WO2004013142A1 - A riboflavin derivative and its manufacture and uses - Google Patents
A riboflavin derivative and its manufacture and uses Download PDFInfo
- Publication number
- WO2004013142A1 WO2004013142A1 PCT/CN2003/000609 CN0300609W WO2004013142A1 WO 2004013142 A1 WO2004013142 A1 WO 2004013142A1 CN 0300609 W CN0300609 W CN 0300609W WO 2004013142 A1 WO2004013142 A1 WO 2004013142A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- riboflavin
- formula
- compound
- preparation
- compound according
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/04—Ortho-condensed systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/02—Stomatological preparations, e.g. drugs for caries, aphtae, periodontitis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/04—Drugs for disorders of the alimentary tract or the digestive system for ulcers, gastritis or reflux esophagitis, e.g. antacids, inhibitors of acid secretion, mucosal protectants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/02—Drugs for dermatological disorders for treating wounds, ulcers, burns, scars, keloids, or the like
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/02—Drugs for skeletal disorders for joint disorders, e.g. arthritis, arthrosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/12—Antihypertensives
Definitions
- the invention relates to a riboflavin derivative and a preparation method and application thereof.
- Riboflavin is a component of the flavin enzyme prosthetic group. It is involved in the complex oxidation process of the body and plays an important pharmacological role. In recent years, pharmacological studies have shown that in addition to treating traditional riboflavin deficiency diseases, riboflavin also has pharmacological effects in the treatment of coronary heart disease, hypertension complications, and burns. However, riboflavin is a water-soluble vitamin, which is easily eliminated from sweat and urine, has a short half-life, and has low bioavailability.
- riboflavin intake when riboflavin intake is insufficient or the demand is increased, it can easily cause cheilitis, cheilitis, glossitis, scrotal dermatitis, etc., which are collectively called oral genital syndrome, as well as various nucleus such as conjunctivitis and bulbitis that affect vision. Flavin deficiency. For a long time, in order to prevent riboflavin deficiency caused by various reasons, a lot of research has been carried out on riboflavin derivatives. It is hoped that a suitable riboflavin derivative can be found as a prodrug and slowly released in vivo.
- the riboflavin is in its original form and can be stored in the body for a long time after a single application to exert the effect of the drug.
- Japanese scholar Kimi 0 and Miyamoto et al Synthesized riboflavin tetrapalmitic acid, tetradecanoate, tetrabutyrate, tetraacetate, and tryptophan tetraester by using riboflavin and fatty acid acyl chloride or anhydride in pyridine.
- these esters cannot be hydrolyzed in saliva and small intestinal fluid. Further research on tetrapalmitate showed that it has no riboflavin biological activity.
- the object of the present invention is to provide a riboflavin derivative and a synthesis method thereof.
- the derivative introduces a long-acting group, which can increase the retention of riboflavin in the body, so that it can increase absorption, delay excretion, and improve biological Utilization, to achieve the purpose of long-term effect.
- the riboflavin derivatives provided by the present invention are riboflavin tetrastearate, tetrapalmitate, trilaurate, tetradecanoate, tetrabutyrate, tetraacetate, and tryptophan. Tetraesters, all riboflavin phosphates except riboflavin phosphate, or ester derivatives on the ribitol hydroxyl group of the thioriboflavin side chain.
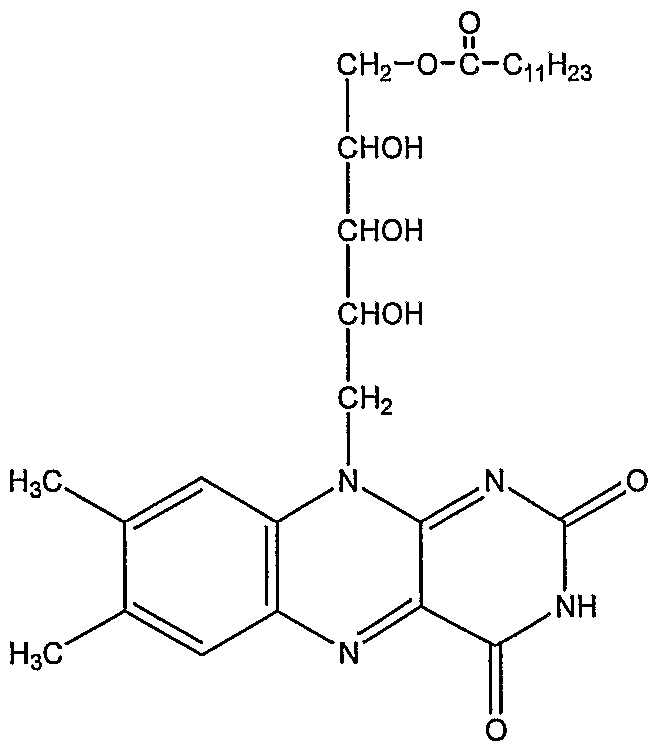
- the riboflavin derivative is a mono-, di-, tri- or tetra-ester compound represented by formula (I), wherein R 2 , R 3 , and R 4 are different, the same, or exist independently.
- the above riboflavin derivatives can be prepared by conventional methods.
- riboflavin-5'-lauric acid monoester represented by the structural formula ( ⁇ ) (
- the riboflavin-5'-lauric acid monoester is prepared by adding lauroyl chloride dropwise to riboflavin or thioriboflavin suspended in a polar organic solvent.
- the polar organic solvent of the suspended riboflavin or thioriboflavin is preferably pyridine or dimethylformamide.
- a second object of the present invention is to provide a riboflavin long-acting preparation using the compound of the present invention as an active ingredient and a preparation method thereof.
- a riboflavin long-acting oil suspension preparation is basically composed of a compound of formula ( ⁇ ) and ethyl oleate.
- camellia oil is also added to the oil suspension preparation, and the weight (volume) ratio of each component is:
- the preferred ratio of the weight (volume) of each component of the oil suspension formulation is:
- the riboflavin long-acting oil suspension preparation of the present invention is prepared by pulverizing the raw materials by a conventional blending method.
- riboflavin derivatives of the present invention have obvious effects in preventing and treating riboflavin deficiency, and overcome the defects that the existing riboflavin is easily discharged from body fluids, has a short half-life, and has low bioavailability.
- the long-acting riboflavin-5'-laurate in the compounds of the present invention is the compound with the highest yield and the best effect.
- four major spectral analyses (including two Dimensional superconducting nuclear magnetic resonance analysis) proves that it is a novel structured, novel riboflavin derivative.
- a long-acting riboflavin sleeve suspension injection prepared by using the riboflavin-5'-laurate, a single intramuscular injection of 150mg / ml, the effect is up to three months, in order to prevent common riboflavin deficiency Disease provides an important medical treatment.
- FIG 1 shows the changes in animal food intake during the experiment.
- FIG. 2 shows the changes in animal weight during the experiment.
- Figure 3 shows the changes in BGRAC of animals during the experiment.
- Figure 4 shows the changes of riboflavin content in the plasma of the animals during the experiment.
- the mixed solvent mother liquor was dried under reduced pressure at 35 ° C to obtain a red solid.
- the solid was washed with diethyl ether, dichloromethane 100mlX5, methanol 50mlX5, dried and crushed, and placed in a phosphorus pentoxide dryer for 48h to obtain 2.60g of red solid. Ester crude.
- the riboflavin was suspended in isobutyric anhydride, and perchloric acid was added dropwise at room temperature. After the addition, the reaction was stirred for 7 hours. The reaction solution was transparent purple, precipitated with ether, crystals were precipitated, filtered, and then dissolved with 30 ml of n-butanol. After crystallization, n-butanol was washed with water several times, dried, and diethyl ether was added to precipitate the crystals, filtered, dried under vacuum, and filtered to obtain a crude product.
- the target compound was obtained by the same separation method as in Example 1.
- Example 5 Preparation of an oil suspension formulation using the compound of the present invention as an active ingredient Formula: 50 mg of the compound of formula ( ⁇ ) (50-150 mg is acceptable)
- the compound of formula (II) was pulverized to 2-10 ⁇ m with an air stream, mixed with ethyl oleate and camellia oil, filled into a 1 ml ampoule, and sterilized to obtain an oil suspension preparation.
- Rat tests were used to determine the effects of long-acting riboflavin monoesters on preventing riboflavin deficiency by measuring urine riboflavin excretion, whole blood glutathione reductase activity coefficient, and plasma riboflavin content. Experimental evaluation of the month.
- AIN-76 was adapted for one week, and was randomly divided into 6 groups according to weight, namely the normal control group, riboflavin deficiency group, riboflavin group, long-acting riboflavin monoester low, medium and high dose groups. Flavin's AIN-76 was served, and tap water was freely consumed, daily food intake was recorded, and body weight was measured every 6 days. At the beginning of the test, orbital blood of normal control animals was taken, anticoagulated, and whole blood glutathione reduction was measured. Enzyme activity coefficient (BGRAC).
- riboflavin was taken.
- Orbital blood and urine were collected from the deficient group, riboflavin group, long-acting riboflavin monoester low, medium and high dose groups, and BGRAC, plasma riboflavin content and urine riboflavin excretion were measured respectively; at the end of the test, they were sacrificed
- the animals were taken out of the liver and kidneys, and their morphological changes were observed with an electron microscope.
- riboflavin-free AIN-76 feed In order to formulate riboflavin-free AIN-76 feed, in addition to the absence of riboflavin in the vitamin complex used, the casein and starch used were immersed in 1% sodium bisulfite for 24 hours before testing. Tap water was washed and dried, and samples were sent to Beijing Institute of Nutrition and Food Safety for analysis of riboflavin content, which was used to formulate feed when qualified. During the trial, a total of 80 kg of riboflavin-free AIN-76 feed was prepared.
- the long-acting riboflavin monoester (75mg / ml) and its solvent mixed oil used in the test were injected into the muscles of the right hind leg of the animal at one time.
- the injection was diluted with the mixed oil according to the dose.
- the dose used in the middle dose group was according to NAS-NC in the United States.
- the recommended riboflavin requirement for rats, the long-acting riboflavin monoester required for 90 days is set to 5.01mg, and the low and high dose groups were halved or doubled respectively; the riboflavin group was injected with riboflavin oil mixture, Grinded from riboflavin and mixed oil in an agate mortar, the dose is equivalent to that of the long-acting riboflavin monoester group; the riboflavin-deficient group was injected with an equal volume of mixed oil.
- BGRAC In the riboflavin-deficient animal test, BGRAC increased significantly at 15 days, and there was a rebound decrease at 60 days, for unknown reasons; in the riboflavin group, the BGRAC test also gradually increased after the start of the experiment, and the amplitude was significantly higher than that of the long-acting riboflavin monoester. Low-, medium- and high-dose groups, in which the amplitude within 30 days of the test was smaller than the riboflavin-deficient group; long-acting riboflavin monoester low-, medium-, and high-dose groups also had an increasing trend during the BGRAC test. Dose group> High dose group ( Figure 3).
- Example 7 Preventive effect of long-acting riboflavin on oral ulcers after chemotherapy
- Example 8 Long-acting riboflavin prevents leukemia bone marrow transplantation chemotherapy mucositis
- Example 10 Long-acting riboflavin for refractory oral ulcers
- the compounds and preparations provided by the present invention are used in the treatment of leukemia for bone marrow transplantation and anti-tumor chemotherapy, various dermatitis caused by radiotherapy, oral and gastrointestinal mucositis, riboflavin deficiency, refractory oral ulcer, coronary heart disease hypertension and hypertension. Can be widely used in medicine for arthritis and arthritis burns.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Life Sciences & Earth Sciences (AREA)
- Veterinary Medicine (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Pharmacology & Pharmacy (AREA)
- Public Health (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Cardiology (AREA)
- Heart & Thoracic Surgery (AREA)
- Urology & Nephrology (AREA)
- Vascular Medicine (AREA)
- Immunology (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Rheumatology (AREA)
- Physical Education & Sports Medicine (AREA)
- Dermatology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicines Containing Material From Animals Or Micro-Organisms (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
Description
Claims
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CNB038137348A CN100413866C (zh) | 2002-08-02 | 2003-07-29 | 核黄素衍生物及其制备方法与应用 |
| US10/522,110 US20060293335A1 (en) | 2002-08-02 | 2003-07-29 | Riboflavin derivative and its manufacture and uses |
| EP03766107A EP1553099A4 (en) | 2002-08-02 | 2003-07-29 | RIBOFLAVIN DERIVATIVE AND THEIR PREPARATION AND USES |
| AU2003257778A AU2003257778A1 (en) | 2002-08-02 | 2003-07-29 | A riboflavin derivative and its manufacture and uses |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN02125917.8 | 2002-08-02 | ||
| CN02125917 | 2002-08-02 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2004013142A1 true WO2004013142A1 (en) | 2004-02-12 |
Family
ID=31193849
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/CN2003/000609 Ceased WO2004013142A1 (en) | 2002-08-02 | 2003-07-29 | A riboflavin derivative and its manufacture and uses |
Country Status (5)
| Country | Link |
|---|---|
| US (1) | US20060293335A1 (zh) |
| EP (1) | EP1553099A4 (zh) |
| CN (1) | CN100413866C (zh) |
| AU (1) | AU2003257778A1 (zh) |
| WO (1) | WO2004013142A1 (zh) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101684120B (zh) * | 2008-09-26 | 2013-07-31 | 深圳信立泰药业股份有限公司 | 核黄素-5'-月桂酸单酯晶型及其制备方法和含有该晶型的药物组合物 |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN104473269A (zh) * | 2014-11-21 | 2015-04-01 | 安徽农业大学 | 一种提高水性溶液中核黄素光稳定性的方法及其应用 |
| CN107308515B (zh) * | 2016-09-29 | 2024-07-05 | 中国人民解放军总医院 | 一种血液中循环肿瘤细胞的灭活系统及应用 |
| KR102260995B1 (ko) * | 2018-11-28 | 2021-06-04 | 국립암센터 | Plk1의 활성 억제제를 유효성분으로 포함하는 암 예방 또는 치료용 약학적 조성물 |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2449003A (en) * | 1945-04-09 | 1948-09-07 | American Cyanamid Co | Water-soluble esters of riboflavin and preparation of same |
| JPH01104075A (ja) * | 1987-10-16 | 1989-04-21 | Mitsubishi Electric Corp | フラビン誘導体およびその製造方法 |
| JPH01132586A (ja) * | 1987-11-17 | 1989-05-25 | Hakusui Chem Ind Ltd | リボフラビン誘導体 |
| EP0364875A1 (de) * | 1988-10-19 | 1990-04-25 | BASF Aktiengesellschaft | Riboflavin-4'5'-cyclo-phosphorsäureesterchlorid, dessen Herstellung und Verwendung zur Herstellung von Riboflavin-5'-phosphat (5'-FMN) bzw. dessen Natriumsalz |
| CN1140992A (zh) * | 1993-10-19 | 1997-01-22 | 拉多帕思有限公司 | 用作抗病毒剂的黄素衍生物 |
Family Cites Families (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3189598A (en) * | 1960-01-14 | 1965-06-15 | Yagi Kunio | Fatty acid esters of riboflavin |
| JPS4955697A (zh) * | 1972-09-30 | 1974-05-30 | ||
| JPS6041611A (ja) * | 1983-08-17 | 1985-03-05 | Sankyo Co Ltd | 血中脂質低下剤 |
| US5554650A (en) * | 1994-07-28 | 1996-09-10 | Southern Research Institute | Antiphlogistic, analgesic, antipyretic injection preparation |
| GB9827727D0 (en) * | 1998-12-16 | 1999-02-10 | Pfizer Ltd | Antiparasitic formulations |
| US6245740B1 (en) * | 1998-12-23 | 2001-06-12 | Amgen Inc. | Polyol:oil suspensions for the sustained release of proteins |
| US20030105104A1 (en) * | 2001-11-27 | 2003-06-05 | Burzynski Stanislaw R. | Formulation of amino acids and riboflavin useful to reduce toxic effects of cytotoxic chemotherapy |
| US6565891B1 (en) * | 2002-08-23 | 2003-05-20 | Tsar Health Private Ltd. | Nutritional supplement for children |
-
2003
- 2003-07-29 US US10/522,110 patent/US20060293335A1/en not_active Abandoned
- 2003-07-29 AU AU2003257778A patent/AU2003257778A1/en not_active Abandoned
- 2003-07-29 WO PCT/CN2003/000609 patent/WO2004013142A1/zh not_active Ceased
- 2003-07-29 EP EP03766107A patent/EP1553099A4/en not_active Withdrawn
- 2003-07-29 CN CNB038137348A patent/CN100413866C/zh not_active Expired - Fee Related
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2449003A (en) * | 1945-04-09 | 1948-09-07 | American Cyanamid Co | Water-soluble esters of riboflavin and preparation of same |
| JPH01104075A (ja) * | 1987-10-16 | 1989-04-21 | Mitsubishi Electric Corp | フラビン誘導体およびその製造方法 |
| JPH01132586A (ja) * | 1987-11-17 | 1989-05-25 | Hakusui Chem Ind Ltd | リボフラビン誘導体 |
| EP0364875A1 (de) * | 1988-10-19 | 1990-04-25 | BASF Aktiengesellschaft | Riboflavin-4'5'-cyclo-phosphorsäureesterchlorid, dessen Herstellung und Verwendung zur Herstellung von Riboflavin-5'-phosphat (5'-FMN) bzw. dessen Natriumsalz |
| CN1140992A (zh) * | 1993-10-19 | 1997-01-22 | 拉多帕思有限公司 | 用作抗病毒剂的黄素衍生物 |
Non-Patent Citations (4)
| Title |
|---|
| KUNIO Y.: "Studies on fatty esters of flavins I. Chemical synthesis of fatty acid esters of riboflavin", THE JOURNAL OF VITAMINOLOGY, vol. 7, 1961, pages 276 - 280, XP003000832 * |
| MAO PU ET AL.: "Synthesis of di,. tri and tetra-stearoyl riboflavines", HUAXUE SHIJIE, no. 3, 2000, pages 142 - 144, XP003000833 * |
| See also references of EP1553099A4 * |
| WU XUEDONG ET AL.: "The activities of long-effect riboflavin in preventing mouth ulceration after chemical therapeutics for leukaemia for enfant", ZHONGGUO XIAOER XIEYE, vol. 2, no. 4, 1997, pages 190 * |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101684120B (zh) * | 2008-09-26 | 2013-07-31 | 深圳信立泰药业股份有限公司 | 核黄素-5'-月桂酸单酯晶型及其制备方法和含有该晶型的药物组合物 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP1553099A4 (en) | 2006-06-28 |
| US20060293335A1 (en) | 2006-12-28 |
| EP1553099A1 (en) | 2005-07-13 |
| CN100413866C (zh) | 2008-08-27 |
| CN1659169A (zh) | 2005-08-24 |
| AU2003257778A1 (en) | 2004-02-23 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| ES2994013T3 (en) | Tyk2 inhibitors and uses thereof | |
| CN116262759B (zh) | 嘧啶三环类化合物及其制备方法和医药用途 | |
| JP6947644B2 (ja) | 重水素化ケノデオキシコール酸誘導体およびこの化合物を含む薬物組成物 | |
| CN101429198B (zh) | 去氢骆驼蓬碱衍生物及其应用 | |
| CN117813312A (zh) | Shp2抑制剂、包含其的药物组合物及其用途 | |
| CN113784963B (zh) | 用作ret激酶抑制剂的化合物及其应用 | |
| JP7713253B2 (ja) | キサンチンオキシダーゼ抑制剤 | |
| CN115477681A (zh) | 一种五环三萜皂苷衍生物及制备方法与应用 | |
| US7354952B2 (en) | Pharmaceutical gallium compositions and methods | |
| EP3560928A1 (en) | Fused imidazole compound having indoleamine 2,3-dioxygenase inhibitory activity | |
| WO2004013142A1 (en) | A riboflavin derivative and its manufacture and uses | |
| WO2004026298A1 (en) | Derivatives of triptolide having high immunosuppressive effect and high water solubility, and uses thereof | |
| CN113214097A (zh) | 治疗阿尔茨海默病的化合物 | |
| CN108368117B (zh) | 一种取代的咪唑并喹唑啉化合物及其药物组合物 | |
| CN110857285B (zh) | 取代吡唑类化合物、其制备方法、药物组合物及用途 | |
| CN117715920A (zh) | 不对称gpr84拮抗剂及其用途 | |
| CN109206435B (zh) | 噻吩并[3,2-d]嘧啶类化合物及其制备方法和医药用途 | |
| CN112358517B (zh) | 一种苯并咪唑衍生物bi305及其制备方法和应用 | |
| WO2019233366A1 (zh) | 选择性a 2a受体拮抗剂 | |
| WO2022262857A1 (zh) | 芳基氧膦类化合物 | |
| CN118439935A (zh) | 一种苯丙烯醇类化合物及其制备方法和应用 | |
| CN119403801A (zh) | Lrrk2抑制剂 | |
| CN120737087A (zh) | 一种取代氮杂尿嘧啶类化合物、药物组合物及其用途 | |
| CN117126162A (zh) | 多取代杂环类化合物及其制备方法和应用 | |
| WO2025011457A1 (zh) | 一种稠合三环化合物及其制备方法和应用 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NI NO NZ OM PG PH PL PT RO RU SC SD SE SG SK SL SY TJ TM TN TR TT TZ UA UG US UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): GH GM KE LS MW MZ SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LU MC NL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 20038137348 Country of ref document: CN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2003766107 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2006293335 Country of ref document: US Ref document number: 10522110 Country of ref document: US |
|
| WWP | Wipo information: published in national office |
Ref document number: 2003766107 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: JP |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: JP |
|
| WWP | Wipo information: published in national office |
Ref document number: 10522110 Country of ref document: US |