EP2029575B1 - Antagonistes spirocycliques substitués des récepteurs cgrp - Google Patents
Antagonistes spirocycliques substitués des récepteurs cgrp Download PDFInfo
- Publication number
- EP2029575B1 EP2029575B1 EP07835747.2A EP07835747A EP2029575B1 EP 2029575 B1 EP2029575 B1 EP 2029575B1 EP 07835747 A EP07835747 A EP 07835747A EP 2029575 B1 EP2029575 B1 EP 2029575B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- unsubstituted
- substituted
- halo
- phenyl
- mmol
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 0 CC(C)(*)[S@](N)=O Chemical compound CC(C)(*)[S@](N)=O 0.000 description 6
- AQBNYNHZFPBXJF-UHFFFAOYSA-N Nc1ncc(CC(C2)(c3c(N4)nccc3)C4=O)c2c1 Chemical compound Nc1ncc(CC(C2)(c3c(N4)nccc3)C4=O)c2c1 AQBNYNHZFPBXJF-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/12—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/40—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil
- A61K31/403—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil condensed with carbocyclic rings, e.g. carbazole
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/04—Drugs for disorders of the alimentary tract or the digestive system for ulcers, gastritis or reflux esophagitis, e.g. antacids, inhibitors of acid secretion, mucosal protectants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
- A61P11/06—Antiasthmatics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P13/00—Drugs for disorders of the urinary system
- A61P13/10—Drugs for disorders of the urinary system of the bladder
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P15/00—Drugs for genital or sexual disorders; Contraceptives
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/06—Antipsoriatics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/02—Drugs for skeletal disorders for joint disorders, e.g. arthritis, arthrosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/04—Centrally acting analgesics, e.g. opioids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/06—Antimigraine agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/08—Antiepileptics; Anticonvulsants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/30—Drugs for disorders of the nervous system for treating abuse or dependence
- A61P25/36—Opioid-abuse
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/04—Antibacterial agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/08—Antiallergic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/12—Antihypertensives
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D409/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms
- C07D409/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings
- C07D413/12—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D417/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00
- C07D417/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing two hetero rings
- C07D417/12—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/04—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/12—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains three hetero rings
- C07D471/20—Spiro-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D519/00—Heterocyclic compounds containing more than one system of two or more relevant hetero rings condensed among themselves or condensed with a common carbocyclic ring system not provided for in groups C07D453/00 or C07D455/00
Definitions
- CGRP Calcitonin Gene-Related Peptide
- CGRP is a naturally occurring 37-amino acid peptide that is generated by tissue-specific alternate processing of calcitonin messenger RNA and is widely distributed in the central and peripheral nervous system.
- CGRP is localized predominantly in sensory afferent and central neurons and mediates several biological actions, including vasodilation.
- CGRP is expressed in alpha- and beta-forms that vary by one and three amino acids in the rat and human, respectively.
- CGRP-alpha and CGRP-beta display similar biological properties.
- CGRP When released from the cell, CGRP initiates its biological responses by binding to specific cell surface receptors that are predominantly coupled to the activation of adenylyl cyclase.
- CGRP receptors have been identified and pharmacologically evaluated in several tissues and cells, including those of brain, cardiovascular, endothelial, and smooth muscle origin.
- CGRP 1 and CGRP 2 Based on pharmacological properties, these receptors are divided into at least two subtypes, denoted CGRP 1 and CGRP 2 .
- CGRP is a potent neuromodulator that has been implicated in the pathology of cerebrovascular disorders such as migraine and cluster headache.
- CGRP-mediated activation of the trigeminovascular system may play a key role in migraine pathogenesis. Additionally, CGRP activates receptors on the smooth muscle of intracranial vessels, leading to increased vasodilation, which is thought to contribute to headache pain during migraine attacks ( Lance, Headache Pathogenesis: Monoamines, Neuropeptides, Purines and Nitric Oxide, Lippincott-Raven Publishers, 1997, 3-9 ).
- the middle meningeal artery the principle artery in the dura mater, is innervated by sensory fibers from the trigeminal ganglion which contain several neuropeptides, including CGRP.
- Trigeminal ganglion stimulation in the cat resulted in increased levels of CGRP, and in humans, activation of the trigeminal system caused facial flushing and increased levels of CGRP in the external jugular vein ( Goadsby et al., Ann. Neurol., 1988, 23, 193-196 ).
- Electrical stimulation of the dura mater in rats increased the diameter of the middle meningeal artery, an effect that was blocked by prior administration of CGRP(8-37), a peptide CGRP antagonist ( Williamson et al., Cephalalgia, 1997, 17, 525-531 ).
- Trigeminal ganglion stimulation increased facial blood flow in the rat, which was inhibited by CGRP(8-37) ( Escott et al., Brain Res. 1995, 669, 93-99 ). Electrical stimulation of the trigeminal ganglion in marmoset produced an increase in facial blood flow that could be blocked by the non-peptide CGRP antagonist BIBN4096BS ( Doods et al., Br. J. Pharmacol., 2000, 129, 420-423 ). Thus the vascular effects of CGRP may be attenuated, prevented or reversed by a CGRP antagonist.
- CGRP-mediated vasodilation of rat middle meningeal artery was shown to sensitize neurons of the trigeminal nucleus caudalis ( Williamson et al., The CGRP Family: Calcitonin Gene-Related Peptide (CGRP), Amylin, and Adrenomedullin, Austin Bioscience, 2000, 245-247 ).
- CGRP Calcitonin Gene-Related Peptide
- Amylin Amylin
- Adrenomedullin RNA derived neuropeptide
- distention of dural blood vessels during migraine headache may sensitize trigeminal neurons.
- Some of the associated symptoms of migraine, including extra-cranial pain and facial allodynia may be the result of sensitized trigeminal neurons ( Burstein et al., Ann. Neurol. 2000, 47, 614-624 ).
- a CGRP antagonist may be beneficial in attenuating, preventing or reversing the effects of neuronal sensitization.
- CGRP antagonists The ability of the compounds of the present invention to act as CGRP antagonists makes them useful pharmacological agents for disorders that involve CGRP in humans and animals, but particularly in humans.
- disorders include migraine and cluster headache ( Doods, Curr Opin Inves Drugs, 2001, 2 (9), 1261-1268 ; Edvinsson et al., Cephalalgia, 1994, 14, 320-327 ); chronic tension type headache ( Ashina et al., Neurology, 2000, 14, 1335-1340 ); pain ( Yu et al., Eur. J.
- non-insulin dependent diabetes mellitus Molina et al., Diabetes, 1990, 39, 260-265 ; vascular disorders; inflammation ( Zhang et al., Pain, 2001, 89, 265 ), arthritis, bronchial hyperreactivity, asthma, ( Foster et al., Ann. NY Acad. Sci., 1992, 657, 397-404 ; Schini et al., Am. J. Physiol., 1994, 267, H2483-H2490 ; Zheng et al., J. Virol., 1993, 67, 5786-5791 ); shock, sepsis ( Beer et al., Crit.
- Urology 2001, 166, 1720-1723 ); allergic dermatitis ( Wallengren, Contact Dermatitis, 2000, 43 (3), 137-143 ); psoriasis; encephalitis, brain trauma, ischaemia, stroke, epilepsy, and neurodegenerative diseases ( Rohrenbeck et al., Neurobiol. of Disease 1999, 6, 15-34 ); skin diseases ( Geppetti and Holzer, Eds., Neurogenic Inflammation, 1996, CRC Press, Boca Raton, FL ), neurogenic cutaneous redness, skin rosaceousness and erythema; tinnitus ( Herzog et al., J.
- WO 2006/029153 disclose classes of compounds that are CGRP antagonists.
- the present invention relates to compounds that are useful as ligands for CGRP receptors, in particular antagonists for CGRP receptors, processes for their preparation, their use in therapy, pharmaceutical compositions comprising them and methods of therapy using them.
- the present invention is directed to compounds of the formula I: (wherein A 1 , E a , E b , E c , R 6 , R 7 , R 10 and R 1 are defined herein) which are antagonists of CGRP receptors and which are useful in the treatment or prevention of diseases in which CGRP is involved, such as migraine.
- the invention is also directed to pharmaceutical compositions comprising these compounds and to these compounds and compositions for use in the prevention or treatment of such diseases in which CGRP is involved.
- the present invention is directed to compounds of the formula I: wherein:
- a 1 is CH 2 .
- a 1 is -N(R 8 )-, wherein R 8 is defined herein.
- a 1 is -NH-.
- R 5a , R 5b and R 5c are independently selected from hydrogen and halo.
- R 5a , R 5b and R 5c are hydrogen.
- R 6 and R 7 are ethyl, which are unsubstituted or substituted with 1-5 fluoro.
- R 6 and R 7 are methyl, which are unsubstituted or substituted with 1-3 fluoro.
- R 6 and R 7 and the carbon atom or atoms to which they are attached join to form a ring selected from cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, tetrahydropyranyl, pyrrolidinyl, and piperidinyl, which ring is unsubstituted or substituted with 1-6 substituents each independently selected from:
- R 8 is selected from: hydrogen, and - C 1-6 alkyl, which is unsubstituted or substituted with 1-5 fluoro.
- R 8 is hydrogen
- R 8 is methyl
- R 10 is hydrogen
- each such variable may be the same or different from each similarly designated variable.
- the compounds of the present invention may contain one or more asymmetric centers and can thus occur as racemates and racemic mixtures, single enantiomers, diastereomeric mixtures and individual diastereomers. Additional asymmetric centers may be present depending upon the nature of the various substituents on the molecule. Each such asymmetric center will independently produce two optical isomers and it is intended that all of the possible optical isomers and diastereomers in mixtures and as pure or partially purified compounds are included within the ambit of this invention. The present invention is meant to comprehend all such isomeric forms of these compounds.
- the present invention includes compounds of formula I wherein one or more hydrogen atoms are replaced by deuterium.
- Tautomers of compounds defined in Formula I are also included within the scope of the present invention.
- racemic mixtures of the compounds may be separated so that the individual enantiomers are isolated.
- the separation can be carried out by methods well known in the art, such as the coupling of a racemic mixture of compounds to an enantiomerically pure compound to form a diastereomeric mixture, followed by separation of the individual diastereomers by standard methods, such as fractional crystallization or chromatography.
- the coupling reaction is often the formation of salts using an enantiomerically pure acid or base.
- the diasteromeric derivatives may then be converted to the pure enantiomers by cleavage of the added chiral residue.
- the racemic mixture of the compounds can also be separated directly by chromatographic methods utilizing chiral stationary phases, which methods are well known in the art.
- any enantiomer of a compound may be obtained by stereoselective synthesis using optically pure starting materials or reagents of known configuration by methods well known in the art.
- halo or halogen as used herein are intended to include chloro, fluoro, bromo and iodo.
- alkyl is intended to mean linear or branched structures having no carbon-to-carbon double or triple bonds.
- C 1-6 alkyl is defined to identify the group as having 1, 2, 3, 4, 5 or 6 carbons in a linear or branched arrangement, such that C 1-6 alkyl specifically includes, but is not limited to, methyl, ethyl, n-propyl, iso-propyl, n-butyl, iso-butyl, tert-butyl, pentyl and hexyl.
- Cycloalkyl is an alkyl, part or all of which which forms a ring of three or more atoms.
- phrases "pharmaceutically acceptable” is employed herein to refer to those compounds, materials, compositions, and/or dosage forms which are, within the scope of sound medical judgment, suitable for use in contact with the tissues of human beings and animals without excessive toxicity, irritation, allergic response, or other problem or complication, commensurate with a reasonable benefit/risk ratio.
- pharmaceutically acceptable salts refer to derivatives wherein the parent compound is modified by making acid or base salts thereof.
- pharmaceutically acceptable salts include, but are not limited to, mineral or organic acid salts of basic residues such as amines; alkali or organic salts of acidic residues such as carboxylic acids; and the like.
- the pharmaceutically acceptable salts include the conventional non-toxic salts or the quaternary ammonium salts of the parent compound formed, for example, from non-toxic inorganic or organic acids.
- such conventional non-toxic salts include those derived from inorganic acids such as hydrochloric, hydrobromic, sulfuric, sulfamic, phosphoric, nitric and the like; and the salts prepared from organic acids such as acetic, propionic, succinic, glycolic, stearic, lactic, malic, tartaric, citric, ascorbic, pamoic, maleic, hydroxymaleic, phenylacetic, glutamic, benzoic, salicylic, sulfanilic, 2-acetoxybenzoic, fumaric, toluenesulfonic, methanesulfonic, ethane disulfonic, oxalic, isethionic, and the like.
- inorganic acids such as hydrochloric, hydrobromic, sulfuric, sulfamic, phosphoric, nitric and the like
- organic acids such as acetic, propionic, succinic, glycolic, stearic, lactic,
- salts may be prepared from pharmaceutically acceptable non-toxic acids, including inorganic and organic acids.
- acids include acetic, benzenesulfonic, benzoic, camphorsulfonic, citric, ethanesulfonic, fumaric, gluconic, glutamic, hydrobromic, hydrochloric, isethionic, lactic, maleic, malic, mandelic, methanesulfonic, mucic, nitric, pamoic, pantothenic, phosphoric, succinic, sulfuric, tartaric, p-toluenesulfonic acid, and the like.
- the salts are citric, hydrobromic, hydrochloric, maleic, phosphoric, sulfuric, fumaric, and tartaric acids. It will be understood that, as used herein, references to the compounds of Formula I are meant to also include the pharmaceutically acceptable salts.
- Exemplifying the invention is the use of the compounds disclosed in the Examples and herein.
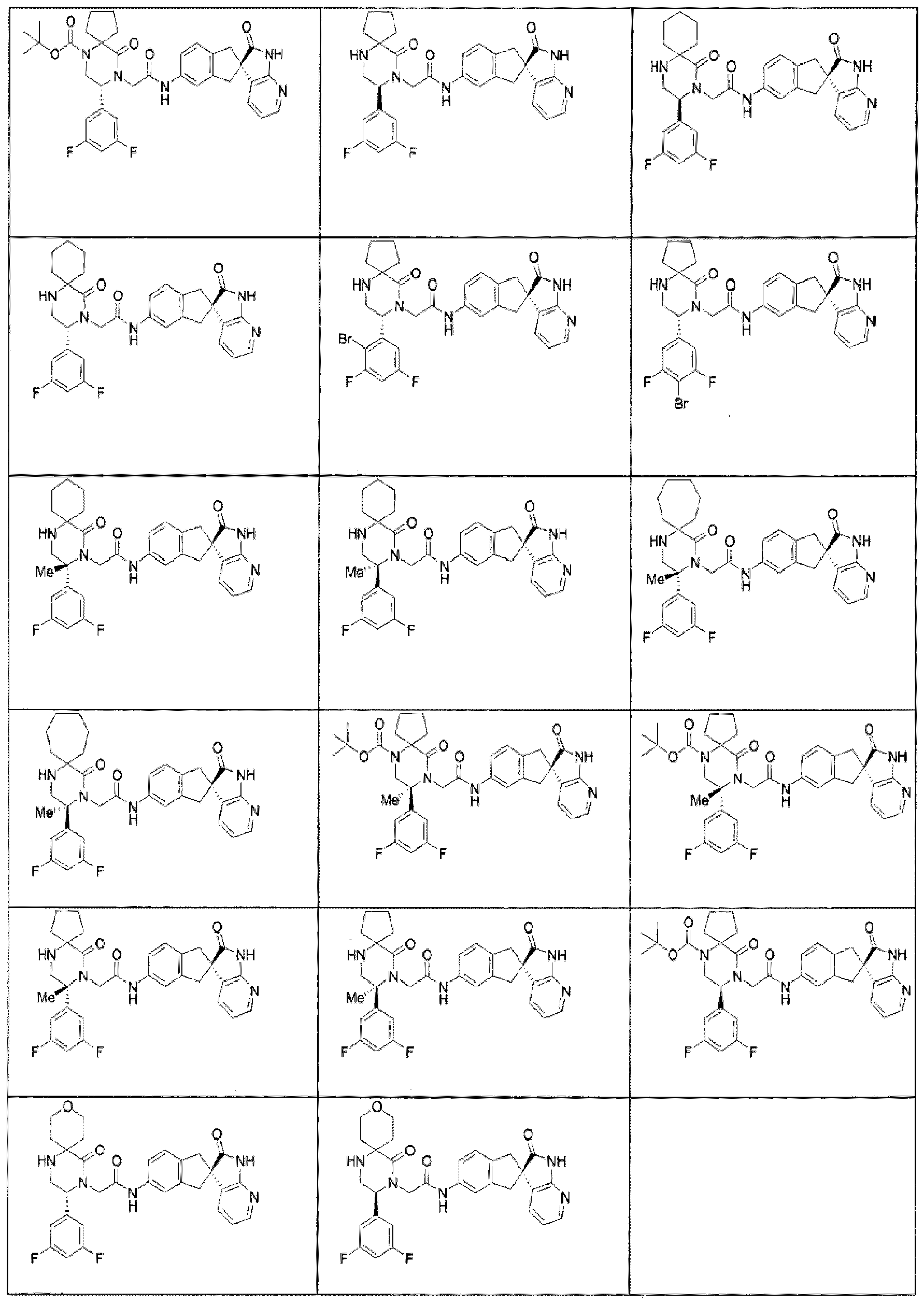
- Specific compounds within the present invention include a compound which is selected from the group consisting of the compounds disclosed in the following Examples and pharmaceutically acceptable salts thereof and individual diastereomers thereof.
- the subject compounds are useful in a method of antagonism of CGRP receptors in a patient such as a mammal in need of such antagonism comprising the administration of an effective amount of the compound.
- the present invention is directed to the compounds disclosed herein for use as antagonists of CGRP receptors.
- a variety of other mammals can be treated according to the method disclosed.
- the present invention is further directed to a method for the manufacture of a medicament for antagonism of CGRP receptors activity in humans and animals comprising combining a compound of the present invention with a pharmaceutical carrier or diluent.
- the subject treated in the present methods is generally a mammal, for example a human being, male or female, in whom antagonism of CGRP receptor activity is desired.
- therapeutically effective amount means the amount of the subject compound that will elicit the biological or medical response of a tissue, system, animal or human that is being sought by the researcher, veterinarian, medical doctor or other clinician.
- treatment refers both to the treatment and to the prevention or prophylactic therapy of the mentioned conditions, particularly in a patient who is predisposed to such disease or disorder.
- composition as used herein is intended to encompass a product comprising the specified ingredients in the specified amounts, as well as any product which results, directly or indirectly, from combination of the specified ingredients in the specified amounts.
- Such term in relation to pharmaceutical composition is intended to encompass a product comprising the active ingredient(s), and the inert ingredient(s) that make up the carrier, as well as any product which results, directly or indirectly, from combination, complexation or aggregation of any two or more of the ingredients, or from dissociation of one or more of the ingredients, or from other types of reactions or interactions of one or more of the ingredients.
- the pharmaceutical compositions of the present invention encompass any composition made by admixing a compound of the present invention and a pharmaceutically acceptable carrier.
- pharmaceutically acceptable it is meant the carrier, diluent or excipient must be compatible with the other ingredients of the formulation and not deleterious to the recipient thereof.
- NATIVE RECEPTOR BINDING ASSAY The binding of 125 I-CGRP to receptors in SK-N-MC cell membranes was carried out essentially as described ( Edvinsson et al. (2001) Eur. J. Pharmacol. 415, 39-44 ). Briefly, membranes (25 ⁇ g) were incubated in 1 mL of binding buffer [10 mM HEPES, pH 7.4, 5 mM MgCl 2 and 0.2% bovine serum albumin (BSA)] containing 10 pM 125 I-CGRP and antagonist.
- binding buffer 10 mM HEPES, pH 7.4, 5 mM MgCl 2 and 0.2% bovine serum albumin (BSA)
- the assay was terminated by filtration through GFB glass fibre filter plates (PerkinElmer) that had been blocked with 0.5% polyethyleneimine for 3 h.
- the filters were washed three times with ice-cold assay buffer (10 mM HEPES, pH 7.4 and 5 mM MgCl 2 ), then the plates were air dried. Scintillation fluid (50 ⁇ L) was added and the radioactivity was counted on a Topcount (Packard Instrument). Data analysis was carried out by using Prism and the K i was determined by using the Cheng-Prusoff equation (Cheng & Prusoff (1973) Biochem. Pharmacol. 22 , 3099-3108).
- RECOMBINANT RECEPTOR Human CL receptor (Genbank accession number L76380) was subcloned into the expression vector pIREShyg2 (BD Biosciences Clontech) as a 5'NheI and 3' PmeI fragment. Human RAMP 1 (Genbank accession number AJ001014) was subcloned into the expression vector pIRESpuro2 (BD Biosciences Clontech) as a 5'NheI and 3'NotI fragment.
- HEK 293 cells human embryonic kidney cells; ATCC #CRL-1573
- DMEM fetal bovine serum
- FBS fetal bovine serum
- penicillin 100 ⁇ g/mL streptomycin
- Stable cell line generation was accomplished by co-transfecting 10 ⁇ g of DNA with 30 ⁇ g Lipofectamine 2000 (Invitrogen) in 75 cm 2 flasks.
- CL receptor and RAMP1 expression constructs were co-transfected in equal amounts. Twenty-four hours after transfection the cells were diluted and selective medium (growth medium + 300 ⁇ g/mL hygromycin and 1 ⁇ g/mL puromycin) was added the following day. A clonal cell line was generated by single cell deposition utilizing a FACS Vantage SE (Becton Dickinson). Growth medium was adjusted to 150 ⁇ g/mL hygromycin and 0.5 ⁇ g/mL puromycin for cell propagation.
- selective medium growth medium + 300 ⁇ g/mL hygromycin and 1 ⁇ g/mL puromycin
- RECOMBINANT RECEPTOR BINDING ASSAY Cells expressing recombinant human CL receptor/RAMP1 were washed with PBS and harvested in harvest buffer containing 50 mM HEPES, 1 mM EDTA and Complete protease inhibitors (Roche). The cell suspension was disrupted with a laboratory homogenizer and centrifuged at 48,000 g to isolate membranes. The pellets were resuspended in harvest buffer plus 250 mM sucrose and stored at - 70°C.
- binding assays 20 ⁇ g of membranes were incubated in 1 ml binding buffer (10 mM HEPES, pH 7.4, 5 mM MgCl 2 , and 0.2% BSA) for 3 hours at room temperature containing 10 pM 125 I-hCGRP (GE Healthcare) and antagonist.
- the assay was terminated by filtration through 96-well GFB glass fiber filter plates (PerkinElmer) that had been blocked with 0.05% polyethyleneimine.
- the filters were washed 3 times with ice-cold assay buffer (10 mM HEPES, pH 7.4 and 5 mM MgCl 2 ). Scintillation fluid was added and the plates were counted on a Topcount (Packard).
- Non-specific binding was determined and the data analysis was carried out with the apparent dissociation constant (K i ) determined by using a non-linear least squares fitting the bound CPM data to the equation below:
- K i apparent dissociation constant
- Y obsd ⁇ Y max - Y min ⁇ % I max - % I min / 100 + Y min + Y max - Y min ⁇ 100 - % I max / 100 ⁇
- Y is observed CPM bound
- Y max is total bound counts
- Y min is non specific bound counts
- (Y max - Y min ) is specific bound counts
- % I max is the maximum percent inhibition
- % I min is the minimum percent inhibition
- radiolabel is the probe
- the K d is the apparent dissociation constant for the radioligand for the receptor as determined by Hot saturation experiments.
- RECOMBINANT RECEPTOR FUNCTIONAL ASSAY Cells were plated in complete growth medium at 85,000 cells/well in 96-well poly-D-lysine coated plates (Corning) and cultured for ⁇ 19 h before assay. Cells were washed with PBS and then incubated with inhibitor for 30 min at 37 °C and 95% humidity in Cellgro Complete Serum-Free/Low-Protein medium (Mediatech, Inc.) with L-glutamine and 1 g/L BSA. Isobutyl-methylxanthine was added to the cells at a concentration of 300 ⁇ M and incubated for 30 min at 37 °C.
- the compounds of the following examples had activity as antagonists of the CGRP receptor in the aforementioned assays, generally with a K i or IC 50 value of less than about 50 ⁇ M. Such a result is indicative of the intrinsic activity of the compounds in use as antagonists of CGRP receptors.
- the ability of the compounds of the present invention to act as CGRP antagonists makes them useful pharmacological agents for disorders that involve CGRP in humans and animals, but particularly in humans.
- the compounds of the present invention have utility in treating, preventing, ameliorating, controlling or reducing the risk of one or more of the following conditions or diseases: headache; migraine; cluster headache; chronic tension type headache; pain; chronic pain; neurogenic inflammation and inflammatory pain; neuropathic pain; eye pain; tooth pain; diabetes; non-insulin dependent diabetes mellitus; vascular disorders; inflammation; arthritis; bronchial hyperreactivity, asthma; shock; sepsis; opiate withdrawal syndrome; morphine tolerance; hot flashes in men and women; allergic dermatitis; psoriasis; encephalitis; brain trauma; epilepsy; neurodegenerative diseases; skin diseases; neurogenic cutaneous redness, skin rosaceousness and erythema; inflammatory bowel disease, irritable bowel syndrome, cystitis; and other conditions that may be treated or prevented by antagonism of CGRP receptors.
- headache migraine; cluster headache; chronic tension type headache; pain; chronic pain; neurogenic inflammation and inflammatory pain; neuropathic pain; eye
- the subject compounds are further useful in a method for the prevention, treatment, control, amelioration, or reduction of risk of the diseases, disorders and conditions noted herein.
- the subject compounds are further useful in a method for the prevention, treatment, control, amelioration, or reduction of risk of the aforementioned diseases, disorders and conditions in combination with other agents.
- the compounds of the present invention may be used in combination with one or more other drugs in the treatment, prevention, control, amelioration, or reduction of risk of diseases or conditions for which compounds of Formula I or the other drugs may have utility, where the combination of the drugs together are safer or more effective than either drug alone.
- Such other drug(s) may be administered, by a route and in an amount commonly used therefor, contemporaneously or sequentially with a compound of Formula I.
- a pharmaceutical composition in unit dosage form containing such other drugs and the compound of Formula I is preferred.
- the combination therapy may also include therapies in which the compound of Formula I and one or more other drugs are administered on different overlapping schedules.
- compositions of the present invention include those that contain one or more other active ingredients, in addition to a compound of Formula I.
- the present compounds may be used in conjunction with an an anti-migraine agent, such as ergotamine and dihydroergotamine, or other serotonin agonists, especially a 5-HT 1B/1D agonist, for example sumatriptan, naratriptan, zolmitriptan, eletriptan, almotriptan, frovatriptan, donitriptan, and rizatriptan, a 5-HT 1D agonist such as PNU-142633 and a 5-HT 1F agonist such as LY334370; a cyclooxygenase inhibitor, such as a selective cyclooxygenase-2 inhibitor, for example rofecoxib, etoricoxib, celecoxib, valdecoxib or paracoxib; a non-steroidal anti-inflammatory agent or a cytokine-suppressing anti-inflammatory agent, for example with a compound such as ibuprofen, ketoprofen, fen
- the instant compounds may be administered with an analgesic such as aspirin, acetaminophen, phenacetin, fentanyl, sufentanil, methadone, acetyl methadol, buprenorphine or morphine.
- an analgesic such as aspirin, acetaminophen, phenacetin, fentanyl, sufentanil, methadone, acetyl methadol, buprenorphine or morphine.
- an interleukin inhibitor such as an interleukin-1 inhibitor; an NK-1 receptor antagonist, for example aprepitant; an NMDA antagonist; an NR2B antagonist; a bradykinin-1 receptor antagonist; an adenosine A1 receptor agonist; a sodium channel blocker, for example lamotrigine; an opiate agonist such as levomethadyl acetate or methadyl acetate; a lipoxygenase inhibitor, such as an inhibitor of 5-lipoxygenase; an alpha receptor antagonist, for example indoramin; an alpha receptor agonist; a vanilloid receptor antagonist; a renin inhibitor; a granzyme B inhibitor; a substance P antagonist; an endothelin antagonist; a norepinephrin precursor; anti-anxiety agents such as diazepam, alprazolam, chlordiazepoxide and chlorazepate; serotonin 5HT 2
- an interleukin inhibitor such as an
- the present compounds may be used in conjunction with gap junction inhibitors; neuronal calcium channel blockers such as civamide; AMPA/KA antagonists such as LY293558; sigma receptor agonists; and vitamin B2.
- the present compounds may be used in conjunction with ergot alkaloids other than ergotamine and dihydroergotamine, for example ergonovine, ergonovine, methylergonovine, metergoline, ergoloid mesylates, dihydroergocornine, dihydroergocristine, dihydroergocryptine, dihydro- ⁇ -ergocryptine, dihydro- ⁇ -ergocryptine, ergotoxine, ergocornine, ergocristine, ergocryptine, ⁇ -ergocryptine, ⁇ -ergocryptine, ergosine, ergostane, bromocriptine, or methysergide.
- ergonovine ergonovine, methylergonovine, metergoline, ergoloid mesylates
- dihydroergocornine dihydroergocristine
- dihydroergocryptine dihydro- ⁇ -ergocryptine
- the present compounds may be used in conjunction with a beta-adrenergic antagonist such as timolol, propanolol, atenolol, metoprolol or nadolol, and the like; a MAO inhibitor, for example phenelzine; a calcium channel blocker, for example flunarizine, diltiazem, amlodipine, felodipine, nisolipine, isradipine, nimodipine, lomerizine, verapamil, nifedipine, or prochlorperazine; neuroleptics such as olanzapine, droperidol, prochlorperazine, chlorpromazine and quetiapine; an anticonvulsant such as topiramate, zonisamide, tonabersat, carabersat, levetiracetam, lamotrigine, tiagabine, gabapentin, pregabalin or di
- the present compounds may be used in conjunction with a potentiator such as caffeine, an H2-antagonist, simethicone, aluminum or magnesium hydroxide; a decongestant such as oxymetazoline, epinephrine, naphazoline, xylometazoline, propylhexedrine, or levo-desoxy-ephedrine; an antitussive such as caramiphen, carbetapentane, or dextromethorphan; a diuretic; a prokinetic agent such as metoclopramide or domperidone; a sedating or non-sedating antihistamine such as acrivastine, azatadine, bromodiphenhydramine, brompheniramine, carbinoxamine, chlorpheniramine, clemastine, dexbrompheniramine, dexchlorpheniramine, diphenhydramine, doxylamine, loratadine, phenindamine, pheniramine
- the present compounds are used in conjunction with an anti-migraine agent, such as: ergotamine or dihydroergotamine; a 5-HT 1 agonist, especially a 5-HT 1B/1D agonist, in particular, sumatriptan, naratriptan, zolmitriptan, eletriptan, almotriptan, frovatriptan, donitriptan, avitriptan and rizatriptan, and other serotonin agonists; and a cyclooxygenase inhibitor, such as a selective cyclooxygenase-2 inhibitor, in particular, rofecoxib, etoricoxib, celecoxib, valdecoxib or paracoxib.
- an anti-migraine agent such as: ergotamine or dihydroergotamine; a 5-HT 1 agonist, especially a 5-HT 1B/1D agonist, in particular, sumatriptan, naratriptan, zolmitriptan, ele
- the above combinations include combinations of a compound of the present invention not only with one other active compound, but also with two or more other active compounds.
- compounds of the present invention may be used in combination with other drugs that are used in the prevention, treatment, control, amelioration, or reduction of risk of the diseases or conditions for which compounds of the present invention are useful.
- Such other drugs may be administered, by a route and in an amount commonly used therefore, contemporaneously or sequentially with a compound of the present invention.
- a pharmaceutical composition containing such other drugs in addition to the compound of the present invention is preferred.
- the pharmaceutical compositions of the present invention include those that also contain one or more other active ingredients, in addition to a compound of the present invention.
- the weight ratio of the compound of the compound of the present invention to the other active ingredient(s) may be varied and will depend upon the effective dose of each ingredient. Generally, an effective dose of each will be used. Thus, for example, when a compound of the present invention is combined with another agent, the weight ratio of the compound of the present invention to the other agent will generally range from about 1000:1 to about 1:1000, or from about 200:1 to about 1:200. Combinations of a compound of the present invention and other active ingredients will generally also be within the aforementioned range, but in each case, an effective dose of each active ingredient should be used.
- the compound of the present invention and other active agents may be administered separately or in conjunction.
- the administration of one element may be prior to, concurrent to, or subsequent to the administration of other agent(s), and via the same or different routes of administration.
- the compounds of the present invention may be administered by oral, parenteral (e.g., intramuscular, intraperitoneal, intravenous, ICV, intracisternal injection or infusion, subcutaneous injection, or implant), by inhalation spray, nasal, vaginal, rectal, sublingual, or topical routes of administration and may be formulated, alone or together, in suitable dosage unit formulations containing conventional non-toxic pharmaceutically acceptable carriers, adjuvants and vehicles appropriate for each route of administration.
- parenteral e.g., intramuscular, intraperitoneal, intravenous, ICV, intracisternal injection or infusion, subcutaneous injection, or implant
- inhalation spray nasal, vaginal, rectal, sublingual, or topical routes of administration
- nasal, vaginal, rectal, sublingual, or topical routes of administration may be formulated, alone or together, in suitable dosage unit formulations containing conventional non-toxic pharmaceutically acceptable carriers, adjuvants and vehicles appropriate for each route of administration.
- the compounds of the invention are effective for
- compositions for the administration of the compounds of this invention may conveniently be presented in dosage unit form and may be prepared by any of the methods well known in the art of pharmacy. All methods include the step of bringing the active ingredient into association with the carrier which constitutes one or more accessory ingredients.
- the pharmaceutical compositions are prepared by uniformly and intimately bringing the active ingredient into association with a liquid carrier or a finely divided solid carrier or both, and then, if necessary, shaping the product into the desired formulation.
- the active compound is included in an amount sufficient to produce the desired effect upon the process or condition of diseases.
- composition is intended to encompass a product comprising the specified ingredients in the specified amounts, as well as any product which results, directly or indirectly, from combination of the specified ingredients in the specified amounts.
- compositions containing the active ingredient may be in a form suitable for oral use, for example, as tablets, troches, lozenges, aqueous or oily suspensions, dispersible powders or granules, emulsions, solutions, hard or soft capsules, or syrups or elixirs.
- Compositions intended for oral use may be prepared according to any method known to the art for the manufacture of pharmaceutical compositions and such compositions may contain one or more agents selected from the group consisting of sweetening agents, flavoring agents, coloring agents and preserving agents in order to provide pharmaceutically elegant and palatable preparations. Tablets contain the active ingredient in admixture with non-toxic pharmaceutically acceptable excipients which are suitable for the manufacture of tablets.
- excipients may be for example, inert diluents, such as calcium carbonate, sodium carbonate, lactose, calcium phosphate or sodium phosphate; granulating and disintegrating agents, for example, corn starch, or alginic acid; binding agents, for example starch, gelatin or acacia; and lubricating agents, for example magnesium stearate, stearic acid or talc.
- the tablets may be uncoated or they may be coated by known techniques to delay disintegration and absorption in the gastrointestinal tract and thereby provide a sustained action over a longer period.
- a time delay material such as glyceryl monostearate or glyceryl distearate may be employed.
- Oral tablets may also be coated by the techniques described in the U.S. Patents 4,256,108 ; 4,166,452 ; and 4,265,874 to form osmotic therapeutic tablets for control release.
- Oral tablets may also be formulated for immediate release, such as fast melt tablets or wafers, rapid dissolve tablets or fast dissolve films.
- Formulations for oral use may also be presented as hard gelatin capsules wherein the active ingredient is mixed with an inert solid diluent, for example, calcium carbonate, calcium phosphate or kaolin, or as soft gelatin capsules wherein the active ingredient is mixed with water or an oil medium, for example peanut oil, liquid paraffin, or olive oil.
- an inert solid diluent for example, calcium carbonate, calcium phosphate or kaolin
- water or an oil medium for example peanut oil, liquid paraffin, or olive oil.
- Aqueous suspensions contain the active materials in admixture with excipients suitable for the manufacture of aqueous suspensions.
- excipients are suspending agents, for example sodium carboxymethylcellulose, methylcellulose, hydroxy-propylmethylcellulose, sodium alginate, polyvinyl-pyrrolidone, gum tragacanth and gum acacia; dispersing or wetting agents may be a naturally-occurring phosphatide, for example lecithin, or condensation products of an alkylene oxide with fatty acids, for example polyoxyethylene stearate, or condensation products of ethylene oxide with long chain aliphatic alcohols, for example heptadecaethyleneoxycetanol, or condensation products of ethylene oxide with partial esters derived from fatty acids and a hexitol such as polyoxyethylene sorbitol monooleate, or condensation products of ethylene oxide with partial esters derived from fatty acids and hexitol anhydrides, for example polyethylene sorbitan monoo
- the aqueous suspensions may also contain one or more preservatives, for example ethyl, or n-propyl, p-hydroxybenzoate, one or more coloring agents, one or more flavoring agents, and one or more sweetening agents, such as sucrose or saccharin.
- preservatives for example ethyl, or n-propyl, p-hydroxybenzoate
- coloring agents for example ethyl, or n-propyl, p-hydroxybenzoate
- coloring agents for example ethyl, or n-propyl, p-hydroxybenzoate
- flavoring agents for example ethyl, or n-propyl, p-hydroxybenzoate
- sweetening agents such as sucrose or saccharin.
- Oily suspensions may be formulated by suspending the active ingredient in a vegetable oil, for example arachis oil, olive oil, sesame oil or coconut oil, or in a mineral oil such as liquid paraffin.
- the oily suspensions may contain a thickening agent, for example beeswax, hard paraffin or cetyl alcohol. Sweetening agents such as those set forth above, and flavoring agents may be added to provide a palatable oral preparation. These compositions may be preserved by the addition of an anti-oxidant such as ascorbic acid.
- Dispersible powders and granules suitable for preparation of an aqueous suspension by the addition of water provide the active ingredient in admixture with a dispersing or wetting agent, suspending agent and one or more preservatives.
- a dispersing or wetting agent e.g., glycerol, glycerol, glycerol, glycerol, glycerol, glycerol, glycerin, glycerin, glycerin, glycerin, glycerin, sorbitol, sorbitol, sorbitol, sorbitol, sorbitol, sorbitol, sorbitol, sorbitol, sorbitol, sorbitol, glycerol, glycerol, glycerol, glycerol, glycerol, glycerol, glycerol, glycerol, glycerol
- the pharmaceutical compositions of the invention may also be in the form of oil-in-water emulsions.
- the oily phase may be a vegetable oil, for example olive oil or arachis oil, or a mineral oil, for example liquid paraffin or mixtures of these.
- Suitable emulsifying agents may be naturally- occurring gums, for example gum acacia or gum tragacanth, naturally-occurring phosphatides, for example soy bean, lecithin, and esters or partial esters derived from fatty acids and hexitol anhydrides, for example sorbitan monooleate, and condensation products of the said partial esters with ethylene oxide, for example polyoxyethylene sorbitan monooleate,
- the emulsions may also contain sweetening and flavoring agents.
- Syrups and elixirs may be formulated with sweetening agents, for example glycerol, propylene glycol, sorbitol or sucrose. Such formulations may also contain a demulcent, a preservative and flavoring and coloring agents.
- sweetening agents for example glycerol, propylene glycol, sorbitol or sucrose.
- Such formulations may also contain a demulcent, a preservative and flavoring and coloring agents.
- the pharmaceutical compositions may be in the form of a sterile injectable aqueous or oleagenous suspension.
- This suspension may be formulated according to the known art using those suitable dispersing or wetting agents and suspending agents which have been mentioned above.
- the sterile injectable preparation may also be a sterile injectable solution or suspension in a non-toxic parenterally-acceptable diluent or solvent, for example as a solution in 1,3-butane diol.
- the acceptable vehicles and solvents that may be employed are water, Ringer's solution and isotonic sodium chloride solution.
- sterile, fixed oils are conventionally employed as a solvent or suspending medium.
- any bland fixed oil may be employed including synthetic mono- or diglycerides.
- fatty acids such as oleic acid find use in the preparation of injectables.
- the compounds of the present invention may also be administered in the form of suppositories for rectal administration of the drug.
- These compositions can be prepared by mixing the drug with a suitable non-irritating excipient which is solid at ordinary temperatures but liquid at the rectal temperature and will therefore melt in the rectum to release the drug.
- suitable non-irritating excipient which is solid at ordinary temperatures but liquid at the rectal temperature and will therefore melt in the rectum to release the drug.
- Such materials are cocoa butter and polyethylene glycols.
- transdermal patches may also be used for topical administration.
- compositions and method of the present invention may further comprise other therapeutically active compounds as noted herein which are usually applied in the treatment of the above mentioned pathological conditions.
- an appropriate dosage level will generally be about 0.01 to 500 mg per kg patient body weight per day which can be administered in single or multiple doses.
- a suitable dosage level may be about 0.01 to 250 mg/kg per day, about 0.05 to 100 mg/kg per day, or about 0.1 to 50 mg/kg per day. Within this range the dosage may be 0.05 to 0.5, 0.5 to 5 or 5 to 50 mg/kg per day.
- the compositions are may be provided in the form of tablets containing 1.0 to 1000 milligrams of the active ingredient, particularly 1.0, 5.0, 10.0, 15.0.
- the compounds may be administered on a regimen of 1 to 4 times per day, or may be administered once or twice per day.
- the compounds of the present invention are administered at a daily dosage of from about 0.1 milligram to about 100 milligram per kilogram of animal body weight, given as a single daily dose or in divided doses two to six times a day, or in sustained release form.
- the total daily dosage is from about 1.0 milligrams to about 1000 milligrams, or from about 1 milligrams to about 50 milligrams. In the case of a 70 kg adult human, the total daily dose will generally be from about 7 milligrams to about 350 milligrams. This dosage regimen may be adjusted to provide the optimal therapeutic response.
- the compounds of the present invention can be prepared readily according to the following Schemes and specific examples, or modifications thereof, using readily available starting materials, reagents and conventional synthesis procedures. In these reactions, it is also possible to make use of variants which are themselves known to those of ordinary skill in this art but are not mentioned in greater detail.
- the general procedures for making the compounds claimed in this invention can be readily understood and appreciated by one skilled in the art from viewing the following Schemes.
- Lactam 1 can be deprotonated with the strong base sodium hydride, in THF, to provide the amide anion which smoothly reacts with the electrophilic methyl bromoacetate. Subsequent treatment of this ester product, still in THF, with aqueous sodium hydroxide gives the acid 2. Acid 2 can be coupled to the known aniline 3 ( Bell, I. M., et al., PCT Int. Appl., WO 2004082605 A2 20040930 ) employing the standard peptide coupling reagent combination of EDCI, HOAt and triethylamine, in DMF, to provided the claimed compound 4. Analogs of lactam 1, if not commercially available, can be prepared by a variety of common methods, one of which is illustrated in Scheme 2, for the preparation of Intermediate 1.
- Anhydride 5 can be converted to the bis-ester 6, though heating in methanol, in the presence of HCl (generated from the reaction of TMSCl with MeOH).
- the less hindered ester of 6 can be selectively hydrolyzed with potassium carbonate, in a mixture of THF, water and MeOH over the course of a few days.
- This acid can be converted in situ to the corresponding acid chloride utilizing oxalyl chloride and a catalytic amount of DMF, in DCM, at ambient temperature.
- excess amine or an amine hydrochloride-triethylamine mixture
- the amide 8 yields the ketone 9 upon treatment with a Grignard reagent, or alternative organometallics, in THF at reduced temperatures.
- a Grignard reagent or alternative organometallics
- the pyrrolidinyl amide analog of 8 can be of similar utility.
- Aryl ketone 9 can then be transformed into the sulfinamide 10 according to the one-pot procedure of Ellman and coworkers, Tetrahedron Lett., 1999, 40, 6709-6712 .
- Treatment of 10 with anhydrous HCl in MeOH removes the t-butyl sulfinyl group, and upon addition of sufficient triethylamine, produces Intermediate 1 (compound 11), where heating is employed as necessary.
- the aryl ketone 9 (from Scheme 2) can be transformed into the sulfinimine 19 utilizing Ti(OEt) 4 in THF at elevated temperatures.
- the sulfmimine can then be treated with a Grignard reagent, or alternative organometallics, in THF at reduced temperatures to produce the sulfinamide 20.
- Treatment of 20 with anhydrous HCl in MeOH removes the t-butyl sulfinyl group, and upon addition of sufficient triethylamine, produces the lactam 21, under refluxing conditions in toluene. Lactam 21 can be deprotonated with the strong base potassium hydride, in THF, to provide the amide anion which smoothly reacts with methyl bromoacetate.
- Scheme 7 illustrates a route to the 3-aminopyridine 41.
- 7-Azaindole ( 33 ) may be protected with a variety of protecting groups, such as the 2-(trimethylsilyl)ethoxymethyl group shown in Scheme 7.
- protecting groups such as the 2-(trimethylsilyl)ethoxymethyl group shown in Scheme 7.
- treatment of 34 with pyridine hydrobromide perbromide provides the dibromoazaoxindole 35, which may be reduced to the corresponding azaoxindole 36 by reaction with zinc.
- Bis-alkylation of the azaoxindole 36 with 1,4-dibromobutan-2-one [ de Meijere et al. (2001) Eur. J. Org. Chem.
- Spiroazaoxindole intermediates such as those illustrated in these schemes ( vide supra ), may be resolved to give pure enantiomers using techniques familiar to those skilled in the art. For example, chromatography of the suitable intermediates on a chiral column can be used to provide the individual stereoisomers. Resolution may also be effected by other methodologies, such as fractional crystallization of diastereomeric salts, and it may be carried out on other synthetic intermediates or on the final products. Alternatively, an asymmetric synthesis of a key intermediate could be used to provide an enantiomerically enriched final product.
- Scheme 10 illustrates a route to 6-substituted piperidin-2-ones, based upon addition of a Grignard reagent (R'MgBr) to the tert -butylsulfinyl imine 100, in analogy with methodology developed by Ellman and coworkers ( Ellman et al., Acc. Chem. Res., 2002, 35, 984-995 ).
- Grignard reagent which usually proceeds with high diastereoselectivity
- the mixture may be heated to reflux to effect deprotection of the resulting sulfinamide and cyclization to provide predominantly one enantiomer of the piperidinone 101.
- organometallic reagent such as an organolithium (R'Li), which may also be used to provide lactam product 101 under similar conditions.
- R" methyl bromoacetate
- Scheme 16 illustrates methodology for synthesis of piperazinone intermediates, such as 133 and 135.
- Alkylation of an ⁇ -aminoester with bromide 129 may be used to provide the aminoketone 130.
- a variety of bases, including NaHCO 3 , K 2 CO 3 , and Na 3 PO 4 may be utilized in this alkylation reaction.
- the aminoketone product may be subjected to reductive amination with ammonium acetate to give the corresponding diamine, which usually undergoes cyclization in situ to provide piperazinone 131.
- the ester although not limited to methyl ester, may then be hydrolyzed by an appropriate base, such as NaOH, to provide acid 137.
- Acid 137 may then be coupled to aniline 138 using a variety of peptide coupling reagent combinations, such as EDCI and HOAt, in the presence of an appropriate base, such as triethylamine, in an appropriate solvent, such as DMF, to yield compound 139 .
- an appropriate base such as triethylamine
- an appropriate solvent such as DMF
- the lactam intermediates such as 1, may be obtained from commercial sources or prepared according to Scheme 2, such as 11. While the methodology shown in Scheme 2 is exemplified using anhydride 5, it is understood that it may be applied to a variety of substrates, such as those described herein, in order to provide various lactam intermediates.
- Scheme 18 illustrates the synthesis of key intermediates that are analogous to those in Scheme 2 but of a more general structure.
- Anhydrides of general structure 140 can be converted to a number of bis-esters analogous to 141, using an appropriate alcohol, such as MeOH, and an acid catalyst, such as HCl. Selective deprotection of one ester may be accomplished using a mild base, such as K 2 CO 3 , in an appropriate solvent mixture, to provide acid 142. Acid 142, may then be coupled with an appropriate amine, such as methylmethoxyamine or pyrrolidine, using an appropriate amide-forming reagent combination, such as oxalyl chloride and catalytic DMF, to provide an amide, such as 143, in preparation for ketone formation.
- an appropriate alcohol such as MeOH
- an acid catalyst such as HCl.
- Selective deprotection of one ester may be accomplished using a mild base, such as K 2 CO 3 , in an appropriate solvent mixture, to provide acid 142.
- Acid 142 may then be coupled with an appropriate amine, such as methylmethoxyamine or pyrrolidine, using an appropriate
- a variety of organometallic reagents such as BrMgR 11 may then be allowed to react with 143, to provide ketone 144 .
- Conversion of ketone 144 to sulfinamide 145 may be achieved using a variety of known methodology, [see, for example Ellman et al. (1987) Tetrahedron Lett. 40, 6709-6712 ].
- Conversion of compound 145 into key lactam intermediate 146 may be achieved using an appropriate acid, such as HCl, followed by an appropriate base, such as triethylamine to allow spontaneous lactamization in an appropriate solvent, such as MeOH, at a temperature ranging from 0-150 °C.
- Ketone 144 (Scheme 18) may be converted to the sulfinimine 160 utilizing an appropriate dehydration reagent, such as titanium tetraethoxide, in a solvent, such at THF, in a temperature range of 20-150 °C.
- an appropriate organometallic reagent such as R 10 MgBr
- an aprotic solvent such as DCM
- Conversion of compound 161 into key lactam intermediate 162 may be achieved using an appropriate acid, such as HCl, followed by an appropriate base, such as triethylamine to allow lactamization in an appropriate solvent, such as MeOH or toluene, at a temperature ranging from 0-150 °C.
- Lactam of general structure 162 may be alkylated with an electrophile of general formula BrH 2 CCO 2 Me after deprotonation with a suitable base, such as potassium hydride, in an appropriate solvent, such as THF to provide 163.
- Dealkylation of the methyl ester in 163 may be effected using KOTMS in an appropriate solvent, such as THF, in a temperature range of 0 to 40 °C, to provide the potassium carboxylate 164 .
- an appropriate solvent such as THF
- hydrolysis may be achieved under a variety of basic or acidic conditions.
- Compound 164 may be elaborated in an analogous manner as compound 2 to provide claimed compounds.
- aldehyde 181 it may be advantageous or necessary to access aldehyde 181 via a two-step procedure in which the ester is reduced to the corresponding alcohol and the alcohol is oxidized to afford aldehyde 181 using, for example, Swern conditions.
- Reductive amination of 181 with a suitable amino ester under standard conditions can be used to provide the amine 182, and this may be deprotected and cyclized under acidic conditions to give piperazinone 183 .
- Piperazinone 183 may be further elaborated in analogy with Scheme 16 to give key acid intermediates like 186 .
- Activation of the amine B may also be a useful strategy in cases where the amine is relatively unreactive.
- various protecting group strategies familiar to one skilled in the art of organic synthesis may be employed to allow preparation of a particular compound of the present invention.
- the final product may be further modified, for example, by manipulation of substituents.
- manipulations may include, but are not limited to, reduction, oxidation, alkylation, acylation, and hydrolysis reactions which are commonly known to those skilled in the art.
- Step E Methyl (5 S )-5- ⁇ [( S )- tert -butylsulfinyl]amino ⁇ -5-(3,5-difluorophenyl)-2,2-dimethylpentanoate
- Step D Benzyl 5- ⁇ [( S )- tert -butylsulfinyl]imino ⁇ -2,2-dimethylpentanoate
- Step B 3,3-Dibromo-1- ⁇ [2-(trimethylsilyl)ethoxy]methyl ⁇ -1,3-dihydro-2 H -pyrrolo[2,3-b]pyridin-2-one
- Step C 1- ⁇ [2-(Trimethylsilyl)ethoxy]methyl ⁇ -1,3-dihydro-2 H -pyrrolo[2,3-b]pyridin-2-one
- Step A ( ⁇ )-5-Nitro-1'- ⁇ [2-(trimethylsilyl)ethoxy]methyl ⁇ -1,3-dihydrospiro[indene-2,3'-pyrrolo[2,3-b]pyridin]-2'(1' H )-one
- Step B ( ⁇ )-5-Amino-1'- ⁇ [2-(trimethylsilyl)ethoxy]methyl ⁇ -1,3-dihydrospiro[indene-2,3'-pyrrolo[2,3- b ]pyridin]-2'(1' H )-one
- Step C tert -Butyl ( R )-(2'-oxo-1'- ⁇ [2-(trimethylsilyl)ethoxy]methyl ⁇ -1,1',2',3-tetrahydrospiro[indene-2,3'-pyrrolo[2,3-b]pyridin]-5-yl)carbamate
- the enantiomers were resolved by HPLC, utilizing a ChiralPak AD column and eluting with EtOH.
- the first major peak to elute was tert -butyl ( S )-(2'-oxo-1'- ⁇ [2-(trimethylsilyl)ethoxy]methyl ⁇ -1,1',2',3-tetrahydrospiro[indene-2,3'-pyrrolo[2,3- b ]pyridin]-5-yl)carbamate
- the second major peak to elute was tert -butyl ( R )-(2'-oxo-1'- ⁇ [2-(trimethylsilyl)ethoxy]methyl ⁇ -1,1',2',3-tetrahydrospiro[indene-2,3'-pyrrolo[2,3- b ]pyridin]-5-yl)carbamate, the title compound.
- MS: m / z 482 (M + 1).
- Step D ( R )-5-Amino-1,3-dihydrospiro[indene-2,3'-pyrrolo[2,3-b]pyridin]-2'(1' H )-one
- Step B ( ⁇ )-Ethyl [6-(3,5-difluorophenyl)-3,3-dimethyl-2-oxopiperazin-1-yl]acetate
- Step C tert -Butyl (5 R )-5-(3,5-difluorophenyl)-4-(2-ethoxy-2-oxoethyl)-2,2-dimethyl-3-oxopiperazine-1-carboxylate
- the crude product was purified by silica gel chromatography, eluting with a gradient of hexane:EtOAc - 100:0 to 0:100, to give the racemic product.
- the enantiomers were separated by SFC, using a Chiralcel OD column and eluting with CO 2 :MeOH - 80:20.
- the first major peak to elute was tert -butyl (5 S )-5-(3,5-difluorophenyl)-4-(2-ethoxy-2-oxoethyl)-2,2-dimethyl-3-oxopiperazine-1-carboxylate and the second major peak to elute was tert-butyl (5 R )-5-(3,5-difluorophenyl)-4-(2-ethoxy-2-oxoethyl)-2,2-dimethyl-3-oxopiperazine-1-carboxylate, the title compound.
- MS: m / z 371 (M - C 4 H 7 ).
- Step D Lithium [(6 R )-4-( tert -butoxycarbonyl)-6-(3,5-difluorophenyl)-3,3-dimethyl-2-oxopiperazin-1-yl]acetate
- Step B ( S )-[6-(3-Fluoro-4-methylphenyl)-3,3-dimethyl-2-oxopiperidin-1-yl]acetic acid
- the crude product was purified directly by HPLC using a reversed phase C18 column and eluting with a gradient of H 2 O:CH 3 CN:CF 3 CO 2 H - 90:10:0.1 to 5:95:0.1.
- the compound was further purified by SFC, utilizing a ChiralPak AD column and eluting with CO 2 :MeOH -91:9, to give the title compound.
- MS: m / z 294 (M + 1).
- Step B ⁇ ( ⁇ )-3,3-Dimethyl-2-oxo-6-[3-(trifluoromethyl)phenyl]piperidin-1-yl ⁇ acetic acid
- Step B Methyl [(3 R )-3,4-dimethyl-2-oxo-6-phenylpiperazin-1-yl]acetate
- Step C Lithium [(3R)-3,4-dimethyl-2-oxo-6-phenylpiperazin-1-yl]acetate
- Step B Methyl 1- ⁇ [2-(3,5-difluorophenyl)-2-oxoethyl]amino ⁇ cyclopentanecarboxylate
- Step C Ethyl [(8 R )-8-(3,5-difluorophenyl)-10-oxo-6,9-diazaspiro[4.5]dec-9-yl]acetate
- the reaction mixture was heated to 60 °C for 72 h then allowed to cool.
- the reaction mixture was quenched with saturated aqueous NaHCO 3 and then extracted with EtOAc (3 ⁇ 50 mL).
- the combined organic extracts were dried over Na 2 SO 4 , filtered, and concentrated in vacuo.
- the crude product was purified by HPLC using a reversed phase C 18 column and eluting with a gradient of H 2 O:CH 3 CN:CF 3 CO 2 H - 90:10:0.1 to 5:95:0.1.
- the product-containing fractions were combined, basified with saturated aqueous NaHCO 3 , and extracted with EtOAc.
- the organic extracts were dried over Na 2 SO 4 , filtered, and concentrated in vacuo to give the racemic product.
- the enantiomers were separated by SFC, using a ChiralPak AD column and eluting with CO 2 :MeOH - 90:10.
- the first major peak to elute was ethyl [(8 S )-8-(3,5-difluorophenyl)-10-oxo-6,9-diazaspiro[4.5]dec-9-yl]acetate
- the second major peak to elute was ethyl [(8 R )-8-(3,5-difluorophenyl)-10-oxo-6,9-diazaspiro[4.5]dec-9-yl]acetate, the title compound.
- MS: m / z 353 (M + 1).
- Step D Lithium [(8 R )-8-(3,5-difluorophenyl)-10-oxo-6,9-diazaspiro[4.5]dec-9-yl]acetate
- Step A Ethyl (5 S )-5- ⁇ [( S )-tert-butylsulfinyl]amino ⁇ -5-(3,5-difluorophenyl)pentanoate
- Step B (6 S )-6-(3,5-Difluorophenyl)-piperidin-2-one
- Step C tert -Butyl (2 S )-2-(3,5-difluorophenyl)-6-oxopiperidine-1-carboxylate
- Step D tert -Butyl (6 S )-6-(3,5-difluorophenyl)-3,3-diethylpiperidin-2-one-1-carboxylate
- Step F [(6 S )-6-(3,5-Difluorophenyl)-3,3-diethyl-2-oxopiperidin-1-yl]acetic acid
- Step B Ethyl [(8 R) -10-oxo-8-phenyl-6,9-diazaspiro[4.5]dec-9-yl]acetate
- the combined organic extracts were dried over Na 2 SO 4 , filtered, and concentrated in vacuo.
- the crude product was purified by HPLC using a reversed phase C 18 column and eluting with a gradient of H 2 O:CH 3 CN:CF 3 CO 2 H - 90:10:0.1 to 5:95:0.1.
- the product-containing fractions were combined, basified with saturated aqueous NaHCO 3 , and extracted with EtOAc.
- the organic extracts were dried over Na 2 SO 4 , filtered, and concentrated in vacuo to give the racemic product.
- the enantiomers were separated by HPLC, using a Chiralcel OD column and eluting with hexane:EtOH - 60:40.
- the first major peak to elute was ethyl [(8 S )-10-oxo-8-phenyl-6,9-diazaspiro[4.5]dec-9-yl]acetate and the second major peak to elute was ethyl [(8 R )-10-oxo-8-phenyl-6,9-diazaspiro[4.5]dec-9-yl]acetate, the title compound.
- MS: m / z 317 (M + 1).
- Step C [(8 R )-10-Oxo-8-phenyl-6,9-diazaspiro[4.5]dec-9-yl]acetic acid hydrochloride
- Step B Methyl 2- ⁇ [2-(3,5-difluorophenyl)-2-oxoethyl]amino ⁇ -2-ethylbutanoate
- Step C Methyl [(6 R )-6-(3,5-difluorophenyl)-3,3-diethyl-2-oxopiperazin-1-yl]acetate
- the stirred reaction mixture was heated at 50 °C for 18 h, then at 70 °C for 24 h, and allowed to cool.
- the mixture was quenched with saturated aqueous NaHCO 3 and then extracted with EtOAc (2 ⁇ 30 mL).
- the combined organic extracts were dried over Na 2 SO 4 , filtered, and concentrated in vacuo .
- the crude product was purified by HPLC using a reversed phase C 18 column and eluting with a gradient of H 2 O:CH 3 CN:CF 3 CO 2 H - 90:10:0.1 to 5:95:0.1.
- the product-containing fractions were combined, basified with saturated aqueous NaHCO 3 , and extracted with EtOAc.
- Step D [(6 R )-6-(3,5-Difluorophenyl)-3,3-diethyl-2-oxopiperazin-1-yl]acetic acid hydrochloride
- Step A [(8 R )-8-(2-Bromo-3,5-difluorophenyl)-10-oxo-6,9-diazaspiro[4.5]dec-9-yl]acetic acid
- Step B Ethyl [8-(3,5-difluorophenyl)-10-oxo-6,9-diazaspiro[4.5]dec-9-yl]acetate
- Step C tert -Butyl (8 R )-8-(3,5-difluorophenyl)-9-(2-ethoxy-2-oxoethyl)-10-oxo-6,9-diazaspiro[4.5]decane-6-carboxylate
- the crude product was purified by silica gel chromatography, eluting with a gradient of hexane:EtOAc - 95:5 to 50:50, to give the racemic product.
- the enantiomers were separated by HPLC, using a Chiralcel OD column and eluting with hexane: i -PrOH:Et 2 NH - 60:40:0.1.
- the first major peak to elute was tert -butyl (8 S )-8-(3,5-difluorophenyl)-9-(2-ethoxy-2-oxoethyl)-10-oxo-6,9-diazaspiro[4.5]decane-6-carboxylate and the second major peak to elute was tert -butyl (8 R )-8-(3,5-difluorophenyl)-9-(2-ethoxy-2-oxoethyl)-10-oxo-6,9-diazaspiro[4.5]decane-6-carboxylate, the title compound.
- MS: m / z 397 (M - C 4 H 7 ).
- Step D Lithium [(8 R )-6-( tert -butoxycarbonyl)-8-(3,5-difluorophenyl)-10-oxo-6,9-diazaspiro[4.5]dec-9-yl]acetate
- Step A Di- tert -butyl [1-(3,5-difluorophenyl)ethyl]imidodicarbonate
- Step B tert -Butyl 2-[( tert -butoxycarbonyl)amino]-2-(3,5-difluorophenyl)propanoate
- Step C tert -Butyl [1-(3,5-difluorophenyl)-1-methyl-2-oxoethyl]carbamate
- Step E Methyl 1- ⁇ [2-[( tert -butoxycarbonyl)amino]-2-(3,5-difluorophenyl)propyl]amino ⁇ cyclohexanecarboxylate
- Step F Methyl 1- ⁇ [2-amino-2-(3,5-difluorophenyl)propyl]amino ⁇ cyclohexanecarboxylate
- Step G (3 R )-3-(3,5-Difluorophenyl)-3-methyl-1,4-diazaspiro[5.5]undccan-5-one
- the crude product was purified by silica gel chromatography, eluting with a gradient of EtOAc:MeOH - 100:0 to 92:8, to give the racemic product.
- the enantiomers were separated by HPLC, using a ChiralPak AD column and eluting with hexane:EtOH:Et 2 NH - 40:60:0.1.
- the first major peak to elute was (3 R )-3-(3,5-difluorophenyl)-3-methyl-1,4-diazaspiro[5.5]undecan-5-one, the title compound, and the second major peak to elute was (3 S )-3-(3,5-difluorophenyl)-3-methyl-1,4-diazaspiro[5.5]undecan-5-one.
- MS: m / z 295 (M + 1).
- Step H tert -Butyl (3 R )-3-(3,5-difluorophenyl)-3-methyl-5-oxo-1,4-diazaspiro[5.5]undecane-1-carboxylate
- Step I tert -Butyl (3 R )-3-(3,5-difluorophenyl)-4-(2-ethoxy-2-oxoethyl)-3-methyl-5-oxo-1,4-diazaspiro[5.5]undecane-1- carboxylate
- Step J Lithium [(3 R )-1-( tert -butoxycarbonyl)-3-(3,5-difluorophenyl)-3-methyl-5-oxo-1,4-diazaspiro[5.5]undec-4-yl]acetate
- Step B ( ⁇ )-3-Nitro-1'- ⁇ [2-(trimethylsilyl)ethoxy]methyl ⁇ -5,7-dihydrospiro[cyclopenta[ b ]pyridine-6,3'-pyrrolo[2,3- b ]pyridin]-2'(1' H )-one
- Step C ( ⁇ )-3-Amino-1'- ⁇ [2-(trimethylsilyl)ethoxy]methyl ⁇ -5,7-dihydrospiro[cyclopenta[ b ]pyridine-6,3-pyrrolo[2,3- b ]pyridin]-2'(1' H )-one
- Step D 3-Amino-5,7-dihydrospiro[cyclopenta[ b ]pyridine-6,3'-pyrrolo[2,3- b ]pyridinl-2'(1' H )-one, isomer A
- reaction mixture was purified directly by HPLC using a reversed phase C 18 column and eluting with a gradient of H 2 O:CH 3 CN:CF 3 CO 2 H - 90:10:0.1 to 5:95:0.1. Lyophilization provided the racemic title compound as the TFA salt.
- the enantiomers were resolved by HPLC, utilizing a ChiralPak AD column and eluting with EtOH.
- the first major peak to elute was 3-amino-5,7-dihydrospiro[cyclopenta[ b ]pyridine-6,3'-pyrrolo[2,3- b ]pyridin]-2'(1' H )-one, isomer A, the title compound, and the second major peak to elute was 3-amino-5,7-dihydrospiro[cyclopenta[ b ]pyridine-6,3'-pyrrolo[2,3- b ]pyridin]-2'(1' H )-one, isomer B.
- MS: m / z 253 (M + 1).
- Step D ( ⁇ )-Sodium 2'-oxo-1',2',5,7-tetrahydrospiro[cyclopenta[ c ]pyridine-6,3'-pyrrolo[2,3- b ]pyridine]-3-carboxylate
- Step E ( ⁇ )- tert -Butyl(2'-oxo-1',2',5,7-tetrahydrospiro[cyclopenta[c]pyridine-6,3-pyrrolo[2,3- b ]pyridin]-3-yl)carbamate
- Step F 3-Amino-5,7-dihydrospiro[cyclopenta[ c ]pyridine-6,3'-pyrrolo[2,3- b ]pyridin]-2'(1' H )-one, isomer A
- the first major peak to elute was 3-amino-5,7-dihydrospiro[cyclopenta[ c ]pyridine-6,3'-pyrrolo[2,3- b ]pyridin]-2'(1' H )-one, isomer A, the title compound, and the second major peak to elute was 3-amino-5,7-dihydrospiro[cyclopenta[ c ]pyridine-6,3'-pyrrolo[2,3- b ]pyridin]-2'(1' H )-one, isomer B.
- MS: m / z 253 (M + 1).
- Step D ( ⁇ )-2'-Oxo-1'- ⁇ [2-(trimethylsilyl)ethoxy]methyl ⁇ -1',2',5,7-tetrahydrospiro[cyclopenta[ b ]pyridine-6,3'-pyrrolo[2,3-b]pyridine]-2-carbonitrile
- Step E ( ⁇ )-2'-Oxo-1',2',5,7-tetrahydrospiro[cyclopenta[ b ]pyridine-6,3'-pyrrolo[2,3- b ]pyridine]-2-carboxylic acid
- Step F ( ⁇ )- tert -Butyl (2'-oxo-1',2',5,7-tetrahydrospiro[cyclopenta[ b ]pyridine-6,3'-pyrrolo[2,3- b ] pyridin]-2-yl)carbamate
- Step G ( ⁇ )-2-Amino-5,7-dihydrospiro[cyclopenta[ b ]pyridine-6,3'-pyrrolo[2,3- b ]pyridin]-2'(1' H )-one
- Step A 1-Benzyl 2-methyl (2 R )-2-(3-oxopropyl)pyrrolidine-1,2-dicarboxylate
- Step B 1-Benzyl 2-methyl (2 R )-2-((3 E )-3- ⁇ [( S )-tert-butylsulfinyl]imino ⁇ propyl)pyrrolidine-1,2-dicarboxylate
- Step C 1-Benzyl 2-methyl (2 R )-2-[(3 S )-3- ⁇ [(S)-tert-butylsulfonyl]amino ⁇ -3-(3,5-difluorophenyl)propyl]pyrrolidine-1,2-dicarboxylate
- Step D Benzyl (5 R ,8 S )-8-(3,5-difluorophenyl)-6-oxo-1,7-diazaspiro[4.5]decane-1-carboxylate
- Step E [(5 R ,8 S )-1-[(Benzyloxy)carbonyl]-8-(3,5-difluorophenyl)-6-oxo-1,7-diazaspiro[4,5]dec-7-yl]acetic acid
- Step B N -[(4 S )-3-Methyl-2,5-dioxo-1',3'-dihydrospiro[imidazolidine-4,2'-inden]-5'-yl-2-((6 ⁇ )-2-oxo-6-phenylpiperidin-1-yl)acetamide
- Step A 2-[(6 S )-(3,5-Difluorophenyl)-3,3-dimethyl-2-oxopiperidin-1-yl]- N -((2 R )-2'-oxo-1'- ⁇ [2-(trimethylsilyl)ethoxy]methyl ⁇ -1,1',2',3-tetrahydrospiro[indene-2,3'-pyrrolo[2,3-b]pyridin]-5-yl]acetamide
- Step B 2-[(6 S )-6-(3,5-Difluorophenyl)-3,3-dimethyl-2-oxopiperidin-1-yl]- N -methyl- N -((2 R )-2'-oxo-1'- ⁇ [2-(trimethylsilyl)ethoxy]methyl ⁇ -1,1',2',3-tetrahydrospiro[indene-2,3'-pyrrolo[2,3-b]pyridin]-5-yl]acetamide
- Step C 2-[(6 S )-6-(3,5-Difluorophenyl)-3,3-dimethyl-2-oxopiperidin-1-yl]- N -methyl- N -[(2 R )-2'-oxo-1,1',2',3-tetrahydrospiro[indene-2,3'-pyrrolo[2,3-b]pyridin]-5-yl]acetamide
- reaction mixture was purified directly by HPLC using a reversed phase C18 column and eluting with a gradient of H 2 O:CH 3 CN:CF 3 CO 2 H - 90:10:0.1 to 5:95:0.1.
- the pure, product-containing fractions were combined, basified with saturated aqueous NaHCO 3 , and extracted with EtOAc. The organic extracts were dried over Na 2 SO 4 , filtered, and concentrated in vacuo to give the title compound.
- reaction mixture was purified directly by HPLC using a reversed phase C 18 column and eluting with a gradient of H 2 O:CH 3 CN:CF 3 CO 2 H - 90:10:0.1 to 5:95:0.1. Lyophilization provided the title compound.
- MS: m / z 547 (M + 1).
- the reaction mixture was diluted with EtOAc (5 mL) and washed successively with 10% citric acid (2 mL), H 2 O (2 mL), saturated aqueous NaHCO 3 (2 mL), and brine (2 mL).
- the organic layer was dried over Na 2 SO 4 , filtered, and concentrated in vacuo.
- the residue was purified by silica gel chromatography, eluting with CH 2 Cl 2 :EtOAc - 100:0 to 0:100, to give the Boc-protected product.
- the Boc-protected product was dissolved in EtOAc (3 mL), the solution was cooled to 0 °C, and HCl (g) was bubbled in for 1 min.
- the reaction mixture was diluted with EtOAc (500 mL) and washed successively with 10% citric acid (100 mL), H 2 O (100 mL), saturated aqueous NaHCO 3 (100 mL), and brine (100 mL).
- the organic layer was dried over Na 2 SO 4 , filtered, and concentrated in vacuo.
- the residue was purified by silica gel chromatography, eluting with CH 2 Cl 2 :MeOH - 100:0 to 90:10, to give the Boc-protected product.
- the Boc-protected product was dissolved in EtOAc (75 mL), the solution was cooled to 0 °C, and HCl (g) was bubbled in for 2 min.
- reaction mixture was purified directly by HPLC using a reversed phase C18 column and eluting with a gradient of H 2 O:CH 3 CN:CF 3 CO 2 H - 90:10:0.1 to 5:95:0.1. Lyophilization provided the title compound as the TFA salt.
- MS: m / z 558 (M + 1).
- reaction mixture was purified directly by HPLC using a reversed phase C18 column and eluting with a gradient of H 2 O:CH 3 CN:CF 3 CO 2 H - 90:10:0.1 to 5:95:0.1. Lyophilization provided the title compound as the TFA salt.
- MS: m/ z 558 (M + 1).
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Public Health (AREA)
- Animal Behavior & Ethology (AREA)
- Veterinary Medicine (AREA)
- General Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Neurosurgery (AREA)
- Neurology (AREA)
- Biomedical Technology (AREA)
- Pain & Pain Management (AREA)
- Pulmonology (AREA)
- Diabetes (AREA)
- Dermatology (AREA)
- Rheumatology (AREA)
- Cardiology (AREA)
- Endocrinology (AREA)
- Addiction (AREA)
- Immunology (AREA)
- Heart & Thoracic Surgery (AREA)
- Oncology (AREA)
- Physical Education & Sports Medicine (AREA)
- Psychiatry (AREA)
- Communicable Diseases (AREA)
- Hematology (AREA)
- Obesity (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Emergency Medicine (AREA)
- Reproductive Health (AREA)
- Urology & Nephrology (AREA)
- Epidemiology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Claims (18)
- Composé de la formule I:
et des sels pharmaceutiquement acceptables de celui-ci et des énantiomères et des diastéréoisomères individuels de celui-ci. - Composé selon la revendication 1, dans lequel A1 est -NH-.
- Composé selon la revendication 1, dans lequel A1 est -CH2-.
- Composé selon la revendication 1, dans lequel Ea est -C(R5a)=.
- Composé selon la revendication 1, dans lequel Eb est -C(R5b)= ou -N=.
- Composé selon la revendication 1, dans lequel Ec est -C(R5c)=.
- Composé selon l'une quelconque des revendications 1, 4, 5 et 6, dans lequel R5a, R5b et R5c sont un hydrogène.
- Composé selon la revendication 1, dans lequel R6 et R7 sont un méthyle.
- Composé selon la revendication 1, dans lequel R6 et R7 sont un éthyle.
- Composé selon la revendication 1, dans lequel R6 et R7 accompagnés de l'atome de carbone auquel ils sont attachés se joignent pour former un cycle choisi parmi un cyclopropyle, un cyclobutyle, un cyclopentyle, un cyclohexyle, un cycloheptyle, un tétrahydropyranyle, un pyrrolidinyle et un pipéridinyle, lequel cycle est non substitué ou substitué avec 1-6 substituants chacun indépendamment choisis parmi:(1) un -C1-6 alkyle, qui est non substitué ou substitué avec 1-3 halo,(2) un phényle, dans lequel le phényle est optionnellement condensé au cycle et lequel phényle est non substitué ou substitué avec 1-3 substituants chacun indépendamment choisis parmi: un halo, -ORa et un -C1-4 alkyle, qui est non substitué ou substitué avec 1-3 fluoro, et(3) un halo.
- Composé selon la revendication 1, dans lequel R8 est choisi parmi: un hydrogène et un -C1-6 alkyle, qui est non substitué ou substitué avec 1-5 fluoro.
- Composé selon la revendication 1, dans lequel R10 est un hydrogène.
- Composé selon la revendication 1, dans lequel R11 est un phényle qui est non substitué ou substitué avec 1-5 halo.
- Composition pharmaceutique qui comprend un support inerte et un composé selon l'une quelconque des revendications précédentes.
- Composé selon l'une quelconque des revendications 1-15 pour une utilisation dans le traitement, le contrôle, l'amélioration ou la réduction du risque de céphalée, de migraine ou de céphalée vasculaire de Horton chez un patient mammifère en ayant besoin qui comprend l'administration au patient d'une quantité thérapeutiquement efficace du composé selon la revendication 1.
- Composé pour une utilisation selon la revendication 17 pour une utilisation en combinaison avec une quantité thérapeutiquement efficace d'un deuxième agent choisi parmi des agonistes de la sérotonine, des analgésiques, des agents anti-inflammatoires, des antihypertenseurs et des anticonvulsifs.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US79907106P | 2006-05-09 | 2006-05-09 | |
| PCT/US2007/010953 WO2008020902A1 (fr) | 2006-05-09 | 2007-05-04 | Antagonistes spirocycliques substitués des récepteurs cgrp |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP2029575A1 EP2029575A1 (fr) | 2009-03-04 |
| EP2029575B1 true EP2029575B1 (fr) | 2014-10-22 |
Family
ID=38556368
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP07835747.2A Active EP2029575B1 (fr) | 2006-05-09 | 2007-05-04 | Antagonistes spirocycliques substitués des récepteurs cgrp |
| EP07776804.2A Active EP2024358B1 (fr) | 2006-05-09 | 2007-05-04 | Antagonistes de récepteurs cgrp monocycliques substitués |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP07776804.2A Active EP2024358B1 (fr) | 2006-05-09 | 2007-05-04 | Antagonistes de récepteurs cgrp monocycliques substitués |
Country Status (23)
| Country | Link |
|---|---|
| US (3) | US7629338B2 (fr) |
| EP (2) | EP2029575B1 (fr) |
| JP (2) | JP4435288B2 (fr) |
| KR (1) | KR20090018107A (fr) |
| CN (1) | CN101443323A (fr) |
| AR (1) | AR060872A1 (fr) |
| AU (1) | AU2007284994B2 (fr) |
| BR (1) | BRPI0711352A2 (fr) |
| CA (1) | CA2650932C (fr) |
| CR (1) | CR10415A (fr) |
| DO (1) | DOP2007000093A (fr) |
| EA (1) | EA200870521A1 (fr) |
| EC (1) | ECSP088872A (fr) |
| GE (1) | GEP20115140B (fr) |
| HN (1) | HN2008001667A (fr) |
| IL (1) | IL195110A0 (fr) |
| MA (1) | MA30558B1 (fr) |
| MX (1) | MX2008014353A (fr) |
| NO (1) | NO20085110L (fr) |
| PE (1) | PE20080253A1 (fr) |
| TW (1) | TW200813008A (fr) |
| WO (2) | WO2008020902A1 (fr) |
| ZA (1) | ZA200809275B (fr) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2017001433A1 (fr) | 2015-06-29 | 2017-01-05 | Galderma Research & Development | Composés d'antagoniste de récepteur cgrp pour le traitement topique de troubles cutanés |
Families Citing this family (54)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CA2580020A1 (fr) * | 2004-09-09 | 2006-03-23 | Ian M. Bell | Antagonistes des recepteurs cgrp d'aryle-spirolactame |
| AU2005285109B2 (en) * | 2004-09-13 | 2011-03-03 | Merck Sharp & Dohme Corp. | Bicyclic anilide spirolactam CGRP receptor antagonists |
| WO2008020902A1 (fr) * | 2006-05-09 | 2008-02-21 | Merck & Co., Inc. | Antagonistes spirocycliques substitués des récepteurs cgrp |
| EP2091533B1 (fr) | 2006-12-08 | 2012-11-28 | Merck Sharp & Dohme Corp. | Composés spirocycliques contraints servant d'antagonistes des récepteurs du cgrp |
| WO2008112159A2 (fr) | 2007-03-12 | 2008-09-18 | Merck & Co., Inc. | Antagonistes des récepteurs cgrp d'anilide spirolactame monocyclique |
| GB0706914D0 (en) | 2007-04-10 | 2007-05-16 | Isis Innovation | Novel adenovirus vectors |
| US8372859B2 (en) | 2007-04-11 | 2013-02-12 | Merck, Sharp & Dohme Corp. | CGRP receptor antagonists with tertiary amide, sulfonamide, carbamate and urea end groups |
| US8173655B2 (en) | 2007-04-16 | 2012-05-08 | Merck, Sharp & Dohme Corp. | Bicyclic anilide heterocyclic CGRP receptor antagonists |
| WO2008130524A1 (fr) * | 2007-04-16 | 2008-10-30 | Merck & Co., Inc. | Antagonistes des récepteurs du cgrp aryl hétérocycliques |
| WO2008153849A1 (fr) * | 2007-06-05 | 2008-12-18 | Merck & Co., Inc. | Antagonistes des récepteurs cgrp à base de carboxamide hétérocyclique |
| JP2010529120A (ja) | 2007-06-07 | 2010-08-26 | メルク・シャープ・エンド・ドーム・コーポレイション | 三環式アニリド複素環cgrp受容体アンタゴニスト |
| EP2065381A1 (fr) | 2007-10-18 | 2009-06-03 | Boehringer Ingelheim Pharma GmbH & Co. KG | Antagonistes du CGRP |
| MX2010003849A (es) | 2007-10-18 | 2010-04-27 | Boehringer Ingelheim Int | Antagonistas de cgrp. |
| US8829006B2 (en) | 2007-11-22 | 2014-09-09 | Boehringer Ingelheim International Gmbh | Compounds |
| CA2711367A1 (fr) * | 2008-02-19 | 2009-08-27 | Merck Sharp & Dohme Corp. | Antagonistes de recepteurs de cgrp de type imidazobenzazepine |
| JP2011515480A (ja) * | 2008-03-28 | 2011-05-19 | メルク・シャープ・エンド・ドーム・コーポレイション | 単環式cgrp受容体アンタゴニスト |
| JP2012511500A (ja) * | 2008-08-20 | 2012-05-24 | メルク・シャープ・エンド・ドーム・コーポレイション | Cgrp受容体アンタゴニストとしての分岐末端を有する非アミドリンカー |
| CA2735821A1 (fr) * | 2008-09-08 | 2010-03-11 | Merck Sharp & Dohme Corp. | Antagonistes du recepteur cgrp d'amide monocyclique |
| CA2737711A1 (fr) * | 2008-09-18 | 2010-03-25 | Merck Sharp & Dohme Corp. | Antagonistes des recepteurs du cgrp a base de dihydroimidazolone bicyclique |
| JP2012504625A (ja) * | 2008-10-03 | 2012-02-23 | メルク・シャープ・エンド・ドーム・コーポレイション | Cgrp受容体アンタゴニスト |
| AU2009302712A1 (en) * | 2008-10-10 | 2010-04-15 | Merck Sharp & Dohme Corp. | CGRP receptor antagonists |
| WO2010077752A1 (fr) | 2008-12-17 | 2010-07-08 | Merck Sharp & Dohme Corp. | Dérivés d'imidazolinone en tant qu'antagonistes de récepteurs cgrp |
| US8765763B2 (en) | 2009-06-05 | 2014-07-01 | Boehringer Ingelheim International Gmbh | Substituted piperazines as CGRP antagonists |
| WO2011005731A2 (fr) | 2009-07-08 | 2011-01-13 | Merck Sharp & Dohme Corp. | Procédé de production d'antagoniste du récepteur cgrp |
| EP2458991A4 (fr) * | 2009-07-27 | 2012-12-12 | Merck Sharp & Dohme | Antagonistes radio-marqués du cgrp |
| CA2798831A1 (fr) | 2010-05-11 | 2011-11-17 | Pfizer Inc. | Composes morpholiniques convenant comme antagonistes de recepteurs de mineralcorticoides |
| CN103096893B (zh) | 2010-06-04 | 2016-05-04 | 阿尔巴尼分子研究公司 | 甘氨酸转运体-1抑制剂、其制备方法及其用途 |
| TWI487706B (zh) | 2010-11-12 | 2015-06-11 | Merck Sharp & Dohme | 六氫吡啶酮甲醯胺氮雜茚滿cgrp受體拮抗劑 |
| EP2654421B1 (fr) | 2010-12-22 | 2016-10-19 | Merck Sharp & Dohme Corp. | Indanecarboxamides condensés comme antagonistes des récepteurs du cgrp |
| AU2012231293A1 (en) * | 2011-03-18 | 2013-08-29 | Merck Sharp & Dohme Corp. | Piperidine carboxamide spirohydantoin CGRP receptor antagonists |
| EP2685826B1 (fr) * | 2011-03-18 | 2016-02-03 | Merck Sharp & Dohme Corp. | Pipéridinone carboxamide spirohydantoïne, antagonistes du récepteur cgrp |
| KR20220164616A (ko) * | 2012-03-14 | 2022-12-13 | 머크 샤프 앤드 돔 코포레이션 | Cgrp 수용체 길항제의 제조 방법 |
| US9487523B2 (en) | 2012-03-14 | 2016-11-08 | Merck Sharp & Dohme Corp. | Process for making CGRP receptor antagonists |
| WO2013169567A1 (fr) | 2012-05-09 | 2013-11-14 | Merck Sharp & Dohme Corp. | Antagonistes de récepteur de cgrp à base de spirolactame |
| EP2846798B1 (fr) | 2012-05-09 | 2018-04-04 | Merck Sharp & Dohme Corp. | Antagonistes de récepteur de cgrp à base de spirolactame aliphatique |
| US9174989B2 (en) | 2012-05-09 | 2015-11-03 | Merck Sharp & Dohme Corp. | Process for making CGRP receptor antagonists |
| PE20161405A1 (es) * | 2013-12-24 | 2017-01-18 | Harvard College | Analogos de cortistatina y sintesis y usos de los mismos |
| US12168004B2 (en) | 2014-02-05 | 2024-12-17 | Merck Sharp & Dohme Llc | Treatment of migraine |
| KR102448369B1 (ko) | 2014-02-05 | 2022-09-28 | 머크 샤프 앤드 돔 엘엘씨 | Cgrp-활성 화합물에 대한 정제 제제 |
| TWI799849B (zh) | 2015-04-24 | 2023-04-21 | 美商安美基公司 | 治療或預防偏頭痛之方法 |
| CN109843889B (zh) | 2016-08-01 | 2022-03-15 | 阿普廷伊克斯股份有限公司 | 螺-内酰胺nmda调节剂和使用其的方法 |
| EP3366286A1 (fr) | 2017-02-22 | 2018-08-29 | Johannes Keller | Composés destinés au traitement de septicémie |
| GB201707938D0 (en) | 2017-05-17 | 2017-06-28 | Univ Sheffield | Compounds |
| AU2019207915B2 (en) | 2018-01-12 | 2025-09-04 | Amgen Inc. | PAC1 antibodies and uses thereof |
| US12090148B2 (en) | 2020-07-29 | 2024-09-17 | Allergan Pharmaceuticals International Limited | Treatment of migraine |
| US12383545B1 (en) | 2018-06-08 | 2025-08-12 | Allergan Pharmaceuticals International Limited | Treatment of migraine |
| MX2023007575A (es) | 2020-12-22 | 2023-09-21 | Allergan Pharmaceuticals Int Ltd | Tratamiento de la migraña. |
| EP4301362A1 (fr) | 2021-03-02 | 2024-01-10 | CGRP Diagnostics GmbH | Traitement et/ou réduction de l'apparition de migraine |
| US20250109188A1 (en) | 2021-08-24 | 2025-04-03 | Cgrp Diagnostics Gmbh | Preventative treatment of migraine |
| AU2022353124A1 (en) | 2021-09-27 | 2024-04-11 | Allergan Pharmaceuticals International Limited | Combination comprising atogepant for treating migraine |
| WO2023208124A1 (fr) * | 2022-04-29 | 2023-11-02 | 熙源安健医药(上海)有限公司 | Dérivé d'azaindane de pipéridine carboxamide, son procédé de préparation et son utilisation |
| WO2024134683A1 (fr) * | 2022-12-21 | 2024-06-27 | Msn Laboratories Private Limited, R&D Center | Nouvelle méthode de préparation de composés intermédiaires utilisés dans le traitement de la migraine |
| CN115947697A (zh) * | 2022-12-28 | 2023-04-11 | 苏州汉德创宏生化科技有限公司 | 一种3-氧代吗啉-4-羧酸叔丁酯的合成方法 |
| CN118930542A (zh) * | 2024-07-25 | 2024-11-12 | 电子科技大学长三角研究院(衢州) | 抗猴痘病毒和天花病毒的新型结构化合物及其组合应用 |
Family Cites Families (37)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4166452A (en) | 1976-05-03 | 1979-09-04 | Generales Constantine D J Jr | Apparatus for testing human responses to stimuli |
| US4256108A (en) | 1977-04-07 | 1981-03-17 | Alza Corporation | Microporous-semipermeable laminated osmotic system |
| US4265874A (en) | 1980-04-25 | 1981-05-05 | Alza Corporation | Method of delivering drug with aid of effervescent activity generated in environment of use |
| JP2001294574A (ja) * | 2000-04-11 | 2001-10-23 | Suntory Ltd | カルシトニン受容体作動物質 |
| EP1606286B1 (fr) | 2003-03-14 | 2011-05-11 | Merck Sharp & Dohme Corp. | Antagonistes benodiazepine spirohydantoin des recepteurs de cgrp |
| ATE516803T1 (de) | 2003-03-14 | 2011-08-15 | Merck Sharp & Dohme | Aryl-spirohydantoin-cgrp-rezeptor-antagonisten |
| CA2518852A1 (fr) * | 2003-03-14 | 2004-09-30 | Merck & Co., Inc. | Antagonistes des recepteurs du cgrp d'anilide spirohydantoine monocyclique |
| US7202251B2 (en) | 2003-03-14 | 2007-04-10 | Merck & Co., Inc. | Bicyclic anilide spirohydantoin CGRP receptor antagonists |
| JO2355B1 (en) | 2003-04-15 | 2006-12-12 | ميرك شارب اند دوم كوربوريشن | Hereditary calcitonin polypeptide receptor antagonists |
| DE602004027021D1 (de) | 2003-04-15 | 2010-06-17 | Merck Sharp & Dohme | Cgrp-rezeptorantagonisten |
| AU2004252150B2 (en) | 2003-06-26 | 2008-11-20 | Merck Sharp & Dohme Corp. | Benzodiazepine CGRP receptor antagonists |
| ATE461196T1 (de) | 2003-06-26 | 2010-04-15 | Merck Sharp & Dohme | Benzodiazepin-cgrp-rezeptor-antagonisten |
| JP2007523870A (ja) | 2003-07-15 | 2007-08-23 | メルク エンド カムパニー インコーポレーテッド | ヒドロキシピリジンcgrp受容体拮抗薬 |
| JP4705922B2 (ja) * | 2004-01-29 | 2011-06-22 | メルク・シャープ・エンド・ドーム・コーポレイション | Cgrp受容体拮抗薬 |
| CA2579221A1 (fr) | 2004-09-08 | 2006-03-16 | Merck & Co., Inc. | Antagonistes de recepteurs cgrp d'anilide spirolactame monocyclique |
| CA2579717A1 (fr) | 2004-09-09 | 2006-03-23 | Merck & Co., Inc. | Antagonistes des recepteurs cgrp sous forme de spirolactame d'anilide tricyclique |
| CA2580020A1 (fr) | 2004-09-09 | 2006-03-23 | Ian M. Bell | Antagonistes des recepteurs cgrp d'aryle-spirolactame |
| ATE518852T1 (de) | 2004-09-13 | 2011-08-15 | Merck Sharp & Dohme | Tricyclische anilid-spirohydantoin-cgrp- rezeptorantagonisten |
| AU2005285109B2 (en) | 2004-09-13 | 2011-03-03 | Merck Sharp & Dohme Corp. | Bicyclic anilide spirolactam CGRP receptor antagonists |
| US7390798B2 (en) | 2004-09-13 | 2008-06-24 | Merck & Co., Inc. | Carboxamide spirolactam CGRP receptor antagonists |
| JP2008515895A (ja) | 2004-10-07 | 2008-05-15 | メルク エンド カムパニー インコーポレーテッド | Cgrp受容体拮抗薬 |
| MX2007004393A (es) | 2004-10-13 | 2007-04-25 | Merck & Co Inc | Antagonistas del receptor a peptido relacionado con el gen calcitonina. |
| JP2008516957A (ja) * | 2004-10-14 | 2008-05-22 | メルク エンド カムパニー インコーポレーテッド | Cgrp受容体拮抗薬 |
| DE602005027230D1 (de) | 2004-10-22 | 2011-05-12 | Merck Sharp & Dohme | Cgrp-rezeptorantagonisten |
| EP1856100B1 (fr) | 2005-01-18 | 2010-05-05 | Merck Sharp & Dohme Corp. | Antagonistes de recepteurs du peptide lie au gene de la calcitonine (cgrp) |
| ATE537170T1 (de) | 2005-03-14 | 2011-12-15 | Merck Sharp & Dohme | Cgrp-rezeptorantagonisten |
| WO2007016087A2 (fr) | 2005-07-29 | 2007-02-08 | Merck & Co., Inc. | Antagonistes des récepteurs de cgrp de benzodiazépine hétérocyclique |
| AU2006316646A1 (en) | 2005-11-18 | 2007-05-31 | Merck Sharp & Dohme Corp. | Spirolactam bicyclic CGRP receptor antagonists |
| EP1951042B1 (fr) | 2005-11-18 | 2012-10-10 | Merck Sharp & Dohme Corp. | Antagonistes du récepteur cgrp spirolactame tricycliques |
| CA2629421A1 (fr) | 2005-11-18 | 2007-05-31 | Merck & Co., Inc. | Antagonistes du recepteur cgrp tricyclique de la spirohydantoine |
| WO2007061677A2 (fr) | 2005-11-18 | 2007-05-31 | Merck & Co., Inc. | Antagoniste du recepteur cgpr de spirolactam aryle |
| EP1963532A4 (fr) | 2005-12-07 | 2010-07-21 | Univ Utah Res Found | Methylation de l'adn en tant que cible pour le diagnostic et le traitement de la leucemie lymphocytaire chronique (llc) |
| WO2008020902A1 (fr) | 2006-05-09 | 2008-02-21 | Merck & Co., Inc. | Antagonistes spirocycliques substitués des récepteurs cgrp |
| CA2658573A1 (fr) | 2006-07-21 | 2008-01-24 | Vertex Pharmaceuticals Incorporated | Antagonistes des recepteurs cgrp |
| WO2008112159A2 (fr) | 2007-03-12 | 2008-09-18 | Merck & Co., Inc. | Antagonistes des récepteurs cgrp d'anilide spirolactame monocyclique |
| US8372859B2 (en) | 2007-04-11 | 2013-02-12 | Merck, Sharp & Dohme Corp. | CGRP receptor antagonists with tertiary amide, sulfonamide, carbamate and urea end groups |
| US8173655B2 (en) | 2007-04-16 | 2012-05-08 | Merck, Sharp & Dohme Corp. | Bicyclic anilide heterocyclic CGRP receptor antagonists |
-
2007
- 2007-05-04 WO PCT/US2007/010953 patent/WO2008020902A1/fr not_active Ceased
- 2007-05-04 EA EA200870521A patent/EA200870521A1/ru unknown
- 2007-05-04 CN CNA2007800167648A patent/CN101443323A/zh active Pending
- 2007-05-04 KR KR1020087029977A patent/KR20090018107A/ko not_active Withdrawn
- 2007-05-04 GE GEAP200711004A patent/GEP20115140B/en unknown
- 2007-05-04 WO PCT/US2007/010952 patent/WO2007133491A1/fr not_active Ceased
- 2007-05-04 BR BRPI0711352-8A patent/BRPI0711352A2/pt not_active IP Right Cessation
- 2007-05-04 EP EP07835747.2A patent/EP2029575B1/fr active Active
- 2007-05-04 CA CA2650932A patent/CA2650932C/fr not_active Expired - Fee Related
- 2007-05-04 EP EP07776804.2A patent/EP2024358B1/fr active Active
- 2007-05-04 AU AU2007284994A patent/AU2007284994B2/en not_active Ceased
- 2007-05-04 JP JP2009509766A patent/JP4435288B2/ja not_active Expired - Fee Related
- 2007-05-04 MX MX2008014353A patent/MX2008014353A/es active IP Right Grant
- 2007-05-07 US US11/800,626 patent/US7629338B2/en active Active
- 2007-05-08 TW TW096116294A patent/TW200813008A/zh unknown
- 2007-05-08 DO DO2007000093A patent/DOP2007000093A/es unknown
- 2007-05-08 PE PE2007000557A patent/PE20080253A1/es not_active Application Discontinuation
- 2007-05-08 AR ARP070101991A patent/AR060872A1/es not_active Application Discontinuation
-
2008
- 2008-06-19 US US12/214,460 patent/US7625901B2/en active Active
- 2008-10-29 ZA ZA200809275A patent/ZA200809275B/xx unknown
- 2008-10-30 CR CR10415A patent/CR10415A/es not_active Application Discontinuation
- 2008-11-04 IL IL195110A patent/IL195110A0/en unknown
- 2008-11-07 HN HN2008001667A patent/HN2008001667A/es unknown
- 2008-11-07 EC EC2008008872A patent/ECSP088872A/es unknown
- 2008-12-04 MA MA31438A patent/MA30558B1/fr unknown
- 2008-12-08 NO NO20085110A patent/NO20085110L/no not_active Application Discontinuation
-
2009
- 2009-07-08 JP JP2009161479A patent/JP2010013449A/ja not_active Ceased
- 2009-11-09 US US12/614,726 patent/US7893079B2/en active Active
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2017001433A1 (fr) | 2015-06-29 | 2017-01-05 | Galderma Research & Development | Composés d'antagoniste de récepteur cgrp pour le traitement topique de troubles cutanés |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP2029575B1 (fr) | Antagonistes spirocycliques substitués des récepteurs cgrp | |
| EP2091533B1 (fr) | Composés spirocycliques contraints servant d'antagonistes des récepteurs du cgrp | |
| EP1791843B1 (fr) | Antagonistes de recepteurs cgrp d'anilide spirolactame monocyclique | |
| AU2008262403B2 (en) | Carboxamide heterocyclic CGRP receptor antagonists | |
| EP2134342B1 (fr) | Antagonistes des récepteurs cgrp d'anilide spirolactame monocyclique | |
| EP2654423B1 (fr) | Antagonistes azaindane carboxamides hétérocycliques fusionnés de récepteurs du cgrp | |
| EP2685826B1 (fr) | Pipéridinone carboxamide spirohydantoïne, antagonistes du récepteur cgrp | |
| EP1954135B1 (fr) | Antagonistes du récepteur cgrp spirolactame-aryle | |
| EP2146715A1 (fr) | Antagonistes du récepteur cgrp comprenant des groupes terminaux amide tertiaire, sulfamide, carbamate et urée | |
| EP2654421B1 (fr) | Indanecarboxamides condensés comme antagonistes des récepteurs du cgrp | |
| US20110306626A1 (en) | Imidazolinone derivatives as cgrp receptor antagonists | |
| EP2146572B1 (fr) | Antagonistes des récepteurs cgrp hétérocycliques d'anilide bicyclique | |
| EP2271346B1 (fr) | Antagonistes des récepteurs cgrp monocycliques | |
| EP2299813B1 (fr) | Quinoléines 3- et 6-substituées ramifiées en tant qu antagonistes de récepteur cgrp | |
| US8143266B2 (en) | Aryl heterocyclic CGRP receptor antagonists | |
| EP2326176B1 (fr) | Lieurs non amidiques avec terminaison ramifiée constituant des antagonistes de récepteurs de peptides associés au gène de la calcitonine | |
| US20110172205A1 (en) | Monocyclic amide cgrp receptor antagonists | |
| US8507477B2 (en) | 3- and 6-quinolines with N-attached heterocyclic CGRP receptor antagonists |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| 17P | Request for examination filed |
Effective date: 20081209 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IS IT LI LT LU LV MC MT NL PL PT RO SE SI SK TR |
|
| AX | Request for extension of the european patent |
Extension state: AL BA HR MK RS |
|
| RAP1 | Party data changed (applicant data changed or rights of an application transferred) |
Owner name: MERCK SHARP & DOHME CORP. |
|
| 17Q | First examination report despatched |
Effective date: 20100325 |
|
| RAP1 | Party data changed (applicant data changed or rights of an application transferred) |
Owner name: MERCK SHARP & DOHME CORP. |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| INTG | Intention to grant announced |
Effective date: 20140521 |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IS IT LI LT LU LV MC MT NL PL PT RO SE SI SK TR |
|
| AX | Request for extension of the european patent |
Extension state: AL BA HR MK RS |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: REF Ref document number: 692577 Country of ref document: AT Kind code of ref document: T Effective date: 20141115 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R096 Ref document number: 602007038998 Country of ref document: DE Effective date: 20141204 |
|
| REG | Reference to a national code |
Ref country code: NL Ref legal event code: VDEP Effective date: 20141022 |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: MK05 Ref document number: 692577 Country of ref document: AT Kind code of ref document: T Effective date: 20141022 |
|
| REG | Reference to a national code |
Ref country code: LT Ref legal event code: MG4D |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141022 Ref country code: IS Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20150222 Ref country code: ES Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141022 Ref country code: FI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141022 Ref country code: PT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20150223 Ref country code: NL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141022 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: AT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141022 Ref country code: SE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141022 Ref country code: CY Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141022 Ref country code: LV Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141022 Ref country code: GR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20150123 Ref country code: PL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141022 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R097 Ref document number: 602007038998 Country of ref document: DE |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: CZ Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141022 Ref country code: EE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141022 Ref country code: SK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141022 Ref country code: RO Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141022 Ref country code: DK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141022 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141022 |
|
| 26N | No opposition filed |
Effective date: 20150723 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MC Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141022 Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20150531 Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20150531 Ref country code: LU Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20150504 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: MM4A |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141022 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 10 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20150504 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141022 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 11 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: HU Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT; INVALID AB INITIO Effective date: 20070504 Ref country code: BG Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141022 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: TR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141022 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141022 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 12 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R081 Ref document number: 602007038998 Country of ref document: DE Owner name: MERCK SHARP & DOHME LLC, RAHWAY, US Free format text: FORMER OWNER: MERCK SHARP & DOHME CORP., RAHWAY, N.J., US |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: 732E Free format text: REGISTERED BETWEEN 20220908 AND 20220914 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 17 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 20250409 Year of fee payment: 19 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20250410 Year of fee payment: 19 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20250409 Year of fee payment: 19 |