a cluster plot of SARS-CoV-2-specific sequences. An igraph network of up to 1000 SARS-CoV-2-specific sequences is shown.
- Each node represents a single unique read with identical IGHV, IGHJ and complementarity determining region 3 (CDR3) amino acid sequences. Nodes were connected by edges defined by a Levenshtein distance of ⁇ 1 for the CDR3 amino acid sequence. The size of the node was the percent frequency of each unique read. Changes in number, frequency and clusters of SARS-CoV-2-specific sequences over time following primary and booster vaccination. Healthy volunteer 1 received a primary dose of mRNA SARS-CoV-2 vaccine (monovalent BNT162b2) followed by a second dose 21 days later. After Figure 7A, the fifth dose of the bivalent BNT162b2 vaccine was administered.
- SARS-CoV-2-specific sequences were searched for SARS-CoV-2-specific sequences in the BCR repertoire data. Time course of number, frequency and clustering of SARS-CoV-2-specific sequences after primary and booster vaccination. Healthy volunteer 2 received the fourth dose of monovalent mRNA-1273 and then SARS-CoV-2-specific sequences were measured 2, 6 and 9 days after the booster vaccination. Time course of SARS-CoV-2 specific sequence numbers, frequency and clusters after mRNA vaccination in hematopoietic stem cell transplant recipients receiving tixagevimab/silgavimab.
- the recipient (T/C patient 1) was administered tixagevimab/silgavimab (T/C) 338 days after cord blood transplantation and then the mRNA vaccine (time indicated by the Vaccination arrow).
- Immune cell reconstitution of helper T cells CD3+CD4+
- class-switched B cells CD19+CD27+IgD-
- plasmablasts CD19+CD27+CD38+
- Anti-SARS-CoV-2 antibody titers were measured using the Abbott SARS-CoV-2 IgG II Quant kit (Abbott) and the Roche Elecsys anti-SARS-CoV-2 S kit (Roche).
- T/C tixagevimab/silgavimab. Number, frequency and clusters of SARS-CoV-2-specific sequences over time following mRNA vaccination in hematopoietic stem cell transplant recipients receiving tixagevimab/silgavimab. In T/C patient 2, the recipient received tixagevimab/silgavimab 212 days after unrelated bone marrow transplant and then the mRNA vaccine (time indicated by the Vaccination arrow).
- Distribution of CDR3 amino acid lengths of SARS-CoV-2-specific sequences with different CDR3 amino acid distances Percent frequency of SARS-CoV-2-specific sequences with different CDR3 lengths detected in this study is shown. VJ usage frequency of SARS-CoV-2 specific sequences with different CDR3 amino acid distances.
- the percentage of VJ usage frequency of SARS-CoV-2 specific sequences detected in this study is shown in a bubble chart.
- the X-axis shows IGHV genes
- the Y-axis shows IGHJ genes
- the size of the bubble indicates the frequency of usage.
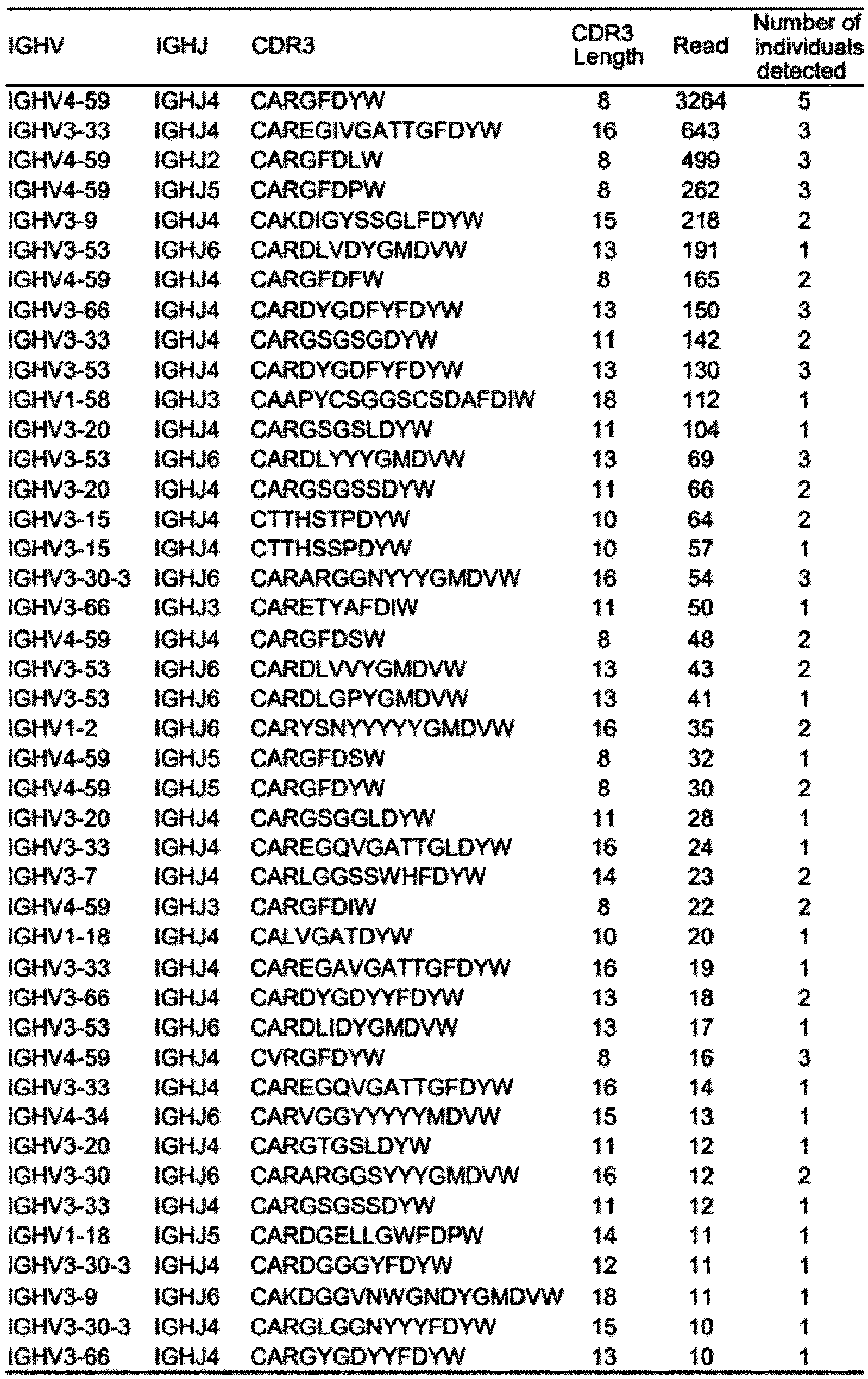
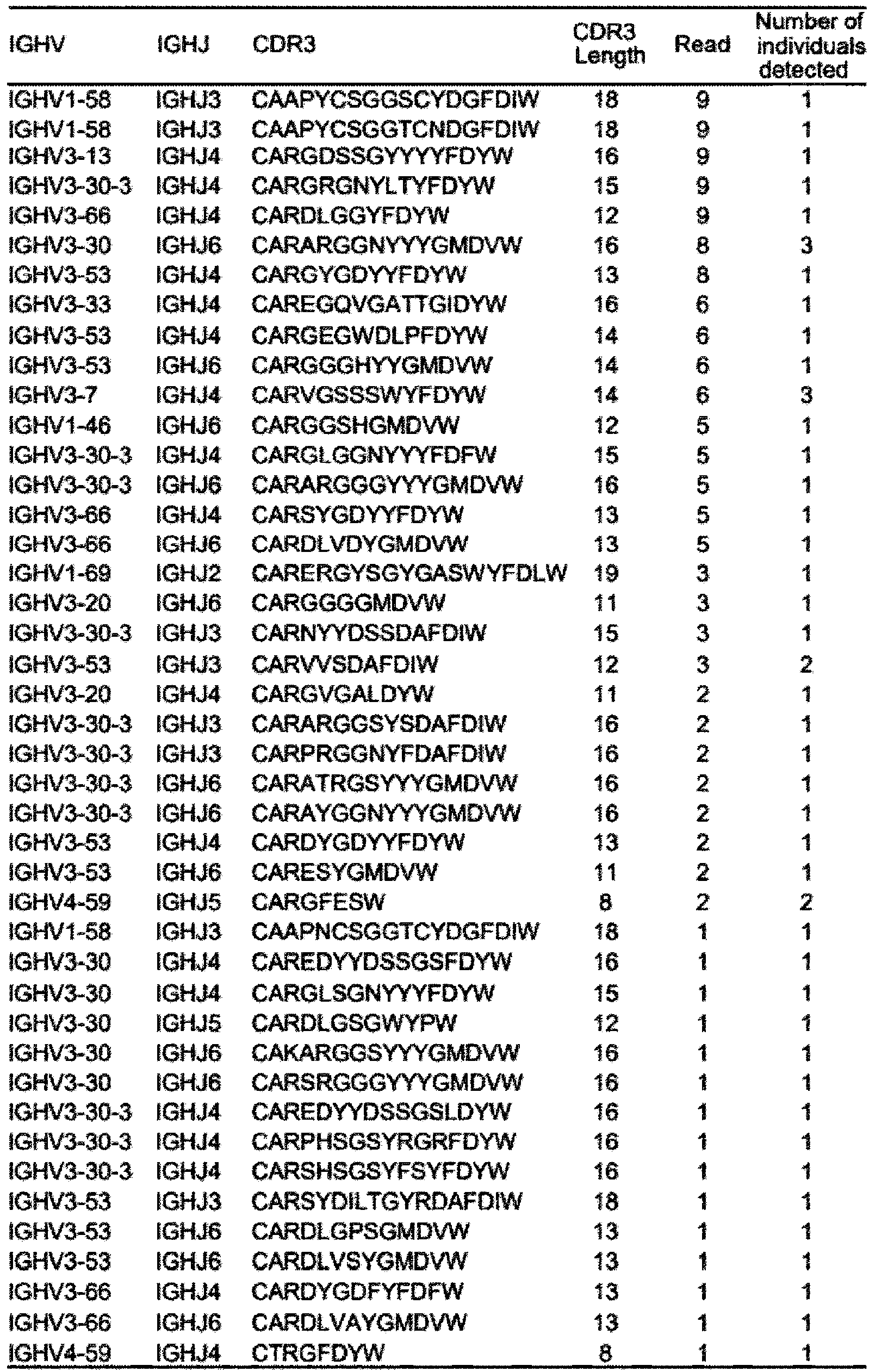
- Characteristics of SARS-CoV-2-specific sequences induced by SARS-CoV-2 infection or vaccination Alignment of SARS-CoV-2-specific sequences from infected and vaccinated individuals.
- the CDR3 amino acid sequences of IGHV4-59, IGHV3-33, IGHV3-53, IGHV3-66, and IGHV3-9, which frequently appear in infected and vaccinated individuals (Table 3A and Table 3B, SARS-COV-2-specific sequences (perfect match) from infected and vaccinated individuals), were aligned using ClustalOmega, and sequence logos were drawn using the ggseqlogo package. Characteristics of SARS-CoV-2-specific sequences induced by SARS-CoV-2 infection or vaccination. Probability of generation (pGen) values were compared.
- a histogram of pGen values for 8,977 SARS-CoV-2-specific sequences in the CoV-AbDab database and 86 perfectly matched SARS-CoV-2-specific sequences (FoundCoV) detected in infected and vaccinated individuals is shown.
- pGen values were calculated from the CDR3 amino acid sequence of each sequence using the OLGA package. Sequences with high pGen values have a high occurrence probability, and sequences with low pGen values occur rarely. Occupancy frequency of the dominant individual clones and the top 10 clones in vaccinated healthy volunteers. Individual clones with an occupancy frequency above 0.1% in vaccinated healthy volunteers are indicated by dots.
- the X-axis shows IGHV genes
- the Y-axis shows IGHJ genes
- the size of the bubble shows the percentage frequency of usage.
- Characteristics of SARS-CoV-2-specific sequences Distribution of CDR3 amino acid lengths of SARS-CoV-2-specific sequences with different CDR3 amino acid distances in the OAS database. The read abundance of SARS-CoV-2-specific sequences with different CDR3 amino acid lengths reported in the OAS database from 2011 to 2020 is shown.
- SARS-CoV-2-specific sequences with different CDR3 edit distances (LV0, LV1, LV2) are shown in separate bar graphs. Alignment of SARS-CoV-2 specific sequences with IGHV4-59, 8 amino acid CDR3 sequences.
- Sequence logos were generated with the ggseqlogo package. Comparison of probability of generation (pGen) values of SARS-CoV-2-specific sequences. Histograms show the pGen values of 8,977 SARS-CoV-2-specific sequences in the CoV-Ab database (CoV-AbDab) and 372 exact matches found in the OAS database (FoundCoV (OAS)). The X-axis shows density, and the Y-axis shows the logarithmic value (log10) of pGen. It was confirmed that vaccination with an mRNA vaccine against SARS-CoV-2 increased antibody sequences that bind to various strains (mutated strains, Omicron strain variants) in the same test subject.
- sequences matching the database in the BCR repertoire (antibody sequences with a Levenshtein distance of 0 amino acid difference in CDR3, and antibody sequences with a Levenshtein distance of 1 or less) increased within about two weeks.
- sequences matching the database in the BCR repertoire (antibody sequences with a Levenshtein distance of 0 amino acid difference in CDR3 and antibody sequences with a Levenshtein distance of 1 or less) increased within about one week.
- 1 shows an illustration of B cell subset classification. After cancer immunotherapy (booster vaccination), activated B cells increased within about a week. After cancer immunotherapy (booster vaccination), activated B cells increased within about a week.
- activated B cells increased within about one week, and a greater increase was observed in patients who developed irAE (patients 1, 2, 6, 10, and 11).
- the first antigen stimulation (vaccination) after hematopoietic stem cell transplantation increased sequences matching the database in the BCR repertoire (antibody sequences with a Levenshtein distance of 0 amino acid difference in CDR3, and antibody sequences with a Levenshtein distance of 1 or less) within two weeks after the first antigen stimulation.
- the number of sequences matching the database in the BCR repertoire (antibody sequences with a Levenshtein distance of 0 amino acid difference in CDR3, and antibody sequences with a Levenshtein distance of 1 or less) increased within one week after the second or subsequent antigen stimulation.
- the number of sequences that matched the BCR repertoire database (containing approximately 1,300 sequences independently collected from citations from research papers) increased within about a week.
- Figure 23A shows an in vitro experiment for collecting sequences to expand the database used.
- the immune response evaluation method of the present invention is a method for evaluating immune responses to specific antigens using antigen receptor repertoire analysis.
- the immune response evaluation method of the present invention may be referred to as the "Quantification of Antigen-specific Antibody Sequence (QASAS) method.”
- QASAS Quality of Antigen-specific Antibody Sequence
- An example of the immune response evaluation method of the present invention is outlined in Figure 1.
- the method for evaluating an immune response of the present invention includes a step A of preparing repertoire data [corresponding to FIG. 1(ii)] containing a group of sequences of antigen recognition sites of immune cell receptors (antigen receptors) obtained from a sample of a subject [corresponding to FIG. 1(i)], and a step B of comparing a database [corresponding to FIG. 1(iii)] containing a group of sequences of antigen recognition sites of immune cell receptors for specific antigens with the repertoire data [corresponding to FIG. 1(ii)], and detecting, from the repertoire data [corresponding to FIG. 1(ii)], a sequence that is identical to the sequence contained in the database [corresponding to FIG.
- the method of the present invention can identify sequences that show an immunologically appropriate reaction, that is, sequences that can truly react to antigens, from the many sequences contained in the repertoire data (ii) and database (iii), and therefore can evaluate immune responses at the level of mRNA expression, rather than at the level of protein expression as in conventional antibody titer measurements by ELISA.
- the immune response evaluation method of the present invention can further include a step C of deriving the number (corresponding to the "number of matching sequences (clones)" in FIG. 1) and/or frequency (corresponding to the "proportion of matching sequences” in FIG. 1) in the repertoire data of the sequences detected in the step B. This makes it possible to determine the degree of the immune response.
- the immune response evaluation method of the present invention may further include a step D of confirming the change over time in the number and/or frequency of sequences matching the database (i.e., the change in the "number of matching sequences (clones)” or the “percentage of matching sequences” on the "Day” axis in FIG. 1).
- This allows the change in the immune response for each collection time of the sample to be investigated. Note that while antibody proteins take time to reach a detectable level after an immune response and remain in the body for a long time due to their long half-life, mRNAs reach a detectable level quickly after an immune response and do not remain in the body for a long time due to their short half-life.
- the immune response can be quickly detected based on the mRNA level, and humoral immune activity can be grasped in real time.
- Step A repertoire data of sequences of antigen recognition sites of immune cell receptors obtained from a subject's sample (corresponding to FIG. 1(i)) is prepared.
- a repertoire is the totality of time-varying adaptive immune receptors possessed by a subject (immune repertoire).
- the biological sample that is the source of repertoire data is not particularly limited as long as it contains immune cells, and examples include body fluid samples such as blood and lymph, and tissue homogenates, and the like, preferably body fluid samples, more preferably blood (even more preferably peripheral blood mononuclear cells).
- the repertoire data includes sequences of antigen recognition sites of immune cell receptors.
- B cells or T cells can be selected as immune cells.
- preferred examples include shapes [a] to [c], [e], and [f].
- preferred examples include shapes [d] and [f].
- the immune cells from which the antigen recognition site sequences are derived include immune cells specific to a particular antigen, but also randomly include immune cells that are not specific to a particular antigen, with no distinction being made between them.
- the immune cells from which the antigen recognition site sequences are derived are not immune cells specific to a particular antigen narrowed down from all immune cells obtained from the above biological sample, but can typically include all immune cells identifiable from the above biological sample (for example, all B cells or all T cells identifiable from peripheral blood mononuclear cells).
- the repertoire data may include information obtained from a single subject, or may include information obtained from multiple subjects with common subject attributes.
- the sequence of the antigen recognition site of the immune cell receptor includes a gene sequence that determines the clonotype of the immune cell.
- the gene sequence may be any sequence that forms a complementarity determining region by rearrangement.
- the gene sequence may be the V and J segments of the TCR ⁇ chain, TCR ⁇ chain, and immunoglobulin L chain, as well as the V, D, and J segments of the TCR ⁇ chain, TCR ⁇ chain, and immunoglobulin H chain.
- the complementarity determining region may be at least one of CDR1, CDR2, and CDR3, but preferably includes at least CDR3.
- the repertoire data may be obtained by a repertoire analysis method.
- the repertoire analysis method is known, and involves determining the sequence (gene sequence) of the antigen recognition site of an immune cell receptor from a biological sample of a subject by next-generation sequencing.
- the repertoire data can be obtained by extracting total RNA from the biological sample of the subject, synthesizing cDNA, amplifying the gene sequence of the immune cell receptor, determining the sequence on a large scale by next-generation sequencing, and assigning the sequence (gene sequence) region of the antigen recognition site.
- nucleic acid amplification it is preferable to uniformly amplify the gene sequence of the immune cell receptor without bias (non-biased gene amplification), and the design of a multiplex primer panel for such non-biased gene amplification can also be selected based on known techniques.
- the number of sequences included in the repertoire data is not particularly limited, and may be determined according to the scale of one analysis of the next-generation sequencing used and the number of subjects who provided the repertoire data. Specifically, the number of sequences included in the repertoire data may be, for example, 100,000 to 10 million, 300,000 to 5 million, or 500,000 to 3 million immune cell receptors.
- the repertoire analysis method applicable to the present invention includes not only bulk analysis, which analyzes gene expression in immune cells at the cell population level, but also single-cell analysis, which analyzes gene expression in immune cells at the individual cell level.
- Subject is not particularly limited as long as it is a subject for which an immune response needs to be evaluated, and can be set according to the purpose of application of the method of the present invention.
- Specific examples of subjects include subjects exposed to a specific antigen, subjects receiving cancer immunotherapy, and subjects suffering from an autoimmune disease.
- biological species of subjects include humans and non-human animals (including, but not limited to, mice, rats, zebrafish, etc.).
- Subjects exposed to a specific antigen is not particularly limited as long as it is an event that leads to activation of an immune response, and examples thereof include vaccination and infection.
- subjects vaccinated with a specific antigen As subjects exposed to a specific antigen, subjects vaccinated with a specific antigen can be selected.
- the immune response of the vaccine can be evaluated not only by identifying the sequence that reacted with the vaccine in the subject's body at the time of sample collection, but also by evaluating (including predicting) the scientific validity of the vaccine (e.g., whether the vaccine exerts a predetermined function in accordance with its design). Examples of cases in which the subject is selected include form [a], form [b], and form [f].
- the method of the present invention is highly useful in that it can selectively identify immune responses to specific antigens from repertoire data, and therefore can effectively identify immune responses to specific antigens even in the case of immune responses to a vaccine.
- the type of vaccine is not particularly limited, and specific examples include nucleic acid vaccines (mRNA vaccines, DNA vaccines), viral vector vaccines, recombinant protein vaccines, inactivated vaccines, and live vaccines.
- the vaccine is preferably a vaccine designed against a pathogen described below, and preferably a vaccine designed against a virus described below, more preferably a coronavirus or influenza virus (coronavirus vaccine or influenza virus vaccine).
- the method of the present invention is capable of identifying an immune response to a specific antigen at the sequence level of the antibody's complementarity determining region, and is also capable of identifying antigen information such as the type of strain (mutant, variant) or epitope (antibody binding site) from the sequence information of the antibody's complementarity determining region. Therefore, it is possible to confirm the type of strain (mutant, variant) and the ability to recognize epitopes of antibodies produced by vaccines that use the blueprint (genetic information) of a specific antigen as an active ingredient, particularly nucleic acid vaccines, from antigen information such as epitopes linked to the sequence of the complementarity determining region in the database. From this perspective, preferred examples of vaccine types include nucleic acid vaccines (mRNA vaccines, DNA vaccines), and more preferably RNA vaccines (mRNA vaccines).
- subjects infected with a specific antigen As subjects exposed to a specific antigen, subjects infected with a specific antigen can be selected. In this case, the immune response in a specific infectious disease can be evaluated. Examples of cases in which such subjects are selected include form [c] and form [f].
- the infection is not particularly limited as long as it is an infection caused by a specific antigen, and specific examples include infections caused by antigenic organisms such as viruses, bacteria, and fungi.
- Viruses are not particularly limited, and examples include coronaviruses, influenza viruses, cytomegaloviruses, rotaviruses, herpes viruses, human immunodeficiency viruses, dengue viruses, etc.
- Coronaviruses include cold coronaviruses (HCoV-229E, HCoV-OC43, HCoV-NL63, HCoV-HKU1), severe acute respiratory syndrome coronavirus (SARS-CoV), Middle East respiratory syndrome coronavirus (MERS-CoV), and novel coronavirus (SARS-CoV-2).
- Influenza viruses include type A (including any combination of 16 types of hemagglutinin (HA) H1-H16 and 9 types of neuraminidase (NA) N1-N9), type B, and type C.
- Bacteria are not particularly limited, but examples include Pseudomonas aeruginosa, Legionella pneumophila, Yersinia spp., Escherichia coli, Vibrio bacillus, Haemophilus influenzae, gram-negative bacillus, Mycobacterium tuberculosis, etc.
- Fungi are not particularly limited, but examples include Candida and Aspergillus.
- antigens that are pathogens include not only antigens for which infection has been confirmed and the species and strains have been identified, but also pathogens such as mutant strains that may newly emerge in the future.
- antigen information can be identified from sequence information related to the repertoire data via an existing database, so that in the diagnosis of infectious diseases, not only can it be determined what type of pathogen is causing the infection, but it can also be determined whether or not an existing vaccine can be applied to the newly emerging pathogen.
- Subjects with high levels of other antibodies Subjects exposed to a specific antigen (subjects described in the above items "1-2-1-1" and "1-2-1-2") may also have high levels of other antibodies at the time of specimen collection.
- the other antibodies refer to antibodies different from those produced by exposure to the specific antigen. Examples of cases in which such subjects are selected include forms [a] to [c] and [f].
- Subjects who have high levels of other antibodies include subjects who have been administered an antibody drug, vaccinated, and/or infected. Below, subjects who have been administered an antibody drug are also referred to as “subject (S1),” subjects who have been vaccinated are also referred to as “subject (S2),” and subjects who have been infected are also referred to as “subject (S3).”
- the subject (S1) is not particularly limited, but specific examples include subjects who have undergone immunosuppressive treatment or patients suffering from autoimmune diseases.
- immunosuppressive treatment include the treatments described in section 1-2-3 below.
- autoimmune diseases include the diseases described in section 1-2-4 below.
- An example of an antibody drug is an antibody drug administered to a subject who has undergone immunosuppressive treatment, for example, to counter exposure to a specific antigen.
- examples of antibody drugs when the specific antigen is the new coronavirus include tixagevimab/silgavimab, casirivimab/imdevimab, sotrovimab, etc.
- Other examples of antibody drugs include antibody drugs used to treat autoimmune diseases. This antibody drug can be appropriately selected by a person skilled in the art depending on the type of autoimmune disease.
- the vaccine that satisfies the condition for subject (S2) is a different vaccine from the vaccine that satisfies the condition for the subject in the above item "1-2-1-1".
- the other vaccine include a vaccine that produces antibodies different from the antibodies produced by the vaccine that satisfies the condition for the subject in the above item "1-2-1-1” (i.e., a specific antigen), a vaccine designed to target another antigen (an antigen other than the specific antigen), etc.
- the pathogen in the subject (S3) is not particularly limited. However, when the subject in the above item "1-2-1-2" further becomes the subject (S3), the pathogen that satisfies the condition of the subject (S3) is another pathogen that is different (at least at the strain class level) from the pathogen (i.e., the specific antigen) that satisfies the condition of the subject in the above item "1-2-1-2". Specifically, the other pathogen can be appropriately selected from the pathogens listed in the above item "1-2-1-2".
- the method of the present invention can selectively identify an immune response to a specific antigen from repertoire data without being affected by the amount of antibody protein in the body, so that if an immune response to a specific antigen occurs, it can be identified even in subjects who have high levels of other antibodies.
- the relationship between the timing of administration of the antibody drug and the timing of antigen exposure is not particularly limited, as long as the biological sample related to the repertoire data is obtained while the antibody related to the antibody drug is retained in the body. Therefore, within the above limits, the timing of administration of the antibody drug may be before antigen exposure, simultaneously with antigen exposure, or after antigen exposure, and is preferably before antigen exposure.
- Subjects who have undergone cancer immunotherapy In the body of a subject who has undergone cancer immunotherapy (a treatment that strengthens the body's inherent immune response to cancer), immune cells re-recognize cancer antigens that were previously tolerant. In principle, this re-recognition of immune cells to cancer antigens (neoantigens) is accompanied by a change in the immune environment (activation of immune response) similar to that of antigen exposure.
- the method of the present invention can evaluate immune responses activated by antigen exposure, and therefore immune responses activated by cancer immunotherapy can also be evaluated by the same mechanism. Therefore, subjects who have undergone cancer immunotherapy can be selected as subjects. Examples of cases in which such subjects are selected include form [d] and form [f].
- a cancer antigen can be selected as the specific antigen.
- Cancer immunotherapy involves treatments that release the brakes placed on the immune system by cancer, such as the administration of immune checkpoint inhibitors, which activate cancer immunity by inhibiting immunosuppressive signal transduction.
- immune checkpoint inhibitors include anti-PD-1 antibodies, anti-PD-L1 antibodies, and anti-CTLA4 antibodies.
- the immune response of a subject who has undergone cancer immunotherapy it is possible to determine whether the prescribed function of cancer immunotherapy is being exerted (i.e., the effectiveness of cancer immunotherapy). Furthermore, according to the method of the present invention in the case where the subject is selected, it is possible to identify an antigen that matches the T cell receptor database as a clinically significant T cell receptor sequence from sequence information related to the T cell receptor repertoire data of a sample collected after cancer immunotherapy, and therefore an antigen (typically a peptide) that binds to the T cell receptor sequence can be identified as a true neoantigen.
- an antigen typically a peptide
- subjects who have received immunosuppressive treatment can be selected. Examples of cases in which such subjects are selected include case b4 of form [b] and form [f]. As the specific antigen, a vaccine can be selected.
- Immunosuppressive treatments include, but are not limited to, hematopoietic stem cell transplantation, administration of B cell depletion therapy (chemotherapy), etc.
- chemotherapy B cell depletion therapy
- When used to evaluate the immune response of a subject who has undergone immunosuppressive treatment for example, it can be determined whether or not the immune response weakened by immunosuppressive treatment has recovered due to antigen stimulation (vaccination, etc.) after immunosuppressive treatment.
- the subject When the subject is selected, it is sufficient that the subject receives antigen stimulation with a specific antigen after the immunosuppressive treatment, and the subject may or may not receive stimulation with an antigen (this antigen may be the same as the specific antigen or may be an antigen different from the specific antigen) before the immunosuppressive treatment. Even if the subject receives stimulation with the same antigen as the specific antigen before the immunosuppressive treatment, the immune system is reset by the immunosuppressive treatment. For this reason, in the method of the present invention, even if the subject receives stimulation with the same antigen as the specific antigen before the immunosuppressive treatment, the reaction due to stimulation with the specific antigen received after the immunosuppressive treatment is considered to be the primary immune reaction (see Figure 22A).
- Subjects suffering from an autoimmune disease As subjects, subjects suffering from an autoimmune disease can be selected. Examples of cases in which such subjects are selected include form [e] and form [f]. As the specific antigen, an autoantigen can be selected.
- Autoimmune diseases are not particularly limited, but examples include myasthenia gravis (autoantigen: acetylcholine receptor), immune thrombocytopenia (ITP) (autoantigen: platelets), and type 1 diabetes (autoantigen: ⁇ cells).
- autoantigen acetylcholine receptor
- ITP immune thrombocytopenia
- ⁇ cells type 1 diabetes
- the method of the present invention allows for the onset or progression of an autoimmune disease to be evaluated by identifying sequences from sequence information related to immune cell receptor repertoire data obtained from the subject that match a database of immune cell receptors for autoantigens that cause the autoimmune disease.
- the method of the present invention can also be used to evaluate side effects after drug administration.
- a database group for autoantigens is constructed, and the off-target effect of the drug can be evaluated by confirming that administration of the drug increases the number of matching sequences in the database for a target other than the designed target of the drug.
- a SARS-CoV-2 vaccine is administered, if repertoire analysis shows an increase in matching sequences in the myocardial database, rather than in the SARS-CoV-2 database, among the countless databases that make up the database group, it can be evaluated that myocarditis occurs as a side effect. This makes it possible to screen for new drugs to predict the appearance of unexpected side effects (antibodies that react to autoantigens), making it possible to develop new drugs that are safer.
- Time of Acquisition of Repertoire Data There is no particular limitation on the time of acquisition of the subject's repertoire data (i.e., the time of collection of the biological sample that is the source of the repertoire data), and it can be selected from any time when an immune response needs to be evaluated. Preferably, a time is selected that coincides with the activation time of the mRNA of the immune cell receptor for a specific antigen (i.e., the time of clonal proliferation of immune cells that have responded to a specific antigen in the repertoire).
- a specific activation time of the mRNA does not depend on the type of specific antigen, and can be, for example, 4 to 18 days after immune stimulation by a specific antigen.
- the time period may be, for example, 11 to 20 days or 11 to 18 days, preferably 12 to 16 days, and more preferably 13 to 15 days, after antigen stimulation for the primary response; and in the case of evaluating a secondary, tertiary, ..., or n-th response (n is an integer representing the number of immune responses.
- secondary and subsequent responses are collectively also referred to as “secondary and subsequent responses" (e.g., when evaluating booster vaccination of a vaccine or cancer immunotherapy), the time period may be, for example, 3 to 10 days or 4 to 10 days, preferably 5 to 9 days, and more preferably 6 to 8 days, after antigen stimulation for the secondary, tertiary, ..., or n-th response, respectively.
- step B a database of sequences of antigen recognition sites for specific antigens [corresponding to FIG. 1(iii)] is collated with the repertoire data [corresponding to FIG. 1(ii)], and sequences that are identical to the sequences included in the database at the amino acid sequence level or that are identical except for two or less amino acid residues [corresponding to "sequences matching the database" in FIG. 1] are detected from the repertoire data.
- the database contains sequences of antigen recognition sites for specific antigens.
- the specific antigens are the same as the specific antigens described above in "1-2. Subjects.”
- the sequences of the antigen recognition sites are as described above as the sequences of the antigen recognition sites in "1-1. Repertoire data.”
- the database may also contain additional information (hereinafter simply referred to as "additional information") such as antibody sequences, nanobody sequences, and variable region sequences for a specific antigen, the presence or absence of neutralizing activity of the antibody, epitope regions in a specific antigen, and species and strains (mutants, variants) of the antigenic organism.
- additional information such as antibody sequences, nanobody sequences, and variable region sequences for a specific antigen, the presence or absence of neutralizing activity of the antibody, epitope regions in a specific antigen, and species and strains (mutants, variants) of the antigenic organism.
- CoV-AbDab a database managed by the Oxford Protein Informatics Group at the Department of Statistics, University of Oxford
- CoV-AbDab contains information on ⁇ -coronaviruses such as SARS-CoV-2, SARS-CoV-1, and MERS-CoV, and includes information on the sequences of antibodies that can bind to each ⁇ -coronavirus, nanobody sequences, variable region sequences, the presence or absence of neutralizing activity of the antibodies, epitope regions, species and strains (mutations, variants) of each ⁇ -coronavirus, etc.
- the database may be a publicly available database as long as it contains the above information, or may be a database created by collecting data for each specific antigen.
- the sequence of the antigen recognition site for a specific antigen to be included in the database can be appropriately obtained using a known method. Examples of the method for collecting data include the following methods [1] to [3]. To collect the database, any of the methods [1] to [3] may be used, or two or more of the methods may be used in combination. [1] Collection from existing literature and/or public databases [2] Collection of sequences through in vitro experiments [3] Collection of sequences through in silico
- sequences of known antigen recognition sites for a specific antigen are selected from existing literature and/or public databases, and information to be included in the database can be compiled by associating additional known information as necessary.
- the method of [2] above includes, for example, a step of preparing a labeled protein by labeling a protein synthesized from an antigen sequence, a step of sorting cells that bind to the labeled protein from among a population of immune cells, and a step of acquiring the sequences of the sorted cells as information to be collected in a database.
- the sequences of antigen recognition sites of immune cell receptors can be collected using machine learning to predict antigen/antigen receptor binding.
- binding prediction include antigen/epitope binding analysis using a protein language model (e.g., Bioinformatics, Volume 37, Issue Supplement_1, July 2021, Pages i237-i244, and Bioinformatics, Volume 39, Issue 1, January 2023, btac820), protein structure analysis using a diffusion model (e.g., Nature. 2024 May 8. doi: 10.1038/s41586-024-07487-w), and binding analysis thereof.
- a protein language model e.g., Bioinformatics, Volume 37, Issue Supplement_1, July 2021, Pages i237-i244, and Bioinformatics, Volume 39, Issue 1, January 2023, btac820
- protein structure analysis e.g., Nature. 2024 May 8. doi: 10.1038/s41586-024-07487-w
- immune cells capable of binding to a specific antigen are obtained from a group of subjects in which an immune reaction has occurred due to the specific antigen, and sequences of antigen recognition sites of immune cell receptors can be collected by repertoire analysis.
- the method combining the above-mentioned [2] and [3] preferably includes the following steps.
- FIG. 2 A conceptual diagram of an example of creating a database by this method is shown in FIG. 2.
- the activation time of immune cell receptor mRNA i.e., the time of clonal proliferation of immune cells reacting to a specific antigen in the repertoire
- clonal proliferation period based on the predetermined period after antigen stimulation (clonal proliferation period) described above in "1-3.
- Time of acquiring repertoire data it is possible to acquire time-series immune cell receptor repertoire data including before the activation (T bf : before clonal proliferation), the activation period (T ex : clonal proliferation period), and after the activation (T af : after clonal proliferation: reduction of the proliferated clone).
- T bf before clonal proliferation
- T ex clonal proliferation period
- T af after clonal proliferation: reduction of the proliferated clone.
- huge amounts of data for example, about 200,000 reads each
- a sequence that is identical to the sequences contained in the database at the amino acid sequence level or is identical to the sequences contained in the database in a portion other than two or less amino acid residues is, in other words, a sequence that is a completely identical amino acid sequence or an amino acid sequence with a mismatch of one to two residues (preferably one residue) with respect to the sequence in the database (similar sequence).
- Levenshtein distance method Any method capable of comparing amino acid sequences can be used to detect matching sequences.
- Levenshtein distance method can detect amino acid sequences whose Levenshtein distance (edit distance, i.e., the minimum number of single-character substitutions, insertions, and deletions required to make one string match another) is 0 to 2 (preferably 0 to 1). Therefore, among matching sequences, the Levenshtein distance of a completely identical sequence is 0, and the Levenshtein distance of a similar sequence is 1 to 2 (preferably 1).
- step C the number (corresponding to "number of matching sequences (clones)" in FIG. 1) and/or frequency (corresponding to "proportion of matching sequences” in FIG. 1) in the repertoire data of the sequences identified in step B is derived.
- step C can be optionally included regardless of the purpose of application of the method of the present invention.
- step C the number of sequences detected as being identical or similar to sequences in the database (matching sequences) contained in the repertoire data and/or the frequency at which they appear are derived.
- the number and/or frequency can be derived appropriately based on known methods. The greater the number and/or frequency of sequences identified as being identical or similar to sequences in the database (matching sequences) in the repertoire data, the stronger and/or more frequently an immune response to a specific antigen occurs.
- step D the change over time in the number and/or frequency of sequences matching the database obtained in step C is confirmed.
- Examples of cases in which the present invention includes step D include form [a], form [b], form [d], form [e] case e2 and case e3, and form [f].
- step D is not required.
- the activation period of immune cell receptor mRNA i.e., the clonal proliferation period of immune cells that have responded to a specific antigen in the repertoire
- the activation period after antigen stimulation for a primary response is, for example, 11-20 days or 11-18 days, preferably 12-16 days, and more preferably 13-15 days
- the activation period after antigen stimulation for a secondary or subsequent response is, for example, 3-10 days or 4-10 days, preferably 5-9 days, and more preferably 6-8 days.
- the period when an increase in the number and/or frequency of sequences matching the database is observed corresponds to either the activation period after antigen stimulation for a primary response or the activation period after antigen stimulation for a secondary or subsequent response, it is possible to determine whether stimulation with a specific antigen caused a primary response or a secondary response. Specifically, if an increase in the number and/or frequency of sequences matching the database is observed 11 to 20 days or 11 to 18 days (preferably 12 to 16 days, more preferably 13 to 15 days) after stimulation with a specific antigen, the immune response caused by the stimulation can be determined to be a primary response.
- the immune response caused by the stimulation can be determined to be a secondary or subsequent response.
- Process E Process E', Process F, Process G
- the method of the present invention may further include other steps as long as the effects of the present invention are not impaired.
- the other steps may be selected depending on the application purpose of the method of the present invention. The decision can be made based on the above.
- Step E and Step E' When an immune response is evaluated for repertoire data of a subject vaccinated with a nucleic acid vaccine, the method of the present invention may further include step E and/or step E'.
- the case including step E include case a1 to case a3 of form [a] and form [f].
- Examples of the case including step E' include case a4 of form [a] and form [f].
- step E the scientific validity of the nucleic acid vaccine is evaluated by checking the information contained in the database for sequences confirmed to have an increase over time in the number and/or frequency of sequences matching the database.
- the information contained in the database is selected from the group consisting of: [1] information on the presence or absence of neutralizing activity against an antigen, [2] information on the species or strain of the antigen, and [3] information on the epitope.
- the evaluation of scientific validity is selected from the group consisting of: [1] evaluating the nucleic acid vaccine as having clinical efficacy in producing antibodies with neutralizing activity when it is confirmed that the neutralizing activity is present; [2] evaluating the nucleic acid vaccine as being compatible with the target antigen when it is confirmed that the species or strain is the same as the species or strain of the target antigen for which the nucleic acid vaccine was designed; and [3] evaluating the nucleic acid vaccine as being compatible with the target epitope when it is confirmed that the epitope is the same as the target epitope for which the nucleic acid vaccine was designed.
- the above case [1] corresponds to case a1 of form [a]
- the above case [2] corresponds to case a2 of form [a]
- the above case [3] corresponds to case a3 of form [a].
- Process E allows for confirmation of the scientific validity of a nucleic acid vaccine that contains the blueprint (genetic information) of a specific antigen as an active ingredient, that it can produce antibodies with neutralizing activity in the body, that it can produce antibodies that can recognize the target antigen (species or strain) as designed, or that it can produce antibodies that can recognize the target epitope as designed.
- step E' if the sequence confirmed to have an increase over time in the number and/or frequency of sequences matching the database is identical (i.e., a perfect match) to the sequence contained in the database at the amino acid sequence level, the scientific validity of the nucleic acid vaccine is evaluated as having the ability to produce antibodies whose antigen recognition site perfectly matches that of antibodies acquired through infection. According to the present invention, it is possible to detect many sequences that perfectly match the antigen recognition site of an immune cell receptor.
- amino acid sequence of the antigen recognition site sequence is, for example, a sequence consisting of 10 amino acids, there are 20 types of amino acids, so theoretically 20 ⁇ 10 types of sequences are required to find a perfect match, and considering this, it is recognized that the ability of the present invention to detect many perfect matches is surprisingly efficient.
- step E' makes it possible to confirm the scientific validity that a nucleic acid vaccine administered to a healthy subject can produce antibodies in the body of the healthy subject that completely match antibodies (having sequences included in the database) acquired by a patient as a result of infection with an antigen such as a virus.
- Step F When evaluating immune responses to repertoire data of a subject who has been inoculated with a vaccine (i.e., a specific antigen) and who has a high level of other antibodies (i.e., antibodies different from the antibodies produced by inoculation of the vaccine as the specific antigen) in his/her body at the time of specimen collection, the method of the present invention may further include step F.
- cases including step F include cases b1 to b3 of form [b] and form [f].
- the subject in this case can be selected from subjects (S1) to (S3) described above in "1-2-1-3.
- Subjects with high levels of other antibodies Specifically, this includes subjects who have been vaccinated with a specific antigen, who have been administered an antibody drug, who have been vaccinated with another vaccine, and/or who have been infected.
- the vaccine is determined to be functional if the number and/or frequency of sequences matching the database increases over time.
- other antibodies specifically, antibodies related to antibody drugs, antibodies produced in an immune response to other vaccines, or antibodies produced in an immune response to pathogens
- S1 to S3 due to administration of antibody drugs, inoculation with other vaccines, or infection.
- these other antibodies are maintained at high levels in the bodies of subjects (S1) to (S3).
- the method of the present invention can selectively identify immune responses to specific antigens from repertoire data without being affected by the amount of antibody protein in the body, so even if a subject has high levels of other antibodies, if an immune response occurs to the vaccine as a specific antigen, that immune response can be selectively identified.
- Process G When an immune response is evaluated for repertoire data of a subject who has undergone immunosuppressive treatment such as hematopoietic stem cell transplantation or B cell depletion therapy, the method of the present invention may further include step G.
- the case including step G include case b4 of form [b] and form [f].
- Step F may further include step G of judging whether or not immune function has been restored based on whether or not there has been an increase over time in the number and/or frequency of sequences matching the database. For example, when an increase in the number and/or frequency of sequences matching the database is observed in the repertoire data of a subject who has been vaccinated with a specific antigen after undergoing immunosuppressive treatment, it is possible to confirm that immune function that had been weakened by the immunosuppressive treatment has been restored.
- Step H In the case where the repertoire data prepared in step A is obtained from a specimen of a subject who was inoculated with a vaccine (existing vaccine) against a virus strain ST1 that has previously emerged, and the database used for collation in step B includes a group of sequences of antigen recognition sites of immune cell receptors against a virus strain ST2 (e.g., a mutant strain of the virus strain ST1) for which the effectiveness of the vaccine (existing vaccine) is unknown, the present invention can further include a step H of evaluating that the vaccine (existing vaccine) is also effective against the virus strain ST2 when a sequence detected in step B is present. In this case, it becomes possible to determine that the existing vaccine is effectively used against the newly emerged virus strain. Conversely, when no sequence is detected in step B, it becomes possible to determine that a new vaccine needs to be developed.
- the vaccine existing vaccine
- PBMCs Peripheral blood mononuclear cells
- PBMC samples were stored at ⁇ 80°C until analysis using CELLBANKER (ZENOGEN PHARMA, Fukushima, Japan).
- Total RNA was extracted with TRIzol LS (Thermo Fisher Scientific, Waltham, MA, USA) and purified using the RNeasy Mini Kit (Qiagen, Hilden, Germany) according to the manufacturer's protocol.
- RNA quantity and purity were measured using an Agilent 2200 TapeStation (Agilent Technologies, Santa Clara, CA, USA). Serum samples were obtained by centrifuging blood samples at 1000 xg for 10 min at room temperature and immediately transferring to a freezer kept at -80°C.
- SARS-CoV-2-specific immunoglobulin antibody assays Three fully automated commercially available immunoassays were used.
- the Abbott SARS-CoV-2 IgG II Quant (Abbott Laboratories, Sligo, Ireland) is a chemiluminescent microparticle immunoassay (CMIA) designed for the quantitative measurement of IgG antibodies against the receptor-binding domain (RBD) of the S1 subunit of the spike protein of SARS-CoV-2. Tests were performed on an Abbott Architect i2000SR system (Abbott Laboratories).
- the Roche Elecsys anti-SARS-CoV-2 S (Roche Diagnostics, Basel, Switzerland) is an electrochemiluminescent immunoassay (ECLIA) for the quantitative measurement of total Ig antibodies against the RBD of the spike protein of SARS-CoV-2. Tests were performed on a Roche Cobas e601 system (Roche Diagnostics).
- the Atellica IM SARS CoV-2 IgG (Siemens Healthcare Diagnostics, Er Weg, Germany) is a chemiluminescent immunoassay (CLIA) designed to quantitatively measure IgG antibodies against the RBD of the S1 subunit of the spike protein of SARS-CoV-2. The test was performed on an Atellica IM automated analyzer (Siemens Healthcare Diagnostics).
- Antibody titers against nucleocapsid protein were measured using the QuaResearch COVID-19 Human IgM IgG ELISA Kit (nucleocapsid protein) (Cellspect, Inc., RCOEL961-N, Iwate, Japan). This kit detects antibody titers based on the indirect ELISA method and comes with different antigenic proteins immobilized.
- the ELISA kit (nucleocapsid protein) plate has SARS-CoV-2 recombinant nucleocapsid protein (full-length) expressed in E. coli immobilized on it. Nucleocapsid protein in serum samples was measured according to the manufacturer's measurement protocol.
- PBMCs were stained for 20 min at 4°C with the following anti-human antibodies for T cell lines: CD3 APC, CD4 BV510, and CD8 BV711 (all BD Biosciences, San Diego, CA, USA), and for B cell lines: CD19 BV510, CD27 BV421, IgD BV711, and CD38 BV510 (all Biolegend, San Diego, CA, USA). Isotype-matched antibodies were used as controls.
- Flow cytometry analysis was performed using a BD FACSAria III instrument (BD Biosciences).
- CD3+CD4+ cells were defined as helper T cells.
- CD19+CD27+IgD- were defined as class-switched B cells.
- CD19+CD27+CD38+ were defined as plasmablasts.
- B cell receptor repertoire analysis was performed using a non-biased next-generation sequencer developed by Repertoire Genesis Co., Ltd. Briefly, cDNA was synthesized from total RNA using polyT18 primer (BSL-18E) and Superscript III reverse transcriptase (Invitrogen, California, USA). After double-stranded (ds)-cDNA was synthesized, P10EA/P20EA dsDNA adapters were ligated and cut with NotI restriction enzyme. Nested PCR was performed with IgG constant region-specific primers (CG1 and CG2) and P20EA using KAPA HiFi DNA Polymerase (Kapa Biosystems, Woburn, MA, USA).
- Amplicon libraries were prepared by amplifying the second PCR products using P22EA-ST1 and CG-ST1-R. Index (barcode) sequences were added by amplification using the Nextera XT Index Kit v2 Set A (Illumina, San Diego, CA, USA). Sequencing was performed using an Illumina MiSeq paired-end platform (2 ⁇ 300 bp). BCR sequences were assigned based on identity to reference sequences in the international ImMunoGeneTics information system® (IMGT) database (http://www.imgt.org) using repertoire analysis software developed in-house by Repertoire Genesis Co., Ltd. (Osaka, Japan).
- IMGT international ImMunoGeneTics information system®
- COVID-19-specific antibody sequences were downloaded from CoV-AbDab (http://opig.stats.ox.ac.uk/webapps/covabdab/). Data updated on December 20, 2022, containing 12,004 entries, were used as reference. Unpaired antibody sequences reported from 2011 to 2021 were downloaded from The Observed Antibody Space database (OAS, http://opig.stats.ox.ac.uk/webapps/oas/) based on the "heavy chain” and "IGHG” attributes. A total of 260,856,092 sequences were used to validate the method.
- sequences with the "Binds to" or “Neutralizing Vs” attribute of SARS-CoV2 were considered to be SARS-CoV-2 specific sequences regardless of the mutant.
- sequences with the "Neutralizing Vs” attribute of SARS-CoV2 were classified as neutralizing antibodies (Neut+), and sequences that did not include SARS-CoV-2 in the "Neutralising Vs” attribute and included SARS-CoV-2 in the "Not Neutralising Vs” attribute were classified as non-neutralizing antibodies (Neut-).
- CoV-AbDab Analysis of OAS public databases using CoV-AbDab The usefulness of CoV-AbDab was verified using previously reported repertoire data before and after the COVID-19 pandemic. From the obtained 12,004 CoV-AbDab data, a reference table with 8,977 identical V and J gene names and CDR3 amino acids of human sequences was used to search for SARS-CoV-2-specific sequences in public databases and the data of the above patient cohort ( Figure 2A). First, a total of 260,856,092 sequences published between 2011 and 2021 from the OAS public database were analyzed using CoV-AbDab.
- BCR repertoire data from healthy volunteers who received the mRNA SARS-CoV-2 vaccine using CoV-AbDab was analyzed BCR repertoires from blood samples from healthy volunteers who received the first and second doses of the mRNA SARS-CoV-2 vaccine (monovalent BNT162b2 [B.1.1.529], Pfizer).
- Case 1 (T/C patient 1): A 63-year-old woman with acute myeloid leukemia with myelodysplasia-related changes underwent cord blood transplantation (CBT). She was administered tixagevimab/silgavimab 338 days after CBT, and received the fourth dose (second dose after CBT) of mRNA vaccine (bivalent BNT162b2) 348 days after CBT.
- Case 2 (T/C patient 2): A 40-year-old man with B-cell acute lymphoblastic leukemia underwent unrelated bone marrow transplantation (u-BMT). Tixagevimab/silgavimab was administered 212 days after u-BMT, and the first mRNA vaccine (monovalent BNT162b2) was administered 218 days after u-BMT.
- Vaccination responses after hematopoietic stem cell transplantation depend on T and B cell reconstitution.
- flow cytometry analysis was performed to evaluate T and B cell reconstitution.
- the presence of helper T cells (CD3+CD4+), class-switched B cells (CD19+CD27+IgD-), and plasmablasts (CD19+CD27+CD38+) was confirmed ( Figure 4A, Figure 4B).
- BCR repertoire analysis using CoV-AbDab clearly detected responses to mRNA SARS-CoV-2 vaccination even after tixagevimab/silgavimab administration ( Figure 4A, Figure 4B, Figure 8).
- SARS-CoV-2-specific sequences frequently contained IGHV4-59 and short CDR3 amino acids. These sequences were also detected most frequently in multiple individuals and post-pandemic samples in the results of this experiment, but some were also detected in pre-pandemic samples in 2019, albeit at a much lower frequency. IGHV4-59 sequences pair with the light chain of IGKV3-20/IGKJ1 and have not been reported to have neutralizing activity. There are also reports that public IGHV4-59/IGKV3 antibodies bind to the S2 domain and cross-react with SARS-CoV-1. This sequence has the potential to react with a wide range of coronavirus strains.
- SARS-CoV-2-specific sequences are characterized by short CDR3 sequences and high pGen values. Since sequences with high pGen values among known SARS-CoV-2-specific sequences increase significantly after infection or vaccination, it is believed that the initial immune response upon exposure to unknown COVID-19 antigens is often mediated by more abundant, common SARS-CoV-2-specific sequences. According to the method of this example, early infection immune responses can be reliably detected using known SARS-CoV-2-specific sequences with abundant high pGen values in any individual.
- the method of this embodiment has the following advantages:
- the antibodies that are measured using conventional methods have a long half-life, remaining in the blood for several months after production.
- the activity of the mRNA that is measured in the present invention is short-lived, making it possible to monitor activity on an ongoing basis without being affected by SARS-CoV-2 vaccination, infection, antibody therapy, etc.
- B cells were activated approximately two weeks after the initial vaccination or initial exposure by infection as a primary immune response. Furthermore, B cells were activated approximately one week after booster vaccination as a secondary immune response.
- the method of this embodiment can provide useful information about individual antibodies.
- the method of this embodiment can obtain useful information about the characteristics of multiple antibodies with neutralizing/non-neutralizing activity.
- the frequency of occurrence of neutralizing antibody sequences varies depending on the sample, and is expected to be an indicator of the immune defense of infected individuals.
- the detection of non-neutralizing antibodies is also a noteworthy indicator.
- an ELISpot assay has been developed to measure human IgG-secreting B cells stimulated with R-848 and IL-2 in vitro as a method for evaluating B cell responses after infection or to vaccines, but such ELISpot assays cannot report immune responses in real time or provide additional information.
- the usefulness of the method of this embodiment is not limited to confirmation of vaccine response after administration of antibody drugs, and it can be inferred that it is useful, for example, for estimating the degree of immune reconstitution after hematopoietic stem cell transplantation.
- Immune reconstitution after hematopoietic stem cell transplantation has been evaluated by total lymphocyte count, lymphocyte subset analysis, and cytokine profile analysis, but these assays are not accurate biomarkers.
- almost all hematopoietic stem cell transplant patients receive mRNA SARS-CoV-2 vaccination 3-6 months after transplantation.
- the method of this embodiment makes it possible to evaluate humoral immune reconstitution at the time of vaccination without being affected by prior infection, vaccination, or antibody therapy.
- the method of this embodiment can be used to evaluate humoral immune reconstitution after B cell depletion therapy.
- antigen-specific antibody sequences can be quantified by BCR repertoire analysis using CoV-AbDab to evaluate the response to SARS-CoV-2 vaccination.
- This example reveals that there is reactivity to mRNA SARS-CoV-2 vaccination even after administration of tixagevimab/silgavimab.
- the method of this example can be applied to clarifying disease mechanisms in the field of antigen-antibody reactions and to developing therapeutic drugs.
- Test Example 3 The same subject was vaccinated multiple times with the mRNA vaccine against SARS-CoV-2.
- the first and second doses were the original strain vaccine, the fifth dose was the original strain and BA.4/5 bivalent vaccine, and the sixth dose was the Omicron XBB vaccine.
- the spike protein which is the target of the vaccine, is composed of the S1 and S2 portions, and it is known that the RBD region in S1 is important for infection.
- BNT-162b2 (Pfizer) and mRNA-1273 (Moderna) are mRNA vaccines that target the entire S1 and S2 spike proteins of SARS-CoV-2, while MAFB-7256a (Daiichi Sankyo Vaccine), approved in Japan in 2024, is an mRNA vaccine against RBD. Information on epitopes is linked to the BCR/antibody sequence in CoV-AbDab.
- the matching sequences that increased 7 days after vaccination with BNT162b2 were for various regions of the spike protein, such as S1, RBD, and S2, while the sequences that increased 7 days after vaccination with MAFB-7256a were for RBD. Therefore, the scientific validity of the mRNA vaccine (validity for the type of target epitope) was confirmed.
- activated B cells CD21low B cells, class-switched B cells, plasmablasts, plasma cells
- activated B cells 7 days after cancer immunotherapy (booster immunization).
- activated B cells increased 7 days after cancer immunotherapy (booster immunization)
- a greater increase was observed in cases that developed irAE (side effect: autoimmune disease after administration of immune checkpoint inhibitors) (specifically, patients 1, 2, 6, 10, and 11).
- Patient 1 underwent hematopoietic stem cell transplantation after one SARS-CoV-2 infection, and then received a vaccine (specific antigen) against SARS-CoV-2.
- Patient 2 underwent hematopoietic stem cell transplantation after two SARS-CoV-2 infections, and then received a vaccine (specific antigen) against SARS-CoV-2.
- Patient 3 underwent hematopoietic stem cell transplantation after two SARS-CoV-2 infections, and then received a vaccine (specific antigen) against SARS-CoV-2 after three SARS-CoV-2 infections.
- the first antigen stimulation (vaccination) after hematopoietic stem cell transplantation increased the number of matching sequences two weeks after the first antigen stimulation.
- the second or subsequent antigen stimulation (vaccination) after hematopoietic stem cell transplantation increased the number of matching sequences one week after the second or subsequent antigen stimulation. In other words, it was confirmed that the immunity acquired by antigen stimulation before hematopoietic stem cell transplantation was reset by hematopoietic stem cell transplantation.
- the timing is about two weeks after the antigen stimulation after hematopoietic stem cell transplantation, it can be determined that the immune response to be analyzed is due to the first antigen stimulation after hematopoietic stem cell transplantation, and if the timing is about one week after the antigen stimulation after hematopoietic stem cell transplantation, it can be determined that the immune response to be analyzed is due to the second or subsequent antigen stimulation after hematopoietic stem cell transplantation.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Immunology (AREA)
- Engineering & Computer Science (AREA)
- Molecular Biology (AREA)
- Biomedical Technology (AREA)
- Organic Chemistry (AREA)
- Hematology (AREA)
- Virology (AREA)
- Urology & Nephrology (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Biochemistry (AREA)
- Cell Biology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Analytical Chemistry (AREA)
- Biophysics (AREA)
- Food Science & Technology (AREA)
- General Physics & Mathematics (AREA)
- Pathology (AREA)
- Biotechnology (AREA)
- Microbiology (AREA)
- Genetics & Genomics (AREA)
- Physics & Mathematics (AREA)
- Gastroenterology & Hepatology (AREA)
- Communicable Diseases (AREA)
- Zoology (AREA)
- Toxicology (AREA)
- Tropical Medicine & Parasitology (AREA)
- Pulmonology (AREA)
- Measuring Or Testing Involving Enzymes Or Micro-Organisms (AREA)
Abstract
Description
項1. 被験者の検体から取得した、免疫細胞レセプターの抗原認識部位の配列の群を含むレパトアデータを用意する工程Aと、
特定の抗原に対する免疫細胞レセプターの抗原認識部位の配列の群を含むデータベースと、前記レパトアデータとを照合し、前記レパトアデータの中から、前記データベースに含まれる前記配列とアミノ酸配列レベルで同一又は2個以下のアミノ酸残基以外の部分で同一である配列を検出する工程Bと、を含む、免疫反応の評価方法。
これにより、前記検体の採取時期において前記被験者の生体内で免疫学的に反応した配列を特定できる。
項2. 前記工程Bにより検出される配列の、前記レパトアデータにおける数及び/又は頻度を導出する工程Cをさらに含む、項1に記載の方法。
項3. 前記数及び/又は頻度の経時変化を確認する工程Dをさらに含む、項2に記載の方法。
項4. 前記工程Dにおいて、前記経時変化として増大が認められた時期が、前記特定の抗原による刺激後11~20日及び3~10日のいずれに該当するかを確認する、項3に記載の方法。
項5. 前記被験者が、前記特定の抗原の暴露を受けた被験者である、項1~4のいずれかに記載の方法。
項6. 前記特定の抗原の暴露がワクチンの接種である、項5に記載の方法。
項7. 前記ワクチンが核酸ワクチンである、項6に記載の方法。
項8. 前記ワクチンがコロナウイルスワクチン又はインフルエンザワクチンである、項6に記載の方法。
項9. 前記数及び/又は頻度の経時変化として増大が確認された前記配列について、前記データベースに収載された付加情報を確認することで、前記核酸ワクチンの科学的妥当性を評価する工程Eをさらに含み、
前記付加情報が、[1]抗原に対する中和活性の有無の情報、[2]抗原の種又は株の情報、及び[3]エピトープの情報からなる群より選択され、
前記科学的妥当性の評価が、[1]前記中和活性が有ることを確認した場合に、前記核酸ワクチンが中和活性を持つ抗体を産生する臨床的有効性を持つと評価すること、[2]前記種又は前記株が、前記核酸ワクチンの設計上の標的抗原の種又は株と同じであることを確認した場合に、前記核酸ワクチンが前記標的抗原に適合していると評価すること、及び[3]前記エピトープが、前記核酸ワクチンの設計上の標的エピトープと同じであることを確認した場合に、前記核酸ワクチンが前記標的エピトープに適合していると評価すること、からなる群より選択される、項7又は8に記載の方法。
項10. 前記数及び/又は頻度の経時変化として増大が確認された前記配列が、前記データベースに含まれる前記配列とアミノ酸配列レベルで同一である場合に、前記核酸ワクチンの科学的妥当性として、感染による獲得免疫による抗体と抗原認識部位が完全一致する抗体の産生能を有すると評価する工程E’をさらに含む、項7~9のいずれかに記載の方法。
項11. 前記特定の抗原の暴露が感染である、項5に記載の方法。
項12. 前記特定の抗原がコロナウイルス又はインフルエンザウイルスである、項11に記載の方法。
項13. 前記被験者が、抗体薬を投与された、ワクチンを接種された、並びに/若しくは、感染を受けた被験者であり、
前記抗体薬、前記ワクチン、及び前記感染の病原体が、前記特定の抗原の暴露により生じる抗体とは異なる他の抗体を前記被験者の体内に保有させる、項5~12のいずれかに記載の方法。
項14. 前記特定の抗原がワクチンであり、
前記数及び/又は頻度が経時的に増大する場合に、前記ワクチンが機能すると判断する工程Fをさらに含む、項13に記載の方法。
項15. 前記検体が、前記特定の抗原に対する免疫細胞レセプターのmRNAの活性化時期に採取されたものである、項5~14のいずれかに記載の方法。
項16. 前記活性化時期が、前記暴露後11~20日又は3~10日である、項15に記載の方法。
項17. 前記被験者ががん免疫療法を受けた被験者である、項1~3のいずれかに記載の方法。
項18. 前記検体が、前記特定の抗原に対する免疫細胞レセプターのmRNAの活性化時期に採取されたものである、項16に記載の方法。
項19. 前記活性化時期が、前記がん免疫療法後11~20日又は3~10日である、項17に記載の方法。
項20. 前記被験者が、免疫抑制処置を受けた被験者である、項1~16のいずれかに記載の方法。
項21. 前記免疫抑制処置が、造血幹細胞移植又はB細胞枯渇療法の投与である、項20に記載の方法。
項22. 前記免疫抑制処置が、造血幹細胞移植又はB細胞枯渇療法であり、前記数及び/又は頻度の経時変化が増大したか否かに基づいて、免疫機能の回復の有無を判定する工程Gをさらに含む、項21に記載の方法。
項23. 前記被験者が自己免疫疾患患者である、項1~16のいずれかに記載の方法。
項24. 前記レパトアデータが、過去に出現したウイルス株ST1に対するワクチンの接種を受けた被験者の検体から取得したものであり、
前記特定の抗原が、前記ワクチンによる有効性が未知であるウイルス株ST2であり、
前記検出される配列が存在する場合に、前記ワクチンが前記ウイルス株ST2にも有効と評価する工程Hをさらに含む、項1又は2に記載の方法。
項25. 前記データベースが、前記特定の抗原による免疫反応が起きた調査対象からなる集団から収集した、前記特定の抗原に対する前記免疫細胞レセプターの抗原認識部位の配列を含み、以下の工程により得られるものである、請求項1~24のいずれかに記載の方法:
前記調査対象それぞれの検体から、前記免疫細胞レセプターの抗原認識部位の配列の群を含むレパトアデータであって、前記特定の抗原に対する免疫細胞レセプターのmRNAの活性化時期Tex、前記活性化時期の前Tbf、及び前記活性化時期の後Tafにおけるレパトアデータを時系列で取得する工程、及び
前記活性化時期Texにおいて増殖が認められる免疫細胞レセプターの抗原認識部位の配列を、前記データベースに収集すべき情報として選定する工程。
項26. 活性化時期Texが、前記特定の抗原による刺激後11~20日又は3~10日である、項25に記載の方法。
項27. 前記免疫細胞が、T細胞又はB細胞である、項1~26のいずれかに記載の方法。
形態[a]ワクチンの科学的妥当性の評価
事例a1:中和活性を持つ抗体の産生能
事例a2:標的抗原の株(変異株、バリアント)の種類
事例a3:標的エピトープの種類
事例a4:獲得免疫による抗体と抗原認識部位が完全一致する抗体の産生能
形態[b]ワクチンの免疫反応の評価
事例b1:抗体薬投与による抗体保有中のワクチンの免疫反応の評価
事例b2:他のワクチン接種による抗体保有中のワクチンの免疫反応の評価
事例b3:感染による抗体保有中のワクチンの免疫反応の評価
事例b4:免疫抑制処理後のワクチン接種による免疫反応の回復評価
形態[c]感染症の診断
事例c1:病原体の特定
事例c2:新たな病原体に適合する既存ワクチンの適合性判定
形態[d]がん免疫の評価
事例d1:がん免疫療法の奏功性の判断
事例d2:真のネオアンチゲンの同定
形態[e]自己抗原に対する反応評価
事例e1:自己免疫疾患の発症診断
事例e2:自己免疫疾患の病勢診断
事例e3:投薬後の副作用評価
形態[f]網羅的バイオセンサとしての利用
工程Aでは、被験者の検体から取得した、免疫細胞レセプターの抗原認識部位の配列のレパトアデータ〔図1(i)に相当〕を用意する。
レパトアは、被験者が有している経時変化性の獲得免疫受容体の総体(免疫レパトア)である。
被験者としては、免疫反応の評価を要する被験者であれば特に限定されず、本発明の方法の適用目的に応じて設定することができる。被験者の具体例としては、特定の抗原の暴露を受けた被験者、がん免疫療法を受けた被験者、及び自己免疫疾患に罹患した被験者等が挙げられる。また、被験者の生物種としては、ヒト及び非ヒト動物(特に限定されないが、例えば、マウス、ラット、ゼブラフィッシュ等)が挙げられる。
特定の抗原の暴露としては、免疫反応の活性化をもたらすイベントであれば特に限定されず、例えば、ワクチンの接種及び感染が挙げられる。
特定の抗原の暴露を受けた被験者として、特定の抗原としてワクチンの接種を受けた被験者を選択することができる。この場合、ワクチンの免疫反応の評価としては、検体の採取時期において被験者の生体内でワクチンに対して免疫反応した配列を特定することにより、当該ワクチンに対する免疫反応性を確認するだけでなく、当該ワクチンの科学的妥当性(例えば、当該ワクチンが、その設計に適った所定の機能を発揮しているか等)を評価(予測を含む)することも挙げられる。当該被験者が選択される場合の例として、形態[a]、形態[b]、形態[f]が挙げられる。
特定の抗原の暴露を受けた被験者として、特定の抗原の感染を受けた被験者を選択することができる。この場合、特定の感染症における免疫反応を評価できる。当該被験者が選択される場合の例として、形態[c]、形態[f]が挙げられる。
特定の抗原の暴露を受けた被験者(上記項目「1-2-1-1」及び上記項目「1-2-1-2」に記載の被験者)は、検体採取時に、さらに、他の抗体を高いレベルで保持してもよい。他の抗体とは、当該特定の抗原の暴露により生じる抗体とは異なる抗体をいう。当該被験者が選択される場合の例として、形態[a]~形態[c]、形態[f]が挙げられる。
がん免疫療法(本来身体に備わっているがんに対する免疫の反応を強化する治療法)を受けた被験者の体内では、それまで寛容であったがん抗原に対して免疫細胞の再認識が行われる。このがん抗原(ネオアンチゲン)に対する免疫細胞の再認識は、原理的に、抗原暴露と同様の免疫環境の変化(免疫反応の活性化)を伴う。上記のとおり、本発明の方法は、抗原の暴露により活性化した免疫反応を評価できるため、がん免疫療法により活性化した免疫反応も同様の機序で評価することができる。従って、被験者として、がん免疫療法を受けた被験者を選択できる。当該被験者が選択される場合の例として、形態[d]、形態[f]が挙げられる。特定の抗原としては、がん抗原を選択できる。
被験者として、免疫抑制処置を受けた被験者を選択できる。当該被験者が選択される場合の例として、形態[b]の事例b4、形態[f]が挙げられる。特定の抗原としては、ワクチンを選択できる。
被験者として、自己免疫疾患に罹患した被験者を選択できる。当該被験者が選択される場合の例として、形態[e]、形態[f]が挙げられる。特定の抗原としては、自己抗原を選択できる。
被験者のレパトアデータの取得時期(つまり、レパトアデータ源となる生体試料の採取時期)については特に限定されず、免疫反応の評価を要する任意の時期から選択できる。好ましくは、特定の抗原に対する免疫細胞レセプターのmRNAの活性化時期(つまり、レパトアにおいて特定の抗原に反応した免疫細胞のクローン性増殖の時期)と合致する時期が選択される。具体的なmRNAの活性化時期としては、特定の抗原の種類に依存せず、例えば、特定の抗原による免疫刺激後4~18日が挙げられる。より具体的には、免疫反応のうち一次反応を評価する場合(例えば、感染、ワクチンの初回接種、又はがん免疫療法の免疫を評価する場合)で、一次反応のための抗原刺激後例えば11~20日又は11~18日、好ましくは12~16日、より好ましくは13~15日が挙げられ、二次反応、三次反応、・・・、又はn次反応(nは免疫反応の回数を表す整数である。これら二次以降の反応をまとめて「二次以降反応」とも記載する。)を評価する場合(例えば、ワクチンのブースター接種、又はがん免疫療法を評価する場合)で、それぞれ、二次反応、三次反応、・・・、又はn次反応のための抗原刺激後、例えば3~10日、又は4~10日、好ましくは5~9日、より好ましくは6~8日が挙げられる。
工程Bでは、特定の抗原に対する抗原認識部位の配列のデータベース〔図1(iii)に相当〕と、前記レパトアデータ〔図1(ii)に相当〕とを照合し、前記レパトアデータの中から、前記データベースに含まれる前記配列と、アミノ酸配列レベルで同一又は2個以下のアミノ酸残基以外の部分で同一である配列〔図1の「データベースと一致する配列」に相当〕を検出する。
データベースには、特定の抗原に対する抗原認識部位の配列が収載されている。特定の抗原については、上記「1-2.被験者」において述べた特定の抗原と同じである。抗原認識部位の配列については、上記「1-1.レパトアデータ」において抗原認識部位の配列として説明した通りである。
このようなデータベースの例としては、公開されているものとして、CoV-AbDab(オックスフォード大学統計学部のオックスフォード・プロテイン・インフォーマティクスグループにより管理されるデータベース)が挙げられる。CoV-AbDabは、SARS-CoV-2、SARS-CoV-1、MERS-CoVなどのβコロナウイルスに関する情報が収載されており、各βコロナウイルスに結合可能な抗体の配列・ナノボディ配列・可変領域配列、当該抗体の中和活性の有無、エピトープ領域、各βコロナウイルスの種・株(変異株、バリアント)、等の情報が含まれる。
本発明において、データベースとしては、上記の情報が含まれていれば、公開されているものされているものを用いてもよいし、特定の抗原ごとに、独自にデータ収集して作成されたものであってもよい。データベースに収載すべき特定の抗原に対する抗原認識部位の配列は公知の方法を用いて適宜取得することができる。データ収集の方法としては、以下の〔1〕~〔3〕に示す方法が挙げられる。データベースの収集のために、これら〔1〕~〔3〕に示す方法のうちいずれかの方法を用いてもよいし、2以上の方法を組み合わせて用いてもよい。
〔1〕既存の文献及び/又は公共データベースからの収集
〔2〕in vitro実験による配列収集
〔3〕in silicoによる配列収集
・前記調査対象それぞれの検体から、前記免疫細胞レセプターの抗原認識部位の配列の群を含むレパトアデータであって、前記特定の抗原に対する免疫細胞レセプターのmRNAの活性化時期Tex、前記活性化時期の前Tbf、及び前記活性化時期の後Tafにおけるレパトアデータを時系列で取得する工程、及び
・前記活性化時期Texにおいて増殖が認められる免疫細胞レセプターの抗原認識部位の配列を、前記データベースに収集すべき情報として選定する工程。
上述の公共データベース及び独自に作成されたデータベースを、あらゆる免疫反応(感染、がん免疫療法、自己免疫反応等)に係る抗原に対する抗原認識部位の配列の情報及び付加的情報を網羅するように収集することで、図3に模式的に示すような大規模なデータセット群を構築できる。このような大規模データセット群を準備しておくことで、抗原受容体レパトアをバイオセンサとして使用することで、生体内で起きている、あらゆる免疫反応(感染、がん免疫療法、自己免疫反応等)を網羅的に、少量の検体でとらえることが可能となる。当該大規模データセット群を使用する例として、形態[f]が挙げられる。
特定の抗原に対する抗原認識部位の配列のデータベース〔図1(iii)に相当〕と、前記レパトアデータ〔図1(ii)に相当〕との照合により、データベースに含まれる配列と、アミノ酸配列レベルで同一又は2個以下(好ましくは1以下)のアミノ酸残基以外の部分で同一である配列〔図1の「データベースと一致する配列」に相当〕を、レパトアデータから検出する。データベースに含まれる配列と、アミノ酸配列レベルで同一又は2個以下のアミノ酸残基以外の部分で同一である配列とは、言い換えれば、データベース中の配列に対し、完全同一のアミノ酸配列又はミスマッチが1~2残基(好ましくは1残基)であるアミノ酸配列(類似配列)に該当する配列である。
工程Cでは、工程Bにより特定される配列の、前記レパトアデータにおける数〔図1の「一致する配列の数(クローン)」に相当〕及び/又は頻度〔図1の「一致する配列の割合」に相当〕を導出する。本発明において、工程Cは、本発明の方法の適用の目的に依存せず任意で含むことができる。
工程Dでは、工程Cで得られた、データベースと一致する配列の数及び/又は頻度の経時変化を確認する。本発明が工程Dを含む場合の例として、形態[a]、形態[b]、形態[d]、形態[e]の事例e2及び事例e3、形態[f]が挙げられる。本発明の方法が形態[c]又は形態[e]の事例e1に適用される場合は、工程Dは要しない。
本発明の方法は、上記工程A~工程Dを含む場合、さらに、本発明の効果を損なわない限り、他の工程を含むことができる。他の工程は、本発明の方法の適用目的に応じて決定することができる。
核酸ワクチンの接種を受けた被験者のレパトアデータについて免疫反応を評価する場合、本発明の方法は、さらに工程E及び/又は工程E’を含むことができる。当該工程Eを含む場合の例として、形態[a]の事例a1~事例a3、形態[f]が挙げられる。工程E’を含む場合の例として、形態[a]の事例a4、形態[f]が挙げられる。
ワクチン(すなわち特定の抗原)の接種を受けた被験者であって、且つ検体採取時に他の抗体(すなわち特定の抗原としてのワクチンの接種により産生される抗体とは異なる抗体)を体内に高いレベルで保有する被験者のレパトアデータについて免疫反応を評価する場合、本発明の方法は、さらに工程Fを含むことができる。工程Fを含む場合の例として、形態[b]の事例b1~事例b3、形態[f]が挙げられる。
造血幹細胞移植又はB細胞枯渇療法等の免疫抑制処理を受けた被験者のレパトアデータについて免疫反応を評価する場合、本発明の方法は、さらに工程Gを含むことができる。工程Gを含む場合の例として、形態[b]の事例b4、形態[f]が挙げられる。
工程Aで用意するレパトアデータが、過去に出現したウイルス株ST1に対するワクチン(既存のワクチン)の接種を受けた被験者の検体から取得したものであり、工程Bで照合に用いるデータベースが、当該ワクチン(既存のワクチン)による有効性が未知であるウイルス株ST2(例えば、ウイルス株ST1の変異株等)に対する免疫細胞レセプターの抗原認識部位の配列の群を含むものである場合、本発明は、工程Bにより検出される配列が存在する場合に、当該ワクチン(既存のワクチン)がウイルス株ST2にも有効と評価する工程Hをさらに含むことができる。この場合、新たに出現したウイルス株に対して、既存のワクチンを有効利用するという判断が可能になる。反対に、工程Bにより配列が検出されなかった場合は、新たなワクチンの開発が必要であることを判断することが可能になる。
[材料及び方法]
患者
SARS-CoV-2感染後の免疫反応を評価するため、2020年8月から2020年12月にかけて神戸大学医学部附属病院にてCOVID-19患者3名から末梢血サンプルを時系列的に採取した。すべての患者は軽症と分類され、免疫抑制剤は投与されていなかった。COVID-19は、SARS-CoV-2の定量的逆転写-ポリメラーゼ連鎖反応アッセイ(rtPCR)により確認された。
末梢血サンプルは、ヘパリン含有チューブを用いて採取した。末梢血単核細胞(PBMC)は、Ficoll-Paque Plus(GE Healthcare, Little Chalfont, UK)およびSepMate-50チューブ(STEMCELL Technologies, Vancouver, Canada)を用いて密度勾配遠心分離により血液から製造者のプロトコル(STEMCELL Technologies)に従って分離された。PBMCサンプルは、CELLBANKER(ZENOGEN PHARMA、福島、日本)を用いて、分析まで-80℃で保存した。Total RNAは、TRIzol LS(Thermo Fisher Scientific, Waltham, MA, USA)で抽出し、RNeasy Mini Kit(Qiagen, Hilden, Germany)を用いて製造者のプロトコルに従って精製した。RNA量及び純度は、Agilent 2200 TapeStation(Agilent Technologies, Santa Clara, CA, USA)を用いて測定した。血清サンプルは、血液サンプルを室温で1000×gで10分間遠心分離し、直ちに-80℃に保たれたフリーザーに移すことによって得た。
3種類の完全自動化された市販のイムノアッセイを使用した。Abbott SARS-CoV-2 IgG II Quant (Abbott Laboratories, Sligo, Ireland) は、SARS-CoV-2のスパイクタンパク質のS1サブユニットの受容体結合ドメイン(RBD)に対するIgG抗体の定量測定のためにデザインされた化学発光微粒子免疫測定法(CMIA)である。検査はAbbott Architect i2000SR system (Abbott Laboratories)で行われた。Roche Elecsys anti-SARS-CoV-2 S(Roche Diagnostics, Basel, Switzerland)は、SARS-CoV-2のスパイクタンパク質のRBDに対する総Ig抗体を定量的に測定するための電気化学発光免疫測定法(ECLIA)である。検査はRoche Cobas e601システム(Roche Diagnostics社製)で実施した。Atellica IM SARS CoV-2 IgG (Siemens Healthcare Diagnostics, Erlangen, Germany) は、SARS-CoV-2のスパイクタンパク質のS1サブユニットのRBDに対するIgG抗体を定量的に測定するために設計された化学発光免疫測定法(CLIA)である。検査はAtellica IM自動分析装置(Siemens Healthcare Diagnostics)で行われた。
PBMCを、T細胞系については以下の抗ヒト抗体、すなわちCD3 APC、CD4 BV510およびCD8 BV711(all BD Biosciences, San Diego, CA, USA)、B細胞系についてはCD19 BV510、CD27 BV421, IgD BV711およびCD38 BV510(all Biolegend, San Diego, CA, USA)を使って4℃で20分間染色した。アイソタイプマッチ抗体をコントロールとして使用した。フローサイトメトリー解析は、BD FACSAria III装置(BD Biosciences)を用いて行った。CD3+CD4+細胞はヘルパーT細胞として定義した。CD19+CD27+IgD-はクラススイッチB細胞として定義した。CD19+CD27+CD38+はプラズマブラストと定義した。
BCR レパトア解析は、Repertoire Genesis株式会社が開発した非バイアス次世代シーケンサーを用いて行った。要すれば、polyT18 プライマー(BSL-18E)と Superscript III 逆転写酵素(Invitrogen、カリフォルニア、米国)を用いて全RNAからcDNAを合成した。二本鎖(ds)-cDNAを合成した後、P10EA/P20EA dsDNAアダプターをライゲーションし、NotI制限酵素で切断した。KAPA HiFi DNA Polymerase(Kapa Biosystems, Woburn, MA, USA)を用いて、IgG定常領域特異的プライマー(CG1およびCG2)およびP20EAを用いてネステッドPCRを実施した。P22EA-ST1およびCG-ST1-Rを用いて第2PCR産物を増幅することにより、アンプリコンライブラリーを調製した。インデックス(バーコード)配列は、Nextera XT Index Kit v2 Set A(Illumina, San Diego, CA, USA)を用いた増幅により追加した。配列決定は、Illumina MiSeqペアエンドプラットフォーム(2×300 bp)を用いて実施した。BCR配列は、Repertoire Genesis株式会社(大阪、日本)が独自に開発したレパトア解析ソフトウェアを用いて、国際ImMunoGeneTics information system(R) (IMGT) データベース(http://www.imgt.org)の参照配列との同一性に基づいて割り当てられた。
データ解析とグラフ化は、Rソフトウェア(バージョン4.0.2)に実装されているパッケージを使用した。レーベンシュタイン距離(編集距離)はstringdist 0.9.10パッケージを用いて算出した。ネットワーク解析は、Rに実装されたigraph 1.2.6 (https://igraph.org/r/)を用いて行った。1,000番目に頻度の高いBCRクローンタイプ(ノード)は、CDR3の1アミノ酸配列以下または1アミノ酸配列以下のLevenshtein距離で定義されるエッジで接続された。ネットワークはFruchtermanとReingoldによるforce-directed layoutアルゴリズムを用いて作成した。グラフィックスはggplot2バージョン3.3.2を用いて描画した。ヒトIGH配列の生成確率(pGen)値は、OLGAパッケージを使用して計算された(Sethna Z, Elhanati Y, Callan CG, Walczak AM, Mora T. OLGA: fast computation of generation probabilities of B- and T-cell receptor amino acid sequences and motifs. Bioinformatics. 2019;35(17):2974-2981.)。
COVID-19に特異的な抗体配列は、CoV-AbDab(http://opig.stats.ox.ac.uk/webapps/covabdab/)からダウンロードした。12,004件のエントリーを含む2022年12月20日に更新されたデータを参照として使用した。2011年から2021年に報告された対にならない抗体配列は、「重鎖」と「IGHG」属性に基づいてThe Observed Antibody Spaceデータベース(OAS、http://opig.stats.ox.ac.uk/webapps/oas/)からダウンロードされた。合計260,856,092の配列が本方法の検証に使用された。COVID-19パンデミック前の健康なボランティア12人からの合計1,259,140個のIgG抗体配列はすでに報告されている(Kitaura K, Yamashita H, Ayabe H, Shini T, Matsutani T, Suzuki R. Different Somatic Hypermutation Levels among Antibody Subclasses Disclosed by a New Next-Generation Sequencing-Based Antibody Repertoire Analysis. Front Immunol. 2017;8:389.)。クエリー配列と同一のVおよびJ遺伝子名で、CDR3のアミノ酸配列の違い(レーベンシュタイン距離、「CDR3AA Distance」)が0~2の配列(一致する配列)をデータベースから取得した。CoV-AbDabのうち、SARS-CoV2の「Binds to」または「Neutralizing Vs」属性を持つ配列は、変異体に関わらずSARS-CoV-2特異的な配列とした。また、SARS-CoV2の「Neutralizing Vs」属性を持つ配列を中和抗体(Neut+)とし、「Neutralising Vs」属性にSARS-CoV-2を含まず、「Not Neutralising Vs」属性にSARS-CoV-2含む配列を非中和抗体(Neut-)と分類した。
ELISA法による抗SARS-CoV-2スパイクIgG抗体測定におけるチキサゲビマブ/シルガビマブ投与による影響
抗SARS-CoV-2スパイク抗体測定に対するチキサゲビマブ/シルガビマブの影響を検討するため、異なるELISA法を用いた代表的な自動定量抗SARS-CoV-2スパイク免疫測定器3種類(Abbott Laboratories、Roche Diagnostics、Siemens Healthcare Diagnostics)により投与前後の時系列で抗体測定が行われた。チキサゲビマブ/シルガビマブの適応となる血液悪性腫瘍患者17名(B細胞枯渇療法n=8、造血幹細胞移植n=6、化学療法n=3)が登録された。予想通り、投与後の抗体価は全例で極めて高く、長期にわたって高い値が維持された(図1)。
COVID-19パンデミック前後の既報のレパトアデータを用いて、CoV-AbDabの有用性を検証した。得られた12,004個のCoV-AbDabデータから、8,977個の同一のVおよびJ遺伝子名とヒト配列のCDR3アミノ酸を持つ参照テーブルを用い、公開データベースと上記患者のコホートのデータからSARS-CoV-2特異的配列を検索した(図2A)。まず、OAS公開データベースから2011年から2021年の間に公開された合計260,856,092個の配列をCoV-AbDabを用いて解析した。V及びJ遺伝子名、CDR3アミノ酸配列の完全な配列一致は、パンデミック前のデータでは2019年以前の67,856,692配列中わずか132配列(0.00019%)とほとんど検出されなかった一方、2020年以降はより多くの配列(0.022%, 8,441,259配列中 の 1,817配列)が検出された(表1、OASデータベースにおけるSARS-CoV-2特異的配列の出現頻度(%)、括弧内の数字は、Disease属性がSARS-CoV-2に分類されたサンプルのデータを示す。)。SARS-CoV-2特異的配列に類似した配列を検出するため、CDR3のアミノ酸差が1または2のレーベンシュタイン(LV)距離を持つ抗体配列を検索した。LV距離が1の類似配列は2019年以前に4,641個(0.0068%)、2020年以降に12,251個(0.15%)検出された。LV距離が2の類似配列は、2019年以前は59,356個(0.087%)、2020年以降は27,484個(0.33%)検出された。2019年以前に対する2020年以降の検出率をS/N比とすると、最も良いS/N比は完全に一致した配列(LV0)で110.6、LV1で21.2、LV2で3.7であった。CDR3配列の距離によって、感度やS/N比は変化する。これらの結果から、抗体レパトアデータからSARS-CoV-2特異的な抗体配列を検出するためには、CoV-AbDab参照配列を用いた計算機的アプローチが有効であることが示された。
SARS-CoV-2感染後の免疫応答が、CoV-AbDabを用いたBCRレパトア解析で検査できるかどうかを試みた。原発性COVID-19患者3名から採取した17の血液サンプルについて、BCRレパトアの解析を行った。合計4,768,753個の配列が得られ、そのうちVおよびJ遺伝子とCDR3のアミノ酸割り当てを持つ2,858,278個のインフレーム配列が使用された。次に、CDR3配列の中にCoV-AbDabのCDR3配列と同一のものがあるかどうかを解析した(図2B)。予想通り、患者のサンプルからは、CoV-AbDabのCDR3配列と一致する配列がいくつか検出された。発症直後は一致する配列は検出されなかったが、発症後徐々に増加し、2週間前後でピークに達した(図2B)。
CoV-AbDabを用いたBCRレパトア解析によりワクチン接種後の免疫応答が検出できるかどうかを検証するため、mRNA SARS-CoV-2ワクチン(一価BNT162b2 [B.1.1.529], Pfizer)を1回目と2回目に接種した健康ボランティアの血液サンプルについてBCRレパトアを解析した。
同種造血幹細胞移植を受けた血液悪性腫瘍患者2名を対象に、チキサゲビマブ/シルガビマブ(T/C)投与後のmRNA SARS-CoV-2ワクチン接種に対する反応性を、CoV-AbDabによるBCRレパトア解析により評価した。
症例2(T/C患者2):B細胞性急性リンパ芽球性白血病を発症した40歳男性が、非血縁者間骨髄移植(u-BMT)を受けた。u-BMTから212日後にチキサゲビマブ/シルガビマブを投与し、u-BMTから218日後に最初のmRNAワクチン(1価のBNT162b2)を接種した。
最後に、SARS-CoV-2特異的配列の特徴を明らかにした。まず、参照配列の属性から、中和能または非中和能を決定した(図5A)。誘導された抗体の中和能はグループ間で異なり、ワクチン接種者では中和抗体が多く誘導され、少数の感染者では非中和抗体が多く誘導された。各サンプルに含まれる個々のSARS-CoV-2特異的配列の頻度を調べたところ、完全に一致した配列の頻度割合は、1~2個のアミノ酸が不一致の配列よりも有意に高かった(平均±SD: 0. LV0では029±0.088、LV1では0.0079±0.034、LV2では0.0074±0.041、Kruskal-WallisテストとDunn-Bonferroniポストホックテスト、LV0 vs. LV1: p<0.0001、LV0 vs. LV2: p<0.0001, LV2 vs. LV3: not significant)(図5B)。さらに、一致した抗体配列のCDR3長とVとJの組み合わせの使用率を調べた(図5C、図5D)。最も完全一致した配列はIGHV4-59/IGHJ4で、次いでIGHV3-33/IGHJ4であった。1アミノ酸のミスマッチがある配列は、IGHV1-8/IGHJ4、IGHV3-30-3/IGHJ4またはIGHJ6、IGHV3-9/IGHJ4またはIGHV3-33/IGHJ4を高い頻度で持っていた。これらのSARS-CoV-2特異的な配列は、複数の個体で検出され、公的な抗体特性を示していた(表3A及び表3B、感染者とワクチン接種者のSARS-CoV-2特異的配列(完全一致)。)。SARS-CoV-2特異的な配列として検出されたのは86配列種のみであった。完全に一致した配列の大半は、8アミノ酸の短いCDR3とIGHV4-59/IGHJ4配列であることが判明した(図6A)。既知のSARS-CoV-2特異的配列のうち、生成確率(pGen)が高いものは、感染者やワクチン接種者に多く見られた(図6B)。これらのパターンは、OASデータベースの検索結果と類似していたことから、本実施例の方法の妥当性が証明された(図9~図12)。
今回、「BCRレパトアデータ」と「標的抗原に結合するBCR配列のデータベース」を用いて、タンパク質発現ではなくmRNA発現のレベルでmRNA SARS-CoV-2ワクチン反応を評価した。実際には、この抗原特異的抗体配列の定量化方法により、チキサゲビマブ/シルガビマブ投与後にもmRNA SARS-CoV-2ワクチン接種に対する反応があることが明確に示された(図4A及び図4B)。循環型バリアントは、個々の地域で経時的に変化する。米国では現在循環している亜種の90%以上に対してチキサゲビマブ/シルガビマブが無効であることが確認されたため、FDAは2023年1月にチキサゲビマブ/シルガビマブの緊急使用認可を改訂している。現在、チキサゲビマブ/シルガビマブ以外のオミクロン株を含む新型亜種に対する様々な抗SARS-CoV-2モノクローナル抗体が開発中である。各国で適応となる抗体製剤の種類はウイルス変異により異なるが、どのタイプの抗SARS-CoV-2抗体投与後にも本実施例の方法は適用可能であると類推できる。
CoV-AbDab内のBCR/抗体配列には、株(変異株及びバリアント)に関する情報が紐づけられている。健常ボランティア1~3のBCRレパトアについて、SARS-CoV-2に対するmRNAワクチンの接種直前及び接種後7日,14日でのデータベースと一致する配列(CDR3のアミノ酸差が0のレーベンシュタイン距離を持つ抗体配列、及び1以下のレーベンシュタイン距離を持つ抗体配列)について試験例1と同様にして解析を行い、ワクチンにより産生される抗体が結合する株(変異株及びオミクロン株のバリアント)を特定した。結果を図17A~図17Cに示す。
同一被験者にSARS-CoV-2に対するmRNAワクチンを複数回接種した。1回目、2回目は起源株ワクチンを、5回目は起源株及びBA.4/5の2価ワクチンを、6回目にオミクロンXBBワクチンを接種した。1回目接種直前、1回目接種後7日,14日、2回目接種直前(1回目接種後21日)、2回目接種後3日,7日(1回目接種後24日,28日)、5回目接種直前、5回目接種後3日,7日、6回目接種直前、及び6回目接種後7日,12日でのBCRレパトアにおいて、データベースと一致する配列について試験例1と同様にして解析を行い、ワクチンにより産生される抗体が結合する株(変異株及びオミクロン株のバリアント)を特定した。結果を図18に示す。
ワクチンの標的であるスパイク蛋白はS1及びS2部分から構成されており、感染にはS1内のRBD領域が重要であることが知られている。BNT-162b2(ファイザー)、mRNA-1273(モデルナ)は、SARS-CoV-2のS1及びS2のスパイク蛋白全体を標的にするmRNAワクチンである一方、2024年日本で承認されたMAFB-7256a(第一三共ワクチン)は、RBDに対するmRNAワクチンである。CoV-AbDab内のBCR/抗体配列にはエピトープに関する情報が紐づけられている。ワクチン接種直前及びワクチン接種後7日,14日でのBCRレパトアについて、データベースと一致する配列について試験例1と同様に解析し、エピトープの特定を行った。結果を図19に示す。図19中、実線矢印は、エピトープがRBD(BNT-162b2、MAFB-7256aの標的)である配列を示し、点線矢印はエピトープがS2(BNT-162b2の標的)である配列を示し、破線矢印はエピトープが他の部位(RBDの可能性もある部位)である配列を示す。
[1]感染、ワクチン初回接種、又はワクチンブースター接種による免疫細胞のクローン性増殖の時期
感染による特定の抗原(SARS-CoV-2)の暴露、ワクチンの初回接種による特定の抗原(SARS-CoV-2に対するワクチン)の暴露、及びワクチンの2回目以降の接種(ブースター接種)による特定の抗原(SARS-CoV-2に対する様々なワクチン)の暴露それぞれについて、BCRレパトア中のデータベースと一致する配列(CDR3のアミノ酸差が0のレーベンシュタイン距離を持つ抗体配列、及び1以下のレーベンシュタイン距離を持つ抗体配列)を、時系列で、試験例1と同様に解析した。結果を図20A(感染又は初回接種による一次反応)及び図20B(ブースター接種による二次以降反応)に示す。
B細胞のサブセット分類を行い、抗体産生に繋がる活性化B細胞(CD21lowB細胞、クラススイッチB細胞、プラズマブラスト、形質細胞)が、同様のタイミングで増加していることの確認を行った。
図20Aに示すように、感染又はワクチン初回接種後においては、BCRレパトア中のデータベースと一致する配列は2週間程度で増大し、図20Bに示すように、ブースター接種後においては、BCRレパトア中のデータベースと一致する配列は1週間程度で増大した。また、図21B、図21Dに示すように、がん免疫療法(追加免疫)後においても、活性化B細胞は1週間程度で増大した。つまり、免疫細胞のクローン性増殖の時期は、共通して、免疫反応の一次反応の場合には抗原刺激後2週間程度、免疫反応の二次反応の場合には抗原刺激後1週間程度であることが確認できた。
造血幹細胞移植後の患者1~3へのワクチン接種による免疫反応を評価した。図22A及び図22Bに、患者1~3の背景及び解析結果を示す。
論文からの引用により、インフルエンザHA抗原に結合可能なB細胞レセプターの抗原認識部位の約1300配列を独自に収集し、データベースを作成した。このデータベースを用い、インフルエンザ不活化ワクチン接種後における免疫反応(被験者はインフルエンザに既感染であるため、当該ワクチン接種による免疫反応は二次以降反応である。)BCRレパトアの解析を行った結果、図23Aに示す通り、一致する配列が接種1週間後に増大(インフルエンザHA抗原の刺激による免疫反応)を確認できた。さらに、図23Bに示すin vitro実験により配列を収集し、このデータベースをさらに拡充した。配列収集を継続することによりさらに拡充されるデータベースは、同様に、当該免疫反応の評価に用いることができる。
Claims (27)
- 被験者の検体から取得した、免疫細胞レセプターの抗原認識部位の配列の群を含むレパトアデータを用意する工程Aと、
特定の抗原に対する免疫細胞レセプターの抗原認識部位の配列の群を含むデータベースと、前記レパトアデータとを照合し、前記レパトアデータの中から、前記データベースに含まれる前記配列とアミノ酸配列レベルで同一又は2個以下のアミノ酸残基以外の部分で同一である配列を検出する工程Bと、を含む、免疫反応の評価方法。 - 前記工程Bにより検出される配列の、前記レパトアデータにおける数及び/又は頻度を導出する工程Cをさらに含む、請求項1に記載の方法。
- 前記数及び/又は頻度の経時変化を確認する工程Dをさらに含む、請求項2に記載の方法。
- 前記工程Dにおいて、前記経時変化として増大が認められた時期が、前記特定の抗原による刺激後11~20日及び3~10日のいずれに該当するかを確認する、請求項3に記載の方法。
- 前記被験者が、前記特定の抗原の暴露を受けた被験者である、請求項1~4のいずれかに記載の方法。
- 前記特定の抗原の暴露がワクチンの接種である、請求項5に記載の方法。
- 前記ワクチンが核酸ワクチンである、請求項6に記載の方法。
- 前記ワクチンがコロナウイルスワクチン又はインフルエンザワクチンである、請求項6に記載の方法。
- 前記数及び/又は頻度の経時変化として増大が確認された前記配列について、前記データベースに収載された付加情報を確認することで、前記核酸ワクチンの科学的妥当性を評価する工程Eをさらに含み、
前記付加情報が、[1]抗原に対する中和活性の有無の情報、[2]抗原の種又は株の情報、及び[3]エピトープの情報からなる群より選択され、
前記科学的妥当性の評価が、[1]前記中和活性が有ることを確認した場合に、前記核酸ワクチンが中和活性を持つ抗体を産生する臨床的有効性を持つと評価すること、[2]前記種又は前記株が、前記核酸ワクチンの設計上の標的抗原の種又は株と同じであることを確認した場合に、前記核酸ワクチンが前記標的抗原に適合していると評価すること、及び[3]前記エピトープが、前記核酸ワクチンの設計上の標的エピトープと同じであることを確認した場合に、前記核酸ワクチンが前記標的エピトープに適合していると評価すること、からなる群より選択される、請求項7に記載の方法。 - 前記数及び/又は頻度の経時変化として増大が確認された前記配列が、前記データベースに含まれる前記配列とアミノ酸配列レベルで同一である場合に、前記核酸ワクチンの科学的妥当性として、感染による獲得免疫による抗体と抗原認識部位が完全一致する抗体の産生能を有すると評価する工程E’をさらに含む、請求項7に記載の方法。
- 前記特定の抗原の暴露が感染である、請求項5に記載の方法。
- 前記特定の抗原がコロナウイルス又はインフルエンザウイルスである、請求項11に記載の方法。
- 前記被験者が、抗体薬を投与された、ワクチンを接種された、並びに/若しくは、感染を受けた被験者であり、
前記抗体薬、前記ワクチン、及び前記感染の病原体が、前記特定の抗原の暴露により生じる抗体とは異なる他の抗体を前記被験者の体内に保有させる、請求項5に記載の方法。 - 前記特定の抗原がワクチンであり、
前記数及び/又は頻度が経時的に増大する場合に、前記ワクチンが機能すると判断する工程Fをさらに含む、請求項13に記載の方法。 - 前記検体が、前記特定の抗原に対する免疫細胞レセプターのmRNAの活性化時期に採取されたものである、請求項5に記載の方法。
- 前記活性化時期が、前記暴露後11~20日又は3~10日である、請求項15に記載の方法。
- 前記被験者ががん免疫療法を受けた被験者である、請求項1~3のいずれかに記載の方法。
- 前記検体が、前記特定の抗原に対する免疫細胞レセプターのmRNAの活性化時期に採取されたものである、請求項16に記載の方法。
- 前記活性化時期が、前記がん免疫療法後11~20日又は3~10日である、請求項17に記載の方法。
- 前記被験者が、免疫抑制処置を受けた被験者である、請求項1~4のいずれかに記載の方法。
- 前記免疫抑制処置が、造血幹細胞移植又はB細胞枯渇療法の投与である、請求項20に記載の方法。
- 前記免疫抑制処置が、造血幹細胞移植又はB細胞枯渇療法であり、前記数及び/又は頻度の経時変化が増大したか否かに基づいて、免疫機能の回復の有無を判定する工程Gをさらに含む、請求項21に記載の方法。
- 前記被験者が自己免疫疾患患者である、請求項1~4のいずれかに記載の方法。
- 前記レパトアデータが、過去に出現したウイルス株ST1に対するワクチンの接種を受けた被験者の検体から取得したものであり、
前記特定の抗原が、前記ワクチンによる有効性が未知であるウイルス株ST2であり、
前記検出される配列が存在する場合に、前記ワクチンが前記ウイルス株ST2にも有効と評価する工程Hをさらに含む、請求項1に記載の方法。 - 前記データベースが、前記特定の抗原による免疫反応が起きた調査対象からなる集団から収集した、前記特定の抗原に対する前記免疫細胞レセプターの抗原認識部位の配列を含み、以下の工程により得られるものである、請求項1に記載の方法:
前記調査対象それぞれの検体から、前記免疫細胞レセプターの抗原認識部位の配列の群を含むレパトアデータであって、前記特定の抗原に対する免疫細胞レセプターのmRNAの活性化時期Tex、前記活性化時期の前Tbf、及び前記活性化時期の後Tafにおけるレパトアデータを時系列で取得する工程、及び
前記活性化時期Texにおいて増殖が認められる免疫細胞レセプターの抗原認識部位の配列を、前記データベースに収集すべき情報として選定する工程。 - 活性化時期Texが、前記特定の抗原による刺激後11~20日又は3~10日である、請求項25に記載の方法。
- 前記免疫細胞が、T細胞又はB細胞である、請求項1に記載の方法。
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2025527941A JPWO2024257764A1 (ja) | 2023-06-12 | 2024-06-11 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2023-096200 | 2023-06-12 | ||
| JP2023096200 | 2023-06-12 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2024257764A1 true WO2024257764A1 (ja) | 2024-12-19 |
Family
ID=93851993
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2024/021195 Pending WO2024257764A1 (ja) | 2023-06-12 | 2024-06-11 | 免疫反応の評価方法 |
Country Status (2)
| Country | Link |
|---|---|
| JP (1) | JPWO2024257764A1 (ja) |
| WO (1) | WO2024257764A1 (ja) |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2013524848A (ja) * | 2010-05-06 | 2013-06-20 | シーケンタ インコーポレイテッド | クロノタイププロファイルを用いた健康状態および疾患状態のモニタリング |
| WO2022270631A1 (ja) * | 2021-06-25 | 2022-12-29 | Repertoire Genesis株式会社 | T細胞エピトープ配列を同定する方法およびその応用 |
-
2024
- 2024-06-11 WO PCT/JP2024/021195 patent/WO2024257764A1/ja active Pending
- 2024-06-11 JP JP2025527941A patent/JPWO2024257764A1/ja active Pending
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2013524848A (ja) * | 2010-05-06 | 2013-06-20 | シーケンタ インコーポレイテッド | クロノタイププロファイルを用いた健康状態および疾患状態のモニタリング |
| WO2022270631A1 (ja) * | 2021-06-25 | 2022-12-29 | Repertoire Genesis株式会社 | T細胞エピトープ配列を同定する方法およびその応用 |
Non-Patent Citations (24)
| Title |
|---|
| ANONYMOUS YOHEI, YAKUSHIJIN KIMIKAZU, OHJI GOH, MATSUTANI TAKAJI, HOJO WATARU, SAKAI HIRONORI, MATSUMOTO SAKUYA, WATANABE MARIKA, : " Response to mRNA SARS-CoV-2 vaccination evaluated by B-cell receptor repertoire after tixagevimab/cilgavimab administration", BRITISH JOURNAL OF HAEMATOLOGY, JOHN WILEY, HOBOKEN, USA, vol. 202, no. 3, 1 August 2023 (2023-08-01), Hoboken, USA, pages 504 - 516, XP093247834, ISSN: 0007-1048, DOI: 10.1111/bjh.18932 * |
| BAHREMAND TYAO JAMILL CPISZCZEK JGRANT JMSMOLINA K.: "COVID-19 hospitalisations in immunocompromised individuals in the Omicron era: a population-based observational study using surveillance data in British Columbia, Canada.", LANCET REG HEALTH AM., vol. 20, 2023, pages 100461 |
| BIOINFORMATICS., vol. 35, no. 17, 2019, pages 2974 - 2981 |
| DAVIS JAGRANGER KROUBAL K ET AL.: "Efficacy of tixagevimab-cilgavimab in preventing SARS-CoV-2 for patients with B-cell malignancies.", BLOOD., vol. 141, no. 2, 2023, pages 200 - 203 |
| DHAKAL BABEDIN SFENSKE T ET AL.: "Response to SARS-CoV-2 vaccination in patients after hematopoietic cell transplantation and CAR T-cell therapy.", BLOOD., vol. 138, no. 14, 2021, pages 1278 - 1281 |
| EINARSDOTTIR SMARTNER ANICKLASSON M ET AL.: "Reduced immunogenicity of a third COVID-19 vaccination among recipients of allogeneic hematopoietic stem cell transplantation.", HAEMATOLOGICA., vol. 107, no. 6, 2022, pages 1479 - 1482 |
| FUNAKOSHI YOHEI, OHJI GOH, YAKUSHIJIN KIMIKAZU, EBISAWA KEI, ARAKAWA YU, SAEGUSA JUN, MATSUMOTO HISAYUKI, IMANISHI TAKAMITSU, FUKU: "Massive surge of mRNA expression of clonal B-cell receptor in patients with COVID-19", HELIYON, ELSEVIER LTD, GB, vol. 7, no. 8, 1 August 2021 (2021-08-01), GB , pages e07748, XP093247821, ISSN: 2405-8440, DOI: 10.1016/j.heliyon.2021.e07748 * |
| FUNAKOSHI YYAKUSHIJIN KOHJI G ET AL.: "Limited increase in antibody titers following mRNA SARS-CoV-2 vaccination for more than 3 years after final dose of anti-CD20 antibody.", INT J HEMATOL., vol. 115, no. 1, 2022, pages 7 - 10 |
| FUNAKOSHI YYAKUSHIJIN KOHJI G ET AL.: "Safety and immunogenicity of the COVID-19 vaccine BNT162b2 in patients undergoing chemotherapy for solid cancer.", J INFECT CHEMOTHER., vol. 28, no. 4, 2022, pages 516 - 520 |
| IMAI MITO MKISO M ET AL.: "Efficacy of Antiviral Agents against Omicron Subvariants BQ.1.1 and XBB.", N ENGL J MED., vol. 388, no. 1, 2023, pages 89 - 91 |
| JONDREVILLE LD'AVENI MLABUSSIERE-WALLET H ET AL.: "Pre-exposure prophylaxis with tixagevimab/cilgavimab (AZD7442) prevents severe SARS-CoV-2 infection in recipients of allogeneic hematopoietic stem cell transplantation during the Omicron wave: a multicentric retrospective study of SFGM-TC.", J HEMATOL ONCOL., vol. 15, no. 1, 2022, pages 169 |
| JUNG KSHIN SNAM M ET AL.: "Performance evaluation of three automated quantitative immunoassays and their correlation with a surrogate virus neutralization test in coronavirus disease 19 patients and pre-pandemic controls.", J CLIN LAB ANAL., vol. 35, no. 9, 2021, pages e23921 |
| KITAURA KYAMASHITA HAYABE HSHINI TMATSUTANI TSUZUKI R: "Different Somatic Hypermutation Levels among Antibody Subclasses Disclosed by a New Next-Generation Sequencing-Based Antibody Repertoire Analysis", FRONT IMMUNOL., vol. 8, 2017, pages 389 |
| KOTAGIRI PRASANTI, MESCIA FEDERICA, RAE WILLIAM M., BERGAMASCHI LAURA, TUONG ZEWEN K., TURNER LORINDA, HUNTER KELVIN, GERBER PEHUÉ: "B cell receptor repertoire kinetics after SARS-CoV-2 infection and vaccination", CELL REPORTS, ELSEVIER INC, US, vol. 38, no. 7, 1 February 2022 (2022-02-01), US , pages 110393, XP093247825, ISSN: 2211-1247, DOI: 10.1016/j.celrep.2022.110393 * |
| LEVIN MJUSTIANOWSKI ADE WIT S ET AL.: "Intramuscular AZD7442 (Tixagevimab-Cilgavimab) for Prevention of Covid-19.", N ENGL J MED., vol. 386, no. 23, 2022, pages 2188 - 2200 |
| MALAHE SRKHOEK RASDALM V ET AL.: "Clinical Characteristics and Outcomes of Immunocompromised Patients With Coronavirus Disease 2019 Caused by the Omicron Variant: A Prospective", OBSERVATIONAL STUDY. CLIN INFECT DIS, vol. 76, no. 3, 2023, pages 172 - 178 |
| MASSARWEH AELIAKIM-RAZ NSTEMMER A ET AL.: "Evaluation of Seropositivity Following BNT162b2 Messenger RNA Vaccination for SARS-CoV-2 in Patients Undergoing Treatment for Cancer.", JAMA ONCOL., vol. 7, no. 8, 2021, pages 1133 - 1140 |
| SATO, Kayoko et al. Evaluation of immune responses induced by influenza vaccines using anti body repertoire analysis. 日本免疫学会総会・学術集会記録, 2021, 2-C-WS14-13-P, (Proceedings of the Japanese Society for Immunology) entire text, see abstract * |
| STUVER RSHAH GLKORDE NS ET AL.: "Activity of AZD7442 (tixagevimab-cilgavimab) against Omicron SARS-CoV-2 in patients with hematologic malignancies.", CANCER CELL, vol. 40, no. 6, 2022, pages 590 - 591 |
| TAENAKA ROBARA TKOHNO KAOKI KOGAWA R.: "Infections with the SARS-CoV-2 Omicron variant show a similar outcome as infections with the previous variants in patients with hematologic malignancies.", ANN HEMATOL., vol. 101, no. 8, 2022, pages 1877 - 1878 |
| TAN JYWEE LETAN YH ET AL.: "Favorable outcomes of COVID-19 in vaccinated hematopoietic stem cell transplant recipients: A single-center experience.", TRANSPL INFECT DIS., 2023, pages e14024 |
| VIJENTHIRA AGONG IYFOX TA ET AL.: "Outcomes of patients with hematologic malignancies and COVID-19: a systematic review and meta-analysis of 3377 patients.", BLOOD., vol. 136, no. 25, 2020, pages 2881 - 2892 |
| WATANABE MYAKUSHIJIN KFUNAKOSHI Y ET AL.: "The Safety and Immunogenicity of the BNT162b2 mRNA COVID-19 Vaccine in Japanese Patients after Allogeneic Stem Cell Transplantation.", VACCINES (BASEL)., vol. 2, 2022, pages 10 |
| XHAARD AXHAARD CD'AVENI M ET AL.: "Risk factors for a severe form of COVID-19 after allogeneic haematopoietic stem cell transplantation: a Societe Francophone de Greffe de Moelle et de Therapie cellulaire (SFGM-TC) multicentre cohort study", BR J HAEMATOL., vol. 192, no. 5, 2021, pages e121 - e124 |
Also Published As
| Publication number | Publication date |
|---|---|
| JPWO2024257764A1 (ja) | 2024-12-19 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Sureshchandra et al. | Single-cell profiling of T and B cell repertoires following SARS-CoV-2 mRNA vaccine | |
| Uzzan et al. | Ulcerative colitis is characterized by a plasmablast-skewed humoral response associated with disease activity | |
| Elyanow et al. | T cell receptor sequencing identifies prior SARS-CoV-2 infection and correlates with neutralizing antibodies and disease severity | |
| JP7430136B2 (ja) | 疲弊したt細胞に関連する疾患を治療するための方法および組成物 | |
| Mysore et al. | Protective heterologous T cell immunity in COVID-19 induced by the trivalent MMR and Tdap vaccine antigens | |
| Kuri-Cervantes et al. | Immunologic perturbations in severe COVID-19/SARS-CoV-2 infection | |
| Zheng et al. | TCR repertoire and CDR3 motif analyses depict the role of αβ T cells in Ankylosing spondylitis | |
| Dalai et al. | Clinical validation of a novel T-cell receptor sequencing assay for identification of recent or prior severe acute respiratory syndrome coronavirus 2 infection | |
| Malipiero et al. | Assessment of humoral and cellular immunity induced by the BNT162b2 SARS-CoV-2 vaccine in healthcare workers, elderly people, and immunosuppressed patients with autoimmune disease | |
| Strauli et al. | Statistical inference of a convergent antibody repertoire response to influenza vaccine | |
| Tian et al. | Dengue-specific CD8+ T cell subsets display specialized transcriptomic and TCR profiles | |
| Chaisawangwong et al. | Cross-reactivity of SARS-CoV-2–and influenza A–specific T cells in individuals exposed to SARS-CoV-2 | |
| Satti et al. | Current approaches toward identifying a correlate of immune protection from tuberculosis | |
| Hellgren et al. | Modulation of innate immune response to mRNA vaccination after SARS-CoV-2 infection or sequential vaccination in humans | |
| Staub et al. | Case series of four re-infections with a SARS-CoV-2 B. 1.351 variant, Luxembourg, February 2021 | |
| Ozer et al. | Coincident rapid expansion of two SARS-CoV-2 lineages with enhanced infectivity in Nigeria | |
| Rha et al. | SARS-CoV-2 spike-specific nasal-resident CD49a+ CD8+ memory T cells exert immediate effector functions with enhanced IFN-γ production | |
| Lee et al. | Robust immune response to the BNT162b mRNA vaccine in an elderly population vaccinated 15 months after recovery from COVID-19 | |
| Mayer-Blackwell et al. | mRNA vaccination boosts S-specific T cell memory and promotes expansion of CD45RAint TEMRA-like CD8+ T cells in COVID-19 recovered individuals | |
| WO2022049600A1 (en) | Methods for assessing t cell-specific immune response against sars-cov-2, and implementations thereof | |
| Thompson et al. | Heterologous versus homologous boosting elicits qualitatively distinct, BA. 5–cross-reactive T cells in transplant recipients | |
| Gittelman et al. | Diagnosis and tracking of SARS-CoV-2 infection by T-cell receptor sequencing | |
| Kenfack et al. | Comparative evaluation of SARS-CoV-2 serological tests shows significant variability in performance across different years of infection and between the tests | |
| CA3013119A1 (en) | Tb biomarkers | |
| WO2024257764A1 (ja) | 免疫反応の評価方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24823379 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2025527941 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2025527941 Country of ref document: JP |
|
| ENP | Entry into the national phase |
Ref document number: 2024823379 Country of ref document: EP Effective date: 20260112 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2024823379 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2024823379 Country of ref document: EP Effective date: 20260112 |
|
| ENP | Entry into the national phase |
Ref document number: 2024823379 Country of ref document: EP Effective date: 20260112 |
|
| ENP | Entry into the national phase |
Ref document number: 2024823379 Country of ref document: EP Effective date: 20260112 |
|
| ENP | Entry into the national phase |
Ref document number: 2024823379 Country of ref document: EP Effective date: 20260112 |