CN111574466A - A novel triazole-containing allyl benzoate compound and its preparation method and application - Google Patents
A novel triazole-containing allyl benzoate compound and its preparation method and application Download PDFInfo
- Publication number
- CN111574466A CN111574466A CN202010504490.4A CN202010504490A CN111574466A CN 111574466 A CN111574466 A CN 111574466A CN 202010504490 A CN202010504490 A CN 202010504490A CN 111574466 A CN111574466 A CN 111574466A
- Authority
- CN
- China
- Prior art keywords
- triazol
- triazole
- arh
- formula
- phenyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D249/00—Heterocyclic compounds containing five-membered rings having three nitrogen atoms as the only ring hetero atoms
- C07D249/02—Heterocyclic compounds containing five-membered rings having three nitrogen atoms as the only ring hetero atoms not condensed with other rings
- C07D249/08—1,2,4-Triazoles; Hydrogenated 1,2,4-triazoles
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/64—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with three nitrogen atoms as the only ring hetero atoms
- A01N43/647—Triazoles; Hydrogenated triazoles
- A01N43/653—1,2,4-Triazoles; Hydrogenated 1,2,4-triazoles
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Agronomy & Crop Science (AREA)
- Pest Control & Pesticides (AREA)
- Plant Pathology (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Dentistry (AREA)
- General Health & Medical Sciences (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Environmental Sciences (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Plural Heterocyclic Compounds (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
Abstract
Description
技术领域technical field
本发明涉及一种新型含三唑的烯丙基苯甲酸酯类化合物及其制备方法和应用。The invention relates to a novel triazole-containing allyl benzoate compound and a preparation method and application thereof.
背景技术Background technique
含氮杂环化合物具有广泛的生物活性,因此在农药创制过程中意义重大。三唑类杀菌剂属于有机杂环氮类化合物,是麦角甾醇合成抑制剂,对危害作物生长的多数真菌病害均有良好防治效果。近几十年以来,三唑类杀菌剂以其高效、广谱、低毒等优势占据了极大市场,由于多数三唑类杀菌剂同时还具有植物生长调节功能,所以三唑类抗真菌药物类型的研究备受关注。迄今为止,数万个三唑类化合物先后被合成发现,而其中1,2,4-三氮唑类化合物占据了大部分,其中已经商品化的品种达到40多种,例如三唑酮(triadimefon)、三唑醇(triadimenol)、丙环唑(propiconazole)、多效唑(Paclobutrazol)等等。随着三唑类杀菌剂在农业上的频繁使用,一些缺点也随之暴露出来,其单一的活性位点,由于频繁使用使得真菌的抗药性越来越强从而使其失去了高效性。Nitrogen-containing heterocyclic compounds have a wide range of biological activities and are therefore of great significance in the creation of pesticides. Triazole fungicides belong to organic heterocyclic nitrogen compounds, are ergosterol synthesis inhibitors, and have good control effects on most fungal diseases that harm crop growth. In recent decades, triazole fungicides have occupied a huge market with their advantages of high efficiency, broad spectrum and low toxicity. Because most triazole fungicides also have plant growth regulation functions, so triazole fungicides are antifungal. Research on drug types has received much attention. So far, tens of thousands of triazole compounds have been synthesized and discovered, of which 1,2,4-triazole compounds occupy most of them, of which more than 40 species have been commercialized, such as triadimefon (triadimefon). ), triadimenol, propiconazole, Paclobutrazol and the like. With the frequent use of triazole fungicides in agriculture, some disadvantages have also been exposed. Their single active site, due to frequent use, makes fungi more and more resistant to drugs and thus loses its high efficiency.
设计合成新型的三唑类杀菌剂对我们开发高效、低毒、低残留的新型农药具有重要意义。The design and synthesis of new triazole fungicides is of great significance for us to develop new pesticides with high efficiency, low toxicity and low residues.
发明内容SUMMARY OF THE INVENTION
针对现有技术存在的上述技术问题,本发明的目的在于提供一种新型含三唑的烯丙基苯甲酸酯类化合物及其制备方法和应用。本发明是基于烯效唑结构为母体,保持烯效唑的苯环、三唑环和碳碳双键不变,用氢与三氟甲基取代苯环上的氯,用苯环替代烯效唑的叔丁基,羟基上引入酯基、芳环合成酯类化合物以考察对化合物生物活性的影响,设计合成了三唑类系列化合物。In view of the above-mentioned technical problems existing in the prior art, the purpose of the present invention is to provide a novel triazole-containing allyl benzoate compound and its preparation method and application. The present invention is based on the uniconazole structure as the parent body, keeps the benzene ring, the triazole ring and the carbon-carbon double bond of the uniconazole unchanged, replaces the chlorine on the benzene ring with hydrogen and trifluoromethyl, and replaces the olefin effect with the benzene ring The tert-butyl group of azoles, ester groups and aromatic rings were introduced into the hydroxyl groups to synthesize ester compounds to investigate the influence on the biological activity of the compounds, and a series of triazole compounds were designed and synthesized.
所述的一种新型含三唑的烯丙基苯甲酸酯类化合物,其特征在于其结构式如式(Ⅰ)所示:Described a kind of novel allyl benzoate compound containing triazole is characterized in that its structural formula is shown in formula (I):
式(Ⅰ)中:取代基R1为苯基或取代苯基,所述取代苯基的苯环上的取代基数量为一个或多个,各个取代基各自独立地选自H、卤素、C1-C4烷基、甲氧基或硝基;取代基R2为氢或三氟甲基。In formula (I): the substituent R 1 is a phenyl group or a substituted phenyl group, the number of substituent groups on the benzene ring of the substituted phenyl group is one or more, and each substituent group is independently selected from H, halogen, C1 -C4 alkyl, methoxy or nitro; substituent R 2 is hydrogen or trifluoromethyl.
所述的一种新型含三唑的烯丙基苯甲酸酯类化合物,其特征在于式(Ⅰ)中R1为下列之一:苯基、2-甲基苯基、2-氯苯基、2,4-二氯苯基、3-甲氧基苯基、2,6-二氯苯基、2-氟苯基、3-氟苯基、4-甲基苯基、2,3-二氯苯基、4-叔丁基苯基、4-正丙基苯基、4-甲氧基苯基、3-甲基苯基、4-氟苯基、3-硝基苯基、3-氯苯基。Said novel triazole-containing allyl benzoate compound is characterized in that R in formula (I) is one of the following: phenyl, 2-methylphenyl, 2-chlorophenyl, 2,4-Dichlorophenyl, 3-methoxyphenyl, 2,6-dichlorophenyl, 2-fluorophenyl, 3-fluorophenyl, 4-methylphenyl, 2,3-dichlorophenyl Chlorophenyl, 4-tert-butylphenyl, 4-n-propylphenyl, 4-methoxyphenyl, 3-methylphenyl, 4-fluorophenyl, 3-nitrophenyl, 3- Chlorophenyl.
所述的一种新型含三唑的烯丙基苯甲酸酯类化合物的制备方法,其特征在于包括以下步骤:Described a kind of preparation method of novel triazole-containing allyl benzoate compound is characterized in that comprising the following steps:
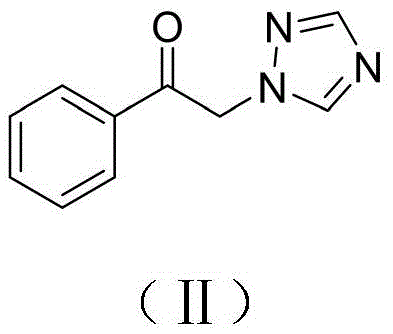
1)ɑ-溴代苯乙酮和1,2,4-三氮唑混合在有机溶剂A中,控温至0℃以下,滴加三乙胺后,于室温下搅拌反应,TLC跟踪反应进程;反应结束后抽滤除去反应生成的三乙胺盐酸盐,滤液经脱溶后,用有机溶剂B重结晶处理制得如式(Ⅱ)所示1-苯基-2-(1H-1,2,4-三唑-1-基)乙-1-酮;1) ɑ-Bromoacetophenone and 1,2,4-triazole are mixed in organic solvent A, the temperature is controlled to below 0°C, after triethylamine is added dropwise, the reaction is stirred at room temperature, and the reaction progress is tracked by TLC After the reaction is finished, suction filtration removes the triethylamine hydrochloride that the reaction generates, and after the filtrate is desolubilized, the 1-phenyl-2-(1H-1 shown in the formula (II) is obtained by recrystallization with the organic solvent B. ,2,4-triazol-1-yl)ethan-1-one;
2)将步骤1)所得式(Ⅱ)所示1-苯基-2-(1H-1,2,4-三唑-1-基)乙-1-酮、苯甲醛或对三氟甲基苯甲醛、哌啶在有机试剂C中加热回流反应,TLC跟踪反应进程;待反应结束后,冷却至室温,将反应液转移至分液漏斗内,水洗后分层,有机相再用饱和食盐水洗涤1-3次后,经无水硫酸钠干燥,过滤,滤液浓缩除去溶剂,浓缩物经柱层析分离提纯处理制得如式(Ⅲ)所示的(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)丙-2-烯-1-酮类化合物;2) 1-phenyl-2-(1H-1,2,4-triazol-1-yl)ethan-1-one, benzaldehyde or p-trifluoromethyl represented by formula (II) obtained in step 1) Benzaldehyde and piperidine are heated and refluxed in organic reagent C, and the reaction process is tracked by TLC; after the reaction is completed, cooled to room temperature, the reaction solution is transferred to a separatory funnel, washed with water and layered, and the organic phase is then saturated brine. After washing 1-3 times, it is dried over anhydrous sodium sulfate, filtered, the filtrate is concentrated to remove the solvent, and the concentrate is separated and purified by column chromatography to obtain (E)-1,3-diphenyl shown in formula (III). base-2-(1H-1,2,4-triazol-1-yl)prop-2-en-1-one compounds;
3)步骤2)所得式(Ⅲ)所示的(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)丙-2-烯-1-酮类化合物分散在有机试剂D中,加入PEG 600和硼氢化钠,在0℃冰浴条件下搅拌反应,TLC跟踪反应进程;待反应结束后,将反应液脱溶,脱溶浓缩物中滴加0.5~2N盐酸溶液,用有机试剂E萃取并合并有机相,随后经无水硫酸钠干燥,过滤,旋干溶剂处理制得如式(Ⅳ)所示的(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)丙-2-烯-1-醇类化合物;3) (E)-1,3-diphenyl-2-(1H-1,2,4-triazol-1-yl)prop-2-ene-1 represented by formula (III) obtained in step 2) -The ketone compound is dispersed in the organic reagent D, PEG 600 and sodium borohydride are added, the reaction is stirred under the condition of ice bath at 0°C, and the reaction progress is tracked by TLC; 0.5-2N hydrochloric acid solution was added dropwise, extracted with organic reagent E, and the organic phases were combined, then dried over anhydrous sodium sulfate, filtered, and spin-dried for solvent treatment to obtain (E)-1,3- as shown in formula (IV). Diphenyl-2-(1H-1,2,4-triazol-1-yl)prop-2-en-1-ol compounds;
4)将步骤3)所得如式(Ⅳ)所示的(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)丙-2-烯-1-醇类化合物分散在有机试剂F中,并加入DMAP、EDC和取代苯甲酸,室温下搅拌反应,TLC跟踪反应进程;待反应结束后,水洗,再用饱和食盐水洗涤,无水硫酸钠干燥,过滤,经柱层析分离提纯处理制得如式(Ⅰ)所示的一种新型含三唑的烯丙基苯甲酸酯类化合物;4) (E)-1,3-diphenyl-2-(1H-1,2,4-triazol-1-yl)prop-2-ene represented by formula (IV) obtained in step 3) The -1-alcohol compounds were dispersed in the organic reagent F, and DMAP, EDC and substituted benzoic acid were added, the reaction was stirred at room temperature, and the reaction progress was tracked by TLC; Sodium drying, filtration, separation and purification by column chromatography to obtain a novel triazole-containing allyl benzoate compound as shown in formula (I);
其中,所述R1为苯基或取代苯基,所述取代苯基的苯环上的取代基数量为一个或多个,各个取代基各自独立地选自H、卤素、C1-C4烷基、甲氧基或硝基;取代基R2为氢或三氟甲基。Wherein, the R 1 is phenyl or substituted phenyl, the number of substituents on the benzene ring of the substituted phenyl is one or more, and each substituent is independently selected from H, halogen, C1-C4 alkyl , methoxy or nitro; substituent R 2 is hydrogen or trifluoromethyl.
所述的一种新型含三唑的烯丙基苯甲酸酯类化合物的制备方法,其特征在于步骤1)中的有机溶剂A为酮类溶剂,优选为丙酮;有机溶剂B为醇类溶剂,优选为异丙醇;步骤2)中的有机溶剂C为苯类溶剂,优选为甲苯;步骤3)中的有机试剂D为醇类溶剂,优选为甲醇;步骤3)中的有机试剂E和步骤4)中的有机溶剂F均为二氯甲烷。The method for preparing a novel triazole-containing allyl benzoate compound is characterized in that the organic solvent A in step 1) is a ketone solvent, preferably acetone; the organic solvent B is an alcohol solvent, It is preferably isopropanol; the organic solvent C in the step 2) is a benzene solvent, preferably toluene; the organic reagent D in the step 3) is an alcohol solvent, preferably methanol; the organic reagent E in the step 3) and the step The organic solvent F in 4) is all methylene chloride.
所述的一种新型含三唑的烯丙基苯甲酸酯类化合物的制备方法,其特征在于步骤1)中,所述ɑ-溴代苯乙酮和1,2,4-三氮唑的投料摩尔比为1:1.1~1.5;步骤2)中,式(Ⅱ)所示的1-苯基-2-(1H-1,2,4-三唑-1-基)乙-1-酮和(对三氟甲基)苯甲醛的投料摩尔比为1:1.1~1.5;步骤3)中,式(Ⅲ)所示的(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)丙-2-烯-1-酮类化合物和硼氢化钠的投料摩尔比为1:1.1~1.5;步骤4)中,式(Ⅳ)所示的(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)丙-2-烯-1-醇类化合物和取代苯甲酸的投料摩尔比为1:1.1~1.5。The preparation method of described a kind of novel triazole-containing allyl benzoate compound is characterized in that in step 1), described ɑ-bromoacetophenone and 1,2,4-triazole are prepared. The feeding molar ratio is 1:1.1-1.5; in step 2), 1-phenyl-2-(1H-1,2,4-triazol-1-yl)ethan-1-one represented by formula (II) and the molar ratio of (p-trifluoromethyl)benzaldehyde is 1:1.1~1.5; in step 3), (E)-1,3-diphenyl-2-(1H- The molar ratio of 1,2,4-triazol-1-yl)prop-2-en-1-one compound and sodium borohydride is 1:1.1~1.5; in step 4), shown in formula (IV) The molar ratio of (E)-1,3-diphenyl-2-(1H-1,2,4-triazol-1-yl) prop-2-en-1-ol compound and substituted benzoic acid It is 1:1.1~1.5.
所述的一种新型含三唑的烯丙基苯甲酸酯类化合物的制备方法,其特征在于步骤1)中有机溶剂A的体积用量以ɑ-溴代苯乙酮的物质的量计为1~2mL/mmol,步骤2)中有机溶剂C的体积用量以式(Ⅱ)所示的1-苯基-2-(1H-1,2,4-三唑-1-基)乙-1-酮的物质的量计为1~1.5mL/mmol,步骤3)中有机溶剂D的体积用量以式(Ⅲ)所示的(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)丙-2-烯-1-酮类化合物的物质的量计为2~3mL/mmol,步骤4)中有机溶剂F的体积用量以式(Ⅳ)所示的(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)丙-2-烯-1-醇类化合物的物质的量计为6~8mL/mmol。The preparation method of described a kind of novel allyl benzoate compound containing triazole is characterized in that in step 1), the volumetric consumption of organic solvent A is counted as 1 with the amount of substance of ɑ-bromoacetophenone. ~2mL/mmol, the volume dosage of organic solvent C in step 2) is 1-phenyl-2-(1H-1,2,4-triazol-1-yl)b-1- The amount of the ketone substance is calculated as 1~1.5mL/mmol, and the volume dosage of the organic solvent D in step 3) is the (E)-1,3-diphenyl-2-(1H-1 represented by the formula (III) , 2,4-triazol-1-yl) prop-2-en-1-one compound substance is calculated as 2~3mL/mmol, and the volumetric amount of organic solvent F in step 4) is calculated as formula (IV) The amount of the (E)-1,3-diphenyl-2-(1H-1,2,4-triazol-1-yl)prop-2-en-1-ol compound shown is calculated as 6~8mL/mmol.
所述的一种新型含三唑的烯丙基苯甲酸酯类化合物的制备方法,其特征在于步骤2)和步骤4)中柱层析分离的洗脱剂均采用体积比1:0.5~2的乙酸乙酯和石油醚混合液。The method for preparing a novel triazole-containing allyl benzoate compound is characterized in that the eluents in step 2) and step 4) are separated by column chromatography in a volume ratio of 1:0.5 to 2 ethyl acetate and petroleum ether mixture.
所述的一种新型含三唑的烯丙基苯甲酸酯类化合物在制备杀菌剂中的应用。The application of the novel triazole-containing allyl benzoate compound in the preparation of fungicides.
进一步地,所述新型含三唑的烯丙基苯甲酸酯类化合物用于制备抑制油菜菌核病菌的杀菌剂。Further, the novel triazole-containing allyl benzoate compound is used to prepare a bactericide for inhibiting Sclerotinia sclerotiorum.
本发明新型含三唑的烯丙基苯甲酸酯类化合物的合成工艺路线如下:The synthetic route of the novel allyl benzoate compound containing triazole of the present invention is as follows:
与现有技术相比,本发明的有益效果主要体现在:本发明提供了一种新型含三唑的烯丙基苯甲酸酯类化合物及其制备方法与其制备杀菌剂中的应用,其制备方法简单、操作方便,得到的化合物在50ppm有效浓度下对油菜菌核病菌抑制活性最好,抑制率达到了71.4%,本发明所述化合物为具有杀菌活性的新化合物,为新农药的研发提供了基础。Compared with the prior art, the beneficial effects of the present invention are mainly reflected in: the present invention provides a novel triazole-containing allyl benzoate compound and its preparation method and its application in the preparation of fungicides, and its preparation method Simple and convenient to operate, the obtained compound has the best inhibitory activity against Sclerotinia sclerotiorum under the effective concentration of 50 ppm, and the inhibition rate reaches 71.4%. Base.
具体实施方式Detailed ways
下面结合具体实施例对本发明作进一步说明,但本发明的保护范围并不限于此。The present invention will be further described below with reference to specific embodiments, but the protection scope of the present invention is not limited thereto.
实施例1(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)烯丙基苯甲酸酯的制备Example 1 Preparation of (E)-1,3-diphenyl-2-(1H-1,2,4-triazol-1-yl)allyl benzoate
1)1-苯基-2-(1H-1,2,4-三唑-1-基)乙-1-酮(Ⅱ)的合成:1) Synthesis of 1-phenyl-2-(1H-1,2,4-triazol-1-yl)ethan-1-one (II):
在500mL三口烧瓶中依次加入ɑ-溴代苯乙酮(19.90g,0.10mol)、1,2,4-三氮唑(7.60g,0.11mol),加入150mL丙酮作溶剂,用冰盐浴控温在0℃以下,通过恒压滴液漏斗逐滴缓慢滴加三乙胺(11.13g,0.11mol)。滴加完毕后,继续反应1h,去冰浴,室温下继续搅拌1h,TLC(VEA/VPE=1/1,v/v)跟踪反应进程。待反应结束后,抽滤除去反应生成的三乙胺盐酸盐,三乙胺盐酸盐用丙酮淋洗,合并淋洗液和滤液后,经脱溶得到红色油状物,冷却固化,用20mL异丙醇重结晶制得式(Ⅱ)所示的1-苯基-2-(1H-1,2,4-三唑-1-基)乙-1-酮;Add ɑ-bromoacetophenone (19.90g, 0.10mol) and 1,2,4-triazole (7.60g, 0.11mol) to a 500mL three-necked flask in turn, add 150mL of acetone as a solvent, and control with an ice-salt bath. When the temperature was below 0°C, triethylamine (11.13 g, 0.11 mol) was slowly added dropwise through a constant pressure dropping funnel. After the dropwise addition, the reaction was continued for 1 h, the ice bath was removed, and the stirring was continued for 1 h at room temperature, and the reaction progress was followed by TLC ( VE A /V PE =1/1, v/v). After the reaction, the triethylamine hydrochloride generated by the reaction was removed by suction filtration, and the triethylamine hydrochloride was rinsed with acetone. After combining the eluent and the filtrate, a red oil was obtained by precipitation, which was cooled and solidified. 1-phenyl-2-(1H-1,2,4-triazol-1-yl)ethan-1-one represented by formula (II) was obtained by recrystallization from isopropanol;
2)(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)丙-2-烯-1-酮(Ⅲ-1)的合成:2) Synthesis of (E)-1,3-diphenyl-2-(1H-1,2,4-triazol-1-yl)prop-2-en-1-one (III-1):
在装有分水器、温度计、冷凝管的100mL三口烧瓶中一次加入式(Ⅱ)所示的1-苯基-2-(1H-1,2,4-三唑-1-基)乙-1-酮(5.62g,30.00mmol),苯甲醛(3.50g,33.00mmol),加入30mL甲苯作溶剂,滴加哌啶(0.20g,2.35mmol)作为催化剂,加热回流反应6h,TLC(VEA/VPE=1/1,v/v)跟踪反应进程。待反应结束后,冷却至室温,将反应液转移至分液漏斗内,水洗后分层,有机相再用饱和食盐水洗涤两次(每次洗涤采用的饱和食盐水体积20mL),无水硫酸钠干燥,过滤,滤液浓缩除去溶剂,浓缩残留物经柱层析分离提纯(洗脱剂为体积比1:1石油醚和乙酸乙酯)制得式(Ⅲ-1)所示的(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)丙-2-烯-1-酮;1-phenyl-2-(1H-1,2,4-triazol-1-yl)ethyl- 1-ketone (5.62g, 30.00mmol), benzaldehyde (3.50g, 33.00mmol), add 30mL of toluene as a solvent, dropwise add piperidine (0.20g, 2.35mmol) as a catalyst, heat under reflux for 6h, TLC (V EA /V PE = 1/1, v/v) to track the progress of the reaction. After the reaction was completed, it was cooled to room temperature, and the reaction solution was transferred to a separatory funnel. After washing, the layers were separated. The organic phase was washed twice with saturated brine (the volume of saturated brine used for each washing was 20 mL), and anhydrous sulfuric acid Dry over sodium, filter, concentrate the filtrate to remove the solvent, and separate and purify the concentrated residue by column chromatography (eluent is 1:1 petroleum ether and ethyl acetate by volume) to obtain (E) represented by formula (III-1) -1,3-diphenyl-2-(1H-1,2,4-triazol-1-yl)prop-2-en-1-one;
3)(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)丙-2-烯-1-醇(Ⅳ-1)的合成:3) Synthesis of (E)-1,3-diphenyl-2-(1H-1,2,4-triazol-1-yl)prop-2-en-1-ol (IV-1):
将式(Ⅲ-1)所示的(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)丙-2-烯-1-酮(1.93g,7.00mmol)加入到50mL圆底烧瓶中,加入15mL甲醇作溶剂,冰浴控温在0℃,加入PEG600(0.50g,0.83mmol),然后在搅拌下分批加入硼氢化钠(0.42g,11.00mmol),在0℃冰浴下反应4h,TLC(VEA/VPE=1/2,v/v)跟踪反应进程。待反应结束后,将反应液脱溶,脱溶浓缩物中滴加1N盐酸溶液,用二氯甲烷萃取三次(每次萃取用二氯甲烷15mL),合并有机相,随后经无水硫酸钠干燥,过滤,旋干溶剂制得式(Ⅳ-1)所示的(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)丙-2-烯-1-醇;(E)-1,3-diphenyl-2-(1H-1,2,4-triazol-1-yl)prop-2-en-1-one ( 1.93g, 7.00mmol) was added to a 50mL round-bottomed flask, 15mL of methanol was added as a solvent, the temperature was controlled at 0°C in an ice bath, PEG600 (0.50g, 0.83mmol) was added, and sodium borohydride (0.42 mmol) was added in batches under stirring. g, 11.00 mmol), reacted at 0°C under ice bath for 4 h, TLC (V EA /V PE =1/2, v/v) followed the reaction progress. After the reaction was completed, the reaction solution was desolubilized, 1N hydrochloric acid solution was added dropwise to the precipitation concentrate, extracted three times with dichloromethane (15 mL of dichloromethane was used for each extraction), the organic phases were combined, and then dried over anhydrous sodium sulfate. , filtered, and spin-dried to obtain (E)-1,3-diphenyl-2-(1H-1,2,4-triazol-1-yl)propan-2 represented by formula (IV-1) -en-1-ol;
4)(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)烯丙基苯甲酸酯(C1)的合成:4) Synthesis of (E)-1,3-diphenyl-2-(1H-1,2,4-triazol-1-yl)allylbenzoate (C1):
将式(Ⅳ-1)所示的(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)丙-2-烯-1-醇(0.20g,0.72mmol)、苯甲酸(0.10g,0.80mmol)、DMAP(0.09g,0.72mmol)、EDC(0.28g,1.44mmol)加入到5mL二氯甲烷中,室温下反应4h,随后将反应液转移至分液漏斗内,经水洗(3×5mL),再用饱和食盐水洗涤(2×5mL),无水硫酸钠干燥,过滤,滤液浓缩除去溶剂,浓缩残留物经柱层析分离提纯(洗脱剂为体积比1:1石油醚和乙酸乙酯)制得(C1)化合物。(E)-1,3-diphenyl-2-(1H-1,2,4-triazol-1-yl)prop-2-en-1-ol ( 0.20g, 0.72mmol), benzoic acid (0.10g, 0.80mmol), DMAP (0.09g, 0.72mmol), EDC (0.28g, 1.44mmol) were added to 5mL of dichloromethane, and reacted at room temperature for 4h, then the reaction The liquid was transferred to a separatory funnel, washed with water (3×5 mL), washed with saturated brine (2×5 mL), dried over anhydrous sodium sulfate, filtered, the filtrate was concentrated to remove the solvent, and the concentrated residue was separated and purified by column chromatography (The eluent is 1:1 petroleum ether and ethyl acetate by volume) to prepare compound (C1).
其中式(Ⅲ)和式(Ⅳ)结构式中的取代基R2均为氢,分别形成了式(Ⅲ-1)和式(Ⅳ-1)的分子结构。Wherein, the substituents R 2 in the structural formulae of formula (III) and formula (IV) are all hydrogen, respectively forming the molecular structures of formula (III-1) and formula (IV-1).
(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)烯丙基苯甲酸酯:黄色油状物,收率52.36%;1H NMR(500MHz,CDCl3)δ:8.08(s,1H,triazole-H),7.68(s,1H,triazole-H),7.60-7.56(m,1H,ArH),7.48-7.42(m,5H,ArH),7.38-7.33(m,4H,ArH),7.21-7.17(m,3H,ArH),7.03(s,1H,CH),6.95(s,1H,=CH),6.84-6.79(m,2H,ArH).HRMS(ESI)for C24H19N3O2m/z:Calculated,382.1550,Found,382.1556[M+H]+.(E)-1,3-diphenyl-2-(1H-1,2,4-triazol-1-yl)allylbenzoate: yellow oil, yield 52.36%; 1 H NMR (500MHz, CDCl 3 )δ: 8.08(s,1H,triazole-H), 7.68(s,1H,triazole-H), 7.60-7.56(m,1H,ArH), 7.48-7.42(m,5H,ArH) ),7.38-7.33(m,4H,ArH),7.21-7.17(m,3H,ArH),7.03(s,1H,CH),6.95(s,1H,=CH),6.84-6.79(m,2H ,ArH).HRMS(ESI)for C 24 H 19 N 3 O 2 m/z:Calculated,382.1550,Found,382.1556[M+H] + .
实施例2(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)烯丙基2-甲基苯甲酸酯(C2)的制备Example 2 (E) Preparation of 1,3-diphenyl-2-(1H-1,2,4-triazol-1-yl)allyl 2-methylbenzoate (C2)
将实施例1步骤4)中的苯甲酸替换为同等摩尔量的2-甲基苯甲酸,其他操作同实施例1,制得(C2)化合物。Substitute the benzoic acid in step 4) of Example 1 with 2-methylbenzoic acid in the same molar amount, and other operations are the same as in Example 1, to prepare compound (C2).
(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)烯丙基2-甲基苯甲酸酯:黄色油状物,收率42.62%;1H NMR(500MHz,CDCl3)δ:8.09(s,1H,triazole-H),7.98(d,J=7.8Hz,1H,ArH),7.66(s,1H,triazole-H),7.45-7.42(m,2H,ArH),7.41-7.31(m,4H,ArH),7.27-7.21(m,2H,ArH),7.20-7.15(m,3H,ArH),7.02(s,1H,CH),6.93(s,1H,=CH),6.82-6.79(m,2H,ArH),2.56(s,3H,CH3).HRMS(ESI)for C25H21N3O2 m/z:Calculated,396.1707,Found,396.1712[M+H]+.(E)-1,3-diphenyl-2-(1H-1,2,4-triazol-1-yl)allyl 2-methylbenzoate: yellow oil, yield 42.62% ; 1 H NMR (500MHz, CDCl 3 )δ: 8.09(s, 1H, triazole-H), 7.98(d, J=7.8Hz, 1H, ArH), 7.66(s, 1H, triazole-H), 7.45- 7.42(m,2H,ArH),7.41-7.31(m,4H,ArH),7.27-7.21(m,2H,ArH),7.20-7.15(m,3H,ArH),7.02(s,1H,CH) ,6.93(s,1H,=CH),6.82-6.79(m,2H,ArH),2.56(s,3H,CH 3 ).HRMS(ESI)for C 25 H 21 N 3 O 2 m/z:Calculated ,396.1707,Found,396.1712[M+H] + .
实施例3(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)烯丙基2-氯苯甲酸酯(C3)的制备Example 3 Preparation of (E)-1,3-diphenyl-2-(1H-1,2,4-triazol-1-yl)allyl 2-chlorobenzoate (C3)
将实施例1步骤4)中的苯甲酸替换为同等摩尔量的2-氯苯甲酸,其他操作同实施例1,制得(C3)化合物。Substitute the benzoic acid in step 4) of Example 1 with 2-chlorobenzoic acid in the same molar amount, and other operations are the same as those in Example 1 to prepare compound (C3).
(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)烯丙基2-氯苯甲酸酯:黄色油状物,收率60.21%;1H NMR(500MHz,CDCl3)δ:8.09(s,1H,triazole-H),7.87(dd,J=7.8,1.3Hz,1H,ArH),7.67(s,1H,triazole-H),7.48-7.41(m,4H,ArH),7.38-7.31(m,4H,ArH),7.23-7.17(m,3H,ArH),7.03(s,1H,CH),6.98(s,1H,=CH),6.84-6.77(m,2H,ArH).HRMS(ESI)forC24H18ClN3O2m/z:Calculated,416.1160,Found,416.1166[M+H]+.(E)-1,3-diphenyl-2-(1H-1,2,4-triazol-1-yl)allyl 2-chlorobenzoate: yellow oil, yield 60.21%; 1 H NMR (500MHz, CDCl 3 )δ: 8.09(s,1H,triazole-H),7.87(dd,J=7.8,1.3Hz,1H,ArH),7.67(s,1H,triazole-H),7.48 -7.41(m,4H,ArH),7.38-7.31(m,4H,ArH),7.23-7.17(m,3H,ArH),7.03(s,1H,CH),6.98(s,1H,=CH) ,6.84-6.77(m,2H,ArH).HRMS(ESI)forC 24 H 18 ClN 3 O 2 m/z: Calculated,416.1160,Found,416.1166[M+H] + .
实施例4(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)烯丙基2,4-二氯苯甲酸酯(C4)的制备Example 4 (E) of 1,3-diphenyl-2-(1H-1,2,4-triazol-1-yl)allyl 2,4-dichlorobenzoate (C4) preparation
将实施例1步骤4)中的苯甲酸替换为同等摩尔量的2,4-二氯苯甲酸,其他操作同实施例1,制得(C4)化合物。Substitute the benzoic acid in step 4) of Example 1 with 2,4-dichlorobenzoic acid in the same molar amount, and other operations are the same as those in Example 1, to prepare compound (C4).
(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)烯丙基2,4-二氯苯甲酸酯:白色油状物,收率42.17%;1H NMR(500MHz,CDCl3)δ:8.09(s,1H,triazole-H),7.85(d,J=8.5Hz,1H,ArH),7.64(s,1H,triazole-H),7.48(d,J=2.0Hz,1H,ArH),7.44-7.40(m,2H,ArH),7.39-7.33(m,3H,ArH),7.32(dd,J=8.5,2.0Hz,1H,ArH),7.23-7.17(m,3H,ArH),7.02(s,1H,CH),6.96(s,1H,=CH),6.84-6.77(m,2H,ArH).HRMS(ESI)for C24H17Cl2N3O2 m/z:Calculated,450.0771,Found,450.0776[M+H]+.(E)-1,3-Diphenyl-2-(1H-1,2,4-triazol-1-yl)allyl 2,4-dichlorobenzoate: white oil, yield 42.17%; 1 H NMR (500MHz, CDCl 3 )δ: 8.09(s,1H,triazole-H),7.85(d,J=8.5Hz,1H,ArH),7.64(s,1H,triazole-H), 7.48 (d, J=2.0Hz, 1H, ArH), 7.44-7.40 (m, 2H, ArH), 7.39-7.33 (m, 3H, ArH), 7.32 (dd, J=8.5, 2.0Hz, 1H, ArH) ),7.23-7.17(m,3H,ArH),7.02(s,1H,CH),6.96(s,1H,=CH),6.84-6.77(m,2H,ArH).HRMS(ESI)for C 24 H 17 Cl 2 N 3 O 2 m/z: Calculated, 450.0771, Found, 450.0776[M+H] + .
实施例5(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)烯丙基3-甲氧基苯甲酸酯(C5)的制备Example 5 Preparation of (E)-1,3-diphenyl-2-(1H-1,2,4-triazol-1-yl)allyl 3-methoxybenzoate (C5)
将实施例1步骤4)中的苯甲酸替换为同等摩尔量的3-甲氧基苯甲酸,其他操作同实施例1,制得(C5)化合物。Substitute the benzoic acid in step 4) of Example 1 with 3-methoxybenzoic acid in the same molar amount, and other operations are the same as those in Example 1, to prepare compound (C5).
(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)烯丙基3-甲氧基苯甲酸酯:白色蜡状物,收率25.35%;1H NMR(500MHz,CDCl3)δ:8.07(s,1H,triazole-H),8.06-8.02(m,2H,ArH),7.69(s,1H,triazole-H),7.44-7.40(m,2H,ArH),7.37-7.31(m,3H,ArH),7.21-7.16(m,3H,ArH),7.00(s,1H,CH),6.95(s,1H,=CH),6.94-6.91(m,2H,ArH),6.83-6.79(m,2H,ArH),3.83(s,3H,OCH3).HRMS(ESI)for C25H21N3O3 m/z:Calculated,412.1656,Found,412.1661[M+H]+.(E)-1,3-Diphenyl-2-(1H-1,2,4-triazol-1-yl)allyl 3-methoxybenzoate: white wax, yield 25.35%; 1 H NMR (500 MHz, CDCl 3 ) δ: 8.07 (s, 1H, triazole-H), 8.06-8.02 (m, 2H, ArH), 7.69 (s, 1H, triazole-H), 7.44-7.40 (m, 2H, ArH), 7.37-7.31 (m, 3H, ArH), 7.21-7.16 (m, 3H, ArH), 7.00 (s, 1H, CH), 6.95 (s, 1H, =CH), 6.94 -6.91(m,2H,ArH),6.83-6.79(m,2H,ArH),3.83(s, 3H , OCH3 ) .HRMS (ESI)for C25H21N3O3 m /z:Calculated, 412.1656,Found,412.1661[M+H] + .
实施例6(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)烯丙基2,6-二氯苯甲酸酯(C6)的制备Example 6 (E) of 1,3-diphenyl-2-(1H-1,2,4-triazol-1-yl)allyl 2,6-dichlorobenzoate (C6) preparation
将实施例1步骤4)中的苯甲酸替换为同等摩尔量的2,6-二氯苯甲酸,其他操作同实施例1,制得(C6)化合物。Substitute the benzoic acid in step 4) of Example 1 with 2,6-dichlorobenzoic acid in the same molar amount, and other operations are the same as those in Example 1, to prepare compound (C6).
(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)烯丙基2,6-二氯苯甲酸酯:黄色油状物,收率47.66%;1H NMR(500MHz,CDCl3)δ:8.08(s,1H,triazole-H),7.78(s,1H,triazole-H),7.52-7.49(m,2H,ArH),7.46-7.41(m,1H,ArH),7.39-7.35(m,3H,ArH),7.30-7.25(m,2H,ArH),7.21-7.17(m,3H,ArH),7.12(s,1H,CH),6.87(s,1H,=CH),6.82-6.78(m,2H,ArH).HRMS(ESI)for C24H17Cl2N3O2 m/z:Calculated,450.0771,Found,450.0776[M+H]+.(E)-1,3-Diphenyl-2-(1H-1,2,4-triazol-1-yl)allyl 2,6-dichlorobenzoate: yellow oil, yield 47.66%; 1 H NMR (500 MHz, CDCl 3 ) δ: 8.08(s,1H,triazole-H),7.78(s,1H,triazole-H),7.52-7.49(m,2H,ArH),7.46-7.41 (m,1H,ArH),7.39-7.35(m,3H,ArH),7.30-7.25(m,2H,ArH),7.21-7.17(m,3H,ArH),7.12(s,1H,CH), 6.87(s,1H,=CH),6.82-6.78(m,2H,ArH).HRMS(ESI)for C 24 H 17 Cl 2 N 3 O 2 m/z: Calculated,450.0771,Found,450.0776[M+ H] + .
实施例7(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)烯丙基2-氟苯甲酸酯(C7)的制备Example 7 Preparation of (E)-1,3-diphenyl-2-(1H-1,2,4-triazol-1-yl)allyl 2-fluorobenzoate (C7)
将实施例1步骤4)中的苯甲酸替换为同等摩尔量的2-氟苯甲酸,其他操作同实施例1,制得(C7)化合物。Substitute the benzoic acid in step 4) of Example 1 with 2-fluorobenzoic acid in an equivalent molar amount, and other operations are the same as those in Example 1, to prepare compound (C7).
(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)烯丙基2-氟苯甲酸酯:黄色油状物,收率56.35%;1HNMR(500MHz,CDCl3)δ:8.07(s,1H,triazole-H),8.00-7.92(m,1H,ArH),7.72(s,1H,triazole-H),7.55-7.47(m,1H,ArH),7.46-7.40(m,2H,ArH),7.38-7.29(m,3H,ArH),7.22-7.10(m,5H,ArH),7.04(s,1H,CH),7.02(s,1H,=CH),6.86-6.78(m,2H,ArH).HRMS(ESI)for C24H18FN3O2 m/z:Calculated,400.1456,Found,400.1461[M+H]+.(E)-1,3-diphenyl-2-(1H-1,2,4-triazol-1-yl)allyl 2-fluorobenzoate: yellow oil, yield 56.35%; 1 HNMR (500MHz, CDCl 3 )δ: 8.07(s,1H,triazole-H),8.00-7.92(m,1H,ArH),7.72(s,1H,triazole-H),7.55-7.47(m,1H) ,ArH),7.46-7.40(m,2H,ArH),7.38-7.29(m,3H,ArH),7.22-7.10(m,5H,ArH),7.04(s,1H,CH),7.02(s, 1H,=CH),6.86-6.78(m,2H,ArH).HRMS(ESI)for C 24 H 18 FN 3 O 2 m/z:Calculated,400.1456,Found,400.1461[M+H] + .
实施例8(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)烯丙基3-氟苯甲酸酯(C8)的制备Example 8 Preparation of (E)-1,3-diphenyl-2-(1H-1,2,4-triazol-1-yl)allyl 3-fluorobenzoate (C8)
将实施例1步骤4)中的苯甲酸替换为同等摩尔量的3-氟苯甲酸,其他操作同实施例1,制得(C8)化合物。Substitute the benzoic acid in step 4) of Example 1 with 3-fluorobenzoic acid in an equivalent molar amount, and other operations are the same as those in Example 1, to prepare compound (C8).
(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)烯丙基3-氟苯甲酸酯:黄色油状物,收率58.34%;1H NMR(500MHz,CDCl3)δ:8.09(s,1H,triazole-H),7.90-7.85(m,1H,ArH),7.79-7.74(m,1H,ArH),7.65(s,1H,triazole-H),7.46-7.40(m,3H,ArH),7.39-7.33(m,3H,ArH),7.31-7.26(m,1H,ArH),7.23-7.17(m,3H,ArH),7.02(s,1H,CH),6.94(s,1H,=CH),6.88-6.78(m,2H,ArH).HRMS(ESI)for C24H18FN3O2 m/z:Calculated,400.1456,Found,400.1461[M+H]+.(E)-1,3-diphenyl-2-(1H-1,2,4-triazol-1-yl)allyl 3-fluorobenzoate: yellow oil, yield 58.34%; 1 H NMR (500MHz, CDCl 3 )δ:8.09(s,1H,triazole-H),7.90-7.85(m,1H,ArH),7.79-7.74(m,1H,ArH),7.65(s,1H, triazole-H),7.46-7.40(m,3H,ArH),7.39-7.33(m,3H,ArH),7.31-7.26(m,1H,ArH),7.23-7.17(m,3H,ArH),7.02 (s,1H,CH),6.94(s,1H,=CH),6.88-6.78(m,2H,ArH).HRMS(ESI) for C 24 H 18 FN 3 O 2 m/z: Calculated, 400.1456, Found,400.1461[M+H] + .
实施例9(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)烯丙基4-甲基苯甲酸酯(C9)的制备Example 9 Preparation of (E)-1,3-diphenyl-2-(1H-1,2,4-triazol-1-yl)allyl 4-methylbenzoate (C9)
将实施例1步骤4)中的苯甲酸替换为同等摩尔量的4-甲基苯甲酸,其他操作同实施例1,制得(C9)化合物。Substitute the benzoic acid in step 4) of Example 1 with 4-methylbenzoic acid in the same molar amount, and other operations are the same as in Example 1, to prepare compound (C9).
(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)烯丙基4-甲基苯甲酸酯:白色油状物,收率44.72%;1H NMR(500MHz,CDCl3)δ:8.07(s,1H,triazole-H),7.97(d,J=8.2Hz,2H,ArH),7.69(s,1H,triazole-H),7.45-7.40(m,3H,ArH),7.36-7.32(m,4H,ArH),7.22-7.16(m,3H,ArH),7.01(s,1H,CH),6.95(s,1H,=CH),6.84-6.78(m,2H,ArH),2.40(s,3H,CH3).HRMS(ESI)for C25H21N3O2 m/z:Calculated,396.1707,Found,396.1712[M+H]+.(E)-1,3-diphenyl-2-(1H-1,2,4-triazol-1-yl)allyl 4-methylbenzoate: white oil, yield 44.72% ; 1 H NMR (500MHz, CDCl 3 )δ: 8.07(s,1H,triazole-H),7.97(d,J=8.2Hz,2H,ArH),7.69(s,1H,triazole-H),7.45- 7.40(m,3H,ArH),7.36-7.32(m,4H,ArH),7.22-7.16(m,3H,ArH),7.01(s,1H,CH),6.95(s,1H,=CH), 6.84-6.78(m, 2H, ArH), 2.40(s, 3H, CH 3 ).HRMS(ESI) for C 25 H 21 N 3 O 2 m/z: Calculated, 396.1707, Found, 396.1712[M+H] + .
实施例10(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)烯丙基2,3-二氯苯甲酸酯(C10)的制备Example 10 (E)-1,3-diphenyl-2-(1H-1,2,4-triazol-1-yl)allyl 2,3-dichlorobenzoate (C10) preparation
将实施例1步骤4)中的苯甲酸替换为同等摩尔量的2,3-二氯苯甲酸,其他操作同实施例1,制得(C10)化合物。Substitute the benzoic acid in step 4) of Example 1 with 2,3-dichlorobenzoic acid in the same molar amount, and other operations are the same as those in Example 1, to prepare compound (C10).
(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)烯丙基2,3-二氯苯甲酸酯:黄色油状物,收率30.33%;1H NMR(500MHz,CDCl3)δ:8.08(s,1H,triazole-H),8.03(d,J=8.3Hz,2H,ArH),7.71(s,1H,triazole-H),7.50-7.45(m,3H,ArH),7.35-7.31(m,3H,ArH),7.21-7.15(m,3H,ArH),7.02(s,1H,CH),6.95(s,1H,=CH),6.84-6.78(m,2H,ArH).HRMS(ESI)forC24H17Cl2N3O2 m/z:Calculated,450.0771,Found,450.0776[M+H]+.(E)-1,3-Diphenyl-2-(1H-1,2,4-triazol-1-yl)allyl 2,3-dichlorobenzoate: yellow oil, yield 30.33%; 1 H NMR (500MHz, CDCl 3 )δ: 8.08(s,1H,triazole-H),8.03(d,J=8.3Hz,2H,ArH),7.71(s,1H,triazole-H), 7.50-7.45(m, 3H, ArH), 7.35-7.31(m, 3H, ArH), 7.21-7.15(m, 3H, ArH), 7.02(s, 1H, CH), 6.95(s, 1H, =CH) ),6.84-6.78(m,2H,ArH).HRMS(ESI)forC 24 H 17 Cl 2 N 3 O 2 m/z:Calculated,450.0771,Found,450.0776[M+H] + .
实施例11(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)烯丙基4-叔丁基苯甲酸酯(C11)的制备Example 11 Preparation of (E)-1,3-diphenyl-2-(1H-1,2,4-triazol-1-yl)allyl 4-tert-butylbenzoate (C11)
将实施例1步骤4)中的苯甲酸替换为同等摩尔量的4-叔丁基苯甲酸,其他操作同实施例1,制得(C11)化合物。Substitute the benzoic acid in step 4) of Example 1 with 4-tert-butylbenzoic acid in the same molar amount, and other operations are the same as those in Example 1 to prepare compound (C11).
(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)烯丙基4-叔丁基苯甲酸酯:白色蜡状物,收率44.20%;1H NMR(500MHz,CDCl3)δ:8.10(s,1H,triazole-H),7.67(dd,J=7.8,1.5Hz,1H,ArH),7.64(s,1H,triazole-H),7.62(dd,J=8.0,1.5Hz,1H,ArH),7.44-7.41(m,2H,ArH),7.41-7.32(m,4H,ArH),7.29(d,J=7.9Hz,1H,ArH),7.24-7.20(m,3H,ArH),7.02(s,1H,CH),6.94(s,1H,=CH),6.83-6.80(m,2H,ArH),1.56(s,9H,t-Bu).HRMS(ESI)forC28H27N3O2 m/z:Calculated,438.2176,Found,438.2182[M+H]+.(E)-1,3-Diphenyl-2-(1H-1,2,4-triazol-1-yl)allyl 4-tert-butylbenzoate: white wax, yield 44.20%; 1 H NMR (500 MHz, CDCl 3 ) δ: 8.10 (s, 1H, triazole-H), 7.67 (dd, J=7.8, 1.5 Hz, 1H, ArH), 7.64 (s, 1H, triazole-H) ),7.62(dd,J=8.0,1.5Hz,1H,ArH),7.44-7.41(m,2H,ArH),7.41-7.32(m,4H,ArH),7.29(d,J=7.9Hz,1H ,ArH),7.24-7.20(m,3H,ArH),7.02(s,1H,CH),6.94(s,1H,=CH),6.83-6.80(m,2H,ArH),1.56(s,9H ,t-Bu).HRMS(ESI)forC 28 H 27 N 3 O 2 m/z: Calculated,438.2176,Found,438.2182[M+H] + .
实施例12(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)烯丙基4-正丙基苯甲酸酯(C12)的制备Example 12 Preparation of (E)-1,3-diphenyl-2-(1H-1,2,4-triazol-1-yl)allyl 4-n-propylbenzoate (C12)
将实施例1步骤4)中的苯甲酸替换为同等摩尔量的4-正丙基苯甲酸,其他操作同实施例1,制得(C12)化合物。Substitute the benzoic acid in step 4) of Example 1 with 4-n-propylbenzoic acid in the same molar amount, and other operations are the same as those in Example 1, to prepare compound (C12).
(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)烯丙基4-正丙基苯甲酸酯:白色蜡状物,收率48.96%;1H NMR(500MHz,CDCl3)δ:8.07(s,1H,triazole-H),8.00(d,J=8.2Hz,2H,ArH),7.69(s,1H,triazole-H),7.45-7.41(m,2H,ArH),7.36-7.31(m,3H,ArH),7.26(d,J=8.4Hz,2H,ArH),7.22-7.17(m,3H,ArH),7.01(s,1H,CH),6.95(s,1H,=CH),6.81(dd,J=7.8,1.4Hz,2H,ArH),2.65(t,J=7.5Hz,2H,CH2),1.71-1.59(m,2H,CH2),0.94(t,J=7.3Hz,3H,CH3).HRMS(ESI)for C27H25N3O2 m/z:Calculated,424.2020,Found,424.2025[M+H]+.(E)-1,3-Diphenyl-2-(1H-1,2,4-triazol-1-yl)allyl 4-n-propylbenzoate: white wax, yield 48.96%; 1 H NMR (500MHz, CDCl 3 )δ: 8.07(s,1H,triazole-H),8.00(d,J=8.2Hz,2H,ArH),7.69(s,1H,triazole-H), 7.45-7.41(m,2H,ArH),7.36-7.31(m,3H,ArH),7.26(d,J=8.4Hz,2H,ArH),7.22-7.17(m,3H,ArH),7.01(s ,1H,CH),6.95(s,1H,=CH),6.81(dd,J=7.8,1.4Hz,2H,ArH),2.65(t,J=7.5Hz,2H,CH 2 ),1.71-1.59 (m, 2H, CH 2 ), 0.94 (t, J=7.3 Hz, 3H, CH 3 ). HRMS (ESI) for C 27 H 25 N 3 O 2 m/z: Calculated, 424.2020, Found, 424.2025 [M +H] + .
实施例13(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)烯丙基4-甲氧基苯甲酸酯(C13)的制备Example 13 Preparation of (E)-1,3-diphenyl-2-(1H-1,2,4-triazol-1-yl)allyl 4-methoxybenzoate (C13)
将实施例1步骤4)中的苯甲酸替换为同等摩尔量的4-甲氧基苯甲酸,其他操作同实施例1,制得(C13)化合物。Substitute the benzoic acid in step 4) of Example 1 with 4-methoxybenzoic acid in the same molar amount, and other operations are the same as those in Example 1, to prepare compound (C13).
(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)烯丙基4-甲氧基苯甲酸酯:白色蜡状物,收率27.43%;1H NMR(500MHz,CDCl3)δ:8.07(s,1H,triazole-H),8.06-8.02(m,2H,ArH),7.69(s,1H,triazole-H),7.42(d,J=7.0Hz,2H,ArH),7.36-7.29(m,3H,ArH),7.20-7.15(m,3H,ArH),7.00(s,1H,CH),6.95(s,1H,=CH),6.92(d,J=8.9Hz,2H,ArH),6.83-6.79(m,2H,ArH),3.82(s,3H,OCH3).HRMS(ESI)for C25H21N3O3 m/z:Calculated,412.1656,Found,412.1661[M+H]+.(E)-1,3-Diphenyl-2-(1H-1,2,4-triazol-1-yl)allyl 4-methoxybenzoate: white wax, yield 27.43%; 1 H NMR (500 MHz, CDCl 3 ) δ: 8.07(s,1H,triazole-H),8.06-8.02(m,2H,ArH),7.69(s,1H,triazole-H),7.42(d ,J=7.0Hz,2H,ArH),7.36-7.29(m,3H,ArH),7.20-7.15(m,3H,ArH),7.00(s,1H,CH),6.95(s,1H,=CH ), 6.92(d, J=8.9Hz, 2H, ArH), 6.83-6.79(m, 2H, ArH), 3.82(s, 3H, OCH 3 ). HRMS(ESI) for C 25 H 21 N 3 O 3 m/z: Calculated,412.1656,Found,412.1661[M+H] + .
实施例14(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)烯丙基3-甲基苯甲酸酯(C14)的制备Example 14 Preparation of (E)-1,3-diphenyl-2-(1H-1,2,4-triazol-1-yl)allyl 3-methylbenzoate (C14)
将实施例1步骤4)中的苯甲酸替换为同等摩尔量的3-甲基苯甲酸,其他操作同实施例1,制得(C14)化合物。Substitute the benzoic acid in step 4) of Example 1 with 3-methylbenzoic acid in the same molar amount, and other operations are the same as those in Example 1, to prepare compound (C14).
(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)烯丙基3-甲基苯甲酸酯:白色油状物,收率55.64%;1H NMR(500MHz,CDCl3)δ:8.07(s,1H,triazole-H),7.89(d,J=7.6Hz,2H,ArH),7.70(s,1H,triazole-H),7.46-7.41(m,2H,ArH),7.36-7.30(m,5H,ArH),7.20-7.15(m,3H,ArH),7.03(s,1H,CH),6.95(s,1H,=CH),6.83-6.78(m,2H,ArH),2.38(s,3H,CH3).HRMS(ESI)for C25H21N3O2 m/z:Calculated,396.1707,Found,396.1712[M+H]+.(E)-1,3-diphenyl-2-(1H-1,2,4-triazol-1-yl)allyl 3-methylbenzoate: white oil, yield 55.64% ; 1 H NMR (500MHz, CDCl 3 )δ: 8.07(s, 1H, triazole-H), 7.89(d, J=7.6Hz, 2H, ArH), 7.70(s, 1H, triazole-H), 7.46- 7.41(m, 2H, ArH), 7.36-7.30(m, 5H, ArH), 7.20-7.15(m, 3H, ArH), 7.03(s, 1H, CH), 6.95(s, 1H, =CH), 6.83-6.78(m, 2H, ArH), 2.38(s, 3H, CH 3 ).HRMS(ESI) for C 25 H 21 N 3 O 2 m/z: Calculated, 396.1707, Found, 396.1712[M+H] + .
实施例15(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)烯丙基4-氟苯甲酸酯(C15)的制备Example 15 Preparation of (E)-1,3-diphenyl-2-(1H-1,2,4-triazol-1-yl)allyl 4-fluorobenzoate (C15)
将实施例1步骤4)中的苯甲酸替换为同等摩尔量的4-氟苯甲酸,其他操作同实施例1,制得(C15)化合物。Substitute the benzoic acid in step 4) of Example 1 with 4-fluorobenzoic acid in the same molar amount, and other operations are the same as those in Example 1, to prepare compound (C15).
(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)烯丙基4-氟苯甲酸酯:黄色油状物,收率47.25%;1H NMR(500MHz,CDCl3)δ:8.08(s,1H,triazole-H),7.66(s,1H,triazole-H),7.44-7.39(m,3H,ArH),7.38-7.31(m,4H,ArH),7.21-7.15(m,3H,ArH),7.13-7.09(m,2H,ArH),7.02(s,1H,CH),6.95(s,1H,=CH),6.83-6.79(m,2H,ArH).HRMS(ESI)for C24H18FN3O2m/z:Calculated,400.1456,Found,400.1461[M+H]+.(E)-1,3-diphenyl-2-(1H-1,2,4-triazol-1-yl)allyl 4-fluorobenzoate: yellow oil, yield 47.25%; 1 H NMR (500MHz, CDCl 3 )δ: 8.08(s,1H,triazole-H),7.66(s,1H,triazole-H),7.44-7.39(m,3H,ArH),7.38-7.31(m, 4H, ArH), 7.21-7.15(m, 3H, ArH), 7.13-7.09(m, 2H, ArH), 7.02(s, 1H, CH), 6.95(s, 1H, =CH), 6.83-6.79( m,2H,ArH).HRMS(ESI)for C 24 H 18 FN 3 O 2 m/z: Calculated,400.1456,Found,400.1461[M+H] + .
实施例16(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)烯丙基3-硝基苯甲酸酯(C16)的制备Example 16 Preparation of (E)-1,3-diphenyl-2-(1H-1,2,4-triazol-1-yl)allyl 3-nitrobenzoate (C16)
将实施例1步骤4)中的苯甲酸替换为同等摩尔量的3-硝基苯甲酸,其他操作同实施例1,制得(C16)化合物。Substitute benzoic acid in step 4) of Example 1 with 3-nitrobenzoic acid in an equivalent molar amount, and other operations are the same as those in Example 1, to prepare compound (C16).
(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)烯丙基3-硝基苯甲酸酯:黄色蜡状物,收率15.16%;1H NMR(500MHz,CDCl3)δ:8.95-8.89(m,1H,ArH),8.47-8.43(m,1H,ArH),8.41-8.38(m,1H,ArH),8.11(s,1H,triazole-H),7.68(t,J=8.0Hz,1H,ArH),7.63(s,1H,triazole-H),7.44-7.37(m,5H,ArH),7.25-7.20(m,3H,ArH),7.07(s,1H,CH),6.94(s,1H,=CH),6.84-6.81(m,2H,ArH).HRMS(ESI)for C24H18N4O4 m/z:Calculated,427.1401,Found,427.1406[M+H]+.(E)-1,3-Diphenyl-2-(1H-1,2,4-triazol-1-yl)allyl 3-nitrobenzoate: yellow wax, yield 15.16 %; 1 H NMR (500 MHz, CDCl 3 ) δ: 8.95-8.89 (m, 1H, ArH), 8.47-8.43 (m, 1H, ArH), 8.41-8.38 (m, 1H, ArH), 8.11 (s, 1H, triazole-H), 7.68(t, J=8.0Hz, 1H, ArH), 7.63(s, 1H, triazole-H), 7.44-7.37(m, 5H, ArH), 7.25-7.20(m, 3H ,ArH),7.07(s,1H,CH),6.94(s,1H,=CH),6.84-6.81(m,2H,ArH).HRMS(ESI) for C 24 H 18 N 4 O 4 m/z :Calculated,427.1401,Found,427.1406[M+H] + .
实施例17(E)-1,3-二苯基-2-(1H-1,2,4-三唑-1-基)烯丙基3-硝基苯甲酸酯的制备Example 17 Preparation of (E)-1,3-diphenyl-2-(1H-1,2,4-triazol-1-yl)allyl 3-nitrobenzoate
1)1-苯基-2-(1H-1,2,4-三唑-1-基)乙-1-酮(Ⅱ)的合成:1) Synthesis of 1-phenyl-2-(1H-1,2,4-triazol-1-yl)ethan-1-one (II):
在500mL三口烧瓶中依次加入ɑ-溴代苯乙酮(19.90g,0.10mol)、1,2,4-三氮唑(7.60g,0.11mol),加入150mL丙酮作溶剂,用冰盐浴控温在0℃以下,通过恒压滴液漏斗逐滴缓慢滴加三乙胺(11.1g,0.11mol)。滴加完毕后,继续反应1h,去冰浴,室温下继续搅拌1h,TLC(VEA/VPE=1/1,v/v)跟踪反应进程。待反应结束后,抽滤除去反应生成的三乙胺盐酸盐,三乙胺盐酸盐用丙酮淋洗,合并淋洗液和滤液后,经脱溶得到红色油状物,冷却固化,用20mL异丙醇重结晶制得式(Ⅱ)所示的1-苯基-2-(1H-1,2,4-三唑-1-基)乙-1-酮;Add ɑ-bromoacetophenone (19.90g, 0.10mol) and 1,2,4-triazole (7.60g, 0.11mol) to a 500mL three-necked flask in turn, add 150mL of acetone as a solvent, and control with an ice-salt bath. When the temperature was below 0°C, triethylamine (11.1 g, 0.11 mol) was slowly added dropwise through a constant pressure dropping funnel. After the dropwise addition, the reaction was continued for 1 h, the ice bath was removed, and the stirring was continued for 1 h at room temperature, and the reaction progress was followed by TLC ( VE A /V PE =1/1, v/v). After the reaction, the triethylamine hydrochloride generated by the reaction was removed by suction filtration, and the triethylamine hydrochloride was rinsed with acetone. After combining the eluent and the filtrate, a red oil was obtained by precipitation, which was cooled and solidified. 1-phenyl-2-(1H-1,2,4-triazol-1-yl)ethan-1-one represented by formula (II) was obtained by recrystallization from isopropanol;
2)(E)-1-苯基-2-(1H-1,2,4-三唑-1-基)-3-(4-(三氟甲基)苯基)丙-2-烯-1-酮(Ⅲ-2)的合成:2) (E)-1-phenyl-2-(1H-1,2,4-triazol-1-yl)-3-(4-(trifluoromethyl)phenyl)prop-2-ene- Synthesis of 1-keto (Ⅲ-2):
在装有分水器、温度计、冷凝管的100mL三口烧瓶中依次加入式(Ⅱ)所示的1-苯基-2-(1H-1,2,4-三唑-1-基)乙-1-酮(5.62g,30.00mol),对三氟甲基苯甲醛(5.75g,33.00mmol),加入30mL甲苯作溶剂,滴加哌啶(0.20g,2.35mmol)作为催化剂,加热回流反应7h,TLC(VEA/VPE=1/1,v/v)跟踪反应进程。待反应结束后,冷却至室温,将反应液转移至分液漏斗内,水洗后分层,有机相再用饱和食盐水洗涤两次(每次洗涤采用的饱和食盐水体积20mL),无水硫酸钠干燥,过滤,滤液浓缩除去溶剂,浓缩残留物经柱层析分离提纯(洗脱剂为体积比1:1石油醚和乙酸乙酯)制得式(Ⅲ-2)所示的(E)-1-苯基-2-(1H-1,2,4-三唑-1-基)-3-(4-(三氟甲基)苯基)丙-2-烯-1-酮;1-phenyl-2-(1H-1,2,4-triazol-1-yl)ethyl- 1-ketone (5.62g, 30.00mol), p-trifluoromethylbenzaldehyde (5.75g, 33.00mmol), add 30mL of toluene as a solvent, dropwise add piperidine (0.20g, 2.35mmol) as a catalyst, heat under reflux for 7h , TLC ( VE A /V PE =1/1, v/v) followed the progress of the reaction. After the reaction was completed, it was cooled to room temperature, and the reaction solution was transferred to a separatory funnel. After washing, the layers were separated. The organic phase was washed twice with saturated brine (volume of saturated brine used in each washing was 20 mL), and anhydrous sulfuric acid Dry over sodium, filter, concentrate the filtrate to remove the solvent, and separate and purify the concentrated residue by column chromatography (eluent is 1:1 petroleum ether and ethyl acetate by volume) to obtain (E) represented by formula (III-2) -1-phenyl-2-(1H-1,2,4-triazol-1-yl)-3-(4-(trifluoromethyl)phenyl)prop-2-en-1-one;
3)(E)-1-苯基-2-(1H-1,2,4-三唑-1-基)-3-(4-(三氟甲基)苯基)丙-2-烯-1-醇(Ⅳ-2)的合成:3) (E)-1-phenyl-2-(1H-1,2,4-triazol-1-yl)-3-(4-(trifluoromethyl)phenyl)prop-2-ene- Synthesis of 1-ol (IV-2):
将式(Ⅲ-2)所示的(E)-1-苯基-2-(1H-1,2,4-三唑-1-基)-3-(4-(三氟甲基)苯基)丙-2-烯-1-酮(2.40g,7.00mmol)加入到50mL圆底烧瓶中,加入15mL甲醇作溶剂,冰浴控温在0℃,加入PEG 600(0.50g,0.83mmol),然后在搅拌下分批加入硼氢化钠(0.42g,11.0mmol),在0℃冰浴下反应4h,TLC(VEA/VPE=1/2,v/v)跟踪反应进程。待反应结束后,将反应液脱溶,脱溶浓缩物中滴加1N盐酸溶液,用二氯甲烷萃取三次(每次萃取用二氯甲烷15mL),合并有机相,经无水硫酸钠干燥,过滤,旋干溶剂制得式(Ⅳ-2)所示的(E)-1-苯基-2-(1H-1,2,4-三唑-1-基)-3-(4-(三氟甲基)苯基)丙-2-烯-1-醇;(E)-1-phenyl-2-(1H-1,2,4-triazol-1-yl)-3-(4-(trifluoromethyl)benzene represented by formula (III-2) base) prop-2-en-1-one (2.40g, 7.00mmol) was added to a 50mL round-bottomed flask, 15mL of methanol was added as a solvent, the temperature was controlled at 0°C in an ice bath, and PEG 600 (0.50g, 0.83mmol) was added Then, sodium borohydride (0.42 g, 11.0 mmol) was added in batches with stirring, and the reaction was carried out under ice bath at 0 °C for 4 h, and the reaction progress was followed by TLC ( VE A /V PE =1/2, v/v). After the reaction was completed, the reaction solution was desolubilized, 1N hydrochloric acid solution was added dropwise to the precipitation concentrate, extracted three times with dichloromethane (15 mL of dichloromethane was used for each extraction), the organic phases were combined and dried over anhydrous sodium sulfate, Filter, spin dry the solvent to obtain (E)-1-phenyl-2-(1H-1,2,4-triazol-1-yl)-3-(4-( trifluoromethyl)phenyl)prop-2-en-1-ol;
4)(E)-1-苯基-2-(1H-1,2,4-三唑-1-基)-3-(4-(三氟甲基)苯基)烯丙基苯甲酸酯(B1)的合成:4) (E)-1-phenyl-2-(1H-1,2,4-triazol-1-yl)-3-(4-(trifluoromethyl)phenyl)allylbenzoic acid Synthesis of Ester (B1):
将式(Ⅳ-2)所示的(E)-1-苯基-2-(1H-1,2,4-三唑-1-基)-3-(4-(三氟甲基)苯基)丙-2-烯-1-醇(0.25g,0.72mmol)、苯甲酸(0.10g,0.80mmol)、DMAP(0.09g,0.72mmol)、EDC(0.28g,1.44mmol)加入到5mL二氯甲烷中,室温下反应4h,随后将反应液转移至分液漏斗内,经水洗(3×5mL),再用饱和食盐水洗涤(2×5mL),无水硫酸钠干燥,过滤,滤液浓缩除去溶剂,浓缩残留物经柱层析分离提纯(洗脱剂为体积比1:1石油醚和乙酸乙酯)制得式(B1)化合物。(E)-1-phenyl-2-(1H-1,2,4-triazol-1-yl)-3-(4-(trifluoromethyl)benzene represented by formula (IV-2) yl)prop-2-en-1-ol (0.25 g, 0.72 mmol), benzoic acid (0.10 g, 0.80 mmol), DMAP (0.09 g, 0.72 mmol), EDC (0.28 g, 1.44 mmol) were added to 5 mL of dilute In methyl chloride, react at room temperature for 4 h, then transfer the reaction solution to a separatory funnel, wash with water (3×5 mL), then with saturated brine (2×5 mL), dry over anhydrous sodium sulfate, filter, and concentrate the filtrate The solvent is removed, and the concentrated residue is separated and purified by column chromatography (eluent is petroleum ether and ethyl acetate in a volume ratio of 1:1) to obtain the compound of formula (B1).
其中式(Ⅲ)和式(Ⅳ)结构式中的取代基R2均为三氟甲基,分别形成了式(Ⅲ-2)和式(Ⅳ-2)的分子结构。Wherein, the substituents R 2 in the structural formulae of formula (III) and formula (IV) are all trifluoromethyl groups, respectively forming the molecular structures of formula (III-2) and formula (IV-2).
(E)-1-苯基-2-(1H-1,2,4-三唑-1-基)-3-(4-(三氟甲基)苯基)烯丙基苯甲酸酯:黄色油状物,收率46.37%;1H NMR(500MHz,CDCl3)δ:8.10(s,1H,triazole-H),8.09-8.07(m,2H,ArH),7.67(s,1H,triazole-H),7.59(t,J=7.5Hz,1H,ArH),7.49-7.42(m,6H,ArH),7.40-7.33(m,3H,ArH),7.04(s,1H,CH),6.99(s,1H,=CH),6.96(d,J=8.3Hz,2H,ArH).HRMS(ESI)for C25H18F3N3O2 m/z:Calculated,450.1424,Found,450.1423[M+H]+.(E)-1-Phenyl-2-(1H-1,2,4-triazol-1-yl)-3-(4-(trifluoromethyl)phenyl)allylbenzoate: Yellow oil, yield 46.37%; 1 H NMR (500MHz, CDCl 3 )δ: 8.10(s,1H,triazole-H),8.09-8.07(m,2H,ArH),7.67(s,1H,triazole-H) H), 7.59(t, J=7.5Hz, 1H, ArH), 7.49-7.42(m, 6H, ArH), 7.40-7.33(m, 3H, ArH), 7.04(s, 1H, CH), 6.99( s,1H,=CH),6.96(d,J=8.3Hz,2H,ArH).HRMS(ESI)for C 25 H 18 F 3 N 3 O 2 m/z: Calculated,450.1424,Found,450.1423[M +H] + .
实施例18(E)-1-苯基-2-(1H-1,2,4-三唑-1-基)-3-(4-(三氟甲基)苯基)烯丙基2-甲基苯甲酸酯(B2)的制备Example 18(E)-1-phenyl-2-(1H-1,2,4-triazol-1-yl)-3-(4-(trifluoromethyl)phenyl)allyl 2- Preparation of methyl benzoate (B2)
将实施例17步骤4)中的苯甲酸替换为同等摩尔量的2-甲基苯甲酸,其他操作同实施例17,制得(B2)化合物。Substitute the benzoic acid in step 4) of Example 17 with 2-methylbenzoic acid in the same molar amount, and other operations are the same as those in Example 17, to obtain compound (B2).
(E)-1-苯基-2-(1H-1,2,4-三唑-1-基)-3-(4-(三氟甲基)苯基)烯丙基2-甲基苯甲酸酯:黄色油状物,收率58.38%;1H NMR(500MHz,CDCl3)δ:8.10(s,1H,triazole-H),8.01-7.94(m,1H,ArH),7.65(s,1H,triazole-H),7.48-7.42(m,5H,ArH),7.39-7.33(m,3H,ArH),7.26(t,J=7.4Hz,2H,ArH),7.03(s,1H,CH),6.96(s,2H,ArH),6.94(s,1H,=CH),2.57(s,3H,CH3).HRMS(ESI)for C26H20F3N3O2 m/z:Calculated,464.1580,Found,464.1577[M+H]+.(E)-1-Phenyl-2-(1H-1,2,4-triazol-1-yl)-3-(4-(trifluoromethyl)phenyl)allyl 2-methylbenzene Formate: yellow oil, yield 58.38%; 1 H NMR (500MHz, CDCl 3 )δ: 8.10(s, 1H, triazole-H), 8.01-7.94(m, 1H, ArH), 7.65(s, 1H, triazole-H), 7.48-7.42(m, 5H, ArH), 7.39-7.33(m, 3H, ArH), 7.26(t, J=7.4Hz, 2H, ArH), 7.03(s, 1H, CH) ), 6.96(s, 2H, ArH), 6.94(s, 1H, =CH), 2.57(s, 3H, CH 3 ). HRMS(ESI) for C 26 H 20 F 3 N 3 O 2 m/z: Calculated,464.1580,Found,464.1577[M+H] + .
实施例19(E)-1-苯基-2-(1H-1,2,4-三唑-1-基)-3-(4-(三氟甲基)苯基)烯丙基2-氟苯甲酸酯(B3)的制备Example 19 (E)-1-phenyl-2-(1H-1,2,4-triazol-1-yl)-3-(4-(trifluoromethyl)phenyl)allyl 2- Preparation of Fluorobenzoate (B3)
将实施例17步骤4)中的苯甲酸替换为同等摩尔量的2-氟苯甲酸,其他操作同实施例17,制得(B3)化合物。Substitute the benzoic acid in step 4) of Example 17 with 2-fluorobenzoic acid in the same molar amount, and other operations are the same as those in Example 17, to obtain compound (B3).
(E)-1-苯基-2-(1H-1,2,4-三唑-1-基)-3-(4-(三氟甲基)苯基)烯丙基2-氟苯甲酸酯:黄色油状物,收率72.35%;1H NMR(500MHz,CDCl3)δ:8.08(s,1H,triazole-H),8.01-7.94(m,1H,ArH),7.69(s,1H,triazole-H),7.61-7.52(m,1H,ArH),7.48-7.42(m,4H,ArH),7.38-7.32(m,3H,ArH),7.23(t,J=7.6Hz,1H,ArH),7.20-7.14(m,1H,ArH),7.07(s,1H,CH),7.04(s,1H,=CH),6.96(d,J=8.2Hz,2H,ArH).HRMS(ESI)for C25H17F4N3O2 m/z:Calculated,468.1330,Found,468.1327[M+H]+.(E)-1-Phenyl-2-(1H-1,2,4-triazol-1-yl)-3-(4-(trifluoromethyl)phenyl)allyl 2-fluorobenzyl Ester: yellow oil, yield 72.35%; 1 H NMR (500MHz, CDCl 3 )δ: 8.08(s,1H,triazole-H),8.01-7.94(m,1H,ArH),7.69(s,1H) ,triazole-H),7.61-7.52(m,1H,ArH),7.48-7.42(m,4H,ArH),7.38-7.32(m,3H,ArH),7.23(t,J=7.6Hz,1H, ArH),7.20-7.14(m,1H,ArH),7.07(s,1H,CH),7.04(s,1H,=CH),6.96(d,J=8.2Hz,2H,ArH).HRMS(ESI ) for C 25 H 17 F 4 N 3 O 2 m/z: Calculated,468.1330,Found,468.1327[M+H] + .
实施例20(E)-1-苯基-2-(1H-1,2,4-三唑-1-基)-3-(4-(三氟甲基)苯基)烯丙基4-叔丁基苯甲酸酯(B4)的制备Example 20(E)-1-phenyl-2-(1H-1,2,4-triazol-1-yl)-3-(4-(trifluoromethyl)phenyl)allyl 4- Preparation of tert-butyl benzoate (B4)
将实施例17步骤4)中的苯甲酸替换为同等摩尔量的4-叔丁基苯甲酸,其他操作同实施例17,制得(B4)化合物。Substitute the benzoic acid in step 4) of Example 17 with 4-tert-butylbenzoic acid in the same molar amount, and other operations are the same as those in Example 17, to obtain compound (B4).
(E)-1-苯基-2-(1H-1,2,4-三唑-1-基)-3-(4-(三氟甲基)苯基)烯丙基4-叔丁基苯甲酸酯:白色蜡状物,收率55.16%;1H NMR(500MHz,CDCl3)δ:8.08(s,1H,triazole-H),8.05-8.01(m,2H,ArH),7.68(s,1H,triazole-H),7.50-7.48(m,2H,ArH),7.47-7.42(m,4H,ArH),7.38-7.32(m,3H,ArH),7.03(s,1H,CH),6.98(s,1H,=CH),6.95(d,J=8.3Hz,2H,ArH),1.34(s,9H,t-Bu).HRMS(ESI)for C29H26F3N3O2 m/z:Calculated,506.2050,Found,506.2061[M+H]+.(E)-1-Phenyl-2-(1H-1,2,4-triazol-1-yl)-3-(4-(trifluoromethyl)phenyl)allyl 4-tert-butyl Benzoate: white wax, yield 55.16%; 1 H NMR (500 MHz, CDCl 3 ) δ: 8.08 (s, 1H, triazole-H), 8.05-8.01 (m, 2H, ArH), 7.68 ( s,1H,triazole-H),7.50-7.48(m,2H,ArH),7.47-7.42(m,4H,ArH),7.38-7.32(m,3H,ArH),7.03(s,1H,CH) ,6.98(s,1H,=CH),6.95(d,J=8.3Hz,2H,ArH),1.34(s,9H,t-Bu).HRMS(ESI)for C 29 H 26 F 3 N 3 O 2 m/z: Calculated,506.2050,Found,506.2061[M+H] + .
实施例21(E)-1-苯基-2-(1H-1,2,4-三唑-1-基)-3-(4-(三氟甲基)苯基)烯丙基3-氯苯甲酸酯(B5)的制备Example 21 (E)-1-phenyl-2-(1H-1,2,4-triazol-1-yl)-3-(4-(trifluoromethyl)phenyl)allyl 3- Preparation of Chlorobenzoate (B5)
将实施例17步骤4)中的苯甲酸替换为同等摩尔量的3-氯苯甲酸,其他操作同实施例17,制得(B5)化合物。Substitute the benzoic acid in step 4) of Example 17 with 3-chlorobenzoic acid in the same molar amount, and other operations are the same as those in Example 17, to obtain compound (B5).
(E)-1-苯基-2-(1H-1,2,4-三唑-1-基)-3-(4-(三氟甲基)苯基)烯丙基3-氯苯甲酸酯:黄色油状物,收率48.75%;1H NMR(500MHz,CDCl3)δ:8.10(s,1H,triazole-H),8.04(t,J=1.8Hz,1H,ArH),7.98-7.94(m,1H,ArH),7.64(s,1H,triazole-H),7.59-7.55(m,1H,ArH),7.46(d,J=8.3Hz,2H,ArH),7.43-7.36(m,6H,ArH),7.04(s,1H,CH),6.97(s,2H,ArH),6.95(s,1H,=CH).HRMS(ESI)for C25H17ClF3N3O2 m/z:Calculated,484.1034,Found,484.1028[M+H]+.(E)-1-Phenyl-2-(1H-1,2,4-triazol-1-yl)-3-(4-(trifluoromethyl)phenyl)allyl 3-chlorobenzyl Ester: yellow oil, yield 48.75%; 1 H NMR (500 MHz, CDCl 3 ) δ: 8.10 (s, 1H, triazole-H), 8.04 (t, J=1.8 Hz, 1H, ArH), 7.98- 7.94(m,1H,ArH),7.64(s,1H,triazole-H),7.59-7.55(m,1H,ArH),7.46(d,J=8.3Hz,2H,ArH),7.43-7.36(m ,6H,ArH),7.04(s,1H,CH),6.97(s,2H,ArH),6.95(s,1H,=CH).HRMS(ESI) for C 25 H 17 ClF 3 N 3 O 2 m /z:Calculated,484.1034,Found,484.1028[M+H] + .
实施例22(E)-1-苯基-2-(1H-1,2,4-三唑-1-基)-3-(4-(三氟甲基)苯基)烯丙基4-正丙基苯甲酸酯(B6)的制备Example 22(E)-1-phenyl-2-(1H-1,2,4-triazol-1-yl)-3-(4-(trifluoromethyl)phenyl)allyl 4- Preparation of n-propyl benzoate (B6)
将实施例17步骤4)中的苯甲酸替换为同等摩尔量的4-正丙基苯甲酸,其他操作同实施例17,制得(B6)化合物。Substitute the benzoic acid in step 4) of Example 17 with 4-n-propylbenzoic acid in the same molar amount, and other operations are the same as those in Example 17, to obtain compound (B6).
(E)-1-苯基-2-(1H-1,2,4-三唑-1-基)-3-(4-(三氟甲基)苯基)烯丙基4-正丙基苯甲酸酯:白色蜡状物,收率55.32%;1H NMR(500MHz,CDCl3)δ:8.10(s,1H,triazole-H),8.00(d,J=8.2Hz,2H,ArH),7.72(s,1H,triazole-H),7.58(t,J=7.4Hz,2H,ArH),7.45-7.41(m,3H,ArH),7.40-7.32(m,4H,ArH),7.02(s,1H,CH),6.99(s,1H,=CH),6.96(d,J=8.2Hz,2H,ArH),2.65(t,J=7.5Hz,2H,CH2),1.70-1.61(m,2H,CH2),0.94(t,J=7.3Hz,3H,CH3).HRMS(ESI)for C28H24F3N3O2 m/z:Calculated,492.1893,Found,492.1861[M+H]+.(E)-1-Phenyl-2-(1H-1,2,4-triazol-1-yl)-3-(4-(trifluoromethyl)phenyl)allyl 4-n-propyl Benzoate: white wax, yield 55.32%; 1 H NMR (500 MHz, CDCl 3 ) δ: 8.10 (s, 1H, triazole-H), 8.00 (d, J=8.2 Hz, 2H, ArH) ,7.72(s,1H,triazole-H),7.58(t,J=7.4Hz,2H,ArH),7.45-7.41(m,3H,ArH),7.40-7.32(m,4H,ArH),7.02( s, 1H, CH), 6.99(s, 1H, =CH), 6.96(d, J=8.2Hz, 2H, ArH), 2.65(t, J=7.5Hz, 2H, CH 2 ), 1.70-1.61( m, 2H, CH 2 ), 0.94 (t, J=7.3 Hz, 3H, CH 3 ). HRMS(ESI) for C 28 H 24 F 3 N 3 O 2 m/z: Calculated, 492.1893, Found, 492.1861[ M+H] + .
实施例23(E)-1-苯基-2-(1H-1,2,4-三唑-1-基)-3-(4-(三氟甲基)苯基)烯丙基4-甲氧基苯甲酸酯(B7)的制备Example 23 (E)-1-phenyl-2-(1H-1,2,4-triazol-1-yl)-3-(4-(trifluoromethyl)phenyl)allyl 4- Preparation of Methoxybenzoate (B7)
将实施例17步骤4)中的苯甲酸替换为同等摩尔量的4-甲氧基苯甲酸,其他操作同实施例17,制得(B7)化合物。Substitute the benzoic acid in step 4) of Example 17 with 4-methoxybenzoic acid in the same molar amount, and other operations are the same as those in Example 17, to obtain compound (B7).
(E)-1-苯基-2-(1H-1,2,4-三唑-1-基)-3-(4-(三氟甲基)苯基)烯丙基4-甲氧基苯甲酸酯:白色蜡状物,收率44.10%;1H NMR(500MHz,CDCl3)δ:8.08(s,1H,triazole-H),8.06-8.03(m,2H,ArH),7.67(s,1H,triazole-H),7.46(d,J=8.3Hz,2H,ArH),7.43-7.40(m,2H,ArH),7.38-7.33(m,3H,ArH),7.00(s,1H,CH),6.98(s,1H,=CH),6.97-6.94(m,4H,ArH),3.86(s,3H,OCH3).HRMS(ESI)for C26H20F3N3O3 m/z:Calculated,480.1530,Found,480.1530[M+H]+.(E)-1-Phenyl-2-(1H-1,2,4-triazol-1-yl)-3-(4-(trifluoromethyl)phenyl)allyl 4-methoxy Benzoate: white wax, yield 44.10%; 1 H NMR (500 MHz, CDCl 3 ) δ: 8.08 (s, 1H, triazole-H), 8.06-8.03 (m, 2H, ArH), 7.67 ( s,1H,triazole-H),7.46(d,J=8.3Hz,2H,ArH),7.43-7.40(m,2H,ArH),7.38-7.33(m,3H,ArH),7.00(s,1H ,CH),6.98(s,1H, = CH),6.97-6.94( m ,4H,ArH),3.86(s, 3H , OCH3 ) .HRMS (ESI)for C26H20F3N3O3 m/z: Calculated,480.1530,Found,480.1530[M+H] + .
实施例24(E)-1-苯基-2-(1H-1,2,4-三唑-1-基)-3-(4-(三氟甲基)苯基)烯丙基2,4-二氯苯甲酸酯(B8)的制备Example 24 (E)-1-phenyl-2-(1H-1,2,4-triazol-1-yl)-3-(4-(trifluoromethyl)phenyl)allyl 2, Preparation of 4-dichlorobenzoate (B8)
将实施例17步骤4)中的苯甲酸替换为同等摩尔量的2,4-二氯苯甲酸,其他操作同实施例17,制得(B8)化合物。Substitute the benzoic acid in step 4) of Example 17 with 2,4-dichlorobenzoic acid in the same molar amount, and other operations are the same as those in Example 17, to obtain compound (B8).
(E)-1-苯基-2-(1H-1,2,4-三唑-1-基)-3-(4-(三氟甲基)苯基)烯丙基2,4-二氯苯甲酸酯:黄色油状物,收率66.16%;1H NMR(500MHz,CDCl3)δ:8.10(s,1H,triazole-H),7.85(d,J=8.5Hz,1H,ArH),7.63(s,1H,triazole-H),7.49-7.45(m,3H,ArH),7.39-7.35(m,4H,ArH),7.34-7.31(m,2H,ArH),7.04(s,1H,CH),7.01(s,1H,=CH),6.95(d,J=8.2Hz,2H,ArH).(E)-1-Phenyl-2-(1H-1,2,4-triazol-1-yl)-3-(4-(trifluoromethyl)phenyl)allyl 2,4-di Chlorobenzoate: yellow oil, yield 66.16%; 1 H NMR (500 MHz, CDCl 3 ) δ: 8.10 (s, 1H, triazole-H), 7.85 (d, J=8.5 Hz, 1H, ArH) ,7.63(s,1H,triazole-H),7.49-7.45(m,3H,ArH),7.39-7.35(m,4H,ArH),7.34-7.31(m,2H,ArH),7.04(s,1H ,CH),7.01(s,1H,=CH),6.95(d,J=8.2Hz,2H,ArH).
实施例25(E)-1-苯基-2-(1H-1,2,4-三唑-1-基)-3-(4-(三氟甲基)苯基)烯丙基3-硝基苯甲酸酯(B9)的制备Example 25 (E)-1-phenyl-2-(1H-1,2,4-triazol-1-yl)-3-(4-(trifluoromethyl)phenyl)allyl 3- Preparation of nitrobenzoate (B9)
将实施例17步骤4)中的苯甲酸替换为同等摩尔量的3-硝基苯甲酸,其他操作同实施例17,制得(B9)化合物。Substitute the benzoic acid in step 4) of Example 17 with 3-nitrobenzoic acid in the same molar amount, and other operations are the same as those in Example 17, to obtain compound (B9).
(E)-1-苯基-2-(1H-1,2,4-三唑-1-基)-3-(4-(三氟甲基)苯基)烯丙基3-硝基苯甲酸酯:白色蜡状物,收率23.17%;1H NMR(500MHz,CDCl3)δ:8.93-8.89(m,1H,ArH),8.48-8.43(m,1H,ArH),8.40(d,J=7.9Hz,1H,ArH),8.13(s,1H,triazole-H),7.70(t,J=8.0Hz,1H,ArH),7.67(s,1H,triazole-H),7.49-7.39(m,7H,ArH),7.09(s,1H,CH),6.99(s,1H,=CH),6.98(d,J=8.3Hz,2H,ArH).HRMS(ESI)for C25H17F3N4O4 m/z:Calculated,495.1275,Found,495.1271[M+H]+.(E)-1-Phenyl-2-(1H-1,2,4-triazol-1-yl)-3-(4-(trifluoromethyl)phenyl)allyl 3-nitrobenzene Formate: white wax, yield 23.17%; 1 H NMR (500 MHz, CDCl 3 ) δ: 8.93-8.89 (m, 1H, ArH), 8.48-8.43 (m, 1H, ArH), 8.40 (d ,J=7.9Hz,1H,ArH),8.13(s,1H,triazole-H),7.70(t,J=8.0Hz,1H,ArH),7.67(s,1H,triazole-H),7.49-7.39 (m, 7H, ArH), 7.09 (s, 1H, CH), 6.99 (s, 1H, =CH), 6.98 (d, J = 8.3 Hz, 2H, ArH). HRMS (ESI) for C 25 H 17 F 3 N 4 O 4 m/z: Calculated, 495.1275, Found, 495.1271[M+H] + .
实施例26(E)-1-苯基-2-(1H-1,2,4-三唑-1-基)-3-(4-(三氟甲基)苯基)烯丙基2,3-二氯苯甲酸酯(B10)的制备Example 26 (E)-1-phenyl-2-(1H-1,2,4-triazol-1-yl)-3-(4-(trifluoromethyl)phenyl)allyl 2, Preparation of 3-dichlorobenzoate (B10)
将实施例17步骤4)中的苯甲酸替换为同等摩尔量的2,3-二氯苯甲酸,其他操作同实施例17,制得(B10)化合物。Substitute benzoic acid in step 4) of Example 17 with 2,3-dichlorobenzoic acid in the same molar amount, and other operations are the same as those in Example 17, to prepare compound (B10).
(E)-1-苯基-2-(1H-1,2,4-三唑-1-基)-3-(4-(三氟甲基)苯基)烯丙基2,3-二氯苯甲酸酯:黄色油状物,收率33.25%;1H NMR(500MHz,CDCl3)δ:8.11(s,1H,triazole-H),7.67(dd,J=7.8,1.6Hz,1H,ArH),7.64(s,1H,triazole-H),7.47(d,J=8.3Hz,2H,ArH),7.45-7.42(m,2H,ArH),7.41-7.36(m,3H,ArH),7.31-7.27(m,2H,ArH),7.04(s,1H,CH),6.99(s,1H,=CH),6.95(d,J=8.3Hz,2H,ArH).HRMS(ESI)for C25H16Cl2F3N3O2 m/z:Calculated,518.0644,Found,518.0644[M+H]+.(E)-1-Phenyl-2-(1H-1,2,4-triazol-1-yl)-3-(4-(trifluoromethyl)phenyl)allyl 2,3-di Chlorobenzoate: yellow oil, yield 33.25%; 1 H NMR (500 MHz, CDCl 3 ) δ: 8.11 (s, 1H, triazole-H), 7.67 (dd, J=7.8, 1.6 Hz, 1H, ArH), 7.64(s, 1H, triazole-H), 7.47(d, J=8.3Hz, 2H, ArH), 7.45-7.42(m, 2H, ArH), 7.41-7.36(m, 3H, ArH), 7.31-7.27(m,2H,ArH),7.04(s,1H,CH),6.99(s,1H,=CH),6.95(d,J=8.3Hz,2H,ArH).HRMS(ESI)for C 25 H 16 Cl 2 F 3 N 3 O 2 m/z: Calculated, 518.0644, Found, 518.0644[M+H] + .
实施例27杀菌活性测试Example 27 Bactericidal activity test
试验对象:番茄早疫病菌(Alternari asolani)、小麦赤霉病菌(Gibberellazeae)、马铃薯晚疫病菌(Phytophthora infestans)、辣椒疫霉病菌(Phytophthoracapsici)、油菜菌核(Sclerotinia sclerotiorum)、黄瓜灰霉病菌(Botrytis cinerea)、水稻纹枯病菌(Rhizoctonia solani)、黄瓜枯萎病菌(Fusarium oxysporum)、花生褐斑病菌(Cercospora arachidicola)、苹果轮纹病菌(Physalosporapiricola)。Test objects: Alternari assolani, Gibberellazeae, Phytophthora infestans, Phytophthoracapsici, Sclerotinia sclerotiorum, Botrytis cucumber (Phytophthora infestans) Botrytis cinerea), Rhizoctonia solani, Fusarium oxysporum, Cercospora arachidicola, Physalosporapiricola.
将实施例1~16制备的式(C1)~式(C16)所示化合物,将实施例17~26制备的式(B1)~式(B10)所示化合物分别标记为待测化合物,并按以下方式进行杀菌活性测试:The compounds represented by formulas (C1) to (C16) prepared in Examples 1 to 16 and the compounds represented by formulas (B1) to (B10) prepared in Examples 17 to 26 were respectively marked as compounds to be tested, and the The bactericidal activity test was performed in the following ways:
试验处理:各待测化合物用DMSO溶解成1%EC母液备用。采用抑菌圈法,评价待测化合物在50ppm剂量下对试验靶标的室内杀菌活性,另设溶剂清水对照(QCK),另设有效含量为50ppm的氟唑菌酰胺对照(YCK)。Experimental treatment: each compound to be tested was dissolved in DMSO into a 1% EC stock solution for later use. The inhibition zone method was used to evaluate the indoor bactericidal activity of the tested compounds against the test target at a dose of 50 ppm. A solvent clear water control (QCK) and a fluoxamid control (YCK) with an effective content of 50 ppm were also set up.
试验方法:用移液枪吸取150微升上述配置的EC母液,溶于2.85mL的吐温水中,配成待测化合物的有效浓度为500ppm的药液。用移液枪吸取1mL药液放入已灭菌的培养皿中,再放入9mL的PDA培养基,摇匀,冷却。用打孔器打取圆形菌饼后用接种针挑至培养皿中央,然后将培养皿置于培养箱27℃中培养,48~72h后测量菌落直径。菌落纯生长量为菌落平均直径与菌饼直径的差值,抑菌率(%)计算方法参照如下公式进行计算。Test method: suck 150 microliters of the EC mother solution prepared above with a pipette, dissolve it in 2.85 mL of Tween water, and prepare a medicinal solution with an effective concentration of 500 ppm of the compound to be tested. Pipette 1mL of liquid medicine into a sterilized petri dish, then put 9mL of PDA medium, shake well, and cool. Use a hole punch to punch out the circular bacterial cake and pick it to the center of the petri dish with an inoculating needle, then place the petri dish in an incubator at 27°C for cultivation, and measure the colony diameter after 48 to 72 hours. The pure growth of the colony is the difference between the average diameter of the colony and the diameter of the bacterial cake, and the calculation method of the bacteriostatic rate (%) is calculated by referring to the following formula.
上述计算公式中的对照菌落纯生长量,是指清水对照(QCK)测试下的菌落纯生长量。The pure growth of the control colony in the above calculation formula refers to the pure growth of the colony under the clear water control (QCK) test.
杀菌活性测试结果如表1所示。The bactericidal activity test results are shown in Table 1.
表1 50ppm下各化合物的杀菌活性(%防效)Table 1 Bactericidal activity (% control effect) of each compound at 50 ppm
由表可知,C系列化合物对油菜菌核表现出了不错的抑制率,大部分化合物抑菌率都超过了60%,其中C1、C7、C14对油菜菌核的抑菌率超过了70%,C8、C13对水稻纹枯的抑菌率也超过了60%,C7、C11、C13、C16对苹果轮纹的抑菌率都达到了58.3%,但均不及对照药剂氟唑菌酰胺。It can be seen from the table that the C series compounds showed a good inhibitory rate on rape sclerotium, and most of the compounds had an inhibitory rate of more than 60%. Among them, C1, C7, and C14 had an inhibitory rate of more than 70% on rape sclerotium. The antibacterial rate of C8 and C13 to rice sheath blight also exceeded 60%, and the antibacterial rate of C7, C11, C13, and C16 to apple ring striae reached 58.3%, but they were not as good as the control agent fluoxamid.
由表可知,B系列各化合物均表现出一定的活性,其中B1、B2、B3、B5、B6、B7、B8、B9化合物对于油菜菌核的抑制率均超过了50%,B7对小麦赤霉的抑制率超过了50%,B1、B3、B6、B7对于水稻纹枯的防治率超过了50%,总体来说B系列的杀菌活性不是非常理想,与对照药剂氟唑菌酰胺相比,对辣椒疫霉的防治率高于氟唑菌酰胺,部分化合物对小麦赤霉的防治率也要高于氟唑菌酰胺。It can be seen from the table that the compounds in the B series all showed a certain activity, among which the compounds B1, B2, B3, B5, B6, B7, B8, and B9 all had more than 50% inhibition rates on rape sclerotia, and B7 had an inhibitory effect on the sclerotium of wheat. The inhibition rate of B1, B3, B6, and B7 for rice sheath blight exceeded 50%. Overall, the bactericidal activity of the B series is not very ideal. The control rate of Phytophthora capsicum was higher than that of Fluconazole, and the control rate of some compounds was also higher than that of Fluconazole.
本说明书所述的内容仅仅是对发明构思实现形式的列举,本发明的保护范围不应当被视为仅限于实施例所陈述的具体形式。The content described in this specification is only an enumeration of the realization forms of the inventive concept, and the protection scope of the present invention should not be regarded as being limited to the specific forms stated in the embodiments.
Claims (9)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202010504490.4A CN111574466B (en) | 2020-06-05 | 2020-06-05 | A kind of allyl benzoate compound containing triazole and preparation method and application thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202010504490.4A CN111574466B (en) | 2020-06-05 | 2020-06-05 | A kind of allyl benzoate compound containing triazole and preparation method and application thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN111574466A true CN111574466A (en) | 2020-08-25 |
| CN111574466B CN111574466B (en) | 2021-08-03 |
Family
ID=72122276
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202010504490.4A Active CN111574466B (en) | 2020-06-05 | 2020-06-05 | A kind of allyl benzoate compound containing triazole and preparation method and application thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN111574466B (en) |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS59186964A (en) * | 1983-04-05 | 1984-10-23 | Sumitomo Chem Co Ltd | Production of alcoholic compound |
| CN101445488A (en) * | 2007-11-27 | 2009-06-03 | 西南大学 | Triazoles compounds with antimicrobial activity and preparation method and pharmaceutical use thereof |
| CN104370892A (en) * | 2014-10-27 | 2015-02-25 | 湖南大学 | 1-(7-methoxybenzofuran-5-yl)-3-(2-methoxyphenyl)-2-(1,2,4-triazole-1-yl) allyl alcohol |
| CN104557888A (en) * | 2015-01-23 | 2015-04-29 | 邵阳学院 | Application of furan phenol allyl alcohol derivative used as herbicide |
-
2020
- 2020-06-05 CN CN202010504490.4A patent/CN111574466B/en active Active
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS59186964A (en) * | 1983-04-05 | 1984-10-23 | Sumitomo Chem Co Ltd | Production of alcoholic compound |
| CN101445488A (en) * | 2007-11-27 | 2009-06-03 | 西南大学 | Triazoles compounds with antimicrobial activity and preparation method and pharmaceutical use thereof |
| CN104370892A (en) * | 2014-10-27 | 2015-02-25 | 湖南大学 | 1-(7-methoxybenzofuran-5-yl)-3-(2-methoxyphenyl)-2-(1,2,4-triazole-1-yl) allyl alcohol |
| CN104557888A (en) * | 2015-01-23 | 2015-04-29 | 邵阳学院 | Application of furan phenol allyl alcohol derivative used as herbicide |
Non-Patent Citations (2)
| Title |
|---|
| YONG KI MIN,等: "New Lead Compounds for Brassinosteroid Biosynthesis Inhibitors", 《BIOORGANIC & MEDICINAL CHEMISTRY LETTERS》 * |
| 李春芳,等: "新型含氟三唑类化合物的合成及生物活性研究", 《青岛科技大学学报( 自然科学版)》 * |
Also Published As
| Publication number | Publication date |
|---|---|
| CN111574466B (en) | 2021-08-03 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN111548320B (en) | 1,3,4-oxadiazole hydrazide compounds and preparation method and application thereof | |
| CN112979627A (en) | Pyrazole bi-1, 2, 4-oxadiazole substituted benzamide compounds and preparation method and application thereof | |
| LU86745A1 (en) | USE OF AROMATIC AND HETEROCYCLIC AMINES AS DRUGS AND AGROCHEMICALS | |
| CN103059010A (en) | 1,4-benzoxazinone-1,2,3-triazole compound having antifungal activity, and its preparation method | |
| CN111574466A (en) | A novel triazole-containing allyl benzoate compound and its preparation method and application | |
| CN108069915B (en) | A kind of pyrazinamide compound and its preparation method and application and a kind of bactericide | |
| CN105541796B (en) | A kind of synthetic method of the triazole Bipyridine compounds of NH 1,2,3 | |
| CN108440506B (en) | Antifungal agent-enol ether compound containing 1,2, 3-triazole structure | |
| KR101663064B1 (en) | Novel N-(2-(azulen-1-yl)-2-substituted vinyl)-sulfonamide derivatives and its preparation | |
| JP2782463B2 (en) | 4-Substituted phenyl-1,2,4-triazolin-5-one derivatives and fungicides for agricultural and horticultural use | |
| CN104961648B (en) | Novel acylhydrazone derivatives containing anthracene and o-phenanthroline, preparation method and application thereof | |
| CN110483405B (en) | Kealiinine derivatives, preparation thereof and application thereof in resisting plant viruses and germs | |
| CN106220633B (en) | A kind of application of 1,2,4- triazole derivatives of the structure of benzopyrazines containing chlorine as fungicide | |
| CN106432245B (en) | A kind of 1,2,4- triazole derivatives of the structure containing benzopyrazines and its preparation method and application | |
| CN106336415B (en) | A kind of 1,2,4- triazole derivatives of chloride benzopyrazines structure and its preparation method and application | |
| CN110590760B (en) | 2,1-Benzisoxazole Derivatives and Their Synthesis and Application | |
| JPS62175460A (en) | Heteroaromatic acetylene useful as antihypertensive | |
| JPH01230562A (en) | Phenyltriazole derivative and insecticide | |
| CN106243110B (en) | A kind of 1,2,4- triazole derivatives of the structure of benzopyrazines containing methoxyl group and its preparation method and application | |
| CN105859645B (en) | A kind of triazole compounds substituted containing phenyl ring and halogen and its production and use | |
| CN106234385B (en) | A kind of application of 1,2,4- triazole derivatives of the structure containing benzopyrazines as fungicide | |
| CN111620827A (en) | Novel allyl ether compound containing triazole as well as preparation method and application thereof | |
| CN110734403A (en) | (S)-2-(1H-pyrazole-4-carboxamido) propyl benzoate compound and its preparation method and application | |
| CN106234372A (en) | A kind of hydrazone compounds containing methoxyl group benzopyrazines structure is as the application of antibacterial | |
| CN106172418B (en) | A kind of application of hydrazone compounds of the structure of benzopyrazines containing chlorine as fungicide |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant | ||
| EE01 | Entry into force of recordation of patent licensing contract | ||
| EE01 | Entry into force of recordation of patent licensing contract |
Application publication date: 20200825 Assignee: Wenzhou Jinhua International Trade Co.,Ltd. Assignor: JIANG University OF TECHNOLOGY Contract record no.: X2023330000144 Denomination of invention: An allyl benzoate compound containing triazole and its preparation method and application Granted publication date: 20210803 License type: Common License Record date: 20230311 |
|
| EE01 | Entry into force of recordation of patent licensing contract |
Application publication date: 20200825 Assignee: Hubei Zhiwei Electric Power Engineering Construction Co.,Ltd. Assignor: JIANG University OF TECHNOLOGY Contract record no.: X2023980035930 Denomination of invention: A triazole containing allyl group benzoate compound and its preparation method and application Granted publication date: 20210803 License type: Common License Record date: 20230525 |
|
| EE01 | Entry into force of recordation of patent licensing contract | ||
| EE01 | Entry into force of recordation of patent licensing contract |
Application publication date: 20200825 Assignee: Guangzhou Fangshao Technology Co.,Ltd. Assignor: JIANG University OF TECHNOLOGY Contract record no.: X2023980036218 Denomination of invention: A triazole containing Allyl group benzoate compound and its preparation method and application Granted publication date: 20210803 License type: Common License Record date: 20230602 |
|
| EE01 | Entry into force of recordation of patent licensing contract | ||
| EE01 | Entry into force of recordation of patent licensing contract |
Application publication date: 20200825 Assignee: Linhai Hongkang Lighting Co.,Ltd. Assignor: JIANG University OF TECHNOLOGY Contract record no.: X2023980047308 Denomination of invention: A triazole containing allyl benzoate compound and its preparation method and application Granted publication date: 20210803 License type: Common License Record date: 20231116 |
|
| EE01 | Entry into force of recordation of patent licensing contract |