EP0585546B1 - Matériau d'enregistrement photographique. - Google Patents
Matériau d'enregistrement photographique. Download PDFInfo
- Publication number
- EP0585546B1 EP0585546B1 EP93109236A EP93109236A EP0585546B1 EP 0585546 B1 EP0585546 B1 EP 0585546B1 EP 93109236 A EP93109236 A EP 93109236A EP 93109236 A EP93109236 A EP 93109236A EP 0585546 B1 EP0585546 B1 EP 0585546B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- formula
- alkyl
- residues
- recording material
- compound
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
Classifications
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03C—PHOTOSENSITIVE MATERIALS FOR PHOTOGRAPHIC PURPOSES; PHOTOGRAPHIC PROCESSES, e.g. CINE, X-RAY, COLOUR, STEREO-PHOTOGRAPHIC PROCESSES; AUXILIARY PROCESSES IN PHOTOGRAPHY
- G03C7/00—Multicolour photographic processes or agents therefor; Regeneration of such processing agents; Photosensitive materials for multicolour processes
- G03C7/30—Colour processes using colour-coupling substances; Materials therefor; Preparing or processing such materials
- G03C7/392—Additives
- G03C7/39208—Organic compounds
- G03C7/39232—Organic compounds with an oxygen-containing function
Definitions
- the invention relates to a photographic recording material with at least one silver halide layer, the hydroquinone diether determined in one layer as Contains light stabilizers.
- Color developer - developed which is in accordance oxidation product resulting from the silver image of the developer substances with the color coupler with formation of a dye image reacts.
- a color developer are usually aromatic, containing primary amino groups Compounds, especially those of the p-phenylenediamine type, used.
- a light stabilizer Agents are essentially phenolic compounds, in particular Derivatives of hydroquinone, either added to the couplers or are linked in the form of substituents to the coupler molecules (DE-B-1 547 803, DE-A-26 17 826, DE-A-29 52 511, JP-N-53 070 822, JP-N-54 070 830, JP-N-54 073 032).
- Hydroquinone dialkyl ethers are already available as Light stabilizers have been described, e.g.
- N-alkylated aminophenol ethers are used as light stabilizers have been proposed, for example for the stabilization of pyrazoloazole couplers generated image dyes, cf. EP-A-0 273 412.
- the known light stabilizers However, they do not yet meet the requirements placed on them in every respect.
- the invention has for its object a color photographic recording material to provide associated silver halide emulsion layers Contains cyan, magenta and yellow couplers and by adding a light stabilizer against the fading of the chromogen formed dye under the Influence of light has been stabilized.
- An alkyl radical represented by R 1 to R 6 can be straight-chain or branched, unsubstituted or substituted and can contain up to 18 carbon atoms; Examples are methyl, ethyl, propyl, isopropyl, butyl, tert-butyl, tert-amyl, hexyl, tert-hexyl, octyl, dodecyl; Examples of substituents are halogen (eg chlorine), hydroxy, alkoxy, alkoxycarbonyl, dialkylamino.
- a cycloalkyl radical represented by R 1 , R 3 or R 4 is in particular cyclohexyl or cyclopentyl.
- An aryl radical represented by R 1 is, in particular, phenyl, optionally substituted, for example with alkylsulfonyl, alkyl and / or alkoxy.
- An acyl radical represented by or contained in R 3 , R 4 or R 5 is derived from an aliphatic or aromatic mono- or dicarboxylic acid, from a carbonic acid monoester or from a carbamic acid, examples of such acyl radicals are acetyl, 2-ethylhexanoyl, p-hydroxybenzoyl , Nn-butylcarbamoyl, Nt-butylcarbamoyl, N-dodecylcarbamoyl, hexyloxycarbonyl, dodecyloxycarbonyl.
- An acyl radical represented by R 5 can also be derived from phosphoric acid, phosphonic acid or phosphorous acid.
- the hydroquinone diether compound corresponds to the formula I, in which at least one of the radicals R 3 and R 4 is tertiary alkyl.
- the hydroquinone diether compound corresponds to the formula I in which -XR 5 is -OH, -O-CH 2 -CH 2 -OH or -O-acyl, the acyl radical being derived from aliphatic or aromatic carboxylic acids, from Carbamic acid, carbonic acid monoesters or phosphoric acid, phosphonic acid or phosphorous acid.
- hydroquinone diether compound corresponds to the formula I, where n is 0 or 1.
- hydroquinone diether compounds according to the invention are given below.
- An alkyl radical represented by R 26 or R 51 is an alkyl radical, as can also be represented by R 1 to R 6 .
- An alkyl radical represented by R 31 or R 32 can be straight-chain or branched and contain up to 18 carbon atoms.
- a cycloalkyl radical represented by R 31 , R 32 or R 51 is a cycloalkyl radical, as can also be represented by R 1 , R 3 or R 4 .
- An aryl radical represented by R 51 is an aryl radical, as can also be represented by R 1 , R 3 or R 4 .
- An acyl radical represented by R 26 is an acyl radical, as can also be represented by R 3 , R 4 or R 5 .
- the color photographic recording material according to the invention contains a sequence of several photosensitive Silver halide emulsion layers with each assigned Color couplers and possibly other auxiliary layers such as especially protective layers and between the photosensitive Layers of non-photosensitive binder layers arranged, whereby according to the present invention at least one of the existing photosensitive silver halide emulsion layers a compound of the invention Formula I in combination with a color coupler assigned.
- a compound of the invention Formula I in combination with a color coupler assigned.
- one according to the invention Compound of formula I combined with at least a compound of one of the formulas II, III, IV and V used.
- synergistic effects occur, i.e.
- connection of formula I is a compound of one of the formulas II, III, IV and V are even better protected against light than by a compound of formula I alone.
- the connections of the formula I are with compounds one of formulas II, III, IV and V in a ratio of 1:10 to 10: 1, preferably 1: 4 to 4: 1.
- the compounds of the formulas used according to the invention I, II, III, IV and V act primarily as Light stabilizer, i.e. that from the color coupler Azomethine dyes formed in the chromogenic development show considerable in their presence increased stability against exposure to light on.
- the compounds of the invention take over the function of a whole or in part Oil generator for the color coupler, i.e. you can alone or together with other known oil formers as Coupler solvent can be used. Thereby they make compounds of the invention preferably 50 to 100 wt .-% of the total amount of oil in the respective Layer off. Accordingly, they are preferred in the 0.2 to 2 times the amount by weight, based on the color couplers used together.
- Oil generator for the color coupler i.e. you can alone or together with other known oil formers as Coupler solvent can be used.
- Coupler solvent can be used.
- they make compounds of the invention preferably 50 to 100 wt .-% of the total amount of oil in the respective Layer off. Accordingly,
- the compounds of the formulas used according to the invention I to V are used as a solution in aprotic (hydrophobic) Solvents, e.g. Ethyl acetate, during incorporation into the casting solution for the layer in question with the respective color coupler.
- aprotic (hydrophobic) Solvents e.g. Ethyl acetate
- the training takes place in the usual way, where appropriate other auxiliary solvents and / or high-boiling Coupler solvents, so-called oil formers, are used can be.
- Silver halide can be used as halide chloride, Contain bromide or iodide or mixtures thereof.
- the halide content can be at least one 0 to 15 mol% layer of iodide, 0 to 100 mol% consist of chloride and 0 to 100 mol% of bromide.
- silver bromide iodide emulsions with high chloride content up to pure silver chloride emulsions are used.
- It can are mainly compact crystals, e.g. are regular cubic or octahedral or transitional forms can have. But preferably also platelet-shaped crystals are present, their average Ratio of diameter to thickness is preferably at least 5: 1, the diameter a grain is defined as the diameter of a Circle with a circle content corresponding to the projected Area of the grain.
- the layers can also have tabular silver halide crystals, where the ratio of diameter to thickness is significantly larger than 5: 1, e.g. 12: 1 to 30: 1.
- the silver halide grains can also be multi-layered Have grain structure, in the simplest case with an inner and an outer grain area (core / shell), the halide composition and / or other modifications, such as Endowments of the individual Grain areas are different.
- the middle The grain size of the emulsions is preferably between 0.2 ⁇ m and 2.0 ⁇ m, the grain size distribution can be both be homo- and heterodisperse. Homodisperse grain size distribution means 95% of the grains are not deviate more than ⁇ 30% from the average grain size.
- the emulsions can also other silver salts, e.g. contain organic silver salts, such as silver benzotriazolate or silver behenate.
- Two or more types of silver halide emulsions which are made separately, as a mixture be used.
- the emulsions can be chemically and in the usual way or be spectrally sensitized; you can also by suitable additives must be stabilized.
- Suitable chemical Sensitizers, spectral sensitizing dyes and stabilizers are, for example, in Research Disclosure 17643 (December 1978); referred special attention is paid to chapters III, IV and VI.
- the color photographic recording material according to the invention contains at least one red sensitive, at least one sensitive to green and at least one sensitive to blue Silver halide emulsion layer.
- the photosensitive Layers in a known manner using suitable sensitizing dyes spectrally sensitized; in the blue sensitive silver halide emulsion layer can yourself due to the existing sensitivity No need to add sensitizing dyes.
- Suitable green sensitizers are, for example 9-ethyl carbocyanines with benzoxazole, naphthoxazole or a benzoxazole and a benzthiazole as basic end groups as well as benzimidazocarbocyanines, which also may be further substituted and also at least a sulfoalkyl group on heterocyclic nitrogen must contain.
- Each of the light-sensitive layers mentioned can consist of consist of a single layer or in a known manner, e.g. with the so-called double layer arrangement, too two or more silver halide emulsion partial layers include (DE-C-1 121 470).
- negative films usually red sensitive silver halide emulsion layers arranged closer to the substrate than those sensitive to green Silver halide emulsion layers and these again closer than blue-sensitive, which is generally between green sensitive layers and blue sensitive Layers a non-photosensitive yellow filter layer.
- blue-sensitive which is generally between green sensitive layers and blue sensitive Layers a non-photosensitive yellow filter layer.
- the means for preventing the Incorrect diffusion of developer oxidation products included can.
- Color photographic recording materials according to the invention usually contain in spatial and spectral Assignment to the silver halide emulsion layers different spectral sensitivity color coupler to generate the different part color images Cyan, magenta and yellow, with at least one of the color couplers together with one of the compounds according to the invention of formula I, preferably in combination with at least one compound of one of the formulas II, III, IV and V, the relevant silver halide emulsion layer assigned.
- Color coupler is in such a spatial relationship to the silver halide emulsion layer, that an interaction between them is possible, the a pictorial correspondence between that at the Development formed silver picture and that from the color coupler color image generated.
- This is in the Usually achieved in that the color coupler together with the hydroquinone diether compound of formula I, preferably in combination with at least one connection one of the formulas II, III, IV and V, in the silver halide emulsion layer itself is included or in one adjacent to this, if necessary, non-light-sensitive Binder layer.
- Spectral assignment means that the Spectral sensitivity of each of the photosensitive Silver halide emulsion layers and the color of the the spatially assigned color coupler Partial color image in a certain relationship to each other stand, each of the spectral sensitivities (Red, green, blue) another color of the concerned Partial color image (generally e.g. the colors cyan, Magenta or yellow in this order) is.
- the radicals R 61 , S, T and U represent hydrogen, alkyl, aralkyl, aryl, alkoxy, aroxy, alkylthio, arylthio, amino, anilino, acylamino, cyano, alkoxycarbonyl, carbamoyl , Sulfamoyl, where these radicals can be further substituted.
- Y stands for hydrogen or one for color coupling removable residue such as a halogen atom or a via an oxygen atom, a sulfur atom or a Nitrogen atom preferably attached to the coupling point cyclic group.
- the split group is one cyclic group
- the link to the Coupling point of the coupler molecule either directly above an atom that is part of a ring, e.g. B. a Nitrogen atom, or indirectly via an intermediate Link has been made.
- Such Cleavable groups are known in large numbers, e.g. B. as escape groups of 2 equivalent magnetic couplers.
- cleavable groups attached via oxygen correspond to the formula - O - R 62 in which R 62 represents an acyclic or cyclic organic radical, for example alkyl, aryl, a heterocyclic group or acyl which is derived, for example, from an organic carbon or sulfonic acid.
- R 62 denotes an optionally substituted phenyl group.
- heterocyclic Rings that have a ring nitrogen atom with the Coupling point of the purple coupler are connected.
- the heterocyclic rings often contain adjacent to which mediates the bond to the coupler molecule Nitrogen atom activating groups, e.g. B. carbonyl or Sulfonyl groups or double bonds.
- the cleavable group is attached to the Coupling point of the coupler is bound, it can for the rest of a diffusible carbocyclic or heterocyclic mercapto compound, which inhibit the development of silver halide can
- inhibitor residues are numerous than at the coupling point of couplers, also purple couplers bonded cleavable group described e.g. in US-A-3,227,554.
- pyrazoloazole couplers of the formulas VIa to VIg those of the formulas VId and VIe are preferably used according to the invention together with a hydroquinone diether compound of the formula I.
- at least one of the radicals R 61 and S or at least one of the radicals R 61 and T preferably represents a secondary alkyl or tertiary alkyl radical, ie a radical of the formula wherein R 63 and R 64 are alkyl and R 65 is H or a substituent.
- Possible substituents are alkyl, aryl, cycloalkyl, hydroxy, halogen, -COOH, -SO 3 H, -SO 2 H, alkoxy, aryloxy, alkylthio, arylthio, nitro, sulfonyl, sulfamoyl, sulfonylamino, acylamino, carbamoyl, acyloxy, alkoxycarbonyl, Aryloxycarbamoyl, ureido, carbamoyloxy, alkoxycarbonylamino, aryloxycarbonylamino, alkoxycarbonyloxy and aryloxycarbonyloxy.
- Preferred substituents are alkyl, sulfonyl, sulfonylamino, Sulfamoyl, ureido, acylamino, carbamoyl, alkoxy, Aryloxy and alkoxycarbonylamino.
- the color couplers can be 4-equivalent couplers, but also act as 2-equivalent couplers.
- the latter are derived from the 4 equivalent couplers in that they have a in the coupling point Contain substituents in the coupling is split off.
- To the 2 equivalent couplers are count those that are colorless, as well as those which have an intense intrinsic color, which at Color coupling disappears or by the color of the generated Image dye is replaced (mask coupler), but also the white couplers, which react with color developer oxidation products essentially colorless Products result.
- the reaction with color developer oxidation products in freedom is set, either directly or after the primary split off one or more other groups have been split off (e.g. DE-A-27 03 145, DE-A-28 55 697, DE-A-31 05 026, DE-A-33 19 428), a certain desired photographic effectiveness unfolded, e.g. as a development inhibitor or accelerator.
- 2-equivalent couplers are the known DIR couplers as well as DAR or. FAR coupler.
- couplers used especially those according to the invention preferably used magenta couplers of the type Pyrazoloazoles, for example of the formulas (VId) and (VIe) can also be in polymeric form, e.g. as polymer latex apply.
- High molecular weight color couplers are described, for example in DE-C-1 297 417, DE-A-24 07 569, DE-A-31 48 125, DE-A-32 17 200, DE-A-33 20 079, DE-A-33 24 932, DE-A-33 31 743, DE-A-33 40 376, EP-A-27 284, US-A-4 080 211.
- the high molecular color couplers are usually by polymerizing ethylenically unsaturated monomers Color couplers manufactured.
- the color couplers used can also be those that Dyes with poor or limited mobility deliver.
- a mobility that is measured in this way is that the contours of chromogenic development formed discrete dye stains run and are smeared into each other.
- This degree of mobility is to be distinguished on the one hand from the usual Case of complete immobility in photographic Layers used in conventional photographic materials for the color couplers or from them Dyes produced is aimed at a possible to achieve high sharpness, and on the other hand by the case of complete mobility of the dyes, the for example in color diffusion processes becomes.
- the latter dyes usually have about at least one group they are in alkaline Make medium soluble.
- the extent of the invention aimed weak mobility can be controlled are made by varying substituents, for example the solubility in the organic medium of the oil former or the affinity for the binder matrix in a more targeted manner Way to influence.
- the color photographic Recording material of the present Invention contain other additives, such as Antioxidants, dye stabilizers and Means for influencing the mechanical and electrostatic Properties as well as UV absorbers.
- additional connections are combined with the compounds of the invention, i.e. in the same Binding agent layer or in adjacent to each other Binder layers used.
- additives to improve the dye, Coupler and whiteness stability and to reduce the Color veils can include the following chemical classes include: hydroquinones, 5-, 6-, 7- and 8-hydroxychromanes, 5-hydroxycoumarans, spirochromanes, Spiroindanes, p-alkoxyphenols, sterically hindered Phenols, gallic acid derivatives, methylenedioxybenzenes, Aminophenols, sterically hindered amines, derivatives with esterified or etherified phenolic hydroxyl groups, Derivatives with acylated amino groups, metal complexes.
- Ultraviolet absorbing couplers such as Cyan couplers of the ⁇ -naphthol type
- ultraviolet absorbing Polymers are used. These ultraviolet absorbents can be done by pickling in a special Layer fixed.
- the invention is used to produce color photographic images color photographic recording material, assigned to at least one silver halide emulsion layer a color coupler and a connection of the Formula I contains, with a color developer compound developed.
- Can be used as a color developer compound use all developer connections that the Have ability in the form of their oxidation product Color couplers react to azomethine dyes.
- Suitable Color developer compounds are aromatic at least compounds containing a primary amino group of the p-phenylenediamine type, for example N, N-dialkyl-p-phenylenediamine, such as N, N-diethyl-p-phenylenediamine, 1- (N-ethyl-N-methylsulfonamidoethyl) -3-methyl-p-phenylenediamine, 1- (N-ethyl-N-hydroxyethyl-3-methyl-p-phenylenediamine, 1- (N-ethyl-N-3-hydroxypropyl) -3-methyl-p-phenylenediamine and 1- (N-ethyl-N-methoxyethyl) -3-methyl-p-phenylenediamine.
- N, N-dialkyl-p-phenylenediamine such as N, N-diethyl-p-phenylenediamine, 1- (N-ethyl-N-methylsulfon
- the material is usually bleached and fixed. Bleaching and fixing can be carried out separately or together.
- the usual compounds can be used as bleaching agents, for example Fe 3+ salts and Fe 3+ complex salts such as ferricyanides, dichromates, water-soluble cobalt complexes, etc.
- Particularly preferred are iron III complexes of aminopolycarboxylic acids, in particular, for example, ethylenediaminetetraacetic acid, N-hydroxyethylethylenediamine triacetic acid, alkyliminodicarboxylic acids and alkyliminodicarboxylic acids of corresponding phosphonic acids.
- Persulphates are also suitable as bleaching agents.
- a color photographic recording material suitable for a rapid processing process was produced by applying the following layers in the order given to a support made of paper coated on both sides with polyethylene.
- the quantities given relate to 1 m 2 .
- the corresponding amounts of AgNO 3 are given for the silver halide application.
- UV absorber UV-1 The following compound (UV absorber UV-1) was used in layers 5 and 7.
- Samples 2 to 9 were made in the same manner as sample 1 produced with the difference that the layer 4 additionally 0.31 g of light stabilizer of the structural formulas VP-1 to VP-3 or light stabilizers according to the invention the structural formula I was added.
- Both Samples 10 to 13, 14 to 17 and 18 to 20 were the Coupler VI-1 shown in the table below Quantity replaced by other couplers of formula VI.
- coupler VI-23 there was an additional 4th layer 0.34 g DBP and 0.04 g CPM through 0.38 g 2,4-di-tert-pentylphenol replaced (samples 18 to 20).
- the example shows that the compounds of formula I the light stability of the image dyes is significantly improved.
- connections VP-1 to VP-3 according to GB-A 2 135 788 cause a significantly less improvement in light stability.
- Sample 14 serves as a comparison as described under Example 1.
- Samples 21 to 23 according to the invention differ from sample 14 in that in the green-sensitive layer DBP and TKP were replaced by 0.38 g of light stabilizer of the formula I. Processing and testing were carried out as described in Example 1 (Table 2).
- Example 2 shows that the light stabilizers according to the invention of formula I as an oil generator in combination with Pyrazoloazole couplers can be used. Sensitivity, Gradation and maximum density are comparable with DBP / TKP, the light stability is significantly increased.
- Example 3 the following compounds are used in addition to the compounds already mentioned:
- Samples 25 and 26 according to the invention were in the manufactured in the same way as the sample 24 with the Difference that the oil generator TKP in layers 6 and 7 replaced by compounds I-14 and I-17 has been.
- coupler VI-7 was replaced by the Coupler VI-14 replaced, TKP (comparison) served as oil generator.
- samples 28 to 30 according to the invention differs from sample 27 in that CPM in the layers 6 and 7 replaced by compounds I-4, I-14 and I-17 has been.
- a suitable one for a rapid processing process color photographic material was prepared as in example 1, sample 1, with the difference that in layer 4 instead of magenta coupler VI-1 the Magenta coupler VI-6 in an amount of 0.41 g and instead of the oil-forming mixture only 0.38 g CPM were used (sample 31).
- Sample 32-52 was produced in the same way as sample 31, with the difference that the light stabilizers VP-1, VP-3, VP-4 or light stabilizers according to the invention, which are given in table 1, were additionally added to layer 4. Processing, irradiation and evaluation of the samples were carried out as in Example 1, with the difference that 6.10 6 was irradiated with 10.10. The results are shown in Table 4.
- Example 4 shows that by using light stabilizers Formula I improves light stability can be (samples 33, 34, 35, 38) and that by Compounds of formula I achieved light stability can be further improved if the connections of the formula I in combination with compounds of the formulas II, III, IV and V can be used (samples 39-52).
- Sample 31 from Example 4 serves as a comparison.
- Samples 53-59 are made in the same way like sample 31 from example 4, with the difference that the oil former in layer 4 by the invention Compounds of formula I is replaced and other compounds one of the formulas II, III, IV and V or the Comparative compound VP-3 can be added.
- Example 5 shows that the light stabilizers according to the invention Formula I also the function of a Oil can take over without being disadvantageous Effects on sensitivity, gradation and Maximum density can be observed.
- the light stability is due to the combination according to the invention with compounds of the formula II, III, IV or V again clearly elevated.
- link VP-3 is here significantly less effective.
- Samples 61-72 were made in the same manner like sample 60 with the difference that the layer 3 the added light stabilizers listed in Table 6 were. Samples 65-72 also became the magenta coupler VI-1 by those given in Table 6 (VI-5, VI-23) replaced.
- the samples obtained were then exposed behind a graduated gray wedge.
- the materials were then processed in the following manner using the processing baths listed below: development 210 s, 33 ° C bleaching 50 s, 20 ° C Fix 60 s, 20 ° C Water 120 s, 20 ° C Dry.
- Example 6 shows the improvement in light stability by using compounds of formula I. (Samples 61, 66, 70) and the increased effectiveness again when combined with connections one of the Formulas II, III, IV and V as light stabilizers. As Comparison of compounds used VP-1, VP-3 and VP-4 are significantly less effective.
Landscapes
- Physics & Mathematics (AREA)
- Spectroscopy & Molecular Physics (AREA)
- General Physics & Mathematics (AREA)
- Silver Salt Photography Or Processing Solution Therefor (AREA)
Claims (12)
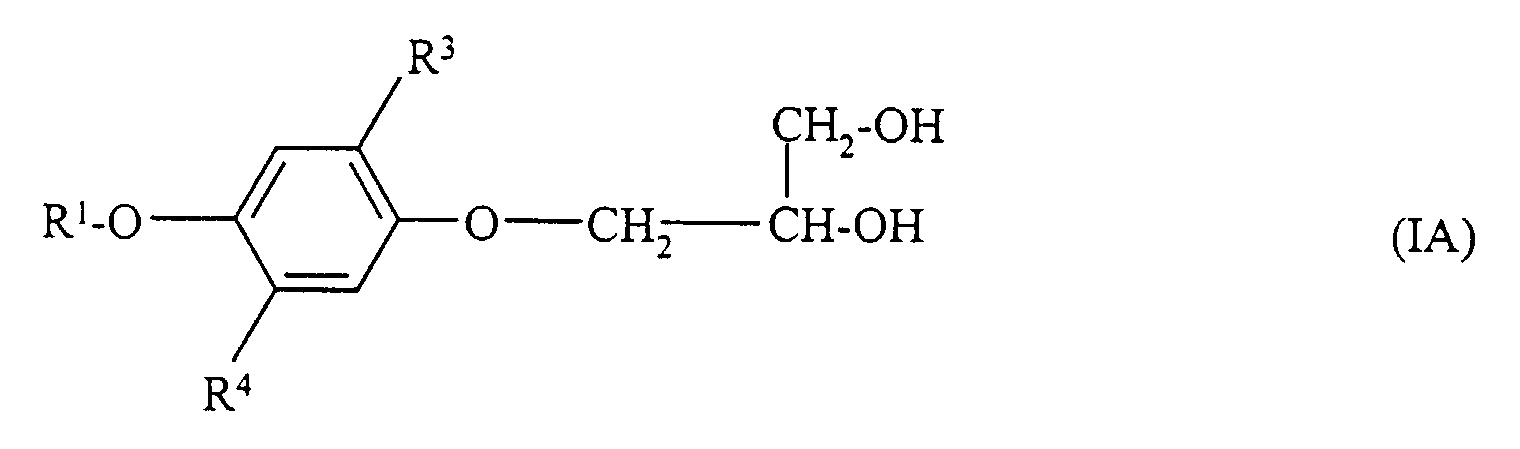
- Matériau d'enregistrement photographique couleur comportant au moins une couche d'émulsion d'halogénure d'argent sensible au rouge à laquelle est associé un coupleur cyan, au moins une couche d'émulsion d'halogénure d'argent sensible au vert à laquelle est associé un coupleur magenta et au moins une couche d'émulsion d'halogénure d'argent sensible au bleu à laquelle est associé un coupleur jaune, qui contient dans au moins l'une de ses couches un composé hydroquinonediéther, caractérisé en ce que le composé hydroquinonediéther est un composé de formule I à l'exception des composés de formule IA et en ce qu'un composé de formule I à l'exception des composés de formule IA est associé en combinaison avec un coupleur à au moins l'une des couches d'émulsion d'halogénure d'argent présentes : où
- R1
- représente alkyle, cycloalkyle, aryle,
- R2
- représente H, alkyle,
- R3, R4
- représentent H, alkyle, cycloalkyle, halogène, alcoxy, aroxy, acyloxy, alkylthio, arylthio, acyle, sulfonyle, sulfamoyle, acylamino, sulfonylamino, nitro,
- R5
- représente H, acyle, alkyle,
- X
- représente -O- ou -NR6-,
- R6
- représente H ou alkyle,
- n
- représente 0 ou un nombre entier de 1 à 3.
- Matériau d'enregistrement selon la revendication 1 caractérisé en ce que, dans la formule I, au moins l'un des restes R3 et R4 représente alkyle tertiaire.
- Matériau d'enregistrement selon l'une des revendications 1 et 2 caractérisé en ce que, dans la formule I, le reste -X-R5 représente -OH, -O-CH2- CH2-OH ou -O-acyle, le reste acyle dérivant d'un acide carboxylique aliphatique ou aromatique, d'un acide carbamique, d'un monoester d'acide carbonique ou de l'acide phosphoreux.
- Matériau d'enregistrement selon l'une des revendications 1 à 3 caractérisé en ce que, dans la formule I, la lettre n représente 0 ou 1.
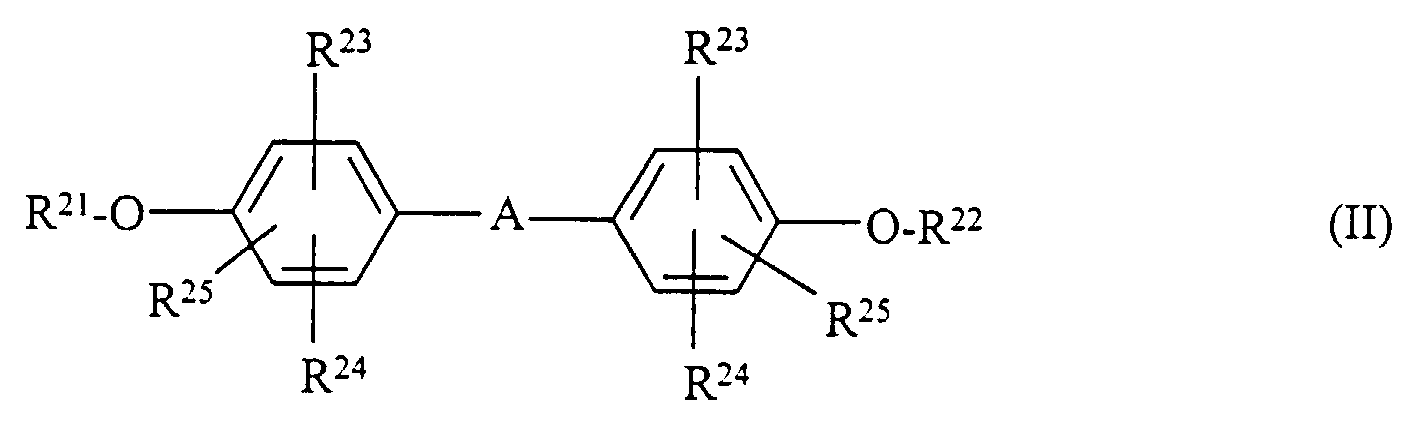
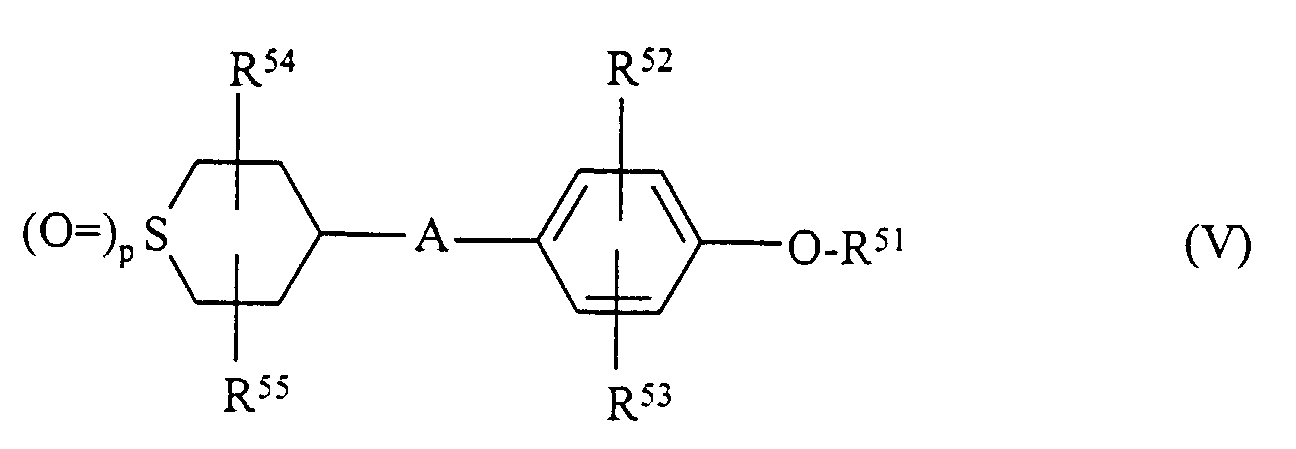
- Matériau d'enregistrement selon l'une des revendications 1 à 4 caractérisé en ce qu'il contient en combinaison avec un composé hydroquinonediéther de formule I au moins un composé de l'une des formules II, III, IV et V où
- R21, R22
- représentent des restes comme R5,
- R23, R24, R25
- représentent des restes comme R3,
- A
- représente une simple liaison, -S(=O)m-, alkylène ou -NR26-,
- R26
- représente alkyle ou acyle,
- m
- représente 0, 1 ou 2 ;
- R31, R32
- représentent alkyle, cycloalkyle,
- R33, R34
- représentent des restes comme R3 ;
- R41, R42
- représentent des restes comme R5,
- R43, R44 R45, R46
- représentent des restes comme R3, à condition qu'au moins l'un des restes R44 et R45 ne représente pas alkyle ;
- R51
- représente alkyle, cycloalkyle, aryle,
- R52, R53, R54, R55
- représentent des restes comme R3,
- p
- représente 0, 1 ou 2.
- Matériau d'enregistrement selon la revendication 5 caractérisé en ce que, dans la formule II, A représente alkylène et R21 et R22 représentent H.
- Matériau d'enregistrement selon la revendication 5 caractérisé en ce que, dans la formule III, au moins l'un des restes R33 et R34 représente alkyle.
- Matériau d'enregistrement selon la revendication 5 caractérisé en ce que, dans la formule IV, au moins l'un des restes R43 et R46 et/ou au moins l'un des restes R44 et R45 représentent H.
- Matériau d'enregistrement selon la revendication 5 caractérisé en ce que, dans la formule V, R54 et R55 représentent H.
- Matériau d'enregistrement selon l'une des revendications 1 à 9 caractérisé en ce qu'à au moins une couche d'émulsion d'halogénure d'argent sensible au vert est associé un composé de formule I en combinaison avec un coupleur magenta de formule VI où, dans la formule VI :
- R61
- représente H, alkyle, aralkyle ou aryle,
- Y
- représente H ou un groupe libérable par couplage,
- Za, Zb, Zc
- représentent un groupe méthine éventuellement substitué, =N- ou -NH-, où la liaison Za-Zb ou la liaison Zb-Zc représente une double liaison et l'autre liaison représente une simple liaison.
- Matériau d'enregistrement selon la revendication 10 caractérisé en ce que le coupleur magenta correspond à l'une des formules VId et VIe où R61 représente H, alkyle, aralkyle ou aryle, et S et T représentent l'hydrogène, alkyle, aralkyle, aryle, alcoxy, aroxy, alkylthio, arylthio, amino, anilino, acylamino, cyano, alcoxycarbonyle, carbamoyle ou sulfamoyle, où au moins l'un des restes R61 et S (dans la formule VId) ou au moins l'un des restes R61 et T (dans la formule VIe) représente un reste alkyle secondaire ou alkyle tertiaire, et où Y représente l'hydrogène ou un reste clivable lors du couplage couleur.
- Matériau d'enregistrement selon la revendication 10 caractérisé en ce que le composé de formule I ou la combinaison d'un composé de formule I avec au moins un composé de l'une des formules II, III, IV et V est contenu en la quantité massique de 0,2 à 2 fois par rapport au coupleur magenta de formule VI, dans la couche d'émulsion d'halogénure d'argent sensible au vert.
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE4323512A DE4323512A1 (de) | 1992-09-01 | 1993-07-14 | Fotografisches Aufzeichnungsmaterial |
| US08/109,436 US5362615A (en) | 1992-09-01 | 1993-08-20 | Photographic recording material containing a hydroquinone diether compound |
| JP5237370A JPH06194796A (ja) | 1992-09-01 | 1993-08-30 | 写真記録材料 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE4229132 | 1992-09-01 | ||
| DE4229132 | 1992-09-01 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| EP0585546A2 EP0585546A2 (fr) | 1994-03-09 |
| EP0585546A3 EP0585546A3 (fr) | 1995-08-09 |
| EP0585546B1 true EP0585546B1 (fr) | 1998-09-23 |
Family
ID=6466952
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP93109236A Expired - Lifetime EP0585546B1 (fr) | 1992-09-01 | 1993-06-08 | Matériau d'enregistrement photographique. |
Country Status (2)
| Country | Link |
|---|---|
| EP (1) | EP0585546B1 (fr) |
| DE (1) | DE59309004D1 (fr) |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5491054A (en) * | 1994-12-22 | 1996-02-13 | Eastman Kodak Company | Photographic elements containing 2-equivalent pyrazolone magenta dye forming couplers and stabilizing compounds |
| US5484696A (en) * | 1994-12-22 | 1996-01-16 | Eastman Kodak Company | Photographic elements containing 2-equivalent pyrazolone magenta dye forming couplers and fade reducing compounds |
| US5565312A (en) * | 1995-04-26 | 1996-10-15 | Eastman Kodak Company | Photographic elements containing magenta dye forming couplers and fade reducing compounds-L |
| US6312881B1 (en) * | 2000-01-14 | 2001-11-06 | Eastman Kodak Company | Photographic element with yellow dye-forming coupler and stabilizing compounds |
Family Cites Families (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS59125732A (ja) * | 1983-01-07 | 1984-07-20 | Fuji Photo Film Co Ltd | ハロゲン化銀カラ−写真感光材料 |
| JPS60262159A (ja) * | 1984-06-08 | 1985-12-25 | Fuji Photo Film Co Ltd | ハロゲン化銀カラ−写真感光材料 |
| DE3784150T2 (de) * | 1986-12-25 | 1993-06-03 | Konishiroku Photo Ind | Lichtempfindliches photographisches silberhalogenidmaterial. |
| US5302863A (en) * | 1993-01-29 | 1994-04-12 | Hewlett-Packard Company | CMOS peak amplitude detector |
-
1993
- 1993-06-08 EP EP93109236A patent/EP0585546B1/fr not_active Expired - Lifetime
- 1993-06-08 DE DE59309004T patent/DE59309004D1/de not_active Expired - Fee Related
Also Published As
| Publication number | Publication date |
|---|---|
| DE59309004D1 (de) | 1998-10-29 |
| EP0585546A3 (fr) | 1995-08-09 |
| EP0585546A2 (fr) | 1994-03-09 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| DE4323512A1 (de) | Fotografisches Aufzeichnungsmaterial | |
| DE69223582T2 (de) | Purpurkuppler enthaltend photographisches material und verfahren | |
| DE3209996C2 (de) | Farbphotographisches Silberhalogenid-Aufzeichnungsmaterial | |
| DE3426656A1 (de) | Lichtempfindliches farbphotographisches silberhalogenid-aufzeichnungsmaterial | |
| EP0401612B1 (fr) | Produit photographique pour la reproduction couleur avec un coupleur DIR | |
| DE3021207A1 (de) | Lichtempfindliches (farb)photographisches aufzeichnungsmaterial | |
| DE3784150T2 (de) | Lichtempfindliches photographisches silberhalogenidmaterial. | |
| DE69212548T2 (de) | Farbphotographische Materialien enthaltend einen Magenta-Kuppler, einen DIR-Kuppler und eine Carbonamid-Verbindung | |
| DE69026737T2 (de) | Farbphotographische Silberhalogenidmaterialien | |
| EP0415056B1 (fr) | Matériau d'enregistrement pour photographie en couleurs à un coupleur libérant un composé photographiquement actif | |
| DE3633364A1 (de) | Farbfotografisches aufzeichnungsmaterial mit einem farbkuppler vom pyrazoloazol-typ | |
| DE4031644A1 (de) | Lichtempfindliches photographisches silberhalogenidmaterial | |
| EP0257451B1 (fr) | Matériau d'enregistrement photographique couleur | |
| EP0585546B1 (fr) | Matériau d'enregistrement photographique. | |
| EP0401613B1 (fr) | Produit photographique de reproduction couleur avec un coupleur DIR | |
| EP0421221B1 (fr) | Produit pour la reproduction photographique couleur contenant un coupleur DIR | |
| DE3779717T2 (de) | Photographisches silberhalogenidmaterial und verfahren zur bildung eines farbbildes darauf. | |
| EP0254151B1 (fr) | Matériau photographique couleur contenant des coupleurs | |
| DE4307194A1 (de) | Farbfotografisches Aufzeichnungsmaterial mit einem Farbkuppler vom Pyrazoloazol-Typ | |
| DE3323448C2 (fr) | ||
| EP0572894B1 (fr) | Produit photographique d'enregistrement en couleur avec un coupleur DIR cyan | |
| DE3536244A1 (de) | Farbphotographisches silberhalogenidmaterial | |
| DE3626219A1 (de) | Farbfotografisches aufzeichnungsmaterial mit einem gelb-dir-kuppler | |
| DE3636824A1 (de) | Farbfotografisches aufzeichnungsmaterial mit einem gelb-dir-kuppler | |
| EP0563638B1 (fr) | Matériau photographique d'enregistrement |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Kind code of ref document: A2 Designated state(s): DE FR GB IT NL |
|
| PUAL | Search report despatched |
Free format text: ORIGINAL CODE: 0009013 |
|
| AK | Designated contracting states |
Kind code of ref document: A3 Designated state(s): DE FR GB IT NL |
|
| 17P | Request for examination filed |
Effective date: 19950831 |
|
| 17Q | First examination report despatched |
Effective date: 19960524 |
|
| GRAG | Despatch of communication of intention to grant |
Free format text: ORIGINAL CODE: EPIDOS AGRA |
|
| GRAG | Despatch of communication of intention to grant |
Free format text: ORIGINAL CODE: EPIDOS AGRA |
|
| GRAH | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOS IGRA |
|
| GRAH | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOS IGRA |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): DE FR GB IT NL |
|
| REF | Corresponds to: |
Ref document number: 59309004 Country of ref document: DE Date of ref document: 19981029 |
|
| GBT | Gb: translation of ep patent filed (gb section 77(6)(a)/1977) |
Effective date: 19981211 |
|
| ET | Fr: translation filed | ||
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: 746 Effective date: 19990305 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: D6 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed | ||
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20000505 Year of fee payment: 8 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20000509 Year of fee payment: 8 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: NL Payment date: 20000516 Year of fee payment: 8 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 20000919 Year of fee payment: 8 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20010608 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20020101 |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 20010608 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20020228 |
|
| NLV4 | Nl: lapsed or anulled due to non-payment of the annual fee |
Effective date: 20020101 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20020403 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES;WARNING: LAPSES OF ITALIAN PATENTS WITH EFFECTIVE DATE BEFORE 2007 MAY HAVE OCCURRED AT ANY TIME BEFORE 2007. THE CORRECT EFFECTIVE DATE MAY BE DIFFERENT FROM THE ONE RECORDED. Effective date: 20050608 |