KR20060017618A - 로르녹시캄 또는 로르녹시캄-유사체의 용도 - Google Patents
로르녹시캄 또는 로르녹시캄-유사체의 용도 Download PDFInfo
- Publication number
- KR20060017618A KR20060017618A KR1020057022636A KR20057022636A KR20060017618A KR 20060017618 A KR20060017618 A KR 20060017618A KR 1020057022636 A KR1020057022636 A KR 1020057022636A KR 20057022636 A KR20057022636 A KR 20057022636A KR 20060017618 A KR20060017618 A KR 20060017618A
- Authority
- KR
- South Korea
- Prior art keywords
- lornoxicam
- cyclopenta
- pyridine
- methyl
- dihydrochloride
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/54—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with at least one nitrogen and one sulfur as the ring hetero atoms, e.g. sulthiame
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/44—Non condensed pyridines; Hydrogenated derivatives thereof
- A61K31/4427—Non condensed pyridines; Hydrogenated derivatives thereof containing further heterocyclic ring systems
- A61K31/4439—Non condensed pyridines; Hydrogenated derivatives thereof containing further heterocyclic ring systems containing a five-membered ring with nitrogen as a ring hetero atom, e.g. omeprazole
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/28—Drugs for disorders of the nervous system for treating neurodegenerative disorders of the central nervous system, e.g. nootropic agents, cognition enhancers, drugs for treating Alzheimer's disease or other forms of dementia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
Landscapes
- Health & Medical Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Veterinary Medicine (AREA)
- Medicinal Chemistry (AREA)
- Public Health (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Epidemiology (AREA)
- Engineering & Computer Science (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Organic Chemistry (AREA)
- Biomedical Technology (AREA)
- Neurosurgery (AREA)
- Neurology (AREA)
- Cardiology (AREA)
- Hospice & Palliative Care (AREA)
- Heart & Thoracic Surgery (AREA)
- Vascular Medicine (AREA)
- Psychiatry (AREA)
- Urology & Nephrology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Nitrogen And Oxygen Or Sulfur-Condensed Heterocyclic Ring Systems (AREA)
- Saccharide Compounds (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Heterocyclic Carbon Compounds Containing A Hetero Ring Having Oxygen Or Sulfur (AREA)
Abstract
Description
Claims (3)
- 알츠하이머병(AE) 또는 동맥경화증의 치료 또는 억제를 위한 의약 조성물을 제조하기 위해,- 시클로옥시게나제 1 및 시클로옥시게나제 2(COX 1 및 COX 2)를 저지하고,- 생리학적인 조건 하에서 혈액/뇌-배리어를 넘을 수 없으며,- 아밀로이드 전구 단백질(APP)의 프로스타글란딘 E2-유도된 유도 작용을 감소시키는, 로르녹시캄 또는 로르녹시캄-유사체의 용도.
- 제 1 항에 있어서,상기 물질의 투여가 바람직하지 않은 심각한 결과들을 전혀 야기하지 않는 것을 특징으로 하는, 로르녹시캄 또는 로르녹시캄-유사체의 용도.
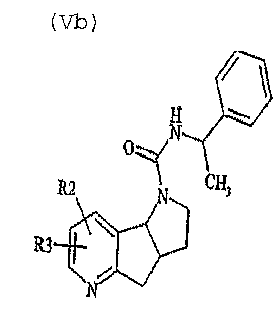
- 제 1 항 또는 제 2 항에 있어서,상기 로르녹시캄-유사체가6-클로르-4-(1-(에톡시카르바모일옥시)에톡시)-2-메틸-N-(2-피리딜)-2H-티에노-(2,3-e)-1,2-티아진-3-카르복실산아미드-1,1-디옥시드,6-클로르-4-하이드록시-2-메틸-3-(2-피리딜-카르바모일)-2H-티에노[3,2-e]1,2-티아진-1,1-디옥시드,(+)-[3aα,8bα]-1,2,3,3a,4,8b-헥사하이드로피롤로[2',3':3,4]-시클로펜타[1,2-b]피리딘-디하이드로클로라이드;(+)-[3aS-(3aα,8bα]-1,2,3,3a,4,8b-헥사하이드로피롤로[3',2':4,5]-시클로펜타[1,2-c]피리딘-디하이드로클로라이드;(-)-[3aR-(3aα,8bα]-1,2,3,3a,4,8b-헥사하이드로피롤로[3',2':4,5]-시클로펜타[1,2-c]피리딘-디하이드로클로라이드;(-)-[3aα,8bα]-1,2,3,3a,4,8b-헥사하이드로피롤로[2',3':3,4]-시클로펜타[1,2-b]피리딘-디하이드로클로라이드;(-)[3aα,8bα]-1,2,3,3a,4,8b-헥사하이드로-1-메틸-피롤로-[2',3':3,4]-시클로펜타[1,2-b]피리딘-디하이드로클로라이드;(+)-[3aS-(3aα,8bα]-1,2,3,3a,4,8b-헥사하이드로-1-메틸-피롤로-[3',2':4,5]-시클로펜타[1,2-c]피리딘-디하이드로클로라이드;(+)-[3aα,8bα]-1,2,3,3a,4,8b-헥사하이드로-1-메틸-피롤로-[2',3':3,4]-시클로펜타[1,2-b]피리딘-디하이드로클로라이드;(+)-[3aS-(3aα,8bα]-1,2,3,3a,4,8b-헥사하이드로-1-메틸-피롤로-[3',2':4,5]-시클로펜타[1,2-c]피리딘-디하이드로클로라이드 그룹으로부터 선택되는 것을 특징으로 하는, 로르녹시캄 또는 로르녹시캄-유사체의 용도.
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AT0081903A AT413944B (de) | 2003-05-27 | 2003-05-27 | Verwendung von oxicam-verbindungen |
| ATA819/2003 | 2003-05-27 | ||
| PCT/AT2004/000185 WO2004105766A2 (de) | 2003-05-27 | 2004-05-27 | Verwendung von lornoxicam oder lornoxicam-analoga verbindungen |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| KR20060017618A true KR20060017618A (ko) | 2006-02-24 |
| KR101134244B1 KR101134244B1 (ko) | 2012-04-09 |
Family
ID=33479909
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020057022636A Expired - Fee Related KR101134244B1 (ko) | 2003-05-27 | 2004-05-27 | 로르녹시캄 또는 로르녹시캄-유사체의 용도 |
Country Status (13)
| Country | Link |
|---|---|
| US (2) | US8129370B2 (ko) |
| EP (1) | EP1626726B1 (ko) |
| JP (1) | JP5597339B2 (ko) |
| KR (1) | KR101134244B1 (ko) |
| CN (3) | CN1842338A (ko) |
| AT (2) | AT413944B (ko) |
| CA (1) | CA2526816C (ko) |
| DE (1) | DE502004001499D1 (ko) |
| DK (1) | DK1626726T3 (ko) |
| ES (1) | ES2271893T3 (ko) |
| PL (1) | PL1626726T3 (ko) |
| RU (1) | RU2366425C2 (ko) |
| WO (1) | WO2004105766A2 (ko) |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CA2563575C (en) * | 2004-04-29 | 2012-12-18 | Binder, Eva | Enantiomerically pure hexahydropyrrolocyclopentapyridine derivatives |
| CA2742771A1 (en) * | 2008-11-13 | 2010-05-20 | Modgene, Llc | Modification of amyloid-beta load in non-brain tissue |
| CA2853961C (en) | 2011-11-01 | 2016-09-20 | Modgene, Llc | Compositions and methods for reduction of amyloid-beta load |
| CN115227655A (zh) * | 2022-08-17 | 2022-10-25 | 海南锦瑞制药有限公司 | 注射用氯诺昔康的制备方法 |
Family Cites Families (18)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| AU518216B2 (en) | 1977-09-06 | 1981-09-17 | Hafslund Nycomed Pharma Aktiengesellschaft | Thienothiazine derivatives |
| JPS58146040A (ja) * | 1982-02-24 | 1983-08-31 | Pioneer Video Kk | 光学式情報記録原盤 |
| DE3371082D1 (de) | 1982-09-09 | 1987-05-27 | Hoffmann La Roche | Thieno(2,3-e)-1,2-thiazine derivatives |
| NZ226477A (en) * | 1987-10-29 | 1990-07-26 | Cl Pharma | Substituted thieno (2,3-e)-1,2 thiazine derivatives and pharmaceutical compositions |
| US5192753A (en) * | 1991-04-23 | 1993-03-09 | Mcgeer Patrick L | Anti-rheumatoid arthritic drugs in the treatment of dementia |
| AT400437B (de) * | 1993-12-14 | 1995-12-27 | Chem Pharm Forsch Gmbh | Neue n-heterocyclische thienothiazincarboxamide, verfahren zu ihrer herstellung und ihre verwendung |
| AT400567B (de) * | 1993-12-14 | 1996-01-25 | Chem Pharm Forsch Gmbh | Neue arylsubstituierte thienothiazinderivate, verfahren zu ihrer herstellung und ihre verwendung |
| EP0658559A1 (de) * | 1993-12-14 | 1995-06-21 | Chemisch Pharmazeutische Forschungsgesellschaft m.b.H. | Thienothiazinderivate, Verfahren zu ihrer Herstellung und ihre Verwendung als 5-dipoxygenase und Cyclooxygenaseinhibitoren |
| US5643960A (en) * | 1994-04-15 | 1997-07-01 | Duke University | Method of delaying onset of alzheimer's disease symptoms |
| US6184248B1 (en) * | 1996-09-05 | 2001-02-06 | Robert K. K. Lee | Compositions and methods for treatment of neurological disorders and neurodegenerative diseases |
| CA2279651A1 (en) | 1996-09-05 | 1998-03-12 | Massachusetts Institute Of Technology | Compositions and methods for treatment of neurological disorders and neurodegenerative diseases |
| WO2001021259A2 (en) * | 1999-09-21 | 2001-03-29 | Emory University | Use and compositions for treating platelet-related disorders using anagrelide |
| CA2406383A1 (en) * | 2000-04-13 | 2001-10-25 | Mayo Foundation For Medical Education And Research | A.beta.42 lowering agents |
| US6429223B1 (en) * | 2000-06-23 | 2002-08-06 | Medinox, Inc. | Modified forms of pharmacologically active agents and uses therefor |
| PE20020322A1 (es) * | 2000-08-18 | 2002-05-10 | Pharmacia Corp | Formulacion oral de rapida disolucion de un inhibidor de la ciclooxigenasa 2 |
| JP2002363104A (ja) | 2001-04-04 | 2002-12-18 | Sankyo Co Ltd | 炎症性サイトカイン産生抑制剤の新規医薬用途 |
| US7008614B2 (en) * | 2001-08-20 | 2006-03-07 | Fuji Photo Film Co., Ltd. | Liposome containing hydrophobic iodine compound and X-ray contrast medium for radiograph comprising the liposome |
| US20030225054A1 (en) * | 2002-06-03 | 2003-12-04 | Jingwu Duan | Combined use of tace inhibitors and COX2 inhibitors as anti-inflammatory agents |
-
2003
- 2003-05-27 AT AT0081903A patent/AT413944B/de not_active IP Right Cessation
-
2004
- 2004-05-27 EP EP04734972A patent/EP1626726B1/de not_active Expired - Lifetime
- 2004-05-27 CN CNA2004800143442A patent/CN1842338A/zh active Pending
- 2004-05-27 CN CN2011100997401A patent/CN102218067A/zh active Pending
- 2004-05-27 DE DE502004001499T patent/DE502004001499D1/de not_active Expired - Lifetime
- 2004-05-27 KR KR1020057022636A patent/KR101134244B1/ko not_active Expired - Fee Related
- 2004-05-27 AT AT04734972T patent/ATE339206T1/de active
- 2004-05-27 JP JP2006529424A patent/JP5597339B2/ja not_active Expired - Fee Related
- 2004-05-27 CN CNA2008101754015A patent/CN101450057A/zh active Pending
- 2004-05-27 ES ES04734972T patent/ES2271893T3/es not_active Expired - Lifetime
- 2004-05-27 DK DK04734972T patent/DK1626726T3/da active
- 2004-05-27 US US10/558,117 patent/US8129370B2/en not_active Expired - Fee Related
- 2004-05-27 RU RU2005136864/15A patent/RU2366425C2/ru not_active IP Right Cessation
- 2004-05-27 WO PCT/AT2004/000185 patent/WO2004105766A2/de not_active Ceased
- 2004-05-27 CA CA2526816A patent/CA2526816C/en not_active Expired - Fee Related
- 2004-05-27 PL PL04734972T patent/PL1626726T3/pl unknown
-
2009
- 2009-04-03 US US12/418,339 patent/US20090239852A1/en not_active Abandoned
Also Published As
| Publication number | Publication date |
|---|---|
| CA2526816C (en) | 2012-11-27 |
| US8129370B2 (en) | 2012-03-06 |
| EP1626726B1 (de) | 2006-09-13 |
| DK1626726T3 (da) | 2006-12-27 |
| CA2526816A1 (en) | 2004-12-09 |
| CN102218067A (zh) | 2011-10-19 |
| US20090239852A1 (en) | 2009-09-24 |
| KR101134244B1 (ko) | 2012-04-09 |
| US20070275958A1 (en) | 2007-11-29 |
| WO2004105766A3 (de) | 2005-03-03 |
| RU2366425C2 (ru) | 2009-09-10 |
| CN1842338A (zh) | 2006-10-04 |
| DE502004001499D1 (de) | 2006-10-26 |
| JP2006528206A (ja) | 2006-12-14 |
| ATE339206T1 (de) | 2006-10-15 |
| EP1626726A2 (de) | 2006-02-22 |
| ES2271893T3 (es) | 2007-04-16 |
| CN101450057A (zh) | 2009-06-10 |
| WO2004105766A2 (de) | 2004-12-09 |
| RU2005136864A (ru) | 2006-06-10 |
| PL1626726T3 (pl) | 2007-02-28 |
| ATA8192003A (de) | 2005-11-15 |
| AT413944B (de) | 2006-07-15 |
| JP5597339B2 (ja) | 2014-10-01 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR102220175B1 (ko) | 헤테로사이클릭 글루타미나아제 억제제 | |
| WO2009033281A1 (en) | Cancer combination therapy with a selective inhibitor of histone deacetylase hdac1, hdac2 and/or hdac3 and a microtubule stabilizer | |
| JP6630374B2 (ja) | τリン酸化を阻害する方法 | |
| US20100324029A1 (en) | Therapeutic agents | |
| US20100222320A1 (en) | Triazole derivatives for treating alzheimer's disease and related conditions | |
| JP5747142B2 (ja) | Dyrkを阻害する化合物を含有する医薬組成物 | |
| KR20100101055A (ko) | 세포핵에서 세포질로의 gsk3의 이동을 억제하는 화합물을 함유하는 세포핵에서 세포질로의 gsk3 이동 관련 질환의 치료 또는 예방용 약학적 조성물 | |
| JP2010517934A (ja) | インドール化合物 | |
| TW200824678A (en) | Methods and compositions for the treatment of neurodegenerative disorders | |
| CA2720908A1 (fr) | Molecules inhibant une voie metabolique impliquant la proteine tyrosine kinase syk et procede d'identification de ces molecules | |
| JP2018514524A (ja) | 5−芳香族アルキニル基置換ベンズアミド系化合物並びにその製造方法、薬物組成物及び使用 | |
| KR20170117170A (ko) | 위 배출을 감소시키기 위한 방법 및 조성물 | |
| BR112014031531B1 (pt) | Composto, composição farmacêutica, métodos de tratamento ou prevenção de uma patologia relacionada com ab, e, de tratamento ou prevenção de mal de alzheimer em um paciente em necessidade do mesmo | |
| JPH08239369A (ja) | 一酸化窒素合成酵素の阻害物質としての2− アミノ− 1,3− チアジン | |
| US12310967B2 (en) | Compositions and methods of treatment for neurological disorders comprising motor neuron diseases | |
| JP2016528266A (ja) | 位相変位睡眠障害を処置するためのV1aアンタゴニスト | |
| WO2022271840A1 (en) | Selective indazole lrrk2 inhibitors and methods for use thereof | |
| CA2676051A1 (en) | Insulin sensitisers and methods of treatment | |
| KR20060017618A (ko) | 로르녹시캄 또는 로르녹시캄-유사체의 용도 | |
| WO2023004280A1 (en) | Selective pyrazole lrrk2 inhibitors and methods for use thereof | |
| JP2021522235A (ja) | 抗がんインデン、インダン、アザインデン、アザインダン、医薬組成物および使用 | |
| KR102571296B1 (ko) | P-tau를 감소시키고 인지를 개선하는 알로스테릭 코르티코트로핀-방출 인자 수용체 1 (crfr1) 길항제 | |
| CA2658627A1 (en) | Imidazothiazole derivatives as mark inhibitors | |
| CN108997328B (zh) | 亚氨基噻二嗪二氧化物衍生物及其用途 | |
| FR2774596A1 (fr) | Utilisation de composes selenies dans la prevention et le traitement de la maladie d'alzheimer |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0105 | International application |
St.27 status event code: A-0-1-A10-A15-nap-PA0105 |
|
| PG1501 | Laying open of application |
St.27 status event code: A-1-1-Q10-Q12-nap-PG1501 |
|
| P11-X000 | Amendment of application requested |
St.27 status event code: A-2-2-P10-P11-nap-X000 |
|
| P13-X000 | Application amended |
St.27 status event code: A-2-2-P10-P13-nap-X000 |
|
| R15-X000 | Change to inventor requested |
St.27 status event code: A-3-3-R10-R15-oth-X000 |
|
| R16-X000 | Change to inventor recorded |
St.27 status event code: A-3-3-R10-R16-oth-X000 |
|
| A201 | Request for examination | ||
| P11-X000 | Amendment of application requested |
St.27 status event code: A-2-2-P10-P11-nap-X000 |
|
| P13-X000 | Application amended |
St.27 status event code: A-2-2-P10-P13-nap-X000 |
|
| PA0201 | Request for examination |
St.27 status event code: A-1-2-D10-D11-exm-PA0201 |
|
| N231 | Notification of change of applicant | ||
| PN2301 | Change of applicant |
St.27 status event code: A-3-3-R10-R13-asn-PN2301 St.27 status event code: A-3-3-R10-R11-asn-PN2301 |
|
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
St.27 status event code: A-1-2-D10-D21-exm-PE0902 |
|
| T11-X000 | Administrative time limit extension requested |
St.27 status event code: U-3-3-T10-T11-oth-X000 |
|
| T11-X000 | Administrative time limit extension requested |
St.27 status event code: U-3-3-T10-T11-oth-X000 |
|
| E13-X000 | Pre-grant limitation requested |
St.27 status event code: A-2-3-E10-E13-lim-X000 |
|
| P11-X000 | Amendment of application requested |
St.27 status event code: A-2-2-P10-P11-nap-X000 |
|
| P13-X000 | Application amended |
St.27 status event code: A-2-2-P10-P13-nap-X000 |
|
| E701 | Decision to grant or registration of patent right | ||
| PE0701 | Decision of registration |
St.27 status event code: A-1-2-D10-D22-exm-PE0701 |
|
| GRNT | Written decision to grant | ||
| PR0701 | Registration of establishment |
St.27 status event code: A-2-4-F10-F11-exm-PR0701 |
|
| PR1002 | Payment of registration fee |
St.27 status event code: A-2-2-U10-U12-oth-PR1002 Fee payment year number: 1 |
|
| PG1601 | Publication of registration |
St.27 status event code: A-4-4-Q10-Q13-nap-PG1601 |
|
| R18-X000 | Changes to party contact information recorded |
St.27 status event code: A-5-5-R10-R18-oth-X000 |
|
| PN2301 | Change of applicant |
St.27 status event code: A-5-5-R10-R11-asn-PN2301 |
|
| PN2301 | Change of applicant |
St.27 status event code: A-5-5-R10-R14-asn-PN2301 |
|
| FPAY | Annual fee payment |
Payment date: 20141230 Year of fee payment: 4 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 4 |
|
| FPAY | Annual fee payment |
Payment date: 20160106 Year of fee payment: 5 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 5 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 6 |
|
| FPAY | Annual fee payment |
Payment date: 20180213 Year of fee payment: 7 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 7 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 8 |
|
| FPAY | Annual fee payment |
Payment date: 20191224 Year of fee payment: 9 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 9 |
|
| PC1903 | Unpaid annual fee |
St.27 status event code: A-4-4-U10-U13-oth-PC1903 Not in force date: 20210331 Payment event data comment text: Termination Category : DEFAULT_OF_REGISTRATION_FEE |
|
| PC1903 | Unpaid annual fee |
St.27 status event code: N-4-6-H10-H13-oth-PC1903 Ip right cessation event data comment text: Termination Category : DEFAULT_OF_REGISTRATION_FEE Not in force date: 20210331 |