EP4582144A2 - Mittel, verwendungen und verfahren zur behandlung von synukleinopathie - Google Patents
Mittel, verwendungen und verfahren zur behandlung von synukleinopathie Download PDFInfo
- Publication number
- EP4582144A2 EP4582144A2 EP25154059.7A EP25154059A EP4582144A2 EP 4582144 A2 EP4582144 A2 EP 4582144A2 EP 25154059 A EP25154059 A EP 25154059A EP 4582144 A2 EP4582144 A2 EP 4582144A2
- Authority
- EP
- European Patent Office
- Prior art keywords
- synuclein
- alpha
- antibody
- human
- seq
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/395—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/395—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum
- A61K39/39533—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum against materials from animals
- A61K39/3955—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum against materials from animals against proteinaceous materials, e.g. enzymes, hormones, lymphokines
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K51/00—Preparations containing radioactive substances for use in therapy or testing in vivo
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/02—Drugs for disorders of the nervous system for peripheral neuropathies
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/14—Drugs for disorders of the nervous system for treating abnormal movements, e.g. chorea, dyskinesia
- A61P25/16—Anti-Parkinson drugs
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/28—Drugs for disorders of the nervous system for treating neurodegenerative disorders of the central nervous system, e.g. nootropic agents, cognition enhancers, drugs for treating Alzheimer's disease or other forms of dementia
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IG], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IG], e.g. monoclonal or polyclonal antibodies against material from animals or humans
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/68—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving proteins, peptides or amino acids
- G01N33/6893—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving proteins, peptides or amino acids related to diseases not provided for elsewhere
- G01N33/6896—Neurological disorders, e.g. Alzheimer's disease
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/505—Medicinal preparations containing antigens or antibodies comprising antibodies
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/20—Immunoglobulins specific features characterized by taxonomic origin
- C07K2317/21—Immunoglobulins specific features characterized by taxonomic origin from primates, e.g. man
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/30—Immunoglobulins specific features characterized by aspects of specificity or valency
- C07K2317/33—Crossreactivity, e.g. for species or epitope, or lack of said crossreactivity
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/30—Immunoglobulins specific features characterized by aspects of specificity or valency
- C07K2317/34—Identification of a linear epitope shorter than 20 amino acid residues or of a conformational epitope defined by amino acid residues
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/56—Immunoglobulins specific features characterized by immunoglobulin fragments variable (Fv) region, i.e. VH and/or VL
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/56—Immunoglobulins specific features characterized by immunoglobulin fragments variable (Fv) region, i.e. VH and/or VL
- C07K2317/565—Complementarity determining region [CDR]
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/76—Antagonist effect on antigen, e.g. neutralization or inhibition of binding
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/90—Immunoglobulins specific features characterized by (pharmaco)kinetic aspects or by stability of the immunoglobulin
- C07K2317/92—Affinity (KD), association rate (Ka), dissociation rate (Kd) or EC50 value
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N2800/00—Detection or diagnosis of diseases
- G01N2800/28—Neurological disorders
- G01N2800/2835—Movement disorders, e.g. Parkinson, Huntington, Tourette
Definitions
- the present invention relates to a novel class of monoclonal antibody that specifically binds to alpha-synuclein, as well as to methods of using these molecules and their alpha-synuclein binding fragments in the treatment and diagnosis of synucleinopathies.
- Synucleinopathies also known as Lewy body diseases (LBDs) are characterized by deposition of intracellular protein aggregates that are microscopically visible as Lewy bodies (LBs) and/or Lewy neurites, where the protein alpha-synuclein is the major component ( Jellinger, Mov Disord. 2012 Jan;27(1):8-30 ; McKeith et al., Neurology (1996) 47:1113-24 ).
- Synucleinopathies include Parkinson's disease (PD) (including idiopathic and inherited forms of Parkinson's disease) and Diffuse Lewy Body (DLB) disease (also known as Dementia with Lewy Bodies (DLB), Lewy body variant of Alzheimer's disease (LBV), Combined Alzheimer's and Parkinson disease (CAPD), pure autonomic failure (PAF) and multiple system atrophy (MSA; e.g., Olivopontocerebellar Atrophy, Striatonigral Degeneration and Shy-Drager Syndrome)).
- PD Parkinson's disease
- DLB Diffuse Lewy Body
- LBV Dementia with Lewy Bodies
- LBV Lewy body variant of Alzheimer's disease
- CAPD Combined Alzheimer's and Parkinson disease
- MSA multiple system atrophy
- Olivopontocerebellar Atrophy Striatonigral Degeneration and Shy-Drager Syndrome
- Synucleinopathies frequently have degeneration of the dopaminergic nigrostriatal system, responsible for the core motor deficits in Parkinsonism (rigidity, bradykinesia, resting tremor), but there is also widespread occurrence of Lewy bodies and dystrophic Lewy neurites in the central, peripheral and autonomic nervous system and brain regions and other organs associated with non-motor dysfunctions, such as dementia and autonomic nervous system deficits.
- Several of the non-motor signs and symptoms are thought to precede motor symptoms in Parkinson's disease and other synucleinopathies. Such early signs include, for example, REM sleep behaviour disorder (RBD) and loss of smell and constipation ( Mahowald et al., Neurology (2010) 75:488-489 ).
- Synucleinopathies continue to be a common cause for movement disorders and cognitive deterioration in the aging population ( Galasko et al., Arch. Neurol. (1994) 51:888-95 ).
- Alpha-synuclein is a member of a family of proteins including beta- and gamma-synuclein and synoretin. Alpha-synuclein is expressed in the normal state associated with synapses and is believed to play a role in regulating synaptic vesicle release and thereby affecting neural communication, plasticity, learning and memory.
- alpha-synuclein with a central role in PD pathogenesis.
- the protein can aggregate to form intracellular insoluble fibrils in pathological conditions.
- synuclein accumulates in LBs ( Spillantini et al., Nature (1997) 388:839-40 ; Takeda et al., J. Pathol. (1998) 152:367-72 ; Wakabayashi et al., Neurosci. Lett. (1997) 239:45-8 ).
- Mutations in the alpha-synuclein gene as well as duplications and triplications of the gene co-segregate with rare familial forms of parkinsonism ( Kruger et al., Nature Gen.
- Naturally occurring alpha-synuclein auto-antibodies have been shown to be present in both PD patients and healthy controls ( Smith et al. 2012, PLoS One. 2012;7(12):e52285 ; Maetzler et al. 2014, PLoS One. 2014 Feb 21;9(2):e88604 , Papachroni et al. 2007 J Neurochem. 2007 May;101(3):749-56 and Woulfe et al. 2002, Neurology. 2002 May 14;58(9):1435-6 ), sometimes increased levels of auto-antibodies to alpha-synuclein in PD ( Gruden et al. 2011, J Neuroimmunol.
- mice Over expression of alpha-synuclein in transgenic mice mimics some pathological aspects of Lewy body disease.
- Several different transgenic lines of mice over-expressing alpha-synuclein have been generated in the last ten years (described in reviews: Koehler et al 2014, PLoS One. 2013 May 31;8(5):e64649 ; Fleming and Chesselet, 2006,Behav Pharmacol. 2006 Sep;17(5-6):383-91 ; Springer and Kahle 2006,Curr Neurol Neurosci Rep. 2006 Sep;6(5):432-6 ).
- alpha-synuclein gene and posttranslational modifications of the protein can create alpha-synuclein protein forms that have enhanced capacity to form aggregated and/or toxic forms of alpha-synuclein ( Beyer and Ariza, Mol Neurobiol. 2013 Apr;47(2):509-24 ).
- the precise pathological species of alpha-synuclein remains unknown.
- Various misfolded/aggregated/secreted species ranging from oligomers to fibrils, and different post-translational modifications have been associated with toxicity but there is no consensus on which is most important, if indeed there even is a single toxic species.
- a C-terminal antibody 274 which targets an epitope that involves alpha-synuclein residues 120-140 was also shown to have an effect in a preclinical model on spreading of the pathology from cell to cell.
- antibodies targeting conformational species such as oligomers and fibrils of alpha-synuclein have been shown to be able to at least reduce the levels of these presumably toxic alpha-synuclein species ( Lindström et al. 2014, Neurobiol Dis. 2014 Sep;69:134-43 and Spencer et al. 2014, Mol Ther.
- amino acids that are important for binding of 9E4 are asparagine 122 and tyrosine 125 (according to an alanine scan presented in patent US20140127131 ), and this means that this antibody cannot bind alpha-synuclein which is truncated at amino acids 119, and 122, which are some of the major truncated species in Parkinson brain tissue ( Kellie et al. Sci Rep. 2014;4:5797 ). The same would be the case for the antibody 274 and antibody mab47 ( US8,632,776 ).
- amino terminal antibodies would possibly not be able to bind to some of the major truncated species that lack the first amino acids of alpha-synuclein, such as alpha-synuclein truncated to amino acids 5-140.
- one suggested mechanism of action is the prevention of truncation at amino acids 119-122 in extracellular space, as the antibody will bind to the same region where the protease that will cleave alpha-synuclein ( Games et al. 2014, J Neurosci. 2014 Jul 9;34(28):9441-54 ).
- a similar mechanism of action could also be found with antibodies in close proximity of the site, and therefore many antibodies around this region would be expected to have this activity.
- antibodies such as "GM37” and "GM285", described in the Examples
- GM37 and GM285 antibodies
- the antibodies of the invention are capable of binding to other oligomeric forms of alpha-synuclein and altering their uptake by other CNS resident cells in a manner that reduce the spreading of disease.
- the antibodies of the invention such as GM37 and 285, were surprisingly found to be superior to prior art antibodies such as 9E4 in binding to different alpha-synuclein species in human brain, and has a surprising superior effect on clearing extracellular alpha-synuclein and normalising impaired synaptic transmission induced by the presence of abnormal alpha-synuclein in vivo. Further illustrating their therapeutic capabilities, the antibodies of the invention, such as GM37 and 285, are able to prevent the appearance of a disease related motor phenotype in a rat model for Parkinson's disease.
- antibodies GM37 and GM285 are able to inhibit seeding of aggregation and phosphorylation of endogenous alpha-synuclein induced by extracellular added recombinant pathological alpha-synuclein seeds in primary mouse neurons.
- Antibodies such as GM37 and 285 can also inhibit seeding of alpha-synuclein pathology into dopaminergic neurons in vivo using a mouse model for Parkinson's disease, further supporting the therapeutic capability of these antibodies in preventing the cell to cell propagation of pathology.
- GM37 and GM285 as new therapeutic agents capable of modifying disease through inhibition of the mechanism by which the disease pathology spreads between the neurons Parkinson's patients.
- GM37 antibody In a further aspect of the invention is provided 3 amino acid variants of the GM37 antibody. All the variants have similar functional readouts as the parent antibody, GM37, but with improved properties for manufacturability.
- the variants reduce the risk of post-translational modification occurring within the binding domain of the GM37 antibody and provide some improvement in the production of the antibody. This is advantageous because large scale clinical or commercial manufacturing of antibodies is complicated and expensive, and providing a homogenous product in pharmaceutical medicaments is crucial in particular for immunoglobulins and proteins.
- the invention relates to novel monoclonal antibodies, and antigen-binding fragments thereof, capable of specifically binding an epitope within amino acids 112-117 in alpha-synuclein (SEQ ID NO:9 (ILEDMP)).
- the epitope bound by the antibodies or antibody-binding fragments thereof of the invention such as exemplary antibody "GM37", or "GM285", is referred to herein as "the 112-117 epitope".
- the antibodies of the present invention specifically bind to an epitope within the 112-117 epitope and may, according to one embodiment, compete with antibody GM37 or GM285 for binding to an epitope within amino acids 112-117.
- antibodies or antigen-binding fragments thereof according to the invention may compete for binding to an epitope within amino acids 112-117 of human alpha-synuclein with a heavy chain consisting of a variable domain of SEQ ID NO:7 and a light chain consisting of a variable domain of SEQ ID NO:8.
- Such competitive binding inhibition can be determined using assays and methods well known in the art, for example using an unlabelled binding assay such as surface plasmon resonance (SPR).
- SPR surface plasmon resonance
- the GM285 antibody binds an epitope within residues 112-117 of alpha-synuclein comprising residues 112-115 of alpha-synuclein (ILED; SEQ ID NO:19).
- the invention relates to monoclonal antibody GM37, its variants (e.g., GM37 Variant 1, GM37 Variant 2 and GM37 Variant 3), or GM285.
- GM37 monoclonal antibody
- its variants e.g., GM37 Variant 1, GM37 Variant 2 and GM37 Variant 3
- the invention provides a monoclonal antibody GM37, its variants (e.g., GM37 Variant 1, GM37 Variant 2 and GM37 Variant 3), or GM285, and encompasses such antibodies as well as derivatives thereof that possess a sufficient number (e.g., 1, 2, or 3) light chain CDRs and a sufficient number (e.g., 1, 2, or 3) heavy chain CDRs to form a binding site capable of specifically binding to human synuclein.
- a sufficient number e.g., 1, 2, or 3
- light chain CDRs e.g., 1, 2, or 3
- heavy chain CDRs e.g., 1, 2, or 3
- such antibodies will possess the three light chain CDRs and three heavy chain CDRs, as defined below.
- the numbering of amino acid residues in this region is according to IMGT ® , the international ImMunoGeneTics information system ® or, Kabat, E. A., Wu, T. T., Perry, H. M., Gottesmann, K. S. & Foeller, C. (1991). Sequences of Proteins of Immunological Interest, 5th edit., NIH Publication no. 91-3242 U.S. Department of Health and Human Services ; Chothia, C. & Lesk, A. M. (1987). Canonical structures For The Hypervariable domains Of Immunoglobulins. J. Mol. Biol. 196, 901-917 .
- the monoclonal antibody or antigen-binding fragments thereof possesses a synuclein antigen-binding fragment comprising or consisting of:
- the monoclonal antibody or antigen-binding fragments thereof possesses a synuclein antigen-binding fragment comprising or consisting of:
- the monoclonal antibody or antigen-binding fragments thereof possesses a synuclein antigen-binding fragment comprising or consisting of:
- the monoclonal antibody or antigen-binding fragments thereof possesses a synuclein antigen-binding fragment comprising an amino acid sequence (in its CDRs, its variable domains, its framework residues or in its constant domains) that differs from that of naturally occurring anti-alpha-synuclein antibodies, and that exhibits (relative to such naturally occurring anti-alpha-synuclein antibodies):
- the antibodies and antigen-binding fragments thereof of the invention may be used in a method to treat, diagnose or image synucleinopathies, such as Parkinson's disease ((PD), including idiopathic and inherited forms of Parkinson's disease), Diffuse Lewy Body Disease (DLBD), Lewy body variant of Alzheimer's disease (LBV), Gauchers Disease (GD), Combined Alzheimer's and Parkinson disease (CAPD), pure autonomic failure and multiple system atrophy.
- Parkinson's disease including idiopathic and inherited forms of Parkinson's disease
- DLBD Diffuse Lewy Body Disease
- LBV Lewy body variant of Alzheimer's disease
- GD Gauchers Disease
- CAPD Combined Alzheimer's and Parkinson disease
- alpha-synuclein is synonymous with “the alpha-synuclein protein” and refers to any of the alpha-synuclein protein isoforms (identified in, for example, UniProt as P37840, 1-3).
- the amino acid numbering of alpha-synuclein is given with respect to SEQ ID NO:10 as shown below, with methionine (M) being amino acid residue1:
- the present invention relates to antibodies and to fragments of antibodies that are capable of specifically binding to alpha-synuclein, and in particular to human alpha-synuclein.
- the antibodies and fragments thereof exhibit the ability to specifically bind to an epitope within 112-117 of human alpha-synuclein.
- antibody in the context of the present invention refers to an immunoglobulin molecule or according to some embodiments of the invention, a fragment of an immunoglobulin molecule which has the ability to specifically bind to an epitope of a molecule ("antigen").
- Naturally occurring antibodies typically comprise a tetramer which is usually composed of at least two heavy (H) chains and at least two light (L) chains. Each heavy chain is comprised of a heavy chain variable domain (abbreviated herein as VH) and a heavy chain constant domain, usually comprised of three domains (CH1, CH2 and CH3).
- Heavy chains can be of any isotype, including IgG (IgG1, IgG2, IgG3 and IgG4 subtypes), IgA (IgA1 and IgA2 subtypes), IgM and IgE.
- Each light chain is comprised of a light chain variable domain (abbreviated herein as VL) and a light chain constant domain (CL).
- Light chains include kappa chains and lambda chains.
- the heavy and light chain variable domain is typically responsible for antigen recognition, while the heavy and light chain constant domain may mediate the binding of the immunoglobulin to host tissues or factors, including various cells of the immune system (e.g., effector cells) and the first component (C1q) of the classical complement system.
- VH and VL regions can be further subdivided into regions of hypervariability, termed “complementarity determining regions,” that are interspersed with regions of more conserved sequence, termed “framework regions” (FR).
- FR frame regions
- Each VH and VL is composed of three CDR Domains and four FR Domains arranged from amino-terminus to carboxy-terminus in the following order: FR1-CDR1-FR2-CDR2-FR3-CDR3-FR4.
- the variable domains of the heavy and light chains contain a binding domain that interacts with an antigen.
- antibodies and their antigen-binding fragments that have been "isolated” so as to exist in a physical milieu distinct from that in which it may occur in nature or that have been modified so as to differ from a naturally occurring antibody in amino acid sequence.
- epitope means an antigenic determinant capable of specific binding to an antibody.
- Epitopes usually consist of surface groupings of molecules such as amino acids or sugar side chains and usually have specific three dimensional structural characteristics, as well as specific charge characteristics. Conformational and linear epitopes are distinguished in that the binding to the former, but not the latter, is always lost in the presence of denaturing solvents.
- the epitope may comprise amino acid residues directly involved in the binding and other amino acid residues, which are not directly involved in the binding, such as amino acid residues which are effectively blocked by the specifically antigen-binding peptide (in other words, the amino acid residue is within the footprint of the specifically antigen-binding peptide).
- 112-117 epitope refers to a region of human alpha-synuclein that contains at least 4 of the 6 amino acid residues of 112-117 human alpha-synuclein, which epitope does not include any residue from 1-111 (including any residue from 106-111) of human alpha-synuclein, nor any residue from 118-140 (including residue 118-120) of human alpha-synuclein.
- an antibody is said to be capable of specifically binding to an epitope within the "112-117 epitope” if it is capable of specifically binding to human alpha-synuclein by binding to at least 4 of the 6 amino acid residues of the 112-117 epitope.
- the term "antigen-binding fragment of an antibody” means a fragment, portion, region or domain of an antibody (regardless of how it is produced ( e.g., via cleavage, recombinantly, synthetically, etc. )) that is capable of specifically binding to an epitope, and thus the term “antigen-binding” is intended to mean the same as “epitope-binding” so that, for example, an "antigen-binding fragment of an antibody” is intended to be the same as an “epitope-binding fragment of an antibody”.
- An antigen-binding fragment may contain 1, 2, 3, 4, 5 or all 6 of the CDR Domains of such antibody and, although capable of specifically binding to such epitope, may exhibit a specificity, affinity or selectivity toward such epitope that differs from that of such antibody.
- an antigen-binding fragment will contain all 6 of the CDR Domains of such antibody.
- An antigen-binding fragment of an antibody may be part of, or comprise, a single polypeptide chain (e.g ., an scFv), or may be part of, or comprise, two or more polypeptide chains, each having an amino-terminus and a carboxyl terminus (e.g., a diabody, a Fab fragment, a Fab 2 fragment, etc .) .
- Fragments of antibodies that exhibit antigen-binding ability can be obtained, for example, by protease cleavage of intact antibodies.
- the two domains of the Fv fragment, VL and VH are naturally encoded by separate genes, or polynucleotides that encode such gene sequences (e.g., their encoding cDNA) can be joined, using recombinant methods, by a flexible linker that enables them to be made as a single protein chain in which the VL and VH regions associate to form monovalent antigen-binding molecules (known as single-chain Fv (scFv); see e.g., Bird et al. , (1988) Science 242:423-426 ; and Huston et al. (1988) Proc. Natl. Acad. Sci. (U.S.A.) 85:5879-5883 ).
- scFv single-chain Fv
- a flexible linker that is too short (e.g., less than about 9 residues) to enable the VL and VH regions of a single polypeptide chain to associate together
- a bispecific antibody, diabody, or similar molecule in which two such polypeptide chains associate together to form a bivalent antigen-binding molecule
- antigen-binding fragments encompassed within the present invention include (i) a Fab' or Fab fragment, a monovalent fragment consisting of the VL, VH, CL and CH1 domains, or a monovalent antibody as described in WO2007059782 ; (ii) F(ab')2 fragments, bivalent fragments comprising two Fab fragments linked by a disulfide bridge at the hinge domain; (iii) an Fd fragment consisting essentially of the VH and CH1 domains; (iv) a Fv fragment consisting essentially of a VL and VH domains, (v) a dAb fragment ( Ward et al., Nature 341, 544-546 (1989 )), which consists essentially of a VH domain and also called domain antibodies ( Holt et al; Trends Biotechnol.
- GM37 variants refers to the deaminated variants 1,2 or 3, wherein variant 1 has a N54S substitution, variant 2 has a N54Q substitution and variant 3 has a N54H compared to the GM37 antibody described herein above.
- antibody GM285 is intended to include an antibody or antigen-binding fragment thereof comprising or consisting of the Heavy Chain as given in CDR1-3 SEQ ID NOs:20-22 and the Light Chain CDR1-3 as given in SEQ ID NOs:23-25.
- the antibody GM37 or antigen-binding fragment thereof may comprise or consist of the heavy chain variable domain of SEQ ID NO:26 and/or the light chain variable domain of SEQ ID NO:27.
- the antibody GM37 may be an IgG antibody comprising a heavy chain consisting of a variable domain of SEQ ID NO:26 and a constant domain of SEQ ID NO:28 together with a light chain consisting of a variable domain of SEQ ID NO:27 and a kappa constant domain of SEQ ID NO:29.
- the GM285 antibody specifically binds an epitope within the sequence 112-115 (ILED; SEQ ID NO:19) of human alpha-synuclein (SEQ ID NO:10).
- human antibody (which may be abbreviated to "humAb” or “HuMab”), as used herein, is intended to include antibodies having variable and constant domains derived from human germline immunoglobulin sequences.
- the human antibodies of the invention may include amino acid residues not encoded by human germline immunoglobulin sequences (e.g., mutations introduced by random or site-specific mutagenesis in vitro or during gene rearrangement or by somatic mutation in vivo).
- humanized antibodies preserve all CDR sequences (for example, a humanized mouse antibody which contains all six CDRs from the mouse antibodies).
- humanized antibodies have one or more CDRs (one, two, three, four, five, six) which are altered with respect to the original antibody, which are also termed one or more CDRs "derived from" one or more CDRs from the original antibody.
- CDRs one, two, three, four, five, six
- the ability to humanize an antigen is well known (see, e.g ., US Patents No. 5,225,539 ; 5,530,101 ; 5,585,089 ; 5,859,205 ; 6,407,213 ; 6,881,557 ).
- an antibody or an antigen-binding fragment thereof is said to "specifically" bind a region of another molecule (i.e., an epitope) if it reacts or associates more frequently, more rapidly, with greater duration and/or with greater affinity or avidity with that epitope relative to alternative epitopes.
- the antibody, or antigen-binding fragment thereof, of the invention binds at least 10-fold more strongly to its target (human alpha synuclein) than to another molecule; preferably at least 50-fold more strongly and more preferably at least 100-fold more strongly.
- the antibody, or antigen-binding fragment thereof binds under physiological conditions, for example, in vivo.
- binding in the context of the binding of an antibody to a predetermined antigen typically refers to binding with an affinity corresponding to a KD of about 10 -7 M or less, such as about 10 -8 M or less, such as about 10 -9 M or less when determined by for instance surface plasmon resonance (SPR) technology in either a BIAcore ® 3000 or T200instrument using the antigen as the ligand and the antibody as the analyte, and binds to the predetermined antigen with an affinity corresponding to a KD that is at least ten-fold lower, such as at least 100 fold lower, for instance at least 1,000 fold lower, such as at least 10,000 fold lower, for instance at least 100,000 fold lower than its affinity for binding to a non-specific antigen (e.g., BSA, casein) other than the predetermined antigen or a closely-related antigen.
- a non-specific antigen e.g., BSA, casein
- the amount with which the affinity is lower is dependent on the KD of the antibody, so that when the KD of the antibody is very low (that is, the antibody is highly specific), then the amount with which the affinity for the antigen is lower than the affinity for a non-specific antigen may be at least 10,000 fold.
- kd (sec -1 or 1/s), as used herein, refers to the dissociation rate constant of a particular antibody-antigen interaction. Said value is also referred to as the koff value.
- ka (M-1 x sec-1 or 1/Msec), as used herein, refers to the association rate constant of a particular antibody-antigen interaction.
- KD (M) refers to the dissociation equilibrium constant of a particular antibody-antigen interaction and is obtained by dividing the kd by the ka.
- KA (M-1 or 1/M), as used herein, refers to the association equilibrium constant of a particular antibody-antigen interaction and is obtained by dividing the ka by the kd.
- the term "capability of inhibiting protease truncation of alpha-synuclein fibrils” includes the capability of inhibiting calpain-1 induced formation of fragment 1-119-122 of human alpha synuclein in primary cortical neurons (see Example 5 ).
- the term "capability, when administered chronically, to restore motor function in a rat model of Parkinson's disease” include the capability to reduce or eliminate motor asymmetry in a rat recombinant adeno-associated viral vector (rAAV) model of Parkinson's Disease (see Example 8 ).
- substitutions of acceptor for donor amino acids in the CDRs to include reflects a balance of competing considerations. Such substitutions are potentially advantageous in decreasing the number of mouse amino acids in a humanized antibody and consequently decreasing potential immunogenicity. However, substitutions can also cause changes of affinity, and significant reductions in affinity are preferably avoided. Positions for substitution within CDRs and amino acids to substitute can also be selected empirically.
- a single amino acid alteration of a CDR residue can result in loss of functional binding ( Rudikoff, S. etc. (1982) "Single Amino Acid Substitution Altering Antigen-Binding Specificity," Proc. Natl. Acad. Sci. (USA) 79(6):1979-1983 ) provides a means for systematically identifying alternative functional CDR sequences.
- a polynucleotide encoding the CDR is mutagenized (for example via random mutagenesis or by a site-directed method (e.g ., polymerase chain-mediated amplification with primers that encode the mutated locus)) to produce a CDR having a substituted amino acid residue.
- the BLOSUM62.iij substitution score for that substitution can be identified.
- the BLOSUM system provides a matrix of amino acid substitutions created by analyzing a database of sequences for trusted alignments ( Eddy, S.R. (2004) “Where Did The BLOSUM62 Alignment Score Matrix Come From?,” Nature Biotech. 22(8):1035-1036 ; Henikoff, J.G. (1992) "Amino acid substitution matrices from protein blocks," Proc. Natl. Acad. Sci. (USA) 89:10915-10919 ; Karlin, S. et al.
- an antigen-binding fragment comprising the resultant CDR fails to bind to alpha-synuclein, for example, then the BLOSUM62.iij substitution score is deemed to be insufficiently conservative, and a new candidate substitution is selected and produced having a higher substitution score.
- the BLOSUM62.iij substitution score will be 0, and more conservative changes (such as to aspartate, asparagine, glutamine, or lysine) are preferred.
- the invention thus contemplates the use of random mutagenesis to identify improved CDRs.
- conservative substitutions may be defined by substitutions within the classes of amino acids reflected in one or more of the following three tables:
- sequence of CDR variants of encompassed antibodies or their antigen-binding fragments may differ from the sequence of the CDR of the parent antibody, GM37, GM37 var 1-3, or 285, through substitutions; for instance substituted 4 amino acid residue, 3 amino acid residue, 2 amino acid residue or 1 of the amino acid residues.
- substitutions for instance substituted 4 amino acid residue, 3 amino acid residue, 2 amino acid residue or 1 of the amino acid residues.
- the amino acids in the CDR regions may be substituted with conservative substitutions, as defined in the 3 tables above.
- treatment means ameliorating, slowing, attenuating or reversing the progress or severity of a disease or disorder, or ameliorating, slowing, attenuating or reversing one or more symptoms or side effects of such disease or disorder.
- an “effective amount,” when applied to an antibody or antigen-binding fragments thereof, of the invention, refers to an amount sufficient, at dosages and for periods of time necessary, to achieve an intended biological effect or a desired therapeutic result including, without limitation, clinical results.
- the phrase "therapeutically effective amount" when applied to an antibody or antigen-binding fragments thereof, of the invention is intended to denote an amount of the antibody, or antigen-binding fragment thereof, that is sufficient to ameliorate, palliate, stabilize, reverse, slow, attenuate or delay the progression of a disorder or disease state, or of a symptom of the disorder or disease.
- the method of the present invention provides for administration of the antibody, or antigen-binding fragment thereof, in combinations with other compounds. In such instances, the "effective amount" is the amount of the combination sufficient to cause the intended biological effect.
- a therapeutically effective amount of an anti-alpha-synuclein antibody or antigen-binding fragment thereof of the invention may vary according to factors such as the disease state, age, sex, and weight of the individual, and the ability of the anti-alpha-synuclein antibody to elicit a desired response in the individual.
- a therapeutically effective amount is also one in which any toxic or detrimental effects of the antibody or antibody portion are outweighed by the therapeutically beneficial effects.
- the present invention particularly relates to a monoclonal antibody capable of specifically binding to an epitope within amino acids 112-117 of human alpha-synuclein (SEQ ID NO:9 (ILEDMP)).
- the antibody is capable of competing with the antibody GM37 for binding to an epitope within the 112-117 amino acids of alpha-synuclein.
- the antibodies of the present invention exemplified by GM37 its variants GM37 var 1-3 and GM285, and their alpha-synuclein binding fragments are capable of binding the toxic alpha-synuclein fragment consisting of residues 1-119/122 of alpha-synuclein and neutralizing its toxicity (for example, by extracellular binding to the alpha-synuclein fragment and thereby preventing it from being taken up by cells.
- the antibodies of the present invention which are capable of binding to an epitope within amino acids 112-117 of alpha-synuclein are superior to prior art antibodies such as antibody 9E4 in binding to toxic alpha-synuclein species in human brain, and have superior effects on clearing extracellular alpha-synuclein and normalising an impaired synaptic transmission induced by alpha-synuclein in vivo.
- the antibodies of the invention are also able to ameliorate the appearance of a relevant motor phenotype in a rat model for Parkinson's disease.
- the antibodies may be in a composition together with a pharmaceutically acceptable carrier, diluent and/or stabilizer.
- the antibodies of the invention may be used in therapy.
- the antibodies of the invention may be used in treating synucleinopathies such as Parkinson's disease (including idiopathic inherited forms of Parkinson's disease), Gaucher's Disease, Diffuse Lewy Body Disease (DLBD), Lewy body variant of Alzheimer's disease (LBV), Combined Alzheimer's and Parkinson disease, pure autonomic failure and multiple system atrophy.
- synucleinopathies such as Parkinson's disease (including idiopathic inherited forms of Parkinson's disease), Gaucher's Disease, Diffuse Lewy Body Disease (DLBD), Lewy body variant of Alzheimer's disease (LBV), Combined Alzheimer's and Parkinson disease, pure autonomic failure and multiple system atrophy.
- the treatment envisioned by the present invention may be chronic and the patient may be treated at least 2 weeks, such as at least for 1 month, 6, months, 1 year or more.
- the antibodies of antigen-binding fragments thereof of the present invention may be produced in different cell lines, such as a human cell line, a mammal non-human cell line, and insect cell line, for example a CHO cell line, HEK cell line, BHK-21 cell line, murine cell line (such as a myeloma cell line), fibrosarcoma cell line, PER.C6 cell line, HKB-11 cell line, CAP cell line and HuH-7 human cell line ( Dumont et al, 2015, Crit Rev Biotechnol. Sep 18:1-13 ., the contents which is included herein by reference).
- insect cell line for example a CHO cell line, HEK cell line, BHK-21 cell line, murine cell line (such as a myeloma cell line), fibrosarcoma cell line, PER.C6 cell line, HKB-11 cell line, CAP cell line and HuH-7 human cell line ( Dumont et al, 2015, Crit Rev Biotechnol. Sep 18
- the antibodies of the present invention may for example be monoclonal antibodies produced by the hybridoma method first described by Kohler et al., Nature 256, 495 (1975 ), or may be produced by recombinant DNA methods. Monoclonal antibodies may also be isolated from phage antibody libraries using the techniques described in, for example, Clackson et al., Nature 352, 624-628 (1991 ) and Marks et al., J. Mol. Biol. 222, 581-597 (1991 ). Monoclonal antibodies may be obtained from any suitable source.
- the antibody of the invention is a human antibody.
- Human monoclonal antibodies directed against alpha-synuclein may be generated using transgenic or transchromosomal mice carrying parts of the human immune system rather than the mouse system.
- transgenic and transchromosomic mice include mice referred to herein as HuMAb mice and KM mice, respectively.
- the HuMAb mouse contains a human immunoglobulin gene minilocus that encodes unrearranged human heavy variable and constant ( ⁇ and Y) and light variable and constant ( ⁇ ) chain immunoglobulin sequences, together with targeted mutations that inactivate the endogenous ⁇ and ⁇ chain loci ( Lonberg, N. et al., Nature 368, 856-859 (1994 )). Accordingly, the mice exhibit reduced expression of mouse IgM or Ig ⁇ and in response to immunization, the introduced human heavy and light chain transgenes, undergo class switching and somatic mutation to generate high affinity human IgG, ⁇ monoclonal antibodies (Lonberg, N. et al.
- HuMAb mice The preparation of HuMAb mice is described in detail in Taylor, L. et al., Nucleic Acids Research 20, 6287-6295 (1992 ), Chen, J. et al., International Immunology 5, 647-656 (1993 ), Tuaillon et al., J. Immunol. 152, 2912-2920 (1994 ), Taylor, L.
- mice have a JKD disruption in their endogenous light chain (kappa) genes (as described in Chen et al., EMBO J. 12, 811-820 (1993 )), a CMD disruption in their endogenous heavy chain genes (as described in Example 1 of WO 01/14424 ), and a KCo5 human kappa light chain transgene (as described in Fishwild et al., Nature Biotechnology 14, 845-851 (1996 )).
- kappa endogenous light chain
- CMD disruption in their endogenous heavy chain genes as described in Example 1 of WO 01/14424
- KCo5 human kappa light chain transgene as described in Fishwild et al., Nature Biotechnology 14, 845-851 (1996 )
- the HCo7 mice have a HCo7 human heavy chain transgene (as described in US 5,770,429 ), the HCo12 mice have a HCo12 human heavy chain transgene (as described in Example 2 of WO 01/14424 ), the HCo17 mice have a HCo17 human heavy chain transgene (as described in Example 2 of WO 01/09187 ) and the HCo20 mice have a HCo20 human heavy chain transgene.
- the resulting mice express human immunoglobulin heavy and kappa light chain transgenes in a background homozygous for disruption of the endogenous mouse heavy and kappa light chain loci.
- the endogenous mouse kappa light chain gene has been homozygously disrupted as described in Chen et al., EMBO J. 12, 811-820 (1993 ) and the endogenous mouse heavy chain gene has been homozygously disrupted as described in Example 1 of WO 01/09187 .
- This mouse strain carries a human kappa light chain transgene, KCo5, as described in Fishwild et al., Nature Biotechnology 14, 845-851 (1996 ).
- This mouse strain also carries a human heavy chain transchromosome composed of chromosome 14 fragment hCF (SC20) as described in WO 02/43478 .
- HCo12-Balb/c, HCo17-Balb/c and HCo20-Balb/c mice can be generated by crossing HCo12, HCo17 and HCo20 to KCo5[J/K](Balb) as described in WO 09/097006 .
- the endogenous mouse kappa light chain gene has been homozygously disrupted as described in Chen et al., EMBO J. 12, 811-820 (1993 ) and the endogenous mouse heavy chain gene has been homozygously disrupted as described in Example 1 of WO 01/09187 .
- This mouse strain carries a human kappa light chain transgene, KCo5, as described in Fishwild et al., Nature Biotechnology 14, 845-851 (1996 ).
- This mouse strain also carries a human heavy chain trans-chromosome composed of chromosome 14 antigen-binding fragment hCF (SC20) as described in WO 02/43478 .
- Splenocytes from these transgenic mice may be used to generate hybridomas that secrete human monoclonal antibodies according to well-known techniques.
- Human monoclonal or polyclonal antibodies of the present invention, or antibodies of the present invention originating from other species may also be generated transgenically through the generation of another non-human mammal or plant that is transgenic for the immunoglobulin heavy and light chain sequences of interest and production of the antibody in a recoverable form therefrom.
- antibodies may be produced in, and recovered from, the milk of goats, cows, or other mammals. See for instance US 5,827,690 , US 5,756,687 , US 5,750,172 and US 5,741,957 .
- An F(ab') antigen-binding fragment may also be produced by binding Fab'-described below via a thioether bond or a disulfide bond.
- a Fab' antigen-binding fragment is an antibody antigen-binding fragment obtained by cutting a disulfide bond of the hinge domain of the F(ab')2.
- a Fab'- antigen-binding fragment may be obtained by treating an F(ab')2 antigen-binding fragment with a reducing agent, such as dithiothreitol.
- Antibody antigen-binding fragment may also be generated by expression of nucleic acids encoding such antigen-binding fragments in recombinant cells (see for instance Evans et al., J. Immunol. Meth.
- Anti-alpha-synuclein antibodies, and antigen-binding fragments thereof, of the invention also include single chain antibodies.
- Single chain antibodies are peptides in which the heavy and light chain Fv regions are connected.
- the present invention provides a single-chain Fv (scFv) wherein the heavy and light chains in the Fv of an anti-alpha-synuclein antibody of the present invention are joined with a flexible peptide linker (typically of about 10, 12, 15 or more amino acid residues) in a single peptide chain.
- a flexible peptide linker typically of about 10, 12, 15 or more amino acid residues
- Chemiluminescent labels may be employed (e.g., luminol, luciferase, luciferin, and aequorin). Such diagnosis and detection can also be accomplished by coupling the diagnostic molecule of the present invention to detectable substances including, but not limited to, various enzymes, enzymes including, but not limited to, horseradish peroxidase, alkaline phosphatase, beta-galactosidase, or acetylcholinesterase, or to prosthetic group complexes such as, but not limited to, streptavidin/biotin and avidin/biotin.
- Chemiluminescent labels may be employed (e.g., luminol, luciferase, luciferin, and aequorin). Such diagnosis and detection can also be accomplished by coupling the diagnostic molecule of the present invention to detectable substances including, but not limited to, various enzymes, enzymes including, but not limited to, horseradish peroxidase, alkaline phosphatase, beta-galactosidase, or acetylcholinesterase, or to prosthetic group complexes such as, but not limited to, streptavidin/biotin and avidin/biotin.
- Paramagnetic labels can also be employed, and are preferably detected using Positron Emission Tomography (PET) or Single-Photon Emission Computed Tomography (SPECT).
- Such paramagnetic labels include, but are not limited to compounds containing paramagnetic ions of Aluminum (Al), Barium (Ba), Calcium (Ca), Cerium (Ce), Dysprosium (Dy), Erbium (Er), Europium (Eu), Gandolinium (Gd), Holmium (Ho), Iridium (Ir), Lithium (Li), Magnesium (Mg), Manganese (Mn), Molybdenum (M), Neodymium (Nd), Osmium (Os), Oxygen (O), Palladium (Pd), Platinum (Pt), Rhodium (Rh), Ruthenium (Ru), Samarium (Sm), Sodium (Na), Strontium (Sr), Terbium (Tb), Thulium (Tm), Tin (Sn), Titanium (Ti
- the anti-alpha-synuclein antibody of the invention may be labelled with a fluorescent label, a chemiluminescent label, a paramagnetic label, a radioisotopic label or an enzyme label.
- the labelled antibody may be used in detecting or measuring the presence or amount of said alpha-synuclein in the brain of a subject.
- This method may comprise the detection or measurement of in vivo imaging of anti-alpha-synuclein antibody bound to said alpha-synuclein and may comprises ex vivo imaging of said anti-alpha-synuclein antibody bound to said alpha-synuclein.
- the invention relates to an expression vector encoding one or more polypeptide chains of an antibody of the invention or an antigen-binding fragment thereof.
- Such expression vectors may be used for recombinant production of the antibodies and antigen-binding fragments of the invention.
- An expression vector in the context of the present invention may be any suitable DNA or RNA vector, including chromosomal, non-chromosomal, and synthetic nucleic acid vectors (a nucleic acid sequence comprising a suitable set of expression control elements).
- suitable DNA or RNA vector including chromosomal, non-chromosomal, and synthetic nucleic acid vectors (a nucleic acid sequence comprising a suitable set of expression control elements).
- examples of such vectors include derivatives of SV40, bacterial plasmids, phage DNA, baculovirus, yeast plasmids, vectors derived from combinations of plasmids and phage DNA, and viral nucleic acid (RNA or DNA) vectors.
- an anti-alpha-synuclein antibody-encoding nucleic acid is comprised in a naked DNA or RNA vector, including, for example, a linear expression element (as described in, for instance, Sykes and Johnston, Nat Biotech 12, 355-59 (1997 )), a compacted nucleic acid vector (as described in for instance US 6,077,835 and/or WO 00/70087 ), a plasmid vector such as pBR322, pUC 19/18, or pUC 118/119, a "midge" minimally-sized nucleic acid vector (as described in, for instance, Schakowski et al., Mol Ther 3, 793-800 (2001 )), or as a precipitated nucleic acid vector construct, such as a CaPO 4 -precipitated construct (as described in, for instance, WO 00/46147 , Benvenisty and Reshef, PNAS USA 83, 9551-55 (1986 ), Wigler
- the vector is suitable for expression of anti-alpha-synuclein antibodies or antigen-binding fragments thereof in a bacterial cell.
- expression vectors such as BlueScript (Stratagene), plN vectors ( Van Heeke & Schuster, J Biol Chem 264, 5503-5509 (1989 ), pET vectors (Novagen, Madison, WI) and the like).
- An expression vector may also or alternatively be a vector suitable for expression in a yeast system. Any vector suitable for expression in a yeast system may be employed. Suitable vectors include, for example, vectors comprising constitutive or inducible promoters such as alpha factor, alcohol oxidase and PGH (reviewed in: F. Ausubel et al., ed. Current Protocols in Molecular Biology, Greene Publishing and Wiley InterScience New York (1987 ), Grant et al., Methods in Enzymol 153, 516-544 (1987 ), Mattanovich, D. et al. Methods Mol. Biol. 824, 329-358 (2012 ), Celik, E. et al. Biotechnol. Adv.
- constitutive or inducible promoters such as alpha factor, alcohol oxidase and PGH
- anti-alpha-synuclein antibody-encoding nucleic acids may comprise or be associated with any suitable promoter, enhancer, and other expression-facilitating elements.
- suitable promoter, enhancer, and other expression-facilitating elements include strong expression promoters (e.g., human CMV IE promoter/enhancer as well as RSV, SV40, SL3-3, MMTV, and HIV LTR promoters), effective poly (A) termination sequences, an origin of replication for plasmid product in E. coli, an antibiotic resistance gene as selectable marker, and/or a convenient cloning site (e.g., a polylinker).
- Nucleic acids may also comprise an inducible promoter as opposed to a constitutive promoter such as CMV IE (the skilled artisan will recognize that such terms are actually descriptors of a degree of gene expression under certain conditions).
- the antibodies of antigen-binding fragments thereof of the present invention may be produced in different cell lines, such as a human cell line, a mammal non-human cell line, and insect cell line, for example a CHO cell line, HEK cell line, BHK-21 cell line, murine cell line (such as a myeloma cell line), fibrosarcoma cell line, PER.C6 cell line, HKB-11 cell line, CAP cell line and HuH-7 human cell line ( Dumont et al, 2015, Crit Rev Biotechnol. Sep 18:1-13 ., the contents which is included herein by reference).
- insect cell line for example a CHO cell line, HEK cell line, BHK-21 cell line, murine cell line (such as a myeloma cell line), fibrosarcoma cell line, PER.C6 cell line, HKB-11 cell line, CAP cell line and HuH-7 human cell line ( Dumont et al, 2015, Crit Rev Biotechnol. Sep 18
- the invention relates to a recombinant eukaryotic or prokaryotic host cell, such as a transfectoma, which produces an antibody or an antigen-binding domain thereof of the invention as defined herein or a bispecific molecule of the invention as defined herein.
- host cells include yeast, bacteria, and mammalian cells, such as CHO or HEK cells.
- the present invention provides a cell comprising a nucleic acid stably integrated into the cellular genome that comprises a sequence coding for expression of an anti-alpha-synuclein antibody of the present invention or an antigen-binding fragment thereof.
- the present invention provides a cell comprising a non-integrated nucleic acid, such as a plasmid, cosmid, phagemid, or linear expression element, which comprises a sequence coding for expression of an anti-alpha-synuclein antibody of the invention.
- a non-integrated nucleic acid such as a plasmid, cosmid, phagemid, or linear expression element, which comprises a sequence coding for expression of an anti-alpha-synuclein antibody of the invention.
- the invention relates to a method for producing an anti-alpha-synuclein antibody of the invention, said method comprising the steps of a) culturing a hybridoma or a host cell of the invention as described herein above, and b) purifying the antibody of the invention from the culture media.
- the invention relates to a preparation that, as such term is used herein, comprises an anti-alpha-synuclein antibody as defined herein, and that is substantially free of naturally-arising antibodies that are either not capable of binding to alpha-synuclein or that do not materially alter the anti-alpha-synuclein functionality of the preparation.
- a preparation does not encompass naturally-arising serum, or a purified derivative of such serum, that comprises a mixture of an anti-alpha-synuclein antibody and another antibody that does not alter the functionality of the anti-alpha-synuclein antibody of the preparation; wherein such functionality is selected from the group consisting of:
- the invention particularly relates to preparations of such an anti-alpha-synuclein antibody having a structural change in its amino acid sequence (in any of its CDRs, variable domains, framework residues and/or constant domains) relative to the structure of a naturally-occurring anti-alpha-synuclein antibody, wherein said structural change causes the anti-alpha-synuclein monoclonal antibody to exhibit a markedly altered functionality (i.e., more than a 20% difference, more than a 40% difference, more than a 60% difference, more than an 80% difference, more than a 100% difference, more than a 150% difference, more than a 2-fold difference, more than a 4-fold difference, more than a 5-fold difference, or more than a 10-fold difference in functionality) relative to the functionality exhibited by said naturally-occurring anti-alpha-synuclein antibody; wherein such functionality is:
- substantially free of naturally-arising antibodies refers to the complete absence of such naturally-arising antibodies in such preparations, or of the inclusion of a concentration of such naturally-arising antibodies in such preparations that does not materially affect the alpha-synuclein-binding properties of the preparations.
- An antibody is said to be “isolated” if it has no naturally-arising counterpart or has been separated or purified from components which naturally accompany it.
- naturally-arising antibodies refers to antibodies (including naturally-arising autoantibodies) elicited within living humans or other animals, as a natural consequence to the functioning of their immune systems.
- the preparations of the present invention do not exclude, and indeed explicitly encompass, such preparations that contain an anti-alpha-synuclein antibody and a deliberately added additional antibody capable of binding to an epitope that is not possessed by alpha-synuclein.
- Such preparations particularly include embodiments thereof wherein the preparation exhibits enhanced efficacy in treating synucleinopathies such as Parkinson's disease (including idiopathic and inherited form of Parkinson's disease), Gaucher's Disease, Diffuse Lewy Body Disease (DLBD), Lewy body variant of Alzheimer's disease (LBV), Combined Alzheimer's and Parkinson disease, pure autonomic failure and multiple system atrophy.
- synucleinopathies such as Parkinson's disease (including idiopathic and inherited form of Parkinson's disease), Gaucher's Disease, Diffuse Lewy Body Disease (DLBD), Lewy body variant of Alzheimer's disease (LBV), Combined Alzheimer's and Parkinson disease, pure autonomic failure and multiple system atrophy.
- the invention relates to a pharmaceutical composition
- a pharmaceutical composition comprising:
- compositions may be formulated with pharmaceutically acceptable carriers or diluents as well as any other known adjuvants and excipients in accordance with conventional techniques such as those disclosed in Remington: The Science and Practice of Pharmacy, 22nd Edition, Gennaro, Ed., Mack Publishing Co., Easton, PA, 2013 .
- compositions should be suitable for the chosen compound of the present invention and the chosen mode of administration. Suitability for carriers and other components of pharmaceutical compositions is determined based on the lack of significant negative impact on the desired biological properties of the chosen compound or pharmaceutical composition of the present invention (e.g., less than a substantial impact (10% or less relative inhibition, 5% or less relative inhibition, etc.)) on epitope binding.
- a pharmaceutical composition of the present invention may also include diluents, fillers, salts, buffers, detergents (e.g., a non-ionic detergent, such as Tween-20 or Tween- 80), stabilizers (e.g., sugars or protein-free amino acids), preservatives, tissue fixatives, solubilizers, and/or other materials suitable for inclusion in a pharmaceutical composition.
- the diluent is selected to not to affect the biological activity of the combination. Examples of such diluents are distilled water, physiological phosphate-buffered saline, Ringer's solutions, dextrose solution, and Hank's solution.
- compositions or formulation may also include other carriers, or non-toxic, nontherapeutic, non-immunogenic stabilizers and the like.
- the compositions may also include large, slowly metabolized macromolecules, such as proteins, polysaccharides like chitosan, polylactic acids, polyglycolic acids and copolymers (e.g., latex functionalized sepharose, agarose, cellulose, and the like), polymeric amino acids, amino acid copolymers, and lipid aggregates (e.g., oil droplets or liposomes).
- the actual dosage levels of the active ingredients in the pharmaceutical compositions of the present invention may be varied so as to obtain an amount of the active ingredient which is effective to achieve the desired therapeutic response for a particular patient, composition, and mode of administration.
- the selected dosage level will depend upon a variety of pharmacokinetic factors including the activity of the particular compositions of the present invention employed, or the amide thereof, the route of administration, the time of administration, the rate of excretion of the particular compound being employed, the duration of the treatment, other drugs, compounds and/or materials used in combination with the particular compositions employed, the age, sex, weight, condition, general health and prior medical history of the patient being treated, and like factors well known in the medical arts.
- the pharmaceutical composition may be administered by any suitable route and mode, including: parenteral, topical, oral or intranasal means for prophylactic and/or therapeutic treatment.
- a pharmaceutical composition of the present invention is administered parenterally.
- parenteral administration and “administered parenterally” as used herein means modes of administration other than enteral and topical administration, usually by injection, and include epidermal, intravenous, intramuscular, intraarterial, intrathecal, intracapsular, intraorbital, intracardiac, intradermal, intraperitoneal, intratendinous, transtracheal, subcutaneous, subcuticular, intraarticular, subcapsular, subarachnoid, intraspinal, intracranial, intrathoracic, epidural and intrasternal injection and infusion. Additional suitable routes of administering a compound of the present invention in vivo and in vitro are well known in the art and may be selected by those of ordinary skill in the art.
- pharmaceutical composition is administered by intravenous or sub
- Pharmaceutically acceptable carriers include any and all suitable solvents, dispersion media, coatings, antibacterial and antifungal agents, isotonicity agents, antioxidants and absorption delaying agents, and the like that are physiologically compatible with a compound of the present invention.
- aqueous and non-aqueous carriers examples include water, saline, phosphate buffered saline, ethanol, dextrose, polyols (such as glycerol, propylene glycol, polyethylene glycol, and the like), and suitable mixtures thereof, vegetable oils, such as olive oil, corn oil, peanut oil, cottonseed oil, and sesame oil, carboxymethyl cellulose colloidal solutions, tragacanth gum and injectable organic esters, such as ethyl oleate, and/or various buffers.
- Other carriers are well known in the pharmaceutical arts.
- Pharmaceutically acceptable carriers include sterile aqueous solutions or dispersions and sterile powders for the extemporaneous preparation of sterile injectable solutions or dispersion.
- sterile aqueous solutions or dispersions and sterile powders for the extemporaneous preparation of sterile injectable solutions or dispersion.
- the use of such media and agents for pharmaceutically active substances is known in the art. Except insofar as any conventional media or agent is incompatible with the active compound, use thereof in the pharmaceutical compositions of the present invention is contemplated.
- compositions of the present invention may also comprise pharmaceutically acceptable antioxidants for instance (1) water soluble antioxidants, such as ascorbic acid, cysteine hydrochloride, sodium bisulfate, sodium metabisulfite, sodium sulfite and the like; (2) oil-soluble antioxidants, such as ascorbyl palmitate, butylated hydroxyanisole (BHA), butylated hydroxytoluene (BHT), lecithin, propyl gallate, alpha- tocopherol, and the like; and (3) metal chelating agents, such as citric acid, ethylenediamine tetraacetic acid (EDTA), sorbitol, tartaric acid, phosphoric acid, and the like.
- water soluble antioxidants such as ascorbic acid, cysteine hydrochloride, sodium bisulfate, sodium metabisulfite, sodium sulfite and the like
- oil-soluble antioxidants such as ascorbyl palmitate, butylated hydroxyanisole (BHA), butylated
- Such carriers may include gelatin, glyceryl monostearate, glyceryl distearate, biodegradable, biocompatible polymers such as ethylene vinyl acetate, polyanhydrides, polyglycolic acid, collagen, polyorthoesters, and polylactic acid alone or with a wax, or other materials well known in the art. Methods for the preparation of such formulations are generally known to those skilled in the art. See, e.g., Sustained and Controlled Release Drug Delivery Systems, J. R. Robinson, ed., Marcel Dekker, Inc., New York, 1978 .
- the compounds of the present invention may be formulated to ensure proper distribution in vivo.
- Pharmaceutically acceptable carriers for parenteral administration include sterile aqueous solutions or dispersions and sterile powders for the extemporaneous preparation of sterile injectable solutions or dispersion.
- the use of such media and agents for pharmaceutically active substances is known in the art. Except insofar as any conventional media or agent is incompatible with the active compound, use thereof in the pharmaceutical compositions of the present invention is contemplated. Supplementary active compounds may also be incorporated into the compositions.
- Dosage regimens in the above methods of treatment and uses described herein are adjusted to provide the optimum desired response (e.g., a therapeutic response). For example, a single bolus may be administered, several divided doses may be administered over time or the dose may be proportionally reduced or increased as indicated by the exigencies of the therapeutic situation.
- Parenteral compositions may be formulated in dosage unit form for ease of administration and uniformity of dosage.
- Dosage unit form as used herein refers to physically discrete units suited as unitary dosages for the subjects to be treated; each unit contains a predetermined quantity of active compound calculated to produce the desired therapeutic effect in association with the required pharmaceutical carrier.
- the treatment is chronic, and is preferably for at least 2 weeks, such as at least for 1 month, 6, months, 1 year or more.
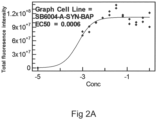
- the protein solution was incubated 170 ⁇ l aliquots in 96 well plate with a 70 ⁇ m diameter ceramic bead in each well at 200 rpm in Vortemp 56 shaker incubator (Labnet International, Edison, NJ, USA), at 37°C for 7 days, and the formation of fibrils was followed by adding thioflavin T and measuring fluorescence in one of the wells.
- the recombinant alpha-synuclein containing amino acids 1-60 was dissolved in water to give a concentration of 1 mg/ml.
- A-Syn-AAKK-BAP A-Syn-BAAK-BAP, A-Syn-BBAA-BAP, A-Syn-BBKK-BAP (SEQ ID Nos:11-14) were produced. These constructs contained linear stretches of human alpha-synuclein (A), human Beta-synuclein (B) and chicken alpha-synuclein (K). Gene were cloned containing a Biotin Acceptor Peptide (BAP) tag fused to the C-terminus of the ligands in order to facilitate site specific biotinylation of each of the ligands.
- BAP Biotin Acceptor Peptide
- Mammalian expression vectors were constructed carrying the different alpha-synuclein BAP tag fusion constructs (ASynBAP).
- the ligands were expressed in HEK 293 cells using transient transfection (Genmab A/S).
- HuMab-Synuclein were derived from the immunizations of HuMAb mouse strain HCo17-BALB/c and HCo12-BALB/c mice, double knock out for the mouse immunoglobulin (Ig) heavy and mouse kappa light chain, which prevents the expression of antibodies that are completely murine (human monoclonal antibody; Medarex Inc., San Jose, CA, USA).
- the various mouse strains were made transgenic by the insertion of human Ig heavy and human Ig kappa light chain loci and differ in the number of human VH (variable domain of heavy chain) and VL (variable domain of light chain) genes.
- IP intraperitoneally
- SC subcutaneously
- mice were additionally boosted twice intravenously (IV) with 10 ⁇ g alpha-synuclein immunogen protein in 100 ⁇ L PBS, four and three days before fusion.
- CFA complete Freund's adjuvant
- IFA incomplete Freund's adjuvant
- the immunization protocols are shown in Fig 1 .
- Antibody 285 came from an immunization protocol where Human ⁇ -Synuclein-monomer 1-140 was used for the first 4 immunizations. If there was no titer, the immunization was continued with fibrils (ip/sc), otherwise it was continued with monomer.
- HuMAb mice with sufficient antigen-specific titer development as defined above were sacrificed and the spleen and lymph nodes flanking the abdominal aorta and caval vein were collected. Fusion of splenocytes and lymph node cells with a mouse myeloma cell line was done by electrofusion using a CEEF 50 Electrofusion System (Cyto Pulse Sciences, Glen Burnie, MD, USA), essentially according to the manufacturer's instructions.
- Fused cells were seeded in fusion medium containing 10% Fetal Clone I Bovine serum (Perbio), 1 mM sodium pyruvate (Cambrex), 0.5 U/mL penicillin, 0.5 U/mL streptomycin (Cambrex), 50 ⁇ M 2-mercaptoethanol (Invitrogen), 600 ng/mL interleukin 6 (IL-6) (Strathmann), 1 x HAT (Sigma) and 0.5 mg/mL kanamycin (Invitrogen) in HyQ mADCF-Mab (Perbio).
- the screening of anti-alpha-synuclein antibodies was performed using a high throughput suspension ELISA format using automated liquid handling systems (Genmab A/S).
- the reading of the plates was performed by two systems, the FMAT 8200 from Applied Biosystems was used to read 384 well plates and the ImageXpress Velos Cytometer from Molecular Devices was used to read the 1536 well plates.
- the sera or supernatant potentially containing alpha-synuclein specific antibodies were added to the beads to allow binding to alpha-Synuclein and/or alpha-synuclein derived constructs.
- the binding of the anti-alpha-synuclein antibodies is detected using a fluorescent conjugate, DyLight649 conjugated goat antihuman IgG, Fc specific.
- an anti-alpha-synuclein sera pool is used as a negative control in the 384 well format titer screening while human ChromPure IgG is used in the 1536 well format 8-bead based assay.
- Hybridoma cells from the best primary wells were seeded in semisolid medium made from 40% CloneMedia (Genetix, Hampshire, UK) and 60% HyQ 2x complete medium (Hyclone, Waltham, USA).
- a well of a Genetix black 6-well plate was seeded.
- 25 sub clones were picked, using the ClonePix system (Genetix).
- the sub clones were picked in harvest medium.
- the supernatants of the sub clones were screened again for Synuclein-specific human IgG binding and the human IgG concentration was measured using Octet (Fortebio, Menlo Park, USA).
- RNA was prepared from 0.2 to 5x106 hybridoma cells and 5'-RACE-Complementary DNA (cDNA) was prepared from 100 ng total RNA, using the SMART RACE cDNA Amplification kit (Clontech), according to the manufacturer's instructions.
- VH and VL coding regions were amplified by PCR and cloned directly, in frame, in the p33G1f and p33Kappa expression vectors (containing the human IgG1/kappa constant domain encoding sequences), by ligation independent cloning ( Aslanidis, C. and P.J. de Jong, Nucleic Acids Res 1990;18(20): 6069-74 ).
- Comparator antibody 9E4 was generated based on the VH and VL sequence derived from hybridoma PTA-8221 ( US patent 20080175838 ) (SEQ ID NO 42 and 43)
- Antibodies were produced by transfection in HEK293 6E cells using the pTT5 vectors and PElpro as a transient transfection agent (National Research Council of Canada). In short, The heavy and light chains were transfected into HEK293 cells using PElpro (VWR), and cells were supplemented with TN1 (Sigma) 24 hours after transfection. Cells were grown until the viability approached 50%, and yield of antibody measured by easy IgG titre (Thermo). Culture supernatant was filtered over 0.2 ⁇ m dead-end filters, loaded on 5 mL Protein A columns (rProtein A FF, Amersham Bioscience) and eluted with 0.1 M citric acid-NaOH, pH 3.
- the eluate was immediately neutralized with 2M Tris-HCl, pH 9 and dialyzed to 12.6 mM NaH 2 PO 4 , 140 mM NaCl, pH 7.4 (B.Braun), O/N. After dialysis, samples were sterile-filtered over 0.2 ⁇ m dead-end filters. Purity was determined by SDS-PAGE and concentration was measured by nephelometry and absorbance at 280 nm. Purified antibodies were aliquoted and stored at -80°C.
- BIAcore ® 3000 Real time binding of the antibodies to alpha-synuclein was measured using a BIAcore ® 3000.
- a capture surface was prepared by amine-coupling a polyclonal rabbit Anti-Mouse antibody (part of Mouse Antibody Capture Kit, GE Healthcare, Cat. no: BR-1008-38) in first flow cell (Fc1) and second flow cell (Fc2) of a CM5 chip (BIAcore ® ).
- the mouse antibody was captured in Fc2 at the concentration required to achieve a ligand level of around 500RU.
- the baseline was allowed to stabilize for 10min before injecting analyte (ASynBAP) in Fc1-2 at 30 ⁇ l/min.
- ASynBAP was run at 100-3200nM and 25-3200RU, respectively.
- the peptide arrays were incubated with a 1/1000 dilution of an antibody peroxidase conjugate (SBA, cat. nr. 2010-05) for one hour at 25°C. After washing, the peroxidase substrate 2,2'-azino-di-3-ethylbenzthiazoline sulfonate (ABTS) and 2 ⁇ l/ml of 3 percent H2O2 were added. After one hour, the color development was measured. The color development was quantified with a charge coupled device (CCD) - camera and an image processing system. For data processing the values were obtained from the CCD camera range from 0 to 3000 mAU, similar to a standard 96-well plate ELISA-reader.
- SBA antibody peroxidase conjugate
- ABTS 2,2'-azino-di-3-ethylbenzthiazoline sulfonate
- H2O2 2 ⁇ l/ml of 3 percent H2O2
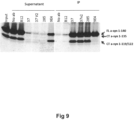
- Example 4 Immunoprecipitation of alpha-synuclein from human brain homogenates of cingulate cortex from patients with dementia with Lewy bodies
- the yield of the immunoprecipitation was visualized by Western blotting with detection mouse monoclonal anti-human alpha-synuclein antibody, (4B12, Thermo Scientific).
- the patterns of bands representing different molecular weight forms of alpha-synuclein being pulled down differ between the 37, 37 variant 2 and 285 antibodies and the comparator antibody 9E4 in that the 37, 37v2 and 285 antibody can immunoprecipitate the major alpha-synuclein species, the full length alpha-synuclein (FL asyn 1-140) and the C-terminal terminal truncated species (1-135 and 1-119/122), while antibody 9E4 cannot immunoprecipitate the truncated species 1-119/122.
- Figure 9 .
- Example 5 Inhibition of protease truncation of alpha-synuclein fibrils by antibodies in cell culture
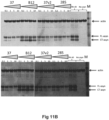
- Recombinant alpha-synuclein monomers and fibrils can be taken up by primary neurons in culture. As shown schematically in Figure 10 , after uptake of the alpha-synuclein in neurons, it can be processed by intracellular proteases, such as Calpain I, with the major protease sensitive site at amino acid 119/122. To investigate truncation of alpha-synuclein by proteases mouse primary cortical neurons were prepared as described in Elvang et al. 2009 ( Elvang et al.. J Neurochem. 2009;110(5):1377-87 ) and treated with cytarabine on DIV3 (3 days in vitro) to inhibit astrocyte growth.
- proteases such as Calpain I
- the neurons were treated with sonicated (5 min at 50 % power in cup horn sonicator) pre-formed alpha-synuclein fibrils (PFFs) at an end concentration of 0.7 ⁇ M alone or together with antibodies in the indicated concentrations. After 24 hours of incubation, the media was harvested and the cells were lysed. Western Blots was run on both media and cell lysate using the 4B12 antibody ( Fig 11A ) (Pierce MA1-90346) and a secondary anti-mouse antibody. After probing with 4B12 + anti-mouse, the blots were stripped and reprobed with an anti-human-IgG antibody.
- alpha-synuclein can be cleaved by Calpain-1.
- the cleavage site of Calpain-1 for fibrillized alpha-synuclein has been found to be in the region 114-122 ( Mishizen-Eberz, J of Neurochem, 86, 836-847, 2003 ).
- the epitope of 285 overlaps with the epitope of 37 and it would also be expected to inhibit the protease cleavage.
- the amino acid sequence of 37v2 only differs from 37 at one amino acid in CDR and has similar binding as 37, so it would also be expected to inhibit protease cleavage in similar manner to 37.
- Alpha-synuclein on Western Blots were detected with 1904/4B12 antibody (Abcam), which has an epitope in the region 103-108 and therefore binds to both the FL and the C-terminally truncated alpha-synuclein ( Fig. 11B ).
- Abcam 1904/4B12 antibody
- both GM37, 37v2 and GM285 have a dose dependent inhibition of protease cleavage, with almost complete inhibition of the cleavage at high concentration of the antibody.
- mouse primary cortical neurons prepared as above are plated in 96 well plates (15,000 cells per well).
- DIV in vitro culture
- 50% of media is changed and supplemented with cytosine arabinoside (final conc. of 1uM).
- DIV 6 half of the media is changed with glia conditioned media along with alpha synuclein fibrillary material, either crude fibril seeds or pure seeds.
- the crude fibril seeds are made from recombinant monomeric human alpha-synuclein, which was isolated from bacteria and the monomers were filtered through an Amicon Ultra 100.000 cut off filter (Millipore cat. No UFC510096) and adjusted to concentration of 1mg/ml in PBS, pH 7.4.
- the monomer solution was incubated in thermomixer at 37C with continuous mixing (800rpm) until plateu is reached (evaluated by daily measures with Thioflavin S). To minimize evaporation a drop of mineral oil was added to cover the solution.
- the total time for incubation was 5-7 days,
- the pure seeds are made from crude fibril seeds that are centrifuged to purify them and the aggregated pellet is resuspended in fresh PBS and sonicated.
- the antibodies are added once on DIV 6 along with alpha-synuclein crude seeds.
- Half of the media in the primary neurons is replaced with glia conditioned media every week to maintain them up to DIV21.
- the neurons are fixed and stained for Phospho-synuclein using a rabbit antibody specific for phosphorylation of alpha-synuclein at amino acid S129 (abcam 51253), followed by a fluorescently labelled anti-rabbit antibody, fluorescence is quantified using automated fluorescent microscopy, Cellomics Arrayscan.
- Nuclei were detected in one channel and defined the number of valid cells.
- Phosphorylated alpha-synuclein spots were detected in another channel in a pre-defined ring-formed area surrounding the nucleus, thus representing the cytoplasm of the cells. The average number of spots per cell was calculated.
- Example of cell staining is shown in Fig 12A .
- Phosphorylated alpha synuclein spots are not seen in untreated neurons. Neurons incubated with crude or pure seeds (1-10ng per well) induce phosphorylation of alpha synuclein ( Fig. 12A ). In neurites, phosphorylated synuclein appears as spots or punctate and some of the phospho-synuclein in the neurites appear elongated.
- cells were harvested in phosphate buffered saline solution (PBS) and centrifuged. Pellet was resuspended in 1% triton buffer with protease inhibitors. Samples were kept on ice for 15 min. followed by sonication. The samples were centrifuged at 100,000x g for 30 min. at 4C. The supernatant is collected and labelled as soluble fraction. The pellet was washed once in triton buffer and resuspended in 1% SDS buffer followed by sonication. Samples were centrifuged again at 100,000xg for 30 min. The supernatant is collected as insoluble fraction.
- PBS phosphate buffered saline solution
- the protein concentrations were measured and samples were run on 4-12% SDS_PAGE gel, blotted on membranes and alpha synuclein and phosphorylated alpha synuclein (S129P) are detected by 4B12/1904 antibody (Thermo scientific: MA1-90346-human synuclein), S129P-asyn antibody (abcam 51253) and mouse synuclein antibody (cell signalling- D37A6), respectively.
- 4B12/1904 antibody Thermo scientific: MA1-90346-human synuclein
- S129P-asyn antibody abcam 51253
- mouse synuclein antibody cell signalling- D37A6
- Figure 12 B shows the Western blot of the soluble and insoluble fraction from the primary neurons with and without crude seeds. As can be seen from Figure 12B the addition of the seeds lead to accumulation of endogenous mouse alpha-synuclein and p-S129-alpha-synuclein and multimers of phosphorylated mouse alpha-synuclein in the insoluble fraction of the cells.
- alpha synuclein synuclein seeds were used at conc. of 6.6nM (10ng/ well). Different concentration of antibody and alpha- synuclein seeds were added together on DIV 6, to make a dose response (starting from highest antibody conc. at 133nM down to 133 pM). The neurons were again fixed and stained for Phospho-synuclein (abcam 51253) and fluorescence from cells was quantified using automated fluorescent microscopy, Cellomics arrayscan. The spots/puncta per cell were counted in Cellomics arrayscan.
- both antibody 37, 37v2 and antibody 285 reduced alpha synuclein phosphorylation in neurons in a dose dependent manner with similar maximal inhibition for 37, 37v2 and 285 (around 70-75%) and EC50 around 5 nM.
- F28-snca transgenic and age-matched control male mice (CRO breeding, Taconic Europe A/S) aged 4 to 6 months were single-housed in controlled temperature (22 ⁇ 1.5°C) and humidity conditions (55-65%) and kept in a 12:12 hour light/dark cycle (lights on at 06:00h). Food and water were available ad libitum.
- mice were anesthetized with an intraperitoneal (i.p.) injection of urethane (1.2 g/kg). Mice were then mounted in a stereotaxic frame, their temperature adjusted to 37.5°C via a heating pad, and the skull was exposed.
- i.p. intraperitoneal
- a platinum wire was placed in the frontal bone to act as a reference, and an additional hole was drilled for insertion of the recording and stimulating electrodes in the hippocampus, at the following coordinates according to the atlas of Paxinos and Franklin ( Paxinos and Franklin's the Mouse Brain in Stereotaxic Coordinates, 4th Edition, 2001 ): recording, 1.5-1.7 mm posterior to Bregma, 1.0-1.2 mm lateral to the midline, 1.4-1.7 mm below the surface of the brain; stimulation, 1.8-2.0 mm posterior to Bregma, 1.5-1.7 mm lateral to the midline, 1.5-1.7 mm below the surface of the brain. Animals were left in the stereotaxic frame through the whole duration of the recordings and their level of anesthesia was regularly checked.
- fEPSP Field potentials
- basal synaptic transmission was assessed by the relationship between stimulation intensity and slope of the evoked fEPSP (input-output relationship).
- the different intensities of stimulation were 0, 25, 50, 75, 100, 150, 200, 300, 400, and 500 ⁇ A, and were applied successively in increasing order, with 2 to 3 repeats for each intensity.
- Basal synaptic transmission was found to be significantly impaired in F28-snca transgenic compared to age-matched control mice.
- the identified impairments in basal synaptic transmission in F28-snca transgenic mice were used to test the GM37, GM285 and comparator h9E4 for their ability to block the alpha synuclein mediated effect.
- Basal synaptic transmission in GM37-treated transgenic mice was not significantly different from basal synaptic transmission in PBS-treated littermates, indicating a full reversal of the impairment ( Fig 15 ).
- GM285 also induced a significant reversal of the impairment in basal synaptic transmission in F28-snca transgenic mice ( Fig 16 ). Basal synaptic transmission in GM285-treated transgenic mice was not significantly different from basal synaptic transmission in PBS-treated littermates, indicating a full reversal of the impairment.
- Example 8 Microdialysis to assess human alpha-synuclein in the brain of awake freely moving animals
- the push-pull microdialysis method was used to assess the levels of human alpha-synuclein in brain interstitial fluid (ISF). Mice were single-housed in controlled temperature (22 ⁇ 1.5°C) and humidity conditions (55-65%) and kept on a 12:12 hour light/dark cycle (lights on at 06:00h). Food and water were available ad libitum. The current study was performed in the hippocampus of F28-snca transgenic mice (50-54 weeks old).
- ISF brain interstitial fluid
- mice were anaesthetized with isoflurane and an intracerebral guide cannula (CMA) was stereotaxically implanted into the brain, positioning the microdialysis probe in the hippocampus (co-ordinates of probe tip: 3.1 mm posterior and 2.8 mm lateral from bregma, and 1.3 mm relative dura mater) according to the atlas of Paxinos and Franklin 2001.
- Anchor screws and acrylic cement were used for the fixation of the guide cannulas. After implantation of the cannula mice were allowed to recover from the surgery for 2-3 days before dialysis.
- a 2-mm, 1000 kDa cut-off CMA probe was inserted through the guide cannula.
- a probe was connected to a microdialysis peristaltic pump with two channels (MAB20; Microbiotech) and operated in push-pull mode.
- the inlet tubing of the microdialysis probe was connected to a peristaltic pump perfusing the probe with artificial cerebrospinal fluid (aCSF; in mM: 147 NaCl, 2.7 KCl, 1.2 CaCl 2 , 0.85 MgCl 2 ).
- the peristaltic pump was also connected to the outlet tubing in order to prevent perfusion fluid loss from the probe, by pulling the fluid through the tubing.
- the average of the two-three basal values (4h-6h) prior to antibody treatment was taken as baseline and set to 100% for each animal. Data was evaluated using two-way analysis of variance (ANOVA) with repeated measures to evaluate statistical relevance.
- the administration of GM37 induced a larger reduction in human alpha-synuclein in the hippocampus of F28 mice compared to both the comparator antibody, human 9E4, and the isotype control (anti-HEL). ( Fig. 17 ).
- Targeted overexpression of human alpha-synuclein to dopaminergic neurons in the rat midbrain can be achieved using a recombinant adeno-associated viral vector (rAAV) and is associated with a progressive loss of dopaminergic cells in the substantia nigra as well as motor impairments.
- rAAV adeno-associated viral vector
- mice were dosed with GM 37 at 30 mg/kg i.p, GM 3715 mg/kg i.v. , or vehicle ip (PBS).One day later the mice were anesthetized and stereotactically injected in one hemisphere with 2 ul of recombinant human alpha-Syn crude seeds, made as described previously (Example 6) (total of 2 ⁇ g crude seeds per animal).
- mice received weekly i.p. or i.v. injections of antibodies until sacrifice at 45 days. Groups of 15 mice/group were dosed either iv w. GM37 15mg/kg, ip with GM37 30mg/kg, or PBS (10 ml/kg) ip once weekly.
- Plasma and CSF samples were analysed to determine the concentration of Human IgG by MSD.
- mouse anti-human IgG (clone MH16-1 (M1268) was used for capture, plasma or CSF was incubated in the well, followed by a sulfo-TAG goat anti-human as the detection antibody (MSD cat no: R32AJ-1). Plates were analysed from electrochemiluminesence by MSD.
- the antibody levels in plasma are shown in Figure 20B and show a dose dependent increase in antibody plasma concentration and accumulation of antibody in plasma during the six weeks.
- the antibody levels in csf are shown in Figure 20C , and show that around 0.1% of antibody level in plasma can be measured in csf.
- mice were anesthetized, transcardially perfused with PBS, followed by perfusion with neutral buffered paraformaldehyde (4%). The brains were removed and incubated overnight for post-fixation in neutral buffered paraformaldehyde. Immunohistochemistry was performed on 45 ⁇ m thick serial sections by Neuroscience associates. Briefly, Using MultiBrain ® technology, up to 25 mouse brains were embedded together per block, into 3 blocks, freeze-sectioned at 45 ⁇ m thickness in the coronal plane, and collected into cups containing antigen preserve solution.
- Quantitation of pSyn pathology was performed by manual counting immunoreactive positive cells from images at 10 x magnification from 5-7 sections covering the entire substantia nigra from every sixth section. The counting was performed blinded. Cell counts in amygdala and nigra were analysed by a one-way ANOVA followed by Bonferoni t-test, where the effect of GM37 antibody was compared to PBS treatment.
- treatment with antibody GM37 reduced the number of intra-cellular inclusions in Substantia Nigra significantly when compared to PBS control, with either ip or iv treatment.
- the data shows that antibody GM37 could have therapeutic effect in PD by blocking entry of extracellular pathological alpha-synuclein into neurons, by blocking its propagation between neurons and/or facilitating clearance from the ISF by uptake into microglia.
- treatment with antibody GM37 could have a therapeutic effect on loss of dopaminergic cells and development of motor deficits in PD.
- Example 11 Manufactability of GM37 and GM37 variants
- the anti-alpha-synuclein antibodies are produced in mammalian cell culture under conditions that mimic the production conditions that will be used for producing clinical grade material for use in patients. It is well known that proteins produced in this manner undergo post-translational modifications that can impact both therapeutic potency of the antibody as well as biophysical attributes that affect the stability of the antibody over time. Empirical knowledge ascertained from decades of studies identified a set of post-translational modifications known to provide risk for the developability of a specific molecule. These post-translational modifications have been shown to correlate with amino acid strings present in the primary sequence of the heavy and light chain proteins. Algorithms have been generated that can identify these sequences and determine the potential risk they will have on the manufacturability and developability of a therapeutic antibody.
- Amino acid residues 54 is an asparagine(N) followed by a glycine(G) at position 55.
- the N54 is at high risk for spontaneous deamidation.
- To mitigate this risk we generated a set of 3 variants that replace the asparagine(N) with serine(S), glutamine(Q) or histidine(H). All 3 variants were produced in mammalian cell culture using transient transfection methods (example 1.5). All 3 variants showed similar expression and purification properties as GM37wt. ( Fig. 23 ).
- transient transfections were performed using CHOK1SV GS-KO cells which had been in culture for minimum 2 weeks. Cells were sub-cultured 24 hours prior to transfection. All transfections were carried out via electroporation using Gene Pulse XCell (Bio-Rad). For each transfection, viable cells were resuspended in pre-warmed CD-CHO media supplemented with 6mM L-glutamine to 2.86x10 7 cells/ml. 40 ⁇ g of each established SGV DNA containing the appropriate heavy and light chains were aliquoted into each cuvette (Bio-Rad, GenePulser cuvette, 0.4 cm gap, 165-2088) and 700 ⁇ l cell suspension added.
- Cells were electroporated at 300V, 900 ⁇ F. Transfected cells were transferred to rep-warmed media in Erlenmeyer flasks and the contents of the cuvettes rinsed twice with prewarmed media were also transferred to the flasks. Transfectant cultures were incubated in a shaking incubator at 36.5°C, 5% CO 2 , 85% humidity, 140 rpm for 6 days. Cell viability was measured at the time of harvest using a Cedex HiRes automated cell counter (Rosche).
- a competition assay was performed using preincubation of a fixed concentration (0.3 ⁇ g/ml) of each of the following antibodies, GM37 (named GM 37wt), GM37 variant1, GM37 variant2 and GM37 variant3 with a range of 0-1000 nM human alpha-synuclein for 60 minutes at room temperature. The remaining unbound antibody was captured and measured on ELISA plates coated with 100 ng/ml of recombinant human alpha-synuclein using an anti-human detection antibody by electrochemiluminesence (MSD, Gathersburg, MD).
- the IC50s of the interaction are 1.9nM, 1.6nM, 2.1nM and 1.4nM for GM37 wt, GM37variant1, GM37variant2 and GM37variant3, respectively (as determined using Prism Graphpad ® ).
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Biomedical Technology (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Neurosurgery (AREA)
- Neurology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Organic Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Immunology (AREA)
- Molecular Biology (AREA)
- Biochemistry (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Hematology (AREA)
- Urology & Nephrology (AREA)
- Microbiology (AREA)
- Physics & Mathematics (AREA)
- Epidemiology (AREA)
- Biophysics (AREA)
- Genetics & Genomics (AREA)
- Psychiatry (AREA)
- Hospice & Palliative Care (AREA)
- Psychology (AREA)
- Pathology (AREA)
- Cell Biology (AREA)
- Biotechnology (AREA)
- Food Science & Technology (AREA)
- Analytical Chemistry (AREA)
- General Physics & Mathematics (AREA)
- Mycology (AREA)
- Endocrinology (AREA)
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| GBGB1512203.9A GB201512203D0 (en) | 2015-07-13 | 2015-07-13 | Agents,uses and methods |
| EP16738736.4A EP3322722B1 (de) | 2015-07-13 | 2016-07-12 | Mittel, verwendung und verfahren zur behandlung von synukleinopathien |
| PCT/EP2016/066476 WO2017009312A1 (en) | 2015-07-13 | 2016-07-12 | Agents, uses and methods for the treatment of synucleinopathy |
Related Parent Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP16738736.4A Division EP3322722B1 (de) | 2015-07-13 | 2016-07-12 | Mittel, verwendung und verfahren zur behandlung von synukleinopathien |
| EP16738736.4A Division-Into EP3322722B1 (de) | 2015-07-13 | 2016-07-12 | Mittel, verwendung und verfahren zur behandlung von synukleinopathien |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP4582144A2 true EP4582144A2 (de) | 2025-07-09 |
| EP4582144A3 EP4582144A3 (de) | 2025-07-16 |
Family
ID=54013851
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP25154059.7A Pending EP4582144A3 (de) | 2015-07-13 | 2016-07-12 | Mittel, verwendungen und verfahren zur behandlung von synukleinopathie |
| EP16738736.4A Active EP3322722B1 (de) | 2015-07-13 | 2016-07-12 | Mittel, verwendung und verfahren zur behandlung von synukleinopathien |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP16738736.4A Active EP3322722B1 (de) | 2015-07-13 | 2016-07-12 | Mittel, verwendung und verfahren zur behandlung von synukleinopathien |
Country Status (40)
| Country | Link |
|---|---|
| US (12) | US10800836B2 (de) |
| EP (2) | EP4582144A3 (de) |
| JP (4) | JP7012004B2 (de) |
| KR (2) | KR102793383B1 (de) |
| CN (3) | CN121736097A (de) |
| AR (1) | AR105336A1 (de) |
| AU (1) | AU2016292896B2 (de) |
| CL (1) | CL2018000075A1 (de) |
| CO (1) | CO2017012941A2 (de) |
| CR (2) | CR20230300A (de) |
| DK (1) | DK3322722T3 (de) |
| DO (1) | DOP2018000013A (de) |
| EA (1) | EA036499B1 (de) |
| EC (1) | ECSP18002645A (de) |
| ES (1) | ES3028882T3 (de) |
| FI (1) | FI3322722T3 (de) |
| GB (1) | GB201512203D0 (de) |
| GE (2) | GEP20207151B (de) |
| HR (1) | HRP20250624T1 (de) |
| HU (1) | HUE071541T2 (de) |
| IL (1) | IL256501B (de) |
| JO (1) | JO3692B1 (de) |
| LT (1) | LT3322722T (de) |
| MA (1) | MA42439B1 (de) |
| MX (2) | MX2018000504A (de) |
| MY (1) | MY194944A (de) |
| NI (1) | NI201800006A (de) |
| PE (1) | PE20181049A1 (de) |
| PH (1) | PH12018500016A1 (de) |
| PL (1) | PL3322722T3 (de) |
| PT (1) | PT3322722T (de) |
| RS (1) | RS66840B1 (de) |
| RU (1) | RU2765303C2 (de) |
| SI (1) | SI3322722T1 (de) |
| SV (1) | SV2018005612A (de) |
| TN (1) | TN2018000005A1 (de) |
| TW (1) | TWI729992B (de) |
| UA (1) | UA125501C2 (de) |
| WO (1) | WO2017009312A1 (de) |
| ZA (1) | ZA201708607B (de) |
Families Citing this family (30)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB201512203D0 (en) * | 2015-07-13 | 2015-08-19 | Lundbeck & Co As H | Agents,uses and methods |
| US11542332B2 (en) | 2016-03-26 | 2023-01-03 | Bioatla, Inc. | Anti-CTLA4 antibodies, antibody fragments, their immunoconjugates and uses thereof |
| CA3043651A1 (en) | 2016-11-15 | 2018-05-24 | H. Lundbeck A/S | Agents, uses and methods for the treatment of synucleinopathy |
| BR112018016717A2 (pt) | 2016-12-16 | 2018-12-26 | H Lundbeck As | agentes, usos e métodos |
| US10364286B2 (en) * | 2016-12-22 | 2019-07-30 | H. Lundbeck A/S | Monoclonal anti-alpha-synuclein antibodies for preventing tau aggregation |
| JP6908709B2 (ja) | 2017-01-06 | 2021-07-28 | エービーエル バイオ インコーポレイテッド | 抗α−syn抗体およびその使用 |
| EA201991720A1 (ru) | 2017-02-17 | 2020-01-20 | Бристол-Маерс Сквибб Компани | Антитела к альфа-синуклеину и их применения |
| MX389282B (es) * | 2017-03-27 | 2025-03-20 | Chase Therapeutics Corp | Composiciones y métodos para tratar sinucleinopatías. |
| US11220538B2 (en) | 2017-05-01 | 2022-01-11 | The Trustees Of The University Of Pennsylvania | Monoclonal antibodies against alpha-synuclein fibrils |
| US11155608B2 (en) | 2017-08-23 | 2021-10-26 | The Trustees Of The University Of Pennsylvania | Monoclonal antibodies against pathological alpha-synuclein, and methods using same |
| AU2018370279B2 (en) * | 2017-11-17 | 2022-11-03 | Abl Bio Inc. | Antibodies to a-synuclein and uses thereof |
| EP3725806A4 (de) | 2017-12-14 | 2022-03-30 | ABL Bio Inc. | Bispezifischer antikörper gegen a-syn/igf1r und seine verwendung |
| GB201720975D0 (en) | 2017-12-15 | 2018-01-31 | Ucb Biopharma Sprl | Anti-alpha synuclein antibodies |
| GB201720970D0 (en) | 2017-12-15 | 2018-01-31 | Ucb Biopharma Sprl | Antibodies |
| CA3088687A1 (en) | 2018-01-26 | 2019-08-01 | Quantum-Si Incorporated | Machine learning enabled pulse and base calling for sequencing devices |
| US20210032369A1 (en) * | 2018-02-12 | 2021-02-04 | The Scripps Research Institute | Methods related to parkinson?s disease and synucleinopathies |
| EP3833435B1 (de) * | 2018-08-09 | 2025-09-24 | F. Hoffmann-La Roche AG | Bestimmung von morbus parkinson |
| TWI734279B (zh) * | 2018-12-14 | 2021-07-21 | 美商美國禮來大藥廠 | 抗α-突觸核蛋白抗體及其用途 |
| MX2021012608A (es) * | 2019-04-18 | 2021-11-12 | Ac Immune Sa | Nuevas moleculas para terapia y diagnostico. |
| JP2022531177A (ja) * | 2019-04-29 | 2022-07-06 | トーマス・ジェファーソン・ユニバーシティ | 神経変性障害を治療するための方法 |
| US20250340623A1 (en) * | 2019-12-04 | 2025-11-06 | Ac Immune Sa | Novel Molecules for Therapy and Diagnosis |
| KR102466943B1 (ko) * | 2020-04-02 | 2022-11-14 | 한국과학기술연구원 | 갭을 갖는 바이오 센서를 이용한 파킨슨병 모니터링 방법 및 시스템 |
| CN111537738A (zh) * | 2020-05-18 | 2020-08-14 | 南通大学附属医院 | 用于超早期帕金森病检测的试剂盒 |
| EP4395894A4 (de) * | 2021-09-01 | 2025-08-06 | Vaxxinity Inc | Verfahren zur prävention und behandlung von synukleinopathien |
| AU2022348798A1 (en) | 2021-09-16 | 2024-03-14 | H. Lundbeck A/S | Compositions and methods for treating synucleinopathies |
| CN117430686B (zh) * | 2022-07-22 | 2025-07-01 | 深圳市安群生物工程有限公司 | α-突触核蛋白抗原表位肽及测定唾液中SNCA试剂盒和在帕金森病诊断中的应用 |
| WO2024177999A2 (en) * | 2023-02-21 | 2024-08-29 | The Regents Of The University Of Michigan | Human aggregated alpha-synuclein binding molecules |
| CN116077524A (zh) * | 2023-02-27 | 2023-05-09 | 广西馨海药业科技有限公司 | 氯化铷在制备治疗帕金森综合征的药物中的用途 |
| WO2025076635A1 (en) * | 2023-10-12 | 2025-04-17 | Universite Laval | Anti-alpha-synuclein antibodies and uses thereof |
| US20250282855A1 (en) | 2024-03-05 | 2025-09-11 | H. Lundbeck A/S | Compositions and methods for treating multiple system atrophy (msa) |
Citations (48)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4179337A (en) | 1973-07-20 | 1979-12-18 | Davis Frank F | Non-immunogenic polypeptides |
| US4495285A (en) | 1981-10-30 | 1985-01-22 | Kimihiro Shimizu | Plasminogen activator derivatives |
| US4609546A (en) | 1982-06-24 | 1986-09-02 | Japan Chemical Research Co., Ltd. | Long-acting composition |
| US4681581A (en) | 1983-12-05 | 1987-07-21 | Coates Fredrica V | Adjustable size diaper and folding method therefor |
| US4735210A (en) | 1985-07-05 | 1988-04-05 | Immunomedics, Inc. | Lymphographic and organ imaging method and kit |
| US4766106A (en) | 1985-06-26 | 1988-08-23 | Cetus Corporation | Solubilization of proteins for pharmaceutical compositions using polymer conjugation |
| US4946778A (en) | 1987-09-21 | 1990-08-07 | Genex Corporation | Single polypeptide chain binding molecules |
| WO1992003918A1 (en) | 1990-08-29 | 1992-03-19 | Genpharm International, Inc. | Transgenic non-human animals capable of producing heterologous antibodies |
| US5102990A (en) | 1989-08-09 | 1992-04-07 | Rhomed Incorporated | Direct radiolabeling of antibodies and other proteins with technetium or rhenium |
| US5101827A (en) | 1985-07-05 | 1992-04-07 | Immunomedics, Inc. | Lymphographic and organ imaging method and kit |
| WO1992022645A1 (en) | 1991-06-14 | 1992-12-23 | Genpharm International, Inc. | Transgenic immunodeficient non-human animals |
| WO1993001227A1 (en) | 1991-07-08 | 1993-01-21 | University Of Massachusetts At Amherst | Thermotropic liquid crystal segmented block copolymer |
| US5225539A (en) | 1986-03-27 | 1993-07-06 | Medical Research Council | Recombinant altered antibodies and methods of making altered antibodies |
| WO1994025585A1 (en) | 1993-04-26 | 1994-11-10 | Genpharm International, Inc. | Transgenic non-human animals capable of producing heterologous antibodies |
| US5530101A (en) | 1988-12-28 | 1996-06-25 | Protein Design Labs, Inc. | Humanized immunoglobulins |
| US5545807A (en) | 1988-10-12 | 1996-08-13 | The Babraham Institute | Production of antibodies from transgenic animals |
| US5545806A (en) | 1990-08-29 | 1996-08-13 | Genpharm International, Inc. | Ransgenic non-human animals for producing heterologous antibodies |
| US5589466A (en) | 1989-03-21 | 1996-12-31 | Vical Incorporated | Induction of a protective immune response in a mammal by injecting a DNA sequence |
| US5625126A (en) | 1990-08-29 | 1997-04-29 | Genpharm International, Inc. | Transgenic non-human animals for producing heterologous antibodies |
| US5633425A (en) | 1990-08-29 | 1997-05-27 | Genpharm International, Inc. | Transgenic non-human animals capable of producing heterologous antibodies |
| US5648471A (en) | 1987-12-03 | 1997-07-15 | Centocor, Inc. | One vial method for labeling antibodies with Technetium-99m |
| US5661016A (en) | 1990-08-29 | 1997-08-26 | Genpharm International Inc. | Transgenic non-human animals capable of producing heterologous antibodies of various isotypes |
| US5697902A (en) | 1985-07-05 | 1997-12-16 | Immunomedics, Inc. | Method for imaging and treating organs and tissues |
| US5741957A (en) | 1989-12-01 | 1998-04-21 | Pharming B.V. | Transgenic bovine |
| US5750172A (en) | 1987-06-23 | 1998-05-12 | Pharming B.V. | Transgenic non human mammal milk |
| US5756687A (en) | 1993-03-09 | 1998-05-26 | Genzyme Transgenics Corporation | Isolation of components of interest from milk |
| WO1998024884A1 (en) | 1996-12-02 | 1998-06-11 | Genpharm International | Transgenic non-human animals capable of producing heterologous antibodies |
| US5770429A (en) | 1990-08-29 | 1998-06-23 | Genpharm International, Inc. | Transgenic non-human animals capable of producing heterologous antibodies |
| US5789650A (en) | 1990-08-29 | 1998-08-04 | Genpharm International, Inc. | Transgenic non-human animals for producing heterologous antibodies |
| US5814318A (en) | 1990-08-29 | 1998-09-29 | Genpharm International Inc. | Transgenic non-human animals for producing heterologous antibodies |
| US5827690A (en) | 1993-12-20 | 1998-10-27 | Genzyme Transgenics Corporatiion | Transgenic production of antibodies in milk |
| US5859205A (en) | 1989-12-21 | 1999-01-12 | Celltech Limited | Humanised antibodies |
| US5874299A (en) | 1990-08-29 | 1999-02-23 | Genpharm International, Inc. | Transgenic non-human animals capable of producing heterologous antibodies |
| US5877397A (en) | 1990-08-29 | 1999-03-02 | Genpharm International Inc. | Transgenic non-human animals capable of producing heterologous antibodies of various isotypes |
| US5973972A (en) | 1995-11-03 | 1999-10-26 | Samsung Electronics, Co., Ltd. | Precharge system for a semiconductor memory device |
| US6077835A (en) | 1994-03-23 | 2000-06-20 | Case Western Reserve University | Delivery of compacted nucleic acid to cells |
| WO2000046147A2 (en) | 1999-02-03 | 2000-08-10 | Biosante Pharmaceuticals, Inc. | Therapeutic calcium phosphate particles and methods of manufacture and use |
| WO2000070087A1 (en) | 1999-05-14 | 2000-11-23 | Copernicus Therapeutics, Inc. | Automated nucleic acid compaction device |
| WO2001009187A2 (en) | 1999-07-29 | 2001-02-08 | Medarex, Inc. | Human monoclonal antibodies to her2/neu |
| WO2001014424A2 (en) | 1999-08-24 | 2001-03-01 | Medarex, Inc. | Human ctla-4 antibodies and their uses |
| WO2002043478A2 (en) | 2000-11-30 | 2002-06-06 | Medarex, Inc. | Transgenic transchromosomal rodents for making human antibodies |
| US6407213B1 (en) | 1991-06-14 | 2002-06-18 | Genentech, Inc. | Method for making humanized antibodies |
| US6881557B2 (en) | 2001-07-12 | 2005-04-19 | Arrowsmith Technologies Llp | Super humanized antibodies |
| WO2007059782A1 (en) | 2005-11-28 | 2007-05-31 | Genmab A/S | Recombinant monovalent antibodies and methods for production thereof |
| US20080175838A1 (en) | 2003-10-31 | 2008-07-24 | Elan Pharmaceuticals, Inc. | Prevention And Treatment of Synucleinopathic And Amyloidogenic Disease |
| WO2009097006A2 (en) | 2007-08-10 | 2009-08-06 | Medarex, Inc. | Hco32 and hco27 and related examples |
| US20120308572A1 (en) | 2010-02-26 | 2012-12-06 | Bioarctic Neuroscience Ab | Protofibril-Binding Antibodies and Their Use in Therapeutic and Diagnostic Methods for Parkinson's Disease, Dementia with Lewy Bodies and Other Alpha-Synucleinopathies |
| US20140127131A1 (en) | 2012-10-08 | 2014-05-08 | Neotope Biosciences Limited | Antibodies recognizing alpha-synuclein |
Family Cites Families (38)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB1512203A (en) | 1974-05-08 | 1978-05-24 | Aspin F | Rolls for use in profiling film |
| AU636562B2 (en) | 1986-12-15 | 1993-05-06 | British Technology Group Usa, Inc. | Monomeric phthalocyanine reagents |
| WO2002004482A1 (en) | 2000-07-07 | 2002-01-17 | Panacea Pharmaceuticals, Inc. | Methods for preventing neural tissue damage and for the treatment of alpha-synuclein diseases |
| WO2003000714A2 (en) | 2001-06-22 | 2003-01-03 | Panacea Pharmaceuticals, Inc. | Compositions and methods for preventing protein aggregation in neurodegenerative diseases |
| US8829198B2 (en) | 2007-10-31 | 2014-09-09 | Proteotech Inc | Compounds, compositions and methods for the treatment of beta-amyloid diseases and synucleinopathies |
| TW200509968A (en) | 2002-11-01 | 2005-03-16 | Elan Pharm Inc | Prevention and treatment of synucleinopathic disease |
| US9034337B2 (en) * | 2003-10-31 | 2015-05-19 | Prothena Biosciences Limited | Treatment and delay of outset of synucleinopathic and amyloidogenic disease |
| US8506959B2 (en) | 2002-11-01 | 2013-08-13 | Neotope Biosciences Limited | Prevention and treatment of synucleinopathic and amyloidogenic disease |
| HUE034320T2 (en) | 2003-05-19 | 2018-02-28 | Prothena Biosciences Ltd | Truncated fragments of alpha-synuclein in lewy-body disease |
| US7358331B2 (en) * | 2003-05-19 | 2008-04-15 | Elan Pharmaceuticals, Inc. | Truncated fragments of alpha-synuclein in Lewy body disease |
| WO2005047860A2 (en) * | 2003-11-08 | 2005-05-26 | Elan Pharmaceuticals, Inc. | Antibodies to alpha-synuclein |
| JP2006031250A (ja) | 2004-07-14 | 2006-02-02 | Fuji Xerox Co Ltd | 通信装置およびその制御方法 |
| MX2007001679A (es) | 2004-08-09 | 2007-05-23 | Elan Pharm Inc | Prevencion y tratamiento de la enfermedad sinucleinopatica y amiloidogenica. |
| US20060205024A1 (en) | 2005-03-08 | 2006-09-14 | Sun Health Research Institute | Method to diagnose and evaluate progression of Alzheimer's disease |
| WO2007011907A2 (en) * | 2005-07-19 | 2007-01-25 | University Of Rochester | Alpha-synuclein antibodies and methods related thereto |
| EP3067066B1 (de) | 2007-02-23 | 2019-03-27 | Prothena Biosciences Limited | Verhinderung und behandlung synukleinopathischer und amyloidogener krankheiten |
| DK3067066T3 (da) | 2007-02-23 | 2019-05-20 | Prothena Biosciences Ltd | Forebyggelse og behandling af synukleinopatisk og amyloidogenisk sygdom |
| CN101754682B (zh) * | 2007-05-16 | 2014-11-12 | 布里格姆妇女医院 | 突触核蛋白病的治疗 |
| EP3470079A1 (de) | 2008-04-29 | 2019-04-17 | BioArctic AB | Antikörper und impfstoffe zur verwendung in therapeutischen und diagnostischen verfahren für erkrankungen im zusammenhang mit alpha-synuklein |
| KR101781228B1 (ko) * | 2008-12-19 | 2017-09-22 | 바이오겐 인터내셔널 뉴로사이언스 게엠베하 | 인간 항-알파-시누클레인 자가항체 |
| NZ595630A (en) | 2009-04-09 | 2013-04-26 | Amicus Therapeutics Inc | Methods for preventing and/or treating degenerative disorders of the central nervous system |
| EP2366714A1 (de) | 2010-03-03 | 2011-09-21 | Dr. Rentschler Holding GmbH & Co. KG | Natürlich auftretende Autoantikörper gegen Alpha-Synuklein, die die Aggregation und Zytotoxizität von Alpha-Synuklein inhibieren |
| GB201008682D0 (en) | 2010-05-25 | 2010-07-07 | Vib Vzw | Epitope tag for affinity based applications |
| SG189174A1 (en) | 2010-10-11 | 2013-05-31 | Biogen Idec Internat Neuroscience Gmbh | Human anti-tau antibodies |
| MX357193B (es) * | 2011-06-23 | 2018-06-29 | Univ Zuerich | Moleculas de union anti-alfa sinucleina. |
| JP2013059866A (ja) | 2011-09-12 | 2013-04-04 | Seiko Epson Corp | 液体噴射装置、液体噴射装置の制御方法および液体噴射装置の制御プログラム |
| ES2937409T3 (es) * | 2011-10-28 | 2023-03-28 | Prothena Biosciences Ltd | Anticuerpos humanizados que reconocen la alfa-sinucleína |
| CN108623682A (zh) | 2012-07-03 | 2018-10-09 | 华盛顿大学 | 针对tau的抗体 |
| US9855276B2 (en) | 2012-10-25 | 2018-01-02 | The General Hospital Corporation | Combination therapies for the treatment of Alzheimer's disease and related disorders |
| US9534044B2 (en) | 2013-02-28 | 2017-01-03 | United Arab Emirates University | Alpha-synuclein antibodies and uses thereof |
| US10513555B2 (en) | 2013-07-04 | 2019-12-24 | Prothena Biosciences Limited | Antibody formulations and methods |
| MY176237A (en) * | 2013-11-21 | 2020-07-24 | Hoffmann La Roche | Anti-alpha-synuclein antibodies and methods of use |
| WO2016061389A2 (en) | 2014-10-16 | 2016-04-21 | Genentech, Inc. | Anti-alpha-synuclein antibodies and methods of use |
| HK1245078A1 (zh) | 2015-05-07 | 2018-08-24 | Axovant Sciences Gmbh | 治疗神经退行性疾病的方法 |
| GB201512203D0 (en) | 2015-07-13 | 2015-08-19 | Lundbeck & Co As H | Agents,uses and methods |
| PT3463435T (pt) | 2016-06-02 | 2021-11-22 | Medimmune Ltd | Anticorpos para alfa-sinucleína e as suas utilizações |
| CA3043651A1 (en) | 2016-11-15 | 2018-05-24 | H. Lundbeck A/S | Agents, uses and methods for the treatment of synucleinopathy |
| US10364286B2 (en) | 2016-12-22 | 2019-07-30 | H. Lundbeck A/S | Monoclonal anti-alpha-synuclein antibodies for preventing tau aggregation |
-
2015
- 2015-07-13 GB GBGB1512203.9A patent/GB201512203D0/en not_active Ceased
-
2016
- 2016-07-11 JO JOP/2016/0140A patent/JO3692B1/ar active
- 2016-07-12 KR KR1020177037733A patent/KR102793383B1/ko active Active
- 2016-07-12 TW TW105121902A patent/TWI729992B/zh active
- 2016-07-12 JP JP2018501163A patent/JP7012004B2/ja active Active
- 2016-07-12 FI FIEP16738736.4T patent/FI3322722T3/fi active
- 2016-07-12 DK DK16738736.4T patent/DK3322722T3/da active
- 2016-07-12 SI SI201631902T patent/SI3322722T1/sl unknown
- 2016-07-12 CN CN202511963296.1A patent/CN121736097A/zh active Pending
- 2016-07-12 AR ARP160102110A patent/AR105336A1/es unknown
- 2016-07-12 EP EP25154059.7A patent/EP4582144A3/de active Pending
- 2016-07-12 WO PCT/EP2016/066476 patent/WO2017009312A1/en not_active Ceased
- 2016-07-12 RS RS20250497A patent/RS66840B1/sr unknown
- 2016-07-12 PE PE2018000066A patent/PE20181049A1/es unknown
- 2016-07-12 EP EP16738736.4A patent/EP3322722B1/de active Active
- 2016-07-12 KR KR1020257011170A patent/KR20250051158A/ko active Pending
- 2016-07-12 RU RU2017145653A patent/RU2765303C2/ru active
- 2016-07-12 PT PT167387364T patent/PT3322722T/pt unknown
- 2016-07-12 MY MYPI2017001926A patent/MY194944A/en unknown
- 2016-07-12 US US15/207,859 patent/US10800836B2/en active Active
- 2016-07-12 ES ES16738736T patent/ES3028882T3/es active Active
- 2016-07-12 CR CR20230300A patent/CR20230300A/es unknown
- 2016-07-12 AU AU2016292896A patent/AU2016292896B2/en active Active
- 2016-07-12 CN CN201680039417.6A patent/CN107709361A/zh active Pending
- 2016-07-12 MA MA42439A patent/MA42439B1/fr unknown
- 2016-07-12 TN TNP/2018/000005A patent/TN2018000005A1/en unknown
- 2016-07-12 CN CN202210997939.4A patent/CN115925921A/zh active Pending
- 2016-07-12 PL PL16738736.4T patent/PL3322722T3/pl unknown
- 2016-07-12 HR HRP20250624TT patent/HRP20250624T1/hr unknown
- 2016-07-12 HU HUE16738736A patent/HUE071541T2/hu unknown
- 2016-07-12 GE GEAP201614670A patent/GEP20207151B/en unknown
- 2016-07-12 GE GEAP202014670A patent/GEAP202014670A/en unknown
- 2016-07-12 MX MX2018000504A patent/MX2018000504A/es unknown
- 2016-07-12 CR CR20180021A patent/CR20180021A/es unknown
- 2016-07-12 LT LTEPPCT/EP2016/066476T patent/LT3322722T/lt unknown
- 2016-07-12 UA UAA201800664A patent/UA125501C2/uk unknown
- 2016-07-12 EA EA201792616A patent/EA036499B1/ru not_active IP Right Cessation
-
2017
- 2017-11-14 US US15/812,410 patent/US10358484B2/en active Active
- 2017-11-14 US US15/812,251 patent/US10358482B2/en active Active
- 2017-11-14 US US15/812,363 patent/US10364285B2/en active Active
- 2017-11-14 US US15/812,303 patent/US10358483B2/en active Active
- 2017-12-15 CO CONC2017/0012941A patent/CO2017012941A2/es unknown
- 2017-12-18 ZA ZA2017/08607A patent/ZA201708607B/en unknown
- 2017-12-22 IL IL256501A patent/IL256501B/en unknown
-
2018
- 2018-01-03 PH PH12018500016A patent/PH12018500016A1/en unknown
- 2018-01-09 CL CL2018000075A patent/CL2018000075A1/es unknown
- 2018-01-11 SV SV2018005612A patent/SV2018005612A/es unknown
- 2018-01-11 NI NI201800006A patent/NI201800006A/es unknown
- 2018-01-11 DO DO2018000013A patent/DOP2018000013A/es unknown
- 2018-01-12 EC ECIEPI20182645A patent/ECSP18002645A/es unknown
- 2018-01-12 MX MX2023009483A patent/MX2023009483A/es unknown
-
2019
- 2019-06-17 US US16/443,249 patent/US10640554B2/en active Active
- 2019-06-17 US US16/443,187 patent/US10647763B2/en active Active
- 2019-06-17 US US16/443,225 patent/US10647764B2/en active Active
-
2020
- 2020-06-08 US US16/895,471 patent/US11524995B2/en active Active
- 2020-08-27 US US17/004,447 patent/US11421024B2/en active Active
- 2020-11-03 US US17/088,307 patent/US11542323B2/en active Active
-
2021
- 2021-08-24 JP JP2021136539A patent/JP7138322B2/ja active Active
-
2022
- 2022-08-29 JP JP2022136108A patent/JP7608404B2/ja active Active
- 2022-11-22 US US17/992,403 patent/US20230312693A1/en active Pending
-
2024
- 2024-12-18 JP JP2024221962A patent/JP2025041734A/ja active Pending
Patent Citations (53)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4179337A (en) | 1973-07-20 | 1979-12-18 | Davis Frank F | Non-immunogenic polypeptides |
| US4495285A (en) | 1981-10-30 | 1985-01-22 | Kimihiro Shimizu | Plasminogen activator derivatives |
| US4495285B1 (de) | 1981-10-30 | 1986-09-23 | Nippon Chemiphar Co | |
| US4609546A (en) | 1982-06-24 | 1986-09-02 | Japan Chemical Research Co., Ltd. | Long-acting composition |
| US4681581A (en) | 1983-12-05 | 1987-07-21 | Coates Fredrica V | Adjustable size diaper and folding method therefor |
| US4766106A (en) | 1985-06-26 | 1988-08-23 | Cetus Corporation | Solubilization of proteins for pharmaceutical compositions using polymer conjugation |
| US5697902A (en) | 1985-07-05 | 1997-12-16 | Immunomedics, Inc. | Method for imaging and treating organs and tissues |
| US5101827A (en) | 1985-07-05 | 1992-04-07 | Immunomedics, Inc. | Lymphographic and organ imaging method and kit |
| US4735210A (en) | 1985-07-05 | 1988-04-05 | Immunomedics, Inc. | Lymphographic and organ imaging method and kit |
| US5225539A (en) | 1986-03-27 | 1993-07-06 | Medical Research Council | Recombinant altered antibodies and methods of making altered antibodies |
| US5750172A (en) | 1987-06-23 | 1998-05-12 | Pharming B.V. | Transgenic non human mammal milk |
| US4946778A (en) | 1987-09-21 | 1990-08-07 | Genex Corporation | Single polypeptide chain binding molecules |
| US5648471A (en) | 1987-12-03 | 1997-07-15 | Centocor, Inc. | One vial method for labeling antibodies with Technetium-99m |
| US5545807A (en) | 1988-10-12 | 1996-08-13 | The Babraham Institute | Production of antibodies from transgenic animals |
| US5530101A (en) | 1988-12-28 | 1996-06-25 | Protein Design Labs, Inc. | Humanized immunoglobulins |
| US5585089A (en) | 1988-12-28 | 1996-12-17 | Protein Design Labs, Inc. | Humanized immunoglobulins |
| US5589466A (en) | 1989-03-21 | 1996-12-31 | Vical Incorporated | Induction of a protective immune response in a mammal by injecting a DNA sequence |
| USRE35500E (en) | 1989-08-09 | 1997-05-06 | Aberlyn Capital Management Limited Partnership | Direct radiolabeling of antibodies and other proteins with technetium or rhenium |
| US5102990A (en) | 1989-08-09 | 1992-04-07 | Rhomed Incorporated | Direct radiolabeling of antibodies and other proteins with technetium or rhenium |
| US5741957A (en) | 1989-12-01 | 1998-04-21 | Pharming B.V. | Transgenic bovine |
| US5859205A (en) | 1989-12-21 | 1999-01-12 | Celltech Limited | Humanised antibodies |
| US5877397A (en) | 1990-08-29 | 1999-03-02 | Genpharm International Inc. | Transgenic non-human animals capable of producing heterologous antibodies of various isotypes |
| US5625126A (en) | 1990-08-29 | 1997-04-29 | Genpharm International, Inc. | Transgenic non-human animals for producing heterologous antibodies |
| US5633425A (en) | 1990-08-29 | 1997-05-27 | Genpharm International, Inc. | Transgenic non-human animals capable of producing heterologous antibodies |
| US5661016A (en) | 1990-08-29 | 1997-08-26 | Genpharm International Inc. | Transgenic non-human animals capable of producing heterologous antibodies of various isotypes |
| US5569825A (en) | 1990-08-29 | 1996-10-29 | Genpharm International | Transgenic non-human animals capable of producing heterologous antibodies of various isotypes |
| US5545806A (en) | 1990-08-29 | 1996-08-13 | Genpharm International, Inc. | Ransgenic non-human animals for producing heterologous antibodies |
| WO1992003918A1 (en) | 1990-08-29 | 1992-03-19 | Genpharm International, Inc. | Transgenic non-human animals capable of producing heterologous antibodies |
| US5874299A (en) | 1990-08-29 | 1999-02-23 | Genpharm International, Inc. | Transgenic non-human animals capable of producing heterologous antibodies |
| US5770429A (en) | 1990-08-29 | 1998-06-23 | Genpharm International, Inc. | Transgenic non-human animals capable of producing heterologous antibodies |
| US5789650A (en) | 1990-08-29 | 1998-08-04 | Genpharm International, Inc. | Transgenic non-human animals for producing heterologous antibodies |
| US5814318A (en) | 1990-08-29 | 1998-09-29 | Genpharm International Inc. | Transgenic non-human animals for producing heterologous antibodies |
| WO1992022645A1 (en) | 1991-06-14 | 1992-12-23 | Genpharm International, Inc. | Transgenic immunodeficient non-human animals |
| US6407213B1 (en) | 1991-06-14 | 2002-06-18 | Genentech, Inc. | Method for making humanized antibodies |
| WO1993001227A1 (en) | 1991-07-08 | 1993-01-21 | University Of Massachusetts At Amherst | Thermotropic liquid crystal segmented block copolymer |
| US5756687A (en) | 1993-03-09 | 1998-05-26 | Genzyme Transgenics Corporation | Isolation of components of interest from milk |
| WO1994025585A1 (en) | 1993-04-26 | 1994-11-10 | Genpharm International, Inc. | Transgenic non-human animals capable of producing heterologous antibodies |
| US5827690A (en) | 1993-12-20 | 1998-10-27 | Genzyme Transgenics Corporatiion | Transgenic production of antibodies in milk |
| US6077835A (en) | 1994-03-23 | 2000-06-20 | Case Western Reserve University | Delivery of compacted nucleic acid to cells |
| US5973972A (en) | 1995-11-03 | 1999-10-26 | Samsung Electronics, Co., Ltd. | Precharge system for a semiconductor memory device |
| WO1998024884A1 (en) | 1996-12-02 | 1998-06-11 | Genpharm International | Transgenic non-human animals capable of producing heterologous antibodies |
| WO2000046147A2 (en) | 1999-02-03 | 2000-08-10 | Biosante Pharmaceuticals, Inc. | Therapeutic calcium phosphate particles and methods of manufacture and use |
| WO2000070087A1 (en) | 1999-05-14 | 2000-11-23 | Copernicus Therapeutics, Inc. | Automated nucleic acid compaction device |
| WO2001009187A2 (en) | 1999-07-29 | 2001-02-08 | Medarex, Inc. | Human monoclonal antibodies to her2/neu |
| WO2001014424A2 (en) | 1999-08-24 | 2001-03-01 | Medarex, Inc. | Human ctla-4 antibodies and their uses |
| WO2002043478A2 (en) | 2000-11-30 | 2002-06-06 | Medarex, Inc. | Transgenic transchromosomal rodents for making human antibodies |
| US6881557B2 (en) | 2001-07-12 | 2005-04-19 | Arrowsmith Technologies Llp | Super humanized antibodies |
| US20080175838A1 (en) | 2003-10-31 | 2008-07-24 | Elan Pharmaceuticals, Inc. | Prevention And Treatment of Synucleinopathic And Amyloidogenic Disease |
| WO2007059782A1 (en) | 2005-11-28 | 2007-05-31 | Genmab A/S | Recombinant monovalent antibodies and methods for production thereof |
| WO2009097006A2 (en) | 2007-08-10 | 2009-08-06 | Medarex, Inc. | Hco32 and hco27 and related examples |
| US20120308572A1 (en) | 2010-02-26 | 2012-12-06 | Bioarctic Neuroscience Ab | Protofibril-Binding Antibodies and Their Use in Therapeutic and Diagnostic Methods for Parkinson's Disease, Dementia with Lewy Bodies and Other Alpha-Synucleinopathies |
| US8632776B2 (en) | 2010-02-26 | 2014-01-21 | Bioarctic Neuroscience Ab | Protofibril-binding antibodies and their use in therapeutic and diagnostic methods for Parkinson's disease, dementia with lewy bodies and other α-synucleinopathies |
| US20140127131A1 (en) | 2012-10-08 | 2014-05-08 | Neotope Biosciences Limited | Antibodies recognizing alpha-synuclein |
Non-Patent Citations (139)
| Title |
|---|
| "Canonical structures For The Hypervariable domains Of Immunoglobulins", J. MOL. BIOL., vol. 196, 1987, pages 901 - 917 |
| "Remington: The Science and Practice of Pharmacy", 2013, MACK PUBLISHING CO. |
| "Sustained and Controlled Release Drug Delivery Systems", 1978, MARCEL DEKKER, INC. |
| "UniProt", Database accession no. P37840 |
| ALTSCHUL, S.F.: "Amino Acid Substitution Matrices From An Information Theoretic Perspective", J. MOL. BIOL., vol. 219, 1991, pages 555 - 565, XP024010245, DOI: 10.1016/0022-2836(91)90193-A |
| ASLANIDIS, C.P.J. DE JONG, NUCLEIC ACIDS RES, vol. 18, no. 20, 1990, pages 6069 - 74 |
| BAE ET AL., J NEUROSCI., vol. 32, no. 39, 26 September 2012 (2012-09-26), pages 13454 - 69 |
| BARDERAS, R. ET AL.: "Affinity Maturation Of Antibodies Assisted By In Silico Modeling", PROC. NATL. ACAD. SCI., 2008 |
| BENVENISTYRESHEF, PNAS USA, vol. 83, 1986, pages 9551 - 55 |
| BESONG-AGBO ET AL., NEUROLOGY, vol. 80, no. 2, 8 January 2013 (2013-01-08), pages 169 - 75 |
| BEYERARIZA, MOL NEUROBIOL., vol. 47, no. 2, April 2013 (2013-04-01), pages 509 - 24 |
| BOER, E. ET AL., APPL. MICROBIOL. BIOTECHNOL., vol. 77, no. 3, 2007, pages 513 - 523 |
| BOSTROM, J. ET AL.: "Improving Antibody Binding Affinity And Specificity For Therapeutic Development", METHODS MOL. BIOL., vol. 525, 2009, pages 353 - 376 |
| CARTER, P. ET AL.: "Humanization Of An Anti-p185her2 Antibody For Human Cancer Therapy", PROC. NATL. ACAD. SCI. (U.S.A., vol. 89, 1992, pages 4285 - 4289, XP000275844, DOI: 10.1073/pnas.89.10.4285 |
| CELIK, E. ET AL., BIOTECHNOL. ADV., vol. 30, no. 5, 2012, pages 1108 - 1118 |
| CHEN ET AL., EMBO J., vol. 12, 1993, pages 811 - 820 |
| CHEN, J. ET AL., INTERNATIONAL IMMUNOLOGY, vol. 5, 1993, pages 647 - 656 |
| CHOTHIA, C. ET AL.: "Canonical Structures For The Hypervariable domains Of Immunoglobulins", J. MOL. BIOL., vol. 196, 1987, pages 901 - 917 |
| CLACKSON ET AL., NATURE, vol. 352, 1991, pages 624 - 628 |
| CO, M. S. ET AL.: "Humanized Antibodies For Antiviral Therapy", PROC. NATL. ACAD. SCI. (U.S.A., vol. 88, 1991, pages 2869 - 2873, XP000200583, DOI: 10.1073/pnas.88.7.2869 |
| CO, M.S. ET AL.: "Chimeric And Humanized Antibodies With Specificity For The CD33 Antigen", J. IMMUNOL., vol. 148, 1992, pages 1149 - 1154 |
| CONWAY ET AL., PROC NATL ACAD SCI USA, vol. 97, 2000, pages 571 - 576 |
| CORAROPEARSON, SOMATIC CELL GENETICS, vol. 2, 1981, pages 603 |
| CREIGHTON: "Proteins: Structure and Molecular Properties", 1984, W. H. FREEMAN AND COMPANY |
| DUFTY ET AL., AM J OF PATHOL, vol. 170, no. 5, May 2007 (2007-05-01) |
| DUMONT ET AL., CRIT REV BIOTECHNOL., vol. 18, 2015, pages 1 - 13 |
| EDDY, S.R: "Where Did The BLOSUM62 Alignment Score Matrix Come From?", NATURE BIOTECH., vol. 22, no. 8, 2004, pages 1035 - 1036 |
| ELVANG ET AL., J NEUROCHEM., vol. 110, no. 5, 2009, pages 1377 - 87 |
| EVANS ET AL., J. IMMUNOL. METH., vol. 184, 1995, pages 123 - 38 |
| FINLAY, W.J. ET AL.: "Affinity Maturation Of A Humanized Rat Antibody For Anti-RAGE Therapy: Comprehensive Mutagenesis Reveals A High Level Of Mutational Plasticity Both Inside And Outside The Complementarity-Determining Regions", J. MOL. BIOL., vol. 388, no. 3, 2009, pages 541 - 558, XP026046499, DOI: 10.1016/j.jmb.2009.03.019 |
| FISHWILD, D. ET AL., NATURE BIOTECHNOLOGY, vol. 14, 1996, pages 845 - 851 |
| FLEMINGCHESSELET, BEHAV PHARMACOL., vol. 17, no. 5-6, September 2006 (2006-09-01), pages 383 - 91 |
| GALASKO ET AL., ARCH. NEUROL., vol. 51, 1994, pages 888 - 95 |
| GAMES ET AL., AM J OF PATHOL, vol. 182, no. 3, March 2013 (2013-03-01) |
| GAMES ET AL., J NEUROSCI., vol. 34, no. 28, 9 July 2014 (2014-07-09), pages 9441 - 54 |
| GLASER ET AL., J. IMMUNOLOGY, vol. 149, 1992, pages 3903 |
| GONZALES, N.R. ET AL.: "SDR Grafting Of A Murine Antibody Using Multiple Human Germline Templates To Minimize Its Immunogenicity", MOL. IMMUNOL., vol. 41, 2004, pages 863 - 872, XP002402422, DOI: 10.1016/j.molimm.2004.03.041 |
| GORMAN, S. D. ET AL.: "Reshaping A Therapeutic CD4 Antibody", PROC. NATL. ACAD. SCI. (U.S.A., vol. 88, 1991, pages 4181 - 4185, XP000368701, DOI: 10.1073/pnas.88.10.4181 |
| GRANT ET AL., METHODS IN ENZYMOL, vol. 153, 1987, pages 516 - 544 |
| GRUDEN ET AL., J NEUROIMMUNOL., vol. 233, no. 1-2, April 2011 (2011-04-01), pages 221 - 7 |
| GRUDEN ET AL., NEUROIMMUNOMODULATION, vol. 19, no. 6, 2012, pages 334 - 42 |
| GUNASEKARAN ET AL., JBC, vol. 285, 2010, pages 19637 - 1 |
| GUSTCHINA, E.: "Affinity Maturation By Targeted Diversification Of The CDR-H2 Loop Of A Monoclonal Fab Derived From A Synthetic Naive Human Antibody Library And Directed Against The Internal Trimeric Coiled-Coil Of Gp41 Yields A Set Of Fabs With Improved HIV-1 Neutralization Potency And Breadth", VIROLOGY, vol. 393, no. 1, 2009, pages 112 - 119, XP026641691, DOI: 10.1016/j.virol.2009.07.019 |
| HACKEL, B.J. ET AL.: "Stability And CDR Composition Biases Enrich Binder Functionality Landscapes", J. MOL. BIOL., vol. 401, no. 1, 2010, pages 84 - 96, XP027143129, DOI: 10.1016/j.jmb.2010.06.004 |
| HALL ET AL., EXP NEUROL., vol. 264, February 2015 (2015-02-01), pages 8 - 13 |
| HANSENLI, TRENDS MOL MED., vol. 18, no. 5, May 2012 (2012-05-01), pages 248 - 55 |
| HARDING, F.LONBERG, N., ANN. N. Y. ACAD. SCI, vol. 764, 1995, pages 536 - 546 |
| HENIKOFF, J.G.: "Amino acid substitution matrices from protein blocks", PROC. NATL. ACAD. SCI. (USA, vol. 89, 1992, pages 10915 - 10919, XP002599751, DOI: 10.1073/pnas.89.22.10915 |
| HOLT ET AL., TRENDS BIOTECHNOL., vol. 2, no. 2, November 2003 (2003-11-01), pages 484 - 90 |
| HUSTON ET AL., PNAS USA, vol. 85, 1988, pages 5879 - 5883 |
| HUSTON ET AL., PROC. NATL. ACAD. SCI. (U.S.A., vol. 85, 1988, pages 5879 - 5883 |
| JELLINGER, MOV DISORD., vol. 27, no. 1, January 2012 (2012-01-01), pages 8 - 30 |
| JUNGHANS ET AL.: "Cancer Chemotherapy and Biotherapy", 1996, LIPPINCOTT RAVEN, pages: 655 - 686 |
| KARLIN, S. ET AL.: "Methods For Assessing The Statistical Significance Of Molecular Sequence Features By Using General Scoring Schemes", PROC. NATL. ACAD. SCI. (USA, vol. 87, 1990, pages 2264 - 2268, XP001030853, DOI: 10.1073/pnas.87.6.2264 |
| KELLIE ET AL., SCI REP., vol. 4, 2014, pages 5797 |
| KELLIE JFHIGGS RERYDER JWMAJOR ABEACH TGADLER CHMERCHANT KKNIERMAN MD: "Quantitative measurement of intact alpha-synuclein proteoforms from post-mortem control and Parkinson's disease brain tissue by mass spectrometry", SCI REP., vol. 4, 23 July 2014 (2014-07-23), pages 5797 |
| KETTLEBOROUGH, C. A. ET AL.: "Humanization Of A Mouse Monoclonal Antibody By CDR-Grafting: The Importance Of Framework Residues On Loop Conformation", PROTEIN ENGINEERING, vol. 4, 1991, pages 773 - 3783 |
| KIRIK ET AL., J NEUROSCI., vol. 22, no. 7, 1 April 2002 (2002-04-01), pages 2780 - 91 |
| KIRIKBJORKLUND, TRENDS NEUROSCI., vol. 26, no. 7, July 2003 (2003-07-01), pages 386 - 92 |
| KOEHLER ET AL., PLOS ONE, vol. 8, no. 5, 31 May 2013 (2013-05-31), pages e64649 |
| KOHLER ET AL., NATURE, vol. 256, 1975, pages 495 |
| KRAUSE, J.C. ET AL.: "An Insertion Mutation That Distorts Antibody Binding Site Architecture Enhances Function Of A Human Antibody", MBIO, vol. 2, no. 1, 2011, pages e00345 - 10, XP055448443, DOI: 10.1128/mBio.00345-10 |
| KRUGER ET AL., NATURE GEN., vol. 18, 1998, pages 106 - 8 |
| KUAN, C.T. ET AL.: "Affinity-Matured Anti-Glycoprotein NMB Recombinant Immunotoxins Targeting Malignant Gliomas And Melanomas", INT. J. CANCER, 2010 |
| KURTH, M. ET AL.: "Site-Specific Conjugation Of A Radioiodinated Phenethylamine Derivative To A Monoclonal Antibody Results In Increased Radioactivity Localization In Tumor", J. MED. CHEM., vol. 36, no. 9, 1993, pages 1255 - 1261, XP055032229, DOI: 10.1021/jm00061a017 |
| LABRIJN ET AL., PNAS, vol. 110, no. 113, 2013, pages 5145 - 5150 |
| LEE ET AL., NAT REV NEUROL., vol. 10, no. 2, February 2014 (2014-02-01), pages 92 - 8 |
| LEETROJANOWSKI, NEURON., vol. 52, no. 1, 5 October 2006 (2006-10-05), pages 33 - 8 |
| LI, P. ET AL., APPL. BIOCHEM. BIOTECHNOL., vol. 142, no. 2, 2007, pages 105 - 124 |
| LINDEGREN, S. ET AL.: "Chloramine-T In High-Specific-Activity Radioiodination Of Antibodies Using N-Succinimidyl-3-(Trimethylstannyl)Benzoate As An Intermediate", NUCL. MED. BIOL., vol. 25, no. 7, 1998, pages 659 - 665, XP004149436, DOI: 10.1016/S0969-8051(98)00033-X |
| LINDSTROM ET AL., NEUROBIOL DIS., vol. 69, September 2014 (2014-09-01), pages 134 - 43 |
| LOBUGLIO, A.F. ET AL.: "Mouse/Human Chimeric Monoclonal Antibody In Man: Kinetics And Immune Response", PROC. NATL. ACAD. SCI. (U.S.A., vol. 86, 1989, pages 4220 - 4224, XP002100301, DOI: 10.1073/pnas.86.11.4220 |
| LONBERG, N. ET AL., NATURE, vol. 368, 1994, pages 856 - 859 |
| LONBERG, N.HUSZAR, D., INTERN. REV. IMMUNOL., vol. 13, 1995, pages 65 - 93 |
| LORENZO ET AL., PLOSONE, December 2015 (2015-12-01) |
| LUK ET AL., SCIENCE, vol. 338, no. 6109, 16 November 2012 (2012-11-16), pages 949 - 53 |
| MABRY ET AL., PEDS, vol. 23, no. 3, 2010, pages 115 - 127 |
| MAEDA, H. ET AL.: "Construction Of Reshaped Human Antibodies With HIV-Neutralizing Activity", HUMAN ANTIBODIES HYBRIDOMA, vol. 2, 1991, pages 124 - 134 |
| MAETZLER ET AL., PLOS ONE, vol. 9, no. 2, 21 February 2014 (2014-02-21), pages e88604 |
| MAHOWALD ET AL., NEUROLOGY, vol. 75, 2010, pages 488 - 489 |
| MARKS ET AL., J. MOL. BIOL., vol. 222, 1991, pages 581 - 597 |
| MATTANOVICH, D. ET AL., METHODS MOL. BIOL., vol. 824, 2012, pages 329 - 358 |
| MCCAFFERTY ET AL., NATURE, vol. 348, 1990, pages 552 - 554 |
| MCKEITH ET AL., NEUROLOGY, vol. 47, 1996, pages 1113 - 24 |
| METZ ET AL., PROTEIN ENGINEERING, vol. 25, no. 10, 2012, pages 571 - 580 |
| MISHIZEN-EBERZ, BIOCHEMISTRY, vol. 44, 2005, pages 7818 - 7829 |
| MISHIZEN-EBERZ, J, NEUROCHEM, vol. 86, 2003, pages 836 - 847 |
| MONTGOMERY, D. L. ET AL.: "Affinity Maturation And Characterization Of A Human Monoclonal Antibody Against HIV-1 gp41", MABS, vol. 1, no. 5, 2009, pages 462 - 474, XP055568716, DOI: 10.4161/mabs.1.5.9214 |
| MOORE ET AL., MABS, vol. 3, no. 6, 2011, pages 546 - 557 |
| NEMANI ET AL., NEURON., vol. 65, no. 1, 14 January 2010 (2010-01-14), pages 66 - 79 |
| PACHECO ET AL., J NEUROCHEM., vol. 132, no. 6, March 2015 (2015-03-01), pages 731 - 4 |
| PAPACHRONI ET AL., J NEUROCHEM., vol. 101, no. 3, May 2007 (2007-05-01), pages 749 - 56 |
| PAUMIER ET AL., PLOS ONE, vol. 8, no. 8, 1 August 2013 (2013-08-01), pages e70274 |
| PAXINOSFRANKLIN'S, MOUSE BRAIN IN STEREOTAXIC COORDINATES, 2001 |
| PLUCKTHUN: "The Pharmacology of Monoclonal Antibodies", vol. 113, 1994, SPRINGER-VERLAG, pages: 269 - 315 |
| PNAS USA, vol. 90, no. 14, 1993, pages 6444 - 8 |
| POLYMEROPOULOS ET AL., SCIENCE, vol. 276, 1997, pages 2045 - 7 |
| REA, D.W. ET AL.: "Site-specifically radioiodinated antibody for targeting tumors", CANCER RES., vol. 50, 1990, pages 857 - 861 |
| REVETS ET AL., EXPERT OPIN BIOL THER., vol. 5, January 2005 (2005-01-01), pages 11 - 24 |
| RIDGWAY ET AL., PROTEIN ENGINEERING, vol. 9, 1996, pages 617 - 621 |
| RIECHMANN, L. ET AL.: "Reshaping Human Antibodies for Therapy", NATURE, vol. 332, 1988, pages 323 - 327, XP002007067, DOI: 10.1038/332323a0 |
| RITCHIE ET AL., HEALTH, vol. 4, 2012, pages 1167 - 1177 |
| RUDIKOFF, S.: "Single Amino Acid Substitution Altering Antigen-Binding Specificity", PROC. NATL. ACAD. SCI. (USA, vol. 79, no. 6, 1982, pages 1979 - 1983, XP002986051, DOI: 10.1073/pnas.79.6.1979 |
| SATO, K. ET AL., CANCER RES, vol. 53, 1993, pages 851 - 856 |
| SCHAKOWSKI ET AL., MOL THER, vol. 3, 2001, pages 793 - 800 |
| SCHIER ET AL., J. MOL. BIO., vol. 263, 1996, pages 551 |
| SHAHADUZZAMAN ET AL., PLOS ONE, vol. 10, no. 2, 6 February 2015 (2015-02-06), pages e0116841 |
| SMITH ET AL., PLOS ONE, vol. 7, no. 12, 2012, pages e52285 |
| SPENCER ET AL., MOL THER., vol. 22, no. 10, October 2014 (2014-10-01), pages 1753 - 67 |
| SPILLANTINI ET AL., NATURE, vol. 388, 1997, pages 839 - 40 |
| SPRETER VON KREUDENSTEIN ET AL., MABS, vol. 5, no. 5, 2013, pages 646 - 654 |
| SPRINGERKAHLE, CURR NEUROL NEUROSCI REP., vol. 6, no. 5, September 2006 (2006-09-01), pages 432 - 6 |
| STEIDL, S. ET AL.: "In Vitro Affinity Maturation Of Human GM-CSF Antibodies By Targeted CDR-Diversification", MOL. IMMUNOL., vol. 46, no. 1, 2008, pages 135 - 144, XP025472134, DOI: 10.1016/j.molimm.2008.07.013 |
| STROP ET AL., JMB, vol. 420, 2012, pages 204 - 219 |
| SYKESJOHNSTON, NAT BIOTECH, vol. 12, 1997, pages 355 - 59 |
| TAKEDA ET AL., J. PATHOL., vol. 152, 1998, pages 367 - 72 |
| TAYLOR, L. ET AL., INTERNATIONAL IMMUNOLOGY, vol. 6, 1994, pages 579 - 591 |
| TAYLOR, L. ET AL., NUCLEIC ACIDS RESEARCH, vol. 20, 1992, pages 6287 - 6295 |
| TEMPEST, P.R. ET AL.: "Reshaping A Human Monoclonal Antibody To Inhibit Human Respiratory Syncytial Virus Infection in vivo", BIO/TECHNOLOGY, vol. 9, 1991, pages 266 - 271, XP000986147, DOI: 10.1038/nbt0391-266 |
| TOFARIS ET AL., J NEUROSCI., vol. 26, no. 15, 12 April 2006 (2006-04-12), pages 3942 - 50 |
| TRAN ET AL., CELL REP., vol. 7, no. 6, 26 June 2014 (2014-06-26), pages 2054 - 65 |
| TUAILLON ET AL., J. IMMUNOL., vol. 152, 1994, pages 2912 - 2920 |
| ULUSOY ET AL., EUR J NEUROSCI., vol. 32, no. 3, August 2010 (2010-08-01), pages 409 - 22 |
| VAIKATH ET AL., NEUROBIOL DIS., vol. 79, 2015, pages 81 - 99 |
| VAN DER VAART, J.M., METHODS MOL. BIOL., vol. 178, 2002, pages 349 - 357 |
| VAN HEEKESCHUSTER, J BIOL CHEM, vol. 264, 1989, pages 5503 - 5509 |
| VEKRELLIS ET AL., LANCET NEUROL., vol. 10, no. 11, November 2011 (2011-11-01), pages 1015 - 25 |
| VERHOEYEN, M.: "Reshaping Human Antibodies: Grafting An Antilysozyme Activity", SCIENCE, vol. 239, 1988, pages 1534 - 1536, XP002949269, DOI: 10.1126/science.2451287 |
| VOLLES ET AL., J. BIOCHEM., vol. 42, 2003, pages 7871 - 7878 |
| WAKABAYASHI ET AL., NEUROSCI. LETT., vol. 239, 1997, pages 45 - 8 |
| WAKAMATSU ET AL., NEUROBIOL AGING, vol. 29, no. 4, April 2008 (2008-04-01), pages 574 - 85 |
| WARD ET AL., NATURE, vol. 341, 1989, pages 544 - 546 |
| WESTERLUND M ET AL., MOL CELL NEUROSCI., vol. 39, no. 4, December 2008 (2008-12-01), pages 586 - 91 |
| WIGLER ET AL., CELL, vol. 14, 1978, pages 725 |
| WOULFE ET AL., NEUROLOGY, vol. 58, no. 9, 14 May 2002 (2002-05-14), pages 1435 - 6 |
| WU ET AL., PROC. NATL. ACAD. SCI. (U.S.A., vol. 95, 1998, pages 6037 |
| XU LDALY TGAO CFLOTTE TRSONG SBYRNE BJSANDS MSPONDER KPMAINGAY M ET AL.: "In this model it has been shown that human alpha-synuclein expression leads to neurodegeneration of dopaminergic neurons", CNS SPECTR., vol. 10, no. 3, March 2005 (2005-03-01), pages 235 - 44 |
| YANAMANDRA, PLOS ONE, vol. 6, no. 4, 25 April 2011 (2011-04-25), pages e18513 |
| YELTON ET AL., J. IMMUNOLOGY, vol. 155, 1995, pages 1994 |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20230312693A1 (en) | Agents, Uses and Methods for the Treatment of Synucleinopathy | |
| US12421303B2 (en) | Agents, uses and methods for the treatment of synucleinopathy | |
| HK40127009A (en) | Agents, uses and methods for the treatment of synucleinopathy | |
| CA2989735C (en) | Antibodies binding alpha-synuclein and uses thereof for the treatment of synucleinopathy | |
| HK1254358B (en) | Agents, uses and methods for the treatment of synucleinopathy | |
| OA18689A (en) | Agents, Uses and Methods for the Treatment of Synucleinopathy |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE APPLICATION HAS BEEN PUBLISHED |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R079 Free format text: PREVIOUS MAIN CLASS: A61P0025280000 Ipc: C07K0016180000 |
|
| PUAL | Search report despatched |
Free format text: ORIGINAL CODE: 0009013 |
|
| AC | Divisional application: reference to earlier application |
Ref document number: 3322722 Country of ref document: EP Kind code of ref document: P |
|
| AK | Designated contracting states |
Kind code of ref document: A2 Designated state(s): AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO RS SE SI SK SM TR |
|
| AK | Designated contracting states |
Kind code of ref document: A3 Designated state(s): AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO RS SE SI SK SM TR |
|
| RIC1 | Information provided on ipc code assigned before grant |
Ipc: C07K 16/18 20060101AFI20250610BHEP Ipc: A61K 39/395 20060101ALI20250610BHEP Ipc: A61K 39/00 20060101ALI20250610BHEP Ipc: A61P 25/00 20060101ALI20250610BHEP Ipc: A61P 25/02 20060101ALI20250610BHEP Ipc: A61P 25/16 20060101ALI20250610BHEP Ipc: A61P 25/28 20060101ALI20250610BHEP |
|
| REG | Reference to a national code |
Ref country code: HK Ref legal event code: DE Ref document number: 40127009 Country of ref document: HK |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: REQUEST FOR EXAMINATION WAS MADE |
|
| 17P | Request for examination filed |
Effective date: 20260115 |
|
| RAV | Requested validation state of the european patent: fee paid |
Extension state: MA Effective date: 20260115 |
|
| RAX | Requested extension states of the european patent have changed |
Extension state: BA Payment date: 20260115 Extension state: ME Payment date: 20260115 |