WO2025128564A1 - Salt forms of a glp-1 receptor modulator - Google Patents
Salt forms of a glp-1 receptor modulator Download PDFInfo
- Publication number
- WO2025128564A1 WO2025128564A1 PCT/US2024/059371 US2024059371W WO2025128564A1 WO 2025128564 A1 WO2025128564 A1 WO 2025128564A1 US 2024059371 W US2024059371 W US 2024059371W WO 2025128564 A1 WO2025128564 A1 WO 2025128564A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- compound
- salt form
- salt
- xrpd pattern
- erbumine
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/14—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing three or more hetero rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
Definitions
- FIG. 30 are TGA and DSC thermograms of Compound 1 erbumine salt Form D.
- FIG. 34 is a 1 H NMR spectrum of Compound 1 erbumine salt Form E.
- Compound 1 sodium salt Form B is characterized by all of the following:
- Compound 1 sodium salt Form B is characterized by (1) an XRPD pattern substantially in accordance with FIG. 12 and at least one of the following:
- Compound 1 sodium salt Form B is characterized by (1) an XRPD pattern substantially in accordance with FIG. 12 and:
- Compound 1 sodium salt Form D is characterized by an XRPD pattern on a 2 Theta scale, ⁇ 0.20, comprising three or more peaks selected from 5.55, 11.08, 12.23, 12.47, 12.84, 13.05, 13.88, 14.10, 14.73, 15.56, 16.47, 17.09, 17.85, 18.29, 18.63,
- Compound 1 sodium salt Form D is characterized by an XRPD pattern on a 2 Theta scale, ⁇ 0.20, comprising five or more peaks selected from 5.55, 11.08, 12.23, 12.47, 12.84, 13.05, 13.88, 14.10, 14.73, 15.56, 16.47, 17.09, 17.85, 18.29, 18.63,
- Compound 1 sodium salt Form D is characterized by (1) an
- 1 erbumine salt is characterized as Compound 1 erbumine salt Form A (1 : 1.0) (stoichiometric ratio of Compound 1 : erbumine).
- Compound 1 erbumine salt Form A is characterized by an
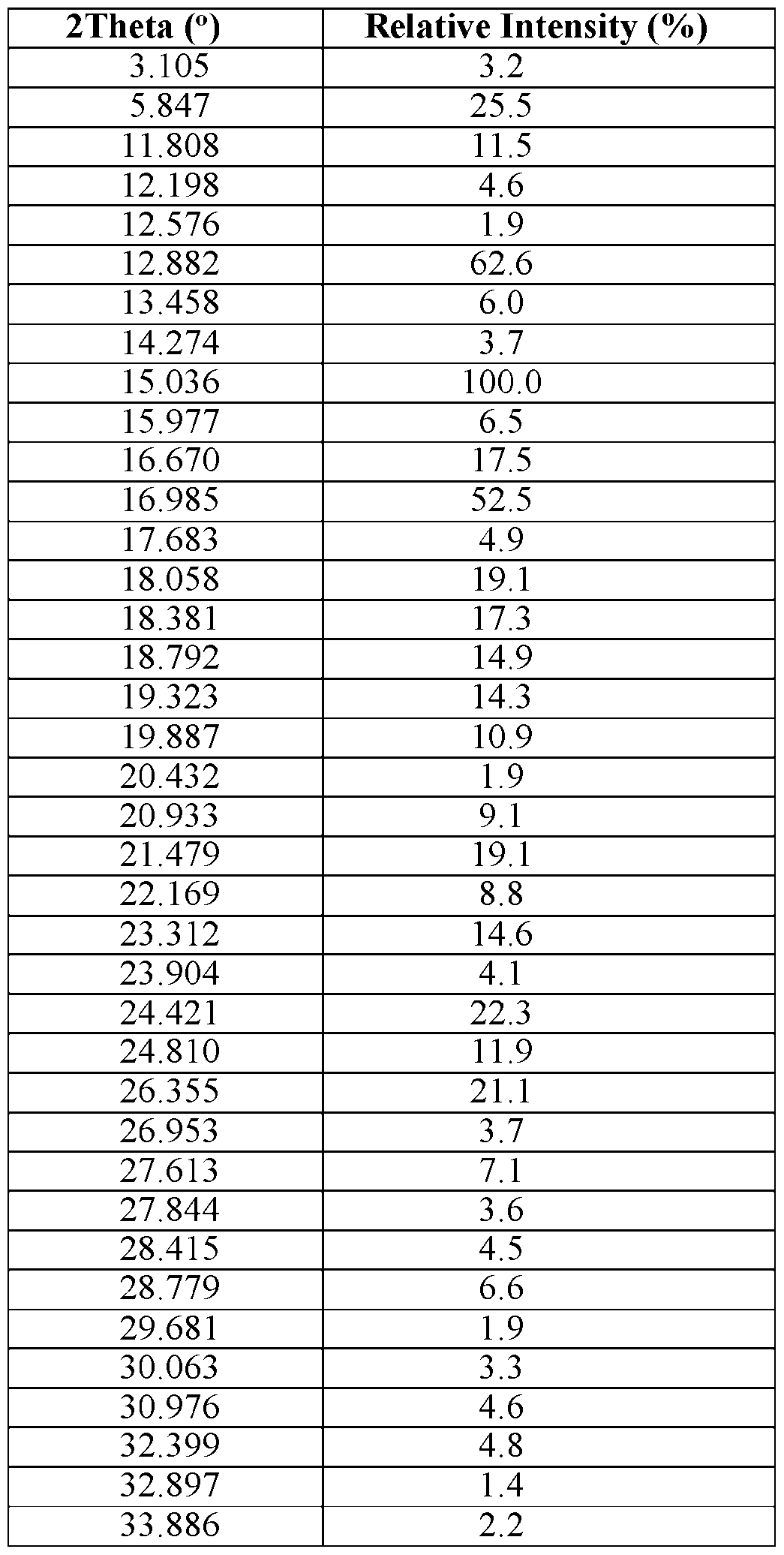
- Compound 1 erbumine salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ⁇ 0.20, comprising peaks at 9.46, 10.26, 11.90, 19.40, and 20.28.
- Compound 1 erbumine salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ⁇ 0.20, comprising all of the peaks 9.46, 10.26, 11.24, 11.90, 16.94, 17.40, 18.73, 19.40, 20.28, 20.95, and 22.12.
- Compound 1 erbumine salt Form A is characterized by the XRPD pattern substantially in accordance with FIG. 20.
- Compound 1 erbumine salt Form A is characterized by the DSC thermogram substantially in accordance with FIG. 21 indicating decomposition upon melting.
- Compound 1 erbumine salt Form A is characterized by the TGA thermogram substantially in accordance with FIG. 22.
- Compound 1 erbumine salt Form A is characterized by the ’l l NMR spectrum in d6-DMSO substantially in accordance with FIG. 23 indicating no detectable residual solvent.
- Compound 1 erbumine salt Form A is characterized by at least one of the following:
- Compound 1 erbumine salt Form A is characterized by at least two of the following:
- Compound 1 erbumine salt Form A is characterized by (1) an
- Compound 1 erbumine crystalline salt is characterized as Compound 1 erbumine salt Form B.
- the XRPD pattern of Compound 1 erbumine salt Form B was recorded in reflection geometry.
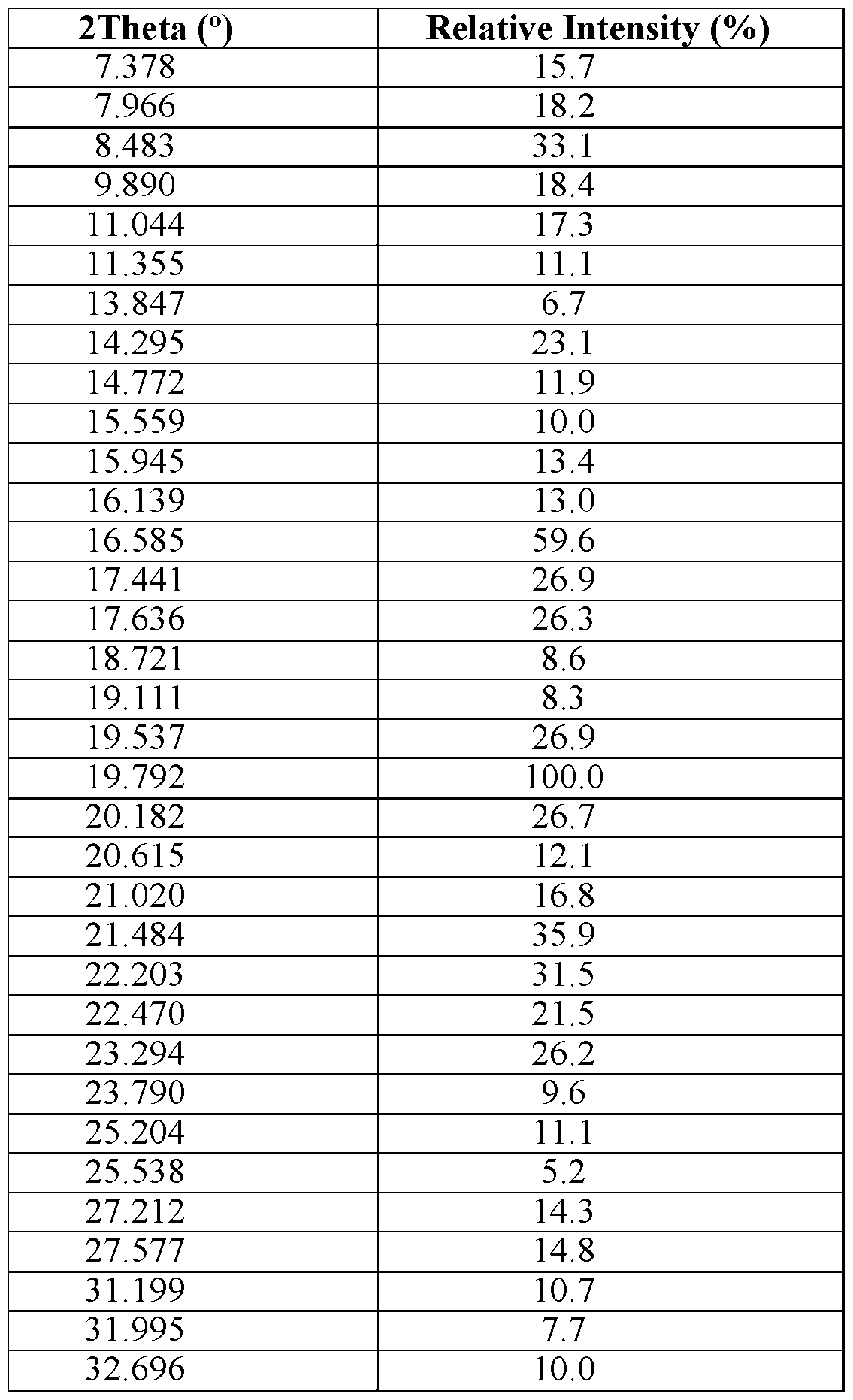
- Compound 1 erbumine salt Form B is characterized by an XRPD pattern on a 2 Theta scale, ⁇ 0.20, comprising five or more peaks selected from 4.74, 5.52, 7.39, 9.40, 9.84, 11.04, 12.35, 14.29, 15.09, 16.49, 17.76, 18.09, 19.26, 20.07, 20.57, 21.06,
- Compound 1 erbumine salt Form B is characterized by an XRPD pattern on a 2 Theta scale, ⁇ 0.20, comprising ten or more peaks selected from 4.74, 5.52, 7.39, 9.40, 9.84, 11.04, 12.35, 14.29, 15.09, 16.49, 17.76, 18.09, 19.26, 20.07, 20.57, 21.06,
- Compound 1 erbumine salt Form B is characterized by an XRPD pattern on a 2 Theta scale, ⁇ 0.20, comprising peaks at 9.84, 11.04, and 19.26.
- Compound 1 erbumine salt Form B is characterized by an XRPD pattern on a 2 Theta scale, ⁇ 0.20, comprising peaks at 9.84, 11.04, 19.26, 20.07, and 21.06.
- Compound 1 erbumine salt Form B is characterized by an XRPD pattern on a 2 Theta scale, ⁇ 0.20, comprising peaks at 5.52, 9.84, 11.04, 17.76, 18.09, 19.26, 20.07, 21.06, 22.14, and 23.72.
- Compound 1 erbumine salt Form B is characterized by an XRPD pattern on a 2 Theta scale, ⁇ 0.20, comprising all of the peaks 4.74, 5.52, 7.39, 9.40, 9.84, 11.04, 12.35, 14.29, 15.09, 16.49, 17.76, 18.09, 19.26, 20.07, 20.57, 21.06, 22.14, and 23.72.
- Compound 1 erbumine salt Form B is characterized by the XRPD pattern substantially in accordance with FIG. 24. [00233] A list of the peaks from the XRPD pattern for Compound 1 erbumine salt Form B is provided in Table 6 below.
- Compound 1 erbumine salt Form D is characterized by the TGA and DSC thermograms substantially in accordance with FIG. 30.
- Compound 1 erbumine salt Form D is characterized by the TGA and DSC thermograms showing endothermic peak onsets at about 116 °C - 120 °C and 174 °C - 178 °C.
- Compound 1 erbumine salt Form D is characterized by the TGA and DSC thermograms showing endothermic peak onsets at about 117.8 and about 176.3 °C.
- Compound 1 erbumine salt Form D is characterized by the 'HNMR spectrum in d6-DMSO substantially in accordance with FIG. 31 indicating no residual solvent.
- Compound 1 erbumine salt Form D is characterized by at least one of the following:
- Compound 1 erbumine salt Form D is characterized by at least one of the following:
- Compound 1 erbumine salt Form D is characterized by at least two of the following:
- Compound 1 erbumine salt Form D is characterized by (1) an XRPD pattern substantially in accordance with FIG. 29 and at least one of the following:
- Compound 1 erbumine salt Form D is characterized by (1) an XRPD pattern substantially in accordance with FIG. 29 and:
- Compound 1 erbumine crystalline salt is characterized as Compound 1 erbumine salt Form E.
- Compound 1 erbumine salt is characterized as Compound 1 erbumine salt Form E (1 :1.0) (stoichiometric ratio of Compound 1 : erbumine)
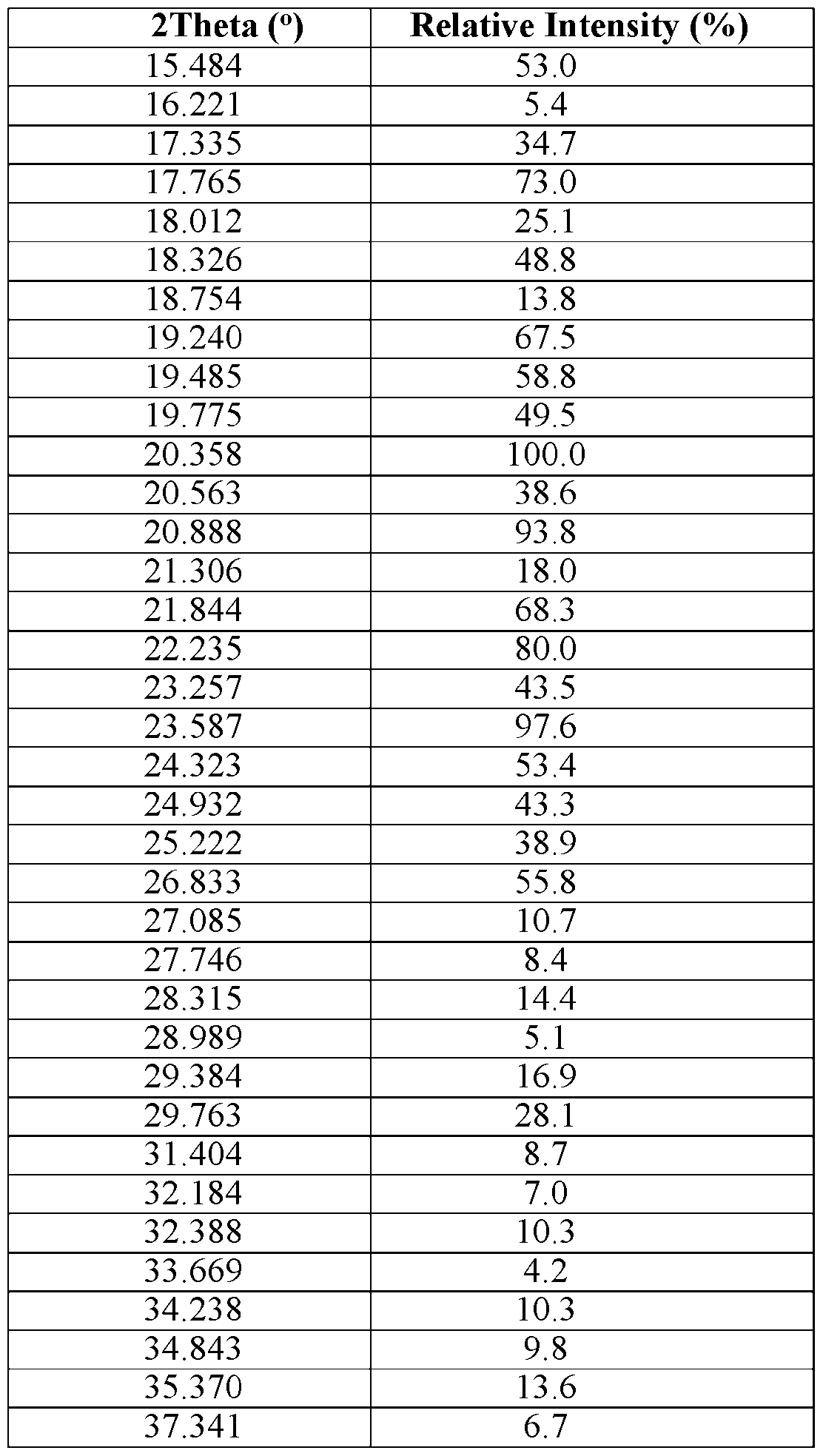
- Compound 1 erbumine salt Form E is characterized by an XRPD pattern on a 2 Theta scale, ⁇ 0.20, comprising one or more peaks selected from 5.85, 7.49, 8.43, 9.62, 10.43, 11.28, 11.68, 12.53, 13.25, 13.71, 14.82, 15.12, 15.51, 16.37, 17.26, 17.61, 18.20, 18.71, 19.31, 19.91, 20.84, 21.93, 22.37, 23.26, 24.13, 25.96, 27.22, and 30.28.
- Compound 1 erbumine salt Form E is characterized by an XRPD pattern on a 2 Theta scale, ⁇ 0.20, comprising three or more peaks selected from 5.85, 7.49, 8.43, 9.62, 10.43, 11.28, 11.68, 12.53, 13.25, 13.71, 14.82, 15.12, 15.51, 16.37, 17.26, 17.61, 18.20, 18.71, 19.31, 19.91, 20.84, 21.93, 22.37, 23.26, 24.13, 25.96, 27.22, and 30.28.
- Compound 1 erbumine salt Form E is characterized by an XRPD pattern on a 2 Theta scale, ⁇ 0.20, comprising five or more peaks selected from 5.85, 7.49, 8.43, 9.62, 10.43, 11.28, 11.68, 12.53, 13.25, 13.71, 14.82, 15.12, 15.51, 16.37, 17.26, 17.61, 18.20, 18.71, 19.31, 19.91, 20.84, 21.93, 22.37, 23.26, 24.13, 25.96, 27.22, and 30.28.
- Compound 1 erbumine salt Form E is characterized by an XRPD pattern on a 2 Theta scale, ⁇ 0.20, comprising ten or more peaks selected from 5.85, 7.49,
- Compound 1 erbumine salt Form E is characterized by an XRPD pattern on a 2 Theta scale, ⁇ 0.20, comprising peaks at 11.68, 18.71, and 19.31.
- Compound 1 erbumine salt Form E is characterized by an XRPD pattern on a 2 Theta scale, ⁇ 0.20, comprising peaks at 11.68, 18.20, 18.71, 19.31, and 19.91.
- Compound 1 erbumine salt Form E is characterized by an XRPD pattern on a 2 Theta scale, ⁇ 0.20, comprising peaks at 5.85, 11.68, 13.25, 18.20, 18.71, 19.31, 19.91, 23.26, 24.13, and 25.96.
- Compound 1 erbumine salt Form E is characterized by an XRPD pattern on a 2 Theta scale, ⁇ 0.20, comprising all of the peaks 5.85, 7.49, 8.43, 9.62,
- Compound 1 erbumine salt Form E is characterized by the XRPD pattern substantially in accordance with FIG. 32.
- Compound 1 erbumine salt Form E is characterized by the TGA and DSC thermograms substantially in accordance with FIG. 33.
- Compound 1 erbumine salt Form E is characterized by a DSC thermogram showing endothermic peak onsets at about 0.6 °C - 1.0 °C and about 159 °C - 163 °C.
- Compound 1 erbumine salt Form E is characterized by a TGA thermogram showing about 1.0 - 1.2 percent weight loss from 25 °C to 100 °C.
- Compound 1 erbumine salt Form E is characterized by the TGA and DSC thermograms showing endothermic peak onsets at about 0.8 °C and about 161 °C and showing about 1.1 percent weight loss from 25 °C to 100 °C.
- Compound 1 erbumine salt Form E is characterized by the T H NMR spectrum in d6-DMSO substantially in accordance with FIG. 34 indicating 0.4 percent residual solvent.
- Compound 1 erbumine salt Form E is characterized by at least one of the following:
- Compound 1 erbumine salt Form E is characterized by at least one of the following:
- Compound 1 erbumine salt Form E is characterized by at least two of the following:
- Compound 1 erbumine salt Form E is characterized by all of the following:
- Compound 1 erbumine salt Form E is characterized by (1) an XRPD pattern substantially in accordance with FIG. 32 and at least one of the following:
- Compound 1 erbumine salt Form E is characterized by (1) an XRPD pattern substantially in accordance with FIG. 32 and: (2) a DSC thermogram substantially in accordance with FIG. 33; and
- Compound 1 erbumine crystalline salt is characterized as Compound 1 erbumine salt Form F.
- Compound 1 erbumine salt is characterized as Compound 1 erbumine salt Form F (1 :1.0) (stoichiometric ratio of Compound 1 : erbumine).
- Compound 1 erbumine salt Form F is characterized by an XRPD pattern on a 2 Theta scale, ⁇ 0.20, comprising one or more peaks selected from 3.17, 5.12, 5.78, 6.47, 7.67, 8.91, 9.72, 10.46, 11.24, 11.96, 13.46, 14.41, 14.80, 15.70, 16.38, 17.11, 17.40, 18.06, 18.38, 18.80, 19.59, 20.63, 20.86, 21.16, 21.94, 22.79, 23.66, 24.580 26.17, and 26.82.
- Compound 1 erbumine salt Form F is characterized by an XRPD pattern on a 2 Theta scale, ⁇ 0.20, comprising three or more peaks selected from 3.17, 5.12, 5.78, 6.47, 7.67, 8.91, 9.72, 10.46, 11.24, 11.96, 13.46, 14.41, 14.80, 15.70, 16.38, 17.11, 17.40, 18.06, 18.38, 18.80, 19.59, 20.63, 20.86, 21.16, 21.94, 22.79, 23.66, 24.580 26.17, and
- Compound 1 erbumine salt Form F is characterized by an XRPD pattern on a 2 Theta scale, ⁇ 0.20, comprising five or more peaks selected from 3.17, 5.12, 5.78, 6.47, 7.67, 8.91, 9.72, 10.46, 11.24, 11.96, 13.46, 14.41, 14.80, 15.70, 16.38, 17.11, 17.40, 18.06, 18.38, 18.80, 19.59, 20.63, 20.86, 21.16, 21.94, 22.79, 23.66, 24.580 26.17, and
- Compound 1 erbumine salt Form F is characterized by an XRPD pattern on a 2 Theta scale, ⁇ 0.20, comprising ten or more peaks selected from 3.17, 5.12, 5.78, 6.47, 7.67, 8.91, 9.72, 10.46, 11.24, 11.96, 13.46, 14.41, 14.80, 15.70, 16.38, 17.11, 17.40, 18.06, 18.38, 18.80, 19.59, 20.63, 20.86, 21.16, 21.94, 22.79, 23.66, 24.580 26.17, and 26.82.
- Compound 1 erbumine salt Form F is characterized by an XRPD pattern on a 2 Theta scale, ⁇ 0.20, comprising peaks at 11.24, 18.38, and 20.86.
- Compound 1 erbumine salt Form F is characterized by an XRPD pattern on a 2 Theta scale, ⁇ 0.20, comprising peaks at 9.72, 11.24, 18.06, 18.38, and 20.86.
- Compound 1 erbumine salt Form E is characterized by an XRPD pattern on a 2 Theta scale, ⁇ 0.20, comprising peaks at 3.17, 9.72, 10.46, 11.24, 13.46, 18.06, 18.38, 18.80, 20.63, and 20.86.
- Compound 1 erbumine salt Form F is characterized by an XRPD pattern on a 2 Theta scale, ⁇ 0.20, comprising all of the peaks 3.17, 5.12, 5.78, 6.47, 7.67, 8.91, 9.72, 10.46, 11.24, 11.96, 13.46, 14.41, 14.80, 15.70, 16.38, 17.11, 17.40, 18.06, 18.38, 18.80, 19.59, 20.63, 20.86, 21.16, 21.94, 22.79, 23.66, 24.580 26.17, and 26.82.
- Compound 1 erbumine salt Form F is characterized by the XRPD pattern substantially in accordance with FIG. 35.
- Compound 1 erbumine salt Form F is characterized by the DSC and TGA thermograms substantially in accordance with FIG. 36.
- Compound 1 erbumine salt Form F is characterized by the DSC and TGA thermograms showing endothermic peak onsets about 36 °C - 41 °C, about 116 °C - 120 °C, and about 144 °C - 148 °C and showing about 1.0 - 1.2 percent weight loss from 30 °C to 80 °C and about 6.5 - 8.5 percent weight loss from 80 °C to 150 °C.
- Compound 1 erbumine salt Form F is characterized by the DSC and TGA thermograms showing endothermic peak onsets at about 37.9 °C, 117.7 °C, and 146.0 °C and showing about 1.1 percent weight loss from 30 °C to 80 °C and about 7.5 percent weight loss from 80 °C to 150 °C.
- Compound 1 erbumine salt Form F is characterized by the ’H NMR spectrum in d6-DMSO substantially in accordance with FIG. 37 indicating no residual solvent.
- Compound 1 erbumine salt Form F is characterized by at least one of the following:
- Compound 1 erbumine salt Form F is characterized by at least one of the following:
- Compound 1 erbumine salt Form F is characterized by at least two of the following:
- Compound 1 erbumine salt Form F is characterized by all of the following:
- Compound 1 erbumine salt Form F is characterized by (1) an XRPD pattern substantially in accordance with FIG. 35 and at least one of the following:
- Compound 1 erbumine salt Form F is characterized by (1) an XRPD pattern substantially in accordance with FIG. 35 and:
- the salt is Compound 1 L-arginine salt.
- Compound 1 L-arginine salt is amorphous.
- Compound 1 L-arginine salt is characterized by the XRPD pattern substantially in accordance with FIG. 38.
- the salt is Compound 1 L-lysine salt.
- Compound 1 L-lysine salt is crystalline.
- Compound 1 L-lysine crystalline salt is characterized as Compound 1 L-lysine salt Form A.
- Compound 1 L-lysine crystalline salt is characterized as Compound 1 L-lysine salt Form A (1:0.4) (stoichiometric ratio of Compound 1: lysine).
- Compound 1 L-lysine salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ⁇ 0.20, comprising one or more peaks selected from 9.68,
- Compound 1 L-lysine salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ⁇ 0.20, comprising three or more peaks selected from 9.68,
- Compound 1 L-lysine salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ⁇ 0.20, comprising five or more peaks selected from 9.68,
- Compound 1 L-lysine salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ⁇ 0.20, comprising peaks at 9.68, 19.38, and 26.03.
- Compound 1 L-lysine salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ⁇ 0.20, comprising peaks at 9.68, 17.71, 19.38, 22.83, and 26.03.
- Compound 1 L-lysine salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ⁇ 0.20, comprising all of the peaks 9.68, 13.32, 17.71, 17.96, 19.38, 20.47, 22.83, 23.83, 24.45, and 26.03.
- Compound 1 L-lysine salt is characterized by the XRPD pattern substantially in accordance with FIG. 39.
- administration can be, for example, orally, nasally, parenterally (intravenous, intramuscular, or subcutaneous), topically, transdermally, intravaginally, intravesically, intraci stemally, or rectally, in the form of solid, semi-solid, lyophilized powder, or liquid dosage forms, such as, for example, tablets, suppositories, pills, soft elastic and hard gelatin capsules, powders, solutions, suspensions, aerosols, and the like, preferably in unit dosage forms suitable for simple administration of precise dosages.
- compositions will include a conventional pharmaceutical excipient and an amorphous or crystalline form of the present disclosure as the/an active agent, and, in addition, may include other medicinal agents, pharmaceutical agents, excipients, adjuvants, and so on.
- Compositions of the disclosure may be used in combination with anticancer or other agents that are generally administered to a patient being treated for cancer.
- Adjuvants include preserving, wetting, suspending, sweetening, flavoring, perfuming, emulsifying, and dispensing agents. Prevention of the action of microorganisms can be ensured by various antibacterial and antifungal agents, for example, parabens, chlorobutanol, phenol, sorbic acid, and the like. It may also be desirable to include isotonic agents, for example sugars, sodium chloride, and the like. Prolonged absorption of the injectable pharmaceutical form can be brought about by the use of agents delaying absorption, for example, aluminum monostearate, and gelatin.
- a pharmaceutical composition of the disclosure may also contain minor amounts of auxiliary substances such as wetting or emulsifying agents, pH buffering agents, antioxidants, and the like, such as, for example, citric acid, sorbitan, monolaurate, triethanolamine oleate, butylated hydroxytoluene, and so on.
- auxiliary substances such as wetting or emulsifying agents, pH buffering agents, antioxidants, and the like, such as, for example, citric acid, sorbitan, monolaurate, triethanolamine oleate, butylated hydroxytoluene, and so on.
- compositions suitable for parenteral injection may comprise physiologically acceptable sterile aqueous or nonaqueous solutions, dispersions, suspensions or emulsions, and sterile powders for reconstitution into sterile injectable solutions or dispersions.
- suitable aqueous and nonaqueous excipients, diluents, solvents, or vehicles include water, ethanol, polyols (propyleneglycol, polyethyleneglycol, glycerol, and the like), suitable mixtures thereof, vegetable oils (such as olive oil), and injectable organic esters such as ethyl oleate.
- Proper fluidity can be maintained, for example, by the use of a coating such as lecithin, by the maintenance of the required particle size in the case of dispersions, and by the use of surfactants.
- One preferable route of administration is oral, using a convenient daily dosage regimen that can be adjusted according to the degree of severity of the disease-state to be treated.
- Solid dosage forms for oral administration include capsules, tablets, pills, powders, and granules.
- the active compound is admixed with at least one inert customary excipient such as sodium citrate or dicalcium phosphate or
- fillers or extenders as for example, starches, lactose, sucrose, glucose, mannitol, and silicic acid
- binders as for example, cellulose derivatives, starch, alignates, gelatin, polyvinylpyrrolidone, sucrose, and gum acacia
- humectants as for example, glycerol
- disintegrating agents as for example, agar- agar, calcium carbonate, potato or tapioca starch, alginic acid, croscarmellose sodium, complex silicates, and sodium carbonate
- solution retarders as for example paraffin
- absorption accelerators as for example, quaternary ammonium compounds
- wetting agents as for example, cetyl alcohol, and glycerol
- Solid dosage forms as described above can be prepared with coatings and shells, such as enteric coatings and others well known in the art. They may contain pacifying agents and can also be of such composition that they release the active compound or compounds in a certain part of the intestinal tract in a delayed manner. Examples of embedded compositions that can be used are polymeric substances and waxes. The active compounds can also be in microencapsulated form, if appropriate, with one or more of the above-mentioned excipients.
- Liquid dosage forms for oral administration include pharmaceutically acceptable emulsions, solutions, suspensions, syrups, and elixirs. Such dosage forms are prepared, for example, by dissolving, dispersing, and so on., crystalline forms of Compound 1, and optional pharmaceutical adjuvants in an excipient, such as, for example, water, saline, aqueous dextrose, glycerol, ethanol, and the like; solubilizing agents and emulsifiers, as for example, ethyl alcohol, isopropyl alcohol, ethyl carbonate, ethyl acetate, benzyl alcohol, benzyl benzoate, propyleneglycol, 1,3-butyleneglycol, and dimethylformamide; oils, in particular, cottonseed oil, groundnut oil, com germ oil, olive oil, castor oil, and sesame oil, glycerol, tetrahydrofurfuryl alcohol, polyethylenegly
- Suspensions in addition to the active compounds, may contain suspending agents, as for example, ethoxylated isostearyl alcohols, polyoxyethylene sorbitol, and sorbitan esters, microcrystalline cellulose, aluminum metahydroxide, bentonite, agar-agar, and tragacanth, or mixtures of these substances, and the like.
- suspending agents as for example, ethoxylated isostearyl alcohols, polyoxyethylene sorbitol, and sorbitan esters, microcrystalline cellulose, aluminum metahydroxide, bentonite, agar-agar, and tragacanth, or mixtures of these substances, and the like.
- compositions for rectal administrations are, for example, suppositories that can be prepared by mixing the crystalline forms of Compound 1 with for example suitable non-irritating excipients such as cocoa butter, polyethyleneglycol, or a suppository wax, which are solid at ordinary temperatures but liquid at body temperature and therefore melt while in a suitable body cavity and release the active component therein.
- suitable non-irritating excipients such as cocoa butter, polyethyleneglycol, or a suppository wax, which are solid at ordinary temperatures but liquid at body temperature and therefore melt while in a suitable body cavity and release the active component therein.
- Dosage forms for topical administration of a compound of this disclosure include ointments, powders, sprays, and inhalants.
- the active component is admixed under sterile conditions with a physiologically acceptable excipient and any preservatives, buffers, or propellants as may be required.
- Ophthalmic formulations, eye ointments, powders, and solutions are also contemplated as being within the scope of this disclosure.
- the pharmaceutically acceptable compositions will contain about 1% to about 99% by weight of a crystalline form of Compound 1, and 99% to 1% by weight of a suitable pharmaceutical excipient. In one example, the composition will be between about 5% and about 75% by weight of a crystalline form of Compound 1, with the rest being suitable pharmaceutical excipients.
- Actual methods of preparing such dosage forms are known, or will be apparent, to those skilled in this art; for example, see Remington's Pharmaceutical Sciences, 21 st Ed., (Lippincott, Williams and Wilkins Philadelphia, PA, 2006).
- the composition to be administered will, in any event, contain a therapeutically effective amount of a crystalline form of Compound 1 for treatment of a disease-state in accordance with the teachings of this disclosure.
- the crystalline forms of Compound 1 are administered in a therapeutically effective amount which will vary depending upon a variety of factors including the activity of Compound 1, the metabolic stability and length of action of Compound 1, the age, body weight, general health, sex, diet, mode, and time of administration, rate of excretion, drug combination, the severity of the particular disease-states, and the host undergoing therapy.
- the crystalline forms of Compound 1 can be administered to a patient at dosage levels in the range of about 0.1 to about 1,000 mg per day. For a normal human adult having a body weight of about 70 kilograms, a dosage in the range of about 0.01 to about 100 mg per kilogram of body weight per day is an example. The specific dosage used, however, can vary.
- the dosage can depend on a number of factors including the requirements of the patient, the severity of the condition being treated, and the pharmacological activity of the compound being used.
- the determination of optimum dosages for a particular patient is well known to one of ordinary skill in the art.
- the compounds provided herein can be formulated into pharmaceutical compositions using methods available in the art and those disclosed herein. Any of the solid forms provided herein can be provided in the appropriate pharmaceutical composition and be administered by a suitable route of administration.
- the methods provided herein encompass administering pharmaceutical compositions comprising at least one solid form provided herein and one or more compatible and pharmaceutically acceptable carriers.
- carrier includes a diluent, adjuvant (e.g., Freund’s adjuvant (complete and incomplete)), excipient, or vehicle with which the therapeutic is administered.
- Such pharmaceutical carriers can be sterile liquids, such as water and oils including petroleum, animal, vegetable, or oils of synthetic origin, such as peanut oil, soybean oil, mineral oil, sesame oil, and the like. Water can be used as a carrier when the pharmaceutical composition is administered intravenously. Saline solutions, aqueous dextrose, and glycerol solutions can also be employed as liquid carriers, particularly for injectable solutions. Examples of suitable pharmaceutical carriers are described in Martin, E.W., Remington 's Pharmaceutical Sciences.
- compositions, or compounds provided herein may be administered by any route known in the art.
- routes of administration include, but are not limited to, inhalation, intraarterial, intradermal, intramuscular, intraperitoneal, intravenous, nasal, parenteral, pulmonary, and subcutaneous routes.
- a pharmaceutical composition or compound provided herein is administered parenterally.
- compositions for parenteral administration can be emulsions or sterile solutions.
- Parenteral compositions may include, for example, propylene glycol, polyethylene glycol, vegetable oils, and injectable organic esters (e.g., ethyl oleate). These compositions can also contain wetting, isotonizing, emulsifying, dispersing, and stabilizing agents. Sterilization can be carried out in several ways, for example, using a bacteriological filter, via radiation, or via heating.
- Parenteral compositions can also be prepared in the form of sterile solid compositions which can be dissolved at the time of use in sterile water, or any other injectable sterile medium.
- compositions provided herein is a pharmaceutical composition, or a single unit dosage form.
- Pharmaceutical compositions, and single unit dosage forms provided herein comprise a prophylactically, or therapeutically effective amount of one, or more prophylactic, or therapeutic compounds.
- the pharmaceutical composition may comprise one or more pharmaceutical excipients.
- Any suitable pharmaceutical excipient may be used, wherein a person of ordinary skill in the art is capable of selecting suitable pharmaceutical excipients.
- suitable excipients include starch, glucose, lactose, sucrose, gelatin, malt, rice, flour, chalk, silica gel, sodium stearate, glycerol monostearate, talc, sodium chloride, dried skim milk, glycerol, propylene glycol, water, ethanol, and the like.
- compositions, or dosage form Whether a particular excipient is suitable for incorporation into a pharmaceutical composition, or dosage form depends on a variety of factors well known in the art including, but not limited to, the way in which the dosage form will be administered to a subject and the specific compound in the dosage form.
- the composition, or single unit dosage form if desired, can also contain minor amounts of wetting, or emulsifying agents, or pH buffering agents. Accordingly, the pharmaceutical excipients provided below are intended to be illustrative, and not limiting. Additional pharmaceutical excipients include, for example, those described in the Handbook of Pharmaceutical Excipients, Rowe et al. (Eds.) 6th Ed. (2009), incorporated by reference herein in its entirety.
- the pharmaceutical composition comprises an anti-foaming agent.
- Any suitable anti-foaming agent may be used.
- the anti-foaming agent is selected from an alcohol, an ether, an oil, a wax, a silicone, a surfactant, and combinations thereof.
- the anti-foaming agent is selected from a mineral oil, a vegetable oil, ethylene bis stearamide, a paraffin wax, an ester wax, a fatty alcohol wax, a long-chain fatty alcohol, a fatty acid soap, a fatty acid ester, a silicon glycol, a fluorosilicone, a polyethylene glycol-polypropylene glycol copolymer, polydimethylsiloxane-silicon dioxide, ether, octyl alcohol, capryl alcohol, sorbitan trioleate, ethyl alcohol, 2-ethyl-hexanol, dimethicone, oleyl alcohol, simethicone, and combinations thereof.
- the pharmaceutical composition comprises a co-solvent.
- co-solvents include ethanol, poly(ethylene) glycol, butylene glycol, di methyl acetamide, glycerin, and propylene glycol.
- the pharmaceutical composition comprises a buffer.
- buffers include acetate, borate, carbonate, lactate, malate, phosphate, citrate, hydroxide, diethanolamine, monoethanolamine, glycine, methionine, guar gum, and monosodium glutamate.
- the pharmaceutical composition comprises a carrier, or filler.
- carriers, or fillers include lactose, maltodextrin, mannitol, sorbitol, chitosan, stearic acid, xanthan gum, and guar gum.
- the pharmaceutical composition comprises a surfactant.
- surfactants include /-alpha tocopherol, benzalkonium chloride, benzethonium chloride, cetrimide, cetylpyridinium chloride, docusate sodium, glyceryl behenate, glyceryl monooleate, lauric acid, macrogol 15 hydroxystearate, myristyl alcohol, phospholipids, polyoxyethylene alkyl ethers, polyoxyethylene sorbitan fatty acid esters, polyoxyethylene stearates, poly oxylglyceri des, sodium lauryl sulfate, sorbitan esters, and vitamin E polyethylene(glycol) succinate.
- the pharmaceutical composition comprises an anti-caking agent.
- anti -caking agents include calcium phosphate (tribasic), hydroxymethyl cellulose, hydroxypropyl cellulose, and magnesium oxide.
- Other excipients that may be used with the pharmaceutical compositions include, for example, albumin, antioxidants, antibacterial agents, antifungal agents, bioabsorbable polymers, chelating agents, controlled release agents, diluents, dispersing agents, dissolution enhancers, emulsifying agents, gelling agents, ointment bases, penetration enhancers, preservatives, solubilizing agents, solvents, stabilizing agents, and sugars.
- the pharmaceutical composition comprises a solvent.
- the solvent is saline solution, such as a sterile isotonic saline solution, or dextrose solution.
- the solvent is water for injection.
- the pharmaceutical compositions are in a particulate form, such as a microparticle or a nanoparticle.
- Microparticles, and nanoparticles may be formed from any suitable material, such as a polymer, or a lipid.
- the microparticles, or nanoparticles are micelles, liposomes, or polymersomes.
- anhydrous pharmaceutical compositions, and dosage forms comprising a compound, since, in some embodiments, water can facilitate the degradation of some compounds.
- Anhydrous pharmaceutical compositions, and dosage forms provided herein can be prepared using anhydrous, or low moisture containing ingredients, and low moisture, or low humidity conditions.
- Pharmaceutical compositions, and dosage forms that comprise lactose, and at least one active ingredient that comprises a primary, or secondary amine can be anhydrous if substantial contact with moisture, and/or humidity during manufacturing, packaging, and/or storage is expected.
- An anhydrous pharmaceutical composition can be prepared and stored such that its anhydrous nature is maintained. Accordingly, anhydrous compositions can be packaged using materials known to prevent exposure to water such that they can be included in suitable formulary kits. Examples of suitable packaging include, but are not limited to, hermetically sealed foils, plastics, unit dose containers (e.g., vials), blister packs, and strip packs.
- Lactose-free compositions can comprise excipients that are well known in the art and are listed, for example, in the U.S. Pharmocopeia (USP) SP (XXI)/NF (XVI).
- USP U.S. Pharmocopeia
- XXI XXI/NF
- lactose-free compositions comprise an active ingredient, a binder/fdler, and a lubricant in pharmaceutically compatible, and pharmaceutically acceptable amounts.
- Exemplary lactose-free dosage forms comprise an active ingredient, microcrystalline cellulose, pre gelatinized starch, and magnesium stearate.
- compositions, and dosage forms that comprise one, or more excipients that reduce the rate by which a compound will decompose.
- excipients which are referred to herein as “stabilizers,” include, but are not limited to, antioxidants such as ascorbic acid, pH buffers, or salt buffers.
- parenteral dosage forms can be administered to subjects by various routes including, but not limited to, subcutaneous, intravenous (including bolus injection), intramuscular, and intraarterial. Because their administration typically bypasses subjects’ natural defenses against contaminants, parenteral dosage forms are typically sterile, or capable of being sterilized prior to administration to a subject. Examples of parenteral dosage forms include, but are not limited to, solutions ready for injection, dry products ready to be dissolved or suspended in a pharmaceutically acceptable vehicle for injection, suspensions ready for injection, and emulsions.
- Suitable vehicles that can be used to provide parenteral dosage forms are well known to those skilled in the art. Examples include, but are not limited to Water for Injection USP; aqueous vehicles such as, but not limited to, Sodium Chloride Injection, Ringer’s Injection, Dextrose Injection, Dextrose, and Sodium Chloride Injection, and Lactated Ringer’s Injection; water miscible vehicles such as, but not limited to, ethyl alcohol, polyethylene glycol, and polypropylene glycol; and non-aqueous vehicles such as, but not limited to, corn oil, cottonseed oil, peanut oil, sesame oil, ethyl oleate, isopropyl myristate, and benzyl benzoate.
- aqueous vehicles such as, but not limited to, Sodium Chloride Injection, Ringer’s Injection, Dextrose Injection, Dextrose, and Sodium Chloride Injection, and Lactated Ringer’s Injection
- Excipients that increase the solubility of one, or more of the antibodies disclosed herein can also be incorporated into the parenteral dosage forms.
- the doctor will determine the posology which he considers most appropriate according to a preventive, or curative treatment, and according to the age, weight, condition, and other factors specific to the subject to be treated.
- compositions provided herein is a pharmaceutical composition, or a single unit dosage form.
- Pharmaceutical compositions, and single unit dosage forms provided herein comprise a prophylactically, or therapeutically effective amount of one, or more prophylactic, or therapeutic antibodies, or antigen binding fragments thereof.
- the amount of the compound, or composition which will be effective in the prevention, or treatment of a disorder, or one, or more symptoms thereof will vary with the nature, and severity of the disease, or condition, and the route by which the compound is administered.
- the frequency and dosage will also vary according to factors specific for each subject depending on the specific therapy (e g., therapeutic or prophylactic agents) administered, the severity of the disorder, disease, or condition, the route of administration, as well as age, body, weight, response, and the past medical history of the subject.
- Effective doses may be extrapolated from dose-response curves derived from in vitro, or animal model test systems.
- exemplary doses of a composition include milligram, or microgram amounts of the compound per kilogram of subject, or sample weight (e.g., about 10 micrograms per kilogram to about 50 milligrams per kilogram, about 100 micrograms per kilogram to about 25 milligrams per kilogram, or about 100 microgram per kilogram to about 10 milligrams per kilogram).
- the dosage of the compound provided herein, based on weight of the compound, administered to prevent, treat, manage, or ameliorate a disorder, or one, or more symptoms thereof in a subject is 0.1 mg/kg, 1 mg/kg, 2 mg/kg, 3 mg/kg, 4 mg/kg, 5 mg/kg, 6 mg/kg, 10 mg/kg, or 15 mg/kg or more of a subject’s body weight.
- the dosage of the composition, or a composition provided herein administered to prevent, treat, manage, or ameliorate a disorder, or one, or more symptoms thereof in a subject is 0.1 mg to 200 mg, 0.1 mg to 100 mg, 0.1 mg to 50 mg, 0.1 mg to 25 mg, 0.1 mg to 20 mg, 0.1 mg to 15 mg, 0.1 mg to 10 mg, 0.1 mg to 7.5 mg, 0.1 mg to 5 mg, 0.1 to 2.5 mg, 0.25 mg to 20 mg, 0.25 to 15 mg, 0.25 to 12 mg, 0.25 to 10 mg, 0.25 mg to 7.5 mg, 0.25 mg to 5 mg, 0.25 mg to 2.5 mg, 0.5 mg to 20 mg, 0.5 to 15 mg, 0.5 to 12 mg, 0.5 to 10 mg, 0.5 mg to 7.5 mg, 0.5 mg to 5 mg, 0.5 mg to 2.5 mg, 1 mg to 20 mg, 1 mg to 15 mg, 1 mg to 12 mg, 0.5 to 10 mg, 0.5 mg to 7.5 mg, 0.5 mg to 5 mg, 0.5 mg to 2.5 mg, 1 mg to 20
- the dose can be administered according to a suitable schedule, for example, once, two times, three times, or four times weekly. It may be necessary to use dosages of the compound outside the ranges disclosed herein in some cases, as will be apparent to those of ordinary skill in the art. Furthermore, it is noted that the clinician, or treating physician will know how, and when to interrupt, adjust, or terminate therapy in conjunction with subject response.
- treatment, or prevention can be initiated with one, or more loading doses of a compound, or composition provided herein followed by one, or more maintenance doses.
- a dose of a compound, or composition provided herein can be administered to achieve a steady-state concentration of the compound in blood, or serum of the subject.
- the steady-state concentration can be determined by measurement according to techniques available to those of skill or can be based on the physical characteristics of the subject such as height, weight, and age.
- administration of the same composition may be repeated, and the administrations may be separated by at least one day, two days, three days, five days, ten days, fifteen days, thirty days, forty-five days, two months, seventy-five days, three months, or six months.
- administration of the same prophylactic, or therapeutic agent may be repeated, and the administration may be separated by at least one day, two days, three days, five days, ten days, fifteen days, thirty days, forty-five days, two months, seventy-five days, three months, or six months.
- the compounds are administered to a mammal, in certain embodiments, a human, in a pharmaceutically acceptable dosage suitable for administration form such as those known in the art, and those discussed herein, intravenously as a bolus or by continuous infusion over a period of time, by intramuscular, intraperitoneal, intra-cerebrospinal, subcutaneous, intra-articular, intrasynovial, intrathecal, or intratumoral routes.
- the compounds also are suitably administered by peritumoral, intralesional, or perilesional routes, to exert local as well as systemic therapeutic effects.
- the compounds are administered to a mammal, in certain embodiments, a human, in a pharmaceutically acceptable dosage suitable for oral administration form such as those known in the art, and those discussed herein.
- a pharmaceutically acceptable dosage suitable for oral administration form such as those known in the art, and those discussed herein.
- the compounds of this disclosure may be administered orally to a human as a liquid, or solid form.
- Solid dosage forms include capsules, tablets, pills, powders, and granules.
- the chemical entity is mixed with one or more pharmaceutically acceptable excipients, such as sodium citrate or dicalcium phosphate and/or: a) fillers or extenders such as starches, lactose, sucrose, glucose, mannitol, and silicic acid, b) binders such as, for example, carboxymethylcellulose, alginates, gelatin, polyvinylpyrrolidinone, sucrose, and acacia, c) humectants such as glycerol, d) disintegrating agents such as agar-agar, calcium carbonate, potato or tapioca starch, alginic acid, certain silicates, and sodium carbonate, e) solution retarding agents such as paraffin, f) absorption accelerators such as quaternary ammonium compounds, g) wetting agents such as, for example, cetyl alcohol and glycerol monostearate, h) absorbents such as kaolin and bentonit
- the dosage form may also comprise buffering agents.
- Solid compositions of a similar type may also be employed as fillers in soft and hard-filled gelatin capsules using such excipients as lactose or milk sugar as well as high molecular weight polyethylene glycols and the like.
- Non-limiting examples of disorders associated with diabetes include, obesity, obesity-related disorders, metabolic syndrome, neuropathy, nephropathy (e.g., diabetic nephropathy), retinopathy, diabetic cardiomyopathy, cataract, macroangiopathy, osteopenia, hyperosmolar diabetic coma, infectious disease (e.g., respiratory infection, urinary tract infection, gastrointestinal infection, dermal soft tissue infections, inferior limb infection), diabetic gangrene, xerostomia, hypacusis, cerebrovascular disorder, diabetic cachexia, delayed wound healing, diabetic dyslipidemia peripheral blood circulation disorder, cardiovascular risk factors, (e.g., coronary artery disease, peripheral artery disease, cerebrovascular disease, hypertension, and risk factors related to unmanaged cholesterol, and/or lipid levels, and/or inflammation), NASH, bone fracture, and cognitive dysfunction
- nephropathy e.g., diabetic nephropathy
- retinopathy diabetic cardiomyopathy
- cataract cataract
- macroangiopathy osteop
- diseases, or conditions related to diabetes include, pre-diabetes, hyperlipidemia (e.g., hypertriglyceridemia, hypercholesterolemia, high LDL- cholesterolemia, low HDL-cholesterolemia, postprandial hyperlipemia), metabolic syndrome (e.g., metabolic disorder where activation of GLP-1R is beneficial, metabolic syndrome X), hypertension, impaired glucose tolerance (IGT), insulin resistance, and sarcopenia.
- hyperlipidemia e.g., hypertriglyceridemia, hypercholesterolemia, high LDL- cholesterolemia, low HDL-cholesterolemia, postprandial hyperlipemia
- metabolic syndrome e.g., metabolic disorder where activation of GLP-1R is beneficial
- metabolic syndrome X e.g., metabolic disorder where activation of GLP-1R is beneficial
- hypertension e.g., impaired glucose tolerance (IGT), insulin resistance, and sarcopenia.
- ITT impaired glucose tolerance
- the disease, or condition is diabetes, and obesity (diabesity).
- the compounds described herein are useful in improving the therapeutic effectiveness of metformin.
- the disease or condition is a disorder of a metabolically important tissue.
- the disease, or condition is a fatty liver disease.
- Fatty liver diseases include, but are not limited to, non-alcoholic fatty acid liver disease (NAFLD) or metabolic dysfunction-associated fatty liver disease (MAFLD) , steatohepatitis, non-alcoholic steatohepatitis (NASH), fatty liver disease resulting from hepatitis, fatty liver disease resulting from obesity, fatty liver disease resulting from diabetes, fatty liver disease resulting from insulin resistance, fatty liver disease resulting from hypertriglyceridemia, Abetalipoproteinemia, glycogen storage diseases, Weber-Christian disease, Wolman’s disease, acute fatty liver of pregnancy, and lipodystrophy.
- NAFLD non-alcoholic fatty acid liver disease
- MAFLD metabolic dysfunction-associated fatty liver disease
- NASH non-alcoholic steatohepatitis
- fatty liver disease resulting from obesity fatty liver disease resulting from diabetes
- fatty liver disease resulting from insulin resistance fatty liver disease resulting from
- Non-alcoholic fatty liver disease represents a spectrum of disease occurring in the absence of alcohol abuse, and is typically characterized by the presence of steatosis (fat in the liver).
- NAFLD is believed to be linked to a variety of conditions, e.g., metabolic syndrome (including obesity, diabetes, and hypertriglyceridemia), and insulin resistance. It can cause liver disease in adults and children, and may ultimately lead to cirrhosis (Skelly et al., J Hepatol 2001; 35: 195-9; Chitturi et al., Hepatology 2002; 35(2):373-9).
- NAFLD nonalcoholic fatty liver or NAFL
- NAFL nonalcoholic fatty liver or NAFL
- NASH non-alcoholic steatohepatitis
- the subject is a pediatric subject (e.g., 6-16 years old; or 6-12 years old; or 6-10 years old). In certain embodiments, the subject is an adult subject.
- diseases, or conditions in metabolically important tissues include, joint disorders (e.g., osteoarthritis, secondary osteoarthritis), steatosis (e.g. in the liver); gall stones; gallbladder disorders; gastroesophageal reflux; sleep apnea; hepatitis; fatty liver; bone disorder characterized by altered bone metabolism, such as osteoporosis, including post-menopausal osteoporosis, poor bone strength, osteopenia, Paget's disease, osteolytic metastasis in cancer patients, osteodystrophy in liver disease, and the altered bone metabolism caused by renal failure, or hemodialysis, bone fracture, bone surgery, aging, pregnancy, protection against bone fractures, and malnutrition polycystic ovary syndrome; renal disease (e.g., chronic renal failure, glomerulonephritis, glomerulosclerosis, nephrotic syndrome, hypertensive nephrosclerosis, end-stage renal disease);
- renal disease e.g.,
- the chemical entities described herein can be used for treating surgical trauma by improving recovery after surgery, and/or by preventing the catabolic reaction caused by surgical trauma.
- the disease, or condition is a cardiovascular disease.
- cardiovascular disease include, congestive heart failure, atherosclerosis, arteriosclerosis, coronary heart disease, or peripheral artery disease, stroke, coronary artery disease, congestive heart failure, coronary heart disease, hypertension, cardiac failure, cerebrovascular disorder (e.g., cerebral infarction), vascular dysfunction, myocardial infarction, elevated blood pressure (e.g., 130/85 mm Hg or higher), and prothrombotic state (exemplified by high fibrinogen, or plasminogen activator inhibitor in the blood).
- the disease, or condition is a neurological disorder (e.g., neurodegenerative disorder), or a psychiatric disorder.
- neurological disorders include, brain insulin resistance, mild cognitive impairment (MCI), Alzheimer's disease (AD), Parkinson's disease (PD), anxiety, dementia (e.g., senile dementia), traumatic brain injury, Huntington's chores, tardive dyskinesia, hyperkinesia, mania, Morbus Parkinson, steel-Richard syndrome, Down's syndrome, myasthenia gravis, nerve trauma, brain trauma, vascular amyloidosis, cerebral hemorrhage I with amyloidosis, brain inflammation, Friedrich's ataxia, acute confusion disorder, amyotrophic lateral sclerosis (ALS), glaucoma, and apoptosis-mediated degenerative diseases of the central nervous system (e.g., Creutzfeld-Jakob Disease, bovine spongiform encephalopathy (mad cow disease),
- MCI mild cognitive impairment
- AD Alzheimer'
- Non-limiting examples of psychiatric disorders include drug dependence/addiction (narcotics, amphetamines, and attention deficit/hyperactivity disorder (ADHD).
- drug dependence/addiction narcotics, amphetamines, and attention deficit/hyperactivity disorder (ADHD).
- ADHD attention deficit/hyperactivity disorder
- the chemical entities described herein can be useful in improving behavioral response to addictive drugs, decreasing drug dependence, prevention drug abuse relapse, and relieving anxiety caused by the absence of a given addictive substance.
- the chemical entities described herein are useful in improving learning, and memory by enhancing neuronal plasticity, and facilitation of cellular differentiation, and also in preserving dopamine neurons, and motor function in Morbus Parkinson.
- the disease, or condition is impaired fasting glucose (IFG), impaired fasting glycemia (IFG), hyperglycemia, insulin resistance (impaired glucose homeostasis), hyperinsulinemia, elevated blood levels of fatty acids, or glycerol, a hypoglycemic condition, insulin resistant syndrome, paresthesia caused by hyperinsulinemia, hyperlipidemia, hypercholesteremia, impaired wound healing, leptin resistance, glucose intolerance, increased fasting glucose, dyslipidemia (e.g., hyperlipidemia, atherogenic dyslipidemia characterized by high triglycerides and low HDL cholesterol), glucagonoma, hyperuric acidemia, hypoglycemia (e.g., nighttime hypoglycemia), and concomitant comatose endpoint associated with insulin.
- IGF impaired fasting glucose
- IGF impaired fasting glycemia
- hyperglycemia insulin resistance
- hyperinsulinemia elevated blood levels of fatty acids
- glycerol a hypoglyce
- the compounds described herein can reduce, or slow down the progression of borderline type, impaired fasting glucose, or impaired fasting glycemia into diabetes.
- the disease, or condition is an autoimmune disorder.
- autoimmune disorders include, multiple sclerosis, experimental autoimmune encephalomyelitis, autoimmune disorder is associated with immune rejection, graft versus host disease, uveitis, optic neuropathies, optic neuritis, transverse myelitis, inflammatory bowel disease, rheumatoid arthritis, ankylosing spondylitis, systemic lupus erythematosus, myasthenia gravis, and Graves’ disease.
- the disease, or condition is a stomach, or intestine related disorder.
- these disorders include, ulcers of any etiology (e.g. peptic ulcers, Zollinger-Ellison syndrome, drug-induced ulcers, ulcers related to infections, or other pathogens), digestion disorders, malabsorption, short bowel syndrome, cul-de-sac syndrome, inflammatory bowel diseases (Crohn’s disease, and ulcerative colitis), celiac sprue, hypogammaglobulinemic sprue, chemotherapy, and/or radiation therapy-induced mucositis, and diarrhea, gastrointestinal inflammation, short bowel syndrome, ulcerative colitis, gastric mucosal injury (e.g., gastric mucosal injury caused by aspirin), small intestinal mucosal injury, and cachexia (e.g., cancerous cachexia, tuberculous cachexia, cachexia associated with blood disease, cachexia associated with endocrine disease, cachexia associated with infectious
- the compounds described herein can be used to reduce body weight (e.g., excess body weight), prevent body weight gain, induce weight loss, decrease body fat, or reduce food intake in a subject (e.g., a subject in need thereof).
- the weight increase in a subject may be attributed to excessive ingestion of food, or unbalanced diets, or may be weight increase derived from a concomitant drug (e.g., insulin sensitizers having a PPARy agonist-like action, such as troglitazone, rosiglitazone, englitazone, ciglitazone, pioglitazone, and the like).
- the weight increase may be weight increase before reaching obesity, or may be weight increase in an obese subject.
- the weight increase may also be medication-induced weight gain, or weight gain subsequent to cessation of smoking.
- the compounds described herein can be used to maintain body weight following weight loss,
- the weight loss is caused by injectable medications.
- the injectable medication is selected from the group consisting of CT-388, CT-868, Mounjaro®, Zepbound®, Wegovy®, Ozempic®, Zepbound®, and Saxenda®, as well as others.
- the condition, disease, or disorder is an eating disorder, such as hyperphagia, binge eating, bulimia, or compulsive eating.
- the disease, or condition is an inflammatory disorder.
- inflammatory disorders include, chronic rheumatoid arthritis, spondylitis deformans, arthritis deformans, lumbago, gout, post-operational or post-traumatic inflammation, bloating, neuralgia, laryngopharyngitis, cystitis, pneumonia, pancreatitis, enteritis, inflammatory bowel disease (including inflammatory large bowel disease), inflammation in metabolically important tissues including liver, fat, pancreas, kidney, and gut, and a proinfl ammatory state (e.g., elevated levels of proinflammatory cytokines or, markers of inflammation-like C-reactive protein in the blood).
- a proinfl ammatory state e.g., elevated levels of proinflammatory cytokines or, markers of inflammation-like C-reactive protein in the blood.
- kits for the treatment that include the administration of an effective amount of compounds provided herein.
- the methods encompass the step of administering to the subject in need thereof an amount of a compound described herein effective for the treatment of disease, or condition in combination with a second agent effective for the treatment, or prevention of the disease, or condition.
- the compound is in the form of a pharmaceutical composition, or dosage form, as described elsewhere herein.
- the subject is a treatment naive subject.
- the subject has previously received therapy. For instance, in certain embodiments, the subject has not responded to a single agent treatment regimen. In some embodiments, the subject has received a prior incretin treatment.
- the incretin treatment is selected from exenatide (Byetta®, Bydureon®), liraglutide (Victoza®), sitagliptin (Januvia®, Janumet®, Janumet® XR, Juvisync®), saxagliptin (Onglyza®, Kombiglyze® XR), alogliptin (Nesina®, Kazano®, Oseni®), linagliptin (Tradjenta®, Jentadueto®), semaglutide, Wegovy®, Mounjaro®, and Rybelsus®, as well as others.
- the subject is a subject that discontinued some other therapy because of one or more adverse events associated with the other therapy.
- the subject has received some other therapy and discontinued that therapy prior to administration of a method provided herein.
- the subject has received therapy and continues to receive that therapy along with administration of a compound provided herein.
- the compounds described herein can be co-administered with other therapy for treatment of the disease or condition according to the judgment of one of skill in the art.
- the methods or compositions provided herein can be co-administered with a reduced dose of the other therapy for the treatment of the disease or condition.
- a compound provided herein is provided in the form of a kit
- the procedure is a diagnostic assay. In certain embodiments, the procedure is a therapeutic procedure.
- the kit further comprises a solvent for the reconstitution of the compound.
- the compound is provided in the form of a pharmaceutical composition.
- kits can include a compound, or composition provided herein, an optional second agent, or composition, and instructions providing information to a health care provider regarding usage for treating the disorder. Instructions may be provided in printed form, or in the form of an electronic medium such as a floppy disc, CD, or DVD, or in the form of a website address where such instructions may be obtained.
- a unit dose of a compound, or a composition provided herein, or a second agent, or composition can include a dosage such that when administered to a subject, a therapeutically, or prophylactically effective plasma level of the compound, or composition can be maintained in the subject for at least one day.
- a compound, or composition can be included as a sterile aqueous pharmaceutical composition, or dry powder (e.g., lyophilized) composition.
- suitable packaging includes a solid matrix, or material customarily used in a system, and capable of holding within fixed limits a compound provided herein, and/or a second agent suitable for administration to a subject.
- materials include glass, and plastic (e.g., polyethylene, polypropylene, and polycarbonate) bottles, vials, paper, plastic, plastic-foil laminated envelopes, and the like. If e- beam sterilization techniques are employed, the packaging should have sufficiently low density to permit sterilization of the contents.
- Another aspect relates to labeled crystalline forms of the present disclosure (radio- labeled, fluorescent-labeled, and so on) that would be useful not only in imaging techniques but also in assays, both in vitro and in vivo.
- the present disclosure further includes isotopically-labeled crystalline forms of the present disclosure.
- An “isotopically” or “radio-labeled” compound is a crystalline form of the present disclosure where one or more atoms are replaced or substituted by an atom having an atomic mass or mass number different from the atomic mass or mass number typically found in nature (that is, naturally occurring).
- Suitable radionuclides that may be incorporated in crystalline forms of the present disclosure include but are not limited to 2 H (also written as D for deuterium), 3 H (also written as T for tritium), n C, 13 C, 14 C, 13 N, 15 N, 1?
- radionuclide that is incorporated in the instant radio-labeled compounds will depend on the specific application of that radio-labeled compound. For example, for in vitro metalloprotease labeling and competition assays, compounds that incorporate 3 H, 14 C, 82 Br, 123 I, 131 I, or 35 S will generally be most useful. For radio-imaging applications "C, 18 F, 125 I, 123 I, 124 I, 131 I, 75 Br, 76 Br, or 77 Br will generally be most useful.
- the crystalline forms described herein in which one or more hydrogens is/are replaced by deuterium, such as hydrogen bonded to a carbon atom exhibit increased resistance to metabolism and are thus useful for increasing the half-life of any compound when administered to a mammal, particularly a human. They can also be useful as tools for pharmacokinetic studies.
- a “radio-labeled” or “labeled compound” is a compound that has incorporated at least one radionuclide.
- the radionuclide is selected from the group consisting of 3 H, 14 C, 125 1, 35 S, and 82 Br.
- the present disclosure can further include synthetic methods for incorporating radioisotopes into crystalline forms of the present disclosure. Synthetic methods for incorporating radioisotopes into organic compounds are well known in the art, and a person of ordinary skill in the art will readily recognize the methods applicable for the compounds of disclosure.
- a labeled compound of the disclosure can be used in a screening assay to identify/evaluate compounds. For example, a newly synthesized or identified compound (that is, test compound) which is labeled can be evaluated. Conversely, in some other screening assays, the standard compound is labeled, and test compounds are unlabeled. Accordingly, the concentration of the labeled standard compound is monitored in order to evaluate the competition between the standard compound and the test compound, and the relative binding affinity of the test compound is thus ascertained.
- X-ray diffraction patterns were recorded at ambient conditions in transmission geometry with a STOE STADI P diffractometer (Cu Kai radiation, primary Ge-monochromator, Mythen IK silicon strip detector, angular range 1.5° to 50.5° 2Theta, 0.02° 2Theta step size, 48 seconds step time).
- Compound 1 was used as starting material in preparation of the salt forms.
- Antisolvents were added to the clear solutions and the hazy solutions from the cooling experiments. Suspensions obtained after antisolvent addition were filtered through a 0.45 pm nylon membrane filter by centrifugation at 14,000 rpm. After being dried at 50 °C under vacuum for two hours, the collected solids were analyzed by XRPD. Solids obtained: Compound 1 erbumine salt Form A, Compound 1 erbumine salt Form B, Compound 1 TRIS salt.
- Example 2G Preparation of Compound 1 Sodium Salt Form C and Sodium Salt Form D
- Compound 1 400 mg was weighed into a 20 mL glass vial and 29 mg of (1.05 equivalent) NaOH was added into the glass vial. 4 mL Acetonitrile (5% water) was added into the glass vial under stirring at 25 °C and a small amount of Compound 1 sodium salt Form C crystal seed was added to the suspension. After stirring for one day, solids were collected by filtration and dried to afford Compound 1 sodium salt Form C as a hydrate at 86.39% yield. Compound 1 sodium salt Form D was obtained by the same process.
- erbumine salt Form E is the most thermodynamically stable salt form among Compound 1 Erbumine Salt Form A, Form E, and Form F.
- Example 5 Solubility of Compound 1 Erbumine Salt Form E and Sodium Salt Form C
- Compound 1 Erbumine Salt Form E and Sodium Salt Form C (8 mg each) were each weighed into a 8 mL glass vial. 4mL of solubility medium was added. Obtained suspensions were stirred at 37°C at 600 rpm and sampled at 2 hours and at 24 hours. Filtration and supernatants were analyzed by HPLC for solubility.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Diabetes (AREA)
- General Chemical & Material Sciences (AREA)
- Emergency Medicine (AREA)
- Hematology (AREA)
- Obesity (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Endocrinology (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
Abstract
The present disclosure relates to salts of a GLP-1R agonist, Compound 1. The disclosure also relates to crystalline or amorphous salts of Compound 1. The disclosure also relates to pharmaceutical compositions comprising the salts of Compound 1. The disclosure further relates to methods of treating a disease, disorder, or syndrome using a salt of compound 1 as disclosed herein.
Description
SALT FORMS OF A GLP-1 RECEPTOR MODULATOR
CROSS-REFERENCES TO RELATED APPLICATIONS
[0001] This application is a PCT International Application, claiming the benefit of International Application No. PCT/CN2023/137926, filed on December 11, 2023, the entire content of which is herein incorporated by reference.
TECHNICAL FIELD
[0002] The present disclosure relates to salt forms (e.g., crystalline and/or amorphous forms) of a GLP-1 receptor modulator, 2-(((2S,4S)-4-((2-((2,4-difluorophenoxy)methyl)pyrimidin-4- yl)oxy)-2-methylpiperidin-l-yl)methyl)-l-(((S)-oxetan-2-yl)methyl)-lH-benzo[d]imidazole-6- carboxylic acid. The disclosure also relates to processes for preparing thereof; pharmaceutical compositions; and uses thereof in treating a disease, disorder, or syndrome mediated at least in part by modulating in vivo activity of a GLP-1 receptor.
BACKGROUND
[0003] Diabetes is a serious chronic disease that occurs when the pancreas does not produce enough insulin, or when the body cannot effectively use the insulin it produces. Complications of diabetes include damage to the heart, blood vessels, eyes, kidneys, and nerves. Diabetes can increase risk of heart disease, and stroke. The results include serious effects on quality of life, health, and mortality. WHO Global Report on Diabetes, 2016, World Health Organization. As of 2017, approximately 462 million individuals worldwide, about 6.28% of the population was affected by type 2 diabetes, and this prevalence was increasing measurably. Khan et al., 2020, J. Epidemiol. Glob. Health 10(1): 107-111. The global economic burden of diabetes in 2015 was estimated to be $1.3T and estimated to increase to $2. IT by 2030. Bommer et al., 2018, Diabetes Care 41(5):963-970. Approximately 90-95% of all diabetes cases are type 2 diabetes. Tripathi & Srivastava, 2016, Med. Sci. Monit. 12(7):RA130-147.
[0004] The glucagon-like peptide-1 receptor (GLP-1 receptor, or GLP-1R) has emerged as a potential target for treating type 2 diabetes. Its ligand, glucagon-like peptide-1 (GLP-1) enhances glucose-induced insulin secretion and increases insulin synthesis among many other effects. Doyle and Egan, 2007, Pharmacol. Ther. 113 (3) :546-593. GLP-1 is known to delay gastric emptying, suppress food intake, increase satiety, and reduce weight in humans. Shah and Vella,
2014, Rev Endocr Metab Disord. 15(3): 181-187. Activating the GLP-1 receptor has been shown to have beneficial effects on insulin secretion and the maintenance of beta cell glucose sensing, transcription, synthesis, proliferation, and survival. Doyle and Egan, 2007, supra. While the GLP-1 receptor is a promising therapeutic target, only a handful of GLP-1 receptor drugs have been approved to date, and most, or all of these are peptide, or polypeptide drugs.
[0005] There is a need for additional therapies for treating metabolic diseases, and conditions, like type 2 diabetes. Small molecules targeting GLP-1 receptor should provide safe, stable, and easy to administer therapeutics for metabolic diseases, and conditions such as type 2 diabetes. Suitable salt forms of these compounds that have favorable properties related to processing, manufacturing, storage stability, and/or usefulness as a drug are consistently sought and remain vital to drug development.
SUMMARY
[0006] The present disclosure provides salts of Compound 1, 2-(((2S,4S)-4-((2-((2,4- difluorophenoxy)rnethyl)pyrimidin-4-yl)oxy)-2-methylpiperidin-l-yl)methyl)-l-(((S)-oxetan-2- yl)methyl)-lH-benzo[d]imidazole-6-carboxylic acid, which has the structure:
Compound 1
[0007] Compound 1 is disclosed as Compound 485 in PCT Application No.
PCT/US2022/014156, published as WO 2022165076.
[0008] In one aspect, what is disclosed is salts of Compound 1. In another embodiment, what is disclosed is hydrates, anhydrates, and/or solvates of salts of Compound 1.
[0009] In one embodiment, what is disclosed is Compound 1 tris (hydroxymethyl)aminomethane (TRIS) salt, Compound 1 L-arginine salt, Compound 1 L-Lysine salt, Compound 1 sodium salt, and Compound 1 erbumine salt.
[0010] In one embodiment, the salts of Compound 1 are solids. In a further embodiment, the salts are amorphous or crystalline. In a further embodiment, the salts are amorphous. In a further embodiment, the salts are crystalline.
[0011] In a further embodiment, the crystalline salt form is selected from the group consisting of Compound 1 TRIS salt Form A, Compound 1 sodium salt Form A, Compound 1 sodium salt Form B, Compound 1 sodium salt Form C, Compound 1 sodium salt Form D, Compound 1 erbumine salt Form A, Compound 1 erbumine salt Form B, Compound 1 erbumine salt Form C, Compound 1 erbumine salt Form D, Compound 1 erbumine salt Form E, Compound 1 erbumine salt Form F, and Compound 1 L-Lysine salt Form A.
[0012] In some embodiments, specific crystalline salt forms of an active pharmaceutical ingredient (API) can have advantages over other crystalline or amorphous salt forms, such as ease of preparation, increased stability during storage or processing, more favorable solubility, ease of formulation, ease of handling, and increased bioavailability. Because of the potential advantages associated with one pure crystalline salt form, it is also desirable to prevent or minimize polymorphic conversion (i.e., conversion of one crystal salt form to another; or conversion between one crystal salt form and amorphous form) when two or more salt forms of the API can exist. The stability during both the preparation of formulations containing a salt form (e.g. a crystalline form), and during storage of a pharmaceutical dosage form containing a salt form (e g. a crystalline form) is an important determinant for drug development. The novel salt forms (e.g. crystalline and/or amorphous forms) of Compound 1 described herein are directed toward this and other important ends.
[0013] In one aspect, the disclosure provides a pharmaceutical composition comprising a crystalline salt form of Compound 1 as described herein and a pharmaceutically acceptable excipient.
[0014] In another aspect, the disclosure includes a method of treating a disease, disorder, or syndrome mediated at least in part by modulating in vivo activity of GLP-1R, comprising administering to a subject in need thereof a crystalline salt form as described herein, or a pharmaceutical composition described herein.
[0015] In one embodiment of this aspect, the disease, disorder, or syndrome mediated at least in part by modulating in vivo activity of GLP-1R is a metabolic disease.
[0016] Also provided herein is the use of a crystalline salt form of Compound 1 as described herein, and compositions thereof, for the treatment of a metabolic disease, or condition. Also provided herein is the use of a crystalline salt form of Compound 1 as described herein, and compositions thereof, for the treatment of type 2 diabetes, obesity with or without type 2 diabetes, and/or hyperglycemia.
BRIEF DESCRIPTION OF THE FIGURES
[0017] FIG. 1 is an XRPD pattern of Compound 1 amorphous form.
[0018] FIG. 2 is an mDSC thermogram of Compound 1 amorphous form.
[0019] FIG. 3 is a TGA thermogram of Compound 1 amorphous form.
[0021] FIG. 5 is an XRPD pattern of Compound 1 TRIS salt Form A.
[0022] FIG. 6 is a DSC thermogram of TRIS salt Form A.
[0023] FIG. 7 is a TGA thermogram of TRIS salt Form A.
[0024] FIG. 8 is a 'H NMR spectrum of TRIS salt Form A
[0025] FIG. 9 is an XRPD pattern of Compound 1 sodium salt Form A.
[0026] FIG. 10 is a DSC thermogram of sodium salt Form A.
[0027] FIG. 11 is a TGA thermogram of sodium salt Form A.
[0028] FIG. 12 is an XRPD pattern of Compound 1 sodium salt Form B.
[0029] FIG. 13 is a DSC thermogram of Compound 1 sodium salt Form B.
[0030] FIG. 14 is a TGA thermogram of Compound 1 sodium salt Form B.
[0031] FIG. 15 is a ’H NMR spectrum of Compound 1 sodium salt Form B [0032] FIG. 16A is an XRPD pattern of Compound 1 sodium salt Form C.
[0033] FIG. 16B is an XRPD pattern of Compound 1 sodium salt Form D.
[0034] FIG. 17A are TGA and DSC thermograms of Compound 1 sodium salt Form C.
[0035] FIG. 17B is a TG-FTIR thermograms of Compound 1 sodium salt Form D.
[0036] FIG. 17C is a DSC thermograms of Compound 1 sodium salt Form D.
[0037] FIG. 18 is an FT-IR spectrum of Compound 1 sodium salt Form C.
[0039] FIG. 20 is an XRPD pattern of Compound 1 erbumine salt Form A.
[0040] FIG. 21 is a DSC thermogram of Compound 1 erbumine salt Form A.
[0041] FIG. 22 is a TGA thermogram of Compound 1 erbumine salt Form A.
[0043] FIG. 24 is an XRPD pattern of Compound 1 erbumine salt Form B.
[0044] FIG. 25 is an XRPD pattern of Compound 1 erbumine salt Form C.
[0045] FIG. 26 is a DSC thermogram of Compound 1 erbumine salt Form C.
[0046] FIG. 27 is a TGA thermogram of Compound 1 erbumine salt Form C.
[0047] FIG. 28 is a 1 H NMR spectrum of Compound 1 erbumine salt Form C.
[0048] FIG. 29 is an XRPD pattern of Compound 1 Compound 1 erbumine salt Form D.
[0049] FIG. 30 are TGA and DSC thermograms of Compound 1 erbumine salt Form D.
[0050] FIG. 31 is a 1 H NMR spectrum of Compound 1 erbumine salt Form D.
[0051] FIG. 32 is an XRPD pattern of Compound 1 erbumine salt Form E.
[0052] FIG. 33 are TGA and DSC thermograms of Compound lerbumine salt Form E.
[0053] FIG. 34 is a 1 H NMR spectrum of Compound 1 erbumine salt Form E.
[0054] FIG. 35 is an XRPD pattern of Compound 1 Compound lerbumine salt Form F.
[0055] FIG. 36 are TGA and DSC thermograms of Compound 1 erbumine salt Form F.
[0057] FIG. 38 is an XRPD pattern of Compound 1 L-arginine salt.
[0058] FIG. 39 is an XRPD pattern of Compound 1 L-lysine salt Form A.
DETAILED DESCRIPTION
[0059] As used herein, the following definitions shall apply unless otherwise indicated.
[0060] For purposes of this disclosure, the chemical elements are identified in accordance with the Periodic Table of the Elements, CAS version, Handbook of Chemistry and Physics, 95th Ed. Additionally, general principles of organic chemistry are described in "Organic Chemistry,” 2nd Ed., Thomas Sorrell, University Science Books, Sausalito: 2006, and "March’s Advanced Organic Chemistry,” 7th Ed., Ed.: Smith, M.B. and March, J., John Wiley & Sons, New York: 2013, the entire contents of which are hereby incorporated by reference.
[0061] As used herein, the term “about” or “approximate” or “approximately” includes (and describes) embodiments that are directed to that value or parameter per se. In certain embodiments, the term “about” or “approximate” or “approximately” includes the indicated amount ± 10%. In other embodiments, the term “about” approximate” or “approximately” includes the indicated amount ± 5%. In certain other embodiments, the term “about” approximate” or “approximately” includes the indicated amount ± 1%.
[0062] As used herein, the term “crystalline” refers to compounds in a solid state having a periodic and repeating three-dimensional internal arrangement of atoms, ions or molecules characteristic of crystals, for example, arranged in fixed geometric patterns or lattices that have rigid long range order. The term crystalline does not necessarily mean that the compound exists as crystals, but that it has a crystal-like internal structural arrangement.
[0063] As used herein, the term “amorphous” refers to a material having diffuse scattering pattern present, but no evidence for Bragg peaks in the XRPD pattern, as well as to noncrystalline solid in which the atoms and molecules are not organized in a definite lattice pattern.
[0064] “Patient” for the purposes of the present disclosure includes humans and any other animals, particularly mammals. Thus, the methods are applicable to both human therapy and veterinary applications. In a preferred embodiment, the patient is a mammal, and in a most preferred embodiment, the patient is human. Examples of the preferred mammals include mice, rats, other rodents, rabbits, dogs, cats, swine, cattle, sheep, horses, and primates.
[0065] “Therapeutically effective amount” is an amount of a crystalline salt form of the present disclosure that, when administered to a patient, ameliorates a symptom of the disease. The amount of the crystalline salt form of the present disclosure which constitutes a “therapeutically effective amount” will vary depending on the compound, the disease state and its severity, the age of the patient to be treated, and the like. The therapeutically effective amount can be determined routinely by one of ordinary skill in the art having regard to his own knowledge and to this disclosure.
[0066] The phrase “pharmaceutically acceptable” is employed herein to refer to those compounds, materials, compositions, and/or dosage forms which are, within the scope of sound medical judgment, suitable for use in contact with the tissues of human beings and animals without excessive toxicity, irritation, allergic response, immunogenicity or other problem or complication, commensurate with a reasonable benefit risk ratio. In addition, guidelines for impurities and residual solvents levels in crystalline and amorphous pharmaceuticals are provided by ICH in “Impurities: Guidelines for Residual Solvents Q3C(R6)” posted at htps://database.ich.org/sites/default/files/Q3C-R6_Guideline_ErrorCorrection 2019_0410_0.pdf (last visited December 01, 2023).
[0067] As used herein, the phrase “pharmaceutically acceptable excipient” refers to a pharmaceutically acceptable material, composition, or vehicle, such as a liquid or solid filler, diluent, solvent, or encapsulating material. Excipients are generally safe, non-toxic and neither biologically nor otherwise undesirable and include excipients that are acceptable for veterinary use as well as human pharmaceutical use. In one embodiment, each component is “pharmaceutically acceptable” as defined herein. See, e.g., Remington: The Science and Practice
of Pharmacy, 21st ed.; Lippincott Williams & Wilkins: Philadelphia, Pa., 2005; Handbook of Pharmaceutical Excipients, 6th ed.; Rowe et al, Eds.; The Pharmaceutical Press and the American Pharmaceutical Association: 2009; Handbook of Pharmaceutical Additives, 3rd ed.; Ash and Ash Eds.; Gower Publishing Company: 2007; Pharmaceutical Preformulation and Formulation, 2nd ed.; Gibson Ed.; CRC Press LLC: Boca Raton, Fla., 2009.
[0068] The terms “treat,” “treating,” “treatment,” and the like refer to eliminating, reducing, or ameliorating a disease or condition, and/or symptoms associated therewith. Although not precluded, treating a disease or condition does not require that the disease, condition, or symptoms associated therewith be completely eliminated. The terms contemplate administering a therapeutically effective amount of a compound of the present disclosure to a subject in need of such treatment. The treatment can be orientated symptomatically, for example, to suppress symptoms. It can be achieved over a short period, be oriented over a medium term, or can be a long-term treatment, for example within the context of a maintenance therapy.
[0069] The terms “prevent,” “preventing,” and “prevention” refer to a method of preventing the onset of a disease or condition and/or its attendant symptoms or barring a subject from acquiring a disease. As used herein, “prevent,” “preventing,” and “prevention” also include delaying the onset of a disease and/or its attendant symptoms and reducing a subject's risk of acquiring a disease. The terms “prevent,” “preventing” and “prevention” may include “prophylactic treatment,” which refers to reducing the probability of redeveloping a disease or condition, or of a recurrence of a previously controlled disease or condition, in a subject who does not have, but is at risk of or is susceptible to, redeveloping a disease or condition or a recurrence of the disease or condition.
[0070] In general, the nomenclature used in this application is based on naming conventions adopted by the international union of pure and applied chemistry (IUPAC). Chemical structures shown herein were prepared using CHEMDRAW®. Any open valency appearing on a carbon, oxygen, or nitrogen atom in the structures herein indicates the presence of a hydrogen atom.
[0071] Unless otherwise stated, all XRPD peaks are reported on a 2 Theta (29) scale, ± 0.20.
EMBODIMENTS
[0072] In one aspect, the disclosure relates to a salt of Compound 1, also known as 2- (((2S,4S)-4-((2-((2,4-difluorophenoxy)methyl)pyrimidin-4-yl)oxy)-2-methylpiperidin-l-
yl)methyl)-l-(((S)-oxetan-2-yl)methyl)-lH-benzo[d]imidazole-6-carboxylic acid. Compound 1 has the structure depicted below.
Compound 1
[0073] In one embodiment, the salt is crystalline or amorphous. In one embodiment, the salt is crystalline. In one embodiment, the salt is amorphous. In one embodiment, the salt is a mixture of crystalline and amorphous forms.
[0074] In one embodiment, the salt is a hydrate, anhydrate, or solvate.
[0075] In one embodiment, the salt of Compound 1 is selected from the group consisting of Compound 1 TRIS salt, Compound 1 sodium salt, Compound 1 L-arginine salt, Compound 1 L- lysine salt, and Compound 1 erbumine salt.
[0076] In another embodiment, the crystalline salt of Compound 1 is selected from the group consisting of Compound 1 TRIS salt Form A, Compound 1 sodium salt Form A, Compound 1 sodium salt Form B, Compound 1 sodium salt Form C, Compound 1 sodium salt Form D, Compound 1 erbumine salt Form A, Compound 1 erbumine salt Form B, Compound 1 erbumine salt Form C, Compound 1 erbumine salt Form D, Compound 1 erbumine salt Form E, Compound 1 erbumine salt Form F, and Compound 1 L-lysine salt Form A.
[0077] These and other aspects and embodiments will now be described in more detail.
Compound 1
[0078] As noted above, this disclosure relates to a salt of Compound 1. A number of methods can be used to prepare Compound 1, for example, see Example 485 of PCT Application No. PCT/US2022/014156, published as WO 2022165076. Compound 1 prepared according to the procedure in WO 2022165076 is isolated as an amorphous form. Amorphous Compound 1 can also be prepared by slow evaporation from THF or DCM, or by fast evaporation from MeOH, THF, EA, DCM, or 2-MeTHF.
[0079] Amorphous Compound 1 was used as the starting material to make the salts of the present disclosure.
[0080] FIG. 1 is an XRPD pattern of amorphous Compound 1.
[0081] FIG. 2 is an mDSC thermogram of amorphous Compound 1.
[0082] FIG. 3 is a TGA thermogram of amorphous Compound 1.
[0083] FIG. 4 is a 1 H NMR spectrum of amorphous Compound 1 in DMSO
Compound 1 TRIS Salt
[0084] In one embodiment, the salt is Compound 1 TRIS salt. In one embodiment, Compound 1 TRIS salt is crystalline. In a further embodiment, Compound 1 TRIS crystalline salt is characterized as Compound 1 TRIS salt Form A. In a further embodiment, Compound 1 TRIS salt is characterized as Compound 1 TRIS salt Form A (1 : 1) (stoichiometric ratio of Compound 1: TRIS).
[0085] The XRPD pattern of Compound 1 TRIS salt was recorded in reflection geometry.
[0086] In another embodiment, Compound 1 TRIS salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 3.92, 7.41, 7.83,
8.40, 10.76, 11.73, 12.26, 13.87, 14.17, 14.89, 15.29, 15.94, 16.38, 16.77, 17.44, 17.77, 18.21,
18.93, 19.28, 19.63, 19.92, 20.21, 20.46, 21.03, 21.42, 21.90, 22.29, 22.80, 23.46, 24.16, 24.39, 24.678, 24.86, 25.38, 25.62, 26.27, 26.51, 27.47, 27.78, 28.09, 28.58, 29.20, 29.91, 30.78, 31.49, 32.19, 32.80, and 33.47.
[0087] In another embodiment, Compound 1 TRIS salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising three or more peaks selected from 3.92, 7.41, 7.83,
8.40, 10.76, 11.73, 12.26, 13.87, 14.17, 14.89, 15.29, 15.94, 16.38, 16.77, 17.44, 17.77, 18.21,
18.93, 19.28, 19.63, 19.92, 20.21, 20.46, 21.03, 21.42, 21.90, 22.29, 22.80, 23.46, 24.16, 24.39, 24.678, 24.86, 25.38, 25.62, 26.27, 26.51, 27.47, 27.78, 28.09, 28.58, 29.20, 29.91, 30.78, 31.49, 32.19, 32.80, and 33.47.
[0088] In another embodiment, Compound 1 TRIS salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising five or more peaks selected from 3.92, 7.41, 7.83,
8.40, 10.76, 11.73, 12.26, 13.87, 14.17, 14.89, 15.29, 15.94, 16.38, 16.77, 17.44, 17.77, 18.21,
18.93, 19.28, 19.63, 19.92, 20.21, 20.46, 21.03, 21.42, 21.90, 22.29, 22.80, 23.46, 24.16, 24.39,
24.678, 24.86, 25.38, 25.62, 26.27, 26.51, 27.47, 27.78, 28.09, 28.58, 29.20, 29.91, 30.78, 31.49, 32.19, 32.80, and 33.47.
[0089] In another embodiment, Compound 1 TRIS salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising ten or more peaks selected from 3.92, 7.41, 7.83, 8.40, 10.76, 11.73, 12.26, 13.87, 14.17, 14.89, 15.29, 15.94, 16.38, 16.77, 17.44, 17.77, 18.21, 18.93, 19.28, 19.63, 19.92, 20.21, 20.46, 21.03, 21.42, 21.90, 22.29, 22.80, 23.46, 24.16, 24.39, 24.678, 24.86, 25.38, 25.62, 26.27, 26.51, 27.47, 27.78, 28.09, 28.58, 29.20, 29.91, 30.78, 31.49, 32.19, 32.80, and 33.47.
[0090] In another embodiment, Compound 1 TRIS salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising peaks at 10.76, 16.77, and 18.93.
[0091] In another embodiment, Compound 1 TRIS salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising peaks at 10.76, 15.94, 16.77, 18.93, and 22.80.
[0092] In another embodiment, Compound 1 TRIS salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising peaks at 3.94, 10.76, 15.94, 16.77, 17.77, 18.93, 19.92, 20.21, 22.80, and 24.68.
[0093] In another embodiment, Compound 1 TRIS salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising all of the peaks 6.05, 9.53, 10.83, 11.95, 12.80, 12.88, 13.27, 14.24, 15.03, 15.71, 17.51, 18.00, 18.55, 19.51, 20.00, 20.57, 21.10, 21.50, 22.08, 22.46, 23.64, 23.81, 24.55, 25.22, 25.43, 27.07, 27.97, 28.53, 29.19, 29.64, 29.96, 31.63, 32.58, 34.44, 35.09, and 35.54.
[0094] In another embodiment, Compound 1 TRIS salt Form A is characterized by the XRPD pattern substantially in accordance with FIG. 5.
[0095] A list of the peaks from the XRPD pattern for Compound 1 TRIS salt Form A is provided in Table 1 below.
[0096] In another embodiment, Compound 1 TRIS salt Form A is characterized by a DSC thermogram substantially in accordance with FIG. 6.
[0097] In another embodiment, Compound 1 TRIS salt Form A is characterized by a DSC thermogram showing a melting peak onset temperature about 155 °C - 159 °C and peak temperature about 156 °C - 160 °C.
[0098] In another embodiment, Compound 1 TRIS salt Form A is characterized by a DSC thermogram showing a melting peak onset temperature about 156.6 °C and peak temperature about 158.0 °C.
[0099] In another embodiment, Compound 1 TRIS salt Form A is characterized by the TGA thermogram substantially in accordance with FIG. 7.
[00100] In another embodiment, Compound 1 TRIS salt Form A is characterized by a TGA thermogram showing about 0.5-0.7 percent weight loss at 150 °C.
[00101] In another embodiment, Compound 1 TRIS salt Form A is characterized by the TGA thermogram showing about 0.6 percent weight loss at 150 °C.
[00102] In another embodiment, Compound 1 TRIS salt Form A is characterized by the
NMR spectrum in d6-DMSO substantially in accordance with FIG. 8 indicating no detectable residual solvent.
[00103] In another embodiment, Compound 1 TRIS salt Form A is characterized by at least one of the following:
(1) an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 3.92, 7.41, 7.83, 8.40, 10.76, 11.73, 12.26, 13.87, 14.17, 14.89, 15.29, 15.94, 16.38, 16.77, 17.44, 17.77, 18.21, 18.93, 19.28, 19.63, 19.92, 20.21, 20.46, 21.03, 21.42, 21.90, 22.29, 22.80, 23.46, 24.16, 24.39, 24.678, 24.86, 25.38, 25.62, 26.27, 26.51, 27.47, 27.78, 28.09, 28.58, 29.20, 29.91, 30.78, 31.49, 32.19, 32.80, and 33.47;
(2) an DSC thermogram substantially in accordance with FIG. 6; and/or
(3) a TGA thermogram substantially in accordance with FIG. 7.
[00104] In another embodiment, Compound 1 TRIS salt Form A is characterized by at least one of the following:
(1) an XRPD pattern substantially in accordance with FIG. 5;
(2) an DSC thermogram substantially in accordance with FIG. 6; and/or
(3) a TGA thermogram substantially in accordance with FIG. 7.
[00105] In another embodiment, Compound 1 TRIS salt Form A is characterized by at least two of the following:
(1) an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 3.92, 7.41, 7.83, 8.40, 10.76, 11.73, 12.26, 13.87, 14.17, 14.89, 15.29, 15.94, 16.38, 16.77, 17.44, 17.77, 18.21 , 18.93, 19.28, 19.63, 19.92, 20.21 , 20.46, 21.03, 21.42, 21.90,
22.29, 22.80, 23.46, 24.16, 24.39, 24.678, 24.86, 25.38, 25.62, 26.27, 26.51, 27.47, 27.78, 28.09, 28.58, 29.20, 29.91, 30.78, 31.49, 32.19, 32.80, and 33.47;
(2) an DSC thermogram substantially in accordance with FIG. 6; and/or
(3) a TGA thermogram substantially in accordance with FIG. 7.
[00106] In another embodiment, Compound 1 TRIS salt Form A is characterized by all of the following:
(1) an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 3.92, 7.41, 7.83, 8.40, 10.76, 11.73, 12.26, 13.87, 14.17, 14.89, 15.29, 15.94, 16.38, 16.77, 17.44, 17.77, 18.21, 18.93, 19.28, 19.63, 19.92, 20.21, 20.46, 21.03, 21.42, 21.90, 22.29, 22.80, 23.46, 24.16, 24.39, 24.678, 24.86, 25.38, 25.62, 26.27, 26.51, 27.47, 27.78, 28.09, 28.58, 29.20, 29.91, 30.78, 31.49, 32.19, 32.80, and 33.47;
(2) an DSC thermogram substantially in accordance with FIG. 6; and
(3) a TGA thermogram substantially in accordance with FIG. 7.
[00107] In another embodiment, Compound 1 TRIS salt Form A is characterized by (1) an XRPD pattern substantially in accordance with FIG. 5 and at least one of the following:
(2) an DSC thermogram substantially in accordance with FIG. 6; and/or
(3) a TGA thermogram substantially in accordance with FIG. 7.
[00108] In another embodiment, Compound 1 TRIS salt Form A is characterized by (1) an XRPD pattern substantially in accordance with FIG. 5 and:
(2) an DSC thermogram substantially in accordance with FIG. 6; and
(3) a TGA thermogram substantially in accordance with FIG. 7.
Compound 1 Sodium Salt Form A
[00109] In one embodiment, the salt is Compound 1 sodium salt. In another embodiment, Compound 1 sodium salt is crystalline. In another embodiment, Compound 1 sodium crystalline salt is characterized as Compound 1 sodium salt Form A. In a further embodiment, Compound 1 sodium salt is characterized as Compound 1 sodium salt Form A ( 1 : 1.1) (stoichiometric ratio of Compound 1: Na+).
[00110] The XRPD pattern of Compound 1 sodium salt Form A was recorded in reflection geometry.
[00111] In another embodiment, Compound 1 sodium salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 4.79, 9.60, 11.10, 11.88, 13.06, 14.05, 16.20, 18.14, 21.11, 21.81, 23.86, 26.07, 29.00, and 29.94.
[00112] In another embodiment, Compound 1 sodium salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising three or more peaks selected from 4.79, 9.60, 11.10, 11.88, 13.06, 14.05, 16.20, 18.14, 21.11, 21.81, 23.86, 26.07, 29.00, and 29.94.
[00113] In another embodiment, Compound 1 sodium salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising five or more peaks selected from 4.79, 9.60, 11.10, 11.88, 13.06, 14.05, 16.20, 18.14, 21.11, 21.81, 23.86, 26.07, 29.00, and 29.94.
[00114] In another embodiment, Compound 1 sodium salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising ten or more peaks selected from 4.79, 9.60, 11.10, 11.88, 13.06, 14.05, 16.20, 18.14, 21.11, 21.81, 23.86, 26.07, 29.00, and 29.94.
[00115] In another embodiment, Compound 1 sodium salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising peaks at 4.79, 9.60, and 21.81.
[00116] In another embodiment, Compound 1 sodium salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising peaks at 4.79, 9.60, 18.14, 21.81, and 23.86.
[00117] In another embodiment, Compound 1 sodium salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising peaks at 4.79, 9.60, 11.10, 14.05, 16.20, 18.14, 21.81, 23.86, 29.00, and 29.94.
[00118] In another embodiment, Compound 1 sodium salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising all of the peaks 4.79, 9.60, 11.10, 11.88, 13.06, 14.05, 16.20, 18.14, 21.11, 21.81, 23.86, 26.07, 29.00, and 29.94.
[00119] In another embodiment, Compound 1 sodium salt Form A is characterized by the XRPD pattern substantially in accordance with FIG. 9.
[00120] A list of the peaks from the XRPD pattern for Compound 1 sodium salt Form A is provided in Table 2 below.
[00121] In another embodiment, Compound 1 sodium salt Form A is characterized by a DSC thermogram substantially in accordance with FIG. 10.
[00122] In another embodiment, Compound 1 sodium salt Form A is characterized by a DSC thermogram showing desolvation peak onset at about 110 °C - 114 °C.
[00123] In another embodiment, Compound 1 sodium salt Form A is characterized by a DSC thermogram showing desolvation peak onset at about 112 °C.
[00124] In another embodiment, Compound 1 sodium salt Form A is characterized by a TGA thermogram substantially in accordance with FIG. 11.
[00125] In another embodiment, Compound 1 sodium salt Form A is characterized by a TGA thermogram showing about 4.0 - 6.0 percent weight loss at 150 °C.
[00126] In another embodiment, Compound 1 sodium salt Form A is characterized by a TGA thermogram showing about 5.0 percent weight loss at 150 °C.
[00127] In another embodiment, Compound 1 sodium salt Form A is characterized by
NMR spectrum in d6-DMSO indicating 2.8 percent detectable residual solvent.
[00128] In another embodiment, Compound 1 sodium salt Form A is characterized by at least one of the following:
(1) an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 4.79, 9.60, 11.10, 11.88, 13.06, 14.05, 16.20, 18.14, 21.1 1, 21.81, 23.86, 26.07, 29.00, and 29.94;
(2) an DSC thermogram substantially in accordance with FIG. 10; and/or
(3) a TGA thermogram substantially in accordance with FIG. 11.
[00129] In another embodiment, Compound 1 sodium salt Form A is characterized by at least one of the following:
(1) an XRPD pattern substantially in accordance with FIG. 9;
(2) an DSC thermogram substantially in accordance with FIG. 10; and/or
(3) a TGA thermogram substantially in accordance with FIG. 11.
[00130] In another embodiment, Compound 1 sodium salt Form A is characterized by at least two of the following:
(1) an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 4.79, 9.60, 11.10, 11.88, 13.06, 14.05, 16.20, 18.14, 21.11, 21.81, 23.86, 26.07, 29.00, and 29.94;
(2) an DSC thermogram substantially in accordance with FIG. 10; and/or
(3) a TGA thermogram substantially in accordance with FIG. 11.
[00131] In another embodiment, Compound 1 sodium salt Form A is characterized by all of the following:
(1) an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 4.79, 9.60, 11.10, 11.88, 13.06, 14.05, 16.20, 18.14, 21.11, 21.81, 23.86, 26.07, 29.00, and 29.94;
(2) an DSC thermogram substantially in accordance with FIG. 10; and
(3) a TGA thermogram substantially in accordance with FIG. 11.
[00132] In another embodiment, Compound 1 sodium salt Form A is characterized by (1) an XRPD pattern substantially in accordance with FIG. 9 and at least one of the following:
(2) an DSC thermogram substantially in accordance with FIG. 6; and/or
(3) a TGA thermogram substantially in accordance with FIG. 7.
[00133] In another embodiment, Compound 1 sodium salt Form A is characterized by (1) an XRPD pattern substantially in accordance with FIG. 9 and:
(2) an DSC thermogram substantially in accordance with FIG. 10; and
(3) a TGA thermogram substantially in accordance with FIG. 11.
Compound 1 Sodium Salt Form B
[00134] In one embodiment, Compound 1 sodium crystalline salt is characterized as Compound 1 sodium salt Form B. In a further embodiment, Compound 1 sodium salt is characterized as Compound 1 sodium salt Form B (1 :1.1) (stoichiometric ratio of Compound 1 : Na+).
[00135] The XRPD pattern of Compound 1 sodium salt Form B was recorded in reflection geometry.
[00136] In another embodiment, Compound 1 sodium salt Form B is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 4.49, 4.58, 6.63, 8.84, 9.34, 11.03, 11.74, 13.18, 14.03, 15.44, 15.92, 16.22, 17.11, 17.82, 18.19, 18.85,
19.83, 21.10, 21.73, 22.63, 23.60, 24.36, 24.57, 25.23, 25.72, 26.81, 28.10, 28.77, 29.45, 30.89,
31.60, 35.58, and 35.97.
[00137] In another embodiment, Compound 1 sodium salt Form B is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising three or more peaks selected from 4.49, 4.58, 6.63, 8.84, 9.34, 11.03, 11.74, 13.18, 14.03, 15.44, 15.92, 16.22, 17.11, 17.82, 18.19, 18.85,
19.83, 21.10, 21.73, 22.63, 23.60, 24.36, 24.57, 25.23, 25.72, 26.81, 28.10, 28.77, 29.45, 30.89,
31.60, 35.58, and 35.97.
[00138] In another embodiment, Compound 1 sodium salt Form B is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising five or more peaks selected from 4.49, 4.58, 6.63, 8.84, 9.34, 11.03, 11.74, 13.18, 14.03, 15.44, 15.92, 16.22, 17.11, 17.82, 18.19, 18.85,
19.83, 21.10, 21.73, 22.63, 23.60, 24.36, 24.57, 25.23, 25.72, 26.81, 28.10, 28.77, 29.45, 30.89,
31.60, 35.58, and 35.97.
[00139] In another embodiment, Compound 1 sodium salt Form B is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising ten or more peaks selected from 4.49, 4.58, 6.63, 8.84, 9.34, 11.03, 11.74, 13.18, 14.03, 15.44, 15.92, 16.22, 17.11, 17.82, 18.19, 18.85,
19.83, 21.10, 21.73, 22.63, 23.60, 24.36, 24.57, 25.23, 25.72, 26.81, 28.10, 28.77, 29.45, 30.89,
31.60, 35.58, and 35.97.
[00140] In another embodiment, Compound 1 sodium salt Form B is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising peaks at 4.49, 6.63, and 8.84.
[00141] In another embodiment, Compound 1 sodium salt Form B is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising peaks at 4.49, 4.58, 6.63, 8.84, and 9.34. [00142] In another embodiment, Compound 1 sodium salt Form B is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising peaks at 4.49, 4.58, 6.63, 8.84, 9.34, 18.85, 19.83, 21.10, 25.23, and 28.77.
[00143] In another embodiment, Compound 1 sodium salt Form B is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising all of the peaks 4.49, 4.58, 6.63, 8.84, 9.34,
11.03, 11.74, 13.18, 14.03, 15.44, 15.92, 16.22, 17.11, 17.82, 18.19, 18.85, 19.83, 21.10, 21.73, 22.63, 23.60, 24.36, 24.57, 25.23, 25.72, 26.81, 28.10, 28.77, 29.45, 30.89, 31.60, 35.58, and 35.97.
[00144] In another embodiment, Compound 1 sodium salt Form B is characterized by the XRPD pattern substantially in accordance with FIG. 12.
[00145] A list of the peaks from the XRPD pattern for Compound 1 sodium salt Form B is provided in Table 3 below.
Table 3. XRPD Peaks of Compound 1 Sodium Salt Form B
[00146] In another embodiment, Compound 1 sodium salt Form B is characterized by a DSC thermogram substantially in accordance with FIG. 13.
[00147] In another embodiment, Compound 1 sodium salt Form B is characterized by a DSC thermogram showing desolvation peak onset about 111 °C - 115 °C.
[00148] In another embodiment, Compound 1 sodium salt Form B is characterized by a DSC thermogram showing desolvation peak onset at about 113.4 °C.
[00149] In another embodiment, Compound 1 sodium salt Form B is characterized by a TGA thermogram substantially in accordance with FIG. 14.
[00150] In another embodiment, Compound 1 sodium salt Form B is characterized by a TGA thermogram showing about 5.5 - 7.5 percent weight loss at 150 °C.
[00151] In another embodiment, Compound 1 sodium salt Form B is characterized by a TGA thermogram showing about 6.5 percent weight loss at 150 °C.
[00152] In another embodiment, Compound 1 sodium salt Form B is characterized by the ’H NMR spectrum in d6-DMSO substantially in accordance with FIG. 15 indicating 5.7 percent residual solvent.
[00153] In another embodiment, Compound 1 sodium salt Form B is characterized by at least one of the following:
(1) an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 4.49, 4.58, 6.63, 8.84, 9.34, 11.03, 11.74, 13.18, 14.03, 15.44, 15.92, 16.22, 17.11, 17.82, 18.19, 18.85, 19.83, 21.10, 21.73, 22.63, 23.60, 24.36, 24.57, 25.23, 25.72, 26.81, 28.10, 28.77, 29.45, 30.89, 31.60, 35.58, and 35.97;
(2) an DSC thermogram substantially in accordance with FIG. 13; and/or
(3) a TGA thermogram substantially in accordance with FIG. 14.
[00154] In another embodiment, Compound 1 sodium salt Form B is characterized by at least one of the following:
(1) an XRPD pattern substantially in accordance with FIG. 12;
(2) an DSC thermogram substantially in accordance with FIG. 13; and/or
(3) a TGA thermogram substantially in accordance with FIG. 14.
[00155] In another embodiment, Compound 1 sodium salt Form B is characterized by at least two of the following:
(1) an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 4.49, 4.58, 6.63, 8.84, 9.34, 11.03, 11.74, 13.18, 14.03, 15.44, 15.92, 16.22, 17.11, 17.82, 18.19, 18.85, 19.83, 21.10, 21.73, 22.63, 23.60, 24.36, 24.57, 25.23, 25.72, 26.81, 28.10, 28.77, 29.45, 30.89, 31.60, 35.58, and 35.97;
(2) an DSC thermogram substantially in accordance with FIG. 13; and/or
(3) a TGA thermogram substantially in accordance with FIG. 14;
[00156] In another embodiment, Compound 1 sodium salt Form B is characterized by all of the following:
(1) an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 4.49, 4.58, 6.63, 8.84, 9.34, 11.03, 11.74, 13.18, 14.03, 15.44, 15.92, 16.22, 17.11, 17.82, 18.19, 18.85, 19.83, 21.10, 21.73, 22.63, 23.60, 24.36, 24.57, 25.23, 25.72, 26.81, 28.10, 28.77, 29.45, 30.89, 31.60, 35.58, and 35.97;
(2) an DSC thermogram substantially in accordance with FIG. 13; and
(3) a TGA thermogram substantially in accordance with FIG. 14;
[00157] In another embodiment, Compound 1 sodium salt Form B is characterized by (1) an XRPD pattern substantially in accordance with FIG. 12 and at least one of the following:
(2) an DSC thermogram substantially in accordance with FIG. 13; and/or
(3) a TGA thermogram substantially in accordance with FIG. 14.
[00158] In another embodiment, Compound 1 sodium salt Form B is characterized by (1) an XRPD pattern substantially in accordance with FIG. 12 and:
(2) an DSC thermogram substantially in accordance with FIG. 13; and
(3) a TGA thermogram substantially in accordance with FIG. 14.
Compound 1 Sodium Salt Form C
[00159] In one embodiment, Compound 1 sodium crystalline salt is characterized as Compound 1 sodium salt Form C. In a further embodiment, Compound 1 sodium salt is a hydrate characterized as Compound 1 sodium salt Form C (1 :0.9) (stoichiometric ratio of Compound 1: Na+).
[00160] The XRPD pattern of Compound 1 sodium salt Form C was recorded in reflection geometry.
[00161] In another embodiment, Compound 1 sodium salt Form C is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 3.11, 5.85, 11.81, 12.20, 12.58, 12.88, 13.46, 14.27, 15.04, 15.98, 16.67, 16.99, 17.68, 18.06, 18.38, 18.79, 19.32, 19.89, 20.43, 20.93, 21.48, 22.17, 23.31, 23.90, 24.42, 24.81, 26.36, 26.95, 27.61, 27.84,
28.42, 28.779, 29.68, 30.06, 30.98, 32.40, 32.90, 33.89, 34.50, 35.13, 36.19, and 37.01.
[00162] In another embodiment, Compound 1 sodium salt Form C is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising three or more peaks selected from 3.11, 5.85, 11.81, 12.20, 12.58, 12.88, 13.46, 14.27, 15.04, 15.98, 16.67, 16.99, 17.68, 18.06, 18.38, 18.79, 19.32, 19.89, 20.43, 20.93, 21.48, 22.17, 23.31, 23.90, 24.42, 24.81, 26.36, 26.95, 27.61, 27.84, 28.42, 28.779, 29.68, 30.06, 30.98, 32.40, 32.90, 33.89, 34.50, 35.13, 36.19, and 37.01.
[00163] In another embodiment, Compound 1 sodium salt Form C is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising five or more peaks selected from 3.11, 5.85, 11.81, 12.20, 12.58, 12.88, 13.46, 14.27, 15.04, 15.98, 16.67, 16.99, 17.68, 18.06, 18.38, 18.79, 19.32, 19.89, 20.43, 20.93, 21.48, 22.17, 23.31, 23.90, 24.42, 24.81, 26.36, 26.95, 27.61, 27.84, 28.42, 28.779, 29.68, 30.06, 30.98, 32.40, 32.90, 33.89, 34.50, 35.13, 36.19, and 37.01.
[00164] In another embodiment, Compound 1 sodium salt Form C is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising ten or more peaks selected from 3.11, 5.85, 11.81, 12.20, 12.58, 12.88, 13.46, 14.27, 15.04, 15.98, 16.67, 16.99, 17.68, 18.06, 18.38, 18.79, 19.32, 19.89, 20.43, 20.93, 21.48, 22.17, 23.31, 23.90, 24.42, 24.81, 26.36, 26.95, 27.61, 27.84,
28.42, 28.779, 29.68, 30.06, 30.98, 32.40, 32.90, 33.89, 34.50, 35.13, 36.19, and 37.01.
[00165] In another embodiment, Compound 1 sodium salt Form C is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising peaks at 12.88, 15.04, and 16.99.
[00166] In another embodiment, Compound 1 sodium salt Form C is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising peaks at 5.85, 12.88, 15.04, 16.99, and
24.42.
[00167] In another embodiment, Compound 1 sodium salt Form C is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising peaks at 5.85, 12.88, 15.04, 16.67, 16.99, 18.06, 18.38, 21.48, 24.42, and 26.36.
[00168] In another embodiment, Compound 1 sodium salt Form C is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising all of the peaks 3.11, 5.85, 11.81, 12.20, 12.58, 12.88, 13.46, 14.27, 15.04, 15.98, 16.67, 16.99, 17.68, 18.06, 18.38, 18.79, 19.32, 19.89,
20.43, 20.93, 21.48, 22.17, 23.31, 23.90, 24.42, 24.81, 26.36, 26.95, 27.61, 27.84, 28.42, 28.779, 29.68, 30.06, 30.98, 32.40, 32.90, 33.89, 34.50, 35.13, 36.19, and 37.01.
[00169] In another embodiment, Compound 1 sodium salt Form C is characterized by the
XRPD pattern substantially in accordance with FIG. 16A.
[00170] A list of the peaks from the XRPD pattern for Compound 1 sodium salt Form C is provided in Table 4 below.
[00171] In another embodiment, Compound 1 sodium salt Form C is characterized by the TGA and DSC thermograms substantially in accordance with FIG. 17A.
[00172] In another embodiment, Compound 1 sodium salt Form C is characterized by a DSC thermogram showing endothermic peak onset at about 37 °C - 41 °C and 126 °C - 130 °C.
[00173] In another embodiment, Compound 1 sodium salt Form C is characterized by a TGA thermogram showing about 0.8 - 1.0 percent weight loss from 30 °C to 75 °C and about 4 - 5 percent weight loss from 80 °C to 190 °C.
[00174] In another embodiment, Compound 1 sodium salt Form C is characterized by the TGA and DSC thermograms showing about 0.9 percent weight loss from 30 °C to 75 °C and about 3.1 percent weight loss from 80 °C to 190 °C, and showing endothermic peak onset at about 39.3 °C and about 127.9 °C.
[00175] In another embodiment, Compound 1 sodium salt Form C is characterized by the FT- IR substantially in accordance with FIG. 18.
[00176] In another embodiment, Compound 1 sodium salt Form C is characterized by the 'H NMR spectrum in d6-DMSO substantially in accordance with FIG. 19.
[00177] In another embodiment, Compound 1 sodium salt Form C is characterized by at least one of the following:
(1) an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 3.11, 5.85, 11.81, 12.20, 12.58, 12.88, 13.46, 14.27, 15.04, 15.98, 16.67, 16.99, 17.68, 18.06, 18.38, 18.79, 19.32, 19.89, 20.43, 20.93, 21.48, 22.17, 23.31, 23.90, 24.42, 24.81, 26.36, 26.95, 27.61, 27.84, 28.42, 28.779, 29.68, 30.06, 30.98, 32.40, 32.90, 33.89, 34.50, 35.13, 36.19, and 37.01;
(2) a TGA thermogram substantially in accordance with FIG. 17A;
(3) a DSC thermogram substantially in accordance with FIG. 17A; and/or
(4) an FT-IR spectrum substantially in accordance with FIG. 18.
[00178] In another embodiment, Compound 1 sodium salt Form C is characterized by at least one of the following:
(1) an XRPD an XRPD pattern substantially in accordance with FIG. 16A;
(2) a TGA thermogram substantially in accordance with FIG. 17A;
(3) a DSC thermogram substantially in accordance with FIG. 17A; and/or
(4) an FT-IR spectrum substantially in accordance with FIG. 18.
[00179] In another embodiment, Compound 1 sodium salt Form C is characterized by at least two of the following:
(1) an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 3.11, 5.85, 11.81, 12.20, 12.58, 12.88, 13.46, 14.27, 15.04, 15.98, 16.67, 16.99,
17.68, 18.06, 18.38, 18.79, 19.32, 19.89, 20.43, 20.93, 21.48, 22.17, 23.31, 23.90, 24.42, 24.81,
26.36, 26.95, 27.61, 27.84, 28.42, 28.779, 29.68, 30.06, 30.98, 32.40, 32.90, 33.89, 34.50, 35.13,
36.19, and 37.01;
(2) a TGA thermogram substantially in accordance with FIG. 17A;
(3) a DSC thermogram substantially in accordance with FIG. 17A; and/or
(4) an FT-IR spectrum substantially in accordance with FIG. 18.
[00180] In another embodiment, Compound 1 sodium salt Form C is characterized by at least three of the following:
(1) an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 3.11, 5.85, 11.81, 12.20, 12.58, 12.88, 13.46, 14.27, 15.04, 15.98, 16.67, 16.99,
17.68, 18.06, 18.38, 18.79, 19.32, 19.89, 20.43, 20.93, 21.48, 22.17, 23.31, 23.90, 24.42, 24.81,
26.36, 26.95, 27.61, 27.84, 28.42, 28.779, 29.68, 30.06, 30.98, 32.40, 32.90, 33.89, 34.50, 35.13,
36.19, and 37.01;
(2) a TGA thermogram substantially in accordance with FIG. 17A;
(3) a DSC thermogram substantially in accordance with FIG. 17A; and/or
(4) an FT-IR spectrum substantially in accordance with FIG. 18.
[00181] In another embodiment, Compound 1 sodium salt Form C is characterized by all of the following:
(1) an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 3.11, 5.85, 11.81, 12.20, 12.58, 12.88, 13.46, 14.27, 15.04, 15.98, 16.67, 16.99,
17.68, 18.06, 18.38, 18.79, 19.32, 19.89, 20.43, 20.93, 21.48, 22.17, 23.31, 23.90, 24.42, 24.81,
26.36, 26.95, 27.61, 27.84, 28.42, 28.779, 29.68, 30.06, 30.98, 32.40, 32.90, 33.89, 34.50, 35.13,
36.19, and 37.01;
(2) a TGA thermogram substantially in accordance with FIG. 17A;
(3) a DSC thermogram substantially in accordance with FIG. 17A; and
(4) an FT-IR spectrum substantially in accordance with FIG. 18.
[00182] In another embodiment, Compound 1 sodium salt Form C is characterized by (1) an XRPD pattern substantially in accordance with FIG. 16A and at least one of the following:
(2) a TGA thermogram substantially in accordance with FIG. 17A;
(3) a DSC thermogram substantially in accordance with FIG. 17A; and/or
(4) an FT-IR spectrum substantially in accordance with FIG. 18.
[00183] In another embodiment, Compound 1 sodium salt Form C is characterized by (1) an XRPD pattern substantially in accordance with FIG. 16A and at least two of the following:
(2) a TGA thermogram substantially in accordance with FIG. 17A;
(3) a DSC thermogram substantially in accordance with FIG. 17A; and/or
(4) an FT-IR spectrum substantially in accordance with FIG. 18.
[00184] In another embodiment, Compound 1 sodium salt Form C is characterized by (1) an XRPD pattern substantially in accordance with FIG. 16A and all of the following:
(2) a TGA thermogram substantially in accordance with FIG. 17A;
(3) a DSC thermogram substantially in accordance with FIG. 17A; and
(4) an FT-IR spectrum substantially in accordance with FIG. 18.
Compound 1 Sodium Salt Form D
[00185] In one embodiment, Compound 1 sodium crystalline salt is characterized as Compound 1 sodium salt Form D. In another embodiment, Compound 1 sodium salt is a hydrate characterized as Compound 1 sodium salt Form D (1 : 1) (stoichiometric ratio of Compound 1 : Na1).
[00186] The XRPD pattern of Compound 1 sodium salt Form D was recorded in transmission geometry.
[00187] In another embodiment, Compound 1 sodium salt Form D is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 5.55,
11.08, 12.23, 12.47, 12.84, 13.05, 13.88, 14.10, 14.73, 15.56, 16.47, 17.09, 17.85, 18.29, 18.63,
19.10, 19.63, 20.31, 20.66, 21.03, 21.80, 22.30, 23.51, 24.57, 25.09, 25.43, 26.13, 26.43, 27.55,
27.96, 28.60, 29.02, 29.67, 30.54, 30.94, 31.85, 34.56, 35.00, 36.02, and 39.50.
[00188] In another embodiment, Compound 1 sodium salt Form D is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising three or more peaks selected from 5.55, 11.08, 12.23, 12.47, 12.84, 13.05, 13.88, 14.10, 14.73, 15.56, 16.47, 17.09, 17.85, 18.29, 18.63,
19.10, 19.63, 20.31, 20.66, 21.03, 21.80, 22.30, 23.51, 24.57, 25.09, 25.43, 26.13, 26.43, 27.55,
27.96, 28.60, 29.02, 29.67, 30.54, 30.94, 31.85, 34.56, 35.00, 36.02, and 39.50.
[00189] In another embodiment, Compound 1 sodium salt Form D is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising five or more peaks selected from 5.55, 11.08, 12.23, 12.47, 12.84, 13.05, 13.88, 14.10, 14.73, 15.56, 16.47, 17.09, 17.85, 18.29, 18.63,
19.10, 19.63, 20.31, 20.66, 21.03, 21.80, 22.30, 23.51, 24.57, 25.09, 25.43, 26.13, 26.43, 27.55,
27.96, 28.60, 29.02, 29.67, 30.54, 30.94, 31.85, 34.56, 35.00, 36.02, and 39.50.
[00190] In another embodiment, Compound 1 sodium salt Form D is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising ten or more peaks selected from 5.55, 11.08, 12.23, 12.47, 12.84, 13.05, 13.88, 14.10, 14.73, 15.56, 16.47, 17.09, 17.85, 18.29, 18.63,
19.10, 19.63, 20.31, 20.66, 21.03, 21.80, 22.30, 23.51, 24.57, 25.09, 25.43, 26.13, 26.43, 27.55,
27.96, 28.60, 29.02, 29.67, 30.54, 30.94, 31.85, 34.56, 35.00, 36.02, and 39.50.
[00191] In another embodiment, Compound 1 sodium salt Form D is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising all of the peaks 5.55, 11.08, 12.23, 12.47, 12.84, 13.05, 13.88, 14.10, 14.73, 15.56, 16.47, 17.09, 17.85, 18.29, 18.63, 19.10, 19.63, 20.31,
20.66, 21.03, 21.80, 22.30, 23.51, 24.57, 25.09, 25.43, 26.13, 26.43, 27.55, 27.96, 28.60, 29.02,
29.67, 30.54, 30.94, 31.85, 34.56, 35.00, 36.02, and 39.50.
[00192] In another embodiment, Compound 1 sodium salt Form D is characterized by the XRPD pattern substantially in accordance with FIG. 16B.
[00193] A list of the peaks from the XRPD pattern for Compound 1 sodium salt Form C is provided in Table 4B below.
[00194] In another embodiment, Compound 1 sodium salt Form D is characterized by a TG- FTIR thermograms substantially in accordance with FIG. 17B.
[00195] In another embodiment, Compound 1 sodium salt Form D is characterized by a DSC thermograms substantially in accordance with FIG. 17C.
[00196] In another embodiment, Compound 1 sodium salt Form D is characterized by a DSC thermogram showing a first endothermal event at 65.4 °C with an onset at 36.5 °C. Then, the melting peak is observed at 129.4 °C, with an onset at 115.9°C.
[00197] In another embodiment, Compound 1 sodium salt Form C is characterized by at least one of the following:
(1) an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 5.55, 11.08, 12.23, 12.47, 12.84, 13.05, 13.88, 14.10, 14.73, 15.56, 16.47, 17.09,
17.85, 18.29, 18.63, 19.10, 19.63, 20.31, 20.66, 21.03, 21.80, 22.30, 23.51, 24.57, 25.09, 25.43,
26.13, 26.43, 27.55, 27.96, 28.60, 29.02, 29.67, 30.54, 30.94, 31.85, 34.56, 35.00, 36.02, and 39.50;
(2) a TG-FTIR thermogram substantially in accordance with FIG. 17B; and/or
(3) a DSC thermogram substantially in accordance with FIG. 17C.
[00198] In another embodiment, Compound 1 sodium salt Form D is characterized by at least two of the following:
(1) an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 5.55, 11.08, 12.23, 12.47, 12.84, 13.05, 13.88, 14.10, 14.73, 15.56, 16.47, 17.09,
17.85, 18.29, 18.63, 19.10, 19.63, 20.31, 20.66, 21.03, 21.80, 22.30, 23.51, 24.57, 25.09, 25.43,
26.13, 26.43, 27.55, 27.96, 28.60, 29.02, 29.67, 30.54, 30.94, 31.85, 34.56, 35.00, 36.02, and 39.50;
(2) a TG-FTIR thermogram substantially in accordance with FIG. 17B; and/or
(3) a DSC thermogram substantially in accordance with FIG. 17C.
[00199] In another embodiment, Compound 1 sodium salt Form D is characterized by all of the following:
(1) an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 5.55, 11.08, 12.23, 12.47, 12.84, 13.05, 13.88, 14.10, 14.73, 15.56, 16.47, 17.09,
17.85, 18.29, 18.63, 19.10, 19.63, 20.31, 20.66, 21.03, 21.80, 22.30, 23.51, 24.57, 25.09, 25.43,
26.13, 26.43, 27.55, 27.96, 28.60, 29.02, 29.67, 30.54, 30.94, 31.85, 34.56, 35.00, 36.02, and 39.50;
(2) a TG-FTIR thermogram substantially in accordance with FIG. 17B; and
(3) a DSC thermogram substantially in accordance with FIG. 17C.
[00200] In another embodiment, Compound 1 sodium salt Form D is characterized by (1) an
XRPD pattern substantially in accordance with FIG. 16B and at least one of the following:
(2) a TG-FTIR thermogram substantially in accordance with FIG. 17B; and/or
(3) a DSC thermogram substantially in accordance with FIG. 17C.
[00201] In another embodiment, Compound 1 sodium salt Form D is characterized by (1) an
XRPD pattern substantially in accordance with FIG. 16B and:
(2) a TG-FTIR thermogram substantially in accordance with FIG. 17B; and
(3) a DSC thermogram substantially in accordance with FIG. 17C.
Compound 1 Erbumine Salt Form A
[00202] In one embodiment, the salt is Compound 1 erbumine salt. In another embodiment, Compound 1 erbumine salt is crystalline. In one embodiment, Compound 1 erbumine crystalline salt is characterized as Compound 1 erbumine salt Form A. In a further embodiment, Compound
1 erbumine salt is characterized as Compound 1 erbumine salt Form A (1 : 1.0) (stoichiometric ratio of Compound 1 : erbumine).
[00203] The XRPD pattern of Compound 1 erbumine salt Form A was recorded in reflection geometry.
[00204] In another embodiment, Compound 1 erbumine salt Form A is characterized by an
XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 9.46,
10.26, 11.24, 11.90, 16.94, 17.40, 18.73, 19.40, 20.28, 20.95, and 22.12.
[00205] In another embodiment, Compound 1 erbumine salt Form A is characterized by an
XRPD pattern on a 2 Theta scale, ± 0.20, comprising three or more peaks selected from 9.46,
10.26, 11.24, 11.90, 16.94, 17.40, 18.73, 19.40, 20.28, 20.95, and 22.12.
[00206] In another embodiment, Compound 1 erbumine salt Form A is characterized by an
XRPD pattern on a 2 Theta scale, ± 0.20, comprising five or more peaks selected from 9.46,
10.26, 11.24, 11.90, 16.94, 17.40, 18.73, 19.40, 20.28, 20.95, and 22.12.
[00207] In another embodiment, Compound 1 erbumine salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising peaks at 10.26, 11.90, and 19.40.
[00208] In another embodiment, Compound 1 erbumine salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising peaks at 9.46, 10.26, 11.90, 19.40, and 20.28.
[00209] In another embodiment, Compound 1 erbumine salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising all of the peaks 9.46, 10.26, 11.24, 11.90, 16.94, 17.40, 18.73, 19.40, 20.28, 20.95, and 22.12.
[00210] In another embodiment, Compound 1 erbumine salt Form A is characterized by the XRPD pattern substantially in accordance with FIG. 20.
[00211] A list of the peaks from the XRPD pattern for Compound 1 erbumine salt Form A is provided in Table 5 below.
[00212] In another embodiment, Compound 1 erbumine salt Form A is characterized by the XRPD pattern substantially in accordance with FIG. 20.
[00213] In another embodiment, Compound 1 erbumine salt Form A is characterized by the DSC thermogram substantially in accordance with FIG. 21 indicating decomposition upon melting.
[00214] In another embodiment, Compound 1 erbumine salt Form A is characterized by the TGA thermogram substantially in accordance with FIG. 22.
[00215] In another embodiment, Compound 1 erbumine salt Form A is characterized by the ’l l NMR spectrum in d6-DMSO substantially in accordance with FIG. 23 indicating no detectable residual solvent.
[00216] In another embodiment, Compound 1 erbumine salt Form A is characterized by at least one of the following:
(1) an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 9.46, 10.26, 11.24, 11.90, 16.94, 17.40, 18.73, 19.40, 20.28, 20.95, and 22.12;
(2) an DSC thermogram substantially in accordance with FIG. 21; and/or
(3) a TGA thermogram substantially in accordance with FIG. 22.
[00217] In another embodiment, Compound 1 erbumine salt Form A is characterized by at least one of the following:
(1) an XRPD pattern substantially in accordance with FIG. 20;
(2) an DSC thermogram substantially in accordance with FIG. 21; and/or
(3) a TGA thermogram substantially in accordance with FIG. 22.
[00218] In another embodiment, Compound 1 erbumine salt Form A is characterized by at least two of the following:
(1) an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 9.46, 10.26, 11.24, 11.90, 16.94, 17.40, 18.73, 19.40, 20.28, 20.95, and 22.12;
(2) an DSC thermogram substantially in accordance with FIG. 21; and/or
(3) a TGA thermogram substantially in accordance with FIG. 22.
[00219] In another embodiment, Compound 1 erbumine salt Form A is characterized by all of the following:
(1) an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 9.46, 10.26, 11.24, 11.90, 16.94, 17.40, 18.73, 19.40, 20.28, 20.95, and 22.12;
(2) an DSC thermogram substantially in accordance with FIG. 21; and
(3) a TGA thermogram substantially in accordance with FIG. 22.
[00220] In another embodiment, Compound 1 erbumine salt Form A is characterized by (1) an
XRPD pattern substantially in accordance with FIG. 20 and at least one of the following:
(2) an DSC thermogram substantially in accordance with FIG. 21; and/or
(3) a TGA thermogram substantially in accordance with FIG. 22.
[00221] In another embodiment, Compound 1 erbumine salt Form A is characterized by (1) an XRPD pattern substantially in accordance with FIG. 20 and:
(2) an DSC thermogram substantially in accordance with FIG. 21; and
(3) a TGA thermogram substantially in accordance with FIG. 22.
Compound 1 Erbumine Salt Form B
[00222] In one embodiment, Compound 1 erbumine crystalline salt is characterized as Compound 1 erbumine salt Form B.
[00223] The XRPD pattern of Compound 1 erbumine salt Form B was recorded in reflection geometry.
[00224] In another embodiment, Compound 1 erbumine salt Form B is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 4.74, 5.52, 7.39, 9.40, 9.84, 11.04, 12.35, 14.29, 15.09, 16.49, 17.76, 18.09, 19.26, 20.07, 20.57, 21.06,
22.14, and 23.72.
[00225] In another embodiment, Compound 1 erbumine salt Form B is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising three or more peaks selected from 4.74, 5.52, 7.39, 9.40, 9.84, 11.04, 12.35, 14.29, 15.09, 16.49, 17.76, 18.09, 19.26, 20.07, 20.57, 21.06,
22.14, and 23.72.
[00226] In another embodiment, Compound 1 erbumine salt Form B is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising five or more peaks selected from 4.74, 5.52, 7.39, 9.40, 9.84, 11.04, 12.35, 14.29, 15.09, 16.49, 17.76, 18.09, 19.26, 20.07, 20.57, 21.06,
22.14, and 23.72.
[00227] In another embodiment, Compound 1 erbumine salt Form B is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising ten or more peaks selected from 4.74, 5.52, 7.39, 9.40, 9.84, 11.04, 12.35, 14.29, 15.09, 16.49, 17.76, 18.09, 19.26, 20.07, 20.57, 21.06,
22.14, and 23.72.
[00228] In another embodiment, Compound 1 erbumine salt Form B is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising peaks at 9.84, 11.04, and 19.26.
[00229] In another embodiment, Compound 1 erbumine salt Form B is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising peaks at 9.84, 11.04, 19.26, 20.07, and 21.06.
[00230] In another embodiment, Compound 1 erbumine salt Form B is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising peaks at 5.52, 9.84, 11.04, 17.76, 18.09, 19.26, 20.07, 21.06, 22.14, and 23.72.
[00231] In another embodiment, Compound 1 erbumine salt Form B is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising all of the peaks 4.74, 5.52, 7.39, 9.40, 9.84, 11.04, 12.35, 14.29, 15.09, 16.49, 17.76, 18.09, 19.26, 20.07, 20.57, 21.06, 22.14, and 23.72.
[00232] In another embodiment, Compound 1 erbumine salt Form B is characterized by the XRPD pattern substantially in accordance with FIG. 24.
[00233] A list of the peaks from the XRPD pattern for Compound 1 erbumine salt Form B is provided in Table 6 below.
Compound 1 Erbumine Salt Form C
[00234] In one embodiment, Compound 1 erbumine crystalline salt is characterized as Compound 1 erbumine salt Form C. In a further embodiment, Compound 1 erbumine salt is characterized as Compound 1 erbumine salt Form C (1:1.0) (stoichiometric ratio of Compound 1 : erbumine).
[00235] The XRPD pattern of Compound 1 erbumine salt Form C was recorded in reflection geometry.
[00236] In another embodiment, Compound 1 erbumine salt Form C is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 4.00, 7.38, 7.97, 8.48, 9.89, 11.04, 11.36, 13.85, 14.30, 14.77, 15.60, 15.95, 16.14, 16.59, 17.44, 17.64, 18.72, 19.11, 19.54, 19.79, 20.18, 20.62, 21.02, 21.48, 22.20, 22.47, 23.29, 23.79, 25.20, 25.54, 27.21, 27.577, 31.20, 32.00, and 32.70.
[00237] In another embodiment, Compound 1 erbumine salt Form C is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising three or more peaks selected from 4.00, 7.38, 7.97, 8.48, 9.89, 11.04, 11.36, 13.85, 14.30, 14.77, 15.60, 15.95, 16.14, 16.59, 17.44, 17.64,
18.72, 19.11, 19.54, 19.79, 20.18, 20.62, 21.02, 21.48, 22.20, 22.47, 23.29, 23.79, 25.20, 25.54,
27.21, 27.577, 31.20, 32.00, and 32.70.
[00238] In another embodiment, Compound 1 erbumine salt Form C is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising five or more peaks selected from 4.00, 7.38, 7.97, 8.48, 9.89, 11.04, 11.36, 13.85, 14.30, 14.77, 15.60, 15.95, 16.14, 16.59, 17.44, 17.64,
18.72, 19.11, 19.54, 19.79, 20.18, 20.62, 21.02, 21.48, 22.20, 22.47, 23.29, 23.79, 25.20, 25.54,
27.21, 27.577, 31.20, 32.00, and 32.70.
[00239] In another embodiment, Compound 1 erbumine salt Form C is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising ten or more peaks selected from 4.00, 7.38, 7.97, 8.48, 9.89, 11.04, 11.36, 13.85, 14.30, 14.77, 15.60, 15.95, 16.14, 16.59, 17.44, 17.64,
18.72, 19.11, 19.54, 19.79, 20.18, 20.62, 21.02, 21.48, 22.20, 22.47, 23.29, 23.79, 25.20, 25.54,
27.21, 27.577, 31.20, 32.00, and 32.70.
[00240] In another embodiment, Compound 1 erbumine salt Form C is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising peaks at 16.59, 19.79, and 21.48.
[00241] In another embodiment, Compound 1 erbumine salt Form C is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising peaks at 8.48, 16.59, 19.79, 21.48, and 22.20.
[00242] In another embodiment, Compound 1 erbumine salt Form C is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising peaks at 8.48, 16.59, 17.44, 17.64, 19.54, 19.79, 20.18, 21.48, 22.20, and 23.29.
[00243] In another embodiment, Compound 1 erbumine salt Form C is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising all of the peaks 4.00, 7.38, 7.97, 8.48, 9.89, 11.04, 11.36, 13.84, 14.30, 14.77, 15.60, 15.95, 16.14, 16.59, 17.44, 17.64, 18.72, 19.11, 19.58, 19.79, 20.18, 20.62, 21.02, 21.48, 22.20, 22.47, 23.29, 23.79, 25.20, 25.54, 27.21, 27.577, 31.20, 32.00, and 32.70.
[00244] In another embodiment, Compound 1 erbumine salt Form C is characterized by the XRPD pattern substantially in accordance with FIG. 25.
[00245] A list of the peaks from the XRPD pattern for Compound 1 erbumine salt Form C is provided in Table 7 below.
[00246] In another embodiment, Compound 1 erbumine salt Form C is characterized by the DSC thermogram substantially in accordance with FIG. 26 indicating decomposition upon melting.
[00247] In another embodiment, Compound 1 erbumine salt Form C is characterized by the TGA thermogram substantially in accordance with FIG. 27.
[00248] In another embodiment, Compound 1 erbumine salt Form C is characterized by the 1 H NMR spectrum in d6-DMSO substantially in accordance with FIG. 28.
[00249] In another embodiment, Compound 1 erbumine salt Form C is characterized by at least one of the following:
(1) an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 4.00, 7.38, 7.97, 8.48, 9.89, 11.04, 11.36, 13.85, 14.30, 14.77, 15.60, 15.95, 16.14,
16.59, 17.44, 17.64, 18.72, 19.11, 19.54, 19.79, 20.18, 20.62, 21.02, 21.48, 22.20, 22.47, 23.29,
23.79, 25.20, 25.54, 27.21, 27.577, 31.20, 32.00, and 32.70;
(2) an DSC thermogram substantially in accordance with FIG. 26; and/or
(3) a TGA thermogram substantially in accordance with FIG. 27.
[00250] In another embodiment, Compound 1 erbumine salt Form C is characterized by at least two of the following:
(1) an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 4.00, 7.38, 7.97, 8.48, 9.89, 11.04, 11.36, 13.85, 14.30, 14.77, 15.60, 15.95, 16.14,
16.59, 17.44, 17.64, 18.72, 19.11, 19.54, 19.79, 20.18, 20.62, 21.02, 21.48, 22.20, 22.47, 23.29,
23.79, 25.20, 25.54, 27.21, 27.577, 31.20, 32.00, and 32.70;
(2) an DSC thermogram substantially in accordance with FIG. 26; and/or
(3) a TGA thermogram substantially in accordance with FIG. 27.
[00251] In another embodiment, Compound 1 erbumine salt Form C is characterized by all of the following:
(1) an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 4.00, 7.38, 7.97, 8.48, 9.89, 11.04, 11.36, 13.85, 14.30, 14.77, 15.60, 15.95, 16.14,
16.59, 17.44, 17.64, 18.72, 19.11, 19.54, 19.79, 20.18, 20.62, 21.02, 21.48, 22.20, 22.47, 23.29,
23.79, 25.20, 25.54, 27.21, 27.577, 31.20, 32.00, and 32.70;
(2) an DSC thermogram substantially in accordance with FIG. 26; and
(3) a TGA thermogram substantially in accordance with FIG. 27.
[00252] In another embodiment, Compound 1 erbumine salt Form C is characterized by (1) an XRPD pattern substantially in accordance with FIG. 25 and at least one of the following:
(2) an DSC thermogram substantially in accordance with FIG. 26; and/or
(3) a TGA thermogram substantially in accordance with FIG. 27.
[00253] In another embodiment, Compound 1 erbumine salt Form C is characterized by (1) an XRPD pattern substantially in accordance with FIG. 25 and:
(2) an DSC thermogram substantially in accordance with FIG. 26; and
(3) a TGA thermogram substantially in accordance with FIG. 27.
Compound 1 Erbumine Salt Form D
[00254] In one embodiment, Compound 1 erbumine crystalline salt is characterized as Compound 1 erbumine salt Form D. In a further embodiment, Compound 1 erbumine salt is characterized as Compound 1 erbumine salt Form D (1:0.4) (stoichiometric ratio of Compound 1 : erbumine).
[00255] The XRPD pattern of Compound 1 erbumine salt Form D was recorded in reflection geometry.
[00256] In another embodiment, Compound 1 erbumine salt Form D is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 3.19, 5.77, 6.50, 8.86, 9.30, 9.69, 10.59, 11.24, 11.71, 11.96, 12.47, 12.70, 13.38, 14.01, 14.77, 15.48, 16.22, 17.34, 17.77, 18.01, 18.33, 18.75, 19.24, 19.49, 19.78, 20.36, 20.56, 20.89, 21.31, 21.84, 22.24, 23.26, 23.59, 24.32, 24.93, 25.22, 26.83, 27.09, 27.75, 28.32, 28.99, 29.38, 29.76, 31.40, 32.18, 32.39, 33.67, 34.24 , 34.84, 35.37, and 37.34.
[00257] In another embodiment, Compound 1 erbumine salt Form D is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising three or more peaks selected from 3.19, 5.77, 6.50, 8.86, 9.30, 9.69, 10.59, 11.24, 11.71, 11.96, 12.47, 12.70, 13.38, 14.01, 14.77, 15.48, 16.22, 17.34, 17.77, 18.01, 18.33, 18.75, 19.24, 19.49, 19.78, 20.36, 20.56, 20.89, 21.31, 21.84, 22.24, 23.26, 23.59, 24.32, 24.93, 25.22, 26.83, 27.09, 27.75, 28.32, 28.99, 29.38, 29.76, 31.40, 32.18, 32.39, 33.67, 34.24 , 34.84, 35.37, and 37.34.
[00258] In another embodiment, Compound 1 erbumine salt Form D is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising five or more peaks selected from 3.19, 5.77, 6.50, 8.86, 9.30, 9.69, 10.59, 11.24, 11.71, 11.96, 12.47, 12.70, 13.38, 14.01, 14.77, 15.48, 16.22, 17.34, 17.77, 18.01, 18.33, 18.75, 19.24, 19.49, 19.78, 20.36, 20.56, 20.89, 21.31, 21.84, 22.24, 23.26, 23.59, 24.32, 24.93, 25.22, 26.83, 27.09, 27.75, 28.32, 28.99, 29.38, 29.76, 31.40, 32.18, 32.39, 33.67, 34.24 , 34.84, 35.37, and 37.34.
[00259] In another embodiment, Compound 1 erbumine salt Form D is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising ten or more peaks selected from 3.19, 5.77, 6.50, 8.86, 9.30, 9.69, 10.59, 11.24, 11.71, 11.96, 12.47, 12.70, 13.38, 14.01, 14.77, 15.48, 16.22, 17.34, 17.77, 18.01, 18.33, 18.75, 19.24, 19.49, 19.78, 20.36, 20.56, 20.89, 21.31, 21.84, 22.24,
23.26, 23.59, 24.32, 24.93, 25.22, 26.83, 27.09, 27.75, 28.32, 28.99, 29.38, 29.76, 31.40, 32.18, 32.39, 33.67, 34.24 , 34.84, 35.37, and 37.34.
[00260] In another embodiment, Compound 1 erbumine salt Form D is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising peaks at 9.30, 20.36, and 23.59.
[00261] In another embodiment, Compound 1 erbumine salt Form D is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising peaks at 9.30, 20.36, 20.89, 22.24, and 23.59.
[00262] In another embodiment, Compound 1 erbumine salt Form D is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising peaks at 9.30, 10.59, 14.01, 17.77, 19.24, 19.49, 20.36, 20.89, 22.24, and 23.59.
[00263] In another embodiment, Compound 1 erbumine salt Form D is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising all of the peaks 3.19, 5.77, 6.50, 8.86, 9.30, 9.69, 10.59, 11.24, 11.71, 11.96, 12.47, 12.70, 13.38, 14.01, 14.77, 15.48, 16.22, 17.34, 17.77, 18.01, 18.33, 18.75, 19.24, 19.49, 19.78, 20.36, 20.56, 20.89, 21.31, 21.84, 22.24, 23.26, 23.59, 24.32, 24.93, 25.22, 26.83, 27.09, 27.75, 28.32, 28.99, 29.38, 29.76, 31.40, 32.18, 32.39, 33.67, 34.24 , 34.84, 35.37, and 37.34.
[00264] In another embodiment, Compound 1 erbumine salt Form D is characterized by the XRPD pattern substantially in accordance with FIG. 29.
[00265] A list of the peaks from the XRPD pattern for Compound 1 erbumine salt Form D is provided in Table 8 below.
[00266] In another embodiment, Compound 1 erbumine salt Form D is characterized by the TGA and DSC thermograms substantially in accordance with FIG. 30.
[00267] In another embodiment, Compound 1 erbumine salt Form D is characterized by the TGA and DSC thermograms showing endothermic peak onsets at about 116 °C - 120 °C and 174 °C - 178 °C.
[00268] In another embodiment, Compound 1 erbumine salt Form D is characterized by the TGA and DSC thermograms showing endothermic peak onsets at about 117.8 and about 176.3 °C.
[00269] In another embodiment, Compound 1 erbumine salt Form D is characterized by the 'HNMR spectrum in d6-DMSO substantially in accordance with FIG. 31 indicating no residual solvent.
[00270] In another embodiment, Compound 1 erbumine salt Form D is characterized by at least one of the following:
(1) an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 3.19, 5.77, 6.50, 8.86, 9.30, 9.69, 10.59, 11.24, 11.71, 11.96, 12.47, 12.70, 13.38,
14.01, 14.77, 15.48, 16.22, 17.34, 17.77, 18.01, 18.33, 18.75, 19.24, 19.49, 19.78, 20.36, 20.56,
20.89, 21.31, 21.84, 22.24, 23.26, 23.59, 24.32, 24.93, 25.22, 26.83, 27.09, 27.75, 28.32, 28.99,
29.38, 29.76, 31.40, 32.18, 32.39, 33.67, 34.24 , 34.84, 35.37, and 37.34;
(2) a TGA thermogram substantially in accordance with FIG. 30; and/or
(3) a DSC thermogram substantially in accordance with FIG. 30.
[00271] In another embodiment, Compound 1 erbumine salt Form D is characterized by at least one of the following:
(1) an XRPD pattern substantially in accordance with FIG. 29;
(2) a TGA thermogram substantially in accordance with FIG. 30; and/or
(3) a DSC thermogram substantially in accordance with FIG. 30.
[00272] In another embodiment, Compound 1 erbumine salt Form D is characterized by at least two of the following:
(1) an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 3.19, 5.77, 6.50, 8.86, 9.30, 9.69, 10.59, 11.24, 11.71, 11.96, 12.47, 12.70, 13.38,
14.01, 14.77, 15.48, 16.22, 17.34, 17.77, 18.01, 18.33, 18.75, 19.24, 19.49, 19.78, 20.36, 20.56,
20.89, 21.31, 21.84, 22.24, 23.26, 23.59, 24.32, 24.93, 25.22, 26.83, 27.09, 27.75, 28.32, 28.99,
29.38, 29.76, 31.40, 32.18, 32.39, 33.67, 34.24 , 34.84, 35.37, and 37.34;
(2) a TGA thermogram substantially in accordance with FIG. 30; and/or
(3) a DSC thermogram substantially in accordance with FIG. 30.
[00273] In another embodiment, Compound 1 erbumine salt Form D is characterized by all of the following:
(1) an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 3.19, 5.77, 6.50, 8.86, 9.30, 9.69, 10.59, 11.24, 11.71, 11.96, 12.47, 12.70, 13.38, 14.01, 14.77, 15.48, 16.22, 17.34, 17.77, 18.01, 18.33, 18.75, 19.24, 19.49, 19.78, 20.36, 20.56, 20.89, 21.31, 21.84, 22.24, 23.26, 23.59, 24.32, 24.93, 25.22, 26.83, 27.09, 27.75, 28.32, 28.99, 29.38, 29.76, 31.40, 32.18, 32.39, 33.67, 34.24 , 34.84, 35.37, and 37.34;
(2) a TGA thermogram substantially in accordance with FIG. 30; and
(3) a TGA thermogram substantially in accordance with FIG. 30.
[00274] In another embodiment, Compound 1 erbumine salt Form D is characterized by (1) an XRPD pattern substantially in accordance with FIG. 29 and at least one of the following:
(2) a TGA thermogram substantially in accordance with FIG. 30; and/or
(3) a DSC thermogram substantially in accordance with FIG. 30.
[00275] In another embodiment, Compound 1 erbumine salt Form D is characterized by (1) an XRPD pattern substantially in accordance with FIG. 29 and:
(2) a TGA thermogram substantially in accordance with FIG. 30; and
(3) a DSC thermogram substantially in accordance with FIG. 30.
Compound 1 Erbumine Salt Form E
[00276] In one embodiment, Compound 1 erbumine crystalline salt is characterized as Compound 1 erbumine salt Form E. In a further embodiment, Compound 1 erbumine salt is characterized as Compound 1 erbumine salt Form E (1 :1.0) (stoichiometric ratio of Compound 1 : erbumine)
[00277] The XRPD pattern of Compound 1 erbumine salt Form E was recorded in reflection geometry.
[00278] In another embodiment, Compound 1 erbumine salt Form E is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 5.85, 7.49, 8.43, 9.62, 10.43, 11.28, 11.68, 12.53, 13.25, 13.71, 14.82, 15.12, 15.51, 16.37, 17.26, 17.61, 18.20, 18.71, 19.31, 19.91, 20.84, 21.93, 22.37, 23.26, 24.13, 25.96, 27.22, and 30.28.
[00279] In another embodiment, Compound 1 erbumine salt Form E is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising three or more peaks selected from 5.85, 7.49, 8.43, 9.62, 10.43, 11.28, 11.68, 12.53, 13.25, 13.71, 14.82, 15.12, 15.51, 16.37, 17.26, 17.61, 18.20, 18.71, 19.31, 19.91, 20.84, 21.93, 22.37, 23.26, 24.13, 25.96, 27.22, and 30.28.
[00280] In another embodiment, Compound 1 erbumine salt Form E is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising five or more peaks selected from 5.85, 7.49, 8.43, 9.62, 10.43, 11.28, 11.68, 12.53, 13.25, 13.71, 14.82, 15.12, 15.51, 16.37, 17.26, 17.61, 18.20, 18.71, 19.31, 19.91, 20.84, 21.93, 22.37, 23.26, 24.13, 25.96, 27.22, and 30.28.
[00281] In another embodiment, Compound 1 erbumine salt Form E is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising ten or more peaks selected from 5.85, 7.49,
8.43, 9.62, 10.43, 11.28, 11.68, 12.53, 13.25, 13.71, 14.82, 15.12, 15.51, 16.37, 17.26, 17.61, 18.20, 18.71, 19.31, 19.91, 20.84, 21.93, 22.37, 23.26, 24.13, 25.96, 27.22, and 30.28.
[00282] In another embodiment, Compound 1 erbumine salt Form E is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising peaks at 11.68, 18.71, and 19.31.
[00283] In another embodiment, Compound 1 erbumine salt Form E is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising peaks at 11.68, 18.20, 18.71, 19.31, and 19.91.
[00284] In another embodiment, Compound 1 erbumine salt Form E is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising peaks at 5.85, 11.68, 13.25, 18.20, 18.71, 19.31, 19.91, 23.26, 24.13, and 25.96.
[00285] In another embodiment, Compound 1 erbumine salt Form E is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising all of the peaks 5.85, 7.49, 8.43, 9.62,
10.43, 11.28, 11.68, 12.53, 13.25, 13.71, 14.82, 15.12, 15.51, 16.37, 17.26, 17.61, 18.20, 18.71, 19.31, 19.91, 20.84, 21.93, 22.37, 23.26, 24.13, 25.96, 27.22, and 30.28.
[00286] In another embodiment, Compound 1 erbumine salt Form E is characterized by the XRPD pattern substantially in accordance with FIG. 32.
[00287] A list of the peaks from the XRPD pattern for Compound 1 erbumine salt Form E is provided in Table 9 below.
[00288] In another embodiment, Compound 1 erbumine salt Form E is characterized by the TGA and DSC thermograms substantially in accordance with FIG. 33.
[00289] In another embodiment, Compound 1 erbumine salt Form E is characterized by a DSC thermogram showing endothermic peak onsets at about 0.6 °C - 1.0 °C and about 159 °C - 163 °C.
[00290] In another embodiment, Compound 1 erbumine salt Form E is characterized by a TGA thermogram showing about 1.0 - 1.2 percent weight loss from 25 °C to 100 °C.
[00291] In another embodiment, Compound 1 erbumine salt Form E is characterized by the TGA and DSC thermograms showing endothermic peak onsets at about 0.8 °C and about 161 °C and showing about 1.1 percent weight loss from 25 °C to 100 °C.
[00292] In another embodiment, Compound 1 erbumine salt Form E is characterized by the TH NMR spectrum in d6-DMSO substantially in accordance with FIG. 34 indicating 0.4 percent residual solvent.
[00293] In another embodiment, Compound 1 erbumine salt Form E is characterized by at least one of the following:
(1) an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 5.85, 7.49, 8.43, 9.62, 10.43, 11.28, 11.68, 12.53, 13.25, 13.71, 14.82, 15.12,
15.51, 16.37, 17.26, 17.61, 18.20, 18.71, 19.31, 19.91, 20.84, 21.93, 22.37, 23.26, 24.13, 25.96,
27.22, and 30.28;
(2) a TGA thermogram substantially in accordance with FIG. 33; and/or
(3) a DSC thermogram substantially in accordance with FIG. 33.
[00294] In another embodiment, Compound 1 erbumine salt Form E is characterized by at least one of the following:
(1) an XRPD pattern substantially in accordance with FIG. 32;
(2) a TGA thermogram substantially in accordance with FIG. 33; and/or
(3) a DSC thermogram substantially in accordance with FIG. 33.
[00295] In another embodiment, Compound 1 erbumine salt Form E is characterized by at least two of the following:
(1) an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 5.85, 7.49, 8.43, 9.62, 10.43, 11.28, 11.68, 12.53, 13.25, 13.71, 14.82, 15.12,
15.51, 16.37, 17.26, 17.61, 18.20, 18.71, 19.31, 19.91, 20.84, 21.93, 22.37, 23.26, 24.13, 25.96,
27.22, and 30.28;
(2) a TGA thermogram substantially in accordance with FIG. 33; and/or
(3) a DSC thermogram substantially in accordance with FIG. 33.
[00296] In another embodiment, Compound 1 erbumine salt Form E is characterized by all of the following:
(1) an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 5.85, 7.49, 8.43, 9.62, 10.43, 11.28, 11.68, 12.53, 13.25, 13.71, 14.82, 15.12,
15.51, 16.37, 17.26, 17.61, 18.20, 18.71, 19.31, 19.91, 20.84, 21.93, 22.37, 23.26, 24.13, 25.96,
27.22, and 30.28;
(2) a TGA thermogram substantially in accordance with FIG. 33; and
(3) a DSC thermogram substantially in accordance with FIG. 33.
[00297] In another embodiment, Compound 1 erbumine salt Form E is characterized by (1) an XRPD pattern substantially in accordance with FIG. 32 and at least one of the following:
(2) a DSC thermogram substantially in accordance with FIG. 33; and/or
(3) a TGA thermogram substantially in accordance with FIG. 33.
[00298] In another embodiment, Compound 1 erbumine salt Form E is characterized by (1) an XRPD pattern substantially in accordance with FIG. 32 and:
(2) a DSC thermogram substantially in accordance with FIG. 33; and
(3) a TGA thermogram substantially in accordance with FIG. 33.
Compound 1 Erbumine Salt Form F
[00299] In one embodiment, Compound 1 erbumine crystalline salt is characterized as Compound 1 erbumine salt Form F. In a further embodiment, Compound 1 erbumine salt is characterized as Compound 1 erbumine salt Form F (1 :1.0) (stoichiometric ratio of Compound 1 : erbumine).
[00300] The XRPD pattern of Compound 1 erbumine salt Form F was recorded in reflection geometry.
[00301] In another embodiment, Compound 1 erbumine salt Form F is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 3.17, 5.12, 5.78, 6.47, 7.67, 8.91, 9.72, 10.46, 11.24, 11.96, 13.46, 14.41, 14.80, 15.70, 16.38, 17.11, 17.40, 18.06, 18.38, 18.80, 19.59, 20.63, 20.86, 21.16, 21.94, 22.79, 23.66, 24.580 26.17, and 26.82.
[00302] In another embodiment, Compound 1 erbumine salt Form F is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising three or more peaks selected from 3.17, 5.12, 5.78, 6.47, 7.67, 8.91, 9.72, 10.46, 11.24, 11.96, 13.46, 14.41, 14.80, 15.70, 16.38, 17.11, 17.40, 18.06, 18.38, 18.80, 19.59, 20.63, 20.86, 21.16, 21.94, 22.79, 23.66, 24.580 26.17, and
26.82.
[00303] In another embodiment, Compound 1 erbumine salt Form F is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising five or more peaks selected from 3.17, 5.12, 5.78, 6.47, 7.67, 8.91, 9.72, 10.46, 11.24, 11.96, 13.46, 14.41, 14.80, 15.70, 16.38, 17.11, 17.40, 18.06, 18.38, 18.80, 19.59, 20.63, 20.86, 21.16, 21.94, 22.79, 23.66, 24.580 26.17, and
26.82.
[00304] In another embodiment, Compound 1 erbumine salt Form F is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising ten or more peaks selected from 3.17, 5.12, 5.78, 6.47, 7.67, 8.91, 9.72, 10.46, 11.24, 11.96, 13.46, 14.41, 14.80, 15.70, 16.38, 17.11, 17.40, 18.06, 18.38, 18.80, 19.59, 20.63, 20.86, 21.16, 21.94, 22.79, 23.66, 24.580 26.17, and 26.82.
[00305] In another embodiment, Compound 1 erbumine salt Form F is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising peaks at 11.24, 18.38, and 20.86.
[00306] In another embodiment, Compound 1 erbumine salt Form F is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising peaks at 9.72, 11.24, 18.06, 18.38, and 20.86.
[00307] In another embodiment, Compound 1 erbumine salt Form E is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising peaks at 3.17, 9.72, 10.46, 11.24, 13.46, 18.06, 18.38, 18.80, 20.63, and 20.86.
[00308] In another embodiment, Compound 1 erbumine salt Form F is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising all of the peaks 3.17, 5.12, 5.78, 6.47, 7.67, 8.91, 9.72, 10.46, 11.24, 11.96, 13.46, 14.41, 14.80, 15.70, 16.38, 17.11, 17.40, 18.06, 18.38, 18.80, 19.59, 20.63, 20.86, 21.16, 21.94, 22.79, 23.66, 24.580 26.17, and 26.82.
[00309] In another embodiment, Compound 1 erbumine salt Form F is characterized by the XRPD pattern substantially in accordance with FIG. 35.
[00310] A list of the peaks from the XRPD pattern for Compound 1 erbumine salt Form F is provided in Table 10 below.
[00311] In another embodiment, Compound 1 erbumine salt Form F is characterized by the DSC and TGA thermograms substantially in accordance with FIG. 36.
[00312] In another embodiment, Compound 1 erbumine salt Form F is characterized by the DSC and TGA thermograms showing endothermic peak onsets about 36 °C - 41 °C, about 116 °C - 120 °C, and about 144 °C - 148 °C and showing about 1.0 - 1.2 percent weight loss from 30 °C to 80 °C and about 6.5 - 8.5 percent weight loss from 80 °C to 150 °C.
[00313] In another embodiment, Compound 1 erbumine salt Form F is characterized by the DSC and TGA thermograms showing endothermic peak onsets at about 37.9 °C, 117.7 °C, and 146.0 °C and showing about 1.1 percent weight loss from 30 °C to 80 °C and about 7.5 percent weight loss from 80 °C to 150 °C.
[00314] In another embodiment, Compound 1 erbumine salt Form F is characterized by the ’H NMR spectrum in d6-DMSO substantially in accordance with FIG. 37 indicating no residual solvent.
[00315] In another embodiment, Compound 1 erbumine salt Form F is characterized by at least one of the following:
(1) an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 3.17, 5.12, 5.78, 6.47, 7.67, 8.91, 9.72, 10.46, 11.24, 11.96, 13.46, 14.41, 14.80, 15.70, 16.38, 17.11, 17.40, 18.06, 18.38, 18.80, 19.59, 20.63, 20.86, 21.16, 21.94, 22.79, 23.66, 24.580 26.17, and 26.82;
(2) a TGA thermogram substantially in accordance with FIG. 36; and/or
(3) a DSC thermogram substantially in accordance with FIG. 36.
[00316] In another embodiment, Compound 1 erbumine salt Form F is characterized by at least one of the following:
(1) an XRPD pattern substantially in accordance with FIG. 35;
(2) a TGA thermogram substantially in accordance with FIG. 36; and/or
(3) a DSC thermogram substantially in accordance with FIG. 36.
[00317] In another embodiment, Compound 1 erbumine salt Form F is characterized by at least two of the following:
(1) an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 3.17, 5.12, 5.78, 6.47, 7.67, 8.91, 9.72, 10.46, 11.24, 11.96, 13.46, 14.41, 14.80, 15.70, 16.38, 17.11, 17.40, 18.06, 18.38, 18.80, 19.59, 20.63, 20.86, 21.16, 21.94, 22.79, 23.66, 24.580 26.17, and 26.82;
(2) a TGA thermogram substantially in accordance with FIG. 36; and/or
(3) a DSC thermogram substantially in accordance with FIG. 36.
[00318] In another embodiment, Compound 1 erbumine salt Form F is characterized by all of the following:
(1) an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 3.17, 5.12, 5.78, 6.47, 7.67, 8.91, 9.72, 10.46, 11.24, 11.96, 13.46, 14.41, 14.80, 15.70, 16.38, 17.11, 17.40, 18.06, 18.38, 18.80, 19.59, 20.63, 20.86, 21.16, 21.94, 22.79, 23.66, 24.580 26.17, and 26.82;
(2) a TGA thermogram substantially in accordance with FIG. 36; and
(3) a DSC thermogram substantially in accordance with FIG. 36.
[00319] In another embodiment, Compound 1 erbumine salt Form F is characterized by (1) an XRPD pattern substantially in accordance with FIG. 35 and at least one of the following:
(2) a DSC thermogram substantially in accordance with FIG. 36; and/or
(3) a TGA thermogram substantially in accordance with FIG. 36.
[00320] In another embodiment, Compound 1 erbumine salt Form F is characterized by (1) an XRPD pattern substantially in accordance with FIG. 35 and:
(2) a DSC thermogram substantially in accordance with FIG. 36; and
(3) a TGA thermogram substantially in accordance with FIG. 36.
Compound 1 L-Arginine Salt
[00321] In one embodiment, the salt is Compound 1 L-arginine salt. In a further embodiment, Compound 1 L-arginine salt is amorphous.
[00322] In another embodiment, Compound 1 L-arginine salt is characterized by the XRPD pattern substantially in accordance with FIG. 38.
Compound 1 L-Lysine Salt
[00323] In one embodiment, the salt is Compound 1 L-lysine salt. In another embodiment, Compound 1 L-lysine salt is crystalline. In another embodiment, Compound 1 L-lysine crystalline salt is characterized as Compound 1 L-lysine salt Form A. In a further embodiment, Compound 1 L-lysine crystalline salt is characterized as Compound 1 L-lysine salt Form A (1:0.4) (stoichiometric ratio of Compound 1: lysine).
[00324] The XRPD pattern of Compound 1 L-lysine salt Form A was recorded in reflection geometry.
[00325] In another embodiment, Compound 1 L-lysine salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 9.68,
13.32, 17.71, 17.96, 19.38, 20.47, 22.83, 23.83, 24.45, and 26.03.
[00326] In another embodiment, Compound 1 L-lysine salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising three or more peaks selected from 9.68,
13.32, 17.71, 17.96, 19.38, 20.47, 22.83, 23.83, 24.45, and 26.03.
[00327] In another embodiment, Compound 1 L-lysine salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising five or more peaks selected from 9.68,
13.32, 17.71, 17.96, 19.38, 20.47, 22.83, 23.83, 24.45, and 26.03.
[00328] In another embodiment, Compound 1 L-lysine salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising peaks at 9.68, 19.38, and 26.03.
[00329] In another embodiment, Compound 1 L-lysine salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising peaks at 9.68, 17.71, 19.38, 22.83, and 26.03.
[00330] In another embodiment, Compound 1 L-lysine salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising all of the peaks 9.68, 13.32, 17.71, 17.96, 19.38, 20.47, 22.83, 23.83, 24.45, and 26.03.
[00331] In another embodiment, Compound 1 L-lysine salt is characterized by the XRPD pattern substantially in accordance with FIG. 39.
[00332] A list of the peaks from the XRPD pattern for Compound 1 L-lysine salt Form A is provided in Table 11 below.
General Administration
[00333] Administration of the amorphous or crystalline forms of the present disclosure, in pure form or in an appropriate pharmaceutical composition, can be carried out via any of the accepted modes of administration or agents for serving similar utilities. Thus, administration can be, for example, orally, nasally, parenterally (intravenous, intramuscular, or subcutaneous), topically, transdermally, intravaginally, intravesically, intraci stemally, or rectally, in the form of solid, semi-solid, lyophilized powder, or liquid dosage forms, such as, for example, tablets, suppositories, pills, soft elastic and hard gelatin capsules, powders, solutions, suspensions, aerosols, and the like, preferably in unit dosage forms suitable for simple administration of precise dosages.
[00334] The compositions will include a conventional pharmaceutical excipient and an amorphous or crystalline form of the present disclosure as the/an active agent, and, in addition, may include other medicinal agents, pharmaceutical agents, excipients, adjuvants, and so on. Compositions of the disclosure may be used in combination with anticancer or other agents that are generally administered to a patient being treated for cancer. Adjuvants include preserving, wetting, suspending, sweetening, flavoring, perfuming, emulsifying, and dispensing agents. Prevention of the action of microorganisms can be ensured by various antibacterial and antifungal agents, for example, parabens, chlorobutanol, phenol, sorbic acid, and the like. It may also be desirable to include isotonic agents, for example sugars, sodium chloride, and the like. Prolonged absorption of the injectable pharmaceutical form can be brought about by the use of agents delaying absorption, for example, aluminum monostearate, and gelatin.
[00335] If desired, a pharmaceutical composition of the disclosure may also contain minor amounts of auxiliary substances such as wetting or emulsifying agents, pH buffering agents,
antioxidants, and the like, such as, for example, citric acid, sorbitan, monolaurate, triethanolamine oleate, butylated hydroxytoluene, and so on.
[00336] Compositions suitable for parenteral injection may comprise physiologically acceptable sterile aqueous or nonaqueous solutions, dispersions, suspensions or emulsions, and sterile powders for reconstitution into sterile injectable solutions or dispersions. Examples of suitable aqueous and nonaqueous excipients, diluents, solvents, or vehicles include water, ethanol, polyols (propyleneglycol, polyethyleneglycol, glycerol, and the like), suitable mixtures thereof, vegetable oils (such as olive oil), and injectable organic esters such as ethyl oleate. Proper fluidity can be maintained, for example, by the use of a coating such as lecithin, by the maintenance of the required particle size in the case of dispersions, and by the use of surfactants. [00337] One preferable route of administration is oral, using a convenient daily dosage regimen that can be adjusted according to the degree of severity of the disease-state to be treated. [00338] Solid dosage forms for oral administration include capsules, tablets, pills, powders, and granules. In such solid dosage forms, the active compound is admixed with at least one inert customary excipient such as sodium citrate or dicalcium phosphate or (a) fillers or extenders, as for example, starches, lactose, sucrose, glucose, mannitol, and silicic acid, (b) binders, as for example, cellulose derivatives, starch, alignates, gelatin, polyvinylpyrrolidone, sucrose, and gum acacia, (c) humectants, as for example, glycerol, (d) disintegrating agents, as for example, agar- agar, calcium carbonate, potato or tapioca starch, alginic acid, croscarmellose sodium, complex silicates, and sodium carbonate, (e) solution retarders, as for example paraffin, (f) absorption accelerators, as for example, quaternary ammonium compounds, (g) wetting agents, as for example, cetyl alcohol, and glycerol monostearate, magnesium stearate, and the like (h) adsorbents, as for example, kaolin and bentonite, and (i) lubricants, as for example, talc, calcium stearate, magnesium stearate, solid polyethylene glycols, sodium lauryl sulfate, or mixtures thereof. In the case of capsules, tablets, and pills, the dosage forms may also comprise buffering agents.
[00339] Solid dosage forms as described above can be prepared with coatings and shells, such as enteric coatings and others well known in the art. They may contain pacifying agents and can also be of such composition that they release the active compound or compounds in a certain part of the intestinal tract in a delayed manner. Examples of embedded compositions that can be used
are polymeric substances and waxes. The active compounds can also be in microencapsulated form, if appropriate, with one or more of the above-mentioned excipients.
[00340] Liquid dosage forms for oral administration include pharmaceutically acceptable emulsions, solutions, suspensions, syrups, and elixirs. Such dosage forms are prepared, for example, by dissolving, dispersing, and so on., crystalline forms of Compound 1, and optional pharmaceutical adjuvants in an excipient, such as, for example, water, saline, aqueous dextrose, glycerol, ethanol, and the like; solubilizing agents and emulsifiers, as for example, ethyl alcohol, isopropyl alcohol, ethyl carbonate, ethyl acetate, benzyl alcohol, benzyl benzoate, propyleneglycol, 1,3-butyleneglycol, and dimethylformamide; oils, in particular, cottonseed oil, groundnut oil, com germ oil, olive oil, castor oil, and sesame oil, glycerol, tetrahydrofurfuryl alcohol, polyethyleneglycols and fatty acid esters of sorbitan; or mixtures of these substances, and the like, to thereby form a solution or suspension.
[00341] Suspensions, in addition to the active compounds, may contain suspending agents, as for example, ethoxylated isostearyl alcohols, polyoxyethylene sorbitol, and sorbitan esters, microcrystalline cellulose, aluminum metahydroxide, bentonite, agar-agar, and tragacanth, or mixtures of these substances, and the like.
[00342] Compositions for rectal administrations are, for example, suppositories that can be prepared by mixing the crystalline forms of Compound 1 with for example suitable non-irritating excipients such as cocoa butter, polyethyleneglycol, or a suppository wax, which are solid at ordinary temperatures but liquid at body temperature and therefore melt while in a suitable body cavity and release the active component therein.
[00343] Dosage forms for topical administration of a compound of this disclosure include ointments, powders, sprays, and inhalants. The active component is admixed under sterile conditions with a physiologically acceptable excipient and any preservatives, buffers, or propellants as may be required. Ophthalmic formulations, eye ointments, powders, and solutions are also contemplated as being within the scope of this disclosure.
[00344] Generally, depending on the intended mode of administration, the pharmaceutically acceptable compositions will contain about 1% to about 99% by weight of a crystalline form of Compound 1, and 99% to 1% by weight of a suitable pharmaceutical excipient. In one example, the composition will be between about 5% and about 75% by weight of a crystalline form of Compound 1, with the rest being suitable pharmaceutical excipients.
[00345] Actual methods of preparing such dosage forms are known, or will be apparent, to those skilled in this art; for example, see Remington's Pharmaceutical Sciences, 21st Ed., (Lippincott, Williams and Wilkins Philadelphia, PA, 2006). The composition to be administered will, in any event, contain a therapeutically effective amount of a crystalline form of Compound 1 for treatment of a disease-state in accordance with the teachings of this disclosure.
[00346] The crystalline forms of Compound 1, are administered in a therapeutically effective amount which will vary depending upon a variety of factors including the activity of Compound 1, the metabolic stability and length of action of Compound 1, the age, body weight, general health, sex, diet, mode, and time of administration, rate of excretion, drug combination, the severity of the particular disease-states, and the host undergoing therapy. The crystalline forms of Compound 1 can be administered to a patient at dosage levels in the range of about 0.1 to about 1,000 mg per day. For a normal human adult having a body weight of about 70 kilograms, a dosage in the range of about 0.01 to about 100 mg per kilogram of body weight per day is an example. The specific dosage used, however, can vary. For example, the dosage can depend on a number of factors including the requirements of the patient, the severity of the condition being treated, and the pharmacological activity of the compound being used. The determination of optimum dosages for a particular patient is well known to one of ordinary skill in the art.
Pharmaceutical Compositions, Treatment Methods, and Uses
[00347] The compounds provided herein can be formulated into pharmaceutical compositions using methods available in the art and those disclosed herein. Any of the solid forms provided herein can be provided in the appropriate pharmaceutical composition and be administered by a suitable route of administration.
[00348] The methods provided herein encompass administering pharmaceutical compositions comprising at least one solid form provided herein and one or more compatible and pharmaceutically acceptable carriers. The term “carrier” includes a diluent, adjuvant (e.g., Freund’s adjuvant (complete and incomplete)), excipient, or vehicle with which the therapeutic is administered. Such pharmaceutical carriers can be sterile liquids, such as water and oils including petroleum, animal, vegetable, or oils of synthetic origin, such as peanut oil, soybean oil, mineral oil, sesame oil, and the like. Water can be used as a carrier when the pharmaceutical composition is administered intravenously. Saline solutions, aqueous dextrose, and glycerol
solutions can also be employed as liquid carriers, particularly for injectable solutions. Examples of suitable pharmaceutical carriers are described in Martin, E.W., Remington 's Pharmaceutical Sciences.
[00349] In clinical practice the pharmaceutical compositions, or compounds provided herein may be administered by any route known in the art. Exemplary routes of administration include, but are not limited to, inhalation, intraarterial, intradermal, intramuscular, intraperitoneal, intravenous, nasal, parenteral, pulmonary, and subcutaneous routes. In some embodiments, a pharmaceutical composition or compound provided herein is administered parenterally.
[00350] The compositions for parenteral administration can be emulsions or sterile solutions. Parenteral compositions may include, for example, propylene glycol, polyethylene glycol, vegetable oils, and injectable organic esters (e.g., ethyl oleate). These compositions can also contain wetting, isotonizing, emulsifying, dispersing, and stabilizing agents. Sterilization can be carried out in several ways, for example, using a bacteriological filter, via radiation, or via heating. Parenteral compositions can also be prepared in the form of sterile solid compositions which can be dissolved at the time of use in sterile water, or any other injectable sterile medium. [00351] In some embodiments, a composition provided herein is a pharmaceutical composition, or a single unit dosage form. Pharmaceutical compositions, and single unit dosage forms provided herein comprise a prophylactically, or therapeutically effective amount of one, or more prophylactic, or therapeutic compounds.
[00352] The pharmaceutical composition may comprise one or more pharmaceutical excipients. Any suitable pharmaceutical excipient may be used, wherein a person of ordinary skill in the art is capable of selecting suitable pharmaceutical excipients. Non-limiting examples of suitable excipients include starch, glucose, lactose, sucrose, gelatin, malt, rice, flour, chalk, silica gel, sodium stearate, glycerol monostearate, talc, sodium chloride, dried skim milk, glycerol, propylene glycol, water, ethanol, and the like. Whether a particular excipient is suitable for incorporation into a pharmaceutical composition, or dosage form depends on a variety of factors well known in the art including, but not limited to, the way in which the dosage form will be administered to a subject and the specific compound in the dosage form. The composition, or single unit dosage form, if desired, can also contain minor amounts of wetting, or emulsifying agents, or pH buffering agents. Accordingly, the pharmaceutical excipients provided below are intended to be illustrative, and not limiting. Additional pharmaceutical excipients include, for
example, those described in the Handbook of Pharmaceutical Excipients, Rowe et al. (Eds.) 6th Ed. (2009), incorporated by reference herein in its entirety.
[00353] In some embodiments, the pharmaceutical composition comprises an anti-foaming agent. Any suitable anti-foaming agent may be used. In some aspects, the anti-foaming agent is selected from an alcohol, an ether, an oil, a wax, a silicone, a surfactant, and combinations thereof. In some aspects, the anti-foaming agent is selected from a mineral oil, a vegetable oil, ethylene bis stearamide, a paraffin wax, an ester wax, a fatty alcohol wax, a long-chain fatty alcohol, a fatty acid soap, a fatty acid ester, a silicon glycol, a fluorosilicone, a polyethylene glycol-polypropylene glycol copolymer, polydimethylsiloxane-silicon dioxide, ether, octyl alcohol, capryl alcohol, sorbitan trioleate, ethyl alcohol, 2-ethyl-hexanol, dimethicone, oleyl alcohol, simethicone, and combinations thereof.
[00354] In some embodiments, the pharmaceutical composition comprises a co-solvent. Illustrative examples of co-solvents include ethanol, poly(ethylene) glycol, butylene glycol, di methyl acetamide, glycerin, and propylene glycol.
[00355] In some embodiments, the pharmaceutical composition comprises a buffer. Illustrative examples of buffers include acetate, borate, carbonate, lactate, malate, phosphate, citrate, hydroxide, diethanolamine, monoethanolamine, glycine, methionine, guar gum, and monosodium glutamate.
[00356] In some embodiments, the pharmaceutical composition comprises a carrier, or filler. Illustrative examples of carriers, or fillers include lactose, maltodextrin, mannitol, sorbitol, chitosan, stearic acid, xanthan gum, and guar gum.
[00357] In some embodiments, the pharmaceutical composition comprises a surfactant. Illustrative examples of surfactants, include /-alpha tocopherol, benzalkonium chloride, benzethonium chloride, cetrimide, cetylpyridinium chloride, docusate sodium, glyceryl behenate, glyceryl monooleate, lauric acid, macrogol 15 hydroxystearate, myristyl alcohol, phospholipids, polyoxyethylene alkyl ethers, polyoxyethylene sorbitan fatty acid esters, polyoxyethylene stearates, poly oxylglyceri des, sodium lauryl sulfate, sorbitan esters, and vitamin E polyethylene(glycol) succinate.
[00358] In some embodiments, the pharmaceutical composition comprises an anti-caking agent. Illustrative examples of anti -caking agents include calcium phosphate (tribasic), hydroxymethyl cellulose, hydroxypropyl cellulose, and magnesium oxide.
[00359] Other excipients that may be used with the pharmaceutical compositions include, for example, albumin, antioxidants, antibacterial agents, antifungal agents, bioabsorbable polymers, chelating agents, controlled release agents, diluents, dispersing agents, dissolution enhancers, emulsifying agents, gelling agents, ointment bases, penetration enhancers, preservatives, solubilizing agents, solvents, stabilizing agents, and sugars. Specific examples of each of these agents are described, for example, in the Handbook of Pharmaceutical Excipients, Rowe et al. (Eds.) 6th Ed. (2009), The Pharmaceutical Press, incorporated by reference herein in its entirety. [00360] In some embodiments, the pharmaceutical composition comprises a solvent. In some aspects, the solvent is saline solution, such as a sterile isotonic saline solution, or dextrose solution. In some aspects, the solvent is water for injection.
[00361] In some embodiments, the pharmaceutical compositions are in a particulate form, such as a microparticle or a nanoparticle. Microparticles, and nanoparticles may be formed from any suitable material, such as a polymer, or a lipid. In some aspects, the microparticles, or nanoparticles are micelles, liposomes, or polymersomes.
[00362] Further provided herein are anhydrous pharmaceutical compositions, and dosage forms comprising a compound, since, in some embodiments, water can facilitate the degradation of some compounds.
[00363] Anhydrous pharmaceutical compositions, and dosage forms provided herein can be prepared using anhydrous, or low moisture containing ingredients, and low moisture, or low humidity conditions. Pharmaceutical compositions, and dosage forms that comprise lactose, and at least one active ingredient that comprises a primary, or secondary amine can be anhydrous if substantial contact with moisture, and/or humidity during manufacturing, packaging, and/or storage is expected.
[00364] An anhydrous pharmaceutical composition can be prepared and stored such that its anhydrous nature is maintained. Accordingly, anhydrous compositions can be packaged using materials known to prevent exposure to water such that they can be included in suitable formulary kits. Examples of suitable packaging include, but are not limited to, hermetically sealed foils, plastics, unit dose containers (e.g., vials), blister packs, and strip packs.
[00365] Lactose-free compositions provided herein can comprise excipients that are well known in the art and are listed, for example, in the U.S. Pharmocopeia (USP) SP (XXI)/NF (XVI). In general, lactose-free compositions comprise an active ingredient, a binder/fdler, and a
lubricant in pharmaceutically compatible, and pharmaceutically acceptable amounts. Exemplary lactose-free dosage forms comprise an active ingredient, microcrystalline cellulose, pre gelatinized starch, and magnesium stearate.
[00366] Also provided are pharmaceutical compositions, and dosage forms that comprise one, or more excipients that reduce the rate by which a compound will decompose. Such excipients, which are referred to herein as “stabilizers,” include, but are not limited to, antioxidants such as ascorbic acid, pH buffers, or salt buffers.
Parenteral Dosage Forms
[00367] In certain embodiments, provided are parenteral dosage forms. Parenteral dosage forms can be administered to subjects by various routes including, but not limited to, subcutaneous, intravenous (including bolus injection), intramuscular, and intraarterial. Because their administration typically bypasses subjects’ natural defenses against contaminants, parenteral dosage forms are typically sterile, or capable of being sterilized prior to administration to a subject. Examples of parenteral dosage forms include, but are not limited to, solutions ready for injection, dry products ready to be dissolved or suspended in a pharmaceutically acceptable vehicle for injection, suspensions ready for injection, and emulsions.
[00368] Suitable vehicles that can be used to provide parenteral dosage forms are well known to those skilled in the art. Examples include, but are not limited to Water for Injection USP; aqueous vehicles such as, but not limited to, Sodium Chloride Injection, Ringer’s Injection, Dextrose Injection, Dextrose, and Sodium Chloride Injection, and Lactated Ringer’s Injection; water miscible vehicles such as, but not limited to, ethyl alcohol, polyethylene glycol, and polypropylene glycol; and non-aqueous vehicles such as, but not limited to, corn oil, cottonseed oil, peanut oil, sesame oil, ethyl oleate, isopropyl myristate, and benzyl benzoate.
[00369] Excipients that increase the solubility of one, or more of the antibodies disclosed herein can also be incorporated into the parenteral dosage forms.
[00370] In human therapeutics, the doctor will determine the posology which he considers most appropriate according to a preventive, or curative treatment, and according to the age, weight, condition, and other factors specific to the subject to be treated.
[00371] In certain embodiments, a composition provided herein is a pharmaceutical composition, or a single unit dosage form. Pharmaceutical compositions, and single unit dosage
forms provided herein comprise a prophylactically, or therapeutically effective amount of one, or more prophylactic, or therapeutic antibodies, or antigen binding fragments thereof.
[00372] The amount of the compound, or composition which will be effective in the prevention, or treatment of a disorder, or one, or more symptoms thereof will vary with the nature, and severity of the disease, or condition, and the route by which the compound is administered. The frequency and dosage will also vary according to factors specific for each subject depending on the specific therapy (e g., therapeutic or prophylactic agents) administered, the severity of the disorder, disease, or condition, the route of administration, as well as age, body, weight, response, and the past medical history of the subject. Effective doses may be extrapolated from dose-response curves derived from in vitro, or animal model test systems. [00373] In certain embodiments, exemplary doses of a composition include milligram, or microgram amounts of the compound per kilogram of subject, or sample weight (e.g., about 10 micrograms per kilogram to about 50 milligrams per kilogram, about 100 micrograms per kilogram to about 25 milligrams per kilogram, or about 100 microgram per kilogram to about 10 milligrams per kilogram). In certain embodiments, the dosage of the compound provided herein, based on weight of the compound, administered to prevent, treat, manage, or ameliorate a disorder, or one, or more symptoms thereof in a subject is 0.1 mg/kg, 1 mg/kg, 2 mg/kg, 3 mg/kg, 4 mg/kg, 5 mg/kg, 6 mg/kg, 10 mg/kg, or 15 mg/kg or more of a subject’s body weight.
[00374] In another embodiment, the dosage of the composition, or a composition provided herein administered to prevent, treat, manage, or ameliorate a disorder, or one, or more symptoms thereof in a subject is 0.1 mg to 200 mg, 0.1 mg to 100 mg, 0.1 mg to 50 mg, 0.1 mg to 25 mg, 0.1 mg to 20 mg, 0.1 mg to 15 mg, 0.1 mg to 10 mg, 0.1 mg to 7.5 mg, 0.1 mg to 5 mg, 0.1 to 2.5 mg, 0.25 mg to 20 mg, 0.25 to 15 mg, 0.25 to 12 mg, 0.25 to 10 mg, 0.25 mg to 7.5 mg, 0.25 mg to 5 mg, 0.25 mg to 2.5 mg, 0.5 mg to 20 mg, 0.5 to 15 mg, 0.5 to 12 mg, 0.5 to 10 mg, 0.5 mg to 7.5 mg, 0.5 mg to 5 mg, 0.5 mg to 2.5 mg, 1 mg to 20 mg, 1 mg to 15 mg, 1 mg to 12 mg, 1 mg to 10 mg, 1 mg to 7.5 mg, 1 mg to 5 mg, or 1 mg to 2.5 mg.
[00375] The dose can be administered according to a suitable schedule, for example, once, two times, three times, or four times weekly. It may be necessary to use dosages of the compound outside the ranges disclosed herein in some cases, as will be apparent to those of ordinary skill in the art. Furthermore, it is noted that the clinician, or treating physician will
know how, and when to interrupt, adjust, or terminate therapy in conjunction with subject response.
[00376] Different therapeutically effective amounts may be applicable for different diseases, and conditions, as will be readily known by those of ordinary skill in the art. Similarly, amounts sufficient to prevent, manage, treat, or ameliorate such disorders, but insufficient to cause, or sufficient to reduce, adverse effects associated with the antibodies, or antigen binding fragments thereof provided herein are also encompassed by the described dosage amounts, and dose frequency schedules herein. Further, when a subject is administered multiple dosages of a composition provided herein, not all of the dosages need be the same. For example, the dosage administered to the subject may be increased to improve the prophylactic, or therapeutic effect of the composition, or it may be decreased to reduce one, or more side effects that a particular subject is experiencing.
[00377] In certain embodiments, treatment, or prevention can be initiated with one, or more loading doses of a compound, or composition provided herein followed by one, or more maintenance doses.
[00378] In certain embodiments, a dose of a compound, or composition provided herein can be administered to achieve a steady-state concentration of the compound in blood, or serum of the subject. The steady-state concentration can be determined by measurement according to techniques available to those of skill or can be based on the physical characteristics of the subject such as height, weight, and age.
[00379] In certain embodiments, administration of the same composition may be repeated, and the administrations may be separated by at least one day, two days, three days, five days, ten days, fifteen days, thirty days, forty-five days, two months, seventy-five days, three months, or six months. In other embodiments, administration of the same prophylactic, or therapeutic agent may be repeated, and the administration may be separated by at least one day, two days, three days, five days, ten days, fifteen days, thirty days, forty-five days, two months, seventy-five days, three months, or six months.
Therapeutic Applications
[00380] For therapeutic applications, the compounds are administered to a mammal, in certain embodiments, a human, in a pharmaceutically acceptable dosage suitable for administration form
such as those known in the art, and those discussed herein, intravenously as a bolus or by continuous infusion over a period of time, by intramuscular, intraperitoneal, intra-cerebrospinal, subcutaneous, intra-articular, intrasynovial, intrathecal, or intratumoral routes. The compounds also are suitably administered by peritumoral, intralesional, or perilesional routes, to exert local as well as systemic therapeutic effects. In certain embodiments, the compounds are administered to a mammal, in certain embodiments, a human, in a pharmaceutically acceptable dosage suitable for oral administration form such as those known in the art, and those discussed herein. For example, the compounds of this disclosure may be administered orally to a human as a liquid, or solid form. Solid dosage forms include capsules, tablets, pills, powders, and granules.
[00381] In such solid dosage forms, the chemical entity is mixed with one or more pharmaceutically acceptable excipients, such as sodium citrate or dicalcium phosphate and/or: a) fillers or extenders such as starches, lactose, sucrose, glucose, mannitol, and silicic acid, b) binders such as, for example, carboxymethylcellulose, alginates, gelatin, polyvinylpyrrolidinone, sucrose, and acacia, c) humectants such as glycerol, d) disintegrating agents such as agar-agar, calcium carbonate, potato or tapioca starch, alginic acid, certain silicates, and sodium carbonate, e) solution retarding agents such as paraffin, f) absorption accelerators such as quaternary ammonium compounds, g) wetting agents such as, for example, cetyl alcohol and glycerol monostearate, h) absorbents such as kaolin and bentonite clay, and i) lubricants such as talc, calcium stearate, magnesium stearate, solid polyethylene glycols, sodium lauryl sulfate, and mixtures thereof. In the case of capsules, tablets and pills, the dosage form may also comprise buffering agents. Solid compositions of a similar type may also be employed as fillers in soft and hard-filled gelatin capsules using such excipients as lactose or milk sugar as well as high molecular weight polyethylene glycols and the like.
[00382] The compounds provided herein may be useful for the treatment of any disease or condition described herein (e.g., a metabolic disease or condition). In certain embodiments, the disease, or condition is any disease, or condition that benefits from modulation of GLP-1 receptor activity. In certain embodiments, the disease, or condition is any disease, or condition that benefits from agonizing GLP-1 receptor activity. In certain embodiments, the methods reduce blood glucose levels. In certain embodiments, the methods promote insulin synthesis, stimulate insulin secretion, increase the mass of P-cells, modulate gastric acid secretion,
modulate gastric emptying, and/or decrease glucagon production. In certain embodiments, the disease, or condition is type 2 diabetes.
[00383] In certain embodiments, the disease, or condition is obesity, or one, or more diseases, or conditions associated with obesity. Non-limiting examples of obesity, and obesity related conditions include symptomatic obesity, simple obesity, childhood obesity, morbid obesity, and abdominal obesity (central obesity characterized by abdominal adiposity). Non-limiting examples of symptomatic obesity include endocrine obesity (e.g., Cushing syndrome, hypothyroidism, insulinoma, obese type II diabetes, pseudohypoparathyroidism, hypogonadism), hypothalamic obesity, hereditary obesity (e.g., Prader-Willi syndrome, Laurence-Moon-Biedl syndrome), and drug-induced obesity (e.g., steroid, phenothiazine, insulin, sulfonylurea agent, or P-blocker-induced obesity). In certain embodiments, the disease or condition is obesity with diabetes. In certain embodiments, the disease or condition is obesity without diabetes.
[00384] Examples of such diseases, and conditions associated with obesity include, without limitation, glucose tolerance disorders, diabetes (e.g., type 2 diabetes, obese diabetes), lipid metabolism abnormality, hyperlipidemia, hypertension, cardiac failure, hyperuricemia, gout, fatty liver (including non-alcoholic steatohepatitis (NASH) or metabolic dysfunction-associated steatohepatitis (MASH)), coronary heart disease (e.g., myocardial infarction, angina pectoris), cerebral infarction (e.g., brain thrombosis, transient cerebral ischemic attack), bone, or articular disease (e.g., knee osteoarthritis, hip osteoarthritis, spondylitis deformans, lumbago), sleep apnea syndrome, obesity hypoventilation syndrome (Pickwickian syndrome), menstrual disorder (e.g., abnormal menstrual cycle, abnormality of menstrual flow and cycle, amenorrhea, abnormal catamenial symptom), visceral obesity syndrome, and metabolic syndrome. In certain embodiments, the compounds described herein can be used to treat subjects exhibiting symptoms of both obesity, and insulin deficiency.
[00385] In some embodiments, the disease, or condition is diabetes. Non-limiting examples of diabetes include, type 1 diabetes, type 2 diabetes (e g., diet-treated type 2-diabetes, sulfonylurea- treated type 2-diabetes, a far-advanced stage type 2-diabetes, long-term insulin-treated type 2- diabetes), diabetes mellitus (e.g., non-insulin-dependent diabetes mellitus, insulin-dependent diabetes mellitus), gestational diabetes, obese diabetes, autoimmune diabetes, and borderline type diabetes.
[00386] In some embodiments, the disease or condition is associated with diabetes (e.g., a complication of diabetes). Non-limiting examples of disorders associated with diabetes include, obesity, obesity-related disorders, metabolic syndrome, neuropathy, nephropathy (e.g., diabetic nephropathy), retinopathy, diabetic cardiomyopathy, cataract, macroangiopathy, osteopenia, hyperosmolar diabetic coma, infectious disease (e.g., respiratory infection, urinary tract infection, gastrointestinal infection, dermal soft tissue infections, inferior limb infection), diabetic gangrene, xerostomia, hypacusis, cerebrovascular disorder, diabetic cachexia, delayed wound healing, diabetic dyslipidemia peripheral blood circulation disorder, cardiovascular risk factors, (e.g., coronary artery disease, peripheral artery disease, cerebrovascular disease, hypertension, and risk factors related to unmanaged cholesterol, and/or lipid levels, and/or inflammation), NASH, bone fracture, and cognitive dysfunction
[00387] Other non-limiting examples of diseases, or conditions related to diabetes include, pre-diabetes, hyperlipidemia (e.g., hypertriglyceridemia, hypercholesterolemia, high LDL- cholesterolemia, low HDL-cholesterolemia, postprandial hyperlipemia), metabolic syndrome (e.g., metabolic disorder where activation of GLP-1R is beneficial, metabolic syndrome X), hypertension, impaired glucose tolerance (IGT), insulin resistance, and sarcopenia.
[00388] In some embodiments, the disease, or condition is diabetes, and obesity (diabesity). In certain embodiments, the compounds described herein are useful in improving the therapeutic effectiveness of metformin.
[00389] In some embodiments, the disease or condition is a disorder of a metabolically important tissue.
[00390] In some embodiments, the disease, or condition is a fatty liver disease. Fatty liver diseases include, but are not limited to, non-alcoholic fatty acid liver disease (NAFLD) or metabolic dysfunction-associated fatty liver disease (MAFLD) , steatohepatitis, non-alcoholic steatohepatitis (NASH), fatty liver disease resulting from hepatitis, fatty liver disease resulting from obesity, fatty liver disease resulting from diabetes, fatty liver disease resulting from insulin resistance, fatty liver disease resulting from hypertriglyceridemia, Abetalipoproteinemia, glycogen storage diseases, Weber-Christian disease, Wolman’s disease, acute fatty liver of pregnancy, and lipodystrophy.
[00391] Non-alcoholic fatty liver disease (NAFLD) represents a spectrum of disease occurring in the absence of alcohol abuse, and is typically characterized by the presence of steatosis (fat in
the liver). NAFLD is believed to be linked to a variety of conditions, e.g., metabolic syndrome (including obesity, diabetes, and hypertriglyceridemia), and insulin resistance. It can cause liver disease in adults and children, and may ultimately lead to cirrhosis (Skelly et al., J Hepatol 2001; 35: 195-9; Chitturi et al., Hepatology 2002; 35(2):373-9). The severity of NAFLD ranges from the relatively benign isolated predominantly macrovesicular steatosis (i.e., nonalcoholic fatty liver or NAFL) to non-alcoholic steatohepatitis (NASH) (Angulo et al., J Gastroenterol Hepatol 2002; 17 SuppkS 186-90). In certain embodiments, the subject is a pediatric subject (e.g., 6-16 years old; or 6-12 years old; or 6-10 years old). In certain embodiments, the subject is an adult subject.
[00392] Other non-limiting examples of diseases, or conditions in metabolically important tissues include, joint disorders (e.g., osteoarthritis, secondary osteoarthritis), steatosis (e.g. in the liver); gall stones; gallbladder disorders; gastroesophageal reflux; sleep apnea; hepatitis; fatty liver; bone disorder characterized by altered bone metabolism, such as osteoporosis, including post-menopausal osteoporosis, poor bone strength, osteopenia, Paget's disease, osteolytic metastasis in cancer patients, osteodystrophy in liver disease, and the altered bone metabolism caused by renal failure, or hemodialysis, bone fracture, bone surgery, aging, pregnancy, protection against bone fractures, and malnutrition polycystic ovary syndrome; renal disease (e.g., chronic renal failure, glomerulonephritis, glomerulosclerosis, nephrotic syndrome, hypertensive nephrosclerosis, end-stage renal disease); muscular dystrophy, angina pectoris, acute, or chronic diarrhea, testicular dysfunction, respiratory dysfunction, frailty, sexual dysfunction (e.g., erectile dysfunction), and geriatric syndrome. In certain embodiments, the chemical entities described herein can be used for treating surgical trauma by improving recovery after surgery, and/or by preventing the catabolic reaction caused by surgical trauma. [00393] In some embodiments, the disease, or condition is a cardiovascular disease. Nonlimiting examples of cardiovascular disease include, congestive heart failure, atherosclerosis, arteriosclerosis, coronary heart disease, or peripheral artery disease, stroke, coronary artery disease, congestive heart failure, coronary heart disease, hypertension, cardiac failure, cerebrovascular disorder (e.g., cerebral infarction), vascular dysfunction, myocardial infarction, elevated blood pressure (e.g., 130/85 mm Hg or higher), and prothrombotic state (exemplified by high fibrinogen, or plasminogen activator inhibitor in the blood).
[00394] In some embodiments, the disease, or condition is a neurological disorder (e.g., neurodegenerative disorder), or a psychiatric disorder. Non-limiting examples of neurological disorders include, brain insulin resistance, mild cognitive impairment (MCI), Alzheimer's disease (AD), Parkinson's disease (PD), anxiety, dementia (e.g., senile dementia), traumatic brain injury, Huntington's chores, tardive dyskinesia, hyperkinesia, mania, Morbus Parkinson, steel-Richard syndrome, Down's syndrome, myasthenia gravis, nerve trauma, brain trauma, vascular amyloidosis, cerebral hemorrhage I with amyloidosis, brain inflammation, Friedrich's ataxia, acute confusion disorder, amyotrophic lateral sclerosis (ALS), glaucoma, and apoptosis-mediated degenerative diseases of the central nervous system (e.g., Creutzfeld-Jakob Disease, bovine spongiform encephalopathy (mad cow disease), chronic wasting syndrome).
[00395] Non-limiting examples of psychiatric disorders include drug dependence/addiction (narcotics, amphetamines, and attention deficit/hyperactivity disorder (ADHD). The chemical entities described herein can be useful in improving behavioral response to addictive drugs, decreasing drug dependence, prevention drug abuse relapse, and relieving anxiety caused by the absence of a given addictive substance.
[00396] In certain embodiments, the chemical entities described herein are useful in improving learning, and memory by enhancing neuronal plasticity, and facilitation of cellular differentiation, and also in preserving dopamine neurons, and motor function in Morbus Parkinson.
[00397] In some embodiments, the disease, or condition is impaired fasting glucose (IFG), impaired fasting glycemia (IFG), hyperglycemia, insulin resistance (impaired glucose homeostasis), hyperinsulinemia, elevated blood levels of fatty acids, or glycerol, a hypoglycemic condition, insulin resistant syndrome, paresthesia caused by hyperinsulinemia, hyperlipidemia, hypercholesteremia, impaired wound healing, leptin resistance, glucose intolerance, increased fasting glucose, dyslipidemia (e.g., hyperlipidemia, atherogenic dyslipidemia characterized by high triglycerides and low HDL cholesterol), glucagonoma, hyperuric acidemia, hypoglycemia (e.g., nighttime hypoglycemia), and concomitant comatose endpoint associated with insulin.
[00398] In certain embodiments, the compounds described herein can reduce, or slow down the progression of borderline type, impaired fasting glucose, or impaired fasting glycemia into diabetes.
[00399] In some embodiments, the disease, or condition is an autoimmune disorder. Nonlimiting examples of autoimmune disorders include, multiple sclerosis, experimental autoimmune encephalomyelitis, autoimmune disorder is associated with immune rejection, graft versus host disease, uveitis, optic neuropathies, optic neuritis, transverse myelitis, inflammatory bowel disease, rheumatoid arthritis, ankylosing spondylitis, systemic lupus erythematosus, myasthenia gravis, and Graves’ disease.
[00400] In some embodiments, the disease, or condition is a stomach, or intestine related disorder. Non-limiting examples of these disorders include, ulcers of any etiology (e.g. peptic ulcers, Zollinger-Ellison syndrome, drug-induced ulcers, ulcers related to infections, or other pathogens), digestion disorders, malabsorption, short bowel syndrome, cul-de-sac syndrome, inflammatory bowel diseases (Crohn’s disease, and ulcerative colitis), celiac sprue, hypogammaglobulinemic sprue, chemotherapy, and/or radiation therapy-induced mucositis, and diarrhea, gastrointestinal inflammation, short bowel syndrome, ulcerative colitis, gastric mucosal injury (e.g., gastric mucosal injury caused by aspirin), small intestinal mucosal injury, and cachexia (e.g., cancerous cachexia, tuberculous cachexia, cachexia associated with blood disease, cachexia associated with endocrine disease, cachexia associated with infectious disease, cachexia caused by acquired immunodeficiency syndrome).
[00401] In some embodiments, the compounds described herein can be used to reduce body weight (e.g., excess body weight), prevent body weight gain, induce weight loss, decrease body fat, or reduce food intake in a subject (e.g., a subject in need thereof). In certain embodiments, the weight increase in a subject may be attributed to excessive ingestion of food, or unbalanced diets, or may be weight increase derived from a concomitant drug (e.g., insulin sensitizers having a PPARy agonist-like action, such as troglitazone, rosiglitazone, englitazone, ciglitazone, pioglitazone, and the like). Alternatively, the weight increase may be weight increase before reaching obesity, or may be weight increase in an obese subject. The weight increase may also be medication-induced weight gain, or weight gain subsequent to cessation of smoking. In some embodiments, the compounds described herein can be used to maintain body weight following weight loss, In certain embodiments, the weight loss is caused by injectable medications. In certain embodiments, the injectable medication is selected from the group consisting of CT-388, CT-868, Mounjaro®, Zepbound®, Wegovy®, Ozempic®, Zepbound®, and Saxenda®, as well as others.
[00402] In some embodiments, the condition, disease, or disorder is an eating disorder, such as hyperphagia, binge eating, bulimia, or compulsive eating.
[00403] In some embodiments, the disease, or condition is an inflammatory disorder. Nonlimiting examples of inflammatory disorders include, chronic rheumatoid arthritis, spondylitis deformans, arthritis deformans, lumbago, gout, post-operational or post-traumatic inflammation, bloating, neuralgia, laryngopharyngitis, cystitis, pneumonia, pancreatitis, enteritis, inflammatory bowel disease (including inflammatory large bowel disease), inflammation in metabolically important tissues including liver, fat, pancreas, kidney, and gut, and a proinfl ammatory state (e.g., elevated levels of proinflammatory cytokines or, markers of inflammation-like C-reactive protein in the blood).
[00404] In some embodiments, the disease, or condition is cancer. Suitable examples of cancer include, breast cancer (e.g., invasive ductal breast cancer, noninvasive ductal breast cancer, inflammatory breast cancer), prostate cancer (e.g., hormone-dependent prostate cancer, hormone-independent prostate cancer), pancreatic cancer (e.g., ductal pancreatic cancer), gastric cancer (e.g., papillary adenocarcinoma, mucous adenocarcinoma, adenosquamous carcinoma), lung cancer (e.g., non-small cell lung cancer, small-cell lung cancer, malignant mesothelioma), colon cancer (e.g., gastrointestinal stromal tumor), rectal cancer (e.g., gastrointestinal stromal tumor), colorectal cancer (e.g., familial colorectal cancer, hereditary non-polyposis colorectal cancer, gastrointestinal stromal tumor), small intestinal cancer (e.g., non-Hodgkin's lymphoma, gastrointestinal stromal tumor), esophageal cancer, duodenal cancer, tongue cancer, pharyngeal cancer (e.g., nasopharyngeal cancer, oropharynx cancer, hypopharyngeal cancer), salivary gland cancer, brain tumor (e.g., pineal astrocytoma, pilocytic astrocytoma, diffuse astrocytoma, anaplastic astrocytoma), neurilemmoma, liver cancer (e.g., primary liver cancer, extrahepatic bile duct cancer), renal cancer (e.g., renal cell cancer, transitional cell cancer of the renal pelvis and ureter), bile duct cancer, endometrial cancer, uterine cervical cancer, ovarian cancer (e.g., epithelial ovarian cancer, extragonadal germ cell tumor, ovarian germ cell tumor, ovarian tumor of low malignant potential), bladder cancer, urethral cancer, skin cancer (e.g., intraocular (ocular) melanoma, Merkel cell carcinoma), hemangioma, malignant lymphoma, malignant melanoma, thyroid cancer (e.g., medullary thyroid cancer), parathyroid cancer, nasal cavity cancer, sinus cancer, bone tumor (e.g., osteosarcoma, Ewing tumor, uterine sarcoma, soft tissue sarcoma), angiofibroma, sarcoma of the retina, penis cancer, testicular tumor, pediatric solid
tumor (e.g., Wilms’ tumor, childhood kidney tumor), Kaposi's sarcoma, Kaposi's sarcoma caused by AIDS, tumor of maxillary sinus, fibrous histiocytoma, leiomyosarcoma, rhabdomyosarcoma, and leukemia (e.g., acute myeloid leukemia, acute lymphoblastic leukemia).
[00405] In certain embodiments, provided herein are methods for the treatment that include the administration of an effective amount of compounds provided herein. In certain embodiments, the methods encompass the step of administering to the subject in need thereof an amount of a compound described herein effective for the treatment of disease, or condition in combination with a second agent effective for the treatment, or prevention of the disease, or condition. In certain embodiments, the compound is in the form of a pharmaceutical composition, or dosage form, as described elsewhere herein.
[00406] In certain embodiments, the subject is a treatment naive subject. In further embodiments, the subject has previously received therapy. For instance, in certain embodiments, the subject has not responded to a single agent treatment regimen. In some embodiments, the subject has received a prior incretin treatment. In some embodiments, the incretin treatment is selected from exenatide (Byetta®, Bydureon®), liraglutide (Victoza®), sitagliptin (Januvia®, Janumet®, Janumet® XR, Juvisync®), saxagliptin (Onglyza®, Kombiglyze® XR), alogliptin (Nesina®, Kazano®, Oseni®), linagliptin (Tradjenta®, Jentadueto®), semaglutide, Wegovy®, Mounjaro®, and Rybelsus®, as well as others.
[00407] In certain embodiments, the subject is a subject that discontinued some other therapy because of one or more adverse events associated with the other therapy. In certain embodiments, the subject has received some other therapy and discontinued that therapy prior to administration of a method provided herein. In further embodiments, the subject has received therapy and continues to receive that therapy along with administration of a compound provided herein. The compounds described herein can be co-administered with other therapy for treatment of the disease or condition according to the judgment of one of skill in the art. In certain embodiments, the methods or compositions provided herein can be co-administered with a reduced dose of the other therapy for the treatment of the disease or condition.
Kits
[00408] In some embodiments, a compound provided herein is provided in the form of a kit
(i.e., a packaged combination of reagents in predetermined amounts with instructions for
performing a procedure). In some embodiments, the procedure is a diagnostic assay. In certain embodiments, the procedure is a therapeutic procedure.
[00409] In some embodiments, the kit further comprises a solvent for the reconstitution of the compound. In some embodiments, the compound is provided in the form of a pharmaceutical composition.
[00410] In some embodiments, the kits can include a compound, or composition provided herein, an optional second agent, or composition, and instructions providing information to a health care provider regarding usage for treating the disorder. Instructions may be provided in printed form, or in the form of an electronic medium such as a floppy disc, CD, or DVD, or in the form of a website address where such instructions may be obtained. A unit dose of a compound, or a composition provided herein, or a second agent, or composition, can include a dosage such that when administered to a subject, a therapeutically, or prophylactically effective plasma level of the compound, or composition can be maintained in the subject for at least one day. In some embodiments, a compound, or composition can be included as a sterile aqueous pharmaceutical composition, or dry powder (e.g., lyophilized) composition.
[00411] In some embodiments, suitable packaging is provided. As used herein, “packaging” includes a solid matrix, or material customarily used in a system, and capable of holding within fixed limits a compound provided herein, and/or a second agent suitable for administration to a subject. Such materials include glass, and plastic (e.g., polyethylene, polypropylene, and polycarbonate) bottles, vials, paper, plastic, plastic-foil laminated envelopes, and the like. If e- beam sterilization techniques are employed, the packaging should have sufficiently low density to permit sterilization of the contents.
Labeled Compounds and Assay Methods
[00412] Another aspect relates to labeled crystalline forms of the present disclosure (radio- labeled, fluorescent-labeled, and so on) that would be useful not only in imaging techniques but also in assays, both in vitro and in vivo.
[00413] The present disclosure further includes isotopically-labeled crystalline forms of the present disclosure. An “isotopically” or “radio-labeled” compound is a crystalline form of the present disclosure where one or more atoms are replaced or substituted by an atom having an atomic mass or mass number different from the atomic mass or mass number typically found in
nature (that is, naturally occurring). Suitable radionuclides that may be incorporated in crystalline forms of the present disclosure include but are not limited to 2H (also written as D for deuterium), 3H (also written as T for tritium), nC, 13C, 14C, 13N, 15N, 1?O, 17O, 18O, 18F, 35S, 36C1, 82Br, 75Br, 76Br, 77Br, 123I, 124I, 125I, and 131I. The radionuclide that is incorporated in the instant radio-labeled compounds will depend on the specific application of that radio-labeled compound. For example, for in vitro metalloprotease labeling and competition assays, compounds that incorporate 3H, 14C, 82Br, 123I, 131I, or 35S will generally be most useful. For radio-imaging applications "C, 18F, 125I, 123I, 124I, 131I, 75Br, 76Br, or 77Br will generally be most useful. In some embodiments, the crystalline forms described herein in which one or more hydrogens is/are replaced by deuterium, such as hydrogen bonded to a carbon atom. Such compounds exhibit increased resistance to metabolism and are thus useful for increasing the half-life of any compound when administered to a mammal, particularly a human. They can also be useful as tools for pharmacokinetic studies.
[00414] It is understood that a “radio-labeled” or “labeled compound” is a compound that has incorporated at least one radionuclide. In some embodiments, the radionuclide is selected from the group consisting of 3H, 14C, 1251, 35S, and 82Br.
[00415] The present disclosure can further include synthetic methods for incorporating radioisotopes into crystalline forms of the present disclosure. Synthetic methods for incorporating radioisotopes into organic compounds are well known in the art, and a person of ordinary skill in the art will readily recognize the methods applicable for the compounds of disclosure.
[00416] A labeled compound of the disclosure can be used in a screening assay to identify/evaluate compounds. For example, a newly synthesized or identified compound (that is, test compound) which is labeled can be evaluated. Conversely, in some other screening assays, the standard compound is labeled, and test compounds are unlabeled. Accordingly, the concentration of the labeled standard compound is monitored in order to evaluate the competition between the standard compound and the test compound, and the relative binding affinity of the test compound is thus ascertained.
EXAMPLES
Instrumental Techniques
[00420] XRPD Method 3
[00421] X-ray diffraction patterns were recorded at ambient conditions in transmission geometry with a STOE STADI P diffractometer (Cu Kai radiation, primary Ge-monochromator, Mythen IK silicon strip detector, angular range 1.5° to 50.5° 2Theta, 0.02° 2Theta step size, 48 seconds step time).
Example 1: Salt Studies
[00430] Compound 1 was used as starting material in preparation of the salt forms.
[00431] Slurry Equilibration. About 40 mg of Compound 1 and 1 equivalent of acid (e.g., L-lysine or L-arginine) or base (e.g., erbumine, sodium hydroxide, or TRIS) was added into a screening solvent (e.g., methanol, ethyl acetate, or dichloromethane) in a 2 mL glass vial. The resulting mixtures were stirred at 50 °C for three days, and 25 °C for at least two days. The
resulting suspensions were filtered through a 0.45 pm nylon membrane filter by centrifugation at 14,000 rpm. After being dried at 50 °C under vacuum for two hours, the collected solids were analyzed by XRPD. Solids obtained: Compound 1 sodium salt Form A, Compound 1 sodium salt Form B; Compound 1 erbumine salt Form A, Compound 1 erbumine salt Form B; and Compound 1 L-lysine.
[00432] By Cooling Clear solutions and hazy suspensions obtained from the slurry equilibration experiments were cooled to 5 °C to precipitate solids. Hazy suspensions were obtained when NaOH, L-arginine, and L-Lysine were the counterions. Clear solutions were obtained when erbumine was the counter ion. A gel was obtained when TRIS was the counterion.
[00433] By Antisolvent addition. Antisolvents were added to the clear solutions and the hazy solutions from the cooling experiments. Suspensions obtained after antisolvent addition were filtered through a 0.45 pm nylon membrane filter by centrifugation at 14,000 rpm. After being dried at 50 °C under vacuum for two hours, the collected solids were analyzed by XRPD. Solids obtained: Compound 1 erbumine salt Form A, Compound 1 erbumine salt Form B, Compound 1 TRIS salt.
[00434] By Slow Evaporation. Clear solutions and the hazy solutions from the cooling experiments were treated by slow evaporation at room temperature.
Example 2A: Preparation of Compound 1 Erbumine Salt Form A
[00435] Compound 1 (100 mg), 19.5 pL (1.05 eq.) of erbumine, and 0.3 mb of methanol were added into a 8 mL glass vial. The mixture was stirred at 50 °C for 2 hours and then 25 °C until the solution was clear. MTBE (1.2 mL) was then added. Seed crystals of Compound 1 erbumine salt Form A were added. After about 2 days, the solids were collected by centrifugation filtration and dried to afford Compound 1 erbumine salt Form A.
Example 2B: Preparation of Compound 1 Erbumine Salt Form C
[00436] Compound 1 (100 mg), 19.5 pL (1.05 eq.) of erbumine, and 0.25 mL ofDCM were added into a 8 mL glass vial. The mixture was stirred at 50 °C for 2 hours and then 25 °C until the solution was clear. Heptane (1 mL) was then added. Seed crystals of Compound 1 erbumine
salt Form B were added. After about 2 days, the solids were collected by centrifugation filtration and dried to afford Compound 1 erbumine salt Form C.
Example 2C: Preparation of Compound 1 Erbumine Salt Form D
[00437] Compound 1 (100 mg), 19.5 pL (1.05 eq.) of erbumine, and 0.3 m of methanol were added into a 8 m glass vial. The mixture was stirred at 50 °C for 2 hours and then 25 °C until the solution was clear. MTBE (6 mb) was then added. Seed crystals of Compound 1 erbumine salt Form A were added. After about 2 days, the solids were collected by centrifugation filtration and dried to afford Compound 1 erbumine salt Form D.
Example 2D: Preparation of Compound 1 Erbumine Salt Form E
[00438] Compound 1 (200 mg), 39 pL (1.05 eq.) of erbumine, and 2 mb of acetonitrile aqueous solution (ACN/water=991/9) were added into a 8-mL glass vial. The mixture was stirred at 25 °C for 1 day, and the solids were collected by filtration and dried to afford Compound 1 erbumine salt Form E as a hydrate at 60% yield.
Example 2E: Preparation of Compound 1 Erbumine Salt Form F
[00439] Compound 1 (40 mg), 19.5 pL (1.05 eq.) of erbumine, and 60 pL of methanol were added into a 8 mL glass vial. The mixture was stirred at 50 °C for 2 hours and then 25 °C until the solution was clear. MTBE (0.4 mL) was then added. The solids were collected by centrifugation filtration and dried to afford Compound 1 erbumine salt Form F.
Example 2F: Preparation of Compound 1 Sodium Salt Form A
[00440] Compound 1 (100 mg) and 7.6 mg ofNaOH (1.05 equiv.) were weighed into an 8 mL glass vial along with 0.5 mL ethyl acetate. The mixture was stirred at 50 °C for about 5 minutes to give a suspension. About 2 mg Compound 1 sodium salt Form A crystals seed were added to the suspension. After stirring at 50 °C for two hours, the suspension was cooled to room temperature for about 6 days. The solids were then collected by filtration and dried to afford Compound 1 sodium salt Form A as a hydrate at 86.39% yield.
Example 2G: Preparation of Compound 1 Sodium Salt Form C and Sodium Salt Form D [00441] Compound 1 (400 mg) was weighed into a 20 mL glass vial and 29 mg of (1.05 equivalent) NaOH was added into the glass vial. 4 mL Acetonitrile (5% water) was added into the glass vial under stirring at 25 °C and a small amount of Compound 1 sodium salt Form C crystal seed was added to the suspension. After stirring for one day, solids were collected by filtration and dried to afford Compound 1 sodium salt Form C as a hydrate at 86.39% yield. Compound 1 sodium salt Form D was obtained by the same process.
Example 3: Relative Stability Testing of Erbumine Salt Form A, Salt Form E, and Salt Form F
[00442] The water activity experiments were conducted at 25 °C in acetonitrile/water systems to determine critical water activity between erbumine salt Form A and erbumine salt Form E. [00443] About 5 mg of erbumine salt Form A and 5 mg of erbumine salt Form E were added into 0.2 mL saturated solutions of the acetonitrile/water systems. The obtained suspensions were stirred at 25°C. Solid parts (wet cakes) were isolated by filtration and investigated by XRPD. Table 12 below show the results of the testing.
Table 12.
[00444] The water activity experiments were also conducted at 25°C in acetonitrile/water systems to determine critical water activity between erbumine salt Pattern E and erbumine salt Pattern F.
[00445] About 5 mg of erbumine salt Form F was add into the suspension (Table 12), and with 0.2 mL saturated solutions Form E in the acetonitrile/water systems were added. The obtained suspensions were stirred at 25 °C. Solid parts (wet cakes) were isolated by filtration and investigated by XRPD. Table 13 below show the results of the testing.
Table 13.
[00446] Table 12 and Table 13 show that in the solvent systems tested, erbumine salt Form E was obtained in most of the solvent systems. Thus, erbumine salt Form E is the most thermodynamically stable salt form among Compound 1 Erbumine Salt Form A, Form E, and Form F.
Example 4: Bulk Stability of Compound 1 Erbumine Salt Form E and Sodium Salt Form C
[00447] Compound 1 Erbumine Salt Form E and Sodium Salt Form C were each placed at 25°C/92.5% RH in an open container, at 40°C/75% RH in an open container, and at 60°C in a closed container for 1 week. The samples after the stress were characterized by XRPD and HPLC and inspected for color change. Table 14 shows the test results.
Table 14.
Results of Bulk Stability Test of Erbumine Salt Form E and Sodium Salt Form C
[00448] Table 14 shows that Compound 1 Erbumine Salt Form E and Sodium Salt Form C are stable under stressed conditions. There were no form changes and nor change of color.
Example 5: Solubility of Compound 1 Erbumine Salt Form E and Sodium Salt Form C [00449] Compound 1 Erbumine Salt Form E and Sodium Salt Form C (8 mg each) were each weighed into a 8 mL glass vial. 4mL of solubility medium was added. Obtained suspensions were stirred at 37°C at 600 rpm and sampled at 2 hours and at 24 hours. Filtration and supernatants were analyzed by HPLC for solubility.
Table 15.
Other Embodiments
[00450J The foregoing disclosure has been described in some detail by way of illustration and example, for purposes of clarity and understanding. The disclosure has been described with reference to various specific and preferred embodiments and techniques. However, it should be understood that many variations and modifications can be made while remaining within the spirit and scope of the disclosure. It will be obvious to one of skill in the art that changes and modifications can be practiced within the scope of the appended claims. Therefore, it is to be understood that the above description is intended to be illustrative and not restrictive.
[00451] The scope of the disclosure should, therefore, be determined not with reference to the above description, but should instead be determined with reference to the following appended claims, along with the full scope of equivalents to which such claims are entitled.
Claims
Compound 1 wherein said salt is selected from the group consisting of Compound 1 TRIS salt, Compound 1 L-arginine salt, Compound 1 L-lysine salt, Compound 1 sodium salt, and Compound 1 erbumine salt.
2. The salt of Compound 1 of claim 1, wherein said salt is a solvate, hydrate, or anhydrate.
3. The salt of Compound 1 of claims 1 or 2, wherein said salt is amorphous or crystalline.
4. The salt of Compound 1 of any one of claims 1-3, wherein said salt is crystalline and is selected from the group consisting of Compound 1 TRIS salt Form A, Compound 1 sodium salt Form A, Compound 1 sodium salt Form B, Compound 1 sodium salt Form C, Compound 1 sodium salt Form D, Compound 1 erbumine salt Form A, Compound 1 erbumine salt Form B, Compound 1 erbumine salt Form C, Compound 1 erbumine salt Form D, Compound 1 erbumine salt Form E, Compound 1 erbumine salt Form F, and Compound 1 L-lysine Form A.
5. The salt of Compound 1 of claim 4, wherein Compound 1 TRIS salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 3.92, 7.41, 7.83, 8.40, 10.76, 11.73, 12.26, 13.87, 14.17, 14.89, 15.29, 15.94, 16.38, 16.77, 17.44, 17.77, 18.21, 18.93, 19.28, 19.63, 19.92, 20.21, 20.46, 21.03, 21.42, 21.90, 22.29, 22.80, 23.46, 24.16, 24.39, 24.678, 24.86, 25.38, 25.62, 26.27, 26.51, 27.47, 27.78, 28.09, 28.58, 29.20, 29.91, 30.78, 31.49, 32.19, 32.80, and 33.47.
6. The salt of Compound 1 of claim 4, wherein Compound 1 TRIS salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising three or more peaks selected from 3.92, 7.41, 7.83, 8.40, 10.76, 11.73, 12.26, 13.87, 14.17, 14.89, 15.29, 15.94, 16.38, 16.77, 17.44, 17.77, 18.21, 18.93, 19.28, 19.63, 19.92, 20.21, 20.46, 21.03, 21.42, 21.90, 22.29, 22.80, 23.46, 24.16, 24.39, 24.678, 24.86, 25.38, 25.62, 26.27, 26.51, 27.47, 27.78, 28.09, 28.58, 29.20, 29.91, 30.78, 31.49, 32.19, 32.80, and 33.47.
7. The salt of Compound 1 of claim 4, wherein Compound 1 TRIS salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising all of the peaks 3.92, 7.41, 7.83, 8.40, 10.76, 11.73, 12.26, 13.87, 14.17, 14.89, 15.29, 15.94, 16.38, 16.77, 17.44,
17.77, 18.21, 18.93, 19.28, 19.63, 19.92, 20.21, 20.46, 21.03, 21.42, 21.90, 22.29, 22.80, 23.46, 24.16, 24.39, 24.678, 24.86, 25.38, 25.62, 26.27, 26.51, 27.47, 27.78, 28.09, 28.58, 29.20, 29.91,
30.78, 31.49, 32.19, 32.80, and 33.47.
8. The salt of Compound 1 of any one of claims 1-7, wherein Compound 1 TRIS salt Form A is characterized by the XRPD pattern substantially in accordance with FIG. 5.
9. The salt of Compound 1 of any one of claims 1-8, wherein Compound 1 TRIS salt Form A is characterized by a DSC thermogram substantially in accordance with FIG. 6.
10. The salt of Compound 1 of any one of claims 1-9, wherein Compound 1 TRIS salt Form A is characterized by a weight loss of about 0.6 weight percent at about 150 °C in a TGA thermogram.
11. The salt of Compound 1 of claim 4, wherein Compound 1 sodium salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 4.79, 9.60, 11.10, 11.88, 13.06, 14.05, 16.20, 18.14, 21.11, 21.81, 23.86, 26.07, 29.00, and 29.94.
12. The salt of Compound 1 of claim 4, wherein Compound 1 sodium salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising three or more peaks
selected from 4.79, 9.60, 11.10, 11.88, 13.06, 14.05, 16.20, 18.14, 21.11, 21.81, 23.86, 26.07, 29.00, and 29.94.
13. The salt of Compound 1 of claim 4, wherein Compound 1 sodium salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising all of the peaks 4.79, 9.60, 11.10, 11.88, 13.06, 14.05, 16.20, 18.14, 21.11, 21.81, 23.86, 26.07, 29.00, and 29.94.
14. The salt of Compound 1 of any one of claims 11-13, wherein Compound 1 sodium salt Form A is characterized by the XRPD pattern substantially in accordance with FIG. 9.
15. The salt of Compound 1 of any one of claims 11-14, wherein Compound 1 sodium salt Form A is characterized by a DSC thermogram substantially in accordance with FIG. 10.
16. The salt of Compound 1 of any one of claims 11-15, wherein Compound 1 sodium salt Form A is characterized by a weight loss of about 5.0 weight percent at about 150 °C in a TGA thermogram.
17. The salt of Compound 1 of claim 4, wherein Compound 1 sodium salt Form B is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 4.49, 4.58, 6.63, 8.84, 9.34, 11.03, 11.74, 13.18, 14.03, 15.44, 15.92, 16.22, 17.11, 17.82, 18.19, 18.85, 19.83, 21.10, 21.73, 22.63, 23.60, 24.36, 24.57, 25.23, 25.72, 26.81, 28.10, 28.77, 29.45, 30.89, 31.60, 35.58, and 35.97.
18. The salt of Compound 1 of claim 4, wherein Compound 1 sodium salt Form B is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising three or more peaks selected from 4.49, 4.58, 6.63, 8.84, 9.34, 11.03, 11.74, 13.18, 14.03, 15.44, 15.92, 16.22, 17.11, 17.82, 18.19, 18.85, 19.83, 21.10, 21.73, 22.63, 23.60, 24.36, 24.57, 25.23, 25.72, 26.81, 28.10, 28.77, 29.45, 30.89, 31.60, 35.58, and 35.97.
19. The salt of Compound 1 of claim 4, wherein Compound 1 sodium salt Form B is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising all of the peaks 4.49,
4.58, 6.63, 8.84, 9.34, 11.03, 11.74, 13.18, 14.03, 15.44, 15.92, 16.22, 17.11, 17.82, 18.19, 18.85,
19.83, 21.10, 21.73, 22.63, 23.60, 24.36, 24.57, 25.23, 25.72, 26.81, 28.10, 28.77, 29.45, 30.89, 31.60, 35.58, and 35.97.
20. The salt of Compound 1 of any one of claims 17-19, wherein Compound 1 sodium salt Form B is characterized by the XRPD pattern substantially in accordance with FIG. 12.
21. The salt of Compound 1 of any one of claims 17-20, wherein Compound 1 sodium salt Form B is characterized by a DSC thermogram substantially in accordance with FIG. 13.
22. The salt of Compound 1 of any one of claims 17-21, wherein Compound 1 sodium salt Form B is characterized by a weight loss of about 6.5 weight percent at about 150 °C in a TGA thermogram.
23. The salt of Compound 1 of claim 4, wherein Compound 1 sodium salt Form C is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 3.11, 5.85, 11.81, 12.20, 12.58, 12.88, 13.46, 14.27, 15.04, 15.98, 16.67, 16.99, 17.68, 18.06, 18.38, 18.79, 19.32, 19.89, 20.43, 20.93, 21.48, 22.17, 23.31, 23.90, 24.42, 24.81, 26.36, 26.95, 27.61, 27.84, 28.42, 28.779, 29.68, 30.06, 30.98, 32.40, 32.90, 33.89, 34.50, 35.13, 36.19, and 37.01.
24. The salt of Compound 1 of claim 4, wherein Compound 1 sodium salt Form C is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising three or more peaks selected from 3.11, 5.85, 11.81, 12.20, 12.58, 12.88, 13.46, 14.27, 15.04, 15.98, 16.67, 16.99, 17.68, 18.06, 18.38, 18.79, 19.32, 19.89, 20.43, 20.93, 21.48, 22.17, 23.31, 23.90, 24.42, 24.81, 26.36, 26.95, 27.61, 27.84, 28.42, 28.779, 29.68, 30.06, 30.98, 32.40, 32.90, 33.89, 34.50, 35.13, 36.19, and 37.01.
25. The salt of Compound 1 of claim 4, wherein Compound 1 sodium salt Form C is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising all of the peaks 3.11, 5.85, 11.81, 12.20, 12.58, 12.88, 13.46, 14.27, 15.04, 15.98, 16.67, 16.99, 17.68, 18.06, 18.38,
18.79, 19.32, 19.89, 20.43, 20.93, 21.48, 22.17, 23.31, 23.90, 24.42, 24.81, 26.36, 26.95, 27.61,
27.84, 28.42, 28.779, 29.68, 30.06, 30.98, 32.40, 32.90, 33.89, 34.50, 35.13, 36.19, and 37.01.
26. The salt of Compound 1 of any one of claims 23-25, wherein Compound 1 sodium salt Form C is characterized by the XRPD pattern substantially in accordance with FIG. 16A.
27. The salt of Compound 1 of any one of claims 23-26, wherein Compound 1 sodium salt Form C is characterized by a DSC-TGA thermogram substantially in accordance with FIG. 17A.
28. The salt of Compound 1 of any one of claims 23-27, wherein Compound 1 sodium salt Form C is characterized by a weight loss of about 0.8 weight percent from 25 °C to 70 °C in a TGA thermogram.
29. The salt of Compound 1 of claim 4, wherein Compound 1 sodium salt Form D is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 5.55, 11.08, 12.23, 12.47, 12.84, 13.05, 13.88, 14.10, 14.73, 15.56, 16.47, 17.09, 17.85, 18.29, 18.63, 19.10, 19.63, 20.31, 20.66, 21.03, 21.80, 22.30, 23.51, 24.57, 25.09, 25.43, 26.13, 26.43, 27.55, 27.96, 28.60, 29.02, 29.67, 30.54, 30.94, 31.85, 34.56, 35.00, 36.02, and
39.50.
30. The salt of Compound 1 of claim 4, wherein Compound 1 sodium salt Form D is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising three or more peaks selected from 5.55, 11.08, 12.23, 12.47, 12.84, 13.05, 13.88, 14.10, 14.73, 15.56, 16.47, 17.09, 17.85, 18.29, 18.63, 19.10, 19.63, 20.31, 20.66, 21.03, 21.80, 22.30, 23.51, 24.57, 25.09, 25.43, 26.13, 26.43, 27.55, 27.96, 28.60, 29.02, 29.67, 30.54, 30.94, 31.85, 34.56, 35.00, 36.02, and
39.50.
31. The salt of Compound 1 of claim 4, wherein Compound 1 sodium salt Form D is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising all of the peaks 5.55, 11.08, 12.23, 12.47, 12.84, 13.05, 13.88, 14.10, 14.73, 15.56, 16.47, 17.09, 17.85, 18.29, 18.63, 19.10, 19.63, 20.31, 20.66, 21.03, 21.80, 22.30, 23.51, 24.57, 25.09, 25.43, 26.13, 26.43, 27.55,
27.96, 28.60, 29.02, 29.67, 30.54, 30.94, 31.85, 34.56, 35.00, 36.02, and 39.50.
32. The salt of Compound 1 of any one of claims 29-31, wherein Compound 1 sodium salt Form D is characterized by the XRPD pattern substantially in accordance with FIG. 16B.
33. The salt of Compound 1 of any one of claims 29-32, wherein Compound 1 sodium salt Form D is characterized by a TG-FTIR thermogram substantially in accordance with FIG. 17B.
34. The salt of Compound 1 of any one of claims 29-33, wherein Compound 1 sodium salt Form D is characterized by a DSC thermogram substantially in accordance with FIG. 17C.
35. The salt of Compound 1 of claim 4, wherein Compound 1 erbumine salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 9.46, 10.26, 11.24, 11.90, 16.94, 17.40, 18.73, 19.40, 20.28, 20.95, and 22.12.
36. The salt of Compound 1 of claim 4, wherein Compound 1 erbumine salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising three or more peaks selected from 9.46, 10.26, 11.24, 11.90, 16.94, 17.40, 18.73, 19.40, 20.28, 20.95, and 22.12.
37. The salt of Compound 1 of claim 4, wherein Compound 1 erbumine salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising all of the peaks 9.46, 10.26, 11.24, 11.90, 16.94, 17.40, 18.73, 19.40, 20.28, 20.95, and 22.12.
38. The salt of Compound 1 of any one of claims 35-37, wherein Compound 1 erbumine salt Form A is characterized by the XRPD pattern substantially in accordance with FIG. 20.
39. The salt of Compound 1 of claim 4, wherein Compound 1 erbumine salt Form B is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 4.74, 5.52, 7.39, 9.40, 9.84, 11.04, 12.35, 14.29, 15.09, 16.49, 17.76, 18.09, 19.26, 20.07, 20.57, 21.06, 22.14, and 23.72.
40. The salt of Compound 1 of claim 4, wherein Compound 1 erbumine salt Form B is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising three or more peaks selected from 4.74, 5.52, 7.39, 9.40, 9.84, 11.04, 12.35, 14.29, 15.09, 16.49, 17.76, 18.09, 19.26, 20.07, 20.57, 21.06, 22.14, and 23.72.
41. The salt of Compound 1 of claim 4, wherein Compound 1 erbumine salt Form B is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising all of the peaks 4.74, 5.52, 7.39, 9.40, 9.84, 11.04, 12.35, 14.29, 15.09, 16.49, 17.76, 18.09, 19.26, 20.07, 20.57, 21.06, 22.14, and 23.72.
42. The salt of Compound 1 of any one of claims 39-41, wherein Compound 1 erbumine salt Form B is characterized by the XRPD pattern substantially in accordance with FIG. 24.
43. The salt of Compound 1 of claim 4, wherein Compound 1 erbumine salt Form C is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 4.00, 7.38, 7.97, 8.48, 9.89, 11.04, 11.36, 13.84, 14.30, 14.77, 15.60, 15.95, 16.14, 16.59, 17.44, 17.64, 18.72, 19.11, 19.58, 19.79, 20.18, 20.62, 21.02, 21.48, 22.20, 22.47, 23.29, 23.79, 25.20, 25.54, 27.21, 27.577, 31.20, 32.00, and 32.70.
44. The salt of Compound 1 of claim 4, wherein Compound 1 erbumine salt Form C is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising three or more peaks selected from 4.00, 7.38, 7.97, 8.48, 9.89, 11.04, 11.36, 13.84, 14.30, 14.77, 15.60, 15.95, 16.14, 16.59, 17.44, 17.64, 18.72, 19.11, 19.58, 19.79, 20.18, 20.62, 21.02, 21.48, 22.20, 22.47, 23.29, 23.79, 25.20, 25.54, 27.21, 27.577, 31.20, 32.00, and 32.70.
45. The salt of Compound 1 of claim 4, wherein Compound 1 erbumine salt Form C is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising all of the peaks 4.00, 7.38, 7.97, 8.48, 9.89, 11.04, 11.36, 13.84, 14.30, 14.77, 15.60, 15.95, 16.14, 16.59, 17.44, 17.64, 18.72, 19.11, 19.58, 19.79, 20.18, 20.62, 21.02, 21.48, 22.20, 22.47, 23.29, 23.79, 25.20, 25.54, 27.21, 27.577, 31.20, 32.00, and 32.70.
46. The salt of Compound 1 of any one of claims 43-45, wherein Compound 1 erbumine salt Form C is characterized by the XRPD pattern substantially in accordance with FIG. 25.
47. The salt of Compound 1 of claim 4, wherein Compound 1 erbumine salt Form D is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 3.19, 5.77, 6.50, 8.86, 9.30, 9.69, 10.59, 11.24, 11.71, 11.96, 12.47, 12.70, 13.38,
14.01, 14.77, 15.48, 16.22, 17.34, 17.77, 18.01, 18.33, 18.75, 19.24, 19.49, 19.78, 20.36, 20.56,
20.89, 21.31, 21.84, 22.24, 23.26, 23.59, 24.32, 24.93, 25.22, 26.83, 27.09, 27.75, 28.32, 28.99,
29.38, 29.76, 31.40, 32.18, 32.39, 33.67, 34.24 , 34.84, 35.37, and 37.34.
48. The salt of Compound 1 of claim 4, wherein Compound 1 erbumine salt Form D is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising three or more peaks selected from 3.19, 5.77, 6.50, 8.86, 9.30, 9.69, 10.59, 11.24, 11.71, 11.96, 12.47, 12.70, 13.38,
14.01, 14.77, 15.48, 16.22, 17.34, 17.77, 18.01, 18.33, 18.75, 19.24, 19.49, 19.78, 20.36, 20.56,
20.89, 21.31, 21.84, 22.24, 23.26, 23.59, 24.32, 24.93, 25.22, 26.83, 27.09, 27.75, 28.32, 28.99,
29.38, 29.76, 31.40, 32.18, 32.39, 33.67, 34.24 , 34.84, 35.37, and 37.34.
49. The salt of Compound 1 of claim 4, wherein Compound 1 erbumine salt Form D is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising all of the peaks 3.19, 5.77, 6.50, 8.86, 9.30, 9.69, 10.59, 11.24, 11.71, 11.96, 12.47, 12.70, 13.38, 14.01, 14.77, 15.48, 16.22, 17.34, 17.77, 18.01, 18.33, 18.75, 19.24, 19.49, 19.78, 20.36, 20.56, 20.89, 21.31, 21.84, 22.24, 23.26, 23.59, 24.32, 24.93, 25.22, 26.83, 27.09, 27.75, 28.32, 28.99, 29.38, 29.76, 31.40, 32.18, 32.39, 33.67, 34.24 , 34.84, 35.37, and 37.34.
50. The salt of Compound 1 of any one of claims 47-49, wherein Compound 1 erbumine salt Form D is characterized by the XRPD pattern substantially in accordance with FIG. 29.
51. The salt of Compound 1 of any one of claims 47-50, wherein Compound 1 erbumine salt Form D is characterized by a DSC-TGA thermogram substantially in accordance with FIG. 30.
52. The salt of Compound 1 of claim 4, wherein Compound 1 erbumine salt Form E is
characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 5.85, 7.49, 8.43, 9.62, 10.43, 11.28, 11.68, 12.53, 13.25, 13.71, 14.82, 15.12, 15.51, 16.37, 17.26, 17.61, 18.20, 18.71, 19.31, 19.91, 20.84, 21.93, 22.37, 23.26, 24.13, 25.96, 27.22, and 30.28.
53. The salt of Compound 1 of claim 4, wherein Compound 1 erbumine salt Form E is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising three or more peaks selected from 5.85, 7.49, 8.43, 9.62, 10.43, 11.28, 11.68, 12.53, 13.25, 13.71, 14.82, 15.12, 15.51, 16.37, 17.26, 17.61, 18.20, 18.71, 19.31, 19.91, 20.84, 21.93, 22.37, 23.26, 24.13, 25.96, 27.22, and 30.28.
54. The salt of Compound 1 of claim 4, wherein Compound 1 erbumine salt Form E is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising all of the peaks 5.85, 7.49, 8.43, 9.62, 10.43, 11.28, 11.68, 12.53, 13.25, 13.71, 14.82, 15.12, 15.51, 16.37, 17.26, 17.61, 18.20, 18.71, 19.31, 19.91, 20.84, 21.93, 22.37, 23.26, 24.13, 25.96, 27.22, and 30.28.
55. The salt of Compound 1 of any one of claims 52-54, wherein Compound 1 erbumine salt Form E is characterized by the XRPD pattern substantially in accordance with FIG. 32.
56. The salt of Compound 1 of any one of claims 52-55, wherein Compound 1 erbumine salt Form E is characterized by a DSC-TGA thermogram substantially in accordance with FIG. 33.
57. The salt of Compound 1 of any one of claims 52-56, wherein Compound 1 erbumine salt Form E is characterized by about 1.1 percent weight loss from 25 °C to 100 °C in a TGA thermogram.
58. The salt of Compound 1 of claim 4, wherein Compound 1 erbumine salt Form F is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 3.17, 5.12, 5.78, 6.47, 7.67, 8.91, 9.72, 10.46, 11.24, 11.96, 13.46, 14.41, 14.80, 15.70, 16.38, 17.11, 17.40, 18.06, 18.38, 18.80, 19.59, 20.63, 20.86, 21.16, 21.94, 22.79, 23.66, 24.580 26.17, and 26.82.
59. The salt of Compound 1 of claim 4, wherein Compound 1 erbumine salt Form F is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising three or more peaks selected from 3.17, 5.12, 5.78, 6.47, 7.67, 8.91, 9.72, 10.46, 11.24, 11.96, 13.46, 14.41, 14.80, 15.70, 16.38, 17.11, 17.40, 18.06, 18.38, 18.80, 19.59, 20.63, 20.86, 21.16, 21.94, 22.79, 23.66, 24.580 26.17, and 26.82.
60. The salt of Compound 1 of claim 4, wherein Compound 1 erbumine salt Form F is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising all of the peaks 3.17, 5.12, 5.78, 6.47, 7.67, 8.91, 9.72, 10.46, 11.24, 11.96, 13.46, 14.41, 14.80, 15.70, 16.38, 17.11, 17.40, 18.06, 18.38, 18.80, 19.59, 20.63, 20.86, 21.16, 21.94, 22.79, 23.66, 24.580 26.17, and 26.82.
61. The salt of Compound 1 of any one of claims 58-60, wherein Compound 1 erbumine salt Form F is characterized by the XRPD pattern substantially in accordance with FIG. 35.
62. The salt of Compound 1 of any one of claims 58-61, wherein Compound 1 erbumine salt Form F is characterized by a DSC-TGA thermogram substantially in accordance with FIG. 36.
63. The salt of Compound 1 of any one of claims 58-62, wherein Compound 1 erbumine salt Form F is characterized by about 1.1 percent weight loss from 30 °C to 80 °C in a TGA thermogram.
64. The salt of Compound 1 of claim 3, wherein said salt is Compound 1 L-arginine salt as characterized substantially in accordance with FIG. 38.
65. The salt of Compound 1 of claim 4, wherein Compound 1 L-lysine salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising one or more peaks selected from 9.68, 13.32, 17.71, 17.96, 19.38, 20.47, 22.83, 23.83, 24.45, and 26.03.
66. The salt of Compound 1 of claim 4, wherein Compound 1 L-lysine salt Form A is
characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising three or more peaks selected from 9.68, 13.32, 17.71, 17.96, 19.38, 20.47, 22.83, 23.83, 24.45, and 26.03.
67. The salt of Compound 1 of claim 4, wherein Compound 1 L-lysine salt Form A is characterized by an XRPD pattern on a 2 Theta scale, ± 0.20, comprising all of the peaks 9.68, 13.32, 17.71, 17.96, 19.38, 20.47, 22.83, 23.83, 24.45, and 26.03.
68. The salt of Compound 1 of claim 4, wherein said salt is Compound 1 L-lysine salt Form A as characterized substantially in accordance with FIG. 39.
69. A pharmaceutical composition comprising a salt of Compound 1 as recited in any one of the preceding claims and one or more pharmaceutically acceptable excipients.
70. A method of treating a metabolic disease or disorder, comprising administering to a subject in need thereof a salt of Compound 1 any one of claims 1-68, or a pharmaceutical composition of claim 69.
71. A salt of Compound 1 of any one of claims 1-68, or the pharmaceutical composition of claim 69 for use in therapy.
72. A salt of Compound 1 of any one of claims 1-68, or the pharmaceutical composition of claim 69, for use in the treatment of a metabolic disease, or disorder.
73. Use of A salt of Compound 1 of any one of claims 1-68, or a pharmaceutical composition of claim 69 for treating a metabolic disease or disorder.
74. Use of a salt of Compound 1 of any one of claims 1-68, or a pharmaceutical composition of claim 69 for the manufacture of a medicament for treating a metabolic disease or disorder.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CNPCT/CN2023/137926 | 2023-12-11 | ||
| CN2023137926 | 2023-12-11 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025128564A1 true WO2025128564A1 (en) | 2025-06-19 |
Family
ID=94083674
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2024/059371 Pending WO2025128564A1 (en) | 2023-12-11 | 2024-12-10 | Salt forms of a glp-1 receptor modulator |
Country Status (3)
| Country | Link |
|---|---|
| AR (1) | AR134641A1 (en) |
| TW (1) | TW202527936A (en) |
| WO (1) | WO2025128564A1 (en) |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2022165076A1 (en) | 2021-01-28 | 2022-08-04 | Carmot Therapeutics, Inc. | Gpcr receptor agonists, pharmaceutical compositions comprising the same, and methods for their use |
| WO2023182869A1 (en) * | 2022-03-25 | 2023-09-28 | Ildong Pharmaceutical Co., Ltd. | Novel salt of glp-1 receptor agonist compound, preparation method thereof and pharmaceutical composition comprising thereof |
| WO2024008174A1 (en) * | 2022-07-07 | 2024-01-11 | 杭州德睿智药科技有限公司 | Salt of glp-1r agonist, preparation method therefor and use thereof |
-
2024
- 2024-12-10 WO PCT/US2024/059371 patent/WO2025128564A1/en active Pending
- 2024-12-11 TW TW113148242A patent/TW202527936A/en unknown
- 2024-12-11 AR ARP240103420A patent/AR134641A1/en unknown
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2022165076A1 (en) | 2021-01-28 | 2022-08-04 | Carmot Therapeutics, Inc. | Gpcr receptor agonists, pharmaceutical compositions comprising the same, and methods for their use |
| WO2023182869A1 (en) * | 2022-03-25 | 2023-09-28 | Ildong Pharmaceutical Co., Ltd. | Novel salt of glp-1 receptor agonist compound, preparation method thereof and pharmaceutical composition comprising thereof |
| WO2024008174A1 (en) * | 2022-07-07 | 2024-01-11 | 杭州德睿智药科技有限公司 | Salt of glp-1r agonist, preparation method therefor and use thereof |
Non-Patent Citations (16)
| Title |
|---|
| "Handbook of Chemistry and Physics", 2006, UNIVERSITY SCIENCE BOOKS |
| "Handbook of Pharmaceutical Additives", 2007, GOWER PUBLISHING COMPANY |
| "March's Advanced Organic Chemistry", 2013, JOHN WILEY & SONS |
| "Remington: The Science and Practice of Pharmacy", 2005, LIPPINCOTT WILLIAMS & WILKINS |
| "The Pharmaceutical Press and the American Pharmaceutical Association", 2009, CRC PRESS |
| ANGULO ET AL., J GASTROENTEROL HEPATOL, vol. 17, 2002, pages 186 - 90 |
| BOMMER ET AL., DIABETES CARE, vol. 41, no. 5, 2018, pages 963 - 970 |
| CHITTURI ET AL., HEPATOLOGY, vol. 35, no. 2, 2002, pages 373 - 9 |
| DEEPAK GUPTA ET AL: "Salts of Therapeutic Agents: Chemical, Physicochemical, and Biological Considerations", MOLECULES, vol. 23, no. 7, 14 July 2018 (2018-07-14), pages 1719, XP055718934, DOI: 10.3390/molecules23071719 * |
| DOYLEEGAN, PHARMACOL. THER., vol. 113, no. 3, 2007, pages 546 - 593 |
| ICH, IMPURITIES: GUIDELINES FOR RESIDUAL SOLVENTS Q3C(R6, 1 December 2023 (2023-12-01), Retrieved from the Internet <URL:https://database.ich.org/sites/default/files/Q3C-R6_GuidelineErrorCorrection2019_0410_O.pdf> |
| KHAN ET AL., J. EPIDEMIOL. GLOB. HEALTH, vol. 10, no. 1, 2020, pages 107 - 111 |
| SHAHVELLA, REV ENDOCR METAB DISORD., vol. 15, no. 3, 2014, pages 181 - 187 |
| SKELLY ET AL., J HEPATOL, vol. 35, 2001, pages 195 - 9 |
| SKRDLA PETER J: "Estimating the maximal solubility advantage of drug salts", INTERNATIONAL JOURNAL OF PHARMACEUTICS, ELSEVIER, AMSTERDAM, NL, vol. 595, 21 January 2021 (2021-01-21), XP086486923, ISSN: 0378-5173, [retrieved on 20210121], DOI: 10.1016/J.IJPHARM.2021.120228 * |
| TRIPATHISRIVASTAVA, MED. SCI. MONIT., vol. 12, no. 7, 2016, pages RA130 - 147 |
Also Published As
| Publication number | Publication date |
|---|---|
| AR134641A1 (en) | 2026-02-04 |
| TW202527936A (en) | 2025-07-16 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| TWI831325B (en) | Naphthyridine derivatives as ATR inhibitors and preparation methods thereof | |
| JP7237544B2 (en) | Bile acid recycling inhibitors for the treatment of cholestatic liver disease in children | |
| JP6227741B2 (en) | Solid form of 3- (6- (1- (2,2-difluorobenzo [d] [1,3] dioxol-5-yl) cyclopropanecarboxamido) -3-methylpyridin-2-yl) benzoic acid | |
| CN101715450B (en) | Azaindole derivatives as CFTR modulators | |
| JP5389030B2 (en) | Isothiazolopyridinone useful for the treatment of (especially) cystic fibrosis | |
| JP5686596B2 (en) | Regulators of cystic fibrosis transmembrane conductance regulator | |
| JP6217939B2 (en) | Bile acid reuse inhibitors for the treatment of hyperbileemia and cholestatic liver disease | |
| CA2852991C (en) | Modulators of atp-binding cassette transporters | |
| JP5885359B2 (en) | Isoquinoline regulator of ATP binding cassette transporter | |
| CN118834208A (en) | Solid state form | |
| TW200815422A (en) | Heteroaryl derivatives as cytokine inhibitors | |
| JP2017530175A (en) | Cocrystals of modulators of cystic fibrosis transmembrane conductance regulator | |
| EA030430B1 (en) | Compositions and methods for modulating farnesoid x receptors | |
| HUE026220T2 (en) | Pyridyl derivatives as CFTR modulators | |
| EP2655369A1 (en) | Compositions and methods for modulating farnesoid x receptors | |
| US20230374030A1 (en) | Solid-state forms of relugolix | |
| TW201900652A (en) | A method for preparing a heterocyclic derivative compound, a composition comprising the same, and a hydrate of the compound | |
| BR112019018418A2 (en) | crystalline forms of obeticolic acid | |
| WO2025128564A1 (en) | Salt forms of a glp-1 receptor modulator | |
| EA028059B1 (en) | Solid state forms of n-((s)-2,3-dihydroxy-propyl)-3-(2-fluoro-4-iodo-phenylamino)isonicotinamide | |
| WO2025128561A1 (en) | Crystalline forms of a glp-1 receptor modulator | |
| ES2970120T3 (en) | Potassium salt monohydrate of a thienopyridone derivative and its preparation process | |
| WO2025011259A1 (en) | Crystal form of resmetirom, and preparation method therefor and use thereof | |
| US20260091022A1 (en) | Pharmaceutical composition for treating fatty liver disease | |
| US20250041279A1 (en) | Therapeutic medicine for heart disease or skeletal muscle disease |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24828679 Country of ref document: EP Kind code of ref document: A1 |