EP0442767B1 - Verfahren und Vorrichtung zum Behandeln eines natürlichen Gases - Google Patents
Verfahren und Vorrichtung zum Behandeln eines natürlichen Gases Download PDFInfo
- Publication number
- EP0442767B1 EP0442767B1 EP91400092A EP91400092A EP0442767B1 EP 0442767 B1 EP0442767 B1 EP 0442767B1 EP 91400092 A EP91400092 A EP 91400092A EP 91400092 A EP91400092 A EP 91400092A EP 0442767 B1 EP0442767 B1 EP 0442767B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- gas
- phase
- additive
- water
- process according
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
- VNWKTOKETHGBQD-UHFFFAOYSA-N methane Chemical compound C VNWKTOKETHGBQD-UHFFFAOYSA-N 0.000 title claims description 56
- 238000000034 method Methods 0.000 title claims description 41
- 239000003345 natural gas Substances 0.000 title claims description 25
- 239000007789 gas Substances 0.000 claims description 124
- 239000000654 additive Substances 0.000 claims description 73
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 71
- 239000012071 phase Substances 0.000 claims description 62
- 230000000996 additive effect Effects 0.000 claims description 54
- 238000004519 manufacturing process Methods 0.000 claims description 42
- 239000008346 aqueous phase Substances 0.000 claims description 36
- 150000002430 hydrocarbons Chemical class 0.000 claims description 34
- 229930195733 hydrocarbon Natural products 0.000 claims description 33
- 239000002904 solvent Substances 0.000 claims description 31
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 claims description 30
- 238000005260 corrosion Methods 0.000 claims description 30
- 239000007788 liquid Substances 0.000 claims description 28
- 239000007791 liquid phase Substances 0.000 claims description 28
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 claims description 21
- 239000004215 Carbon black (E152) Substances 0.000 claims description 18
- 238000000926 separation method Methods 0.000 claims description 15
- 239000007792 gaseous phase Substances 0.000 claims description 14
- 150000001875 compounds Chemical class 0.000 claims description 10
- 238000004064 recycling Methods 0.000 claims description 10
- 238000001816 cooling Methods 0.000 claims description 7
- 239000002253 acid Substances 0.000 claims description 6
- 229920006395 saturated elastomer Polymers 0.000 claims description 6
- HZAXFHJVJLSVMW-UHFFFAOYSA-N 2-Aminoethan-1-ol Chemical compound NCCO HZAXFHJVJLSVMW-UHFFFAOYSA-N 0.000 claims description 4
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims description 4
- YNAVUWVOSKDBBP-UHFFFAOYSA-N Morpholine Chemical compound C1COCCN1 YNAVUWVOSKDBBP-UHFFFAOYSA-N 0.000 claims description 4
- HQABUPZFAYXKJW-UHFFFAOYSA-N butan-1-amine Chemical compound CCCCN HQABUPZFAYXKJW-UHFFFAOYSA-N 0.000 claims description 4
- PAFZNILMFXTMIY-UHFFFAOYSA-N cyclohexylamine Chemical compound NC1CCCCC1 PAFZNILMFXTMIY-UHFFFAOYSA-N 0.000 claims description 4
- WGYKZJWCGVVSQN-UHFFFAOYSA-N propylamine Chemical compound CCCN WGYKZJWCGVVSQN-UHFFFAOYSA-N 0.000 claims description 4
- 238000005057 refrigeration Methods 0.000 claims description 4
- 238000009834 vaporization Methods 0.000 claims description 4
- 238000005406 washing Methods 0.000 claims description 4
- NVJUHMXYKCUMQA-UHFFFAOYSA-N 1-ethoxypropane Chemical compound CCCOCC NVJUHMXYKCUMQA-UHFFFAOYSA-N 0.000 claims description 2
- XNWFRZJHXBZDAG-UHFFFAOYSA-N 2-METHOXYETHANOL Chemical compound COCCO XNWFRZJHXBZDAG-UHFFFAOYSA-N 0.000 claims description 2
- XTHFKEDIFFGKHM-UHFFFAOYSA-N Dimethoxyethane Chemical compound COCCOC XTHFKEDIFFGKHM-UHFFFAOYSA-N 0.000 claims description 2
- PIICEJLVQHRZGT-UHFFFAOYSA-N Ethylenediamine Chemical compound NCCN PIICEJLVQHRZGT-UHFFFAOYSA-N 0.000 claims description 2
- HPNMFZURTQLUMO-UHFFFAOYSA-N diethylamine Chemical compound CCNCC HPNMFZURTQLUMO-UHFFFAOYSA-N 0.000 claims description 2
- NKDDWNXOKDWJAK-UHFFFAOYSA-N dimethoxymethane Chemical compound COCOC NKDDWNXOKDWJAK-UHFFFAOYSA-N 0.000 claims description 2
- POLCUAVZOMRGSN-UHFFFAOYSA-N dipropyl ether Chemical compound CCCOCCC POLCUAVZOMRGSN-UHFFFAOYSA-N 0.000 claims description 2
- WEHWNAOGRSTTBQ-UHFFFAOYSA-N dipropylamine Chemical compound CCCNCCC WEHWNAOGRSTTBQ-UHFFFAOYSA-N 0.000 claims description 2
- VNKYTQGIUYNRMY-UHFFFAOYSA-N methoxypropane Chemical compound CCCOC VNKYTQGIUYNRMY-UHFFFAOYSA-N 0.000 claims description 2
- XCVNDBIXFPGMIW-UHFFFAOYSA-N n-ethylpropan-1-amine Chemical compound CCCNCC XCVNDBIXFPGMIW-UHFFFAOYSA-N 0.000 claims description 2
- BDERNNFJNOPAEC-UHFFFAOYSA-N propan-1-ol Chemical compound CCCO BDERNNFJNOPAEC-UHFFFAOYSA-N 0.000 claims description 2
- 238000011084 recovery Methods 0.000 claims description 2
- 230000001172 regenerating effect Effects 0.000 claims description 2
- 230000000295 complement effect Effects 0.000 claims 3
- 210000002196 fr. b Anatomy 0.000 claims 1
- 210000003918 fraction a Anatomy 0.000 claims 1
- 230000007797 corrosion Effects 0.000 description 16
- 239000000203 mixture Substances 0.000 description 14
- 230000002401 inhibitory effect Effects 0.000 description 13
- 239000003112 inhibitor Substances 0.000 description 9
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 7
- 230000015572 biosynthetic process Effects 0.000 description 7
- 238000010586 diagram Methods 0.000 description 7
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 6
- 238000012856 packing Methods 0.000 description 5
- 238000009833 condensation Methods 0.000 description 4
- 230000005494 condensation Effects 0.000 description 4
- 238000009434 installation Methods 0.000 description 4
- RWSOTUBLDIXVET-UHFFFAOYSA-N Dihydrogen sulfide Chemical compound S RWSOTUBLDIXVET-UHFFFAOYSA-N 0.000 description 3
- 238000009835 boiling Methods 0.000 description 3
- 229910002092 carbon dioxide Inorganic materials 0.000 description 3
- 230000018044 dehydration Effects 0.000 description 3
- 238000006297 dehydration reaction Methods 0.000 description 3
- 150000004677 hydrates Chemical class 0.000 description 3
- 229910000037 hydrogen sulfide Inorganic materials 0.000 description 3
- 239000003507 refrigerant Substances 0.000 description 3
- 230000008016 vaporization Effects 0.000 description 3
- 239000013078 crystal Substances 0.000 description 2
- 230000000694 effects Effects 0.000 description 2
- WGCNASOHLSPBMP-UHFFFAOYSA-N hydroxyacetaldehyde Natural products OCC=O WGCNASOHLSPBMP-UHFFFAOYSA-N 0.000 description 2
- 229940087646 methanolamine Drugs 0.000 description 2
- 238000002156 mixing Methods 0.000 description 2
- 239000000047 product Substances 0.000 description 2
- 238000010521 absorption reaction Methods 0.000 description 1
- 150000001412 amines Chemical class 0.000 description 1
- 239000000872 buffer Substances 0.000 description 1
- 229910052799 carbon Inorganic materials 0.000 description 1
- 239000001569 carbon dioxide Substances 0.000 description 1
- -1 compounds hydrocarbon Chemical class 0.000 description 1
- 239000000470 constituent Substances 0.000 description 1
- 238000010908 decantation Methods 0.000 description 1
- 238000007599 discharging Methods 0.000 description 1
- 230000009977 dual effect Effects 0.000 description 1
- 239000004744 fabric Substances 0.000 description 1
- 230000005484 gravity Effects 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- 239000002808 molecular sieve Substances 0.000 description 1
- 150000002894 organic compounds Chemical class 0.000 description 1
- 238000004806 packaging method and process Methods 0.000 description 1
- URGAHOPLAPQHLN-UHFFFAOYSA-N sodium aluminosilicate Chemical compound [Na+].[Al+3].[O-][Si]([O-])=O.[O-][Si]([O-])=O URGAHOPLAPQHLN-UHFFFAOYSA-N 0.000 description 1
- 238000001179 sorption measurement Methods 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 239000013589 supplement Substances 0.000 description 1
- 230000009466 transformation Effects 0.000 description 1
- 238000000844 transformation Methods 0.000 description 1
- 239000012808 vapor phase Substances 0.000 description 1
Images
Classifications
-
- F—MECHANICAL ENGINEERING; LIGHTING; HEATING; WEAPONS; BLASTING
- F17—STORING OR DISTRIBUTING GASES OR LIQUIDS
- F17D—PIPE-LINE SYSTEMS; PIPE-LINES
- F17D1/00—Pipe-line systems
- F17D1/02—Pipe-line systems for gases or vapours
- F17D1/04—Pipe-line systems for gases or vapours for distribution of gas
- F17D1/05—Preventing freezing
-
- E—FIXED CONSTRUCTIONS
- E21—EARTH OR ROCK DRILLING; MINING
- E21B—EARTH OR ROCK DRILLING; OBTAINING OIL, GAS, WATER, SOLUBLE OR MELTABLE MATERIALS OR A SLURRY OF MINERALS FROM WELLS
- E21B43/00—Methods or apparatus for obtaining oil, gas, water, soluble or meltable materials or a slurry of minerals from wells
- E21B43/34—Arrangements for separating materials produced by the well
-
- F—MECHANICAL ENGINEERING; LIGHTING; HEATING; WEAPONS; BLASTING
- F17—STORING OR DISTRIBUTING GASES OR LIQUIDS
- F17D—PIPE-LINE SYSTEMS; PIPE-LINES
- F17D1/00—Pipe-line systems
- F17D1/005—Pipe-line systems for a two-phase gas-liquid flow
Definitions
- the present invention relates to a method and a device for implementing and regenerating corrosion inhibiting additives and / or hydrates for transporting and treating a natural gas.
- Such a process, with recycling of the solvent phase comprising the additives, is known from FR-A-2 618 876.
- the natural gas is saturated with water at the temperature of production; during transport, the gas generally undergoes a temperature drop which causes a condensation of part of the water, but which can also under certain conditions cause the formation of hydrate crystals, which are inclusion compounds hydrocarbon molecules in crystal structures formed by water molecules and which form at a temperature significantly above 0 ° C.
- hydrate crystals which are inclusion compounds hydrocarbon molecules in crystal structures formed by water molecules and which form at a temperature significantly above 0 ° C.
- the formation of hydrates in a gas pipeline can lead to plugging and stopping production. To avoid this, it is necessary either to dehydrate the gas before transporting it, or to inject a hydrate inhibitor such as methanol or ethylene glycol into the gas.
- the gas is generally treated in a washing unit with glycol to adjust the water dew point to the value imposed for transport, the latter being carried out under single-phase conditions; in the second case, the inhibitor is introduced into the gas just after the wellhead and the transport is carried out at least partially under two-phase conditions.
- the gas which can come from several different wells collected on the same pipeline, is generally dehydrated to obtain a lower water dew point than that required by transport; this second dehydration step can be carried out in most cases either by absorption of water in glycol, or by adsorption of water on molecular sieves; the dehydration process thus implemented may be different from that used on the production site to ensure the water dew point necessary for transport.
- This second dehydration step is essential if we want to be able to cool the gas to a relatively low temperature, which can for example be between -10 and -40 ° C, in order to extract the liquids from natural gas, that is to say hydrocarbons other than methane which can be delivered liquid at room temperature. Under these conditions, the additives that have been injected for transport (hydrate formation inhibitors and corrosion inhibitors) are absorbed during the treatment and are not recycled.
- the method according to the invention corresponds to a new implementation of these anti-hydrate and / or anti-corrosion additives which allows their recycling.
- normally liquid liquid under normal conditions of temperature and pressure.
- the proportion by weight of anti-hydrate solvent in water is generally from 10 to 70% and preferably from 20 to 50%.
- the anti-hydrate additive and the water at least one anti-corrosion additive, non-hydrocarbon at least partially miscible with water or dispersible in the water and preferably vaporizing at a boiling temperature lower than that of water or forming with water an azeotrope whose boiling temperature is lower than that of water, so that it can be entrained by the gas during step (a) of the process.
- the proportion of aqueous liquid phase introduced into the contact zone generally corresponds to 0.05 to 5% by weight of the mass flow rate of gas to be treated and advantageously from 0.1 to 1%, the contacting step s generally performing at a temperature and at a pressure corresponding substantially to that of the gases leaving the production well, for example approximately at 20 to 100 ° C. under 0.1 to 25 MPa.
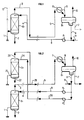
- FIG. 1 The principle of the process according to the invention is illustrated by the diagram in FIG. 1, applied by way of example to a natural gas containing methane, associated higher hydrocarbons, acid gases (carbon dioxide, hydrogen sulfide) and saturated in water under production temperature and pressure conditions.
- the natural gas leaving the production wellhead arrives via the pipe 1, at the bottom of a contacting enclosure G1 preferably substantially vertical. It is brought into contact, in the contact zone G1 preferably operating against the current, with a mixture consisting of water, at least one hydrate inhibiting solvent alone or in admixture with at least one corrosion inhibiting additive and coming from line 4.
- a gas phase laden with solvent and additive is removed at the head via line 3.
- an aqueous phase substantially free of solvent and additive is withdrawn through line 2.
- the overhead gas phase is transported in line 3 over a distance which may be several kilometers and arrives via line 5 at the reception terminal where the gas can be treated before it is dispatched into the commercial network.
- the gas flowing in line 5 is cooled to the low temperature necessary for treatment in the heat exchanger E1 by a refrigerant external to the process, which causes partial condensation; this cooling does not cause a hydrate formation phenomenon due to the presence of the inhibiting solvent in the gas in a sufficiently large amount.
- the cooled mixture leaving the exchanger E1 via line 6 consists of a condensate comprising an aqueous liquid phase which contains most of the water, solvent and additive which were in the gas leaving the contact area G1 via the conduit 3, and of a so-called poor gas phase depleted in heavy hydrocarbons.
- the phenomena of hydrate formation and corrosion do not occur, because they are inhibited by the presence of the anti-hydrate solvent and the anti-corrosion additive which protect the whole of the installation.
- One of the advantages of the process according to the invention is that the anti-hydrate and anti-corrosion additives which are used are effective over the entire installation, that is to say the contact zone G1 between the gas and additives on the production site, the transport pipe which allows the gas to be transported from the production zone to the reception terminal and the treatment zone during which natural gas is separated from water and hydrocarbons the heavier.
- part of the gas to be transported (conduit 12) can be directly mixed with the gas leaving the contact area G1 through the conduit 3, without having to pass through the contact area G1.
- natural gas is generally produced by several wells. In this case it is possible to collect the effluents from several different wells on a single process according to the invention; for this, the gas coming from certain wells can be introduced into the process according to the invention via line 1, while the gas coming from other wells can be introduced into the process through line 12.
- natural gas is produced by two main sites and it is assumed to contain methane, associated higher hydrocarbons and to be saturated with water under the conditions of temperature and pressure of production.
- the outgoing natural gas of a production wellhead is treated as described above for FIG. 1.
- the natural gas leaving another production wellhead arrives via the conduit 21. It is brought into contact, in the contact zone G2, with a mixture consisting of water and hydrate-inhibiting solvent coming from line 24.
- a gas phase laden with solvent is removed at the head, through line 23.
- an aqueous phase substantially free of solvent is drawn off via line 22.
- the overhead gas phase is transported in line 23 and it is mixed in line 25 with the gas coming from the first production site and circulating in line 3.
- All of the gas is transported over a distance which can be several kilometers and arrives via line 5 at the reception terminal where the gas can be treated before it is sent to the commercial network.
- the gas flowing in line 5 is cooled to the low temperature necessary for treatment in the heat exchanger E1 by a refrigerant external to the process, which causes partial condensation; this cooling does not cause a hydrate formation phenomenon due to the presence of the inhibiting solvent in the gas in a sufficiently large amount.
- the cooled mixture leaving the exchanger E1 via line 6 consists of an aqueous liquid phase which contains most of the water and the solvent which were on the one hand in the gas leaving the contact zone G1 via line 3 and secondly in the gas leaving the contact zone G2 via line 23, of a liquid hydrocarbon phase consisting of the heaviest hydrocarbons in the gas and of a so-called lean poor gas phase heavy hydrocarbons.
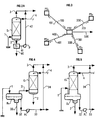
- FIG. 3 shows an example of a production diagram operating with four wells distant from each other, denoted respectively PS1, PS2, PS3 and PS4.
- the gas is conveyed via lines 100 from the well PS1, 200 from the well PS2, 300 from the well PS3, 400 from the well PS4 to a central processing platform PTC.
- the gas is cooled so as to obtain an aqueous phase and a partially dehydrated gas, the water dew point of which complies with the transport specification which imposes a value, for example less than or equal to -10 ° C.
- the gas thus obtained is compressed by a compressor placed on the PTC platform and evacuated via line 500.
- the aqueous phase is returned to the production wells PS1, PS2, PS3 and PS4 by the pumps which return, via the pipes 101, 201, 301 and 401, the flow rates of the aqueous phase proportional to the flow rates of gas conveyed by the pipes 100, 200, 300 and 400.
- a contactor which makes it possible to charge the product gas as an additive and to evacuate an aqueous phase substantially free of the additive which it contained at the start.
- an additive reserve which is renewed periodically, enables regular add-ons to compensate for the loss of additive.
- natural gas is produced accompanied by hydrocarbon condensates, that is to say that the effluent leaving the well consists of a gas phase and a fraction of liquids, composed of hydrocarbons. the heaviest; in most cases, an aqueous liquid phase is also present at the outlet of the well.
- the diagram of the process according to the invention may be slightly different to take into account the liquid hydrocarbon phase; this variant is illustrated by the Figure 4: the condensate gas leaving the production wellhead arrives via line 1 and enters the upper part of a separator flask B2 in which the 3 phases are separated: the aqueous phase, consisting of water deposit, is withdrawn through the conduit 30; the liquid hydrocarbon phase is drawn off through line 32, taken up by pump P3 and evacuated through line 33; the gaseous phase is drawn off through line 31 and brought into contact, in contact zone G1, with a mixture consisting of water, solvent and additives and coming from line 4.
- the condensate gas leaving the production wellhead arrives via line 1 and enters the upper part of a separator flask B2 in which the 3 phases are separated: the aqueous phase, consisting of water deposit, is withdrawn through the conduit 30; the liquid hydrocarbon phase is drawn off through line 32, taken up by pump P3 and evacuated through line 33; the gaseous phase is drawn off through line 31
- FIG. 5 A variant of the case of condensate gas production is illustrated in FIG. 5: in this case, the separating flask B2 and the contact area G1 are integrated into a single item of equipment in order to gain compactness, a criterion which is particularly interesting in the case of production at sea.
- the condensate gas leaving the production wellhead arrives via line 1 and enters the separator flask B2 in which are separated the liquid hydrocarbon phase, an aqueous phase consisting of reservoir water and water from the contact zone G1 in direct relation with the upper part of the separator B2 and a gas phase which is brought into contact against the current, in the contact zone G1, with a mixture consisting of water, solvent and additives and coming from line 4.
- a gas phase loaded with solvent and additives is evacuated at the head, via line 3, which is transported to the reception terminal.
- the aqueous phase substantially free of solvent and additives is mixed with the aqueous phase of deposit water, decanted and drawn off through line 2.
- the liquid hydrocarbon phase is drawn off from the flask B2 through line 32, taken up by pump P3 and evacuated through line 33; this phase can either be transported by an independent pipe to a reception terminal, or mixed with the gas circulating in the pipe 3, in which case the transport under these conditions is carried out in two-phase regime.
- This variant allows the packing G1 to play a dual role: on the one hand it makes it possible to make contact between the aqueous phase arriving via line 4 and the gas arriving through line 1; on the other hand it makes it possible to stop the liquid droplets entrained by the gas and thus improve the separation between phases.
- the installation shown diagrammatically in FIG. 5 can be carried out on land, on a platform at sea, or under the sea.
- the gas does not contain hydrocarbon condensate at the outlet of the well
- the water evacuated through line 2 can be sent directly to the sea provided that the additive has been sufficiently purified in the contact column G1.
- the gas is then transported by an underwater pipe in single-phase conditions.
- the gas contains a hydrocarbon condensate at the outlet of the well, after separation, this condensate is preferably remixed with the gas so as to carry out simultaneous transport in two-phase conditions, which makes it possible to transport the two phases in a single pipe. It may be necessary to raise the pressure level before transport, which can be done either after mixing by a two-phase pump or compressor, or after mixing by passing the gas through a compressor and the condensate through a pump.
- the anti-hydrate solvent can advantageously be, for example, methanol. It can also be chosen, for example, from the following solvents: methylpropylether, ethylpropylether, dipropylether, methyltertiobutylether, dimethoxymethane, dimethoxyethane, ethanol, methoxyethanol, propanol, used alone or as a mixture.
- the anti-corrosion additive may preferably be chosen from organic compounds of the chemical family of amines, such as diethylamine, propylamine, butylamine, triethylamine, dipropylamine, ethylpropylamine, ethanolamine, cyclohexylamine, pyrrid morpholine, ethylenediamine, used alone or as a mixture.
- organic compounds of the chemical family of amines such as diethylamine, propylamine, butylamine, triethylamine, dipropylamine, ethylpropylamine, ethanolamine, cyclohexylamine, pyrrid morpholine, ethylenediamine, used alone or as a mixture.
- the corrosion inhibitor additive is dispersible in water and if its boiling temperature is higher than that of water, said additive can be recovered and recycled as shown in the diagram in FIG. 2A: according to this diagram, the natural gas leaving the production wellhead arrives via line 1. It is brought into contact, in the contact zone G1, with a mixture consisting of water, hydrate inhibiting solvent and d corrosive inhibiting additive coming from line 4. A gas phase containing essentially solvent is removed at the head via line 3.
- the aqueous phase substantially freed of solvent, but still containing the majority of the corrosion-inhibiting additive which has not been entrained by the gas leaves the contact zone G1 via line 2, and enters the separator S1 in which the water is separated from the corrosion inhibiting additive; the water, practically completely free of solvent and of corrosion-inhibiting additive, leaves S1 through line 40; the corrosion inhibitor additive leaves S1 through line 41, is taken up by pump P4 and sent via line 42 into line 3 in order to be remixed with the gas coming from contact zone G1 and circulating in line 3 to inhibit corrosion during gas transport to the processing terminal.
- the separator S1 can be of different types such as for example coalescer, decanter, extractor, distiller, centrifuge.
- the refrigeration temperature required to extract the heaviest hydrocarbons from the gas is a function of the gas pressure and the desired recovery rate; she may be for example between +10 and -60 ° C and preferably between -10 and -40 ° C for a gas pressure for example between 0.1 and 25 MPa and preferably between 0.2 and 10 MPa.
- This refrigeration can be provided either by an external refrigeration cycle, or by other means such as for example the expansion of the gas in a turbine or an expansion valve.
- the dehydrated gas leaving the cooling step (c) can be subjected to an additional treatment.
- the solvent leaving this washing zone can then be regenerated by lowering the pressure and / or heating and recycled.
- the at least partially dehydrated and deacidified gas is withdrawn.
- the contact zone used during step (a) can be achieved by means of a column of plates or of a column with packing.
- Different packings can be used, in particular so-called “structured” packings which are regularly arranged in the contact zone. It will also be possible to use packings formed from metallic fabrics assembled in the form of cylindrical buffers with a diameter equal to the internal diameter of the contact column.
- any other device known to a person skilled in the art making it possible to make such contact between the liquid phase and the gas phase can also be used.
- a device can for example be constituted by a centrifugal contactor in which the counter-current flow of the two phases takes place no longer under the effect of gravity but under the effect of a centrifugal force, with a view to make a device contact of a reduced volume.
- a natural gas is produced on a site, it enters the method according to the invention through line 1. Its pressure is 7.5 MPa (abs) and its temperature is 40 ° C; its composition is given in table 1 and it is saturated with water. Its flow is 123 tonnes / h, which corresponds to 3.5 MNm3 / day.
- the head gas phase is transported in line 3 which is an underwater pipeline 0.25 m in diameter over a distance of 11.2 km and arrives via line 5 at the receiving terminal where its pressure is 6.95 MPa due to the pressure drop in the pipeline.
- the gas is cooled to a temperature of -15 ° C in the heat exchanger E1 by a refrigerant external to the process; this cooling causes partial condensation of the gas.
- the cooled mixture leaving the exchanger E1 via line 6 consists of the non-condensed gas and, on the one hand, 226 kg / h of an aqueous liquid phase of a mixture of water, methanol and triethylamine, d 'other part of 410 kg / h of a liquid hydrocarbon phase.
Landscapes
- Engineering & Computer Science (AREA)
- Mechanical Engineering (AREA)
- General Engineering & Computer Science (AREA)
- Mining & Mineral Resources (AREA)
- Life Sciences & Earth Sciences (AREA)
- Geology (AREA)
- Fluid Mechanics (AREA)
- Environmental & Geological Engineering (AREA)
- Physics & Mathematics (AREA)
- General Life Sciences & Earth Sciences (AREA)
- Geochemistry & Mineralogy (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Pipeline Systems (AREA)
- Devices And Processes Conducted In The Presence Of Fluids And Solid Particles (AREA)
- Separation By Low-Temperature Treatments (AREA)
Claims (22)
- Verfahren zum Behandeln und Transportieren eines Erdgases, das aus mindestens einem Produktions-Bohrloch stammt, zu einem Empfangs- und Behandlungs-Terminal, dadurch gekennzeichnet, daß es die folgenden Stufen umfaßt :a) man kontaktiert unter geeigneten Kontaktierbedingungen mindestens einen Teil des genannten Gases, das mindestens mit Wasser gesättigt ist, in mindestens einer Kontaktzone (G₁) mit einer flüssigen Phase (4), die mindestens zum Teil aus einer Recyclisierung (der nachstehend beschriebenen Stufe (e)) stammt und gleichzeitig Wasser und mindestens ein Antihydrat-Additiv enthält, bei dem es sich um eine von Wasser verschiedene, normalerweise flüssige Nicht-Kohlenwasserstoff-Verbindung handelt, die mindestens teilweise mit Wasser mischbar ist und in reinem Zustand oder in Form eines Azeotrops bei einer Temperatur unterhalb der Verdampfungstemperatur des Wassers in der Weise verdampft, daß man eine wäßrige flüssige Phase (2), die im wesentlichen kein Additiv enthält, verglichen mit der genannten recyclisierten flüssigen Phase, und eine gasförmige Phase (3), die den Wasserdampf und praktisch das gesamte Additiv enthält, erhält;b) man transportiert die genannte gasförmige Phase der Stufe (a) unter geeigneten Transportbedingungen in einer Leitung (3) in mindestens eine Wärmeaustauschzone (E₁) des genannten Terminals;c) man kühlt die genannte gasförmige Phase, die aus der Stufe (b) stammt, unter geeigneten Bedingungen in der Wärmeaustauschzone so ab, daß sie teilweise kondensiert und man ein nicht-kondensiertes Gas erhält, wobei das erhaltene Kondensat mindestens eine wäßrige Phase umfaßt, die mindestens einen Teil des genannten Additivs enthält;d) man trennt die wäßrige Phase von dem nicht-kondensierten Gas unter geeigneten Bedingungen in einer Trennzone (B₁) und zieht das nicht-kondensierte genannte Gas (10) ab; unde) man recyclisiert die wäßrige Phase der Stufe (d) in die Stufe (a), indem man sie in einer anderen Leitung (8, 9) in die Kontaktzone transportiert.
- Verfahren nach Anspruch 1, in dem der Gewichtsmengenanteil des Antihydrat-Additivs in der recyclisierten flüssigen Phase 10 bis 70 %, vorzugsweise 20 bis 50 %, beträgt.
- Verfahren nach Anspruch 1, in dem man das genannte Gas mit der recyclisierten flüssigen Phase in Kontakt bringt, die außerdem mindestens ein Antikorrosions-Additiv enthält, bei dem es sich um eine von Wasser verschiedene, normalerweise flüssige Nicht-Kohlenwasserstoff-Verbindung handelt, die mindestens teilweise mit Wasser mischbar oder in Wasser dispergierbar ist und in reinem Zustand oder in Form eines Azeotrops bei einer Temperatur unterhalb der Verdampfungstemperatur des Wassers verdampft.
- Verfahren nach Anspruch 1, in dem man das genannte Gas mit der recyclisierten flüssigen Phase in Kontakt bringt, die außerdem mindestens ein Antikorrosions-Additiv enthält, bei dem es sich um eine von Wasser verschiedene, normalerweise flüssige Nicht-Kohlenwasserstoff-Verbindung handelt, die in Wasser dispergierbar ist, in dem es von der aus der Stufe (a) stammenden wäßrigen Phase durch eine zusätzliche Trennstufe unter geeigneten Trennbedingungen abgetrennt und mit der aus der Stufe (a) stammenden gasförmigen Phase wieder gemischt wird.
- Verfahren nach Anspruch 3 oder 4, in dem die Gewichtsmengenanteile der recyclisierten flüssigen Phase die folgenden sind :- 0,1 bis 5 %, vorzugsweise 0,3 bis 1 %, Antikorrosions-Additiv- 10 bis 70 %, vorzugsweise 20 bis 50 %, Antihydrat-Additiv- 29,9 bis 89,9 %, vorzugsweise 49,7 bis 79,7 %, Wasser.
- Verfahren nach einem der Ansprüche 1 bis 5, in dem in der Stufe (a) der Mengenanteil der recyclisierten flüssigen Phase, bezogen auf die Massendurchflußmenge des aus dem Bohrloch austretenden Gases, 0,05 bis 5 Gew.-%, vorzugsweise 0,1 bis 1 Gew.-%, beträgt, wobei die Temperatur im wesentlichen zwischen 20 und 100°C liegt und der Druck bei 0,1 bis 25 MPa liegt.
- Verfahren nach einem der Ansprüche 1 bis 6, dadurch gekennzeichnet, daß im Verlaufe der Stufe (c) das Kondensat eine wäßrige Phase und eine flüssige Kohlenwasserstoffphase umfaßt, wobei die Kohlenwasserstoffphase im Verlaufe der Stufe (d) durch Dekantieren von der wäßrigen Phase getrennt und abgezogen wird.
- Verfahren nach einem der Ansprüche 1 bis 7, dadurch gekennzeichnet, daß man das aus dem Produktions-Bohrloch austretende Gas in mindestens zwei Fraktionen aufteilt, wobei eine erste Fraktion A des genannten Gases der Stufe (a) unterworfen wird und eine zweite Fraktion B, die nicht der Stufe (a) unterworfen wird, mit der aus der Stufe (a) austretenden gasförmigen Phase gemischt wird.
- Verfahren nach einem der Ansprüche 1 bis 8, dadurch gekennzeichnet, daß das genannte Produktionsgas von mindestens zwei verschiedenen Bohrlöchern produziert wird und daß die Stufe (a) in mindestens zwei unterschiedlichen Kontaktzonen durchgeführt wird und daß die aus diesen genannten Kontaktzonen austretenden gasförmigen Phasen vor Durchführung der Stufe (b) miteinander gemischt werden.
- Verfahren nach einem der Ansprüche 1 bis 9, dadurch gekennzeichnet, daß es sich bei dem Antihydrat-Additiv um mindestens eine Verbindung handelt, die ausgewählt wird aus der Gruppe, die gebildet wird von Methanol, Methylpropyläther, Ethylpropyläther, Dipropyläther, Methyl-tert-butyläther, Dimethoxymethan, Dimethoxyethan, Ethanol, Methoxyethanol und Propanol, wobei das bevorzugte Additiv Methanol ist.
- Verfahren nach einem der Ansprüche 3 bis 10, dadurch gekennzeichnet, daß es sich bei dem Antikorrosions-Additiv um mindestens eine Verbindung handelt, die ausgewählt wird aus der Gruppe, die gebildet wird von Diethylamin, Propylamin, Butylamin, Triethylamin, Dipropylamin, Ethylpropylamin, Ethanolamin, Cyclohexylamin, Pyridylmorpholin und Ethylendiamin.
- Verfahren nach einem der Ansprüche 1 bis 11, dadurch gekennzeichnet, daß die Abkühlungstemperatur der Stufe (c) zwischen +10 und -60°C, vorzugsweise zwischen -10 und -40°C, liegt.
- Verfahren nach einem der Ansprüche 1 bis 12, dadurch gekennzeichnet, daß das aus dem Produktions-Bohrloch austretende Gas ein Kohlenwasserstoff-Kondensat enthält, das vor Durchführung der Stufe (a) in einer Trennzone abgetrennt wird und daß man die aus der genannten Trennung resultierende gasförmige Phase in die Kontaktzone einführt.
- Verfahren nach Anspruch 13, dadurch gekennzeichnet, daß das Kohlenwasserstoff-Kondensat und die gasförmige Phase, die aus der Stufe (a) austreten, vor Durchführung der Stufe (b) wieder miteinander gemischt werden, und daß die Stufe (b) in einem 2-Phasen-Zustand durchgeführt wird.
- Verfahren nach einem der Ansprüche 1 bis 14, dadurch gekennzeichnet, daß die Stufe (a) unter dem Meer durchgeführt wird, wobei das Gas im Verlauf der Stufe (b) durch eine Unterwasser-Leitung transportiert wird.
- Verfahren nach einem der Ansprüche 1 bis 15, dadurch gekennzeichnet, daß das aus der Stufe (d) austretende Gas einer ergänzenden Behandlung unterworfen wird durch Waschen in der Kälte mit einem Lösungsmittel, das als Additiv im Verlaufe der Stufe (a) verwendet wird, um mindestens einen Teil der in dem genannten Gas enthaltenen sauren Gase zu eliminieren.
- Vorrichtung zum Transportieren und Behandeln eines Erdgases, dadurch gekennzeichnet, daß sie in Kombination umfaßt:- mindestens einen Behälter (G₁), in dem das Erdgas unter Druck und vorzugsweise im Gegenstrom mit einer wäßrigen flüssigen Phase in Kontakt gebracht wird, die mindestens ein Additiv enthält, der ein erstes Ende und ein zweites Ende aufweist,- Mittel (1) zur Einführung des genannten Gases, die mit dem zweiten Ende des Behälters in Verbindung stehen, und gegebenenfalls Transportmittel (3, 5), wie sie nachstehend definiert werden;- Mittel (4) zur Einführung der genannten wäßrigen flüssigen Phase, die mindestens ein Additiv enthält, die Recyclisierungs-Mittel (P₁, 9, 8) der genannten flüssigen Phase mit dem ersten Ende des Behälters verbinden,- Mittel (2) zum Abziehen einer wäßrigen flüssigen Phase, die praktisch kein Additiv enthält, die mit dem zweiten Ende des Behälters verbunden sind,- Mittel zum Abziehen und zum Transport (3, 5) einer unter Druck stehenden gasförmigen Phase, die praktisch das gesamte Additiv enthält, die das erste Ende des Behälters (G₁) mit unter Druck stehenden Wärmeaustausch-Mitteln E₁ verbinden,- Mittel (B₁) zur Trennung einer wäßrigen flüssigen Phase von dem nicht-kondensierten und behandelten Gas, die mit den Wärmeaustausch-Mitteln E₁ verbunden sind,- Mittel (10) zur Rückgewinnung des nicht-kondensierten und behandelten Gases, die mit den Trenn-Mitteln (B₁) verbunden sind,- Mittel (8) zum Abziehen der wäßrigen Phase, die mit den Trenn-Mitteln verbunden sind, und- Mittel (P₁, 9, 8) zur Recyclisierung der wäßrigen Phase, die mit Abzugs-Mitteln verbunden sind, die Mittel (4) zur Einführung der wäßrigen flüssigen Phase enthalten, die mit dem ersten Ende des Behälters (G₁) verbunden sind.
- Vorrichtung nach Anspruch 17, die aufweist Mittel zur Trennung des Erdgases von Kondensaten, die mit Mitteln (1) zur Einführung des Gases verbunden sind, die einen ersten Ausgang (30) zum Abziehen einer wäßrigen Phase, einen zweiten Ausgang (31) zum Abziehen des zu behandelnden Gases, der mit dem zweiten Ende des Behälters G₁) verbunden ist, und einen dritten Ausgang für ein Kohlenwasserstoff-Kondensat aufweisen, der entweder mit den Transportmitteln (3, 5) oder mit einem Empfangs-Terminal oder mit Transportmitteln (3, 5) und dem Terminal verbunden ist.
- Vorrichtung nach Anspruch 17 oder 18, die einen komplementären Separator S₁ für Wasser und das Additiv aufweist, der mit den Mitteln (2) zum Abziehen der wäßrigen flüssigen Phase verbunden ist, die einen Ausgang (40) zum Abziehen von Wasser und einen Ausgang (41, 42) zum Abziehen des Additivs aufweist, der mit den Transport-Mitteln (3, 5) verbunden ist.
- Vorrichtung nach einem der Ansprüche 16 bis 19, die Mittel (11) zur Additiv-Zufuhr aufweist, die mit den Recyclisierungs-Mitteln (P₁, 9, 4) in Verbindung stehen.
- Vorrichtung nach einem der Ansprüche 16 bis 19, die Mittel zum Waschen des behandelten Gases aufweist, die mit Trennmitteln (B₁) verbunden sind.
- Verwendung der Vorrichtung nach einem der Ansprüche 16 bis 21 bei der Verwendung und Regenerierung von Additiven für den Transport und die Behandlung eines Erdgases.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| FR9000757A FR2657416B1 (fr) | 1990-01-23 | 1990-01-23 | Procede et dispositif pour le transport et le traitement d'un gaz naturel. |
| FR9000757 | 1990-01-23 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP0442767A1 EP0442767A1 (de) | 1991-08-21 |
| EP0442767B1 true EP0442767B1 (de) | 1994-07-20 |
Family
ID=9393030
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP91400092A Expired - Lifetime EP0442767B1 (de) | 1990-01-23 | 1991-01-16 | Verfahren und Vorrichtung zum Behandeln eines natürlichen Gases |
Country Status (9)
| Country | Link |
|---|---|
| US (1) | US5127231A (de) |
| EP (1) | EP0442767B1 (de) |
| JP (1) | JP3074394B2 (de) |
| AU (1) | AU640988B2 (de) |
| CA (1) | CA2034806C (de) |
| DE (1) | DE69102899T2 (de) |
| FR (1) | FR2657416B1 (de) |
| MY (1) | MY106171A (de) |
| NO (1) | NO176534C (de) |
Families Citing this family (35)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2691503B1 (fr) * | 1992-05-20 | 1997-07-25 | Inst Francais Du Petrole | Procede pour le traitement et le transport d'un gaz naturel sortant d'un puits de gaz. |
| US5420370A (en) * | 1992-11-20 | 1995-05-30 | Colorado School Of Mines | Method for controlling clathrate hydrates in fluid systems |
| US5432292A (en) * | 1992-11-20 | 1995-07-11 | Colorado School Of Mines | Method for controlling clathrate hydrates in fluid systems |
| US5639925A (en) * | 1992-11-20 | 1997-06-17 | Colorado School Of Mines | Additives and method for controlling clathrate hydrates in fluid systems |
| WO1994024413A1 (en) * | 1993-04-08 | 1994-10-27 | Bp Chemicals Limited | Method for inhibiting solids formation and blends for use therein |
| US5460728A (en) * | 1993-12-21 | 1995-10-24 | Shell Oil Company | Method for inhibiting the plugging of conduits by gas hydrates |
| FR2715692B1 (fr) * | 1993-12-23 | 1996-04-05 | Inst Francais Du Petrole | Procédé de prétraitement d'un gaz naturel contenant de l'hydrogène sulfure. |
| US5648575A (en) * | 1995-01-10 | 1997-07-15 | Shell Oil Company | Method for inhibiting the plugging of conduits by gas hydrates |
| AR001674A1 (es) * | 1995-04-25 | 1997-11-26 | Shell Int Research | Método para inhibir la obstrucción de conductos por hidrato de gas |
| FR2735210B1 (fr) * | 1995-06-06 | 1997-07-18 | Inst Francais Du Petrole | Procede de recyclage d'un additif dispersant utilise pour le transport d'un gaz a condensat ou d'un petrole avec gaz associe en presence d'hydrates |
| FR2735211B1 (fr) * | 1995-06-06 | 1997-07-18 | Inst Francais Du Petrole | Procede de transport d'un fluide tel un gaz sec, susceptible de former des hydrates |
| FR2753720B1 (fr) * | 1996-09-24 | 1998-11-27 | Procede de deshydratation et de degazolinage d'un gaz, comportant un etage de refroidissement preliminaire | |
| FR2753719B1 (fr) * | 1996-09-24 | 1998-11-27 | Procede de deshydratation et de degazolinage d'un gaz, comportant deux etapes complementaires de regeneration du solvant | |
| DE19709373A1 (de) † | 1997-03-07 | 1998-09-10 | Manfred Veenker | Leitung für gefährdende Fluide und Verfahren zu deren Herstellung |
| US5853458A (en) * | 1997-04-28 | 1998-12-29 | Gavlin Associates, Inc | Glycol solvents and method thereof |
| GB2366802B (en) * | 1997-06-17 | 2002-07-03 | Inst Francais Du Petrole | Process for degasolining a gas containing condensable hydrocarbons |
| FR2764609B1 (fr) * | 1997-06-17 | 2000-02-11 | Inst Francais Du Petrole | Procede de degazolinage d'un gaz contenant des hydrocarbures condensables |
| US6153100A (en) * | 1998-12-30 | 2000-11-28 | Phillips Petroleum Company | Removing iron salts from NGL streams |
| US6177597B1 (en) * | 1999-07-06 | 2001-01-23 | Gavlin Associates, Inc. | Glycol solvents and process |
| CA2391447C (en) * | 1999-11-24 | 2010-10-19 | Shell Canada Limited | A method for recovering water soluble surfactants |
| CN100510000C (zh) * | 2001-08-15 | 2009-07-08 | 协同化学公司 | 降低管线中硫化铁沉积物的方法和组合物 |
| US6688324B2 (en) * | 2002-01-08 | 2004-02-10 | Cooper Cameron Corporation | Valve for hydrate forming environments |
| RU2209363C1 (ru) * | 2002-06-17 | 2003-07-27 | Общество с ограниченной ответственностью НТЦ "Адгезивнефтегаз" | Способ регенерации ингибиторов осушки и очистки природного газа |
| WO2004038279A2 (en) * | 2002-10-23 | 2004-05-06 | Saudi Arabian Oil Company | Controlled superheating of natural gas for transmission |
| WO2006110192A1 (en) * | 2005-04-07 | 2006-10-19 | Exxonmobil Upstream Research Company | Recovery of kinetic hydrate inhibitor |
| US7875103B2 (en) * | 2006-04-26 | 2011-01-25 | Mueller Environmental Designs, Inc. | Sub-micron viscous impingement particle collection and hydraulic removal system |
| GB2447027A (en) * | 2006-09-21 | 2008-09-03 | Statoil Asa | Prevention of solid gas hydrate build-up |
| JP2008255364A (ja) * | 2008-06-19 | 2008-10-23 | Japan Energy Corp | 自動車用液化石油ガス組成物 |
| FR2939694B1 (fr) * | 2008-12-16 | 2010-12-17 | Inst Francais Du Petrole | Procede de deshydratation partielle d'un gaz par absorption sur un solvant regenerable par demixtion a temperature ambiante |
| US20110259794A1 (en) * | 2010-04-23 | 2011-10-27 | Chevron U.S.A. Inc. | Removing chlathrate inhibitors from contaminated petroleum streams |
| GB2507429B8 (en) | 2011-07-01 | 2021-01-06 | Equinor Energy As | A method and system for lowering the water dew point of a hydrocarbon fluid stream subsea |
| US8940067B2 (en) | 2011-09-30 | 2015-01-27 | Mueller Environmental Designs, Inc. | Swirl helical elements for a viscous impingement particle collection and hydraulic removal system |
| CN104812876B (zh) | 2012-11-26 | 2019-04-02 | 挪威国家石油公司 | 自井流的结合的气体脱水和液体抑制 |
| GB2526604B (en) | 2014-05-29 | 2020-10-07 | Equinor Energy As | Compact hydrocarbon wellstream processing |
| US9334722B1 (en) * | 2015-11-18 | 2016-05-10 | Mubarak Shater M. Taher | Dynamic oil and natural gas grid production system |
Family Cites Families (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3330124A (en) * | 1963-07-05 | 1967-07-11 | Lummus Co | Process for removal of water from light hydrocarbon fluid mixtures by distillation |
| US3262278A (en) * | 1963-08-19 | 1966-07-26 | Exxon Research Engineering Co | Increased ethylene recovery by ethane addition |
| GB1131003A (en) * | 1967-02-24 | 1968-10-16 | Shell Int Research | Process and apparatus for the dehydration of a gas |
| US3899312A (en) * | 1969-08-21 | 1975-08-12 | Linde Ag | Extraction of odorizing sulfur compounds from natural gas and reodorization therewith |
| US3925047A (en) * | 1970-12-24 | 1975-12-09 | Phillips Petroleum Co | Removal of moisture from a natural gas stream by contacting with a liquid desiccant-antifreeze agent and subsequently chilling |
| US4132535A (en) * | 1976-11-17 | 1979-01-02 | Western Chemical Company | Process for injecting liquid in moving natural gas streams |
| US4416333A (en) * | 1982-04-20 | 1983-11-22 | Shell Oil Company | Corrosion inhibiting process for a remotely located deep corrosive gas well |
| FR2570162B1 (fr) * | 1984-09-07 | 1988-04-08 | Inst Francais Du Petrole | Procede et dispositif de compression et de transport d'un gaz contenant une fraction liquide |
| FR2618876B1 (fr) * | 1987-07-30 | 1989-10-27 | Inst Francais Du Petrole | Procede de traitement et de transport d'un gaz contenant du methane et de l'eau |
-
1990
- 1990-01-23 FR FR9000757A patent/FR2657416B1/fr not_active Expired - Lifetime
-
1991
- 1991-01-16 DE DE69102899T patent/DE69102899T2/de not_active Expired - Fee Related
- 1991-01-16 EP EP91400092A patent/EP0442767B1/de not_active Expired - Lifetime
- 1991-01-21 NO NO910225A patent/NO176534C/no not_active IP Right Cessation
- 1991-01-22 US US07/643,620 patent/US5127231A/en not_active Expired - Lifetime
- 1991-01-22 MY MYPI91000096A patent/MY106171A/en unknown
- 1991-01-23 CA CA002034806A patent/CA2034806C/fr not_active Expired - Lifetime
- 1991-01-23 JP JP03006275A patent/JP3074394B2/ja not_active Expired - Fee Related
- 1991-02-12 AU AU70949/91A patent/AU640988B2/en not_active Expired
Also Published As
| Publication number | Publication date |
|---|---|
| AU7094991A (en) | 1991-08-15 |
| FR2657416A1 (fr) | 1991-07-26 |
| JP3074394B2 (ja) | 2000-08-07 |
| AU640988B2 (en) | 1993-09-09 |
| MY106171A (en) | 1995-03-31 |
| US5127231A (en) | 1992-07-07 |
| NO176534C (no) | 1995-04-19 |
| DE69102899T2 (de) | 1994-11-17 |
| NO176534B (no) | 1995-01-09 |
| CA2034806A1 (fr) | 1991-07-24 |
| DE69102899D1 (de) | 1994-08-25 |
| NO910225L (no) | 1991-07-24 |
| CA2034806C (fr) | 2002-03-19 |
| FR2657416B1 (fr) | 1994-02-11 |
| JPH0586379A (ja) | 1993-04-06 |
| EP0442767A1 (de) | 1991-08-21 |
| NO910225D0 (no) | 1991-01-21 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0442767B1 (de) | Verfahren und Vorrichtung zum Behandeln eines natürlichen Gases | |
| CA2096714C (fr) | Procede pour le traitement et le transport d'un gaz naturel sortant d'un puits de gaz | |
| CA2239758C (fr) | Procede de degazolinage d'un gaz contenant des hydrocarbures condensables | |
| CN104812876B (zh) | 自井流的结合的气体脱水和液体抑制 | |
| EP0783031B1 (de) | Verfahren zur Entfernung von Wasser, Saüren und Benzin aus Erdgas, unter Verwendung eines Lösungsmittelgemisches | |
| EP0770667B1 (de) | Verfahren zur Trocknung von Gasen mittels Glycol mit anschliessender Raffinierung von gasförmigen Abflüssen | |
| EP0796134B1 (de) | Verfahren zum behandeln von erdgas, das wasser und kondensierbare kohlenwasserstoffe enthält | |
| EP0267819B1 (de) | Integrierter Prozess zur Behandlung eines feuchten methanhaltigen Gases mit dem Ziel, das Wasser daraus zu entfernen | |
| EP2878589B1 (de) | Flexibles verfahren für die aufbereitung von lösungsmitteln, das zum abbau von erdgas verwendet wird | |
| FR2814379A1 (fr) | Procede de desacidification d'un gaz par absorption dans un solvant avec un controle de la temperature | |
| FR2814378A1 (fr) | Procede de pretraitement d'un gaz naturel contenant des gaz acides | |
| FR2618876A1 (fr) | Procede de traitement et de transport d'un gaz contenant du methane et de l'eau | |
| FR2760653A1 (fr) | Procede de desacidification avec production de gaz acides en phase liquide | |
| CA2443009A1 (fr) | Procede de desacidification d`un gaz naturel | |
| EP0835921B1 (de) | Verfahren zur Entfernung von Wasser und Benzin aus einem Gas unter Verwendung einer Vorkühlung | |
| JPH10102076A (ja) | 二つの補助溶媒再生工程から成る、ガスの脱水および脱ガスの方法 | |
| EP0768106B1 (de) | Verfahren zur Fraktionierung eines mehrere trennbare Komponenten enthaltenden Fluids wie z.B. Erdgas | |
| FR2914684A1 (fr) | Procede de recyclage d'agents retardateurs de formation d'hydrates | |
| WO2013076433A1 (fr) | Procédé de traitement d'effluent gazeux en tête de distillation atmosphérique |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): DE GB IT NL |
|
| 17P | Request for examination filed |
Effective date: 19920103 |
|
| 17Q | First examination report despatched |
Effective date: 19930506 |
|
| ITF | It: translation for a ep patent filed | ||
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): DE GB IT NL |
|
| REF | Corresponds to: |
Ref document number: 69102899 Country of ref document: DE Date of ref document: 19940825 |
|
| GBT | Gb: translation of ep patent filed (gb section 77(6)(a)/1977) |
Effective date: 19940819 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed | ||
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: IF02 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 20030205 Year of fee payment: 13 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20040803 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES;WARNING: LAPSES OF ITALIAN PATENTS WITH EFFECTIVE DATE BEFORE 2007 MAY HAVE OCCURRED AT ANY TIME BEFORE 2007. THE CORRECT EFFECTIVE DATE MAY BE DIFFERENT FROM THE ONE RECORDED. Effective date: 20050116 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: NL Payment date: 20090127 Year of fee payment: 19 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20090122 Year of fee payment: 19 |
|
| REG | Reference to a national code |
Ref country code: NL Ref legal event code: V1 Effective date: 20100801 |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 20100116 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20100801 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20100116 |