CN113953473A - Covering slag for continuous casting crystallizer and preparation method thereof - Google Patents
Covering slag for continuous casting crystallizer and preparation method thereof Download PDFInfo
- Publication number
- CN113953473A CN113953473A CN202111385464.5A CN202111385464A CN113953473A CN 113953473 A CN113953473 A CN 113953473A CN 202111385464 A CN202111385464 A CN 202111385464A CN 113953473 A CN113953473 A CN 113953473A
- Authority
- CN
- China
- Prior art keywords
- parts
- continuous casting
- slag
- covering slag
- mold flux
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 239000002893 slag Substances 0.000 title claims abstract description 60
- 238000009749 continuous casting Methods 0.000 title claims abstract description 21
- 238000002360 preparation method Methods 0.000 title abstract description 10
- 238000005266 casting Methods 0.000 claims abstract description 46
- 239000000843 powder Substances 0.000 claims abstract description 35
- MCMNRKCIXSYSNV-UHFFFAOYSA-N ZrO2 Inorganic materials O=[Zr]=O MCMNRKCIXSYSNV-UHFFFAOYSA-N 0.000 claims abstract description 21
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N silicon dioxide Inorganic materials O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 claims abstract description 18
- 229910052681 coesite Inorganic materials 0.000 claims abstract description 16
- 229910052906 cristobalite Inorganic materials 0.000 claims abstract description 16
- 239000000377 silicon dioxide Substances 0.000 claims abstract description 16
- 229910052682 stishovite Inorganic materials 0.000 claims abstract description 16
- 229910052905 tridymite Inorganic materials 0.000 claims abstract description 16
- KKCBUQHMOMHUOY-UHFFFAOYSA-N Na2O Inorganic materials [O-2].[Na+].[Na+] KKCBUQHMOMHUOY-UHFFFAOYSA-N 0.000 claims abstract description 13
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 claims abstract description 7
- 229910001634 calcium fluoride Inorganic materials 0.000 claims abstract description 7
- 229910052593 corundum Inorganic materials 0.000 claims abstract description 7
- JEIPFZHSYJVQDO-UHFFFAOYSA-N iron(III) oxide Inorganic materials O=[Fe]O[Fe]=O JEIPFZHSYJVQDO-UHFFFAOYSA-N 0.000 claims abstract description 7
- 229910001845 yogo sapphire Inorganic materials 0.000 claims abstract description 7
- 230000004907 flux Effects 0.000 claims description 35
- 238000002156 mixing Methods 0.000 claims description 5
- 239000002245 particle Substances 0.000 claims description 5
- 238000000227 grinding Methods 0.000 claims description 3
- 238000000034 method Methods 0.000 claims description 3
- 230000001681 protective effect Effects 0.000 claims description 3
- 238000001035 drying Methods 0.000 claims description 2
- 238000003801 milling Methods 0.000 claims description 2
- 238000004519 manufacturing process Methods 0.000 claims 4
- 238000009413 insulation Methods 0.000 abstract description 5
- 230000000694 effects Effects 0.000 abstract description 4
- 229910001208 Crucible steel Inorganic materials 0.000 abstract description 2
- 239000007788 liquid Substances 0.000 description 19
- 229910000831 Steel Inorganic materials 0.000 description 17
- 239000010959 steel Substances 0.000 description 17
- 230000000052 comparative effect Effects 0.000 description 16
- 238000012546 transfer Methods 0.000 description 13
- FUJCRWPEOMXPAD-UHFFFAOYSA-N Li2O Inorganic materials [Li+].[Li+].[O-2] FUJCRWPEOMXPAD-UHFFFAOYSA-N 0.000 description 10
- 239000002994 raw material Substances 0.000 description 8
- 230000009286 beneficial effect Effects 0.000 description 6
- 239000006229 carbon black Substances 0.000 description 5
- 230000002349 favourable effect Effects 0.000 description 4
- 238000002844 melting Methods 0.000 description 4
- 230000008018 melting Effects 0.000 description 4
- 239000000126 substance Substances 0.000 description 4
- 238000012360 testing method Methods 0.000 description 4
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 4
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 3
- 238000013329 compounding Methods 0.000 description 3
- 238000001816 cooling Methods 0.000 description 3
- 230000001050 lubricating effect Effects 0.000 description 3
- 238000005461 lubrication Methods 0.000 description 3
- 230000003647 oxidation Effects 0.000 description 3
- 238000007254 oxidation reaction Methods 0.000 description 3
- RVTZCBVAJQQJTK-UHFFFAOYSA-N oxygen(2-);zirconium(4+) Chemical compound [O-2].[O-2].[Zr+4] RVTZCBVAJQQJTK-UHFFFAOYSA-N 0.000 description 3
- 229910001928 zirconium oxide Inorganic materials 0.000 description 3
- 238000002425 crystallisation Methods 0.000 description 2
- 230000008025 crystallization Effects 0.000 description 2
- XUCJHNOBJLKZNU-UHFFFAOYSA-M dilithium;hydroxide Chemical compound [Li+].[Li+].[OH-] XUCJHNOBJLKZNU-UHFFFAOYSA-M 0.000 description 2
- 239000010439 graphite Substances 0.000 description 2
- 229910002804 graphite Inorganic materials 0.000 description 2
- 239000012535 impurity Substances 0.000 description 2
- 239000000463 material Substances 0.000 description 2
- 229910052664 nepheline Inorganic materials 0.000 description 2
- 239000010434 nepheline Substances 0.000 description 2
- 230000008569 process Effects 0.000 description 2
- 239000011435 rock Substances 0.000 description 2
- 238000003756 stirring Methods 0.000 description 2
- 238000010998 test method Methods 0.000 description 2
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- BPQQTUXANYXVAA-UHFFFAOYSA-N Orthosilicate Chemical compound [O-][Si]([O-])([O-])[O-] BPQQTUXANYXVAA-UHFFFAOYSA-N 0.000 description 1
- WUKWITHWXAAZEY-UHFFFAOYSA-L calcium difluoride Chemical compound [F-].[F-].[Ca+2] WUKWITHWXAAZEY-UHFFFAOYSA-L 0.000 description 1
- 229910052799 carbon Inorganic materials 0.000 description 1
- 239000000498 cooling water Substances 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- 230000007797 corrosion Effects 0.000 description 1
- 238000005260 corrosion Methods 0.000 description 1
- 238000005336 cracking Methods 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- 239000000203 mixture Substances 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- GALOTNBSUVEISR-UHFFFAOYSA-N molybdenum;silicon Chemical compound [Mo]#[Si] GALOTNBSUVEISR-UHFFFAOYSA-N 0.000 description 1
- SIWVEOZUMHYXCS-UHFFFAOYSA-N oxo(oxoyttriooxy)yttrium Chemical compound O=[Y]O[Y]=O SIWVEOZUMHYXCS-UHFFFAOYSA-N 0.000 description 1
- 238000011056 performance test Methods 0.000 description 1
- 239000011819 refractory material Substances 0.000 description 1
- 239000000523 sample Substances 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 230000002195 synergetic effect Effects 0.000 description 1
- 238000004017 vitrification Methods 0.000 description 1
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B22—CASTING; POWDER METALLURGY
- B22D—CASTING OF METALS; CASTING OF OTHER SUBSTANCES BY THE SAME PROCESSES OR DEVICES
- B22D11/00—Continuous casting of metals, i.e. casting in indefinite lengths
- B22D11/10—Supplying or treating molten metal
- B22D11/11—Treating the molten metal
- B22D11/111—Treating the molten metal by using protecting powders
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02P—CLIMATE CHANGE MITIGATION TECHNOLOGIES IN THE PRODUCTION OR PROCESSING OF GOODS
- Y02P10/00—Technologies related to metal processing
- Y02P10/20—Recycling
Landscapes
- Engineering & Computer Science (AREA)
- Mechanical Engineering (AREA)
- Continuous Casting (AREA)
- Treatment Of Steel In Its Molten State (AREA)
Abstract
The application relates to the field of cast steel, and particularly discloses a covering slag for a continuous casting crystallizer and a preparation method thereof. The continuous casting crystallizer casting powder comprises the following components of SiO224.00-31.00 parts of CaO26.00-33.00 parts of Al2O35.40-5.70 parts of Fe2O3Not more than 3.00 parts of MgO not more than 3.00 parts of Na2O and Li26.50-10.50 parts of O and CaF23.50-7.50 parts of ZrO24.5-5.5 parts of Y2O33.5-4.5 parts; the preparation method comprises the following steps: the components are uniformly mixed and ground to obtain the finished product of the covering slag powder slag, and the covering slag powder slag has the advantage of good heat insulation effect.
Description
Technical Field
The application relates to the field of cast steel, in particular to covering slag for a continuous casting crystallizer and a preparation method thereof.
Background
The crystallizer covering slag is a functional material which influences the quality of a casting blank and the stability of a continuous casting process, and during the casting process of molten steel, granular or powdery covering slag is continuously added on the liquid level of the molten steel of the crystallizer, so that the covering slag forms a three-layer structure comprising a liquid slag layer, a sintered layer and a powder slag layer due to the high temperature of the molten steel. The basic functions of the mold flux are as follows: (1) the exposed molten steel is insulated and thermally insulated; (2) secondary oxidation of molten steel is prevented; (3) absorbing impurities floating to the surface of molten steel; (4) and a liquid film is filled between the wall of the crystallizer and the shell of the casting billet to play a role of lubrication (5) for controlling the heat transfer of the casting billet to the crystallizer. The casting powder can effectively solve the problems of longitudinal cracking, sinking, steel leakage and the like on the surface of the steel billet.
At present, the heat insulation effect of the casting powder is not good enough, so that the casting blank is longitudinally cracked due to the fact that the outer wall of the crystallizer is cooled too fast to the casting blank, the phenomenon of affecting the quality of the steel blank is very common, the heat transfer of the casting powder can be effectively controlled by increasing carbon black, the proportion of the added carbon black is not easy to calculate, the proportion of the carbon black is too high, the carbon black easily enters the casting blank, the carbon content of the surface of the casting blank is increased, the quality of the casting blank is affected, the proportion of the carbon black is too low, and the heat transfer process is difficult to control.
Disclosure of Invention
In order to improve the heat insulation effect of the covering slag, the application provides the continuous casting crystallizer covering slag and the preparation method thereof.
The continuous casting crystallizer covering slag and the preparation method thereof adopt the following technical scheme:
in a first aspect, a mold flux for a continuous casting mold comprises the following composition SiO224.00-31.00 parts of CaO26.00-33.00 parts of Al2O35.40-5.70 parts of Fe2O3Not more than 3.00 parts of MgO not more than 3.00 parts of Na2O and Li26.50-10.50 parts of O and CaF23.50-7.50 parts of ZrO24.5-5.5 parts of Y2O33.5 to 4.5 portions.
By adopting the technical scheme, the zirconia has the advantages of high melting point, difficult oxidation and the like, has stable chemical property, good heat insulation effect, corrosion resistance and oxidation resistance under specific high-temperature environment, is non-volatile and pollution-free, is a refractory material with good performance,
when the mold flux is added to the liquid level of the molten steel, the mold flux is melted due to the high temperature of the molten steel, and the mold flux forms a three-layer structure, namely a liquid slag layer, a sintered layer and a powder slag layer. Because the crystallizer rocks, make and form the gap between casting blank and the crystallizer wall, the liquid slag layer flows into between casting blank and the crystallizer wall, play the role of lubricating and controlling heat transfer, because add zirconia in the covering slag, zirconia is high temperature resistant, chemical stability, and the heat-proof quality is good, can be better reduce the heat transfer efficiency of covering slag, reduce because the casting blank cooling rate is too fast, thus make the casting blank produce the cracked possibility, in addition, still avoided the phenomenon that the covering slag carburizes to the casting blank surface.
The zirconium oxide is added into the covering slag, the heat transfer efficiency of the covering slag is well controlled, but the zirconium oxide increases the viscosity of a liquid slag layer of the covering slag, and the liquid slag layer is not beneficial to permeating into a gap between a crystallizer and a casting blank, so that the casting blank is not easy to pull out from the crystallizer, and even a viscous breakout accident is caused.
The zirconium oxide and the yttrium oxide have synergistic effect, so that the heat transfer performance of the covering slag is improved, the viscosity of liquid covering slag is not weakened, and the possibility of generating cracks in a casting blank is reduced.
Alternatively, the Na2O and Li2The weight ratio of 0 is 1:1-2: 1.
By adopting the technical scheme, Na2O and Li2O can destroy the chain structure of the casting powder, reduce the viscosity of the liquid slag layer of the casting powder, and simultaneously reduce the crystallization temperature, Na2O and Li20 is more than 2:1, the casting powder is easy to separate out nepheline and is not beneficial to lubrication, and Na2O and Li20 is less than 1:1 by weight, affecting the mold fluxCrystallization rate and vitrification degree.
Optionally, the CaO and SiO2In a weight ratio of 0.95: 1-1.15:1.
By adopting the technical scheme, CaO and SiO2The alkalinity is an important index of the capability of reacting the covering slag to absorb inclusions in the molten steel, and the alkalinity is properly improved, so that the covering slag is favorable for absorbing the inclusions in the molten steel.
On the other hand, the preparation method of the continuous casting crystallizer casting powder is to prepare SiO224.00-31.00 parts of CaO26.00-33.00 parts of Al2O35.40-5.70 parts of Fe2O3Not more than 3.00 parts of MgO not more than 3.00 parts of Na2O and Li26.50-10.50 parts of O and CaF23.50-7.50 parts of ZrO24.5-5.5 parts of Y2O33.5 to 4.5 portions of the powder slag of the finished product of the covering slag are obtained after uniform mixing and grinding.
Optionally, the finished mold flux powder is dried.
By adopting the technical scheme, the content of water in the casting powder is reduced, so that the quality of a casting blank is not influenced.
Optionally, the milling process uses a ball mill.
Optionally, the particle size of the finished product of the covering slag powder is 100-200 meshes.
By adopting the technical scheme, the particle density is fine, which is beneficial to melting the casting powder, but the particle density is too fine, so that the liquid film is not beneficial to controlling the heat transfer temperature after passing.
Optionally, the finished mold flux is stored in a dry environment.
In summary, the present application has the following beneficial effects:
1. when the mold flux is added to the liquid level of the molten steel, the mold flux is melted due to the high temperature of the molten steel, and the mold flux forms a three-layer structure, namely a liquid slag layer, a sintered layer and a powder slag layer. Because the crystallizer rocks, make and form the gap between casting blank and the crystallizer wall, the liquid slag layer flows into between casting blank and the crystallizer wall, play the role of lubricating and controlling heat transfer, because add zirconia in the covering slag, zirconia is high temperature resistant, chemical stability, and the heat-proof quality is good, can be better reduce the heat transfer efficiency of covering slag, reduce because the casting blank cooling rate is too fast, thus make the casting blank produce the cracked possibility, in addition, still avoided the phenomenon that the covering slag carburizes to the casting blank surface.
2. CaO and SiO2The alkalinity is an important index of the capability of reacting the covering slag to absorb inclusions in the molten steel, and the alkalinity is properly improved, so that the covering slag is favorable for absorbing the inclusions in the molten steel.
Detailed Description
The present application will be described in further detail with reference to examples.
Examples
Example 1
The continuous casting crystallizer casting powder comprises the following components of SiO224.00kg、CaO26.00kg、Al2O35.40kg、Fe2O33.00kg、MgO3.00kg、Na2O and Li206.5kg、CaF23.50-7.50 parts of ZrO24.5-5.5 parts of Y2O33.5-4.5 parts of Na2O and Li2The weight ratio of 0 is 1: 1.
A preparation method of continuous casting crystallizer casting powder comprises the following steps: mixing 24.00kgSiO2、26.00kgCaO、5.40kgAl2O3、3.00kgFe2O3、3.00kgMgO、3.50kgCaF2、4.5kgZrO2、3.5kgY2O3、6.50kgNa2O and Li20, uniformly mixing, grinding by using a ball mill, and drying to obtain the finished product of the covering slag powder, wherein the particle size of the finished product of the covering slag powder corresponds to 100 meshes, and Na is contained in the powder2O and Li20 weight ratio of 1:1, CaO and SiO2The weight ratio of (1.08): 1.
examples 2 to 9 and comparative examples 1 to 2
Examples 2 to 9 and comparative examples 1 to 2 are different from example 1 in the ratio of raw materials, and specific ratios of raw materials are shown in tables 1 to 1 and tables 1 to 2.
TABLE 1-1 raw material ratios of examples 2-9 and comparative examples 1-2
Tables 1-1 the mass of each example was set to 1kg and the remaining data were homogeneously expressed as follows:
TABLE 1-2 raw material compounding ratio of examples 2-9 and comparative examples 1-2
Performance test
Viscosity test: and testing the viscosity of the casting powder according to a standard YB/T185-2017 continuous casting powder viscosity test method.
The heat transfer efficiency test method comprises the following steps: and testing the heat flux density of the casting powder by using an HF-200 crystallizer slag film heat flux simulator.
The heat flow test slag film preparation step: at 1300 ℃, 350g of prepared slag materials are uniformly mixed and then put into a graphite crucible, the graphite crucible is added into a silicon-molybdenum furnace, the temperature is raised, the melting and the stirring are uniform, after the temperature of a hearth reaches 1400 ℃, a stirring rod is used for uniformly melting protective slag, a lifting arm is started after the position of a sensor is positioned at the center of the furnace tube to drive a positioning sensor to descend to the slag liquid level, and the sensor can immediately and automatically ascend when contacting the liquid level. And the lifting arm descends again to drive the copper probe to be immersed into the slag liquid, the sensor starts timing, and the sensor below the slag liquid surface is taken out after 80s to obtain a solid liquid film adhered to the sensor and the water temperature of the inlet and the outlet automatically acquired by the computer.
The heat flux density of the slag film is calculated as follows: phi W C delta T/(F1000)
Phi-heat flow density, Mw m-2(ii) a Cooling water flow rate of W-heat flow sensor, kg.s-1(ii) a Delta T-temperature difference of water at the inlet and the outlet of the heat flow sensor, DEG C; effective heat transfer area, m, of F-heat flow sensor2(ii) a C-specific Heat of Water, kg · (kg ℃)-1。
The following table shows the viscosity and heat flow density for examples 2-9 and comparative examples 1-2:
TABLE 2 viscosity vs. Heat flow Density for examples 2-9 and comparative examples 1-2
Example 2 improved SiO compared to example 12Content of (3), data show, SiO2In a specific range, SiO2The content is increased, so that the protective slag forms a glass phase, which is beneficial to lubricating a casting blank;
compared with the embodiment 1, the embodiment 3 improves the content of CaO, and the data show that the content of CaO is in a specific range, the content of CaO is improved, the viscosity of the casting powder is obviously reduced, and the capacity of absorbing impurities is improved;
al in example 4 in comparison with example 12O3The content is unchanged, and the viscosity is unchanged;
fe in example 5 compared to example 12O3While the viscosity of the mold flux was not changed, it was confirmed that the content of (B) in Fe was reduced2O3The content of (b) does not affect the viscosity of the mold flux within a specific range;
compared with the embodiment 1, the MgO content in the embodiment 6 is reduced, the viscosity of the mold flux is increased, the MgO content is in a specific range, the MgO can reduce the viscosity and the solidifying point of the mold flux, the fluidity of the mold flux is increased, and the chemical stability of the mold flux is favorable;
CaF in example 6 in comparison with example 12The content is reduced, the viscosity of the casting powder is increased, and CaF is increased within a specific content range2The content of (3) can reduce the viscosity of the mold flux;
example 1 and examples 7 to 8 in combination with comparative examples 1 to 2 found that ZrO2In a specific range, ZrO2Increase in the content ofAnd the viscosity of the casting powder is increased, the heat flow density of the casting powder is reduced, the heat insulation performance of the casting powder is improved, and the possibility that the casting blank cracks due to the fact that the cooling speed of the casting blank is too high is reduced.
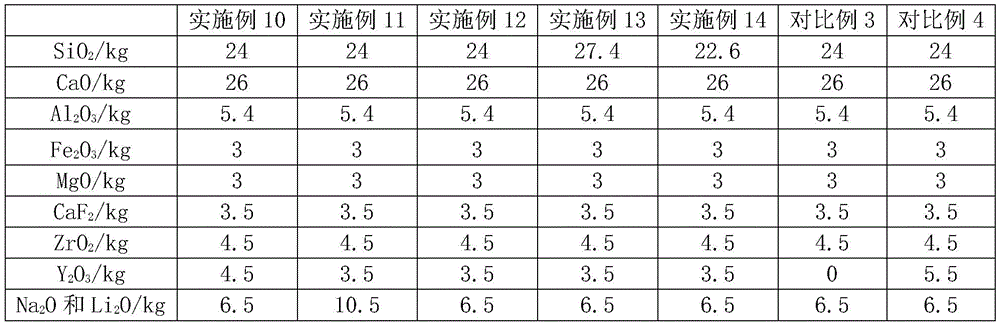
Examples 10 to 14 and comparative examples 3 to 4
Examples 10 to 14 and comparative examples 3 to 4 are different from example 1 in the ratio of raw materials, and specific ratios of raw materials are shown in tables 3 to 1 and 3 to 2.
TABLE 3-1 raw material compounding ratios of examples 10 to 14 and comparative examples 3 to 4
The mass of each example is set to 1kg in tables 3-1 and the remaining data are homogeneously expressed as follows:
TABLE 3-2 raw material compounding ratios of examples 10 to 14 and comparative examples 3 to 4
| Example 10 | Example 11 | Example 12 | Example 13 | Example 14 | Comparative example 3 | Comparative example 4 | |
| SiO2/kg | 0.30 | 0.29 | 0.30 | 0.33 | 0.29 | 0.32 | 0.29 |
| CaO/kg | 0.32 | 0.31 | 0.33 | 0.31 | 0.33 | 0.34 | 0.32 |
| Al2O3/kg | 0.07 | 0.06 | 0.07 | 0.07 | 0.07 | 0.07 | 0.07 |
| Fe2O3/kg | 0.04 | 0.04 | 0.04 | 0.04 | 0.04 | 0.04 | 0.04 |
| MgO/kg | 0.04 | 0.04 | 0.04 | 0.04 | 0.04 | 0.04 | 0.04 |
| CaF2/kg | 0.04 | 0.04 | 0.04 | 0.04 | 0.04 | 0.05 | 0.04 |
| ZrO2/kg | 0.06 | 0.05 | 0.06 | 0.05 | 0.06 | 0.06 | 0.06 |
| Y2O3/kg | 0.06 | 0.04 | 0.04 | 0.04 | 0.04 | 0.00 | 0.07 |
| Na2O and Li2O/kg | 0.08 | 0.13 | 0.08 | 0.08 | 0.08 | 0.09 | 0.08 |
| Na2O/Li20 | 1 | 1 | 2 | 1 | 1 | 1 | 1 |
| CaO/SiO2 | 1.08 | 1.08 | 1.08 | 0.95 | 1.15 | 1.08 | 1.08 |
The following table shows the viscosity and heat flow density for examples 10-14 and comparative examples 3-4:
TABLE 4 viscosity vs. Heat flow Density for examples 10-14 and comparative examples 3-4
Example 10 increased Y compared to example 12O3In a specific content range, Y is increased2O3The viscosity of the mold flux is reduced, and the heat flux density of the mold flux is reduced;
example 1 and example 10 in combination with comparative examples 3 to 4, it was found that within a specific content range, Y2O3The higher the content of (A), the lower the viscosity of the mold flux, and with ZrO2In cooperation, the heat flux density of the mold flux is reduced, that is, the heat insulating property of the mold flux is synergistically increased.
Example 11 increased Na over example 12O and Li20 content of Na in the mold flux2O and Li20 can destroy the network structure of the silicate, and can reduce the viscosity of the mold flux within a specific content range.
Example 12 increased Na over example 12O/Li20, in a specific content range, Na2The increase in O content increases the tendency to crystallize, and nepheline is likely to precipitate, which is not favorable for lubrication.
In comparison with example 1, examples 13 to 14 show that in the specified content range, CaO/SiO2The ratio of (2) is increased, namely the alkalinity is increased, the crystallization is improved, and the heat transfer is reduced.
The present embodiment is only for explaining the present application, and it is not limited to the present application, and those skilled in the art can make modifications of the present embodiment without inventive contribution as needed after reading the present specification, but all of them are protected by patent law within the scope of the claims of the present application.
Claims (8)
1. The covering slag for the continuous casting crystallizer is characterized by comprising the following components of SiO224.00-31.00 parts of CaO26.00-33.00 parts of Al2O35.40-5.70 parts of Fe2O3Not more than 3.00 parts of MgO not more than 3.00 parts of Na2O and Li2O6.50-10.50 part of CaF23.50-7.50 parts of ZrO24.5-5.5 parts of Y2O33.5 to 4.5 portions.
2. The mold flux for a continuous casting crystallizer according to claim 1, characterized in that: the Na is2O and Li2The weight ratio of 0 is 1:1-2: 1.
3. The mold flux for a continuous casting crystallizer according to claim 1, characterized in that: the CaO and SiO2In a weight ratio of 0.95: 1-1.15:1.
4. A method for preparing the mold flux for the continuous casting mold according to any one of claims 1 to 3, characterized in that: mixing SiO224.00-31.00 parts of CaO26.00-33.00 parts of Al2O35.40-5.70 parts of Fe2O3Not more than 3.00 parts of MgO not more than 3.00 parts of Na2O and Li26.50-10.50 parts of O and CaF23.50-7.50 parts of ZrO24.5-5.5 parts of Y2O33.5 to 4.5 portions of the powder slag of the finished product of the covering slag are obtained after uniform mixing and grinding.
5. The method for producing the mold flux for a continuous casting crystallizer according to claim 4, characterized in that: and drying the finished product of the covering slag powder slag.
6. The method for producing the mold flux for a continuous casting crystallizer according to claim 4, characterized in that: the milling process uses a ball mill.
7. The method for producing the mold flux for a continuous casting crystallizer according to claim 4, characterized in that: the particle size of the finished product of the protective slag powder is 100 meshes and 200 meshes.
8. The method for producing the mold flux for a continuous casting crystallizer according to claim 4, characterized in that: and storing the finished product of the casting powder in a dry environment.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202111385464.5A CN113953473B (en) | 2021-11-22 | 2021-11-22 | Covering slag for continuous casting crystallizer and preparation method thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202111385464.5A CN113953473B (en) | 2021-11-22 | 2021-11-22 | Covering slag for continuous casting crystallizer and preparation method thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN113953473A true CN113953473A (en) | 2022-01-21 |
| CN113953473B CN113953473B (en) | 2022-12-30 |
Family
ID=79471321
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202111385464.5A Active CN113953473B (en) | 2021-11-22 | 2021-11-22 | Covering slag for continuous casting crystallizer and preparation method thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN113953473B (en) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN115647313A (en) * | 2022-11-14 | 2023-01-31 | 鞍钢股份有限公司 | A ladle covering agent suitable for straight up SPHC steel grades and its application method |
| CN117340210A (en) * | 2023-09-27 | 2024-01-05 | 南京钢铁股份有限公司 | High basicity continuous casting covering slag for high-alumina steel |
| CN120624755A (en) * | 2025-08-13 | 2025-09-12 | 洛阳科丰冶金新材料有限公司 | A special protective slag for electromagnetic ejection of duplex stainless steel |
Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH03193248A (en) * | 1989-12-25 | 1991-08-23 | Sumitomo Metal Ind Ltd | Mold powder for continuously casting steel |
| KR19990050955A (en) * | 1997-12-17 | 1999-07-05 | 이구택 | Zirconia-containing mold flux for continuous casting of medium carbon steel |
| JP2006247735A (en) * | 2005-03-14 | 2006-09-21 | Sumitomo Metal Ind Ltd | Mold powder for continuous casting of steel |
| CN102006950A (en) * | 2009-07-07 | 2011-04-06 | 住友金属工业株式会社 | Mold flux for continuous casting of steel and continuous casting method of steel using the same |
| CN104607607A (en) * | 2014-12-31 | 2015-05-13 | 中南大学 | Fluoride-free mould flux for continuous casting of zirconium-containing medium-carbon steel |
| CN107498014A (en) * | 2017-09-06 | 2017-12-22 | 中南大学 | One kind contains ZrO2Automobile using TWIP steel covering slag and its application |
| CN109095896A (en) * | 2018-08-21 | 2018-12-28 | 北京利尔高温材料股份有限公司 | A kind of ladle slag line brick of useless submersed nozzle slag line material pug |
| CN113084105A (en) * | 2021-03-31 | 2021-07-09 | 重庆大学 | Low-reactivity casting powder containing yttrium oxide for high-aluminum steel |
-
2021
- 2021-11-22 CN CN202111385464.5A patent/CN113953473B/en active Active
Patent Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH03193248A (en) * | 1989-12-25 | 1991-08-23 | Sumitomo Metal Ind Ltd | Mold powder for continuously casting steel |
| KR19990050955A (en) * | 1997-12-17 | 1999-07-05 | 이구택 | Zirconia-containing mold flux for continuous casting of medium carbon steel |
| JP2006247735A (en) * | 2005-03-14 | 2006-09-21 | Sumitomo Metal Ind Ltd | Mold powder for continuous casting of steel |
| CN102006950A (en) * | 2009-07-07 | 2011-04-06 | 住友金属工业株式会社 | Mold flux for continuous casting of steel and continuous casting method of steel using the same |
| CN104607607A (en) * | 2014-12-31 | 2015-05-13 | 中南大学 | Fluoride-free mould flux for continuous casting of zirconium-containing medium-carbon steel |
| CN107498014A (en) * | 2017-09-06 | 2017-12-22 | 中南大学 | One kind contains ZrO2Automobile using TWIP steel covering slag and its application |
| CN109095896A (en) * | 2018-08-21 | 2018-12-28 | 北京利尔高温材料股份有限公司 | A kind of ladle slag line brick of useless submersed nozzle slag line material pug |
| CN113084105A (en) * | 2021-03-31 | 2021-07-09 | 重庆大学 | Low-reactivity casting powder containing yttrium oxide for high-aluminum steel |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN115647313A (en) * | 2022-11-14 | 2023-01-31 | 鞍钢股份有限公司 | A ladle covering agent suitable for straight up SPHC steel grades and its application method |
| CN115647313B (en) * | 2022-11-14 | 2023-08-18 | 鞍钢股份有限公司 | Ladle covering agent suitable for SPHC steel grade straight up and use method |
| CN117340210A (en) * | 2023-09-27 | 2024-01-05 | 南京钢铁股份有限公司 | High basicity continuous casting covering slag for high-alumina steel |
| CN120624755A (en) * | 2025-08-13 | 2025-09-12 | 洛阳科丰冶金新材料有限公司 | A special protective slag for electromagnetic ejection of duplex stainless steel |
| CN120624755B (en) * | 2025-08-13 | 2025-11-18 | 洛阳科丰冶金新材料有限公司 | Special covering slag for electromagnetic ejection duplex stainless steel |
Also Published As
| Publication number | Publication date |
|---|---|
| CN113953473B (en) | 2022-12-30 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN113953473B (en) | Covering slag for continuous casting crystallizer and preparation method thereof | |
| CN100584487C (en) | A preparation method of pre-melted continuous casting mold powder | |
| CN106270429B (en) | Hypoergia covering slag and preparation method | |
| JP7272477B2 (en) | Mold powder for continuous casting of Al-containing hypo-peritectic steel and continuous casting method | |
| JP6674093B2 (en) | Mold powder for continuous casting of steel and continuous casting method | |
| CN104923754A (en) | High-drawing-speed sheet billet peritectic steel continuous casting crystallizer casting powder and preparation method thereof | |
| CN101758174A (en) | Mold powder capable of effectively controlling peritectic steel thick slab narrow face depression defect | |
| CN101612654A (en) | The high basicity covering slag of the inferior peritectic steel plate slab crystallizer of carbon heat flux uniformity in the control | |
| JP4337748B2 (en) | Mold powder for continuous casting of steel | |
| CN102990025A (en) | Medium carbon steel casting powder for H-section steel continuous casting | |
| CN102652990B (en) | Casting powder for medium carbon peritectic steel continuous casting crystallizer | |
| JP3179358B2 (en) | Mold powder for continuous casting | |
| CN102304634B (en) | Refining agent for smelting tin bronze alloy | |
| CN114535524A (en) | Covering agent for semi-continuous casting crystallizer for copper-iron alloy | |
| CN113996771A (en) | Pre-melted crystallizer casting powder for non-oriented silicon steel and preparation method thereof | |
| CN113714477A (en) | Formula and production process of low-carbon covering slag containing titanium stainless steel | |
| CN101612652A (en) | Continuous casting mold flux for low-nickel and high-manganese stainless steel and preparation method thereof | |
| JP3610885B2 (en) | Mold powder and continuous casting method | |
| CN116422847A (en) | Low-reactivity continuous casting mold flux for rare earth steel with high rare earth content and preparation method thereof | |
| JP2000051998A (en) | Method for continuously casting lead-containing steel | |
| CN114769533B (en) | Functional protection material of continuous casting crystallizer for industrial pure iron | |
| JP2000218348A (en) | Mold powder for continuous casting and continuous casting method | |
| CN110560649B (en) | A New Type of High Alumina Steel Molding Slag and Its Application | |
| CN115870464B (en) | A high iron copper alloy continuous casting crystallizer protection slag and its preparation method and application | |
| JP7716005B2 (en) | Exothermic mold powder |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |