EP0061209B1 - Analogues de prostaglandines et leur procédé de préparation - Google Patents
Analogues de prostaglandines et leur procédé de préparation Download PDFInfo
- Publication number
- EP0061209B1 EP0061209B1 EP82200192A EP82200192A EP0061209B1 EP 0061209 B1 EP0061209 B1 EP 0061209B1 EP 82200192 A EP82200192 A EP 82200192A EP 82200192 A EP82200192 A EP 82200192A EP 0061209 B1 EP0061209 B1 EP 0061209B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- reaction
- compound
- olefin
- epoxy
- bond
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C405/00—Compounds containing a five-membered ring having two side-chains in ortho position to each other, and having oxygen atoms directly attached to the ring in ortho position to one of the side-chains, one side-chain containing, not directly attached to the ring, a carbon atom having three bonds to hetero atoms with at the most one bond to halogen, and the other side-chain having oxygen atoms attached in gamma-position to the ring, e.g. prostaglandins ; Analogues or derivatives thereof
- C07C405/0008—Analogues having the carboxyl group in the side-chains replaced by other functional groups
- C07C405/0016—Analogues having the carboxyl group in the side-chains replaced by other functional groups containing only hydroxy, etherified or esterified hydroxy groups
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C405/00—Compounds containing a five-membered ring having two side-chains in ortho position to each other, and having oxygen atoms directly attached to the ring in ortho position to one of the side-chains, one side-chain containing, not directly attached to the ring, a carbon atom having three bonds to hetero atoms with at the most one bond to halogen, and the other side-chain having oxygen atoms attached in gamma-position to the ring, e.g. prostaglandins ; Analogues or derivatives thereof
- C07C405/0008—Analogues having the carboxyl group in the side-chains replaced by other functional groups
- C07C405/0025—Analogues having the carboxyl group in the side-chains replaced by other functional groups containing keto groups
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D303/00—Compounds containing three-membered rings having one oxygen atom as the only ring hetero atom
- C07D303/02—Compounds containing oxirane rings
- C07D303/12—Compounds containing oxirane rings with hydrocarbon radicals, substituted by singly or doubly bound oxygen atoms
- C07D303/14—Compounds containing oxirane rings with hydrocarbon radicals, substituted by singly or doubly bound oxygen atoms by free hydroxyl radicals
Definitions
- This invention relates to a process of making a novel class of prostaglandin-like compounds.
- a key feature of this synthesis is the trans opening of the oxirane by nucleophilic attack.
- this synthesis solves the problem of the trans substitution at the 12-position, it creates the new problem of isomer selective synthesis of all cis 3,5-dihydroxy-1,2-epoxycyclopentane.
- prostaglandin-type compounds (V) have been synthesized from 1,2-epoxycyclopentane (IV).
- this further deviation from the natural prostaglandin structure results in an erratic biological activity of the compounds thus obtained; some of them act as prostaglandin agonists in one test, and as antagonists in another.
- (VI) is synthesized by reacting cyclopenten-3-ol, a relatively expensive starting material, with t-butyl hydroperoxide in the presence of a vanadium catalyst. Typically a 45% isolated yield is obtained after 2 days. The reaction time is less (about 1 day) if the t-butyl hydroperoxide is replaced with a peracid, but then a significant amount of the trans isomer is formed (cis:trans ratio about 4:1).
- olefins can quite readily be converted to the corresponding alpha-epoxy alcohols when they are reacted with singlet oxygen in the presence of a suitable oxidation catalyst.
- the reaction is fast, gives high yields, has a high selectivity towards the epoxy alcohol, and uses an inexpensive starting material.
- the present invention is a process for the photo-oxidative conversion of olefins to alpha-epoxy alcohols using a vanadium catalyst.
- the present invention also encompasses a process for making prostaglandin analogues using cyclopentene as a relatively inexpensive starting material.
- the first step in this process is the conversion of cyclopentene to cis-2,3-epoxycyclopentan-1-ol by reacting cyclopentene with singlet oxygen in the presence of a catalytic amount of a catalyst containing a transition metal of group IV B, V B, or VI B of the Periodic Table, excluding chromium.
- Application of this process to the conversion of other olefins to the corresponding alpha-epoxy alcohols is also within the scope of this invention.
- the next step is a nucleophilic attack on the oxirane bond with an alkynyl-alane reagent, resulting in a regio-selective trans opening.
- an alkynyl-alane reagent Prior to this reaction, the hydroxyl group is protected against nucleophilic attack with a suitable protecting group.
- the substituent at the 8-position is attached via a Williamson synthesis. Deprotection and tailoring of the oxidation states at the 9-position and in the substituent chains leads to the desired prostaglandin analogues, as described more fully hereinafter.
- the present invention encompasses a new class of prostaglandin analogues which are characterized by an oxygen atom replacing the methylene group at the 7-position, and the absence of a hydroxyl substituent at the 11-position.
- Members of this class have been found to possess cytoprotective properties which make them useful for the prevention and treatment of gastric ulcers.
- olefin herein is meant an aliphatic hydrocarbon having at least one double bond and at least one allylic hydrogen.
- mono-olefin herein is meant an olefin with one double bond.
- corresponding alpha epoxy alcohol herein is meant that when the olefin starting material has its double bond between the carbon atoms m and m+1, the alpha-epoxy alcohol formed is the m-hydroxy-(m+1), (m+2)-epoxy compound or the (m+1)-hydroxy-(m-1), m epoxy compound.
- heterogeneous catalyst herein is meant a catalyst which is in the solid state and is not soluble in the reaction mixture.
- homogeneous catalyst herein is meant a catalyst which is soluble in the reaction mixture.
- acetylacetonate acetylacetonate
- stream oxygen herein is meant oxygen molecules in the lowest excited electronic state that has spin multiplicity of one, generally denoted as the 'Ag state.
- photo-sensitizer herein is meant an organic compound which can be excited to the triplet state by adsorption of visible light.
- catalytic amount herein is meant an amount significantly less than a stoichiometric amount which is sufficient to act as a catalyst in the reaction.
- prostaglandin analogue herein is meant a compound which is structurally similar to the naturally occurring prostaglandins.
- 7-oxa-11-deoxy prostaglandin analogue herein is meant a compound structurally similar to the natural prostaglandins of the E-series or F-series, characterized by an oxa group replacing the methylene group at the 7-position, and by the absence of a hydroxyl substituent at the 11-position.
- suitable protecting group herein is meant any base stable protecting group for alcohols.
- suitable protecting group examples thereof are ethers, in particular methoxymethyl, t-butyl, benzyl, dimethyl-t-butylsilyl and methylthiomethyl ether. Protection of alcoholic hydroxyl groups is discussed by Reese, in “Protective Groups in Organic Chemistry", McOmie, editor, Plenum Press (1973) p. 95 et seq., which is incorporated herein by reference.
- the hydroxyl group can be deprotected selectively by reaction with tetrabutyl ammonium fluoride. The procedure is described by E. J. Corey, et al., J. Am. Chem. Soc., 94, 6190 (1972), incorporated herein by reference.
- alkynyl alane reagent herein is meant the dialkylalkynylaluminum compound formed by reaction of dialkylaluminum chloride with a lithio-1-alkyne.
- the first aspect of this invention is a process for converting olefins to the corresponding alpha-epoxy alcohols comprising reacting an olefin with singlet oxygen and converting the reaction product to an alpha-epoxy alcohol by in situ rearrangement in the presence of a catalytic amount of a heterogeneous or homogeneous catalyst containing a transition metal of group IVB, VB or VIB of the Periodic Table, excluding chromium.
- a heterogeneous or homogeneous catalyst containing a transition metal of group IVB, VB or VIB of the Periodic Table, excluding chromium.
- the present oxidation process has general applicability to all olefins capable of reaction with singlet oxygen, that is, alkyl substituted olefins having at least one allylic hydrogen.
- alkyl substituted olefins having at least one allylic hydrogen Preferred herein are dialkyl substituted olefins.
- the double bond may be acyclic, as in alkenes, or semi-cyclic, as in sabinene and beta- pinene, or endocyclic, as in the cycloalkenes.
- Olefins having functional substituents, such as halogens, carboxyl radicals, etc., at the alpha-position with respect to the double bond are less suitable for the oxidation reaction disclosed herein, and fall outside the scope of this invention.
- Singlet oxygen from any source may be used in the olefin oxidation reaction.
- singlet oxygen can be generated by contacting ground-state oxygen with a photo-sensitizer which has been excited by irradiation with visible light, by the reaction of sodium hypochlorite with hydrogen peroxide; by reaction of hydrogen peroxide with bromine in an alkaline medium; by decomposition of the 1:1 adduct of triphenyl phosphite and ozone generated by passage of ozone into a solution of triphenyl phosphite in methylene chloride; by thermal decomposition of epidioxides; or by microwave discharge in a stream of gaseous oxygen.
- singlet oxygen is preferably generated by contacting ground-state molecular oxygen with a suitable photosensitizer which is activated by irradiation with visible light.
- Suitable sensitizers are those organic compounds which have a large molar absorptivity in the visible part of the electromagnetic spectrum, a high quantum yield of triplet formation, a long triplet lifetime, a low tendency toward hydrogen abstraction and self-oxidation, and a triplet energy not far above the energy of singlet oxygen to permit efficient energy transfer to oxygen. Many common dyes meet these requirements adequately.
- Typical classes of dyes that can advantageously be used in the olefin oxidation process of this invention are the xanthenes (rose bengal, erythrosin, eosin, fluorescein), the thiasines (methylene blue), the porphyrins (chlorophyll a and b, hematoporphyrin), the porphins and the phthalocyanines and mixtures thereof.
- xanthenes rose bengal, erythrosin, eosin, fluorescein
- the thiasines methylene blue
- the porphyrins chlorophyll a and b, hematoporphyrin
- the porphins and the phthalocyanines and mixtures thereof are disclosed in the Denny reference, cited supra, and incorporated herein by reference.
- Preferred photo-sensitizers for the present invention are the phthalocyanines and tetraphenyl porphin, and most preferred is tetraphenyl porphin.
- the amount of photosensitizer should neither be very low nor very high. At very low concentrations the sensitizer may not absorb all the available useful light. At too high a concentration, it absorbs all the useful light within a short distance from its entrance to the solution and depletes oxygen in that region of the reaction vessel.
- Preferred amounts of sensitizer range from about 0.01% to about 2.5%, more preferably amounts range from about 0.05% to about 1.3%.
- any source of visible light is suitable for the activation of the sensitizer.
- the source should strongly emit light of the wavelength corresponding with the absorptivity maximum of the sensitizer.
- a sodium vapor discharge tube is particularly suitable for use in combination with tetraphenyl porphin.
- Any catalyst capable of converting the reaction product of the olefin with singlet oxygen, presumably a hydroperoxide, to an alpha-epoxy alcohol is suitable for use in the olefin oxidation reaction of the present invention.
- Suitable catalysts are those which contain a transition metal of the groups IVB, VB, or VIB of the periodic table. Although both heterogeneous and homogeneous catalyst systems can be used, homogeneous catalysts are preferred for their superior selectivity.
- Suitable homogeneous catalysts are soluble salts and metallo-organic complexes of transition metals of group IVB, VB, or VIB of the Periodic Table, excluding chromium.
- Preferred herein are the soluble salts and metallo organic complexes of vanadium and molybdenum.
- examples are vanadylacetylacetonate, molybdenylacetyl acetonate, molybdenum hexacarbonyl, tungsten hexacarbonyl, and vanadium carbonyl.
- Other examples of suitable catalysts are disclosed by Allison, et al., Ind. & Eng. Chem. (Prod. Res. and Dev.) 5 (1966) 166, incorporated herein by reference.
- Preferred catalysts are those containing vanadium or molybdenum; more preferred are those containing vanadium (IV), and most preferred is vanadylacetyl acetonate.
- the amount of catalyst should be sufficient to ensure instantaneous conversion of the hydroperoxides. Much higher levels of catalyst may adversely affect the efficiency of the photo-sensitization reaction, as most catalyst systems absorb visible light.
- Suitable levels of catalyst range from about 0.1 % to about 2.5%. Preferred levels range from 0.7% to 1.3%.
- the superior yields of alpha epoxy alcohol obtained with the process of the present invention in comparison with art disclosed processes is believed to be, at least in part, due to the presence of the catalyst at the time the reaction intermediate, the hydroperoxide, is formed.
- the hydroperoxide is instantaneously converted to the epoxy alcohol by the action of the catalyst.
- the concentration of hydroperoxide remains low throughout the reaction period, and side reactions resulting in ketones and alkenols are suppressed.
- the olefin oxidation reaction of this invention does not require a solvent, better yields are obtained when a solvent is present.
- the reaction mixture may contain from about 5% to about 98% of the solvent. Alcohols should not be used, however, as they tend to interact too strongly with the active sites of the catalyst and make it inactive.
- the reaction mixture should be substantially water-free, as water interacts with the catalyst as well. Examples of suitable solvents are methylene dichloride and toluene.
- the olefin oxidation process of this invention can be used for the conversion of any olefin capable of reaction with singlet oxygen to the corresponding alpha-epoxy alcohol.
- suitable olefins are cyclopentene, cis-9-octadecenoic acid esters, cis-4-octene, 2,3-dimethyl-2-butene, alpha-pinene and beta pinene.
- the olefin is a cyclic alkene, the reaction is highly selective to the cis epoxy alcohol. Thus, oxidation of cyclopentene yields cis 2,3-epoxycyclopentan-1-ol.
- the process of this invention can be used for the conversion of a broad spectrum of olefins to the corresponding alpha epoxy alcohols. All conversion reactions are characterized by a high conversion, a short reaction time and a high yield of the epoxy alcohol.
- Methyl oleate (14.83 g, 0.05 mol) was photooxidized in the manner described in Example I (0.15 g tetraphenylporphin, 0.132 g VO(acac) 2 ). The reaction mixture was concentrated, diluted with 0.2L ether, and washed with water (2 x 0.11) and brine (2 x 0.11). After drying with magnesium sulfate and concentrating, the crude product was chromatographed on silica gel to give the pure epoxy alcohol as a mixture of diastereomers. Isolated yields and reaction conditions are given in Table I.
- Cis-4-octene and 2,3-dimethyl 2-butene were oxidized in the manner described in Example I, and the distilled yields determined. The results are given in Table I. Other olefins capable of reaction with singlet oxygen are oxidized in the same manner. Similar results are obtained.
- Example II The cyclopentene oxidation of Example I was repeated with Mo(CO 6 ), Ti(i-PrO) 4 and VO(acac) 2 as oxidation catalysts. The reactions were run on 0.4 mole of cyclopentene using methylene dichloride as solvent. The conversions after three hours and the product distributions obtained with the different catalysts are given in Table II.
- the second aspect of this invention is a process for synthesizing 7-oxa-11-deoxy prostaglandin analogues comprising the steps of
- Cyclopentene (VII) is oxidized with singlet oxygen to cis-2,3-epoxycyclopentan-1-ol (VI) in the manner described above. Then, the hydroxyl group is protected with a suitable protecting group, e.g. methoxymethyl. The protected cyclopentane compound is reacted with the alkynylalane reagent.
- the alkynylalane reagent is formed in situ by reaction of dimethyl aluminum chloride with a lithiated 1- alkyne.
- Any 1-alkyne is suitable for the purpose of the present invention.
- Preferred 1-alkynes are 1-hexyne and 1-octyne, and derivatives thereof. The most preferred are the 1-octynes.
- the 1-octyne may have substituents at the 3 and/or 4 positions.
- substituted 1-octynes suitable for use in prostanoid synthesis are (R,S)-1-octyn-3-ol, (R)-1-octyn-3-ol, (S)-1-octyn-3-ol, 3-methyl(S)-1-octyn-3-ol, 4-methyl(R,S)-1-octyn-3-ol, 4-methyl(R)-1-octyn-3-ol, 4-methyl(S)-1-octyn-3-ol, 4,4 dimethyl(R,S)-1-octyn-3-ol, 4,4 dimethyl-(R)-1-octyn-3-ol, 4,4 dimethyl-(S)-1-octyn-3-ol, 4,4 dimethyl-(S)-1-octyn-3-ol, (R,S)-1-octyn-4-ol, (R)-1-octyn-4-ol, (S)-1-
- alkynylalane reagents herein are those derived from 1-octyne, (R,S)-1-octyn-3-ol; (S)-1-octyn-3-ol, and 3-methyl(S)-1-octyn-3-ol. Most highly preferred are the alkynylalane reagents derived from (R,S)-1-octyn-3-ol and (S)-1-octyn-3-ol.
- the 1-octyne contains an alcoholic hydroxyl group
- this group has to be protected prior to the reaction with dimethyl aluminum chloride, by e.g. reaction to an ether in a manner similar to the protection reaction for the alcoholic hydroxyl in compound I.
- the t-butyl ether generally is a suitable protective group for the 1-octynol hydroxyl group.
- the alkynylalane reagent thus formed is reacted with compound (VI) to give the corresponding 3 beta- octynyl-alpha-1-methoxymethoxy alpha cyclopentan-2-ol (VIII).
- Compound (VIII) is reacted to compound (IX) by a Williamson synthesis.
- the carboxylic hydroxyl group needs to be protected prior to the exposure of the molecule to sodium hydride.
- the t-butyl ester is a suitable protecting group.
- Any omega-iodo substituted carboxylic acid is suitable for this reaction.
- Preferred carboxylic acids are carboxylic acids containing from 3 to 12 carbon atoms. Particularly suitable are the omega-iodo alkanoic acid derivatives of the formula: wherein R 1 is or and R 2 and R 3 are each
- omega-iodo derivatives of hexenoic acid and 4-hexanoic acid are preferred herein. Most preferred is the hexanoic acid derivative.
- 2,3-Epoxymethoxymethoxycyclopentane (1) was prepared in accordance with the invention as follows.
- alcohol (2) is reacted with t-butyl-6-iodo-3-hexenoate, t-butyl-6-iodo-4-hexenoate, t-butyl-6-iodo-2,2-dimethyl-hexanoate, t-butyl 6-iodo-2,2 diethyl-hexanoate, t-butyl-6-iodo-2 methoxymethoxy-hexanoate, and the corresponding prost-13,14-ynoates are obtained.
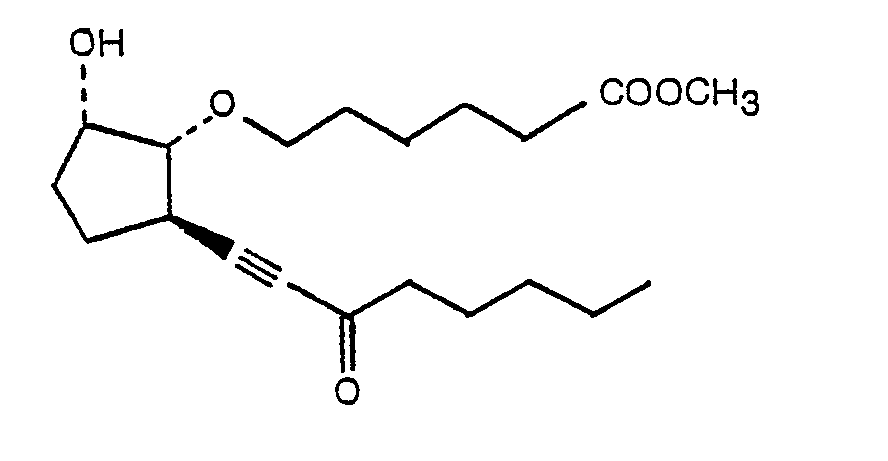
- Compound (4) belongs to the novel class of 7-oxa-prostanoic acid derivatives of this invention.
- the keto-analogues of (4) can be prepared therefrom by oxidation with chromium trioxide (Example VIII). If the 15-hydroxy is protected with TFA prior to the reaction with chromium trioxide (Example IX), the 9-hydroxy is oxidized selectively (Example X).
- Ketone (11) (220mg, 0.5mmol) was dissolved in 1.4ml CH 2 Cl 2 and cooled to -15°C under argon; trifluoroacetic acid (1.4mi, cooled to 0°C) was added, with stirring. After 10 min. the cooling bath was removed, and the reaction stirred for 90 min. more. The reaction mixture was concentrated under vacuum and passed through a silica gel column (elution with 94:3:3 CHCl 3 :methanol:acetic acid).
- R 5 is C 4 H 9 .
- R 1 is preferably
- the C 13 ⁇ C 14 bond is preferably a double bond or a triple bond, more preferably a triple bond.
- R 3 is R 4 is preferably CH 2 , most preferably CH 2 .
- a number of the 7-oxa prostanoic acid derivatives has been tested for cytoprotective activity in rats.
- Sprague-Dawley rats are fasted for 24 hours prior to the pretreatment.
- the pretreatment is oral administration of a 5mg per kg body weight dose of the prostanoid in 2.5ml of a vehicle consisting of Tween® 80 (0.75%) and the balance water.
- the control animals are given 2.5ml of the vehicle, without prostanoid.
- absolute ethanol is administered orally to the animals, in a dose of 10mi per kg body weight.
- One hour later the animals are sacrificed, their stomachs dissected out, opened along the greater curvature and the mucose examined for lesions.
- the average lesion length is expressed as percentage of the average lesion length found in the stomach mucosa of control animals.
- this test measures the active cytoprotective properties of the prostanoids, independent of the gastric secretion inhibition that these compounds may or may not show.
- the test is discussed more fully by Robert et al., Gastroenterology 77 (1979) 433, incorporated herein by reference.
- methyl-7-oxa-9-oxo-Prostanoate derivatives were tested for cytoprotective properties.
- the compounds differed with respect to the nature of the C 13 ⁇ C 14 bond (triple, cis or trans double, or single), the substituent(s) at the 15-position and at the 16-position. The following results were obtained.
- the substituent at the 15-position be either oxo or (S)OH. If the substituent is oxo, the C 13 ⁇ C 14 bond preferably is a triple bond.
- oxo and S(OH) appear to be the preferred substituents at the 15-position.
- Example XIX The optically pure compounds of Example XIX were tested for cytoprotective activity. The following results were obtained (Table III).
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Pharmacology & Pharmacy (AREA)
- Engineering & Computer Science (AREA)
- Animal Behavior & Ethology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Catalysts (AREA)
- Epoxy Compounds (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
- Low-Molecular Organic Synthesis Reactions Using Catalysts (AREA)
Claims (10)
R2 représente C = 0 ou
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AT82200192T ATE15368T1 (de) | 1981-03-02 | 1982-02-18 | Prostaglandin-analoga und verfahren zu ihrer herstellung. |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US06/239,765 US4345984A (en) | 1981-03-02 | 1981-03-02 | Novel prostaglandin analogues and process for making same |
| US239765 | 1981-03-02 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP0061209A1 EP0061209A1 (fr) | 1982-09-29 |
| EP0061209B1 true EP0061209B1 (fr) | 1985-09-04 |
Family
ID=22903636
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP82200192A Expired EP0061209B1 (fr) | 1981-03-02 | 1982-02-18 | Analogues de prostaglandines et leur procédé de préparation |
Country Status (12)
| Country | Link |
|---|---|
| US (1) | US4345984A (fr) |
| EP (1) | EP0061209B1 (fr) |
| JP (1) | JPS57206654A (fr) |
| AT (1) | ATE15368T1 (fr) |
| AU (2) | AU548052B2 (fr) |
| CA (1) | CA1210016A (fr) |
| DE (1) | DE3265945D1 (fr) |
| ES (3) | ES510008A0 (fr) |
| IE (1) | IE52623B1 (fr) |
| MX (1) | MX7354E (fr) |
| NZ (1) | NZ199807A (fr) |
| ZA (1) | ZA821102B (fr) |
Families Citing this family (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4383904A (en) * | 1981-09-21 | 1983-05-17 | Celanese Corporation | Photochemical epoxidation |
| US4576962A (en) * | 1983-10-19 | 1986-03-18 | The Procter & Gamble Company | Prostaglandin analogues |
| IL75230A (en) * | 1984-05-18 | 1989-07-31 | Glaxo Group Ltd | Aminocyclopentyl ethers and their preparation and pharmaceutical formulation |
| US4851555A (en) * | 1984-10-25 | 1989-07-25 | Scientific Design Company, Inc. | Process for preparing alkylene oxides from alkylene carbonates |
| EP0267878A1 (fr) * | 1986-11-14 | 1988-05-18 | Ciba-Geigy Ag | Dérivés de l'adénine N9-substitués par le groupement cyclopentyle |
| JPH08501541A (ja) * | 1992-09-16 | 1996-02-20 | ヘキスト・アクチェンゲゼルシャフト | 不飽和化合物の増感された光酸素付加のための方法 |
| US6674781B1 (en) * | 2002-08-19 | 2004-01-06 | The Boeing Company | Method and system for fueling a closed cycle chemical oxygen iodine laser |
| ES2432063T3 (es) * | 2009-06-16 | 2013-11-29 | Crystal Pharma, S.A.U. | Procedimiento para la obtención de 17-espirolactonas en esteroides |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| NL300754A (fr) * | 1962-11-20 | |||
| US4175201A (en) * | 1973-05-18 | 1979-11-20 | University Of Chicago | Chemical synthesis |
| US4021369A (en) * | 1973-06-29 | 1977-05-03 | Sun Ventures, Inc. | Ion-exchanged transition metal catalysts for the direct oxidation of olefins to epoxyalcohols |
| US4123445A (en) * | 1974-11-13 | 1978-10-31 | Sun Ventures, Inc. | Novel homogeneous transition-metal catalyzed geometric isomerization of cyclic epoxy alcohols |
| US4180672A (en) * | 1976-08-13 | 1979-12-25 | Teijin Limited | 2-Organothio-2-cyclopentenones, organothio-cyclopentanes derived therefrom |
-
1981
- 1981-03-02 US US06/239,765 patent/US4345984A/en not_active Expired - Lifetime
-
1982
- 1982-02-18 DE DE8282200192T patent/DE3265945D1/de not_active Expired
- 1982-02-18 EP EP82200192A patent/EP0061209B1/fr not_active Expired
- 1982-02-18 AT AT82200192T patent/ATE15368T1/de not_active IP Right Cessation
- 1982-02-19 ZA ZA821102A patent/ZA821102B/xx unknown
- 1982-02-19 AU AU80632/82A patent/AU548052B2/en not_active Ceased
- 1982-02-23 NZ NZ199807A patent/NZ199807A/en unknown
- 1982-03-01 MX MX829947U patent/MX7354E/es unknown
- 1982-03-01 IE IE450/82A patent/IE52623B1/en unknown
- 1982-03-01 ES ES510008A patent/ES510008A0/es active Granted
- 1982-03-01 CA CA000397348A patent/CA1210016A/fr not_active Expired
- 1982-03-02 JP JP57032953A patent/JPS57206654A/ja active Pending
-
1983
- 1983-02-18 ES ES519903A patent/ES8500224A1/es not_active Expired
- 1983-02-18 ES ES519902A patent/ES8403451A1/es not_active Expired
-
1985
- 1985-08-22 AU AU46558/85A patent/AU4655885A/en not_active Abandoned
Non-Patent Citations (2)
| Title |
|---|
| Römpp's Chemie Lexikon, Stuttgart, 1975, S. 3234-35 * |
| TETRAHEDRON LETTERS, no. 32, August 1974, pages 2737-2738; J.E. LYONS: "The stereoselective oxidation of cyclohexene to cis-1,2-epoxycyclohexane-3-01 in the presence of (C5H5V(CO)4)" * |
Also Published As
| Publication number | Publication date |
|---|---|
| ES519903A0 (es) | 1984-10-01 |
| MX7354E (es) | 1988-07-19 |
| ES8307738A1 (es) | 1983-07-01 |
| AU548052B2 (en) | 1985-11-21 |
| ES510008A0 (es) | 1983-07-01 |
| ATE15368T1 (de) | 1985-09-15 |
| CA1210016A (fr) | 1986-08-19 |
| JPS57206654A (en) | 1982-12-18 |
| US4345984A (en) | 1982-08-24 |
| IE52623B1 (en) | 1988-01-06 |
| NZ199807A (en) | 1985-11-08 |
| DE3265945D1 (en) | 1985-10-10 |
| ES519902A0 (es) | 1984-03-16 |
| ZA821102B (en) | 1983-01-26 |
| AU8063282A (en) | 1982-09-09 |
| IE820450L (en) | 1982-09-02 |
| ES8500224A1 (es) | 1984-10-01 |
| ES8403451A1 (es) | 1984-03-16 |
| EP0061209A1 (fr) | 1982-09-29 |
| AU4655885A (en) | 1985-12-05 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Newton et al. | Steric control in prostaglandin synthesis involving bicyclic and tricyclic intermediates | |
| JPH0459306B2 (fr) | ||
| Finch et al. | Total synthesis of dl-9-deoxyprostaglandin E1 | |
| JP3568219B2 (ja) | 19−ノルビタミンd化合物の調製 | |
| EP0061209B1 (fr) | Analogues de prostaglandines et leur procédé de préparation | |
| Demuth et al. | Photocycloadditions of 2‐(trimethylsilyloxy)‐1, 3‐butadiene to 2‐cycloalkenones. Access to the basic pentalenolactone skeleton | |
| Greene et al. | The total synthesis of prostaglandins by the tropolone route | |
| EP0081892B1 (fr) | Procédé pour préparer de dérivés de bicycloheptane et dérivés de bicycloheptane | |
| Davies et al. | Rhodium carbenoid mediated cyclisations. Part 4. Synthetic approaches to oxepanes related to zoapatanol | |
| US4496759A (en) | Novel prostaglandin analogues and process for making same | |
| Duncia et al. | Stereoselective and regioselective Lewis acid catalyzed ene reactions of. alpha.-substituted acrylate esters | |
| US4608388A (en) | Novel [4,2,0]bicyclooctane derivatives with valuable therapeutic properties | |
| Kwon et al. | Chemistry of singlet oxygen. 53. Environmental effects on the photooxygenation of 2-methoxy-3-methyl-2-cyclopenten-1-one | |
| CA1221963A (fr) | Analogues de prostaglandine, et leur fabrication | |
| EP0362816B1 (fr) | Dérivés de l'acide cyclopentèneheptanoique et méthode pour leur préparation | |
| NZ211848A (en) | 7-oxa-11-deoxy prostaglandin analogues and production thereof | |
| Suginome et al. | Photoinduced molecular transformations. Part 152. Ring expansion based on a sensitized [2+ 2] photoaddition of enol ethers of cyclic ketones with olefins, followed by a β-scission of alkoxyl radicals generated from the resulting cyclobutanols. Two-carbon ring expansion of β-indanone, β-tetralone and β-suberone | |
| FI78289C (fi) | Foerfarande foer framstaellning av terapeutiskt anvaendbara bicyklo/3.2.0/heptan-6-onoximeterderivat. | |
| Cantrell | Reactivity of photochemically excited 3-acylthiophenes, 3-acylfurans, and the formylthiophenes and furans | |
| US5434300A (en) | Method for preparing dodecahydro-3A,6,6,9A-tetramethylnaphtho [2,1-B]furan and novel haloethyl decalin derivatives | |
| Goodwin et al. | Synthesis of two new maytansinoid model compounds from carbohydrate precursors | |
| Takeda et al. | Cope rearrangement of some germacrane-type furan sesquiterpenes. Part IV. Preparation of some cyclodeca-cis, trans-diene derivatives and their Cope rearrangements | |
| JP2503073B2 (ja) | 2―置換―2―シクロペンテノン類およびそれを含有する骨形成促進剤又は制ガン剤 | |
| Jiang et al. | Total synthesis of dioxane analogs related to zoapatanol | |
| Liljebris et al. | Ligand-controlled palladium-catalyzed intramolecular reactions of phenyl-substituted prostaglandin F2α analogues |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Designated state(s): AT BE CH DE FR GB IT LU NL SE |
|
| 17P | Request for examination filed |
Effective date: 19830311 |
|
| ITF | It: translation for a ep patent filed | ||
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Designated state(s): AT BE CH DE FR GB IT LI LU NL SE |
|
| REF | Corresponds to: |
Ref document number: 15368 Country of ref document: AT Date of ref document: 19850915 Kind code of ref document: T |
|
| REF | Corresponds to: |
Ref document number: 3265945 Country of ref document: DE Date of ref document: 19851010 |
|
| ET | Fr: translation filed | ||
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19860228 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed | ||
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: AT Payment date: 19870225 Year of fee payment: 6 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: NL Payment date: 19870228 Year of fee payment: 6 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Effective date: 19890218 Ref country code: AT Effective date: 19890218 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SE Effective date: 19890219 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LI Effective date: 19890228 Ref country code: CH Effective date: 19890228 Ref country code: BE Effective date: 19890228 |
|
| BERE | Be: lapsed |
Owner name: THE PROCTER & GAMBLE CY Effective date: 19890228 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Effective date: 19890901 |
|
| NLV4 | Nl: lapsed or anulled due to non-payment of the annual fee | ||
| GBPC | Gb: european patent ceased through non-payment of renewal fee | ||
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19891027 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Effective date: 19891101 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST |
|
| EUG | Se: european patent has lapsed |
Ref document number: 82200192.1 Effective date: 19900118 |