JP5114935B2 - Liquid resin composition for electronic components, and electronic component device using the same - Google Patents
Liquid resin composition for electronic components, and electronic component device using the same Download PDFInfo
- Publication number
- JP5114935B2 JP5114935B2 JP2006332101A JP2006332101A JP5114935B2 JP 5114935 B2 JP5114935 B2 JP 5114935B2 JP 2006332101 A JP2006332101 A JP 2006332101A JP 2006332101 A JP2006332101 A JP 2006332101A JP 5114935 B2 JP5114935 B2 JP 5114935B2
- Authority
- JP
- Japan
- Prior art keywords
- electronic component
- resin composition
- liquid resin
- electronic
- liquid
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 239000007788 liquid Substances 0.000 title claims description 96
- 239000011342 resin composition Substances 0.000 title claims description 66
- 229920000647 polyepoxide Polymers 0.000 claims description 61
- 239000003822 epoxy resin Substances 0.000 claims description 55
- 239000004065 semiconductor Substances 0.000 claims description 37
- 239000003795 chemical substances by application Substances 0.000 claims description 34
- 229920001296 polysiloxane Polymers 0.000 claims description 22
- 229920005989 resin Polymers 0.000 claims description 21
- 239000011347 resin Substances 0.000 claims description 21
- 239000000203 mixture Substances 0.000 claims description 18
- 239000004850 liquid epoxy resins (LERs) Substances 0.000 claims description 14
- 238000007789 sealing Methods 0.000 claims description 14
- 239000000758 substrate Substances 0.000 claims description 12
- LNEPOXFFQSENCJ-UHFFFAOYSA-N haloperidol Chemical class C1CC(O)(C=2C=CC(Cl)=CC=2)CCN1CCCC(=O)C1=CC=C(F)C=C1 LNEPOXFFQSENCJ-UHFFFAOYSA-N 0.000 claims description 11
- 239000011256 inorganic filler Substances 0.000 claims description 10
- 229910003475 inorganic filler Inorganic materials 0.000 claims description 10
- 150000004982 aromatic amines Chemical class 0.000 claims description 9
- 125000003700 epoxy group Chemical group 0.000 claims description 9
- 229910052751 metal Inorganic materials 0.000 claims description 9
- 239000002736 nonionic surfactant Substances 0.000 claims description 9
- 150000002460 imidazoles Chemical class 0.000 claims description 8
- 125000005375 organosiloxane group Chemical group 0.000 claims description 8
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 6
- 125000000524 functional group Chemical group 0.000 claims description 5
- 239000002184 metal Substances 0.000 claims description 5
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 2
- 125000001424 substituent group Chemical group 0.000 claims description 2
- 125000002883 imidazolyl group Chemical group 0.000 claims 2
- -1 glycidyl ester Chemical class 0.000 description 36
- 239000002245 particle Substances 0.000 description 24
- 239000004094 surface-active agent Substances 0.000 description 17
- IISBACLAFKSPIT-UHFFFAOYSA-N bisphenol A Chemical compound C=1C=C(O)C=CC=1C(C)(C)C1=CC=C(O)C=C1 IISBACLAFKSPIT-UHFFFAOYSA-N 0.000 description 15
- 239000000047 product Substances 0.000 description 15
- 239000007787 solid Substances 0.000 description 15
- PXKLMJQFEQBVLD-UHFFFAOYSA-N bisphenol F Chemical compound C1=CC(O)=CC=C1CC1=CC=C(O)C=C1 PXKLMJQFEQBVLD-UHFFFAOYSA-N 0.000 description 14
- 238000002156 mixing Methods 0.000 description 14
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 13
- 239000000126 substance Substances 0.000 description 13
- 230000000694 effects Effects 0.000 description 11
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 10
- 229920001971 elastomer Polymers 0.000 description 10
- 239000005060 rubber Substances 0.000 description 10
- 229910052782 aluminium Inorganic materials 0.000 description 9
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 9
- 150000001412 amines Chemical class 0.000 description 9
- 150000001875 compounds Chemical class 0.000 description 9
- 239000000463 material Substances 0.000 description 9
- 238000000034 method Methods 0.000 description 9
- 238000000465 moulding Methods 0.000 description 9
- 229910000679 solder Inorganic materials 0.000 description 9
- 238000012360 testing method Methods 0.000 description 9
- 238000006243 chemical reaction Methods 0.000 description 7
- 230000000052 comparative effect Effects 0.000 description 7
- 230000035882 stress Effects 0.000 description 7
- PISLZQACAJMAIO-UHFFFAOYSA-N 2,4-diethyl-6-methylbenzene-1,3-diamine Chemical compound CCC1=CC(C)=C(N)C(CC)=C1N PISLZQACAJMAIO-UHFFFAOYSA-N 0.000 description 6
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 6
- 239000000853 adhesive Substances 0.000 description 6
- 230000001070 adhesive effect Effects 0.000 description 6
- 238000011156 evaluation Methods 0.000 description 6
- 229930185605 Bisphenol Natural products 0.000 description 5
- 239000004593 Epoxy Substances 0.000 description 5
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 5
- 150000008065 acid anhydrides Chemical class 0.000 description 5
- 235000014113 dietary fatty acids Nutrition 0.000 description 5
- 239000000194 fatty acid Substances 0.000 description 5
- 229930195729 fatty acid Natural products 0.000 description 5
- 239000003063 flame retardant Substances 0.000 description 5
- 239000011521 glass Substances 0.000 description 5
- XEEYBQQBJWHFJM-UHFFFAOYSA-N iron Substances [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 5
- 239000010410 layer Substances 0.000 description 5
- 229920003986 novolac Polymers 0.000 description 5
- 229920001721 polyimide Polymers 0.000 description 5
- 230000035939 shock Effects 0.000 description 5
- 238000003860 storage Methods 0.000 description 5
- 239000011800 void material Substances 0.000 description 5
- BRLQWZUYTZBJKN-UHFFFAOYSA-N Epichlorohydrin Chemical compound ClCC1CO1 BRLQWZUYTZBJKN-UHFFFAOYSA-N 0.000 description 4
- KFSLWBXXFJQRDL-UHFFFAOYSA-N Peracetic acid Chemical compound CC(=O)OO KFSLWBXXFJQRDL-UHFFFAOYSA-N 0.000 description 4
- UUQQGGWZVKUCBD-UHFFFAOYSA-N [4-(hydroxymethyl)-2-phenyl-1h-imidazol-5-yl]methanol Chemical compound N1C(CO)=C(CO)N=C1C1=CC=CC=C1 UUQQGGWZVKUCBD-UHFFFAOYSA-N 0.000 description 4
- 239000011258 core-shell material Substances 0.000 description 4
- 239000007822 coupling agent Substances 0.000 description 4
- 239000003085 diluting agent Substances 0.000 description 4
- 238000010438 heat treatment Methods 0.000 description 4
- ZFSLODLOARCGLH-UHFFFAOYSA-N isocyanuric acid Chemical compound OC1=NC(O)=NC(O)=N1 ZFSLODLOARCGLH-UHFFFAOYSA-N 0.000 description 4
- 239000011777 magnesium Substances 0.000 description 4
- QWVGKYWNOKOFNN-UHFFFAOYSA-N o-cresol Chemical compound CC1=CC=CC=C1O QWVGKYWNOKOFNN-UHFFFAOYSA-N 0.000 description 4
- 230000035699 permeability Effects 0.000 description 4
- XNGIFLGASWRNHJ-UHFFFAOYSA-N phthalic acid Chemical compound OC(=O)C1=CC=CC=C1C(O)=O XNGIFLGASWRNHJ-UHFFFAOYSA-N 0.000 description 4
- RIOQSEWOXXDEQQ-UHFFFAOYSA-N triphenylphosphine Chemical compound C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 RIOQSEWOXXDEQQ-UHFFFAOYSA-N 0.000 description 4
- RUEBPOOTFCZRBC-UHFFFAOYSA-N (5-methyl-2-phenyl-1h-imidazol-4-yl)methanol Chemical compound OCC1=C(C)NC(C=2C=CC=CC=2)=N1 RUEBPOOTFCZRBC-UHFFFAOYSA-N 0.000 description 3
- AZQWKYJCGOJGHM-UHFFFAOYSA-N 1,4-benzoquinone Chemical compound O=C1C=CC(=O)C=C1 AZQWKYJCGOJGHM-UHFFFAOYSA-N 0.000 description 3
- RNFJDJUURJAICM-UHFFFAOYSA-N 2,2,4,4,6,6-hexaphenoxy-1,3,5-triaza-2$l^{5},4$l^{5},6$l^{5}-triphosphacyclohexa-1,3,5-triene Chemical compound N=1P(OC=2C=CC=CC=2)(OC=2C=CC=CC=2)=NP(OC=2C=CC=CC=2)(OC=2C=CC=CC=2)=NP=1(OC=1C=CC=CC=1)OC1=CC=CC=C1 RNFJDJUURJAICM-UHFFFAOYSA-N 0.000 description 3
- 229910017944 Ag—Cu Inorganic materials 0.000 description 3
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 3
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 3
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 description 3
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 3
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 3
- UMHKOAYRTRADAT-UHFFFAOYSA-N [hydroxy(octoxy)phosphoryl] octyl hydrogen phosphate Chemical compound CCCCCCCCOP(O)(=O)OP(O)(=O)OCCCCCCCC UMHKOAYRTRADAT-UHFFFAOYSA-N 0.000 description 3
- 230000001133 acceleration Effects 0.000 description 3
- 150000005215 alkyl ethers Chemical class 0.000 description 3
- 238000013329 compounding Methods 0.000 description 3
- 230000002950 deficient Effects 0.000 description 3
- GYZLOYUZLJXAJU-UHFFFAOYSA-N diglycidyl ether Chemical compound C1OC1COCC1CO1 GYZLOYUZLJXAJU-UHFFFAOYSA-N 0.000 description 3
- 239000005350 fused silica glass Substances 0.000 description 3
- 238000005227 gel permeation chromatography Methods 0.000 description 3
- 229910052739 hydrogen Inorganic materials 0.000 description 3
- 239000001257 hydrogen Substances 0.000 description 3
- 238000005470 impregnation Methods 0.000 description 3
- 150000002500 ions Chemical class 0.000 description 3
- 229910052742 iron Inorganic materials 0.000 description 3
- 229910052749 magnesium Inorganic materials 0.000 description 3
- 238000005259 measurement Methods 0.000 description 3
- 229910052759 nickel Inorganic materials 0.000 description 3
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N phenol group Chemical group C1(=CC=CC=C1)O ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 3
- 229920000058 polyacrylate Polymers 0.000 description 3
- 239000011164 primary particle Substances 0.000 description 3
- 230000009467 reduction Effects 0.000 description 3
- 239000000377 silicon dioxide Substances 0.000 description 3
- 229920002379 silicone rubber Polymers 0.000 description 3
- 239000004945 silicone rubber Substances 0.000 description 3
- 229910052725 zinc Inorganic materials 0.000 description 3
- 239000011701 zinc Substances 0.000 description 3
- 229940005561 1,4-benzoquinone Drugs 0.000 description 2
- FRASJONUBLZVQX-UHFFFAOYSA-N 1,4-naphthoquinone Chemical compound C1=CC=C2C(=O)C=CC(=O)C2=C1 FRASJONUBLZVQX-UHFFFAOYSA-N 0.000 description 2
- NADHCXOXVRHBHC-UHFFFAOYSA-N 2,3-dimethoxycyclohexa-2,5-diene-1,4-dione Chemical compound COC1=C(OC)C(=O)C=CC1=O NADHCXOXVRHBHC-UHFFFAOYSA-N 0.000 description 2
- VPWNQTHUCYMVMZ-UHFFFAOYSA-N 4,4'-sulfonyldiphenol Chemical compound C1=CC(O)=CC=C1S(=O)(=O)C1=CC=C(O)C=C1 VPWNQTHUCYMVMZ-UHFFFAOYSA-N 0.000 description 2
- RQEOBXYYEPMCPJ-UHFFFAOYSA-N 4,6-diethyl-2-methylbenzene-1,3-diamine Chemical compound CCC1=CC(CC)=C(N)C(C)=C1N RQEOBXYYEPMCPJ-UHFFFAOYSA-N 0.000 description 2
- PLIKAWJENQZMHA-UHFFFAOYSA-N 4-aminophenol Chemical compound NC1=CC=C(O)C=C1 PLIKAWJENQZMHA-UHFFFAOYSA-N 0.000 description 2
- LTPBRCUWZOMYOC-UHFFFAOYSA-N Beryllium oxide Chemical compound O=[Be] LTPBRCUWZOMYOC-UHFFFAOYSA-N 0.000 description 2
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 2
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 2
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 2
- UQSXHKLRYXJYBZ-UHFFFAOYSA-N Iron oxide Chemical compound [Fe]=O UQSXHKLRYXJYBZ-UHFFFAOYSA-N 0.000 description 2
- 229920000877 Melamine resin Polymers 0.000 description 2
- 229920000459 Nitrile rubber Polymers 0.000 description 2
- 239000005062 Polybutadiene Substances 0.000 description 2
- 239000004642 Polyimide Substances 0.000 description 2
- 239000006087 Silane Coupling Agent Substances 0.000 description 2
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 2
- 229920006311 Urethane elastomer Polymers 0.000 description 2
- XLOMVQKBTHCTTD-UHFFFAOYSA-N Zinc monoxide Chemical compound [Zn]=O XLOMVQKBTHCTTD-UHFFFAOYSA-N 0.000 description 2
- MCMNRKCIXSYSNV-UHFFFAOYSA-N Zirconium dioxide Chemical compound O=[Zr]=O MCMNRKCIXSYSNV-UHFFFAOYSA-N 0.000 description 2
- 239000002253 acid Substances 0.000 description 2
- 229920000800 acrylic rubber Polymers 0.000 description 2
- 239000012190 activator Substances 0.000 description 2
- 239000000654 additive Substances 0.000 description 2
- 150000001299 aldehydes Chemical class 0.000 description 2
- 125000002723 alicyclic group Chemical group 0.000 description 2
- 239000004844 aliphatic epoxy resin Substances 0.000 description 2
- WNROFYMDJYEPJX-UHFFFAOYSA-K aluminium hydroxide Chemical compound [OH-].[OH-].[OH-].[Al+3] WNROFYMDJYEPJX-UHFFFAOYSA-K 0.000 description 2
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 2
- 125000003277 amino group Chemical group 0.000 description 2
- 150000001450 anions Chemical class 0.000 description 2
- 229910052787 antimony Inorganic materials 0.000 description 2
- WATWJIUSRGPENY-UHFFFAOYSA-N antimony atom Chemical compound [Sb] WATWJIUSRGPENY-UHFFFAOYSA-N 0.000 description 2
- ADCOVFLJGNWWNZ-UHFFFAOYSA-N antimony trioxide Chemical compound O=[Sb]O[Sb]=O ADCOVFLJGNWWNZ-UHFFFAOYSA-N 0.000 description 2
- 239000006229 carbon black Substances 0.000 description 2
- 239000000460 chlorine Substances 0.000 description 2
- 229910052801 chlorine Inorganic materials 0.000 description 2
- 239000011248 coating agent Substances 0.000 description 2
- 238000000576 coating method Methods 0.000 description 2
- 229910017052 cobalt Inorganic materials 0.000 description 2
- 239000010941 cobalt Substances 0.000 description 2
- GUTLYIVDDKVIGB-UHFFFAOYSA-N cobalt atom Chemical compound [Co] GUTLYIVDDKVIGB-UHFFFAOYSA-N 0.000 description 2
- 239000003086 colorant Substances 0.000 description 2
- 229910052802 copper Inorganic materials 0.000 description 2
- 239000010949 copper Substances 0.000 description 2
- 150000007973 cyanuric acids Chemical class 0.000 description 2
- 239000000539 dimer Substances 0.000 description 2
- ZZTCPWRAHWXWCH-UHFFFAOYSA-N diphenylmethanediamine Chemical compound C=1C=CC=CC=1C(N)(N)C1=CC=CC=C1 ZZTCPWRAHWXWCH-UHFFFAOYSA-N 0.000 description 2
- 239000000945 filler Substances 0.000 description 2
- 238000011049 filling Methods 0.000 description 2
- 238000005040 ion trap Methods 0.000 description 2
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 2
- VTHJTEIRLNZDEV-UHFFFAOYSA-L magnesium dihydroxide Chemical compound [OH-].[OH-].[Mg+2] VTHJTEIRLNZDEV-UHFFFAOYSA-L 0.000 description 2
- 239000000347 magnesium hydroxide Substances 0.000 description 2
- 229910001862 magnesium hydroxide Inorganic materials 0.000 description 2
- 229910000000 metal hydroxide Inorganic materials 0.000 description 2
- 150000004692 metal hydroxides Chemical class 0.000 description 2
- 239000003094 microcapsule Substances 0.000 description 2
- 238000013508 migration Methods 0.000 description 2
- 230000005012 migration Effects 0.000 description 2
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 description 2
- 230000001590 oxidative effect Effects 0.000 description 2
- AFEQENGXSMURHA-UHFFFAOYSA-N oxiran-2-ylmethanamine Chemical compound NCC1CO1 AFEQENGXSMURHA-UHFFFAOYSA-N 0.000 description 2
- 238000002161 passivation Methods 0.000 description 2
- 230000035515 penetration Effects 0.000 description 2
- 150000004965 peroxy acids Chemical class 0.000 description 2
- 239000005011 phenolic resin Substances 0.000 description 2
- 150000002989 phenols Chemical class 0.000 description 2
- 229910052698 phosphorus Inorganic materials 0.000 description 2
- 239000011574 phosphorus Substances 0.000 description 2
- 229920002857 polybutadiene Polymers 0.000 description 2
- 150000007519 polyprotic acids Polymers 0.000 description 2
- 150000004756 silanes Chemical class 0.000 description 2
- 229920005573 silicon-containing polymer Polymers 0.000 description 2
- 229910052719 titanium Inorganic materials 0.000 description 2
- 239000010936 titanium Substances 0.000 description 2
- IMNIMPAHZVJRPE-UHFFFAOYSA-N triethylenediamine Chemical compound C1CN2CCN1CC2 IMNIMPAHZVJRPE-UHFFFAOYSA-N 0.000 description 2
- BIKXLKXABVUSMH-UHFFFAOYSA-N trizinc;diborate Chemical compound [Zn+2].[Zn+2].[Zn+2].[O-]B([O-])[O-].[O-]B([O-])[O-] BIKXLKXABVUSMH-UHFFFAOYSA-N 0.000 description 2
- XAEWLETZEZXLHR-UHFFFAOYSA-N zinc;dioxido(dioxo)molybdenum Chemical compound [Zn+2].[O-][Mo]([O-])(=O)=O XAEWLETZEZXLHR-UHFFFAOYSA-N 0.000 description 2
- VOORKBJLBDXKGM-UHFFFAOYSA-N (2,3-diethylphenyl)-phenylmethanediamine Chemical compound CCC1=CC=CC(C(N)(N)C=2C=CC=CC=2)=C1CC VOORKBJLBDXKGM-UHFFFAOYSA-N 0.000 description 1
- WYTZZXDRDKSJID-UHFFFAOYSA-N (3-aminopropyl)triethoxysilane Chemical compound CCO[Si](OCC)(OCC)CCCN WYTZZXDRDKSJID-UHFFFAOYSA-N 0.000 description 1
- JYEUMXHLPRZUAT-UHFFFAOYSA-N 1,2,3-triazine Chemical group C1=CN=NN=C1 JYEUMXHLPRZUAT-UHFFFAOYSA-N 0.000 description 1
- XGINAUQXFXVBND-UHFFFAOYSA-N 1,2,6,7,8,8a-hexahydropyrrolo[1,2-a]pyrimidine Chemical compound N1CC=CN2CCCC21 XGINAUQXFXVBND-UHFFFAOYSA-N 0.000 description 1
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 1
- FBHPRUXJQNWTEW-UHFFFAOYSA-N 1-benzyl-2-methylimidazole Chemical compound CC1=NC=CN1CC1=CC=CC=C1 FBHPRUXJQNWTEW-UHFFFAOYSA-N 0.000 description 1
- XZKLXPPYISZJCV-UHFFFAOYSA-N 1-benzyl-2-phenylimidazole Chemical compound C1=CN=C(C=2C=CC=CC=2)N1CC1=CC=CC=C1 XZKLXPPYISZJCV-UHFFFAOYSA-N 0.000 description 1
- HIQAWCBKWSQMRQ-UHFFFAOYSA-N 16-methylheptadecanoic acid;2-methylprop-2-enoic acid;propan-2-ol;titanium Chemical compound [Ti].CC(C)O.CC(=C)C(O)=O.CC(=C)C(O)=O.CC(C)CCCCCCCCCCCCCCC(O)=O HIQAWCBKWSQMRQ-UHFFFAOYSA-N 0.000 description 1
- IEKHISJGRIEHRE-UHFFFAOYSA-N 16-methylheptadecanoic acid;propan-2-ol;titanium Chemical compound [Ti].CC(C)O.CC(C)CCCCCCCCCCCCCCC(O)=O.CC(C)CCCCCCCCCCCCCCC(O)=O.CC(C)CCCCCCCCCCCCCCC(O)=O IEKHISJGRIEHRE-UHFFFAOYSA-N 0.000 description 1
- ZEGDFCCYTFPECB-UHFFFAOYSA-N 2,3-dimethoxy-1,4-benzoquinone Natural products C1=CC=C2C(=O)C(OC)=C(OC)C(=O)C2=C1 ZEGDFCCYTFPECB-UHFFFAOYSA-N 0.000 description 1
- AIACLXROWHONEE-UHFFFAOYSA-N 2,3-dimethylcyclohexa-2,5-diene-1,4-dione Chemical compound CC1=C(C)C(=O)C=CC1=O AIACLXROWHONEE-UHFFFAOYSA-N 0.000 description 1
- JGYUBHGXADMAQU-UHFFFAOYSA-N 2,4,6-triethylbenzene-1,3-diamine Chemical compound CCC1=CC(CC)=C(N)C(CC)=C1N JGYUBHGXADMAQU-UHFFFAOYSA-N 0.000 description 1
- AHDSRXYHVZECER-UHFFFAOYSA-N 2,4,6-tris[(dimethylamino)methyl]phenol Chemical compound CN(C)CC1=CC(CN(C)C)=C(O)C(CN(C)C)=C1 AHDSRXYHVZECER-UHFFFAOYSA-N 0.000 description 1
- SENUUPBBLQWHMF-UHFFFAOYSA-N 2,6-dimethylcyclohexa-2,5-diene-1,4-dione Chemical compound CC1=CC(=O)C=C(C)C1=O SENUUPBBLQWHMF-UHFFFAOYSA-N 0.000 description 1
- YSUQLAYJZDEMOT-UHFFFAOYSA-N 2-(butoxymethyl)oxirane Chemical compound CCCCOCC1CO1 YSUQLAYJZDEMOT-UHFFFAOYSA-N 0.000 description 1
- YCUKMYFJDGKQFC-UHFFFAOYSA-N 2-(octan-3-yloxymethyl)oxirane Chemical compound CCCCCC(CC)OCC1CO1 YCUKMYFJDGKQFC-UHFFFAOYSA-N 0.000 description 1
- HJEORQYOUWYAMR-UHFFFAOYSA-N 2-[(2-butylphenoxy)methyl]oxirane Chemical compound CCCCC1=CC=CC=C1OCC1OC1 HJEORQYOUWYAMR-UHFFFAOYSA-N 0.000 description 1
- SEFYJVFBMNOLBK-UHFFFAOYSA-N 2-[2-[2-(oxiran-2-ylmethoxy)ethoxy]ethoxymethyl]oxirane Chemical compound C1OC1COCCOCCOCC1CO1 SEFYJVFBMNOLBK-UHFFFAOYSA-N 0.000 description 1
- WTYYGFLRBWMFRY-UHFFFAOYSA-N 2-[6-(oxiran-2-ylmethoxy)hexoxymethyl]oxirane Chemical compound C1OC1COCCCCCCOCC1CO1 WTYYGFLRBWMFRY-UHFFFAOYSA-N 0.000 description 1
- CDAWCLOXVUBKRW-UHFFFAOYSA-N 2-aminophenol Chemical compound NC1=CC=CC=C1O CDAWCLOXVUBKRW-UHFFFAOYSA-N 0.000 description 1
- LYNVLWLRSACENL-UHFFFAOYSA-N 2-decyl-1h-imidazole Chemical compound CCCCCCCCCCC1=NC=CN1 LYNVLWLRSACENL-UHFFFAOYSA-N 0.000 description 1
- KKOHCQAVIJDYAF-UHFFFAOYSA-N 2-dodecylbenzenesulfonic acid;propan-2-ol;titanium Chemical compound [Ti].CC(C)O.CCCCCCCCCCCCC1=CC=CC=C1S(O)(=O)=O.CCCCCCCCCCCCC1=CC=CC=C1S(O)(=O)=O.CCCCCCCCCCCCC1=CC=CC=C1S(O)(=O)=O KKOHCQAVIJDYAF-UHFFFAOYSA-N 0.000 description 1
- YTWBFUCJVWKCCK-UHFFFAOYSA-N 2-heptadecyl-1h-imidazole Chemical compound CCCCCCCCCCCCCCCCCC1=NC=CN1 YTWBFUCJVWKCCK-UHFFFAOYSA-N 0.000 description 1
- LXBGSDVWAMZHDD-UHFFFAOYSA-N 2-methyl-1h-imidazole Chemical compound CC1=NC=CN1 LXBGSDVWAMZHDD-UHFFFAOYSA-N 0.000 description 1
- VTWDKFNVVLAELH-UHFFFAOYSA-N 2-methylcyclohexa-2,5-diene-1,4-dione Chemical compound CC1=CC(=O)C=CC1=O VTWDKFNVVLAELH-UHFFFAOYSA-N 0.000 description 1
- ZCUJYXPAKHMBAZ-UHFFFAOYSA-N 2-phenyl-1h-imidazole Chemical compound C1=CNC(C=2C=CC=CC=2)=N1 ZCUJYXPAKHMBAZ-UHFFFAOYSA-N 0.000 description 1
- RLQZIECDMISZHS-UHFFFAOYSA-N 2-phenylcyclohexa-2,5-diene-1,4-dione Chemical compound O=C1C=CC(=O)C(C=2C=CC=CC=2)=C1 RLQZIECDMISZHS-UHFFFAOYSA-N 0.000 description 1
- HXLAEGYMDGUSBD-UHFFFAOYSA-N 3-[diethoxy(methyl)silyl]propan-1-amine Chemical compound CCO[Si](C)(OCC)CCCN HXLAEGYMDGUSBD-UHFFFAOYSA-N 0.000 description 1
- ZYAASQNKCWTPKI-UHFFFAOYSA-N 3-[dimethoxy(methyl)silyl]propan-1-amine Chemical compound CO[Si](C)(OC)CCCN ZYAASQNKCWTPKI-UHFFFAOYSA-N 0.000 description 1
- IKYAJDOSWUATPI-UHFFFAOYSA-N 3-[dimethoxy(methyl)silyl]propane-1-thiol Chemical compound CO[Si](C)(OC)CCCS IKYAJDOSWUATPI-UHFFFAOYSA-N 0.000 description 1
- OXYZDRAJMHGSMW-UHFFFAOYSA-N 3-chloropropyl(trimethoxy)silane Chemical compound CO[Si](OC)(OC)CCCCl OXYZDRAJMHGSMW-UHFFFAOYSA-N 0.000 description 1
- MECNWXGGNCJFQJ-UHFFFAOYSA-N 3-piperidin-1-ylpropane-1,2-diol Chemical compound OCC(O)CN1CCCCC1 MECNWXGGNCJFQJ-UHFFFAOYSA-N 0.000 description 1
- SJECZPVISLOESU-UHFFFAOYSA-N 3-trimethoxysilylpropan-1-amine Chemical compound CO[Si](OC)(OC)CCCN SJECZPVISLOESU-UHFFFAOYSA-N 0.000 description 1
- UUEWCQRISZBELL-UHFFFAOYSA-N 3-trimethoxysilylpropane-1-thiol Chemical compound CO[Si](OC)(OC)CCCS UUEWCQRISZBELL-UHFFFAOYSA-N 0.000 description 1
- XDLMVUHYZWKMMD-UHFFFAOYSA-N 3-trimethoxysilylpropyl 2-methylprop-2-enoate Chemical compound CO[Si](OC)(OC)CCCOC(=O)C(C)=C XDLMVUHYZWKMMD-UHFFFAOYSA-N 0.000 description 1
- JNRLEMMIVRBKJE-UHFFFAOYSA-N 4,4'-Methylenebis(N,N-dimethylaniline) Chemical compound C1=CC(N(C)C)=CC=C1CC1=CC=C(N(C)C)C=C1 JNRLEMMIVRBKJE-UHFFFAOYSA-N 0.000 description 1
- TYOXIFXYEIILLY-UHFFFAOYSA-N 5-methyl-2-phenyl-1h-imidazole Chemical compound N1C(C)=CN=C1C1=CC=CC=C1 TYOXIFXYEIILLY-UHFFFAOYSA-N 0.000 description 1
- ULKLGIFJWFIQFF-UHFFFAOYSA-N 5K8XI641G3 Chemical compound CCC1=NC=C(C)N1 ULKLGIFJWFIQFF-UHFFFAOYSA-N 0.000 description 1
- OAOABCKPVCUNKO-UHFFFAOYSA-N 8-methyl Nonanoic acid Chemical compound CC(C)CCCCCCC(O)=O OAOABCKPVCUNKO-UHFFFAOYSA-N 0.000 description 1
- 239000004925 Acrylic resin Substances 0.000 description 1
- 229920000178 Acrylic resin Polymers 0.000 description 1
- 229910052582 BN Inorganic materials 0.000 description 1
- PZNSFCLAULLKQX-UHFFFAOYSA-N Boron nitride Chemical compound N#B PZNSFCLAULLKQX-UHFFFAOYSA-N 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 1
- VGGSQFUCUMXWEO-UHFFFAOYSA-N Ethene Chemical compound C=C VGGSQFUCUMXWEO-UHFFFAOYSA-N 0.000 description 1
- 239000005977 Ethylene Substances 0.000 description 1
- PIICEJLVQHRZGT-UHFFFAOYSA-N Ethylenediamine Chemical compound NCCN PIICEJLVQHRZGT-UHFFFAOYSA-N 0.000 description 1
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerol Natural products OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 1
- UEEJHVSXFDXPFK-UHFFFAOYSA-N N-dimethylaminoethanol Chemical compound CN(C)CCO UEEJHVSXFDXPFK-UHFFFAOYSA-N 0.000 description 1
- 229910052779 Neodymium Inorganic materials 0.000 description 1
- FQYUMYWMJTYZTK-UHFFFAOYSA-N Phenyl glycidyl ether Chemical compound C1OC1COC1=CC=CC=C1 FQYUMYWMJTYZTK-UHFFFAOYSA-N 0.000 description 1
- 229920001214 Polysorbate 60 Polymers 0.000 description 1
- 239000004793 Polystyrene Substances 0.000 description 1
- 229910052581 Si3N4 Inorganic materials 0.000 description 1
- BLRPTPMANUNPDV-UHFFFAOYSA-N Silane Chemical compound [SiH4] BLRPTPMANUNPDV-UHFFFAOYSA-N 0.000 description 1
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 1
- AWMVMTVKBNGEAK-UHFFFAOYSA-N Styrene oxide Chemical compound C1OC1C1=CC=CC=C1 AWMVMTVKBNGEAK-UHFFFAOYSA-N 0.000 description 1
- ATJFFYVFTNAWJD-UHFFFAOYSA-N Tin Chemical compound [Sn] ATJFFYVFTNAWJD-UHFFFAOYSA-N 0.000 description 1
- GSEJCLTVZPLZKY-UHFFFAOYSA-N Triethanolamine Chemical compound OCCN(CCO)CCO GSEJCLTVZPLZKY-UHFFFAOYSA-N 0.000 description 1
- 239000007983 Tris buffer Substances 0.000 description 1
- QCWXUUIWCKQGHC-UHFFFAOYSA-N Zirconium Chemical compound [Zr] QCWXUUIWCKQGHC-UHFFFAOYSA-N 0.000 description 1
- NOZAQBYNLKNDRT-UHFFFAOYSA-N [diacetyloxy(ethenyl)silyl] acetate Chemical compound CC(=O)O[Si](OC(C)=O)(OC(C)=O)C=C NOZAQBYNLKNDRT-UHFFFAOYSA-N 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- NIXOWILDQLNWCW-UHFFFAOYSA-N acrylic acid group Chemical group C(C=C)(=O)O NIXOWILDQLNWCW-UHFFFAOYSA-N 0.000 description 1
- 230000000996 additive effect Effects 0.000 description 1
- 229910052784 alkaline earth metal Inorganic materials 0.000 description 1
- 150000003973 alkyl amines Chemical class 0.000 description 1
- 125000000217 alkyl group Chemical group 0.000 description 1
- 150000001343 alkyl silanes Chemical class 0.000 description 1
- 239000002518 antifoaming agent Substances 0.000 description 1
- 125000003118 aryl group Chemical group 0.000 description 1
- 239000011324 bead Substances 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 229910052797 bismuth Inorganic materials 0.000 description 1
- JCXGWMGPZLAOME-UHFFFAOYSA-N bismuth atom Chemical compound [Bi] JCXGWMGPZLAOME-UHFFFAOYSA-N 0.000 description 1
- 229910001451 bismuth ion Inorganic materials 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 229910000019 calcium carbonate Inorganic materials 0.000 description 1
- 239000000378 calcium silicate Substances 0.000 description 1
- 229910052918 calcium silicate Inorganic materials 0.000 description 1
- OYACROKNLOSFPA-UHFFFAOYSA-N calcium;dioxido(oxo)silane Chemical compound [Ca+2].[O-][Si]([O-])=O OYACROKNLOSFPA-UHFFFAOYSA-N 0.000 description 1
- 238000011088 calibration curve Methods 0.000 description 1
- 239000003990 capacitor Substances 0.000 description 1
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 1
- 239000000969 carrier Substances 0.000 description 1
- 238000005266 casting Methods 0.000 description 1
- 239000000919 ceramic Substances 0.000 description 1
- 239000007795 chemical reaction product Substances 0.000 description 1
- 239000004927 clay Substances 0.000 description 1
- 229910052570 clay Inorganic materials 0.000 description 1
- 239000002131 composite material Substances 0.000 description 1
- 238000009833 condensation Methods 0.000 description 1
- 230000005494 condensation Effects 0.000 description 1
- 238000010276 construction Methods 0.000 description 1
- PMHQVHHXPFUNSP-UHFFFAOYSA-M copper(1+);methylsulfanylmethane;bromide Chemical compound Br[Cu].CSC PMHQVHHXPFUNSP-UHFFFAOYSA-M 0.000 description 1
- 230000007797 corrosion Effects 0.000 description 1
- 238000005260 corrosion Methods 0.000 description 1
- 238000004132 cross linking Methods 0.000 description 1
- 229910002026 crystalline silica Inorganic materials 0.000 description 1
- 230000001351 cycling effect Effects 0.000 description 1
- 229960002887 deanol Drugs 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- GDVKFRBCXAPAQJ-UHFFFAOYSA-A dialuminum;hexamagnesium;carbonate;hexadecahydroxide Chemical compound [OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[Mg+2].[Mg+2].[Mg+2].[Mg+2].[Mg+2].[Mg+2].[Al+3].[Al+3].[O-]C([O-])=O GDVKFRBCXAPAQJ-UHFFFAOYSA-A 0.000 description 1
- CRGRWBQSZSQVIE-UHFFFAOYSA-N diazomethylbenzene Chemical compound [N-]=[N+]=CC1=CC=CC=C1 CRGRWBQSZSQVIE-UHFFFAOYSA-N 0.000 description 1
- JJQZDUKDJDQPMQ-UHFFFAOYSA-N dimethoxy(dimethyl)silane Chemical compound CO[Si](C)(C)OC JJQZDUKDJDQPMQ-UHFFFAOYSA-N 0.000 description 1
- WHGNXNCOTZPEEK-UHFFFAOYSA-N dimethoxy-methyl-[3-(oxiran-2-ylmethoxy)propyl]silane Chemical compound CO[Si](C)(OC)CCCOCC1CO1 WHGNXNCOTZPEEK-UHFFFAOYSA-N 0.000 description 1
- 125000000118 dimethyl group Chemical group [H]C([H])([H])* 0.000 description 1
- 239000004205 dimethyl polysiloxane Substances 0.000 description 1
- HASCQPSFPAKVEK-UHFFFAOYSA-N dimethyl(phenyl)phosphine Chemical compound CP(C)C1=CC=CC=C1 HASCQPSFPAKVEK-UHFFFAOYSA-N 0.000 description 1
- XXBDWLFCJWSEKW-UHFFFAOYSA-N dimethylbenzylamine Chemical compound CN(C)CC1=CC=CC=C1 XXBDWLFCJWSEKW-UHFFFAOYSA-N 0.000 description 1
- HTDKEJXHILZNPP-UHFFFAOYSA-N dioctyl hydrogen phosphate Chemical compound CCCCCCCCOP(O)(=O)OCCCCCCCC HTDKEJXHILZNPP-UHFFFAOYSA-N 0.000 description 1
- XMQYIPNJVLNWOE-UHFFFAOYSA-N dioctyl hydrogen phosphite Chemical compound CCCCCCCCOP(O)OCCCCCCCC XMQYIPNJVLNWOE-UHFFFAOYSA-N 0.000 description 1
- KZHJGOXRZJKJNY-UHFFFAOYSA-N dioxosilane;oxo(oxoalumanyloxy)alumane Chemical compound O=[Si]=O.O=[Si]=O.O=[Al]O[Al]=O.O=[Al]O[Al]=O.O=[Al]O[Al]=O KZHJGOXRZJKJNY-UHFFFAOYSA-N 0.000 description 1
- NJLLQSBAHIKGKF-UHFFFAOYSA-N dipotassium dioxido(oxo)titanium Chemical compound [K+].[K+].[O-][Ti]([O-])=O NJLLQSBAHIKGKF-UHFFFAOYSA-N 0.000 description 1
- KPUWHANPEXNPJT-UHFFFAOYSA-N disiloxane Chemical class [SiH3]O[SiH3] KPUWHANPEXNPJT-UHFFFAOYSA-N 0.000 description 1
- VTIXMGZYGRZMAW-UHFFFAOYSA-N ditridecyl hydrogen phosphite Chemical compound CCCCCCCCCCCCCOP(O)OCCCCCCCCCCCCC VTIXMGZYGRZMAW-UHFFFAOYSA-N 0.000 description 1
- XHWQYYPUYFYELO-UHFFFAOYSA-N ditridecyl phosphite Chemical compound CCCCCCCCCCCCCOP([O-])OCCCCCCCCCCCCC XHWQYYPUYFYELO-UHFFFAOYSA-N 0.000 description 1
- 239000000975 dye Substances 0.000 description 1
- 239000003480 eluent Substances 0.000 description 1
- 238000007720 emulsion polymerization reaction Methods 0.000 description 1
- FWDBOZPQNFPOLF-UHFFFAOYSA-N ethenyl(triethoxy)silane Chemical compound CCO[Si](OCC)(OCC)C=C FWDBOZPQNFPOLF-UHFFFAOYSA-N 0.000 description 1
- NKSJNEHGWDZZQF-UHFFFAOYSA-N ethenyl(trimethoxy)silane Chemical compound CO[Si](OC)(OC)C=C NKSJNEHGWDZZQF-UHFFFAOYSA-N 0.000 description 1
- WOXXJEVNDJOOLV-UHFFFAOYSA-N ethenyl-tris(2-methoxyethoxy)silane Chemical compound COCCO[Si](OCCOC)(OCCOC)C=C WOXXJEVNDJOOLV-UHFFFAOYSA-N 0.000 description 1
- 230000006355 external stress Effects 0.000 description 1
- 239000010419 fine particle Substances 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 238000007429 general method Methods 0.000 description 1
- 239000003365 glass fiber Substances 0.000 description 1
- 235000011187 glycerol Nutrition 0.000 description 1
- 229910052736 halogen Inorganic materials 0.000 description 1
- NEXSMEBSBIABKL-UHFFFAOYSA-N hexamethyldisilane Chemical compound C[Si](C)(C)[Si](C)(C)C NEXSMEBSBIABKL-UHFFFAOYSA-N 0.000 description 1
- 229910001701 hydrotalcite Inorganic materials 0.000 description 1
- 229960001545 hydrotalcite Drugs 0.000 description 1
- 150000003949 imides Chemical class 0.000 description 1
- 230000001771 impaired effect Effects 0.000 description 1
- 239000012212 insulator Substances 0.000 description 1
- 230000010354 integration Effects 0.000 description 1
- 239000011229 interlayer Substances 0.000 description 1
- 229940079865 intestinal antiinfectives imidazole derivative Drugs 0.000 description 1
- 238000004898 kneading Methods 0.000 description 1
- FPYJFEHAWHCUMM-UHFFFAOYSA-N maleic anhydride Chemical compound O=C1OC(=O)C=C1 FPYJFEHAWHCUMM-UHFFFAOYSA-N 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- JDSHMPZPIAZGSV-UHFFFAOYSA-N melamine Chemical compound NC1=NC(N)=NC(N)=N1 JDSHMPZPIAZGSV-UHFFFAOYSA-N 0.000 description 1
- 150000007974 melamines Chemical class 0.000 description 1
- 229910044991 metal oxide Inorganic materials 0.000 description 1
- 150000004706 metal oxides Chemical class 0.000 description 1
- UJNZOIKQAUQOCN-UHFFFAOYSA-N methyl(diphenyl)phosphane Chemical compound C=1C=CC=CC=1P(C)C1=CC=CC=C1 UJNZOIKQAUQOCN-UHFFFAOYSA-N 0.000 description 1
- BFXIKLCIZHOAAZ-UHFFFAOYSA-N methyltrimethoxysilane Chemical compound CO[Si](C)(OC)OC BFXIKLCIZHOAAZ-UHFFFAOYSA-N 0.000 description 1
- 229910000476 molybdenum oxide Inorganic materials 0.000 description 1
- 229910052863 mullite Inorganic materials 0.000 description 1
- PHQOGHDTIVQXHL-UHFFFAOYSA-N n'-(3-trimethoxysilylpropyl)ethane-1,2-diamine Chemical compound CO[Si](OC)(OC)CCCNCCN PHQOGHDTIVQXHL-UHFFFAOYSA-N 0.000 description 1
- LIBWSLLLJZULCP-UHFFFAOYSA-N n-(3-triethoxysilylpropyl)aniline Chemical compound CCO[Si](OCC)(OCC)CCCNC1=CC=CC=C1 LIBWSLLLJZULCP-UHFFFAOYSA-N 0.000 description 1
- KBJFYLLAMSZSOG-UHFFFAOYSA-N n-(3-trimethoxysilylpropyl)aniline Chemical compound CO[Si](OC)(OC)CCCNC1=CC=CC=C1 KBJFYLLAMSZSOG-UHFFFAOYSA-N 0.000 description 1
- NXPPAOGUKPJVDI-UHFFFAOYSA-N naphthalene-1,2-diol Chemical compound C1=CC=CC2=C(O)C(O)=CC=C21 NXPPAOGUKPJVDI-UHFFFAOYSA-N 0.000 description 1
- QEFYFXOXNSNQGX-UHFFFAOYSA-N neodymium atom Chemical compound [Nd] QEFYFXOXNSNQGX-UHFFFAOYSA-N 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- JRZJOMJEPLMPRA-UHFFFAOYSA-N olefin Natural products CCCCCCCC=C JRZJOMJEPLMPRA-UHFFFAOYSA-N 0.000 description 1
- 229920000620 organic polymer Polymers 0.000 description 1
- PQQKPALAQIIWST-UHFFFAOYSA-N oxomolybdenum Chemical compound [Mo]=O PQQKPALAQIIWST-UHFFFAOYSA-N 0.000 description 1
- 230000000737 periodic effect Effects 0.000 description 1
- XPPWLXNXHSNMKC-UHFFFAOYSA-N phenylboron Chemical class [B]C1=CC=CC=C1 XPPWLXNXHSNMKC-UHFFFAOYSA-N 0.000 description 1
- 150000003003 phosphines Chemical class 0.000 description 1
- HWLDNSXPUQTBOD-UHFFFAOYSA-N platinum-iridium alloy Chemical compound [Ir].[Pt] HWLDNSXPUQTBOD-UHFFFAOYSA-N 0.000 description 1
- 230000010287 polarization Effects 0.000 description 1
- 229920000435 poly(dimethylsiloxane) Polymers 0.000 description 1
- 229920003217 poly(methylsilsesquioxane) Polymers 0.000 description 1
- 229920001921 poly-methyl-phenyl-siloxane Polymers 0.000 description 1
- 229920000768 polyamine Polymers 0.000 description 1
- 239000004848 polyfunctional curative Substances 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- 229920002223 polystyrene Polymers 0.000 description 1
- 238000003918 potentiometric titration Methods 0.000 description 1
- 238000007639 printing Methods 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- 238000010992 reflux Methods 0.000 description 1
- 239000003566 sealing material Substances 0.000 description 1
- 125000000467 secondary amino group Chemical group [H]N([*:1])[*:2] 0.000 description 1
- 238000004062 sedimentation Methods 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 229910000077 silane Inorganic materials 0.000 description 1
- 239000010703 silicon Substances 0.000 description 1
- 229910052710 silicon Inorganic materials 0.000 description 1
- HBMJWWWQQXIZIP-UHFFFAOYSA-N silicon carbide Chemical compound [Si+]#[C-] HBMJWWWQQXIZIP-UHFFFAOYSA-N 0.000 description 1
- 229910010271 silicon carbide Inorganic materials 0.000 description 1
- 235000012239 silicon dioxide Nutrition 0.000 description 1
- HQVNEWCFYHHQES-UHFFFAOYSA-N silicon nitride Chemical compound N12[Si]34N5[Si]62N3[Si]51N64 HQVNEWCFYHHQES-UHFFFAOYSA-N 0.000 description 1
- 229920002050 silicone resin Polymers 0.000 description 1
- IYMSIPPWHNIMGE-UHFFFAOYSA-N silylurea Chemical compound NC(=O)N[SiH3] IYMSIPPWHNIMGE-UHFFFAOYSA-N 0.000 description 1
- 239000000243 solution Substances 0.000 description 1
- 239000000600 sorbitol Substances 0.000 description 1
- 229910052596 spinel Inorganic materials 0.000 description 1
- 239000011029 spinel Substances 0.000 description 1
- 238000005728 strengthening Methods 0.000 description 1
- 229920003048 styrene butadiene rubber Polymers 0.000 description 1
- TXDNPSYEJHXKMK-UHFFFAOYSA-N sulfanylsilane Chemical compound S[SiH3] TXDNPSYEJHXKMK-UHFFFAOYSA-N 0.000 description 1
- 125000001302 tertiary amino group Chemical group 0.000 description 1
- 238000010998 test method Methods 0.000 description 1
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 1
- 229920001187 thermosetting polymer Polymers 0.000 description 1
- 125000003396 thiol group Chemical group [H]S* 0.000 description 1
- 230000009974 thixotropic effect Effects 0.000 description 1
- 239000011135 tin Substances 0.000 description 1
- 229910052718 tin Inorganic materials 0.000 description 1
- 150000003609 titanium compounds Chemical class 0.000 description 1
- OGIDPMRJRNCKJF-UHFFFAOYSA-N titanium oxide Inorganic materials [Ti]=O OGIDPMRJRNCKJF-UHFFFAOYSA-N 0.000 description 1
- 230000007704 transition Effects 0.000 description 1
- 229910052723 transition metal Inorganic materials 0.000 description 1
- TUQOTMZNTHZOKS-UHFFFAOYSA-N tributylphosphine Chemical compound CCCCP(CCCC)CCCC TUQOTMZNTHZOKS-UHFFFAOYSA-N 0.000 description 1
- GQIUQDDJKHLHTB-UHFFFAOYSA-N trichloro(ethenyl)silane Chemical compound Cl[Si](Cl)(Cl)C=C GQIUQDDJKHLHTB-UHFFFAOYSA-N 0.000 description 1
- CPUDPFPXCZDNGI-UHFFFAOYSA-N triethoxy(methyl)silane Chemical compound CCO[Si](C)(OCC)OCC CPUDPFPXCZDNGI-UHFFFAOYSA-N 0.000 description 1
- DQZNLOXENNXVAD-UHFFFAOYSA-N trimethoxy-[2-(7-oxabicyclo[4.1.0]heptan-4-yl)ethyl]silane Chemical compound C1C(CC[Si](OC)(OC)OC)CCC2OC21 DQZNLOXENNXVAD-UHFFFAOYSA-N 0.000 description 1
- BPSIOYPQMFLKFR-UHFFFAOYSA-N trimethoxy-[3-(oxiran-2-ylmethoxy)propyl]silane Chemical compound CO[Si](OC)(OC)CCCOCC1CO1 BPSIOYPQMFLKFR-UHFFFAOYSA-N 0.000 description 1
- FIQMHBFVRAXMOP-UHFFFAOYSA-N triphenylphosphane oxide Chemical compound C=1C=CC=CC=1P(C=1C=CC=CC=1)(=O)C1=CC=CC=C1 FIQMHBFVRAXMOP-UHFFFAOYSA-N 0.000 description 1
- 239000005050 vinyl trichlorosilane Substances 0.000 description 1
- UKRDPEFKFJNXQM-UHFFFAOYSA-N vinylsilane Chemical compound [SiH3]C=C UKRDPEFKFJNXQM-UHFFFAOYSA-N 0.000 description 1
- 235000012431 wafers Nutrition 0.000 description 1
- 150000003752 zinc compounds Chemical class 0.000 description 1
- 239000011787 zinc oxide Substances 0.000 description 1
- BNEMLSQAJOPTGK-UHFFFAOYSA-N zinc;dioxido(oxo)tin Chemical compound [Zn+2].[O-][Sn]([O-])=O BNEMLSQAJOPTGK-UHFFFAOYSA-N 0.000 description 1
- 229910052845 zircon Inorganic materials 0.000 description 1
- 229910052726 zirconium Inorganic materials 0.000 description 1
- 150000003755 zirconium compounds Chemical class 0.000 description 1
- GFQYVLUOOAAOGM-UHFFFAOYSA-N zirconium(iv) silicate Chemical compound [Zr+4].[O-][Si]([O-])([O-])[O-] GFQYVLUOOAAOGM-UHFFFAOYSA-N 0.000 description 1
Landscapes
- Epoxy Resins (AREA)
- Structures Or Materials For Encapsulating Or Coating Semiconductor Devices Or Solid State Devices (AREA)
- Wire Bonding (AREA)
- Compositions Of Macromolecular Compounds (AREA)
Description
本発明は、厳しい信頼性を要求される電子部品装置の封止用に特に好適な電子部品用液状樹脂組成物、及びこの組成物で封止された素子を備えた電子部品装置に関する。 The present invention relates to a liquid resin composition for electronic components that is particularly suitable for sealing electronic component devices that require strict reliability, and an electronic component device that includes an element sealed with the composition.
従来から、トランジスタ、IC等の電子部品装置の素子封止の分野では生産性、コスト等の面から樹脂封止が主流となり、エポキシ樹脂組成物が広く用いられている。この理由としては、エポキシ樹脂が作業性、成形性、電気特性、耐湿性、耐熱性、機械特性、インサート品との接着性等の諸特性にバランスがとれているためである。COB(Chip on Board)、COG(Chip on Glass)、TCP(Tape Carrier Package)等のベアチップ実装した半導体装置においては電子部品用液状樹脂組成物が封止材として広く使用されている。また、半導体素子をセラミック、ガラス/エポキシ樹脂、ガラス/イミド樹脂またはポリイミドフィルム等を基板とする配線基板上に直接バンプ接続してなる半導体装置(フリップチップ)では、バンプ接続した半導体素子と配線基板の間隙(ギャップ)を充填するアンダーフィル材として電子部品用液状樹脂組成物が使用されている。これらの電子部品用液状樹脂組成物は電子部品を温湿度や機械的な外力から保護するために重要な役割を果たしている。 Conventionally, in the field of element sealing of electronic component devices such as transistors and ICs, resin sealing has been the mainstream in terms of productivity and cost, and epoxy resin compositions have been widely used. This is because the epoxy resin is balanced in various properties such as workability, moldability, electrical properties, moisture resistance, heat resistance, mechanical properties, and adhesiveness with inserts. Liquid resin compositions for electronic components are widely used as sealing materials in semiconductor devices mounted on bare chips such as COB (Chip on Board), COG (Chip on Glass), and TCP (Tape Carrier Package). Further, in a semiconductor device (flip chip) in which a semiconductor element is directly bump-connected to a wiring board using ceramic, glass / epoxy resin, glass / imide resin or polyimide film as a substrate, the bump-connected semiconductor element and wiring board A liquid resin composition for electronic parts is used as an underfill material for filling the gap. These liquid resin compositions for electronic parts play an important role in protecting electronic parts from temperature and humidity and mechanical external force.
また、耐湿接着力、低応力性に優れた封止用エポキシ樹脂組成物、及びこれにより封止された素子を備えた信頼性(耐湿性、耐熱衝撃性)の高い電子部品装置を提供するため、(A)液状エポキシ樹脂、(B)液状芳香族アミンを含む硬化剤、(C)ゴム粒子、(D)無機充填剤を含有してなる封止用エポキシ樹脂組成物、及びこの封止用エポキシ樹脂組成物により封止された素子を備えた電子部品装置が開示されている(例えば特許文献1参照。)。
しかしながら半導体の進歩は著しく、バンプ接続を行うフリップチップ方式ではバンプ数の増加に伴いバンプピッチ、バンプ高さが小さくなり、結果として狭ギャップ化が進んでいる。高集積化に伴いチップサイズも大きくなり、アンダーフィル材には狭ギャップで大面積を流動する特性が求められてきた。また、狭ギャップ化とともにバンプ数が増加し、バンプピッチも狭くなるため、アンダーフィル材の流動経路も複雑になり、ボイドが発生し易くなる。さらには、アンダーフィル材は配線基板と半導体素子の間隙を充填した後、半導体素子周辺部に一定量シミ出し、フィレットを形成するが、フィレットの形状が不均一であると温度サイクル時などにフィレットがクラックしたり、基板或いは半導体素子と剥離を生じたりすることがある。この様なボイドの発生や不均一なフィレットの形成はフリップチップ半導体装置の信頼性に大きな影響を与える。 However, the progress of semiconductors is remarkable, and in the flip-chip method in which bump connection is performed, the bump pitch and bump height are reduced as the number of bumps is increased, and as a result, the gap is being narrowed. With higher integration, the chip size has also increased, and underfill materials have been required to have a characteristic of flowing over a large area with a narrow gap. Further, since the number of bumps is increased and the bump pitch is narrowed along with the narrowing of the gap, the flow path of the underfill material becomes complicated, and voids are easily generated. In addition, the underfill material fills the gap between the wiring board and the semiconductor element, and then a certain amount of spots are formed on the periphery of the semiconductor element to form a fillet. However, if the shape of the fillet is not uniform, the fillet may be damaged during temperature cycling. May crack or peel off from the substrate or semiconductor element. The generation of such voids and the formation of non-uniform fillets greatly affects the reliability of the flip chip semiconductor device.
以上のようにアンダーフィル材を例にとると、半導体の進歩とともに電子部品用液状樹脂組成物にはいろいろな課題の解決が要求されている。本発明は、このような事情に鑑みなされたもので、狭ギャップでの流動性が良好であり、ボイド発生がなく、フィレット形成性に優れた電子部品用液状樹脂組成物、及びこれにより封止された信頼性(耐湿性、耐熱衝撃性)の高い電子部品装置を提供することを目的とする。 As described above, taking the underfill material as an example, the liquid resin composition for electronic parts is required to solve various problems as the semiconductor advances. The present invention has been made in view of such circumstances, a liquid resin composition for electronic parts having excellent fluidity in a narrow gap, no generation of voids, and excellent fillet-forming properties, and sealing by this An object of the present invention is to provide an electronic component device having high reliability (moisture resistance and thermal shock resistance).
本発明者らは上記の課題を解決するために鋭意検討を重ねた結果、硬化剤としては液状芳香族アミンを含む硬化剤と界面活性剤を用いることで上記の目的を達成し本発明を完成するに至った。 As a result of intensive studies to solve the above problems, the present inventors have achieved the above object by using a curing agent containing a liquid aromatic amine and a surfactant as the curing agent, and completed the present invention. It came to do.
本発明は、以下に関する。 The present invention relates to the following.
1.電子部品の封止に用いられる電子部品用液状樹脂組成物であって、(A)液状エポキシ樹脂、(B)液状芳香族アミンを含む硬化剤、(C)非イオン性界面活性剤、(D)無機充填剤を含有する電子部品用液状樹脂組成物。 1. A liquid resin composition for electronic parts used for sealing electronic parts, wherein (A) a liquid epoxy resin, (B) a curing agent containing a liquid aromatic amine, (C) a nonionic surfactant, (D) A liquid resin composition for electronic parts containing an inorganic filler.
2.(C)成分の非イオン性界面活性剤が、エポキシ基と反応する官能基を有し、かつ重量平均分子量500以上5000以下のオルガノシロキサンとエポキシ樹脂との反応で得られるシリコーン変性エポキシ樹脂である前記1.記載の電子部品用液状樹脂組成物。 2. A silicone-modified epoxy resin obtained by a reaction between an epoxy resin and an organosiloxane having a functional group that reacts with an epoxy group and a nonionic surfactant as the component (C) having a weight average molecular weight of 500 or more and 5000 or less. 2. The liquid resin composition for electronic parts as described in 1 above.
3.配線基板上に電子部品を直接バンプ接続する電子部品装置に用いられる前記1.又は2.記載の電子部品用液状樹脂組成物。 3. The liquid resin composition for electronic parts as described in 1 or 2 above, which is used in an electronic part apparatus for directly bump-connecting electronic parts on a wiring board.
4. バンプが鉛を含まない金属である前記3.記載の電子部品用液状樹脂組成物。 4. The liquid resin composition for electronic parts as described in 3 above, wherein the bump is a metal containing no lead.
5. 電子部品の長辺の長さが5mm以上であり、かつ電子部品装置を構成する配線基板と前記電子部品のバンプ接続面の距離が80μm以下である前記3.または4.記載の電子部品用液状樹脂組成物。 5. The electronic component according to 3 or 4 above, wherein the length of the long side of the electronic component is 5 mm or more, and the distance between the wiring board constituting the electronic component device and the bump connection surface of the electronic component is 80 μm or less. Liquid resin composition.
6. 電子部品の長辺の長さが5mm以上であり、かつ前記電子部品が誘電率3.0以下の誘電体層を有する半導体素子である前記3.〜5.いずれかに記載の電子部品用液状樹脂組成物。 6. The electronic component according to any one of 3. to 5. above, wherein the electronic component is a semiconductor element having a dielectric layer having a dielectric constant of 3.0 or less, wherein the length of the long side of the electronic component is 5 mm or more. Liquid resin composition.
7. 前記1.〜6.のいずれかに記載の電子部品用液状樹脂組成物により封止された電子部品を備えた電子部品装置。 7. An electronic component device comprising an electronic component sealed with the liquid resin composition for electronic components according to any one of 1. to 6. above.
8. バンプが鉛を含まない金属である前記7.記載の電子部品装置。 8. The electronic component device as described in 7 above, wherein the bump is a metal containing no lead.
9. 電子部品の長辺の長さが5mm以上であり、かつ電子部品装置を構成する配線基板と前記電子部品のバンプ接続面の距離が80μm以下である前記7.または8.記載の電子部品装置。 9. The electronic component according to 7 or 8 above, wherein the length of the long side of the electronic component is 5 mm or more and the distance between the wiring board constituting the electronic component device and the bump connection surface of the electronic component is 80 μm or less. apparatus.
10. 電子部品の長辺の長さが5mm以上であり、かつ前記電子部品が誘電率3.0以下の誘電体層を有する半導体素子である前記7.〜9.いずれかに記載の電子部品装置。 10. The electronic component as described in any one of 7 to 9 above, wherein the electronic component is a semiconductor element having a long side length of 5 mm or more and the electronic component having a dielectric layer having a dielectric constant of 3.0 or less. apparatus.
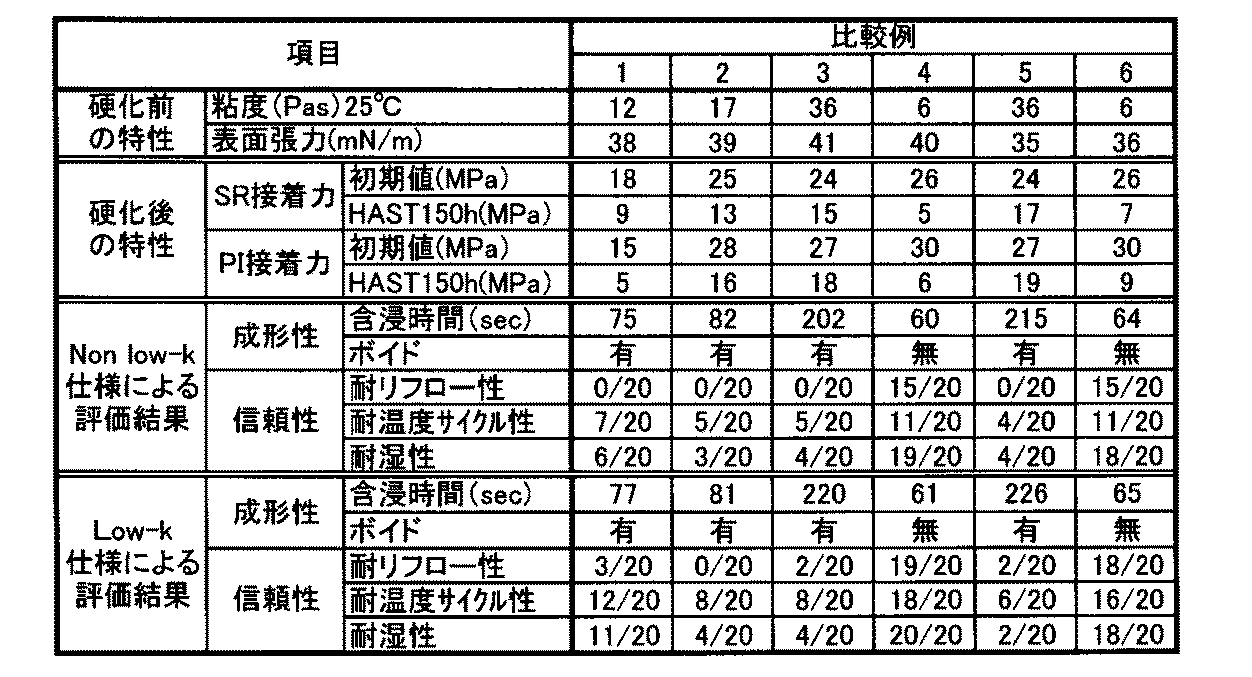
本発明の電子部品用液状樹脂組成物は、表面張力を小さくすることで成形時のボイドを抑制し、吸湿後においても各種基材との接着力が高いため、この電子部品用液状樹脂組成物を用いて素子を封止すれば信頼性に優れる電子部品装置を得ることができるので、その工業的価値は大である。 Since the liquid resin composition for electronic parts of the present invention suppresses voids during molding by reducing the surface tension and has high adhesive strength with various substrates even after moisture absorption, this liquid resin composition for electronic parts Since the electronic component device having excellent reliability can be obtained by sealing the element using, its industrial value is great.
本発明で用いる(A)成分の液状エポキシ樹脂としては、一分子中に1個以上のエポキシ基を有するもので常温で液状であれば制限はなく、電子部品用液状樹脂組成物で一般に使用されている液状エポキシ樹脂を用いることができる。本発明で使用できるエポキシ樹脂としては、例えば、ビスフェノールA、ビスフェノールF、ビスフェノールAD、ビスフェノールS、水添ビスフェノールA等のジグリシジルエーテル型エポキシ樹脂、オルソクレゾールノボラック型エポキシ樹脂を代表とするフェノール類とアルデヒド類のノボラック樹脂をエポキシ化したもの、フタル酸、ダイマー酸等の多塩基酸とエピクロルヒドリンの反応により得られるグリシジルエステル型エポキシ樹脂、p―アミノフェノール、ジアミノジフェニルメタン、イソシアヌル酸等のアミン化合物とエピクロルヒドリンの反応により得られるグリシジルアミン型エポキシ樹脂、オレフィン結合を過酢酸等の過酸により酸化して得られる線状脂肪族エポキシ樹脂、脂環族エポキシ樹脂等が挙げられ、これらを単独で用いても2種以上を組み合わせて用いてもよい。なかでも、流動性の観点からは液状ビスフェノール型エポキシ樹脂が好ましく、耐熱性、接着性及び流動性の観点から液状グリシジルアミン型エポキシ樹脂が好ましい。 The liquid epoxy resin of component (A) used in the present invention is not limited as long as it has one or more epoxy groups in one molecule and is liquid at room temperature, and is generally used in liquid resin compositions for electronic components. A liquid epoxy resin can be used. Examples of epoxy resins that can be used in the present invention include diglycidyl ether type epoxy resins such as bisphenol A, bisphenol F, bisphenol AD, bisphenol S, hydrogenated bisphenol A, and phenols typified by orthocresol novolac type epoxy resins. Epoxidized aldehyde novolak resin, glycidyl ester epoxy resin obtained by reaction of polybasic acid such as phthalic acid and dimer acid and epichlorohydrin, amine compound such as p-aminophenol, diaminodiphenylmethane, isocyanuric acid and epichlorohydrin Glycidylamine-type epoxy resin obtained by the above reaction, linear aliphatic epoxy resin obtained by oxidizing olefinic bonds with peracid such as peracetic acid, alicyclic epoxy resin, etc. May be used in combination of two or more kinds thereof may be used alone. Among these, a liquid bisphenol type epoxy resin is preferable from the viewpoint of fluidity, and a liquid glycidylamine type epoxy resin is preferable from the viewpoint of heat resistance, adhesiveness, and fluidity.
上記した2種のエポキシ樹脂はいずれか1種を単独で用いても2種以上を組合わせて用いてもよいが、その配合量は、その性能を発揮するために液状エポキシ樹脂全量に対して合わせて20質量%以上とすることが好ましく、30質量%以上がより好ましく、50質量%以上とすることがさらに好ましい。 The above two types of epoxy resins may be used alone or in combination of two or more, but the blending amount is based on the total amount of the liquid epoxy resin in order to exhibit its performance. In total, it is preferably 20% by mass or more, more preferably 30% by mass or more, and further preferably 50% by mass or more.
また、本発明の電子部品用液状樹脂組成物には、本発明の効果が達成される範囲内であれば固形エポキシ樹脂を併用することもできるが、成形時の流動性の観点から併用する固形エポキシ樹脂はエポキシ樹脂全量に対して20質量%以下とすることが好ましい。さらに、これらのエポキシ樹脂の純度、特に加水分解性塩素量はICなど素子上のアルミ配線腐食に係わるため少ない方が好ましく、耐湿性の優れた電子部品用液状樹脂組成物を得るためには500ppm以下であることが好ましい。ここで、加水分解性塩素量とは試料のエポキシ樹脂1gをジオキサン30mlに溶解し、1N−KOHメタノール溶液5mlを添加して30分間リフラックス後、電位差滴定により求めた値を尺度としたものである。 Further, in the liquid resin composition for electronic parts of the present invention, a solid epoxy resin can be used in combination as long as the effect of the present invention is achieved, but the solid resin used in combination from the viewpoint of fluidity during molding. The epoxy resin is preferably 20% by mass or less based on the total amount of the epoxy resin. Further, the purity of these epoxy resins, particularly the amount of hydrolyzable chlorine, is preferably less because it relates to the corrosion of aluminum wiring on ICs and other elements. In order to obtain a liquid resin composition for electronic parts having excellent moisture resistance, 500 ppm. The following is preferable. Here, the amount of hydrolyzable chlorine is a value obtained by dissolving 1 g of an epoxy resin of a sample in 30 ml of dioxane, adding 5 ml of a 1N-KOH methanol solution and refluxing for 30 minutes, and then obtaining by potentiometric titration. is there.
本発明に用いられる液状芳香族アミンを含む硬化剤(B)は、常温で液状の芳香環を有するアミンを含むものであれば特に制限はなく、これらを例示すればジエチルトルエンジアミン、1−メチル−3,5−ジエチル−2,4−ジアミノベンゼン、1−メチル−3,5−ジエチル−2,6−ジアミノベンゼン、1,3,5−トリエチル−2,6−ジアミノベンゼン、3,3´−ジエチル−4,4´−ジアミノジフェニルメタン、3,5,3´,5´−テトラメチル−4,4´−ジアミノジフェニルメタンなどが挙げられる。これらの液状芳香族アミン化合物は、例えば、市販品として、jERキュア−W、jERキュア−Z(ジャパンエポキシレジン株式会社製商品名)、カヤハードA−A、カヤハードA−B、カヤハードA−S(日本化薬株式会社製商品名)、トートアミンHM−205(東都化成株式会社製商品名)、アデカハードナーEH−101(旭電化工業株式会社製商品名)、エポミックQ−640、エポミックQ−643(三井化学株式会社製商品名)、DETDA80(Lonza社製商品名)等が入手可能で、これらは単独で用いても2種類以上を組み合わせて用いてもよい。 The curing agent (B) containing a liquid aromatic amine used in the present invention is not particularly limited as long as it contains an amine having a liquid aromatic ring at room temperature, and examples thereof include diethyltoluenediamine and 1-methyl. -3,5-diethyl-2,4-diaminobenzene, 1-methyl-3,5-diethyl-2,6-diaminobenzene, 1,3,5-triethyl-2,6-diaminobenzene, 3,3 ' -Diethyl-4,4'-diaminodiphenylmethane, 3,5,3 ', 5'-tetramethyl-4,4'-diaminodiphenylmethane and the like. These liquid aromatic amine compounds are, for example, commercially available products such as jER Cure-W, jER Cure-Z (trade name, manufactured by Japan Epoxy Resin Co., Ltd.), Kayahard A-A, Kayahard AB, Kayahard AS ( Nippon Kayaku Co., Ltd. product name), Totoamine HM-205 (Toto Kasei Co., Ltd. product name), Adeka Hardener EH-101 (Asahi Denka Kogyo Co., Ltd. product name), Epomic Q-640, Epomic Q-643 (Trade name manufactured by Mitsui Chemicals, Inc.), DETDA80 (trade name manufactured by Lonza), etc. are available, and these may be used alone or in combination of two or more.
硬化剤に含まれる液状芳香族アミンとしては、保存安定性の観点からは、3,3´−ジエチル−4,4´−ジアミノジフェニルメタン及びジエチルトルエンジアミンが好ましく、硬化剤はこれらのいずれか又はこれらの混合物を主成分とすることが好ましい。ジエチルトルエンジアミンとしては、3,5−ジエチルトルエン−2,4−ジアミン及び3,5−ジエチルトルエン−2,6−ジアミンが挙げられ、これらを単独で用いても混合物を用いてもよいが、3,5−ジエチルトルエン−2,4−ジアミンを60質量%以上含むものが好ましい。 As the liquid aromatic amine contained in the curing agent, 3,3′-diethyl-4,4′-diaminodiphenylmethane and diethyltoluenediamine are preferable from the viewpoint of storage stability, and the curing agent is any one of these or these It is preferable to use a mixture of Examples of diethyltoluenediamine include 3,5-diethyltoluene-2,4-diamine and 3,5-diethyltoluene-2,6-diamine, and these may be used alone or as a mixture. What contains 60 mass% or more of 3,5-diethyltoluene-2,4-diamine is preferable.
また、本発明の電子部品用液状樹脂組成物には、本発明の効果が達成される範囲内であれば液状芳香族アミンを含む硬化剤以外に、フェノール性硬化剤、酸無水物等の電子部品用液状樹脂組成物で一般に使用されている硬化剤を併用することができ、固形アミン、イミダゾール化合物等の固形硬化剤も併用することもできる。なかでも、耐熱性の観点からは、固形アミンが好ましく、流動性の観点からは、アリル化フェノールノボラック樹脂が好ましい。 In addition, the liquid resin composition for electronic parts of the present invention includes, in addition to a curing agent containing a liquid aromatic amine, an electron such as a phenolic curing agent or an acid anhydride as long as the effects of the present invention are achieved. Curing agents generally used in liquid resin compositions for parts can be used in combination, and solid curing agents such as solid amines and imidazole compounds can also be used in combination. Among these, solid amines are preferable from the viewpoint of heat resistance, and allylated phenol novolac resins are preferable from the viewpoint of fluidity.
他の硬化剤を併用する場合、液状芳香族アミンを含む硬化剤(B)の配合量は、その性能を発揮するために硬化剤全量に対して60質量%以上とすることが好ましい。(A)成分と固形エポキシ樹脂を含むエポキシ樹脂と(B)硬化剤全量との当量比は特に制限はないが、それぞれの未反応分を少なく抑えるために、エポキシ樹脂に対して硬化剤を0.7当量以上1.6当量以下の範囲に設定することが好ましく、0.8当量以上1.4当量以下がより好ましく、0.9当量以上1.2当量以下がさらに好ましい。 When other curing agents are used in combination, the blending amount of the curing agent (B) containing the liquid aromatic amine is preferably 60% by mass or more based on the total amount of the curing agent in order to exhibit its performance. The equivalent ratio of the component (A) and the epoxy resin containing the solid epoxy resin and the total amount of the (B) curing agent is not particularly limited. It is preferably set in the range of not less than 0.7 equivalents and not more than 1.6 equivalents, more preferably not less than 0.8 equivalents and not more than 1.4 equivalents, and still more preferably not less than 0.9 equivalents and not more than 1.2 equivalents.
本発明に用いられる(C)成分の界面活性剤は、非イオン性の界面活性剤であれば特に制限はなく、例えばポリオキシエチレンアルキルエーテル系、ポリオキシアルキレンアルキルエーテル系、ソルビタン脂肪酸エステル系、ポリオキシエチレンソルビタン脂肪酸エステル系、ポリオキシエチレンソルビトール脂肪酸エステル系、グリセリン脂肪酸エステル系、ポリオキシエチレン脂肪酸エステル系、ポリオキシエチレンアルキルアミン系、アルキルアルカノールアミド系、ポリエーテル変性シリコーン系、アラルキル変性シリコーン系、ポリエステル変性シリコーン系、ポリアクリル系などの界面活性剤が挙げられ、なかでもポリエーテル変性シリコーン系界面活性剤およびアラルキル変性シリコーン系界面活性剤が電子部品用液状樹脂組成物の表面張力低減に効果が有る。これらの非イオン性界面活性剤は、例えば、市販品としてBYK−307、BYK−333、BYK−377、BYK−323(ビックケミー・ジャパン製商品名)等が入手可能である。また、非イオン性以外の界面活性剤を併用しても良い。 The surfactant of component (C) used in the present invention is not particularly limited as long as it is a nonionic surfactant. For example, polyoxyethylene alkyl ether type, polyoxyalkylene alkyl ether type, sorbitan fatty acid ester type, Polyoxyethylene sorbitan fatty acid ester, polyoxyethylene sorbitol fatty acid ester, glycerin fatty acid ester, polyoxyethylene fatty acid ester, polyoxyethylene alkylamine, alkylalkanolamide, polyether-modified silicone, aralkyl-modified silicone , Polyester-modified silicone and polyacrylic surfactants, among others, polyether-modified silicone surfactants and aralkyl-modified silicone surfactants are liquid resin groups for electronic components. Effect is in the surface tension reduction of thing. As these nonionic surfactants, for example, BYK-307, BYK-333, BYK-377, BYK-323 (trade names manufactured by Big Chemie Japan) and the like are available as commercial products. Further, a surfactant other than nonionic may be used in combination.
さらに、非イオン性界面活性剤としてシリコーン変性エポキシ樹脂を添加することができる。シリコーン変性エポキシ樹脂はエポキシ基と反応する官能基を有するオルガノシロキサンとエポキシ樹脂との反応物として得ることができるが、常温で液状であることが好ましい。シリコーン変性エポキシ樹脂は液体の表面に局在化し、液体の表面張力を下げることができる。これにより濡れ性が高く、流動しやすくなるため、狭ギャップへの浸透性向上や巻き込みボイドの低減に効果がある。 Furthermore, a silicone-modified epoxy resin can be added as a nonionic surfactant. The silicone-modified epoxy resin can be obtained as a reaction product of an organosiloxane having a functional group that reacts with an epoxy group and an epoxy resin, but is preferably liquid at room temperature. The silicone-modified epoxy resin is localized on the surface of the liquid and can reduce the surface tension of the liquid. As a result, the wettability is high and the fluid is easy to flow, which is effective in improving the permeability to a narrow gap and reducing entrainment voids.
ここでエポキシ基と反応する官能基を有するオルガノシロキサンを例示すれば、アミノ基、カルボキシル基、水酸基、フェノール性水酸基、メルカプト基等を1分子中に1個以上有するジメチルシロキサン、ジフェニルシロキサン、メチルフェニルシロキサン等が挙げられる。これらのオルガノシロキサンは、例えば、市販品としてBY16−799、BY16−871、BY16−004(東レ・ダウコーニング・シリコーン製商品名)やX−22−1821、KF−8010(信越化学製商品名)等が入手可能である。オルガノシロキサンの分子構造は直鎖状、分枝鎖状のいずれであっても良い。シリコーン変性エポキシ樹脂は、原料エポキシ樹脂100質量部に対してエポキシ基と反応可能な官能基を有するシリコーン成分を50〜150質量部反応させたものが好ましい。 Examples of organosiloxanes having functional groups that react with epoxy groups are dimethylsiloxane, diphenylsiloxane, methylphenyl having at least one amino group, carboxyl group, hydroxyl group, phenolic hydroxyl group, mercapto group, etc. in one molecule. Examples thereof include siloxane. These organosiloxanes are, for example, commercially available products BY16-799, BY16-871, BY16-004 (trade name made by Toray Dow Corning Silicone), X-22-1821, KF-8010 (trade name made by Shin-Etsu Chemical). Etc. are available. The molecular structure of the organosiloxane may be either linear or branched. The silicone-modified epoxy resin is preferably obtained by reacting 50 to 150 parts by mass of a silicone component having a functional group capable of reacting with an epoxy group with respect to 100 parts by mass of the raw material epoxy resin.
オルガノシロキサンの重量平均分子量としては500以上5000以下の範囲が好ましく、100以上3000以下の範囲がより好ましい。この理由としては500未満では樹脂系との相溶性が良くなり過ぎて添加剤としての効果が発揮されず、5000mm2/sを超えると樹脂系との相溶性が悪くなり過ぎてシリコーン変性エポキシ樹脂が成形時に分離・染み出しを発生し、接着性や外観を損なうためである。本発明において、重量平均分子量はゲルパーミエーションクロマトグラフィー法(GPC)により標準ポリスチレンによる検量線を用いて測定する。具体的には、GPCとしてポンプ(株式会社日立製作所製L−6200型)、カラム(TSKgel−G5000HXLおよびTSKgel−G2000HXL、いずれも東ソー株式会社製商品名)、検出器(株式会社日立製作所製L−3300RI型)を用い、テトラヒドロフランを溶離液として温度30℃、流量1.0ml/minの条件で測定した結果を参照する。 The weight average molecular weight of the organosiloxane is preferably in the range of 500 to 5000, and more preferably in the range of 100 to 3000. The reason for this is that if it is less than 500, the compatibility with the resin system becomes too good and the effect as an additive is not exhibited, and if it exceeds 5000 mm 2 / s, the compatibility with the resin system becomes too bad and the silicone-modified epoxy resin becomes poor. This is because, during molding, separation and oozing occur and the adhesiveness and appearance are impaired. In the present invention, the weight average molecular weight is measured by gel permeation chromatography (GPC) using a standard polystyrene calibration curve. Specifically, as GPC, a pump (L-6200 model manufactured by Hitachi, Ltd.), a column (TSKgel-G5000HXL and TSKgel-G2000HXL, both of which are trade names manufactured by Tosoh Corporation), a detector (L-manufactured by Hitachi, Ltd.) 3300RI type), and the result of measurement using tetrahydrofuran as an eluent at a temperature of 30 ° C. and a flow rate of 1.0 ml / min is referred to.
シリコーン変性エポキシ樹脂を得るためのエポキシ樹脂としては電子部品用液状樹脂組成物の樹脂系に相溶するものであれば特に制限は無く、電子部品用液状樹脂組成物に一般に使用されているエポキシ樹脂を用いることができ、たとえば、ビスフェノールA、ビスフェノールF、ビスフェノールAD、ビスフェノールS、ナフタレンジオール、水添ビスフェノールA等とエピクロルヒドリンの反応により得られるグリシジルエーテル型エポキシ樹脂、オルソクレゾールノボラック型エポキシ樹脂をはじめとするフェノール類とアルデヒド類とを縮合又は共縮合させて得られるノボラック樹脂をエポキシ化したノボラック型エポキシ樹脂、フタル酸、ダイマー酸等の多塩基酸とエピクロルヒドリンの反応により得られるグリシジルエステル型エポキシ樹脂、ジアミノジフェニルメタン、イソシアヌル酸等のポリアミンとエピクロルヒドリンの反応により得られるグリシジルアミン型エポキシ樹脂、オレフィン結合を過酢酸等の過酸で酸化して得られる線状脂肪族エポキシ樹脂、脂環族エポキシ樹脂などが挙げられ、これらを単独で用いても2種以上を組み合わせて用いてもよいが、常温液状のものが好ましい。特に好ましいのはビスフェノールA型エポキシ樹脂、ビスフェノールF型エポキシ樹脂、ビスフェノールAD型エポキシ樹脂である。 The epoxy resin for obtaining the silicone-modified epoxy resin is not particularly limited as long as it is compatible with the resin system of the liquid resin composition for electronic parts, and is generally used for the liquid resin composition for electronic parts. For example, glycidyl ether type epoxy resins, orthocresol novolac type epoxy resins obtained by the reaction of bisphenol A, bisphenol F, bisphenol AD, bisphenol S, naphthalene diol, hydrogenated bisphenol A and the like with epichlorohydrin Glycidyl ester type obtained by reaction of a polybasic acid such as phthalic acid and dimer acid with epichlorohydrin obtained by epoxidizing a novolak resin obtained by condensation or cocondensation of phenols and aldehydes Glycidylamine type epoxy resin obtained by reaction of polyamine such as poxy resin, diaminodiphenylmethane, isocyanuric acid and epichlorohydrin, linear aliphatic epoxy resin obtained by oxidizing olefin bond with peracid such as peracetic acid, alicyclic epoxy Resins and the like can be mentioned, and these may be used alone or in combination of two or more, but those at room temperature are preferred. Particularly preferred are bisphenol A type epoxy resin, bisphenol F type epoxy resin, and bisphenol AD type epoxy resin.
本発明では耐熱衝撃性向上、半導体素子への応力低減などの観点から各種可撓剤を配合することができる。可撓剤としては、特に制限は無いがゴム粒子が好ましく、それらを例示すればスチレン−ブタジエンゴム(SBR)、ニトリル−ブタジエンゴム(NBR)、ブタジエンゴム(BR)、ウレタンゴム(UR)、アクリルゴム(AR)等のゴム粒子が挙げられる。なかでも耐熱性、耐湿性の観点からアクリルゴムからなるゴム粒子が好ましく、コアシェル型アクリル系重合体、すなわちコアシェル型アクリルゴム粒子がより好ましい。 In the present invention, various flexible agents can be blended from the viewpoint of improving thermal shock resistance and reducing stress on the semiconductor element. The flexible agent is not particularly limited, but rubber particles are preferable. Examples thereof include styrene-butadiene rubber (SBR), nitrile-butadiene rubber (NBR), butadiene rubber (BR), urethane rubber (UR), and acrylic. Examples thereof include rubber particles such as rubber (AR). Among these, rubber particles made of acrylic rubber are preferable from the viewpoint of heat resistance and moisture resistance, and core-shell type acrylic polymers, that is, core-shell type acrylic rubber particles are more preferable.
また、上記以外のゴム粒子としてシリコーンゴム粒子も好適に用いることができ、それらを例示すれば、直鎖状のポリジメチルシロキサン、ポリメチルフェニルシロキサン、ポリジフェニルシロキサンなどのポリオルガノシロキサンを架橋したシリコーンゴム粒子、シリコーンゴム粒子の表面をシリコーンレジンで被覆したもの、乳化重合等で得られる固形シリコーン粒子のコアとアクリル樹脂などの有機重合体のシェルからなるコア−シェル重合体粒子等が挙げられる。これらのシリコーン重合体粒子の形状は無定形であっても球形であっても使用することができるが、電子部品用液状樹脂組成物の成形性に関わる粘度を低く抑えるためには球形のものを用いることが好ましい。これらのシリコーン重合体粒子は東レ・ダウコーニング・シリコーン株式会社、信越化学株式会社等から市販品が入手可能である。 Silicone rubber particles can also be suitably used as the rubber particles other than those described above. For example, a silicone obtained by crosslinking a polyorganosiloxane such as a linear polydimethylsiloxane, polymethylphenylsiloxane, or polydiphenylsiloxane. Examples thereof include rubber particles, those obtained by coating the surface of silicone rubber particles with a silicone resin, and core-shell polymer particles comprising a core of solid silicone particles obtained by emulsion polymerization and a shell of an organic polymer such as an acrylic resin. These silicone polymer particles can be used in an amorphous or spherical shape, but in order to keep the viscosity related to the moldability of the liquid resin composition for electronic parts low, a spherical one is used. It is preferable to use it. These silicone polymer particles are commercially available from Toray Dow Corning Silicone Co., Ltd. and Shin-Etsu Chemical Co., Ltd.
これらのゴム粒子の1次粒径は組成物を均一に変性するためには細かい方が良好であり、1次粒子径が0.05μm以上10μm以下の範囲であることが好ましく、0.1μm以上5μm以下の範囲であることが更に好ましい。1次粒径が0.05μm未満では液状エポキシ樹脂組成物への分散性に劣る傾向があり、10μmを超えると低応力化改善効果が低くなる傾向や、電子部品用液状樹脂組成物としての微細間隙への浸透性・流動性が低下しボイド、未充填を招きやすくなる傾向がある。 The primary particle diameter of these rubber particles is preferably finer in order to uniformly modify the composition, and the primary particle diameter is preferably in the range of 0.05 μm to 10 μm, preferably 0.1 μm or more. More preferably, it is in the range of 5 μm or less. If the primary particle size is less than 0.05 μm, the dispersibility in the liquid epoxy resin composition tends to be inferior, and if it exceeds 10 μm, the effect of reducing stress reduction tends to be low, and the fineness as a liquid resin composition for electronic components There is a tendency for the permeability and fluidity to the gap to be lowered, and voids and unfilling to occur easily.
これらのゴム粒子の配合量は、充填剤を除く電子部品用液状樹脂組成物全体の1質量%以上30質量%以下の範囲に設定されるのが好ましく、より好ましくは2質量%以上20質量%以下である。ゴム粒子の配合量が1質量%未満では低応力効果が低くなる傾向があり、30質量%を超えると電子部品用液状樹脂組成物の粘度が上昇し成形性(流動特性)に劣る傾向がある。 The blending amount of these rubber particles is preferably set in a range of 1% by mass or more and 30% by mass or less, and more preferably 2% by mass or more and 20% by mass of the entire liquid resin composition for electronic parts excluding the filler. It is as follows. When the blending amount of the rubber particles is less than 1% by mass, the low stress effect tends to be low, and when it exceeds 30% by mass, the viscosity of the liquid resin composition for electronic parts tends to increase and the moldability (flow characteristics) tends to be inferior. .
上記(A)〜(C)成分と共に用いられる(D)成分の無機充填剤としては、例えば、溶融シリカ、結晶シリカ等のシリカ、炭酸カルシウム、クレー、酸化アルミナ等のアルミナ、窒化珪素、炭化珪素、窒化ホウ素、珪酸カルシウム、チタン酸カリウム、窒化アルミ、ベリリア、ジルコニア、ジルコン、フォステライト、ステアタイト、スピネル、ムライト、チタニア等の粉体、又はこれらを球形化したビーズ、ガラス繊維などが挙げられる。 Examples of the inorganic filler of the component (D) used together with the components (A) to (C) include silica such as fused silica and crystalline silica, alumina such as calcium carbonate, clay and alumina oxide, silicon nitride and silicon carbide. , Boron nitride, calcium silicate, potassium titanate, aluminum nitride, beryllia, zirconia, zircon, fosterite, steatite, spinel, mullite, titania, etc., or spherical beads of these, glass fibers, etc. .
さらに、難燃効果のある無機充填剤としては水酸化アルミニウム、水酸化マグネシウム、硼酸亜鉛、モリブデン酸亜鉛等が挙げられる。これらの無機充填剤は単独で用いても2種類以上を組み合わせて用いてもよい。なかでも溶融シリカが好ましく、電子部品用液状樹脂組成物の微細間隙への流動性・浸透性の観点からは球形シリカがより好ましい。 Furthermore, examples of the inorganic filler having a flame retardant effect include aluminum hydroxide, magnesium hydroxide, zinc borate, and zinc molybdate. These inorganic fillers may be used alone or in combination of two or more. Of these, fused silica is preferred, and spherical silica is more preferred from the viewpoint of fluidity and permeability into the fine gaps of the liquid resin composition for electronic components.
無機充填剤の平均粒径は、特に球形シリカの場合、0.3μm以上10μm以下の範囲が好ましく、平均粒径0.5μm以上5μm以下の範囲がより好ましい。平均粒径が0.5μm未満では液状樹脂への分散性に劣る傾向や電子部品用液状樹脂組成物にチキソトロピック性が付与されて流動特性に劣る傾向があり、10μmを超えるとフィラ沈降を起こしやすくなる傾向や、電子部品用液状樹脂組成物としての微細間隙への浸透性・流動性が低下してボイド・未充填を招きやすくなる傾向がある。無機充填剤の配合量は、電子部品用液状樹脂組成物全体の20質量%以上90質量%以下の範囲に設定されるのが好ましく、より好ましくは25質量%以上80質量%以下、さらに好ましくは30質量%以上60質量%以下である。配合量が20質量%未満では熱膨張係数の低減効果が低くなる傾向があり、90質量%を超えると電子部品用液状樹脂組成物の粘度が上昇し、流動性・浸透性およびディスペンス性の低下を招く傾向がある。 The average particle size of the inorganic filler is preferably in the range of 0.3 μm to 10 μm, and more preferably in the range of 0.5 μm to 5 μm, particularly in the case of spherical silica. If the average particle size is less than 0.5 μm, the dispersibility in the liquid resin tends to be inferior, or the liquid resin composition for electronic parts tends to have thixotropic properties and the flow properties tend to be inferior. If it exceeds 10 μm, filler sedimentation occurs. There is a tendency that it becomes easy, and the permeability / fluidity to the fine gap as the liquid resin composition for electronic parts tends to be lowered, and voids / unfilled tend to be caused. The blending amount of the inorganic filler is preferably set in the range of 20% by mass or more and 90% by mass or less of the entire liquid resin composition for electronic parts, more preferably 25% by mass or more and 80% by mass or less, and still more preferably. 30% by mass or more and 60% by mass or less. When the blending amount is less than 20% by mass, the effect of reducing the thermal expansion coefficient tends to be low. When the blending amount exceeds 90% by mass, the viscosity of the liquid resin composition for electronic parts increases, and the fluidity / penetration and dispensing properties decrease. Tend to invite.
本発明の電子部品用液状樹脂組成物には必要に応じて、樹脂と無機充填剤或いは樹脂と電子部品の構成部材との界面接着を強固にする目的でカップリング剤を使用することができる。これらのカップリング剤には特に制限はなく、従来公知のものを用いることができるが、たとえば、1級及び/又は2級及び/又は3級アミノ基を有するシラン化合物、エポキシシラン、メルカプトシラン、アルキルシラン、ウレイドシラン、ビニルシラン等の各種シラン系化合物、チタン系化合物、アルミニウムキレート類、アルミニウム/ジルコニウム系化合物等が挙げられる。これらを例示すると、ビニルトリクロロシラン、ビニルトリエトキシシラン、ビニルトリス(β−メトキシエトキシ)シラン、γ−メタクリロキシプロピルトリメトキシシラン、β−(3,4−エポキシシクロヘキシル)エチルトリメトキシシラン、γ−グリシドキシプロピルトリメトキシシラン、γ−グリシドキシプロピルメチルジメトキシシラン、ビニルトリアセトキシシラン、γ−メルカプトプロピルトリメトキシシラン、γ−アミノプロピルトリメトキシシラン、γ−アミノプロピルメチルジメトキシシラン、γ−アミノプロピルトリエトキシシラン、γ−アミノプロピルメチルジエトキシシラン、γ−アニリノプロピルトリメトキシシラン、γ−アニリノプロピルトリエトキシシラン、γ−(N,N−ジメチル)アミノプロピルトリメトキシシラン、γ−(N,N−ジエチル)アミノプロピルトリメトキシシラン、γ−(N,N−ジブチル)アミノプロピルトリメトキシシラン、γ−(N−メチル)アニリノプロピルトリメトキシシラン、γ−(N−エチル)アニリノプロピルトリメトキシシラン、γ−(N,N−ジメチル)アミノプロピルトリエトキシシラン、γ−(N,N−ジエチル)アミノプロピルトリエトキシシラン、γ−(N,N−ジブチル)アミノプロピルトリエトキシシラン、γ−(N−メチル)アニリノプロピルトリエトキシシラン、γ−(N−エチル)アニリノプロピルトリエトキシシラン、γ−(N,N−ジメチル)アミノプロピルメチルジメトキシシラン、γ−(N,N−ジエチル)アミノプロピルメチルジメトキシシラン、γ−(N,N−ジブチル)アミノプロピルメチルジメトキシシラン、γ−(N−メチル)アニリノプロピルメチルジメトキシシラン、γ−(N−エチル)アニリノプロピルメチルジメトキシシラン、N−(トリメトキシシリルプロピル)エチレンジアミン、N−(ジメトキシメチルシリルイソプロピル)エチレンジアミン、メチルトリメトキシシラン、ジメチルジメトキシシラン、メチルトリエトキシシラン、γ−クロロプロピルトリメトキシシラン、ヘキサメチルジシラン、ビニルトリメトキシシラン、γ−メルカプトプロピルメチルジメトキシシラン等のシラン系カップリング剤、イソプロピルトリイソステアロイルチタネート、イソプロピルトリス(ジオクチルパイロホスフェート)チタネート、イソプロピルトリ(N−アミノエチル−アミノエチル)チタネート、テトラオクチルビス(ジトリデシルホスファイト)チタネート、テトラ(2,2−ジアリルオキシメチル−1−ブチル)ビス(ジトリデシル)ホスファイトチタネート、ビス(ジオクチルパイロホスフェート)オキシアセテートチタネート、ビス(ジオクチルパイロホスフェート)エチレンチタネート、イソプロピルトリオクタノイルチタネート、イソプロピルジメタクリルイソステアロイルチタネート、イソプロピルトリドデシルベンゼンスルホニルチタネート、イソプロピルイソステアロイルジアクリルチタネート、イソプロピルトリ(ジオクチルホスフェート)チタネート、イソプロピルトリクミルフェニルチタネート、テトライソプロピルビス(ジオクチルホスファイト)チタネート等のチタネート系カップリング剤などが挙げられ、これらの1種を単独で用いても2種類以上を組み合わせて用いてもよい。カップリング剤の配合量は、液状樹脂組成物に対して0.1〜3.0質量%であることが好ましく、0.2〜2.5質量%であることがより好ましく、0.2〜2.5質量%であることがさらに好ましい。0.1質量%未満では基板と液状樹脂組成物の硬化物の密着性が低下する傾向があり、3.0質量%を超えるとボイドが発生しやすくなる傾向がある。 In the liquid resin composition for electronic parts of the present invention, a coupling agent can be used for the purpose of strengthening the interfacial adhesion between the resin and the inorganic filler or between the resin and the component of the electronic part, if necessary. These coupling agents are not particularly limited and conventionally known ones can be used. For example, silane compounds having primary and / or secondary and / or tertiary amino groups, epoxy silane, mercaptosilane, Examples thereof include various silane compounds such as alkyl silane, ureido silane, and vinyl silane, titanium compounds, aluminum chelates, and aluminum / zirconium compounds. Examples of these are vinyltrichlorosilane, vinyltriethoxysilane, vinyltris (β-methoxyethoxy) silane, γ-methacryloxypropyltrimethoxysilane, β- (3,4-epoxycyclohexyl) ethyltrimethoxysilane, γ-glycol. Sidoxypropyltrimethoxysilane, γ-glycidoxypropylmethyldimethoxysilane, vinyltriacetoxysilane, γ-mercaptopropyltrimethoxysilane, γ-aminopropyltrimethoxysilane, γ-aminopropylmethyldimethoxysilane, γ-aminopropyl Triethoxysilane, γ-aminopropylmethyldiethoxysilane, γ-anilinopropyltrimethoxysilane, γ-anilinopropyltriethoxysilane, γ- (N, N-dimethyl) aminopropyltrimethoxy Silane, γ- (N, N-diethyl) aminopropyltrimethoxysilane, γ- (N, N-dibutyl) aminopropyltrimethoxysilane, γ- (N-methyl) anilinopropyltrimethoxysilane, γ- (N -Ethyl) anilinopropyltrimethoxysilane, γ- (N, N-dimethyl) aminopropyltriethoxysilane, γ- (N, N-diethyl) aminopropyltriethoxysilane, γ- (N, N-dibutyl) amino Propyltriethoxysilane, γ- (N-methyl) anilinopropyltriethoxysilane, γ- (N-ethyl) anilinopropyltriethoxysilane, γ- (N, N-dimethyl) aminopropylmethyldimethoxysilane, γ- (N, N-diethyl) aminopropylmethyldimethoxysilane, γ- (N, N-dibutyl) aminopropy Rumethyldimethoxysilane, γ- (N-methyl) anilinopropylmethyldimethoxysilane, γ- (N-ethyl) anilinopropylmethyldimethoxysilane, N- (trimethoxysilylpropyl) ethylenediamine, N- (dimethoxymethylsilylisopropyl) ) Silane coupling agents such as ethylenediamine, methyltrimethoxysilane, dimethyldimethoxysilane, methyltriethoxysilane, γ-chloropropyltrimethoxysilane, hexamethyldisilane, vinyltrimethoxysilane, γ-mercaptopropylmethyldimethoxysilane, isopropyl Triisostearoyl titanate, isopropyl tris (dioctyl pyrophosphate) titanate, isopropyl tri (N-aminoethyl-aminoethyl) titanate, tetraoctane Rubis (ditridecyl phosphite) titanate, tetra (2,2-diallyloxymethyl-1-butyl) bis (ditridecyl) phosphite titanate, bis (dioctyl pyrophosphate) oxyacetate titanate, bis (dioctyl pyrophosphate) ethylene titanate, Isopropyltrioctanoyl titanate, isopropyldimethacrylisostearoyl titanate, isopropyltridodecylbenzenesulfonyl titanate, isopropylisostearoyl diacryl titanate, isopropyltri (dioctylphosphate) titanate, isopropyltricumylphenyl titanate, tetraisopropylbis (dioctylphosphite) titanate And titanate coupling agents such as A seed may be used independently or may be used in combination of 2 or more types. The blending amount of the coupling agent is preferably 0.1 to 3.0% by mass, more preferably 0.2 to 2.5% by mass, and more preferably 0.2 to 2.5% by mass with respect to the liquid resin composition. More preferably, it is 2.5 mass%. If the amount is less than 0.1% by mass, the adhesion between the substrate and the cured product of the liquid resin composition tends to decrease, and if it exceeds 3.0% by mass, voids tend to be generated.
本発明の電子部品用液状樹脂組成物には、必要に応じて液状エポキシ樹脂(A)を含むエポキシ樹脂と液状芳香族アミンを含む硬化剤(B)の反応を促進する硬化促進剤を用いることができる。硬化促進剤には特に制限はなく、従来公知のものを用いることができる。これらを例示すれば1,8−ジアザ−ビシクロ(5,4,0)ウンデセン−7、1,5−ジアザ−ビシクロ(4,3,0)ノネン、5、6−ジブチルアミノ−1,8−ジアザ−ビシクロ(5,4,0)ウンデセン−7等のシクロアミジン化合物及びこれらの誘導体、
トリエチレンジアミン、ベンジルジメチルアミン、トリエタノールアミン、ジメチルアミノエタノール、トリス(ジメチルアミノメチル)フェノール等の三級アミン化合物及びこれらの誘導体、
2−メチルイミダゾール、2−エチル−4−メチルイミダゾール、2−フェニルイミダゾール、2−フェニル−4−メチルイミダゾール、1−ベンジルー2−フェニルイミダゾール、1−ベンジルー2−メチルイミダゾール、2−フェニル−4,5−ジヒドロキシメチルイミダゾール、2−フェニル−4−メチル−5−ヒドロキシメチルイミダゾール、2,4−ジアミノ−6−(2´−メチルイミダゾリル−(1´))−エチル−s−トリアジン、2−ヘプタデシルイミダゾール等のイミダゾール化合物及びこれらの誘導体、
トリブチルホスフィン等のトリアルキルホスフィン、ジメチルフェニルホスフィン等のジアルキルアリールホスフィン、メチルジフェニルホスフィン等のアルキルジアリールホスフィン、トリフェニルホスフィン、アルキル基置換トリフェニルホスフィンなどの有機ホスフィン類及びこれらの誘導体、
及びこれらの化合物に無水マレイン酸、1,4−ベンゾキノン、2,5−トルキノン、1,4−ナフトキノン、2,3−ジメチルベンゾキノン、2,6−ジメチルベンゾキノン、2,3−ジメトキシ−5−メチル−1,4−ベンゾキノン、2,3−ジメトキシ−1,4−ベンゾキノン、フェニル−1,4−ベンゾキノン等のキノン化合物、ジアゾフェニルメタン、フェノール樹脂等のπ結合をもつ化合物を付加してなる分子内分極を有する化合物、及びこれらの誘導体
などが挙げられ、さらには2−エチル−4−メチルイミダゾールテトラフェニルボレート、N−メチルモルホリンテトラフェニルボレート等のフェニルボロン塩等などが挙げられ、これらの1種を単独で用いても2種以上を組合わせて用いてもよい。
In the liquid resin composition for electronic parts of the present invention, a curing accelerator that accelerates the reaction between the epoxy resin containing the liquid epoxy resin (A) and the curing agent (B) containing the liquid aromatic amine is used as necessary. Can do. There is no restriction | limiting in particular in a hardening accelerator, A conventionally well-known thing can be used. For example, 1,8-diaza-bicyclo (5,4,0) undecene-7, 1,5-diaza-bicyclo (4,3,0) nonene, 5,6-dibutylamino-1,8- Cycloamidine compounds such as diaza-bicyclo (5,4,0) undecene-7 and derivatives thereof,
Tertiary amine compounds such as triethylenediamine, benzyldimethylamine, triethanolamine, dimethylaminoethanol, tris (dimethylaminomethyl) phenol, and derivatives thereof;
2-methylimidazole, 2-ethyl-4-methylimidazole, 2-phenylimidazole, 2-phenyl-4-methylimidazole, 1-benzyl-2-phenylimidazole, 1-benzyl-2-methylimidazole, 2-phenyl-4, 5-dihydroxymethylimidazole, 2-phenyl-4-methyl-5-hydroxymethylimidazole, 2,4-diamino-6- (2′-methylimidazolyl- (1 ′))-ethyl-s-triazine, 2-hepta Imidazole compounds such as decylimidazole and derivatives thereof,
Trialkylphosphine such as tributylphosphine, dialkylarylphosphine such as dimethylphenylphosphine, alkyldiarylphosphine such as methyldiphenylphosphine, organic phosphines such as triphenylphosphine and alkyl group-substituted triphenylphosphine, and derivatives thereof,
And maleic anhydride, 1,4-benzoquinone, 2,5-toluquinone, 1,4-naphthoquinone, 2,3-dimethylbenzoquinone, 2,6-dimethylbenzoquinone, 2,3-dimethoxy-5-methyl Molecule formed by adding a quinone compound such as -1,4-benzoquinone, 2,3-dimethoxy-1,4-benzoquinone, phenyl-1,4-benzoquinone, a compound having a π bond such as diazophenylmethane, phenol resin Examples thereof include compounds having internal polarization, and derivatives thereof, and further include phenylboron salts such as 2-ethyl-4-methylimidazole tetraphenylborate and N-methylmorpholine tetraphenylborate. The seeds may be used alone or in combination of two or more.
また、潜在性を有する硬化促進剤として、常温固体のアミノ基を有する化合物、特にイミダゾール誘導体をコアとして、常温固体のエポキシ化合物のシェルを被覆してなるコア−シェル粒子が挙げられ、市販品としてアミキュア(味の素製商品名)や、マイクロカプセル化されたイミダゾール誘導体をビスフェノールA型エポキシ樹脂、ビスフェノールF型エポキシ樹脂に分散させたノバキュア(旭化成ケミカルズ製商品名)などが使用できる。潜在性を有する硬化促進剤とは、ある特定の温度等の条件で硬化促進機能が発現されるもので、例えば通常の硬化促進剤が、マイクロカプセル等で保護されたり各種化合物と付加した塩の構造となっていたりするものが挙げられる。この場合、特定の温度を超えるとマイクロカプセルや付加物から硬化促進剤が開放される。 Further, as a curing accelerator having a potential, a core-shell particle formed by coating a shell of a normal temperature solid epoxy compound with a core having a normal temperature solid amino group, particularly an imidazole derivative as a core, is a commercially available product. Amicure (trade name, manufactured by Ajinomoto Co., Inc.), Novacure (trade name, manufactured by Asahi Kasei Chemicals Co., Ltd.) in which a microencapsulated imidazole derivative is dispersed in a bisphenol A type epoxy resin or a bisphenol F type epoxy resin can be used. A latent curing accelerator is a substance that exhibits a curing acceleration function under conditions such as a specific temperature. For example, a normal curing accelerator is protected by a microcapsule or the like or added with various compounds. There are things that are structured. In this case, when a specific temperature is exceeded, the curing accelerator is released from the microcapsules and adducts.
硬化促進剤は、イミダゾール誘導体が好ましく、硬化性と成形性の両立の観点からは、2−フェニル−4,5−ジヒドロキシメチルイミダゾール、2−フェニル−4−メチル−5−ヒドロキシメチルイミダゾール、2,4−ジアミノ−6−(2´−メチルイミダゾリル−(1´))−エチル−s−トリアジン、2−ヘプタデシルイミダゾールが好ましく、保存安定性の観点からは上記ノバキュア(旭化成ケミカルズ製商品名)が好ましく、特にフェニル基及び水酸基を置換基として有するイミダゾール誘導体である2−フェニル−4,5−ジヒドロキシメチルイミダゾール、2−フェニル−4−メチル−5−ヒドロキシメチルイミダゾールが好ましい。 The curing accelerator is preferably an imidazole derivative. From the viewpoint of achieving both curability and moldability, 2-phenyl-4,5-dihydroxymethylimidazole, 2-phenyl-4-methyl-5-hydroxymethylimidazole, 2, 4-Diamino-6- (2′-methylimidazolyl- (1 ′))-ethyl-s-triazine and 2-heptadecylimidazole are preferred, and from the viewpoint of storage stability, the above Novacure (trade name, manufactured by Asahi Kasei Chemicals) is Particularly preferred are 2-phenyl-4,5-dihydroxymethylimidazole and 2-phenyl-4-methyl-5-hydroxymethylimidazole, which are imidazole derivatives having a phenyl group and a hydroxyl group as a substituent.
硬化促進剤の配合量は、硬化促進効果が達成される量であれば特に制限されるものではないが、(A)液状エポキシ樹脂と固形エポキシ樹脂を含むエポキシ樹脂に対して0.2質量%以上5質量%以下が好ましく、0.5質量%以上3質量%以下がより好ましい。0.2質量%未満では低温での硬化性に劣る傾向があり、5質量%を超えると硬化速度が速すぎて制御が困難になったりポットライフ、シェルライフ等の保存安定性が劣ったりする傾向がある。また、0.2質量%よりも少ないと硬化促進剤の添加効果が十分に発揮されず、結果として成形時のボイドの発生を抑制できなくなるとともに、反り低減効果も不十分になる傾向がある。 Although the compounding quantity of a hardening accelerator will not be restrict | limited especially if the hardening acceleration effect is achieved, it is 0.2 mass% with respect to the epoxy resin containing (A) liquid epoxy resin and solid epoxy resin. The content is preferably 5% by mass or less and more preferably 0.5% by mass or more and 3% by mass or less. If it is less than 0.2% by mass, the curability at low temperature tends to be inferior, and if it exceeds 5% by mass, the curing rate is too fast and it becomes difficult to control, and the storage stability such as pot life and shell life is inferior. Tend. On the other hand, when the amount is less than 0.2% by mass, the effect of adding the curing accelerator is not sufficiently exhibited, and as a result, generation of voids during molding cannot be suppressed and the effect of reducing warpage tends to be insufficient.
なお、硬化促進剤が、硬化促進成分単体でなく、潜在性を有する硬化促進剤のように硬化促進剤成分とエポキシ化合物、エポキシ樹脂成分を有する硬化促進剤を配合する際は、分散しているエポキシ樹脂等を含め、全てを硬化促進剤と考え、その他のエポキシ樹脂に対する配合量は、0.1質量%以上40質量%以下が好ましく、0.5質量%以上20質量%以下がより好ましく、0.8質量%以上10質量%以下がさらに好ましい。0.1質量%未満では低温での硬化性に劣る傾向があり、40質量%を超えると硬化速度が速すぎて制御が困難になったりポットライフ、シェルライフ等の保存安定性が劣ったりする傾向がある。 It should be noted that the curing accelerator is not a curing accelerating component alone but is dispersed when blending a curing accelerator component, an epoxy compound, and a curing accelerator having an epoxy resin component like a latent curing accelerator. Considering everything as a curing accelerator, including epoxy resins, the blending amount with respect to other epoxy resins is preferably 0.1% by mass or more and 40% by mass or less, more preferably 0.5% by mass or more and 20% by mass or less, 0.8 mass% or more and 10 mass% or less are more preferable. If it is less than 0.1% by mass, the curability at low temperature tends to be inferior, and if it exceeds 40% by mass, the curing rate is too high and control becomes difficult, and the storage stability such as pot life and shell life is inferior. Tend.
また、本発明の電子部品用液状樹脂組成物には、必要に応じてイオントラップ剤をIC等の半導体素子の耐マイグレーション性、耐湿性及び高温放置特性を向上させる観点から含有することができる。イオントラップ剤としては特に制限はなく、従来公知のものを用いることができ、特に下記組成式(I)で表されるハイドロタルサイトまたは(II)で表されるビスマスの含水酸化物が好ましい。 Further, the liquid resin composition for electronic parts of the present invention can contain an ion trap agent as necessary from the viewpoint of improving the migration resistance, moisture resistance and high temperature storage characteristics of a semiconductor element such as an IC. There is no restriction | limiting in particular as an ion trap agent, A conventionally well-known thing can be used, Especially the hydrotalcite represented by the following compositional formula (I) or the hydrous oxide of bismuth represented by (II) is preferable.
(化1)
Mg1−XAlX(OH)2(CO3)X/2・mH2O (I)
(式(I)中、0<X≦0.5、mは正の数)
(化2)
BiOx(OH)y(NO3)z (II)
(式(II)中、0.9≦x≦1.1、 0.6≦y≦0.8、
0.2≦z≦0.4)
これらイオントラップ剤の添加量としてはハロゲンイオンなどの陰イオンを捕捉できる十分量であれば特に制限はないが、耐マイグレーション性の観点から液状樹脂組成物に対して0.1質量%以上3.0質量%以下が好ましく、さらに好ましくは0.3質量%以上1.5質量%以下である。イオントラップ剤の平均粒径は0.1μm以上3.0μm以下が好ましく、最大粒径は10μm以下が好ましい。なお、上記式(I)の化合物は市販品として協和化学工業株式会社製商品名DHT−4Aとして入手可能である。また、上記式(II)の化合物は市販品としてIXE500(東亞合成株式会社製商品名)として入手可能である。また必要に応じてその他の陰イオン交換体を添加することもできるたとえば、マグネシウム、アルミニウム、チタン、ジルコニウム、アンチモン等から選ばれる元素の含水酸化物等が挙げられ、これらを単独又は2種以上を組み合わせて用いることができる。
(Chemical formula 1)
Mg 1-X Al X (OH) 2 (CO 3 ) X / 2 · mH 2 O (I)
(In formula (I), 0 <X ≦ 0.5, m is a positive number)
(Chemical formula 2)
BiO x (OH) y (NO 3 ) z (II)
(In formula (II), 0.9 ≦ x ≦ 1.1, 0.6 ≦ y ≦ 0.8,
0.2 ≦ z ≦ 0.4)
The addition amount of these ion trapping agents is not particularly limited as long as it is a sufficient amount capable of capturing anions such as halogen ions, but from the viewpoint of migration resistance, it is 0.1% by mass or more based on the liquid resin composition. 0 mass% or less is preferable, More preferably, it is 0.3 mass% or more and 1.5 mass% or less. The average particle size of the ion trapping agent is preferably 0.1 μm or more and 3.0 μm or less, and the maximum particle size is preferably 10 μm or less. In addition, the compound of the said formula (I) is available as Kyowa Chemical Industry Co., Ltd. brand name DHT-4A as a commercial item. The compound of the above formula (II) is commercially available as IXE500 (trade name, manufactured by Toagosei Co., Ltd.). In addition, other anion exchangers can be added as necessary, for example, hydrated oxides of elements selected from magnesium, aluminum, titanium, zirconium, antimony, etc., and these can be used alone or in combination of two or more. They can be used in combination.
本発明の電子部品用液状樹脂組成物には、その他の添加剤として、染料、カーボンブラック、酸化チタン、鉛丹等の着色剤、難燃剤、希釈剤、レベリング剤、他の可撓化剤、消泡剤などを必要に応じて配合することができる。 In the liquid resin composition for electronic parts of the present invention, as other additives, coloring agents such as dyes, carbon black, titanium oxide, and lead tan, flame retardants, diluents, leveling agents, other flexing agents, An antifoaming agent etc. can be mix | blended as needed.
難燃剤としては、臭素化エポキシ樹脂や三酸化アンチモンを用いることができるが、ノンハロゲン、ノンアンチモンの難燃剤を用いるのが好ましい。たとえば、赤リン、フェノール樹脂等の熱硬化性樹脂等で被覆された赤リン、リン酸エステル、酸化トリフェニルホスフィン等のリン化合物、メラミン、メラミン誘導体、メラミン変性フェノール樹脂、トリアジン環を有する化合物、シアヌル酸誘導体、イソシアヌル酸誘導体等の窒素含有化合物、シクロホスファゼン等のリン及び窒素含有化合物、ジシクロペンタジエニル鉄等の金属錯体化合物、酸化亜鉛、錫酸亜鉛、硼酸亜鉛、モリブデン酸亜鉛等の亜鉛化合物、酸化鉄、酸化モリブデン等の金属酸化物、水酸化アルミニウム、水酸化マグネシウム等の金属水酸化物、下記組成式(III)で示される複合金属水酸化物などが挙げられる。 A brominated epoxy resin or antimony trioxide can be used as the flame retardant, but it is preferable to use a halogen-free or non-antimony flame retardant. For example, red phosphorus coated with a thermosetting resin such as red phosphorus, phenol resin, phosphorous ester, phosphorus compound such as triphenylphosphine oxide, melamine, melamine derivative, melamine modified phenolic resin, compound having triazine ring, Nitrogen-containing compounds such as cyanuric acid derivatives and isocyanuric acid derivatives, phosphorus and nitrogen-containing compounds such as cyclophosphazene, metal complex compounds such as dicyclopentadienyl iron, zinc oxide, zinc stannate, zinc borate, and zinc molybdate Examples thereof include zinc compounds, metal oxides such as iron oxide and molybdenum oxide, metal hydroxides such as aluminum hydroxide and magnesium hydroxide, and composite metal hydroxides represented by the following composition formula (III).
(化3)
p(M1 aOb)・q(M2 cOd)・r(M3 cOd)・mH2O
(III)
(組成式(III)で、M1、M2及びM3は互いに異なる金属元素を示し、a、b、c、d、p、q及びmは正の数、rは0又は正の数を示す。)
上記組成式(III)中のM1、M2及びM3は互いに異なる金属元素であれば特に制限はないが、難燃性の観点からは、M1が第3周期の金属元素、IIA族のアルカリ土類金属元素、IVB族、IIB族、VIII族、IB族、IIIA族及びIVA族に属する金属元素から選ばれ、M2がIIIB〜IIB族の遷移金属元素から選ばれることが好ましく、M1がマグネシウム、カルシウム、アルミニウム、スズ、チタン、鉄、コバルト、ニッケル、銅及び亜鉛から選ばれ、M2が鉄、コバルト、ニッケル、銅及び亜鉛から選ばれることがより好ましい。流動性の観点からは、M1がマグネシウム、M2が亜鉛又はニッケルで、r=0のものが好ましい。p、q及びrのモル比は特に制限はないが、r=0で、p/qが1/99〜1/1であることが好ましい。なお、金属元素の分類は、典型元素をA亜族、遷移元素をB亜族とする長周期型の周期律表に基づいて行った。上記した難燃剤は1種を単独で用いても2種以上を組み合わせて用いてもよい。
(Chemical formula 3)
p (M 1 a O b ) · q (M 2 c O d ) · r (M 3 c O d ) · mH 2 O
(III)
(In the composition formula (III), M 1 , M 2 and M 3 represent different metal elements, a, b, c, d, p, q and m are positive numbers, and r is 0 or a positive number. Show.)
M 1 , M 2 and M 3 in the composition formula (III) are not particularly limited as long as they are different metal elements, but from the viewpoint of flame retardancy, M 1 is a metal element of the third period, group IIA alkaline earth metal elements, IVB group, IIB group, VIII, IB, selected from metal elements belonging to group IIIA and group IVA, it is preferable that M 2 is selected from transition metal elements of group IIIB~IIB, More preferably, M 1 is selected from magnesium, calcium, aluminum, tin, titanium, iron, cobalt, nickel, copper and zinc, and M 2 is selected from iron, cobalt, nickel, copper and zinc. From the viewpoint of fluidity, it is preferable that M 1 is magnesium, M 2 is zinc or nickel, and r = 0. The molar ratio of p, q and r is not particularly limited, but preferably r = 0 and p / q is 1/99 to 1/1. The classification of the metal elements was performed based on a long-period type periodic table in which the typical element is the A subgroup and the transition element is the B subgroup. The above flame retardants may be used alone or in combination of two or more.
希釈剤として、粘度調整のためエポキシ基を有する反応性希釈剤を混合しても良い。エポキシ基を有する反応性希釈剤としては例えばn-ブチルグリシジルエーテル、バーサティック酸グリシジルエーテル、スチレンオキサイド、エチルヘキシルグリシジルエーテル、フェニルグリシジルエーテル、ブチルフェニルグリシジルエーテル、1,6-ヘキサンジオールジグリシジルエーテル、ネオペンチルグリコールジグリシジルエーテル、ジエチレングリコールジグリシジルエーテル、トリメチロールプロパントリグリシジルエーテルが挙げられる。これらの1種を単独で用いても2種以上を組合わせて用いてもよい。 As a diluent, you may mix the reactive diluent which has an epoxy group for viscosity adjustment. Examples of the reactive diluent having an epoxy group include n-butyl glycidyl ether, versatic acid glycidyl ether, styrene oxide, ethylhexyl glycidyl ether, phenyl glycidyl ether, butyl phenyl glycidyl ether, 1,6-hexanediol diglycidyl ether, neodymium Examples include pentyl glycol diglycidyl ether, diethylene glycol diglycidyl ether, and trimethylolpropane triglycidyl ether. One of these may be used alone, or two or more may be used in combination.
本発明の電子部品用液状樹脂組成物は、上記各種成分を均一に分散混合できるのであれば、いかなる手法を用いても調製できるが、一般的な手法として、所定の配合量の成分を秤量し、らいかい機、ミキシングロール、プラネタリミキサ等を用いて混合、混練し、必要に応じて脱泡することによって得ることができる。 The liquid resin composition for electronic parts of the present invention can be prepared by any method as long as the above various components can be uniformly dispersed and mixed. However, as a general method, a component having a predetermined blending amount is weighed. It can be obtained by mixing, kneading using a raking machine, mixing roll, planetary mixer or the like, and defoaming as necessary.
本発明で得られる電子部品用液状樹脂組成物により電子部品を封止して得られる電子部品装置としては、リードフレーム、配線済みのテープキャリア、リジッド及びフレキシブル配線板、ガラス、シリコンウエハ等の支持部材に、半導体チップ、トランジスタ、ダイオード、サイリスタ等の能動素子、コンデンサ、抵抗体、抵抗アレイ、コイル、スイッチ等の受動素子などの素子を搭載し、必要な部分を本発明の電子部品用液状樹脂組成物で封止して得られる電子部品装置などが挙げられる。特に配線基板上に電子部品を直接バンプ接続する電子部品装置の封止に用いられることが好ましい。例えば、リジッド及びフレキシブル配線板やガラス上に形成した配線に半導体素子をバンプ接続によるフリップチップボンディングした半導体装置が対象となる。具体的な例としてはフリップチップBGA/LGAやCOF(Chip On Film)等の半導体装置が挙げられ、本発明で得られる電子部品用液状樹脂組成物は信頼性に優れたフリップチップ用のアンダーフィル材として好適である。また、プリント回路板にも本発明の液状樹脂組成物は有効に使用できる。 Electronic component devices obtained by sealing electronic components with the liquid resin composition for electronic components obtained in the present invention include support for lead frames, wired tape carriers, rigid and flexible wiring boards, glass, silicon wafers, etc. An element such as an active element such as a semiconductor chip, a transistor, a diode, or a thyristor, or a passive element such as a capacitor, a resistor, a resistor array, a coil, or a switch is mounted on the member, and the necessary part is a liquid resin for electronic components according to the present invention. Examples thereof include an electronic component device obtained by sealing with a composition. In particular, it is preferably used for sealing an electronic component device in which an electronic component is directly bump-connected on a wiring board. For example, a semiconductor device in which a semiconductor element is flip-chip bonded by bump connection to a rigid and flexible wiring board or a wiring formed on glass is an object. Specific examples include semiconductor devices such as flip chip BGA / LGA and COF (Chip On Film), and the liquid resin composition for electronic components obtained by the present invention is an underfill for flip chip with excellent reliability. Suitable as a material. The liquid resin composition of the present invention can also be used effectively for printed circuit boards.
本発明の電子部品用液状樹脂組成物が特に好適なフリップチップの分野としては、配線基板と半導体素子を接続するバンプ材質が従来の鉛含有はんだではなく、Sn−Ag−Cu系などの鉛フリーはんだを用いたフリップチップ半導体部品であり、従来の鉛はんだと比較して物性的に脆い鉛フリーはんだバンプ接続をしたフリップチップに対しても良好な信頼性を維持できる。 In the field of flip chip in which the liquid resin composition for electronic parts of the present invention is particularly suitable, the bump material for connecting the wiring substrate and the semiconductor element is not a conventional lead-containing solder, but a lead-free material such as Sn-Ag-Cu. It is a flip chip semiconductor component using solder, and good reliability can be maintained even for a flip chip having a lead-free solder bump connection that is physically brittle compared to conventional lead solder.
さらには、半導体素子のサイズが長い方の辺で5mm以上である大形素子に対して好適であり、電子部品を構成する配線基板と半導体素子のバンプ接続面の距離が80μm以下であるフリップチップ接続に対しても良好な流動性と充填性を示し、耐湿性、耐熱衝撃性等の信頼性にも優れた半導体装置を提供することができる。また、近年半導体素子の高速化に伴い低誘電率の層間絶縁膜が半導体素子に形成されているが、これら低誘電絶縁体は機械強度が弱く、外部からの応力で破壊する故障が発生し易い。この傾向は素子が大きくなる程顕著になり、アンダーフィル材からの応力低減が求められており、半導体素子のサイズが長い方の辺で5mm以上であり、誘電率3.0以下の誘電体層を有する半導体素子を搭載するフリップチップ半導体装置に対しても優れた信頼性を提供できる。 Furthermore, the flip chip is suitable for a large element having a semiconductor element size of 5 mm or more on the longer side, and the distance between the wiring board constituting the electronic component and the bump connection surface of the semiconductor element is 80 μm or less. It is possible to provide a semiconductor device that exhibits good fluidity and filling properties for connection and is excellent in reliability such as moisture resistance and thermal shock resistance. In recent years, an interlayer insulating film having a low dielectric constant is formed on a semiconductor element as the speed of the semiconductor element increases. However, these low dielectric insulators have low mechanical strength and are liable to break down due to external stress. . This tendency becomes more prominent as the element becomes larger, and stress reduction from the underfill material is required. The dielectric layer having a semiconductor element size of 5 mm or more on the longer side and a dielectric constant of 3.0 or less. Excellent reliability can also be provided for a flip-chip semiconductor device on which a semiconductor element having the above is mounted.
本発明の電子部品用液状樹脂組成物を用いて電子部品を封止する方法としては、ディスペンス方式、注型方式、印刷方式等が挙げられる。 Examples of a method for sealing an electronic component using the liquid resin composition for an electronic component of the present invention include a dispensing method, a casting method, and a printing method.
次に実施例により本発明をさらに具体的に説明するが、本発明はこれらの実施例に限定されるものではない。 EXAMPLES Next, although an Example demonstrates this invention further more concretely, this invention is not limited to these Examples.
実施例および比較例において行った特性試験の試験方法を以下にまとめて示す。なお、使用した電子部品用液状樹脂組成物の諸特性及び含浸時間、ボイドの観察、各種信頼性の評価は以下の方法及び条件で行った。(5)〜(9)の含浸時間、ボイドの観察、信頼性の評価に使用した半導体装置は次の2種類のフリップチップBGAを使用した。 The test methods of the characteristic tests performed in the examples and comparative examples are summarized below. In addition, various characteristics and impregnation time of the used liquid resin composition for electronic components, observation of a void, and evaluation of various reliability were performed with the following method and conditions. The following two types of flip chip BGAs were used as semiconductor devices used for the impregnation time, void observation, and reliability evaluation of (5) to (9).
(Non low−k仕様フリップチップBGA)
諸元は、チップサイズ20×20×0.55tmm(回路はアルミのデイジーチェーン接続、パッシベーション:日立化成デュポンマイクロシステムズ製ポリイミド膜商品名HD4000)、バンプ:はんだボール(Sn−Ag−Cu、Φ80μm、 7,744pin、)、バンプピッチ:190μm、基板:FR−5(日立化成工業製ソルダーレジスト商品名SR7000、60×60×0.8tmm)、チップ/基板間のギャップ:50μmである。
(Non low-k specification flip chip BGA)
Specifications are: chip size 20 × 20 × 0.55 tmm (circuit is aluminum daisy chain connection, passivation: polyimide film product name HD4000 manufactured by Hitachi Chemical DuPont Microsystems), bump: solder ball (Sn—Ag—Cu, Φ80 μm, 7, 744 pin,), bump pitch: 190 μm, substrate: FR-5 (trade name SR7000 manufactured by Hitachi Chemical Co., Ltd., 60 × 60 × 0.8 tmm), and chip / substrate gap: 50 μm.
(Low−k仕様フリップチップBGA)
諸元は、チップサイズ20×20×0.55tmm(誘電率2.7の誘電体層を3層形成、回路はアルミのデイジーチェーン接続、パッシベーション:ポリイミド膜HD4000,日立化成デュポンマイクロシステムズ製商品名)、バンプ:はんだボール(Sn−Ag−Cu、Φ80μm、7,744pin、)、バンプピッチ:190μm、基板:FR−5(日立化成工業製ソルダーレジスト商品名SR7000、60×60×0.8tmm)、チップ/基板間のギャップ:50μmである。
(Low-k specification flip chip BGA)
Specifications are: chip size 20 × 20 × 0.55 tmm (dielectric layer with a dielectric constant of 2.7, three layers are formed, aluminum daisy chain connection, passivation: polyimide film HD4000, product name manufactured by Hitachi Chemical DuPont Microsystems) ), Bumps: solder balls (Sn—Ag—Cu, Φ80 μm, 7,744 pin), bump pitch: 190 μm, substrate: FR-5 (trade name SR7000, Hitachi Chemical Co., Ltd., solder resist product name, 60 × 60 × 0.8 tmm) Chip / substrate gap: 50 μm.
半導体装置は、上記BGAへ電子部品用液状樹脂組成物をディスペンス方式でアンダーフィルし、165℃、2時間の加熱条件で硬化することで作製した。また、各種試験片の硬化条件も165℃、2時間とした。 The semiconductor device was produced by underfilling the above BGA with a liquid resin composition for electronic components by a dispensing method and curing it at 165 ° C. for 2 hours. Also, the curing conditions for various test pieces were 165 ° C. and 2 hours.
(1)粘度
電子部品用液状樹脂組成物の25℃での粘度をE型粘度計(コーン角度3°、回転数5rpm)を用いて測定した。
(1) Viscosity The viscosity at 25 ° C. of the liquid resin composition for electronic parts was measured using an E-type viscometer (cone angle 3 °, rotation speed 5 rpm).
(2)表面張力
電子部品用液状樹脂組成物の25℃での表面張力を表面張力測定器DCAT11(英弘精機株式会社製)を用い、プレート法(19.9mm×10mm×0.2mm、白金−イリジウム製ウィルヘルミプレート)により測定した。
(2) Surface tension The surface tension at 25 ° C. of the liquid resin composition for electronic parts was measured using a plate tension method (19.9 mm × 10 mm × 0.2 mm, platinum − Iridium Wilhelmi plate).
(3)接着力・SR接着力
ソルダーレジストSR7000(日立化成工業製商品名)の表面に電子部品用液状樹脂組成物を直径3mm高さ3mmに成形した試験片を作製し、ボンドテスターDS100型(DAGE製)を用いて、ヘッドスピード50μm/sec、25℃の条件でせん断応力をかけ、成形品がソルダーレジストから剥離する強度を測定した。この測定は、試験片成形直後、及び130℃、85%RHのHAST(高度加速寿命試験、Highly Accelerated Temperature and Humidity Stress Test)条件下で150h処理後に行った。
(3) Adhesive strength / SR adhesive strength A test piece obtained by molding a liquid resin composition for electronic parts to a surface of a solder resist SR7000 (trade name, manufactured by Hitachi Chemical Co., Ltd.) with a diameter of 3 mm and a height of 3 mm was prepared. The strength at which the molded product peels from the solder resist was measured by applying a shear stress under the conditions of a head speed of 50 μm / sec and 25 ° C. This measurement was performed immediately after molding of the test piece and after the treatment for 150 hours under HAST (Highly Accelerated Temperature and Humidity Stress Test) conditions at 130 ° C. and 85% RH.
(4)PI接着力
感光性ポリイミドHD4000(日立化成デュポンマイクロシステムズ製商品名)の表面に電子部品用液状樹脂組成物を直径3mm高さ3mmに成形した試験片を作製し、ボンドテスターDS100型(DAGE製)を用いて、ヘッドスピード50μm/sec、25℃の条件でせん断応力をかけ、成形品が感光性ポリイミドから剥離する強度を測定した。この測定は、試験片成形直後、及び130℃、85%RHのHAST条件下で150h処理後に行った。
(4) PI adhesive strength A test piece was prepared by molding a liquid resin composition for electronic parts to a surface of photosensitive polyimide HD4000 (trade name, manufactured by Hitachi Chemical DuPont Microsystems) to a diameter of 3 mm and a height of 3 mm. The strength at which the molded product peels from the photosensitive polyimide was measured by applying a shear stress under the conditions of a head speed of 50 μm / sec and 25 ° C. This measurement was performed immediately after molding of the test piece and after 150 h treatment under HAST conditions of 130 ° C. and 85% RH.
(5)含浸時間
半導体装置を110℃に加熱したホットプレート上に置き、デイスペンサーを用いて電子部品用液状樹脂組成物の所定量をチップの側面(1辺)に滴下し、樹脂組成物が対向する側面に浸透するまでの時間を測定した。
(5) Impregnation time The semiconductor device is placed on a hot plate heated to 110 ° C., and a predetermined amount of the liquid resin composition for electronic components is dropped on the side surface (one side) of the chip using a dispenser. The time until penetration into the opposite side was measured.
(6)ボイド観察
電子部品用液状樹脂組成物をアンダーフィル及び硬化した半導体装置の内部を超音波探傷装置AT−5500(日立建機製)で観察し、ボイドの有無を調べた。
(6) Void Observation The inside of the semiconductor device obtained by underfilling and curing the liquid resin composition for electronic components was observed with an ultrasonic flaw detector AT-5500 (manufactured by Hitachi Construction Machinery Co., Ltd.) to examine the presence or absence of voids.
(7)耐リフロー性
電子部品用液状樹脂組成物をアンダーフィル及び硬化した半導体装置を120℃/12時間加熱乾燥した後、85℃、60%RH下で168時間吸湿させ、遠赤外線加熱方式のリフロー炉(予熱150℃〜180℃で50秒、ピーク温度260℃、250℃以上の加熱時間40秒)中を3回通した後、内部を超音波探傷装置で観察し、樹脂硬化物とチップ及び基板との剥離、樹脂硬化物のクラックの有無を調べ不良パッケージ数/評価パッケージ数で評価した。
(7) Reflow resistance A semiconductor device obtained by underfilling and curing a liquid resin composition for electronic parts is heated and dried at 120 ° C. for 12 hours, and then absorbed at 168 hours under 85 ° C. and 60% RH, and the far infrared heating system is used. After passing through the reflow furnace (preheating 150 ° C-180 ° C for 50 seconds, peak temperature 260 ° C, heating time 40 ° C or more 40 seconds) three times, the inside was observed with an ultrasonic flaw detector, and the cured resin and chip In addition, the presence or absence of peeling from the substrate and cracks in the cured resin was examined and evaluated by the number of defective packages / number of evaluation packages.
(8)耐温度サイクル性
電子部品用液状樹脂組成物をアンダーフィル及び硬化した半導体装置を−50℃〜150℃、各30分のヒートサイクルで1000サイクル処理し、導通試験を行いアルミ配線及びパッドの断線不良を調べ、不良パッケージ数/評価パッケージ数で評価した。
(8) Temperature cycle resistance The semiconductor device obtained by underfilling and curing the liquid resin composition for electronic parts is processed at -50 ° C. to 150 ° C. with a heat cycle of 30 minutes for 1000 cycles, conducting a continuity test, and aluminum wiring and pads. The disconnection failure was checked and evaluated by the number of defective packages / number of evaluation packages.
(9)耐湿信頼性
電子部品用液状樹脂組成物をアンダーフィル及び硬化した半導体装置を130℃、85%RHのHAST条件下で150h処理後、アルミ配線及びパッドの断線有無を導通試験より確認し、不良パッケージ数/評価パッケージ数で評価した。
(9) Moisture resistance reliability After underfilling and curing the semiconductor resin liquid resin composition for electronic parts under HAST conditions of 130 ° C and 85% RH for 150 hours, the presence or absence of disconnection of aluminum wiring and pads was confirmed by continuity test. The number of defective packages / the number of evaluation packages was evaluated.
(実施例1〜13、比較例1〜6)
(A)液状エポキシ樹脂としてビスフェノールFをエポキシ化して得られるエポキシ当量160の液状ジエポキシ樹脂(エポキシ樹脂1、ジャパンエポキシレジン株式会社製商品名jER806)、アミノフェノールをエポキシ化して得られるエポキシ当量95の3官能液状エポキシ樹脂(エポキシ樹脂2、ジャパンエポキシレジン株式会社製商品名jER630)を用意した。
(Examples 1-13, Comparative Examples 1-6)
(A) Epoxy equivalent 160 liquid diepoxy resin obtained by epoxidizing bisphenol F as a liquid epoxy resin (Epoxy resin 1, trade name jER806 manufactured by Japan Epoxy Resin Co., Ltd.), epoxy equivalent 95 obtained by epoxidizing aminophenol A trifunctional liquid epoxy resin (epoxy resin 2, trade name jER630 manufactured by Japan Epoxy Resin Co., Ltd.) was prepared.
(B)硬化剤として活性水素当量45のジエチルトルエンジアミン(液状アミン1、ジャパンエポキシレジン株式会社製商品名jERキュアW)、活性水素当量63のジエチル−ジアミノ−ジフェニルメタン(液状アミン2、日本化薬株式会社製商品名カヤハードA−A)、比較のために、活性水素当量63のテトラメチル−ジアミノ−ジフェニルメタン(固形アミン、日本化薬株式会社製商品名カヤボンドC−200S)、酸無水物当量168の液状酸無水物(日立化成工業株式会社製商品名HN5500)を用意した。 (B) Diethyltoluenediamine having 45 active hydrogen equivalents (Liquid amine 1, trade name jER Cure W manufactured by Japan Epoxy Resin Co., Ltd.), diethyl-diamino-diphenylmethane having 63 active hydrogen equivalents (Liquid amine 2, Nippon Kayaku) Trade name Kayahard AA), for comparison, tetramethyl-diamino-diphenylmethane (solid amine, Nippon Kayaku Co., Ltd. trade name Kayabond C-200S), acid anhydride equivalent 168 for comparison. Liquid acid anhydride (trade name HN5500, manufactured by Hitachi Chemical Co., Ltd.) was prepared.
(C)非イオン性界面活性剤としてポリエーテル変性シリコーン系界面活性剤(ビックケミー製商品名BYK−377、界面活性剤1)、アラルキル変性シリコーン系界面活性剤(ビックケミー製商品名BYK−323、界面活性剤2)、水酸基当量750、重量平均分子量1600のフェノール変性シリコーン(東レ・ダウコーニング・シリコーン株式会社製商品名BY16−799)とビスフェノールF型液状エポキシ樹脂(jER806)を重量比1/1で加熱混融して得られたシリコーン変性エポキシ樹脂(界面活性剤3)、活性水素当量130、重量平均分子量470のフェノール変性シリコーン(東レ・ダウコーニング・シリコーン株式会社製商品名BY16−871)とビスフェノールF型液状エポキシ樹脂(ジャパンエポキシレジン株式会社製商品名jER806)を重量比1/1で加熱混融して得られたシリコーン変性エポキシ樹脂(界面活性剤4)、水酸基当量3500、重量平均分子量8500のカルビノール変性シリコーン(東レ・ダウコーニング・シリコーン製商品名BY16−004)とビスフェノールF型液状エポキシ樹脂(ジャパンエポキシレジン株式会社製商品名jER806)を重量比1/1で加熱混融して得られたシリコーン変性エポキシ樹脂(界面活性剤5)、ポリオキシエチレンアルキルエーテル系界面活性剤(東邦化学工業株式会社製商品名ペグノールL−4、界面活性剤6)を用意した。 (C) Polyether-modified silicone surfactant (BIC Chemie trade name BYK-377, surfactant 1), aralkyl-modified silicone surfactant (BIC Chemie trade name BYK-323, interface) as nonionic surfactant Activator 2), phenol-modified silicone having a hydroxyl equivalent weight of 750 and a weight average molecular weight of 1600 (trade name BY16-799 manufactured by Toray Dow Corning Silicone Co., Ltd.) and bisphenol F type liquid epoxy resin (jER806) at a weight ratio of 1/1. Silicone-modified epoxy resin (surfactant 3) obtained by heat-mixing, phenol-modified silicone (trade name BY16-871 manufactured by Toray Dow Corning Silicone Co., Ltd.) and bisphenol having an active hydrogen equivalent weight of 130 and a weight average molecular weight of 470 F-type liquid epoxy resin (Japan Epoxy Resin Co., Ltd., trade name jER806), a silicone-modified epoxy resin (surfactant 4) obtained by heating and fusing at a 1/1 weight ratio, a carbinol-modified silicone having a hydroxyl group equivalent of 3500 and a weight average molecular weight of 8500 (Toray Silicone modified epoxy resin (interface) obtained by heating and fusing Dow Corning Silicone product name BY16-004) and bisphenol F type liquid epoxy resin (Japan Epoxy Resin Co., Ltd. product name jER806) at a weight ratio of 1/1. Activators 5) and polyoxyethylene alkyl ether surfactants (trade name Pegnol L-4, surfactant 6 manufactured by Toho Chemical Industry Co., Ltd.) were prepared.
可撓化剤としてジメチル型固形シリコーンゴム粒子の表面がエポキシ基で修飾された、平均粒径2μmの球状のシリコーン微粒子(東レ・ダウコーニング・シリコーン株式会社製商品名トレフィルE−601)、硬化促進剤として2−フェニル−4,5−ジヒドロキシメチルイミダゾール、シランカップリング剤としてγ−グリシドキシプロピルトリメトキシシラン(チッソ株式会社製商品名サイラエースS510)、着色剤としてカーボンブラック(三菱化学株式会社製商品名MA‐100)、イオントラップ剤としてビスマス系イオントラップ剤(東亞合成株式会社製商品名IXE−500)、(D)無機充填剤として平均粒径1μmの球状溶融シリカを用意した。 Spherical silicone fine particles with a mean particle size of 2 μm (trade name Treffil E-601 manufactured by Toray Dow Corning Silicone Co., Ltd.) whose surface of dimethyl type solid silicone rubber particles is modified with an epoxy group as a flexibilizing agent, acceleration of curing 2-phenyl-4,5-dihydroxymethylimidazole as the agent, γ-glycidoxypropyltrimethoxysilane (trade name: Silaace S510, manufactured by Chisso Corporation) as the silane coupling agent, and carbon black (manufactured by Mitsubishi Chemical Corporation) as the colorant (Trade name MA-100), a bismuth ion trapping agent (trade name IXE-500, manufactured by Toagosei Co., Ltd.) as an ion trapping agent, and (D) spherical fused silica having an average particle diameter of 1 μm as an inorganic filler.
これらをそれぞれ下記表1から表3に示す組成で配合し、三本ロール及び真空擂潰機にて混練分散した後、実施例1〜13及び比較例1〜6の電子部品用液状樹脂組成物を作製した。なお表1〜3中の配合単位は質量部であり、また空欄は配合無しを表す。
各種評価結果を表4から表6に示す。
本発明における(C)成分の非イオン性界面活性剤を含まない比較例1及び2では、表面張力が大きく、HAST処理後の接着力、ボイド、耐湿信頼性及び耐温度サイクル性が著しく劣っていた。また、(C)成分の界面活性剤を含み硬化剤が固形アミンの比較例3、(C)成分の界面活性剤を含まず硬化剤が固形アミンの比較例5は高粘度のため含浸時間が長く生産に劣り、またボイド起因によって耐湿信頼性及び耐熱衝撃性が低下した。さらに、(C)成分の界面活性剤を含み硬化剤が液状酸無水物の比較例4、(C)成分の界面活性剤を含まず硬化剤が液状酸無水物の比較例6は、成形性には優れるものの耐湿接着力が低いため、耐リフロー性、及び耐温度サイクル性、耐湿信頼性が著しく劣っていた。これに対して、実施例1〜13は、表面張力が低く成形時のボイド性、HAST処理後の接着性の向上が図られ、耐リフロー性、耐温度サイクル性及び耐湿性の各種信頼性に優れる。 In Comparative Examples 1 and 2 that do not contain the nonionic surfactant of component (C) in the present invention, the surface tension is large, and the adhesive strength, void, moisture resistance reliability, and temperature cycle resistance after HAST treatment are extremely inferior. It was. In addition, the comparative example 3 in which the curing agent is a solid amine containing a surfactant (C) and the curing agent is a solid amine that does not contain the surfactant (C), and the curing agent is a solid amine. It was inferior in production for a long time, and the moisture resistance reliability and thermal shock resistance decreased due to voids. Furthermore, the comparative example 4 in which the surfactant (C) contains a curing agent and is a liquid acid anhydride, and the comparative example 6 in which the curing agent does not contain a surfactant (C) and a liquid acid anhydride is a moldability. However, the reflow resistance, the temperature cycle resistance, and the moisture resistance reliability were remarkably inferior due to the low moisture adhesion strength. On the other hand, in Examples 1 to 13, the surface tension is low, the void property at the time of molding and the adhesion after HAST treatment are improved, and various reliability of reflow resistance, temperature cycle resistance and moisture resistance is achieved. Excellent.
Claims (12)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2006332101A JP5114935B2 (en) | 2005-12-08 | 2006-12-08 | Liquid resin composition for electronic components, and electronic component device using the same |
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2005354720 | 2005-12-08 | ||
| JP2005354720 | 2005-12-08 | ||
| JP2006332101A JP5114935B2 (en) | 2005-12-08 | 2006-12-08 | Liquid resin composition for electronic components, and electronic component device using the same |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2007182561A JP2007182561A (en) | 2007-07-19 |
| JP5114935B2 true JP5114935B2 (en) | 2013-01-09 |
Family
ID=38338910
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2006332101A Active JP5114935B2 (en) | 2005-12-08 | 2006-12-08 | Liquid resin composition for electronic components, and electronic component device using the same |
Country Status (1)
| Country | Link |
|---|---|
| JP (1) | JP5114935B2 (en) |
Families Citing this family (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP5277537B2 (en) * | 2005-12-08 | 2013-08-28 | 日立化成株式会社 | Liquid resin composition for electronic components and electronic component device using the same |
| JP5692212B2 (en) * | 2005-12-08 | 2015-04-01 | 日立化成株式会社 | Liquid resin composition for electronic components and electronic component device using the same |
| WO2007066763A1 (en) | 2005-12-08 | 2007-06-14 | Hitachi Chemical Co., Ltd. | Liquid resin composition for electronic element and electronic element device |
| JP2010095702A (en) * | 2008-09-18 | 2010-04-30 | Sumitomo Bakelite Co Ltd | Resin composition, liquid resin composition for sealing semiconductor, liquid resin composition for underfill, and semiconductor device |
| JP5445005B2 (en) * | 2009-10-05 | 2014-03-19 | 日立化成株式会社 | Epoxy resin composition, semiconductor sealing resin composition, and semiconductor device |
| JP5579764B2 (en) * | 2012-03-05 | 2014-08-27 | 信越化学工業株式会社 | Liquid epoxy resin composition for underfill and semiconductor device sealed with the composition |
| JP2015012273A (en) * | 2013-07-02 | 2015-01-19 | Tdk株式会社 | Dust core and electronic components |
| JP6679849B2 (en) * | 2015-07-01 | 2020-04-15 | 味の素株式会社 | Resin composition |
| JP7000698B2 (en) * | 2017-03-31 | 2022-01-19 | 昭和電工マテリアルズ株式会社 | Resin composition for underfill, manufacturing method of semiconductor device and semiconductor device |
| US20190345325A1 (en) * | 2018-05-11 | 2019-11-14 | Samsung Electronics Co., Ltd. | Resin composition for printed circuit board and integrated circuit package, and product using the same |
| JP7241389B2 (en) * | 2019-02-21 | 2023-03-17 | ナミックス株式会社 | Liquid epoxy resin composition and cured product obtained by curing it |
Family Cites Families (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS6284147A (en) * | 1985-10-07 | 1987-04-17 | Shin Etsu Chem Co Ltd | epoxy resin composition |
| JPH02281023A (en) * | 1989-04-21 | 1990-11-16 | Sumitomo Bakelite Co Ltd | Curable epoxy resin composition |
| JP3351974B2 (en) * | 1996-12-05 | 2002-12-03 | 住友ベークライト株式会社 | Liquid injection underfill material |
| JP3835715B2 (en) * | 1997-02-18 | 2006-10-18 | 住友ベークライト株式会社 | Liquid injection sealed underfill material |
| JP4053152B2 (en) * | 1998-09-07 | 2008-02-27 | 住友ベークライト株式会社 | Thermosetting resin composition and semiconductor device thereof |
| JP2002121259A (en) * | 2000-10-19 | 2002-04-23 | Sumitomo Bakelite Co Ltd | Liquid sealing resin composition, method for producing semiconductor device and semiconductor device |
| JP2004182479A (en) * | 2002-11-29 | 2004-07-02 | Mitsubishi Rayon Co Ltd | Surface-modified spherical silica, method for producing the same, and resin composition for encapsulating semiconductor |
| JP4009853B2 (en) * | 2003-05-23 | 2007-11-21 | 信越化学工業株式会社 | Liquid epoxy resin composition and flip chip type semiconductor device |
| JP4337405B2 (en) * | 2003-05-30 | 2009-09-30 | 住友ベークライト株式会社 | Method for producing liquid epoxy resin composition for semiconductor encapsulation, and semiconductor device using liquid epoxy resin composition for semiconductor encapsulation |
| JP2005146104A (en) * | 2003-11-14 | 2005-06-09 | Shin Etsu Chem Co Ltd | Liquid epoxy resin composition and semiconductor device |
| JP4557148B2 (en) * | 2003-12-12 | 2010-10-06 | 信越化学工業株式会社 | Liquid epoxy resin composition and semiconductor device |
-
2006
- 2006-12-08 JP JP2006332101A patent/JP5114935B2/en active Active
Also Published As
| Publication number | Publication date |
|---|---|
| JP2007182561A (en) | 2007-07-19 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN102675599B (en) | Method for manufacturing semiconductor device | |
| JP5228426B2 (en) | Liquid resin composition for electronic component sealing and electronic component device using the same | |
| CN105315618B (en) | Electronic component-use liquid resin composition and its manufacturing method and electronic part apparatus | |
| CN104629262B (en) | Electronic component-use liquid resin composition and electronic part apparatus | |
| CN101321799B (en) | Liquid resin composition for electronic element and electronic element device | |
| CN101851388B (en) | Liquid resin composition for electronic component and electronic component device | |
| JP5692212B2 (en) | Liquid resin composition for electronic components and electronic component device using the same | |
| JP2013028659A (en) | Epoxy resin liquid sealing material for underfill and electric component apparatus using the same | |
| CN101313006A (en) | Liquid resin composition for electronic component and electronic component device | |
| JP7167912B2 (en) | Liquid encapsulating resin composition, electronic component device, and method for manufacturing electronic component device | |
| JP5114935B2 (en) | Liquid resin composition for electronic components, and electronic component device using the same | |
| JP5277537B2 (en) | Liquid resin composition for electronic components and electronic component device using the same | |
| JP2007182562A (en) | Liquid resin composition for electronic element and electronic element device | |
| JP2004051734A (en) | Liquid epoxy resin composition and semiconductor device | |
| JP6286959B2 (en) | Epoxy resin composition, electronic component device, and method of manufacturing electronic component device | |
| JP2018048276A (en) | Underfill material and electronic component device using the same | |
| JP6686433B2 (en) | Underfill resin composition, electronic component device, and method for manufacturing electronic component device | |
| JP2015054952A (en) | Epoxy resin composition, electronic component device, and method of manufacturing electronic component device | |
| JP2017028050A (en) | Underfill material and electronic component device using the same | |
| JP7786113B2 (en) | Liquid underfill resin composition, electronic component device, and method for manufacturing electronic component device | |
| JP2019081816A (en) | Liquid resin composition for underfill, electronic part device and method for manufacturing electronic part device | |
| JP2021014588A (en) | Resin composition for underfill, electronic component device and method for producing electronic component device |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20090608 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20110928 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20111004 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20111202 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20120110 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20120312 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20120918 |
|
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20121001 |
|
| R151 | Written notification of patent or utility model registration |
Ref document number: 5114935 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R151 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20151026 Year of fee payment: 3 |
|
| S531 | Written request for registration of change of domicile |
Free format text: JAPANESE INTERMEDIATE CODE: R313531 |
|
| S533 | Written request for registration of change of name |
Free format text: JAPANESE INTERMEDIATE CODE: R313533 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20151026 Year of fee payment: 3 |
|
| R350 | Written notification of registration of transfer |
Free format text: JAPANESE INTERMEDIATE CODE: R350 |
|
| S531 | Written request for registration of change of domicile |
Free format text: JAPANESE INTERMEDIATE CODE: R313531 |
|
| S533 | Written request for registration of change of name |
Free format text: JAPANESE INTERMEDIATE CODE: R313533 |
|
| R350 | Written notification of registration of transfer |
Free format text: JAPANESE INTERMEDIATE CODE: R350 |
|
| S531 | Written request for registration of change of domicile |
Free format text: JAPANESE INTERMEDIATE CODE: R313531 |
|
| R350 | Written notification of registration of transfer |
Free format text: JAPANESE INTERMEDIATE CODE: R350 |
|
| S801 | Written request for registration of abandonment of right |
Free format text: JAPANESE INTERMEDIATE CODE: R311801 |
|
| ABAN | Cancellation due to abandonment | ||
| R350 | Written notification of registration of transfer |
Free format text: JAPANESE INTERMEDIATE CODE: R350 |