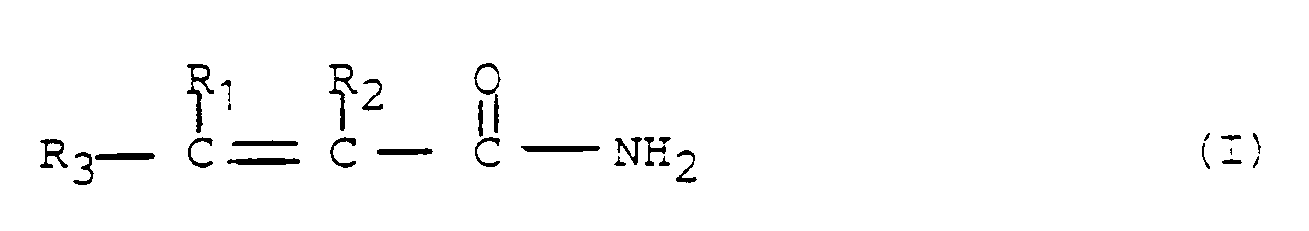EP0447967B1 - Gélification de polymères contenant un acrylamide par l'alcool furfurylique et des aldéhydes hydrodispersibles - Google Patents
Gélification de polymères contenant un acrylamide par l'alcool furfurylique et des aldéhydes hydrodispersibles Download PDFInfo
- Publication number
- EP0447967B1 EP0447967B1 EP91103945A EP91103945A EP0447967B1 EP 0447967 B1 EP0447967 B1 EP 0447967B1 EP 91103945 A EP91103945 A EP 91103945A EP 91103945 A EP91103945 A EP 91103945A EP 0447967 B1 EP0447967 B1 EP 0447967B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- acid
- sodium
- water
- acrylamide
- ammonium
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
- 0 CC(CN*)=C Chemical compound CC(CN*)=C 0.000 description 2
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K8/00—Compositions for drilling of boreholes or wells; Compositions for treating boreholes or wells, e.g. for completion or for remedial operations
- C09K8/60—Compositions for stimulating production by acting on the underground formation
- C09K8/84—Compositions based on water or polar solvents
- C09K8/86—Compositions based on water or polar solvents containing organic compounds
- C09K8/88—Compositions based on water or polar solvents containing organic compounds macromolecular compounds
- C09K8/887—Compositions based on water or polar solvents containing organic compounds macromolecular compounds containing cross-linking agents
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K8/00—Compositions for drilling of boreholes or wells; Compositions for treating boreholes or wells, e.g. for completion or for remedial operations
- C09K8/02—Well-drilling compositions
- C09K8/04—Aqueous well-drilling compositions
- C09K8/06—Clay-free compositions
- C09K8/12—Clay-free compositions containing synthetic organic macromolecular compounds or their precursors
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K8/00—Compositions for drilling of boreholes or wells; Compositions for treating boreholes or wells, e.g. for completion or for remedial operations
- C09K8/50—Compositions for plastering borehole walls, i.e. compositions for temporary consolidation of borehole walls
- C09K8/504—Compositions based on water or polar solvents
- C09K8/506—Compositions based on water or polar solvents containing organic compounds
- C09K8/508—Compositions based on water or polar solvents containing organic compounds macromolecular compounds
- C09K8/512—Compositions based on water or polar solvents containing organic compounds macromolecular compounds containing cross-linking agents
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10S—TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10S507/00—Earth boring, well treating, and oil field chemistry
- Y10S507/903—Crosslinked resin or polymer
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10S—TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10S507/00—Earth boring, well treating, and oil field chemistry
- Y10S507/935—Enhanced oil recovery
Definitions
- the present invention relates to a novel process for crosslinking water-soluble polymers utilizing an organic crosslinking system composed of a furan derivative and a water dispersible aldehyde.
- gelled or crosslinked water-soluble polymers are useful in enhanced oil recovery operations. They have been used to alter the permeability of underground formations in order to enhance the effectiveness of water flooding operations. Polymers along with an appropriate crosslinking system are injected in an aqueous solution into the formation. The polymers then permeate into and gel in the regions having the highest water permeability. Any fluids injected into the formation in subsequent water flooding operations, will then be diverted away from the regions in which the gel formed to areas of the formation now having a higher water permeability.
- EP-A-0 180 406 discloses a method of treating a subterranean formation for reducing its permeability by contacting it with an aqueous polymerizable composition comprising a polymerization monomer, a polymerization catalyst having a latent period, and an alcoholic compound which is a cyclic ether containing a hydroxymethylene substituent on a carbon atom bonded to the ether oxygen, or an aliphatic alcohol having a carbon-to-carbon double bond or a carbon-to-carbon triple bond.
- the organic crosslinking system of the present invention is formed from:
- Furan derivatives suitable for use in the present invention are of the formula: wherein R9, R10, R11, and R12, are independently selected from hydrogen, methyl, hydroxyl, carbinol, carboxyl, -CH2COOH, acyl amide, mercaptan and primary amine radicals, and at least one R9, R10, R11 and R12 radical must be selected from hydroxyl, carbinol, carboxyl, -CH2COOH, acyl amide, mercaptan and primary amine radicals.
- Suitable furan derivatives include but are not limited to those selected from furfuryl acetate, furanamine, furandiamine, furantriamine, furantetramine, furancarbinol, furandicarbinol (furandimethanol), furantricarbinol, furantetracarbinol, hydroxyfuran, dihydroxyfuran, trihydroxyfuran, tetrahydroxyfuran, furancarboxylic acid, furandicarboxylic acid, furantricarboxylic acid, furantetracarboxylic acid, furanmethylenecarboxylic acid, furanbis(methylenecarboxylic acid), furantris(methylenecarboxylic acid) and furantetrakis(methylenecarboxylic acid), mercaptofuran, and combinations of two or more thereof.
- the preferred furan derivative for the practice of the present invention is 2-furancarbinol, furfuryl alcohol mercaptofuran, furandicarbinol, furfuryl acetate and furan
- suitable aldehyde or aldehyde generating compounds can be selected from aliphatic aldehydes, aliphatic dialdehydes, and aromatic aldehydes, aromatic dialdehydes.
- Preferred aldehydes or aldehyde generating compounds can be selected from formaldehyde, paraformaldehyde, acetaldehyde, propionaldehyde, butyraldehyde, isobutyraldehyde, valeraldehyde, heptaldehyde, decanal, glyoxal, glutaraldehyde, terephthaldehyde, hexamethylenetetramine and mixtures thereof.
- the amount of furan derivative used in preparing the gelled compositions of the invention will be in the range of 0.005 to 5.0 wt.%, preferably from in the range of 0.01 to 2.0 wt.% and most preferably from in the range of 0.05 to 1.0 wt% based on the total weight of the gelable composition.
- the amount of aldehyde or aldehyde generating compound used will be in the range of 0.005 to 5.0 wt.%, preferably in the range of 0.01 to 2.0 wt.% and most preferably in the range of 0.05 to 1.0 wt% based on the total weight of the gelable composition.
- the molar ratio of furan derivative to aldehyde or aldehyde generating compound will be in the broad range of 30:1 to 1:30 with a more preferred range of 2:1 to 1:4.
- water-soluble polymer As used in this application, the term water-soluble polymer, copolymers, and terpolymers refers to those polymers which are truly water-soluble or those which are dispersible in water or other aqueous medium to form a stable colloidal suspension which can be pumped into a formation and gelled therein.
- the water-soluble polymers which are suitable for use in the present invention include those which contain from 5 to 100 mole percent of at least one monomer of the formula: wherein R1, R2, and R3 are independently selected from hydrogen and alkyl radicals containing from 1 to 3 carbon atoms, of which acrylamide and methacrylamide are the preferred examples; and from 0 to 95 mole percent of at least one monomer selected from
- the presently preferred class of water-soluble polymers are those selected from homopolymers of acrylamide, homopolymers of methacrylamide, copolymers of acrylamide and acrylic acid, copolymers of acrylamide and sodium acrylate, copolymers of acylamide and N-vinyl-2-pyrrolidone, copolymers of acrylamide and sodium 2-acrylamido-2-methylpropane sulfonate, copolymers of acrylamide and 2-acrylamido-2-ethylpropane sulfonic acid, terpolymers of acrylamide, N-vinyl-2-pyrrolidone, and sodium 2-acrylamido-2-methylpropane sulfonate and terpolymers of acrylamide, N-vinyl-2-pyrrolidone and 2-acrylamido-2-methylpropane sulfonic acid.
- the ratio of the monomers in the above-described polymers is not critical; provided however, that at least 5 mole percent of acrylamide is present in the above-described polymers.
- Particularly preferred are terpolymers of N-vinyl-2-pyrrolidone, acrylamide and sodium 2-acrylamido-2-methylpropane sulfonate wherein said monomers are provided in a 30 to 15 to 55 weight percent terpolymer. Additionally within the scope of this invention is the use of combinations of copolymers and terpolymers utilizing the above listed monomers.
- gelled polymers to alter the water permeability of underground formations is well known to those skilled in the art.
- an aqueous solution containing the polymer and a crosslinking agent is transmitted into the formation from a well in fluid communication with the formation so that the aqueous solution can diffuse into the more water permeable portions of the formation and alter that water permeability by gelling therein.
- the present invention can be used in a similar manner.
- An aqueous solution containing the water-soluble polymer, aldehyde or aldehyde generating compound, and furan derivative is pumped into the formation so that it alters the water permeability of the formation in a similar manner when gelation takes place.
- the aqueous solution containing the water soluble polymer, aldehyde or aldehyde generating compound and furan derivative can be pumped into the formation in one slug, or alternatively the components may be sequentially injected in a manner to provide an appropriate concentration in the formation.
- the present invention's advantage lies in the fact that the onset of gelation is delayed allowing the aqueous solution to penetrate further into the formation.
- the nature of the underground formation treated is not critical to the practice of the present invention.
- the organic crosslinkers will gel the water-soluble polymers in fresh water, salt water, or brines, as well as at a temperature range of from 93 to 205°C (200 to 400°F).
- the purpose of this example is to demonstrate the delayed gelation of a water-soluble polymer by a furan derivate and an aldehyde. Additionally this example demonstrates the long term stability of a gel formed with a water-soluble polymer, a furan derivative and an aldehyde.
- a one percent solution of a terpolymer composed of 30 wt % of N-vinyl-2-pyrrolidone, 15 wt % of acrylamide, and 55 wt % of sodium 2-acrylamido-2-methylpropane sulfonate was prepared in the following manner. 16.3 ml of an inverse emulsion which contained 32.4 wt % of the above-described active terpolymer was mixed with 500 ml of synthetic sea water.
- the synthetic water used had the following formula:
- 0.18 ml of furfuryl alcohol was added to 200 ml of the polymer and synthetic sea water mixture described above, to provide a 1000 ppm furfuryl alcohol concentration.
- 0.36 ml of furfuryl alcohol was added to 200 ml of the polymer and synthetic sea water mixture to generate a 2000 ppm furfuryl alcohol concentration.
- the ten ampules were sealed under nitrogen and then placed upright in compartmented aluminum boxes with relief holes and placed in an oven and heated to 121°C (250°F). Periodically, the ampules were removed from the oven and the mechanical strength of the gel was determined behind a protective shield.
- the purpose of this example is to demonstrate the delayed gelation of a water-soluble polymer by a furan derivative and an aldehyde. Additionally this example demonstrates the long term stability of a gel formed with a water-soluble polymer, a furan derivative and an aldehyde at high temperatures.
- ampules were prepared in a manner identical to those in Example I except that the aging was conducted at 149°C (300°F).
Landscapes
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Organic Chemistry (AREA)
- Compositions Of Macromolecular Compounds (AREA)
- Addition Polymer Or Copolymer, Post-Treatments, Or Chemical Modifications (AREA)
- Formation And Processing Of Food Products (AREA)
- Plural Heterocyclic Compounds (AREA)
Claims (8)
- Un procédé pour modifier la perméabilité d'une formation souterraine poreuse ayant une température ambiante supérieure à 93°C, caractérisé en ce qu'il consiste à introduire dans ladite formation soit indépendamment, soit en mélange :(a) 0,1 à 5 % d'un polymère soluble dans l'eau constitué de(a1) 100 à 5 moles % d'au moins un monomère de formule (I)
- Le procédé selon la revendication 1 dans lequel ledit polymère (a) soluble dans l'eau est choisi parmi les homopolymères d'acrylamide, les homopolymères de méthacrylamide, les copolymères d 'acrylamide et d'acide acrylique, les copolymères d'acrylamide et d'acrylate de sodium, les copolymères d'acrylamide et de N-vinyl-2-pyrrolidone, les copolymères d'acrylamide et de 2-acrylamido-2-méthylpropane sulfonate de sodium, les copolymères d'acrylamide et d'acide 2-acrylamido-2-méthylpropane sulfonique, les terpolymères de N-vinyl-2-pyrrolidone, d'acrylamide et de 2-acrylamido-2-méthylpropane sulfonate de sodium et les terpolymères d'acrylamide, de N-vinyl-2-pyrrolidone et d'acide 2-acrylamido-2-méthylpropane sulfonique.
- Procédé selon les revendications 1 ou 2 dans lequel le dérivé de furane (c) est choisi parmi l'acétate de furfuryle, la furanamine, la furandiamine, la furantriamine, la furantétramine, le furancarbinol, le furandicarbinol, le furantricarbinol, le furantétracarbinol, l'hydroxyfurane, le dihydroxyfurane, le trihydroxyfurane, le tétrahydroxyfurane, l'acide furancarboxylique, l'acide furandicarboxylique, l'acide furantricarboxylique, l'acide furantétracarboxylique, l'acide furanméthylènecarboxylique, le furanbis(acide méthylène carboxylique), le furantris(acide méthylène carboxylique), le furantétrakis(acide méthylène carboxylique), le mercaptofurane et des combinaisons de deux ou plusieurs de ceux-ci.
- Le procédé selon l'une quelconque des revendications précédentes dans lequel ledit aldéhyde ou ledit composé générant de l'aldéhyde (b) est choisi parmi le formaldéhyde, le paraformaldéhyde, l'acétaldéhyde, le propionaldéhyde, le butyraldéhyde, l'isobutyraldéhyde, le valéraldéhyde, l'heptaldéhyde, le décanal, le glyoxal, le glutaraldéhyde, le téréphtaldéhyde, l'hexaméthylènetétramine et leurs mélanges.
- Le procédé selon l'une quelconque des revendications précédentes dans lequel ledit polymère (a) soluble dans l'eau est un terpolymère de N-vinyl-2-pyrrolidone, d'acrylamide et de 2-acrylamido-2-méthylpropane sulfonate de sodium, ledit dérivé de furane (b) est l'alcool furfurylique et ledit aldéhyde ou composé générant de l'aldéhyde (c) est le formaldéhyde.
- Une composition de gel susceptible d'être obtenue par gélification des constituants suivants:(a) 0,1 à 5 % d'un polymère soluble dans l'eau constitué de :(a1) 100 à 5 moles % d'au moins un monomère de formule I
- La composition de gel selon la revendication 7 dans laquelle un ou plusieurs des constituants (a), (b), (c) et (d) sont comme définis dans l'une quelconque des revendications 2 à 6.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US07/493,665 US5043364A (en) | 1990-03-15 | 1990-03-15 | Gelation of acrylamide-containing polymers with furfuryl alcohol and water dispersible aldehydes |
| US493665 | 1990-03-15 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| EP0447967A2 EP0447967A2 (fr) | 1991-09-25 |
| EP0447967A3 EP0447967A3 (en) | 1991-11-27 |
| EP0447967B1 true EP0447967B1 (fr) | 1994-05-18 |
Family
ID=23961203
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP91103945A Expired - Lifetime EP0447967B1 (fr) | 1990-03-15 | 1991-03-14 | Gélification de polymères contenant un acrylamide par l'alcool furfurylique et des aldéhydes hydrodispersibles |
Country Status (9)
| Country | Link |
|---|---|
| US (1) | US5043364A (fr) |
| EP (1) | EP0447967B1 (fr) |
| AU (1) | AU625168B2 (fr) |
| CA (1) | CA2035423C (fr) |
| EG (1) | EG19032A (fr) |
| ES (1) | ES2053225T3 (fr) |
| MX (1) | MX171813B (fr) |
| NO (1) | NO301079B1 (fr) |
| NZ (1) | NZ237342A (fr) |
Families Citing this family (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5179136A (en) * | 1990-09-10 | 1993-01-12 | Phillips Petroleum Company | Gelatin of acrylamide-containing polymers with aminobenzoic acid compounds and water dispersible aldehydes |
| CA2091489C (fr) * | 1992-04-13 | 2001-05-08 | Ahmad Moradi-Araghi | Procede de gelification de polymeres solubles dans l'eau |
| US5447986A (en) * | 1992-06-29 | 1995-09-05 | Phillips Petroleum Company | Acceleration of gelation of water soluble polymers |
| US5957203A (en) * | 1992-08-31 | 1999-09-28 | Union Oil Company Of California | Ultra-high temperature stable gels |
| US5246073A (en) * | 1992-08-31 | 1993-09-21 | Union Oil Company Of California | High temperature stable gels |
| US5335733A (en) * | 1992-08-31 | 1994-08-09 | Union Oil Company Of California | Method for delaying gelation of a gelable polymer composition injected into a subterranean formation |
| US5617920A (en) * | 1992-08-31 | 1997-04-08 | Union Oil Company Of California | Method for modifying gelation time of organically crosslinked, aqueous gels |
| US5423380A (en) * | 1994-02-22 | 1995-06-13 | Phillips Petroleum Company | Process for treating oil-bearing formation |
| GB9426025D0 (en) * | 1994-12-22 | 1995-02-22 | Smith Philip L U | Oil and gas field chemicals |
| US6465397B1 (en) | 2000-02-11 | 2002-10-15 | Clariant Finance (Bvi) Limited | Synthetic crosslinked copolymer solutions and direct injection to subterranean oil and gas formations |
| EA012088B1 (ru) * | 2004-06-17 | 2009-08-28 | Статойлгидро Аса | Обработка скважин |
| WO2005124100A1 (fr) * | 2004-06-17 | 2005-12-29 | Statoil Asa | Traitement de puits |
| WO2006114623A2 (fr) * | 2005-04-26 | 2006-11-02 | Statoilhydro Asa | Procede de construction et de traitement de puits |
| US20080224087A1 (en) * | 2007-03-14 | 2008-09-18 | Ezell Ryan G | Aqueous-Based Insulating Fluids and Related Methods |
| US20080223596A1 (en) * | 2007-03-14 | 2008-09-18 | Ryan Ezell | Aqueous-Based Insulating Fluids and Related Methods |
| GB2450502B (en) * | 2007-06-26 | 2012-03-07 | Statoil Asa | Microbial enhanced oil recovery |
Family Cites Families (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3242020A (en) * | 1963-09-27 | 1966-03-22 | Dow Chemical Co | Gelled alcohol explosive composition |
| US3373812A (en) * | 1965-07-09 | 1968-03-19 | Gulf Research Development Co | Method of permeably consolidating incompetent sands with a heat-curable resin |
| US4244826A (en) * | 1978-07-17 | 1981-01-13 | Phillips Petroleum Company | Gelled acidic well treating composition and process |
| US4532052A (en) * | 1978-09-28 | 1985-07-30 | Halliburton Company | Polymeric well treating method |
| GB2100269B (en) * | 1981-06-12 | 1985-06-12 | Nat Res Dev | Preparation of particulate gels |
| US4539348A (en) * | 1982-12-16 | 1985-09-03 | Celanese Corporation | Water-swellable crosslinked polymeric microgel particles and aqueous dispersions of organic film-forming resins containing the same |
| US4939203A (en) * | 1983-07-18 | 1990-07-03 | Cities Service Oil And Gas Corporation | Gel for retarding water flow |
| US4600057A (en) * | 1984-10-23 | 1986-07-15 | Halliburton Company | Method of reducing the permeability of a subterranean formation |
-
1990
- 1990-03-15 US US07/493,665 patent/US5043364A/en not_active Expired - Fee Related
-
1991
- 1991-01-31 CA CA002035423A patent/CA2035423C/fr not_active Expired - Fee Related
- 1991-03-06 AU AU72641/91A patent/AU625168B2/en not_active Ceased
- 1991-03-06 NZ NZ237342A patent/NZ237342A/en unknown
- 1991-03-13 EG EG14891A patent/EG19032A/xx active
- 1991-03-14 MX MX024906A patent/MX171813B/es unknown
- 1991-03-14 EP EP91103945A patent/EP0447967B1/fr not_active Expired - Lifetime
- 1991-03-14 ES ES91103945T patent/ES2053225T3/es not_active Expired - Lifetime
- 1991-03-14 NO NO911026A patent/NO301079B1/no unknown
Also Published As
| Publication number | Publication date |
|---|---|
| NO301079B1 (no) | 1997-09-08 |
| CA2035423C (fr) | 1994-10-18 |
| EP0447967A3 (en) | 1991-11-27 |
| NZ237342A (en) | 1992-06-25 |
| ES2053225T3 (es) | 1994-07-16 |
| NO911026L (no) | 1991-09-16 |
| EP0447967A2 (fr) | 1991-09-25 |
| AU7264191A (en) | 1991-09-19 |
| EG19032A (en) | 1994-04-30 |
| CA2035423A1 (fr) | 1991-09-16 |
| NO911026D0 (no) | 1991-03-14 |
| AU625168B2 (en) | 1992-07-02 |
| MX171813B (es) | 1993-11-16 |
| US5043364A (en) | 1991-08-27 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0447967B1 (fr) | Gélification de polymères contenant un acrylamide par l'alcool furfurylique et des aldéhydes hydrodispersibles | |
| US5399269A (en) | Gelation of water soluble polymers | |
| EP0390137B1 (fr) | Changement de perméabilité de formations souterraines à haute température | |
| US4098337A (en) | Method of improving injectivity profiles and/or vertical conformance in heterogeneous formations | |
| EP0115836B1 (fr) | Polymères utilisables dans la recouverte et l'ouvraison de ressources minérales | |
| US4617132A (en) | Method of altering the permeability of a hydrocarbon-containing subterranean formation | |
| US5480933A (en) | Acceleration of gelation of water soluble polymers | |
| US5829527A (en) | Compositions and applications thereof of water-soluble copolymers comprising an ampholytic imidazolium inner salt | |
| US4254249A (en) | Copolymers of N,N-dimethylacrylamide and acrylamide | |
| EP0475325B1 (fr) | Gélatine constituée par des polymères contenant de l'acrylamide et des composés de l'acide aminobenzoique et des aldéhydes dispersibles dans l'eau | |
| NO173510B (no) | Gelerbar vannopploeselig polymerblanding og fremgangsmaate til stimulert oljeutvinning ved bruk av en slik blanding | |
| US4665987A (en) | Prepartially crosslinked gel for retarding fluid flow | |
| US4690219A (en) | Acidizing using n-vinyl lactum/unsaturated amide copolymers | |
| US4326970A (en) | Copolymers of N,N-dimethylacrylamide and acrylamide | |
| EP0446865B1 (fr) | Gelification de polymères contenant un acrylamide par des hydroxyphényle-alcanols | |
| US4481316A (en) | Method for inhibiting the degradation of an aqueous solution of an acrylamide polymer in a petroleum recovery process | |
| CA2006981C (fr) | Extraction amelioree d'hydrocarbures grace a une poly(vinylamine) de masse moleculaire elevee, formee sur place | |
| US4578201A (en) | N-vinyl lactam/unsaturated amide copolymers in thickened acid compositions | |
| US4393163A (en) | Method for stabilizing an acrylamide polymer in a petroleum recovery process | |
| CA1053893A (fr) | Stabilisation des solutions de polymeres | |
| EP0212019B1 (fr) | Méthode pour stabiliser des particules fines | |
| JPS61102995A (ja) | 石油回収用重合体組成物 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Kind code of ref document: A2 Designated state(s): ES FR GB |
|
| PUAL | Search report despatched |
Free format text: ORIGINAL CODE: 0009013 |
|
| AK | Designated contracting states |
Kind code of ref document: A3 Designated state(s): ES FR GB |
|
| 17P | Request for examination filed |
Effective date: 19920331 |
|
| 17Q | First examination report despatched |
Effective date: 19920908 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): ES FR GB |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FG2A Ref document number: 2053225 Country of ref document: ES Kind code of ref document: T3 |
|
| ET | Fr: translation filed | ||
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed | ||
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 19980205 Year of fee payment: 8 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 19980219 Year of fee payment: 8 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: ES Payment date: 19980325 Year of fee payment: 8 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19990314 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: ES Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19990315 |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 19990314 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19991130 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FD2A Effective date: 20010503 |