WO1999061702A1 - Procede de floculation pour la fabrication d'une feuille de papier - Google Patents
Procede de floculation pour la fabrication d'une feuille de papier Download PDFInfo
- Publication number
- WO1999061702A1 WO1999061702A1 PCT/FR1999/001209 FR9901209W WO9961702A1 WO 1999061702 A1 WO1999061702 A1 WO 1999061702A1 FR 9901209 W FR9901209 W FR 9901209W WO 9961702 A1 WO9961702 A1 WO 9961702A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- polymer
- paper
- cardboard
- sheet
- monomers
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- D—TEXTILES; PAPER
- D21—PAPER-MAKING; PRODUCTION OF CELLULOSE
- D21H—PULP COMPOSITIONS; PREPARATION THEREOF NOT COVERED BY SUBCLASSES D21C OR D21D; IMPREGNATING OR COATING OF PAPER; TREATMENT OF FINISHED PAPER NOT COVERED BY CLASS B31 OR SUBCLASS D21G; PAPER NOT OTHERWISE PROVIDED FOR
- D21H21/00—Non-fibrous material added to the pulp, characterised by its function, form or properties; Paper-impregnating or coating material, characterised by its function, form or properties
- D21H21/06—Paper forming aids
- D21H21/10—Retention agents or drainage improvers
-
- D—TEXTILES; PAPER
- D21—PAPER-MAKING; PRODUCTION OF CELLULOSE
- D21H—PULP COMPOSITIONS; PREPARATION THEREOF NOT COVERED BY SUBCLASSES D21C OR D21D; IMPREGNATING OR COATING OF PAPER; TREATMENT OF FINISHED PAPER NOT COVERED BY CLASS B31 OR SUBCLASS D21G; PAPER NOT OTHERWISE PROVIDED FOR
- D21H17/00—Non-fibrous material added to the pulp, characterised by its constitution; Paper-impregnating material characterised by its constitution
- D21H17/20—Macromolecular organic compounds
- D21H17/33—Synthetic macromolecular compounds
- D21H17/34—Synthetic macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds
- D21H17/37—Polymers of unsaturated acids or derivatives thereof, e.g. polyacrylates
-
- D—TEXTILES; PAPER
- D21—PAPER-MAKING; PRODUCTION OF CELLULOSE
- D21H—PULP COMPOSITIONS; PREPARATION THEREOF NOT COVERED BY SUBCLASSES D21C OR D21D; IMPREGNATING OR COATING OF PAPER; TREATMENT OF FINISHED PAPER NOT COVERED BY CLASS B31 OR SUBCLASS D21G; PAPER NOT OTHERWISE PROVIDED FOR
- D21H17/00—Non-fibrous material added to the pulp, characterised by its constitution; Paper-impregnating material characterised by its constitution
- D21H17/20—Macromolecular organic compounds
- D21H17/33—Synthetic macromolecular compounds
- D21H17/34—Synthetic macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds
- D21H17/41—Synthetic macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds containing ionic groups
- D21H17/42—Synthetic macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds containing ionic groups anionic
- D21H17/43—Carboxyl groups or derivatives thereof
-
- D—TEXTILES; PAPER
- D21—PAPER-MAKING; PRODUCTION OF CELLULOSE
- D21H—PULP COMPOSITIONS; PREPARATION THEREOF NOT COVERED BY SUBCLASSES D21C OR D21D; IMPREGNATING OR COATING OF PAPER; TREATMENT OF FINISHED PAPER NOT COVERED BY CLASS B31 OR SUBCLASS D21G; PAPER NOT OTHERWISE PROVIDED FOR
- D21H17/00—Non-fibrous material added to the pulp, characterised by its constitution; Paper-impregnating material characterised by its constitution
- D21H17/20—Macromolecular organic compounds
- D21H17/33—Synthetic macromolecular compounds
- D21H17/34—Synthetic macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds
- D21H17/41—Synthetic macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds containing ionic groups
- D21H17/44—Synthetic macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds containing ionic groups cationic
- D21H17/45—Nitrogen-containing groups
- D21H17/455—Nitrogen-containing groups comprising tertiary amine or being at least partially quaternised
Definitions
- the present invention relates to the field of synthetic polymers obtained from water-soluble monomers, or mixtures of such monomers, and to their application
- Goren's polymers could be used as coagulants, and especially for coagulating very fine suspended solids.
- European patent EP 0 201 237 describes a flocculation process in which a polymeric material is added to water to form an aqueous composition, and is used to flocculate the solid materials in suspension in an aqueous suspension,
- Said polymer consisting of a high molecular weight polymer, which is subjected to shearing, this shearing being carried out before or during flocculation and the polymer having to respond to certain intrinsic properties which are indicated in this patent.
- the polymer is a high molecular weight polymer, formed from water-soluble monomers or a mixture of such monomers, and the polymer is subjected to shearing.
- the process described in this patent is characterized in that the shearing can be carried out before or during flocculation.
- Patent EP 0 201 237 also indicates that the polymer used comprises a crosslinked polymer swellable with water which it is possible to shear up to an intrinsic viscosity of at least 4 dl / g. It is also indicated that the aqueous composition containing the polymer material can be a stable and homogeneous composition, the shearing then causing an increase in the intrinsic viscosity of at least 1 dl / g.
- stable and homogeneous designates a polymer composition which is stable when the polymer is in complete equilibrium with water, that is to say that it has reached its ultimate degree of dissolution or swelling.
- the composition is moreover homogeneous in the sense that the polymer remains uniformly dispersed throughout the composition, without having a tendency to precipitate after a few days.
- This document describes in particular numerous applications for the treatment of water, which is obviously the quite main application which is targeted, and the treatment of coal ore.
- the document also indicates that the shearing on the production line, when the flocculating suspension approaches a centrifuge, a filter press, or a belt press, or another step of removing water. It is also noted that the shear can be applied during a step of removing the water which is carried out under a certain shear, preferably in a centrifuge or else in a filter press or in a belt press.
- the present invention relates to a significant improvement in this process and in the corresponding flocculating agents, with surprising advantages in the specific application for the manufacture of a sheet of paper, cardboard or the like, and by operating under specific conditions which will be described below.
- a cross-linked polymer or copolymer formed from a single flocculating agent intended to significantly improve the retention, the formation, the drainage, and other properties of the sheet of paper or cardboard thus obtained, is used.
- suitable water-soluble monomers or mixtures of such monomers are used.
- the method according to the invention is characterized in that the crosslinked polymer is sheared before the introduction or injection into the suspension to be flocculated.
- the suspension containing the polymer is therefore not sheared.
- the monomers can be nonionic, but generally at least part of the monomers used for forming the polymer is ionic.
- the monomers are usually monoethylenically unsaturated monomers, sometimes allylic monomers, but generally vinyl monomers. They are generally acrylic or metacrylic monomers.
- Suitable nonionic monomers are acrylamide, methacrylamide, N-vinylmethylacetamide or N-vinylformamide, vinyl acetate, vinylpyrrolidone, methyl methacrylate or other acrylic esters, or other unsaturated esters ethylenic, or other vinyl monomers insoluble in water such as styrene or acrylonitrile.
- Suitable anionic monomers are, for example, sodium acrylate, sodium methacrylate, sodium itaconate, 2-acrylamido 2-methylpropane sulfonate (AMPS), sulfopropylacrylates or sulfopropylmethacrylates, or other soluble forms in l water of these polymerizable sulfonic or carboxylic acids.
- AMPS 2-acrylamido 2-methylpropane sulfonate
- a sodium vinyl sulfonate, or an allylsulfonate, or a sulfomethylated acrylamide may be used.
- Suitable cationic monomers are dialkylaminoalkyl acrylates and methacrylates, in particular dialkylaminoethyl acrylate, as well as their salts acidified or quaternized by means known to those skilled in the art, such as benzyl chloride, methyl chloride, aryl, alkyl chlorides, dimethylsulfate, and also the dialkylaminoalkylalkylacrylamides or -methacrylamides, as well as their acidified or quaternized salts in a known manner, for example methacrylamido-propyl trimethyl ammonium chloride (MAPTAC) and Mannich products such as quaternized dialkylaminomethylacrylamides.
- the alkyl groups in question are generally C 1 -C 4 alkyl groups.
- the monomers may contain hydrophobic groups, as for example described in patent EP 0 172 723, and it will be possible in certain cases to prefer monomers of allyl ether.
- Crosslinking can be carried out during or after the polymerization, for example by reaction of two polymers soluble with counter ions, or by reaction with formaldehyde or a polyvalent metal compound. Often, crosslinking takes place during polymerization by the addition of a crosslinking agent, and this solution will be much preferred according to the invention. These crosslinking polymerization processes are known.
- Cross-linking agents which can be incorporated include ionic cross-linking agents such as polyvalent metal salts, formaldehyde, glyoxal, or, and preferably, covalent cross-linking agents which will copolymerize with the monomers, preferably diethylenically unsaturated monomers (such as the family of diacrylate esters such as polyethylene glycol diacrylates PEG) or polyethylene, of the type conventionally used for the crosslinking of water-soluble polymers, and in particular methylenebisacrylamide (MBA) or any of the other known acrylic crosslinking agents.
- ionic cross-linking agents such as polyvalent metal salts, formaldehyde, glyoxal, or, and preferably, covalent cross-linking agents which will copolymerize with the monomers, preferably diethylenically unsaturated monomers (such as the family of diacrylate esters such as polyethylene glycol diacrylates PEG) or polyethylene, of the type conventionally used for the crosslink
- the quantity of crosslinking agents, and in particular methylenebisacrylamide (MBA) which can be incorporated according to the invention is generally of the order of 5 to 100, preferably 5 to 40, and according to the best mode, around 20 ppm of crosslinking agent / active material (polymer).
- the polymers which can be used according to the invention can be prepared by a method of polymerization in aqueous solution at low concentration, but the invention relates above all to an emulsion polymerization in reverse phase, that is to say an emulsion polymerization water- in-oil.
- Retention agent systems are also known in the prior art for the manufacture of a sheet of paper, cardboard or the like, which consist of a combination of two retention agents, usually a primary retention agent and a secondary retention agent. It is therefore a system qualified as “dual”.
- Cross-linked flocculating agents such as those described for example in patent EP 0 202 780 are also known in the prior art for treating water, essentially and secondarily paper. It should be noted that it uses a crosslinked product, which is added to the suspension to be flocculated, the flocs then being sheared in the papermaking process, that is to say sheared in and at the same time as the pulp. to paper. The flocs are thus transformed into smaller and shear-resistant flocs, therefore more robust. This document does not therefore suggest and describe any shearing of the polymer itself before introduction into the suspension to be flocculated.
- flocs of a fairly large size are thus formed between the flocculating agent and the fibrous mass of pulp, which are then sheared in order to form flocs which are indicated in the documents. mentioned that they are smaller and more robust.
- the prior art systems of the dual system type require the use of two retention components, and in particular the use of bentonite, which is a very heavy product to use in industry, if only only by the environmental problems it causes. This bentonite is necessary to bring about the reassembly of the sheared flocs, otherwise they do not hold on the drip cloth or paper drainage.
- Such systems can be described as "microparticles", and they include at least two retention agents.
- a single retention agent is used, preferably in reverse water-in-oil emulsion, crosslinked, sheared before its injection, and which leads directly to microflocs without passing through the shearing of larger flocs. and involving the fibrous mass.
- the Applicant considers in fact that it occurs, as a result of the strong shearing carried out on the polymer itself before its injection into the fibrous mass of dough, directly microflocculation, which is therefore a different (and unexpected) process from reducing the size of large flocs (and involving the fibrous mass) into smaller and more robust flocs, and which leads to unforeseen improvements in sheet properties of paper or cardboard.
- this “microflocculation” also avoids the presence of bentonite or of another “dual” second retention agent.
- Reverse emulsion polymerization is very well known to those skilled in the art.
- the injection or introduction of the crosslinked and previously sheared polymer according to the invention is carried out in paper pulp (or fibrous mass to be flocculated) more or less diluted according to the practice of a person skilled in the art, and generally in paper pulp diluted or "thin stock", ie a pulp diluted to about 0.7% - 1.5% of solid materials such as cellulose fibers, any fillers, various usual additives for papermaking.
- part of the crosslinked polymer will be introduced, and sheared according to the invention, at the stage of preparing the thick stock or "thick stock” at approx. 5% or more solids, or even in the preparation of the thick dough.
- an inverse emulsion of the polymer or else the powder obtained from the emulsion by a known drying technique, such as for example by spray drying or “spray-drying”, precipitation with solvent or agglomeration (PEG) and grinding (cf.
- This variant is very advantageous since the dried product behaves substantially, according to the invention, like the emulsion, and this variant therefore provides a solution for using dry products having the advantages of an emulsion, that it is not always possible to prepare by direct polymerization in aqueous phase in gel or in solution.
- the inverse emulsion of the crosslinked polymer is preferably used, or else the crosslinked polymer in solution as obtained by redissolving a powder, as described above, with of course shearing prior to injection. in the dough.
- a comparative example is shown below showing that, if the shearing of the fibrous mass (ie after the addition of the polymer) is carried out in a paper application, no retention is obtained. Without wishing to be limited by any theory, the Applicant considers that this is due to the fact that the cationic charge is not released.
- shear tests can be carried out in the laboratory, at a concentration of the order of 3 - 5 to 10 - 15 g of active material (ie the polymer) / liter, preferably between 5 and 10 g / l, in a material called Ultra Turrax, for example at 10,000 rpm or in a household mixer of the Moulinex type, substantially at same order of magnitude of rotation speed, for a duration which may be between 15 - 30 seconds and 2 - 5 minutes.
- active material ie the polymer
- Ultra Turrax for example at 10,000 rpm or in a household mixer of the Moulinex type
- a person skilled in the art will naturally be able to envisage all the possible equipment making it possible to effect a high shear of the polymer emulsion, diluted to an appropriate value as will be described below, without being limited to the examples above.
- a normal dosage of the agent according to the invention will be such that it leads to approximately 100 to 500 g of active material (polymer) per ton of fibrous material to be treated.
- an ionic recovery (IR as defined in patent EP 0 201 237) from 40 to 50 or which can reach at least 60 or 70%. and even more, up to values greater than or much greater than 100.
- system according to the invention is not expensive, and it therefore combines all the advantages of the systems of linear or crosslinked products unique to shear flocs and “dual” systems with two retention agents and also flock shear.
- Example 1, 2 or 3 The manufacturing examples of the polymer are followed by a Table ("example 1, 2 or 3") indicating the characteristics of the polymer obtained, by a Table (numbered N ° 1, N ° 2 and N ° 3 with the reference of the produced in the preceding tables) in two parts indicating on the one hand the conditions of application test and on the other hand the results of retention, draining and formation, and other similar characteristics.
- RET type of addition of the emulsion solution according to the invention, sheared before injection
- DOS dosage of the retention agent according to the invention
- ChM mineral load% / dry paste
- G80 grammage 80 g
- Weight leaf weight, in grams
- the emulsion is then incorporated with its inverting surfactant in order to subsequently release the polymer in the aqueous phase.
- MBA methylenebis acrylamide, crosslinking agent
- Ionic regain RI (X-Y) / Y x 100 with X: ionicity after shear in meq / g.
- Pulp used mixture of 70% KF bleached hardwood kraft
- G grammage ex.
- G80 grammage of 80 g / m2
- Figure 1 shows the histogram of the first pass retention figures corresponding to Table 1
- Figure 2 shows the histogram of ash retention figures corresponding to Table 1
- Figure 3 shows the histogram of the white water turbidity figures corresponding to table 1
- Figure 4 represents the histogram of the figures of turbidity of white water after 30 minutes of rest corresponding to table 1
- Variant according to the invention (overdose, approx. 1 kg / t)
- the invention makes it possible to completely eliminate the shearing operation of the crosslinked polymer emulsion before introduction into the fibrous mass, provided that the polymer is overdosed.
- we then find, without shearing, the effects of sheared crosslinked emulsions (cf. nonlimiting example of overdose at 1000 g / t, which is completely unusual in this industry).
- This choice can have advantages in certain applications; for example, this avoids the shearing time and the corresponding material, which are two important factors for the end user, and it is possible to increase the drainage and therefore the productivity because it then becomes possible to increase the speed. of the machine.
- the constituents of the organic phase of the emulsion to be synthesized are mixed at room temperature.
- phase of the emulsion to be prepared is prepared by mixing:
- the contents of B are mixed in A with stirring. After mixing the phases, the emulsion is sheared in the mixer for 1 minute to create the reverse emulsion. The emulsion is then degassed by bubbling nitrogen then after 20 minutes, the gradual addition of metabisulfite leads to initiation and then polymerization.
- the emulsion is then incorporated with its inverting surfactant in order to subsequently release the polymer in the aqueous phase.
- MBA methylenebis acrylamide, crosslinking agent
- the product SD 448 B is the product 448 B which has been dried by spray drying or spray drying, then dissolution of the white powder obtained, shearing of the solution with 5-10 g / l of polymer approximately and then used as emulsion 448 B.
- Ionic regain (X-Y) / Y x 100 with X: ionicity after shear in meq / g.
- V1 intrinsic viscosity after shearing in dl / g
- V2 intrinsic viscosity before shearing in dl / g
- Pulp used mixture of 70% KF bleached hardwood kraft
- GCC crushed CaCO3 Bonding in neutral medium with 2% of a dimeric ketene alkyl emulsion.
- the paste used is diluted to a consistency of 1.5%. 2.24 g dry paste is taken, ie 149 g 15% paste and then diluted to 0.4% with clear water.
- G grammage ex.
- G80 grammage of 80 g
- the volume of 560 ml is introduced into the plexiglass cylinder of the automated form and the sequence is started.
- - t 10 s, addition of the polymer.
- - 1 60 s, automatic reduction to 1000 rpm and addition, if necessary, of bentonite.
- Figure 5 shows the histogram of the white water turbidity figures corresponding to Table 2
- Figure 6 shows the drip curve corresponding to Table 2
- the invention provides an important additional advantage with regard to a very marked improvement in the formation of the sheet.
- the formation designates, as is known, qualities of the sheet such as homogeneity, and the like.
- Example 2 The same polymeric agent is used as in Example 2 but at a different dosage (0.1% of polymer relative to the fibrous mass to be flocculated).
- Figure 7 shows the histogram of the white water turbidity figures corresponding to Table 3
- Figure 8 shows the drip curve corresponding to Table 3
- This example shows the effects obtained with a high dosage of retention agent.
- EXAMPLE 1 Type of dough: 70% KF, 10% KR, 20% PM, 20% GCC. Rot speed : 800/30 s CSF: 365 ml pH paste: 7.5 Consistency (before dilution): 1.5% G80
- EXAMPLE 1 Type of dough: 70% KF, 10% KR, 20% PM, 20% GCC. Rot speed : 800/30 s CSF: 365 ml pH paste: 7.5 Consistency (before dilution): 1.5% G80
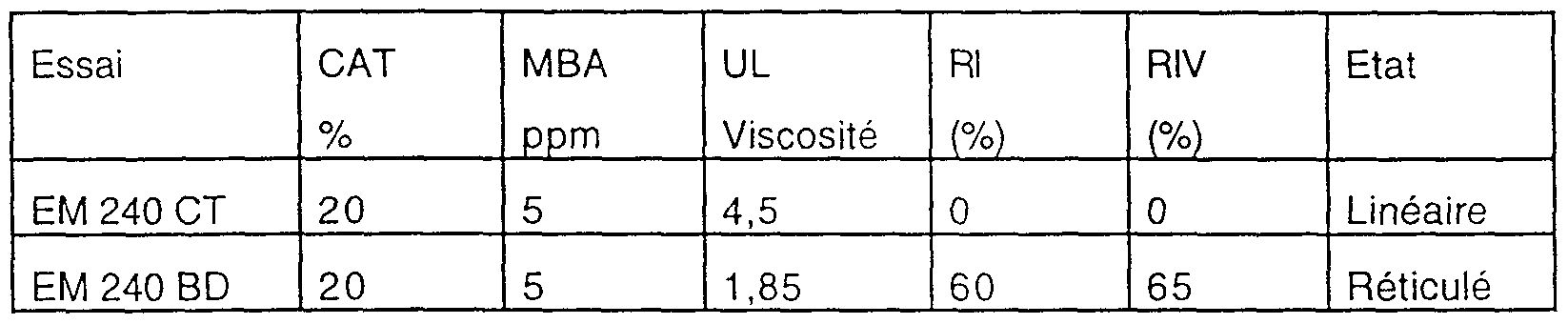
- the SD 240 BD product is the EM 240 BD product, except that it was prepared by solvent precipitation followed by drying and grinding; a white powder is obtained which is put back into solution, shears, then used as the emulsion EM 240 BD.
- EXAMPLE 2 Type of dough: 70% KF, 10% KR, 20% PM, 20% GCC. Rot speed : 800 EM 140 BD CSF: 399 pH paste: 7.5
- the product SD 448 B is the product 448 B except in that it was prepared by precipitation with solvent followed by drying and grinding; a white powder is obtained which is put back into solution, shears, then used as emulsion 448 B.
- EXAMPLE 2 Type of dough: 70% KF, 10% KR, 20% PM, 20% GCC. Rot speed : 800 EM140 BD CSF: 399 paste pH: 7.5
- EXAMPLE 3 Type of dough 70% KF, 10% KR, 20% PM, 20% GCC. Rot speed : 800 140 BD CSF: 399 pH paste: 7.5
Landscapes
- Paper (AREA)
Abstract
Description
Claims
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CA2333508A CA2333508C (fr) | 1998-05-28 | 1999-05-21 | Procede de floculation pour la fabrication de feuilles de papier, de carton ou autres articles semblables, emulsions a orientation transversale comme nouveaux agents de floculation pour cette preparation, et articles resultants |
| EP99920905A EP1090185B1 (fr) | 1998-05-28 | 1999-05-21 | Procede de floculation pour la fabrication d'une feuille de papier |
| AT99920905T ATE273417T1 (de) | 1998-05-28 | 1999-05-21 | Verfahren zur ausflockung bei der herstellung einer papierbahn |
| DE69919356T DE69919356T2 (de) | 1998-05-28 | 1999-05-21 | Verfahren zur ausflockung bei der herstellung einer papierbahn |
| AU38309/99A AU3830999A (en) | 1998-05-28 | 1999-05-21 | Flocculation method for making a paper sheet |
| US09/700,719 US6579417B1 (en) | 1998-05-28 | 1999-05-21 | Flocculation method for making a paper sheet |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| FR98/06876 | 1998-05-28 | ||
| FR9806876A FR2779159B1 (fr) | 1998-05-28 | 1998-05-28 | Procede de floculation pour la fabrication d'une feuille de papier, carton ou analogue, emulsions reticulees comme nouveaux agents floculants de cette preparation, et les articles ainsi obtenus |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO1999061702A1 true WO1999061702A1 (fr) | 1999-12-02 |
Family
ID=9526916
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/FR1999/001209 Ceased WO1999061702A1 (fr) | 1998-05-28 | 1999-05-21 | Procede de floculation pour la fabrication d'une feuille de papier |
Country Status (8)
| Country | Link |
|---|---|
| US (1) | US6579417B1 (fr) |
| EP (1) | EP1090185B1 (fr) |
| AT (1) | ATE273417T1 (fr) |
| AU (1) | AU3830999A (fr) |
| CA (1) | CA2333508C (fr) |
| DE (1) | DE69919356T2 (fr) |
| FR (1) | FR2779159B1 (fr) |
| WO (1) | WO1999061702A1 (fr) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7815771B2 (en) * | 2004-04-29 | 2010-10-19 | Snf S.A.S. | Process for the manufacture of paper and board |
| US20140367059A1 (en) * | 2012-02-01 | 2014-12-18 | Basf Se | Process for the manufacture of paper and paperboard |
Families Citing this family (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20030150575A1 (en) * | 1998-06-04 | 2003-08-14 | Snf Sa | Paper and paperboard production process and corresponding novel retention and drainage aids, and papers and paperboards thus obtained |
| DE10059828A1 (de) * | 2000-12-01 | 2002-06-13 | Clariant Gmbh | Kammförmige Copolymere auf Basis von Acryloyldimethyltaurinsäure |
| US20040087717A1 (en) * | 2002-11-04 | 2004-05-06 | Ge Betz, Inc. | Modified polymeric flocculants with improved performance characteristics |
| GB0518059D0 (en) * | 2005-09-06 | 2005-10-12 | Dow Corning | Delivery system for releasing active ingredients |
| CA3050234C (fr) | 2009-09-15 | 2022-11-08 | Suncor Energy Inc. | Techniques de floculation et de deshydratation de residus fins |
| US9404686B2 (en) | 2009-09-15 | 2016-08-02 | Suncor Energy Inc. | Process for dying oil sand mature fine tailings |
| CA2735330C (fr) | 2009-10-30 | 2015-06-02 | Suncor Energy Inc. | Procedes de sedimentation et de traitement agricole pour le sechage de residus fins |
| CN113248651B (zh) * | 2021-07-12 | 2021-10-01 | 山东诺尔生物科技有限公司 | 一种造纸助留剂及其制备方法和应用 |
| CN113354773B (zh) * | 2021-08-09 | 2021-10-29 | 山东诺尔生物科技有限公司 | 一种两性聚丙烯酰胺造纸助留剂及其制备方法 |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0201237A2 (fr) * | 1985-04-25 | 1986-11-12 | Ciba Specialty Chemicals Water Treatments Limited | Procédés de floculation |
| DE4406624A1 (de) * | 1994-03-01 | 1995-09-07 | Roehm Gmbh | Vernetzte wasserlösliche Polymerdispersionen |
| US5571380A (en) * | 1992-01-08 | 1996-11-05 | Nalco Chemical Company | Papermaking process with improved retention and maintained formation |
| US5630907A (en) * | 1992-12-07 | 1997-05-20 | Basf Aktiengesellschaft | Use of hydrolyzed copolymers of N-vinylcarboxamides and monoethylenically unsaturated carboxylic acids in papermaking |
Family Cites Families (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4759856A (en) * | 1984-04-30 | 1988-07-26 | Allied Colloids, Ltd. | Flocculation processes |
| US5180473A (en) * | 1987-03-20 | 1993-01-19 | Mitsui-Cyanamid, Ltd. | Paper-making process |
| US5254221A (en) * | 1988-04-22 | 1993-10-19 | Allied Colloids Limited | Processes for the production of paper and paper board |
| US5431783A (en) * | 1993-07-19 | 1995-07-11 | Cytec Technology Corp. | Compositions and methods for improving performance during separation of solids from liquid particulate dispersions |
-
1998
- 1998-05-28 FR FR9806876A patent/FR2779159B1/fr not_active Expired - Fee Related
-
1999
- 1999-05-21 US US09/700,719 patent/US6579417B1/en not_active Expired - Lifetime
- 1999-05-21 EP EP99920905A patent/EP1090185B1/fr not_active Revoked
- 1999-05-21 AT AT99920905T patent/ATE273417T1/de not_active IP Right Cessation
- 1999-05-21 DE DE69919356T patent/DE69919356T2/de not_active Revoked
- 1999-05-21 CA CA2333508A patent/CA2333508C/fr not_active Expired - Lifetime
- 1999-05-21 WO PCT/FR1999/001209 patent/WO1999061702A1/fr not_active Ceased
- 1999-05-21 AU AU38309/99A patent/AU3830999A/en not_active Abandoned
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0201237A2 (fr) * | 1985-04-25 | 1986-11-12 | Ciba Specialty Chemicals Water Treatments Limited | Procédés de floculation |
| US5571380A (en) * | 1992-01-08 | 1996-11-05 | Nalco Chemical Company | Papermaking process with improved retention and maintained formation |
| US5630907A (en) * | 1992-12-07 | 1997-05-20 | Basf Aktiengesellschaft | Use of hydrolyzed copolymers of N-vinylcarboxamides and monoethylenically unsaturated carboxylic acids in papermaking |
| DE4406624A1 (de) * | 1994-03-01 | 1995-09-07 | Roehm Gmbh | Vernetzte wasserlösliche Polymerdispersionen |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7815771B2 (en) * | 2004-04-29 | 2010-10-19 | Snf S.A.S. | Process for the manufacture of paper and board |
| US20140367059A1 (en) * | 2012-02-01 | 2014-12-18 | Basf Se | Process for the manufacture of paper and paperboard |
| US9404223B2 (en) * | 2012-02-01 | 2016-08-02 | Basf Se | Process for the manufacture of paper and paperboard |
Also Published As
| Publication number | Publication date |
|---|---|
| FR2779159A1 (fr) | 1999-12-03 |
| FR2779159B1 (fr) | 2000-08-11 |
| CA2333508A1 (fr) | 1999-12-02 |
| EP1090185B1 (fr) | 2004-08-11 |
| EP1090185A1 (fr) | 2001-04-11 |
| CA2333508C (fr) | 2010-05-18 |
| AU3830999A (en) | 1999-12-13 |
| ATE273417T1 (de) | 2004-08-15 |
| US6579417B1 (en) | 2003-06-17 |
| DE69919356D1 (de) | 2004-09-16 |
| DE69919356T2 (de) | 2005-09-08 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1086276B1 (fr) | Procede de fabrication de papier et carton | |
| CA2097127C (fr) | Procede de fabrication d'un papier ou d'un carton a retention amelioree | |
| EP1171487A1 (fr) | Nouveaux procedes de synthese d'agents floculants et coagulants de type polyvinylamine pva, nouveaux agents obtenus, leurs utilisations, et papiers ameliores ainsi obtenus | |
| FR2929963A1 (fr) | Procede de fabrication de papier et carton | |
| WO2013128109A1 (fr) | Nouveau procede de fabrication de papier mettant en oeuvre un copolymere base ayant reagi avec un aldehyde comme agent de resistance a sec, de retention, d'egouttage et de machinabilite | |
| EP3423630B1 (fr) | Procédé de fabrication de papier et de carton | |
| EP0800597B1 (fr) | Procede pour ameliorer la retention dans un procede de fabrication du papier, et utilisation d'un agent de retention a cette fin | |
| WO2012017172A1 (fr) | Procede de fabrication de papier et carton presentant des proprietes de retention et d'egouttage ameliorees | |
| EP1090185B1 (fr) | Procede de floculation pour la fabrication d'une feuille de papier | |
| EP1092064B2 (fr) | Procede de fabrication de papier et carton | |
| EP1740769A1 (fr) | Procede de fabrication de papier et carton, nouveaux agents de retention et d'egouttage correspondants, et papiers et cartons ainsi obtenus | |
| WO2000001757A1 (fr) | Polymeres acryliques en dispersions essentiellement aqueuses | |
| EP4121466B1 (fr) | Nouveaux complexes de polymeres hydrosolubles sous forme d'emulsion inverse et leurs utilisations | |
| EP4189162B1 (fr) | Procede de fabrication de papier et de carton | |
| EP3990700B1 (fr) | Procede de fabrication de papier ou de carton | |
| FR2879631A1 (fr) | Procede pour la fabrication de papier | |
| FR3149009A1 (fr) | Polymère et son procédé de préparation | |
| MXPA97003180A (en) | Process for pa manufacturing |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AL AM AT AU AZ BA BB BG BR BY CA CH CN CU CZ DE DK EE ES FI GB GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MD MG MK MN MW MX NO NZ PL PT RO RU SD SE SG SI SK SL TJ TM TR TT UA UG US UZ VN YU ZA ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): GH GM KE LS MW SD SL SZ UG ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE BF BJ CF CG CI CM GA GN GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| ENP | Entry into the national phase |
Ref document number: 2333508 Country of ref document: CA |
|
| NENP | Non-entry into the national phase |
Ref country code: KR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1999920905 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 09700719 Country of ref document: US |
|
| REG | Reference to national code |
Ref country code: DE Ref legal event code: 8642 |
|
| WWP | Wipo information: published in national office |
Ref document number: 1999920905 Country of ref document: EP |
|
| WWG | Wipo information: grant in national office |
Ref document number: 1999920905 Country of ref document: EP |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: 1999920905 Country of ref document: EP |