KR20170061177A - 결정질 메틸티오니늄 클로라이드(메틸렌 블루) 수화물 - Google Patents
결정질 메틸티오니늄 클로라이드(메틸렌 블루) 수화물 Download PDFInfo
- Publication number
- KR20170061177A KR20170061177A KR1020177013764A KR20177013764A KR20170061177A KR 20170061177 A KR20170061177 A KR 20170061177A KR 1020177013764 A KR1020177013764 A KR 1020177013764A KR 20177013764 A KR20177013764 A KR 20177013764A KR 20170061177 A KR20170061177 A KR 20170061177A
- Authority
- KR
- South Korea
- Prior art keywords
- methylthioninium chloride
- powder
- crystalline
- methylthioninium
- type
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D279/00—Heterocyclic compounds containing six-membered rings having one nitrogen atom and one sulfur atom as the only ring hetero atoms
- C07D279/10—1,4-Thiazines; Hydrogenated 1,4-thiazines
- C07D279/14—1,4-Thiazines; Hydrogenated 1,4-thiazines condensed with carbocyclic rings or ring systems
- C07D279/18—[b, e]-condensed with two six-membered rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/44—Non condensed pyridines; Hydrogenated derivatives thereof
- A61K31/445—Non condensed piperidines, e.g. piperocaine
- A61K31/4515—Non condensed piperidines, e.g. piperocaine having a butyrophenone group in position 1, e.g. haloperidol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/28—Drugs for disorders of the nervous system for treating neurodegenerative disorders of the central nervous system, e.g. nootropic agents, cognition enhancers, drugs for treating Alzheimer's disease or other forms of dementia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
- A61P31/14—Antivirals for RNA viruses
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
- A61P31/14—Antivirals for RNA viruses
- A61P31/18—Antivirals for RNA viruses for HIV
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D279/00—Heterocyclic compounds containing six-membered rings having one nitrogen atom and one sulfur atom as the only ring hetero atoms
- C07D279/10—1,4-Thiazines; Hydrogenated 1,4-thiazines
- C07D279/14—1,4-Thiazines; Hydrogenated 1,4-thiazines condensed with carbocyclic rings or ring systems
- C07D279/18—[b, e]-condensed with two six-membered rings
- C07D279/22—[b, e]-condensed with two six-membered rings with carbon atoms directly attached to the ring nitrogen atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07B—GENERAL METHODS OF ORGANIC CHEMISTRY; APPARATUS THEREFOR
- C07B2200/00—Indexing scheme relating to specific properties of organic compounds
- C07B2200/13—Crystalline forms, e.g. polymorphs
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02A—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE
- Y02A50/00—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE in human health protection, e.g. against extreme weather
- Y02A50/30—Against vector-borne diseases, e.g. mosquito-borne, fly-borne, tick-borne or waterborne diseases whose impact is exacerbated by climate change
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Virology (AREA)
- Oncology (AREA)
- Communicable Diseases (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Molecular Biology (AREA)
- Neurology (AREA)
- Neurosurgery (AREA)
- Biomedical Technology (AREA)
- Epidemiology (AREA)
- Psychiatry (AREA)
- Hospice & Palliative Care (AREA)
- AIDS & HIV (AREA)
- Dermatology (AREA)
- Tropical Medicine & Parasitology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Nitrogen- Or Sulfur-Containing Heterocyclic Ring Compounds With Rings Of Six Or More Members (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Steroid Compounds (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
- Crystals, And After-Treatments Of Crystals (AREA)
Abstract
Description
도 2는 메틸티오니늄 클로라이드 C형의 결정질 2수화물의 특징적인 X선 분말 회절 패턴이다.
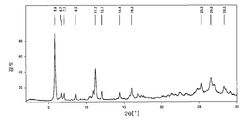
도 3은 메틸티오니늄 클로라이드 D형의 결정질 2수화물의 특징적인 X선 분말 회절 패턴이다.
도 4는 메틸티오니늄 클로라이드 E형의 결정질 1수화물의 특징적인 X선 분말 회절 패턴이다.
Claims (15)
- C형으로서의 결정질 메틸티오니늄 클로라이드 2수화물.
- 제1항 또는 제2항에 따른 실질적으로 C형인 메틸티오니늄 클로라이드 2수화물의 제조 방법으로서, 함수 메틸티오니늄 클로라이드 또는 이의 다양한 수화물의 혼합물 또는 이의 특정한 수화물을 소량의 물을 함유하는 이소프로판올, 1-프로판올, 1-부탄올, 2-부탄올, tert-부탄올, 테트라히드로푸란, 디옥산, 아세톤, 2-부타논 및 아세토니트릴을 포함하는 군으로부터 선택되는 용매 중에 주위 온도에서 C형을 생성하기에 충분한 시간 동안 현탁시키고 교반하며; 그 후 고체를 분리하고; 고체로부터 용매를 제거하는 것인 제조 방법.
- E형으로서의 결정질 메틸티오니늄 클로라이드 1수화물.
- 제4항 또는 제5항에 따른 실질적으로 E형인 메틸티오니늄 클로라이드 1수화물의 제조 방법으로서, 함수 메틸티오니늄 클로라이드 또는 메틸티오니늄 클로라이드의 다양한 수화물의 혼합물 또는 메틸티오니늄 클로라이드의 특정한 수화물을 무수 용매, 바람직하게는 이소프로판올 중에 주위 온도에서 E형을 생성하기에 충분한 시간 동안 현탁시키고 교반하며; 고체를 여과로 분리하고; 진공 건조 또는 건조 가스에 의한 퍼징에 의해 고체로부터 잔류 용매를 제거하는 것인 제조 방법.
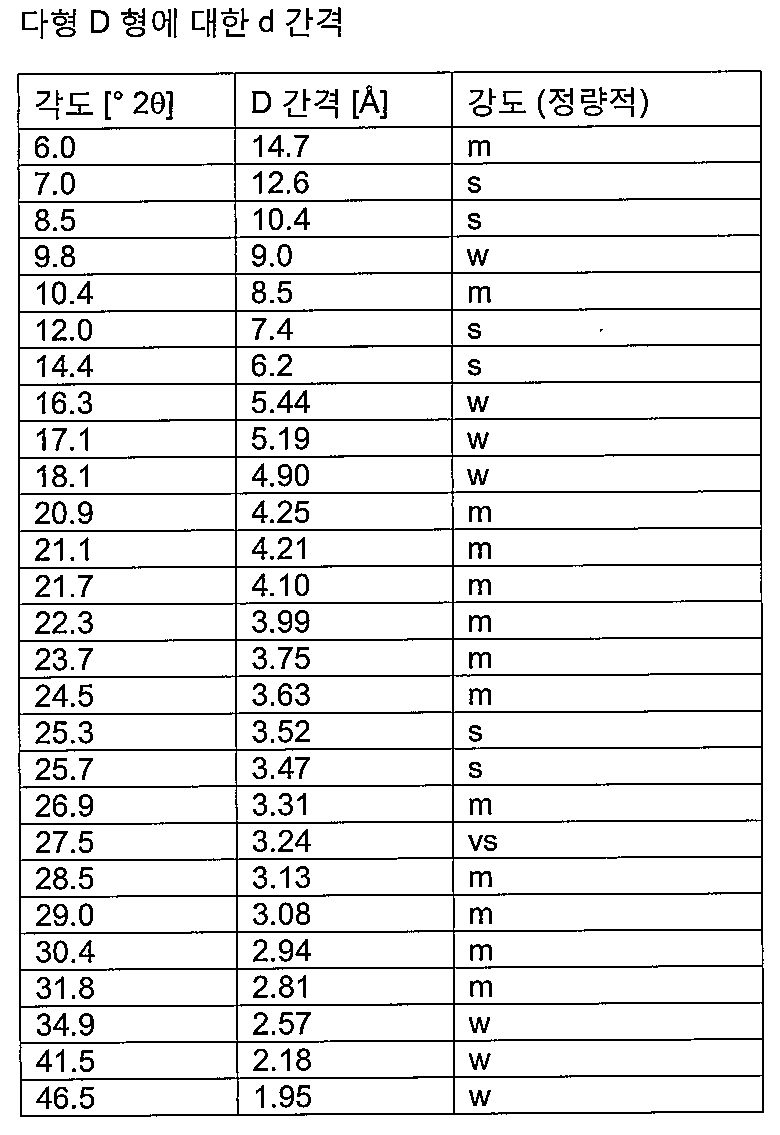
- D형으로서의 결정질 메틸티오니늄 클로라이드 2수화물.
- 메틸티오니늄 클로라이드 5수화물을 아세트산 중에 용해시키고, 톨루엔을 아세트산 용액에 첨가하거나 아세트산 용액을 톨루엔에 첨가함으로써 아세트산 용액을 톨루엔과 합하는 단계;
고체를 침전 직후 여과로 분리하는 단계;
용매를 진공 건조에 의해 또는 불활성 가스 흐름에 의해 제거하는 단계를 포함하는, 제7항 또는 제8항에 따른 실질적으로 D형인 메틸티오니늄 클로라이드 2수화물의 제조 방법으로서, 모든 공정 단계에서의 상대 공기 습도가 50% 미만인 제조 방법. - 실질적으로 B형으로서의 결정질 메틸티오니늄 클로라이드 2수화물.
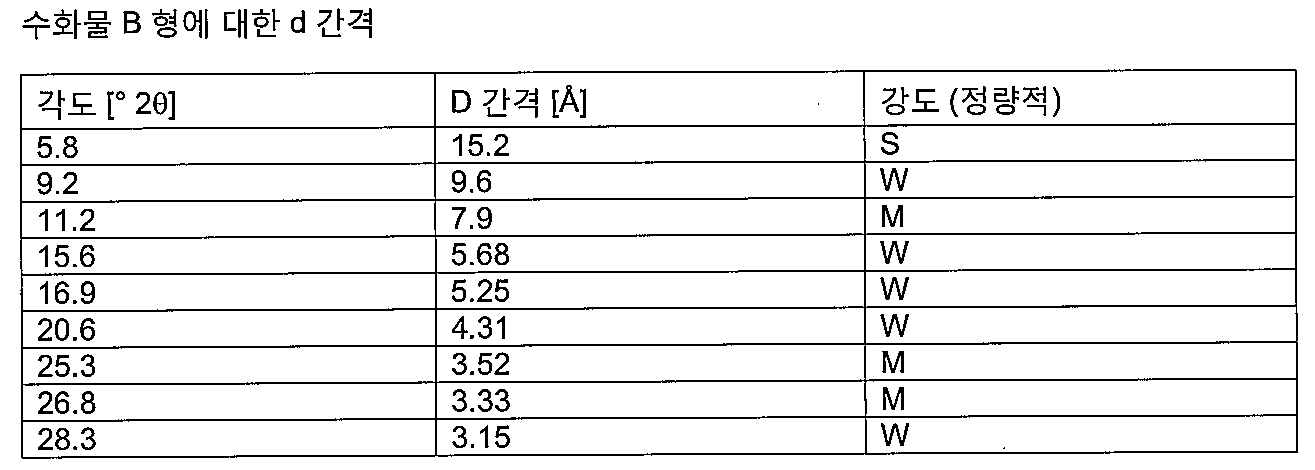
- 고체 메틸티오니늄 클로라이드 5수화물을 대략 실온에서 실질적으로 순수한 B형을 생성하기에 충분한 시간 동안 8 내지 15%의 상대 습도를 갖는 불활성 가스 흐름에 노출시키는 단계를 포함하는, 제10항 또는 제11항에 따른 실질적으로 B형인 메틸티오니늄 클로라이드 2수화물의 제조 방법.
- 제1항, 제2항, 제4항, 제5항, 제7항, 제8항, 제10항 및 제11항 중 어느 한 항에 따른 메틸티오니늄 클로라이드 2수화물 B형, C형 또는 D형 또는 메틸티오니늄 클로라이드 1수화물 E형 및 선택적으로 약학적으로 허용가능한 담체, 부형제 또는 희석제를 포함하는 약학 조성물.
- 치료에 의한 인간 또는 동물 신체의 치료 방법에서 사용하기 위한, 제1항, 제2항, 제4항, 제5항, 제7항, 제8항, 제10항 및 제11항 중 어느 한 항에 따른 메틸티오니늄 클로라이드 2수화물 B형, C형 또는 D형 또는 메틸티오니늄 클로라이드 1수화물 E형.
- 질병 상태의 치료에 사용되는 제1항, 제2항, 제4항, 제5항, 제7항, 제8항, 제10항 및 제11항 중 어느 한 항에 따른 메틸티오니늄 클로라이드 2수화물 B형, C형 또는 D형 또는 메틸티오니늄 클로라이드 1수화물 E형의 용도로서, 상기 질병 상태의 치료는 예를 들어, 타우병증, 알츠하이머병(AD), 피부암, 흑색종, C형 간염, HIV 또는 웨스트 나일 바이러스의 치료인 것인 용도.
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US24539109P | 2009-09-24 | 2009-09-24 | |
| US61/245,391 | 2009-09-24 | ||
| PCT/IB2010/002526 WO2011036558A2 (en) | 2009-09-24 | 2010-09-23 | Crystalline methylthionium chloride (methylene blue) hydrates |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020127009853A Division KR20120099409A (ko) | 2009-09-24 | 2010-09-23 | 결정질 메틸티오니늄 클로라이드(메틸렌 블루) 수화물 |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020187022958A Division KR20180094126A (ko) | 2009-09-24 | 2010-09-23 | 결정질 메틸티오니늄 클로라이드(메틸렌 블루) 수화물 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| KR20170061177A true KR20170061177A (ko) | 2017-06-02 |
Family
ID=43415329
Family Applications (3)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020177013764A Ceased KR20170061177A (ko) | 2009-09-24 | 2010-09-23 | 결정질 메틸티오니늄 클로라이드(메틸렌 블루) 수화물 |
| KR1020187022958A Ceased KR20180094126A (ko) | 2009-09-24 | 2010-09-23 | 결정질 메틸티오니늄 클로라이드(메틸렌 블루) 수화물 |
| KR1020127009853A Ceased KR20120099409A (ko) | 2009-09-24 | 2010-09-23 | 결정질 메틸티오니늄 클로라이드(메틸렌 블루) 수화물 |
Family Applications After (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020187022958A Ceased KR20180094126A (ko) | 2009-09-24 | 2010-09-23 | 결정질 메틸티오니늄 클로라이드(메틸렌 블루) 수화물 |
| KR1020127009853A Ceased KR20120099409A (ko) | 2009-09-24 | 2010-09-23 | 결정질 메틸티오니늄 클로라이드(메틸렌 블루) 수화물 |
Country Status (19)
| Country | Link |
|---|---|
| US (1) | US8765742B2 (ko) |
| EP (4) | EP3792254B1 (ko) |
| JP (1) | JP5806217B2 (ko) |
| KR (3) | KR20170061177A (ko) |
| CN (2) | CN105061358A (ko) |
| AU (1) | AU2010299571B2 (ko) |
| BR (4) | BR112012006638B1 (ko) |
| CA (4) | CA3004822C (ko) |
| DK (4) | DK2963024T3 (ko) |
| ES (4) | ES2960761T3 (ko) |
| FI (1) | FI3792254T3 (ko) |
| HR (3) | HRP20231201T1 (ko) |
| MX (3) | MX381636B (ko) |
| MY (1) | MY159782A (ko) |
| PL (4) | PL3378856T3 (ko) |
| PT (4) | PT2480541E (ko) |
| SG (1) | SG10201408584UA (ko) |
| SI (4) | SI3378856T1 (ko) |
| WO (1) | WO2011036558A2 (ko) |
Families Citing this family (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9925282B2 (en) | 2009-01-29 | 2018-03-27 | The General Hospital Corporation | Cromolyn derivatives and related methods of imaging and treatment |
| PL2480540T3 (pl) * | 2009-09-24 | 2018-05-30 | Wista Laboratories Ltd. | Pentahydrat chlorku metylotioninowego, jego wytwarzanie i zastosowanie farmaceutyczne |
| US10058530B2 (en) | 2012-10-25 | 2018-08-28 | The General Hospital Corporation | Combination therapies for the treatment of Alzheimer's disease and related disorders |
| US9855276B2 (en) | 2012-10-25 | 2018-01-02 | The General Hospital Corporation | Combination therapies for the treatment of Alzheimer's disease and related disorders |
| US10525005B2 (en) | 2013-05-23 | 2020-01-07 | The General Hospital Corporation | Cromolyn compositions and methods thereof |
| AU2014340182B2 (en) | 2013-10-22 | 2019-05-23 | The General Hospital Corporation | Cromolyn derivatives and related methods of imaging and treatment |
| US10765755B1 (en) | 2013-11-20 | 2020-09-08 | University Of South Florida | Preparation and characterization of methylene blue nanoparticles for Alzheimer's disease and other tauopathies |
| SI3487505T1 (sl) | 2016-07-25 | 2023-10-30 | Wista Laboratories Ltd., | Dajanje in odmerek diaminofenotiazinov |
| CN109922800B (zh) | 2016-08-31 | 2023-06-13 | 通用医疗公司 | 与神经退行性疾病相关的神经炎症中的巨噬细胞/小胶质细胞 |
| GB201614834D0 (en) | 2016-09-01 | 2016-10-19 | Wista Lab Ltd | Treatment of dementia |
| MX2020000577A (es) | 2017-07-20 | 2020-09-10 | Aztherapies Inc | Formulaciones en polvo de cromolina sodica e ibuprofeno. |
| JP2021529771A (ja) | 2018-07-02 | 2021-11-04 | ザ ジェネラル ホスピタル コーポレイション | クロモリンナトリウムおよびα−ラクトースの粉末製剤 |
| SG11202106117SA (en) | 2018-12-10 | 2021-07-29 | Massachusetts Gen Hospital | Cromolyn esters and uses thereof |
| WO2021207060A1 (en) | 2020-04-06 | 2021-10-14 | The General Hospital Corporation | Methods of treatment of coronavirus-induced inflammation conditions |
| US20260048066A1 (en) | 2022-05-31 | 2026-02-19 | Wista Laboratories Ltd | Treatment of neurodegenerative disorders utilising methylthioninium (mt)-containing compounds |
| EP4590275A1 (en) | 2022-09-21 | 2025-07-30 | WisTa Laboratories Ltd. | Novel formulations and vehicles |
Family Cites Families (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB9506197D0 (en) | 1995-03-27 | 1995-05-17 | Hoffmann La Roche | Inhibition of tau-tau association. |
| GB0101049D0 (en) | 2001-01-15 | 2001-02-28 | Univ Aberdeen | Materials and methods relating to protein aggregation in neurodegenerative disease |
| GB0322756D0 (en) | 2003-09-29 | 2003-10-29 | Univ Aberdeen | Methods of chemical synthesis |
| PT2322517T (pt) | 2004-09-23 | 2019-07-30 | Wista Lab Ltd | Métodos de síntese química e purificação de compostos de diaminofenotiazínio incluindo cloreto de metiltionínio (mtc) |
| US7790881B2 (en) * | 2004-09-23 | 2010-09-07 | Wista Laboratories Ltd. | Methods of chemical synthesis and purification of diaminophenothiazinium compounds including methylthioninium chloride (MTC) |
| MY153198A (en) * | 2006-03-29 | 2015-01-29 | Wista Lab Ltd | Inhibitors of protein aggregation |
| ES2349322T7 (es) * | 2006-03-29 | 2019-10-17 | Wista Lab Ltd | Sales de 3,7-diamino-10H-fenotiazina y su utilización |
| US8227459B2 (en) * | 2007-04-03 | 2012-07-24 | Prosetta Antiviral Inc. | Diamino-phenothiazinyl derivatives as antiviral treatments |
| US7603854B2 (en) | 2007-04-10 | 2009-10-20 | Illinois Tool Works Inc. | Pneumatically self-regulating valve |
-
2010
- 2010-09-23 KR KR1020177013764A patent/KR20170061177A/ko not_active Ceased
- 2010-09-23 DK DK15176967.6T patent/DK2963024T3/en active
- 2010-09-23 PL PL18170358T patent/PL3378856T3/pl unknown
- 2010-09-23 SI SI201032051T patent/SI3378856T1/sl unknown
- 2010-09-23 FI FIEP20203173.8T patent/FI3792254T3/fi active
- 2010-09-23 BR BR112012006638-2A patent/BR112012006638B1/pt active IP Right Grant
- 2010-09-23 BR BR122019022628A patent/BR122019022628B8/pt active IP Right Grant
- 2010-09-23 PT PT107715161T patent/PT2480541E/pt unknown
- 2010-09-23 CA CA3004822A patent/CA3004822C/en active Active
- 2010-09-23 CN CN201510501874.XA patent/CN105061358A/zh active Pending
- 2010-09-23 MY MYPI2012001322A patent/MY159782A/en unknown
- 2010-09-23 SG SG10201408584UA patent/SG10201408584UA/en unknown
- 2010-09-23 SI SI201031765T patent/SI2963024T1/sl unknown
- 2010-09-23 ES ES20203173T patent/ES2960761T3/es active Active
- 2010-09-23 US US13/497,686 patent/US8765742B2/en active Active
- 2010-09-23 SI SI201031101T patent/SI2480541T1/sl unknown
- 2010-09-23 SI SI201032134T patent/SI3792254T1/sl unknown
- 2010-09-23 PL PL20203173.8T patent/PL3792254T3/pl unknown
- 2010-09-23 CN CN201080053178.2A patent/CN102666511B/zh active Active
- 2010-09-23 JP JP2012530357A patent/JP5806217B2/ja active Active
- 2010-09-23 EP EP20203173.8A patent/EP3792254B1/en active Active
- 2010-09-23 WO PCT/IB2010/002526 patent/WO2011036558A2/en not_active Ceased
- 2010-09-23 DK DK10771516.1T patent/DK2480541T3/en active
- 2010-09-23 PL PL10771516T patent/PL2480541T3/pl unknown
- 2010-09-23 BR BR122019022612A patent/BR122019022612B8/pt active IP Right Grant
- 2010-09-23 EP EP10771516.1A patent/EP2480541B1/en active Active
- 2010-09-23 HR HRP20231201TT patent/HRP20231201T1/hr unknown
- 2010-09-23 EP EP15176967.6A patent/EP2963024B1/en active Active
- 2010-09-23 EP EP18170358.8A patent/EP3378856B1/en active Active
- 2010-09-23 HR HRP20181567TT patent/HRP20181567T2/hr unknown
- 2010-09-23 CA CA3141737A patent/CA3141737C/en active Active
- 2010-09-23 PT PT181703588T patent/PT3378856T/pt unknown
- 2010-09-23 CA CA2773305A patent/CA2773305C/en active Active
- 2010-09-23 ES ES15176967.6T patent/ES2689029T3/es active Active
- 2010-09-23 ES ES18170358T patent/ES2847003T3/es active Active
- 2010-09-23 MX MX2018004983A patent/MX381636B/es unknown
- 2010-09-23 BR BR122019022631A patent/BR122019022631B8/pt active IP Right Grant
- 2010-09-23 CA CA3077911A patent/CA3077911C/en active Active
- 2010-09-23 ES ES10771516.1T patent/ES2557318T3/es active Active
- 2010-09-23 MX MX2015008552A patent/MX355627B/es unknown
- 2010-09-23 PT PT202031738T patent/PT3792254T/pt unknown
- 2010-09-23 PT PT15176967T patent/PT2963024T/pt unknown
- 2010-09-23 KR KR1020187022958A patent/KR20180094126A/ko not_active Ceased
- 2010-09-23 DK DK20203173.8T patent/DK3792254T3/da active
- 2010-09-23 PL PL15176967T patent/PL2963024T3/pl unknown
- 2010-09-23 DK DK18170358.8T patent/DK3378856T3/da active
- 2010-09-23 KR KR1020127009853A patent/KR20120099409A/ko not_active Ceased
- 2010-09-23 MX MX2012003083A patent/MX2012003083A/es active IP Right Grant
- 2010-09-23 AU AU2010299571A patent/AU2010299571B2/en not_active Ceased
-
2021
- 2021-01-19 HR HRP20210086TT patent/HRP20210086T1/hr unknown
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR20170061177A (ko) | 결정질 메틸티오니늄 클로라이드(메틸렌 블루) 수화물 | |
| KR101728574B1 (ko) | 실질적으로 순수한 메틸티오니늄 클로라이드 5수화물 형태 a의 제조 방법 | |
| AU2017228644B2 (en) | Crystalline methylthionium chloride (methylene blue) hydrates | |
| HK40049199B (en) | Crystalline methylthioninium chloride hydrates | |
| HK40049199A (en) | Crystalline methylthioninium chloride hydrates | |
| HK1261428B (en) | Crystalline methylthioninium chloride hydrates | |
| HK1261428A1 (en) | Crystalline methylthioninium chloride hydrates | |
| HK1167653B (en) | Crystalline methylthionium chloride (methylene blue) hydrates | |
| HK1213897B (en) | Crystalline methylthioninium chloride hydrates - part 2 | |
| AU2014253566A1 (en) | Crystalline methylthionium chloride (methylene blue) hydrates |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A107 | Divisional application of patent | ||
| PA0104 | Divisional application for international application |
St.27 status event code: A-0-1-A10-A18-div-PA0104 St.27 status event code: A-0-1-A10-A16-div-PA0104 |
|
| A201 | Request for examination | ||
| PA0201 | Request for examination |
St.27 status event code: A-1-2-D10-D11-exm-PA0201 |
|
| PG1501 | Laying open of application |
St.27 status event code: A-1-1-Q10-Q12-nap-PG1501 |
|
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
St.27 status event code: A-1-2-D10-D21-exm-PE0902 |
|
| T11-X000 | Administrative time limit extension requested |
St.27 status event code: U-3-3-T10-T11-oth-X000 |
|
| AMND | Amendment | ||
| P11-X000 | Amendment of application requested |
St.27 status event code: A-2-2-P10-P11-nap-X000 |
|
| P13-X000 | Application amended |
St.27 status event code: A-2-2-P10-P13-nap-X000 |
|
| E601 | Decision to refuse application | ||
| PE0601 | Decision on rejection of patent |
St.27 status event code: N-2-6-B10-B15-exm-PE0601 |
|
| T11-X000 | Administrative time limit extension requested |
St.27 status event code: U-3-3-T10-T11-oth-X000 |
|
| T13-X000 | Administrative time limit extension granted |
St.27 status event code: U-3-3-T10-T13-oth-X000 |
|
| T13-X000 | Administrative time limit extension granted |
St.27 status event code: U-3-3-T10-T13-oth-X000 |
|
| AMND | Amendment | ||
| E13-X000 | Pre-grant limitation requested |
St.27 status event code: A-2-3-E10-E13-lim-X000 |
|
| P11-X000 | Amendment of application requested |
St.27 status event code: A-2-2-P10-P11-nap-X000 |
|
| P13-X000 | Application amended |
St.27 status event code: A-2-2-P10-P13-nap-X000 |
|
| PX0901 | Re-examination |
St.27 status event code: A-2-3-E10-E12-rex-PX0901 |
|
| PX0601 | Decision of rejection after re-examination |
St.27 status event code: N-2-6-B10-B17-rex-PX0601 |
|
| PA0104 | Divisional application for international application |
St.27 status event code: A-0-1-A10-A18-div-PA0104 St.27 status event code: A-0-1-A10-A16-div-PA0104 |
|
| R18 | Changes to party contact information recorded |
Free format text: ST27 STATUS EVENT CODE: A-3-3-R10-R18-OTH-X000 (AS PROVIDED BY THE NATIONAL OFFICE) |
|
| R18-X000 | Changes to party contact information recorded |
St.27 status event code: A-3-3-R10-R18-oth-X000 |