CN111574415A - Synthetic method of perchloromethylmercaptan - Google Patents
Synthetic method of perchloromethylmercaptan Download PDFInfo
- Publication number
- CN111574415A CN111574415A CN202010461258.7A CN202010461258A CN111574415A CN 111574415 A CN111574415 A CN 111574415A CN 202010461258 A CN202010461258 A CN 202010461258A CN 111574415 A CN111574415 A CN 111574415A
- Authority
- CN
- China
- Prior art keywords
- reaction
- sulfur dichloride
- perchloromethylmercaptan
- trichloromethane
- triethylamine
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- RYFZYYUIAZYQLC-UHFFFAOYSA-N perchloromethyl mercaptan Chemical compound ClSC(Cl)(Cl)Cl RYFZYYUIAZYQLC-UHFFFAOYSA-N 0.000 title claims abstract description 33
- 238000010189 synthetic method Methods 0.000 title claims abstract description 7
- 238000006243 chemical reaction Methods 0.000 claims abstract description 45
- FWMUJAIKEJWSSY-UHFFFAOYSA-N sulfur dichloride Chemical compound ClSCl FWMUJAIKEJWSSY-UHFFFAOYSA-N 0.000 claims abstract description 39
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 claims abstract description 36
- 229960001701 chloroform Drugs 0.000 claims abstract description 33
- 238000000034 method Methods 0.000 claims abstract description 20
- 150000007530 organic bases Chemical class 0.000 claims abstract description 12
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical group CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 claims description 54
- 239000012295 chemical reaction liquid Substances 0.000 claims description 10
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 9
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 claims description 8
- 238000005406 washing Methods 0.000 claims description 8
- 239000003513 alkali Substances 0.000 claims description 7
- 238000001308 synthesis method Methods 0.000 claims description 6
- 230000008569 process Effects 0.000 claims description 5
- 238000003786 synthesis reaction Methods 0.000 claims description 5
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 claims description 4
- 230000015572 biosynthetic process Effects 0.000 claims description 4
- VKYKSIONXSXAKP-UHFFFAOYSA-N hexamethylenetetramine Chemical compound C1N(C2)CN3CN1CN2C3 VKYKSIONXSXAKP-UHFFFAOYSA-N 0.000 claims description 4
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 claims description 4
- 239000000758 substrate Substances 0.000 claims description 4
- 238000002156 mixing Methods 0.000 claims description 3
- 238000004321 preservation Methods 0.000 claims description 3
- 150000003839 salts Chemical class 0.000 claims description 3
- 238000001914 filtration Methods 0.000 claims description 2
- 229910052757 nitrogen Inorganic materials 0.000 claims description 2
- 230000002194 synthesizing effect Effects 0.000 claims description 2
- VHHHONWQHHHLTI-UHFFFAOYSA-N hexachloroethane Chemical compound ClC(Cl)(Cl)C(Cl)(Cl)Cl VHHHONWQHHHLTI-UHFFFAOYSA-N 0.000 claims 1
- 235000010299 hexamethylene tetramine Nutrition 0.000 claims 1
- QGJOPFRUJISHPQ-UHFFFAOYSA-N Carbon disulfide Chemical compound S=C=S QGJOPFRUJISHPQ-UHFFFAOYSA-N 0.000 abstract description 21
- 239000007788 liquid Substances 0.000 abstract description 15
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 abstract description 4
- 239000002253 acid Substances 0.000 abstract description 4
- 239000011230 binding agent Substances 0.000 abstract description 4
- 239000003054 catalyst Substances 0.000 abstract description 4
- 239000000460 chlorine Substances 0.000 abstract description 4
- 229910052801 chlorine Inorganic materials 0.000 abstract description 4
- 239000002994 raw material Substances 0.000 abstract description 3
- 238000003756 stirring Methods 0.000 description 14
- 239000000047 product Substances 0.000 description 11
- 239000006227 byproduct Substances 0.000 description 8
- 238000010438 heat treatment Methods 0.000 description 7
- 238000000967 suction filtration Methods 0.000 description 7
- ILWRPSCZWQJDMK-UHFFFAOYSA-N triethylazanium;chloride Chemical compound Cl.CCN(CC)CC ILWRPSCZWQJDMK-UHFFFAOYSA-N 0.000 description 5
- KZBUYRJDOAKODT-UHFFFAOYSA-N Chlorine Chemical compound ClCl KZBUYRJDOAKODT-UHFFFAOYSA-N 0.000 description 4
- JLTDJTHDQAWBAV-UHFFFAOYSA-N N,N-dimethylaniline Chemical compound CN(C)C1=CC=CC=C1 JLTDJTHDQAWBAV-UHFFFAOYSA-N 0.000 description 4
- 230000000052 comparative effect Effects 0.000 description 4
- 238000004519 manufacturing process Methods 0.000 description 4
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- 239000003795 chemical substances by application Substances 0.000 description 3
- 230000000694 effects Effects 0.000 description 3
- 229920001971 elastomer Polymers 0.000 description 3
- 239000005060 rubber Substances 0.000 description 3
- 239000013064 chemical raw material Substances 0.000 description 2
- 238000001514 detection method Methods 0.000 description 2
- IXCSERBJSXMMFS-UHFFFAOYSA-N hydrogen chloride Substances Cl.Cl IXCSERBJSXMMFS-UHFFFAOYSA-N 0.000 description 2
- 229910000041 hydrogen chloride Inorganic materials 0.000 description 2
- 239000000463 material Substances 0.000 description 2
- 239000003960 organic solvent Substances 0.000 description 2
- AOJFQRQNPXYVLM-UHFFFAOYSA-N pyridin-1-ium;chloride Chemical compound [Cl-].C1=CC=[NH+]C=C1 AOJFQRQNPXYVLM-UHFFFAOYSA-N 0.000 description 2
- 230000035484 reaction time Effects 0.000 description 2
- 239000002351 wastewater Substances 0.000 description 2
- LDVVMCZRFWMZSG-OLQVQODUSA-N (3ar,7as)-2-(trichloromethylsulfanyl)-3a,4,7,7a-tetrahydroisoindole-1,3-dione Chemical compound C1C=CC[C@H]2C(=O)N(SC(Cl)(Cl)Cl)C(=O)[C@H]21 LDVVMCZRFWMZSG-OLQVQODUSA-N 0.000 description 1
- 239000005745 Captan Substances 0.000 description 1
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 1
- 239000005789 Folpet Substances 0.000 description 1
- 244000043261 Hevea brasiliensis Species 0.000 description 1
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 1
- LSDPWZHWYPCBBB-UHFFFAOYSA-N Methanethiol Chemical compound SC LSDPWZHWYPCBBB-UHFFFAOYSA-N 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 230000003213 activating effect Effects 0.000 description 1
- 230000000844 anti-bacterial effect Effects 0.000 description 1
- 239000003899 bactericide agent Substances 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 229940117949 captan Drugs 0.000 description 1
- QGJOPFRUJISHPQ-NJFSPNSNSA-N carbon disulfide-14c Chemical compound S=[14C]=S QGJOPFRUJISHPQ-NJFSPNSNSA-N 0.000 description 1
- 229950005499 carbon tetrachloride Drugs 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 230000007547 defect Effects 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 230000007613 environmental effect Effects 0.000 description 1
- KQTVWCSONPJJPE-UHFFFAOYSA-N etridiazole Chemical compound CCOC1=NC(C(Cl)(Cl)Cl)=NS1 KQTVWCSONPJJPE-UHFFFAOYSA-N 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 239000006260 foam Substances 0.000 description 1
- HKIOYBQGHSTUDB-UHFFFAOYSA-N folpet Chemical compound C1=CC=C2C(=O)N(SC(Cl)(Cl)Cl)C(=O)C2=C1 HKIOYBQGHSTUDB-UHFFFAOYSA-N 0.000 description 1
- 239000007789 gas Substances 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- 229910052739 hydrogen Inorganic materials 0.000 description 1
- NGDSBQHTMKGUQU-UHFFFAOYSA-N methenamine hydrochloride Chemical compound Cl.C([N@@](C1)C2)[N@]3C[N@@]2C[N@@]1C3 NGDSBQHTMKGUQU-UHFFFAOYSA-N 0.000 description 1
- XAUGWFWQVYXATQ-UHFFFAOYSA-N n-phenylbenzenesulfonamide Chemical compound C=1C=CC=CC=1S(=O)(=O)NC1=CC=CC=C1 XAUGWFWQVYXATQ-UHFFFAOYSA-N 0.000 description 1
- 229920003052 natural elastomer Polymers 0.000 description 1
- 229920001194 natural rubber Polymers 0.000 description 1
- -1 organic base hydrochloride Chemical class 0.000 description 1
- 239000000575 pesticide Substances 0.000 description 1
- 230000001737 promoting effect Effects 0.000 description 1
- 238000013040 rubber vulcanization Methods 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 229920003051 synthetic elastomer Polymers 0.000 description 1
- 239000005061 synthetic rubber Substances 0.000 description 1
- VZGDMQKNWNREIO-UHFFFAOYSA-N tetrachloromethane Chemical compound ClC(Cl)(Cl)Cl VZGDMQKNWNREIO-UHFFFAOYSA-N 0.000 description 1
- 229960002447 thiram Drugs 0.000 description 1
- KUAZQDVKQLNFPE-UHFFFAOYSA-N thiram Chemical compound CN(C)C(=S)SSC(=S)N(C)C KUAZQDVKQLNFPE-UHFFFAOYSA-N 0.000 description 1
- 238000004073 vulcanization Methods 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C319/00—Preparation of thiols, sulfides, hydropolysulfides or polysulfides
- C07C319/02—Preparation of thiols, sulfides, hydropolysulfides or polysulfides of thiols
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
Abstract
The invention discloses a synthetic method of perchloromethylmercaptan, which is obtained by taking trichloromethane and sulfur dichloride as reaction raw materials, organic base as a catalyst and an acid-binding agent. The method is safe and simple, has strong operability, avoids the use of carbon disulfide and chlorine, effectively solves the potential safety hazard caused by the use of carbon disulfide and chlorine, has higher safety, ensures that the obtained product is yellow oily liquid in appearance, has the yield of more than 85 percent and the density of more than 1.69, meets the use requirement, and has industrial application value.
Description
Technical Field
The invention relates to a synthetic method of perchloromethylmercaptan as an organic chemical raw material, in particular to a synthetic method of perchloromethylmercaptan, which has the advantages of simple and safe production process, small waste water amount, environmental protection and high product yield, and belongs to the technical field of organic synthesis.
Background
The perchloromethylmercaptan is an important chemical raw material for pesticide bactericides such as captan, folpet, terrazole and the like, and the perchloromethylmercaptan is also a key synthetic raw material of a rubber scorch retarder V.E (also called as a scorch retarder E). The scorch retarder V.E is developed by Bayer company in Germany and is dedicated to light-colored rubber vulcanization, is pollution-free and non-colored, does not foam during high-temperature mixing, is effective for sulfenamides, and is more suitable for vulcanization systems of mercaptan and thiuram accelerators. The rubber is widely applied to various industries such as daily life, medical industry, rail transit industry and the like at present, and has an irreplaceable effect in the life of people. Natural rubber and synthetic rubber are widely used in the current society and daily life, and with the continuous development of economy, the demand of rubber in various industries is continuously increased, so that the demand of the scorch retarder V.E is also continuously increased, which also results in the large increase of the demand of perchloromethylmercaptan.
At present, the synthesis process of perchloromethylmercaptan generally adopts a method of introducing chlorine gas into carbon disulfide, and the process has the problems of long reaction time, low product yield and escape of carbon disulfide and chlorine gas from a system in the reaction process, the escape of carbon disulfide has certain potential safety hazard, the escape of chlorine gas needs a tail gas absorption device, and simultaneously has larger smell, thus influencing the physical health of an operator and improving the production cost.
Disclosure of Invention
Aiming at the defects in the prior art, the invention provides a synthetic method of perchloromethylmercaptan, which takes trichloromethane and sulfur dichloride as substrates and takes organic base as a catalyst and an acid-binding agent, so that the perchloromethylmercaptan is conveniently and rapidly synthesized.
The specific technical scheme of the invention is as follows:
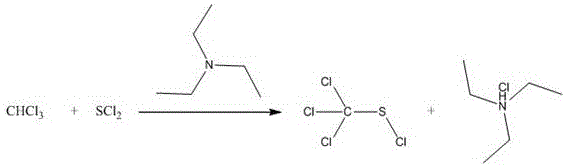
a process for synthesizing perchloromethylmercaptan features that trichloromethane and sulfur dichloride are used as substrate and the perchloromethylmercaptan is synthesized in the presence of organic alkali. The organic base is used as a catalyst and an acid-binding agent, so that the action of activating the trichloromethane is achieved, the leaving of hydrogen is promoted, and the reaction activity of the trichloromethane is improved; on the other hand, the reaction kettle plays a role in capturing by-product hydrogen chloride so as to ensure that the reaction is carried out in a forward direction.
Further, the organic base may be any organic base which can react with hydrogen chloride to form a salt and has a promoting effect on the activity of chloroform, and may be triethylamine, pyridine, urotropine and the like, and the commonly used organic base is triethylamine.
Furthermore, when the organic base is triethylamine, the yield of the product is higher, and the operability is stronger. The reaction equation is as follows:
further, in the above synthesis method, the molar ratio of nitrogen in chloroform, sulfur dichloride and organic base is 1.05-1.2: 1: 1-1.2, preferably 1.05-1.1: 1:1.
furthermore, the synthesis method does not need to carry out reaction in an organic solvent, and the cost is lower. The method specifically comprises the following steps:
(1) mixing trichloromethane and organic alkali, and then dropwise adding sulfur dichloride for reaction;
(2) and treating the reaction liquid after the reaction to obtain perchloromethylmercaptan.
Further, in the step (1), sulfur dichloride is dripped at the temperature of 0-20 ℃, and the heat preservation reaction is continuously carried out at the temperature after the dripping is finished. The dripping time of the common sulfur dichloride is controlled to be 1-3h, and the heat preservation reaction is continued for 1-2h after the dripping of the sulfur dichloride is finished. After the reaction, the reaction solution contains perchloromethane alcohol and organic base hydrochloride, and also contains a trace amount of unreacted trichloromethane and organic base.
Further, in the step (2), the post-treatment process of the reaction solution is as follows: and after the reaction, filtering to remove salts formed in the reaction, washing the residual reaction solution with water to remove unreacted organic alkali, and distilling the residual reaction solution to remove trichloromethane to obtain perchloromethylmercaptan.
Compared with the traditional process for preparing perchloromethylmercaptan by introducing chlorine gas, carbon disulfide and dilute hydrochloric acid, the method has the following advantages:
1. trichloromethane and sulfur dichloride are used as substrates, so that the use of carbon disulfide and chlorine is avoided, the potential safety hazard caused by the use of carbon disulfide and chlorine is effectively solved, and the safety is higher.
2. The invention uses organic alkali as catalyst and acid-binding agent, which is more beneficial to the forward reaction, the reaction temperature is lower, and the reaction time is short.
3. The method has the advantages of safe and simple production process and strong operability, the obtained product is yellow oily liquid in appearance, the yield is more than 85 percent, the density is more than 1.69, the use requirement is met, and the method has industrial application value.
4. The method does not need to use an organic solvent, has lower cost, simple post-treatment and less generated waste water, and is more environment-friendly.
Detailed Description
The present invention is further illustrated by the following examples, which are intended to be purely exemplary and are not intended to be limiting.
In the following examples, the yield was calculated as: quality of the obtained product/theoretical quality of the product.
Example 1
154.10g of trichloromethane and 124.39g of triethylamine are added into a 1L reaction kettle, the temperature is controlled to be 10 ℃, stirring is started, 126.58g of sulfur dichloride is slowly dripped into the system, after about 2 hours, the dripping of the sulfur dichloride is finished, the temperature is kept, stirring is continued for about 2 hours, and the reaction is finished. And (3) carrying out suction filtration on the obtained reaction liquid to remove a byproduct triethylamine hydrochloride, then adding about 100g of water to wash to remove trace unreacted triethylamine, then heating the collected lower layer liquid to 65 ℃, distilling off trace unreacted trichloromethane to finally obtain 210.6g of yellow oily liquid, namely perchloromethylmercaptan, wherein the yield is 92.21% in terms of sulfur dichloride, and the density is 1.69 by detection.
The synthetic experiment of the scorch retarder E is carried out by taking perchloromethylmercaptan as a raw material, and the specific method comprises the following steps: controlling the system temperature to be 10 ℃, dropwise adding perchloromethylmercaptan into a reaction system containing N-phenyl benzene sulfonamide, solvent oil and a sodium hydroxide solution, centrifuging and drying after the reaction is finished, wherein the purity of the obtained antiscorching agent E is 99.15%, and the yield and purity of the antiscorching agent E are similar to those of the antiscorching agent E synthesized by purchased perchloromethylmercaptan, so that the use requirement is met.
Example 2
151g of trichloromethane and 116.34g of triethylamine are added into a 1L reaction kettle, the temperature is controlled to be 10 ℃, stirring is started, 118.39g of sulfur dichloride is slowly dripped into the system, after about 2 hours, the dripping of the sulfur dichloride is finished, the temperature is kept, stirring is continued for about 2 hours, and the reaction is finished. And carrying out suction filtration on the obtained reaction liquid to remove a byproduct triethylamine hydrochloride, then adding about 100g of water for washing to remove trace unreacted triethylamine, then heating the collected lower layer liquid to 65 ℃, distilling out trace unreacted trichloromethane, and finally obtaining 199.2g of yellow oily liquid, namely perchloromethane thiol, wherein the yield is 93.25% in terms of sulfur dichloride, and the density is 1.69 through detection, so that the use requirement is met.
Example 3
167.28g of trichloromethane and 118.15g of triethylamine are added into a 1L reaction kettle, the temperature is controlled to be 0 ℃, stirring is started, 120.23g of sulfur dichloride is slowly dripped into the system, after about 1 hour, the dripping of the sulfur dichloride is finished, the temperature is kept, stirring is continued for about 1 hour, and the reaction is finished. And (3) carrying out suction filtration on the obtained reaction liquid to remove a byproduct triethylamine hydrochloride, then adding about 100g of water for washing to remove trace unreacted triethylamine, then heating the collected lower layer liquid to 65 ℃, distilling out trace unreacted trichloromethane, and finally obtaining 195.78g of yellow oily liquid, namely perchloromethylmercaptan, wherein the yield is 90.24% in terms of sulfur dichloride, and the detected density is 1.69, so that the use requirement is met.
Example 4
Adding 135.63g of trichloromethane and 104.50g of triethylamine into a 1L reaction kettle, controlling the temperature to be 20 ℃, starting stirring, slowly dropwise adding 106.34g of sulfur dichloride into the system, after about 2 hours, completing dropwise adding of sulfur dichloride, keeping the temperature, continuously stirring for about 2 hours, and completing the reaction. And (3) carrying out suction filtration on the obtained reaction liquid to remove a byproduct triethylamine hydrochloride, then adding about 100g of water for washing to remove trace unreacted triethylamine, then heating the collected lower layer liquid to 65 ℃, distilling out trace unreacted trichloromethane, and finally obtaining 175.21g of yellow oily liquid, namely perchloromethylmercaptan, wherein the yield is 91.31% in terms of sulfur dichloride, and the detected density is 1.69, so that the use requirement is met.
Example 5
172.97g of trichloromethane and 133.28g of triethylamine are added into a 1L reaction kettle, the temperature is controlled to be 10 ℃, stirring is started, 135.62g of sulfur dichloride is slowly dripped into the system, after about 3 hours, the dripping of the sulfur dichloride is finished, the temperature is kept, stirring is continuously carried out for about 2 hours, and the reaction is finished. And (3) carrying out suction filtration on the obtained reaction liquid to remove a byproduct triethylamine hydrochloride, then adding about 100g of water for washing to remove trace unreacted triethylamine, then heating the collected lower layer liquid to 65 ℃, distilling out trace unreacted trichloromethane, and finally obtaining 231.25g of yellow oily liquid, namely perchloromethylmercaptan, wherein the yield is 94.50% in terms of sulfur dichloride, and the detected density is 1.69, so that the use requirement is met.
Example 6
152.86g of trichloromethane and 98.77g of pyridine are added into a 1L reaction kettle, the temperature is controlled to be 10 ℃, stirring is started, 128.57g of sulfur dichloride is slowly dripped into the system, after about 2 hours, the dripping of the sulfur dichloride is finished, the temperature is kept, the stirring is continued for about 2 hours, and the reaction is finished. And (3) carrying out suction filtration on the obtained reaction liquid to remove a byproduct, namely pyridine hydrochloride, then adding about 100g of water for washing to remove trace unreacted pyridine, then heating the collected lower layer liquid to 65 ℃, distilling out unreacted trichloromethane, and finally obtaining 207.26g of yellow oily liquid, namely perchloromethylmercaptan, wherein the yield is 89.34% in terms of sulfur dichloride, and the detected density is 1.69, so that the use requirement is met.
Example 7
142.87g of trichloromethane and 47.93g of urotropine are added into a 1L reaction kettle, the temperature is controlled to be 10 ℃, stirring is started, 117.36g of sulfur dichloride is slowly dripped into the system, after about 2 hours, the dripping of the sulfur dichloride is finished, the temperature is kept, the stirring is continued for about 2 hours, and the reaction is finished. And (3) carrying out suction filtration on the obtained reaction liquid, removing byproducts, namely urotropine hydrochloride and unreacted urotropine, adding about 100g of water for washing, heating the collected subnatant to 65 ℃, and distilling out unreacted trichloromethane to finally obtain 185.36g of yellow oily liquid, namely perchloromethylmercaptan, wherein the yield is 87.53% in terms of sulfur dichloride, and the detected density is 1.69, so that the use requirement is met.
Comparative example 1
Perchloromethylmercaptan was synthesized according to the procedure of example 2, except that: the dropwise addition and the reaction temperature were controlled to 40 ℃. The yield of the obtained product is 80.23%, the detected density is 1.67, and the purity of the synthesized V.E is 95.23%, which does not meet the use requirement.
Comparative example 2
Perchloromethylmercaptan was synthesized according to the procedure of example 2, except that: the molar ratio of trichloromethane to sulfur dichloride to triethylamine is 1.3:1: 1.1. The product yield was 93.30%, with a detected density of 1.69. It can be seen that the product yield does not change much with increasing chloroform dosage, but the cost is increased.
Comparative example 3
Perchloromethylmercaptan was synthesized according to the procedure of example 2, except that: n, N-dimethylaniline is used for replacing triethylamine, and the materials are fed according to the molar ratio of 1.05:1:1 of trichloromethane, sulfur dichloride and N, N-dimethylaniline. The yield of the obtained product is 73.65 percent, the detected density is 1.66, and the use requirement is not met.
Comparative example 4
Perchloromethylmercaptan was synthesized according to the procedure of example 2, except that: azodiisopropylnitrile is used for replacing triethylamine, and the materials are fed according to the molar ratio of 1.05:1:1 of trichloromethane, sulfur dichloride and azodiisopropylnitrile. The yield of the obtained product is 42.36 percent, and the detected density is 1.63, so that the use requirement is not met.
Claims (8)
1. A synthetic method of perchloromethylmercaptan is characterized by comprising the following steps: using trichloromethane and sulfur dichloride as substrates, and synthesizing the perchloroethane in the presence of organic alkali.
2. The method of synthesis according to claim 1, comprising the steps of:
(1) mixing trichloromethane and organic alkali, and then dropwise adding sulfur dichloride for reaction;
(2) and treating the reaction liquid after the reaction to obtain perchloromethylmercaptan.
3. The synthesis method according to claim 1 or 2, wherein: the organic base is triethylamine, pyridine or urotropin.
4. The synthesis method according to claim 1 or 2, wherein: the organic base is triethylamine.
5. The synthesis method according to claim 1 or 2, wherein: the molar ratio of the nitrogen in the trichloromethane, the sulfur dichloride and the organic base is 1.05-1.2: 1: 1-1.2, preferably 1.05-1.1: 1:1.
6. the method of synthesis according to claim 2, wherein: in the step (1), sulfur dichloride is dripped at the temperature of 0-20 ℃, and the heat preservation reaction is continuously carried out at the temperature after the dripping is finished.
7. The synthesis method according to claim 2 or 6, wherein: in the step (1), the dripping time of the sulfur dichloride is 1-3h, and the reaction is continued for 1-2h after the dripping of the sulfur dichloride is finished.
8. The method of synthesis according to claim 2, wherein: the post-treatment process of the reaction liquid comprises the following steps: and after the reaction, filtering to remove salts formed in the reaction, washing the residual reaction solution with water to remove unreacted organic alkali, and distilling the residual reaction solution to remove trichloromethane to obtain perchloromethylmercaptan.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202010461258.7A CN111574415B (en) | 2020-05-27 | 2020-05-27 | Synthetic method of perchloromethylmercaptan |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202010461258.7A CN111574415B (en) | 2020-05-27 | 2020-05-27 | Synthetic method of perchloromethylmercaptan |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN111574415A true CN111574415A (en) | 2020-08-25 |
| CN111574415B CN111574415B (en) | 2021-12-14 |
Family
ID=72121642
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202010461258.7A Active CN111574415B (en) | 2020-05-27 | 2020-05-27 | Synthetic method of perchloromethylmercaptan |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN111574415B (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN112094209A (en) * | 2020-09-17 | 2020-12-18 | 山东阳谷华泰化工股份有限公司 | Method for preparing scorch retarder N-phenyl-N-trichloromethylthio benzene sulfonamide |
| CN114957049A (en) * | 2022-06-13 | 2022-08-30 | 河北冠龙农化有限公司 | Synthesis method of N-phenyl-N-trichloromethylthio benzenesulfonamide |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN103360295A (en) * | 2012-04-06 | 2013-10-23 | 英德广农康盛化工有限责任公司 | Device and method for preparing perchloromethylmercaptan |
| CN103755600A (en) * | 2013-12-27 | 2014-04-30 | 爱斯特(成都)医药技术有限公司 | Process for synthesizing N,N-dimethyl-N-phenyl-(N-fluorodichloromethylmercapto)-sulfamide |
| CN107778221A (en) * | 2016-08-26 | 2018-03-09 | 上海雅本化学有限公司 | A kind of preparation technology of 2,3 dichloropyridine |
| CN110950826A (en) * | 2019-12-10 | 2020-04-03 | 湖南海利常德农药化工有限公司 | Preparation method of carbosulfan |
-
2020
- 2020-05-27 CN CN202010461258.7A patent/CN111574415B/en active Active
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN103360295A (en) * | 2012-04-06 | 2013-10-23 | 英德广农康盛化工有限责任公司 | Device and method for preparing perchloromethylmercaptan |
| CN103755600A (en) * | 2013-12-27 | 2014-04-30 | 爱斯特(成都)医药技术有限公司 | Process for synthesizing N,N-dimethyl-N-phenyl-(N-fluorodichloromethylmercapto)-sulfamide |
| CN107778221A (en) * | 2016-08-26 | 2018-03-09 | 上海雅本化学有限公司 | A kind of preparation technology of 2,3 dichloropyridine |
| CN110950826A (en) * | 2019-12-10 | 2020-04-03 | 湖南海利常德农药化工有限公司 | Preparation method of carbosulfan |
Non-Patent Citations (1)
| Title |
|---|
| 尹拥军等: "克菌丹原药合成工艺的改进研究", 《山东化工》 * |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN112094209A (en) * | 2020-09-17 | 2020-12-18 | 山东阳谷华泰化工股份有限公司 | Method for preparing scorch retarder N-phenyl-N-trichloromethylthio benzene sulfonamide |
| CN114957049A (en) * | 2022-06-13 | 2022-08-30 | 河北冠龙农化有限公司 | Synthesis method of N-phenyl-N-trichloromethylthio benzenesulfonamide |
| CN114957049B (en) * | 2022-06-13 | 2023-10-03 | 河北冠龙农化有限公司 | Synthesis method of N-phenyl-N-trichloromethylthiobenzene sulfonamide |
Also Published As
| Publication number | Publication date |
|---|---|
| CN111574415B (en) | 2021-12-14 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN111574415B (en) | Synthetic method of perchloromethylmercaptan | |
| WO2003045904A1 (en) | Process for production of methionine | |
| CN105541683A (en) | Method for preparing tetrathioperoxydicarbamic acid | |
| HK1047105A1 (en) | Process for the production of dithiazolyl disulfides | |
| FR2468597A1 (en) | PROCESS FOR THE PREPARATION OF DITHIAZOLYLDISULFURES | |
| CN110330468B (en) | Synthetic method of 4, 4' -dithiodimorpholine | |
| CA1210011A (en) | Method of preparing dibenzothiazolyl disulfides | |
| JPS61115075A (en) | Manufacture of storable benzthiazole sulfenamides | |
| CN103102326A (en) | Preparation method of rubber accelerator NOBS (N-(oxidiethylene)-2-benzothiazolyl sulfenamide) | |
| CN114163383A (en) | Green production process of nicosulfuron intermediate nicotinamide and sulfamide | |
| CN112250600A (en) | Process method for improving yield of N, N' -diisopropylcarbodiimide product | |
| EP2003116A2 (en) | Process for the production of organic oxides | |
| CN118388378B (en) | A preparation method of 1,4-butanedisulfonyl chloride | |
| US4233461A (en) | Process for preparing colorless methionine | |
| JP7323876B2 (en) | Method for producing clopidogrel sulfate type I crystals | |
| CN108727297A (en) | A kind of hydrogen peroxide oxidation one-step synthesis technique of rubber accelerator dibenzothiazyl disulfide | |
| US2851391A (en) | Processes and products | |
| KR920001765B1 (en) | Method for preparing benzothiazole sulfenamide | |
| JPH0469347A (en) | Method for purifying ethylene compound having fluorine-containing organic group | |
| JPS61229873A (en) | Manufacture of 2-mercaptobenzoxazole | |
| JP4551722B2 (en) | Method for producing pentabromothiophenol | |
| JP4747660B2 (en) | Method for producing dithiosulfate compound | |
| CN107954909A (en) | A kind of method that isothiocyanates is prepared using ethyl chloroformate | |
| RU2381066C1 (en) | Catalyst and method of oxidising sodium sulphide | |
| US5276206A (en) | Method of preparing 2-mercaptobenzothiazole and benzothiazole |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |