KR20170108005A - 디오스민의 제조방법 - Google Patents
디오스민의 제조방법 Download PDFInfo
- Publication number
- KR20170108005A KR20170108005A KR1020177020436A KR20177020436A KR20170108005A KR 20170108005 A KR20170108005 A KR 20170108005A KR 1020177020436 A KR1020177020436 A KR 1020177020436A KR 20177020436 A KR20177020436 A KR 20177020436A KR 20170108005 A KR20170108005 A KR 20170108005A
- Authority
- KR
- South Korea
- Prior art keywords
- sodium
- potassium
- diosmin
- hydroxide
- hesperidin
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07H—SUGARS; DERIVATIVES THEREOF; NUCLEOSIDES; NUCLEOTIDES; NUCLEIC ACIDS
- C07H1/00—Processes for the preparation of sugar derivatives
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07H—SUGARS; DERIVATIVES THEREOF; NUCLEOSIDES; NUCLEOTIDES; NUCLEIC ACIDS
- C07H17/00—Compounds containing heterocyclic radicals directly attached to hetero atoms of saccharide radicals
- C07H17/04—Heterocyclic radicals containing only oxygen as ring hetero atoms
- C07H17/06—Benzopyran radicals
- C07H17/065—Benzo[b]pyrans
- C07H17/07—Benzo[b]pyran-4-ones
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/14—Vasoprotectives; Antihaemorrhoidals; Drugs for varicose therapy; Capillary stabilisers
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- General Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Biochemistry (AREA)
- Molecular Biology (AREA)
- Genetics & Genomics (AREA)
- Biotechnology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Veterinary Medicine (AREA)
- General Chemical & Material Sciences (AREA)
- Cardiology (AREA)
- Animal Behavior & Ethology (AREA)
- Pharmacology & Pharmacy (AREA)
- Public Health (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Heart & Thoracic Surgery (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Medicinal Chemistry (AREA)
- Vascular Medicine (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Nitrogen Condensed Heterocyclic Rings (AREA)
- Saccharide Compounds (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Cephalosporin Compounds (AREA)
Abstract
Description
Claims (17)
- 헤스페리딘(hesperidin)으로부터 디오스민(diosmin)의 제조 방법으로서,
a) 헤스페리딘을 C2-C4 카복실산의 무수물로 아실화하는 단계;
b) 단계 a)에서 얻어진 혼합물을, 수성 매질 중에서, 요오드 및 브로민으로부터 얻어진 할로겐으로 처리하는 단계;
c) 3.5 내지 6.5 범위의 pH값에 도달하도록 단계 b)에서 얻어진 혼합물을 무기 염기로 처리하는 단계;
d) 단계 c)에서 얻어진 아실화된 디오스민을 무기 염기에 의한 처리에 의해 탈아실화하는 단계를 포함하되,
상기 방법 전체를 통해서 유기 용매가 첨가되지 않는, 헤스페리딘으로부터 디오스민의 제조 방법. - 제1항에 있어서, 단계 a)에서 아세트산나트륨 및 아세트산칼륨으로부터 선택된 촉매가 이용되는, 헤스페리딘으로부터 디오스민의 제조 방법.
- 제1항 또는 제2항에 있어서, 단계 a)의 상기 C2-C4 카복실산의 상기 무수물은 아세트산 무수물인, 헤스페리딘으로부터 디오스민의 제조 방법.
- 제1항 내지 제3항 중 어느 한 항에 있어서, 단계 b)는,
a) 화학량론적 양의 상기 할로겐을 이용해서, 또는
b) 화학량론적 양의 할로겐화물 및 화학량론적 양의 산화제를 이용해서, 또는
c) 촉매량의 상기 할로겐 및 화학량론적 양의 산화제를 이용해서
수행되는, 헤스페리딘으로부터 디오스민의 제조 방법. - 제1항 내지 제3항 중 어느 한 항에 있어서, 단계 b)는 촉매량의 할로겐화물 및 화학량론적 양의 산화제를 이용해서 수행되는, 헤스페리딘으로부터 디오스민의 제조 방법.
- 제4항 또는 제5항에 있어서, 상기 산화제는 과산화수소, 과탄산나트륨, 과탄산칼륨, 과붕산나트륨, 과붕산칼륨, 과망간산나트륨, 과망간산칼륨, 중크롬산나트륨, 중크롬산칼륨, 및 이들의 수화물로 이루어진 군으로부터 선택되는, 헤스페리딘으로부터 디오스민의 제조 방법.
- 제6항에 있어서, 상기 산화제는 과산화수소인, 헤스페리딘으로부터 디오스민의 제조 방법.
- 제6항에 있어서, 상기 산화제는 과탄산나트륨, 과탄산칼륨, 과붕산나트륨, 과붕산칼륨 및 이들의 수화물로부터 선택되는, 헤스페리딘으로부터 디오스민의 제조 방법.
- 제4항 내지 제8항 중 어느 한 항에 있어서, 상기 요오드화물은 요오드화나트륨, 요오드화칼륨, 요오드화칼슘, 요오드화마그네슘 및 이들의 혼합물로부터 선택되는, 헤스페리딘으로부터 디오스민의 제조 방법.
- 제9항에 있어서, 상기 요오드화물은 요오드화나트륨, 요오드화칼륨 및 이들의 혼합물로부터 선택되는, 헤스페리딘으로부터 디오스민의 제조 방법.
- 제4항 내지 제8항 중 어느 한 항에 있어서, 상기 브로민화물은 브로민화나트륨, 브로민화칼륨, 브로민화칼슘, 브로민화마그네슘, 및 이들의 혼합물로부터 선택되는, 헤스페리딘으로부터 디오스민의 제조 방법.
- 제1항 내지 제11항 중 어느 한 항에 있어서, 단계 c)의 상기 무기 염기는 수산화나트륨, 수산화칼륨, 수산화칼슘, 수산화마그네슘, 탄산나트륨, 탄산칼륨, 탄산칼슘, 탄산마그네슘, 이들의 수화물, 및 이들의 혼합물로부터 선택되는, 헤스페리딘으로부터 디오스민의 제조 방법.
- 제12항에 있어서, 상기 무기 염기는 수산화나트륨, 수산화칼륨 및 이들의 혼합물로부터 선택되는, 헤스페리딘으로부터 디오스민의 제조 방법.
- 제1항 내지 제13항 중 어느 한 항에 있어서, 단계 c)에서 상기 pH값은 4.5 내지 5.5의 범위인, 헤스페리딘으로부터 디오스민의 제조 방법.
- 제1항 내지 제14항 중 어느 한 항에 있어서, 단계 d) 후에 얻어진 상기 디오스민은 수성 매질 중에서 재결정화되는, 헤스페리딘으로부터 디오스민의 제조 방법.
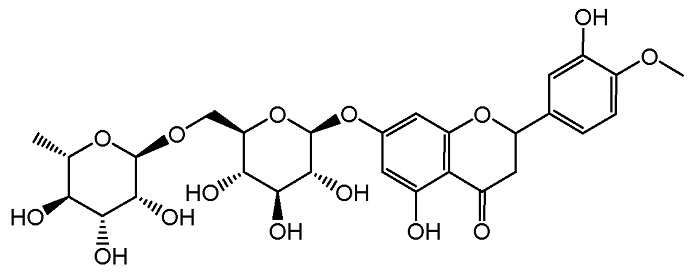
- 제1항 내지 제15항 중 어느 한 항의 방법에 의해 얻어질 수 있는 디오스민.
- 제16항에 있어서, 1000 ppm 미만의 할로겐 함량을 지니고, 그리고 잔류 유기 용매가 없는, 디오스민.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020207031757A KR102309525B1 (ko) | 2015-02-03 | 2016-02-02 | 디오스민의 제조방법 |
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP15153537.4A EP3053930A1 (en) | 2015-02-03 | 2015-02-03 | Process for the preparation of diosmin |
| EP15153537.4 | 2015-02-03 | ||
| PCT/EP2016/052165 WO2016124585A1 (en) | 2015-02-03 | 2016-02-02 | Process for the preparation of diosmin |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020207031757A Division KR102309525B1 (ko) | 2015-02-03 | 2016-02-02 | 디오스민의 제조방법 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| KR20170108005A true KR20170108005A (ko) | 2017-09-26 |
| KR102216702B1 KR102216702B1 (ko) | 2021-02-18 |
Family
ID=52477555
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020207031757A Active KR102309525B1 (ko) | 2015-02-03 | 2016-02-02 | 디오스민의 제조방법 |
| KR1020177020436A Active KR102216702B1 (ko) | 2015-02-03 | 2016-02-02 | 디오스민의 제조방법 |
Family Applications Before (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020207031757A Active KR102309525B1 (ko) | 2015-02-03 | 2016-02-02 | 디오스민의 제조방법 |
Country Status (24)
| Country | Link |
|---|---|
| US (2) | US10287310B2 (ko) |
| EP (2) | EP3053930A1 (ko) |
| KR (2) | KR102309525B1 (ko) |
| CN (1) | CN107108675B (ko) |
| AR (1) | AR103583A1 (ko) |
| BR (1) | BR112017016430B1 (ko) |
| CA (1) | CA2974860A1 (ko) |
| EA (1) | EA032402B1 (ko) |
| ES (1) | ES2803213T3 (ko) |
| HR (1) | HRP20201102T1 (ko) |
| HU (1) | HUE049997T2 (ko) |
| LT (1) | LT3253772T (ko) |
| MA (1) | MA40703B1 (ko) |
| MX (1) | MX372637B (ko) |
| MY (1) | MY183290A (ko) |
| PH (1) | PH12017501244A1 (ko) |
| PL (1) | PL3253772T3 (ko) |
| PT (1) | PT3253772T (ko) |
| RS (1) | RS60494B1 (ko) |
| SI (1) | SI3253772T1 (ko) |
| TW (1) | TWI715551B (ko) |
| UA (1) | UA123046C2 (ko) |
| UY (1) | UY36549A (ko) |
| WO (1) | WO2016124585A1 (ko) |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN106967138A (zh) * | 2017-04-19 | 2017-07-21 | 成都百特万合医药科技有限公司 | 基于二甲亚砜体系的地奥司明生产方法 |
| AR122887A1 (es) | 2020-07-09 | 2022-10-12 | Servier Lab | Procedimiento de preparación de la diosmina |
| AR126366A1 (es) | 2021-07-06 | 2023-10-11 | Servier Lab | Procedimiento para la preparación de diosmina y de fracción flavonoica |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4078137A (en) * | 1975-05-16 | 1978-03-07 | Hommel Aktiengesellschaft | Process for manufacturing a flavone derivative |
| WO2000011009A2 (fr) * | 1998-08-19 | 2000-03-02 | Innokem, S.A.R.L. | Procede de fabrication industrielle de diosmine a partir de l'hesperidine par reaction avec de l'iode et de la pyridine |
Family Cites Families (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| ES440427A1 (es) | 1975-08-25 | 1977-03-01 | Espanola Prod Quimicos | Un procedimiento de obtencion de flavonas. |
| DE2740950A1 (de) | 1977-09-10 | 1979-03-22 | Merck Patent Gmbh | Verfahren zur herstellung von flavonen |
| IT1150612B (it) | 1982-03-04 | 1986-12-17 | Prophin Lab Spa | Procedimento per la preparazione del 1',5,7-tridrossi-4'-metossiflavone-7-rutinoside (diosmina) |
| DE3514123C2 (de) | 1985-04-19 | 1994-12-08 | Kabelmetal Ag | Verfahren zur Herstellung von Durchlaufkokillen für Stranggießmaschinen |
| IT1191620B (it) | 1985-05-30 | 1988-03-23 | Edmond Pharma Srl | Procedimento per la preparazione della diosmina |
| GB9717629D0 (en) * | 1997-08-21 | 1997-10-22 | Johnson Matthey Plc | Removal of residual organic solvents |
| CN102653549A (zh) * | 2011-12-28 | 2012-09-05 | 长沙富能生物技术有限公司 | 一种符合ep7版质量标准的地奥司明原料药合成方法 |
-
2015
- 2015-02-03 EP EP15153537.4A patent/EP3053930A1/en not_active Withdrawn
-
2016
- 2016-01-19 TW TW105101530A patent/TWI715551B/zh active
- 2016-02-02 WO PCT/EP2016/052165 patent/WO2016124585A1/en not_active Ceased
- 2016-02-02 CN CN201680006218.5A patent/CN107108675B/zh active Active
- 2016-02-02 EP EP16702557.6A patent/EP3253772B8/en active Active
- 2016-02-02 ES ES16702557T patent/ES2803213T3/es active Active
- 2016-02-02 HU HUE16702557A patent/HUE049997T2/hu unknown
- 2016-02-02 US US15/544,174 patent/US10287310B2/en active Active
- 2016-02-02 CA CA2974860A patent/CA2974860A1/en active Pending
- 2016-02-02 MA MA40703A patent/MA40703B1/fr unknown
- 2016-02-02 KR KR1020207031757A patent/KR102309525B1/ko active Active
- 2016-02-02 MY MYPI2017702825A patent/MY183290A/en unknown
- 2016-02-02 BR BR112017016430-2A patent/BR112017016430B1/pt active IP Right Grant
- 2016-02-02 SI SI201630841T patent/SI3253772T1/sl unknown
- 2016-02-02 PL PL16702557T patent/PL3253772T3/pl unknown
- 2016-02-02 KR KR1020177020436A patent/KR102216702B1/ko active Active
- 2016-02-02 AR ARP160100287A patent/AR103583A1/es active IP Right Grant
- 2016-02-02 LT LTEP16702557.6T patent/LT3253772T/lt unknown
- 2016-02-02 UA UAA201707326A patent/UA123046C2/uk unknown
- 2016-02-02 RS RS20200814A patent/RS60494B1/sr unknown
- 2016-02-02 MX MX2017010026A patent/MX372637B/es active IP Right Grant
- 2016-02-02 PT PT167025576T patent/PT3253772T/pt unknown
- 2016-02-02 EA EA201791280A patent/EA032402B1/ru not_active IP Right Cessation
- 2016-02-02 HR HRP20201102TT patent/HRP20201102T1/hr unknown
- 2016-02-03 UY UY0001036549A patent/UY36549A/es active IP Right Grant
-
2017
- 2017-07-05 PH PH12017501244A patent/PH12017501244A1/en unknown
-
2019
- 2019-04-02 US US16/373,118 patent/US10711025B2/en active Active
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4078137A (en) * | 1975-05-16 | 1978-03-07 | Hommel Aktiengesellschaft | Process for manufacturing a flavone derivative |
| WO2000011009A2 (fr) * | 1998-08-19 | 2000-03-02 | Innokem, S.A.R.L. | Procede de fabrication industrielle de diosmine a partir de l'hesperidine par reaction avec de l'iode et de la pyridine |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US8283464B2 (en) | Process for synthesizing and purifying sucralose | |
| WO2010092592A2 (en) | Process for the preparation of diosmin | |
| US10711025B2 (en) | Process for the preparation of diosmin | |
| CN103880903A (zh) | 一种泰乐菌素类大环内酯及其衍生物的制备方法 | |
| NO340888B1 (no) | Framgangsmåte for framstilling av ioheksol | |
| HK1241885A1 (en) | Process for the preparation of diosmin | |
| HK1241885B (zh) | 制备地奥司明的方法 | |
| JP5197106B2 (ja) | ハロゲン化フタル酸化合物の製造法 | |
| CN101918349B (zh) | 制备双醋瑞因的方法 | |
| CN107011146A (zh) | 一种艾地苯醌的制备方法 | |
| EP1961730A1 (en) | Method for producing polymerizable hydroxydiamantyl ester compound | |
| CN118561739A (zh) | 一种5-氨基酮戊酸盐酸盐中间体的制备方法 | |
| EP2522651B1 (en) | Process for the purification of 2,6-diisopropyl phenol | |
| CA2856324C (en) | Process for the preparation of drospirenone | |
| JP2005298459A (ja) | 3−ヒドロキシカルボン酸類およびこれに対応するラクトン類の製造法 | |
| CN104447887A (zh) | 甲基-5-脱氧-l-阿拉伯糖的制备方法 | |
| JPH0710800A (ja) | 2,3−ジメトキシ−5−メチル−1,4−ベンゾキノンの製造法 | |
| ZA200400842B (en) | Process for the preparation of 5-formylphtalide. |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0105 | International application |
St.27 status event code: A-0-1-A10-A15-nap-PA0105 |
|
| PG1501 | Laying open of application |
St.27 status event code: A-1-1-Q10-Q12-nap-PG1501 |
|
| R17-X000 | Change to representative recorded |
St.27 status event code: A-3-3-R10-R17-oth-X000 |
|
| A201 | Request for examination | ||
| AMND | Amendment | ||
| P11-X000 | Amendment of application requested |
St.27 status event code: A-2-2-P10-P11-nap-X000 |
|
| P13-X000 | Application amended |
St.27 status event code: A-2-2-P10-P13-nap-X000 |
|
| PA0201 | Request for examination |
St.27 status event code: A-1-2-D10-D11-exm-PA0201 |
|
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
St.27 status event code: A-1-2-D10-D21-exm-PE0902 |
|
| AMND | Amendment | ||
| E13-X000 | Pre-grant limitation requested |
St.27 status event code: A-2-3-E10-E13-lim-X000 |
|
| P11-X000 | Amendment of application requested |
St.27 status event code: A-2-2-P10-P11-nap-X000 |
|
| P13-X000 | Application amended |
St.27 status event code: A-2-2-P10-P13-nap-X000 |
|
| R17-X000 | Change to representative recorded |
St.27 status event code: A-3-3-R10-R17-oth-X000 |
|
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
St.27 status event code: A-1-2-D10-D21-exm-PE0902 |
|
| E601 | Decision to refuse application | ||
| PE0601 | Decision on rejection of patent |
St.27 status event code: N-2-6-B10-B15-exm-PE0601 |
|
| X091 | Application refused [patent] | ||
| A107 | Divisional application of patent | ||
| AMND | Amendment | ||
| E13-X000 | Pre-grant limitation requested |
St.27 status event code: A-2-3-E10-E13-lim-X000 |
|
| P11-X000 | Amendment of application requested |
St.27 status event code: A-2-2-P10-P11-nap-X000 |
|
| P13-X000 | Application amended |
St.27 status event code: A-2-2-P10-P13-nap-X000 |
|
| PA0104 | Divisional application for international application |
St.27 status event code: A-0-1-A10-A18-div-PA0104 St.27 status event code: A-0-1-A10-A16-div-PA0104 |
|
| PX0901 | Re-examination |
St.27 status event code: A-2-3-E10-E12-rex-PX0901 |
|
| PX0701 | Decision of registration after re-examination |
St.27 status event code: A-3-4-F10-F13-rex-PX0701 |
|
| X701 | Decision to grant (after re-examination) | ||
| N231 | Notification of change of applicant | ||
| PN2301 | Change of applicant |
St.27 status event code: A-3-3-R10-R13-asn-PN2301 St.27 status event code: A-3-3-R10-R11-asn-PN2301 |
|
| P11-X000 | Amendment of application requested |
St.27 status event code: A-2-2-P10-P11-nap-X000 |
|
| P13-X000 | Application amended |
St.27 status event code: A-2-2-P10-P13-nap-X000 |
|
| GRNT | Written decision to grant | ||
| PR0701 | Registration of establishment |
St.27 status event code: A-2-4-F10-F11-exm-PR0701 |
|
| PR1002 | Payment of registration fee |
St.27 status event code: A-2-2-U10-U12-oth-PR1002 Fee payment year number: 1 |
|
| PG1601 | Publication of registration |
St.27 status event code: A-4-4-Q10-Q13-nap-PG1601 |
|
| R18-X000 | Changes to party contact information recorded |
St.27 status event code: A-5-5-R10-R18-oth-X000 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 4 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 5 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 6 |
|
| U11 | Full renewal or maintenance fee paid |
Free format text: ST27 STATUS EVENT CODE: A-4-4-U10-U11-OTH-PR1001 (AS PROVIDED BY THE NATIONAL OFFICE) Year of fee payment: 6 |