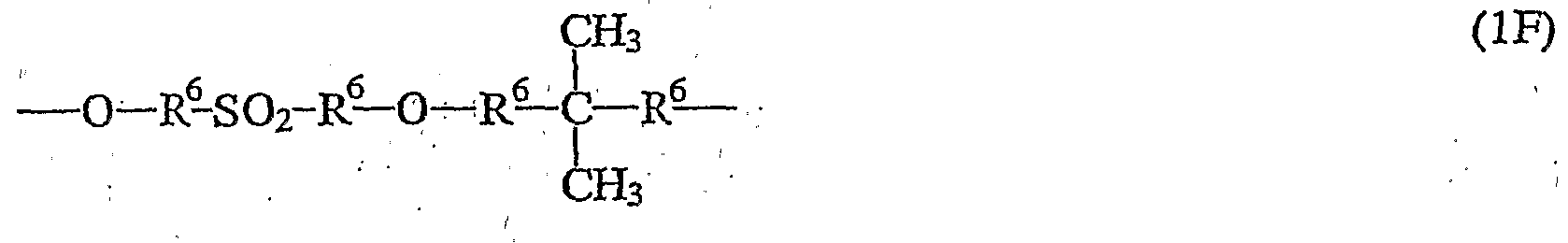WO2003014201A2 - Membranen für ionentransport - Google Patents
Membranen für ionentransport Download PDFInfo
- Publication number
- WO2003014201A2 WO2003014201A2 PCT/EP2002/007585 EP0207585W WO03014201A2 WO 2003014201 A2 WO2003014201 A2 WO 2003014201A2 EP 0207585 W EP0207585 W EP 0207585W WO 03014201 A2 WO03014201 A2 WO 03014201A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- acid
- polymeric
- membrane
- membranes
- base
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L81/00—Compositions of macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing sulfur with or without nitrogen, oxygen or carbon only; Compositions of polysulfones; Compositions of derivatives of such polymers
- C08L81/06—Polysulfones; Polyethersulfones
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D67/00—Processes specially adapted for manufacturing semi-permeable membranes for separation processes or apparatus
- B01D67/0081—After-treatment of organic or inorganic membranes
- B01D67/0093—Chemical modification
- B01D67/00931—Chemical modification by introduction of specific groups after membrane formation, e.g. by grafting
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D69/00—Semi-permeable membranes for separation processes or apparatus characterised by their form, structure or properties; Manufacturing processes specially adapted therefor
- B01D69/14—Dynamic membranes
- B01D69/141—Heterogeneous membranes, e.g. containing dispersed material; Mixed matrix membranes
- B01D69/1411—Heterogeneous membranes, e.g. containing dispersed material; Mixed matrix membranes containing dispersed material in a continuous matrix
- B01D69/14111—Heterogeneous membranes, e.g. containing dispersed material; Mixed matrix membranes containing dispersed material in a continuous matrix with nanoscale dispersed material, e.g. nanoparticles
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D71/00—Semi-permeable membranes for separation processes or apparatus characterised by the material; Manufacturing processes specially adapted therefor
- B01D71/06—Organic material
- B01D71/52—Polyethers
- B01D71/522—Aromatic polyethers
- B01D71/5221—Polyaryletherketone
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D71/00—Semi-permeable membranes for separation processes or apparatus characterised by the material; Manufacturing processes specially adapted therefor
- B01D71/06—Organic material
- B01D71/66—Polymers having sulfur in the main chain, with or without nitrogen, oxygen or carbon only
- B01D71/68—Polysulfones; Polyethersulfones
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D71/00—Semi-permeable membranes for separation processes or apparatus characterised by the material; Manufacturing processes specially adapted therefor
- B01D71/06—Organic material
- B01D71/76—Macromolecular material not specifically provided for in a single one of groups B01D71/08 - B01D71/74
- B01D71/80—Block polymers
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D71/00—Semi-permeable membranes for separation processes or apparatus characterised by the material; Manufacturing processes specially adapted therefor
- B01D71/06—Organic material
- B01D71/76—Macromolecular material not specifically provided for in a single one of groups B01D71/08 - B01D71/74
- B01D71/82—Macromolecular material not specifically provided for in a single one of groups B01D71/08 - B01D71/74 characterised by the presence of specified groups, e.g. introduced by chemical after-treatment
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J41/00—Anion exchange; Use of material as anion exchangers; Treatment of material for improving the anion exchange properties
- B01J41/08—Use of material as anion exchangers; Treatment of material for improving the anion exchange properties
- B01J41/12—Macromolecular compounds
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J47/00—Ion-exchange processes in general; Apparatus therefor
- B01J47/12—Ion-exchange processes in general; Apparatus therefor characterised by the use of ion-exchange material in the form of ribbons, filaments, fibres or sheets, e.g. membranes
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J5/00—Manufacture of articles or shaped materials containing macromolecular substances
- C08J5/20—Manufacture of shaped structures of ion-exchange resins
- C08J5/22—Films, membranes or diaphragms
- C08J5/2206—Films, membranes or diaphragms based on organic and/or inorganic macromolecular compounds
- C08J5/2218—Synthetic macromolecular compounds
- C08J5/2256—Synthetic macromolecular compounds based on macromolecular compounds obtained by reactions other than those involving carbon-to-carbon bonds, e.g. obtained by polycondensation
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J5/00—Manufacture of articles or shaped materials containing macromolecular substances
- C08J5/20—Manufacture of shaped structures of ion-exchange resins
- C08J5/22—Films, membranes or diaphragms
- C08J5/2206—Films, membranes or diaphragms based on organic and/or inorganic macromolecular compounds
- C08J5/2275—Heterogeneous membranes
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M8/00—Fuel cells; Manufacture thereof
- H01M8/04—Auxiliary arrangements, e.g. for control of pressure or for circulation of fluids
- H01M8/04082—Arrangements for control of reactant parameters, e.g. pressure or concentration
- H01M8/04197—Preventing means for fuel crossover
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M8/00—Fuel cells; Manufacture thereof
- H01M8/10—Fuel cells with solid electrolytes
- H01M8/1016—Fuel cells with solid electrolytes characterised by the electrolyte material
- H01M8/1018—Polymeric electrolyte materials
- H01M8/102—Polymeric electrolyte materials characterised by the chemical structure of the main chain of the ion-conducting polymer
- H01M8/1023—Polymeric electrolyte materials characterised by the chemical structure of the main chain of the ion-conducting polymer having only carbon, e.g. polyarylenes, polystyrenes or polybutadiene-styrenes
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M8/00—Fuel cells; Manufacture thereof
- H01M8/10—Fuel cells with solid electrolytes
- H01M8/1016—Fuel cells with solid electrolytes characterised by the electrolyte material
- H01M8/1018—Polymeric electrolyte materials
- H01M8/102—Polymeric electrolyte materials characterised by the chemical structure of the main chain of the ion-conducting polymer
- H01M8/1025—Polymeric electrolyte materials characterised by the chemical structure of the main chain of the ion-conducting polymer having only carbon and oxygen, e.g. polyethers, sulfonated polyetheretherketones [S-PEEK], sulfonated polysaccharides, sulfonated celluloses or sulfonated polyesters
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M8/00—Fuel cells; Manufacture thereof
- H01M8/10—Fuel cells with solid electrolytes
- H01M8/1016—Fuel cells with solid electrolytes characterised by the electrolyte material
- H01M8/1018—Polymeric electrolyte materials
- H01M8/102—Polymeric electrolyte materials characterised by the chemical structure of the main chain of the ion-conducting polymer
- H01M8/1027—Polymeric electrolyte materials characterised by the chemical structure of the main chain of the ion-conducting polymer having carbon, oxygen and other atoms, e.g. sulfonated polyethersulfones [S-PES]
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M8/00—Fuel cells; Manufacture thereof
- H01M8/10—Fuel cells with solid electrolytes
- H01M8/1016—Fuel cells with solid electrolytes characterised by the electrolyte material
- H01M8/1018—Polymeric electrolyte materials
- H01M8/102—Polymeric electrolyte materials characterised by the chemical structure of the main chain of the ion-conducting polymer
- H01M8/103—Polymeric electrolyte materials characterised by the chemical structure of the main chain of the ion-conducting polymer having nitrogen, e.g. sulfonated polybenzimidazoles [S-PBI], polybenzimidazoles with phosphoric acid, sulfonated polyamides [S-PA] or sulfonated polyphosphazenes [S-PPh]
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M8/00—Fuel cells; Manufacture thereof
- H01M8/10—Fuel cells with solid electrolytes
- H01M8/1016—Fuel cells with solid electrolytes characterised by the electrolyte material
- H01M8/1018—Polymeric electrolyte materials
- H01M8/102—Polymeric electrolyte materials characterised by the chemical structure of the main chain of the ion-conducting polymer
- H01M8/1032—Polymeric electrolyte materials characterised by the chemical structure of the main chain of the ion-conducting polymer having sulfur, e.g. sulfonated-polyethersulfones [S-PES]
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M8/00—Fuel cells; Manufacture thereof
- H01M8/10—Fuel cells with solid electrolytes
- H01M8/1016—Fuel cells with solid electrolytes characterised by the electrolyte material
- H01M8/1018—Polymeric electrolyte materials
- H01M8/1039—Polymeric electrolyte materials halogenated, e.g. sulfonated polyvinylidene fluorides
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M8/00—Fuel cells; Manufacture thereof
- H01M8/10—Fuel cells with solid electrolytes
- H01M8/1016—Fuel cells with solid electrolytes characterised by the electrolyte material
- H01M8/1018—Polymeric electrolyte materials
- H01M8/1041—Polymer electrolyte composites, mixtures or blends
- H01M8/1044—Mixtures of polymers, of which at least one is ionically conductive
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2323/00—Details relating to membrane preparation
- B01D2323/30—Cross-linking
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2381/00—Characterised by the use of macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing sulfur with or without nitrogen, oxygen, or carbon only; Polysulfones; Derivatives of such polymers
- C08J2381/06—Polysulfones; Polyethersulfones
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M2300/00—Electrolytes
- H01M2300/0017—Non-aqueous electrolytes
- H01M2300/0065—Solid electrolytes
- H01M2300/0082—Organic polymers
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/30—Hydrogen technology
- Y02E60/50—Fuel cells
Definitions
- the present invention relates to ionically crosslinked polymers and ionically crosslinked polymers with inorganic contents.
- Polymers that are used in membranes are, for example, polyarylenes, such as polyphenylene and polypyrene, aromatic polyvinyl compounds, such as polystyrene and polyvinylpyridine, polyphenylene vinylene, aromatic polyethers, such as polyphenylene oxide, aromatic polythioethers, such as polyphenylene sulfide, polysulfones, such as ®Radel R, and polyether ketones, such as PEK.
- polyarylenes such as polyphenylene and polypyrene
- aromatic polyvinyl compounds such as polystyrene and polyvinylpyridine
- aromatic polyethers such as polyphenylene oxide
- aromatic polythioethers such as polyphenylene sulfide
- polysulfones such as ®Radel R
- polyether ketones such as PEK.
- they also include polypyrroles, polythiophenes, polyazoles, such as polybenzimidazole, polyanilines, polya
- membranes are doped with concentrated phosphoric acid or sulfuric acid and serve as proton conductors in so-called polyelectrolyte membrane fuel cells (PEM fuel cells).
- PEM fuel cells polyelectrolyte membrane fuel cells
- MEE membrane electrode assembly
- a disadvantage of these membranes is their mechanical instability with a low modulus of elasticity, a low tensile strength and a low upper flow limit, and their relatively high permeability to hydrogen, oxygen and methanol.
- DE 196 22 337 describes a ner process for the production of covalently crosslinked ionomer membranes which is based on an alkylation reaction of polymers containing sulfinate groups, polymer blends and polymer (blend) membranes.
- the covalent network has good resistance to hydrolysis even at higher temperatures.
- WO 99/02756 and WO 99/02755 disclose ionically crosslinked acid-base polymer blends and Porymer (blend) membranes.
- An advantage of the ionically crosslinked acid-base blend membranes is that the ionic bonds are flexible, the polymers / membranes do not dry out so easily even at higher temperatures because of the hydrophilicity of the acid-base groups, and therefore the polymers / membranes also do not become brittle at higher temperatures.
- IEC proton exchange capacity of the polymeric acid
- B- proportion of the base added
- Anion exchangers naturally also contain the anions of the acid used for the oxidation as the counter ion.
- the polymeric acid used must be proton-neutralized, otherwise complexing occurs when the components are mixed in.
- Membrane is also positive charge, which opposes the transport of the protons.
- the polymer according to the invention should have a low volume resistivity, preferably less than or equal to 200 ohm ⁇ cm at 25 ° C. in water, and a low permeability for hydrogen, oxygen and methanol.
- Another object was to provide a polymer that can be used in fuel cells.
- the polymer should be suitable for use in direct methanol fuel cells.
- the object of the invention was also to provide a process for the preparation of the ionically and optionally covalently crosslinked polymer which can be carried out in a simple manner, inexpensively and on an industrial scale.
- anion exchangers which have hydroxyl ions as the counterion and which have been processed into membranes with known cation exchangers, preferably those mentioned in WO 99/02756 and WO 99/02755, have a higher proton conductivity than the control in which the anion exchangers have halogen anions as the counterion exhibit.
- the polymeric anion exchanger is diluted with a solution containing hydroxyl ions, preferably an aqueous, e.g. NaOH in water, added and the negative counterions are exchanged with the excess of hydroxyl ions.
- the anion exchanger is then rinsed with demineralized water to the pH of the wash water. This pH is preferably between 6.5 and 7.5. Then dried and dissolved in a suitable, preferably aprotic, solvent.
- the polymeric acid is added in the salt form, preferably a mono-, di-, tri-, or tetravalent cation is used.
- one or more polymeric bases also dissolved in an aprotic solvent, can be added to the mixture.
- the mixture is processed into a membrane according to the prior art.
- the polymeric acid is still in the salt form after drying.
- An acidic cation exchange resin is used to convert them into the necessary acid form. Any other known process for converting to the acid group is also suitable which excludes the anions from reacting with the anion exchanger and as a result of which the hydroxyl ions are displaced.
- the polymeric acid in the membrane is now exchanged, it is in the protonated form and in parallel there is the anion exchanger with the hydroxyl ion in the membrane. In the subsequent further processing of the membrane into the fuel cell, it must be ensured that the membrane is never exposed to exchangeable anions.
- a cation exchanger and an anion exchanger may also be mixed with one or more polymeric bases and processed into a membrane without the membrane again containing low-molecular-weight anions, such as F “ , Cl “ , Br “ , J " or other is brought into contact.
- the membranes show a reduced methanol permeability with simultaneously increased proton conductivity (measured in water) compared to the control.
- part of the invention is a new process for the production of acid-base blends with nanodispersed, sparingly soluble salts and oxides, titanium and zirconium salts being particularly preferred.
- An acid-base blend is a polymer or polymer mixture which carries at least one group which releases protons in an aqueous environment and at least one group which fixes protons.
- the principle of the acid-base interaction is described in detail in the publications WO 99/02755 and WO 99/02756. All the production methods of acid-base blends and acid-base blend membranes that have been described and disclosed so far always result in aftertreatment in dilute protonic acids.
- zirconyl (ZrO 2+ ) and titanyl cation (TiO 2+ ) are particularly preferred. It was surprisingly found that polymeric acids exchanged with zirconyl (ZrO + ) and / or titanyl cations (TiO 2+ ), in particular sulfonic acids, with polymeric bases, for example polybenzimidazoles (PBI), polyvinylpyridine (PVP and P4VP), poly aminated Allow (ether) sulfones and aminated polyaryl ether ketones to be mixed homogeneously with one another in an aprotic solvent such as NMP, DMAc and DMSO.
- PBI polybenzimidazoles
- PVP and P4VP poly aminated Allow (ether) sulfones and aminated polyaryl ether ketones
- converting a sulfonic acid into its zirconyl salt can e.g. proceed as follows:
- IEC ion exchange capacity
- the polymeric acid can be protonated or in the cation-exchanged form, ⁇ a + , K + , Li + , Ca + , Mg 2+ are preferred.
- the water-soluble, but soluble in aprotic solvents is an IEC up to approximately 1.8.
- the exchanged acid is filtered off and carefully dried in vacuo at low temperature, preferably below 50 ° C.
- the solvent is NMP or DMAc
- the resulting solution can be mixed immediately thereafter with a solution of a polymeric base and / or a polymeric anion exchanger or its reduced preform in an aprotic solvent, for example PBI in DMAc and processed into a membrane.
- the processing into a membrane takes place, for example, by doctoring to a thin film on a suitable surface.
- the salt form of the polymeric acid must still be brought into its protonated form.
- the zirconyl (ZrO 2+ ) or titanyl cation (TiO 2+ ) reacts with water to form sparingly soluble zirconium dioxide or titanium dioxide.
- the polymeric acid undergoes protonation and the acid-base interaction can develop.
- the product obtained is an acid-base blend with molecularly dispersed zirconium or titanium dioxide.
- the advantage of this method is not only the simplified ecological and economic representation of acid-base blends with molecularly distributed oxides, but the membranes can be coated with a catalyst before activation with water, in particular in the case of fuel cell applications, and processed further to form a membrane electrode assembly and only then, at the latest when the fuel cell is in operation, does the protonation of the polymeric acid take place.
- blends containing at least one polymeric cation exchanger, one polymeric anion exchanger, molecularly dispersed metal oxide and / or one polymeric base in which a non-oxidized or only partially oxidized preform is used instead of the anion exchanger.
- the use of the reduced precursor is particularly advantageous, in particular in the case of polymeric triphenylmethane dyes.
- the following compound represents a non-oxidized form and its oxidized form of such a weakly basic anion exchanger.
- a film produced by the above process containing at least one polymeric zirconyl (ZrO 2+ ) and / or titanyl cation (TiO 2+ ) exchanged acid and a polymeric base and / or a polymeric anion exchanger is or after-treated with phosphoric acid (diluted to concentrated) or diluted Sulfuric acid converted to the protonated form.
- This method has the advantage that no mono- or divalent metal-containing waste acids or alkalis are produced in order to generate a protonated zirconium phosphate or titanium phosphate or the corresponding sulfates from the membrane.
- molecularly dispersed metal salts or oxides in particular of zirconium dioxide, titanium dioxide, zirconium phosphate, titanium phosphate, the corresponding hydrogen phosphates, sulfates and hydrogen sulfates, is a reduced methanol diffusion through the membrane, with an increased proton conductivity in the membrane.
- This has the advantages already described in the art.
- the acid-base interaction can still develop.
- the process according to the invention is used to produce new acid-base blends, acid-anion exchanger blends, acid-anion exchanger-base blends with molecularly dispersed oxides or salts.
- the membranes can be used to generate energy by electrochemical means.
- membrane fuel cells H2 or direct methanol fuel cells
- They can be used in electrochemical cells, secondary batteries, electrolysis cells, in membrane separation processes such as gas separation, pervaporation, perstraction, reverse osmosis, electrodialysis and diffusion dialysis.
- part of the invention is the use of polymer-bound dyes which have at least two heteroatoms. These dyes must have at least two boundary structures. It was surprisingly found that the water transport numbers through the membrane decrease in fuel cell operation for each proton transported when using dyes, in particular polymer-bound dyes. It was also found that the methanol permeability due to the added dyes was lower than the control without the dyes. The same effects were also observed when polyaniline was added to the membrane. However, care must be taken to ensure that the electron conductivity of the polyaniline is not sufficient across the entire membrane thickness. Additions of 2 to 10% by weight of the polyaniline are completely sufficient for the effects.
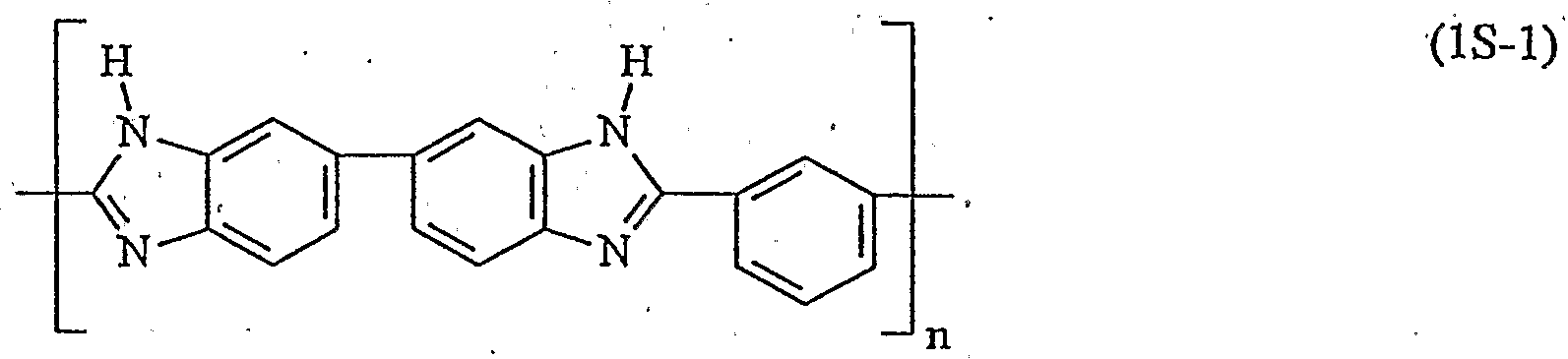
- the radicals R 6 independently of one another identical or different 1,2-phenylene, 1,3-phenylene, 1,4-phenylene, 4,4'-biphenyl, a divalent radical of a heteroaromatic, a divalent radical of a C 10 ⁇ aromatics , a divalent radical of a C 14 aromatic and / or a divalent pyrene radical.
- a C 10 -Aromateri is naphthalene, a C X4 aromatics phenanthrene.
- the substitution pattern of the aromatic and / or heteroaromatic is arbitrary, in the case of phenylene, for example, R 6 can be ortho-, meta- and para-ptienylene.
- the radicals R 7 , R 8 and R 9 denote one, four or three-membered aromatic or heteroaromatic groups and the radicals U, which are the same within a repeating unit, are. for an oxygen atom, a sulfur atom or an amino group which carries a hydrogen atom, a group having 1-20 carbon atoms, preferably a branched or unbranched alkyl or alkoxy group, or an aryl group as a further radical.
- polymers having recurring units of the general formula (1) belong to homopolymers and copolymers, for example random copolymers, such as Victrex ® 720 P and Astrel ®.
- Very particularly preferred polymers are polyaryl ethers, polyaryl thioethers, polysulfones, polyether ketones, poly pyrroles, polythiophenes, polyazoles, polyphenylenes.
- u4MHi polymers with recurring units of the general formula (1A-1), (IB-i), (1C-1), (II-1), (1G-1), (1E-1), (lH-, 1), (ll-l), (1F-1), (1J-1), (1K-1), (1L-1), (1M-1) ; and / or (1N-1).
- n denotes the number of repeating units along a macromolecule chain of the polymer.
- This number of repeating units of the general formula (1) along a macromolecule chain of the crosslinked polymer is preferably an integer greater than or equal to 10, in particular greater than or equal to 100.
- the number of repeating units of the general formula (1A), (1B), ' (1C), (1D), (1E), (IF), (IG), (1H), (II), (IJ), (IK), (IL), (IM), (IN), (1Q ) > (IP), (IQ), (1R), (IS) and / or (IT) along a macromolecule chain of the crosslinked polymer an integer greater than or equal to 10, in particular greater than or equal to 100.
- the number average molecular weight of the macromolecule chain is greater than 25,000 g / mol, advantageously greater than 50,000 g / mol, in particular greater than 100,000 g / mol.
Landscapes
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Engineering & Computer Science (AREA)
- Manufacturing & Machinery (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Chemical & Material Sciences (AREA)
- Electrochemistry (AREA)
- Sustainable Energy (AREA)
- Sustainable Development (AREA)
- Health & Medical Sciences (AREA)
- Organic Chemistry (AREA)
- Crystallography & Structural Chemistry (AREA)
- Inorganic Chemistry (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Materials Engineering (AREA)
- Transplantation (AREA)
- Dispersion Chemistry (AREA)
- Nanotechnology (AREA)
- Composite Materials (AREA)
- Manufacture Of Macromolecular Shaped Articles (AREA)
Abstract
Description
Claims
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AU2002336925A AU2002336925A1 (en) | 2001-07-07 | 2002-07-08 | Membranes for ion transport |
| DE10293515T DE10293515D2 (de) | 2001-07-07 | 2002-07-08 | Membranen für Ionentransport |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE10134793A DE10134793A1 (de) | 2001-07-07 | 2001-07-07 | Membranen für Ionentransport |
| DE10134793.6 | 2001-07-07 | ||
| DE10158006 | 2001-11-22 | ||
| DE10158006.1 | 2001-11-22 |
Publications (4)
| Publication Number | Publication Date |
|---|---|
| WO2003014201A2 true WO2003014201A2 (de) | 2003-02-20 |
| WO2003014201A8 WO2003014201A8 (de) | 2003-07-31 |
| WO2003014201A9 WO2003014201A9 (de) | 2003-10-30 |
| WO2003014201A3 WO2003014201A3 (de) | 2005-04-07 |
Family
ID=27614234
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2002/007585 Ceased WO2003014201A2 (de) | 2001-07-07 | 2002-07-08 | Membranen für ionentransport |
Country Status (2)
| Country | Link |
|---|---|
| AU (1) | AU2002336925A1 (de) |
| WO (1) | WO2003014201A2 (de) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2004098773A3 (de) * | 2003-05-06 | 2005-02-10 | Forschungszentrum Juelich Gmbh | Katalysatorschicht enthaltend einen sauren ionenaustauscher und bestimmte basische polymere, geeignete katalysatorpaste, sowie herstellungsverfahren derselben |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE19622337C1 (de) | 1996-06-04 | 1998-03-12 | Dlr Deutsche Forschungsanstalt | Vernetzung von modifizierten Engineering Thermoplasten |
| WO1999002756A1 (en) | 1997-07-14 | 1999-01-21 | Symetrix Corporation | Method and apparatus for fabrication of thin films by chemical vapor deposition |
| WO1999002755A1 (en) | 1997-07-11 | 1999-01-21 | Applied Materials, Inc. | Chemical vapor deposition manifold |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4380590A (en) * | 1978-09-19 | 1983-04-19 | Rohm And Haas Company | Emulsion copolymer cation exchange resins |
| US4508852A (en) * | 1983-09-22 | 1985-04-02 | Albany International Corp. | Compositions and method of preparation by chlorosulfonation of difficultly sulfonatable poly(ether sulfone) |
-
2002
- 2002-07-08 WO PCT/EP2002/007585 patent/WO2003014201A2/de not_active Ceased
- 2002-07-08 AU AU2002336925A patent/AU2002336925A1/en not_active Abandoned
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE19622337C1 (de) | 1996-06-04 | 1998-03-12 | Dlr Deutsche Forschungsanstalt | Vernetzung von modifizierten Engineering Thermoplasten |
| WO1999002755A1 (en) | 1997-07-11 | 1999-01-21 | Applied Materials, Inc. | Chemical vapor deposition manifold |
| WO1999002756A1 (en) | 1997-07-14 | 1999-01-21 | Symetrix Corporation | Method and apparatus for fabrication of thin films by chemical vapor deposition |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2004098773A3 (de) * | 2003-05-06 | 2005-02-10 | Forschungszentrum Juelich Gmbh | Katalysatorschicht enthaltend einen sauren ionenaustauscher und bestimmte basische polymere, geeignete katalysatorpaste, sowie herstellungsverfahren derselben |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2003014201A9 (de) | 2003-10-30 |
| WO2003014201A3 (de) | 2005-04-07 |
| AU2002336925A1 (en) | 2003-02-24 |
| WO2003014201A8 (de) | 2003-07-31 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1639153B1 (de) | Composites und compositemembranen | |
| DE60214166T2 (de) | Polymerelektrolyt für eine brennstoffzelle des festpolymertyps und brennstoffzelle | |
| DE102009020232B4 (de) | Sulfonierte Polyperfluorcyclobutan-Polyphenylen-Polymere für PEM-Brennstoffzellenanwendungen | |
| EP1073690B1 (de) | Säure-base-polymerblends und ihre verwendung in membranprozessen | |
| EP2443172B1 (de) | Aromatische polyethersulfon-blockcopolymere | |
| DE102014009170A1 (de) | Kombinatorisches Materialsystem für Ionenaustauschermembranen und dessen Verwendung in elektrochemischen Prozessen | |
| WO2001084657A2 (de) | Polymere membranen | |
| DE19919881A1 (de) | Organisch-Anorganische Komposites und Kompositmembranen aus Ionomeren oder Ionomerblends und aus Schicht- oder Gerätsilicaten | |
| EP1432741A2 (de) | Sulfinatgruppen enthaltende oligomere und polymere und verfahren zu ihrer herstellung | |
| DE60033681T2 (de) | Komposit-ionenaustauschmembranen | |
| EP1392765A1 (de) | Modifikation von verstreckten folien | |
| EP1373364B1 (de) | Sulfoniertes polyetherketonketon | |
| DE60029731T2 (de) | Fester Polymerelektrolyt mit hoher Dauerhaftigkeit | |
| WO2003060012A1 (de) | Funktionalisierte hauptkettenpolymere | |
| DE60014713T2 (de) | Membranen aus Pfropfpolymeren und Ionenaustauschmenbranen daraus | |
| DE102022120196A1 (de) | Seitenkettenfunktionalisierte Polystyrole als Membranmaterialien für alkalische Wasserelektrolyseure, Brennstoffzellen und Flow-Batterien | |
| EP1292634B1 (de) | Perfluorsulfonsäure-menbranen, verfahren zu ihrer herstellung und verwendung für brennstoffzellen | |
| WO2003014201A2 (de) | Membranen für ionentransport | |
| EP1673832B1 (de) | Schichtstrukturen und verfahren zu deren herstellung | |
| DE3143804A1 (de) | Mikroporoese ionenaustauschermembran und verfahren zu ihrer herstellung | |
| DE10134793A1 (de) | Membranen für Ionentransport | |
| DE10295737B4 (de) | Kovalent vernetzter Komposit, kovalente vernetzte Kompositmembran, Verfahren zu deren Herstellung und Verwendung der Membranen | |
| DE10296977T5 (de) | Elektrodenstruktur für Polymerelektrolytbrennstoffzellen | |
| WO2008034399A1 (de) | Mischungen sulfonierter und phosphonierter polymere | |
| EP4302351A2 (de) | Membran für protonenleitung |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A2 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NO NZ OM PH PL PT RO RU SD SE SG SI SK SL TJ TM TN TR TT TZ UA UG US UZ VN YU ZA ZM ZW Kind code of ref document: A2 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BY BZ CA CH CN CO CR CU CZ DE DM DZ EC EE ES FI GB GD GE GH HR HU ID IL IN IS JP KE KG KP KR LC LK LR LS LT LU LV MA MD MG MN MW MX MZ NO NZ OM PH PL PT RU SD SE SG SI SK SL TJ TM TN TR TZ UA UG US UZ VN YU ZA ZM |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A2 Designated state(s): GH GM KE LS MW MZ SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR IE IT LU MC NL PT SE SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG Kind code of ref document: A2 Designated state(s): GH GM KE LS MW MZ SD SL SZ UG ZM ZW AM AZ BY KG KZ RU TJ TM AT BE BG CH CY CZ DK EE ES FI FR GB GR IE IT LU MC PT SE SK TR BF BJ CF CG CI GA GN GQ GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| CFP | Corrected version of a pamphlet front page | ||
| CR1 | Correction of entry in section i |
Free format text: IN PCT GAZETTE 08/2003 UNDER (30) REPLACE "10156575.5, 21 NOVEMBER 2001 (21.11.2001)" BY "10158006.1 22 NOVEMBER 2001 (22.11.2001)" |
|
| COP | Corrected version of pamphlet |
Free format text: PAGES 1-13, DESCRIPTION, REPLACED BY NEW PAGES 1-16; PAGE 14, CLAIMS, REPLACED BY A NEW PAGE 17; DUE TO LATE TRANSMITTAL BY THE RECEIVING OFFICE |
|
| REF | Corresponds to |
Ref document number: 10293515 Country of ref document: DE Date of ref document: 20040527 Kind code of ref document: P |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 10293515 Country of ref document: DE |
|
| 122 | Ep: pct application non-entry in european phase | ||
| NENP | Non-entry into the national phase |
Ref country code: JP |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: JP |
|
| REG | Reference to national code |
Ref country code: DE Ref legal event code: 8607 |
|
| DPE2 | Request for preliminary examination filed before expiration of 19th month from priority date (pct application filed from 20040101) |