KR20170077286A - 연속 세포 배양에서 목적 폴리펩티드 또는 바이러스를 생산하는 방법 - Google Patents
연속 세포 배양에서 목적 폴리펩티드 또는 바이러스를 생산하는 방법 Download PDFInfo
- Publication number
- KR20170077286A KR20170077286A KR1020177017512A KR20177017512A KR20170077286A KR 20170077286 A KR20170077286 A KR 20170077286A KR 1020177017512 A KR1020177017512 A KR 1020177017512A KR 20177017512 A KR20177017512 A KR 20177017512A KR 20170077286 A KR20170077286 A KR 20170077286A
- Authority
- KR
- South Korea
- Prior art keywords
- cell
- cells
- culture
- cell culture
- culture system
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N7/00—Viruses; Bacteriophages; Compositions thereof; Preparation or purification thereof
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12M—APPARATUS FOR ENZYMOLOGY OR MICROBIOLOGY; APPARATUS FOR CULTURING MICROORGANISMS FOR PRODUCING BIOMASS, FOR GROWING CELLS OR FOR OBTAINING FERMENTATION OR METABOLIC PRODUCTS, i.e. BIOREACTORS OR FERMENTERS
- C12M3/00—Tissue, human, animal or plant cell, or virus culture apparatus
- C12M3/006—Cell injection or fusion devices
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/0018—Culture media for cell or tissue culture
- C12N5/0056—Xeno-free medium
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N9/00—Enzymes; Proenzymes; Compositions thereof; Processes for preparing, activating, inhibiting, separating or purifying enzymes
- C12N9/14—Hydrolases (3)
- C12N9/48—Hydrolases (3) acting on peptide bonds (3.4)
- C12N9/50—Proteinases, e.g. Endopeptidases (3.4.21-3.4.25)
- C12N9/64—Proteinases, e.g. Endopeptidases (3.4.21-3.4.25) derived from animal tissue
- C12N9/6421—Proteinases, e.g. Endopeptidases (3.4.21-3.4.25) derived from animal tissue from mammals
- C12N9/6489—Metalloendopeptidases (3.4.24)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12P—FERMENTATION OR ENZYME-USING PROCESSES TO SYNTHESISE A DESIRED CHEMICAL COMPOUND OR COMPOSITION OR TO SEPARATE OPTICAL ISOMERS FROM A RACEMIC MIXTURE
- C12P21/00—Preparation of peptides or proteins
- C12P21/02—Preparation of peptides or proteins having a known sequence of two or more amino acids, e.g. glutathione
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2500/00—Specific components of cell culture medium
- C12N2500/98—Xeno-free medium and culture conditions
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2710/00—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA dsDNA viruses
- C12N2710/00011—Details
- C12N2710/00051—Methods of production or purification of viral material
- C12N2710/00052—Methods of production or purification of viral material relating to complementing cells and packaging systems for producing virus or viral particles
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Y—ENZYMES
- C12Y304/00—Hydrolases acting on peptide bonds, i.e. peptidases (3.4)
- C12Y304/24—Metalloendopeptidases (3.4.24)
- C12Y304/24087—ADAMTS13 endopeptidase (3.4.24.87)
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02P—CLIMATE CHANGE MITIGATION TECHNOLOGIES IN THE PRODUCTION OR PROCESSING OF GOODS
- Y02P20/00—Technologies relating to chemical industry
- Y02P20/50—Improvements relating to the production of bulk chemicals
- Y02P20/52—Improvements relating to the production of bulk chemicals using catalysts, e.g. selective catalysts
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Organic Chemistry (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Genetics & Genomics (AREA)
- Biomedical Technology (AREA)
- Biotechnology (AREA)
- General Engineering & Computer Science (AREA)
- Microbiology (AREA)
- Biochemistry (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Molecular Biology (AREA)
- Virology (AREA)
- Immunology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- General Chemical & Material Sciences (AREA)
- Cell Biology (AREA)
- Sustainable Development (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
- Preparation Of Compounds By Using Micro-Organisms (AREA)
- Enzymes And Modification Thereof (AREA)
- Peptides Or Proteins (AREA)
Abstract
Description
Claims (19)
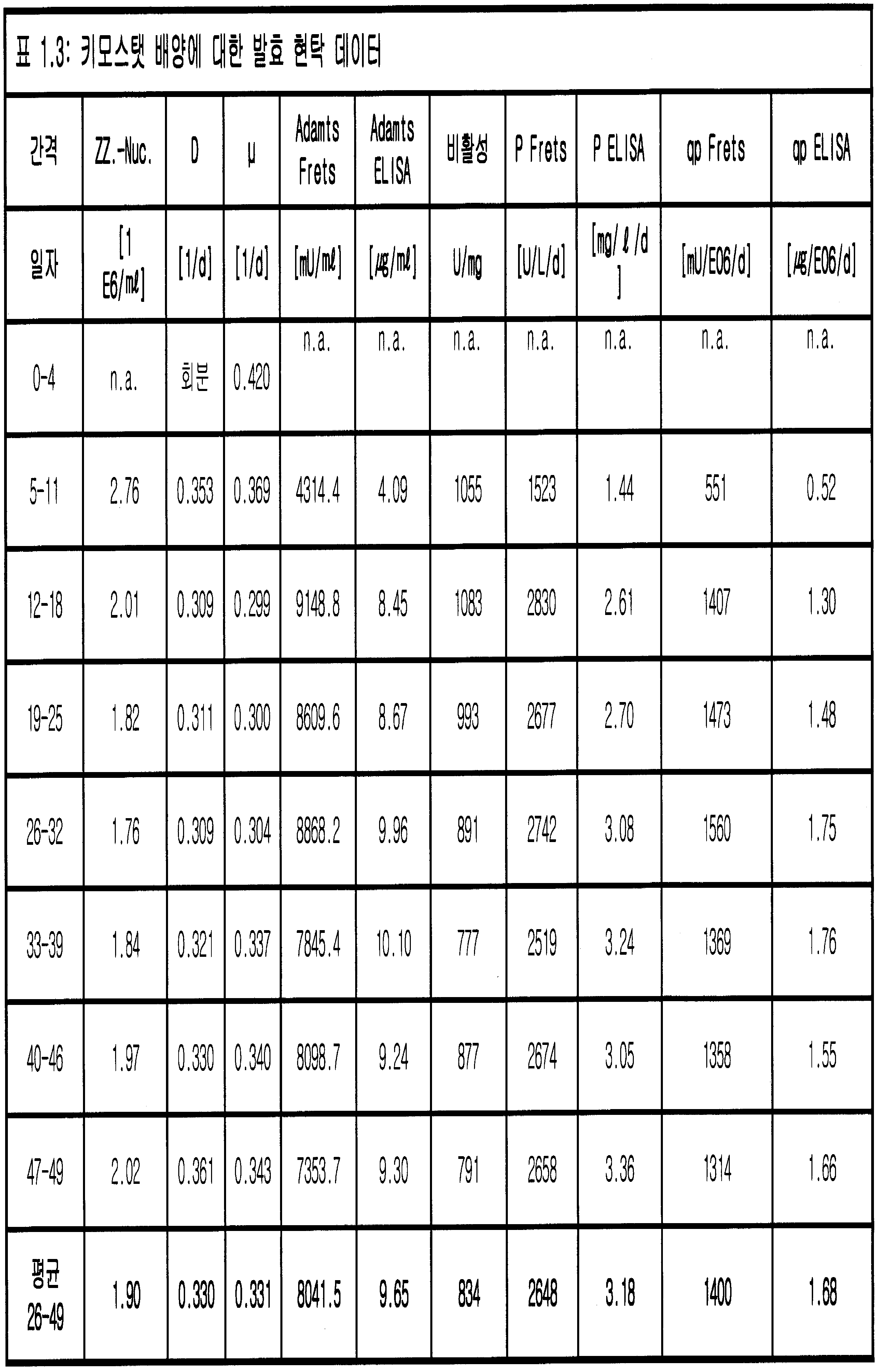
- (a) 연속 세포 배양 시스템에서 폴리펩티드를 발현하는 포유동물 세포를 배양하는 단계이며, 상기 세포 배양 시스템은 세포 유지 장치를 포함하고 2 d-1 미만의 희석 속도 (D) 및 2 X 107개 세포/㎖ 미만의 세포 밀도를 갖는, 단계; 및
(b) 상기 세포 배양 시스템으로부터 제거된 배지로부터 상기 폴리펩티드를 회수하는 단계를 포함하고,
여기서 상기 세포는 트롬보스폰딘 타입 1 모티프 13을 갖는 디스인테그린-유사 및 금속단백분해효소 (ADAMTS13) 단백질을 발현하도록 유전적으로 변형되는 것인,
연속 세포 배양에서 폴리펩티드를 생산하는 방법. - 제1항에 있어서, 상기 세포 유지 장치는 90% 미만의 세포 유지 비율을 갖는 것인 방법.
- 제1항에 있어서, 상기 세포 배양 시스템은 0.2 d-1 내지 0.8 d-1의 비 성장 속도를 갖는 것인 방법.
- 제2항에 있어서, 상기 세포 배양 시스템은 0.2 d-1 내지 0.8 d-1의 비 성장 속도를 갖는 것인 방법.
- 제1항 내지 제4항 중 어느 한 항에 있어서, 상기 희석 속도는 0.1 d-1 내지 1.0 d-1인 방법.
- 제1항 내지 제4항 중 어느 한 항에 있어서, 상기 세포 밀도는 1 X 107개 세포/㎖ 미만인 방법.
- 제1항 내지 제4항 중 어느 한 항에 있어서, 상기 세포 배양 시스템은 1.2 내지 5의 희석 속도와 비 성장 속도의 비율 (D/μ)을 갖는 것인 방법.
- 제1항 내지 제4항 중 어느 한 항에 있어서, 상기 세포는 20일 초과 동안 상기 세포 배양 시스템에서 배양되는 것인 방법.
- 제1항 내지 제4항 중 어느 한 항에 있어서, 상기 세포는 40일 초과 동안 상기 세포 배양 시스템에서 배양되는 것인 방법.
- 제1항 내지 제4항 중 어느 한 항에 있어서, 상기 세포는 50일 초과 동안 상기 세포 배양 시스템에서 배양되는 것인 방법.
- 제1항 내지 제4항 중 어느 한 항에 있어서, 상기 희석 속도 및 세포 밀도는 세포가 상기 세포 배양 시스템에서 배양되는 시간의 적어도 80% 동안 지속되는 것인 방법.
- 제1항 내지 제4항 중 어느 한 항에 있어서, 상기 세포 유지 장치는 마크로다공성 미립담체를 포함하는 것인 방법.
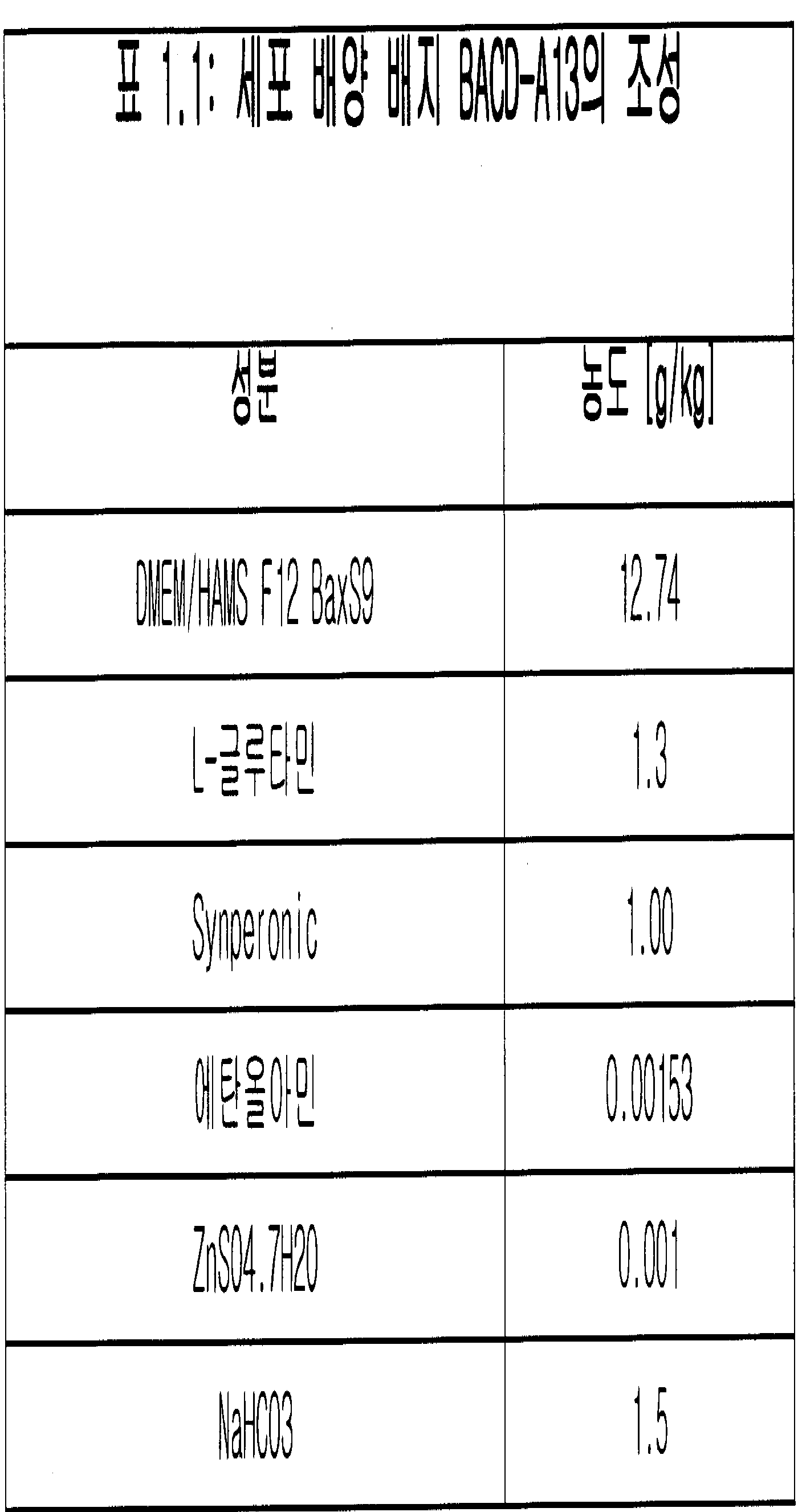
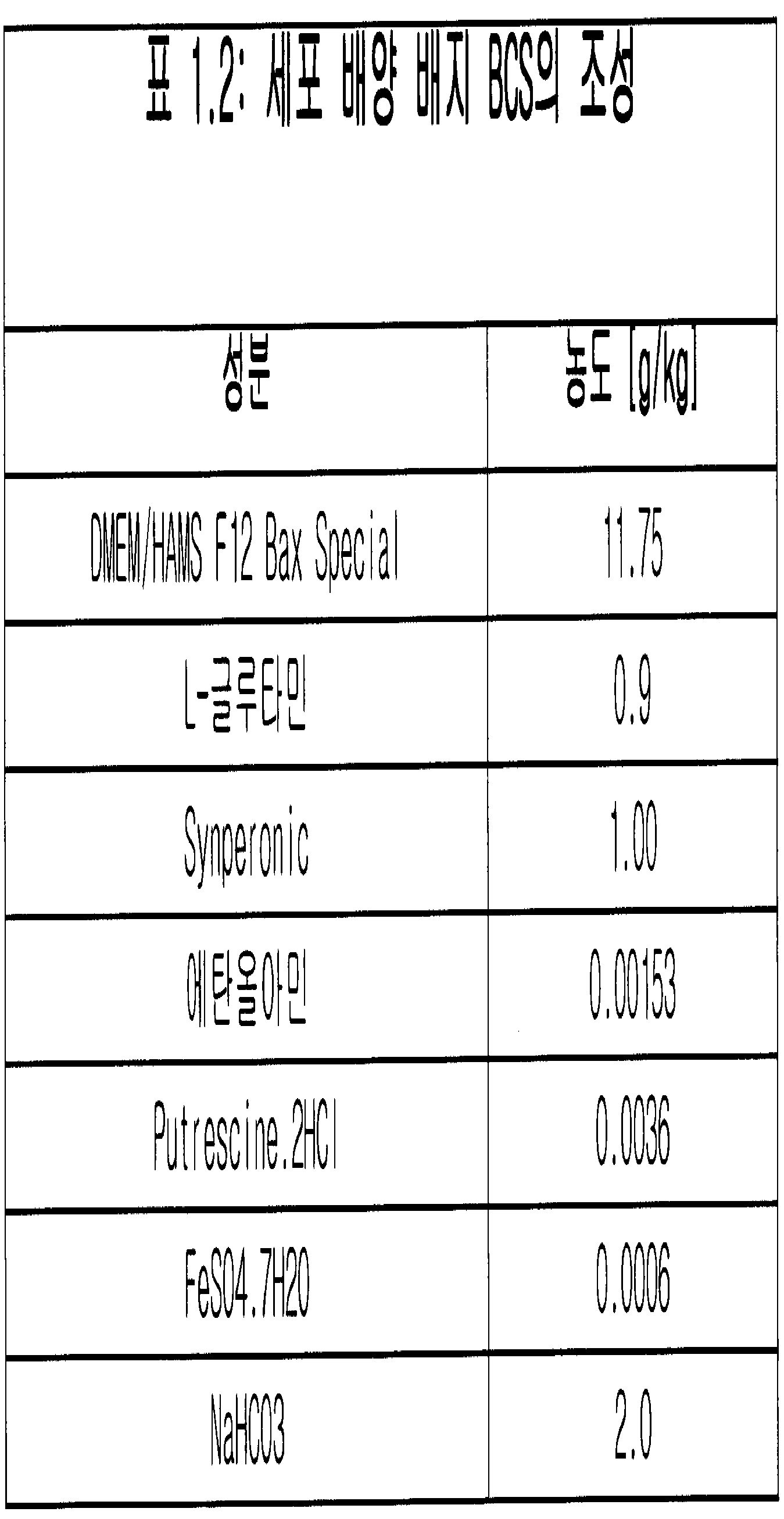
- 제1항 내지 제4항 중 어느 한 항에 있어서, 상기 세포는 혈청이 없는 배지에서 배양되는 것인 방법.
- 제1항 내지 제4항 중 어느 한 항에 있어서, 상기 세포는 적어도 250 ℓ의 배지에서 배양되는 것인 방법.
- 제1항 내지 제4항 중 어느 한 항에 있어서, 상기 세포는 비고정 의존성 세포인 방법.
- 제1항 내지 제4항 중 어느 한 항에 있어서, 배양 단계에 앞서, 현탁 상태에서 세포를 사전 배양하는 단계를 더 포함하는 것인 방법.
- 제1항 내지 제4항 중 어느 한 항에 있어서, 상기 세포는 CHO 세포인 방법.
- 제1항에 있어서,
(a) 연속 세포 배양 시스템에서 재조합 ADAMTS13 단백질을 발현하는 비고정 의존성 포유동물 세포를 배양하는 단계이며, 상기 세포 배양 시스템은 90% 미만의 세포 유지 비율을 갖는 세포 유지 장치를 포함하고 0.1 d-1 내지 1.0 d- 1 의 희석 속도 (D) 및 1 X 107개 세포/㎖ 미만의 세포 밀도를 갖는, 단계; 및
(b) 상기 세포 배양 시스템으로부터 제거된 배지로부터 ADAMTS13 단백질을 회수하는 단계를 포함하는,
연속 세포 배양에서 ADAMTS13 단백질을 생산하는 방법. - 제1항 내지 제4항 중 어느 한 항에 있어서, 상기 희석 속도는 약 1.0 d-1 미만, 약 0.9 d-1 미만, 약 0.8 d-1 미만, 약 0.7 d-1 미만, 또는 약 0.6 d-1 미만으로 지속되는 방법.
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US23031309P | 2009-07-31 | 2009-07-31 | |
| US61/230,313 | 2009-07-31 | ||
| PCT/EP2010/061191 WO2011012725A1 (en) | 2009-07-31 | 2010-08-02 | Method of producing a polypeptide or virus of interest in a continuous cell culture |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020127004996A Division KR101752789B1 (ko) | 2009-07-31 | 2010-08-02 | 연속 세포 배양에서 목적 폴리펩티드 또는 바이러스를 생산하는 방법 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| KR20170077286A true KR20170077286A (ko) | 2017-07-05 |
| KR101885046B1 KR101885046B1 (ko) | 2018-08-02 |
Family
ID=42668436
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020177017512A Active KR101885046B1 (ko) | 2009-07-31 | 2010-08-02 | 연속 세포 배양에서 목적 폴리펩티드 또는 바이러스를 생산하는 방법 |
| KR1020127004996A Active KR101752789B1 (ko) | 2009-07-31 | 2010-08-02 | 연속 세포 배양에서 목적 폴리펩티드 또는 바이러스를 생산하는 방법 |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020127004996A Active KR101752789B1 (ko) | 2009-07-31 | 2010-08-02 | 연속 세포 배양에서 목적 폴리펩티드 또는 바이러스를 생산하는 방법 |
Country Status (22)
| Country | Link |
|---|---|
| US (4) | US8580554B2 (ko) |
| EP (2) | EP3480292A1 (ko) |
| JP (2) | JP5808742B2 (ko) |
| KR (2) | KR101885046B1 (ko) |
| CN (1) | CN102549142B (ko) |
| AU (1) | AU2010277490B2 (ko) |
| BR (1) | BR112012002137B8 (ko) |
| CA (1) | CA2769354A1 (ko) |
| CO (1) | CO6491119A2 (ko) |
| DK (1) | DK2459697T3 (ko) |
| EA (1) | EA022219B1 (ko) |
| ES (1) | ES2705206T3 (ko) |
| HR (1) | HRP20182165T1 (ko) |
| IN (1) | IN2012DN00899A (ko) |
| MX (2) | MX2012001274A (ko) |
| NZ (1) | NZ597742A (ko) |
| PL (1) | PL2459697T3 (ko) |
| PT (1) | PT2459697T (ko) |
| SG (1) | SG178176A1 (ko) |
| SI (1) | SI2459697T1 (ko) |
| TR (1) | TR201900213T4 (ko) |
| WO (1) | WO2011012725A1 (ko) |
Families Citing this family (24)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EA202090067A3 (ru) | 2010-07-08 | 2020-07-31 | Баксалта Инкорпорейтид | Способ получения рекомбинантного adamts13 в культуре клеток |
| WO2012098055A1 (en) * | 2011-01-17 | 2012-07-26 | F. Hoffmann-La Roche Ag | Separation apparatus and use thereof |
| CN103517919B (zh) | 2011-05-13 | 2016-11-16 | 欧克塔医药公司 | 在重组fviii的生产中提高真核细胞生产率的方法 |
| DK2837680T3 (da) | 2011-07-01 | 2020-04-27 | Amgen Inc | Mammaliacellekultur |
| CN203129697U (zh) | 2013-02-05 | 2013-08-14 | 客贝利(厦门)休闲用品有限公司 | 一种帐篷架杆 |
| LT2782997T (lt) | 2011-11-24 | 2018-05-25 | Genethon | Kintamo masto lentivirusinio vektoriaus gamybos sistema, suderinama su pramoniniu farmaciniu pritaikymu |
| US9574167B2 (en) | 2013-02-15 | 2017-02-21 | Pharyx, Inc. | Methods and apparatus for independent control of product and reactant concentrations |
| JP6608705B2 (ja) | 2013-03-08 | 2019-11-20 | ジェンザイム・コーポレーション | 治療用タンパク質の連続的な精製 |
| US9217168B2 (en) | 2013-03-14 | 2015-12-22 | Momenta Pharmaceuticals, Inc. | Methods of cell culture |
| WO2014146933A1 (en) | 2013-03-19 | 2014-09-25 | Cmc Biologics A/S | A method for producing a product (e.g. polypeptide) in a continuous cell culture fermentation process. |
| CA2920934C (en) * | 2013-08-30 | 2022-12-06 | Glaxosmithkline Biologicals S.A. | Large scale production of viruses in cell culture |
| US11390663B2 (en) | 2013-10-11 | 2022-07-19 | Regeneron Pharmaceuticals, Inc. | Metabolically optimized cell culture |
| TWI671312B (zh) | 2014-01-17 | 2019-09-11 | 美商健臻公司 | 無菌層析法及製法 |
| TWI709569B (zh) | 2014-01-17 | 2020-11-11 | 美商健臻公司 | 無菌層析樹脂及其用於製造方法的用途 |
| WO2015149144A2 (en) | 2014-04-01 | 2015-10-08 | Advantech Bioscience Farmacêutica Ltda. | Stabilization of factor viii without calcium as an excipient |
| WO2015149143A2 (en) | 2014-04-01 | 2015-10-08 | Advantech Bioscience Farmacêutica Ltda. | Stable factor viii formulations with low sugar-glycine |
| AU2015274238B2 (en) * | 2014-06-13 | 2020-11-05 | Csl Limited | Improved production of recombinant von Willebrand factor in a bioreactor |
| CN116083345A (zh) | 2016-01-26 | 2023-05-09 | 勃林格殷格翰国际公司 | 对连续流动搅拌釜反应器细胞培养系统的连接灌注 |
| EP3443064A1 (en) * | 2016-04-15 | 2019-02-20 | Boehringer Ingelheim International GmbH | Cell retention device and method |
| HRP20210163T1 (hr) * | 2016-12-21 | 2021-03-19 | F. Hoffmann - La Roche Ag | Kontrola rasta eukariotskih stanica |
| IL307155B2 (en) | 2018-05-01 | 2025-11-01 | Amgen Inc | Antibodies with modulated glycan profiles |
| WO2019239780A1 (ja) * | 2018-06-15 | 2019-12-19 | 富士フイルム株式会社 | 細胞培養方法、細胞培養装置、及び生産物の製造方法 |
| HK40046210A (en) | 2018-08-31 | 2021-10-29 | Genzyme Corporation | Sterile chromatography resin and use thereof in manufacturing processes |
| CN113355297A (zh) * | 2021-06-23 | 2021-09-07 | 吉林冠界生物技术有限公司 | 一种灌流式培养全悬浮mdck细胞生产重组禽流感病毒的方法 |
Family Cites Families (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5019512A (en) | 1989-03-17 | 1991-05-28 | Baxter International Inc. | Spin filter for removing substantially cell-free culture medium from suspension cell culture system |
| US5626734A (en) | 1995-08-18 | 1997-05-06 | University Technologies International, Inc. | Filter for perfusion cultures of animal cells and the like |
| BR0114374A (pt) | 2000-10-02 | 2003-12-30 | Novo Nordisk As | Preparação compreendendo uma pluralidade de polipeptìdeos de fator vii, métodos para a determinação do padrão de glicoforma de fator vii e de polipeptìdeos relacionados com fator vii, e para a produção da dita preparação, formulação farmacêutica, métodos para o tratamento de uma sìndrome responsiva a fator vii, para a prevenção de sangramento indesejado, para a prevenção de coagulação sanguìnea indesejada, e para prevenção de reações mediadas por fator de tecido, e, uso da preparação |
| IL161858A0 (en) * | 2001-11-28 | 2005-11-20 | Sandoz Ag | Cell culture process |
| TWI384069B (zh) | 2004-08-27 | 2013-02-01 | Pfizer Ireland Pharmaceuticals | 多胜肽之製法 |
| DK1902141T3 (da) | 2005-06-17 | 2012-05-07 | Baxter Int | ADAMTS 13-holdige sammensætninger med thrombolytisk virkning |
| CN102643777A (zh) * | 2006-01-04 | 2012-08-22 | 巴克斯特国际公司 | 无寡肽的细胞培养基 |
| DK2574677T3 (da) * | 2007-12-27 | 2017-11-06 | Baxalta GmbH | Fremgangsmåder til celledyrkning |
-
2010
- 2010-07-30 US US12/847,921 patent/US8580554B2/en active Active
- 2010-08-02 CA CA2769354A patent/CA2769354A1/en not_active Abandoned
- 2010-08-02 KR KR1020177017512A patent/KR101885046B1/ko active Active
- 2010-08-02 NZ NZ597742A patent/NZ597742A/en unknown
- 2010-08-02 EP EP18199855.0A patent/EP3480292A1/en not_active Withdrawn
- 2010-08-02 MX MX2012001274A patent/MX2012001274A/es active IP Right Grant
- 2010-08-02 HR HRP20182165TT patent/HRP20182165T1/hr unknown
- 2010-08-02 IN IN899DEN2012 patent/IN2012DN00899A/en unknown
- 2010-08-02 EA EA201270213A patent/EA022219B1/ru not_active IP Right Cessation
- 2010-08-02 PL PL10737916T patent/PL2459697T3/pl unknown
- 2010-08-02 AU AU2010277490A patent/AU2010277490B2/en active Active
- 2010-08-02 EP EP10737916.6A patent/EP2459697B1/en active Active
- 2010-08-02 TR TR2019/00213T patent/TR201900213T4/tr unknown
- 2010-08-02 DK DK10737916.6T patent/DK2459697T3/en active
- 2010-08-02 KR KR1020127004996A patent/KR101752789B1/ko active Active
- 2010-08-02 JP JP2012522190A patent/JP5808742B2/ja active Active
- 2010-08-02 PT PT10737916T patent/PT2459697T/pt unknown
- 2010-08-02 BR BR112012002137A patent/BR112012002137B8/pt active IP Right Grant
- 2010-08-02 ES ES10737916T patent/ES2705206T3/es active Active
- 2010-08-02 WO PCT/EP2010/061191 patent/WO2011012725A1/en not_active Ceased
- 2010-08-02 CN CN201080036754.2A patent/CN102549142B/zh active Active
- 2010-08-02 SG SG2012006599A patent/SG178176A1/en unknown
- 2010-08-02 SI SI201031835T patent/SI2459697T1/sl unknown
-
2012
- 2012-01-27 MX MX2014008515A patent/MX353610B/es unknown
- 2012-02-06 CO CO12020130A patent/CO6491119A2/es unknown
-
2013
- 2013-10-10 US US14/050,710 patent/US9650612B2/en active Active
-
2015
- 2015-01-14 JP JP2015004718A patent/JP6177260B2/ja active Active
-
2017
- 2017-04-12 US US15/486,181 patent/US10221400B2/en active Active
-
2019
- 2019-01-18 US US16/252,216 patent/US10745672B2/en active Active
Non-Patent Citations (4)
| Title |
|---|
| BIOTECHNOLOGY AND BIOENGINEERING, 1995, vol. 48, no. 3, pages 289-300.* * |
| BIOTECHNOLOGY AND BIOENGINEERING,2003, vol. 82, no. 7, pages 751-765.* * |
| JOURNAL OF BIOTECHNOLOGY, 1992, vol. 22, no. 1-2, pages 31-40.* * |
| JOURNAL OF BIOTECHNOLOGY, 1992, vol. 22, no. 1-2, pages 41-50.* * |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR101752789B1 (ko) | 연속 세포 배양에서 목적 폴리펩티드 또는 바이러스를 생산하는 방법 | |
| KR101143796B1 (ko) | 개선된 세포 배양 방법 | |
| Van der Velden-de Groot | Microcarrier technology, present status and perspective | |
| Thyden et al. | Recycling spent animal cell culture media using the thermally resistant microalga Chlorella sorokiniana | |
| JP2859679B2 (ja) | 新規細胞株 | |
| Hong et al. | Development of an alternating tangential flow (ATF) perfusion‐based transient gene expression (TGE) bioprocess for universal influenza vaccine | |
| HK40008436A (en) | Method of producing a polypeptide or virus of interest in a continuous cell culture | |
| Zhang | Approaches to optimizing animal cell culture process: substrate metabolism regulation and protein expression improvement | |
| HK1169439B (en) | Method of producing a polypeptide or virus of interest in a continuous cell culture | |
| HK1169439A (en) | Method of producing a polypeptide or virus of interest in a continuous cell culture | |
| CN118667752A (zh) | 一种用于生产人骨形态发生蛋白的细胞培养方法 | |
| HK40117190A (zh) | 一种用於生产人骨形态发生蛋白的细胞培养方法 | |
| Rodríguez et al. | Experimental factorial design for the study of some variables in roller bottles cell culture |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A107 | Divisional application of patent | ||
| A201 | Request for examination | ||
| PA0104 | Divisional application for international application |
Comment text: Divisional Application for International Patent Patent event code: PA01041R01D Patent event date: 20170626 Application number text: 1020127004996 Filing date: 20120227 |
|
| PA0201 | Request for examination | ||
| PG1501 | Laying open of application | ||
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
Comment text: Notification of reason for refusal Patent event date: 20170913 Patent event code: PE09021S01D |
|
| E701 | Decision to grant or registration of patent right | ||
| PE0701 | Decision of registration |
Patent event code: PE07011S01D Comment text: Decision to Grant Registration Patent event date: 20180528 |
|
| PR0701 | Registration of establishment |
Comment text: Registration of Establishment Patent event date: 20180727 Patent event code: PR07011E01D |
|
| PR1002 | Payment of registration fee |
Payment date: 20180727 End annual number: 3 Start annual number: 1 |
|
| PG1601 | Publication of registration | ||
| PR1001 | Payment of annual fee |
Payment date: 20210628 Start annual number: 4 End annual number: 4 |
|
| PR1001 | Payment of annual fee |
Payment date: 20220627 Start annual number: 5 End annual number: 5 |
|
| PR1001 | Payment of annual fee |
Payment date: 20240625 Start annual number: 7 End annual number: 7 |
|
| PR1001 | Payment of annual fee |
Payment date: 20250630 Start annual number: 8 End annual number: 8 |