KR20110008043A - 전착 방법 및 전착 장치 - Google Patents
전착 방법 및 전착 장치 Download PDFInfo
- Publication number
- KR20110008043A KR20110008043A KR1020107023293A KR20107023293A KR20110008043A KR 20110008043 A KR20110008043 A KR 20110008043A KR 1020107023293 A KR1020107023293 A KR 1020107023293A KR 20107023293 A KR20107023293 A KR 20107023293A KR 20110008043 A KR20110008043 A KR 20110008043A
- Authority
- KR
- South Korea
- Prior art keywords
- plating
- electrolyte
- cell
- electrodeposition
- current
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 238000000034 method Methods 0.000 title claims abstract description 47
- 238000004070 electrodeposition Methods 0.000 title claims description 68
- 238000009713 electroplating Methods 0.000 claims abstract description 44
- 239000008151 electrolyte solution Substances 0.000 claims abstract description 20
- 238000007747 plating Methods 0.000 claims description 280
- 239000003792 electrolyte Substances 0.000 claims description 147
- 239000000758 substrate Substances 0.000 claims description 58
- 239000007769 metal material Substances 0.000 claims description 49
- 239000002245 particle Substances 0.000 claims description 38
- 230000002441 reversible effect Effects 0.000 claims description 33
- 229910052751 metal Inorganic materials 0.000 claims description 32
- 239000002184 metal Substances 0.000 claims description 32
- 229910052759 nickel Inorganic materials 0.000 claims description 17
- 230000001360 synchronised effect Effects 0.000 claims description 17
- 150000002500 ions Chemical class 0.000 claims description 13
- 238000013019 agitation Methods 0.000 claims description 12
- 229910045601 alloy Inorganic materials 0.000 claims description 9
- 239000000956 alloy Substances 0.000 claims description 9
- 229910052799 carbon Inorganic materials 0.000 claims description 8
- 229910052804 chromium Inorganic materials 0.000 claims description 8
- 229910052802 copper Inorganic materials 0.000 claims description 8
- 229910052697 platinum Inorganic materials 0.000 claims description 7
- 229910052718 tin Inorganic materials 0.000 claims description 7
- 229910052721 tungsten Inorganic materials 0.000 claims description 7
- 229910052725 zinc Inorganic materials 0.000 claims description 7
- 229910052796 boron Inorganic materials 0.000 claims description 6
- 229910052742 iron Inorganic materials 0.000 claims description 6
- 229910052709 silver Inorganic materials 0.000 claims description 6
- 229910052737 gold Inorganic materials 0.000 claims description 5
- 229910052750 molybdenum Inorganic materials 0.000 claims description 5
- 229910052763 palladium Inorganic materials 0.000 claims description 5
- 229910052703 rhodium Inorganic materials 0.000 claims description 5
- 229910052707 ruthenium Inorganic materials 0.000 claims description 5
- WYTGDNHDOZPMIW-RCBQFDQVSA-N alstonine Natural products C1=CC2=C3C=CC=CC3=NC2=C2N1C[C@H]1[C@H](C)OC=C(C(=O)OC)[C@H]1C2 WYTGDNHDOZPMIW-RCBQFDQVSA-N 0.000 claims description 4
- 229910052745 lead Inorganic materials 0.000 claims description 4
- 229910052698 phosphorus Inorganic materials 0.000 claims description 4
- 229910052717 sulfur Inorganic materials 0.000 claims description 4
- 239000007864 aqueous solution Substances 0.000 claims description 2
- 230000001413 cellular effect Effects 0.000 claims description 2
- 230000000399 orthopedic effect Effects 0.000 claims description 2
- 229910052760 oxygen Inorganic materials 0.000 claims description 2
- 238000000576 coating method Methods 0.000 abstract description 81
- 239000011248 coating agent Substances 0.000 abstract description 71
- 210000004027 cell Anatomy 0.000 description 260
- 238000000151 deposition Methods 0.000 description 49
- PXHVJJICTQNCMI-UHFFFAOYSA-N nickel Substances [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 49
- 239000010410 layer Substances 0.000 description 48
- 230000008021 deposition Effects 0.000 description 47
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 38
- 229910002804 graphite Inorganic materials 0.000 description 29
- 239000010439 graphite Substances 0.000 description 29
- 239000004593 Epoxy Substances 0.000 description 25
- 230000010287 polarization Effects 0.000 description 13
- 238000002474 experimental method Methods 0.000 description 12
- 239000000463 material Substances 0.000 description 12
- 239000011156 metal matrix composite Substances 0.000 description 12
- 230000008569 process Effects 0.000 description 11
- 230000000694 effects Effects 0.000 description 10
- 239000011133 lead Substances 0.000 description 9
- 230000015572 biosynthetic process Effects 0.000 description 8
- 239000002131 composite material Substances 0.000 description 8
- 238000005260 corrosion Methods 0.000 description 8
- 230000007797 corrosion Effects 0.000 description 8
- 238000009826 distribution Methods 0.000 description 8
- 238000003786 synthesis reaction Methods 0.000 description 8
- 235000019589 hardness Nutrition 0.000 description 7
- 238000004519 manufacturing process Methods 0.000 description 7
- -1 polypropylene Polymers 0.000 description 7
- 230000008859 change Effects 0.000 description 6
- 239000011651 chromium Substances 0.000 description 6
- 238000013461 design Methods 0.000 description 6
- 239000000835 fiber Substances 0.000 description 6
- XEEYBQQBJWHFJM-UHFFFAOYSA-N iron Substances [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 6
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 6
- 239000010936 titanium Substances 0.000 description 6
- 150000001875 compounds Chemical class 0.000 description 5
- 229910003460 diamond Inorganic materials 0.000 description 5
- 239000010432 diamond Substances 0.000 description 5
- 238000005259 measurement Methods 0.000 description 5
- 150000002739 metals Chemical class 0.000 description 5
- 239000000843 powder Substances 0.000 description 5
- 230000002829 reductive effect Effects 0.000 description 5
- 230000004044 response Effects 0.000 description 5
- 238000007873 sieving Methods 0.000 description 5
- 239000011135 tin Substances 0.000 description 5
- 241000288673 Chiroptera Species 0.000 description 4
- 239000004952 Polyamide Substances 0.000 description 4
- 239000004743 Polypropylene Substances 0.000 description 4
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 4
- 239000000654 additive Substances 0.000 description 4
- 229910052782 aluminium Inorganic materials 0.000 description 4
- VTYYLEPIZMXCLO-UHFFFAOYSA-L calcium carbonate Substances [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 4
- 230000007547 defect Effects 0.000 description 4
- 229920001971 elastomer Polymers 0.000 description 4
- 239000003822 epoxy resin Substances 0.000 description 4
- 230000006870 function Effects 0.000 description 4
- 229910001092 metal group alloy Inorganic materials 0.000 description 4
- 229920002647 polyamide Polymers 0.000 description 4
- 229920000647 polyepoxide Polymers 0.000 description 4
- 229920001155 polypropylene Polymers 0.000 description 4
- 239000005060 rubber Substances 0.000 description 4
- 239000010935 stainless steel Substances 0.000 description 4
- 229910001220 stainless steel Inorganic materials 0.000 description 4
- 238000003756 stirring Methods 0.000 description 4
- 239000000126 substance Substances 0.000 description 4
- 229910052719 titanium Inorganic materials 0.000 description 4
- 238000005299 abrasion Methods 0.000 description 3
- XECAHXYUAAWDEL-UHFFFAOYSA-N acrylonitrile butadiene styrene Chemical compound C=CC=C.C=CC#N.C=CC1=CC=CC=C1 XECAHXYUAAWDEL-UHFFFAOYSA-N 0.000 description 3
- 239000004676 acrylonitrile butadiene styrene Substances 0.000 description 3
- 229920000122 acrylonitrile butadiene styrene Polymers 0.000 description 3
- 230000000996 additive effect Effects 0.000 description 3
- 230000008901 benefit Effects 0.000 description 3
- 229910021393 carbon nanotube Inorganic materials 0.000 description 3
- 239000002041 carbon nanotube Substances 0.000 description 3
- 238000006243 chemical reaction Methods 0.000 description 3
- 239000013078 crystal Substances 0.000 description 3
- 230000007423 decrease Effects 0.000 description 3
- 239000010419 fine particle Substances 0.000 description 3
- 239000012535 impurity Substances 0.000 description 3
- 229910052500 inorganic mineral Inorganic materials 0.000 description 3
- 238000003780 insertion Methods 0.000 description 3
- 230000037431 insertion Effects 0.000 description 3
- 238000001465 metallisation Methods 0.000 description 3
- 239000011707 mineral Substances 0.000 description 3
- 235000010755 mineral Nutrition 0.000 description 3
- 239000000203 mixture Substances 0.000 description 3
- 239000002055 nanoplate Substances 0.000 description 3
- KDLHZDBZIXYQEI-UHFFFAOYSA-N palladium Substances [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 3
- 230000000704 physical effect Effects 0.000 description 3
- 229920001343 polytetrafluoroethylene Polymers 0.000 description 3
- 239000004810 polytetrafluoroethylene Substances 0.000 description 3
- 239000010948 rhodium Substances 0.000 description 3
- 229910052710 silicon Inorganic materials 0.000 description 3
- 239000000243 solution Substances 0.000 description 3
- 230000007704 transition Effects 0.000 description 3
- 229920000049 Carbon (fiber) Polymers 0.000 description 2
- 229920002160 Celluloid Polymers 0.000 description 2
- VYZAMTAEIAYCRO-UHFFFAOYSA-N Chromium Chemical compound [Cr] VYZAMTAEIAYCRO-UHFFFAOYSA-N 0.000 description 2
- 229910001030 Iron–nickel alloy Inorganic materials 0.000 description 2
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 2
- 229910000831 Steel Inorganic materials 0.000 description 2
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 2
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 2
- 239000010405 anode material Substances 0.000 description 2
- 238000013459 approach Methods 0.000 description 2
- 239000000440 bentonite Substances 0.000 description 2
- 229910000278 bentonite Inorganic materials 0.000 description 2
- SVPXDRXYRYOSEX-UHFFFAOYSA-N bentoquatam Chemical compound O.O=[Si]=O.O=[Al]O[Al]=O SVPXDRXYRYOSEX-UHFFFAOYSA-N 0.000 description 2
- 235000010216 calcium carbonate Nutrition 0.000 description 2
- 235000012241 calcium silicate Nutrition 0.000 description 2
- 238000004140 cleaning Methods 0.000 description 2
- 239000011247 coating layer Substances 0.000 description 2
- 238000005336 cracking Methods 0.000 description 2
- 230000003247 decreasing effect Effects 0.000 description 2
- 238000009429 electrical wiring Methods 0.000 description 2
- 239000011888 foil Substances 0.000 description 2
- 239000011361 granulated particle Substances 0.000 description 2
- LNEPOXFFQSENCJ-UHFFFAOYSA-N haloperidol Chemical compound C1CC(O)(C=2C=CC(Cl)=CC=2)CCN1CCCC(=O)C1=CC=C(F)C=C1 LNEPOXFFQSENCJ-UHFFFAOYSA-N 0.000 description 2
- 229910052738 indium Inorganic materials 0.000 description 2
- 239000007788 liquid Substances 0.000 description 2
- 229910052749 magnesium Inorganic materials 0.000 description 2
- 239000011159 matrix material Substances 0.000 description 2
- 229910021645 metal ion Inorganic materials 0.000 description 2
- 239000011859 microparticle Substances 0.000 description 2
- 239000012764 mineral filler Substances 0.000 description 2
- 239000003973 paint Substances 0.000 description 2
- 238000010422 painting Methods 0.000 description 2
- 238000005240 physical vapour deposition Methods 0.000 description 2
- 238000005498 polishing Methods 0.000 description 2
- 229920006122 polyamide resin Polymers 0.000 description 2
- 229920000642 polymer Polymers 0.000 description 2
- 229920000307 polymer substrate Polymers 0.000 description 2
- 239000004800 polyvinyl chloride Substances 0.000 description 2
- 239000008262 pumice Substances 0.000 description 2
- 230000009467 reduction Effects 0.000 description 2
- 150000004760 silicates Chemical class 0.000 description 2
- 238000004513 sizing Methods 0.000 description 2
- 125000006850 spacer group Chemical group 0.000 description 2
- 239000010959 steel Substances 0.000 description 2
- 229920002397 thermoplastic olefin Polymers 0.000 description 2
- 239000004408 titanium dioxide Substances 0.000 description 2
- 229910052720 vanadium Inorganic materials 0.000 description 2
- 229910000859 α-Fe Inorganic materials 0.000 description 2
- XMWRBQBLMFGWIX-UHFFFAOYSA-N C60 fullerene Chemical compound C12=C3C(C4=C56)=C7C8=C5C5=C9C%10=C6C6=C4C1=C1C4=C6C6=C%10C%10=C9C9=C%11C5=C8C5=C8C7=C3C3=C7C2=C1C1=C2C4=C6C4=C%10C6=C9C9=C%11C5=C5C8=C3C3=C7C1=C1C2=C4C6=C2C9=C5C3=C12 XMWRBQBLMFGWIX-UHFFFAOYSA-N 0.000 description 1
- 206010008531 Chills Diseases 0.000 description 1
- 229910000640 Fe alloy Inorganic materials 0.000 description 1
- 229910001374 Invar Inorganic materials 0.000 description 1
- 229910003271 Ni-Fe Inorganic materials 0.000 description 1
- 239000004698 Polyethylene Substances 0.000 description 1
- 235000017848 Rubus fruticosus Nutrition 0.000 description 1
- 244000078534 Vaccinium myrtillus Species 0.000 description 1
- IUHFWCGCSVTMPG-UHFFFAOYSA-N [C].[C] Chemical compound [C].[C] IUHFWCGCSVTMPG-UHFFFAOYSA-N 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 239000000853 adhesive Substances 0.000 description 1
- 230000001070 adhesive effect Effects 0.000 description 1
- 238000002048 anodisation reaction Methods 0.000 description 1
- 235000021029 blackberry Nutrition 0.000 description 1
- 230000000903 blocking effect Effects 0.000 description 1
- 229910000019 calcium carbonate Inorganic materials 0.000 description 1
- 229910052918 calcium silicate Inorganic materials 0.000 description 1
- 239000000378 calcium silicate Substances 0.000 description 1
- OYACROKNLOSFPA-UHFFFAOYSA-N calcium;dioxido(oxo)silane Chemical compound [Ca+2].[O-][Si]([O-])=O OYACROKNLOSFPA-UHFFFAOYSA-N 0.000 description 1
- 239000004917 carbon fiber Substances 0.000 description 1
- 239000003575 carbonaceous material Substances 0.000 description 1
- 239000000919 ceramic Substances 0.000 description 1
- 239000000805 composite resin Substances 0.000 description 1
- 230000001010 compromised effect Effects 0.000 description 1
- 239000004020 conductor Substances 0.000 description 1
- 238000010924 continuous production Methods 0.000 description 1
- 238000005520 cutting process Methods 0.000 description 1
- 210000001787 dendrite Anatomy 0.000 description 1
- 238000010586 diagram Methods 0.000 description 1
- 238000004090 dissolution Methods 0.000 description 1
- 239000000428 dust Substances 0.000 description 1
- 238000003487 electrochemical reaction Methods 0.000 description 1
- 239000002659 electrodeposit Substances 0.000 description 1
- 238000005323 electroforming Methods 0.000 description 1
- 238000005538 encapsulation Methods 0.000 description 1
- 239000000945 filler Substances 0.000 description 1
- 239000010408 film Substances 0.000 description 1
- 238000009501 film coating Methods 0.000 description 1
- 238000001914 filtration Methods 0.000 description 1
- 238000007667 floating Methods 0.000 description 1
- 239000000446 fuel Substances 0.000 description 1
- 239000003365 glass fiber Substances 0.000 description 1
- 230000005484 gravity Effects 0.000 description 1
- 238000000227 grinding Methods 0.000 description 1
- 230000001771 impaired effect Effects 0.000 description 1
- 239000007943 implant Substances 0.000 description 1
- 238000002347 injection Methods 0.000 description 1
- 239000007924 injection Substances 0.000 description 1
- 239000010416 ion conductor Substances 0.000 description 1
- 238000007733 ion plating Methods 0.000 description 1
- UGKDIUIOSMUOAW-UHFFFAOYSA-N iron nickel Chemical compound [Fe].[Ni] UGKDIUIOSMUOAW-UHFFFAOYSA-N 0.000 description 1
- 231100000518 lethal Toxicity 0.000 description 1
- 230000001665 lethal effect Effects 0.000 description 1
- 238000011068 loading method Methods 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- 239000012528 membrane Substances 0.000 description 1
- 150000001247 metal acetylides Chemical class 0.000 description 1
- 229910044991 metal oxide Inorganic materials 0.000 description 1
- 150000004706 metal oxides Chemical class 0.000 description 1
- 239000005300 metallic glass Substances 0.000 description 1
- VNWKTOKETHGBQD-UHFFFAOYSA-N methane Chemical compound C VNWKTOKETHGBQD-UHFFFAOYSA-N 0.000 description 1
- 239000010445 mica Substances 0.000 description 1
- 229910052618 mica group Inorganic materials 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 238000000465 moulding Methods 0.000 description 1
- 239000002159 nanocrystal Substances 0.000 description 1
- 150000004767 nitrides Chemical class 0.000 description 1
- 239000011255 nonaqueous electrolyte Substances 0.000 description 1
- 230000001590 oxidative effect Effects 0.000 description 1
- 239000011236 particulate material Substances 0.000 description 1
- 230000000737 periodic effect Effects 0.000 description 1
- 229910000889 permalloy Inorganic materials 0.000 description 1
- 229920003023 plastic Polymers 0.000 description 1
- 239000004033 plastic Substances 0.000 description 1
- 239000004417 polycarbonate Substances 0.000 description 1
- 229920000515 polycarbonate Polymers 0.000 description 1
- 229920001690 polydopamine Polymers 0.000 description 1
- 229920000573 polyethylene Polymers 0.000 description 1
- 239000002861 polymer material Substances 0.000 description 1
- 229920000915 polyvinyl chloride Polymers 0.000 description 1
- 238000002360 preparation method Methods 0.000 description 1
- 239000000376 reactant Substances 0.000 description 1
- 230000010076 replication Effects 0.000 description 1
- 230000000284 resting effect Effects 0.000 description 1
- 230000000630 rising effect Effects 0.000 description 1
- 238000007650 screen-printing Methods 0.000 description 1
- 230000035939 shock Effects 0.000 description 1
- 239000000377 silicon dioxide Substances 0.000 description 1
- 241000894007 species Species 0.000 description 1
- 230000003068 static effect Effects 0.000 description 1
- 230000002194 synthesizing effect Effects 0.000 description 1
- 235000012222 talc Nutrition 0.000 description 1
- 239000010409 thin film Substances 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 1
- 238000004018 waxing Methods 0.000 description 1
- 238000004260 weight control Methods 0.000 description 1
- 229910052726 zirconium Inorganic materials 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C25—ELECTROLYTIC OR ELECTROPHORETIC PROCESSES; APPARATUS THEREFOR
- C25D—PROCESSES FOR THE ELECTROLYTIC OR ELECTROPHORETIC PRODUCTION OF COATINGS; ELECTROFORMING; APPARATUS THEREFOR
- C25D5/00—Electroplating characterised by the process; Pretreatment or after-treatment of workpieces
- C25D5/18—Electroplating using modulated, pulsed or reversing current
-
- C—CHEMISTRY; METALLURGY
- C25—ELECTROLYTIC OR ELECTROPHORETIC PROCESSES; APPARATUS THEREFOR
- C25D—PROCESSES FOR THE ELECTROLYTIC OR ELECTROPHORETIC PRODUCTION OF COATINGS; ELECTROFORMING; APPARATUS THEREFOR
- C25D17/00—Constructional parts, or assemblies thereof, of cells for electrolytic coating
-
- C—CHEMISTRY; METALLURGY
- C25—ELECTROLYTIC OR ELECTROPHORETIC PROCESSES; APPARATUS THEREFOR
- C25D—PROCESSES FOR THE ELECTROLYTIC OR ELECTROPHORETIC PRODUCTION OF COATINGS; ELECTROFORMING; APPARATUS THEREFOR
- C25D17/00—Constructional parts, or assemblies thereof, of cells for electrolytic coating
- C25D17/007—Current directing devices
-
- C—CHEMISTRY; METALLURGY
- C25—ELECTROLYTIC OR ELECTROPHORETIC PROCESSES; APPARATUS THEREFOR
- C25D—PROCESSES FOR THE ELECTROLYTIC OR ELECTROPHORETIC PRODUCTION OF COATINGS; ELECTROFORMING; APPARATUS THEREFOR
- C25D21/00—Processes for servicing or operating cells for electrolytic coating
- C25D21/12—Process control or regulation
- C25D21/14—Controlled addition of electrolyte components
-
- C—CHEMISTRY; METALLURGY
- C25—ELECTROLYTIC OR ELECTROPHORETIC PROCESSES; APPARATUS THEREFOR
- C25D—PROCESSES FOR THE ELECTROLYTIC OR ELECTROPHORETIC PRODUCTION OF COATINGS; ELECTROFORMING; APPARATUS THEREFOR
- C25D5/00—Electroplating characterised by the process; Pretreatment or after-treatment of workpieces
- C25D5/003—Electroplating using gases, e.g. pressure influence
-
- C—CHEMISTRY; METALLURGY
- C25—ELECTROLYTIC OR ELECTROPHORETIC PROCESSES; APPARATUS THEREFOR
- C25D—PROCESSES FOR THE ELECTROLYTIC OR ELECTROPHORETIC PRODUCTION OF COATINGS; ELECTROFORMING; APPARATUS THEREFOR
- C25D5/00—Electroplating characterised by the process; Pretreatment or after-treatment of workpieces
- C25D5/60—Electroplating characterised by the structure or texture of the layers
- C25D5/615—Microstructure of the layers, e.g. mixed structure
- C25D5/617—Crystalline layers
-
- C—CHEMISTRY; METALLURGY
- C25—ELECTROLYTIC OR ELECTROPHORETIC PROCESSES; APPARATUS THEREFOR
- C25D—PROCESSES FOR THE ELECTROLYTIC OR ELECTROPHORETIC PRODUCTION OF COATINGS; ELECTROFORMING; APPARATUS THEREFOR
- C25D5/00—Electroplating characterised by the process; Pretreatment or after-treatment of workpieces
- C25D5/60—Electroplating characterised by the structure or texture of the layers
- C25D5/615—Microstructure of the layers, e.g. mixed structure
- C25D5/619—Amorphous layers
-
- C—CHEMISTRY; METALLURGY
- C25—ELECTROLYTIC OR ELECTROPHORETIC PROCESSES; APPARATUS THEREFOR
- C25D—PROCESSES FOR THE ELECTROLYTIC OR ELECTROPHORETIC PRODUCTION OF COATINGS; ELECTROFORMING; APPARATUS THEREFOR
- C25D17/00—Constructional parts, or assemblies thereof, of cells for electrolytic coating
- C25D17/001—Apparatus specially adapted for electrolytic coating of wafers, e.g. semiconductors or solar cells
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Electrochemistry (AREA)
- Materials Engineering (AREA)
- Metallurgy (AREA)
- Organic Chemistry (AREA)
- Crystallography & Structural Chemistry (AREA)
- Automation & Control Theory (AREA)
- Electroplating Methods And Accessories (AREA)
Abstract
Description
도 1a는 도 1의 2개의 인접 셀의 확대도면이다.
도 2는 18개의 셀 멀티-셀 컴파트먼트, 즉, 도 1의 컴파트먼트 B1의 18개의 파트를 동시에 도금하는 전기 배선 개략도를 도시하고 있는데, 각 스트링은 직렬 구성으로 3개의 파트를 포함한다.
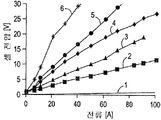
도 3은 도금 셀의 다수의 워크피스(workpieces)에 대한 전압-전류 프로파일을 도시하고 있다.
도 4는 DC 도금을 위한 도금 셀의 다양한 코딩 레벨에서의 워크피스에 대한 전압-전류 프로파일을 도시하고 있다.
도 5는 펄스 전착을 위한 다양한 코팅 레벨에서의 워크피스에 대한 전압-전류 프로파일을 도시하고 있다.
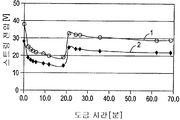
도 6은 3-파트 및 4-파트 직렬 도금 스트링에 대한 전압-시간 프로파일을 도시하고 있다.
도 7은 3-단계 도금 프로파일을 사용하는 흑연/에폭시 튜브의 6개의 3-파트 스트링에 대한 전압-시간 프로파일을 도시하고 있다.
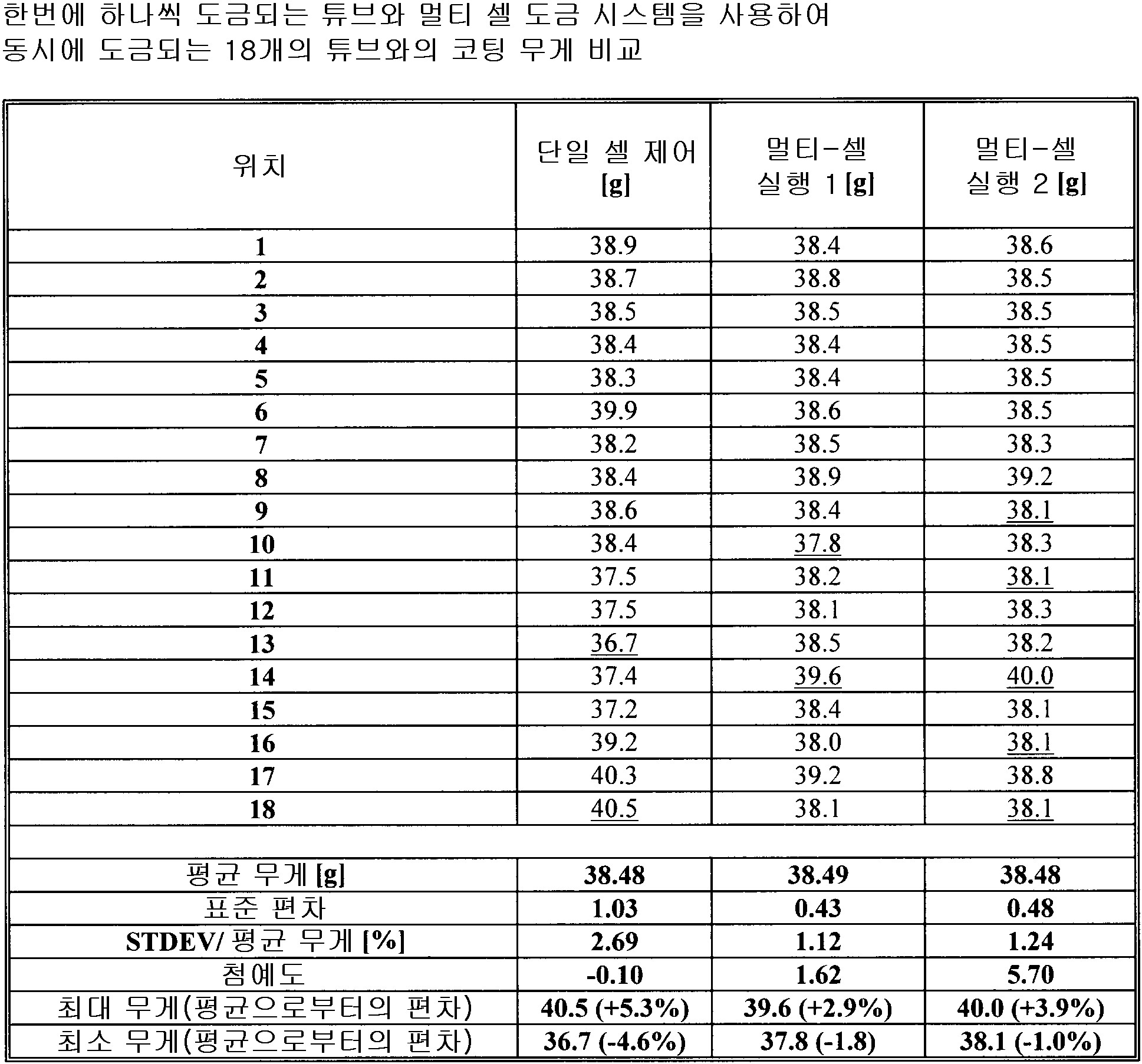
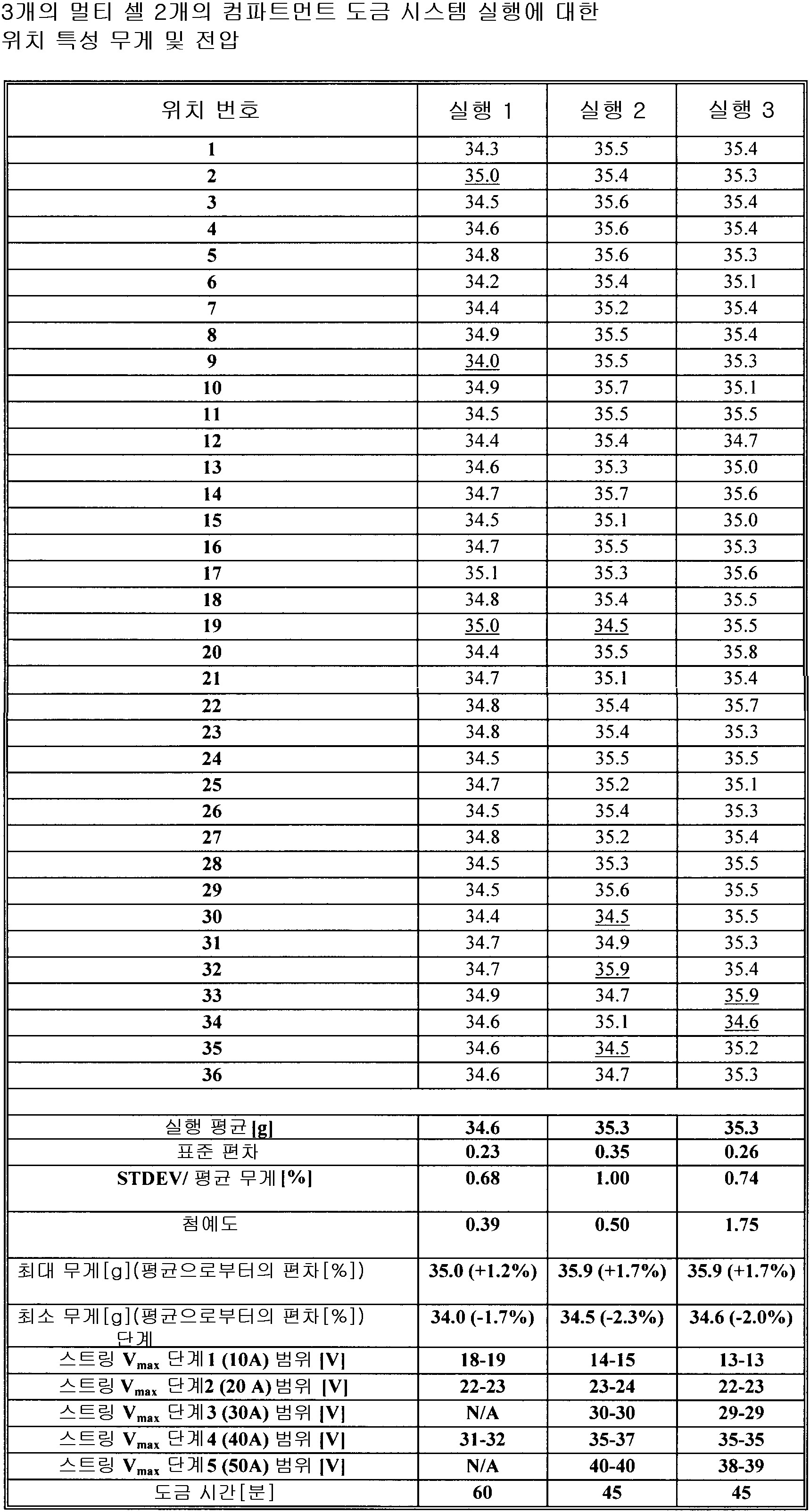
도 8은 단일 셀에서 도금되는 파트 및 멀티-도금 시스템에서 도금되는 파트에 대한 코팅 두께 프로파일을 도시하고 있는데, 단일 셀 탱크 및 멀티-셀 탱크 두께 프로파일 비교를 제공한다.
Claims (12)
- 적어도 2개의 영구 또는 임시 기판 각각에 금속 물질층을 동시에 전착하는(electrodepositing) 방법으로서,
(a) 복수의 이온적으로(ionically) 서로 통하는 전착 영역(electrodepositing zones))을 직렬로 전기 접속시키는 단계와.
(b) 단일 소스로부터 상기 이온적으로 서로 통하는 전착 영역 중 적어도 2개로 전기 전력을 직렬로 공급하는 단계와,
(c) 상기 이온적으로 서로 통하는 전착 영역 사이에서 공유되는 수용액 전해액에 상기 적어도 2개의 기판의 각 기판을 침수시키는 단계와,
(d) 각 기판에 음전하를 공급하고 각 기판에 동일한 전류를 제공하는 단계를 포함하는
전착 방법.
- 제 1 항에 있어서,
복수의 도금된 파트를 동시에 준비하되, 각 파트는 적어도 일부에 전착된 금속 물질층을 포함하고,
각 전착 영역은 적어도 하나의 캐소드 영역을 구비하고, 내부의 기판은 음극성(cathodic)이되고,
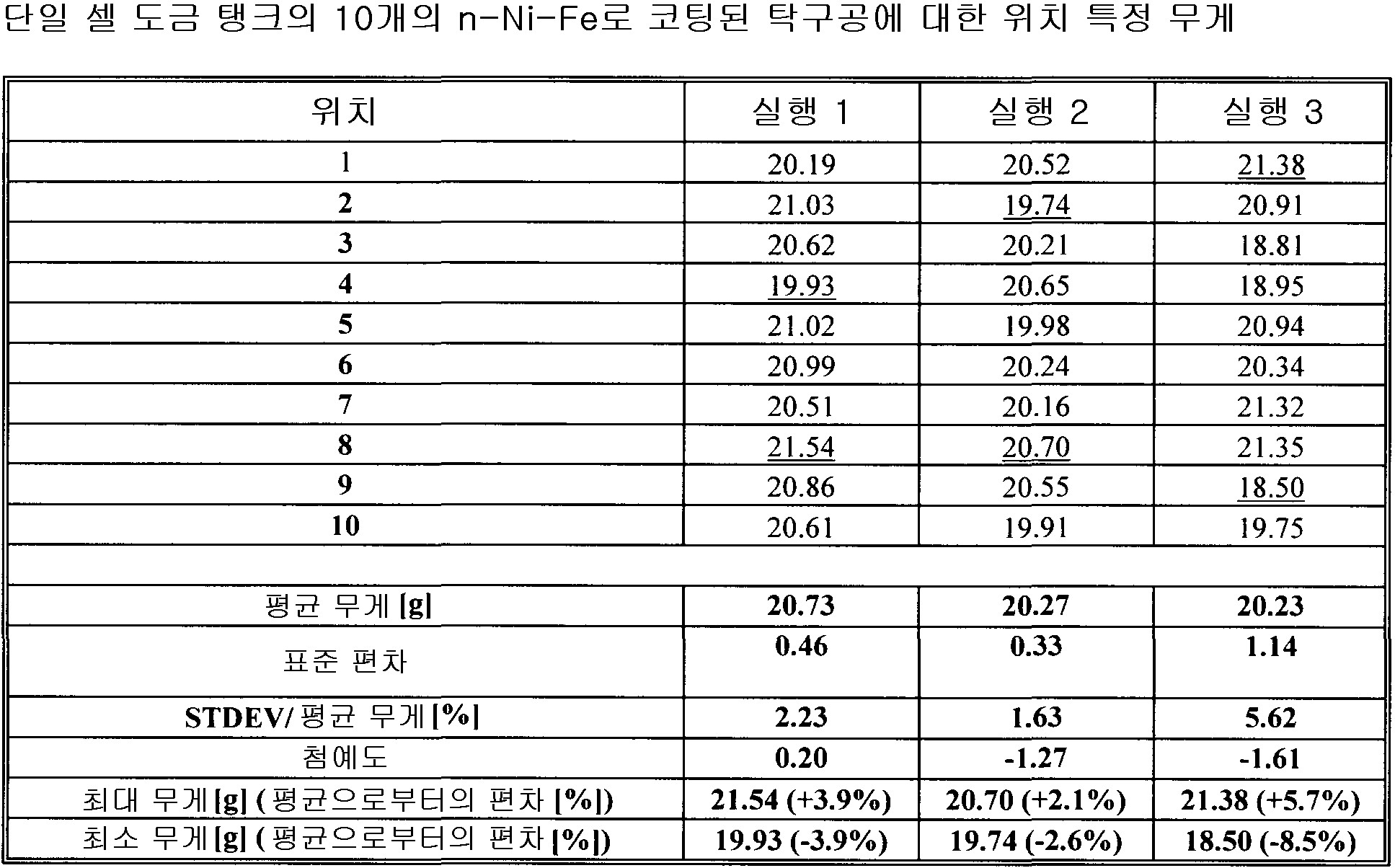
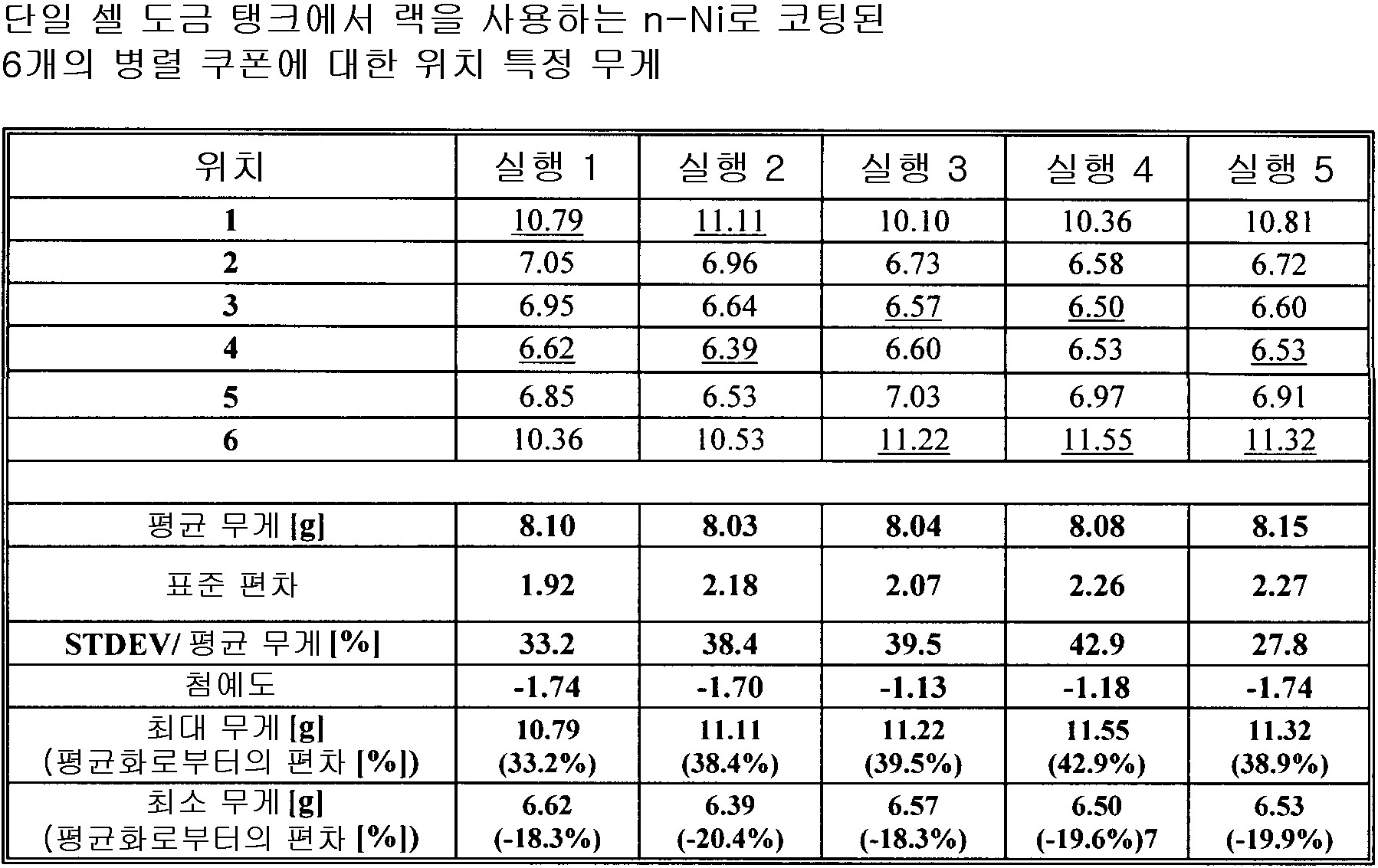
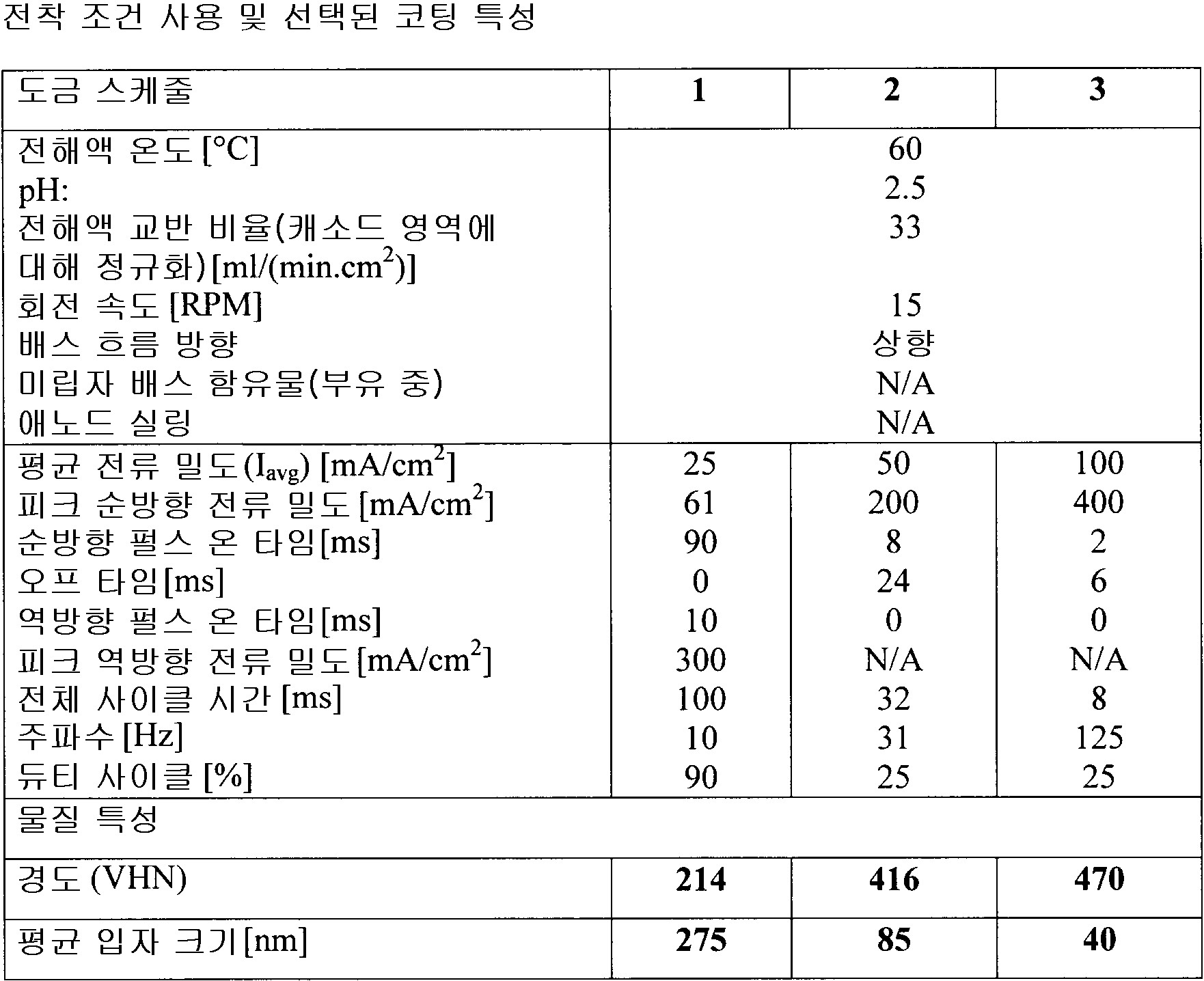
각 전착 영역의 전착 파라미터는, 5 내지 10,000mA/cm2 범위의 평균 전류 밀도와, 0.1 내지 10,000ms 범위의 순방향 펄스 온 타임과, 0 내지 10,000ms 범위의 펄스 오프와, 0 내지 500ms 범위의 역방향 펄스 온 타임과, 5 내지 10,000 mA/cm2 범위의 피크 순방향 전류 밀도와, 5 내지 20,000mA/cm2 범위의 피크 역방향 전류 밀도와, 0 내지 1,000Hz 범위의 주파수와, 5 내지 100% 범위의 듀티 사이클과, 0 내지 100℃ 범위의 전해액 온도이며, 동작 전극은 상기 기판이거나 애노드 영역을 구성하고, 0 내지 1,000 RPM 범위의 회전 속도와, 0 내지 12 범위의 전해액 pH와, 1 내지 6,000ml/(min·cm2) 범위의 전해액 교반 속도(electrolyte agitation rate)을 가지며, 상기 애노드 영역에서 기하구조적 애노드 표면적의 0-95% 사이를 덮으며, 전해액 전기화학적으로 불활성 미립자 함유물은 체적 0 내지 70% 범위이며, 얻어진 상기 동시에 도금되는 파트의 파트 대 파트 변동은 ±20% 미만의 평균 층 무게 대 최대 층 무게 비율 및 ±20% 미만의 평균 층 무게 대 층 무게 표준 편차 비율 및 4개 이상의 기판의 경우에는 10 미만의 첨예도(kurtosis)에 의해 나타나는
전착 방법.
- 제 2 항에 있어서,
적어도 4개의 물품은 2개의 직렬 스트링에서 동시에 전착되고 각 스트링은 상이한 전력원에 의해 전력이 공급되며,
상기 전력원은 동기화되어 전기도금 영역으로부터 전착 영역으로의 전압 변동을 최소화시키는
전착 방법.
- 제 3 항에 있어서,
상기 전착 파라미터는 각 전착된 금속 물질층이 20 마이크론 내지 5cm 범위의 두께를 갖도록 선택되고,
얻어진 파트 대 파트 변동성은 ±20% 미만의 평균 층 두께 대 최대 층 두께의 비율 및 ±20% 미만의 평균 층 두께 대 층 두게 표준 편차의 비율 및 4개 이상의 기판의 경우에 10 미만의 첨예도에 의해 나타나는
전착 방법.
- 제 2 항에 있어서,
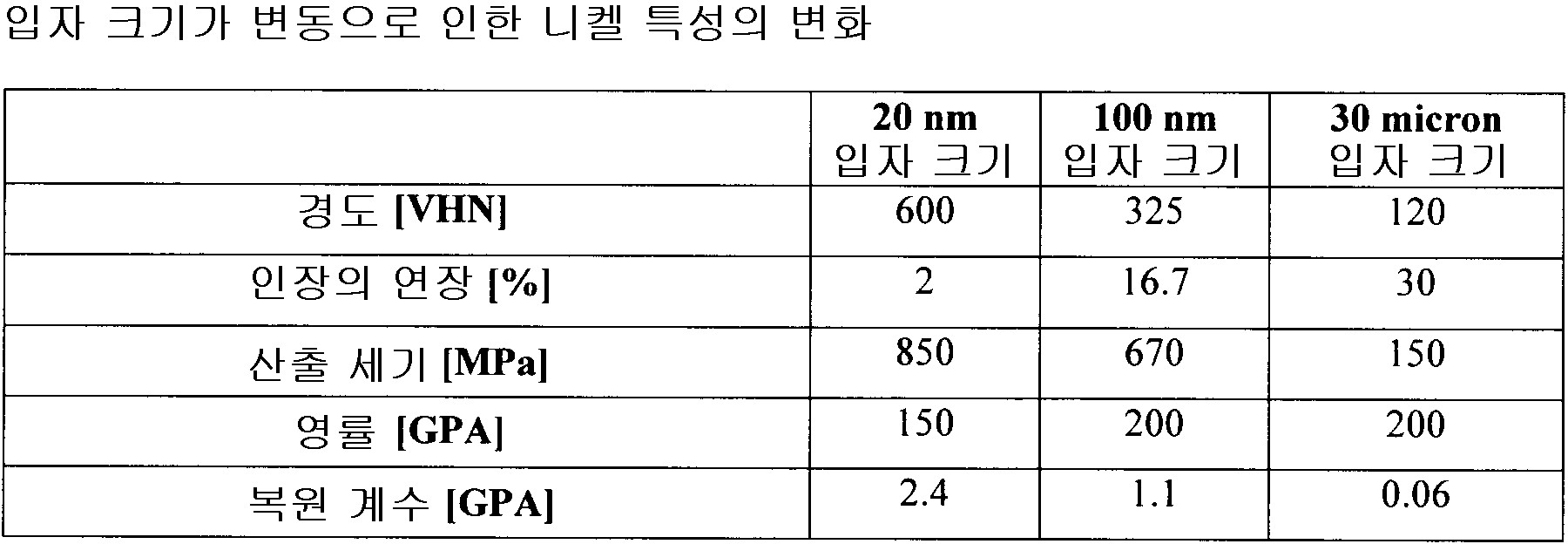
상기 전착 파라미터는 상기 전착된 금속 물질층이 2nm 내지 5,000nm 범위의 평균 입자 크기, 5,000nm 초과 평균 입자 크기를 갖는 조립 마이크로구조 및 무정형 마이크로구조로 구성되는 그룹으로부터 선택되는 동일한 마이크로 구조를 갖도록 선택되는
전착 방법. - 제 2 항에 있어서,
상기 전착 파라미터는 모든 상기 증착된 금속 물질층이 동일한 등급의 입자 크기를 갖도록 선택되는
전착 방법.
- 제 2 항에 있어서,
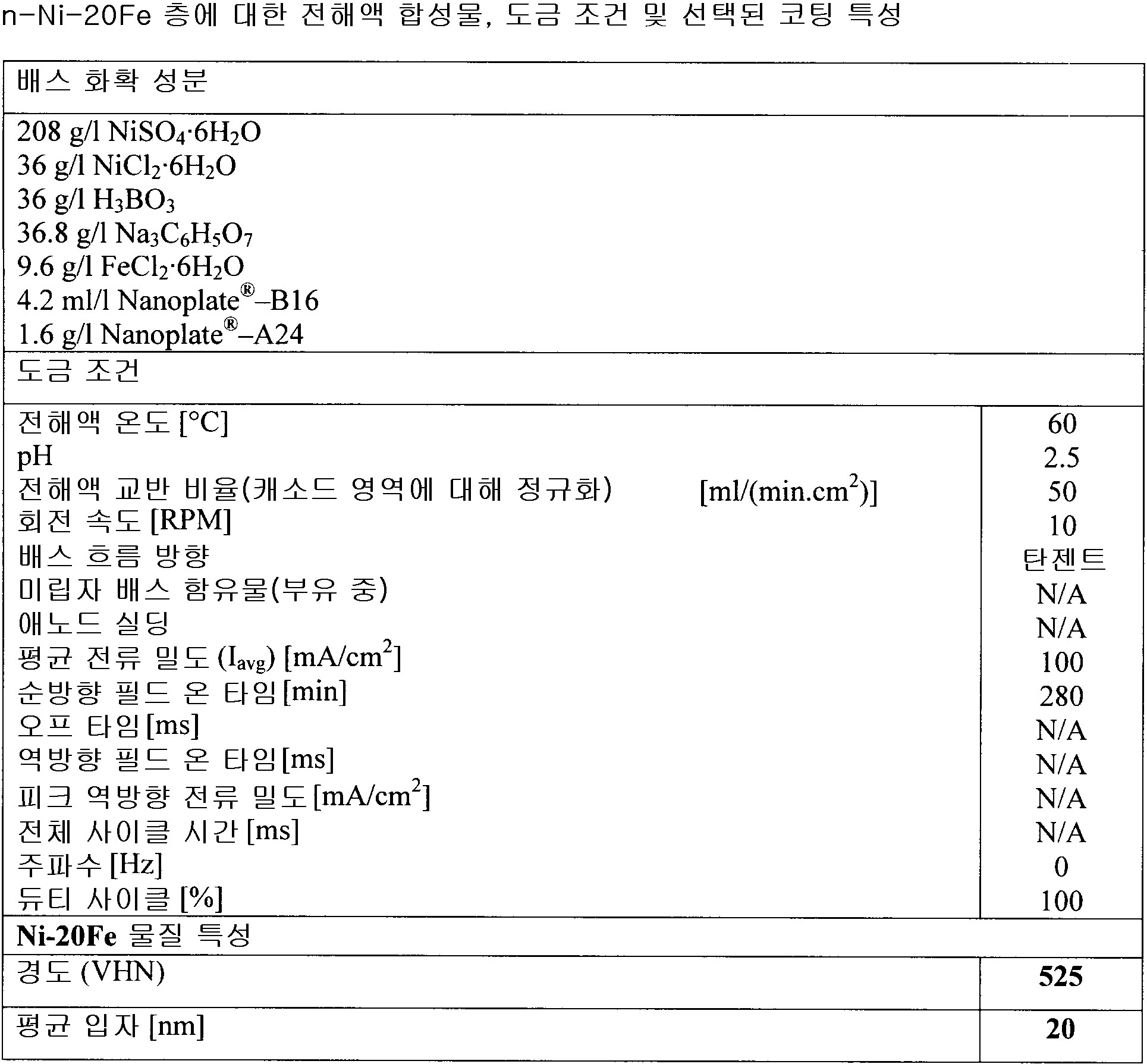
상기 금속 물질은 Ag, Au, Cu, Co, Cr, Mo, Ni, Sn, Fe, Pd, Pb, Pt, Rh, Ru 및 Zn으로 구성되는 그룹으로부터 선택되는 하나 이상의 원소의 금속 또는 합금이며, 선택적으로 하나 이상의 원소는 B, P, C, S 및 W로 구성되는 그룹으로부터 선택되는
전착 방법.
- 제 2 항에 있어서,
상기 금속 물질은
a. Ag, Au, Cu, Co, Cr, Mo, Ni, Sn, Fe, Pd, Pb, Pt, Rh, Ru 및 Zn로 구성되는 그룹으로부터 선택되는 하나 이상의 금속
b. C, O 및 S로 구성되는 그룹으로부터 선택되는 적어도 하나의 원소 및
c. 선택적으로 B, P 및 W로 구성되는 그룹으로부터 선택되는 적어도 하나의 원소
를 포함하는
전착 방법.
- 제 2 항에 있어서,
전착은 교정용 보철(an orthopedic prosthesis), 총신(gun barrel), 몰드(mold), 스포츠 제품, 셀룰러 전화 또는 자동차 부품의 기판으로 수행되는
전착 방법.
- 제 2 항에 있어서,
전착은 총신 내부로 수행되는
전착 방법.
- 직렬 전기 접속의 적어도 2개의 기판의 표면상으로 금속 물질을 동시에 전착하는 장치로서,
(a) 증착될 상기 금속 물질의 이온을 함유하는 전해액 용액으로 채워진 전해액 우물과,
(b) 직렬로 전기 접속되는 적어도 2개의 도금 셀과,
(c) 상기 우물로부터 각 도금 셀에 상기 전해액 용액을 공급하고 상기 전해액 용액을 상기 중앙 전해액 우물로 귀환시키는 전해액 순환 루프와,
(d) 각 도금 셀은
(i) 적어도 하나의 애노드와,
(ii) 시빙 전극(a thieving electrode)에 대해 선택적으로 위치되는 도금될 임시 또는 영구 기판 중 하나를 수용하고 유지할 수 있는 캐소드와,
(iii) 분배기 플레이트, 동기화 전력 공급기 및 셀들 사이의 구불구불한 전해액 경로로 구성되는 그룹으로부터 선택되는 도금 셀들 사이의 전압 차 및 분로 전류를 최소화시키는 수단을 포함하고,
(e) 적어도 2개의 도금 셀에 전기적으로 접속되는 적어도 하나의 전력원을 포함하는
전착 장치.
- 적어도 2개의 전력 공급기를 사용하며 직렬 전기 접속되는 적어도 4개의 기판의 표면으로 금속 물질을 동시에 전착하는 장치로서,
(a) 증착될 상기 금속 물질의 이온을 함유하는 전해액 용액으로 채워지는 전해액 우물과,
(b) 직렬로 전기 접속되는 적어도 2개의 도금 셀과,
(c) 각각 직렬로 전기접속되는 적어도 2개의 도금 셀의 적어도 2개의 스트링과,
(d) 상기 우물로부터 각 도금 셀로 상기 전해액 용액을 공급하고 상기 전해액 용액을 상기 전해액 우물로 귀환시키는 전해액 순환 루프와,
(e) 각각 상이한 도금 셀 스트링을 전기 접속시키는 적어도 2개의 전력 공급기 - 상기 전력 공급기는 도금 사이클 동안에 항상 전류 온 타임, 오프 타임, 역방향 타임 및 각 전류 밀도에 대해 동기화됨 - 와,
(f) 각 도금 셀은
(i) 적어도 하나의 애노드와,
(ii) 시빙 전극에 대해 선택적으로 위치되는 도금될 임시 또는 영구 기판 중 하나를 수용하고 유지할 수 있는 캐소드와,
(iii) 증착될 상기 금속 물질의 이온을 함유하는 전해액과,
(iv) 분배기 플레이트, 동기화 전력 공급기 및 셀들 사이의 구불구불한 전해액 경로로 구성되는 그룹으로부터 선택되는 도금 셀들 사이의 전압 차 및 분로 전류를 최소화시키는 수단을 포함하는
전착 장치.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US12/081,623 US8062496B2 (en) | 2008-04-18 | 2008-04-18 | Electroplating method and apparatus |
| US12/081,623 | 2008-04-18 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| KR20110008043A true KR20110008043A (ko) | 2011-01-25 |
Family
ID=41198720
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020107023293A Ceased KR20110008043A (ko) | 2008-04-18 | 2009-03-04 | 전착 방법 및 전착 장치 |
Country Status (8)
| Country | Link |
|---|---|
| US (2) | US8062496B2 (ko) |
| EP (1) | EP2262928A1 (ko) |
| KR (1) | KR20110008043A (ko) |
| CN (1) | CN102007232B (ko) |
| BR (1) | BRPI0910587A2 (ko) |
| CA (1) | CA2716394A1 (ko) |
| MX (1) | MX2010010658A (ko) |
| WO (1) | WO2009127037A1 (ko) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20150118080A (ko) * | 2012-07-27 | 2015-10-21 | 인스티튜토 슈페리어 테크니코 | 수지상 조직을 갖는 니켈-코발트 피막의 전착법 |
| KR20190036619A (ko) * | 2017-09-28 | 2019-04-05 | (주)포인텍 | 전기도금라인의 무선행거에 장착된 기판에 인가되는 전류의 제어방법 |
| KR102012731B1 (ko) * | 2018-12-06 | 2019-08-21 | 주식회사 에이엔씨코리아 | 6 가 크롬도금액 및 이를 이용한 크랙프리 펄스 전기도금방법 |
Families Citing this family (38)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2958300B1 (fr) * | 2010-03-31 | 2012-05-04 | Snecma | Dispositif pour controler des caracteristiques physiques d'un bain d'electrodeposition metallique. |
| AU2011288299B2 (en) * | 2010-08-11 | 2015-11-19 | Outotec Oyj | Apparatus for use in electrorefining and electrowinning |
| JP5469038B2 (ja) | 2010-11-12 | 2014-04-09 | 株式会社オティックス | 燃料系部品の製造方法および燃料系部品 |
| US8512541B2 (en) * | 2010-11-16 | 2013-08-20 | Trevor Pearson | Electrolytic dissolution of chromium from chromium electrodes |
| US9951436B2 (en) * | 2011-10-27 | 2018-04-24 | Garmor Inc. | Composite graphene structures |
| KR101506910B1 (ko) * | 2012-09-27 | 2015-03-30 | 티디케이가부시기가이샤 | 이방성 도금 방법 및 박막 코일 |
| JP6134396B2 (ja) | 2013-03-08 | 2017-05-24 | ガーマー インク.Garmor, Inc. | ホストにおけるグラフェン同伴 |
| CA2903987C (en) | 2013-03-08 | 2018-05-01 | University Of Central Florida Research Foundation, Inc. | Large scale oxidized graphene production for industrial applications |
| US9533897B2 (en) * | 2013-03-12 | 2017-01-03 | Radical Waters International Ltd. | Method for electro-chemical activation of water |
| US9828290B2 (en) | 2014-08-18 | 2017-11-28 | Garmor Inc. | Graphite oxide entrainment in cement and asphalt composite |
| EP3268311B1 (en) | 2015-03-13 | 2023-11-08 | University of Central Florida Research Foundation, Inc. | Uniform dispersing of graphene nanoparticles in a host |
| JP6522777B2 (ja) | 2015-03-23 | 2019-05-29 | ガーマー インク.Garmor, Inc. | 酸化グラフェンを用いた設計複合構造体 |
| KR101979575B1 (ko) | 2015-04-13 | 2019-05-17 | 갈모어 인코포레이티드 | 콘크리트 또는 아스팔트와 같은 호스트 중의 그래파이트 옥사이드 강화된 섬유 |
| WO2016200469A1 (en) | 2015-06-09 | 2016-12-15 | Garmor Inc. | Graphite oxide and polyacrylonitrile based composite |
| WO2017053204A1 (en) | 2015-09-21 | 2017-03-30 | Garmor Inc. | Low-cost, high-performance composite bipolar plate |
| EP3532543B1 (en) | 2016-10-26 | 2022-07-13 | Asbury Graphite of North Carolina, Inc. | Additive coated particles for low high performance materials |
| US10240245B2 (en) | 2017-06-28 | 2019-03-26 | Honeywell International Inc. | Systems, methods, and anodes for enhanced ionic liquid bath plating of turbomachine components and other workpieces |
| US11974752B2 (en) | 2019-12-12 | 2024-05-07 | Covidien Lp | Electrically enhanced retrieval of material from vessel lumens |
| US12004803B2 (en) | 2021-03-15 | 2024-06-11 | Covidien Lp | Thrombectomy treatment system |
| US10874411B2 (en) | 2018-06-22 | 2020-12-29 | Covidien Lp | Electrically enhanced retrieval of material from vessel lumens |
| US12318126B2 (en) | 2021-06-25 | 2025-06-03 | Covidien Lp | Current generator for a medical treatment system |
| US11058444B2 (en) | 2017-12-11 | 2021-07-13 | Covidien Lp | Electrically enhanced retrieval of material from vessel lumens |
| CN108716017B (zh) * | 2018-06-19 | 2023-07-28 | 昆山硕凯自动化科技有限公司 | 一种连续点蜡轴 |
| CN108707951B (zh) * | 2018-06-19 | 2023-08-01 | 昆山硕凯自动化科技有限公司 | 一种连续点蜡槽 |
| KR102639119B1 (ko) | 2018-12-31 | 2024-02-20 | 엘지디스플레이 주식회사 | 전기 도금 장치 및 이를 이용한 전기 도금 방법 |
| WO2020160531A1 (en) * | 2019-02-01 | 2020-08-06 | Lumishield Technologies Incorporated | Methods and compositions for improved adherence of organic coatings to materials |
| US11612430B2 (en) | 2019-03-19 | 2023-03-28 | Covidien Lp | Electrically enhanced retrieval of material from vessel lumens |
| US11523838B2 (en) | 2019-06-12 | 2022-12-13 | Covidien Lp | Retrieval of material from corporeal lumens |
| US11791061B2 (en) | 2019-09-12 | 2023-10-17 | Asbury Graphite North Carolina, Inc. | Conductive high strength extrudable ultra high molecular weight polymer graphene oxide composite |
| US12320025B2 (en) * | 2020-09-11 | 2025-06-03 | University Of Cincinnati | Electrochemical deposition of functionalized high entropy alloys |
| US11963713B2 (en) | 2021-06-02 | 2024-04-23 | Covidien Lp | Medical treatment system |
| CN113668039A (zh) * | 2021-08-17 | 2021-11-19 | Oppo广东移动通信有限公司 | 挂具组件和设备组件 |
| US11944374B2 (en) | 2021-08-30 | 2024-04-02 | Covidien Lp | Electrical signals for retrieval of material from vessel lumens |
| CN114108048B (zh) * | 2021-11-19 | 2023-05-23 | 南京航空航天大学 | 一种提高晶圆级阵列微结构电铸厚度均匀性的方法 |
| US20230323553A1 (en) * | 2022-04-12 | 2023-10-12 | The Regents Of The University Of Colorado, A Body Corporate | Pulsed electrodeposition for reversible metal electrodeposition to control metal film morphology and optical properties |
| CN115679398B (zh) * | 2022-11-17 | 2023-06-16 | 重庆太蓝新能源有限公司 | 参比电极的电镀方法及电池 |
| CN116240611B (zh) * | 2023-02-23 | 2025-07-25 | 京东方科技集团股份有限公司 | 一种电镀板、电镀装置以及电镀方法 |
| DE102023206303A1 (de) * | 2023-07-03 | 2025-01-09 | Semsysco Gmbh | Verteilungssystem für einen elektrischen Strom für unterschiedliche elektrolytische Oberflächenbehandlungen auf mindestens zwei unterschiedlichen Substratoberflächen eines Substrats |
Family Cites Families (35)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3269932A (en) | 1961-09-01 | 1966-08-30 | Exxon Research Engineering Co | Multi-cell reactor in parallel |
| US3365382A (en) | 1964-09-09 | 1968-01-23 | Adrian T. Godschalx | Electrical distribution system for continuous plating apparatus |
| US4077864A (en) * | 1973-09-10 | 1978-03-07 | General Dynamics | Electroforming anode shields |
| US3909368A (en) * | 1974-07-12 | 1975-09-30 | Louis W Raymond | Electroplating method and apparatus |
| US4155821A (en) * | 1974-11-25 | 1979-05-22 | Falconbridge Nickel Mines Limited | Electrowinning metal from chloride solution |
| US4197169A (en) * | 1978-09-05 | 1980-04-08 | Exxon Research & Engineering Co. | Shunt current elimination and device |
| US4277317A (en) * | 1979-11-26 | 1981-07-07 | Exxon Research & Engineering Co. | Shunt current elimination and device employing tunneled protective current |
| US4377445A (en) * | 1980-11-07 | 1983-03-22 | Exxon Research And Engineering Co. | Shunt current elimination for series connected cells |
| JPS5928597A (ja) * | 1982-08-05 | 1984-02-15 | Ibiden Co Ltd | 炭素電極棒の電気メッキ方法及びそのメッキ装置 |
| US4565607A (en) | 1984-03-09 | 1986-01-21 | Energy Conversion Devices, Inc. | Method of fabricating an electroplated substrate |
| US4686014A (en) | 1984-11-23 | 1987-08-11 | Pellegrino Peter P | Turbulent cell electroplating method and apparatus |
| US4935109A (en) * | 1988-05-23 | 1990-06-19 | General Dynamics Corp., Pomona Div. | Double-cell electroplating apparatus and method |
| US5000827A (en) | 1990-01-02 | 1991-03-19 | Motorola, Inc. | Method and apparatus for adjusting plating solution flow characteristics at substrate cathode periphery to minimize edge effect |
| US5358610A (en) * | 1992-07-20 | 1994-10-25 | Fuji Photo Film Co., Ltd. | Method for electrolytic treatment |
| US5352266A (en) | 1992-11-30 | 1994-10-04 | Queen'university At Kingston | Nanocrystalline metals and process of producing the same |
| US5433797A (en) | 1992-11-30 | 1995-07-18 | Queen's University | Nanocrystalline metals |
| US5312532A (en) * | 1993-01-15 | 1994-05-17 | International Business Machines Corporation | Multi-compartment eletroplating system |
| US5421987A (en) | 1993-08-30 | 1995-06-06 | Tzanavaras; George | Precision high rate electroplating cell and method |
| US5756874A (en) | 1995-10-10 | 1998-05-26 | Eosystems, Inc. | Electrochemical cell for processing organic wastes |
| US5776327A (en) | 1996-10-16 | 1998-07-07 | Mitsubishi Semiconuctor Americe, Inc. | Method and apparatus using an anode basket for electroplating a workpiece |
| DE19736351C1 (de) * | 1997-08-21 | 1998-10-01 | Atotech Deutschland Gmbh | Verfahren und Vorrichtung zum präzisen Galvanisieren von Werkstücken |
| US6027631A (en) | 1997-11-13 | 2000-02-22 | Novellus Systems, Inc. | Electroplating system with shields for varying thickness profile of deposited layer |
| US6491806B1 (en) * | 2000-04-27 | 2002-12-10 | Intel Corporation | Electroplating bath composition |
| US6776891B2 (en) * | 2001-05-18 | 2004-08-17 | Headway Technologies, Inc. | Method of manufacturing an ultra high saturation moment soft magnetic thin film |
| US20050205425A1 (en) * | 2002-06-25 | 2005-09-22 | Integran Technologies | Process for electroplating metallic and metall matrix composite foils, coatings and microcomponents |
| DE10311575B4 (de) * | 2003-03-10 | 2007-03-22 | Atotech Deutschland Gmbh | Verfahren zum elektrolytischen Metallisieren von Werkstücken mit Bohrungen mit einem hohen Aspektverhältnis |
| US6884335B2 (en) * | 2003-05-20 | 2005-04-26 | Novellus Systems, Inc. | Electroplating using DC current interruption and variable rotation rate |
| US20060042932A1 (en) * | 2004-08-25 | 2006-03-02 | Rosenzweig Mark A | Apparatus and method for electroplating a workpiece |
| US7435324B2 (en) * | 2004-09-02 | 2008-10-14 | Micron Technology, Inc. | Noncontact localized electrochemical deposition of metal thin films |
| US7354354B2 (en) | 2004-12-17 | 2008-04-08 | Integran Technologies Inc. | Article comprising a fine-grained metallic material and a polymeric material |
| US7387578B2 (en) * | 2004-12-17 | 2008-06-17 | Integran Technologies Inc. | Strong, lightweight article containing a fine-grained metallic layer |
| US7320832B2 (en) | 2004-12-17 | 2008-01-22 | Integran Technologies Inc. | Fine-grained metallic coatings having the coefficient of thermal expansion matched to the one of the substrate |
| US7425255B2 (en) * | 2005-06-07 | 2008-09-16 | Massachusetts Institute Of Technology | Method for producing alloy deposits and controlling the nanostructure thereof using negative current pulsing electro-deposition |
| DE102005032738B3 (de) * | 2005-07-08 | 2006-11-23 | Siemens Ag | Verfahren und Vorrichtung zum Bearbeiten wenigstens zweier Werkstücke mittels elektrochemischer Behandlung |
| US9005420B2 (en) * | 2007-12-20 | 2015-04-14 | Integran Technologies Inc. | Variable property electrodepositing of metallic structures |
-
2008
- 2008-04-18 US US12/081,623 patent/US8062496B2/en active Active
-
2009
- 2009-03-04 WO PCT/CA2009/000264 patent/WO2009127037A1/en not_active Ceased
- 2009-03-04 MX MX2010010658A patent/MX2010010658A/es active IP Right Grant
- 2009-03-04 KR KR1020107023293A patent/KR20110008043A/ko not_active Ceased
- 2009-03-04 CN CN2009801136044A patent/CN102007232B/zh not_active Expired - Fee Related
- 2009-03-04 EP EP09733070A patent/EP2262928A1/en not_active Withdrawn
- 2009-03-04 BR BRPI0910587A patent/BRPI0910587A2/pt not_active IP Right Cessation
- 2009-03-04 CA CA2716394A patent/CA2716394A1/en not_active Abandoned
-
2011
- 2011-10-07 US US13/268,175 patent/US20120024696A1/en not_active Abandoned
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20150118080A (ko) * | 2012-07-27 | 2015-10-21 | 인스티튜토 슈페리어 테크니코 | 수지상 조직을 갖는 니켈-코발트 피막의 전착법 |
| KR20190036619A (ko) * | 2017-09-28 | 2019-04-05 | (주)포인텍 | 전기도금라인의 무선행거에 장착된 기판에 인가되는 전류의 제어방법 |
| KR102012731B1 (ko) * | 2018-12-06 | 2019-08-21 | 주식회사 에이엔씨코리아 | 6 가 크롬도금액 및 이를 이용한 크랙프리 펄스 전기도금방법 |
Also Published As
| Publication number | Publication date |
|---|---|
| US20120024696A1 (en) | 2012-02-02 |
| US8062496B2 (en) | 2011-11-22 |
| MX2010010658A (es) | 2010-11-09 |
| CA2716394A1 (en) | 2009-10-22 |
| US20100006445A1 (en) | 2010-01-14 |
| BRPI0910587A2 (pt) | 2015-09-22 |
| EP2262928A1 (en) | 2010-12-22 |
| CN102007232B (zh) | 2013-05-01 |
| CN102007232A (zh) | 2011-04-06 |
| WO2009127037A1 (en) | 2009-10-22 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR20110008043A (ko) | 전착 방법 및 전착 장치 | |
| CN101903566B (zh) | 具有可变性质的金属性结构 | |
| CN103261479B (zh) | 纳米层压黄铜合金的材料及其电化学沉积方法 | |
| EP1516076B1 (en) | Process for electroplating metallic and metall matrix composite foils, coatings and microcomponents | |
| Bacher et al. | Fabrication of LIGA mold inserts | |
| CN103726083A (zh) | 纳晶和非晶金属及合金作为涂层的实施方法 | |
| US11527782B1 (en) | Pulse reverse current high rate electrodeposition and charging while mitigating the adverse effects of dendrite formation | |
| CN104911676B (zh) | 具有滤过膜的碱性电镀浴 | |
| US8425751B1 (en) | Systems and methods for the electrodeposition of a nickel-cobalt alloy | |
| CN101432467A (zh) | 通过电流分布的变化控制电沉积的铜镀层的硬度 | |
| Celis et al. | Electroplating technology | |
| US20070014997A1 (en) | Tool and method of making and using the same | |
| Marita et al. | Preparation of Ni-Co alloy by electrodeposition | |
| Yelton et al. | Electroplated coatings for friction, lubrication, and wear technology | |
| Fallah | Fabrication and optimization of 3D metallic parts using electroplating of additively manufactured parts | |
| Petrović | Technology improvement of copper plating on the constituents of internal crusher gauges | |
| CN118360640A (zh) | 电镀装置 | |
| HK1075278B (en) | Process for electroplating metallic and metal matrix composite foils, coatings and microcomponents | |
| HK1105218A (en) | Process for electroplating metallic and metal matrix composite foils and microcomponents |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0105 | International application |
Patent event date: 20101018 Patent event code: PA01051R01D Comment text: International Patent Application |
|
| PG1501 | Laying open of application | ||
| A201 | Request for examination | ||
| PA0201 | Request for examination |
Patent event code: PA02012R01D Patent event date: 20140304 Comment text: Request for Examination of Application |
|
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
Comment text: Notification of reason for refusal Patent event date: 20150508 Patent event code: PE09021S01D |
|
| E601 | Decision to refuse application | ||
| PE0601 | Decision on rejection of patent |
Patent event date: 20150905 Comment text: Decision to Refuse Application Patent event code: PE06012S01D Patent event date: 20150508 Comment text: Notification of reason for refusal Patent event code: PE06011S01I |