EP1529093B1 - Odorisation du gaz avec des phenols et/ou des ethers de phenol - Google Patents
Odorisation du gaz avec des phenols et/ou des ethers de phenol Download PDFInfo
- Publication number
- EP1529093B1 EP1529093B1 EP03784163A EP03784163A EP1529093B1 EP 1529093 B1 EP1529093 B1 EP 1529093B1 EP 03784163 A EP03784163 A EP 03784163A EP 03784163 A EP03784163 A EP 03784163A EP 1529093 B1 EP1529093 B1 EP 1529093B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- ester
- carbon atoms
- alkyl
- acrylic acid
- methyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
- 0 *C1[N+3](*)c(cccc2)c2OC1* Chemical compound *C1[N+3](*)c(cccc2)c2OC1* 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10L—FUELS NOT OTHERWISE PROVIDED FOR; NATURAL GAS; SYNTHETIC NATURAL GAS OBTAINED BY PROCESSES NOT COVERED BY SUBCLASSES C10G OR C10K; LIQUIFIED PETROLEUM GAS; USE OF ADDITIVES TO FUELS OR FIRES; FIRE-LIGHTERS
- C10L3/00—Gaseous fuels; Natural gas; Synthetic natural gas obtained by processes not covered by subclass C10G, C10K; Liquefied petroleum gas
- C10L3/003—Additives for gaseous fuels
- C10L3/006—Additives for gaseous fuels detectable by the senses
Definitions
- the present invention relates to the use of mixtures containing alkyl acrylates and / or alkyl methacrylates and phenols and / or phenol ethers for the odorization of natural gas, a process for the odorization of natural gas and natural gas containing these mixtures.
- the city and coke oven gas previously used for public gas supply contained intensely smelling components and therefore had a strong odor, so that escaping gas could be easily perceived.
- Gas odorization means the addition of odor-intensive substances acting as warning or alarm substances (odorizing agents) to otherwise odorless gases.
- Natural gas consists mainly of methane (typical methane contents are in the range 50 to 99 wt.%, Usually in the range 60 to 90 wt.%) And, depending on the origin, besides different proportions of ethane, propane and higher molecular weight hydrocarbons included.
- gas is therefore odorized by the addition of odorous substances.
- DVGW German Association of Gas and Water.
- TFT tetrahydrothiophene
- mercaptans or thioethers is also common.
- shock odorization up to three times the amount of odorant is added to the gas as compared to conventional odorization.
- the shock odorization is used, for example, when commissioning new networks or line sections for faster achievement of minimum odorant concentration or to detect small leaks in the gas installation.
- THT and mercaptans are ideal for reliable odorization of gas.
- combustion of such odorized gases produces sulfur oxides as combustion products - hundreds of tons per year across the country.
- JP-B-51-007481 mentions that acrylic acid alkyl esters such as methyl acrylate, ethyl acrylate and butyl acrylate are known to have weak odorant properties for fuel gases and are of practically no importance in this respect.
- the document describes and claims allyl acrylate as an effective odorizing component.
- JP-A-55-104393 discloses that odorants containing an alkyne and at least 2 compounds selected from a group consisting of methyl acrylate, ethyl acrylate, methymethacrylate, allyl methacrylate, ethyl propionate, methyl n-butyrate, Methyl iso-butyrate and prenyl acrylate is, and optionally tert-butylmercaptan, are suitable for the odorization of fuel gases.
- JP-B-51-034841 "odor thresholds" of various substances were determined, with n-valeric acid, n-butyric acid, isobutyraldehyde and various methylamines having low odor odor thresholds. Nevertheless, it was found that ethyl acrylate or n-valeric acid used alone, because of their odor properties, not sufficiently odorizing act.
- the optimized mixture comprised ethyl acrylate, n-valeric acid and triethylamine, this mixture containing equal parts by weight of n-valeric acid and triethylamine and 30 to 80% by weight of ethyl acrylate.
- Another object of the present invention is a method for the odorization of fuel gases with a methane content of at least 60 wt .-% with the mixtures according to the invention.
- Another object of the present invention are fuel gases with a methane content of at least 60 wt .-% containing the mixtures of the invention.
- mixtures according to the invention are excellent alternatives to known sulfur-free odorizing agents.
- the alkyl or alkenyl groups of up to 5 carbon atoms may be methyl, ethyl, ethenyl, n-propyl, 1-propen-1-yl, 2-propen-1-yl, 2-propyl , 1-propen-2-yl, n-butyl, 1-butenyl, 2-butenyl, 3-butenyl, sec-butyl, 1-ethylethenyl, 1-methyl-1-propenyl, 1-methyl-2-propenyl, 1,3-butadien-1-yl, 1,3-butadien-2-yl, 1-methylene-3-butenyl, 2 Methylpropyl, 2-methyl-1-propenyl, 2-methyl-2-propenyl, 1,1-dimethylethyl, n-pentyl, 1-pentenyl, 2-pentenyl, 3-pentenyl, 4 Pentenyl, 1,3-pentadienyl, 2,4-pentadien
- the alkoxy groups having 1 to 4 carbon atoms may be methoxy, ethoxy, n-propoxy, iso-propoxy, n-butoxy, sec-butoxy, iso-butoxy or tert-butoxy groups.
- Advantageous compounds of the formula (I) which contain at most 2 alkyl groups, at most 2 alkenyl groups, at most 2 acyl groups or at most 2 alkoxy groups.
- inventively preferred compounds of formula (I) are liquid at 25 ° C and 1013 mbar. These liquids can also be highly viscous or oily.
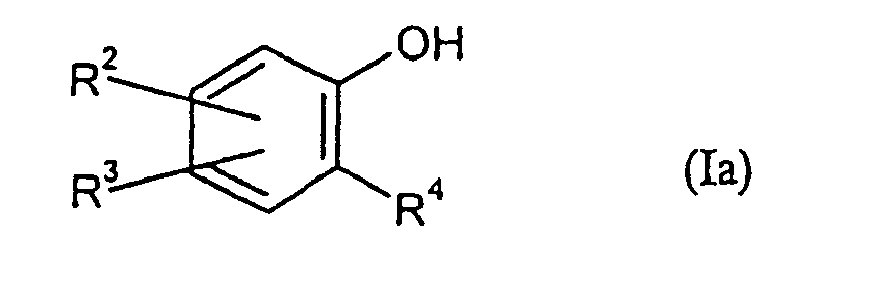
- ortho-phenols and 2,3-, 2,4-, 2,5- or 2,6-substituted phenols.
- ortho-anisoles in particular ortho-anisoles, as well as 2,3-, 2,4-, 2,5- or 2,6-disubstituted anisoles are preferred.
- the acrylic acid C 1 -C 6 -alkyl esters are advantageously selected from the group comprising methyl acrylate, ethyl acrylate, n-propyl acrylate, isopropyl acrylate, n-butyl acrylate, iso-butyl acrylate, acrylic acid tert. butyl ester, n-pentyl acrylate, acrylic acid iso-pentyl ester and n-hexyl acrylate.
- acrylic acid C 1 -C 4 -alkyl esters in particular methyl acrylate, ethyl acrylate, n-propyl acrylate, isopropyl acrylate, n-butyl acrylate and iso-butyl acrylate.
- Very particularly preferred acrylic acid C 1 -C 4 -alkyl esters are methyl acrylate, ethyl acrylate and n-butyl acrylate.
- the methacrylic acid-C 1 -C 6 -alkyl esters are advantageously selected from the group comprising methyl methacrylate, ethyl methacrylate, n-propyl methacrylate, isopropyl methacrylate, n-butyl methacrylate, iso-butyl methacrylate, methacrylic acid tert. butyl ester, n-pentyl methacrylate, iso-pentyl methacrylate and n-hexyl methacrylate.
- C 1 -C 4 -alkyl methacrylates in particular methyl methacrylate, ethyl methacrylate, n-propyl methacrylate, isopropyl methacrylate, Methacrylic acid n-butyl ester and methacrylic acid iso-butyl ester.
- Very particularly preferred methacrylic acid-C 1 -C 4 alkyl esters are methyl methacrylate and ethyl methacrylate.
- Preferred odorants contain at least 2 or at least 3 acrylic acid alkyl esters, at least 2 or at least 3 alkyl methacrylates or at least one acrylic acid alkyl ester and at least one methacrylic acid alkyl ester.
- Particularly preferred odorizing agents contain at least 2 or at least 3 acrylic acid alkyl esters.
- the acrylate mixtures contain the low molecular weight alkyl acrylate and the higher molecular weight alkyl acrylate preferably in a weight ratio of 9: 1 to 1: 9, preferably from 7: 3 to 3: 7.
- the odorant contains side by side at least two acrylic acid C 1 -C 4 alkyl esters, most preferably methyl acrylate and ethyl ester.
- the compounds of the formula (I) can be used in the mixtures according to the invention in amounts of 1 to 100, preferably 10 to 100, in particular 20 to 50 parts by weight per 1000 parts by weight of acrylic acid C 1 -C 6 -alkyl ester and / or methacrylic acid-C 1 -C 6- alkyl esters are used.
- the odorant according to the invention can be added, for example, to increase the stability of common antioxidants.
- examples include vitamin C and derivatives (eg ascorbyl palmitate, ascorbyl acetate), tocopherols and derivatives (eg vitamin E, vitamin E acetate), vitamin A and derivatives (vitamin A - palmitate) phenolic benzylamines, formic acid, acetic acid, benzoic acid, sorbic acid, Hexamethylenetetramine, tert-butylhydroxytoluene, tert-butylhydroxyanisole, ⁇ -hydroxy acids (eg citric acid, lactic acid, malic acid), hydroquinone monomethyl ether.
- Preferred antioxidants are tert-butylhydroxytoluene (BHT, Jonol), tert-butylhydroxyanisole, hydroquinone monomethyl ether and ⁇ -tocopherol.
- the odorants can be added to an odorant also several antioxidants.
- the odorants contain one, two or three antioxidants, preferably one or two antioxidants.
- the antioxidants are preferably used in amounts of 0.01 to 5, in particular 0.05 to 2, especially 0.1 to 1 parts by weight per 1000 parts by weight of acrylic acid alkyl ester and / or methacrylic acid ester.
- the total amount of antioxidants in the odorant is usually in the range 0.001-1% by weight, preferably in the range 0.01-0.5% by weight, particularly preferably in the range 0.05-0.25% by weight.
- the amount of odorant with respect to the gas to be odorized is typically in the range 5 - 100 mg / m 3, preferably 5: - 50 mg / m 3, particularly preferably 10-40 mg / m 3 and most preferably 12-30 mg / m 3 .
- Me-Ac methyl acrylate; Et-Ac: ethyl acrylate; Bu-Ac: n-butyl acrylate; Me-Me: methyl methacrylate; Et-Me: ethyl methacrylate; Bu-Me: n-butyl methacrylate; BHT: tert-butylhydroxytoluene; BHA: tert-butylhydroxyanisole; Hydr: hydroquinone monomethyl ether
- the odorants according to the invention were evaluated in concentrations of 10, 25 and 50 mg / m 3 natural gas (methane content: 85 wt .-%) with regard to their odor odor and their warning intensity against unodorated natural gas (blank). These concentrations correspond to the typical concentrations of odorant in natural gas under normal conditions or in shock odorization.
- the experiment was carried out at room temperature (about 20 ° C) such that in a gas stream in a tube, the odorant is metered. At the end of this 2 m long tube (within the tube is the homogenization), the exiting odorized gas is evaluated by a group of trained examiners (8 to 12 people) odor. The rating was on a scale from 1 (very weak / very little warning) to 10 (very strong / very warning), the values given are mean values. The industry standard THT was given the value 10.
- Table 2 shows the ratings for phenols of the formula (Ia), the execution was carried out as described in Example 1.
- Table 2 ⁇ / u> material material Me-Ac Et-Ac Bu-Ac rating 2-methoxyphenol 3 37 60 - 8.5 2-methoxyphenol 3 97 - - 8.5 3-methyl phenol 3 20 70 17 7.5 2-ethylphenol 3 37 60 - 8.5 2-ethylphenol 3 17 50 30 8th 2-isopropyl phenol 3 37 60 - 8.5 2-tert-butylphenol 3 37 60 - 8.5 2-methyl-5-isopropylphenol 3 37 60 - 8th 2-methyl-5-isopropylphenol 3 27 60 10 8th 2-methoxy-4-methylphenol 3 37 60 - 8.5 2-methoxy-4-methylphenol 3 - 97 - 8.5 2-tert-butyl-4-methylphenol 3 37 60 - 8th 2,6-diisopropylphenol 3 37 60 - 7.5
- Table 3 shows the ratings for anisole of the formula (Ib), the procedure was carried out as described in Example 1.
- Table 3 shows the ratings for anisole of the formula (Ib), the procedure was carried out as described in Example 1.
- Table 3 ⁇ u> Table 3: ⁇ / u> material material Me-Ac Et-Ac Bu-Ac rating anisole 3 37 60 - 8.5 anisole 3 97 - - 8th 2-Methylanisole 3 37 60 - 8.5 4-allylanisole 3 37 60 - 8.5 4-methylanisole 3 37 60 - 8.5 2-Methylanisole 3 17 50 30 7.5 2-Methylanisole 3 - 97 - 8th 4-methylanisole 3 27 60 10 8th 4-allylanisole 3 10 60 27 7.5
- Table 4 shows the evaluations for compounds of the formula (Ic), the procedure was carried out as described in Example 1.
- Table 4 ⁇ / u> material material Me-Ac Et-Ac Bu-Ac rating 2-methyl benzoxazole 3 37 60 - 8.5 2-methyl benzoxazole 3 45 30 22 7.5 2-Ethylbenzoxazol 3 37 60 - 8th 2-Acetylbenzoxazol 3 37 60 - 7.5
- Table 5 shows for comparison evaluations for phenols and phenol ethers, which are not in the context of the invention, the implementation was carried out as described in Example 1.
- Table 5 ⁇ u> Table 5: ⁇ / u> material material Me-Ac Et-Ac rating 4-aminophenol 3 37 60 3 3-nitroanisole 3 37 60 4 5-amino-2-methoxyphenol 3 37 60 3 2-Cyclohexylphenol 3 37 60 5 2,6-dimethyl-4-nitrophenol 3 37 60 2 4-Phenoxyphenol 3 37 60 3
- Table 6 shows odorants according to the invention with acrylates containing antioxidants. ⁇ u> Table 6: ⁇ / u> material material Me-Ac Et-Ac Bu-Ac BHT BHA Hydraulic 2-methoxyphenol 3 60 36.9 - 0.1.
- Table 7 shows odorants according to the invention with methacrylates containing antioxidants.
- Table 8 shows odorants according to the invention containing mixtures of compounds of the formula (I) according to the invention and antioxidants.
- Table 9 shows odorants according to the invention with acrylates and methacrylates containing 0.1 parts by weight of antioxidants (BHT or BHA).
- BHT antioxidants
Landscapes
- Chemical & Material Sciences (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Solid-Sorbent Or Filter-Aiding Compositions (AREA)
- Treating Waste Gases (AREA)
- Liquid Carbonaceous Fuels (AREA)
Claims (15)
- Utilisation de mélanges contenant au moins un acrylate d'alkyle en C1-C6 ou au moins un méthacrylate d'alkyle en C1-C6, au moins un composé de formule (1)
pour l'odorisation de gaz combustible ayant une teneur en méthane d'au moins 60 % en masse. - Utilisation selon la revendication 1, caractérisée en ce que les composés de formule (I) sont choisis parmi des composés de formule (Ia)
- Utilisation selon la revendication 1, caractérisée en ce que les composés de formule (I) sont choisis parmi les composés de formule (Ib)
- Utilisation selon la revendication 1, caractérisée en ce que les composés de formule (I) sont choisis parmi les composés de formule (Ic) ou (Id)
- Utilisation selon au moins l'une des revendications 1 à 4, caractérisée en ce que les composés de formule (I) sont liquides à 25°C et sous 1013 mbar.
- Utilisation selon l'une quelconque des revendications 1 à 5, caractérisée en ce que les acrylates d'alkyle en C1-C6 sont choisis parmi l'acrylate de méthyle, l'acrylate d'éthyle, l'acrylate de n-propyle, l'acrylate d'isopropyle, l'acrylate de n-butyle, l'acrylate d'isobutyle, l'acrylate de tert-butyle, l'acrylate de n-pentyle, l'acrylate d'isopentyle, l'acrylate de n-hexyle.
- Utilisation selon l'une quelconque des revendications 1 à 5, caractérisée en ce que les méthacrylates d'alkyle en C1-C6 sont choisis parmi le méthacrylate de méthyle, le méthacrylate d'éthyle, le méthacrylate de n-propyle, le méthacrylate d'isopropyle, le méthacrylate de n-butyle, le méthacrylate d'isobutyle, le méthacrylate de tert-butyle, le méthacrylate de n-pentyle, le méthacrylate d'isopentyle et le méthacrylate de n-hexyle.
- Utilisation selon au moins l'une des revendications 1 à 7, caractérisée en ce que les mélanges contiennent au moins deux acrylates d'alkyle en C1-C6.
- Utilisation selon au moins l'une des revendications 1 à 8, caractérisée en ce que les mélanges contiennent les composés de formule (I) en une proportion en masse de 1 à 100 pour 1000 parties en masse d'acrylates d'alkyle en C1-C6 et/ou de méthacrylates d'alkyle en C1-C6.
- Gaz combustible ayant une teneur en méthane d'au moins 60 % en masse, contenant au moins un acrylate d'alkyle en C1-C6 ou au moins un méthacrylate d'alkyle en C1-C6, au moins un composé de formule (I) et éventuellement un antioxydant.
- Gaz combustible selon la revendication 10, caractérisé en ce que le gaz combustible est du gaz naturel.
- Gaz naturel selon la revendication 10 ou 11, caractérisé en ce que l'antioxydant est choisi parmi un tert-butylhydroxytoluène, un tert-butylhydroxyanisole, l'éther monométhylique d'hydroquinone et l'α-tocophérol.
- Procédé d'odorisation de gaz combustible ayant une teneur en méthane d'au moins 60 % en masse, caractérisé en ce que l'on ajoute au gaz combustible un mélange contenant au moins un acrylate d'alkyle en C1-C6 ou au moins un méthacrylate d'alkyle en C1-C6, au moins un composé de formule (I), et éventuellement un antioxydant.
- Procédé selon la revendication 13, caractérisé en ce que la quantité totale d'antioxydants dans le mélange est de 0,001-1 % en masse.
- Procédé selon la revendication 13 ou 14, caractérisé en ce que l'on ajoute le mélange au gaz combustible en une quantité de 5-100 mg/m3 de gaz.
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE10235756A DE10235756A1 (de) | 2002-08-05 | 2002-08-05 | Gasodorierung mit Phenolen und/oder Phenolethern |
| DE10235756 | 2002-08-05 | ||
| PCT/EP2003/008597 WO2004015040A1 (fr) | 2002-08-05 | 2003-08-02 | Odorisation du gaz avec des phenols et/ou des ethers de phenol |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP1529093A1 EP1529093A1 (fr) | 2005-05-11 |
| EP1529093B1 true EP1529093B1 (fr) | 2007-05-16 |
Family
ID=30469446
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP03784163A Expired - Lifetime EP1529093B1 (fr) | 2002-08-05 | 2003-08-02 | Odorisation du gaz avec des phenols et/ou des ethers de phenol |
Country Status (5)
| Country | Link |
|---|---|
| EP (1) | EP1529093B1 (fr) |
| AT (1) | ATE362505T1 (fr) |
| AU (1) | AU2003258565A1 (fr) |
| DE (2) | DE10235756A1 (fr) |
| WO (1) | WO2004015040A1 (fr) |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE602005020843D1 (de) | 2004-12-22 | 2010-06-02 | Symrise Gmbh & Co Kg | Auf acrylat und acetophenon basierendes odoriermittel für wasserstoff |
| WO2016019074A1 (fr) | 2014-07-30 | 2016-02-04 | Georgia-Pacific Consumer Products Lp | Distributeurs d'assainisseur d'air, cartouches, systèmes et procédés associés |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3509054A (en) * | 1958-05-05 | 1970-04-28 | Ethyl Corp | Liquid hydrocarbon compositions of boron esters |
| JPS55104393A (en) * | 1979-02-02 | 1980-08-09 | Nippon Zeon Co Ltd | Fuel gas odorant |
| JPS55149391A (en) * | 1979-05-10 | 1980-11-20 | Riken Koryo Kogyo Kk | Odorant for fuel gas |
| US4487613A (en) * | 1983-09-26 | 1984-12-11 | International Flavors & Fragrances Inc. | Odorization of combustible hydrocarbon gases |
| DE19837066A1 (de) * | 1998-08-17 | 2000-02-24 | Haarmann & Reimer Gmbh | Odorierung von Gas |
-
2002
- 2002-08-05 DE DE10235756A patent/DE10235756A1/de not_active Withdrawn
-
2003
- 2003-08-02 WO PCT/EP2003/008597 patent/WO2004015040A1/fr not_active Ceased
- 2003-08-02 DE DE50307299T patent/DE50307299D1/de not_active Expired - Lifetime
- 2003-08-02 EP EP03784163A patent/EP1529093B1/fr not_active Expired - Lifetime
- 2003-08-02 AU AU2003258565A patent/AU2003258565A1/en not_active Abandoned
- 2003-08-02 AT AT03784163T patent/ATE362505T1/de not_active IP Right Cessation
Also Published As
| Publication number | Publication date |
|---|---|
| DE10235756A1 (de) | 2004-02-19 |
| EP1529093A1 (fr) | 2005-05-11 |
| WO2004015040A1 (fr) | 2004-02-19 |
| DE50307299D1 (de) | 2007-06-28 |
| AU2003258565A1 (en) | 2004-02-25 |
| ATE362505T1 (de) | 2007-06-15 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1329495B1 (fr) | Composition pour l'odorisation d'un gaz | |
| EP1537193B1 (fr) | Agents odorants a faible teneur en soufre destines a des gaz liquides | |
| EP1694801B1 (fr) | Odorisation de gaz combustible a l'aide d'odorisants pauvres en soufre | |
| EP1927646B1 (fr) | Agent odorant doté d'une stabilité améliorée | |
| EP2066766B1 (fr) | Agent odorant pauvre en soufre à stabilité améliorée | |
| EP1529093B1 (fr) | Odorisation du gaz avec des phenols et/ou des ethers de phenol | |
| EP1529092B1 (fr) | Odorisation de gaz par des cetones | |
| WO2004015038A1 (fr) | Odorisation de gaz contenant des acides carboxyliques et alcynes | |
| CN101260331A (zh) | 具有改善的稳定性的气体加味剂 | |
| WO2004024853A1 (fr) | Alcoxypyrazines d'odorisation de gaz | |
| EP1529091B1 (fr) | Ester d'odorisation de gaz combustibles | |
| WO2004015039A1 (fr) | Composes d'azote pour odoriser le gaz | |
| JPS5842234B2 (ja) | ネンリヨウガスヨウフシユウザイ |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| 17P | Request for examination filed |
Effective date: 20050307 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LI LU MC NL PT RO SE SI SK TR |
|
| AX | Request for extension of the european patent |
Extension state: AL LT LV MK |
|
| DAX | Request for extension of the european patent (deleted) | ||
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| RIN1 | Information on inventor provided before grant (corrected) |
Inventor name: KAESLER, HERIBERT Inventor name: MANSFELD, GERD Inventor name: BERTRAM, HEINZ-JUERGEN Inventor name: EILERS, JOERG Inventor name: WOLF, FLORIAN |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LI LU MC NL PT RO SE SI SK TR |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20070516 |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D Free format text: NOT ENGLISH |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D Free format text: LANGUAGE OF EP DOCUMENT: GERMAN |
|
| REF | Corresponds to: |
Ref document number: 50307299 Country of ref document: DE Date of ref document: 20070628 Kind code of ref document: P |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20070816 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: ES Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20070827 |
|
| NLV1 | Nl: lapsed or annulled due to failure to fulfill the requirements of art. 29p and 29m of the patents act | ||
| GBV | Gb: ep patent (uk) treated as always having been void in accordance with gb section 77(7)/1977 [no translation filed] |
Effective date: 20070516 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FD4D |
|
| EN | Fr: translation not filed | ||
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BG Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20070816 Ref country code: IE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20070516 Ref country code: PT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20071016 Ref country code: SI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20070516 Ref country code: CZ Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20070516 Ref country code: DK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20070516 Ref country code: NL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20070516 |
|
| BERE | Be: lapsed |
Owner name: RUHRGAS A.G. Effective date: 20070831 Owner name: SYMRISE G.M.B.H. & CO. KG Effective date: 20070831 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20070516 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| 26N | No opposition filed |
Effective date: 20080219 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MC Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20070831 Ref country code: GR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20070817 Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20070831 Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20070831 Ref country code: GB Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20070516 Ref country code: IT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20070516 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: RO Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20070516 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20080111 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20070831 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: AT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20070802 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: EE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20070516 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: CY Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20070516 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20070802 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: HU Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20071117 Ref country code: TR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20070516 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 20090821 Year of fee payment: 7 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R119 Ref document number: 50307299 Country of ref document: DE Effective date: 20110301 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20110301 |