WO2009151103A1 - ゴム組成物及びそれを用いたタイヤ - Google Patents
ゴム組成物及びそれを用いたタイヤ Download PDFInfo
- Publication number
- WO2009151103A1 WO2009151103A1 PCT/JP2009/060712 JP2009060712W WO2009151103A1 WO 2009151103 A1 WO2009151103 A1 WO 2009151103A1 JP 2009060712 W JP2009060712 W JP 2009060712W WO 2009151103 A1 WO2009151103 A1 WO 2009151103A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- rubber
- polymer
- block
- polymerization
- group
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L9/00—Compositions of homopolymers or copolymers of conjugated diene hydrocarbons
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B60—VEHICLES IN GENERAL
- B60C—VEHICLE TYRES; TYRE INFLATION; TYRE CHANGING; CONNECTING VALVES TO INFLATABLE ELASTIC BODIES IN GENERAL; DEVICES OR ARRANGEMENTS RELATED TO TYRES
- B60C1/00—Tyres characterised by the chemical composition or the physical arrangement or mixture of the composition
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L15/00—Compositions of rubber derivatives
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L21/00—Compositions of unspecified rubbers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L53/00—Compositions of block copolymers containing at least one sequence of a polymer obtained by reactions only involving carbon-to-carbon unsaturated bonds; Compositions of derivatives of such polymers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L7/00—Compositions of natural rubber
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08C—TREATMENT OR CHEMICAL MODIFICATION OF RUBBERS
- C08C19/00—Chemical modification of rubber
- C08C19/30—Addition of a reagent which reacts with a hetero atom or a group containing hetero atoms of the macromolecule
- C08C19/42—Addition of a reagent which reacts with a hetero atom or a group containing hetero atoms of the macromolecule reacting with metals or metal-containing groups
- C08C19/44—Addition of a reagent which reacts with a hetero atom or a group containing hetero atoms of the macromolecule reacting with metals or metal-containing groups of polymers containing metal atoms exclusively at one or both ends of the skeleton
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K3/00—Use of inorganic substances as compounding ingredients
- C08K3/01—Use of inorganic substances as compounding ingredients characterized by their specific function
- C08K3/013—Fillers, pigments or reinforcing additives
Definitions
- the present invention relates to a rubber composition and a tire using the rubber composition, and more particularly to a rubber composition capable of improving the fracture characteristics and wear resistance of the tire.
- the phase separation interface present in the rubber composition for a tire tread can be mainly divided into a filler / polymer interface and a polymer / polymer interface.
- Patent Document 1 an incompatible polymer blend system having a phase separation interface between a polymer and a polymer has a plurality of blocks, and each block is compatible with a different polymer phase. It is disclosed that the interfacial affinity between a polymer and a polymer is improved by blending a block copolymer, which can improve fracture characteristics, wear resistance, and the like.
- the filler / polymer interface existing in the rubber composition tends to concentrate strain at the time of rubber deformation as compared with the polymer / polymer interface, and the filler / polymer interface. It was found that the interface of was likely to be the starting point of fracture. Therefore, the influence of the polymer / polymer interface is smaller than that of the filler / polymer interface, and the effect of improving the polymer / polymer interface affinity has not been achieved effectively.
- an object of the present invention is to effectively suppress the strain concentrated on the filler / polymer interface, thereby effectively exhibiting the effect of improving the polymer / polymer interface affinity, and to provide the tire fracture characteristics and wear resistance. It is providing the rubber composition which can improve this. Another object of the present invention is to provide a tire excellent in fracture characteristics and wear resistance using the rubber composition in a tread portion.
- the present inventor has found that the incompatible polymer blend forming the polymer phase A and the polymer phase B is compatible with the polymer phase A but not with the polymer phase B.
- At least one rubber component comprising a block copolymer comprising a block a that is soluble and a block b that is compatible with the polymer phase B but is incompatible with the polymer phase A, and further comprises an incompatible polymer blend
- the inventors have found that the introduction of at least one functional group into can improve fracture characteristics and wear resistance, and have completed the present invention.
- the rubber composition of the present invention is composed of at least two kinds of rubber components, and with respect to the incompatible polymer blend forming the polymer phase A and the polymer phase B, A block copolymer comprising a block a comprising the same monomer unit as the monomer unit forming the polymer phase A and a block b comprising the same monomer unit as the monomer unit forming the polymer phase B Blended, At least one of the rubber components has at least one functional group.
- incompatibility of polymer phase A and polymer phase B of incompatible polymer blend incompatibility of block copolymer block a and block b, and block of polymer phase A or polymer phase B and block copolymer of incompatible polymer blend
- the incompatibility of b or block a is determined by pressing a rubber composition composed of the target combination into a sheet using a test roll and pressing it at 150 ° C. for 30 minutes in a 15 cm ⁇ 15 cm ⁇ 1 cm test mold. Vulcanization is performed to prepare a vulcanized sheet, and the obtained vulcanized sheet is evaluated by observing it with a microtome as an ultrathin section using a scanning probe microscope.
- phase separation structure If the phase separation structure is confirmed, the polymer phase and / or block is incompatible. In order to determine compatibility or incompatibility, it is possible to determine whether the peak is bimodal from the temperature dispersion curve of tan ⁇ , or whether multiple glass transition temperatures of the blend polymer are observed by DSC measurement. A method for determining whether or not the phase separation structure extends to several tens of microns can be used.
- one of the polymer phase A and the polymer phase B contains natural rubber and / or polyisoprene rubber, and the other polymer phase contains synthetic rubber other than polyisoprene rubber.
- the synthetic rubber other than the polyisoprene rubber is preferably a polybutadiene rubber.
- the weight average molecular weight of the rubber component is 200,000 or more, and the weight average molecular weights of the block a and the block b forming the block copolymer are both 50,000 to 500,000.
- the “weight average molecular weight” is a value in terms of polystyrene measured by gel permeation chromatography (GPC).
- the blending amount of the block copolymer is preferably 0.3 to 20 parts by mass with respect to a total of 100 parts by mass of the rubber components.
- the tire of the present invention is characterized in that the rubber composition is used for any tire member.
- the tire member is preferably a tread portion, a sidewall portion, a side reinforcing rubber, a bead portion, or a rubber chafer.
- block a which is compatible with polymer phase A but incompatible with polymer phase B, and polymer phase B
- block b which is compatible but incompatible with polymer phase A
- at least one functional group is introduced into at least one rubber component constituting the incompatible polymer blend.
- the rubber composition of the present invention is composed of at least two kinds of rubber components, and the same amount as the monomer unit forming the polymer phase A with respect to the incompatible polymer blend forming the polymer phase A and the polymer phase B.
- a block copolymer comprising a block a comprising a body unit and a block b comprising a monomer unit identical to the monomer unit forming the polymer phase B, wherein at least one of the rubber components is at least one It has one functional group.
- the reinforcing effect between the filler and the polymer widely known as a bound rubber is increased.
- the effect of improving the polymer / polymer interface affinity produced by the blending of the block copolymer with the incompatible polymer blend is effective. As a result, the fracture characteristics and wear resistance of the tire can be greatly improved.
- the incompatible polymer blend of the rubber composition of the present invention comprises at least two kinds of rubber components, forms a polymer phase A and a polymer phase B, and the polymer phase A and the polymer phase B are incompatible.
- the rubber component include natural rubber (NR) and synthetic rubber.
- Specific examples of the synthetic rubber include polyisoprene rubber (IR), styrene-butadiene copolymer rubber (SBR), and polybutadiene rubber. (BR), ethylene-propylene-diene rubber (EPDM), chloroprene rubber (CR), isobutylene isoprene rubber (IIR), halogenated butyl rubber, acrylonitrile-butadiene rubber (NBR), and the like.
- the rubber component may be either an unmodified polymer or a modified polymer, but at least one of the rubber components constituting the polymer phase A and the polymer phase B has at least one functional group (modified polymer). Is required).
- a functional group a functional group having an affinity for a filler such as carbon black and silica is preferable, and a functional group containing tin, a functional group containing silicon, and a functional group containing nitrogen are more preferable.
- the ratio of the rubber component having at least one functional group in the incompatible polymer blend is preferably 20% by mass or more from the viewpoint of reinforcing the filler / polymer interface.
- one of the polymer phase A and the polymer phase B contains natural rubber and / or polyisoprene rubber, and the other polymer phase is polyisoprene rubber. It is preferable to contain a synthetic rubber other than the above. That is, an incompatible polymer blend can be easily formed by using natural rubber and / or polyisoprene rubber and synthetic rubber other than the polyisoprene rubber as the rubber component of the rubber composition of the present invention. . Moreover, as a synthetic rubber other than polyisoprene rubber, polybutadiene rubber is preferable.
- SBR / BR SBR / NR, and the like may be mentioned as a combination of the rubber component constituting the polymer phase A and the polymer phase B forming the incompatible polymer blend.
- the object of the present invention can be achieved by using any of the above rubber components as the modified polymer.
- the production method of the modified natural rubber is not particularly limited.
- a polar group-containing monomer is added to natural rubber latex, and the polar group-containing monomer is added.
- the natural rubber latex used for the production of the modified natural rubber is not particularly limited.
- field latex ammonia-treated latex, centrifugal concentrated latex, deproteinized latex treated with a surfactant or an enzyme, and a combination thereof. A thing etc. can be used.
- the polar group-containing monomer added to the natural rubber latex is not particularly limited as long as it has at least one polar group in the molecule and can be graft-polymerized with the natural rubber molecule.
- the polar group-containing monomer preferably has a carbon-carbon double bond in the molecule for graft polymerization with a natural rubber molecule, and is preferably a polar group-containing vinyl monomer. .
- polar group examples include amino group, imino group, nitrile group, ammonium group, imide group, amide group, hydrazo group, azo group, diazo group, hydroxyl group, carboxyl group, carbonyl group, epoxy group, oxycarbonyl
- Preferred examples include a group, sulfide group, disulfide group, sulfonyl group, sulfinyl group, thiocarbonyl group, nitrogen-containing heterocyclic group, oxygen-containing heterocyclic group and alkoxysilyl group.
- monomers containing polar groups may be used alone or in combination of two or more.
- Examples of the monomer containing an amino group include polymerizable monomers containing at least one amino group selected from primary, secondary, and tertiary amino groups in one molecule.
- a tertiary amino group-containing monomer such as dialkylaminoalkyl (meth) acrylate is particularly preferable.
- These amino group-containing monomers may be used alone or in a combination of two or more.
- acrylamide methacrylamide, 4-vinylaniline, aminomethyl (meth) acrylate, aminoethyl (meth) acrylate, aminopropyl (meth) acrylate, aminobutyl (meth) ) Acrylate and the like.
- the secondary amino group-containing monomer includes (1) anilinostyrene, ⁇ -phenyl-p-anilinostyrene, ⁇ -cyano-p-anilinostyrene, ⁇ -cyano- ⁇ -methyl-p. -Anilinostyrene, ⁇ -chloro-p-anilinostyrene, ⁇ -carboxy-p-anilinostyrene, ⁇ -methoxycarbonyl-p-anilinostyrene, ⁇ - (2-hydroxyethoxy) carbonyl-p-anilino
- Anilinostyrenes such as styrene, ⁇ -formyl-p-anilinostyrene, ⁇ -formyl- ⁇ -methyl-p-anilinostyrene, ⁇ -carboxy- ⁇ -carboxy- ⁇ -phenyl-p-anilinostyrene, (2) 1-anilinophenyl-1,3-butadiene, 1-anil
- examples of the tertiary amino group-containing monomer include N, N-disubstituted aminoalkyl (meth) acrylate and N, N-disubstituted aminoalkyl (meth) acrylamide.
- examples of the N, N-disubstituted aminoalkyl (meth) acrylate include N, N-dimethylaminoethyl (meth) acrylate, N, N-diethylaminoethyl (meth) acrylate, N, N-dipropylaminoethyl (meth)
- Preferable examples include acrylate, N, N-dioctylaminoethyl (meth) acrylate, N-methyl-N-ethylaminoethyl (meth) acrylate, and the like.
- N, N-disubstituted aminoalkyl (meth) acrylamide examples include N, N-dimethylaminopropyl (meth) acrylamide, N, N-diethylaminopropyl (meth) acrylamide, N, N-dioctylaminopropyl (meta) Preferred examples include acrylamide.
- Examples of the monomer containing a nitrile group include (meth) acrylonitrile, vinylidene cyanide and the like. These nitrile group-containing monomers may be used alone or in a combination of two or more.
- Examples of the monomer containing a hydroxyl group include polymerizable monomers having at least one primary, secondary, and tertiary hydroxyl group in one molecule.
- Examples of such monomers include hydroxyl group-containing unsaturated carboxylic acid monomers, hydroxyl group-containing vinyl ether monomers, hydroxyl group-containing vinyl ketone monomers, and the like.
- specific examples of the hydroxyl group-containing monomer include 2-hydroxyethyl (meth) acrylate, 2-hydroxypropyl (meth) acrylate, 3-hydroxypropyl (meth) acrylate, and 2-hydroxybutyl (meth) acrylate.
- Hydroxyalkyl (meth) acrylates such as 3-hydroxybutyl (meth) acrylate and 4-hydroxybutyl (meth) acrylate; polyalkylene glycols such as polyethylene glycol and polypropylene glycol (the number of alkylene glycol units is, for example, 2 to 23 Mono (meth) acrylates; N-hydroxymethyl (meth) acrylamide, N- (2-hydroxyethyl) (meth) acrylamide, N, N-bis (2-hydroxyethyl) (meth) acrylamide, etc.
- Hydroxyl group-containing unsaturated amides o-hydroxy Hydroxyl group-containing vinyl such as tylene, m-hydroxystyrene, p-hydroxystyrene, o-hydroxy- ⁇ -methylstyrene, m-hydroxy- ⁇ -methylstyrene, p-hydroxy- ⁇ -methylstyrene, p-vinylbenzyl alcohol Aromatic compounds etc. are mentioned. Among these, hydroxyl group-containing unsaturated carboxylic acid monomers, hydroxyalkyl (meth) acrylates, and hydroxyl group-containing vinyl aromatic compounds are preferable, and hydroxyl group-containing unsaturated carboxylic acid monomers are particularly preferable.
- examples of the hydroxyl group-containing unsaturated carboxylic acid-based monomer include esters such as acrylic acid, methacrylic acid, itaconic acid, fumaric acid, maleic acid, amides, and anhydrides. Among these, Particularly preferred are esters such as acrylic acid and methacrylic acid. These hydroxyl group-containing monomers may be used alone or in a combination of two or more.
- Examples of the monomer containing a carboxyl group include unsaturated carboxylic acids such as (meth) acrylic acid, maleic acid, fumaric acid, itaconic acid, tetraconic acid and cinnamic acid; non-phthalic acids such as phthalic acid, succinic acid and adipic acid.
- unsaturated carboxylic acids such as (meth) acrylic acid, maleic acid, fumaric acid, itaconic acid, tetraconic acid and cinnamic acid
- non-phthalic acids such as phthalic acid, succinic acid and adipic acid.
- free carboxyl group-containing esters such as monoesters of a polymerizable polycarboxylic acid and a hydroxyl group-containing unsaturated compound such as (meth) allyl alcohol and 2-hydroxyethyl (meth) acrylate, and salts thereof.
- unsaturated carboxylic acids are particularly preferred.
- These carboxyl group-containing monomers may be used
- Examples of the monomer containing an epoxy group include (meth) allyl glycidyl ether, glycidyl (meth) acrylate, and 3,4-oxycyclohexyl (meth) acrylate. These epoxy group-containing monomers may be used alone or in combination of two or more.
- the nitrogen-containing heterocyclic ring includes pyrrole, histidine, imidazole, triazolidine, triazole, triazine, pyridine, pyrimidine, pyrazine, indole, quinoline, purine, phenazine, pteridine, Examples include melamine.
- the nitrogen-containing heterocycle may contain other heteroatoms in the ring.
- a monomer containing a pyridyl group as a nitrogen-containing heterocyclic group 2-vinylpyridine, 3-vinylpyridine, 4-vinylpyridine, 5-methyl-2-vinylpyridine, 5-ethyl-2- Examples include vinyl compounds containing a pyridyl group such as vinyl pyridine, among which 2-vinyl pyridine, 4-vinyl pyridine and the like are particularly preferable.
- These nitrogen-containing heterocyclic group-containing monomers may be used alone or in a combination of two or more.
- Examples of the monomer containing the alkoxysilyl group include (meth) acryloxymethyltrimethoxysilane, (meth) acryloxymethylmethyldimethoxysilane, (meth) acryloxymethyldimethylmethoxysilane, (meth) acryloxymethyltrimethoxysilane.
- the graft polymerization of the polar group-containing monomer to the natural rubber molecule is performed by emulsion polymerization.
- the polar group-containing monomer is added to a solution obtained by adding water and, if necessary, an emulsifier to a natural rubber latex, and a polymerization initiator is further added. It is preferable to polymerize the polar group-containing monomer by stirring at the temperature.
- an emulsifier may be added to the natural rubber latex in advance, or after emulsifying the polar group-containing monomer with the emulsifier, May be added.
- an emulsifier which can be used for emulsification of a natural rubber latex and / or a polar group containing monomer
- Nonionic surfactants such as polyoxyethylene lauryl ether, are mentioned.
- the polymerization initiator is not particularly limited, and various polymerization initiators for emulsion polymerization can be used, and the addition method is not particularly limited.
- Examples of commonly used polymerization initiators include benzoyl peroxide, hydrogen peroxide, cumene hydroperoxide, tert-butyl hydroperoxide, di-tert-butyl peroxide, 2,2-azobisisobutyronitrile, 2,2-azobis (2-diaminopropane) hydrochloride, 2,2-azobis (2-diaminopropane) dihydrochloride, 2,2-azobis (2,4-dimethylvaleronitrile), potassium persulfate, sodium persulfate And ammonium persulfate.
- a redox polymerization initiator In order to lower the polymerization temperature, it is preferable to use a redox polymerization initiator.
- the reducing agent to be combined with the peroxide in the redox polymerization initiator include tetraethylenepentamine, mercaptans, acidic sodium sulfite, reducing metal ions, ascorbic acid and the like.
- a preferred combination of a peroxide and a reducing agent in the redox polymerization initiator includes a combination of tert-butyl hydroperoxide and tetraethylenepentamine.
- the addition amount of the polymerization initiator is preferably in the range of 1 to 100 mol%, more preferably in the range of 10 to 100 mol% with respect to the polar group-containing monomer.
- the above-mentioned components are charged into a reaction vessel and reacted at 30 to 80 ° C. for 10 minutes to 7 hours to obtain a modified natural rubber latex in which the polar group-containing monomer is graft copolymerized with natural rubber molecules.
- the modified natural rubber latex is coagulated, washed, and then dried using a dryer such as a vacuum dryer, an air dryer, a drum dryer or the like to obtain a modified natural rubber.
- the coagulant used for coagulating the modified natural rubber latex is not particularly limited, and examples thereof include acids such as formic acid and sulfuric acid, and salts such as sodium chloride.
- the graft amount of the polar group-containing monomer is preferably in the range of 0.01 to 5.0% by mass, preferably in the range of 0.01 to 1.0% by mass with respect to the rubber component in the natural rubber latex. Is more preferable. If the graft amount of the polar group-containing monomer is less than 0.01% by mass, the low loss and wear resistance of the rubber composition may not be sufficiently improved. If the graft amount of the polar group-containing monomer exceeds 5.0% by mass, natural physical properties such as viscoelasticity and SS characteristics (stress-strain curve in a tensile tester) will be greatly changed. The natural physical properties inherent to natural rubber are impaired, and the processability of the rubber composition may be greatly deteriorated.
- the modified synthetic rubber is not particularly limited, but is a conjugated diene polymer, that is, a conjugated diene compound homopolymer or an aromatic vinyl compound and a conjugated diene compound.
- a conjugated diene compound as a monomer alone or a mixture of a monomeric aromatic vinyl compound and a conjugated diene compound is used as a polymerization initiator.
- the polymerization active site is modified with various modifiers, and (2) the monomer is polymerized using a polymerization initiator having a functional group.
- the conjugated diene polymer having a polymerization active site may be one produced by anionic polymerization or one produced by coordination polymerization.
- Examples of the conjugated diene compound as a monomer include 1,3-butadiene, isoprene, 1,3-pentadiene, 2,3-dimethylbutadiene, 2-phenyl-1,3-butadiene, 1,3-hexadiene, and the like. Among these, 1,3-butadiene and isoprene are preferable.
- examples of the aromatic vinyl compound as a monomer include styrene, p-methylstyrene, m-methylstyrene, p-tert-butylstyrene, ⁇ -methylstyrene, chloromethylstyrene, vinyltoluene and the like.
- the polymerization initiator is preferably a lithium compound, more preferably hydrocarbyl lithium and a lithium amide compound.
- hydrocarbyl lithium is used as the polymerization initiator, a polymer having a hydrocarbyl group at the polymerization initiation terminal and the other terminal being a polymerization active site is obtained.
- a lithium amide compound is used as a polymerization initiator, a polymer having a nitrogen-containing functional group at the polymerization initiation terminal and a polymerization active site at the other terminal is obtained, and the polymer is modified with a modifier. And can be used as a modified synthetic rubber.
- the amount of the polymerization initiator used is preferably in the range of 0.2 to 20 mmol per 100 g of monomer.
- the lithium amide compounds include lithium hexamethylene imide, lithium pyrrolidide, lithium piperidide, lithium heptamethylene imide, lithium dodecamethylene imide, lithium dimethylamide, lithium diethylamide, lithium dipropylamide, lithium dibutylamide, lithium Dihexylamide, lithium diheptylamide, lithium dioctylamide, lythym-2-ethylhexylamide, lithium didecylamide, lithium-N-methylpiperazide, lithium ethylpropylamide, lithium ethylbutyramide, lithium methylbutyramide, lithium ethylbenzylamide, Lithium methylphenethyl amide and the like can be mentioned.
- lithium hexamethylene imide, lithium pyrrolidide, lithium Cyclic lithium amide compounds such as mupperizide, lithium heptamethylene imide and lithium dodecamethylene imide are preferred, and lithium hexamethylene imide and lithium pyrrolidide are particularly preferred.
- a modified synthetic rubber can be produced by polymerizing monomers in a hydrocarbon solvent inert to the polymerization reaction.
- hydrocarbon solvents inert to the polymerization reaction include propane, n-butane, isobutane, n-pentane, isopentane, n-hexane, cyclohexane, propene, 1-butene, isobutene, trans-2-butene, cis -2-butene, 1-pentene, 2-pentene, 1-hexene, 2-hexene, benzene, toluene, xylene, ethylbenzene, methylcyclopentane, methylcyclohexane and the like. These may be used alone or in combination of two or more.
- the anionic polymerization may be performed in the presence of a randomizer.
- the randomizer can control the microstructure of the conjugated diene compound portion of the polymer. More specifically, the randomizer can control the amount of vinyl bonds in the conjugated diene compound portion of the polymer, or can control the conjugated diene content in the polymer. It has the effect of randomizing the compound unit and the aromatic vinyl compound unit.
- randomizer examples include dimethoxybenzene, tetrahydrofuran, dimethoxyethane, diethylene glycol dibutyl ether, diethylene glycol dimethyl ether, bistetrahydrofurylpropane, triethylamine, pyridine, N-methylmorpholine, N, N, N ′, N′-tetramethylethylenediamine, 1 , 2-dipiperidinoethane, potassium-t-amylate, potassium-t-butoxide, sodium-t-amylate and the like.
- the amount of these randomizers used is preferably in the range of 0.01 to 100 molar equivalents per mole of polymerization initiator. Moreover, you may mix and use.
- the anionic polymerization is preferably carried out by solution polymerization, and the concentration of the monomer in the polymerization reaction solution is preferably in the range of 5 to 50% by mass, and more preferably in the range of 10 to 30% by mass.
- the content of the aromatic vinyl compound in the monomer mixture may be appropriately selected according to the amount of the aromatic vinyl compound in the target copolymer. it can.
- the polymerization mode is not particularly limited, and may be batch type or continuous type.
- the polymerization temperature for the anionic polymerization is preferably in the range of 0 to 150 ° C, more preferably in the range of 20 to 130 ° C.
- the polymerization can be carried out under a generated pressure, but it is usually preferred to carry out the polymerization under a pressure sufficient to keep the monomer used in a substantially liquid phase.
- a rare earth metal compound as a polymerization initiator, and the following (a) component, (b) component, and (c) component may be used in combination. Further preferred.
- this coordination polymerization a conjugated diene polymer having a polymerization active site is obtained.
- the component (a) used for the coordination polymerization is selected from a rare earth metal compound, a complex compound of a rare earth metal compound and a Lewis base, and the like.
- rare earth metal compounds include rare earth element carboxylates, alkoxides, ⁇ -diketone complexes, phosphates and phosphites
- Lewis bases include acetylacetone, tetrahydrofuran, pyridine, N, N. -Dimethylformamide, thiophene, diphenyl ether, triethylamine, organophosphorus compound, monovalent or divalent alcohol and the like.
- the rare earth element of the rare earth metal compound lanthanum, neodymium, praseodymium, samarium and gadolinium are preferable, and among these, neodymium is particularly preferable.
- Specific examples of the component (a) include neodymium tri-2-ethylhexanoate, complex compounds thereof with acetylacetone, neodymium trineodecanoate, complex compounds thereof with acetylacetone, neodymium tri-n-butoxide, and the like. It is done.
- These (a) components may be used individually by 1 type, or 2 or more types may be mixed and used for them.
- the component (b) used for the coordination polymerization is selected from organoaluminum compounds.
- organoaluminum compound a trihydrocarbyl aluminum compound represented by the formula: R 3 Al, a hydrocarbyl aluminum hydride represented by the formula: R 2 AlH or RAlH 2 (wherein R is independently And hydrocarbylaluminoxane compounds having a hydrocarbon group having 1 to 30 carbon atoms.
- the organoaluminum compound include trialkylaluminum, dialkylaluminum hydride, alkylaluminum dihydride, alkylaluminoxane, and the like. These compounds may be used alone or in combination of two or more.
- (b) component it is preferable to use aluminoxane and another organoaluminum compound together.
- the component (c) used in the coordination polymerization is a compound having a hydrolyzable halogen or a complex compound thereof with a Lewis base; an organic halide having a tertiary alkyl halide, benzyl halide or allyl halide; a non-coordinating anion And an ionic compound comprising a counter cation.
- Specific examples of the component (c) include alkylaluminum dichloride, dialkylaluminum chloride, silicon tetrachloride, tin tetrachloride, complexes of zinc chloride with Lewis bases such as alcohol, magnesium chloride and Lewis such as alcohol.
- Examples thereof include complexes with bases, benzyl chloride, t-butyl chloride, benzyl bromide, t-butyl bromide, triphenylcarbonium tetrakis (pentafluorophenyl) borate and the like.
- bases benzyl chloride, t-butyl chloride, benzyl bromide, t-butyl bromide, triphenylcarbonium tetrakis (pentafluorophenyl) borate and the like.
- the polymerization initiator is preliminarily used, if necessary, using the same conjugated diene compound and / or aromatic vinyl compound as the polymerization monomer. May be prepared. Further, part or all of the component (a) or the component (c) may be supported on an inert solid. The amount of each of the above components can be appropriately set. Usually, the component (a) is 0.001 to 0.5 mmol per 100 g of the monomer. The molar ratio of component (b) / component (a) is preferably 5 to 1000, and component (c) / component (a) is preferably in the range of 0.5 to 10.
- the polymerization temperature in the coordination polymerization is preferably in the range of -80 to 150 ° C, more preferably in the range of -20 to 120 ° C.
- a hydrocarbon solvent inert to the reaction exemplified in the above-mentioned anionic polymerization can be used, and the concentration of the monomer in the reaction solution is the same as in the case of anionic polymerization.
- the reaction pressure in coordination polymerization is the same as that in the case of anionic polymerization, and it is desirable that the raw material used for the reaction substantially removes reaction inhibitors such as water, oxygen, carbon dioxide, and protic compounds.
- the modifier used is preferably a nitrogen-containing compound, a silicon-containing compound or a tin-containing compound.
- a nitrogen-containing functional group, a silicon-containing functional group, or a tin-containing functional group can be introduced by a modification reaction.
- the polymer having the polymerization active site is synthesized using a polymerization initiator having a functional group and already has at least one functional group, the polymerization terminator usually used in the polymerization reaction is reacted. In addition to the system, the polymerization reaction may be stopped.
- the nitrogen-containing compound that can be used as the modifier preferably has a substituted or unsubstituted amino group, amide group, imino group, imidazole group, nitrile group, or pyridyl group.
- Suitable nitrogen-containing compounds as the modifier include isocyanate compounds such as diphenylmethane diisocyanate, crude MDI, trimethylhexamethylene diisocyanate, tolylene diisocyanate, 4,4′-bis (diethylamino) benzophenone, 4- (dimethylamino) benzophenone, Examples include 4- (diethylamino) benzophenone, 4-dimethylaminobenzylideneaniline, 4-dimethylaminobenzylidenebutylamine, dimethylimidazolidinone, N-methylpyrrolidone, and 1-butylaziridine.
- silicon-containing compound that can be used as the modifier examples include 3-glycidoxypropyltrimethoxysilane, 3-glycidoxypropyltriethoxysilane, and N- (1-methylpropylidene) -3- (tri Ethoxysilyl) -1-propanamine, N- (1,3-dimethylbutylidene) -3- (triethoxysilyl) -1-propanamine, N- (3-triethoxysilylpropyl) -4,5-dihydro Imidazole, 3-methacryloyloxypropyltrimethoxysilane, 3-isocyanatopropyltriethoxysilane, 3-triethoxysilylpropyl succinic anhydride, 3- (1-hexamethyleneimino) propyl (triethoxy) silane, (1- Hexamethyleneimino) methyl (trimethoxy) silane, 3-diethylaminopropyl (triethoxy) si
- the coupling agent represented is also preferred.
- R 1 in formula (I) include a methyl group, an ethyl group, an n-butyl group, a neophyll group, a cyclohexyl group, an n-octyl group, and a 2-ethylhexyl group.
- the coupling agent of the formula (I) specifically, SnCl 4 , R 1 SnCl 3 , R 1 2 SnCl 2 , R 1 3 SnCl, SiCl 4 , R 1 SiCl 3 , R 1 2 SiCl 2 , R 1 3 SiCl and the like are preferable, and SnCl 4 and SiCl 4 are particularly preferable.
- the modification reaction of the polymerization active site by the modifier is preferably performed by a solution reaction, and the solution may contain a monomer used at the time of polymerization.
- the reaction mode of the modification reaction is not particularly limited, and may be a batch type or a continuous type.
- the reaction temperature of the modification reaction is not particularly limited as long as the reaction proceeds, and the reaction temperature of the polymerization reaction may be employed as it is.
- the amount of the modifying agent used is preferably in the range of 0.25 to 3.0 mol, more preferably in the range of 0.5 to 1.5 mol, with respect to 1 mol of the polymerization initiator used for producing the polymer.
- the rubber component constituting the incompatible polymer blend preferably has a weight average molecular weight of 200,000 or more. If the weight average molecular weight of the rubber component is less than 200,000, the unvulcanized viscosity will be too low, the torque during kneading will not be applied, and the kneading may be insufficient.
- the block copolymer used in the rubber composition of the present invention comprises a block a comprising the same monomer unit as the monomer unit forming the polymer phase A, and a single monomer having the same monomer unit forming the polymer phase B. It consists of a block b consisting of body units.
- each polymer phase is composed of a rubber component formed from two or more types of monomer units, or is composed of two or more types of rubber components, the corresponding block is also composed of two or more types of monomers. It will be formed from units.
- the block a of the block copolymer is formed from the same monomer unit as the monomer unit forming the polymer phase A, it is compatible with the polymer phase A but incompatible with the polymer phase B.
- the block b of the block copolymer is formed from the same monomer unit as the monomer unit forming the polymer phase B, so that it is compatible with the polymer phase B but incompatible with the polymer phase A. . That is, the block a and the block b are incompatible with each other, but this allows each block to enter only the corresponding polymer phase, thereby improving fracture characteristics, wear resistance, and the like.
- the block copolymer is not particularly limited, but a conjugated diene polymer, that is, a polymer of a conjugated diene compound or a polymer of an aromatic vinyl compound and a conjugated diene compound is preferable.
- the block copolymer is obtained by, for example, polymerizing a monomer conjugated diene compound or a mixture of a monomer aromatic vinyl compound and a conjugated diene compound using a polymerization initiator, thereby converting the monomer into a polymer.
- a block a is produced by carrying out a polymerization reaction to a rate of at least 5% by mass, preferably 10% by mass or more, and then a conjugated diene compound as a monomer or a mixture of an aromatic vinyl compound as a monomer and a conjugated diene compound Can be obtained by a method of producing a block b by further polymerization.
- the block copolymer may be produced by anionic polymerization or may be produced by coordination polymerization.
- the block copolymer may be an unmodified polymer or a modified polymer.
- the form of the modified polymer obtained varies depending on the type of the modifying agent, the form of the modified polymer is not particularly limited, and may take any form such as a linear polymer, a branched polymer, and a star polymer. Of these modified polymer forms, linear polymers are preferred.
- conjugated diene compound, aromatic vinyl compound and polymerization initiator examples include those exemplified as the conjugated diene compound, aromatic vinyl compound, and polymerization initiator that can be used in the modified synthetic rubber. The same thing can be illustrated.
- the anionic polymerization, coordination polymerization and modification reaction can also be carried out in the same manner as described in the method for producing the modified synthetic rubber.
- the weight average molecular weights of the block a and the block b are preferably 50,000 to 500,000, respectively.
- the weight average molecular weight of the block a or the block b is less than 50,000, it is difficult to be entangled with the rubber component constituting the incompatible polymer blend, and the effect of improving the interfacial affinity between the polymer / polymer cannot be sufficiently secured.
- it exceeds 500,000 it tends to be difficult to synthesize block copolymers.
- the block a and the block b forming the block copolymer are not particularly limited, but preferably have the same weight average molecular weight.
- the weight average molecular weight of each block refers to the weight average molecular weight of each polymer obtained by synthesizing polymers using the block a and the block b as models.
- the blending amount of the block copolymer is preferably 0.3 to 20 parts by mass, and 0.3 to 9 parts by mass with respect to a total of 100 parts by mass of the rubber components constituting the incompatible polymer blend. More preferably.
- the blending amount of the block copolymer with respect to 100 parts by mass of the rubber component is less than 0.3 parts by mass, the effect of improving the interfacial affinity between the polymer and the polymer cannot be sufficiently obtained, while when it exceeds 20 parts by mass, the rubber composition There exists a tendency for the physical property of a physical property to fall.
- the rubber composition of the present invention preferably further contains 10 to 100 parts by weight, more preferably 20 to 80 parts by weight of the filler with respect to 100 parts by weight of the total rubber components constituting the incompatible polymer blend. Further preferred. If the blending amount of the filler with respect to 100 parts by mass of the rubber component is less than 10 parts by mass, the fracture characteristics and wear resistance of the vulcanized rubber are not sufficient, while if it exceeds 100 parts by mass, the workability deteriorates. Tend.
- carbon black and silica are preferable.
- the carbon black is preferably FEF, SRF, HAF, ISAF, or SAF grade, and more preferably HAF, ISAF, or SAF grade.
- silica wet silica and dry silica are preferable, and wet silica is more preferable.
- the rubber composition of the present invention comprises, in addition to the above-mentioned rubber component, a block copolymer, a filler, a compounding agent usually used in the rubber industry, such as a silane coupling agent, a softener, stearic acid, an anti-aging agent, zinc Hana, a vulcanization accelerator, a vulcanizing agent, and the like can be produced by appropriately selecting and blending them within a range that does not impair the object of the present invention, and kneading, heating, extruding, and the like.
- a compounding agent usually used in the rubber industry such as a silane coupling agent, a softener, stearic acid, an anti-aging agent, zinc Hana, a vulcanization accelerator, a vulcanizing agent, and the like.
- the tire of the present invention is characterized by using the above-described rubber composition in the tread portion.
- the tire uses a rubber composition in the tread portion that suppresses strain concentrated on the filler / polymer interface and effectively exhibits an effect of improving the polymer / polymer interface affinity, and has a tensile strength. Fracture characteristics such as (Tb) and elongation at break (Eb) and wear resistance are greatly improved.
- the tire of the present invention is not particularly limited except that the above rubber composition is used for the tread portion, and can be produced according to a conventional method.
- inert gas such as nitrogen, argon, helium other than normal or the air which adjusted oxygen partial pressure, can be used.
- HC block> A nitrogen-substituted 5 liter autoclave was charged with 2.4 kg of cyclohexane and 100 g of 1,3-butadiene (block a component) in a nitrogen atmosphere, and the catalyst A was further added to the resulting solution. Polymerization was carried out for minutes. The reaction conversion rate of 1,3-butadiene at this time was almost 100%. Next, a cyclohexane solution containing 100 g of isoprene (block b component) was added to the polymerization reaction system, and polymerization was further performed for 60 minutes.
- HC block block
- the weight average molecular weight (Mw) of a 100,000
- the weight average molecular weight (Mw) of block b 100,000
- the HC block from which the weight average molecular weights of the block a and the block b differ was synthesize
- LC Block> In a pressure-resistant glass container with an internal volume of about 900 ml that has been dried and purged with nitrogen, 283 g of cyclohexane, 35 g of 1,3-butadiene (component of block a), 0.015 mmol of 2,2-ditetrahydrofurylpropane are injected as a cyclohexane solution, and n After adding 0.50 mmol of -butyllithium (n-BuLi), a polymerization reaction was carried out in a hot water bath at 50 ° C. equipped with a stirrer for 1.5 hours. The reaction conversion rate of 1,3-butadiene at this time was almost 100%.
- weight average molecular weight (Mw) and microstructure of the polymer (rubber component and block copolymer) produced as described above were measured by the following methods.
- Weight average molecular weight (Mw) Gel permeation chromatography [GPC: Tosoh HLC-8020, column: Tosoh GMH-XL (two in series), detector: differential refractometer (RI)] on the basis of monodisperse polystyrene, The weight average molecular weight (Mw) in terms of polystyrene in the state without denaturation stop was determined. In addition, about the block copolymer, the weight average molecular weight (Mw) of each block of a block copolymer was calculated
- microstructure of the polymer was determined by an infrared method (Morero method).
- Elongation at break Elongation at break (Eb) was measured in accordance with JIS K6251-1993.
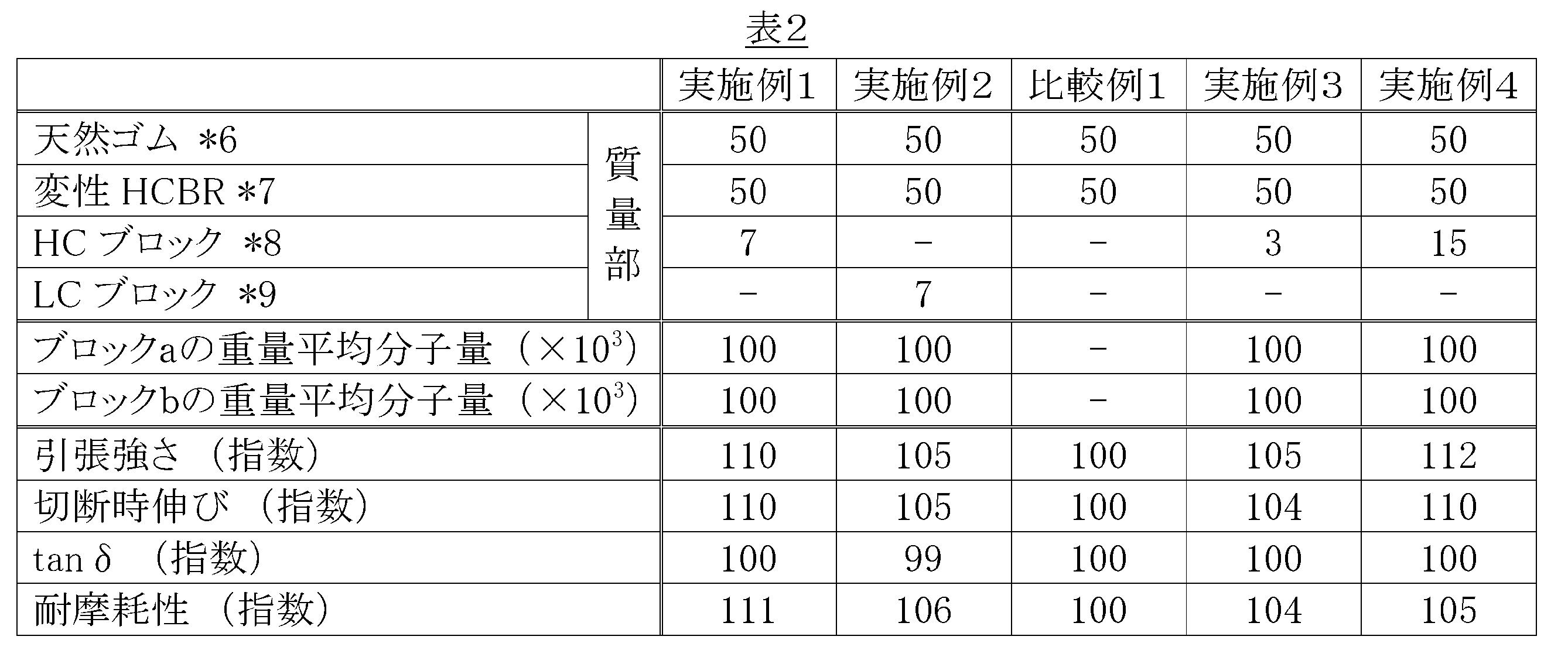
- Table 2 the elongation at break in Comparative Example 1 was set to 100.
- Table 3 the elongation at break in Example 6 was measured. Elongation was taken as 100, and in Table 4, the elongation at break of Comparative Example 5 was taken as 100, and the results were shown as an index. The larger the index value, the better the fracture resistance.
- tan ⁇ The loss tangent (tan ⁇ ) was measured at a temperature of 50 ° C., a strain of 5%, and a frequency of 15 Hz using a viscoelasticity measuring apparatus [manufactured by Rheometrics Co., Ltd.]
- tan ⁇ of Example 6 was set to 100
- tan ⁇ of Comparative Example 5 was set to 100 and displayed as an index. It shows that it is excellent in low-loss property, so that an index value is small.
- the rubber compositions of Examples 1 to 4 containing at least one rubber component constituting the incompatible polymer blend as a modified polymer and further containing a block copolymer are the same incompatible polymer blend. It can be seen that the fracture resistance and wear resistance can be greatly improved as compared with the rubber composition of Comparative Example 1 in which no block copolymer is blended.
- Example 9 Comparative Example 7 and Comparative Example 8 in Table 4, it is obtained by blending at least one rubber component constituting the incompatible polymer blend with a modified polymer and further adding a block copolymer.
- the effect is an effect obtained only by using at least one rubber component constituting the incompatible polymer blend as a modified polymer (Comparative Example 8), and an effect obtained only by blending a block copolymer (Comparison). It can be seen that this is far superior to the sum of Example 2). Similar results are obtained from the comparison of Comparative Example 3, Example 10, Comparative Example 5 and Comparative Example 6, and the comparison of Comparative Example 4, Example 11, Comparative Example 7 and Comparative Example 8.
Landscapes
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Mechanical Engineering (AREA)
- Compositions Of Macromolecular Compounds (AREA)
- Tires In General (AREA)
Abstract
Description
ポリマー相Aを形成する単量体単位と同一の単量体単位からなるブロックaと、ポリマー相Bを形成する単量体単位と同一の単量体単位からなるブロックbとからなるブロックコポリマーを配合してなり、
前記ゴム成分の少なくとも一種が、少なくとも一つの官能基を有することを特徴とする。
R1 aZXb ・・・ (I)
[式中、R1は、それぞれ独立して炭素数1~20のアルキル基、炭素数3~20のシクロアルキル基、炭素数6~20のアリール基及び炭素数7~20のアラルキル基からなる群から選択され;Zは、スズ又はケイ素であり;Xは、それぞれ独立して塩素又は臭素であり;aは0~3で、bは1~4で、但し、a+b=4である]で表されるカップリング剤も好ましい。式(I)のカップリング剤で変性することで、変性合成ゴムの耐コールドフロー性を改良することができる。なお、式(I)のカップリング剤で変性して得られる変性合成ゴムは、少なくとも一種のスズ-炭素結合又はケイ素-炭素結合を有する。
触媒成分としてバーサチック酸ネオジム(0.09ミリモル)のシクロヘキサン溶液、メチルアルミノキサン(MAO:東ソーアクゾ社製PMAO)(1.8ミリモル)のトルエン溶液、水素化ジイソブチルアルミニウム(DIBAH:関東化学社製)(5.0ミリモル)及びジエチルアルミニウムクロリド(0.18ミリモル)のトルエン溶液と、1,3-ブタジエン(4.5ミリモル)とを50℃で30分間反応熟成させた触媒Aを得た。次に、窒素置換された5リットルのオートクレーブに、窒素雰囲気下、シクロヘキサン2.4kg及び1,3-ブタジエン300gを仕込み、得られた溶液中に更に触媒Aを加えて、80℃で60分間重合を行った。この際の1,3-ブタジエンの反応転化率は、ほぼ100%であった。その後、重合反応系に2,4-ジ-tert-ブチル-p-クレゾール1.5gを含むメタノール溶液を添加し、重合反応を停止させ、更に常法に従って乾燥して重合体A(未変性HCBR:重量平均分子量(Mw)=30万,シス-1,4結合量=97%)を得た。
窒素置換された5リットルのオートクレーブに、窒素雰囲気下、シクロヘキサン2.4kg及び1,3-ブタジエン300gを仕込み、得られた溶液中に更に上記触媒Aを加えて、80℃で60分間重合を行った。この際の1,3-ブタジエンの反応転化率は、ほぼ100%であった。次に、重合反応系に末端変性剤として4,4'-ビス(ジエチルアミノ)ベンゾフェノン(0.55ミリモル)を加え、更に30分間変性反応を行った。その後、重合反応系に2,4-ジ-tert-ブチル-p-クレゾール1.5gを含むメタノール溶液を添加し、重合反応を停止させ、更に常法に従って乾燥して重合体B(変性HCBR:重量平均分子量(Mw)=30万,シス-1,4結合量=97%)を得た。
窒素置換された5リットルのオートクレーブに、窒素雰囲気下、シクロヘキサン2.4kg及び1,3-ブタジエン300gを仕込み、得られた溶液中に更に上記触媒Aを加えて、80℃で60分間重合を行った。この際の1,3-ブタジエンの反応転化率は、ほぼ100%であった。次に、重合反応系に末端変性剤として1-ブチルアジリジン(0.55ミリモル)を加え、更に30分間変性反応を行った。その後、重合反応系に2,4-ジ-tert-ブチル-p-クレゾール1.5gを含むメタノール溶液を添加し、重合反応を停止させ、更に常法に従って乾燥して重合体C(変性HCBR:重量平均分子量(Mw)=30万,シス-1,4結合量=96%)を得た。
乾燥し、窒素置換した内容積約900mlの耐圧ガラス容器に、シクロヘキサン283g、1,3-ブタジエン100g、2,2-ジテトラヒドロフリルプロパン0.015mmolをシクロヘキサン溶液として注入し、更にn-ブチルリチウム(n-BuLi)0.50mmolを加えた後、攪拌装置を備えた50℃の温水浴中で4.5時間重合反応を行った。この際の1,3-ブタジエンの反応転化率は、ほぼ100%であった。その後、重合反応系に2,6-ジ-t-ブチル-p-クレゾール(BHT)のイソプロパノール溶液(BHT濃度:5質量%)0.5mLを添加し、重合反応を停止させ、更に常法に従って乾燥して重合体D(未変性LCBR:重量平均分子量(Mw)=30万,シス-1,4結合量=32%)を得た。
乾燥し、窒素置換した内容積約900mlの耐圧ガラス容器に、シクロヘキサン283g、1,3-ブタジエン100g、2,2-ジテトラヒドロフリルプロパン0.015mmolをシクロヘキサン溶液として注入し、更にn-ブチルリチウム(n-BuLi)0.50mmolを加えた後、攪拌装置を備えた50℃の温水浴中で4.5時間重合反応を行った。この際の1,3-ブタジエンの反応転化率は、ほぼ100%であった。次に、重合反応系に変性剤として四塩化スズ(0.50mmol)のシクロヘキサン溶液を加え、攪拌しながら更に50℃で30分間変性反応を行った。その後、重合反応系に2,6-ジ-t-ブチル-p-クレゾール(BHT)のイソプロパノール溶液(BHT濃度:5質量%)0.5mLを添加し、重合反応を停止させ、更に常法に従って乾燥して重合体E(変性LCBR:重量平均分子量(Mw)=30万,シス-1,4結合量=31%)を得た。
窒素置換された5リットルのオートクレーブに、窒素雰囲気下、シクロヘキサン2.4kg及び1,3-ブタジエン(ブロックa成分)100gを仕込み、得られた溶液中に更に上記触媒Aを加えて、80℃で60分間重合を行った。この際の1,3-ブタジエンの反応転化率は、ほぼ100%であった。次に、重合反応系にイソプレン(ブロックb成分)100gを含むシクロへキサン溶液を添加し、更に60分間重合を行った。その後、重合反応系に2,4-ジ-tert-ブチル-p-クレゾール1.5gを含むメタノール溶液を添加し、重合反応を停止させ、更に常法に従って乾燥して重合体F(HCブロック:ブロックaの重量平均分子量(Mw)=10万,ブロックbの重量平均分子量(Mw)=10万,シス-1,4結合量=96%)を得た。なお、1,3-ブタジエン及びイソプレンの使用量を変えることで、ブロックa及びブロックbの重量平均分子量が異なるHCブロックを合成した(表3参照)。
乾燥し、窒素置換した内容積約900mlの耐圧ガラス容器に、シクロヘキサン283g、1,3-ブタジエン(ブロックa成分)35g、2,2-ジテトラヒドロフリルプロパン0.015mmolをシクロヘキサン溶液として注入し、更にn-ブチルリチウム(n-BuLi)0.50mmolを加えた後、攪拌装置を備えた50℃の温水浴中で1.5時間重合反応を行った。この際の1,3-ブタジエンの反応転化率は、ほぼ100%であった。次に、重合反応系にイソプレン(ブロックb成分)35gを含むシクロへキサン溶液を添加し、更に1.5時間重合を行った。その後、重合反応系に2,6-ジ-t-ブチル-p-クレゾール(BHT)のイソプロパノール溶液(BHT濃度:5質量%)0.5mLを添加し、重合反応を停止させ、更に常法に従って乾燥して重合体G(LCブロック:ブロックaの重量平均分子量(Mw)=100×103,ブロックbの重量平均分子量(Mw)=100×103,シス-1,4結合量=30%)を得た。
ゲルパーミエーションクロマトグラフィー[GPC:東ソー製HLC-8020、カラム:東ソー製GMH-XL(2本直列)、検出器:示差屈折率計(RI)]で単分散ポリスチレンを基準として、各重合体の変性停止なしの状態でのポリスチレン換算重量平均分子量(Mw)を求めた。なお、ブロックコポリマーについては、ブロックコポリマーの各ブロックの重量平均分子量(Mw)を求めた。
重合体のミクロ構造を赤外法(モレロ法)で求めた。
次に、表1に示す配合処方のゴム組成物を調製し、更に、該ゴム組成物を160℃で15分間加硫して加硫ゴムに対し、引張強さ(Tb)、切断時伸び(Eb)、tanδ及び耐摩耗性を測定・評価した。結果を表2~4に示す。
JIS K6251-1993に準拠して、引張強さ(Tb)を測定し、表2においては比較例1の引張強さを100とし、表3においては実施例6の引張強さを100とし、表4においては比較例5の引張強さを100として指数表示した。指数値が大きい程、耐破壊性が良好であることを示す。
JIS K6251-1993に準拠して、切断時伸び(Eb)を測定し、表2においては比較例1の切断時伸びを100とし、表3においては実施例6の切断時伸びを100とし、表4においては比較例5の切断時伸びを100として指数表示した。指数値が大きい程、耐破壊性が良好であることを示す。
粘弾性測定装置[レオメトリックス社製]を用い、温度50℃、歪み5%、周波数15Hzで損失正接(tanδ)を測定し、表2においては比較例1のtanδを100とし、表3においては実施例6のtanδを100とし、表4においては比較例5のtanδを100として指数表示した。指数値が小さい程、低ロス性に優れることを示す。
ランボーン型摩耗試験機を用い、室温におけるスリップ率60%での摩耗量を測定し、表2においては比較例1の摩耗量の逆数を100とし、表3においては実施例6の摩耗量の逆数を100とし、表4においては比較例5の摩耗量の逆数を100として指数表示した。指数値が大きい程、摩耗量が少なく、耐摩耗性に優れることを示す。
*2 上記の方法で調製したポリブタジエンゴム,使用したポリブタジエンゴムの種類を表2~4に示す.なお,いずれのポリブタジエンゴムも天然ゴムと非相溶性を示し,非相溶ポリマーブレンドを形成していた.
*3 上記の方法で調製したブロックコポリマー,使用したブロックコポリマーの種類及びゴム成分の合計100質量部に対する配合量を表2~4に示す.
*4 N-(1,3-ジメチルブチル)-N'-フェニル-p-フェニレンジアミン.
*5 N-シクロヘキシル-2-ベンゾチアゾリルスルフェンアミド.
*7 上記製造例2に従い製造された変性HCBR.
*8 上記製造例6に従い製造されたHCブロック.
*9 上記製造例7に従い製造されたLCブロック.
*11 上記製造例2に従い製造された変性HCBR.
*12 上記製造例6に従い製造されたHCブロック.
*14 上記製造例1に従い製造された未変性HCBR.
*15 上記製造例2に従い製造された変性HCBR.
*16 上記製造例3に従い製造された変性HCBR.
*17 上記製造例4に従い製造された未変性LCBR.
*18 上記製造例5に従い製造された変性LCBR.
*19 上記製造例6に従い製造されたHCブロック.
*20 上記製造例7に従い製造されたLCブロック.
Claims (8)
- 少なくとも二種類のゴム成分からなり、ポリマー相A及びポリマー相Bを形成する非相溶ポリマーブレンドに対し、
ポリマー相Aを形成する単量体単位と同一の単量体単位からなるブロックaと、ポリマー相Bを形成する単量体単位と同一の単量体単位からなるブロックbとからなるブロックコポリマーを配合してなり、
前記ゴム成分の少なくとも一種が、少なくとも一つの官能基を有することを特徴とするゴム組成物。 - 前記ポリマー相A及びポリマー相Bの内の一方が天然ゴム及び/又はポリイソプレンゴムを含み、他方のポリマー相がポリイソプレンゴム以外の合成ゴムを含むことを特徴とする請求項1に記載のゴム組成物。
- ゴム成分の重量平均分子量が20万以上であり、ブロックコポリマーを形成するブロックa及びブロックbの重量平均分子量がいずれも5万~50万であることを特徴する請求項1に記載のゴム組成物。
- 前記ポリイソプレンゴム以外の合成ゴムがポリブタジエンゴムであることを特徴とする請求項2に記載のゴム組成物。
- 前記ブロックコポリマーの配合量が、前記ゴム成分の合計100質量部に対し0.3~20質量部であることを特徴とする請求項1に記載のゴム組成物。
- 前記ゴム成分の合計100質量部に対し、更に充填剤を10~100質量部含有することを特徴とする請求項1に記載のゴム組成物。
- 請求項1~6のいずれかに記載のゴム組成物をタイヤ部材のいずれかに用いたことを特徴とするタイヤ。
- 前記タイヤ部材が、トレッド部、サイドウォール部、サイド補強ゴム、ビード部又はゴムチェーファーであることを特徴とする請求項7に記載のタイヤ。
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US12/996,964 US8222337B2 (en) | 2008-06-11 | 2009-06-11 | Rubber composition and tire using the same |
| JP2010516886A JP5548615B2 (ja) | 2008-06-11 | 2009-06-11 | ゴム組成物及びそれを用いたタイヤ |
| EP09762533A EP2289992B1 (en) | 2008-06-11 | 2009-06-11 | Rubber composition and tire using the same |
| CN2009801293223A CN102105525B (zh) | 2008-06-11 | 2009-06-11 | 橡胶组合物及使用该橡胶组合物的轮胎 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2008153110 | 2008-06-11 | ||
| JP2008-153110 | 2008-06-11 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2009151103A1 true WO2009151103A1 (ja) | 2009-12-17 |
Family
ID=41416807
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2009/060712 Ceased WO2009151103A1 (ja) | 2008-06-11 | 2009-06-11 | ゴム組成物及びそれを用いたタイヤ |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US8222337B2 (ja) |
| EP (1) | EP2289992B1 (ja) |
| JP (1) | JP5548615B2 (ja) |
| KR (1) | KR20110019418A (ja) |
| CN (1) | CN102105525B (ja) |
| WO (1) | WO2009151103A1 (ja) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2019535887A (ja) * | 2016-11-09 | 2019-12-12 | 株式会社ブリヂストン | ポリブタジエンとポリイソプレンとの高シス−1,4ブロックコポリマー |
Families Citing this family (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9546237B2 (en) | 2012-06-06 | 2017-01-17 | Bridgestone Corporation | Stabilization of polymers that contain a hydrolyzable functionality |
| JP2017501289A (ja) | 2013-12-12 | 2017-01-12 | ファイアストン ポリマーズ エルエルシー | 官能化ポリマーを含むポリマー組成物の製造方法 |
| KR101770482B1 (ko) | 2014-12-17 | 2017-08-22 | 주식회사 엘지화학 | 변성 공액디엔계 중합체 및 이를 포함하는 고무 조성물 |
| KR101770883B1 (ko) | 2014-12-17 | 2017-08-23 | 주식회사 엘지화학 | 변성 공액디엔계 중합체의 제조방법 및 이를 이용하여 제조한 변성 공액디엔계 중합체 |
| US20190256690A1 (en) * | 2016-06-08 | 2019-08-22 | Bridgestone Corporation | Rubber Composition |
| US20220372197A1 (en) * | 2019-09-05 | 2022-11-24 | Asahi Kasei Kabushiki Kaisha | Conjugated Diene-Based Polymer, Production Method for Conjugated Diene-Based Polymer, Conjugated Diene-Based Polymer Composition and Rubber Composition |
Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH01129660A (ja) | 1987-11-16 | 1989-05-22 | Canon Inc | 画像合成装置 |
| JPH029660A (ja) | 1988-06-29 | 1990-01-12 | Brother Ind Ltd | 手動印字装置 |
| JPH08283465A (ja) * | 1995-04-14 | 1996-10-29 | Yokohama Rubber Co Ltd:The | タイヤトレッド用ゴム組成物 |
| JP2002012702A (ja) * | 2000-06-30 | 2002-01-15 | Nippon Zeon Co Ltd | ゴム組成物 |
| JP2002256108A (ja) * | 2001-02-27 | 2002-09-11 | Kuraray Co Ltd | ゴム組成物 |
| JP2005133017A (ja) * | 2003-10-31 | 2005-05-26 | Yokohama Rubber Co Ltd:The | サイドウォール用ゴム組成物 |
| JP2007191611A (ja) * | 2006-01-20 | 2007-08-02 | Bridgestone Corp | 変性ポリブタジエンゴム配合ゴム組成物及びタイヤ |
Family Cites Families (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4098401A (en) * | 1976-08-18 | 1978-07-04 | Weyerhaeuser Company | Furniture container |
| JPS62112640A (ja) * | 1985-11-09 | 1987-05-23 | Toyo Tire & Rubber Co Ltd | タイヤトレツド用ゴム組成物 |
| US5679744A (en) * | 1994-11-11 | 1997-10-21 | The Yokohama Rubber Co., Ltd. | Rubber composition |
| JP3601110B2 (ja) * | 1995-04-13 | 2004-12-15 | ユニマテック株式会社 | アクリル系エラストマー加硫成形品 |
| JP4067151B2 (ja) | 1996-07-19 | 2008-03-26 | 横浜ゴム株式会社 | タイヤ用ゴム組成物 |
| US6084022A (en) * | 1997-09-22 | 2000-07-04 | The Goodyear Tire & Rubber Company | Tire tread compositions containing asymmetrically tin-coupled polybutadiene rubber |
| JP4194825B2 (ja) * | 2002-11-18 | 2008-12-10 | 横浜ゴム株式会社 | ゴム組成物 |
| WO2008004675A1 (en) | 2006-07-06 | 2008-01-10 | Bridgestone Corporation | Rubber composition and pneumatic tire using the same |
-
2009
- 2009-06-11 EP EP09762533A patent/EP2289992B1/en not_active Not-in-force
- 2009-06-11 KR KR1020117000534A patent/KR20110019418A/ko not_active Ceased
- 2009-06-11 US US12/996,964 patent/US8222337B2/en not_active Expired - Fee Related
- 2009-06-11 WO PCT/JP2009/060712 patent/WO2009151103A1/ja not_active Ceased
- 2009-06-11 JP JP2010516886A patent/JP5548615B2/ja not_active Expired - Fee Related
- 2009-06-11 CN CN2009801293223A patent/CN102105525B/zh not_active Expired - Fee Related
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH01129660A (ja) | 1987-11-16 | 1989-05-22 | Canon Inc | 画像合成装置 |
| JPH029660A (ja) | 1988-06-29 | 1990-01-12 | Brother Ind Ltd | 手動印字装置 |
| JPH08283465A (ja) * | 1995-04-14 | 1996-10-29 | Yokohama Rubber Co Ltd:The | タイヤトレッド用ゴム組成物 |
| JP2002012702A (ja) * | 2000-06-30 | 2002-01-15 | Nippon Zeon Co Ltd | ゴム組成物 |
| JP2002256108A (ja) * | 2001-02-27 | 2002-09-11 | Kuraray Co Ltd | ゴム組成物 |
| JP2005133017A (ja) * | 2003-10-31 | 2005-05-26 | Yokohama Rubber Co Ltd:The | サイドウォール用ゴム組成物 |
| JP2007191611A (ja) * | 2006-01-20 | 2007-08-02 | Bridgestone Corp | 変性ポリブタジエンゴム配合ゴム組成物及びタイヤ |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP2289992A4 * |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2019535887A (ja) * | 2016-11-09 | 2019-12-12 | 株式会社ブリヂストン | ポリブタジエンとポリイソプレンとの高シス−1,4ブロックコポリマー |
| US11230622B2 (en) | 2016-11-09 | 2022-01-25 | Bridgestone Corporation | High cis-1,4 block copolymers of polybutadiene and polyisoprene |
| US11884767B2 (en) | 2016-11-09 | 2024-01-30 | Bridgestone Corporation | High cis-1,4 block copolymers of polybutadiene and polyisoprene |
Also Published As
| Publication number | Publication date |
|---|---|
| CN102105525A (zh) | 2011-06-22 |
| EP2289992A1 (en) | 2011-03-02 |
| JP5548615B2 (ja) | 2014-07-16 |
| EP2289992B1 (en) | 2012-12-19 |
| EP2289992A4 (en) | 2011-11-02 |
| KR20110019418A (ko) | 2011-02-25 |
| JPWO2009151103A1 (ja) | 2011-11-17 |
| CN102105525B (zh) | 2013-03-27 |
| US8222337B2 (en) | 2012-07-17 |
| US20110092633A1 (en) | 2011-04-21 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US5959039A (en) | Rubber composition having both high and low molecular weight polymer components, for use in tires | |
| JP5548615B2 (ja) | ゴム組成物及びそれを用いたタイヤ | |
| JPH07292161A (ja) | タイヤトレッド用ゴム組成物 | |
| WO2019044889A1 (ja) | タイヤ用ゴム組成物 | |
| JPWO2007081026A1 (ja) | ゴム組成物及びそれを用いた空気入りタイヤ | |
| TWI829906B (zh) | 氫化共軛二烯系聚合物、聚合物組成物、交聯體及輪胎 | |
| JPWO2008047582A1 (ja) | サイドゴム用ゴム組成物及びそれを用いた重荷重用空気入りラジアルタイヤ | |
| JP7657722B2 (ja) | 重合体組成物、架橋重合体、及びタイヤ | |
| JP5193623B2 (ja) | ゴム組成物及びそれを用いた空気入りタイヤ | |
| JP2019094390A (ja) | 変性共役ジエン系重合体の製造方法、重合体組成物、架橋体及びタイヤ | |
| CN103189443B (zh) | 橡胶组合物及其制造方法和轮胎 | |
| JPH1053671A (ja) | ゴム組成物 | |
| CN104937037B (zh) | 聚合物组合物、交联聚合物、轮胎和聚合物 | |
| JP4846250B2 (ja) | ゴム組成物及びそれを用いた空気入りタイヤ | |
| JP2004107384A (ja) | 共役ジエン系ゴム、ゴム組成物、及び共役ジエン系ゴムの製造方法 | |
| JP5464781B2 (ja) | ゴム組成物及びそれを用いたタイヤ | |
| JP2011148956A (ja) | ゴム組成物及び該ゴム組成物を用いたトレッド及びタイヤ | |
| JP5121300B2 (ja) | ゴム組成物及びそれを用いた空気入りタイヤ | |
| JP4134732B2 (ja) | ゴム組成物 | |
| JP2003221471A (ja) | ゴム組成物およびその製造方法 | |
| CN119907815A (zh) | 氢化共轭二烯系聚合物以及氢化共轭二烯系聚合物的制造方法 | |
| JP4770161B2 (ja) | 空気入りタイヤ | |
| JP7458373B2 (ja) | 重合体組成物、架橋重合体、及びタイヤ | |
| JP2026003265A (ja) | 共役ジエン系重合体、重合体組成物、架橋体及びタイヤ | |
| WO2024058265A1 (ja) | 共役ジエン系重合体、重合体組成物、架橋物、及びタイヤ |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200980129322.3 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 09762533 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2010516886 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 12996964 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2009762533 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 20117000534 Country of ref document: KR Kind code of ref document: A |