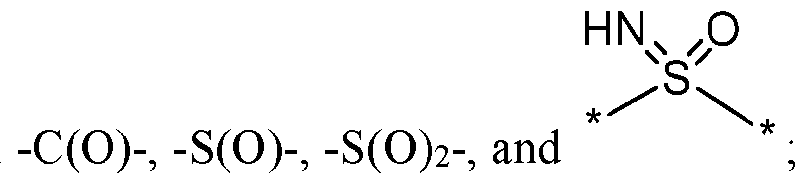WO2024259048A2 - Thiazolopyridin-7(4h)-one wrn inhibitors - Google Patents
Thiazolopyridin-7(4h)-one wrn inhibitors Download PDFInfo
- Publication number
- WO2024259048A2 WO2024259048A2 PCT/US2024/033732 US2024033732W WO2024259048A2 WO 2024259048 A2 WO2024259048 A2 WO 2024259048A2 US 2024033732 W US2024033732 W US 2024033732W WO 2024259048 A2 WO2024259048 A2 WO 2024259048A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- independently selected
- membered
- optionally substituted
- ring
- nitrogen
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D513/00—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for in groups C07D463/00, C07D477/00 or C07D499/00 - C07D507/00
- C07D513/02—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for in groups C07D463/00, C07D477/00 or C07D499/00 - C07D507/00 in which the condensed system contains two hetero rings
- C07D513/04—Ortho-condensed systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D519/00—Heterocyclic compounds containing more than one system of two or more relevant hetero rings condensed among themselves or condensed with a common carbocyclic ring system not provided for in groups C07D453/00 or C07D455/00
Definitions
- the invention provides bicyclic compounds and compositions, the use thereof and methods using the compounds, for inhibiting Werner Syndrome RecQ DNA helicase (WRN) and methods of treating disease using said compounds, in particular the use in treating cancer, and in particular the treatment of cancer characterized as microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR), including colorectal, gastric and endometrial cancer.
- WRN Werner Syndrome RecQ DNA helicase
- MSI-H microsatellite instability-high
- dMMR mismatch repair deficient
- the invention also provides the use of said compounds as research chemicals, intermediate compounds, combinations, processes and formulations.
- Loss of DNA mismatch repair is a common initiating event in cancer development occurring in 10-30% of colorectal, endometrial, ovarian and gastric cancers (Aaltonen, L. A. et al. Clues to the pathogenesis of familial colorectal cancer, Science 260, 812-816 (1993), Bonneville R et al., Landscape of Microsatellite Instability Across 39 Cancer Types. JCO Precis Oncol. 1 : PO.17.00073 (2017)). Cancers that are deficient in mismatch repair (dMMR) have a high mutational burden, and frequent deletion and insertion events in repetitive DNA tracts, a phenotype known as microsatellite instability (MSI).
- MSI microsatellite instability
- WRN helicase is a synthetic lethal target in microsatellite unstable cancers. Nature 568, 551-556 (2019). Kategaya, L., Perumal, S. K., Hager, J. H. & Belmont, L. D. Werner syndrome helicase is required for the survival of cancer cells with microsatellite instability. iScience 13, 488-497 (2019), Lieb, S. et al. Werner syndrome helicase is a selective vulnerability of microsatellite instability-high tumor cells. eLife 8, e43333 (2019)). WRN is synthetically lethal with MSI cancers.
- WRN helicase provides a DNA repair and maintenance function that is essential for cell survival in MSI cancers. Recently, the mechanism of WRN dependence has been elucidated.
- dinucleotide TA repeats are selectively unstable in MSI cells and undergo large scale expansions. These expanded TA repeats form secondary DNA structures that require the WRN helicase for unwinding (van Wietmarschen, N. et al. Repeat expansions confer WRN dependence in microsatellite-unstable cancers. Nature 586, 292-298, 2020).
- WRN or upon WRN helicase inhibition
- expanded TA repeats in MSI cells are subject to nuclease cleavage and chromosome breakage.
- inhibiting the WRN helicase is an attractive strategy for the treatment of MSI-H cancers.
- the invention provides compounds, pharmaceutically acceptable salts thereof, pharmaceutical compositions thereof and combinations thereof, said compounds being inhibitors of Werner Syndrome RecQ DNA Helicase (WRN).
- WRN Werner Syndrome RecQ DNA Helicase
- the invention further provides methods of treating, preventing, or ameliorating a disease or condition, comprising administering to a subject in need thereof an effective amount of a WRN inhibitor.
- the invention also provides compounds, pharmaceutically acceptable salts thereof, pharmaceutical compositions thereof and combinations thereof, said compounds being useful for the treatment of cancer, in particular cancers characterized as microsatellite instability-high (MSI- H) or mismatch repair deficient (dMMR). Also provided are compounds that bind to, and/or inhibit WRN, and are therefore useful as research chemicals, e.g., as a chemical probe, and as tool compounds. Various embodiments of the invention are described herein.
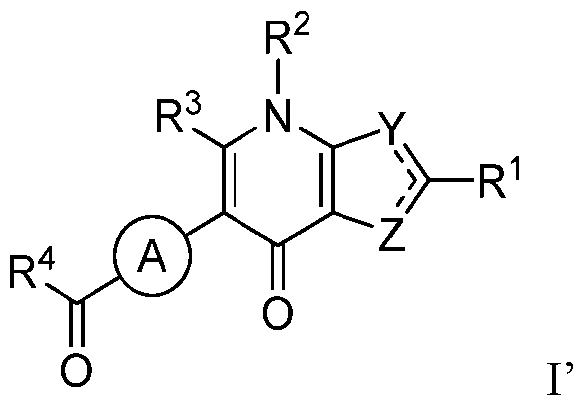
- the disclosure provides a compound of Formula I, or a pharmaceutically acceptable salt thereof: wherein R 1 , R 2 , R 3 , R 4 , Y, Z, L and Ring A are as described and defined herein.
- the invention provides a pharmaceutical composition comprising a compound of Formula I of the present invention and one or more pharmaceutically acceptable carriers.
- the invention provides a combination, in particular a pharmaceutical combination, comprising a compound of Formula I of the present invention and one or more therapeutically active agents.
- the invention provides a compound of Formula I of the present invention for use as a medicament, in particular for the treatment of a disorder or disease which can be treated by WRN inhibition.
- the invention provides a compound of Formula I of the present invention for use in the treatment of cancer, particularly wherein the cancer is characterized as microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR).
- MSI-H microsatellite instability-high
- dMMR mismatch repair deficient
- the invention provides a method of treating a disorder or disease which can be treated by WRN inhibition in a subject, comprising administering to the subject a therapeutically effective amount of a compound of Formula I of the present invention.
- the invention provides a method of treating cancer in a subject, more particularly wherein the cancer is characterized as microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR), comprising administering to the subject a therapeutically effective amount of a compound of Formula I of the present invention.
- MSI-H microsatellite instability-high
- dMMR mismatch repair deficient
- the invention provides the use of a compound of Formula I of the present invention in the manufacture of a medicament for the treatment of a disorder or disease which can be treated by WRN inhibition.
- the invention provides a compound of Formula I of the present invention for use as a research chemical, for example as a chemical probe or as a tool compound.
- the invention provides a solid form, process or intermediate as described herein.
- the disclosure provides a compound of Formula I, or a pharmaceutically acceptable salt thereof:
- Ring A represents: a) a 4-7 membered saturated or partially unsaturated bivalent monocyclic ring system selected from carbocyclylene or heterocyclylene (having 1-4 heteroatoms independently selected from nitrogen, oxygen, and sulfur); or b) a 4-12 membered saturated or partially unsaturated bivalent bicyclic ring system that is fused, bridged, or spirocyclic selected from carbocyclylene or heterocyclylene (having 1- 4 heteroatoms independently selected from nitrogen, oxygen, and sulfur); wherein Ring A is substituted with 0-4 independently selected R A substituents; each of Z and Y is selected from N and S, wherein the 5-membered ring comprising Z and Y is aromatic; wherein - denotes a single or double bond and wherein Y is N and Z is S, or Y is S and Z is
- L is a linker selected from
- R 1 is selected from groups a) to e): a) a 5-6 membered monocyclic heteroaryl (having 1-4 heteroatoms independently selected from nitrogen, oxygen, and sulfur) optionally substituted with 1-3 groups independently selected from halogen, Ci-Cealkyl, haloCi-Cealkyl, Cs-Cecycloalkyl, Ci-Cealkoxy, and C3- Cscycloalkoxy, wherein said 5-6 membered monocyclic heteroaryl is further substituted with 0-3 independently selected R A ; b) a 9-10 membered bicyclic heteroaryl (having 1-4 heteroatoms independently selected from nitrogen, oxygen, and sulfur) optionally substituted with 1 or 2 groups independently selected from C 1 -C 6 aliphatic, C 3 -C 6 cycloalkyl, C 1 -C 6 alkoxy, and C 3 -C 6 cycloalkoxy, wherein said 9-10 membered bicyclic heteroaryl is further substituted
- the invention provides a method of treating a disorder or disease which can be treated by WRN inhibition in a subject, comprising administering to the subject a therapeutically effective amount of a compound of Formula I, or a pharmaceutically acceptable salt thereof, of the present invention.
- the disclosure provides a compound of Formula I’, or a pharmaceutically acceptable salt thereof: I’
- R 4 is selected from one of a), b), a)
- R 4 is a Ring B that is selected from the group consisting of: whe p y g p g ula I’; and wherein: any substituents that are present on Ring B selected from R 4A , R 4B , R 4C , R 4D , R 4E , and R F are each independently selected from hydrogen; -OH, halogen; -CN; C 1 -C 4 alkyl; C 2 - C4alkenyl; C2-C4alkynyl; C1-C4alkoxy; haloC1-C4alkyl; C1-C3alkyl substituted with -OH, -OCH3, or -OCH2CH3; haloC1-C4alkoxy; C3-C6cycloalkyl; C3-C6cycloalkyl
- the invention provides a method of treating a disorder or disease which can be treated by WRN inhibition in a subject, comprising administering to the subject a therapeutically effective amount of a compound of Formula I’, or a pharmaceutically acceptable salt thereof, of the present invention.
- the disclosure provides a compound of Formula I’’, or a pharmaceutically acceptable salt thereof: I’’ wherein Ring A represents: a) a 4-7 membered saturated or partially unsaturated bivalent monocyclic ring system selected from carbocyclylene or heterocyclylene (having 1-4 heteroatoms independently selected from nitrogen, oxygen, and sulfur); or b) a 4-12 membered saturated or partially unsaturated bivalent bicyclic ring system that is fused, bridged, or spirocyclic selected from carbocyclylene or heterocyclylene (having 1- 4 heteroatoms independently selected from nitrogen, oxygen, and sulfur); wherein Ring A is substituted with 0-4 independently selected R A substituents; each of Z and Y is selected from N and S, wherein the 5-membered ring comprising Z and Y is aromatic; wherein ----- denotes a single or double bond and wherein Y is N and Z is S, or Y is S and Z is N; L is a linker selected from
- the invention provides a method of treating a disorder or disease which can be treated by WRN inhibition in a subject, comprising administering to the subject a therapeutically effective amount of a compound of Formula I”, or a pharmaceutically acceptable salt thereof, of the present invention.
- Stereocenters marked with “&1” or “andl” indicate that the compound material has a mixture of R and S- configured stereoisomers with respect to the marked stereocenter and is in the same relative configuration to each other if they share the same label such as “andl” or “&1”.
- aliphatic or “aliphatic group,” as used herein, means a straight-chain (i.e., unbranched) or branched, substituted or unsubstituted hydrocarbon chain that is completely saturated or that contains one or more units of unsaturation, or a monocyclic hydrocarbon or bicyclic hydrocarbon that is completely saturated or that contains one or more units of unsaturation, but which is not aromatic (also referred to herein as “carbocycle,” “cycloaliphatic” or “cycloalkyl”), that has a single point of attachment to the rest of the molecule.
- aliphatic groups contain 1-6 aliphatic carbon atoms.
- aliphatic groups contain 1-5 aliphatic carbon atoms. In other embodiments, aliphatic groups contain 1-4 aliphatic carbon atoms. In still other embodiments, aliphatic groups contain 1-3 aliphatic carbon atoms, and in yet other embodiments, aliphatic groups contain 1-2 aliphatic carbon atoms.
- “cycloaliphatic” (or “carbocycle” or “cycloalkyl”) refers to a monocyclic C3-C6 hydrocarbon that is completely saturated or that contains one or more units of unsaturation, but which is not aromatic, that has a single point of attachment to the rest of the molecule.
- Suitable aliphatic groups include, but are not limited to, linear or branched, substituted or unsubstituted alkyl, alkenyl, alkynyl groups and hybrids thereof such as (cycloalkyl)alkyl, (cycloalkenyl)alkyl or (cycloalkyl)alkenyl.
- bridged bicyclic refers to any bicyclic ring system, i.e., carbocyclic or heterocyclic, saturated or partially unsaturated, having at least one bridge.
- a “bridge” is an unbranched chain of atoms or an atom or a valence bond connecting two bridgeheads, where a “bridgehead” is any skeletal atom of the ring system which is bonded to three or more skeletal atoms (excluding hydrogen).
- a bridged bicyclic group has 5-12 ring members and 0-4 heteroatoms independently selected from nitrogen, oxygen, or sulfur unless otherwise specified, a bridged bicyclic group is optionally substituted with one or more substituents as set forth for aliphatic groups. Additionally or alternatively, any substitutable nitrogen of abridged bicyclic group is optionally substituted.
- alkyl refers to a C1-12 straight or branched saturated aliphatic group. In certain instances, alkyl refers to a Cns straight or branched saturated aliphatic group or a C1-6 straight or branched saturated aliphatic group. The term “lower alkyl” refers to a C1.4 straight or branched alkyl group.
- Exemplary lower alkyl groups are methyl (-CH3), ethyl (-CH2CH3), propyl, isopropyl (also referred to interchangeably herein as 2-propyl, iPr, ‘Pr and i-Pr), butyl, isobutyl (also referred to interchangeably herein as 2-butyl, iBu, ‘Bu and i-Bu) and tert-butyl (also referred to interchangeably herein as 2-methyl-2 -butyl, tBu, ’Bu and t-Bu).
- alkenyl refers to a C2-12 straight or branched partially unsaturated aliphatic group comprising at least one unsaturated carbon carbon double bond. In certain instances, alkenyl refers to a C2-8 or a C2-6 straight or branched partially unsaturated aliphatic group comprising at least one unsaturated carbon carbon double bond.
- lower alkenyl refers to a C2-4 straight or branched partially unsaturated aliphatic group comprising at least one unsaturated carbon carbon double bond.
- alkynyl refers to a C2-12 straight or branched partially unsaturated aliphatic group comprising at least one unsaturated carbon carbon triple bond. In certain instances, alkynyl refers to a C2-8 or a C2-6 straight or branched partially unsaturated aliphatic group comprising at least one unsaturated carbon carbon triple bond.
- lower alkynyl refers to a C2-4 straight or branched partially unsaturated aliphatic group comprising at least one unsaturated carbon carbon triple bond. Exemplary lower alkynyl groups are ethynyl, 1-propynyl, 2-propynyl, 1- butynyl, 2-butynyl, and 3-butynyl.
- haloalkyl refers to a straight or branched alkyl group that is substituted with one or more halogen atoms.
- lower haloalkyl refers to a C1-4 straight or branched alkyl group that is substituted with one or more halogen atoms.
- heteroatom means one or more of oxygen, sulfur, nitrogen, phosphorus, or silicon (including, any oxidized form of nitrogen, sulfur, phosphorus, or silicon; the quaternized form of any basic nitrogen or; a substitutable nitrogen of a heterocyclic ring, for example N (as in 3,4-dihydro-2/f-pyrrolyl), NH (as in pyrrolidinyl) or NR + (as in N-substituted pyrrolidinyl).
- the term “unsaturated,” as used herein, means that a moiety has one or more units of unsaturation.
- the term “bivalent C 1-8 (or C 1-6 i.e., C 1 -C 6 ) saturated or unsaturated, straight or branched, hydrocarbon chain,” refers to bivalent alkylene, alkenylene, and alkynylene chains that are straight or branched as defined herein.
- the term “bivalent,” to describe a cyclic (and noncyclic) group refers to, for example, bivalent carbocyclylene, phenylene, heterocyclylene, and heteroarylene that are bivalent moieties of carbocycles, phenyls, heterocycles, and heteroaryls described herein.
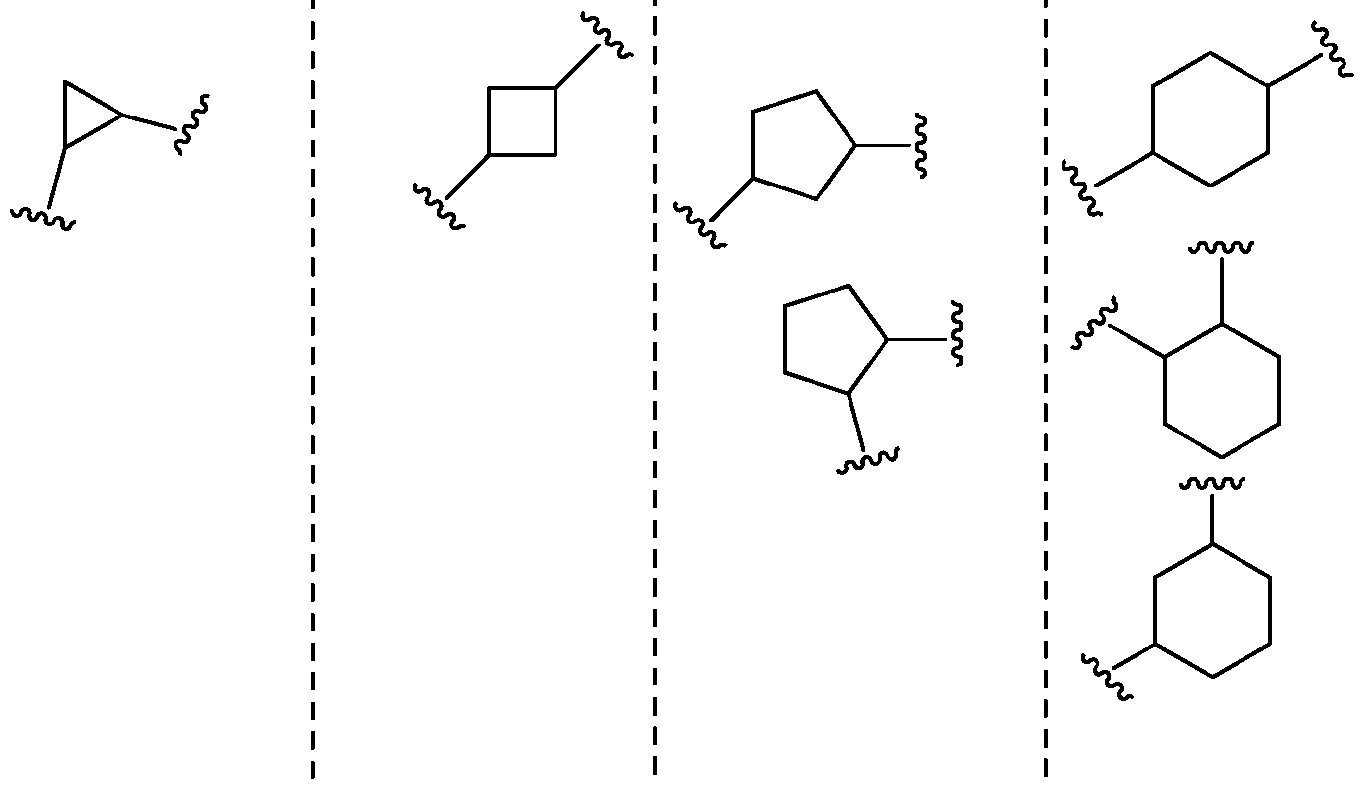
- Non- limiting examples include . [ at is bivalent as described above (i.e., attached at two different points to the rest of the compound).
- Non-limiting examples include cyclopropylene, cyclobutylene, cyclopentylene, or cyclohexylene as shown below.
- a carbocyclylene may be saturated as in the examples shown above or partially unsaturated as in the examples shown below.
- a carbocyclylene may be multi-cyclic, for example, bicyclic or tricyclic. Such multi- cyclic carbocyclylene systems may be saturated or partially unsaturated (while one ring of the bicyclic system may be aromatic it is to be understood that multi-cyclic ring systems that are not in their entirety aromatic may also fall under the definition of carbocyclylene).
- the rings may form bridged, fused, or spiro systems. Non-limiting examples are shown below. spirocyclic bicyclic carbocyclylenes fused bicyclic carbocyclylenes bridged bicyclic carbocyclylenes
- Heterocyclylene refers to a heterocyclic or heterocyclyl moiety that is bivalent as described above (i.e., attached at two different points to the rest of the compound) and may also be saturated or partially unsaturated. Non-limiting examples include those shown below. Heterocyclylene is understood to include bicyclic heterocyclylene systems. Non-limiting examples of bicyclic heterocyclylene moieties are also shown below and said bicyclic systems may be spirocyclic, fused, or bridged and may be saturated or partially unsaturated.
- Phenylene refers to a phenyl moiety that is bivalent as described above
- “Arylene” as used herein refers to an mono or multi-cyclic aryl (i.e., phenyl or a multi- cyclic aryl) moiety that is bivalent as described above (i.e., attached at two different points to the rest of the compound), wherein the arylene group contains no heteroatoms. Examples are shown below.
- “Heteroarylene,” as used herein refers to a mono or multi-cyclic aryl ring system that contains at least one heteroatom wherein the ring system is bivalent as described above (i.e., attached at two different points to the rest of the compound). Examples are shown below.
- alkylene refers to a bivalent alkyl group.
- An “alkylene chain” is a polymethylene group, i.e., -(CH2) n - wherein n is a positive integer, preferably from 1 to 6, from 1 to 4, from 1 to 3, from 1 to 2, or from 2 to 3.
- a substituted alkylene chain is a polymethylene group in which one or more methylene hydrogen atoms are replaced with a substituent. Suitable substituents include those described below for a substituted aliphatic group.
- Carbocyclyl (or heterocyclyl, aryl, phenyl, or heteroaryl) fused to” another phenyl, aryl, heteroaryl, carbocyclyl, or heterocyclyl, for example, a “phenyl or pyridyl” as used herein, may be referred to as “partially unsaturated” without said “carbocyclyl (or heterocyclyl, aryl, phenyl, or heteroaryl) fused to” the other ring requiring further unsaturation besides the carbon carbon bond which it shares with the ring to which it is fused (i.e., the “phenyl or pyridyl”). This is illustrated below. partially unsaturated cyclopentyl fused to phenyl, i.e., "cyclopentyl fused to phenyl"
- a further example below shows a carbocyclyl moiety fused to a Ring B as defined in the embodiments herein.
- Said carbocyclyl does not explicitly require a descriptor of “partially unsaturated” to describe said carbocyclyl because it shares two carbons with the aromatic pyridine to which it is fused.
- Such language is used herein to describe such systems, for example, “R 4A and R 4B , along with their intervening atoms, join to form 4-7-membered carbocyclyl that is fused to Ring B” as shown in the image below.
- Ring B may refer to a monocyclic ring (i.e., the pyridine shown below and its substituents which do not form a fused ring), without any further fused rings created by its substituents (i.e., R 4A and R 4B ). Any further fused ring created by the substituents of Ring B is described as being “fused to Ring B.” Likewise, R 4A and R 4B , along with their intervening atoms, join to form 4-7-membered carbocyclyl or heterocyclyl (having 1-4 heteroatoms independently selected from nitrogen, oxygen, and sulfur) that is fused to Ring B (not pictured), is subject to the same interpretation. embered car ocyclyl that is
- alkenylene refers to a bivalent alkenyl group.
- a substituted alkenylene chain is a polymethylene group containing at least one double bond in which one or more hydrogen atoms are replaced with a substituent. Suitable substituents include those described below for a substituted aliphatic group.
- halogen means F, Cl, Br, or I.
- aryl used alone or as part of a larger moiety as in “aralkyl,” “aralkoxy,” or
- aryloxyalkyl refers to monocyclic or bicyclic ring systems having a total of five to fourteen ring members, wherein at least one ring in the system is aromatic and wherein each ring in the system contains 3 to 7 ring members.
- aryl may be used interchangeably with the term “aryl ring.”
- aryl refers to an aromatic ring system which includes, but not limited to, phenyl, biphenyl, naphthyl, anthracyl and the like, which may bear one or more substituents.
- aryl is a group in which an aromatic ring is fused to one or more non-aromatic rings, such as indanyl, phthalimidyl, naphthimidyl, phenanthridinyl, or tetrahydronaphthyl, and the like.
- heteroaryl and “heteroar-,” used alone or as part of a larger moiety, e.g., “heteroaralkyl,” or “heteroaralkoxy,” refer to groups having 5 to 10 ring atoms, preferably 5, 6, 9 or 10 ring atoms; having 6, 10, or 14 n electrons shared in a cyclic array; and having, in addition to carbon atoms, from one to five heteroatoms.
- heteroatom refers to nitrogen, oxygen, or sulfur, and includes any oxidized form of nitrogen or sulfur, and any quatemized form of a basic nitrogen.
- Heteroaryl groups include, without limitation, pyrazinyl, pyrazolyl, pyridazinyl, pyridyl, pyrimidinyl, triazinyl, thienyl, furanyl, pyrrolyl, imidazolyl, pyrazolyl, triazolyl (i.e., 1,2,3-triazolyl), 1,2,4-triazolyl, 1,2,5-triazolyl, 1,3,4-triazolyl, tetrazolyl, oxazolyl, isoxazolyl, oxadiazolyl, 1,2,3-oxadiazolyl, 1,2,4-oxadiazolyl, 1,2,5-oxadiazolyl, 1,3,4-oxadiazolyl, thiazolyl, isothiazolyl, thiadiazolyl, indolizinyl, purinyl, naphthyridinyl, and pteridinyl.
- heteroaryl and “heteroar-,” as used herein, also include groups in which a heteroaromatic ring is fused to one or more aryl, cycloaliphatic, or heterocyclyl rings, where unless otherwise specified, the radical or point of attachment is on the heteroaromatic ring or on one of the rings to which the heteroaromatic ring is fused.
- Nonlimiting examples include indolyl, isoindolyl, benzothienyl, benzofuranyl, dibenzofuranyl, indazolyl, indolizinyl, isoindolin-l-only, l,2-dihydro-3H- pyrrolo[3, 4-c]pyri din-3 -onyl, 2,3-dihydro-lH-pyrrolo[3,4-c]pyridin-l-onyl, imidazo[l,2- a]pyridyl, imidazo[l,5-a]pyridyl, pyrazolo[l,5-a]pyridyl, pyrrolo[l,2-b]pyridazinyl, pyrrolo[l,2- a]pyrimidinyl, imidazo[l,2-b]pyridazinyl, imidazo[l,2-a]pyrimidinyl, benzimidazolyl, benzthiazolyl,
- heteroaryl group may be mono- or bicyclic.
- heteroaryl may be used interchangeably with the terms “heteroaryl ring,” “heteroaryl group,” or “heteroaromatic,” any of which terms include rings that are optionally substituted.
- heteroarylkyl refers to an alkyl group substituted by a heteroaryl, wherein the alkyl and heteroaryl portions independently are optionally substituted.
- heterocycle refers to the substituent * 3 .
- heterocyclyl refers to the substituent * 3 .
- heterocyclic radical refers to the substituent * 3 .
- heterocyclic ring refers to a stable 5- to 7-membered monocyclic or 7-10-membered bicyclic heterocyclic moiety that is either saturated or partially unsaturated, and having, in addition to carbon atoms, one or more, preferably one to four, heteroatoms, as defined above.
- Said 7-10-membered bicyclic heterocyclic moiety that is partially unsaturated may include an aryl or heteroaryl ring fused to a non-aromatic ring.
- said 7-10-membered bicyclic heterocyclic moiety may include a bicyclic heterocyclyl as shown below:
- the term “nitrogen” includes a substituted nitrogen.
- the nitrogen in a saturated or partially unsaturated ring having 0-3 heteroatoms selected from oxygen, sulfur or nitrogen, the nitrogen may be N (as in 3,4-di hydro-2// pyrrol y I), NH (as in pyrrol i di nyl), or + NR (as in //-substituted pyrrol idinyl).
- a heterocyclic ring can be attached to its pendant group at any heteroatom or carbon atom that results in a stable structure and any of the ring atoms can be optionally substituted.
- saturated or partially unsaturated heterocyclic radicals include, without limitation, oxetanyl, azetidinyl, tetrahydrofuranyl, tetrahydrothiophenyl pyrrolidinyl, piperidinyl, pyrrolinyl, tetrahydroquinolinyl, tetrahydroisoquinolinyl, decahydroquinolinyl, oxazolidinyl, piperazinyl, dioxanyl, dioxolanyl, diazepinyl, oxazepinyl, thiazepinyl, morpholinyl, 2-oxa-6- azaspiro[3.3]heptane, and quinuclidinyl.
- heterocycle used interchangeably herein, and also include groups in which a heterocyclyl ring is fused to one or more aryl, heteroaryl, or cycloaliphatic rings, such as indolinyl, 3//-indolyl, chromanyl, phenanthridinyl, or tetrahydroquinolinyl.
- a heterocyclyl group may be mono- or bicyclic.
- heterocyclyl alkyl refers to an alkyl group substituted by a heterocyclyl, wherein the alkyl and heterocyclyl portions independently are optionally substituted.
- Heterocyclyloxy refers to an -OR group wherein the R is a heterocyclyl. Nonlimiting examples are shown below.
- partially unsaturated refers to a ring moiety that includes at least one double or triple bond.
- partially unsaturated is intended to encompass rings having multiple sites of unsaturation, but is not intended to include aryl or heteroaryl moieties, as herein defined.
- compounds of the invention may contain “optionally substituted” moieties.
- substituted whether preceded by the term “optionally” or not, means that one or more hydrogens of the designated moiety are replaced with a suitable substituent.
- an “optionally substituted” group may have a suitable substituent at each substitutable position of the group, and when more than one position in any given structure may be substituted with more than one substituent selected from a specified group, the substituent may be either the same or different at every position.
- Combinations of substituents envisioned by this invention are preferably those that result in the formation of stable or chemically feasible compounds.
- stable refers to compounds that are not substantially altered when subjected to conditions to allow for their production, detection, and, in certain embodiments, their recovery, purification, and use for one or more of the purposes disclosed herein.
- Suitable monovalent substituents on R° are independently halogen, -(CH 2 )o 2 R*, -(haloR*), -(CH 2 ) 0 2 OH, -(CH 2 ) O 2 OR’, -(CH 2 )O 2 CH(OR’) 2 ; -O(haloR’), -CN, -N 3 , -(CH 2 ) 0 2 C(O)R’, -(CH 2 ) O 2 C(O)OH, -(CH 2 ) O 2 C(O)OR*, -(CH 2 ) O 2 SR’, -(CH 2 ) O 2 SH, -(CH 2 ) O 2 NH 2 , - (CH 2 )O- 2 NHR*, -(CH 2 ) O 2 NR* 2 , -NO 2 , -SiR* 3
- Suitable divalent substituents that are bound to vicinal substitutable carbons of an “optionally substituted” group include: -O(CR* 2 )2-3O- wherein each independent occurrence of R* is selected from hydrogen, C1-6 aliphatic which may be substituted as defined below, or an unsubstituted 5-6-membered saturated, partially unsaturated, or aryl ring having 0- 4 heteroatoms independently selected from nitrogen, oxygen, or sulfur.
- Suitable substituents on the aliphatic group of R* include halogen, -R*, -(haloR*), -OH, -OR*, -O(haloR’), -CN, -C(O)OH, -C(O)OR*, -NH 2 , -NHR*, -NR* 2 , or -NO2, wherein each R* is unsubstituted or where preceded by “halo” is substituted only with one or more halogens, and is independently C 1-4 aliphatic, -CH 2 Ph, -0(CH2)o iPh, or a 5-6- membered saturated, partially unsaturated, or aryl ring having 0-4 heteroatoms independently selected from nitrogen, oxygen, or sulfur.
- Suitable substituents on a substitutable nitrogen of an “optionally substituted” group include -R ⁇ -NR ⁇ , -C(O)R f , -C(O)OR t , -C(O)C(O)R f , -C(O)CH 2 C(O)R t , -S(O) 2 R t , -S(O) 2 NR t 2, -C ⁇ NR ⁇ , -C(NH)NR t 2 , or -N(R t )S(O) 2 R t ; wherein each R ; is independently hydrogen, Ci-6 aliphatic which may be substituted as defined below, unsubstituted -OPh, or an unsubstituted 5-6-membered saturated, partially unsaturated, or aryl ring having 0-4 heteroatoms independently selected from nitrogen, oxygen, or sulfur, or, notwithstanding the definition above, two independent occurrences of R’ 1 ', taken together with
- Suitable substituents on the aliphatic group of R are independently halogen, -R*, -(haloR*), -OH, -OR*, -O(haloR*), -CN, -C(O)OH, -C(O)OR*, -NH 2 , -NHR*, -NR* 2 , or -NO 2 , wherein each R* is unsubstituted or where preceded by “halo” is substituted only with one or more halogens, and is independently C 1-4 aliphatic, -CH 2 Ph, -0(CH 2 )o iPh, or a 5-6- membered saturated, partially unsaturated, or aryl ring having 0-4 heteroatoms independently selected from nitrogen, oxygen, or sulfur.
- the term “pharmaceutically acceptable salt” refers to those salts which are, within the scope of sound medical judgment, suitable for use in contact with the tissues of humans and lower animals without undue toxicity, irritation, allergic response and the like, and are commensurate with a reasonable benefit/risk ratio.
- Pharmaceutically acceptable salts are well known in the art. For example, S. M. Berge et al., describe pharmaceutically acceptable salts in detail in J. Pharmaceutical Sciences, 1977, 66, 1-19.
- Pharmaceutically acceptable salts of the compounds of this invention include those derived from suitable inorganic and organic acids and bases.
- Examples of pharmaceutically acceptable, nontoxic acid addition salts are salts of an amino group formed with inorganic acids such as hydrochloric acid, hydrobromic acid, phosphoric acid, sulfuric acid and perchloric acid or with organic acids such as acetic acid, oxalic acid, maleic acid, tartaric acid, citric acid, succinic acid or malonic acid or by using other methods used in the art such as ion exchange.
- inorganic acids such as hydrochloric acid, hydrobromic acid, phosphoric acid, sulfuric acid and perchloric acid
- organic acids such as acetic acid, oxalic acid, maleic acid, tartaric acid, citric acid, succinic acid or malonic acid or by using other methods used in the art such as ion exchange.
- salts include adipate, alginate, ascorbate, aspartate, benzenesulfonate, benzoate, bisulfate, borate, butyrate, camphorate, camphorsulfonate, citrate, cyclopentanepropionate, digluconate, dodecyl sulfate, ethanesulfonate, formate, fumarate, glucoheptonate, glycerophosphate, gluconate, hemisulfate, heptanoate, hexanoate, hydroiodide, 2- hydroxy-ethanesulfonate, lactobionate, lactate, laurate, lauryl sulfate, malate, maleate, malonate, methanesulfonate, 2-naphthalenesulfonate, nicotinate, nitrate, oleate, oxalate, palmitate, pamoate, pect
- Salts derived from appropriate bases include alkali metal, alkaline earth metal, ammonium and N (C, 4alkyl)4 salts.
- Representative alkali or alkaline earth metal salts include sodium, lithium, potassium, calcium, magnesium, and the like.
- Further pharmaceutically acceptable salts include, when appropriate, nontoxic ammonium, quaternary ammonium, and amine cations formed using counterions such as halide, hydroxide, carboxylate, sulfate, phosphate, nitrate, loweralkyl sulfonate and aryl sulfonate.
- structures depicted herein are also meant to include all isomeric (e.g., enantiomeric, diastereomeric, and geometric (or conformational)) forms of the structure; for example, the R and S configurations for each asymmetric center, Z and E double bond isomers, Z and E conformational isomers and R a (or M) and S a (or P) atropisomers. Therefore, single stereochemical isomers as well as enantiomeric, diastereomeric, and geometric (or conformational) mixtures of the present compounds are within the scope of the invention. Unless otherwise stated, all tautomeric forms of the compounds of the invention are within the scope of the invention.
- structures depicted herein are also meant to include compounds that differ only in the presence of one or more isotopically enriched atoms.
- compounds having the present structures including the replacement of hydrogen by deuterium or tritium, or the replacement of a carbon by a 13 C- or 14 C-enriched carbon are within the scope of this invention.
- Such compounds are useful, for example, as analytical tools, as probes in biological assays, or as therapeutic agents in accordance with the present invention.
- Ring A of a provided compound may be substituted with one or more deuterium atoms.
- the disclosure provides a compound of Formula I, or a pharmaceutically acceptable salt thereof: wherein Ring A represents: a) a 4-7 membered saturated or partially unsaturated bivalent monocyclic ring system selected from carbocyclylene and heterocyclylene (having 1-4 heteroatoms independently selected from nitrogen, oxygen, and sulfur); or b) a 4-12 membered saturated or partially unsaturated bivalent bicyclic ring system that is fused, bridged, or spirocyclic selected from carbocyclylene or heterocyclylene (having 1- 4 heteroatoms independently selected from nitrogen, oxygen, and sulfur); wherein Ring A is substituted with 0-4 independently selected R A substituents; each of Z and Y is selected from N and S, wherein the 5-membered ring comprising Z and Y is aromatic; wherein ----- denotes a single or double bond and wherein Y is N and Z is S, or Y is S and Z is N; -L- is a linker selected from ;
- the disclosure provides a compound of Formula I x , or a pharmaceutically acceptable salt thereof: wherein Ring A represents: a) a 4-7 membered saturated or partially unsaturated bivalent monocyclic ring system selected from carbocyclylene and heterocyclylene (having 1-4 heteroatoms independently selected from nitrogen, oxygen, and sulfur); or b) a 4-12 membered saturated or partially unsaturated bivalent bicyclic ring system that is fused, bridged, or spirocyclic selected from carbocyclylene or heterocyclylene (having 1- 4 heteroatoms independently selected from nitrogen, oxygen, and sulfur); wherein Ring A is substituted with 0-4 independently selected R A substituents; each of Z and Y is selected from N and S, wherein if Y is S, then Z is N or wherein if Y is N, then Z is S and wherein the 5-membered ring comprising Z and Y is aromatic; R 1 is selected from groups a) to e): a)
- the disclosure provides a compound of Formula F, or a pharmaceutically acceptable salt thereof: wherein Ring A represents: b) a 4-7 membered saturated or partially unsaturated bivalent monocyclic ring system selected from carbocyclylene and heterocyclylene (having 1-4 heteroatoms independently selected from nitrogen, oxygen, and sulfur); or c) a 4-12 membered saturated or partially unsaturated bivalent bicyclic ring system that is fused, bridged, or spirocyclic selected from carbocyclylene or heterocyclylene (having 1- 4 heteroatoms independently selected from nitrogen, oxygen, and sulfur); wherein Ring A is substituted with 0-4 independently selected R A substituents; each of Z and Y is selected from N and S, wherein if Y is S, then Z is N or wherein if Y is N, then Z is S and wherein the 5-membered ring comprising Z and Y is aromatic;
- R 1 is selected from groups a) to e): a) a 5-6 membered monocyclic heteroaryl (having 1 -4 heteroatoms independently selected from nitrogen, oxygen, and sulfur) optionally substituted with 1-3 groups independently selected from halogen, C 1 -C 6 alkyl, haloC 1 -C 6 alkyl, C 3 -C 6 cycloalkyl, C 1 -C 6 alkoxy, and C 3 - C6cycloalkoxy, wherein said 5-6 membered monocyclic heteroaryl is further substituted with 0-3 independently selected R A ; b) a 9-10 membered bicyclic heteroaryl (having 1-4 heteroatoms independently selected from nitrogen, oxygen, and sulfur) optionally substituted with 1 or 2 groups independently selected from C1-C6aliphatic, C3-C6cycloalkyl, C1-C6alkoxy, and C3-C6cycloalkoxy, wherein said 9-10 membered bicyclic heteroaryl is further
- the disclosure provides a compound of Formula I”, or a pharmaceutically acceptable salt thereof: wherein Ring A represents: a) a 4-7 membered saturated or partially unsaturated bivalent monocyclic ring system selected from carbocyclylene or heterocyclylene (having 1-4 heteroatoms independently selected from nitrogen, oxygen, and sulfur); or b) a 4-12 membered saturated or partially unsaturated bivalent bicyclic ring system that is fused, bridged, or spirocyclic selected from carbocyclylene or heterocyclylene (having 1- 4 heteroatoms independently selected from nitrogen, oxygen, and sulfur); wherein Ring A is substituted with 0-4 independently selected R A substituents; each of Z and Y is selected from N and S, wherein the 5-membered ring comprising Z and Y is aromatic; wherein ----- denotes a single or double bond and wherein Y is N and Z is S, or Y is S and Z is N; L is a linker selected from ; R 1 is
- the present invention provides a compound of Formula I, I’, I x , I y , or I’, wherein R 4 is selected from one of a), b), and c): a) R 4 is a Ring B that is selected from the group consisting of: wher and wherein: any substituents that are present on Ring B selected from R 4A , R 4B , R 4C , R 4D , R 4E , and R 4F are each independently selected from hydrogen; halogen; -OH; -CN; C 1 -C 4 alkyl; C 2 - C4alkenyl; C2-C4alkynyl; C1-C4alkoxy; haloC1-C4alkyl; C1-C3alkyl substituted with -OH, -OCH3, or -OCH2CH3; halo
- Ring A is a) a 4-7 membered saturated or partially unsaturated bivalent monocyclic ring system selected from carbocyclylene or heterocyclylene (having 1-4 heteroatoms independently selected from nitrogen, oxygen, and sulfur); or b) a 4-12 membered saturated or partially unsaturated bivalent bicyclic ring system that is fused, bridged, or spirocyclic selected from carbocyclylene or heterocyclylene (having 1- 4 heteroatoms independently selected from nitrogen, oxygen, and sulfur), wherein Ring A is substituted with 0-4 independently selected R A substituents.
- Ring A is a 4-7 membered saturated or partially unsaturated bivalent monocyclic ring system selected from carbocyclylene and heterocyclylene (having 1-4 heteroatoms independently selected from nitrogen, oxygen, and sulfur), wherein Ring A is substituted with 0-4 independently selected R A substituents.
- Ring A is a 4- 7 membered saturated or partially unsaturated bivalent monocyclic carbocyclylene, wherein Ring A is substituted with 0-4 independently selected R A substituents.
- Ring A is a 4-7 membered saturated or partially unsaturated bivalent monocyclic heterocyclylene (having 1- 4 heteroatoms independently selected from nitrogen, oxygen, and sulfur), wherein Ring A is substituted with 0-4 independently selected R A substituents.
- Ring A is a 4-12 membered saturated or partially unsaturated bivalent bicyclic ring system that is fused, bridged, or spirocyclic selected from carbocyclylene and heterocyclylene (having 1-4 heteroatoms independently selected from nitrogen, oxygen, and sulfur), wherein Ring A is substituted with 0-4 independently selected R A substituents.
- Ring A is a 4-12 membered saturated or partially unsaturated bivalent bicyclic ring system that is fused, bridged, or spirocyclic and is a carbocyclylene, wherein Ring A is substituted with 0-4 independently selected R A substituents.
- Ring A is a 4-12 membered saturated or partially unsaturated bivalent bicyclic ring system that is fused, bridged, or spirocyclic and is a heterocyclylene (having 1-4 heteroatoms independently selected from nitrogen, oxygen, and sulfur), wherein Ring A is substituted with 0-4 independently selected R A substituents.
- Ring A is a 4-12 membered saturated or partially unsaturated bivalent bicyclic ring system comprising 2 fused rings. In some embodiments, Ring A is a 4-12 membered saturated or partially unsaturated bivalent bicyclic ring system comprising a spirocyclic ring system. In some embodiments, Ring A is a 4-12 membered saturated or partially unsaturated bivalent bicyclic ring system comprising a bridged ring system.
- Ring A is selected from and wherein Ring A is substituted with 0-4 independently selected R A substituents.
- Ring A is . [0078] In some embodiments, Ring . [0079] In some embodiments, Ring . [0080] In some embodiments, Ring cted from one of the substituents of Table 1, Table 1a or Table 1b.
- R 1 is selected from groups a) to e): a) a 5-6 membered monocyclic heteroaryl (having 1-4 heteroatoms independently selected from nitrogen, oxygen, and sulfur) optionally substituted with 1-3 groups independently selected from halogen, C1-C6alkyl, haloC1-C6alkyl, C3-C6cycloalkyl, C1-C6alkoxy, and C3- C 6 cycloalkoxy, wherein said 5-6 membered monocyclic heteroaryl is further substituted with 0-3 independently selected R A ; b) a 9-10 membered bicyclic heteroaryl (having 1-4 heteroatoms independently selected from nitrogen, oxygen, and sulfur) optionally substituted with 1 or 2 groups independently selected from C 1 -C 6 aliphatic, C 3 -C 6 cycloalkyl, C 1 -C 6 alkoxy, and C 3 -C 6 cycloalkoxy, wherein said 9-10 membere
- R 1 is a 5-6 monocyclic membered heteroaryl (having 1-4 heteroatoms independently selected from nitrogen, oxygen, and sulfur) optionally substituted with 1 - 3 groups independently selected from C 1 -C 6 alkyl, C 1 -C 6 alkoxy, C 3 -C 6 cycloalkyl, and C 3 - C6cycloalkoxy, wherein said 5-6 membered monocyclic heteroaryl is further substituted with 0-2 independently selected R A .
- R 1 is a 4- or 6-membered saturated or partially unsaturated monocyclic heterocyclyl (having 1-2 heteroatoms independently selected from nitrogen, oxygen, and sulfur), said heterocyclyl substituted with 0-2 groups independently selected from halogen, oxo, -NR2, optionally substituted C1-4 aliphatic, -OR, azetidinyl optionally substituted with 1 or 2 independently selected halogen, and pyrrolidinyl optionally substituted with 1 or 2 independently selected halogen.
- R 1 is a 6-8 membered saturated or partially unsaturated bridged bicyclic heterocyclyl (having 1-4 heteroatoms independently selected from nitrogen, oxygen, and sulfur), said heterocyclyl substituted with 0-2 groups independently selected from halogen, oxo, -NR 2 , optionally substituted C 1 - 4 aliphatic, -OR, azetidinyl optionally substituted with 1 or 2 independently selected halogen, and pyrrolidinyl optionally substituted with 1 or 2 independently selected halogen.
- R 1 is a 3-7 membered optionally substituted carbocyclyl.
- R 1 is an optionally substituted C 2 -C 4 alkenyl.
- R 1 is cyclopropyl substituted C2-C4alkenyl. In some embodiments, R 1 is methyl substituted C2alkenyl. [0083] In some embodiments, R 1 is a 6-membered partially unsaturated heterocyclyl (having 1 oxygen atom).
- R 1 is a 6-membered heteroaryl (having 1 nitrogen atom), said heteroaryl may be optionally substituted with 1 or 2 groups independently selected from C1- C 6 alkyl, C 1 -C 6 alkoxy, C 3 -C 6 cycloalkyl, and C 3 -C 6 cycloalkoxy, wherein said heteroaryl is further substituted with 0-1 R A , wherein R A is an optionally substituted C 1-6 aliphatic group.
- R 1 is a 6-membered heteroaryl (having 2 nitrogen atoms), said heteroaryl may be optionally substituted with 1 or 2 groups independently selected from C1-C6alkyl, C1-C6alkoxy, C 3 -C 6 cycloalkyl, and C 3 -C 6 cycloalkoxy, wherein said heteroaryl is further substituted with 0-1 R A , wherein R A is an optionally substituted C1-6 aliphatic group.
- R 1 is a bicyclic 9-10 membered heteroaryl (having 1-4 heteroatoms independently selected from nitrogen, oxygen, and sulfur) optionally substituted with 1 or 2 groups independently selected from C1-C6alkyl, C3-C6cycloalkyl, C1-C6alkoxy, and C3- C6cycloalkoxy, wherein said bicyclic 9-10 membered heteroaryl is further substituted with 0-3 independently selected R A .
- R 1 is a 5-membered heteroaryl (having 1-3 heteroatoms independently selected from nitrogen, oxygen, and sulfur) optionally substituted with 1 or 2 groups independently selected from C1-C6alkyl, C1-C6alkoxy, C3-C6cycloalkyl, and C3-C6cycloalkoxy, wherein said 5-membered heteroaryl is optionally further substituted with 0-2 independently selected R A .
- R 1 is a 5-membered heteroaryl (having 1-3 heteroatoms independently selected from nitrogen, oxygen, and sulfur) optionally substituted with 1 or 2 groups independently selected from C 1 -C 6 alkyl, C 1 -C 6 alkoxy, C 3 -C 6 cycloalkyl, and C 3 -C 6 cycloalkoxy.
- R 1 is a 5-membered heteroaryl (having 2 nitrogen atoms) substituted with 1 or 2 groups independently selected from C1-C6alkoxy, C3-C6cycloalkyl, and C3-C6cycloalkoxy, wherein said 5-membered heteroaryl is optionally further substituted with 0-1 R A , wherein R A is hydroxyl substituted C 1 -C 4 alkyl.
- R 1 is a 5-6 membered heteroaryl (having 1-4 heteroatoms is independently selected from nitrogen, oxygen, and sulfur) optionally substituted with one group of C 1 -C 6 alkoxy or C 3 -C 6 cycloalkyl, wherein said 5-6 membered heteroaryl is optionally further substituted with 0-3 independently selected R A .
- R 1 is pyridyl substituted with C1-C4alkoxy and further substituted with 0-2 R A substituents.
- R 1 is 5-membered heteroaryl (having 1 heteroatom independently selected from nitrogen, oxygen, and sulfur, and 0 or 1 additional ring nitrogen atoms), wherein said 5-membered heteroaryl is optionally substituted with Ci-Cealkyl, or C3- Cscycloalkyl and further substituted with 0-2 R A substituents.
- R 1 is a) a 5-6 membered saturated or partially unsaturated heterocyclyl (having 1-2 heteroatoms independently selected from nitrogen, oxygen, and sulfur), said heterocyclyl substituted with 0-2 groups independently selected from halogen, oxo, -NR2, optionally substituted C1-4 aliphatic, -OR, azetidinyl optionally substituted with 1 or 2 independently selected halogen, and pyrrolidinyl optionally substituted with 1 or 2 independently selected halogen; or b) a 6-8 membered saturated or partially unsaturated bridged bicyclic heterocyclyl (having 1- 4 heteroatoms independently selected from nitrogen, oxygen, and sulfur), said heterocyclyl substituted with 0-2 groups independently selected from halogen, oxo, -NR2, optionally substituted C1-4 aliphatic, -OR, azetidinyl optionally substituted with 1 or 2 independently selected halogen, and pyrrolidinyl optional
- R 1 is a 5-6 membered saturated or partially unsaturated heterocyclyl (having 1-2 heteroatoms independently selected from nitrogen, oxygen, and sulfur), said heterocyclyl substituted with 0-2 groups independently selected from halogen, oxo, -NR2, optionally substituted C1-4 aliphatic, -OR, azetidinyl optionally substituted with 1 or 2 independently selected halogen, and pyrrolidinyl optionally substituted with 1 or 2 independently selected halogen.
- R 1 is a 5-6 membered heteroaryl (having 1-4 heteroatoms independently selected from nitrogen, oxygen, and sulfur) substituted with 1 group selected from Ci-C6alkoxy and C3-C6cycloalkyl, wherein said 5-6 membered heteroaryl is further substituted with 0-3 independently selected R A .
- R 1 is
- R 1 is
- R 1 is , Table 1a or Table 1b.
- R A is independently selected at each occurrence from the group consisting of optionally substituted phenyl, optionally substituted 5-6 membered heteroaryl (having 1-4 heteroatoms independently selected from nitrogen, oxygen, and sulfur), optionally substituted 4-7 membered saturated or partially unsaturated heterocyclyl (having 1-2 heteroatoms independently selected from nitrogen, oxygen, and sulfur), halogen, optionally substituted C 1 -C 6 aliphatic, hydroxy-C 1 -C 6 alkyl, haloC 1 -C 6 alkyl, an optionally substituted C 3 - C 6 cycloalkyl, haloC 3 -C 6 cycloalkyl, C 1 -C 6 alkoxy, haloC 1 -C 6 alkoxy, C 3 -C 6 cycloalkoxy, haloC 3 - C6cycloalkoxy, C1-
- R A is independently selected at each occurrence from the group consisting of halogen, -OR, or an optionally substituted C 1-6 aliphatic group. In some embodiments, R A is independently selected at each occurrence from a halogen. In some embodiments, R A is independently selected at each occurrence from an -OR. In some embodiments R A is an optionally substituted 5-6 membered heteroaryl ring (having 1-4 heteroatoms independently selected from nitrogen, oxygen, and sulfur). In some embodiments, R A is an optionally substituted C3- C6cycloalkyl. In some embodiments, R A is independently selected at each occurrence from an optionally substituted C 1-6 aliphatic group.
- R A is as selected from one of the substituents of Table 1, Table 1a or Table 1b.
- R 2 is C(R B )2C(O)N(R)R 2A .
- R 2 is C(R B ) 2 C(R B ) 2 C(O)N(R)R 2A .
- R 2 is C(R B ) 2 C(R B ) 2 N(R)C(O)N(R)R 2A .
- R 2 is C(R B )2C(R B )2N(R)C(O)R 2A .
- R 2 is CH2C(O)N(H)R 2A . In some embodiments, R 2 is CH2CH2C(O)N(H)R 2A . In some embodiments, R 2 is CH 2 CH 2 N(R)C(O)N(R)R 2A . In some embodiments, R 2 is CH 2 CH 2 N(H)C(O)R 2A .
- R 2 is C(R B )2C(O)N(H)R 2A , wherein R 2A is phenyl or bicyclo[1.1.1]pentyl optionally substituted with 1, 2, or 3 substituents independently selected from halogen, C1-C4alkyl, or haloC1- C 4 alkyl.
- R 2 is C(R B ) 2 C(O)N(H)R 2A , wherein R 2A is phenyl optionally substituted with 1, 2, or 3 substituents independently selected from halogen, C 1 -C 4 alkyl, or haloC 1 - C4alkyl.
- R 2 is C(R B )2C(O)N(H)R 2A , wherein R 2A is bicyclo[1.1.1]pentyl optionally substituted with 1, 2, or 3 substituents independently selected from halogen, C1-C4alkyl, or haloC 1 -C 4 alkyl. [00102] In some embodiments, R 2 is
- R 2 is
- R 2 is as selected from one of the substituents of Table 1, Table la or Table lb.
- R B is independently selected at each occurrence from hydrogen, -CH3, and -CH2CH3, or two R B taken together with the carbon to which they are attached form a cyclopropyl ring.
- R B is independently selected at each occurrence from hydrogen, -CH3, and -CH2CH3.
- R B is hydrogen.
- two R B taken together with the carbon to which they are attached form a cyclopropyl ring.
- R B is as selected from one of the substituents of Table 1, Table la or Table lb.
- R 2A is phenyl, pyridyl, cubanyl, a saturated or partially unsaturated 4-8 membered monocyclic ring, a saturated or partially unsaturated bridged, fused, or spirocyclic 5-, 6-, 7-, 8-, 9-, 10-, 11-, or 12-membered ring, wherein said saturated or partially unsaturated monocyclic ring, or saturated or partially unsaturated bridged, fused, or spirocyclic ring contains 0, 1, 2, 3, or 4 heteroatoms independently selected from nitrogen, oxygen, and sulfur; and wherein said phenyl, pyridyl, cubanyl, saturated or partially unsaturated monocyclic ring, or saturated or partially unsaturated bridged, fused, or spirocyclic ring are each optionally substituted with 1, 2, or 3 substituents independently selected from halogen, Ci-C4aliphatic, haloCi-C4alkyl, C3-C6cycl
- R 2A is 2-benzimidazolyl, 2-naphthyl, or 3-quinolinyl, each of which is optionally substituted with 1, 2, or 3 substituents independently selected from halogen, C1-C4aliphatic, haloC1-C4alkyl and - OH.
- R 2A is phenyl optionally substituted with 1, 2, or 3 substituents independently selected from halogen, C1-C4alkyl, haloC1-C4alkyl, C3-C6cycloalkyl, haloC3-C6- cycloalkyl, -OH, -CN, C 1 -C 4 alkoxy, haloC 1 -C 4 alkoxy, and –SF 5 , two substituents on adjacent atoms of the phenyl together with their intervening atoms form a 4-7 membered carbocyclyl fused to the phenyl, and two substituents on adjacent atoms of the phenyl together with their intervening atoms form a 4-7 membered heterocyclyl (having 1-4 heteroatoms independently selected from nitrogen, oxygen, and sulfur) fused to the phenyl.
- substituents on adjacent atoms of the phenyl together with their intervening atoms form a 4-7 membered carb
- R 2A is phenyl optionally substituted with 1, 2, or 3 substituents independently selected from halogen, C 1 -C 4 alkyl, haloC 1 -C 4 alkyl, C 3 -C 6 cycloalkyl, haloC 3 -C 6- cycloalkyl, -OH, -CN, C1-C4alkoxy, haloC1-C4alkoxy, and –SF5.
- R 2A is phenyl optionally substituted with 1, 2, or 3 substituents independently selected from halogen, C1- C 4 alkyl, and haloC 1 -C 4 alkyl.
- R 2A is phenyl optionally substituted with a halogen, C1-C4alkyl, and haloC1-C4alkyl. In some embodiments, R 2A is phenyl optionally substituted with 2 substituents independently selected from halogen, C1-C4alkyl, and haloC1- C 4 alkyl. In some embodiments, R 2A is phenyl optionally substituted with 3 substituents independently selected from halogen, C 1 -C 4 alkyl, and haloC 1 -C 4 alkyl.
- R 2A is pyridyl optionally substituted with 1, 2, or 3 substituents independently selected from halogen, C1-C4alkyl, haloC1-C4alkyl, C3-C6cycloalkyl, haloC3-C6- cycloalkyl, -OH, -CN, C 1 -C 4 alkoxy, haloC 1 -C 4 alkoxy, and –SF 5 , two substituents on adjacent atoms of the pyridyl together with their intervening atoms form a 4-7 membered carbocyclyl fused to the pyridyl, and two substituents on adjacent atoms of the pyridyl together with their intervening atoms form a 4-7 membered heterocyclyl (having 1-4 heteroatoms independently selected from nitrogen, oxygen, and sulfur) fused to the pyridyl.
- R 2A is pyridyl optionally substituted with a halogen, C 1 -C 4 alkyl, or haloC 1 -C 4 alkyl. In some embodiments, R 2A is pyridyl optionally substituted with 2 substituents independently selected from halogen, C1-C4alkyl, and haloC1- C4alkyl. In some embodiments, R 2A is pyridyl optionally substituted with 3 substituents independently selected from halogen, C 1 -C 4 alkyl, and haloC 1 -C 4 alkyl.
- R 2A is cubanyl, a saturated or partially unsaturated 4-8 membered monocyclic ring, a saturated or partially unsaturated bridged, fused, or spirocyclic 5-, 6-, 7-, 8-, 9-, 10-, 11-, or 12-membered ring, wherein said saturated or partially unsaturated monocyclic ring, or saturated or partially unsaturated bridged, fused, or spirocyclic ring contains 0, 1, 2, 3, or 4 heteroatoms independently selected from nitrogen, oxygen, and sulfur; and wherein said cubanyl, partially unsaturated monocyclic ring, or saturated or partially unsaturated bridged, fused, or spirocyclic ring are each optionally substituted with 1, 2, or 3 substituents independently selected from halogen, C1-C4alkyl, haloC1-C4alkyl, C3-C6cycloalkyl, haloC3-C6cycloalkyl, -OH, -
- R 2A is bicyclo[1.1.1]pentyl optionally substituted with 1, 2, or 3 substituents independently selected from halogen, C1-C4alkyl, and haloC1-C4alkyl.
- R 2A is bicyclo[2.2.2]octyl optionally substituted with 1, 2, or 3 substituents independently selected from halogen, C 1 -C 4 alkyl, and haloC 1 -C 4 alkyl.
- R 2A is bicyclo[3.1.0]pentyl optionally substituted with 1, 2, or 3 substituents independently selected from halogen, C 1 -C 4 alkyl, and haloC1-C4alkyl.
- R 2A is cubanyl optionally substituted with 1, 2, or 3 substituents independently selected from halogen, C1-C4alkyl, and haloC1-C4alkyl.
- R 2A is 2-oxabicyclo[2.1.1]hexan-4-yl optionally substituted with 1, 2, or 3 substituents independently selected from halogen, C1-C4alkyl, and haloC1-C4alkyl.
- R 2A is a saturated or partially unsaturated bridged 5-, 6-, 7-, 8-, 9-, 10-, 11-, or 12-membered ring, which contains 0, 1, 2, 3, or 4 heteroatoms independently selected from nitrogen, oxygen, and sulfur; and wherein said bridged ring is optionally substituted with 1, 2, or 3 substituents independently selected from halogen, C1-C4alkyl, haloC1-C4alkyl, C3- C 6 cycloalkyl, haloC 3 -C 6 cycloalkyl, -OH, -CN, C 1 -C 4 alkoxy, haloC 1 -C 4 alkoxy, C 3 -C 6 cycloalkoxy, haloC 3 -C 6 cycloalkoxy and –SF 5 .
- R 2A is a saturated or partially unsaturated fused 5-, 6-, 7-, 8-, 9- , 10-, 11-, or 12-membered ring, which contains 0, 1, 2, 3, or 4 heteroatoms independently selected from nitrogen, oxygen, and sulfur; and wherein said fused ring is optionally substituted with 1, 2, or 3 substituents independently selected from halogen, C1-C4alkyl, haloC1-C4alkyl, C3-C6- cycloalkyl, haloC3-C6cycloalkyl, -OH, -CN, C1-C4alkoxy, haloC1-C4alkoxy, C3-C6cycloalkoxy, haloC 3 -C 6 cycloalkoxy and –SF 5 .
- R 2A is a saturated or partially unsaturated spirocyclic 5-, 6-, 7-, 8-, 9-, 10-, 11-, or 12-membered ring, which contains 0, 1, 2, 3, or 4 heteroatoms independently selected from nitrogen, oxygen, and sulfur; and wherein said spirocyclic ring is optionally substituted with 1, 2, or 3 substituents independently selected from halogen, C 1 -C 4 alkyl, haloC 1 - C4alkyl, C3-C6cycloalkyl, haloC3-C6cycloalkyl, -OH, -CN, C1-C4alkoxy, haloC1-C4alkoxy, C3-C6- cycloalkoxy, haloC3-C6cycloalkoxy and –SF5.
- R 2A is bicyclo[1.1.1]pentyl optionally substituted with 1, 2, or 3 substituents independently selected from halogen, C1-C4alkyl, haloC1-C4alkyl, C3-C6cycloalkyl, haloC3-C6cycloalkyl, -OH, -CN, C1-C4alkoxy, haloC1-C4alkoxy, C3-C6cycloalkoxy, haloC3-C6- cycloalkoxy and –SF 5 .
- R 2A is bicyclo[1.1.1]pentyl optionally substituted with 1, 2, or 3 substituents independently selected from halogen, C1-C4alkyl, and haloC1-C4alkyl. In some embodiments, R 2A is bicyclo[1.1.1]pentyl optionally substituted with a halogen, C1- C 4 alkyl, or haloC 1 -C 4 alkyl. In some embodiments, R 2A is bicyclo[1.1.1]pentyl optionally substituted with 2 substituents independently selected from halogen, C 1 -C 4 alkyl, and haloC 1 - C4alkyl.
- R 2A is bicyclo[1.1.1]pentyl optionally substituted with 3 substituents independently selected from halogen, C1-C4alkyl, and haloC1-C4alkyl.
- R 2A is Ring F selected from the group consisting of: , wherein x, y, and q are independently selected , , , p y from O, NR 15 , CHR 15 or CR 15 R 15 ; R 15 is independently selected from H, halogen, C1-C4aliphatic, haloC1-C4alkyl, C3-C6cycloalkyl, haloC3-C6cycloalkyl, -OH, -CN, C 1 -C 4 alkoxy, haloC 1 -C 4 alkoxy, C 3 -C 6 cycloalkoxy, haloC 3 -C 6 cycloalkoxy and –SF 5 .
- R 2A is Ring F of the following structu , wherein R 15 is selected from halogen, C 1-C4aliphatic, haloC1-C4alkyl, C3-C6cycloalkyl, haloC3- C6cycloalkyl, -OH, -CN, C1-C4alkoxy, haloC1-C4alkoxy, C3-C6cycloalkoxy, haloC3-C6- cycloalkoxy and –SF 5 .
- R 2A is 2-benzimidazolyl, 2-naphthyl, or 3-quinolinyl, each of which is optionally substituted with 1, 2, or 3 substituents independently selected from halogen, C1-4alkyl and -OH.
- R 2A is 2-benzimidazolyl optionally substituted with 1, 2, or 3 substituents independently selected from halogen, C 1 - 4 alkyl and -OH.
- R 2A is 3-quinolinyl optionally substituted with 1, 2, or 3 substituents independently selected from halogen, C1-4alkyl, and -OH.
- R 2A is phenyl comprising a -CF 3 substituent or pyridyl comprising a -CF3 substituent.
- R 2A is bicyclo[1.1.1]pentyl comprising a -CF3 substituent or bicyclo[1.1.1]pentyl comprising a -CHF 2 substituent.
- R 2A is bicyclo[2.2.2]octyl comprising a -CF 3 substituent or bicyclo[2.2.2]octyl comprising a -CHF 2 substituent.
- R 2A is bicyclo[3.1.0]pentyl comprising a -CF3 substituent or bicyclo[3.1.0]pentyl comprising a -CHF2 substituent.
- R 2A is cubanyl comprising a -CF 3 substituent or cubanyl comprising a -CHF 2 substituent. [00125] In some embodiments, R 2A is as selected from one of the substituents of Table 1, Table 1a or Table 1b.
- R 3 is hydrogen, C 1 -C 4 alkyl, C 3 -C 5 cycloalkyl, C 1 - C4alkoxy, -NHR 3A , -N(R 3A )2 or C1-C4alkylthio each of which, besides hydrogen, is optionally substituted with -OH, 1-5 independently selected halogen, or C1-C4alkoxy.
- R 3 is hydrogen.
- R 3 is C 1 -C 4 alkyl optionally substituted with -OH, 1-5 independently selected halogen, or C 1 -C 4 alkoxy.
- R 3 is C1-C4alkyl.
- R 3 is -CH2CH3. In some embodiments, R 3 is -CH3. In some embodiments, R 3 is C3-C5cycloalkyl, C1-C4alkoxy, -NHR 3A , -N(R 3A )2 or C1- C 4 alkylthio optionally substituted with -OH, 1-5 independently selected halogen, or C 1 -C 4 alkoxy. In some embodiments, R 3 is C3-C5cycloalkyl optionally substituted with -OH, 1-5 independently selected halogen, or C1-C4alkoxy.
- R 3 is C1-C4alkoxy optionally substituted with -OH, 1-5 independently selected halogen, or C 1 -C 4 alkoxy.
- R 3 is - NHR 3A optionally substituted with -OH, 1-5 independently selected halogen, or C1-C4alkoxy.
- R 3 is -N(R 3A )2 optionally substituted with -OH, 1-5 independently selected halogen, or C 1 -C 4 alkoxy.
- R 3 is C 1 -C 4 alkylthio optionally substituted with - OH, 1-5 independently selected halogen, or C 1 -C 4 alkoxy.
- R 3 is selected from the group consisting of C1-C4alkyl and C3-C5cycloalkyl. [00128] In some embodiments, R 3 is as selected from one of the substituents of Table 1, Table 1a or Table 1b. [00129] As described generally above, each R 3A is independently selected at each occurrence from C1-C4alkyl. In some embodiments, R 3A is -CH3. In some embodiments, R 3A is -CH2CH3. In some embodiments, R 3A is propyl. In some embodiments, R 3A is butyl. [00130] In some embodiments, R 3A is as selected from one of the substituents of Table 1, Table 1a or Table 1b.
- L is a linker selected from -C(O)-, -S(O)-, -S(O) 2 -, and .
- linker L is -C(O)-.
- linker L is -S(O)-.
- linker L is -S(O)2-.
- linker L i is as selected from one of the substituents of Table 1, Table 1a or Table 1b.
- R 4 is selected from one of a), b), and c): a) R 4 is a Ring B that is selected from the group consisting of
- any substituents that are present on Ring B selected from R 4A , R 4B , R 4C , R 4D , R 4E , and R 4F are each independently selected from hydrogen; halogen; -OH; -CN; C 1 -C 4 alkyl; C 2 - C4alkenyl; C2-C4alkynyl; C1-C4alkoxy; haloC1-C4alkyl; C1-C3alkyl substituted with -OH, -OCH3, or -OCH2CH3; haloC1-C4alkoxy; C3-C6cycloalkyl; C3-C6cycloalkoxy; and NR 13 R 14 ; or R 4A and R 4B , along with their intervening atoms, join to form 4-7 membered optionally substituted carbocyclyl, 4-7 membered optionally substituted heterocyclyl, or 5-6 membered optionally substituted heteroary
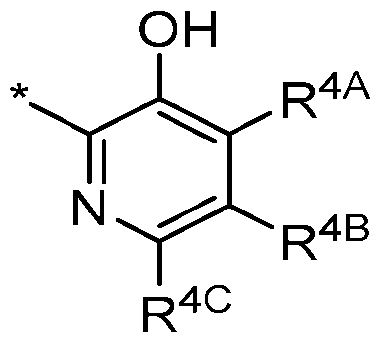
- R 4 is Ring B of the following structure: wherein * is a point of attachment to L n er a s onded to Ring A in Formula I; and wherein: R 4A , R 4C , and R 4D are each independently selected from hydrogen; halogen; -CN; C1- C 4 alkyl; C 2 -C 4 alkenyl; C 2 -C 4 alkynyl; haloC 1 -C 4 alkyl; C 1 -C 3 alkyl substituted with -OH, - OCH 3 , or -OCH 2 CH 3 ; haloC 1 -C 4 alkoxy; C 3 -C 6 cycloalkyl; C 3 -C 6 cycloalkoxy; and NR 13 R 14 ; or R 4C and R 4D , along with their intervening atoms, join to form 4-7 membered carbocyclyl or 4-7 membered heterocyclyl (having 1-4 heteroatom
- R 4 is Ring B of the following structure: wherein * is a point of attachment to L Ring A in Formula I; and wherein: R 4A is -OCH3, -OCH2CH3, or -OCHF2; R 4C and R 4D are each independently selected from hydrogen; -CN; C 1 -C 4 alkyl; C 2 - C 4 alkenyl; C 2 -C 4 alkynyl; haloC 1 -C 4 alkyl; C 1 -C 3 alkyl substituted with -OH, -OCH 3 , or - OCH2CH3; haloC1-C4alkoxy; C3-C6cycloalkyl; C3-C6cycloalkoxy; and NR 13 R 14 ; and R 13 is independently selected at each occurrence from hydrogen and C 1 -C 4 alkyl optionally substituted with -OH, -OCH 3 , or -OCH 2 CH 3 ; R 14 is hydrogen or
- R 4 is Ring B of the following structure: wherein * is a point of attachment to L that is bonded to Ring A in Formula I; and wherein: R 4A is -OCH3, -OCH2CH3, or -OCHF2; R 4C and R 4D are each independently selected from hydrogen; -CN; C 1 -C 4 alkyl; C 2 - and R 4D are each independently selected from hydrogen; -CN; C1-C4alkyl; C2-C4alkenyl; C2- C4alkynyl; haloC1-C4alkyl; C1-C3alkyl substituted with -OH, -OCH3, or -OCH2CH3; haloC 1 -C 4 alkoxy; C 3 -C 6 cycloalkyl; C 3 -C 6 cycloalkoxy; and NR 13 R 14 ; and R 13 is independently selected at each occurrence from hydrogen and C 1 -C 4 alkyl optionally
- R 4 is Ring B of the following structure: wherein * is a point of attachment to L a s o e o Ring A in Formula I; and wherein: R 4A , R 4C , and R 4D are each independently selected from hydrogen; halogen; and C 1 - C4alkyl.
- R 4 is Ring B of the following structure: wherein * is a point of attachment to L that is bonded to Ring A in Formula I; and wherein: R 4A , R 4B , and R 4C are each independently selected from hydrogen; halogen; -CN; C1- C4alkyl; C2-C4alkenyl; C2-C4alkynyl; C1-C4alkoxy; haloC1-C4alkyl; C1-C3alkyl substituted with -OH, -OCH 3 , or -OCH 2 CH 3 ; haloC 1 -C 4 alkoxy; C 3 -C 6 cycloalkyl; C 3 -C 6 cycloalkoxy; and NR 13 R 14 ; or R 4A and R 4B , along with their intervening atoms, join to form 4-7 membered carbocyclyl or heterocyclyl (having 1-4 heteroatoms independently selected from nitrogen, oxygen, and
- R 4 is Ring B of the following structure: wherein * is a point of attachment to L that is bonded to Ring A in Formula I; and wherein: R 4A and R 4B , along with their intervening atoms, join to form 4-7-membered carbocyclyl or heterocyclyl (having 1-4 heteroatoms independently selected from nitrogen, oxygen, and sulfur) that is fused to Ring B; and R 4C is hydrogen.
- R 4 is Ring B of the following structure: wherein * is a point of attachment to Ring A in Formula I; and wherein: R 4A and R 4B , along with their intervening atoms, join to form 5-membered heterocyclyl (having 1 oxygen atom) that is fused to Ring B; and R 4C is hydrogen.
- R 4 is Ring B of the following structure: wherein * is a point of attachment to L that is bonded to Ring A in Formula I; and wherein: R 4A , R 4B , and R 4D are each independently selected from hydrogen; halogen; -CN; C1- C 4 alkyl; C 2 -C 4 alkenyl; C 2 -C 4 alkynyl; C 1 -C 4 alkoxy; haloC 1 -C 4 alkyl; C 1 -C 3 alkyl substituted with -OH, -OCH 3 , or -OCH 2 CH 3 ; haloC 1 -C 4 alkoxy; C 3 -C 6 cycloalkyl; C 3 -C 6 cycloalkoxy; and NR 13 R 14 ; or R 4A and R 4B , along with their intervening atoms, join to form 4-7 membered carbocyclyl or heterocyclyl (having 1-4 heteroatoms independently
- R 4 is Ring B of the following structure: wherein * is a point of attachment to L Ring A in Formula I; and wherein: R 4A and R 4D are each hydrogen; and R 4B is C 1 -C 4 alkyl.
- R 4 is Ring B of the following structure: wherein * is a point of attachment to L that is bonded to Ring A in Formula I; and wherein: R 4A and R 4C are each independently selected from hydrogen; halogen; -CN; C 1 -C 4 alkyl; C2-C4alkenyl; C2-C4alkynyl; C1-C4alkoxy; haloC1-C4alkyl; C1-C3alkyl substituted with - OH, -OCH 3 , or -OCH 2 CH 3 ; haloC 1 -C 4 alkoxy; C 3 -C 6 cycloalkyl; C 3 -C 6 cycloalkoxy; and NR 13 R 14 ; and R 13 is independently selected at each occurrence from hydrogen and C 1 -C 4 alkyl optionally substituted with -OH, -OCH3, or -OCH2CH3; R 14 is hydrogen; or NR 13 R 14 forms
- R 4 is Ring B of the following structure: wherein * is a point of attachment to Ring A in Formula I; and wherein: R 4A and R 4C are each independently selected from hydrogen and C 1 -C 4 alkyl.
- R 4 is Ring B of the following structure: wherein * is a point of attachment to L t at s bonded to Ring A in Formula I; and wherein: R 4A , R 4B , R 4C , R 4D , and R 4E are each independently selected from hydrogen; halogen; -CN; C1-C4alkyl; C2-C4alkenyl; C2-C4alkynyl; C1-C4alkoxy; haloC1-C4alkyl; C1-C3alkyl substituted with -OH, -OCH3, or -OCH2CH3; haloC1-C4alkoxy; C3-C6cycloalkyl; C3- C
- R 4 is Ring B of the following structure: wherein * is a point of attachment to L that is bonded to Ring A in Formula I; and wherein: R 4A , R 4B , R 4C , R 4D , and R 4E are each independently selected from hydrogen; halogen; C1- C 4 alkyl; and C 1 -C 4 alkoxy; or R 4C and R 4D , along with their intervening atoms, join to form a 4- 7-membered heterocyclyl (having 1-3 nitrogen atoms) fused to Ring B; and R 4A , R 4B , and R 4E are each hydrogen.
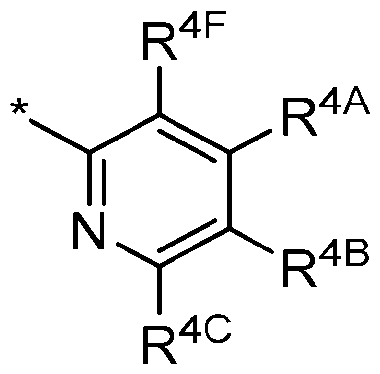
- R 4 is Ring B of the following structure: wherein * is a point of attachment to L Ring A in Formula I; and wherein: R 4F and R 4A , along with their intervening atoms, join to form 5-6 membered heteroaryl (having 1-4 heteroatoms independently selected from nitrogen, oxygen, and sulfur) fused to Ring B; and R 4B and R 4C are each independently selected from hydrogen; halogen; -CN; C1-C4alkyl; C2-C4alkenyl; C2-C4alkynyl; haloC1-C4alkyl; C1-C3alkyl substituted with -OH, -OCH3, or -OCH2CH3; haloC1-C4alkoxy; C3-C6cycloalkyl; C3-C6cycloalkoxy; and NR 13 R 14 ; R 13 is independently selected at each occurrence from hydrogen and C1-C4alkyl optionally substituted with -OH
- R 4 is Ring B of the following structure: wherein * is a point of attachment to Ring A in Formula I; and wherein: R 4F and R 4A , along with their intervening atoms, join to form 5-6-membered heteroaryl (having 1-2 nitrogen atoms) fused to Ring B; and R 4B and R 4C are each hydrogen.
- R 4 is Ring B of the following structure: wherein * is a point of attachment to L Ring A in Formula I; and wherein: R 4A , R 4C , R 4D , and R 4F are each independently selected from hydrogen; halogen; -CN; C 1 - C4alkyl; C2-C4alkenyl; C2-C4alkynyl; C1-C4alkoxy; haloC1-C4alkyl; C1-C3alkyl substituted with -OH, -OCH 3 , or -OCH 2 CH 3 ; haloC 1 -C 4 alkoxy; C 3 -C 6 cycloalkyl; C 3 -C 6 cycloalkoxy; and NR 13 R 14 ; or R 4C and R 4D , along with their intervening atoms, join to form 4-7 membered optionally substituted carbocyclyl or optionally substituted heterocyclyl (having 1-4 heteroatom
- R 4 is a 5-membered heteroaryl (having 1 heteroatom independently selected from nitrogen, oxygen, and sulfur and 0, 1, 2, or 3 additional ring nitrogen atoms), wherein said heteroaryl is substituted with 0-4 groups independently selected from halogen, -OH, -CN, C1-C4alkyl, haloC1-C4alkyl, C3-C6cycloalkyl, and C1-C4alkoxy.
- R 4 is a 5-membered heteroaryl (having 1 heteroatom independently selected from nitrogen, oxygen, and sulfur and 0, 1, 2, or 3 additional ring nitrogen atoms), wherein said heteroaryl is substituted with 0-4 groups independently selected from OH, - CH3, -CHF2, cyclopropyl, and -OCH3.
- R 4 is a C 1 -C 4 alkyl, C 1 -C 4 alkoxy, or C 3 -C 6 cycloalkyl, each of which is substituted with 0-3 groups independently selected from halogen, -CN, -OH, C1-C4alkyl, C1-C4alkoxy, optionally substituted 5-6-membered heterocyclyl, and optionally substituted 5-6- membered heterocyclyloxy.
- R 4 is a C 1 -C 4 alkyl, substituted with 0-3 independently selected halogen, -CN, -OH, C 1 -C 4 alkyl, and C 1 -C 4 alkoxy.
- R 4 is a C1-C4alkoxy, substituted with 0-3 independently selected halogen, -CN, -OH, C1-C4alkyl, and C1-C4alkoxy. In some embodiments, R 4 is a C3-C6cycloalkyl, substituted with 0-3 independently selected halogen, -CN, -OH, C 1 -C 4 alkyl, and C 1 -C 4 alkoxy. [00157] In some embodiments, R 4 is an isoxazolyl substituted with -OH or C1-C4alkoxy.
- R 4 is a 5-membered heteroaryl (having 1 heteroatom independently selected from nitrogen, oxygen, and sulfur and 0, 1, 2, or 3 additional ring nitrogen atoms) selected from the group consisting of thiophenyl, imidazolyl, pyrazolyl, tetrazolyl, thiazolyl, isothiazolyl, 1,2,5-thiadiazolyl, 1,3,4-thiadiazolyl, oxazolyl, isoxazolyl, 1,2,4- oxadiazolyl, 1,2,3-triazolyl, 1,2,4-triazolyl, wherein said heteroaryl is substituted with 0-4 groups independently selected from halogen, -OH, -CN, C1-C4alkyl, haloC1-C4alkyl, C3-C6cycloalkyl, and C1-C4alkoxy.
- R 4 is , , , , , , , , ,
- R 4 is as shown in a substituent of Table 1, Table la or Table lb.
- each R is independently hydrogen, or an optionally substituted Ci-ealiphatic group, an optionally substituted phenyl, an optionally substituted 3-7 membered saturated or partially unsaturated carbocyclic ring, an optionally substituted 3-7 membered saturated or partially unsaturated heterocyclic ring (having 1-2 heteroatoms independently selected from nitrogen, oxygen, and sulfur), or an optionally substituted 5-6 membered heteroaryl ring (having 1-4 heteroatoms independently selected from nitrogen, oxygen, and sulfur); or two R groups on the same atom are taken together with the same atom to form an optionally substituted 4-7 membered saturated ring, 4-7 membered partially unsaturated ring, or 5-6 membered heteroaryl ring (wherein said 4-7 membered saturated ring and 4-7 membered partially unsaturated ring has 0-3 heteroatoms independently selected from nitrogen, oxygen, and
- each R is independently hydrogen, or an optionally substituted Ci-6aliphatic group, an optionally substituted phenyl, an optionally substituted 3-7 membered saturated or partially unsaturated carbocyclic ring, an optionally substituted 3-7 membered saturated or partially unsaturated heterocyclic ring (having 1-2 heteroatoms independently selected from nitrogen, oxygen, and sulfur), or an optionally substituted 5-6 membered heteroaryl ring (having 1-4 heteroatoms independently selected from nitrogen, oxygen, and sulfur).
- two R groups on the same atom are taken together with the same atom to form an optionally substituted 4-7 membered saturated ring, partially unsaturated ring, or heteroaryl ring (wherein said 4-7 membered saturated ring, partially unsaturated ring, or heteroaryl ring, has 0-3 heteroatoms independently selected from nitrogen, oxygen, and sulfur).
- each R is independently hydrogen or a Ci-6 alkyl. In some embodiments, R is hydrogen.
- each R is as selected from one or more of the substituents of Table 1, Table la or Table lb.
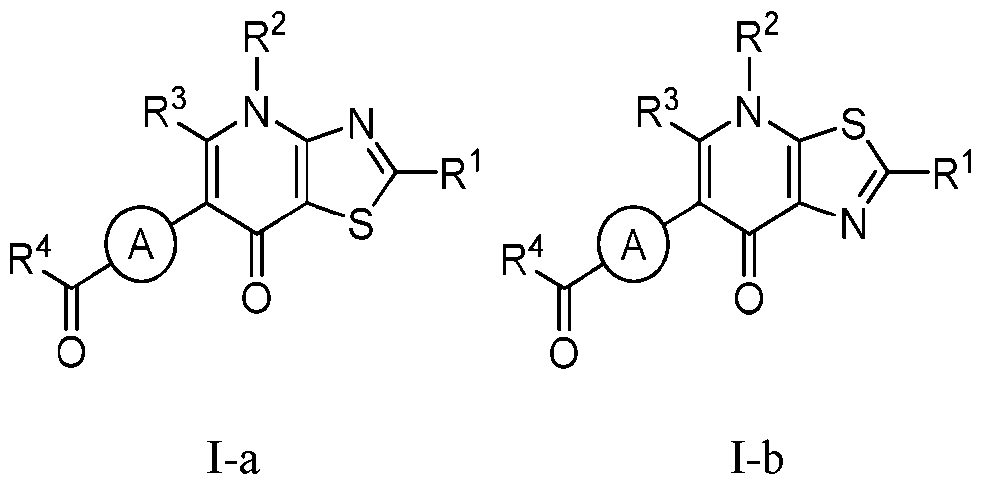
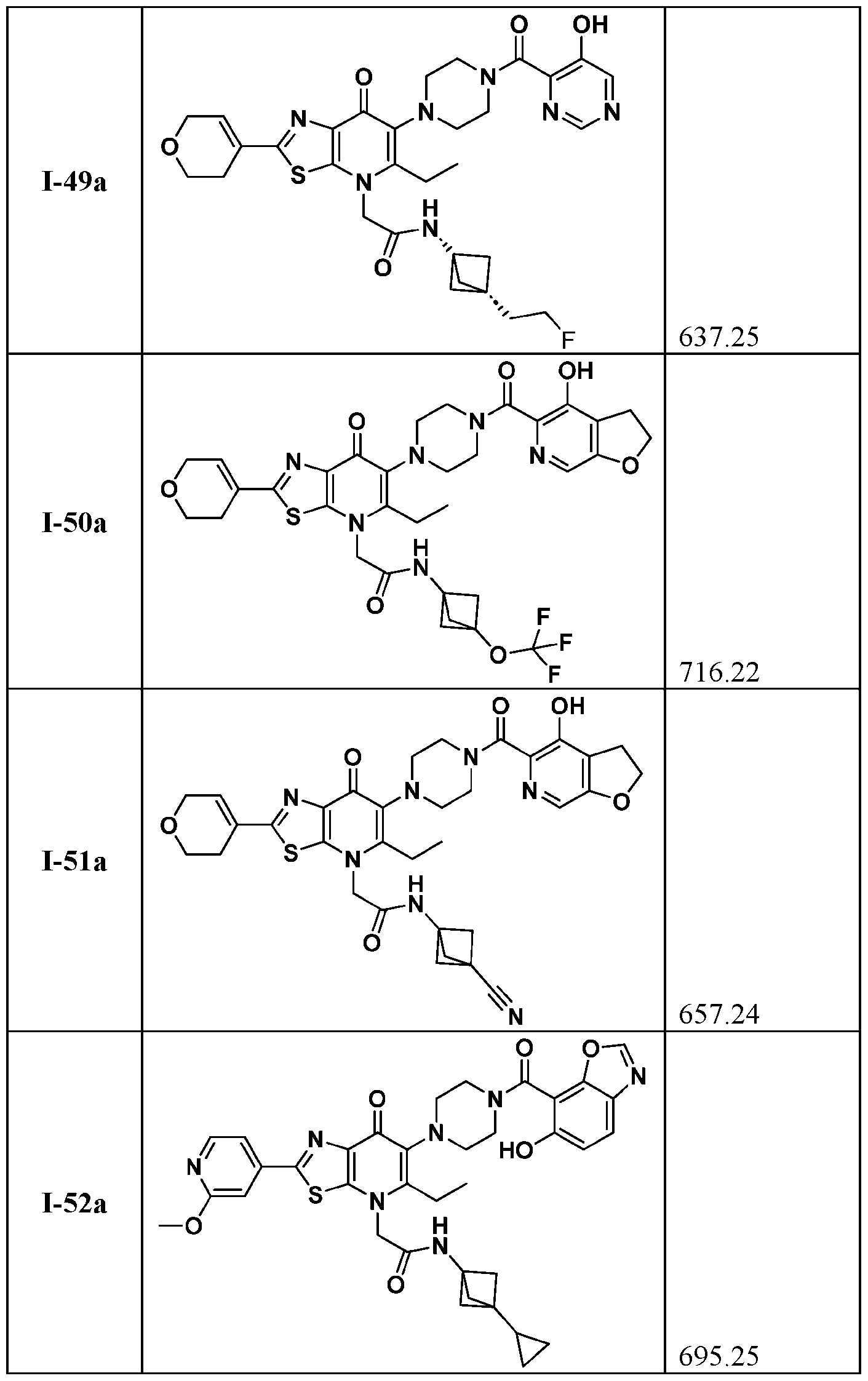
- the compound of Formula I is a compound of Formula I-a, Formula I-b, or a pharmaceutically acceptable salt thereof:
- Ring A, R 1 , R 2 , R 3 , and R 4 are as defined herein, both singly and in combination.
- the compound of Formula l is a compound of Formula I-a, or I-b or a pharmaceutically acceptable salt thereof wherein Ring A, R 1 , R 2 , R 3 , and R 4 , are as defined herein, both singly and in combination and R 2a
- the compound of Formula I is a compound of Formula I-a, Formula I-b, or a pharmaceutically acceptable salt thereof: wherein R 1 , R 2 , R 3 , and R 4 , are as defined herein, both singly and in combination, Ring A is
- the compound of Formula I is a compound of Formula ILa, Formula Il-b, Formula ILc, or a pharmaceutically acceptable salt thereof: wherein Ring A, Linker L, R 2 , R 3 , Y, Z and R 4 , are as defined herein, both singly and in combination.
- the compound of Formula I is a compound of Formula ILd
- the compound of Formula I is a compound of Formula III-a, Formula III-b, Formula III-c, or a pharmaceutically acceptable salt thereof: wherein Ring A, R 1 , R 2 , Y, Z and R 3 , are as defined herein, both singly and in combination, and wherein: X is CH, CR 7 , or N; R 5 is -OH or halogen; R 6 is halogen, C1-4alkyl, or C1-4alkoxy; each R 7 is independently hydrogen, halogen, C 1-4 alkyl, or C 1-4 alkoxy; R 8 is C 1-4 alkyl; each of the 0-2 instances of R 9 is independently a hydrogen or C1-4alkyl.
- X is CH or N; R 5 is -OH or fluoro; R 6 is fluoro, -CH 3 , or -OCH 3 ; each R 7 is independently hydrogen, fluoro, -CH3, or -OCH3; R 8 is -CH3; each instance of R 9 is independently a hydrogen or -CH 3 .
- the compound of Formula I is a compound of Formula IV-a, Formula IV-b, Formula IV-c, or a pharmaceutically acceptable salt thereof: wherei ation and wherein: R 5 is -OH or halogen; and R 6 is halogen, C1-4alkyl, or C1-4alkoxy.
- R 5 is -OH or fluoro
- R 6 is fluoro, -CH3, or -OCH3.
- the compound of Formula I is a compound of Formula V-a, Formula V-b, Formula V-c, or a pharmaceutically acceptable salt thereof:
- Ring A, R 2 , Y, Z and R 3 are as defined herein, both singly and in combination, and wherein:
- X is CH, CR 7 , or N; and each R 7 is independently hydrogen, halogen, Ci.4alkyl, or Ci.4alkoxy. In some embodiments: X is CH or N; and R 7 is hydrogen, fluoro, -CH3, or -OCH3.
- the compound of Formula I is a compound of Formula Vl-a, Formula Vl-b, Formula VI-c, or a pharmaceutically acceptable salt thereof: VI-c wherein Ring A, R 2 , Y, Z and R 3 , are as defined herein, both singly and in combination, and wherein:
- R 8 is Ci-4alkyl; and each of the 0-2 instances of R 9 are independently a hydrogen or Ci.4alkyl. In some embodiments: R 8 is -CH3; and each instance of R 9 is independently a hydrogen or -CH3.
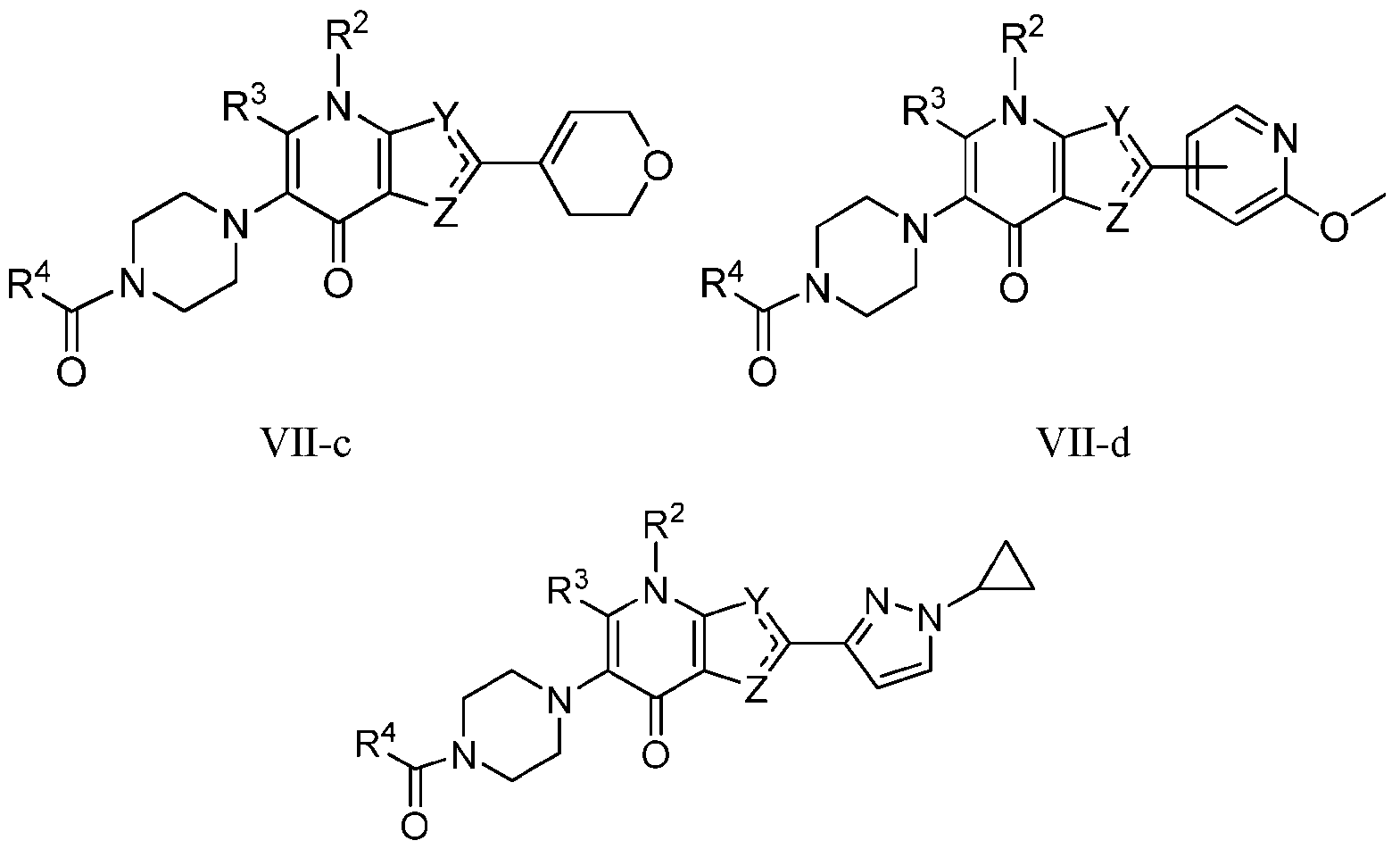
- the compound of Formula I is a compound of Formula Vll-a
- Vll-a Vll-b wherein R 1 , R 2 , R 3 , R A and R 4 , are as defined herein, both singly and in combination.
- the compound of Formula I is a compound of Formula Vll-a, or Vll-b or a pharmaceutically acceptable salt thereof: wherein R 1 , R 2 , R 3 , R A and R 4 , are as defined herein, both singly and in combination and R 2a is selected from
- the compound of Formula I is a compound of Formula VII-c, Formula Vll-d, Formula VH-e, or a pharmaceutically acceptable salt thereof:
- the compound of Formula I is a compound of Formula Vlll-a, Vlll-b, Formula VIII-c, or a pharmaceutically acceptable salt thereof: vm-c wherein R 1 , R 2 , Y, Z and R 3 , are as defined herein, both singly and in combination, and wherein: X is CH, CR 7 , or N; R 5 is -OH or halogen; R 6 is halogen, C1-4alkyl, or C1-4alkoxy; each R 7 is independently hydrogen, halogen, C 1-4 alkyl, or C 1-4 alkoxy; R 8 is C1-4alkyl; and each of the 0-2 instances of R 9 are independently a hydrogen or C1-4alkyl.
- the compound of Formula I is a compound of Formula IX-a, Formula IX-b, Formula IX-c, or a pharmaceutically acceptable salt thereof: wherein Y, Z, R 2 and R 3 , are as defined herein, both singly and in combination and wherein: R 5 is -OH or halogen; and R 6 is halogen, C1-4alkyl, or C1-4alkoxy. In some embodiments: R 5 is -OH or fluoro; and R 6 is fluoro, -CH 3 , or -OCH 3 .
- the compound of Formula I is a compound of Formula X-a, Formula X-b, Formula X-c, or a pharmaceutically acceptable salt thereof: wherein Y, Z, R 2 and R 3 , are as defined herein, both singly and in combination, and wherein: X is CH, CR 7 , or N; and each R 7 is independently hydrogen, halogen, C 1-4 alkyl, or C 1-4 alkoxy. [00180] In some embodiments: X is CH or N; and each R 7 is independently hydrogen, fluoro, -CH3, or -OCH3.
- the compound of Formula I is a compound of Formula XI-a, Formula XI-b, Formula XI-c, or a pharmaceutically acceptable salt thereof: wherein Y, Z, R 2 and R 3 , are as defined herein, both singly and in combination, and wherein: R 8 is C1-4alkyl; and each of the 0-2 instances of R 9 are independently a hydrogen or C 1-4 alkyl. In some embodiments: R 8 is -CH 3 ; and each instance of R 9 is independently a hydrogen or -CH 3 .
- the compound of Formula I is a compound of Formula X-a, Formula X-b, Formula X-c, or a pharmaceutically acceptable salt thereof:
- R 2 is C(R B )2C(O)NH
- R B is independently selected at each occurrence from hydrogen or -CH 3 ;
- R 2A is phenyl, pyridyl, or bicyclo[1.1.1]pentyl each of which is optionally substituted with 1, or 2, or 3 substituents independently selected from halogen, C1-C4alkyl, haloC1-C4alkyl, C3-C6- cycloalkyl, haloC 3 -C 6 cycloalkyl, -OH, -CN, C 1 -C 4 alkoxy, haloC 1 -C 4 alkoxy, and –SF 5 ; or R 2A is 2-benzimidazolyl, 2-naphthyl, or 3-quinolinyl, each of which is optionally,
- the compound of Formula I is a compound of Formula X-d, Formula X-e Formula X-f, or a pharmaceutically acceptable salt thereof:
- R 2 is C(R B )2C(O)NH
- R B is independently selected at each occurrence from hydrogen or -CH 3 ;
- R 2A is phenyl, pyridyl, or bicyclo[1.1.1]pentyl each of which is optionally substituted with 1, or 2, or 3 substituents independently selected from halogen, C1-C4alkyl, haloC1-C4alkyl, C3-C6- cycloalkyl, haloC 3 -C 6 cycloalkyl, -OH, -CN, C 1 -C 4 alkoxy, haloC 1 -C 4 alkoxy, and –SF 5 ; or R 2A is 2-benzimidazolyl, 2-naphthyl, or 3-quinolinyl, each of which is optionally substitute
- the compound of Formula I is selected from one of those depicted in Table 1, Table 1a, or Table 1b, or a pharmaceutically acceptable salt thereof.
- Table 1a, or 1b identifies compounds by their IUPAC name and Table 2
- Table 2a lists the same compounds and shows their chemical structure.
- Table 1a’s, or Table 1b’s name for a compound and Table 2's, Table 2a’s, and Table 2b’s structure for that same compound;
- Table 2's, Table 2a’s, and Table 2b’s compound structures will dominate and identify the compound corresponding to each respective compound number (I-#) in Table 1, Table 1a, or Table 1b.
- compositions, methods of treatment and uses of compounds [00185]
- the present invention provides a pharmaceutical composition comprising a compound of the present invention, or a pharmaceutically acceptable salt thereof, and a pharmaceutically acceptable carrier.
- the composition comprises at least two pharmaceutically acceptable carriers, such as those described herein.
- the pharmaceutical composition can be formulated for particular routes of administration such as oral administration, parenteral administration (e.g. by injection, infusion, transdermal or topical administration), and rectal administration, in particular oral administration. Topical administration may also pertain to inhalation or intranasal application.
- compositions of the present invention can be made up in a solid form (including, without limitation, capsules, tablets, pills, granules, powders or suppositories), or in a liquid form (including, without limitation, solutions, suspensions or emulsions). Tablets may be either film coated or enteric coated according to methods known in the art.
- the pharmaceutical compositions are tablets or gelatin capsules comprising the active ingredient together with one or more of: a) diluents, e.g., lactose, dextrose, sucrose, mannitol, sorbitol, cellulose and/or glycine; b) lubricants, e.g., silica, talcum, stearic acid, its magnesium or calcium salt and/or polyethylene glycol; for tablets also c) binders, e.g., magnesium aluminum silicate, starch paste, gelatin, tragacanth, methylcellulose, sodium carboxymethylcellulose and/or polyvinylpyrrolidone; if desired d) disintegrants, e.g., starches, agar, alginic acid or its sodium salt, or effervescent mixtures; and e) absorbents, colorants, flavors and sweeteners.
- diluents e.g., lactose, dextrose,
- Typical approaches to solubilize compounds for parenteral administration are the optimization of the pH or the use of co-solvents (e.g. PEG300, PEG400, propylene glycol, or ethanol). If these approaches are, for any reason, not feasible, the use of surfactants may be considered (e.g. Tween® 80 or Cremophor EL®). Cyclodextrins are established as safe solubilizing agents. Compounds with a high solubility in natural oils may be solubilized in parenteral fat emulsions. [00187] There is also provided a pharmaceutical composition comprising a compound of Formula I as described herein, or a pharmaceutically acceptable salt thereof, and one or more pharmaceutically acceptable carriers.
- co-solvents e.g. PEG300, PEG400, propylene glycol, or ethanol.
- surfactants e.g. Tween® 80 or Cremophor EL®
- Cyclodextrins are established as safe solubilizing agents.
- the compounds of Formula I of the present invention in free form or in pharmaceutically acceptable salt form, exhibit valuable pharmacological properties, e.g. WRN inhibiting properties, e.g. as indicated in vitro tests as provided in the next sections, and are therefore indicated for therapy, or for use as research chemicals, e.g. as a chemical probe, and as tool compounds.
- WRN inhibiting properties e.g. as indicated in vitro tests as provided in the next sections
- research chemicals e.g. as a chemical probe
- tool compounds e.g. as a chemical probe
- a compound of Formula I as described herein, as a research chemical, for example tool compound or chemical probe, in particular for research on WRN.
- a compound of Formula I as described herein, or a pharmaceutically acceptable salt thereof for use in the treatment of cancer.
- Cancers that may be treated by WRN inhibition include cancers that are characterized as microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR).
- dMMR mismatch repair deficient
- a compound of Formula I as described herein, or a pharmaceutically acceptable salt thereof may be useful in the treatment of a cancer that is characterized as microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR).
- a compound of Formula I as described herein, or a pharmaceutically acceptable salt thereof, for use as a medicament for use as a medicament.
- said use is: ⁇ for the treatment of a disease that is treated by WRN inhibition, ⁇ for the treatment of cancer, ⁇ for the treatment of cancer that is characterized as microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR), ⁇ for the treatment of cancer that is characterized as microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR), such as colorectal, gastric, prostate, endometrial, adrenocortical, uterine, cervical, esophageal, breast, kidney and ovarian cancer, ⁇ for the treatment of cancer that is characterized as microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR) is selected from colorectal, gastric, prostate and endometrial cancer, or ⁇ for the treatment of cancer wherein
- a method of: ⁇ modulating WRN activity in a subject comprising administering to the subject a therapeutically effective amount of the compound of Formula I as described herein, or a pharmaceutically acceptable salt thereof, ⁇ inhibiting WRN in a subject, wherein the method comprises administering to the subject a therapeutically effective amount of the compound of Formula I as described herein, or a pharmaceutically acceptable salt thereof, ⁇ treating a disorder or disease which can be treated by WRN inhibition in a subject, comprising administering to the subject a therapeutically effective amount of the compound of Formula I as described herein, or a pharmaceutically acceptable salt thereof, ⁇ treating cancer in a subject, comprising administering to the subject a therapeutically effective amount of the compound of Formula I as described herein, or a pharmaceutically acceptable salt thereof, ⁇ treating cancer in a subject, comprising administering a compound of Formula I as described herein, wherein the cancer is characterized as microsatellite instability- high (MSI-H) or mismatch repair de
- the cancer characterized as microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR) is selected from colorectal, gastric, prostate, endometrial, adrenocortical, uterine, cervical, esophageal, breast, kidney and ovarian cancer. More particularly, the cancer characterized as microsatellite instability-high (MSI- H) or mismatch repair deficient (dMMR) is selected from colorectal, gastric, prostate and endometrial cancer.
- Examples include uterine corpus endometrial carcinoma, colon adenocarcinoma, stomach adenocarcinoma, rectal adenocarcinoma, adrenocortical carcinoma, uterine carcinosarcoma, cervical squamous cell carcinoma, endocervical adenocarcinoma, esophageal carcinoma, breast carcinoma, kidney renal clear cell carcinoma, prostate cancer and ovarian serous cystadenocarcinoma.
- a compound of Formula I as described herein, or a pharmaceutically acceptable salt thereof ⁇ in therapy, ⁇ in the manufacture of a medicament, ⁇ in the manufacture of a medicament for the treatment of cancer.
- said cancer is characterized as microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR), ⁇ in the manufacture of a medicament for treatment of a disease which may be treated by WRN inhibition, wherein in particular, the cancer is characterized by microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR), for example colorectal, gastric, prostate, endometrial, adrenocortical, uterine, cervical, esophageal, breast, kidney and ovarian cancer, in particular, colorectal, gastric, prostate or endometrial cancer, or uterine corpus endometrial carcinoma, colon adenocarcinoma, stomach adenocarcinoma, rectal adenocarcinoma, adrenocortical carcinoma, uterine carcinosarcoma, cervical squamous cell carcinoma, endocervical adenocarcinoma, esophageal carcinoma,
- the subject has or is identified as having a microsatellite instable (MSI-H) cancer, e.g., in reference to a control, e.g., a normal, subject.
- MSI-H microsatellite instable
- the subject has MSI-H advanced solid tumors, a colorectal cancer (CRC), endometrial, uterine, stomach or other MSI-H cancer.
- CRC colorectal
- endometrial or stomach cancer which cancer has or is identified as having a microsatellite instability (MSI-H), e.g., in reference to a control, e.g., a normal, subject.
- MSI-H microsatellite instable
- the compounds can be present in the form of one of the possible stereoisomers or as mixtures thereof, for example as pure optical isomers, or as stereoisomer mixtures, such as racemates and diastereoisomer mixtures, depending on the number of asymmetric carbon atoms.
- the present invention is meant to include all such possible stereoisomers, including racemic mixtures, diasteriomeric mixtures and optically pure forms.
- Optically active (R)- and (S)-stereoisomers may be prepared using chiral synthons or chiral reagents, or resolved using conventional techniques. If the compound contains a double bond, the substituent may be E or Z configuration.
- the cycloalkyl substituent may have a cis- or trans-configuration. All tautomeric forms are also intended to be included. [00196] Any formula given herein is intended to represent unlabeled forms as well as isotopically labeled forms of the compounds, in addition to the deuteration specifically claimed in Formula I. Isotopically labeled compounds have structures depicted by the formulae given herein except that one or more atoms are replaced by an atom having a selected atomic mass or mass number. Isotopes that can be incorporated into compounds of the invention include, for example, isotopes of hydrogen.
- isotopes particularly deuterium (i.e., 2H or D) may afford certain therapeutic advantages resulting from greater metabolic stability, for example increased in vivo half-life or reduced dosage requirements or an improvement in therapeutic index or tolerability.
- deuterium in this context is regarded as a substituent of a compound of the present invention.
- concentration of deuterium may be defined by the isotopic enrichment factor.
- isotopic enrichment factor as used herein means the ratio between the isotopic abundance and the natural abundance of a specified isotope.
- a substituent in a compound of this invention is denoted as being deuterium, such compound has an isotopic enrichment factor for each designated deuterium atom of at least 3500 (52.5% deuterium incorporation at each designated deuterium atom), at least 4000 (60% deuterium incorporation), at least 4500 (67.5% deuterium incorporation), at least 5000 (75% deuterium incorporation), at least 5500 (82.5% deuterium incorporation), at least 6000 (90% deuterium incorporation), at least 6333.3 (95% deuterium incorporation), at least 6466.7 (97% deuterium incorporation), at least 6600 (99% deuterium incorporation), or at least 6633.3 (99.5% deuterium incorporation).
- isotopic enrichment factor can be applied to any isotope in the same manner as described for deuterium.
- isotopes that can be incorporated into compounds of the invention include isotopes of hydrogen, carbon, nitrogen, oxygen, phosphorous, fluorine, and chlorine, such as 3H, 11C, 13C, 14C, 15N, 18F 3 IP, 32P, 35S, 36CI, 1231, 1241, and 1251, respectively. Accordingly it should be understood that the invention includes compounds that incorporate one or more of any of the aforementioned isotopes, including for example, radioactive isotopes, such as 3H and 14C, orthose into which non-radioactive isotopes, such as 2H and 13C are present.
- radioactive isotopes such as 3H and 14C
- orthose into which non-radioactive isotopes such as 2H and 13C are present.
- Such isotopically labelled compounds are useful in metabolic studies (with 14C), reaction kinetic studies (with, for example 2H or 3H), detection or imaging techniques, such as positron emission tomography (PET) or single-photon emission computed tomography (SPECT) including drug or substrate tissue distribution assays, or in radioactive treatment of patients.
- PET positron emission tomography
- SPECT single-photon emission computed tomography
- an 18F or labeled compound may be particularly desirable for PET or SPECT studies.
- Isotopically-labeled compounds of the present invention can generally be prepared by conventional techniques known to those skilled in the art or by processes analogous to those described in the accompanying Examples and Preparations using an appropriate isotopically-labeled reagents in place of the nonlabeled reagent previously employed.
- a “compound of the present invention” or a “compound of Formula I” includes a zwitterion thereof, a non-zwitterion thereof (non-charged form), or a pharmaceutically acceptable salt of said zwitterionic or non-zwitterionic form thereof.
- “Zwitterion” or “zwitterionic form” means a compound containing both positive and negatively charged functional groups.
- cancer refers to a disease characterized by the rapid and uncontrolled growth of aberrant cells. Cancer cells can spread locally or through the bloodstream and lymphatic system to other parts of the body. Examples of various cancers are described herein and include but are not limited to colorectal, gastric, endometrial, prostate, adrenocortical, uterine, cervical, esophageal, breast, kidney, ovarian cancer and the like.
- WRN inhibitor or “WRN helicase inhibitor” as used herein means a compound that inhibits Werner Syndrome RecQ DNA helicase (WRN).
- WRN refers to the protein of Werner Syndrome RecQ DNA helicase.
- WRN includes mutants, fragments, variants, isoforms, and homologs of full-length wild-type WRN.
- the protein is encoded by the WRN gene (Entrez gene ID 7486; Ensembl ID ENSG00000165392). Exemplary WRN sequences are available at the Uniprot database under accession number Q 14191.
- Disease or condition mediated by WRN includes a disease or condition, such as cancer, which is treated by WRN inhibition.
- this may include cancers characterized as microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR).
- MSI-H microsatellite instability-high
- dMMR mismatch repair deficient
- “Microsatellite unstable cancer,” “microsatellite instability-high cancer,” “microsatellite high cancer” and “MSI-high cancer,” “MSIhi” and “MSI-H” when used herein, are used interchangeably, and describe cancers that have a high number of alterations in the length of simple repetitive genomic sequences within microsatellites.
- MSI-H or dMMR tumor status for patients can be performed using, e.g., polymerase chain reaction (PCR) tests for MSI-H status or immunohistochemistry (IHC) tests for dMMR.
- PCR polymerase chain reaction
- IHC immunohistochemistry
- Microsatellite instability can be found in colorectal cancer, gastric cancer and endometrial cancer in particular, but also in adrenocortical, uterine, cervical, esophageal, breast, kidney, prostate and ovarian cancers.
- microsatellite high cancers include uterine corpus endometrial carcinoma, colon adenocarcinoma, stomach adenocarcinoma, rectal adenocarcinoma, adrenocortical carcinoma, uterine carcinosarcoma, cervical squamous cell carcinoma, endocervical adenocarcinoma, esophageal carcinoma, breast carcinoma, kidney renal clear cell carcinoma and ovarian serous cystadenocarcinoma.
- a cancer that has “defective mismatch repair” (dMMR) or “dMMR character” includes cancer types associated with documented MLH1, PMS2, MSH2, MSH3, MSH6, MLH3, and PMS1 mutations or epigenetic silencing, microsatellite fragile sites, or other gene inactivation mechanisms, including but not limited to cancers of the lung, breast, kidney, large intestine, ovary, prostate, upper aerodigestive tract, stomach, endometrium, liver, pancreas, haematopoietic and lymphoid tissue, skin, thyroid, pleura, autonomic ganglia, central nervous system, soft tissue, pediatric rhabdoid sarcomas, melanomas and other cancers.
- dMMR defective mismatch repair
- a cell or cancer with “defective” mismatch repair has a significantly reduced (e.g., at least about 25%, 30%, 40%, 50%, 60%, 70%, 80% or 90% decrease) amount of mismatch repair. In some cases, a cell or cancer which is defective in mismatch repair will perform no mismatch repair.
- composition refers to a compound of the invention, or a pharmaceutically acceptable salt thereof, together with at least one pharmaceutically acceptable carrier, in a form suitable for oral or parenteral administration.
- the term “pharmaceutically acceptable carrier” refers to a substance useful in the preparation or use of a pharmaceutical composition and includes, for example, suitable diluents, solvents, dispersion media, surfactants, antioxidants, preservatives, isotonic agents, buffering agents, emulsifiers, absorption delaying agents, salts, drug stabilizers, binders, excipients, disintegration agents, lubricants, wetting agents, sweetening agents, flavoring agents, dyes, and combinations thereof, as would be known to those skilled in the art (see, for example, Remington The Science and Practice of Pharmacy, 22nd Ed. Pharmaceutical Press, 2013, pp. 1049-1070).
- synthetic lethality and “synthetically lethal” are used to refer to reduced cell viability and/or a reduced rate of cell proliferation caused by a combination of mutations or approaches to cause loss of function (e.g., RNA interference or protein function inhibition) in two or more genes but not by the loss of function of only one of these genes.
- loss of function e.g., RNA interference or protein function inhibition
- a therapeutically effective amount of a compound of the present invention refers to an amount of the compound of the present invention that will elicit the biological or medical response of a subject, for example, reduction or inhibition of an enzyme or a protein activity, or ameliorate symptoms, alleviate conditions, slow or delay disease progression, or prevent a disease, etc.
- the methods of the invention comprise administration of a therapeutically effective amount of a compound herein.
- a therapeutically effective amount refers to the amount of the compound of the present invention that, when administered to a subject, is effective to (1) at least partially alleviate, prevent and/or ameliorate a condition, or a disorder or a disease (i) mediated by WRN, or (ii) associated with WRN activity, or (iii) characterized by activity (normal or abnormal) of WRN; or (2) reduce or inhibit the activity of WRN.
- a therapeutically effective amount refers to the amount of the compound of the present invention that, when administered to a cell, or a tissue, or a non-cellular biological material, or a medium, is effective to at least partially reducing or inhibiting the activity of WRN, or reducing WRN protein levels.
- the term “subject” refers to primates (e g., humans, male or female), dogs, rabbits, guinea pigs, pigs, rats and mice.
- the subject is a primate, a rat or a mouse.
- the subject is a human.
- the term “inhibit,” “inhibition” or “inhibiting” refers to the reduction or suppression of a given condition, symptom, or disorder, or disease, or a significant decrease in the baseline activity of a biological activity or process.
- the term “treat,” “treating” or “treatment” of any disease or disorder refers to alleviating or ameliorating the disease or disorder (i.e., slowing or arresting the development of the disease or at least one of the clinical symptoms thereof); or alleviating or ameliorating at least one physical parameter or biomarker associated with the disease or disorder, including those which may not be discernible to the patient.
- the term “prevent,” “preventing” or “prevention” of any disease or disorder refers to the prophylactic treatment of the disease or disorder; or delaying the onset or progression of the disease or disorder.
- a subject is “in need of’ a treatment if such subject would benefit biologically, medically or in quality of life from such treatment.
- May j oin means j oins or does not j oin.
- any asymmetric atom (e.g., carbon or the like) of the compound(s) of the present invention can be present in racemic or enantiomerically enriched, for example the (R)-, (S)- or (R, S)-configuration.
- each asymmetric atom has at least 50 % enantiomeric excess, at least 60 % enantiomeric excess, at least 70 % enantiomeric excess, at least 80 % enantiomeric excess, at least 90 % enantiomeric excess, at least 95 % enantiomeric excess, or at least 99 % enantiomeric excess in the (R)- or (S)- configuration.
- Substituents at atoms with unsaturated double bonds may, if possible, be present in cis-(Z)- or trans-(E)- form.
- a compound of the present invention can be in the form of one of the possible stereoisomers, rotamers, atropisomers, tautomers or mixtures thereof, for example, as substantially pure geometric (cis or trans) stereoisomers, diastereomers, optical isomers (antipodes), racemates or mixtures thereof.
- Any resulting mixtures of stereoisomers can be separated on the basis of the physicochemical differences of the constituents, into the pure or substantially pure geometric or optical isomers, diastereomers, racemates, for example, by chromatography and/or fractional crystallization.
- Any resulting racemates of compounds of the present invention or of intermediates can be resolved into the optical antipodes by known methods, e.g., by separation of the diastereomeric salts thereof, obtained with an optically active acid or base, and liberating the optically active acidic or basic compound.
- a basic moiety may thus be employed to resolve the compounds of the present invention into their optical antipodes, e.g., by fractional crystallization of a salt formed with an optically active acid, e.g., tartaric acid, dibenzoyl tartaric acid, diacetyl tartaric acid, di-O,O’-p-toluoyl tartaric acid, mandelic acid, malic acid or camphor- 10-sulfonic acid.
- Racemic compounds of the present invention or racemic intermediates can also be resolved by chiral chromatography, e.g., high pressure liquid chromatography (HPLC) using a chiral adsorbent.
- Compounds of the invention i.e. compounds of Formula I that contain groups capable of acting as donors and/or acceptors for hydrogen bonds may be capable of forming co-crystals with suitable co-crystal formers.
- These co-crystals may be prepared from compounds of Formula I by known co-crystal forming procedures. Such procedures include grinding, heating, cosubliming, co-melting, or contacting in solution compounds of Formula I with the co-crystal former under crystallization conditions and isolating co-crystals thereby formed.
- Suitable cocrystal formers include those described in WO 2004/078163. Hence the invention further provides co-crystals comprising a compound of Formula I.
- the compounds of the present invention can also be obtained in the form of their hydrates, or include other solvents used for their crystallization.
- solvates refers to a molecular complex of a compound of the present invention (including pharmaceutically acceptable salts thereof) with one or more solvent molecules.
- solvent molecules are those commonly used in the pharmaceutical art, which are known to be innocuous to the recipient, e.g., water, ethanol, and the like.
- hydrate refers to the complex where the solvent molecule is water.
- composition or combination of the present invention may, for example, be in unit dosage of about 1-1000 mg of active ingredient(s) for a subject of about 50-70 kg-
- “Combination” refers to either a fixed combination in one dosage unit form, or a combined administration where a compound of Formula I, or a pharmaceutically acceptable salt thereof, and a combination partner (e.g. another drug as explained below, also referred to as “therapeutic agent” or “co-agent”) may be administered independently at the same time or separately within time intervals, especially where these time intervals allow that the combination partners show a cooperative, e.g. synergistic effect.
- the single components may be packaged in a kit or separately.
- One or both of the components e.g., powders or liquids
- co-administration or “combined administration” or the like as utilized herein are meant to encompass administration of the selected combination partner to a single subject in need thereof (e.g. a patient), and are intended to include treatment regimens in which the agents are not necessarily administered by the same route of administration or at the same time.
- pharmaceutical combination as used herein means a product that results from the mixing or combining of more than one therapeutic agent and includes both fixed and non-fixed combinations of the therapeutic agents.
- fixed combination means that the therapeutic agents, e.g. a compound of the present invention and a combination partner, are both administered to a patient simultaneously in the form of a single entity or dosage.
- non-fixed combination means that the therapeutic agents, e.g. a compound of the present invention and a combination partner, are both administered to a patient as separate entities either simultaneously, concurrently or sequentially with no specific time limits, wherein such administration provides therapeutically effective levels of the two compounds in the body of the patient.
- cocktail therapy e.g. the administration of three or more therapeutic agents.
- the combinations described herein can include a compound of Formula I and one or more additional therapeutic agents, e.g., one or more anti-cancer agents, cytotoxic or cytostatic agents, hormone treatment, vaccines, and/or other immunotherapies.
- the combination is further administered or used in combination with other therapeutic treatment modalities, including surgery, radiation, cryosurgery, and/or thermotherapy.
- Such combination therapies may advantageously utilize lower dosages of the administered therapeutic agents, thus avoiding possible toxicities or complications associated with the treatment.
- a combination comprising a compound of Formula I as described herein, or a pharmaceutically acceptable salt thereof, as described herein, and one or more additional therapeutically active agents.
- the additional therapeutic agent is, for example, a chemical compound, peptide, antibody, antibody fragment or nucleic acid, which is therapeutically active or enhances the therapeutic activity when administered to a patient in combination with a compound of the present disclosure.
- an additional therapeutically active agent is:
- Liposome injection (DaunoXome®), dexamethasone, docetaxel (Taxotere®), doxorubicin hydrochloride (Adriamycin®, Rubex®), etoposide (Vepesid®), fludarabine phosphate (Fludara®), 5-fluorouracil (Adrucil®, Efudex®), flutamide (Eulexin®), tezacitibine, Gemcitabine (difluorodeoxycitidine), hydroxyurea (Hydrea®), Idarubicin (Idamycin®), ifosfamide (IFEXO), irinotecan
- the additional therapeutically active agent is:
- a chemotherapy selected from anastrozole (Arimidex®), bicalutamide (Casodex®), bleomycin sulfate (Blenoxane®), busulfan (Myleran®), busulfan injection (Busulfex®), capecitabine (Xeloda®), N4-pentoxycarbonyl-5-deoxy-5-fluorocytidine, carboplatin (Parapl atin®), carmustine (BiCNU®), chlorambucil (Leukeran®), cisplatin (Platinol®), cladribine (Leustatin®), cyclophosphamide (Cytoxan® or Neosar®), cytarabine, cytosine arabinoside (Cytosar-U®), cytarabine liposome injection (DepoCyt®), dacarbazine (DTIC-Dome®), dactinomycin (Actinomycin D
- a PD-1 inhibitor selected from spartalizumab (Novartis), nivolumab (Bristol-Myers Squibb), pembrolizumab (Merck & Co), pidilizumab (CureTech), MED 10680 (Medimmune), cemiplimab (REGN2810, Regeneron), dostarlimab (TSR-042, Tesaro), PF- 06801591 (Pfizer), tislelizumab (BGB-A317, Beigene), BGB-108 (Beigene), INCSHR1210 (Incyte), balstilimab (AGEN2035, Agenus), sintilimab (InnoVent), toripalimab (Shanghai Junshi Bioscience), camrelizumab (Jiangsu Hengrui Medicine Co.), AMP -224 (Amplimmune), penpulimab (Akeso Biopharma Inc), zimberelimab
- the additional therapeutically active agent is an inhibitor of PD-1, e g., human PD-1.
- the immunomodulator is an inhibitor of PD-L1, e g., human PD-L1.
- the inhibitor of PD-1 or PD-L1 is an antibody molecule to PD-1 or PD-L1.
- the additional therapeutically active agent is an anti- PD-1 antibody molecule.
- the PD-1 inhibitor is an anti-PD-1 antibody molecule as described in US 2015/0210769, published on July 30, 2015, entitled “Antibody Molecules to PD- 1 and Uses Thereof.”
- a combination of a compound of Formula I or a pharmaceutically acceptable salt thereof, and a chemotherapy, and a PD-1 inhibitor are selected from those described above.
- the PD-1 inhibitor e.g., tislelizumab
- the PD-1 inhibitor is administered at a flat dose of between about 100 mg to about 600 mg.
- the PD-1 inhibitor is administered at a dose of between about 100 mg to about 500 mg.
- the PD-1 inhibitor is administered at a dose of between about 100 mg to about 400 mg.
- the PD-1 inhibitor is administered at a dose of between about 100 mg to about 300 mg.
- the PD- 1 inhibitor is administered at a dose of between about 100 mg to about 200 mg. In some embodiments, the PD-1 inhibitor is administered at a dose of between about 200 mg to about 600 mg. In some embodiments, the PD-1 inhibitor is administered at a dose of between about 200 mg to about 500 mg. In some embodiments, the PD-1 inhibitor is administered at a dose of between about 200 mg to about 400 mg. In some embodiments, the PD-1 inhibitor is administered at a dose of between about 200 mg to about 300 mg. In some embodiments, the PD-1 inhibitor is administered at a dose of between about 300 mg to about 600 mg. In some embodiments, the PD- 1 inhibitor is administered at a dose of between about 300 mg to about 500 mg.
- the PD-1 inhibitor is administered at a dose of between about 300 mg to about 400 mg. In some embodiments, the PD-1 inhibitor is administered at a dose of between about 400 mg to about 600 mg. In some embodiments, the PD-1 inhibitor is administered at a dose of between about 400 mg to about 500 mg. In some embodiments, the PD-1 inhibitor is administered at a dose of between about 500 mg to about 600 mg. In some embodiments, the PD-1 inhibitor is administered at a dose of between about 600 mg to about 700 mg. In some embodiments, the PD- 1 inhibitor is administered at a dose of between about 700 mg to about 800 mg. In some embodiments, the PD-1 inhibitor is administered at a dose of between about 800 mg to about 900 mg. In some embodiments, the PD-1 inhibitor is administered at a dose of between about 900 mg to about 1000 mg.
- the PD-1 inhibitor (e.g., tislelizumab) is administered at a flat dose of about 100 mg. In some embodiments, the PD-1 inhibitor is administered at a dose of about 200 mg. In some embodiments, the PD-1 inhibitor is administered at a dose of about 300 mg. In some embodiments, the PD-1 inhibitor is administered at a dose of about 400 mg. In some embodiments, the PD-1 inhibitor is administered at a dose of about 500 mg. In some embodiments, the PD-1 inhibitor is administered at a dose of about 600 mg. In some embodiments, the PD-1 inhibitor is administered at a dose of about 700 mg. In some embodiments, the PD-1 inhibitor is administered at a dose of about 800 mg. In some embodiments, the PD-1 inhibitor is administered at a dose of about 900 mg. In some embodiments, the PD-1 inhibitor is administered at a dose of about 1000 mg.
- the PD-1 inhibitor is administered at a dose of about 200 mg. In some embodiments, the PD-1 inhibitor is administered at
- the PD-1 inhibitor (e.g., tislelizumab) is administered once every ten weeks. In some embodiments, the PD-1 inhibitor is administered once every nine weeks. In some embodiments, the PD-1 inhibitor is administered once every eight weeks. In some embodiments, the PD-1 inhibitor is administered once every seven weeks. In some embodiments, the PD-1 inhibitor is administered once every six weeks. In some embodiments, the PD-1 inhibitor is administered once every five weeks. In some embodiments, the PD-1 inhibitor is administered once every four weeks. In some embodiments, the PD-1 inhibitor is administered once every three weeks. In some embodiments, the PD-1 inhibitor is administered once every two weeks. In some embodiments, the PD-1 inhibitor is administered once every week.
- the PD-1 inhibitor is administered once every nine weeks.
- the PD-1 inhibitor e.g., tislelizumab
- the PD-1 inhibitor is administered intravenously.
- the PD-1 inhibitor (e.g., tislelizumab) is administered over a period of about 20 minutes to 40 minutes (e.g., about 30 minutes). In some embodiments, the PD- 1 inhibitor is administered over a period of about 30 minutes. In some embodiments, the PD-1 inhibitor is administered over a period of about an hour. In some embodiments, the PD-1 inhibitor is administered over a period of about two hours. In some embodiments, the PD-1 inhibitor is administered over a period of about three hours. In some embodiments, the PD-1 inhibitor is administered over a period of about four hours. In some embodiments, the PD-1 inhibitor is administered over a period of about five hours. In some embodiments, the PD-1 inhibitor is administered over a period of about six hours.
- the PD-1 inhibitor e.g., tislelizumab
- the PD-1 inhibitor is administered over a period of about 20 minutes to 40 minutes (e.g., about 30 minutes). In some embodiments, the PD- 1 inhibitor is
- the PD-1 inhibitor (e.g., tislelizumab) is administered at a dose between about 300 mg to about 500 mg (e.g., about 400 mg), intravenously, once every four weeks. In some embodiments, the PD-1 inhibitor is administered at a dose between about 200 mg to about 400 mg (e.g., about 300 mg), intravenously, once every three weeks. In some embodiments, tislelizumab is administered at a dose of 400 mg, once every four weeks. In some embodiments, tislelizumab is administered at a dose of 300 mg, once every three weeks.
- the PD-1 inhibitor (e.g., tislelizumab) is administered at a dose between about 300 mg to about 500 mg (e.g., about 400 mg), intravenously, over a period of about 20 minutes to about 40 minutes (e.g., about 30 minutes), once every two weeks.
- the PD-1 inhibitor is administered at a dose between about 200 mg to about 400 mg (e g., about 300 mg), intravenously, over a period of about 20 minutes to about 40 minutes (e.g., about 30 minutes), once every three weeks.
- the PD-1 inhibitor (e.g., tislelizumab) is administered at a dose of about 100 mg per week. For example, if a 10-week dose is given to a patient, then the PD-1 inhibitor (e.g., tislelizumab) can be given at 1000 mg. If a 9-week dose is given, then the PD-1 inhibitor (e.g., tislelizumab) can be given at 900 mg. If an 8-week dose is given, then the PD-1 inhibitor (e.g., tislelizumab) can be given at 800 mg.
- a 10-week dose is given to a patient, then the PD-1 inhibitor (e.g., tislelizumab) can be given at 1000 mg. If a 9-week dose is given, then the PD-1 inhibitor (e.g., tislelizumab) can be given at 900 mg. If an 8-week dose is given, then the PD-1 inhibitor (e.g
- the PD-1 inhibitor e.g., tislelizumab
- the PD-1 inhibitor can be given at 700 mg.
- a 6-week dose is given, then the PD-1 inhibitor (e.g., tislelizumab) can be given at 600 mg.
- a 5-week dose is given, then the PD-1 inhibitor (e.g., tislelizumab) can be given at 500 mg.
- a 4-week dose is given, then the PD-1 inhibitor (e.g., tislelizumab) can be given at 400 mg.
- the PD-1 inhibitor e.g., tislelizumab
- the PD-1 inhibitor can be given at 300 mg.
- the PD-1 inhibitor e.g., tislelizumab
- the PD-1 inhibitor can be given at 200 mg.
- the PD-1 inhibitor e.g., tislelizumab
- the PD-1 inhibitor can be given at 100 mg.
- an anti-PD-1 antibody such as tislelizumab
- it can be administered at a dose of 200 mg as an intravenous infusion, once every three weeks.
- tislelizumab can be administered at a dose of 300 mg as an intravenous infusion, once every four weeks.
- an anti-PD-1 antibody such as tislelizumab
- it can be administered at a dose of 300 mg as an intravenous infusion, once every three weeks.
- tislelizumab can be administered at a dose of 400 mg as an intravenous infusion, once every four weeks.
- the invention provides a product comprising a compound of the present invention and at least one other therapeutic agent as a combined preparation for simultaneous, separate or sequential use in therapy.
- the therapy is the treatment of a disease or condition mediated by WRN.
- Products provided as a combined preparation include a composition comprising the compound of Formula I and the other therapeutic agent(s) together in the same pharmaceutical composition, or the compound of the present invention and the other therapeutic agent(s) in separate form, e.g. in the form of a kit.
- the invention provides a kit comprising two or more separate pharmaceutical compositions, at least one of which contains a compound of the present invention.
- the kit comprises means for separately retaining said compositions, such as a container, divided bottle, or divided foil packet.
- a container, divided bottle, or divided foil packet An example of such a kit is a blister pack, as typically used for the packaging of tablets, capsules and the like.
- the kit of the invention may be used for administering different dosage forms, for example, oral and parenteral, for administering the separate compositions at different dosage intervals, or for titrating the separate compositions against one another.
- the kit of the invention typically comprises directions for administration.
- the compound of the present invention and the other therapeutic agent may be manufactured and/or formulated by the same or different manufacturers. Moreover, the compound of the present invention and the other therapeutic may be brought together into a combination therapy: (i) prior to release of the combination product to physicians (e g. in the case of a kit comprising the compound of the present invention and the other therapeutic agent); (ii) by the physician themselves (or under the guidance of the physician) shortly before administration; (iii) in the patient themselves, e.g. during sequential administration of the compound of the present invention and the other therapeutic agent.
- the invention provides the use of a compound of the present invention for treating a disease or condition mediated by WRN, wherein the medicament is prepared for administration with another therapeutic agent.
- the invention also provides the use of another therapeutic agent for treating a disease or condition mediated by WRN, wherein the medicament is administered with a compound of the present invention.
- the invention also provides a compound of the present invention for use in treating a disease or condition mediated by WRN, wherein the compound of the present invention is prepared for administration with another therapeutic agent.
- the invention also provides another therapeutic agent for use in treating a disease or condition mediated by WRN, wherein the other therapeutic agent is prepared for administration with a compound of the present invention.
- the invention also provides a compound of the present invention for use in treating a disease or condition mediated by WRN, wherein the compound of the present invention is administered with another therapeutic agent.
- the invention also provides another therapeutic agent for use in a method of treating a disease or condition mediated by WRN, wherein the other therapeutic agent is administered with a compound of the present invention.
- the invention also provides the use of a compound of the present invention for treating a disease or condition mediated by WRN, wherein the patient has previously (e.g. within 24 hours) been treated with another therapeutic agent.
- the invention also provides the use of another therapeutic agent for treating a disease or condition mediated by WRN, wherein the patient has previously (e.g. within 24 hours) been treated with compound of the present invention.
- the appropriate halothiazole may undergo nucleophilic aromatic substitution with a protected amine as shown in Step 1.
- Bicyclic heterocycle F is produced via intramolecular condensation reaction of E.
- Protecting group chemistry produces G then the R 2 substituent addition to the compound is initiated by reacting G with tert-butyl 2-bromoacetate to produce Intermediate-7.
- R 1 is then attached using a boronic acid thereof by cross-coupling Intermediate-7 with said boronic acid to produce H.
- Deprotections followed by amide forming reactions may be used to finish adding substituents R 4 and R 2 to the compound as shown in Steps 9-13, to arrive at compounds of the disclosure such as 1-2.
- ISfeSCU sodium sulfate
- EDCI N-(3-dimethylaminopropyl)-N’ -ethylcarbodiimide hydrochloride
- LCMS Liquid chromatography–mass spectrometry
- HPLC High-performance liquid chromatography
- NH4Cl ammonium chloride
- K 3 PO 4 tripotassium phosphate
- TFA trifluoroacetic acid
- NBS N-bromosuccinimide
- DIEA N,N-diisopropylethylamine
- TsOH 4-methylbenzenesulfonic acid
- DMSO-d 6 deuterated dimethyl sulfoxide
- NMP N-methyl-2-pyrrolidone
- DBU 1,8-diazabicyclo[5.4.0]und
- Step 1 Synthesis of 4-chloro-5-methoxy-6-methylpyrimidine [00320] To a mixture of 4,6-dichloro-5-methoxypyrimidine (30.00 g, 167.6 mmol, 1.0 eq) in THF (300 mL) was added a 3 M solution of MeMgBr (61.45 mL, 184.4 mmol, 1.1 eq) in diethyl ether dropwise at 0 °C and then the mixture was stirred at 5 °C for 1 h. The resulting mixture was poured into H 2 O (200 mL) and extracted with EtOAc (100 mL*3).
- Step 2 Synthesis of methyl 5-methoxy-6-methylpyrimidine-4-carboxylate [00321] To a mixture of 4-chloro-5-methoxy-6-methylpyrimidine (22.00 g, 138.7 mmol, 1.0 eq) in MeOH (250 mL) was added Pd(dppf)Cl 2 -CH 2 Cl 2 (6.80 g, 8.32 mmol, 0.06 eq) and TEA (28.1 g, 278 mmol, 2.0 eq). The reaction was purged with CO (50 psi) and stirred at 50 °C overnight. The resulting mixture was filtered, and the filtrate was concentrated under reduced pressure.
- Step 3 Synthesis of 5-hydroxy-6-methylpyrimidine-4-carboxylic acid [00322] A mixture of methyl 5-methoxy-6-methylpyrimidine-4-carboxylate (16.00 g, 87.83 mmol, 1.0 eq) in HBr solution (aq.) (68.5 mL, 68%) was stirred at 50 °C overnight. Then HI solution (aq.) (67.2 mL, 56%) was added and stirred at 50 °C for 6 h.
- Step 1 Synthesis of 2-methoxypyridine-4-carbothioamide
- 2-methoxypyridine-4-carbothioamide To a solution of 2-methoxyisonicotinonitrile (15.10 g, 112.6 mmol, 1.0 eq) in DMF (400 mL) was added MgCl2 (10.72 g, 112.6 mmol, 1.0 eq) and the mixture was stirred at room temperature for 15 mins. NaHS (25.02 g, 337.7 mmol, 3.0 eq) was added to the mixture and the resulting mixture was stirred at room temperature for 14 h. The reaction mixture was poured into water (1.5 L) and extracted with EtOAc (1 L * 3).
- Step 2 Synthesis of ethyl 4-hydroxy-2-(2-methoxypyridin-4-yl)thiazole-5-carboxylate [00327] To a solution of 2-methoxypyridine-4-carbothioamide (20.00 g, 118.9 mmol, 1.0 eq) in EtOH (400 mL) was added pyridine (28.21 g, 356.7 mmol, 3.0 eq) and diethyl 2-bromomalonate (28.42 g, 118.9 mmol, 1.0 eq), and then the mixture was stirred at 80 °C for 1 h. The reaction mixture was cooled to room temperature and then filtered.
- Step 3 Synthesis of ethyl 2-(2-methoxypyridin-4-yl)-4- (((trifluoromethyl)sulfonyl)oxy)thiazole-5-carboxylate [00330] To a solution of ethyl 4-hydroxy-2-(2-methoxypyridin-4-yl)thiazole-5-carboxylate (16.62 g, 59.28 mmol, 1.0 eq) and pyridine (14.07 g, 177.8 mmol, 3.0 eq) in DCM (100 mL) was added Tf2O (25.09 g, 88.92 mmol, 1.5 eq) at 0 °C.
- Step 4 Synthesis of ethyl 4-(bis(4-methoxybenzyl)amino)-2-(2-methoxypyridin-4- yl)thiazole-5-carboxylate
- ethyl 2-(2-methoxypyridin-4-yl)-4- (((trifluoromethyl)sulfonyl)oxy)thiazole-5-carboxylate 25.00 g, 60.63 mmol, 1.0 eq
- bis(4- methoxybenzyl)amine 23.40 g, 90.94 mmol, 1.5 eq
- 1,4-dioxane 400 mL
- DIEA 23.51 g, 181.9 mmol, 3.0 eq
- Step 5 Synthesis of 1-(4-(bis(4-methoxybenzyl)amino)-2-(2-methoxypyridin-4- yl)thiazol-5-yl)pentane-1,3-dione
- ethyl 4-(bis(4-methoxybenzyl)amino)-2-(2-methoxypyridin-4- yl)thiazole-5-carboxylate 500 mg, 962 ⁇ mol, 1.0 eq
- butan-2-one (278 mg, 3.85 mmol, 4.0 eq) in 2-methyltetrahydrofuran (4 mL) was added LiHMDS (1 M in THF, 3.85 mL, 4.0 eq) at room temperature and then the mixture was stirred at 80°C for 2 h.
- Step 6 Synthesis of 1-(4-(bis(4-methoxybenzyl)amino)-2-(2-methoxypyridin-4- yl)thiazol-5-yl)-2-bromopentane-1,3-dione
- 1-(4-(bis(4-methoxybenzyl)amino)-2-(2-methoxypyridin-4-yl)thiazol- 5-yl)pentane-1,3-dione (2.47 g, 4.53 mmol, 1.0 eq) in DCM (25 mL) was added TsOH-H2O (78 mg, 453 ⁇ mol, 0.1 eq) and NBS (806 mg, 4.53 mmol, 1.0 eq) and then the mixture was stirred at room temperature for 1 h.
- Step 2 Synthesis of 5-ethyl-2-(2-methoxypyridin-4-yl)-6-(piperazin-1-yl)thiazolo[4,5- b]pyridin-7(4H)-one trifluoroacetate
- a solution of tert-butyl 4-(1-(4-(bis(4-methoxybenzyl)amino)-2-(2-methoxypyridin-4- yl)thiazol-5-yl)-1,3-dioxopentan-2-yl)piperazine-1-carboxylate (1.00 g, 1.37 mmol, 1.0 eq) in TFA (10 mL) was stirred at 70 °C for 4 h, and then the reaction mixture was concentrated in vacuo to afford the title compound, which was used into the next step without further purification.
- Step 1 Synthesis of tert-butyl 4-(4-(2-((2-chloro-4-(trifluoromethyl)phenyl)amino)-2- oxoethyl)-5-ethyl-2-(2-methoxypyridin-4-yl)-7-oxo-4,7-dihydrothiazolo[4,5-b]pyridin-6- yl)piperazine-1-carboxylate [00351] To a solution of tert-butyl 4-(5-ethyl-2-(2-methoxypyridin-4-yl)-7-oxo-4,7- dihydrothiazolo[4,5-b]pyridin-6-yl)piperazine-1-carboxylate (Intermediate-9) (285 mg, 604 ⁇ mol, 1.0 eq) in DMF (10 mL) was added DIEA (234 mg, 1.81 mmol, 3.0 eq) and N-(2-chloro
- Step 2 Synthesis of N-(2-chloro-4-(trifluoromethyl)phenyl)-2-(5-ethyl-2-(2- methoxypyridin-4-yl)-7-oxo-6-(piperazin-1-yl)thiazolo[4,5-b]pyridin-4(7H)-yl)acetamide hydrochloride [00354] To a solution of tert-butyl 4-(4-(2-((2-chloro-4-(trifluoromethyl)phenyl)amino)-2- oxoethyl)-5-ethyl-2-(2-methoxypyridin-4-yl)-7-oxo-4,7-dihydrothiazolo[4,5-b]pyridin-6- yl)piperazine-1-carboxylate (180 mg, 255 ⁇ mol, 1.0 eq) in 1,4-dioxane (2.7 mL) was added a solution of
- Step 2 Synthesis of 2-(2-bromo-6-(4-(tert-butoxycarbonyl)piperazin-1-yl)-5-ethyl-7- oxothiazolo[5,4-b]pyridin-4(7H)-yl)acetic acid
- Step 3 Synthesis of tert-butyl 4-(2-bromo-4-(2-((2-chloro-4- (trifluoromethyl)phenyl)amino)-2-oxoethyl)-5-ethyl-7-oxo-4,7-dihydrothiazolo[5,4-b]pyridin-6- yl)piperazine-1-carboxylate [00364] To a solution of 2-(2-bromo-6-(4-(tert-butoxycarbonyl)piperazin-1-yl)-5-ethyl-7- oxothiazolo[5,4-b]pyridin-4(7H)-yl)acetic acid (2.50 g, 4.99 mmol, 1.0 eq) and 2-chloro-4- (trifluoromethyl)aniline (1.02 g, 5.24 mmol, 1.05 eq) in pyridine (25 mL) and DCM (25 mL)
- Step 4 Synthesis of 2-(2-bromo-5-ethyl-7-oxo-6-(piperazin-1-yl)thiazolo[5,4- b]pyridin-4(7H)-yl)-N-(2-chloro-4-(trifluoromethyl)phenyl)acetamide trifluoroacetate [00367] To a solution of tert-butyl 4-(2-bromo-4-(2-((2-chloro-4- (trifluoromethyl)phenyl)amino)-2-oxoethyl)-5-ethyl-7-oxo-4,7-dihydrothiazolo[5,4-b]pyridin-6- yl)piperazine-1-carboxylate (50 mg, 74 ⁇ mol, 1.0 eq) in DCM (1.5 mL) was added TFA (0.5 mL), and the resulting mixture was stirred at room temperature for 0.5 h.
- the resulting mixture was diluted with water (20 mL).
- the resulting mixture was extracted with EtOAc (3 * 20 mL).
- the combined organic layers were washed with brine (20 mL), dried over anhydrous Na2SO4. After filtration, the filtrate was concentrated under reduced pressure to afford the title compound, which was used into the next step directly without purification.
- Step 1 Synthesis of tert-butyl 4-(5-ethyl-2-(2-methoxypyridin-4-yl)-7-oxo-4-(2-oxo- 2-((3-(trifluoromethyl)bicyclo[l .1. l]pentan-l-yl)amino)ethyl)-4,7-dihydrothiazolo[4,5- b]pyridin-6-yl)piperazine- 1 -carboxylate
- Bicine buffer solution was purchased from Alfa Aesar (Tewksbury, MA) and compound NSC-617145 was purchased from Tocris (Minneapolis, MN).
- DNA duplex was synthesized at BGI (Shenzhen, China) and was composed of strand 1 with the sequence 5’-GCACTGGCCGTCGTTTTACGGTCG-3’ (SEQ ID NO.: 1) and strand 2 with the sequence 5’-TCCAAGTAAAACGACGGCCAGTGC-3’ (SEQ ID NO.: 2). DNA strands were annealed by heating to 95 o C for 5 minutes followed by slow cooling to room temperature.
- DMSO concentration was 1% and the reference compound concentration (NSC-617145) used for the minimal signal control was 20 ⁇ M.
- the reaction was stopped by the addition of the ADP-Glo Kit components (Promega; Madison, WI) as directed and the relative luminescence units (RLU) were read on an Envision 2104 (Perkin Elmer; Waltham, MA).
- WRN protein production [00392] Molecular Biology and virus production. The DNA encoding human Werner helicase (Uniprot Q14191, amino acids 517-1235 with L1074F point mutation) was generated with codon- optimization for E.coli expression and subcloned into the pFastBac vector with a TEV cleavable 8xHis tag (WRN-BV08).
- Sf9 cells grown in SF900II media were infected with 1.200 WRN-BV08 P2 virus and incubated for protein expression for 72 h at 27°C.
- the WRN protein was purified using the following protocol.
- the cell pellets were thawed and resuspended in buffer A (50 mM Tris, pH 7.5, 500 mM NaCl, 1 mM TCEP, 10% Glycerol) supplemented with 0.5% CHAPS, ImM PMSF, Ipg/ml Leupeptin, Ipg/ml Pepstatin, and the Pierce Universal Nuclease and cocktail tablet.
- Untagged WRN protein was eluted from the column with buffer A supplemented with 20 mM imidazole, dialyzed overnight into buffer B (50 mM Tris, pH 7.5, 1 mM TCEP, 10% Glycerol) supplemented with 150 mM NaCl and loaded onto a Heparin column. Proteins were eluted with a step gradient of buffer B supplemented with 150 mM, 200 mM, 300 mM and 500 mM NaCl.
- WRN containing fractions were pooled and concentrated prior to loading on to size exclusion chromatography using a HiLoad 16/600 Superdex TM 200 pg column (GE Healthcare) in buffer C (20 mM HEPES, pH 7.5, 250 mM NaCl, 0.25 mM TCEP, 2.5% Glycerol).
- Example 3 Method for determining effect on p21 induction in cells.
- the colon carcinoma cell line HCT116 was obtained from ATCC and cultured in growth medium consisting of Mccoy's 5A Medium (Gibco 16600108) supplemented with 10% FBS (Transgene FS201-02) and 100 units/mL penicillin-streptomycin (Gibco 15140122) and maintained at 37 °C under 5% CO2.
- 2,000 cells in 30pL of culture media were plated per well to Poly-D-Lysine 384 Well Black Clear Plates (Biocoat 356663) and incubated overnight at 37 °C under 5% CO2.
- compounds were serially diluted in DMSO for a total of 11 test concentrations.
- the typical starting concentration of cpds was lOuM with 2-fold dilutions.
- 150nL of diluted compound was added in duplicate to the assay plate, using an Echo 655 (Labcyte).
- the plate was centrifuged at 500 RPM for 1 min and then incubated at 37 °C under 5% CO2 for 24h. After 24h, medium was removed, and cells were fixed by adding 40pL of 4% paraformaldehyde solution to each well and incubated for 20 min at room temperature.
- the plate was then washed 4 times with lOOpL per well of wash buffer (PBS with 0.1% Tween- 20) using a microplate washer.
- Each plate contained DMSO control (low control) and an internal reference WRN inhibitor (high control) respectively.
- WRN inhibitor high control
- the 800nm/700nm ratio was calculated for each well to give fold p21 induction and then percent activation for each compound well was calculated as follows ((100 x (ratio cpd well-ratio low control)/(ratio high control - ratio low control)).
- EC50 values for each compound was generated after non-linear regression curve fitting using commercially available software.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Nitrogen And Oxygen Or Sulfur-Condensed Heterocyclic Ring Systems (AREA)
Abstract
Description
Claims
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202480044318.1A CN121419979A (en) | 2023-06-13 | 2024-06-13 | Thiazolidine-7(4H)-one WRN inhibitor |
Applications Claiming Priority (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202363507932P | 2023-06-13 | 2023-06-13 | |
| US63/507,932 | 2023-06-13 | ||
| US202363519756P | 2023-08-15 | 2023-08-15 | |
| US63/519,756 | 2023-08-15 | ||
| US202363613655P | 2023-12-21 | 2023-12-21 | |
| US63/613,655 | 2023-12-21 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2024259048A2 true WO2024259048A2 (en) | 2024-12-19 |
| WO2024259048A3 WO2024259048A3 (en) | 2025-02-27 |
Family
ID=93852894
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2024/033732 Pending WO2024259048A2 (en) | 2023-06-13 | 2024-06-13 | Thiazolopyridin-7(4h)-one wrn inhibitors |
Country Status (2)
| Country | Link |
|---|---|
| CN (1) | CN121419979A (en) |
| WO (1) | WO2024259048A2 (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US12344609B2 (en) | 2023-06-08 | 2025-07-01 | Nimbus Wadjet, Inc. | WRN inhibitors |
| WO2025215527A2 (en) | 2024-04-10 | 2025-10-16 | Novartis Ag | Pharmaceutical combinations and uses thereof |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102816175B (en) * | 2011-06-09 | 2015-12-16 | 上海汇伦生命科技有限公司 | A kind of heterocycle pyridine compounds, its intermediate, preparation method and purposes |
| WO2018229683A1 (en) * | 2017-06-15 | 2018-12-20 | Novartis Ag | 5,6-fused-bicyclic compounds and compositions for the treatment of parasitic diseases |
| CN117425657A (en) * | 2021-05-26 | 2024-01-19 | 诺华股份有限公司 | Triazolo-pyrimidine analogues for the treatment of disorders associated with Werner syndrome RECQ helicase (WRN) inhibition |
| CN120225197A (en) * | 2022-11-18 | 2025-06-27 | 诺华股份有限公司 | Drug combinations and their uses |
| JP2025540756A (en) * | 2022-12-05 | 2025-12-16 | 江蘇恒瑞医薬股▲ふん▼有限公司 | Triazole compounds, their preparation methods and medicinal uses |
-
2024
- 2024-06-13 WO PCT/US2024/033732 patent/WO2024259048A2/en active Pending
- 2024-06-13 CN CN202480044318.1A patent/CN121419979A/en active Pending
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US12344609B2 (en) | 2023-06-08 | 2025-07-01 | Nimbus Wadjet, Inc. | WRN inhibitors |
| US12421233B2 (en) | 2023-06-08 | 2025-09-23 | Nimbus Wadjet, Inc. | WRN inhibitors |
| US12528806B2 (en) | 2023-06-08 | 2026-01-20 | Nimbus Wadjet, Inc. | WRN inhibitors |
| WO2025215527A2 (en) | 2024-04-10 | 2025-10-16 | Novartis Ag | Pharmaceutical combinations and uses thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2024259048A3 (en) | 2025-02-27 |
| CN121419979A (en) | 2026-01-27 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP7720840B2 (en) | Tricyclic heterocycles as FGFR inhibitors | |
| WO2023287896A1 (en) | Tricyclic compounds as inhibitors of kras | |
| RU2665462C2 (en) | 5-azaindazole compounds and methods for use thereof | |
| AU2013225533B2 (en) | Amido spirocyclic amide and sulfonamide derivatives | |
| WO2024015731A1 (en) | Fused tricyclic compounds as inhibitors of kras g12v mutants | |
| KR20220100879A (en) | Bicyclic heterocycles as FGFR inhibitors | |
| WO2025014877A2 (en) | Triazolo wrn inhibitors | |
| WO2023122140A1 (en) | Parp1 inhibitors | |
| WO2024259048A2 (en) | Thiazolopyridin-7(4h)-one wrn inhibitors | |
| WO2024220887A1 (en) | [1,2,4]triazolo[1,5-a]pyrimidinone wrn inhibitors | |
| AU2024286830A1 (en) | Wrn inhibitors | |
| WO2024254602A1 (en) | Oxazolopyridin-7(4h)-one wrn inhibitors | |
| WO2024215923A2 (en) | Cyclic vinyl sulfone wrn inhibitors | |
| WO2025250620A1 (en) | Tricyclic fused wrn inhibitors | |
| WO2026015855A1 (en) | Spirocyclic wrn inhibitors | |
| TW202604522A (en) | Tricyclic fused wrn inhibitors | |
| WO2025137599A1 (en) | Wrn inhibitors | |
| WO2026060143A1 (en) | Kras inhibitors | |
| CN118696036A (en) | PARP1 inhibitors | |
| EA051810B1 (en) | QUINOLINE COMPOUNDS AS KRAS INHIBITORS | |
| HK1227874A1 (en) | Tricyclic pi3k inhibitor compounds and methods of use | |
| HK1227874A (en) | Tricyclic pi3k inhibitor compounds and methods of use |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24824123 Country of ref document: EP Kind code of ref document: A2 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2024824123 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2024824123 Country of ref document: EP Effective date: 20260113 |
|
| ENP | Entry into the national phase |
Ref document number: 2024824123 Country of ref document: EP Effective date: 20260113 |
|
| ENP | Entry into the national phase |
Ref document number: 2024824123 Country of ref document: EP Effective date: 20260113 |
|
| ENP | Entry into the national phase |
Ref document number: 2024824123 Country of ref document: EP Effective date: 20260113 |